WO1991015478A1 - Crop-selective herbicidal sulfonamides - Google Patents
Crop-selective herbicidal sulfonamides Download PDFInfo
- Publication number
- WO1991015478A1 WO1991015478A1 PCT/US1991/001075 US9101075W WO9115478A1 WO 1991015478 A1 WO1991015478 A1 WO 1991015478A1 US 9101075 W US9101075 W US 9101075W WO 9115478 A1 WO9115478 A1 WO 9115478A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- amino
- methyl
- compound
- sulfonyl
- benzoate
- Prior art date
Links
- 0 C*c(c(*)ccc1)c1C#* Chemical compound C*c(c(*)ccc1)c1C#* 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D521/00—Heterocyclic compounds containing unspecified hetero rings
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N47/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid
- A01N47/08—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom not being member of a ring and having no bond to a carbon or hydrogen atom, e.g. derivatives of carbonic acid the carbon atom having one or more single bonds to nitrogen atoms
- A01N47/28—Ureas or thioureas containing the groups >N—CO—N< or >N—CS—N<
- A01N47/36—Ureas or thioureas containing the groups >N—CO—N< or >N—CS—N< containing the group >N—CO—N< directly attached to at least one heterocyclic ring; Thio analogues thereof
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C205/00—Compounds containing nitro groups bound to a carbon skeleton
- C07C205/49—Compounds containing nitro groups bound to a carbon skeleton the carbon skeleton being further substituted by carboxyl groups
- C07C205/57—Compounds containing nitro groups bound to a carbon skeleton the carbon skeleton being further substituted by carboxyl groups having nitro groups and carboxyl groups bound to carbon atoms of six-membered aromatic rings of the carbon skeleton
- C07C205/58—Compounds containing nitro groups bound to a carbon skeleton the carbon skeleton being further substituted by carboxyl groups having nitro groups and carboxyl groups bound to carbon atoms of six-membered aromatic rings of the carbon skeleton the carbon skeleton being further substituted by halogen atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C205/00—Compounds containing nitro groups bound to a carbon skeleton
- C07C205/49—Compounds containing nitro groups bound to a carbon skeleton the carbon skeleton being further substituted by carboxyl groups
- C07C205/57—Compounds containing nitro groups bound to a carbon skeleton the carbon skeleton being further substituted by carboxyl groups having nitro groups and carboxyl groups bound to carbon atoms of six-membered aromatic rings of the carbon skeleton
- C07C205/59—Compounds containing nitro groups bound to a carbon skeleton the carbon skeleton being further substituted by carboxyl groups having nitro groups and carboxyl groups bound to carbon atoms of six-membered aromatic rings of the carbon skeleton the carbon skeleton being further substituted by singly-bound oxygen atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C255/00—Carboxylic acid nitriles
- C07C255/01—Carboxylic acid nitriles having cyano groups bound to acyclic carbon atoms
- C07C255/32—Carboxylic acid nitriles having cyano groups bound to acyclic carbon atoms having cyano groups bound to acyclic carbon atoms of a carbon skeleton containing at least one six-membered aromatic ring
- C07C255/41—Carboxylic acid nitriles having cyano groups bound to acyclic carbon atoms having cyano groups bound to acyclic carbon atoms of a carbon skeleton containing at least one six-membered aromatic ring the carbon skeleton being further substituted by carboxyl groups, other than cyano groups
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C309/00—Sulfonic acids; Halides, esters, or anhydrides thereof
- C07C309/78—Halides of sulfonic acids
- C07C309/86—Halides of sulfonic acids having halosulfonyl groups bound to carbon atoms of six-membered aromatic rings of a carbon skeleton
- C07C309/89—Halides of sulfonic acids having halosulfonyl groups bound to carbon atoms of six-membered aromatic rings of a carbon skeleton containing carboxyl groups bound to the carbon skeleton
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C323/00—Thiols, sulfides, hydropolysulfides or polysulfides substituted by halogen, oxygen or nitrogen atoms, or by sulfur atoms not being part of thio groups
- C07C323/50—Thiols, sulfides, hydropolysulfides or polysulfides substituted by halogen, oxygen or nitrogen atoms, or by sulfur atoms not being part of thio groups containing thio groups and carboxyl groups bound to the same carbon skeleton
- C07C323/62—Thiols, sulfides, hydropolysulfides or polysulfides substituted by halogen, oxygen or nitrogen atoms, or by sulfur atoms not being part of thio groups containing thio groups and carboxyl groups bound to the same carbon skeleton having the sulfur atom of at least one of the thio groups bound to a carbon atom of a six-membered aromatic ring of the carbon skeleton
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F7/00—Compounds containing elements of Groups 4 or 14 of the Periodic System
- C07F7/02—Silicon compounds
- C07F7/08—Compounds having one or more C—Si linkages
- C07F7/10—Compounds having one or more C—Si linkages containing nitrogen having a Si-N linkage
Definitions
- This invention relates to novel sulfonamides and their use as crop selective herbicides.
- EP-A-120,814 discloses herbicidal sulfonylureas of the formula
- A is C 1 -C 6 haloalkyl
- R 1 is H, halogen, NO 2 , CN, C 1 -C 4 alkyl,
- C 1 -C 4 haloalkyl C 1 -C 4 alkoxy, C 1 -C 4 alkylthio, C 1 -C 4 alkylsulfinyl, C 1 -C 4 alkylsulfonyl, COR 6 , NR 7 R 8 , CONR 9 R 10 or
- R 6 is C 1 -C 4 alkoxy, C 1 -C 4 haloalkoxy,
- herbicides because of world-wide food shortages. In addition, herbicides which are selective to important crops such as corn are particularly necessary.
- this invention comprises novel compounds of Formula I, agriculturally suitable compositions containing them, and their method-of-use as preemergence and/or postemergence herbicides or plant growth regulants:
- A is OR 1 or N(CH 3 ) 2 ;
- R 2 is CH 2 F, CHF 2 , CHFCH 3 or CH 2 CN;
- X is CH 3 or OCH 3 ;
- Y is H, Cl, CH 3 , C 2 H 5 , OCH 3 or OCF 2 H;
- alkyl denotes straight chain or branched alkyl, e.g. methyl, ethyl, n-propyl or isopropyl.
- the total number of carbon atoms in a substituent group is indicated by the C i -C j prefix where i and j are numbers 1 to 3.
- C 1 -C 3 alkyl would designate methyl through propyl (both n-propyl and isopropyl).
- A is OR 1 ;
- R 1 is CH 3 , CH 2 CH 3 or CH(CH 3 ) 2 .
- wheat and barley are:
- A is OR 1 , R 1 is CH 2 CH 3 , R 2 is CH 2 F, X is OCH 3 , Y is Cl).
- This invention also comprises novel compounds, such as the sulfonyl benzoates of Formula II, useful as intermediates for preparation of the compounds of Formula I:
- A is OR 1 or N(CH 3 ) 2 ;
- R 2 is CH 2 F, CHF 2 , CHFCH 3 or CH 2 CN;
- Z 1 is Cl or NHSiR 3 R 4 R 5 ;
- R 3 is C 1 -C 4 alkyl
- R 4 is C 1 -C 4 alkyl
- R 5 is C 1 -C 4 alkyl.
- R 3 and R 4 are CH 3 and R 5 is C(CH 3 ) 3 .
- the compounds of Formula (1) which corresponds to Formula I, can be prepared by one or more of the methods described below.
- the proper choice of reaction sequences for a given compound will be known to one skilled in the art.
- Equation 1 many of the compounds of Formula (1) are prepared by reacting a silyl sulfonamide of Formula (2) with a pyrimidine
- R 3 , R 4 , and R 5 are
- reaction is carried out at 0°C to 50°C in a solvent such as acetonitrile, dioxane, or
- tetrahydrofuran in the presence of a fluoride ion source such as cesium fluoride, or tetrabutylammonium fluoride for 0.1 to 2 hours.
- a fluoride ion source such as cesium fluoride, or tetrabutylammonium fluoride for 0.1 to 2 hours.
- a catalytic amount of base such as 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU), increases the reaction rate.
- DBU 1,8-diazabicyclo[5.4.0]undec-7-ene
- Equation 2 Equation 2
- Sulfonyl chlorides of Formula (5) may be reacted with cyanate anion in the presence of
- the reaction is carried out by mixing one equivalent of sulfonyl chloride (5), pyrimidine amine (6), a metal cyanate, such as potassium cyanate, and a catalytic amount of an amine base, such as Aliquat ® 336 (Tricaprylymethylammonium chloride), in a solvent such as acetonitrile for 0.2 to 10 days.
- a tetraalkylammonium cyanate such as tetraethylammonium cyanate, to effect this reaction by the method described in U.S. Patent 4,604,131 herein incorporated by reference.
- Silyl sulfonamides of Formula (2) are prepared by reacting sulfonyl chlorides of Formula (5) with a trialkylsilyl amine of Formula (7), such as
- Equation 4 t-butyldimethylsilyl amine
- reaction is carried out at 0°C to 30°C in a solvent, such as dichloromethane, in the presence of
- the sulfonyl chlorides of Formula (5) and the silyl sulfonamides of Formula (2) correspond to the intermediates of Formula II.
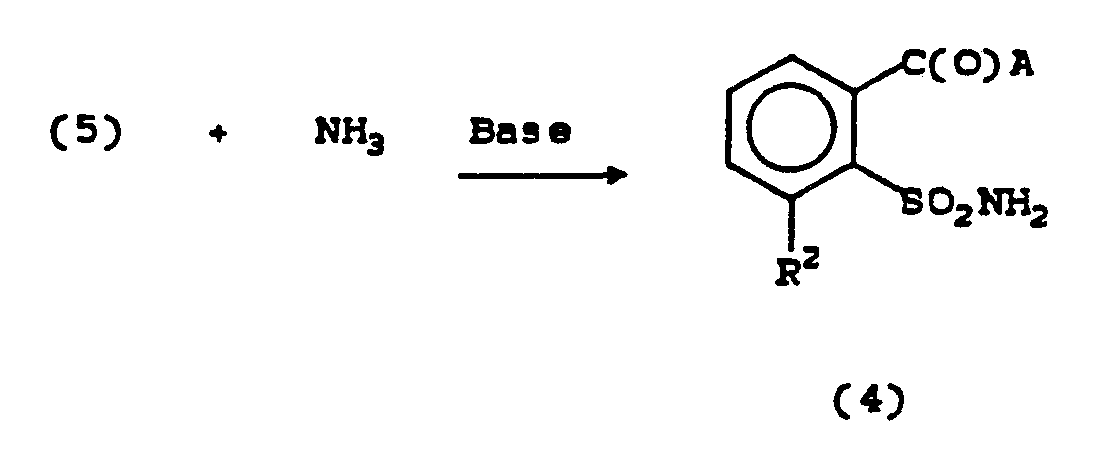
- Sulfonyl chlorides of Formula (5) can be reacted with no more than 2 equivalents of ammonia or, alternatively, 1 equivalent of ammonia and 1 equivalent of a base to give sulfonamides of Formula
- Equation 5 Equation 5
- Sulfonamides of Formula (4) tend to be unstable and can be difficult to isolate.
- Sulfonyl chlorides of Formula (5) are prepared by the methods shown in Equations 6 and 7.
- Sulfur-containing compounds of Formula (8) are oxidized with chlorine as shown in Equation 6.
- R 6 is
- Equation 6 The reaction of Equation 6 is carried out by contacting compounds of Formula (8) in a solvent, such as acetic acid or propionic acid, with at least 3.0 equivalents of chlorine in the presence of at least 2.5 equivalents of water for 0.2 to 5 hours at -20°C to 30°C.
- a solvent such as acetic acid or propionic acid
- chlorides of Formula (5) may be prepared from the corresponding anilines of Formula (9) by a Meerwein reaction.
- aniline is diazotized and then reacted with sulfur dioxide and cupric chloride analogous to the teachings of Yale and Sowinski, J. Org. Chem. 25 , 1824 (1960).
- the hydrochloride salts of anilines of Formula (9) can be diazotized in an organic solvent with an alkyl nitrite and reacted with sulfur dioxide to give sulfonyl chlorides (5) analogous to the teachings of M. Doyle, J. Org. Chem. 42, 2426,2431 (1977).
- the pyrimidine carbamates of Formula (3) and the pyrimidine amines of Formula (6) are prepared by the methods described and referenced in EP-A-72,347, EP-A-164,269, EP-A-173,498, U.S. Patent 4,540,782, and U.S. Patent 4,666,506, herein incorporated by reference.
- Agriculturally suitable salts of compounds of Formula I are also useful herbicides and can be prepared in a number of ways known to the art. For example, metal salts can be made by contacting
- an alkali or alkaline earth metal salt having a sufficiently basic anion e.g., hydroxide, alkoxide, carbonate or hydroxide.
- Quaternary amine salts can be made by similar techniques.
- Salts of compounds of Formula I can also be prepared by exchange of one cation for another.
- Cationic exchange can be effected by direct contact of an aqueous solution of a salt of a compound of Formula I (e.g., alkali or quaternary amine salt) with a solution containing the cation to be
- Exchange may also be effected by passing an aqueous solution of a salt of a compound of Formula I (e.g., an alkali metal or quaternary amine salt) through a column packed with a cation exchange resin containing the cation to be exchanged for that of the original salt and the desired product is eluted from the column.
- a salt of a compound of Formula I e.g., an alkali metal or quaternary amine salt
- a cation exchange resin containing the cation to be exchanged for that of the original salt and the desired product is eluted from the column.
- Acid addition salts useful in this invention, can be obtained by reacting a compound of Formula I with a suitable acid, e.g., p-toluenesulfonic acid, trichloroacetic acid or the like.
- a suitable acid e.g., p-toluenesulfonic acid, trichloroacetic acid or the like.
- the crude thioether was stirred in a mixture of dichloromethane (200 mL) and 6N hydrochloric acid (25 mL) at 0°C, as 5% sodium hypochlorite (180 mL) was slowly added. The reaction mixture was then stirred at 0°C for 2 hours. The dichloromethane layer was washed with water, dried over magnesium sulfate, filtered, concentrated, and chromatographed on silica gel eluting with 25% ethyl acetate in hexanes to give 1.7 g of the title compound as a yellow solid.
- Methyl 3-Formyl-2-nitrobenzoate Methyl 3-(dibromomethyl)-2-nitrobenzoate (62.6 g), silver nitrate (109 g), 1,2-dimethoxyethane (500 mL), and water (400 mL) were heated at reflux
- chlorobutane gave 20.9 g of the title compound as an orange solid.
- the crude thioether was stirred in a mixture of dichloromethane (400 mL) and 6N hydrochloric acid (28 mL) at 0°C, as 5% sodium hypochlorite (204 mL) was slowly added. The reaction mixture was stirred at 0°C for 2.5 hours. The dichloromethane layer was washed with water, dried over magnesium sulfate, filtered, concentrated, and triturated with hexanes to give 5.17 g of the title compound as a yellow solid.
- Methyl 3-(Difluoromethyl)-2-[[[[(4-methoxy-2- pyrimidinyl)amino]carbonyl]amino]sulfonyl]benzoate A mixture of methyl 3-(difluoromethyl)-2- [[[(1,1-dimethylethyl)dimethylsilyl]amino]sulfonyl]- benzoate (0.22 g), O-phenyl-N-(4-methoxy- 2-pyrimidinyl)carbamate (0.15 g), and acetonitrile (2.0 mL) were stirred and cooled in an ice-acetone bath. A stock solution of 1 M tetrabutylammonium fluoride containing 8 mol % 1,8-diazabicyclo-
- Titanium tetrachloride (6.25 mL) was added to dry diethylether ( 250 mL) at -78°C and was sti rred under a nitrogen atmosphere.
- the resulting yellow suspension was allowed to warm to -50°C and was recooled to -78°C prior to adding 1.4 oolar methyllithium in diethylether (41.6 mL).
- the mixture was warmed to -30°C and the resulting purple mixture was cooled to -60°C and transferred by cannula into an adj acent f lask containing methyl 3-formyl- 2-nitrobenzoate (11.6 g) and dry tetrahydrofuran (250 mL).
- the resulting mixture was allowed to warm to room temperature and was stirred under a nitrogen atmosphere for about 18 hours.
- the reaction was poured onto a mixture of ice (100 mL) and 3N
- Useful formulations of the compounds of Formula I can be prepared in conventional ways. They include dusts, granules, pellets, solutions, suspensions, emulsions, wettable powders, emulsifiable
- Sprayable formulations can be extended in suitable media and used at spray volumes of from a few liters to several hundred liters per hectare. High strength compositions are primarily used as intermediates for further formulation.
- the formulations broadly, contain about 0.1% to 99% by weight of active ingredient(s) and at least one of
- Wettable Powders 20-90 0-74 1-10 Oil Suspensions, 3-50 40-95 0-15
- compositions Active ingredient plus at least one of a Surfactant or a Diluent equals 100 weight percent.
- Lower or higher levels of active ingredient can, of course, be present depending on the intended use and the physical properties of the compound. Higher ratios of surfactant to active ingredient are sometimes desirable, and are achieved by incorporation into the formulation or by tank mixing.
- Emulsifiers Annual MC Publishing Corp., Kidgewood, New Jersey, as well as Sisely and Wood, "Encyclopedia of Surface Active Agents", Chemical Publishing Co., Inc., New York, 1964, list surfactants and recommended uses. All formulations can contain minor amounts of additives to reduce foaming, caking, corrosion, microbiological growth, etc.
- compositions are well known. Solutions are prepared by simply mixing the ingredients. Fine solid compositions are made by blending and, usually, grinding as in a hammer or fluid energy mill. Suspensions are prepared by wet milling (see, for example, Littler, U.S. Patent
- Granules and pellets may be made by spraying the active material upon preformed granular carriers or by agglomeration techniques. See J. E. Browning, "Agglomeration", Chemical Engineering,
- the surfactant is sprayed upon the active ingredient in a blender and the mixture sifted through a U. S. S. No. 40 sieve (0.42 mm openings) prior to packaging.
- the concentrate may be formulated further for practical use.
- the ingredients are thoroughly blended.
- the liquid surfactant is added by spraying upon the solid ingredients in the blender. After grinding in a hammer mill to produce particles essentially all below 100 microns, the material is reblended and sifted through a U.S.S. No. 50 sieve (0.3 mm opening) and packaged.
- the ingredients are blended and ground together in a sand mill to produce particles essentially all under 5 microns in size.
- the ingredients are combined and ground together in a sand mill to produce particles essentially all below 3 microns.
- the product can be used directly, extended with oils, or emulsified in water.
- the ingredients are ground together in a sand mi l l unti l the solid particles have been reduced to under about 5 microns.
- the resulting thick suspension may be applied directly, but preferably after being extended with oils or emulsified in water.
- the ingredients are ground together in a ball or roller mill until the solid particles have been reduced to diameters under 10 microns.
- the ingredients are thoroughly blended, passed through an air mill, to produce an average particle size under 15 microns, reblended, and sifted through a U.S.S. No. 50 sieve (0.3 mm opening) before packaging.
- the ingredients are blended in a rotating mixer and water sprayed on to accomplish granulation. When most of the material has reached the desired range of 1.0 to 0.42 cm (U.S.S. #18 to 40 sieves), the granules are removed, dried, and screened. Oversized material is crushed to produce additional material in the desired range. These granules contain % active ingredient.
- Example J The ingredients are blended, coarsely hammermilled and the air milled to produce particles of active essentially all below 10 microns in diameter. The product is reblended before packaging.
- Example J The ingredients are blended, coarsely hammermilled and the air milled to produce particles of active essentially all below 10 microns in diameter. The product is reblended before packaging.
- the ingredients are blended, hammer-milled and then moistened with about 12% water.
- the mixture is extruded as cylinders about 3 mm diameter which are cut to produce pellets about 3 mm long. These may be used directly after drying, or the dried pellets may be crushed to pass a U.S.S. No. 20 sieve (0.84 mm openings).
- the granules held on a U.S.S. No. 40 sieve (0.42 mm openings) may be packaged for use and the fines recycled.
- Example L The ingredients are blended and then ground in a hammermill to produce particles with an average particle size less than 25 microns in diameter. The material is reblended and sifted through a U.S.S. No. 50 sieve (0.3 mm opening) before being packaged.
- Example L The ingredients are blended and then ground in a hammermill to produce particles with an average particle size less than 25 microns in diameter. The material is reblended and sifted through a U.S.S. No. 50 sieve (0.3 mm opening) before being packaged.
- Example L Example L
- the ingredients are blended and ground in a hammer mill to produce a high strength concentrate essentially all passing a U.S.S. No. 50 sieve (0.3 mm openings). This material may then be formulated in a variety of ways.
- the salt is added directly to the water with stirring to produce the solution, which may then be packaged for use.
- UTILITY is added directly to the water with stirring to produce the solution, which may then be packaged for use.
- important argonomic crops such as barley (Hordeum vulgare), corn (Zea mays), rice (Orysa sativa), and wheat (Triticum aestivum).
- Grass weeds controlled include, but are not limited to, barnyardgrass (Echinochloa crus-galli), black- grass (Alopecurus myosuroides), Bromus spp., foxtail
- weeds controlled include, but are not limited to, cocklebur (Xanthium pensylvanicum), jimsonweed (Datura stramonium), lambsquarters
- Rates of application for compounds of this invention are determined by a number of factors.
- the subject compounds should be applied at rates from 0.001 to 20 kg/ha with a preferred rate range of from 0.004 to 0.25 kg/ha.
- Compounds of this invention may be used alone or in combination with other commercial herbicides, insecticides, or fungicides.
- the following list exemplifies some of the herbicides suitable for use in mixtures.
- a combination of compounds from this invention with one or more of the following herbicides may be particularly useful for weed control.
- DPX-M6316 3-[[[[[(4-methoxy-6-methyl-1,3,5-triazin- 2-yl)amino]carbonyl]amino]sulfonyl]- 2-thiophenecarboxylic acid, methyl ester
- PPG-1013 5-[2-chloro-4-(trifluoromethyl)- phenoxy]-2-nitroacetophenone
- Herbicidal properties of the subject compounds were discovered in a series of greenhouse tests.
- Croperus rotundus tubers were planted and treated preemergence with test chemicals dissolved in a non-phytotoxic solvent. At the same time, these crop and weed species were also treated with postemergence applications of test chemicals. Plants ranged in height from two to eighteen cm (two to three leaf stage) for postemergence treatments. Treated plants and controls were maintained in a greenhouse for approximately sixteen days, after which all species were compared to controls and visually evaluated.
- Plant iresponse ratings are based on a scale of 0 to 10 where 0 is no effect and 10 is complete control.
- a dash (-) response means no test result.
- soybean (Glycine max), sugar beet (Beta vulgaris), velvetleaf (Abutilon theophrasti), wheat (Triticum aestiyum), wild buckwheat (Polygonum convolyulus), and wild oat (Ayena fatua) and purple nutsedge (Cyperus rotundus) tubers were planted and treated preemergence with test chemicals dissolved in a non-phytotoxic solvent. At the same time, these crop and weed species were also treated with
- Plants ranged in height from two to eighteen cm (one to four leaf stage) for postemergence treatments. Treated plants and controls were maintained in a greenhouse for twelve to sixteen days, after which all species were compared to controls and visually evaluated. Plant response ratings, summarized in Table B, are based on a scale of 0 to 10 where 0 is no effect and 10 is complete control. A dash (-) response means no test result.
- barnyardgrass Echinochloa crus-galli
- blackgrass Alopecurus myosuroides
- chickweed Stellaria media
- cocklebur Xanthium pensylyanicum
- corn Zea mays
- cotton Gossypium hirsutum
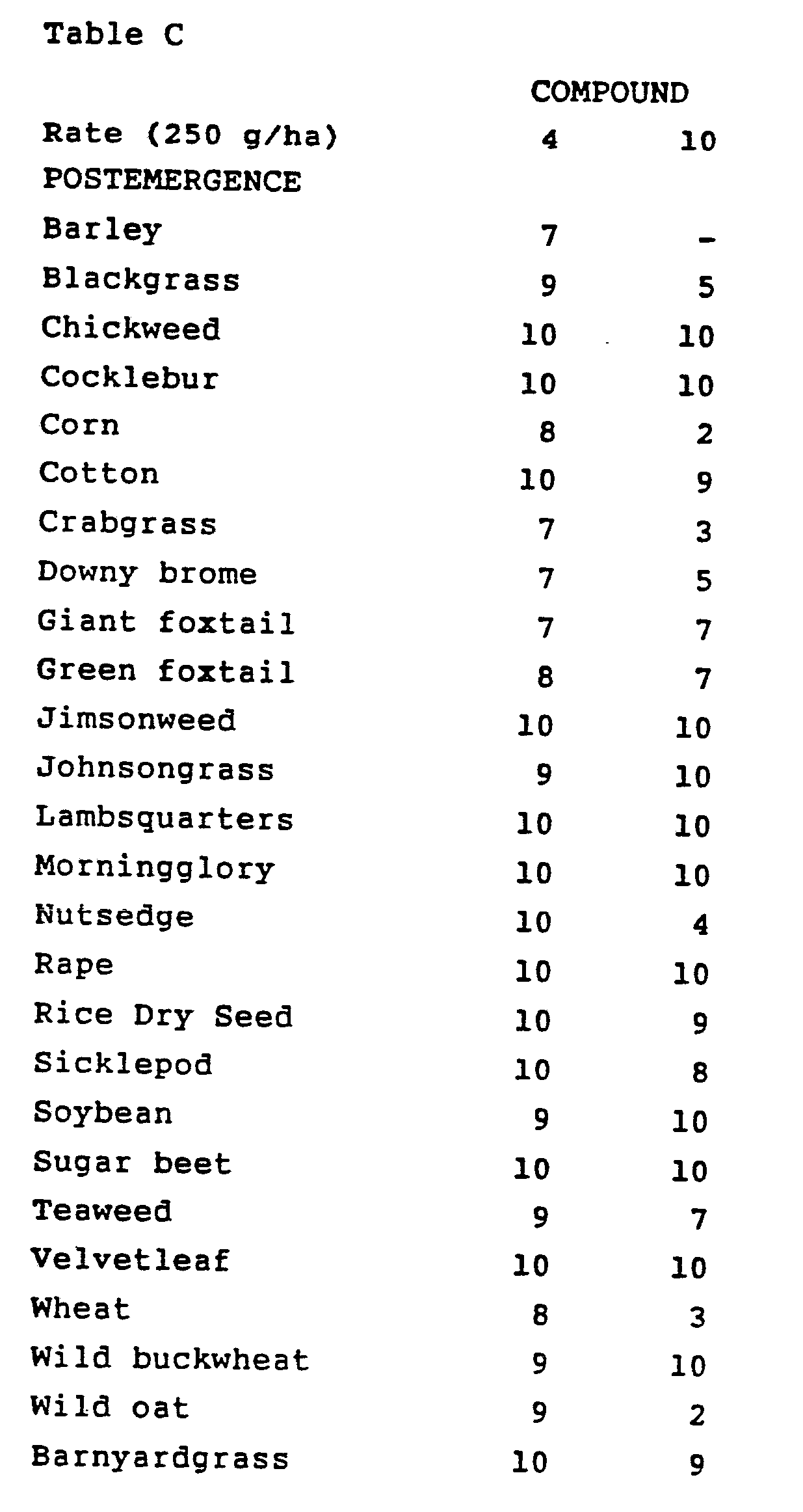
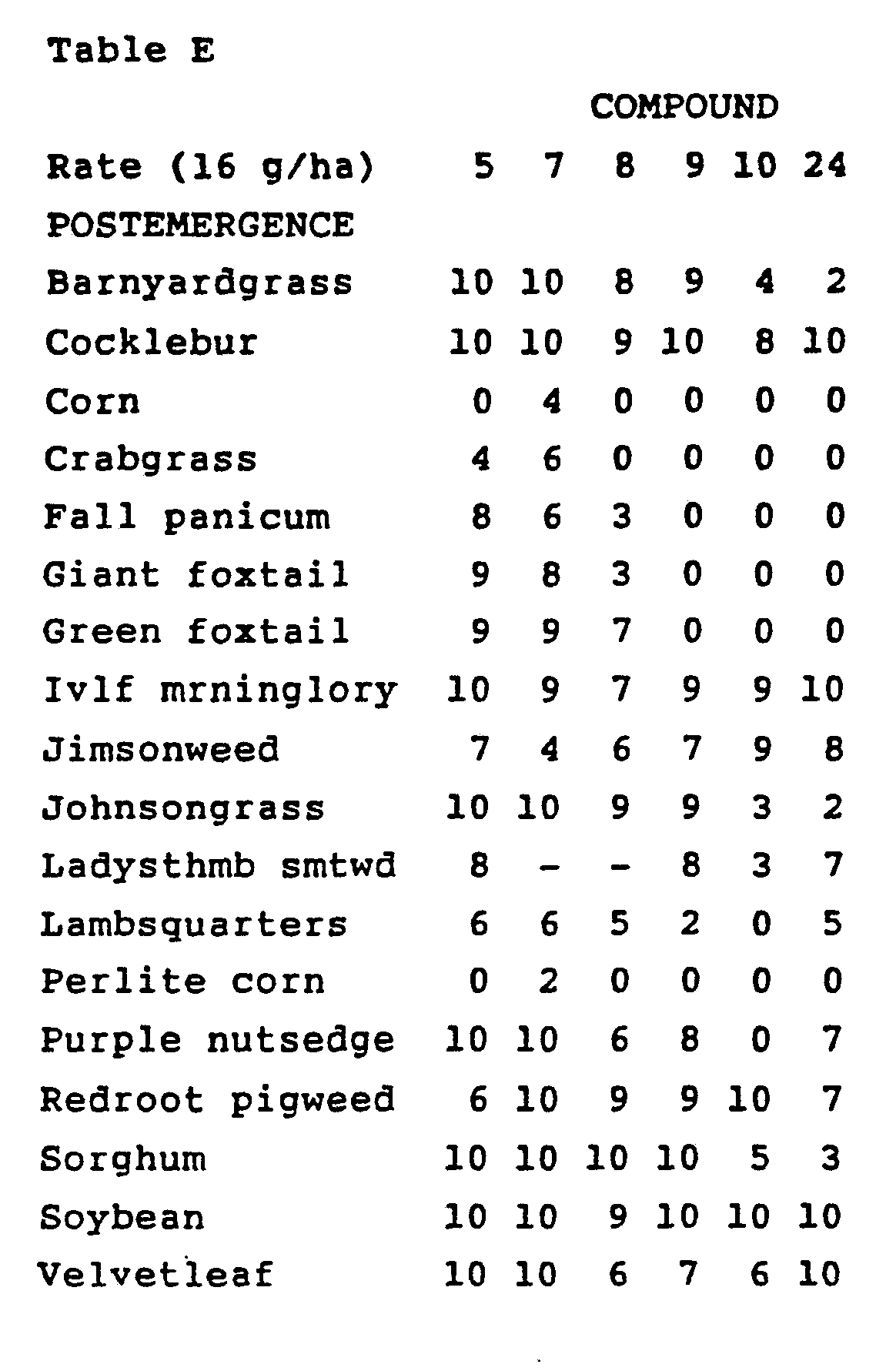
- Croperus rotundus tubers were planted and treated preemergence with test chemicals dissolved in a non-phytotoxic solvent. At the same time, these crop and weed species were also treated with postemergence applications of test chemicals. Plants ranged in height from two to eighteen cm (two to three leaf stage) for postemergence treatments. Treated plants and controls were maintained in a greenhouse for approximately 24 days, after which all species were compared to controls and visually evaluated. Plant response ratings, summarized in Table C, are reported on a 0 to 10 scale where 0 is no effect and 10 is complete control. A dash (-) response means no test result.
- preemergence application to water that covered the soil surface
- postemergence application to plants that were in the one-to-four leaf stage
- a sandy loam soil was used for the preemergence and postemergence tests, while a silt loam soil was used in the paddy test. Water depth was approximately 2.5 cm for the paddy test and was maintained at this level for the duration of the test,
- Plant species in the paddy test consisted of barnyardgrass (Echinochloa crus-galli), rice (Oryza sativa), and umbrella sedge (Cyperus difformis).
- cocklebur Xanthium pensylvanicum
- corn Zea mays
- soil surface exposed and soil surface covered with perlite crabgrass (Digitaria spp.), fall panicum (Panicum dichotomiflorum), giant foxtail (Setaria faberii), green foxtail (Setaria viridis), ivyleaf morningglory (Ipomoea hederacea), jimsonweed (Datura stramonium), johnsongrass (Sorghum
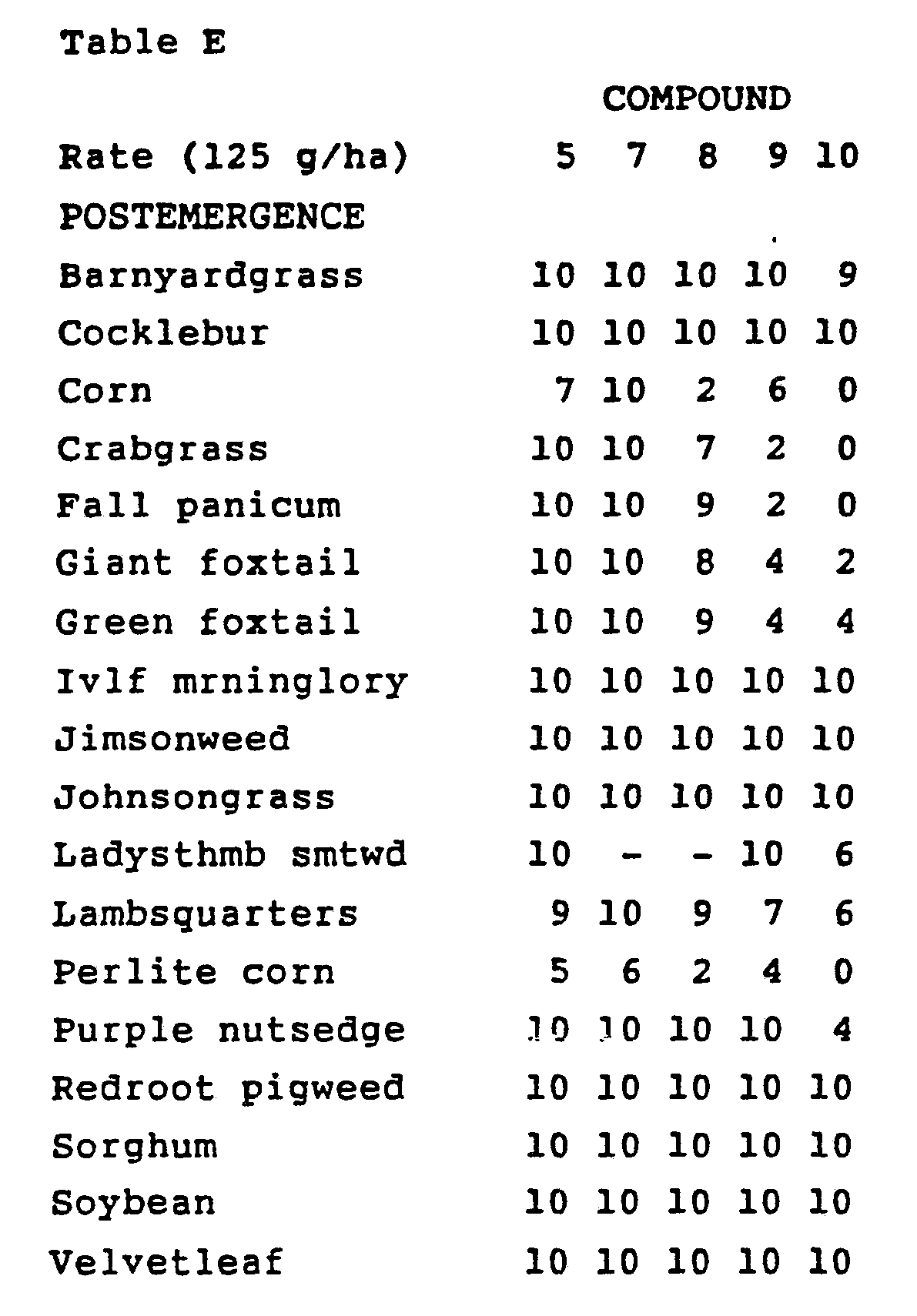
- Treated plants and controls were maintained in a greenhouse for approximately 24 days, after which all species were compared to controls and visually evaluated.
- the ratings, summarized in Table E, are based on a scale of 0 to 10 where 0 is no effect and 10 is complete control.
- a dash (-) response means no test result.
- Plastic pots were partially filled with silt loam soil. The soil was then saturated with water.
- Indica and Japonica rice (Oryza sativa) seedlings at the 2.0 to 2.5 leaf stage, seeds selected from barnyardgrass (Echinochloa crus-galli), bulrush
- a dash (-) response means no test result.
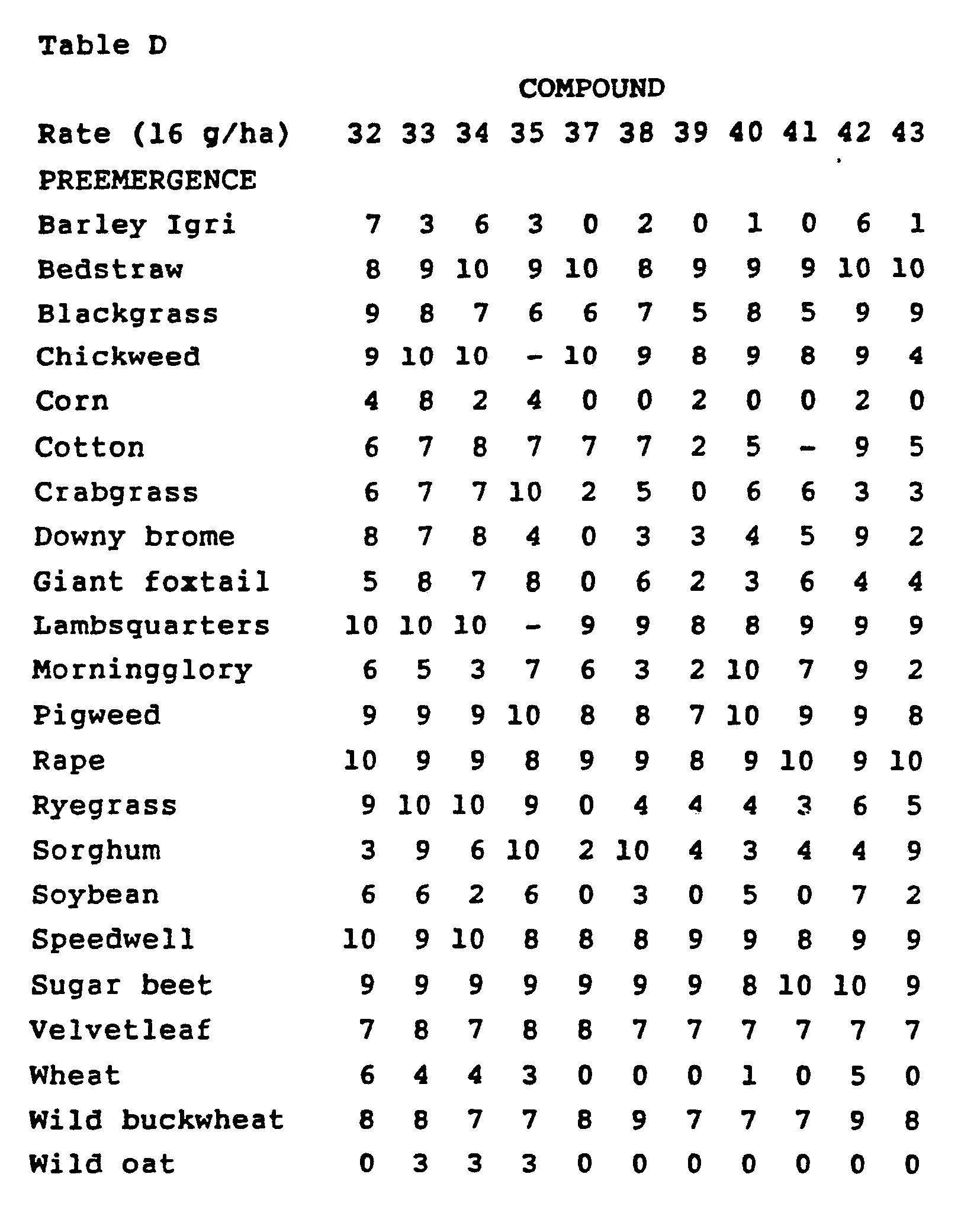
- Crop and weed species include winter barley (Hordeum vulgare cv. 'Igri'), bedstraw (Galium aparine), blackgrass (Alopecurus
- Blackgrass and wild oat were treated postemergence at two growth stages. The first stage (1) was when the plants had two to three leaves. The second stage (2) was when the plants had approximately four leaves or in the initial stages of tillering. Treated plants and untreated controls were maintained in a greenhouse for approximately 21 to 28 days, after which all treated plants were compared to untreated controls and visually
- Plant response ratings summarized in Table G, are based upon a 0 to 10 scale where 0 is no effect and 10 is complete control. A dash response (-) means no test result.
- Plants ranged in height from four to twenty cm (two to three leaf stage) when post-emergence applications were applied. Treated plants and controls were grown under greenhouse conditions for approximately
- Test chemical Application rates for the test chemical are shown in Table H. Plant response ratings, summarized in Table H, are from 0 to 10 where 0 is no injury and 10 is complete control. A dash (-) response means no test result.
Abstract
Description
Claims
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| BR919106301A BR9106301A (en) | 1990-04-02 | 1991-02-26 | SULPHONAMIDES, SELECTIVE HERBICIDES FOR CROPS |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US50318290A | 1990-04-02 | 1990-04-02 | |
| US503,182 | 1990-04-02 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO1991015478A1 true WO1991015478A1 (en) | 1991-10-17 |
Family
ID=24001052
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US1991/001075 WO1991015478A1 (en) | 1990-04-02 | 1991-02-26 | Crop-selective herbicidal sulfonamides |
Country Status (5)
| Country | Link |
|---|---|
| EP (1) | EP0523067A1 (en) |
| JP (1) | JP2799246B2 (en) |
| BR (1) | BR9106301A (en) |
| CA (1) | CA2079589A1 (en) |
| WO (1) | WO1991015478A1 (en) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0537585A2 (en) * | 1991-10-15 | 1993-04-21 | E.I. Du Pont De Nemours And Company | Process for preparing sulfonylureas |
| WO2002007519A1 (en) * | 2000-07-25 | 2002-01-31 | Bayer Cropscience Gmbh | Herbicide agent |
| EP1431278A1 (en) * | 2001-09-28 | 2004-06-23 | IHARA CHEMICAL INDUSTRY Co., Ltd. | Process for producing (2-nitrophenyl)acetonitrile derivative and intermediate therefor |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0073627A2 (en) * | 1981-08-24 | 1983-03-09 | E.I. Du Pont De Nemours And Company | Herbicidal sulfonamides |
| EP0096002A2 (en) * | 1982-05-26 | 1983-12-07 | Ciba-Geigy Ag | N-phenylsulfonyl-N'-pyrimidinyl- and -triazinyl-urea |
| EP0181526A1 (en) * | 1984-10-19 | 1986-05-21 | Chugai Seiyaku Kabushiki Kaisha | Benzoic acid derivatives |
| WO1989009214A1 (en) * | 1988-03-24 | 1989-10-05 | E.I. Du Pont De Nemours And Company | Fluoroalkoxy amino triazines for control of weeds in sugar beets |
-
1991
- 1991-02-26 WO PCT/US1991/001075 patent/WO1991015478A1/en not_active Application Discontinuation
- 1991-02-26 CA CA 2079589 patent/CA2079589A1/en not_active Abandoned
- 1991-02-26 EP EP19910905178 patent/EP0523067A1/en not_active Withdrawn
- 1991-02-26 JP JP3505118A patent/JP2799246B2/en not_active Expired - Fee Related
- 1991-02-26 BR BR919106301A patent/BR9106301A/en unknown
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0073627A2 (en) * | 1981-08-24 | 1983-03-09 | E.I. Du Pont De Nemours And Company | Herbicidal sulfonamides |
| EP0096002A2 (en) * | 1982-05-26 | 1983-12-07 | Ciba-Geigy Ag | N-phenylsulfonyl-N'-pyrimidinyl- and -triazinyl-urea |
| EP0181526A1 (en) * | 1984-10-19 | 1986-05-21 | Chugai Seiyaku Kabushiki Kaisha | Benzoic acid derivatives |
| WO1989009214A1 (en) * | 1988-03-24 | 1989-10-05 | E.I. Du Pont De Nemours And Company | Fluoroalkoxy amino triazines for control of weeds in sugar beets |
Cited By (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0537585A2 (en) * | 1991-10-15 | 1993-04-21 | E.I. Du Pont De Nemours And Company | Process for preparing sulfonylureas |
| EP0537585A3 (en) * | 1991-10-15 | 1993-06-16 | E.I. Du Pont De Nemours And Company | Process for preparing sulfonylureas |
| WO2002007519A1 (en) * | 2000-07-25 | 2002-01-31 | Bayer Cropscience Gmbh | Herbicide agent |
| US6770594B2 (en) | 2000-07-25 | 2004-08-03 | Aventis Cropscience | Herbicidal compositions |
| AU2001289654B2 (en) * | 2000-07-25 | 2006-02-02 | Bayer Cropscience Ag | Herbicide agent |
| HRP20030040B1 (en) * | 2000-07-25 | 2011-06-30 | Bayer Cropscience Ag | Herbicide agent |
| EP1431278A1 (en) * | 2001-09-28 | 2004-06-23 | IHARA CHEMICAL INDUSTRY Co., Ltd. | Process for producing (2-nitrophenyl)acetonitrile derivative and intermediate therefor |
| EP1431278A4 (en) * | 2001-09-28 | 2006-01-25 | Ihara Chemical Ind Co | Process for producing (2-nitrophenyl)acetonitrile derivative and intermediate therefor |
Also Published As
| Publication number | Publication date |
|---|---|
| JP2799246B2 (en) | 1998-09-17 |
| JPH05505796A (en) | 1993-08-26 |
| CA2079589A1 (en) | 1991-10-03 |
| BR9106301A (en) | 1993-04-13 |
| EP0523067A1 (en) | 1993-01-20 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO1992009587A1 (en) | Herbicidal oxazine ethers | |
| WO1991010653A1 (en) | Herbicidal pyrimidines and triazines | |
| EP0440659A1 (en) | Substituted phenyltriazolopyrimidine herbicides | |
| US4906282A (en) | Herbicidal sulfonamides | |
| US5356862A (en) | Herbicidal sulfonylureas | |
| US4946494A (en) | Herbicidal pyridine sulfonylureas | |
| AU8549891A (en) | Herbicidal pyrrolesulfonylureas | |
| US5125954A (en) | Herbicidal oxabicycloalkane ethers | |
| US5071469A (en) | Herbicidal benzylsulfonamides | |
| WO1993003020A1 (en) | Herbicidal quinoxalinyloxy ethers | |
| US5127936A (en) | Substituted phenyltriazolopyrimidine herbicides | |
| WO1991015478A1 (en) | Crop-selective herbicidal sulfonamides | |
| US5032166A (en) | Herbicidal pyridine sulfonylureas | |
| US5069709A (en) | Herbicidal thiadiazolo pyrimidines | |
| WO1989000994A2 (en) | Herbicidal sulfonamides | |
| US4995901A (en) | Herbicidal sulfonamides | |
| US5480856A (en) | 1,2,4-triazolo[1,2-a]pyridazine-1,3(2H)-dione herbicides for citrus, sugarcane, oil palm and the like | |
| WO1989004605A1 (en) | Herbicidal alkanoylpyridinesulfonylureas | |
| WO1989002700A2 (en) | Herbicidal o-carbomethoxysulfonylureas | |
| WO1992013861A1 (en) | Herbicidal bicyclic ethers | |
| US5175314A (en) | Herbicidal oxatricyclic ethers | |
| US4881968A (en) | Herbicidal sulfonamides | |
| US5407901A (en) | Herbicidal oxatricyclic ethers | |
| WO1990010634A1 (en) | Selective oxabicycloalkanes | |
| WO1992013453A1 (en) | Herbicides for citrus, sugarcane, oil palm and the like |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): BR CA JP US |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): AT BE CH DE DK ES FR GB GR IT LU NL SE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1991905178 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2079589 Country of ref document: CA |
|
| WWP | Wipo information: published in national office |
Ref document number: 1991905178 Country of ref document: EP |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: 1991905178 Country of ref document: EP |