USRE38460E1 - Huperzine A derivatives, their preparation and their use - Google Patents
Huperzine A derivatives, their preparation and their use Download PDFInfo
- Publication number
- USRE38460E1 USRE38460E1 US09/799,127 US79912701A USRE38460E US RE38460 E1 USRE38460 E1 US RE38460E1 US 79912701 A US79912701 A US 79912701A US RE38460 E USRE38460 E US RE38460E
- Authority
- US
- United States
- Prior art keywords
- huperzine
- cholinesterase
- treatment
- administering
- patient
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
- ZRJBHWIHUMBLCN-YQEJDHNASA-N huperzine A Chemical class N1C(=O)C=CC2=C1C[C@H]1\C(=C/C)[C@]2(N)CC(C)=C1 ZRJBHWIHUMBLCN-YQEJDHNASA-N 0.000 title claims abstract description 24
- 238000002360 preparation method Methods 0.000 title description 6
- -1 hydroxy, amino Chemical group 0.000 claims abstract description 31
- 238000000034 method Methods 0.000 claims abstract description 31
- 125000000217 alkyl group Chemical group 0.000 claims abstract description 24
- 125000003545 alkoxy group Chemical group 0.000 claims abstract description 9
- 125000002541 furyl group Chemical group 0.000 claims abstract description 6
- 229910052736 halogen Inorganic materials 0.000 claims abstract description 6
- 150000002367 halogens Chemical group 0.000 claims abstract description 6
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 claims abstract description 6
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims abstract description 6
- 125000004029 hydroxymethyl group Chemical group [H]OC([H])([H])* 0.000 claims abstract description 5
- ZRJBHWIHUMBLCN-SEQYCRGISA-N Huperzine A Natural products N1C(=O)C=CC2=C1C[C@H]1/C(=C/C)[C@]2(N)CC(C)=C1 ZRJBHWIHUMBLCN-SEQYCRGISA-N 0.000 claims description 36
- ZRJBHWIHUMBLCN-UHFFFAOYSA-N Shuangyiping Natural products N1C(=O)C=CC2=C1CC1C(=CC)C2(N)CC(C)=C1 ZRJBHWIHUMBLCN-UHFFFAOYSA-N 0.000 claims description 36
- ZRJBHWIHUMBLCN-BMIGLBTASA-N rac-huperzine A Natural products N1C(=O)C=CC2=C1C[C@@H]1C(=CC)[C@@]2(N)CC(C)=C1 ZRJBHWIHUMBLCN-BMIGLBTASA-N 0.000 claims description 36
- 108090000322 Cholinesterases Proteins 0.000 claims description 27
- 230000000694 effects Effects 0.000 claims description 25
- 150000001875 compounds Chemical class 0.000 claims description 21
- 239000001257 hydrogen Substances 0.000 claims description 16
- 229910052739 hydrogen Inorganic materials 0.000 claims description 16
- 230000002401 inhibitory effect Effects 0.000 claims description 16
- 102100033639 Acetylcholinesterase Human genes 0.000 claims description 15
- 108010022752 Acetylcholinesterase Proteins 0.000 claims description 15
- 230000005764 inhibitory process Effects 0.000 claims description 10
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 claims description 10
- 206010027175 memory impairment Diseases 0.000 claims description 9
- 229940022698 acetylcholinesterase Drugs 0.000 claims description 8
- 150000002431 hydrogen Chemical group 0.000 claims description 8
- 230000001713 cholinergic effect Effects 0.000 claims description 6
- 108010053652 Butyrylcholinesterase Proteins 0.000 claims description 5
- 102100032404 Cholinesterase Human genes 0.000 claims description 4
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims description 4
- 239000002904 solvent Substances 0.000 claims description 4
- 102000003914 Cholinesterases Human genes 0.000 claims 26
- 229940048961 cholinesterase Drugs 0.000 claims 26
- 206010028372 Muscular weakness Diseases 0.000 claims 1
- 230000006735 deficit Effects 0.000 claims 1
- 125000002485 formyl group Chemical class [H]C(*)=O 0.000 claims 1
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims 1
- 230000036473 myasthenia Effects 0.000 claims 1
- 125000004453 alkoxycarbonyl group Chemical group 0.000 abstract description 6
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 abstract description 6
- 239000000544 cholinesterase inhibitor Substances 0.000 abstract description 6
- 125000003236 benzoyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C(*)=O 0.000 abstract description 3
- 125000004925 dihydropyridyl group Chemical group N1(CC=CC=C1)* 0.000 abstract description 3
- 229910052757 nitrogen Inorganic materials 0.000 abstract description 3
- 125000004433 nitrogen atom Chemical group N* 0.000 abstract description 3
- 125000005942 tetrahydropyridyl group Chemical group 0.000 abstract description 3
- 229940100578 Acetylcholinesterase inhibitor Drugs 0.000 abstract 1
- HEDRZPFGACZZDS-MICDWDOJSA-N Trichloro(2H)methane Chemical compound [2H]C(Cl)(Cl)Cl HEDRZPFGACZZDS-MICDWDOJSA-N 0.000 description 30
- 238000005160 1H NMR spectroscopy Methods 0.000 description 17
- 239000002504 physiological saline solution Substances 0.000 description 17
- 229930000680 A04AD01 - Scopolamine Natural products 0.000 description 14
- STECJAGHUSJQJN-GAUPFVANSA-N Hyoscine Natural products C1([C@H](CO)C(=O)OC2C[C@@H]3N([C@H](C2)[C@@H]2[C@H]3O2)C)=CC=CC=C1 STECJAGHUSJQJN-GAUPFVANSA-N 0.000 description 14
- STECJAGHUSJQJN-UHFFFAOYSA-N N-Methyl-scopolamin Natural products C1C(C2C3O2)N(C)C3CC1OC(=O)C(CO)C1=CC=CC=C1 STECJAGHUSJQJN-UHFFFAOYSA-N 0.000 description 14
- STECJAGHUSJQJN-FWXGHANASA-N scopolamine Chemical compound C1([C@@H](CO)C(=O)O[C@H]2C[C@@H]3N([C@H](C2)[C@@H]2[C@H]3O2)C)=CC=CC=C1 STECJAGHUSJQJN-FWXGHANASA-N 0.000 description 14
- 229960002646 scopolamine Drugs 0.000 description 14
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 12
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 12
- 102000004190 Enzymes Human genes 0.000 description 11
- 108090000790 Enzymes Proteins 0.000 description 11
- 229940088598 enzyme Drugs 0.000 description 11
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 10
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical compound C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 10
- 241000700159 Rattus Species 0.000 description 10
- 238000006243 chemical reaction Methods 0.000 description 10
- 239000000243 solution Substances 0.000 description 8
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 7
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 6
- 238000012360 testing method Methods 0.000 description 6
- 0 *n1c2c(ccc1=O)[C@@]1(C)CC(C)=CC(C2)/C1=C\C Chemical compound *n1c2c(ccc1=O)[C@@]1(C)CC(C)=CC(C2)/C1=C\C 0.000 description 5
- 241000699670 Mus sp. Species 0.000 description 5
- 239000000203 mixture Substances 0.000 description 5
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 5
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 5
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 4
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 4
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 4
- 238000004587 chromatography analysis Methods 0.000 description 4
- 239000012043 crude product Substances 0.000 description 4
- ASUTZQLVASHGKV-JDFRZJQESA-N galanthamine Chemical compound O1C(=C23)C(OC)=CC=C2CN(C)CC[C@]23[C@@H]1C[C@@H](O)C=C2 ASUTZQLVASHGKV-JDFRZJQESA-N 0.000 description 4
- 230000015654 memory Effects 0.000 description 4
- 238000000926 separation method Methods 0.000 description 4
- 238000004809 thin layer chromatography Methods 0.000 description 4
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 3
- ITQTTZVARXURQS-UHFFFAOYSA-N CC1=CC=CN=C1 Chemical compound CC1=CC=CN=C1 ITQTTZVARXURQS-UHFFFAOYSA-N 0.000 description 3
- 238000005481 NMR spectroscopy Methods 0.000 description 3
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 3
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 3
- 229930013930 alkaloid Natural products 0.000 description 3
- 150000003797 alkaloid derivatives Chemical class 0.000 description 3
- 239000002585 base Substances 0.000 description 3
- 238000001816 cooling Methods 0.000 description 3
- 238000000338 in vitro Methods 0.000 description 3
- 239000012046 mixed solvent Substances 0.000 description 3
- 239000003208 petroleum Substances 0.000 description 3
- 230000000144 pharmacologic effect Effects 0.000 description 3
- 239000000047 product Substances 0.000 description 3
- 210000002966 serum Anatomy 0.000 description 3
- 239000000741 silica gel Substances 0.000 description 3
- 229910002027 silica gel Inorganic materials 0.000 description 3
- 230000001988 toxicity Effects 0.000 description 3
- 231100000419 toxicity Toxicity 0.000 description 3
- HPOIPOPJGBKXIR-UHFFFAOYSA-N 3,6-dimethoxy-10-methyl-galantham-1-ene Natural products O1C(C(=CC=2)OC)=C3C=2CN(C)CCC23C1CC(OC)C=C2 HPOIPOPJGBKXIR-UHFFFAOYSA-N 0.000 description 2
- XKRFYHLGVUSROY-UHFFFAOYSA-N Argon Chemical compound [Ar] XKRFYHLGVUSROY-UHFFFAOYSA-N 0.000 description 2
- JEHZLMRXLITQHB-UHFFFAOYSA-N C.C.CC.CC.CC.CCc1ccccc1.Cc1cccc2ccccc12.c1ccncc1 Chemical compound C.C.CC.CC.CC.CCc1ccccc1.Cc1cccc2ccccc12.c1ccncc1 JEHZLMRXLITQHB-UHFFFAOYSA-N 0.000 description 2
- ZRJBHWIHUMBLCN-JYXJEATCSA-N C/C=C1\C2C=C(C)C[C@]1(N)C1=C(C2)NC(=O)C=C1 Chemical compound C/C=C1\C2C=C(C)C[C@]1(N)C1=C(C2)NC(=O)C=C1 ZRJBHWIHUMBLCN-JYXJEATCSA-N 0.000 description 2
- IUPGWMZFHKUORT-UHFFFAOYSA-N CC1=C(CO)C=NC=C1O Chemical compound CC1=C(CO)C=NC=C1O IUPGWMZFHKUORT-UHFFFAOYSA-N 0.000 description 2
- RHPUJHQBPORFGV-UHFFFAOYSA-N CC1=C(O)C=CC(Cl)=C1 Chemical compound CC1=C(O)C=CC(Cl)=C1 RHPUJHQBPORFGV-UHFFFAOYSA-N 0.000 description 2
- RFWWKVKUMUEILD-UHFFFAOYSA-N COC1=CC(Cl)=CC(C)=C1O Chemical compound COC1=CC(Cl)=CC(C)=C1O RFWWKVKUMUEILD-UHFFFAOYSA-N 0.000 description 2
- LPCKPBWOSNVCEL-UHFFFAOYSA-N Chlidanthine Natural products O1C(C(=CC=2)O)=C3C=2CN(C)CCC23C1CC(OC)C=C2 LPCKPBWOSNVCEL-UHFFFAOYSA-N 0.000 description 2
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 2
- IAZDPXIOMUYVGZ-WFGJKAKNSA-N Dimethyl sulfoxide Chemical compound [2H]C([2H])([2H])S(=O)C([2H])([2H])[2H] IAZDPXIOMUYVGZ-WFGJKAKNSA-N 0.000 description 2
- 241001090156 Huperzia serrata Species 0.000 description 2
- PIJVFDBKTWXHHD-UHFFFAOYSA-N Physostigmine Natural products C12=CC(OC(=O)NC)=CC=C2N(C)C2C1(C)CCN2C PIJVFDBKTWXHHD-UHFFFAOYSA-N 0.000 description 2
- 210000000170 cell membrane Anatomy 0.000 description 2
- 239000003153 chemical reaction reagent Substances 0.000 description 2
- 238000011161 development Methods 0.000 description 2
- 235000013305 food Nutrition 0.000 description 2
- 229960003980 galantamine Drugs 0.000 description 2
- BGLNUNCBNALFOZ-WMLDXEAASA-N galanthamine Natural products COc1ccc2CCCC[C@@]34C=CCC[C@@H]3Oc1c24 BGLNUNCBNALFOZ-WMLDXEAASA-N 0.000 description 2
- ASUTZQLVASHGKV-UHFFFAOYSA-N galanthamine hydrochloride Natural products O1C(=C23)C(OC)=CC=C2CN(C)CCC23C1CC(O)C=C2 ASUTZQLVASHGKV-UHFFFAOYSA-N 0.000 description 2
- IYVSXSLYJLAZAT-NOLJZWGESA-N lycoramine Natural products CN1CC[C@@]23CC[C@H](O)C[C@@H]2Oc4cccc(C1)c34 IYVSXSLYJLAZAT-NOLJZWGESA-N 0.000 description 2
- 229960001697 physostigmine Drugs 0.000 description 2
- PIJVFDBKTWXHHD-HIFRSBDPSA-N physostigmine Chemical compound C12=CC(OC(=O)NC)=CC=C2N(C)[C@@H]2[C@@]1(C)CCN2C PIJVFDBKTWXHHD-HIFRSBDPSA-N 0.000 description 2
- 238000010992 reflux Methods 0.000 description 2
- 238000011160 research Methods 0.000 description 2
- 230000002441 reversible effect Effects 0.000 description 2
- 239000007787 solid Substances 0.000 description 2
- 239000000758 substrate Substances 0.000 description 2
- 238000003786 synthesis reaction Methods 0.000 description 2
- SMBBQHHYSLHDHF-UHFFFAOYSA-M 2-acetyloxyethyl(trimethyl)azanium;iodide Chemical compound [I-].CC(=O)OCC[N+](C)(C)C SMBBQHHYSLHDHF-UHFFFAOYSA-M 0.000 description 1
- GALNBQVDMJRFGJ-UHFFFAOYSA-M 2-butanoyloxyethyl(trimethyl)azanium;iodide Chemical compound [I-].CCCC(=O)OCC[N+](C)(C)C GALNBQVDMJRFGJ-UHFFFAOYSA-M 0.000 description 1
- FQRQWPNYJOFDLO-UHFFFAOYSA-N 2-hydroxy-4,6-dimethoxybenzaldehyde Chemical compound COC1=CC(O)=C(C=O)C(OC)=C1 FQRQWPNYJOFDLO-UHFFFAOYSA-N 0.000 description 1
- VMZCDNSFRSVYKQ-UHFFFAOYSA-N 2-phenylacetyl chloride Chemical compound ClC(=O)CC1=CC=CC=C1 VMZCDNSFRSVYKQ-UHFFFAOYSA-N 0.000 description 1
- MJUHIAWSJRKXSH-UHFFFAOYSA-N 2-pyridin-3-ylacetaldehyde Chemical compound O=CCC1=CC=CN=C1 MJUHIAWSJRKXSH-UHFFFAOYSA-N 0.000 description 1
- 101710108761 Acetylcholinesterase-1 Proteins 0.000 description 1
- 208000024827 Alzheimer disease Diseases 0.000 description 1
- VHUUQVKOLVNVRT-UHFFFAOYSA-N Ammonium hydroxide Chemical compound [NH4+].[OH-] VHUUQVKOLVNVRT-UHFFFAOYSA-N 0.000 description 1
- CKJRQPWAXMFUCK-UHFFFAOYSA-N C.CC(C)=O Chemical compound C.CC(C)=O CKJRQPWAXMFUCK-UHFFFAOYSA-N 0.000 description 1
- WEGYGNROSJDEIW-UHFFFAOYSA-N CC(=O)C1=CC=CN=C1 Chemical compound CC(=O)C1=CC=CN=C1 WEGYGNROSJDEIW-UHFFFAOYSA-N 0.000 description 1
- NXHUHEUOFBLGGT-UHFFFAOYSA-N CC.CCc1ccccc1 Chemical compound CC.CCc1ccccc1 NXHUHEUOFBLGGT-UHFFFAOYSA-N 0.000 description 1
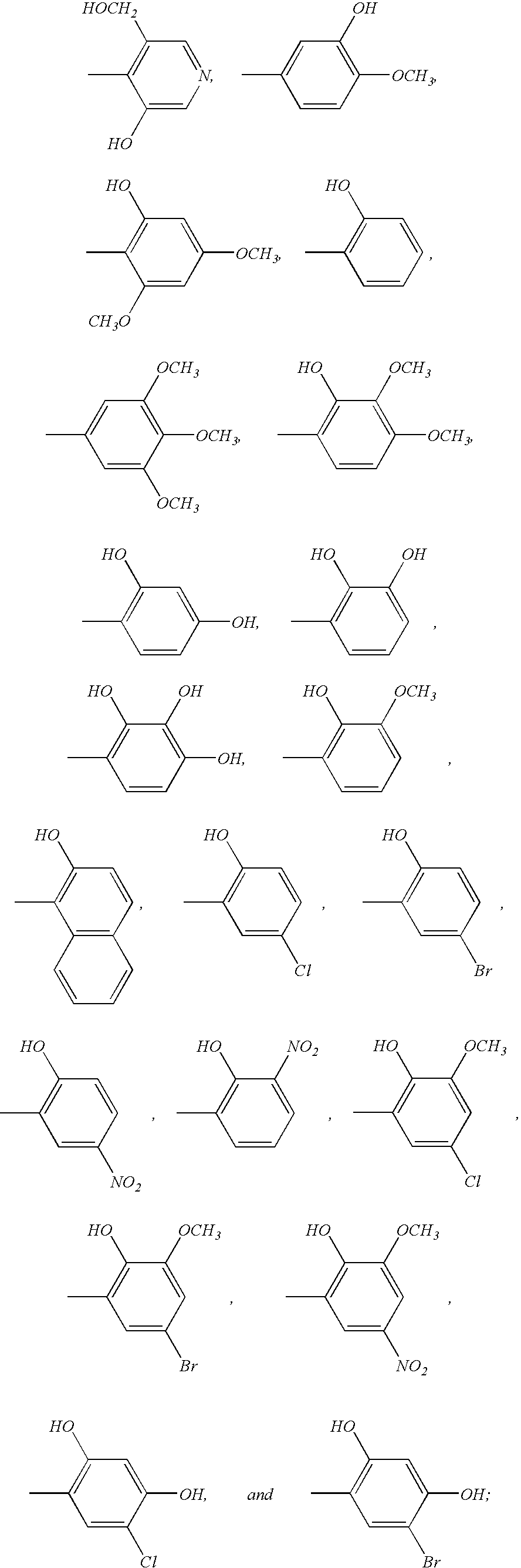
- PHZREHARUPHHCU-UHFFFAOYSA-N CC1=C(O)C(O)=C(O)C=C1 Chemical compound CC1=C(O)C(O)=C(O)C=C1 PHZREHARUPHHCU-UHFFFAOYSA-N 0.000 description 1
- PGSWEKYNAOWQDF-UHFFFAOYSA-N CC1=C(O)C(O)=CC=C1 Chemical compound CC1=C(O)C(O)=CC=C1 PGSWEKYNAOWQDF-UHFFFAOYSA-N 0.000 description 1
- AQDKZPFDOWHRDZ-UHFFFAOYSA-N CC1=C(O)C([N+](=O)[O-])=CC=C1 Chemical compound CC1=C(O)C([N+](=O)[O-])=CC=C1 AQDKZPFDOWHRDZ-UHFFFAOYSA-N 0.000 description 1
- BBORPPIIXOVCGX-UHFFFAOYSA-N CC1=C(O)C=C(O)C(Br)=C1 Chemical compound CC1=C(O)C=C(O)C(Br)=C1 BBORPPIIXOVCGX-UHFFFAOYSA-N 0.000 description 1
- BVKBTTMNHDZLII-UHFFFAOYSA-N CC1=C(O)C=C(O)C(Cl)=C1 Chemical compound CC1=C(O)C=C(O)C(Cl)=C1 BVKBTTMNHDZLII-UHFFFAOYSA-N 0.000 description 1
- FNYDIAAMUCQQDE-UHFFFAOYSA-N CC1=C(O)C=C(O)C=C1 Chemical compound CC1=C(O)C=C(O)C=C1 FNYDIAAMUCQQDE-UHFFFAOYSA-N 0.000 description 1
- IWJGMJHAIUBWKT-UHFFFAOYSA-N CC1=C(O)C=CC(Br)=C1 Chemical compound CC1=C(O)C=CC(Br)=C1 IWJGMJHAIUBWKT-UHFFFAOYSA-N 0.000 description 1
- KDQPMQNHVQVVMR-UHFFFAOYSA-N CC1=C(O)C=CC([N+](=O)[O-])=C1 Chemical compound CC1=C(O)C=CC([N+](=O)[O-])=C1 KDQPMQNHVQVVMR-UHFFFAOYSA-N 0.000 description 1
- BBOCZFGVXFNCTC-UHFFFAOYSA-N CC1=C(O)C=CC2=C1C=CC=C2 Chemical compound CC1=C(O)C=CC2=C1C=CC=C2 BBOCZFGVXFNCTC-UHFFFAOYSA-N 0.000 description 1
- QWVGKYWNOKOFNN-UHFFFAOYSA-N CC1=C(O)C=CC=C1 Chemical compound CC1=C(O)C=CC=C1 QWVGKYWNOKOFNN-UHFFFAOYSA-N 0.000 description 1
- CKKOGHDGZJIUJF-UHFFFAOYSA-N CC1=CN(C)CC=C1 Chemical compound CC1=CN(C)CC=C1 CKKOGHDGZJIUJF-UHFFFAOYSA-N 0.000 description 1
- SQYIVWVLOAIJDI-UHFFFAOYSA-N CC1=CN(C)CCC1 Chemical compound CC1=CN(C)CCC1 SQYIVWVLOAIJDI-UHFFFAOYSA-N 0.000 description 1
- QROGIFZRVHSFLM-UHFFFAOYSA-N CC=CC1=CC=CC=C1 Chemical compound CC=CC1=CC=CC=C1 QROGIFZRVHSFLM-UHFFFAOYSA-N 0.000 description 1
- IGWQTPINFQSICW-UHFFFAOYSA-N CC=CC1=CC=CO1 Chemical compound CC=CC1=CC=CO1 IGWQTPINFQSICW-UHFFFAOYSA-N 0.000 description 1
- IFNDEOYXGHGERA-UHFFFAOYSA-N COC1=C(O)C=C(C)C=C1 Chemical compound COC1=C(O)C=C(C)C=C1 IFNDEOYXGHGERA-UHFFFAOYSA-N 0.000 description 1
- CFBDXCCCOMMRPK-UHFFFAOYSA-N COC1=C(OC)C(O)=C(C)C=C1 Chemical compound COC1=C(OC)C(O)=C(C)C=C1 CFBDXCCCOMMRPK-UHFFFAOYSA-N 0.000 description 1
- PKNAATJMQOUREZ-UHFFFAOYSA-N COC1=C(OC)C(OC)=C(C(C)=O)C=C1 Chemical compound COC1=C(OC)C(OC)=C(C(C)=O)C=C1 PKNAATJMQOUREZ-UHFFFAOYSA-N 0.000 description 1
- UGZGMKGTJRTCFV-UHFFFAOYSA-N COC1=CC(Br)=CC(C)=C1O Chemical compound COC1=CC(Br)=CC(C)=C1O UGZGMKGTJRTCFV-UHFFFAOYSA-N 0.000 description 1
- KCIZTNZGSBSSRM-UHFFFAOYSA-N COC1=CC(C)=CC(OC)=C1OC Chemical compound COC1=CC(C)=CC(OC)=C1OC KCIZTNZGSBSSRM-UHFFFAOYSA-N 0.000 description 1
- LXUHYMCTFZFCCV-UHFFFAOYSA-N COC1=CC(O)=C(C)C(OC)=C1 Chemical compound COC1=CC(O)=C(C)C(OC)=C1 LXUHYMCTFZFCCV-UHFFFAOYSA-N 0.000 description 1
- WNJLQISFTXLCRT-UHFFFAOYSA-N COC1=CC([N+](=O)[O-])=CC(C)=C1O Chemical compound COC1=CC([N+](=O)[O-])=CC(C)=C1O WNJLQISFTXLCRT-UHFFFAOYSA-N 0.000 description 1
- WBHAUHHMPXBZCQ-UHFFFAOYSA-N COC1=CC=CC(C)=C1O Chemical compound COC1=CC=CC(C)=C1O WBHAUHHMPXBZCQ-UHFFFAOYSA-N 0.000 description 1
- 229940122601 Esterase inhibitor Drugs 0.000 description 1
- ZQPQGKQTIZYFEF-WCVJEAGWSA-N Huperzine Natural products C1([C@H]2[C@H](O)C(=O)N[C@H]2[C@@H](O)C=2C=CC=CC=2)=CC=CC=C1 ZQPQGKQTIZYFEF-WCVJEAGWSA-N 0.000 description 1
- 241000699666 Mus <mouse, genus> Species 0.000 description 1
- 229910019142 PO4 Inorganic materials 0.000 description 1
- 206010036631 Presenile dementia Diseases 0.000 description 1
- 229940124639 Selective inhibitor Drugs 0.000 description 1
- DBMJMQXJHONAFJ-UHFFFAOYSA-M Sodium laurylsulphate Chemical compound [Na+].CCCCCCCCCCCCOS([O-])(=O)=O DBMJMQXJHONAFJ-UHFFFAOYSA-M 0.000 description 1
- 239000004141 Sodium laurylsulphate Substances 0.000 description 1
- 150000008065 acid anhydrides Chemical class 0.000 description 1
- 150000001263 acyl chlorides Chemical class 0.000 description 1
- 150000001298 alcohols Chemical class 0.000 description 1
- 150000001299 aldehydes Chemical class 0.000 description 1
- 239000003513 alkali Substances 0.000 description 1
- 150000001408 amides Chemical class 0.000 description 1
- 125000003277 amino group Chemical group 0.000 description 1
- 235000011114 ammonium hydroxide Nutrition 0.000 description 1
- 229910052786 argon Inorganic materials 0.000 description 1
- 239000007853 buffer solution Substances 0.000 description 1
- 239000003795 chemical substances by application Substances 0.000 description 1
- OEYIOHPDSNJKLS-UHFFFAOYSA-N choline Chemical compound C[N+](C)(C)CCO OEYIOHPDSNJKLS-UHFFFAOYSA-N 0.000 description 1
- 229960001231 choline Drugs 0.000 description 1
- 238000004737 colorimetric analysis Methods 0.000 description 1
- 238000009833 condensation Methods 0.000 description 1
- 230000005494 condensation Effects 0.000 description 1
- 230000007547 defect Effects 0.000 description 1
- 238000013461 design Methods 0.000 description 1
- 238000010586 diagram Methods 0.000 description 1
- 230000008034 disappearance Effects 0.000 description 1
- 238000010828 elution Methods 0.000 description 1
- 239000002329 esterase inhibitor Substances 0.000 description 1
- 238000000605 extraction Methods 0.000 description 1
- 230000006870 function Effects 0.000 description 1
- 239000007789 gas Substances 0.000 description 1
- 235000008216 herbs Nutrition 0.000 description 1
- 230000009191 jumping Effects 0.000 description 1
- 150000007522 mineralic acids Chemical class 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 239000002808 molecular sieve Substances 0.000 description 1
- 229930014626 natural product Natural products 0.000 description 1
- 239000003960 organic solvent Substances 0.000 description 1
- KJIFKLIQANRMOU-UHFFFAOYSA-N oxidanium;4-methylbenzenesulfonate Chemical compound O.CC1=CC=C(S(O)(=O)=O)C=C1 KJIFKLIQANRMOU-UHFFFAOYSA-N 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 1
- 239000010452 phosphate Substances 0.000 description 1
- LGRFSURHDFAFJT-UHFFFAOYSA-N phthalic anhydride Chemical compound C1=CC=C2C(=O)OC(=O)C2=C1 LGRFSURHDFAFJT-UHFFFAOYSA-N 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- BDERNNFJNOPAEC-UHFFFAOYSA-N propan-1-ol Chemical compound CCCO BDERNNFJNOPAEC-UHFFFAOYSA-N 0.000 description 1
- 125000004076 pyridyl group Chemical group 0.000 description 1
- 238000007086 side reaction Methods 0.000 description 1
- 238000010898 silica gel chromatography Methods 0.000 description 1
- URGAHOPLAPQHLN-UHFFFAOYSA-N sodium aluminosilicate Chemical compound [Na+].[Al+3].[O-][Si]([O-])=O.[O-][Si]([O-])=O URGAHOPLAPQHLN-UHFFFAOYSA-N 0.000 description 1
- 235000019333 sodium laurylsulphate Nutrition 0.000 description 1
- 239000007858 starting material Substances 0.000 description 1
- 230000002194 synthesizing effect Effects 0.000 description 1
- 231100000331 toxic Toxicity 0.000 description 1
- 230000002588 toxic effect Effects 0.000 description 1
- 231100000820 toxicity test Toxicity 0.000 description 1
- 229940086542 triethylamine Drugs 0.000 description 1
- 230000003936 working memory Effects 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/06—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings linked by a carbon chain containing only aliphatic carbon atoms
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/28—Drugs for disorders of the nervous system for treating neurodegenerative disorders of the central nervous system, e.g. nootropic agents, cognition enhancers, drugs for treating Alzheimer's disease or other forms of dementia
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D221/00—Heterocyclic compounds containing six-membered rings having one nitrogen atom as the only ring hetero atom, not provided for by groups C07D211/00 - C07D219/00
- C07D221/02—Heterocyclic compounds containing six-membered rings having one nitrogen atom as the only ring hetero atom, not provided for by groups C07D211/00 - C07D219/00 condensed with carbocyclic rings or ring systems
- C07D221/22—Bridged ring systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/12—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/14—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D405/00—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom
- C07D405/02—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing two hetero rings
- C07D405/12—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing two hetero rings linked by a chain containing hetero atoms as chain links
Definitions
- the present invention relates to semi-synthesis of natural product, and particularly to alkaloid and analogues thereof.
- the present invention has made use of the excellent resources of Chinese herbs in China to design a semi-synthesis starting from huperzine A, in the hope of finding among a great variety of huperzine A derivatives compounds having better treatment effect and lower toxicity than the existing huperzine A.
- the present invention is carried out through the following steps:
- Y is —C ⁇ O or —R′′
- Y is ⁇ CH
- R is C 1 -C 5 lower alkyl
- R′ is hydrogen, C 1 -C 5 lower alkyl, pyridoyl, benzoyl substituted by C 1 -C 5 lower alkyloxy;
- R′′ is hydrogen and C 1 -C 5 lower alkyl.
- R is C 1 -C 5 lower alkyl
- X is hydrogen, C 1 -C 5 lower alkyloxy, carboxy, alkyloxycarbonyl, pyridyl, dihydro or tetrahydropyridyl substituted by C 1 -C 5 lower alkyl at the nitrogen atom;
- —(CH 2 ) m COOZ group, m 0-5, Z is hydrogen or C 1 -C 5 lower alkyl;
- —CH ⁇ CH—G group, G is phenyl, furanyl, carboxy, alkyloxycarbonyl;
- R′ is hydrogen, C 1 -C 5 lower alkyl, pyridoyl, benzoyl substituted by C 1 -C 5 lower alkyloxy;
- R′′ is hydrogen and C 1 - 5 lower alkyl.
- X is hydrogen, C 1 -C 5 lower alkyl, C 1 -C 5 lower alkyloxy, nitro, halogen, hydroxy, hydroxymethyl, amino substituted by bis-C 1 -C 5 lower alkyl; —CH ⁇ CH—G group, G is phenyl, furanyl;
- R′ is hydrogen and C 1 -C 5 lower alkyl.
- a calorimetric method reported by Ellman was used to determine the effect of inhibition of the pharmaceutical on enzyme activity.
- the total volume of the solution of the enzyme activity reaction was 4 ml, which contained 0.3 mmol/L of acetylcholin iodide (acetylcholinesterase substrate), or 0.4 mmol/L of butyrylcholin iodide (butyrylcholinesterase substrate), 1 ml of the buffer solution of 25 mmol/L of phosphate of pH 7.4, and water to make up to 4 ml (including the amount of the enzyme solution and the testing reagent added afterwards). After the solution was kept at 37° C.
- a diagram was plotted with the percentage of the remaining enzyme activity against gram molecule concentration of the pharmaceutical, so as to obtain the dose of the pharmaceutical at 50% of inhibition of the enzyme activity, i.e., IC 50 .
- the results of the test showed that all the derivatives exhibited inhibition effect on acetylcholinesterase; derivatives No. 1, No. 17, No. 18 and No. 19 exhibited remarkable inhibition effect on acetylcholinesterase; their effect was slightly weaker than that of huperzine A, but apparently stronger than that of physostigmine and galanthamine. They exhibited weaker inhibition effect on butyrylcholinesterase (pseudo enzyme) than huperzine A. Derivatives No. 1 and No.
- mice The critical toxicity test on mice indicated that LD50 of derivatives No. 18 and No. 19 were smaller than, only 1 ⁇ 3 of, that of the compound of formula (I) (see Table 5).
- mice were administered, and tested after 24 hours. comparison with the physiological saline group P ⁇ 0.01 **comparison with scopolamine group *P ⁇ 0.05, **P ⁇ 0.01
- IR ⁇ max 3420, 2900, 1660, 1620, 1580, 1560, 1500, 1460, 1420, 1380, 1350, 1280, 1205, 1150, 1120, 1080, 1010, 970, 910, 840, 790, 755(s), 650, 630, 610, 520 cm ⁇ 1
- IR ⁇ max 2940, 1660(s), 1590, 1560, 1500, 1565, 1420, 1370, 1330, 1300, 1230, 1190, 1130(s), 1005, 960, 930, 840, 770, 735, 725, 660, 605, 540, 530 cm ⁇ 1 .
- IR ⁇ max 3500-2800(br), 1665(s), 1625(s), 1605(s), 1550, 1540, 1450, 1345, 1300, 1230, 1180, 1120, 1080, 975 cm ⁇ 1 .
- IR ⁇ max 3500-2800(br), 1660(s), 1625(s), 1545, 1460, 1370, 1275, 1240, 1120, 1080, 1060, 1020, 835, 775, 740(s), 640, 630, 525 cm ⁇ 1 .
- IR ⁇ max 3450-3360, 2940-2980, 1660(s), 1620(s), 1555, 1465(s), 1255(s), 1190, 1170, 1115, 1085, 970, 930, 840, 780, 740, 680, 640, 630, 520 cm ⁇ 1 .
- IR ⁇ max 3600-3400(br), 2950-2850(br), 1660(s), 1632, 1620, 1550, 1465, 1445, 1300(m), 1175, 1115, 975, 825, 750(s), 690(s), 630, 620, 520 cm ⁇ 1 .
Landscapes
- Organic Chemistry (AREA)
- Chemical & Material Sciences (AREA)
- Health & Medical Sciences (AREA)
- Neurosurgery (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Biomedical Technology (AREA)
- Neurology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Psychiatry (AREA)
- Hospice & Palliative Care (AREA)
- General Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
wherein n is 0 or 1, X is H, (C1-C5) alkyl; (C1-C5) alkyloxy; nitro, halogen, carboxy, alkyloxycarbonyl, hydroxymethyl, hydroxy, amino substituted by bis-(C1-C5) alkyl; —(CH2)m COOZ, wherein m=0-5, Z is H or (C1-C5) alkyl; —CH═CH—G, wherein G is phenyl, furanyl, carboxy, alkyloxycarbonyl; and dihydro- or tetrahydro-pyridyl substituted by (C1-C5) alkyl at the nitrogen atom; R′ is H, (C1-C5) alkyl, pyridoyl, benzoyl substituted by (C1-C5) alkyloxy; R″ is H or (C1-C5) alkyl; processes for their use as acetylcholinesterase inhibitor.
Description
This application is the national phase of PCT/CN95/00100, filed Dec. 26, 1995, published as WO96/20176 on Jul. 4, 1996.
The present invention relates to semi-synthesis of natural product, and particularly to alkaloid and analogues thereof.
In the past ten years a lot of researches have been made in foreign countries about application of choline esterase inhibitor to enhance the function of the intracerebral cholinergic system for the treatment of presenile dementia. Although delightful results of research have been obtained, there still exist some defects; at the time of producing the treatment effect, there occurs a relatively serious toxic side reaction; and the duration of the effect is relatively short.
In recent years China has isolated from a Chinese herb Lycopodium serratum Thunb., a new alkaloid huperzine A (5R, 9R, 11E)-5-amino-11-ethylidene-5,6,9,10-tetrahydro-7-methyl-5,9-methylene cycloocta [b] pyridyl-2(1H)-one having the formula (I)
Upon a pharmacological study it is proved that it exhibits highly effective reversible anticholinesterase activity, and has selective inhibitory effect on intracerebral acetylcholinesterase (U.S. Pat. No. 5,177,082). In foreign countries modification of the structure of huperzine A has been carried out and analogues of huperzine A have been synthesized in hopes of discovering therefrom compounds having anticholinesterase activity [J. Org. Chem. 56, 1991(4636-4645)] and finding a method of synthesizing huperzine A [U.S. Pat. No. 4,929,731]. However, neither a good method nor an analogue having a better effect has been found so far.
The present invention has made use of the excellent resources of Chinese herbs in China to design a semi-synthesis starting from huperzine A, in the hope of finding among a great variety of huperzine A derivatives compounds having better treatment effect and lower toxicity than the existing huperzine A.
The present invention is carried out through the following steps:
1. Using alcohols such as ethanol as extraction solvent, from the plant Lycopodium serratum Thunb., the residue obtained is concentrated, and then treated with an inorganic acid (such as hydrochloric acid). The aqueous layer is neutralized with alkali (such as ammonia water, NaOH), after which an organic solvent (such as chloroform) is used to extract the total alkaloid. After the treatment, separation by layer chromatography is carried out to produce the compound of the formula (I).
2. After condensation is carried out between the compound of the formula (I), and the corresponding substituted aldehyde or the corresponding substituted acyl chloride or acid anhydride in anhydrous solvent, a compound of the formula (II) is obtained.
n=0,1, X is hydrogen, C1-C5 lower alkyl, C1-C5 lower alkyloxy, nitro, halogen, carboxy, alkyloxycarbonyl, hydroxymethyl, hydroxy, amino substituted by bis-C1-C5 lower alkyl; —(CH2)m COOZ group, m=0-5, Z is hydrogen or C1-C5 lower alkyl; —CH═CH—G group, G is phenyl, furanyl, carboxy, alkyloxycarbonyl; dihydro or tetrahydropyridyl substituted by C1-C5 lower alkyl at the nitrogen atom;
R′ is hydrogen, C1-C5 lower alkyl, pyridoyl, benzoyl substituted by C1-C5 lower alkyloxy;
R″ is hydrogen and C1-C5 lower alkyl.
n=0,1, X is hydrogen, C1-C5 lower alkyloxy, carboxy, alkyloxycarbonyl, pyridyl, dihydro or tetrahydropyridyl substituted by C1-C5 lower alkyl at the nitrogen atom; —(CH2)m COOZ group, m=0-5, Z is hydrogen or C1-C5 lower alkyl; —CH═CH—G group, G is phenyl, furanyl, carboxy, alkyloxycarbonyl;
R′ is hydrogen, C1-C5 lower alkyl, pyridoyl, benzoyl substituted by C1-C5 lower alkyloxy;
R″ is hydrogen and C1-5 lower alkyl.
n=0, X is hydrogen, C1-C5 lower alkyl, C1-C5 lower alkyloxy, nitro, halogen, hydroxy, hydroxymethyl, amino substituted by bis-C1-C5 lower alkyl; —CH═CH—G group, G is phenyl, furanyl;
R′ is hydrogen and C1-C5 lower alkyl.
Huperzine A derivatives:
| No. | Y | R | R′ | R″ |
| No. 1 | C═O | HOOC—CH2CH2— | H | H |
| No. 2 | C═O | C6H5CH2— | H | H |
| No. 3 | C═O |
|
CH3 | CH3 |
| ═ | ||||
| No. 4 | C═O |
|
H | H |
| ═ | ||||
| No. 5 | C═O |
|
|
H |
| ═ | ═ | |||
| No. 6 | C═O |
|
H | H |
| ═ | ||||
| No. 7 | C═O | 4-CH3OC6H4— | H | H |
| No. 8 | C—O | 2,3,4-(CH3O)3C6H2— | H | H |
| No. 9 | C═O | 2,3,4-(CH3O)3C6H2— |
|
H |
| — | ||||
| No. 10 | C═O | (CH3)2CH— | H | H |
| No. 11 | C═O | C6H5— | H | H |
| No. 12 | C═O | 2-py | H | H |
| No. 13 | C═O | 3-py | 3-py-CO— | H |
| No. 14 | C═O | 4-py | H | H |
| No. 15 | C═O | 2-HOOC—C6H4— | H | H |
| No. 16 | C═O | trans-HOOCCH2CH═CH— | H | H |
| No. 17 | CH |
|
H | — |
| No. 18 | CH |
|
H | — |
| No. 19 | CH |
|
H | — |
| No. 20 | CH |
|
H | — |
| No. 21 | CH |
|
H | — |
| No. 22 | CH |
|
H | — |
| No. 23 | CH |
|
H | — |
| No. 24 | CH |
|
H | — |
| No. 25 | CH |
|
H | — |
| No. 26 | CH |
|
H | — |
| No. 27 | CH |
|
H | — |
| No. 28 | CH |
|
H | — |
| No. 29 | CH |
|
H | — |
| No. 30 | CH |
|
H | — |
| No. 31 | CH |
|
H | — |
| No. 32 | CH |
|
H | — |
| No. 33 | CH |
|
H | — |
| No. 34 | CH |
|
H | — |
| No. 35 | CH |
|
H | — |
| No. 36 | CH |
|
H | — |
| No. 37 | CH |
|
H | — |
| No. 38 | CH |
|
H | — |
Pharmacological effect of huperzine A derivatives:
In the present invention a calorimetric method reported by Ellman was used to determine the effect of inhibition of the pharmaceutical on enzyme activity. The total volume of the solution of the enzyme activity reaction was 4 ml, which contained 0.3 mmol/L of acetylcholin iodide (acetylcholinesterase substrate), or 0.4 mmol/L of butyrylcholin iodide (butyrylcholinesterase substrate), 1 ml of the buffer solution of 25 mmol/L of phosphate of pH 7.4, and water to make up to 4 ml (including the amount of the enzyme solution and the testing reagent added afterwards). After the solution was kept at 37° C. for 5 minutes, 0.1-0.2 ml of enzyme solution (red cell membrane or serum of rat) was added, or 0.1-0.3 ml of the testing reagent was added at the same time. The solution was kept at the same temperature for another 8 minutes. Then 1 ml of 3% of sodium lauryl sulphate was added to terminate the reaction. Finally 1 ml of 0.2% 5,5-dithio-2,2′-nitro-benzoic acid solution was added for developing. “721” spectrophotometer was used to determine the density of light at 440 nm. A solution without addition of the pharmaceutical to inhibit the enzyme activity was used as a control (100%). A diagram was plotted with the percentage of the remaining enzyme activity against gram molecule concentration of the pharmaceutical, so as to obtain the dose of the pharmaceutical at 50% of inhibition of the enzyme activity, i.e., IC50. The results of the test showed that all the derivatives exhibited inhibition effect on acetylcholinesterase; derivatives No. 1, No. 17, No. 18 and No. 19 exhibited remarkable inhibition effect on acetylcholinesterase; their effect was slightly weaker than that of huperzine A, but apparently stronger than that of physostigmine and galanthamine. They exhibited weaker inhibition effect on butyrylcholinesterase (pseudo enzyme) than huperzine A. Derivatives No. 1 and No. 17 exhibited greater selective inhibition effect on acetylcholinesterase than huperzine A (see Tables 1, 2 and 6). A study on kinetics of enzyme indicated that the combination of derivatives No. 17, No. 18 and No. 19 respectively with acetylcholinesterase was reversible.
The two memory model test, mouse passive escape operation (Platform jumping method) and 8-arm maze spatial distinction operation of rat, indicated that both derivatives No. 18 and No. 19 exhibited very strong effect, similar to that of the compound of formula (I), on improving memory (see Tables 3 and 4).
The critical toxicity test on mice indicated that LD50 of derivatives No. 18 and No. 19 were smaller than, only ⅓ of, that of the compound of formula (I) (see Table 5).
| TABLE 1 |
| In-vitro anticholinesterase effect of huperzine |
| A derivatives (determined by colorimetric method) |
| concentration for inhibition | ||
| of 50% of enzyme activity | ||
| (IC50 μM) |
| acetylcholin- | butyrylcholin- | ||
| esterase | esterase | IC50 ratio | |
| Compound No. | (AChE) | (BuChE) | BuChE/AChE |
| 1 | 0.348 | 380.19 | 1092.5 |
| 2 | 9.05 | >346.7 | |
| 3 | 3.63 | >331.1 | |
| 4 | >12.88 | 58.9 | |
| 5 | >10.96 | >275.4 | |
| 6 | >12.3 | >309.1 | |
| 17 | 0.172 | 199.5 | 1159.9 |
| 18 | 0.151 | 107.2 | 709.9 |
| 19 | 0.145 | 104.7 | 722.1 |
| 10 | >15.85 | 109.6 | |
| 11 | >14.45 | 363 | |
| compound(I) | 0.06309 | 63.09 | 1000 |
| Physostigmine | 0.251 | 1.259 | 5.02 |
| galanthamine | 1.995 | 12.59 | 6.3 |
AChE taken from rat's red cell membrane. BuChE taken from rat's serum.
| TABLE 2 |
| In-vitro anticholinesterase effect of huperzine |
| A derivatives |
| concentration for | ||
| inhibition of 50% | ||
| of enzyme activity | ||
| (IC50 μM) |
| acetyl- | butyryl- | |||
| cholin- | cholin- | |||
| esterase | esterase | IC50 ratio | ||
| Compound No. | MW | (AChE) | (BuChE) | BuChE/AChE |
| huperzine A | 242 | 0.0977 | 100.0 | 1023.3 |
| 1 | 342 | 0.3475 | 380.2 | 1094.1 |
| 12 | 376 | 0.1259 | 251.2 | 1995.2 |
| 15 | 390 | 0.6310 | 502.0 | 795.6 |
| 17 | 428 | 0.1718 | 199.5 | 1161.2 |
| 18 | 376 | 0.1514 | 107.2 | 708.2 |
| 19 | 406 | 0.1445 | 104.7 | 724.6 |
| 20 | 346 | 0.1778 | 125.9 | 708.1 |
| 21 | 420 | 0.1413 | 158.5 | 1121.3 |
| 22 | 406 | 0.1259 | 125.9 | 1000.0 |
| 23 | 362 | 0.2512 | 199.5 | 794.2 |
| 24 | 362 | 0.1000 | 158.5 | 1585.0 |
| 25 | 378 | 0.1585 | 251.2 | 1584.9 |
| 27 | 356 | 0.1259 | 100.0 | 794.3 |
| MW: molecular weight | ||||
AChE taken from homogenate of rat's cortex. BuChE taken from rat's serum.
| TABLE 3 |
| Improvement by huperzine A derivatives on the |
| condition of memory impairment caused by scopolamine in |
| passive escape operation |
| No. | delitescence of | ||
| dosage | of | get-down Δ | |
| Group | (mg/kg ip + po) | mice | (sec ± SEM) |
| physiological saline + | −+− | 20 | 71.9 ± 12.9 |
| physiological saline | |||
| scopolamine + | 2+ − | 20 | 29.5 ± 2.7 |
| physiological saline | |||
| scopolamine + | 2 + 0.2 | 20 | 67.7 ± 11.7** |
| derivative No. 18 | 3 + 0.3 | 20 | 64.0 ± 9.8* |
| 2 + 0.4 | 20 | 48.7 ± 6.9 | |
| physiological saline + | −+− | 18 | 81.7 ± 19.0 |
| physiological saline | |||
| scopolamine + | 2+ − | 23 | 32.8 ± 8.2 |
| physiological saline | |||
| scopolamine + | 2 + 0.1 | 11 | 48.9 ± 12.6 |
| derivative No. 19 | 2 + 0.2 | 16 | 71.6 ± 14.5 |
| 2 + 0.3 | 21 | 99.8 ± 16.4** | |
| 2 + 0.4 | 18 | 92.2 ± 15.5* | |
| Δ: Once trained, the mice were administered, and tested after 24 hours. comparison with the physiological saline group P < 0.01 | |||
| **comparison with scopolamine group *P < 0.05, **P < 0.01 | |||
| TABLE 4 |
| Improvement by huperzine derivatives on the |
| condition of memory impairment caused by scopolamine in |
| spatial distinction operation |
| No. of mistakes in | |
| operation before reaching | |
| the standard Δ | |
| X + SEM) |
| dosage | No. | refer- | ||
| (mg/kg | of | ence | working | |
| Group | ip + po) | rats | memory | memory |
| physiological saline + | −+− | 24 | 0.42 ± 0.1 | 0.08 ± 0.01 |
| physiological saline | ||||
| scopolamine + | 0.2+ − | 6 | 1.67 ± 0.21 | 0.33 ± 0.42 |
| physiological saline | ||||
| scopolamine + | 0.2 ± 0.1 | 6 | 1.33 ± 0.21 | 1.33 ± |
| derivative No. 19 | 0.49** | |||
| 0.2 ± 0.3 | 6 | 0.33 ± | 0.17 ± | |
| 0.21** | 0.17** | |||
| physiological saline + | −+− | 24 | 0.33 ± 0.13 | 0.08 ± 0.006 |
| physiological saline | ||||
| scopolamine + | 0.125+ − | 6 | 2.0 ± 0.45 | 2.0 ± 0.52 |
| physiological saline | ||||
| scopolamine + | 0.124 + 0.2 | 6 | 0.67 ± | 0.33 ± |
| derivative No. 18 | 0.33** | 0.13** | ||
| physiological saline + | −+− | 14 | 0.21 ± 0.11 | 0.07 ± 0.07 |
| physiological saline | ||||
| scopolamine + | 0.15+ − | 7 | 2.14 ± 0.14 | 2.57 ± 0.29 |
| physiological saline | ||||
| scopolamine + | 0.15 + 0.25 | 10 | 0.57 ± | 0.86 ± |
| huperzine A | 0.30** | 0.14** | ||
| Δ: 8-arm maze test. After trained to reach the standard (no. of mistake less than one per day for three successive days), the rats were tested. The rat first entering the arm on which there is no food is taken as the reference emory mistake. The rat re-entering the arm on which there is food is taken as the working memory mistake, comparison with the physiological saline group P > 0.01 | ||||
| **comparison with scopolamine group P < 0.01 | ||||
| TABLE 5 |
| Critical toxicity of huperzine A derivatives on |
| mice (Bliss method) |
| mg/kg p.o. (95% confidence limit)* |
| Compound | LD50 | LD50 | ||
| compound (I) | 3.1(3.5-3.8) | 4.6(4.2-5.1) | ||
| derivative No. 18 | 9.6(7.3-12.5) | 14.4(12.9-16.4) | ||
| derivative No. 19 | 11.1(9.6-12.9) | 14.1(15.5-20.5) | ||
| *10 mice per group, with equal no. of males and females. | ||||
Test of each pharmaceutical was carried out using 4-5 dosage group.
Mortality within 7 days was observed.
| TABLE 6 |
| In-vitro anticholinesterase effect of huperzine |
| A derivatives |
| inhibition activity | |||
| Compound | AChE | BuChE |
| No. | 1(μM) | IC50(M) | 10(μM) | BuChE/AChE |
| (18) | 90.2 | 8.4 × 10−8 | 0 | >500 |
| (20) | 83.7 | 9.4 × 10−8 | 0 | >500 |
| (21) | 84.9 | 1.0 × 107 | 0 | >500 |
| (22) | 68.7 | 0 | ||
| (23) | 34.4 | 0 | ||
| (15) | 60.8 | 0 | ||
| (24) | 78.3 | 2.8 × 10−7 | 0 | >100 |
| (25) | 62.0 | 0 | ||
| (27) | 84.0 | 1.1 × 10−7 | 3.3 | >500 |
| (26) | 79.7 | 1.8 × 10−7 | 1.1 | >300 |
| (16) | 80.7 | 2.1 × 10−7 | 1.8 | >200 |
| (14) | 76.4 | 3.6 × 10−7 | 2.5 | >100 |
| (44) | 85.6 | 1.1 × 10−7 | 1.8 | >500 |
| (45) | 87.0 | 1.0 × 10−7 | 1.8 | >500 |
| (46) | 78.7 | 2.9 × 10−7 | 1.2 | >100 |
| E-2020 | 86.2 | 6.6 × 10−8 | 2.9 | >500 |
| huperzine A | 88.3 | 1.2 × 10−7 | 0 | >500 |
The results of the above pharmacological study show that derivatives No. 17, No. 18 and No. 19 are highly effective selective inhibitors of acetylcholinesterase and have lower critical toxicity than compound (I). Hence, it can be deduced that they have prospect of clinical application and development for use in the treatment of relieving serious amyasthenia and of dysmnesia caused by failure in central cholinergic system.
Compound (I) (0.50 mmole) into 5 ml three-neck flask was weight out. 20 ml of anhydrous pyridine was added to dissolve compound (I). While cooling in ice-bath, phenyl acetyl chloride (0.55 mmole) was added dropwise. Then the whole mixture was stirred overnight at room temperature (25° C.). When thin-layer chromatography indicated substantial disappearance of the starting materials, the reaction was stopped. Pressure was reduced by means of water pump to vaporize pyridine. Separation was carried out using silica gel layer chromatography. Elution was carried out with dichloromethane:acetone:methanol=5:45:5 to produce the crude product, which was recrystallized using a mixed solvent of acetone and petroleum ether to produce the product with 75% yield.
UV λmax=229 nm (∈=17360); λmax=316 nm(∈=9320); [α]D 25° C.=29.43; 1HNMR: [CDCl3]; 3H 6.31(1H,d,J=9.9 Hz); 4H 7.20 (1H,d,J=9.9 Hz); 10H 2.89(2H,m); 9H 3.52 (1H,m); 8H 5.38(1H,d,J=5.0 Hz); 14-H 1.62(3H,d,J=6.7 Hz); 13-H 5.08(1H,q,J=6.7 Hz); 6-H 2.15,2.45(2H,m); 12-H 1.50(3H,s); 2′,4′-H 7.36(2H,m); 3′,5′-H 7.29(2H,m); 6′-H 7.24(1H,m); 7′-H 3.59(s); MS(m/z) 360(M+) 345 269 252 227 224 210(100%) 91; mp: 171-173° C.; IR: υmax 3280, 1660(s), 1620(s), 1550, 1450, 1350, 1300, 1175, 1130, 840, 620 cm−1
The same process was used to prepare derivatives No. 3, No. 5, No. 6, No. 7, No. 8, No. 9, No. 10, No. 11, and No. 13.
Compound (I) (0.5 mmole) into 50 ml three-neck flask was weight out. 20 ml of anhydrous pyricline to dissolve compound (I) was added. While cooling in ice-bath, DBU (0.6 mmole) was first added, and then 0.55 mmole of hydrochloric acid pyridine-2-formyl chloride was added. The whole mixture was placed overnight at room temperature. When thin-layer chromatography determination indicated substantial completion of the reaction, pressure was reduced by means of water pump to vaporize pyridine. Separation was carried out using silica gel layer chromatography. Elutioni was carried out with dichloromethane:acetone:methanol=50:45:5 to produce the crude product, which was recrystallized using a mixed solvent of acetone and petroleum ether to produce the product with 74% yield.
UV λmax=226 nm (∈=1.35×104); λmax=264 nm (∈=5.4·103); λmax=315 nm (∈=5730); [α]D=77.85°; 1HNMR: [CDCl3]; 3-H 6.36(1H,J=9.2 Hz); 4-H 7.44(1H,J=9.2 Hz); 10-H 3.05(2H,m); 9-H 3.74(1H,m); 8-H 5.42(1H,d,J=4.7 Hz); 14-H 1.65(3H,d,J=6.6 Hz); 13-H 5.35(1H,q=6.6 Hz); 6-H 2.42(2H,s); 12-H 1.55(3H,s); 2′-H 8.58(1H,m); 4′-H 7.85(1H,m); 5′-H 7.48(1H,m); 6′-H′ 8.15(1H,m); MS(m/z) 347(M+), 241(100%) 169, 149, 106, 95, 79, 71, 55; mp: 170-171° C.; IR: umax 3450, 2900, 1660(s), 1615(s), 1530(s), 1460, 1300, 1180, 1140, 1000, 830, 750 cm−1
The same process was used to prepare derivatives No. 12 and No. 14.
Compound (I) (0.5 mmole) was weight out into 50 ml three-neck flask. Anhydrous ethanol and 4,6-dimethoxy-2-hydroxy benzaldehyde (0.51 mmole) were added. The mixture was heated slightly under reflux. Through a water segregator part of the ethanol was vaporized continuously. The solvent in the reaction was constantly replenished. The reaction was carried out for several hours and the reaction state was constantly determined by means of thin-layer chromatography. When the reaction was completed, the pressure was reduced to remove the ethanol to produce a solid crude product, which was recrystallized using a mixed solvent of acetone and petroleum ether to produce the product with 92% yield.
The same process was used to prepare derivatives No. 17, 18, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, and 43.
Derivative No. 19
m.p. 207-210° C.; MS: m/z 406(M+); 391(M+-CH3), 351(M+-C4H7); 1H NMR (CDCl3); 1-H 13.12(1H,br,s); 3-H 6.41(1H,d,J=9.1 Hz); 4-H 7.25(1H,d,J=9.1 Hz); 6-H 2.85 (2H,s); 8-H 3.45(1H,d,J=4.7 Hz); 9-H 3.61(1H,m); 10-H 2.85(2H,m); 12-H3 1.60(3H,s); 13-H 5.22(1H,q,J=7.7 Hz); 14-H3 1.25(3H,d,J=7.7 Hz); 2′-OH 14.58(1H,br,s); 3′-H 5.57 (1H,s); 4′-OCH3 3.79(3H,s); 5′-H 5.85(1H,s); 6′-OCH3 3.70 (3H,s); 7′-H 8.15(1H,s); IR: υmax 3480(m), 2960(s), 2930(s), 2870(s), 1670(s), 1620(s), 1540(s), 1450(s), 1330 (s), 1300 (s), 1218(sh), 1186(m), 1155(s), 1110(s), 1080(m), 1050(m), 1000(m), 930(m), 840(s), 730(m), 670(m), 610(m), 520(s) cm−1.
Derivative No. 20
1H NMR (CDCl3): 1-H 12.72(1H,br,s); 3-H 6.34(1H,d, J=9.4 Hz); 4-H 7.11(1H,d,J=9.4 Hz); 6a-H 2.79(1H,d,J=16.0 Hz); 6b-H 2.75(1H,d,J=16.0 Hz); 8-H 5.41(1H,d,J=5.0 Hz); 9-H 3.63(1H,m); 10a-H 2.95(1H,dd,J=16.6, 4.9 Hz); 10b-H 2.27(1H,d,J=16.6 Hz); 12-H 1.57(3H,s); 13-H 5.07(1H,q,J=6.8 Hz); 14-H 1.55(3H,d,J=6.8 Hz); 2′-OH 13.77(1H,br,s); 3′-H 6.94(1H,d,J=8.4 Hz); 4′-H 7.30(1H,t with small splitts, J=8.4 Hz); 5′-H 6.85(1H,t with small splitts, J=7.4 Hz); 6′-H 7.22(1H,dd,J=7.4, 1.6 Hz); 7′-H 8.31(1H,s); MS: 346(M+), 331, 317, 303, 291, 253, 239, 226, 210, 197, 184, 167, 128, 121, 97. IR: υmax 3420, 2900, 1660, 1620, 1580, 1560, 1500, 1460, 1420, 1380, 1350, 1280, 1205, 1150, 1120, 1080, 1010, 970, 910, 840, 790, 755(s), 650, 630, 610, 520 cm−1
Derivative No. 21;
1H NMR (CDCl3); 1-H 12.87(1H,br,s); 3-H 6.32(1H,d, J=9.4 Hz); 4-H 7.07(1H,d,J=9.4 Hz); 6a-H 2.79(1H,d,J=16.0 Hz); 6b-H 2.75(1H,d,J=16.0 Hz); 8-H 5.43(1H,d,J=5.0 Hz); 9-H 3.63(1H,m); 10a-H 2.97(1H,dd,J=16.0, 4.0 Hz); 10b-H 2.18(1H,d,J=16.0 Hz); 12-H 1.58(3H,s); 13-H 5.14(1H,q,J=6.8 Hz); 14-H 1.57(3H,d,J=6.8 Hz); 2′,6′-2H 7.04(2H,s); 7′-H 8.25(1H,s); 3′,5′-2(OCH3) 3.87(6H,s); 4′-OCH3 3.84 (3H,s); MS: 420(M+), 405(M+-CH3), 391, 365, 351, 313, 284, 239, 226, 210, 196, 181, 140, 124. IR: υmax 2940, 1660(s), 1590, 1560, 1500, 1565, 1420, 1370, 1330, 1300, 1230, 1190, 1130(s), 1005, 960, 930, 840, 770, 735, 725, 660, 605, 540, 530 cm−1.
Derivative No. 22
1H NMR(CDCl3): 1-H 13.03(1H,br,s); 3-H 6.34(1H,d,J=9.4 Hz); 4-H 7.14(1H,d,J=9.4 Hz); 6-H2 2.73(2H,s); 8-H 5.39(1H,d,J=4.7 Hz); 9-H 3.61(1H,m); 10a-H 2.94(1H,dd, J=16.8, 4.9 Hz); 10b-H 2.27(1H,d,J=16.8 Hz); 12-H 1.55 (3H,s); 13-H 5.09(1H,q,J=6.8 Hz); 14-H 1.54(3H,d,J=6.8 Hz); 2′-OH 14.39(1H,br.s); 3′-OCH3 3.87(3H,s); 4′-OCH3 3.85(3H,s); 5′-H 6.43(1H,d,J=8.7 Hz); 6′-H 6.91(1H,d,J=8.7 Hz); 7′-H 8.14(1H,s); MS: 406(M+), 391(M+-CH3), 373, 351, 239, 226, 197, 182, 167, 139, 101; IR: υmax 3450-3350 (br), 2940(m), 1660(s), 1620(s), 1555, 1510, 1455, 1415, 1290, 1270(s), 1210, 1110(s), 1060, 970, 930, 835, 785, 695, 675, 640, 625 cm−1.
Derivative No. 23
1H NMR(d6-DMSO): 1-H 11.48(1H,br,s); 3-H 6.13(1H, d,J=9.4 Hz); 4-H 7.07(1H,d,J=9.4 Hz); 6a-H 2.68(1H,d,J=16.7 Hz); 6b-H 2.59(1H,d,J=16.7 Hz); 8-H 5.48(1H,d,J=4.7 Hz); 9-H 3.65(1H,m); 10a-H 2.77(1H,dd,J=16.3, 4.0 Hz); 10b-H 2.28(1H,d,J=16.3 Hz); 12-H 1.59(3H,s); 13-H 5.05 (1H,q,J=6.8 Hz); 14-H 1.60(3H,d,J=6.8 Hz); 2′-OH 14.40 (1H,br,s); 3′-H 6.18(1H,s); 4′-OH 10.09(1H,br,s); 5′-H 6.30 (1H,d, with small splitts, J=8.4 Hz); 6′-H 7.31(1H,d,J=8.4 Hz); 7′-H 8.47(1H,s); MS: 362(M+), 347, 319, 307, 242, 226, 210, 197, 167, 137. IR: υmax 3500-2800(br), 1665(s), 1625(s), 1605(s), 1550, 1540, 1450, 1345, 1300, 1230, 1180, 1120, 1080, 975 cm−1.
Derivative No. 24
1H-NMR(CDCl3): 1-H 13.09(1H,br,s); 3-H 6.37(1H,d,J=9.4 Hz); 4-H 7.15(1H,d,J=9.4 Hz); 6-H2 2.76(2H,s); 8-H 5.42(1H,d,J=5.0 Hz); 9-H 3.60(1H,m); 10a-H 2.96(1H,dd, J=16.5, 4.9 Hz); 10b-H 2.32(1H,d,J=16.5 Hz); 12-H 1.56 (3H,s); 13-H 5.11(1H,q,J=6.8 Hz); 14-H 1.58(3H,d,J=6.8 Hz); 2′-OH 13.09(1H,br,s); 3′-OH 9.83(1H,br,s); 4′-H 6.68 (1H,dd,J=8.1, 1.5 Hz); 5′-H 6.57(1H,t,J=7.8 Hz); 6′-H 6.91 (1H,dd,J=7.6, 1.5 Hz); 7′-H 8.08(1H,s); MS: 362(M+), 347 (M+-CH3), 307, 253, 239, 226 197, 167, 149, 138, 137, 109, 92, 81. IR: υmax 3500-2800(br), 1660(s), 1625(s), 1545, 1460, 1370, 1275, 1240, 1120, 1080, 1060, 1020, 835, 775, 740(s), 640, 630, 525 cm−1.
Derivative No. 26
1HNMR(CDCl3): 1-H 13.10(1H,br,s); 3-H 6.33(1H,d J=9.4 Hz); 4-H 7.12(1H,d,J=9.4 Hz); 6-H2 2.75(2H,s); 8-H 5.41(1H,d,J=4.8 Hz); 9-H 3.62(1H,m); 10a-H 2.95(1H,dd, J=16.5, 4.9 Hz); 10b-H 2.27(1H,d,J=16.5 Hz); 12-H3 1.57 (3H,s); 13-H 5.08(1H,q,J=6.8 Hz); 14-H 1.54(3H,d,J=6.8 Hz); 2′-OH 14.36(1H,br,s); 3′-OCH3 3.86(3H,s); 4′-H 6.83 (1H,dd,J=7.9, 1.5 Hz); 5′-H 6.76 (1H,t,J=7.9 Hz); 6′-H 6.89(1H,dd,J=7.9, 1.5 Hz); 7′-H 8.27(1H,s); MS: 376(M+), 361, 346, 331, 306, 270, 253, 239, 226, 210, 197, 167, 152, 109, 106, 82. IR: υmax 3450-3360, 2940-2980, 1660(s), 1620(s), 1555, 1465(s), 1255(s), 1190, 1170, 1115, 1085, 970, 930, 840, 780, 740, 680, 640, 630, 520 cm−1.
Derivative No. 27
1HNMR(CDCl3): 1-H 12.80(1H,br,s); 3-H 6.34(1H,d,J=9.4 Hz); 4-H 7.07(1H,d,J=9.4 Hz); 6-H2 2.72(2H,s); 8-H 5.40(1H,d,J=5.0 Hz); 9-H 3.61(1H,m); 10a-H 2.94(1H,dd, J=16.2, 3.6 Hz); 10b-H 2.17(1H,d,J=16.2 Hz); 12-H 1.56 (3H,s); 13-H 5.08(1H,q,J=6.8 Hz); 14-H 1.57(3H,d,J=6.8 Hz); 2′,6′-2H 7.45(2H,dd,J=6.8, 1.4 Hz); 3′,5′,8′-3H 7.30 (3H,m); 4′-H 7.06(1H,m); 7′-H 6.99 (1H,d,J=15.9 Hz); 9′-H 8.06(1H,d,J=8.1 Hz); MS: 356(M+), 341(M+-CH3), 327, 301, 265, 237, 226, 210, 197, 167, 131, 115, 91, 77. IR: υmax 3600-3400(br), 2950-2850(br), 1660(s), 1632, 1620, 1550, 1465, 1445, 1300(m), 1175, 1115, 975, 825, 750(s), 690(s), 630, 620, 520 cm−1.
Derivative No. 28
1HNMR(CDCl3): 1-H 12.76(1H,br,s); 3-H 6.38(1H,d,J=9.3 Hz); 4-H 7.12(1H,d,J=9.3 Hz); 6a-H 2.81(1H,d,J=16.0 Hz); 6b-H 2.78(1H,d,J=16.0 Hz); 8-H 5.46(1H,br,s); 9-H 3.67(1H,m); 10a-H 3.00(1H,dd,J=16.4, 4.5 Hz); 10b-H 2.31 (1H,d,J=16.4 Hz); 12-H 1.62(3H,s); 13-H 5.07(1H,q,J=6.6 Hz); 14-H 1.60(3H,d,J=6.6 Hz); 2′-OH 13.76(1H,br,s); 3′-H 6.94(1H,d,J=8.7 Hz); 4′-H 7.29(1H,dd,J=8.7, 2.3 Hz); 5′,6′,7′,8′-4H 7.25(4H,m); 11′-H 8.30(1H,s); MS(m/z): 396(M+, 3%), 381(M+-CH3,1), 226(2), 172(4), 144(2), 127(1), 115 (5), 85(13), 71(22), 57(100). IR(KBr): 3408, 1660, 1626, 1479, 1280 cm−1.
Derivative No. 30
1HNMR(CDCl3): 1-H 10.92(1H,br,s); 3-H 6.42(1H,d,J=9.3 Hz); 4-H 7.15(1H,d,J=9.3 Hz); 6a-H 2.82(1H,d,J=17.2 Hz); 6b-H 2.76(1H,d,J=17.2 Hz); 8-H 5.47(1H,br,s); 9-H 3.69(1H,m); 10a-H 2.99(1H,dd,J=16.0, 3.7 Hz); 10b-H 2.30 (1H,d,J=16.0 Hz); 12-H 1.63(3H,s); 13-H 5.07(1H,q,J=6.8 Hz); 14-H 1.61(3H,d,J=6.8 Hz); 3′-H 6.89(1H,d,J=6.8 Hz); 4′,6′-2H 7.42(2H,m); 7′-H 8.30(1H,s); MS(m/z): 426(M++2,43%), 424(M+, 43), 411[(M++2)-CH3,42], 409(M+-CH3, 45), 369(11), 289(5), 253(18), 239(33), 226(100), 210(54), 197(47), 167(31), 128(31), 115(46), 91(32), 77(59), 57(81). IR(KBr): 3400, 1660, 1630, 1475, 1280, 823 cm−1.
Derivative No. 32
1HNMR(CDCl3): 1-H 12.70(1H,br,s); 3-H 6.39(1H,d,J=9.4 Hz); 4-H 7.11(1H,d,J=9.4 Hz); 6a-H 2.81(1H,d,J=16.2 Hz); 6b-H 2.77(1H,d,J=16.2 Hz); 8-H 5.45(1H,br,s); 9-H 3.67(1H,m); 10a-H 2.99(1H,dd,J=16.7, 4.9 Hz); 10b-H 2.31 (1H,d,J=16.7 Hz); 12-H 1.62(3H,s); 13-H 5.06(1H,q,J=6.8 Hz); 14-H 1.60(3H,d,J=6.8 Hz); 2′-OH 13.80(1H,br,s); 3′-H 6.89(1H,d,J=8.7 Hz); 4′-H 7.42(1H,dd,J=8.7, 2.4 Hz); 6′-H 7.39(1H,d,J=2.4 Hz); 7′-H 8.29(1H,s); MS(m/z): 392(M++1,11%), 391(M+,45), 376(M+-CH3,60), 242(18), 226(53), 101(28), 83(53), 59(100). IR(KBr): 3419, 1660, 1616, 1549, 1336, 833 cm−1.
Derivative No. 33
1HNMR(CDCl3): 1-H 12.40(1H,br,s); 3-H 6.42(1H,d,J=9.5 Hz); 4-H 7.15(1H,d,J=9.5 Hz); 6a-H 2.84(1H,d,J=16.3 Hz); 6b-H 2.77(1H,d,J=16.3 Hz); 8-H 5.47(1H,br,s); 9-H 3.70(1H,m); 10a-H 3.00(1H,dd,J=16.0, 4.9 Hz); 10b-H 2.39 (1H,d,J=16.0 Hz); 12-H 1.58(3H,s); 13-H 5.12(1H,q,J=6.8 Hz); 14-H 1.63(3H,d,J=6.8 Hz); 2′-OH 15.59(1H,br,s); 4′-H 8.16(1H,d,J=7.8 Hz); 5′-H 6.77(1H,t,J=7.8 Hz); 6′-H 7.46 (1H,d,J=7.8 Hz); 7′-H 8.24(1H,s); MS(m/z): 391(M+,66%), 376(M+-CH3,41), 356(45), 328(18), 288(14), 239(15), 226 (100), 210(25), 197(22), 83(61). IR(KBr): 3431, 1662, 1633, 1529, 1352, 1242, 754 cm−1.
Derivative No. 34
1HNMR(CDCl3): 1-H 12.87(1H,br,s); 3-H 6.38(1H,d,J=9.6 Hz); 4-H 7.13(1H,d,J=9.6 Hz); 6-H2 2.78(2H,s); 8-H 5.46(1H,br,s); 9-H 3.67(1H,m); 10a-H 3.00(1H,dd,J=16.5, 4.9 Hz); 10b-H 2.32(1H,d,J=16.5 Hz); 12-H 1.62(3H,s); 13-H 5.09(1H,q,J=6.7 Hz); 14-H 1.59(3H,d,J=6.7 Hz); 2′-OH 14.42(1H,br,s); 3′-OCH3 3.90(3H,s); 4′,6′-2H 6.86 (2H,m); 7′-H 8.22(1H,s); MS(m/z): 412(M++2,0.3%), 410 (M+,1.2), 395(M+-CH3,0.5), 239(1.4), 216(2), 83(100). IR(KBr): 3421, 2933, 1660, 1618, 1471, 1253, 839 cm−1.
Derivative No. 35
1HNMR(CDCl3): 1-H 12.80(1H,br,s); 3-H 6.38(1H,d,J=9.3 Hz); 4-H 7.13(1H,d,J=9.3 Hz); 6-H2 2.78(2H,s); 8-H 5.46(1H,br,s); 9-H 3.67(1H,m); 10a-H 3.00(1H,dd,J=16.7, 5.1 Hz); 10b-H 2.32(1H,d,J=16.7 Hz); 12-H 1.62(3H,s); 13-H 5.08(1H,q,J=6.8 Hz); 14-H 1.59(3H,d,J=6.8 Hz); 2′-OH 14.45(1H,br,s); 3′-OCH3 3.89(3H,s); 4′,6′-2H 7.00 (2H,m); 7′-H 8.21(1H,s); MS(m/z): 456(M+2,79), 454(M+, 7a), 441[(M++2)-CH3, 40], 439(M+-CH3,42), 401(7), 399 (8), 285(14), 239(24), 226(100), 210(33), 197(37), 83(84). IR(KBr): 3431, 1660, 1624, 1471, 1252 cm−1.
Compound (I) (0.5 mmole) was weight out into 50 μl three-neck flask. 20 ml of anhydrous pyridine was added to dissolve compound (I). While cooling in ice-bath, o-phthalic acid anhydride was added. The mixture was stirred overnight at room temperature. When thin-layer chromatography determination indicated substantial completion of the reaction, the pressure was reduced by means of water pump to vaporize pyridine. Separation was carried out using silica gel layer chromatography. The development agent dichloromethane:methanol=3:1 was used to produce the crude product, which was recrystallized using propanol to produce white powders of derivative No. 15 with 78% yield.
The same process was used to prepare derivatives No. 1 to No. 16.
1H NMR(d6-DMSO): 3-H 6.09(1H,d,J=9.4 Hz); 6-H2 2.51(2H,s); 8-H 5.44(1H,d,J=4.8 Hz); 9-H 3.56(1H,m); 10a-H 2.66(1H,dd,J=16.8, 4.6 Hz); 10b-H 2.09(1H,d,J=16.8 Hz); 12-H3 1.51(3H,s); 13-H 5.60(1H,q,J=6.8 Hz); 14-H3 1.62(3H,d,J=6.8 Hz); 3′,4′,5′ and 4-H 7.51(4H,m); 6′-H 7.73(1H,d,J=7.5 Hz); N-H(amide) 7.90(1H,br,s); COO-H 8.46(1H,br,s); IR: υmax 3650-2400, 1710, 1655, 1605, 1546, 1450, 1300 cm−1 MS m/z: 372(M+), 357, 343, 329, 242, 227, 187, 147, 104(100).
Under argon gas stream 150 mg (0.620 mmole) of huperzine A and 200 mg of 4A molecular sieve were suspended in 4 ml of benzene. 76 μl (0.86 mmol) of pyridine-3-acetaldehyde and 20 mg of p-toluene sulphonic acid monohydrate were added. After reflux for three hours, the mixture was allowed to cool, and then neutralized with triethyl amine. The reaction solution was filtered with celite. After concentration, the residue was refined through silica gel column chromatography (methanol:dichloromethane=1:20) to produce derivative No. 44, a colorless solid (183 mg, 89% yield).
The same process was used to prepare derivatives No. 45 and No. 46, with 84% and 82% yield respectively.
Derivative No. 44
NMR(CDCL3) 1.64(3H,d,J=7.0 Hz), 1.65(3H,s); 2.25 (1H,d,J=16.0 Hz), 2.82(1H,d,J=17.0 Hz); 2.86(1H,d,J=16.0 Hz), 3.07(1H,dd,J=5.0,17.0 Hz) 8.65-3.75(1H,m); 5.14(1H, q,J=7.0 Hz), 5.50(1H,d,J=5.0 Hz); 6.34(1H,d,J=8.5 Hz), 7.09(1H,d,J=9.5 Hz); 7.40(1H,dd,J=5.0,8.0 Hz), 8.28(1H,dt, J=2.0,8.0 Hz), 8.47(1H,s); 8.70-8.72(1H,m), 8.94-8.96(1H, m), MS: 331(M+), 316(base), 226; HRMS calculated value: molecular formula C22H21N2O(M+): 331.16846; experimental value: 331.16888
Derivative No. 45
NMR(CDCL3) 1.64(3H,d,J=7.0 Hz), 1.65(3H,s); 2.25 (1H,d,J=16.0 Hz), 2.82(1H,d,J=16.5 Hz); 2.86(1H,d,J=16.0 Hz), 3.07(1H,dd,J=5.0,16.5 Hz),3.61-3.70(1H,m); 5.10(1H, q,J=7.0 Hz), 5.50(1H,d,J=4.0 Hz); 6.39(1H,d,J=9.5 Hz), 7.05(1H,d,J=9.5 Hz); 7.11-7.73(2H,m), 8.41(1H,s), 8.73-8.79(2H,m), MS: 331(M+), 316(base), 226; HRMS calculated value: molecular formula C22H21N3O(M+): 331.16846; experimental value: 331.18836
Derivative No. 46
NMR(CDCL3) 1.64(3H,d,J=6.5 Hz), 1.65(3H,s); 2.24 (1H,d,J=16.0 Hz), 2.82(1H,d,J=16.5 Hz); 2.85(1H,d,J=16.0 Hz), 3.07(1H,dd,J=5.0,16.5 Hz),3.67-3.70(1H,m); 3.96(6H, s), 5.22(1H,q,J=6.5 Hz), 5.30(1H,s); 5.47-5.49(1H,m), 6.39 (1H,d,J=9.5 Hz), 7.12(1H,s), 7.15(1H,d,J=9.5 Hz), 8.29(1H, s); MS: 406(M−,base), 391, 228, 167 HRMS calculated value: molecular formula C24H26N2O4(M+): 406.18926; experimental value: 406.18949
Claims (25)
2. A method of treating myasthenia amyasthenia by inhibiting cholinesterase comprising administering a huperzine A compound according to claim 1 10to a patient in need of such treatment in an amount effective to inhibit cholinesterase.
3. A method of treating dysmnesia, caused by a failure in central cholinergic system by inhibiting cholinesterase comprising administering a huperzine A compound according to claim 1 10to a patient in need of such treatment in an amount effective to inhibit cholinesterase.
4. A method of inhibiting cholinesterase comprising administering a huperzine A compound according to claim 1 10in an amount effective to inhibit cholinesterase to a patient in need of such treatment.
5. A method of inhibiting cholinesterase according to claim 4 , wherein said cholinesterase is an acetylcholinesterase.
6. A method of inhibiting cholinesterase according to claim 4 , wherein the cholinesterase is butyrylcholinesterase.
7. A method of improving memory impairment comprising administering a huperzine A compound according to claim 1 10in an amount effective to improve mnemory imnpairmientmemory impairment to a patient in need of such treatment.
8. A compound according to claim 1 10, wherein R″ and Y are ═CH, prepared by a process of condensing huperzine A and the corresponding substituted aldehyde in an anhydrous solvent.
9. A method of inhibiting cholinesterase according to claim 4 , wherein said huperzine A compound exhibits weaker inhibition effect on butyrylcholinesterase than huperzine A.
wherein
Y and —R″ together form a ═CH group;
wherein X is selected from the group consisting of hydrogen, C
1
-C
5
lower alkoxy, nitro, halogen, hydroxymethyl, hydroxy, amino substituted by two C
1
-C
5
lower alkyls and —CH═CH—G,
wherein G is phenyl or furanyl; and
R′ is hydrogen.
14. A method of treating amyasthenia by inhibiting cholinesterase comprising administering a huperzine A compound according to claim 11 to a patient in need of such treatment in an amount effective to inhibit cholinesterase.
15. A method of treating amyasthenia by inhibiting cholinesterase comprising administering a huperzine A compound according to claim 12 to a patient in need of such treatment in an amount effective to inhibit cholinesterase.
16. A method of treating amyasthenia by inhibiting cholinesterase comprising administering a huperzine A compound according to claim 13 to a patient in need of such treatment in an amount effective to inhibit cholinesterase.
17. A method of treating dysmnesia caused by a failure in central cholinergic system by inhibiting cholinesterase comprising administering a huperzine A compound according to claim 11 to a patient in need of such treatment in an amount effective to inhibit cholinesterase.
18. A method of treatment dysmnesia caused by a failure in central cholinergic system by inhibiting cholinesterase comprising administering a huperzine A compound according to claim 12 to a patient in need of such treatment in an amount effective to inhibit cholinesterase.
19. A method of treating dysmnesia caused by a failure in central cholinergic system by inhibiting cholinesterase comprising administering a huperzine A compound according to claim 13 to a patient in need of such treatment in an amount effective to inhibit cholinesterase.
20. A method of inhibiting cholinesterase comprising administering a huperzine A compound according to claim 11 in an amount effective to inhibit colinesterase to a patient in need of such treatment.
21. A method of inhibiting cholinesterase comprising administering a huperzine A compound according to claim 12 in an amount effective to inhibit colinesterase to a patient in need of such treatment.
22. A method of inhibiting cholinesterase comprising administering a huperzine A compound according to claim 13 in an amount effective to inhibit colinesterase to a patient in need of such treatment.
23. A method of improving memory impairment comprising administering a huperzine A compound according to claim 11 in an amount effective to improve memory impairment to a patient in need of such treatment.
24. A method of improving memory impairment comprising administering a huperzine A compound according to claim 12 in an amount effective to improve memory impairment to a patient in need of such treatment.
25. A method of improving memory impairment comprising administering a huperzine A compound according to claim 13 in an amount effective to improve memory impairment to a patient in need of such treatment.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US09/799,127 USRE38460E1 (en) | 1994-12-28 | 1995-12-26 | Huperzine A derivatives, their preparation and their use |
Applications Claiming Priority (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN94114057A CN1125725A (en) | 1994-12-28 | 1994-12-28 | First-kind "Haikelin" alkali derivant and its usage |
| CN94114057 | 1994-12-28 | ||
| PCT/CN1995/000100 WO1996020176A1 (en) | 1994-12-28 | 1995-12-26 | Huperzine a derivatives, their preparation and their use |
| US08/860,524 US5929084A (en) | 1994-12-28 | 1995-12-26 | Huperzine A derivatives, their preparation and their use |
| US09/799,127 USRE38460E1 (en) | 1994-12-28 | 1995-12-26 | Huperzine A derivatives, their preparation and their use |
Related Parent Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US08/860,524 Reissue US5929084A (en) | 1994-12-28 | 1995-12-26 | Huperzine A derivatives, their preparation and their use |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| USRE38460E1 true USRE38460E1 (en) | 2004-03-09 |
Family
ID=31891686
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US09/799,127 Expired - Fee Related USRE38460E1 (en) | 1994-12-28 | 1995-12-26 | Huperzine A derivatives, their preparation and their use |
Country Status (1)
| Country | Link |
|---|---|
| US (1) | USRE38460E1 (en) |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20060264455A1 (en) * | 2005-05-23 | 2006-11-23 | Schachter Steven C | Use of huperzine for disorders |
| US20070270454A1 (en) * | 2006-05-17 | 2007-11-22 | Industrial Technology Research Institute | Huperzine a compound for treatment of Alzheimer's disease |
| US20110098265A1 (en) * | 2009-10-28 | 2011-04-28 | Neuroscience, Inc. | Methods for reducing cravings and impulses associated with addictive and compulsive behaviors |
| US20110224245A1 (en) * | 2008-09-04 | 2011-09-15 | President And Fellows Of Harvard College | Treatment Of Neurological Disorders Using Huperzine |
| US8053577B2 (en) * | 2006-07-05 | 2011-11-08 | Hesheng Zhang | Derivatives of 5,9-methanocycloocta[b]pyridin-2-(1H)-one, their preparation and use as analgesics |
| WO2012121863A1 (en) | 2011-03-04 | 2012-09-13 | Yale University | (--)- huperzine a processes and related compositions and methods of treatment |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4929731A (en) | 1989-02-21 | 1990-05-29 | University Of Pittsburgh | Method for the synthesis of huperzine A and analogs thereof and compounds useful therein |
| US4968672A (en) | 1987-01-02 | 1990-11-06 | The United States Of America As Represented By The Department Of Health And Human Services | Adenosine receptor prodrugs |
| US5104880A (en) | 1991-05-01 | 1992-04-14 | Mayo Foundation For Medical Education And Research | Huperzine a analogs as acetylcholinesterase inhibitors |
| US5177082A (en) | 1985-11-05 | 1993-01-05 | Yu Chao Mei | Huperzines and analogs |
| US5684018A (en) | 1994-12-13 | 1997-11-04 | Merck & Co., Inc. | Acyloxyisopropyl carbamates as prodrugs for amine drugs |
-
1995
- 1995-12-26 US US09/799,127 patent/USRE38460E1/en not_active Expired - Fee Related
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5177082A (en) | 1985-11-05 | 1993-01-05 | Yu Chao Mei | Huperzines and analogs |
| US4968672A (en) | 1987-01-02 | 1990-11-06 | The United States Of America As Represented By The Department Of Health And Human Services | Adenosine receptor prodrugs |
| US4929731A (en) | 1989-02-21 | 1990-05-29 | University Of Pittsburgh | Method for the synthesis of huperzine A and analogs thereof and compounds useful therein |
| US5104880A (en) | 1991-05-01 | 1992-04-14 | Mayo Foundation For Medical Education And Research | Huperzine a analogs as acetylcholinesterase inhibitors |
| US5684018A (en) | 1994-12-13 | 1997-11-04 | Merck & Co., Inc. | Acyloxyisopropyl carbamates as prodrugs for amine drugs |
Non-Patent Citations (5)
| Title |
|---|
| Kozikowski et al., "Synthesis of Huperzine A and its Analogues and their Anticholinesterase Activity", J. Org. Chem., 56:4636-4645 (1991). |
| Liu et al., Can. J. Chem, 64(4):837-839 (1986). |
| Qian et al., Tetrahedron Lett., 30(16):2089-90 (1989). |
| Tang et al., Acta Pharmacologica Sinica, 15(2):107-110 (1994) (Abstract Only). |
| Xiong et al., Acta Pharmacologica Sinica, 16(1):21-25 (1995). |
Cited By (13)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8193212B2 (en) | 2005-05-23 | 2012-06-05 | President And Fellows Of Harvard College | Use of huperzine for neuropathic pain |
| US20060264454A1 (en) * | 2005-05-23 | 2006-11-23 | Schachter Steven C | Use of huperzine for neuropathic pain |
| US20100234416A1 (en) * | 2005-05-23 | 2010-09-16 | Schachter Steven C | Use of huperzine for disorders |
| US8093262B2 (en) | 2005-05-23 | 2012-01-10 | President And Fellows Of Harvard College | Use of huperzine for disorders |
| US20060264455A1 (en) * | 2005-05-23 | 2006-11-23 | Schachter Steven C | Use of huperzine for disorders |
| US8822492B2 (en) | 2005-05-23 | 2014-09-02 | President And Fellows Of Harvard College | Use of huperzine for disorders |
| US20070270454A1 (en) * | 2006-05-17 | 2007-11-22 | Industrial Technology Research Institute | Huperzine a compound for treatment of Alzheimer's disease |
| US8097726B2 (en) | 2006-05-17 | 2012-01-17 | Industrial Technology Research Institute | Huperzine a compound for treatment of alzheimer's disease |
| US8053577B2 (en) * | 2006-07-05 | 2011-11-08 | Hesheng Zhang | Derivatives of 5,9-methanocycloocta[b]pyridin-2-(1H)-one, their preparation and use as analgesics |
| US20110224245A1 (en) * | 2008-09-04 | 2011-09-15 | President And Fellows Of Harvard College | Treatment Of Neurological Disorders Using Huperzine |
| US20110098265A1 (en) * | 2009-10-28 | 2011-04-28 | Neuroscience, Inc. | Methods for reducing cravings and impulses associated with addictive and compulsive behaviors |
| WO2012121863A1 (en) | 2011-03-04 | 2012-09-13 | Yale University | (--)- huperzine a processes and related compositions and methods of treatment |
| EP3434669A1 (en) | 2011-03-04 | 2019-01-30 | Yale University | Intermediate compounds utilizable in synthesis of (-) huperzine and process for obtain one of these intermediate compounds |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US5929084A (en) | Huperzine A derivatives, their preparation and their use | |
| EP1709047B1 (en) | Azabenzofuran substituted thioureas as inhibitors of viral replication | |
| EP0181793B1 (en) | Piperidine derivatives, their preparation and their therapeutical application | |
| Stillings et al. | . alpha.-Adrenoreceptor reagents. 3. Synthesis of some 2-substituted 1, 4-benzodioxans as selective presynaptic. alpha. 2-adrenoreceptor antagonists | |
| KR20090079257A (en) | Substituted 3-isobutyl-9,10-dimethoxy-1,3,4,6,7,11b-hexahydro-2h-pyrido[2,1-a]isoquinolin-2-ol compounds and methods relating thereto | |
| EP0606366B1 (en) | Substituted phenserines and phenylcarbamates of (-)-eseroline, (-)-n1-noreseroline, and (-)-n1-benzylnoreseroline; as specific inhibitors of acetylcholinesterase | |
| SK20199A3 (en) | Ether muscarinic antagonists | |
| US5171750A (en) | Substituted phenserines as specific inhibitors of acetylcholinesterase | |
| US5721267A (en) | Chemotherapeutic pyrrolocarbazole derivatives | |
| RU2103271C1 (en) | Carbamic acid derivatives and their pharmaceutically acceptable acid-additive salts, method of their synthesis (variants) and pharmaceutical composition | |
| USRE38460E1 (en) | Huperzine A derivatives, their preparation and their use | |
| JP4564713B2 (en) | Nitrogen heterocyclic compounds, and methods for making nitrogen heterocyclic compounds and intermediates thereof | |
| KR100195421B1 (en) | Derivatives of 6-aminooctahydroindolizinetriol | |
| US7112593B2 (en) | N-aryl-substituted cyclic amine derivative and medicine containing the same as active ingredient | |
| FI98459C (en) | Process for the preparation of therapeutically useful 5-ethano-2,3,4,5-1H-2-benzazepin-7-ol carbamate derivatives | |
| FR2623808A1 (en) | NOVEL SUBSTITUTED FLAVONOID (BENZYL-4 PIPERAZINYL-1) -2 OXO-2 ETHYLENE DERIVATIVES, PROCESSES FOR PREPARING THEM AND PHARMACEUTICAL COMPOSITIONS CONTAINING SAME | |
| US4970214A (en) | Quinoline substituted oxomethyl or thioxomethyl glycine derivatives and aldose reductase inhibition therewith | |
| US6187783B1 (en) | Phenanthridinium derivatives | |
| JPH0517471A (en) | Furan derivative | |
| FR2498183A1 (en) | NOVEL GUANIDINOCYCLOHEXANECARBOXYLIC ACID DERIVATIVES, PROCESS FOR THEIR PREPARATION AND THERAPEUTIC USE THEREOF | |
| CZ282419B6 (en) | BENZO(c)PHENANTHRIDINE DERIVATIVES, PROCESS OF THEIR PREPARATION AND PHARMACEUTICAL COMPOSITION CONTAINING THEREOF | |
| FR2783521A1 (en) | New thrombin-inhibiting N-arylsulfonyl arginine derivatives useful as antithrombotic agents | |
| FR2761065A1 (en) | N- (ARGINYL) BENZENESULFONAMIDE DERIVATIVES, THEIR PREPARATION AND THEIR THERAPEUTIC USE | |
| KR100427127B1 (en) | NEW BENZO[3,4]CYCLOBUTA[1,2-c]PYRROLE COMPOUNDS, A PROCESS FOR THEIR PREPARATION AND PHARMACEUTICAL COMPOSITIONS CONTAINING THEM | |
| JPH0227344B2 (en) | SURUHONIRUPIPERAJINJUDOTAI |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| FPAY | Fee payment |
Year of fee payment: 8 |
|
| FEPP | Fee payment procedure |
Free format text: PAYOR NUMBER ASSIGNED (ORIGINAL EVENT CODE: ASPN); ENTITY STATUS OF PATENT OWNER: LARGE ENTITY |
|
| REMI | Maintenance fee reminder mailed | ||
| LAPS | Lapse for failure to pay maintenance fees |