WO1988005533A1 - Amplification of signals from optical fibers - Google Patents
Amplification of signals from optical fibers Download PDFInfo
- Publication number
- WO1988005533A1 WO1988005533A1 PCT/US1988/000041 US8800041W WO8805533A1 WO 1988005533 A1 WO1988005533 A1 WO 1988005533A1 US 8800041 W US8800041 W US 8800041W WO 8805533 A1 WO8805533 A1 WO 8805533A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- polymer
- fiber
- glass
- fluorescent
- photoactive moieties
- Prior art date
Links
Classifications
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N21/00—Investigating or analysing materials by the use of optical means, i.e. using sub-millimetre waves, infrared, visible or ultraviolet light
- G01N21/62—Systems in which the material investigated is excited whereby it emits light or causes a change in wavelength of the incident light
- G01N21/63—Systems in which the material investigated is excited whereby it emits light or causes a change in wavelength of the incident light optically excited
- G01N21/64—Fluorescence; Phosphorescence
- G01N21/6428—Measuring fluorescence of fluorescent products of reactions or of fluorochrome labelled reactive substances, e.g. measuring quenching effects, using measuring "optrodes"
- G01N21/643—Measuring fluorescence of fluorescent products of reactions or of fluorochrome labelled reactive substances, e.g. measuring quenching effects, using measuring "optrodes" non-biological material
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K11/00—Luminescent, e.g. electroluminescent, chemiluminescent materials
- C09K11/06—Luminescent, e.g. electroluminescent, chemiluminescent materials containing organic luminescent materials
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/53—Immunoassay; Biospecific binding assay; Materials therefor
- G01N33/543—Immunoassay; Biospecific binding assay; Materials therefor with an insoluble carrier for immobilising immunochemicals
- G01N33/551—Immunoassay; Biospecific binding assay; Materials therefor with an insoluble carrier for immobilising immunochemicals the carrier being inorganic
- G01N33/552—Glass or silica
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N21/00—Investigating or analysing materials by the use of optical means, i.e. using sub-millimetre waves, infrared, visible or ultraviolet light
- G01N21/62—Systems in which the material investigated is excited whereby it emits light or causes a change in wavelength of the incident light
- G01N21/63—Systems in which the material investigated is excited whereby it emits light or causes a change in wavelength of the incident light optically excited
- G01N21/64—Fluorescence; Phosphorescence
- G01N21/6428—Measuring fluorescence of fluorescent products of reactions or of fluorochrome labelled reactive substances, e.g. measuring quenching effects, using measuring "optrodes"
- G01N2021/6432—Quenching
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N21/00—Investigating or analysing materials by the use of optical means, i.e. using sub-millimetre waves, infrared, visible or ultraviolet light
- G01N21/75—Systems in which material is subjected to a chemical reaction, the progress or the result of the reaction being investigated
- G01N21/77—Systems in which material is subjected to a chemical reaction, the progress or the result of the reaction being investigated by observing the effect on a chemical indicator
- G01N21/7703—Systems in which material is subjected to a chemical reaction, the progress or the result of the reaction being investigated by observing the effect on a chemical indicator using reagent-clad optical fibres or optical waveguides
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N21/00—Investigating or analysing materials by the use of optical means, i.e. using sub-millimetre waves, infrared, visible or ultraviolet light
- G01N21/75—Systems in which material is subjected to a chemical reaction, the progress or the result of the reaction being investigated
- G01N21/77—Systems in which material is subjected to a chemical reaction, the progress or the result of the reaction being investigated by observing the effect on a chemical indicator
- G01N21/78—Systems in which material is subjected to a chemical reaction, the progress or the result of the reaction being investigated by observing the effect on a chemical indicator producing a change of colour
- G01N21/81—Indicating humidity
Definitions
- This invention relates generally to the use of optical fibers for measuring various chemical and biochemical parameters in fluid or body tissue.
- optical fibers for such analytic purposes is known and described, for example, in Hirschfeld, U.S. Patent No. 4,577,109, entitled “Remote Multi-Position Information Gathering System and Method.” Such techniques are also described by F.P. Milanovich, T.B. Hirschfeld, F.T. Wang, S.M. Klainer, and -D. Walt in "Novel Optical Fiber Techniques for Medical Application,” published in the Proceedings of the SPIE 28th Annual International Technical Symposium on Optics and Electrooptics, Volume 494.
- a sensor consisting typically of a fluorescent dye is attached to the distal end of an optical fiber, preferably of diameter suitable for in vivo application.
- Light of a suitable wavelength from an appropriate source, is used to illuminate the distal end of the optical fiber, visible light, for example, having a wavelength typically between about 400 and 800 nm.
- the light is propagated along the fiber to its distal end.
- a fraction of the light is reflected at the fiberliquid interface and returns along the fiber.
- a second fraction of the light is absorbed by the fluorescent dye, and light is then emitted therefrom in a band centered usually but not always at a longer wavelength.
- the intensity of this band is dependent upon the interaction of the dye with the property or substance measured in the body fluid or tissue.
- This fluorescent light is also propagated along the return path of the fiber and measured using suitable optical equipment. The ratio of the wavelengths is determined and the parameter of interest measured.
- Fluorescent molecules have been attached to the distal end of an optical fiber or to a separate piece of glass, e.g., a glass bead, by a technique which is used in the immobilization of enzymes. See Methods of Enzymoloqy, vol. XLIV, Ed. Klaus Mosbach, Academic Press, pp. 134-148 (1976) . A fundamental problem encountered with such a technique has been the failure to provide sufficient amounts of indicator at the distal end of the fiber.
- the prior art describes tw.o primary approaches to the problem. In the first of these, a porous substrate is used so that more surface area is available to which indicator moieties may bind, while in the second, as described, for example, in U.S. Patent No. 4,269,516 to Lubbers et al. , indicator is provided in solution form, which, solution is separated from the external environment by a membrane.
- a glass surface for example the distal end of an optical fiber (or a glass bead which is subsequently attached to the fiber), is treated so that it is porous. It is then reacted with a suitable agent such as 3-aminopropyltriethoxy silane, and a series of reactions is carried out culminating in the covalent attachment of a biologically and/or chemically active molecule, e.g., a fluorescent species, to the surface of the glass.
- a biologically and/or chemically active molecule e.g., a fluorescent species
- silanized glass referred to also hereinafter as aminoalkyl glass or fiber
- the next step is realized by reacting a fluorophore such as fluorescein isothiocyanate dissolved in an aprotic solvent with the aminoalkyl fiber
- a dye immobilized on glass in this manner is pHsensitive in the physiological range and is relatively 0 stable.
- porous glass that is, a special alkali borosilicate glass which has been heat treated to separate the glass into two phases.
- the phase which is rich in boric acid and 5 alkali is leached out with acid to leave a porous, high-
- SUBSTITUTE SHEET silica structure The porous structure is then silanized and treated with the desired fluorescent species. The resulting treated glass is then attached to the distal end of an optical fiber. Porous glass provides more surface area available for silanizing and thus many more fluorescent molecules can be attached.
- a fiber optic sensing device comprising an optical fiber to one end of which is attached a polymer including a plurality of photoactive moieties spaced apart from each other at a preselected distance. It is still another object of the invention to provide such a device wherein the preselected distance is sufficient to minimize chemical or physical interaction between the photoactive moieties while optimizing the density thereof. It is yet another object of the invention to provide such a device wherein the photoactive moieties are covalently attached to the polymer through functional groups, such as through ethers, amides, or the like. It is a further object of the invention to provide such a device wherein the polymer is inherently fluorescent and includes at least one monomer unit which is itself photoactive.
- a fiber optic sensing device comprising a fiber to one end of which is attached a polymer including a plurality of photoactive moieties.
- the photoactive moieties which may be chromophores or lumophores are positioned along the polymer in such a way as to optimize the distance between the moieties. That is, chemical or physical interaction (e.g., concentration quenching, exciton interaction, steric interference) is minimized while the density of photoactive moieties is optimized.
- the high density of photoactive moieties achieved with the present invention provides an apparatus of substantially increased sensitivity.
- the photoactive moieties may be attached as side groups, i.e. pendant to the main polymer chain. In such a case, each photoactive moiety is attached through one bond to the polymer backbone. In an alternative embodiment of the invention, in which the polymers are designated "inherently fluorescent,” each photoactive moiety is in the backbone of the polymer, i.e. is bound to the remainder of the polymer through two or more bo ds.
- a method of forming a fiber optic device involves either sequential attachment of labeled monomer units to a riber optic tip, or, alternatively, attachment (covalent or otherwise) of a preformed polymer to a fiber optic tip; the preformed polymer may be labeled with photoactive moieties before attachment or after.
- Potential uses of the device include, inter alia, use as a pH sensor, an oxygen sensor, an electrolyte sensor and a blood gas sensor.
- photoactive moieties are molecular species which undergo a detectable change in electronic configuration in response to interaction with light.
- the detectable change corresponds to one or more chemical or physiological parameters.
- Chrophors are atoms or groups of atoms that serve as units in light absorption.
- a chromophore includes the electronic substructure of a dye that absorbs light and thereby causes the material to be colored.
- a change in the environment of a chromophore can change the amount of light absorbed at a particular wavelength or the amount of light absorbed over a range of wavelengths.
- chromophores useful herein include cresol red and phenol red.
- Suitable chromophores also include lumophores such as pyrenes , perylenes and their derivatives, and other paracyclic aromatic compounds.
- Liophores are atoms or groups of atoms that serve as units in light emission. All compounds which are capable of undergoing fluorescence or phosphorescence include a lumophore.
- “Chemical or physical interaction” between the photoactive moieties of a polymer as used herein includes any interaction which (1) changes the electronic absorption spectrum obtained; (2) changes the electronic emission spectrum obtained; (3) changes the quantum yield of emission; (4) interferes with the accurate measurement of particular chemical or physiological parameters in a sample or (5) destabilizes the polymer structure.
- a “substantially nonporous” material is a material having a porosity less than about 5%______ (vol./vol.).
- optical fiber of the present invention is typically between about' 50 ⁇ and about 600 ⁇ in diameter.
- the sensing device is useful for in vivo applications, for example to measure pH or oxygen concentration, in which case the diameter should be between about 50 ⁇ and about 250 ⁇ .
- the fiber may be comprised of virtually any material so long as it is an optical conductor at the wavelengths of interest.
- the fiber may be an organic material such as polymethylmethacrylate, polystyrene, polymethylphenyl siloxane or deuterated methyl methacrylate, or it may be an inorganic material such as glass.
- the polymer which is attached to the fiber tip can be any stable polymer which can be derivatized so as to contain photoactive moieties.
- the polymer must be optically transparent both to exciting light, i.e. light directed through the fiber into the polymer, and to light emitted from the photoactive moieties attached thereto.
- Examples of polymers suitable for use herein include polyacrylamides, polylysines, polymethacrylates, polyurethanes, polyethers, and polysiloxanes such as polydimethyl and polymethylphenyl siloxane.
- the polymer chain is preferably less than about 200 monomer units in length, to ensure that substantially all photoactive moieties are maintained fairly close to the fiber tip.
- the polymer may be attached to the fiber in a number of ways.
- the polymer is attached to the fiber covalently, typically through an aminoalkyl or other linking group (see Example 4).
- Suitable linking groups for glass and other fibers may be prepared from the following: diaminoalkanes, diaminoaryl compounds, diisothiocyanate-substituted aryl compounds, alkyldialdehydes, arylalkyl dialdehydes, arylalkyldiamines, alkyl dicarboxylic acid derivatives, arylalkyl decarboxylic acid derivatives, aryl dicarboxylic acid derivatives, alkyl diols, aryl diols, arylalkyl diols, and the like.
- the polymer may be attached to the polymer by adsorption or electrostatic interaction. In the latter case, the polymer is attached to the fiber by forming a salt of the polymer and a linking group
- the polymer is of the "inherently fluorescent" type, it must be provided with areas of reactivity so that photoactive moieties can bind to the polymer backbone.
- the photoactive moieties may bind directly to the polymer backbone or may bind to the polymer through functional groups such as ethers, amines, amides including acrylamides, esters, alkyl moieties having from about two to about twenty-two carbon atoms, arylalkyl, etc.
- Suitable photoactive moieties for use herein include chromophores, lumophores and combinations thereof. Chromophores include, for example, cresol red and phenol red. Lumophores include both fluorophores and phosphors. Examples of suitable fluorophores for use herein are fluorescein, perylene, pyrene and their derivatives. An examples of a suitable phosphor is rose bengal.
- the reactive areas on the polymer are distributed along the polymer chain such that the photoactive moieties are spaced apart by at least about 4 angstroms, that is, the distance between photoactive moieties is determined by the relative frequency of the species in the polymer backbone. This frequency can be achieved during preparation of the polymer by diluting the photoactive moiety, i.e. by introducing a nonphotoactive comonomer during polymerization. Alternatively, with a preformed polymer having reactive functional groups to which ' photoactive moieties will bind, the frequency of these moieties can be controlled by dilution during the reaction attaching the pendant moieties or by using a nonphotoactive moiety which can compete for binding to the functional groups on the polymer chain.
- spacing of at least about 14 angstroms is preferred depending on the environment and on the photoactive moieties selected, i.e. where there is a potential for interaction at a greater distance.
- the distance factor is key to the present invention, as the distance between photoactive moieties is carefully preselected so as to minimize chemical or physical interaction therebetween, while optimizing the density thereof close to the fiber tip.
- the inventors herein have demonstrated that, generally, the amount of light recaptured by the fiber from the photoactive moieties is strongly dependent on the density ⁇ of these groups. The strength of the signal ultimately generated is also dependent on the distance of the active moieties from the end of the fiber to these groups.
- the fiber tip will be provided with a plurality of polymer chains all similarly bonded to the fiber.
- the spacing of photoactive moieties is then controlled not only along a single polymer chain but also between polymer chains. This is accomplished by controlling the number of polymer chains attached to a given area of the fiber tip, for example, by minimizing the number of attachment sites on the substrate or by reacting some of the sites with nonphotoactive polymer chains.
- a primary use of the above-described sensing device is as a pH sensor.
- the fiber tip is placed into contact with a fluid or tissue, light is directed along the fiber toward the fiber-sample interface so that photoactive species are caused to fluoresce, and the ratio of fluorescence to reflected light is determined. This ratio changes with pH in a predictable fashion; thus, pH may be determined by interpolating along a curve prepared from known pH/ratio points.
- a fiber optic sensing device which comprises a fiber tip to one end of which is attached an inherently fluorescent polymer.
- the polymer has as part of its molecular structure a chromophore, lumophore or the like, i.e. it is actually formed from one or more photoactive monomer units.
- a second advantage is that with inherently fluorescent polymers, as with the polymers having pendant photoactive moieties, there is no separate dye which can migrate out of the polymer matrix. This promotes long- term stability of the device and allows the material to be used in contexts where dye toxicity could otherwise be a problem.
- the inherently fluorescent polymers are prepared by reaction of a photoactive moiety with a reactive monomeric species under polymerization conditions.
- the photoactive moieties may be introduced within a straight-chain polymer or as cross-linking functionalities in a cross-linked polymer (see Examples 16 and 17).
- Suitable photoactive moieties are as described earlier, and particularly suitable photoactive moieties for use in the inherently fluorescent polymer application are derivatives of pyrene.
- Preferred polymers include polycarbonamides, polyurethanes, polyamides, polyimides, polyesters, and polysiloxanes such as polymethylsiloxanes, polymethylphenylsiloxanes and the like.
- a preferred use of the fiber optic sensing device which incorporates an inherently fluorescent polymer at the optical fiber tip is as an oxygen sensor.
- the photoactive moieties incorporated in the polymer are fluorescent species which are quenchable by oxygen.
- the device is constructed so that fluorescence emission varies with (typically is inversely related to) the oxygen concentration of its environment, which may be gaseous or aqueous (e.g., blood).
- the present invention also encompasses a method of attaching labeled polymeric species — inherently fluorescent or otherwise -to a fiber optic tip.
- the method involves sequential addition of monomer or oligomer species to a linking group present on the fiber. At least a fraction of these species is photoactive.
- the method involves attachment of a preformed polymer to the fiber tip, which polymer may be labeled with photoactive moieties prior to attachment or after. The attachment of the preformed polymer may be covalent, or it may be effected by adsorption or electrostatic binding.
- This compound is a fluorophore having an acryloyl substituent on an aromatic ring, and in one embodiment is given by the following structure
- the compound may be further substituted on the aromatic rings, e.g., with halogen substituents or lower alkyl or alkoxy groups.
- a fluorescent acrylamide is then polymerizable (with or without nonfluorescent acrylamide monomers) in the presence of a catalyst such as 2,2'- azobisisobutyronitrile to form fluorescent polymers having the structure
- x and y represent the number of nonfluorescent and fluorescent monomer units in the polymeric structure, respectively, and are selected so as to provide the requisite spacing between photoactive moieties.
- x and y each range from about 1 to about 200, and the sum of x and y is also, typically, less than about 200.
- F represents the major portion of the molecule given by
- the group -NH- is derived from the primary amino group of the aminoalkyl glass.
- the acid glass 1 was prepared as follows:
- Example 2 Preparation of Acid Chloride Glass
- the acid glass of Example 1 was treated with SOCI2 as follows: A 10% solution of thionyl chloride (Aldrich) in dry chloroform was prepared. 0.5 g of acid glass was added to 20 ml of the thionyl chloride solution and refluxed for four hours. The glass was then rinsed with chloroform and dried under vacuum for thirty minutes. o o o
- Example 3 Preparat ion of N-Hydroxy Succinic (NHS) Glass
- N-hydroxysuccinimide was reacted with N-hydroxysuccinimide as follows : o
- reaction was carried out as follows: 3.8 g of N-hydroxysuccinimide (Aldrich) and 4.45 ml of triethylamine (Aldrich) were added to 50 ml of anhydrous stirred chloroform. This solution was then cooled to 0°C. Acid chloride glass was added and the suspension was stirred for an additional forty minutes. The temperature of the suspension was maintained at 0°C during this stirring period. The glass was filtered, and the beads were washed with chloroform and dried in vacuo at 25°C for thirty minutes.
- the purpose of treating the acid chloride glass with N-hydroxy succinimide was to protect the treated glass from hydrolysis in subsequent treatment.
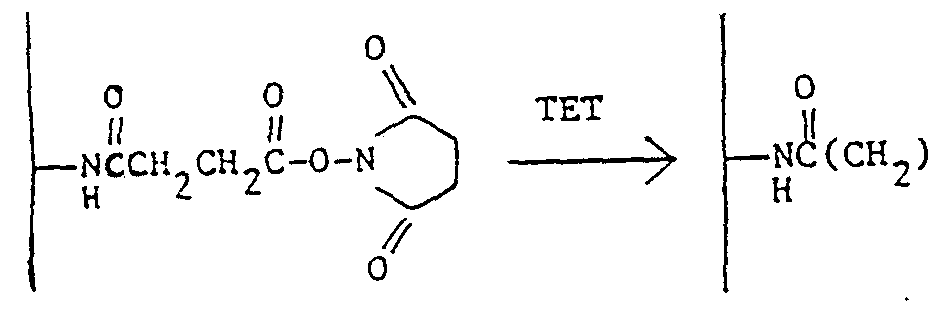
- Example 4 Treatment of NHS Glass with Triethylenetetramine (TET)
- TET Triethylenetetramine
- the NHS glass from Example 3 is added with stirring to a mixture of 50 ml of anhydrous chloroform and 8 ml of triethylenetetramine (Aldrich) .
- the glass mixture is stirred for one hour, and the beads are then filtered, washed with chloroform and dried in vacuo at 25°C for thirty ⁇ . minutes.
- the reaction may be represented as
- the treated, aminoalkyl glass 4. has numerous nitrogen functionalities to which fluorescent groups may be attached.
- Glutaraldehyde glass (represented by structure 5_) was prepared as follows. A solution of 25% glutaraldehyde was diluted to 2.5% in 0.1 M Na3P04, pH 7, to._50 ml. The aminoalkyl glass prepared in the preceding Example was immersed for one hour and then rinsed with deionized water.
- the pendant group F is derived from 8_; i.e., it is the group 8a.
- FITC fluorescein isothiocyanate
- the glass to which the fluorescent species was attached was nonporous. It was treated to append aminoalkyl groups but eliminated the tedious step of attachment of a porous bead.
- the polymeric technique thus achieves a result which is similar to that achieved with porous glass, the porous glass route, however, being less desirable due to attachment problems.
- Example 8 Glass Fiber Treated with FITC
- aminoalkyl glass fiber as in Example 7(b) was treated as follows: The fiber was soaked first in a solution containing 5 mg of FITC in 100 ml of acetone for one hour and then in deionized water for one hour. No observable fluorescent pH effect could be measured using the procedure of Example 7(c) .
- Example 9 Glass Fiber Treated with High Molecular Weight Polylysine and FITC
- a glutaraldehyde-treated aminopr ⁇ pyl glass fiber (prepared as in Example 5) was placed in a solution of 4 mg of 540,000 D average molecular weight (M v ) polylysine in 4 ml of deionized water and allowed to soak for one hour at 23°C. The fiber was then allowed to stand in a deionized water wash for one hour after which time it was treated with a. solution of 5 mg FITC/100 ml acetone for one hour. Its pH-dependent fluorescence was measured. At pH 6.8 the ratio of fluorescence to reflected light was 0.11, and at pH 7.4 it was 0.21.
- a glutaraldehyde-treated aminopropyl glass fiber (prepared as in Example 5) was placed in a solution of 4 mg of 4,000 D average molecular_weight ( M w) polylysine (Polysciences) in deionized water and allowed to soak for one hour at 23°C. The fiber was then placed in a deionized water wash for one hour and immediately treated with a solution of 5 mg FITC/100 ml acetone for one hour. The fiber was then rewashed in deionized water for one hour and its pH-dependent fluorescence measured. The ratio of fluorescent to reflected light at pH 6.8 was 0.35 and at pH 7.4 was 0.52.
- Electrostatically Bound Fluorescent Polymer A fluorescent polymer is prepared having pendant fluorescent groups and pendant carboxyl groups.
- the polymer is caused to react with activated glass (i.e., the aminoalkyl glass of Example 4) having basic groups, resulting in the following:
- C represents one of a multiplicity of activated groups on glass
- D represents a repeating unit ⁇ in the polymer described above (wherein F represents a pendant fluorescent group) and E represents the resulting salt. This may be accomplished by reacting aminopropyl glass C with a fluorescently labeled polymer D. The resulting ionic salt bridges are then stabilized by drying the product in vacuo.
- Example 12 Adsorption of a Fluorescent Polymer on an Aminoalkyl Glass Surface
- a polymer having pendant fluorescent groups is prepared, e.g., a copolymer of acrylamide and the acryloyl derivative 8. of fluorescein prepared as described in Example 7(b) but in the absence of aminopropyl glass.
- An aminopropyl glass fiber tip is immersed in a 0.01 molar aqueous solution of this copolymer for two days. The fiber is then rinsed with chloroform for three days to remove the nonadsorbed polymer and is dried in vacuo at 25°C for one hour.
- Example 13 Immunoassay Embodiment In the enzyme-linked immunoassay (ELISA) technique, an enzyme is immobilized on the surface of a test tube or cuvette. See, for example, Chang, Biochemical Applications of Immobilized Enzymes and Proteins, Vol. 2, Plenum Press, 1977, Chapters 30-33. This technique is improved by the amplification technique of the present invention as follows.
- ELISA enzyme-linked immunoassay
- An acetaminophen-specific antibody is treated with a one hundredfold molecular excess of acryloyl chloride, and taken into solution with deionized water to which a twofold quantity of acrylamide hasJseen added together with a suitable amount of AIBN.
- An aminopropyl glass optical fiber is treated with an excess of acryloyl chloride, washed, added to the acrylated antibod /AIBN solution and warmed at a sufficient temperature to promote graft polymerization of the antibody derivative to the silica surface without denaturing the protein.
- an acetaminophen-alkaline phosphatase conjugate is incubated with the immobilized antibody, filtered, washed, and the unknown quantity of acetaminophen-analyte added. The residual bound enzyme activity is then measured and the quantity of acetaminophen calculated.
- fluorescent species such as compound 8. and FITC are used and the ratio of fluorescent light to reflected light is measured.
- active species other than fluorescent species may be used.
- phenol red, cresol red and neutral red may be used. These dyes change color with pH.
- These dyes have functional groups which are capable of reacting with functional groups of monomers and polymers and can be incorporated in polymer molecules formed in situ or in preformed polymers. The following example will serve for purposes of illustration.
- Cresol red (0.038 g) is dissolved in 30 ml of chloroform. N-Bromosuccinimide (0.017 g) is added along with benzoyl peroxide (0.005 g) . The solution is refluxed for one hour and allowed to cool to room temperature. The succinimide is removed by filtration and the bromomethyl cresol red is crystallized from acetic acid. To a solution of dimethylformamide (25 ml) containing the bromoethyl cresol red derivative (0.025 g) is added a tenfold molar excess of allylamine. The reaction mixture is heated with stirring at 50°C under argon in the dark for six hours. The reaction mixture is then cooled to room temperature and the solvent removed under high vacuum.
- the reaction product of the bromomethyl cresol red and allylamine is dissolved in DMF (50 ml) together with 1 g of acrylamide and lOmg 2,2'- azobisisobutyronitrile.
- DMF 50 ml
- aminoalkyl glass in the form of a fiber tip, . which has been previously treated with a mixture of acryloyl chloride (0.10 g), pyridine (20 ml), and N,-N'- dimethylamino pyridine (0.010 g) for two hours.
- the resulting amidated fiber is heated with the bromomethyl cresol red allylamine product and acrylamide is formed at the end of the fiber.
- I I the remainder of the molecule is as in I.
- Ill is the glass fiber to which a copolymer of acrylamide and II is covalently attached.
- Example 15 Preparation and Use of a Fluorescent Polycarbonamide
- the well-rinsed fiber was inserted through a condenser attached to a flask charged with diaminodecane (137 mg) , sebacoyl chloride ( 190 mg ) , perylene-3,9-dicarboxylic acid chloride ( 3 mg ) , triethylamine (107 mg) and chloroform ( 100 ml ) .
- the solution was refluxed, and a fluorescent polymer was allowed to form and grow from the end of the fiber. After one hour, the fiber was removed and rinsed.
- fluorescent green light When properly filtered blue light was launched into the fiber, fluorescent green light could be collected (after it had traveled back along the optical fiber) by use of a dichroic mirror and appropriate optical filter. The intensity of the green light was inversely related to the amount of oxygen in the environment surrounding the polymer tip. This, was observed when the polymer tip was in a gaseous or aqueous environment.
- Example 16 Preparation of a Fluorescent Silicone A sample of the diallyl ester of perylene-3,9- dicarboxylic acid (3 mg) was mixed with a 1 ml sample of a methylhydrodimethyl siloxane copolymer, and a small amount of platinum catalyst was added. Upon heating, a homogeneous fluorescent green or yellow material was formed. The intensity of the fluorescence emission of this material varies with the oxygen content of its environment. The material may be mounted at the end of a fiber by a mechanical means such as a capillary, and the fiber can then be used as an optrode.
- a mechanical means such as a capillary
- a piece of 240 ⁇ ID CelgardTM hollow porous fiber was slipped over the end of a 140 ⁇ plastic-clad silica optical fiber.
- a sample of the partially cured polymer was then wicked into the hollow fiber.
- the fiber was then heated while the polymer cured to form a solid material.
- the fluorescence emission from this tip when monitored as described in Example 1, was inversely related to the oxygen concentration of its environment. This was observed in both gaseous and aqueous environments, including canine blood. This behavior could still be observed when the optical fiber had been subjected to steam sterilization,
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Immunology (AREA)
- Engineering & Computer Science (AREA)
- Physics & Mathematics (AREA)
- Molecular Biology (AREA)
- Urology & Nephrology (AREA)
- Analytical Chemistry (AREA)
- Pathology (AREA)
- General Physics & Mathematics (AREA)
- Hematology (AREA)
- General Health & Medical Sciences (AREA)
- Biomedical Technology (AREA)
- Biochemistry (AREA)
- Inorganic Chemistry (AREA)
- Medicinal Chemistry (AREA)
- Materials Engineering (AREA)
- Food Science & Technology (AREA)
- Organic Chemistry (AREA)
- Microbiology (AREA)
- Cell Biology (AREA)
- Biotechnology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Optics & Photonics (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Investigating Or Analysing Materials By The Use Of Chemical Reactions (AREA)
- Investigating, Analyzing Materials By Fluorescence Or Luminescence (AREA)
Abstract
A fiber optic sensing device for measuring a chemical or physiological parameter of a body fluid or tissue. To one end of the fiber is attached a polymer including a plurality of photoactive moieties selected from the group consisting of chromophores and lumophores, the photoactive moieties spaced apart so as to minimize chemical or physical interaction therebetween while optimizing the density of photoactive moieties. In one embodiment, a polymer chain is covalently bound to photoactive moieties through functional groups such as esters, amides, or the like. In a second embodiment, a polymer chain is inherently fluorescent and is formed from at least one monomeric unit. These devices are particularly useful as pH and oxygen sensors.
Description
AMPLIFICATION OF SIGNALS
FROM OPTICAL FIBERS
Description
Technical Field
This invention relates generally to the use of optical fibers for measuring various chemical and biochemical parameters in fluid or body tissue.
Background
The use of optical fibers for such analytic purposes is known and described, for example, in Hirschfeld, U.S. Patent No. 4,577,109, entitled "Remote Multi-Position Information Gathering System and Method." Such techniques are also described by F.P. Milanovich, T.B. Hirschfeld, F.T. Wang, S.M. Klainer, and -D. Walt in "Novel Optical Fiber Techniques for Medical Application," published in the Proceedings of the SPIE 28th Annual International Technical Symposium on Optics and Electrooptics, Volume 494.
Briefly summarizing the method of the prior art, a sensor consisting typically of a fluorescent dye is attached to the distal end of an optical fiber, preferably of diameter suitable for in vivo application. Light of a suitable wavelength, from an appropriate source, is used to illuminate the distal end of the optical fiber, visible light, for example, having a wavelength typically between about 400 and 800 nm. The light is propagated along the fiber to its distal end.
A fraction of the light is reflected at the fiberliquid interface and returns along the fiber. A second fraction of the light is absorbed by the fluorescent dye, and light is then emitted therefrom in a band centered usually but not always at a longer wavelength. The intensity of this band is dependent upon the interaction of the dye with the property or substance measured in the body fluid or tissue. This fluorescent light is also propagated along the return path of the fiber and measured using suitable optical equipment. The ratio of the wavelengths is determined and the parameter of interest measured.
Fluorescent molecules have been attached to the distal end of an optical fiber or to a separate piece of glass, e.g., a glass bead, by a technique which is used in the immobilization of enzymes. See Methods of Enzymoloqy, vol. XLIV, Ed. Klaus Mosbach, Academic Press, pp. 134-148 (1976) . A fundamental problem encountered with such a technique has been the failure to provide sufficient amounts of indicator at the distal end of the fiber. The prior art describes tw.o primary approaches to the problem. In the first of these, a porous substrate is used so that more surface area is available to which indicator moieties may bind, while in the second, as described, for example, in U.S. Patent No. 4,269,516 to Lubbers et al. , indicator is provided in solution form, which, solution is separated from the external environment by a membrane.
In the former procedure, a glass surface, for example the distal end of an optical fiber (or a glass bead which is subsequently attached to the fiber), is treated so that it is porous. It is then reacted with a suitable agent such as 3-aminopropyltriethoxy silane, and a series of reactions is carried out culminating in
the covalent attachment of a biologically and/or chemically active molecule, e.g., a fluorescent species, to the surface of the glass. These reactions may be represented as follows:
Glass Surface
Si-OH + EtO-
1.0 (Amino ropy1triethoxys!l ne)
Now representing the silanized glass (referred to also hereinafter as aminoalkyl glass or fiber) as
the next step is realized by reacting a fluorophore such as fluorescein isothiocyanate dissolved in an aprotic solvent with the aminoalkyl fiber
A dye immobilized on glass in this manner is pHsensitive in the physiological range and is relatively 0 stable. However, to achieve a satisfactory degree of response, it has been necessary to employ porous glass, that is, a special alkali borosilicate glass which has been heat treated to separate the glass into two phases. Typically, the phase which is rich in boric acid and 5 alkali is leached out with acid to leave a porous, high-
SUBSTITUTE SHEET
silica structure. The porous structure is then silanized and treated with the desired fluorescent species. The resulting treated glass is then attached to the distal end of an optical fiber. Porous glass provides more surface area available for silanizing and thus many more fluorescent molecules can be attached.
However, this technique is difficult to carry out. The most practical way to accomplish the intended result is to provide a bead of porous glass, s-ilanize it, react it with the fluorescent species, and then attach the glass bead to the distal end of an optical fiber. This attachment is difficult to effect because of the small size of the beads and the ease with which the pores in the glass are occluded. The technique of using a solution behind a membrane also suffers from serious drawbacks. Most importantly, the phenomena of concentration quenching comes into play, which severely limits the amount of dye that can be in close proximity to the end of the fiber. here fluorescent dyes are present in concentrations higher than about 10 JM, for example, concentration quenching occurs and results in a substantial loss of accuracy. Several processes are believed to be responsible for concentration quenching: (1) the increased probability of self-absorption at higher densities or concentrations of fluorophores; (2) formation of dimers or higher aggregates which are normally less fluorescent than the monomer; and (3) reaction of excited molecules with ground state molecules to form excimers, which again have emission spectra quite different from the monomer spectrum. See, e.g. , Guilbault, Practical Fluorescence, New York: Marcel Dekker, Inc., 1973. Similar problems arise in the case of absorbing dyes, where exciton interaction —
that is, interaction of dipoles on neighboring groups or molecules -is a factor at higher densities. See Birks, Photophysics of Aromatic Molecules, London: Wiley & Sons, 1970.
Disclosure of the Invention
Accordingly, it is an object of the present invention to overcome the aforementioned disadvantages of the prior art. It is another object of the invention to provide a fiber optic sensing device, comprising an optical fiber to one end of which is attached a polymer including a plurality of photoactive moieties spaced apart from each other at a preselected distance. It is still another object of the invention to provide such a device wherein the preselected distance is sufficient to minimize chemical or physical interaction between the photoactive moieties while optimizing the density thereof. It is yet another object of the invention to provide such a device wherein the photoactive moieties are covalently attached to the polymer through functional groups, such as through ethers, amides, or the like. It is a further object of the invention to provide such a device wherein the polymer is inherently fluorescent and includes at least one monomer unit which is itself photoactive.
It is still a further object of the invention to provide such a device wherein the inherently fluorescent polymer is quenchable by oxygen.
It is another object of the invention to provide a method of making fiber optic sensing devices, comprising attaching a plurality of monomeric units,
sequentially or as a preformed polymer, to an optical fiber tip, wherein at least a fraction of the monomeric units include photoactive moieties.
Additional objects, advantages and novel features of the invention will be set forth in part in the description which follows, and in part will become apparent to those skilled in the art on examination of the following, or may be learned by practice of the invention. In one aspect of the invention, a fiber optic sensing device is provided, comprising a fiber to one end of which is attached a polymer including a plurality of photoactive moieties. The photoactive moieties, which may be chromophores or lumophores are positioned along the polymer in such a way as to optimize the distance between the moieties. That is, chemical or physical interaction (e.g., concentration quenching, exciton interaction, steric interference) is minimized while the density of photoactive moieties is optimized. The high density of photoactive moieties achieved with the present invention provides an apparatus of substantially increased sensitivity.
The photoactive moieties may be attached as side groups, i.e. pendant to the main polymer chain. In such a case, each photoactive moiety is attached through one bond to the polymer backbone. In an alternative embodiment of the invention, in which the polymers are designated "inherently fluorescent," each photoactive moiety is in the backbone of the polymer, i.e. is bound to the remainder of the polymer through two or more bo ds.
In another aspect of the invention, a method of forming a fiber optic device is provided. The method involves either sequential attachment of labeled monomer
units to a riber optic tip, or, alternatively, attachment (covalent or otherwise) of a preformed polymer to a fiber optic tip; the preformed polymer may be labeled with photoactive moieties before attachment or after.
Potential uses of the device include, inter alia, use as a pH sensor, an oxygen sensor, an electrolyte sensor and a blood gas sensor.
Modes for Carrying Out the Invention
As used herein, "photoactive" moieties are molecular species which undergo a detectable change in electronic configuration in response to interaction with light. In the present- invention, the detectable change corresponds to one or more chemical or physiological parameters.
"Chromophores" are atoms or groups of atoms that serve as units in light absorption. For example, a chromophore includes the electronic substructure of a dye that absorbs light and thereby causes the material to be colored. A change in the environment of a chromophore can change the amount of light absorbed at a particular wavelength or the amount of light absorbed over a range of wavelengths. Examples of chromophores useful herein include cresol red and phenol red.
Suitable chromophores also include lumophores such as pyrenes , perylenes and their derivatives, and other paracyclic aromatic compounds.
"Lumophores" are atoms or groups of atoms that serve as units in light emission. All compounds which are capable of undergoing fluorescence or phosphorescence include a lumophore.
"Chemical or physical interaction" between the photoactive moieties of a polymer as used herein,
includes any interaction which (1) changes the electronic absorption spectrum obtained; (2) changes the electronic emission spectrum obtained; (3) changes the quantum yield of emission; (4) interferes with the accurate measurement of particular chemical or physiological parameters in a sample or (5) destabilizes the polymer structure.
A "substantially nonporous" material is a material having a porosity less than about 5%______ (vol./vol.).
"Inherently fluorescent" polymers, as used herein, are polymers formed from one or more photoactive monomer units, so that photoactive moieties are actually present within the polymer backbone so that each is bound to the remainder of the polymer chain through two or more bonds. This is in contrast to polymers in which the photoactive moieties are pendant to the polymer chain, where each is bound through only one bond to the remainder of the polymeric structure. The optical fiber of the present invention is typically between about' 50 μ and about 600 μ in diameter. In a preferred embodiment, the sensing device is useful for in vivo applications, for example to measure pH or oxygen concentration, in which case the diameter should be between about 50 μ and about 250 μ.
The fiber may be comprised of virtually any material so long as it is an optical conductor at the wavelengths of interest. For example, the fiber may be an organic material such as polymethylmethacrylate, polystyrene, polymethylphenyl siloxane or deuterated methyl methacrylate, or it may be an inorganic material such as glass. As noted earlier, successful use of substantially nonporous materials in this application —
- -
that is, to give a highly sensitive device -represents a significant improvement in the art.
The polymer which is attached to the fiber tip can be any stable polymer which can be derivatized so as to contain photoactive moieties. In addition, the polymer must be optically transparent both to exciting light, i.e. light directed through the fiber into the polymer, and to light emitted from the photoactive moieties attached thereto. Examples of polymers suitable for use herein include polyacrylamides, polylysines, polymethacrylates, polyurethanes, polyethers, and polysiloxanes such as polydimethyl and polymethylphenyl siloxane. The polymer chain is preferably less than about 200 monomer units in length, to ensure that substantially all photoactive moieties are maintained fairly close to the fiber tip.
The polymer may be attached to the fiber in a number of ways. In a preferred embodiment, the polymer is attached to the fiber covalently, typically through an aminoalkyl or other linking group (see Example 4). Suitable linking groups for glass and other fibers may be prepared from the following: diaminoalkanes, diaminoaryl compounds, diisothiocyanate-substituted aryl compounds, alkyldialdehydes, arylalkyl dialdehydes, arylalkyldiamines, alkyl dicarboxylic acid derivatives, arylalkyl decarboxylic acid derivatives, aryl dicarboxylic acid derivatives, alkyl diols, aryl diols, arylalkyl diols, and the like. Alternatively, the polymer may be attached to the polymer by adsorption or electrostatic interaction. In the latter case, the polymer is attached to the fiber by forming a salt of the polymer and a linking group on the fiber.
Unless the polymer is of the "inherently fluorescent" type, it must be provided with areas of
reactivity so that photoactive moieties can bind to the polymer backbone. The photoactive moieties may bind directly to the polymer backbone or may bind to the polymer through functional groups such as ethers, amines, amides including acrylamides, esters, alkyl moieties having from about two to about twenty-two carbon atoms, arylalkyl, etc.
Suitable photoactive moieties for use herein include chromophores, lumophores and combinations thereof. Chromophores include, for example, cresol red and phenol red. Lumophores include both fluorophores and phosphors. Examples of suitable fluorophores for use herein are fluorescein, perylene, pyrene and their derivatives. An examples of a suitable phosphor is rose bengal.
The reactive areas on the polymer are distributed along the polymer chain such that the photoactive moieties are spaced apart by at least about 4 angstroms, that is, the distance between photoactive moieties is determined by the relative frequency of the species in the polymer backbone. This frequency can be achieved during preparation of the polymer by diluting the photoactive moiety, i.e. by introducing a nonphotoactive comonomer during polymerization. Alternatively, with a preformed polymer having reactive functional groups to which' photoactive moieties will bind, the frequency of these moieties can be controlled by dilution during the reaction attaching the pendant moieties or by using a nonphotoactive moiety which can compete for binding to the functional groups on the polymer chain.
In some cases, depending on the environment and on the photoactive moieties selected, i.e. where there is a potential for interaction at a greater
distance, spacing of at least about 14 angstroms is preferred. The distance factor is key to the present invention, as the distance between photoactive moieties is carefully preselected so as to minimize chemical or physical interaction therebetween, while optimizing the density thereof close to the fiber tip. The inventors herein have demonstrated that, generally, the amount of light recaptured by the fiber from the photoactive moieties is strongly dependent on the density^of these groups. The strength of the signal ultimately generated is also dependent on the distance of the active moieties from the end of the fiber to these groups. Thus, and as accomplished by the present invention, it is advantageous to have a very high density of photoactive moieties at the fiber tip, as close to the end of the fiber as possible.
Generally, the fiber tip will be provided with a plurality of polymer chains all similarly bonded to the fiber. The spacing of photoactive moieties is then controlled not only along a single polymer chain but also between polymer chains. This is accomplished by controlling the number of polymer chains attached to a given area of the fiber tip, for example, by minimizing the number of attachment sites on the substrate or by reacting some of the sites with nonphotoactive polymer chains.
A primary use of the above-described sensing device, a preferred embodiment herein, is as a pH sensor. The fiber tip is placed into contact with a fluid or tissue, light is directed along the fiber toward the fiber-sample interface so that photoactive species are caused to fluoresce, and the ratio of fluorescence to reflected light is determined. This ratio changes with pH in a predictable fashion; thus, pH
may be determined by interpolating along a curve prepared from known pH/ratio points.
In an alternative embodiment of the invention, a fiber optic sensing device is provided which comprises a fiber tip to one end of which is attached an inherently fluorescent polymer. The polymer has as part of its molecular structure a chromophore, lumophore or the like, i.e. it is actually formed from one or more photoactive monomer units. Such a device provides a number of advantages: first, as with the embodiment described earlier, a very high optical density may be achieved without interfering effects such as concentration quenching. This allows for a very thin, yet highly absorbing, polymer layer. The thinness of the polymer layer causes an increase in the amount of emitted light which enters the fiber and allows, also, for a mechanically sturdy configuration. A second advantage is that with inherently fluorescent polymers, as with the polymers having pendant photoactive moieties, there is no separate dye which can migrate out of the polymer matrix. This promotes long- term stability of the device and allows the material to be used in contexts where dye toxicity could otherwise be a problem. The inherently fluorescent polymers are prepared by reaction of a photoactive moiety with a reactive monomeric species under polymerization conditions. The photoactive moieties may be introduced within a straight-chain polymer or as cross-linking functionalities in a cross-linked polymer (see Examples 16 and 17). Suitable photoactive moieties are as described earlier, and particularly suitable photoactive moieties for use in the inherently fluorescent polymer application are derivatives of pyrene. Preferred
polymers include polycarbonamides, polyurethanes, polyamides, polyimides, polyesters, and polysiloxanes such as polymethylsiloxanes, polymethylphenylsiloxanes and the like. A preferred use of the fiber optic sensing device which incorporates an inherently fluorescent polymer at the optical fiber tip is as an oxygen sensor. In such an application, the photoactive moieties incorporated in the polymer are fluorescent species which are quenchable by oxygen. The device is constructed so that fluorescence emission varies with (typically is inversely related to) the oxygen concentration of its environment, which may be gaseous or aqueous (e.g., blood). The present invention also encompasses a method of attaching labeled polymeric species — inherently fluorescent or otherwise -to a fiber optic tip. In one embodiment, the method involves sequential addition of monomer or oligomer species to a linking group present on the fiber. At least a fraction of these species is photoactive. In another embodiment, the method involves attachment of a preformed polymer to the fiber tip, which polymer may be labeled with photoactive moieties prior to attachment or after. The attachment of the preformed polymer may be covalent, or it may be effected by adsorption or electrostatic binding.
In developing the device of the present invention, the inventors herein discovered a new and useful fluorescent compound attachable in a simple, one- step reaction to a polymer chain. This compound is a fluorophore having an acryloyl substituent on an aromatic ring, and in one embodiment is given by the following structure
It will be apparent to those skilled in the art that the compound may be further substituted on the aromatic rings, e.g., with halogen substituents or lower alkyl or alkoxy groups. Such a fluorescent acrylamide is then polymerizable (with or without nonfluorescent acrylamide monomers) in the presence of a catalyst such as 2,2'- azobisisobutyronitrile to form fluorescent polymers having the structure
In the above structure, x and y represent the number of nonfluorescent and fluorescent monomer units in the polymeric structure, respectively, and are selected so as to provide the requisite spacing between photoactive moieties. Typically, x and y each range from about 1 to about 200, and the sum of x and y is also, typically, less than about 200. F represents the major portion of the molecule given by
Pσlyirer
It is to be understood that while the invention has been described in conjunction with specific embodiments thereof, that the foregoing description as well as the examples which follow are intended to illustrate and not limit the scope of the invention, which is defined by the appended claims. Other aspects, advantages and modifications within the scope of the invention will be apparent to those skilled in the art to which the invention pertains.
Example 1 Preparation of Acid Glass
In 1. and generally hereinafter, the group -NH- is derived from the primary amino group of the aminoalkyl glass. The acid glass 1, was prepared as follows:
Succinic anhydride (Aldrich, 3.0g) was dissolved in anhydrous tetrahydrofuran (THF) (100 ml). Silanized glass, i.e. aminoalkyl glass (Pierce Chemical, 1 g) , was added and the solution was refluxed for four hours. The glass was then washed, with 30 ml of acetone and dried in vacuo at 25°C for thirty minutes.
Example 2 Preparation of Acid Chloride Glass The acid glass of Example 1 was treated with SOCI2 as follows: A 10% solution of thionyl chloride (Aldrich) in dry chloroform was prepared. 0.5 g of acid glass was added to 20 ml of the thionyl chloride solution and refluxed for four hours. The glass was then rinsed with chloroform and dried under vacuum for thirty minutes. o o o
If soci. /( « f— NH-C(CH ) ;-OH -NH-C CCH ) C-Cl
Example 3 Preparat ion of N-Hydroxy Succinic (NHS) Glass The acid chloride glass of Example 2 was reacted with N-hydroxysuccinimide as follows : o
The reaction was carried out as follows: 3.8 g of N-hydroxysuccinimide (Aldrich) and 4.45 ml of triethylamine (Aldrich) were added to 50 ml of anhydrous stirred chloroform. This solution was then cooled to 0°C. Acid chloride glass was added and the suspension was stirred for an additional forty minutes. The temperature of the suspension was maintained at 0°C during this stirring period. The glass was filtered,
and the beads were washed with chloroform and dried in vacuo at 25°C for thirty minutes.
The purpose of treating the acid chloride glass with N-hydroxy succinimide was to protect the treated glass from hydrolysis in subsequent treatment.
Example 4 Treatment of NHS Glass with Triethylenetetramine (TET) The NHS glass from Example 3 is added with stirring to a mixture of 50 ml of anhydrous chloroform and 8 ml of triethylenetetramine (Aldrich) . The glass mixture is stirred for one hour, and the beads are then filtered, washed with chloroform and dried in vacuo at 25°C for thirty^.minutes. The reaction may be represented as
2C-NH(CH2)2NH(CH2)2mi(CH2)2 H2
The treated, aminoalkyl glass 4. has numerous nitrogen functionalities to which fluorescent groups may be attached.
-
Example 5
Glutaraldehyde glass (represented by structure 5_) was prepared as follows. A solution of 25% glutaraldehyde was diluted to 2.5% in 0.1 M Na3P04, pH 7, to._50 ml. The aminoalkyl glass prepared in the preceding Example was immersed for one hour and then rinsed with deionized water.
Example 6 Glutaryl Chloride (qlut-Cl) Glass
o o o o
-NH + CI-Cιι(CH2)3Cιι-C1 -> (—NH-C-(CH2 ,))3,Cc :---cCc1ι
2.5 ml of glutaryl dichloride [source?] was diluted to 10 ml in anhydrous THF. Aminoalkyl glass (400 mg) as prepared in Example 4 was immersed for one hour in this solution and then'rinsed with dry acetone.
Example 7 Attachment of a Fluorescent Polymer to Aminoalkyl Glass (a) Preparation of N-substituted acrylamide derivative of fluorescein: Two hundred mg of fluorescein amine 1_ (Aldrich) were dissolved in a mixture of 1.0 ml anhydrous dimethyl formamide and 25 ml anhydrous chloroform, followed b -addition of 0.56 ml of
acryloyl chloride (Aldrich) . The reaction mixture was stirred and maintained at a temperature of 25°C. After sixty minutes, the pH of the reaction mixture was found to be about 3, indicating formation of HC1 and thus reaction of the fluorescein amine with the acryloyl chloride. The solvent was stripped off in vacuo leaving an amber oil. The product was presumed to have the formula 8.
(b) Copolymerization of Acrylamide and 13 with Aminoalkyl Glass: 400 ml of 8> were dissolved in 30 ml tetrahydrofuran together with 1 g acrylamide and 10 mg of 2,2' -azobisisobutryo nitrile (AIBN) (Polysciences) as catalyst. Aminoalkyl glass in the form of a fiber tip (prepared as in Example 4) was activated in 1 ml acryloyl chloride for one hour, immersed in the acrylamide solution, and heated at 50°C for twenty hours. A copolymer of 8. and acrylamide linked to the aminoalkyl glass resulted in the structure given by 9_, wherein x and y indicate the numbers of the respective monomer units.
The monomer units in all likelihood occur in some more or less random order. The pendant group F is derived from 8_; i.e., it is the group 8a.
Polymer
Chain
8a
(c) Testing of Fiber Tip _: The fiber tip 9_ was tested as a pH sensor by immersion first in a buffer solution at pH 6.8 and then in a second buffer solution at pH 7.8. The ratio of the intensity of light resulting from fluorescence to that of the reflected light was found to be 0.81 at the lower pH and 1.1 at the higher pH.
(d) Comparison with a Porous Glass Bead to which FITC is Directly Attached: A porous glass bead (OD = 177 μ, average pore size = 500), was glued to the end of an optical fiber and activated by treatment with a inopropyl triethoxy silane (Petrarch) 10 wt.% aqueous solution, pH 3.45, treatment time sixty minutes). Due to the porous nature of the glass, many aminoalkyl groups were presumed attached. The fiber was then rinsed in distilled water and air dried at 90°C.
Ten mg fluorescein isothiocyanate (FITC) (Molecular Probes) was dissolved in 5 ml acetone and the tip of the fiber described above was immersed in the solution for sixty minutes at room temperature. The fiber was rinsed with acetone, and tested as in Example 7(c) above. The ratio of the intensity of light
resulting from fluorescence to that of the reflected light was found to be 0.54 at pH 6.8 and 1.29 at pH 7.8.
In example 7(c) the glass to which the fluorescent species was attached was nonporous. It was treated to append aminoalkyl groups but eliminated the tedious step of attachment of a porous bead.
The polymeric technique thus achieves a result which is similar to that achieved with porous glass, the porous glass route, however, being less desirable due to attachment problems.
Example 8 Glass Fiber Treated with FITC By way of further comparison, aminoalkyl glass fiber as in Example 7(b) was treated as follows: The fiber was soaked first in a solution containing 5 mg of FITC in 100 ml of acetone for one hour and then in deionized water for one hour. No observable fluorescent pH effect could be measured using the procedure of Example 7(c) .
Example 9 Glass Fiber Treated with High Molecular Weight Polylysine and FITC A glutaraldehyde-treated aminoprόpyl glass fiber (prepared as in Example 5) was placed in a solution of 4 mg of 540,000 D average molecular weight (Mv) polylysine in 4 ml of deionized water and allowed to soak for one hour at 23°C. The fiber was then allowed to stand in a deionized water wash for one hour after which time it was treated with a. solution of 5 mg FITC/100 ml acetone for one hour. Its pH-dependent fluorescence was measured. At pH 6.8 the ratio of
fluorescence to reflected light was 0.11, and at pH 7.4 it was 0.21.
Example 10 Glass Fiber Treated with
Low Molecular Weight Polylysine and FITC
A glutaraldehyde-treated aminopropyl glass fiber (prepared as in Example 5) was placed in a solution of 4 mg of 4,000 D average molecular_weight (Mw) polylysine (Polysciences) in deionized water and allowed to soak for one hour at 23°C. The fiber was then placed in a deionized water wash for one hour and immediately treated with a solution of 5 mg FITC/100 ml acetone for one hour. The fiber was then rewashed in deionized water for one hour and its pH-dependent fluorescence measured. The ratio of fluorescent to reflected light at pH 6.8 was 0.35 and at pH 7.4 was 0.52.
From the results of Examples 9 and 10, it may be concluded that the polylysine was attached covalently to the aminopropyl glass and that FITC molecules were attached covalently as pendant groups to the polylysine chains.
Example "11
Electrostatically Bound Fluorescent Polymer A fluorescent polymer is prepared having pendant fluorescent groups and pendant carboxyl groups. The polymer is caused to react with activated glass (i.e., the aminoalkyl glass of Example 4) having basic groups, resulting in the following:
where C represents one of a multiplicity of activated groups on glass, D represents a repeating unit^ in the polymer described above (wherein F represents a pendant fluorescent group) and E represents the resulting salt. This may be accomplished by reacting aminopropyl glass C with a fluorescently labeled polymer D. The resulting ionic salt bridges are then stabilized by drying the product in vacuo.
Example 12 Adsorption of a Fluorescent Polymer on an Aminoalkyl Glass Surface A polymer having pendant fluorescent groups is prepared, e.g., a copolymer of acrylamide and the acryloyl derivative 8. of fluorescein prepared as described in Example 7(b) but in the absence of aminopropyl glass.
An aminopropyl glass fiber tip is immersed in a 0.01 molar aqueous solution of this copolymer for two days. The fiber is then rinsed with chloroform for three days to remove the nonadsorbed polymer and is dried in vacuo at 25°C for one hour.
Example 13 Immunoassay Embodiment In the enzyme-linked immunoassay (ELISA) technique, an enzyme is immobilized on the surface of a
test tube or cuvette. See, for example, Chang, Biochemical Applications of Immobilized Enzymes and Proteins, Vol. 2, Plenum Press, 1977, Chapters 30-33. This technique is improved by the amplification technique of the present invention as follows.
An acetaminophen-specific antibody is treated with a one hundredfold molecular excess of acryloyl chloride, and taken into solution with deionized water to which a twofold quantity of acrylamide hasJseen added together with a suitable amount of AIBN. An aminopropyl glass optical fiber is treated with an excess of acryloyl chloride, washed, added to the acrylated antibod /AIBN solution and warmed at a sufficient temperature to promote graft polymerization of the antibody derivative to the silica surface without denaturing the protein.
Next, an acetaminophen-alkaline phosphatase conjugate is incubated with the immobilized antibody, filtered, washed, and the unknown quantity of acetaminophen-analyte added. The residual bound enzyme activity is then measured and the quantity of acetaminophen calculated.
In the examples above, fluorescent species such as compound 8. and FITC are used and the ratio of fluorescent light to reflected light is measured. As noted, active species other than fluorescent species may be used. For example, phenol red, cresol red and neutral red may be used. These dyes change color with pH. These dyes have functional groups which are capable of reacting with functional groups of monomers and polymers and can be incorporated in polymer molecules formed in situ or in preformed polymers. The following example will serve for purposes of illustration.
Example 14 Incorporation of Cresol Red in a Polymer Chain
Cresol red (0.038 g) is dissolved in 30 ml of chloroform. N-Bromosuccinimide (0.017 g) is added along with benzoyl peroxide (0.005 g) . The solution is refluxed for one hour and allowed to cool to room temperature. The succinimide is removed by filtration and the bromomethyl cresol red is crystallized from acetic acid. To a solution of dimethylformamide (25 ml) containing the bromoethyl cresol red derivative (0.025 g) is added a tenfold molar excess of allylamine. The reaction mixture is heated with stirring at 50°C under argon in the dark for six hours. The reaction mixture is then cooled to room temperature and the solvent removed under high vacuum.
The reaction product of the bromomethyl cresol red and allylamine is dissolved in DMF (50 ml) together with 1 g of acrylamide and lOmg 2,2'- azobisisobutyronitrile. Into this solution is placed aminoalkyl glass, in the form of a fiber tip,.which has been previously treated with a mixture of acryloyl chloride (0.10 g), pyridine (20 ml), and N,-N'- dimethylamino pyridine (0.010 g) for two hours. The resulting amidated fiber is heated with the bromomethyl cresol red allylamine product and acrylamide is formed at the end of the fiber.
A simplified reaction scheme is as follows:
Δ
Cresol ced + N-bromosuccinimide ^
CH 2-NH-CH 2-CH=CH
I I
II +• C.H2=CH-C0-NH H-CO-CH=CH . Ill
In II, the remainder of the molecule is as in I. Ill is the glass fiber to which a copolymer of acrylamide and II is covalently attached.
It will be apparent that a new and useful method of making optical fibers having enhanced capacity for fluorescence, etc., and new and useful optical fibers having such enhanced properties have been provided.
Example 15 Preparation and Use of a Fluorescent Polycarbonamide The cleaved end of a silica core step index, glass-on-glass optical fiber, was treated with a 10% solution of -a inopropyltriethoxysilane in toluene overnight at 100°C. The well-rinsed fiber was inserted through a condenser attached to a flask charged with diaminodecane (137 mg) , sebacoyl chloride (190 mg) , perylene-3,9-dicarboxylic acid chloride (3 mg), triethylamine (107 mg) and chloroform (100 ml). The solution was refluxed, and a fluorescent polymer was
allowed to form and grow from the end of the fiber. After one hour, the fiber was removed and rinsed.
When properly filtered blue light was launched into the fiber, fluorescent green light could be collected (after it had traveled back along the optical fiber) by use of a dichroic mirror and appropriate optical filter. The intensity of the green light was inversely related to the amount of oxygen in the environment surrounding the polymer tip. This, was observed when the polymer tip was in a gaseous or aqueous environment.
Example 16 Preparation of a Fluorescent Silicone A sample of the diallyl ester of perylene-3,9- dicarboxylic acid (3 mg) was mixed with a 1 ml sample of a methylhydrodimethyl siloxane copolymer, and a small amount of platinum catalyst was added. Upon heating, a homogeneous fluorescent green or yellow material was formed. The intensity of the fluorescence emission of this material varies with the oxygen content of its environment. The material may be mounted at the end of a fiber by a mechanical means such as a capillary, and the fiber can then be used as an optrode. For example, a piece of 240 μ ID Celgard™ hollow porous fiber was slipped over the end of a 140 μ plastic-clad silica optical fiber. A sample of the partially cured polymer was then wicked into the hollow fiber. The fiber was then heated while the polymer cured to form a solid material. The fluorescence emission from this tip, when monitored as described in Example 1, was inversely related to the oxygen concentration of its environment. This was observed in both gaseous and aqueous environments, including canine
blood. This behavior could still be observed when the optical fiber had been subjected to steam sterilization,
Claims
1. A fiber optic sensing device for measuring a chemical or physiological parameter of a body fluid or tissue, comprising an optical fiber to one end of which is attached a polymer including a plurality of photoactive moieties selected from the group consisting of chromophores and lumophores, the photoactive moieties spaced apart from each other at a distance sufficient to minimize chemical or physical interaction therebetween while optimizing the density thereof.
2. The device of claim 1, wherein the polymer is covalently bound to the fiber.
3. The device of claim 2, wherein the polymer is covalently bound to the fiber through an aminoalkyl linking group.
4. The device of claim 1, wherein the polymer is ionically bound to the fiber.
5. The method of claim 1, wherein the polymer is attached to the fiber by adsorption,
6. The device of claim 1, wherein the polymer is selected from the group consisting of polyacrylamides, polylysines, polymethacrylates, polyurethanes, polyethers, polysiloxanes and mixtures thereof.
7. The device of claim 1, wherein the distance between the photoactive moieties is such that concentration quenching or exciton interaction is substantially prevented.
8. The device of claim 1, wherein a plurality of polymer chains are attached to the fiber, and wherein the chains are spaced apart so that the distance between active moieties on different chains is greater than about 4 angstroms.
9. The device of claim 1, wherein the polymer is an inherently fluorescent polymer.
10. An inherently fluorescent polymer including a plurality of photoactive moieties selected from the group consisting of chromophores, lumophores and combinations thereof, the photoactive moieties spaced apart from each other at a distance sufficient to minimize chemical or physical interaction therebetween while optimizing the density thereof.
11. A polymer having the structure
where x represents the number of nonfluorescent monomer units, y represents the number of fluorescent monomer units, and F represents a fluorophore moiety having the structure
10
15
20
25
30
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US433987A | 1987-01-16 | 1987-01-16 | |
| US004,339 | 1987-01-16 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO1988005533A1 true WO1988005533A1 (en) | 1988-07-28 |
Family
ID=21710282
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US1988/000041 WO1988005533A1 (en) | 1987-01-16 | 1988-01-12 | Amplification of signals from optical fibers |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO1988005533A1 (en) |
Cited By (32)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2650076A1 (en) * | 1989-07-20 | 1991-01-25 | Commissariat Energie Atomique | FIBER OPTIC ACTIVE CHEMICAL SENSOR AND PROCESS FOR PRODUCING THE SAME |
| US5015715A (en) * | 1989-08-16 | 1991-05-14 | Puritan-Bennett Corporation | Method for bonding an analyte-sensitive dye compound to an addition-cure silicone |
| EP0448052A2 (en) * | 1990-03-20 | 1991-09-25 | Minnesota Mining And Manufacturing Company | Method for making a gas sensing element |
| US5182353A (en) * | 1989-08-16 | 1993-01-26 | Puritan-Bennett Corporation | Method for bonding an analyte-sensitive dye compound to an addition-cure silicone |
| US5219527A (en) * | 1989-08-16 | 1993-06-15 | Puritan-Bennett Corporation | Sensor element and method for making the same |
| US5262192A (en) * | 1992-04-27 | 1993-11-16 | Puritan-Bennett Corporation | Method and compositions for manufacture of chemical sensors |
| US5266271A (en) * | 1992-05-22 | 1993-11-30 | Puritan-Bennett Corporation | Microsensor copolymer and method of manufacture |
| US5271073A (en) * | 1990-08-10 | 1993-12-14 | Puritan-Bennett Corporation | Optical fiber sensor and method of manufacture |
| US5277872A (en) * | 1990-10-16 | 1994-01-11 | Puritan-Bennett Corporation | Optical fiber pH microsensor and method of manufacture |
| US5296381A (en) * | 1991-08-08 | 1994-03-22 | Minnesota Mining & Manufacturing Co. | Sensing elements and methods for making and using same |
| US5326531A (en) * | 1992-12-11 | 1994-07-05 | Puritan-Bennett Corporation | CO2 sensor using a hydrophilic polyurethane matrix and process for manufacturing |
| US5330718A (en) * | 1989-08-16 | 1994-07-19 | Puritan-Bennett Corporation | Sensor element and method for making the same |
| US5409666A (en) * | 1991-08-08 | 1995-04-25 | Minnesota Mining And Manufacturing Company | Sensors and methods for sensing |
| US5453248A (en) * | 1992-03-09 | 1995-09-26 | Optical Sensors Incorporated | Cross-linked gas permeable membrane of a cured perfluorinated urethane polymer, and optical gas sensors fabricated therewith |
| WO1995029959A1 (en) * | 1994-05-02 | 1995-11-09 | Ciba-Geigy Ag | Polymerisable compositon and the use thereof |
| WO1995030148A1 (en) * | 1994-05-02 | 1995-11-09 | Ciba-Geigy Ag | OPTICAL SENSOR SYSTEM FOR DETERMINING pH VALUES AND IONIC STRENGTHS |
| US5536783A (en) * | 1993-06-10 | 1996-07-16 | Optical Sensors Incorporated | Fluorescent polymers useful in conjunction with optical PH sensors |
| US5808307A (en) * | 1996-02-07 | 1998-09-15 | Director-General Of Agency Of Industrial Science And Technology | Method of detecting ultraviolet laser beam and method of recording information |
| WO2007059449A2 (en) * | 2005-11-10 | 2007-05-24 | Beckman Coulter, Inc. | Covalently attached nile blue derivatives for optical sensors |
| US8222047B2 (en) | 2008-09-23 | 2012-07-17 | Quanterix Corporation | Ultra-sensitive detection of molecules on single molecule arrays |
| US8236574B2 (en) | 2010-03-01 | 2012-08-07 | Quanterix Corporation | Ultra-sensitive detection of molecules or particles using beads or other capture objects |
| US8242203B2 (en) | 2005-11-10 | 2012-08-14 | Eric Bakker | Covalently attached nile blue derivatives for optical sensors |
| US8415171B2 (en) | 2010-03-01 | 2013-04-09 | Quanterix Corporation | Methods and systems for extending dynamic range in assays for the detection of molecules or particles |
| US8460879B2 (en) | 2006-02-21 | 2013-06-11 | The Trustees Of Tufts College | Methods and arrays for target analyte detection and determination of target analyte concentration in solution |
| US9110025B2 (en) | 2010-03-01 | 2015-08-18 | Quanterix Corporation | Methods and systems for extending dynamic range in assays for the detection of molecules or particles |
| US9678068B2 (en) | 2010-03-01 | 2017-06-13 | Quanterix Corporation | Ultra-sensitive detection of molecules using dual detection methods |
| US9809838B2 (en) | 2007-08-30 | 2017-11-07 | Trustees Of Tufts College | Methods for determining the concentration of an analyte in solution |
| US9932626B2 (en) | 2013-01-15 | 2018-04-03 | Quanterix Corporation | Detection of DNA or RNA using single molecule arrays and other techniques |
| US9952237B2 (en) | 2011-01-28 | 2018-04-24 | Quanterix Corporation | Systems, devices, and methods for ultra-sensitive detection of molecules or particles |
| US10393759B2 (en) | 2011-04-12 | 2019-08-27 | Quanterix Corporation | Methods of determining a treatment protocol for and/or a prognosis of a patient's recovery from a brain injury |
| WO2020160835A1 (en) * | 2019-02-08 | 2020-08-13 | Universite De Franche-Comte | Method of manufacture of an optical fibre-based ph measurement sensor |
| US11237171B2 (en) | 2006-02-21 | 2022-02-01 | Trustees Of Tufts College | Methods and arrays for target analyte detection and determination of target analyte concentration in solution |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3612866A (en) * | 1969-07-08 | 1971-10-12 | Brian Stevens | Instrument for determining oxygen quantities by measuring oxygen quenching of fluorescent radiation |
| US4018884A (en) * | 1975-06-26 | 1977-04-19 | Hoffmann-La Roche Inc. | Fluorogenic materials and labeling techniques |
| US4200110A (en) * | 1977-11-28 | 1980-04-29 | United States Of America | Fiber optic pH probe |
| US4577109A (en) * | 1980-10-06 | 1986-03-18 | Regents Of The University Of California | Remote multi-position information gathering system and method |
-
1988
- 1988-01-12 WO PCT/US1988/000041 patent/WO1988005533A1/en unknown
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3612866A (en) * | 1969-07-08 | 1971-10-12 | Brian Stevens | Instrument for determining oxygen quantities by measuring oxygen quenching of fluorescent radiation |
| US4018884A (en) * | 1975-06-26 | 1977-04-19 | Hoffmann-La Roche Inc. | Fluorogenic materials and labeling techniques |
| US4200110A (en) * | 1977-11-28 | 1980-04-29 | United States Of America | Fiber optic pH probe |
| US4577109A (en) * | 1980-10-06 | 1986-03-18 | Regents Of The University Of California | Remote multi-position information gathering system and method |
Cited By (63)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0410851A1 (en) * | 1989-07-20 | 1991-01-30 | Commissariat A L'energie Atomique | Fiberoptic active chemical sensor and fabrication method of the same |
| US5028395A (en) * | 1989-07-20 | 1991-07-02 | Commissariat A L'energie Atomique | Active chemical sensor with optical fiber and its production process |
| FR2650076A1 (en) * | 1989-07-20 | 1991-01-25 | Commissariat Energie Atomique | FIBER OPTIC ACTIVE CHEMICAL SENSOR AND PROCESS FOR PRODUCING THE SAME |
| US5330718A (en) * | 1989-08-16 | 1994-07-19 | Puritan-Bennett Corporation | Sensor element and method for making the same |
| US5015715A (en) * | 1989-08-16 | 1991-05-14 | Puritan-Bennett Corporation | Method for bonding an analyte-sensitive dye compound to an addition-cure silicone |
| US5182353A (en) * | 1989-08-16 | 1993-01-26 | Puritan-Bennett Corporation | Method for bonding an analyte-sensitive dye compound to an addition-cure silicone |
| US5219527A (en) * | 1989-08-16 | 1993-06-15 | Puritan-Bennett Corporation | Sensor element and method for making the same |
| EP0448052A2 (en) * | 1990-03-20 | 1991-09-25 | Minnesota Mining And Manufacturing Company | Method for making a gas sensing element |
| EP0448052A3 (en) * | 1990-03-20 | 1992-02-26 | Minnesota Mining And Manufacturing Company | Gas sensing element and method for making and using same |
| US5271073A (en) * | 1990-08-10 | 1993-12-14 | Puritan-Bennett Corporation | Optical fiber sensor and method of manufacture |
| US5378432A (en) * | 1990-10-16 | 1995-01-03 | Puritan-Bennett Corporation | Optical fiber pH microsensor and method of manufacture |
| US5277872A (en) * | 1990-10-16 | 1994-01-11 | Puritan-Bennett Corporation | Optical fiber pH microsensor and method of manufacture |
| US5498549A (en) * | 1991-08-08 | 1996-03-12 | Minnesota Mining And Manufacturing Company | Sensors and methods for sensing |
| US5409666A (en) * | 1991-08-08 | 1995-04-25 | Minnesota Mining And Manufacturing Company | Sensors and methods for sensing |
| US5296381A (en) * | 1991-08-08 | 1994-03-22 | Minnesota Mining & Manufacturing Co. | Sensing elements and methods for making and using same |
| US5631340A (en) * | 1992-03-09 | 1997-05-20 | Optical Sensors Incorporated | Cross-linked gas permeable membrane of a cured perfluorinated urethane polymer, and optical gas sensors fabricated therewith |
| US5453248A (en) * | 1992-03-09 | 1995-09-26 | Optical Sensors Incorporated | Cross-linked gas permeable membrane of a cured perfluorinated urethane polymer, and optical gas sensors fabricated therewith |
| US5262192A (en) * | 1992-04-27 | 1993-11-16 | Puritan-Bennett Corporation | Method and compositions for manufacture of chemical sensors |
| US5326585A (en) * | 1992-04-27 | 1994-07-05 | Puritan-Bennett Corporation | Method and compositions for manufacture of chemical sensors |
| US5266271A (en) * | 1992-05-22 | 1993-11-30 | Puritan-Bennett Corporation | Microsensor copolymer and method of manufacture |
| US5326531A (en) * | 1992-12-11 | 1994-07-05 | Puritan-Bennett Corporation | CO2 sensor using a hydrophilic polyurethane matrix and process for manufacturing |
| US5536783A (en) * | 1993-06-10 | 1996-07-16 | Optical Sensors Incorporated | Fluorescent polymers useful in conjunction with optical PH sensors |
| WO1995029959A1 (en) * | 1994-05-02 | 1995-11-09 | Ciba-Geigy Ag | Polymerisable compositon and the use thereof |
| WO1995030148A1 (en) * | 1994-05-02 | 1995-11-09 | Ciba-Geigy Ag | OPTICAL SENSOR SYSTEM FOR DETERMINING pH VALUES AND IONIC STRENGTHS |
| EP0881241A2 (en) * | 1994-05-02 | 1998-12-02 | Novartis AG | Copolymers and dyes for optical pH sensors |
| US5852126A (en) * | 1994-05-02 | 1998-12-22 | Novartis Ag | Polymerisable composition and the use thereof |
| EP0881241A3 (en) * | 1994-05-02 | 1998-12-23 | Novartis AG | Copolymers and dyes for optical pH sensors |
| US5922612A (en) * | 1994-05-02 | 1999-07-13 | Novartis Corporation | Optical sensor system for determining pH values and ionic strengths |
| US5808307A (en) * | 1996-02-07 | 1998-09-15 | Director-General Of Agency Of Industrial Science And Technology | Method of detecting ultraviolet laser beam and method of recording information |
| WO2007059449A3 (en) * | 2005-11-10 | 2007-08-09 | Beckman Coulter Inc | Covalently attached nile blue derivatives for optical sensors |
| US8242203B2 (en) | 2005-11-10 | 2012-08-14 | Eric Bakker | Covalently attached nile blue derivatives for optical sensors |
| WO2007059449A2 (en) * | 2005-11-10 | 2007-05-24 | Beckman Coulter, Inc. | Covalently attached nile blue derivatives for optical sensors |
| US11874279B2 (en) | 2006-02-21 | 2024-01-16 | Trustees Of Tufts College | Methods and arrays for target analyte detection and determination of target analyte concentration in solution |
| US11237171B2 (en) | 2006-02-21 | 2022-02-01 | Trustees Of Tufts College | Methods and arrays for target analyte detection and determination of target analyte concentration in solution |
| US8460879B2 (en) | 2006-02-21 | 2013-06-11 | The Trustees Of Tufts College | Methods and arrays for target analyte detection and determination of target analyte concentration in solution |
| US8460878B2 (en) | 2006-02-21 | 2013-06-11 | The Trustees Of Tufts College | Methods and arrays for detecting cells and cellular components in small defined volumes |
| US8492098B2 (en) | 2006-02-21 | 2013-07-23 | The Trustees Of Tufts College | Methods and arrays for target analyte detection and determination of reaction components that affect a reaction |
| US10261089B2 (en) | 2006-02-21 | 2019-04-16 | Trustees Of Tufts College | Methods and arrays for target analyte detection and determination of target analyte concentration in solution |
| US9395359B2 (en) | 2006-02-21 | 2016-07-19 | Trustees Of Tufts College | Methods and arrays for target analyte detection and determination of target analyte concentration in solution |
| US9809838B2 (en) | 2007-08-30 | 2017-11-07 | Trustees Of Tufts College | Methods for determining the concentration of an analyte in solution |
| US8222047B2 (en) | 2008-09-23 | 2012-07-17 | Quanterix Corporation | Ultra-sensitive detection of molecules on single molecule arrays |
| US8846415B2 (en) | 2008-09-23 | 2014-09-30 | Quanterix Corporation | Ultra-sensitive detection of molecules on single molecule arrays |
| US9846155B2 (en) | 2010-03-01 | 2017-12-19 | Quanterix Corporation | Methods and systems for extending dynamic range in assays for the detection of molecules or particles |
| US10989713B2 (en) | 2010-03-01 | 2021-04-27 | Quanterix Corporation | Methods and systems for extending dynamic range in assays for the detection of molecules or particles |
| US9678068B2 (en) | 2010-03-01 | 2017-06-13 | Quanterix Corporation | Ultra-sensitive detection of molecules using dual detection methods |
| US9482662B2 (en) | 2010-03-01 | 2016-11-01 | Quanterix Corporation | Ultra-sensitive detection of molecules or particles using beads or other capture objects |
| US9310360B2 (en) | 2010-03-01 | 2016-04-12 | Quanterix Corporation | Ultra-sensitive detection of molecules or particles using beads or other capture objects |
| US8236574B2 (en) | 2010-03-01 | 2012-08-07 | Quanterix Corporation | Ultra-sensitive detection of molecules or particles using beads or other capture objects |
| US9110025B2 (en) | 2010-03-01 | 2015-08-18 | Quanterix Corporation | Methods and systems for extending dynamic range in assays for the detection of molecules or particles |
| US11619631B2 (en) | 2010-03-01 | 2023-04-04 | Quanterix Corporation | Ultra-sensitive detection of molecules or particles using beads or other capture objects |
| US9551663B2 (en) | 2010-03-01 | 2017-01-24 | Quanterix Corporation | Methods and systems for extending dynamic range in assays for the detection of molecules or particles |
| US10725032B2 (en) | 2010-03-01 | 2020-07-28 | Quanterix Corporation | Ultra-sensitive detection of molecules or particles using beads or other capture objects |
| US8415171B2 (en) | 2010-03-01 | 2013-04-09 | Quanterix Corporation | Methods and systems for extending dynamic range in assays for the detection of molecules or particles |
| US9952237B2 (en) | 2011-01-28 | 2018-04-24 | Quanterix Corporation | Systems, devices, and methods for ultra-sensitive detection of molecules or particles |
| US11112415B2 (en) | 2011-01-28 | 2021-09-07 | Quanterix Corporation | Systems, devices, and methods for ultra-sensitive detection of molecules or particles |
| US11275092B2 (en) | 2011-04-12 | 2022-03-15 | Quanterix Corporation | Methods of determining a treatment protocol for and/or a prognosis of a patient's recovery from a brain injury |
| US10393759B2 (en) | 2011-04-12 | 2019-08-27 | Quanterix Corporation | Methods of determining a treatment protocol for and/or a prognosis of a patient's recovery from a brain injury |
| US10640814B2 (en) | 2013-01-15 | 2020-05-05 | Quanterix Corporation | Detection of DNA or RNA using single molecule arrays and other techniques |
| US9932626B2 (en) | 2013-01-15 | 2018-04-03 | Quanterix Corporation | Detection of DNA or RNA using single molecule arrays and other techniques |
| FR3092666A1 (en) * | 2019-02-08 | 2020-08-14 | Universite De Franche-Comte | A method of manufacturing an optical fiber-based pH measurement sensor. |
| WO2020160835A1 (en) * | 2019-02-08 | 2020-08-13 | Universite De Franche-Comte | Method of manufacture of an optical fibre-based ph measurement sensor |
| US20220128478A1 (en) * | 2019-02-08 | 2022-04-28 | Universite De Franche-Comte | Method of manufacture of an optical fibre-based ph measurement sensor |
| US11846589B2 (en) | 2019-02-08 | 2023-12-19 | Universite De Franche-Comte | Method of manufacture of an optical fibre-based pH measurement sensor |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US5700897A (en) | Method for making fluorescent polymers | |
| WO1988005533A1 (en) | Amplification of signals from optical fibers | |
| US5019350A (en) | Fluorescent polymers | |
| US5028395A (en) | Active chemical sensor with optical fiber and its production process | |
| US5405975A (en) | Fluorescent ion-selective diaryldiaza crown ether conjugates | |
| CA2415338C (en) | Detection of analytes in aqueous environments | |
| US4712865A (en) | Dye containing silicon polymer composition | |
| EP0455730B1 (en) | Material and method for oxygen sensing | |
| JPH09502256A (en) | Optical sensor for measuring pH in liquids | |
| JP2010535903A (en) | HPTS-mono and bis-Cys-MA polymerizable fluorescent dyes used in analyte sensors | |
| EP0862741B1 (en) | Optical sensor system for the determination of ph values independently of ionic strength | |
| JPH0216435A (en) | Composition, apparatus and method for detection of ion component | |
| JP3810260B2 (en) | Method for determination of triaza-cryptands and alkali ions | |
| EP0205232A1 (en) | Amplification of signals from optical fibers | |
| Marks et al. | An innovative strategy for immobilization of receptor proteins on to an optical fiber by use of poly (pyrrole–biotin) | |
| US5015715A (en) | Method for bonding an analyte-sensitive dye compound to an addition-cure silicone | |
| JPH06192213A (en) | Amino substituted phenol red as ph indicator and preparation | |
| Wang et al. | Binding characteristics of avidin and surface immobilized octylbiotin: implications for the development of dynamically modified optical fiber sensors | |
| AU662925B2 (en) | Sensors and methods for sensing | |
| Sloan et al. | A fibre‐optic calcium ion sensor using a calcein derivative | |
| EP0889324A1 (en) | Sensors for the determination of potassium ions | |
| JPS61258201A (en) | Copolymer molecule bonded solid base material product and manufacture thereof | |
| AU662169B2 (en) | Exciplex-based sensors and methods for sensing | |
| Bhatia et al. | Fiber optic-based immunosensors: a progress report | |
| JPH06180287A (en) | Sensor element for measuring quantity of dissolving oxygen in sample |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): JP |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): AT BE CH DE FR GB IT LU NL SE |