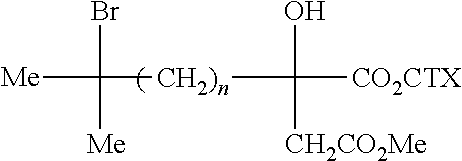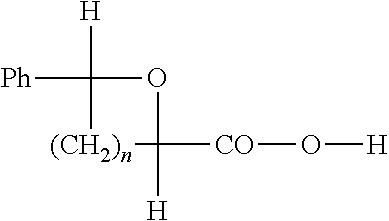USRE45128E1 - Cephalotaxane derivatives and their processes of preparation and purification - Google Patents
Cephalotaxane derivatives and their processes of preparation and purification Download PDFInfo
- Publication number
- USRE45128E1 USRE45128E1 US13/661,677 US201213661677A USRE45128E US RE45128 E1 USRE45128 E1 US RE45128E1 US 201213661677 A US201213661677 A US 201213661677A US RE45128 E USRE45128 E US RE45128E
- Authority
- US
- United States
- Prior art keywords
- hydroxy
- cephalotaxine
- ester
- methyl
- butanedioate
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related, expires
Links
- 0 CC(C)*C(C(Cl)=O)=O Chemical compound CC(C)*C(C(Cl)=O)=O 0.000 description 66
- HYFHYPWGAURHIV-DJWDSZJHSA-N COC(=O)C[C@](O)(CCCC(C)(C)O)C(=O)OC1C(OC)=C[C@]23CCCN2CCC2=CC4=C(C=C2[C@H]13)OCO4 Chemical compound COC(=O)C[C@](O)(CCCC(C)(C)O)C(=O)OC1C(OC)=C[C@]23CCCN2CCC2=CC4=C(C=C2[C@H]13)OCO4 HYFHYPWGAURHIV-DJWDSZJHSA-N 0.000 description 8
- YUSBYRHFSXBSJT-OJNLXCMQSA-N COC(=O)CC(O)(CCCC(C)(C)O)C(=O)OC1C(OC)=CC23CCCN2CCC2=CC4=C(C=C2C13)OCO4.COC(=O)C[C@](O)(CCCC(C)(C)O)C(=O)OC1C(OC)=C[C@]23CCCN2CCC2=CC4=C(C=C2[C@H]13)OCO4 Chemical compound COC(=O)CC(O)(CCCC(C)(C)O)C(=O)OC1C(OC)=CC23CCCN2CCC2=CC4=C(C=C2C13)OCO4.COC(=O)C[C@](O)(CCCC(C)(C)O)C(=O)OC1C(OC)=C[C@]23CCCN2CCC2=CC4=C(C=C2[C@H]13)OCO4 YUSBYRHFSXBSJT-OJNLXCMQSA-N 0.000 description 5
- RLJJTHIETVMNAO-UHFFFAOYSA-N [H]OC(=O)C1(CC(=O)OC)CCCC(C)(C)O1 Chemical compound [H]OC(=O)C1(CC(=O)OC)CCCC(C)(C)O1 RLJJTHIETVMNAO-UHFFFAOYSA-N 0.000 description 5
- QCQAAADYVNCDTL-UHFFFAOYSA-N COC(=O)CC(O)(CC(C)(C)O)C(=O)OC1C(OC)=CC23CCCN2CCC2=CC4=C(C=C2C13)OCO4 Chemical compound COC(=O)CC(O)(CC(C)(C)O)C(=O)OC1C(OC)=CC23CCCN2CCC2=CC4=C(C=C2C13)OCO4 QCQAAADYVNCDTL-UHFFFAOYSA-N 0.000 description 4
- YUSBYRHFSXBSJT-UVDZPDOLSA-N COC(=O)C[C@](O)(CCCC(C)(C)O)C(=O)OC1C(OC)=C[C@]23CCCN2CCC2=CC4=C(C=C2[C@H]13)OCO4.COC(=O)C[C@](O)(CCCC(C)(C)O)C(=O)OC1C(OC)=C[C@]23CCCN2CCC2=CC4=C(C=C2[C@H]13)OCO4 Chemical compound COC(=O)C[C@](O)(CCCC(C)(C)O)C(=O)OC1C(OC)=C[C@]23CCCN2CCC2=CC4=C(C=C2[C@H]13)OCO4.COC(=O)C[C@](O)(CCCC(C)(C)O)C(=O)OC1C(OC)=C[C@]23CCCN2CCC2=CC4=C(C=C2[C@H]13)OCO4 YUSBYRHFSXBSJT-UVDZPDOLSA-N 0.000 description 4
- YUSBYRHFSXBSJT-UMPMLZQQSA-N COC(=O)CC(O)(CCCC(C)(C)O)C(=O)OC1C(OC)=C[C@]23CCCN2CCC2=CC4=C(C=C2[C@H]13)OCO4.COC(=O)C[C@@](O)(CCCC(C)(C)O)C(=O)OC1C(OC)=C[C@]23CCCN2CCC2=C(C=C4OCOC4=C2)[C@H]13 Chemical compound COC(=O)CC(O)(CCCC(C)(C)O)C(=O)OC1C(OC)=C[C@]23CCCN2CCC2=CC4=C(C=C2[C@H]13)OCO4.COC(=O)C[C@@](O)(CCCC(C)(C)O)C(=O)OC1C(OC)=C[C@]23CCCN2CCC2=C(C=C4OCOC4=C2)[C@H]13 YUSBYRHFSXBSJT-UMPMLZQQSA-N 0.000 description 3
- JPTHFFGLKVWXQY-UHFFFAOYSA-N C1=CC=C2C(=C1)CCN1CCCC13CCCC23 Chemical compound C1=CC=C2C(=C1)CCN1CCCC13CCCC23 JPTHFFGLKVWXQY-UHFFFAOYSA-N 0.000 description 2
- BYOABXSVTBOUEB-UHFFFAOYSA-N CC1(C)CCCC2(CC(=O)OC2=O)O1 Chemical compound CC1(C)CCCC2(CC(=O)OC2=O)O1 BYOABXSVTBOUEB-UHFFFAOYSA-N 0.000 description 2
- NYGKGGIFRQFDRB-HFFOVNNJSA-N COC(=O)CC1(C(=O)OC2C(OC)=C[C@]34CCCN3CCC3=C(C=C5OCOC5=C3)[C@H]24)CCCC(C)(C)O1 Chemical compound COC(=O)CC1(C(=O)OC2C(OC)=C[C@]34CCCN3CCC3=C(C=C5OCOC5=C3)[C@H]24)CCCC(C)(C)O1 NYGKGGIFRQFDRB-HFFOVNNJSA-N 0.000 description 2
- TWWQUFGDVHWLBQ-UHFFFAOYSA-N COC1=[N+](C)C=CC=C1.[Br-] Chemical compound COC1=[N+](C)C=CC=C1.[Br-] TWWQUFGDVHWLBQ-UHFFFAOYSA-N 0.000 description 2
- FGBUEMLYNJISCW-UHFFFAOYSA-N COC=[N+](C)C.[Cl-] Chemical compound COC=[N+](C)C.[Cl-] FGBUEMLYNJISCW-UHFFFAOYSA-N 0.000 description 2
- YMNCVRSYJBNGLD-BLAYRMRBSA-N [H]C1(O)C(OC)=C[C@]23CCCN2CCC2=CC4=C(C=C2[C@H]13)OCO4 Chemical compound [H]C1(O)C(OC)=C[C@]23CCCN2CCC2=CC4=C(C=C2[C@H]13)OCO4 YMNCVRSYJBNGLD-BLAYRMRBSA-N 0.000 description 2
- NKMCHVMAKQPPEK-UHFFFAOYSA-N [H]OC(=O)C(O)(CCC=C(C)C)CC(=O)OC Chemical compound [H]OC(=O)C(O)(CCC=C(C)C)CC(=O)OC NKMCHVMAKQPPEK-UHFFFAOYSA-N 0.000 description 2
- RZRIFGMWEQEBTG-UHFFFAOYSA-N [H]OC(=O)C1(CC(=O)OC)CC([H])(C2=CC=CC=C2)O1 Chemical compound [H]OC(=O)C1(CC(=O)OC)CC([H])(C2=CC=CC=C2)O1 RZRIFGMWEQEBTG-UHFFFAOYSA-N 0.000 description 2
- JNLVZJFVFCIJNZ-UHFFFAOYSA-N [H]OC(=O)C1([H])CC([H])(C2=CC=CC=C2)O1 Chemical compound [H]OC(=O)C1([H])CC([H])(C2=CC=CC=C2)O1 JNLVZJFVFCIJNZ-UHFFFAOYSA-N 0.000 description 2
- CSMDYOZVZCWFEA-SVJBVINKSA-N [H][C@@]1([C@H](OC(=O)C2(CC(=O)OC)CCCC(C)(C)O2)C2=C3C=C(OC)C=CC3=NC=C2)C[C@@H]2CC[N@@]1CC2C=C Chemical compound [H][C@@]1([C@H](OC(=O)C2(CC(=O)OC)CCCC(C)(C)O2)C2=C3C=C(OC)C=CC3=NC=C2)C[C@@H]2CC[N@@]1CC2C=C CSMDYOZVZCWFEA-SVJBVINKSA-N 0.000 description 2
- HVQSZPCVPKESBR-UEACTRMWSA-N *.[H][C@@](OC(=O)CC(O)(CCC=C(C)C)C(=O)OC(C)(C)C)(C1=CC=CC=C1)C(O)(C1=CC=CC=C1)C1=CC=CC=C1 Chemical compound *.[H][C@@](OC(=O)CC(O)(CCC=C(C)C)C(=O)OC(C)(C)C)(C1=CC=CC=C1)C(O)(C1=CC=CC=C1)C1=CC=CC=C1 HVQSZPCVPKESBR-UEACTRMWSA-N 0.000 description 1
- LQQIOOYNWWPTHX-FDOABKJOSA-N *.[H][C@@](OC(=O)CC(O)(CCC=C(C)C)C(=O)OCC)(C1=CC=CC=C1)C(O)(C1=CC=CC=C1)C1=CC=CC=C1 Chemical compound *.[H][C@@](OC(=O)CC(O)(CCC=C(C)C)C(=O)OCC)(C1=CC=CC=C1)C(O)(C1=CC=CC=C1)C1=CC=CC=C1 LQQIOOYNWWPTHX-FDOABKJOSA-N 0.000 description 1
- RDXXKPMOWHVFLT-UHFFFAOYSA-N C.C.C1=CC=C2C(=C1)CCN1CCCC13CCCC23.C1=CC=C2C(=C1)CCN1CCCC13CCCC23.CO.O Chemical compound C.C.C1=CC=C2C(=C1)CCN1CCCC13CCCC23.C1=CC=C2C(=C1)CCN1CCCC13CCCC23.CO.O RDXXKPMOWHVFLT-UHFFFAOYSA-N 0.000 description 1
- DGDFJBILAKELJU-UHFFFAOYSA-N C.C1=CC=C2C(=C1)CCN1CCCC13CCCC23.COC Chemical compound C.C1=CC=C2C(=C1)CCN1CCCC13CCCC23.COC DGDFJBILAKELJU-UHFFFAOYSA-N 0.000 description 1
- PZJMZZBWYSHBJC-NRXBRZBTSA-N C.[3H]C(C)OC(=O)C(O)(CC(=O)OC)CC(C)(C)Br Chemical compound C.[3H]C(C)OC(=O)C(O)(CC(=O)OC)CC(C)(C)Br PZJMZZBWYSHBJC-NRXBRZBTSA-N 0.000 description 1
- JXQMQISDEMIKDH-NRXBRZBTSA-N C.[3H]C(C)OC(=O)C(O)(CC(=O)OC)CC(C)(C)N Chemical compound C.[3H]C(C)OC(=O)C(O)(CC(=O)OC)CC(C)(C)N JXQMQISDEMIKDH-NRXBRZBTSA-N 0.000 description 1
- UHEPJGULSIKKTP-UHFFFAOYSA-N CC(=O)CCC=C(C)C Chemical compound CC(=O)CCC=C(C)C UHEPJGULSIKKTP-UHFFFAOYSA-N 0.000 description 1
- JQZPSXAAOOQGHU-XDOQXNTLSA-N CC(C(O)C1=CC=CC=C1)[NH+](C)C.CC(C(O)C1=CC=CC=C1)[NH+](C)C.COC(=O)CC1(C(=O)[O-])CCCC(C)(C)O1.COC(=O)C[C@@]1(C(=O)[O-])CCCC(C)(C)O1 Chemical compound CC(C(O)C1=CC=CC=C1)[NH+](C)C.CC(C(O)C1=CC=CC=C1)[NH+](C)C.COC(=O)CC1(C(=O)[O-])CCCC(C)(C)O1.COC(=O)C[C@@]1(C(=O)[O-])CCCC(C)(C)O1 JQZPSXAAOOQGHU-XDOQXNTLSA-N 0.000 description 1
- GAXAFAIDVKXADL-UHFFFAOYSA-N CC(C)(C)OC(C)(CC(O1)=O)C1=O Chemical compound CC(C)(C)OC(C)(CC(O1)=O)C1=O GAXAFAIDVKXADL-UHFFFAOYSA-N 0.000 description 1
- HAVJATCHLFRDHY-UHFFFAOYSA-N CC(C)(CCC(CC(OC)=O)(C(OC(C1c2c3)C(OC)=CC1(CCC1)N1CCc2cc1c3OCO1)=O)O)O Chemical compound CC(C)(CCC(CC(OC)=O)(C(OC(C1c2c3)C(OC)=CC1(CCC1)N1CCc2cc1c3OCO1)=O)O)O HAVJATCHLFRDHY-UHFFFAOYSA-N 0.000 description 1
- DRFNZQPZTGLTJC-UHFFFAOYSA-N CC(C)=CCCC(O)(CC(=O)O)C(=O)O Chemical compound CC(C)=CCCC(O)(CC(=O)O)C(=O)O DRFNZQPZTGLTJC-UHFFFAOYSA-N 0.000 description 1
- SPQXEXUSESBANH-PPHPATTJSA-N CC(C)C(O)C1=CC=CC=C1.CC(C)[C@H](O)C1=CC=CC=C1 Chemical compound CC(C)C(O)C1=CC=CC=C1.CC(C)[C@H](O)C1=CC=CC=C1 SPQXEXUSESBANH-PPHPATTJSA-N 0.000 description 1
- AJBWTDQJHHDXPL-UHFFFAOYSA-N CC1(C)CCCC(CC(=O)O)(C(=O)O)O1 Chemical compound CC1(C)CCCC(CC(=O)O)(C(=O)O)O1 AJBWTDQJHHDXPL-UHFFFAOYSA-N 0.000 description 1
- RVZKHHXSPMCABK-UHFFFAOYSA-M CC1=CC23CCCN2CCC2=CC4=C(C=C2C3C1C)OCO4.CC1=CC23CCCN2CCC2=CC4=C(C=C2C3C1O)OCO4.CC1=CC23CCCN2CCC2=CC4=C(C=C2C3C1[Li]O)OCO4 Chemical compound CC1=CC23CCCN2CCC2=CC4=C(C=C2C3C1C)OCO4.CC1=CC23CCCN2CCC2=CC4=C(C=C2C3C1O)OCO4.CC1=CC23CCCN2CCC2=CC4=C(C=C2C3C1[Li]O)OCO4 RVZKHHXSPMCABK-UHFFFAOYSA-M 0.000 description 1
- SVXDEAYDJNELMG-UHFFFAOYSA-N CC1=CC23CCCN2CCC2=CC4=C(C=C2C3C1O)OCO4 Chemical compound CC1=CC23CCCN2CCC2=CC4=C(C=C2C3C1O)OCO4 SVXDEAYDJNELMG-UHFFFAOYSA-N 0.000 description 1
- IOAISUCAQCEHTA-UHFFFAOYSA-N CC1=CC=C(C(C)C)C(O)=C1.CC1=CC=C(C(C)C)C(O)=C1 Chemical compound CC1=CC=C(C(C)C)C(O)=C1.CC1=CC=C(C(C)C)C(O)=C1 IOAISUCAQCEHTA-UHFFFAOYSA-N 0.000 description 1
- VURMTZQBPHFFHT-KSUKSCHBSA-N CCC(O)(CC(C)(C)O)C(=O)O.O=C=O.O=C=O.[H]C1(O)C(OC)=C[C@]23CCCN2CCC2=CC4=C(C=C2[C@H]13)OCO4.[H]C1(OC(=O)C(O)(CC)CC(C)(C)O)C(OC)=C[C@]23CCCN2CCC2=CC4=C(C=C2[C@H]13)OCO4 Chemical compound CCC(O)(CC(C)(C)O)C(=O)O.O=C=O.O=C=O.[H]C1(O)C(OC)=C[C@]23CCCN2CCC2=CC4=C(C=C2[C@H]13)OCO4.[H]C1(OC(=O)C(O)(CC)CC(C)(C)O)C(OC)=C[C@]23CCCN2CCC2=CC4=C(C=C2[C@H]13)OCO4 VURMTZQBPHFFHT-KSUKSCHBSA-N 0.000 description 1
- BEDDBBCLHNGXGD-UHFFFAOYSA-N CCOC(=O)C(=O)CCC=C(C)C Chemical compound CCOC(=O)C(=O)CCC=C(C)C BEDDBBCLHNGXGD-UHFFFAOYSA-N 0.000 description 1
- BLDRTRSHVDPKCC-PIVQAISJSA-N CCOC(=O)C(O)(CCC=C(C)C)CC(=O)N1C(=O)OC[C@H]1C(C)C Chemical compound CCOC(=O)C(O)(CCC=C(C)C)CC(=O)N1C(=O)OC[C@H]1C(C)C BLDRTRSHVDPKCC-PIVQAISJSA-N 0.000 description 1
- NBOWWOOUDLBQFP-UHFFFAOYSA-N CCOC(=O)C(O)(CCC=C(C)C)CC(=O)OC Chemical compound CCOC(=O)C(O)(CCC=C(C)C)CC(=O)OC NBOWWOOUDLBQFP-UHFFFAOYSA-N 0.000 description 1
- ZGHABFQROAWMEO-FYWCMSFXSA-N CCOC(=O)C(O)(CCC=C(C)C)CC(=O)O[C@H]1C[C@@H](C)CC[C@@H]1C(C)C Chemical compound CCOC(=O)C(O)(CCC=C(C)C)CC(=O)O[C@H]1C[C@@H](C)CC[C@@H]1C(C)C ZGHABFQROAWMEO-FYWCMSFXSA-N 0.000 description 1
- ZZAIVSWZNPVDRU-UHFFFAOYSA-N CCOC(=O)C(O)(CCCC(C)(C)O)CC(=O)OC Chemical compound CCOC(=O)C(O)(CCCC(C)(C)O)CC(=O)OC ZZAIVSWZNPVDRU-UHFFFAOYSA-N 0.000 description 1
- VGAAPSXZTVDZDA-UHFFFAOYSA-N CCOC(=O)C1(CC(=O)OC)CCCC(C)(C)O1 Chemical compound CCOC(=O)C1(CC(=O)OC)CCCC(C)(C)O1 VGAAPSXZTVDZDA-UHFFFAOYSA-N 0.000 description 1
- MCTWTZJPVLRJOU-UHFFFAOYSA-N CN1C=CN=C1 Chemical compound CN1C=CN=C1 MCTWTZJPVLRJOU-UHFFFAOYSA-N 0.000 description 1
- DBMBGGDHXQBQRV-HUJVSABWSA-N CO/C1=C/[C@]23CCC[N@@]2CCC2=CC4=C(C=C2[C@@H]3C1OC(=O)C(C)(C)C)OCO4 Chemical compound CO/C1=C/[C@]23CCC[N@@]2CCC2=CC4=C(C=C2[C@@H]3C1OC(=O)C(C)(C)C)OCO4 DBMBGGDHXQBQRV-HUJVSABWSA-N 0.000 description 1
- QYFPNDFLFFKFJA-DDWIOCJRSA-N COC(=O)C(O)C1=CC=CC=C1.COC(=O)[C@H](O)C1=CC=CC=C1 Chemical compound COC(=O)C(O)C1=CC=CC=C1.COC(=O)[C@H](O)C1=CC=CC=C1 QYFPNDFLFFKFJA-DDWIOCJRSA-N 0.000 description 1
- FRWAHJPNYAKABR-UHFFFAOYSA-N COC(=O)C1(CC(=O)O)CCCC(C)(C)O1 Chemical compound COC(=O)C1(CC(=O)O)CCCC(C)(C)O1 FRWAHJPNYAKABR-UHFFFAOYSA-N 0.000 description 1
- AZYCUHYELMASNW-UHFFFAOYSA-N COC(=O)CC(C)(O)CCC=C(C)C Chemical compound COC(=O)CC(C)(O)CCC=C(C)C AZYCUHYELMASNW-UHFFFAOYSA-N 0.000 description 1
- CRKAFXGWDZSGMZ-UHFFFAOYSA-N COC(=O)CC(O)(CC(C)(C)O)C(=O)OC1C(C)=CC23CCCN2CCC2=CC4=C(C=C2C13)OCO4 Chemical compound COC(=O)CC(O)(CC(C)(C)O)C(=O)OC1C(C)=CC23CCCN2CCC2=CC4=C(C=C2C13)OCO4 CRKAFXGWDZSGMZ-UHFFFAOYSA-N 0.000 description 1
- JXGPHRXZPLKQSG-HFFOVNNJSA-N COC(=O)CC(O)(CCCC(C)(C)Br)C(=O)OC1/C(OC)=C\[C@]23CCC[N@@]2CCC2=CC4=C(C=C2[C@H]13)OCO4 Chemical compound COC(=O)CC(O)(CCCC(C)(C)Br)C(=O)OC1/C(OC)=C\[C@]23CCC[N@@]2CCC2=CC4=C(C=C2[C@H]13)OCO4 JXGPHRXZPLKQSG-HFFOVNNJSA-N 0.000 description 1
- OGDMWEGFKVYJBO-RFVHGSKJSA-N COC(=O)CC1(C(=O)O)CCC(C)(C)O1.COC(=O)C[C@@]1(C(=O)O)CCCC(C)(C)O1 Chemical compound COC(=O)CC1(C(=O)O)CCC(C)(C)O1.COC(=O)C[C@@]1(C(=O)O)CCCC(C)(C)O1 OGDMWEGFKVYJBO-RFVHGSKJSA-N 0.000 description 1
- BSIMQAIOFVEVRB-UHFFFAOYSA-N COC(=O)CC1(C(=O)OC(=O)C2=C(Cl)C=C(Cl)C=C2Cl)CCCC(C)(C)O1 Chemical compound COC(=O)CC1(C(=O)OC(=O)C2=C(Cl)C=C(Cl)C=C2Cl)CCCC(C)(C)O1 BSIMQAIOFVEVRB-UHFFFAOYSA-N 0.000 description 1
- KFKJLIDOAZUCPU-UHFFFAOYSA-N COC(=O)CC1(C(=O)OC(C(=O)OC)C2=CC=CC=C2)CCCC(C)(C)O1 Chemical compound COC(=O)CC1(C(=O)OC(C(=O)OC)C2=CC=CC=C2)CCCC(C)(C)O1 KFKJLIDOAZUCPU-UHFFFAOYSA-N 0.000 description 1
- NZMCOLUBUOTIQN-ALAWQYECSA-N COC(=O)CC1(C(=O)OC(C(=O)OC)C2=CC=CC=C2)CCCC(C)(C)O1.COC(=O)C[C@@]1(C(=O)OC(C(=O)OC)C2=CC=CC=C2)CCCC(C)(C)O1 Chemical compound COC(=O)CC1(C(=O)OC(C(=O)OC)C2=CC=CC=C2)CCCC(C)(C)O1.COC(=O)C[C@@]1(C(=O)OC(C(=O)OC)C2=CC=CC=C2)CCCC(C)(C)O1 NZMCOLUBUOTIQN-ALAWQYECSA-N 0.000 description 1
- KVMCTPIFXVKQIZ-UHFFFAOYSA-N COC(=O)CC1(C(=O)OC)CCCC(C)(C)O1 Chemical compound COC(=O)CC1(C(=O)OC)CCCC(C)(C)O1 KVMCTPIFXVKQIZ-UHFFFAOYSA-N 0.000 description 1
- BQTBLRNYILFSAV-FUUGTPEESA-N COC(=O)CC1(C(=O)OC2CC(C)CC[C@@H]2C(C)C)CCCC(C)(C)O1 Chemical compound COC(=O)CC1(C(=O)OC2CC(C)CC[C@@H]2C(C)C)CCCC(C)(C)O1 BQTBLRNYILFSAV-FUUGTPEESA-N 0.000 description 1
- UEZAWEBDADCYTD-NZAKVUCASA-N COC(=O)CC1(C(=O)OC2C[C@@H](C)CC[C@@H]2C(C)C)CCCC(C)(C)O1.COC(=O)C[C@@]1(C(=O)OC2CC(C)CC[C@@H]2C(C)C)CCCC(C)(C)O1 Chemical compound COC(=O)CC1(C(=O)OC2C[C@@H](C)CC[C@@H]2C(C)C)CCCC(C)(C)O1.COC(=O)C[C@@]1(C(=O)OC2CC(C)CC[C@@H]2C(C)C)CCCC(C)(C)O1 UEZAWEBDADCYTD-NZAKVUCASA-N 0.000 description 1
- RLJJTHIETVMNAO-LLVKDONJSA-N COC(=O)C[C@@]1(C(=O)O)CCCC(C)(C)O1 Chemical compound COC(=O)C[C@@]1(C(=O)O)CCCC(C)(C)O1 RLJJTHIETVMNAO-LLVKDONJSA-N 0.000 description 1
- NYGKGGIFRQFDRB-DJWDSZJHSA-N COC(=O)C[C@@]1(C(=O)OC2/C(OC)=C\[C@]34CCC[N@@]3CCC3=CC5=C(C=C3[C@H]24)OCO5)CCCC(C)(C)O1 Chemical compound COC(=O)C[C@@]1(C(=O)OC2/C(OC)=C\[C@]34CCC[N@@]3CCC3=CC5=C(C=C3[C@H]24)OCO5)CCCC(C)(C)O1 NYGKGGIFRQFDRB-DJWDSZJHSA-N 0.000 description 1
- JXGPHRXZPLKQSG-DJWDSZJHSA-N COC(=O)C[C@](O)(CCCC(C)(C)Br)C(=O)OC1/C(OC)=C\[C@]23CCC[N@@]2CCC2=CC4=C(C=C2[C@H]13)OCO4 Chemical compound COC(=O)C[C@](O)(CCCC(C)(C)Br)C(=O)OC1/C(OC)=C\[C@]23CCC[N@@]2CCC2=CC4=C(C=C2[C@H]13)OCO4 JXGPHRXZPLKQSG-DJWDSZJHSA-N 0.000 description 1
- YUSBYRHFSXBSJT-BYAFONPQSA-N COC(=O)C[C@](O)(CCCC(C)(C)O)C(=O)OC1C(OC)=C[C@@]23CCCN2CCC2=CC4=C(C=C2[C@@H]13)OCO4.COC(=O)C[C@](O)(CCCC(C)(C)O)C(=O)OC1C(OC)=C[C@]23CCCN2CCC2=CC4=C(C=C2C13)OCO4 Chemical compound COC(=O)C[C@](O)(CCCC(C)(C)O)C(=O)OC1C(OC)=C[C@@]23CCCN2CCC2=CC4=C(C=C2[C@@H]13)OCO4.COC(=O)C[C@](O)(CCCC(C)(C)O)C(=O)OC1C(OC)=C[C@]23CCCN2CCC2=CC4=C(C=C2C13)OCO4 YUSBYRHFSXBSJT-BYAFONPQSA-N 0.000 description 1
- ZJUDOSYLDNWDBG-VCWVRYCNSA-N COC(C([C@H]12)OC(C3N(Cc4ccccc4)C3c3ccccc3)=O)=C[C@H](CCC3)C1N3CCc1c2cc2OCOc2c1 Chemical compound COC(C([C@H]12)OC(C3N(Cc4ccccc4)C3c3ccccc3)=O)=C[C@H](CCC3)C1N3CCc1c2cc2OCOc2c1 ZJUDOSYLDNWDBG-VCWVRYCNSA-N 0.000 description 1
- ORCVESYJMGHNQN-GDSHKQBBSA-N COC(C)=O.[H]C(C)(C)CC(=O)C(=O)Cl.[H]C(C)(C)CC(=O)C(=O)OC([3H])C Chemical compound COC(C)=O.[H]C(C)(C)CC(=O)C(=O)Cl.[H]C(C)(C)CC(=O)C(=O)OC([3H])C ORCVESYJMGHNQN-GDSHKQBBSA-N 0.000 description 1
- RYTUQTKNXOITRX-RCEVZZFNSA-N COC1=C[C@]23CCCN2CCC2=CC4=C(C=C2[C@@H]3C1(O)Cl)OCO4.O=C=O.O=C=O.O=C=O.O=C=O.O=C=O.[H]C(C)(C)CC(=O)C(=O)Cl.[H]C(C)(C)CC(=O)C(=O)OC([3H])C.[H]C(C)(C)CC(=O)CC.[H]C(C)(C)C[C@@](O)(CC)C(=O)O.[H]C(C)(C)C[C@@](O)(CC)C(=O)O.[H]C(C)(C)C[C@@](O)(CC)C(=O)OC1([H])/C(OC)=C\[C@]23CCC[N@]2CCC2=CC4=C(C=C2[C@H]13)OCO4.[H]C1(O)C(OC)=C[C@]23CCCN2CCC2=CC4=C(C=C2[C@H]13)OCO4.[H]C1=C2C3=CC4=C(C=C3CCN3CCC[C@]23C(OC(=O)[C@](O)(CC)CC([H])(C)C)[C@@]1([H])C)OCO4.[Li]CC(=O)OC Chemical compound COC1=C[C@]23CCCN2CCC2=CC4=C(C=C2[C@@H]3C1(O)Cl)OCO4.O=C=O.O=C=O.O=C=O.O=C=O.O=C=O.[H]C(C)(C)CC(=O)C(=O)Cl.[H]C(C)(C)CC(=O)C(=O)OC([3H])C.[H]C(C)(C)CC(=O)CC.[H]C(C)(C)C[C@@](O)(CC)C(=O)O.[H]C(C)(C)C[C@@](O)(CC)C(=O)O.[H]C(C)(C)C[C@@](O)(CC)C(=O)OC1([H])/C(OC)=C\[C@]23CCC[N@]2CCC2=CC4=C(C=C2[C@H]13)OCO4.[H]C1(O)C(OC)=C[C@]23CCCN2CCC2=CC4=C(C=C2[C@H]13)OCO4.[H]C1=C2C3=CC4=C(C=C3CCN3CCC[C@]23C(OC(=O)[C@](O)(CC)CC([H])(C)C)[C@@]1([H])C)OCO4.[Li]CC(=O)OC RYTUQTKNXOITRX-RCEVZZFNSA-N 0.000 description 1
- XQEXYXORGFOFSL-MBXYGVAOSA-N COC1=C[C@]23CCCN2CCC2=CC4=C(C=C2[C@@H]3C1C)OCO4 Chemical compound COC1=C[C@]23CCCN2CCC2=CC4=C(C=C2[C@@H]3C1C)OCO4 XQEXYXORGFOFSL-MBXYGVAOSA-N 0.000 description 1
- PIETWHXITPJPML-SDFUFFHRSA-O [3H]C(C)OC(=O)C1(CC(=O)OC)CCC(C)(C)O1.[3H]C(C)OC(=O)C1(CC(=O)OC)CCCC(C)(C)O1.[H+] Chemical compound [3H]C(C)OC(=O)C1(CC(=O)OC)CCC(C)(C)O1.[3H]C(C)OC(=O)C1(CC(=O)OC)CCCC(C)(C)O1.[H+] PIETWHXITPJPML-SDFUFFHRSA-O 0.000 description 1
- ROKZBUZPHUUZIL-BXGMVJJPSA-N [H]C(N)(C1=CC=CC=C1)C([H])(O)C(=O)OC1C(OC)=C[C@]23CCCN2CCC2=CC4=C(C=C2[C@H]13)OCO4 Chemical compound [H]C(N)(C1=CC=CC=C1)C([H])(O)C(=O)OC1C(OC)=C[C@]23CCCN2CCC2=CC4=C(C=C2[C@H]13)OCO4 ROKZBUZPHUUZIL-BXGMVJJPSA-N 0.000 description 1
- MOBCZHBJOSVHDP-BXGMVJJPSA-N [H]C(O)(C(=O)OC1C(OC)=C[C@]23CCCN2CCC2=CC4=C(C=C2[C@H]13)OCO4)C([H])(N=[N+]=[N-])C1=CC=CC=C1 Chemical compound [H]C(O)(C(=O)OC1C(OC)=C[C@]23CCCN2CCC2=CC4=C(C=C2[C@H]13)OCO4)C([H])(N=[N+]=[N-])C1=CC=CC=C1 MOBCZHBJOSVHDP-BXGMVJJPSA-N 0.000 description 1
- UZYHMZKTETUFAH-YDJKICJBSA-N [H]C(O)(C(=O)OC1C(OC)=C[C@]23CCCN2CCC2=CC4=C(C=C2[C@H]13)OCO4)C([H])(NC(=O)C1=CC=CC=C1)C1=CC=CC=C1 Chemical compound [H]C(O)(C(=O)OC1C(OC)=C[C@]23CCCN2CCC2=CC4=C(C=C2[C@H]13)OCO4)C([H])(NC(=O)C1=CC=CC=C1)C1=CC=CC=C1 UZYHMZKTETUFAH-YDJKICJBSA-N 0.000 description 1
- LFGPGTINMCQRLH-QTTZXPTASA-N [H]C(O)(C(=O)OC1C(OC)=C[C@]23CCCN2CCC2=CC4=C(C=C2[C@H]13)OCO4)C([H])(NC(=O)OC(C)(C)C)C1=CC=CC=C1 Chemical compound [H]C(O)(C(=O)OC1C(OC)=C[C@]23CCCN2CCC2=CC4=C(C=C2[C@H]13)OCO4)C([H])(NC(=O)OC(C)(C)C)C1=CC=CC=C1 LFGPGTINMCQRLH-QTTZXPTASA-N 0.000 description 1
- LVFXTDPVZBRPBJ-BICURDJASA-N [H]C([H])(C1=CC=CC=C1)C([H])(O)C(=O)OC1C(OC)=C[C@]23CCCN2CCC2=CC4=C(C=C2[C@H]13)OCO4 Chemical compound [H]C([H])(C1=CC=CC=C1)C([H])(O)C(=O)OC1C(OC)=C[C@]23CCCN2CCC2=CC4=C(C=C2[C@H]13)OCO4 LVFXTDPVZBRPBJ-BICURDJASA-N 0.000 description 1
- LKEJEBLPKJFPSB-UHFFFAOYSA-N [H]C1(C(=O)OC2C(OC)=CC34CCCN3CCC3=CC5=C(C=C3C24)OCO5)OC1([H])C1=CC=CC=C1 Chemical compound [H]C1(C(=O)OC2C(OC)=CC34CCCN3CCC3=CC5=C(C=C3C24)OCO5)OC1([H])C1=CC=CC=C1 LKEJEBLPKJFPSB-UHFFFAOYSA-N 0.000 description 1
- BICYHFCMBIQKSC-XPGOMIJWSA-N [H]C1(C(=O)OC2C(OC)=C[C@]34CCCN3CCC3=CC5=C(C=C3[C@H]24)OCO5)N(CC2=CC=CC=C2)C1([H])C1=CC=CC=C1 Chemical compound [H]C1(C(=O)OC2C(OC)=C[C@]34CCCN3CCC3=CC5=C(C=C3[C@H]24)OCO5)N(CC2=CC=CC=C2)C1([H])C1=CC=CC=C1 BICYHFCMBIQKSC-XPGOMIJWSA-N 0.000 description 1
- JAWXNFPNLLZJDA-JQEAMFEWSA-N [H]C1(C(=O)OC2C(OC)=C[C@]34CCCN3CCC3=CC5=C(C=C3[C@H]24)OCO5)OC(C)(C)OC1([H])C1=CC=CC=C1 Chemical compound [H]C1(C(=O)OC2C(OC)=C[C@]34CCCN3CCC3=CC5=C(C=C3[C@H]24)OCO5)OC(C)(C)OC1([H])C1=CC=CC=C1 JAWXNFPNLLZJDA-JQEAMFEWSA-N 0.000 description 1
- LKEJEBLPKJFPSB-BXGMVJJPSA-N [H]C1(C(=O)OC2C(OC)=C[C@]34CCCN3CCC3=CC5=C(C=C3[C@H]24)OCO5)OC1([H])C1=CC=CC=C1 Chemical compound [H]C1(C(=O)OC2C(OC)=C[C@]34CCCN3CCC3=CC5=C(C=C3[C@H]24)OCO5)OC1([H])C1=CC=CC=C1 LKEJEBLPKJFPSB-BXGMVJJPSA-N 0.000 description 1
- PCPOTAFKNLXKBL-LHZOPFOASA-N [H]C1(C2=CC=CC=C2)CC(OC)OC1([H])C(=O)OC1C(OC)=C[C@]23CCCN2CCC2=CC4=C(C=C2[C@H]13)OCO4 Chemical compound [H]C1(C2=CC=CC=C2)CC(OC)OC1([H])C(=O)OC1C(OC)=C[C@]23CCCN2CCC2=CC4=C(C=C2[C@H]13)OCO4 PCPOTAFKNLXKBL-LHZOPFOASA-N 0.000 description 1
- VTLBMKFADKLSJR-UHFFFAOYSA-N [H]C1(C2=CC=CC=C2)N=C(C2=CC=CC=C2)OC1([H])C(=O)OC1C(OC)=CC23CCCN2CCC2=CC4=C(C=C2C13)OCO4 Chemical compound [H]C1(C2=CC=CC=C2)N=C(C2=CC=CC=C2)OC1([H])C(=O)OC1C(OC)=CC23CCCN2CCC2=CC4=C(C=C2C13)OCO4 VTLBMKFADKLSJR-UHFFFAOYSA-N 0.000 description 1
- VTLBMKFADKLSJR-YDJKICJBSA-N [H]C1(C2=CC=CC=C2)N=C(C2=CC=CC=C2)OC1([H])C(=O)OC1C(OC)=C[C@]23CCCN2CCC2=CC4=C(C=C2[C@H]13)OCO4 Chemical compound [H]C1(C2=CC=CC=C2)N=C(C2=CC=CC=C2)OC1([H])C(=O)OC1C(OC)=C[C@]23CCCN2CCC2=CC4=C(C=C2[C@H]13)OCO4 VTLBMKFADKLSJR-YDJKICJBSA-N 0.000 description 1
- PBBMIWPUJGABCF-UHFFFAOYSA-N [H]C1(OC)OC([H])(C(=O)OC2C(OC)=CC34CCCN3CCC3=CC5=C(C=C3C24)OCO5)C([H])(C2=CC=CC=C2)N1C(=O)C1=CC=CC=C1 Chemical compound [H]C1(OC)OC([H])(C(=O)OC2C(OC)=CC34CCCN3CCC3=CC5=C(C=C3C24)OCO5)C([H])(C2=CC=CC=C2)N1C(=O)C1=CC=CC=C1 PBBMIWPUJGABCF-UHFFFAOYSA-N 0.000 description 1
- NFMVLKMWVNLQKT-WQPBPYDNSA-M [H]C1([Li]O)C(OC)=C[C@]23CCCN2CCC2=CC4=C(C=C2[C@H]13)OCO4 Chemical compound [H]C1([Li]O)C(OC)=C[C@]23CCCN2CCC2=CC4=C(C=C2[C@H]13)OCO4 NFMVLKMWVNLQKT-WQPBPYDNSA-M 0.000 description 1
- JTRCDIHUHTYUCA-WQPBPYDNSA-M [H]C1([Na]O)C(OC)=C[C@]23CCCN2CCC2=CC4=C(C=C2[C@H]13)OCO4 Chemical compound [H]C1([Na]O)C(OC)=C[C@]23CCCN2CCC2=CC4=C(C=C2[C@H]13)OCO4 JTRCDIHUHTYUCA-WQPBPYDNSA-M 0.000 description 1
- JAWPJOMGMQIYIL-UHFFFAOYSA-N [H]OC(=O)C(O)(CC=C(C)C)CC(=O)OC Chemical compound [H]OC(=O)C(O)(CC=C(C)C)CC(=O)OC JAWPJOMGMQIYIL-UHFFFAOYSA-N 0.000 description 1
- BGUKHRANUOQOCN-VQHVLOKHSA-N [H]OC(=O)C([H])(O)C/C=C(\C)C1=CC=CC=C1 Chemical compound [H]OC(=O)C([H])(O)C/C=C(\C)C1=CC=CC=C1 BGUKHRANUOQOCN-VQHVLOKHSA-N 0.000 description 1
- DZBAKHOTFNKRDF-UHFFFAOYSA-N [H]OC(=O)C1(CC(=O)OC)CCC(C)(C)O1 Chemical compound [H]OC(=O)C1(CC(=O)OC)CCC(C)(C)O1 DZBAKHOTFNKRDF-UHFFFAOYSA-N 0.000 description 1
- TXZPDXJCATVTPS-UHFFFAOYSA-N [H]OC(=O)C1(CC(=O)OC)OC1([H])C1=CC=CC=C1 Chemical compound [H]OC(=O)C1(CC(=O)OC)OC1([H])C1=CC=CC=C1 TXZPDXJCATVTPS-UHFFFAOYSA-N 0.000 description 1
- KFLQWXPNNNRIED-KHTNLEEKSA-N [H][C@@]1([C@H](O)C2=C3C=C(C)C=CC3=NC=C2)C[C@@H]2CC[N@@]1CC2C=C Chemical compound [H][C@@]1([C@H](O)C2=C3C=C(C)C=CC3=NC=C2)C[C@@H]2CC[N@@]1CC2C=C KFLQWXPNNNRIED-KHTNLEEKSA-N 0.000 description 1
- PWRUSBVMFQRZNS-LOESWOACSA-N [H][C@@]1([C@H](OC(=O)C2(CC(=O)OC)CCCC(C)(C)O2)C2=C3C=C(OC)C=CC3=NC=C2)C[C@@H]2CC[N@@]1C[C@@H]2C=C.[H][C@@]1([C@H](OC(=O)[C@]2(CC(=O)OC)CCCC(C)(C)O2)C2=C3C=C(OC)C=CC3=NC=C2)C[C@@H]2CC[N@@]1C[C@@H]2C=C Chemical compound [H][C@@]1([C@H](OC(=O)C2(CC(=O)OC)CCCC(C)(C)O2)C2=C3C=C(OC)C=CC3=NC=C2)C[C@@H]2CC[N@@]1C[C@@H]2C=C.[H][C@@]1([C@H](OC(=O)[C@]2(CC(=O)OC)CCCC(C)(C)O2)C2=C3C=C(OC)C=CC3=NC=C2)C[C@@H]2CC[N@@]1C[C@@H]2C=C PWRUSBVMFQRZNS-LOESWOACSA-N 0.000 description 1
- CSMDYOZVZCWFEA-HVKWCGAPSA-N [H][C@@]1([C@H](OC(=O)[C@]2(CC(=O)OC)CCCC(C)(C)O2)C2=C3C=C(OC)C=CC3=NC=C2)C[C@@H]2CC[N@@]1CC2C=C Chemical compound [H][C@@]1([C@H](OC(=O)[C@]2(CC(=O)OC)CCCC(C)(C)O2)C2=C3C=C(OC)C=CC3=NC=C2)C[C@@H]2CC[N@@]1CC2C=C CSMDYOZVZCWFEA-HVKWCGAPSA-N 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D493/00—Heterocyclic compounds containing oxygen atoms as the only ring hetero atoms in the condensed system
- C07D493/02—Heterocyclic compounds containing oxygen atoms as the only ring hetero atoms in the condensed system in which the condensed system contains two hetero rings
- C07D493/10—Spiro-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D491/00—Heterocyclic compounds containing in the condensed ring system both one or more rings having oxygen atoms as the only ring hetero atoms and one or more rings having nitrogen atoms as the only ring hetero atoms, not provided for by groups C07D451/00 - C07D459/00, C07D463/00, C07D477/00 or C07D489/00
- C07D491/12—Heterocyclic compounds containing in the condensed ring system both one or more rings having oxygen atoms as the only ring hetero atoms and one or more rings having nitrogen atoms as the only ring hetero atoms, not provided for by groups C07D451/00 - C07D459/00, C07D463/00, C07D477/00 or C07D489/00 in which the condensed system contains three hetero rings
- C07D491/14—Ortho-condensed systems
- C07D491/147—Ortho-condensed systems the condensed system containing one ring with oxygen as ring hetero atom and two rings with nitrogen as ring hetero atom
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P33/00—Antiparasitic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P33/00—Antiparasitic agents
- A61P33/02—Antiprotozoals, e.g. for leishmaniasis, trichomoniasis, toxoplasmosis
- A61P33/06—Antimalarials
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C57/00—Unsaturated compounds having carboxyl groups bound to acyclic carbon atoms
- C07C57/02—Unsaturated compounds having carboxyl groups bound to acyclic carbon atoms with only carbon-to-carbon double bonds as unsaturation
- C07C57/13—Dicarboxylic acids
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C59/00—Compounds having carboxyl groups bound to acyclic carbon atoms and containing any of the groups OH, O—metal, —CHO, keto, ether, groups, groups, or groups
- C07C59/40—Unsaturated compounds
- C07C59/42—Unsaturated compounds containing hydroxy or O-metal groups
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C59/00—Compounds having carboxyl groups bound to acyclic carbon atoms and containing any of the groups OH, O—metal, —CHO, keto, ether, groups, groups, or groups
- C07C59/40—Unsaturated compounds
- C07C59/42—Unsaturated compounds containing hydroxy or O-metal groups
- C07C59/48—Unsaturated compounds containing hydroxy or O-metal groups containing six-membered aromatic rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C69/00—Esters of carboxylic acids; Esters of carbonic or haloformic acids
- C07C69/66—Esters of carboxylic acids having esterified carboxylic groups bound to acyclic carbon atoms and having any of the groups OH, O—metal, —CHO, keto, ether, acyloxy, groups, groups, or in the acid moiety
- C07C69/73—Esters of carboxylic acids having esterified carboxylic groups bound to acyclic carbon atoms and having any of the groups OH, O—metal, —CHO, keto, ether, acyloxy, groups, groups, or in the acid moiety of unsaturated acids
- C07C69/732—Esters of carboxylic acids having esterified carboxylic groups bound to acyclic carbon atoms and having any of the groups OH, O—metal, —CHO, keto, ether, acyloxy, groups, groups, or in the acid moiety of unsaturated acids of unsaturated hydroxy carboxylic acids
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D203/00—Heterocyclic compounds containing three-membered rings with one nitrogen atom as the only ring hetero atom
- C07D203/04—Heterocyclic compounds containing three-membered rings with one nitrogen atom as the only ring hetero atom not condensed with other rings
- C07D203/06—Heterocyclic compounds containing three-membered rings with one nitrogen atom as the only ring hetero atom not condensed with other rings having no double bonds between ring members or between ring members and non-ring members
- C07D203/08—Heterocyclic compounds containing three-membered rings with one nitrogen atom as the only ring hetero atom not condensed with other rings having no double bonds between ring members or between ring members and non-ring members with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to the ring nitrogen atom
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D263/00—Heterocyclic compounds containing 1,3-oxazole or hydrogenated 1,3-oxazole rings
- C07D263/02—Heterocyclic compounds containing 1,3-oxazole or hydrogenated 1,3-oxazole rings not condensed with other rings
- C07D263/08—Heterocyclic compounds containing 1,3-oxazole or hydrogenated 1,3-oxazole rings not condensed with other rings having one double bond between ring members or between a ring member and a non-ring member
- C07D263/16—Heterocyclic compounds containing 1,3-oxazole or hydrogenated 1,3-oxazole rings not condensed with other rings having one double bond between ring members or between a ring member and a non-ring member with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D303/00—Compounds containing three-membered rings having one oxygen atom as the only ring hetero atom
- C07D303/02—Compounds containing oxirane rings
- C07D303/48—Compounds containing oxirane rings with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, directly attached to ring carbon atoms, e.g. ester or nitrile radicals
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D305/00—Heterocyclic compounds containing four-membered rings having one oxygen atom as the only ring hetero atoms
- C07D305/02—Heterocyclic compounds containing four-membered rings having one oxygen atom as the only ring hetero atoms not condensed with other rings
- C07D305/04—Heterocyclic compounds containing four-membered rings having one oxygen atom as the only ring hetero atoms not condensed with other rings having no double bonds between ring members or between ring members and non-ring members
- C07D305/08—Heterocyclic compounds containing four-membered rings having one oxygen atom as the only ring hetero atoms not condensed with other rings having no double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D307/00—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom
- C07D307/02—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom not condensed with other rings
- C07D307/04—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom not condensed with other rings having no double bonds between ring members or between ring members and non-ring members
- C07D307/10—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom not condensed with other rings having no double bonds between ring members or between ring members and non-ring members with substituted hydrocarbon radicals attached to ring carbon atoms
- C07D307/16—Radicals substituted by carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D309/00—Heterocyclic compounds containing six-membered rings having one oxygen atom as the only ring hetero atom, not condensed with other rings
- C07D309/02—Heterocyclic compounds containing six-membered rings having one oxygen atom as the only ring hetero atom, not condensed with other rings having no double bonds between ring members or between ring members and non-ring members
- C07D309/08—Heterocyclic compounds containing six-membered rings having one oxygen atom as the only ring hetero atom, not condensed with other rings having no double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D317/00—Heterocyclic compounds containing five-membered rings having two oxygen atoms as the only ring hetero atoms
- C07D317/08—Heterocyclic compounds containing five-membered rings having two oxygen atoms as the only ring hetero atoms having the hetero atoms in positions 1 and 3
- C07D317/10—Heterocyclic compounds containing five-membered rings having two oxygen atoms as the only ring hetero atoms having the hetero atoms in positions 1 and 3 not condensed with other rings
- C07D317/32—Heterocyclic compounds containing five-membered rings having two oxygen atoms as the only ring hetero atoms having the hetero atoms in positions 1 and 3 not condensed with other rings with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D453/00—Heterocyclic compounds containing quinuclidine or iso-quinuclidine ring systems, e.g. quinine alkaloids
- C07D453/02—Heterocyclic compounds containing quinuclidine or iso-quinuclidine ring systems, e.g. quinine alkaloids containing not further condensed quinuclidine ring systems
- C07D453/04—Heterocyclic compounds containing quinuclidine or iso-quinuclidine ring systems, e.g. quinine alkaloids containing not further condensed quinuclidine ring systems having a quinolyl-4, a substituted quinolyl-4 or a alkylenedioxy-quinolyl-4 radical linked through only one carbon atom, attached in position 2, e.g. quinine
Definitions
- the present invention relates to a process for preparing cephalotaxane derivatives bearing a side chain. It further relates to highly purified and crystalline forms of harringtonines and their process of preparation by purification of crude alkaloids from natural, synthetic or semi-synthetic sources.
- the acquisition of highly purified and crystalline forms of harringtonines allows their use for blending in pharmaceutical compositions, especially as directed to the treatment of cancer through oral administration.
- cephalotaxanes refers to compounds or salts thereof which have a basic skeleton of formula
- p is equal to 1 or 2 (it being possible for the two units to be identical or different and linked via a single bond or an oxygen atom), which can contain various oxygenated substituents (aliphatic or aromatic ethers, free or esterified alcohols, substituted or free enols and/or phenols, bridged ethers, and more generally any substituent usually encountered in the natural state on compounds of this type).
- oxygenated substituents aliphatic or aromatic ethers, free or esterified alcohols, substituted or free enols and/or phenols, bridged ethers, and more generally any substituent usually encountered in the natural state on compounds of this type).
- Harringtonines are alkaloids which are of high interest in anticancer chemotherapy, in particular on certain haematosarcomas which are multi-resistant to the existing therapies.
- the selectivity of harringtonines, which is based on a novel mechanism of action relating to protein synthesis, is such that this series is favoured with a great future in anticancer therapy.
- Homo-harringtonine the most active member of the series, is active at and above daily doses of 2.5 mg/m 2 of body area per 24 hours, i.e., as a guide, at doses twenty times lower than that for Taxol.
- HHT has already undergone fourteen phase I and II clinical trials and it is the only known product capable of a 70% reinduction of full haematological remissions in patients suffering from chronic myeloid leukaemias that have become resistant to alpha-interferon [S. O'Brien, H. Kantarjian, M. Keating, M. Beran, C. Koler, L. E. Robertson, J. Hester, M. Rios, M. Andreeff and M. Talpaz, Blood, 332 (1995); Leukemia Insights, Vol. 3, No. 1 (1998)].
- Cephalotaxus harringtonia a biosynthetic precursor of the harringtonines, the latter individually representing only a few percent of the total alkaloids.
- harringtonines are mixed with many congeners which have very similar chemical structures.
- HPLC high performance liquid chromatography
- harringtonines are generally relatively non-crystallogenic, as is suggested by the flexibility of their side chains, which are generally branched and aliphatic,
- esters in particular harringtonine and homo-harringtonine, are contaminated with congeners which are themselves biologically active and very difficult to separate out, even by high resolution analytical HPLC,
- cephalotaxanes are alkaloids which have a unique structure in nature, encountered only in the genus Cephalotaxus, which is the only genus of the Cephalotaxacea family.
- the side chains of the various harringtonine congeners are all derived from the methyl hemiester of the primary carboxyl of (2R) citramalic acid 3a (see table p 156) by substitution of the tertiary methyl using alkyl or aralkyl radicals which may themselves be unsubstituted or substituted with tertiary hydroxyls, it then being possible for the latter to form a cyclic ether with a tertiary alcohol (anhydro derivatives).
- the table p 156 shows the main examples of harringtonine congeners, which all have significant cytostatic activity to different degrees. None of the artificial analogous table p 156) and lack significant cytostatic activity.
- Taxaceas contain triterpene alkaloids (taxines), accompanied by non-alkaloid triterpenes, taxanes, which are also of unique structure in nature. Although they are completely different from taxanes in terms of chemical structures and anticancer mechanism of activity, the harringtonines have analogy with taxanes in more than one respect:
- the polycyclic part of the taxanes (baccatins in the broad sense) and of the harringtonines (cephalotaxines) is relatively abundant in renewable parts of the plant, whereas the active molecules (harringtonines and taxanes) are ten to one hundred times less abundant therein,
- the plum yew (Cephalotaxus) is a rare tree, even rarer than the yew (Taxus), and is much less ubiquitous than the latter.
- HHT homo-harringtonine
- harringtonine and isoharringtonine have been the subject of American patent applications for their preparation by extraction [R. G. Powell et al., U.S. Pat. No. 3,793,454 and U.S. Pat. No. 3,870,727].
- Harringtonine has been the subject of a Japanese patent [JP 58-032,880] and deoxyharringtonine has been the subject of an American patent [U.S. Pat. No. 3,959,312].
- cephalotaxine can serve as a springboard for the synthesis of cephalotaxoids and harringtoids which are useful for antitumour (cancerous and non-cancerous tumours), antiparasitic, antifungal, antiviral and antibacterial chemotherapies.
- Harringtonines consist of a complex alkaloid polycyclic alcohol (cephalotaxine), esterified with a side chain, in isolation having no more biological activity than cephalotaxine, but essential for the biological activity of the whole. Saponification of the side chain under harsh conditions leads to the cephalotaxine free base and to harringtonic acids. The attachment of the side chains takes place at the end of the biosynthesis. It has been demonstrated that catabolism leading to this reaction could be triggered in vivo under the influence of environmental or physiological stress exerted on the plant [N. E. Delfel, Phytochemistry, 403 (1980)].
- Cephalotaxine the polycyclic part consisting of 5 fused rings, has a novel arrangement which is unique in nature, i.e. a benzodioxoazepine onto which is fused a spiropyrrolidinopentenediol system.
- Cephalotaxane contains four asymmetric centres: three “asymmetric carbons” and a heterocyclic tertiary aminic nitrogen.
- the only reactive function is a secondary alcohol located in position 3, the methyl enol ether located in position 2 being potentially sensitive to proton attack.
- the whole forms a pseudohelical structure encaging the hydroxyl in the tube formed by the tetrahydrazepine.
- the base cephalotaxine readily forms highly crystallogenic stable salts (for example hydrochlorides and perhydrochlorides).
- This compound and its congeners which are not O-acylated in position 3 are biologically inactive.
- the alkyl chain has at the end either branching constituting an isopropyl bearing (harringtonine HT and homo-harringtonine HHT) or not bearing (deoxy-homo-harringtonine DHT) a tertiary alcohol, or a phenyl radical (for example the neo-harringtonine series most recently isolated).
- the chain can be closed by dehydration between its two tertiary alcohols, for example forming a substituted tetrahydropyran ring.
- the tertiary carboxyl of this complex diester is borne by the single hydroxyl of the cephalotaxine.
- the only chiral centre on the side chain is located (to the ester junction. It contains, besides the first secondary chain, a hydroxyl which, on account of its tertiary nature, does not have the possibility of epimerizing.
- the table p 157 shows synthetically the known processes for preparing harringtonines.
- CTX represents the cephalotaxyl radical of formula:
- cephalotaxine is present in nature in partially racemized form [personal observation; Huang et al., Scientia Sinica, Vol. XXIII, 835 (1980)], the processes of the prior art which use a natural cephalotaxine as starting material can only theoretically result in partially racemized harringtonines.
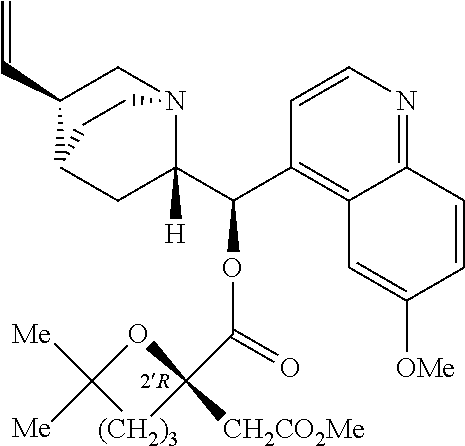
- FIGS. 1A and 1B gives the sequence of synthesis of homo-harringtonine corresponding to the Example 25, where A represents a 2,4,6-trichlorophenyl group, R represents a methyl and R′ represents a cephalotaxyl moiety.
- FIGS. 2A and 2B represents a variant of the process according to the invention, more exactly the semi-synthesis of harrintonines via oxacycloalkane carboxylic acids.
- the substituents R 6 , R 7 , R 8 , R, A, CTX, X and the letters n and m referenced in this scheme are defined in the description.
- FIG. 3 shows a typical DSC curve for a crystalline homoharringtonine.
- FIG. 4 shows an a typical X-ray diffractogram for a crystalline homoharringtonine.
- FIG. 5 shows an IR spectrum, in KBR KBr for a crystalline homoharringtonine.
- FIG. 6 shows a typical DSC curve for a homoarringtonine crystalline harringtonine.
- FIG. 7 shows an a typical IR spectrum, in KBR KBr for a homoharringtonine crystalline harringtonine.
- FIG. 8 shows an HPLC chromatogram of commercial, natural harringtonine using UV detection.
- FIG. 9 shows an enlarged chromatogram of commercial, natural harringtonine using liquid chromatography coupled with mass spectrometry and a UV detector.
- FIG. 10 shows identification of major impurities of commercial natural harringtonine using liquid chromatography coupled with mass spectrometry and a UV detector.
- FIG. 11 shows an enlarged chromatogram of highly purified natural harringtonine using liquid chromatography coupled with mass spectrometry and a UV detector.
- FIG. 12 shows an HPLC chromatogram showing the progression of purity between raw and chromatographed semi-synthetic homoharringonine DS.
- FIG. 13 shows an HPLC chromatogram showing the progression of purity between chromatographed and crystallized semi-synthetic homoharringtonine DS.
- FIG. 14 shows HPLC profiles of three samples of different natural sources.
- Alkaloids natural substances present in the vegetal kingdom allowing at least a cyclic or acyclic basic nitrogen (allowed extensions: animal kingdom, primary amine; refused extensions: amidic nitrogen, because non basic, ex: taxanes) and showing frequently pronounced pharmacological properties.
- Basic or free Alkaloid alkaloid showing a tertiary amine in a non-ionized form generally existing at alkaline pH and soluble in aprotic organic solvents.
- Cephalotaxanes 1 (see table p. 134): this generic term indicates the basic framework, showing diverse oxygenated substitutes (aliphatic or aromatic ether, free or esterified alcohol, enol and/or free or substituted phenol, bridged ether, more generally all substitute usually founded at natural state for this kind of compounds).
- Cephalotaxanes are particular alkaloids today only extracted from the Cephalotaxaceae family which exhibiting the structural formula 1 (Scheme 1). Several substituants may be encountered on this core structure: hydroxyl, ether, acyloxy etc. The eventual presence of some additional double bound or intramolecular bridge achieve to definite cephalotaxanes.
- Cephalotaxines 2 and harringtonines 3 are examples of cephalotaxanes. Several dozen of cephalotaxanes have been isolated from various Cephalotaxus species.
- Cephalotaxines 2 this generic term indicates cephalotaxanes possibly showing at least one of the substitutes described above, a sidechain excepted.
- Cephalotaxines 2 (Scheme 1) are cephalotaxanes without acyloxy side-chain.
- Cephalotaxine 2a and drupacine 2b are example of cephalotaxines.
- Cephalotaxine 2 a cephalotaxine in majority present in the genus Cephalotaxus
- Cephalotaxoids this generic term indicates a non-natural cephalotaxine.
- Harringtonines this generic term indicates cephalotaxane showing at least an alcohol group, a phenol or an enol, esterified by a sidechain and possibly by one of the substitutes described above.
- Harringtonines 3 (Scheme 1) are particular cephalotaxanes formed by attachment of a branched hydroxyacyloxy side-chain at the 3-position of various cephalotaxines moieties.
- Harringtonines are natural esters of cephalotaxines exhibiting generally a strong cytotoxic activity. However the lost only one atom of this minimal structure lead to a dramatic lost of activity (see below).
- harringtonines are harringtonine 3a, homoharringtonine 3b, drupangtonine 3c, anhydroharringtonine 3d and neoharringtonine 3e.
- Harringtonine one of the main alkaloids bearing a sidechain in position 3.
- Harringtoids this generic term indicates a non-natural harringtonine, where sidechain is an ester radical showing at least 3 carbon atoms.
- Sidechain of the harringtoids indicates an ester formed between one of the hydroxyl group and a carboxylic acid showing at least 3 carbon atoms allowing usually a tertiary alcohol tertiaire in ⁇ the ⁇ (alpha) position and an hydrophobe substitute hydrophobic substituent in ⁇ the ⁇ position relatively relative to the carboxyl group.
- the present invention thus has the advantage of obtaining enantiomerically pure harringtonines even from racemic cephalotaxine, since:
- the asymmetric centre on the side chain is created prior to the esterification step, i.e. the side chain precursor can be obtained in enantiomerically pure form prior to being attached,
- the present invention consists in:
- One part of the present invention thus consists in synthesizing, in particular, anhydroharringtonine, harringtonine, anhydro-homo-harringtonine and homo-harringtonine.
- the present invention also relates to esterifying cephalotaxines or metal alkoxides thereof with N-alkyl- and N-carbamoyl-2-alkylisoserine.
- the hydroxyl located at position 13 on the taxane skeleton made it possible to receive acylation with a relatively bulky chain such as, for example, an N-benzoylphenylisoserine protected in position 2′ (although the attempts to acylate baccatin protected with a chain bearing an (hydroxyl group protected with a benzoyl group all failed).
- a relatively bulky chain such as, for example, an N-benzoylphenylisoserine protected in position 2′ (although the attempts to acylate baccatin protected with a chain bearing an (hydroxyl group protected with a benzoyl group all failed).
- the hydroxyl function of a cephalotaxane is esterified with a 2-carboxyl-2-alkyl-1-oxacycloalkane derivative.
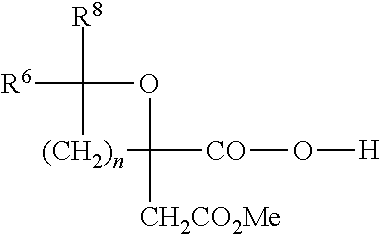
- hydroxyl of a cephalotaxane whose skeleton corresponds to the general formula 1 or, more particularly, a secondary alcohol located in position 3 of a cephalotaxine or metal alkoxides thereof, corresponding respectively to the general formulae CTX—O—H and CTX—O-M, M being a metal and CTX—the cephalotaxyl radical defined above, are esterified with the tertiary carboxylic acid function of a substituted cycloether corresponding to the general formula of the type 3k, or alternatively, preferably, with one of its activated forms, isolated or formed in situ, corresponding to the general formula of the type 3l to give 4c, according to the reaction below:
- R 5 , R 6 and R 8 are, independently of each other,
- a linear or branched and/or cyclic, saturated, unsaturated or aromatic, hydrocarbon-based radical in particular an alkyl, an alkenyl, an alkynyl, a cycloalkyl, a cycloalkenyl, an aryl, a heterocycloalkyl, the said hydrocarbon-based radical bearing or not bearing (a) heteroatom(s), it being possible for R 5 and R 8 to be linked together to form a ring
- n is between 0 and 8.
- A is a leaving group capable of leaving behind a carbocation
- 3l can thus be, in particular: an acid halide, an anhydride, a mixed anhydride or a cyclic anhydride (when R 5 ⁇ —CH 2 CO—).
- the free acid of the type 3k or any activated form of the acids of the type 3l can be used to esterify the cephalotaxanes corresponding to the general formula 1 or, for example, the cephalotaxines of the type CTX—OH.
- the anhydride of 3l can be prepared separately and isolated, and then placed in contact with the alcohol. This is likewise the case for the cyclic anhydrides of the type 3p
- n, R 6 and R 8 have the same meaning as above, and which can be readily prepared from the corresponding diacids in order once again to give 3k by esterification of methanol or alternatively to become attached as above to the alcohol function of a cephalotaxine of the type 2 with, however, a poorer yield than that above, the primary acid function then being methylated conventionally using methanol in the presence of a protonic acid or a Lewis acid, or alternatively using the boron trifluoride etherate/methanol complex or diazomethane.
- All the reagents of the type 3k, 3l and 2, as well as the resulting esters of the type 4c, can be used alone in enantiomerically pure form, or in the form of a racemic mixture or in the form of diastereoisomeric mixtures.
- the intermediates can, in certain cases, not be isolated or formed in situ fleetingly.
- the reaction can take place at between 0° C. and 140° C., with or without an organic solvent, it being possible for these solvents to be alone or as a mixture.
- the esterification of the hydroxyl of cephalotaxane with a 2-carboxyl-2-alkyl-1-oxacycloalkane derivative can be carried out either by acyl transfer to the alcohol or by the carbodiimide method.
- the esterification (a) takes place by placing the acid of the type 3k and the alcohol of the type 2 in contact in solution in a co-solvent and in the presence of an acid catalyst.
- the displacement of the equilibrium can be promoted by adding a dehydrating agent or by azeotropic entrainment or by partition between two immiscible solvents, one of which is miscible with the ester formed and the other with water.
- the acid catalyst can be a protonic acid such as, for example, sulphuric acid, hydrochloric acid, boric acid, preferably para-toluenesulphonic acid, or a Lewis acid which may or may not be supported on a polymer, such as, for example, aluminium chloride, chlorotrimethylsilane or, preferably, boron trifluoride etherate.
- a protonic acid such as, for example, sulphuric acid, hydrochloric acid, boric acid, preferably para-toluenesulphonic acid
- a Lewis acid which may or may not be supported on a polymer, such as, for example, aluminium chloride, chlorotrimethylsilane or, preferably, boron trifluoride etherate.
- an ion-exchange resin or bisulphate on graphite can also be used, for example.
- the dehydrating agent can be, for example, a dehydrating agent consisting of an inorganic salt which is low in water and inert with respect to the reaction, such as magnesium sulphate, sodium sulphate, calcium chloride or, preferably, a molecular sieve.
- an inorganic salt which is low in water and inert with respect to the reaction, such as magnesium sulphate, sodium sulphate, calcium chloride or, preferably, a molecular sieve.
- the esterification (a) uses the same reaction conditions as above. However, in order to result in the formation of the acylium ion characteristic of this method, the ordinary acid catalysts are replaced, for example, with anhydrous sulphuric acid or superacids such as, for example, hydrofluoric acid and its derivatives or antimony pentafluoride.
- anhydrous sulphuric acid or superacids such as, for example, hydrofluoric acid and its derivatives or antimony pentafluoride.
- the esterification (b) consists in using the same physical operating conditions as above, but using 3l, in which A represents a halogen (acid halide), another alkanoyl molecule which is identical (anhydride) or different (mixed anhydride), such as, for example, trifluoroacetyl, 2,4,6-trichlorobenzoyl, formyl, methoxyformyl, sulphonates, phosphates and chlorophosphate.
- A represents a halogen (acid halide), another alkanoyl molecule which is identical (anhydride) or different (mixed anhydride), such as, for example, trifluoroacetyl, 2,4,6-trichlorobenzoyl, formyl, methoxyformyl, sulphonates, phosphates and chlorophosphate.
- n, R 6 and R 8 have the same meaning as above, which can be prepared very simply by treating the corresponding diacid with acetic anhydride, for example under the general operating conditions for the preparation of the mixed anhydrides described below.
- the catalysis can be acidic, as indicated in the above method, or, preferably, alkaline, for example a tertiary base such as pyridine and/or dimethylaminopyridine (DMAP), pyrrolidinopyridine (PPY), triethylamine, or a stronger base such as a hydride, for example calcium hydride.
- the solvent can preferably be an aprotic solvent, for example hexane, toluene, dichloromethane, tetrahydrofuran and/or pyridine.
- esterification (c) is a method similar to the esterification (b). These ester preparation methods can also be advantageously used and involve, for example, 1-acyloxy-1,2,3-triazole or formamidinium or silyl ester or 2-acyloxypyridinium intermediates.
- the carbonyldiimidazole method in which an N-acylimidazole intermediate is involved, can also be used.
- the carbodiimide method uses a dehydrating coupling agent such as a carbodiimide, for example dicyclohexylcarbodiimide (DCC), 1,3-diisopropylcarbo-diimide (DIC) or 1-(3-dimethylaminopropyl)-3-ethyl-carbodiimide.
- a dehydrating coupling agent such as a carbodiimide, for example dicyclohexylcarbodiimide (DCC), 1,3-diisopropylcarbo-diimide (DIC) or 1-(3-dimethylaminopropyl)-3-ethyl-carbodiimide.
- the reaction can be catalysed with a tertiary base such as, for example, pyridine and/or dimethylaminopyridine (DMAP), pyrrolidinopyridine (PPY), triethylamine, 4-morpholinopyridine or any other substituted base.
- a tertiary base such as, for example, pyridine and/or dimethylaminopyridine (DMAP), pyrrolidinopyridine (PPY), triethylamine, 4-morpholinopyridine or any other substituted base.
- DMAP dimethylaminopyridine
- PY pyrrolidinopyridine
- triethylamine triethylamine
- 4-morpholinopyridine 4-morpholinopyridine or any other substituted base.
- N-Hydroxybenzotriazole (HOBt) or N-hydroxysuccinimide (HO-Su) can also be used, for example.
- the molar ratio of 3l relative to 2 can be between 1/1 and 4/1.
- the reaction can preferably be carried out under inert gas at a pressure close to atmospheric pressure, preferably at a temperature of between 0° C. and about 110° C.
- the solvents preferably used are organic solvents such as, for example, toluene and/or dichloromethane and/or chloroform and/or tetrahydrofuran and/or pyridine and/or dimethylformamide.
- n, R 5 , R 6 , R 8 and CTX— have the same meaning as above.
- the substituted carboxylic cycloethers of the type 3k, the substituted ethylenic tertiary (-hydroxy acids of the type 3f, their activated intermediates, and cyclic anhydrides of the type 3p, can be coupled with the cephalotaxines, either in the racemic series, or, more advantageously, in the optically active series.
- n, R 6 , R 8 and CTX— have the same meaning as above, can be carried out by preparative chromatography either in a so-called normal phase, for example on native silica gel as stationary phase and a mixture of organic solvents as mobile phase, or, preferably, in a reverse phase, for example an inert silica grafted with apolar groups such as, for example, organosilyl, cyanoalkyl, phenylalkyl, preferably ocatadecylalkylsilane, chains and a mixture of aqueous solvents as mobile phase.
- a so-called normal phase for example on native silica gel as stationary phase and a mixture of organic solvents as mobile phase
- a reverse phase for example an inert silica grafted with apolar groups such as, for example, organosilyl, cyanoalkyl, phenylalkyl, preferably ocatadecylalkylsilane, chains and a mixture
- R 5 , R 6 , R 8 and CTX— have the same meaning as above, X being a heteroatom such as a halogen.
- the cyclic ethers of the type 4c can, in certain examples, have the particular feature of simultaneously containing a methyl enol ether, for example in position 2 of the cephalotaxines.
- a methyl enol ether for example in position 2 of the cephalotaxines.
- an organic solvent preferably a chlorinated solvent such as, for example, dichloromethane, dichloroethane or chloroform, under controlled conditions, i.e.
- the enol can easily be selectively remethylated as described in the literature (for example by dissolution in methanol in the presence of para-toluenesulphonic acid).
- Another variant consists in placing the product of type 4c in contact with an aqueous acid optionally in a miscible or immiscible co-solvent.
- the acid can be, for example, protonic, and in this case it is an organic or inorganic acid, preferably hydrochloric, tetrafluoroboric or formic acid.
- the diols of the type 4b can be isolated directly without passing through the halo alcohol stage described above.
- n, R 6 , R 8 , R 5 , X and CTX— have the same meaning as above.
- the halo alcohols of the type 4d are particularly suitable for controlled hydrolysis by placing in contact with an aqueous inorganic base such as, for example, dilute sodium hydroxide, sodium carbonate or, preferably, barium carbonate, at a temperature of between 0° C. and 30° C. with stirring, to give the diols of the type 4b.
- an aqueous inorganic base such as, for example, dilute sodium hydroxide, sodium carbonate or, preferably, barium carbonate
- One variant consists in carrying out the in situ hydrolysis of the intermediate halo alcohols of the type 4d at the end of the reaction, preferably carried out by treating the cyclic ether of the type 4c in dichloromethane in the presence of hydrobromic acid in acetic acid, and by directly adding the water or the hydrolysis solution to the reaction medium while cold and with vigorous stirring.
- the process according to the invention is particularly suitable for the preparation of azaharringtonines, nitrogenous analogues of the harringtonines.
- these materials were, for example, subjected to azidolysis by treatment using, for example, an alkaline azide in a solvent such as, for example, ethanol, methanol or dimethylformamide, which, by hydrogenolysis in solution in an organic solvent such as, for example, an alkanol or a lower ester, lead to the corresponding amino alcohol corresponding to the formula 4f (aminodeoxyharrington-ine).
- the amine can then be subjected to amidation under the Schotten-Baumann conditions, i.e. in aqueous media in the presence of an inorganic base as catalyst, to give an amido alcohol of the type 4 e
- Z ⁇ NHCOR or NHCOAr, R and Ar being as defined above, more specifically alkyl or aryl groups, respectively, which may or may not be substituted.
- the amine 4f can also be sec-alkylated to give an alkylaminodeoxyharringtonine (Z ⁇ NHR or Z ⁇ NHAr or Z ⁇ NR 2 or Z ⁇ NHAr 2 , it being possible for the two radicals R and Ar to be identical or independent), or acylated to give amides (Z ⁇ NHCOR or Z ⁇ NHCOAr) or carbamates (Z ⁇ NHCOOR) derived from the corresponding aminodeoxyharringtonine 4f, n, R 5 , R 6 , R 8 , R 9 , X and CTX— being defined as above.
- the cyclic ethers of the type 4c can be suitable for the Ritter reaction in the presence of a nitrile (which can serve as solvent) at a low temperature of between ⁇ 100° C. and +30° C. in the presence of an acid such as sulphuric acid, perchloric acid or, preferably, tetrafluoroboric acid, to give an acylaminodeoxyharringtonine (Z ⁇ NHCOR or Z ⁇ NHCOAr) derived from the corresponding aminodeoxyharringtonine 4f.
- a nitrile which can serve as solvent
- an acid such as sulphuric acid, perchloric acid or, preferably, tetrafluoroboric acid
- cephalotaxines of the type 2 is carried out according to the procedure indicated below.
- cephalotaxines of the type 2 can be prepared according to the methods described in the literature, either by synthesis or by extraction. In the latter case, since no method uses a direct placing in contact of the plant starting material with an aqueous acid, it has been found to be advantageous to describe this in the present invention.
- the fresh or dry plant starting material is placed in contact for 24 h with an acidified aqueous-organic mixture using a dilute inorganic acid or a weak organic acid, so as to bring the pH to between 1 and 4, preferably 3.
- the inorganic acid can be, for example, sulphuric acid or hydrochloric acid and the organic acid can be citric acid, lactic acid or tartaric acid, for example; the organic solvent can be, for example, a lower alkanol, a ketone, tetrahydrofuran or any other water-miscible solvent used in extraction by those skilled in the art.
- the water content is between 20 and 80%, preferably 50%.
- the solution obtained can be directly chromatographed or basified in order to be counter-extracted, since, in contrast with the methods described in the literature, it contains no chlorophyll and/or plant fat.
- the counter-extraction using a water-immiscible organic solvent such as a lower ester or, preferably, a lower halogenated hydrocarbon, more particularly dichloromethane gives a mixture of total alkaloids isolated in the form of a white powder.
- a water-immiscible organic solvent such as a lower ester or, preferably, a lower halogenated hydrocarbon, more particularly dichloromethane.
- cephalotaxine which forms an integral part of the novel process for the semi-synthesis of harringtonines, thus involves reverse-phase chromatography, which has never been used for this purpose.
- This reverse-phase chromatography uses as stationary phase, for example, an inert silica grafted with apolar groups such as, for example, organosilyl, cyanoalkyl, phenylalkyl or, preferably, octadecylalkylsilane chains such as those encountered commercially, and a mixture of aqueous solvents as mobile phase, preferably water itself (without organic solvent); the pH is adjusted, i.e. to between 2 and 4, with an inorganic acid such as hydrochloric or phosphoric or sulphuric acid. It is also advantageous to add an additive such as, for example, aqueous ammonia or triethylamine. According to this process, which is economically very advantageous since it avoids the use of organic solvent and allows the reuse of the stationary phase for virtually hundreds of operations, the cephalotaxine is obtained in a quantitative recovery yield and with a purity of greater than 95%.
- apolar groups such as, for example, organosilyl, cyan
- the above method makes it possible to obtain not only laevorotatory cephalotaxines naturally present in the plant material, but also racemic cephalotaxines also present in the natural state.
- M is a metal, more particularly an alkali metal such as sodium, potassium or lithium, or a transition metal, for example zirconium, titanium or zinc
- alkoxide is of great value for acylating more readily in this form the hindered hydroxyls of the cephalotaxanes and most particularly for coupling this cephalotaxane with acylating precursors of the side chains, automatically leading to the harringtonines which are the subject of the present invention.
- a metal hydride, an alkyl-metal, an amide or, more generally, any agent capable of exchanging or of giving up a metal atom can be used.
- a metal hydride such as, for example, potassium hydride, lithium hydride or, more particularly, sodium hydride
- a cephalotaxane metal alkoxide which can, for example, serve as an in situ substrate in order to attach, for example, a suitably substituted alkyl, acyl or alkylsilyl group.
- the organic solvent can be a suitable aprotic solvent such as an ether, more particularly tetrahydrofuran, a liquid aromatic hydrocarbon, preferably toluene or, more generally, any organic solvent which is liquid under the temperature and pressure conditions used and which has no appreciable reactivity towards the reagent.
- the temperature of the reaction medium can be between ⁇ 90° C. and +30° C.
- the preparation of these carboxylic cycloethers can be carried out by cyclization of the substituted ethylenic tertiary (-hydroxy acids of the type 3f according to the following scheme:
- substituted carboxylic cycloethers of the type 3k such as, for example, A or B of formulae:
- the preparation of these carboxylic cycloethers can be carried out by cyclization concomitant with the formation of the acylating species.
- acids whose tertiary alcohol is free such as 3f, cyclize spontaneously by the action of a dehydrating agent required for a certain technique for acylating a cephalotaxine alcohol mentioned above and then esterifying the latter to give 4c.
- the ethylenic tertiary alcohol of the 3f type leads, under anhydrous operating conditions, to the isolation of the acylating intermediate 3l, mentioned above, or, by hydrolysis, to the isolation of the acids of the type 3k.
- the procedure used is strictly the one described for coupling involving the formation of an acylating species in situ but in the absence of a substrate of the cephalotaxine type.
- these substituted carboxylic cycloethers of the type 3k can be prepared by deprotecting the tertiary carboxyl of the suitably substituted precursor 3h,
- R 5 , R 8 , R 6 and n being defined as above and GP representing a protecting group for the acids, with, as a specific case, GP ⁇ R.
- R 8 , R 6 and n being defined as above, can be prepared by total saponification of the corresponding diesters 3i such that R 5 ⁇ CH 2 CO 2 R, followed by mild selective methylation of the intermediate diacid 3r such that R 5 ⁇ CH 2 CO 2 H.
- R 8 , R 6 , GP and n being defined as above, which can be coupled with the cephalotaxines using the methods described above in order to ensure the absence of transesterification during the coupling of the tertiary acids which form the subject of the present invention.
- the preparation of these carboxylic cycloethers can be carried out by regioselective methanolysis of the corresponding cyclic anhydride.
- the diacid 3r leads, by self-dehydration, to the cyclic anhydride 3p, which is a good acylating agent for alcohols, such that by methanolysis 3k is also preferentially obtained such that R 5 ⁇ CH 2 CO 2 Me, which constitutes an additional preparation method.
- the diacids of the type 3s such that R 5 ⁇ CH 2 CO 2 H can be obtained by saponification of the corresponding diesters of the type 3g such that R 5 ⁇ CH 2 CO 2 Me by placing the latter in contact with an excess of base in an aqueous or aqueous-alcoholic medium,
- the alcohol can be a lower alcohol such as methanol, ethanol or, preferably, isopropanol
- the base can be, for example, an alkali metal or alkaline earth metal base or a rare earth metal hydroxide or aqueous ammonia.
- the diacids obtained above can then be converted into substituted ethylenic tertiary (-hydroxy acids of the type 3f such, that R 5 ⁇ CH 2 CO 2 Me by selective mono-esterification of their primary carboxyl, using methanol in the presence of a protonic acid or a Lewis acid or alternatively using the boron trifluoride etherate/methanol complex or diazomethane.
- the substituted ethylenic tertiary (-hydroxy esters of the type 3g can be prepared according to the scheme outlined below:
- R 6 , R 8 , m and R 5 having the same meaning as above.
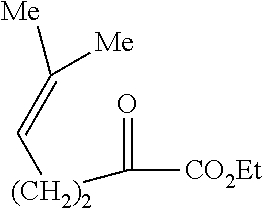
- ethylenic esters of the type 3g can be prepared according to the numerous methods described in the literature for similar cases such as, for example, (-hydroxyalkylation of the corresponding 1-alkyl- or 1-alkenyl-1-keto ester of the type 9.
- the (-hydroxyalkylation reaction of the 1-alkyl- or 1-alkenyl-1-keto ester of the type 9, conducted in the presence of a chiral inducing agent such as sparteine or quinine can give a significant enantiomeric enrichment, which can be further enhanced by fractional crystallization.
- keto esters of the type 9 are themselves conventionally obtained by C-semi-acylation of the carbanion of the corresponding alkyl or alkenyl halides of the type 10 with a dialkyl oxalate.
- an epimeric mixture is formed by combination with a chiral alcohol or amine.
- the reactions for esterifying a hindered secondary alcohol function of a cephalotaxine with oxacycloalkanecarboxylic acids of the type 3k above can also be applied to the esterification of another chiral alcohol in order to convert a racemic mixture, or one with partial enantiomeric enrichment of acids of the type 3k, into a diastereoisomeric mixture on which all of the non-chiral separation methods become applicable.
- the above methods are also applicable without modification to the amidation of chiral primary or secondary amines.
- oxacycloalkanecarboxylic acids of the type 3k or their ethylenic linear precursors are reacted with a chiral alcohol, denoted by R*OH, or an amide R′*R*NH (it being possible for R′* to be replaced with a hydrogen)
- R*OH a chiral alcohol
- R′*R*NH an amide
- the alcohol or the amide must preferably be hindered and bear their chiral centre at their site of binding with the tertiary carboxyl of the acid of the type 3k.
- the alcohol can be, for example, menthol, borneol, valinol or, preferably, quinine.
- the amine can be, for example, ephedrine; more generally, any commercial chiral alcohol or amide can be used.
- R 5 , R 6 , R 8 and A having the same meaning as above, R* having the same meaning as R, but being chiral.
- ( ⁇ )-quinine which, like the cephalotaxines, is an alkaloid with a sterically hindered secondary alcohol function, reacts with the racemic mixture of the oxacycloalkanecarboxylic acids of the type 3k to give the mixture of the two corresponding epimers 3v such that R 5 ⁇ CH 2 CO 2 Me and 3w such that R 5 ⁇ CH 2 CO 2 Me:
- the binary mixtures of epimers obtained by the combination with a chiral compound can be separated, for example, by fractional crystallization, by distillation, by counter-current liquid-liquid partition and, given the high added value of these intermediates, by any common preparative chromatography technique, for example normal phase chromatography, exclusion chromatography, preferably in reverse or normal grafted phase. Since these methods are synergistic, they can advantageously be combined in order to improve the diastereoisomeric purity.
- the regeneration of the oxacycloalkane-carboxylic acids of the type 3k in enantiomerically pure form can be carried out by total hydrolysis followed by selective remethylation of the primary carboxyl of the suitably selected diastereoisomer (see above sequence 3j (3k or, when it is an ester bond with an oxygen in the benzyl position (see for example quinine above), by simple hydrogenolysis.
- sequence 3j 3k or, when it is an ester bond with an oxygen in the benzyl position (see for example quinine above
- the double saponification of 3v followed by selective remethylation gave a product which was entirely identical to the (2R)-anhydroharringtonic acid of the type 3k above.
- the enantiomer of non-natural configuration (2S) can, after having undergone the same conversions as its (2R) enantiomer, be exploited, for example, for the purposes of structure-activity relationship studies.
- the racemic mixtures are resolved by formation of salts with a chiral basic species.
- the racemic mixtures of oxacycloalkane carboxylic acids of the type 3k can form a salt with a chiral amine by simple placing together in solution in an organic solvent.
- oxacycloalkane carboxylic acids of the type 3k including those formed in situ from ethylenic tertiary (-hydroxy acids of the type 3f)
- most of the methods described above for separating the esters and amides formed with 3k are applicable (for example chromatography), since the salts formed are generally highly crystallogenic, it is fractional crystallization which is preferably carried out to resolve the acids of the type 3k.
- the solvents used, alone or as a mixture can preferably be polar organic solvents which may or may not be combined with water, such as, for example, ketones, alcohols and lower esters.
- the reaction to form the salt preferably takes place at a temperature of between 0 and 100° C.
- the recrystallization can be carried out by redissolving the salt in a mixture whose solvent power can be adjusted with precision by means of the use of the above combinations of solvents and by varying the temperature according to the standard techniques practised by those skilled in the art.
- the salt is decomposed in the presence, for example, of a dilute aqueous acid such as hydrochloric acid or sulphuric acid.
- the extraction of the enantiomer of the regenerated acid can be carried out using a water-immiscible organic solvent such as, for example, a lower ester.
- racemic mixture of (2R or 2S)-anhydroharringtonic acids of the type 3k can be resolved, for example, by placing them in contact with ( ⁇ )-ephedrine, followed by fractional recrystallization in an ethyl acetate/methanol mixture.
- the 2R-anhydroharringtonic acid of the type 3k is then regenerated by placing the purified salt in contact with 2N hydrochloric acid and continuously extracting the acidic aqueous phase with ethyl acetate.
- oxacycloalkanecarboxylic acids of the type 3k described above can be subjected to preparative chiral chromatography.
- the final products are purified by HPLC to give final products for pharmaceutical use.
- the diastereoisomeric purity (with, as a specific case, the enantiomeric purity) can, moreover, lead to therapeutic aberrations; for example, it is well known that quinine (see above formula) is an antimalaria agent, whereas one of its diastereoisomers is a cardiac antifibrillant.
- R 6 , R 8 , R 5 , n and CTX— being defined as above, can be freed of its epimer at the same time as its other related impurities by preparative reverse-phase chromatography using a grafted reverse phase of octadecylsilane type as stationary phase and a suitably adjusted methanol/water mixture as mobile phase.
- This process gives a product whose sum of related impurities is less than 0.5% and for which none of these impurities taken individually exceeds 0.1%.
- the present invention concerns a process for the preparation of sidechain-bearing cephalotaxane of the following formula and/or a salt thereof ⁇ -CO—O—CTX where ⁇ (“omega”) is a representative radical of the chain terminal moiety and —CO— is the carbonyl of the ester group bonded to cephalotaxane; the ⁇ -CO— radical is corresponding:
- n is included between 0 and 8;
- Z is oxygen, nitrogen or sulfur heteroatom;
- R 5 , R 6 and R 8 are independently hydrogen
- hydrocarbon radical saturated, insaturated or aromatic, linear or ramified and/or cyclic, especially alkyl, alkenyl, alkynyl, cycloalkyl, cycloalkenyl, aryl, heterocycloalkyl, of said radical including or not heteroatom(s);
- R 6 and R 8 may be included in a cycle;
- R 5 , R 6 and R 8 are as defined above;
- n, R 5 , R 6 and R 8 are as defined above; Z and Q 2 are independently oxygen, nitrogen or sulfur heteroatom; Q1 is carbon, silicium or phosphorus atom; R 9 and R 10 are independently hydrogen, alkoxy, hydrocarbon radical, including or not heteroatom(s), saturated, unsaturated or aromatic, linear or ramified and/or cyclic, especially alkyl, alkenyl, alkynyl, cycloalkyl, cycloalkenyl, aryl, heterocycloalkyl; R 9 and/or R 10 having the ability to be null or taken together to make an heteroatom and/or make a multiple bond with Q 1 , R 9 and R 11 having the ability to be null to make a multiple bond between the two atoms of carbon bearing them; and R 11 is hydrogen, arylcarbonyl, alkoxycarbonyl, aryloxycarbonyl or alkylcarbonyl; where
- CTX is cephalotaxine moiety of the following formula a salt thereof: where p is equal to 1 or 2; the two types of radicals - ⁇ and —CTX above-mentioned being bonded with an ester bond —CO—O— the said process bringing together:
- ⁇ -CO of the following formula:
- A represents:
- Y—O—CTX an activated form of its hydroxyl group of the formula Y—O—CTX, where —O—CTX is as defined above and Y is, either a leaving group to allow a negative charge on oxygen atom by cleavage between Y— and —O—CTX, or to allow a carbocation by cleavage between Y—O— and —CTX;
- Z is an oxygen atom and the cephalotaxane H—O—CTX is a cephalotaxine of the following formula, or a salt thereof:
- R 1 , R 2 , R 3 and R 4 are independently hydrogen, hydroxyl group or alkoxide.
- a cephalotaxane H—O—CTX is cephalotaxine, or a salt thereof, where R 1 is hydroxyl, R 2 is methoxyl, R 3 and R 4 are hydrogen.
- R 5 is preferably hydrogen or —CH 2 —CO—O-Me.
- R 5 is —CH 2 —CO—O-Me
- R 1 ⁇ OH, R2 OMe, R 3 ⁇ R 4 ⁇ H
- A may be ⁇ -CO—CO where ⁇ is defined as above, or an halide.
- A may also be a radical of compound ⁇ -CO-A having the ability to generate cleavage of the bond between carbonyl group and substituent A to provide ⁇ -CO + and A ⁇ .
- A is a radical selected from substituents:
- A may also be 2,4,6-trichlorobenzoyloxy or a radical corresponding to the following formula:
- the reagent of formula ⁇ -CO-A is obtained by contacting an acid ⁇ -CO—OH, as defined above, with 2,4,6-trichlorobenzoyloxy carbonyl-diimidazole in presence of a strong base such as an alkoxide.
- the coupling additive is a substituted carbodiimide and/or a basic additive such as tertiary amine for example.
- the substituted carbodiimide is selected from cyclohexylcarbodiimide (DCC), 1,3-diisopropylcarbodiimide (DIC) and chlorhydrate of 1-(3-dimethylaminopropyl)-3-ethyl-carbodiimide.
- cephalotaxine alcoxide corresponding to the formula M-O—CTX where M and CTX are as defined above, may be obtained by contacting a cephalotaxine of formula H—O—CTX with metal himself, an amidure, a metallic hydride or an alkyl-metal.
- M may be an alkaline metal such as lithium, potassium or sodium.
- the aim of the present invention is also the preparation of new compounds such as:
- n is included between 0 and 8;
- Z is oxygen, nitrogen or sulfur heteroatom;
- R 5 , R 6 and R 8 are independently hydrogen
- hydrocarbon radical saturated, insaturated or aromatic, linear or ramified and/or cyclic, especially alkyl, alkenyl, alkynyl, cycloalkyl, cycloalkenyl, aryl, heterocycloalkyl, of said radical including or not heteroatom(s);
- CTX is as defined above;
- n 2 or 3
- R 6 ⁇ R 8 methyl and R 5 ⁇ OMe or hydroxyl
- n 3 and simultaneously R 6 is hydroxyl, when R 8 is methyl and R 5 is —CH 2 CO 2 CH 3 radical.
- R 5 is preferably the —CH 2 —CO—O—CH 3 radical.
- n, Z, Q 1 , Q 2 , R 5 , R 6 , R 8 , R 9 , R 10 , R 11 and CTX are as defined above.
- n, R 5 , R 6 , R 8 , CTX and Z are as defined above, the said chain is open with an agent and/or a protonic or not protonic electrophilic radical E in aqueous or not aqueous medium, to provide an intermediate compound of the following formula:
- n, CTX, R 5 , R 6 and R 8 are as defined above, E is either hydrogen or the provisionally or definitively fixed eletrophilic radical; the aforementioned intermediate compound may be attacked with an agent or a nucleophilic radical Z′, deliberately added or possibly present in the medium, and when the cyclic side-chain of sidechain-bearing cephalotaxane, and/or a salt thereof, presents the following formula:
- n, R 5 , R 6 , R 8 , R 9 , R 10 and R 11 are as defined above, and Z′ is an heteroatom; the said chain is open by hydrolysis or carefully solvolysis with possibly presence of activation and/or opening additive.
- cephalotaxine esters of the following formulas:
- R 5 , R 6 , R 8 , Z′, X and CTX are as defined above;
- CTX is as defined above
- CTX is as defined above
- n, R 5 , R 6 , R 8 , CTX and Z are as defined above, the said chain is open by treatment with a solution of hydrobromic acid in acetic acid, in an halogenated solvent, preferably dichloromethane, followed by in situ hydrolysis to provide, without isolation of the intermediate, a sidechain-bearing cephalotaxane of the following formula:
- n CTX, R 5 , R 6 et R 8 are defined above.
- ⁇ -CO— is:
- n, Z, R 6 , R 8 , and R 5 are as defined above;
- n, R 5 , R 6 , R 8 , Z, Q 2 , Q 1 , R 9 , R 10 and R 11 are as defined above.
- the stable combination may be represented by an ester of the following formula ⁇ -CO—O- ⁇ * such as ⁇ and ⁇ * are as defined above, the said stable combination is obtained by contacting acid with a chiral alcohol corresponding to the formula HO- ⁇ * such as ⁇ * is as defined above, according the process of invention.
- the stable combination may be represented by an thioester of the following formula ⁇ -CO—S- ⁇ * such as ⁇ and ⁇ * are as defined above, the said stable combination is obtained by contacting acid with a chiral thiol corresponding to the formula HS- ⁇ * such as ⁇ * is as defined above, according the process invention.
- the ionic combination may be represented by a salt just prepared by contacting of acid with a chiral amine corresponding to the either one or the other of the three following formulas: ⁇ -CO—O ⁇ [NH- ⁇ *] + ⁇ -CO—O ⁇ [NH 2 - ⁇ *] + ⁇ -CO—O ⁇ [NH 3 - ⁇ *] + where ⁇ and ⁇ *. are as defined above.
- the bringing into play of a labil bonding based combination is achieved in the form of chromatography with the help of a chiral stationary phase.
- the bringing into play of an interatomic or intermolecular labil bonding based combination, within crystalline lattice, is achieved in the form of fractionated crystallization initiated by a chiral precursor.
- the chiral amine H 2 N- ⁇ * is ( ⁇ )- or (+)-ephedrine corresponding to the following formulas:
- the present invention concerns the following new compounds:
- the said acid is obtained by treatment in aprotic or protic solvant, eventually in the presence of cyclization additive and/or dehydrating agent, the said treatment eventually supported with physical carrying of the water formed.
- R 12 is not a CTX radical as defined above and represents R 5 and/or a protective group of acids and/or a chiral group; then R 12 is removed later, either just by saponification, or by hydrogenolysis, or more generally by the method of the state of art to remove protective groups of acids.
- the cyclization additive is a protic acid such as sulfonic or formic acid, or an aprotic acid, included in immobilized form.
- Z is an oxygen atom.
- the aim of the present invention is also the preparation of the following new compounds:
- the tertiary heterocycloalcane carboxylic acid included its salts and each one of its pure enantiomeric forms or in racemic mixture or in variable composition, corresponding to the following formula:
- R 5 , R 6 and R 8 are as defined above, but are not hydrogen simultaneously. except for compounds corresponding to the exceptions 1 to 6 defined above.
- the tertiary heterocycloalcane carboxylic acid included its salts and each one of its pure enantiomeric forms or in racemic mixture or in variable composition, corresponding to the following formula:
- n is included between 0 and 8
- Z, R 5 , R 6 and R 8 are as defined above, R 5 is not hydrogen, and R 12 is not a CTX radical defined above; except for compounds where Z is oxygen atom and, corresponding to the exceptions 1 to 6 defined above.
- the tertiary oxacycloalcane carboxylic hemiester included its salts and each one of its pure enantiomeric forms or in racemic mixture or in variable composition, corresponding to the following formula:
- R 6 and R 8 are as defined above. except for compounds corresponding to the exceptions 1 to 6 defined above.
- the tertiary oxacycloalcane carboxylic hemiester included its salts and each one of its pure enantiomeric forms or in racemic mixture or in variable composition, corresponding to the following formula:
- R 6 and R 8 are as defined above
- R 12 is an hydrocarbon radical different from CTX as defined above. except for compounds corresponding to the exceptions 1 to 6 defined above.
- the tertiary oxacycloalcane carboxylic hemiester or anhydro-homoharringtonic acid included its salts and each one of its pure enantiomeric forms or in racemic mixture or in variable composition, corresponding to the following formula:
- the tertiary oxacycloalcane carboxylic hemiester or anhydro-harringtonic acid included its salts and each one of its pure enantiomeric forms or in racemic mixture or in variable composition, corresponding to the following formula:
- the tertiary oxacycloalcane carboxylic acid included its salts and each one of its pure enantiomeric forms or in racemic mixture or in variable composition, corresponding to the following formula:
- n and R 5 are as defined above, except for compounds corresponding to the exceptions 1 to 6 defined above.
- the tertiary oxacycloalcane carboxylic acid included its salts and each one of its pure enantiomeric forms or in racemic mixture or in variable composition, corresponding to the following formula:
- n is included between 1 and 8.
- the tertiary oxacycloalcane carboxylic acid included its salts and each one of its pure enantiomeric forms or in racemic mixture or in variable composition, corresponding to the following formula:
- n is included between 0 and 8.
- tertiary oxacycloalcane carboxylic acid or oxanhydroneoharringtonic acid included its salts and each one of its pure enantiomeric forms or in racemic mixture or in variable composition, corresponding to the following formula:
- the tertiary oxacycloalcane carboxylic acid or oxanhydroneohomoharringtonic acid included its salts and each one of its pure enantiomeric forms or in racemic mixture or in variable composition, corresponding to the following formula:
- the tertiary oxacycloalcane carboxylic acid included its salts and each one of its pure enantiomeric forms or in racemic mixture or in variable composition, corresponding to the following formula:
- the tertiary alkene carboxylic acid included its salts and each one of its pure enantiomeric forms or in racemic mixture or in variable composition, corresponding to the following formula:
- R 6 and R 8 are as defined above, but are not hydrogen simultaneously, and R 5 is not hydrogen or heteroatom.
- the tertiary alkene carboxylic acid included its salts and each one of its pure enantiomeric
- the tertiary alkene carboxylic acid included its salts and each one of its pure enantiomeric forms or in racemic mixture or in variable composition, corresponding to the following formula:
- R 6 and R 8 are as defined above but are not hydrogen.
- the tertiary alkene carboxylic acid included its salts and each one of its pure enantiomeric forms or in racemic mixture or in variable composition, corresponding to the following formula:
- the tertiary alkene carboxylic acid included its salts and each one of its pure enantiomeric forms or in racemic mixture or in variable composition, corresponding to the following formula:
- the tertiary alkene carboxylic acid included its salts and each one of its pure enantiomeric forms or in racemic mixture or in variable composition, corresponding to the following formula:
- R 6 and R 8 are as defined above.
- the sidechain-bearing cephalotaxane was purified like a salt by chromatography using a hydrophobic reversed-phase like stationary phase, and a mobile phase without organic solvent like a solution adjusted to a pH 2 to 4.5 with a buffer prepared with an acid and an alkaline or ammonium salt and one or several additive like attenuator of silanol effect, the said cephalotaxine salt was generated from mineral acid under the form of chlorohydrate, sulfate, phosphate, nitrate, perchlorate, or from organic acid under the form of tartrate, malate, citrate or lactate.
- the sidechain-bearing cephalotaxane was purified by a step of chromatographic purification of a natural or semi-synthetic or synthetic homoharringtonine as a for pharmaceutical use, corresponding to the following formula and having the chemical name, cephalotaxine, 4-methyl (2R)-2-hydroxy-2-(4-hydroxy-4-methylpentyl)butanedioate (ester):
- This process of purification and chromatographic control of a natural or semi-synthetic or synthetic homoharringtonine allows to offset the double makes it possible to overcome the two fold insufficiency of enantiomeric purity of the semi-precursors thereof, i.e., both on the sidechain precursor (said referred to as homoharringtonic acid) and the cephalotaxine, whether or not the two said-precursors semi-precursors are each independently generated by total synthesis or semi-synthetic process or natural process within of in the plant (biosynthesis), in fact the withdrawal i.e. the removal of the non natural enantiomer of homoharringtonine showing an having the opposite absolute configuration, by using a chiral stationary phase with on a preparative scale.
- the table p. 157 recapitulates process of preparation of in columns 99-102 summarizes the processes for preparing harringtonines of the prior art.
- FIGS. 1A and 1B gives the sequence of synthesis of homoharringtonine corresponding to the Example 25, where A represents a 2,4,6-trichloro-phenyl group, R represents a methyl and R′ represents a cephalotaxyl moiety.
- FIGS. 2A and 2B represents a variant of the process according to the invention, more exactly the semi-synthesis of harringtonines via oxacycloalcalne carboxylic acids.
- the present invention also concerns highly purified and crystalline form of harringtonines, definite by their solid state analysis patterns, their process of preparation by purification of crude alkaloids from natural, synthetic or semi-synthetic sources, allowing their use as drug substance for blending alone or in combination in pharmaceutical composition particularly useful for treatment of cancer in using oral mode of administration.
- cephalotaxine esters alkaloids isolated from rare and endangered conifers belonging to the Cephalotaxus genus.
- Cephalotaxine and its natural ester are gathered under the generic term of cephalotaxane.
- CML Chronic Myelogenous Leukemia
- Drug agencies such as the U.S. Food and Drug Administration, require a high level of purity including enantiomeric, before approval new agents, particularly when these agents are isolated from natural sources. For example, 0.1% an impurity must be qualified and toxicology studies must be performed. New drugs not enantiomerically pure or as racemic mixture are no longer approved by the FDA. In addition, due to large variation of related impurities profil under environmental conditions, drug agencies are particularly suspicious versus drug substances prepared from direct extraction of organisms.
- Homoharringtonine and harringtonine are present in Cephalotaxus extract as a complex mixture of several dozens of alkaloids (see scheme 1).
- HA and HHT were firstly used as a mixture for therapy of cancer and leukemia in China.
- the level of compliance of drug substances to a given quality required by the FDA is increasing during the process of development of an investigational drug (i.e. from early phase I clinical trial to phase III).
- FDA requires that the profile of impurities in term of related compounds shall be reproducible from batch to batch during the marketing phase, that is very difficult when the drug substance is prepared from natural source.
- An homoharringtonine prepared from natural source was developed by the U.S., the National Cancer Institute and used in its early clinical trials.
- the final drug substance contains three major impurities including two natural congeners and ethyl analog of HHT which is an artifact of purification resulting from transesterification with the solvent of purification.
- HHT ethyl analog of HHT
- FIGS. 8 , 9 , 12 , and 14 show a chromatographic profiles of harringtonines coming from various sources.
- harringtonines including harringtonine and homoharringtonine
- chromatographic purity of the final harringtonine and homoharringtonine drug substance is consistently higher then than 99.8% versus 98.5% for the above cited NCI products (the purest ever previously described) versus 95%-97% for Chinese products, corresponding to 0.2%, 1.5 1.5% and 3-5% of impurities (see FIGS. 6 , 7 and 12 8, 9 and 14).
- cephalotaxine as a precursor of semi-synthetic HA and HHT, is abundant in a renewable part of the tree, this semi-synthetic process overcome overcomes the serious environmental concern induce concerns induced by the destroying destruction of a rare and endanger endangered plant.
- a well definite crystalline form of a drug substance is a very important condition, to have reliable solid final form of drugs useful for example for oral administration.
- HHT and HA would be very promising drugs for the treatment of patients with CML
- the current mode of administration by continuous intravenous central infusion (CIVI) is a strong handicap for the administration of this therapy during several years.
- extra-hematologic toxicity of HHT/HA is very mild, the occurrence of infection due to catheter is the main toxicity of this regimen.
- the use of an oral form of these drugs could be change completely this situation and would extend widely the market of this product.
- the present invention provides natural, synthetic or semi-synthetic harringtonines including their tautomeric forms and their salts of the following formula:
- the total content of impurities, possibly including enantiomeric forms, is lower than 1%, and/or
- the content of the major impurity is lower than 0.9%, and/or
- the chromatographic assay exhibits a harringtonines content higher than 97.5%.
- a preferred embodiment of the invention provides a natural, synthetic or semi-synthetic homoharringtonine including its tautomeric forms and its salts in which:
- the total content of impurities, possibly including enantiomeric forms, is lower than 1%, and/or
- the content of the major impurity is lower than 0.9%, and/or
- the chromatographic assay exhibits a homoharringtonines content higher than 97.5%.
- a further preferred embodiment of the invention provides a natural, synthetic or semi-synthetic harringtonine including its tautomeric forms and its salts in which:
- the total content of impurities, possibly including enantiomeric forms, is lower than 1%, and/or
- the content of the major impurity is lower than 0.9%, and/or
- the chromatographic assay exhibits a harringtonine content higher than 97.5%.
- a further preferred aspect of the invention is a crystalline natural, synthetic or semi-synthetic homoharringtonine having substantially the same DSC curve as set out in FIG. 3 .
- a further embodiment of the invention provides a crystalline natural, synthetic or semi-synthetic homoharringtonine having substantially the same X-ray diffractogram as set out in FIG. 4 , and substantially the same IR spectrum, in KBr as set out in FIG. 5 .
- another embodiment of the invention provides a crystalline natural, synthetic or semi-synthetic homoharringtonine having substantially the same DSC curve as set out in FIG. 3 , and substantially the same X-ray diffractogram as set out in FIG. 4 , and substantially the same IR spectrum, in KBr as set out in FIG. 5 .
- Another preferred embodiment of the invention provides a crystalline natural, synthetic or semi-synthetic harringtonine having substantially the same DSC curve as set out in FIG. 6 .
- a preferred aspect of this invention provides a pharmaceutical composition
- a pharmaceutical composition comprising an effective antitumor amount of a natural, synthetic or semi-synthetic homoharringtonine having substantially the same X-ray diffractogram as set out in FIG. 4 , and substantially the same IR spectrum, in KBr as set out in FIG. 5 , and substantially the same DSC curve as set out in FIG. 3 , together with one or more pharmaceutically acceptable inactive components such as carriers, excipients, adjuvants or diluents.
- Another aspect of the invention provides a pharmaceutical composition
- a pharmaceutical composition comprising an effective antitumor amount of a natural, synthetic or semi-synthetic harringtonine having substantially the same IR spectrum, in KBr as set out in FIG. 7 , and substantially the same DSC curve as set out in FIG. 6 , together with one or more pharmaceutically acceptable inactive components such as carriers, excipients, adjuvants or diluents.
- Another preferred aspect of the invention provide a process of purification of natural, synthetic or semi-synthetic crude harringtonines for the preparation of pure harringtonines exhibiting the above included features including for eventual enantiomeric enrichment, and comprising the successive steps:
- the progression of the process of purification is monitored by HPLC analyses and several termal analysis at the solid state.
- the progression of enantiomeric purity is monitored by optical rotation checking of the dried solid form.
- Another preferred embodiment of the invention is the above process of purification in which the lower C 1-4 alkanol is methanol and the cephalotaxane is harringtonine
- a further preferred aspect of the invention is the above process of purification in which the lower C 1-4 alkanol is methanol and the cephalotaxane is homoharringtonine
- This invention include also a pharmaceutical composition which comprises an antitumor effective amount of at least one above described harringtonine or homoharringtonine with one or more pharmaceutically acceptable carriers, excipients or diluents therefore, including the process for preparing the said solid pharmaceutical composition such as, for examples, tablet, capsule, implant or suppository.
- Another aspect of the invention is the use of at least the above solid form of one harringtonine or homoharringtonine described in the invention for preparing the above pharmaceutical composition as (i) chemotherapeutic agent, (ii) enhancer of other chemotherapeutic agents (iii) for inhibiting tumors growth, (iv) for inhibiting mammalian parasites, (v) as immunosuppressive agent, or (vi) as reversal agent.
- the present invention further describes a method for treating mammalian tumors which comprises administering to a mammal an antitumor effective amount of the solid form of at least one harringtonine or homoharringtonine described in this invention, by parenteral, topic, subcutaneous or anal mode.
- a preferred embodiment of the invention describes a method for treating mammalian tumors which comprises oral administering to a mammal an antitumor effective amount of the solid form of at least one harringtonine or homoharringtonine described in this invention.
- a further preferred embodiment of the invention describes a method for treating mammalian tumors which comprises implantable pharmaceutical preparation administering to a mammal an antitumor effective amount of the solid form of at least one harringtonine or homoharringtonine described in this invention.
- the invention is also concerned with the use of purified and/or solid harringtonines as defined above, for the preparation of pharmaceutical compositions for the treatment of cancers and leukemias particularly acute myelod leukemia (AML), myelodysplastic syndrome (MDS) and myeloproliferative disorders including chronic myelogenous leukemia.
- AML acute myelod leukemia
- MDS myelodysplastic syndrome
- myeloproliferative disorders including chronic myelogenous leukemia.
- p-Toluenesulfonic acid (2.06 g, 10.8 mmol) was added to a stirred solution of ethylenic ester resulting from Example 1 (2.8 g, 10.8 mmol) in toluene (30 ml) and the resulting mixture was stirred at 65° C. for 5 hours. After cooling at room temperature, the mixture was hydrolyzed with saturated sodium hydrogen carbonate solution. The aqueous layer was extracted with ether (3 ⁇ 50 ml), and the organic layers were combined, washed with brine (100 ml), dried over magnesium sulfate and evaporated to dryness.
- IR (ATR) (cm ⁇ 1 ): 3490; 2966; 1731; 1193; 1177; 1152.
- IR (ATR) (cm ⁇ 1 ): 3500; 3019; 2966; 2931; 1716; 1691; 1656; 1219; 1199; 1111.
- the resulting crude product (3.9 g) was purified by column chromatography (dichloromethane/methanol (99:1), silica (15-40 ⁇ m) 160 g) to provide a yellow oil (3.1 g, 67%).
- the crude product thus obtained showed identical characteristics to this obtained above.
- DiastereomericA diastereomeric mixture of ( ⁇ )-quinyl(2′R)-anhydrohomoharringtonate and of ( ⁇ )-quinyl(2′S)-anhydrohomoharringtonate (5 g) was submit submitted to preparative HPLC. Above The mixture was dissolved in buffer (triethylamine (1.55/1000) in deionised deionized water and orthophosphoric acid to adjust pH to 3.
- IR (ATR) (cm ⁇ 1 ): 3488; 2935; 2105; 1748; 1654; 1486; 1223; 1034.
- IR (ATR) (cm ⁇ 1 ): 3299; 2935; 1740; 1654; 1486; 1222 et 1034.
- the resulting crude product (65 mg) was purified by column chromatography (dichloromethane/methanol (98:2), silica (15-40 ⁇ m) 2.5 g) to provide a white solid (41 mg, 56%).
- the product thus obtained showed identical characteristics to this obtained from method above-mentioned.
- the resulting crude product (2 g) was purified by column chromatography (cyclohexane/ethyl acetate (98:2), silica (15-40 ⁇ m) 80 g) to provide an oil (660 mg, 39%).
- the intermediate ⁇ -cetoester showed the following characteristics:
- the 1 H NMR spectra of the crude product showed a diastereomeric mixture ⁇ 60/40.
- the resulting crude product (820 mg) was purified by column chromatography (cyclohexane/ethyl acetate (97:3 to 95:5), silica (15-40 ⁇ m) 80 g) to provide the expected products (361 mg, 35%).
- the separated diastereomers thus obtained like white crystalline compounds showed the following characteristics:
- the second mother liquors allowed prisms composed of ( ⁇ )-cephalotaxine identical to this obtained at the first crystallization.
- the third mother liquors still allowed macled needles (urchins) composed of ( ⁇ )-cephalotaxine.
- Natural harringtonine (5 grams) is injected on a preparative high-pressure liquid chromatography (HPLC) system (Prochrom stainless steel; permanent axial compression; diameter: 80 mm; length: 1000 mm) containing 1000 grams of reverse phase octadecylsilane specially dedicated for basic compounds as stationary phase. Then elution is performed in using a gradient of pH 3 buffered methanol-water solution as mobile phase (pressure 1200 psi). Unwanted fractions are discarded based upon in-line UV spectrophotometric detection.
- HPLC high-pressure liquid chromatography
- Kept fractions are collected in 16 separate containers which each are individually checked in using an analytical HPLC system exhibiting a different selectivity pattern (octadecylsilane as stationary phase and buffered acetonitrile-water system as mobile phase).
- an analytical HPLC system exhibiting a different selectivity pattern
- octadecylsilane as stationary phase
- buffered acetonitrile-water system as mobile phase.
- a dual in-line UV-MS detection is used. After discarding of the fractions representing more than 0.5% of the total content of harringtonine, fractions which complied with pre-established specification were gathered, neutralized then evaporated under reduce pressure. Then crude concentrated solution of harringtonine are alkalinized at pH 8.5 with aqueous ammonia and partitioned with dichloromethane. Resulting organic solution is concentrated under high vacuum. In-process HPLC analysis indicated a total of related compound lower than 1.5%.
- the above raw harringtonine (4.1 grams) is dissolved in methanol (5 ml), at 30° C.
- methanol 5 ml
- the resulting alcoholic solution was filtered on a 0.25 ⁇ sterile Millipore filter to remove microparticles and germs and collected in a sterilized rotary flask.
- deionized water 50 mL
- methanol is completely removed under vacuum at 30° C. in using a decontaminated rotary evaporator.
- heating is stopped and the aqueous solution of harringtonine is kept under vacuum and rotation is continued during appearance of white crystals of pure harringtonine.
- the stirring is continued until no more crystal occurs.
- Routine analytical procedure includes solvent residues, loss on drying, water determination, melting point, IR and NMR spectrum, related compound and assay by HPLC.
- FIGS. 9 and 10 11 compare HPLC chromatogram before and after purification in using this process. Table II shows the comparison of the corresponding related compound content.
- DSC differential scanning calorimetry
- FIG. 7 shows typical infrared spectrum (KBr) for unambiguous identification at the solid state of the crystalline harringtonine obtained by this process.
- a series of sharp absorption bands are noted at 615, 654, 674, 689, 709, 722, 750, 761 805, 850, 928, 989, 1022, 1033, 1062, 1083, 1112, 1162, 1205, 1224, 1262, 1277, 1308, 1340, 1364, 1382, 1438 1486, 1508, 1625, 1656, 1725, 1745, 2883, 2936, 2972, 3079, 3353, 3552 and 3647 cm ⁇ 1
- Crude reaction mixture of raw homoharringtonine contains a potential of 250 grams of homoharringtonine DS together with process impurities such as catalyst, unchanged starting product (anhydro-homo-harringtonine), and some related side product.
- HPLC analysis with UV detection indicated a total of 9% of related impurities.
- Raw semi-synthetic homoharringtonine (550 grams) is injected on a preparative high-pressure liquid chromatography (HPLC) system (Prochrom stainless steel; permanent axial compression; diameter: 450 mm; length: 1000 mm) containing 48,000 grams of reverse phase octadecylsilane specially dedicated for basic compounds as stationary phase. Then elution is performed in using a gradient of pH 3 buffered methanol-water solution as mobile phase (pressure 1200 psi, flow-rate 540 L/hour). Unwanted fractions are discarded based upon by-passed in-line UV spectrophotometric detector.
- HPLC high-pressure liquid chromatography
- Kept fractions are collected in 30 separate stainless steel containers (20 or 50 L each) which are individually checked in using an analytical HPLC system exhibiting a different selectivity pattern (octadecylsilane as stationary phase and buffered acetonitrile-water system as mobile phase) and equipped with a diode array detector. After discarding of the fractions representing more than 0.5% of the total content of homoharringtonine, fractions which complied with pre-established specification were gathered, neutralized then evaporated under reduce pressure in using a mechanically stirred thin film evaporator. Then crude concentrated solution of homoharringtonine are alkalinized at pH 8.5 with aqueous ammonia and partitioned with dichloromethane. Resulting organic solution is concentrated under high vacuum. In-process HPLC analysis indicated a total of related compound lower than 0.5% (see right-side chromatogram on FIG. 12 )
- Routine analytical procedure includes solvent residues, loss on drying, water determination, melting point, IR and NMR spectrum, related compound and assay by HPLC.
- FIG. 13 shows HPLC chromatogram before and after crystallization. Total of related impurities of homoharringtonine DS is 0.03%.
- DSC differential scanning calorimetry
- TD thermogravimetry
- 2D NMR 2D NMR
- solid NMR X-ray powder diffractometry
- FIG. 5 shows typical infrared spectrum (KBr) for unambiguous identification at the solid state of the crystalline homoharringtonine obtained by this process.
- a series of sharp absorption bands are noted at 612, 703, 771, 804, 826, 855, 879, 932, 1029, 1082, 1119, 1135, 1161, 1191, 1229, 1274, 1344, 1367, 1436, 1457, 1488, 1505, 1653, 1743. 2814, 2911, 2958, 3420, and 3552 cm ⁇ 1
- X-ray powder diffraction pattern was collected on a INEL microdiffractomer microdiffractometer, model DIFFRACTINEL. Powdered homoharringtonine DS was packed in a glass capillary tube and was analyzed according to a standard method in the art.
- the X-ray generator was opered operated at 45 kV and 40 mA, using the copper Kalpha line as the radiation source. The sample was rotated along the chi axis and data was collected between 0 and 120 deg 2-theta. A collection time of 1200 sec was used. As showed on shown in FIG.
- the x-ray powder diffraction for this crystalline form of homoharringtonine shows a typical pattern including major reflection peaks at approximately 7.9, 9.2, 10.9, 14.9, 16.0, 17.7, 19.5, 19.7, 21.78, 23.1, 25.3, 25.4 and 25.7 deg 2-theta.
- Natural homoharringtonine (25 grams) is injected on a preparative high-pressure liquid chromatography (HPLC) system (Prochrom stainless steel; permanent axial compression; diameter: 200 mm; length: 1000 mm) containing 12,000 grams of reverse phase octadecylsilane specially dedicated for basic compounds as stationary phase. Then elution is performed in using a gradient of pH 3 buffered methanol-water solution as mobile phase (pressure 1200 psi, flow-rate 120 L/hour). Unwanted fractions are discarded based upon by-passed in-line UV spectrophotometric detector.
- HPLC preparative high-pressure liquid chromatography
- Kept fractions are collected in 22 separate stainless steel containers which are individually checked in using an analytical HPLC system exhibiting a different selectivity pattern (octadecylsilane as stationary phase and buffered acetonitrile-water system as mobile phase) and equipped with a diode array detector. After discarding of the fractions representing more than 0.5% of the total content of homoharringtonine, fractions which complied with pre-established specification were gathered, neutralized then evaporated under reduce pressure in using a mechanically stirred thin film evaporator. Then crude concentrated solution of homoharringtonine are alkalinized at pH 8.5 with aqueous ammonia and partitioned with dichloromethane. Resulting organic solution is concentrated under high vacuum. In-process HPLC analysis indicated a total of related compound lower than 0.5%.
- Routine analytical procedure includes solvent residues, loss on drying, water determination, melting point, IR and NMR spectrum, related compound and assay by HPLC.
- FIG. 14 (bottom right) shows HPLC chromatogram after crystallization. Total of related impurities of homoharringtonine DS is 0.05%.
- DSC differential scanning calorimetry
- TD thermogravimetry
- 2D NMR 2D NMR
- solid NMR X-ray powder diffractometry
- the mother liquors coming from the fractioned crystallization of example 3 areA solution containing (+)-homoharringtonine is concentrated then dissolved in methanol at 30° C. The resulting alcoholic solution is filtered on a 0.25 ⁇ sterile Millipore filter. Then, desionized deionized water is added and methanol is completely removed under vacuum at 30° C. in using a rotary evaporator. After removing methanol, heating is stopped and the aqueous solution of (+)-homoharringtonine is kept under vacuum and rotation is continued during appearance of white crystals of pure (+)-homoharringtonine. The stirring is continued until no more crystal crystalization occurs. The suspension of crystals is poured on a sintered glass filter with house vacuum. The resulting crystalline solid cake is washed two times with cold desionized deionized water (50 mL ⁇ 2). The white crystals are then dried using high vacuum at 60° C. for 48 hours.
- the (+)-homoharringtonine thus obtained present has all the analytical characteristics of the natural homoharringtonine (levogyrous), except its rotatory power which is equal but of opposite sign).
Landscapes
- Organic Chemistry (AREA)
- Chemical & Material Sciences (AREA)
- Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Chemical & Material Sciences (AREA)
- Tropical Medicine & Parasitology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Engineering & Computer Science (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Nitrogen Condensed Heterocyclic Rings (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Epoxy Compounds (AREA)
- Heterocyclic Carbon Compounds Containing A Hetero Ring Having Oxygen Or Sulfur (AREA)
- Pyrane Compounds (AREA)
- Cephalosporin Compounds (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
- Heterocyclic Carbon Compounds Containing A Hetero Ring Having Nitrogen And Oxygen As The Only Ring Hetero Atoms (AREA)
- Low-Molecular Organic Synthesis Reactions Using Catalysts (AREA)
- Nitrogen And Oxygen Or Sulfur-Condensed Heterocyclic Ring Systems (AREA)
Abstract
wherein n=2 (i.e. harringtonine) or n=3 (i.e. homoharringtonine),
in which the total content of impurities, possibly including enantiomeric forms, is lower than 1%, and/or the content of the major impurity is lower than 0.9%, and/or the chromatographic assay exhibits a harringtonines content higher than 97.5%.
Description
where p is equal to 1 or 2 (it being possible for the two units to be identical or different and linked via a single bond or an oxygen atom), which can contain various oxygenated substituents (aliphatic or aromatic ethers, free or esterified alcohols, substituted or free enols and/or phenols, bridged ethers, and more generally any substituent usually encountered in the natural state on compounds of this type).
- Homo: 1 extra carbon.
- Bishomo: 2 extra carbons.
- Nor: a sidechain with 1 carbon less.
- Iso: a sidechain with methylene bearing an hydroxyl group at the place of juxtaterminal carbon.
- Deoxy: the hydroxyl group of juxtaterminal carbon is replaced par hydrogen.
- Anhydro: the two tertiary hydroxyl groups lose a molecule of water to give the corresponding saturated oxygenated heterocycle.
- neo: R6 is a phenyl group at the place of 2-hydroxyisopropyl.
- HPLC: High-Performance Liquid Chromatography.
- NMR: Nuclear Magnetic Resonance.
in which n, R6 and R8 have the same meaning as above,
and which can be readily prepared from the corresponding diacids in order once again to give 3k by esterification of methanol or alternatively to become attached as above to the alcohol function of a cephalotaxine of the
- (a) esterification of the free acid with the alcohol in acid catalysis,
- (b) esterification by acyl transfer via anhydrides or halides,
- (c) esterification by acyl transfer using activated esters,
- (d) esterification with scandium triflate,
- (e) esterification with boron trifluoride etherate,
- (f) esterification by the thioester method.
in which n, R6 and R8 have the same meaning as above,
which can be prepared very simply by treating the corresponding diacid with acetic anhydride, for example under the general operating conditions for the preparation of the mixed anhydrides described below.
in which n, R6, R8 and CTX— have the same meaning as above,
can be carried out by preparative chromatography either in a so-called normal phase, for example on native silica gel as stationary phase and a mixture of organic solvents as mobile phase, or, preferably, in a reverse phase, for example an inert silica grafted with apolar groups such as, for example, organosilyl, cyanoalkyl, phenylalkyl, preferably ocatadecylalkylsilane, chains and a mixture of aqueous solvents as mobile phase.
such that Z═NHCOR or NHCOAr, R and Ar being as defined above, more specifically alkyl or aryl groups, respectively, which may or may not be substituted. The amine 4f can also be sec-alkylated to give an alkylaminodeoxyharringtonine (Z═NHR or Z═NHAr or Z═NR2 or Z═NHAr2, it being possible for the two radicals R and Ar to be identical or independent), or acylated to give amides (Z═NHCOR or Z═NHCOAr) or carbamates (Z═NHCOOR) derived from the corresponding aminodeoxyharringtonine 4f, n, R5, R6, R8, R9, X and CTX— being defined as above.
can be prepared from the substituted ethylenic tertiary (-hydroxy acids of the
R5, R8, R6 and n being defined as above and GP representing a protecting group for the acids, with, as a specific case, GP═R.
R8, R6 and n being defined as above,
can be prepared by total saponification of the corresponding diesters 3i such that R5═CH2CO2R, followed by mild selective methylation of the intermediate diacid 3r such that R5═CH2CO2H.
R8, R6, GP and n being defined as above,
which can be coupled with the cephalotaxines using the methods described above in order to ensure the absence of transesterification during the coupling of the tertiary acids which form the subject of the present invention.
by selective methylation of the primary carboxyl in particular, by placing the reactants in prolonged contact in methanolic solution at room temperature or using the boron trifluoride/methanol complex.
n, R5, R6, R8 and A having the same meaning as above, R* having the same meaning as R, but being chiral.
R6, R8, R5, n and CTX— being defined as above,
can be freed of its epimer at the same time as its other related impurities by preparative reverse-phase chromatography using a grafted reverse phase of octadecylsilane type as stationary phase and a suitably adjusted methanol/water mixture as mobile phase.
Ω-CO—O—CTX
where
Ω (“omega”) is a representative radical of the chain terminal moiety and —CO— is the carbonyl of the ester group bonded to cephalotaxane;
the Ω-CO— radical is corresponding:
where n, R5, R6 and R8 are as defined above;
Z and Q2 are independently oxygen, nitrogen or sulfur heteroatom;
Q1 is carbon, silicium or phosphorus atom;
R9 and R10 are independently hydrogen, alkoxy, hydrocarbon radical, including or not heteroatom(s), saturated, unsaturated or aromatic, linear or ramified and/or cyclic, especially alkyl, alkenyl, alkynyl, cycloalkyl, cycloalkenyl, aryl, heterocycloalkyl;
R9 and/or R10 having the ability to be null or taken together to make an heteroatom and/or make a multiple bond with Q1, R9 and R11 having the ability to be null to make a multiple bond between the two atoms of carbon bearing them; and
R11 is hydrogen, arylcarbonyl, alkoxycarbonyl, aryloxycarbonyl or alkylcarbonyl;
where
—O—CTX is cephalotaxine moiety of the following formula a salt thereof:
where p is equal to 1 or 2;
the two types of radicals -Ω and —CTX above-mentioned being bonded with an ester bond —CO—O—
the said process bringing together:
-
- either carboxylic acid with general formula Ω-CO—OH or a salt thereof;
-
- or an activated form of an acid with general formula Ω-CO-A or a salt thereof, with Ω-CO of the following formula:
where n, Z, R5, R6 and R8 are as defined above;
where Ω-CO of the following formula:
- or an activated form of an acid with general formula Ω-CO-A or a salt thereof, with Ω-CO of the following formula:
m is included between 1 and 8, Z, R5, R6 and R8 are as defined above;
where Ω-CO of the following formula:
where n, R6 and R8 are as defined above;
the reaction has been completed by methylation of the primary carboxyl thus formed, with:
where
m, R5, R6, R8 and CTX are as defined above;
except compound where m=2, R5═CH2CO2CH3, R6═R8=methyl and CTX is as defined above.
where n, CTX, R5, R6 and R8 are as defined above, E is either hydrogen or the provisionally or definitively fixed eletrophilic radical;
the aforementioned intermediate compound may be attacked with an agent or a nucleophilic radical Z′, deliberately added or possibly present in the medium, and
when the cyclic side-chain of sidechain-bearing cephalotaxane, and/or a salt thereof, presents the following formula:
where n, R5, R6, R8, R9, R10 and R11 are as defined above, and Z′ is an heteroatom;
the said chain is open by hydrolysis or carefully solvolysis with possibly presence of activation and/or opening additive.
where n, R5, R6, R8, CTX and Z are as defined above,
the said chain is open by treatment with a solution of hydrobromic acid in acetic acid, in an halogenated solvent, preferably dichloromethane, followed by in situ hydrolysis to provide, without isolation of the intermediate, a sidechain-bearing cephalotaxane of the following formula:
Ω-CO—OH
where Ω radical is as defined according above;
the said formula equivalent to racemic mixture containing compounds of the formulas (+)-Ω-CO—OH and (−)-Ω-CO—OH such as (+)-Ω-CO—OH represents its dextrogyre enantiomer and (−)-Ω-CO—OH represent its levogyre enantiomer, were obtained
a) by contacting of said racemic mixture or one of its activated form of the formula
Ω-CO-A
which is as defined above;
the said racemic mixture or said activated form generating respectively:
Ω-CO—O−[NH-Δ*]+
Ω-CO—O−[NH2-Δ*]+
Ω-CO—O−[NH3-Δ*]+
where Ω and Δ*. are as defined above.
the (−)-methylmandelyl (2′R)-(−)-anhydro-homoharringtonate and the (−)-methylmandelyl (2′S)-(−)-anhydro-homoharringtonate corresponding respectively to the two following formulas:
where n, Z, R5, R6 and R8 are as defined above, the said acid is obtained by treatment in aprotic or protic solvant, eventually in the presence of cyclization additive and/or dehydrating agent, the said treatment eventually supported with physical carrying of the water formed.
where m is included between 1 and 8, Z, R5, R6 and R8 are as defined above, R12 is not a CTX radical as defined above and represents R5 and/or a protective group of acids and/or a chiral group;
then R12 is removed later, either just by saponification, or by hydrogenolysis, or more generally by the method of the state of art to remove protective groups of acids.
where n, Z, R5, R6 and R8 are as defined above, and R5 is not hydrogen;
except for compounds where Z is oxygen atom and,
1°) n=0 and R5 is not —CH2CO2H or —CH2CO2CH3 radical;
2°) n=0 and R5 is —CH2CO2H or —CH2CO2CH3 radical, and R6═R8=methyl or —CH2CO2H or —CH2CO2CH3 radical;
3□°) n=2 and simultaneously R6═R8=methyl, and R5═OMe or hydroxyl;
- 4□°) n=2 and simultaneously R6═R8=methyl, and R5 is —CH2CO2H or —CH2CO2CH3 radical; or methyl;
5°) n=3 and simultaneously R6 is hydroxyl, and R8 is methyl, and R5 is —CH2CO2CH3 radical;
6°) n=3 and simultaneously R6═R8=methyl and R5═OH or methyl or ethyl.- the tertiary oxacycloalcane carboxylic acid, included its salts and each one of its pure enantiomeric forms or in racemic mixture or in variable composition, corresponding to the following formula:
where n is included between 0 and 8, R5, R6 and R8 are as defined above, but are not hydrogen simultaneously.
except for compounds corresponding to the
where n is included between 0 and 8, Z, R5, R6 and R8 are as defined above, R5 is not hydrogen, and R12 is not a CTX radical defined above;
except for compounds where Z is oxygen atom and, corresponding to the
where n is included between 0 and 8, R6 and R8 are as defined above.
except for compounds corresponding to the
where n is included between 0 and 8, R6 and R8 are as defined above, R12 is an hydrocarbon radical different from CTX as defined above.
except for compounds corresponding to the
where n and R5 are as defined above,
except for compounds corresponding to the
to remove the undesired related impurity named 2′-épi-homoharringtonine 2′-epi-homoharringtonine resulting:
a) either from a semi-synthetic process with introduction of in the course of which a synthetic homoharringtonic acid of inadequate enantiomeric purity was introduced, the generated impurity showing having the absolute configuration corresponding to the following formula:
b) or from a synthetic or semi-synthetic process, or from the biosynthetic process in the plant, where in the course of which a cephalotaxine with of inadequate enantiomeric purity was introduced, or in the form of artefact an artifact by partial racemization of the cephalotaxine moiety, the generated impurity showing having strictly identical chromatographic properties with in a non-chiral system, with and having an absolute configuration opposite to the one above (enantiomer) and corresponding to the following formula:
Homoharringtonine and harringtonine are present in Cephalotaxus extract as a complex mixture of several dozens of alkaloids (see scheme 1). For example, HA and HHT were firstly used as a mixture for therapy of cancer and leukemia in China. In the U.S., the level of compliance of drug substances to a given quality required by the FDA is increasing during the process of development of an investigational drug (i.e. from early phase I clinical trial to phase III). In addition, FDA requires that the profile of impurities in term of related compounds shall be reproducible from batch to batch during the marketing phase, that is very difficult when the drug substance is prepared from natural source. An homoharringtonine prepared from natural source was developed by the U.S., the National Cancer Institute and used in its early clinical trials. In despite of a final purification which use a crystallization in ethyl acetate, the final drug substance contains three major impurities including two natural congeners and ethyl analog of HHT which is an artifact of purification resulting from transesterification with the solvent of purification. [He et al., Journ. of Pharm. Biomed. Analysis, 22, pp 541-534 (2000)] The following table, reproduced from reference [He et al., 2000] exemplified the quality of this HHT, which is fact the best quality yielded in using the method of purification the state of art.
| TABLE I |
| Quality Of Various National Cancer Institute (NCI) Clinical Batches Of |
| Homoharringtonine Described In Literature [1] Compared to Semi-Synthetic Batches |
| % Related | % Related | % Related | % Total of | % HPLC | |
| Batch Identification | Comp. A | Comp. b | Comp. c | Rel. Comp. | purity |
| NCI Batch #800528 | 0.1 | 1.3 | 0 | 1.4 | 98.6 |
| NCI Batch #871203 | 0 | 1.0 | 0 | 1.0 | 99.0 |
| NCI Batch #921115 | 0.3 | 3.0 | 0.8 | 4.1 | 95.9 |
| NCI Batch #960625 | 0 | 1.8 | 0.3 | 2.1 | 97.9 |
| NCI Batch #800722 | 0.1 | 0.9 | 0.2 | 1.2 | 98.8 |
| NCI Batch #KS-22-130-2 | 0 | 1.5 | 0.9 | 2.4 | 97.6 |
| Average of National Cancer | 0.1 | 1.6 | 0.4 | 2.0 | 98.0 |
| Institute batches | |||||
| Average of Oncopharm's batches | 0.00 | 0.00 | 0.00 | <0.05 | >99.95 |
| Rate Impurities NCI batches/Impurities | >40 | ||||
| Oncopharm's Batches | |||||
| [1] Stability indicating LC assay of and impurity indentification in homoharringtonine samples, He et al., Journ. of Pharm. Biomed. Analysis, 22, pp 541-534 (2000). | |||||
- (i) at least one chromatographic purification, preferably in reverse phase in aqueous mobile phase such as a lower alkanol or tetrahydrofurane or acetonitrile, purified water, and an acidic buffer, preferably based on phosphoric acid and is salt. Stationary phase may be any standard chemically bound phase preferably an alkylsilane or an alkylnitrile, bounded on an inert core, preferably silicagel;
- (ii) at least one crystallization in water or aqueous solvent containing an organic solvent, preferably a lower C1-4 alkanol,
-
- [α]D 20: −23 (c=0.38; CHCl3).
-
- [α]D 20: +(c=0.36; CHCl3)
-
- [α]D 20: +8 (c=0.19; CHCl3)
homoharringtonine or cephalotaxine, 4-methyl (2R)-2-hydroxy-2-(4-hydroxy-4-methylpentyl) butanedioate (ester)
| TABLE II |
| Impurity Content Decrease After Application Of This Process |
| Before this | After this | ||
| Peak | Related Compound (impurities) | process | process |
| a | Harringtonine (HA) | 93.49 | 99.97 |
| b | HA isomer | 1.76 | 0 |
| c | HA isomer | 1.67 | 0 |
| d | Homoharringtonine | 3.01 | 0 |
| |
4′-dimethyl-HA | 0.01 | 0.03 |
| f | Drupacine analog | 0.05 | 0 |
| Sum of Related Compounds | 6.49 | 0.03 |
| Rate 6.49/0.03 | 216 | |
| DESCRIPTION AND DEFINITION OF HARRINGTONINES |
| EXAMPLES OF HARRINGTONIC ACIDS (R = H), 3 | |
| CEPHALOTAXANES | EXAMPLES OF HARRINGTONINES (R = CTX), 4 |
|
|
|
| CEPHALOTAXINES | R′ | R7 | n | R |
| EXAMPLES OF HARRINGTONIC ACIDS | |
| (not isolated in the natural form as such) |
|
|
H H H H H | (CH3)2C(OH)— (CH3)2C(OH)— (CH3)2C(OH)— (CH3)2CH— —C (CH3)2—O— Ph— | 2 3 1 2 3 1 | H H H H H H | harringtonic acid homoharringtonic acid norharringtonic acid desoxyharringtonic acid anhydrohomoharringtonic acid neoharringtonic acid |
| Examples of cephalotaxines: | EXAMPLES OF HARRINGTONINES (natural) |
| cephalotaxine = CTXOH | H | (CH3)2C(OH)— | 2 | CTX | Harringtonine: HT |
| 2a | H | (CH3)2C(OH)— | 3 | CTX | Homoharringtonine: HHT |
| R1 = OH, R2 = R4 = H, R3 = OMe; | H | (CH3)2C(OH)— | 1 | CTX | Norharringtonine |
| acetylcephalotaxine, 2b | H | (CH3)2CH— | 2 | CTX | Desoxyharringtonine |
| R1 = Ac, R2 = R4 = H, R3 = OMe; | H | (CH3)2CH— | 1 | CTX | Nordesoxyharringtonine |
| cephalotaxinone, 2c | H | (CH3)2CH— | 3 | CTX | Homodesoxyharringtonine |
| R1, R2 = O, R3 = OMe, R4 = H; | H | (CH3)2CH— | 4 | CTX | Bishomodesoxyharringtonine |
| demethylcephalotaxine, 2d | —C(CH3)2—O— | 2 | CTX | Anhydroharringtonine | |
| R1 = OH, R2 = R4 = H, R3 = OH; | H | Ph— | 1 | CTX | Neoharringtonine |
| demethylcephalotaxinone, 2e | H | Ph— | 2 | CTX | Homoneoharringtonine |
| R1, R2 = O, R3 = OH, R4 = H; | |||||
| 11-hydroxycephalotaxine, | |||||
| 2f = 2b + R4 = OH; | |||||
| ESSENCE OF PRIOR ART |
|
|
| i) Impossible on account of the steric hindrance [K. L. Mikolajczak et coll., Tetrahedron, p. 1995, (1972]; |
| ii) HCN, H+; |
| iii) MeOH, H+; |
| i′) Silver salt of 3c [K. L. Mikolajczak et coll. J. Med. Chem., p. 63, (1975)]; |
| iv) 2a/pyridine-dichloromethane; |
| v) according to [T. R. Kelly et coll., Tetrahedron Lett., 3501 (1973)]; |
| ORIGIN of the ANHYDROHARRINGTONINES |
|
|
| [D. Z. Wang et coll., Yaoxue Xuebao, p. 173, (1992)] |
| [D. Z. Wang et coll., Yaoxue Xuebao, p. 178, (1992)] |
Claims (35)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US13/661,677 USRE45128E1 (en) | 1998-03-20 | 2012-10-26 | Cephalotaxane derivatives and their processes of preparation and purification |
Applications Claiming Priority (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| FR9803492 | 1998-03-20 | ||
| FR9803492A FR2776292B1 (en) | 1998-03-20 | 1998-03-20 | CEPHALOTAXANES SUPPORTING THE SIDE CHAIN AND THEIR SYNTHESIS PROCESS |
| US09/270,006 US6831180B1 (en) | 1998-03-20 | 1999-03-16 | Cephalotaxane derivatives and process for their preparation |
| US10/877,067 US7169774B2 (en) | 1998-03-20 | 2004-06-25 | Cephalotaxane derivatives and their processes of preparation and purification |
| US11/440,648 US7842687B2 (en) | 1998-03-20 | 2006-05-25 | Cephalotaxane derivatives and their processes of preparation and purification |
| US13/661,677 USRE45128E1 (en) | 1998-03-20 | 2012-10-26 | Cephalotaxane derivatives and their processes of preparation and purification |
Related Parent Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US11/440,648 Reissue US7842687B2 (en) | 1998-03-20 | 2006-05-25 | Cephalotaxane derivatives and their processes of preparation and purification |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| USRE45128E1 true USRE45128E1 (en) | 2014-09-09 |
Family
ID=9524328
Family Applications (5)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US09/270,006 Expired - Lifetime US6831180B1 (en) | 1998-03-20 | 1999-03-16 | Cephalotaxane derivatives and process for their preparation |
| US09/817,176 Expired - Lifetime US6613900B2 (en) | 1998-03-20 | 2001-03-27 | Cephalotaxane derivatives and process for their preparation |
| US10/877,067 Expired - Lifetime US7169774B2 (en) | 1998-03-20 | 2004-06-25 | Cephalotaxane derivatives and their processes of preparation and purification |
| US11/440,648 Ceased US7842687B2 (en) | 1998-03-20 | 2006-05-25 | Cephalotaxane derivatives and their processes of preparation and purification |
| US13/661,677 Expired - Fee Related USRE45128E1 (en) | 1998-03-20 | 2012-10-26 | Cephalotaxane derivatives and their processes of preparation and purification |
Family Applications Before (4)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US09/270,006 Expired - Lifetime US6831180B1 (en) | 1998-03-20 | 1999-03-16 | Cephalotaxane derivatives and process for their preparation |
| US09/817,176 Expired - Lifetime US6613900B2 (en) | 1998-03-20 | 2001-03-27 | Cephalotaxane derivatives and process for their preparation |
| US10/877,067 Expired - Lifetime US7169774B2 (en) | 1998-03-20 | 2004-06-25 | Cephalotaxane derivatives and their processes of preparation and purification |
| US11/440,648 Ceased US7842687B2 (en) | 1998-03-20 | 2006-05-25 | Cephalotaxane derivatives and their processes of preparation and purification |
Country Status (19)
| Country | Link |
|---|---|
| US (5) | US6831180B1 (en) |
| EP (1) | EP1064285B1 (en) |
| JP (3) | JP4942871B2 (en) |
| KR (1) | KR20010042085A (en) |
| CN (1) | CN1220692C (en) |
| AT (1) | ATE289312T1 (en) |
| AU (1) | AU3270699A (en) |
| CA (1) | CA2324895A1 (en) |
| DE (1) | DE69923768T2 (en) |
| DK (1) | DK1064285T3 (en) |
| ES (1) | ES2237144T3 (en) |
| FR (1) | FR2776292B1 (en) |
| HU (1) | HUP0101375A3 (en) |
| IL (1) | IL138563A0 (en) |
| PL (1) | PL343032A1 (en) |
| PT (1) | PT1064285E (en) |
| TR (1) | TR200003613T2 (en) |
| WO (1) | WO1999048894A1 (en) |
| ZA (1) | ZA200005018B (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10597401B2 (en) | 2015-05-08 | 2020-03-24 | Albany Molecular Research, Inc. | Methods and intermediates for the preparation of omacetaxine and cephalotaxine derivatives thereof |
Families Citing this family (26)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2002074776A2 (en) * | 2001-03-21 | 2002-09-26 | Oncopharm Corporation | Highly purified and crystalline form of harringtonine |
| FR2776292B1 (en) | 1998-03-20 | 2004-09-10 | Oncopharm | CEPHALOTAXANES SUPPORTING THE SIDE CHAIN AND THEIR SYNTHESIS PROCESS |
| AU7940500A (en) * | 2000-10-17 | 2002-04-29 | Oncopharm Corp | New cephalotaxanes, their method of preparation and their use in treatment of cancers, leukemias, parasites including thus resistant to usual chemotherapeutic agents and as reversal agents |
| US7285546B2 (en) | 2000-10-17 | 2007-10-23 | Stragen Pharma S.A. | Cephalotaxanes, their method of preparation and their use in treatment of cancers, leukemias, parasites including those resistant to usual chemotherapeutic agents and as reversal agents |
| US20040082565A1 (en) * | 2002-07-17 | 2004-04-29 | Chemgenex Therapeutics, Inc. | Formulations and methods of administration of cephalotaxines including homoharringtonine |
| CN100396286C (en) * | 2002-12-30 | 2008-06-25 | 北京大学第一医院 | Application of homoharringtonine and harringtonine in preparation of drugs for inhibiting angiogenesis |
| ITMI20051352A1 (en) | 2005-07-15 | 2007-01-16 | Erregierre Spa | PROCESS OF PREPARATION OF ACID 2-METOXYICARBONYLMETHYL-6,6-DIMETHYL-2-TETRAIDROPIRAN CARBOSSYLIC |
| JP5550546B2 (en) * | 2007-04-13 | 2014-07-16 | ケムジェネックス・ファーマシューティカルズ・インコーポレイテッド | Oral cephalotaxin dosage form |
| EP2260041A4 (en) | 2008-03-03 | 2012-04-25 | Sloan Kettering Inst Cancer | CEPHALOTAXUS ESTERS, SYNTHESIS METHODS AND USES THEREOF |
| WO2010103405A2 (en) * | 2009-03-11 | 2010-09-16 | Jean-Pierre Robin | Process for preparing cephalotaxine esters |
| US20100240887A1 (en) * | 2009-03-23 | 2010-09-23 | Yaguang Liu | New methods of producing HHT |
| CN103635476B (en) * | 2011-08-18 | 2016-03-09 | 杭州本生药业有限公司 | The acylated derivatives of homoharringtonine, and its preparation method and application |
| JP6068472B2 (en) | 2011-08-18 | 2017-01-25 | ハンジョウ ベンシェン ファーマシューティカル シーオー., エルティーディー.Hangzhou Bensheng Pharmaceutical Co., Ltd. | Aminated homoharringtonine derivatives, their preparation and use |
| CN103687859B (en) * | 2011-08-18 | 2016-08-17 | 杭州本生药业有限公司 | The amination derivant of homoharringtonine, and its preparation method and application |
| WO2013023622A1 (en) * | 2011-08-18 | 2013-02-21 | 杭州本生药业有限公司 | Acylated derivative of homoharringtonine, preparation method therefor, and application thereof |
| CN102675327B (en) * | 2012-03-01 | 2014-12-10 | 南开大学 | Harringtonine alkaloid and preparation method and application thereof |
| CN102633806A (en) * | 2012-04-05 | 2012-08-15 | 贵州博丰生物科技产业开发有限公司 | Extraction and separation method of harringtonine |
| WO2015101628A1 (en) * | 2013-12-31 | 2015-07-09 | Jean-Pierre Robin | Water soluble crystallin salts of certain harringtonines unambiguously protonated on their alkaloid nitrogen and their use as chemotherapeutic drugs |
| WO2015128463A1 (en) * | 2014-02-28 | 2015-09-03 | Jean-Pierre Robin | Water soluble salts of harringtonine and their pharmaceutical applications |
| WO2015162217A2 (en) * | 2014-04-23 | 2015-10-29 | Jean-Pierre Robin | Crystalline salts of cephalotaxine and their use for its purification including enantiomers |
| CN106866690B (en) * | 2015-12-10 | 2019-10-11 | 南开大学 | harringtonine alkaloids, their preparation method and use |
| EP3275875A1 (en) | 2016-07-28 | 2018-01-31 | INDENA S.p.A. | Dioxolanone intermediate useful in the synthesis of homoharringtonine |
| CN110577506B (en) * | 2018-06-07 | 2023-04-07 | 中国科学院上海有机化学研究所 | Method for synthesizing ester derivative of (-cephalotaxine) and intermediate thereof |
| EP3763715A1 (en) | 2019-07-11 | 2021-01-13 | Robin, Jean-Pierre | Harringtonines salts, in particular retinoates, their process of preparation and their uses in the treatment of leukemias, cancers, autoimmune, skin, alzheimer's and inflammatory bowel diseases and viral infections, combined with myelopoiesis stimulating agents |
| CN114544849B (en) * | 2022-02-22 | 2024-07-19 | 苏州正济医药研究有限公司 | Method for detecting 1H-1,2, 4-triazole-1-formamidine monohydrochloride and impurities |
| CN117099793B (en) * | 2023-07-26 | 2026-04-10 | 广西农业职业技术大学 | Application of plant-derived active substance homoharringtonine in weed control |
Citations (39)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2889339A (en) | 1956-07-02 | 1959-06-02 | Trubek Lab | Dialkyl substituted 2, 3-epoxyacid esters |
| US2889340A (en) | 1956-07-02 | 1959-06-02 | Trubek Lab | Unsaturated dialkyl substituted 2, 3-epoxyacid esters |
| US3246038A (en) | 1961-05-05 | 1966-04-12 | Bayer Ag | Process for the production of 4-[2', 6', 6'-trimethylcyclohexen-(1')-yl]-2-methylbuten-(3)-al |
| US3725437A (en) | 1970-09-16 | 1973-04-03 | Sankyo Chem Ind Ltd | Process for the preparation of {60 -hydroxy-{62 -phenypropionic acid derivatives and alkalimetal-or ammonium salts thereof |
| US3793454A (en) | 1971-11-12 | 1974-02-19 | Us Agriculture | Harringtonine and isopharringtonine for treating l1210 or p388 leukemic tumors in mice |
| US3855245A (en) | 1971-06-16 | 1974-12-17 | Nisshin Flour Milling Co | Process for the manufacture of derivatives of beta-phenylpyruvic acid |
| US3870727A (en) | 1970-04-09 | 1975-03-11 | Us Agriculture | Production of harringtonine and isoharringtonine |
| US3959312A (en) | 1974-12-20 | 1976-05-25 | The United States Of America As Represented By The Secretary Of Agriculture | Synthesis of antitumor alkaloid deoxyharringtonine and its precursor 3'-0-(5-methyl-2-oxohexanoyl)-cephalotaxine |
| US4042617A (en) | 1974-07-27 | 1977-08-16 | Nisshin Flour Milling Co., Ltd. | Process for preparing a 2-methyl-(substituted aryl)-pyruvic acid compound |
| US4131747A (en) | 1975-11-19 | 1978-12-26 | Ono Pharmaceutical Co., Ltd. | Process for preparing α-substituted phenylalkanecarboxylic acid |
| US4132720A (en) | 1978-04-20 | 1979-01-02 | Mcneil Laboratories, Inc. | Alkenylglycidic acid derivatives |
| US4152214A (en) | 1977-10-07 | 1979-05-01 | The United States Of America As Represented By The Secretary Of Agriculture | Production of homodeoxyharringtonine and other cephalotaxine esters by tissue culture |
| US4152333A (en) | 1978-02-22 | 1979-05-01 | The United States Of America As Represented By The Secretary Of Agriculture | Synthetic cephalotaxine esters having antileukemic activity |
| US4154840A (en) | 1976-02-13 | 1979-05-15 | E. R. Squibb & Sons, Inc. | Antihypertensive pipecolic acid derivatives |
| US4178286A (en) | 1976-10-28 | 1979-12-11 | Research Corporation | β-Lactams, their production, intermediates thereto and derivatives thereof |
| US4203996A (en) | 1978-02-22 | 1980-05-20 | The United States Of America As Represented By The Secretary Of Agriculture | Synthetic cephalotaxine esters having antileukemic P388 activity |
| US4252728A (en) | 1978-04-17 | 1981-02-24 | Firmenich Sa | Norbornane and norbornene derivatives |
| JPS5832880A (en) | 1981-08-24 | 1983-02-25 | Isukura Sangyo Kk | Partial synthesis of harringtonine |
| US4409236A (en) | 1978-08-03 | 1983-10-11 | Boehringer Mannheim Gmbh | N-Substituted aziridine-2-carboxylic acid derivatives and immuno-stimulation composition and method |
| US4430339A (en) | 1980-08-29 | 1984-02-07 | Byk Gulden Lomberg Chemische Fabrik Gesellschaft Mit Beschrankter Haftung | Substituted oxiranecarboxylic acids, their preparation and their use as medicaments |
| US4619888A (en) | 1984-08-24 | 1986-10-28 | Fuji Photo Film Co., Ltd. | Heat developable light-sensitive material |
| USH271H (en) | 1984-08-06 | 1987-05-05 | The United States Of America As Represented By The Secretary Of The Army | Treatment of malaria with esters of cephalotaxine |
| US4675318A (en) | 1985-05-28 | 1987-06-23 | Yaguang Liu | Safe antileukemia drug, SAL |
| US4783454A (en) | 1985-05-28 | 1988-11-08 | Yaguang Liu | Process for producing harringtonine and homoharringtonine |
| US4808629A (en) | 1985-05-28 | 1989-02-28 | Yaguang Liu | Safe antileukemia drug, SAL |
| US4849524A (en) | 1986-11-21 | 1989-07-18 | Hoechst Aktiengesellschaft | Process of preparing proline derivatives |
| US4870208A (en) | 1988-06-03 | 1989-09-26 | Monsanto Company | Asymmetric hydrogenolysis of epoxides |
| US5081240A (en) | 1989-07-18 | 1992-01-14 | Sanofi | Process for the preparation of benzothiazepin-one derivatives |
| US5084582A (en) | 1988-07-01 | 1992-01-28 | Societe Nationale Elf Aquitaine | Oxidation of epoxy alcohols into carboxilic epoxy acids |
| WO1992001696A1 (en) | 1990-07-24 | 1992-02-06 | Beecham Group Plc | Cephalosporins and homologues, preparations and pharmaceutical compositions |
| US5256803A (en) | 1990-02-21 | 1993-10-26 | Rhone-Poulenc Rorer S.A. | Process for the preparation of (2R,3R)-cis)-β-phenylglycidic acid |
| US5334730A (en) | 1991-07-04 | 1994-08-02 | Consortium Fur Elektrochemische Industrie Gmbh | Process for the preparation of optically active carboxylic acids and amide intermediates |
| US5502221A (en) | 1991-06-21 | 1996-03-26 | Takasago International Corporation | Process for producing cyclohexylbutyric acid derivative |
| US5677470A (en) | 1994-06-28 | 1997-10-14 | Tanabe Seiyaku Co., Ltd. | Baccatin derivatives and processes for preparing the same |
| US5834467A (en) | 1992-12-22 | 1998-11-10 | Agouron Pharmaceuticals, Inc. | HIV protease inhibitors |
| FR2776292A1 (en) | 1998-03-20 | 1999-09-24 | Oncopharm | Novel cephalotaxane derivatives with a side chain |
| US6107291A (en) | 1997-12-19 | 2000-08-22 | Amgen Inc. | Azepine or larger medium ring derivatives and methods of use |
| FR2818970A1 (en) | 2000-12-29 | 2002-07-05 | Oncopharm | PROCESS FOR THE ENANTIOSELECTIVE PREPARATION OF PHENYLETHYLIMINO-CYCLOALCANES ALPHA SUBSTITUTE ADDUCTS AND 2-ACETOXYACRYLONITRILE, INTERMEDIARIES IN THE ENANTOSELECTIVE SYNTHESIS OF THE HARRINGTONIN SIDE CHAIN |
| US7683050B2 (en) | 2002-07-17 | 2010-03-23 | Chemgenex Pharmaceuticals, Inc. | Formulations and methods of administration of cephalotaxines, including homoharringtonine |
Family Cites Families (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3225069A (en) * | 1963-04-18 | 1965-12-21 | Atlas Chem Ind | Process of producing esters of hydroxymethyletrahydrofuroic acid |
| JPS5764695A (en) * | 1980-10-09 | 1982-04-19 | Kyowa Hakko Kogyo Co Ltd | Novel cephalotaxine derivative |
| JPS57102194A (en) * | 1980-12-17 | 1982-06-25 | Kyowa Hakko Kogyo Co Ltd | Preparation of cephalotaxine and ester thereof |
| US20020128258A1 (en) * | 2001-03-09 | 2002-09-12 | Jean-Pierre Robin | Therapeutical method involving subcutaneous administration of drugs containing cephalotaxine derivatives |
-
1998
- 1998-03-20 FR FR9803492A patent/FR2776292B1/en not_active Expired - Lifetime
-
1999
- 1999-03-16 US US09/270,006 patent/US6831180B1/en not_active Expired - Lifetime
- 1999-03-17 JP JP2000537877A patent/JP4942871B2/en not_active Expired - Fee Related
- 1999-03-17 IL IL13856399A patent/IL138563A0/en unknown
- 1999-03-17 DE DE69923768T patent/DE69923768T2/en not_active Expired - Lifetime
- 1999-03-17 DK DK99942587T patent/DK1064285T3/en active
- 1999-03-17 WO PCT/IB1999/000491 patent/WO1999048894A1/en not_active Ceased
- 1999-03-17 HU HU0101375A patent/HUP0101375A3/en unknown
- 1999-03-17 AT AT99942587T patent/ATE289312T1/en active
- 1999-03-17 PT PT99942587T patent/PT1064285E/en unknown
- 1999-03-17 KR KR1020007010440A patent/KR20010042085A/en not_active Withdrawn
- 1999-03-17 ES ES99942587T patent/ES2237144T3/en not_active Expired - Lifetime
- 1999-03-17 TR TR2000/03613T patent/TR200003613T2/en unknown
- 1999-03-17 PL PL99343032A patent/PL343032A1/en unknown
- 1999-03-17 CN CNB998060445A patent/CN1220692C/en not_active Expired - Lifetime
- 1999-03-17 AU AU32706/99A patent/AU3270699A/en not_active Abandoned
- 1999-03-17 CA CA002324895A patent/CA2324895A1/en not_active Abandoned
- 1999-03-17 EP EP99942587A patent/EP1064285B1/en not_active Expired - Lifetime
-
2000
- 2000-09-20 ZA ZA200005018A patent/ZA200005018B/en unknown
-
2001
- 2001-03-27 US US09/817,176 patent/US6613900B2/en not_active Expired - Lifetime
-
2004
- 2004-06-25 US US10/877,067 patent/US7169774B2/en not_active Expired - Lifetime
-
2006
- 2006-05-25 US US11/440,648 patent/US7842687B2/en not_active Ceased
-
2009
- 2009-03-16 JP JP2009063260A patent/JP5884005B2/en not_active Expired - Fee Related
-
2012
- 2012-10-26 US US13/661,677 patent/USRE45128E1/en not_active Expired - Fee Related
-
2015
- 2015-03-19 JP JP2015056022A patent/JP6087381B2/en not_active Expired - Lifetime
Patent Citations (42)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2889340A (en) | 1956-07-02 | 1959-06-02 | Trubek Lab | Unsaturated dialkyl substituted 2, 3-epoxyacid esters |
| US2889339A (en) | 1956-07-02 | 1959-06-02 | Trubek Lab | Dialkyl substituted 2, 3-epoxyacid esters |
| US3246038A (en) | 1961-05-05 | 1966-04-12 | Bayer Ag | Process for the production of 4-[2', 6', 6'-trimethylcyclohexen-(1')-yl]-2-methylbuten-(3)-al |
| US3870727A (en) | 1970-04-09 | 1975-03-11 | Us Agriculture | Production of harringtonine and isoharringtonine |
| US3725437A (en) | 1970-09-16 | 1973-04-03 | Sankyo Chem Ind Ltd | Process for the preparation of {60 -hydroxy-{62 -phenypropionic acid derivatives and alkalimetal-or ammonium salts thereof |
| US3855245A (en) | 1971-06-16 | 1974-12-17 | Nisshin Flour Milling Co | Process for the manufacture of derivatives of beta-phenylpyruvic acid |
| US3793454A (en) | 1971-11-12 | 1974-02-19 | Us Agriculture | Harringtonine and isopharringtonine for treating l1210 or p388 leukemic tumors in mice |
| US4042617A (en) | 1974-07-27 | 1977-08-16 | Nisshin Flour Milling Co., Ltd. | Process for preparing a 2-methyl-(substituted aryl)-pyruvic acid compound |
| US3959312A (en) | 1974-12-20 | 1976-05-25 | The United States Of America As Represented By The Secretary Of Agriculture | Synthesis of antitumor alkaloid deoxyharringtonine and its precursor 3'-0-(5-methyl-2-oxohexanoyl)-cephalotaxine |
| US4131747A (en) | 1975-11-19 | 1978-12-26 | Ono Pharmaceutical Co., Ltd. | Process for preparing α-substituted phenylalkanecarboxylic acid |
| US4154840A (en) | 1976-02-13 | 1979-05-15 | E. R. Squibb & Sons, Inc. | Antihypertensive pipecolic acid derivatives |
| US4178286A (en) | 1976-10-28 | 1979-12-11 | Research Corporation | β-Lactams, their production, intermediates thereto and derivatives thereof |
| US4152214A (en) | 1977-10-07 | 1979-05-01 | The United States Of America As Represented By The Secretary Of Agriculture | Production of homodeoxyharringtonine and other cephalotaxine esters by tissue culture |
| US4203996A (en) | 1978-02-22 | 1980-05-20 | The United States Of America As Represented By The Secretary Of Agriculture | Synthetic cephalotaxine esters having antileukemic P388 activity |
| US4152333A (en) | 1978-02-22 | 1979-05-01 | The United States Of America As Represented By The Secretary Of Agriculture | Synthetic cephalotaxine esters having antileukemic activity |
| US4252728A (en) | 1978-04-17 | 1981-02-24 | Firmenich Sa | Norbornane and norbornene derivatives |
| US4132720A (en) | 1978-04-20 | 1979-01-02 | Mcneil Laboratories, Inc. | Alkenylglycidic acid derivatives |
| US4409236A (en) | 1978-08-03 | 1983-10-11 | Boehringer Mannheim Gmbh | N-Substituted aziridine-2-carboxylic acid derivatives and immuno-stimulation composition and method |
| US4430339A (en) | 1980-08-29 | 1984-02-07 | Byk Gulden Lomberg Chemische Fabrik Gesellschaft Mit Beschrankter Haftung | Substituted oxiranecarboxylic acids, their preparation and their use as medicaments |
| JPS5832880A (en) | 1981-08-24 | 1983-02-25 | Isukura Sangyo Kk | Partial synthesis of harringtonine |
| USH271H (en) | 1984-08-06 | 1987-05-05 | The United States Of America As Represented By The Secretary Of The Army | Treatment of malaria with esters of cephalotaxine |
| US4619888A (en) | 1984-08-24 | 1986-10-28 | Fuji Photo Film Co., Ltd. | Heat developable light-sensitive material |
| US4675318A (en) | 1985-05-28 | 1987-06-23 | Yaguang Liu | Safe antileukemia drug, SAL |
| US4783454A (en) | 1985-05-28 | 1988-11-08 | Yaguang Liu | Process for producing harringtonine and homoharringtonine |
| US4808629A (en) | 1985-05-28 | 1989-02-28 | Yaguang Liu | Safe antileukemia drug, SAL |
| US4849524A (en) | 1986-11-21 | 1989-07-18 | Hoechst Aktiengesellschaft | Process of preparing proline derivatives |
| US4870208A (en) | 1988-06-03 | 1989-09-26 | Monsanto Company | Asymmetric hydrogenolysis of epoxides |
| US5084582A (en) | 1988-07-01 | 1992-01-28 | Societe Nationale Elf Aquitaine | Oxidation of epoxy alcohols into carboxilic epoxy acids |
| US5081240A (en) | 1989-07-18 | 1992-01-14 | Sanofi | Process for the preparation of benzothiazepin-one derivatives |
| US5256803A (en) | 1990-02-21 | 1993-10-26 | Rhone-Poulenc Rorer S.A. | Process for the preparation of (2R,3R)-cis)-β-phenylglycidic acid |
| WO1992001696A1 (en) | 1990-07-24 | 1992-02-06 | Beecham Group Plc | Cephalosporins and homologues, preparations and pharmaceutical compositions |
| US5502221A (en) | 1991-06-21 | 1996-03-26 | Takasago International Corporation | Process for producing cyclohexylbutyric acid derivative |
| US5334730A (en) | 1991-07-04 | 1994-08-02 | Consortium Fur Elektrochemische Industrie Gmbh | Process for the preparation of optically active carboxylic acids and amide intermediates |
| US5834467A (en) | 1992-12-22 | 1998-11-10 | Agouron Pharmaceuticals, Inc. | HIV protease inhibitors |
| US5677470A (en) | 1994-06-28 | 1997-10-14 | Tanabe Seiyaku Co., Ltd. | Baccatin derivatives and processes for preparing the same |
| US6107291A (en) | 1997-12-19 | 2000-08-22 | Amgen Inc. | Azepine or larger medium ring derivatives and methods of use |
| FR2776292A1 (en) | 1998-03-20 | 1999-09-24 | Oncopharm | Novel cephalotaxane derivatives with a side chain |
| CA2324895A1 (en) | 1998-03-20 | 1999-09-30 | Oncopharm Corporation | Novel cephalotaxane derivatives and process for their preparation |
| US6613900B2 (en) | 1998-03-20 | 2003-09-02 | Oncopharm Corporation | Cephalotaxane derivatives and process for their preparation |
| US6831180B1 (en) | 1998-03-20 | 2004-12-14 | Stragen Pharma Sa. | Cephalotaxane derivatives and process for their preparation |
| FR2818970A1 (en) | 2000-12-29 | 2002-07-05 | Oncopharm | PROCESS FOR THE ENANTIOSELECTIVE PREPARATION OF PHENYLETHYLIMINO-CYCLOALCANES ALPHA SUBSTITUTE ADDUCTS AND 2-ACETOXYACRYLONITRILE, INTERMEDIARIES IN THE ENANTOSELECTIVE SYNTHESIS OF THE HARRINGTONIN SIDE CHAIN |
| US7683050B2 (en) | 2002-07-17 | 2010-03-23 | Chemgenex Pharmaceuticals, Inc. | Formulations and methods of administration of cephalotaxines, including homoharringtonine |
Non-Patent Citations (70)
| Title |
|---|
| Abraham et al., Single Crystal X-Ray Structures of Chemotherapeutic Agents II, Tetrahedron Letters (1969), No. 46, pp. 4085-4086. Pregman Press, Great Britain. |
| Ancliff et al., Synthesis of the ester side chains of some potently antileukemic harringtonia alkaloids from chiral citrates, Chem. Commun. (2006), pp. 3243-3245. |
| Anonymous, Acta Bot. Sin.22:156 (1980) cited by Huang, L, et al., "Cephalotaxus Alkaloids", The Alkaloids vol. XXII, pp. 157 (1988). |
| Arora et al., Crystal and Molecular Structure of Cephalotaxine, J. Org. Chem. (1976), 43(3), pp. 551-554. |
| Auerbach et al., The Total Synthesis of Cephalotaxine, J. Amer. Chem. Soc. (1972), pp. 7172-7173. |
| Berhal et al., Stereoselective Synthesis of Enantiopure Analogues of (-)-Cephalotaxine, Tetrahedron: Asymmetry (2010), 21, pp. 325-332. |
| Berhal et al., Stereoselective Synthesis of Enantiopure Analogues of (−)-Cephalotaxine, Tetrahedron: Asymmetry (2010), 21, pp. 325-332. |
| Cecil R. Smith, Jr. et al., "Harringtonine and Related Cephalotaxine Esters", Anticancer Agents based on Natural Product Models, Chapter II, vol. 16, pp. 391-416, 1980, Academic Press, New York, New York, USA. |
| Ciurdaru et al., PL 103334, CA 121:255399, 1994. |
| D.Z. Wang et al, Yaoxue Xuebao, vol. 27, p. 173 (1992). |
| D.Z. Wang et al, Yaoxue Xuebao, vol. 27, p. 178 (1992). |
| Eckelbarger et al., Synthesis of Antiproliferative Cephalotaxus Esters and Their Evaluation against Several Human Hematopoietic and Solid Tumor Cell Lines: Uncovering Differential Susceptibilities to Multidrug Resistance, Chemistry Eur. J. (2008), 14(14), pp. 4293-4306. |
| Eckelbarger, et al., Strain-Release Rearrangement of N-Vinyl-2-Arylaziridines. Total Synthesis of the Anti-Leukemia Alkaloid (-)-Deoxyharringtonine, J. Am. Chem. Soc. (2006), 128, pp. 10370-10371. |
| Eckelbarger, et al., Strain-Release Rearrangement of N-Vinyl-2-Arylaziridines. Total Synthesis of the Anti-Leukemia Alkaloid (−)-Deoxyharringtonine, J. Am. Chem. Soc. (2006), 128, pp. 10370-10371. |
| French Preliminary Search Report from FR 9803492, issued Dec. 14, 1998. |
| Guanghong et al., The Crystal Structures of Harringtonine and Homoharringtonine, Acta Acad. Med. Sinica (1988), 10(4), pp. 283-287 (with Engl. Translation). |
| H.A. Bates, "Decarbonylation of tetrahydrofuran-2-carboxylic acids and tetrahydropyran-2-carboxylic acids in concentrated sulfuric acid: formation of oxonium ions", Journal of the American Chemical Society, vol. 104, No. 9, 1982, pp. 2490-2493, XP002087583. |
| He et al., Stability-indicating LC assay of and impurity identification in homoharringtonine samples, J. Pharma. and Biomedical Analysis (2000), vol. 22, pp . 541-554. |
| Hiranuma et al., Synthesis of homoharringtonine and Its Derivative by Partial Esterification of Cephalotaxine, Tetrahedron Lett. (1982), 23(34), pp. 3431-3434. |
| Hiranuma et al.; Studies in Cephalotaxus Alkaloids. Stereospecific Total Synthesis of Homoharringtonine, J. Org. Chem. (1983), 48, pp. 5321-5326. |
| Huang et al., Chapter 3: Cephalotaxus Alkaloids, The Alkaloids (1988), vol. XXIII, pp. 157-226. |
| Hudlicky et al., Chapter 5: Synthesis of Cephalotaxine Alkaloids, pp. 639-691, 1998. |
| International Search Report from PCT/IB02/02054 issued Nov. 18, 2002. |
| Isono et al., Total Synthesis (-)-Cephalotaxine, J. Org. Chem. (1995), 60, pp. 115-119. |
| Isono et al., Total Synthesis (−)-Cephalotaxine, J. Org. Chem. (1995), 60, pp. 115-119. |
| Johnson et al., "Dianions Derived from alpha-Halo Acids. The Darzens Condensation Revisited", Journal of Organic Chemsitry, 47:1205-1212 (1982). |
| Johnson et al., "Dianions Derived from α-Halo Acids. The Darzens Condensation Revisited", Journal of Organic Chemsitry, 47:1205-1212 (1982). |
| K.L. Mikolajczack et al, "Preparation and Antitumor Activity of a Rearranged Ester of Cephalotaxine", J. Med. Chem., vol. 18, No. 1, pp. 63-66, (1975). |
| Kantarjian et al., Homoharringtonine History, Current Research, and Future Directions, American Cancer Society (2001), 92(6), pp. 1591-1605. |
| Keng et al., Synthesis of Homoharringtonine and Separation of its Stereoisomers, Asta Chimica Sinica (1985), 43, pp. 161-167. |
| Kexue Tongbao, The Partial Synthesis of Harringtonine, Study the Theory of the Dictatorship of the Proletariat, vol. 20, No. 9, 1975 (with English Translation). |
| Kim et al., Characterization of solvent induced crystalline and amorphous homoharringtonine, Korean J. Chem. Eng. (2009), 26(4), pp. 1090-1093. |
| Kulesza et al., PL 85803, CA 90: 121390 (1979). |
| Lemmens et al., Journal of Organic Chemistry, "Synthesis of alpha, beta-Epoxyacyl Azides and Their Rearrangement to Epoxy Isocyanates and 3- and 4-Oxazolin-2-ones", 49:2231-2235 (1984). |
| Lemmens et al., Journal of Organic Chemistry, "Synthesis of α, β-Epoxyacyl Azides and Their Rearrangement to Epoxy Isocyanates and 3- and 4-Oxazolin-2-ones", 49:2231-2235 (1984). |
| Leroux, "Obtention de nitriles tetrahydrfuranniques et tetrahydropyranniques par action du CuCN surlesc'etones gamma et delta halogenees", Bulletin De La Societe Chimique De France, No. 1, 1968, pp. 344-351, XP002087586, Paris, France, Translator's Reports/Comments, 2011, (with English Translation). |
| Leroux, Preparation of Tetrahydrofuran and Tetrahydropyran Nitriles by the Action of CuCN on gamma-and 8-Halogenated Ketones, Laboratoire de Synthese Organicque, pp. 1-22, 1967. |
| Leroux, Preparation of Tetrahydrofuran and Tetrahydropyran Nitriles by the Action of CuCN on γ-and 8-Halogenated Ketones, Laboratoire de Synthese Organicque, pp. 1-22, 1967. |
| Li et al.; Concise Total Synthesis of (±)-Cephalotaxine via a Transannulation Strategy: Development of a Facile Reductive oxy-Nazarov Cyclization, Organic Letters American Chemical Society, 2011. |
| Liang et al., Partial Synthesis of Harringtonine, Scientia Sinica (1979), 22(11), pp. 1335-1345. |
| Liang Huang et al., "Cephalotaxus Alkaloids", The Alkaloids, vol. 51, Chapter 3, pp. 157-227, 1999, Academic Press, Burlington, MA, USA. |
| M. Nagai et al, "The stereochemistry of protopanaxadiol. The absolute configuration of C(20) of dammarenediol-I- and -II", Tetrahedron Letters, No. 40, 1966, pp. 4797-4801, XP002087585, Oxford, GB. |
| M.A. Miah et al., "Cephalotaxine Alkaloids", The Alkaloids, vol. 51, Chapter 2, pp. 199-264, 1998, Academic Press, Burlington, MA, USA. |
| Miah et al., Chapter 2: Cephalotaxus Alkaloids, The Alkaloids, vol. 51, pp. 199-269, 1998. |
| Mikolajczak et al., Deoxyharringtonine, A New Antitumor Alkaloid from Cephalotaxus: Structure and Synthetic Studies, Tetrahedron (1971), vol. 28, pp. 1995-2001. |
| Nagasaka et al.; 1-Azaspiro[4.4]nonane-2,6-dione and the Separation and Absolute Configurations of its Enantiomers, Tetrahedron: Asymmetry (1997), 8(2), pp. 191-194. |
| Newman, M.S., et al., "The Darzens Glycidic Ester Condensation", Organic Reactions, vol. V, Chapter 10, pp. 413-440 (1949), John Wiley & Sons, New York, NY, USA. |
| O'Dwyer et al., Homoharringtonine-Perspectives on an Active New Natural Product, Journal of Clinical Oncology (1986), 4(10), pp. 1563-1568. |
| P.G.M. Wuts et al, "An electrochemical ketal synthesis from 2-carboxy-2-allyl-tetrahydropyrans", Tetrahedron Letters, vol. 23, No. 39, 1982, pp. 3987-3990, XP002087584, Oxford, GB. |
| Paudler et al., The Alkaloids of Cephalotaxus Drupacea and Cephalotaxus Fortunei, Department of Chemistry, Ohio University (1963), vol. 28, pp. 2194-2197. |
| Planas et al., Stereoselective Synthesis of (-)-Cephalotaxine and C-7 Alkylated Analogues, J. Org. Chem. (2004), vol. 69, pp. 3087-3092. |
| Planas et al., Stereoselective Synthesis of (−)-Cephalotaxine and C-7 Alkylated Analogues, J. Org. Chem. (2004), vol. 69, pp. 3087-3092. |
| Powell et al., Antitumor Alkaloids from Cephalotaxus Harringtonia: Structure and Activity (1972), 61(8), pp. 1227-1230. |
| Powell, Structures of Homoerythrina Alkaloids from Cephalotaxus Harringtonia, Phytochemistry (1972), vol. 11, pp. 1467-1972. |
| Reich et al., Biorg. Med. Chem. Lett. "Derivatives of D-Penicillamine as Potential Antiarthritic Agents", 4(0):1167-70 1994. |
| Robin et al., The First Semi-Synthesis of Enantiopure Homoharringtonine via Anhydrohomoharringtonine from a Preformed Chiral Acyl Moiety, Tetrahedron Lett. (1999), 40, pp. 2931-2934. |
| Shu-Wen et al., A Study on the Deoxyharringtonine and its Analogues, Acta Chimica Sinica, Shanghai Institute of Mayeria Medica (1975), pp. 75-78 (with English Translation). |
| Smith et al., The Genus Cephalotaxus: Source of Homoharringtonine and Related Anticancer Alkaloids, Cancer Treatment Reports 1976), 60(8), pp. 1157-1170. |
| Studies on Alkaloids of Cephlotaxus Sinensis (Rehd. et Will.) Li, Alkaloids Group, Laboratory of Plant Chemistry, Institute of Botany, Academia Botanica Sinica (1980), 22(2). |
| Suffness et al., Chapter 1: Antitumor Alkaloids, The Alkaloids (1985),vol. XXV, pp. 57-69 and 295-298. |
| Takeda et al. Antitumor Activities of Harringtonine and Homoharringtonine, Cephalotaxus Alkaloids Which Are Active Principles From Plant by Intraperitoneal and Oral Administration, J. Pharm. Dyn. (1982), 5, pp. 841-847. |
| Taniguchi et al., Short Synthesis of (--)-Cephalotaxine Using a Radical Cascade, Organic Letters (2008), 10(18), pp. 4129-4131. |
| Thomas Hudlicky et al., "Synthesis of Cephalotaxine Alkaloids", The Alkaloids, vol. 51, Chapter 5, pp. 639-691, 1998, Academic Press, Burlington, MA, USA. |
| U.S. Appl. No. 60/278,673, filed Mar. 21, 2001, Robin et al. |
| Wang et al., Studies on Alkaloids of Cephlotaxus, VII Structures and Semi-Synthesis of Two Anticancer Cephalotaxine Esters, Acta Pharma. Sinica (1992), 27(3), pp. 173-177 (with Engl. Translation). |
| Wang et al., Studies on the Alkaloids of Cephalotaxus IX. Semi-Synthesis of Cephalotaxine Esters and Their Anti-Leukemic Activity, Acta Pharma. Sinica (1992), 23(3), pp. 178-184 (with Engl. Translation). |
| Wenkui et al., Synthesis of Deoxyharringtonine and Separation of its Steromers, Scientia Sinica (1980), 23(7), pp. 835-846. |
| Wen-Kui Huang et al., "Synthesis of Deoxyharringtonine and Separation of its Stereomers", Sci. Sin., vol. 23, No. 7, pp. 835-846,1980, Chinese Academy of Sciences, Beijing, China. |
| Wilmot, Complex Botanical Natural Products: Synthesis of Cephalotaxus Esters and of the C19-Diterpenoid Skeleton of Aconitunm and Delphinium Alkaloids, 2010. |
| Yongji et al., Experimental Studies on the Toxicity of Harringtonine and homoharringtonine, Chinese Med. Journal (1979), 92(3), pp. 175-180. |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10597401B2 (en) | 2015-05-08 | 2020-03-24 | Albany Molecular Research, Inc. | Methods and intermediates for the preparation of omacetaxine and cephalotaxine derivatives thereof |
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| USRE45128E1 (en) | Cephalotaxane derivatives and their processes of preparation and purification | |
| KR100255525B1 (en) | Baccatine iii derivatives | |
| JPH0556357B2 (en) | ||
| US9096550B2 (en) | Bryostatin analogues and methods of making and using thereof | |
| JP2006504753A (en) | Dihydroartemisinin and dihydroartemicitene dimers as anticancer and antiinfectives | |
| JPH02504394A (en) | Stereospecific method for producing furo[3,4-c]pyridine enantiomers, compounds thus obtained and pharmaceutical compositions thereof | |
| US20140343039A1 (en) | Process for preparing cephalotaxine esters | |
| US8796474B1 (en) | Macrolide compounds and methods and intermediates useful for their preparation | |
| El-Feraly et al. | A new method for the preparation of arteether and its C-9 epimer | |
| JP2000511897A (en) | Novel water-soluble analog of 20 (S) camptothecin | |
| CA2441428A1 (en) | Highly purified and crystalline form of harringtonine | |
| US20230026724A1 (en) | Substituted nitrogen heterocyclic compound and anesthetic effect thereof | |
| Riehs et al. | EPC synthesis of (+)-heptelidic acid | |
| CN115043879A (en) | Dihydroartemisinin derivative and preparation method thereof | |
| US7285546B2 (en) | Cephalotaxanes, their method of preparation and their use in treatment of cancers, leukemias, parasites including those resistant to usual chemotherapeutic agents and as reversal agents | |
| Torres-Valencia et al. | Preparation of (2R, 3S)-(−)-and (2S, 3R)-(+)-2, 3-epoxy-2-methylbutanoic acids and some of their esters | |
| JPS5913783A (en) | diterpenoids | |
| Shigemori et al. | Oxidation products of strictosamide by iodosobenzene diacetate | |
| CZ20003451A3 (en) | New Cephalotaxane Derivatives and Process for their Preparation | |
| Baars | Towards a Total Synthesis of Peloruside A and the Preparation of Selected Inositol Derivatives | |
| WO2022143617A1 (en) | Nagilactone compound and use thereof in preparation of anti-tumor drug | |
| JP2004161728A (en) | Nachiterpiocin derivative and anticancer agent containing this as an active ingredient | |
| JP2004315373A (en) | Compound having antitumor activity and angiogenesis inhibitory activity, and method for producing the same |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| FEPP | Fee payment procedure |
Free format text: PAYOR NUMBER ASSIGNED (ORIGINAL EVENT CODE: ASPN); ENTITY STATUS OF PATENT OWNER: LARGE ENTITY |
|
| CC | Certificate of correction | ||
| AS | Assignment |
Owner name: TEVA PHARMACEUTICALS INTERNATIONAL GMBH, SWITZERLA Free format text: COMMERCIAL REGISTER CHANGES;ASSIGNOR:IVAX PHARMACEUTICALS GMBH;REEL/FRAME:043889/0617 Effective date: 20150223 |
|
| AS | Assignment |
Owner name: TEVA PHARMACEUTICALS INTERNATIONAL GMBH, SWITZERLA Free format text: CORRECTIVE ASSIGNMENT TO CORRECT THE ASSIGNOR NAME PREVIOUSLY RECORDED AT REEL: 043889 FRAME: 0617. ASSIGNOR(S) HEREBY CONFIRMS THE CHANGE OF NANE;ASSIGNOR:IVAX INTERNATIONAL GMBH;REEL/FRAME:044710/0595 Effective date: 20150223 |
|
| MAFP | Maintenance fee payment |
Free format text: PAYMENT OF MAINTENANCE FEE, 8TH YEAR, LARGE ENTITY (ORIGINAL EVENT CODE: M1552) Year of fee payment: 8 |
|
| FEPP | Fee payment procedure |
Free format text: MAINTENANCE FEE REMINDER MAILED (ORIGINAL EVENT CODE: REM.); ENTITY STATUS OF PATENT OWNER: LARGE ENTITY |
|
| LAPS | Lapse for failure to pay maintenance fees |
Free format text: PATENT EXPIRED FOR FAILURE TO PAY MAINTENANCE FEES (ORIGINAL EVENT CODE: EXP.); ENTITY STATUS OF PATENT OWNER: LARGE ENTITY |