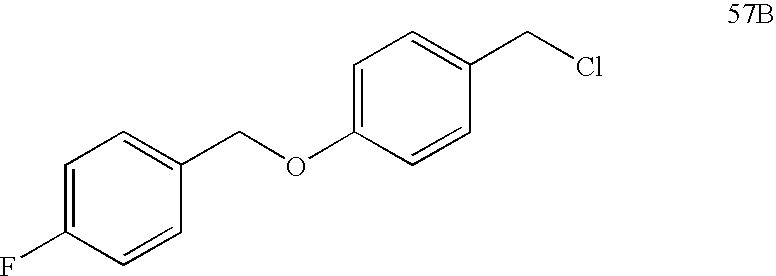
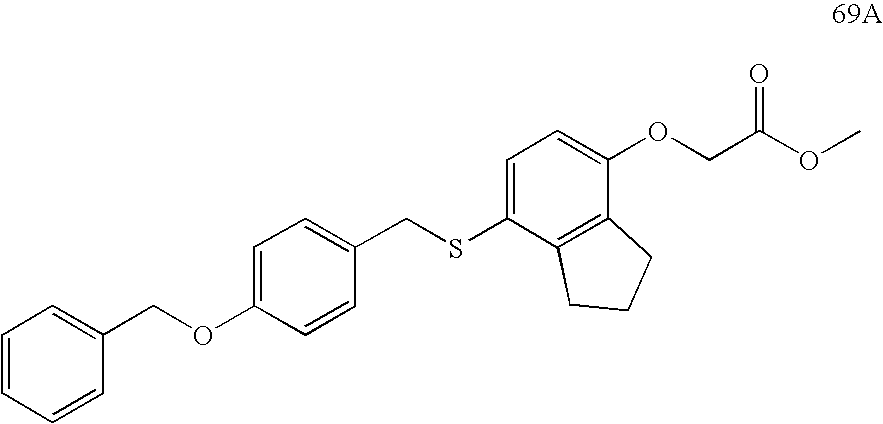
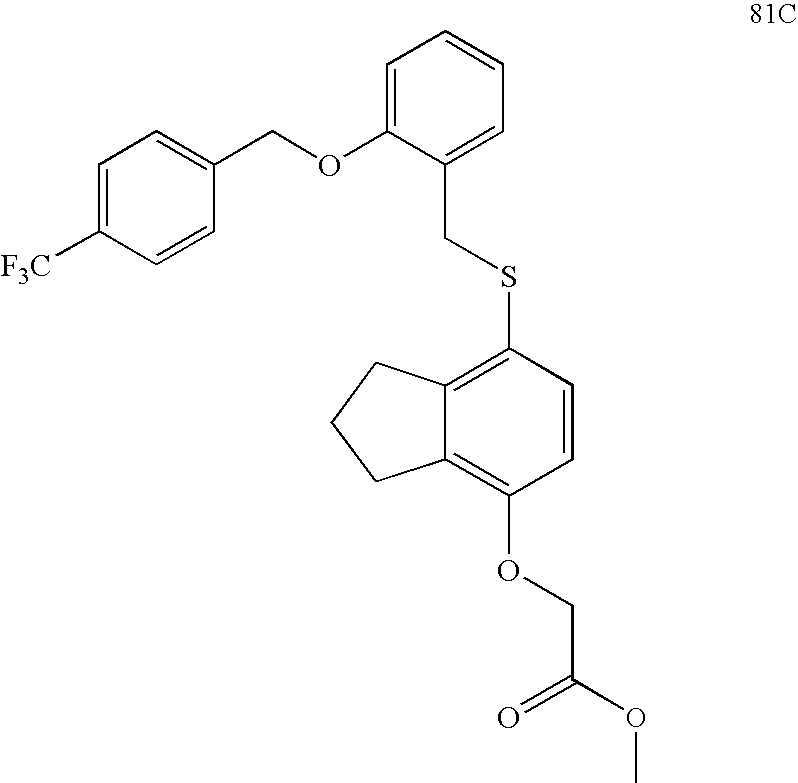
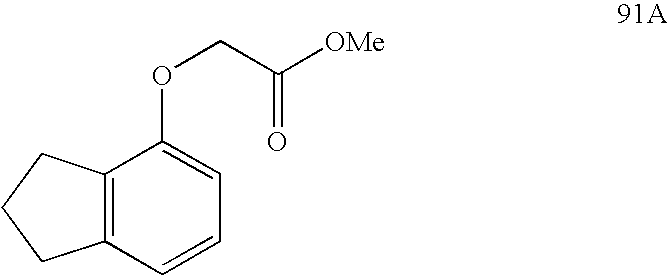
USRE39916E1 - Compounds that modulate PPAR activity and methods for their preparation - Google Patents
Compounds that modulate PPAR activity and methods for their preparation Download PDFInfo
- Publication number
- USRE39916E1 USRE39916E1 US11/288,022 US28802205A USRE39916E US RE39916 E1 USRE39916 E1 US RE39916E1 US 28802205 A US28802205 A US 28802205A US RE39916 E USRE39916 E US RE39916E
- Authority
- US
- United States
- Prior art keywords
- acetic acid
- compound
- methyl
- phenoxy
- trifluoromethyl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related, expires
Links
- 150000001875 compounds Chemical class 0.000 title claims abstract description 690
- 238000000034 method Methods 0.000 title claims abstract description 32
- 230000000694 effects Effects 0.000 title abstract description 13
- 108090000029 Peroxisome Proliferator-Activated Receptors Proteins 0.000 title abstract description 6
- 102000003728 Peroxisome Proliferator-Activated Receptors Human genes 0.000 title abstract description 6
- 238000002360 preparation method Methods 0.000 title description 306
- 239000000203 mixture Substances 0.000 claims abstract description 55
- 150000003839 salts Chemical class 0.000 claims abstract description 27
- 241000124008 Mammalia Species 0.000 claims abstract description 23
- 206010012601 diabetes mellitus Diseases 0.000 claims abstract description 21
- 102000016267 Leptin Human genes 0.000 claims abstract description 13
- 108010092277 Leptin Proteins 0.000 claims abstract description 13
- 208000008589 Obesity Diseases 0.000 claims abstract description 13
- NRYBAZVQPHGZNS-ZSOCWYAHSA-N leptin Chemical compound O=C([C@H](CO)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](CC=1C2=CC=CC=C2NC=1)NC(=O)[C@H](CC(C)C)NC(=O)[C@@H](NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CO)NC(=O)CNC(=O)[C@H](CCC(N)=O)NC(=O)[C@@H](N)CC(C)C)CCSC)N1CCC[C@H]1C(=O)NCC(=O)N[C@@H](CS)C(O)=O NRYBAZVQPHGZNS-ZSOCWYAHSA-N 0.000 claims abstract description 13
- 229940039781 leptin Drugs 0.000 claims abstract description 13
- 235000020824 obesity Nutrition 0.000 claims abstract description 13
- 201000001421 hyperglycemia Diseases 0.000 claims abstract description 8
- 206010060378 Hyperinsulinaemia Diseases 0.000 claims abstract description 7
- 230000036528 appetite Effects 0.000 claims abstract description 7
- 235000019789 appetite Nutrition 0.000 claims abstract description 7
- 230000003451 hyperinsulinaemic effect Effects 0.000 claims abstract description 7
- 201000008980 hyperinsulinism Diseases 0.000 claims abstract description 7
- 208000030814 Eating disease Diseases 0.000 claims abstract description 5
- 208000019454 Feeding and Eating disease Diseases 0.000 claims abstract description 5
- 235000014632 disordered eating Nutrition 0.000 claims abstract description 5
- -1 ethoxy, isopropoxy, n-propoxy, t-butoxy, n-butoxy Chemical group 0.000 claims description 124
- 125000000217 alkyl group Chemical group 0.000 claims description 78
- QTBSBXVTEAMEQO-UHFFFAOYSA-N acetic acid Substances CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 claims description 66
- 229910052739 hydrogen Inorganic materials 0.000 claims description 44
- 239000001257 hydrogen Substances 0.000 claims description 44
- 125000003545 alkoxy group Chemical group 0.000 claims description 43
- 229910052736 halogen Inorganic materials 0.000 claims description 33
- 125000004093 cyano group Chemical group *C#N 0.000 claims description 31
- 150000002367 halogens Chemical class 0.000 claims description 31
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 claims description 31
- 125000001188 haloalkyl group Chemical group 0.000 claims description 27
- 150000002431 hydrogen Chemical class 0.000 claims description 26
- 125000002023 trifluoromethyl group Chemical group FC(F)(F)* 0.000 claims description 22
- 229910052717 sulfur Inorganic materials 0.000 claims description 19
- 229910052760 oxygen Inorganic materials 0.000 claims description 16
- YBYRUZQLTYRCRF-UHFFFAOYSA-N 2-[5-methoxy-2-methyl-4-[[4-[4-(trifluoromethyl)phenyl]phenyl]methylsulfanyl]phenoxy]acetic acid Chemical compound COC1=CC(OCC(O)=O)=C(C)C=C1SCC1=CC=C(C=2C=CC(=CC=2)C(F)(F)F)C=C1 YBYRUZQLTYRCRF-UHFFFAOYSA-N 0.000 claims description 15
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 15
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims description 12
- WPLMYOVJSNXPMK-UHFFFAOYSA-N 2-[5-methoxy-2-methyl-4-[[4-[3-(trifluoromethyl)phenyl]phenyl]methylsulfanyl]phenoxy]acetic acid Chemical compound COC1=CC(OCC(O)=O)=C(C)C=C1SCC1=CC=C(C=2C=C(C=CC=2)C(F)(F)F)C=C1 WPLMYOVJSNXPMK-UHFFFAOYSA-N 0.000 claims description 9
- QZCOZWYSOAUBJR-UHFFFAOYSA-N 2-[4-[[4-(2,4-dichlorophenyl)phenyl]methylsulfanyl]-5-methoxy-2-methylphenoxy]acetic acid Chemical compound COC1=CC(OCC(O)=O)=C(C)C=C1SCC1=CC=C(C=2C(=CC(Cl)=CC=2)Cl)C=C1 QZCOZWYSOAUBJR-UHFFFAOYSA-N 0.000 claims description 8
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 claims description 8
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 claims description 8
- 239000008103 glucose Substances 0.000 claims description 8
- VFQRJNOXLNYYTE-UHFFFAOYSA-N 2-[3-methoxy-4-[[4-[4-(trifluoromethyl)phenyl]phenyl]methylsulfanyl]phenoxy]acetic acid Chemical compound COC1=CC(OCC(O)=O)=CC=C1SCC1=CC=C(C=2C=CC(=CC=2)C(F)(F)F)C=C1 VFQRJNOXLNYYTE-UHFFFAOYSA-N 0.000 claims description 7
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 claims description 7
- 125000004076 pyridyl group Chemical group 0.000 claims description 7
- UXBLXTMVLIEOGI-UHFFFAOYSA-N 2-[4-[[4-(4-fluorophenyl)phenyl]methylsulfanyl]-5-methoxy-2-methylphenoxy]acetic acid Chemical compound COC1=CC(OCC(O)=O)=C(C)C=C1SCC1=CC=C(C=2C=CC(F)=CC=2)C=C1 UXBLXTMVLIEOGI-UHFFFAOYSA-N 0.000 claims description 6
- COOMMSHFMOKEKM-UHFFFAOYSA-N 2-[5-methoxy-2-methyl-4-[(4-phenylphenyl)methylsulfanyl]phenoxy]acetic acid Chemical compound COC1=CC(OCC(O)=O)=C(C)C=C1SCC1=CC=C(C=2C=CC=CC=2)C=C1 COOMMSHFMOKEKM-UHFFFAOYSA-N 0.000 claims description 6
- VVUWGDAIOYKXPE-UHFFFAOYSA-N 2-[5-methoxy-2-methyl-4-[[3-[3-(trifluoromethoxy)phenyl]phenyl]methylsulfanyl]phenoxy]acetic acid Chemical compound COC1=CC(OCC(O)=O)=C(C)C=C1SCC1=CC=CC(C=2C=C(OC(F)(F)F)C=CC=2)=C1 VVUWGDAIOYKXPE-UHFFFAOYSA-N 0.000 claims description 6
- MANTXHYUNLPBHE-UHFFFAOYSA-N 2-[5-methoxy-2-methyl-4-[[3-[4-(trifluoromethyl)phenyl]phenyl]methylsulfanyl]phenoxy]acetic acid Chemical compound COC1=CC(OCC(O)=O)=C(C)C=C1SCC1=CC=CC(C=2C=CC(=CC=2)C(F)(F)F)=C1 MANTXHYUNLPBHE-UHFFFAOYSA-N 0.000 claims description 6
- ONKOHQZZVMTPOM-UHFFFAOYSA-N 2-[5-methoxy-2-methyl-4-[[4-[[4-(trifluoromethyl)phenyl]methoxy]phenyl]methylsulfanyl]phenoxy]acetic acid Chemical compound COC1=CC(OCC(O)=O)=C(C)C=C1SCC(C=C1)=CC=C1OCC1=CC=C(C(F)(F)F)C=C1 ONKOHQZZVMTPOM-UHFFFAOYSA-N 0.000 claims description 6
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims description 6
- 239000000546 pharmaceutical excipient Substances 0.000 claims description 6
- QPQVDUHBPMLTTD-UHFFFAOYSA-N 2-[4-[[4-[2-(3-fluorophenyl)ethenyl]phenyl]methylsulfanyl]-5-methoxy-2-methylphenoxy]acetic acid Chemical compound COC1=CC(OCC(O)=O)=C(C)C=C1SCC(C=C1)=CC=C1C=CC1=CC=CC(F)=C1 QPQVDUHBPMLTTD-UHFFFAOYSA-N 0.000 claims description 5
- HXCKESKWJSVFAR-UHFFFAOYSA-N 2-[5-methoxy-2-methyl-4-[[2-methyl-4-[4-(trifluoromethyl)phenyl]phenyl]methylsulfanyl]phenoxy]acetic acid Chemical compound COC1=CC(OCC(O)=O)=C(C)C=C1SCC1=CC=C(C=2C=CC(=CC=2)C(F)(F)F)C=C1C HXCKESKWJSVFAR-UHFFFAOYSA-N 0.000 claims description 5
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 claims description 5
- 208000001072 type 2 diabetes mellitus Diseases 0.000 claims description 5
- 239000003085 diluting agent Substances 0.000 claims description 4
- 208000035475 disorder Diseases 0.000 claims description 4
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 claims description 4
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 claims description 4
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 claims description 4
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 claims description 4
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 claims description 4
- HEMOEZGWSUFMTR-UHFFFAOYSA-N 2-[5-methoxy-2-methyl-4-[[6-[4-(trifluoromethyl)phenyl]pyridin-3-yl]methylsulfanyl]phenoxy]acetic acid Chemical compound COC1=CC(OCC(O)=O)=C(C)C=C1SCC1=CC=C(C=2C=CC(=CC=2)C(F)(F)F)N=C1 HEMOEZGWSUFMTR-UHFFFAOYSA-N 0.000 claims description 3
- 125000004199 4-trifluoromethylphenyl group Chemical group [H]C1=C([H])C(=C([H])C([H])=C1*)C(F)(F)F 0.000 claims description 3
- 102000051325 Glucagon Human genes 0.000 claims description 3
- 108060003199 Glucagon Proteins 0.000 claims description 3
- 102000018997 Growth Hormone Human genes 0.000 claims description 3
- 108010051696 Growth Hormone Proteins 0.000 claims description 3
- 102000003982 Parathyroid hormone Human genes 0.000 claims description 3
- 108090000445 Parathyroid hormone Proteins 0.000 claims description 3
- 150000003943 catecholamines Chemical class 0.000 claims description 3
- MASNOZXLGMXCHN-ZLPAWPGGSA-N glucagon Chemical compound C([C@@H](C(=O)N[C@H](C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(O)=O)C(C)C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](C)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CO)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CO)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@H](CC=1C=CC=CC=1)NC(=O)[C@@H](NC(=O)CNC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](N)CC=1NC=NC=1)[C@@H](C)O)[C@@H](C)O)C1=CC=CC=C1 MASNOZXLGMXCHN-ZLPAWPGGSA-N 0.000 claims description 3
- 229960004666 glucagon Drugs 0.000 claims description 3
- 239000003862 glucocorticoid Substances 0.000 claims description 3
- 239000000122 growth hormone Substances 0.000 claims description 3
- 239000000199 parathyroid hormone Substances 0.000 claims description 3
- 229960001319 parathyroid hormone Drugs 0.000 claims description 3
- 239000008194 pharmaceutical composition Substances 0.000 claims description 3
- 229940037128 systemic glucocorticoids Drugs 0.000 claims description 3
- 230000001419 dependent effect Effects 0.000 claims description 2
- 230000001747 exhibiting effect Effects 0.000 claims description 2
- 125000002510 isobutoxy group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])O* 0.000 claims description 2
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 claims description 2
- 208000001145 Metabolic Syndrome Diseases 0.000 claims 1
- 201000000690 abdominal obesity-metabolic syndrome Diseases 0.000 claims 1
- 208000035150 Hypercholesterolemia Diseases 0.000 abstract description 12
- 201000001320 Atherosclerosis Diseases 0.000 abstract description 8
- 239000003814 drug Substances 0.000 abstract description 7
- 208000006575 hypertriglyceridemia Diseases 0.000 abstract description 7
- 229940124597 therapeutic agent Drugs 0.000 abstract description 3
- IAZDPXIOMUYVGZ-WFGJKAKNSA-N Dimethyl sulfoxide Chemical compound [2H]C([2H])([2H])S(=O)C([2H])([2H])[2H] IAZDPXIOMUYVGZ-WFGJKAKNSA-N 0.000 description 254
- 238000005160 1H NMR spectroscopy Methods 0.000 description 136
- 238000003786 synthesis reaction Methods 0.000 description 106
- 230000015572 biosynthetic process Effects 0.000 description 103
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 93
- SCBACYIIYHCAPL-UHFFFAOYSA-N methyl 2-[5-methoxy-2-methyl-4-[(4-phenylphenyl)methylsulfanyl]phenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC(OC)=C1SCC1=CC=C(C=2C=CC=CC=2)C=C1 SCBACYIIYHCAPL-UHFFFAOYSA-N 0.000 description 86
- 239000000047 product Substances 0.000 description 65
- 229910001868 water Inorganic materials 0.000 description 62
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 60
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 58
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 57
- 229960000583 acetic acid Drugs 0.000 description 55
- 239000000243 solution Substances 0.000 description 51
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical class CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 49
- NOESYZHRGYRDHS-UHFFFAOYSA-N insulin Chemical compound N1C(=O)C(NC(=O)C(CCC(N)=O)NC(=O)C(CCC(O)=O)NC(=O)C(C(C)C)NC(=O)C(NC(=O)CN)C(C)CC)CSSCC(C(NC(CO)C(=O)NC(CC(C)C)C(=O)NC(CC=2C=CC(O)=CC=2)C(=O)NC(CCC(N)=O)C(=O)NC(CC(C)C)C(=O)NC(CCC(O)=O)C(=O)NC(CC(N)=O)C(=O)NC(CC=2C=CC(O)=CC=2)C(=O)NC(CSSCC(NC(=O)C(C(C)C)NC(=O)C(CC(C)C)NC(=O)C(CC=2C=CC(O)=CC=2)NC(=O)C(CC(C)C)NC(=O)C(C)NC(=O)C(CCC(O)=O)NC(=O)C(C(C)C)NC(=O)C(CC(C)C)NC(=O)C(CC=2NC=NC=2)NC(=O)C(CO)NC(=O)CNC2=O)C(=O)NCC(=O)NC(CCC(O)=O)C(=O)NC(CCCNC(N)=N)C(=O)NCC(=O)NC(CC=3C=CC=CC=3)C(=O)NC(CC=3C=CC=CC=3)C(=O)NC(CC=3C=CC(O)=CC=3)C(=O)NC(C(C)O)C(=O)N3C(CCC3)C(=O)NC(CCCCN)C(=O)NC(C)C(O)=O)C(=O)NC(CC(N)=O)C(O)=O)=O)NC(=O)C(C(C)CC)NC(=O)C(CO)NC(=O)C(C(C)O)NC(=O)C1CSSCC2NC(=O)C(CC(C)C)NC(=O)C(NC(=O)C(CCC(N)=O)NC(=O)C(CC(N)=O)NC(=O)C(NC(=O)C(N)CC=1C=CC=CC=1)C(C)C)CC1=CN=CN1 NOESYZHRGYRDHS-UHFFFAOYSA-N 0.000 description 38
- 235000019439 ethyl acetate Nutrition 0.000 description 37
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 36
- 0 [1*]C1=C(OCC(=O)O)C([3*])=C([4*])C(CCCCCC)=C1[2*] Chemical compound [1*]C1=C(OCC(=O)O)C([3*])=C([4*])C(CCCCCC)=C1[2*] 0.000 description 35
- BVJSUAQZOZWCKN-UHFFFAOYSA-N p-hydroxybenzyl alcohol Chemical compound OCC1=CC=C(O)C=C1 BVJSUAQZOZWCKN-UHFFFAOYSA-N 0.000 description 35
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 33
- 238000006243 chemical reaction Methods 0.000 description 33
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 27
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 24
- URYHYZRICUTYIL-UHFFFAOYSA-N [4-[[4-(trifluoromethyl)phenyl]methoxy]phenyl]methanol Chemical compound C1=CC(CO)=CC=C1OCC1=CC=C(C(F)(F)F)C=C1 URYHYZRICUTYIL-UHFFFAOYSA-N 0.000 description 24
- 239000012267 brine Substances 0.000 description 23
- HPALAKNZSZLMCH-UHFFFAOYSA-M sodium;chloride;hydrate Chemical compound O.[Na+].[Cl-] HPALAKNZSZLMCH-UHFFFAOYSA-M 0.000 description 23
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 21
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 20
- 125000003118 aryl group Chemical group 0.000 description 20
- 125000004429 atom Chemical group 0.000 description 20
- 102000004877 Insulin Human genes 0.000 description 19
- 108090001061 Insulin Proteins 0.000 description 19
- 229940125396 insulin Drugs 0.000 description 19
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 18
- 239000012043 crude product Substances 0.000 description 18
- 239000011541 reaction mixture Substances 0.000 description 17
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 16
- 125000000304 alkynyl group Chemical group 0.000 description 16
- 125000001309 chloro group Chemical group Cl* 0.000 description 16
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 15
- FJDQFPXHSGXQBY-UHFFFAOYSA-L caesium carbonate Chemical compound [Cs+].[Cs+].[O-]C([O-])=O FJDQFPXHSGXQBY-UHFFFAOYSA-L 0.000 description 15
- 125000001072 heteroaryl group Chemical group 0.000 description 15
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 14
- HEDRZPFGACZZDS-MICDWDOJSA-N Trichloro(2H)methane Chemical compound [2H]C(Cl)(Cl)Cl HEDRZPFGACZZDS-MICDWDOJSA-N 0.000 description 14
- 229910000024 caesium carbonate Inorganic materials 0.000 description 14
- HVYWMOMLDIMFJA-DPAQBDIFSA-N cholesterol Chemical compound C1C=C2C[C@@H](O)CC[C@]2(C)[C@@H]2[C@@H]1[C@@H]1CC[C@H]([C@H](C)CCCC(C)C)[C@@]1(C)CC2 HVYWMOMLDIMFJA-DPAQBDIFSA-N 0.000 description 14
- 238000004587 chromatography analysis Methods 0.000 description 14
- 235000019441 ethanol Nutrition 0.000 description 14
- 150000002430 hydrocarbons Chemical group 0.000 description 14
- 239000002904 solvent Substances 0.000 description 14
- 239000010409 thin film Substances 0.000 description 14
- YCVFDTRUBSFXSA-UHFFFAOYSA-N [4-[4-(trifluoromethyl)phenyl]phenyl]methanol Chemical compound C1=CC(CO)=CC=C1C1=CC=C(C(F)(F)F)C=C1 YCVFDTRUBSFXSA-UHFFFAOYSA-N 0.000 description 13
- 125000000753 cycloalkyl group Chemical group 0.000 description 13
- 125000001424 substituent group Chemical group 0.000 description 13
- 150000003573 thiols Chemical class 0.000 description 13
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 12
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical compound [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 description 12
- 125000004432 carbon atom Chemical group C* 0.000 description 12
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 12
- 208000032928 Dyslipidaemia Diseases 0.000 description 11
- 208000017170 Lipid metabolism disease Diseases 0.000 description 11
- KDLHZDBZIXYQEI-UHFFFAOYSA-N Palladium Chemical compound [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 description 11
- 150000002148 esters Chemical class 0.000 description 11
- IYQKPXHVUDFXKX-UHFFFAOYSA-N methyl 2-(5-methoxy-2-methyl-4-sulfanylphenoxy)acetate Chemical compound COC(=O)COC1=CC(OC)=C(S)C=C1C IYQKPXHVUDFXKX-UHFFFAOYSA-N 0.000 description 11
- WMFOQBRAJBCJND-UHFFFAOYSA-M Lithium hydroxide Chemical compound [Li+].[OH-] WMFOQBRAJBCJND-UHFFFAOYSA-M 0.000 description 10
- 125000003342 alkenyl group Chemical group 0.000 description 10
- 210000004027 cell Anatomy 0.000 description 10
- 238000000746 purification Methods 0.000 description 10
- 229910052938 sodium sulfate Inorganic materials 0.000 description 10
- YRXGIYXRCPNLQO-UHFFFAOYSA-N (4-hydroxy-2-methoxy-5-methylphenyl) thiocyanate Chemical compound COC1=CC(O)=C(C)C=C1SC#N YRXGIYXRCPNLQO-UHFFFAOYSA-N 0.000 description 9
- CNJKMXPPKMCCEJ-UHFFFAOYSA-N 2-[[7-[[5-(4-chlorophenyl)-1,2-oxazol-3-yl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetic acid Chemical compound C1=2CCCC=2C(OCC(=O)O)=CC=C1SCC(=NO1)C=C1C1=CC=C(Cl)C=C1 CNJKMXPPKMCCEJ-UHFFFAOYSA-N 0.000 description 9
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 9
- 239000000706 filtrate Substances 0.000 description 9
- 238000009472 formulation Methods 0.000 description 9
- 125000005842 heteroatom Chemical group 0.000 description 9
- 125000000951 phenoxy group Chemical group [H]C1=C([H])C([H])=C(O*)C([H])=C1[H] 0.000 description 9
- 125000005309 thioalkoxy group Chemical group 0.000 description 9
- BWJRRXJWVDNQOQ-UHFFFAOYSA-N 2-[4-[[5-(4-chlorophenyl)-1,2-oxazol-3-yl]methylsulfanyl]-2-methylphenoxy]acetic acid Chemical compound C1=C(OCC(O)=O)C(C)=CC(SCC2=NOC(=C2)C=2C=CC(Cl)=CC=2)=C1 BWJRRXJWVDNQOQ-UHFFFAOYSA-N 0.000 description 8
- 230000037396 body weight Effects 0.000 description 8
- 239000003054 catalyst Substances 0.000 description 8
- 125000000623 heterocyclic group Chemical group 0.000 description 8
- 239000004615 ingredient Substances 0.000 description 8
- 229910052757 nitrogen Inorganic materials 0.000 description 8
- 229920006395 saturated elastomer Polymers 0.000 description 8
- DTQVDTLACAAQTR-UHFFFAOYSA-N trifluoroacetic acid Substances OC(=O)C(F)(F)F DTQVDTLACAAQTR-UHFFFAOYSA-N 0.000 description 8
- 125000004169 (C1-C6) alkyl group Chemical group 0.000 description 7
- YBKKDSJIPWMTNX-UHFFFAOYSA-N 2-[2-butyl-4-[[4-[4-(trifluoromethyl)phenyl]phenyl]methylsulfanyl]phenoxy]acetic acid Chemical compound C1=C(OCC(O)=O)C(CCCC)=CC(SCC=2C=CC(=CC=2)C=2C=CC(=CC=2)C(F)(F)F)=C1 YBKKDSJIPWMTNX-UHFFFAOYSA-N 0.000 description 7
- RZHUKZMLRKWZGB-UHFFFAOYSA-N 2-[2-propyl-4-[[4-[4-(trifluoromethyl)phenyl]phenyl]methylsulfanyl]phenoxy]acetic acid Chemical compound C1=C(OCC(O)=O)C(CCC)=CC(SCC=2C=CC(=CC=2)C=2C=CC(=CC=2)C(F)(F)F)=C1 RZHUKZMLRKWZGB-UHFFFAOYSA-N 0.000 description 7
- LCHAXLZFOCAPKE-UHFFFAOYSA-N 2-[4-[[4-(3,4-dichlorophenyl)phenyl]methylsulfanyl]-5-methoxy-2-methylphenoxy]acetic acid Chemical compound COC1=CC(OCC(O)=O)=C(C)C=C1SCC1=CC=C(C=2C=C(Cl)C(Cl)=CC=2)C=C1 LCHAXLZFOCAPKE-UHFFFAOYSA-N 0.000 description 7
- VNFBVJKCXNXPQO-UHFFFAOYSA-N 2-[4-[[4-[4-chloro-3-(trifluoromethyl)phenyl]phenyl]methylsulfanyl]-2-methylphenoxy]acetic acid Chemical compound C1=C(OCC(O)=O)C(C)=CC(SCC=2C=CC(=CC=2)C=2C=C(C(Cl)=CC=2)C(F)(F)F)=C1 VNFBVJKCXNXPQO-UHFFFAOYSA-N 0.000 description 7
- AQTCHGAIFYVITF-UHFFFAOYSA-N 2-[[4-[[5-[4-(trifluoromethyl)phenyl]-1,2-oxazol-3-yl]methylsulfanyl]-5,6,7,8-tetrahydronaphthalen-1-yl]oxy]acetic acid Chemical compound C1=2CCCCC=2C(OCC(=O)O)=CC=C1SCC(=NO1)C=C1C1=CC=C(C(F)(F)F)C=C1 AQTCHGAIFYVITF-UHFFFAOYSA-N 0.000 description 7
- GHVCJPROFSJODO-UHFFFAOYSA-N 2-[[7-[2-[4-[4-(trifluoromethyl)phenyl]phenyl]ethyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetic acid Chemical compound C1=2CCCC=2C(OCC(=O)O)=CC=C1CCC(C=C1)=CC=C1C1=CC=C(C(F)(F)F)C=C1 GHVCJPROFSJODO-UHFFFAOYSA-N 0.000 description 7
- HRHULDYBWAZVGT-UHFFFAOYSA-N 2-[[7-[2-[4-[4-(trifluoromethyl)phenyl]phenyl]ethylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetic acid Chemical compound C1=2CCCC=2C(OCC(=O)O)=CC=C1SCCC(C=C1)=CC=C1C1=CC=C(C(F)(F)F)C=C1 HRHULDYBWAZVGT-UHFFFAOYSA-N 0.000 description 7
- UQDCLZWSSZJCMF-UHFFFAOYSA-N 2-[[7-[[2-[[4-(trifluoromethyl)phenyl]methoxy]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetic acid Chemical compound C1=2CCCC=2C(OCC(=O)O)=CC=C1SCC1=CC=CC=C1OCC1=CC=C(C(F)(F)F)C=C1 UQDCLZWSSZJCMF-UHFFFAOYSA-N 0.000 description 7
- HSGSQAUAZSYHNY-UHFFFAOYSA-N 2-[[7-[[3-[4-(trifluoromethyl)phenyl]-1,2-oxazol-5-yl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetic acid Chemical compound C1=2CCCC=2C(OCC(=O)O)=CC=C1SCC(ON=1)=CC=1C1=CC=C(C(F)(F)F)C=C1 HSGSQAUAZSYHNY-UHFFFAOYSA-N 0.000 description 7
- VHZIVPTZCQFHTF-UHFFFAOYSA-N 2-[[7-[[3-[[4-(trifluoromethyl)phenyl]methoxy]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetic acid Chemical compound C1=2CCCC=2C(OCC(=O)O)=CC=C1SCC(C=1)=CC=CC=1OCC1=CC=C(C(F)(F)F)C=C1 VHZIVPTZCQFHTF-UHFFFAOYSA-N 0.000 description 7
- ITECITAWVCJVQI-UHFFFAOYSA-N 2-[[7-[[3-[[4-chloro-3-(trifluoromethyl)phenyl]methoxy]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetic acid Chemical compound C1=2CCCC=2C(OCC(=O)O)=CC=C1SCC(C=1)=CC=CC=1OCC1=CC=C(Cl)C(C(F)(F)F)=C1 ITECITAWVCJVQI-UHFFFAOYSA-N 0.000 description 7
- MQQUYERLSYUAIA-UHFFFAOYSA-N 2-[[7-[[4-[[4-(trifluoromethoxy)phenyl]methoxy]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetic acid Chemical compound C1=2CCCC=2C(OCC(=O)O)=CC=C1SCC(C=C1)=CC=C1OCC1=CC=C(OC(F)(F)F)C=C1 MQQUYERLSYUAIA-UHFFFAOYSA-N 0.000 description 7
- DCIFLBVKPAOZLT-UHFFFAOYSA-N 2-[[7-[[4-methoxy-3-[[4-(trifluoromethyl)phenyl]methoxy]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetic acid Chemical compound COC1=CC=C(CSC=2C=3CCCC=3C(OCC(O)=O)=CC=2)C=C1OCC1=CC=C(C(F)(F)F)C=C1 DCIFLBVKPAOZLT-UHFFFAOYSA-N 0.000 description 7
- GVCVLTVAANYNQB-UHFFFAOYSA-N 2-[[8-[[4-[(2,5-dichlorophenyl)methoxy]phenyl]methylsulfanyl]-3,4-dihydro-2h-chromen-5-yl]oxy]acetic acid Chemical compound C1=2OCCCC=2C(OCC(=O)O)=CC=C1SCC(C=C1)=CC=C1OCC1=CC(Cl)=CC=C1Cl GVCVLTVAANYNQB-UHFFFAOYSA-N 0.000 description 7
- CBLNYVUYNXMNOU-UHFFFAOYSA-N 2-[[8-[[4-[4-(trifluoromethyl)phenyl]phenyl]methylsulfanyl]-3,4-dihydro-2h-chromen-5-yl]oxy]acetic acid Chemical compound C1=2OCCCC=2C(OCC(=O)O)=CC=C1SCC(C=C1)=CC=C1C1=CC=C(C(F)(F)F)C=C1 CBLNYVUYNXMNOU-UHFFFAOYSA-N 0.000 description 7
- TWJAIHIMIYYEFE-UHFFFAOYSA-N 2-[[8-[[5-(4-chlorophenyl)-1,2-oxazol-3-yl]methylsulfanyl]-3,4-dihydro-2h-chromen-5-yl]oxy]acetic acid Chemical compound C1=2OCCCC=2C(OCC(=O)O)=CC=C1SCC(=NO1)C=C1C1=CC=C(Cl)C=C1 TWJAIHIMIYYEFE-UHFFFAOYSA-N 0.000 description 7
- 239000007832 Na2SO4 Substances 0.000 description 7
- ISWSIDIOOBJBQZ-UHFFFAOYSA-N Phenol Chemical compound OC1=CC=CC=C1 ISWSIDIOOBJBQZ-UHFFFAOYSA-N 0.000 description 7
- 230000004913 activation Effects 0.000 description 7
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 7
- 239000000284 extract Substances 0.000 description 7
- JNADCJAWSTUCPF-UHFFFAOYSA-N methyl 2-[(8-sulfanyl-3,4-dihydro-2h-chromen-5-yl)oxy]acetate Chemical compound O1CCCC2=C1C(S)=CC=C2OCC(=O)OC JNADCJAWSTUCPF-UHFFFAOYSA-N 0.000 description 7
- JNYTWLSMXNXSRX-UHFFFAOYSA-N methyl 2-[5-methoxy-2-methyl-4-[[4-(2,4,6-trimethylphenyl)phenyl]methylsulfanyl]phenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC(OC)=C1SCC1=CC=C(C=2C(=CC(C)=CC=2C)C)C=C1 JNYTWLSMXNXSRX-UHFFFAOYSA-N 0.000 description 7
- YDCHPLOFQATIDS-UHFFFAOYSA-N methyl 2-bromoacetate Chemical group COC(=O)CBr YDCHPLOFQATIDS-UHFFFAOYSA-N 0.000 description 7
- 239000012044 organic layer Substances 0.000 description 7
- DPHNJPUOMLRELT-UHFFFAOYSA-N 2,3-dihydro-1h-inden-4-ol Chemical compound OC1=CC=CC2=C1CCC2 DPHNJPUOMLRELT-UHFFFAOYSA-N 0.000 description 6
- KVOXUCYWRVOFDX-UHFFFAOYSA-N 2-[4-[[4-[(2,4-dichlorophenyl)methoxy]phenyl]methylsulfanyl]-2-methylphenoxy]acetic acid Chemical compound C1=C(OCC(O)=O)C(C)=CC(SCC=2C=CC(OCC=3C(=CC(Cl)=CC=3)Cl)=CC=2)=C1 KVOXUCYWRVOFDX-UHFFFAOYSA-N 0.000 description 6
- YHUGHNVXVQYASV-UHFFFAOYSA-N 2-[5-methoxy-2-methyl-4-[[2-[[4-(trifluoromethyl)phenyl]methoxy]phenyl]methylsulfanyl]phenoxy]acetic acid Chemical compound COC1=CC(OCC(O)=O)=C(C)C=C1SCC1=CC=CC=C1OCC1=CC=C(C(F)(F)F)C=C1 YHUGHNVXVQYASV-UHFFFAOYSA-N 0.000 description 6
- VYYLCIGYUQXTIM-UHFFFAOYSA-N 2-[[5-methyl-7-[[4-[4-(trifluoromethyl)phenyl]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetic acid Chemical compound C=12CCCC2=C(OCC(O)=O)C(C)=CC=1SCC(C=C1)=CC=C1C1=CC=C(C(F)(F)F)C=C1 VYYLCIGYUQXTIM-UHFFFAOYSA-N 0.000 description 6
- HIPQZFVWHPNRMY-UHFFFAOYSA-N 2-[[8-[[5-[4-(trifluoromethyl)phenyl]-1,2-oxazol-3-yl]methylsulfanyl]-3,4-dihydro-2h-chromen-5-yl]oxy]acetic acid Chemical compound C1=2OCCCC=2C(OCC(=O)O)=CC=C1SCC(=NO1)C=C1C1=CC=C(C(F)(F)F)C=C1 HIPQZFVWHPNRMY-UHFFFAOYSA-N 0.000 description 6
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical compound [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 6
- 239000012359 Methanesulfonyl chloride Substances 0.000 description 6
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 6
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical compound [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 6
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 6
- 150000001298 alcohols Chemical class 0.000 description 6
- ILAHWRKJUDSMFH-UHFFFAOYSA-N boron tribromide Chemical compound BrB(Br)Br ILAHWRKJUDSMFH-UHFFFAOYSA-N 0.000 description 6
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 description 6
- 229910052794 bromium Inorganic materials 0.000 description 6
- 150000001721 carbon Chemical group 0.000 description 6
- 229910052799 carbon Inorganic materials 0.000 description 6
- JRONPIZRZBBOBR-UHFFFAOYSA-N dichlorine tetroxide Inorganic materials ClOCl(=O)(=O)=O JRONPIZRZBBOBR-UHFFFAOYSA-N 0.000 description 6
- 238000003818 flash chromatography Methods 0.000 description 6
- 238000004128 high performance liquid chromatography Methods 0.000 description 6
- QARBMVPHQWIHKH-UHFFFAOYSA-N methanesulfonyl chloride Chemical compound CS(Cl)(=O)=O QARBMVPHQWIHKH-UHFFFAOYSA-N 0.000 description 6
- VQVARLMPFSNMSU-UHFFFAOYSA-N methyl 2-(5-methoxy-2-methyl-4-thiocyanatophenoxy)acetate Chemical compound COC(=O)COC1=CC(OC)=C(SC#N)C=C1C VQVARLMPFSNMSU-UHFFFAOYSA-N 0.000 description 6
- IYBYTAAXISWWBK-UHFFFAOYSA-N methyl 2-[(4-sulfanyl-5,6,7,8-tetrahydronaphthalen-1-yl)oxy]acetate Chemical compound C1CCCC2=C1C(S)=CC=C2OCC(=O)OC IYBYTAAXISWWBK-UHFFFAOYSA-N 0.000 description 6
- NFHFRUOZVGFOOS-UHFFFAOYSA-N palladium;triphenylphosphane Chemical compound [Pd].C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1 NFHFRUOZVGFOOS-UHFFFAOYSA-N 0.000 description 6
- JHJLBTNAGRQEKS-UHFFFAOYSA-M sodium bromide Chemical compound [Na+].[Br-] JHJLBTNAGRQEKS-UHFFFAOYSA-M 0.000 description 6
- 229910000029 sodium carbonate Inorganic materials 0.000 description 6
- 238000003756 stirring Methods 0.000 description 6
- RIOQSEWOXXDEQQ-UHFFFAOYSA-N triphenylphosphine Chemical compound C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1 RIOQSEWOXXDEQQ-UHFFFAOYSA-N 0.000 description 6
- QULQHEYBWUAZAX-UHFFFAOYSA-N 1-(2-bromoethyl)-4-[4-(trifluoromethyl)phenyl]benzene Chemical group C1=CC(C(F)(F)F)=CC=C1C1=CC=C(CCBr)C=C1 QULQHEYBWUAZAX-UHFFFAOYSA-N 0.000 description 5
- IKSNDOVDVVPSMA-UHFFFAOYSA-N 1-(bromomethyl)-4-(trifluoromethyl)benzene Chemical compound FC(F)(F)C1=CC=C(CBr)C=C1 IKSNDOVDVVPSMA-UHFFFAOYSA-N 0.000 description 5
- WJVPZHJRLXHFDD-UHFFFAOYSA-N 2-[2-methyl-4-[(4-phenylmethoxyphenyl)methylsulfanyl]phenoxy]acetic acid Chemical compound C1=C(OCC(O)=O)C(C)=CC(SCC=2C=CC(OCC=3C=CC=CC=3)=CC=2)=C1 WJVPZHJRLXHFDD-UHFFFAOYSA-N 0.000 description 5
- GLMFWFPXBNNBJB-UHFFFAOYSA-N 2-[2-methyl-4-[[4-[5-(trifluoromethyl)pyridin-2-yl]phenyl]methylsulfanyl]phenoxy]acetic acid Chemical compound C1=C(OCC(O)=O)C(C)=CC(SCC=2C=CC(=CC=2)C=2N=CC(=CC=2)C(F)(F)F)=C1 GLMFWFPXBNNBJB-UHFFFAOYSA-N 0.000 description 5
- WYYCVASAOXBPQU-UHFFFAOYSA-N 2-[2-methyl-4-[[4-[[4-(trifluoromethoxy)phenyl]methoxy]phenyl]methylsulfanyl]phenoxy]acetic acid Chemical compound C1=C(OCC(O)=O)C(C)=CC(SCC=2C=CC(OCC=3C=CC(OC(F)(F)F)=CC=3)=CC=2)=C1 WYYCVASAOXBPQU-UHFFFAOYSA-N 0.000 description 5
- VUAOPQZQOPZBCT-UHFFFAOYSA-N 2-[2-methyl-4-[[5-[4-(trifluoromethyl)phenyl]-1,2-oxazol-3-yl]methylsulfanyl]phenoxy]acetic acid Chemical compound C1=C(OCC(O)=O)C(C)=CC(SCC2=NOC(=C2)C=2C=CC(=CC=2)C(F)(F)F)=C1 VUAOPQZQOPZBCT-UHFFFAOYSA-N 0.000 description 5
- JTXHHQDYEHXWPC-UHFFFAOYSA-N 2-[4-(chloromethyl)phenyl]-5-(trifluoromethyl)pyridine Chemical compound N1=CC(C(F)(F)F)=CC=C1C1=CC=C(CCl)C=C1 JTXHHQDYEHXWPC-UHFFFAOYSA-N 0.000 description 5
- PKYBEKBSGPDIKA-UHFFFAOYSA-N 2-[4-[(4-hydroxyphenyl)methylsulfanyl]-2-methylphenoxy]acetic acid Chemical compound C1=C(OCC(O)=O)C(C)=CC(SCC=2C=CC(O)=CC=2)=C1 PKYBEKBSGPDIKA-UHFFFAOYSA-N 0.000 description 5
- XKGDKLFNOAMAJK-UHFFFAOYSA-N 2-[4-[[4-[(2,4-difluorophenyl)methoxy]phenyl]methylsulfanyl]-2-methylphenoxy]acetic acid Chemical compound C1=C(OCC(O)=O)C(C)=CC(SCC=2C=CC(OCC=3C(=CC(F)=CC=3)F)=CC=2)=C1 XKGDKLFNOAMAJK-UHFFFAOYSA-N 0.000 description 5
- HTVWQHWCWRNJSL-UHFFFAOYSA-N 2-[4-[[4-[(4-methoxyphenyl)methoxy]phenyl]methylsulfanyl]-2-methylphenoxy]acetic acid Chemical compound C1=CC(OC)=CC=C1COC(C=C1)=CC=C1CSC1=CC=C(OCC(O)=O)C(C)=C1 HTVWQHWCWRNJSL-UHFFFAOYSA-N 0.000 description 5
- WYORLEBCNSSUNT-UHFFFAOYSA-N 2-[4-[[4-[(4-tert-butylphenyl)methoxy]phenyl]methylsulfanyl]-2-methylphenoxy]acetic acid Chemical compound C1=C(OCC(O)=O)C(C)=CC(SCC=2C=CC(OCC=3C=CC(=CC=3)C(C)(C)C)=CC=2)=C1 WYORLEBCNSSUNT-UHFFFAOYSA-N 0.000 description 5
- JLQGDIDQGNXZJJ-UHFFFAOYSA-N 2-[4-[[5-(4-chlorophenyl)-1,2-oxazol-3-yl]methylsulfanyl]-5-methoxy-2-methylphenoxy]acetic acid Chemical compound COC1=CC(OCC(O)=O)=C(C)C=C1SCC1=NOC(C=2C=CC(Cl)=CC=2)=C1 JLQGDIDQGNXZJJ-UHFFFAOYSA-N 0.000 description 5
- IJMQZWWCERGBDQ-UHFFFAOYSA-N 2-[5-chloro-2-methyl-4-[[4-[4-(trifluoromethyl)phenyl]phenyl]methylsulfanyl]phenoxy]acetic acid Chemical compound C1=C(OCC(O)=O)C(C)=CC(SCC=2C=CC(=CC=2)C=2C=CC(=CC=2)C(F)(F)F)=C1Cl IJMQZWWCERGBDQ-UHFFFAOYSA-N 0.000 description 5
- AJQNSRYZFLLBOI-UHFFFAOYSA-N 2-[5-methoxy-2-methyl-4-[2-[4-[4-(trifluoromethyl)phenyl]phenyl]ethylsulfanyl]phenoxy]acetic acid Chemical compound COC1=CC(OCC(O)=O)=C(C)C=C1SCCC1=CC=C(C=2C=CC(=CC=2)C(F)(F)F)C=C1 AJQNSRYZFLLBOI-UHFFFAOYSA-N 0.000 description 5
- GSPKTIOMMLSIHT-UHFFFAOYSA-N 2-[5-methoxy-2-methyl-4-[[3-[4-(trifluoromethyl)phenyl]-1,2-oxazol-5-yl]methylsulfanyl]phenoxy]acetic acid Chemical compound COC1=CC(OCC(O)=O)=C(C)C=C1SCC1=CC(C=2C=CC(=CC=2)C(F)(F)F)=NO1 GSPKTIOMMLSIHT-UHFFFAOYSA-N 0.000 description 5
- DMXGREBBKGWVCY-UHFFFAOYSA-N 2-[[5-methyl-7-[[4-[5-(trifluoromethyl)pyridin-2-yl]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetic acid Chemical compound C=12CCCC2=C(OCC(O)=O)C(C)=CC=1SCC(C=C1)=CC=C1C1=CC=C(C(F)(F)F)C=N1 DMXGREBBKGWVCY-UHFFFAOYSA-N 0.000 description 5
- KOEHFAAAJFPVKP-UHFFFAOYSA-N 2-[[6-methyl-8-[[4-[[4-(trifluoromethyl)phenyl]methoxy]phenyl]methylsulfanyl]-3,4-dihydro-2h-chromen-5-yl]oxy]acetic acid Chemical compound C=12OCCCC2=C(OCC(O)=O)C(C)=CC=1SCC(C=C1)=CC=C1OCC1=CC=C(C(F)(F)F)C=C1 KOEHFAAAJFPVKP-UHFFFAOYSA-N 0.000 description 5
- AOASBCKNMXGYCQ-UHFFFAOYSA-N 2-[[7-[[3-(4-chlorophenyl)-1,2-oxazol-5-yl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetic acid Chemical compound C1=2CCCC=2C(OCC(=O)O)=CC=C1SCC(ON=1)=CC=1C1=CC=C(Cl)C=C1 AOASBCKNMXGYCQ-UHFFFAOYSA-N 0.000 description 5
- WHSCCONPBHHTQY-UHFFFAOYSA-N 2-[[7-[[3-[5-(trifluoromethyl)pyridin-2-yl]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetic acid Chemical compound C1=2CCCC=2C(OCC(=O)O)=CC=C1SCC(C=1)=CC=CC=1C1=CC=C(C(F)(F)F)C=N1 WHSCCONPBHHTQY-UHFFFAOYSA-N 0.000 description 5
- XRQYYPOXSLDBNG-UHFFFAOYSA-N 2-[[7-[[4-[5-(trifluoromethyl)pyridin-2-yl]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetic acid Chemical compound C1=2CCCC=2C(OCC(=O)O)=CC=C1SCC(C=C1)=CC=C1C1=CC=C(C(F)(F)F)C=N1 XRQYYPOXSLDBNG-UHFFFAOYSA-N 0.000 description 5
- IVINVXPMKZERSK-UHFFFAOYSA-N 2-[[7-[[4-[[4-(trifluoromethyl)phenyl]methoxy]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetic acid Chemical compound C1=2CCCC=2C(OCC(=O)O)=CC=C1SCC(C=C1)=CC=C1OCC1=CC=C(C(F)(F)F)C=C1 IVINVXPMKZERSK-UHFFFAOYSA-N 0.000 description 5
- MADPEGDMHBXTST-UHFFFAOYSA-N 2-[[8-[[3-[[4-(trifluoromethyl)phenyl]methoxy]phenyl]methylsulfanyl]-3,4-dihydro-2h-chromen-5-yl]oxy]acetic acid Chemical compound C1=2OCCCC=2C(OCC(=O)O)=CC=C1SCC(C=1)=CC=CC=1OCC1=CC=C(C(F)(F)F)C=C1 MADPEGDMHBXTST-UHFFFAOYSA-N 0.000 description 5
- GJYCVCVHRSWLNY-UHFFFAOYSA-N 2-butylphenol Chemical compound CCCCC1=CC=CC=C1O GJYCVCVHRSWLNY-UHFFFAOYSA-N 0.000 description 5
- JKSRJLOJDZYJLA-UHFFFAOYSA-N 3-(chloromethyl)-5-(4-chlorophenyl)-1,2-oxazole Chemical compound O1N=C(CCl)C=C1C1=CC=C(Cl)C=C1 JKSRJLOJDZYJLA-UHFFFAOYSA-N 0.000 description 5
- 239000004215 Carbon black (E152) Substances 0.000 description 5
- 241000282693 Cercopithecidae Species 0.000 description 5
- 229910020323 ClF3 Inorganic materials 0.000 description 5
- 108020004414 DNA Proteins 0.000 description 5
- 108010010234 HDL Lipoproteins Proteins 0.000 description 5
- 102000015779 HDL Lipoproteins Human genes 0.000 description 5
- 241001465754 Metazoa Species 0.000 description 5
- 108010044210 PPAR-beta Proteins 0.000 description 5
- 230000029936 alkylation Effects 0.000 description 5
- 238000005804 alkylation reaction Methods 0.000 description 5
- 239000003795 chemical substances by application Substances 0.000 description 5
- 208000029078 coronary artery disease Diseases 0.000 description 5
- 238000010828 elution Methods 0.000 description 5
- 125000005843 halogen group Chemical group 0.000 description 5
- 229930195733 hydrocarbon Natural products 0.000 description 5
- GDCILPBOYQAPBE-UHFFFAOYSA-N methyl 2-(2,6-dimethyl-4-sulfanylphenoxy)acetate Chemical compound COC(=O)COC1=C(C)C=C(S)C=C1C GDCILPBOYQAPBE-UHFFFAOYSA-N 0.000 description 5
- QDEFPBCRTWICJS-UHFFFAOYSA-N methyl 2-(2-methyl-4-sulfanylphenoxy)acetate Chemical compound COC(=O)COC1=CC=C(S)C=C1C QDEFPBCRTWICJS-UHFFFAOYSA-N 0.000 description 5
- RALDOKCMDDMPQM-UHFFFAOYSA-N methyl 2-[2-methyl-4-[2-[4-[4-(trifluoromethyl)phenyl]phenyl]ethylsulfanyl]phenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC=C1SCCC1=CC=C(C=2C=CC(=CC=2)C(F)(F)F)C=C1 RALDOKCMDDMPQM-UHFFFAOYSA-N 0.000 description 5
- UEEACGHNHRRKJP-UHFFFAOYSA-N methyl 2-[4-[(4-acetyloxyphenyl)methylsulfanyl]-2-methylphenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC=C1SCC1=CC=C(OC(C)=O)C=C1 UEEACGHNHRRKJP-UHFFFAOYSA-N 0.000 description 5
- HDZBVDINICUNPE-UHFFFAOYSA-N methyl 2-[5-chloro-2-methyl-4-[[4-[5-(trifluoromethyl)pyridin-2-yl]phenyl]methylsulfanyl]phenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC(Cl)=C1SCC1=CC=C(C=2N=CC(=CC=2)C(F)(F)F)C=C1 HDZBVDINICUNPE-UHFFFAOYSA-N 0.000 description 5
- 229920001223 polyethylene glycol Polymers 0.000 description 5
- 239000000843 powder Substances 0.000 description 5
- 229930195734 saturated hydrocarbon Natural products 0.000 description 5
- 239000000741 silica gel Substances 0.000 description 5
- 229910002027 silica gel Inorganic materials 0.000 description 5
- 239000007787 solid Substances 0.000 description 5
- 125000003107 substituted aryl group Chemical group 0.000 description 5
- 239000011593 sulfur Substances 0.000 description 5
- 239000000725 suspension Substances 0.000 description 5
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 5
- 125000000335 thiazolyl group Chemical group 0.000 description 5
- 229940086542 triethylamine Drugs 0.000 description 5
- 229930195735 unsaturated hydrocarbon Natural products 0.000 description 5
- FAIBIGSVWBOKCX-UHFFFAOYSA-N (4-hydroxy-3,5-dimethylphenyl) thiocyanate Chemical compound CC1=CC(SC#N)=CC(C)=C1O FAIBIGSVWBOKCX-UHFFFAOYSA-N 0.000 description 4
- RXAOSYYRENLMBE-UHFFFAOYSA-N (7-hydroxy-2,3-dihydro-1h-inden-4-yl) thiocyanate Chemical compound OC1=CC=C(SC#N)C2=C1CCC2 RXAOSYYRENLMBE-UHFFFAOYSA-N 0.000 description 4
- KQFSFTCRHMKYBT-UHFFFAOYSA-N 1,3-dichloro-5-(chloromethyl)-2-[[4-(trifluoromethyl)phenyl]methoxy]benzene Chemical compound C1=CC(C(F)(F)F)=CC=C1COC1=C(Cl)C=C(CCl)C=C1Cl KQFSFTCRHMKYBT-UHFFFAOYSA-N 0.000 description 4
- NMALYCCZCNCSEP-UHFFFAOYSA-N 1-(chloromethyl)-4-[(4-methoxyphenyl)methoxy]benzene Chemical compound C1=CC(OC)=CC=C1COC1=CC=C(CCl)C=C1 NMALYCCZCNCSEP-UHFFFAOYSA-N 0.000 description 4
- XLQSXGGDTHANLN-UHFFFAOYSA-N 1-bromo-4-(trifluoromethyl)benzene Chemical compound FC(F)(F)C1=CC=C(Br)C=C1 XLQSXGGDTHANLN-UHFFFAOYSA-N 0.000 description 4
- NXEPZNPQVNREAC-UHFFFAOYSA-N 1-tert-butyl-4-[[4-(chloromethyl)phenoxy]methyl]benzene Chemical compound C1=CC(C(C)(C)C)=CC=C1COC1=CC=C(CCl)C=C1 NXEPZNPQVNREAC-UHFFFAOYSA-N 0.000 description 4
- VNJRLKXMLBJCEP-UHFFFAOYSA-N 2-[2-methyl-4-[(4-phenoxyphenyl)methylsulfanyl]phenoxy]acetic acid Chemical compound C1=C(OCC(O)=O)C(C)=CC(SCC=2C=CC(OC=3C=CC=CC=3)=CC=2)=C1 VNJRLKXMLBJCEP-UHFFFAOYSA-N 0.000 description 4
- OPNSCGQJMSUOHM-UHFFFAOYSA-N 2-[2-methyl-4-[2-[4-[4-(trifluoromethyl)phenyl]phenyl]ethenyl]phenoxy]acetic acid Chemical compound C1=C(OCC(O)=O)C(C)=CC(C=CC=2C=CC(=CC=2)C=2C=CC(=CC=2)C(F)(F)F)=C1 OPNSCGQJMSUOHM-UHFFFAOYSA-N 0.000 description 4
- WLAMDVXFHPQBSD-UHFFFAOYSA-N 2-[2-methyl-4-[2-[4-[4-(trifluoromethyl)phenyl]phenyl]ethylsulfanyl]phenoxy]acetic acid Chemical compound C1=C(OCC(O)=O)C(C)=CC(SCCC=2C=CC(=CC=2)C=2C=CC(=CC=2)C(F)(F)F)=C1 WLAMDVXFHPQBSD-UHFFFAOYSA-N 0.000 description 4
- GDYUMLCDKLRQLB-UHFFFAOYSA-N 2-[2-methyl-4-[[3-[4-(trifluoromethyl)phenyl]-1,2-oxazol-5-yl]methylsulfanyl]phenoxy]acetic acid Chemical compound C1=C(OCC(O)=O)C(C)=CC(SCC=2ON=C(C=2)C=2C=CC(=CC=2)C(F)(F)F)=C1 GDYUMLCDKLRQLB-UHFFFAOYSA-N 0.000 description 4
- VVNHBVDGVFFCFB-UHFFFAOYSA-N 2-[2-methyl-4-[[4-(pyridin-2-ylmethoxy)phenyl]methylsulfanyl]phenoxy]acetic acid Chemical compound C1=C(OCC(O)=O)C(C)=CC(SCC=2C=CC(OCC=3N=CC=CC=3)=CC=2)=C1 VVNHBVDGVFFCFB-UHFFFAOYSA-N 0.000 description 4
- SWZYTFVXAKGIHZ-UHFFFAOYSA-N 2-[2-methyl-4-[[4-[4-(trifluoromethyl)phenyl]phenyl]methylsulfanyl]phenoxy]acetic acid Chemical compound C1=C(OCC(O)=O)C(C)=CC(SCC=2C=CC(=CC=2)C=2C=CC(=CC=2)C(F)(F)F)=C1 SWZYTFVXAKGIHZ-UHFFFAOYSA-N 0.000 description 4
- YJRSXDGYNINSJU-UHFFFAOYSA-N 2-[2-methyl-4-[[4-[[4-(trifluoromethyl)phenyl]methoxy]phenyl]methylsulfanyl]phenoxy]acetic acid Chemical compound C1=C(OCC(O)=O)C(C)=CC(SCC=2C=CC(OCC=3C=CC(=CC=3)C(F)(F)F)=CC=2)=C1 YJRSXDGYNINSJU-UHFFFAOYSA-N 0.000 description 4
- DHTATVFNFJNQCP-UHFFFAOYSA-N 2-[4-[4-(trifluoromethyl)phenyl]phenyl]acetic acid Chemical compound C1=CC(CC(=O)O)=CC=C1C1=CC=C(C(F)(F)F)C=C1 DHTATVFNFJNQCP-UHFFFAOYSA-N 0.000 description 4
- WQYKYZFRSHNBOR-UHFFFAOYSA-N 2-[4-[4-(trifluoromethyl)phenyl]phenyl]ethanol Chemical compound C1=CC(CCO)=CC=C1C1=CC=C(C(F)(F)F)C=C1 WQYKYZFRSHNBOR-UHFFFAOYSA-N 0.000 description 4
- NPBOMWDCBOOOPH-UHFFFAOYSA-N 2-[4-[[4-[(4-chlorophenyl)methoxy]phenyl]methylsulfanyl]-2-methylphenoxy]acetic acid Chemical compound C1=C(OCC(O)=O)C(C)=CC(SCC=2C=CC(OCC=3C=CC(Cl)=CC=3)=CC=2)=C1 NPBOMWDCBOOOPH-UHFFFAOYSA-N 0.000 description 4
- JDPZWQNXCARVHW-UHFFFAOYSA-N 2-[5-chloro-2-methyl-4-[[4-[5-(trifluoromethyl)pyridin-2-yl]phenyl]methylsulfanyl]phenoxy]acetic acid Chemical compound C1=C(OCC(O)=O)C(C)=CC(SCC=2C=CC(=CC=2)C=2N=CC(=CC=2)C(F)(F)F)=C1Cl JDPZWQNXCARVHW-UHFFFAOYSA-N 0.000 description 4
- XXXMKMSHRBZEPM-UHFFFAOYSA-N 2-[5-chloro-2-methyl-4-[[4-[[4-(trifluoromethyl)phenyl]methoxy]phenyl]methylsulfanyl]phenoxy]acetic acid Chemical compound C1=C(OCC(O)=O)C(C)=CC(SCC=2C=CC(OCC=3C=CC(=CC=3)C(F)(F)F)=CC=2)=C1Cl XXXMKMSHRBZEPM-UHFFFAOYSA-N 0.000 description 4
- VSBJGHHHVIQUBJ-UHFFFAOYSA-N 2-[5-chloro-2-methyl-4-[[5-[4-(trifluoromethyl)phenyl]-1,2-oxazol-3-yl]methylsulfanyl]phenoxy]acetic acid Chemical compound C1=C(OCC(O)=O)C(C)=CC(SCC2=NOC(=C2)C=2C=CC(=CC=2)C(F)(F)F)=C1Cl VSBJGHHHVIQUBJ-UHFFFAOYSA-N 0.000 description 4
- KQUOMDCXEGZVBD-UHFFFAOYSA-N 2-[5-methoxy-2-methyl-4-[2-[1-[[4-(trifluoromethyl)phenyl]methyl]indol-3-yl]ethylsulfanyl]phenoxy]acetic acid Chemical compound COC1=CC(OCC(O)=O)=C(C)C=C1SCCC(C1=CC=CC=C11)=CN1CC1=CC=C(C(F)(F)F)C=C1 KQUOMDCXEGZVBD-UHFFFAOYSA-N 0.000 description 4
- HGNIFAAHWIYHSC-UHFFFAOYSA-N 2-[5-methoxy-2-methyl-4-[[4-(2,4,6-trimethylphenyl)phenyl]methylsulfanyl]phenoxy]acetic acid Chemical compound COC1=CC(OCC(O)=O)=C(C)C=C1SCC1=CC=C(C=2C(=CC(C)=CC=2C)C)C=C1 HGNIFAAHWIYHSC-UHFFFAOYSA-N 0.000 description 4
- HYDRDNRHZSRELG-UHFFFAOYSA-N 2-[5-methoxy-2-methyl-4-[[4-[3-(trifluoromethoxy)phenyl]phenyl]methylsulfanyl]phenoxy]acetic acid Chemical compound COC1=CC(OCC(O)=O)=C(C)C=C1SCC1=CC=C(C=2C=C(OC(F)(F)F)C=CC=2)C=C1 HYDRDNRHZSRELG-UHFFFAOYSA-N 0.000 description 4
- RGUZPZUTEDKSJP-UHFFFAOYSA-N 2-[5-methoxy-2-methyl-4-[[4-[4-(trifluoromethyl)phenyl]phenyl]methylsulfinyl]phenoxy]acetic acid Chemical compound COC1=CC(OCC(O)=O)=C(C)C=C1S(=O)CC1=CC=C(C=2C=CC(=CC=2)C(F)(F)F)C=C1 RGUZPZUTEDKSJP-UHFFFAOYSA-N 0.000 description 4
- WWPCHJSYGOWGES-UHFFFAOYSA-N 2-[5-methoxy-2-methyl-4-[[4-[4-(trifluoromethyl)phenyl]phenyl]methylsulfonyl]phenoxy]acetic acid Chemical compound COC1=CC(OCC(O)=O)=C(C)C=C1S(=O)(=O)CC1=CC=C(C=2C=CC(=CC=2)C(F)(F)F)C=C1 WWPCHJSYGOWGES-UHFFFAOYSA-N 0.000 description 4
- WIOTZGHDAYUCBC-UHFFFAOYSA-N 2-[5-methoxy-2-methyl-4-[[5-[4-(trifluoromethyl)phenyl]-1,2-oxazol-3-yl]methylsulfanyl]phenoxy]acetic acid Chemical compound COC1=CC(OCC(O)=O)=C(C)C=C1SCC1=NOC(C=2C=CC(=CC=2)C(F)(F)F)=C1 WIOTZGHDAYUCBC-UHFFFAOYSA-N 0.000 description 4
- DMZAKAJFNOKFPW-UHFFFAOYSA-N 2-[[5-methyl-7-[[4-[5-(trifluoromethyl)pyridin-2-yl]phenyl]methylsulfanyl]-2,3-dihydro-1-benzofuran-4-yl]oxy]acetic acid Chemical compound C=12OCCC2=C(OCC(O)=O)C(C)=CC=1SCC(C=C1)=CC=C1C1=CC=C(C(F)(F)F)C=N1 DMZAKAJFNOKFPW-UHFFFAOYSA-N 0.000 description 4
- FFFODHOLTATZFE-UHFFFAOYSA-N 2-[[6-methyl-8-[[4-[5-(trifluoromethyl)pyridin-2-yl]phenyl]methylsulfanyl]-3,4-dihydro-2h-chromen-5-yl]oxy]acetic acid Chemical compound C=12OCCCC2=C(OCC(O)=O)C(C)=CC=1SCC(C=C1)=CC=C1C1=CC=C(C(F)(F)F)C=N1 FFFODHOLTATZFE-UHFFFAOYSA-N 0.000 description 4
- XWYVNCGVZFRLFM-UHFFFAOYSA-N 2-[[7-[(4-phenylmethoxyphenyl)methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetic acid Chemical compound C1=2CCCC=2C(OCC(=O)O)=CC=C1SCC(C=C1)=CC=C1OCC1=CC=CC=C1 XWYVNCGVZFRLFM-UHFFFAOYSA-N 0.000 description 4
- HZCJEJYMIUUIGS-UHFFFAOYSA-N 2-[[7-[[2-[4-(trifluoromethyl)phenyl]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetic acid Chemical compound C1=2CCCC=2C(OCC(=O)O)=CC=C1SCC1=CC=CC=C1C1=CC=C(C(F)(F)F)C=C1 HZCJEJYMIUUIGS-UHFFFAOYSA-N 0.000 description 4
- JHOBDCQJTGVGCD-UHFFFAOYSA-N 2-[[7-[[3-(2,6-dichlorophenyl)-5-methyl-1,2-oxazol-4-yl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetic acid Chemical compound C=1C=C(OCC(O)=O)C=2CCCC=2C=1SCC1=C(C)ON=C1C1=C(Cl)C=CC=C1Cl JHOBDCQJTGVGCD-UHFFFAOYSA-N 0.000 description 4
- JLHZODXFXKFFSE-UHFFFAOYSA-N 2-[[7-[[3-[4-(trifluoromethyl)phenyl]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetic acid Chemical compound C1=2CCCC=2C(OCC(=O)O)=CC=C1SCC(C=1)=CC=CC=1C1=CC=C(C(F)(F)F)C=C1 JLHZODXFXKFFSE-UHFFFAOYSA-N 0.000 description 4
- GJWYGXOFRHJFDI-UHFFFAOYSA-N 2-[[7-[[4-[(2,4-dichlorophenyl)methoxy]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetic acid Chemical compound C1=2CCCC=2C(OCC(=O)O)=CC=C1SCC(C=C1)=CC=C1OCC1=CC=C(Cl)C=C1Cl GJWYGXOFRHJFDI-UHFFFAOYSA-N 0.000 description 4
- QBFOOBAVYCAUPJ-UHFFFAOYSA-N 2-[[7-[[4-[(2,4-difluorophenyl)methoxy]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetic acid Chemical compound C1=2CCCC=2C(OCC(=O)O)=CC=C1SCC(C=C1)=CC=C1OCC1=CC=C(F)C=C1F QBFOOBAVYCAUPJ-UHFFFAOYSA-N 0.000 description 4
- JPCMVIVXKCMDJB-UHFFFAOYSA-N 2-[[7-[[4-[(2,5-dichlorophenyl)methoxy]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetic acid Chemical compound C1=2CCCC=2C(OCC(=O)O)=CC=C1SCC(C=C1)=CC=C1OCC1=CC(Cl)=CC=C1Cl JPCMVIVXKCMDJB-UHFFFAOYSA-N 0.000 description 4
- MBUJLLWNJDYQII-UHFFFAOYSA-N 2-[[7-[[4-[(3,4-dichlorophenyl)methoxy]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetic acid Chemical compound C1=2CCCC=2C(OCC(=O)O)=CC=C1SCC(C=C1)=CC=C1OCC1=CC=C(Cl)C(Cl)=C1 MBUJLLWNJDYQII-UHFFFAOYSA-N 0.000 description 4
- OXWPTDGCOBSUPD-UHFFFAOYSA-N 2-[[7-[[4-[(3,5-dichlorophenyl)methoxy]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetic acid Chemical compound C1=2CCCC=2C(OCC(=O)O)=CC=C1SCC(C=C1)=CC=C1OCC1=CC(Cl)=CC(Cl)=C1 OXWPTDGCOBSUPD-UHFFFAOYSA-N 0.000 description 4
- PUTRDWOXGQZTNK-UHFFFAOYSA-N 2-[[7-[[4-[(4-chlorophenyl)methoxy]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetic acid Chemical compound C1=2CCCC=2C(OCC(=O)O)=CC=C1SCC(C=C1)=CC=C1OCC1=CC=C(Cl)C=C1 PUTRDWOXGQZTNK-UHFFFAOYSA-N 0.000 description 4
- BONGLINICVKANL-UHFFFAOYSA-N 2-[[7-[[4-[(4-methoxyphenyl)methoxy]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetic acid Chemical compound C1=CC(OC)=CC=C1COC(C=C1)=CC=C1CSC1=CC=C(OCC(O)=O)C2=C1CCC2 BONGLINICVKANL-UHFFFAOYSA-N 0.000 description 4
- UUBMLUGVHDOXPD-UHFFFAOYSA-N 2-[[7-[[4-[4-(trifluoromethyl)phenyl]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetic acid Chemical compound C1=2CCCC=2C(OCC(=O)O)=CC=C1SCC(C=C1)=CC=C1C1=CC=C(C(F)(F)F)C=C1 UUBMLUGVHDOXPD-UHFFFAOYSA-N 0.000 description 4
- KZFZYOOORMPMPV-UHFFFAOYSA-N 2-[[7-[[4-[[4-(trifluoromethyl)phenyl]methyl]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetic acid Chemical compound C1=2CCCC=2C(OCC(=O)O)=CC=C1SCC(C=C1)=CC=C1CC1=CC=C(C(F)(F)F)C=C1 KZFZYOOORMPMPV-UHFFFAOYSA-N 0.000 description 4
- BAFBGCJOZQQZNG-UHFFFAOYSA-N 2-[[7-[[4-[[4-chloro-3-(trifluoromethyl)phenyl]methoxy]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetic acid Chemical compound C1=2CCCC=2C(OCC(=O)O)=CC=C1SCC(C=C1)=CC=C1OCC1=CC=C(Cl)C(C(F)(F)F)=C1 BAFBGCJOZQQZNG-UHFFFAOYSA-N 0.000 description 4
- MNJGBPCXRHBVRS-UHFFFAOYSA-N 2-[[7-[[4-[[4-fluoro-3-(trifluoromethyl)phenyl]methoxy]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetic acid Chemical compound C1=2CCCC=2C(OCC(=O)O)=CC=C1SCC(C=C1)=CC=C1OCC1=CC=C(F)C(C(F)(F)F)=C1 MNJGBPCXRHBVRS-UHFFFAOYSA-N 0.000 description 4
- VDMRRVAEUIADBQ-UHFFFAOYSA-N 2-[[7-[[5-(2-chlorophenyl)-1,2-oxazol-3-yl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetic acid Chemical compound C1=2CCCC=2C(OCC(=O)O)=CC=C1SCC(=NO1)C=C1C1=CC=CC=C1Cl VDMRRVAEUIADBQ-UHFFFAOYSA-N 0.000 description 4
- GQCXEZCBUPCZEW-UHFFFAOYSA-N 2-[[7-[[5-[4-(trifluoromethyl)phenyl]-1,2-oxazol-3-yl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetic acid Chemical compound C1=2CCCC=2C(OCC(=O)O)=CC=C1SCC(=NO1)C=C1C1=CC=C(C(F)(F)F)C=C1 GQCXEZCBUPCZEW-UHFFFAOYSA-N 0.000 description 4
- HEZNHVQMOJSFDE-UHFFFAOYSA-N 2-[[8-[[4-[5-(trifluoromethyl)pyridin-2-yl]phenyl]methylsulfanyl]-3,4-dihydro-2h-chromen-5-yl]oxy]acetic acid Chemical compound C1=2OCCCC=2C(OCC(=O)O)=CC=C1SCC(C=C1)=CC=C1C1=CC=C(C(F)(F)F)C=N1 HEZNHVQMOJSFDE-UHFFFAOYSA-N 0.000 description 4
- CUOCCHDGPRTEAX-UHFFFAOYSA-N 2-[[8-[[4-[[4-(trifluoromethyl)phenyl]methoxy]phenyl]methylsulfanyl]-3,4-dihydro-2h-chromen-5-yl]oxy]acetic acid Chemical compound C1=2OCCCC=2C(OCC(=O)O)=CC=C1SCC(C=C1)=CC=C1OCC1=CC=C(C(F)(F)F)C=C1 CUOCCHDGPRTEAX-UHFFFAOYSA-N 0.000 description 4
- FOPHIEABULMTDJ-UHFFFAOYSA-N 3-(chloromethyl)-5-[4-(trifluoromethyl)phenyl]-1,2-oxazole Chemical compound C1=CC(C(F)(F)F)=CC=C1C1=CC(CCl)=NO1 FOPHIEABULMTDJ-UHFFFAOYSA-N 0.000 description 4
- QXIOWYFFTWXSHE-UHFFFAOYSA-N 5-methoxy-2-methylphenol Chemical compound COC1=CC=C(C)C(O)=C1 QXIOWYFFTWXSHE-UHFFFAOYSA-N 0.000 description 4
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 4
- 241000282414 Homo sapiens Species 0.000 description 4
- 206010022489 Insulin Resistance Diseases 0.000 description 4
- 108010007622 LDL Lipoproteins Proteins 0.000 description 4
- 102000007330 LDL Lipoproteins Human genes 0.000 description 4
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 4
- PCLIMKBDDGJMGD-UHFFFAOYSA-N N-bromosuccinimide Chemical compound BrN1C(=O)CCC1=O PCLIMKBDDGJMGD-UHFFFAOYSA-N 0.000 description 4
- 102000023984 PPAR alpha Human genes 0.000 description 4
- 108010016731 PPAR gamma Proteins 0.000 description 4
- 102000000536 PPAR gamma Human genes 0.000 description 4
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 4
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 4
- OGGXHWFEGSDQCG-UHFFFAOYSA-N [2-methyl-4-[4-(trifluoromethyl)phenyl]phenyl]methanol Chemical compound C1=C(CO)C(C)=CC(C=2C=CC(=CC=2)C(F)(F)F)=C1 OGGXHWFEGSDQCG-UHFFFAOYSA-N 0.000 description 4
- ZABYFMPEEYJQFT-UHFFFAOYSA-N [4-[[4-(trifluoromethyl)phenyl]methyl]phenyl]methanol Chemical compound C1=CC(CO)=CC=C1CC1=CC=C(C(F)(F)F)C=C1 ZABYFMPEEYJQFT-UHFFFAOYSA-N 0.000 description 4
- 239000002253 acid Substances 0.000 description 4
- 125000002947 alkylene group Chemical group 0.000 description 4
- 230000003178 anti-diabetic effect Effects 0.000 description 4
- 239000002585 base Substances 0.000 description 4
- 239000008280 blood Substances 0.000 description 4
- 210000004369 blood Anatomy 0.000 description 4
- 235000012000 cholesterol Nutrition 0.000 description 4
- ZUOUZKKEUPVFJK-UHFFFAOYSA-N diphenyl Chemical compound C1=CC=CC=C1C1=CC=CC=C1 ZUOUZKKEUPVFJK-UHFFFAOYSA-N 0.000 description 4
- 229940079593 drug Drugs 0.000 description 4
- 238000001914 filtration Methods 0.000 description 4
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 description 4
- VKRULNTYXHYAAH-UHFFFAOYSA-N methyl 2-(2,3-dihydro-1h-inden-4-yloxy)acetate Chemical compound COC(=O)COC1=CC=CC2=C1CCC2 VKRULNTYXHYAAH-UHFFFAOYSA-N 0.000 description 4
- LVIWZUAGRHUNED-UHFFFAOYSA-N methyl 2-(2,5-dimethyl-4-sulfanylphenoxy)acetate Chemical compound COC(=O)COC1=CC(C)=C(S)C=C1C LVIWZUAGRHUNED-UHFFFAOYSA-N 0.000 description 4
- FWHNDYVYVYLGPZ-UHFFFAOYSA-N methyl 2-(4-bromo-2-methylphenoxy)acetate Chemical compound COC(=O)COC1=CC=C(Br)C=C1C FWHNDYVYVYLGPZ-UHFFFAOYSA-N 0.000 description 4
- OLWMUEMPOVOVLB-UHFFFAOYSA-N methyl 2-(4-sulfanylphenoxy)acetate Chemical compound COC(=O)COC1=CC=C(S)C=C1 OLWMUEMPOVOVLB-UHFFFAOYSA-N 0.000 description 4
- SYQAXUAVRVDBBM-UHFFFAOYSA-N methyl 2-[(7-sulfanyl-2,3-dihydro-1h-inden-4-yl)oxy]acetate Chemical compound COC(=O)COC1=CC=C(S)C2=C1CCC2 SYQAXUAVRVDBBM-UHFFFAOYSA-N 0.000 description 4
- MMILWMPCFWFEOQ-UHFFFAOYSA-N methyl 2-[2-methyl-4-[(4-phenoxyphenyl)methylsulfanyl]phenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC=C1SCC(C=C1)=CC=C1OC1=CC=CC=C1 MMILWMPCFWFEOQ-UHFFFAOYSA-N 0.000 description 4
- PFHDYWIQGBGKEH-UHFFFAOYSA-N methyl 2-[2-methyl-4-[[4-(pyridin-2-ylmethoxy)phenyl]methylsulfanyl]phenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC=C1SCC(C=C1)=CC=C1OCC1=CC=CC=N1 PFHDYWIQGBGKEH-UHFFFAOYSA-N 0.000 description 4
- IVFRWIRSTLZDTE-UHFFFAOYSA-N methyl 2-[4-[(4-hydroxyphenyl)methylsulfanyl]-2-methylphenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC=C1SCC1=CC=C(O)C=C1 IVFRWIRSTLZDTE-UHFFFAOYSA-N 0.000 description 4
- YGWBGEGHCUNZNY-UHFFFAOYSA-N methyl 2-[4-[(6-chloropyridin-3-yl)methylsulfanyl]-5-methoxy-2-methylphenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC(OC)=C1SCC1=CC=C(Cl)N=C1 YGWBGEGHCUNZNY-UHFFFAOYSA-N 0.000 description 4
- QTRYEZHSTLMULI-UHFFFAOYSA-N methyl 2-[4-[2-(1h-indol-3-yl)ethylsulfanyl]-5-methoxy-2-methylphenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC(OC)=C1SCCC1=CNC2=CC=CC=C12 QTRYEZHSTLMULI-UHFFFAOYSA-N 0.000 description 4
- GNKBDWJKONZPLJ-UHFFFAOYSA-N methyl 2-[4-[[4-[(2,4-difluorophenyl)methoxy]phenyl]methylsulfanyl]-2-methylphenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC=C1SCC(C=C1)=CC=C1OCC1=CC=C(F)C=C1F GNKBDWJKONZPLJ-UHFFFAOYSA-N 0.000 description 4
- OVEHNMBXESCZOA-UHFFFAOYSA-N methyl 2-[5-chloro-2-methyl-4-[[5-[4-(trifluoromethyl)phenyl]-1,2-oxazol-3-yl]methylsulfanyl]phenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC(Cl)=C1SCC1=NOC(C=2C=CC(=CC=2)C(F)(F)F)=C1 OVEHNMBXESCZOA-UHFFFAOYSA-N 0.000 description 4
- DANQLTDPRWASKS-UHFFFAOYSA-N methyl 2-[5-methoxy-2-methyl-4-[2-[1-[[4-(trifluoromethyl)phenyl]methyl]indol-3-yl]ethylsulfanyl]phenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC(OC)=C1SCCC(C1=CC=CC=C11)=CN1CC1=CC=C(C(F)(F)F)C=C1 DANQLTDPRWASKS-UHFFFAOYSA-N 0.000 description 4
- UQPLNGGMMNNHLV-UHFFFAOYSA-N methyl 2-[5-methoxy-2-methyl-4-[[4-[4-(trifluoromethyl)phenyl]phenyl]methylsulfanyl]phenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC(OC)=C1SCC1=CC=C(C=2C=CC(=CC=2)C(F)(F)F)C=C1 UQPLNGGMMNNHLV-UHFFFAOYSA-N 0.000 description 4
- XQUNYNAQUGNOCA-UHFFFAOYSA-N methyl 2-[5-methoxy-2-methyl-4-[[6-[4-(trifluoromethyl)phenyl]pyridin-3-yl]methylsulfanyl]phenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC(OC)=C1SCC1=CC=C(C=2C=CC(=CC=2)C(F)(F)F)N=C1 XQUNYNAQUGNOCA-UHFFFAOYSA-N 0.000 description 4
- PLOOJVWUILFSQJ-UHFFFAOYSA-N methyl 2-[[6-methyl-8-[[4-[5-(trifluoromethyl)pyridin-2-yl]phenyl]methylsulfanyl]-3,4-dihydro-2h-chromen-5-yl]oxy]acetate Chemical compound C1=2OCCCC=2C(OCC(=O)OC)=C(C)C=C1SCC(C=C1)=CC=C1C1=CC=C(C(F)(F)F)C=N1 PLOOJVWUILFSQJ-UHFFFAOYSA-N 0.000 description 4
- GESPOCSITBXWAX-UHFFFAOYSA-N methyl 2-[[6-methyl-8-[[4-[[4-(trifluoromethyl)phenyl]methoxy]phenyl]methylsulfanyl]-3,4-dihydro-2h-chromen-5-yl]oxy]acetate Chemical compound C1=2OCCCC=2C(OCC(=O)OC)=C(C)C=C1SCC(C=C1)=CC=C1OCC1=CC=C(C(F)(F)F)C=C1 GESPOCSITBXWAX-UHFFFAOYSA-N 0.000 description 4
- SVCVLGXPQXRIBA-UHFFFAOYSA-N methyl 2-[[7-[(4-hydroxyphenyl)methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetate Chemical compound C1=2CCCC=2C(OCC(=O)OC)=CC=C1SCC1=CC=C(O)C=C1 SVCVLGXPQXRIBA-UHFFFAOYSA-N 0.000 description 4
- FUMPMIXMNOCGPH-UHFFFAOYSA-N methyl 2-[[7-[2-[4-[4-(trifluoromethyl)phenyl]phenyl]ethyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetate Chemical compound C1=2CCCC=2C(OCC(=O)OC)=CC=C1CCC(C=C1)=CC=C1C1=CC=C(C(F)(F)F)C=C1 FUMPMIXMNOCGPH-UHFFFAOYSA-N 0.000 description 4
- XJIZAXJBHXOMHB-UHFFFAOYSA-N methyl 2-[[7-[[3-[4-(trifluoromethyl)phenyl]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetate Chemical compound C1=2CCCC=2C(OCC(=O)OC)=CC=C1SCC(C=1)=CC=CC=1C1=CC=C(C(F)(F)F)C=C1 XJIZAXJBHXOMHB-UHFFFAOYSA-N 0.000 description 4
- SKEGBMRMIKRRDO-UHFFFAOYSA-N methyl 2-[[7-[[3-[5-(trifluoromethyl)pyridin-2-yl]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetate Chemical compound C1=2CCCC=2C(OCC(=O)OC)=CC=C1SCC(C=1)=CC=CC=1C1=CC=C(C(F)(F)F)C=N1 SKEGBMRMIKRRDO-UHFFFAOYSA-N 0.000 description 4
- COEWJBSTGGPQJI-UHFFFAOYSA-N methyl 2-[[7-[[3-[[4-(trifluoromethyl)phenyl]methoxy]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetate Chemical compound C1=2CCCC=2C(OCC(=O)OC)=CC=C1SCC(C=1)=CC=CC=1OCC1=CC=C(C(F)(F)F)C=C1 COEWJBSTGGPQJI-UHFFFAOYSA-N 0.000 description 4
- HYWJIAOVLDSXNR-UHFFFAOYSA-N methyl 2-[[7-[[4-[(2,5-dichlorophenyl)methoxy]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetate Chemical compound C1=2CCCC=2C(OCC(=O)OC)=CC=C1SCC(C=C1)=CC=C1OCC1=CC(Cl)=CC=C1Cl HYWJIAOVLDSXNR-UHFFFAOYSA-N 0.000 description 4
- 125000002971 oxazolyl group Chemical group 0.000 description 4
- 230000002265 prevention Effects 0.000 description 4
- 102000005962 receptors Human genes 0.000 description 4
- 108020003175 receptors Proteins 0.000 description 4
- 238000001953 recrystallisation Methods 0.000 description 4
- 238000007127 saponification reaction Methods 0.000 description 4
- VGTPCRGMBIAPIM-UHFFFAOYSA-M sodium thiocyanate Chemical compound [Na+].[S-]C#N VGTPCRGMBIAPIM-UHFFFAOYSA-M 0.000 description 4
- 241000894007 species Species 0.000 description 4
- 239000003826 tablet Substances 0.000 description 4
- VZGDMQKNWNREIO-UHFFFAOYSA-N tetrachloromethane Chemical compound ClC(Cl)(Cl)Cl VZGDMQKNWNREIO-UHFFFAOYSA-N 0.000 description 4
- 150000003626 triacylglycerols Chemical class 0.000 description 4
- 239000003981 vehicle Substances 0.000 description 4
- UDQTXCHQKHIQMH-KYGLGHNPSA-N (3ar,5s,6s,7r,7ar)-5-(difluoromethyl)-2-(ethylamino)-5,6,7,7a-tetrahydro-3ah-pyrano[3,2-d][1,3]thiazole-6,7-diol Chemical compound S1C(NCC)=N[C@H]2[C@@H]1O[C@H](C(F)F)[C@@H](O)[C@@H]2O UDQTXCHQKHIQMH-KYGLGHNPSA-N 0.000 description 3
- VDLUKNLXXMUPED-UHFFFAOYSA-N 1-(chloromethyl)-2-[[4-(trifluoromethyl)phenyl]methoxy]benzene Chemical compound C1=CC(C(F)(F)F)=CC=C1COC1=CC=CC=C1CCl VDLUKNLXXMUPED-UHFFFAOYSA-N 0.000 description 3
- IWDQCCZCBOLJCA-UHFFFAOYSA-N 1-(chloromethyl)-3-[[4-(trifluoromethyl)phenyl]methoxy]benzene Chemical compound C1=CC(C(F)(F)F)=CC=C1COC1=CC=CC(CCl)=C1 IWDQCCZCBOLJCA-UHFFFAOYSA-N 0.000 description 3
- AHEFTHIHJDXTRP-UHFFFAOYSA-N 1-(chloromethyl)-4-[2-(3-fluorophenyl)ethenyl]benzene Chemical compound FC1=CC=CC(C=CC=2C=CC(CCl)=CC=2)=C1 AHEFTHIHJDXTRP-UHFFFAOYSA-N 0.000 description 3
- VIAOSIIYSYBGHB-UHFFFAOYSA-N 1-(chloromethyl)-4-[[4-(trifluoromethyl)phenyl]methoxy]benzene Chemical compound C1=CC(C(F)(F)F)=CC=C1COC1=CC=C(CCl)C=C1 VIAOSIIYSYBGHB-UHFFFAOYSA-N 0.000 description 3
- REZVHINADSDRGA-UHFFFAOYSA-N 1-(chloromethyl)-4-[[4-(trifluoromethyl)phenyl]methyl]benzene Chemical compound C1=CC(C(F)(F)F)=CC=C1CC1=CC=C(CCl)C=C1 REZVHINADSDRGA-UHFFFAOYSA-N 0.000 description 3
- ZUIDIHDHRZFIIH-UHFFFAOYSA-N 1-[[4-(chloromethyl)phenoxy]methyl]-2,4-difluorobenzene Chemical compound FC1=CC(F)=CC=C1COC1=CC=C(CCl)C=C1 ZUIDIHDHRZFIIH-UHFFFAOYSA-N 0.000 description 3
- JJGADPYYRCAHGE-UHFFFAOYSA-N 1-chloro-4-[[3-(chloromethyl)phenoxy]methyl]-2-(trifluoromethyl)benzene Chemical compound C1=C(Cl)C(C(F)(F)F)=CC(COC=2C=C(CCl)C=CC=2)=C1 JJGADPYYRCAHGE-UHFFFAOYSA-N 0.000 description 3
- DMXWPVUSYFWZCG-UHFFFAOYSA-N 1-ethenyl-4-[4-(trifluoromethyl)phenyl]benzene Chemical group C1=CC(C(F)(F)F)=CC=C1C1=CC=C(C=C)C=C1 DMXWPVUSYFWZCG-UHFFFAOYSA-N 0.000 description 3
- NHLSXXHLDJYGKB-UHFFFAOYSA-N 2,4-dichloro-1-[[4-(chloromethyl)phenoxy]methyl]benzene Chemical compound C1=CC(CCl)=CC=C1OCC1=CC=C(Cl)C=C1Cl NHLSXXHLDJYGKB-UHFFFAOYSA-N 0.000 description 3
- ZRIDXJOGXXKCEK-UHFFFAOYSA-N 2-(chloromethyl)-9h-fluorene Chemical compound C1=CC=C2C3=CC=C(CCl)C=C3CC2=C1 ZRIDXJOGXXKCEK-UHFFFAOYSA-N 0.000 description 3
- KHSSLZMQGWDTAC-UHFFFAOYSA-N 2-[2-methyl-4-[(4-phenylphenyl)methylsulfanyl]phenoxy]acetic acid Chemical compound C1=C(OCC(O)=O)C(C)=CC(SCC=2C=CC(=CC=2)C=2C=CC=CC=2)=C1 KHSSLZMQGWDTAC-UHFFFAOYSA-N 0.000 description 3
- NBWSCGOUANWCMQ-UHFFFAOYSA-N 2-[2-methyl-4-[2-[4-[4-(trifluoromethyl)phenyl]phenyl]ethyl]phenoxy]acetic acid Chemical compound C1=C(OCC(O)=O)C(C)=CC(CCC=2C=CC(=CC=2)C=2C=CC(=CC=2)C(F)(F)F)=C1 NBWSCGOUANWCMQ-UHFFFAOYSA-N 0.000 description 3
- REOFKAGPIGDCLW-UHFFFAOYSA-N 2-[3-(chloromethyl)phenyl]-5-(trifluoromethyl)pyridine Chemical compound N1=CC(C(F)(F)F)=CC=C1C1=CC=CC(CCl)=C1 REOFKAGPIGDCLW-UHFFFAOYSA-N 0.000 description 3
- IMSHZHYMHGKDON-UHFFFAOYSA-N 2-[4-[4-(trifluoromethyl)phenyl]phenyl]acetyl chloride Chemical compound C1=CC(C(F)(F)F)=CC=C1C1=CC=C(CC(Cl)=O)C=C1 IMSHZHYMHGKDON-UHFFFAOYSA-N 0.000 description 3
- ZXRHBDVPDIVQQY-UHFFFAOYSA-N 2-[4-[[4-[(4-fluorophenyl)methoxy]phenyl]methylsulfanyl]-2-methylphenoxy]acetic acid Chemical compound C1=C(OCC(O)=O)C(C)=CC(SCC=2C=CC(OCC=3C=CC(F)=CC=3)=CC=2)=C1 ZXRHBDVPDIVQQY-UHFFFAOYSA-N 0.000 description 3
- RWRTYMGJKGDYLQ-UHFFFAOYSA-N 2-[[5-methyl-7-[[4-[[4-(trifluoromethyl)phenyl]methoxy]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetic acid Chemical compound C=12CCCC2=C(OCC(O)=O)C(C)=CC=1SCC(C=C1)=CC=C1OCC1=CC=C(C(F)(F)F)C=C1 RWRTYMGJKGDYLQ-UHFFFAOYSA-N 0.000 description 3
- APVMHDWTBQWFCS-UHFFFAOYSA-N 2-[[7-[(4-hydroxyphenyl)methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetic acid Chemical compound C1=2CCCC=2C(OCC(=O)O)=CC=C1SCC1=CC=C(O)C=C1 APVMHDWTBQWFCS-UHFFFAOYSA-N 0.000 description 3
- SNUCWTMFDGLTCI-UHFFFAOYSA-N 2-[[7-[[4-[(4-tert-butylphenyl)methoxy]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetic acid Chemical compound C1=CC(C(C)(C)C)=CC=C1COC(C=C1)=CC=C1CSC1=CC=C(OCC(O)=O)C2=C1CCC2 SNUCWTMFDGLTCI-UHFFFAOYSA-N 0.000 description 3
- CLWMMUCGSPUDJT-UHFFFAOYSA-N 2-[[7-[[4-[[4-(fluoromethyl)phenyl]methoxy]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetic acid Chemical compound C1=2CCCC=2C(OCC(=O)O)=CC=C1SCC(C=C1)=CC=C1OCC1=CC=C(CF)C=C1 CLWMMUCGSPUDJT-UHFFFAOYSA-N 0.000 description 3
- PIWIBUHURPMRHK-UHFFFAOYSA-N 2-[[7-[[4-[[4-fluoro-2-(trifluoromethyl)phenyl]methoxy]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetic acid Chemical compound C1=2CCCC=2C(OCC(=O)O)=CC=C1SCC(C=C1)=CC=C1OCC1=CC=C(F)C=C1C(F)(F)F PIWIBUHURPMRHK-UHFFFAOYSA-N 0.000 description 3
- HTFXWAOSQODIBI-UHFFFAOYSA-N 2-benzyl-1,3-dihydropyrrolo[3,4-c]pyridine Chemical compound C1C2=CC=NC=C2CN1CC1=CC=CC=C1 HTFXWAOSQODIBI-UHFFFAOYSA-N 0.000 description 3
- RDBHHUSDMIDPEB-UHFFFAOYSA-N 4-(chloromethyl)-1-methoxy-2-[[4-(trifluoromethyl)phenyl]methoxy]benzene Chemical compound COC1=CC=C(CCl)C=C1OCC1=CC=C(C(F)(F)F)C=C1 RDBHHUSDMIDPEB-UHFFFAOYSA-N 0.000 description 3
- SLKGLAFNLGTRGT-UHFFFAOYSA-N 5-(chloromethyl)-3-[4-(trifluoromethyl)phenyl]-1,2-oxazole Chemical compound C1=CC(C(F)(F)F)=CC=C1C1=NOC(CCl)=C1 SLKGLAFNLGTRGT-UHFFFAOYSA-N 0.000 description 3
- NVRRCUVMHFKHCL-UHFFFAOYSA-N 9h-fluoren-2-ylmethanol Chemical compound C1=CC=C2C3=CC=C(CO)C=C3CC2=C1 NVRRCUVMHFKHCL-UHFFFAOYSA-N 0.000 description 3
- 229920002134 Carboxymethyl cellulose Polymers 0.000 description 3
- 241000282412 Homo Species 0.000 description 3
- 108060001084 Luciferase Proteins 0.000 description 3
- 239000005089 Luciferase Substances 0.000 description 3
- 241000282567 Macaca fascicularis Species 0.000 description 3
- 238000003820 Medium-pressure liquid chromatography Methods 0.000 description 3
- 229910002651 NO3 Inorganic materials 0.000 description 3
- 229910002666 PdCl2 Inorganic materials 0.000 description 3
- 229920002472 Starch Polymers 0.000 description 3
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 3
- 108010062497 VLDL Lipoproteins Proteins 0.000 description 3
- GWVIKCOWFAWODY-UHFFFAOYSA-N [2-[[4-(trifluoromethyl)phenyl]methoxy]phenyl]methanol Chemical compound OCC1=CC=CC=C1OCC1=CC=C(C(F)(F)F)C=C1 GWVIKCOWFAWODY-UHFFFAOYSA-N 0.000 description 3
- HGTDLKXUWVKLQX-UHFFFAOYSA-N [3-(hydroxymethyl)phenyl]boronic acid Chemical compound OCC1=CC=CC(B(O)O)=C1 HGTDLKXUWVKLQX-UHFFFAOYSA-N 0.000 description 3
- ZFEKDTKGNJHRHJ-UHFFFAOYSA-N [3-[3-(trifluoromethoxy)phenyl]phenyl]methanol Chemical compound OCC1=CC=CC(C=2C=C(OC(F)(F)F)C=CC=2)=C1 ZFEKDTKGNJHRHJ-UHFFFAOYSA-N 0.000 description 3
- BKESULFFLXBNLC-UHFFFAOYSA-N [3-[4-(trifluoromethyl)phenyl]phenyl]methanol Chemical compound OCC1=CC=CC(C=2C=CC(=CC=2)C(F)(F)F)=C1 BKESULFFLXBNLC-UHFFFAOYSA-N 0.000 description 3
- BDBKOHBCYSFAGR-UHFFFAOYSA-N [3-[5-(trifluoromethyl)pyridin-2-yl]phenyl]methanol Chemical compound OCC1=CC=CC(C=2N=CC(=CC=2)C(F)(F)F)=C1 BDBKOHBCYSFAGR-UHFFFAOYSA-N 0.000 description 3
- YIHQOAAXSKRTMJ-UHFFFAOYSA-N [3-[[4-(trifluoromethyl)phenyl]methoxy]phenyl]methanol Chemical compound OCC1=CC=CC(OCC=2C=CC(=CC=2)C(F)(F)F)=C1 YIHQOAAXSKRTMJ-UHFFFAOYSA-N 0.000 description 3
- YGNWAAIMJMNODZ-UHFFFAOYSA-N [3-[[4-chloro-3-(trifluoromethyl)phenyl]methoxy]phenyl]methanol Chemical compound OCC1=CC=CC(OCC=2C=C(C(Cl)=CC=2)C(F)(F)F)=C1 YGNWAAIMJMNODZ-UHFFFAOYSA-N 0.000 description 3
- ZSWIVXLMMVNSCM-UHFFFAOYSA-N [4-(4-fluorophenyl)phenyl]methanol Chemical compound C1=CC(CO)=CC=C1C1=CC=C(F)C=C1 ZSWIVXLMMVNSCM-UHFFFAOYSA-N 0.000 description 3
- YIZSAETVNYHUOQ-UHFFFAOYSA-N [4-[(2,4-dichlorophenyl)methoxy]phenyl]methanol Chemical compound C1=CC(CO)=CC=C1OCC1=CC=C(Cl)C=C1Cl YIZSAETVNYHUOQ-UHFFFAOYSA-N 0.000 description 3
- HPUNAPIDVUGQIR-UHFFFAOYSA-N [4-[(2,4-difluorophenyl)methoxy]phenyl]methanol Chemical compound C1=CC(CO)=CC=C1OCC1=CC=C(F)C=C1F HPUNAPIDVUGQIR-UHFFFAOYSA-N 0.000 description 3
- FNWSEJASZBRBDB-UHFFFAOYSA-N [4-[(4-fluorophenyl)methoxy]phenyl]methanol Chemical compound C1=CC(CO)=CC=C1OCC1=CC=C(F)C=C1 FNWSEJASZBRBDB-UHFFFAOYSA-N 0.000 description 3
- CPNBUCKGQNVQFY-UHFFFAOYSA-N [4-[(4-methoxyphenyl)methoxy]phenyl]methanol Chemical compound C1=CC(OC)=CC=C1COC1=CC=C(CO)C=C1 CPNBUCKGQNVQFY-UHFFFAOYSA-N 0.000 description 3
- SFMRLDLUMUTYMH-UHFFFAOYSA-N [4-[(4-tert-butylphenyl)methoxy]phenyl]methanol Chemical compound C1=CC(C(C)(C)C)=CC=C1COC1=CC=C(CO)C=C1 SFMRLDLUMUTYMH-UHFFFAOYSA-N 0.000 description 3
- SUTCKBSZTXQCDR-UHFFFAOYSA-N [4-[4-chloro-3-(trifluoromethyl)phenyl]phenyl]methanol Chemical compound C1=CC(CO)=CC=C1C1=CC=C(Cl)C(C(F)(F)F)=C1 SUTCKBSZTXQCDR-UHFFFAOYSA-N 0.000 description 3
- VRVOASJNEFKGGI-UHFFFAOYSA-N [4-[5-(trifluoromethyl)pyridin-2-yl]phenyl]methanol Chemical compound C1=CC(CO)=CC=C1C1=CC=C(C(F)(F)F)C=N1 VRVOASJNEFKGGI-UHFFFAOYSA-N 0.000 description 3
- OKCVZMUDSAKIME-UHFFFAOYSA-N [4-methoxy-3-[[4-(trifluoromethyl)phenyl]methoxy]phenyl]methanol Chemical compound COC1=CC=C(CO)C=C1OCC1=CC=C(C(F)(F)F)C=C1 OKCVZMUDSAKIME-UHFFFAOYSA-N 0.000 description 3
- HTDMXATWYNKLLE-UHFFFAOYSA-N [5-[4-(trifluoromethyl)phenyl]-1,2-oxazol-3-yl]methanol Chemical compound O1N=C(CO)C=C1C1=CC=C(C(F)(F)F)C=C1 HTDMXATWYNKLLE-UHFFFAOYSA-N 0.000 description 3
- 230000002402 anti-lipaemic effect Effects 0.000 description 3
- 239000003472 antidiabetic agent Substances 0.000 description 3
- 150000001499 aryl bromides Chemical class 0.000 description 3
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 3
- UHOVQNZJYSORNB-UHFFFAOYSA-N benzene Substances C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 3
- WQZGKKKJIJFFOK-FPRJBGLDSA-N beta-D-galactose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@H]1O WQZGKKKJIJFFOK-FPRJBGLDSA-N 0.000 description 3
- 108010005774 beta-Galactosidase Proteins 0.000 description 3
- 125000002618 bicyclic heterocycle group Chemical group 0.000 description 3
- 239000002775 capsule Substances 0.000 description 3
- 235000010948 carboxy methyl cellulose Nutrition 0.000 description 3
- 239000000969 carrier Substances 0.000 description 3
- 108700010039 chimeric receptor Proteins 0.000 description 3
- 150000001805 chlorine compounds Chemical class 0.000 description 3
- 229940125936 compound 42 Drugs 0.000 description 3
- 150000004292 cyclic ethers Chemical class 0.000 description 3
- 235000014113 dietary fatty acids Nutrition 0.000 description 3
- 201000010099 disease Diseases 0.000 description 3
- 239000006185 dispersion Substances 0.000 description 3
- VHJLVAABSRFDPM-QWWZWVQMSA-N dithiothreitol Chemical compound SC[C@@H](O)[C@H](O)CS VHJLVAABSRFDPM-QWWZWVQMSA-N 0.000 description 3
- MKMLEMDGZCVWJB-UHFFFAOYSA-N ethyl 5-[4-(trifluoromethyl)phenyl]-1,2-oxazole-3-carboxylate Chemical compound O1N=C(C(=O)OCC)C=C1C1=CC=C(C(F)(F)F)C=C1 MKMLEMDGZCVWJB-UHFFFAOYSA-N 0.000 description 3
- PQVSTLUFSYVLTO-UHFFFAOYSA-N ethyl n-ethoxycarbonylcarbamate Chemical compound CCOC(=O)NC(=O)OCC PQVSTLUFSYVLTO-UHFFFAOYSA-N 0.000 description 3
- 239000000194 fatty acid Substances 0.000 description 3
- 229930195729 fatty acid Natural products 0.000 description 3
- 235000011187 glycerol Nutrition 0.000 description 3
- 210000000936 intestine Anatomy 0.000 description 3
- 229940040692 lithium hydroxide monohydrate Drugs 0.000 description 3
- GLXDVVHUTZTUQK-UHFFFAOYSA-M lithium hydroxide monohydrate Substances [Li+].O.[OH-] GLXDVVHUTZTUQK-UHFFFAOYSA-M 0.000 description 3
- 210000004185 liver Anatomy 0.000 description 3
- XHEWAVUQCYLXJT-UHFFFAOYSA-N methyl 2-(2,6-dimethyl-4-thiocyanatophenoxy)acetate Chemical compound COC(=O)COC1=C(C)C=C(SC#N)C=C1C XHEWAVUQCYLXJT-UHFFFAOYSA-N 0.000 description 3
- NBAUXWIBKMUMJV-UHFFFAOYSA-N methyl 2-(2-butyl-4-cyanophenyl)sulfanylacetate Chemical compound C(CCC)C1=C(SCC(=O)OC)C=CC(=C1)C#N NBAUXWIBKMUMJV-UHFFFAOYSA-N 0.000 description 3
- PJKNIMLOIPTHNE-UHFFFAOYSA-N methyl 2-(2-butyl-4-sulfanylphenoxy)acetate Chemical compound CCCCC1=CC(S)=CC=C1OCC(=O)OC PJKNIMLOIPTHNE-UHFFFAOYSA-N 0.000 description 3
- MSXJXHJYKWDCQD-UHFFFAOYSA-N methyl 2-(2-methyl-4-thiocyanatophenoxy)acetate Chemical compound COC(=O)COC1=CC=C(SC#N)C=C1C MSXJXHJYKWDCQD-UHFFFAOYSA-N 0.000 description 3
- UOCOKTZWSBVJPY-UHFFFAOYSA-N methyl 2-(3-methoxy-4-sulfanylphenoxy)acetate Chemical compound COC(=O)COC1=CC=C(S)C(OC)=C1 UOCOKTZWSBVJPY-UHFFFAOYSA-N 0.000 description 3
- WAPRRKJFRACMEK-UHFFFAOYSA-N methyl 2-(3-methoxy-4-thiocyanatophenoxy)acetate Chemical compound COC(=O)COC1=CC=C(SC#N)C(OC)=C1 WAPRRKJFRACMEK-UHFFFAOYSA-N 0.000 description 3
- NOKYLJSWVAASOJ-UHFFFAOYSA-N methyl 2-(5-chloro-2-methyl-4-sulfanylphenoxy)acetate Chemical compound COC(=O)COC1=CC(Cl)=C(S)C=C1C NOKYLJSWVAASOJ-UHFFFAOYSA-N 0.000 description 3
- NJVPEWHCLMBJGT-UHFFFAOYSA-N methyl 2-[(5-methyl-7-sulfanyl-2,3-dihydro-1h-inden-4-yl)oxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=C2CCCC2=C1S NJVPEWHCLMBJGT-UHFFFAOYSA-N 0.000 description 3
- GVZABZVGRKACTH-UHFFFAOYSA-N methyl 2-[2-butyl-4-[[4-[4-(trifluoromethyl)phenyl]phenyl]methylsulfanyl]phenoxy]acetate Chemical compound C1=C(OCC(=O)OC)C(CCCC)=CC(SCC=2C=CC(=CC=2)C=2C=CC(=CC=2)C(F)(F)F)=C1 GVZABZVGRKACTH-UHFFFAOYSA-N 0.000 description 3
- HFUIZAUOZMMGLA-UHFFFAOYSA-N methyl 2-[2-methyl-4-[(4-phenylmethoxyphenyl)methylsulfanyl]phenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC=C1SCC(C=C1)=CC=C1OCC1=CC=CC=C1 HFUIZAUOZMMGLA-UHFFFAOYSA-N 0.000 description 3
- RQXULGGDLSMXHZ-UHFFFAOYSA-N methyl 2-[2-methyl-4-[(4-phenylphenyl)methylsulfanyl]phenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC=C1SCC1=CC=C(C=2C=CC=CC=2)C=C1 RQXULGGDLSMXHZ-UHFFFAOYSA-N 0.000 description 3
- OTXMGZLEOJVEDX-UHFFFAOYSA-N methyl 2-[2-methyl-4-[[3-[4-(trifluoromethyl)phenyl]-1,2-oxazol-5-yl]methylsulfanyl]phenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC=C1SCC1=CC(C=2C=CC(=CC=2)C(F)(F)F)=NO1 OTXMGZLEOJVEDX-UHFFFAOYSA-N 0.000 description 3
- INYFSJMDFOYYBP-UHFFFAOYSA-N methyl 2-[2-methyl-4-[[4-[4-(trifluoromethyl)phenyl]phenyl]methylsulfanyl]phenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC=C1SCC1=CC=C(C=2C=CC(=CC=2)C(F)(F)F)C=C1 INYFSJMDFOYYBP-UHFFFAOYSA-N 0.000 description 3
- VJYCCLULANANMA-UHFFFAOYSA-N methyl 2-[2-methyl-4-[[4-[5-(trifluoromethyl)pyridin-2-yl]phenyl]methylsulfanyl]phenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC=C1SCC1=CC=C(C=2N=CC(=CC=2)C(F)(F)F)C=C1 VJYCCLULANANMA-UHFFFAOYSA-N 0.000 description 3
- UKXLEPRUIDBFGI-UHFFFAOYSA-N methyl 2-[2-methyl-4-[[4-[[4-(trifluoromethoxy)phenyl]methoxy]phenyl]methylsulfanyl]phenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC=C1SCC(C=C1)=CC=C1OCC1=CC=C(OC(F)(F)F)C=C1 UKXLEPRUIDBFGI-UHFFFAOYSA-N 0.000 description 3
- AWVBVZCJSBAUBY-UHFFFAOYSA-N methyl 2-[2-methyl-4-[[4-[[4-(trifluoromethyl)phenyl]methoxy]phenyl]methylsulfanyl]phenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC=C1SCC(C=C1)=CC=C1OCC1=CC=C(C(F)(F)F)C=C1 AWVBVZCJSBAUBY-UHFFFAOYSA-N 0.000 description 3
- OEFTZLHSZDWCLC-UHFFFAOYSA-N methyl 2-[2-propyl-4-[[4-[4-(trifluoromethyl)phenyl]phenyl]methylsulfanyl]phenoxy]acetate Chemical compound C1=C(OCC(=O)OC)C(CCC)=CC(SCC=2C=CC(=CC=2)C=2C=CC(=CC=2)C(F)(F)F)=C1 OEFTZLHSZDWCLC-UHFFFAOYSA-N 0.000 description 3
- SRCZIGQVHPCJAU-UHFFFAOYSA-N methyl 2-[3-methoxy-4-[[4-[4-(trifluoromethyl)phenyl]phenyl]methylsulfanyl]phenoxy]acetate Chemical compound COC1=CC(OCC(=O)OC)=CC=C1SCC1=CC=C(C=2C=CC(=CC=2)C(F)(F)F)C=C1 SRCZIGQVHPCJAU-UHFFFAOYSA-N 0.000 description 3
- NPXDQXBGHRTWPK-UHFFFAOYSA-N methyl 2-[4-[[4-(2,4-dichlorophenyl)phenyl]methylsulfanyl]-5-methoxy-2-methylphenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC(OC)=C1SCC1=CC=C(C=2C(=CC(Cl)=CC=2)Cl)C=C1 NPXDQXBGHRTWPK-UHFFFAOYSA-N 0.000 description 3
- XQQDCVULUIAUNQ-UHFFFAOYSA-N methyl 2-[4-[[4-(3,4-dichlorophenyl)phenyl]methylsulfanyl]-5-methoxy-2-methylphenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC(OC)=C1SCC1=CC=C(C=2C=C(Cl)C(Cl)=CC=2)C=C1 XQQDCVULUIAUNQ-UHFFFAOYSA-N 0.000 description 3
- JDEXWJPKJAGNGW-UHFFFAOYSA-N methyl 2-[4-[[4-(4-fluorophenyl)phenyl]methylsulfanyl]-5-methoxy-2-methylphenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC(OC)=C1SCC1=CC=C(C=2C=CC(F)=CC=2)C=C1 JDEXWJPKJAGNGW-UHFFFAOYSA-N 0.000 description 3
- FXNYKNSKPKCPOP-UHFFFAOYSA-N methyl 2-[4-[[4-[(2,4-dichlorophenyl)methoxy]phenyl]methylsulfanyl]-2-methylphenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC=C1SCC(C=C1)=CC=C1OCC1=CC=C(Cl)C=C1Cl FXNYKNSKPKCPOP-UHFFFAOYSA-N 0.000 description 3
- UCFCYRZSYKTCNK-UHFFFAOYSA-N methyl 2-[4-[[4-[(2,5-dichlorophenyl)methoxy]phenyl]methylsulfanyl]-2-methylphenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC=C1SCC(C=C1)=CC=C1OCC1=CC(Cl)=CC=C1Cl UCFCYRZSYKTCNK-UHFFFAOYSA-N 0.000 description 3
- KXSXAUPIYAHESS-UHFFFAOYSA-N methyl 2-[4-[[4-[(4-chlorophenyl)methoxy]phenyl]methylsulfanyl]-2-methylphenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC=C1SCC(C=C1)=CC=C1OCC1=CC=C(Cl)C=C1 KXSXAUPIYAHESS-UHFFFAOYSA-N 0.000 description 3
- HYUMBMNSTNHLPH-UHFFFAOYSA-N methyl 2-[4-[[4-[(4-fluorophenyl)methoxy]phenyl]methylsulfanyl]-2-methylphenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC=C1SCC(C=C1)=CC=C1OCC1=CC=C(F)C=C1 HYUMBMNSTNHLPH-UHFFFAOYSA-N 0.000 description 3
- WLESGZDLZOTIES-UHFFFAOYSA-N methyl 2-[4-[[4-[(4-methoxyphenyl)methoxy]phenyl]methylsulfanyl]-2-methylphenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC=C1SCC(C=C1)=CC=C1OCC1=CC=C(OC)C=C1 WLESGZDLZOTIES-UHFFFAOYSA-N 0.000 description 3
- TZNXQDFUXBGMBS-UHFFFAOYSA-N methyl 2-[4-[[4-[(4-tert-butylphenyl)methoxy]phenyl]methylsulfanyl]-2-methylphenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC=C1SCC(C=C1)=CC=C1OCC1=CC=C(C(C)(C)C)C=C1 TZNXQDFUXBGMBS-UHFFFAOYSA-N 0.000 description 3
- IWMMVMMNIDBXES-UHFFFAOYSA-N methyl 2-[4-[[4-[4-chloro-3-(trifluoromethyl)phenyl]phenyl]methylsulfanyl]-2-methylphenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC=C1SCC1=CC=C(C=2C=C(C(Cl)=CC=2)C(F)(F)F)C=C1 IWMMVMMNIDBXES-UHFFFAOYSA-N 0.000 description 3
- XRFXLGQDXWWQQP-UHFFFAOYSA-N methyl 2-[4-[[5-(4-chlorophenyl)-1,2-oxazol-3-yl]methylsulfanyl]-2-methylphenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC=C1SCC1=NOC(C=2C=CC(Cl)=CC=2)=C1 XRFXLGQDXWWQQP-UHFFFAOYSA-N 0.000 description 3
- LVGYQBQGWWSGMM-UHFFFAOYSA-N methyl 2-[4-[[5-(4-chlorophenyl)-1,2-oxazol-3-yl]methylsulfanyl]-5-methoxy-2-methylphenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC(OC)=C1SCC1=NOC(C=2C=CC(Cl)=CC=2)=C1 LVGYQBQGWWSGMM-UHFFFAOYSA-N 0.000 description 3
- ISNUIUZHBRJQKF-UHFFFAOYSA-N methyl 2-[5-chloro-2-methyl-4-[[4-[4-(trifluoromethyl)phenyl]phenyl]methylsulfanyl]phenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC(Cl)=C1SCC1=CC=C(C=2C=CC(=CC=2)C(F)(F)F)C=C1 ISNUIUZHBRJQKF-UHFFFAOYSA-N 0.000 description 3
- VMWJGTIBWLCEOC-UHFFFAOYSA-N methyl 2-[5-chloro-2-methyl-4-[[4-[[4-(trifluoromethyl)phenyl]methoxy]phenyl]methylsulfanyl]phenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC(Cl)=C1SCC(C=C1)=CC=C1OCC1=CC=C(C(F)(F)F)C=C1 VMWJGTIBWLCEOC-UHFFFAOYSA-N 0.000 description 3
- QKEKSHDGTKEXDJ-UHFFFAOYSA-N methyl 2-[5-methoxy-2-methyl-4-[[2-[[4-(trifluoromethyl)phenyl]methoxy]phenyl]methylsulfanyl]phenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC(OC)=C1SCC1=CC=CC=C1OCC1=CC=C(C(F)(F)F)C=C1 QKEKSHDGTKEXDJ-UHFFFAOYSA-N 0.000 description 3
- PRPQLYOWUTXFBZ-UHFFFAOYSA-N methyl 2-[5-methoxy-2-methyl-4-[[2-methyl-4-[4-(trifluoromethyl)phenyl]phenyl]methylsulfanyl]phenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC(OC)=C1SCC1=CC=C(C=2C=CC(=CC=2)C(F)(F)F)C=C1C PRPQLYOWUTXFBZ-UHFFFAOYSA-N 0.000 description 3
- XEZKFLNHJSXRQB-UHFFFAOYSA-N methyl 2-[5-methoxy-2-methyl-4-[[3-[3-(trifluoromethoxy)phenyl]phenyl]methylsulfanyl]phenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC(OC)=C1SCC1=CC=CC(C=2C=C(OC(F)(F)F)C=CC=2)=C1 XEZKFLNHJSXRQB-UHFFFAOYSA-N 0.000 description 3
- HWKNOVZBKYEOHE-UHFFFAOYSA-N methyl 2-[5-methoxy-2-methyl-4-[[3-[4-(trifluoromethyl)phenyl]-1,2-oxazol-5-yl]methylsulfanyl]phenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC(OC)=C1SCC1=CC(C=2C=CC(=CC=2)C(F)(F)F)=NO1 HWKNOVZBKYEOHE-UHFFFAOYSA-N 0.000 description 3
- KQTNQUKAFBDSPF-UHFFFAOYSA-N methyl 2-[5-methoxy-2-methyl-4-[[3-[4-(trifluoromethyl)phenyl]phenyl]methylsulfanyl]phenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC(OC)=C1SCC1=CC=CC(C=2C=CC(=CC=2)C(F)(F)F)=C1 KQTNQUKAFBDSPF-UHFFFAOYSA-N 0.000 description 3
- VWUWVLCOBKXIMS-UHFFFAOYSA-N methyl 2-[5-methoxy-2-methyl-4-[[4-[3-(trifluoromethoxy)phenyl]phenyl]methylsulfanyl]phenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC(OC)=C1SCC1=CC=C(C=2C=C(OC(F)(F)F)C=CC=2)C=C1 VWUWVLCOBKXIMS-UHFFFAOYSA-N 0.000 description 3
- PTVHGDJMKHFXQF-UHFFFAOYSA-N methyl 2-[5-methoxy-2-methyl-4-[[4-[3-(trifluoromethyl)phenyl]phenyl]methylsulfanyl]phenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC(OC)=C1SCC1=CC=C(C=2C=C(C=CC=2)C(F)(F)F)C=C1 PTVHGDJMKHFXQF-UHFFFAOYSA-N 0.000 description 3
- HLBPHSXQGYRDST-UHFFFAOYSA-N methyl 2-[5-methoxy-2-methyl-4-[[4-[4-(trifluoromethyl)phenyl]phenyl]methylsulfinyl]phenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC(OC)=C1S(=O)CC1=CC=C(C=2C=CC(=CC=2)C(F)(F)F)C=C1 HLBPHSXQGYRDST-UHFFFAOYSA-N 0.000 description 3
- HHDJDISYASLJPU-UHFFFAOYSA-N methyl 2-[5-methoxy-2-methyl-4-[[4-[4-(trifluoromethyl)phenyl]phenyl]methylsulfonyl]phenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC(OC)=C1S(=O)(=O)CC1=CC=C(C=2C=CC(=CC=2)C(F)(F)F)C=C1 HHDJDISYASLJPU-UHFFFAOYSA-N 0.000 description 3
- KWRBTVOUVZWXSI-UHFFFAOYSA-N methyl 2-[5-methoxy-2-methyl-4-[[4-[5-(trifluoromethyl)pyridin-2-yl]phenyl]methylsulfanyl]phenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC(OC)=C1SCC1=CC=C(C=2N=CC(=CC=2)C(F)(F)F)C=C1 KWRBTVOUVZWXSI-UHFFFAOYSA-N 0.000 description 3
- RTBSBIGLBCOBGP-UHFFFAOYSA-N methyl 2-[5-methoxy-2-methyl-4-[[4-[[4-(trifluoromethyl)phenyl]methoxy]phenyl]methylsulfanyl]phenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC(OC)=C1SCC(C=C1)=CC=C1OCC1=CC=C(C(F)(F)F)C=C1 RTBSBIGLBCOBGP-UHFFFAOYSA-N 0.000 description 3
- PPYLECZZTUJEIG-UHFFFAOYSA-N methyl 2-[5-methoxy-2-methyl-4-[[5-[4-(trifluoromethyl)phenyl]-1,2-oxazol-3-yl]methylsulfanyl]phenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC(OC)=C1SCC1=NOC(C=2C=CC(=CC=2)C(F)(F)F)=C1 PPYLECZZTUJEIG-UHFFFAOYSA-N 0.000 description 3
- KRNNZBNJCAPIIO-UHFFFAOYSA-N methyl 2-[[4-[[5-[4-(trifluoromethyl)phenyl]-1,2-oxazol-3-yl]methylsulfanyl]-5,6,7,8-tetrahydronaphthalen-1-yl]oxy]acetate Chemical compound C1=2CCCCC=2C(OCC(=O)OC)=CC=C1SCC(=NO1)C=C1C1=CC=C(C(F)(F)F)C=C1 KRNNZBNJCAPIIO-UHFFFAOYSA-N 0.000 description 3
- MPCZTZIZHRLHPO-UHFFFAOYSA-N methyl 2-[[5-methyl-7-[[4-[4-(trifluoromethyl)phenyl]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=C2CCCC2=C1SCC(C=C1)=CC=C1C1=CC=C(C(F)(F)F)C=C1 MPCZTZIZHRLHPO-UHFFFAOYSA-N 0.000 description 3
- UTZXQILJDZRVOI-UHFFFAOYSA-N methyl 2-[[5-methyl-7-[[4-[5-(trifluoromethyl)pyridin-2-yl]phenyl]methylsulfanyl]-2,3-dihydro-1-benzofuran-4-yl]oxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=C2CCOC2=C1SCC(C=C1)=CC=C1C1=CC=C(C(F)(F)F)C=N1 UTZXQILJDZRVOI-UHFFFAOYSA-N 0.000 description 3
- KAYODKXXCBUOKZ-UHFFFAOYSA-N methyl 2-[[5-methyl-7-[[4-[5-(trifluoromethyl)pyridin-2-yl]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=C2CCCC2=C1SCC(C=C1)=CC=C1C1=CC=C(C(F)(F)F)C=N1 KAYODKXXCBUOKZ-UHFFFAOYSA-N 0.000 description 3
- VQGMAQIRTOSILA-UHFFFAOYSA-N methyl 2-[[5-methyl-7-[[4-[[4-(trifluoromethyl)phenyl]methoxy]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=C2CCCC2=C1SCC(C=C1)=CC=C1OCC1=CC=C(C(F)(F)F)C=C1 VQGMAQIRTOSILA-UHFFFAOYSA-N 0.000 description 3
- JLUVMQDJKRUNDG-UHFFFAOYSA-N methyl 2-[[7-[(4-acetyloxyphenyl)methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetate Chemical compound C1=2CCCC=2C(OCC(=O)OC)=CC=C1SCC1=CC=C(OC(C)=O)C=C1 JLUVMQDJKRUNDG-UHFFFAOYSA-N 0.000 description 3
- JBWOURYTDKMHQR-UHFFFAOYSA-N methyl 2-[[7-[(4-phenylmethoxyphenyl)methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetate Chemical compound C1=2CCCC=2C(OCC(=O)OC)=CC=C1SCC(C=C1)=CC=C1OCC1=CC=CC=C1 JBWOURYTDKMHQR-UHFFFAOYSA-N 0.000 description 3
- DNASJQNRLJMRPG-UHFFFAOYSA-N methyl 2-[[7-[2-[4-[4-(trifluoromethyl)phenyl]phenyl]ethylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetate Chemical compound C1=2CCCC=2C(OCC(=O)OC)=CC=C1SCCC(C=C1)=CC=C1C1=CC=C(C(F)(F)F)C=C1 DNASJQNRLJMRPG-UHFFFAOYSA-N 0.000 description 3
- DNOJIGAIZBODMQ-UHFFFAOYSA-N methyl 2-[[7-[[2-[4-(trifluoromethyl)phenyl]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetate Chemical compound C1=2CCCC=2C(OCC(=O)OC)=CC=C1SCC1=CC=CC=C1C1=CC=C(C(F)(F)F)C=C1 DNOJIGAIZBODMQ-UHFFFAOYSA-N 0.000 description 3
- NGRZOPVOIQFFGR-UHFFFAOYSA-N methyl 2-[[7-[[2-[[4-(trifluoromethyl)phenyl]methoxy]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetate Chemical compound C1=2CCCC=2C(OCC(=O)OC)=CC=C1SCC1=CC=CC=C1OCC1=CC=C(C(F)(F)F)C=C1 NGRZOPVOIQFFGR-UHFFFAOYSA-N 0.000 description 3
- PJKJZTATBLCBEC-UHFFFAOYSA-N methyl 2-[[7-[[3,5-dichloro-4-[[4-(trifluoromethyl)phenyl]methoxy]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetate Chemical compound C1=2CCCC=2C(OCC(=O)OC)=CC=C1SCC(C=C1Cl)=CC(Cl)=C1OCC1=CC=C(C(F)(F)F)C=C1 PJKJZTATBLCBEC-UHFFFAOYSA-N 0.000 description 3
- BBUSUGUJHBPBNK-UHFFFAOYSA-N methyl 2-[[7-[[3-(2,6-dichlorophenyl)-5-methyl-1,2-oxazol-4-yl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetate Chemical compound C1=2CCCC=2C(OCC(=O)OC)=CC=C1SCC1=C(C)ON=C1C1=C(Cl)C=CC=C1Cl BBUSUGUJHBPBNK-UHFFFAOYSA-N 0.000 description 3
- BRWLOXSQQPEEPO-UHFFFAOYSA-N methyl 2-[[7-[[3-[4-(trifluoromethyl)phenyl]-1,2-oxazol-5-yl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetate Chemical compound C1=2CCCC=2C(OCC(=O)OC)=CC=C1SCC(ON=1)=CC=1C1=CC=C(C(F)(F)F)C=C1 BRWLOXSQQPEEPO-UHFFFAOYSA-N 0.000 description 3
- LFWIIISCRGRPDA-UHFFFAOYSA-N methyl 2-[[7-[[3-[[4-chloro-3-(trifluoromethyl)phenyl]methoxy]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetate Chemical compound C1=2CCCC=2C(OCC(=O)OC)=CC=C1SCC(C=1)=CC=CC=1OCC1=CC=C(Cl)C(C(F)(F)F)=C1 LFWIIISCRGRPDA-UHFFFAOYSA-N 0.000 description 3
- ORUBONIIIKZNQK-UHFFFAOYSA-N methyl 2-[[7-[[4-[(2,4-dichlorophenyl)methoxy]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetate Chemical compound C1=2CCCC=2C(OCC(=O)OC)=CC=C1SCC(C=C1)=CC=C1OCC1=CC=C(Cl)C=C1Cl ORUBONIIIKZNQK-UHFFFAOYSA-N 0.000 description 3
- IIWBMVAJJPPCMZ-UHFFFAOYSA-N methyl 2-[[7-[[4-[(3,4-dichlorophenyl)methoxy]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetate Chemical compound C1=2CCCC=2C(OCC(=O)OC)=CC=C1SCC(C=C1)=CC=C1OCC1=CC=C(Cl)C(Cl)=C1 IIWBMVAJJPPCMZ-UHFFFAOYSA-N 0.000 description 3
- FGCNVUAPCWOSGZ-UHFFFAOYSA-N methyl 2-[[7-[[4-[(3,5-dichlorophenyl)methoxy]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetate Chemical compound C1=2CCCC=2C(OCC(=O)OC)=CC=C1SCC(C=C1)=CC=C1OCC1=CC(Cl)=CC(Cl)=C1 FGCNVUAPCWOSGZ-UHFFFAOYSA-N 0.000 description 3
- YDIDJPRYASPNAN-UHFFFAOYSA-N methyl 2-[[7-[[4-[(4-chlorophenyl)methoxy]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetate Chemical compound C1=2CCCC=2C(OCC(=O)OC)=CC=C1SCC(C=C1)=CC=C1OCC1=CC=C(Cl)C=C1 YDIDJPRYASPNAN-UHFFFAOYSA-N 0.000 description 3
- JVENZGGBNNSTAM-UHFFFAOYSA-N methyl 2-[[7-[[4-[(4-methoxyphenyl)methoxy]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetate Chemical compound C1=2CCCC=2C(OCC(=O)OC)=CC=C1SCC(C=C1)=CC=C1OCC1=CC=C(OC)C=C1 JVENZGGBNNSTAM-UHFFFAOYSA-N 0.000 description 3
- ARFSCPDOWCFLHU-UHFFFAOYSA-N methyl 2-[[7-[[4-[(4-tert-butylphenyl)methoxy]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetate Chemical compound C1=2CCCC=2C(OCC(=O)OC)=CC=C1SCC(C=C1)=CC=C1OCC1=CC=C(C(C)(C)C)C=C1 ARFSCPDOWCFLHU-UHFFFAOYSA-N 0.000 description 3
- YRUUNPXRZDNSAS-UHFFFAOYSA-N methyl 2-[[7-[[4-[4-(trifluoromethyl)phenyl]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetate Chemical compound C1=2CCCC=2C(OCC(=O)OC)=CC=C1SCC(C=C1)=CC=C1C1=CC=C(C(F)(F)F)C=C1 YRUUNPXRZDNSAS-UHFFFAOYSA-N 0.000 description 3
- HVNIATGKLQZTAU-UHFFFAOYSA-N methyl 2-[[7-[[4-[5-(trifluoromethyl)pyridin-2-yl]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetate Chemical compound C1=2CCCC=2C(OCC(=O)OC)=CC=C1SCC(C=C1)=CC=C1C1=CC=C(C(F)(F)F)C=N1 HVNIATGKLQZTAU-UHFFFAOYSA-N 0.000 description 3
- KNCGJRVQNSIMPI-UHFFFAOYSA-N methyl 2-[[7-[[4-[[4-(trifluoromethoxy)phenyl]methoxy]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetate Chemical compound C1=2CCCC=2C(OCC(=O)OC)=CC=C1SCC(C=C1)=CC=C1OCC1=CC=C(OC(F)(F)F)C=C1 KNCGJRVQNSIMPI-UHFFFAOYSA-N 0.000 description 3
- UHMYEWBPNFWLDK-UHFFFAOYSA-N methyl 2-[[7-[[4-[[4-(trifluoromethyl)phenyl]methoxy]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetate Chemical compound C1=2CCCC=2C(OCC(=O)OC)=CC=C1SCC(C=C1)=CC=C1OCC1=CC=C(C(F)(F)F)C=C1 UHMYEWBPNFWLDK-UHFFFAOYSA-N 0.000 description 3
- XWVIVUKZDZOHNE-UHFFFAOYSA-N methyl 2-[[7-[[4-[[4-(trifluoromethyl)phenyl]methyl]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetate Chemical compound C1=2CCCC=2C(OCC(=O)OC)=CC=C1SCC(C=C1)=CC=C1CC1=CC=C(C(F)(F)F)C=C1 XWVIVUKZDZOHNE-UHFFFAOYSA-N 0.000 description 3
- QRBXBUJTMWEYCP-UHFFFAOYSA-N methyl 2-[[7-[[4-[[4-chloro-3-(trifluoromethyl)phenyl]methoxy]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetate Chemical compound C1=2CCCC=2C(OCC(=O)OC)=CC=C1SCC(C=C1)=CC=C1OCC1=CC=C(Cl)C(C(F)(F)F)=C1 QRBXBUJTMWEYCP-UHFFFAOYSA-N 0.000 description 3
- LMLVKIGGDHRBMF-UHFFFAOYSA-N methyl 2-[[7-[[4-[[4-fluoro-3-(trifluoromethyl)phenyl]methoxy]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetate Chemical compound C1=2CCCC=2C(OCC(=O)OC)=CC=C1SCC(C=C1)=CC=C1OCC1=CC=C(F)C(C(F)(F)F)=C1 LMLVKIGGDHRBMF-UHFFFAOYSA-N 0.000 description 3
- NLLDMRFDRAVZQL-UHFFFAOYSA-N methyl 2-[[7-[[4-methoxy-3-[[4-(trifluoromethyl)phenyl]methoxy]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetate Chemical compound C1=2CCCC=2C(OCC(=O)OC)=CC=C1SCC(C=1)=CC=C(OC)C=1OCC1=CC=C(C(F)(F)F)C=C1 NLLDMRFDRAVZQL-UHFFFAOYSA-N 0.000 description 3
- IVKQMXASIHXTAA-UHFFFAOYSA-N methyl 2-[[7-[[5-(2-chlorophenyl)-1,2-oxazol-3-yl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetate Chemical compound C1=2CCCC=2C(OCC(=O)OC)=CC=C1SCC(=NO1)C=C1C1=CC=CC=C1Cl IVKQMXASIHXTAA-UHFFFAOYSA-N 0.000 description 3
- PBDIKPJXZUFSTJ-UHFFFAOYSA-N methyl 2-[[7-[[5-(4-chlorophenyl)-1,2-oxazol-3-yl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetate Chemical compound C1=2CCCC=2C(OCC(=O)OC)=CC=C1SCC(=NO1)C=C1C1=CC=C(Cl)C=C1 PBDIKPJXZUFSTJ-UHFFFAOYSA-N 0.000 description 3
- JNWHOOBDULSEOL-UHFFFAOYSA-N methyl 2-[[7-[[5-[4-(trifluoromethyl)phenyl]-1,2-oxazol-3-yl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetate Chemical compound C1=2CCCC=2C(OCC(=O)OC)=CC=C1SCC(=NO1)C=C1C1=CC=C(C(F)(F)F)C=C1 JNWHOOBDULSEOL-UHFFFAOYSA-N 0.000 description 3
- YPWVYYSFKLYBSW-UHFFFAOYSA-N methyl 2-[[8-[[3-[[4-(trifluoromethyl)phenyl]methoxy]phenyl]methylsulfanyl]-3,4-dihydro-2h-chromen-5-yl]oxy]acetate Chemical compound C1=2OCCCC=2C(OCC(=O)OC)=CC=C1SCC(C=1)=CC=CC=1OCC1=CC=C(C(F)(F)F)C=C1 YPWVYYSFKLYBSW-UHFFFAOYSA-N 0.000 description 3
- SZIJBWDMKZLXJI-UHFFFAOYSA-N methyl 2-[[8-[[4-[(2,5-dichlorophenyl)methoxy]phenyl]methylsulfanyl]-3,4-dihydro-2h-chromen-5-yl]oxy]acetate Chemical compound C1=2OCCCC=2C(OCC(=O)OC)=CC=C1SCC(C=C1)=CC=C1OCC1=CC(Cl)=CC=C1Cl SZIJBWDMKZLXJI-UHFFFAOYSA-N 0.000 description 3
- MLNANIYHSUYOGU-UHFFFAOYSA-N methyl 2-[[8-[[4-[4-(trifluoromethyl)phenyl]phenyl]methylsulfanyl]-3,4-dihydro-2h-chromen-5-yl]oxy]acetate Chemical compound C1=2OCCCC=2C(OCC(=O)OC)=CC=C1SCC(C=C1)=CC=C1C1=CC=C(C(F)(F)F)C=C1 MLNANIYHSUYOGU-UHFFFAOYSA-N 0.000 description 3
- GSLLEYGJXMSGHZ-UHFFFAOYSA-N methyl 2-[[8-[[4-[5-(trifluoromethyl)pyridin-2-yl]phenyl]methylsulfanyl]-3,4-dihydro-2h-chromen-5-yl]oxy]acetate Chemical compound C1=2OCCCC=2C(OCC(=O)OC)=CC=C1SCC(C=C1)=CC=C1C1=CC=C(C(F)(F)F)C=N1 GSLLEYGJXMSGHZ-UHFFFAOYSA-N 0.000 description 3
- VPLSUCWRJIYNEW-UHFFFAOYSA-N methyl 2-[[8-[[4-[[4-(trifluoromethyl)phenyl]methoxy]phenyl]methylsulfanyl]-3,4-dihydro-2h-chromen-5-yl]oxy]acetate Chemical compound C1=2OCCCC=2C(OCC(=O)OC)=CC=C1SCC(C=C1)=CC=C1OCC1=CC=C(C(F)(F)F)C=C1 VPLSUCWRJIYNEW-UHFFFAOYSA-N 0.000 description 3
- JDTILJJBRCBDAB-UHFFFAOYSA-N methyl 2-[[8-[[5-(4-chlorophenyl)-1,2-oxazol-3-yl]methylsulfanyl]-3,4-dihydro-2h-chromen-5-yl]oxy]acetate Chemical compound C1=2OCCCC=2C(OCC(=O)OC)=CC=C1SCC(=NO1)C=C1C1=CC=C(Cl)C=C1 JDTILJJBRCBDAB-UHFFFAOYSA-N 0.000 description 3
- DQYSYWYICGJUHP-UHFFFAOYSA-N methyl 2-[[8-[[5-[4-(trifluoromethyl)phenyl]-1,2-oxazol-3-yl]methylsulfanyl]-3,4-dihydro-2h-chromen-5-yl]oxy]acetate Chemical compound C1=2OCCCC=2C(OCC(=O)OC)=CC=C1SCC(=NO1)C=C1C1=CC=C(C(F)(F)F)C=C1 DQYSYWYICGJUHP-UHFFFAOYSA-N 0.000 description 3
- QGMKBNZQKNECNH-UHFFFAOYSA-N methyl 4-[[4-(trifluoromethyl)phenyl]methyl]benzoate Chemical compound C1=CC(C(=O)OC)=CC=C1CC1=CC=C(C(F)(F)F)C=C1 QGMKBNZQKNECNH-UHFFFAOYSA-N 0.000 description 3
- 125000002950 monocyclic group Chemical group 0.000 description 3
- 231100000252 nontoxic Toxicity 0.000 description 3
- 230000003000 nontoxic effect Effects 0.000 description 3
- CQRYARSYNCAZFO-UHFFFAOYSA-N o-hydroxybenzyl alcohol Natural products OCC1=CC=CC=C1O CQRYARSYNCAZFO-UHFFFAOYSA-N 0.000 description 3
- 239000001301 oxygen Substances 0.000 description 3
- PIBWKRNGBLPSSY-UHFFFAOYSA-L palladium(II) chloride Chemical compound Cl[Pd]Cl PIBWKRNGBLPSSY-UHFFFAOYSA-L 0.000 description 3
- 239000006187 pill Substances 0.000 description 3
- 239000013612 plasmid Substances 0.000 description 3
- 229940068917 polyethylene glycols Drugs 0.000 description 3
- 239000002244 precipitate Substances 0.000 description 3
- 150000003254 radicals Chemical class 0.000 description 3
- 230000009467 reduction Effects 0.000 description 3
- 238000010992 reflux Methods 0.000 description 3
- 125000006413 ring segment Chemical group 0.000 description 3
- 229910000030 sodium bicarbonate Inorganic materials 0.000 description 3
- 229910000104 sodium hydride Inorganic materials 0.000 description 3
- 235000011152 sodium sulphate Nutrition 0.000 description 3
- 239000007909 solid dosage form Substances 0.000 description 3
- 235000019698 starch Nutrition 0.000 description 3
- 239000000126 substance Substances 0.000 description 3
- 210000001519 tissue Anatomy 0.000 description 3
- 125000000876 trifluoromethoxy group Chemical group FC(F)(F)O* 0.000 description 3
- PUPZLCDOIYMWBV-UHFFFAOYSA-N (+/-)-1,3-Butanediol Chemical compound CC(O)CCO PUPZLCDOIYMWBV-UHFFFAOYSA-N 0.000 description 2
- ASGMFNBUXDJWJJ-JLCFBVMHSA-N (1R,3R)-3-[[3-bromo-1-[4-(5-methyl-1,3,4-thiadiazol-2-yl)phenyl]pyrazolo[3,4-d]pyrimidin-6-yl]amino]-N,1-dimethylcyclopentane-1-carboxamide Chemical compound BrC1=NN(C2=NC(=NC=C21)N[C@H]1C[C@@](CC1)(C(=O)NC)C)C1=CC=C(C=C1)C=1SC(=NN=1)C ASGMFNBUXDJWJJ-JLCFBVMHSA-N 0.000 description 2
- UAOUIVVJBYDFKD-XKCDOFEDSA-N (1R,9R,10S,11R,12R,15S,18S,21R)-10,11,21-trihydroxy-8,8-dimethyl-14-methylidene-4-(prop-2-enylamino)-20-oxa-5-thia-3-azahexacyclo[9.7.2.112,15.01,9.02,6.012,18]henicosa-2(6),3-dien-13-one Chemical compound C([C@@H]1[C@@H](O)[C@@]23C(C1=C)=O)C[C@H]2[C@]12C(N=C(NCC=C)S4)=C4CC(C)(C)[C@H]1[C@H](O)[C@]3(O)OC2 UAOUIVVJBYDFKD-XKCDOFEDSA-N 0.000 description 2
- AOSZTAHDEDLTLQ-AZKQZHLXSA-N (1S,2S,4R,8S,9S,11S,12R,13S,19S)-6-[(3-chlorophenyl)methyl]-12,19-difluoro-11-hydroxy-8-(2-hydroxyacetyl)-9,13-dimethyl-6-azapentacyclo[10.8.0.02,9.04,8.013,18]icosa-14,17-dien-16-one Chemical compound C([C@@H]1C[C@H]2[C@H]3[C@]([C@]4(C=CC(=O)C=C4[C@@H](F)C3)C)(F)[C@@H](O)C[C@@]2([C@@]1(C1)C(=O)CO)C)N1CC1=CC=CC(Cl)=C1 AOSZTAHDEDLTLQ-AZKQZHLXSA-N 0.000 description 2
- ABJSOROVZZKJGI-OCYUSGCXSA-N (1r,2r,4r)-2-(4-bromophenyl)-n-[(4-chlorophenyl)-(2-fluoropyridin-4-yl)methyl]-4-morpholin-4-ylcyclohexane-1-carboxamide Chemical compound C1=NC(F)=CC(C(NC(=O)[C@H]2[C@@H](C[C@@H](CC2)N2CCOCC2)C=2C=CC(Br)=CC=2)C=2C=CC(Cl)=CC=2)=C1 ABJSOROVZZKJGI-OCYUSGCXSA-N 0.000 description 2
- SZUVGFMDDVSKSI-WIFOCOSTSA-N (1s,2s,3s,5r)-1-(carboxymethyl)-3,5-bis[(4-phenoxyphenyl)methyl-propylcarbamoyl]cyclopentane-1,2-dicarboxylic acid Chemical compound O=C([C@@H]1[C@@H]([C@](CC(O)=O)([C@H](C(=O)N(CCC)CC=2C=CC(OC=3C=CC=CC=3)=CC=2)C1)C(O)=O)C(O)=O)N(CCC)CC(C=C1)=CC=C1OC1=CC=CC=C1 SZUVGFMDDVSKSI-WIFOCOSTSA-N 0.000 description 2
- IEYBQRZTYDTMSI-UHFFFAOYSA-N (2-chloro-4-hydroxy-5-methylphenyl) thiocyanate Chemical compound CC1=CC(SC#N)=C(Cl)C=C1O IEYBQRZTYDTMSI-UHFFFAOYSA-N 0.000 description 2
- GHYOCDFICYLMRF-UTIIJYGPSA-N (2S,3R)-N-[(2S)-3-(cyclopenten-1-yl)-1-[(2R)-2-methyloxiran-2-yl]-1-oxopropan-2-yl]-3-hydroxy-3-(4-methoxyphenyl)-2-[[(2S)-2-[(2-morpholin-4-ylacetyl)amino]propanoyl]amino]propanamide Chemical compound C1(=CCCC1)C[C@@H](C(=O)[C@@]1(OC1)C)NC([C@H]([C@@H](C1=CC=C(C=C1)OC)O)NC([C@H](C)NC(CN1CCOCC1)=O)=O)=O GHYOCDFICYLMRF-UTIIJYGPSA-N 0.000 description 2
- IUSARDYWEPUTPN-OZBXUNDUSA-N (2r)-n-[(2s,3r)-4-[[(4s)-6-(2,2-dimethylpropyl)spiro[3,4-dihydropyrano[2,3-b]pyridine-2,1'-cyclobutane]-4-yl]amino]-3-hydroxy-1-[3-(1,3-thiazol-2-yl)phenyl]butan-2-yl]-2-methoxypropanamide Chemical compound C([C@H](NC(=O)[C@@H](C)OC)[C@H](O)CN[C@@H]1C2=CC(CC(C)(C)C)=CN=C2OC2(CCC2)C1)C(C=1)=CC=CC=1C1=NC=CS1 IUSARDYWEPUTPN-OZBXUNDUSA-N 0.000 description 2
- JNYAEWCLZODPBN-JGWLITMVSA-N (2r,3r,4s)-2-[(1r)-1,2-dihydroxyethyl]oxolane-3,4-diol Chemical compound OC[C@@H](O)[C@H]1OC[C@H](O)[C@H]1O JNYAEWCLZODPBN-JGWLITMVSA-N 0.000 description 2
- YJLIKUSWRSEPSM-WGQQHEPDSA-N (2r,3r,4s,5r)-2-[6-amino-8-[(4-phenylphenyl)methylamino]purin-9-yl]-5-(hydroxymethyl)oxolane-3,4-diol Chemical compound C=1C=C(C=2C=CC=CC=2)C=CC=1CNC1=NC=2C(N)=NC=NC=2N1[C@@H]1O[C@H](CO)[C@@H](O)[C@H]1O YJLIKUSWRSEPSM-WGQQHEPDSA-N 0.000 description 2
- VIJSPAIQWVPKQZ-BLECARSGSA-N (2s)-2-[[(2s)-2-[[(2s)-2-[[(2s)-2-[[(2s)-2-[[(2s)-2-acetamido-5-(diaminomethylideneamino)pentanoyl]amino]-4-methylpentanoyl]amino]-4,4-dimethylpentanoyl]amino]-4-methylpentanoyl]amino]propanoyl]amino]-5-(diaminomethylideneamino)pentanoic acid Chemical compound NC(=N)NCCC[C@@H](C(O)=O)NC(=O)[C@H](C)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC(C)(C)C)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CCCNC(N)=N)NC(C)=O VIJSPAIQWVPKQZ-BLECARSGSA-N 0.000 description 2
- WWTBZEKOSBFBEM-SPWPXUSOSA-N (2s)-2-[[2-benzyl-3-[hydroxy-[(1r)-2-phenyl-1-(phenylmethoxycarbonylamino)ethyl]phosphoryl]propanoyl]amino]-3-(1h-indol-3-yl)propanoic acid Chemical compound N([C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)O)C(=O)C(CP(O)(=O)[C@H](CC=1C=CC=CC=1)NC(=O)OCC=1C=CC=CC=1)CC1=CC=CC=C1 WWTBZEKOSBFBEM-SPWPXUSOSA-N 0.000 description 2
- STBLNCCBQMHSRC-BATDWUPUSA-N (2s)-n-[(3s,4s)-5-acetyl-7-cyano-4-methyl-1-[(2-methylnaphthalen-1-yl)methyl]-2-oxo-3,4-dihydro-1,5-benzodiazepin-3-yl]-2-(methylamino)propanamide Chemical compound O=C1[C@@H](NC(=O)[C@H](C)NC)[C@H](C)N(C(C)=O)C2=CC(C#N)=CC=C2N1CC1=C(C)C=CC2=CC=CC=C12 STBLNCCBQMHSRC-BATDWUPUSA-N 0.000 description 2
- QFLWZFQWSBQYPS-AWRAUJHKSA-N (3S)-3-[[(2S)-2-[[(2S)-2-[5-[(3aS,6aR)-2-oxo-1,3,3a,4,6,6a-hexahydrothieno[3,4-d]imidazol-4-yl]pentanoylamino]-3-methylbutanoyl]amino]-3-(4-hydroxyphenyl)propanoyl]amino]-4-[1-bis(4-chlorophenoxy)phosphorylbutylamino]-4-oxobutanoic acid Chemical compound CCCC(NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](Cc1ccc(O)cc1)NC(=O)[C@@H](NC(=O)CCCCC1SC[C@@H]2NC(=O)N[C@H]12)C(C)C)P(=O)(Oc1ccc(Cl)cc1)Oc1ccc(Cl)cc1 QFLWZFQWSBQYPS-AWRAUJHKSA-N 0.000 description 2
- IWZSHWBGHQBIML-ZGGLMWTQSA-N (3S,8S,10R,13S,14S,17S)-17-isoquinolin-7-yl-N,N,10,13-tetramethyl-2,3,4,7,8,9,11,12,14,15,16,17-dodecahydro-1H-cyclopenta[a]phenanthren-3-amine Chemical compound CN(C)[C@H]1CC[C@]2(C)C3CC[C@@]4(C)[C@@H](CC[C@@H]4c4ccc5ccncc5c4)[C@@H]3CC=C2C1 IWZSHWBGHQBIML-ZGGLMWTQSA-N 0.000 description 2
- OOKAZRDERJMRCJ-KOUAFAAESA-N (3r)-7-[(1s,2s,4ar,6s,8s)-2,6-dimethyl-8-[(2s)-2-methylbutanoyl]oxy-1,2,4a,5,6,7,8,8a-octahydronaphthalen-1-yl]-3-hydroxy-5-oxoheptanoic acid Chemical compound C1=C[C@H](C)[C@H](CCC(=O)C[C@@H](O)CC(O)=O)C2[C@@H](OC(=O)[C@@H](C)CC)C[C@@H](C)C[C@@H]21 OOKAZRDERJMRCJ-KOUAFAAESA-N 0.000 description 2
- HUWSZNZAROKDRZ-RRLWZMAJSA-N (3r,4r)-3-azaniumyl-5-[[(2s,3r)-1-[(2s)-2,3-dicarboxypyrrolidin-1-yl]-3-methyl-1-oxopentan-2-yl]amino]-5-oxo-4-sulfanylpentane-1-sulfonate Chemical compound OS(=O)(=O)CC[C@@H](N)[C@@H](S)C(=O)N[C@@H]([C@H](C)CC)C(=O)N1CCC(C(O)=O)[C@H]1C(O)=O HUWSZNZAROKDRZ-RRLWZMAJSA-N 0.000 description 2
- MPDDTAJMJCESGV-CTUHWIOQSA-M (3r,5r)-7-[2-(4-fluorophenyl)-5-[methyl-[(1r)-1-phenylethyl]carbamoyl]-4-propan-2-ylpyrazol-3-yl]-3,5-dihydroxyheptanoate Chemical compound C1([C@@H](C)N(C)C(=O)C2=NN(C(CC[C@@H](O)C[C@@H](O)CC([O-])=O)=C2C(C)C)C=2C=CC(F)=CC=2)=CC=CC=C1 MPDDTAJMJCESGV-CTUHWIOQSA-M 0.000 description 2
- FIZPVVAFGBDLRR-UHFFFAOYSA-N (4-hydroxy-2-methoxyphenyl) thiocyanate Chemical compound COC1=CC(O)=CC=C1SC#N FIZPVVAFGBDLRR-UHFFFAOYSA-N 0.000 description 2
- WEAMLHXSIBDPGN-UHFFFAOYSA-N (4-hydroxy-3-methylphenyl) thiocyanate Chemical compound CC1=CC(SC#N)=CC=C1O WEAMLHXSIBDPGN-UHFFFAOYSA-N 0.000 description 2
- YQOLEILXOBUDMU-KRWDZBQOSA-N (4R)-5-[(6-bromo-3-methyl-2-pyrrolidin-1-ylquinoline-4-carbonyl)amino]-4-(2-chlorophenyl)pentanoic acid Chemical compound CC1=C(C2=C(C=CC(=C2)Br)N=C1N3CCCC3)C(=O)NC[C@H](CCC(=O)O)C4=CC=CC=C4Cl YQOLEILXOBUDMU-KRWDZBQOSA-N 0.000 description 2
- STPKWKPURVSAJF-LJEWAXOPSA-N (4r,5r)-5-[4-[[4-(1-aza-4-azoniabicyclo[2.2.2]octan-4-ylmethyl)phenyl]methoxy]phenyl]-3,3-dibutyl-7-(dimethylamino)-1,1-dioxo-4,5-dihydro-2h-1$l^{6}-benzothiepin-4-ol Chemical compound O[C@H]1C(CCCC)(CCCC)CS(=O)(=O)C2=CC=C(N(C)C)C=C2[C@H]1C(C=C1)=CC=C1OCC(C=C1)=CC=C1C[N+]1(CC2)CCN2CC1 STPKWKPURVSAJF-LJEWAXOPSA-N 0.000 description 2
- VUEGYUOUAAVYAS-JGGQBBKZSA-N (6ar,9s,10ar)-9-(dimethylsulfamoylamino)-7-methyl-6,6a,8,9,10,10a-hexahydro-4h-indolo[4,3-fg]quinoline Chemical compound C1=CC([C@H]2C[C@@H](CN(C)[C@@H]2C2)NS(=O)(=O)N(C)C)=C3C2=CNC3=C1 VUEGYUOUAAVYAS-JGGQBBKZSA-N 0.000 description 2
- DEVSOMFAQLZNKR-RJRFIUFISA-N (z)-3-[3-[3,5-bis(trifluoromethyl)phenyl]-1,2,4-triazol-1-yl]-n'-pyrazin-2-ylprop-2-enehydrazide Chemical compound FC(F)(F)C1=CC(C(F)(F)F)=CC(C2=NN(\C=C/C(=O)NNC=3N=CC=NC=3)C=N2)=C1 DEVSOMFAQLZNKR-RJRFIUFISA-N 0.000 description 2
- KZPYGQFFRCFCPP-UHFFFAOYSA-N 1,1'-bis(diphenylphosphino)ferrocene Chemical compound [Fe+2].C1=CC=C[C-]1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=C[C-]1P(C=1C=CC=CC=1)C1=CC=CC=C1 KZPYGQFFRCFCPP-UHFFFAOYSA-N 0.000 description 2
- WSLDOOZREJYCGB-UHFFFAOYSA-N 1,2-Dichloroethane Chemical compound ClCCCl WSLDOOZREJYCGB-UHFFFAOYSA-N 0.000 description 2
- KKHFRAFPESRGGD-UHFFFAOYSA-N 1,3-dimethyl-7-[3-(n-methylanilino)propyl]purine-2,6-dione Chemical compound C1=NC=2N(C)C(=O)N(C)C(=O)C=2N1CCCN(C)C1=CC=CC=C1 KKHFRAFPESRGGD-UHFFFAOYSA-N 0.000 description 2
- BFRCKOLRGYIRKE-UHFFFAOYSA-N 1,4-dichloro-2-[[4-(chloromethyl)phenoxy]methyl]benzene Chemical compound C1=CC(CCl)=CC=C1OCC1=CC(Cl)=CC=C1Cl BFRCKOLRGYIRKE-UHFFFAOYSA-N 0.000 description 2
- IBLMYGXJKQIGSN-UHFFFAOYSA-N 1-(bromomethyl)-2,4-difluorobenzene Chemical compound FC1=CC=C(CBr)C(F)=C1 IBLMYGXJKQIGSN-UHFFFAOYSA-N 0.000 description 2
- JDNPUJCKXLOHOW-UHFFFAOYSA-N 1-(bromomethyl)-4-(trifluoromethoxy)benzene Chemical compound FC(F)(F)OC1=CC=C(CBr)C=C1 JDNPUJCKXLOHOW-UHFFFAOYSA-N 0.000 description 2
- NVNPLEPBDPJYRZ-UHFFFAOYSA-N 1-(bromomethyl)-4-fluorobenzene Chemical compound FC1=CC=C(CBr)C=C1 NVNPLEPBDPJYRZ-UHFFFAOYSA-N 0.000 description 2
- HZQLUIZFUXNFHK-UHFFFAOYSA-N 1-(bromomethyl)-4-phenylbenzene Chemical group C1=CC(CBr)=CC=C1C1=CC=CC=C1 HZQLUIZFUXNFHK-UHFFFAOYSA-N 0.000 description 2
- QZNQSIHCDAGZIA-UHFFFAOYSA-N 1-(bromomethyl)-4-tert-butylbenzene Chemical compound CC(C)(C)C1=CC=C(CBr)C=C1 QZNQSIHCDAGZIA-UHFFFAOYSA-N 0.000 description 2
- OVWCURSGDICEQO-UHFFFAOYSA-N 1-(chloromethyl)-2-methyl-4-[4-(trifluoromethyl)phenyl]benzene Chemical group C1=C(CCl)C(C)=CC(C=2C=CC(=CC=2)C(F)(F)F)=C1 OVWCURSGDICEQO-UHFFFAOYSA-N 0.000 description 2
- BLWLFZYCZRJSPK-UHFFFAOYSA-N 1-(chloromethyl)-3-[3-(trifluoromethoxy)phenyl]benzene Chemical group FC(F)(F)OC1=CC=CC(C=2C=C(CCl)C=CC=2)=C1 BLWLFZYCZRJSPK-UHFFFAOYSA-N 0.000 description 2
- SOWUIXJDGINAKM-UHFFFAOYSA-N 1-(chloromethyl)-3-[4-(trifluoromethyl)phenyl]benzene Chemical group C1=CC(C(F)(F)F)=CC=C1C1=CC=CC(CCl)=C1 SOWUIXJDGINAKM-UHFFFAOYSA-N 0.000 description 2
- HABHGBOCWJYINP-UHFFFAOYSA-N 1-(chloromethyl)-4-(4-fluorophenyl)benzene Chemical group C1=CC(F)=CC=C1C1=CC=C(CCl)C=C1 HABHGBOCWJYINP-UHFFFAOYSA-N 0.000 description 2
- CTNBTUSBSPHZAX-UHFFFAOYSA-N 1-(chloromethyl)-4-[4-(trifluoromethyl)phenyl]benzene Chemical group C1=CC(C(F)(F)F)=CC=C1C1=CC=C(CCl)C=C1 CTNBTUSBSPHZAX-UHFFFAOYSA-N 0.000 description 2
- UYQPSKUPEXAQRJ-UHFFFAOYSA-N 1-(chloromethyl)-4-phenylmethoxybenzene Chemical compound C1=CC(CCl)=CC=C1OCC1=CC=CC=C1 UYQPSKUPEXAQRJ-UHFFFAOYSA-N 0.000 description 2
- LSESHKUFQCFLHB-QPJJXVBHSA-N 1-[(1e)-buta-1,3-dienyl]-2-methoxybenzene Chemical compound COC1=CC=CC=C1\C=C\C=C LSESHKUFQCFLHB-QPJJXVBHSA-N 0.000 description 2
- ONBQEOIKXPHGMB-VBSBHUPXSA-N 1-[2-[(2s,3r,4s,5r)-3,4-dihydroxy-5-(hydroxymethyl)oxolan-2-yl]oxy-4,6-dihydroxyphenyl]-3-(4-hydroxyphenyl)propan-1-one Chemical compound O[C@@H]1[C@H](O)[C@@H](CO)O[C@H]1OC1=CC(O)=CC(O)=C1C(=O)CCC1=CC=C(O)C=C1 ONBQEOIKXPHGMB-VBSBHUPXSA-N 0.000 description 2
- UNILWMWFPHPYOR-KXEYIPSPSA-M 1-[6-[2-[3-[3-[3-[2-[2-[3-[[2-[2-[[(2r)-1-[[2-[[(2r)-1-[3-[2-[2-[3-[[2-(2-amino-2-oxoethoxy)acetyl]amino]propoxy]ethoxy]ethoxy]propylamino]-3-hydroxy-1-oxopropan-2-yl]amino]-2-oxoethyl]amino]-3-[(2r)-2,3-di(hexadecanoyloxy)propyl]sulfanyl-1-oxopropan-2-yl Chemical compound O=C1C(SCCC(=O)NCCCOCCOCCOCCCNC(=O)COCC(=O)N[C@@H](CSC[C@@H](COC(=O)CCCCCCCCCCCCCCC)OC(=O)CCCCCCCCCCCCCCC)C(=O)NCC(=O)N[C@H](CO)C(=O)NCCCOCCOCCOCCCNC(=O)COCC(N)=O)CC(=O)N1CCNC(=O)CCCCCN\1C2=CC=C(S([O-])(=O)=O)C=C2CC/1=C/C=C/C=C/C1=[N+](CC)C2=CC=C(S([O-])(=O)=O)C=C2C1 UNILWMWFPHPYOR-KXEYIPSPSA-M 0.000 description 2
- QXOGPTXQGKQSJT-UHFFFAOYSA-N 1-amino-4-[4-(3,4-dimethylphenyl)sulfanylanilino]-9,10-dioxoanthracene-2-sulfonic acid Chemical compound Cc1ccc(Sc2ccc(Nc3cc(c(N)c4C(=O)c5ccccc5C(=O)c34)S(O)(=O)=O)cc2)cc1C QXOGPTXQGKQSJT-UHFFFAOYSA-N 0.000 description 2
- WVUDHWBCPSXAFN-UHFFFAOYSA-N 1-bromo-3-(trifluoromethoxy)benzene Chemical compound FC(F)(F)OC1=CC=CC(Br)=C1 WVUDHWBCPSXAFN-UHFFFAOYSA-N 0.000 description 2
- ASJCVVGLGXVRGQ-UHFFFAOYSA-N 1-butyl-2-methoxybenzene Chemical compound CCCCC1=CC=CC=C1OC ASJCVVGLGXVRGQ-UHFFFAOYSA-N 0.000 description 2
- VBICKXHEKHSIBG-UHFFFAOYSA-N 1-monostearoylglycerol Chemical compound CCCCCCCCCCCCCCCCCC(=O)OCC(O)CO VBICKXHEKHSIBG-UHFFFAOYSA-N 0.000 description 2
- IRSVDHPYXFLLDS-UHFFFAOYSA-N 2,4-dichloro-1-(chloromethyl)benzene Chemical compound ClCC1=CC=C(Cl)C=C1Cl IRSVDHPYXFLLDS-UHFFFAOYSA-N 0.000 description 2
- NXXYKOUNUYWIHA-UHFFFAOYSA-N 2,6-Dimethylphenol Chemical compound CC1=CC=CC(C)=C1O NXXYKOUNUYWIHA-UHFFFAOYSA-N 0.000 description 2
- WGFNXGPBPIJYLI-UHFFFAOYSA-N 2,6-difluoro-3-[(3-fluorophenyl)sulfonylamino]-n-(3-methoxy-1h-pyrazolo[3,4-b]pyridin-5-yl)benzamide Chemical compound C1=C2C(OC)=NNC2=NC=C1NC(=O)C(C=1F)=C(F)C=CC=1NS(=O)(=O)C1=CC=CC(F)=C1 WGFNXGPBPIJYLI-UHFFFAOYSA-N 0.000 description 2
- FQMZXMVHHKXGTM-UHFFFAOYSA-N 2-(1-adamantyl)-n-[2-[2-(2-hydroxyethylamino)ethylamino]quinolin-5-yl]acetamide Chemical compound C1C(C2)CC(C3)CC2CC13CC(=O)NC1=CC=CC2=NC(NCCNCCO)=CC=C21 FQMZXMVHHKXGTM-UHFFFAOYSA-N 0.000 description 2
- VCUXVXLUOHDHKK-UHFFFAOYSA-N 2-(2-aminopyrimidin-4-yl)-4-(2-chloro-4-methoxyphenyl)-1,3-thiazole-5-carboxamide Chemical compound ClC1=CC(OC)=CC=C1C1=C(C(N)=O)SC(C=2N=C(N)N=CC=2)=N1 VCUXVXLUOHDHKK-UHFFFAOYSA-N 0.000 description 2
- QEBYEVQKHRUYPE-UHFFFAOYSA-N 2-(2-chlorophenyl)-5-[(1-methylpyrazol-3-yl)methyl]-4-[[methyl(pyridin-3-ylmethyl)amino]methyl]-1h-pyrazolo[4,3-c]pyridine-3,6-dione Chemical compound C1=CN(C)N=C1CN1C(=O)C=C2NN(C=3C(=CC=CC=3)Cl)C(=O)C2=C1CN(C)CC1=CC=CN=C1 QEBYEVQKHRUYPE-UHFFFAOYSA-N 0.000 description 2
- WZUODJNEIXSNEU-UHFFFAOYSA-N 2-Hydroxy-4-methoxybenzaldehyde Chemical compound COC1=CC=C(C=O)C(O)=C1 WZUODJNEIXSNEU-UHFFFAOYSA-N 0.000 description 2
- NWEHHVVXCHVPFQ-UHFFFAOYSA-N 2-[5-methoxy-2-methyl-4-[[4-[5-(trifluoromethyl)pyridin-2-yl]phenyl]methylsulfanyl]phenoxy]acetic acid Chemical compound COC1=CC(OCC(O)=O)=C(C)C=C1SCC1=CC=C(C=2N=CC(=CC=2)C(F)(F)F)C=C1 NWEHHVVXCHVPFQ-UHFFFAOYSA-N 0.000 description 2
- PYRKKGOKRMZEIT-UHFFFAOYSA-N 2-[6-(2-cyclopropylethoxy)-9-(2-hydroxy-2-methylpropyl)-1h-phenanthro[9,10-d]imidazol-2-yl]-5-fluorobenzene-1,3-dicarbonitrile Chemical compound C1=C2C3=CC(CC(C)(O)C)=CC=C3C=3NC(C=4C(=CC(F)=CC=4C#N)C#N)=NC=3C2=CC=C1OCCC1CC1 PYRKKGOKRMZEIT-UHFFFAOYSA-N 0.000 description 2
- FMKGJQHNYMWDFJ-CVEARBPZSA-N 2-[[4-(2,2-difluoropropoxy)pyrimidin-5-yl]methylamino]-4-[[(1R,4S)-4-hydroxy-3,3-dimethylcyclohexyl]amino]pyrimidine-5-carbonitrile Chemical compound FC(COC1=NC=NC=C1CNC1=NC=C(C(=N1)N[C@H]1CC([C@H](CC1)O)(C)C)C#N)(C)F FMKGJQHNYMWDFJ-CVEARBPZSA-N 0.000 description 2
- KSQOBVUINFBJNJ-UHFFFAOYSA-N 2-[[7-[[3,5-dichloro-4-[[4-(trifluoromethyl)phenyl]methoxy]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetic acid Chemical compound C1=2CCCC=2C(OCC(=O)O)=CC=C1SCC(C=C1Cl)=CC(Cl)=C1OCC1=CC=C(C(F)(F)F)C=C1 KSQOBVUINFBJNJ-UHFFFAOYSA-N 0.000 description 2
- VVCMGAUPZIKYTH-VGHSCWAPSA-N 2-acetyloxybenzoic acid;[(2s,3r)-4-(dimethylamino)-3-methyl-1,2-diphenylbutan-2-yl] propanoate;1,3,7-trimethylpurine-2,6-dione Chemical compound CC(=O)OC1=CC=CC=C1C(O)=O.CN1C(=O)N(C)C(=O)C2=C1N=CN2C.C([C@](OC(=O)CC)([C@H](C)CN(C)C)C=1C=CC=CC=1)C1=CC=CC=C1 VVCMGAUPZIKYTH-VGHSCWAPSA-N 0.000 description 2
- YSUIQYOGTINQIN-UZFYAQMZSA-N 2-amino-9-[(1S,6R,8R,9S,10R,15R,17R,18R)-8-(6-aminopurin-9-yl)-9,18-difluoro-3,12-dihydroxy-3,12-bis(sulfanylidene)-2,4,7,11,13,16-hexaoxa-3lambda5,12lambda5-diphosphatricyclo[13.2.1.06,10]octadecan-17-yl]-1H-purin-6-one Chemical compound NC1=NC2=C(N=CN2[C@@H]2O[C@@H]3COP(S)(=O)O[C@@H]4[C@@H](COP(S)(=O)O[C@@H]2[C@@H]3F)O[C@H]([C@H]4F)N2C=NC3=C2N=CN=C3N)C(=O)N1 YSUIQYOGTINQIN-UZFYAQMZSA-N 0.000 description 2
- TVTJUIAKQFIXCE-HUKYDQBMSA-N 2-amino-9-[(2R,3S,4S,5R)-4-fluoro-3-hydroxy-5-(hydroxymethyl)oxolan-2-yl]-7-prop-2-ynyl-1H-purine-6,8-dione Chemical compound NC=1NC(C=2N(C(N(C=2N=1)[C@@H]1O[C@@H]([C@H]([C@H]1O)F)CO)=O)CC#C)=O TVTJUIAKQFIXCE-HUKYDQBMSA-N 0.000 description 2
- JFZJMSDDOOAOIV-UHFFFAOYSA-N 2-chloro-5-(trifluoromethyl)pyridine Chemical compound FC(F)(F)C1=CC=C(Cl)N=C1 JFZJMSDDOOAOIV-UHFFFAOYSA-N 0.000 description 2
- LFOIDLOIBZFWDO-UHFFFAOYSA-N 2-methoxy-6-[6-methoxy-4-[(3-phenylmethoxyphenyl)methoxy]-1-benzofuran-2-yl]imidazo[2,1-b][1,3,4]thiadiazole Chemical compound N1=C2SC(OC)=NN2C=C1C(OC1=CC(OC)=C2)=CC1=C2OCC(C=1)=CC=CC=1OCC1=CC=CC=C1 LFOIDLOIBZFWDO-UHFFFAOYSA-N 0.000 description 2
- DFRAKBCRUYUFNT-UHFFFAOYSA-N 3,8-dicyclohexyl-2,4,7,9-tetrahydro-[1,3]oxazino[5,6-h][1,3]benzoxazine Chemical compound C1CCCCC1N1CC(C=CC2=C3OCN(C2)C2CCCCC2)=C3OC1 DFRAKBCRUYUFNT-UHFFFAOYSA-N 0.000 description 2
- WFOVEDJTASPCIR-UHFFFAOYSA-N 3-[(4-methyl-5-pyridin-4-yl-1,2,4-triazol-3-yl)methylamino]-n-[[2-(trifluoromethyl)phenyl]methyl]benzamide Chemical compound N=1N=C(C=2C=CN=CC=2)N(C)C=1CNC(C=1)=CC=CC=1C(=O)NCC1=CC=CC=C1C(F)(F)F WFOVEDJTASPCIR-UHFFFAOYSA-N 0.000 description 2
- NHQDETIJWKXCTC-UHFFFAOYSA-N 3-chloroperbenzoic acid Chemical compound OOC(=O)C1=CC=CC(Cl)=C1 NHQDETIJWKXCTC-UHFFFAOYSA-N 0.000 description 2
- OKVJCVWFVRATSG-UHFFFAOYSA-N 3-hydroxybenzyl alcohol Chemical compound OCC1=CC=CC(O)=C1 OKVJCVWFVRATSG-UHFFFAOYSA-N 0.000 description 2
- ASHGTJPOSUFTGB-UHFFFAOYSA-N 3-methoxyphenol Chemical compound COC1=CC=CC(O)=C1 ASHGTJPOSUFTGB-UHFFFAOYSA-N 0.000 description 2
- LZLIPLUATRVXSB-UHFFFAOYSA-N 4-(bromomethyl)-1-chloro-2-(trifluoromethyl)benzene Chemical compound FC(F)(F)C1=CC(CBr)=CC=C1Cl LZLIPLUATRVXSB-UHFFFAOYSA-N 0.000 description 2
- FMRRFPFZGPDDLZ-UHFFFAOYSA-N 4-Thiocyanatophenol Chemical compound OC1=CC=C(SC#N)C=C1 FMRRFPFZGPDDLZ-UHFFFAOYSA-N 0.000 description 2
- UIADWSXPNFQQCZ-UHFFFAOYSA-N 4-[4-(3,4-dichlorophenyl)-5-phenyl-1,3-oxazol-2-yl]butanoic acid Chemical compound ClC=1C=C(C=CC=1Cl)C=1N=C(OC=1C1=CC=CC=C1)CCCC(=O)O UIADWSXPNFQQCZ-UHFFFAOYSA-N 0.000 description 2
- WYFCZWSWFGJODV-MIANJLSGSA-N 4-[[(1s)-2-[(e)-3-[3-chloro-2-fluoro-6-(tetrazol-1-yl)phenyl]prop-2-enoyl]-5-(4-methyl-2-oxopiperazin-1-yl)-3,4-dihydro-1h-isoquinoline-1-carbonyl]amino]benzoic acid Chemical compound O=C1CN(C)CCN1C1=CC=CC2=C1CCN(C(=O)\C=C\C=1C(=CC=C(Cl)C=1F)N1N=NN=C1)[C@@H]2C(=O)NC1=CC=C(C(O)=O)C=C1 WYFCZWSWFGJODV-MIANJLSGSA-N 0.000 description 2
- MPMKMQHJHDHPBE-RUZDIDTESA-N 4-[[(2r)-1-(1-benzothiophene-3-carbonyl)-2-methylazetidine-2-carbonyl]-[(3-chlorophenyl)methyl]amino]butanoic acid Chemical compound O=C([C@@]1(N(CC1)C(=O)C=1C2=CC=CC=C2SC=1)C)N(CCCC(O)=O)CC1=CC=CC(Cl)=C1 MPMKMQHJHDHPBE-RUZDIDTESA-N 0.000 description 2
- 125000001054 5 membered carbocyclic group Chemical group 0.000 description 2
- WHKRHBLAJFYZKF-UHFFFAOYSA-N 5-(hydroxymethyl)-2-methoxyphenol Chemical compound COC1=CC=C(CO)C=C1O WHKRHBLAJFYZKF-UHFFFAOYSA-N 0.000 description 2
- VKLKXFOZNHEBSW-UHFFFAOYSA-N 5-[[3-[(4-morpholin-4-ylbenzoyl)amino]phenyl]methoxy]pyridine-3-carboxamide Chemical compound O1CCN(CC1)C1=CC=C(C(=O)NC=2C=C(COC=3C=NC=C(C(=O)N)C=3)C=CC=2)C=C1 VKLKXFOZNHEBSW-UHFFFAOYSA-N 0.000 description 2
- XFJBGINZIMNZBW-CRAIPNDOSA-N 5-chloro-2-[4-[(1r,2s)-2-[2-(5-methylsulfonylpyridin-2-yl)oxyethyl]cyclopropyl]piperidin-1-yl]pyrimidine Chemical compound N1=CC(S(=O)(=O)C)=CC=C1OCC[C@H]1[C@@H](C2CCN(CC2)C=2N=CC(Cl)=CN=2)C1 XFJBGINZIMNZBW-CRAIPNDOSA-N 0.000 description 2
- RSIWALKZYXPAGW-NSHDSACASA-N 6-(3-fluorophenyl)-3-methyl-7-[(1s)-1-(7h-purin-6-ylamino)ethyl]-[1,3]thiazolo[3,2-a]pyrimidin-5-one Chemical compound C=1([C@@H](NC=2C=3N=CNC=3N=CN=2)C)N=C2SC=C(C)N2C(=O)C=1C1=CC=CC(F)=C1 RSIWALKZYXPAGW-NSHDSACASA-N 0.000 description 2
- GDUANFXPOZTYKS-UHFFFAOYSA-N 6-bromo-8-[(2,6-difluoro-4-methoxybenzoyl)amino]-4-oxochromene-2-carboxylic acid Chemical compound FC1=CC(OC)=CC(F)=C1C(=O)NC1=CC(Br)=CC2=C1OC(C(O)=O)=CC2=O GDUANFXPOZTYKS-UHFFFAOYSA-N 0.000 description 2
- HCCNBKFJYUWLEX-UHFFFAOYSA-N 7-(6-methoxypyridin-3-yl)-1-(2-propoxyethyl)-3-(pyrazin-2-ylmethylamino)pyrido[3,4-b]pyrazin-2-one Chemical compound O=C1N(CCOCCC)C2=CC(C=3C=NC(OC)=CC=3)=NC=C2N=C1NCC1=CN=CC=N1 HCCNBKFJYUWLEX-UHFFFAOYSA-N 0.000 description 2
- XASOHFCUIQARJT-UHFFFAOYSA-N 8-methoxy-6-[7-(2-morpholin-4-ylethoxy)imidazo[1,2-a]pyridin-3-yl]-2-(2,2,2-trifluoroethyl)-3,4-dihydroisoquinolin-1-one Chemical compound C(N1C(=O)C2=C(OC)C=C(C=3N4C(=NC=3)C=C(C=C4)OCCN3CCOCC3)C=C2CC1)C(F)(F)F XASOHFCUIQARJT-UHFFFAOYSA-N 0.000 description 2
- 229920001817 Agar Polymers 0.000 description 2
- GUBGYTABKSRVRQ-XLOQQCSPSA-N Alpha-Lactose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-XLOQQCSPSA-N 0.000 description 2
- NLXLAEXVIDQMFP-UHFFFAOYSA-N Ammonia chloride Chemical compound [NH4+].[Cl-] NLXLAEXVIDQMFP-UHFFFAOYSA-N 0.000 description 2
- QGZKDVFQNNGYKY-UHFFFAOYSA-O Ammonium Chemical compound [NH4+] QGZKDVFQNNGYKY-UHFFFAOYSA-O 0.000 description 2
- BTBUEUYNUDRHOZ-UHFFFAOYSA-N Borate Chemical compound [O-]B([O-])[O-] BTBUEUYNUDRHOZ-UHFFFAOYSA-N 0.000 description 2
- JQUCWIWWWKZNCS-LESHARBVSA-N C(C1=CC=CC=C1)(=O)NC=1SC[C@H]2[C@@](N1)(CO[C@H](C2)C)C=2SC=C(N2)NC(=O)C2=NC=C(C=C2)OC(F)F Chemical compound C(C1=CC=CC=C1)(=O)NC=1SC[C@H]2[C@@](N1)(CO[C@H](C2)C)C=2SC=C(N2)NC(=O)C2=NC=C(C=C2)OC(F)F JQUCWIWWWKZNCS-LESHARBVSA-N 0.000 description 2
- OJRUSAPKCPIVBY-KQYNXXCUSA-N C1=NC2=C(N=C(N=C2N1[C@H]3[C@@H]([C@@H]([C@H](O3)COP(=O)(CP(=O)(O)O)O)O)O)I)N Chemical compound C1=NC2=C(N=C(N=C2N1[C@H]3[C@@H]([C@@H]([C@H](O3)COP(=O)(CP(=O)(O)O)O)O)O)I)N OJRUSAPKCPIVBY-KQYNXXCUSA-N 0.000 description 2
- NBWLIMHTVLIDAS-UHFFFAOYSA-N CC1=C(CCC(=O)O)C=CC(SCC2=CC=C(OCC3=CC=C(F)C=C3)C=C2)=C1 Chemical compound CC1=C(CCC(=O)O)C=CC(SCC2=CC=C(OCC3=CC=C(F)C=C3)C=C2)=C1 NBWLIMHTVLIDAS-UHFFFAOYSA-N 0.000 description 2
- NMFDJMWYDRUEFH-UHFFFAOYSA-N CC1=C(O)C=C(Cl)C(N=C=S)=C1 Chemical compound CC1=C(O)C=C(Cl)C(N=C=S)=C1 NMFDJMWYDRUEFH-UHFFFAOYSA-N 0.000 description 2
- QFPAICITLFUOQP-UHFFFAOYSA-N CC1=C(OCC(=O)O)C=CC(SCC2=CC=C(OCC3=C(Cl)C=CC(Cl)=C3)C=C2)=C1 Chemical compound CC1=C(OCC(=O)O)C=CC(SCC2=CC=C(OCC3=C(Cl)C=CC(Cl)=C3)C=C2)=C1 QFPAICITLFUOQP-UHFFFAOYSA-N 0.000 description 2
- KCBAMQOKOLXLOX-BSZYMOERSA-N CC1=C(SC=N1)C2=CC=C(C=C2)[C@H](C)NC(=O)[C@@H]3C[C@H](CN3C(=O)[C@H](C(C)(C)C)NC(=O)CCCCCCCCCCNCCCONC(=O)C4=C(C(=C(C=C4)F)F)NC5=C(C=C(C=C5)I)F)O Chemical compound CC1=C(SC=N1)C2=CC=C(C=C2)[C@H](C)NC(=O)[C@@H]3C[C@H](CN3C(=O)[C@H](C(C)(C)C)NC(=O)CCCCCCCCCCNCCCONC(=O)C4=C(C(=C(C=C4)F)F)NC5=C(C=C(C=C5)I)F)O KCBAMQOKOLXLOX-BSZYMOERSA-N 0.000 description 2
- BQXUPNKLZNSUMC-YUQWMIPFSA-N CCN(CCCCCOCC(=O)N[C@H](C(=O)N1C[C@H](O)C[C@H]1C(=O)N[C@@H](C)c1ccc(cc1)-c1scnc1C)C(C)(C)C)CCOc1ccc(cc1)C(=O)c1c(sc2cc(O)ccc12)-c1ccc(O)cc1 Chemical compound CCN(CCCCCOCC(=O)N[C@H](C(=O)N1C[C@H](O)C[C@H]1C(=O)N[C@@H](C)c1ccc(cc1)-c1scnc1C)C(C)(C)C)CCOc1ccc(cc1)C(=O)c1c(sc2cc(O)ccc12)-c1ccc(O)cc1 BQXUPNKLZNSUMC-YUQWMIPFSA-N 0.000 description 2
- AFDHECXZDCGDCN-JXMROGBWSA-N COC(=O)COC1=C(C)C=C(SCC2=CC=C(/C=C/C3=CC=CC(F)=C3)C=C2)C(OC)=C1 Chemical compound COC(=O)COC1=C(C)C=C(SCC2=CC=C(/C=C/C3=CC=CC(F)=C3)C=C2)C(OC)=C1 AFDHECXZDCGDCN-JXMROGBWSA-N 0.000 description 2
- NWVDDPALSKLNPU-UHFFFAOYSA-N COC(=O)COC1=C2CCCC2=C(SCC2=CC(C3=CC=C(Cl)C=C3)=ON2)C=C1 Chemical compound COC(=O)COC1=C2CCCC2=C(SCC2=CC(C3=CC=C(Cl)C=C3)=ON2)C=C1 NWVDDPALSKLNPU-UHFFFAOYSA-N 0.000 description 2
- SVZWWXBNHLFXPT-UHFFFAOYSA-N COC(=O)COC1=CC=C(SCC2=CC=C(OCC3=C(C(F)(F)F)C=C(F)C=C3)C=C2)C2=C1CCC2 Chemical compound COC(=O)COC1=CC=C(SCC2=CC=C(OCC3=C(C(F)(F)F)C=C(F)C=C3)C=C2)C2=C1CCC2 SVZWWXBNHLFXPT-UHFFFAOYSA-N 0.000 description 2
- AKRYKRQSQLDDLP-UHFFFAOYSA-N COC1=CC(O)=C(C)C=C1N=C=S Chemical compound COC1=CC(O)=C(C)C=C1N=C=S AKRYKRQSQLDDLP-UHFFFAOYSA-N 0.000 description 2
- QPQVDUHBPMLTTD-RMKNXTFCSA-N COC1=CC(OCC(=O)O)=C(C)C=C1SCC1=CC=C(/C=C/C2=CC=CC(F)=C2)C=C1 Chemical compound COC1=CC(OCC(=O)O)=C(C)C=C1SCC1=CC=C(/C=C/C2=CC=CC(F)=C2)C=C1 QPQVDUHBPMLTTD-RMKNXTFCSA-N 0.000 description 2
- PKMUHQIDVVOXHQ-HXUWFJFHSA-N C[C@H](C1=CC(C2=CC=C(CNC3CCCC3)S2)=CC=C1)NC(C1=C(C)C=CC(NC2CNC2)=C1)=O Chemical compound C[C@H](C1=CC(C2=CC=C(CNC3CCCC3)S2)=CC=C1)NC(C1=C(C)C=CC(NC2CNC2)=C1)=O PKMUHQIDVVOXHQ-HXUWFJFHSA-N 0.000 description 2
- VTYYLEPIZMXCLO-UHFFFAOYSA-L Calcium carbonate Chemical compound [Ca+2].[O-]C([O-])=O VTYYLEPIZMXCLO-UHFFFAOYSA-L 0.000 description 2
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical group [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 2
- 229940126657 Compound 17 Drugs 0.000 description 2
- 229940126639 Compound 33 Drugs 0.000 description 2
- 229940127007 Compound 39 Drugs 0.000 description 2
- ROSDSFDQCJNGOL-UHFFFAOYSA-N Dimethylamine Chemical compound CNC ROSDSFDQCJNGOL-UHFFFAOYSA-N 0.000 description 2
- QUSNBJAOOMFDIB-UHFFFAOYSA-N Ethylamine Chemical compound CCN QUSNBJAOOMFDIB-UHFFFAOYSA-N 0.000 description 2
- 108010010803 Gelatin Proteins 0.000 description 2
- 229940121710 HMGCoA reductase inhibitor Drugs 0.000 description 2
- WTDHULULXKLSOZ-UHFFFAOYSA-N Hydroxylamine hydrochloride Chemical compound Cl.ON WTDHULULXKLSOZ-UHFFFAOYSA-N 0.000 description 2
- 206010020772 Hypertension Diseases 0.000 description 2
- 239000012097 Lipofectamine 2000 Substances 0.000 description 2
- FEIOASZZURHTHB-UHFFFAOYSA-N Methyl-p-formylbenzoate Natural products COC(=O)C1=CC=C(C=O)C=C1 FEIOASZZURHTHB-UHFFFAOYSA-N 0.000 description 2
- BAVYZALUXZFZLV-UHFFFAOYSA-N Methylamine Chemical compound NC BAVYZALUXZFZLV-UHFFFAOYSA-N 0.000 description 2
- 241000699670 Mus sp. Species 0.000 description 2
- FFDGPVCHZBVARC-UHFFFAOYSA-N N,N-dimethylglycine Chemical compound CN(C)CC(O)=O FFDGPVCHZBVARC-UHFFFAOYSA-N 0.000 description 2
- MZRVEZGGRBJDDB-UHFFFAOYSA-N N-Butyllithium Chemical compound [Li]CCCC MZRVEZGGRBJDDB-UHFFFAOYSA-N 0.000 description 2
- SECXISVLQFMRJM-UHFFFAOYSA-N N-Methylpyrrolidone Chemical compound CN1CCCC1=O SECXISVLQFMRJM-UHFFFAOYSA-N 0.000 description 2
- LVDRREOUMKACNJ-BKMJKUGQSA-N N-[(2R,3S)-2-(4-chlorophenyl)-1-(1,4-dimethyl-2-oxoquinolin-7-yl)-6-oxopiperidin-3-yl]-2-methylpropane-1-sulfonamide Chemical compound CC(C)CS(=O)(=O)N[C@H]1CCC(=O)N([C@@H]1c1ccc(Cl)cc1)c1ccc2c(C)cc(=O)n(C)c2c1 LVDRREOUMKACNJ-BKMJKUGQSA-N 0.000 description 2
- AVYVHIKSFXVDBG-UHFFFAOYSA-N N-benzyl-N-hydroxy-2,2-dimethylbutanamide Chemical compound C(C1=CC=CC=C1)N(C(C(CC)(C)C)=O)O AVYVHIKSFXVDBG-UHFFFAOYSA-N 0.000 description 2
- OPFJDXRVMFKJJO-ZHHKINOHSA-N N-{[3-(2-benzamido-4-methyl-1,3-thiazol-5-yl)-pyrazol-5-yl]carbonyl}-G-dR-G-dD-dD-dD-NH2 Chemical compound S1C(C=2NN=C(C=2)C(=O)NCC(=O)N[C@H](CCCN=C(N)N)C(=O)NCC(=O)N[C@H](CC(O)=O)C(=O)N[C@H](CC(O)=O)C(=O)N[C@H](CC(O)=O)C(N)=O)=C(C)N=C1NC(=O)C1=CC=CC=C1 OPFJDXRVMFKJJO-ZHHKINOHSA-N 0.000 description 2
- QOVYHDHLFPKQQG-NDEPHWFRSA-N N[C@@H](CCC(=O)N1CCC(CC1)NC1=C2C=CC=CC2=NC(NCC2=CN(CCCNCCCNC3CCCCC3)N=N2)=N1)C(O)=O Chemical compound N[C@@H](CCC(=O)N1CCC(CC1)NC1=C2C=CC=CC2=NC(NCC2=CN(CCCNCCCNC3CCCCC3)N=N2)=N1)C(O)=O QOVYHDHLFPKQQG-NDEPHWFRSA-N 0.000 description 2
- AXULCJXNLKWOCT-UHFFFAOYSA-N O=C(O)COC1=CC=C(SCC2=CC=C(OCC3=CC=C(F)C=C3)C=C2)C2=C1CCC2 Chemical compound O=C(O)COC1=CC=C(SCC2=CC=C(OCC3=CC=C(F)C=C3)C=C2)C2=C1CCC2 AXULCJXNLKWOCT-UHFFFAOYSA-N 0.000 description 2
- 239000012124 Opti-MEM Substances 0.000 description 2
- 108010028924 PPAR alpha Proteins 0.000 description 2
- GLUUGHFHXGJENI-UHFFFAOYSA-N Piperazine Chemical group C1CNCCN1 GLUUGHFHXGJENI-UHFFFAOYSA-N 0.000 description 2
- NQRYJNQNLNOLGT-UHFFFAOYSA-N Piperidine Chemical group C1CCNCC1 NQRYJNQNLNOLGT-UHFFFAOYSA-N 0.000 description 2
- 239000002202 Polyethylene glycol Substances 0.000 description 2
- 108700008625 Reporter Genes Proteins 0.000 description 2
- PNUZDKCDAWUEGK-CYZMBNFOSA-N Sitafloxacin Chemical compound C([C@H]1N)N(C=2C(=C3C(C(C(C(O)=O)=CN3[C@H]3[C@H](C3)F)=O)=CC=2F)Cl)CC11CC1 PNUZDKCDAWUEGK-CYZMBNFOSA-N 0.000 description 2
- KEAYESYHFKHZAL-UHFFFAOYSA-N Sodium Chemical compound [Na] KEAYESYHFKHZAL-UHFFFAOYSA-N 0.000 description 2
- AWMVMTVKBNGEAK-UHFFFAOYSA-N Styrene oxide Chemical compound C1OC1C1=CC=CC=C1 AWMVMTVKBNGEAK-UHFFFAOYSA-N 0.000 description 2
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 2
- 229930006000 Sucrose Natural products 0.000 description 2
- 229940100389 Sulfonylurea Drugs 0.000 description 2
- ZMZDMBWJUHKJPS-UHFFFAOYSA-M Thiocyanate anion Chemical compound [S-]C#N ZMZDMBWJUHKJPS-UHFFFAOYSA-M 0.000 description 2
- LJOOWESTVASNOG-UFJKPHDISA-N [(1s,3r,4ar,7s,8s,8as)-3-hydroxy-8-[2-[(4r)-4-hydroxy-6-oxooxan-2-yl]ethyl]-7-methyl-1,2,3,4,4a,7,8,8a-octahydronaphthalen-1-yl] (2s)-2-methylbutanoate Chemical compound C([C@H]1[C@@H](C)C=C[C@H]2C[C@@H](O)C[C@@H]([C@H]12)OC(=O)[C@@H](C)CC)CC1C[C@@H](O)CC(=O)O1 LJOOWESTVASNOG-UFJKPHDISA-N 0.000 description 2
- SPXSEZMVRJLHQG-XMMPIXPASA-N [(2R)-1-[[4-[(3-phenylmethoxyphenoxy)methyl]phenyl]methyl]pyrrolidin-2-yl]methanol Chemical compound C(C1=CC=CC=C1)OC=1C=C(OCC2=CC=C(CN3[C@H](CCC3)CO)C=C2)C=CC=1 SPXSEZMVRJLHQG-XMMPIXPASA-N 0.000 description 2
- IOSLINNLJFQMFF-XMMPIXPASA-N [(2R)-1-[[4-[[3-[(4-fluorophenyl)methylsulfanyl]phenoxy]methyl]phenyl]methyl]pyrrolidin-2-yl]methanol Chemical compound FC1=CC=C(CSC=2C=C(OCC3=CC=C(CN4[C@H](CCC4)CO)C=C3)C=CC=2)C=C1 IOSLINNLJFQMFF-XMMPIXPASA-N 0.000 description 2
- LNUFLCYMSVYYNW-ZPJMAFJPSA-N [(2r,3r,4s,5r,6r)-2-[(2r,3r,4s,5r,6r)-6-[(2r,3r,4s,5r,6r)-6-[(2r,3r,4s,5r,6r)-6-[[(3s,5s,8r,9s,10s,13r,14s,17r)-10,13-dimethyl-17-[(2r)-6-methylheptan-2-yl]-2,3,4,5,6,7,8,9,11,12,14,15,16,17-tetradecahydro-1h-cyclopenta[a]phenanthren-3-yl]oxy]-4,5-disulfo Chemical compound O([C@@H]1[C@@H](COS(O)(=O)=O)O[C@@H]([C@@H]([C@H]1OS(O)(=O)=O)OS(O)(=O)=O)O[C@@H]1[C@@H](COS(O)(=O)=O)O[C@@H]([C@@H]([C@H]1OS(O)(=O)=O)OS(O)(=O)=O)O[C@@H]1[C@@H](COS(O)(=O)=O)O[C@H]([C@@H]([C@H]1OS(O)(=O)=O)OS(O)(=O)=O)O[C@@H]1C[C@@H]2CC[C@H]3[C@@H]4CC[C@@H]([C@]4(CC[C@@H]3[C@@]2(C)CC1)C)[C@H](C)CCCC(C)C)[C@H]1O[C@H](COS(O)(=O)=O)[C@@H](OS(O)(=O)=O)[C@H](OS(O)(=O)=O)[C@H]1OS(O)(=O)=O LNUFLCYMSVYYNW-ZPJMAFJPSA-N 0.000 description 2
- PSLUFJFHTBIXMW-WYEYVKMPSA-N [(3r,4ar,5s,6s,6as,10s,10ar,10bs)-3-ethenyl-10,10b-dihydroxy-3,4a,7,7,10a-pentamethyl-1-oxo-6-(2-pyridin-2-ylethylcarbamoyloxy)-5,6,6a,8,9,10-hexahydro-2h-benzo[f]chromen-5-yl] acetate Chemical compound O([C@@H]1[C@@H]([C@]2(O[C@](C)(CC(=O)[C@]2(O)[C@@]2(C)[C@@H](O)CCC(C)(C)[C@@H]21)C=C)C)OC(=O)C)C(=O)NCCC1=CC=CC=N1 PSLUFJFHTBIXMW-WYEYVKMPSA-N 0.000 description 2
- OCFOSIUUSQYWLW-UHFFFAOYSA-N [3,5-dichloro-2-[[4-(trifluoromethyl)phenyl]methoxy]phenyl]methanol Chemical compound OCC1=CC(Cl)=CC(Cl)=C1OCC1=CC=C(C(F)(F)F)C=C1 OCFOSIUUSQYWLW-UHFFFAOYSA-N 0.000 description 2
- OKRCXSCDWKJWSW-UHFFFAOYSA-N [4-(chloromethyl)phenyl] acetate Chemical compound CC(=O)OC1=CC=C(CCl)C=C1 OKRCXSCDWKJWSW-UHFFFAOYSA-N 0.000 description 2
- PZRPBPMLSSNFOM-UHFFFAOYSA-N [4-(hydroxymethyl)phenyl]boronic acid Chemical compound OCC1=CC=C(B(O)O)C=C1 PZRPBPMLSSNFOM-UHFFFAOYSA-N 0.000 description 2
- XMOLAFPVDQWPNV-UHFFFAOYSA-N [4-[2-(3-fluorophenyl)ethenyl]phenyl]methanol Chemical compound C1=CC(CO)=CC=C1C=CC1=CC=CC(F)=C1 XMOLAFPVDQWPNV-UHFFFAOYSA-N 0.000 description 2
- SMNRFWMNPDABKZ-WVALLCKVSA-N [[(2R,3S,4R,5S)-5-(2,6-dioxo-3H-pyridin-3-yl)-3,4-dihydroxyoxolan-2-yl]methoxy-hydroxyphosphoryl] [[[(2R,3S,4S,5R,6R)-4-fluoro-3,5-dihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy-hydroxyphosphoryl]oxy-hydroxyphosphoryl] hydrogen phosphate Chemical compound OC[C@H]1O[C@H](OP(O)(=O)OP(O)(=O)OP(O)(=O)OP(O)(=O)OC[C@H]2O[C@H]([C@H](O)[C@@H]2O)C2C=CC(=O)NC2=O)[C@H](O)[C@@H](F)[C@@H]1O SMNRFWMNPDABKZ-WVALLCKVSA-N 0.000 description 2
- 238000010521 absorption reaction Methods 0.000 description 2
- 230000002378 acidificating effect Effects 0.000 description 2
- 230000009471 action Effects 0.000 description 2
- 239000002671 adjuvant Substances 0.000 description 2
- 235000010419 agar Nutrition 0.000 description 2
- 239000000556 agonist Substances 0.000 description 2
- 235000010443 alginic acid Nutrition 0.000 description 2
- 229920000615 alginic acid Polymers 0.000 description 2
- 229910052784 alkaline earth metal Inorganic materials 0.000 description 2
- 239000000883 anti-obesity agent Substances 0.000 description 2
- 229940125708 antidiabetic agent Drugs 0.000 description 2
- 239000003524 antilipemic agent Substances 0.000 description 2
- 229940125710 antiobesity agent Drugs 0.000 description 2
- 150000001543 aryl boronic acids Chemical class 0.000 description 2
- XRWSZZJLZRKHHD-WVWIJVSJSA-N asunaprevir Chemical compound O=C([C@@H]1C[C@H](CN1C(=O)[C@@H](NC(=O)OC(C)(C)C)C(C)(C)C)OC1=NC=C(C2=CC=C(Cl)C=C21)OC)N[C@]1(C(=O)NS(=O)(=O)C2CC2)C[C@H]1C=C XRWSZZJLZRKHHD-WVWIJVSJSA-N 0.000 description 2
- 235000012216 bentonite Nutrition 0.000 description 2
- 239000000440 bentonite Substances 0.000 description 2
- 229910000278 bentonite Inorganic materials 0.000 description 2
- SVPXDRXYRYOSEX-UHFFFAOYSA-N bentoquatam Chemical compound O.O=[Si]=O.O=[Al]O[Al]=O SVPXDRXYRYOSEX-UHFFFAOYSA-N 0.000 description 2
- SESFRYSPDFLNCH-UHFFFAOYSA-N benzyl benzoate Chemical compound C=1C=CC=CC=1C(=O)OCC1=CC=CC=C1 SESFRYSPDFLNCH-UHFFFAOYSA-N 0.000 description 2
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 description 2
- 230000033228 biological regulation Effects 0.000 description 2
- 125000001246 bromo group Chemical group Br* 0.000 description 2
- 239000001768 carboxy methyl cellulose Substances 0.000 description 2
- 239000008112 carboxymethyl-cellulose Substances 0.000 description 2
- 229940105329 carboxymethylcellulose Drugs 0.000 description 2
- 238000001311 chemical methods and process Methods 0.000 description 2
- OSASVXMJTNOKOY-UHFFFAOYSA-N chlorobutanol Chemical compound CC(C)(O)C(Cl)(Cl)Cl OSASVXMJTNOKOY-UHFFFAOYSA-N 0.000 description 2
- 238000000576 coating method Methods 0.000 description 2
- 229940125904 compound 1 Drugs 0.000 description 2
- 229940125797 compound 12 Drugs 0.000 description 2
- 229940126543 compound 14 Drugs 0.000 description 2
- 229940125758 compound 15 Drugs 0.000 description 2
- 229940126142 compound 16 Drugs 0.000 description 2
- 229940125782 compound 2 Drugs 0.000 description 2
- 229940125810 compound 20 Drugs 0.000 description 2
- 229940126086 compound 21 Drugs 0.000 description 2
- 229940126208 compound 22 Drugs 0.000 description 2
- 229940125833 compound 23 Drugs 0.000 description 2
- 229940125961 compound 24 Drugs 0.000 description 2
- 229940125851 compound 27 Drugs 0.000 description 2
- 229940127204 compound 29 Drugs 0.000 description 2
- 229940126214 compound 3 Drugs 0.000 description 2
- 229940125878 compound 36 Drugs 0.000 description 2
- 229940125807 compound 37 Drugs 0.000 description 2
- 229940126540 compound 41 Drugs 0.000 description 2
- 229940125844 compound 46 Drugs 0.000 description 2
- 229940127271 compound 49 Drugs 0.000 description 2
- 229940126545 compound 53 Drugs 0.000 description 2
- 229940127113 compound 57 Drugs 0.000 description 2
- 229940125900 compound 59 Drugs 0.000 description 2
- 229940126179 compound 72 Drugs 0.000 description 2
- 125000004122 cyclic group Chemical group 0.000 description 2
- HGCIXCUEYOPUTN-UHFFFAOYSA-N cyclohexene Chemical compound C1CCC=CC1 HGCIXCUEYOPUTN-UHFFFAOYSA-N 0.000 description 2
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 2
- ZSWFCLXCOIISFI-UHFFFAOYSA-N cyclopentadiene Chemical compound C1C=CC=C1 ZSWFCLXCOIISFI-UHFFFAOYSA-N 0.000 description 2
- LPIQUOYDBNQMRZ-UHFFFAOYSA-N cyclopentene Chemical compound C1CC=CC1 LPIQUOYDBNQMRZ-UHFFFAOYSA-N 0.000 description 2
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 2
- 230000003247 decreasing effect Effects 0.000 description 2
- 125000004984 dialkylaminoalkoxy group Chemical group 0.000 description 2
- 235000001916 dieting Nutrition 0.000 description 2
- 230000037228 dieting effect Effects 0.000 description 2
- 239000002552 dosage form Substances 0.000 description 2
- 239000003995 emulsifying agent Substances 0.000 description 2
- 230000001804 emulsifying effect Effects 0.000 description 2
- 239000000839 emulsion Substances 0.000 description 2
- BJXYHBKEQFQVES-NWDGAFQWSA-N enpatoran Chemical compound N[C@H]1CN(C[C@H](C1)C(F)(F)F)C1=C2C=CC=NC2=C(C=C1)C#N BJXYHBKEQFQVES-NWDGAFQWSA-N 0.000 description 2
- GWNFQAKCJYEJEW-UHFFFAOYSA-N ethyl 3-[8-[[4-methyl-5-[(3-methyl-4-oxophthalazin-1-yl)methyl]-1,2,4-triazol-3-yl]sulfanyl]octanoylamino]benzoate Chemical compound CCOC(=O)C1=CC(NC(=O)CCCCCCCSC2=NN=C(CC3=NN(C)C(=O)C4=CC=CC=C34)N2C)=CC=C1 GWNFQAKCJYEJEW-UHFFFAOYSA-N 0.000 description 2
- 125000004705 ethylthio group Chemical group C(C)S* 0.000 description 2
- 235000019197 fats Nutrition 0.000 description 2
- 150000004665 fatty acids Chemical class 0.000 description 2
- 239000000945 filler Substances 0.000 description 2
- 125000001153 fluoro group Chemical group F* 0.000 description 2
- 239000012458 free base Substances 0.000 description 2
- 239000008273 gelatin Substances 0.000 description 2
- 229920000159 gelatin Polymers 0.000 description 2
- 239000007903 gelatin capsule Substances 0.000 description 2
- 235000019322 gelatine Nutrition 0.000 description 2
- 235000011852 gelatine desserts Nutrition 0.000 description 2
- 239000008187 granular material Substances 0.000 description 2
- JAXFJECJQZDFJS-XHEPKHHKSA-N gtpl8555 Chemical compound OC(=O)C[C@H](N)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](C(C)C)C(=O)N1CCC[C@@H]1C(=O)N[C@H](B1O[C@@]2(C)[C@H]3C[C@H](C3(C)C)C[C@H]2O1)CCC1=CC=C(F)C=C1 JAXFJECJQZDFJS-XHEPKHHKSA-N 0.000 description 2
- 125000000592 heterocycloalkyl group Chemical group 0.000 description 2
- 239000002471 hydroxymethylglutaryl coenzyme A reductase inhibitor Substances 0.000 description 2
- 239000003701 inert diluent Substances 0.000 description 2
- 150000007529 inorganic bases Chemical class 0.000 description 2
- 239000002198 insoluble material Substances 0.000 description 2
- 239000008101 lactose Substances 0.000 description 2
- 239000010410 layer Substances 0.000 description 2
- 230000037356 lipid metabolism Effects 0.000 description 2
- 150000002632 lipids Chemical class 0.000 description 2
- 239000008297 liquid dosage form Substances 0.000 description 2
- 229910052744 lithium Inorganic materials 0.000 description 2
- AMXOYNBUYSYVKV-UHFFFAOYSA-M lithium bromide Chemical compound [Li+].[Br-] AMXOYNBUYSYVKV-UHFFFAOYSA-M 0.000 description 2
- RENRQMCACQEWFC-UGKGYDQZSA-N lnp023 Chemical compound C1([C@H]2N(CC=3C=4C=CNC=4C(C)=CC=3OC)CC[C@@H](C2)OCC)=CC=C(C(O)=O)C=C1 RENRQMCACQEWFC-UGKGYDQZSA-N 0.000 description 2
- 210000002540 macrophage Anatomy 0.000 description 2
- 235000019359 magnesium stearate Nutrition 0.000 description 2
- DJKYKADHIYBHCV-UHFFFAOYSA-N methyl 2-(5-chloro-2-methyl-4-thiocyanatophenoxy)acetate Chemical compound COC(=O)COC1=CC(Cl)=C(SC#N)C=C1C DJKYKADHIYBHCV-UHFFFAOYSA-N 0.000 description 2
- SJLPBWORMPHABI-UHFFFAOYSA-N methyl 2-[(6-methyl-8-sulfanyl-3,4-dihydro-2h-chromen-5-yl)oxy]acetate Chemical compound O1CCCC2=C1C(S)=CC(C)=C2OCC(=O)OC SJLPBWORMPHABI-UHFFFAOYSA-N 0.000 description 2
- RIAXMSOMYLFZAR-UHFFFAOYSA-N methyl 2-[2-methyl-4-[2-[4-[4-(trifluoromethyl)phenyl]phenyl]ethenyl]phenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC=C1C=CC1=CC=C(C=2C=CC(=CC=2)C(F)(F)F)C=C1 RIAXMSOMYLFZAR-UHFFFAOYSA-N 0.000 description 2
- LIGJXDWHWYEGGR-UHFFFAOYSA-N methyl 2-[2-methyl-4-[[5-[4-(trifluoromethyl)phenyl]-1,2-oxazol-3-yl]methylsulfanyl]phenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC=C1SCC1=NOC(C=2C=CC(=CC=2)C(F)(F)F)=C1 LIGJXDWHWYEGGR-UHFFFAOYSA-N 0.000 description 2
- AFDHECXZDCGDCN-UHFFFAOYSA-N methyl 2-[4-[[4-[2-(3-fluorophenyl)ethenyl]phenyl]methylsulfanyl]-5-methoxy-2-methylphenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC(OC)=C1SCC(C=C1)=CC=C1C=CC1=CC=CC(F)=C1 AFDHECXZDCGDCN-UHFFFAOYSA-N 0.000 description 2
- ZIHCUSUALKPHDW-UHFFFAOYSA-N methyl 2-[5-methoxy-2-methyl-4-[2-[4-[4-(trifluoromethyl)phenyl]phenyl]ethylsulfanyl]phenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC(OC)=C1SCCC1=CC=C(C=2C=CC(=CC=2)C(F)(F)F)C=C1 ZIHCUSUALKPHDW-UHFFFAOYSA-N 0.000 description 2
- YQPRBQHRBYFONV-UHFFFAOYSA-N methyl 2-[5-methoxy-2-methyl-4-[2-[4-phenyl-1-(trifluoromethyl)cyclohexa-2,4-dien-1-yl]ethylsulfanyl]phenoxy]acetate Chemical compound C1=C(C)C(OCC(=O)OC)=CC(OC)=C1SCCC1(C(F)(F)F)C=CC(C=2C=CC=CC=2)=CC1 YQPRBQHRBYFONV-UHFFFAOYSA-N 0.000 description 2
- ZZHNTGQQEHGREL-UHFFFAOYSA-N methyl 2-[[7-[[3-(4-chlorophenyl)-1,2-oxazol-5-yl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetate Chemical compound C1=2CCCC=2C(OCC(=O)OC)=CC=C1SCC(ON=1)=CC=1C1=CC=C(Cl)C=C1 ZZHNTGQQEHGREL-UHFFFAOYSA-N 0.000 description 2
- FWZSZZJHMDQUIB-UHFFFAOYSA-N methyl 2-[[7-[[4-[[4-(fluoromethyl)phenyl]methoxy]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetate Chemical compound C1=2CCCC=2C(OCC(=O)OC)=CC=C1SCC(C=C1)=CC=C1OCC1=CC=C(CF)C=C1 FWZSZZJHMDQUIB-UHFFFAOYSA-N 0.000 description 2
- ZBELDPMWYXDLNY-UHFFFAOYSA-N methyl 9-(4-bromo-2-fluoroanilino)-[1,3]thiazolo[5,4-f]quinazoline-2-carboximidate Chemical compound C12=C3SC(C(=N)OC)=NC3=CC=C2N=CN=C1NC1=CC=C(Br)C=C1F ZBELDPMWYXDLNY-UHFFFAOYSA-N 0.000 description 2
- 125000002757 morpholinyl group Chemical group 0.000 description 2
- YGBMCLDVRUGXOV-UHFFFAOYSA-N n-[6-[6-chloro-5-[(4-fluorophenyl)sulfonylamino]pyridin-3-yl]-1,3-benzothiazol-2-yl]acetamide Chemical compound C1=C2SC(NC(=O)C)=NC2=CC=C1C(C=1)=CN=C(Cl)C=1NS(=O)(=O)C1=CC=C(F)C=C1 YGBMCLDVRUGXOV-UHFFFAOYSA-N 0.000 description 2
- 125000001280 n-hexyl group Chemical group C(CCCCC)* 0.000 description 2
- 125000000740 n-pentyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 2
- IOMMMLWIABWRKL-WUTDNEBXSA-N nazartinib Chemical compound C1N(C(=O)/C=C/CN(C)C)CCCC[C@H]1N1C2=C(Cl)C=CC=C2N=C1NC(=O)C1=CC=NC(C)=C1 IOMMMLWIABWRKL-WUTDNEBXSA-N 0.000 description 2
- QWVGKYWNOKOFNN-UHFFFAOYSA-N o-cresol Chemical compound CC1=CC=CC=C1O QWVGKYWNOKOFNN-UHFFFAOYSA-N 0.000 description 2
- QIQXTHQIDYTFRH-UHFFFAOYSA-N octadecanoic acid Chemical compound CCCCCCCCCCCCCCCCCC(O)=O QIQXTHQIDYTFRH-UHFFFAOYSA-N 0.000 description 2
- 239000003921 oil Substances 0.000 description 2
- 235000019198 oils Nutrition 0.000 description 2
- 239000002674 ointment Substances 0.000 description 2
- 239000004006 olive oil Substances 0.000 description 2
- 235000008390 olive oil Nutrition 0.000 description 2
- 150000007530 organic bases Chemical class 0.000 description 2
- CTSLXHKWHWQRSH-UHFFFAOYSA-N oxalyl chloride Chemical compound ClC(=O)C(Cl)=O CTSLXHKWHWQRSH-UHFFFAOYSA-N 0.000 description 2
- 229910052763 palladium Inorganic materials 0.000 description 2
- YJVFFLUZDVXJQI-UHFFFAOYSA-L palladium(ii) acetate Chemical compound [Pd+2].CC([O-])=O.CC([O-])=O YJVFFLUZDVXJQI-UHFFFAOYSA-L 0.000 description 2
- 239000003614 peroxisome proliferator Substances 0.000 description 2
- 108091008725 peroxisome proliferator-activated receptors alpha Proteins 0.000 description 2
- 125000003367 polycyclic group Chemical group 0.000 description 2
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 2
- 229960004063 propylene glycol Drugs 0.000 description 2
- 235000013772 propylene glycol Nutrition 0.000 description 2
- 108090000623 proteins and genes Proteins 0.000 description 2
- 125000000719 pyrrolidinyl group Chemical group 0.000 description 2
- 239000011347 resin Substances 0.000 description 2
- 229920005989 resin Polymers 0.000 description 2
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 2
- 239000000377 silicon dioxide Substances 0.000 description 2
- 235000012239 silicon dioxide Nutrition 0.000 description 2
- 235000017557 sodium bicarbonate Nutrition 0.000 description 2
- 239000011780 sodium chloride Substances 0.000 description 2
- 239000012312 sodium hydride Substances 0.000 description 2
- 239000008107 starch Substances 0.000 description 2
- 239000005720 sucrose Substances 0.000 description 2
- 235000000346 sugar Nutrition 0.000 description 2
- 125000000472 sulfonyl group Chemical group *S(*)(=O)=O 0.000 description 2
- 239000000829 suppository Substances 0.000 description 2
- 239000000375 suspending agent Substances 0.000 description 2
- 208000024891 symptom Diseases 0.000 description 2
- 238000012360 testing method Methods 0.000 description 2
- HJUGFYREWKUQJT-UHFFFAOYSA-N tetrabromomethane Chemical compound BrC(Br)(Br)Br HJUGFYREWKUQJT-UHFFFAOYSA-N 0.000 description 2
- RWRDLPDLKQPQOW-UHFFFAOYSA-N tetrahydropyrrole Natural products C1CCNC1 RWRDLPDLKQPQOW-UHFFFAOYSA-N 0.000 description 2
- RAOIDOHSFRTOEL-UHFFFAOYSA-N tetrahydrothiophene Chemical compound C1CCSC1 RAOIDOHSFRTOEL-UHFFFAOYSA-N 0.000 description 2
- FYSNRJHAOHDILO-UHFFFAOYSA-N thionyl chloride Chemical compound ClS(Cl)=O FYSNRJHAOHDILO-UHFFFAOYSA-N 0.000 description 2
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 2
- 238000011830 transgenic mouse model Methods 0.000 description 2
- AQRLNPVMDITEJU-UHFFFAOYSA-N triethylsilane Chemical compound CC[SiH](CC)CC AQRLNPVMDITEJU-UHFFFAOYSA-N 0.000 description 2
- UFTFJSFQGQCHQW-UHFFFAOYSA-N triformin Chemical compound O=COCC(OC=O)COC=O UFTFJSFQGQCHQW-UHFFFAOYSA-N 0.000 description 2
- GETQZCLCWQTVFV-UHFFFAOYSA-N trimethylamine Chemical compound CN(C)C GETQZCLCWQTVFV-UHFFFAOYSA-N 0.000 description 2
- 238000011144 upstream manufacturing Methods 0.000 description 2
- 239000001993 wax Substances 0.000 description 2
- 230000004580 weight loss Effects 0.000 description 2
- 239000000080 wetting agent Substances 0.000 description 2
- UAYWVJHJZHQCIE-UHFFFAOYSA-L zinc iodide Chemical compound I[Zn]I UAYWVJHJZHQCIE-UHFFFAOYSA-L 0.000 description 2
- XUFXOAAUWZOOIT-SXARVLRPSA-N (2R,3R,4R,5S,6R)-5-[[(2R,3R,4R,5S,6R)-5-[[(2R,3R,4S,5S,6R)-3,4-dihydroxy-6-methyl-5-[[(1S,4R,5S,6S)-4,5,6-trihydroxy-3-(hydroxymethyl)-1-cyclohex-2-enyl]amino]-2-oxanyl]oxy]-3,4-dihydroxy-6-(hydroxymethyl)-2-oxanyl]oxy]-6-(hydroxymethyl)oxane-2,3,4-triol Chemical compound O([C@H]1O[C@H](CO)[C@H]([C@@H]([C@H]1O)O)O[C@H]1O[C@@H]([C@H]([C@H](O)[C@H]1O)N[C@@H]1[C@@H]([C@@H](O)[C@H](O)C(CO)=C1)O)C)[C@@H]1[C@@H](CO)O[C@@H](O)[C@H](O)[C@H]1O XUFXOAAUWZOOIT-SXARVLRPSA-N 0.000 description 1
- PQXKWPLDPFFDJP-ZXZARUISSA-N (2r,3s)-2,3-dimethyloxirane Chemical compound C[C@H]1O[C@H]1C PQXKWPLDPFFDJP-ZXZARUISSA-N 0.000 description 1
- VSNNLLQKDRCKCB-UHFFFAOYSA-N (3,5-dichlorophenyl)methanol Chemical compound OCC1=CC(Cl)=CC(Cl)=C1 VSNNLLQKDRCKCB-UHFFFAOYSA-N 0.000 description 1
- IFKWLKCPUIQXPU-UHFFFAOYSA-N (4-bromo-2-methylphenyl)methanol Chemical compound CC1=CC(Br)=CC=C1CO IFKWLKCPUIQXPU-UHFFFAOYSA-N 0.000 description 1
- BUZNFXSCYITJEF-UHFFFAOYSA-N (4-hydroxy-2,5-dimethylphenyl) thiocyanate Chemical compound CC1=CC(SC#N)=C(C)C=C1O BUZNFXSCYITJEF-UHFFFAOYSA-N 0.000 description 1
- FGPPWMUEDVMTNA-UHFFFAOYSA-N (4-hydroxy-5,6,7,8-tetrahydronaphthalen-1-yl) thiocyanate Chemical compound C1CCCC2=C1C(SC#N)=CC=C2O FGPPWMUEDVMTNA-UHFFFAOYSA-N 0.000 description 1
- AXCHZLOJGKSWLV-UHFFFAOYSA-N (4-phenylphenyl)methanol Chemical compound C1=CC(CO)=CC=C1C1=CC=CC=C1 AXCHZLOJGKSWLV-UHFFFAOYSA-N 0.000 description 1
- LYEQNUQZTOQEIH-UHFFFAOYSA-N *.CC.CCCBr.CCCCCCl.CCCCCO.OCCB(O)O.[V].[W].[Y] Chemical compound *.CC.CCCBr.CCCCCCl.CCCCCO.OCCB(O)O.[V].[W].[Y] LYEQNUQZTOQEIH-UHFFFAOYSA-N 0.000 description 1
- SCYULBFZEHDVBN-UHFFFAOYSA-N 1,1-Dichloroethane Chemical compound CC(Cl)Cl SCYULBFZEHDVBN-UHFFFAOYSA-N 0.000 description 1
- FIDRAVVQGKNYQK-UHFFFAOYSA-N 1,2,3,4-tetrahydrotriazine Chemical group C1NNNC=C1 FIDRAVVQGKNYQK-UHFFFAOYSA-N 0.000 description 1
- HHFJHICTIHHPLD-UHFFFAOYSA-N 1,2-dichloro-4-[[4-(chloromethyl)phenoxy]methyl]benzene Chemical compound C1=CC(CCl)=CC=C1OCC1=CC=C(Cl)C(Cl)=C1 HHFJHICTIHHPLD-UHFFFAOYSA-N 0.000 description 1
- 229940058015 1,3-butylene glycol Drugs 0.000 description 1
- RYHBNJHYFVUHQT-UHFFFAOYSA-N 1,4-Dioxane Chemical compound C1COCCO1 RYHBNJHYFVUHQT-UHFFFAOYSA-N 0.000 description 1
- OMZINLIPPVNUOG-UHFFFAOYSA-N 1,4-dichloro-2-(chloromethyl)benzene Chemical compound ClCC1=CC(Cl)=CC=C1Cl OMZINLIPPVNUOG-UHFFFAOYSA-N 0.000 description 1
- WGSFPLUFRPUBCH-UHFFFAOYSA-N 1-(4-chlorophenyl)-3-(4-methoxyphenyl)-3-(4-methylphenyl)sulfonylpropan-1-one Chemical compound C1=CC(OC)=CC=C1C(S(=O)(=O)C=1C=CC(C)=CC=1)CC(=O)C1=CC=C(Cl)C=C1 WGSFPLUFRPUBCH-UHFFFAOYSA-N 0.000 description 1
- HTWOLWSZQAPKQA-UHFFFAOYSA-N 1-(bromomethyl)-3-fluoro-2-(trifluoromethyl)benzene Chemical compound FC1=CC=CC(CBr)=C1C(F)(F)F HTWOLWSZQAPKQA-UHFFFAOYSA-N 0.000 description 1
- KFWJYVSMZTXKKG-UHFFFAOYSA-N 1-(bromomethyl)-4-[4-(trifluoromethyl)phenyl]benzene Chemical compound C1=CC(C(F)(F)F)=CC=C1C1=CC=C(CBr)C=C1 KFWJYVSMZTXKKG-UHFFFAOYSA-N 0.000 description 1
- KQNBRMUBPRGXSL-UHFFFAOYSA-N 1-(bromomethyl)-4-chlorobenzene Chemical compound ClC1=CC=C(CBr)C=C1 KQNBRMUBPRGXSL-UHFFFAOYSA-N 0.000 description 1
- GIGRWGTZFONRKA-UHFFFAOYSA-N 1-(bromomethyl)-4-methoxybenzene Chemical compound COC1=CC=C(CBr)C=C1 GIGRWGTZFONRKA-UHFFFAOYSA-N 0.000 description 1
- QZMCURBKSYOCRT-UHFFFAOYSA-N 1-(chloromethyl)-2-[4-(trifluoromethyl)phenyl]benzene Chemical group C1=CC(C(F)(F)F)=CC=C1C1=CC=CC=C1CCl QZMCURBKSYOCRT-UHFFFAOYSA-N 0.000 description 1
- DDMVJYGBWVQLDZ-UHFFFAOYSA-N 1-(chloromethyl)-4-(4-chlorophenyl)-2-(trifluoromethyl)benzene Chemical group C1=C(CCl)C(C(F)(F)F)=CC(C=2C=CC(Cl)=CC=2)=C1 DDMVJYGBWVQLDZ-UHFFFAOYSA-N 0.000 description 1
- MCHDHQVROPEJJT-UHFFFAOYSA-N 1-(chloromethyl)-4-(trifluoromethyl)benzene Chemical compound FC(F)(F)C1=CC=C(CCl)C=C1 MCHDHQVROPEJJT-UHFFFAOYSA-N 0.000 description 1
- MOHYOXXOKFQHDC-UHFFFAOYSA-N 1-(chloromethyl)-4-methoxybenzene Chemical compound COC1=CC=C(CCl)C=C1 MOHYOXXOKFQHDC-UHFFFAOYSA-N 0.000 description 1
- IATNZRYVIRYKDJ-UHFFFAOYSA-N 1-(chloromethyl)-4-phenoxybenzene Chemical compound C1=CC(CCl)=CC=C1OC1=CC=CC=C1 IATNZRYVIRYKDJ-UHFFFAOYSA-N 0.000 description 1
- KQZLRWGGWXJPOS-NLFPWZOASA-N 1-[(1R)-1-(2,4-dichlorophenyl)ethyl]-6-[(4S,5R)-4-[(2S)-2-(hydroxymethyl)pyrrolidin-1-yl]-5-methylcyclohexen-1-yl]pyrazolo[3,4-b]pyrazine-3-carbonitrile Chemical compound ClC1=C(C=CC(=C1)Cl)[C@@H](C)N1N=C(C=2C1=NC(=CN=2)C1=CC[C@@H]([C@@H](C1)C)N1[C@@H](CCC1)CO)C#N KQZLRWGGWXJPOS-NLFPWZOASA-N 0.000 description 1
- WZZBNLYBHUDSHF-DHLKQENFSA-N 1-[(3s,4s)-4-[8-(2-chloro-4-pyrimidin-2-yloxyphenyl)-7-fluoro-2-methylimidazo[4,5-c]quinolin-1-yl]-3-fluoropiperidin-1-yl]-2-hydroxyethanone Chemical compound CC1=NC2=CN=C3C=C(F)C(C=4C(=CC(OC=5N=CC=CN=5)=CC=4)Cl)=CC3=C2N1[C@H]1CCN(C(=O)CO)C[C@@H]1F WZZBNLYBHUDSHF-DHLKQENFSA-N 0.000 description 1
- YXDVFKLDISXBSU-UHFFFAOYSA-N 1-[4-(4-naphthalen-1-yloxyphenyl)sulfonylphenoxy]naphthalene Chemical compound C1=CC=C2C(OC3=CC=C(C=C3)S(=O)(C=3C=CC(OC=4C5=CC=CC=C5C=CC=4)=CC=3)=O)=CC=CC2=C1 YXDVFKLDISXBSU-UHFFFAOYSA-N 0.000 description 1
- HHAISVSEJFEWBZ-UHFFFAOYSA-N 1-[4-(trifluoromethyl)phenyl]ethanone Chemical compound CC(=O)C1=CC=C(C(F)(F)F)C=C1 HHAISVSEJFEWBZ-UHFFFAOYSA-N 0.000 description 1
- 125000004173 1-benzimidazolyl group Chemical group [H]C1=NC2=C([H])C([H])=C([H])C([H])=C2N1* 0.000 description 1
- ISHYFWKKWKXXPL-UHFFFAOYSA-N 1-bromo-2,4-dichlorobenzene Chemical compound ClC1=CC=C(Br)C(Cl)=C1 ISHYFWKKWKXXPL-UHFFFAOYSA-N 0.000 description 1
- NNMBNYHMJRJUBC-UHFFFAOYSA-N 1-bromo-3-(trifluoromethyl)benzene Chemical compound FC(F)(F)C1=CC=CC(Br)=C1 NNMBNYHMJRJUBC-UHFFFAOYSA-N 0.000 description 1
- WGGLDBIZIQMEGH-UHFFFAOYSA-N 1-bromo-4-ethenylbenzene Chemical compound BrC1=CC=C(C=C)C=C1 WGGLDBIZIQMEGH-UHFFFAOYSA-N 0.000 description 1
- AITNMTXHTIIIBB-UHFFFAOYSA-N 1-bromo-4-fluorobenzene Chemical compound FC1=CC=C(Br)C=C1 AITNMTXHTIIIBB-UHFFFAOYSA-N 0.000 description 1
- 125000004973 1-butenyl group Chemical group C(=CCC)* 0.000 description 1
- 125000004972 1-butynyl group Chemical group [H]C([H])([H])C([H])([H])C#C* 0.000 description 1
- UHUDPGSZCOUKAI-UHFFFAOYSA-N 1-chloro-4-[[4-(chloromethyl)phenoxy]methyl]-2-(trifluoromethyl)benzene Chemical compound C1=C(Cl)C(C(F)(F)F)=CC(COC=2C=CC(CCl)=CC=2)=C1 UHUDPGSZCOUKAI-UHFFFAOYSA-N 0.000 description 1
- LDMOEFOXLIZJOW-UHFFFAOYSA-N 1-dodecanesulfonic acid Chemical class CCCCCCCCCCCCS(O)(=O)=O LDMOEFOXLIZJOW-UHFFFAOYSA-N 0.000 description 1
- ZJSKEGAHBAHFON-UHFFFAOYSA-N 1-ethenyl-3-fluorobenzene Chemical compound FC1=CC=CC(C=C)=C1 ZJSKEGAHBAHFON-UHFFFAOYSA-N 0.000 description 1
- 125000006039 1-hexenyl group Chemical group 0.000 description 1
- FFENXTLBEPCBBG-UHFFFAOYSA-N 1-methoxyethanol Chemical compound [CH2]C(O)OC FFENXTLBEPCBBG-UHFFFAOYSA-N 0.000 description 1
- IIZPXYDJLKNOIY-JXPKJXOSSA-N 1-palmitoyl-2-arachidonoyl-sn-glycero-3-phosphocholine Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCC\C=C/C\C=C/C\C=C/C\C=C/CCCCC IIZPXYDJLKNOIY-JXPKJXOSSA-N 0.000 description 1
- 125000006023 1-pentenyl group Chemical group 0.000 description 1
- 125000006017 1-propenyl group Chemical group 0.000 description 1
- 125000000530 1-propynyl group Chemical group [H]C([H])([H])C#C* 0.000 description 1
- HEWZVZIVELJPQZ-UHFFFAOYSA-N 2,2-dimethoxypropane Chemical compound COC(C)(C)OC HEWZVZIVELJPQZ-UHFFFAOYSA-N 0.000 description 1
- 125000004825 2,2-dimethylpropylene group Chemical group [H]C([H])([H])C(C([H])([H])[H])(C([H])([H])[*:1])C([H])([H])[*:2] 0.000 description 1
- PQXKWPLDPFFDJP-UHFFFAOYSA-N 2,3-dimethyloxirane Chemical compound CC1OC1C PQXKWPLDPFFDJP-UHFFFAOYSA-N 0.000 description 1
- 150000001473 2,4-thiazolidinediones Chemical class 0.000 description 1
- SQKUSGDAQJJKBT-UHFFFAOYSA-N 2,6-dichloro-4-(hydroxymethyl)phenol Chemical compound OCC1=CC(Cl)=C(O)C(Cl)=C1 SQKUSGDAQJJKBT-UHFFFAOYSA-N 0.000 description 1
- JZUPYBRYQINNRE-UHFFFAOYSA-N 2,6-dimethyl-1,4-dioxane Chemical compound CC1COCC(C)O1 JZUPYBRYQINNRE-UHFFFAOYSA-N 0.000 description 1
- PMOSJSPFNDUAFY-UHFFFAOYSA-N 2-(4-bromophenyl)ethanol Chemical compound OCCC1=CC=C(Br)C=C1 PMOSJSPFNDUAFY-UHFFFAOYSA-N 0.000 description 1
- MKHGVMIXRPGHOO-UHFFFAOYSA-N 2-(benzenesulfonyl)-3-phenyloxaziridine Chemical compound C=1C=CC=CC=1S(=O)(=O)N1OC1C1=CC=CC=C1 MKHGVMIXRPGHOO-UHFFFAOYSA-N 0.000 description 1
- UUVDOPTUDWJHFK-UHFFFAOYSA-N 2-(bromomethyl)-1,4-dichlorobenzene Chemical compound ClC1=CC=C(Cl)C(CBr)=C1 UUVDOPTUDWJHFK-UHFFFAOYSA-N 0.000 description 1
- QJYHMWJJNWZWCH-UHFFFAOYSA-N 2-(bromomethyl)pyridine;hydrochloride Chemical compound Cl.BrCC1=CC=CC=N1 QJYHMWJJNWZWCH-UHFFFAOYSA-N 0.000 description 1
- NGNBDVOYPDDBFK-UHFFFAOYSA-N 2-[2,4-di(pentan-2-yl)phenoxy]acetyl chloride Chemical compound CCCC(C)C1=CC=C(OCC(Cl)=O)C(C(C)CCC)=C1 NGNBDVOYPDDBFK-UHFFFAOYSA-N 0.000 description 1
- ACUVPNVTIKDDRA-UHFFFAOYSA-N 2-[2,5-dimethyl-4-[(2-phenylphenyl)methylsulfanyl]phenoxy]acetic acid Chemical compound C1=C(OCC(O)=O)C(C)=CC(SCC=2C(=CC=CC=2)C=2C=CC=CC=2)=C1C ACUVPNVTIKDDRA-UHFFFAOYSA-N 0.000 description 1
- KOINHDMQMQWBQJ-UHFFFAOYSA-N 2-[2,5-dimethyl-4-[(4-phenylmethoxyphenyl)methylsulfanyl]phenoxy]acetic acid Chemical compound C1=C(OCC(O)=O)C(C)=CC(SCC=2C=CC(OCC=3C=CC=CC=3)=CC=2)=C1C KOINHDMQMQWBQJ-UHFFFAOYSA-N 0.000 description 1
- AHDAFGVZAJALJQ-UHFFFAOYSA-N 2-[2,5-dimethyl-4-[(4-phenylphenyl)methylsulfanyl]phenoxy]acetic acid Chemical compound C1=C(OCC(O)=O)C(C)=CC(SCC=2C=CC(=CC=2)C=2C=CC=CC=2)=C1C AHDAFGVZAJALJQ-UHFFFAOYSA-N 0.000 description 1
- OTJXPFXLIASUKS-UHFFFAOYSA-N 2-[2,5-dimethyl-4-[(4-pyrazol-1-ylphenyl)methylsulfanyl]phenoxy]acetic acid Chemical compound C1=C(OCC(O)=O)C(C)=CC(SCC=2C=CC(=CC=2)N2N=CC=C2)=C1C OTJXPFXLIASUKS-UHFFFAOYSA-N 0.000 description 1
- IQHBIRHWXDXIFO-UHFFFAOYSA-N 2-[2,5-dimethyl-4-[[5-(4-methylphenyl)-1,3,4-oxadiazol-2-yl]methylsulfanyl]phenoxy]acetic acid Chemical compound C1=CC(C)=CC=C1C(O1)=NN=C1CSC1=CC(C)=C(OCC(O)=O)C=C1C IQHBIRHWXDXIFO-UHFFFAOYSA-N 0.000 description 1
- KOCPUUDONLBYLP-UHFFFAOYSA-N 2-[2,6-dimethyl-4-[(4-phenylmethoxyphenyl)methylsulfanyl]phenoxy]acetic acid Chemical compound CC1=C(OCC(O)=O)C(C)=CC(SCC=2C=CC(OCC=3C=CC=CC=3)=CC=2)=C1 KOCPUUDONLBYLP-UHFFFAOYSA-N 0.000 description 1
- WZFSQZBYOZGXDT-UHFFFAOYSA-N 2-[2,6-dimethyl-4-[(4-phenylphenyl)methylsulfanyl]phenoxy]acetic acid Chemical compound CC1=C(OCC(O)=O)C(C)=CC(SCC=2C=CC(=CC=2)C=2C=CC=CC=2)=C1 WZFSQZBYOZGXDT-UHFFFAOYSA-N 0.000 description 1
- CHTDVQHTWDNXDA-UHFFFAOYSA-N 2-[2-methyl-4-[(2-phenylphenyl)methylsulfanyl]phenoxy]acetic acid Chemical compound C1=C(OCC(O)=O)C(C)=CC(SCC=2C(=CC=CC=2)C=2C=CC=CC=2)=C1 CHTDVQHTWDNXDA-UHFFFAOYSA-N 0.000 description 1
- COXBOGGXLLXUQH-UHFFFAOYSA-N 2-[2-methyl-4-[(3-phenoxyphenyl)methylsulfanyl]phenoxy]acetic acid Chemical compound C1=C(OCC(O)=O)C(C)=CC(SCC=2C=C(OC=3C=CC=CC=3)C=CC=2)=C1 COXBOGGXLLXUQH-UHFFFAOYSA-N 0.000 description 1
- MZRHDZKDYBACND-UHFFFAOYSA-N 2-[2-methyl-4-[(4-pyrazol-1-ylphenyl)methylsulfanyl]phenoxy]acetic acid Chemical compound C1=C(OCC(O)=O)C(C)=CC(SCC=2C=CC(=CC=2)N2N=CC=C2)=C1 MZRHDZKDYBACND-UHFFFAOYSA-N 0.000 description 1
- AGUVRAUTEUWNDE-UHFFFAOYSA-N 2-[2-methyl-4-[(5-methyl-3-phenyl-1,2-oxazol-4-yl)methylsulfanyl]phenoxy]acetic acid Chemical compound CC=1ON=C(C=2C=CC=CC=2)C=1CSC1=CC=C(OCC(O)=O)C(C)=C1 AGUVRAUTEUWNDE-UHFFFAOYSA-N 0.000 description 1
- JZDQVGBTXMFQEJ-UHFFFAOYSA-N 2-[2-methyl-4-[2-(5-phenylnaphthalen-1-yl)oxyethylsulfanyl]phenoxy]acetic acid Chemical compound C1=C(OCC(O)=O)C(C)=CC(SCCOC=2C3=CC=CC(=C3C=CC=2)C=2C=CC=CC=2)=C1 JZDQVGBTXMFQEJ-UHFFFAOYSA-N 0.000 description 1
- WLBLEWYDVLQENM-UHFFFAOYSA-N 2-[2-methyl-4-[2-[4-[2-(3-phenyl-1-benzofuran-7-yl)ethyl]piperazin-1-yl]ethylsulfanyl]phenoxy]acetic acid Chemical compound C1=C(OCC(O)=O)C(C)=CC(SCCN2CCN(CCC=3C=4OC=C(C=4C=CC=3)C=3C=CC=CC=3)CC2)=C1 WLBLEWYDVLQENM-UHFFFAOYSA-N 0.000 description 1
- VBTRHMDCZOSPCF-UHFFFAOYSA-N 2-[2-methyl-4-[[5-(4-methylphenyl)-1,3,4-oxadiazol-2-yl]methylsulfanyl]phenoxy]acetic acid Chemical compound C1=CC(C)=CC=C1C(O1)=NN=C1CSC1=CC=C(OCC(O)=O)C(C)=C1 VBTRHMDCZOSPCF-UHFFFAOYSA-N 0.000 description 1
- CPOJCOSZSXNCHO-UHFFFAOYSA-N 2-[4-(chloromethyl)phenyl]-1,3,5-trimethylbenzene Chemical group CC1=CC(C)=CC(C)=C1C1=CC=C(CCl)C=C1 CPOJCOSZSXNCHO-UHFFFAOYSA-N 0.000 description 1
- IVKDNORULJUVCY-UHFFFAOYSA-N 2-[4-[(4-phenylmethoxyphenyl)methylsulfanyl]phenoxy]acetic acid Chemical compound C1=CC(OCC(=O)O)=CC=C1SCC(C=C1)=CC=C1OCC1=CC=CC=C1 IVKDNORULJUVCY-UHFFFAOYSA-N 0.000 description 1
- OEBMLZJERWLDRT-UHFFFAOYSA-N 2-[4-[(4-phenylphenyl)methylsulfanyl]phenoxy]acetic acid Chemical compound C1=CC(OCC(=O)O)=CC=C1SCC1=CC=C(C=2C=CC=CC=2)C=C1 OEBMLZJERWLDRT-UHFFFAOYSA-N 0.000 description 1
- PMNOZTIMAHTUMA-UHFFFAOYSA-N 2-[4-[2-(2,6-diphenylpiperidin-1-yl)ethylsulfanyl]-2-methylphenoxy]acetic acid Chemical compound C1=C(OCC(O)=O)C(C)=CC(SCCN2C(CCCC2C=2C=CC=CC=2)C=2C=CC=CC=2)=C1 PMNOZTIMAHTUMA-UHFFFAOYSA-N 0.000 description 1
- CKHPGNJOZMQAON-UHFFFAOYSA-N 2-[4-[2-(2,6-diphenylpiperidin-1-yl)ethylsulfanyl]-5-methoxy-2-methylphenoxy]acetic acid Chemical compound COC1=CC(OCC(O)=O)=C(C)C=C1SCCN1C(C=2C=CC=CC=2)CCCC1C1=CC=CC=C1 CKHPGNJOZMQAON-UHFFFAOYSA-N 0.000 description 1
- JXRIZTMBXFUBJU-UHFFFAOYSA-N 2-[4-[2-(4-bromophenoxy)ethylsulfanyl]-2,6-dimethylphenoxy]acetic acid Chemical compound CC1=C(OCC(O)=O)C(C)=CC(SCCOC=2C=CC(Br)=CC=2)=C1 JXRIZTMBXFUBJU-UHFFFAOYSA-N 0.000 description 1
- WEIKNQBUHODDPD-UHFFFAOYSA-N 2-[4-[2-[3-(4-fluorophenyl)-1-benzothiophen-7-yl]ethylsulfanyl]-2-methylphenoxy]acetic acid Chemical compound C1=C(OCC(O)=O)C(C)=CC(SCCC=2C=3SC=C(C=3C=CC=2)C=2C=CC(F)=CC=2)=C1 WEIKNQBUHODDPD-UHFFFAOYSA-N 0.000 description 1
- ZYKCOVMUVXXFJM-UHFFFAOYSA-N 2-[4-[2-[4-[2-(3-chloro-4-cyclohexylphenyl)ethyl]piperazin-1-yl]ethylsulfanyl]-2-methylphenoxy]acetic acid Chemical compound C1=C(OCC(O)=O)C(C)=CC(SCCN2CCN(CCC=3C=C(Cl)C(C4CCCCC4)=CC=3)CC2)=C1 ZYKCOVMUVXXFJM-UHFFFAOYSA-N 0.000 description 1
- LLFWNIBURHMDBQ-UHFFFAOYSA-N 2-[4-[3-[2-[4-[2-(diethylamino)ethoxy]phenyl]benzimidazol-1-yl]propylsulfanyl]-5-methoxy-2-methylphenoxy]acetic acid Chemical compound C1=CC(OCCN(CC)CC)=CC=C1C1=NC2=CC=CC=C2N1CCCSC1=CC(C)=C(OCC(O)=O)C=C1OC LLFWNIBURHMDBQ-UHFFFAOYSA-N 0.000 description 1
- BYFISCKERYKXGC-UHFFFAOYSA-N 2-[4-[[2-butyl-5-chloro-1-[[4-(1-cyanocyclopentyl)phenyl]methyl]imidazol-4-yl]methylsulfanyl]-2-methylphenoxy]acetic acid Chemical compound ClC=1N(CC=2C=CC(=CC=2)C2(CCCC2)C#N)C(CCCC)=NC=1CSC1=CC=C(OCC(O)=O)C(C)=C1 BYFISCKERYKXGC-UHFFFAOYSA-N 0.000 description 1
- FTVRHGIYMXHIAR-UHFFFAOYSA-N 2-[4-[[3-(2-fluorophenoxy)phenyl]methylsulfanyl]-2,6-dimethylphenoxy]acetic acid Chemical compound CC1=C(OCC(O)=O)C(C)=CC(SCC=2C=C(OC=3C(=CC=CC=3)F)C=CC=2)=C1 FTVRHGIYMXHIAR-UHFFFAOYSA-N 0.000 description 1
- YVVSWZYTKNPZMJ-UHFFFAOYSA-N 2-[4-[[3-(4-chlorophenyl)-1,2,4-oxadiazol-5-yl]methylsulfanyl]-2-methylphenoxy]acetic acid Chemical compound C1=C(OCC(O)=O)C(C)=CC(SCC=2ON=C(N=2)C=2C=CC(Cl)=CC=2)=C1 YVVSWZYTKNPZMJ-UHFFFAOYSA-N 0.000 description 1
- HQMLIKFHVORQSO-UHFFFAOYSA-N 2-[5-methoxy-2-methyl-4-[1-[4-[4-(trifluoromethyl)phenyl]phenyl]ethylsulfanyl]phenoxy]acetic acid Chemical compound COC1=CC(OCC(O)=O)=C(C)C=C1SC(C)C1=CC=C(C=2C=CC(=CC=2)C(F)(F)F)C=C1 HQMLIKFHVORQSO-UHFFFAOYSA-N 0.000 description 1
- GLUDLMOIYFFBCR-UHFFFAOYSA-N 2-[5-methoxy-2-methyl-4-[2-[4-[2-(3-phenyl-1-benzofuran-7-yl)ethyl]piperazin-1-yl]ethylsulfanyl]phenoxy]acetic acid Chemical compound COC1=CC(OCC(O)=O)=C(C)C=C1SCCN1CCN(CCC=2C=3OC=C(C=3C=CC=2)C=2C=CC=CC=2)CC1 GLUDLMOIYFFBCR-UHFFFAOYSA-N 0.000 description 1
- KDXJNYWMXXWFMY-UHFFFAOYSA-N 2-[5-methoxy-2-methyl-4-[[5-(4-phenylphenyl)-2-thiophen-2-yl-4,5-dihydro-1,3-oxazol-4-yl]methylsulfanyl]phenoxy]acetic acid Chemical compound COC1=CC(OCC(O)=O)=C(C)C=C1SCC1C(C=2C=CC(=CC=2)C=2C=CC=CC=2)OC(C=2SC=CC=2)=N1 KDXJNYWMXXWFMY-UHFFFAOYSA-N 0.000 description 1
- KSQSHMPBIHPSDG-UHFFFAOYSA-N 2-[5-methoxy-2-methyl-4-[[6-[4-(trifluoromethoxy)phenyl]pyridin-3-yl]methylsulfanyl]phenoxy]acetic acid Chemical compound COC1=CC(OCC(O)=O)=C(C)C=C1SCC1=CC=C(C=2C=CC(OC(F)(F)F)=CC=2)N=C1 KSQSHMPBIHPSDG-UHFFFAOYSA-N 0.000 description 1
- DLGIZPUAZYHPCA-UHFFFAOYSA-N 2-[[4-[(4-phenylmethoxyphenyl)methylsulfanyl]-5,6,7,8-tetrahydronaphthalen-1-yl]oxy]acetic acid Chemical compound C1=2CCCCC=2C(OCC(=O)O)=CC=C1SCC(C=C1)=CC=C1OCC1=CC=CC=C1 DLGIZPUAZYHPCA-UHFFFAOYSA-N 0.000 description 1
- UTKAHLFEMIUVFS-UHFFFAOYSA-N 2-[[4-[(4-phenylphenyl)methylsulfanyl]-5,6,7,8-tetrahydronaphthalen-1-yl]oxy]acetic acid Chemical compound C1=2CCCCC=2C(OCC(=O)O)=CC=C1SCC(C=C1)=CC=C1C1=CC=CC=C1 UTKAHLFEMIUVFS-UHFFFAOYSA-N 0.000 description 1
- QRYXOMDLTJTEGA-UHFFFAOYSA-N 2-[[5-methoxy-7-[[4-[[4-(trifluoromethyl)phenyl]methoxy]phenyl]methylsulfanyl]-2,3-dihydro-1h-inden-4-yl]oxy]acetic acid Chemical compound C=12CCCC2=C(OCC(O)=O)C(OC)=CC=1SCC(C=C1)=CC=C1OCC1=CC=C(C(F)(F)F)C=C1 QRYXOMDLTJTEGA-UHFFFAOYSA-N 0.000 description 1
- RRTLQRYOJOSPEA-UHFFFAOYSA-N 2-bromo-1,3,5-trimethylbenzene Chemical compound CC1=CC(C)=C(Br)C(C)=C1 RRTLQRYOJOSPEA-UHFFFAOYSA-N 0.000 description 1
- 125000004974 2-butenyl group Chemical group C(C=CC)* 0.000 description 1
- SKCNYHLTRZIINA-UHFFFAOYSA-N 2-chloro-5-(chloromethyl)pyridine Chemical compound ClCC1=CC=C(Cl)N=C1 SKCNYHLTRZIINA-UHFFFAOYSA-N 0.000 description 1
- 125000004182 2-chlorophenyl group Chemical group [H]C1=C([H])C(Cl)=C(*)C([H])=C1[H] 0.000 description 1
- 125000006040 2-hexenyl group Chemical group 0.000 description 1
- 125000004204 2-methoxyphenyl group Chemical group [H]C1=C([H])C(*)=C(OC([H])([H])[H])C([H])=C1[H] 0.000 description 1
- 125000006024 2-pentenyl group Chemical group 0.000 description 1
- 125000003903 2-propenyl group Chemical group [H]C([*])([H])C([H])=C([H])[H] 0.000 description 1
- LCHYEKKJCUJAKN-UHFFFAOYSA-N 2-propylphenol Chemical compound CCCC1=CC=CC=C1O LCHYEKKJCUJAKN-UHFFFAOYSA-N 0.000 description 1
- 125000004189 3,4-dichlorophenyl group Chemical group [H]C1=C([H])C(Cl)=C(Cl)C([H])=C1* 0.000 description 1
- NTLAICDKHHQUGC-UHFFFAOYSA-N 3-(2-bromoethyl)-1h-indole Chemical compound C1=CC=C2C(CCBr)=CNC2=C1 NTLAICDKHHQUGC-UHFFFAOYSA-N 0.000 description 1
- UKBHBRICCXLWJZ-UHFFFAOYSA-N 3-(chloromethyl)-5-(2-chlorophenyl)-1,2-oxazole Chemical compound O1N=C(CCl)C=C1C1=CC=CC=C1Cl UKBHBRICCXLWJZ-UHFFFAOYSA-N 0.000 description 1
- QBWKPGNFQQJGFY-QLFBSQMISA-N 3-[(1r)-1-[(2r,6s)-2,6-dimethylmorpholin-4-yl]ethyl]-n-[6-methyl-3-(1h-pyrazol-4-yl)imidazo[1,2-a]pyrazin-8-yl]-1,2-thiazol-5-amine Chemical compound N1([C@H](C)C2=NSC(NC=3C4=NC=C(N4C=C(C)N=3)C3=CNN=C3)=C2)C[C@H](C)O[C@H](C)C1 QBWKPGNFQQJGFY-QLFBSQMISA-N 0.000 description 1
- 125000000474 3-butynyl group Chemical group [H]C#CC([H])([H])C([H])([H])* 0.000 description 1
- FPHNWFFKQCPXPI-UHFFFAOYSA-N 3-chlorooxolane Chemical compound ClC1CCOC1 FPHNWFFKQCPXPI-UHFFFAOYSA-N 0.000 description 1
- 125000004179 3-chlorophenyl group Chemical group [H]C1=C([H])C(*)=C([H])C(Cl)=C1[H] 0.000 description 1
- 125000006041 3-hexenyl group Chemical group 0.000 description 1
- 125000004207 3-methoxyphenyl group Chemical group [H]C1=C([H])C(*)=C([H])C(OC([H])([H])[H])=C1[H] 0.000 description 1
- 125000006032 3-methyl-3-butenyl group Chemical group 0.000 description 1
- XLWSBDFQAJXCQX-UHFFFAOYSA-N 4-(bromomethyl)-1,2-dichlorobenzene Chemical compound ClC1=CC=C(CBr)C=C1Cl XLWSBDFQAJXCQX-UHFFFAOYSA-N 0.000 description 1
- JVQDTRQMKZMBAU-UHFFFAOYSA-N 4-(bromomethyl)-1-fluoro-2-(trifluoromethyl)benzene Chemical compound FC1=CC=C(CBr)C=C1C(F)(F)F JVQDTRQMKZMBAU-UHFFFAOYSA-N 0.000 description 1
- DHHBTFDTRDVDID-UHFFFAOYSA-N 4-(bromomethyl)-3-(2,6-dichlorophenyl)-5-methyl-1,2-oxazole Chemical compound BrCC1=C(C)ON=C1C1=C(Cl)C=CC=C1Cl DHHBTFDTRDVDID-UHFFFAOYSA-N 0.000 description 1
- QOWSWEBLNVACCL-UHFFFAOYSA-N 4-Bromophenyl acetate Chemical compound OC(=O)CC1=CC=C(Br)C=C1 QOWSWEBLNVACCL-UHFFFAOYSA-N 0.000 description 1
- AZGVPRTXJIEVJG-UHFFFAOYSA-N 4-[[4-(chloromethyl)phenoxy]methyl]-1-fluoro-2-(trifluoromethyl)benzene Chemical compound C1=C(C(F)(F)F)C(F)=CC=C1COC1=CC=C(CCl)C=C1 AZGVPRTXJIEVJG-UHFFFAOYSA-N 0.000 description 1
- CFPZDVAZISWERM-UHFFFAOYSA-N 4-bromo-1,2-dichlorobenzene Chemical compound ClC1=CC=C(Br)C=C1Cl CFPZDVAZISWERM-UHFFFAOYSA-N 0.000 description 1
- XGOCKBMEZPNDPJ-UHFFFAOYSA-N 4-bromo-1-chloro-2-(trifluoromethyl)benzene Chemical compound FC(F)(F)C1=CC(Br)=CC=C1Cl XGOCKBMEZPNDPJ-UHFFFAOYSA-N 0.000 description 1
- BCJVBDBJSMFBRW-UHFFFAOYSA-N 4-diphenylphosphanylbutyl(diphenyl)phosphane Chemical compound C=1C=CC=CC=1P(C=1C=CC=CC=1)CCCCP(C=1C=CC=CC=1)C1=CC=CC=C1 BCJVBDBJSMFBRW-UHFFFAOYSA-N 0.000 description 1
- CKSCMRNFDBWFND-UHFFFAOYSA-N 4-hydroxy-2,3-dihydroinden-1-one Chemical compound OC1=CC=CC2=C1CCC2=O CKSCMRNFDBWFND-UHFFFAOYSA-N 0.000 description 1
- 125000004172 4-methoxyphenyl group Chemical group [H]C1=C([H])C(OC([H])([H])[H])=C([H])C([H])=C1* 0.000 description 1
- 125000004195 4-methylpiperazin-1-yl group Chemical group [H]C([H])([H])N1C([H])([H])C([H])([H])N(*)C([H])([H])C1([H])[H] 0.000 description 1
- SCWNNOCLLOHZIG-UHFFFAOYSA-N 5,6,7,8-tetrahydro-1-naphthol Chemical compound C1CCCC2=C1C=CC=C2O SCWNNOCLLOHZIG-UHFFFAOYSA-N 0.000 description 1
- NSPBTJFAUPZOHK-UHFFFAOYSA-N 5-(chloromethyl)-3-(4-chlorophenyl)-1,2-oxazole Chemical compound O1C(CCl)=CC(C=2C=CC(Cl)=CC=2)=N1 NSPBTJFAUPZOHK-UHFFFAOYSA-N 0.000 description 1
- OCKGFTQIICXDQW-ZEQRLZLVSA-N 5-[(1r)-1-hydroxy-2-[4-[(2r)-2-hydroxy-2-(4-methyl-1-oxo-3h-2-benzofuran-5-yl)ethyl]piperazin-1-yl]ethyl]-4-methyl-3h-2-benzofuran-1-one Chemical compound C1=C2C(=O)OCC2=C(C)C([C@@H](O)CN2CCN(CC2)C[C@H](O)C2=CC=C3C(=O)OCC3=C2C)=C1 OCKGFTQIICXDQW-ZEQRLZLVSA-N 0.000 description 1
- KKFPXGXMSBBNJI-UHFFFAOYSA-N 5-chloro-2-methylphenol Chemical compound CC1=CC=C(Cl)C=C1O KKFPXGXMSBBNJI-UHFFFAOYSA-N 0.000 description 1
- MNQGEQSXFDKAPY-UHFFFAOYSA-N 9h-fluorene-2-carbaldehyde Chemical compound C1=CC=C2C3=CC=C(C=O)C=C3CC2=C1 MNQGEQSXFDKAPY-UHFFFAOYSA-N 0.000 description 1
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 description 1
- 239000005995 Aluminium silicate Substances 0.000 description 1
- VHUUQVKOLVNVRT-UHFFFAOYSA-N Ammonium hydroxide Chemical compound [NH4+].[OH-] VHUUQVKOLVNVRT-UHFFFAOYSA-N 0.000 description 1
- 206010002091 Anaesthesia Diseases 0.000 description 1
- 101710095342 Apolipoprotein B Proteins 0.000 description 1
- 102100040202 Apolipoprotein B-100 Human genes 0.000 description 1
- 108010083590 Apoproteins Proteins 0.000 description 1
- 102000006410 Apoproteins Human genes 0.000 description 1
- 241000416162 Astragalus gummifer Species 0.000 description 1
- XUKUURHRXDUEBC-KAYWLYCHSA-N Atorvastatin Chemical compound C=1C=CC=CC=1C1=C(C=2C=CC(F)=CC=2)N(CC[C@@H](O)C[C@@H](O)CC(O)=O)C(C(C)C)=C1C(=O)NC1=CC=CC=C1 XUKUURHRXDUEBC-KAYWLYCHSA-N 0.000 description 1
- XUKUURHRXDUEBC-UHFFFAOYSA-N Atorvastatin Natural products C=1C=CC=CC=1C1=C(C=2C=CC(F)=CC=2)N(CCC(O)CC(O)CC(O)=O)C(C(C)C)=C1C(=O)NC1=CC=CC=C1 XUKUURHRXDUEBC-UHFFFAOYSA-N 0.000 description 1
- AKCFCWVWRQTNIM-UHFFFAOYSA-N BB.C.C#C.CC.CC.CCCBr.CCCCCCl.CCCCCO.OCCO Chemical compound BB.C.C#C.CC.CC.CCCBr.CCCCCCl.CCCCCO.OCCO AKCFCWVWRQTNIM-UHFFFAOYSA-N 0.000 description 1
- 229910015845 BBr3 Inorganic materials 0.000 description 1
- 229940123208 Biguanide Drugs 0.000 description 1
- XNCOSPRUTUOJCJ-UHFFFAOYSA-N Biguanide Chemical compound NC(N)=NC(N)=N XNCOSPRUTUOJCJ-UHFFFAOYSA-N 0.000 description 1
- 241000283690 Bos taurus Species 0.000 description 1
- CPELXLSAUQHCOX-UHFFFAOYSA-M Bromide Chemical compound [Br-] CPELXLSAUQHCOX-UHFFFAOYSA-M 0.000 description 1
- WCNKPBXPFQNVND-UHFFFAOYSA-N C.CC.CC.CCCCl.CCCO.CCO.CN.N#N Chemical compound C.CC.CC.CCCCl.CCCO.CCO.CN.N#N WCNKPBXPFQNVND-UHFFFAOYSA-N 0.000 description 1
- MURVEEMFEKWKEK-YKVJHHDASA-N C=CCCB(O)O.C=CCCCCC.CCCBr.[2H][2H].[V] Chemical compound C=CCCB(O)O.C=CCCCCC.CCCBr.[2H][2H].[V] MURVEEMFEKWKEK-YKVJHHDASA-N 0.000 description 1
- WOZPPNHSQTYRSA-UHFFFAOYSA-N CC(=O)COC1=CC=C(SCC2=CC=C(OCC3=C(C(F)(F)F)C=C(F)C=C3)C=C2)C2=C1CCC2 Chemical compound CC(=O)COC1=CC=C(SCC2=CC=C(OCC3=C(C(F)(F)F)C=C(F)C=C3)C=C2)C2=C1CCC2 WOZPPNHSQTYRSA-UHFFFAOYSA-N 0.000 description 1
- PIAFYUWOMHWGNR-UHFFFAOYSA-N CC1=C(CCC(=O)O)C=C(O)C(SCC2=CC=C(C3=CC=C(C(F)(F)F)C=C3)C=C2)=C1 Chemical compound CC1=C(CCC(=O)O)C=C(O)C(SCC2=CC=C(C3=CC=C(C(F)(F)F)C=C3)C=C2)=C1 PIAFYUWOMHWGNR-UHFFFAOYSA-N 0.000 description 1
- DWFUGJQDAIBSBB-UHFFFAOYSA-N CC1=C(O)C=CC(N=C=S)=C1 Chemical compound CC1=C(O)C=CC(N=C=S)=C1 DWFUGJQDAIBSBB-UHFFFAOYSA-N 0.000 description 1
- XGQJOJCYTKVLKT-DUXPYHPUSA-N CC1=CC(/C=C/CC2=CC=C(C3=CC=C(C(F)(F)F)C=C3)C=C2)=CC=C1OCC(=O)O Chemical compound CC1=CC(/C=C/CC2=CC=C(C3=CC=C(C(F)(F)F)C=C3)C=C2)=CC=C1OCC(=O)O XGQJOJCYTKVLKT-DUXPYHPUSA-N 0.000 description 1
- VSFVXRLEJMQMQK-UHFFFAOYSA-N CC1=CC(O)=C(C)C=C1N=C=S Chemical compound CC1=CC(O)=C(C)C=C1N=C=S VSFVXRLEJMQMQK-UHFFFAOYSA-N 0.000 description 1
- NXQBRGFTUCQUHD-UHFFFAOYSA-N CC1=CC(SCC2=CC3=C(C=C2)C2=C(C=CC=C2)C3)=CC=C1OCC(=O)O Chemical compound CC1=CC(SCC2=CC3=C(C=C2)C2=C(C=CC=C2)C3)=CC=C1OCC(=O)O NXQBRGFTUCQUHD-UHFFFAOYSA-N 0.000 description 1
- CPXOCABOYRDUEZ-UHFFFAOYSA-N CCCCC1=C(O)C=CC(SC#N)=C1 Chemical compound CCCCC1=C(O)C=CC(SC#N)=C1 CPXOCABOYRDUEZ-UHFFFAOYSA-N 0.000 description 1
- SADDWJMXRSEIHD-UHFFFAOYSA-N CCCCC1=C(OCC(=O)OC)C=CC(SC#N)=C1 Chemical compound CCCCC1=C(OCC(=O)OC)C=CC(SC#N)=C1 SADDWJMXRSEIHD-UHFFFAOYSA-N 0.000 description 1
- XVSMCIRZNONAQE-UHFFFAOYSA-N COC(=O)COC1=C(C)C=C(N=C=S)C(C)=C1 Chemical compound COC(=O)COC1=C(C)C=C(N=C=S)C(C)=C1 XVSMCIRZNONAQE-UHFFFAOYSA-N 0.000 description 1
- XFJXINZNSMIPEA-UHFFFAOYSA-N COC(=O)COC1=C(C)C=C(N=C=S)C(Cl)=C1 Chemical compound COC(=O)COC1=C(C)C=C(N=C=S)C(Cl)=C1 XFJXINZNSMIPEA-UHFFFAOYSA-N 0.000 description 1
- QJJYCBCTNJXRMN-UHFFFAOYSA-N COC(=O)COC1=C(C)C=C(N=C=S)C(OC)=C1 Chemical compound COC(=O)COC1=C(C)C=C(N=C=S)C(OC)=C1 QJJYCBCTNJXRMN-UHFFFAOYSA-N 0.000 description 1
- PBFWPHXFZWSBPL-UHFFFAOYSA-N COC(=O)COC1=C(C)C=C(N=C=S)C=C1 Chemical compound COC(=O)COC1=C(C)C=C(N=C=S)C=C1 PBFWPHXFZWSBPL-UHFFFAOYSA-N 0.000 description 1
- CMIDQEFOSAUIKK-UHFFFAOYSA-N COC(=O)COC1=C(C)C=C(N=C=S)C=C1C Chemical compound COC(=O)COC1=C(C)C=C(N=C=S)C=C1C CMIDQEFOSAUIKK-UHFFFAOYSA-N 0.000 description 1
- WCWAPUWSWCWCOP-UHFFFAOYSA-N COC(=O)COC1=C2CCCCC2=C(N=C=S)C=C1 Chemical compound COC(=O)COC1=C2CCCCC2=C(N=C=S)C=C1 WCWAPUWSWCWCOP-UHFFFAOYSA-N 0.000 description 1
- HNHOEVVLIVZRHS-UHFFFAOYSA-N COC(=O)COC1=CC=C(C(=O)CC2=CC=C(C3=CC=C(C(F)(F)F)C=C3)C=C2)C2=C1CCC2 Chemical compound COC(=O)COC1=CC=C(C(=O)CC2=CC=C(C3=CC=C(C(F)(F)F)C=C3)C=C2)C2=C1CCC2 HNHOEVVLIVZRHS-UHFFFAOYSA-N 0.000 description 1
- XMXFWZYCKMDBHH-UHFFFAOYSA-N COC(=O)COC1=CC=C(N=C=S)C(OC)=C1 Chemical compound COC(=O)COC1=CC=C(N=C=S)C(OC)=C1 XMXFWZYCKMDBHH-UHFFFAOYSA-N 0.000 description 1
- IEMJONPUWNJNME-UHFFFAOYSA-N COC(=O)COC1=CC=C(N=C=S)C=C1 Chemical compound COC(=O)COC1=CC=C(N=C=S)C=C1 IEMJONPUWNJNME-UHFFFAOYSA-N 0.000 description 1
- UXDNUZAMPRZFTL-UHFFFAOYSA-N COC(=O)COC1=CC=C(SCC2=CC3=C(C=C2)C2=C(C=CC=C2)C3)C=C1C Chemical compound COC(=O)COC1=CC=C(SCC2=CC3=C(C=C2)C2=C(C=CC=C2)C3)C=C1C UXDNUZAMPRZFTL-UHFFFAOYSA-N 0.000 description 1
- GXEKVNIVTCGQFZ-UHFFFAOYSA-N COC(=O)COC1=CC=C(SCC2=CC=C(OCC3=C(F)C=C(F)C=C3)C=C2)C2=C1CCC2 Chemical compound COC(=O)COC1=CC=C(SCC2=CC=C(OCC3=C(F)C=C(F)C=C3)C=C2)C2=C1CCC2 GXEKVNIVTCGQFZ-UHFFFAOYSA-N 0.000 description 1
- VPWJKJVLEOIZCB-UHFFFAOYSA-N COC(=O)COC1=CC=C(SCC2=CC=C(OCC3=CC=C(F)C=C3)C=C2)C2=C1CCC2 Chemical compound COC(=O)COC1=CC=C(SCC2=CC=C(OCC3=CC=C(F)C=C3)C=C2)C2=C1CCC2 VPWJKJVLEOIZCB-UHFFFAOYSA-N 0.000 description 1
- LIRNJMGMLJVNSC-UHFFFAOYSA-N COC1=CC(O)=CC=C1N=C=S Chemical compound COC1=CC(O)=CC=C1N=C=S LIRNJMGMLJVNSC-UHFFFAOYSA-N 0.000 description 1
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 1
- 241000282472 Canis lupus familiaris Species 0.000 description 1
- 241000283707 Capra Species 0.000 description 1
- 208000005623 Carcinogenesis Diseases 0.000 description 1
- KKVZAVRSVHUSPL-GQCTYLIASA-N Cassiastearoptene Chemical compound COC1=CC=CC=C1\C=C\C=O KKVZAVRSVHUSPL-GQCTYLIASA-N 0.000 description 1
- PTHCMJGKKRQCBF-UHFFFAOYSA-N Cellulose, microcrystalline Chemical compound OC1C(O)C(OC)OC(CO)C1OC1C(O)C(O)C(OC)C(CO)O1 PTHCMJGKKRQCBF-UHFFFAOYSA-N 0.000 description 1
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 description 1
- 229920001268 Cholestyramine Polymers 0.000 description 1
- 208000017667 Chronic Disease Diseases 0.000 description 1
- KRKNYBCHXYNGOX-UHFFFAOYSA-K Citrate Chemical compound [O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O KRKNYBCHXYNGOX-UHFFFAOYSA-K 0.000 description 1
- VBOSZVRRXDHOMT-UHFFFAOYSA-N ClCC1(CC(=CC=C1)OCC1=CC=CC=C1)F Chemical compound ClCC1(CC(=CC=C1)OCC1=CC=CC=C1)F VBOSZVRRXDHOMT-UHFFFAOYSA-N 0.000 description 1
- 229920002911 Colestipol Polymers 0.000 description 1
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 1
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 description 1
- FBPFZTCFMRRESA-JGWLITMVSA-N D-glucitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-JGWLITMVSA-N 0.000 description 1
- FEWJPZIEWOKRBE-JCYAYHJZSA-N Dextrotartaric acid Chemical compound OC(=O)[C@H](O)[C@@H](O)C(O)=O FEWJPZIEWOKRBE-JCYAYHJZSA-N 0.000 description 1
- 206010012735 Diarrhoea Diseases 0.000 description 1
- 235000019739 Dicalciumphosphate Nutrition 0.000 description 1
- 239000006144 Dulbecco’s modified Eagle's medium Substances 0.000 description 1
- 238000002965 ELISA Methods 0.000 description 1
- LVGKNOAMLMIIKO-UHFFFAOYSA-N Elaidinsaeure-aethylester Natural products CCCCCCCCC=CCCCCCCCC(=O)OCC LVGKNOAMLMIIKO-UHFFFAOYSA-N 0.000 description 1
- IAYPIBMASNFSPL-UHFFFAOYSA-N Ethylene oxide Chemical compound C1CO1 IAYPIBMASNFSPL-UHFFFAOYSA-N 0.000 description 1
- KSWASNDTRBHTNJ-UHFFFAOYSA-N FC(F)(F)C1=CC(C2=CC=C(CCl)C=C2)=CC=C1Cl Chemical compound FC(F)(F)C1=CC(C2=CC=C(CCl)C=C2)=CC=C1Cl KSWASNDTRBHTNJ-UHFFFAOYSA-N 0.000 description 1
- AHEFTHIHJDXTRP-QPJJXVBHSA-N FC1=CC(/C=C/C2=CC=C(CCl)C=C2)=CC=C1 Chemical compound FC1=CC(/C=C/C2=CC=C(CCl)C=C2)=CC=C1 AHEFTHIHJDXTRP-QPJJXVBHSA-N 0.000 description 1
- HWBDTYKIMZPAJV-UHFFFAOYSA-N FC1=CC=C(COC2=CC=C(CCl)C=C2)C=C1 Chemical compound FC1=CC=C(COC2=CC=C(CCl)C=C2)C=C1 HWBDTYKIMZPAJV-UHFFFAOYSA-N 0.000 description 1
- 241000282326 Felis catus Species 0.000 description 1
- 239000004606 Fillers/Extenders Substances 0.000 description 1
- 238000005727 Friedel-Crafts reaction Methods 0.000 description 1
- VZCYOOQTPOCHFL-OWOJBTEDSA-N Fumaric acid Chemical compound OC(=O)\C=C\C(O)=O VZCYOOQTPOCHFL-OWOJBTEDSA-N 0.000 description 1
- 241000206672 Gelidium Species 0.000 description 1
- HEMJJKBWTPKOJG-UHFFFAOYSA-N Gemfibrozil Chemical compound CC1=CC=C(C)C(OCCCC(C)(C)C(O)=O)=C1 HEMJJKBWTPKOJG-UHFFFAOYSA-N 0.000 description 1
- 108010023302 HDL Cholesterol Proteins 0.000 description 1
- 241000463109 Haloprofundus marisrubri Species 0.000 description 1
- CPELXLSAUQHCOX-UHFFFAOYSA-N Hydrogen bromide Chemical compound Br CPELXLSAUQHCOX-UHFFFAOYSA-N 0.000 description 1
- 102000004286 Hydroxymethylglutaryl CoA Reductases Human genes 0.000 description 1
- 108090000895 Hydroxymethylglutaryl CoA Reductases Proteins 0.000 description 1
- 208000031226 Hyperlipidaemia Diseases 0.000 description 1
- 208000013016 Hypoglycemia Diseases 0.000 description 1
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 1
- 229910021578 Iron(III) chloride Inorganic materials 0.000 description 1
- 238000008214 LDL Cholesterol Methods 0.000 description 1
- 108010028554 LDL Cholesterol Proteins 0.000 description 1
- 102000000853 LDL receptors Human genes 0.000 description 1
- 108010001831 LDL receptors Proteins 0.000 description 1
- JVTAAEKCZFNVCJ-UHFFFAOYSA-M Lactate Chemical compound CC(O)C([O-])=O JVTAAEKCZFNVCJ-UHFFFAOYSA-M 0.000 description 1
- 240000007472 Leucaena leucocephala Species 0.000 description 1
- 235000010643 Leucaena leucocephala Nutrition 0.000 description 1
- 102000004895 Lipoproteins Human genes 0.000 description 1
- 108090001030 Lipoproteins Proteins 0.000 description 1
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 description 1
- 241000282560 Macaca mulatta Species 0.000 description 1
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 1
- 240000003183 Manihot esculenta Species 0.000 description 1
- 235000016735 Manihot esculenta subsp esculenta Nutrition 0.000 description 1
- 229930195725 Mannitol Natural products 0.000 description 1
- AFVFQIVMOAPDHO-UHFFFAOYSA-N Methanesulfonic acid Chemical compound CS(O)(=O)=O AFVFQIVMOAPDHO-UHFFFAOYSA-N 0.000 description 1
- 229920000168 Microcrystalline cellulose Polymers 0.000 description 1
- PCZOHLXUXFIOCF-UHFFFAOYSA-N Monacolin X Natural products C12C(OC(=O)C(C)CC)CC(C)C=C2C=CC(C)C1CCC1CC(O)CC(=O)O1 PCZOHLXUXFIOCF-UHFFFAOYSA-N 0.000 description 1
- PHSPJQZRQAJPPF-UHFFFAOYSA-N N-alpha-Methylhistamine Chemical compound CNCCC1=CN=CN1 PHSPJQZRQAJPPF-UHFFFAOYSA-N 0.000 description 1
- 206010028980 Neoplasm Diseases 0.000 description 1
- PVNIIMVLHYAWGP-UHFFFAOYSA-N Niacin Chemical class OC(=O)C1=CC=CN=C1 PVNIIMVLHYAWGP-UHFFFAOYSA-N 0.000 description 1
- NHNBFGGVMKEFGY-UHFFFAOYSA-N Nitrate Chemical compound [O-][N+]([O-])=O NHNBFGGVMKEFGY-UHFFFAOYSA-N 0.000 description 1
- 102000007399 Nuclear hormone receptor Human genes 0.000 description 1
- 108020005497 Nuclear hormone receptor Proteins 0.000 description 1
- KSQOBVUINFBJNJ-UHFFFAOYSA-M O.O=C([O-])COC1=CC=C(SCC2=CC(Cl)=C(OCC3=CC=C(C(F)(F)F)C=C3)C(Cl)=C2)C2=C1CCC2.[Li+] Chemical compound O.O=C([O-])COC1=CC=C(SCC2=CC(Cl)=C(OCC3=CC=C(C(F)(F)F)C=C3)C(Cl)=C2)C2=C1CCC2.[Li+] KSQOBVUINFBJNJ-UHFFFAOYSA-M 0.000 description 1
- RWZBFNOVEOLLEB-UHFFFAOYSA-N OC1=CC=C(N=C=S)C2=C1CCCC2 Chemical compound OC1=CC=C(N=C=S)C2=C1CCCC2 RWZBFNOVEOLLEB-UHFFFAOYSA-N 0.000 description 1
- HIPHYBWWZWRZOV-UHFFFAOYSA-N OC1=CC=C(N=C=S)C=C1 Chemical compound OC1=CC=C(N=C=S)C=C1 HIPHYBWWZWRZOV-UHFFFAOYSA-N 0.000 description 1
- KYQTXGQQLIPALH-UHFFFAOYSA-N OCC1=CC(Cl)=C(OCC2=CC=C(C(F)(F)F)C=C2)C(Cl)=C1 Chemical compound OCC1=CC(Cl)=C(OCC2=CC=C(C(F)(F)F)C=C2)C(Cl)=C1 KYQTXGQQLIPALH-UHFFFAOYSA-N 0.000 description 1
- XMOLAFPVDQWPNV-QPJJXVBHSA-N OCC1=CC=C(/C=C/C2=CC=CC(F)=C2)C=C1 Chemical compound OCC1=CC=C(/C=C/C2=CC=CC(F)=C2)C=C1 XMOLAFPVDQWPNV-QPJJXVBHSA-N 0.000 description 1
- 206010030113 Oedema Diseases 0.000 description 1
- 241000283973 Oryctolagus cuniculus Species 0.000 description 1
- MUBZPKHOEPUJKR-UHFFFAOYSA-N Oxalic acid Chemical compound OC(=O)C(O)=O MUBZPKHOEPUJKR-UHFFFAOYSA-N 0.000 description 1
- 229910019142 PO4 Inorganic materials 0.000 description 1
- 108010015181 PPAR delta Proteins 0.000 description 1
- 235000019483 Peanut oil Nutrition 0.000 description 1
- 241001494479 Pecora Species 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 1
- 229920003171 Poly (ethylene oxide) Polymers 0.000 description 1
- 229920001213 Polysorbate 20 Polymers 0.000 description 1
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 1
- TUZYXOIXSAXUGO-UHFFFAOYSA-N Pravastatin Natural products C1=CC(C)C(CCC(O)CC(O)CC(O)=O)C2C(OC(=O)C(C)CC)CC(O)C=C21 TUZYXOIXSAXUGO-UHFFFAOYSA-N 0.000 description 1
- GOOHAUXETOMSMM-UHFFFAOYSA-N Propylene oxide Chemical compound CC1CO1 GOOHAUXETOMSMM-UHFFFAOYSA-N 0.000 description 1
- RYMZZMVNJRMUDD-UHFFFAOYSA-N SJ000286063 Natural products C12C(OC(=O)C(C)(C)CC)CC(C)C=C2C=CC(C)C1CCC1CC(O)CC(=O)O1 RYMZZMVNJRMUDD-UHFFFAOYSA-N 0.000 description 1
- 240000004808 Saccharomyces cerevisiae Species 0.000 description 1
- VMHLLURERBWHNL-UHFFFAOYSA-M Sodium acetate Chemical compound [Na+].CC([O-])=O VMHLLURERBWHNL-UHFFFAOYSA-M 0.000 description 1
- DBMJMQXJHONAFJ-UHFFFAOYSA-M Sodium laurylsulphate Chemical compound [Na+].CCCCCCCCCCCCOS([O-])(=O)=O DBMJMQXJHONAFJ-UHFFFAOYSA-M 0.000 description 1
- 235000002595 Solanum tuberosum Nutrition 0.000 description 1
- 244000061456 Solanum tuberosum Species 0.000 description 1
- 235000021355 Stearic acid Nutrition 0.000 description 1
- SSZBUIDZHHWXNJ-UHFFFAOYSA-N Stearinsaeure-hexadecylester Natural products CCCCCCCCCCCCCCCCCC(=O)OCCCCCCCCCCCCCCCC SSZBUIDZHHWXNJ-UHFFFAOYSA-N 0.000 description 1
- 108010085012 Steroid Receptors Proteins 0.000 description 1
- 102000007451 Steroid Receptors Human genes 0.000 description 1
- 241000282887 Suidae Species 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-L Sulfate Chemical compound [O-]S([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-L 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 1
- 208000001435 Thromboembolism Diseases 0.000 description 1
- 229920001615 Tragacanth Polymers 0.000 description 1
- 102000040945 Transcription factor Human genes 0.000 description 1
- 108091023040 Transcription factor Proteins 0.000 description 1
- 108010021119 Trichosanthin Proteins 0.000 description 1
- 240000008042 Zea mays Species 0.000 description 1
- 235000005824 Zea mays ssp. parviglumis Nutrition 0.000 description 1
- 235000002017 Zea mays subsp mays Nutrition 0.000 description 1
- 239000001089 [(2R)-oxolan-2-yl]methanol Substances 0.000 description 1
- ALMFIOZYDASRRC-UHFFFAOYSA-N [4-(trifluoromethyl)phenyl]boronic acid Chemical compound OB(O)C1=CC=C(C(F)(F)F)C=C1 ALMFIOZYDASRRC-UHFFFAOYSA-N 0.000 description 1
- 206010000059 abdominal discomfort Diseases 0.000 description 1
- 239000003655 absorption accelerator Substances 0.000 description 1
- 229960002632 acarbose Drugs 0.000 description 1
- XUFXOAAUWZOOIT-UHFFFAOYSA-N acarviostatin I01 Natural products OC1C(O)C(NC2C(C(O)C(O)C(CO)=C2)O)C(C)OC1OC(C(C1O)O)C(CO)OC1OC1C(CO)OC(O)C(O)C1O XUFXOAAUWZOOIT-UHFFFAOYSA-N 0.000 description 1
- 229940022663 acetate Drugs 0.000 description 1
- 230000003213 activating effect Effects 0.000 description 1
- 125000005073 adamantyl group Chemical group C12(CC3CC(CC(C1)C3)C2)* 0.000 description 1
- 210000000577 adipose tissue Anatomy 0.000 description 1
- 239000003463 adsorbent Substances 0.000 description 1
- 239000008272 agar Substances 0.000 description 1
- 239000000783 alginic acid Substances 0.000 description 1
- 229960001126 alginic acid Drugs 0.000 description 1
- 150000004781 alginic acids Chemical class 0.000 description 1
- 239000003513 alkali Substances 0.000 description 1
- 150000001342 alkaline earth metals Chemical class 0.000 description 1
- 150000001348 alkyl chlorides Chemical class 0.000 description 1
- 150000001350 alkyl halides Chemical class 0.000 description 1
- 229910052782 aluminium Inorganic materials 0.000 description 1
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 1
- 235000012211 aluminium silicate Nutrition 0.000 description 1
- VZTDIZULWFCMLS-UHFFFAOYSA-N ammonium formate Chemical compound [NH4+].[O-]C=O VZTDIZULWFCMLS-UHFFFAOYSA-N 0.000 description 1
- 230000037005 anaesthesia Effects 0.000 description 1
- 229940040526 anhydrous sodium acetate Drugs 0.000 description 1
- 239000003242 anti bacterial agent Substances 0.000 description 1
- 230000000844 anti-bacterial effect Effects 0.000 description 1
- 239000003429 antifungal agent Substances 0.000 description 1
- 229940121375 antifungal agent Drugs 0.000 description 1
- 238000013459 approach Methods 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- 125000003710 aryl alkyl group Chemical group 0.000 description 1
- 150000001502 aryl halides Chemical class 0.000 description 1
- 238000003556 assay Methods 0.000 description 1
- 229960005370 atorvastatin Drugs 0.000 description 1
- 125000002393 azetidinyl group Chemical group 0.000 description 1
- 125000004069 aziridinyl group Chemical group 0.000 description 1
- 230000006399 behavior Effects 0.000 description 1
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid Chemical compound OC(=O)C1=CC=CC=C1 WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 description 1
- 229960002903 benzyl benzoate Drugs 0.000 description 1
- AGEZXYOZHKGVCM-UHFFFAOYSA-N benzyl bromide Chemical compound BrCC1=CC=CC=C1 AGEZXYOZHKGVCM-UHFFFAOYSA-N 0.000 description 1
- 125000002619 bicyclic group Chemical group 0.000 description 1
- 229920000080 bile acid sequestrant Polymers 0.000 description 1
- 229940096699 bile acid sequestrants Drugs 0.000 description 1
- 239000011230 binding agent Substances 0.000 description 1
- 238000004166 bioassay Methods 0.000 description 1
- 230000031018 biological processes and functions Effects 0.000 description 1
- 239000004305 biphenyl Substances 0.000 description 1
- 235000010290 biphenyl Nutrition 0.000 description 1
- 239000007844 bleaching agent Substances 0.000 description 1
- 230000036760 body temperature Effects 0.000 description 1
- ZADPBFCGQRWHPN-UHFFFAOYSA-N boronic acid Chemical compound OBO ZADPBFCGQRWHPN-UHFFFAOYSA-N 0.000 description 1
- 239000000872 buffer Substances 0.000 description 1
- 239000006172 buffering agent Substances 0.000 description 1
- 235000019437 butane-1,3-diol Nutrition 0.000 description 1
- 239000011575 calcium Substances 0.000 description 1
- 229910052791 calcium Inorganic materials 0.000 description 1
- 229910000019 calcium carbonate Inorganic materials 0.000 description 1
- 235000010216 calcium carbonate Nutrition 0.000 description 1
- FATUQANACHZLRT-KMRXSBRUSA-L calcium glucoheptonate Chemical compound [Ca+2].OC[C@@H](O)[C@@H](O)[C@H](O)[C@@H](O)C(O)C([O-])=O.OC[C@@H](O)[C@@H](O)[C@H](O)[C@@H](O)C(O)C([O-])=O FATUQANACHZLRT-KMRXSBRUSA-L 0.000 description 1
- 239000001506 calcium phosphate Substances 0.000 description 1
- CJZGTCYPCWQAJB-UHFFFAOYSA-L calcium stearate Chemical compound [Ca+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O CJZGTCYPCWQAJB-UHFFFAOYSA-L 0.000 description 1
- 235000013539 calcium stearate Nutrition 0.000 description 1
- 239000008116 calcium stearate Substances 0.000 description 1
- 230000036952 cancer formation Effects 0.000 description 1
- CREMABGTGYGIQB-UHFFFAOYSA-N carbon carbon Chemical compound C.C CREMABGTGYGIQB-UHFFFAOYSA-N 0.000 description 1
- 239000011203 carbon fibre reinforced carbon Substances 0.000 description 1
- 231100000504 carcinogenesis Toxicity 0.000 description 1
- 239000004359 castor oil Substances 0.000 description 1
- 235000019438 castor oil Nutrition 0.000 description 1
- 150000001768 cations Chemical class 0.000 description 1
- 230000024245 cell differentiation Effects 0.000 description 1
- 230000001413 cellular effect Effects 0.000 description 1
- 229960000541 cetyl alcohol Drugs 0.000 description 1
- 239000003610 charcoal Substances 0.000 description 1
- 239000003153 chemical reaction reagent Substances 0.000 description 1
- 229910001914 chlorine tetroxide Inorganic materials 0.000 description 1
- 229960004926 chlorobutanol Drugs 0.000 description 1
- 125000004218 chloromethyl group Chemical group [H]C([H])(Cl)* 0.000 description 1
- 229960001214 clofibrate Drugs 0.000 description 1
- KNHUKKLJHYUCFP-UHFFFAOYSA-N clofibrate Chemical compound CCOC(=O)C(C)(C)OC1=CC=C(Cl)C=C1 KNHUKKLJHYUCFP-UHFFFAOYSA-N 0.000 description 1
- 239000011248 coating agent Substances 0.000 description 1
- 229940110456 cocoa butter Drugs 0.000 description 1
- 235000019868 cocoa butter Nutrition 0.000 description 1
- 229910052681 coesite Inorganic materials 0.000 description 1
- GMRWGQCZJGVHKL-UHFFFAOYSA-N colestipol Chemical compound ClCC1CO1.NCCNCCNCCNCCN GMRWGQCZJGVHKL-UHFFFAOYSA-N 0.000 description 1
- 229960002604 colestipol Drugs 0.000 description 1
- 229940125773 compound 10 Drugs 0.000 description 1
- 229940125846 compound 25 Drugs 0.000 description 1
- 229940125877 compound 31 Drugs 0.000 description 1
- 229940127573 compound 38 Drugs 0.000 description 1
- 229940125898 compound 5 Drugs 0.000 description 1
- 235000009508 confectionery Nutrition 0.000 description 1
- 238000007796 conventional method Methods 0.000 description 1
- 235000005822 corn Nutrition 0.000 description 1
- 235000012343 cottonseed oil Nutrition 0.000 description 1
- 239000002385 cottonseed oil Substances 0.000 description 1
- 229910052906 cristobalite Inorganic materials 0.000 description 1
- 125000000392 cycloalkenyl group Chemical group 0.000 description 1
- HWEQKSVYKBUIIK-UHFFFAOYSA-N cyclobuta-1,3-diene Chemical compound C1=CC=C1 HWEQKSVYKBUIIK-UHFFFAOYSA-N 0.000 description 1
- CFBGXYDUODCMNS-UHFFFAOYSA-N cyclobutene Chemical compound C1CC=C1 CFBGXYDUODCMNS-UHFFFAOYSA-N 0.000 description 1
- 125000001995 cyclobutyl group Chemical group [H]C1([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- ZXIJMRYMVAMXQP-UHFFFAOYSA-N cycloheptene Chemical compound C1CCC=CCC1 ZXIJMRYMVAMXQP-UHFFFAOYSA-N 0.000 description 1
- 125000000582 cycloheptyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- 125000001559 cyclopropyl group Chemical group [H]C1([H])C([H])([H])C1([H])* 0.000 description 1
- 125000004855 decalinyl group Chemical group C1(CCCC2CCCCC12)* 0.000 description 1
- 230000003111 delayed effect Effects 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 230000018109 developmental process Effects 0.000 description 1
- 125000004663 dialkyl amino group Chemical group 0.000 description 1
- NEFBYIFKOOEVPA-UHFFFAOYSA-K dicalcium phosphate Chemical compound [Ca+2].[Ca+2].[O-]P([O-])([O-])=O NEFBYIFKOOEVPA-UHFFFAOYSA-K 0.000 description 1
- 229940038472 dicalcium phosphate Drugs 0.000 description 1
- 229910000390 dicalcium phosphate Inorganic materials 0.000 description 1
- 235000005911 diet Nutrition 0.000 description 1
- 230000000378 dietary effect Effects 0.000 description 1
- FAMRKDQNMBBFBR-BQYQJAHWSA-N diethyl azodicarboxylate Substances CCOC(=O)\N=N\C(=O)OCC FAMRKDQNMBBFBR-BQYQJAHWSA-N 0.000 description 1
- WYACBZDAHNBPPB-UHFFFAOYSA-N diethyl oxalate Chemical compound CCOC(=O)C(=O)OCC WYACBZDAHNBPPB-UHFFFAOYSA-N 0.000 description 1
- 230000004069 differentiation Effects 0.000 description 1
- UGMCXQCYOVCMTB-UHFFFAOYSA-K dihydroxy(stearato)aluminium Chemical compound CCCCCCCCCCCCCCCCCC(=O)O[Al](O)O UGMCXQCYOVCMTB-UHFFFAOYSA-K 0.000 description 1
- 108700003601 dimethylglycine Proteins 0.000 description 1
- POULHZVOKOAJMA-UHFFFAOYSA-M dodecanoate Chemical compound CCCCCCCCCCCC([O-])=O POULHZVOKOAJMA-UHFFFAOYSA-M 0.000 description 1
- 239000008298 dragée Substances 0.000 description 1
- 239000006196 drop Substances 0.000 description 1
- 230000005584 early death Effects 0.000 description 1
- 239000002702 enteric coating Substances 0.000 description 1
- 238000009505 enteric coating Methods 0.000 description 1
- 239000003797 essential amino acid Substances 0.000 description 1
- 235000020776 essential amino acid Nutrition 0.000 description 1
- BEFDCLMNVWHSGT-UHFFFAOYSA-N ethenylcyclopentane Chemical compound C=CC1CCCC1 BEFDCLMNVWHSGT-UHFFFAOYSA-N 0.000 description 1
- JHVRVXDQXHJMAK-UHFFFAOYSA-N ethyl 2,4-dioxo-4-[4-(trifluoromethyl)phenyl]butanoate Chemical compound CCOC(=O)C(=O)CC(=O)C1=CC=C(C(F)(F)F)C=C1 JHVRVXDQXHJMAK-UHFFFAOYSA-N 0.000 description 1
- 229940093499 ethyl acetate Drugs 0.000 description 1
- FAMRKDQNMBBFBR-UHFFFAOYSA-N ethyl n-ethoxycarbonyliminocarbamate Chemical compound CCOC(=O)N=NC(=O)OCC FAMRKDQNMBBFBR-UHFFFAOYSA-N 0.000 description 1
- LVGKNOAMLMIIKO-QXMHVHEDSA-N ethyl oleate Chemical compound CCCCCCCC\C=C/CCCCCCCC(=O)OCC LVGKNOAMLMIIKO-QXMHVHEDSA-N 0.000 description 1
- 229940093471 ethyl oleate Drugs 0.000 description 1
- 229940117927 ethylene oxide Drugs 0.000 description 1
- 239000013613 expression plasmid Substances 0.000 description 1
- 239000003885 eye ointment Substances 0.000 description 1
- 229940125753 fibrate Drugs 0.000 description 1
- 238000011049 filling Methods 0.000 description 1
- 239000012065 filter cake Substances 0.000 description 1
- 239000012467 final product Substances 0.000 description 1
- 239000000796 flavoring agent Substances 0.000 description 1
- 125000006419 fluorocyclopropyl group Chemical group 0.000 description 1
- 125000003784 fluoroethyl group Chemical group [H]C([H])(F)C([H])([H])* 0.000 description 1
- 235000019138 food restriction Nutrition 0.000 description 1
- 125000002485 formyl group Chemical group [H]C(*)=O 0.000 description 1
- 239000012737 fresh medium Substances 0.000 description 1
- 125000000524 functional group Chemical group 0.000 description 1
- 125000002541 furyl group Chemical group 0.000 description 1
- 210000001035 gastrointestinal tract Anatomy 0.000 description 1
- ZTQSADJAYQOCDD-UHFFFAOYSA-N ginsenoside-Rd2 Natural products C1CC(C2(CCC3C(C)(C)C(OC4C(C(O)C(O)C(CO)O4)O)CCC3(C)C2CC2O)C)(C)C2C1C(C)(CCC=C(C)C)OC(C(C(O)C1O)O)OC1COC1OCC(O)C(O)C1O ZTQSADJAYQOCDD-UHFFFAOYSA-N 0.000 description 1
- 239000012362 glacial acetic acid Substances 0.000 description 1
- 235000001727 glucose Nutrition 0.000 description 1
- 230000002641 glycemic effect Effects 0.000 description 1
- YQEMORVAKMFKLG-UHFFFAOYSA-N glycerine monostearate Natural products CCCCCCCCCCCCCCCCCC(=O)OC(CO)CO YQEMORVAKMFKLG-UHFFFAOYSA-N 0.000 description 1
- SVUQHVRAGMNPLW-UHFFFAOYSA-N glycerol monostearate Natural products CCCCCCCCCCCCCCCCC(=O)OCC(O)CO SVUQHVRAGMNPLW-UHFFFAOYSA-N 0.000 description 1
- ZEMPKEQAKRGZGQ-XOQCFJPHSA-N glycerol triricinoleate Natural products CCCCCC[C@@H](O)CC=CCCCCCCCC(=O)OC[C@@H](COC(=O)CCCCCCCC=CC[C@@H](O)CCCCCC)OC(=O)CCCCCCCC=CC[C@H](O)CCCCCC ZEMPKEQAKRGZGQ-XOQCFJPHSA-N 0.000 description 1
- 210000002216 heart Anatomy 0.000 description 1
- 238000010438 heat treatment Methods 0.000 description 1
- 230000002440 hepatic effect Effects 0.000 description 1
- 231100000304 hepatotoxicity Toxicity 0.000 description 1
- 125000004474 heteroalkylene group Chemical group 0.000 description 1
- BXWNKGSJHAJOGX-UHFFFAOYSA-N hexadecan-1-ol Chemical compound CCCCCCCCCCCCCCCCO BXWNKGSJHAJOGX-UHFFFAOYSA-N 0.000 description 1
- IPCSVZSSVZVIGE-UHFFFAOYSA-M hexadecanoate Chemical compound CCCCCCCCCCCCCCCC([O-])=O IPCSVZSSVZVIGE-UHFFFAOYSA-M 0.000 description 1
- 239000003906 humectant Substances 0.000 description 1
- ZMZDMBWJUHKJPS-UHFFFAOYSA-N hydrogen thiocyanate Natural products SC#N ZMZDMBWJUHKJPS-UHFFFAOYSA-N 0.000 description 1
- 238000005984 hydrogenation reaction Methods 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-M hydrogensulfate Chemical compound OS([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-M 0.000 description 1
- 230000007062 hydrolysis Effects 0.000 description 1
- 238000006460 hydrolysis reaction Methods 0.000 description 1
- 125000004356 hydroxy functional group Chemical group O* 0.000 description 1
- 206010020718 hyperplasia Diseases 0.000 description 1
- 230000002218 hypoglycaemic effect Effects 0.000 description 1
- 239000005457 ice water Substances 0.000 description 1
- 125000002883 imidazolyl group Chemical group 0.000 description 1
- 238000011065 in-situ storage Methods 0.000 description 1
- 230000028709 inflammatory response Effects 0.000 description 1
- 238000002347 injection Methods 0.000 description 1
- 239000007924 injection Substances 0.000 description 1
- 230000003914 insulin secretion Effects 0.000 description 1
- 125000002346 iodo group Chemical group I* 0.000 description 1
- RBTARNINKXHZNM-UHFFFAOYSA-K iron trichloride Chemical compound Cl[Fe](Cl)Cl RBTARNINKXHZNM-UHFFFAOYSA-K 0.000 description 1
- 238000002955 isolation Methods 0.000 description 1
- 125000001786 isothiazolyl group Chemical group 0.000 description 1
- 239000007951 isotonicity adjuster Substances 0.000 description 1
- 125000000842 isoxazolyl group Chemical group 0.000 description 1
- ZLVXBBHTMQJRSX-VMGNSXQWSA-N jdtic Chemical compound C1([C@]2(C)CCN(C[C@@H]2C)C[C@H](C(C)C)NC(=O)[C@@H]2NCC3=CC(O)=CC=C3C2)=CC=CC(O)=C1 ZLVXBBHTMQJRSX-VMGNSXQWSA-N 0.000 description 1
- NLYAJNPCOHFWQQ-UHFFFAOYSA-N kaolin Chemical compound O.O.O=[Al]O[Si](=O)O[Si](=O)O[Al]=O NLYAJNPCOHFWQQ-UHFFFAOYSA-N 0.000 description 1
- 210000003292 kidney cell Anatomy 0.000 description 1
- 229940099584 lactobionate Drugs 0.000 description 1
- JYTUSYBCFIZPBE-AMTLMPIISA-N lactobionic acid Chemical compound OC(=O)[C@H](O)[C@@H](O)[C@@H]([C@H](O)CO)O[C@@H]1O[C@H](CO)[C@H](O)[C@H](O)[C@H]1O JYTUSYBCFIZPBE-AMTLMPIISA-N 0.000 description 1
- 229940070765 laurate Drugs 0.000 description 1
- 239000000787 lecithin Substances 0.000 description 1
- 235000010445 lecithin Nutrition 0.000 description 1
- 229940067606 lecithin Drugs 0.000 description 1
- 230000000670 limiting effect Effects 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- 239000012280 lithium aluminium hydride Substances 0.000 description 1
- 229910003002 lithium salt Inorganic materials 0.000 description 1
- 159000000002 lithium salts Chemical class 0.000 description 1
- 230000007774 longterm Effects 0.000 description 1
- 229960004844 lovastatin Drugs 0.000 description 1
- PCZOHLXUXFIOCF-BXMDZJJMSA-N lovastatin Chemical compound C([C@H]1[C@@H](C)C=CC2=C[C@H](C)C[C@@H]([C@H]12)OC(=O)[C@@H](C)CC)C[C@@H]1C[C@@H](O)CC(=O)O1 PCZOHLXUXFIOCF-BXMDZJJMSA-N 0.000 description 1
- QLJODMDSTUBWDW-UHFFFAOYSA-N lovastatin hydroxy acid Natural products C1=CC(C)C(CCC(O)CC(O)CC(O)=O)C2C(OC(=O)C(C)CC)CC(C)C=C21 QLJODMDSTUBWDW-UHFFFAOYSA-N 0.000 description 1
- 239000000314 lubricant Substances 0.000 description 1
- 125000000040 m-tolyl group Chemical group [H]C1=C([H])C(*)=C([H])C(=C1[H])C([H])([H])[H] 0.000 description 1
- 239000011777 magnesium Substances 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- 238000012423 maintenance Methods 0.000 description 1
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 description 1
- 230000036210 malignancy Effects 0.000 description 1
- 239000000594 mannitol Substances 0.000 description 1
- 235000010355 mannitol Nutrition 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- 230000001404 mediated effect Effects 0.000 description 1
- 239000002609 medium Substances 0.000 description 1
- 238000002844 melting Methods 0.000 description 1
- 230000008018 melting Effects 0.000 description 1
- 239000002207 metabolite Substances 0.000 description 1
- XZWYZXLIPXDOLR-UHFFFAOYSA-N metformin Chemical compound CN(C)C(=N)NC(N)=N XZWYZXLIPXDOLR-UHFFFAOYSA-N 0.000 description 1
- 229960003105 metformin Drugs 0.000 description 1
- GBMDVOWEEQVZKZ-UHFFFAOYSA-N methanol;hydrate Chemical compound O.OC GBMDVOWEEQVZKZ-UHFFFAOYSA-N 0.000 description 1
- KOLORTBZWHYINT-UHFFFAOYSA-N methyl 2-(2,5-dimethyl-4-thiocyanatophenoxy)acetate Chemical compound COC(=O)COC1=CC(C)=C(SC#N)C=C1C KOLORTBZWHYINT-UHFFFAOYSA-N 0.000 description 1
- OMAUDUHRMMRHBV-UHFFFAOYSA-N methyl 2-(2-methylphenoxy)acetate Chemical compound COC(=O)COC1=CC=CC=C1C OMAUDUHRMMRHBV-UHFFFAOYSA-N 0.000 description 1
- DVGFIWUVYWRFLJ-UHFFFAOYSA-N methyl 2-(2-propyl-4-sulfanylphenoxy)acetate Chemical compound CCCC1=CC(S)=CC=C1OCC(=O)OC DVGFIWUVYWRFLJ-UHFFFAOYSA-N 0.000 description 1
- YGMOSUYOYNUHFL-UHFFFAOYSA-N methyl 2-(4-thiocyanatophenoxy)acetate Chemical compound COC(=O)COC1=CC=C(SC#N)C=C1 YGMOSUYOYNUHFL-UHFFFAOYSA-N 0.000 description 1
- BOLRZZUKIOUFOM-UHFFFAOYSA-N methyl 2-[(4-thiocyanato-5,6,7,8-tetrahydronaphthalen-1-yl)oxy]acetate Chemical compound C1CCCC2=C1C(SC#N)=CC=C2OCC(=O)OC BOLRZZUKIOUFOM-UHFFFAOYSA-N 0.000 description 1
- YQJLDAKZPWHHFN-UHFFFAOYSA-N methyl 4,5-dimethyl-2-[[5-[2-(trifluoromethyl)phenyl]furan-2-carbonyl]amino]thiophene-3-carboxylate Chemical compound CC1=C(C)SC(NC(=O)C=2OC(=CC=2)C=2C(=CC=CC=2)C(F)(F)F)=C1C(=O)OC YQJLDAKZPWHHFN-UHFFFAOYSA-N 0.000 description 1
- LSEFCHWGJNHZNT-UHFFFAOYSA-M methyl(triphenyl)phosphanium;bromide Chemical compound [Br-].C=1C=CC=CC=1[P+](C=1C=CC=CC=1)(C)C1=CC=CC=C1 LSEFCHWGJNHZNT-UHFFFAOYSA-M 0.000 description 1
- 244000005700 microbiome Species 0.000 description 1
- 229940016286 microcrystalline cellulose Drugs 0.000 description 1
- 235000019813 microcrystalline cellulose Nutrition 0.000 description 1
- 239000008108 microcrystalline cellulose Substances 0.000 description 1
- 239000002480 mineral oil Substances 0.000 description 1
- 235000010446 mineral oil Nutrition 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- CQDGTJPVBWZJAZ-UHFFFAOYSA-N monoethyl carbonate Chemical compound CCOC(O)=O CQDGTJPVBWZJAZ-UHFFFAOYSA-N 0.000 description 1
- 229910000402 monopotassium phosphate Inorganic materials 0.000 description 1
- 235000019796 monopotassium phosphate Nutrition 0.000 description 1
- 125000004312 morpholin-2-yl group Chemical group [H]N1C([H])([H])C([H])([H])OC([H])(*)C1([H])[H] 0.000 description 1
- 125000004572 morpholin-3-yl group Chemical group N1C(COCC1)* 0.000 description 1
- 125000004573 morpholin-4-yl group Chemical group N1(CCOCC1)* 0.000 description 1
- 210000003205 muscle Anatomy 0.000 description 1
- 229940078490 n,n-dimethylglycine Drugs 0.000 description 1
- MNDYDYTXPOFXLS-UHFFFAOYSA-N n-[[4-(trifluoromethyl)phenyl]methylidene]hydroxylamine Chemical compound ON=CC1=CC=C(C(F)(F)F)C=C1 MNDYDYTXPOFXLS-UHFFFAOYSA-N 0.000 description 1
- 125000005487 naphthalate group Chemical group 0.000 description 1
- 125000001624 naphthyl group Chemical group 0.000 description 1
- 229940097496 nasal spray Drugs 0.000 description 1
- 239000007922 nasal spray Substances 0.000 description 1
- 231100000344 non-irritating Toxicity 0.000 description 1
- 239000012457 nonaqueous media Substances 0.000 description 1
- 102000006255 nuclear receptors Human genes 0.000 description 1
- 108020004017 nuclear receptors Proteins 0.000 description 1
- KKVZAVRSVHUSPL-UHFFFAOYSA-N o-methoxycinnamic aldehyde Natural products COC1=CC=CC=C1C=CC=O KKVZAVRSVHUSPL-UHFFFAOYSA-N 0.000 description 1
- 125000003261 o-tolyl group Chemical group [H]C1=C([H])C(*)=C(C([H])=C1[H])C([H])([H])[H] 0.000 description 1
- OQCDKBAXFALNLD-UHFFFAOYSA-N octadecanoic acid Natural products CCCCCCCC(C)CCCCCCCCC(O)=O OQCDKBAXFALNLD-UHFFFAOYSA-N 0.000 description 1
- 229940049964 oleate Drugs 0.000 description 1
- ZQPPMHVWECSIRJ-KTKRTIGZSA-N oleic acid Chemical compound CCCCCCCC\C=C/CCCCCCCC(O)=O ZQPPMHVWECSIRJ-KTKRTIGZSA-N 0.000 description 1
- PIDFDZJZLOTZTM-KHVQSSSXSA-N ombitasvir Chemical compound COC(=O)N[C@@H](C(C)C)C(=O)N1CCC[C@H]1C(=O)NC1=CC=C([C@H]2N([C@@H](CC2)C=2C=CC(NC(=O)[C@H]3N(CCC3)C(=O)[C@@H](NC(=O)OC)C(C)C)=CC=2)C=2C=CC(=CC=2)C(C)(C)C)C=C1 PIDFDZJZLOTZTM-KHVQSSSXSA-N 0.000 description 1
- 239000003538 oral antidiabetic agent Substances 0.000 description 1
- 238000003305 oral gavage Methods 0.000 description 1
- 150000002895 organic esters Chemical class 0.000 description 1
- 239000003960 organic solvent Substances 0.000 description 1
- 229940039748 oxalate Drugs 0.000 description 1
- 150000002924 oxiranes Chemical class 0.000 description 1
- 125000003854 p-chlorophenyl group Chemical group [H]C1=C([H])C(*)=C([H])C([H])=C1Cl 0.000 description 1
- 125000001037 p-tolyl group Chemical group [H]C1=C([H])C(=C([H])C([H])=C1*)C([H])([H])[H] 0.000 description 1
- NXJCBFBQEVOTOW-UHFFFAOYSA-L palladium(2+);dihydroxide Chemical compound O[Pd]O NXJCBFBQEVOTOW-UHFFFAOYSA-L 0.000 description 1
- VEDDBHYQWFOITD-UHFFFAOYSA-N para-bromobenzyl alcohol Chemical compound OCC1=CC=C(Br)C=C1 VEDDBHYQWFOITD-UHFFFAOYSA-N 0.000 description 1
- 239000012188 paraffin wax Substances 0.000 description 1
- 239000002245 particle Substances 0.000 description 1
- 239000000312 peanut oil Substances 0.000 description 1
- RGSFGYAAUTVSQA-UHFFFAOYSA-N pentamethylene Natural products C1CCCC1 RGSFGYAAUTVSQA-UHFFFAOYSA-N 0.000 description 1
- 125000005010 perfluoroalkyl group Chemical group 0.000 description 1
- 239000002304 perfume Substances 0.000 description 1
- 230000000144 pharmacologic effect Effects 0.000 description 1
- 229960003742 phenol Drugs 0.000 description 1
- 150000002989 phenols Chemical class 0.000 description 1
- WVDDGKGOMKODPV-ZQBYOMGUSA-N phenyl(114C)methanol Chemical compound O[14CH2]C1=CC=CC=C1 WVDDGKGOMKODPV-ZQBYOMGUSA-N 0.000 description 1
- 239000010452 phosphate Substances 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 1
- PJNZPQUBCPKICU-UHFFFAOYSA-N phosphoric acid;potassium Chemical compound [K].OP(O)(O)=O PJNZPQUBCPKICU-UHFFFAOYSA-N 0.000 description 1
- IPNPIHIZVLFAFP-UHFFFAOYSA-N phosphorus tribromide Chemical compound BrP(Br)Br IPNPIHIZVLFAFP-UHFFFAOYSA-N 0.000 description 1
- 230000037081 physical activity Effects 0.000 description 1
- 230000000704 physical effect Effects 0.000 description 1
- 125000004194 piperazin-1-yl group Chemical group [H]N1C([H])([H])C([H])([H])N(*)C([H])([H])C1([H])[H] 0.000 description 1
- 125000004193 piperazinyl group Chemical group 0.000 description 1
- 125000003386 piperidinyl group Chemical group 0.000 description 1
- 229940113116 polyethylene glycol 1000 Drugs 0.000 description 1
- 229940057838 polyethylene glycol 4000 Drugs 0.000 description 1
- 229920005862 polyol Polymers 0.000 description 1
- 150000003077 polyols Chemical class 0.000 description 1
- 239000000256 polyoxyethylene sorbitan monolaurate Substances 0.000 description 1
- 235000010486 polyoxyethylene sorbitan monolaurate Nutrition 0.000 description 1
- 229920000136 polysorbate Polymers 0.000 description 1
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 description 1
- 239000001267 polyvinylpyrrolidone Substances 0.000 description 1
- 229920000036 polyvinylpyrrolidone Polymers 0.000 description 1
- 239000011591 potassium Substances 0.000 description 1
- 229910052700 potassium Inorganic materials 0.000 description 1
- 229910000027 potassium carbonate Inorganic materials 0.000 description 1
- 229960002965 pravastatin Drugs 0.000 description 1
- TUZYXOIXSAXUGO-PZAWKZKUSA-N pravastatin Chemical compound C1=C[C@H](C)[C@H](CC[C@@H](O)C[C@@H](O)CC(O)=O)[C@H]2[C@@H](OC(=O)[C@@H](C)CC)C[C@H](O)C=C21 TUZYXOIXSAXUGO-PZAWKZKUSA-N 0.000 description 1
- 239000003755 preservative agent Substances 0.000 description 1
- 238000002203 pretreatment Methods 0.000 description 1
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 1
- FYPMFJGVHOHGLL-UHFFFAOYSA-N probucol Chemical compound C=1C(C(C)(C)C)=C(O)C(C(C)(C)C)=CC=1SC(C)(C)SC1=CC(C(C)(C)C)=C(O)C(C(C)(C)C)=C1 FYPMFJGVHOHGLL-UHFFFAOYSA-N 0.000 description 1
- 229960003912 probucol Drugs 0.000 description 1
- 230000002035 prolonged effect Effects 0.000 description 1
- 230000001737 promoting effect Effects 0.000 description 1
- LJZPPWWHKPGCHS-UHFFFAOYSA-N propargyl chloride Chemical compound ClCC#C LJZPPWWHKPGCHS-UHFFFAOYSA-N 0.000 description 1
- 239000003380 propellant Substances 0.000 description 1
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000006239 protecting group Chemical group 0.000 description 1
- 230000001681 protective effect Effects 0.000 description 1
- 102000004169 proteins and genes Human genes 0.000 description 1
- 125000004307 pyrazin-2-yl group Chemical group [H]C1=C([H])N=C(*)C([H])=N1 0.000 description 1
- 125000003373 pyrazinyl group Chemical group 0.000 description 1
- USPWKWBDZOARPV-UHFFFAOYSA-N pyrazolidine Chemical group C1CNNC1 USPWKWBDZOARPV-UHFFFAOYSA-N 0.000 description 1
- 125000003226 pyrazolyl group Chemical group 0.000 description 1
- 125000000246 pyrimidin-2-yl group Chemical group [H]C1=NC(*)=NC([H])=C1[H] 0.000 description 1
- 125000004527 pyrimidin-4-yl group Chemical group N1=CN=C(C=C1)* 0.000 description 1
- 125000004528 pyrimidin-5-yl group Chemical group N1=CN=CC(=C1)* 0.000 description 1
- 125000000714 pyrimidinyl group Chemical group 0.000 description 1
- NGXSWUFDCSEIOO-UHFFFAOYSA-N pyrrolidin-3-amine Chemical group NC1CCNC1 NGXSWUFDCSEIOO-UHFFFAOYSA-N 0.000 description 1
- 125000000168 pyrrolyl group Chemical group 0.000 description 1
- 150000003856 quaternary ammonium compounds Chemical class 0.000 description 1
- 125000001453 quaternary ammonium group Chemical group 0.000 description 1
- 238000009790 rate-determining step (RDS) Methods 0.000 description 1
- 210000000664 rectum Anatomy 0.000 description 1
- 230000002829 reductive effect Effects 0.000 description 1
- 238000004007 reversed phase HPLC Methods 0.000 description 1
- 230000000630 rising effect Effects 0.000 description 1
- 238000000926 separation method Methods 0.000 description 1
- 210000002966 serum Anatomy 0.000 description 1
- 239000008159 sesame oil Substances 0.000 description 1
- 235000011803 sesame oil Nutrition 0.000 description 1
- 150000004760 silicates Chemical class 0.000 description 1
- RMAQACBXLXPBSY-UHFFFAOYSA-N silicic acid Chemical compound O[Si](O)(O)O RMAQACBXLXPBSY-UHFFFAOYSA-N 0.000 description 1
- 229960002855 simvastatin Drugs 0.000 description 1
- RYMZZMVNJRMUDD-HGQWONQESA-N simvastatin Chemical compound C([C@H]1[C@@H](C)C=CC2=C[C@H](C)C[C@@H]([C@H]12)OC(=O)C(C)(C)CC)C[C@@H]1C[C@@H](O)CC(=O)O1 RYMZZMVNJRMUDD-HGQWONQESA-N 0.000 description 1
- 210000002027 skeletal muscle Anatomy 0.000 description 1
- 239000002002 slurry Substances 0.000 description 1
- 210000000329 smooth muscle myocyte Anatomy 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- WXMKPNITSTVMEF-UHFFFAOYSA-M sodium benzoate Chemical compound [Na+].[O-]C(=O)C1=CC=CC=C1 WXMKPNITSTVMEF-UHFFFAOYSA-M 0.000 description 1
- 239000004299 sodium benzoate Substances 0.000 description 1
- 235000010234 sodium benzoate Nutrition 0.000 description 1
- 239000012279 sodium borohydride Substances 0.000 description 1
- 229910000033 sodium borohydride Inorganic materials 0.000 description 1
- 239000001509 sodium citrate Substances 0.000 description 1
- NLJMYIDDQXHKNR-UHFFFAOYSA-K sodium citrate Chemical compound O.O.[Na+].[Na+].[Na+].[O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O NLJMYIDDQXHKNR-UHFFFAOYSA-K 0.000 description 1
- BEOOHQFXGBMRKU-UHFFFAOYSA-N sodium cyanoborohydride Chemical compound [Na+].[B-]C#N BEOOHQFXGBMRKU-UHFFFAOYSA-N 0.000 description 1
- 235000019333 sodium laurylsulphate Nutrition 0.000 description 1
- 239000008247 solid mixture Substances 0.000 description 1
- 239000004334 sorbic acid Substances 0.000 description 1
- 235000010199 sorbic acid Nutrition 0.000 description 1
- 229940075582 sorbic acid Drugs 0.000 description 1
- 239000000600 sorbitol Substances 0.000 description 1
- 125000003003 spiro group Chemical group 0.000 description 1
- 239000007921 spray Substances 0.000 description 1
- 238000012289 standard assay Methods 0.000 description 1
- 239000008117 stearic acid Substances 0.000 description 1
- 150000003431 steroids Chemical class 0.000 description 1
- 229910052682 stishovite Inorganic materials 0.000 description 1
- 125000005346 substituted cycloalkyl group Chemical group 0.000 description 1
- KDYFGRWQOYBRFD-UHFFFAOYSA-L succinate(2-) Chemical compound [O-]C(=O)CCC([O-])=O KDYFGRWQOYBRFD-UHFFFAOYSA-L 0.000 description 1
- 150000008163 sugars Chemical class 0.000 description 1
- 150000003457 sulfones Chemical group 0.000 description 1
- 150000003462 sulfoxides Chemical class 0.000 description 1
- 125000004962 sulfoxyl group Chemical group 0.000 description 1
- 230000001629 suppression Effects 0.000 description 1
- 239000004094 surface-active agent Substances 0.000 description 1
- 239000003765 sweetening agent Substances 0.000 description 1
- 239000006188 syrup Substances 0.000 description 1
- 235000020357 syrup Nutrition 0.000 description 1
- 239000000454 talc Substances 0.000 description 1
- 229910052623 talc Inorganic materials 0.000 description 1
- 235000012222 talc Nutrition 0.000 description 1
- 229940095064 tartrate Drugs 0.000 description 1
- CBXCPBUEXACCNR-UHFFFAOYSA-N tetraethylammonium Chemical compound CC[N+](CC)(CC)CC CBXCPBUEXACCNR-UHFFFAOYSA-N 0.000 description 1
- BSYVTEYKTMYBMK-UHFFFAOYSA-N tetrahydrofurfuryl alcohol Chemical compound OCC1CCCO1 BSYVTEYKTMYBMK-UHFFFAOYSA-N 0.000 description 1
- QEMXHQIAXOOASZ-UHFFFAOYSA-N tetramethylammonium Chemical compound C[N+](C)(C)C QEMXHQIAXOOASZ-UHFFFAOYSA-N 0.000 description 1
- 125000003831 tetrazolyl group Chemical group 0.000 description 1
- 238000002560 therapeutic procedure Methods 0.000 description 1
- 125000001544 thienyl group Chemical group 0.000 description 1
- 125000002053 thietanyl group Chemical group 0.000 description 1
- 125000004568 thiomorpholinyl group Chemical group 0.000 description 1
- 210000001685 thyroid gland Anatomy 0.000 description 1
- 102000004217 thyroid hormone receptors Human genes 0.000 description 1
- 108090000721 thyroid hormone receptors Proteins 0.000 description 1
- JOXIMZWYDAKGHI-UHFFFAOYSA-N toluene-4-sulfonic acid Chemical compound CC1=CC=C(S(O)(=O)=O)C=C1 JOXIMZWYDAKGHI-UHFFFAOYSA-N 0.000 description 1
- 238000011200 topical administration Methods 0.000 description 1
- 235000010487 tragacanth Nutrition 0.000 description 1
- 239000000196 tragacanth Substances 0.000 description 1
- 229940116362 tragacanth Drugs 0.000 description 1
- 238000001890 transfection Methods 0.000 description 1
- 230000009261 transgenic effect Effects 0.000 description 1
- 238000003146 transient transfection Methods 0.000 description 1
- 125000001425 triazolyl group Chemical group 0.000 description 1
- 229910052905 tridymite Inorganic materials 0.000 description 1
- 229940070710 valerate Drugs 0.000 description 1
- NQPDZGIKBAWPEJ-UHFFFAOYSA-N valeric acid Chemical compound CCCCC(O)=O NQPDZGIKBAWPEJ-UHFFFAOYSA-N 0.000 description 1
- 210000003556 vascular endothelial cell Anatomy 0.000 description 1
- 235000015112 vegetable and seed oil Nutrition 0.000 description 1
- 239000008158 vegetable oil Substances 0.000 description 1
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 description 1
- 229920002554 vinyl polymer Polymers 0.000 description 1
- 230000003612 virological effect Effects 0.000 description 1
- 102000009310 vitamin D receptors Human genes 0.000 description 1
- 108050000156 vitamin D receptors Proteins 0.000 description 1
- 238000005406 washing Methods 0.000 description 1
- 230000003442 weekly effect Effects 0.000 description 1
- 238000009736 wetting Methods 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C323/00—Thiols, sulfides, hydropolysulfides or polysulfides substituted by halogen, oxygen or nitrogen atoms, or by sulfur atoms not being part of thio groups
- C07C323/10—Thiols, sulfides, hydropolysulfides or polysulfides substituted by halogen, oxygen or nitrogen atoms, or by sulfur atoms not being part of thio groups containing thio groups and singly-bound oxygen atoms bound to the same carbon skeleton
- C07C323/18—Thiols, sulfides, hydropolysulfides or polysulfides substituted by halogen, oxygen or nitrogen atoms, or by sulfur atoms not being part of thio groups containing thio groups and singly-bound oxygen atoms bound to the same carbon skeleton having the sulfur atom of at least one of the thio groups bound to a carbon atom of a six-membered aromatic ring of the carbon skeleton
- C07C323/20—Thiols, sulfides, hydropolysulfides or polysulfides substituted by halogen, oxygen or nitrogen atoms, or by sulfur atoms not being part of thio groups containing thio groups and singly-bound oxygen atoms bound to the same carbon skeleton having the sulfur atom of at least one of the thio groups bound to a carbon atom of a six-membered aromatic ring of the carbon skeleton with singly-bound oxygen atoms bound to carbon atoms of the same non-condensed six-membered aromatic ring
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/04—Anorexiants; Antiobesity agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/06—Antihyperlipidemics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/08—Drugs for disorders of the metabolism for glucose homeostasis
- A61P3/10—Drugs for disorders of the metabolism for glucose homeostasis for hyperglycaemia, e.g. antidiabetics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P5/00—Drugs for disorders of the endocrine system
- A61P5/48—Drugs for disorders of the endocrine system of the pancreatic hormones
- A61P5/50—Drugs for disorders of the endocrine system of the pancreatic hormones for increasing or potentiating the activity of insulin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/10—Drugs for disorders of the cardiovascular system for treating ischaemic or atherosclerotic diseases, e.g. antianginal drugs, coronary vasodilators, drugs for myocardial infarction, retinopathy, cerebrovascula insufficiency, renal arteriosclerosis
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C317/00—Sulfones; Sulfoxides
- C07C317/16—Sulfones; Sulfoxides having sulfone or sulfoxide groups and singly-bound oxygen atoms bound to the same carbon skeleton
- C07C317/22—Sulfones; Sulfoxides having sulfone or sulfoxide groups and singly-bound oxygen atoms bound to the same carbon skeleton with sulfone or sulfoxide groups bound to carbon atoms of six-membered aromatic rings of the carbon skeleton
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C323/00—Thiols, sulfides, hydropolysulfides or polysulfides substituted by halogen, oxygen or nitrogen atoms, or by sulfur atoms not being part of thio groups
- C07C323/10—Thiols, sulfides, hydropolysulfides or polysulfides substituted by halogen, oxygen or nitrogen atoms, or by sulfur atoms not being part of thio groups containing thio groups and singly-bound oxygen atoms bound to the same carbon skeleton
- C07C323/18—Thiols, sulfides, hydropolysulfides or polysulfides substituted by halogen, oxygen or nitrogen atoms, or by sulfur atoms not being part of thio groups containing thio groups and singly-bound oxygen atoms bound to the same carbon skeleton having the sulfur atom of at least one of the thio groups bound to a carbon atom of a six-membered aromatic ring of the carbon skeleton
- C07C323/19—Thiols, sulfides, hydropolysulfides or polysulfides substituted by halogen, oxygen or nitrogen atoms, or by sulfur atoms not being part of thio groups containing thio groups and singly-bound oxygen atoms bound to the same carbon skeleton having the sulfur atom of at least one of the thio groups bound to a carbon atom of a six-membered aromatic ring of the carbon skeleton with singly-bound oxygen atoms bound to acyclic carbon atoms of the carbon skeleton
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C59/00—Compounds having carboxyl groups bound to acyclic carbon atoms and containing any of the groups OH, O—metal, —CHO, keto, ether, groups, groups, or groups
- C07C59/40—Unsaturated compounds
- C07C59/58—Unsaturated compounds containing ether groups, groups, groups, or groups
- C07C59/64—Unsaturated compounds containing ether groups, groups, groups, or groups containing six-membered aromatic rings
- C07C59/66—Unsaturated compounds containing ether groups, groups, groups, or groups containing six-membered aromatic rings the non-carboxylic part of the ether containing six-membered aromatic rings
- C07C59/68—Unsaturated compounds containing ether groups, groups, groups, or groups containing six-membered aromatic rings the non-carboxylic part of the ether containing six-membered aromatic rings the oxygen atom of the ether group being bound to a non-condensed six-membered aromatic ring
- C07C59/70—Ethers of hydroxy-acetic acid, e.g. substitutes on the ring
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C59/00—Compounds having carboxyl groups bound to acyclic carbon atoms and containing any of the groups OH, O—metal, —CHO, keto, ether, groups, groups, or groups
- C07C59/40—Unsaturated compounds
- C07C59/58—Unsaturated compounds containing ether groups, groups, groups, or groups
- C07C59/72—Unsaturated compounds containing ether groups, groups, groups, or groups containing six-membered aromatic rings and other rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D209/00—Heterocyclic compounds containing five-membered rings, condensed with other rings, with one nitrogen atom as the only ring hetero atom
- C07D209/02—Heterocyclic compounds containing five-membered rings, condensed with other rings, with one nitrogen atom as the only ring hetero atom condensed with one carbocyclic ring
- C07D209/04—Indoles; Hydrogenated indoles
- C07D209/10—Indoles; Hydrogenated indoles with substituted hydrocarbon radicals attached to carbon atoms of the hetero ring
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D211/00—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings
- C07D211/04—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings with only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D211/06—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having no double bonds between ring members or between ring members and non-ring members
- C07D211/08—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having no double bonds between ring members or between ring members and non-ring members with hydrocarbon or substituted hydrocarbon radicals directly attached to ring carbon atoms
- C07D211/10—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having no double bonds between ring members or between ring members and non-ring members with hydrocarbon or substituted hydrocarbon radicals directly attached to ring carbon atoms with radicals containing only carbon and hydrogen atoms attached to ring carbon atoms
- C07D211/14—Heterocyclic compounds containing hydrogenated pyridine rings, not condensed with other rings with only hydrogen or carbon atoms directly attached to the ring nitrogen atom having no double bonds between ring members or between ring members and non-ring members with hydrocarbon or substituted hydrocarbon radicals directly attached to ring carbon atoms with radicals containing only carbon and hydrogen atoms attached to ring carbon atoms with hydrocarbon or substituted hydrocarbon radicals attached to the ring nitrogen atom
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D213/00—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members
- C07D213/02—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members
- C07D213/04—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D213/24—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom with substituted hydrocarbon radicals attached to ring carbon atoms
- C07D213/28—Radicals substituted by singly-bound oxygen or sulphur atoms
- C07D213/30—Oxygen atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D213/00—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members
- C07D213/02—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members
- C07D213/04—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D213/24—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom with substituted hydrocarbon radicals attached to ring carbon atoms
- C07D213/28—Radicals substituted by singly-bound oxygen or sulphur atoms
- C07D213/32—Sulfur atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D231/00—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings
- C07D231/02—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings not condensed with other rings
- C07D231/10—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members
- C07D231/12—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to ring carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D233/00—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, not condensed with other rings
- C07D233/54—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, not condensed with other rings having two double bonds between ring members or between ring members and non-ring members
- C07D233/56—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, not condensed with other rings having two double bonds between ring members or between ring members and non-ring members with only hydrogen atoms or radicals containing only hydrogen and carbon atoms, attached to ring carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D233/00—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, not condensed with other rings
- C07D233/54—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, not condensed with other rings having two double bonds between ring members or between ring members and non-ring members
- C07D233/66—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, not condensed with other rings having two double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D233/68—Halogen atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D235/00—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, condensed with other rings
- C07D235/02—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, condensed with other rings condensed with carbocyclic rings or ring systems
- C07D235/04—Benzimidazoles; Hydrogenated benzimidazoles
- C07D235/18—Benzimidazoles; Hydrogenated benzimidazoles with aryl radicals directly attached in position 2
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D249/00—Heterocyclic compounds containing five-membered rings having three nitrogen atoms as the only ring hetero atoms
- C07D249/02—Heterocyclic compounds containing five-membered rings having three nitrogen atoms as the only ring hetero atoms not condensed with other rings
- C07D249/08—1,2,4-Triazoles; Hydrogenated 1,2,4-triazoles
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D261/00—Heterocyclic compounds containing 1,2-oxazole or hydrogenated 1,2-oxazole rings
- C07D261/02—Heterocyclic compounds containing 1,2-oxazole or hydrogenated 1,2-oxazole rings not condensed with other rings
- C07D261/06—Heterocyclic compounds containing 1,2-oxazole or hydrogenated 1,2-oxazole rings not condensed with other rings having two or more double bonds between ring members or between ring members and non-ring members
- C07D261/08—Heterocyclic compounds containing 1,2-oxazole or hydrogenated 1,2-oxazole rings not condensed with other rings having two or more double bonds between ring members or between ring members and non-ring members with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to ring carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D271/00—Heterocyclic compounds containing five-membered rings having two nitrogen atoms and one oxygen atom as the only ring hetero atoms
- C07D271/02—Heterocyclic compounds containing five-membered rings having two nitrogen atoms and one oxygen atom as the only ring hetero atoms not condensed with other rings
- C07D271/06—1,2,4-Oxadiazoles; Hydrogenated 1,2,4-oxadiazoles
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D271/00—Heterocyclic compounds containing five-membered rings having two nitrogen atoms and one oxygen atom as the only ring hetero atoms
- C07D271/02—Heterocyclic compounds containing five-membered rings having two nitrogen atoms and one oxygen atom as the only ring hetero atoms not condensed with other rings
- C07D271/10—1,3,4-Oxadiazoles; Hydrogenated 1,3,4-oxadiazoles
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D295/00—Heterocyclic compounds containing polymethylene-imine rings with at least five ring members, 3-azabicyclo [3.2.2] nonane, piperazine, morpholine or thiomorpholine rings, having only hydrogen atoms directly attached to the ring carbon atoms
- C07D295/04—Heterocyclic compounds containing polymethylene-imine rings with at least five ring members, 3-azabicyclo [3.2.2] nonane, piperazine, morpholine or thiomorpholine rings, having only hydrogen atoms directly attached to the ring carbon atoms with substituted hydrocarbon radicals attached to ring nitrogen atoms
- C07D295/08—Heterocyclic compounds containing polymethylene-imine rings with at least five ring members, 3-azabicyclo [3.2.2] nonane, piperazine, morpholine or thiomorpholine rings, having only hydrogen atoms directly attached to the ring carbon atoms with substituted hydrocarbon radicals attached to ring nitrogen atoms substituted by singly bound oxygen or sulfur atoms
- C07D295/084—Heterocyclic compounds containing polymethylene-imine rings with at least five ring members, 3-azabicyclo [3.2.2] nonane, piperazine, morpholine or thiomorpholine rings, having only hydrogen atoms directly attached to the ring carbon atoms with substituted hydrocarbon radicals attached to ring nitrogen atoms substituted by singly bound oxygen or sulfur atoms with the ring nitrogen atoms and the oxygen or sulfur atoms attached to the same carbon chain, which is not interrupted by carbocyclic rings
- C07D295/088—Heterocyclic compounds containing polymethylene-imine rings with at least five ring members, 3-azabicyclo [3.2.2] nonane, piperazine, morpholine or thiomorpholine rings, having only hydrogen atoms directly attached to the ring carbon atoms with substituted hydrocarbon radicals attached to ring nitrogen atoms substituted by singly bound oxygen or sulfur atoms with the ring nitrogen atoms and the oxygen or sulfur atoms attached to the same carbon chain, which is not interrupted by carbocyclic rings to an acyclic saturated chain
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D307/00—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom
- C07D307/77—Heterocyclic compounds containing five-membered rings having one oxygen atom as the only ring hetero atom ortho- or peri-condensed with carbocyclic rings or ring systems
- C07D307/78—Benzo [b] furans; Hydrogenated benzo [b] furans
- C07D307/79—Benzo [b] furans; Hydrogenated benzo [b] furans with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to carbon atoms of the hetero ring
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D311/00—Heterocyclic compounds containing six-membered rings having one oxygen atom as the only hetero atom, condensed with other rings
- C07D311/02—Heterocyclic compounds containing six-membered rings having one oxygen atom as the only hetero atom, condensed with other rings ortho- or peri-condensed with carbocyclic rings or ring systems
- C07D311/04—Benzo[b]pyrans, not hydrogenated in the carbocyclic ring
- C07D311/58—Benzo[b]pyrans, not hydrogenated in the carbocyclic ring other than with oxygen or sulphur atoms in position 2 or 4
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D333/00—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom
- C07D333/50—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom condensed with carbocyclic rings or ring systems
- C07D333/52—Benzo[b]thiophenes; Hydrogenated benzo[b]thiophenes
- C07D333/54—Benzo[b]thiophenes; Hydrogenated benzo[b]thiophenes with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to carbon atoms of the hetero ring
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D405/00—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom
- C07D405/02—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing two hetero rings
- C07D405/12—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D413/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D413/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings
- C07D413/04—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings directly linked by a ring-member-to-ring-member bond
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D413/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D413/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings
- C07D413/12—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2602/00—Systems containing two condensed rings
- C07C2602/02—Systems containing two condensed rings the rings having only two atoms in common
- C07C2602/04—One of the condensed rings being a six-membered aromatic ring
- C07C2602/08—One of the condensed rings being a six-membered aromatic ring the other ring being five-membered, e.g. indane
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2603/00—Systems containing at least three condensed rings
- C07C2603/02—Ortho- or ortho- and peri-condensed systems
- C07C2603/04—Ortho- or ortho- and peri-condensed systems containing three rings
- C07C2603/06—Ortho- or ortho- and peri-condensed systems containing three rings containing at least one ring with less than six ring members
- C07C2603/10—Ortho- or ortho- and peri-condensed systems containing three rings containing at least one ring with less than six ring members containing five-membered rings
- C07C2603/12—Ortho- or ortho- and peri-condensed systems containing three rings containing at least one ring with less than six ring members containing five-membered rings only one five-membered ring
- C07C2603/18—Fluorenes; Hydrogenated fluorenes
Definitions
- the present invention relates to compounds and pharmaceutical formulations that can be used to treat conditions mediated by nuclear hormone receptors, more specifically, to compounds and pharmaceutical formulations that modulate Peroxisome Proliferator Activation Receptor (PPAR) activity.
- PPAR Peroxisome Proliferator Activation Receptor
- Hypercholesterolemia, dyslipidemia, diabetes, and obesity are well-recognized risk factors in the onset of atherosclerosis and coronary heart disease.
- the diseases are characterized by high levels of cholesterol and lipids in the blood.
- the blood cholesterol pool is generally dependent on dietary uptake of cholesterol from the intestine, and from the biosynthesis of cholesterol throughout the body, especially the liver.
- the majority of cholesterol in plasma is carried on apolipoprotein B-containing lipoproteins, such as low-density lipoproteins (LDL) and very-low-density lipoproteins (VLDL).
- LDL low-density lipoproteins
- VLDL very-low-density lipoproteins
- the risk of coronary artery disease in man increases when LDL and VLDL levels increase.
- high levels of cholesterol carried in high-density lipoproteins (HDL) is protective against coronary artery disease (Am. J. Med., 1977;62:707-714).
- statins represent perhaps the most important class of lipid-lowering drugs. These compounds inhibit HMG-CoA reductase which is implicated in the rate-limiting step in cellular cholesterol biosynthesis.
- Representative statins include atorvastatin, lovastatin, pravastatin, and simvastatin. The effectiveness of these compounds depends on LDL receptor regulation.
- Other important antilipidemia drugs include fibrates such as gemfibril and clofibrate, bile acid sequestrants such as cholestyramine and colestipol, probucol, and nicotinic acid analogs.
- sulfonylureas are generally used to stimulate insulin.
- the biguanide metformin is generally used to improve insulin sensitivity and to decrease hepatic glucose output.
- Acarbose is used to limit postprandial hyperglycemia.
- Thiazolidine 2,4 diones are used to enhance insulin action without increasing insulin secretion.
- Obesity is a chronic disease that is highly prevalent in modern society and is associated not only with a social stigma, but also with decreased life span and numerous medical problems, including diabetes mellitus, insulin resistance, hypertension, hypercholesterolemia, thromboembolic disease, and coronary heart disease.
- Treatment of obesity remains a problem and it is unclear whether dieting results in decreased long-term risk of early death.
- a further important obesity intervention is physical activity. Exercise, however, in general, has been found to be only moderately successful in promoting weight loss. A program combining both dieting and exercise as well as behaviour modification is widely viewed as the optimal approach to weight loss. Studies have demonstrated that the combination of both food restriction and exercise promote a substantial loss of fat while maintaining lean tissue.
- PPAR Peroxisome Proliferator Activation Receptors
- PPARs are implicated in a number of biological processes and disease states including hypercholesterolemia, dyslipidemia, and diabetes.
- PPARs are members of the nuclear receptor superfamily of transcription factors that includes steroid, thyroid, and vitamin D receptors. They play a role in controlling expression of proteins that regulate lipid metabolism.
- the PPARs are activated by fatty acids and fatty acid metabolites.
- PPAR ⁇ for instance, is expressed most abundantly in adipose tissue and at lower levels in skeletal muscle, heart, liver, intestine, kidney, vascular endothelial and smooth muscle cells as well as macrophages.
- PPAR receptors are associated with regulation of insulin sensitivity and blood glucose levels, macrophage differentiation, inflammatory response, and cell differentiation. Accordingly, PPARs have been associated with obesity, diabetes, carcinogenesis, hyperplasia, atherosclerosis, dyslipidemia, and hypercholesterolemia.
- PPAR ⁇ agonists lower plasma triglycerides and LDL cholesterol and are therefore useful in treating hypertriglyceridemia, dyslipidemia and obesity.
- PPAR ⁇ is associated with the development of non-insulin-dependent diabetes mellitus (NIDDM), hypertension, coronary artery disease, dyslipidemia and certain malignancies.
- NIDDM non-insulin-dependent diabetes mellitus
- activation of PPAR ⁇ has been demonstrated to increase HDL levels.
- a PPAR ⁇ selective agonist was reported to have shown a dose-related increase in serum HDL-C and decrease in LDL-C and VLDL-TG in insulin-resistant middle aged rhesus monkeys. (W. R. Oliver et al., PNAS, v. 98, pp. 5306-5311, 2001)
- Antilipidemic, antidiabetic and anti-obesity agents are still considered to have non-uniform effectiveness.
- the effectiveness of antidiabetic and antilipidemic therapies is limited, in part because of poor patient compliance due to unacceptable side effects. These side effects include diarrhea and gastrointestinal discomfort, and in the case of antidiabetics, edema, hypoglycemia and hepatoxicity. Furthermore, each type of drug does not work equally well in all patients.
- the present invention provides compounds capable of modulating PPAR activity.
- Compounds of the present invention are described by Formula I: and pharmaceutically acceptable salts thereof, where:
- X 0 and X 1 are independently absent, O, S, —CH 2 —, —CH 2 —CH 2 —, —CH ⁇ CH—, —CH ⁇ CH—, —S(O) 2 —, or —S(O)—;
- Ar 1 and Ar 2 are each independently unsubstituted or substituted aryl or heteroaryl, provided that Ar 1 is not thiazolyl or oxazolyl; is absent; or when present, is a saturated or unsaturated hydrocarbon chain which is substituted or unsubstituted, wherein said chain has from 1 to 4 atoms so that Ar 1 , X 1 , (CH 2 ) r , and Ar 2 , together form a five to eight membered ring;
- R 1 and R 2 are selected from hydrogen, lower alkyl, lower alkoxy, haloalkyl, —O—(CH 2 ) m CF 3 , halogen, nitro, cyano, —OH, —SH, —CF 3 , —S(O) p alkyl, S(O) p aryl, —(CH 2 ) m OR 5 , —(CH 2 ) m NR 6 R 7 , —COR 5 , CO 2 R 5 , or —NR 6 R 7 , or together with the atoms to which they are attached form a five to eight member ring;
- R 3 and R 4 are selected from hydrogen, lower alkyl, lower alkoxy, haloalkyl, —O—(CH 2 ) m CF 3 , halogen, nitro, cyano, —OH, —SH, —CF 3 , —S(O) p alkyl, S(O) p aryl, —(CH 2 ) m OR 5 , —(CH 2 ) m NR 6 R 7 , —COR 5 , —CO 2 H, —CO 2 R 5 , or —NR 6 R 7 ;
- R 1 -R 4 is H, lower alkyl, lower alkoxy, haloalkyl, —O—(CH 2 ) m CF 3 , halogen, nitro, cyano, —OH, —SH, —CF 3 , —S(O) p alkyl, S(O) p aryl, —(CH 2 ) m OR 5 , —(CH 2 ) m NR 6 R 7 , or NR 6 R 7 ;
- R 5 is hydrogen, alkyl, alkenyl, alkynyl, or aryl
- R 6 and R 7 are each independently hydrogen, alkyl, alkenyl, alkynyl, —COalkyl, —COaryl, cycloalkyl, —CO 2 alkyl, —CO 2 aryl, or R 6 and R 7 together with the atoms to which they are attached form a 4 to 7 membered ring having 1 to 3 heteroatoms;
- n 0 to 5;
- p 0, 1, or 2;
- r 0 to 6.
- the invention also provides a compound of formula (II): and pharmaceutically acceptable salts thereof, where:
- X 0 and X 1 are independently absent, O, S, —CH 2 —, —CH 2 —CH 2 —, —CH ⁇ CH—, —CH ⁇ CH—, —S(O) 2 —, or —S(O)—;
- Ar 1 and Ar 2 are each independently unsubstituted or substituted aryl or heteroaryl, provided that Ar 1 is not thiazolyl or oxazolyl; is absent; or when present, is a saturated or unsaturated, hydrocarbon chain which is substituted or unsubstituted, wherein said chain has from 1 to 4 atoms so that Ar 1 , X 1 , (CH 2 ) r , and Ar 2 , together form a five to eight member ring;
- R 3 and R 4 are selected from hydrogen, lower alkyl, lower alkoxy, haloalkyl, —O—(CH 2 ) m CF 3 , halogen, nitro, cyano, —OH, —SH, —CF 3 , —S(O) p alkyl, S(O) p aryl, —(CH 2 ) m OR 5 , —(CH 2 ) m NR 6 R 7 , —COR 5 , —CO 2 H, —CO 2 R 5 , or —NR 6 R 7 ;
- R 5 is hydrogen, alkyl, alkenyl, alkynyl, or aryl
- R 6 and R 7 are each independently hydrogen, alkyl, alkenyl, alkynyl, —COalkyl, —COaryl, cycloalkyl, —CO 2 alkyl, —CO 2 aryl, —SO 2 alkyl, —SO 2 aryl, or R 6 and R 7 together with the atoms to which they are attached form a 4 to 7 membered ring having 1 to 3 heteroatoms; is a saturated or unsaturated, substituted or unsubstituted hydrocarbon chain or hydrocarbon-heteroatom chain having from 3 to 6 atoms wherein the carbon atom of position 2 is connected to the carbon atom of position 3 to form a five to eight member ring;
- n 0 to 5;
- p 0 to 2;
- r 0 to 6.
- a method of treating, preventing or controlling hypercholesteremia and dyslipidemia in a mammal comprises administering to the mammal in need thereof a therapeutically effective amount of the compounds of the present invention.
- the compounds of the present invention are also useful in the method of the present invention for treating, preventing, or controlling obesity, eating disorders, hyperglycemia, atherosclerosis, hypertriglyceridemia, hyperinsulinemia and diabetes.
- the compounds of the present invention are also useful in the methods of suppressing appetite in a mammal, modulating leptin levels in a mammal, and treating a patient exhibiting glucose disorders associated with circulating glucocorticoids, growth hormone, catecholamines, glucagon, or parathyroid hormone.
- a therapeutically effective amount of the compounds of the present invention are administered to the mammal in need thereof.
- a method for preparing compounds with Formulae I-II, or a pharmaceutically acceptable salt thereof comprises reacting in a solvent in the presence of a base such as cesium carbonate: with where
- X 0 is OH or SH
- n, q, r, R 1 , R 2 , R 3 , R 4 , X 1 , Ar 1 and Ar 2 are as defined above for Formula I;
- R 11 is a lower alkyl
- X is a halogen
- an alternative method for preparing compounds with Formulae I-II, or a pharmaceutically acceptable salt thereof is provided.
- the method of this embodiment comprises reacting
- R 1 -R 4 have any of the meanings defined above, and R 11 is a lower alkyl with: where - - - is a bond or is absent and wherein n, q, r, X 0 , X 1 , Ar 1 and Ar 2 are as defined above for Formula I;
- the double bond may optionally be removed, for instance, by hydrogenation and the resulting ester is preferably hydrolyzed to form the compounds of Formulas I or II.
- the invention provides a process for preparing the compound of formula I-4 which is: or a pharmaceutically acceptable salt thereof, comprising:
- R 1 is hydrogen or together with R 2 forms a 5 membered carbocyclic ring
- R 2 is methoxy or together with R 1 forms a 5 membered carbocyclic ring
- R 3 is hydrogen or methyl
- R 4 is hydrogen
- X 1 is absent or O
- r is 0 or 1.
- halo is fluoro, chloro, bromo, or iodo.
- Alkyl, alkoxy, alkenyl, alkynyl, etc. denote both straight and branched groups; but reference to an individual radical such as “propyl” embraces only the straight chain radical, a branched chain isomer such as “isopropyl” being specifically referred to.
- alkyl refers to a straight or branched, hydrocarbon of from 1 to 11 carbon atoms and includes, for example, methyl, ethyl, n-propyl, isopropyl, n-butyl, sec-butyl, isobutyl, tert-butyl, n-pentyl, n-hexyl, and the like.
- the alkyl group can also be substituted with one or more of the substituents selected from lower alkoxy, lower thioalkoxy, —O(CH 2 ) 1-5 CF 3 , halogen, nitro, cyano, ⁇ O, ⁇ S, —OH, —SH, —CF 3 , —OCF 3 , —CO 2 H, CO 2 C 1 -C 6 alkyl, —NH 2 , —NHC 1 -C 6 alkyl, —CONR′R′′, or —N(C 1 -C 6 alkyl) 2 where R′ and R′′ are independently alkyl, akenyl, alkynyl, aryl, or joined together to form a 4 to 7 membering ring.
- Preferred alkyl groups have from 1 to 6 carbon atoms (C 1 -C 6 alkyl).
- lower alkyl refers to a subset of alkyl which means a straight or branched hydrocarbon radical having from 1 to 6 carbon atoms and includes, for example, methyl, ethyl, n-propyl, isopropyl, n-butyl, sec-butyl, isobutyl, tert-butyl, n-pentyl, n-hexyl, and the like.
- lower alkyl is referred to as “C 1 -C 6 alkyl.”
- haloalkyl refers to a lower alkyl radical, as defined above, bearing at least one halogen substituent, for example, chloromethyl, fluoroethyl, trifluoromethyl, or 1,1,1-trifluoroethyl and the like.
- Haloalkyl can also include perfluoroalkyl wherein all hydrogens of a loweralkyl group are replaced with fluorine atoms.
- alkenyl means a straight or branched unsaturated hydrocarbon radical having from 2 to 12 carbon atoms and includes, for example, ethenyl, 1-propenyl, 2-propenyl, 1-butenyl, 2-butenyl, 1-pentenyl, 2-pentenyl, 3-methyl-3-butenyl, 1-hexenyl, 2-hexenyl, 3-hexenyl, 3-heptenyl, 1-octenyl, 1-nonenyl, 1-decenyl, 1-undecenyl, 1-dodecenyl, and the like.
- alkynyl means a straight or branched hydrocarbon radical having from 2 to 12 carbon atoms having at least one triple bond and includes, for example, 1-propynyl, 1-butynyl, 3-butynyl, 1-pentynyl, 3-pentynyl, 3-methyl-3-butynyl, 1-hexynyl, 3-hexynyl, 3-heptynyl, 1-octynyl, 1-nonynyl, 1-decynyl, 1-undecynyl, 1-dodecynyl, and the like.
- alkylene refers to a divalent group derived from a straight or branched chain saturated hydrocarbon having from 1 to 10 carbon atoms by the removal of two hydrogen atoms, for example, methylene, 1,2-ethylene, 1,1-ethylene, 1,3-propylene, 2,2-dimethylpropylene, and the like.
- the alkylene groups of this invention can be optionally substituted.
- the alkylene group can also be substituted with one or more of the substituents selected from lower alkyl, lower alkoxy, lower thioalkoxy, —O(CH 2 ) 1-5 CF 3 , halogen, nitro, cyano, ⁇ O, ⁇ S, —OH, —SH, —CF 3 , —CO 2 H, —CO 2 C 1 -C 6 alkyl, —NH 2 , —NHC 1 -C 6 alkyl, —CONR′R′′, or —N(C 1 -C 6 alkyl) 2 where R′ and R′′ are independently alkyl, akenyl, alkynyl, aryl, or joined together to form a 4 to 7 member ring.
- Preferred alkylene groups have from 1 to 6 carbon atoms (C 1 -C 6 alkyl).
- cycloalkyl means a hydrocarbon ring containing from 3 to 12 carbon atoms, for example, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, cycloctyl, decalinyl, norpinanyl, and adamantyl.
- the cycloalkyl group may contain double bonds, for example, 3-cyclohexen-1-yl.
- the cycloalkyl ring may be unsubstituted or substituted by 1 to 3 substituents selected from alkyl, alkoxy, thioalkoxy, hydroxy, thiol, nitro, halogen, amino, alkyl and dialkylamino, formyl, carboxyl, CN, —NH—CO—R′, —CO—NHR′—, —CO 2 R′, —COR′, aryl, or heteroaryl, wherein alkyl, aryl, and heteroaryl are as defined herein.
- substituted cycloalkyl groups include fluorocyclopropyl, 2-iodocyclobutyl, 2,3-dimethylcyclopentyl, 2,2-dimethoxycyclohexyl, and 3-phenylcyclopentyl.
- heteroatom as used herein represents oxygen, nitrogen, or sulfur (O, N, or S) as well as sulfoxyl or sulfonyl (SO or SO 2 ) unless otherwise indicated.
- heterocycloalkyl means a monocyclic, fused, bridged, or spiro bicyclic heterocyclic ring systems.
- Monocyclic heterocyclic rings contain from about 3 to 12 ring atoms, with from 1 to 5 heteroatoms selected from N, O, and S, and preferably from 3 to 7 member atoms, in the ring.
- Bicyclic heterocyclics contain from 7 to 17 member atoms, preferably 7 to 12 member atoms, in the ring.
- Bicyclic heterocyclics contain from about 7 to about 17 ring atoms, preferably from 7 to 12 ring atoms.
- Bicyclic heterocyclics rings may be fused, spiro, or bridged ring systems.
- heterocyclic groups include cyclic ethers (oxiranes) such as ethyleneoxide, tetrahydrofuran, dioxane, and substituted cyclic ethers, wherein the substituents are those described above for the alkyl and cycloalkyl groups.
- Typical substituted cyclic ethers include propyleneoxide, phenyloxirane (styrene oxide), cis-2-butene-oxide (2,3-dimethyloxirane), 3-Chlorotetrahydrofuran, 2,6-dimethyl-1,4-dioxane, and the like.
- Heterocycles containing nitrogen are groups such as pyrrolidine, piperidine, piperazine, tetrahydrotriazine, tetrahydropyrazole, and substituted groups such as 3-aminopyrrolidine, 4-methylpiperazin-1-yl, and the like.
- Typical sulfur containing heterocycles include tetrahydrothiophene, dihydro-1,3-dithiol-2-yl, and hexahydrothiepin-4-yl.
- heterocycles include dihydro-oxathiol-4-yl, tetrahydro-oxazolyl, tetrahydro-oxadiazolyl, tetrahydrodioxazolyl, tetrahydrooxathiazolyl, hexahydrotriazinyl, tetrahydro-oxazinyl, morpholinyl, thiomorpholinyl, tetrahydropyrimidinyl, dioxolinyl, octahydrobenzofuranyl, octahydrobenzimidazolyl, and octahydrobenzothiazolyl.
- heterocycles containing sulfur the oxidized sulfur heterocycles containing SO or SO 2 groups are also included. Examples include the sulfoxide and sulfone forms of tetrahydrothiophene.
- hydrocarbon chain refers to a straight hydrocarbon of from 2 to 6 carbon atoms.
- the hydrocarbon chain is optionally substituted with one or more substituents selected from lower alkyl, lower alkoxy, lower thioalkoxy, —O(CH 2 ) 0-2 CF 3 , halogen, nitro, cyano, ⁇ O, ⁇ S, —OH, —SH, —CF 3 , —CO 2 H, —CO 2 (C 1 -C 6 alkyl), —NH 2 , —NHC 1 -C 6 alkyl, —CONR′R′′, or —N(C 1 -C 6 alkyl) 2 where R′ and R′′ are independently alkyl, akenyl, alkynyl, aryl, or joined together to form a 4 to 7 member ring.
- aryl means a cyclic or polycyclic aromatic ring having from 5 to 12 carbon atoms, and being unsubstituted or substituted with up to 4 groups selected from C 1 -C 6 alkyl, cycloalkyl, heteroaryl, dialkylaminoalkoxy, or those recited above as substituents for alkyl.
- aryl includes both monovalent species, for example when Ar 2 is aryl, and divalent species, for example where Ar 1 is aryl.
- aryl groups include, but are not limited to, phenyl, biphenyl, naphthyl, 2-chlorophenyl, 3-chlorophenyl, 4-chlorophenyl, 2-methylphenyl, 3-methylphenyl, 4-methylphenyl, 2-methoxyphenyl, 3-methoxyphenyl, 4-methoxyphenyl, 2-chloro-3-methylphenyl, 2-chloro-4-methylphenyl, 2-chloro-5-methylphenyl, 3-chloro-2-methylphenyl, 3-chloro-4-methylphenyl, 4-chloro-2-methylphenyl, 4-chloro-3-methylphenyl, 5-chloro-2-methylphenyl, 2,3-dichlorophenyl, 2,5-dichlorophenyl, 3,4-dichlorophenyl, 2,3-dimethylphenyl, 3,4-dimethylphenyl, 4-trifluoromethyl and the like.
- heteroaryl means an aromatic mono-, bi-, or or polycyclic ring incorporating one or more (i.e. 1-4) heteroatoms selected from N, O, and S.
- heteroaryl includes both monovalent species, for example where Ar 2 is heteroaryl, and divalent species, for example where Ar 1 is heteroaryl. It is understood that a heterocycle is optionally substituted with up to 4 groups selected from C 1 -C 6 alkyl, cycloalkyl, heteroaryl, dialkylaminoalkoxy, or those recited above as substituents for alkyl.
- Suitable monocyclic heteroaryl include, but are not limited to substituted or unsubstituted thienyl, furanyl, pyrrolyl, imidazolyl, pyrazolyl, isothiazolyl, isoxazolyl, triazolyl, tetrazolyl, pyridinyl, pyrazinyl, pyrimidinyl, piperidinyl, pyrrolidinyl, piperazinyl, azetidinyl, aziridinyl, morpholinyl, thietanyl, oxetaryl.
- Preferred monocyclic diheterocycles include, but are not limited to 1-, 2-, 4-, or 5-imidazolyl, 1-, 3-, 4-, or 5-pyrazolyl, 3-, 4-, or 5-isothiazolyl, 3-, 4-, or 5-isoxazolyl, 1,3-, or 5-triazolyl, 1-, 2-, or 3-tetrazolyl, 2-pyrazinyl, 2-, 4-, or 5-pyrimidinyl, 1- or 2-piperazinyl, 2-, 3-, or 4-morpholinyl.
- suitable bicyclic and polyclic heteroaryl groups include but are not limited to include but are not limited to 1-, 2-, 3-, 5-, 6-, 7-, or 8-indolizinyl, 1-, 3-, 4-, 5-, 6-, or 7-isoindolyl, 2-, 3-, 4-, 5-,6-, or 7-indolyl, 2-, 3-, 4-, 5-, 6-, or 7-indazolyl, 2-, 4-, 5-, 6-, 7-, or 8-purinyl, 1-, 2-, 3-, 4-, 6-, 7-, 8-, or 9-quinolinzinyl, 2-, 3-, 4-, 5-, 6-, 7-, or 8-quinoliyl, 1-, 3-, 4-, 5-, 6-, 7-, or 8-isoquinoliyl, 1-, 4-, 5-, 6-, 7-, or 8-phthalazinyl, 2-, 3-, 4-, 5-, or 6-naphthyridinyl, 2-, 3-, 5-, 6-, 7-, or 8-in
- Typical fused heteroaryl groups include, but are not limited to 2-, 3-, 4-, 5-, 6-, 7-, or 8-quinolinyl, 1-, 3-, 4-, 5-, 6-, 7-, or 8-isoquinolinyl, 2-, 3-, 4-, 5-, 6-, or 7-indolyl, 2-, 3-, 4-, 5-, 6-, or 7-benzo[b]thienyl, 2-, 4-, 5-, 6-, or 7-benzoxazolyl, 2-, 4-, 5-, 6-, or 7-benzimidazolyl, 2-, 4-, 5-, 6-, or 7-benzothiazolyl.
- hydrocarbon-heteroatom chain refers to a hydrocarbon chain wherein one or more carbon atoms are replaced with a heteroatom.
- the hydrocarbon-heteroatom chain is optionally substituted with one or more substituents selected from lower alkyl, lower :alkoxy, lower thioalkoxy, —O(CH 2 ) 0-2 CF 3 , halogen, nitro, cyano, ⁇ O, ⁇ S, —OH, —SH, —CF 3 , —CO 2 H, —CO 2 C 1 -C 6 alkyl, —NH 2 , —NHC 1 -C 6 alkyl, —CONR′R′′, or —N(C 1 -C 6 alkyl) 2 where R′ and R′′ are independently alkyl, akenyl, alkynyl, aryl, or joined together to form a 4 to 7 member ring.
- heteroalkylene refers to an alkylene radical as defined above, that includes one or more heteroatoms such as oxygen, sulfur, or nitrogen (with valence completed by hydrogen or oxygen) in the carbon chain or terminating the carbon chain.
- lower alkoxy and “lower thioalkoxy” as used herein refers to O-alkyl or S-alkyl of from 1 to 6 carbon atoms as defined above for “lower alkyl.”
- cycloalkenyl means a cycloalkyl group having one or more carbon-carbon double.
- Example includes cyclobutene, cyclopentene, cyclohexene, cycloheptene, cyclobutadiene, cyclopentadiene, and the like.
- the symbol “ ” means a bond to a group wherein a 4 to 8 membered ring is formed. Typically this symbol will appear in pairs.
- patient means all mammals including humans. Examples of patients include, humans, cows, dogs, cats, goats, sheep, pigs, and rabbits.
- a “therapeutically effective amount” is an amount of a compound of the present invention that when administered to a patient ameliorates a symptom of dyslipidemia, non-insulin dependent diabetes mellitus, obesity, hyperglycemia, hypercholesteremia, hyperlipidemia, atherosclerosis, hypertriglyceridemia, hyperinsulinemia, glucose disorders associated with circulating glucocorticoids, growth hormone, catecholamines, glucagon, or parathyroid hormone. Additionally, a “therapeutically effective amount” is an amount of a compound of the present invention that when administered to a patient ameliorates a symptom of an eating disorder suppresses appetite, or modulates leptin levels.
- a pharmaceutically acceptable salt refers to the relatively non-toxic, inorganic and organic base or acid addition salts of compounds of the present invention. These salts can be prepared in situ during the final isolation and purification of the compounds or by separately reacting the purified compound in its free form with a suitable organic or inorganic base or acid and isolating the salt thus formed.
- Representative salts include the hydrobromide, hydrochloride, sulfate, bisulfate, nitrate, acetate, oxalate, valerate, oleate, palmitate, stearate, laurate, borate, benzoate, lactate, phosphate, tosylate, citrate, maleate, fumarate, succinate, tartrate, naphthylate mesylate, glucoheptonate, lactobionate, and laurylsulphonate salts, and the like.
- alkali and alkaline earth metals such as sodium, lithium, potassium, calcium, magnesium, and the like
- non-toxic ammonium, quaternary ammonium, and amine cations including, but not limited to ammonium, tetramethylammonium, tetraethylammonium, methylamine, dimethylamine, trimethylamine, triethylamine, ethylamine, and the like.
- the free base form may be regenerated by contacting the salt form with a base. While the free base may differ from the salt form in terms of physical properties, such as solubility, the salts are equivalent to their respective free bases for the purposes of the present invention.
- X 0 and X 1 are independently absent, O, S, —CH 2 —, —CH 2 —CH 2 —, —CH ⁇ CH—, —CH ⁇ CH—, —S(O) 2 —, or —S(O)—;
- Ar 1 and Ar 2 are each independently unsubstituted or substituted aryl or heteroaryl, provided that Ar 1 is not thiazolyl or oxazolyl; is absent; or when present, is a saturated or unsaturated hydrocarbon chain which is substituted or unsubstituted, wherein said chain has from 1 to 4 atoms so that Ar 1 , X 1 , (CH 2 ) r , and Ar 2 , together form a five to eight membered ring;
- R 1 and R 2 are selected from hydrogen, lower alkyl, lower alkoxy, haloalkyl, —O—(CH 2 ) m CF 3 , halogen, nitro, cyano, —OH, —SH, —CF 3 , —S(O) p alkyl, S(O) p aryl, —(CH 2 ) m OR 5 , —(CH 2 ) m NR 6 R 7 , —COR 5 , CO 2 R 5 , or —NR 6 R 7 , or together with the atoms to which they are attached form a five to eight member ring;
- R 3 and R 4 are selected from hydrogen, lower alkyl, lower alkoxy, haloalkyl, —O—(CH 2 ) m CF 3 , halogen, nitro, cyano, —OH, —SH, —CF 3 , —S(O) p alkyl, S(O) p aryl, —(CH 2 ) m OR 5 , —(CH 2 ) m NR 6 R 7 , —COR 5 , —CO 2 H, —CO 2 R 5 , or —NR 6 R 7 ;
- R 1 -R 4 is H, lower alkyl, lower alkoxy, haloalkyl, —O—(CH 2 ) m CF 3 , halogen, nitro, cyano, —OH, —SH, —CF 3 , —S(O) p alkyl, S(O) p aryl, —(CH 2 ) m OR 5 , —(CH 2 ) m NR 6 R 7 , or NR 6 R 7 ;
- R 5 is hydrogen, alkyl, alkenyl, alkynyl, or aryl
- R 6 and R are each independently hydrogen, alkyl, alkenyl, alkynyl, —COalkyl, —COaryl, cycloalkyl, —CO 2 alkyl, —CO 2 aryl, or R 6 and R 7 together with the atoms to which they are attached form a 4 to 7 membered ring having 1 to 3 heteroatoms;
- n 0 to 5;
- p 0, 1, or 2;
- r 0 to 6.
- R 1 , R 2 , R 3 , and R 4 are preferably selected from hydrogen, alkyl, or alkoxy. More preferably, R 2 and R 3 are hydrogen; and R 1 and R 4 are alkyl or alkoxy. In a particularly preferred embodiment of Formula I, R 2 and R 3 are hydrogen; R 1 is alkyl; and R 4 is alkoxy. Preferred alkoxy include methoxy, ethoxy, isopropoxy, n-propoxy, t-butoxy, n-butoxy, or isobutoxy. Similarly, preferred alkyl include methyl, ethyl, isopropyl, n-propyl, t-butyl, n-butyl, or isobutyl. In a most preferred embodiment of Formula I, q is 1, Ar 1 is phenyl, X 1 is absent, r is 0, V 1 is absent, and Ar 2 is 4-trifluoromethylphenyl.
- (CH 2 ) t wherein t is 1 to 4. Additionally, is optionally substituted with at least one substituent, wherein the substituent include but are not limited to lower alkyl, lower alkoxy, lower thioalkoxy, —O(CH 2 ) 0-2 CF 3 , halogen, nitro, cyano, ⁇ O, ⁇ S, —OH, —SH, —CF 3 , OCF 3 , —CO 2 H, —CO 2 C 1 -C 6 alkyl, —NH 2 , —NHC 1 -C 6 alkyl, —CONR′R′′, or —N(C 1 -C 6 alkyl) 2 where R′ and R′′ are independently alkyl, akenyl, alkynyl, aryl, or joined together to form a 4 to 7 member ring.
- the substituent include but are not limited to lower alkyl, lower alkoxy, lower thioalkoxy, —O(CH 2 )
- R 1 and R 2 are joined together to form a five to eight member ring having Formula II.
- a ring includes, for example, cycloalkyl, aryl, heterocycloalkyl, or a heteroaryl rings where each such ring is optionally substituted as described above. and pharmaceutically acceptable salts thereof, wherein:
- X 0 and X 1 are independently absent, O, S, —CH 2 —, —CH 2 —CH 2 —, —CH ⁇ CH—, —CH ⁇ CH—, —S(O) 2 —, or —S(O)—;
- Ar 1 and Ar 2 are each independently unsubstituted or substituted aryl or heteroaryl, provided that Ar 1 is not thiazolyl or oxazolyl; is absent; or when present, a saturated or unsaturated hydrocarbon chain which is substituted or unsubstituted, wherein said chain has from 1 to 4 atoms so that Ar 1 , X 1 , (CH 2 ) r , and Ar 2 , together form a five to eight membered ring;
- R 3 and R 4 are selected from hydrogen, lower alkyl, lower alkoxy, haloalkyl, —O—(CH 2 ) m CF 3 , halogen, nitro, cyano, —OH, —SH, —CF 3 , —S(O) p alkyl, S(O) p aryl, —(CH 2 ) m OR 5 , —(CH 2 ) m NR 6 R 7 , —COR 5 , —CO 2 H, —CO 2 R 5 , or —NR 6 R 7 ;
- R 5 is hydrogen, alkyl, alkenyl, alkynyl, or aryl
- R 6 and R 7 are each independently hydrogen, alkyl, alkenyl, alkynyl, —COalkyl, —COaryl, cycloalkyl, —CO 2 alkyl, —CO 2 aryl, —SO 2 alkyl, —SO 2 aryl, or R 6 and R 7 together with the atoms to which they are attached form a 4 to 7 membered ring having 1 to 3 heteroatoms; is a saturated or unsaturated, substituted or unsubstituted hydrocarbon chain or hydrocarbon-heteroatom chain having from 3 to 6 atoms wherein the carbon atom of position 2 is connected to the carbon atom of position 3 to form a five to eight member ring;
- n 0 to 5;
- p 0 to 2;
- r 0 to 6.
- substituents selected from the group consisting of lower alkyl, lower alkoxy, lower thioalkoxy, —O(CH 2 ) 1-5 CF 3 , halogen, nitro, cyano, ⁇ O, ⁇ S, —OH, —SH, —CF 3 , —CO 2 H, —CO 2 C 1 -C 6 alkyl, —N 2 , —NHC 1 -C 6 alkyl, —OCH 2 O—, and —N(C 1 -C 6 alkyl) 2 .
- R 3 , and R 4 are preferably selected from hydrogen, alkyl, or alkoxy. More preferably, R 3 and R 4 are hydrogen; and is a saturated or unsaturated, unsubstituted hydrocarbon chain having from 3 to 6 atoms wherein the carbon atom of position 2 is connected to the carbon atom of position 3 to form a five to eight member ring such as a cyclopentyl or cyclohexyl ring.
- q is 1
- Ar 1 is phenyl
- X 1 is O
- r is 1
- V 1 is absent
- Ar 2 is 4-trifluoromethylphenyl.
- the substituent include but are not limited to lower alkyl, lower alkoxy, lower thioalkoxy, —O(
- a subset of exemplary compounds of Formula I and Formula II include
- Certain of the compounds of the present invention possess one or more chiral centers and each center may exist in the R or S configuration.
- the present invention includes all diastereomeric, enantiomeric, and epimeric forms as well as the appropriate mixtures thereof. Stereoisomers may be obtained, if desired, by methods known in the art as, for example, the separation of stereoisomers by chiral chromatographic columns. Additionally, the compounds of the present invention may exist as geometric isomers.
- the present invention includes all cis, trans, syn, anti,
- E
- Z isomers as well as the appropriate mixtures thereof
- compounds may exist as tautomers. All tautomers are included within Formulas I and II and are provided by this invention.
- the compounds of the present invention can exist in unsolvated as well as solvated forms with pharmaceutically acceptable solvents such as water, ethanol, and the like.
- the solvated forms are considered equivalent to the unsolvated forms for the purposes of the present invention.
- the present invention includes all pharmaceutically acceptable, non-toxic esters of the compounds of Formulae I and II.
- esters include C 1 -C 6 alkyl esters where the alkyl group is a straight or branched chain.
- Acceptable esters also include C 5 -C 7 cycloalkyl esters as well as arylalkyl esters such as, but not limited to benzyl.
- C 1 -C 4 esters are preferred.
- Esters of the compounds of the present invention may be prepared according to conventional methods.
- the compounds of the present invention are suitable to be administered to a patient for the treatment, control, or prevention of hypercholesteremia, dyslipidemia, obesity, hyperglycemia, hypercholesteremia, atherosclerosis, hypertriglyceridemia, and hyperinsulinemia.
- the compounds of the present invention are also suitable to be administered to a patient for the suppression of appetite and modulation of leptin. Accordingly, the compounds may be administered to a patient alone or as part of a composition that contains other components such as excipients, diluents, and carriers, all of which are well-known in the art.
- compositions can be administered to humans and/or animals either orally, rectally, parenterally (intravenously, intramuscularly, or subcutaneously), intracisternally, intravaginally, intraperitoneally, intravesically, locally (powders, ointments, or drops), or as a buccal or nasal spray.
- compositions suitable for parenteral injection may comprise physiologically acceptable sterile aqueous or nonaqueous solutions, dispersions, suspensions or emulsions, and sterile powders for reconstitution into sterile injectable solutions or dispersions.
- suitable aqueous and nonaqueous carriers, diluents, solvents or vehicles include water, ethanol, polyols (propyleneglycol, polyethyleneglycol, glycerol, and the like), suitable mixtures thereof, vegetable oils (such as olive oil), and injectable organic esters such as ethyl oleate.
- Proper fluidity can be maintained, for example, by the use of a coating such as lecithin, by the maintenance of the required particle size in the case of dispersions and by the use of surfactants.
- compositions may also contain adjuvants such as preserving, wetting, emulsifying, and dispensing agents.
- adjuvants such as preserving, wetting, emulsifying, and dispensing agents.
- Prevention of the action of microorganisms can be ensured by various antibacterial and antifungal agents, for example, parabens, chlorobutanol, phenol, sorbic acid, and the like.
- isotonic agents for example sugars, sodium chloride, and the like.
- Prolonged absorption of the injectable pharmaceutical form can be brought about by the use of agents delaying absorption, for example, aluminum monostearate and gelatin.
- Solid dosage forms for oral administration include capsules, tablets, pills, powders, and granules.
- the active compound is admixed with at least one inert customary excipient (or carrier) such as sodium citrate or dicalcium phosphate or
- fillers or extenders as for example, starches, lactose, sucrose, glucose, mannitol, and silicic acid;
- binders as for example, carboxymethylcellulose, alginates, gelatin, polyvinylpyrrolidone, sucrose, and acacia;
- humectants as for example, glycerol;
- disintegrating agents as for example, agar, calcium carbonate, potato or tapioca starch, alginic acid, certain complex silicates, and sodium carbonate;
- solution retarders as for example paraffin;
- absorption accelerators as for example, quaternary ammonium compounds;
- wetting agents as for example
- compositions of a similar type may also be employed as fillers in soft and hard-filled gelatin capsules using such excipients as lactose or milk sugar, as well as high molecular weight polyethyleneglycols, and the like.
- Solid dosage forms such as tablets, dragees, capsules, pills, and granules can be prepared with coatings and shells, such as enteric coatings and others well-known in the art. They may contain opacifying agents, and can also be of such composition that they release the active compound or compounds in a certain part of the intestinal tract in a delayed manner. Examples of embedding compositions which can be used are polymeric substances and waxes. The active compounds can also be in micro-encapsulated form, if appropriate, with one or more of the above-mentioned excipients.
- Liquid dosage forms for oral administration include pharmaceutically acceptable emulsions, solutions, suspensions, syrups, and elixirs.
- the liquid dosage forms may contain inert diluents commonly used in the art, such as water or other solvents, solubilizing agents and emulsifiers, as for example, ethyl alcohol, isopropyl alcohol, ethyl carbonate, ethyl acetate, benzyl alcohol, benzyl benzoate, propyleneglycol, 1,3-butyleneglycol, dimethylformamide, oils, in particular, cottonseed oil, groundnut oil, corn germ oil, olive oil, castor oil and sesame oil, glycerol, tetrahydrofurfuryl alcohol, polyethyleneglycols and fatty acid esters of sorbitan or mixtures of these substances, and the like.
- inert diluents commonly used in the art, such as water or other solvents, solubilizing
- composition can also include adjuvants, such as wetting agents, emulsifying and suspending agents, sweetening, flavoring, and perfuming agents.
- adjuvants such as wetting agents, emulsifying and suspending agents, sweetening, flavoring, and perfuming agents.
- Suspensions in addition to the active compounds, may contain suspending agents, as for example, ethoxylated isostearyl alcohols, polyoxyethylene sorbitol and sorbitan esters, microcrystalline cellulose, aluminum metahydroxide, bentonite, agar-agar and tragacanth, or mixtures of these substances, and the like.
- suspending agents as for example, ethoxylated isostearyl alcohols, polyoxyethylene sorbitol and sorbitan esters, microcrystalline cellulose, aluminum metahydroxide, bentonite, agar-agar and tragacanth, or mixtures of these substances, and the like.
- compositions for rectal administrations are preferably suppositories which can be prepared by mixing the compounds of the present invention with suitable non-irritating excipients or carriers such as cocoa butter, polyethyleneglycol, or a suppository wax, which are solid at ordinary temperatures, but liquid at body temperature and therefore, melt in the rectum, or vaginal cavity and release the active component.
- suitable non-irritating excipients or carriers such as cocoa butter, polyethyleneglycol, or a suppository wax, which are solid at ordinary temperatures, but liquid at body temperature and therefore, melt in the rectum, or vaginal cavity and release the active component.
- Dosage forms for topical administration of a compound of this invention include ointments, powders, sprays, and inhalants.
- the active component is admixed under sterile conditions with a physiologically acceptable carrier and any preservatives, buffers, or propellants as may be required.
- Ophthalmic formulations, eye ointments, powders, and solutions are also contemplated as being within the scope of this invention.
- the compounds of the present invention can be administered to a patient at dosage levels in the range of about 0.1 to about 2,000 mg per day.
- dosage levels in the range of about 0.1 to about 2,000 mg per day.
- a dosage in the range of about 0.01 to about 10 mg per kilogram of body weight per day is preferable.
- the specific dosage used can vary.
- the dosage can depend on a numbers of factors including the requirements of the patient, the severity of the condition being treated, and the pharmacological activity of the compound being used. The determination of optimum dosages for a particular patient is well-known to those skilled in the art.
- the present invention contains compounds that can be synthesized in a number of ways familiar to one skilled in organic synthesis.
- the compounds outlined herein can be synthesized according to the methods described below, along with methods typically utilized by a synthetic chemist, and combinations or variations of those methods, which are generally known to one skilled in the art of synthetic chemistry.
- the synthetic route of compounds in the present invention is not limited to the methods outlined below. It is assumed one skilled in the art will be able to use the schemes outlined below to synthesize compounds claimed in this invention.
- Individual compounds may require manipulation of the conditions in order to accommodate various functional groups. A variety of protecting groups generally known to one skilled in the art may be required. Purification, if necessary, can be accomplished on a silica gel column eluted with the appropriate organic solvent system. Also, reverse phase HPLC or recrystallization may be employed.
- R 1 , R 2 , R 3 , R 4 , X 0 , X 1 , V 1 , Ar 1 , and Ar 2 are the same as defined above for Formula I;
- R 11 is a lower alkyl
- X is a halogen
- the resulting ester is then preferably hydrolyzed to form the compounds of Formulas I and II.
- compounds of Formulas I and II can be prepared using the synthetic route outlined in Scheme 1-6.
- Scheme 1 covers the preparation of compounds of Formulas I and II wherein X 0 is S, q is 0-3, is absent, X 1 is absent and r is 0.
- Compounds of the general formula A are thiocyanated with a mixture of bromine and sodium thiocyanate to give compounds of the general formula B.
- Compounds of the general formula B are then alkylated with an alkyl haloacarboxylate to give compounds of the general formula C.
- the preferred alkyl haloacarboxylate is methyl bromoacetate.
- Alternate routes to compounds of formula C will be readily apparent to a person skilled in the art of organic synthesis.
- Compounds of the general formula D are then prepared by reduction of C with dithiothreitol in methanol.
- Scheme 2 covers the preparation of compounds of Formula I-II wherein X 0 is O, q is 0-3, X 1 is absent, is absent, and r is 0.
- Compounds of the general formula A are alkylated with an alkyl haloacarboxylate to give compounds of the general formula G.
- the preferred alkyl halocarboxylate is methyl bromoacetate. Alternate routes to compounds of formula G when Y is O will be readily apparent to a person skilled in the art of organic synthesis.
- Scheme 3 covers the preparation of compounds of formula I-II wherein X 0 is —CH 2 —CH 2 —, —C ⁇ C—, q is 0-3, X 1 is absent, is absent, and r is 0.
- Compounds of the general formula A are brominated with bromine, using acetic acid as solvent to give L.
- N-bromosuccinimide may be used in place of bromine and dichloromethane in place of acetic acid as solvent.
- Compounds of the general formula L are then alkylated with an alkyl haloacarboxylate to give compounds of the general formula M.
- the preferred alkyl halocarboxylate is methyl bromoacetate.
- Scheme 4 covers the preparation of compounds of formula I-II wherein X 0 is —CH 2 —CH 2 —, q is 0-3, X 1 is absent, is absent, and r is 0. Accordingly, compounds of the general N can be reduced with hydrogen and palladium as catalyst to give compounds of the general formula P which are then saponified LiOH in THF to give the final compound Q.
- Scheme 5 covers the preparation of compounds of formula I-II wherein X 0 is absent, q is 0, X 1 is absent, is absent, and r is 0 are prepared.
- Compounds of the general formula M are allowed to react with tetrakis (triphenylphosphine) palladium(0) and biphenyl compounds of the general formula HH to give compounds of the general formula R.
- Compounds of the general formula HH are prepared as described in Scheme 10 or are readily available from commercial sources.
- Compounds of the general formula R are then saponified with LiOH in THF to give the final compound S.
- compounds of the general formula X are prepared by reacting aryl boronic acid W with aryl bromide V in the presence of Pd(0) and cesium carbonate. Compounds of the general formula X are then reacted with methanesulfonyl chloride to give chlorides of the general formula Y.
- compounds of the general formula HH are prepared by reacting aryl bromide V with boronic acid FF to give GG.
- Compounds of the general formula GG are then reacted with an alkyl lithium reagent and then quenched with a borate which is hydrolyzed to give compounds of the general formula HH.
- compounds of the general formula NN can be prepared by reacting an appropriately substituted aryl amine LL under Sandmeyer conditions followed by heating to give intermediate MM. The resulting intermediate MM is then reacted with methanesulfonyl chloride to give chlorides of the general formula NN.
- the reaction was flushed with nitrogen and heated to 90° C. for 1 h.
- the reaction was cooled, poured into diethyl ether and washed with water (2 ⁇ 10 ml), brine (1 ⁇ 10 ml) and dried over anhydrous sodium sulfate.
- the organic solution was filtered through silica gel, eluted with diethyl ether, and concentrated.
- the crude product was dissolved in 10 ml dichloromethane. Added to this solution were triethylamine (202 mg, 2 mmol) then methanesulfonyl chloride (184 mg, 1.6 mmol) and allowed to stir at ambient temperature for 18 h.
- the reaction was poured into water and extracted with methylene chloride.
- the reaction mixture was diluted with 5.0 ml of water and made strongly acidic by the addition of 4.0 N hydrochloric acid.
- the mixture was extracted with ethyl acetate (4 ⁇ 30 ml), and the combined extracts were washed with brine (2 ⁇ 50 ml), then dried over anhydrous sodium sulfate and concentrated.
- the crude product was recrystallized from ethyl acetate/hexane. mp 132-134° C.
- Step 5 Preparation of ⁇ 7-[4-(4-trifluoromethyl-benzyl)-benzylsulfanyl]-indan-4-yloxy ⁇ -acetic acid (compound 40)
- the title compound was prepared in the manner analogous to Example 1 using 40D.
- reaction was allowed to come to room temperature and then heated to 45° C. for 45 minutes. The reaction was then cooled, concentrated in vacuo, and the residue taken up in EtOAc. The organic layer was then washed with 2 M HCl (1 ⁇ 100 mL), dried (Na 2 SO 4 ) and the solvent removed in vacuo.
- Step 4 Preparation of ⁇ 7-[4-(4-Chloro-benzyloxy)-benzylsulfanyl]-indan-4-yloxy ⁇ -acetic acid (compound 70)
- the title compound was prepared in the manner analogous to Example 1 using 70A.
- Examples 103-134 were prepared using combinatorial chemistry analogous to that previously described in Examples 1-102.
- the combinatorial chemistry methods useful in the present invention include those where an activated alcohol is contacted with a thiol followed by saponification of the resulting ester to afford the desired products. Such methods can be illustrated by previously described Scheme 1 where compound D is an exemplary thiol, compound Y is an exemplary activated alcohol, and compound F is an exemplary desired product.
- Examples 103-134 were synthesized in the following fashion using either the thiol products 2C, 1D, WW, XX, YY or ZZ and the appropriate activated alcohol or alkyl halide. Each thiol was diluted to 0.15M with CH 3 CN and each activated alcohol diluted to 0.15M with CH 3 CN.
- Each alkylation product was diluted with 3.0 mL of 0.5M LiOH in 4:1 methoxyethanol:H 2 O shaken at 60° C. for 4 h and cooled to ambient temperature. To each reaction, was then added 1.0 mL 1N HCl and 1.0 mL brine. Each reaction was extracted twice with 2.0 mL EtOAc, and the organic layers concentrated to afford the desired products.
- the compounds of the present invention have demonstrated PPAR modulating activity in the standard assays commonly employed by those skilled in the art. Accordingly, such compounds and formulations comprising such compounds are useful for suppressing appetite, modulating leptin, and treating, preventing or controlling hypercholesterolemia, dyslipidemia, obesity, eating disorders, hyperglycemia, atherosclerosis, hypertriglyceridemia, hyperinsulinemia and diabetes.
- Test A Transient Transfections Assay using the HepG2Hepatoma Cell Line.
- HepG2 cells were transiently transfected with an expression plasmids encoding hPPAR ⁇ , hPPAR ⁇ or mPPAR ⁇ chimeric receptors and a reporter containing the yeast upstream activating sequence (UAS) upstream of the viral E1B promoter controlling a luceriferase reporter gene.
- UAS yeast upstream activating sequence
- HepG2 cells were grown in DMEM supplemented with 10% FBS and 1 ⁇ M non-essential amino acid. On the first day, cells were split into 100 mm dishes at 2.5 ⁇ 10 8 /dish and incubated overnight at 37° C./5% CO 2 .
- the cells were transiently transfected with plasmid DNA encoding a chimeric receptor, the luciferase reporter gene; and ⁇ -gal.
- plasmid DNA encoding a chimeric receptor, the luciferase reporter gene; and ⁇ -gal.
- lucifease reporter (PG5E1b) DNA 15 ⁇ g of Gal4-PPAR chimeric receptor DNA, and 1.5 ⁇ g of ⁇ -gal plasmid DNA were mixed with 1.4 ml of opti-MEM in the tube.
- 28 ⁇ l of LipoFectamine-2000 reagent was added to 1.4 ml of opti-MEM in the tube, and incubate for 5 min at RT.
- the diluted LipoFectamine-2000 reagent was combined with the DNA mixture, and incubate for 20 min at RT. After fresh medium was added to each 100 mm dish of cells, 2.8 ml of Lipofectamine2000-DNA mixture was added dropwise to the 100 mm dish containing 14 ml of medium, and incubate 37° C. overnight. On day three cells were trypsinized off the 100 mm dishes and re-plated on 96 well plates. Cells were plated at 2.5 ⁇ 10 4 cells per well in 150 ⁇ l of media and 50 ⁇ l of compound diluted by media was added. The test compound added were in the range from 50 ⁇ M to 50 pM. After addition of compounds, the plates were incubated at 37° C. for 24 hours.
- Subseqently cells were washed with once with 100 ⁇ l of PBS, lysed, and processed for measuring luciferase and ⁇ -gal activity using Dual-Light luciferase kit from Tropix®, according to the manufacturer's recommendations, on an EG&G Bethold MicroLumat LB96P luminometer. EC 50 values were obtained using the GraphPad PrismTM program. Surprisingly, the compounds of the present invention exhibit activity for both PPAR ⁇ and PPAR ⁇ .
- Hep G2-hBeta EC 50 's (“EC 50 ⁇ ”) and Hep G2-hAlpha EC 50 's (“EC 50 ⁇ ”) from greater than zero to about 20 ⁇ M.
- the Hep G2-hBeta EC 50 's and Hep G2-hAlpha EC 50 's for the compounds of the present invention fall within the following 6 groups:
- mice transgenic for human apoA1 were purchased from Jackson laboratories. All animals were allowed normal chow (Ralston-Purina) and water ad libitum in temperature controlled rooms, under a 12-h light, 12-h dark cycle beginning with lights on at 6 AM. During the treatment phase of the study the mice were dosed daily between 6 and 9 AM by oral gavage using a suspension vehicle of 1.5% carboxymethyl-cellulose plus 0.2 percent Tween-20 (CMC/Tween) containing the specifid compounds. Control animals received vehicle alone. Vehicle volume represented 0.25 percent of body weight. Under anesthesia, tail blood was obtained weekly in the morning at the indicated days of study.
- CMC/Tween Tween-20
- tissue samples liver, intestine, fat, and muscle
- tissue samples liver, intestine, fat, and muscle
- Each of the compounds of the present invention that were tested effected a significant increase in HDL over the values observed for the control animals. Furthermore, these compounds resulted in triglyceride levels which were lower than observed in controls.
- Componds of the present invention tested in the hApoAI transgenic mouse model showed a range of HDL-c elevation and triglyceride lowering when dosed at 30 mg/kg/day.
- Example 4 raised HDL-c 97% and lowered triglycerides 65 % relative to the control population,
- Example 6 raised HDL-c 24% and lowered triglycerides 59 % relative to the control population, and
- Example 3 raised HDL-c 9% and lowered triglycerides 70% relative to the control population.
- Cynomolgus monkeys that were either insulin resistant or diabetic (type II) were treated for eight weeks with [5-Methoxy-2-methyl-4-(4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid in a rising dose fashion (0.1 to 1 mg/kg).
- Plasma was sampled bi-weekly and analyzed for glycemics and leptin.
- Body weight was also measured at various time points across the study.
- the diabetic moneky mean body weight data is presented in Table 2 and the insulin resistant monkey mean body weight data is presented in Table 3.
- Plasma leptin values presented as baseline values vs. 8-week treatment, were determined by ELISA and are presented in Table 4.
- the effect of [5-Methoxy-2-methyl-4-(4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid on plasma leptin values in diabetic and insulin resistant obese cynomolgus monkeys is demonstrated by the lesser increase in plasma leptin values seen in the treated animals.
- exogenous insulin requirement The amount of exogenous insulin the diabetic monkeys received to maintain proper glucose levels, the exogenous insulin requirement, is also presented in Table 4 as baseline values vs. 8-week treatment.
- insulin resistant monkeys are not yet diabetic and do not receive exogenous insulin.
- a reduction in exogenous insulin requirements is a measure of improved insulin sensitivity and glucose control.
- Diabetic Controls 89 ⁇ 71 101 ⁇ 73 Diabetic Treated 66 ⁇ 16 18 ⁇ 4 Leptin (ng/ml) Diabetic Controls 6.55 ⁇ 1.38 18.83 ⁇ 0.24 Diabetic Treated 7.22 ⁇ 0.93 10.89 ⁇ 2.7 Insulin Resistant Controls 9.85 ⁇ 0.45 13.60 ⁇ 3.45 Insulin Resistant Treated 7.33 ⁇ 1.16 5.73 ⁇ 2.14
- the compounds of the present invention can be administered alone or in combination with one or more therapeutic agents. These include, for example, other agents for the treatment, control, or prevention of hypercholesteremia, dyslipidemia, obesity, hyperglycemia, hypercholesteremia, atherosclerosis, hypertriglyceridemia, and hyperinsulinemia.
- the compounds of the present invention can be administered alone or in combination with one or more therapeutic agents for the supression of appetite and modulation of leptin.
- the compounds are thus well suited to formulation for convenient administration to mammals for the prevention and treatment of such disorders.
- Formulation 1 Ingredient Amount compound of Formulas I-V 0.5 to 800 mg sodium benzoate 5 mg isotonic saline 1000 mL
- the above ingredients are mixed and dissolved in the saline for IV administration to a patient.
- Formulation 2 Ingredient Amount compound of Formulas I-V 0.5 to 800 mg cellulose, microcrystalline 400 mg stearic acid 5 mg silicon dioxide 10 mg sugar, confectionery 50 mg
- the ingredients are blended to uniformity and pressed into a tablet that is well suited for oral administration to a patient.
- Formulation 3 Ingredient Amount compound of Formulas I-V 0.5 to 800 mg starch, dried 250 mg magnesium stearate 10 mg
- the ingredients are combined and milled to afford material suitable for filling hard gelatin capsules administered to a patient.
- Formulation 4 Ingredient Amount % wt./(total wt.) compound of Formulas I-V 1 to 50 Polyethylene glycol 1000 32 to 75 Polyethylene glycol 4000 16 to 25
- the ingredients are combined via melting and then poured into molds containing 2.5 g total weight.
Abstract
This invention discloses compounds that alter PPAR activity. The invention also discloses pharmaceutically acceptable salts of the compounds, pharmaceutically acceptable compositions comprising the compounds of their salts, and methods of using them as therapeutic agents for treating or preventing disipidemia, hypercholesteremia, obesity, eating disorders, hyperglycemia, atherosclerosis, hypertriglyceridemia, hyperinsulinemia and diabetes in a mammal as well as methods of suppressing appetite and modulating leptin levels in a mammal. The present invention also discloses methods for making the disclosed compounds.
Description
This application is a divisional application of U.S. Ser. No. 10/347,749, filed Jan. 22, 2003, which claims priority to U.S. Provisional Applications Ser. Nos. 60/370,508, filed Apr. 5, 2002 and 60/386,026, filed Jun. 5, 2002.
The present invention relates to compounds and pharmaceutical formulations that can be used to treat conditions mediated by nuclear hormone receptors, more specifically, to compounds and pharmaceutical formulations that modulate Peroxisome Proliferator Activation Receptor (PPAR) activity.
Hypercholesterolemia, dyslipidemia, diabetes, and obesity are well-recognized risk factors in the onset of atherosclerosis and coronary heart disease. The diseases are characterized by high levels of cholesterol and lipids in the blood. The blood cholesterol pool is generally dependent on dietary uptake of cholesterol from the intestine, and from the biosynthesis of cholesterol throughout the body, especially the liver. The majority of cholesterol in plasma is carried on apolipoprotein B-containing lipoproteins, such as low-density lipoproteins (LDL) and very-low-density lipoproteins (VLDL). The risk of coronary artery disease in man increases when LDL and VLDL levels increase. Conversely, high levels of cholesterol carried in high-density lipoproteins (HDL) is protective against coronary artery disease (Am. J. Med., 1977;62:707-714).
The statins represent perhaps the most important class of lipid-lowering drugs. These compounds inhibit HMG-CoA reductase which is implicated in the rate-limiting step in cellular cholesterol biosynthesis. Representative statins include atorvastatin, lovastatin, pravastatin, and simvastatin. The effectiveness of these compounds depends on LDL receptor regulation. Other important antilipidemia drugs include fibrates such as gemfibril and clofibrate, bile acid sequestrants such as cholestyramine and colestipol, probucol, and nicotinic acid analogs.
To date, a number of oral antidiabetic agents have been developed. The most commonly used hypoglygernic drugs are the sulfonylureas. Sulfonylureas are generally used to stimulate insulin. The biguanide metformin is generally used to improve insulin sensitivity and to decrease hepatic glucose output. Acarbose is used to limit postprandial hyperglycemia. Thiazolidine 2,4 diones are used to enhance insulin action without increasing insulin secretion.
Obesity is a chronic disease that is highly prevalent in modern society and is associated not only with a social stigma, but also with decreased life span and numerous medical problems, including diabetes mellitus, insulin resistance, hypertension, hypercholesterolemia, thromboembolic disease, and coronary heart disease. Rissanen et al, British Medical Journal, 301:835-837 (1990). Treatment of obesity remains a problem and it is unclear whether dieting results in decreased long-term risk of early death. A further important obesity intervention is physical activity. Exercise, however, in general, has been found to be only moderately successful in promoting weight loss. A program combining both dieting and exercise as well as behaviour modification is widely viewed as the optimal approach to weight loss. Studies have demonstrated that the combination of both food restriction and exercise promote a substantial loss of fat while maintaining lean tissue.
Peroxisome Proliferator Activation Receptors (PPAR) are implicated in a number of biological processes and disease states including hypercholesterolemia, dyslipidemia, and diabetes. PPARs are members of the nuclear receptor superfamily of transcription factors that includes steroid, thyroid, and vitamin D receptors. They play a role in controlling expression of proteins that regulate lipid metabolism. Furthermore, the PPARs are activated by fatty acids and fatty acid metabolites. There are three PPAR subtypes PPAR α, PPAR β (also referred to as PPAR δ), and PPAR γ. Each receptor shows a different pattern of tissue expression, and differences in activation by structurally diverse compounds. PPAR γ, for instance, is expressed most abundantly in adipose tissue and at lower levels in skeletal muscle, heart, liver, intestine, kidney, vascular endothelial and smooth muscle cells as well as macrophages. PPAR receptors are associated with regulation of insulin sensitivity and blood glucose levels, macrophage differentiation, inflammatory response, and cell differentiation. Accordingly, PPARs have been associated with obesity, diabetes, carcinogenesis, hyperplasia, atherosclerosis, dyslipidemia, and hypercholesterolemia.
In addition, PPARα agonists lower plasma triglycerides and LDL cholesterol and are therefore useful in treating hypertriglyceridemia, dyslipidemia and obesity. PPAR γ is associated with the development of non-insulin-dependent diabetes mellitus (NIDDM), hypertension, coronary artery disease, dyslipidemia and certain malignancies. Finally, activation of PPAR β has been demonstrated to increase HDL levels. (Leibowitz, WO97/28149, August 1997.) More recently, a PPAR β selective agonist was reported to have shown a dose-related increase in serum HDL-C and decrease in LDL-C and VLDL-TG in insulin-resistant middle aged rhesus monkeys. (W. R. Oliver et al., PNAS, v. 98, pp. 5306-5311, 2001)
Antilipidemic, antidiabetic and anti-obesity agents are still considered to have non-uniform effectiveness. The effectiveness of antidiabetic and antilipidemic therapies is limited, in part because of poor patient compliance due to unacceptable side effects. These side effects include diarrhea and gastrointestinal discomfort, and in the case of antidiabetics, edema, hypoglycemia and hepatoxicity. Furthermore, each type of drug does not work equally well in all patients.
For the reasons set forth above, there is a need for novel antilipidemic, antidiabetic, and anti-obesity agents that can be used alone or in combination. Furthermore, activation of multiple PPARs, for instance, PPARβ alone or in combination with the simultaneous activation of PPAR α and/or PPAR γ, may be desirable in formulating a treatment for dyslipidemia in which HDL is increased and LDL lowered.
The present invention provides compounds capable of modulating PPAR activity. Compounds of the present invention are described by Formula I:
and pharmaceutically acceptable salts thereof, where:
and pharmaceutically acceptable salts thereof, where:
X0 and X1 are independently absent, O, S, —CH2—, —CH2—CH2—, —CH═CH—, —CH≡CH—, —S(O)2—, or —S(O)—;
Ar1 and Ar2 are each independently unsubstituted or substituted aryl or heteroaryl, provided that Ar1 is not thiazolyl or oxazolyl;
is absent; or when present,
is a saturated or unsaturated hydrocarbon chain which is substituted or unsubstituted, wherein said chain has from 1 to 4 atoms so that
Ar1, X1, (CH2)r, and Ar2, together form a five to eight membered ring;
is absent; or when present,
is a saturated or unsaturated hydrocarbon chain which is substituted or unsubstituted, wherein said chain has from 1 to 4 atoms so that
Ar1, X1, (CH2)r, and Ar2, together form a five to eight membered ring;
R1 and R2 are selected from hydrogen, lower alkyl, lower alkoxy, haloalkyl, —O—(CH2)mCF3, halogen, nitro, cyano, —OH, —SH, —CF3, —S(O)palkyl, S(O)paryl, —(CH2)mOR5, —(CH2)mNR6R7, —COR5, CO2R5, or —NR6R7, or together with the atoms to which they are attached form a five to eight member ring;
R3 and R4 are selected from hydrogen, lower alkyl, lower alkoxy, haloalkyl, —O—(CH2)mCF3, halogen, nitro, cyano, —OH, —SH, —CF3, —S(O)palkyl, S(O)paryl, —(CH2)mOR5, —(CH2)mNR6R7, —COR5, —CO2H, —CO2R5, or —NR6R7;
provided that at least one of R1-R4 is H, lower alkyl, lower alkoxy, haloalkyl, —O—(CH2)mCF3, halogen, nitro, cyano, —OH, —SH, —CF3, —S(O)palkyl, S(O)paryl, —(CH2)mOR5, —(CH2)mNR6R7, or NR6R7;
R5 is hydrogen, alkyl, alkenyl, alkynyl, or aryl;
R6 and R7 are each independently hydrogen, alkyl, alkenyl, alkynyl, —COalkyl, —COaryl, cycloalkyl, —CO2alkyl, —CO2aryl, or R6 and R7 together with the atoms to which they are attached form a 4 to 7 membered ring having 1 to 3 heteroatoms;
m is 0 to 5;
p is 0, 1, or 2;
q is 0 to 6; and
r is 0 to 6.
The invention also provides a compound of formula (II):
and pharmaceutically acceptable salts thereof, where:
and pharmaceutically acceptable salts thereof, where:
X0 and X1 are independently absent, O, S, —CH2—, —CH2—CH2—, —CH═CH—, —CH≡CH—, —S(O)2—, or —S(O)—;
Ar1 and Ar2 are each independently unsubstituted or substituted aryl or heteroaryl, provided that Ar1 is not thiazolyl or oxazolyl;
is absent; or when present,
is a saturated or unsaturated, hydrocarbon chain which is substituted or unsubstituted, wherein said chain has from 1 to 4 atoms so that
Ar1, X1, (CH2)r, and Ar2, together form a five to eight member ring;
is absent; or when present,
is a saturated or unsaturated, hydrocarbon chain which is substituted or unsubstituted, wherein said chain has from 1 to 4 atoms so that
Ar1, X1, (CH2)r, and Ar2, together form a five to eight member ring;
R3 and R4 are selected from hydrogen, lower alkyl, lower alkoxy, haloalkyl, —O—(CH2)mCF3, halogen, nitro, cyano, —OH, —SH, —CF3, —S(O)palkyl, S(O)paryl, —(CH2)mOR5, —(CH2)mNR6R7, —COR5, —CO2H, —CO2R5, or —NR6R7;
R5 is hydrogen, alkyl, alkenyl, alkynyl, or aryl;
R6 and R7 are each independently hydrogen, alkyl, alkenyl, alkynyl, —COalkyl, —COaryl, cycloalkyl, —CO2alkyl, —CO2aryl, —SO2alkyl, —SO2aryl, or R6 and R7 together with the atoms to which they are attached form a 4 to 7 membered ring having 1 to 3 heteroatoms;
is a saturated or unsaturated, substituted or unsubstituted hydrocarbon chain or hydrocarbon-heteroatom chain having from 3 to 6 atoms wherein the carbon atom of position 2 is connected to the carbon atom of position 3 to form a five to eight member ring;
is a saturated or unsaturated, substituted or unsubstituted hydrocarbon chain or hydrocarbon-heteroatom chain having from 3 to 6 atoms wherein the carbon atom of position 2 is connected to the carbon atom of position 3 to form a five to eight member ring;
m is 0 to 5;
p is 0 to 2;
q is 0 to 6; and
r is 0 to 6.
In still another embodiment of the present invention, a method of treating, preventing or controlling hypercholesteremia and dyslipidemia in a mammal is provided. The method comprises administering to the mammal in need thereof a therapeutically effective amount of the compounds of the present invention. Additionally, the compounds of the present invention are also useful in the method of the present invention for treating, preventing, or controlling obesity, eating disorders, hyperglycemia, atherosclerosis, hypertriglyceridemia, hyperinsulinemia and diabetes. Furthermore, the compounds of the present invention are also useful in the methods of suppressing appetite in a mammal, modulating leptin levels in a mammal, and treating a patient exhibiting glucose disorders associated with circulating glucocorticoids, growth hormone, catecholamines, glucagon, or parathyroid hormone. For each disease state treatable, preventable, or controllable by the method of the present invention, a therapeutically effective amount of the compounds of the present invention are administered to the mammal in need thereof.
In yet another embodiment of the present invention, a method for preparing compounds with Formulae I-II, or a pharmaceutically acceptable salt thereof, is provided. The method of this embodiment comprises reacting
in a solvent in the presence of a base such as cesium carbonate: with
where
in a solvent in the presence of a base such as cesium carbonate: with
where
X0 is OH or SH;
R11 is a lower alkyl; and
X is a halogen.
In yet another embodiment of the present invention, an alternative method for preparing compounds with Formulae I-II, or a pharmaceutically acceptable salt thereof, is provided. The method of this embodiment comprises reacting
where X is a halide, R1-R4 have any of the meanings defined above, and R11 is a lower alkyl with:
where - - - is a bond or is absent and wherein n, q, r, X0, X1,
Ar1 and Ar2 are as defined above for Formula I;
where - - - is a bond or is absent and wherein n, q, r, X0, X1,
Ar1 and Ar2 are as defined above for Formula I;
where - - - is a bond or is absent.
The double bond may optionally be removed, for instance, by hydrogenation and the resulting ester is preferably hydrolyzed to form the compounds of Formulas I or II.
In still another embodiment, the invention provides a process for preparing the compound of formula I-4 which is:
or a pharmaceutically acceptable salt thereof, comprising:
or a pharmaceutically acceptable salt thereof, comprising:
-
- (a) conversion of phenol 1A to the thiocyante 1B;
- (b) alkylation of phenol moiety of thiocyanate 1B to acetoxyester 1C;
- (c) reduction of the thiocyanate moiety in 1C to form thiol 1D;
- (d) alkylation of thiol 1D with chloride 3C to form 4a;
- (e) saponification of the ester moiety in 4a to form I-4;
where
- (a) conversion of phenol 1A to the thiocyante 1B;
R1 is hydrogen or together with R2 forms a 5 membered carbocyclic ring;
R2 is methoxy or together with R1 forms a 5 membered carbocyclic ring;
R3 is hydrogen or methyl;
R4 is hydrogen;
X1 is absent or O; and
r is 0 or 1.
The following definitions are used, unless otherwise described: halo is fluoro, chloro, bromo, or iodo. Alkyl, alkoxy, alkenyl, alkynyl, etc. denote both straight and branched groups; but reference to an individual radical such as “propyl” embraces only the straight chain radical, a branched chain isomer such as “isopropyl” being specifically referred to.
The term “alkyl” as used herein refers to a straight or branched, hydrocarbon of from 1 to 11 carbon atoms and includes, for example, methyl, ethyl, n-propyl, isopropyl, n-butyl, sec-butyl, isobutyl, tert-butyl, n-pentyl, n-hexyl, and the like. The alkyl group can also be substituted with one or more of the substituents selected from lower alkoxy, lower thioalkoxy, —O(CH2)1-5CF3, halogen, nitro, cyano, ═O, ═S, —OH, —SH, —CF3, —OCF3, —CO2H, CO2C1-C6alkyl, —NH2, —NHC1-C6alkyl, —CONR′R″, or —N(C1-C6alkyl)2 where R′ and R″ are independently alkyl, akenyl, alkynyl, aryl, or joined together to form a 4 to 7 membering ring. Preferred alkyl groups have from 1 to 6 carbon atoms (C1-C6alkyl).
The term “lower alkyl” as used herein refers to a subset of alkyl which means a straight or branched hydrocarbon radical having from 1 to 6 carbon atoms and includes, for example, methyl, ethyl, n-propyl, isopropyl, n-butyl, sec-butyl, isobutyl, tert-butyl, n-pentyl, n-hexyl, and the like. Optionally, herein lower alkyl is referred to as “C1-C6alkyl.”
The term “haloalkyl” as used herein refers to a lower alkyl radical, as defined above, bearing at least one halogen substituent, for example, chloromethyl, fluoroethyl, trifluoromethyl, or 1,1,1-trifluoroethyl and the like. Haloalkyl can also include perfluoroalkyl wherein all hydrogens of a loweralkyl group are replaced with fluorine atoms.
The term “alkenyl” means a straight or branched unsaturated hydrocarbon radical having from 2 to 12 carbon atoms and includes, for example, ethenyl, 1-propenyl, 2-propenyl, 1-butenyl, 2-butenyl, 1-pentenyl, 2-pentenyl, 3-methyl-3-butenyl, 1-hexenyl, 2-hexenyl, 3-hexenyl, 3-heptenyl, 1-octenyl, 1-nonenyl, 1-decenyl, 1-undecenyl, 1-dodecenyl, and the like.
The term “alkynyl” means a straight or branched hydrocarbon radical having from 2 to 12 carbon atoms having at least one triple bond and includes, for example, 1-propynyl, 1-butynyl, 3-butynyl, 1-pentynyl, 3-pentynyl, 3-methyl-3-butynyl, 1-hexynyl, 3-hexynyl, 3-heptynyl, 1-octynyl, 1-nonynyl, 1-decynyl, 1-undecynyl, 1-dodecynyl, and the like.
The term “alkylene” as used herein refers to a divalent group derived from a straight or branched chain saturated hydrocarbon having from 1 to 10 carbon atoms by the removal of two hydrogen atoms, for example, methylene, 1,2-ethylene, 1,1-ethylene, 1,3-propylene, 2,2-dimethylpropylene, and the like. The alkylene groups of this invention can be optionally substituted. The alkylene group can also be substituted with one or more of the substituents selected from lower alkyl, lower alkoxy, lower thioalkoxy, —O(CH2)1-5CF3, halogen, nitro, cyano, ═O, ═S, —OH, —SH, —CF3, —CO2H, —CO2C1-C6alkyl, —NH2, —NHC1-C6alkyl, —CONR′R″, or —N(C1-C6alkyl)2 where R′ and R″ are independently alkyl, akenyl, alkynyl, aryl, or joined together to form a 4 to 7 member ring. Preferred alkylene groups have from 1 to 6 carbon atoms (C1-C6 alkyl).
The term “cycloalkyl” means a hydrocarbon ring containing from 3 to 12 carbon atoms, for example, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, cycloctyl, decalinyl, norpinanyl, and adamantyl. Where possible, the cycloalkyl group may contain double bonds, for example, 3-cyclohexen-1-yl. The cycloalkyl ring may be unsubstituted or substituted by 1 to 3 substituents selected from alkyl, alkoxy, thioalkoxy, hydroxy, thiol, nitro, halogen, amino, alkyl and dialkylamino, formyl, carboxyl, CN, —NH—CO—R′, —CO—NHR′—, —CO2R′, —COR′, aryl, or heteroaryl, wherein alkyl, aryl, and heteroaryl are as defined herein. Examples of substituted cycloalkyl groups include fluorocyclopropyl, 2-iodocyclobutyl, 2,3-dimethylcyclopentyl, 2,2-dimethoxycyclohexyl, and 3-phenylcyclopentyl.
The term “heteroatom” as used herein represents oxygen, nitrogen, or sulfur (O, N, or S) as well as sulfoxyl or sulfonyl (SO or SO2) unless otherwise indicated.
The term “heterocycloalkyl” means a monocyclic, fused, bridged, or spiro bicyclic heterocyclic ring systems. Monocyclic heterocyclic rings contain from about 3 to 12 ring atoms, with from 1 to 5 heteroatoms selected from N, O, and S, and preferably from 3 to 7 member atoms, in the ring. Bicyclic heterocyclics contain from 7 to 17 member atoms, preferably 7 to 12 member atoms, in the ring. Bicyclic heterocyclics contain from about 7 to about 17 ring atoms, preferably from 7 to 12 ring atoms. Bicyclic heterocyclics rings may be fused, spiro, or bridged ring systems. Examples of heterocyclic groups include cyclic ethers (oxiranes) such as ethyleneoxide, tetrahydrofuran, dioxane, and substituted cyclic ethers, wherein the substituents are those described above for the alkyl and cycloalkyl groups. Typical substituted cyclic ethers include propyleneoxide, phenyloxirane (styrene oxide), cis-2-butene-oxide (2,3-dimethyloxirane), 3-Chlorotetrahydrofuran, 2,6-dimethyl-1,4-dioxane, and the like. Heterocycles containing nitrogen are groups such as pyrrolidine, piperidine, piperazine, tetrahydrotriazine, tetrahydropyrazole, and substituted groups such as 3-aminopyrrolidine, 4-methylpiperazin-1-yl, and the like. Typical sulfur containing heterocycles include tetrahydrothiophene, dihydro-1,3-dithiol-2-yl, and hexahydrothiepin-4-yl. Other commonly employed heterocycles include dihydro-oxathiol-4-yl, tetrahydro-oxazolyl, tetrahydro-oxadiazolyl, tetrahydrodioxazolyl, tetrahydrooxathiazolyl, hexahydrotriazinyl, tetrahydro-oxazinyl, morpholinyl, thiomorpholinyl, tetrahydropyrimidinyl, dioxolinyl, octahydrobenzofuranyl, octahydrobenzimidazolyl, and octahydrobenzothiazolyl. For heterocycles containing sulfur, the oxidized sulfur heterocycles containing SO or SO2 groups are also included. Examples include the sulfoxide and sulfone forms of tetrahydrothiophene.
The term “hydrocarbon chain” as used herein refers to a straight hydrocarbon of from 2 to 6 carbon atoms. The hydrocarbon chain is optionally substituted with one or more substituents selected from lower alkyl, lower alkoxy, lower thioalkoxy, —O(CH2)0-2CF3, halogen, nitro, cyano, ═O, ═S, —OH, —SH, —CF3, —CO2H, —CO2(C1-C6alkyl), —NH2, —NHC1-C6alkyl, —CONR′R″, or —N(C1-C6alkyl)2 where R′ and R″ are independently alkyl, akenyl, alkynyl, aryl, or joined together to form a 4 to 7 member ring.
The term “aryl” means a cyclic or polycyclic aromatic ring having from 5 to 12 carbon atoms, and being unsubstituted or substituted with up to 4 groups selected from C1-C6alkyl, cycloalkyl, heteroaryl, dialkylaminoalkoxy, or those recited above as substituents for alkyl. The term aryl includes both monovalent species, for example when Ar2 is aryl, and divalent species, for example where Ar1 is aryl. Examples of aryl groups include, but are not limited to, phenyl, biphenyl, naphthyl, 2-chlorophenyl, 3-chlorophenyl, 4-chlorophenyl, 2-methylphenyl, 3-methylphenyl, 4-methylphenyl, 2-methoxyphenyl, 3-methoxyphenyl, 4-methoxyphenyl, 2-chloro-3-methylphenyl, 2-chloro-4-methylphenyl, 2-chloro-5-methylphenyl, 3-chloro-2-methylphenyl, 3-chloro-4-methylphenyl, 4-chloro-2-methylphenyl, 4-chloro-3-methylphenyl, 5-chloro-2-methylphenyl, 2,3-dichlorophenyl, 2,5-dichlorophenyl, 3,4-dichlorophenyl, 2,3-dimethylphenyl, 3,4-dimethylphenyl, 4-trifluoromethyl and the like.
The term “heteroaryl” means an aromatic mono-, bi-, or or polycyclic ring incorporating one or more (i.e. 1-4) heteroatoms selected from N, O, and S. The term heteroaryl includes both monovalent species, for example where Ar2 is heteroaryl, and divalent species, for example where Ar1 is heteroaryl. It is understood that a heterocycle is optionally substituted with up to 4 groups selected from C1-C6 alkyl, cycloalkyl, heteroaryl, dialkylaminoalkoxy, or those recited above as substituents for alkyl. Examples of suitable monocyclic heteroaryl include, but are not limited to substituted or unsubstituted thienyl, furanyl, pyrrolyl, imidazolyl, pyrazolyl, isothiazolyl, isoxazolyl, triazolyl, tetrazolyl, pyridinyl, pyrazinyl, pyrimidinyl, piperidinyl, pyrrolidinyl, piperazinyl, azetidinyl, aziridinyl, morpholinyl, thietanyl, oxetaryl. Preferred monocyclic diheterocycles include, but are not limited to 1-, 2-, 4-, or 5-imidazolyl, 1-, 3-, 4-, or 5-pyrazolyl, 3-, 4-, or 5-isothiazolyl, 3-, 4-, or 5-isoxazolyl, 1,3-, or 5-triazolyl, 1-, 2-, or 3-tetrazolyl, 2-pyrazinyl, 2-, 4-, or 5-pyrimidinyl, 1- or 2-piperazinyl, 2-, 3-, or 4-morpholinyl. Examples of suitable bicyclic and polyclic heteroaryl groups include but are not limited to include but are not limited to 1-, 2-, 3-, 5-, 6-, 7-, or 8-indolizinyl, 1-, 3-, 4-, 5-, 6-, or 7-isoindolyl, 2-, 3-, 4-, 5-,6-, or 7-indolyl, 2-, 3-, 4-, 5-, 6-, or 7-indazolyl, 2-, 4-, 5-, 6-, 7-, or 8-purinyl, 1-, 2-, 3-, 4-, 6-, 7-, 8-, or 9-quinolinzinyl, 2-, 3-, 4-, 5-, 6-, 7-, or 8-quinoliyl, 1-, 3-, 4-, 5-, 6-, 7-, or 8-isoquinoliyl, 1-, 4-, 5-, 6-, 7-, or 8-phthalazinyl, 2-, 3-, 4-, 5-, or 6-naphthyridinyl, 2-, 3-, 5-, 6-, 7-, or 8-quinazolinyl, 3-, 4-, 5-, 6-, 7-, or 8-cinnolinyl, 2-, 4-, 6-, or 7-pteridinyl, 1-, 2-, 3-, 4-, 5-, 6-, 7-, or 8-4aH carbazolyl, 1-, 2-, 3-, 4-, 5-, 6-, 7-, or 8-carbazolyl, 1-, 3-, 4-, 5-, 6-, 7-, 8-, or 9-carbolinyl, 1-, 2-, 3-, 4-, 6-, 7-, 8-, 9-, or 10-phenanthridinyl, 1-, 2-, 3-, 4-, 5-, 6-, 7-, 8-, or 9-acridinyl, 1-, 2-, 4-, 5-, 6-, 7-, 8-, or 9-perimidinyl, 2-, 3-, 4-, 5-, 6-, 8-, 9-, or 10-phenanthrolinyl, 1-, 2-, 3-, 4-, 6-, 7-, 8-, or 9-phenazinyl, 1-, 2-, 3-, 4-, 6-, 7-, 8-, 9-, or 10-phenothiazinyl, 1-, 2-, 3-, 4-, 6-, 7-, 8-, or 9-, or 10phenoxazinyl, 2-, 3-, 4-, 5-, 6-, or 1-, 3-, 4-, 5-, 6-, 7-, 8-, 9-, or 10-benzisoqinolinyl, 2-, 3-, 4-, or thieno[2,3-b]furanyl, 2-, 3-, 5-, 6-, 7-, 8-, 9-, 10-, or 11-7H-pyrazino[2,3-c]carbazolyl, 2-, 3-, 5-, 6-, or 7-2H-furo[3,2-b]-pyranyl, 2-, 3-, 4-, 5-, 7-, or 8-5H-pyrido[2,3-d]-o-oxazinyl, 1-, 3-, or 5-1H-pyrazolo[4,3-d]-oxazolyl, 2-, 4-, or 5-4H-imidazol[4,5-d]thiazolyl, 3-, 5-, or 8-pyrazino[2,3-d]pyridazinyl, 2-, 3-, 5-, or 6-imidazo[2,1-b]thiazolyl, 1-, 3-, 6-, 7-, 8-, or 9-furo[3, 4-c]cinnolinyl, 1-, 2-, 3-, 4-, 5-, 6-, 7-, 8-, 9-, 10, or 11-4H-pyrido[2,3-c]carbazolyl, 2-, 3-, 6-, or 7-imidazo[1,2-b][1,2,4]triazinyl, 7-benzo[b]thienyl, 2-, 4-, 5-, 6-, or 7-benzoxazolyl, 2-, 4-, 5-, 6-, or 7-benzimidazolyl, 2-, 4-, 5-, 6-, or 7-benzothiazolyl, 1-, 2-, 4-, 5-, 6-, 7-, 8-, or 9-benzoxapinyl, 2-, 4-, 5-, 6-, 7-, or 8-benzoxazinyl, 1-, 2-, 3-, 5-, 6-, 7-, 8-, 9-, 10-, or 11-1H-pyrrolo[1,2-b][2]benzazapinyl. Typical fused heteroaryl groups include, but are not limited to 2-, 3-, 4-, 5-, 6-, 7-, or 8-quinolinyl, 1-, 3-, 4-, 5-, 6-, 7-, or 8-isoquinolinyl, 2-, 3-, 4-, 5-, 6-, or 7-indolyl, 2-, 3-, 4-, 5-, 6-, or 7-benzo[b]thienyl, 2-, 4-, 5-, 6-, or 7-benzoxazolyl, 2-, 4-, 5-, 6-, or 7-benzimidazolyl, 2-, 4-, 5-, 6-, or 7-benzothiazolyl.
The term “hydrocarbon-heteroatom chain” as used herein refers to a hydrocarbon chain wherein one or more carbon atoms are replaced with a heteroatom. The hydrocarbon-heteroatom chain is optionally substituted with one or more substituents selected from lower alkyl, lower :alkoxy, lower thioalkoxy, —O(CH2)0-2CF3, halogen, nitro, cyano, ═O, ═S, —OH, —SH, —CF3, —CO2H, —CO2C1-C6alkyl, —NH2, —NHC1-C6alkyl, —CONR′R″, or —N(C1-C6alkyl)2 where R′ and R″ are independently alkyl, akenyl, alkynyl, aryl, or joined together to form a 4 to 7 member ring.
The term “heteroalkylene” as used herein, refers to an alkylene radical as defined above, that includes one or more heteroatoms such as oxygen, sulfur, or nitrogen (with valence completed by hydrogen or oxygen) in the carbon chain or terminating the carbon chain.
The terms “lower alkoxy” and “lower thioalkoxy” as used herein refers to O-alkyl or S-alkyl of from 1 to 6 carbon atoms as defined above for “lower alkyl.”
The term “cycloalkenyl” means a cycloalkyl group having one or more carbon-carbon double. Example includes cyclobutene, cyclopentene, cyclohexene, cycloheptene, cyclobutadiene, cyclopentadiene, and the like.
The symbol “” means a bond to a group wherein a 4 to 8 membered ring is formed. Typically this symbol will appear in pairs.
When a bond is represented by a line such as “- - - ” this is meant to represent that the bond may be absent or present provided that the resultant compound is stable and of satisfactory valency.
The term “patient” means all mammals including humans. Examples of patients include, humans, cows, dogs, cats, goats, sheep, pigs, and rabbits.
A “therapeutically effective amount” is an amount of a compound of the present invention that when administered to a patient ameliorates a symptom of dyslipidemia, non-insulin dependent diabetes mellitus, obesity, hyperglycemia, hypercholesteremia, hyperlipidemia, atherosclerosis, hypertriglyceridemia, hyperinsulinemia, glucose disorders associated with circulating glucocorticoids, growth hormone, catecholamines, glucagon, or parathyroid hormone. Additionally, a “therapeutically effective amount” is an amount of a compound of the present invention that when administered to a patient ameliorates a symptom of an eating disorder suppresses appetite, or modulates leptin levels.
The term “a pharmaceutically acceptable salt” refers to the relatively non-toxic, inorganic and organic base or acid addition salts of compounds of the present invention. These salts can be prepared in situ during the final isolation and purification of the compounds or by separately reacting the purified compound in its free form with a suitable organic or inorganic base or acid and isolating the salt thus formed. Representative salts include the hydrobromide, hydrochloride, sulfate, bisulfate, nitrate, acetate, oxalate, valerate, oleate, palmitate, stearate, laurate, borate, benzoate, lactate, phosphate, tosylate, citrate, maleate, fumarate, succinate, tartrate, naphthylate mesylate, glucoheptonate, lactobionate, and laurylsulphonate salts, and the like. These also include cations based on the alkali and alkaline earth metals, such as sodium, lithium, potassium, calcium, magnesium, and the like, as well as non-toxic ammonium, quaternary ammonium, and amine cations including, but not limited to ammonium, tetramethylammonium, tetraethylammonium, methylamine, dimethylamine, trimethylamine, triethylamine, ethylamine, and the like. (See, for example, Berge S. M., et al., “Pharmaceutical Salts,” J. Pharm. Sci., 1977;66:1-19, which is incorporated herein by reference.) The free base form may be regenerated by contacting the salt form with a base. While the free base may differ from the salt form in terms of physical properties, such as solubility, the salts are equivalent to their respective free bases for the purposes of the present invention.
Compounds of the present invention are described by Formula I:
and pharmaceutically acceptable salts thereof, wherein:
and pharmaceutically acceptable salts thereof, wherein:
X0 and X1 are independently absent, O, S, —CH2—, —CH2—CH2—, —CH═CH—, —CH≡CH—, —S(O)2—, or —S(O)—;
Ar1 and Ar2 are each independently unsubstituted or substituted aryl or heteroaryl, provided that Ar1 is not thiazolyl or oxazolyl;
is absent; or when present,
is a saturated or unsaturated hydrocarbon chain which is substituted or unsubstituted, wherein said chain has from 1 to 4 atoms so that
Ar1, X1, (CH2)r, and Ar2, together form a five to eight membered ring;
is absent; or when present,
is a saturated or unsaturated hydrocarbon chain which is substituted or unsubstituted, wherein said chain has from 1 to 4 atoms so that
Ar1, X1, (CH2)r, and Ar2, together form a five to eight membered ring;
R1 and R2 are selected from hydrogen, lower alkyl, lower alkoxy, haloalkyl, —O—(CH2)mCF3, halogen, nitro, cyano, —OH, —SH, —CF3, —S(O)palkyl, S(O)paryl, —(CH2)mOR5, —(CH2)mNR6R7, —COR5, CO2R5, or —NR6R7, or together with the atoms to which they are attached form a five to eight member ring;
R3 and R4 are selected from hydrogen, lower alkyl, lower alkoxy, haloalkyl, —O—(CH2)mCF3, halogen, nitro, cyano, —OH, —SH, —CF3, —S(O)palkyl, S(O)paryl, —(CH2)mOR5, —(CH2)mNR6R7, —COR5, —CO2H, —CO2R5, or —NR6R7;
provided that at least one of R1-R4 is H, lower alkyl, lower alkoxy, haloalkyl, —O—(CH2)mCF3, halogen, nitro, cyano, —OH, —SH, —CF3, —S(O)palkyl, S(O)paryl, —(CH2)mOR5, —(CH2)mNR6R7, or NR6R7;
R5 is hydrogen, alkyl, alkenyl, alkynyl, or aryl;
R6 and R are each independently hydrogen, alkyl, alkenyl, alkynyl, —COalkyl, —COaryl, cycloalkyl, —CO2alkyl, —CO2aryl, or R6 and R7 together with the atoms to which they are attached form a 4 to 7 membered ring having 1 to 3 heteroatoms;
m is 0 to 5;
p is 0, 1, or 2;
q is 0 to 6; and
r is 0 to 6.
In compounds of Formula I, R1, R2, R3, and R4 are preferably selected from hydrogen, alkyl, or alkoxy. More preferably, R2 and R3 are hydrogen; and R1 and R4 are alkyl or alkoxy. In a particularly preferred embodiment of Formula I, R2 and R3 are hydrogen; R1 is alkyl; and R4 is alkoxy. Preferred alkoxy include methoxy, ethoxy, isopropoxy, n-propoxy, t-butoxy, n-butoxy, or isobutoxy. Similarly, preferred alkyl include methyl, ethyl, isopropyl, n-propyl, t-butyl, n-butyl, or isobutyl. In a most preferred embodiment of Formula I, q is 1, Ar1 is phenyl, X1 is absent, r is 0, V1 is absent, and Ar2 is 4-trifluoromethylphenyl.
In compounds of Formula I,
is preferably (CH2)t wherein t is 1 to 4. Additionally,
is optionally substituted with at least one substituent, wherein the substituent include but are not limited to lower alkyl, lower alkoxy, lower thioalkoxy, —O(CH2)0-2CF3, halogen, nitro, cyano, ═O, ═S, —OH, —SH, —CF3, OCF3, —CO2H, —CO2C1-C6alkyl, —NH2, —NHC1-C6alkyl, —CONR′R″, or —N(C1-C6alkyl)2 where R′ and R″ are independently alkyl, akenyl, alkynyl, aryl, or joined together to form a 4 to 7 member ring.
is preferably (CH2)t wherein t is 1 to 4. Additionally,
is optionally substituted with at least one substituent, wherein the substituent include but are not limited to lower alkyl, lower alkoxy, lower thioalkoxy, —O(CH2)0-2CF3, halogen, nitro, cyano, ═O, ═S, —OH, —SH, —CF3, OCF3, —CO2H, —CO2C1-C6alkyl, —NH2, —NHC1-C6alkyl, —CONR′R″, or —N(C1-C6alkyl)2 where R′ and R″ are independently alkyl, akenyl, alkynyl, aryl, or joined together to form a 4 to 7 member ring.
In a preferred embodiment of Formula I, R1 and R2 are joined together to form a five to eight member ring having Formula II. Such a ring includes, for example, cycloalkyl, aryl, heterocycloalkyl, or a heteroaryl rings where each such ring is optionally substituted as described above.
and pharmaceutically acceptable salts thereof, wherein:
and pharmaceutically acceptable salts thereof, wherein:
X0 and X1 are independently absent, O, S, —CH2—, —CH2—CH2—, —CH═CH—, —CH≡CH—, —S(O)2—, or —S(O)—;
Ar1 and Ar2 are each independently unsubstituted or substituted aryl or heteroaryl, provided that Ar1 is not thiazolyl or oxazolyl;
is absent; or when present,
a saturated or unsaturated hydrocarbon chain which is substituted or unsubstituted, wherein said chain has from 1 to 4 atoms so that
Ar1, X1, (CH2)r, and Ar2, together form a five to eight membered ring;
is absent; or when present,
a saturated or unsaturated hydrocarbon chain which is substituted or unsubstituted, wherein said chain has from 1 to 4 atoms so that
Ar1, X1, (CH2)r, and Ar2, together form a five to eight membered ring;
R3 and R4 are selected from hydrogen, lower alkyl, lower alkoxy, haloalkyl, —O—(CH2)mCF3, halogen, nitro, cyano, —OH, —SH, —CF3, —S(O)palkyl, S(O)paryl, —(CH2)mOR5, —(CH2)mNR6R7, —COR5, —CO2H, —CO2R5, or —NR6R7;
R5 is hydrogen, alkyl, alkenyl, alkynyl, or aryl;
R6 and R7 are each independently hydrogen, alkyl, alkenyl, alkynyl, —COalkyl, —COaryl, cycloalkyl, —CO2alkyl, —CO2aryl, —SO2alkyl, —SO2aryl, or R6 and R7 together with the atoms to which they are attached form a 4 to 7 membered ring having 1 to 3 heteroatoms;
is a saturated or unsaturated, substituted or unsubstituted hydrocarbon chain or hydrocarbon-heteroatom chain having from 3 to 6 atoms wherein the carbon atom of position 2 is connected to the carbon atom of position 3 to form a five to eight member ring;
is a saturated or unsaturated, substituted or unsubstituted hydrocarbon chain or hydrocarbon-heteroatom chain having from 3 to 6 atoms wherein the carbon atom of position 2 is connected to the carbon atom of position 3 to form a five to eight member ring;
m is 0 to 5;
p is 0 to 2;
q is 0 to 6; and
r is 0 to 6.
Preferably,
is —CH2CH2CO—O—, —CH2—CH2—O—CO—, —CH2—CH2—CH2—CH2—, —HC═CH—HC═CH—, —N═CH—HC═CH—, —HC═N—HC═CH—, —HC═CH—N═CH—, —HC═CH—HC═N—, —CH2—CH2—CH2—, —CH2—CH2—O—CH2—, —CH2—O—CH2CH2—, —CH2—HC═CH—CH2—, —CH2—HC═CH—, —CH2CH2—N R4—CH2—, —COCH═CH—O—, —O—CH═CH—CO—, —CH═CH—NR4—, —NR4—CH═CH—, —CH═CH—CH2—, —CH2—CH2—NR4—, —NR4—CH2—CH2—, —O—CH2—CH2—, —CH2—CH2—O—, —CH2—CH2CO—, —CH2—CO—CH2—, —CO—CH2—CH2—, —CH2—CH2—CH2—CO—, —CO—CH2—CH2—CH2—, —CH2—CO—CH2—CH2—, —CH2—CH2—CO—CH2, —CH2—CH2—CH2—NR4—, —NR4—CH2—CH2—CH2—, —O—CH2—CH2—CH2—, —CH2—CH2—CH2—O—, —CO—NR4—CH2—CH2—, NR4CO—CH2—CH2—, —CH2—CH2NR4—CO—, or —CH2—CH2—CO—NR4—. It will be understood that the left-most atom of these groups in attached to the atom labeled “3” in Formula I and the right-most atom of these groups is attached to the atom label “2” in Formula I.
is —CH2CH2CO—O—, —CH2—CH2—O—CO—, —CH2—CH2—CH2—CH2—, —HC═CH—HC═CH—, —N═CH—HC═CH—, —HC═N—HC═CH—, —HC═CH—N═CH—, —HC═CH—HC═N—, —CH2—CH2—CH2—, —CH2—CH2—O—CH2—, —CH2—O—CH2CH2—, —CH2—HC═CH—CH2—, —CH2—HC═CH—, —CH2CH2—N R4—CH2—, —COCH═CH—O—, —O—CH═CH—CO—, —CH═CH—NR4—, —NR4—CH═CH—, —CH═CH—CH2—, —CH2—CH2—NR4—, —NR4—CH2—CH2—, —O—CH2—CH2—, —CH2—CH2—O—, —CH2—CH2CO—, —CH2—CO—CH2—, —CO—CH2—CH2—, —CH2—CH2—CH2—CO—, —CO—CH2—CH2—CH2—, —CH2—CO—CH2—CH2—, —CH2—CH2—CO—CH2, —CH2—CH2—CH2—NR4—, —NR4—CH2—CH2—CH2—, —O—CH2—CH2—CH2—, —CH2—CH2—CH2—O—, —CO—NR4—CH2—CH2—, NR4CO—CH2—CH2—, —CH2—CH2NR4—CO—, or —CH2—CH2—CO—NR4—. It will be understood that the left-most atom of these groups in attached to the atom labeled “3” in Formula I and the right-most atom of these groups is attached to the atom label “2” in Formula I.
In the present embodiment,
optionally substituted with 1 or more substituents selected from the group consisting of lower alkyl, lower alkoxy, lower thioalkoxy, —O(CH2)1-5CF3, halogen, nitro, cyano, ═O, ═S, —OH, —SH, —CF3, —CO2H, —CO2C1-C6alkyl, —N2, —NHC1-C6alkyl, —OCH2O—, and —N(C1-C6alkyl)2.
optionally substituted with 1 or more substituents selected from the group consisting of lower alkyl, lower alkoxy, lower thioalkoxy, —O(CH2)1-5CF3, halogen, nitro, cyano, ═O, ═S, —OH, —SH, —CF3, —CO2H, —CO2C1-C6alkyl, —N2, —NHC1-C6alkyl, —OCH2O—, and —N(C1-C6alkyl)2.
In compounds of Formula II, R3, and R4 are preferably selected from hydrogen, alkyl, or alkoxy. More preferably, R3 and R4 are hydrogen; and
is a saturated or unsaturated, unsubstituted hydrocarbon chain having from 3 to 6 atoms wherein the carbon atom of position 2 is connected to the carbon atom of position 3 to form a five to eight member ring such as a cyclopentyl or cyclohexyl ring. In a most preferred embodiment of Formula II, q is 1, Ar1 is phenyl, X1 is O, r is 1, V1 is absent, and Ar2 is 4-trifluoromethylphenyl.
is a saturated or unsaturated, unsubstituted hydrocarbon chain having from 3 to 6 atoms wherein the carbon atom of position 2 is connected to the carbon atom of position 3 to form a five to eight member ring such as a cyclopentyl or cyclohexyl ring. In a most preferred embodiment of Formula II, q is 1, Ar1 is phenyl, X1 is O, r is 1, V1 is absent, and Ar2 is 4-trifluoromethylphenyl.
Also in this embodiment,
is preferably (CH2)t wherein t is 1 to 4. Additionally,
is optionally substituted with at least one substituent, wherein the substituent include but are not limited to lower alkyl, lower alkoxy, lower thioalkoxy, —O(CH2)0-2CF3, halogen, nitro, cyano, ═O, ═S, —OH, —SH, —CF3, —OCF3, —CO2H, —CO2C1-C6alkyl, —NH2, —NHC1-C6alkyl, —CONR′R″, or —N(C1-C6alkyl)2 where R′ and R″ are independently alkyl, akenyl, alkynyl, aryl, or joined together to form a 4 to 7 member ring.
is preferably (CH2)t wherein t is 1 to 4. Additionally,
is optionally substituted with at least one substituent, wherein the substituent include but are not limited to lower alkyl, lower alkoxy, lower thioalkoxy, —O(CH2)0-2CF3, halogen, nitro, cyano, ═O, ═S, —OH, —SH, —CF3, —OCF3, —CO2H, —CO2C1-C6alkyl, —NH2, —NHC1-C6alkyl, —CONR′R″, or —N(C1-C6alkyl)2 where R′ and R″ are independently alkyl, akenyl, alkynyl, aryl, or joined together to form a 4 to 7 member ring.
Examples of compounds of Formula I and Formula II include
[4-(Biphenyl-4-ylmethylsulfanyl)-5-methoxy-2-methyl-phenoxy]-acetic acid;
[4-(Biphenyl-4-ylmethylsulfanyl)-2-methyl-phenoxy]-acetic acid;
[2-Methyl-4-(4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid;
[5-Methoxy-2-methyl-4-(4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid;
[5-Methoxy-2-methyl-4-(2′,4′,6′-trimethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid;
[4-(4′-Chloro-3′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-2-methyl-phenoxy]-acetic acid;
[4-(2′,4′-Dichloro-biphenyl-4-ylmethylsulfanyl)-5-methoxy-2-methyl-phenoxy]-acetic acid;
[4-(3′,4′-Dichloro-biphenyl-4-ylmethylsulfanyl)-5-methoxy-2-methyl-phenoxy]-acetic acid;
[5-Methoxy-2-methyl-4-(3′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid;
[4-(4′-Fluoro-biphenyl-4-ylmethylsulfanyl)-5-methoxy-2-methyl-phenoxy]-acetic acid;
[5-Methoxy-2-methyl-4-(3′-trifluoromethoxy-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid;
[7-(4′-Trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-indan-4-yloxy]-acetic acid;
[4-(4Benzyloxy-benzylsulfanyl)-2-methyl-phenoxy]-acetic acid;
{5-Methoxy-2-methyl-4-[4-(4-trifluoromethyl-benzyloxy)-benzylsulfanyl]-phenoxy}-acetic acid;
{2-Methyl-4-[4-(4-trifluoromethyl-benzyloxy)-benzylsulfanyl]-phenoxy}-acetic acid;
[5-Methoxy-2-methyl-4-(3′-trifluoromethoxy-biphenyl-3-ylmethylsulfanyl)-phenoxy]-acetic acid;
[4-(9H-Fluoren-2-ylmethylsulfanyl-2-methyl-phenoxy]-acetic acid;
{[5-Methoxy-2-methyl-4-[4-(5-trifluoromethyl-pyridin-2-yl)-benzylsulfanyl]-phenoxy}-acetic acid;
{5-Methoxy-2-methyl-4-[6-4-trifluoromethyl-phenyl)-pyridin-3-ylmethylsulfanyl]-phenoxy}-acetic acid;
[5-Chloro-2-methyl-4-(4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid;
[3-Methoxy-4-(4′-trifluoromethyl-biphenyl)-4-ylmethylsulfanyl)-phenoxy]-acetic acid;
{2-Methyl-4-[2-(4′-trifluoromethyl-biphenyl-4-yl)-ethylsulfanyl]-phenoxy}-acetic acid;
{5-Methoxy-2-methyl-4-[2-(4′-trifluoromethyl-biphenyl-4-yl)-ethylsulfanyl]-phenoxy}-acetic acid;
{2-Methyl-4-[2-(4′-trifluoromethyl-biphenyl-4-yl)-vinyl]-phenoxy}-acetic acid;
{2-Methyl-4-[2-(4′-trifluoromethyl-biphenyl-4-yl)ethyl]-phenoxy}-acetic acid;
{7-[4-(5-Trifluoromethyl-pyridin-2-yl)-benzylsulfanyl]-indan-4-yloxy}-acetic acid;
{5-Methyl-7-[4-(5-trifluoromethyl-pyridin-2-yl)-benzylsulfanyl]-indan-4-yloxy}-acetic acid;
[5-Methyl-7-(4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-indan-4-yloxy]-acetic acid;
(4-{4-[2-(3-Fluoro-phenyl)-vinyl]-benzylsulfanyl}-5-methoxy-2-methyl-phenoxy)-acetic acid;
{2-Methyl-4-[4-(5-trifluoromethyl-pyridin-2-yl)-benzylsulfanyl]-phenoxy}-acetic acid;
{4-[4-(2,4-Difluoro-benzyloxy)-benzylsulfanyl]-2-methyl-phenoxy}-acetic acid;
{4-[4-(2,4-Dichloro-benzyloxy)-benzylsulfanyl]-2-methyl-phenoxy}-acetic acid;
{4-[4-(4-Methoxy-benzyloxy)-benzylsulfanyl]-2-methyl-phenoxy}-acetic acid;
{4-[4-(4-tert-Butyl-benzyloxy)-benzylsulfanyl]-2-methyl-phenoxy}-acetic acid;
{2-Methyl-4-[4-(4-trifluoromethoxy-benzyloxy)-benzylsulfanyl]-phenoxy}-acetic acid;
{6-Methyl-8-[4-(5-trifluoromethyl-pyridine-2-yl)-benzylsulfanyl]-chroman-5-yloxy}-acetic acid;
{5-Chloro-2-methyl-4-[4-(5-trifluoromethyl-pyridin-2-yl)-benzylsulfanyl]-phenoxy}-acetic acid;
[5-hydroxy-2-methyl-4,4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid;
[5-Methoxy-2-methyl-4-(3-methyl-4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid;
{7-[4-(4-trifluoromethyl-benzyl)-benzylsulfanyl]-indan-4-yloxy}-acetic acid;
{4-[5-(4-Chloro-phenyl)-isoxazol-3-ylmethylsulfanyl]-5-methoxy-2-methyl-phenoxy}-acetic acid;
{2-Methyl-4-[5-(4-trifluoromethyl-phenyl)-isoxazol-3-ylmethylsulfanyl]-phenoxy}-acetic acid;
{5-Methoxy-2-methyl-4-[5-(4-trifluoromethyl-phenyl)-isoxazol-3-ylmethylsulfanyl]-phenoxy}-acetic acid;
{7-[5-(4-Trifluoromethyl-phenyl)-isoxazol-3-ylmethylsulfanyl]-indan-4-yloxy}-acetic acid;
{2-Methyl-4-[3-(4-trifluoromethyl-phenyl)-isoxazol-5-ylmethylsulfanyl]-phenoxy}-acetic acid;
{5-Methoxy-2-methyl-4-[3-(4-trifluoromethyl-phenyl)-isoxazol-5-ylmethylsulfanyl]-phenoxy}-acetic acid;
[2-Methyl-4-(4-phenoxy-benzylsulfanyl)-phenoxy]-acetic acid;
[7-(4′-Trifluoromethyl-biphenyl-3-ylmethylsulfanyl)-indan-4-yloxy]-acetic acid;
[5-Methoxy-2-methyl-4-(4′-trifluoromethyl-biphenyl-3-ylmethylsulfanyl)-phenoxy]-acetic acid;
[5-Methoxy-2-methyl-4-(4′-trifluoromethyl-biphenyl-4-ylmethanesulfonyl)-phenoxy]-acetic acid;
[5-Methoxy-2-methyl-4-(4′-trifluoromethyl-biphenyl-4-ylmethanesulfinyl)-phenoxy]-acetic acid;
[2-Propyl-4-(4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid;
{7-[3-(5-Trifluoromethyl-pyridin-2-yl)-benzylsulfanyl]-indan-4-yloxy}-acetic acid;
(5-Methoxy-2-methyl-4-{2-[1-(4-trifluoromethyl-benzyl)-1H-indol-3-yl]-ethylsulfanyl}-phenoxy)-acetic acid;
[7-(4′-Trifluoromethyl-biphenyl-2-ylmethylsulfanyl)-indan-4-yloxy]-acetic acid;
{5-Methoxy-2-methyl-4-[2-(4-trifluoromethyl-benzyloxy)-benzylsulfanyl]-phenoxy}-acetic acid;
{4-[4-(4-Fluoro-benzyloxy)-benzylsulfanyl]-2-methyl-phenoxy}-acetic acid;
{4-[4-(4-Chloro-benzyloxy)-benzylsulfanyl]-2-methyl-phenoxy}-acetic acid;
4-[4-(2,5-Dichloro-benzyloxy)-benzylsulfanyl]-2methyl-phenoxy]-acetic acid;
{2-Methyl-4-[4-(pyridine-2-ylmethoxy)-benzylsulfanyl]-phenoxy}-acetic acid;
{5-Chloro-2-methyl-4-[4-(4-trifluoromethyl-benzyloxy)-benzylsulfanyl]-phenoxy}-acetic acid;
{7-[4-(2,4-Dichloro-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid;
{7-[4-(4-Trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid;
{5-Methoxy-7-[4-(4-trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid;
{7-[4-(4-Fluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid;
{7-[4-(2,4-Difluoro-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid;
{7-[4-(4-tert-Butyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid;
{7-[4-(4-Methoxy-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid;
[7-(4-Benzyloxy-benzylsulfanyl)-indan-4-yloxy]-acetic acid;
{7-[4-(4-Chloro-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid;
{7-[4-(2,5-Dichloro-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid;
{7-[4-(3,4-Dichloro-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid;
{7-[4-(4-Chloro-3-trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid;
{7-[4-(4-Fluoro-3-trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid;
{7-[4-(4-Trifluoromethoxy-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid;
{7-[4-(4-Fluoro-2-trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid;
{7-[4-(3,5-Dichloro-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid;
{7-[4-Methoxy-3-(4-trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid;
{7-[3-(4-Trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid;
{7-[3-(4-Chloro-3-trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid;
{7-[2-(4-Trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid;
{7-[3,5-Dichloro-4-(4-trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid;
{8-[4-(4-Trifluoromethyl-benzyloxy)-benzylsulfanyl]-chroman-5-yloxy}-acetic acid;
{8-[3-(4-Trifluoromethyl-benzyloxy)-benzylsulfanyl]-chroman-5-yloxy}-acetic acid;
{8-[4-(2,5-Dichloro-benzyloxy)-benzylsulfanyl]-chroman-5-yloxy}-acetic acid;
{8-[4-(5-Trifluoromethyl-pyridine-2-yl)-benzylsulfanyl]-chroman-5-yloxy}-acetic acid;
{7-[5-(2-Chloro-phenyl)-isoxazol-3-ylmethylsulfanyl]-indan-4-yloxy}-acetic acid;
{7-[3-(2,6-Dichloro-phenyl)-5-methyl-isoxazol-4-ylmethylsulfanyl]-indan-4-yloxy}-acetic acid;
{7-[3-(4-Trifluoromethyl-phenyl)-isoxazol-5-ylmethylsulfanyl]-indan-4-yloxy}-acetic acid;
{7-[2-(4′-Trifluoromethyl-biphenyl-4-yl)-ethylsulfanyl]-indan-4-yloxy}-acetic acid;
{7-[2-(4′-Trifluoromethyl-biphenyl-4-yl)-ethyl]-indan-4-yloxy}-acetic acid;
{5-Methyl-7-[4-5-trifluoromethyl-pyridin-2-yl)-benzylsulfanyl]-2,3-dihydro-benzofuran-4yloxy}-acetic acid;
[8-(4′-Trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-chroman-5-yloxy]-acetic acid;
{8-[5-(4-Chloro-phenyl)-isoxazol-3-ylmethylsulfanyl]-chroman-5-yloxy}-acetic acid;
{4-[5-(4-Chloro-phenyl)-isoxazol-3-ylmethylsulfanyl]-2-methyl-phenoxy}-acetic acid;
{7-[5-(4-Chloro-phenyl)-isoxazol-3-ylmethylsulfanyl]-indan-4-yloxy}-acetic acid;
{7-[3-(4-Chloro-phenyl)-isoxazol-5-ylmethylsulfanyl]-indan-4-yloxy}-acetic acid;
{5-Chloro2-methyl-4-[5-(4-trifluoromethyl-phenyl)-isoxazol-3-ylmethylsulfanyl]-phenoxy}-acetic acid;
2-[2-butyl-4-({4-[4-(trifluoromethyl)phenyl]-phenyl}methylthio)phenoxy]acetic acid;
{6-methyl-8-[4-(4-trifluoromethyl-benzyloxy)-benzylsulfanyl]-chroman-5-yloxy}-acetic acid;
{4-[5-(4-trifluoromethyl-phenyl)-isoxazol-3-ylmethylsulfanyl]-5,6,7,8-tetrahydro-naphthalen-1-yloxy}-acetic acid;
(4-{2-Butyl-5-chloro-1-[4-(1-cyano-cyclopentyl)-benzyl]-1H-imidazol-4-ylmethylsulfanyl}-2-methyl-phenoxy)-acetic acid;
[4-(5-Biphenyl-4-yl-2-thiophen-2-yl-4,5-dihydro-oxazol-4-ylmethylsulfanyl)-5-methoxy-2-methyl-phenoxy]-acetic acid;
{4-[2-(4-Bromo-phenoxy)-ethylsulfanyl]-2,6-dimethyl-phenoxy}-acetic acid;
[4-(3-{2-[4(2-Diethylamino-ethoxy)-phenyl]-benzimidazol-1-yl}-propylsulfanyl)-5-methoxy-2-methyl-phenoxy]-acetic acid;
[4-(5-Biphenyl-2-thiophen-2-yl-4,5-dihydro-oxazol-4-ylmethylsulfanyl)-2-methyl-phenoxy]-acetic acid;
(4-{2-[3-(4-Fluoro-phenyl)-benzo[b]thiophen-7-yl]-ethylsulfanyl}-2-methyl-phenoxy)-acetic acid;
{2-Methyl-4-[2-(5-phenyl-naphthalen-1-yloxy) ethylsulfanyl]-phenoxy}-acetic acid;
[2-Methyl-4-(3-phenoxy-benzylsulfanyl)-phenoxy]-acetic acid;
[2,5-Dimethyl-4-(5-p-tolyl-1,3,4-oxadiazol-2-ylmethylsulfanyl)-phenoxy]-acetic acid;
[2-Methyl-4-(4-pyrazol-1-yl-benzylsulfanyl)-phenoxy]-acetic acid;
[2-Methyl-4-(5-methyl-3-phenyl-isoxazol-4-ylmethylsulfanyl)-phenoxy]-acetic acid;
[4-(Biphenyl-2-ylmethylsulfanyl)-2-methyl-phenoxy]-acetic acid;
{4-[5-(4-Chloro-phenyl)-isoxazol-3-ylmethylsulfanyl]-2-methyl-phenoxy}-acetic acid;
[2-Methyl-4-(5-p-tolyl-1,3,4-oxadiazol-2-ylmethylsulfanyl)-phenoxy]-acetic acid;
{4-[3-(4-Chloro-phenyl)-1,2,4-oxadiazol-5-ylmethylsulfanyl]-2-methyl-phenoxy}-acetic acid;
[2,5-Dimethyl-4-(4-pyrazol-1-yl-benzylsulfanyl)-phenoxy]-acetic acid;
[4-(Biphenyl-2-ylmethylsulfanyl)-2,5-dimethyl-phenoxy]-acetic acid;
[4-(4-Benzyloxy-benzylsulfanyl)-5,6,7,8-tetrahydro-naphthalen-1-yloxy]-acetic acid;
[4-(4-Benzyloxy-benzylsulfanyl)-2,6-dimethyl-phenoxy]-acetic acid;
[4-(4-Benzyloxy-benzylsulfanyl)-2,5-dimethyl-phenoxy]-acetic acid;
[4-(4-Benzyloxy-benzylsulfanyl)-2-methyl-phenoxy]-acetic acid;
[4-(4-Benzyloxy-benzylsulfanyl)-phenoxy]-acetic acid;
[4-(Biphenyl-4-ylmethylsulfanyl)-5,6,7,8-tetrahydro-naphthalen-1-yloxy]-acetic acid;
[4-(Biphenyl-4-ylmethylsulfanyl)-2,6-dimethyl-phenoxy]-acetic acid;
[4-(Biphenyl-4-ylmethylsulfanyl)-2,5-dimethyl-phenoxy]-acetic acid;
[4-(Biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid;
{4-[3-(2-Fluoro-phenoxy)-benzylsulfanyl]-2,6-dimethyl-phenoxy}-acetic acid;
[4-(2-{4-[2-(3-Chloro-4-cyclohexyl-phenyl)-ethyl]-piperazin-1-yl}-ethylsulfanyl)-2-methyl-phenoxy]-acetic acid;
[5-Methoxy-2-methyl-4-(2-{4-[2-(3-phenyl-benzofuran-7-yl)-ethyl]-piperazin-1-yl}-ethylsulfanyl)-phenoxy]-acetic acid;
{4-[2-(2,6-Diphenyl-piperidin-1-yl)-ethylsulfanyl]-5-methoxy-2-methyl-phenoxy}-acetic acid;
[2-Methyl-4-(2-{4-[2-(3-phenyl-benzofuran-7-yl)-ethyl]-piperazin-1-yl}-ethylsulfanyl)-phenoxy]-acetic acid;
{4-[2-(2,6-Diphenyl-piperidin-1yl)-ethylsulfanyl]-2-methyl-phenoxy}-acetic acid;
and pharmaceutically acceptable salts thereof.
A subset of exemplary compounds of Formula I and Formula II include
[4-(Biphenyl-4-ylmethylsulfanyl)-5-methoxy-2-methyl-phenoxy]-acetic acid;
[5-Methoxy-2-methyl-4-(4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid;
[4-(2′,4′-Dichloro-biphenyl-4-ylmethylsulfanyl)-5-methoxy-2-methyl-phenoxy]-acetic acid;
[5-Methoxy-2-methyl-4-(3′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid;
[4-(4′-Fluorobiphenyl-4-ylmethylsulfanyl)-5-methoxy-2-methyl-phenoxy]-acetic acid;
[7-(4′-Trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-indan-4-yloxy]-acetic acid;
{5-Methoxy-2-methyl-4-[4-(4-trifluoromethyl-benzyloxy)-benzylsulfanyl]-phenoxy}-acetic acid;
[5-Methoxy-2-methyl-4-(3′-trifluoromethoxy-biphenyl-3-ylmethylsulfanyl)-phenoxy]-acetic acid;
{{5-Methoxy-2-methyl-4-[4-(5-trifluoromethyl-pyridin-2-yl)-benzylsulfanyl]-phenoxy}-acetic acid;
{5-Methoxy-2-methyl-4-[6-(4-trifluoromethoxy-phenyl)-pyridin-3-ylmethylsulfanyl]-phenoxy}-acetic acid;
[3-Methoxy-4-(4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid;
{5-Methoxy-2-methyl-4-[2-(4′-trifluoromethyl-biphenyl-4-yl)-ethylsulfanyl]-phenoxy}-acetic acid;
(4-{4-[2-(3-Fluoro-phenyl)-vinyl]-benzylsulfanyl}-5-methoxy-2-methyl-phenoxy)-acetic acid;
[5-Methoxy-2-methyl-4-(3-methyl-4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid;
{4-[5-(4-Chloro-phenyl)-isoxazol-3-ylmethylsulfanyl]-5-methoxy-2-methyl-phenoxy}-acetic acid;
{5-Methoxy-2-methyl 4-[5-(4-trifluoromethyl-phenyl)-isoxazol-3-ylmethylsulfanyl]-phenoxy}-acetic acid;
{5-Methoxy-2-methyl-4-[3-(4-trifluoromethyl-phenyl)-isoxazol-5-ylmethylsulfanyl]-phenoxy}-acetic acid;
[5-Methoxy-2-methyl-4-(4′-trifluoromethyl-biphenyl-3-ylmethylsulfanyl)-phenoxy]-acetic acid;
{7-[4-(4-Trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid;
{5-Methyl-7-[4-(5-trifluoromethyl-pyridin-2-yl)-benzylsulfanyl]-2,3-dihydro-benzofuran-4-yloxy}-acetic acid;
and pharmaceutically acceptable salts thereof.
Certain of the compounds of the present invention possess one or more chiral centers and each center may exist in the R or S configuration. The present invention includes all diastereomeric, enantiomeric, and epimeric forms as well as the appropriate mixtures thereof. Stereoisomers may be obtained, if desired, by methods known in the art as, for example, the separation of stereoisomers by chiral chromatographic columns. Additionally, the compounds of the present invention may exist as geometric isomers. The present invention includes all cis, trans, syn, anti, entgegen (E), and zusammen (Z) isomers as well as the appropriate mixtures thereof
In some situations, compounds may exist as tautomers. All tautomers are included within Formulas I and II and are provided by this invention.
In addition, the compounds of the present invention can exist in unsolvated as well as solvated forms with pharmaceutically acceptable solvents such as water, ethanol, and the like. In general, the solvated forms are considered equivalent to the unsolvated forms for the purposes of the present invention.
The present invention includes all pharmaceutically acceptable, non-toxic esters of the compounds of Formulae I and II. Such esters include C1-C6 alkyl esters where the alkyl group is a straight or branched chain. Acceptable esters also include C5-C7 cycloalkyl esters as well as arylalkyl esters such as, but not limited to benzyl. C1-C4 esters are preferred. Esters of the compounds of the present invention may be prepared according to conventional methods.
The compounds of the present invention are suitable to be administered to a patient for the treatment, control, or prevention of hypercholesteremia, dyslipidemia, obesity, hyperglycemia, hypercholesteremia, atherosclerosis, hypertriglyceridemia, and hyperinsulinemia. The compounds of the present invention are also suitable to be administered to a patient for the suppression of appetite and modulation of leptin. Accordingly, the compounds may be administered to a patient alone or as part of a composition that contains other components such as excipients, diluents, and carriers, all of which are well-known in the art. The compositions can be administered to humans and/or animals either orally, rectally, parenterally (intravenously, intramuscularly, or subcutaneously), intracisternally, intravaginally, intraperitoneally, intravesically, locally (powders, ointments, or drops), or as a buccal or nasal spray.
Compositions suitable for parenteral injection may comprise physiologically acceptable sterile aqueous or nonaqueous solutions, dispersions, suspensions or emulsions, and sterile powders for reconstitution into sterile injectable solutions or dispersions. Examples of suitable aqueous and nonaqueous carriers, diluents, solvents or vehicles include water, ethanol, polyols (propyleneglycol, polyethyleneglycol, glycerol, and the like), suitable mixtures thereof, vegetable oils (such as olive oil), and injectable organic esters such as ethyl oleate. Proper fluidity can be maintained, for example, by the use of a coating such as lecithin, by the maintenance of the required particle size in the case of dispersions and by the use of surfactants.
These compositions may also contain adjuvants such as preserving, wetting, emulsifying, and dispensing agents. Prevention of the action of microorganisms can be ensured by various antibacterial and antifungal agents, for example, parabens, chlorobutanol, phenol, sorbic acid, and the like. It may also be desirable to include isotonic agents, for example sugars, sodium chloride, and the like. Prolonged absorption of the injectable pharmaceutical form can be brought about by the use of agents delaying absorption, for example, aluminum monostearate and gelatin.
Solid dosage forms for oral administration include capsules, tablets, pills, powders, and granules. In such solid dosage forms, the active compound is admixed with at least one inert customary excipient (or carrier) such as sodium citrate or dicalcium phosphate or (a) fillers or extenders, as for example, starches, lactose, sucrose, glucose, mannitol, and silicic acid; (b) binders, as for example, carboxymethylcellulose, alginates, gelatin, polyvinylpyrrolidone, sucrose, and acacia; (c) humectants, as for example, glycerol; (d) disintegrating agents, as for example, agar, calcium carbonate, potato or tapioca starch, alginic acid, certain complex silicates, and sodium carbonate; (e) solution retarders, as for example paraffin; (f) absorption accelerators, as for example, quaternary ammonium compounds; (g) wetting agents, as for example, cetyl alcohol and glycerol monostearate; (h) adsorbents, as for example, kaolin and bentonite; and (i) lubricants, as for example, talc, calcium stearate, magnesium stearate, solid polyethylene glycols, sodium lauryl sulfate, or mixtures thereof. In the case of capsules, tablets, and pills, the dosage forms may also comprise buffering agents.
Solid compositions of a similar type may also be employed as fillers in soft and hard-filled gelatin capsules using such excipients as lactose or milk sugar, as well as high molecular weight polyethyleneglycols, and the like.
Solid dosage forms such as tablets, dragees, capsules, pills, and granules can be prepared with coatings and shells, such as enteric coatings and others well-known in the art. They may contain opacifying agents, and can also be of such composition that they release the active compound or compounds in a certain part of the intestinal tract in a delayed manner. Examples of embedding compositions which can be used are polymeric substances and waxes. The active compounds can also be in micro-encapsulated form, if appropriate, with one or more of the above-mentioned excipients.
Liquid dosage forms for oral administration include pharmaceutically acceptable emulsions, solutions, suspensions, syrups, and elixirs. In addition to the active compounds, the liquid dosage forms may contain inert diluents commonly used in the art, such as water or other solvents, solubilizing agents and emulsifiers, as for example, ethyl alcohol, isopropyl alcohol, ethyl carbonate, ethyl acetate, benzyl alcohol, benzyl benzoate, propyleneglycol, 1,3-butyleneglycol, dimethylformamide, oils, in particular, cottonseed oil, groundnut oil, corn germ oil, olive oil, castor oil and sesame oil, glycerol, tetrahydrofurfuryl alcohol, polyethyleneglycols and fatty acid esters of sorbitan or mixtures of these substances, and the like.
Besides such inert diluents, the composition can also include adjuvants, such as wetting agents, emulsifying and suspending agents, sweetening, flavoring, and perfuming agents.
Suspensions, in addition to the active compounds, may contain suspending agents, as for example, ethoxylated isostearyl alcohols, polyoxyethylene sorbitol and sorbitan esters, microcrystalline cellulose, aluminum metahydroxide, bentonite, agar-agar and tragacanth, or mixtures of these substances, and the like.
Compositions for rectal administrations are preferably suppositories which can be prepared by mixing the compounds of the present invention with suitable non-irritating excipients or carriers such as cocoa butter, polyethyleneglycol, or a suppository wax, which are solid at ordinary temperatures, but liquid at body temperature and therefore, melt in the rectum, or vaginal cavity and release the active component.
Dosage forms for topical administration of a compound of this invention include ointments, powders, sprays, and inhalants. The active component is admixed under sterile conditions with a physiologically acceptable carrier and any preservatives, buffers, or propellants as may be required. Ophthalmic formulations, eye ointments, powders, and solutions are also contemplated as being within the scope of this invention.
The compounds of the present invention can be administered to a patient at dosage levels in the range of about 0.1 to about 2,000 mg per day. For a normal human adult having a body weight of about 70 kilograms, a dosage in the range of about 0.01 to about 10 mg per kilogram of body weight per day is preferable. However, the specific dosage used can vary. For example, the dosage can depend on a numbers of factors including the requirements of the patient, the severity of the condition being treated, and the pharmacological activity of the compound being used. The determination of optimum dosages for a particular patient is well-known to those skilled in the art.
The present invention contains compounds that can be synthesized in a number of ways familiar to one skilled in organic synthesis. The compounds outlined herein can be synthesized according to the methods described below, along with methods typically utilized by a synthetic chemist, and combinations or variations of those methods, which are generally known to one skilled in the art of synthetic chemistry. The synthetic route of compounds in the present invention is not limited to the methods outlined below. It is assumed one skilled in the art will be able to use the schemes outlined below to synthesize compounds claimed in this invention. Individual compounds may require manipulation of the conditions in order to accommodate various functional groups. A variety of protecting groups generally known to one skilled in the art may be required. Purification, if necessary, can be accomplished on a silica gel column eluted with the appropriate organic solvent system. Also, reverse phase HPLC or recrystallization may be employed.
Many of the compounds with Formulas I and II are preferably made by reacting:
in a solvent in the presence of a base such as cesium carbonate, with the aryl halide:
wherein:
in a solvent in the presence of a base such as cesium carbonate, with the aryl halide:
wherein:
n, R1, R2, R3, R4, X0, X1, V1, Ar1, and Ar2 are the same as defined above for Formula I;
R11 is a lower alkyl; and
X is a halogen.
The resulting ester is then preferably hydrolyzed to form the compounds of Formulas I and II. Specifically, compounds of Formulas I and II can be prepared using the synthetic route outlined in Scheme 1-6.
Scheme 1 covers the preparation of compounds of Formulas I and II wherein X0 is S, q is 0-3,
is absent, X1 is absent and r is 0. Compounds of the general formula A are thiocyanated with a mixture of bromine and sodium thiocyanate to give compounds of the general formula B. Compounds of the general formula B are then alkylated with an alkyl haloacarboxylate to give compounds of the general formula C. The preferred alkyl haloacarboxylate is methyl bromoacetate. Alternate routes to compounds of formula C will be readily apparent to a person skilled in the art of organic synthesis. Compounds of the general formula D are then prepared by reduction of C with dithiothreitol in methanol. Compounds of the general formula D are then alkylated with compounds of the general formula Y in the same manner as for B to give E. Compounds of the general formula Y are prepared as described in Scheme 7 (below) or are readily available from commercial sources. Compounds of the general formula E are then, saponified with LiOH in the THF to give the final compound F.
is absent, X1 is absent and r is 0. Compounds of the general formula A are thiocyanated with a mixture of bromine and sodium thiocyanate to give compounds of the general formula B. Compounds of the general formula B are then alkylated with an alkyl haloacarboxylate to give compounds of the general formula C. The preferred alkyl haloacarboxylate is methyl bromoacetate. Alternate routes to compounds of formula C will be readily apparent to a person skilled in the art of organic synthesis. Compounds of the general formula D are then prepared by reduction of C with dithiothreitol in methanol. Compounds of the general formula D are then alkylated with compounds of the general formula Y in the same manner as for B to give E. Compounds of the general formula Y are prepared as described in Scheme 7 (below) or are readily available from commercial sources. Compounds of the general formula E are then, saponified with LiOH in the THF to give the final compound F.
Scheme 2 covers the preparation of compounds of Formula I-II wherein X0 is O, q is 0-3, X1 is absent,
is absent, and r is 0. Compounds of the general formula A are alkylated with an alkyl haloacarboxylate to give compounds of the general formula G. The preferred alkyl halocarboxylate is methyl bromoacetate. Alternate routes to compounds of formula G when Y is O will be readily apparent to a person skilled in the art of organic synthesis. Compounds of the general formula G are then acylated using Friedel-Crafts conditions to give compounds of the general formula H which are then oxidized with m-chloroperoxybenzoic acid followed by hydrolysis to give phenolic compounds of the general formula I. Compounds of the general formula I are then reacted in a similar manner as for D to give after saponification with LiOH in THF, compounds of the general formula K.
is absent, and r is 0. Compounds of the general formula A are alkylated with an alkyl haloacarboxylate to give compounds of the general formula G. The preferred alkyl halocarboxylate is methyl bromoacetate. Alternate routes to compounds of formula G when Y is O will be readily apparent to a person skilled in the art of organic synthesis. Compounds of the general formula G are then acylated using Friedel-Crafts conditions to give compounds of the general formula H which are then oxidized with m-chloroperoxybenzoic acid followed by hydrolysis to give phenolic compounds of the general formula I. Compounds of the general formula I are then reacted in a similar manner as for D to give after saponification with LiOH in THF, compounds of the general formula K.
Scheme 3 covers the preparation of compounds of formula I-II wherein X0 is —CH2—CH2—, —C═C—, q is 0-3, X1 is absent,
is absent, and r is 0. Compounds of the general formula A are brominated with bromine, using acetic acid as solvent to give L. Alternatively, N-bromosuccinimide may be used in place of bromine and dichloromethane in place of acetic acid as solvent. Compounds of the general formula L are then alkylated with an alkyl haloacarboxylate to give compounds of the general formula M. The preferred alkyl halocarboxylate is methyl bromoacetate. Alternate routes to compounds of formula M will be readily apparent to a person skilled in the art of organic synthesis. Compounds of the general formula M are then reacted in the presence of tetrakis (triphenylphosphine)palladium(0) and biphenyl compounds of the general formula EE to give compounds of the general formula N. Compounds of the general formula EE are prepared as described in Scheme 9 or are readily available from commercial sources. Compounds of the general formula N are then saponified with LIOH in THF to give the final compound O.
is absent, and r is 0. Compounds of the general formula A are brominated with bromine, using acetic acid as solvent to give L. Alternatively, N-bromosuccinimide may be used in place of bromine and dichloromethane in place of acetic acid as solvent. Compounds of the general formula L are then alkylated with an alkyl haloacarboxylate to give compounds of the general formula M. The preferred alkyl halocarboxylate is methyl bromoacetate. Alternate routes to compounds of formula M will be readily apparent to a person skilled in the art of organic synthesis. Compounds of the general formula M are then reacted in the presence of tetrakis (triphenylphosphine)palladium(0) and biphenyl compounds of the general formula EE to give compounds of the general formula N. Compounds of the general formula EE are prepared as described in Scheme 9 or are readily available from commercial sources. Compounds of the general formula N are then saponified with LIOH in THF to give the final compound O.
Scheme 4 covers the preparation of compounds of formula I-II wherein X0 is —CH2—CH2—, q is 0-3, X1 is absent,
is absent, and r is 0. Accordingly, compounds of the general N can be reduced with hydrogen and palladium as catalyst to give compounds of the general formula P which are then saponified LiOH in THF to give the final compound Q.
is absent, and r is 0. Accordingly, compounds of the general N can be reduced with hydrogen and palladium as catalyst to give compounds of the general formula P which are then saponified LiOH in THF to give the final compound Q.
Scheme 5 covers the preparation of compounds of formula I-II wherein X0 is absent, q is 0, X1 is absent,
is absent, and r is 0 are prepared. Compounds of the general formula M are allowed to react with tetrakis (triphenylphosphine) palladium(0) and biphenyl compounds of the general formula HH to give compounds of the general formula R. Compounds of the general formula HH are prepared as described in Scheme 10 or are readily available from commercial sources. Compounds of the general formula R are then saponified with LiOH in THF to give the final compound S.
is absent, and r is 0 are prepared. Compounds of the general formula M are allowed to react with tetrakis (triphenylphosphine) palladium(0) and biphenyl compounds of the general formula HH to give compounds of the general formula R. Compounds of the general formula HH are prepared as described in Scheme 10 or are readily available from commercial sources. Compounds of the general formula R are then saponified with LiOH in THF to give the final compound S.
With reference to Scheme 6, compounds of formula I-II wherein X0 is S or O, q is 1-3, X1 is O, and r is 1-3 are prepared using the same conditions utilized for the preparation of K.
With reference to Scheme 7, compounds of the general formula X are prepared by reacting aryl boronic acid W with aryl bromide V in the presence of Pd(0) and cesium carbonate. Compounds of the general formula X are then reacted with methanesulfonyl chloride to give chlorides of the general formula Y.
With reference to Scheme 8, compounds of the general formula CC are prepared wherein an appropriate hydroxy benzyl alcohol AA is alkylated with an appropriate bromide Z. The resulting compound BB is then reacted with methanesulfonyl chloride to give chlorides of the general formula CC.
With reference to Scheme 9, compounds of the general formula EE are prepared by reacting aryl boronic acid V with aryl bromide DD in the presence of Pd(0) and cesium carbonate.
With reference to Scheme 10, compounds of the general formula HH are prepared by reacting aryl bromide V with boronic acid FF to give GG. Compounds of the general formula GG are then reacted with an alkyl lithium reagent and then quenched with a borate which is hydrolyzed to give compounds of the general formula HH.
With reference to Scheme 11, compounds of formula I-II wherein X0 is S or O, q is 1-3, X1 is absent, r is O, and V1 is a saturated or unsaturated hydrocarbon chain which is substituted or unsubstituted are prepared using the same conditions utilized for the preparation of K. Compounds of the general formula NN are prepared as described in Scheme 12 or are readily available from commercial sources.
With reference to Scheme 12, compounds of the general formula NN can be prepared by reacting an appropriately substituted aryl amine LL under Sandmeyer conditions followed by heating to give intermediate MM. The resulting intermediate MM is then reacted with methanesulfonyl chloride to give chlorides of the general formula NN.
Not all compounds of Formulas I-II falling into a given class may be compatible with some of the reaction conditions described. Such restrictions are readily apparent to those skilled in the art of organic synthesis, and alternative methods must then be used.
The following non-limiting descriptions also demonstrate methods for the synthesis of compounds of Formulae I and II.
Synthesis of [4-(Biphenyl-4 ylmethylsulfanyl)-5-methoxy-2-methyl-phenoxy]-acetic acid (compound 1)
Step 1. Preparation of 5-Methoxy-2-methyl-phenol (compound 1A)
2-Hydroxy-4-methoxy-benzaldehyde (3 g, 19.7 mmol), ammonium formate (6.2 g, 99 mmol) and palladium/carbon (900 mg @10%) were added to 26 ml glacial acetic acid and heated at 110° C. for 1 h. The reaction was cooled, filtered, and diluted with water (100 ml). The crude product was extracted with chloroform (3×50 ml), washed with water, brine, and dried over anhydrous sodium sulfate. The resulting solution was concentrated and used for the next step without further purification. MS m/z 139 (M+1).
Step 2. Preparation of 5-Methoxy-2-methyl-4-thiocyanato-phenol (compound 1B)
The product from Example 1A (3.5 g, 25 mmol), sodium thiocyanate (6.48 g, 80 mmol), and sodium bromide (2.6 g, 25 mmol) were dissolved in 30 ml anhydrous methanol. Bromine (4.4 g, 28 mmol) was added drop wise over 15 minutes and allowed to stir at ambient temperature for 1 h. Brine was added (50 ml) and the crude product was extracted into ethyl acetate (3×100 ml). The combined organic extracts were washed with brine, dried over anhydrous sodium sulfate, decanted, and concentrated to afford the title product in good purity. 400 MHz 1H NMR (DMSO-d6) δ 10.13 (s, 1H), 7.25 (s, 1H), 6.54 (s, 1H), 3.77 (s, 3H), 2.0 (s, 3H); MS m/z 196 (M+1).
Step 3. Preparation of (5-Methoxy-2-methyl-4-thiocyanato-phenoxy)-acetic acid methyl ester (compound 1C)
The product from Example 1B (620 mg, 3.2 mmol), methyl bromoacetate (854 mg, 3.5 mmol), and cesium carbonate (3.1 g, 9.6 mmol) were stirred in 10 ml anhydrous acetonitrile at ambient temperature for 1 h. The reaction was filtered through Celite®, concentrated, and purified using normal phase chromatography. 400 MHz 1H NMR (DMSO-d6) δ 7.33 (s, 1H), 6.72 (s, 1H), 4.93 (s, 2H), 3.84 (s, 3H), 3.66 (s, 3H), 2.09 (s, 3H); MS m/z 268 (M+1).
Step 4. Preparation of (4Mercapto-5-methoxy-2-methyl-phenoxy)-acetic acid methyl ester (compound 1D)
The product from Example 1C (1.1 g, 4.1 mmol) and dithiothreitol (824 mg, 5.4 mmol) were dissolved in 20 ml methanol with 2.5 ml water. The solution was refluxed for 4 h, cooled, concentrated, and purified by normal phase chromatography. 400 MHz 1H NMR (DMSO-d6) δ 7.02 (s, 1H), 6.54 (s, 1H), 4.79 (s, 2H), 4.41 (s, 1H), 3.72 (s, 3H), 3.64 (s, 3H), 2.02 (s, 3H); MS m/z 243 (M+1).
Step 5. Preparation of 4-Bromomethyl-biphenyl (compound 1E)
Biphenyl-4-yl methanol (500 mg, 2.72 mmol) phosphorus tribromide (809 mg, 2.99 mmol), and lithium bromide (260 mg, 2.99 mmol) were dissolved in 10 ml DMF and stirred at ambient temperature for 1 h. Water (10 ml) was added and the crude product was extracted into dichloromethane, dried over anhydrous sodium sulfate, filtered through silica gel, and concentrated. MS m/z 167 (M+1-Br).
Step 6. Preparation of [4-(Biphenyl-4-ylmethylsulfanyl)-5-methoxy-2-methyl-phenoxy]-acetic acid methyl ester (compound 1F)
The product from Example 1D (100 mg, 0.38 mmol), the product from Example 1E (92 mg, 0.38 mmol) and cesium carbonate (250 mg, 0.76 mmol) were added to 5 ml acetonitrile and stirred at ambient temperature for 4 hr. The reaction was filtered through Celite®, concentrated and purified through normal phase chromatography. MS m/z 409 (M+1).
Step 7. Preparation of [4-(Biphenyl-4-ylmethylsulfanyl)-5-methoxy-2-methyl-phenoxy]-acetic acid (compound 1)
The product from Example 1F (101 mg) was dissolved in 10 ml THF/water solution (10:1). Lithium hydroxide monohydrate (300 mg) was added and stirred for 30 minutes. 2 Normal aqueous HCl was added to pH<5 and then washed with ethyl acetate. The organic extract was dried over anhydrous sodium sulfate, decanted, and concentrated. The title product was recrystallized from chloroform/hexanes. mp 60-62° C.; 400 MHz 1H NMR (DMSO-d6) δ 12.94 (br(s), 1H), 7.58 (d, 2H, J=8 Hz), 7.52 (d, 2H, J=8 Hz), 7.39 (t, 2H, J=7.2 Hz), 7.30 (m, 3H), 7.02 (s, 1H), 6.52 (s, 1H), 4.69 (s, 2H), 4.01 (s, 2H), 3.73 (s, 3H), 2.01 (s, 3H). MS m/z 393 (M−1). Anal. Calc'd for C32H22O4S.3H2O C, 69.05; H, 5.07; found: C, 69.04; H, 5.35.
Step 1. Preparation of 5-Methoxy-2-methyl-phenol (compound 1A)
2-Hydroxy-4-methoxy-benzaldehyde (3 g, 19.7 mmol), ammonium formate (6.2 g, 99 mmol) and palladium/carbon (900 mg @10%) were added to 26 ml glacial acetic acid and heated at 110° C. for 1 h. The reaction was cooled, filtered, and diluted with water (100 ml). The crude product was extracted with chloroform (3×50 ml), washed with water, brine, and dried over anhydrous sodium sulfate. The resulting solution was concentrated and used for the next step without further purification. MS m/z 139 (M+1).
Step 2. Preparation of 5-Methoxy-2-methyl-4-thiocyanato-phenol (compound 1B)
The product from Example 1A (3.5 g, 25 mmol), sodium thiocyanate (6.48 g, 80 mmol), and sodium bromide (2.6 g, 25 mmol) were dissolved in 30 ml anhydrous methanol. Bromine (4.4 g, 28 mmol) was added drop wise over 15 minutes and allowed to stir at ambient temperature for 1 h. Brine was added (50 ml) and the crude product was extracted into ethyl acetate (3×100 ml). The combined organic extracts were washed with brine, dried over anhydrous sodium sulfate, decanted, and concentrated to afford the title product in good purity. 400 MHz 1H NMR (DMSO-d6) δ 10.13 (s, 1H), 7.25 (s, 1H), 6.54 (s, 1H), 3.77 (s, 3H), 2.0 (s, 3H); MS m/z 196 (M+1).
Step 3. Preparation of (5-Methoxy-2-methyl-4-thiocyanato-phenoxy)-acetic acid methyl ester (compound 1C)
The product from Example 1B (620 mg, 3.2 mmol), methyl bromoacetate (854 mg, 3.5 mmol), and cesium carbonate (3.1 g, 9.6 mmol) were stirred in 10 ml anhydrous acetonitrile at ambient temperature for 1 h. The reaction was filtered through Celite®, concentrated, and purified using normal phase chromatography. 400 MHz 1H NMR (DMSO-d6) δ 7.33 (s, 1H), 6.72 (s, 1H), 4.93 (s, 2H), 3.84 (s, 3H), 3.66 (s, 3H), 2.09 (s, 3H); MS m/z 268 (M+1).
Step 4. Preparation of (4Mercapto-5-methoxy-2-methyl-phenoxy)-acetic acid methyl ester (compound 1D)
The product from Example 1C (1.1 g, 4.1 mmol) and dithiothreitol (824 mg, 5.4 mmol) were dissolved in 20 ml methanol with 2.5 ml water. The solution was refluxed for 4 h, cooled, concentrated, and purified by normal phase chromatography. 400 MHz 1H NMR (DMSO-d6) δ 7.02 (s, 1H), 6.54 (s, 1H), 4.79 (s, 2H), 4.41 (s, 1H), 3.72 (s, 3H), 3.64 (s, 3H), 2.02 (s, 3H); MS m/z 243 (M+1).
Step 5. Preparation of 4-Bromomethyl-biphenyl (compound 1E)
Biphenyl-4-yl methanol (500 mg, 2.72 mmol) phosphorus tribromide (809 mg, 2.99 mmol), and lithium bromide (260 mg, 2.99 mmol) were dissolved in 10 ml DMF and stirred at ambient temperature for 1 h. Water (10 ml) was added and the crude product was extracted into dichloromethane, dried over anhydrous sodium sulfate, filtered through silica gel, and concentrated. MS m/z 167 (M+1-Br).
Step 6. Preparation of [4-(Biphenyl-4-ylmethylsulfanyl)-5-methoxy-2-methyl-phenoxy]-acetic acid methyl ester (compound 1F)
The product from Example 1D (100 mg, 0.38 mmol), the product from Example 1E (92 mg, 0.38 mmol) and cesium carbonate (250 mg, 0.76 mmol) were added to 5 ml acetonitrile and stirred at ambient temperature for 4 hr. The reaction was filtered through Celite®, concentrated and purified through normal phase chromatography. MS m/z 409 (M+1).
Step 7. Preparation of [4-(Biphenyl-4-ylmethylsulfanyl)-5-methoxy-2-methyl-phenoxy]-acetic acid (compound 1)
The product from Example 1F (101 mg) was dissolved in 10 ml THF/water solution (10:1). Lithium hydroxide monohydrate (300 mg) was added and stirred for 30 minutes. 2 Normal aqueous HCl was added to pH<5 and then washed with ethyl acetate. The organic extract was dried over anhydrous sodium sulfate, decanted, and concentrated. The title product was recrystallized from chloroform/hexanes. mp 60-62° C.; 400 MHz 1H NMR (DMSO-d6) δ 12.94 (br(s), 1H), 7.58 (d, 2H, J=8 Hz), 7.52 (d, 2H, J=8 Hz), 7.39 (t, 2H, J=7.2 Hz), 7.30 (m, 3H), 7.02 (s, 1H), 6.52 (s, 1H), 4.69 (s, 2H), 4.01 (s, 2H), 3.73 (s, 3H), 2.01 (s, 3H). MS m/z 393 (M−1). Anal. Calc'd for C32H22O4S.3H2O C, 69.05; H, 5.07; found: C, 69.04; H, 5.35.
Synthesis of [4-Biphenyl-4-ylmethylsulfanyl)-2-methyl-phenoxy]-acetic acid (compound 2)
Step 1. Preparation of 2-Methyl-4-thiocyanato-phenol (compound 2A)
The title compound was prepared in a manner analogous to Example 1B from 2-methylphenol. 400 MHz 1H NMR (DMSO-d6) δ 10.09 (s, 1H), 7.36 (s, 1H), 7.30 (d, 1H, J=8.1 Hz), 6.83 (d, 1H, J=8.1 Hz), 2.08 (s, 3H); MS m/z 166 (M+1).
Step 2. Preparation of (2-Methyl-4-thiocyanato-phenoxy)-acetic acid methyl ester (compound 2B)
The title compound was prepared from 2 Example 2A in a manner analogous to Example 1C. 400 MHz 1H NMR (DMSO-d6) δ 7.46 (s, 1H), 6.95 (d, 1H, J=8.5 Hz), 6.80 (d, 1H, J=8.5 Hz), 4.86 (s, 2H), 3.65 (s, 3H), 2.17 (s, 3H); MS m/z 238 (M+1).
Step 3. Preparation of (4-Mercapto-2-methyl-phenoxy)-acetic acid methyl ester (compound 2C)
The title compound was prepared from (2-methyl-4-thiocyanato-phenoxy)-acetic acid methyl ester in a manner analogous to Example 1C. 400 MHz 1H NMR (DMSO-d6) δ 7.05 (s, 1H), 7.00 (d, 1H, J=10.3 Hz), 6.70 (d, 1H, J=10.3 Hz), 5.00 (s, 1H), 4.73 (s, 1H), 3.63 (s, 3H), 2.09 (s, 3H); MS m/z 213 (M+1).
Step 4. Preparation of [4-(Biphenyl-4-ylmethylsulfanyl)-2-methyl-phenoxy]-acetic acid methyl ester (compound 2D)
The title compound was prepared in the manner analogous to Example 1F using 2C and 1E. MS m/z 379 (M+1).
Step 5. Preparation of [4-(Biphenyl-4ylmethylsulfanyl)-2-methyl-phenoxy]-acetic acid (compound 2)
The title compound was prepared in the manner analogous to Example 1. mp 138° C.; 400 MHz 1H NMR (DMSO-d6) δ 7.59 (d, 2H, J=9.5 Hz), 7.53 (d, 2H, J=9.5 Hz), 7.40 (m, 2H), 7.31 (m, 3H), 7.14 (d, 1H, J=1.7 Hz), 7.09 (d, 1H, J=10.7 Hz), 6.70 (d, 1H, J=10.7 Hz), 4.62 (s, 2H), 4.11 (s, 2H), 2.09 (s, 3H); MS m/z 363 (M−1).
Step 1. Preparation of 2-Methyl-4-thiocyanato-phenol (compound 2A)
The title compound was prepared in a manner analogous to Example 1B from 2-methylphenol. 400 MHz 1H NMR (DMSO-d6) δ 10.09 (s, 1H), 7.36 (s, 1H), 7.30 (d, 1H, J=8.1 Hz), 6.83 (d, 1H, J=8.1 Hz), 2.08 (s, 3H); MS m/z 166 (M+1).
Step 2. Preparation of (2-Methyl-4-thiocyanato-phenoxy)-acetic acid methyl ester (compound 2B)
The title compound was prepared from 2 Example 2A in a manner analogous to Example 1C. 400 MHz 1H NMR (DMSO-d6) δ 7.46 (s, 1H), 6.95 (d, 1H, J=8.5 Hz), 6.80 (d, 1H, J=8.5 Hz), 4.86 (s, 2H), 3.65 (s, 3H), 2.17 (s, 3H); MS m/z 238 (M+1).
Step 3. Preparation of (4-Mercapto-2-methyl-phenoxy)-acetic acid methyl ester (compound 2C)
The title compound was prepared from (2-methyl-4-thiocyanato-phenoxy)-acetic acid methyl ester in a manner analogous to Example 1C. 400 MHz 1H NMR (DMSO-d6) δ 7.05 (s, 1H), 7.00 (d, 1H, J=10.3 Hz), 6.70 (d, 1H, J=10.3 Hz), 5.00 (s, 1H), 4.73 (s, 1H), 3.63 (s, 3H), 2.09 (s, 3H); MS m/z 213 (M+1).
Step 4. Preparation of [4-(Biphenyl-4-ylmethylsulfanyl)-2-methyl-phenoxy]-acetic acid methyl ester (compound 2D)
The title compound was prepared in the manner analogous to Example 1F using 2C and 1E. MS m/z 379 (M+1).
Step 5. Preparation of [4-(Biphenyl-4ylmethylsulfanyl)-2-methyl-phenoxy]-acetic acid (compound 2)
The title compound was prepared in the manner analogous to Example 1. mp 138° C.; 400 MHz 1H NMR (DMSO-d6) δ 7.59 (d, 2H, J=9.5 Hz), 7.53 (d, 2H, J=9.5 Hz), 7.40 (m, 2H), 7.31 (m, 3H), 7.14 (d, 1H, J=1.7 Hz), 7.09 (d, 1H, J=10.7 Hz), 6.70 (d, 1H, J=10.7 Hz), 4.62 (s, 2H), 4.11 (s, 2H), 2.09 (s, 3H); MS m/z 363 (M−1).
Synthesis of [2-Methyl-4-(4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid (compound 3)
Step 1. Preparation of (4′-Trifluoromethyl-biphenyl-4-yl)-methanol (compound 3A)
1-Bromo-4-trifluoromethyl-benzene (814 mg, 3.62 mmol), 4-hydroxymethylphenylboronic acid (600 mg, 3.98 mmol), cesium carbonate (2.36 g, 7.24 mmol), and PdCl2(dppf) (132 mg, 0.181 mmol) were added to 10 ml of a 1:1 solution of DMF/THF. The reaction was flushed with nitrogen and heated to 90° C. for 1 h. The reaction was cooled, poured into diethyl ether and washed with water (2×50 ml), brine (1×50 ml) and dried over anhydrous sodium sulfate. The crude product was filtered through silica gel, eluted: with diethyl ether, and concentrated to provide the title compound. MS m/z 251 (M−1).
Step 2. Preparation of 4-Chloromethyl-4′-trifluoromethyl-biphenyl (compound 3B)
The product from Example 3A was dissolved in 10 ml methylene chloride. Triethylamine (468 mg, 4.62 mol) and methanesulfonyl chloride (422 mg, 3.68 mmol) were then added and stirred for 18 h. The reaction was poured into water and extracted with methylene chloride. The organic solution was dried over anhydrous sodium sulfate, decanted and concentrated to provide the title compound that was used without further purification. MS m/z 235 (M-Cl+1).
Step 3. Preparation of [2-Methyl-4(4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid methyl ester (compound 3C)
The title compound was prepared in the manner analogous to Example 1F using 3B and 2C. MS m/z 447 (M+1).
Step 4. Preparation of [2-Methyl-4-(4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid (compound 3)
The title compound was prepared in the manner analogous to Example 1 using 3C. mp 140-141° C.; 400 MHz 1H NMR (DMSO-d6) δ 12.95 (br(s), 1H), 7.82 (d, 2H, J=8.4 Hz), 7.74 (d, 2H, J=8.4 Hz), 7.61 (d, 2H, J=8.4 Hz), 7.35 (d, 2H, J=8.4 Hz), 7.10 (m, 2H), 6.70 (d, 1H, J=8.4 Hz), 4.62 (s, 2H), 4.13 (s, 2H), 2.08 (s, 3H); MS m/z 431 (M−1). Anal. Calc'd for C23H19F3O3S.0.7 H2O C, 62.07; H, 4.62; found: C, 61.98; H, 4.22.
Step 1. Preparation of (4′-Trifluoromethyl-biphenyl-4-yl)-methanol (compound 3A)
1-Bromo-4-trifluoromethyl-benzene (814 mg, 3.62 mmol), 4-hydroxymethylphenylboronic acid (600 mg, 3.98 mmol), cesium carbonate (2.36 g, 7.24 mmol), and PdCl2(dppf) (132 mg, 0.181 mmol) were added to 10 ml of a 1:1 solution of DMF/THF. The reaction was flushed with nitrogen and heated to 90° C. for 1 h. The reaction was cooled, poured into diethyl ether and washed with water (2×50 ml), brine (1×50 ml) and dried over anhydrous sodium sulfate. The crude product was filtered through silica gel, eluted: with diethyl ether, and concentrated to provide the title compound. MS m/z 251 (M−1).
Step 2. Preparation of 4-Chloromethyl-4′-trifluoromethyl-biphenyl (compound 3B)
The product from Example 3A was dissolved in 10 ml methylene chloride. Triethylamine (468 mg, 4.62 mol) and methanesulfonyl chloride (422 mg, 3.68 mmol) were then added and stirred for 18 h. The reaction was poured into water and extracted with methylene chloride. The organic solution was dried over anhydrous sodium sulfate, decanted and concentrated to provide the title compound that was used without further purification. MS m/z 235 (M-Cl+1).
Step 3. Preparation of [2-Methyl-4(4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid methyl ester (compound 3C)
The title compound was prepared in the manner analogous to Example 1F using 3B and 2C. MS m/z 447 (M+1).
Step 4. Preparation of [2-Methyl-4-(4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid (compound 3)
The title compound was prepared in the manner analogous to Example 1 using 3C. mp 140-141° C.; 400 MHz 1H NMR (DMSO-d6) δ 12.95 (br(s), 1H), 7.82 (d, 2H, J=8.4 Hz), 7.74 (d, 2H, J=8.4 Hz), 7.61 (d, 2H, J=8.4 Hz), 7.35 (d, 2H, J=8.4 Hz), 7.10 (m, 2H), 6.70 (d, 1H, J=8.4 Hz), 4.62 (s, 2H), 4.13 (s, 2H), 2.08 (s, 3H); MS m/z 431 (M−1). Anal. Calc'd for C23H19F3O3S.0.7 H2O C, 62.07; H, 4.62; found: C, 61.98; H, 4.22.
Synthesis of [5-Methoxy-2-methyl-4-(4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid (compound 4)
Step 1. Preparation of [5-Methoxy-2-methyl-4-(4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid methyl ester (compound 4A)
The title compound was prepared in the manner analogous to Example 1F using 1D and 3B. MS m/z 477 (M+1).
Step 2. Preparation of [5-Methoxy-2-methyl-4-(4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid (compound 4)
The title compound was prepared in the manner analogous to Example 1 using 4A. mp 170-171° C.; 400 MHz 1H NMR (DMSO-d6) δ 7.81 (d, 2H, J=8 Hz), 7.60 (d, 2H, J 8.4 Hz), 7.32 (d, 2H, J=8.4 Hz), 7.02 (s, 1H), 6.52 (s, 1H), 4.70 (s, 2H), 4.03 (s, 2H), 3.73 (s, 3H), 2.00 (s, 3H). MS m/z 463 (M+1). Anal. Calc'd for C24H21F3NO4S.0.1 H2O C, 62.09; H, 4.60; found: C, 62.00; H, 4.36.
Step 1. Preparation of [5-Methoxy-2-methyl-4-(4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid methyl ester (compound 4A)
The title compound was prepared in the manner analogous to Example 1F using 1D and 3B. MS m/z 477 (M+1).
Step 2. Preparation of [5-Methoxy-2-methyl-4-(4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid (compound 4)
The title compound was prepared in the manner analogous to Example 1 using 4A. mp 170-171° C.; 400 MHz 1H NMR (DMSO-d6) δ 7.81 (d, 2H, J=8 Hz), 7.60 (d, 2H, J 8.4 Hz), 7.32 (d, 2H, J=8.4 Hz), 7.02 (s, 1H), 6.52 (s, 1H), 4.70 (s, 2H), 4.03 (s, 2H), 3.73 (s, 3H), 2.00 (s, 3H). MS m/z 463 (M+1). Anal. Calc'd for C24H21F3NO4S.0.1 H2O C, 62.09; H, 4.60; found: C, 62.00; H, 4.36.
Synthesis of [5-Methoxy-2-methyl-4-(2′,4′,6′-trimethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid (compound 5)
Step 1. Preparation of [5-Methoxy-2-methyl-4-(2′,4′,6′-trimethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid methyl ester (compound 5A)
2-Bromo-1,3,5-trimethyl-benzene (396 mg, 2mmol), 4-hydroxymethylphenylboronic acid (334 mg, 2.2 mmol), cesium carbonate (1.3 g, 4 mmol), and PdCl2(dppf) (82 mg, 0.1 mmol) were added to 5 ml of a 1:1 solution of DMF/THF. The reaction was flushed with nitrogen and heated to 90° C. for 1 h. The reaction was cooled, poured into diethyl ether and washed with water (2×10 ml), brine (1×10 ml) and dried over anhydrous sodium sulfate. The organic solution was filtered through silica gel, eluted with diethyl ether, and concentrated. The crude product was dissolved in 10 ml dichloromethane. Added to this solution were triethylamine (202 mg, 2 mmol) then methanesulfonyl chloride (184 mg, 1.6 mmol) and allowed to stir at ambient temperature for 18 h. The reaction was poured into water and extracted with methylene chloride. The organic solution was dried over anhydrous sodium sulfate, decanted and concentrated to provide the crude alkyl chloride. The product from Example 1D (387 mg, 1.6 mmol), the crude 4′-chloromethyl-2,4,6-trimethyl-biphenyl, and cesium carbonate (1 g, 3.06 mmol) were stirred in 10 ml acetonitrile for 3 h, filtered, concentrated, and purified by normal phase chromatography to afford the title product. MS m/z 451 (M+1).
Step 2. Preparation of [5-Methoxy-2-methyl-4-(2′,4′,6′-trimethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid
The title compound was prepared in the manner analogous to Example 1 using 5A. mp 141° C.; 400 MHz 1H NMR (DMSO-d6) δ 12.81 (br(s), 1H), 7.04 (d, 2H, J=8.4 Hz), 6.77 (m, 3H), 6.69 (s, 2H), 6.36 (s, 1H), 4.43 (s, 2H), 3.82 (s, 2H), 3.58 (s, 3H), 2.03 (s, 3H), 1.82 (s, 3H), 1.67 (s, 6H). MS m/z 437 (M+1).
Step 1. Preparation of [5-Methoxy-2-methyl-4-(2′,4′,6′-trimethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid methyl ester (compound 5A)
2-Bromo-1,3,5-trimethyl-benzene (396 mg, 2mmol), 4-hydroxymethylphenylboronic acid (334 mg, 2.2 mmol), cesium carbonate (1.3 g, 4 mmol), and PdCl2(dppf) (82 mg, 0.1 mmol) were added to 5 ml of a 1:1 solution of DMF/THF. The reaction was flushed with nitrogen and heated to 90° C. for 1 h. The reaction was cooled, poured into diethyl ether and washed with water (2×10 ml), brine (1×10 ml) and dried over anhydrous sodium sulfate. The organic solution was filtered through silica gel, eluted with diethyl ether, and concentrated. The crude product was dissolved in 10 ml dichloromethane. Added to this solution were triethylamine (202 mg, 2 mmol) then methanesulfonyl chloride (184 mg, 1.6 mmol) and allowed to stir at ambient temperature for 18 h. The reaction was poured into water and extracted with methylene chloride. The organic solution was dried over anhydrous sodium sulfate, decanted and concentrated to provide the crude alkyl chloride. The product from Example 1D (387 mg, 1.6 mmol), the crude 4′-chloromethyl-2,4,6-trimethyl-biphenyl, and cesium carbonate (1 g, 3.06 mmol) were stirred in 10 ml acetonitrile for 3 h, filtered, concentrated, and purified by normal phase chromatography to afford the title product. MS m/z 451 (M+1).
Step 2. Preparation of [5-Methoxy-2-methyl-4-(2′,4′,6′-trimethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid
The title compound was prepared in the manner analogous to Example 1 using 5A. mp 141° C.; 400 MHz 1H NMR (DMSO-d6) δ 12.81 (br(s), 1H), 7.04 (d, 2H, J=8.4 Hz), 6.77 (m, 3H), 6.69 (s, 2H), 6.36 (s, 1H), 4.43 (s, 2H), 3.82 (s, 2H), 3.58 (s, 3H), 2.03 (s, 3H), 1.82 (s, 3H), 1.67 (s, 6H). MS m/z 437 (M+1).
Synthesis of [4-(4′-Chloro-3′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-2-methyl-phenoxy]-acetic acid (compound 6)
Step 1. Preparation of (4′-Chloro-3′-trifluoromethyl-biphenyl-4-yl)-methanol (compound 6A)
The title compound was prepared in the manner analogous to Example 3A using 4-bromo-1-chloro-2-trifluoromethyl-benzene. MS m/z 288 (M+1).
Step 1. Preparation of (4′-Chloro-3′-trifluoromethyl-biphenyl-4-yl)-methanol (compound 6A)
The title compound was prepared in the manner analogous to Example 3A using 4-bromo-1-chloro-2-trifluoromethyl-benzene. MS m/z 288 (M+1).
-
- Step 2. Preparation of 4-Chloro-4′-chloromethyl-3′-trifluoromethyl-biphenyl (compound 6B)
The title compound was prepared in the manner analogous to Example 3B using 6A. MS m/z 305 (M).
Step 3. Preparation of [4-(4′-Chloro-3′-trifluoromethyl-biphenyl-4ylmethylsulfanyl)-2-methyl-phenoxy]-acetic acid methyl ester (compound 6C)
The title compound was prepared in the manner analogous to Example 1F using 2C and 6B. MS m/z 481 (M+1).
Step 4. Preparation of [4-(4′-Chloro-3′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-2-methyl-phenoxy]-acetic acid (compound 6)
The title compound was prepared in the manner analogous to Example 1 using 6C. 400 MHz 1H NMR (DMSO-d6) δ 7.97 (s, 1H), 7.92 (d, 1H, J=10.5 Hz), 7.74 (d, 1H, J=10.5 Hz), 7.64 (d, 2H, J=8.9 Hz), 7.34 (d, 2H, J=8.9 Hz), 7.14 (s, 1H), 7.08 (d, 1H, J=11.0 Hz), 6.70 (d, 1H, J=11.0 Hz), 4.62 (s, 2H), 4.12 (s, 2H), 2.08 (s, 3H); MS m/z 465 (M−1).
- Step 2. Preparation of 4-Chloro-4′-chloromethyl-3′-trifluoromethyl-biphenyl (compound 6B)
Synthesis of [4-(2′,4′-Dichloro-biphenyl-4-ylmethylsulfanyl)-5-methoxy-2-methyl-phenoxyl-acetic acid (compound 7)
Step 1. Preparation of [4(2′,4′-Dichloro-biphenyl-4ylmethylsulfanyl)-5-methoxy-2-methyl-phenoxy]-acetic acid methyl ester (compound 7A)
The title compound was prepared in the manner analogous to Example 5A using 1D and 1-bromo-2,4-dichloro-benzene. MS m/z 479 (M+2).
Step 2. Preparation of [4-(2′,4′-Dichloro-biphenyl-4-ylmethylsulfanyl)-5-methoxy-2-methyl-phenoxy]-acetic acid (compound 7)
The title compound was prepared in the manner analogous to Example 1 using 7A. 400 MHz 1H NMR (DMSO-d6) δ 12.95 (br(s, 1H), 7.67 (dd, 1H, J′=2 Hz, J=8.4 Hz), 7.43 (d, 1H, J=2.4 Hz), 7.37 (s, 1H), 7.28 (s, 4H), 7.02 (s, 1H), 6.52 (s, 1H), 4.70 (s, 2H), 4.02 (s, 2H), 3.72 (s, 3H), 2.01 (s, 3H); MS m/z 465 (M+2).
Step 1. Preparation of [4(2′,4′-Dichloro-biphenyl-4ylmethylsulfanyl)-5-methoxy-2-methyl-phenoxy]-acetic acid methyl ester (compound 7A)
The title compound was prepared in the manner analogous to Example 5A using 1D and 1-bromo-2,4-dichloro-benzene. MS m/z 479 (M+2).
Step 2. Preparation of [4-(2′,4′-Dichloro-biphenyl-4-ylmethylsulfanyl)-5-methoxy-2-methyl-phenoxy]-acetic acid (compound 7)
The title compound was prepared in the manner analogous to Example 1 using 7A. 400 MHz 1H NMR (DMSO-d6) δ 12.95 (br(s, 1H), 7.67 (dd, 1H, J′=2 Hz, J=8.4 Hz), 7.43 (d, 1H, J=2.4 Hz), 7.37 (s, 1H), 7.28 (s, 4H), 7.02 (s, 1H), 6.52 (s, 1H), 4.70 (s, 2H), 4.02 (s, 2H), 3.72 (s, 3H), 2.01 (s, 3H); MS m/z 465 (M+2).
Synthesis of [4-(3′,4′-Dichloro-biphenyl-4-ylmethylsulfanyl)-5-methoxy-2-methyl-phenoxy]-acetic acid (compound 8)
Step 1. Preparation of [4-(3′,4′-Dichloro-biphenyl-4-ylmethylsulfanyl)-5-methoxy-2-methyl-phenoxy]-acetic acid methyl ester (compound 8A)
The title compound was prepared in the manner analogous to Example 5A using 1D and 4-bromo-1,2-dichlorobenzene. MS m/z 479 (M+2).
Step 2. Preparation of [4-(3′,4′-Dichloro-biphenyl-4-ylmethylsulfanyl)-5-methoxy-2-methyl-phenoxy]-acetic acid (compound 8)
The title compound was prepared in the manner analogous to Example 1 using 8A. mp 161-162° C.; 400 MHz 1H NMR (DMSO-d6) δ 7.87 (d, 1H, J=2 Hz), 7.57-7.66 (m, 4H), 7.29 (d, 2H, J=8.4 Hz), 7.01 (s, 1H), 6.52 (s, 1H), 4.69 (s, 2H), 4.02 (s, 2H), 3.72 (s, 3H), 2.03 (s, 3H); MS m/z 494 (M+1).
Step 1. Preparation of [4-(3′,4′-Dichloro-biphenyl-4-ylmethylsulfanyl)-5-methoxy-2-methyl-phenoxy]-acetic acid methyl ester (compound 8A)
The title compound was prepared in the manner analogous to Example 5A using 1D and 4-bromo-1,2-dichlorobenzene. MS m/z 479 (M+2).
Step 2. Preparation of [4-(3′,4′-Dichloro-biphenyl-4-ylmethylsulfanyl)-5-methoxy-2-methyl-phenoxy]-acetic acid (compound 8)
The title compound was prepared in the manner analogous to Example 1 using 8A. mp 161-162° C.; 400 MHz 1H NMR (DMSO-d6) δ 7.87 (d, 1H, J=2 Hz), 7.57-7.66 (m, 4H), 7.29 (d, 2H, J=8.4 Hz), 7.01 (s, 1H), 6.52 (s, 1H), 4.69 (s, 2H), 4.02 (s, 2H), 3.72 (s, 3H), 2.03 (s, 3H); MS m/z 494 (M+1).
Synthesis of [5-Methoxy-2-methyl-4-(3′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid (compound 9)
Step 1. Preparation of [5-Methoxy-2-methyl-4-(3′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid methyl ester (compound 9A)
The title compound was prepared in the manner analogous to Example 5A using 1D and 1-bromo-3-trifluoromethyl-benzene. MS m/z 477 (M+1).
Step 2. Preparation of [5-Methoxy-2-methyl-4-(3′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid (compound 9)
The title compound was prepared in the manner analogous to Example 1 using 9A. mp 138-139° C.; 400 MHz 1H NMR (DMSO-d6) δ 12.95 (br(s), 1H), 7.91 (m, 2), 7.63 (m, 4H), 7.32 (d, 2H, J=8.4 Hz), 7.02 (s, 1H), 6.52 (s, 1H), 4.69 (s, 2H), 4.03 (s, 2H), 3.73 (s, 3H), 2.06 (s, 3H); MS m/z 463 (M+1).
Step 1. Preparation of [5-Methoxy-2-methyl-4-(3′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid methyl ester (compound 9A)
The title compound was prepared in the manner analogous to Example 5A using 1D and 1-bromo-3-trifluoromethyl-benzene. MS m/z 477 (M+1).
Step 2. Preparation of [5-Methoxy-2-methyl-4-(3′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid (compound 9)
The title compound was prepared in the manner analogous to Example 1 using 9A. mp 138-139° C.; 400 MHz 1H NMR (DMSO-d6) δ 12.95 (br(s), 1H), 7.91 (m, 2), 7.63 (m, 4H), 7.32 (d, 2H, J=8.4 Hz), 7.02 (s, 1H), 6.52 (s, 1H), 4.69 (s, 2H), 4.03 (s, 2H), 3.73 (s, 3H), 2.06 (s, 3H); MS m/z 463 (M+1).
Synthesis of [4-(4′-Fluoro-biphenyl-4-ylmethylsulfanyl)-5-methoxy-2-methyl-phenoxy]-acetic acid (compound 10)
Step 1. Preparation of (4′-Fluoro-biphenyl-4-yl)-methanol (compound 10A)
The title compound was prepared in the manner analogous to Example 3A using 1-bromo-4-fluorobenzene. MS m/z 185 (M-H2O).
Step 2. Preparation of 4-Chloromethyl-4′-fluoro-biphenyl (compound 10B).
Step 1. Preparation of (4′-Fluoro-biphenyl-4-yl)-methanol (compound 10A)
The title compound was prepared in the manner analogous to Example 3A using 1-bromo-4-fluorobenzene. MS m/z 185 (M-H2O).
Step 2. Preparation of 4-Chloromethyl-4′-fluoro-biphenyl (compound 10B).
The title compound was prepared in the manner analogous to Example 3B using 10A. MS m/z 222 (M+2).
Step 3. Preparation of [4(4′-Fluoro-biphenyl-4-ylmethylsulfanyl)-5-methoxy-2-methyl-phenoxy]-acetic acid methyl ester (compound 10C)
The title compound was prepared in the manner analogous to Example 1F using 10B and 1D. MS m/z 427 (M+1).
Step 4. Preparation of [4-(4′-Fluoro-biphenyl-4-ylmethylsulfanyl)-5-methoxy-2-methyl-phenoxy]-acetic acid
The title compound was prepared in the manner analogous to Example 1 using 10C. 400 MHz 1H NMR (DMSO-d6) δ 12.93 (br(s), 1H), 7.62 (m, 2H), 7.49 (d, 2H, J=8.4 Hz), 7.27 (d, 2H, J=8.4 Hz), 7.22 (m, 2H), 7.02 (s, 1H), 6.52 (s, 1H), 4.69 (s, 2H), 4.01 (s, 2H), 3.73 (s, 3H), 2.01 (s, 3H). MS m/z 411 (M−1).
Synthesis of [5-Methoxy-2-methyl-4-(3′-trifluoromethoxy-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid (compound 11)
Step 1. Preparation of [5-Methoxy-2-methyl-4(3′-trifluoromethoxy-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid methyl ester (compound 11A)
The title compound was prepared in the manner analogous to Example 5A using 1D and 1-bromo-3-trifluoromethoxy-benzene. MS m/z 493 (M+1).
Step 2. Preparation of [5-Methoxy-2-methyl-4-(3′-trifluoromethoxy-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid (compound 11)
The title compound was prepared in the manner analogous to Example 1 using 11A. mp 137° C.; 400 m/z 1H NMR (DMSO-d6) δ 12.95 (br(s), 1H), 7.66 (d, 1H, J=8.8 Hz), 7.56 (m, 4H), 7.30 (m, 3H), 7.02 (s, 1H), 6.52 (s, 1H), 4.69 (s, 2H), 4.02 (s, 2H), 3.73 (s, 3H), 2.01 (s, 3H). MS m/z 493 (M+1).
Step 1. Preparation of [5-Methoxy-2-methyl-4(3′-trifluoromethoxy-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid methyl ester (compound 11A)
The title compound was prepared in the manner analogous to Example 5A using 1D and 1-bromo-3-trifluoromethoxy-benzene. MS m/z 493 (M+1).
Step 2. Preparation of [5-Methoxy-2-methyl-4-(3′-trifluoromethoxy-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid (compound 11)
The title compound was prepared in the manner analogous to Example 1 using 11A. mp 137° C.; 400 m/z 1H NMR (DMSO-d6) δ 12.95 (br(s), 1H), 7.66 (d, 1H, J=8.8 Hz), 7.56 (m, 4H), 7.30 (m, 3H), 7.02 (s, 1H), 6.52 (s, 1H), 4.69 (s, 2H), 4.02 (s, 2H), 3.73 (s, 3H), 2.01 (s, 3H). MS m/z 493 (M+1).
Synthesis of [7-(4′-Trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-indan-4-yloxy]-acetic acid (compound 12)
Step 1. Preparation of Indan-4-ol (compound 12A)
A mixture of 4-hydroxy-indan-1-one (5.0 g, 33.7 mmol), sodium cyanoborohydride (6.4 g, 101.1 mmol), and zinc iodide (32.3 g, 101.1 mmol) in dichloromethane, was heated at reflux for two hours. The reaction mixture was then filtered through 50 g SiO2 while still warm, eluting further with dichloroethane. The filtrate was collected and concentrated under vacuum. The residue was added to diethyl ether and the resulting white precipitate was filtered off. The filtrate was collected and concentrated in vacuo to give 4.2 g of the title, compound with purity high enough for subsequent use. 400 MHz 1H NMR (DMSO-d6) δ 9.06 (s, 1H), 6.86 (t, 1H, J=7.8 Hz), 6.59 (d, 1H, J=7.8 Hz), 6.48 (d, 1H, J=7.8 Hz), 2.75 (t, 2H, J=7.3 Hz), 2.67 (t, 2H, J=7.3 Hz), 1.92 (m, 2H).
Step 2. Preparation of 7-Thiocyanato-indan-4-ol (compound 12B)
The title compound was prepared in the manner analogous to Example 1B using 12A. MS m/z 192 (M+1).
Step 3. Preparation of (7-Mercapto-indan-4-yloxy)-acetic acid methyl ester (compound 12C)
7-Thiocyanato-indan-4ol (Example 12B) (1.47 g, 7.7 mmol), cesium carbonate (3.77 g, 11.6 mmol) and methyl bromoacetate (1.24 g, 8.08 mmol) were stirred in 20 ml acetonitrile at ambient temperature for 4 h. The reaction was filtered and concentrated. The crude product was treated under the conditions of Example 1D to afford the title product. MS m/z 239 (M+1).
Step 4. Preparation of [7-(4′-Trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-indan-4-yloxy]-acetic acid methyl ester (compound 12D)
The title compound was prepared in the manner analogous to Example 1F using 12C and 3B. MS m/z 473 (M+1).
Step 5. Preparation 17-(4′-Trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-indan-4-yloxy]-acetic acid (compound 12)
The title compound was prepared in the manner analogous to Example 1 using 12D. mp 158-159° C.; 400 MHz 1H NMR (DMSO-d6) δ 12.94 (br(s), 1H), 7.82 (d, 2H, J=8 Hz), 7.74 (d, 2H, J=8.8 Hz), 7.60 (d, 2H, J=6.4 Hz), 7.31 (d, 2H, J=8 Hz), 7.08 (d, 1H, J=8.8 Hz), 6.58 (d, 1H, J=8.4 Hz), 4.61 (s, 2H), 4.06 (s, 2H), 2.72 (m, 4H), 1.90 (q, 2H); MS m/z 457 (M−1).
Step 1. Preparation of Indan-4-ol (compound 12A)
A mixture of 4-hydroxy-indan-1-one (5.0 g, 33.7 mmol), sodium cyanoborohydride (6.4 g, 101.1 mmol), and zinc iodide (32.3 g, 101.1 mmol) in dichloromethane, was heated at reflux for two hours. The reaction mixture was then filtered through 50 g SiO2 while still warm, eluting further with dichloroethane. The filtrate was collected and concentrated under vacuum. The residue was added to diethyl ether and the resulting white precipitate was filtered off. The filtrate was collected and concentrated in vacuo to give 4.2 g of the title, compound with purity high enough for subsequent use. 400 MHz 1H NMR (DMSO-d6) δ 9.06 (s, 1H), 6.86 (t, 1H, J=7.8 Hz), 6.59 (d, 1H, J=7.8 Hz), 6.48 (d, 1H, J=7.8 Hz), 2.75 (t, 2H, J=7.3 Hz), 2.67 (t, 2H, J=7.3 Hz), 1.92 (m, 2H).
Step 2. Preparation of 7-Thiocyanato-indan-4-ol (compound 12B)
The title compound was prepared in the manner analogous to Example 1B using 12A. MS m/z 192 (M+1).
Step 3. Preparation of (7-Mercapto-indan-4-yloxy)-acetic acid methyl ester (compound 12C)
7-Thiocyanato-indan-4ol (Example 12B) (1.47 g, 7.7 mmol), cesium carbonate (3.77 g, 11.6 mmol) and methyl bromoacetate (1.24 g, 8.08 mmol) were stirred in 20 ml acetonitrile at ambient temperature for 4 h. The reaction was filtered and concentrated. The crude product was treated under the conditions of Example 1D to afford the title product. MS m/z 239 (M+1).
Step 4. Preparation of [7-(4′-Trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-indan-4-yloxy]-acetic acid methyl ester (compound 12D)
The title compound was prepared in the manner analogous to Example 1F using 12C and 3B. MS m/z 473 (M+1).
Step 5. Preparation 17-(4′-Trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-indan-4-yloxy]-acetic acid (compound 12)
The title compound was prepared in the manner analogous to Example 1 using 12D. mp 158-159° C.; 400 MHz 1H NMR (DMSO-d6) δ 12.94 (br(s), 1H), 7.82 (d, 2H, J=8 Hz), 7.74 (d, 2H, J=8.8 Hz), 7.60 (d, 2H, J=6.4 Hz), 7.31 (d, 2H, J=8 Hz), 7.08 (d, 1H, J=8.8 Hz), 6.58 (d, 1H, J=8.4 Hz), 4.61 (s, 2H), 4.06 (s, 2H), 2.72 (m, 4H), 1.90 (q, 2H); MS m/z 457 (M−1).
Synthesis of [4-(4-Benzyloxy-benzylsulfanyl)-2-methyl-phenoxy]-acetic acid (compound 13)
Step 1. Preparation of [4-(4-Benzyloxy-benzylsulfanyl)-2-methyl-phenoxy]-acetic acid methyl ester (compound 13A)
The title compound was prepared in the manner analogous to Example 1F using 1-chloromethyl-4-benzyloxy-benzene and 2C. MS m/z 409 (M+1).
Step 2. Preparation of [4-(4-Benzyloxy-benzylsulfanyl)-2-methyl-phenoxy]-acetic acid (compound 13)
The title compound was prepared in the manner analogous to Example 1 and 13A. mp 120-121° C.; 400 MHz 1H NMR (DMSO-d6) δ 7.39-7.25 (m, 5H), 7.15-7.03 (m, 4), 6.85 (d, 2H, J=8.5 Hz), 6.68 (d, 1H, J 8.4 Hz), 5.00 (s, 2H), 4.62 (s, 2H), 4.00 (s, 2H), 2.08 (s, 3H); MS m/z 395 (M+1).
Step 1. Preparation of [4-(4-Benzyloxy-benzylsulfanyl)-2-methyl-phenoxy]-acetic acid methyl ester (compound 13A)
The title compound was prepared in the manner analogous to Example 1F using 1-chloromethyl-4-benzyloxy-benzene and 2C. MS m/z 409 (M+1).
Step 2. Preparation of [4-(4-Benzyloxy-benzylsulfanyl)-2-methyl-phenoxy]-acetic acid (compound 13)
The title compound was prepared in the manner analogous to Example 1 and 13A. mp 120-121° C.; 400 MHz 1H NMR (DMSO-d6) δ 7.39-7.25 (m, 5H), 7.15-7.03 (m, 4), 6.85 (d, 2H, J=8.5 Hz), 6.68 (d, 1H, J 8.4 Hz), 5.00 (s, 2H), 4.62 (s, 2H), 4.00 (s, 2H), 2.08 (s, 3H); MS m/z 395 (M+1).
Synthesis of {5-Methoxy-2-methyl-4-[4-(4-trifluoromethyl-benzyloxy)-benzylsulfanyl]-phenoxy}-acetic acid (compound 14)
Step 1. Preparation of [4-(4-Trifluoromethyl-benzyloxy)-phenyl]-methanol (compound 14A)
4-Hydroxymethyl-phenol (1 g, 8.06 mmol), 1-Chloromethyl-4-trifluoromethyl-benzene (1.57 g, 8.06 mmol), and cesium carbonate (5.26 g, 16.12 mmol) were refluxed in acetonitrile for 20 h, cooled, filtered, and concentrated to give the title compound. MS m/z 265 (M-H2O+1).
Step 2. Preparation of 4-Chloromethyl-(4-trifluoromethyl-benzyloxy-benzene) (compound 14B)
The title compound was prepared in the manner analogous to Example 3B using 14A. MS m/z 265 (M-Cl+1).
Step 3. Preparation of {5-Methoxy-2-methyl-4-[4-(4-trifluoromethyl-benzyloxy)-benzylsulfanyl]-phenoxy}-acetic acid methyl ester (compound 14C)
The title compound was prepared in the manner analogous to Example 1F using 14B and 1D. MS m/z 507 (M+1).
Step 4. Preparation of {5-Methoxy-2-methyl-4-[4-(4-trifluoromethyl-benzyloxy)-benzylsulfanyl]-phenoxy}-acetic acid (compound 14)
The title compound was prepared in the manner analogous to Example 1 using 14C. mp 145° C. 400 MHz 1H NMR (DMSO-d6) δ 12.94 (br(s), 1H), 7.70 (d, 2H, J=8 Hz), 7.76 (d, 2H, J=8 Hz), 7.14 (d, 2H, J=8.8 Hz), 6.97 (s, 1H), 6.87 (m, 2H), 6.50 (s, 1H), 5.14 (s, 2H), 4.68 (s, 2H), 3.91 (s, 2H), 3.71 (s, 3H), 2.00 (s, 3H); MS m/z 491 (M−1).
Step 1. Preparation of [4-(4-Trifluoromethyl-benzyloxy)-phenyl]-methanol (compound 14A)
4-Hydroxymethyl-phenol (1 g, 8.06 mmol), 1-Chloromethyl-4-trifluoromethyl-benzene (1.57 g, 8.06 mmol), and cesium carbonate (5.26 g, 16.12 mmol) were refluxed in acetonitrile for 20 h, cooled, filtered, and concentrated to give the title compound. MS m/z 265 (M-H2O+1).
Step 2. Preparation of 4-Chloromethyl-(4-trifluoromethyl-benzyloxy-benzene) (compound 14B)
The title compound was prepared in the manner analogous to Example 3B using 14A. MS m/z 265 (M-Cl+1).
Step 3. Preparation of {5-Methoxy-2-methyl-4-[4-(4-trifluoromethyl-benzyloxy)-benzylsulfanyl]-phenoxy}-acetic acid methyl ester (compound 14C)
The title compound was prepared in the manner analogous to Example 1F using 14B and 1D. MS m/z 507 (M+1).
Step 4. Preparation of {5-Methoxy-2-methyl-4-[4-(4-trifluoromethyl-benzyloxy)-benzylsulfanyl]-phenoxy}-acetic acid (compound 14)
The title compound was prepared in the manner analogous to Example 1 using 14C. mp 145° C. 400 MHz 1H NMR (DMSO-d6) δ 12.94 (br(s), 1H), 7.70 (d, 2H, J=8 Hz), 7.76 (d, 2H, J=8 Hz), 7.14 (d, 2H, J=8.8 Hz), 6.97 (s, 1H), 6.87 (m, 2H), 6.50 (s, 1H), 5.14 (s, 2H), 4.68 (s, 2H), 3.91 (s, 2H), 3.71 (s, 3H), 2.00 (s, 3H); MS m/z 491 (M−1).
Synthesis of {2-Methyl-4-[4-(4-trifluoromethyl-benzyloxy)-benzylsulfanyl]-phenoxy}-acetic acid (compound 15)
Step 1. Preparation of {2-Methyl-4-[4-(4-trifluoromethyl-benzyloxy)-benzylsulfanyl]-phenoxy}-acetic acid methyl ester (compound 15A)
The title compound was prepared in the manner analogous to Example 1F using 2C and 14B. MS m/z 477 (M+1).
Step 2. Preparation of {2-Methyl-4-[4-(4-trifluoromethyl-benzyloxy)-benzylsulfanyl]-phenoxy}-acetic acid (compound 15)
The title compound was prepared in the manner analogous to Example 1 using 15A. mp 133° C.; 400 MHz 1H NMR (DMSO-d6) δ 7.70 (d, 2H, J=8 Hz), 7.76 (d, 2H, J=8 Hz), 7.14 (d, 2H, J=8.8 Hz), 7.09 (m, 1H), 7.04 (dd, 1H, J=2.4 Hz, J′=8.4 Hz), 6.87 (m, 2H), 6.68 (d, 1H, J=8.4 Hz), 5.14 (s, 2H), 4.61 (s, 2H), 3.99 (s, 2H), 2.08 (s, 3H); MS m/z 461 (M−1).
Step 1. Preparation of {2-Methyl-4-[4-(4-trifluoromethyl-benzyloxy)-benzylsulfanyl]-phenoxy}-acetic acid methyl ester (compound 15A)
The title compound was prepared in the manner analogous to Example 1F using 2C and 14B. MS m/z 477 (M+1).
Step 2. Preparation of {2-Methyl-4-[4-(4-trifluoromethyl-benzyloxy)-benzylsulfanyl]-phenoxy}-acetic acid (compound 15)
The title compound was prepared in the manner analogous to Example 1 using 15A. mp 133° C.; 400 MHz 1H NMR (DMSO-d6) δ 7.70 (d, 2H, J=8 Hz), 7.76 (d, 2H, J=8 Hz), 7.14 (d, 2H, J=8.8 Hz), 7.09 (m, 1H), 7.04 (dd, 1H, J=2.4 Hz, J′=8.4 Hz), 6.87 (m, 2H), 6.68 (d, 1H, J=8.4 Hz), 5.14 (s, 2H), 4.61 (s, 2H), 3.99 (s, 2H), 2.08 (s, 3H); MS m/z 461 (M−1).
Synthesis of [5-Methoxy-2-methyl-4-(3′-trifluoromethoxy-biphenyl-3-ylmethylsulfanyl)-phenoxy]-acetic acid (compound 16)
Step 1. Preparation of (3′-Trifluoromethoxy-biphenyl-3-yl)-methanol (compound 16A)
The title compound was prepared in the manner analogous to Example 3A using 3-hydroxymethylphenylboronic acid and 1-bromo-3-trifluoromethoxy-benzene. MS m/z 251 (M−1).
Step 2. Preparation of 3-Chloromethyl-3′-trifluoromethoxy-biphenyl (compound 16B)
The title compound was prepared in the manner analogous to Example 3B using 16A. MS m/z 251 (M−1).
Step 2. Preparation of [5-Methoxy-2-methyl-4-(3′-trifluoromethoxy-biphenyl-3-ylmethylsulfanyl)-phenoxy]-acetic acid methyl ester (compound 16C)
The title compound was prepared in the manner analogous to Example 1F using 16B and 1D. MS m/z 491 (M−1).
Step 4. Preparation of [5-Methoxy-2-methyl-4-(3′-trifluoromethoxy-biphenyl-3-ylmethylsulfanyl)-phenoxy]-acetic acid (compound 16)
The title compound was prepared in the manner analogous to Example 1 using 16C. mp 92-94° C.; 400 MHz 1H NMR (DMSO-d6) δ 12.96 (br(s), 1H), 7.55-7.24 (m, 8H), 7.01 (s, 1H), 6.52 (s, 1H), 4.68 (s, 2H), 4.03 (s, 2H), 3.72 (s, 3H), 1.99 (s, 3H); MS m/z 479 (M+1).
Step 1. Preparation of (3′-Trifluoromethoxy-biphenyl-3-yl)-methanol (compound 16A)
The title compound was prepared in the manner analogous to Example 3A using 3-hydroxymethylphenylboronic acid and 1-bromo-3-trifluoromethoxy-benzene. MS m/z 251 (M−1).
Step 2. Preparation of 3-Chloromethyl-3′-trifluoromethoxy-biphenyl (compound 16B)
The title compound was prepared in the manner analogous to Example 3B using 16A. MS m/z 251 (M−1).
Step 2. Preparation of [5-Methoxy-2-methyl-4-(3′-trifluoromethoxy-biphenyl-3-ylmethylsulfanyl)-phenoxy]-acetic acid methyl ester (compound 16C)
The title compound was prepared in the manner analogous to Example 1F using 16B and 1D. MS m/z 491 (M−1).
Step 4. Preparation of [5-Methoxy-2-methyl-4-(3′-trifluoromethoxy-biphenyl-3-ylmethylsulfanyl)-phenoxy]-acetic acid (compound 16)
The title compound was prepared in the manner analogous to Example 1 using 16C. mp 92-94° C.; 400 MHz 1H NMR (DMSO-d6) δ 12.96 (br(s), 1H), 7.55-7.24 (m, 8H), 7.01 (s, 1H), 6.52 (s, 1H), 4.68 (s, 2H), 4.03 (s, 2H), 3.72 (s, 3H), 1.99 (s, 3H); MS m/z 479 (M+1).
Synthesis of [4-(9H-Fluoren-2-ylmethylsulfanyl-2-methyl-phenoxy]-acetic acid (compound 17)
Step 1. Preparation of (9H-Fluoren-2-yl)-methanol (compound 17A)
9H-Fluorene-2-carbaldehyde (500 mg, 2.6 mmol) was dissolved in 10 ml methanol. Sodium borohydride (200 mg, 5.2 mmol) was added and allowed to stir at ambient temperature for 1 h. Water (10 ml) was added and the crude product was extracted into ethyl acetate (50 ml), washed with brine (50 ml), dried over anhydrous sodium sulfate, decanted and concentrated to afford the title product in good purity. MS m/z 195 (M+1).
Step 2. Preparation of 2-Chloromethyl-9H-fluorene (compound 17B)
The title compound was prepared in the manner analogous to Example 3B using 17A. MS m/z 179 (M-Cl+1).
Step 3. Preparation of [4-(9H-Fluoren-2-ylmethylsulfanyl-2-methyl-phenoxy]-acetic acid methyl ester (compound 17C)
The title compound was prepared in the manner analogous to Example 1F using 17B and 2C. MS m/z 422 (M+1).
Step 4. Preparation of [4-(9H-Fluoren-2-ylmethylsulfanyl-2-methyl-phenoxy]-acetic acid (compound 17)
The title compound was prepared in the manner analogous to Example 1 using 17C. 400 MHz 1H NMR (DMSO-d6) δ 12.93 (br(s), 1H), 7.80 (d, 1H, J=7.6 Hz), 7.23 (d, 1H, J=8 Hz), 7.5 (d, 1H, J=7.6 Hz), 7.43 (s, 1H), 7.31 (t, 1H, J′=6.4 Hz), 7.24 (m, 2H), 7.15 (m, 1H), 7.08 (dd, 1H, J=2.4 Hz, J=8.4 Hz), 6.7 (d, 1H, J=8.8 Hz), 4.62 (s, 2H), 4.13 (s, 2H), 3.82 (s, 2H), 2.09 (s, 3H). MS m/z 375 (M−1). Anal. Calc'd for C23H20O3S.0.3 H2O C, 72.34; H, 5.44; found: C, 72.46; H, 5.20.
Step 1. Preparation of (9H-Fluoren-2-yl)-methanol (compound 17A)
9H-Fluorene-2-carbaldehyde (500 mg, 2.6 mmol) was dissolved in 10 ml methanol. Sodium borohydride (200 mg, 5.2 mmol) was added and allowed to stir at ambient temperature for 1 h. Water (10 ml) was added and the crude product was extracted into ethyl acetate (50 ml), washed with brine (50 ml), dried over anhydrous sodium sulfate, decanted and concentrated to afford the title product in good purity. MS m/z 195 (M+1).
Step 2. Preparation of 2-Chloromethyl-9H-fluorene (compound 17B)
The title compound was prepared in the manner analogous to Example 3B using 17A. MS m/z 179 (M-Cl+1).
Step 3. Preparation of [4-(9H-Fluoren-2-ylmethylsulfanyl-2-methyl-phenoxy]-acetic acid methyl ester (compound 17C)
The title compound was prepared in the manner analogous to Example 1F using 17B and 2C. MS m/z 422 (M+1).
Step 4. Preparation of [4-(9H-Fluoren-2-ylmethylsulfanyl-2-methyl-phenoxy]-acetic acid (compound 17)
The title compound was prepared in the manner analogous to Example 1 using 17C. 400 MHz 1H NMR (DMSO-d6) δ 12.93 (br(s), 1H), 7.80 (d, 1H, J=7.6 Hz), 7.23 (d, 1H, J=8 Hz), 7.5 (d, 1H, J=7.6 Hz), 7.43 (s, 1H), 7.31 (t, 1H, J′=6.4 Hz), 7.24 (m, 2H), 7.15 (m, 1H), 7.08 (dd, 1H, J=2.4 Hz, J=8.4 Hz), 6.7 (d, 1H, J=8.8 Hz), 4.62 (s, 2H), 4.13 (s, 2H), 3.82 (s, 2H), 2.09 (s, 3H). MS m/z 375 (M−1). Anal. Calc'd for C23H20O3S.0.3 H2O C, 72.34; H, 5.44; found: C, 72.46; H, 5.20.
Synthesis of {{5-Methoxy-2-methyl-4-[4-(5-trifluoromethyl-pyridin-2-yl)-benzylsulfanyl]-phenoxy}-acetic acid (compound 18)
Preparation of [4-(5-Trifluoromethyl-pyridin-2-yl)-phenyl]-methanol (compound 18A)
The title compound was prepared from 2-Chloro-5-trifluoromethyl-pyridine and 4-(hydroxymethyl)boronic acid PdCl2(dppb) catalyst in the manner analogous to Example 3A. 400 MHz 1H NMR (DMSO-d6) δ 8.98 (s, 1H), 8.2 (dd, 1H, J=2.4 Hz, J′=8.4 Hz), 8.09 (m, 3H), 7.42 (d, 2H, J=8.54 Hz), 5.23 (t, 1H), 4.54 (d, 2H, J=6 Hz); MS m/z 254 (M+1).
Preparation of 2-(4-Chloromethyl-phenyl) 5-trifluoromethyl-pyridine (compound 18B)
The title compound was prepared in the manner analogous to Example 3B using 18A. MS m/z 272 (M+1).
Preparation of {5-Methoxy-2-methyl-4-[4-(5-trifluoromethyl-pyridin-2-yl)-benzylsulfanyl]-phenoxy}-acetic acid methyl ester (compound 18C)
The title compound was prepared in the manner analogous to Example 1F using 1D and 18B. MS m/z 478 (M+1).
Preparation of {5-Methoxy-2-methyl-4-[4-(5-trifluoromethyl-pyridin-2-yl)-benzylsulfanyl]-phenoxy}-acetic acid (compound 18)
The title compound was prepared from the product of Example 18C in the manner analogous to Example 1. mp 225° C. (dec.); 400 MHz 1H NMR (DMSO-d6) δ 8.97 (s, 1H), 8.2 (dd, 1H, J=2.4 Hz, J′=8.4 Hz), 8.13 (m, 1H), 8.01 (d, 2H, J=8.4 Hz), 7.34 (s, 2H, J=8.4 Hz), 6.95 (s, 1H), 6.42 (s, 1H), 4.23 (s, 2H), 4.01 (s, 2H), 3.69 (s, 3H), 1.96 (s, 3H) MS m/z 464 (M+1).
Preparation of [4-(5-Trifluoromethyl-pyridin-2-yl)-phenyl]-methanol (compound 18A)
The title compound was prepared from 2-Chloro-5-trifluoromethyl-pyridine and 4-(hydroxymethyl)boronic acid PdCl2(dppb) catalyst in the manner analogous to Example 3A. 400 MHz 1H NMR (DMSO-d6) δ 8.98 (s, 1H), 8.2 (dd, 1H, J=2.4 Hz, J′=8.4 Hz), 8.09 (m, 3H), 7.42 (d, 2H, J=8.54 Hz), 5.23 (t, 1H), 4.54 (d, 2H, J=6 Hz); MS m/z 254 (M+1).
Preparation of 2-(4-Chloromethyl-phenyl) 5-trifluoromethyl-pyridine (compound 18B)
The title compound was prepared in the manner analogous to Example 3B using 18A. MS m/z 272 (M+1).
Preparation of {5-Methoxy-2-methyl-4-[4-(5-trifluoromethyl-pyridin-2-yl)-benzylsulfanyl]-phenoxy}-acetic acid methyl ester (compound 18C)
The title compound was prepared in the manner analogous to Example 1F using 1D and 18B. MS m/z 478 (M+1).
Preparation of {5-Methoxy-2-methyl-4-[4-(5-trifluoromethyl-pyridin-2-yl)-benzylsulfanyl]-phenoxy}-acetic acid (compound 18)
The title compound was prepared from the product of Example 18C in the manner analogous to Example 1. mp 225° C. (dec.); 400 MHz 1H NMR (DMSO-d6) δ 8.97 (s, 1H), 8.2 (dd, 1H, J=2.4 Hz, J′=8.4 Hz), 8.13 (m, 1H), 8.01 (d, 2H, J=8.4 Hz), 7.34 (s, 2H, J=8.4 Hz), 6.95 (s, 1H), 6.42 (s, 1H), 4.23 (s, 2H), 4.01 (s, 2H), 3.69 (s, 3H), 1.96 (s, 3H) MS m/z 464 (M+1).
Synthesis of 45-Methoxy-2-methyl-4-[6-(4-trifluoromethylphenyl)-pyridin-3-ylmethylsulfanyl]-phenoxy}-acetic acid (compound 19)
Step 1. Preparation of [4-(6-Chloro-pyridin-3-ylmethylsulfanyl)-5-methoxy-2-methyl-phenoxy]-acetic acid methyl ester (compound 19A)
The title compound was prepared from 2-Chloro-5-chloromethyl-pyridine and 1D in a manner analogous to Example 1F. MS m/z 370 (M+2).
Step 2. Preparation of {5-Methoxy-2-methyl-4-[6-(4-trifluoromethyl-phenyl)-pyridin-3-ylmethylsulfanyl]-phenoxy}-acetic acid methyl ester (compound 19B)
The title compound was prepared from the product of Example 19A and 1-bromo-4-trifluoromethyl-benzene in a manner analogous to Example 3A. MS m/z 478 (M+1).
Step 3. Preparation of {5-Methoxy-2-methyl-4-[6-(4-trifluoromethyl-phenyl)-pyridin-3-ylmethylsulfanyl]-phenoxy}-acetic acid (compound 19)
The title compound was prepared from the product of Example 19B in the manner analogous to Example 1. mp 203° C. (dec.); 400 MHz 1H NMR (DMSO-d6) δ 8.34 (d, 1H, J=1.6 Hz), 8.21 (d, 2H, J=8 Hz), 7.92 (d, 1H, J=8.4 Hz), 7.77 (d, 2H, J=8.4 Hz), 7.65 (dd, 1H, J′=2.4 Hz, J=8.4 Hz), 6.93 (s, 1H), 6.39 (s, 1H), 4.15 (s, 2H), 4.00 (s, 2H), 3.66 (s, 3H), 1.95 (s, 3H). MS m/z 464 (M+1).
Step 1. Preparation of [4-(6-Chloro-pyridin-3-ylmethylsulfanyl)-5-methoxy-2-methyl-phenoxy]-acetic acid methyl ester (compound 19A)
The title compound was prepared from 2-Chloro-5-chloromethyl-pyridine and 1D in a manner analogous to Example 1F. MS m/z 370 (M+2).
Step 2. Preparation of {5-Methoxy-2-methyl-4-[6-(4-trifluoromethyl-phenyl)-pyridin-3-ylmethylsulfanyl]-phenoxy}-acetic acid methyl ester (compound 19B)
The title compound was prepared from the product of Example 19A and 1-bromo-4-trifluoromethyl-benzene in a manner analogous to Example 3A. MS m/z 478 (M+1).
Step 3. Preparation of {5-Methoxy-2-methyl-4-[6-(4-trifluoromethyl-phenyl)-pyridin-3-ylmethylsulfanyl]-phenoxy}-acetic acid (compound 19)
The title compound was prepared from the product of Example 19B in the manner analogous to Example 1. mp 203° C. (dec.); 400 MHz 1H NMR (DMSO-d6) δ 8.34 (d, 1H, J=1.6 Hz), 8.21 (d, 2H, J=8 Hz), 7.92 (d, 1H, J=8.4 Hz), 7.77 (d, 2H, J=8.4 Hz), 7.65 (dd, 1H, J′=2.4 Hz, J=8.4 Hz), 6.93 (s, 1H), 6.39 (s, 1H), 4.15 (s, 2H), 4.00 (s, 2H), 3.66 (s, 3H), 1.95 (s, 3H). MS m/z 464 (M+1).
Synthesis of [5-Chloro-2-methyl-4-(4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid (compound 20)
Step 1. Preparation of 5-Chloro-2-methyl-4-thiocyanato-phenol (compound 20A)
The title compound was prepared in a manner analogous to Example 1B from 5-chloro-2-methyl-phenol. 400 MHz 1H NMR (DMSO-d6) δ 10.60 (s, 1H), 7.54 (s, 1H), 6.98 (s, 1H), 2.07 (s, 3H).
Step 2. Preparation of (5-Chloro-2-methyl-4-thiocyanato-phenoxy)-acetic acid methyl ester(compound 20B)
The title compound was prepared from the product of Example 20A in a manner analogous to Example 1C. 400 MHz 1H NMR (DMSO-d6) δ 7.62 (s, 1H), 7.40 (s, 1H), 4.94 (s, 2H), 3.65 (s, 3H), 2.15 (s, 3H).
Step 3. Preparation of (5-Chloro-4-mercapto-2-methyl-phenoxy)-acetic acid methyl ester (compound 20C)
The title compound was prepared from (5-chloro-2-methyl-4-thiocyanato-phenoxy)-acetic acid methyl ester in a manner analogous to Example 1D. 400 MHz 1H NMR (DMSO-d6) δ 7.27 (s, 1H), 6.97 (s, 1H), 5.31 (s, 1H), 4.79 (s, 2H), 3.64 (s, 3H), 2.07 (s, 3H).
Step 4. Preparation of [5-Chloro-2-methyl-4-(4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)phenoxy]-acetic acid methyl ester (compound 20D)
The title compound was prepared in the manner analogous to Example 1F using 3B and 20C. 400 MHz 1H NMR (DMSO-d6) δ 7.82 (d, 2H, J=8.3 Hz), 7.74 (d, 2H, J=8.3 Hz), 7.62 (d, 2H, J=8.3 Hz), 7.39 (d, 2H, J=8.3 Hz), 7.27 (s, 1H), 7.00 (s, 1H), 4.82 (s, 2H), 4.20 (s, 2H), 3.64 (s, 3H), 2.09 (s, 3H); MS m/z 480 (M+).
Step 5. Preparation of [5-Chloro-2-methyl-4-(4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid (compound 20)
The title compound was prepared in the manner analogous to Example 1 using 20D. mp 161-162° C.; 400 MHz 1H NMR (DMSO-d6) δ 7.83 (d, 2H, J=8.3 Hz), 7.74 (d, 2H, J=8.3 Hz), 7.62 (d, 2H, J=8.3 Hz), 7.51 (d, 2H, J=8.3 Hz), 7.26 (s, 1H), 6.95 (s, 1H), 4.69 (s, 2H), 4.20 (s, 2H), 2.08 (s, 3H); MS m/z 467 (M+1).
Step 1. Preparation of 5-Chloro-2-methyl-4-thiocyanato-phenol (compound 20A)
The title compound was prepared in a manner analogous to Example 1B from 5-chloro-2-methyl-phenol. 400 MHz 1H NMR (DMSO-d6) δ 10.60 (s, 1H), 7.54 (s, 1H), 6.98 (s, 1H), 2.07 (s, 3H).
Step 2. Preparation of (5-Chloro-2-methyl-4-thiocyanato-phenoxy)-acetic acid methyl ester(compound 20B)
The title compound was prepared from the product of Example 20A in a manner analogous to Example 1C. 400 MHz 1H NMR (DMSO-d6) δ 7.62 (s, 1H), 7.40 (s, 1H), 4.94 (s, 2H), 3.65 (s, 3H), 2.15 (s, 3H).
Step 3. Preparation of (5-Chloro-4-mercapto-2-methyl-phenoxy)-acetic acid methyl ester (compound 20C)
The title compound was prepared from (5-chloro-2-methyl-4-thiocyanato-phenoxy)-acetic acid methyl ester in a manner analogous to Example 1D. 400 MHz 1H NMR (DMSO-d6) δ 7.27 (s, 1H), 6.97 (s, 1H), 5.31 (s, 1H), 4.79 (s, 2H), 3.64 (s, 3H), 2.07 (s, 3H).
Step 4. Preparation of [5-Chloro-2-methyl-4-(4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)phenoxy]-acetic acid methyl ester (compound 20D)
The title compound was prepared in the manner analogous to Example 1F using 3B and 20C. 400 MHz 1H NMR (DMSO-d6) δ 7.82 (d, 2H, J=8.3 Hz), 7.74 (d, 2H, J=8.3 Hz), 7.62 (d, 2H, J=8.3 Hz), 7.39 (d, 2H, J=8.3 Hz), 7.27 (s, 1H), 7.00 (s, 1H), 4.82 (s, 2H), 4.20 (s, 2H), 3.64 (s, 3H), 2.09 (s, 3H); MS m/z 480 (M+).
Step 5. Preparation of [5-Chloro-2-methyl-4-(4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid (compound 20)
The title compound was prepared in the manner analogous to Example 1 using 20D. mp 161-162° C.; 400 MHz 1H NMR (DMSO-d6) δ 7.83 (d, 2H, J=8.3 Hz), 7.74 (d, 2H, J=8.3 Hz), 7.62 (d, 2H, J=8.3 Hz), 7.51 (d, 2H, J=8.3 Hz), 7.26 (s, 1H), 6.95 (s, 1H), 4.69 (s, 2H), 4.20 (s, 2H), 2.08 (s, 3H); MS m/z 467 (M+1).
Synthesis of [3-Methoxy-4-(4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid (compound 21)
Step 1. Preparation of 3-Methoxy-4-thiocyanato-phenol (compound 21A)
The title compound was prepared in a manner analogous to Example 1B from 3-methoxy-phenol. 400 MHz 1H NMR (DMSO-d6) δ 10.21 (s, 1H), 7.34 (d, 1H, J=8.3 Hz), 6.53 (s, 1H), 6.43 (d, 1H, J=8.3 Hz), 3.81 (s, 3H).
Step 2. Preparation of (3-Methoxy-4-thiocyanato-phenoxy)-acetic acid methyl ester (compound 21B)
The title compound was prepared from the product of Example 21A in a manner analogous to Example 1C. MS m/z 227 (M-CN).
Step 4. Preparation of (4-Mercapto-3-methoxy-phenoxy)-acetic acid methyl ester (compound 21C)
The title compound was prepared from (3-Methoxy-4-thiocyanato-phenoxy)-acetic acid methyl ester in a manner analogous to Example 1D. 400 MHz 1H NMR (DMSO-d6) δ 7.13 (d, 1H, J=8.5 Hz), 6.56 (s, 1H), 6.39 (d, 1H, J=8.5 Hz), 4.72 (s, 2H), 4.51 (s, 1H), 3.75 (s, 3H), 3.64 (s, 3H).
Step 5. Preparation of [3-Methoxy-4-(4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid methyl ester (compound 21D)
The title compound was prepared in the manner analogous to Example 1F using 3B and 21C. 400 MHz 1H NMR (DMSO-d6) δ 7.81 (d, 2H, J=8.3 Hz), 7.53 (d, 2H, J=8.3 Hz), 7.59 (d, 2H, J=8.3 Hz), 7.32 (d, 2H, J=8.3 Hz), 7.11 (d, 1H, J=8.5 Hz), 6.56 (s, 1H), 6.37 (d, 1H, J=8.5 Hz), 4.73 (s, 2H), 4.04 (s, 2H), 3.76 (s, 3H), 3.63 (s, 3H); MS m/z 463 (M+1).
Step 6. Preparation of [3-Methoxy-4-(4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid (compound 21)
The title compound was prepared in the manner analogous to Example 1 using 21D. 400 MHz 1H NMR (DMSO-d6) δ 7.83 (d, 2H, J=8.3 Hz), 7.76 (d, 2H, J=8.3 Hz), 7.61 (d, 2H, J=8.3 Hz), 7.34 (d, 2H, J=8.3 Hz), 7.13 (d, 1H, J=8.5 Hz), 6.56 (s, 1H), 6.39 (d, 1H, J=8.5 Hz), 4.63 (s, 2H), 4.06 (s, 2H), 3.77 (s, 3H); MS m/z 449 (M+1). Anal. Calc'd for C23H19F3O4S, C, 61.60; H, 4.27; found: C, 61.35; H, 4.25.
Step 1. Preparation of 3-Methoxy-4-thiocyanato-phenol (compound 21A)
The title compound was prepared in a manner analogous to Example 1B from 3-methoxy-phenol. 400 MHz 1H NMR (DMSO-d6) δ 10.21 (s, 1H), 7.34 (d, 1H, J=8.3 Hz), 6.53 (s, 1H), 6.43 (d, 1H, J=8.3 Hz), 3.81 (s, 3H).
Step 2. Preparation of (3-Methoxy-4-thiocyanato-phenoxy)-acetic acid methyl ester (compound 21B)
The title compound was prepared from the product of Example 21A in a manner analogous to Example 1C. MS m/z 227 (M-CN).
Step 4. Preparation of (4-Mercapto-3-methoxy-phenoxy)-acetic acid methyl ester (compound 21C)
The title compound was prepared from (3-Methoxy-4-thiocyanato-phenoxy)-acetic acid methyl ester in a manner analogous to Example 1D. 400 MHz 1H NMR (DMSO-d6) δ 7.13 (d, 1H, J=8.5 Hz), 6.56 (s, 1H), 6.39 (d, 1H, J=8.5 Hz), 4.72 (s, 2H), 4.51 (s, 1H), 3.75 (s, 3H), 3.64 (s, 3H).
Step 5. Preparation of [3-Methoxy-4-(4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid methyl ester (compound 21D)
The title compound was prepared in the manner analogous to Example 1F using 3B and 21C. 400 MHz 1H NMR (DMSO-d6) δ 7.81 (d, 2H, J=8.3 Hz), 7.53 (d, 2H, J=8.3 Hz), 7.59 (d, 2H, J=8.3 Hz), 7.32 (d, 2H, J=8.3 Hz), 7.11 (d, 1H, J=8.5 Hz), 6.56 (s, 1H), 6.37 (d, 1H, J=8.5 Hz), 4.73 (s, 2H), 4.04 (s, 2H), 3.76 (s, 3H), 3.63 (s, 3H); MS m/z 463 (M+1).
Step 6. Preparation of [3-Methoxy-4-(4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid (compound 21)
The title compound was prepared in the manner analogous to Example 1 using 21D. 400 MHz 1H NMR (DMSO-d6) δ 7.83 (d, 2H, J=8.3 Hz), 7.76 (d, 2H, J=8.3 Hz), 7.61 (d, 2H, J=8.3 Hz), 7.34 (d, 2H, J=8.3 Hz), 7.13 (d, 1H, J=8.5 Hz), 6.56 (s, 1H), 6.39 (d, 1H, J=8.5 Hz), 4.63 (s, 2H), 4.06 (s, 2H), 3.77 (s, 3H); MS m/z 449 (M+1). Anal. Calc'd for C23H19F3O4S, C, 61.60; H, 4.27; found: C, 61.35; H, 4.25.
Synthesis of {2-Methyl-4-[2-(4′-trifluoromethyl-biphenyl-4-yl)-ethylsulfanyl]-phenoxy}-acetic acid (compound 22)
Step 1. Preparation of 2-(4′-Trifluoromethyl-biphenyl-4-yl)-ethanol (compound 22A)
A mixture of 2-(4-bromo-phenyl)-ethanol (2.3 ml, 3.3 g, 16.4 mmol), 4-trifluoromethylphenylboronic acid (5.0 g, 26.3 mmol), 1.0 M aqueous sodium carbonate solution (44.0 ml), and tetrakis(triphenylphosphine)palladium (0.98 g, 0.85 mmol) in 180 ml of ethanol and 180 ml of toluene was heated at reflux for 4 h. The cooled reaction mixture was diluted with 500 ml of ethyl acetate and filtered through a bed of Celite filter-aid. The filtrate was washed with 5% aqueous sodium carbonate solution (2×750 ml) and brine (3×750 ml), then dried over anhydrous sodium sulfate and concentrated. The crude product was purified by normal phase chromatography. MS m/z 266 (M).
Step 2. Preparation of 4-(2-Bromo-ethyl)-4′-trifluoromethyl-biphenyl (compound 22B)
A solution of 2-(4′-trifluoromethyl-biphenyl-4-yl)-ethanol (2.8 g, 10.3 mmol) and carbon tetrabromide (3.8 g, 11.5 mmol) in 50 ml of dichloromethane was cooled in ice, and triphenylphosphine (2.9 g, 11.1 mmol) was added in portions over 10 minutes. The mixture was stirred at room temperature for 18 h, and the solvent was evaporated. The residue was stirred in 75 ml of ether, and the mixture was filtered. The insoluble material was washed on the funnel with fresh ether (3×75 ml). The combined ether filtrates were concentrated, and the crude product was purified by normal phase chromatography. MS m/z 328 (M−1).
Step 3. Preparation of {2-Methyl-4-[2-(4′-trifluoromethyl-biphenyl-4-yl)-ethylsulfanyl]-phenoxy}-acetic acid methyl ester (compound 22C)
A solution 4-(2-bromo-ethyl)-4′-trifluoromethyl-biphenyl (0.66 g, 2.0 mmol) and (4-mercapto-2-methyl-phenoxy)-acetic acid methyl ester (0.42 g, 2.0 mmol) in 10 ml of acetonitrile was treated with cesium carbonate (1.3 g, 4.0 mmol), and the mixture was stirred at room temperature for 18 h. The reaction mixture was added to 200 ml of brine and extracted with ethyl acetate (4×75 ml). The combined extracts were washed with brine (2×200 ml), then dried over anhydrous sodium sulfate and concentrated. The crude product was purified by normal phase chromatography. MS m/z 461 (M+1).
Step 4. Preparation of {2-Methyl-4-[2-(4′-trifluoromethyl-biphenyl-4-yl)-ethylsulfanyl]-phenoxy}acetic acid (compound 22)
A solution of {2-methyl-4-[2-(4′-trifluoromethyl-biphenyl-4-yl)-ethylsulfanyl]-phenoxy}-acetic acid methyl ester (0.78 g, 1.7 mmol) in 10 ml of tetrahydrofuran and 2.0 ml of water was treated with lithium hydroxide monohydrate (0.21 g, 5.0 mmol), and the mixture was stirred at room temperature for 2 h. The reaction mixture was diluted with 5.0 ml of water and made strongly acidic by the addition of 4.0 N hydrochloric acid. The mixture was extracted with ethyl acetate (4×30 ml), and the combined extracts were washed with brine (2×50 ml), then dried over anhydrous sodium sulfate and concentrated. The crude product was recrystallized from ethyl acetate/hexane. mp 132-134° C. IR (thin film) cm−1: 1741, 1709, 1490, 1326, 1239, 1110; 400 MHz 1H NMR (DMSO-d6) δ 7.82 (d, 2H, J=8.0 Hz), 7.74 (d, 2H, J=8.0 Hz), 7.61 (d, 2H, J=8.3 Hz), 7.31 (d, 2H, J=8.3 Hz), 7.15 (m, 2H), 6.75 (d, 1H, J=8.3 Hz), 4.64 (s, 2H), 3.11 (t, 2H, J=7.6 Hz), 2.82 (t, 2H, J=7.6 Hz), 2.12 (s, 3H); MS m/z 447 (M+1). Anal. Calc'd for C24H21F3O3S: C, 64.56; H, 4.74; found: C, 64.45; H, 4.58.
Step 1. Preparation of 2-(4′-Trifluoromethyl-biphenyl-4-yl)-ethanol (compound 22A)
A mixture of 2-(4-bromo-phenyl)-ethanol (2.3 ml, 3.3 g, 16.4 mmol), 4-trifluoromethylphenylboronic acid (5.0 g, 26.3 mmol), 1.0 M aqueous sodium carbonate solution (44.0 ml), and tetrakis(triphenylphosphine)palladium (0.98 g, 0.85 mmol) in 180 ml of ethanol and 180 ml of toluene was heated at reflux for 4 h. The cooled reaction mixture was diluted with 500 ml of ethyl acetate and filtered through a bed of Celite filter-aid. The filtrate was washed with 5% aqueous sodium carbonate solution (2×750 ml) and brine (3×750 ml), then dried over anhydrous sodium sulfate and concentrated. The crude product was purified by normal phase chromatography. MS m/z 266 (M).
Step 2. Preparation of 4-(2-Bromo-ethyl)-4′-trifluoromethyl-biphenyl (compound 22B)
A solution of 2-(4′-trifluoromethyl-biphenyl-4-yl)-ethanol (2.8 g, 10.3 mmol) and carbon tetrabromide (3.8 g, 11.5 mmol) in 50 ml of dichloromethane was cooled in ice, and triphenylphosphine (2.9 g, 11.1 mmol) was added in portions over 10 minutes. The mixture was stirred at room temperature for 18 h, and the solvent was evaporated. The residue was stirred in 75 ml of ether, and the mixture was filtered. The insoluble material was washed on the funnel with fresh ether (3×75 ml). The combined ether filtrates were concentrated, and the crude product was purified by normal phase chromatography. MS m/z 328 (M−1).
Step 3. Preparation of {2-Methyl-4-[2-(4′-trifluoromethyl-biphenyl-4-yl)-ethylsulfanyl]-phenoxy}-acetic acid methyl ester (compound 22C)
A solution 4-(2-bromo-ethyl)-4′-trifluoromethyl-biphenyl (0.66 g, 2.0 mmol) and (4-mercapto-2-methyl-phenoxy)-acetic acid methyl ester (0.42 g, 2.0 mmol) in 10 ml of acetonitrile was treated with cesium carbonate (1.3 g, 4.0 mmol), and the mixture was stirred at room temperature for 18 h. The reaction mixture was added to 200 ml of brine and extracted with ethyl acetate (4×75 ml). The combined extracts were washed with brine (2×200 ml), then dried over anhydrous sodium sulfate and concentrated. The crude product was purified by normal phase chromatography. MS m/z 461 (M+1).
Step 4. Preparation of {2-Methyl-4-[2-(4′-trifluoromethyl-biphenyl-4-yl)-ethylsulfanyl]-phenoxy}acetic acid (compound 22)
A solution of {2-methyl-4-[2-(4′-trifluoromethyl-biphenyl-4-yl)-ethylsulfanyl]-phenoxy}-acetic acid methyl ester (0.78 g, 1.7 mmol) in 10 ml of tetrahydrofuran and 2.0 ml of water was treated with lithium hydroxide monohydrate (0.21 g, 5.0 mmol), and the mixture was stirred at room temperature for 2 h. The reaction mixture was diluted with 5.0 ml of water and made strongly acidic by the addition of 4.0 N hydrochloric acid. The mixture was extracted with ethyl acetate (4×30 ml), and the combined extracts were washed with brine (2×50 ml), then dried over anhydrous sodium sulfate and concentrated. The crude product was recrystallized from ethyl acetate/hexane. mp 132-134° C. IR (thin film) cm−1: 1741, 1709, 1490, 1326, 1239, 1110; 400 MHz 1H NMR (DMSO-d6) δ 7.82 (d, 2H, J=8.0 Hz), 7.74 (d, 2H, J=8.0 Hz), 7.61 (d, 2H, J=8.3 Hz), 7.31 (d, 2H, J=8.3 Hz), 7.15 (m, 2H), 6.75 (d, 1H, J=8.3 Hz), 4.64 (s, 2H), 3.11 (t, 2H, J=7.6 Hz), 2.82 (t, 2H, J=7.6 Hz), 2.12 (s, 3H); MS m/z 447 (M+1). Anal. Calc'd for C24H21F3O3S: C, 64.56; H, 4.74; found: C, 64.45; H, 4.58.
Synthesis of {5-Methoxy-2-methyl-4-[2-(4′-trifluoromethyl-biphenyl-4-yl)-ethylsulfanyl]-phenoxy}-acetic acid (compound 23)
Step 1. Preparation of {5-Methoxy-2-methyl-4-[2-(4-trifluoromethyl-biphenyl-4-yl)-ethylsulfanyl]-phenoxy}-acetic acid methyl ester (compound 23A)
The title compound was prepared in the manner analogous to Example 22C using 4-(2-bromo-ethyl)-4′-trifluoromethyl-biphenyl and 1D. MS m/z 491 (M+1).
Step 2. Preparation of {5-Methoxy-2-methyl-4-[2-(4′-trifluoromethyl-biphenyl-4-yl)-ethylsulfanyl]-phenoxy}-acetic acid (compound 23)
The title compound was prepared in the manner analogous to Example 1 using {5-methoxy-2-methyl-4-[2-(4′-trifluoromethyl-biphenyl-4-yl)-ethylsulfanyl]-phenoxy}-acetic acid methyl ester. mp 169-171° C.; IR (thin film) cm−1: 1718, 1500, 1330, 1162, 1109, 1052; 400 MHz 1H NMR (DMSO-d6) δ 7.82 (d, 2H, J=8.3 Hz), 7.74 (d, 2H, J=8.6 Hz), 7.61 (d, 2H, J=8.3 Hz), 7.31 (d, 2H, J=8.3 Hz), 7.06 (s, 1H), 6.53 (s, 1H), 4.69 (s, 2H), 3.73 (s, 3H), 3.02 (t, 2H, J=7.5 Hz), 2.78 (t, 2H, J=7.5 Hz), 2.06 (s, 3H); MS m/z 477 (M+1). Anal. Calc'd for C25H23F3O4S: C, 63.02; H, 4.87; found: C, 62.77; H, 4.62.
Step 1. Preparation of {5-Methoxy-2-methyl-4-[2-(4-trifluoromethyl-biphenyl-4-yl)-ethylsulfanyl]-phenoxy}-acetic acid methyl ester (compound 23A)
The title compound was prepared in the manner analogous to Example 22C using 4-(2-bromo-ethyl)-4′-trifluoromethyl-biphenyl and 1D. MS m/z 491 (M+1).
Step 2. Preparation of {5-Methoxy-2-methyl-4-[2-(4′-trifluoromethyl-biphenyl-4-yl)-ethylsulfanyl]-phenoxy}-acetic acid (compound 23)
The title compound was prepared in the manner analogous to Example 1 using {5-methoxy-2-methyl-4-[2-(4′-trifluoromethyl-biphenyl-4-yl)-ethylsulfanyl]-phenoxy}-acetic acid methyl ester. mp 169-171° C.; IR (thin film) cm−1: 1718, 1500, 1330, 1162, 1109, 1052; 400 MHz 1H NMR (DMSO-d6) δ 7.82 (d, 2H, J=8.3 Hz), 7.74 (d, 2H, J=8.6 Hz), 7.61 (d, 2H, J=8.3 Hz), 7.31 (d, 2H, J=8.3 Hz), 7.06 (s, 1H), 6.53 (s, 1H), 4.69 (s, 2H), 3.73 (s, 3H), 3.02 (t, 2H, J=7.5 Hz), 2.78 (t, 2H, J=7.5 Hz), 2.06 (s, 3H); MS m/z 477 (M+1). Anal. Calc'd for C25H23F3O4S: C, 63.02; H, 4.87; found: C, 62.77; H, 4.62.
Synthesis of {2-Methyl-4-[2-(4′-trifluoromethyl-biphenyl-4-yl)-vinyl]-phenoxy}-acetic acid (compound 24)
Step 1. Preparation of (4-Bromo-2-methyl-phenoxy)-acetic acid methyl ester (compound 24A)
A solution of o-tolyloxy-acetic acid methyl ester (Belleney J., et al., J. Heterocyclic Chem., 1984; 21:1431; 3.7 g, 20.5 mmol) in 70 ml of acetonitrile was treated in portions over 10 minutes with N-bromosuccinimide (3.8 g, 21.3 mmol). The mixture was stirred at room temperature for 18 h, and the solvent was evaporated. The residue was stirred in 75 ml of carbon tetrachloride, and the mixture was filtered. The insoluble material was washed on the funnel with fresh carbon tetrachloride (2×50 ml). The combined filtrates were concentrated, and the crude product was purified by normal phase chromatography. MS m/z 258 (M−1).
Step 2. Preparation of 4-Trifluoromethyl-4′-vinyl-biphenyl (compound 24B)
The title compound was prepared in the manner analogous to Example 3A using 1-bromo-4-vinyl-benzene and 4-trifluoromethylphenylboronic acid. MS m/z 248 (M).
Step 3. Preparation of {2-Methyl-4-[2-(4′-trifluoromethyl-biphenyl-4-yl)-vinyl]-phenoxy}-acetic acid methyl ester and {2-methyl-4-[2-(4′-trifluoromethyl-biphenyl-4-yl)-vinyl]-phenoxy}-acetic acid (compound 24)
A mixture of 4-trifluoromethyl-4′-vinyl-biphenyl (1.9 g, 7.7 mmol), (4-Bromo-2-methyl-phenoxy)-acetic acid methyl ester (2.0 g, 7.7 mmol), anhydrous sodium acetate (1.2 g, 14.6 mmol), N,N-dimethylglycine (0.23 g, 2.2 mmol), and palladium acetate (0.025 g, 0.11 mmol) in 10 ml of 1-methyl-pyrrolidin-2-one was heated at 130° C. for 10 h. The reaction mixture was partitioned between 250 ml of brine and 300 ml of ethyl acetate. The total mixture was filtered through a bed of Celite filter-aid. The organic layer was washed with 5% aqueous sodium carbonate solution (3×250 ml) and brine (2×250 ml), then dried over anhydrous sodium sulfate and concentrated. The residue was purified by normal phase chromatography to give {2-methyl-4-[2-(4′-trifluoromethyl-biphenyl-4-yl)-vinyl]-phenoxy}-acetic acid methyl ester; MS m/z 427 (M+1).
Step 1. Preparation of (4-Bromo-2-methyl-phenoxy)-acetic acid methyl ester (compound 24A)
A solution of o-tolyloxy-acetic acid methyl ester (Belleney J., et al., J. Heterocyclic Chem., 1984; 21:1431; 3.7 g, 20.5 mmol) in 70 ml of acetonitrile was treated in portions over 10 minutes with N-bromosuccinimide (3.8 g, 21.3 mmol). The mixture was stirred at room temperature for 18 h, and the solvent was evaporated. The residue was stirred in 75 ml of carbon tetrachloride, and the mixture was filtered. The insoluble material was washed on the funnel with fresh carbon tetrachloride (2×50 ml). The combined filtrates were concentrated, and the crude product was purified by normal phase chromatography. MS m/z 258 (M−1).
Step 2. Preparation of 4-Trifluoromethyl-4′-vinyl-biphenyl (compound 24B)
The title compound was prepared in the manner analogous to Example 3A using 1-bromo-4-vinyl-benzene and 4-trifluoromethylphenylboronic acid. MS m/z 248 (M).
Step 3. Preparation of {2-Methyl-4-[2-(4′-trifluoromethyl-biphenyl-4-yl)-vinyl]-phenoxy}-acetic acid methyl ester and {2-methyl-4-[2-(4′-trifluoromethyl-biphenyl-4-yl)-vinyl]-phenoxy}-acetic acid (compound 24)
A mixture of 4-trifluoromethyl-4′-vinyl-biphenyl (1.9 g, 7.7 mmol), (4-Bromo-2-methyl-phenoxy)-acetic acid methyl ester (2.0 g, 7.7 mmol), anhydrous sodium acetate (1.2 g, 14.6 mmol), N,N-dimethylglycine (0.23 g, 2.2 mmol), and palladium acetate (0.025 g, 0.11 mmol) in 10 ml of 1-methyl-pyrrolidin-2-one was heated at 130° C. for 10 h. The reaction mixture was partitioned between 250 ml of brine and 300 ml of ethyl acetate. The total mixture was filtered through a bed of Celite filter-aid. The organic layer was washed with 5% aqueous sodium carbonate solution (3×250 ml) and brine (2×250 ml), then dried over anhydrous sodium sulfate and concentrated. The residue was purified by normal phase chromatography to give {2-methyl-4-[2-(4′-trifluoromethyl-biphenyl-4-yl)-vinyl]-phenoxy}-acetic acid methyl ester; MS m/z 427 (M+1).
During the above sodium carbonate washings, a precipitate formed and was removed by filtration. The solid was stirred for 18 h in a solution of 150 ml of water, 50 ml of methanol, and 50 ml of 4.0 N hydrochloric acid. The acidified product was filtered and recrystallized from aqueous acetonitrile to give {2-methyl-4-[2-(4′-trifluoromethyl-biphenyl-4-yl)-vinyl]-phenoxy}-acetic acid. mp 243-245° C.; IR (thin film) cm−1: 1746, 1717, 1502, 1323, 1125, 1069; 400 MHz 1H NMR (DMSO-d6) δ 7.88 (d, 2H, J=8.0 Hz), 7.76 (d, 2H, J=8.3 Hz), 7.71 (d, 2H, J=8.5 Hz), 7.64 (d, 2H, J=8.5 Hz), 7.43 (d, 1H, J=1.7 Hz), 7.32 (dd, 1H, J=2.1, 8.5 Hz), 7.21 (d, 1H, J=16.5 Hz), 7.10 (d, 1H, J=16.4 Hz), 6.79 (d, 1H, J=8.5 Hz), 4.68 (s, 2H), 2.18 (s, 3H); MS m/z 413 (M+1). Anal. Calc'd for C24H19F3O3: C, 69.90; H, 4.64; found: C, 69.77; H, 4.57.
Synthesis of {2-Methyl-4-[2-(4′-trifluoromethyl-biphenyl-4-yl)-ethyl]-phenoxy}-acetic acid (compound 25)
A solution of {2-methyl-4-[2-4′-trifluoromethyl-biphenyl-4-yl)-vinyl]-phenoxy}-acetic acid (0.98 g, 2.4 mmol) in 100 ml of tetrahydrofuran was hydrogenated over 0.16 g of 20% palladium on carbon catalyst. The catalyst was removed by filtration, and the filtrate was evaporated. The crude product was recrystallized from aqueous acetonitrile. mp 174-176° C.; IR (thin film) cm−1: 1747, 1711, 1500, 1318, 1160, 1123; 400 MHz 1H NMR (DMSO-d6) δ 7.83 (d, 2H, J=8.1 Hz), 7.74 (d, 2H, J=8.3 Hz), 7.60 (d, 2H, J=6.5 Hz), 7.32 (d, 2H, J=8.3 Hz), 7.01 (d, 1H, J=2.0 Hz), 6.94 (dd, 1H, J=2.0, 8.3 Hz), 6.66 (d, 1H, J=8.3 Hz), 4.59 (s, 2H), 2.83 (m, 2H), 2.76 (m, 2H), 2.11 (s, 3H); MS m/z 413 (M−1). Anal. Calc'd for C24H21F3O3: C, 69.56; H, 5.11; found: C, 69.28; H, 4.96.
Synthesis of {7-[4-(5-Trifluoromethyl-pyridin-2-yl)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 26)
Step 1. Preparation of {7-[4-(5-Trifluoromethyl-pyridin-2-yl)-benzylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 26A)
The title compound was prepared in the manner analogous to Example 1F using 18B and 12C. MS m/z 474 (M+1).
Step 2. Preparation of {7-[4-(5-Trifluoromethyl-pyridin-2-yl)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 26)
The title compound was prepared from the product of Example 26A in the manner analogous to Example 1. mp 220° C. (dec.); 400 MHz 1H NMR (DMSO-d6) δ 12.94 (s, 1H), 8.97 (s, 1H), 8.2 (dd, 1H, J=2 Hz, J′=8.8 Hz), 8.13 (d, 1H, J=8.4 Hz), 8.01 (d, 2H, J=8.4 Hz), 7.34 (d, 2H, J=8.8 Hz), 7.07 (d, 1H, J=8.4 Hz), 6.57 (d, 1H, J=8 Hz), 4.61 (s, 2), 4.07 (s, 2H), 2.72 (m, 4H), 1.89 (m, 2H). MS m/z 460 (M+1).
Step 1. Preparation of {7-[4-(5-Trifluoromethyl-pyridin-2-yl)-benzylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 26A)
The title compound was prepared in the manner analogous to Example 1F using 18B and 12C. MS m/z 474 (M+1).
Step 2. Preparation of {7-[4-(5-Trifluoromethyl-pyridin-2-yl)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 26)
The title compound was prepared from the product of Example 26A in the manner analogous to Example 1. mp 220° C. (dec.); 400 MHz 1H NMR (DMSO-d6) δ 12.94 (s, 1H), 8.97 (s, 1H), 8.2 (dd, 1H, J=2 Hz, J′=8.8 Hz), 8.13 (d, 1H, J=8.4 Hz), 8.01 (d, 2H, J=8.4 Hz), 7.34 (d, 2H, J=8.8 Hz), 7.07 (d, 1H, J=8.4 Hz), 6.57 (d, 1H, J=8 Hz), 4.61 (s, 2), 4.07 (s, 2H), 2.72 (m, 4H), 1.89 (m, 2H). MS m/z 460 (M+1).
Synthesis of {5-Methyl-7-[4-(5-trifluoromethyl-pyridin-2-yl)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 27)
Step 1. Preparation of {5-Methyl-7-[4-(5-trifluoromethyl-pyridin-2-yl)-benzylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 27A)
The title compound was prepared from the products of Example 18B and (7-mercapto-5-methyl-indan-4-yloxy)-acetic acid methyl ester (prepared in a similar manner as described for Example 12C) in a manner analogous to Example 1F. MS m/z 488 (M+1).
Step 2. Preparation of {5-Methyl-7-[4-(5-trifluoromethyl-pyridin-2-yl)-benzylsulfanyl]-indan-4yloxy}-acetic acid (compound 27)
The title compound was prepared from the product of Example 27A in the manner analogous to Example 1. mp 186° C.; 400 MHz 1H NMR (DMSO-d6) δ 12.94 (s, 1H), 8.97 (s, 1H), 8.24 (dd, 1H, J=2 Hz, J′=8.8 Hz), 8.14 (d, 1H, J=8.4 Hz), 8.01 (d, 2H, J=8.4 Hz), 7.34 (d, 2H, J=8.8 Hz), 6.99 (s, 1H), 4.41 (s, 2H), 4.13 (s, 2H), 2.83 (t, 2H, J=7.2 Hz), 2.62 (t, 2H, J=7.2 Hz), 2.13 (s, 3H), 1.87 (m, 2H). MS m/z 474 (M+1).
Step 1. Preparation of {5-Methyl-7-[4-(5-trifluoromethyl-pyridin-2-yl)-benzylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 27A)
The title compound was prepared from the products of Example 18B and (7-mercapto-5-methyl-indan-4-yloxy)-acetic acid methyl ester (prepared in a similar manner as described for Example 12C) in a manner analogous to Example 1F. MS m/z 488 (M+1).
Step 2. Preparation of {5-Methyl-7-[4-(5-trifluoromethyl-pyridin-2-yl)-benzylsulfanyl]-indan-4yloxy}-acetic acid (compound 27)
The title compound was prepared from the product of Example 27A in the manner analogous to Example 1. mp 186° C.; 400 MHz 1H NMR (DMSO-d6) δ 12.94 (s, 1H), 8.97 (s, 1H), 8.24 (dd, 1H, J=2 Hz, J′=8.8 Hz), 8.14 (d, 1H, J=8.4 Hz), 8.01 (d, 2H, J=8.4 Hz), 7.34 (d, 2H, J=8.8 Hz), 6.99 (s, 1H), 4.41 (s, 2H), 4.13 (s, 2H), 2.83 (t, 2H, J=7.2 Hz), 2.62 (t, 2H, J=7.2 Hz), 2.13 (s, 3H), 1.87 (m, 2H). MS m/z 474 (M+1).
Synthesis of [5-Methyl-7-(4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-indan-4-yloxy]-acetic acid (compound 28)
Step 1. Preparation of [5-Methyl-7-(4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-indan-4-yloxy]-acetic acid methyl ester (compound 28A)
The title compound was prepared from the product of Example 3B and (7-Mercapto-5-methyl-indan-4-yloxy)-acetic acid methyl ester in a manner analogous to Example 1F. MS m/z 487 (M+1).
Step 2. Preparation of [5-Methyl-7-(4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-indan-4-yloxy]-acetic acid (compound 28)
The title compound was prepared from the product of Example 28A in the manner analogous to Example 1. mp 177° C.; 400 MHz 1H NMR (DMSO-d6) δ 12.82 (s, 1H), 7.82(d, 2H, J=8 Hz), 7.75 (d, J=8 Hz), 7.61 (d, 2H, J=8 Hz), 7.35 (d, 2H, J=8 Hz), 6.99 (s, 1H), 4.41 (s, 2H), 4.12 (s, 2H), 2.83 (t, 2H, J=7.2 Hz), 2.62 (t, 2H, J=7.2 Hz), 2.13 (s, 3H), 1.89 (m, 2H). MS m/z 473 (M+1).
Step 1. Preparation of [5-Methyl-7-(4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-indan-4-yloxy]-acetic acid methyl ester (compound 28A)
The title compound was prepared from the product of Example 3B and (7-Mercapto-5-methyl-indan-4-yloxy)-acetic acid methyl ester in a manner analogous to Example 1F. MS m/z 487 (M+1).
Step 2. Preparation of [5-Methyl-7-(4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-indan-4-yloxy]-acetic acid (compound 28)
The title compound was prepared from the product of Example 28A in the manner analogous to Example 1. mp 177° C.; 400 MHz 1H NMR (DMSO-d6) δ 12.82 (s, 1H), 7.82(d, 2H, J=8 Hz), 7.75 (d, J=8 Hz), 7.61 (d, 2H, J=8 Hz), 7.35 (d, 2H, J=8 Hz), 6.99 (s, 1H), 4.41 (s, 2H), 4.12 (s, 2H), 2.83 (t, 2H, J=7.2 Hz), 2.62 (t, 2H, J=7.2 Hz), 2.13 (s, 3H), 1.89 (m, 2H). MS m/z 473 (M+1).
Synthesis of (4-{4-[2-(3-Fluoro-phenyl)-vinyl]-benzylsulfanyl}-5-methoxy-2-methyl-phenoxy)-acetic acid (compound 29)
Step 1. Preparation of {4-[2-(3-Fluoro-phenyl)-vinyl]-phenyl}-methanol (compound 29A)
(4-Bromo-phenyl)-methanol (1 g, 5.35 mmol), 3-fluorostyrene (718 mg, 5.89 mmol), palladium acetate (60 mg, 0.3 mmol), and triphenylphosphine (140 mg, 0.6 mmol) were heated in triethylamine at 90° C. in a sealed tube for 18 h. The reaction was concentrated and purified by normal phase chromatography to afford the title compound. MS m/z 227 (M−1).
Step 2. Preparation of {4-[2-(3-Fluoro-phenyl)-vinyl]-phenyl}-chloromethane (compound 29B)
The title compound was prepared from the product of Example 29A in a manner analogous to Example 3B. MS m/z 245 (M−1).
Step 3. Preparation of (4-{4-[2-(3-Fluoro-phenyl)-vinyl]-benzylsulfanyl}-5-methoxy-2-methyl-phenoxy)-acetic acid methyl ester (compound 29C)
The title compound was prepared from the product of Example 29B and the product of Example 1D in a manner analogous to Example 1F. MS m/z 453 (M+1).
Step 4. Preparation of (4-{4-[2-(3-Fluoro-phenyl)-vinyl]-benzylsulfanyl}-5-methoxy-2-methyl-phenoxy)-acetic acid (compound 29)
The title compound was prepared from the product of Example 29C in the manner analogous to Example 1. mp 154° C. (dec.); 400 MHz 1H NMR (DMSO-d6) δ 12.94 (s, 1H), 7.46-7.34 (m, 5H), 7.27-7.15 (m, 4H), 7.06-6.99 (m, 2H), 6.52 (s, 1H), 4.69 (s, 2H), 3.98 (s, 2H), 3.73 (s, 3H), 2.00 (s, 3H), MS m/z 439 (M+1).
Step 1. Preparation of {4-[2-(3-Fluoro-phenyl)-vinyl]-phenyl}-methanol (compound 29A)
(4-Bromo-phenyl)-methanol (1 g, 5.35 mmol), 3-fluorostyrene (718 mg, 5.89 mmol), palladium acetate (60 mg, 0.3 mmol), and triphenylphosphine (140 mg, 0.6 mmol) were heated in triethylamine at 90° C. in a sealed tube for 18 h. The reaction was concentrated and purified by normal phase chromatography to afford the title compound. MS m/z 227 (M−1).
Step 2. Preparation of {4-[2-(3-Fluoro-phenyl)-vinyl]-phenyl}-chloromethane (compound 29B)
The title compound was prepared from the product of Example 29A in a manner analogous to Example 3B. MS m/z 245 (M−1).
Step 3. Preparation of (4-{4-[2-(3-Fluoro-phenyl)-vinyl]-benzylsulfanyl}-5-methoxy-2-methyl-phenoxy)-acetic acid methyl ester (compound 29C)
The title compound was prepared from the product of Example 29B and the product of Example 1D in a manner analogous to Example 1F. MS m/z 453 (M+1).
Step 4. Preparation of (4-{4-[2-(3-Fluoro-phenyl)-vinyl]-benzylsulfanyl}-5-methoxy-2-methyl-phenoxy)-acetic acid (compound 29)
The title compound was prepared from the product of Example 29C in the manner analogous to Example 1. mp 154° C. (dec.); 400 MHz 1H NMR (DMSO-d6) δ 12.94 (s, 1H), 7.46-7.34 (m, 5H), 7.27-7.15 (m, 4H), 7.06-6.99 (m, 2H), 6.52 (s, 1H), 4.69 (s, 2H), 3.98 (s, 2H), 3.73 (s, 3H), 2.00 (s, 3H), MS m/z 439 (M+1).
Synthesis of {2-Methyl-4-[4-(5-trifluoromethyl-pyridin-2-yl)-benzylsulfanyl]-phenoxy}-acetic acid (compound 30)
Step 1. Preparation of {2-Methyl-4-[4-(5-trifluoromethyl-pyridin-2-yl)-benzylsulfanyl]-phenoxy}-acetic acid methyl ester (compound 30A)
The title compound was prepared from the product of Example 2C and the product of Example 18B in a manner analogous to Example 1F. MS m/z 448 (M+1).
Preparation of {2-Methyl-4-[4-(5-trifluoromethyl-pyridin-2-yl)-benzylsulfanyl]-phenoxy}-acetic acid
The title compound was prepared from the product of Example 30A in the manner analogous to Example 1. 400 MHz 1H NMR (DMSO-d6) δ 8.97 (s, 1H), 8.2 (dd, 1H, J=2 Hz, J′=8.8 Hz), 8.13 (d, 1H, J=8.4 Hz), 8.01 (d, 2H, J=8.4 Hz), 7.36 (d, 2H, J=8.4 Hz), 7.07 (s, 1H), 7.01 (dd, 1H, J=8.4 Hz, J′=2.4 Hz), 6.57 (d, 1H, J=8.8 Hz), 4.09 (s, 2H), 4.06 (s, 2H), 2.04 (s, 3H). MS m/z 434 (M+1).
Step 1. Preparation of {2-Methyl-4-[4-(5-trifluoromethyl-pyridin-2-yl)-benzylsulfanyl]-phenoxy}-acetic acid methyl ester (compound 30A)
The title compound was prepared from the product of Example 2C and the product of Example 18B in a manner analogous to Example 1F. MS m/z 448 (M+1).
Preparation of {2-Methyl-4-[4-(5-trifluoromethyl-pyridin-2-yl)-benzylsulfanyl]-phenoxy}-acetic acid
The title compound was prepared from the product of Example 30A in the manner analogous to Example 1. 400 MHz 1H NMR (DMSO-d6) δ 8.97 (s, 1H), 8.2 (dd, 1H, J=2 Hz, J′=8.8 Hz), 8.13 (d, 1H, J=8.4 Hz), 8.01 (d, 2H, J=8.4 Hz), 7.36 (d, 2H, J=8.4 Hz), 7.07 (s, 1H), 7.01 (dd, 1H, J=8.4 Hz, J′=2.4 Hz), 6.57 (d, 1H, J=8.8 Hz), 4.09 (s, 2H), 4.06 (s, 2H), 2.04 (s, 3H). MS m/z 434 (M+1).
Synthesis of {4-[4-(2,4-Difluoro-benzyloxy)-benzylsulfanyl]-2-methyl-phenoxy}-acetic acid (compound 31)
Step 1. Preparation of [4-(2,4-Difluoro-benzyloxy)phenyl]-methanol (compound 31A)
The title compound was prepared in the manner analogous to Example 14A using 1-bromomethyl-2,4-difluoro-benzene and 4-hydroxymethyl-phenol. MS m/z 233 (M-OH).
Step 2. Preparation of 1-(4-Chloromethyl-phenoxymethyl)-2,4-difluoro-benzene (compound 31B)
The title compound was prepared in the manner analogous to Example 3B using 31A. MS m/z 233 (M-Cl).
Step 3. Preparation of {4-[4-(2,4-Difluoro-benzyloxy)-benzylsulfanyl]-2-methyl-phenoxy}-acetic acid methyl ester (compound 31C)
The title compound was prepared in the manner analogous to Example 1F using 31B and 2C. MS m/z 445 (M+1).
Step 4. Preparation of {4-[4-(2,4-Difluoro-benzyloxy)-benzylsulfanyl]-2-methyl-phenoxy}-acetic acid
To a solution of the product from Example 31C (1.5 g, 3.3 mmol) in a mixture of 20 mL of tetrahydrofuran and 4 mL of water was added lithium hydroxide monohydrate (0.42 g, 9.9 mmol). The reaction mixture was stirred at room temperature for 18 hrs and then evaporated to afford a residue, which was suspended in 50 mL of water. The mixture was acidified with 1N hydrochloric acid to pH 2. The precipitated solid was collected be filtration, washed with water, and then dried to provide the title compound without any further purification. mp 139-141° C.; IR (KBr) cm−1: 3081, 2917, 1735, 1604, 1508, 1233; 400 MHz 1H NMR (DMSO-d6): δ 7.51-7.59 (m, 1H), 7.02-7.31 (m, 6H), 6.85-6.92 (m, 2H), 6.68 (d, 1H, J=8.6 Hz), 5.01 (s, 2H), 4.60 (s, 2H), 4.00 (s, 2H), 2.08 (s, 3H); MS m/z 429 (M−1). Anal. Calc'd for C23H20F2O4S: C, 64.17; H, 4.68; found: C, 64.11; H, 4.59.
Step 1. Preparation of [4-(2,4-Difluoro-benzyloxy)phenyl]-methanol (compound 31A)
The title compound was prepared in the manner analogous to Example 14A using 1-bromomethyl-2,4-difluoro-benzene and 4-hydroxymethyl-phenol. MS m/z 233 (M-OH).
Step 2. Preparation of 1-(4-Chloromethyl-phenoxymethyl)-2,4-difluoro-benzene (compound 31B)
The title compound was prepared in the manner analogous to Example 3B using 31A. MS m/z 233 (M-Cl).
Step 3. Preparation of {4-[4-(2,4-Difluoro-benzyloxy)-benzylsulfanyl]-2-methyl-phenoxy}-acetic acid methyl ester (compound 31C)
The title compound was prepared in the manner analogous to Example 1F using 31B and 2C. MS m/z 445 (M+1).
Step 4. Preparation of {4-[4-(2,4-Difluoro-benzyloxy)-benzylsulfanyl]-2-methyl-phenoxy}-acetic acid
To a solution of the product from Example 31C (1.5 g, 3.3 mmol) in a mixture of 20 mL of tetrahydrofuran and 4 mL of water was added lithium hydroxide monohydrate (0.42 g, 9.9 mmol). The reaction mixture was stirred at room temperature for 18 hrs and then evaporated to afford a residue, which was suspended in 50 mL of water. The mixture was acidified with 1N hydrochloric acid to pH 2. The precipitated solid was collected be filtration, washed with water, and then dried to provide the title compound without any further purification. mp 139-141° C.; IR (KBr) cm−1: 3081, 2917, 1735, 1604, 1508, 1233; 400 MHz 1H NMR (DMSO-d6): δ 7.51-7.59 (m, 1H), 7.02-7.31 (m, 6H), 6.85-6.92 (m, 2H), 6.68 (d, 1H, J=8.6 Hz), 5.01 (s, 2H), 4.60 (s, 2H), 4.00 (s, 2H), 2.08 (s, 3H); MS m/z 429 (M−1). Anal. Calc'd for C23H20F2O4S: C, 64.17; H, 4.68; found: C, 64.11; H, 4.59.
Synthesis of {4-[4-(2,4-Dichloro-benzyloxy)-benzylsulfanyl]-2-methyl-phenoxy}-acetic acid (compound 32)
Step 1. Preparation of [4-(2,4-Dichloro-benzyloxy)-phenyl]-methanol (compound 32A)
The title compound was prepared in the manner analogous to Example 14A using 2,4-dichloro-1-chloromethyl-benzene and 4-hydroxymethyl-phenol. MS m/z 265 (M-OH).
Step 2. Preparation of 2,4-Dichloro-1-(4-chloromethyl-phenoxymethyl)-benzene (compound 32B)
The title compound was prepared in the manner analogous to Example 3B using 32A. MS m/z 265 (M-Cl).
Step 3. Preparation of {4-[4-(2,4-Dichloro-benzyloxy)-benzylsulfanyl]-2-methyl-phenoxy}-acetic acid methyl ester (compound 32C)
The title compound was prepared in the manner analogous to Example 1F using 32B and 2C. MS m/z 477 (M+1).
Step 4. Preparation of {4-[4-(2,4-Dichloro-benzyloxy)-benzylsulfanyl]-2-methyl-phenoxy}-acetic acid (compound 32)
The title compound was prepared in the manner analogous to Example 1 using 32C. mp 143-145° C.; IR (KBr) cm−1: 3062, 2936, 1724, 1492, 1227, 1192; 400 MHz 1H NMR (DMSO-d6): δ 12.97 (br(s), 1H), 7.64 (d, 1H, J=2.0 Hz), 7.54 (d, 1H, J=8.3 Hz), 7.42 (dd, 1H, J=8.3, 2.0 Hz), 7.01-7.20 (m, 4H), 6.84-6.92 (m, 2H), 6.69 (d, 1H, J=8.5 Hz), 5.05 (s, 2H), 4.62 (s, 2H), 4.01 (s, 2H), 2.08 (s, 3H); MS m/z 461 (M−1). Anal. Calc'd for C23H20Cl2O4S: C, 59.62; H, 4.35; found: C, 59.33; H, 4.28.
Step 1. Preparation of [4-(2,4-Dichloro-benzyloxy)-phenyl]-methanol (compound 32A)
The title compound was prepared in the manner analogous to Example 14A using 2,4-dichloro-1-chloromethyl-benzene and 4-hydroxymethyl-phenol. MS m/z 265 (M-OH).
Step 2. Preparation of 2,4-Dichloro-1-(4-chloromethyl-phenoxymethyl)-benzene (compound 32B)
The title compound was prepared in the manner analogous to Example 3B using 32A. MS m/z 265 (M-Cl).
Step 3. Preparation of {4-[4-(2,4-Dichloro-benzyloxy)-benzylsulfanyl]-2-methyl-phenoxy}-acetic acid methyl ester (compound 32C)
The title compound was prepared in the manner analogous to Example 1F using 32B and 2C. MS m/z 477 (M+1).
Step 4. Preparation of {4-[4-(2,4-Dichloro-benzyloxy)-benzylsulfanyl]-2-methyl-phenoxy}-acetic acid (compound 32)
The title compound was prepared in the manner analogous to Example 1 using 32C. mp 143-145° C.; IR (KBr) cm−1: 3062, 2936, 1724, 1492, 1227, 1192; 400 MHz 1H NMR (DMSO-d6): δ 12.97 (br(s), 1H), 7.64 (d, 1H, J=2.0 Hz), 7.54 (d, 1H, J=8.3 Hz), 7.42 (dd, 1H, J=8.3, 2.0 Hz), 7.01-7.20 (m, 4H), 6.84-6.92 (m, 2H), 6.69 (d, 1H, J=8.5 Hz), 5.05 (s, 2H), 4.62 (s, 2H), 4.01 (s, 2H), 2.08 (s, 3H); MS m/z 461 (M−1). Anal. Calc'd for C23H20Cl2O4S: C, 59.62; H, 4.35; found: C, 59.33; H, 4.28.
Synthesis of {4-[4-(4-Methoxy-benzyloxy)-benzylsulfanyl]-2-methyl-phenoxy}-acetic acid (compound 33)
Step 1. Preparation of [4-(4-Methoxy-benzyloxy)-phenyl]-methanol (compound 33A)
The title compound was prepared in the manner analogous to Example 14A using 1-chloromethyl-4-methoxy-benzene and 4-hydroxymethyl-phenol. MS m/z 227 (M-OH).
Step 2. Preparation of 4-Methoxy-1-(4-chloromethyl-phenoxymethyl)-benzene (compound 33B)
The title compound was prepared in the manner analogous to Example 3B using 33A. MS m/z 227 (M-Cl).
Step 3. Preparation of {4-[4-(4Methoxy-benzyloxy)-benzylsulfanyl]-2-methyl-phenoxy}-acetic acid methyl ester (compound 33C)
The title compound was prepared in the manner analogous to Example 1F using 33B and 2C. MS m/z 439 (M+1).
Step 4. Preparation of {4-[4(4-Methoxy-benzyloxy)-benzylsulfanyl]-2-methyl-phenoxy}-acetic acid (compound 33)
The title compound was prepared in the manner analogous to Example 1 using 33C. mp 150-152° C.; HPLC: area %=96.69, r.t.=2.93 min., γ=214 nm, mobile phase=acetonitrile/water with 0.10% TFA; IR (KBr) cm−1: 2929, 1728, 1707, 1513, 1491, 1225; 400 MHz 1H NMR (DMSO-d6): δ 12.98 (br(s), 1H), 7.27-7.23 (m, 2H), 7.02-7.16 (m, 4H), 6.81-6.91 (m, 4H), 6.69 (d, 1H, J=8.6 Hz), 4.91 (s, 2H), 4.61 (s, 2H), 3.99 (s, 2H), 3.69 (s, 3H), 2.08 (s, 3H); MS m/z 423 (M−1). Anal. Calc'd for C24H24O3S: C, 67.90; H, 5.70; found: C, 67.48; H, 5.59.
Step 1. Preparation of [4-(4-Methoxy-benzyloxy)-phenyl]-methanol (compound 33A)
The title compound was prepared in the manner analogous to Example 14A using 1-chloromethyl-4-methoxy-benzene and 4-hydroxymethyl-phenol. MS m/z 227 (M-OH).
Step 2. Preparation of 4-Methoxy-1-(4-chloromethyl-phenoxymethyl)-benzene (compound 33B)
The title compound was prepared in the manner analogous to Example 3B using 33A. MS m/z 227 (M-Cl).
Step 3. Preparation of {4-[4-(4Methoxy-benzyloxy)-benzylsulfanyl]-2-methyl-phenoxy}-acetic acid methyl ester (compound 33C)
The title compound was prepared in the manner analogous to Example 1F using 33B and 2C. MS m/z 439 (M+1).
Step 4. Preparation of {4-[4(4-Methoxy-benzyloxy)-benzylsulfanyl]-2-methyl-phenoxy}-acetic acid (compound 33)
The title compound was prepared in the manner analogous to Example 1 using 33C. mp 150-152° C.; HPLC: area %=96.69, r.t.=2.93 min., γ=214 nm, mobile phase=acetonitrile/water with 0.10% TFA; IR (KBr) cm−1: 2929, 1728, 1707, 1513, 1491, 1225; 400 MHz 1H NMR (DMSO-d6): δ 12.98 (br(s), 1H), 7.27-7.23 (m, 2H), 7.02-7.16 (m, 4H), 6.81-6.91 (m, 4H), 6.69 (d, 1H, J=8.6 Hz), 4.91 (s, 2H), 4.61 (s, 2H), 3.99 (s, 2H), 3.69 (s, 3H), 2.08 (s, 3H); MS m/z 423 (M−1). Anal. Calc'd for C24H24O3S: C, 67.90; H, 5.70; found: C, 67.48; H, 5.59.
Synthesis of {4-[4-(4-tert-Butyl-benzyloxy)-benzylsulfanyl]-2-methyl-phenoxy}-acetic acid (compound 34)
Step 1. Preparation of [4-(4-tert-Butyl-benzyloxy)-phenyl]-methanol (compound 34A)
The title compound was prepared in the manner analogous to Example 14A using 1-bromomethyl-4-tert-butyl-benzene and 4-hydroxymethyl-phenol. MS m/z 253 (M-OH).
Step 2. Preparation of 4-tert-Butyl-1-(4-chloromethyl-phenoxymethyl)-benzene (compound 34B)
The title compound was prepared in the manner analogous to Example 3B using 34A. MS m/z 253 (M-Cl).
Step 3. Preparation of {4-[4-(4-tert-Butyl-benzyloxy)-benzylsulfanyl]-2-methyl-phenoxy}-acetic acid methyl ester (compound 34C)
The title compound was prepared in the manner analogous to Example 1F using 34B and 2C. MS m/z 465 (M+1).
Step 4. Preparation of {4-[4-(4-tert-Butyl-benzyloxy)-benzylsulfanyl]-2-methyl-phenoxy}-acetic acid (compound 34)
The title compound was prepared in the manner analogous to Example 1 using 34C. mp 135-137° C.; IR (KBr) cm−1: 2961, 2908, 1751, 1495, 1233, 1194; 400 MHz 1H NMR (DMSO-d6): δ 13.00 (br(s), 1H), 7.27-7.38 (m, 4H), 7.02-7.17 (m, 4H), 6.82-6.88 (m, 2H), 6.69 (d, 1H, J=8.5 Hz), 4.96 (s, 2H), 4.61 (s, 2H), 3.99 (s, 2H), 2.08 (s, 3H), 1.22 (s, 9H); MS m/z 451 (M+1). Anal. Calc'd for C27H30O4S: C, 71.97; H, 6.71; found: C, 71.66; H, 6.52.
Step 1. Preparation of [4-(4-tert-Butyl-benzyloxy)-phenyl]-methanol (compound 34A)
The title compound was prepared in the manner analogous to Example 14A using 1-bromomethyl-4-tert-butyl-benzene and 4-hydroxymethyl-phenol. MS m/z 253 (M-OH).
Step 2. Preparation of 4-tert-Butyl-1-(4-chloromethyl-phenoxymethyl)-benzene (compound 34B)
The title compound was prepared in the manner analogous to Example 3B using 34A. MS m/z 253 (M-Cl).
Step 3. Preparation of {4-[4-(4-tert-Butyl-benzyloxy)-benzylsulfanyl]-2-methyl-phenoxy}-acetic acid methyl ester (compound 34C)
The title compound was prepared in the manner analogous to Example 1F using 34B and 2C. MS m/z 465 (M+1).
Step 4. Preparation of {4-[4-(4-tert-Butyl-benzyloxy)-benzylsulfanyl]-2-methyl-phenoxy}-acetic acid (compound 34)
The title compound was prepared in the manner analogous to Example 1 using 34C. mp 135-137° C.; IR (KBr) cm−1: 2961, 2908, 1751, 1495, 1233, 1194; 400 MHz 1H NMR (DMSO-d6): δ 13.00 (br(s), 1H), 7.27-7.38 (m, 4H), 7.02-7.17 (m, 4H), 6.82-6.88 (m, 2H), 6.69 (d, 1H, J=8.5 Hz), 4.96 (s, 2H), 4.61 (s, 2H), 3.99 (s, 2H), 2.08 (s, 3H), 1.22 (s, 9H); MS m/z 451 (M+1). Anal. Calc'd for C27H30O4S: C, 71.97; H, 6.71; found: C, 71.66; H, 6.52.
Synthesis of {2-Methyl-4-[4-(4-trifluoromethoxy-benzyloxy)-benzylsulfanyl]-phenoxy}-acetic acid (compound 35)
Step 1. Preparation of [4-(4-Acetoxy-benzylsulfanyl)-2-methyl-phenoxy]-acetic acid methyl ester (compound 35A)
The title compound was prepared in the manner analogous to Example 1F using acetic acid 4-chloromethyl-phenyl ester and 2C. MS m/z 361 (M+1).
Step 2. Preparation [4-(4Hydroxy-benzylsulfanyl)-2-methyl-phenoxy]-acetic acid (compound 35B)
The title compound was prepared in the manner analogous to Example 1 using 35A. MS m/z 303 (M−1).
Step 3. Preparation of [4-(4-Hydroxy-benzylsulfanyl)-2-methyl-phenoxy]-acetic acid methyl ester (compound 35C)
To a solution of the product from Example 35B (0.43 g, 1.4 mmol) in 14 mL of 2,2-dimethoxy propane was added 1.4 mL of concentrated hydrochloric acid. The reaction mixture was stirred at room temperature for 18 hours. The mixture was concentrated and purified by flash chromatography to provide the title compound. MS m/z 317 (M−1).
Step 4. Preparation of {2-Methyl-4-[4(4-trifluoromethoxy-benzyloxy)-benzylsulfanyl]-phenoxy}-acetic acid methyl ester (compound 35D)
The title compound was prepared in the manner analogous to Example 1F using 1-bromomethyl-4-trifluoromethoxy-benzene and 35C. MS m/z 493 (M+1).
Step 5. Preparation of {2-Methyl-4-[4-(4-trifluoromethoxy-benzyloxy)-benzylsulfanyl]-phenoxy}-acetic acid (compound 35)
The title compound was prepared in the manner analogous to Example 1 using 35D. mp 141-142° C.; 400 MHz 1H NMR (DMSO-d6): δ 12.96 (br(s), 1H), 7.46-7.57 (m, 2H), 7.28-7.38 (m, 2H), 7.00-7.20 (m, 4H), 6.82-6.91 (m, 2H), 6.69 (d, 1H, J=8.6 Hz), 5.05 (s, 2H), 4.62 (s, 2H), 4.00 (s, 2H), 2.08 (s, 3H); MS m/z 477 (M−1). Anal. Calc'd for C24H21F3O3S: C, 60.25; H, 4.42; found: C, 59.92; H, 4.07.
Step 1. Preparation of [4-(4-Acetoxy-benzylsulfanyl)-2-methyl-phenoxy]-acetic acid methyl ester (compound 35A)
The title compound was prepared in the manner analogous to Example 1F using acetic acid 4-chloromethyl-phenyl ester and 2C. MS m/z 361 (M+1).
Step 2. Preparation [4-(4Hydroxy-benzylsulfanyl)-2-methyl-phenoxy]-acetic acid (compound 35B)
The title compound was prepared in the manner analogous to Example 1 using 35A. MS m/z 303 (M−1).
Step 3. Preparation of [4-(4-Hydroxy-benzylsulfanyl)-2-methyl-phenoxy]-acetic acid methyl ester (compound 35C)
To a solution of the product from Example 35B (0.43 g, 1.4 mmol) in 14 mL of 2,2-dimethoxy propane was added 1.4 mL of concentrated hydrochloric acid. The reaction mixture was stirred at room temperature for 18 hours. The mixture was concentrated and purified by flash chromatography to provide the title compound. MS m/z 317 (M−1).
Step 4. Preparation of {2-Methyl-4-[4(4-trifluoromethoxy-benzyloxy)-benzylsulfanyl]-phenoxy}-acetic acid methyl ester (compound 35D)
The title compound was prepared in the manner analogous to Example 1F using 1-bromomethyl-4-trifluoromethoxy-benzene and 35C. MS m/z 493 (M+1).
Step 5. Preparation of {2-Methyl-4-[4-(4-trifluoromethoxy-benzyloxy)-benzylsulfanyl]-phenoxy}-acetic acid (compound 35)
The title compound was prepared in the manner analogous to Example 1 using 35D. mp 141-142° C.; 400 MHz 1H NMR (DMSO-d6): δ 12.96 (br(s), 1H), 7.46-7.57 (m, 2H), 7.28-7.38 (m, 2H), 7.00-7.20 (m, 4H), 6.82-6.91 (m, 2H), 6.69 (d, 1H, J=8.6 Hz), 5.05 (s, 2H), 4.62 (s, 2H), 4.00 (s, 2H), 2.08 (s, 3H); MS m/z 477 (M−1). Anal. Calc'd for C24H21F3O3S: C, 60.25; H, 4.42; found: C, 59.92; H, 4.07.
Synthesis of {6-Methyl-8-[4-(5-trifluoromethyl-pyridine-2-yl)-benzylsulfanyl]-chroman-5-yloxy}-acetic acid (compound 36)
Step 1. {6-Methyl-8-[4-(5-trifluoromethyl-pyridine-2-yl)-benzylsulfanyl]-chroman-5-yloxy}-acetic acid methyl ester (compound 36A)
The title compound was prepared in the manner analogous to Example 1F using (8-mercapto-6-methyl-chroman-5-yloxy)-acetic acid methyl ester and 18B. MS m/z 504 (M+1).
Step 2. {6-Methyl-8-[4-(5-trifluoromethyl-pyridine-2-yl)-benzylsulfanyl]-chroman-5-yloxy}-acetic acid methyl ester (compound 36)
The title compound was prepared in the manner analogous to Example 1 using 36A. mp 156-157° C.; IR (KBr) cm−1: 2928, 1731, 1710, 1603, 1329, 1113, 1082; 400 MHz 1H NMR (DMSO-d6): δ 12.84 (br(s), 1H), 8.97 (s, 1H), 8.22 (dd, 1H, J=8.4, 2.0 Hz), 8.12 (d, 1H, J=8.4 Hz), 8.04 (d, 2H, J=8.2 Hz), 7.42 (d, 2H, J=8.2 Hz), 6.88 (s, 1H), 4.29 (s, 2H), 4.05-4.14 (m, 4H), 2.64 (t, 2H, J=6.3 Hz), 2.04 (s, 3H), 1.81 (pentet, 2H); MS m/z 490 (M+1). Anal. Calc'd for C25H22F3NO4S: C, 61.34; H, 4.53; N, 2.86; found: C, 60.96; H, 4.48; N, 2.79.
Step 1. {6-Methyl-8-[4-(5-trifluoromethyl-pyridine-2-yl)-benzylsulfanyl]-chroman-5-yloxy}-acetic acid methyl ester (compound 36A)
The title compound was prepared in the manner analogous to Example 1F using (8-mercapto-6-methyl-chroman-5-yloxy)-acetic acid methyl ester and 18B. MS m/z 504 (M+1).
Step 2. {6-Methyl-8-[4-(5-trifluoromethyl-pyridine-2-yl)-benzylsulfanyl]-chroman-5-yloxy}-acetic acid methyl ester (compound 36)
The title compound was prepared in the manner analogous to Example 1 using 36A. mp 156-157° C.; IR (KBr) cm−1: 2928, 1731, 1710, 1603, 1329, 1113, 1082; 400 MHz 1H NMR (DMSO-d6): δ 12.84 (br(s), 1H), 8.97 (s, 1H), 8.22 (dd, 1H, J=8.4, 2.0 Hz), 8.12 (d, 1H, J=8.4 Hz), 8.04 (d, 2H, J=8.2 Hz), 7.42 (d, 2H, J=8.2 Hz), 6.88 (s, 1H), 4.29 (s, 2H), 4.05-4.14 (m, 4H), 2.64 (t, 2H, J=6.3 Hz), 2.04 (s, 3H), 1.81 (pentet, 2H); MS m/z 490 (M+1). Anal. Calc'd for C25H22F3NO4S: C, 61.34; H, 4.53; N, 2.86; found: C, 60.96; H, 4.48; N, 2.79.
Synthesis of {5-Chloro-2-methyl-4-[4-(5-trifluoromethyl-pyridin-2-yl)-benzylsulfanyl]-phenoxy}-acetic acid (compound 37)
Step 1. Preparation of {5-Chloro-2-methyl-4-[4-(5-trifluoromethyl-pyridin-2-yl)-benzylsulfanyl]-phenoxy}-acetic acid methyl ester (compound 37A)
Compound 37A was prepared in the manner analogous to Example 1F using the products from Example 18B and Example 20C. MS m/z 482 (M+).
Step 2. Preparation of {5-chloro-2-methyl-4-[4-(5-trifluoromethyl-pyridin-2-yl)-benzylsulfanyl]-phenoxy}-acetic acid (compound 37)
The title compound was prepared in the manner analogous to Example 1 using 37A. IR cm−1: 1708, 1122; 400 MHz 1H NMR (DMSO-d6) δ 8.97 (s, 1H), 8.22 (d, 1H, J=8.3 Hz), 8.12 (d, 1H, J=8.3 Hz), 8.04 (d, 2H, J=8.3 Hz), 7.43 (d, 2H, J=8.3 Hz), 7.25 (s, 1H), 6.94 (s, 1H), 4.70 (s, 2H), 4.21 (s, 2H), 2.06 (s, 3H). Anal. Calc'd for C22H17ClF3NO3S; C, 56.47, H, 3.66, N 2.99; found: C, 56.48, H, 3.28, N 3.04.
Step 1. Preparation of {5-Chloro-2-methyl-4-[4-(5-trifluoromethyl-pyridin-2-yl)-benzylsulfanyl]-phenoxy}-acetic acid methyl ester (compound 37A)
Compound 37A was prepared in the manner analogous to Example 1F using the products from Example 18B and Example 20C. MS m/z 482 (M+).
Step 2. Preparation of {5-chloro-2-methyl-4-[4-(5-trifluoromethyl-pyridin-2-yl)-benzylsulfanyl]-phenoxy}-acetic acid (compound 37)
The title compound was prepared in the manner analogous to Example 1 using 37A. IR cm−1: 1708, 1122; 400 MHz 1H NMR (DMSO-d6) δ 8.97 (s, 1H), 8.22 (d, 1H, J=8.3 Hz), 8.12 (d, 1H, J=8.3 Hz), 8.04 (d, 2H, J=8.3 Hz), 7.43 (d, 2H, J=8.3 Hz), 7.25 (s, 1H), 6.94 (s, 1H), 4.70 (s, 2H), 4.21 (s, 2H), 2.06 (s, 3H). Anal. Calc'd for C22H17ClF3NO3S; C, 56.47, H, 3.66, N 2.99; found: C, 56.48, H, 3.28, N 3.04.
Synthesis of [5-hydroxy-2-methyl-4-(4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxyl-acetic acid (compound 38)
A solution of [5-Methoxy-2-methyl-4-(4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid, prepared according to Example 4,(1.0 g, 2.2 mmol) in 75 mL DCM at 0° C. was treated with dropwise addition of BBr3 (5.5 mL of a 1.0 M solution in DCM). After 30 minutes, the reaction was carefully quenched with 50% NH4OH. The reaction was then acidified to pH 1 with conc. HCl, and extracted with EtOAc. The organic layer was dried (Na2SO4) and concentrated in vacuo. The crude reaction mixture was then taken up in MeOH, followed by addition of 50 μL H2SO4, and then refluxing for 3 hours. The reaction was then diluted with EtOAc, washed 1×50 mL water, dried (Na2SO4), and the reaction concentrated in vacuo. The resulting ester was purified by recrystallization from EtOAC/Hexanes. The ester was then saponified in the same manner as described for Example 1, to give the title compound in 37% overall yield. IR cm−1:3408, 1752, 1323; 400 MHz 1H NMR (DMSO-d6) δ 9.56 (s, 1H), 7.82 (d, 2H, J=8.4 Hz), 7.74 (d, 2H, J=8.4 Hz), 7.31 (d, 2H, J=8.4 Hz), 6.92 (s, 1H), 6.30 (s, 1H), 4.55 (s, 2H), 4.00 (s, 2H), 1.95 (s, 3H). Anal. Calc'd for C23H19F3NO4S.0.1 H2O C, 61.35; H, 4.30; found: C, 61.08; H, 3.92.
Synthesis of [5-Methoxy-2-methyl-4(3-methyl-4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid (compound 39)
Step 1. Preparation of (3-Methyl-4′-trifluoromethyl-biphenyl-4-yl)-methanol (compound 39A)
The title compound was prepared in the manner analogous to Example 3A using (4-bromo-2-methyl-phenyl)-methanol and 4-(trifluoromethyl)benzeneboronic acid. MS m/z 249 (M-OH).
Step 2. Preparation of 4chloromethyl-3-methyl-4′-trifluoromethyl-biphenyl (compound 39B)
The title compound was prepared in the manner analogous to Example 3B using 39A. 400 MHz 1H NMR (DMSO-d6) δ 7.84 (d, 2H, J=8.3 Hz), 7.76 (d, 2H, J=8.3 Hz), 7.57 (s, 1H), 7.48 (m, 2H), 4.80 (s, 2H), 2.41 (s, 3H).
Step 3. Preparation of [5-Methoxy-2-methyl-4-(3-methyl-4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid methyl ester (compound 39C)
The title compound was prepared in the manner analogous to Example 1F using 1D and 39B. MS m/z 491 (M+1).
Step 4. Preparation of [5-Methoxy-2-methyl-4-(methyl-4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid (compound 39)
The title compound was prepared in the manner analogous to Example 1 using 39C. IR cm−1: 1740, 1322; 400 MHz 1H NMR (DMSO-d6) δ 7.81 (d, 2H, J=8.1 Hz), 7.73 (d, 2H, J=8.1 Hz), 7.51 (s, 1H), 7.39 (d, 1H, J=7.8 Hz), 7.16 (d, 1H, J=7.8 Hz), 7.04 (s, 1H), 6.53 (s, 1H), 4.71 (s, 2H), 4.00 (s, 2H), 3.73 (s, 3H), 2.39 (s, 3H), 2.02 (s, 3H); MS m/z 477 (M+1). Anal. Calc'd for C25H23F3O4S.0.1 H2O; C, 62.78; H, 4.89; found: C, 62.57; H, 4.82.
Step 1. Preparation of (3-Methyl-4′-trifluoromethyl-biphenyl-4-yl)-methanol (compound 39A)
The title compound was prepared in the manner analogous to Example 3A using (4-bromo-2-methyl-phenyl)-methanol and 4-(trifluoromethyl)benzeneboronic acid. MS m/z 249 (M-OH).
Step 2. Preparation of 4chloromethyl-3-methyl-4′-trifluoromethyl-biphenyl (compound 39B)
The title compound was prepared in the manner analogous to Example 3B using 39A. 400 MHz 1H NMR (DMSO-d6) δ 7.84 (d, 2H, J=8.3 Hz), 7.76 (d, 2H, J=8.3 Hz), 7.57 (s, 1H), 7.48 (m, 2H), 4.80 (s, 2H), 2.41 (s, 3H).
Step 3. Preparation of [5-Methoxy-2-methyl-4-(3-methyl-4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid methyl ester (compound 39C)
The title compound was prepared in the manner analogous to Example 1F using 1D and 39B. MS m/z 491 (M+1).
Step 4. Preparation of [5-Methoxy-2-methyl-4-(methyl-4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid (compound 39)
The title compound was prepared in the manner analogous to Example 1 using 39C. IR cm−1: 1740, 1322; 400 MHz 1H NMR (DMSO-d6) δ 7.81 (d, 2H, J=8.1 Hz), 7.73 (d, 2H, J=8.1 Hz), 7.51 (s, 1H), 7.39 (d, 1H, J=7.8 Hz), 7.16 (d, 1H, J=7.8 Hz), 7.04 (s, 1H), 6.53 (s, 1H), 4.71 (s, 2H), 4.00 (s, 2H), 3.73 (s, 3H), 2.39 (s, 3H), 2.02 (s, 3H); MS m/z 477 (M+1). Anal. Calc'd for C25H23F3O4S.0.1 H2O; C, 62.78; H, 4.89; found: C, 62.57; H, 4.82.
Synthesis of {7-[4-(4-trifluoromethyl-benzyl)-benzylsulfanyl]-indan-4yloxy}-acetic acid (compound 40)
Step 1. Preparation of 4-(4-trifluoromethyl-benzyl)-benzoic acid methyl ester (compound 40A)
A solution of 1-bromo-4-trifluoromethyl-benzene (10.0 g, 44.4 mmol) in THF at −78° C. was treated with dropwise addition of n-butyl lithium (33.3 mL of a 1.6 M solution in hexanes). After 20 minutes, 4-formyl-benzoic acid methyl ester in 50 mL THF was added. The reaction was allowed to come to room temperature and after 1 H, quenched with sat. NH4Cl. The reaction was then concentrated in vacuo, taken up in EtOAc, and washed with 2 M HCl (1×100 mL), brine (1×100 mL), dried (Na2SO4) and the solvent removed in vacuo to give the alcohol intermediate. Purification by flash column chromatography (gradient elution: 5% EtOAc/hexane to 40% EtOAc/hexane) gave 6.2 g of the alcohol intermediate. 4.0 g (12.9 mmol) of the intermediate was then hydrogenated in EtOAc using 0.5 g of 10% Pd(OH)2/C as catalyst. Filtration through Celite®, and concentration in vacuo gave the title compound (3.60 g, 95%). MS m/z 295 (M+1).
Step 2. Preparation of [4-(4-trifluoromethyl-benzyl)-phenyl]-methanol (compound 40B)
A solution of 40A (3.6 g, 12.2 mmol) in 75 mL THF at room temperature was treated portionwise with lithium aluminum hydride (0.97 g, 25.6 mmol). After 1 hour, the reaction mixture was carefully quenched with sat. NH4Cl. The reaction mixture was then extracted with EtOAc, and the organic layer washed with 2 M HCl (1×50 mL), brine (1×50 mL), dried (Na2SO4) and the solvent removed in vacuo. Purification by flash column chromatography (gradient elution: 5% EtOAc/hexane to 40% EtOAc/hexane) gave the title compound (2.8 g, 86%). MS m/z 265 (M−1).
Step 3. Preparation of Chloro-[4-(4-trifluoromethyl-benzyl)-phenyl]-methane (compound 40C)
The title compound was prepared in a similar manner as described for 3B using 40B and thionyl chloride. 400 MHz 1H NMR (DMSO-d6) 7.59 (d, 2H, J=8.4 Hz), 7.40 (d, 2H, J=8.4 Hz), 7.31 (d, 2H, J=8.4 Hz), 7.20 (d, 2H, J=8.4 Hz), 4.67 (s, 2H), 3.99 (s, 2H).
Step 4. Preparation of {7-[4-(4-Trifluoromethyl-benzyl)-benzylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 40D)
The title compound was prepared in the manner analogous to Example 1F using the product from Example 12C and Example 40C. MS m/z 487 (M+1).
Step 5. Preparation of {7-[4-(4-trifluoromethyl-benzyl)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 40)
The title compound was prepared in the manner analogous to Example 1 using 40D. IR cm−1: 1745, 1704, 1325; 400 MHz 1H NMR (DMSO-d6) δ 7.58 (d, 2H, J=8.1 Hz), 7.37 (d, 2H, J=8.1 Hz), 7.09 (s, 2H), 7.05 (d, 2H, J=8.3 Hz), 6.55 (d, 1H, J=8.3 Hz), 4.60 (s, 2H), 3.94 (s, 4H), 2.71 (t, 2H, J=8.3 Hz), 2.58 (t, 2H, J=8.3 Hz), 1.81 (m, 2H); MS m/z 473 (M+1). Anal. Calc'd for C26H23F3O3S.0.1 H2O, C, 65.84; H, 4.93; found: C, 65.58; H, 4.96.
Step 1. Preparation of 4-(4-trifluoromethyl-benzyl)-benzoic acid methyl ester (compound 40A)
A solution of 1-bromo-4-trifluoromethyl-benzene (10.0 g, 44.4 mmol) in THF at −78° C. was treated with dropwise addition of n-butyl lithium (33.3 mL of a 1.6 M solution in hexanes). After 20 minutes, 4-formyl-benzoic acid methyl ester in 50 mL THF was added. The reaction was allowed to come to room temperature and after 1 H, quenched with sat. NH4Cl. The reaction was then concentrated in vacuo, taken up in EtOAc, and washed with 2 M HCl (1×100 mL), brine (1×100 mL), dried (Na2SO4) and the solvent removed in vacuo to give the alcohol intermediate. Purification by flash column chromatography (gradient elution: 5% EtOAc/hexane to 40% EtOAc/hexane) gave 6.2 g of the alcohol intermediate. 4.0 g (12.9 mmol) of the intermediate was then hydrogenated in EtOAc using 0.5 g of 10% Pd(OH)2/C as catalyst. Filtration through Celite®, and concentration in vacuo gave the title compound (3.60 g, 95%). MS m/z 295 (M+1).
Step 2. Preparation of [4-(4-trifluoromethyl-benzyl)-phenyl]-methanol (compound 40B)
A solution of 40A (3.6 g, 12.2 mmol) in 75 mL THF at room temperature was treated portionwise with lithium aluminum hydride (0.97 g, 25.6 mmol). After 1 hour, the reaction mixture was carefully quenched with sat. NH4Cl. The reaction mixture was then extracted with EtOAc, and the organic layer washed with 2 M HCl (1×50 mL), brine (1×50 mL), dried (Na2SO4) and the solvent removed in vacuo. Purification by flash column chromatography (gradient elution: 5% EtOAc/hexane to 40% EtOAc/hexane) gave the title compound (2.8 g, 86%). MS m/z 265 (M−1).
Step 3. Preparation of Chloro-[4-(4-trifluoromethyl-benzyl)-phenyl]-methane (compound 40C)
The title compound was prepared in a similar manner as described for 3B using 40B and thionyl chloride. 400 MHz 1H NMR (DMSO-d6) 7.59 (d, 2H, J=8.4 Hz), 7.40 (d, 2H, J=8.4 Hz), 7.31 (d, 2H, J=8.4 Hz), 7.20 (d, 2H, J=8.4 Hz), 4.67 (s, 2H), 3.99 (s, 2H).
Step 4. Preparation of {7-[4-(4-Trifluoromethyl-benzyl)-benzylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 40D)
The title compound was prepared in the manner analogous to Example 1F using the product from Example 12C and Example 40C. MS m/z 487 (M+1).
Step 5. Preparation of {7-[4-(4-trifluoromethyl-benzyl)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 40)
The title compound was prepared in the manner analogous to Example 1 using 40D. IR cm−1: 1745, 1704, 1325; 400 MHz 1H NMR (DMSO-d6) δ 7.58 (d, 2H, J=8.1 Hz), 7.37 (d, 2H, J=8.1 Hz), 7.09 (s, 2H), 7.05 (d, 2H, J=8.3 Hz), 6.55 (d, 1H, J=8.3 Hz), 4.60 (s, 2H), 3.94 (s, 4H), 2.71 (t, 2H, J=8.3 Hz), 2.58 (t, 2H, J=8.3 Hz), 1.81 (m, 2H); MS m/z 473 (M+1). Anal. Calc'd for C26H23F3O3S.0.1 H2O, C, 65.84; H, 4.93; found: C, 65.58; H, 4.96.
Synthesis of {4-[5-(4-Chloro-phenyl)-isoxazol-3-ylmethylsulfanyl]-5-methoxy-2-methyl-phenoxy}-acetic acid (compound 41)
Step 1. Preparation of {4-[5-(4-Chloro-phenyl)-isoxazol-3-ylmethylsulfanyl]-5-methoxy-2-methyl-phenoxy}-acetic acid methyl ester (compound 41A)
The title compound was prepared in the manner analogous to Example 1F using 1D and 3-chloromethyl-5-(4-chloro-phenyl)-isoxazole. MS m/z 434 (M+1).
Step 2. Preparation of {4-[5-(4-Chloro-phenyl)-isoxazol-3-ylmethylsulfanyl]-5-methoxy-2-methyl-phenoxy}-acetic acid (compound 41)
The title compound was prepared in the manner analogous to Example 1 using 41A. IR cm−1: 1747, 1432; 400 MHz 1H NMR (DMSO-d6) δ 7.81 (d, 2H, J=8.8 Hz), 7.53 (d, 2H, J=8.8 Hz), 7.05 (s, 1H), 6.92 (s, 1H), 6.52 (s, 1H), 4.69 (s, 2H), 4.01 (s, 2H), 3.71 (s, 3H), 2.00 (s, 3H); MS m/z 420 (M+1). Anal. Calc'd for C20H18ClNO5S, C 57.21; H, 4.32 N, 3.34; found: C, 56.84; H, 4.62, N, 2.96.
Step 1. Preparation of {4-[5-(4-Chloro-phenyl)-isoxazol-3-ylmethylsulfanyl]-5-methoxy-2-methyl-phenoxy}-acetic acid methyl ester (compound 41A)
The title compound was prepared in the manner analogous to Example 1F using 1D and 3-chloromethyl-5-(4-chloro-phenyl)-isoxazole. MS m/z 434 (M+1).
Step 2. Preparation of {4-[5-(4-Chloro-phenyl)-isoxazol-3-ylmethylsulfanyl]-5-methoxy-2-methyl-phenoxy}-acetic acid (compound 41)
The title compound was prepared in the manner analogous to Example 1 using 41A. IR cm−1: 1747, 1432; 400 MHz 1H NMR (DMSO-d6) δ 7.81 (d, 2H, J=8.8 Hz), 7.53 (d, 2H, J=8.8 Hz), 7.05 (s, 1H), 6.92 (s, 1H), 6.52 (s, 1H), 4.69 (s, 2H), 4.01 (s, 2H), 3.71 (s, 3H), 2.00 (s, 3H); MS m/z 420 (M+1). Anal. Calc'd for C20H18ClNO5S, C 57.21; H, 4.32 N, 3.34; found: C, 56.84; H, 4.62, N, 2.96.
Synthesis of {2-Methyl-4-[5-(4-trifluoromethyl-phenyl)-isoxazol-3-ylmethylsulfanyl]-phenoxy}-acetic acid (compound 42)
Step 1. Preparation of 5-(4-Trifluoromethyl-phenyl)-isoxazole-3-carboxylic acid ethyl ester (compound 42A)
Sodium hydride (1.6 g, 63.7 mmol, 95%) was added to a solution of 1(4-trifluoromethyl-phenyl)-ethanone (10.0 g, 53.1 mmol) and oxalic acid diethyl ester (8.7 mL, 63.7 mmol) in 75 mL dry DMF at 0° C. The reaction was allowed to come to room temperature and then heated to 45° C. for 45 minutes. The reaction was then cooled, concentrated in vacuo, and the residue taken up in EtOAc. The organic layer was then washed with 2 M HCl (1×100 mL), dried (Na2SO4) and the solvent removed in vacuo. Purification by flash column chromatography (gradient elution: 5% EtOAc/hexane to 55% EtOAc/hexane) gave the intermediate 2,4-dioxo-4-(4-trifluoromethyl-phenyl)-butyric acid ethyl ester (12.2 g, 80%) which was then taken up in EtOH and refluxed in the presence of hydroxyl amine hydrochloride (10.2 g, 132.3 mmol) for 3H. The reaction was then cooled, diluted with EtOAc, washed with dilute NaHCO3, brine, dried (Na2SO4), and concentrated in vacuo. Recrystallization from EtOAc/hexane gave 5.2 g of the title compound. MS m/z 286 (M+1).
Step 2. Preparation of [5-(4-Trifluoromethyl-phenyl)-isoxazol-3-yl]-methanol (compound 42B)
The title compound was prepared in a manner analogous to Example 40B using 42A. MS m/z 244 (M+1).
Step 3. Preparation of 3-chloromethyl-5-(4-trifluoromethyl-phenyl)-isoxazole (compound 42C)
The title compound was prepared in a manner analogous to Example 3B using 42B. MS m/z 262 (M+1).
Step 4. Preparation of {2-methyl-4-[5-(4-trifluoromethyl-phenyl)-isoxazol-3-ylmethylsulfanyl]-phenoxy}-acetic acid methyl ester (compound 42
The title compound was prepared in a manner analogous to Example 1F using 42C and 2C. MS m/z 438 (M+1).
Step 5. Preparation of {2Methyl-4-[5-(4-trifluoromethyl-phenyl)-isoxazol-3-ylmethylsulfanyl]-phenoxy}-acetic acid (compound 42)
The title compound was prepared in a manner analogous to Example 1 using 42D. IR cm−1:1746, 1326; 400 MHz 1H NMR (DMSO-d6) δ 12.97 (br(s), 1H), 8.02 (d, 2H, J=8.0 Hz), 7.83 (d, 2H, J=8.0 Hz), 7.20 (s, 1H), 7.15 (s, 2H), 6.71 (d, 1H, J=8.5 Hz), 4.63 (s, 2H), 4.14 (s, 2H), 2.08 (s, 3H); MS m/z 424 (M+1). Anal. Calc'd for C20H16F3NO4S C, 56.73; H, 3.81; N, 3.31 found: C, 56.59; H, 3.58; N, 3.22.
Step 1. Preparation of 5-(4-Trifluoromethyl-phenyl)-isoxazole-3-carboxylic acid ethyl ester (compound 42A)
Sodium hydride (1.6 g, 63.7 mmol, 95%) was added to a solution of 1(4-trifluoromethyl-phenyl)-ethanone (10.0 g, 53.1 mmol) and oxalic acid diethyl ester (8.7 mL, 63.7 mmol) in 75 mL dry DMF at 0° C. The reaction was allowed to come to room temperature and then heated to 45° C. for 45 minutes. The reaction was then cooled, concentrated in vacuo, and the residue taken up in EtOAc. The organic layer was then washed with 2 M HCl (1×100 mL), dried (Na2SO4) and the solvent removed in vacuo. Purification by flash column chromatography (gradient elution: 5% EtOAc/hexane to 55% EtOAc/hexane) gave the intermediate 2,4-dioxo-4-(4-trifluoromethyl-phenyl)-butyric acid ethyl ester (12.2 g, 80%) which was then taken up in EtOH and refluxed in the presence of hydroxyl amine hydrochloride (10.2 g, 132.3 mmol) for 3H. The reaction was then cooled, diluted with EtOAc, washed with dilute NaHCO3, brine, dried (Na2SO4), and concentrated in vacuo. Recrystallization from EtOAc/hexane gave 5.2 g of the title compound. MS m/z 286 (M+1).
Step 2. Preparation of [5-(4-Trifluoromethyl-phenyl)-isoxazol-3-yl]-methanol (compound 42B)
The title compound was prepared in a manner analogous to Example 40B using 42A. MS m/z 244 (M+1).
Step 3. Preparation of 3-chloromethyl-5-(4-trifluoromethyl-phenyl)-isoxazole (compound 42C)
The title compound was prepared in a manner analogous to Example 3B using 42B. MS m/z 262 (M+1).
Step 4. Preparation of {2-methyl-4-[5-(4-trifluoromethyl-phenyl)-isoxazol-3-ylmethylsulfanyl]-phenoxy}-acetic acid methyl ester (compound 42
The title compound was prepared in a manner analogous to Example 1F using 42C and 2C. MS m/z 438 (M+1).
Step 5. Preparation of {2Methyl-4-[5-(4-trifluoromethyl-phenyl)-isoxazol-3-ylmethylsulfanyl]-phenoxy}-acetic acid (compound 42)
The title compound was prepared in a manner analogous to Example 1 using 42D. IR cm−1:1746, 1326; 400 MHz 1H NMR (DMSO-d6) δ 12.97 (br(s), 1H), 8.02 (d, 2H, J=8.0 Hz), 7.83 (d, 2H, J=8.0 Hz), 7.20 (s, 1H), 7.15 (s, 2H), 6.71 (d, 1H, J=8.5 Hz), 4.63 (s, 2H), 4.14 (s, 2H), 2.08 (s, 3H); MS m/z 424 (M+1). Anal. Calc'd for C20H16F3NO4S C, 56.73; H, 3.81; N, 3.31 found: C, 56.59; H, 3.58; N, 3.22.
Synthesis of {5-Methoxy-2methyl-4-[5-(4-trifluoromethyl-phenyl)-isoxazol-3-ylmethylsulfanyl]-phenoxy}-acetic acid (compound 43)
Step 1. Preparation of {5-Methoxy-2-methyl-4-[5-(4-trifluoromethyl-phenyl)-isoxazol-3-ylmethylsulfanyl]-phenoxy}-acetic acid methyl ester (compound 43A)
The title compound was prepared in a manner analogous to Example 1F using 42C and 1D. MS m/z 468 (M+1).
Step 2. Preparation of {5-Methoxy-2-methyl-4-15-(4-trifluoromethyl-phenyl)-isoxazol-3-ylmethylsulfanyl]-phenoxy}-acetic acid (compound 43)
The title compound was prepared in a manner analogous to Example 1 using 43A. IR cm−1: 1745, 1322; 400 MHz 1H NMR (DMSO-d6) δ 12.96 (br(s), 1H), 8.01 (d, 2H, J=8.3 Hz), 7.83 (d, 2H, J=8.3 Hz), 7.07 (s, 2H), 6.52 (s, 1H), 4.70 (s, 2H), 4.03 (s, 2H), 3.71 (s, 3H), 1.99 (s, 3H); MS m/z 454 (M+1). Anal. Calc'd for C21H18F3NO3S. 0.1H2O C, 54.46; H, 3.92; N, 3.01 found: C, 54.54; H, 3.74; N, 2.93.
Step 1. Preparation of {5-Methoxy-2-methyl-4-[5-(4-trifluoromethyl-phenyl)-isoxazol-3-ylmethylsulfanyl]-phenoxy}-acetic acid methyl ester (compound 43A)
The title compound was prepared in a manner analogous to Example 1F using 42C and 1D. MS m/z 468 (M+1).
Step 2. Preparation of {5-Methoxy-2-methyl-4-15-(4-trifluoromethyl-phenyl)-isoxazol-3-ylmethylsulfanyl]-phenoxy}-acetic acid (compound 43)
The title compound was prepared in a manner analogous to Example 1 using 43A. IR cm−1: 1745, 1322; 400 MHz 1H NMR (DMSO-d6) δ 12.96 (br(s), 1H), 8.01 (d, 2H, J=8.3 Hz), 7.83 (d, 2H, J=8.3 Hz), 7.07 (s, 2H), 6.52 (s, 1H), 4.70 (s, 2H), 4.03 (s, 2H), 3.71 (s, 3H), 1.99 (s, 3H); MS m/z 454 (M+1). Anal. Calc'd for C21H18F3NO3S. 0.1H2O C, 54.46; H, 3.92; N, 3.01 found: C, 54.54; H, 3.74; N, 2.93.
Synthesis of {7-[5-(4-Trifluoromethyl-phenyl)-isoxazol-3-ylmethylsulfanyl]-indan-4-yloxy}-acetic acid (compound 44)
Step 1. Preparation of {7-[5-(4-Trifluoromethyl-phenyl)-isoxazol-3-ylmethylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 44A)
The title compound was prepared in a manner analogous to Example 1F using 42C and 12C. MS m/z 464 (M+1).
Step 2. Preparation of {7-[5-(4-Trifluoromethyl-phenyl)-isoxazol-3-ylmethylsulfanyl]-indan-4-yloxy}-acetic acid (compound 44)
The title compound was prepared in a manner analogous to Example 1 using 44A. 400 MHz 1H NMR (DMSO-d6) δ 12.94 (br(s), 1H), 8.01 (d, 2H, J=8.4 Hz), 7.83 (d, 2H, J=8.4 Hz), 7.09 (s, 2H), 6.58 (d, 1H, J=8.6 Hz), 4.62 (s, 2H), 4.08 (s, 2H), 2.77 (m, 4H), 1.89 (m, 2H); MS m/z 450 (M+1). Anal. Calc'd for C22H18F3NO4S C, 58.79; H, 4.04; N, 3.12 found: C, 58.59; H, 3.80; N, 3.01.
Step 1. Preparation of {7-[5-(4-Trifluoromethyl-phenyl)-isoxazol-3-ylmethylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 44A)
The title compound was prepared in a manner analogous to Example 1F using 42C and 12C. MS m/z 464 (M+1).
Step 2. Preparation of {7-[5-(4-Trifluoromethyl-phenyl)-isoxazol-3-ylmethylsulfanyl]-indan-4-yloxy}-acetic acid (compound 44)
The title compound was prepared in a manner analogous to Example 1 using 44A. 400 MHz 1H NMR (DMSO-d6) δ 12.94 (br(s), 1H), 8.01 (d, 2H, J=8.4 Hz), 7.83 (d, 2H, J=8.4 Hz), 7.09 (s, 2H), 6.58 (d, 1H, J=8.6 Hz), 4.62 (s, 2H), 4.08 (s, 2H), 2.77 (m, 4H), 1.89 (m, 2H); MS m/z 450 (M+1). Anal. Calc'd for C22H18F3NO4S C, 58.79; H, 4.04; N, 3.12 found: C, 58.59; H, 3.80; N, 3.01.
Synthesis of {2-Methyl-4-[3-(4-trifluoromethyl-phenyl)-isoxazol-5-ylmethylsulfanyl]-phenoxy}-acetic acid (compound 45)
Step 1. Preparation of 5-chloromethyl-3-(4-trifluoromethyl-phenyl)-isoxazole (compound 45A)
A solution of 4-trifluoromethyl-benzaldehyde oxime (8.9 g, 47.1 mmol) in 100 mL DCM was added to a rapidly stirred solution of propargyl chloride (47.1 mmol), triethyl amine (4.71 mmol) and 91 mL of commercial bleach (6.5% by weight) all in 50 mL DCM at 0° C. After 1 hour the layers were separated and the organic layer dried (Na2SO4), and concentrated in vacuo. Purification by flash column chromatography (gradient elution: 5% EtOAc/hexane to 25% EtOAc/hexane) gave the title compound (2.9 g, 23%) MS m/z 262 (M+1).
Step 2. Preparation of {2-Methyl-4-[3-(4-trifluoromethyl-phenyl)-isoxazol-5-ylmethylsulfanyl]-phenoxy}-acetic acid methyl ester (compound 45B)
The title compound was prepared in a manner analogous to Example 1F using 45A and 2C. MS m/z 438 (M+1).
Step 3. Preparation of {2-Methyl-4-[3-(4-trifluoromethyl-phenyl)-isoxazol-5-ylmethylsulfanyl]-3-phenoxy}-acetic acid (compound 45)
The title compound was prepared in a manner analogous to Example 1 using 45B. IR cm−1: 1747; 400 MHz 1H NMR (DMSO-d6) δ 12.97 (br(s), 1H), 7.99 (d, 2H, J=8.0 Hz), 7.81 (d, 2H, J=8.0 Hz), 7.21 (s, 1H), 7.16 (d, 1H, J=8.5 Hz), 6.86 (s, 1H), 6.74 (d, 1H, J=8.5 Hz), 4.63 (s, 2H), 4.30 (s, 2H), 2.09 (s, 3H); MS m/z 424 (M+1).
Step 1. Preparation of 5-chloromethyl-3-(4-trifluoromethyl-phenyl)-isoxazole (compound 45A)
A solution of 4-trifluoromethyl-benzaldehyde oxime (8.9 g, 47.1 mmol) in 100 mL DCM was added to a rapidly stirred solution of propargyl chloride (47.1 mmol), triethyl amine (4.71 mmol) and 91 mL of commercial bleach (6.5% by weight) all in 50 mL DCM at 0° C. After 1 hour the layers were separated and the organic layer dried (Na2SO4), and concentrated in vacuo. Purification by flash column chromatography (gradient elution: 5% EtOAc/hexane to 25% EtOAc/hexane) gave the title compound (2.9 g, 23%) MS m/z 262 (M+1).
Step 2. Preparation of {2-Methyl-4-[3-(4-trifluoromethyl-phenyl)-isoxazol-5-ylmethylsulfanyl]-phenoxy}-acetic acid methyl ester (compound 45B)
The title compound was prepared in a manner analogous to Example 1F using 45A and 2C. MS m/z 438 (M+1).
Step 3. Preparation of {2-Methyl-4-[3-(4-trifluoromethyl-phenyl)-isoxazol-5-ylmethylsulfanyl]-3-phenoxy}-acetic acid (compound 45)
The title compound was prepared in a manner analogous to Example 1 using 45B. IR cm−1: 1747; 400 MHz 1H NMR (DMSO-d6) δ 12.97 (br(s), 1H), 7.99 (d, 2H, J=8.0 Hz), 7.81 (d, 2H, J=8.0 Hz), 7.21 (s, 1H), 7.16 (d, 1H, J=8.5 Hz), 6.86 (s, 1H), 6.74 (d, 1H, J=8.5 Hz), 4.63 (s, 2H), 4.30 (s, 2H), 2.09 (s, 3H); MS m/z 424 (M+1).
Synthesis of {5-Methoxy-2-methyl-4-[3(4-trifluoromethyl-phenyl)-isoxazol-5-ylmethylsulfanyl]-phenoxy}-acetic acid (compound 46)
Step 1. Preparation of {5-Methoxy-2-methyl-4-[3-(4-trifluoromethyl-phenyl)-isoxazol-5-ylmethylsulfanyl]-phenoxy}-acetic acid methyl ester (compound 46A)
The title compound was prepared in a manner analogous to Example 1F using 45A and 1D. MS m/z 468 (M+1).
Step 2. Preparation of {5-Methoxy-2-methyl-4-[3-(4-trifluoromethyl-phenyl)-isoxazol-5-ylmethylsulfanyl]-phenoxy}-acetic acid (compound 46)
The title compound was prepared in a manner analogous to Example 1 using 46A. IR cm−1:1752, 1711; 400 MHz 1H NMR (DMSO-d6) δ 12.96 (br(s), 1H), 7.98 (d, 2H, J=8.1 Hz), 7.81 (d,2H, J=8.1 Hz), 7.09 (s, 1H), 6.80 (s, 1H), 6.54 (s, 1H), 4.71 (s, 2H), 4.18 (s, 2H), 3.79 (s, 3H), 2.00 (s, 3H); MS m/z 454 (M+1).
Step 1. Preparation of {5-Methoxy-2-methyl-4-[3-(4-trifluoromethyl-phenyl)-isoxazol-5-ylmethylsulfanyl]-phenoxy}-acetic acid methyl ester (compound 46A)
The title compound was prepared in a manner analogous to Example 1F using 45A and 1D. MS m/z 468 (M+1).
Step 2. Preparation of {5-Methoxy-2-methyl-4-[3-(4-trifluoromethyl-phenyl)-isoxazol-5-ylmethylsulfanyl]-phenoxy}-acetic acid (compound 46)
The title compound was prepared in a manner analogous to Example 1 using 46A. IR cm−1:1752, 1711; 400 MHz 1H NMR (DMSO-d6) δ 12.96 (br(s), 1H), 7.98 (d, 2H, J=8.1 Hz), 7.81 (d,2H, J=8.1 Hz), 7.09 (s, 1H), 6.80 (s, 1H), 6.54 (s, 1H), 4.71 (s, 2H), 4.18 (s, 2H), 3.79 (s, 3H), 2.00 (s, 3H); MS m/z 454 (M+1).
Synthesis of [2-Methyl-4-(4-phenoxy-benzylsulfanyl)-phenoxy]-acetic acid (compound 47)
Step 1. Preparation of [2-Methyl-4-(4-phenoxy-benzylsulfanyl)-phenoxy]-acetic acid methyl ester (compound 47A)
The title compound was prepared in the manner analogous to Example 1F using 1-chloromethyl-4-phenoxy-benzene and 2C. MS m/z 321 (M-methylacetate).
Step 2. Preparation of [2-Methyl-4-(4-phenoxy-benzylsulfanyl)-phenoxy]-acetic acid (compound 47)
The title compound was prepared in the manner analogous to Example 1 using 47A. 400 MHz 1H NMR (DMSO-d6) δ 12.95 (br(s), 1H), 7.33 (m, 2H), 7.22 (m, 2H), 7.07 (m, 3H), 6.93 (m, 2H), 6.87 (m, 2H), 6.71 (d, 1H, J=8.3 Hz), 4.62 (s, 2H), 4.04 (s, 2H), 2.09 (s, 3H). MS m/z 379 (M−1).
Step 1. Preparation of [2-Methyl-4-(4-phenoxy-benzylsulfanyl)-phenoxy]-acetic acid methyl ester (compound 47A)
The title compound was prepared in the manner analogous to Example 1F using 1-chloromethyl-4-phenoxy-benzene and 2C. MS m/z 321 (M-methylacetate).
Step 2. Preparation of [2-Methyl-4-(4-phenoxy-benzylsulfanyl)-phenoxy]-acetic acid (compound 47)
The title compound was prepared in the manner analogous to Example 1 using 47A. 400 MHz 1H NMR (DMSO-d6) δ 12.95 (br(s), 1H), 7.33 (m, 2H), 7.22 (m, 2H), 7.07 (m, 3H), 6.93 (m, 2H), 6.87 (m, 2H), 6.71 (d, 1H, J=8.3 Hz), 4.62 (s, 2H), 4.04 (s, 2H), 2.09 (s, 3H). MS m/z 379 (M−1).
Synthesis of [7-(4′-Trifluoromethyl-biphenyl-3-ylmethylsulfanyl)-indan-4-yloxy]-acetic acid (compound 48)
Step 1. Preparation of (4′-Trifluoromethyl-biphenyl-3-yl)-methanol (compound 48A)
The title compound was prepared in the manner analogous to Example 3A with 1-bromo-4-trifluoromethyl-benzene and 3-(hydroxymethyl)phenyl boronic acid. MS m/z 251 (M−1).
Step 2. Preparation of 3-Chloromethyl-4′-trifluoromethyl-biphenyl (compound 48B)
The title compound was prepared in the manner analogous to Example 3B using 48A. MS m/z 236 (M+1-Cl).
Step 3. Preparation of [7-(4′-Trifluoromethyl-biphenyl-3-ylmethylsulfanyl)-indan-4-yloxy]-acetic acid methyl ester (compound 48C)
The title compound was prepared in the manner analogous to Example 1F using 12C and 48B. MS m/z 473 (M+1).
Step 4. Preparation of [7-(4′-Trifluoromethyl-biphenyl-3-ylmethylsulfanyl)-indan-4-yloxy]-acetic acid (compound 48)
The title compound was prepared in the manner analogous to Example 1 using 48C. 400 MHz 1H NMR (DMSO-d6) δ 12.96 (br(s), 1H), 7.75 (d, 2H, J=8.3 Hz), 7.67 (d, 2H, J=8.1 Hz), 7.52 (m, 1H), 7.37 (t, 1H, J=7.6 Hz), 7.29 (m, 2H), 7.13 (d, 1H, J=8.5 Hz), 6.61 (d, 1H, J=8.5 Hz), 4.63 (s, 2H), 4.05 (s, 2H), 2.71 (t, 2H), 2.58 (t, 2H), 1.81 (m, 2H). MS m/z 459 (M+1).
Step 1. Preparation of (4′-Trifluoromethyl-biphenyl-3-yl)-methanol (compound 48A)
The title compound was prepared in the manner analogous to Example 3A with 1-bromo-4-trifluoromethyl-benzene and 3-(hydroxymethyl)phenyl boronic acid. MS m/z 251 (M−1).
Step 2. Preparation of 3-Chloromethyl-4′-trifluoromethyl-biphenyl (compound 48B)
The title compound was prepared in the manner analogous to Example 3B using 48A. MS m/z 236 (M+1-Cl).
Step 3. Preparation of [7-(4′-Trifluoromethyl-biphenyl-3-ylmethylsulfanyl)-indan-4-yloxy]-acetic acid methyl ester (compound 48C)
The title compound was prepared in the manner analogous to Example 1F using 12C and 48B. MS m/z 473 (M+1).
Step 4. Preparation of [7-(4′-Trifluoromethyl-biphenyl-3-ylmethylsulfanyl)-indan-4-yloxy]-acetic acid (compound 48)
The title compound was prepared in the manner analogous to Example 1 using 48C. 400 MHz 1H NMR (DMSO-d6) δ 12.96 (br(s), 1H), 7.75 (d, 2H, J=8.3 Hz), 7.67 (d, 2H, J=8.1 Hz), 7.52 (m, 1H), 7.37 (t, 1H, J=7.6 Hz), 7.29 (m, 2H), 7.13 (d, 1H, J=8.5 Hz), 6.61 (d, 1H, J=8.5 Hz), 4.63 (s, 2H), 4.05 (s, 2H), 2.71 (t, 2H), 2.58 (t, 2H), 1.81 (m, 2H). MS m/z 459 (M+1).
Synthesis of [5-Methoxy-2-methyl-4-(4′-trifluoromethyl-biphenyl-3-ylmethylsulfanyl)-phenoxy]-acetic acid (compound 49)
Step 1. Preparation of [5-Methoxy-2-methyl-4-(4′-trifluoromethyl-biphenyl-3-ylmethylsulfanyl)-phenoxy]-acetic acid methyl ester (compound 49A)
The title compound was prepared in the manner analogous to Example 1F using 48B and 1D. MS m/z 477 (M+1).
Step 2. Preparation of [5-Methoxy-2-methyl-4-(4′-trifluoromethyl-biphenyl-3-ylmethylsulfanyl)-phenoxy]-acetic acid (compound 49)
The title compound was prepared in the manner analogous to Example 1 using 49A. 400 MHz 1H NMR (DMSO-d6) δ 12.96 (br(s), 1H), 7.73 (d, 2H, J=9 Hz), 7.71 (d, 2H, J=9 Hz), 7.50 (m, 1H), 7.42 (m, 1H), 7.36 (t, 1H, J=7.8 Hz), 7.28 (m, 1H), 6.99 (s, 1H), 6.53 (s, 1H), 4.70 (s, 2H), 4.03 (s, 2H), 3.73 (s, 3H), 1.99 (s, 3H). MS m/z 463 (M+1).
Step 1. Preparation of [5-Methoxy-2-methyl-4-(4′-trifluoromethyl-biphenyl-3-ylmethylsulfanyl)-phenoxy]-acetic acid methyl ester (compound 49A)
The title compound was prepared in the manner analogous to Example 1F using 48B and 1D. MS m/z 477 (M+1).
Step 2. Preparation of [5-Methoxy-2-methyl-4-(4′-trifluoromethyl-biphenyl-3-ylmethylsulfanyl)-phenoxy]-acetic acid (compound 49)
The title compound was prepared in the manner analogous to Example 1 using 49A. 400 MHz 1H NMR (DMSO-d6) δ 12.96 (br(s), 1H), 7.73 (d, 2H, J=9 Hz), 7.71 (d, 2H, J=9 Hz), 7.50 (m, 1H), 7.42 (m, 1H), 7.36 (t, 1H, J=7.8 Hz), 7.28 (m, 1H), 6.99 (s, 1H), 6.53 (s, 1H), 4.70 (s, 2H), 4.03 (s, 2H), 3.73 (s, 3H), 1.99 (s, 3H). MS m/z 463 (M+1).
Synthesis of [5-Methoxy-2-methyl-4-(4′-trifluoromethyl-biphenyl-4-ylmethanesulfonyl)-phenoxy]-acetic acid (compound 50)
Step 1. Preparation of [5-Methoxy-2-methyl-4-(4′-trifluoromethyl-biphenyl-4-ylmethanesulfonyl)-phenoxy]-acetic acid methyl ester (compound 50A)
Example 4 (200 mg) was dissolved in 5 ml dichloromethane. Excess m-chloroperbenzoic acid (300 mg) was added and the reaction was allowed to stir 3 h. The solvent was removed under vacuum and the crude product was purified by MPLC. MS m/z 509 (M+1).
Step 2. Preparation of [5-Methoxy-2-methyl-4-(4′-trifluoromethyl-biphenyl-4-ylmethanesulfonyl)-phenoxy]-acetic acid (compound 50)
The title compound was prepared in the manner analogous to Example 1 using 50A. 400 MHz 1H NMR (DMSO-d6) δ 13.13 (br(s), 1H), 7.85 (d, 2H, J=8.1 Hz), 7.77 (d, 2H, J=8.3 Hz), 7.66 (d, 2H, J=8.6 Hz), 7.3 (m, 3H), 6.76 (s, 1H), 4.89 (s, 2H), 4.67 (s, 2H), 3.96 (s, 3H), 2.04 (s, 3H). MS m/z 495 (M+1).
Step 1. Preparation of [5-Methoxy-2-methyl-4-(4′-trifluoromethyl-biphenyl-4-ylmethanesulfonyl)-phenoxy]-acetic acid methyl ester (compound 50A)
Example 4 (200 mg) was dissolved in 5 ml dichloromethane. Excess m-chloroperbenzoic acid (300 mg) was added and the reaction was allowed to stir 3 h. The solvent was removed under vacuum and the crude product was purified by MPLC. MS m/z 509 (M+1).
Step 2. Preparation of [5-Methoxy-2-methyl-4-(4′-trifluoromethyl-biphenyl-4-ylmethanesulfonyl)-phenoxy]-acetic acid (compound 50)
The title compound was prepared in the manner analogous to Example 1 using 50A. 400 MHz 1H NMR (DMSO-d6) δ 13.13 (br(s), 1H), 7.85 (d, 2H, J=8.1 Hz), 7.77 (d, 2H, J=8.3 Hz), 7.66 (d, 2H, J=8.6 Hz), 7.3 (m, 3H), 6.76 (s, 1H), 4.89 (s, 2H), 4.67 (s, 2H), 3.96 (s, 3H), 2.04 (s, 3H). MS m/z 495 (M+1).
Synthesis of [5-Methoxy-2-methyl-4-(4′-trifluoromethyl-biphenyl-4-ylmethanesulfinyl)-phenoxy]-acetic acid (compound 51)
Step 1. Preparation of [5-Methoxy-2-methyl-4-(4′-trifluoromethyl-biphenyl-4-ylmethanesulfinyl)-phenoxy]-acetic acid methyl ester (compound 51A)
Example 4 (0.5 g, 1.0 mmol) was dissolved in 25 ml dichloromethane followed by the addition of 2-benzenesulfonyl-3-phenyl-oxaziridine (0.274, 1.04 mmol). The reaction was stirred 1 h. 10 ml water was added, the layers were separated, and dichloromethane solution was dried over anhydrous sodium sulfate, decanted, and concentrated. The product was recrystallized from ethyl acetate to give the title product. MS m/z 493 (M+1).
Step 2. Preparation of [5-Methoxy-2-methyl-4-(4′-trifluoromethyl-biphenyl-4-ylmethanesulfinyl)-phenoxy]-acetic acid (compound 51)
The title compound was prepared in the manner analogous to Example 1 using 51A. 400 MHz 1H NMR (DMSO-d6) δ 7.83 (d, 2H, J=8.78 Hz), 7.76 (d, 2H, J=8.30 Hz), 7.59 (d, 2H, J=8.30 Hz), 7.11 (d, 2H, J=8.30 Hz), 7.02 (s, 1H), 6.64 (s, 1H), 4.79 (s, 2H), 4.20 (d, 1H, J=12.7 Hz), 3.91 (d, 1H, J=12.4 Hz), 3.76 (s, 3H), 2.03 (s, 3H). MS m/z 479 (M+1).
Step 1. Preparation of [5-Methoxy-2-methyl-4-(4′-trifluoromethyl-biphenyl-4-ylmethanesulfinyl)-phenoxy]-acetic acid methyl ester (compound 51A)
Example 4 (0.5 g, 1.0 mmol) was dissolved in 25 ml dichloromethane followed by the addition of 2-benzenesulfonyl-3-phenyl-oxaziridine (0.274, 1.04 mmol). The reaction was stirred 1 h. 10 ml water was added, the layers were separated, and dichloromethane solution was dried over anhydrous sodium sulfate, decanted, and concentrated. The product was recrystallized from ethyl acetate to give the title product. MS m/z 493 (M+1).
Step 2. Preparation of [5-Methoxy-2-methyl-4-(4′-trifluoromethyl-biphenyl-4-ylmethanesulfinyl)-phenoxy]-acetic acid (compound 51)
The title compound was prepared in the manner analogous to Example 1 using 51A. 400 MHz 1H NMR (DMSO-d6) δ 7.83 (d, 2H, J=8.78 Hz), 7.76 (d, 2H, J=8.30 Hz), 7.59 (d, 2H, J=8.30 Hz), 7.11 (d, 2H, J=8.30 Hz), 7.02 (s, 1H), 6.64 (s, 1H), 4.79 (s, 2H), 4.20 (d, 1H, J=12.7 Hz), 3.91 (d, 1H, J=12.4 Hz), 3.76 (s, 3H), 2.03 (s, 3H). MS m/z 479 (M+1).
Synthesis of [2-Propyl-4-(4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid (compound 52)
Step 1. Preparation of [2-Propyl-4-(4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid methyl ester (compound 52A)
The title compound was prepared in the manner analogous to Example 1F using 48B and (4-Mercapto-2-propyl-phenoxy)-acetic acid methyl ester, prepared from 2-propylphenol in a manner analogous to Example 2. MS m/z 475 (M+1).
Step 2. Preparation of [2-Propyl-4-(4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid (compound 52)
The title compound was prepared in the manner analogous to Example 1 using 52A. 400 MHz 1H NMR (DMSO-d6) δ 7.81 (d, 2H, J=8.5 Hz), 7.74 (d, 2H, J=8.5 Hz), 7.60 (d, 2H, J=8.1 Hz), 7.33 (d, 2H, J=8.3 Hz), 7.10 (m, 1H), 7.02 (m, 1H), 6.71 (d, 1H, J=8.5 Hz), 4.60 (s, 2H), 4.11 (s, 2H), 1.43 (m, 2H), 0.77 (m, 3H). MS m/z 459 (M−1).
Step 1. Preparation of [2-Propyl-4-(4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid methyl ester (compound 52A)
The title compound was prepared in the manner analogous to Example 1F using 48B and (4-Mercapto-2-propyl-phenoxy)-acetic acid methyl ester, prepared from 2-propylphenol in a manner analogous to Example 2. MS m/z 475 (M+1).
Step 2. Preparation of [2-Propyl-4-(4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid (compound 52)
The title compound was prepared in the manner analogous to Example 1 using 52A. 400 MHz 1H NMR (DMSO-d6) δ 7.81 (d, 2H, J=8.5 Hz), 7.74 (d, 2H, J=8.5 Hz), 7.60 (d, 2H, J=8.1 Hz), 7.33 (d, 2H, J=8.3 Hz), 7.10 (m, 1H), 7.02 (m, 1H), 6.71 (d, 1H, J=8.5 Hz), 4.60 (s, 2H), 4.11 (s, 2H), 1.43 (m, 2H), 0.77 (m, 3H). MS m/z 459 (M−1).
Synthesis of {7-[3-(5-Trifluoromethyl-pyridin-2-yl)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 53)
Step 1. Preparation of [3-(5-Trifluoromethyl-pyridin-2-yl)-phenyl]-methanol (compound 53A)
The title compound was prepared in the manner analogous to Example 3A using 3-(hydroxymethyl)phenyl boronic acid and 2-chloro-5-trifluoromethyl-pyridine. MS m/z 253 (M+1).
Step 2. Preparation of 2-(3-Chloromethyl-phenyl)-5-trifluoromethyl-pyridine (compound 53B)
The title compound was prepared in the manner analogous to Example 3B using 53A. MS m/z 237 (M+1-Cl).
Step 3. Preparation of {7-[3-(5-Trifluoromethyl-pyridin-2-yl)-benzylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 53C)
The title compound was prepared in the manner analogous to Example 1F using 12C and 53B. MS m/z 474 (M+1).
Step 4. Preparation of {7 -[3-(5-Trifluoromethyl-pyridin-2-yl)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 53)
The title compound was prepared in the manner analogous to Example 1 using 53C. 400 MHz 1H NMR (DMSO-d6) δ 8.98 (m, 1H), 8.23 (dd, 1H, J=1.7 Hz, J′=8.5 Hz), 7.99 (m, 2H), 7.87 (s, 1H), 7.39 (t, 1H, J=7.6 Hz), 7.33 (m, 1H), 7.11 (d, 1H, J=8.5 Hz), 6.58 (d, 1H, J=8.5 Hz), 4.61 (s, 2H), 4.08 (s, 2H), 2.70 (t, 2H, J=7.3 Hz), 2.63 (t, 2H, J=7.6 Hz), 1.84 (m, 2H). MS m/z 460 (M+1).
Step 1. Preparation of [3-(5-Trifluoromethyl-pyridin-2-yl)-phenyl]-methanol (compound 53A)
The title compound was prepared in the manner analogous to Example 3A using 3-(hydroxymethyl)phenyl boronic acid and 2-chloro-5-trifluoromethyl-pyridine. MS m/z 253 (M+1).
Step 2. Preparation of 2-(3-Chloromethyl-phenyl)-5-trifluoromethyl-pyridine (compound 53B)
The title compound was prepared in the manner analogous to Example 3B using 53A. MS m/z 237 (M+1-Cl).
Step 3. Preparation of {7-[3-(5-Trifluoromethyl-pyridin-2-yl)-benzylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 53C)
The title compound was prepared in the manner analogous to Example 1F using 12C and 53B. MS m/z 474 (M+1).
Step 4. Preparation of {7 -[3-(5-Trifluoromethyl-pyridin-2-yl)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 53)
The title compound was prepared in the manner analogous to Example 1 using 53C. 400 MHz 1H NMR (DMSO-d6) δ 8.98 (m, 1H), 8.23 (dd, 1H, J=1.7 Hz, J′=8.5 Hz), 7.99 (m, 2H), 7.87 (s, 1H), 7.39 (t, 1H, J=7.6 Hz), 7.33 (m, 1H), 7.11 (d, 1H, J=8.5 Hz), 6.58 (d, 1H, J=8.5 Hz), 4.61 (s, 2H), 4.08 (s, 2H), 2.70 (t, 2H, J=7.3 Hz), 2.63 (t, 2H, J=7.6 Hz), 1.84 (m, 2H). MS m/z 460 (M+1).
Synthesis of (5-Methoxy-2-methyl-4-{2-[1-(4-trifluoromethyl-benzyl)-1H-indol-3-yl]-ethylsulfanyl}-phenoxy)-acetic acid (compound 54)
Step 1. Preparation of {4-[2-(1H-Indol-3-yl)-ethylsulfanyl]-5-methoxy-2-methyl-phenoxy}-acetic acid methyl ester (compound 54A)
The compound 1D (1.622 g, 6.7 mmol) and 3-(2-bromo-ethyl)-1H-indole (1.50 g, 6.69 mmol) was dissolved in 5 ml DMF. Potassium carbonate (1.11 g, 8.03 mmol) was added followed by stirring at room temperature for 3 h. The reaction was filtered, concentrated and purified by MPLC to give the title compound. MS m/z 225 (M+1).
Step 2. Preparation of (5-Methoxy-2-methyl-4-{2-[1-(4-trifluoromethyl-benzyl)-1H-indol-3-yl]-ethylsulfanyl}-phenoxy)-acetic acid methyl ester (compound 54B)
The compound 54A (0.300 g, 0.778 mmol) was dissolved in 5 ml DMF. NaH was added and stirred for ½ h. 4-trifluoromethylbenzyl bromide (0.223 g, 0.934 mmol) was added and the reaction was stirred for 1.5 h. 10 ml 2N HCl was added to pH<4. The DMF solution was partitioned between 30 ml water and 30 ml ethyl acetate. The organic solution was washed 2×30 ml water 1×30 ml brine, dried over sodium sulfate, decanted, concentrated and purified by MPLC to give the title product. MS m/z 544 (m+1).
Step 3. Preparation of (5-Methoxy-2-methyl-4-{2-[1-(4-trifluoromethyl-benzyl)-1H-indol-3-yl]-ethylsulfanyl}-phenoxy)-acetic acid (compound 54)
The title compound was prepared in the manner analogous to Example 1 using 54B. 400 MHz 1H NMR (DMSO-d6) δ 13.0 (br(s), 1H), 7.60 (d, 1H, J=8.1 Hz), 7.42 (d, 1H, J=7.96 Hz), 7.31 (m, 4H), 7.04 (m, 2H), 6.96 (m, 1H), 6.53 (s, 1H), 5.43 (s, 2H), 4.70 (s, 2H), 3.72 (s, 3H), 3.03 (t, 2H, J=8.8 Hz), 2.86 (t, 2H, J=7.6 Hz), 2.04 (s, 3H). MS m/z 530 (M+1).
Step 1. Preparation of {4-[2-(1H-Indol-3-yl)-ethylsulfanyl]-5-methoxy-2-methyl-phenoxy}-acetic acid methyl ester (compound 54A)
The compound 1D (1.622 g, 6.7 mmol) and 3-(2-bromo-ethyl)-1H-indole (1.50 g, 6.69 mmol) was dissolved in 5 ml DMF. Potassium carbonate (1.11 g, 8.03 mmol) was added followed by stirring at room temperature for 3 h. The reaction was filtered, concentrated and purified by MPLC to give the title compound. MS m/z 225 (M+1).
Step 2. Preparation of (5-Methoxy-2-methyl-4-{2-[1-(4-trifluoromethyl-benzyl)-1H-indol-3-yl]-ethylsulfanyl}-phenoxy)-acetic acid methyl ester (compound 54B)
The compound 54A (0.300 g, 0.778 mmol) was dissolved in 5 ml DMF. NaH was added and stirred for ½ h. 4-trifluoromethylbenzyl bromide (0.223 g, 0.934 mmol) was added and the reaction was stirred for 1.5 h. 10 ml 2N HCl was added to pH<4. The DMF solution was partitioned between 30 ml water and 30 ml ethyl acetate. The organic solution was washed 2×30 ml water 1×30 ml brine, dried over sodium sulfate, decanted, concentrated and purified by MPLC to give the title product. MS m/z 544 (m+1).
Step 3. Preparation of (5-Methoxy-2-methyl-4-{2-[1-(4-trifluoromethyl-benzyl)-1H-indol-3-yl]-ethylsulfanyl}-phenoxy)-acetic acid (compound 54)
The title compound was prepared in the manner analogous to Example 1 using 54B. 400 MHz 1H NMR (DMSO-d6) δ 13.0 (br(s), 1H), 7.60 (d, 1H, J=8.1 Hz), 7.42 (d, 1H, J=7.96 Hz), 7.31 (m, 4H), 7.04 (m, 2H), 6.96 (m, 1H), 6.53 (s, 1H), 5.43 (s, 2H), 4.70 (s, 2H), 3.72 (s, 3H), 3.03 (t, 2H, J=8.8 Hz), 2.86 (t, 2H, J=7.6 Hz), 2.04 (s, 3H). MS m/z 530 (M+1).
Synthesis of [7-(4′-Trifluoromethyl-biphenyl-2-ylmethylsulfanyl)-indan-4-yloxy]-acetic acid (compound 55)
Step 1. Preparation of [7-(4′-Trifluoromethyl-biphenyl-2-ylmethylsulfanyl)-indan-4-yloxy]-acetic acid methyl ester (compound 55A)
The title compound was prepared in the manner analogous to Example 1F using 12C, and 2-chloromethyl-4′-trifluoromethyl-biphenyl, which was prepared in a manner analogous to Examples 3A and 3B. MS m/z 473 (M+1).
Step 2. Preparation of [7-(4′-Trifluoromethyl-biphenyl-2-ylmethylsulfanyl)-indan-4-yloxy]-acetic acid (compound 55)
The title compound was prepared in the manner analogous to Example 1 using 55A. 400 MHz 1H NMR (DMSO-d6) δ 12.97 (br(s), 1H), 7.68 (d, 2H, J=8.5 Hz), 7.32 (m, 5H), 7.11 (m, 1H), 6.77 (d, 1H, J=8.5 Hz), 6.47 (d, 1H, J=8.3 Hz), 4.62 (s, 2H), 3.87 (s, 2H), 2.70 (m, 2H), 1.76 (m, 2H). MS m/z 459 (M+1).
Step 1. Preparation of [7-(4′-Trifluoromethyl-biphenyl-2-ylmethylsulfanyl)-indan-4-yloxy]-acetic acid methyl ester (compound 55A)
The title compound was prepared in the manner analogous to Example 1F using 12C, and 2-chloromethyl-4′-trifluoromethyl-biphenyl, which was prepared in a manner analogous to Examples 3A and 3B. MS m/z 473 (M+1).
Step 2. Preparation of [7-(4′-Trifluoromethyl-biphenyl-2-ylmethylsulfanyl)-indan-4-yloxy]-acetic acid (compound 55)
The title compound was prepared in the manner analogous to Example 1 using 55A. 400 MHz 1H NMR (DMSO-d6) δ 12.97 (br(s), 1H), 7.68 (d, 2H, J=8.5 Hz), 7.32 (m, 5H), 7.11 (m, 1H), 6.77 (d, 1H, J=8.5 Hz), 6.47 (d, 1H, J=8.3 Hz), 4.62 (s, 2H), 3.87 (s, 2H), 2.70 (m, 2H), 1.76 (m, 2H). MS m/z 459 (M+1).
Synthesis of {5-Methoxy-2-methyl-4-[2-(4-trifluoromethyl-benzyloxy)-benzylsulfanyl]-phenoxy}-acetic acid (compound 56)
Step 1. Preparation of {5-Methoxy-2-methyl-4-[2-(4-trifluoromethyl-benzyloxy)-benzylsulfanyl]-phenoxy}-acetic acid methyl ester (compound 56A)
The title compound was prepared in the manner analogous to Example 1F using 81A and 1D. MS m/z 507 (M+1).
Step 2. Preparation of 5-Methoxy-2-methyl-4-[2-(4-trifluoromethyl-benzyloxy)-benzylsulfanyl]-phenoxy}-acetic acid (compound 56)
The title compound was prepared in the manner analogous to Example 1 using 56A. 400 MHz 1H NMR (DMSO-d6) δ 12.95 (br(s), 1H), 7.67 (m, 4H), 7.12 (m, 2H), 6.96 (m, 2H), 6.80 (m, 1H), 6.49 (s, 1H), 5.19 (s, 2H), 4.68 (s, 2H), 3.99 (s, 2H), 3.67 (s, 3H), 1.96 (s, 3H). MS m/z 493 (M+1).
Step 1. Preparation of {5-Methoxy-2-methyl-4-[2-(4-trifluoromethyl-benzyloxy)-benzylsulfanyl]-phenoxy}-acetic acid methyl ester (compound 56A)
The title compound was prepared in the manner analogous to Example 1F using 81A and 1D. MS m/z 507 (M+1).
Step 2. Preparation of 5-Methoxy-2-methyl-4-[2-(4-trifluoromethyl-benzyloxy)-benzylsulfanyl]-phenoxy}-acetic acid (compound 56)
The title compound was prepared in the manner analogous to Example 1 using 56A. 400 MHz 1H NMR (DMSO-d6) δ 12.95 (br(s), 1H), 7.67 (m, 4H), 7.12 (m, 2H), 6.96 (m, 2H), 6.80 (m, 1H), 6.49 (s, 1H), 5.19 (s, 2H), 4.68 (s, 2H), 3.99 (s, 2H), 3.67 (s, 3H), 1.96 (s, 3H). MS m/z 493 (M+1).
Synthesis of {4-[4-(4-Fluoro-benzyloxy)-benzylsulfanyl]-2-methyl-phenoxy}-acetic acid (compound 57)
Step 1. Preparation of [4-(4-Fluoro-benzyloxy)-phenyl]-methanol (compound 57A)
The title compound was prepared in the manner analogous to Example 14A using 1-bromomethyl-4-fluoro-benzene and 4-hydroxymethyl-phenol. MS m/z 215 (M-OH).
Step 2. Preparation of 1-(4-Chloromethyl-phenoxymethyl)-4difluoro-benzene (compound 57B)
The title compound was prepared in the manner analogous to Example 3B using 57A. MS m/z 215 (M-Cl).
Step 3. Preparation of {4-[4-(4-Fluoro-benzyloxy)-benzylsulfanyl]-2-methyl-phenoxy}-acetic acid methyl ester (compound 57C)
The title compound was prepared in the manner analogous to Example 1F using 57B and 2C. MS m/z 427 (M+1).
Step 4. Preparation of {4-[4-(4-Fluoro-benzyloxy)-benzylsulfanyl]-2-methyl-phenoxy}-acetic acid (compound 57)
The title compound was prepared in the manner analogous to Example 1 using the product from Example 57C. mp 154-155° C.; IR (KBr) cm−1: 3060, 2925, 1727, 1495, 1228, 1156; 400 MHz 1H NMR (DMSO-d6): δ 13.00 (br(s), 1H), 7.37-7.48 (m, 2H), 6.99-7.22 (m, 6H), 6.80-6.90 (m, 2H), 6.69 (d, 1H, J=8.5 Hz), 4.98 (s, 2H), 4.61 (s, 2H), 4.00 (s, 2H), 2.08 (s, 3H); MS m/z 411 (M−1). Anal. Calc'd for C23H21FO4S: C, 66.97; H, 5.13; found: C, 66.64; H, 4.88.
Step 1. Preparation of [4-(4-Fluoro-benzyloxy)-phenyl]-methanol (compound 57A)
The title compound was prepared in the manner analogous to Example 14A using 1-bromomethyl-4-fluoro-benzene and 4-hydroxymethyl-phenol. MS m/z 215 (M-OH).
Step 2. Preparation of 1-(4-Chloromethyl-phenoxymethyl)-4difluoro-benzene (compound 57B)
The title compound was prepared in the manner analogous to Example 3B using 57A. MS m/z 215 (M-Cl).
Step 3. Preparation of {4-[4-(4-Fluoro-benzyloxy)-benzylsulfanyl]-2-methyl-phenoxy}-acetic acid methyl ester (compound 57C)
The title compound was prepared in the manner analogous to Example 1F using 57B and 2C. MS m/z 427 (M+1).
Step 4. Preparation of {4-[4-(4-Fluoro-benzyloxy)-benzylsulfanyl]-2-methyl-phenoxy}-acetic acid (compound 57)
The title compound was prepared in the manner analogous to Example 1 using the product from Example 57C. mp 154-155° C.; IR (KBr) cm−1: 3060, 2925, 1727, 1495, 1228, 1156; 400 MHz 1H NMR (DMSO-d6): δ 13.00 (br(s), 1H), 7.37-7.48 (m, 2H), 6.99-7.22 (m, 6H), 6.80-6.90 (m, 2H), 6.69 (d, 1H, J=8.5 Hz), 4.98 (s, 2H), 4.61 (s, 2H), 4.00 (s, 2H), 2.08 (s, 3H); MS m/z 411 (M−1). Anal. Calc'd for C23H21FO4S: C, 66.97; H, 5.13; found: C, 66.64; H, 4.88.
Synthesis of {4-[4-(4-Chloro-benzyloxy)-benzylsulfanyl]-2-methyl-phenoxy}-acetic acid (compound 58)
Step 1. Preparation of {4-[4(4-Chloro-benzyloxy)-benzylsulfanyl]-2-methyl-phenoxy}-acetic acid methyl ester (compound 58A)
The title compound was prepared in the manner analogous to Example 1F using I-bromomethyl-4-chloro-benzene and the product from Example 35C. MS m/z 443 (M+1).
Step 2. Preparation of {4-[4-(4-Chloro-benzyloxy)-benzylsulfanyl]-2-methyl-phenoxy}-acetic acid (compound 58)
The title compound was prepared in the manner analogous to Example 1 using 58A. mp 164° C.; IR (KBr) cm−1: 3062, 1727, 1612, 1493, 1226, 1193; 400 MHz 1H NMR (DMSO-d6): δ 12.99 (br(s), 1H), 7.36-7.44 (m, 4H), 7.01-7.17 (m, 4H), 6.81-6.89 (m, 2H), 6.69 (d, 1H, J=8.5 Hz), 5.02 (s, 2H), 4.61 (s, 2H), 4.00 (s, 2H), 2.08 (s, 3H); MS m/z 427 (M−1); Anal. Calc'd for C23H21ClO4S: C, 64.40; H, 4.93; found: C, 64.43; H, 4.81.
Step 1. Preparation of {4-[4(4-Chloro-benzyloxy)-benzylsulfanyl]-2-methyl-phenoxy}-acetic acid methyl ester (compound 58A)
The title compound was prepared in the manner analogous to Example 1F using I-bromomethyl-4-chloro-benzene and the product from Example 35C. MS m/z 443 (M+1).
Step 2. Preparation of {4-[4-(4-Chloro-benzyloxy)-benzylsulfanyl]-2-methyl-phenoxy}-acetic acid (compound 58)
The title compound was prepared in the manner analogous to Example 1 using 58A. mp 164° C.; IR (KBr) cm−1: 3062, 1727, 1612, 1493, 1226, 1193; 400 MHz 1H NMR (DMSO-d6): δ 12.99 (br(s), 1H), 7.36-7.44 (m, 4H), 7.01-7.17 (m, 4H), 6.81-6.89 (m, 2H), 6.69 (d, 1H, J=8.5 Hz), 5.02 (s, 2H), 4.61 (s, 2H), 4.00 (s, 2H), 2.08 (s, 3H); MS m/z 427 (M−1); Anal. Calc'd for C23H21ClO4S: C, 64.40; H, 4.93; found: C, 64.43; H, 4.81.
Synthesis of 4-[4-(2,5-Dichloro-benzyloxy)-benzylsulfanyl]-2-methyl-phenoxy}-acetic acid (compound 59)
Step 1. Preparation of {4-[4-(2,5-Dichloro-benzyloxy)-benzylsulfanyl]-2-methyl-phenoxy}-acetic acid methyl ester (compound 59A)
The title compound was prepared in the manner analogous to Example 1F using 2-bromomethyl-1,4-dichloro-benzene and the product prepared from Example 35C. m/z 265 (M−211).
Step 2. Preparation of {4-[4-(2,4-Dichlorobenzyloxy)-benzylsulfanyl]-2-methyl-phenoxy}-acetic acid (compound 59)
The title compound was prepared in the manner analogous to Example 1 using 59A. mp 101-103° C.; IR (KBr) cm−1: 3077, 1715, 1608, 1496, 1437, 1234; 400 MHz 1H NMR (DMSO-d6): δ 7.37-7.63 (m, 3H), 6.99-7.20 (m, 4H), 6.85-6.94 (m, 2H), 6.63 (d, 1H, J=8.5 Hz), 5.05 (s, 2H), 4.45 (s, 2H), 3.99 (s, 2H), 2.07 (s, 3H); HPLC: area %=97.61, r.t.=5.56 min., λ=214 nm, mobile phase=acetonitrile/water with 0.10% TFA; MS m/z 461 (M−1). Anal. Calc'd for C23H20Cl2O4S: C, 59.62; H, 4.35; found: C, 58.04; H, 4.26.
Step 1. Preparation of {4-[4-(2,5-Dichloro-benzyloxy)-benzylsulfanyl]-2-methyl-phenoxy}-acetic acid methyl ester (compound 59A)
The title compound was prepared in the manner analogous to Example 1F using 2-bromomethyl-1,4-dichloro-benzene and the product prepared from Example 35C. m/z 265 (M−211).
Step 2. Preparation of {4-[4-(2,4-Dichlorobenzyloxy)-benzylsulfanyl]-2-methyl-phenoxy}-acetic acid (compound 59)
The title compound was prepared in the manner analogous to Example 1 using 59A. mp 101-103° C.; IR (KBr) cm−1: 3077, 1715, 1608, 1496, 1437, 1234; 400 MHz 1H NMR (DMSO-d6): δ 7.37-7.63 (m, 3H), 6.99-7.20 (m, 4H), 6.85-6.94 (m, 2H), 6.63 (d, 1H, J=8.5 Hz), 5.05 (s, 2H), 4.45 (s, 2H), 3.99 (s, 2H), 2.07 (s, 3H); HPLC: area %=97.61, r.t.=5.56 min., λ=214 nm, mobile phase=acetonitrile/water with 0.10% TFA; MS m/z 461 (M−1). Anal. Calc'd for C23H20Cl2O4S: C, 59.62; H, 4.35; found: C, 58.04; H, 4.26.
Synthesis of {2-Methyl-4-[4-(pyridine-2-ylmethoxy)-benzylsulfanyl]-phenoxy}-acetic acid (compound 60)
Step 1. Preparation of {2-Methyl-4-[4-(pyridine-2-ylmethoxy)-benzylsulfanyl]-phenoxy}-acetic acid methyl ester (compound 60A)
The title compound was prepared in the manner analogous to Example 1F using 2-bromomethyl-pyridine hydrochloride and the product prepared from Example 35C. MS m/z 410 (M+1).
Step 2. Preparation of {2-Methyl-4-[4-(pyridine-2-ylmethoxy)-benzylsulfanyl]-phenoxy}-acetic acid (compound 60)
The title compound was prepared in the manner analogous to Example 1 using 60A. mp 150° C.; IR (KBr) cm−1: 2917, 1723, 1605, 1511, 1490,1217; 400 MHz 1H NMR (DMSO-d6): δ 12.95 (brs, 1H), 8.49-8.55 (s, 1H), 7.73-7.81 (m, 1H), 7.40-7.48 (m, 1H), 7.24-7.33 (m, 1H), 7.00-7.19 (m, 4H), 6.83-6.93 (m, 2H), 6.69 (d, 1H, J=8.5 Hz), 5.08 (s, 2H), 4.62 (s, 2H), 4.00 (s, 2H), 2.08 (s, 3H); HPLC: area %=96.24, r.t.=1.95 min., γ=214 nm, mobile phase=acetonitrile/water with 0.10% TFA; MS m/z 396 (M+1). Anal. Calc'd for C22H21NO4S: C, 66.82; H, 5.35; N, 3.54; found: C, 66.12; H, 5.09; N, 3.44.
Step 1. Preparation of {2-Methyl-4-[4-(pyridine-2-ylmethoxy)-benzylsulfanyl]-phenoxy}-acetic acid methyl ester (compound 60A)
The title compound was prepared in the manner analogous to Example 1F using 2-bromomethyl-pyridine hydrochloride and the product prepared from Example 35C. MS m/z 410 (M+1).
Step 2. Preparation of {2-Methyl-4-[4-(pyridine-2-ylmethoxy)-benzylsulfanyl]-phenoxy}-acetic acid (compound 60)
The title compound was prepared in the manner analogous to Example 1 using 60A. mp 150° C.; IR (KBr) cm−1: 2917, 1723, 1605, 1511, 1490,1217; 400 MHz 1H NMR (DMSO-d6): δ 12.95 (brs, 1H), 8.49-8.55 (s, 1H), 7.73-7.81 (m, 1H), 7.40-7.48 (m, 1H), 7.24-7.33 (m, 1H), 7.00-7.19 (m, 4H), 6.83-6.93 (m, 2H), 6.69 (d, 1H, J=8.5 Hz), 5.08 (s, 2H), 4.62 (s, 2H), 4.00 (s, 2H), 2.08 (s, 3H); HPLC: area %=96.24, r.t.=1.95 min., γ=214 nm, mobile phase=acetonitrile/water with 0.10% TFA; MS m/z 396 (M+1). Anal. Calc'd for C22H21NO4S: C, 66.82; H, 5.35; N, 3.54; found: C, 66.12; H, 5.09; N, 3.44.
Synthesis of {5-Chloro-2-methyl-4-[4-(4-trifluoromethyl-benzyloxy)-benzylsulfanyl]-phenoxy}-acetic acid (compound 61)
Step 1. Preparation of (5-Chloro-2-methyl-4-[4-(4trifluoromethyl-benzyloxy)-benzyl-sulfanyl]-phenoxy}-acetic acid methyl ester (compound 61A)
The title compound was prepared in the manner analogous to Example 1F using 14B and 20C. MS m/z 265 (M−245).
Step 2. Preparation of {5-Chloro-2-methyl-4-[4-(4-trifluoromethyl-benzyloxy)-benzyl-sulfanyl]-phenoxy}-acetic acid (compound 61)
The title compound was prepared in the manner analogous to Example 1 using 61A. mp 142-143° C.; IR (KBr) cm−1: 3074, 1747, 1321, 1234, 1175, 1124; 400 MHz 1H NMR (DMSO-d6): δ 13.01 (br(s), 1H), 7.67-7.73 (m, 2H), 7.57-7.64 (m, 2H), 7.16-7.25 (m, 4H), 6.87-6.95 (m, 4H), 5.15 (s, 2H), 4.69 (s, 2H), 4.07 (s, 2H), 2.07 (s, 3H); MS m/z 495 (M−1). Anal. Calc'd for C24H20ClF3O4S: C, 58.01; H, 4.06; found: C, 57.73; H, 4.06.
Step 1. Preparation of (5-Chloro-2-methyl-4-[4-(4trifluoromethyl-benzyloxy)-benzyl-sulfanyl]-phenoxy}-acetic acid methyl ester (compound 61A)
The title compound was prepared in the manner analogous to Example 1F using 14B and 20C. MS m/z 265 (M−245).
Step 2. Preparation of {5-Chloro-2-methyl-4-[4-(4-trifluoromethyl-benzyloxy)-benzyl-sulfanyl]-phenoxy}-acetic acid (compound 61)
The title compound was prepared in the manner analogous to Example 1 using 61A. mp 142-143° C.; IR (KBr) cm−1: 3074, 1747, 1321, 1234, 1175, 1124; 400 MHz 1H NMR (DMSO-d6): δ 13.01 (br(s), 1H), 7.67-7.73 (m, 2H), 7.57-7.64 (m, 2H), 7.16-7.25 (m, 4H), 6.87-6.95 (m, 4H), 5.15 (s, 2H), 4.69 (s, 2H), 4.07 (s, 2H), 2.07 (s, 3H); MS m/z 495 (M−1). Anal. Calc'd for C24H20ClF3O4S: C, 58.01; H, 4.06; found: C, 57.73; H, 4.06.
Synthesis of {7-[4-(2,4Dichloro-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 62)
Step 1. Preparation of {7-[4(2,4-Dichloro-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 62A)
The title compound was prepared in the manner analogous to Example 1F using 12C and 4chloromethyl-(2,4-dichloro-benzyloxy-benzene) prepared from 4-hydroxymethyl-phenol and 1-chloromethyl-2,4-dichloro-benzene in the manner analogous to Examples 14A and 14B. MS m/z 503 (M+1).
Step 2. Preparation of {7-[4-(2,4-Dichloro-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 62)
The title compound was prepared in the manner analogous to Example 1 using 62A. mp 149-150° C.; IR (KBr) cm−1: 3089, 1734, 1512, 1473, 1295, 1234; 400 MHz 1H NMR (DMSO-d6): δ 13.11 (br(s), 1H), 7.40-7.71 (m, 3H), 6.88-7.19 (m, 5H), 6.58 (d, 1H, J=8.5 Hz), 5.07 (s, 2H), 4.61 (s, 2H), 3.96 (s, 2H), 2.77 (t, 2H, J=7.4 Hz), 2.69 (t, 2H, J=7.4 Hz), 1.91 (pentet, 2H); MS m/z 487 (M−1). Anal. Calc'd for C25H22Cl2O4S: C, 61.35; H, 4.53; found: C, 60.95; H, 4.41.
Step 1. Preparation of {7-[4(2,4-Dichloro-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 62A)
The title compound was prepared in the manner analogous to Example 1F using 12C and 4chloromethyl-(2,4-dichloro-benzyloxy-benzene) prepared from 4-hydroxymethyl-phenol and 1-chloromethyl-2,4-dichloro-benzene in the manner analogous to Examples 14A and 14B. MS m/z 503 (M+1).
Step 2. Preparation of {7-[4-(2,4-Dichloro-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 62)
The title compound was prepared in the manner analogous to Example 1 using 62A. mp 149-150° C.; IR (KBr) cm−1: 3089, 1734, 1512, 1473, 1295, 1234; 400 MHz 1H NMR (DMSO-d6): δ 13.11 (br(s), 1H), 7.40-7.71 (m, 3H), 6.88-7.19 (m, 5H), 6.58 (d, 1H, J=8.5 Hz), 5.07 (s, 2H), 4.61 (s, 2H), 3.96 (s, 2H), 2.77 (t, 2H, J=7.4 Hz), 2.69 (t, 2H, J=7.4 Hz), 1.91 (pentet, 2H); MS m/z 487 (M−1). Anal. Calc'd for C25H22Cl2O4S: C, 61.35; H, 4.53; found: C, 60.95; H, 4.41.
Synthesis of {7-[4-(4-Trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 63)
Step 1. Preparation of {7-[4-(4-Trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 63A)
The title compound was prepared in the manner analogous to Example 1F using 14B and 12C. MS m/z 265 (M−237).
Step 2. Preparation of {7-[4-(4-Trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 63)
The title compound was prepared in the manner analogous to Example 1 using 63A. mp 145° C.; IR (KBr) cm−1: 2968, 1740, 1510, 1325, 1248, 1110; 400 MHz 1H NMR (DMSO-d6): δ 12.99 (br(s), 1H), 7.66-7.75 (m, 2H), 7.55-7.65 (m, 2H); 7.01-7.15 (m, 3H) 6.83-6.90 (m, 2H), 6.56 (d, 1H, J=8.5 Hz), 5.14 (s, 2H), 4.60 (s, 2H), 3.93 (s, 2H), 2.74 (t, 2H, t, J=7.6 Hz), 2.65 (t, 2H, J=7.4 Hz), 1.87 (pentet, 2H); MS m/z 487 (M−1). Anal. Calc'd for C26H23F3O4S: C, 63.92; H, 4.75; found: C, 63.95; H, 4.65.
Step 1. Preparation of {7-[4-(4-Trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 63A)
The title compound was prepared in the manner analogous to Example 1F using 14B and 12C. MS m/z 265 (M−237).
Step 2. Preparation of {7-[4-(4-Trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 63)
The title compound was prepared in the manner analogous to Example 1 using 63A. mp 145° C.; IR (KBr) cm−1: 2968, 1740, 1510, 1325, 1248, 1110; 400 MHz 1H NMR (DMSO-d6): δ 12.99 (br(s), 1H), 7.66-7.75 (m, 2H), 7.55-7.65 (m, 2H); 7.01-7.15 (m, 3H) 6.83-6.90 (m, 2H), 6.56 (d, 1H, J=8.5 Hz), 5.14 (s, 2H), 4.60 (s, 2H), 3.93 (s, 2H), 2.74 (t, 2H, t, J=7.6 Hz), 2.65 (t, 2H, J=7.4 Hz), 1.87 (pentet, 2H); MS m/z 487 (M−1). Anal. Calc'd for C26H23F3O4S: C, 63.92; H, 4.75; found: C, 63.95; H, 4.65.
Synthesis of {5-Methyl-7-[4-(4-trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 64)
Step 1. Preparation of {5-Methyl-7-[4-(4-trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 64A)
The title compound was prepared in the manner analogous to Example 1F using 14B and (7-mercapto-5-methyl-indan-4-yloxy)-acetic acid methyl ester, which was prepared in a similar manner as described for Example 12C. MS m/z 265 (M−251).
Step 2. Preparation of {5-Methyl-7-[4-(4-trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 64)
The title compound was prepared in the manner analogous to Example 1 using 64A. mp 165-167° C.; IR (KBr) cm−1: 3039, 1707, 1516, 1329, 1163, 1112; 400 MHz 1H NMR (DMSO-d6) δ 12.81 (br(s), 1H), 7.66-7.74 (m, 2H), 7.56-7.64 (m, 2H); 7.11-7.19 (m, 2H), 6.94 (s, 1H), 6.85-6.91 (m, 2H), 5.15 (s, 2H), 4.40 (s, 2H), 3.99 (s, 2H), 2.81 (t, 2H, J=7.4 Hz), 2.59 (t, 2H, J=7.4 Hz), 2.13 (s, 3H), 1.86 (pentet, 2H); MS m/z 501 (M−1). Anal. Calc'd for C27H23F3O4S: C, 64.53; H, 5.01; found: C, 64.33; H, 4.82.
Step 1. Preparation of {5-Methyl-7-[4-(4-trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 64A)
The title compound was prepared in the manner analogous to Example 1F using 14B and (7-mercapto-5-methyl-indan-4-yloxy)-acetic acid methyl ester, which was prepared in a similar manner as described for Example 12C. MS m/z 265 (M−251).
Step 2. Preparation of {5-Methyl-7-[4-(4-trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 64)
The title compound was prepared in the manner analogous to Example 1 using 64A. mp 165-167° C.; IR (KBr) cm−1: 3039, 1707, 1516, 1329, 1163, 1112; 400 MHz 1H NMR (DMSO-d6) δ 12.81 (br(s), 1H), 7.66-7.74 (m, 2H), 7.56-7.64 (m, 2H); 7.11-7.19 (m, 2H), 6.94 (s, 1H), 6.85-6.91 (m, 2H), 5.15 (s, 2H), 4.40 (s, 2H), 3.99 (s, 2H), 2.81 (t, 2H, J=7.4 Hz), 2.59 (t, 2H, J=7.4 Hz), 2.13 (s, 3H), 1.86 (pentet, 2H); MS m/z 501 (M−1). Anal. Calc'd for C27H23F3O4S: C, 64.53; H, 5.01; found: C, 64.33; H, 4.82.
Synthesis of {7-[4-(4-Fluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 65)
Step 1. Preparation of {7-[4-(4-Fluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 65A)
The title compound was prepared in the manner analogous to Example 1F using 12C and 4-chloromethyl-(4-fluoro-benzyloxy-benzene), prepared from 4-hydroxymethyl-phenol and 1-bromomethyl-4-fluoro-benzene in a manner analogous to Examples 14A and 14B. MS m/z 453 (M+1).
Step 2. Preparation of {7-[4-(4-Fluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 65)
The title compound was prepared in the manner analogous to Example 1 using 65A. mp 153-155° C.; IR (KBr) cm−1: 3117, 3028, 1731, 1512, 1471, 1231; 400 MHz 1H NMR (DMSO-d6): δ 12.94 (br(s), 1H), 7.39-7.47 (m, 2H), 7.01-7.20 (m, 5H); 6.82-6.89 (m, 2H) 6.56 (d, 1H, J=8.4 Hz), 4.99 (s, 2H), 4.61 (s, 2H), 3.94 (s, 2H), 2.75 (t, 2H, J=7.5 Hz), 2.67 (t, 2H, J=7.5 Hz), 1.89 (pentet, 2H); MS m/z 437 (M−1). Anal. Calc'd for C25H23FO4S: C, 68.48; H, 5.29; found: C, 68.24; H, 5.15.
Step 1. Preparation of {7-[4-(4-Fluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 65A)
The title compound was prepared in the manner analogous to Example 1F using 12C and 4-chloromethyl-(4-fluoro-benzyloxy-benzene), prepared from 4-hydroxymethyl-phenol and 1-bromomethyl-4-fluoro-benzene in a manner analogous to Examples 14A and 14B. MS m/z 453 (M+1).
Step 2. Preparation of {7-[4-(4-Fluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 65)
The title compound was prepared in the manner analogous to Example 1 using 65A. mp 153-155° C.; IR (KBr) cm−1: 3117, 3028, 1731, 1512, 1471, 1231; 400 MHz 1H NMR (DMSO-d6): δ 12.94 (br(s), 1H), 7.39-7.47 (m, 2H), 7.01-7.20 (m, 5H); 6.82-6.89 (m, 2H) 6.56 (d, 1H, J=8.4 Hz), 4.99 (s, 2H), 4.61 (s, 2H), 3.94 (s, 2H), 2.75 (t, 2H, J=7.5 Hz), 2.67 (t, 2H, J=7.5 Hz), 1.89 (pentet, 2H); MS m/z 437 (M−1). Anal. Calc'd for C25H23FO4S: C, 68.48; H, 5.29; found: C, 68.24; H, 5.15.
Synthesis of {7-[4-(2,4-Difluoro-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 66)
Step 1. Preparation of {17-[4(2,4-Difluoro-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 66A)
The title compound was prepared in the manner analogous to Example 1F using 12C and 4chloromethyl-(2,4-difluoro-benzyloxy-benzene) prepared from 4-hydroxymethyl-phenol and 1-bromomethyl-2,4-difluoro-benzene in the manner analogous to Examples 14A and 14B. MS m/z 471 (M+1).
Step 2. Preparation of {7-[4-(2,4-Difluoro-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 66)
The title compound was prepared in the manner analogous to Example 1 using 66A. mp 158-160° C.; IR (KBr) cm−1: 3065, 3043, 1751, 1510, 1433, 1239; 400 MHz 1H NMR (DMSO-d6): δ 12.94 (br(s), 1H), 7.51-7.61 (m, 1H), 7.21-7.30 (m, 1H); 7.01-7.16 (m, 4H) 6.83-6.92 (m, 2H), 6.57 (d, 1H, J=8.5 Hz), 5.01 (s, 2H), 4.61 (s, 2H), 3.94 (s, 2H), 2.76 (t, 2H, J=7.4 Hz), 2.68 (t, 2H, J=7.4 Hz), 1.90 (pentet, 2H); MS m/z 455 (M−1). Anal. Calc'd for C25H22F2O4S: C, 65.78; H, 4.86; found: C, 65.58; H, 4.83.
Step 1. Preparation of {17-[4(2,4-Difluoro-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 66A)
The title compound was prepared in the manner analogous to Example 1F using 12C and 4chloromethyl-(2,4-difluoro-benzyloxy-benzene) prepared from 4-hydroxymethyl-phenol and 1-bromomethyl-2,4-difluoro-benzene in the manner analogous to Examples 14A and 14B. MS m/z 471 (M+1).
Step 2. Preparation of {7-[4-(2,4-Difluoro-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 66)
The title compound was prepared in the manner analogous to Example 1 using 66A. mp 158-160° C.; IR (KBr) cm−1: 3065, 3043, 1751, 1510, 1433, 1239; 400 MHz 1H NMR (DMSO-d6): δ 12.94 (br(s), 1H), 7.51-7.61 (m, 1H), 7.21-7.30 (m, 1H); 7.01-7.16 (m, 4H) 6.83-6.92 (m, 2H), 6.57 (d, 1H, J=8.5 Hz), 5.01 (s, 2H), 4.61 (s, 2H), 3.94 (s, 2H), 2.76 (t, 2H, J=7.4 Hz), 2.68 (t, 2H, J=7.4 Hz), 1.90 (pentet, 2H); MS m/z 455 (M−1). Anal. Calc'd for C25H22F2O4S: C, 65.78; H, 4.86; found: C, 65.58; H, 4.83.
Synthesis of {7-[4-(4-tert-Butyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 67)
Step 1. Preparation of {7-[4(4-tert-Butyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 67A)
The title compound was prepared in the manner analogous to Example 1F using 12C and 4-chloromethyl-(4-tert-butyl-benzyloxy-benzene) prepared from 4-hydroxymethyl-phenol and 1-bromomethyl-4-tert-butyl-benzene in the manner analogous to Examples 14A and 14B. m/z 491 (M+1).
Step 2. Preparation of {7-[4(4-tert-Butyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 67)
The title compound was prepared in the manner analogous to Example 1 using 67A. mp 152-153° C.; IR (KBr) cm−1: 3134, 3032, 1745, 1708, 1473, 1228; 400 MHz 1H NMR (DMSO-d6): δ 12.94 (br(s), 1H), 7.25-7.39 (m, 4H), 7.01-7.14 (m, 3H), 6.80-6.89 (m, 2H), 6.57 (d, 1H, J=8.5 Hz), 4.97 (s, 2H), 4.61 (s, 2H), 3.93 (s, 2H), 2.75 (t, 2H, J=7.5 Hz), 2.67 (t, 2H, J=7.5 Hz), 1.89 (pentet, 2H), 1.22 (s, 9H); MS m/z 475 (M−1). Anal. Calc'd for C29H32O4S: C, 73.08; H, 6.77; found: C, 72.97; H, 6.84.
Step 1. Preparation of {7-[4(4-tert-Butyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 67A)
The title compound was prepared in the manner analogous to Example 1F using 12C and 4-chloromethyl-(4-tert-butyl-benzyloxy-benzene) prepared from 4-hydroxymethyl-phenol and 1-bromomethyl-4-tert-butyl-benzene in the manner analogous to Examples 14A and 14B. m/z 491 (M+1).
Step 2. Preparation of {7-[4(4-tert-Butyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 67)
The title compound was prepared in the manner analogous to Example 1 using 67A. mp 152-153° C.; IR (KBr) cm−1: 3134, 3032, 1745, 1708, 1473, 1228; 400 MHz 1H NMR (DMSO-d6): δ 12.94 (br(s), 1H), 7.25-7.39 (m, 4H), 7.01-7.14 (m, 3H), 6.80-6.89 (m, 2H), 6.57 (d, 1H, J=8.5 Hz), 4.97 (s, 2H), 4.61 (s, 2H), 3.93 (s, 2H), 2.75 (t, 2H, J=7.5 Hz), 2.67 (t, 2H, J=7.5 Hz), 1.89 (pentet, 2H), 1.22 (s, 9H); MS m/z 475 (M−1). Anal. Calc'd for C29H32O4S: C, 73.08; H, 6.77; found: C, 72.97; H, 6.84.
Synthesis of {7-[4-(4-Methoxy-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 68)
Step 1. Preparation of {7-[4-(4-Methoxy-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 68A)
The title compound was prepared in the manner analogous to Example 1F using 12C and 4-chloromethyl-(4-methoxy-benzyloxy-benzene) prepared from 4-hydroxymethyl-phenol and 1-bromomethyl-4-methoxy-benzene in the manner analogous to Examples 14A and 14B. MS m/z 465 (M+1).
Step 2. Preparation of {7-[4-(4-Methoxy-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 68)
The title compound was prepared in the manner analogous to Example 1 using 68A. mp 183-185° C.; IR (KBr) cm−1: 3015, 2588, 1742, 1716, 1514, 1243; 400 MHz 1H NMR (DMSO-d6): δ 12.97 (br(s), 1H), 7.26-7.34 (m, 2H), 7.06-7.13 (m, 2H) 7.04 (d, 1H, J=8.5 Hz), 6.80-6.92 (m, 4H), 6.56 (d, 1H, J=8.5 Hz), 4.92 (s, 2H), 4.60 (s, 2H), 3.93 (s, 2H), 3.69 (s, 3H), 2.75 (t, 2H, J=7.3 Hz), 2.67 (t, 2H, J=7.3 Hz), 1.89 (pentet, 2H); MS m/z 449 (M−1). Anal. Calc'd for C26H26O5S: C, 69.31; H, 5.82; found: C, 69.00; H, 5.74.
Step 1. Preparation of {7-[4-(4-Methoxy-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 68A)
The title compound was prepared in the manner analogous to Example 1F using 12C and 4-chloromethyl-(4-methoxy-benzyloxy-benzene) prepared from 4-hydroxymethyl-phenol and 1-bromomethyl-4-methoxy-benzene in the manner analogous to Examples 14A and 14B. MS m/z 465 (M+1).
Step 2. Preparation of {7-[4-(4-Methoxy-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 68)
The title compound was prepared in the manner analogous to Example 1 using 68A. mp 183-185° C.; IR (KBr) cm−1: 3015, 2588, 1742, 1716, 1514, 1243; 400 MHz 1H NMR (DMSO-d6): δ 12.97 (br(s), 1H), 7.26-7.34 (m, 2H), 7.06-7.13 (m, 2H) 7.04 (d, 1H, J=8.5 Hz), 6.80-6.92 (m, 4H), 6.56 (d, 1H, J=8.5 Hz), 4.92 (s, 2H), 4.60 (s, 2H), 3.93 (s, 2H), 3.69 (s, 3H), 2.75 (t, 2H, J=7.3 Hz), 2.67 (t, 2H, J=7.3 Hz), 1.89 (pentet, 2H); MS m/z 449 (M−1). Anal. Calc'd for C26H26O5S: C, 69.31; H, 5.82; found: C, 69.00; H, 5.74.
Synthesis of [7-(4Benzyloxy-benzylsulfanyl)-indan-4-yloxy]-acetic acid (compound 69)
Step 1. Preparation of [7-(4-Benzyloxy-benzylsulfanyl)-indan-4-yloxy]-acetic acid methyl ester (compound 69A)
The title compound was prepared in the manner analogous to Example 1F using 12C and 1-benzyloxy-4-chloromethyl-benzene prepared from 4-hydroxymethyl-phenol and 4-bromomethyl-benzene in the manner analogous to Examples 14A and 14B. MS m/z 435 (M+1).
Step 2. Preparation of [7-(4-Benzyloxy-benzylsulfanyl)-indan-4-yloxy]-acetic acid (compound 69)
The title compound was prepared in the manner analogous to Example 1 using 69A. mp 145-147° C.; IR (KBr) cm−1: 3066, 2584, 1742, 1511, 1234; 400 MHz 1H NMR (DMSO-d6): δ 12.94 (br(s), 1H), 7.21-7.44 (m, 5H), 6.99-7.15 (m, 3H) 6.80-6.90 (m, 2H), 6.56 (d, 1H, J=8.3 Hz), 5.01 (s, 2H), 4.61 (s, 2H), 3.93 (s, 2H), 2.75 (t, 2H, J=7.3 Hz), 2.67 (t, 2H, J=7.3 Hz), 1.89 (pentet, 2H); MS m/z 419 (M−1). Anal. Calc'd for C25H24O4S: C, 71.40; H, 5.75; found: C, 71.46; H, 5.75.
Step 1. Preparation of [7-(4-Benzyloxy-benzylsulfanyl)-indan-4-yloxy]-acetic acid methyl ester (compound 69A)
The title compound was prepared in the manner analogous to Example 1F using 12C and 1-benzyloxy-4-chloromethyl-benzene prepared from 4-hydroxymethyl-phenol and 4-bromomethyl-benzene in the manner analogous to Examples 14A and 14B. MS m/z 435 (M+1).
Step 2. Preparation of [7-(4-Benzyloxy-benzylsulfanyl)-indan-4-yloxy]-acetic acid (compound 69)
The title compound was prepared in the manner analogous to Example 1 using 69A. mp 145-147° C.; IR (KBr) cm−1: 3066, 2584, 1742, 1511, 1234; 400 MHz 1H NMR (DMSO-d6): δ 12.94 (br(s), 1H), 7.21-7.44 (m, 5H), 6.99-7.15 (m, 3H) 6.80-6.90 (m, 2H), 6.56 (d, 1H, J=8.3 Hz), 5.01 (s, 2H), 4.61 (s, 2H), 3.93 (s, 2H), 2.75 (t, 2H, J=7.3 Hz), 2.67 (t, 2H, J=7.3 Hz), 1.89 (pentet, 2H); MS m/z 419 (M−1). Anal. Calc'd for C25H24O4S: C, 71.40; H, 5.75; found: C, 71.46; H, 5.75.
Synthesis of {7-[4-(4-Chloro-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 70)
Step 1. Preparation of {7-[4(4Chloro-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 70A)
The title compound was prepared in the manner analogous to Example 1F using 12C and 1-(4-chloro-benzyloxy) 4chloromethyl benzene prepared from 1-bromomethyl-4-chloro-benzene and 4-hydroxymethyl-phenol in the manner analogous to Examples 14A and 14B. MS m/z 469 (M+1).
Step 4. Preparation of {7-[4-(4-Chloro-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 70)
The title compound was prepared in the manner analogous to Example 1 using 70A. mp 170-172° C.; IR (KBr) cm−1: 3054, 25.77, 1731, 1710, 1471, 1234; 400 MHz 1H NMR (DMSO-d6): δ 12.95 (br(s), 1H), 7.39 (s, 4H), 6.99-7.25 (m, 3H), 6.79-6.97 (m, 2H), 6.56 (d, 1H, J=8.4 Hz), 5.01 (s, 2H), 4.60 (s, 2H), 3.93 (s, 2H), 2.74 (t, 2H, J=7.4 Hz), 2.66 (t, 2H, J=7.4 Hz), 1.88 (pentet, 2H); MS m/z 453 (M−1). Anal. Calc'd for C25H23ClO4S: C, 66.00; H, 5.10; found: C, 65.95; H, 4.97.
Step 1. Preparation of {7-[4(4Chloro-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 70A)
The title compound was prepared in the manner analogous to Example 1F using 12C and 1-(4-chloro-benzyloxy) 4chloromethyl benzene prepared from 1-bromomethyl-4-chloro-benzene and 4-hydroxymethyl-phenol in the manner analogous to Examples 14A and 14B. MS m/z 469 (M+1).
Step 4. Preparation of {7-[4-(4-Chloro-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 70)
The title compound was prepared in the manner analogous to Example 1 using 70A. mp 170-172° C.; IR (KBr) cm−1: 3054, 25.77, 1731, 1710, 1471, 1234; 400 MHz 1H NMR (DMSO-d6): δ 12.95 (br(s), 1H), 7.39 (s, 4H), 6.99-7.25 (m, 3H), 6.79-6.97 (m, 2H), 6.56 (d, 1H, J=8.4 Hz), 5.01 (s, 2H), 4.60 (s, 2H), 3.93 (s, 2H), 2.74 (t, 2H, J=7.4 Hz), 2.66 (t, 2H, J=7.4 Hz), 1.88 (pentet, 2H); MS m/z 453 (M−1). Anal. Calc'd for C25H23ClO4S: C, 66.00; H, 5.10; found: C, 65.95; H, 4.97.
Synthesis of {7-[4-(2,5-Dichloro-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 71)
Step 1. Preparation of {7-[4(2,5-Dichloro-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 71A)
The title compound was prepared in the manner analogous to Example 1F using 12C and 1,4-Dichloro-2-(4-chloromethyl-phenoxymethyl)-benzene prepared from 1,4-dichloro-2-chloromethyl-benzene and 4-hydroxymethyl-phenol in the manner analogous to Examples 14A and 14B. MS m/z 265 (M−237).
Step 2. Preparation of {7-[4-(2,5-Dichloro-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 71)
The title compound was prepared in the manner analogous to Example 1 using 71A. mp 158-160° C.; IR (KBr) cm−1: 3070, 2573, 1747, 1716, 1236, 1106; 400 MHz 1H NMR (DMSO-d6): δ 12.95 (br(s), 1H), 7.57-7.62 (m, 1H), 7.47-7.53 (m, 1H), 7.38-7.45 (m, 1H), 7.08-7.16 (m, 2H), 7.04 (d, 1H, J=8.5 Hz), 6.85-6.92 (m, 2H), 6.56 (d, 1H, J=8.5 Hz), 5.06 (s, 2H), 4.61 (s, 2H), 3.94 (s, 2H), 2.75 (t, 2H, J=7.3 Hz), 2.67 (t, 2H, J=7.3 Hz), 1.89 (pentet, 2H); MS m/z 487 (M−1). Anal. Calc'd for C25H22Cl2O4S: C, 61.35; H, 4.53; found: C, 61.24; H, 4.43.
Step 1. Preparation of {7-[4(2,5-Dichloro-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 71A)
The title compound was prepared in the manner analogous to Example 1F using 12C and 1,4-Dichloro-2-(4-chloromethyl-phenoxymethyl)-benzene prepared from 1,4-dichloro-2-chloromethyl-benzene and 4-hydroxymethyl-phenol in the manner analogous to Examples 14A and 14B. MS m/z 265 (M−237).
Step 2. Preparation of {7-[4-(2,5-Dichloro-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 71)
The title compound was prepared in the manner analogous to Example 1 using 71A. mp 158-160° C.; IR (KBr) cm−1: 3070, 2573, 1747, 1716, 1236, 1106; 400 MHz 1H NMR (DMSO-d6): δ 12.95 (br(s), 1H), 7.57-7.62 (m, 1H), 7.47-7.53 (m, 1H), 7.38-7.45 (m, 1H), 7.08-7.16 (m, 2H), 7.04 (d, 1H, J=8.5 Hz), 6.85-6.92 (m, 2H), 6.56 (d, 1H, J=8.5 Hz), 5.06 (s, 2H), 4.61 (s, 2H), 3.94 (s, 2H), 2.75 (t, 2H, J=7.3 Hz), 2.67 (t, 2H, J=7.3 Hz), 1.89 (pentet, 2H); MS m/z 487 (M−1). Anal. Calc'd for C25H22Cl2O4S: C, 61.35; H, 4.53; found: C, 61.24; H, 4.43.
Synthesis of {7-[4-(3,4-Dichloro-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 72)
Step 1. Preparation of {7-[4(3,4Dichloro-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 72A)
The title compound was prepared in the manner analogous to Example 1F using 12C and 1,2-Dichloro-4-(4-chloromethyl-phenoxymethyl)-benzene prepared from 1-bromomethyl-3,4-dichloro-benzene and 4-hydroxymethyl-phenol in the manner analogous to Examples 14A and 14B. MS m/z 503 (M+1).
Step 2. Preparation of {7-[4-(3,4Dichloro-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 72)
The title compound was prepared in the manner analogous to Example 1 using 72A. mp 168° C.; IR (KBr) cm−1: 3084, 3039, 1744, 1708, 1244, 1226; 400 MHz 1H NMR (DMSO-d6): δ 12.95 (br(s), 1H), 7.57-7.67 (m, 2H), 7.37 (dd, 1H, J=8.3, 2.0 Hz), 7.01-7.14 (m, 3H), 6.82-6.89 (m, 2H), 6.56 (d, 1H, J=8.4 Hz), 5.04 (s, 2H), 4.61 (s, 2H), 3.93 (s, 2H), 2.75 (t, 2H, J=7.4 Hz), 2.66 (t, 2H, J=7.4 Hz), 1.88 (pentet, 2H); MS m/z 487 (M−1). Anal. Calc'd for C25H22Cl2O4S: C, 61.35; H, 4.53; found: C, 61.13; H, 4.38.
Step 1. Preparation of {7-[4(3,4Dichloro-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 72A)
The title compound was prepared in the manner analogous to Example 1F using 12C and 1,2-Dichloro-4-(4-chloromethyl-phenoxymethyl)-benzene prepared from 1-bromomethyl-3,4-dichloro-benzene and 4-hydroxymethyl-phenol in the manner analogous to Examples 14A and 14B. MS m/z 503 (M+1).
Step 2. Preparation of {7-[4-(3,4Dichloro-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 72)
The title compound was prepared in the manner analogous to Example 1 using 72A. mp 168° C.; IR (KBr) cm−1: 3084, 3039, 1744, 1708, 1244, 1226; 400 MHz 1H NMR (DMSO-d6): δ 12.95 (br(s), 1H), 7.57-7.67 (m, 2H), 7.37 (dd, 1H, J=8.3, 2.0 Hz), 7.01-7.14 (m, 3H), 6.82-6.89 (m, 2H), 6.56 (d, 1H, J=8.4 Hz), 5.04 (s, 2H), 4.61 (s, 2H), 3.93 (s, 2H), 2.75 (t, 2H, J=7.4 Hz), 2.66 (t, 2H, J=7.4 Hz), 1.88 (pentet, 2H); MS m/z 487 (M−1). Anal. Calc'd for C25H22Cl2O4S: C, 61.35; H, 4.53; found: C, 61.13; H, 4.38.
Synthesis of {7-[4-(4Chloro-3-trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 73)
Step 1. Preparation of {7-[4-(4-Chloro-3-trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 73A)
The title compound was prepared in the manner analogous to Example 1F using 12C and 1-Chloro-4-(4-chloromethyl-phenoxymethyl)-2-trifluoromethyl-benzene prepared from 1-bromomethyl-4-chloro-3-trifluoromethyl-benzene and 4-hydroxymethyl-phenol in the manner analogous to Examples 14A and 14B. MS m/z 299 (M−237).
Step 2. Preparation of {7-[4-(4-Chloro-3-trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 73)
The title compound was prepared in the manner analogous to Example 1 using 73A. mp 151-152° C.; IR (KBr) cm−1: 3076, 3050, 1748, 1426, 1244, 1109; 400 MHz 1H NMR (DMSO-d6): δ 12.94 (br(s), 1H), 7.87 (s, 1H), 7.68-7.72 (m, 2H) 7.00-7.15 (m, 3H), 6.82-6.92 (m, 2H), 6.56 (d, 1H, J=8.5 Hz), 5.12 (s, 2H), 4.61 (s, 2H), 3.94 (s, 2H), 2.74 (t, 2H, J=7.5 Hz), 2.66 (t, 2H, J=7.5 Hz), 1.87 (pentet, 2H); MS m/z 521 (M−1). Anal. Calc'd for C26H22ClF3O4S: C, 59.71; H, 4.24; found: C, 59.54; H, 4.11.
Step 1. Preparation of {7-[4-(4-Chloro-3-trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 73A)
The title compound was prepared in the manner analogous to Example 1F using 12C and 1-Chloro-4-(4-chloromethyl-phenoxymethyl)-2-trifluoromethyl-benzene prepared from 1-bromomethyl-4-chloro-3-trifluoromethyl-benzene and 4-hydroxymethyl-phenol in the manner analogous to Examples 14A and 14B. MS m/z 299 (M−237).
Step 2. Preparation of {7-[4-(4-Chloro-3-trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 73)
The title compound was prepared in the manner analogous to Example 1 using 73A. mp 151-152° C.; IR (KBr) cm−1: 3076, 3050, 1748, 1426, 1244, 1109; 400 MHz 1H NMR (DMSO-d6): δ 12.94 (br(s), 1H), 7.87 (s, 1H), 7.68-7.72 (m, 2H) 7.00-7.15 (m, 3H), 6.82-6.92 (m, 2H), 6.56 (d, 1H, J=8.5 Hz), 5.12 (s, 2H), 4.61 (s, 2H), 3.94 (s, 2H), 2.74 (t, 2H, J=7.5 Hz), 2.66 (t, 2H, J=7.5 Hz), 1.87 (pentet, 2H); MS m/z 521 (M−1). Anal. Calc'd for C26H22ClF3O4S: C, 59.71; H, 4.24; found: C, 59.54; H, 4.11.
Synthesis of {7-[4-(4-Fluoro-3-trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 74)
Step 1. Preparation of {7-[4-(4-Fluoro-3-trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 74A)
The title compound was prepared in the manner analogous to Example 1F using 12C and 4-(4-Chloromethyl-phenoxymethyl)-1-fluoro-2-trifluoromethyl-benzene prepared from 1-bromomethyl-4-fluoro-3-trifluoromethyl-benzene and 4-hydroxymethyl-phenol in the manner analogous to Examples 14A and 14B. MS m/z 283 (M−237).
Step 2. Preparation of {7-[4-(4-Fluoro-3-trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 74)
The title compound was prepared in the manner analogous to Example 1 using 74A. mp 116-117° C.; IR (KBr) cm−1: 3028, 1741, 1704, 1511, 1235, 1110; 400 MHz 1H NMR (DMSO-d6): δ 12.96 (br(s), 1H), 7.74-7.85 (m, 2H), 7.45-7.54 (m, 1H), 7.08-7.15 (m, 2H), 7.04 (d, 1H, J=8.5 Hz), 6.84-6.91 (m, 2H), 6.56 (d, 1H, J=8.5 Hz), 5.09 (s, 2H), 4.61 (s, 2H), 3.94 (s, 2H), 2.75 (t, 2H, J=7.5 Hz), 2.66 (t, 2H, J=7.5 Hz), 1.88 (pentet, 2H); MS m/z 505 (M−1). Anal. Calc'd for C26H22F4O4S: C, 61.65; H, 4.38; found: C, 61.50; H, 4.07.
Step 1. Preparation of {7-[4-(4-Fluoro-3-trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 74A)
The title compound was prepared in the manner analogous to Example 1F using 12C and 4-(4-Chloromethyl-phenoxymethyl)-1-fluoro-2-trifluoromethyl-benzene prepared from 1-bromomethyl-4-fluoro-3-trifluoromethyl-benzene and 4-hydroxymethyl-phenol in the manner analogous to Examples 14A and 14B. MS m/z 283 (M−237).
Step 2. Preparation of {7-[4-(4-Fluoro-3-trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 74)
The title compound was prepared in the manner analogous to Example 1 using 74A. mp 116-117° C.; IR (KBr) cm−1: 3028, 1741, 1704, 1511, 1235, 1110; 400 MHz 1H NMR (DMSO-d6): δ 12.96 (br(s), 1H), 7.74-7.85 (m, 2H), 7.45-7.54 (m, 1H), 7.08-7.15 (m, 2H), 7.04 (d, 1H, J=8.5 Hz), 6.84-6.91 (m, 2H), 6.56 (d, 1H, J=8.5 Hz), 5.09 (s, 2H), 4.61 (s, 2H), 3.94 (s, 2H), 2.75 (t, 2H, J=7.5 Hz), 2.66 (t, 2H, J=7.5 Hz), 1.88 (pentet, 2H); MS m/z 505 (M−1). Anal. Calc'd for C26H22F4O4S: C, 61.65; H, 4.38; found: C, 61.50; H, 4.07.
Synthesis of {7-[4-(4-Trifluoromethoxy-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 75)
Step 1. Preparation of [7-(4-Acetoxy-benzylsulfanyl)-indan-4-yloxy]-acetic acid methyl ester (compound 75A)
The title compound was prepared in the manner analogous to Example 35A using acetic acid 4-chloromethyl-phenyl ester and 12C. MS m/z 387 (M+1).
Step 2. Preparation of [7-(4-Hydroxy-benzylsulfanyl)-indan-4-yloxy]-acetic acid (compound 75B)
The title compound was prepared in the manner analogous to Example 35B using 75A. MS m/z 391 (M−1).
Step 3. Preparation of [7-(4-Hydroxy-benzylsulfanyl)-indan-4-yloxy]-acetic acid methyl ester (compound 75C)
The title compound was prepared in the manner analogous to Example 35C using the product from Example 75B. MS m/z 237 (M−107).
Step 4. Preparation of {7-[4-(4-Trifluoromethoxy-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 75D)
The title compound was prepared in the manner analogous to Example 1F using 1-bromomethyl-4-trifluoromethoxy-benzene and 75C. MS m/z 519 (M+1).
Step 5. Preparation of {7-[4-(4-Trifluoromethoxy-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 75)
The title compound was prepared in the manner analogous to Example 1 using 75D. mp 140-142° C.; IR (KBr) cm−1: 3072, 3043, 1724, 1511, 1226, 1156; 400 MHz 1H NMR (DMSO-d6): δ 12.95 (br(s), 1H), 7.47-7.57 (m, 2H), 7.29-7.39 (m, 2H), 7.00-7.16 (m, 3H), 6.81-6.91 (m, 2H), 6.56 (d, 1H, J=8.5 Hz), 5.05 (s, 2H), 4.61 (s, 2H), 3.94 (s, 2H), 2.75 (t, 2H, J=7.4 Hz), 2.67 (t, 2H, J=7.4 Hz), 1.88 (pentet, 2H) MS m/z 503 (M−1). Anal. Calc'd for C26H23F3O3S: C, 61.90; H, 4.60; found: C, 61.53; H, 4.36.
Step 1. Preparation of [7-(4-Acetoxy-benzylsulfanyl)-indan-4-yloxy]-acetic acid methyl ester (compound 75A)
The title compound was prepared in the manner analogous to Example 35A using acetic acid 4-chloromethyl-phenyl ester and 12C. MS m/z 387 (M+1).
Step 2. Preparation of [7-(4-Hydroxy-benzylsulfanyl)-indan-4-yloxy]-acetic acid (compound 75B)
The title compound was prepared in the manner analogous to Example 35B using 75A. MS m/z 391 (M−1).
Step 3. Preparation of [7-(4-Hydroxy-benzylsulfanyl)-indan-4-yloxy]-acetic acid methyl ester (compound 75C)
The title compound was prepared in the manner analogous to Example 35C using the product from Example 75B. MS m/z 237 (M−107).
Step 4. Preparation of {7-[4-(4-Trifluoromethoxy-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 75D)
The title compound was prepared in the manner analogous to Example 1F using 1-bromomethyl-4-trifluoromethoxy-benzene and 75C. MS m/z 519 (M+1).
Step 5. Preparation of {7-[4-(4-Trifluoromethoxy-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 75)
The title compound was prepared in the manner analogous to Example 1 using 75D. mp 140-142° C.; IR (KBr) cm−1: 3072, 3043, 1724, 1511, 1226, 1156; 400 MHz 1H NMR (DMSO-d6): δ 12.95 (br(s), 1H), 7.47-7.57 (m, 2H), 7.29-7.39 (m, 2H), 7.00-7.16 (m, 3H), 6.81-6.91 (m, 2H), 6.56 (d, 1H, J=8.5 Hz), 5.05 (s, 2H), 4.61 (s, 2H), 3.94 (s, 2H), 2.75 (t, 2H, J=7.4 Hz), 2.67 (t, 2H, J=7.4 Hz), 1.88 (pentet, 2H) MS m/z 503 (M−1). Anal. Calc'd for C26H23F3O3S: C, 61.90; H, 4.60; found: C, 61.53; H, 4.36.
Synthesis of {7-[4-(4-Fluoro-2-trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 76)
Step 1. Preparation of {7-[4-(4-Fuoro-2-trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 76A)
The title compound was prepared in the manner analogous to Example 1F using 1-bromomethyl-fluoro-2-trifluoromethyl-benzene and the product prepared from Example 75C. MS m/z 283
Step 2. Preparation of {7-[4-(4-Fluoro-2-trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 76)
The title compound was prepared in the manner analogous to Example 1 using 76A. mp 125-127° C.; IR (KBr) cm−1: 3132, 3032, 1742, 1708, 1244, 1110; 400 MHz 1H NMR (DMSO-d6): δ 12.95 (br(s), 1H), 7.71-7.81 (m, 1H), 7.65 (dd, 1H, J=9.3, 2.6 Hz), 7.51-7.61 (m, 1H), 7.01-7.19 (m, 3H), 6.81-6.91 (m, 2H), 6.57 (d, 1H, J=8.5 Hz), 5.11 (s, 2H), 4.61 (s, 2H), 3.95 (s, 2H), 2.75 (t, 2H, J=7.4 Hz), 2.66 (t, 2H, J=7.4 Hz), 1.89 (pentet, 2H); MS m/z 502 (M−1). Anal. Calc'd for C26H22F4O4S: C, 61.65; H, 4.38; found: C, 61.28; H, 4.12.
Step 1. Preparation of {7-[4-(4-Fuoro-2-trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 76A)
The title compound was prepared in the manner analogous to Example 1F using 1-bromomethyl-fluoro-2-trifluoromethyl-benzene and the product prepared from Example 75C. MS m/z 283
Step 2. Preparation of {7-[4-(4-Fluoro-2-trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 76)
The title compound was prepared in the manner analogous to Example 1 using 76A. mp 125-127° C.; IR (KBr) cm−1: 3132, 3032, 1742, 1708, 1244, 1110; 400 MHz 1H NMR (DMSO-d6): δ 12.95 (br(s), 1H), 7.71-7.81 (m, 1H), 7.65 (dd, 1H, J=9.3, 2.6 Hz), 7.51-7.61 (m, 1H), 7.01-7.19 (m, 3H), 6.81-6.91 (m, 2H), 6.57 (d, 1H, J=8.5 Hz), 5.11 (s, 2H), 4.61 (s, 2H), 3.95 (s, 2H), 2.75 (t, 2H, J=7.4 Hz), 2.66 (t, 2H, J=7.4 Hz), 1.89 (pentet, 2H); MS m/z 502 (M−1). Anal. Calc'd for C26H22F4O4S: C, 61.65; H, 4.38; found: C, 61.28; H, 4.12.
Synthesis of {7-[4-(3,5-Dichloro-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 77)
Step 1. Preparation of {7-[4(3,5-Dichloro-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 77A)
The product from Example 75C (0.42 g, 1 mmoles), (3,5-dichlorophenyl)-methanol (0.24 g, 1.3 mmoles), triphenyl phosphine (0.38 g, 1.5 mmoles) and 0.23 mL of diethyl azodicarboxylate (0.25 g, 1.5 mmoles) were dissolved in 9 mL of tetrahydrofuran. The reaction mixture was stirred at room temperature under nitrogen for 18 hrs. The reaction mixture was evaporated to give a residue, which was flash chromatographed (silica gel, 20% ethyl acetate in hexane) to afford the title compound in good purity. MS m/z 265
Step 2. Preparation of {7-[4-(3,5-Dichloro-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 77)
The title compound was prepared in the manner analogous to Example 1 using 77A. mp 134-135° C.; IR (KBr) cm−1: 3070, 1747, 1708, 1572, 1432, 1244; 400 MHz 1H NMR (DMSO-d6): δ 12.95 (br(s), 1H), 7.51-7.53 (m, 1H), 7.42-7.45 (m, 2H), 7.07-7.13 (m, 2H), 7.04 (d, 1H, J=8.6 Hz), 6.83-6.90 (m, 2H), 6.56 (d, 1H, J=8.6 Hz), 5.05 (s, 2H), 4.61 (s, 2H), 3.93 (s, 2H), 2.75 (t, 2H, J=7.5 Hz), 2.66 (t, 2H, 7.5 Hz), 1.88 (pentet, 2H); MS m/z 487 (M−1). Anal. Calc'd for C25H22Cl2O4S: C, 61.35; H, 4.53; found: C, 61.01; H, 436.
Step 1. Preparation of {7-[4(3,5-Dichloro-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 77A)
The product from Example 75C (0.42 g, 1 mmoles), (3,5-dichlorophenyl)-methanol (0.24 g, 1.3 mmoles), triphenyl phosphine (0.38 g, 1.5 mmoles) and 0.23 mL of diethyl azodicarboxylate (0.25 g, 1.5 mmoles) were dissolved in 9 mL of tetrahydrofuran. The reaction mixture was stirred at room temperature under nitrogen for 18 hrs. The reaction mixture was evaporated to give a residue, which was flash chromatographed (silica gel, 20% ethyl acetate in hexane) to afford the title compound in good purity. MS m/z 265
Step 2. Preparation of {7-[4-(3,5-Dichloro-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 77)
The title compound was prepared in the manner analogous to Example 1 using 77A. mp 134-135° C.; IR (KBr) cm−1: 3070, 1747, 1708, 1572, 1432, 1244; 400 MHz 1H NMR (DMSO-d6): δ 12.95 (br(s), 1H), 7.51-7.53 (m, 1H), 7.42-7.45 (m, 2H), 7.07-7.13 (m, 2H), 7.04 (d, 1H, J=8.6 Hz), 6.83-6.90 (m, 2H), 6.56 (d, 1H, J=8.6 Hz), 5.05 (s, 2H), 4.61 (s, 2H), 3.93 (s, 2H), 2.75 (t, 2H, J=7.5 Hz), 2.66 (t, 2H, 7.5 Hz), 1.88 (pentet, 2H); MS m/z 487 (M−1). Anal. Calc'd for C25H22Cl2O4S: C, 61.35; H, 4.53; found: C, 61.01; H, 436.
Synthesis of {7-[4-Methoxy-3-(4-trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 78)
Step 1. Preparation of [4-Methoxy-3-(4-trifluoromethyl-benzyloxy)-phenyl]-methanol (compound 78A)
The title compound was prepared in the manner analogous to Example 14A using 1-bromomethyl-4-trifluoromethyl-benzene and 5-hydroxymethyl-2-methoxy-phenol. MS m/z 295 (M-OH).
Step 2. Preparation of 4-Chloromethyl-1-methoxy-2-(4-trifluoromethyl-benzyloxy)-benzene (compound 78B)
The title compound was prepared in the manner analogous to Example 3B using 78A. MS m/z 295 (M-Cl).
Step 3. Preparation of {7-[4-Methoxy-3-(4-trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 78C)
The title compound was prepared in the manner analogous to Example 1F using 78B and 12C. MS m/z 373 (M−159).
Step 4. Preparation of {7-[4-Methoxy-3-(4-trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 78)
The title compound was prepared in the manner analogous to Example 1 using 78C. mp 150-151° C.; IR (KBr) cm−1: 3046, 1722, 1515, 1328, 1232, 1106; 400 MHz 1H NMR (DMSO-d6): δ 12.95 (br(s), 1H), 7.71 (d, 2H, J=8.1 Hz), 7.58 (d, 2H, J=8.1 Hz), 7.03 (d, 1H, J=8.5 Hz), 6.70-6.86 (m, 3H), 6.57 (d, 1H, J=8.5 Hz), 5.01 (s, 2H), 4.61 (s, 2H), 3.89 (s, 2H), 3.69 (s, 3H), 2.75 (t, 2H, J=7.3 Hz), 2.62 (t, 2H, J=7.3 Hz), 1.87 (pentet, 2H); MS m/z 517 (M−1). Anal. Calc'd for C27 H25F3O3S: C, 62.54; H, 4.86; found: C, 62.54; H, 4.71.
Step 1. Preparation of [4-Methoxy-3-(4-trifluoromethyl-benzyloxy)-phenyl]-methanol (compound 78A)
The title compound was prepared in the manner analogous to Example 14A using 1-bromomethyl-4-trifluoromethyl-benzene and 5-hydroxymethyl-2-methoxy-phenol. MS m/z 295 (M-OH).
Step 2. Preparation of 4-Chloromethyl-1-methoxy-2-(4-trifluoromethyl-benzyloxy)-benzene (compound 78B)
The title compound was prepared in the manner analogous to Example 3B using 78A. MS m/z 295 (M-Cl).
Step 3. Preparation of {7-[4-Methoxy-3-(4-trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 78C)
The title compound was prepared in the manner analogous to Example 1F using 78B and 12C. MS m/z 373 (M−159).
Step 4. Preparation of {7-[4-Methoxy-3-(4-trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 78)
The title compound was prepared in the manner analogous to Example 1 using 78C. mp 150-151° C.; IR (KBr) cm−1: 3046, 1722, 1515, 1328, 1232, 1106; 400 MHz 1H NMR (DMSO-d6): δ 12.95 (br(s), 1H), 7.71 (d, 2H, J=8.1 Hz), 7.58 (d, 2H, J=8.1 Hz), 7.03 (d, 1H, J=8.5 Hz), 6.70-6.86 (m, 3H), 6.57 (d, 1H, J=8.5 Hz), 5.01 (s, 2H), 4.61 (s, 2H), 3.89 (s, 2H), 3.69 (s, 3H), 2.75 (t, 2H, J=7.3 Hz), 2.62 (t, 2H, J=7.3 Hz), 1.87 (pentet, 2H); MS m/z 517 (M−1). Anal. Calc'd for C27 H25F3O3S: C, 62.54; H, 4.86; found: C, 62.54; H, 4.71.
Synthesis of {7-[3-(4-Trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 79)
Step 1. Preparation of [3-(4-trifluoromethyl-benzyloxy)-phenyl]-methanol (compound 79A)
The title compound was prepared in the manner analogous to Example 14A using 1-bromomethyl-4-trifluoromethyl-benzene and 3-hydroxymethyl-phenol. MS m/z 265(M-OH).
Step 2. Preparation of 1-(4-trifluoromethyl-benzyloxy)-3-chloromethyl-benzene (compound 79B)
The title compound was prepared in the manner analogous to Example 3B using 79A. MS m/z 265 (M-Cl).
Step 3. Preparation of {7-[3-(4-Trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 79C)
The title compound was prepared in the manner analogous to Example 1F using 79B and 12C. MS m/z 503 (M+1).
Step 4. Preparation of {7-[3-(4-Trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 79)
The title compound was prepared in the manner analogous to Example 1 using 79C. mp 145-146° C.; IR (KBr) cm−1: 3076, 3028, 1705, 1318, 1232, 1109; 400 MHz 1H NMR (DMSO-d6): δ 12.94 (br(s), 1H), 7.71 (d, 2H, J=8.2 Hz), 7.59 (d, 2H, J=8.0 Hz), 7.10-7.19 (m, 1H), 7.05 (d, 1H, J=8.5 Hz), 6.75-6.86 (m, 4H), 6.58 (d, 1H, J=8.5 Hz), 5.08 (s, 2H), 4.61 (s, 2H), 3.95 (s, 2H), 2.75 (t, 2H, J=7.5 Hz), 2.64 (t, 2H, J=7.5 Hz), 1.88 (pentet, 2H); MS m/z 487 (M−1). Anal. Calc'd for C26H23F3O4S: C, 63.92; H, 4.75; found: C, 63.78; H, 4.53.
Step 1. Preparation of [3-(4-trifluoromethyl-benzyloxy)-phenyl]-methanol (compound 79A)
The title compound was prepared in the manner analogous to Example 14A using 1-bromomethyl-4-trifluoromethyl-benzene and 3-hydroxymethyl-phenol. MS m/z 265(M-OH).
Step 2. Preparation of 1-(4-trifluoromethyl-benzyloxy)-3-chloromethyl-benzene (compound 79B)
The title compound was prepared in the manner analogous to Example 3B using 79A. MS m/z 265 (M-Cl).
Step 3. Preparation of {7-[3-(4-Trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 79C)
The title compound was prepared in the manner analogous to Example 1F using 79B and 12C. MS m/z 503 (M+1).
Step 4. Preparation of {7-[3-(4-Trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 79)
The title compound was prepared in the manner analogous to Example 1 using 79C. mp 145-146° C.; IR (KBr) cm−1: 3076, 3028, 1705, 1318, 1232, 1109; 400 MHz 1H NMR (DMSO-d6): δ 12.94 (br(s), 1H), 7.71 (d, 2H, J=8.2 Hz), 7.59 (d, 2H, J=8.0 Hz), 7.10-7.19 (m, 1H), 7.05 (d, 1H, J=8.5 Hz), 6.75-6.86 (m, 4H), 6.58 (d, 1H, J=8.5 Hz), 5.08 (s, 2H), 4.61 (s, 2H), 3.95 (s, 2H), 2.75 (t, 2H, J=7.5 Hz), 2.64 (t, 2H, J=7.5 Hz), 1.88 (pentet, 2H); MS m/z 487 (M−1). Anal. Calc'd for C26H23F3O4S: C, 63.92; H, 4.75; found: C, 63.78; H, 4.53.
Synthesis of {7-[3-(4-Chloro-3-trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 80)
Step 1. Preparation of [3-(4Chloro-3-trifluoromethyl-benzyloxy)-phenyl]-methanol (compound 80A)
The title compound was prepared in the manner analogous to Example 14A using 4-bromomethyl-1chloro-2-trifluoromethyl-benzene and 3-hydroxymethyl-phenol. MS m/z 299 (M-OH).
Step 2. Preparation of 1-Chloro-4-(3-chloromethyl-phenoxymethyl)-2-trifluoromethyl-benzene (compound 80B)
The title compound was prepared in the manner analogous to Example 3B using 80A. MS m/z 299 (M-Cl).
Step 3. Preparation of {7-[3-(4-Chloro-3-trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 80C)
The title compound was prepared in the manner analogous to Example 1F using 80B and 12C. MS m/z 537 (M+1).
Step 4. Preparation of {7-[3-(4-Chloro-3-trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 80)
The title compound was prepared in the manner analogous to Example 1 using 80C. mp 121-122° C.; IR (KBr) cm−1: 3027, 2584, 1742, 1255, 1129, 1109; 400 MHz 1H NMR (DMSO-d6): δ 12.95 (br(s), 1H), 7.87 (s, 1H), 7.64-7.75 (m, 2H), 7.09-7.19 (m, 1H), 7.01-7.09 (m, 1H), 6.73-6.88 (m, 3H), 6.56 (d, 1H, J=8.5 Hz), 5.06 (s, 2H), 4.60 (s, 2H), 3.95 (s, 2H), 2.74 (t, 2H, J=7.4 Hz), 2.63 (t, 2H, J=7.4 Hz), 1.87 (pentet, 2H); MS m/z 523 (M+1). Anal. Calc'd for C26H22ClF3O4S: C, 59.71; H, 4.24; found: C, 59.45; H, 4.08.
Step 1. Preparation of [3-(4Chloro-3-trifluoromethyl-benzyloxy)-phenyl]-methanol (compound 80A)
The title compound was prepared in the manner analogous to Example 14A using 4-bromomethyl-1chloro-2-trifluoromethyl-benzene and 3-hydroxymethyl-phenol. MS m/z 299 (M-OH).
Step 2. Preparation of 1-Chloro-4-(3-chloromethyl-phenoxymethyl)-2-trifluoromethyl-benzene (compound 80B)
The title compound was prepared in the manner analogous to Example 3B using 80A. MS m/z 299 (M-Cl).
Step 3. Preparation of {7-[3-(4-Chloro-3-trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 80C)
The title compound was prepared in the manner analogous to Example 1F using 80B and 12C. MS m/z 537 (M+1).
Step 4. Preparation of {7-[3-(4-Chloro-3-trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 80)
The title compound was prepared in the manner analogous to Example 1 using 80C. mp 121-122° C.; IR (KBr) cm−1: 3027, 2584, 1742, 1255, 1129, 1109; 400 MHz 1H NMR (DMSO-d6): δ 12.95 (br(s), 1H), 7.87 (s, 1H), 7.64-7.75 (m, 2H), 7.09-7.19 (m, 1H), 7.01-7.09 (m, 1H), 6.73-6.88 (m, 3H), 6.56 (d, 1H, J=8.5 Hz), 5.06 (s, 2H), 4.60 (s, 2H), 3.95 (s, 2H), 2.74 (t, 2H, J=7.4 Hz), 2.63 (t, 2H, J=7.4 Hz), 1.87 (pentet, 2H); MS m/z 523 (M+1). Anal. Calc'd for C26H22ClF3O4S: C, 59.71; H, 4.24; found: C, 59.45; H, 4.08.
Synthesis of {7-[2-(4-Trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 81)
Step 1. Preparation of [2-(4-Trifluoromethyl-benzyloxy)-phenyl]-methanol (compound 81A)
The title compound was prepared in the manner analogous to Example 14A using 1-bromomethyl-4-trifluoromethyl-benzene and 2-hydroxymethyl-phenol. MS m/z 265 (M-OH).
Step 2. Preparation of 1-(4-trifluoromethyl-benzyloxy)-2-chloromethyl-benzene (compound 81B)
The title compound was prepared in the manner analogous to Example 3B using 81A. MS m/z 265 (M-Cl).
Step 3. Preparation of {7-[2-(4-Trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 81C)
The title compound was prepared in the manner analogous to Example 1F using 81B and 12C. MS m/z 503 (M+1).
Step 4. Preparation of {7-[2-(4-Trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 81)
The title compound was prepared in the manner analogous to Example 1 using 81C. mp 150-152° C.; IR (KBr) cm−1: 3074, 3042, 1701, 1324, 1124, 1099; 400 MHz 1H NMR (DMSO-d6): δ 7.58-7.75 (m, 2H), 6.92-7.20 (m, 4H), 6.80 (t, 1H, J=7.5 Hz), 6.51 (d, 1H, J=8.5 Hz), 5.16 (s, 2H), 4.57 (s, 2H), 3.99 (s, 2H), 2.70 (t, 2H, J=7.5 Hz), 2.62 (t, 2H, J=7.5 Hz), 1.80 (pentet, 2H); MS m/z 487 (M−1). Anal. Calc'd for C26H23F3O4S: C, 63.92; H, 4.75; found: C, 63.54; H, 4.52.
Step 1. Preparation of [2-(4-Trifluoromethyl-benzyloxy)-phenyl]-methanol (compound 81A)
The title compound was prepared in the manner analogous to Example 14A using 1-bromomethyl-4-trifluoromethyl-benzene and 2-hydroxymethyl-phenol. MS m/z 265 (M-OH).
Step 2. Preparation of 1-(4-trifluoromethyl-benzyloxy)-2-chloromethyl-benzene (compound 81B)
The title compound was prepared in the manner analogous to Example 3B using 81A. MS m/z 265 (M-Cl).
Step 3. Preparation of {7-[2-(4-Trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 81C)
The title compound was prepared in the manner analogous to Example 1F using 81B and 12C. MS m/z 503 (M+1).
Step 4. Preparation of {7-[2-(4-Trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 81)
The title compound was prepared in the manner analogous to Example 1 using 81C. mp 150-152° C.; IR (KBr) cm−1: 3074, 3042, 1701, 1324, 1124, 1099; 400 MHz 1H NMR (DMSO-d6): δ 7.58-7.75 (m, 2H), 6.92-7.20 (m, 4H), 6.80 (t, 1H, J=7.5 Hz), 6.51 (d, 1H, J=8.5 Hz), 5.16 (s, 2H), 4.57 (s, 2H), 3.99 (s, 2H), 2.70 (t, 2H, J=7.5 Hz), 2.62 (t, 2H, J=7.5 Hz), 1.80 (pentet, 2H); MS m/z 487 (M−1). Anal. Calc'd for C26H23F3O4S: C, 63.92; H, 4.75; found: C, 63.54; H, 4.52.
Synthesis of {7-[3-Dichloro-4-(4-trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid lithium salt (compound 82)
Step 1. Preparation of [3,5-Dichloro-(4-trifluoromethyl-benzyloxy)-phenyl]-methanol (compound 82A)
The title compound was prepared in the manner analogous to Example 14A using 1-bromomethyl-4-trifluoromethyl-benzene and 2,6-dichloro-4-hydroxymethyl-phenol. MS m/z 191 (M−159).
Step 2. Preparation of 1,3-Dichloro-5-chloromethyl-2-(4-trifluoromethyl-benzyloxy)-benzene (compound 82B)
The title compound was prepared in the manner analogous to Example 3B using 82A. MS m/z 333 (M-Cl).
Step 3. Preparation of {7-[3,5-Dichloro-4-(4-trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 82C)
The title compound was prepared in the manner analogous to Example 1F using 82B and 12C. MS m/z 571 (M+1).
Step 4. Preparation of The Lithium Salt of {7-[3,5-Dichloro-4(4-trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 82)
The title compound, in unprotonated form, was prepared in the manner analogous to Example 1 using 82C. mp 235° C. dec; IR (KBr) cm−1: 3414, 1622, 1591, 1472, 1326, 1265; 400 MHz 1H NMR (DMSO-d6): δ 7.66-7.77 (m, 4H), 7.26 (s, 2H), 6.99 (d, 1H, J=8.5 Hz), 6.45 (d, 1H, J=8.5 Hz), 5.03 (s, 2H), 4.02 (s, 2H), 3.92 (s, 2H), 2.73 (t, 2H, J=7.3 Hz), 2.65 (t, 2H, J=7.3 Hz), 1.88 (pentet, 2H); MS m/z 555 (M−1). Anal. Calc'd for C26H20Cl2F3O4S; Li: 0.50 H2O: C, 54.56; H, 3.70; Li, 1.21; H2O, 1.57; found: C, 54.63; H, 3.68; Li, 1.46; H2O, 1.61.
Step 1. Preparation of [3,5-Dichloro-(4-trifluoromethyl-benzyloxy)-phenyl]-methanol (compound 82A)
The title compound was prepared in the manner analogous to Example 14A using 1-bromomethyl-4-trifluoromethyl-benzene and 2,6-dichloro-4-hydroxymethyl-phenol. MS m/z 191 (M−159).
Step 2. Preparation of 1,3-Dichloro-5-chloromethyl-2-(4-trifluoromethyl-benzyloxy)-benzene (compound 82B)
The title compound was prepared in the manner analogous to Example 3B using 82A. MS m/z 333 (M-Cl).
Step 3. Preparation of {7-[3,5-Dichloro-4-(4-trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 82C)
The title compound was prepared in the manner analogous to Example 1F using 82B and 12C. MS m/z 571 (M+1).
Step 4. Preparation of The Lithium Salt of {7-[3,5-Dichloro-4(4-trifluoromethyl-benzyloxy)-benzylsulfanyl]-indan-4-yloxy}-acetic acid (compound 82)
The title compound, in unprotonated form, was prepared in the manner analogous to Example 1 using 82C. mp 235° C. dec; IR (KBr) cm−1: 3414, 1622, 1591, 1472, 1326, 1265; 400 MHz 1H NMR (DMSO-d6): δ 7.66-7.77 (m, 4H), 7.26 (s, 2H), 6.99 (d, 1H, J=8.5 Hz), 6.45 (d, 1H, J=8.5 Hz), 5.03 (s, 2H), 4.02 (s, 2H), 3.92 (s, 2H), 2.73 (t, 2H, J=7.3 Hz), 2.65 (t, 2H, J=7.3 Hz), 1.88 (pentet, 2H); MS m/z 555 (M−1). Anal. Calc'd for C26H20Cl2F3O4S; Li: 0.50 H2O: C, 54.56; H, 3.70; Li, 1.21; H2O, 1.57; found: C, 54.63; H, 3.68; Li, 1.46; H2O, 1.61.
Synthesis of {8-[4-(4-Trifluoromethyl-benzyloxy)-benzylsulfanyl]-chroman-5-yloxy}-acetic acid (compound 83)
Step 1. Preparation of {8-[4-(4-Trifluoromethyl-benzyloxy)-benzylsulfanyl]-chroman-5-yloxy}-acetic acid methyl ester (compound 83A)
The title compound was prepared in the manner analogous to Example 1F using (8-mercapto-chroman-5-yloxy)-acetic acid methyl ester and 14B. MS m/z 519 (M+1).
Step 2. Preparation of {8-[4(4-Trifluoromethyl-benzyloxy)-benzylsulfanyl]-chroman-5-yloxy}-acetic acid (compound 83)
The title compound was prepared in the manner analogous to Example 1 using 83A. mp 143-144° C.; HPLC: area %=96.02, r.t.=3.770 min, γ=214 nm, mobile phase=acetonitrile/water w/0.10% TFA; IR (KBr) cm−1: 3042, 2581, 1739, 1714, 1325, 1116; 400 MHz 1H NMR (DMSO-d6): δ 12.95 (br(s), 1H), 7.66-7.74 (m, 2H), 7.56-7.63 (m, 2H), 7.10-7.17 (m, 2H), 6.92 (d, 1H, J=8.7 Hz), 6.83-6.89 (m, 2H), 6.26 (d, 1H, J=8.5 Hz), 5.13 (s, 2H), 4.58 (s, 2H), 4.08 (t, 2H, J=4.8 Hz), 3.90 (s, 2H), 2.55 (t, 2H, J=6.5 Hz), 1.83 (pentet, 2H); MS m/z 503 (M−1). Anal. Calc'd for C26H23F3O5S: C, 61.90; H, 4.60; found: C, 61.41; H, 4.50.
Step 1. Preparation of {8-[4-(4-Trifluoromethyl-benzyloxy)-benzylsulfanyl]-chroman-5-yloxy}-acetic acid methyl ester (compound 83A)
The title compound was prepared in the manner analogous to Example 1F using (8-mercapto-chroman-5-yloxy)-acetic acid methyl ester and 14B. MS m/z 519 (M+1).
Step 2. Preparation of {8-[4(4-Trifluoromethyl-benzyloxy)-benzylsulfanyl]-chroman-5-yloxy}-acetic acid (compound 83)
The title compound was prepared in the manner analogous to Example 1 using 83A. mp 143-144° C.; HPLC: area %=96.02, r.t.=3.770 min, γ=214 nm, mobile phase=acetonitrile/water w/0.10% TFA; IR (KBr) cm−1: 3042, 2581, 1739, 1714, 1325, 1116; 400 MHz 1H NMR (DMSO-d6): δ 12.95 (br(s), 1H), 7.66-7.74 (m, 2H), 7.56-7.63 (m, 2H), 7.10-7.17 (m, 2H), 6.92 (d, 1H, J=8.7 Hz), 6.83-6.89 (m, 2H), 6.26 (d, 1H, J=8.5 Hz), 5.13 (s, 2H), 4.58 (s, 2H), 4.08 (t, 2H, J=4.8 Hz), 3.90 (s, 2H), 2.55 (t, 2H, J=6.5 Hz), 1.83 (pentet, 2H); MS m/z 503 (M−1). Anal. Calc'd for C26H23F3O5S: C, 61.90; H, 4.60; found: C, 61.41; H, 4.50.
Synthesis of {8-[3-(4-Trifluoromethyl-benzyloxy)-benzylsulfanyl]-chroman-5-yloxy}-acetic acid (compound 84)
Step 1. Preparation of {8-[3-(4-Trifluoromethyl-benzyloxy)-benzylsulfanyl]-chroman-5-yloxy}-acetic acid methyl ester (compound 84A)
The title compound was prepared in the manner analogous to Example 1F using (8-mercapto-chroman-5-yloxy)-acetic acid methyl ester and 79B. MS m/z 519 (M+1).
Step 2. Preparation of {8-[3-(4-Trifluoromethyl-benzyloxy)-benzylsulfanyl]-chroman-5-yloxy}-acetic acid (compound 84)
The title compound was prepared in the manner analogous to Example 1 using 84A. mp 113-115° C.; HPLC: area %=97.30, r.t.=3.140 min, γ=214 nm, mobile phase=acetonitrile/water w/0.10% TFA; IR (KBr) cm−: 2952, 2577, 1742, 1582, 1323, 1120; 400 MHz 1H NMR (DMSO-d6): δ 7.67-7.74 (m, 2H), 7.56-7.63 (m, 2H), 7.13 (t, 1H, J=7.8 Hz), 6.76-6.93 (m, 4H), 6.22 (d, 1H, J=8.7 Hz), 5.08 (s, 2H), 4.45 (s, 2H), 4.08 (t, 2H, J=4.8 Hz), 3.91 (s, 2H), 2.54 (t, 2H, J=6.6 Hz), 1.82 (pentet, 2H); MS m/z 505 (M+1). Anal. Calc'd for C26H23F3O5S: C, 61.90; H, 4.60; found: C, 61.49; H, 4.44.
Step 1. Preparation of {8-[3-(4-Trifluoromethyl-benzyloxy)-benzylsulfanyl]-chroman-5-yloxy}-acetic acid methyl ester (compound 84A)
The title compound was prepared in the manner analogous to Example 1F using (8-mercapto-chroman-5-yloxy)-acetic acid methyl ester and 79B. MS m/z 519 (M+1).
Step 2. Preparation of {8-[3-(4-Trifluoromethyl-benzyloxy)-benzylsulfanyl]-chroman-5-yloxy}-acetic acid (compound 84)
The title compound was prepared in the manner analogous to Example 1 using 84A. mp 113-115° C.; HPLC: area %=97.30, r.t.=3.140 min, γ=214 nm, mobile phase=acetonitrile/water w/0.10% TFA; IR (KBr) cm−: 2952, 2577, 1742, 1582, 1323, 1120; 400 MHz 1H NMR (DMSO-d6): δ 7.67-7.74 (m, 2H), 7.56-7.63 (m, 2H), 7.13 (t, 1H, J=7.8 Hz), 6.76-6.93 (m, 4H), 6.22 (d, 1H, J=8.7 Hz), 5.08 (s, 2H), 4.45 (s, 2H), 4.08 (t, 2H, J=4.8 Hz), 3.91 (s, 2H), 2.54 (t, 2H, J=6.6 Hz), 1.82 (pentet, 2H); MS m/z 505 (M+1). Anal. Calc'd for C26H23F3O5S: C, 61.90; H, 4.60; found: C, 61.49; H, 4.44.
Synthesis of {8-[4-(2,5-Dichloro-benzyloxy)-benzylsulfanyl]-chroman-5-yloxy}-acetic acid (compound 85)
Step 1. Preparation of {8-[4-(2,5-Dichloro-benzyloxy)-benzylsulfanyl]-chroman-5-yloxy}-acetic acid methyl ester (compound 85A)
The title compound was prepared in the manner analogous to Example 1F using (8-mercapto-chroman-5-yloxy)-acetic acid methyl ester and 1,4-Dichloro-2-(4-chloromethyl-phenoxymethyl)-benzene prepared in the manner analogous to Example 71A. MS m/z 519 (M+1).
Step 2. Preparation of {8-[4-(2,5-Dichloro-benzyloxy)-benzylsulfanyl]-chroman-5-yloxy}-acetic acid (compound 85)
The title compound was prepared in the manner analogous to Example 1 using 85A. mp 98-100° C.; IR (KBr) cm−1: 3038, 2854, 1729, 1508, 1240, 1124; 400 MHz 1H NMR (MSOd-6): δ 12.95 (br(s), 1H), 7.57-7.62 (m, 1H), 7.39-7.53 (m, 2H), 7.12-7.20 (m, 2H), 6.84-6.96 (m, 3H), 6.26 (d, 1H, J=8.6 Hz), 5.05 (s, 2H), 4.58 (s, 2H), 4.09 (t, 2H, J=4.9 Hz), 3.92 (s, 2H), 2.55 (t, 2H, J=6.5 Hz), 1.83 (pentet, 2H); MS m/z 503 (M−1). Anal. Calc'd for C25H22Cl2O5S: C, 59.41; H, 4.39; found: C, 59.20; H, 4.20.
Step 1. Preparation of {8-[4-(2,5-Dichloro-benzyloxy)-benzylsulfanyl]-chroman-5-yloxy}-acetic acid methyl ester (compound 85A)
The title compound was prepared in the manner analogous to Example 1F using (8-mercapto-chroman-5-yloxy)-acetic acid methyl ester and 1,4-Dichloro-2-(4-chloromethyl-phenoxymethyl)-benzene prepared in the manner analogous to Example 71A. MS m/z 519 (M+1).
Step 2. Preparation of {8-[4-(2,5-Dichloro-benzyloxy)-benzylsulfanyl]-chroman-5-yloxy}-acetic acid (compound 85)
The title compound was prepared in the manner analogous to Example 1 using 85A. mp 98-100° C.; IR (KBr) cm−1: 3038, 2854, 1729, 1508, 1240, 1124; 400 MHz 1H NMR (MSOd-6): δ 12.95 (br(s), 1H), 7.57-7.62 (m, 1H), 7.39-7.53 (m, 2H), 7.12-7.20 (m, 2H), 6.84-6.96 (m, 3H), 6.26 (d, 1H, J=8.6 Hz), 5.05 (s, 2H), 4.58 (s, 2H), 4.09 (t, 2H, J=4.9 Hz), 3.92 (s, 2H), 2.55 (t, 2H, J=6.5 Hz), 1.83 (pentet, 2H); MS m/z 503 (M−1). Anal. Calc'd for C25H22Cl2O5S: C, 59.41; H, 4.39; found: C, 59.20; H, 4.20.
Synthesis of {8-[4-(5-Trifluoromethyl-pyridine-2-yl)-benzylsulfanyl]-chroman-5-yloxy}-acetic acid (compound 86)
Step 1. {8-[4-(5-Trifluoromethyl-pyridine-2-yl)-benzylsulfanyl]-chroman-5-yloxy}-acetic acid methyl ester (compound 86A)
The title compound was prepared in the manner analogous to Example 1F using (8-mercapto-chroman-5-yloxy)-acetic acid methyl ester and 18B. MS m/z 490 (M+1).
Step 2. {8-[4-(5-Trifluoromethyl-pyridine-2-yl)-benzylsulfanyl]-chroman-5-yloxy}-acetic acid (compound 86)
The title compound was prepared in the manner analogous to Example 1 using 86A. mp 178-179° C.; IR (KBr) cm−1: 3034, 2577, 1707, 1604, 1332, 1111; 400 MHz 1H NMR (DMSO-d6): δ 12.95 (br(s), 1H), 8.97 (s, 1H), 7.99-8.24 (m, 4H), 7.33-7.40 (m, 2H), 6.95 (d, 2H, J=8.5 Hz), 6.26 (d, 2H, J=8.6 Hz), 4.57 (s, 2H), 4.11 (t, 2H, J=4.9 Hz), 4.04 (s, 2H), 2.56 (t, 2H, J=6.5 Hz), 1.84 (pentet, 2H); MS m/z 476 (M+1). Anal. Calc'd for C24H20F3NO4S: C, 60.63; H, 4.24; N, 2.95; found: C, 60.31; H, 4.24; N, 3.02.
Step 1. {8-[4-(5-Trifluoromethyl-pyridine-2-yl)-benzylsulfanyl]-chroman-5-yloxy}-acetic acid methyl ester (compound 86A)
The title compound was prepared in the manner analogous to Example 1F using (8-mercapto-chroman-5-yloxy)-acetic acid methyl ester and 18B. MS m/z 490 (M+1).
Step 2. {8-[4-(5-Trifluoromethyl-pyridine-2-yl)-benzylsulfanyl]-chroman-5-yloxy}-acetic acid (compound 86)
The title compound was prepared in the manner analogous to Example 1 using 86A. mp 178-179° C.; IR (KBr) cm−1: 3034, 2577, 1707, 1604, 1332, 1111; 400 MHz 1H NMR (DMSO-d6): δ 12.95 (br(s), 1H), 8.97 (s, 1H), 7.99-8.24 (m, 4H), 7.33-7.40 (m, 2H), 6.95 (d, 2H, J=8.5 Hz), 6.26 (d, 2H, J=8.6 Hz), 4.57 (s, 2H), 4.11 (t, 2H, J=4.9 Hz), 4.04 (s, 2H), 2.56 (t, 2H, J=6.5 Hz), 1.84 (pentet, 2H); MS m/z 476 (M+1). Anal. Calc'd for C24H20F3NO4S: C, 60.63; H, 4.24; N, 2.95; found: C, 60.31; H, 4.24; N, 3.02.
Synthesis of {7-[5-(2-Chloro-phenyl)-isoxazol-3-ylmethylsulfanyl]-indan-4-yloxy}-acetic acid (compound 87)
Step 1. Preparation of {7-[5-(2-Chloro-phenyl)-isoxazol-3-ylmethylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 87A)
The title compound was prepared in the manner analogous to Example 1F using commercially available 3-chloromethyl-5-(2-chloro-phenyl)isoxazole and 12C. MS m/z 430 (M+1).
Step 2. Preparation of {7-[5-(2-Chloro-phenyl)-isoxazol-3-ylmethylsulfanyl]-indan-4-yloxy}-acetic acid (compound 87)
The title compound was prepared in the manner analogous to Example 1 using 87A. mp 161-162° C.; IR (KBr) cm−1: 3177, 3065, 1706, 1475, 1432, 1233; 400 MHz 1H NMR (DMSO-d6): δ 13.00 (bs, 1H), 7.78-7.85 (m, 1H), 7.58-7.64 (m, 1H), 7.42-7.53 (m, 2H), 7.15 (d, 1H, J=8.6 Hz), 6.84 (s, 1H), 6.60 (d, 1H, J=8.6 Hz), 4.62 (s, 2H), 4.09 (s, 2H), 2.72-2.82 (m, 4H), 1.91 (pentet, 2H); MS m/z 416 (M+1). Anal. Calc'd for C21H18ClNO4S: C, 60.65; H, 4.36; N, 3.37; found: C, 60.56; H, 4.28; N, 3.16.
Step 1. Preparation of {7-[5-(2-Chloro-phenyl)-isoxazol-3-ylmethylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 87A)
The title compound was prepared in the manner analogous to Example 1F using commercially available 3-chloromethyl-5-(2-chloro-phenyl)isoxazole and 12C. MS m/z 430 (M+1).
Step 2. Preparation of {7-[5-(2-Chloro-phenyl)-isoxazol-3-ylmethylsulfanyl]-indan-4-yloxy}-acetic acid (compound 87)
The title compound was prepared in the manner analogous to Example 1 using 87A. mp 161-162° C.; IR (KBr) cm−1: 3177, 3065, 1706, 1475, 1432, 1233; 400 MHz 1H NMR (DMSO-d6): δ 13.00 (bs, 1H), 7.78-7.85 (m, 1H), 7.58-7.64 (m, 1H), 7.42-7.53 (m, 2H), 7.15 (d, 1H, J=8.6 Hz), 6.84 (s, 1H), 6.60 (d, 1H, J=8.6 Hz), 4.62 (s, 2H), 4.09 (s, 2H), 2.72-2.82 (m, 4H), 1.91 (pentet, 2H); MS m/z 416 (M+1). Anal. Calc'd for C21H18ClNO4S: C, 60.65; H, 4.36; N, 3.37; found: C, 60.56; H, 4.28; N, 3.16.
Synthesis of {7-[3-(2,6-Dichloro-phenyl)-5-methyl-isoxazol-4-ylmethylsulfanyl]-indan-4-yloxy}-acetic acid (compound 88)
Step 1. Preparation of {7-[3-(2,6-Dichloro-phenyl)-5-methyl-isoxazol-4-ylmethylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 88A)
The title compound was prepared in the manner analogous to Example 1F using commercially available 4-bromomethyl-3-(2,6-dichloro-phenyl)-5-methyl-isoxazole and 12C. MS m/z 478 (M+1).
Step 2. Preparation of {7-[3-(2,6-Dichloro-phenyl)-5-methyl-isoxazol-4-ylmethylsulfanyl]-indan-4-yloxy}-acetic acid (compound 88)
The title compound was prepared in the manner analogous to Example 1 using 88A. mp 151-152° C.; IR (KBr) cm−1: 3084, 1743, 1430, 1277, 1245, 1106; 400 MHz 1H NMR (DMSO-d6): δ 12.97 (br(s), 1H), 7.47-7.70 (m, 3H), 6.90 (d, 1H, J=8.5 Hz), 6.52 (d, 1H, J=8.5 Hz), 4.63 (s, 2H), 3.58 (s, 2H), 2.75 (t, 2H, J=7.6 Hz), 2.65 (t, 2H, J=7.6 Hz), 2.07 (s, 3H), 1.89 (pentet, 2H) MS m/z 464 (M+1). Anal. Calc'd for C22H19Cl2NO4S: C, 56.90; H, 4.12; N, 3.02; found: C, 56.51; H, 3.96; N, 2.95.
Step 1. Preparation of {7-[3-(2,6-Dichloro-phenyl)-5-methyl-isoxazol-4-ylmethylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 88A)
The title compound was prepared in the manner analogous to Example 1F using commercially available 4-bromomethyl-3-(2,6-dichloro-phenyl)-5-methyl-isoxazole and 12C. MS m/z 478 (M+1).
Step 2. Preparation of {7-[3-(2,6-Dichloro-phenyl)-5-methyl-isoxazol-4-ylmethylsulfanyl]-indan-4-yloxy}-acetic acid (compound 88)
The title compound was prepared in the manner analogous to Example 1 using 88A. mp 151-152° C.; IR (KBr) cm−1: 3084, 1743, 1430, 1277, 1245, 1106; 400 MHz 1H NMR (DMSO-d6): δ 12.97 (br(s), 1H), 7.47-7.70 (m, 3H), 6.90 (d, 1H, J=8.5 Hz), 6.52 (d, 1H, J=8.5 Hz), 4.63 (s, 2H), 3.58 (s, 2H), 2.75 (t, 2H, J=7.6 Hz), 2.65 (t, 2H, J=7.6 Hz), 2.07 (s, 3H), 1.89 (pentet, 2H) MS m/z 464 (M+1). Anal. Calc'd for C22H19Cl2NO4S: C, 56.90; H, 4.12; N, 3.02; found: C, 56.51; H, 3.96; N, 2.95.
Synthesis of {7-[3-(4-Trifluoromethyl-phenyl)-isoxazol-5-ylmethylsulfanyl]-indan-4-yloxy}-acetic acid (compound 89)
Step 1. Preparation of {7-[3-(4-Trifluoromethyl-phenyl)-isoxazol-5-ylmethylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 89A)
The title compound was prepared in the manner analogous to Example 1F using 42C and 12C. MS m/z 464 (M+1).
Step 2. Preparation of {7-[3-(4-Trifluoromethyl-phenyl)-isoxazol-5-ylmethylsulfanyl]-indan-4-yloxy}-acetic acid (compound 89)
The title compound was prepared in the manner analogous to Example 1 using 89A. mp 166-168° C.; HPLC: area %=96.95; r.t.=3.140 min, γ=214 nm, mobile phase=acetonitrile/water w/0.10% TFA. IR (KBr) cm−1: 3140, 3085, 1742, 1322, 1255, 1109; 400 MHz 1H NMR (DMSO-d6): δ 12.96 (br(s), 1H), 7.99 (d, 2H, J=8.3 Hz), 7.81 (d, 2H, J=8.3 Hz), 7.14 (d, 1H, J=8.4 Hz), 6.81 (s, 1H), 6.61 (d, 1H, J=8.4 Hz), 4.63 (s, 2H), 4.23 (s, 2H), 2.76 (t, 4H, J=7.5 Hz), 1.91 (pentet, 2H); MS m/z 450 (M+1). Anal. Calc'd for C22H18F3NO4S: C, 58.79; H, 4.04; N, 3.12; found: C, 58.38; H, 3.92; N, 2.95.
Step 1. Preparation of {7-[3-(4-Trifluoromethyl-phenyl)-isoxazol-5-ylmethylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 89A)
The title compound was prepared in the manner analogous to Example 1F using 42C and 12C. MS m/z 464 (M+1).
Step 2. Preparation of {7-[3-(4-Trifluoromethyl-phenyl)-isoxazol-5-ylmethylsulfanyl]-indan-4-yloxy}-acetic acid (compound 89)
The title compound was prepared in the manner analogous to Example 1 using 89A. mp 166-168° C.; HPLC: area %=96.95; r.t.=3.140 min, γ=214 nm, mobile phase=acetonitrile/water w/0.10% TFA. IR (KBr) cm−1: 3140, 3085, 1742, 1322, 1255, 1109; 400 MHz 1H NMR (DMSO-d6): δ 12.96 (br(s), 1H), 7.99 (d, 2H, J=8.3 Hz), 7.81 (d, 2H, J=8.3 Hz), 7.14 (d, 1H, J=8.4 Hz), 6.81 (s, 1H), 6.61 (d, 1H, J=8.4 Hz), 4.63 (s, 2H), 4.23 (s, 2H), 2.76 (t, 4H, J=7.5 Hz), 1.91 (pentet, 2H); MS m/z 450 (M+1). Anal. Calc'd for C22H18F3NO4S: C, 58.79; H, 4.04; N, 3.12; found: C, 58.38; H, 3.92; N, 2.95.
Synthesis of {7-[2-(4′-Trifluoromethyl-biphenyl-4yl)-ethylsulfanyl]-indan-4-yloxy}-acetic acid (compound 90)
Step 1. Preparation of {7-[2-(4′-Trifluoromethyl-biphenyl-4-yl)-ethylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 90A)
The title compound was prepared in the manner analogous to Example 1F using 12C and 4-(2-bromo-ethyl)-4′-trifluoromethyl-biphenyl (compound 22B). MS m/z 487 (M+1).
Step 2. Preparation of {7-[2-(4′-Trifluoromethyl-biphenyl-4-yl)-ethylsulfanyl]-indan-4-yloxy}-acetic acid (compound 90)
The title compound was prepared in the manner analogous to Example 1 using 90A. mp 170-172° C.; IR (thin film) cm−1: 1724, 1471, 1327, 1239, 1175, 1110; 400 MHz 1H NMR (DMSO-d6) δ 12.96 (br s, 1H), 7.82 (d, 2H, J=8.3 Hz), 7.74 (d, 2H, J=8.3 Hz), 7.61 (d, 2H, J=8.3 Hz), 7.31 (d, 2H, J=8.3 Hz), 7.12 (d, 2H, J=8.5 Hz), 6.62 (d, 1H, J=8.5 Hz), 4.63 (s, 2H), 3.08 (t, 2H, J=7.6 Hz), 2.80 (m, 6H), 1.96 (m, 2H); MS m/z 471 (M−1). Anal. Calc'd for C26H23F3O3S: C, 66.09; H, 4.91; found: C, 65.95; H, 4.63.
Step 1. Preparation of {7-[2-(4′-Trifluoromethyl-biphenyl-4-yl)-ethylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 90A)
The title compound was prepared in the manner analogous to Example 1F using 12C and 4-(2-bromo-ethyl)-4′-trifluoromethyl-biphenyl (compound 22B). MS m/z 487 (M+1).
Step 2. Preparation of {7-[2-(4′-Trifluoromethyl-biphenyl-4-yl)-ethylsulfanyl]-indan-4-yloxy}-acetic acid (compound 90)
The title compound was prepared in the manner analogous to Example 1 using 90A. mp 170-172° C.; IR (thin film) cm−1: 1724, 1471, 1327, 1239, 1175, 1110; 400 MHz 1H NMR (DMSO-d6) δ 12.96 (br s, 1H), 7.82 (d, 2H, J=8.3 Hz), 7.74 (d, 2H, J=8.3 Hz), 7.61 (d, 2H, J=8.3 Hz), 7.31 (d, 2H, J=8.3 Hz), 7.12 (d, 2H, J=8.5 Hz), 6.62 (d, 1H, J=8.5 Hz), 4.63 (s, 2H), 3.08 (t, 2H, J=7.6 Hz), 2.80 (m, 6H), 1.96 (m, 2H); MS m/z 471 (M−1). Anal. Calc'd for C26H23F3O3S: C, 66.09; H, 4.91; found: C, 65.95; H, 4.63.
Synthesis of {7-[2-(4′-Trifluoromethyl-biphenyl-4-yl)-ethyl]-indan-4-yloxy}-acetic acid (compound 91)
Step 1. Preparation of (Indan-4-yloxy)-acetic acid methyl ester (compound 91A)
Compound 91A was prepared from indan-4-ol and bromo-acetic acid methyl ester in the manner analogous to Example 1C. 400 MHz 1H NMR (DMSO-d6) δ 7.00 (t, 1H, J=7.8 Hz), 6.79 (d, 1H, J=7.3 Hz), 6.56 (d, 1H, J=8.1 Hz), 4.74 (s, 2H), 3.63 (s, 3H), 2.77 (m, 4H), 1.95 (m, 2H).
Step 2. Preparation of (4′-Trifluoromethyl-biphenyl-4-yl)-acetic acid (compound 91B)
A mixture of (4-bromo-phenyl)-acetic acid (10.2 g, 47.4 mmol), 4-trifluoromethylphenylboronic acid (10.0 g, 52.7 mmol), and 50% water-wet 5% palladium on charcoal catalyst (4.6 g) in 50 ml of water and 8.0 ml of 2-propanol was treated dropwise over 30 minutes with a solution of sodium carbonate (6.8 g, 64.2 mmol) in 18 ml of water. The mixture was heated at 65-70° C. for 3 h, then cooled to 40° C. and treated with 13.0 ml of a solution of 2-propanol/water/2.0 N aqueous sodium hydroxide solution (70/15/1). The reaction mixture was filtered through a bed of Celite filter-aid, and the filter cake was washed 5× with the above 2-propanol/water/2.0 N aqueous sodium hydroxide solution. The combined filtrates were diluted with 125 ml of water, and the solution was digested on the steam bath with charcoal and filtered. The filtrate was diluted with an additional 150 ml of water and made strongly acidic by the addition of 4.0 N hydrochloric acid. The precipitated product was filtered and suspended in 350 ml of water plus 50 ml of methanol. The new mixture was stirred for several hours and filtered again. The crude product was recrystallized from aqueous acetonitrile. A sample recrystallized a second time from aqueous acetonitrile had mp 158-160° C.; MS m/z 280 (M).
Step 3. Preparation of (4′-Trifluoromethyl-biphenyl-4-yl)-acetyl chloride (compound 91C)
A suspension 91B (2.0 g, 7.1 mmol) and 5 drops of N,N-dimethylformamide in 30 ml of dichloromethane was cooled in ice and treated dropwise with a solution of oxalyl chloride (0.70 ml, 1.0 g, 8.0 mmol) in 10 ml of dichloromethane. The ice bath was removed, and the mixture was stirred at room temperature for 3 h. The solution was filtered, and the filtrate was evaporated. The residue quickly crystallized to yield the acid chloride intermediate, which was used immediately in the next step.
Step 4. Preparation of {7-[2-(4′-Trifluoromethyl-biphenyl-4-yl-acetyl]-indan-4-yloxy}-acetic acid methyl ester (compound 91D)
A solution of 91C (2.1 g, 7.0 mmol) in 25 ml of 1,2-dichloro-ethane was cooled in ice and treated with anhydrous ferric chloride (1.2 g, 7.4 mmol). The mixture was stirred, and a solution of 91A (1.5 g, 7.3 mmol) in 10 ml of 1,2-dichloro-ethane was added dropwise. The mixture was stirred at room temperature for 18 h. The reaction mixture was added to 300 g of ice and of brine and extracted with ethyl acetate (4×100 ml). The combined extracts were washed with 5% aqueous sodium bicarbonate solution (4×250 ml) and brine (1×250 ml), then dried over anhydrous sodium sulfate and concentrated. The crude product was purified by normal phase chromatography. A sample recrystallized from ethyl acetate/hexane had mp 141-143° C.; MS m/z 467 (M−1).
Step 5. Preparation of {7-[2-(4′-Trifluoromethyl-biphenyl-4-yl)-ethyl]-indan-4-yloxy}-acetic acid methyl ester (compound 91E)
A solution of 91D (1.0 g, 2.1 mmol) in 10.0 ml of trifluoroacetic acid was treated dropwise with triethylsilane (1.5 ml, 1.1 g, 9.4 mmol). The mixture was stirred at room temperature for 3 h and then added to 200 g of ice and water. The precipitated solid was extracted out with ethyl acetate (4×100 ml). The combined extracts were washed with brine (1×250 ml), 5% aqueous sodium bicarbonate solution (4×250 ml), and brine again, then dried over anhydrous sodium sulfate and concentrated. The crude product was purified by normal phase chromatography. 400 MHz 1H NMR (DMSO-d6) δ 7.82 (d, 2H, J=8.3 Hz), 7.75 (d, 2H, J=8.3 Hz), 7.61 (d, 2H, J=8.3 Hz), 7.30 (d, 2H, J=8.3 Hz), 6.90 (d, 1H, J=8.3 Hz), 6.53 (d, 1H, J=8.3 Hz), 4.71 (s, 2H), 3.63 (s, 2H), 2.77 (m, 8H), 1.94 (m, 2H).
Step 6. Preparation of {7-[2-(4′-Trifluoromethyl-biphenyl-4-yl)-ethyl]-indan-4-yloxy}-acetic acid (compound 91)
The title compound was prepared in the manner analogous to Example 1 using 91E. mp 188-190° C.; IR (thin film) cm−1: 1745, 1322, 1252, 1170, 1112, 1071; 400 MHz 1H NMR (DMSO-d6) δ 12.89 (br s, 1H), 7.83 (d, 2H, J=8.1 Hz), 7.74 (d, 2H, J=8.5 Hz), 7.61 (d, 2H, J=8.1 Hz), 7.30 (d, 2H, J=8.3 Hz), 6.90 (d, 1H, J=8.3 Hz), 6.51 (d, 1H, J=8.3 Hz), 4.58 (s, 2H), 2.75(m, 8H), 2.45 (m, 2H); MS m/z 439 (M−1). Anal. Calc'd for C26H23F3O3: C, 70.90; H, 5.26; found: C, 70.92; H, 5.01.
Step 1. Preparation of (Indan-4-yloxy)-acetic acid methyl ester (compound 91A)
Compound 91A was prepared from indan-4-ol and bromo-acetic acid methyl ester in the manner analogous to Example 1C. 400 MHz 1H NMR (DMSO-d6) δ 7.00 (t, 1H, J=7.8 Hz), 6.79 (d, 1H, J=7.3 Hz), 6.56 (d, 1H, J=8.1 Hz), 4.74 (s, 2H), 3.63 (s, 3H), 2.77 (m, 4H), 1.95 (m, 2H).
Step 2. Preparation of (4′-Trifluoromethyl-biphenyl-4-yl)-acetic acid (compound 91B)
A mixture of (4-bromo-phenyl)-acetic acid (10.2 g, 47.4 mmol), 4-trifluoromethylphenylboronic acid (10.0 g, 52.7 mmol), and 50% water-wet 5% palladium on charcoal catalyst (4.6 g) in 50 ml of water and 8.0 ml of 2-propanol was treated dropwise over 30 minutes with a solution of sodium carbonate (6.8 g, 64.2 mmol) in 18 ml of water. The mixture was heated at 65-70° C. for 3 h, then cooled to 40° C. and treated with 13.0 ml of a solution of 2-propanol/water/2.0 N aqueous sodium hydroxide solution (70/15/1). The reaction mixture was filtered through a bed of Celite filter-aid, and the filter cake was washed 5× with the above 2-propanol/water/2.0 N aqueous sodium hydroxide solution. The combined filtrates were diluted with 125 ml of water, and the solution was digested on the steam bath with charcoal and filtered. The filtrate was diluted with an additional 150 ml of water and made strongly acidic by the addition of 4.0 N hydrochloric acid. The precipitated product was filtered and suspended in 350 ml of water plus 50 ml of methanol. The new mixture was stirred for several hours and filtered again. The crude product was recrystallized from aqueous acetonitrile. A sample recrystallized a second time from aqueous acetonitrile had mp 158-160° C.; MS m/z 280 (M).
Step 3. Preparation of (4′-Trifluoromethyl-biphenyl-4-yl)-acetyl chloride (compound 91C)
A suspension 91B (2.0 g, 7.1 mmol) and 5 drops of N,N-dimethylformamide in 30 ml of dichloromethane was cooled in ice and treated dropwise with a solution of oxalyl chloride (0.70 ml, 1.0 g, 8.0 mmol) in 10 ml of dichloromethane. The ice bath was removed, and the mixture was stirred at room temperature for 3 h. The solution was filtered, and the filtrate was evaporated. The residue quickly crystallized to yield the acid chloride intermediate, which was used immediately in the next step.
Step 4. Preparation of {7-[2-(4′-Trifluoromethyl-biphenyl-4-yl-acetyl]-indan-4-yloxy}-acetic acid methyl ester (compound 91D)
A solution of 91C (2.1 g, 7.0 mmol) in 25 ml of 1,2-dichloro-ethane was cooled in ice and treated with anhydrous ferric chloride (1.2 g, 7.4 mmol). The mixture was stirred, and a solution of 91A (1.5 g, 7.3 mmol) in 10 ml of 1,2-dichloro-ethane was added dropwise. The mixture was stirred at room temperature for 18 h. The reaction mixture was added to 300 g of ice and of brine and extracted with ethyl acetate (4×100 ml). The combined extracts were washed with 5% aqueous sodium bicarbonate solution (4×250 ml) and brine (1×250 ml), then dried over anhydrous sodium sulfate and concentrated. The crude product was purified by normal phase chromatography. A sample recrystallized from ethyl acetate/hexane had mp 141-143° C.; MS m/z 467 (M−1).
Step 5. Preparation of {7-[2-(4′-Trifluoromethyl-biphenyl-4-yl)-ethyl]-indan-4-yloxy}-acetic acid methyl ester (compound 91E)
A solution of 91D (1.0 g, 2.1 mmol) in 10.0 ml of trifluoroacetic acid was treated dropwise with triethylsilane (1.5 ml, 1.1 g, 9.4 mmol). The mixture was stirred at room temperature for 3 h and then added to 200 g of ice and water. The precipitated solid was extracted out with ethyl acetate (4×100 ml). The combined extracts were washed with brine (1×250 ml), 5% aqueous sodium bicarbonate solution (4×250 ml), and brine again, then dried over anhydrous sodium sulfate and concentrated. The crude product was purified by normal phase chromatography. 400 MHz 1H NMR (DMSO-d6) δ 7.82 (d, 2H, J=8.3 Hz), 7.75 (d, 2H, J=8.3 Hz), 7.61 (d, 2H, J=8.3 Hz), 7.30 (d, 2H, J=8.3 Hz), 6.90 (d, 1H, J=8.3 Hz), 6.53 (d, 1H, J=8.3 Hz), 4.71 (s, 2H), 3.63 (s, 2H), 2.77 (m, 8H), 1.94 (m, 2H).
Step 6. Preparation of {7-[2-(4′-Trifluoromethyl-biphenyl-4-yl)-ethyl]-indan-4-yloxy}-acetic acid (compound 91)
The title compound was prepared in the manner analogous to Example 1 using 91E. mp 188-190° C.; IR (thin film) cm−1: 1745, 1322, 1252, 1170, 1112, 1071; 400 MHz 1H NMR (DMSO-d6) δ 12.89 (br s, 1H), 7.83 (d, 2H, J=8.1 Hz), 7.74 (d, 2H, J=8.5 Hz), 7.61 (d, 2H, J=8.1 Hz), 7.30 (d, 2H, J=8.3 Hz), 6.90 (d, 1H, J=8.3 Hz), 6.51 (d, 1H, J=8.3 Hz), 4.58 (s, 2H), 2.75(m, 8H), 2.45 (m, 2H); MS m/z 439 (M−1). Anal. Calc'd for C26H23F3O3: C, 70.90; H, 5.26; found: C, 70.92; H, 5.01.
Synthesis of {5-Methyl-7-[4-(5-trifluoromethyl-pyridin-2-yl)-benzylsulfanyl]-2,3-dihydro-benzofuran-4-yloxy}-acetic acid (compound 92)
Step 1. Preparation of {5-Methyl-7-[4-(5-trifluoromethyl-pyridin-2-yl)-benzylsulfanyl]-2,3-dihydro-benzofuran-4-yloxy}-acetic acid methyl ester (compound 92A)
The title compound was prepared in the manner analogous to Example 1F using 18B and 7-mercapto-5-methyl-2,3-dihydro-benzofuran-4-yloxy)-acetic acid methyl ester (prepared in a similar manner as described for Example 12C). mp 94-95° C.; MS m/z 490 (M+1).
Step 2. Preparation of (5-Methyl-7-[4(5-trifluoromethyl-pyridin-2-yl)-benzylsulfanyl]-2,3-dihydro-benzofuran-4-yloxy)-acetic acid (compound 92)
The title compound was prepared in the manner analogous to Example 1 using 92A. mp 155-157° C.; IR (thin film) cm−1: 1732, 1587, 1416, 1331, 1211, 1129; 400 MHz 1H NMR (DMSO-d6) δ 12.89 (br s, 1H), 8.97 (m, 1H), 8.21 (dd, 1H, J=2.0, 8.5 Hz), 8.12 (d, 1H, J=8.3 Hz), 8.02 (d, 2H, J=8.3 Hz), 7.34 (d, 2H, J=8.5 Hz), 6.83 (s, 1H), 4.56 (s, 2H), 4.48 (t, 2H, J=8.7 Hz), 4.08 (s, 2H), 3.24 (t, 2H, J=8.7 Hz), 2.02 (s, 3H); MS m/z 474 (M−1). Anal. Calc'd for C24H20F3NO4S: C, 60.63; H, 4.24; N, 2.95; found: C, 60.54; H, 4.19; N, 2.94.
Step 1. Preparation of {5-Methyl-7-[4-(5-trifluoromethyl-pyridin-2-yl)-benzylsulfanyl]-2,3-dihydro-benzofuran-4-yloxy}-acetic acid methyl ester (compound 92A)
The title compound was prepared in the manner analogous to Example 1F using 18B and 7-mercapto-5-methyl-2,3-dihydro-benzofuran-4-yloxy)-acetic acid methyl ester (prepared in a similar manner as described for Example 12C). mp 94-95° C.; MS m/z 490 (M+1).
Step 2. Preparation of (5-Methyl-7-[4(5-trifluoromethyl-pyridin-2-yl)-benzylsulfanyl]-2,3-dihydro-benzofuran-4-yloxy)-acetic acid (compound 92)
The title compound was prepared in the manner analogous to Example 1 using 92A. mp 155-157° C.; IR (thin film) cm−1: 1732, 1587, 1416, 1331, 1211, 1129; 400 MHz 1H NMR (DMSO-d6) δ 12.89 (br s, 1H), 8.97 (m, 1H), 8.21 (dd, 1H, J=2.0, 8.5 Hz), 8.12 (d, 1H, J=8.3 Hz), 8.02 (d, 2H, J=8.3 Hz), 7.34 (d, 2H, J=8.5 Hz), 6.83 (s, 1H), 4.56 (s, 2H), 4.48 (t, 2H, J=8.7 Hz), 4.08 (s, 2H), 3.24 (t, 2H, J=8.7 Hz), 2.02 (s, 3H); MS m/z 474 (M−1). Anal. Calc'd for C24H20F3NO4S: C, 60.63; H, 4.24; N, 2.95; found: C, 60.54; H, 4.19; N, 2.94.
Synthesis of [8-(4′-Trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-chroman-5-yloxy]-acetic acid (compound 93)
Step 1. Preparation of [8-(4′-Trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-chroman-5-yloxy]-acetic acid methyl ester (compound 93A)
The title compound was prepared in the manner analogous to Example 1F using 18B and (8-mercapto-chroman-5-yloxy)-acetic acid methyl ester mp 122-124° C.; MS m/z 489 (M+1).
Step 2. Preparation of [8-(4′-Trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-chroman-5-yloxy]-acetic acid (compound 93)
The title compound was prepared in the manner analogous to Example 1 using 93A. mp 170-172° C.; IR (thin film) cm−1: 1717, 1584, 1473, 1330, 1231, 1118; 400 MHz 1H NMR (DMSO-d6) δ 12.94 (br s, 1H), 7.82 (d, 2H, J=8.1 Hz), 7.74 (d, 2H, J=8.5 Hz), 7.60 (d, 2H, J=8.3 Hz), 7.34 (d, 2H, J=8.3 Hz), 6.96 (d, 2H, J=8.8 Hz), 6.28 (d, 2H, J=8.8 Hz), 4.58 (s, 2H), 4.11 (t, 2H, J=5.0 Hz), 4.03 (s, 2H), 2.56 (t, 2H, J=6.5 Hz), 1.84 (m, 2H); MS m/z 473 (M−1). Anal. Calc'd for C25H21F3O4S: C, 63.28; H, 4.46; found: C, 63.28; H, 4.26.
Step 1. Preparation of [8-(4′-Trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-chroman-5-yloxy]-acetic acid methyl ester (compound 93A)
The title compound was prepared in the manner analogous to Example 1F using 18B and (8-mercapto-chroman-5-yloxy)-acetic acid methyl ester mp 122-124° C.; MS m/z 489 (M+1).
Step 2. Preparation of [8-(4′-Trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-chroman-5-yloxy]-acetic acid (compound 93)
The title compound was prepared in the manner analogous to Example 1 using 93A. mp 170-172° C.; IR (thin film) cm−1: 1717, 1584, 1473, 1330, 1231, 1118; 400 MHz 1H NMR (DMSO-d6) δ 12.94 (br s, 1H), 7.82 (d, 2H, J=8.1 Hz), 7.74 (d, 2H, J=8.5 Hz), 7.60 (d, 2H, J=8.3 Hz), 7.34 (d, 2H, J=8.3 Hz), 6.96 (d, 2H, J=8.8 Hz), 6.28 (d, 2H, J=8.8 Hz), 4.58 (s, 2H), 4.11 (t, 2H, J=5.0 Hz), 4.03 (s, 2H), 2.56 (t, 2H, J=6.5 Hz), 1.84 (m, 2H); MS m/z 473 (M−1). Anal. Calc'd for C25H21F3O4S: C, 63.28; H, 4.46; found: C, 63.28; H, 4.26.
Synthesis of {8-[5-(4-Trifluoromethyl-phenyl)-isoxazol-3-ylmethylsulfanyl]-chroman-5-yloxy}-acetic acid (compound 94)
Step 1. Preparation of {8-[5-(4-Trifluoromethyl-phenyl)-isoxazol-3-ylmethylsulfanyl]-chroman-5-yloxy}-acetic acid methyl ester (compound 94A)
The title compound was prepared in the manner analogous to Example 1F using 42C and (8-mercapto-chroman-5-yloxy)-acetic acid methyl ester. mp 112-113° C.; MS m/z 480 (M+1).
Step 2. Preparation of {8-[5-(4-Trifluoromethyl-phenyl)-isoxazol-3-ylmethylsulfanyl]-chroman-5-yloxy}-acetic acid (compound 94)
The title compound was prepared in the manner analogous to Example 1 using 94A. The crude produce was recrystallized from ethyl acetate/hexane to yield the final product. mp 171-173° C.; IR (thin film) cm−1: 1722, 1432, 1322, 1232, 1103, 1065; 400 MHz 1H NMR (DMSO-d6) δ 12.94 (br s, 1H), 8.01 (d, 2H, J=8.1 Hz), 7.84 (d, 2H, J=8.3 Hz), 7.09 (s, 1H), 7.03 (d, 1H, J=8.5 Hz), 6.29 (d, 1H, J=8.5 Hz), 4.59 (s, 2H), 4.08 (t, 2H, J=5.0 Hz), 4.04 (s, 2H), 2.55 (t, 2H, J=6.5 Hz), 1.83 (m, 2H); MS m/z 464 (M−1). Anal. Calc'd for C22H18F3NO5S; C, 56.77; H, 3.90; N, 3.01; found: C, 56.80; H, 3.58; N, 3.07.
Step 1. Preparation of {8-[5-(4-Trifluoromethyl-phenyl)-isoxazol-3-ylmethylsulfanyl]-chroman-5-yloxy}-acetic acid methyl ester (compound 94A)
The title compound was prepared in the manner analogous to Example 1F using 42C and (8-mercapto-chroman-5-yloxy)-acetic acid methyl ester. mp 112-113° C.; MS m/z 480 (M+1).
Step 2. Preparation of {8-[5-(4-Trifluoromethyl-phenyl)-isoxazol-3-ylmethylsulfanyl]-chroman-5-yloxy}-acetic acid (compound 94)
The title compound was prepared in the manner analogous to Example 1 using 94A. The crude produce was recrystallized from ethyl acetate/hexane to yield the final product. mp 171-173° C.; IR (thin film) cm−1: 1722, 1432, 1322, 1232, 1103, 1065; 400 MHz 1H NMR (DMSO-d6) δ 12.94 (br s, 1H), 8.01 (d, 2H, J=8.1 Hz), 7.84 (d, 2H, J=8.3 Hz), 7.09 (s, 1H), 7.03 (d, 1H, J=8.5 Hz), 6.29 (d, 1H, J=8.5 Hz), 4.59 (s, 2H), 4.08 (t, 2H, J=5.0 Hz), 4.04 (s, 2H), 2.55 (t, 2H, J=6.5 Hz), 1.83 (m, 2H); MS m/z 464 (M−1). Anal. Calc'd for C22H18F3NO5S; C, 56.77; H, 3.90; N, 3.01; found: C, 56.80; H, 3.58; N, 3.07.
Synthesis of {8-[5-(4-Chloro-phenyl)-isoxazol-3-ylmethylsulfanyl]-chroman-5-yloxy}-acetic acid (compound 95)
Step 1. Preparation of {8-[5-(4-Chloro-phenyl)-isoxazol-3-ylmethylsulfanyl]-chroman-5-yloxy}-acetic acid methyl ester (compound 95A)
The title compound was prepared in the manner analogous to Example 1F using 3-chloromethyl-5-(4-chloro-phenyl)-isoxazole and (8-mercapto-chroman-5-yloxy)-acetic acid methyl ester. mp 131-133° C.; MS m/z 446 (M+1).
Step 2. Preparation of {8-[5-(4Chloro-phenyl)-isoxazol-3-ylmethylsulfanyl]-chroman-5-yloxy}-acetic acid (compound 95)
The title compound was prepared in the manner analogous to Example 1 using 95A. mp 181-183° C.; IR (thin film) cm−1: 1723, 1612, 1479, 1428, 1231, 1134; 400 MHz 1H NMR (DMSO-d6) δ 12.95 (br s, 1H), 7.82 (m, 2H), 7.54 (m, 2H), 7.02 (d, 1H, J=8.8 Hz), 6.94 (s, 1H), 6.29 (d, 1H, J=8.5 Hz), 4.59 (s, 2H), 4.08 (t, 2H, J=5.0 Hz), 4.01 (s, 2H), 2.55 (t, 2H, J=6.6 Hz), 1.83 (m, 2H); MS m/z 430 (M−1). Anal. Calc'd for C21H18ClNO5S: C, 58.40; H, 4.20; N, 3.24; found: C, 58.30; H, 3.91; N, 3.28.
Step 1. Preparation of {8-[5-(4-Chloro-phenyl)-isoxazol-3-ylmethylsulfanyl]-chroman-5-yloxy}-acetic acid methyl ester (compound 95A)
The title compound was prepared in the manner analogous to Example 1F using 3-chloromethyl-5-(4-chloro-phenyl)-isoxazole and (8-mercapto-chroman-5-yloxy)-acetic acid methyl ester. mp 131-133° C.; MS m/z 446 (M+1).
Step 2. Preparation of {8-[5-(4Chloro-phenyl)-isoxazol-3-ylmethylsulfanyl]-chroman-5-yloxy}-acetic acid (compound 95)
The title compound was prepared in the manner analogous to Example 1 using 95A. mp 181-183° C.; IR (thin film) cm−1: 1723, 1612, 1479, 1428, 1231, 1134; 400 MHz 1H NMR (DMSO-d6) δ 12.95 (br s, 1H), 7.82 (m, 2H), 7.54 (m, 2H), 7.02 (d, 1H, J=8.8 Hz), 6.94 (s, 1H), 6.29 (d, 1H, J=8.5 Hz), 4.59 (s, 2H), 4.08 (t, 2H, J=5.0 Hz), 4.01 (s, 2H), 2.55 (t, 2H, J=6.6 Hz), 1.83 (m, 2H); MS m/z 430 (M−1). Anal. Calc'd for C21H18ClNO5S: C, 58.40; H, 4.20; N, 3.24; found: C, 58.30; H, 3.91; N, 3.28.
Synthesis of {4-[5-(4-Chloro-phenyl)-isoxazol-3-ylmethylsulfanyl]-2-methyl-phenoxy}-acetic acid (compound 96)
Step 1. Preparation of {4-[5-(4-Chloro-phenyl)-isoxazol-3-ylmethylsulfanyl]-2-methyl-phenoxy}-acetic acid methyl ester (compound 96A)
The title compound was prepared in the manner analogous to Example 1F using 3-chloromethyl-5-(4-chloro-phenyl)-isoxazole and 2C. mp 79-80° C.; MS m/z 404 (M+1).
Step 2. Preparation of {4-[5-(4-Chloro-phenyl)-isoxazol-3-ylmethylsulfanyl]-2-methyl-phenoxy}-acetic acid (compound 96)
The title compound was prepared in the manner analogous to Example 1 using 96A. mp 152-153° C.; IR (thin film) cm−1: 1724, 1495, 1433, 1309, 1225, 1196; 400 MHz 1H NMR (DMSO-d6) δ 12.96 (br s, 1H), 7.82 (m, 2H), 7.54 (m, 2H), 7.19 (d, 1H, J=1.5 Hz), 7.12 (dd, 1H, J=2.1, 8.4 Hz), 6.97 (s, 1H), 6.72 (d, 1H, J=8.5 Hz), 4.63 (s, 2H), 4.11 (s, 2H), 2.09 (s, 3H); MS m/z 388 (M−1). Anal. Calc'd for C19H16ClNO4S: C, 58.54; H, 4.14; N, 3.59; found: C, 58.53; H, 4.08; N, 3.50.
Step 1. Preparation of {4-[5-(4-Chloro-phenyl)-isoxazol-3-ylmethylsulfanyl]-2-methyl-phenoxy}-acetic acid methyl ester (compound 96A)
The title compound was prepared in the manner analogous to Example 1F using 3-chloromethyl-5-(4-chloro-phenyl)-isoxazole and 2C. mp 79-80° C.; MS m/z 404 (M+1).
Step 2. Preparation of {4-[5-(4-Chloro-phenyl)-isoxazol-3-ylmethylsulfanyl]-2-methyl-phenoxy}-acetic acid (compound 96)
The title compound was prepared in the manner analogous to Example 1 using 96A. mp 152-153° C.; IR (thin film) cm−1: 1724, 1495, 1433, 1309, 1225, 1196; 400 MHz 1H NMR (DMSO-d6) δ 12.96 (br s, 1H), 7.82 (m, 2H), 7.54 (m, 2H), 7.19 (d, 1H, J=1.5 Hz), 7.12 (dd, 1H, J=2.1, 8.4 Hz), 6.97 (s, 1H), 6.72 (d, 1H, J=8.5 Hz), 4.63 (s, 2H), 4.11 (s, 2H), 2.09 (s, 3H); MS m/z 388 (M−1). Anal. Calc'd for C19H16ClNO4S: C, 58.54; H, 4.14; N, 3.59; found: C, 58.53; H, 4.08; N, 3.50.
Synthesis of {7-[5-(4-Chloro-phenyl)-isoxazol-3-ylmethylsulfanyl]-indan-4-yloxy}-acetic acid (compound 97)
Step 1. Preparation of {7-[5-(4-Chloro-phenyl)-isoxazol-3-ylmethylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 97A)
The title compound was prepared in the manner analogous to Example 1F using 3-chloromethyl-5-(4-chloro-phenyl)-isoxazole and 12C. mp 112-114° C.; MS m/z 430 (M+1).
Step 2. Preparation of {7-[5-(4-Chloro-phenyl)-isoxazol-3-ylmethylsulfanyl]-indan-4-yloxy}-acetic acid (compound 97)
The title compound was prepared in the manner analogous to Example 1 using 97A. mp 157-159° C.; IR (thin film) cm−1: 1731, 1612, 1465, 1433, 1231, 1110; 400 MHz 1H NMR (DMSO-d6) δ 7.81 (m, 2H), 7.54 (m, 2H), 7.10 (d, 1H, J=8.3 Hz), 6.95 (s, 1H), 6.59 (d, 1H, J=8.5 Hz), 4.62 (s, 2H), 4.06 (s, 2H), 2.77 (m, 4H), 1.92 (m, 2H); MS m/z 414 (M−1). Anal. Calc'd for C21H18ClNO4S: C, 60.65; H, 4.36; N, 3.37; found: C, 60.62; H, 4.10; N, 3.31.
Step 1. Preparation of {7-[5-(4-Chloro-phenyl)-isoxazol-3-ylmethylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 97A)
The title compound was prepared in the manner analogous to Example 1F using 3-chloromethyl-5-(4-chloro-phenyl)-isoxazole and 12C. mp 112-114° C.; MS m/z 430 (M+1).
Step 2. Preparation of {7-[5-(4-Chloro-phenyl)-isoxazol-3-ylmethylsulfanyl]-indan-4-yloxy}-acetic acid (compound 97)
The title compound was prepared in the manner analogous to Example 1 using 97A. mp 157-159° C.; IR (thin film) cm−1: 1731, 1612, 1465, 1433, 1231, 1110; 400 MHz 1H NMR (DMSO-d6) δ 7.81 (m, 2H), 7.54 (m, 2H), 7.10 (d, 1H, J=8.3 Hz), 6.95 (s, 1H), 6.59 (d, 1H, J=8.5 Hz), 4.62 (s, 2H), 4.06 (s, 2H), 2.77 (m, 4H), 1.92 (m, 2H); MS m/z 414 (M−1). Anal. Calc'd for C21H18ClNO4S: C, 60.65; H, 4.36; N, 3.37; found: C, 60.62; H, 4.10; N, 3.31.
Synthesis of {7-[3-(4-Chloro-phenyl)-isoxazol-5-ylmethylsulfanyl]-indan-4-yloxy}-acetic acid (compound 98)
Step 1. Preparation of {7-[3-(4-Chloro-phenyl)-isoxazol-5-ylmethylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 98A)
The title compound was prepared in the manner analogous to Example 1F using 5-chloromethyl-3-(4-chloro-phenyl)-isoxazole and 12C. mp 92-94° C.; MS m/z 430 (M+1).
Step 2. Preparation of {7-[3-(4-Chloro-phenyl)-isoxazol-5-ylmethylsulfanyl]-indan-4-yloxy}-acetic acid
The title compound was prepared in the manner analogous to Example 1 using 98A. mp 158-160° C.; IR (thin film) cm−1: 1741, 1574, 1476, 1429, 1252, 1110; 400 MHz 1H NMR (DMSO-d6) δ 7.78 (m, 2H), 7.51 (m, 2H), 7.13 (d, 1H, J=8.6 Hz), 6.72 (s, 1H), 6.60 (d, 1H, J=8.5 Hz), 4.62 (s, 2H), 4.20 (s, 2H), 2.76 (m, 4H), 1.91 (m, 2H); MS m/z 414 (M−1). Anal. Calc'd for C21H18ClNO4S: C, 60.65; H, 4.36; N, 3.37 found: C, 60.55; H, 4.17; N, 3.34.
Step 1. Preparation of {7-[3-(4-Chloro-phenyl)-isoxazol-5-ylmethylsulfanyl]-indan-4-yloxy}-acetic acid methyl ester (compound 98A)
The title compound was prepared in the manner analogous to Example 1F using 5-chloromethyl-3-(4-chloro-phenyl)-isoxazole and 12C. mp 92-94° C.; MS m/z 430 (M+1).
Step 2. Preparation of {7-[3-(4-Chloro-phenyl)-isoxazol-5-ylmethylsulfanyl]-indan-4-yloxy}-acetic acid
The title compound was prepared in the manner analogous to Example 1 using 98A. mp 158-160° C.; IR (thin film) cm−1: 1741, 1574, 1476, 1429, 1252, 1110; 400 MHz 1H NMR (DMSO-d6) δ 7.78 (m, 2H), 7.51 (m, 2H), 7.13 (d, 1H, J=8.6 Hz), 6.72 (s, 1H), 6.60 (d, 1H, J=8.5 Hz), 4.62 (s, 2H), 4.20 (s, 2H), 2.76 (m, 4H), 1.91 (m, 2H); MS m/z 414 (M−1). Anal. Calc'd for C21H18ClNO4S: C, 60.65; H, 4.36; N, 3.37 found: C, 60.55; H, 4.17; N, 3.34.
Synthesis of {5-Chloro-2-methyl-4-[5-(4-trifluoromethyl-phenyl)-isoxazol-3-ylmethylsulfanyl]-phenoxy}-acetic acid (compound 99)
Step 1. Preparation of {5-Chloro-2-methyl-4-[5-(4-trifluoromethyl-phenyl)-isoxazol-3-ylmethylsulfanyl]-phenoxy}-acetic acid methyl ester (compound 99A)
The title compound was prepared in the manner analogous to Example 1F using 3-chloromethyl-5-(4-chloro-phenyl)-isoxazole and 20C. mp 120-121° C.; MS m/z 472 (M+1).
Step 2. Preparation of {5-Chloro-2-methyl-4-[5-(4-trifluoromethyl-phenyl)-isoxazol-3-ylmethylsulfanyl]-phenoxy}-acetic acid (compound 99)
The title compound was prepared in the manner analogous to Example 1 using 99A. mp 180-182° C.; IR (thin film) cm−1: 1743, 1484, 1325, 1236, 1165, 1111; 400 MHz 1H NMR (DMSO-d6) δ 13.03 (br s, 1H), 8.03 (d, 2H, J=8.1 Hz), 7.84 (d, 2H, J=8.3 Hz), 7.33 (s, 1H), 7.14 (s, 1H), 6.96 (s, 1H), 4.71 (s, 2H), 4.23 (s, 2H), 2.07 (s, 3H); MS m/z 456 (M−1). Anal. Calc'd for C20H15ClF3NO4S: C, 52.47; H, 3.30; N, 3.06; found: C, 52.39; H, 3.02; N, 2.86.
Step 1. Preparation of {5-Chloro-2-methyl-4-[5-(4-trifluoromethyl-phenyl)-isoxazol-3-ylmethylsulfanyl]-phenoxy}-acetic acid methyl ester (compound 99A)
The title compound was prepared in the manner analogous to Example 1F using 3-chloromethyl-5-(4-chloro-phenyl)-isoxazole and 20C. mp 120-121° C.; MS m/z 472 (M+1).
Step 2. Preparation of {5-Chloro-2-methyl-4-[5-(4-trifluoromethyl-phenyl)-isoxazol-3-ylmethylsulfanyl]-phenoxy}-acetic acid (compound 99)
The title compound was prepared in the manner analogous to Example 1 using 99A. mp 180-182° C.; IR (thin film) cm−1: 1743, 1484, 1325, 1236, 1165, 1111; 400 MHz 1H NMR (DMSO-d6) δ 13.03 (br s, 1H), 8.03 (d, 2H, J=8.1 Hz), 7.84 (d, 2H, J=8.3 Hz), 7.33 (s, 1H), 7.14 (s, 1H), 6.96 (s, 1H), 4.71 (s, 2H), 4.23 (s, 2H), 2.07 (s, 3H); MS m/z 456 (M−1). Anal. Calc'd for C20H15ClF3NO4S: C, 52.47; H, 3.30; N, 3.06; found: C, 52.39; H, 3.02; N, 2.86.
Synthesis of 2-[2-butyl-4-({4-[4-(trifluoromethyl)phenyl]phenyl}methylthio)phenoxy]acetic acid (compound 100)
Step 1. Preparation of 2-((1E)buta-1,3-dienyl)-1-methoxybenzene (compound 100A)
Methyltriphenylphosphonium bromide (42.9 g, 0.12 mol) was suspended in 400 ml of anhydrous THF under nitrogen and cooled to −78° C. Sodium hydride (60% in mineral oil, 6.0 g, 0.15 mol) was added portionwise. The reaction mixture was allowed to warm up slowly to room temperature and stirred at the same temperature for 1 h, then 2-methoxycinnamaldehyde (16.2 g, 0.10 mol) in 200 ml of THF was added dropwise at room temperature, and stirred at the same temperature for 3 h. Water (200 ml) and diethyl ether (800 ml) were added. The organic layer was separated, dried over sodium sulfate, concentrated, and purified using normal phase chromatography to afford the title product. 400 MHz 1H NMR (CDCl3) δ 7.48-6.56 (m, 7H), 5.32 (d, 1H), 5.16 (d, 11H), 3.84 (s, 3H).
Step 2. Preparation of 2-butyl-1-methoxybenzene (compound 100B)
A mixture of the product from example 100A (12.8 g, 0.08 mol) and palladium/carbon (10%, 50% water, 12 g) in 400 ml of ethyl acetate was hydrogenated at 50 psi, at room temperature overnight, then filtered through Celite®, and concentrated to give 100B. 400 MHz 1H NMR (CDCl3) δ 7.16 (m, 2H), 6.85 (m, 2H), 3.81 (s, 3H), 2.61 (m, 2H), 1.57 (m, 2H), 1.38 (m, 2H), 0.92 (t, 3H).
Step 3. Preparation of 2-butylphenol (compound 100C)
To a stirred solution of the product from example 100B (13.1 g, 0.08 mol) in 400 ml of dichloromethane at −78° C. was added dropwise a solution of boron tribromide (100.2 g, 0.4 mol) in 200 ml of dichloromethane. After the completion of addition of boron tribromide, the reaction mixture was maintained at −78° C. for 1 h, then allowed to reach room temperature and stirred at the same temperature overnight. The mixture was cooled to 0° C., and carefully quenched with 100 ml of water. The mixture was extracted with ethyl acetate, washed with brine, dried over sodium sulfate, and concentrated to give 100C. 400 MHz 1H NMR (CDCl3) δ 7.10 (m, 2H), 6.86 (m, 1H), 6.78 (d, 1H), 4.61 (brs, 1H), 2.61 (m, 2H), 1.60 (m, 2H), 1.40 (m, 2H), 0.96 (t, 3H).
Step 4. Preparation of (3-butyl-4-hydroxyphenyl)thiocarbonitrile (compound 100D)
The title compound was prepared in the manner analogous to Example 1B with the product from example 100C (2.38 g, 0.016 mol), sodium thiocyanate (5.14 g, 0.063 mol), sodium bromide (1.63 g, 0.016 mol), and bromine (2.8 g, 0.017 mol) in methanol. 400 MHz 1H NMR (CDCl3) δ 7.30 (m, 2H), 6.81 (d, 1H), 5.49 (brs, 1H), 2.60 (m, 2H), 1.59 (m, 2H), 1.38 (m, 2H), 0.95 (t, 3H).
Step 5. Preparation of methyl 2-(2-butyl-4-cyanothiophenoxy)acetate (compound 100E)
The title compound was prepared in the manner analogous to Example 1C with the product from example 100D (2.80 g, 0.014 mol), methyl bromoacetate (2.28 g, 0.015 mol), and cesium carbonate (6.60 g, 0.020 mol), in 100 ml of acetonitrile. 400 MHz 1H NMR (CDCl3) δ 7.38 (m, 2H), 6.72 (d, 1H), 4.66 (s, 2H), 3.80 (s, 3H), 2.67 (m, 2H), 1.57 (m, 2H), 1.38 (m, 2H), 0.98 (t, 3H).
Step 6. Preparation of methyl 2-(2-butyl-4-sulfanylphenoxy) acetate (compound 100F)
The title compound was prepared in the manner analogous to Example 1D with the product from example 100E (2.79 g, 10.0 mol), dithiothreitol (3.08 g, 20.0 mmol), and 0.2 M potassium dihydrogenphosphate (15 ml) in 60 ml of methanol. 400 MHz 1H NMR (CDCl3) δ 7.11 (m, 2H), 6.60 (d, 1H), 4.61 (s, 2H), 3.81 (s, 3H), 3.36 (s, 1H), 2.63 (m, 2H), 1.57 (m, 2H), 1.38 (m, 2H), 0.98 (t, 3H).
Step 7. Preparation of methyl 2-[2-butyl-4-({4-[4-(trifluoromethyl)phenyl]phenyl}methylthio)phenoxy]acetate (compound 100G)
The title compound was prepared in the manner analogous to Example 1F using 100F and 1-(bromomethyl)-4-[4-(trifluoromethyl)phenyl]benzene prepared from and phosphorous tribromide and (4′-trifluoromethyl-biphenyl-4-yl)-methanol in a manner analogous to Example 3B. 400 MHz 1H NMR (CDCl3) δ 7.68 (m, 4H), 7.51 (d, 2H), 7.29 (d, 2H), 7.15 (m, 2H), 6.60 (d, 1H), 4.62 (s, 2H), 4.04 (s, 2H), 3.79 (s, 3H), 2.60 (m, 2H), 1.55 (m, 2H), 1.35 (m, 2H), 0.88 (t, 3H).
Step 8. Preparation of 2-[2-butyl-4-({4-[4-(trifluoromethyl)phenyl]phenyl}methylthio)phenoxy]acetic acid (compound 100)
The title compound was prepared in the manner analogous to Example 1 with the product from example 100G. mp 155-157° C.; 400 MHz 1H NMR (DMSOd-6) δ 7.88 (d, 2H), 7.80 (d, 2H), 7.65 (d, 2H), 7.38 (d, 2H), 7.17 (dd, 1H), 7.09 (d, 1H), 6.79 (d, 1H), 4.65 (s, 2H), 4.17 (s, 2H), 2.50 (m, 2H), 1.43 (m, 2H), 1.22 (m, 2H), 0.81 (t, 3H). MS m/z 473 (M−1). Anal. Calc'd for C26H25O3SF3: C, 65.81; H, 5.31; Found: C, 65.95; H, 5.36.
Step 1. Preparation of 2-((1E)buta-1,3-dienyl)-1-methoxybenzene (compound 100A)
Methyltriphenylphosphonium bromide (42.9 g, 0.12 mol) was suspended in 400 ml of anhydrous THF under nitrogen and cooled to −78° C. Sodium hydride (60% in mineral oil, 6.0 g, 0.15 mol) was added portionwise. The reaction mixture was allowed to warm up slowly to room temperature and stirred at the same temperature for 1 h, then 2-methoxycinnamaldehyde (16.2 g, 0.10 mol) in 200 ml of THF was added dropwise at room temperature, and stirred at the same temperature for 3 h. Water (200 ml) and diethyl ether (800 ml) were added. The organic layer was separated, dried over sodium sulfate, concentrated, and purified using normal phase chromatography to afford the title product. 400 MHz 1H NMR (CDCl3) δ 7.48-6.56 (m, 7H), 5.32 (d, 1H), 5.16 (d, 11H), 3.84 (s, 3H).
Step 2. Preparation of 2-butyl-1-methoxybenzene (compound 100B)
A mixture of the product from example 100A (12.8 g, 0.08 mol) and palladium/carbon (10%, 50% water, 12 g) in 400 ml of ethyl acetate was hydrogenated at 50 psi, at room temperature overnight, then filtered through Celite®, and concentrated to give 100B. 400 MHz 1H NMR (CDCl3) δ 7.16 (m, 2H), 6.85 (m, 2H), 3.81 (s, 3H), 2.61 (m, 2H), 1.57 (m, 2H), 1.38 (m, 2H), 0.92 (t, 3H).
Step 3. Preparation of 2-butylphenol (compound 100C)
To a stirred solution of the product from example 100B (13.1 g, 0.08 mol) in 400 ml of dichloromethane at −78° C. was added dropwise a solution of boron tribromide (100.2 g, 0.4 mol) in 200 ml of dichloromethane. After the completion of addition of boron tribromide, the reaction mixture was maintained at −78° C. for 1 h, then allowed to reach room temperature and stirred at the same temperature overnight. The mixture was cooled to 0° C., and carefully quenched with 100 ml of water. The mixture was extracted with ethyl acetate, washed with brine, dried over sodium sulfate, and concentrated to give 100C. 400 MHz 1H NMR (CDCl3) δ 7.10 (m, 2H), 6.86 (m, 1H), 6.78 (d, 1H), 4.61 (brs, 1H), 2.61 (m, 2H), 1.60 (m, 2H), 1.40 (m, 2H), 0.96 (t, 3H).
Step 4. Preparation of (3-butyl-4-hydroxyphenyl)thiocarbonitrile (compound 100D)
The title compound was prepared in the manner analogous to Example 1B with the product from example 100C (2.38 g, 0.016 mol), sodium thiocyanate (5.14 g, 0.063 mol), sodium bromide (1.63 g, 0.016 mol), and bromine (2.8 g, 0.017 mol) in methanol. 400 MHz 1H NMR (CDCl3) δ 7.30 (m, 2H), 6.81 (d, 1H), 5.49 (brs, 1H), 2.60 (m, 2H), 1.59 (m, 2H), 1.38 (m, 2H), 0.95 (t, 3H).
Step 5. Preparation of methyl 2-(2-butyl-4-cyanothiophenoxy)acetate (compound 100E)
The title compound was prepared in the manner analogous to Example 1C with the product from example 100D (2.80 g, 0.014 mol), methyl bromoacetate (2.28 g, 0.015 mol), and cesium carbonate (6.60 g, 0.020 mol), in 100 ml of acetonitrile. 400 MHz 1H NMR (CDCl3) δ 7.38 (m, 2H), 6.72 (d, 1H), 4.66 (s, 2H), 3.80 (s, 3H), 2.67 (m, 2H), 1.57 (m, 2H), 1.38 (m, 2H), 0.98 (t, 3H).
Step 6. Preparation of methyl 2-(2-butyl-4-sulfanylphenoxy) acetate (compound 100F)
The title compound was prepared in the manner analogous to Example 1D with the product from example 100E (2.79 g, 10.0 mol), dithiothreitol (3.08 g, 20.0 mmol), and 0.2 M potassium dihydrogenphosphate (15 ml) in 60 ml of methanol. 400 MHz 1H NMR (CDCl3) δ 7.11 (m, 2H), 6.60 (d, 1H), 4.61 (s, 2H), 3.81 (s, 3H), 3.36 (s, 1H), 2.63 (m, 2H), 1.57 (m, 2H), 1.38 (m, 2H), 0.98 (t, 3H).
Step 7. Preparation of methyl 2-[2-butyl-4-({4-[4-(trifluoromethyl)phenyl]phenyl}methylthio)phenoxy]acetate (compound 100G)
The title compound was prepared in the manner analogous to Example 1F using 100F and 1-(bromomethyl)-4-[4-(trifluoromethyl)phenyl]benzene prepared from and phosphorous tribromide and (4′-trifluoromethyl-biphenyl-4-yl)-methanol in a manner analogous to Example 3B. 400 MHz 1H NMR (CDCl3) δ 7.68 (m, 4H), 7.51 (d, 2H), 7.29 (d, 2H), 7.15 (m, 2H), 6.60 (d, 1H), 4.62 (s, 2H), 4.04 (s, 2H), 3.79 (s, 3H), 2.60 (m, 2H), 1.55 (m, 2H), 1.35 (m, 2H), 0.88 (t, 3H).
Step 8. Preparation of 2-[2-butyl-4-({4-[4-(trifluoromethyl)phenyl]phenyl}methylthio)phenoxy]acetic acid (compound 100)
The title compound was prepared in the manner analogous to Example 1 with the product from example 100G. mp 155-157° C.; 400 MHz 1H NMR (DMSOd-6) δ 7.88 (d, 2H), 7.80 (d, 2H), 7.65 (d, 2H), 7.38 (d, 2H), 7.17 (dd, 1H), 7.09 (d, 1H), 6.79 (d, 1H), 4.65 (s, 2H), 4.17 (s, 2H), 2.50 (m, 2H), 1.43 (m, 2H), 1.22 (m, 2H), 0.81 (t, 3H). MS m/z 473 (M−1). Anal. Calc'd for C26H25O3SF3: C, 65.81; H, 5.31; Found: C, 65.95; H, 5.36.
Preparation of {6-methyl-8-[4-(4-trifluoromethyl-benzyloxy)-benzyl-sulfanyl]-chroman-5-yloxy}-acetic acid (compound 101)
Step 1. {6-methyl-8-[4-(4-trifluoromethyl-benzyloxy)-benzylsulfanyl]-chroman-5-yloxy}-acetic acid methyl ester (compound 101A)
The title compound was prepared in the manner analogous to Example 1F using (8-mercapto-6-methyl-chroman-5-yloxy)-acetic acid methyl ester and the product from Example 14B. MS m/z 533 (M+1).
Step 2. {6-methyl-8-[4-(4-trifluoromethyl-benzyloxy)-benzylsulfanyl]-chroman-5-yloxy}-acetic acid (compound 101)
2N KOH solution (5 ml) was added to a stirred slurry of the product from Example 101A (0.4g, 0.75 mmol) in 2-3 ml of methanol, and the mixture was heated briefly on a steambath until nearly clear, then stirred at room temp. After 3 hours the mixture was diluted with 15-20 ml of ice-water and acidified with H3PO4. After 15 minutes the precipitate was filtered off, rinsed 3× with water, and dried to afford the title product, 0.2g. 51% 400 MHz 1H NMR (DMSO-d6) δ 7.70 (d, 2H, J=8 Hz), 7.59 (d, 2H, J=8 Hz), 7.18 (d, 2H, J=8.5 Hz), 6.88 (d, 2H, J=8.5 Hz), 6.83 (s, 1H), 5.14 (s, 2H), 4.21 (s, 2H), 4.06 (t, 2H, J=4.9Hz), 3.95 (s, 2H), 2.63 (t, 2H, J=6.3Hz), 2.03 (s, 3H), 1.79 (quint, 2H); MS m/z 517 (M−1).
Step 1. {6-methyl-8-[4-(4-trifluoromethyl-benzyloxy)-benzylsulfanyl]-chroman-5-yloxy}-acetic acid methyl ester (compound 101A)
The title compound was prepared in the manner analogous to Example 1F using (8-mercapto-6-methyl-chroman-5-yloxy)-acetic acid methyl ester and the product from Example 14B. MS m/z 533 (M+1).
Step 2. {6-methyl-8-[4-(4-trifluoromethyl-benzyloxy)-benzylsulfanyl]-chroman-5-yloxy}-acetic acid (compound 101)
2N KOH solution (5 ml) was added to a stirred slurry of the product from Example 101A (0.4g, 0.75 mmol) in 2-3 ml of methanol, and the mixture was heated briefly on a steambath until nearly clear, then stirred at room temp. After 3 hours the mixture was diluted with 15-20 ml of ice-water and acidified with H3PO4. After 15 minutes the precipitate was filtered off, rinsed 3× with water, and dried to afford the title product, 0.2g. 51% 400 MHz 1H NMR (DMSO-d6) δ 7.70 (d, 2H, J=8 Hz), 7.59 (d, 2H, J=8 Hz), 7.18 (d, 2H, J=8.5 Hz), 6.88 (d, 2H, J=8.5 Hz), 6.83 (s, 1H), 5.14 (s, 2H), 4.21 (s, 2H), 4.06 (t, 2H, J=4.9Hz), 3.95 (s, 2H), 2.63 (t, 2H, J=6.3Hz), 2.03 (s, 3H), 1.79 (quint, 2H); MS m/z 517 (M−1).
Preparation of {4-[5-(4-trifluoromethyl-phenyl)-isoxazol-3-ylmethylsulfanyl]-5,6,7,8-tetrahydro-naphthalen-1-yloxy}-acetic acid (compound 102)
Step 1. {4-[5-(4-trifluoromethyl-phenyl)-isoxazol-3-ylmethylsulfanyl]-5,6,7,8-tetrahydro-naphthalen-1-yloxy}-acetic acid methyl ester (compound 102A)
The title compound was prepared in the manner analogous to Example 1F using (4-mercapto-5,6,7,8-tetrahydro-naphthalen-1-yloxy)-acetic acid methyl ester and the product from Example 42C; recrystallization from methanol afforded the title compound. MS m/z 478 (M+1).
Step 2. {4-[5-(4-trifluoromethyl-phenyl)-isoxazol-3-ylmethylsulfanyl]-5,6,7,8-tetrahydro-naphthalen-1-yloxy}-acetic acid (compound 102)
The title compound was prepared in the manner analogous to Example 1 using the product from Example 102A. mp 167-170° C.; 400 MHz 1H NMR (DMSO-d6) δ 8.01 (d, 2H, J=8 Hz), 7.83 (d, 2H, J=8.3 Hz), 7.14 (d, 2H, J=8.5 Hz), 7.07 (s, 1H), 6.58 (d, 2H, J=8.8 Hz), 4.56 (s, 2H), 4.07 (s, 2H), 2.64 (m, 2H), 2.53 (m, 2H), 1.60 (m, 4H); MS m/z 464 (M+1).
The compounds of the present invention can also be prepared using combinatorial chemistry methods. In particular, compounds of Examples 103-134 were prepared using combinatorial chemistry analogous to that previously described in Examples 1-102. The combinatorial chemistry methods useful in the present invention include those where an activated alcohol is contacted with a thiol followed by saponification of the resulting ester to afford the desired products. Such methods can be illustrated by previously described Scheme 1 where compound D is an exemplary thiol, compound Y is an exemplary activated alcohol, and compound F is an exemplary desired product.
Step 1. {4-[5-(4-trifluoromethyl-phenyl)-isoxazol-3-ylmethylsulfanyl]-5,6,7,8-tetrahydro-naphthalen-1-yloxy}-acetic acid methyl ester (compound 102A)
The title compound was prepared in the manner analogous to Example 1F using (4-mercapto-5,6,7,8-tetrahydro-naphthalen-1-yloxy)-acetic acid methyl ester and the product from Example 42C; recrystallization from methanol afforded the title compound. MS m/z 478 (M+1).
Step 2. {4-[5-(4-trifluoromethyl-phenyl)-isoxazol-3-ylmethylsulfanyl]-5,6,7,8-tetrahydro-naphthalen-1-yloxy}-acetic acid (compound 102)
The title compound was prepared in the manner analogous to Example 1 using the product from Example 102A. mp 167-170° C.; 400 MHz 1H NMR (DMSO-d6) δ 8.01 (d, 2H, J=8 Hz), 7.83 (d, 2H, J=8.3 Hz), 7.14 (d, 2H, J=8.5 Hz), 7.07 (s, 1H), 6.58 (d, 2H, J=8.8 Hz), 4.56 (s, 2H), 4.07 (s, 2H), 2.64 (m, 2H), 2.53 (m, 2H), 1.60 (m, 4H); MS m/z 464 (M+1).
The compounds of the present invention can also be prepared using combinatorial chemistry methods. In particular, compounds of Examples 103-134 were prepared using combinatorial chemistry analogous to that previously described in Examples 1-102. The combinatorial chemistry methods useful in the present invention include those where an activated alcohol is contacted with a thiol followed by saponification of the resulting ester to afford the desired products. Such methods can be illustrated by previously described Scheme 1 where compound D is an exemplary thiol, compound Y is an exemplary activated alcohol, and compound F is an exemplary desired product.
| Example | ||
| No. | Name | MS m/z |
| 103 | (4-{2-Butyl-5-chloro-1-[4-(1-cyano-cyclopentyl)- | 552.37 |
| benzyl]-1H-imidazol-4-ylmethylsulfanyl}-2-methyl- | ||
| phenoxy)-acetic acid | ||
| 104 | [4-(5-Biphenyl-4-yl-2-thiopen-2-yl-4,5-dihydro- | 546.33 |
| oxazol-4-ylmethylsulfanyl)-5-methoxy-2-methyl- | ||
| phenoxy]-acetic acid | ||
| 105 | {4-[2-(4-Bromo-phenoxy)-ethylsulfanyl]-2,6- | 451.33 |
| dimethyl-phenoxy}-acetic acid | ||
| 106 | [4-(3-{2-[4-(2-Diethylamino-ethoxy)-phenyl]- | 578.43 |
| benzimidazol-1-yl]-propylsulfanyl)-5-methoxy-2- | ||
| methyl-phenoxy]-acetic acid | ||
| 107 | [4-(5-Biphenyl-4-yl-2-thiophen-2-yl-4,5-dihydro- | 516.31 |
| oxazol-4-ylmethylsulfanyl)-2-methyl-phenoxy]- | ||
| acetic acid | ||
| 108 | (4-{2-[3-(4-Fluoro-phenyl)-benzo[b]thiophen-7-yl]- | 451.24 |
| ethylsulfanyl}-2-methyl-phenoxy)-acetic acid | ||
| 109 | {2-Methyl-4-[2-(5-phenyl-naphthalen-1-yloxy)- | 443.3 |
| ethylsulfanyl]-phenoxy}-acetic acid | ||
| 110 | [2-Methyl-4-(3-phenoxy-benzylsulfanyl)-phenoxy]- | 379.23 |
| acetic acid | ||
| 111 | [2,5-Dimethyl-4-(5-p-tolyl-1,3,4-oxadiazol-2- | 385.22 |
| ylmethylsulfanyl)-phenoxy]-acetic acid | ||
| 112 | [2-Methyl-4-(4-pyrazol-1-yl-benzylsulfanyl)- | 355.2 |
| phenoxy]-acetic acid | ||
| 113 | [2-Methyl-4-(5-methyl-3-phenyl-isoxazol-4- | 370.2 |
| ylmethylsulfanyl)-phenoxy]-acetic acid | ||
| 114 | [4-(Biphenyl-2-ylmethylsulfanyl)-2-methyl- | 363.2 |
| phenoxy]-acetic acid | ||
| 115 | {4-[5-(4-Chloro-phenyl)-isoxazol-3- | 390.16 |
| ylmethylsulfanyl]-2-methyl-phenoxy}-acetic acid | ||
| 116 | [2-Methyl-4-(5-p-tolyl-1,3,4-oxadizol-2- | 371.19 |
| ylmethylsulfanyl)-phenoxy]-acetic acid | ||
| 117 | {4-[3-(4-Chloro-phenyl)-1,2,4-oxadiazol-5- | 391.14 |
| ylmethylsulfanyl]-2-methyl-phenoxy}-acetic acid | ||
| 118 | [2,5-Dimethyl-4-(4-pyrazol-1-yl-benzylsulfanyl)- | 369.2 |
| phenoxy]-acetic acid | ||
| 119 | [4-(Biphenyl-2-ylmethylsulfanyl)-2,5-dimethyl- | 377.19 |
| phenoxy]-acetic acid | ||
| 120 | [4-(4-Benzyloxy-benzylsulfanyl)-5,6,7,8-tetrahydro- | 433.41 |
| naphthalen-1-yloxy]-acetic acid | ||
| 121 | [4-(4-Benzyloxy-benzylsulfanyl)-2,6-dimethyl- | 407.39 |
| phenoxy]-acetic acid | ||
| 122 | [4-(4-Benzyloxy-benzylsulfanyl)-2,5-dimethyl- | 407.38 |
| phenoxy]-acetic acid | ||
| 123 | [4-(4-Benzyloxy-benzylsulfanyl)-2-methyl- | 393.38 |
| phenoxy]-acetic acid | ||
| 124 | [4-(4-Benzyloxy-benzylsulfanyl)-phenoxy]-acetic | 379.35 |
| acid | ||
| 125 | [4-(Biphenyl-4-ylmethylsulfanyl)-5,6,7,8-tetrahydro- | 403.4 |
| naphthalen-1-yloxy]-acetic acid | ||
| 126 | [4-(Biphenyl-4-ylmethylsulfanyl)-2,6-dimethyl- | 377.38 |
| phenoxy]-acetic acid | ||
| 127 | [4-(Biphenyl-4-ylmethylsulfanyl)-2,5-dimethyl- | 377.37 |
| phenoxy]-acetic acid | ||
| 128 | [4-(Biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic | 349.32 |
| acid | ||
| 129 | {4-[3-(2-Fluoro-phenoxy)-benzylsulfanyl]-2,6- | 411.2 |
| dimethyl-phenoxy}-acetic acid | ||
| 130 | [4-(2-{4-[2-(3-Chloro-4-cyclohexyl-phenyl)-ethyl]- | 531.31 |
| piperazin-1-yl}-ethylsulfanyl)-2-methyl-phenoxy]- | ||
| acetic acid | ||
| 131 | [5-Methoxy-2-methyl-4-(2-{4-[2-(3-phenyl- | 561.37 |
| benzofuran-7-yl)-ethyl]-piperazin-1-yl}- | ||
| ethylsulfanyl)-phenoxy]-acetic acid | ||
| 132 | {4-[2-(2,6-Diphenyl-piperidin-1-yl)-ethylsulfanyl]- | 492.31 |
| 5-methoxy-2-methyl-phenoxy}-acetic acid | ||
| 133 | [2-Methyl-4-(2-{4-[2-(3-phenyl- | 531.32 |
| benzofuran-7-yl)-ethyl]-piperazin-1-yl)- | ||
| ethylsulfanyl)-phenoxy]-acetic acid | ||
| 134 | {4-[2-(2,6-Diphenyl-piperidin-1-yl)-ethylsulfanyl]- | 462.25 |
| 2-methyl-phenoxy}-acetic acid | ||
The preparation of Examples 103-134 is further described below.
Preparation of Thiols used in Combinatorial Methods
The title compound was prepared in a manner analogous to compound 1B. 400 MHz 1H NMR (DOMSO-d6) δ 10.0 (s, 1H), 7.35 (s, 1H), 6.73 (s, 1H), 2.3 (s, 3H), 2.04 (s, 3H); MS m/z 180 (m+1).
The title compound was prepared from compound WWA in a manner analogous to compound 1C. 400 MHz 1H NMR (DMSO-d6) δ 7.07 (s, 1H), 6.50 (s, 1H), 4.56 (s, 2H), 3.76 (s, 3H), (s, 1H), 2.26 (s, 3H), 2.17 (s, 3H); MS m/z 252 (m+1).
The title compound was prepared from compound WWB in a manner analogous to compound 1D. 400 MHz 1H NMR (DMSO-d6) δ 7.07 (s, 1H), 6.50 (s, 1H), 4.56 (s, 2H), 3.76 (s, 3H), 3.07 (s, 1H), 2.26 (s, 3H), 2.17 (s, 3H); MS m/z 227 (M+1).
Compound XXA was prepared from 2,6-dimethylphenol in a similar manner as described for compound 1B. 400 MHz 1H NMR (DMSO-d6) δ 8.96 (s, 1H), 7.22 (s, 2H), 2.13 (s, 6H).
Compound XXB was prepared from compound XXA in a similar manner as described for compound 1C to give 2.5 g (46%) of the title compound pure enough for subsequent use 400 MHz 1H NMR (DMSO-d6) δ 7.11 (s, 2H), 4.41 (s, 2H), 3.63 (s, 3H), 2.14 (s, 6H).
Compound XX was prepared from compound XXB in a similar manner as described for compound 1D to give, after purification by flash column chromatography (gradient elution: 100% hexanes to 30% EtOAc/hexanes), 1.8 g (82%) of the title compound. 400 MHz 1H NMR (DMSO-d6) δ 6.90 (s, 2H), 5.51 (s, 1H), 4.39 (s, 2H), 3.66 (s, 3H), 2.10 (s, 6H); MS m/z 225 (M−1).
5,6,7,8-Tetrahydro-naphthalen-1-ol (1 g, 6.8 mmol) was dissolved in 25 ml acetonitrile. Sodium thiocyanate (1.76 g, 22 mmol) and sodium bromide (0.7 g, 6.8 mmol) were added and stirred for 5 minutes at ambient temperature. Bromine (1.2 g, 7.48 mmol) was added drop wise over 5 minutes. The orange solution was allowed to stir two hours. Brine was added and the crude product was extracted twice into ethyl acetate. The combined organic extracts were washed once with brine, dried over anhydrous sodium sulfate, decanted and concentrated. Normal phase chromatography afforded the title product, 1.28 g, 92%. 400 MHz 1H MR (DMSO-d6) δ 11.1 (s, 1H), 7.40 (d, 1H, J=8.8 Hz), 6.61 (d, 1H, 8.8 Hz), 2.78 (m, 2H), 2.59 (m, 2H), 1.70 (m, 4H). MS m/z 278 (m+1)
Preparation of (4-Thiocyanato-5,6,7,8-tetrahydro-naphthalen-1-yloxy)-acetic acid methyl ester (compound YYB)
The title compound was prepared in the manner analogous to example 1C utilizing compound YYA. 400 MHz 1H NMR (DMSO-d6) δ 7.4 (d, 1H, J=8.8 Hz), 6.80 (d, 1H, 8.8 Hz), 4.84 (s, 2H), 3.64 (s, 3H), 2.78 (m, 2H), 2.59 (m, 2H), 1.70 (m, 4H). MS m/z 278 (m+1).
Preparation of (4-Mercapto-5,6,7,8tetrahydro-naphthalen-1-yloxy)-acetic acid methyl ester (compound YY)
The title compound was prepared in the manner analogous to example 1D utilizing compound YYB. 400 MHz 1H NMR (DMSO-d6) δ 7.08 (d, 1H, J=8.8 Hz), 6.55 (d, 1H, 8.8 Hz), 4.71 (s, 1H), 4.70 (s, 2H), 3.63 (s, 3H), 2.45 (m, 2H), 2.44 (m, 2H), 1.65 (m, 4H). MS m/z 253 (M+1).
Preparation of (4-Thiocyanato-5,6,7,8-tetrahydro-naphthalen-1-yloxy)-acetic acid methyl ester (compound YYB)
The title compound was prepared in the manner analogous to example 1C utilizing compound YYA. 400 MHz 1H NMR (DMSO-d6) δ 7.4 (d, 1H, J=8.8 Hz), 6.80 (d, 1H, 8.8 Hz), 4.84 (s, 2H), 3.64 (s, 3H), 2.78 (m, 2H), 2.59 (m, 2H), 1.70 (m, 4H). MS m/z 278 (m+1).
Preparation of (4-Mercapto-5,6,7,8tetrahydro-naphthalen-1-yloxy)-acetic acid methyl ester (compound YY)
The title compound was prepared in the manner analogous to example 1D utilizing compound YYB. 400 MHz 1H NMR (DMSO-d6) δ 7.08 (d, 1H, J=8.8 Hz), 6.55 (d, 1H, 8.8 Hz), 4.71 (s, 1H), 4.70 (s, 2H), 3.63 (s, 3H), 2.45 (m, 2H), 2.44 (m, 2H), 1.65 (m, 4H). MS m/z 253 (M+1).
The title compound was prepared in the manner analogous to Example 1B using phenol. MS m/z 152 (M+1)
The title compound was prepared in the manner analogous to Example 1C using ZZA. MS m/z 224 (M+1)
The title compound was prepared in the manner analogous to Example 1D using ZZB. MS m/z 197 (M−1)
Preparation of Examples 103-134
Alcohol Preparation:
The appropriate alcohols in the salt form (0.65 mmol) were dissolved in 3.0 mL of low water MeOH. MP-CO3 (3.21 mmol/g, 3.70 equivalents to alcohol, 2.41 mmol) was then added to each vial containing the alcohol and shaken at ambient temperature for 3 h. The samples were then filtered into tared vials and concentrated.
Alcohol Activation:
Each alcohol sample was then diluted to 0.15M with DCM and 1.0 ML of each delivered to a reaction tube. PS-morpholine (4.0 mmol/g, 2 equivalents to alcohol, 0.3 mmol) and 25 μL methanesulfonyl chloride was then added to each reaction tube. The reaction tubes were shaken at ambient temperature for 16 h. The samples were then filtered into collection tubes, the resin rinsed with two 1.0 mL aliquots of DCM, and concentrated.
Alkylation:
Examples 103-134 were synthesized in the following fashion using either the thiol products 2C, 1D, WW, XX, YY or ZZ and the appropriate activated alcohol or alkyl halide. Each thiol was diluted to 0.15M with CH3CN and each activated alcohol diluted to 0.15M with CH3CN. 1.0 mL aliquots of each thiol (0.15 mmol) and 1.0 mL aliquots of each activated alcohol (1.0 equivalents, 0.15 mmol) were then delivered to a reaction tube and 100 mg Cs2CO3 (2 equivalents, 0.3 mmol) was added. The reaction tubes were shaken at 60° C. for 2.5 h. The reaction mixtures were filtered into collection tubes, the resin rinsed with two 1.0 mL aliquots of CH3CN, and concentrated.
Saponification:
Examples 103-134 were synthesized in the following fashion using the products from the alkylation step discussed above. Each alkylation product was diluted with 3.0 mL of 0.5M LiOH in 4:1 methoxyethanol:H2O shaken at 60° C. for 4 h and cooled to ambient temperature. To each reaction, was then added 1.0 mL 1N HCl and 1.0 mL brine. Each reaction was extracted twice with 2.0 mL EtOAc, and the organic layers concentrated to afford the desired products.
Alcohol Activation:
Each alcohol sample was then diluted to 0.15M with DCM and 1.0 ML of each delivered to a reaction tube. PS-morpholine (4.0 mmol/g, 2 equivalents to alcohol, 0.3 mmol) and 25 μL methanesulfonyl chloride was then added to each reaction tube. The reaction tubes were shaken at ambient temperature for 16 h. The samples were then filtered into collection tubes, the resin rinsed with two 1.0 mL aliquots of DCM, and concentrated.
Alkylation:
Examples 103-134 were synthesized in the following fashion using either the thiol products 2C, 1D, WW, XX, YY or ZZ and the appropriate activated alcohol or alkyl halide. Each thiol was diluted to 0.15M with CH3CN and each activated alcohol diluted to 0.15M with CH3CN. 1.0 mL aliquots of each thiol (0.15 mmol) and 1.0 mL aliquots of each activated alcohol (1.0 equivalents, 0.15 mmol) were then delivered to a reaction tube and 100 mg Cs2CO3 (2 equivalents, 0.3 mmol) was added. The reaction tubes were shaken at 60° C. for 2.5 h. The reaction mixtures were filtered into collection tubes, the resin rinsed with two 1.0 mL aliquots of CH3CN, and concentrated.
Saponification:
Examples 103-134 were synthesized in the following fashion using the products from the alkylation step discussed above. Each alkylation product was diluted with 3.0 mL of 0.5M LiOH in 4:1 methoxyethanol:H2O shaken at 60° C. for 4 h and cooled to ambient temperature. To each reaction, was then added 1.0 mL 1N HCl and 1.0 mL brine. Each reaction was extracted twice with 2.0 mL EtOAc, and the organic layers concentrated to afford the desired products.
The compounds of the present invention have demonstrated PPAR modulating activity in the standard assays commonly employed by those skilled in the art. Accordingly, such compounds and formulations comprising such compounds are useful for suppressing appetite, modulating leptin, and treating, preventing or controlling hypercholesterolemia, dyslipidemia, obesity, eating disorders, hyperglycemia, atherosclerosis, hypertriglyceridemia, hyperinsulinemia and diabetes.
A. Selectivity Measurements
1. Test A. Transient Transfections Assay using the HepG2Hepatoma Cell Line.
HepG2 cells were transiently transfected with an expression plasmids encoding hPPARα, hPPARβ or mPPARγ chimeric receptors and a reporter containing the yeast upstream activating sequence (UAS) upstream of the viral E1B promoter controlling a luceriferase reporter gene. In addition, the plasmid pRSVβ-gal was used to control the transfection efficiency, HepG2 cells were grown in DMEM supplemented with 10% FBS and 1 μM non-essential amino acid. On the first day, cells were split into 100 mm dishes at 2.5×108/dish and incubated overnight at 37° C./5% CO2. On the second day the cells were transiently transfected with plasmid DNA encoding a chimeric receptor, the luciferase reporter gene; and β-gal. For each 100 mm dish, 15 μg of lucifease reporter (PG5E1b) DNA, 15 μg of Gal4-PPAR chimeric receptor DNA, and 1.5 μg of β-gal plasmid DNA were mixed with 1.4 ml of opti-MEM in the tube. 28 μl of LipoFectamine-2000 reagent was added to 1.4 ml of opti-MEM in the tube, and incubate for 5 min at RT. The diluted LipoFectamine-2000 reagent was combined with the DNA mixture, and incubate for 20 min at RT. After fresh medium was added to each 100 mm dish of cells, 2.8 ml of Lipofectamine2000-DNA mixture was added dropwise to the 100 mm dish containing 14 ml of medium, and incubate 37° C. overnight. On day three cells were trypsinized off the 100 mm dishes and re-plated on 96 well plates. Cells were plated at 2.5×104 cells per well in 150 μl of media and 50 μl of compound diluted by media was added. The test compound added were in the range from 50 μM to 50 pM. After addition of compounds, the plates were incubated at 37° C. for 24 hours. Subseqently cells were washed with once with 100 μl of PBS, lysed, and processed for measuring luciferase and β-gal activity using Dual-Light luciferase kit from Tropix®, according to the manufacturer's recommendations, on an EG&G Bethold MicroLumat LB96P luminometer. EC50 values were obtained using the GraphPad Prism™ program. Surprisingly, the compounds of the present invention exhibit activity for both PPARα and PPARβ. Compounds of the present invention exhibited a range of Hep G2-hBeta EC50's (“EC50β”) and Hep G2-hAlpha EC50's (“EC50α”) from greater than zero to about 20 μM. Specifically, as shown in Table 1, the Hep G2-hBeta EC50's and Hep G2-hAlpha EC50's for the compounds of the present invention fall within the following 6 groups:
I) >0-300 nM
II) >300-500 nM
III) >500-1000 nM
IV) >1000-2000 nM
V) >2000-5000 nM
VI) >5000 nM
| TABLE 1 | ||
| Ec50β nM | EC50α nM | |
| Compound | (Group) | (Group) |
| 1 | I | I |
| 3 | I | V |
| 4 | I | I |
| 5 | IV | — |
| 7 | I | I |
| 9 | I | I |
| 10 | I | II |
| 11 | I | III |
| 12 | I | — |
| 14 | I | I |
| 15 | I | — |
| 16 | I | I |
| 17 | IV | V |
| 18 | I | VI |
| 19 | I | I |
| 20 | I | V |
| 21 | I | I |
| 22 | IV | III |
| 23 | I | I |
| 25 | V | IV |
| 26 | I | — |
| 27 | V | III |
| 28 | IV | IV |
| 29 | I | I |
| 30 | IV | IV |
| 31 | VI | IV |
| 32 | II | IV |
| 33 | IV | IV |
| 34 | I | I |
| 35 | II | I |
| 36 | V | I |
| 37 | I | — |
| 38 | I | — |
| 39 | I | — |
| 40 | III | — |
| 41 | I | V |
| 42 | I | V |
| 43 | I | III |
| 44 | III | VI |
| 45 | I | — |
| 46 | I | I |
| 47 | VI | — |
| 48 | I | V |
| 49 | I | II |
| 50 | V | — |
| 51 | V | — |
| 52 | I | III |
| 53 | I | IV |
| 54 | II | — |
| 57 | V | IV |
| 58 | V | — |
| 59 | IV | VI |
| 60 | VI | — |
| 61 | II | — |
| 62 | IV | VI |
| 63 | I | V |
| 64 | VI | — |
| 65 | VI | — |
| 66 | VI | — |
| 67 | I | — |
| 68 | IV | — |
| 69 | V | — |
| 70 | III | — |
| 71 | III | — |
| 72 | IV | — |
| 73 | I | V |
| 74 | I | V |
| 75 | III | — |
| 76 | IV | — |
| 77 | II | — |
| 78 | II | — |
| 79 | II | VI |
| 80 | IV | — |
| 81 | I | VI |
| 84 | II | VI |
| 85 | IV | — |
| 87 | V | — |
| 88 | VI | — |
| 89 | IV | — |
| 90 | III | — |
| 91 | V | — |
| 92 | I | I |
| 93 | III | — |
| 97 | III | VI |
| 98 | V | — |
| 99 | II | — |
| 100 | III | — |
B. Effect of PPAR Modulators on Lipid and Human Apoprotein A1 Concentrations in the hApoA1 Transgenic Mouse
Mice, transgenic for human apoA1, were purchased from Jackson laboratories. All animals were allowed normal chow (Ralston-Purina) and water ad libitum in temperature controlled rooms, under a 12-h light, 12-h dark cycle beginning with lights on at 6 AM. During the treatment phase of the study the mice were dosed daily between 6 and 9 AM by oral gavage using a suspension vehicle of 1.5% carboxymethyl-cellulose plus 0.2 percent Tween-20 (CMC/Tween) containing the specifid compounds. Control animals received vehicle alone. Vehicle volume represented 0.25 percent of body weight. Under anesthesia, tail blood was obtained weekly in the morning at the indicated days of study. At termination, tissue samples (liver, intestine, fat, and muscle) were taken to study effects on genes effecting lipid metabolism. Each of the compounds of the present invention that were tested effected a significant increase in HDL over the values observed for the control animals. Furthermore, these compounds resulted in triglyceride levels which were lower than observed in controls. Componds of the present invention tested in the hApoAI transgenic mouse model showed a range of HDL-c elevation and triglyceride lowering when dosed at 30 mg/kg/day. For instance, Example 4 raised HDL-c 97% and lowered triglycerides 65 % relative to the control population, Example 6 raised HDL-c 24% and lowered triglycerides 59 % relative to the control population, and Example 3 raised HDL-c 9% and lowered triglycerides 70% relative to the control population.
5 C. Effect of Compounds of the Invention on Insulin Resistant or Diabetic Cynomolgus Monkeys
Cynomolgus monkeys that were either insulin resistant or diabetic (type II) were treated for eight weeks with [5-Methoxy-2-methyl-4-(4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid in a rising dose fashion (0.1 to 1 mg/kg). Plasma was sampled bi-weekly and analyzed for glycemics and leptin. Body weight was also measured at various time points across the study. The diabetic moneky mean body weight data is presented in Table 2 and the insulin resistant monkey mean body weight data is presented in Table 3.
| TABLE 2 |
| Diabetic Monkey Mean Body Weight |
| Mean | ||||||||
| −7 wk | −4 wk | Baseline | 2 wk | 5 wk | 7 wk | 8 wk | ||
| Diab Ctl | 8.10 | 8.05 | 8.08 | 8.17 | 8.18 | 8.17 | 8.19 |
| Mean | |||||||
| Diab Ctl | 3.77 | 3.77 | 3.77 | 3.74 | 3.70 | 3.70 | 3.64 |
| StDev | |||||||
| Diab Ctl | 1.89 | 1.88 | 1.89 | 1.87 | 1.85 | 1.85 | 1.82 |
| SEM | |||||||
| Diab Txt | 8.84 | 8.91 | 8.87 | 8.75 | 8.37 | 8.08 | 7.80 |
| Mean | |||||||
| Diab Txt | 3.73 | 3.68 | 3.70 | 3.61 | 3.52 | 3.34 | 3.20 |
| StDev | |||||||
| Diab Txt | 1.32 | 1.30 | 1.31 | 1.28 | 1.24 | 1.18 | 1.13 |
| SEM | |||||||
| IR = insulin resistant, Diab = diabetic, Ctl = control, Txt = experimental, StDev = standard deviation, SEM = standard error of the mean | |||||||
| TABLE 3 |
| Insulin Resistant Monkey Mean Body Weight |
| Mean | ||||||||
| −7 wk | −4 wk | Baseline | 2 wk | 5 wk | 7 wk | 8 wk | ||
| IR Ctl Mean | 6.18 | 6.15 | 6.17 | 6.04 | 6.00 | 5.94 | 5.91 |
| IR Ctl StDev | 3.48 | 3.43 | 3.46 | 3.22 | 3.19 | 3.16 | 3.17 |
| IR Ctl SEM | 1.74 | 1.72 | 1.73 | 1.61 | 1.60 | 1.58 | 1.59 |
| IR Txt Mean | 5.25 | 5.32 | 5.29 | 5.26 | 5.04 | 4.91 | 4.71 |
| IR Txt | 3.17 | 3.22 | 3.19 | 3.42 | 3.40 | 3.34 | 3.20 |
| StDev | |||||||
| IR Txt SEM | 0.95 | 0.97 | 0.96 | 1.03 | 1.03 | 1.01 | 0.96 |
| IR = insulin resistant, Diab = diabetic, Ctl = control, Txt = treated, StDev = standard deviation, SEM = standard error of the mean | |||||||
Plasma leptin values, presented as baseline values vs. 8-week treatment, were determined by ELISA and are presented in Table 4. The effect of [5-Methoxy-2-methyl-4-(4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid on plasma leptin values in diabetic and insulin resistant obese cynomolgus monkeys is demonstrated by the lesser increase in plasma leptin values seen in the treated animals.
The amount of exogenous insulin the diabetic monkeys received to maintain proper glucose levels, the exogenous insulin requirement, is also presented in Table 4 as baseline values vs. 8-week treatment. By definition, insulin resistant monkeys are not yet diabetic and do not receive exogenous insulin. A reduction in exogenous insulin requirements is a measure of improved insulin sensitivity and glucose control.
| TABLE 4 |
| Leptin and Exogenous Insulin |
| Pre-Treatment | Post-Treatment | ||
| Exogenous Insulin | ||||
| Requirement (u/day) | ||||
| Diabetic Controls | 89 ± 71 | 101 ± 73 | ||
| Diabetic Treated | 66 ± 16 | 18 ± 4 | ||
| Leptin (ng/ml) | ||||
| Diabetic Controls | 6.55 ± 1.38 | 18.83 ± 0.24 | ||
| Diabetic Treated | 7.22 ± 0.93 | 10.89 ± 2.7 | ||
| Insulin Resistant Controls | 9.85 ± 0.45 | 13.60 ± 3.45 | ||
| Insulin Resistant Treated | 7.33 ± 1.16 | 5.73 ± 2.14 | ||
The compounds of the present invention can be administered alone or in combination with one or more therapeutic agents. These include, for example, other agents for the treatment, control, or prevention of hypercholesteremia, dyslipidemia, obesity, hyperglycemia, hypercholesteremia, atherosclerosis, hypertriglyceridemia, and hyperinsulinemia. The compounds of the present invention can be administered alone or in combination with one or more therapeutic agents for the supression of appetite and modulation of leptin.
The compounds are thus well suited to formulation for convenient administration to mammals for the prevention and treatment of such disorders.
The following examples further illustrate typical formulations provided by the invention.
| Formulation 1 |
| Ingredient | Amount |
| compound of Formulas I-V | 0.5 to 800 | mg |
| sodium benzoate | 5 | mg |
| isotonic saline | 1000 | mL |
The above ingredients are mixed and dissolved in the saline for IV administration to a patient.
| Formulation 2 |
| Ingredient | Amount |
| compound of Formulas I-V | 0.5 to 800 | mg |
| cellulose, microcrystalline | 400 | mg |
| stearic acid | 5 | mg |
| silicon dioxide | 10 | mg |
| sugar, confectionery | 50 | mg |
The ingredients are blended to uniformity and pressed into a tablet that is well suited for oral administration to a patient.
| Formulation 3 |
| Ingredient | Amount |
| compound of Formulas I-V | 0.5 to 800 | mg |
| starch, dried | 250 | mg |
| magnesium stearate | 10 | mg |
The ingredients are combined and milled to afford material suitable for filling hard gelatin capsules administered to a patient.
| Formulation 4 |
| Ingredient | Amount % wt./(total wt.) | ||
| compound of Formulas I-V | 1 to 50 | ||
| Polyethylene glycol 1000 | 32 to 75 | ||
| Polyethylene glycol 4000 | 16 to 25 | ||
The ingredients are combined via melting and then poured into molds containing 2.5 g total weight.
While embodiments of the invention have been illustrated and described, it is not intended that these embodiments illustrate and describe all possible forms of the invention. Rather, the words used in the specification are words of description rather than limitation, and it is understood that various changes may be made without departing from the spirit and scope of the invention.
Claims (24)
and pharmaceutically acceptable salts thereof, wherein:
X0 and S; X1 is absent; O, S, —CH2—, —CH2—CH2—, or —CH═CH—;
is absent;
R1 and R2 are selected from hydrogen, lower alkyl, lower alkoxy, haloalkyl, —O—(CH2)mCF3, halogen, nitro, cyano, —OH, —SH or —CF3;
R3 and R4 are selected from hydrogen, lower alkyl, lower alkoxy, haloalkyl, —O—(CH2)mCF3, halogen, nitro, cyano, —OH, —SH or —CF3;
provided that at least one of R1-R4 is H, lower alkyl, lower alkoxy, haloalkyl, —O—(CH2)mCF3, halogen, nitro, cyano, —OH, —SH or —CF3;
m is 0 to 5;
q is 0 to 1; and
r is 0 to 1.
2. The compund of claim 1 , wherein
X1 is absent or 0;
Ar1 and Ar2 are each independently unsubstituted or substituted phenyl or pyridinyl;
R1 is hydrogen;
R2 is lower alkyl, lower alkoxy, haloalkyl, —O—(CH2)mCF3, halogen, nitro, cyano, —OH, —SH or —CF3;
R3 is lower alkyl, lower alkoxy, haloalkyl, —O—(CH2)mCF3, halogen, nitro, cyano, —OH, —SH or —CF3;
R4 is hydrogen, lower alkyl, lower alkoxy, haloalkyl, —O—CH2)mCF3, halogen, nitro, cyano, —OH, —SH or —CF3;
m is 0 to 5;
q is 1; and
r is 0 to 1.
3. The compund of claim 1 , wherein
X1 is absent;
Ar1 is phenyl;
Ar2 is substituted phenyl or pyridinyl;
R1 is hydrogen;
R2 is lower alkyl, lower alkoxy, haloalkyl, —O—(CH2)mCF3, —OH or —SH;
R3 is lower alkyl or haloalkyl;
R4 is hydrogen;
m is 0 to 5;
q is 1; and
r is 0.
4. The compund of claim 1 , wherein
X1 is absent or 0;
Ar1 is phenyl;
Ar2 is substituted phenyl;
R1 is hydrogen;
R2 is lower alkoxy;
R3 is lower alkyl;
R4 is hydrogen;
q is 1; and
r is 0 to 1.
5. The compund of claim 1 , wherein
X1 is absent or 0;
Ar1 is phenyl;
Ar2 is substituted phenyl;
R1 is hydrogen;
R2 is methoxy;
R3 is methyl;
R4 is hydrogen;
q is 1; and
r is 0 to 1.
6. The compound of claim 1 , wherein q is 1.
7. The compound of claim 1 , wherein Ar1 is substituted or unsubstituted phenyl.
8. The compound of claim 1 , wherein Ar2 is 4-trifluoromethylphenyl.
9. The compound of claim 1 , wherein:
R1, R2, R3, and R4 are selected from hydrogen, alkyl, or alkoxy.
10. The compound of claim 1 , wherein:
R2 and R3 are hydrogen; and
R1 and R4 are alkyl or alkoxy.
11. The compound of claim 1 , wherein:
R1 is alkyl;
R2 and R3 are hydrogen; and
R4 is alkoxy.
12. The compound of claim 1 , wherein:
R1 is methyl, ethyl, isopropyl, n-propyl, t-butyl, n-butyl, or isobutyl;
R2 and R3 are hydrogen; and
R4 is methyoxy, ethoxy, isopropoxy, n-propoxy, t-butoxy, n-butoxy, or isobutoxy.
13. A compound selected from:
[4-(Biphenyl-4-ylmethylsulfanyl)-5-methoxy-2-methylphenoxy]-acetic acid;
[5-Methoxy-2-methyl-4-(4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid;
[4-(2′,4′-Dichloro-biphenyl-4-ylmethylsulfanyl)-5-methoxy-2-methyl-phenoxy]-acetic acid;
[5-Methoxy-2-methyl-4-(3′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid;
[4-(4′-Fluoro-biphenyl-4-ylmethylsulfanyl)-5-methoxy-2-methyl-phenoxy]-acetic acid;
[5-Methoxy-2-methyl-4-[4-(4-trifluoromethyl-benzyloxy)-benzylsulfanyl]-phenoxy]-acetic acid;
[5-Methoxy-2-methyl-4-(3′-trifluoromethoxy-biphenyl-3-ylmethylsulfanyl)-phenoxy]-acetic acid;
{{-Methoxy-2-methyl-4-[4-(5-trifluoromethyl-pyridin-2-yl)-benzylsulfanyl]-phenoxy}-acetic acid;
{5-Methoxy-2-methyl-4-[6-(4-trifluoromethyl-phenyl)-pyridin-3-ylmethylsulfanyl]-phenoxy}-acetic acid;
[3-Methoxy-4-(4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid;
(4-{4-[2-(3-Fluoro-phenyl)-vinyl]benzylsulfanyl}-5-methoxy-2-methyl-phenoxy)-acetic acid;
[5-Methoxy-2-methyl-4-(3-methyl-4′-trifluoromethyl-biphenyl-4-ylmethylsulfanyl)-phenoxy]-acetic acid;
[5-Methoxy-2-methyl-4-(4′-trifluoromethyl-biphenyl-3-ylmethylsulfanyl)-phenoxy]-acetic acid;
and pharmaceutically acceptable salts thereof.
14. A pharmaceutical composition comprising a compound of claim 1 or a pharmaceutically acceptable salt thereof admixed with a carrier, diluent, or excipient.
15. A method of treating or controlling non-insulin dependent diabetes mellitus in a mammal comprising administering to the mammal in need thereof a therapeutically effective amount of a compound of claim 1 formula (I):
or a pharmaceutically acceptable salt thereof wherein:
X
0
is S; X
1
is absent, O, S, —CH
2
—, —CH
2
—CH
2
—, or —CH═CH;
is absent; R 1 and R 2 are selected from hydrogen, lower alkyl, lower alkoxy, haloalkyl,—O—(CH 2)m CF 3 , halogen, nitro, cyano, —OH, —SH or —CF 3;
R 3 and R 4 are selected from hydrogen, lower alkyl, lower alkoxy, haloalkyl,—O—(CH 2)m CF 3 , halogen, nitro, cyano, —OH, —SH or —CF 3 , provided that at least one of R 1-R 4 is H, lower alkyl, lower alkoxy, haloalkyl,—O—(CH 2)m CF 3 , halogen, nitro, cyano, —OH, —SH or —CF 3 ; m is 0 to 5;
q is 0 to 1; and
r is 0 to 1 .
16. A method of treating or controlling obesity in a mammal comprising adminsitering to the mammal in need thereof a therapeutically effective amount of a compound of claim 1 formula (I):
or a pharmaceutically acceptable salt thereof, wherein:
X
0
is S; X
1
is absent, O, S, —CH
2
—, —CH
2
—CH
2
—, or —CH═CH;
is absent; R 1 and R 2 are selected from hydrogen, lower alkyl, lower alkoxy, haloalkyl,—O—(CH 2)m CF 3 , halogen, nitro, cyano, —OH, —SH or —CF 3;
R 3 and R 4 are selected from hydrogen, lower alkyl, lower alkoxy, haloalkyl,—O—(CH 2)m CF 3 , halogen, nitro, cyano, —OH, —SH or —CF 3 , provided that at least one of R 1-R 4 is H, lower alkyl, lower alkoxy, haloalkyl,—O—(CH 2)m CF 3 , halogen, nitro, cyano, —OH, —SH or —CF 3 ; m is 0 to 5;
q is 0 to 1; and
r is 0 to 1 .
17. A method of treating or controlling eating disorders in a mammal comprising administering to the mammal in need thereof a therapeutically effective amount of a compound of claim 1 .
18. A method of supressing appetite in a mammal comprising administering to the mammal in need thereof a therapeutically effective amount of a compound of claim 1 .
19. A method of modulating leptin levels in a mammal comprising administering to the mammal in need thereof a therapeutically effective amount of a compound of claim 1 .
20. A method of treating or controlling hyperglycemia in a mammal comprising administering to the mammal in need thereof a therapeutically effective amount of a compound of claim 1 .
21. A method of treating or controlling hyperinsulinemia in a mammal comprising administering to the mammal in need thereof a therapeutically effective amount of a compound of claim 1 .
22. A method of treating a patient exhibiting glucose disorders associated with circulating glucocorticoids, growth hormone, catecholamines, glucagon, or parathyroid hormone, comprising administering to the patient a therapeutically effective amount of a compound of claim 1 .
23. A method of treating non-insuline dependent diabetes mellitus in a patient comprising administering to the patient in need of treatment a composition according to claim 14 .
24. A method of treating metabolic syndrome in a patient comprising administering to the patient in need of treatment a composition according to claim 14 comprising a compound of formula (I):
or a pharmaceutically acceptable salt thereof, wherein:
X
0
is S; X
1
is absent, O, S, —CH
2
—, —CH
2
—CH
2
—, or —CH═CH;
is absent; R 1 and R 2 are selected from hydrogen, lower alkyl, lower alkoxy, haloalkyl,—O—(CH 2)m CF 3 , halogen, nitro, cyano, —OH, —SH or —CF 3;
R 3 and R 4 are selected from hydrogen, lower alkyl, lower alkoxy, haloalkyl,—O—(CH 2)m CF 3 , halogen, nitro, cyano, —OH, —SH or —CF 3 , provided that at least one of R 1-R 4 is H, lower alkyl, lower alkoxy, haloalkyl,—O—(CH 2)m CF 3 , halogen, nitro, cyano, —OH, —SH or —CF 3 ; m is 0 to 5;
q is 0 to 1; and
r is 0 to 1;
and a pharmacuetically acceptable carrier, diluent or vehicle.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US11/288,022 USRE39916E1 (en) | 2002-04-05 | 2005-11-28 | Compounds that modulate PPAR activity and methods for their preparation |
Applications Claiming Priority (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US37050802P | 2002-04-05 | 2002-04-05 | |
| US38602602P | 2002-06-05 | 2002-06-05 | |
| US10/347,749 US6875780B2 (en) | 2002-04-05 | 2003-01-22 | Compounds that modulate PPAR activity and methods for their preparation |
| US10/979,617 US6939875B2 (en) | 2002-04-05 | 2004-11-02 | Compounds that modulate PPAR activity and methods for their preparation |
| US11/288,022 USRE39916E1 (en) | 2002-04-05 | 2005-11-28 | Compounds that modulate PPAR activity and methods for their preparation |
Related Parent Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US10/979,617 Reissue US6939875B2 (en) | 2002-04-05 | 2004-11-02 | Compounds that modulate PPAR activity and methods for their preparation |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| USRE39916E1 true USRE39916E1 (en) | 2007-11-06 |
Family
ID=28794388
Family Applications (4)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US10/347,749 Expired - Fee Related US6875780B2 (en) | 2002-04-05 | 2003-01-22 | Compounds that modulate PPAR activity and methods for their preparation |
| US10/979,617 Ceased US6939875B2 (en) | 2002-04-05 | 2004-11-02 | Compounds that modulate PPAR activity and methods for their preparation |
| US10/979,629 Expired - Fee Related US6964983B2 (en) | 2002-04-05 | 2004-11-02 | Compounds that modulate PPAR activity and methods for their preparation |
| US11/288,022 Expired - Fee Related USRE39916E1 (en) | 2002-04-05 | 2005-11-28 | Compounds that modulate PPAR activity and methods for their preparation |
Family Applications Before (3)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US10/347,749 Expired - Fee Related US6875780B2 (en) | 2002-04-05 | 2003-01-22 | Compounds that modulate PPAR activity and methods for their preparation |
| US10/979,617 Ceased US6939875B2 (en) | 2002-04-05 | 2004-11-02 | Compounds that modulate PPAR activity and methods for their preparation |
| US10/979,629 Expired - Fee Related US6964983B2 (en) | 2002-04-05 | 2004-11-02 | Compounds that modulate PPAR activity and methods for their preparation |
Country Status (35)
| Country | Link |
|---|---|
| US (4) | US6875780B2 (en) |
| EP (1) | EP1494989A2 (en) |
| JP (2) | JP3816922B2 (en) |
| KR (1) | KR100687166B1 (en) |
| CN (1) | CN1649820A (en) |
| AP (1) | AP1772A (en) |
| AR (1) | AR039239A1 (en) |
| AU (1) | AU2003212578A1 (en) |
| BR (1) | BR0309169A (en) |
| CA (1) | CA2481246C (en) |
| CO (1) | CO5640080A2 (en) |
| DO (1) | DOP2003000620A (en) |
| EA (2) | EA200700969A1 (en) |
| GE (1) | GEP20074244B (en) |
| GT (3) | GT200300075A (en) |
| HN (1) | HN2003000113A (en) |
| HR (1) | HRP20040916A2 (en) |
| IL (1) | IL164015A0 (en) |
| IS (1) | IS7429A (en) |
| MA (1) | MA27189A1 (en) |
| MX (1) | MXPA04009727A (en) |
| MY (1) | MY134199A (en) |
| NI (1) | NI200300042A (en) |
| NO (1) | NO20044795L (en) |
| NZ (1) | NZ554477A (en) |
| OA (1) | OA12859A (en) |
| PA (1) | PA8570701A1 (en) |
| PE (1) | PE20040307A1 (en) |
| PL (1) | PL373651A1 (en) |
| RS (1) | RS82904A (en) |
| TN (1) | TNSN04191A1 (en) |
| TW (1) | TWI249522B (en) |
| UA (1) | UA76838C2 (en) |
| UY (1) | UY27749A1 (en) |
| WO (1) | WO2003084916A2 (en) |
Families Citing this family (84)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7517884B2 (en) * | 1998-03-30 | 2009-04-14 | Kalypsys Inc. | Sulfonyl-substituted bicyclic compounds as modulators of PPAR |
| GB0029974D0 (en) * | 2000-12-08 | 2001-01-24 | Glaxo Group Ltd | Chemical compounds |
| AU2002336532C1 (en) | 2001-09-14 | 2008-10-16 | Amgen, Inc | Linked biaryl compounds |
| US6833380B2 (en) | 2002-03-07 | 2004-12-21 | Warner-Lambert Company, Llc | Compounds that modulate PPAR activity and methods of preparation |
| AU2003243947A1 (en) * | 2002-06-24 | 2004-01-06 | Sugatsune Kogyo Co., Ltd. | Umbrella storage device and umbrella holding lock |
| US7396850B2 (en) * | 2003-01-06 | 2008-07-08 | Eli Lilly And Company | Pyrazole derivative as PPAR modulator |
| US7244763B2 (en) * | 2003-04-17 | 2007-07-17 | Warner Lambert Company Llc | Compounds that modulate PPAR activity and methods of preparation |
| US7820837B2 (en) * | 2003-05-30 | 2010-10-26 | Takeda Pharmaceutical Company Limited | Condensed ring compound |
| DE602004022179D1 (en) | 2003-09-19 | 2009-09-03 | Janssen Pharmaceutica Nv | 4 - ((PHENOXYALKYL) THIO) -PHENOXY ACETIC ACIDS AND ANALOGUES |
| BRPI0414554A (en) * | 2003-10-28 | 2006-11-07 | Reddys Lab Ltd Dr | compounds and their uses in medicine: process for the preparation of these and pharmaceutical compositions containing them |
| CN1897939A (en) * | 2003-10-31 | 2007-01-17 | 詹森药业有限公司 | Phenoxyacetic acids derivatives useful as peroxisome proliferator-activated receptor (ppar) dual agonists |
| CA2543239A1 (en) | 2003-11-05 | 2005-06-02 | F. Hoffmann-La Roche Ag | Heteroaryl derivatives as ppar activators |
| AU2004291262C1 (en) * | 2003-11-05 | 2011-08-11 | F. Hoffmann-La Roche Ag | Phenyl derivatives as PPAR agonists |
| RU2367654C2 (en) * | 2003-11-05 | 2009-09-20 | Ф.Хоффманн-Ля Рош Аг | Benzannealed compounds as ppar activators |
| WO2005054213A1 (en) * | 2003-12-02 | 2005-06-16 | Shionogi & Co., Ltd. | Isoxazole derivative having agonistic activity against peroxisome proliferator-activated receptor |
| US7507832B2 (en) | 2003-12-22 | 2009-03-24 | Eli Lilly And Company | Triazole PPAR modulators |
| EP1706386A1 (en) | 2003-12-22 | 2006-10-04 | Eli Lilly And Company | Bicyclic derivatives as ppar modulators |
| TW200523247A (en) * | 2003-12-25 | 2005-07-16 | Takeda Pharmaceutical | 3-(4-benzyloxyphenyl) propanoic acid derivatives |
| GB0403148D0 (en) * | 2004-02-12 | 2004-03-17 | Smithkline Beecham Corp | Chemical compounds |
| ES2433466T3 (en) * | 2004-02-27 | 2013-12-11 | Amgen, Inc | Compounds, pharmaceutical compositions and methods for use in the treatment of metabolic disorders |
| ES2314630T3 (en) * | 2004-03-09 | 2009-03-16 | F. Hoffmann-La Roche Ag | DERIVATIVES OF PIRAZOLIL-INDOLILO IN QUALITY OF ACTIVITIES. |
| US7119214B2 (en) * | 2004-04-13 | 2006-10-10 | Cephalon France | Thio-substituted tricyclic and bicyclic aromatic methanesulfinyl derivatives |
| ES2347578T3 (en) * | 2004-05-05 | 2010-11-02 | High Point Pharmaceuticals, Llc | DERIVATIVES OF THE PHENOXYACETIC ACID AS PPAR AGONISTS. |
| ES2352085T3 (en) * | 2004-05-05 | 2011-02-15 | High Point Pharmaceuticals, Llc | NEW COMPOUNDS, THEIR PREPARATION AND USE. |
| JP4981662B2 (en) * | 2004-05-05 | 2012-07-25 | ハイ・ポイント・ファーマスーティカルズ、エルエルシー | New compounds, their preparation and use |
| KR20070026503A (en) | 2004-05-14 | 2007-03-08 | 아이알엠 엘엘씨 | Compounds and compositions as ppar modulators |
| CN1984894A (en) * | 2004-05-14 | 2007-06-20 | Irm责任有限公司 | Compounds and compositions as PPAR modulators |
| AU2005247172A1 (en) * | 2004-05-25 | 2005-12-08 | Pfizer Products Inc. | Use of PPAR agonists to treat ruminants |
| AU2005247473A1 (en) * | 2004-05-25 | 2005-12-08 | Metabolex, Inc. | Substituted triazoles as modulators of PPAR and methods of their preparation |
| US20060014785A1 (en) * | 2004-05-25 | 2006-01-19 | Metabolex, Inc. | Bicyclic, substituted triazoles as modulators of ppar and methods of their preparation |
| RU2384565C2 (en) * | 2004-07-01 | 2010-03-20 | Ф.Хоффманн-Ля Рош Аг | Phenyl derivatives containing acetylene group |
| US7622491B2 (en) * | 2004-08-13 | 2009-11-24 | Metabolex Inc. | Modulators of PPAR and methods of their preparation |
| MY145712A (en) * | 2004-09-15 | 2012-03-30 | Janssen Pharmaceutica Nv | 4-((phenoxyalkyl)thio)-phenoxyacetic acids and analogs |
| US20070190079A1 (en) * | 2004-10-29 | 2007-08-16 | Kalypsys, Inc. | Methods for the selective modulation of ppar |
| DK1805158T3 (en) * | 2004-10-29 | 2018-08-06 | Kalypsys Inc | SULFONYL-SUBSTITUTED BICYCLIC COMPOUNDS AS MODULATORS OF PPAR |
| CA2590533C (en) * | 2004-11-23 | 2010-09-07 | Warner-Lambert Company Llc | 7-(2h-pyrazol-3-yl)-3,5-dihydroxy-heptanoic acid derivatives as hmg co-a reductase inhibitors for the treatment of lipidemia |
| US7465804B2 (en) * | 2005-05-20 | 2008-12-16 | Amgen Inc. | Compounds, pharmaceutical compositions and methods for their use in treating metabolic disorders |
| KR101333101B1 (en) * | 2005-06-30 | 2013-11-26 | 하이 포인트 파마슈티칼스, 엘엘씨 | Phenoxy acetic acids as ppar delta activators |
| CA2621406A1 (en) * | 2005-09-07 | 2007-03-15 | Plexxikon, Inc. | Pparactive compounds |
| JO3006B1 (en) * | 2005-09-14 | 2016-09-05 | Janssen Pharmaceutica Nv | Novel Lysine Salts of 4-((Phenoxy Alkyl)Thio)-Phenoxy Acetic Acid Derivatives |
| US7582803B2 (en) * | 2005-09-14 | 2009-09-01 | Amgen Inc. | Conformationally constrained 3-(4-hydroxy-phenyl)-substituted-propanoic acids useful for treating metabolic disorders |
| US7741317B2 (en) | 2005-10-21 | 2010-06-22 | Bristol-Myers Squibb Company | LXR modulators |
| CA2624383C (en) * | 2005-10-25 | 2014-03-11 | Kalypsys, Inc. | Salts of modulators of ppar and methods of treating metabolic disorders |
| US7888376B2 (en) | 2005-11-23 | 2011-02-15 | Bristol-Myers Squibb Company | Heterocyclic CETP inhibitors |
| EA201101085A1 (en) | 2005-12-22 | 2012-05-30 | ХАЙ ПОЙНТ ФАРМАСЬЮТИКАЛЗ, ЭлЭлСи | PHENOXYACIC ACIDS AS PPAR DELTA ACTIVATORS |
| CN101007798B (en) * | 2006-01-24 | 2011-01-26 | 中国人民解放军军事医学科学院毒物药物研究所 | Benzodioxole derivatives and their preparation method and medicinal uses |
| EP1999098A2 (en) * | 2006-03-09 | 2008-12-10 | High Point Pharmaceuticals, LLC | Compounds that modulate ppar activity, their preparation and use |
| US8383660B2 (en) | 2006-03-10 | 2013-02-26 | Pfizer Inc. | Dibenzyl amine compounds and derivatives |
| AU2007225208A1 (en) * | 2006-03-14 | 2007-09-20 | Amgen Inc. | Bicyclic carboxylic acid derivatives useful for treating metabolic disorders |
| UY30288A1 (en) * | 2006-04-18 | 2007-08-31 | Janssen Pharmaceutica Nv | DERIVATIVES OF BENZOAZEPIN-OXI-ACETIC ACID AS PPAR-DELTA AGONISTS USED TO INCREASE HDL-C. REDUCE LDL-C AND REDUCE CHOLESTEROL |
| US20070249519A1 (en) * | 2006-04-20 | 2007-10-25 | Kalypsys, Inc. | Methods for the upregulation of glut4 via modulation of ppar delta in adipose tissue and for the treatment of disease |
| AR060937A1 (en) * | 2006-05-16 | 2008-07-23 | Kalypsys Inc | SUBSTITUTED BICYCLIC COMPOUNDS WITH SULFONYL AS PPAR MODULATORS |
| CA2662305C (en) * | 2006-09-07 | 2012-04-17 | Amgen Inc. | Heterocyclic gpr40 modulators |
| EP2061760A1 (en) * | 2006-09-07 | 2009-05-27 | Amgen, Inc | Benzo-fused compounds for use in treating metabolic disorders |
| WO2008070496A2 (en) | 2006-12-01 | 2008-06-12 | Bristol-Myers Squibb Company | N- ( (3-benzyl) -2, 2- (bis-phenyl) -propan-1-amine derivatives as cetp inhibitors for the treatment of atherosclerosis and cardiovascular diseases |
| US8519145B2 (en) | 2006-12-02 | 2013-08-27 | Seoul National University Industry Foundation | Aryl compounds as PPAR ligands and their use |
| WO2008091863A1 (en) | 2007-01-23 | 2008-07-31 | Kalypsys, Inc. | Sulfonyl-substituted bicyclic compounds as ppar modulators for the treatment of non-alcoholic steatohepatitis |
| EP2129667A2 (en) * | 2007-02-23 | 2009-12-09 | Eli Lilly And Company | Peroxisome proliferator activated receptor modulators |
| US8329722B2 (en) * | 2007-03-13 | 2012-12-11 | Merck Sharp & Dohme Corp. | Inhibitors of janus kinases and/or 3-phosphoinositide-dependent protein kinase-1 |
| AU2008241490B2 (en) * | 2007-04-16 | 2011-07-21 | Amgen Inc. | Substituted biphenyl phenoxy-, thiophenyl- and aminophenylpropanoic acid GPR40 modulators |
| US8759355B2 (en) * | 2007-07-25 | 2014-06-24 | Institute Of Pharmacology And Toxicology Academy Of Military Medical Sciences P.L.A. | Aryl pyrimidine derivatives, preparation methods and pharmaceutical uses thereof |
| CN102083783A (en) | 2007-10-10 | 2011-06-01 | 安姆根有限公司 | Substituted biphenyl GPR40 modulators |
| CA2708391A1 (en) * | 2007-12-13 | 2009-06-25 | Sri International | Ppar-delta ligands and methods of their use |
| EP2260017A1 (en) * | 2008-03-06 | 2010-12-15 | Amgen, Inc | Conformationally constrained carboxylic acid derivatives useful for treating metabolic disorders |
| WO2009153496A2 (en) | 2008-05-26 | 2009-12-23 | Genfit | Ppar agonist compounds, preparation and uses |
| WO2010018547A1 (en) * | 2008-08-13 | 2010-02-18 | Pfizer Inc. | Aminoquinoline compounds |
| EP2358656B1 (en) * | 2008-10-15 | 2014-01-01 | Amgen, Inc | Spirocyclic gpr40 modulators |
| WO2012178142A1 (en) * | 2011-06-23 | 2012-12-27 | Metabolic Solutions Development Company, Llc | Ppar-sparing compounds and combinations fort the treatment of diabetes and other metabolic diseases |
| CN104487542B (en) * | 2012-07-06 | 2016-10-12 | 默克专利股份有限公司 | Double mesomorphic compounds and mesogenic media |
| WO2014170786A1 (en) | 2013-04-17 | 2014-10-23 | Pfizer Inc. | N-piperidin-3-ylbenzamide derivatives for treating cardiovascular diseases |
| EP3756661A1 (en) | 2013-09-09 | 2020-12-30 | vTv Therapeutics LLC | Use of a ppar-delta agonist for treating muscle atrophy |
| WO2016055901A1 (en) | 2014-10-08 | 2016-04-14 | Pfizer Inc. | Substituted amide compounds |
| CN106831437B (en) * | 2016-02-03 | 2019-06-21 | 华中师范大学 | Ester type compound and its preparation method and application containing nitroethenyl group |
| UY37806A (en) | 2017-07-11 | 2020-01-31 | Vertex Pharma | CARBOXAMIDES AS MODULATORS OF SODIUM CHANNELS |
| BR112020005489A2 (en) | 2017-09-22 | 2020-09-24 | Jubilant Epipad Llc, | compound of formula (i), compound of formula (ii), compound of formula (iii), process of preparing compounds of formula (i), process of preparing compounds of formula (ii), process of preparing compounds of formula (iii), pharmaceutical composition, method for inhibiting one or more pad families in a cell, method for treating a condition mediated by one or more pads, use of the compound, method for treating and / or preventing a condition mediated by one or more more disorders of the pad family, method for treating rheumatoid arthritis and cancer treatment method |
| ES2941512T3 (en) | 2017-10-18 | 2023-05-23 | Jubilant Epipad LLC | Imidazo-pyridine compounds as PAD inhibitors |
| CN111386265A (en) | 2017-11-06 | 2020-07-07 | 朱比连特普罗德尔有限责任公司 | Pyrimidine derivatives as inhibitors of PD1/PD-L1 activation |
| KR20200092346A (en) | 2017-11-24 | 2020-08-03 | 주빌런트 에피스크라이브 엘엘씨 | Heterocyclic compounds as PRMT5 inhibitors |
| SG11202008950PA (en) | 2018-03-13 | 2020-10-29 | Jubilant Prodel LLC | Bicyclic compounds as inhibitors of pd1/pd-l1 interaction/activation |
| CN113574055A (en) | 2019-01-18 | 2021-10-29 | 阿斯利康(瑞典)有限公司 | PCSK9 inhibitors and methods of use thereof |
| BR112021026384A2 (en) * | 2019-06-28 | 2022-02-08 | Sumitomo Chemical Co | Acrylate derivative, intermediate compound for its use and production |
| CN112824394A (en) * | 2019-11-21 | 2021-05-21 | 中国科学院上海药物研究所 | PPARs-FXR multi-target small molecule agonist and preparation method and application thereof |
| CA3185909A1 (en) | 2020-07-22 | 2022-01-27 | Reneo Pharmaceuticals, Inc. | Crystalline ppar-delta agonist |
| WO2023147309A1 (en) | 2022-01-25 | 2023-08-03 | Reneo Pharmaceuticals, Inc. | Use of ppar-delta agonists in the treatment of disease |
Citations (20)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE3526235A1 (en) | 1984-11-16 | 1986-05-22 | Bayer Ag, 5090 Leverkusen | LIQUID CRYSTAL COMPOSITIONS |
| DE4002374A1 (en) | 1990-01-27 | 1991-08-01 | Merck Patent Gmbh | New partly fluorinated uncondensed poly:cyclic cpds. - useful in LCD medium, having low viscosity and wide mesophase range |
| EP0578054A1 (en) | 1992-07-09 | 1994-01-12 | Hoechst Aktiengesellschaft | Meta-substituted six membered aromatic rings for use in liquid crystal mixtures |
| EP0625513A1 (en) | 1993-05-17 | 1994-11-23 | Canon Kabushiki Kaisha | Mesomorphic compound, liquid crystal composition containing it, liquid crystal device using the composition, display apparatus and method |
| WO1996004228A1 (en) | 1994-08-04 | 1996-02-15 | Sumitomo Chemical Company, Limited | Dihalopropene compounds, insecticidal/acaricidal agents containing same, and intermediates for their production |
| JPH09194418A (en) | 1996-01-18 | 1997-07-29 | Sumitomo Chem Co Ltd | Dihalopropene compound, its use and its production intermediate |
| WO1997028137A1 (en) | 1996-02-02 | 1997-08-07 | Merck & Co., Inc. | Heterocyclic derivatives as antidiabetic and antiobesity agents |
| WO1997028149A1 (en) | 1996-02-02 | 1997-08-07 | Merck & Co., Inc. | Method for raising hdl cholesterol levels |
| EP0930299A1 (en) | 1996-08-19 | 1999-07-21 | Japan Tobacco Inc. | Propionic acid derivatives and applications thereof |
| WO1999046232A1 (en) | 1998-03-10 | 1999-09-16 | Ono Pharmaceutical Co., Ltd. | Carboxylic acid derivatives and drugs containing the same as the active ingredient |
| WO2001000603A1 (en) | 1999-06-25 | 2001-01-04 | Glaxo Group Limited | Thiazole and oxazole derivatives and their pharmaceutical use |
| WO2001016120A1 (en) | 1999-08-27 | 2001-03-08 | Eli Lilly And Company | Biaryl-oxa(thia)zole derivatives and their use as ppars modulators |
| WO2002050048A1 (en) | 2000-12-20 | 2002-06-27 | Glaxo Group Limited | Thia- and oxazoles and their use as ppars activators |
| WO2002062774A1 (en) | 2000-12-20 | 2002-08-15 | Glaxo Group Limited | Thiazole derivatives for treating ppar related disorders |
| WO2002092590A1 (en) | 2001-05-11 | 2002-11-21 | Glaxo Group Limited | Furan and thiophene derivatives that activate human peroxisome proliferator activated receptors |
| WO2002100403A1 (en) | 2001-06-07 | 2002-12-19 | Eli Lilly And Company | Modulators of peroxisome proliferator activated receptors (ppar) |
| WO2003024395A2 (en) | 2001-09-14 | 2003-03-27 | Tularik Inc. | Linked biaryl compounds |
| US20030207915A1 (en) * | 2002-03-07 | 2003-11-06 | Xue-Min Cheng | Compounds that modulate PPAR activity and methods of preparation |
| US20030207916A1 (en) * | 2002-03-07 | 2003-11-06 | Xue-Min Cheng | Compounds that modulate PPAR activity and methods of preparation |
| US20030207924A1 (en) * | 2002-03-07 | 2003-11-06 | Xue-Min Cheng | Compounds that modulate PPAR activity and methods of preparation |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2001032621A1 (en) * | 1999-10-29 | 2001-05-10 | Wakunaga Pharmaceutical Co., Ltd. | Novel indole derivatives and drugs containing the same as the active ingredient |
-
2003
- 2003-01-22 US US10/347,749 patent/US6875780B2/en not_active Expired - Fee Related
- 2003-03-06 NI NI200300042A patent/NI200300042A/en unknown
- 2003-03-24 EP EP03708403A patent/EP1494989A2/en not_active Withdrawn
- 2003-03-24 JP JP2003582115A patent/JP3816922B2/en not_active Expired - Fee Related
- 2003-03-24 OA OA1200400261A patent/OA12859A/en unknown
- 2003-03-24 IL IL16401503A patent/IL164015A0/en unknown
- 2003-03-24 MX MXPA04009727A patent/MXPA04009727A/en active IP Right Grant
- 2003-03-24 CA CA002481246A patent/CA2481246C/en not_active Expired - Fee Related
- 2003-03-24 AP APAP/P/2004/003135A patent/AP1772A/en active
- 2003-03-24 PL PL03373651A patent/PL373651A1/en unknown
- 2003-03-24 DO DO2003000620A patent/DOP2003000620A/en unknown
- 2003-03-24 UA UA20041008021A patent/UA76838C2/en unknown
- 2003-03-24 KR KR1020047015763A patent/KR100687166B1/en not_active IP Right Cessation
- 2003-03-24 WO PCT/IB2003/001121 patent/WO2003084916A2/en active Application Filing
- 2003-03-24 AU AU2003212578A patent/AU2003212578A1/en not_active Abandoned
- 2003-03-24 RS YUP-829/04A patent/RS82904A/en unknown
- 2003-03-24 GE GEAP8431A patent/GEP20074244B/en unknown
- 2003-03-24 EA EA200700969A patent/EA200700969A1/en unknown
- 2003-03-24 NZ NZ554477A patent/NZ554477A/en unknown
- 2003-03-24 CN CNA038097915A patent/CN1649820A/en active Pending
- 2003-03-24 BR BR0309169-4A patent/BR0309169A/en not_active IP Right Cessation
- 2003-03-24 EA EA200401153A patent/EA009518B1/en not_active IP Right Cessation
- 2003-04-01 GT GT200300075A patent/GT200300075A/en unknown
- 2003-04-01 GT GT200300075BL patent/GT200300075BA/en unknown
- 2003-04-01 PE PE2003000327A patent/PE20040307A1/en not_active Application Discontinuation
- 2003-04-01 GT GT200300075AK patent/GT200300075AA/en unknown
- 2003-04-02 MY MYPI20031235A patent/MY134199A/en unknown
- 2003-04-03 AR ARP030101164A patent/AR039239A1/en unknown
- 2003-04-03 UY UY27749A patent/UY27749A1/en not_active Application Discontinuation
- 2003-04-04 TW TW092107732A patent/TWI249522B/en not_active IP Right Cessation
- 2003-04-04 PA PA20038570701A patent/PA8570701A1/en unknown
- 2003-04-04 HN HN2003000113A patent/HN2003000113A/en unknown
-
2004
- 2004-08-30 IS IS7429A patent/IS7429A/en unknown
- 2004-09-17 CO CO04092873A patent/CO5640080A2/en not_active Application Discontinuation
- 2004-09-28 MA MA27877A patent/MA27189A1/en unknown
- 2004-10-04 TN TNP2004000191A patent/TNSN04191A1/en unknown
- 2004-10-05 HR HRP20040916 patent/HRP20040916A2/en not_active Application Discontinuation
- 2004-11-02 US US10/979,617 patent/US6939875B2/en not_active Ceased
- 2004-11-02 US US10/979,629 patent/US6964983B2/en not_active Expired - Fee Related
- 2004-11-04 NO NO20044795A patent/NO20044795L/en not_active Application Discontinuation
-
2005
- 2005-11-28 US US11/288,022 patent/USRE39916E1/en not_active Expired - Fee Related
- 2005-12-14 JP JP2005360431A patent/JP2006151985A/en not_active Withdrawn
Patent Citations (22)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE3526235A1 (en) | 1984-11-16 | 1986-05-22 | Bayer Ag, 5090 Leverkusen | LIQUID CRYSTAL COMPOSITIONS |
| DE4002374A1 (en) | 1990-01-27 | 1991-08-01 | Merck Patent Gmbh | New partly fluorinated uncondensed poly:cyclic cpds. - useful in LCD medium, having low viscosity and wide mesophase range |
| EP0578054A1 (en) | 1992-07-09 | 1994-01-12 | Hoechst Aktiengesellschaft | Meta-substituted six membered aromatic rings for use in liquid crystal mixtures |
| US5447656A (en) * | 1992-07-09 | 1995-09-05 | Hoechst Aktiengesellschaft | Meta-substituted aromatic compounds having six-membered rings, for use in liquid-crystal mixtures |
| EP0625513A1 (en) | 1993-05-17 | 1994-11-23 | Canon Kabushiki Kaisha | Mesomorphic compound, liquid crystal composition containing it, liquid crystal device using the composition, display apparatus and method |
| WO1996004228A1 (en) | 1994-08-04 | 1996-02-15 | Sumitomo Chemical Company, Limited | Dihalopropene compounds, insecticidal/acaricidal agents containing same, and intermediates for their production |
| JPH09194418A (en) | 1996-01-18 | 1997-07-29 | Sumitomo Chem Co Ltd | Dihalopropene compound, its use and its production intermediate |
| WO1997028137A1 (en) | 1996-02-02 | 1997-08-07 | Merck & Co., Inc. | Heterocyclic derivatives as antidiabetic and antiobesity agents |
| WO1997028149A1 (en) | 1996-02-02 | 1997-08-07 | Merck & Co., Inc. | Method for raising hdl cholesterol levels |
| EP0930299A1 (en) | 1996-08-19 | 1999-07-21 | Japan Tobacco Inc. | Propionic acid derivatives and applications thereof |
| WO1999046232A1 (en) | 1998-03-10 | 1999-09-16 | Ono Pharmaceutical Co., Ltd. | Carboxylic acid derivatives and drugs containing the same as the active ingredient |
| US6506757B1 (en) * | 1998-03-10 | 2003-01-14 | Ono Pharmaceutical Co., Ltd. | Carboxylic acid derivatives and drugs containing the same as the active ingredient |
| WO2001000603A1 (en) | 1999-06-25 | 2001-01-04 | Glaxo Group Limited | Thiazole and oxazole derivatives and their pharmaceutical use |
| WO2001016120A1 (en) | 1999-08-27 | 2001-03-08 | Eli Lilly And Company | Biaryl-oxa(thia)zole derivatives and their use as ppars modulators |
| WO2002050048A1 (en) | 2000-12-20 | 2002-06-27 | Glaxo Group Limited | Thia- and oxazoles and their use as ppars activators |
| WO2002062774A1 (en) | 2000-12-20 | 2002-08-15 | Glaxo Group Limited | Thiazole derivatives for treating ppar related disorders |
| WO2002092590A1 (en) | 2001-05-11 | 2002-11-21 | Glaxo Group Limited | Furan and thiophene derivatives that activate human peroxisome proliferator activated receptors |
| WO2002100403A1 (en) | 2001-06-07 | 2002-12-19 | Eli Lilly And Company | Modulators of peroxisome proliferator activated receptors (ppar) |
| WO2003024395A2 (en) | 2001-09-14 | 2003-03-27 | Tularik Inc. | Linked biaryl compounds |
| US20030207915A1 (en) * | 2002-03-07 | 2003-11-06 | Xue-Min Cheng | Compounds that modulate PPAR activity and methods of preparation |
| US20030207916A1 (en) * | 2002-03-07 | 2003-11-06 | Xue-Min Cheng | Compounds that modulate PPAR activity and methods of preparation |
| US20030207924A1 (en) * | 2002-03-07 | 2003-11-06 | Xue-Min Cheng | Compounds that modulate PPAR activity and methods of preparation |
Non-Patent Citations (8)
| Title |
|---|
| Belleney, J. et al., Heterocyclic Chem., 1984;21:1431. |
| Nanteuil De G et al.: "5-Imidaol-yl-1H Benzimidazoles Inhibiteurs De L Interleukine-l: Une Nouevlle Voie Pour Le Traitement De L' Arthrose" XP001147539. |
| Nishimura, Koji: "Preparation of Indole Derivatives as Chymase Inhibitors and Drugs containing the same as the active Ingredient" XP002258311. |
| Rissanen et al., British Medical Journal,301:835-837 (1990). |
| S.M. Berge et al., "Pharmaceutical Salts", Journal of Pharmaceutical Sciences, 1977:66:1-19. |
| T. Gordon et al., The American Journal of Medicine, 1977;62:707-714. |
| U.S. Appl. No. 10/774,260, filed Feb. 6, 2004. |
| W.R. Oliver et al., PNAS, vol. 98, pp. 5306-5311, (2001). |
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| USRE39916E1 (en) | Compounds that modulate PPAR activity and methods for their preparation | |
| US7244763B2 (en) | Compounds that modulate PPAR activity and methods of preparation | |
| JP5225984B2 (en) | Compounds and methods for modulating FXR | |
| US20030207924A1 (en) | Compounds that modulate PPAR activity and methods of preparation | |
| US6867224B2 (en) | Compounds that modulate PPAR activity and methods of preparation | |
| NZ535603A (en) | Substituted phenylacetic acids | |
| JPWO2002046176A1 (en) | Activator of peroxisome proliferator-activated receptor | |
| EP1492528A1 (en) | Antidiabetic agents |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| FPAY | Fee payment |
Year of fee payment: 4 |
|
| REMI | Maintenance fee reminder mailed | ||
| LAPS | Lapse for failure to pay maintenance fees |