USH1074H - Bacterio-electric leaching of metals - Google Patents
Bacterio-electric leaching of metals Download PDFInfo
- Publication number
- USH1074H USH1074H US07/486,039 US48603990A USH1074H US H1074 H USH1074 H US H1074H US 48603990 A US48603990 A US 48603990A US H1074 H USH1074 H US H1074H
- Authority
- US
- United States
- Prior art keywords
- iron
- metals
- slurry
- coal
- cathode
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Abandoned
Links
- 229910052751 metal Inorganic materials 0.000 title claims abstract description 59
- 239000002184 metal Substances 0.000 title claims abstract description 59
- 150000002739 metals Chemical class 0.000 title claims description 40
- 238000002386 leaching Methods 0.000 title description 15
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 claims abstract description 75
- 238000000034 method Methods 0.000 claims abstract description 49
- 241000894006 Bacteria Species 0.000 claims abstract description 20
- 239000003245 coal Substances 0.000 claims description 23
- 229910052683 pyrite Inorganic materials 0.000 claims description 23
- VTLYFUHAOXGGBS-UHFFFAOYSA-N Fe3+ Chemical compound [Fe+3] VTLYFUHAOXGGBS-UHFFFAOYSA-N 0.000 claims description 20
- 239000002002 slurry Substances 0.000 claims description 16
- CWYNVVGOOAEACU-UHFFFAOYSA-N Fe2+ Chemical compound [Fe+2] CWYNVVGOOAEACU-UHFFFAOYSA-N 0.000 claims description 12
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 claims description 12
- 229910001448 ferrous ion Inorganic materials 0.000 claims description 12
- 244000005700 microbiome Species 0.000 claims description 11
- 241000605222 Acidithiobacillus ferrooxidans Species 0.000 claims description 10
- QAOWNCQODCNURD-UHFFFAOYSA-L Sulfate Chemical compound [O-]S([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-L 0.000 claims description 6
- 230000002378 acidificating effect Effects 0.000 claims description 6
- 239000002803 fossil fuel Substances 0.000 claims description 6
- 238000002485 combustion reaction Methods 0.000 claims description 3
- 150000003467 sulfuric acid derivatives Chemical class 0.000 claims description 3
- 229910052782 aluminium Inorganic materials 0.000 claims description 2
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 claims description 2
- 230000001939 inductive effect Effects 0.000 claims description 2
- 239000000203 mixture Substances 0.000 claims description 2
- GCICAPWZNUIIDV-UHFFFAOYSA-N lithium magnesium Chemical compound [Li].[Mg] GCICAPWZNUIIDV-UHFFFAOYSA-N 0.000 claims 1
- 230000008569 process Effects 0.000 abstract description 41
- 229910052742 iron Inorganic materials 0.000 abstract description 35
- 239000005864 Sulphur Substances 0.000 abstract description 31
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 abstract description 27
- 230000001590 oxidative effect Effects 0.000 abstract description 14
- 238000000926 separation method Methods 0.000 abstract description 8
- -1 cationic metals Chemical class 0.000 abstract description 6
- 239000000463 material Substances 0.000 abstract description 4
- 230000005012 migration Effects 0.000 abstract description 4
- 238000013508 migration Methods 0.000 abstract description 4
- 125000002091 cationic group Chemical group 0.000 abstract 2
- 238000007254 oxidation reaction Methods 0.000 description 21
- NIFIFKQPDTWWGU-UHFFFAOYSA-N pyrite Chemical compound [Fe+2].[S-][S-] NIFIFKQPDTWWGU-UHFFFAOYSA-N 0.000 description 20
- 239000011028 pyrite Substances 0.000 description 20
- 230000003647 oxidation Effects 0.000 description 18
- 230000001580 bacterial effect Effects 0.000 description 14
- 229910052500 inorganic mineral Inorganic materials 0.000 description 13
- 239000011707 mineral Substances 0.000 description 13
- 239000013049 sediment Substances 0.000 description 13
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Chemical compound [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 description 10
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 9
- 150000001768 cations Chemical class 0.000 description 9
- 239000010949 copper Substances 0.000 description 9
- 229910052802 copper Inorganic materials 0.000 description 8
- 239000002253 acid Substances 0.000 description 7
- 238000011084 recovery Methods 0.000 description 7
- PXHVJJICTQNCMI-UHFFFAOYSA-N Nickel Chemical compound [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 description 6
- 238000007792 addition Methods 0.000 description 6
- 238000006243 chemical reaction Methods 0.000 description 6
- 238000000605 extraction Methods 0.000 description 6
- 239000012528 membrane Substances 0.000 description 6
- UQSXHKLRYXJYBZ-UHFFFAOYSA-N iron oxide Inorganic materials [Fe]=O UQSXHKLRYXJYBZ-UHFFFAOYSA-N 0.000 description 5
- 239000007800 oxidant agent Substances 0.000 description 5
- 239000002245 particle Substances 0.000 description 5
- 229910052697 platinum Inorganic materials 0.000 description 5
- 238000001556 precipitation Methods 0.000 description 5
- 230000007928 solubilization Effects 0.000 description 5
- 238000005063 solubilization Methods 0.000 description 5
- 229910003556 H2 SO4 Inorganic materials 0.000 description 4
- 229920002301 cellulose acetate Polymers 0.000 description 4
- 238000004090 dissolution Methods 0.000 description 4
- 230000000694 effects Effects 0.000 description 4
- 229910001447 ferric ion Inorganic materials 0.000 description 4
- RUTXIHLAWFEWGM-UHFFFAOYSA-H iron(3+) sulfate Chemical compound [Fe+3].[Fe+3].[O-]S([O-])(=O)=O.[O-]S([O-])(=O)=O.[O-]S([O-])(=O)=O RUTXIHLAWFEWGM-UHFFFAOYSA-H 0.000 description 4
- 229910000360 iron(III) sulfate Inorganic materials 0.000 description 4
- 230000001172 regenerating effect Effects 0.000 description 4
- 239000007787 solid Substances 0.000 description 4
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical compound [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 description 3
- 239000003795 chemical substances by application Substances 0.000 description 3
- 229910052935 jarosite Inorganic materials 0.000 description 3
- 239000011777 magnesium Substances 0.000 description 3
- 239000011159 matrix material Substances 0.000 description 3
- 229910052759 nickel Inorganic materials 0.000 description 3
- 230000002829 reductive effect Effects 0.000 description 3
- 229910052725 zinc Inorganic materials 0.000 description 3
- 239000011701 zinc Substances 0.000 description 3
- VYZAMTAEIAYCRO-UHFFFAOYSA-N Chromium Chemical compound [Cr] VYZAMTAEIAYCRO-UHFFFAOYSA-N 0.000 description 2
- 241000605118 Thiobacillus Species 0.000 description 2
- 230000001133 acceleration Effects 0.000 description 2
- 230000015572 biosynthetic process Effects 0.000 description 2
- 229920002678 cellulose Polymers 0.000 description 2
- 239000001913 cellulose Substances 0.000 description 2
- 238000001311 chemical methods and process Methods 0.000 description 2
- 229910052804 chromium Inorganic materials 0.000 description 2
- 239000011651 chromium Substances 0.000 description 2
- 238000009826 distribution Methods 0.000 description 2
- 238000006056 electrooxidation reaction Methods 0.000 description 2
- 231100001261 hazardous Toxicity 0.000 description 2
- 235000013980 iron oxide Nutrition 0.000 description 2
- BAUYGSIQEAFULO-UHFFFAOYSA-L iron(2+) sulfate (anhydrous) Chemical compound [Fe+2].[O-]S([O-])(=O)=O BAUYGSIQEAFULO-UHFFFAOYSA-L 0.000 description 2
- VBMVTYDPPZVILR-UHFFFAOYSA-N iron(2+);oxygen(2-) Chemical class [O-2].[Fe+2] VBMVTYDPPZVILR-UHFFFAOYSA-N 0.000 description 2
- 229910000359 iron(II) sulfate Inorganic materials 0.000 description 2
- 230000028161 membrane depolarization Effects 0.000 description 2
- 239000004058 oil shale Substances 0.000 description 2
- 239000010970 precious metal Substances 0.000 description 2
- 239000000047 product Substances 0.000 description 2
- 230000009467 reduction Effects 0.000 description 2
- 238000007670 refining Methods 0.000 description 2
- 230000000717 retained effect Effects 0.000 description 2
- 239000003476 subbituminous coal Substances 0.000 description 2
- 239000000758 substrate Substances 0.000 description 2
- 229910052569 sulfide mineral Inorganic materials 0.000 description 2
- 241000605272 Acidithiobacillus thiooxidans Species 0.000 description 1
- 229910002703 Al K Inorganic materials 0.000 description 1
- BVKZGUZCCUSVTD-UHFFFAOYSA-L Carbonate Chemical compound [O-]C([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-L 0.000 description 1
- 229910017518 Cu Zn Inorganic materials 0.000 description 1
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 description 1
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 1
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical compound [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 description 1
- 229910000831 Steel Inorganic materials 0.000 description 1
- 238000009825 accumulation Methods 0.000 description 1
- 238000003916 acid precipitation Methods 0.000 description 1
- 238000004458 analytical method Methods 0.000 description 1
- 150000001450 anions Chemical class 0.000 description 1
- 239000002956 ash Substances 0.000 description 1
- 239000006227 byproduct Substances 0.000 description 1
- 239000000470 constituent Substances 0.000 description 1
- 238000011109 contamination Methods 0.000 description 1
- 229910001779 copper mineral Inorganic materials 0.000 description 1
- 238000005260 corrosion Methods 0.000 description 1
- 230000007797 corrosion Effects 0.000 description 1
- 238000000354 decomposition reaction Methods 0.000 description 1
- 238000000151 deposition Methods 0.000 description 1
- 230000008021 deposition Effects 0.000 description 1
- 238000006477 desulfuration reaction Methods 0.000 description 1
- 230000023556 desulfurization Effects 0.000 description 1
- 230000003009 desulfurizing effect Effects 0.000 description 1
- 239000000428 dust Substances 0.000 description 1
- 230000005684 electric field Effects 0.000 description 1
- 238000005868 electrolysis reaction Methods 0.000 description 1
- 238000000921 elemental analysis Methods 0.000 description 1
- 230000007613 environmental effect Effects 0.000 description 1
- 238000001704 evaporation Methods 0.000 description 1
- 230000008020 evaporation Effects 0.000 description 1
- 239000010881 fly ash Substances 0.000 description 1
- 238000009291 froth flotation Methods 0.000 description 1
- 239000011521 glass Substances 0.000 description 1
- 229910052598 goethite Inorganic materials 0.000 description 1
- PCHJSUWPFVWCPO-UHFFFAOYSA-N gold Chemical compound [Au] PCHJSUWPFVWCPO-UHFFFAOYSA-N 0.000 description 1
- 229910052737 gold Inorganic materials 0.000 description 1
- 239000010931 gold Substances 0.000 description 1
- BHEPBYXIRTUNPN-UHFFFAOYSA-N hydridophosphorus(.) (triplet) Chemical compound [PH] BHEPBYXIRTUNPN-UHFFFAOYSA-N 0.000 description 1
- 229910052739 hydrogen Inorganic materials 0.000 description 1
- 239000001257 hydrogen Substances 0.000 description 1
- AEIXRCIKZIZYPM-UHFFFAOYSA-M hydroxy(oxo)iron Chemical compound [O][Fe]O AEIXRCIKZIZYPM-UHFFFAOYSA-M 0.000 description 1
- 238000011065 in-situ storage Methods 0.000 description 1
- 229920000592 inorganic polymer Polymers 0.000 description 1
- SZVJSHCCFOBDDC-UHFFFAOYSA-N iron(II,III) oxide Inorganic materials O=[Fe]O[Fe]O[Fe]=O SZVJSHCCFOBDDC-UHFFFAOYSA-N 0.000 description 1
- 229910021519 iron(III) oxide-hydroxide Inorganic materials 0.000 description 1
- 238000002955 isolation Methods 0.000 description 1
- 229910052744 lithium Inorganic materials 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- 230000014759 maintenance of location Effects 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 230000007246 mechanism Effects 0.000 description 1
- 230000002503 metabolic effect Effects 0.000 description 1
- 230000004060 metabolic process Effects 0.000 description 1
- 229910052976 metal sulfide Inorganic materials 0.000 description 1
- 150000001457 metallic cations Chemical class 0.000 description 1
- 150000001455 metallic ions Chemical class 0.000 description 1
- 238000001465 metallisation Methods 0.000 description 1
- 230000007935 neutral effect Effects 0.000 description 1
- 239000012188 paraffin wax Substances 0.000 description 1
- 229920000867 polyelectrolyte Polymers 0.000 description 1
- 150000003839 salts Chemical class 0.000 description 1
- 238000007790 scraping Methods 0.000 description 1
- 229910052710 silicon Inorganic materials 0.000 description 1
- 239000010703 silicon Substances 0.000 description 1
- 239000002689 soil Substances 0.000 description 1
- 241000894007 species Species 0.000 description 1
- 229910001220 stainless steel Inorganic materials 0.000 description 1
- 239000010935 stainless steel Substances 0.000 description 1
- 239000010959 steel Substances 0.000 description 1
- 230000004936 stimulating effect Effects 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- 150000004763 sulfides Chemical class 0.000 description 1
- 229910001656 zinc mineral Inorganic materials 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22B—PRODUCTION AND REFINING OF METALS; PRETREATMENT OF RAW MATERIALS
- C22B3/00—Extraction of metal compounds from ores or concentrates by wet processes
- C22B3/18—Extraction of metal compounds from ores or concentrates by wet processes with the aid of microorganisms or enzymes, e.g. bacteria or algae
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02P—CLIMATE CHANGE MITIGATION TECHNOLOGIES IN THE PRODUCTION OR PROCESSING OF GOODS
- Y02P10/00—Technologies related to metal processing
- Y02P10/20—Recycling
Definitions
- the present invention relates to the biological beneficiation of an ore body or fossil fuel utilizing an applied electrical field in conjunction with living microorganisms and/or their metabolic by-products to effect the release of metallic ions or mineral components from the ore body.
- Coal desulfurization is highly desirable in that it is believed that combustion of coal, and specifically high-sulphur content coals, is a contributing factor to acid rain and other environmental problems. Numerous methods for desulfurizing coal have been attempted. Physical separation, bacterial oxidation and chemical processes can all be demonstrated to produce the desired effect in laboratory or pilot plant scale tests. However, each of these processes suffers because they are either too expensive or too difficult to accomplish on the massive scale involved. Physical separation processes are difficult because of the small particle size necessary. Bacterial oxidation suffers because of the long exposure times required (on the order of 5-15 days). Chemical processes suffer from the expense of the large quantity of chemicals required.
- U.S. Pat. No. 4,775,627 discloses a process whereby pyrite particles and high-sulphur content coal are modified so as to be more hydrophyllic, and more easily separated by conventional means, such as froth flotation. Ground coal particles are preconditioned by subjecting the pyrite to thiophilic bacteria adapted to the process, permitting the bacteria to alter the hydrophobicity of the pyrite particles.
- U.S. Pat. No. 4,822,413 discloses a process to extract metals from an ore containing one or more metallic sulphides using a leach liquor containing bacterially-generated ferric ions in order to effect metal sulfide dissolution.
- the metals removed are capable of dissolution in an acidic ferric sulfate solution (such as copper) or are more easily removed by subsequent processing (such as gold) as a result of exposure to the acidic ferric sulfate solution.
- Suitable bacteria such as Thiobacillus ferrooxidans may be used to increase dissolution of metals in certain circumstances.
- the phenomenon of bacterio-electric leaching of metals from minerals combines two processes having a common feature--the bacterial oxidation of Fe +2 in an acid solution.
- the first process is the oxidative leaching of pyritic minerals by iron-oxidizing bacteria (most commonly thiobacilli)--the so-called "pyrite cycle”.
- the second process is the acceleration of the oxidation of reduced iron by iron-oxidizing thiobacilli by subjecting the process to an applied electric potential and the resulting current.
- the pyrite cycle is a chain reaction in which ferric ions produced by bacterial oxidation react with the sulphur of a pyritic mineral in order to oxidize the pyrite. This oxidation releases ferrous ions, enabling the chemo-lithotrophic oxidation of the pyrite, and sustains the pyrite decomposition by regenerating Fe +3 as the oxidizing agent. Pyrite oxidation and solubilization are limited by the concentration of ferric ions, and their accessibility to the mineral substrate. Both the concentration and accessibility of Fe +3 ions are affected by the precipitation of bacterially-oxidized iron from solution as an Fe +3 sulfato complex.
- Precipitation usually occurs initially in the form of a metastable amorphous hydrated ferric sulfate. As illustrated in FIG. 1, the presence of jarosite directing cations and excess sulfate causes the Fe +3 sulfato complex to be converted to a stable crystalline jarosite. Under more alkaline conditions the deposits comprise iron oxides and oxyhydroxides that are distinguishable from bacterial oxidation products.
- Iron oxidizing bacteria are essential to the pyrite cycle by regenerating the soluble Fe +3 lixiviant under acid conditions permitting extraction of metals from sulfide minerals and coal--conditions under which abiotic auto-oxidation of iron occurs slowly.
- the acceleration of electro-oxidation of reduced iron by iron oxidizing thiobacilli occurs when an electro-oxidizable metal mineral liberates Fe +2 under the influence of an electric potential.
- FIG. 2 in the presence of iron-oxidizing thiobacilli and in an acid environment, the flow of electric current and solubilization of iron is accelerated through depolarization of cathodic sites by bacterially oxidized iron in solution.
- Fe 0 corrodes in an acidic aqueous environment
- Fe +2 passes into solution as electrons are transferred from Fe 0 at anodic sites to cathodic sites on the metal.
- the cathodic sites must be depolarized by a loss of electrons to an oxidizing agent in order for the process to continue.
- hydrogen ions may serve as the oxidizing agent, however when Fe +3 is present in such environment, it competes at the cathodes with H + for available electrons. If the Fe +2 produced is oxidized by the thiobacillus, a chain reaction of iron oxidation and reduction occurs analogous to that of the pyrite cycle.
- Pyrite is electrically conductive, therefore by subjecting pyritic materials to an applied potential in a galvanic cell containing an iron oxidizing microorganism, under acid conditions, solubilization of the pyritic component and liberation of associated cations from the mineral matrix will be enhanced by the combination of electro- and bacterial-oxidation. Electrophoretic migration of cations to the cathode in a galvanic cell may be utilized as a mechanism to separate and recover the cations.
- the process of the present invention may be utilized to separate valuable metals from low-grade ores or to remove ash metals and sulphur from pyrite coals.
- the method of the present invention comprises adding iron-oxidizing bacteria and/or sulphur oxidizing bacteria to a slurry containing an ore body, inducing a voltage between a pair of electrodes across the slurry, and recovering metals from the ore body at one of the cathodes.
- comminuted ores are mixed in a vessel with a lixiviant, such as pH 2.5 H 2 SO 4 , and a pair of platinum electrodes are suspended within the vessel. While the voltage required to produce the desired effect may depend upon various process conditions, application of a voltage of between about 1 and about 25 volts has been found to produce the electrophoretic separation of metals desired.
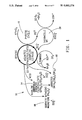
- FIG. 1 is a diagramatic representation of the relationship of sulfate to pyrite leaching and the formation of oxidized iron sediments;
- FIG. 2 is a diagramatic representation of the corrosion of steel by iron oxidizing bacteria
- FIG. 3 is a schematic representation of the bacterioelectric leaching apparatus of the present invention.
- FIG. 4 is a graphic representation of metal deposits on the cathode of the apparatus of FIG. 3;
- FIG. 5 is a graphic representation of galvanic separation of metals from a copper ore
- FIG. 6 is a schematic representation of a galvanic sandwich
- FIG. 7 is a schematic representation of metal deposition in concentric zones about the cathode of the device of FIG. 6;
- FIGS. 8(a and b) are graphic representation of metal recovery from coal in a galvanic sandwich of FIG. 6.
- FIGS. 1 and 2 illustrate schematically separate processes involved in the present invention.
- FIG. 1 represents the relationship of sulfate to pyrite leaching and the formation of oxidized iron sediments by iron oxidizing bacteria such as Thiobacillus ferrooxidans.
- Ferric ions produced by bacterial oxidation in the pyrite cycle 12 react with the sulphur of a pyritic mineral to oxidize it, releasing Fe +2 for the chemo-lithotrophic oxidation and maintains pyrite composition by regenerating the Fe +3 oxidant.
- Pyrite oxidation and solubilization are limited by the concentration of the ferric oxidizing agent and its accessibility to the mineral substrate.
- Precipitation usually occurs as an amorphous-hydrated ferric sulfate 14, however the presence of jarosite-directing cations and excess sulfate causes the Fe +3 sulfato complex to be converted into a stable jarosite 16. Under alkaline conditions iron oxides and oxyhydroxide 18 are formed. Iron oxidizing bacteria 20 are essential to the process by regenerating the soluble Fe +3 leaching agent which permit extraction of metals from sulfide minerals and coal. Without such bacteria the Fe +3 sulfato complex is converted into magnetite or goethite,
- FIG. 2 illustrates the electro-oxidation of a metal (iron) under the influence of an electric potential.
- the flow of electric current and solubilization of iron is accelerated through depolarization of cathodic sites 22 by bacterially-oxidized iron in an acid environment.
- Fe +2 26 passes into solution. If the Fe +2 is oxidized by the bacteria, a chain reaction of iron oxidation and reduction occurs and is self-sustaining, modulated by the precipitation of the typical sediments of chemo-lithotrophic iron oxidation.
- the reaction is driven by applying an external voltage to the system, thereby accelerating dissolution of the metal, and forming more Fe +2 for bacterial oxidation, which in turn causes an increased accumulation of the Fe +3 sediment.
- an apparatus illustrated in FIG. 2 involves, for instance, a metal (iron) coupon 28 to conduct the electric potential
- the process can be driven if an electrically-conductive ore body is suspended in an appropriate galvanic cell.
- the pyritic component of a mineral contained within a galvanic cell having an iron-oxidizing microorganism therein, under acid conditions may be solubilized by a combination of electro- and bacterial-oxidation, with a concurrent liberation of cations retained within the mineral matrix.
- Electrophoretic migration of cations to the cathode of the galvanic cell assists collection of the separated cationic metals. While the metallic cations of interest migrate to the cathode, their associated anion (usually sulfate) will likewise migrate.
- the metals may be separated by conventional means after being liberated from the ore body.
- the apparatus of FIG. 3 demonstrates the bacterioelectric extraction of metals from ore bodies.
- An appropriate container 30, such as a glass beaker, is filled with a leaching agent 40, in this case sulfuric acid at a pH of from about 0.8 to about 4.5. It has been found that in the apparatus of FIG. 3 a pH of about 2.5 is preferable.
- An anode 42 and cathode 44 are suspended within the lixiviant, with the electrodes retained within chambers 46, 48 respectively. Both electrodes in the examples set forth herein are constructed of platinum (the source of the platinum recovered and illustrated in FIG. 4. Powdered mineral specimens are compacted against the anode within chamber 46 and then wrapped in ashless filter paper 50.
- the liberated metals from the ore are collected on the cathode 44 or in the surrounding cathode chamber 48. It should be understood that the metals collected may be removed from solution at any point in their migration between the anode and cathode, as with filters.
- the leaching agent 40 is preferably agitated, as by an air-driven magnetic stirrer 52.
- the metals recovered on the cathode are illustrated in FIG. 4--the ordinant of this Figure is not delineated in definitive units of measure, as the graphic representation illustrates merely the relative quantities of the various metals recovered.
- FIG. 5 illustrates the quantities of five metals recovered in the apparatus of FIG. 3 with and without the addition of iron oxidizing thiobacilli and soluble iron.
- the apparatus was operated for 72 hours at 10 volts.
- One gram of powdered ore was pretreated by shaking for 2 days in 50 mls of pH 2.5 H 2 SO 4 , containing 5 ⁇ 10 10 T. ferrooxidans cells and 10 mg FeSO 4 7H 2 O solution. In each case, the amount recovered with such additions was greater than that without.
- PSOC 667 an Iowa subbituminous coal with approximately 13% total sulphur and 4.5% pyritic sulphur, was analyzed in the apparatus of FIG. 3. 0.75 grams of powdered coal was added to the anode chamber, with or without added microorganisms. The container was filled with 200 ml of pH 2.5 H 2 SO 4 and the apparatus operated at 10 volts for 64 hours. Sediments were collected separately from the cathode and main chambers. Solutes were obtained by evaporation of the filtered lixiviant.
- a galvanic sandwich illustrated in FIG. 6, comprises a container or dish 54, having a membrane filter 56 atop a cellulose pad 58.
- the pad 58 is soaked with a dilute sulfuric acid solution and either overlies or surrounds a stainless steel anode 60.
- the anode 60 is maintained in contact with the ore specimen 70 within the pad.
- approximately 0.3-0.4 gm of powdered ore was utilized.
- a platinum cathode 72 rests on the surface of the membrane filter 56 below a layer of paraffin 74 held in place by a lid 76 of container 54.
- the membrane filter is preferably comprised of cellulose acetate.
- the sulfuric acid is provided at a pH of from about 0.8 to about 4.5, preferably about 2.5, and electric current was provided for 20 hours at 10 volts.
- Electrophoretic separation of the metals present in the ore specimen are deposited in zones 78 concentric to the cathode 72 (FIG. 7).
- the various zones 78 may be cut from the membrane filter 56 and analyzed by appropriate techniques to determine relative amount of the various metals.
- the process of the present invention is useful to recover sulphur from high-sulphur coals, such as the PSOC 667 sample of Table 1.
- This coal contains approximately 13% total sulphur and 4-5% pyritic sulphur.
- the total amount of sulphur recovered using the bacterio-electric process of the present invention is approximately 20% greater than by the use of the galvanic process only.
- areas experiencing soil contamination from metals resulting from dumping may be cleansed by the process of the present invention.
- radionuclides and hazardous organics preferentially complexed with metals subject to removal by this process may be removed from contaminated areas.
- products of combustion such as fly ash and bag-house dust removed from industrial processes may be cleansed of specific metals by this process.
Landscapes
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Life Sciences & Earth Sciences (AREA)
- Geochemistry & Mineralogy (AREA)
- Biotechnology (AREA)
- Microbiology (AREA)
- Environmental & Geological Engineering (AREA)
- General Life Sciences & Earth Sciences (AREA)
- Biochemistry (AREA)
- Geology (AREA)
- Manufacturing & Machinery (AREA)
- Materials Engineering (AREA)
- Mechanical Engineering (AREA)
- Metallurgy (AREA)
- Organic Chemistry (AREA)
- Manufacture And Refinement Of Metals (AREA)
Abstract
The separation of cationic materials from an ore body is assisted by the application of an electric potential, and resulting current, to the ore body, in association with iron or sulphur oxidizing bacteria. The combined process induces migration of cationic metals to a cathode suspended within the ore body so that the cationic metal can be preferentially separated from the ore body.
Description
The United States Government has rights in this invention pursuant to Contract No. DE-AC07-76ID01570 between the United States Department of Energy and EG&G Idaho, Inc.
The present invention relates to the biological beneficiation of an ore body or fossil fuel utilizing an applied electrical field in conjunction with living microorganisms and/or their metabolic by-products to effect the release of metallic ions or mineral components from the ore body.
Processes for removing sulphur from coal, or for extracting desirable metals from an ore body, are well-known. Coal desulfurization is highly desirable in that it is believed that combustion of coal, and specifically high-sulphur content coals, is a contributing factor to acid rain and other environmental problems. Numerous methods for desulfurizing coal have been attempted. Physical separation, bacterial oxidation and chemical processes can all be demonstrated to produce the desired effect in laboratory or pilot plant scale tests. However, each of these processes suffers because they are either too expensive or too difficult to accomplish on the massive scale involved. Physical separation processes are difficult because of the small particle size necessary. Bacterial oxidation suffers because of the long exposure times required (on the order of 5-15 days). Chemical processes suffer from the expense of the large quantity of chemicals required.
For example, U.S. Pat. No. 4,775,627 discloses a process whereby pyrite particles and high-sulphur content coal are modified so as to be more hydrophyllic, and more easily separated by conventional means, such as froth flotation. Ground coal particles are preconditioned by subjecting the pyrite to thiophilic bacteria adapted to the process, permitting the bacteria to alter the hydrophobicity of the pyrite particles. U.S. Pat. No. 4,822,413 discloses a process to extract metals from an ore containing one or more metallic sulphides using a leach liquor containing bacterially-generated ferric ions in order to effect metal sulfide dissolution. The metals removed are capable of dissolution in an acidic ferric sulfate solution (such as copper) or are more easily removed by subsequent processing (such as gold) as a result of exposure to the acidic ferric sulfate solution. Suitable bacteria, such as Thiobacillus ferrooxidans may be used to increase dissolution of metals in certain circumstances.
It has been proposed in U.S. Pat. No. 4,043,884 to upgrade the kerogen components of oil shale by leaching carbonate materials from the oil shale to produce a porous residue, forming a slurry of the residue with a reductive electrolytically active solution, and then subjecting the slurry to reductive electrolysis. The reduced residue is then more easily separated from the electrolyzed slurry.
The phenomenon of bacterio-electric leaching of metals from minerals combines two processes having a common feature--the bacterial oxidation of Fe+2 in an acid solution. The first process is the oxidative leaching of pyritic minerals by iron-oxidizing bacteria (most commonly thiobacilli)--the so-called "pyrite cycle". The second process is the acceleration of the oxidation of reduced iron by iron-oxidizing thiobacilli by subjecting the process to an applied electric potential and the resulting current.
The pyrite cycle is a chain reaction in which ferric ions produced by bacterial oxidation react with the sulphur of a pyritic mineral in order to oxidize the pyrite. This oxidation releases ferrous ions, enabling the chemo-lithotrophic oxidation of the pyrite, and sustains the pyrite decomposition by regenerating Fe+3 as the oxidizing agent. Pyrite oxidation and solubilization are limited by the concentration of ferric ions, and their accessibility to the mineral substrate. Both the concentration and accessibility of Fe+3 ions are affected by the precipitation of bacterially-oxidized iron from solution as an Fe+3 sulfato complex.
Precipitation usually occurs initially in the form of a metastable amorphous hydrated ferric sulfate. As illustrated in FIG. 1, the presence of jarosite directing cations and excess sulfate causes the Fe+3 sulfato complex to be converted to a stable crystalline jarosite. Under more alkaline conditions the deposits comprise iron oxides and oxyhydroxides that are distinguishable from bacterial oxidation products.
Iron oxidizing bacteria are essential to the pyrite cycle by regenerating the soluble Fe+3 lixiviant under acid conditions permitting extraction of metals from sulfide minerals and coal--conditions under which abiotic auto-oxidation of iron occurs slowly.
The acceleration of electro-oxidation of reduced iron by iron oxidizing thiobacilli occurs when an electro-oxidizable metal mineral liberates Fe+2 under the influence of an electric potential. As illustrated in FIG. 2, in the presence of iron-oxidizing thiobacilli and in an acid environment, the flow of electric current and solubilization of iron is accelerated through depolarization of cathodic sites by bacterially oxidized iron in solution. When Fe0 corrodes in an acidic aqueous environment, Fe+2 passes into solution as electrons are transferred from Fe0 at anodic sites to cathodic sites on the metal. The cathodic sites must be depolarized by a loss of electrons to an oxidizing agent in order for the process to continue. In an acid environment, hydrogen ions may serve as the oxidizing agent, however when Fe+3 is present in such environment, it competes at the cathodes with H+ for available electrons. If the Fe+2 produced is oxidized by the thiobacillus, a chain reaction of iron oxidation and reduction occurs analogous to that of the pyrite cycle.
Pyrite is electrically conductive, therefore by subjecting pyritic materials to an applied potential in a galvanic cell containing an iron oxidizing microorganism, under acid conditions, solubilization of the pyritic component and liberation of associated cations from the mineral matrix will be enhanced by the combination of electro- and bacterial-oxidation. Electrophoretic migration of cations to the cathode in a galvanic cell may be utilized as a mechanism to separate and recover the cations. The process of the present invention may be utilized to separate valuable metals from low-grade ores or to remove ash metals and sulphur from pyrite coals.
The method of the present invention comprises adding iron-oxidizing bacteria and/or sulphur oxidizing bacteria to a slurry containing an ore body, inducing a voltage between a pair of electrodes across the slurry, and recovering metals from the ore body at one of the cathodes. Specifically, comminuted ores are mixed in a vessel with a lixiviant, such as pH 2.5 H2 SO4, and a pair of platinum electrodes are suspended within the vessel. While the voltage required to produce the desired effect may depend upon various process conditions, application of a voltage of between about 1 and about 25 volts has been found to produce the electrophoretic separation of metals desired. While there is no process limitation for a maximum voltage applied, cost considerations will limit the maximum voltage to a reasonably low level e.g., less than 100 volts. It has been found that the addition of various salts to the reaction components can substantially increase conductivity of the reaction matrix, and thereby increase the metal recovery. For example, the addition of sulfates of lithium, magnesium and aluminum will increase the conductivity.
FIG. 1 is a diagramatic representation of the relationship of sulfate to pyrite leaching and the formation of oxidized iron sediments;
FIG. 2 is a diagramatic representation of the corrosion of steel by iron oxidizing bacteria;
FIG. 3 is a schematic representation of the bacterioelectric leaching apparatus of the present invention;
FIG. 4 is a graphic representation of metal deposits on the cathode of the apparatus of FIG. 3;
FIG. 5 is a graphic representation of galvanic separation of metals from a copper ore;
FIG. 6 is a schematic representation of a galvanic sandwich;
FIG. 7 is a schematic representation of metal deposition in concentric zones about the cathode of the device of FIG. 6; and
FIGS. 8(a and b) are graphic representation of metal recovery from coal in a galvanic sandwich of FIG. 6.
FIGS. 1 and 2 illustrate schematically separate processes involved in the present invention. FIG. 1 represents the relationship of sulfate to pyrite leaching and the formation of oxidized iron sediments by iron oxidizing bacteria such as Thiobacillus ferrooxidans. Ferric ions produced by bacterial oxidation in the pyrite cycle 12 react with the sulphur of a pyritic mineral to oxidize it, releasing Fe+2 for the chemo-lithotrophic oxidation and maintains pyrite composition by regenerating the Fe+3 oxidant. Pyrite oxidation and solubilization are limited by the concentration of the ferric oxidizing agent and its accessibility to the mineral substrate. Precipitation usually occurs as an amorphous-hydrated ferric sulfate 14, however the presence of jarosite-directing cations and excess sulfate causes the Fe+3 sulfato complex to be converted into a stable jarosite 16. Under alkaline conditions iron oxides and oxyhydroxide 18 are formed. Iron oxidizing bacteria 20 are essential to the process by regenerating the soluble Fe+3 leaching agent which permit extraction of metals from sulfide minerals and coal. Without such bacteria the Fe+3 sulfato complex is converted into magnetite or goethite,
FIG. 2 illustrates the electro-oxidation of a metal (iron) under the influence of an electric potential. The flow of electric current and solubilization of iron is accelerated through depolarization of cathodic sites 22 by bacterially-oxidized iron in an acid environment. As electrons are transferred from Fe0 at an anode 24 to a cathode 22, Fe +2 26 passes into solution. If the Fe+2 is oxidized by the bacteria, a chain reaction of iron oxidation and reduction occurs and is self-sustaining, modulated by the precipitation of the typical sediments of chemo-lithotrophic iron oxidation. The reaction is driven by applying an external voltage to the system, thereby accelerating dissolution of the metal, and forming more Fe+2 for bacterial oxidation, which in turn causes an increased accumulation of the Fe+3 sediment. While an apparatus illustrated in FIG. 2 involves, for instance, a metal (iron) coupon 28 to conduct the electric potential, the process can be driven if an electrically-conductive ore body is suspended in an appropriate galvanic cell. For example, because pyrite is electrically conductive, the pyritic component of a mineral contained within a galvanic cell having an iron-oxidizing microorganism therein, under acid conditions, may be solubilized by a combination of electro- and bacterial-oxidation, with a concurrent liberation of cations retained within the mineral matrix. Electrophoretic migration of cations to the cathode of the galvanic cell assists collection of the separated cationic metals. While the metallic cations of interest migrate to the cathode, their associated anion (usually sulfate) will likewise migrate. The metals may be separated by conventional means after being liberated from the ore body.
The apparatus of FIG. 3 demonstrates the bacterioelectric extraction of metals from ore bodies. An appropriate container 30, such as a glass beaker, is filled with a leaching agent 40, in this case sulfuric acid at a pH of from about 0.8 to about 4.5. It has been found that in the apparatus of FIG. 3 a pH of about 2.5 is preferable. An anode 42 and cathode 44 are suspended within the lixiviant, with the electrodes retained within chambers 46, 48 respectively. Both electrodes in the examples set forth herein are constructed of platinum (the source of the platinum recovered and illustrated in FIG. 4. Powdered mineral specimens are compacted against the anode within chamber 46 and then wrapped in ashless filter paper 50. The liberated metals from the ore are collected on the cathode 44 or in the surrounding cathode chamber 48. It should be understood that the metals collected may be removed from solution at any point in their migration between the anode and cathode, as with filters. The leaching agent 40 is preferably agitated, as by an air-driven magnetic stirrer 52. The metals recovered on the cathode are illustrated in FIG. 4--the ordinant of this Figure is not delineated in definitive units of measure, as the graphic representation illustrates merely the relative quantities of the various metals recovered.
The addition of soluble iron and iron oxidizing microorganisms (Thiobacillus ferrooxidans) to the ore prior to extraction of cationic metals results in enhanced recovery of copper and other cations in the iron sediment collected in the cathode chamber. The results illustrated in FIG. 4 are obtained by analyzing for metals on the surface of the platinum electrode. In FIG. 5, the metals were collected with filters, dried and weighed. The apparatus of FIG. 3 was operated for 24 hours under the following process parameters:
1 gm powdered ore
350 mls H2 SO4
pH 2.5
5×1010 cells Thiobacillus ferrooxidans
10 volts applied
10 mg FeSO4 7H2 O
FIG. 5 illustrates the quantities of five metals recovered in the apparatus of FIG. 3 with and without the addition of iron oxidizing thiobacilli and soluble iron. The apparatus was operated for 72 hours at 10 volts. One gram of powdered ore was pretreated by shaking for 2 days in 50 mls of pH 2.5 H2 SO4, containing 5×1010 T. ferrooxidans cells and 10 mg FeSO4 7H2 O solution. In each case, the amount recovered with such additions was greater than that without.
Galvanic metal recovery from all coals using the process of FIG. 3 is not uniformly enhanced by the presence of iron oxidizing microorganisms. However, these results indicate that metal recovery from certain coals is consistently enhanced with the bacterio-electric process disclosed above.
PSOC 667, an Iowa subbituminous coal with approximately 13% total sulphur and 4.5% pyritic sulphur, was analyzed in the apparatus of FIG. 3. 0.75 grams of powdered coal was added to the anode chamber, with or without added microorganisms. The container was filled with 200 ml of pH 2.5 H2 SO4 and the apparatus operated at 10 volts for 64 hours. Sediments were collected separately from the cathode and main chambers. Solutes were obtained by evaporation of the filtered lixiviant. 190.5 mg of solids were recovered from the cathode and main chambers in both soluble and suspended form (which does not include materials plated on the cathode, consisting primarily of iron, copper, nickel and sulphur). The 190.5 mg of recovered solids contained approximately 66 mg of metals, distributed as indicated in Table 1.
TABLE 1
__________________________________________________________________________
Distribution of Metals Leached from PSOC Coal (Mg)
Total Atom
Atom
Solids Fe Ca Al K Si Cu S Fe S
__________________________________________________________________________
Galvanic
190.5
54.02
11.03
0.71
0.15
0.43
0.07
40.51
0.967
1.24
Process
Bacterio-
239.9
91.00
1.53
0.72
-- 0.04
0.04
48.60
1.592
1.489
electric
process
__________________________________________________________________________
By comparison, leaching of the PSOC 667 coal with combined bacterial (5×1010 cells T. ferrooxidans added) and electric leaching separated approximately 239 mg of recovered solids, with approximately 93.3 mg of metals contained therein. While the combined bacterial and electric treatment results in enhanced metal extraction from PSOC 667 coal, the accompanying increased precipitation of iron is not an indicator of increased recovery of sulphur, which would be expected if bacterial oxidation had released iron from pyrite. While not attempted herein, Applicants believe that removal of sulphur as well as metals from such coal samples would be enhanced by providing mixed bacterial populations having the ability to readily oxidize elemental sulphur as well as reduced iron. Such mixed bacterial populations may include other species of Thiobacillus, such as T. thiooxidans, and other microbes capable of metabolism in an acidic environment, such as Acidophillium sp. and Leptospirillium sp. Also, bacteria capable of stimulating iron and sulphur oxidixing bacteria may be added.
In order to determine the nature of the cationic metals extracted from coal by the inventive process, the apparatus of FIG. 3 was modified to better determine the results of the process.
A galvanic sandwich, illustrated in FIG. 6, comprises a container or dish 54, having a membrane filter 56 atop a cellulose pad 58. The pad 58 is soaked with a dilute sulfuric acid solution and either overlies or surrounds a stainless steel anode 60. The anode 60 is maintained in contact with the ore specimen 70 within the pad. In the apparatus of FIG. 6, approximately 0.3-0.4 gm of powdered ore was utilized. A platinum cathode 72 rests on the surface of the membrane filter 56 below a layer of paraffin 74 held in place by a lid 76 of container 54. The membrane filter is preferably comprised of cellulose acetate. The sulfuric acid is provided at a pH of from about 0.8 to about 4.5, preferably about 2.5, and electric current was provided for 20 hours at 10 volts. Electrophoretic separation of the metals present in the ore specimen are deposited in zones 78 concentric to the cathode 72 (FIG. 7). The various zones 78 may be cut from the membrane filter 56 and analyzed by appropriate techniques to determine relative amount of the various metals.
It was determined that the zone closest to the cathode contained mostly nickel, while the next closest zone contained mostly nickel and chromium, the next contained primarily iron and sulphur and the farthest from the cathode contained copper and chromium. While the technique of FIGS. 6 and 7 does not permit complete separation of the metals recovered from a coal specimen, it does provide for partial separation and permits a means to better appreciate the metal complexes that are formed.
Significant amounts of copper and zinc are present in the sediment from the bacterio-electric treatment of FIG. 3, but these elements are not collected on the cathode port membrane of a corresponding leaching system without iron oxidizing bacteria added thereto.
An Illinois # 6 coal (PSOC 1322) having about 1.28% pyritic sulphur was subjected to bacterio-electric leaching in the vessel of FIG. 3. It was determined that the cellulose or cellulose acetate filters placed across the cathode port 80 were the site of sediment deposition for some coals. Although the sediment obtained by galvanic leaching with addition of the iron-oxidizing microorganisms differs from the abiotic galvanic sediment having more of the amorphous hydrated Fe+3 SO4, both appear to be inorganic polymers (polyelectrolytes) based on their retention by the filter. Sediments were easily separated by scraping from the cellulose acetate or ashless filter paper and analyzed for metal content.
TABLE 2 __________________________________________________________________________ Distribution of Metals Leache from PSOC 1322Illinois # 6 Coal (Mg) Al Si S Ca Mn Fe Cu Zn K Br TOTAL __________________________________________________________________________ Bac- wt % 1.04 0.25 4.53 0.19 0.89 92.69 0.16 0.25 -- -- 100 terized atomic % 2.06 0.47 7.54 0.25 0.86 88.48 0.13 0.21 -- -- -- No Bac- wt % -- 1.16 1.01 0.24 1.60 92.07 -- -- 0.44 3.48 100 teria atomic -- 2.78 1.75 0.33 1.60 91.01 -- -- 0.62 2.41 -- __________________________________________________________________________
The elemental analysis set forth in Table 2 indicates that significant amounts of copper and zinc are present in the sediment from the bacterio-electric process but are not collected on the cathode port membrane when the process is run without iron-oxidizing bacteria. Applicants believe that either the additional Fe+3 provided by way of bacterial action or by direct bacterial action on copper and zinc minerals, results in greater release of copper and zinc from the bound mineral form.
Because of the concern for sulphur emissions resulting from the burning of coal, it is highly desireable to remove a significant portion of the sulphur in high-sulphur coals prior to burning.
Powdered PSOC 667 Iowa subbituminous coal containing about 4.5% pyritic sulphur was placed at the anode of a galvanic sandwich of FIG. 6. The system was operated at 10 volts for 48 hours. Analysis of the bacterially-sedimented iron (FIG. 8A) is distinguishable from iron oxidized abiotically (FIG. 8B) by the relative amounts of sulphur recovered. The bacterially oxidized Fe+3 complex migrates toward the cathode where it forms a distinct zone that differs from the zone containing Fe+3 complexes formed by abiotic oxidation, as set forth above. A larger proportion of sulphur is found in deposits formed on cellulose acetate filters from iron oxidized by Thiobacillus ferrooxidans then that oxidized abiotically. The data in Table 2 above indicate that the bacterized leach treatment yields a sediment with a significantly greater amount of sulphur than an abiotic treatment. Presumably, recovery of nonmetallic constituents such as sulphur, silicon and phosphorous are complexed with heavier cations recovered by the process.
The process of the present invention is useful to recover sulphur from high-sulphur coals, such as the PSOC 667 sample of Table 1. This coal contains approximately 13% total sulphur and 4-5% pyritic sulphur. As illustrated in Table 1, the total amount of sulphur recovered using the bacterio-electric process of the present invention is approximately 20% greater than by the use of the galvanic process only.
While the process of the present invention has been disclosed herein in a relatively small laboratory apparatus, such processes are intended merely to demonstrate the feasibility of the inventive process. It is contemplated that the process can be most economically carried out in one of three environments: (1) an in situ process wherein the bacteria and electric potential are introduced directly into a subterranean ore body; (2) after an ore body is mined, precious metals or non-precious metals (including hazardous metals) may be recovered from a slurry either prior to refining or as a component of a refining process; or (3) prior to burning, sulphur may be removed from a slurry of coal particles.
It is believed that areas experiencing soil contamination from metals resulting from dumping may be cleansed by the process of the present invention. For example, radionuclides and hazardous organics preferentially complexed with metals subject to removal by this process may be removed from contaminated areas. Also, products of combustion such as fly ash and bag-house dust removed from industrial processes may be cleansed of specific metals by this process.
It is to be understood that while the process of the present invention has been described with reference to an acidic environment, the process is equally susceptible to operation in neutral and basic environments. The limiting factor is the isolation of suitable bacteria having the capability to complex with the desired metals. Applicants have noted galvanic metal extraction in lixiviants having a pH as high as 13.0.
While a preferred embodiment of the invention has been disclosed, various modes of carrying out the principles disclosed herein are contemplated as being within the scope of the following claims. Therefore, it is understood that the scope of the invention is not to be limited except as otherwise set forth in the claims.
Claims (4)
1. A method of removing metals from fossil fuels, comprising:
a. forming and agitating a fossil fuel slurry containing the fossil fuel and sulfuric acid at a Ph of about 2.5 in a vessel;
b. treating the slurry with the iron-oxidizing microorganism, Thiobacillus Ferrooxidans;
c. inserting a pair of electrodes comprising an anode and a cathode into the vessel;
d. applying a voltage of about 10 volts between the electrodes across at least a portion of the vessel to electrolytically dissolve and redeposit the metals; and
e. recovering the electro-deposited metals between the anode and the cathode.
2. The method of claim 1, further comprising increasing the conductivity of the slurry by adding a sulfate salt to the slurry, wherein the sulfate salt is selected from the group consisting of lithium magnesium or aluminum.
3. A method of removing metals from coal prior to combustion of the coal, comprising:
a. forming an acidic slurry of the coal and sulfuric acid at a Ph of about 2.5;
b. adding the iron-oxidating bacteria, Thiobacillus ferrooxidans to the slurry;
c. inducing a voltage of about 10 volts across the slurry between a pair of electrodes; and
d. recovering the metals adjacent one of the electrodes.
4. A method of removing iron pyrite from fossil fuels, comprising:
a. forming a fossil fuel slurry containing the iron pyrite in mixture with sulfuric acid solution at a Ph of about 2.5 and an iron-oxidizing microorganism including Thiobacillus ferrooxidans;
b. electrolytically dissolving the iron pyrite at anodic potential in the presence of the iron-oxidizing microorganism to form Fe+2 in sulfate solution. The Fe+2 being oxidized to Fe+3 by the action of the iron-oxidizing microorganism;
c. electrolytically converting Fe+3 at cathodic potential to Fe+2 and to iron metal; and
d. recovering the iron metal from the slurry.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US07/486,039 USH1074H (en) | 1990-02-27 | 1990-02-27 | Bacterio-electric leaching of metals |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US07/486,039 USH1074H (en) | 1990-02-27 | 1990-02-27 | Bacterio-electric leaching of metals |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| USH1074H true USH1074H (en) | 1992-07-07 |
Family
ID=23930367
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US07/486,039 Abandoned USH1074H (en) | 1990-02-27 | 1990-02-27 | Bacterio-electric leaching of metals |
Country Status (1)
| Country | Link |
|---|---|
| US (1) | USH1074H (en) |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5766930A (en) * | 1995-06-02 | 1998-06-16 | Geobiotics, Inc. | Method of biotreatment for solid materials in a nonstirred surface bioreactor |
| US5885825A (en) * | 1990-08-24 | 1999-03-23 | Brookhaven Science Associates | Biochemical transformation of coals |
| US6083730A (en) * | 1993-12-03 | 2000-07-04 | Geobiotics, Inc. | Nonstirred bioreactor for processing refractory sulfide concentrates and method for operating same |
| US6107065A (en) * | 1995-06-02 | 2000-08-22 | Geobiotics, Inc. | Nonstirred bioreactor for processing refractory sulfide concentrates and method for operating same |
| US6387239B1 (en) * | 1999-11-17 | 2002-05-14 | Bhp Minerals International, Inc. | Recovery of metals from ore |
| CN110629023A (en) * | 2019-10-17 | 2019-12-31 | 中南大学 | A multifunctional biometallurgical reactor |
Citations (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3982995A (en) | 1975-05-07 | 1976-09-28 | The University Of Southern California | Method of converting oil shale into a fuel |
| US4043884A (en) | 1976-08-23 | 1977-08-23 | University Of Southern California | Electrolytic hydrogenation of leached oil shale components |
| US4193854A (en) | 1977-12-23 | 1980-03-18 | Union Carbide Corporation | Heavy metal removal from wastewater sludge |
| US4206288A (en) | 1978-05-05 | 1980-06-03 | Union Carbide Corporation | Microbial desulfurization of coal |
| US4392402A (en) | 1979-05-31 | 1983-07-12 | E.C.H. Will (Gmbh & Co.) | Apparatus for severing running paper webs or the like |
| US4711849A (en) | 1983-08-29 | 1987-12-08 | General Mining Union Corporation, Limited | Construction of selectable shuttle cloning vectors for Thiobacillus ferrooxidans |
| US4748118A (en) | 1983-09-09 | 1988-05-31 | General Mining Union Corporation Limited | Construction of arsenic resistance vectors for Thiobacillus ferrooxidans |
| US4775627A (en) | 1986-04-22 | 1988-10-04 | The Ohio State University, A Branch Of The State Government | Coal desulfurization using bacteria adaptation and bacterial modification of pyrite surfaces |
| US4822413A (en) | 1986-03-13 | 1989-04-18 | Davy Mckee (Stockton) Limited | Extraction of metal values from ores or concentrates |
| US4845034A (en) | 1985-01-22 | 1989-07-04 | Houston Industries Incorporated | Biochemically reacting substrates in subterranean cavities |
-
1990
- 1990-02-27 US US07/486,039 patent/USH1074H/en not_active Abandoned
Patent Citations (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3982995A (en) | 1975-05-07 | 1976-09-28 | The University Of Southern California | Method of converting oil shale into a fuel |
| US4043884A (en) | 1976-08-23 | 1977-08-23 | University Of Southern California | Electrolytic hydrogenation of leached oil shale components |
| US4193854A (en) | 1977-12-23 | 1980-03-18 | Union Carbide Corporation | Heavy metal removal from wastewater sludge |
| US4206288A (en) | 1978-05-05 | 1980-06-03 | Union Carbide Corporation | Microbial desulfurization of coal |
| US4392402A (en) | 1979-05-31 | 1983-07-12 | E.C.H. Will (Gmbh & Co.) | Apparatus for severing running paper webs or the like |
| US4711849A (en) | 1983-08-29 | 1987-12-08 | General Mining Union Corporation, Limited | Construction of selectable shuttle cloning vectors for Thiobacillus ferrooxidans |
| US4748118A (en) | 1983-09-09 | 1988-05-31 | General Mining Union Corporation Limited | Construction of arsenic resistance vectors for Thiobacillus ferrooxidans |
| US4845034A (en) | 1985-01-22 | 1989-07-04 | Houston Industries Incorporated | Biochemically reacting substrates in subterranean cavities |
| US4822413A (en) | 1986-03-13 | 1989-04-18 | Davy Mckee (Stockton) Limited | Extraction of metal values from ores or concentrates |
| US4775627A (en) | 1986-04-22 | 1988-10-04 | The Ohio State University, A Branch Of The State Government | Coal desulfurization using bacteria adaptation and bacterial modification of pyrite surfaces |
Non-Patent Citations (4)
| Title |
|---|
| Lararoff and Dugan, "Bacterio-Electric Separation of Leached Metals from Coals and Sulfide Minerals", 197th American Chemical Society Mtg., Apr. 9-14, 1989. |
| Lazaroff and Dugan, "Bacterio-Electric Removal of Metals from Coal", Bioprocessing of Coals, Tyson's Corner, Va., Aug. 13-18, 1989. |
| Lazaroff, Dugan, and Wey, "Review of Biological Processing and Interactions with Coal", Annual Program Review, Jan. 30, 1990. |
| Srivastava et al., "Coal Bioprocessing: A Research Needs Assessment", Chemical Engineering Progress (after Mar. 27, 1989). |
Cited By (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5885825A (en) * | 1990-08-24 | 1999-03-23 | Brookhaven Science Associates | Biochemical transformation of coals |
| US6083730A (en) * | 1993-12-03 | 2000-07-04 | Geobiotics, Inc. | Nonstirred bioreactor for processing refractory sulfide concentrates and method for operating same |
| US6410304B2 (en) | 1995-06-02 | 2002-06-25 | Geobiotics, Llc | Method of biotreatment for solid materials in a nonstirred surface bioreactor |
| US6107065A (en) * | 1995-06-02 | 2000-08-22 | Geobiotics, Inc. | Nonstirred bioreactor for processing refractory sulfide concentrates and method for operating same |
| US6159726A (en) * | 1995-06-02 | 2000-12-12 | Geobiotics, Inc. | Method of biotreatment for solid materials in a nonstirred surface bioreactor |
| US5766930A (en) * | 1995-06-02 | 1998-06-16 | Geobiotics, Inc. | Method of biotreatment for solid materials in a nonstirred surface bioreactor |
| US20030013166A1 (en) * | 1995-06-02 | 2003-01-16 | Geobiotics, Inc., A California Corporation | Method of biotreatment for solid materials in a nonstirred surface bioreactor |
| US6855527B2 (en) | 1995-06-02 | 2005-02-15 | Geobiotics Llc | Method of biotreatment for solid materials in a nonstirred surface bioreactor |
| US20050112741A1 (en) * | 1995-06-02 | 2005-05-26 | Geobiotics, Inc., A California Corporation | Method of biotreatment for solid materials in a nonstirred surface bioreactor |
| US7416882B2 (en) | 1995-06-02 | 2008-08-26 | Geobiotics, Llc | Method of biotreatment for solid materials in a nonstirred surface bioreactor |
| US20080311647A1 (en) * | 1995-06-02 | 2008-12-18 | Kohr William J | Method of Biotreating a Solid Material Comprising an Organic Compound |
| US8021870B2 (en) | 1995-06-02 | 2011-09-20 | Geobiotics, Llc | Method of biotreating a solid material comprising an organic compound |
| US8030055B2 (en) | 1995-06-02 | 2011-10-04 | Geobiotics, Llc | Method of biotreatment for solid materials in a nonstirred surface bioreactor |
| US6387239B1 (en) * | 1999-11-17 | 2002-05-14 | Bhp Minerals International, Inc. | Recovery of metals from ore |
| CN110629023A (en) * | 2019-10-17 | 2019-12-31 | 中南大学 | A multifunctional biometallurgical reactor |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Brombacher et al. | Biohydrometallurgical processing of solids: a patent review | |
| RU2086682C1 (en) | Hydrometallurgical method of extracting precious metals from persistent sulfide ore | |
| Yuehua et al. | The effect of silver-bearing catalysts on bioleaching of chalcopyrite | |
| Pedersen | Characterization and electrodialytic treatment of wood combustion fly ash for the removal of cadmium | |
| Hiskey | Thiourea leaching of gold and silver—technology update and additional applications | |
| Nakazawa et al. | Effect of activated carbon on the bioleaching of chalcopyrite concentrate | |
| Miller et al. | The low-potential hydrophobic state of pyrite in amyl xanthate flotation with nitrogen | |
| Sato et al. | Effect of silver chloride on the bioleaching of chalcopyrite concentrate | |
| Ubaldini et al. | Biooxidation of arsenopyrite to improve gold cyanidation: study of some parameters and comparison with grinding | |
| Natarajan et al. | Role of galvanic interactions in the bioleaching of Duluth gabbro copper-nickel sulfides | |
| Smith et al. | TREATMENT OPTIONS | |
| Parga et al. | Recovery of silver and gold from cyanide solution by magnetic species formed in the electrocoagulation process | |
| Langhans et al. | Biooxidation of an arsenic-bearing refractory gold ore | |
| KR20230008292A (en) | Method for selective recovery of copper and gold from waste printed circuit boards | |
| Dan et al. | Enrichment of ferric iron on mineral surface during bioleaching of chalcopyrite | |
| USH1074H (en) | Bacterio-electric leaching of metals | |
| Torma | Impact of biotechnology on metal extractions | |
| Kölle et al. | Reduced sulphur compounds in sandy aquifers and their interactions with groundwater | |
| Twidwell et al. | Removal of selenium oxyanions from mine waters utilizing elemental iron and galvanically coupled metals | |
| Tripathy et al. | Effect of chemical pretreatment on bacterial desulphurisation of Assam coal | |
| Abbruzzese et al. | Preparatory bioleaching to the conventional cyanidation of arsenical gold ores | |
| Pesic et al. | Electrochemistry of Thiobacillus ferrooxidans interactions with pyrite | |
| Kumari et al. | Electrochemical aspects of leaching of ocean nodules in the presence and absence of microorganisms | |
| Pradhan et al. | Microbial leaching process to recover valuable metals from spent petroleum catalyst using iron oxidizing bacteria | |
| Mirajkar et al. | Growth and attachment of Thiobacillus ferrooxidans during sulfide mineral leaching |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AS | Assignment |
Owner name: UNITED STATES OF AMERICA, AS REPRESENTED BY THE SE Free format text: ASSIGNMENT OF ASSIGNORS INTEREST.;ASSIGNORS:LAZAROFF, NORMAN;DUGAN, PATRICK R.;REEL/FRAME:005307/0873;SIGNING DATES FROM 19900213 TO 19900219 |
|
| STCF | Information on status: patent grant |
Free format text: PATENTED CASE |