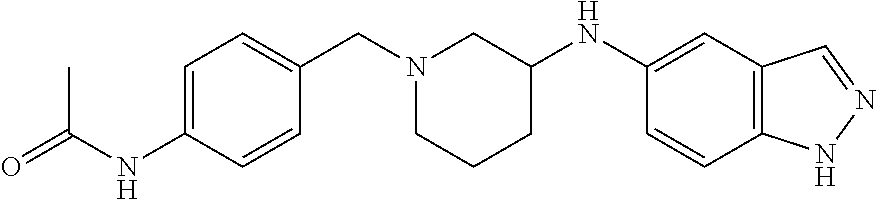
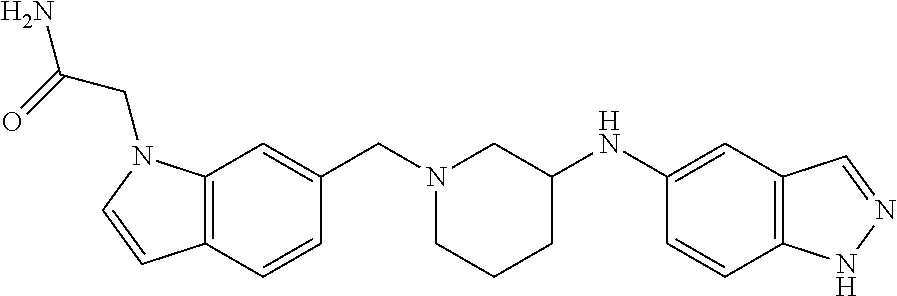
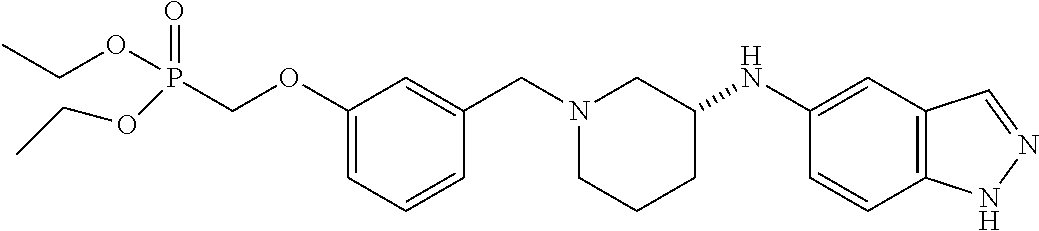
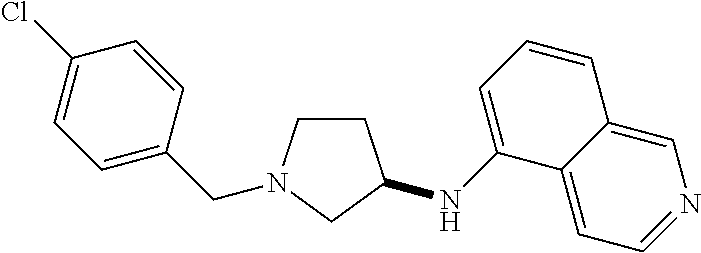
US8604218B2 - Cytoskeletal active rho kinase inhibitor compounds, composition and use - Google Patents
Cytoskeletal active rho kinase inhibitor compounds, composition and use Download PDFInfo
- Publication number
- US8604218B2 US8604218B2 US13/273,043 US201113273043A US8604218B2 US 8604218 B2 US8604218 B2 US 8604218B2 US 201113273043 A US201113273043 A US 201113273043A US 8604218 B2 US8604218 B2 US 8604218B2
- Authority
- US
- United States
- Prior art keywords
- compound
- indazol
- piperidin
- ylamino
- amine
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active, expires
Links
- 0 [1*]CN1CCC(N([2*])[3*])C1 Chemical compound [1*]CN1CCC(N([2*])[3*])C1 0.000 description 8
- SJWGREBZKQINRB-UHFFFAOYSA-N C1=NNC2=CC=C(CC3CCCNC3)C=C12 Chemical compound C1=NNC2=CC=C(CC3CCCNC3)C=C12 SJWGREBZKQINRB-UHFFFAOYSA-N 0.000 description 3
- HOTGJYAYVQJOMO-UHFFFAOYSA-N C1=CC(NC2CCNC2)=C2C=CN=CC2=C1 Chemical compound C1=CC(NC2CCNC2)=C2C=CN=CC2=C1 HOTGJYAYVQJOMO-UHFFFAOYSA-N 0.000 description 2
- HMLQFHNWIUXVQH-UHFFFAOYSA-N C1=CC=C(CN2CCC(CC3=C4C=CNC4=NC=C3)C2)C=C1 Chemical compound C1=CC=C(CN2CCC(CC3=C4C=CNC4=NC=C3)C2)C=C1 HMLQFHNWIUXVQH-UHFFFAOYSA-N 0.000 description 2
- RBMBPNNACSCUKT-UHFFFAOYSA-N C1=CC=C(CN2CCC(CC3=CC=NC=C3)C2)C=C1 Chemical compound C1=CC=C(CN2CCC(CC3=CC=NC=C3)C2)C=C1 RBMBPNNACSCUKT-UHFFFAOYSA-N 0.000 description 2
- IDUQOMMMWYKMON-UHFFFAOYSA-N C1=CC=C(CN2CCCC(CC3=C/C4=C(\C=C/3)NN=C4)CC2)C=C1 Chemical compound C1=CC=C(CN2CCCC(CC3=C/C4=C(\C=C/3)NN=C4)CC2)C=C1 IDUQOMMMWYKMON-UHFFFAOYSA-N 0.000 description 2
- RKQBCMBGDPFJCP-UHFFFAOYSA-N C1=CC=C(CN2CCCC(CC3=C4C=CN=CC4=CC=C3)CC2)C=C1 Chemical compound C1=CC=C(CN2CCCC(CC3=C4C=CN=CC4=CC=C3)CC2)C=C1 RKQBCMBGDPFJCP-UHFFFAOYSA-N 0.000 description 2
- BYEVHWXXLJEEPC-UHFFFAOYSA-N C1=CC=C(CN2CCCC(CC3=C4C=CNC4=NC=C3)C2)C=C1 Chemical compound C1=CC=C(CN2CCCC(CC3=C4C=CNC4=NC=C3)C2)C=C1 BYEVHWXXLJEEPC-UHFFFAOYSA-N 0.000 description 2
- XNVWZORZAVTITP-UHFFFAOYSA-N C1=CC=C(CN2CCCC(CC3=CC=NC=C3)C2)C=C1 Chemical compound C1=CC=C(CN2CCCC(CC3=CC=NC=C3)C2)C=C1 XNVWZORZAVTITP-UHFFFAOYSA-N 0.000 description 2
- YNHPTTHUFMALFY-UHFFFAOYSA-N CC(C)(C)C(=O)N1N=CC2=CC(N)=CC=C21 Chemical compound CC(C)(C)C(=O)N1N=CC2=CC(N)=CC=C21 YNHPTTHUFMALFY-UHFFFAOYSA-N 0.000 description 2
- QPTCSNDPDWGXLB-UHFFFAOYSA-N CC(C)(C)C(=O)N1N=CC2=CC([N+](=O)[O-])=CC=C21 Chemical compound CC(C)(C)C(=O)N1N=CC2=CC([N+](=O)[O-])=CC=C21 QPTCSNDPDWGXLB-UHFFFAOYSA-N 0.000 description 2
- RJUCQPDAUKCDOB-UHFFFAOYSA-N CC1=NNC2=CC=C(CC3CCCN(CC4=CC=CC=C4)C3)C=C21 Chemical compound CC1=NNC2=CC=C(CC3CCCN(CC4=CC=CC=C4)C3)C=C21 RJUCQPDAUKCDOB-UHFFFAOYSA-N 0.000 description 2
- HOPPMEHKDBCSGB-UHFFFAOYSA-N CCSC1=CC=C(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)C=C1 Chemical compound CCSC1=CC=C(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)C=C1 HOPPMEHKDBCSGB-UHFFFAOYSA-N 0.000 description 2
- GSQBEYKCKBBITC-UHFFFAOYSA-N COC1=CC=C(CN2C=CC3=C2N=CC=C3Br)C=C1 Chemical compound COC1=CC=C(CN2C=CC3=C2N=CC=C3Br)C=C1 GSQBEYKCKBBITC-UHFFFAOYSA-N 0.000 description 2
- XSBZPYALIBDMDC-UHFFFAOYSA-N CS(=O)(=O)CC1=CC(CN2CCCC(CC3=CC=C4NN=CC4=C3)C2)=CC=C1 Chemical compound CS(=O)(=O)CC1=CC(CN2CCCC(CC3=CC=C4NN=CC4=C3)C2)=CC=C1 XSBZPYALIBDMDC-UHFFFAOYSA-N 0.000 description 2
- XURBMOAVKHTIQR-UHFFFAOYSA-N CSC1=CC=C(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)C=C1 Chemical compound CSC1=CC=C(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)C=C1 XURBMOAVKHTIQR-UHFFFAOYSA-N 0.000 description 2
- XURBMOAVKHTIQR-GOSISDBHSA-N CSC1=CC=C(CN2CCC[C@H](CC3=CC4=C(C=C3)NN=C4)C2)C=C1 Chemical compound CSC1=CC=C(CN2CCC[C@H](CC3=CC4=C(C=C3)NN=C4)C2)C=C1 XURBMOAVKHTIQR-GOSISDBHSA-N 0.000 description 2
- IQUZIVDCVRBBFR-GOSISDBHSA-N CSC1=CC=C(CN2CC[C@@H](NC3=CC=CC4=CN=CC=C43)C2)C=C1 Chemical compound CSC1=CC=C(CN2CC[C@@H](NC3=CC=CC4=CN=CC=C43)C2)C=C1 IQUZIVDCVRBBFR-GOSISDBHSA-N 0.000 description 2
- NRQKKBVJTBNYKS-UHFFFAOYSA-N NC1=NNC2=CC=C(CC3CCCN(CC4=CC=CC=C4)C3)C=C21 Chemical compound NC1=NNC2=CC=C(CC3CCCN(CC4=CC=CC=C4)C3)C=C21 NRQKKBVJTBNYKS-UHFFFAOYSA-N 0.000 description 2
- MHRFMAYQDRORGH-UHFFFAOYSA-N NC1=NON=C1C1=CC=C(CC2CCCN(CC3=CC=CC=C3)C2)C=C1 Chemical compound NC1=NON=C1C1=CC=C(CC2CCCN(CC3=CC=CC=C3)C2)C=C1 MHRFMAYQDRORGH-UHFFFAOYSA-N 0.000 description 2
- OSWIFSSLZNYWMR-UHFFFAOYSA-N NC1=NON=C1C1=CC=C(CC2CCN(CC3=CC=CC=C3)C2)C=C1 Chemical compound NC1=NON=C1C1=CC=C(CC2CCN(CC3=CC=CC=C3)C2)C=C1 OSWIFSSLZNYWMR-UHFFFAOYSA-N 0.000 description 2
- DSNWZCXVNWVASE-UHFFFAOYSA-N BrC1=CC=C(Br)C(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)=C1 Chemical compound BrC1=CC=C(Br)C(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)=C1 DSNWZCXVNWVASE-UHFFFAOYSA-N 0.000 description 1
- KRPLHKCUZXPUIT-KRWDZBQOSA-N BrC1=CC=C(CN2CCC[C@@H](CC3=CC4=C(C=C3)NN=C4)C2)C=C1 Chemical compound BrC1=CC=C(CN2CCC[C@@H](CC3=CC4=C(C=C3)NN=C4)C2)C=C1 KRPLHKCUZXPUIT-KRWDZBQOSA-N 0.000 description 1
- SOSQXTINASWATM-UHFFFAOYSA-N BrC1=CC=C2C(=C1)C=NN2C1CCCCO1 Chemical compound BrC1=CC=C2C(=C1)C=NN2C1CCCCO1 SOSQXTINASWATM-UHFFFAOYSA-N 0.000 description 1
- YCDBEBUQCHHGFO-UHFFFAOYSA-N BrC1=CC=CC(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)=C1 Chemical compound BrC1=CC=CC(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)=C1 YCDBEBUQCHHGFO-UHFFFAOYSA-N 0.000 description 1
- OSMYTGVDGJBFFI-UHFFFAOYSA-N C#CC1=CC=C(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)C=C1 Chemical compound C#CC1=CC=C(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)C=C1 OSMYTGVDGJBFFI-UHFFFAOYSA-N 0.000 description 1
- MJPZIFUKMFCEFN-UHFFFAOYSA-N C#CC1=CC=C(CN2CCCC(CC3=CC=C4NN=CC4=C3)C2)C=C1O Chemical compound C#CC1=CC=C(CN2CCCC(CC3=CC=C4NN=CC4=C3)C2)C=C1O MJPZIFUKMFCEFN-UHFFFAOYSA-N 0.000 description 1
- OSMYTGVDGJBFFI-FQEVSTJZSA-N C#CC1=CC=C(CN2CCC[C@@H](CC3=CC4=C(C=C3)NN=C4)C2)C=C1 Chemical compound C#CC1=CC=C(CN2CCC[C@@H](CC3=CC4=C(C=C3)NN=C4)C2)C=C1 OSMYTGVDGJBFFI-FQEVSTJZSA-N 0.000 description 1
- OSMYTGVDGJBFFI-HXUWFJFHSA-N C#CC1=CC=C(CN2CCC[C@H](CC3=CC4=C(C=C3)NN=C4)C2)C=C1 Chemical compound C#CC1=CC=C(CN2CCC[C@H](CC3=CC4=C(C=C3)NN=C4)C2)C=C1 OSMYTGVDGJBFFI-HXUWFJFHSA-N 0.000 description 1
- MIMFSAOPASEOEY-HXUWFJFHSA-N C#CC1=CC=C(CN2CC[C@@H](NC3=CC=CC4=CN=CC=C43)C2)C=C1 Chemical compound C#CC1=CC=C(CN2CC[C@@H](NC3=CC=CC4=CN=CC=C43)C2)C=C1 MIMFSAOPASEOEY-HXUWFJFHSA-N 0.000 description 1
- JTISNCNTXOMJDH-UHFFFAOYSA-N C#CC1=CC=CC(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)=C1 Chemical compound C#CC1=CC=CC(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)=C1 JTISNCNTXOMJDH-UHFFFAOYSA-N 0.000 description 1
- LXCXVDDNMONTHW-UHFFFAOYSA-N C(#CC1CC1)C1=CC=C(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)C=C1 Chemical compound C(#CC1CC1)C1=CC=C(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)C=C1 LXCXVDDNMONTHW-UHFFFAOYSA-N 0.000 description 1
- SFATVUFGJJLYSY-UHFFFAOYSA-N C(c(cc1)cc2c1OCCO2)N(CCC1)CC1Nc(cc1)cc2c1[nH]nc2 Chemical compound C(c(cc1)cc2c1OCCO2)N(CCC1)CC1Nc(cc1)cc2c1[nH]nc2 SFATVUFGJJLYSY-UHFFFAOYSA-N 0.000 description 1
- QNBWBURSLYDEDL-UHFFFAOYSA-N C(c1cc([nH]cc2)c2cc1)N(CCC1)CC1Nc(cc1)cc2c1[nH]nc2 Chemical compound C(c1cc([nH]cc2)c2cc1)N(CCC1)CC1Nc(cc1)cc2c1[nH]nc2 QNBWBURSLYDEDL-UHFFFAOYSA-N 0.000 description 1
- OPAWRULAUSLMKC-UHFFFAOYSA-N C1=CC(CC2CCCNC2)=CC=N1 Chemical compound C1=CC(CC2CCCNC2)=CC=N1 OPAWRULAUSLMKC-UHFFFAOYSA-N 0.000 description 1
- DZIRLFMOZVWNDB-UHFFFAOYSA-N C1=CC(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)=CC(C2CC2)=C1 Chemical compound C1=CC(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)=CC(C2CC2)=C1 DZIRLFMOZVWNDB-UHFFFAOYSA-N 0.000 description 1
- CLCMIAAQLBZPHZ-UHFFFAOYSA-N C1=CC(CN2CCCC(NC3=CC=CC4=CN=CC=C43)C2)=CC(C2CC2)=C1 Chemical compound C1=CC(CN2CCCC(NC3=CC=CC4=CN=CC=C43)C2)=CC(C2CC2)=C1 CLCMIAAQLBZPHZ-UHFFFAOYSA-N 0.000 description 1
- ZOHRKOKZKGNCNH-OAQYLSRUSA-N C1=CC(CN2CC[C@@H](NC3=CC=CC4=CN=CC=C43)C2)=CC(C2CC2)=C1 Chemical compound C1=CC(CN2CC[C@@H](NC3=CC=CC4=CN=CC=C43)C2)=CC(C2CC2)=C1 ZOHRKOKZKGNCNH-OAQYLSRUSA-N 0.000 description 1
- ZOHRKOKZKGNCNH-NRFANRHFSA-N C1=CC(CN2CC[C@H](NC3=CC=CC4=CN=CC=C43)C2)=CC(C2CC2)=C1 Chemical compound C1=CC(CN2CC[C@H](NC3=CC=CC4=CN=CC=C43)C2)=CC(C2CC2)=C1 ZOHRKOKZKGNCNH-NRFANRHFSA-N 0.000 description 1
- ALCJHEHAIODSCG-UHFFFAOYSA-N C1=CC(NC2CCCNC2)=C2C=CN=CC2=C1 Chemical compound C1=CC(NC2CCCNC2)=C2C=CN=CC2=C1 ALCJHEHAIODSCG-UHFFFAOYSA-N 0.000 description 1
- HOTGJYAYVQJOMO-LLVKDONJSA-N C1=CC(N[C@@H]2CCNC2)=C2C=CN=CC2=C1 Chemical compound C1=CC(N[C@@H]2CCNC2)=C2C=CN=CC2=C1 HOTGJYAYVQJOMO-LLVKDONJSA-N 0.000 description 1
- HOTGJYAYVQJOMO-NSHDSACASA-N C1=CC(N[C@H]2CCNC2)=C2C=CN=CC2=C1 Chemical compound C1=CC(N[C@H]2CCNC2)=C2C=CN=CC2=C1 HOTGJYAYVQJOMO-NSHDSACASA-N 0.000 description 1
- GHAUYOIWJLUEFX-UHFFFAOYSA-N C1=CC2=C(C=C1CN1CCCC(CC3=CC=C4NN=CC4=C3)C1)OCCO2 Chemical compound C1=CC2=C(C=C1CN1CCCC(CC3=CC=C4NN=CC4=C3)C1)OCCO2 GHAUYOIWJLUEFX-UHFFFAOYSA-N 0.000 description 1
- QCNRZPGBBHCWOF-UHFFFAOYSA-N C1=CC2=C(C=CC(CN3CCCC(CC4=CC5=C(C=C4)NN=C5)C3)=C2)N1 Chemical compound C1=CC2=C(C=CC(CN3CCCC(CC4=CC5=C(C=C4)NN=C5)C3)=C2)N1 QCNRZPGBBHCWOF-UHFFFAOYSA-N 0.000 description 1
- JFALGMUHNDTCBC-UHFFFAOYSA-N C1=CC2=C(C=CC(CN3CCCC(CC4=CC=C5NN=CC5=C4)C3)=C2)O1 Chemical compound C1=CC2=C(C=CC(CN3CCCC(CC4=CC=C5NN=CC5=C4)C3)=C2)O1 JFALGMUHNDTCBC-UHFFFAOYSA-N 0.000 description 1
- VHAWMNRFJKTDEQ-UHFFFAOYSA-N C1=CC2=C(C=CC(CN3CCCC(CC4=CC=C5NN=CC5=C4)C3)=C2)S1 Chemical compound C1=CC2=C(C=CC(CN3CCCC(CC4=CC=C5NN=CC5=C4)C3)=C2)S1 VHAWMNRFJKTDEQ-UHFFFAOYSA-N 0.000 description 1
- JFALGMUHNDTCBC-KRWDZBQOSA-N C1=CC2=C(C=CC(CN3CCC[C@@H](CC4=CC5=C(C=C4)NN=C5)C3)=C2)O1 Chemical compound C1=CC2=C(C=CC(CN3CCC[C@@H](CC4=CC5=C(C=C4)NN=C5)C3)=C2)O1 JFALGMUHNDTCBC-KRWDZBQOSA-N 0.000 description 1
- VHAWMNRFJKTDEQ-KRWDZBQOSA-N C1=CC2=C(C=CC(CN3CCC[C@@H](CC4=CC5=C(C=C4)NN=C5)C3)=C2)S1 Chemical compound C1=CC2=C(C=CC(CN3CCC[C@@H](CC4=CC5=C(C=C4)NN=C5)C3)=C2)S1 VHAWMNRFJKTDEQ-KRWDZBQOSA-N 0.000 description 1
- AVVXFJKNWSEZNN-UHFFFAOYSA-N C1=CC2=C(C=NN2)C=C1CC1CCCN(CC2=CC=C(C3CC3)C=C2)C1 Chemical compound C1=CC2=C(C=NN2)C=C1CC1CCCN(CC2=CC=C(C3CC3)C=C2)C1 AVVXFJKNWSEZNN-UHFFFAOYSA-N 0.000 description 1
- AZBHFXSUXJDAPF-UHFFFAOYSA-N C1=CC2=C(C=NN2)C=C1CC1CCCN(CC2=CC=C(N3CCCC3)C=C2)C1 Chemical compound C1=CC2=C(C=NN2)C=C1CC1CCCN(CC2=CC=C(N3CCCC3)C=C2)C1 AZBHFXSUXJDAPF-UHFFFAOYSA-N 0.000 description 1
- WGQCQHGUIZKNIZ-UHFFFAOYSA-N C1=CC2=C(C=NN2)C=C1CC1CCCN(CC2=CC=C(N3CCOCC3)C=C2)C1 Chemical compound C1=CC2=C(C=NN2)C=C1CC1CCCN(CC2=CC=C(N3CCOCC3)C=C2)C1 WGQCQHGUIZKNIZ-UHFFFAOYSA-N 0.000 description 1
- KVEMFHJXZGVMPV-UHFFFAOYSA-N C1=CC2=C(C=NN2)C=C1CC1CCCN(CC2=CC=C(SC3CC3)C=C2)C1 Chemical compound C1=CC2=C(C=NN2)C=C1CC1CCCN(CC2=CC=C(SC3CC3)C=C2)C1 KVEMFHJXZGVMPV-UHFFFAOYSA-N 0.000 description 1
- RICMFQMYQKOCRU-UHFFFAOYSA-N C1=CC2=C(N=CC=C2CC2CCCNC2)N1 Chemical compound C1=CC2=C(N=CC=C2CC2CCCNC2)N1 RICMFQMYQKOCRU-UHFFFAOYSA-N 0.000 description 1
- IYFZCOPNWBUVLL-UHFFFAOYSA-N C1=CC2=CC=C(CN3CCCC(CC4=CC5=C(C=C4)NN=C5)C3)C=C2C1 Chemical compound C1=CC2=CC=C(CN3CCCC(CC4=CC5=C(C=C4)NN=C5)C3)C=C2C1 IYFZCOPNWBUVLL-UHFFFAOYSA-N 0.000 description 1
- NWQAWCOYBNUWPT-UHFFFAOYSA-N C1=CC2=CN=CC=C2C(NC2CCCN(CC3=CC=C(C4CC4)C=C3)C2)=C1 Chemical compound C1=CC2=CN=CC=C2C(NC2CCCN(CC3=CC=C(C4CC4)C=C3)C2)=C1 NWQAWCOYBNUWPT-UHFFFAOYSA-N 0.000 description 1
- XPXJREKHQWNIQZ-UHFFFAOYSA-N C1=CC2=CN=CC=C2C(NC2CCCN(CC3=CC=C(SC4CC4)C=C3)C2)=C1 Chemical compound C1=CC2=CN=CC=C2C(NC2CCCN(CC3=CC=C(SC4CC4)C=C3)C2)=C1 XPXJREKHQWNIQZ-UHFFFAOYSA-N 0.000 description 1
- LMORZGYOIOJSPZ-OAQYLSRUSA-N C1=CC2=CN=CC=C2C(N[C@@H]2CCN(CC3=CC=C(C4CC4)C=C3)C2)=C1 Chemical compound C1=CC2=CN=CC=C2C(N[C@@H]2CCN(CC3=CC=C(C4CC4)C=C3)C2)=C1 LMORZGYOIOJSPZ-OAQYLSRUSA-N 0.000 description 1
- SPMSXTGVXFPBBK-LJQANCHMSA-N C1=CC2=CN=CC=C2C(N[C@@H]2CCN(CC3=CC=C(SC4CC4)C=C3)C2)=C1 Chemical compound C1=CC2=CN=CC=C2C(N[C@@H]2CCN(CC3=CC=C(SC4CC4)C=C3)C2)=C1 SPMSXTGVXFPBBK-LJQANCHMSA-N 0.000 description 1
- LMORZGYOIOJSPZ-NRFANRHFSA-N C1=CC2=CN=CC=C2C(N[C@H]2CCN(CC3=CC=C(C4CC4)C=C3)C2)=C1 Chemical compound C1=CC2=CN=CC=C2C(N[C@H]2CCN(CC3=CC=C(C4CC4)C=C3)C2)=C1 LMORZGYOIOJSPZ-NRFANRHFSA-N 0.000 description 1
- SPMSXTGVXFPBBK-IBGZPJMESA-N C1=CC2=CN=CC=C2C(N[C@H]2CCN(CC3=CC=C(SC4CC4)C=C3)C2)=C1 Chemical compound C1=CC2=CN=CC=C2C(N[C@H]2CCN(CC3=CC=C(SC4CC4)C=C3)C2)=C1 SPMSXTGVXFPBBK-IBGZPJMESA-N 0.000 description 1
- RJFPALOZHSVMLV-UHFFFAOYSA-N C1=CC=C(C2=CC=C(CN3CCCC(CC4=CC5=C(C=C4)NN=C5)C3)C=C2)C=C1 Chemical compound C1=CC=C(C2=CC=C(CN3CCCC(CC4=CC5=C(C=C4)NN=C5)C3)C=C2)C=C1 RJFPALOZHSVMLV-UHFFFAOYSA-N 0.000 description 1
- YRCNFZIFNSAAQU-UHFFFAOYSA-N C1=CC=C(CN2CCC(NC3=CC=CC4=CN=CC=C43)C2)C=C1 Chemical compound C1=CC=C(CN2CCC(NC3=CC=CC4=CN=CC=C43)C2)C=C1 YRCNFZIFNSAAQU-UHFFFAOYSA-N 0.000 description 1
- KUASAJSJEMCJQG-SFHVURJKSA-N C1=CC=C(CN2CCC[C@@H](CC3=CC4=C(C=C3)NN=C4)C2)C=C1 Chemical compound C1=CC=C(CN2CCC[C@@H](CC3=CC4=C(C=C3)NN=C4)C2)C=C1 KUASAJSJEMCJQG-SFHVURJKSA-N 0.000 description 1
- REEDREJFHLXSDP-UHFFFAOYSA-N C1=CC=C2C=NC=CC2=C1.C1=CC=C2NN=CC2=C1.C1=CC=NC=C1.C1=CN=C2NC=CC2=C1.NC1=NON=C1C1=CC=CC=C1.[CH2+][CH2-].[CH2+][CH2-].[CH2+][CH2-].[CH2+][CH2-].[CH2+][CH2-] Chemical compound C1=CC=C2C=NC=CC2=C1.C1=CC=C2NN=CC2=C1.C1=CC=NC=C1.C1=CN=C2NC=CC2=C1.NC1=NON=C1C1=CC=CC=C1.[CH2+][CH2-].[CH2+][CH2-].[CH2+][CH2-].[CH2+][CH2-].[CH2+][CH2-] REEDREJFHLXSDP-UHFFFAOYSA-N 0.000 description 1
- YSRFOUDJXTXASQ-UHFFFAOYSA-N C1=CN(C2=CC=C(CN3CCCC(CC4=CC5=C(C=C4)NN=C5)C3)C=C2)C=N1 Chemical compound C1=CN(C2=CC=C(CN3CCCC(CC4=CC5=C(C=C4)NN=C5)C3)C=C2)C=N1 YSRFOUDJXTXASQ-UHFFFAOYSA-N 0.000 description 1
- DFUMRKWBUMKFJP-UHFFFAOYSA-N C1=CSC(C2=CC=C(CN3CCCC(CC4=CC5=C(C=C4)NN=C5)C3)C=C2)=C1 Chemical compound C1=CSC(C2=CC=C(CN3CCCC(CC4=CC5=C(C=C4)NN=C5)C3)C=C2)=C1 DFUMRKWBUMKFJP-UHFFFAOYSA-N 0.000 description 1
- ZMNNILZLLPJBKO-UHFFFAOYSA-N C=CC1=CC=C(CN2CCCC(CC3=CC=C4NN=CC4=C3)C2)C=C1 Chemical compound C=CC1=CC=C(CN2CCCC(CC3=CC=C4NN=CC4=C3)C2)C=C1 ZMNNILZLLPJBKO-UHFFFAOYSA-N 0.000 description 1
- ZTPOUQBXFHLGIW-UHFFFAOYSA-N CC(=O)C1=CC=C(CN2CCCC(CC3=CC=C4NN=CC4=C3)C2)C=C1 Chemical compound CC(=O)C1=CC=C(CN2CCCC(CC3=CC=C4NN=CC4=C3)C2)C=C1 ZTPOUQBXFHLGIW-UHFFFAOYSA-N 0.000 description 1
- KZBFZVPALPFZQF-UHFFFAOYSA-N CC(=O)CC1=CC=C(CN2CCC(NC3=CC=CC4=CN=CC=C43)C2)C=C1 Chemical compound CC(=O)CC1=CC=C(CN2CCC(NC3=CC=CC4=CN=CC=C43)C2)C=C1 KZBFZVPALPFZQF-UHFFFAOYSA-N 0.000 description 1
- KAKOBZKMOKWDKI-UHFFFAOYSA-N CC(=O)CC1=CC=C(CN2CCCC(NC3=CC=CC4=CN=CC=C43)C2)C=C1 Chemical compound CC(=O)CC1=CC=C(CN2CCCC(NC3=CC=CC4=CN=CC=C43)C2)C=C1 KAKOBZKMOKWDKI-UHFFFAOYSA-N 0.000 description 1
- BXIUTHMATQYGKF-UHFFFAOYSA-N CC(=O)NC1=CC=C(CN2CCC(CC3=C/C4=C(\C=C/3)NN=C4)C2)C=C1 Chemical compound CC(=O)NC1=CC=C(CN2CCC(CC3=C/C4=C(\C=C/3)NN=C4)C2)C=C1 BXIUTHMATQYGKF-UHFFFAOYSA-N 0.000 description 1
- UKSZQMZQSJTYCI-UHFFFAOYSA-N CC(=O)NC1=CC=C(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)C=C1 Chemical compound CC(=O)NC1=CC=C(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)C=C1 UKSZQMZQSJTYCI-UHFFFAOYSA-N 0.000 description 1
- FENPVOQLZCBPRG-UHFFFAOYSA-N CC(=O)NCC1=CC=CC(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)=C1 Chemical compound CC(=O)NCC1=CC=CC(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)=C1 FENPVOQLZCBPRG-UHFFFAOYSA-N 0.000 description 1
- SYHPIDBQMFMQEL-UHFFFAOYSA-N CC(=O)NCCOC1=CC=CC(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)=C1 Chemical compound CC(=O)NCCOC1=CC=CC(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)=C1 SYHPIDBQMFMQEL-UHFFFAOYSA-N 0.000 description 1
- VGRJFJGRVSPHHY-UHFFFAOYSA-N CC(C)(C)C(=O)N1N=CC2=CC(CC3CCCNC3)=CC=C21 Chemical compound CC(C)(C)C(=O)N1N=CC2=CC(CC3CCCNC3)=CC=C21 VGRJFJGRVSPHHY-UHFFFAOYSA-N 0.000 description 1
- QTIIUQZMZASULO-UHFFFAOYSA-N CC(C)(C)C(=O)N1N=CC2=CC(CC3CCNC3)=CC=C21 Chemical compound CC(C)(C)C(=O)N1N=CC2=CC(CC3CCNC3)=CC=C21 QTIIUQZMZASULO-UHFFFAOYSA-N 0.000 description 1
- SELLOUCVOPBGBM-UHFFFAOYSA-N CC(C)(C)C([n]1ncc2c1ccc(NC1CNCC1)c2)=O Chemical compound CC(C)(C)C([n]1ncc2c1ccc(NC1CNCC1)c2)=O SELLOUCVOPBGBM-UHFFFAOYSA-N 0.000 description 1
- ZPWDZAHAZDAIAD-UHFFFAOYSA-N CC(C)(C)OC(=O)CCC1=CC=C(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)C=C1 Chemical compound CC(C)(C)OC(=O)CCC1=CC=C(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)C=C1 ZPWDZAHAZDAIAD-UHFFFAOYSA-N 0.000 description 1
- YNQDETMNTZZLQA-UHFFFAOYSA-N CC(C)(C)OC(=O)CN1C=CC2=C1C=CC(CN1CCCC(CC3=CC4=C(C=C3)NN=C4)C1)=C2 Chemical compound CC(C)(C)OC(=O)CN1C=CC2=C1C=CC(CN1CCCC(CC3=CC4=C(C=C3)NN=C4)C1)=C2 YNQDETMNTZZLQA-UHFFFAOYSA-N 0.000 description 1
- CBVPKCZQOWASBY-UHFFFAOYSA-N CC(C)(C)OC(=O)N1CCCC(CC2=CC=C(C3=NON=C3N)C=C2)C1 Chemical compound CC(C)(C)OC(=O)N1CCCC(CC2=CC=C(C3=NON=C3N)C=C2)C1 CBVPKCZQOWASBY-UHFFFAOYSA-N 0.000 description 1
- DTKAEILRSFSCEA-UHFFFAOYSA-N CC(C)(C)OC(=O)N1CCCC(CC2=CC=C3C(=C2)C=NN3C(=O)C(C)(C)C)C1 Chemical compound CC(C)(C)OC(=O)N1CCCC(CC2=CC=C3C(=C2)C=NN3C(=O)C(C)(C)C)C1 DTKAEILRSFSCEA-UHFFFAOYSA-N 0.000 description 1
- BKHSELUFPBCGDB-UHFFFAOYSA-N CC(C)(C)OC(=O)N1CCCC(CC2=CC=C3C(=C2)C=NN3C2CCCCO2)C1 Chemical compound CC(C)(C)OC(=O)N1CCCC(CC2=CC=C3C(=C2)C=NN3C2CCCCO2)C1 BKHSELUFPBCGDB-UHFFFAOYSA-N 0.000 description 1
- AFKFDGPLOQZWBL-CQSZACIVSA-N CC(C)(C)OC(=O)N1CC[C@@H](NC2=C3C=CN=CC3=CC=C2)C1 Chemical compound CC(C)(C)OC(=O)N1CC[C@@H](NC2=C3C=CN=CC3=CC=C2)C1 AFKFDGPLOQZWBL-CQSZACIVSA-N 0.000 description 1
- HIJAOOXBAOYEEI-UHFFFAOYSA-N CC(C)(C)OC(=O)NCC1=CC=CC(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)=C1 Chemical compound CC(C)(C)OC(=O)NCC1=CC=CC(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)=C1 HIJAOOXBAOYEEI-UHFFFAOYSA-N 0.000 description 1
- IVWYMSQXWXGHGG-UHFFFAOYSA-N CC(C)C1=CC=C(CN2CCCC(CC3=CC=C4NN=CC4=C3)C2)C=C1 Chemical compound CC(C)C1=CC=C(CN2CCCC(CC3=CC=C4NN=CC4=C3)C2)C=C1 IVWYMSQXWXGHGG-UHFFFAOYSA-N 0.000 description 1
- GUZAKJSDQLLFNI-UHFFFAOYSA-N CC(C)CC1=CC=C(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)C=C1 Chemical compound CC(C)CC1=CC=C(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)C=C1 GUZAKJSDQLLFNI-UHFFFAOYSA-N 0.000 description 1
- OEMSDRIHNWLIIC-UHFFFAOYSA-N CC(C)OC1=CC=C(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)C=C1 Chemical compound CC(C)OC1=CC=C(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)C=C1 OEMSDRIHNWLIIC-UHFFFAOYSA-N 0.000 description 1
- MWRMSZIKVCVCTM-UHFFFAOYSA-N CC1=C(C)C(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)=CC=C1 Chemical compound CC1=C(C)C(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)=CC=C1 MWRMSZIKVCVCTM-UHFFFAOYSA-N 0.000 description 1
- SICJOKJVGHGFLM-UHFFFAOYSA-N CC1=C(O)C=C(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)C=C1 Chemical compound CC1=C(O)C=C(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)C=C1 SICJOKJVGHGFLM-UHFFFAOYSA-N 0.000 description 1
- WCWHSCQNYFXGFN-UHFFFAOYSA-N CC1=C(OCCO)C=C(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)C=C1 Chemical compound CC1=C(OCCO)C=C(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)C=C1 WCWHSCQNYFXGFN-UHFFFAOYSA-N 0.000 description 1
- OGCVTGZBRPXYAI-IBGZPJMESA-N CC1=CC(C)=C(CN2CCC[C@@H](CC3=CC4=C(C=C3)NN=C4)C2)C=C1 Chemical compound CC1=CC(C)=C(CN2CCC[C@@H](CC3=CC4=C(C=C3)NN=C4)C2)C=C1 OGCVTGZBRPXYAI-IBGZPJMESA-N 0.000 description 1
- OGCVTGZBRPXYAI-UHFFFAOYSA-N CC1=CC=C(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)C(C)=C1 Chemical compound CC1=CC=C(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)C(C)=C1 OGCVTGZBRPXYAI-UHFFFAOYSA-N 0.000 description 1
- GVPWJAHSXNVQOH-IBGZPJMESA-N CC1=CC=C(CN2CCC[C@@H](CC3=CC4=C(C=C3)NN=C4)C2)C=C1 Chemical compound CC1=CC=C(CN2CCC[C@@H](CC3=CC4=C(C=C3)NN=C4)C2)C=C1 GVPWJAHSXNVQOH-IBGZPJMESA-N 0.000 description 1
- GVPWJAHSXNVQOH-LJQANCHMSA-N CC1=CC=C(CN2CCC[C@H](CC3=CC4=C(C=C3)NN=C4)C2)C=C1 Chemical compound CC1=CC=C(CN2CCC[C@H](CC3=CC4=C(C=C3)NN=C4)C2)C=C1 GVPWJAHSXNVQOH-LJQANCHMSA-N 0.000 description 1
- UQCGQZYLCBENSJ-LJQANCHMSA-N CC1=CC=C(CN2CC[C@@H](NC3=CC=CC4=CN=CC=C43)C2)C=C1 Chemical compound CC1=CC=C(CN2CC[C@@H](NC3=CC=CC4=CN=CC=C43)C2)C=C1 UQCGQZYLCBENSJ-LJQANCHMSA-N 0.000 description 1
- UQCGQZYLCBENSJ-IBGZPJMESA-N CC1=CC=C(CN2CC[C@H](NC3=CC=CC4=CN=CC=C43)C2)C=C1 Chemical compound CC1=CC=C(CN2CC[C@H](NC3=CC=CC4=CN=CC=C43)C2)C=C1 UQCGQZYLCBENSJ-IBGZPJMESA-N 0.000 description 1
- WMRUQASEKGKODU-UHFFFAOYSA-N CC1=CC=CC(CN2CCCC(CC3=CC=C4NN=CC4=C3)C2)=C1 Chemical compound CC1=CC=CC(CN2CCCC(CC3=CC=C4NN=CC4=C3)C2)=C1 WMRUQASEKGKODU-UHFFFAOYSA-N 0.000 description 1
- KCYOJSRVIMCKPQ-UHFFFAOYSA-N CCC1=CC=C(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)C=C1 Chemical compound CCC1=CC=C(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)C=C1 KCYOJSRVIMCKPQ-UHFFFAOYSA-N 0.000 description 1
- KCYOJSRVIMCKPQ-FQEVSTJZSA-N CCC1=CC=C(CN2CCC[C@@H](CC3=CC4=C(C=C3)NN=C4)C2)C=C1 Chemical compound CCC1=CC=C(CN2CCC[C@@H](CC3=CC4=C(C=C3)NN=C4)C2)C=C1 KCYOJSRVIMCKPQ-FQEVSTJZSA-N 0.000 description 1
- JZQKCPXZTCIHHA-UHFFFAOYSA-N CCCCC1=CC=C(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)C=C1 Chemical compound CCCCC1=CC=C(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)C=C1 JZQKCPXZTCIHHA-UHFFFAOYSA-N 0.000 description 1
- XFSXAENBZQZJTB-UHFFFAOYSA-N CCOC(=O)COC1=CC(CN2CCCC(CC3=CC=C4NN=CC4=C3)C2)=CC=C1 Chemical compound CCOC(=O)COC1=CC(CN2CCCC(CC3=CC=C4NN=CC4=C3)C2)=CC=C1 XFSXAENBZQZJTB-UHFFFAOYSA-N 0.000 description 1
- KJQNEZDOUKEZQC-UHFFFAOYSA-N CCOC1=CC(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)=CC=C1 Chemical compound CCOC1=CC(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)=CC=C1 KJQNEZDOUKEZQC-UHFFFAOYSA-N 0.000 description 1
- YQKFVLFBINJXCF-NRFANRHFSA-N CCOP(=O)(COC1=CC=CC(CN2CCC[C@@H](CC3=CC4=C(C=C3)NN=C4)C2)=C1)OCC Chemical compound CCOP(=O)(COC1=CC=CC(CN2CCC[C@@H](CC3=CC4=C(C=C3)NN=C4)C2)=C1)OCC YQKFVLFBINJXCF-NRFANRHFSA-N 0.000 description 1
- RHKBSAUTYIOMTM-UHFFFAOYSA-N CCS(=O)(=O)CC1=CC=CC(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)=C1 Chemical compound CCS(=O)(=O)CC1=CC=CC(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)=C1 RHKBSAUTYIOMTM-UHFFFAOYSA-N 0.000 description 1
- RHKBSAUTYIOMTM-FQEVSTJZSA-N CCS(=O)(=O)CC1=CC=CC(CN2CCC[C@@H](CC3=CC4=C(C=C3)NN=C4)C2)=C1 Chemical compound CCS(=O)(=O)CC1=CC=CC(CN2CCC[C@@H](CC3=CC4=C(C=C3)NN=C4)C2)=C1 RHKBSAUTYIOMTM-FQEVSTJZSA-N 0.000 description 1
- RHKBSAUTYIOMTM-HXUWFJFHSA-N CCS(=O)(=O)CC1=CC=CC(CN2CCC[C@H](CC3=CC4=C(C=C3)NN=C4)C2)=C1 Chemical compound CCS(=O)(=O)CC1=CC=CC(CN2CCC[C@H](CC3=CC4=C(C=C3)NN=C4)C2)=C1 RHKBSAUTYIOMTM-HXUWFJFHSA-N 0.000 description 1
- CNFJHJMGGNMYDX-HXUWFJFHSA-N CCS(=O)(=O)NC1=CC=CC(CN2CC[C@@H](NC3=CC=CC4=CN=CC=C43)C2)=C1 Chemical compound CCS(=O)(=O)NC1=CC=CC(CN2CC[C@@H](NC3=CC=CC4=CN=CC=C43)C2)=C1 CNFJHJMGGNMYDX-HXUWFJFHSA-N 0.000 description 1
- URERQTDVKMXXIH-UHFFFAOYSA-N CN(C)C1=CC=C(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)C=C1 Chemical compound CN(C)C1=CC=C(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)C=C1 URERQTDVKMXXIH-UHFFFAOYSA-N 0.000 description 1
- VSEDUJDPDZLERR-UHFFFAOYSA-N CN(C)CC1=CC=C(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)C=C1 Chemical compound CN(C)CC1=CC=C(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)C=C1 VSEDUJDPDZLERR-UHFFFAOYSA-N 0.000 description 1
- YDRJPMRDNAGPOO-UHFFFAOYSA-N CN(C)CCCOC1=CC=C(CN2CCC(CC3=C/C4=C(\C=C/3)NN=C4)C2)C=C1 Chemical compound CN(C)CCCOC1=CC=C(CN2CCC(CC3=C/C4=C(\C=C/3)NN=C4)C2)C=C1 YDRJPMRDNAGPOO-UHFFFAOYSA-N 0.000 description 1
- VFWRVGYJFVXOFU-UHFFFAOYSA-N CN(C1=CC=CC(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)=C1)S(C)(=O)=O Chemical compound CN(C1=CC=CC(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)=C1)S(C)(=O)=O VFWRVGYJFVXOFU-UHFFFAOYSA-N 0.000 description 1
- RTNYUODICVDMEL-QFIPXVFZSA-N CN1CCN(C(=O)COC2=CC=CC(CN3CCC[C@@H](CC4=CC5=C(C=C4)NN=C5)C3)=C2)CC1 Chemical compound CN1CCN(C(=O)COC2=CC=CC(CN3CCC[C@@H](CC4=CC5=C(C=C4)NN=C5)C3)=C2)CC1 RTNYUODICVDMEL-QFIPXVFZSA-N 0.000 description 1
- CEJKPZFRJSYAIH-UHFFFAOYSA-N COC1=C(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)C=CC=C1 Chemical compound COC1=C(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)C=CC=C1 CEJKPZFRJSYAIH-UHFFFAOYSA-N 0.000 description 1
- QSMSEMLDTFXSHH-UHFFFAOYSA-N COC1=CC=C(CN2C=CC3=C2N=CC=C3CC2CCCN(C(=O)OC(C)(C)C)C2)C=C1 Chemical compound COC1=CC=C(CN2C=CC3=C2N=CC=C3CC2CCCN(C(=O)OC(C)(C)C)C2)C=C1 QSMSEMLDTFXSHH-UHFFFAOYSA-N 0.000 description 1
- UGTSHNFNZYDEGQ-UHFFFAOYSA-N COC1=CC=C(CN2CCC(NC3=CC=CC4=CN=CC=C43)C2)C=C1 Chemical compound COC1=CC=C(CN2CCC(NC3=CC=CC4=CN=CC=C43)C2)C=C1 UGTSHNFNZYDEGQ-UHFFFAOYSA-N 0.000 description 1
- CINLZTAGAYPKGA-UHFFFAOYSA-N COC1=CC=C(CN2CCCC(NC3=CC=CC4=CN=CC=C43)C2)C=C1 Chemical compound COC1=CC=C(CN2CCCC(NC3=CC=CC4=CN=CC=C43)C2)C=C1 CINLZTAGAYPKGA-UHFFFAOYSA-N 0.000 description 1
- YZCNVWXECIKZSY-GOSISDBHSA-N COC1=CC=C(CN2CCC[C@H](CC3=CC4=C(C=C3)NN=C4)C2)C=C1 Chemical compound COC1=CC=C(CN2CCC[C@H](CC3=CC4=C(C=C3)NN=C4)C2)C=C1 YZCNVWXECIKZSY-GOSISDBHSA-N 0.000 description 1
- VGHPJXFGZMGPLU-UHFFFAOYSA-N COC1=CC=C(CN2N=CC3=CC(Br)=CC=C32)C=C1 Chemical compound COC1=CC=C(CN2N=CC3=CC(Br)=CC=C32)C=C1 VGHPJXFGZMGPLU-UHFFFAOYSA-N 0.000 description 1
- GBMNKFPFRVCIBQ-UHFFFAOYSA-N COC1=CC=C(CN2N=CC3=CC(CC4CCCN(C(=O)OC(C)(C)C)C4)=CC=C32)C=C1 Chemical compound COC1=CC=C(CN2N=CC3=CC(CC4CCCN(C(=O)OC(C)(C)C)C4)=CC=C32)C=C1 GBMNKFPFRVCIBQ-UHFFFAOYSA-N 0.000 description 1
- UCJIXVPTONWDEH-SFHVURJKSA-N COc1ccc(CN(CCC2)C[C@H]2Nc(cc2)cc3c2[nH]nc3)cc1 Chemical compound COc1ccc(CN(CCC2)C[C@H]2Nc(cc2)cc3c2[nH]nc3)cc1 UCJIXVPTONWDEH-SFHVURJKSA-N 0.000 description 1
- VRKKBAZVPLUPBC-UHFFFAOYSA-N CS(=O)(=O)C1=CC=C(CN2CCC(CC3=C/C4=C(\C=C/3)NN=C4)C2)C=C1 Chemical compound CS(=O)(=O)C1=CC=C(CN2CCC(CC3=C/C4=C(\C=C/3)NN=C4)C2)C=C1 VRKKBAZVPLUPBC-UHFFFAOYSA-N 0.000 description 1
- IERPXRYTDDRYGB-UHFFFAOYSA-N CS(=O)(=O)C1=CC=C(CN2CCC(NC3=CC=CC4=CN=CC=C43)C2)C=C1 Chemical compound CS(=O)(=O)C1=CC=C(CN2CCC(NC3=CC=CC4=CN=CC=C43)C2)C=C1 IERPXRYTDDRYGB-UHFFFAOYSA-N 0.000 description 1
- LBRDAFAZGKXSNZ-UHFFFAOYSA-N CS(=O)(=O)C1=CC=C(CN2CCCC(CC3=CC=C4NN=CC4=C3)C2)C=C1 Chemical compound CS(=O)(=O)C1=CC=C(CN2CCCC(CC3=CC=C4NN=CC4=C3)C2)C=C1 LBRDAFAZGKXSNZ-UHFFFAOYSA-N 0.000 description 1
- XSYWGPNWBBDYPP-UHFFFAOYSA-N CS(=O)(=O)C1=CC=C(CN2CCCC(NC3=CC=CC4=CN=CC=C43)C2)C=C1 Chemical compound CS(=O)(=O)C1=CC=C(CN2CCCC(NC3=CC=CC4=CN=CC=C43)C2)C=C1 XSYWGPNWBBDYPP-UHFFFAOYSA-N 0.000 description 1
- MAFUFVVAIBQBIN-UHFFFAOYSA-N CS(=O)(=O)C1=CC=CC(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)=C1 Chemical compound CS(=O)(=O)C1=CC=CC(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)=C1 MAFUFVVAIBQBIN-UHFFFAOYSA-N 0.000 description 1
- KGTZZTNXTGOGIF-UHFFFAOYSA-N CS(=O)(=O)CC1=CC(CN2CCC(CC3=C/C4=C(\C=C/3)NN=C4)C2)=CC=C1 Chemical compound CS(=O)(=O)CC1=CC(CN2CCC(CC3=C/C4=C(\C=C/3)NN=C4)C2)=CC=C1 KGTZZTNXTGOGIF-UHFFFAOYSA-N 0.000 description 1
- KGTZZTNXTGOGIF-SFHVURJKSA-N CS(=O)(=O)CC1=CC(CN2CC[C@@H](CC3=C/C4=C(\C=C/3)NN=C4)C2)=CC=C1 Chemical compound CS(=O)(=O)CC1=CC(CN2CC[C@@H](CC3=C/C4=C(\C=C/3)NN=C4)C2)=CC=C1 KGTZZTNXTGOGIF-SFHVURJKSA-N 0.000 description 1
- KGTZZTNXTGOGIF-GOSISDBHSA-N CS(=O)(=O)CC1=CC(CN2CC[C@H](CC3=C/C4=C(\C=C/3)NN=C4)C2)=CC=C1 Chemical compound CS(=O)(=O)CC1=CC(CN2CC[C@H](CC3=C/C4=C(\C=C/3)NN=C4)C2)=CC=C1 KGTZZTNXTGOGIF-GOSISDBHSA-N 0.000 description 1
- OOAUTNTZJBWXIP-UHFFFAOYSA-N CS(=O)(=O)CC1=CC=C(CN2CCCC(CC3=CC=C4NN=CC4=C3)C2)C=C1 Chemical compound CS(=O)(=O)CC1=CC=C(CN2CCCC(CC3=CC=C4NN=CC4=C3)C2)C=C1 OOAUTNTZJBWXIP-UHFFFAOYSA-N 0.000 description 1
- XSBZPYALIBDMDC-IBGZPJMESA-N CS(=O)(=O)CC1=CC=CC(CN2CCC[C@@H](CC3=CC4=C(C=C3)NN=C4)C2)=C1 Chemical compound CS(=O)(=O)CC1=CC=CC(CN2CCC[C@@H](CC3=CC4=C(C=C3)NN=C4)C2)=C1 XSBZPYALIBDMDC-IBGZPJMESA-N 0.000 description 1
- XSBZPYALIBDMDC-LJQANCHMSA-N CS(=O)(=O)CC1=CC=CC(CN2CCC[C@H](CC3=CC4=C(C=C3)NN=C4)C2)=C1 Chemical compound CS(=O)(=O)CC1=CC=CC(CN2CCC[C@H](CC3=CC4=C(C=C3)NN=C4)C2)=C1 XSBZPYALIBDMDC-LJQANCHMSA-N 0.000 description 1
- VZHDHNDBXDORPP-UHFFFAOYSA-N CS(=O)(=O)NC1=C(Cl)C=CC(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)=C1 Chemical compound CS(=O)(=O)NC1=C(Cl)C=CC(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)=C1 VZHDHNDBXDORPP-UHFFFAOYSA-N 0.000 description 1
- JVACKTOSODNYNH-HXUWFJFHSA-N CS(=O)(=O)NC1=CC(CN2CCC[C@@H](NC3=CC=CC4=CN=CC=C43)C2)=CC=C1 Chemical compound CS(=O)(=O)NC1=CC(CN2CCC[C@@H](NC3=CC=CC4=CN=CC=C43)C2)=CC=C1 JVACKTOSODNYNH-HXUWFJFHSA-N 0.000 description 1
- JVACKTOSODNYNH-FQEVSTJZSA-N CS(=O)(=O)NC1=CC(CN2CCC[C@H](NC3=CC=CC4=CN=CC=C43)C2)=CC=C1 Chemical compound CS(=O)(=O)NC1=CC(CN2CCC[C@H](NC3=CC=CC4=CN=CC=C43)C2)=CC=C1 JVACKTOSODNYNH-FQEVSTJZSA-N 0.000 description 1
- ZRMHZVWQKSOWAN-LJQANCHMSA-N CS(=O)(=O)NC1=CC=CC(CN2CC[C@@H](NC3=CC=CC4=CN=CC=C43)C2)=C1 Chemical compound CS(=O)(=O)NC1=CC=CC(CN2CC[C@@H](NC3=CC=CC4=CN=CC=C43)C2)=C1 ZRMHZVWQKSOWAN-LJQANCHMSA-N 0.000 description 1
- ZRMHZVWQKSOWAN-IBGZPJMESA-N CS(=O)(=O)NC1=CC=CC(CN2CC[C@H](NC3=CC=CC4=CN=CC=C43)C2)=C1 Chemical compound CS(=O)(=O)NC1=CC=CC(CN2CC[C@H](NC3=CC=CC4=CN=CC=C43)C2)=C1 ZRMHZVWQKSOWAN-IBGZPJMESA-N 0.000 description 1
- XAZGHTYPFHLQFK-UHFFFAOYSA-N CSC1=CC(C(=O)N2CCCC(CC3=CC4=C(C=C3)N(C(=O)C(C)(C)C)N=C4)C2)=CC=C1 Chemical compound CSC1=CC(C(=O)N2CCCC(CC3=CC4=C(C=C3)N(C(=O)C(C)(C)C)N=C4)C2)=CC=C1 XAZGHTYPFHLQFK-UHFFFAOYSA-N 0.000 description 1
- JUFDTFNTPCSEGY-UHFFFAOYSA-N CSC1=CC(C(=O)N2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)=CC=C1 Chemical compound CSC1=CC(C(=O)N2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)=CC=C1 JUFDTFNTPCSEGY-UHFFFAOYSA-N 0.000 description 1
- HLIQOCNLZCWQNE-UHFFFAOYSA-N CSC1=CC(CN2CCCC(CC3=CC=C4NN=CC4=C3)C2)=CC=C1 Chemical compound CSC1=CC(CN2CCCC(CC3=CC=C4NN=CC4=C3)C2)=CC=C1 HLIQOCNLZCWQNE-UHFFFAOYSA-N 0.000 description 1
- RDHNSEHZSJYUQE-UHFFFAOYSA-N CSC1=CC=C(CN2CCCC(CC3=CC=C4C(=C3)C=NN4C(=O)C(C)(C)C)C2)C=C1 Chemical compound CSC1=CC=C(CN2CCCC(CC3=CC=C4C(=C3)C=NN4C(=O)C(C)(C)C)C2)C=C1 RDHNSEHZSJYUQE-UHFFFAOYSA-N 0.000 description 1
- DMQNUCUGOVADJT-UHFFFAOYSA-N CSC1=CC=C(CN2CCCC(NC3=CC=CC4=CN=CC=C43)C2)C=C1 Chemical compound CSC1=CC=C(CN2CCCC(NC3=CC=CC4=CN=CC=C43)C2)C=C1 DMQNUCUGOVADJT-UHFFFAOYSA-N 0.000 description 1
- XURBMOAVKHTIQR-SFHVURJKSA-N CSC1=CC=C(CN2CCC[C@@H](CC3=CC4=C(C=C3)NN=C4)C2)C=C1 Chemical compound CSC1=CC=C(CN2CCC[C@@H](CC3=CC4=C(C=C3)NN=C4)C2)C=C1 XURBMOAVKHTIQR-SFHVURJKSA-N 0.000 description 1
- IQUZIVDCVRBBFR-SFHVURJKSA-N CSC1=CC=C(CN2CC[C@H](NC3=CC=CC4=CN=CC=C43)C2)C=C1 Chemical compound CSC1=CC=C(CN2CC[C@H](NC3=CC=CC4=CN=CC=C43)C2)C=C1 IQUZIVDCVRBBFR-SFHVURJKSA-N 0.000 description 1
- ADLCSKFWWMPPMZ-MJGOQNOKSA-N C[C@@H](O)COC1=CC=CC(CN2CCC[C@@H](CC3=CC4=C(C=C3)NN=C4)C2)=C1 Chemical compound C[C@@H](O)COC1=CC=CC(CN2CCC[C@@H](CC3=CC4=C(C=C3)NN=C4)C2)=C1 ADLCSKFWWMPPMZ-MJGOQNOKSA-N 0.000 description 1
- ADLCSKFWWMPPMZ-IEBWSBKVSA-N C[C@@H](O)COC1=CC=CC(CN2CCC[C@H](CC3=CC4=C(C=C3)NN=C4)C2)=C1 Chemical compound C[C@@H](O)COC1=CC=CC(CN2CCC[C@H](CC3=CC4=C(C=C3)NN=C4)C2)=C1 ADLCSKFWWMPPMZ-IEBWSBKVSA-N 0.000 description 1
- HXQOQIFVXSMLGR-UHFFFAOYSA-N ClC1=C(Cl)C=C(CN2CCC(CC3=C/C4=C(\C=C/3)NN=C4)C2)C=C1 Chemical compound ClC1=C(Cl)C=C(CN2CCC(CC3=C/C4=C(\C=C/3)NN=C4)C2)C=C1 HXQOQIFVXSMLGR-UHFFFAOYSA-N 0.000 description 1
- DMPFVMAOEFGDNT-UHFFFAOYSA-N ClC1=CC=C(CN2CCC(NC3=CC=CC4=CN=CC=C43)C2)C=C1Cl Chemical compound ClC1=CC=C(CN2CCC(NC3=CC=CC4=CN=CC=C43)C2)C=C1Cl DMPFVMAOEFGDNT-UHFFFAOYSA-N 0.000 description 1
- HMBSLPNVVKIQGR-UHFFFAOYSA-N ClC1=CC=C(CN2CCCC(NC3=CC=CC4=CN=CC=C43)C2)C=C1Cl Chemical compound ClC1=CC=C(CN2CCCC(NC3=CC=CC4=CN=CC=C43)C2)C=C1Cl HMBSLPNVVKIQGR-UHFFFAOYSA-N 0.000 description 1
- CDEPQRNHNKZDNP-KRWDZBQOSA-N ClC1=CC=C(CN2CCC[C@@H](CC3=CC4=C(C=C3)NN=C4)C2)C=C1 Chemical compound ClC1=CC=C(CN2CCC[C@@H](CC3=CC4=C(C=C3)NN=C4)C2)C=C1 CDEPQRNHNKZDNP-KRWDZBQOSA-N 0.000 description 1
- YHDXNNQMAZXDKA-OAHLLOKOSA-N ClC1=CC=C(CN2CCC[C@H](CC3=CC4=C(C=C3)NN=C4)C2)C=C1Cl Chemical compound ClC1=CC=C(CN2CCC[C@H](CC3=CC4=C(C=C3)NN=C4)C2)C=C1Cl YHDXNNQMAZXDKA-OAHLLOKOSA-N 0.000 description 1
- CDEPQRNHNKZDNP-QGZVFWFLSA-N ClC1=CC=C(CN2CCC[C@H](CC3=CC=C4NN=CC4=C3)C2)C=C1 Chemical compound ClC1=CC=C(CN2CCC[C@H](CC3=CC=C4NN=CC4=C3)C2)C=C1 CDEPQRNHNKZDNP-QGZVFWFLSA-N 0.000 description 1
- UGWBCYQPCXKMGF-GOSISDBHSA-N ClC1=CC=C(CN2CC[C@@H](NC3=CC=CC4=CN=CC=C43)C2)C=C1 Chemical compound ClC1=CC=C(CN2CC[C@@H](NC3=CC=CC4=CN=CC=C43)C2)C=C1 UGWBCYQPCXKMGF-GOSISDBHSA-N 0.000 description 1
- UGWBCYQPCXKMGF-SFHVURJKSA-N ClC1=CC=C(CN2CC[C@H](NC3=CC=CC4=CN=CC=C43)C2)C=C1 Chemical compound ClC1=CC=C(CN2CC[C@H](NC3=CC=CC4=CN=CC=C43)C2)C=C1 UGWBCYQPCXKMGF-SFHVURJKSA-N 0.000 description 1
- UNJROQMNHYROMI-UHFFFAOYSA-N ClC1=CC=C(OC2=CC=CC(CN3CCCC(CC4=CC5=C(C=C4)NN=C5)C3)=C2)C=C1 Chemical compound ClC1=CC=C(OC2=CC=CC(CN3CCCC(CC4=CC5=C(C=C4)NN=C5)C3)=C2)C=C1 UNJROQMNHYROMI-UHFFFAOYSA-N 0.000 description 1
- LOUNGIXPHUIIKS-UHFFFAOYSA-N FC(F)(F)C1=CC(CN2CCC(CC3=C/C4=C(\C=C/3)NN=C4)C2)=CC=C1 Chemical compound FC(F)(F)C1=CC(CN2CCC(CC3=C/C4=C(\C=C/3)NN=C4)C2)=CC=C1 LOUNGIXPHUIIKS-UHFFFAOYSA-N 0.000 description 1
- SSRBFIKGFONJBY-UHFFFAOYSA-N FC(F)(F)C1=CC(CN2CCC(NC3=CC=CC4=CN=CC=C43)C2)=CC=C1 Chemical compound FC(F)(F)C1=CC(CN2CCC(NC3=CC=CC4=CN=CC=C43)C2)=CC=C1 SSRBFIKGFONJBY-UHFFFAOYSA-N 0.000 description 1
- KJWZJDPKSHCNRO-UHFFFAOYSA-N FC(F)(F)C1=CC(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)=CC=C1 Chemical compound FC(F)(F)C1=CC(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)=CC=C1 KJWZJDPKSHCNRO-UHFFFAOYSA-N 0.000 description 1
- KTQNVPQFJGMQDM-UHFFFAOYSA-N FC(F)(F)C1=CC(CN2CCCC(NC3=CC=CC4=CN=CC=C43)C2)=CC=C1 Chemical compound FC(F)(F)C1=CC(CN2CCCC(NC3=CC=CC4=CN=CC=C43)C2)=CC=C1 KTQNVPQFJGMQDM-UHFFFAOYSA-N 0.000 description 1
- WQMNDVFTXGOPOF-UHFFFAOYSA-N FC(F)(F)C1=CC(OC2=CC=CC(CN3CCCC(CC4=CC5=C(C=C4)NN=C5)C3)=C2)=CC=C1 Chemical compound FC(F)(F)C1=CC(OC2=CC=CC(CN3CCCC(CC4=CC5=C(C=C4)NN=C5)C3)=C2)=CC=C1 WQMNDVFTXGOPOF-UHFFFAOYSA-N 0.000 description 1
- DIYSKYLEJWUPCS-UHFFFAOYSA-N FC(F)(F)OC1=CC=C(CN2CCCC(CC3=CC=C4NN=CC4=C3)C2)C=C1 Chemical compound FC(F)(F)OC1=CC=C(CN2CCCC(CC3=CC=C4NN=CC4=C3)C2)C=C1 DIYSKYLEJWUPCS-UHFFFAOYSA-N 0.000 description 1
- QOYNRBLJACVNTD-UHFFFAOYSA-N FC(c1cc(CN(CCC2)CC2Nc(cc2)cc3c2[nH]nc3)ccc1)(F)F Chemical compound FC(c1cc(CN(CCC2)CC2Nc(cc2)cc3c2[nH]nc3)ccc1)(F)F QOYNRBLJACVNTD-UHFFFAOYSA-N 0.000 description 1
- PYRLAXGCKDALJK-HNNXBMFYSA-N FC1=CC=C(CN2CCC[C@@H](CC3=CC4=C(C=C3)NN=C4)C2)C=C1F Chemical compound FC1=CC=C(CN2CCC[C@@H](CC3=CC4=C(C=C3)NN=C4)C2)C=C1F PYRLAXGCKDALJK-HNNXBMFYSA-N 0.000 description 1
- PYRLAXGCKDALJK-OAHLLOKOSA-N FC1=CC=C(CN2CCC[C@H](CC3=CC4=C(C=C3)NN=C4)C2)C=C1F Chemical compound FC1=CC=C(CN2CCC[C@H](CC3=CC4=C(C=C3)NN=C4)C2)C=C1F PYRLAXGCKDALJK-OAHLLOKOSA-N 0.000 description 1
- VRRKUAYXLMKPJI-UHFFFAOYSA-N N#CC1=CC(CN2CCC(CC3=C/C4=C(\C=C/3)NN=C4)C2)=CC=C1 Chemical compound N#CC1=CC(CN2CCC(CC3=C/C4=C(\C=C/3)NN=C4)C2)=CC=C1 VRRKUAYXLMKPJI-UHFFFAOYSA-N 0.000 description 1
- DJUWCELEOPHKCD-UHFFFAOYSA-N N#CC1=CC(CN2CCC(NC3=CC=CC4=CN=CC=C43)C2)=CC=C1 Chemical compound N#CC1=CC(CN2CCC(NC3=CC=CC4=CN=CC=C43)C2)=CC=C1 DJUWCELEOPHKCD-UHFFFAOYSA-N 0.000 description 1
- OJPMQDKCBRAGIK-UHFFFAOYSA-N N#CC1=CC(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)=CC=C1 Chemical compound N#CC1=CC(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)=CC=C1 OJPMQDKCBRAGIK-UHFFFAOYSA-N 0.000 description 1
- HGIRVGIKIQKKOC-UHFFFAOYSA-N N#CC1=CC(CN2CCCC(NC3=CC=CC4=CN=CC=C43)C2)=CC=C1 Chemical compound N#CC1=CC(CN2CCCC(NC3=CC=CC4=CN=CC=C43)C2)=CC=C1 HGIRVGIKIQKKOC-UHFFFAOYSA-N 0.000 description 1
- RIAOHPSVUILRSJ-UHFFFAOYSA-N N#CC1=CC=C(CN2CCCC(CC3=CC=C4NN=CC4=C3)C2)C=C1 Chemical compound N#CC1=CC=C(CN2CCCC(CC3=CC=C4NN=CC4=C3)C2)C=C1 RIAOHPSVUILRSJ-UHFFFAOYSA-N 0.000 description 1
- QISWBLDEUBIFPE-UHFFFAOYSA-N NC(=O)C1=CC=CC(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)=C1 Chemical compound NC(=O)C1=CC=CC(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)=C1 QISWBLDEUBIFPE-UHFFFAOYSA-N 0.000 description 1
- ZBPIZZDXLOMYEJ-UHFFFAOYSA-N NC(=O)CN1C=CC2=C1C=C(CN1CCCC(CC3=CC4=C(C=C3)NN=C4)C1)C=C2 Chemical compound NC(=O)CN1C=CC2=C1C=C(CN1CCCC(CC3=CC4=C(C=C3)NN=C4)C1)C=C2 ZBPIZZDXLOMYEJ-UHFFFAOYSA-N 0.000 description 1
- ZBPIZZDXLOMYEJ-SFHVURJKSA-N NC(=O)CN1C=CC2=C1C=C(CN1CCC[C@@H](CC3=CC4=C(C=C3)NN=C4)C1)C=C2 Chemical compound NC(=O)CN1C=CC2=C1C=C(CN1CCC[C@@H](CC3=CC4=C(C=C3)NN=C4)C1)C=C2 ZBPIZZDXLOMYEJ-SFHVURJKSA-N 0.000 description 1
- ZBPIZZDXLOMYEJ-GOSISDBHSA-N NC(=O)CN1C=CC2=C1C=C(CN1CCC[C@H](CC3=CC4=C(C=C3)NN=C4)C1)C=C2 Chemical compound NC(=O)CN1C=CC2=C1C=C(CN1CCC[C@H](CC3=CC4=C(C=C3)NN=C4)C1)C=C2 ZBPIZZDXLOMYEJ-GOSISDBHSA-N 0.000 description 1
- MTYHEFMPHIKGIH-UHFFFAOYSA-N NC(=O)CN1C=CC2=C1C=CC(CN1CCCC(CC3=CC4=C(C=C3)NN=C4)C1)=C2 Chemical compound NC(=O)CN1C=CC2=C1C=CC(CN1CCCC(CC3=CC4=C(C=C3)NN=C4)C1)=C2 MTYHEFMPHIKGIH-UHFFFAOYSA-N 0.000 description 1
- SKQQGYXUNKXKFY-UHFFFAOYSA-N NC(=O)COC1=CC=CC(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)=C1 Chemical compound NC(=O)COC1=CC=CC(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)=C1 SKQQGYXUNKXKFY-UHFFFAOYSA-N 0.000 description 1
- XJBMIEOOJSNFLV-GOSISDBHSA-N NC(=O)COC1=CC=CC(CN2CC[C@@H](NC3=CC=CC4=CN=CC=C43)C2)=C1 Chemical compound NC(=O)COC1=CC=CC(CN2CC[C@@H](NC3=CC=CC4=CN=CC=C43)C2)=C1 XJBMIEOOJSNFLV-GOSISDBHSA-N 0.000 description 1
- UIWBKOYHKDISKA-UHFFFAOYSA-N NC1=C(Cl)C=CC(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)=C1 Chemical compound NC1=C(Cl)C=CC(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)=C1 UIWBKOYHKDISKA-UHFFFAOYSA-N 0.000 description 1
- PXNZHWOGIVAURZ-UHFFFAOYSA-N NC1=CC=C(C2=NON=C2N)C=C1 Chemical compound NC1=CC=C(C2=NON=C2N)C=C1 PXNZHWOGIVAURZ-UHFFFAOYSA-N 0.000 description 1
- AUKYDYPKRKRRHH-UHFFFAOYSA-N NC1=NON=C1C1=CC=C(CC2CCCNC2)C=C1 Chemical compound NC1=NON=C1C1=CC=C(CC2CCCNC2)C=C1 AUKYDYPKRKRRHH-UHFFFAOYSA-N 0.000 description 1
- ZEFURAOTBQBPON-UHFFFAOYSA-N NC1=NON=C1C1=CC=C([N+](=O)[O-])C=C1 Chemical compound NC1=NON=C1C1=CC=C([N+](=O)[O-])C=C1 ZEFURAOTBQBPON-UHFFFAOYSA-N 0.000 description 1
- JEMWDCCPNFBZSH-UHFFFAOYSA-N NCC1=CC=C(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)C=C1 Chemical compound NCC1=CC=C(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)C=C1 JEMWDCCPNFBZSH-UHFFFAOYSA-N 0.000 description 1
- LYBUMCJKEHKKFR-UHFFFAOYSA-N NCC1=CC=CC(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)=C1 Chemical compound NCC1=CC=CC(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)=C1 LYBUMCJKEHKKFR-UHFFFAOYSA-N 0.000 description 1
- KBWIUPKUMFYIER-UHFFFAOYSA-N NCCOC1=CC(CN2CCCC(CC3=CC=C4NN=CC4=C3)C2)=CC=C1 Chemical compound NCCOC1=CC(CN2CCCC(CC3=CC=C4NN=CC4=C3)C2)=CC=C1 KBWIUPKUMFYIER-UHFFFAOYSA-N 0.000 description 1
- MGCVDAMNDBRKJO-UHFFFAOYSA-N NS(=O)(=O)C1=CC=CC(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)=C1 Chemical compound NS(=O)(=O)C1=CC=CC(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)=C1 MGCVDAMNDBRKJO-UHFFFAOYSA-N 0.000 description 1
- GNXDSMNDOZIRSA-NRFANRHFSA-N O=C(COC1=CC=CC(CN2CCC[C@@H](CC3=CC4=C(C=C3)NN=C4)C2)=C1)N1CCOCC1 Chemical compound O=C(COC1=CC=CC(CN2CCC[C@@H](CC3=CC4=C(C=C3)NN=C4)C2)=C1)N1CCOCC1 GNXDSMNDOZIRSA-NRFANRHFSA-N 0.000 description 1
- IKPCABDNMUSPOD-NRFANRHFSA-N O=C(COC1=CC=CC(CN2CCC[C@@H](CC3=CC4=C(C=C3)NN=C4)C2)=C1)NC1=CC=CN=C1 Chemical compound O=C(COC1=CC=CC(CN2CCC[C@@H](CC3=CC4=C(C=C3)NN=C4)C2)=C1)NC1=CC=CN=C1 IKPCABDNMUSPOD-NRFANRHFSA-N 0.000 description 1
- CVQXJHBUWUKUAE-JOCHJYFZSA-N O=C(COC1=CC=CC(CN2CC[C@@H](NC3=CC=CC4=CN=CC=C43)C2)=C1)N1CCOCC1 Chemical compound O=C(COC1=CC=CC(CN2CC[C@@H](NC3=CC=CC4=CN=CC=C43)C2)=C1)N1CCOCC1 CVQXJHBUWUKUAE-JOCHJYFZSA-N 0.000 description 1
- PJBDOXAGDHFJHF-HSZRJFAPSA-N O=C(COc1cccc(CN(CCC2)C[C@@H]2Nc2ccc3[nH]ncc3c2)c1)Nc1cccnc1 Chemical compound O=C(COc1cccc(CN(CCC2)C[C@@H]2Nc2ccc3[nH]ncc3c2)c1)Nc1cccnc1 PJBDOXAGDHFJHF-HSZRJFAPSA-N 0.000 description 1
- ZMIXJFMYMCAXCD-UHFFFAOYSA-N O=C(O)CN1C=CC2=C1C=C(CN1CCCC(CC3=CC4=C(C=C3)NN=C4)C1)C=C2 Chemical compound O=C(O)CN1C=CC2=C1C=C(CN1CCCC(CC3=CC4=C(C=C3)NN=C4)C1)C=C2 ZMIXJFMYMCAXCD-UHFFFAOYSA-N 0.000 description 1
- PJEYLOAHNROVAG-UHFFFAOYSA-N O=C(O)CN1C=CC2=C1C=CC(CN1CCCC(CC3=CC4=C(C=C3)NN=C4)C1)=C2 Chemical compound O=C(O)CN1C=CC2=C1C=CC(CN1CCCC(CC3=CC4=C(C=C3)NN=C4)C1)=C2 PJEYLOAHNROVAG-UHFFFAOYSA-N 0.000 description 1
- FJMDCZYCYZXYLO-UHFFFAOYSA-N O=C(O)COC1=CC(CN2CCCC(CC3=CC=C4NN=CC4=C3)C2)=CC=C1 Chemical compound O=C(O)COC1=CC(CN2CCCC(CC3=CC=C4NN=CC4=C3)C2)=CC=C1 FJMDCZYCYZXYLO-UHFFFAOYSA-N 0.000 description 1
- FJMDCZYCYZXYLO-KRWDZBQOSA-N O=C(O)COC1=CC=CC(CN2CCC[C@@H](CC3=CC4=C(C=C3)NN=C4)C2)=C1 Chemical compound O=C(O)COC1=CC=CC(CN2CCC[C@@H](CC3=CC4=C(C=C3)NN=C4)C2)=C1 FJMDCZYCYZXYLO-KRWDZBQOSA-N 0.000 description 1
- UPWDVLSJGHGBFP-GOSISDBHSA-N O=C(O)COC1=CC=CC(CN2CC[C@@H](NC3=CC=CC4=CN=CC=C43)C2)=C1 Chemical compound O=C(O)COC1=CC=CC(CN2CC[C@@H](NC3=CC=CC4=CN=CC=C43)C2)=C1 UPWDVLSJGHGBFP-GOSISDBHSA-N 0.000 description 1
- QRWUYEJXAHMNJP-QGZVFWFLSA-N OC(O)COC1=CC=CC(CN2CCC[C@H](CC3=CC4=C(C=C3)NN=C4)C2)=C1 Chemical compound OC(O)COC1=CC=CC(CN2CCC[C@H](CC3=CC4=C(C=C3)NN=C4)C2)=C1 QRWUYEJXAHMNJP-QGZVFWFLSA-N 0.000 description 1
- KQOZEFZRNJQPPK-UHFFFAOYSA-N OC1=C(I)C=CC(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)=C1 Chemical compound OC1=C(I)C=CC(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)=C1 KQOZEFZRNJQPPK-UHFFFAOYSA-N 0.000 description 1
- UDXCQFBTKWGMEB-UHFFFAOYSA-N OC1=CC=CC(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)=C1 Chemical compound OC1=CC=CC(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)=C1 UDXCQFBTKWGMEB-UHFFFAOYSA-N 0.000 description 1
- AGDXETYAOSQBRW-UHFFFAOYSA-N OCC#CC1=CC=C(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)C=C1 Chemical compound OCC#CC1=CC=C(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)C=C1 AGDXETYAOSQBRW-UHFFFAOYSA-N 0.000 description 1
- ILHMLLPVURAPFF-UHFFFAOYSA-N OCC1=CC=C(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)C=C1 Chemical compound OCC1=CC=C(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)C=C1 ILHMLLPVURAPFF-UHFFFAOYSA-N 0.000 description 1
- DEONFZLPQMBQJM-UHFFFAOYSA-N OCCC#CC1=CC=C(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)C=C1 Chemical compound OCCC#CC1=CC=C(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)C=C1 DEONFZLPQMBQJM-UHFFFAOYSA-N 0.000 description 1
- KZUAMEIXAPIXQT-UHFFFAOYSA-N OCCCOC1=CC(CN2CCCC(CC3=CC=C4NN=CC4=C3)C2)=CC=C1 Chemical compound OCCCOC1=CC(CN2CCCC(CC3=CC=C4NN=CC4=C3)C2)=CC=C1 KZUAMEIXAPIXQT-UHFFFAOYSA-N 0.000 description 1
- KJWNNRMGCHCQGS-UHFFFAOYSA-N OCCN1C=CC2=C1C=C(CN1CCCC(CC3=CC4=C(C=C3)NN=C4)C1)C=C2 Chemical compound OCCN1C=CC2=C1C=C(CN1CCCC(CC3=CC4=C(C=C3)NN=C4)C1)C=C2 KJWNNRMGCHCQGS-UHFFFAOYSA-N 0.000 description 1
- KJWNNRMGCHCQGS-IBGZPJMESA-N OCCN1C=CC2=C1C=C(CN1CCC[C@@H](CC3=CC4=C(C=C3)NN=C4)C1)C=C2 Chemical compound OCCN1C=CC2=C1C=C(CN1CCC[C@@H](CC3=CC4=C(C=C3)NN=C4)C1)C=C2 KJWNNRMGCHCQGS-IBGZPJMESA-N 0.000 description 1
- KJWNNRMGCHCQGS-LJQANCHMSA-N OCCN1C=CC2=C1C=C(CN1CCC[C@H](CC3=CC4=C(C=C3)NN=C4)C1)C=C2 Chemical compound OCCN1C=CC2=C1C=C(CN1CCC[C@H](CC3=CC4=C(C=C3)NN=C4)C1)C=C2 KJWNNRMGCHCQGS-LJQANCHMSA-N 0.000 description 1
- ORAVSFOSBKCBDN-UHFFFAOYSA-N OCCN1C=CC2=C1C=CC(CN1CCCC(CC3=CC4=C(C=C3)NN=C4)C1)=C2 Chemical compound OCCN1C=CC2=C1C=CC(CN1CCCC(CC3=CC4=C(C=C3)NN=C4)C1)=C2 ORAVSFOSBKCBDN-UHFFFAOYSA-N 0.000 description 1
- WVMPOEPIOOMWER-UHFFFAOYSA-N OCCN1CCC2=C1C=C(CN1CCCC(CC3=CC4=C(C=C3)NN=C4)C1)C=C2 Chemical compound OCCN1CCC2=C1C=C(CN1CCCC(CC3=CC4=C(C=C3)NN=C4)C1)C=C2 WVMPOEPIOOMWER-UHFFFAOYSA-N 0.000 description 1
- VITGPBWGHJMQQF-UHFFFAOYSA-N OCCOC1=CC(CN2CCC(CC3=C/C4=C(\C=C/3)NN=C4)C2)=CC=C1 Chemical compound OCCOC1=CC(CN2CCC(CC3=C/C4=C(\C=C/3)NN=C4)C2)=CC=C1 VITGPBWGHJMQQF-UHFFFAOYSA-N 0.000 description 1
- BELRNBSYCBEOFC-UHFFFAOYSA-N OCCOC1=CC(CN2CCCC(CC3=CC=C4NN=CC4=C3)C2)=CC=C1 Chemical compound OCCOC1=CC(CN2CCCC(CC3=CC=C4NN=CC4=C3)C2)=CC=C1 BELRNBSYCBEOFC-UHFFFAOYSA-N 0.000 description 1
- VDEVCBVUHZCKCU-HXUWFJFHSA-N OCCOC1=CC(CN2CCC[C@@H](NC3=CC=CC4=CN=CC=C43)C2)=CC=C1 Chemical compound OCCOC1=CC(CN2CCC[C@@H](NC3=CC=CC4=CN=CC=C43)C2)=CC=C1 VDEVCBVUHZCKCU-HXUWFJFHSA-N 0.000 description 1
- VDEVCBVUHZCKCU-FQEVSTJZSA-N OCCOC1=CC(CN2CCC[C@H](NC3=CC=CC4=CN=CC=C43)C2)=CC=C1 Chemical compound OCCOC1=CC(CN2CCC[C@H](NC3=CC=CC4=CN=CC=C43)C2)=CC=C1 VDEVCBVUHZCKCU-FQEVSTJZSA-N 0.000 description 1
- VITGPBWGHJMQQF-SFHVURJKSA-N OCCOC1=CC(CN2CC[C@@H](CC3=C/C4=C(\C=C/3)NN=C4)C2)=CC=C1 Chemical compound OCCOC1=CC(CN2CC[C@@H](CC3=C/C4=C(\C=C/3)NN=C4)C2)=CC=C1 VITGPBWGHJMQQF-SFHVURJKSA-N 0.000 description 1
- VITGPBWGHJMQQF-GOSISDBHSA-N OCCOC1=CC(CN2CC[C@H](CC3=C/C4=C(\C=C/3)NN=C4)C2)=CC=C1 Chemical compound OCCOC1=CC(CN2CC[C@H](CC3=C/C4=C(\C=C/3)NN=C4)C2)=CC=C1 VITGPBWGHJMQQF-GOSISDBHSA-N 0.000 description 1
- MEFIIXOVGADQIR-UHFFFAOYSA-N OCCOC1=CC=C(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)C=C1 Chemical compound OCCOC1=CC=C(CN2CCCC(CC3=CC4=C(C=C3)NN=C4)C2)C=C1 MEFIIXOVGADQIR-UHFFFAOYSA-N 0.000 description 1
- YBJHGAVHUZKRLT-UHFFFAOYSA-N OCCOC1=CC=CC(CN2CCC(CC3=CC4=C(C=C3)NN=C4)CC2)=C1 Chemical compound OCCOC1=CC=CC(CN2CCC(CC3=CC4=C(C=C3)NN=C4)CC2)=C1 YBJHGAVHUZKRLT-UHFFFAOYSA-N 0.000 description 1
- HRCKLYFDWWYCLS-UHFFFAOYSA-N OCCOC1=CC=CC(CN2CCC(NC3=CC=CC4=CN=CC=C43)C2)=C1 Chemical compound OCCOC1=CC=CC(CN2CCC(NC3=CC=CC4=CN=CC=C43)C2)=C1 HRCKLYFDWWYCLS-UHFFFAOYSA-N 0.000 description 1
- BELRNBSYCBEOFC-SFHVURJKSA-N OCCOC1=CC=CC(CN2CCC[C@@H](CC3=CC4=C(C=C3)NN=C4)C2)=C1 Chemical compound OCCOC1=CC=CC(CN2CCC[C@@H](CC3=CC4=C(C=C3)NN=C4)C2)=C1 BELRNBSYCBEOFC-SFHVURJKSA-N 0.000 description 1
- BELRNBSYCBEOFC-GOSISDBHSA-N OCCOC1=CC=CC(CN2CCC[C@H](CC3=CC4=C(C=C3)NN=C4)C2)=C1 Chemical compound OCCOC1=CC=CC(CN2CCC[C@H](CC3=CC4=C(C=C3)NN=C4)C2)=C1 BELRNBSYCBEOFC-GOSISDBHSA-N 0.000 description 1
- HRCKLYFDWWYCLS-LJQANCHMSA-N OCCOC1=CC=CC(CN2CC[C@@H](NC3=CC=CC4=CN=CC=C43)C2)=C1 Chemical compound OCCOC1=CC=CC(CN2CC[C@@H](NC3=CC=CC4=CN=CC=C43)C2)=C1 HRCKLYFDWWYCLS-LJQANCHMSA-N 0.000 description 1
- HRCKLYFDWWYCLS-IBGZPJMESA-N OCCOC1=CC=CC(CN2CC[C@H](NC3=CC=CC4=CN=CC=C43)C2)=C1 Chemical compound OCCOC1=CC=CC(CN2CC[C@H](NC3=CC=CC4=CN=CC=C43)C2)=C1 HRCKLYFDWWYCLS-IBGZPJMESA-N 0.000 description 1
- XEQSNPVXRPAPMJ-OAQYLSRUSA-N OCC[n](cc1)c2c1ccc(CN(CCC1)C[C@@H]1Nc(cc1)cc3c1[nH]nc3)c2 Chemical compound OCC[n](cc1)c2c1ccc(CN(CCC1)C[C@@H]1Nc(cc1)cc3c1[nH]nc3)c2 XEQSNPVXRPAPMJ-OAQYLSRUSA-N 0.000 description 1
- WXSOYZKWKZNVQL-GHTZIAJQSA-N OC[C@@H](O)COC1=CC=CC(CN2CCC[C@@H](CC3=CC4=C(C=C3)NN=C4)C2)=C1 Chemical compound OC[C@@H](O)COC1=CC=CC(CN2CCC[C@@H](CC3=CC4=C(C=C3)NN=C4)C2)=C1 WXSOYZKWKZNVQL-GHTZIAJQSA-N 0.000 description 1
- WXSOYZKWKZNVQL-RXVVDRJESA-N OC[C@H](O)COC1=CC=CC(CN2CCC[C@@H](CC3=CC4=C(C=C3)NN=C4)C2)=C1 Chemical compound OC[C@H](O)COC1=CC=CC(CN2CCC[C@@H](CC3=CC4=C(C=C3)NN=C4)C2)=C1 WXSOYZKWKZNVQL-RXVVDRJESA-N 0.000 description 1
- NVXGZQGKWLMDKQ-UHFFFAOYSA-N OCc1ccc(CN(CCC2)CC2Nc(cc2)cc3c2[nH]nc3)cc1 Chemical compound OCc1ccc(CN(CCC2)CC2Nc(cc2)cc3c2[nH]nc3)cc1 NVXGZQGKWLMDKQ-UHFFFAOYSA-N 0.000 description 1
- ZEAHQEHAYJCLND-UHFFFAOYSA-N Oc1cc(CN(CCC2)CC2Nc(cc2)cc3c2[nH]nc3)ccc1 Chemical compound Oc1cc(CN(CCC2)CC2Nc(cc2)cc3c2[nH]nc3)ccc1 ZEAHQEHAYJCLND-UHFFFAOYSA-N 0.000 description 1
- NEKHXYRCMNVPKS-UHFFFAOYSA-N [CH2-][C+]1CC[NH+]([CH2-])C1 Chemical compound [CH2-][C+]1CC[NH+]([CH2-])C1 NEKHXYRCMNVPKS-UHFFFAOYSA-N 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/12—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D231/00—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings
- C07D231/54—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings condensed with carbocyclic rings or ring systems
- C07D231/56—Benzopyrazoles; Hydrogenated benzopyrazoles
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/41—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with two or more ring hetero atoms, at least one of which being nitrogen, e.g. tetrazole
- A61K31/415—1,2-Diazoles
- A61K31/416—1,2-Diazoles condensed with carbocyclic ring systems, e.g. indazole
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0048—Eye, e.g. artificial tears
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P27/00—Drugs for disorders of the senses
- A61P27/02—Ophthalmic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P27/00—Drugs for disorders of the senses
- A61P27/02—Ophthalmic agents
- A61P27/06—Antiglaucoma agents or miotics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/14—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D403/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00
- C07D403/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings
- C07D403/12—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D405/00—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom
- C07D405/14—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D409/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms
- C07D409/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D413/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D413/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings
- C07D413/12—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D413/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D413/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing three or more hetero rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F9/00—Methods or devices for treatment of the eyes; Devices for putting-in contact lenses; Devices to correct squinting; Apparatus to guide the blind; Protective devices for the eyes, carried on the body or in the hand
- A61F9/007—Methods or devices for eye surgery
- A61F9/00781—Apparatus for modifying intraocular pressure, e.g. for glaucoma treatment
Definitions
- This invention relates to synthetic cytoskeletal active compounds, such as rho-associated kinase (ROCK) inhibiting compounds, and the methods of making such compounds.
- the invention also relates to using such compounds in the prevention or treatment of diseases or disorders that are affected or can be assisted by altering the integrity or rearrangement of the cytoskeleton, including but not exclusive of actomyosin interactions, tight junctional and focal adhesion complexes, for example, the treatment of disorders in which intraocular pressure is elevated, such as primary open-angle glaucoma.
- Glaucoma is an ophthalmic disease that leads to irreversible visual impairment. It is the fourth most common cause of blindness and the second most common cause of visual loss in the United States, and the most common cause of irreversible visual loss among African-Americans. Generally speaking, the disease is characterized by a progressive optic neuropathy caused at least in part by deleterious effects resulting from increased intraocular pressure. In normal individuals, intraocular pressures range from 12 to 20 mm Hg, averaging approximately 16 mm Hg. However, in individuals suffering from primary open angle glaucoma, intraocular pressures generally rise above 22 to 30 mm Hg. In angle closure or acute glaucoma intraocular pressure can reach as high as 70 mm Hg leading to blindness within only a few days.
- Open-angle glaucoma constitutes approximately 90% of all primary glaucomas and is characterized by abnormally high resistance to fluid (aqueous humor) drainage from the eye.
- Normal resistance is required to maintain an intraocular pressure sufficient to maintain the shape of the eye for optical integrity.
- This resistance is provided by the trabecular meshwork, a complex, multilaminar tissue consisting of specialized cells with a dense actomyosin cytoskeletal network, collagenous beams and extracellular matrix.
- the resistance of the trabecular meshwork normally is such that intraocular pressure is ⁇ 16 mm Hg, a pressure at which aqueous humor leaves the eye at the same rate at which it is produced (2.5 ⁇ L/minute). In the glaucomatous eye, the rate of aqueous humor production remains constant, while it is the increased resistance to outflow that is responsible for the elevated intraocular pressure.
- Typical treatments for glaucoma comprise a variety of pharmaceutical approaches for reducing intraocular pressure (IOP), each with their drawbacks.
- IOP intraocular pressure
- Beta-blockers and carbonic anhydrase inhibitors reduce aqueous humor production, which is needed to nourish the avascular lens and corneal endothelial cells, and the prostaglandins effect the uvealscleral outflow pathway, which only accounts for 10% of the total outflow facility.
- Pharmacological agents which target the trabecular meshwork may provide relief to the significant numbers of patients that do not respond adequately to current IOP-lowering medications and/or cannot tolerate the side effects associated with these agents. Additionally, these molecules may prove beneficial as adjunctive therapy in combination with other classes of IOP-lowering medications.
- U.S. Pat. Nos. 6,586,425, 6,110,912, and 5,798,380 disclose a method for the treatment of glaucoma using compounds that affect the actin filament integrity of the eye to enhance aqueous humor outflow.
- These patents also specifically disclose kinase inhibitors as well as latrunculin-A, latrunculin-B, swinholide-A, and jasplakinolide, which cause a perturbation of the actin cytoskeleton and tight junctional complexes in the trabecular meshwork or the modulation of its interactions with the underlying membrane.
- Perturbation of the cytoskeleton and the associated adhesions reduces the resistance of aqueous humor flow through the trabecular meshwork and thereby reduces intraocular pressure.
- Wound healing is another approach in which these classes of molecules can aid in modulating IOP.
- Trabeculectomy is the most common form of glaucoma filtration surgery and remains as the first-line therapy for surgical reduction of pharmacologically uncontrolled intraocular pressure in primary open angle glaucoma. This procedure establishes a limbal fistula through which aqueous humor drains into the subconjunctival space establishing a filtering bleb to lower intraocular pressure. The success of the procedure is highly dependent on pharmacological modulation/inhibition of wound healing.
- a major advance in the surgical management of glaucoma has been the use of antimetabolites to prevent scarring after glaucoma filtration surgery.
- Postoperative scarring of the filtering bleb is the most crucial factor in determining the short and long-term outcome of modern glaucoma filtration surgery.
- the antimetabolites mitomycin C (MMC) and 5-fluorouracil (5-FU) are widely used to suppress scarring and thus failure of the filtering bleb.
- MMC mitomycin C
- 5-fluorouracil 5-fluorouracil
- MMC exhibits a high incidence of severe post-application complications, as does 5-FU; although its side effects mainly affect the corneal epithelium its clinical use is limited by severe pain and discomfort to the patient. No sufficient method has been established to achieve satisfying postoperative long-term surgical results with only minimal or no side effects for the patient.
- ROCK inhibitors impact diverse physiological functions associated with cytoskeletal rearrangement leading to changes in cell morphology, cell contractility, cell motility and cytokinesis. They play a key role in modulating focal adhesion and stress fiber formation in trabecular meshwork cells which express a dense, dynamic cytoskeletal network. Thus, altering the contractility of these cells leads to drainage-surface expansion of the trabecular meshwork and Schlemm's canal.
- the present invention is directed to compounds of Formula I and Formula II, which are inhibitors of rho kinase.
- the present invention is also directed to pharmaceutical compositions comprising such compounds and a pharmaceutically acceptable carrier.
- the present invention is also directed to a method of preventing or treating diseases or conditions associated with cellular relaxation and/or changes in cell-substratum adhesions
- the invention provides a method of reducing intraocular pressure, including treating glaucoma such as primary open-angle glaucoma; a method of treating constriction of the visual field; a method of inhibiting wound healing after trabeculectomy; a method of treating posterior capsule opacification following extracapsular cataract extraction and intraocular lens implantation; a method of inhibiting angiogenesis; a method of modulating fluid transport on the ocular surface; a method of controlling vasospasm; a method of increasing tissue perfusion; a method of neuroprotection; and a method of vasoprotection to atherogenic agents.
- the methods comprise the steps of identifying a subject in need of treatment, and administering to the subject a compound of Formula I or Formula II, in an amount effective to alter the actin cytoskeleton, such as by inhibiting actomyosin interactions.
- FIG. 1 shows the observed aqueous humor concentrations of the test compounds at 0.5, 2, and 4 hours after instillation of the compounds in the animal eyes.
- FIGS. 2-1 to 2 - 3 show the intraocular pressure of animals after treated with the test compounds or vehicle.
- the inventors of the present invention have discovered compounds that are cytoskeletal active agents, which modify cell contractility, cell-cell and cell-substrate interactions, for example by inhibiting actomyosin interactions. These compounds contain structural features that render them suitable for use as therapeutic agents, especially for use in topical formulations, for example for use in the treatment of ophthalmic disorders.
- the structures described herein provide new compounds with therapeutic utility.
- Halo substituents are taken from fluorine, chlorine, bromine, and iodine.
- Alkyl refers to groups of from 1 to 12 carbon atoms inclusively, either straight chained or branched, more preferably from 1 to 8 carbon atoms inclusively, and most preferably 1 to 6 carbon atoms inclusively.
- Alkenyl refers to groups of from 2 to 12 carbon atoms inclusively, either straight or branched containing at least one double bond but optionally containing more than one double bond.
- Alkynyl refers to groups of from 2 to 12 carbon atoms inclusively, either straight or branched containing at least one triple bond but optionally containing more than one triple bond, and additionally optionally containing one or more double bonded moieties.
- Alkoxy refers to the group alkyl-O— wherein the alkyl group is as defined above including optionally substituted alkyl groups as also defined above.
- Alkenoxy refers to the group alkenyl-O— wherein the alkenyl group is as defined above including optionally substituted alkenyl groups as also defined above.
- Alkynoxy refers to the group alkynyl-O— wherein the alkynyl group is as defined above including optionally substituted alkynyl groups as also defined above.
- Aryl refers to an unsaturated aromatic carbocyclic group of from 6 to 14 carbon atoms inclusively having a single ring (e.g., phenyl) or multiple condensed rings (e.g., naphthyl or anthryl). Preferred aryls include phenyl, naphthyl and the like.
- Arylalkyl refers to aryl-alkyl- groups preferably having from 1 to 6 carbon atoms inclusively in the alkyl moiety and from 6 to 10 carbon atoms inclusively in the aryl moiety. Such arylalkyl groups are exemplified by benzyl, phenethyl and the like.
- Arylalkenyl refers to aryl-alkenyl- groups preferably having from 2 to 6 carbon atoms in the alkenyl moiety and from 6 to 10 carbon atoms inclusively in the aryl moiety.
- Arylalkynyl refers to aryl-alkynyl- groups preferably having from 2 to 6 carbon atoms inclusively in the alkynyl moiety and from 6 to 10 carbon atoms inclusively in the aryl moiety.
- Cycloalkyl refers to cyclic alkyl groups of from 3 to 12 carbon atoms inclusively having a single cyclic ring or multiple condensed rings which can be optionally substituted with from 1 to 3 alkyl groups.
- Such cycloalkyl groups include, by way of example, single ring structures such as cyclopropyl, cyclobutyl, cyclopentyl, cyclooctyl, 1-methylcyclopropyl, 2-methylcyclopentyl, 2-methylcyclooctyl, and the like, or multiple ring structures such as adamantyl, and the like.
- Cycloalkenyl refers to cyclic alkenyl groups of from 4 to 12 carbon atoms inclusively having a single cyclic ring or multiple condensed rings and at least one point of internal unsaturation, which can be optionally substituted with from 1 to 3 alkyl groups.
- suitable cycloalkenyl groups include, for instance, cyclobut-2-enyl, cyclopent-3-enyl, cyclooct-3-enyl and the like.
- Cycloalkylalkyl refers to cycloalkyl-alkyl- groups preferably having from 1 to 6 carbon atoms inclusively in the alkyl moiety and from 6 to 10 carbon atoms inclusively in the cycloalkyl moiety. Such cycloalkylalkyl groups are exemplified by cyclopropylmethyl, cyclohexylethyl and the like.
- Cycloalkylalkenyl refers to cycloalkyl-alkenyl- groups preferably having from 2 to 6 carbon atoms inclusively in the alkenyl moiety and from 6 to 10 carbon atoms inclusively in the cycloalkyl moiety. Such cycloalkylalkenyl groups are exemplified by cyclohexylethenyl and the like.
- Cycloalkylalkynyl refers to cycloalkyl-alkynyl- groups preferably having from 2 to 6 carbon atoms inclusively in the alkynyl moiety and from 6 to 10 carbon atoms inclusively in the cycloalkyl moiety. Such cycloalkylalkynyl groups are exemplified by cyclopropylethynyl and the like.
- Heteroaryl refers to a monovalent aromatic heterocyclic group of from 1 to 10 carbon atoms inclusively and 1 to 4 heteroatoms inclusively selected from oxygen, nitrogen and sulfur within the ring.
- Such heteroaryl groups can have a single ring (e.g., pyridyl or furyl) or multiple condensed rings (e.g., indolizinyl or benzothienyl).
- Heteroarylalkyl refers to heteroaryl-alkyl- groups preferably having from 1 to 6 carbon atoms inclusively in the alkyl moiety and from 6 to 10 atoms inclusively in the heteroaryl moiety. Such heteroarylalkyl groups are exemplified by pyridylmethyl and the like.
- Heteroarylalkenyl refers to heteroaryl-alkenyl- groups preferably having from 2 to 6 carbon atoms inclusively in the alkenyl moiety and from 6 to 10 atoms inclusively in the heteroaryl moiety.
- Heteroarylalkynyl refers to heteroaryl-alkynyl- groups preferably having from 2 to 6 carbon atoms inclusively in the alkynyl moiety and from 6 to 10 atoms inclusively in the heteroaryl moiety.
- Heterocycle refers to a saturated or unsaturated group having a single ring or multiple condensed rings, from 1 to 8 carbon atoms inclusively and from 1 to 4 hetero atoms inclusively selected from nitrogen, sulfur or oxygen within the ring.
- Such heterocyclic groups can have a single ring (e.g., piperidinyl or tetrahydrofuryl) or multiple condensed rings (e.g., indolinyl, dihydrobenzofuran or quinuclidinyl).
- Preferred heterocycles include piperidinyl, pyrrolidinyl and tetrahydrofuryl.
- Heterocycle-alkyl refers to heterocycle-alkyl- groups preferably having from 1 to 6 carbon atoms inclusively in the alkyl moiety and from 6 to 10 atoms inclusively in the heterocycle moiety. Such heterocycle-alkyl groups are exemplified by morpholino-ethyl, pyrrolidinylmethyl, and the like.
- Heterocycle-alkenyl refers to heterocycle-alkenyl- groups preferably having from 2 to 6 carbon atoms inclusively in the alkenyl moiety and from 6 to 10 atoms inclusively in the heterocycle moiety.
- Heterocycle-alkynyl refers to heterocycle-alkynyl- groups preferably having from 2 to 6 carbon atoms inclusively in the alkynyl moiety and from 6 to 10 atoms inclusively in the heterocycle moiety.
- heterocycles and heteroaryls include, but are not limited to, furan, thiophene, thiazole, oxazole, pyrrole, imidazole, pyrazole, pyridine, pyrazine, pyrimidine, pyridazine, indolizine, isoindole, indole, indazole, purine, quinolizine, isoquinoline, quinoline, phthalazine, naphthylpyridine, quinoxaline, quinazoline, cinnoline, pteridine, carbazole, carboline, phenanthridine, acridine, phenanthroline, isothiazole, phenazine, isoxazole, phenoxazine, phenothiazine, imidazolidine, imidazoline, piperidine, piperazine, pyrrolidine, indoline and the like.
- positions occupied by hydrogen in the foregoing groups can be further substituted with substituents exemplified by, but not limited to, hydroxy, oxo, nitro, methoxy, ethoxy, alkoxy, substituted alkoxy, trifluoromethoxy, haloalkoxy, fluoro, chloro, bromo, iodo, halo, methyl, ethyl, propyl, butyl, alkyl, alkenyl, alkynyl, substituted alkyl, trifluoromethyl, haloalkyl, hydroxyalkyl, alkoxyalkyl, thio, alkylthio, acyl, carboxy, alkoxycarbonyl, carboxamido, substituted carboxamido, alkylsulfonyl, alkylsulfinyl, alkylsulfonylamino, sulfonamido, substituted sulfonamido
- heteroatom-containing substituent refers to substituents containing at least one non-halogen heteroatom.
- substituents include, but are not limited to, hydroxy, oxo, nitro, methoxy, ethoxy, alkoxy, substituted alkoxy, trifluoromethoxy, haloalkoxy, hydroxyalkyl, alkoxyalkyl, thio, alkylthio, acyl, carboxy, alkoxycarbonyl, carboxamido, substituted carboxamido, alkylsulfonyl, alkylsulfonylamino, sulfonamido, substituted sulfonamido, cyano, amino, substituted amino, alkylamino, dialkylamino, aminoalkyl, acylamino, amidino, amidoximo, hydroxamoyl, aryloxy, pyridyl, imidazolyl, heteroaryl, substituted hetero
- “Pharmaceutically acceptable salts” are salts that retain the desired biological activity of the parent compound and do not impart undesired toxicological effects.
- Pharmaceutically acceptable salt forms include various polymorphs as well as the amorphous form of the different salts derived from acid or base additions.
- the acid addition salts can be formed with inorganic or organic acids.
- Such acids include hydrochloric, hydrobromic, sulfuric, phosphoric, citric, acetic, propionic, benzoic, napthoic, oxalic, succinic, maleic, fumaric, malic, adipic, lactic, tartaric, salicylic, methanesulfonic, 2-hydroxyethanesulfonic, toluenesulfonic, benzenesulfonic, camphorsulfonic, and ethanesulfonic acids.
- the pharmaceutically acceptable base addition salts can be formed with metal or organic counterions and include, but are not limited to, alkali metal salts such as sodium or potassium; alkaline earth metal salts such as magnesium or calcium; and ammonium or tetraalkyl ammonium salts, i.e., NX 4 + (wherein X is C 1-4 .
- Tautomers are compounds that can exist in one or more forms, called tautomeric forms, which can interconvert by way of a migration of one or more hydrogen atoms in the compound accompanied by a rearrangement in the position of adjacent double bonds. These tautomeric forms are in equilibrium with each other, and the position of this equilibrium will depend on the exact nature of the physical state of the compound. It is understood that where tautomeric forms are possible, the current invention relates to all possible tautomeric forms.
- Solidvates are addition complexes in which a compound of Formula I or Formula II is combined with a pharmaceutically acceptable cosolvent in some fixed proportion.
- Cosolvents include, but are not limited to, water, methanol, ethanol, 1-propanol, isopropanol, 1-butanol, isobutanol, tert-butanol, acetone, methyl ethyl ketone, acetonitrile, ethyl acetate, benzene, toulene, xylene(s), ethylene glycol, dichloromethane, 1,2-dichloroethane, N-methylformamide, N,N-dimethylformamide, N-methylacetamide, pyridine, dioxane, and diethyl ether. Hydrates are solvates in which the cosolvent is water. It is to be understood that the definitions of compounds in Formula I and Formula II encompass all possible hydrates and solvates, in any proportion, which possess the stated
- the rho kinase inhibitor compounds useful for this invention include compounds of general Formula I and Formula II, and/or tautomers thereof, and/or pharmaceutically-acceptable salts, and/or solvates, and/or hydrates thereof.
- a compound according to Formula I or Formula II can exist in several diastereomeric forms.
- the general structures of Formula I and Formula II include all diastereomeric forms of such materials, when not specified otherwise.
- Formula I and Formula II also include mixtures of compounds of these Formulae, including mixtures of enantiomers, diastereomers and/or other isomers in any proportion.
- R 1 is aryl or heteroaryl, optionally substituted;
- Q is C ⁇ O, SO 2 , or (CR 4 R 5 ) n3 ;
- n 1 is 1, 2, or 3;
- n 2 is 1 or 2;
- n 3 is 0, 1, 2, or 3; wherein the ring represented by
- R 2 is selected from the following heteroaryl systems, optionally substituted:
- R 3 -R 7 are independently H, alkyl, alkenyl, alkynyl, cycloalkyl, cycloalkenyl, cycloalkylalkyl, cycloalkylalkenyl, or cycloalkylalkynyl optionally substituted.
- the preferred R 1 is substituted aryl, the more preferred R 1 is substituted phenyl, the preferred Q is (CR 4 R 5 ) n3 , the more preferred Q is CH 2 , the preferred n 1 is 1 or 2, the preferred n 2 is 1, the preferred n 3 is 1 or 2, and the preferred R 3 -R 7 are H.
- R 2 is 5-indazolyl or 6-indazolyl (R 2 -1), optionally substituted.
- R 2 -1 is substituted by one or more alkyl or halo substituents.
- R 2 -1 is substituted by one or more amino, alkylamino, hydroxyl, or alkoxy substituents.
- R 2 -1 is unsubstituted.
- the invention is represented by Formula I in which R 2 is 5-isoquinolinyl or 6-isoquinolinyl (R 2 -2), optionally substituted.
- R 2 -2 is substituted by one or more alkyl or halo substituents.
- R 2 -2 is substituted by one or more amino, alkylamino, hydroxyl, or alkoxy substituents.
- R 2 -2 is unsubstituted.
- the invention is represented by Formula I in which R 2 is 4-pyridyl or 3-pyridyl (R 2 -3), optionally substituted.
- R 2 -3 is substituted by one or more alkyl or halo substituents.
- R 2 -3 is substituted by one or more amino, alkylamino, hydroxyl, or alkoxy substituents.
- R 2 -3 is unsubstituted.
- the invention is represented by Formula I in which R 2 is 7-azaindol-4-yl or 7-azaindol-5-yl (R 2 -4), optionally substituted.
- R 2 -4 is substituted by one or more alkyl or halo substituents.
- R 2 -4 is substituted by one or more amino, alkylamino, hydroxyl, or alkoxy substituents.
- R 2 -4 is unsubstituted.
- the invention is represented by Formula I in which R 2 is 4-(3-amino-1,2,5-oxadiazol-4-yl)phenyl or 3-(3-amino-1,2,5-oxadiazol-4-yl)phenyl (R 2 -5), optionally substituted.
- R 2 -5 is unsubstituted.
- the invention is represented by Formula I in which R 2 is one of the groups R 2 -1-R 2 -5, substituted by one or more alkyl, halo, amino, alkylamino, hydroxyl, or alkoxy substituents.
- R 2 is substituted by one or more alkyl or halo substituents.
- R 2 is substituted by one or more amino, alkylamino, hydroxyl, or alkoxy substituents.
- the invention is represented by Formula I in which R 2 is one of the groups R 2 -1-R 2 -5, and is unsubstituted.
- the invention is represented by Formula I in which Q is (CR 4 R 5 ) n3 , and n 3 is 1 or 2.
- the invention is represented by Formula I in which Q is (CH 2 ) n3 , and n 3 is 1.
- the invention is represented by Formula I in which R 1 is aryl or heteroaryl substituted with one or more alkenyl, alkynyl, aryl, arylalkyl, arylalkenyl, arylalkynyl, heteroaryl, heteroarylalkyl, heteroarylalkenyl, heteroarylalkynyl, cycloalkyl, cycloalkenyl, cycloalkylalkyl, cycloalkylalkenyl, cycloalkylalkynyl, heterocycle, (heterocycle)alkyl, (heterocycle)alkenyl, or (heterocycle)alkynyl substituents, optionally further substituted.
- Compounds exemplifying embodiment 11 include compounds 1.009, 1.010, 1.011, 1.012, 1.020, 1.021, 1.030, 1.034, 1.037, 1.044, 1.047, 1.076, 1.077, 1.083, 2.010, 2.011, 2.019, 2.020, 2.022, 2.023, and 2.031, shown below in Table I.
- the invention is represented by Formula I in which R 1 is aryl or heteroaryl substituted with one or more heteroatom-containing substituents, with the proviso that if the R 1 substituent is acyclic and is connected to R 1 by a carbon atom, then this substituent contains at least one nitrogen or sulfur atom, with the second proviso that if the substituent is acyclic and is connected to R 1 by an oxygen or nitrogen atom, then this substituent contains at least one additional oxygen, nitrogen or sulfur atom, and with the third proviso that if the substituent is connected to R 1 by a sulfone linkage “—SO 2 —”, then R 2 is not nitrogen- or oxygen-substituted R 2 -2.
- the heteroatom-containing substituent is connected to R 1 by an oxygen or nitrogen atom.
- the heteroatom-containing substituent is connected to R 1 by a sulfide linkage, “—S—”.
- Compounds exemplifying embodiment 12 include compounds 1.001, 1.002, 1.004, 1.005, 1.038, 1.048, 1.055, 1.056, 2.002, 2.003, 2.005, 2.007, 1.003, 1.006, 1.007, 1.018, 1.039, 1.051, 1.058, 1.060, 1.084, 1.085, 1.086, 1.087, 1.088, 1.090, 1.091, 1.092, 1.093, 1.094, 1.095, 1.096, 1.097, 1.098, 1.102, 1.111, 1.113, 1.115, 1.116, 1.117, 1.118, 1.120, 1.121, 1.123, 1.124, 1.125, 1.126, 1.127, 1.128, 1.129, 1.130, 2.004, 2.008, 2.032, 2.033, 2.034, 2.035, 2.036, 2.037, 2.038, 2.039, 2.040, 2.041, 2.042, 2.043, 2.044, 1.008, 1.017, 1.026, 1.0
- the invention is represented by Formula I in which R 1 is aryl or heteroaryl substituted with one or more alkyl, alkenyl, alkynyl, aryl, arylalkyl, arylalkenyl, arylalkynyl, heteroaryl, heteroarylalkyl, heteroarylalkenyl, heteroarylalkynyl, cycloalkyl, cycloalkenyl, cycloalkylalkyl, cycloalkylalkenyl, cycloalkylalkynyl, heterocycle, (heterocycle)alkyl, (heterocycle)alkenyl, or (heterocycle)alkynyl substituents, which are further substituted with one or more heteroatom-containing substituents, with the proviso that if the R 1 substituent is acyclic and its heteroatom-containing substituent falls on the carbon by which it is attached to R 1 , then the heteroatom-containing substituents, which are further
- Compounds exemplifying embodiment 13 include compounds 1.019, 1.027, 1.028, 1.029, 1.035, 1.041, 1.042, 1.043, 1.057, 1.061, 1.099, 1.101, 1.103, 1.104, 1.105, 1.106, 1.107, 1.108, 1.109, 1.112, 1.114, 1.119, and 1.122, shown below in Table I.
- the invention is represented by Formula I in which R 1 is aryl or heteroaryl substituted with one or more alkenyl, alkynyl, aryl, arylalkyl, arylalkenyl, arylalkynyl, heteroaryl, heteroarylalkyl, heteroarylalkenyl, heteroarylalkynyl, cycloalkyl, cycloalkenyl, cycloalkylalkyl, cycloalkylalkenyl, cycloalkylalkynyl, heterocycle, (heterocycle)alkyl, (heterocycle)alkenyl, or (heterocycle)alkynyl substituents, optionally further substituted, and R 2 is 5-indazolyl (R 2 -1) or 5-isoquinolinyl (R 2 -2), optionally substituted.
- R 2 is 5-indazolyl (R 2 -1), optionally substituted by one or more alkyl, halo, amino, alkylamino, hydroxyl, or alkoxy substituents.
- R 2 is 5-isoquinolinyl (R 2 -2), optionally substituted by one or more alkyl, halo, amino, alkylamino, hydroxyl, or alkoxy substituents.
- R 2 is unsubstituted.
- Compounds exemplifying embodiment 14 include compounds 1.009, 1.010, 1.011, 1.012, 1.020, 1.021, 1.030, 1.034, 1.037, 1.044, 1.047, 1.076, 1.077, 1.083, 2.010, 2.011, 2.019, 2.020, 2.022, 2.023, and 2.031, shown below in Table I.
- the invention is represented by Formula I in which R 1 is aryl or heteroaryl substituted with one or more heteroatom-containing substituents, and R 2 is 5-indazolyl (R 2 -1) or 5-isoquinolinyl (R 2 -2), optionally substituted, with the proviso that if the R 1 substituent is acyclic and is connected to R 1 by a carbon atom, then this substituent contains at least one nitrogen or sulfur atom, with the second proviso that if the substituent is acyclic and is connected to R 1 by an oxygen or nitrogen atom, then this substituent contains at least one additional oxygen, nitrogen or sulfur atom, and with the third proviso that if the substituent is connected to R 1 by a sulfone linkage “—SO 2 —”, then R 2 is not nitrogen- or oxygen-substituted R 2 -2.
- R 2 is 5-indazolyl (R 2 -1), optionally substituted by one or more alkyl, halo, amino, alkylamino, hydroxyl, or alkoxy substituents.
- R 2 is 5-isoquinolinyl (R 2 -2), optionally substituted by one or more alkyl, halo, amino, alkylamino, hydroxyl, or alkoxy substituents.
- R 2 is unsubstituted.
- the heteroatom-containing substituent is connected to R 1 by an oxygen or nitrogen atom.
- the heteroatom-containing substituent is connected to R 1 by a sulfide linkage, “—S—”.
- Compounds exemplifying embodiment 15 include compounds 1.001, 1.002, 1.004, 1.005, 1.038, 1.048, 1.055, 1.056, 2.002, 2.003, 2.005, 2.007, 1.003, 1.006, 1.007, 1.018, 1.039, 1.051, 1.058, 1.060, 1.084, 1.085, 1.086, 1.087, 1.088, 1.090, 1.091, 1.092, 1.093, 1.094, 1.095, 1.096, 1.097, 1.098, 1.102, 1.111, 1.113, 1.115, 1.116, 1.117, 1.118, 1.120, 1.121, 1.123, 1.124, 1.125, 1.126, 1.127, 1.128, 1.129, 1.130, 2.004, 2.008, 2.032, 2.033, 2.034, 2.035, 2.036, 2.037, 2.038, 2.039, 2.040, 2.041, 2.042, 2.043, 2.044, 1.008, 1.017, 1.026, 1.0
- the invention is represented by Formula I in which R 1 is aryl or heteroaryl substituted with one or more alkyl, alkenyl, alkynyl, aryl, arylalkyl, arylalkenyl, arylalkynyl, heteroaryl, heteroarylalkyl, heteroarylalkenyl, heteroarylalkynyl, cycloalkyl, cycloalkenyl, cycloalkylalkyl, cycloalkylalkenyl, cycloalkylalkynyl, heterocycle, (heterocycle)alkyl, (heterocycle)alkenyl, or (heterocycle)alkynyl substituents, at least one of which is further substituted with one or more heteroatom-containing substituents, and R 2 is 5-indazolyl (R 2 -1) or 5-isoquinolinyl (R 2 -2), optionally substituted, with the proviso that
- R 2 is 5-indazolyl (R 2 -1), optionally substituted by one or more alkyl, halo, amino, alkylamino, hydroxyl, or alkoxy substituents.
- R 2 is 5-isoquinolinyl (R 2 -2), optionally substituted by one or more alkyl, halo, amino, alkylamino, hydroxyl, or alkoxy substituents.
- R 2 is unsubstituted.
- Compounds exemplifying embodiment 16 include compounds 1.019, 1.027, 1.028, 1.029, 1.035, 1.041, 1.042, 1.043, 1.057, 1.061, 1.099, 1.101, 1.103, 1.104, 1.105, 1.106, 1.107, 1.108, 1.109, 1.112, 1.114, 1.119, and 1.122, shown below in Table I.
- Ar is a monocyclic or bicyclic aryl or heteroaryl ring, such as phenyl;
- X is from 1 to 3 substituents on Ar, each independently in the form Y—Z, in which Z is attached to Ar;
- Y is one or more substituents on Z, and each is chosen independently from H, halogen, or the heteroatom-containing substituents, including but not limited to OR 8 , NR 8 R 9 , NO 2 , SR 8 , SOR 8 , SO 2 R 8 , SO 2 NR 8 R 9 , NR 8 SO 2 R 9 , OCF 3 , CONR 8 R 9 , NR 8 C( ⁇ O)R 9 , NR 8 C( ⁇ O)OR 9 , OC( ⁇ O)NR 8 R 9 , or NR 8 C( ⁇ O)NR 9 R 10 ;
- Z is chosen independently from alkyl, alkenyl, alkynyl, aryl, arylalkyl, arylalkenyl
- the preferred Y is H, halogen, OR 8 , NR 8 R 9 , NO 2 , SR 8 , SOR 8 , SO 2 R 8 , SO 2 NR 8 R 9 , NR 8 SO 2 R 9 , OCF 3 , CONR 8 R 9 , NR 8 C( ⁇ O)R 9 , NR 8 C( ⁇ O)OR 9 , OC( ⁇ O)NR 8 R 9 , or NR 8 C( ⁇ O)NR 9 R 10
- the more preferred Y is H, halogen, OR 8 , SR 8 , SOR 8 , SO 2 R 8 , SO 2 NR 8 R 9 , NR 8 SO 2 R 9 , CONR 8 R 9 , or NR 8 C( ⁇ O)NR 9 R 10
- the preferred Z is alkyl, alkenyl, alkynyl, cycloalkyl, cycloalkenyl, cycloalkylalkyl, or is absent; the more preferred Z is alkyl
- R 2 is 5-indazolyl or 6-indazolyl (R 2 -1), optionally substituted.
- R 2 -1 is substituted by one or more alkyl or halo substituents.
- R 2 -1 is substituted by one or more amino, alkylamino, hydroxyl, or alkoxy substituents.
- R 2 -1 is unsubstituted.
- the invention is represented by Formula II in which R 2 is 5-isoquinolinyl or 6-isoquinolinyl (R 2 -2), optionally substituted.
- R 2 -2 is substituted by one or more alkyl or halo substituents.
- R 2 -2 is substituted by one or more amino, alkylamino, hydroxyl, or alkoxy substituents.
- R 2 -2 is unsubstituted.
- the invention is represented by Formula II in which R 2 is 4-pyridyl or 3-pyridyl (R 2 -3), optionally substituted.
- R 2 -3 is substituted by one or more alkyl or halo substituents.
- R 2 -3 is substituted by one or more amino, alkylamino, hydroxyl, or alkoxy substituents.
- R 2 -3 is unsubstituted.
- the invention is represented by Formula II in which R 2 is 7-azaindol-4-yl or 7-azaindol-5-yl (R 2 -4), optionally substituted.
- R 2 -4 is substituted by one or more alkyl or halo substituents.
- R 2 -4 is substituted by one or more amino, alkylamino, hydroxyl, or alkoxy substituents.
- R 2 -4 is unsubstituted.
- the invention is represented by Formula II in which R 2 is 4-(3-amino-1,2,5-oxadiazol-4-yl)phenyl or 3-(3-amino-1,2,5-oxadiazol-4-yl)phenyl (R 2 -5), optionally substituted.
- R 2 -5 is unsubstituted.
- the invention is represented by Formula II in which R 2 is one of the groups R 2 -1-R 2 -5, substituted by one or more alkyl, halo, amino, alkylamino, hydroxyl, or alkoxy substituents.
- R 2 is substituted by one or more alkyl or halo substituents.
- R 2 is substituted by one or more amino, alkylamino, hydroxyl, or alkoxy substituents.
- the invention is represented by Formula II in which R 2 is one of the groups R 2 -1-R 2 -5, and is unsubstituted.
- the invention is represented by Formula II in which Q is (CR 4 R 5 ) n3 , and n 3 is 1 or 2.
- the invention is represented by Formula II in which Q is (CH 2 ) n3 , and n 3 is 1.
- the invention is represented by Formula II in which Z is alkenyl, alkynyl, aryl, arylalkyl, arylalkenyl, arylalkynyl, heteroaryl, heteroarylalkyl, heteroarylalkenyl, heteroarylalkynyl, cycloalkyl, cycloalkylalkenyl, cycloalkylalkynyl, cycloalkenyl, cycloalkylalkyl, heterocycle, (heterocycle)alkyl, (heterocycle)alkenyl, or (heterocycle)alkynyl.
- Compounds exemplifying embodiment 11 include compounds 1.009, 1.010, 1.011, 1.012, 1.020, 1.021, 1.030, 1.034, 1.037, 1.044, 1.047, 1.076, 1.077, 1.083, 2.010, 2.011, 2.019, 2.020, 2.022, 2.023, and 2.031, shown below in Table I.
- the invention is represented by Formula II in which Z is absent, Y is a heteroatom-containing substituent, including but not limited to OR 8 , NR 8 R 9 , SR 8 , SOR 8 , SO 2 R 8 , SO 2 NR 8 R 9 , NR 8 SO 2 R 9 , CONR 8 R 9 , NR 8 C( ⁇ O)R 9 , NR 8 C( ⁇ O)OR 9 , OC( ⁇ O)NR 8 R 9 , or NR 8 C( ⁇ O)NR 9 R 10 , with the proviso that if the substituent Y is acyclic and is connected to Ar by a carbon atom, then this substituent contains at least one nitrogen or sulfur atom, with the second proviso that if the substituent Y is acyclic and is connected to Ar by an oxygen or nitrogen atom, then this substituent contains at least one additional oxygen, nitrogen or sulfur atom, and with the third proviso that if the substituent Y is connected to Ar by a
- the heteroatom-containing substituent is connected to R 1 by an oxygen or nitrogen atom.
- the heteroatom-containing substituent is connected to R 1 by a sulfide linkage, “—S—”.
- Compounds exemplifying embodiment 12 include compounds 1.001, 1.002, 1.004, 1.005, 1.038, 1.048, 1.055, 1.056, 2.002, 2.003, 2.005, 2.007, 1.003, 1.006, 1.007, 1.018, 1.039, 1.051, 1.058, 1.060, 1.084, 1.085, 1.086, 1.087, 1.088, 1.090, 1.091, 1.092, 1.093, 1.094, 1.095, 1.096, 1.097, 1.098, 1.102, 1.111, 1.113, 1.115, 1.116, 1.117, 1.118, 1.120, 1.121, 1.123, 1.124, 1.125, 1.126, 1.127, 1.128, 1.129, 1.130, 2.004, 2.008, 2.032, 2.033, 2.034, 2.035, 2.036, 2.037, 2.038, 2.039, 2.040, 2.041, 2.042, 2.043, 2.044, 1.008, 1.017, 1.026, 1.0
- the invention is represented by Formula II in which Z is alkyl, alkenyl, alkynyl, aryl, arylalkyl, arylalkenyl, arylalkynyl, heteroaryl, heteroarylalkyl, heteroarylalkenyl, heteroarylalkynyl, cycloalkyl, cycloalkenyl, cycloalkylalkyl, cycloalkylalkenyl, cycloalkylalkynyl, heterocycle, (heterocycle)alkyl, (heterocycle)alkenyl, or (heterocycle)alkynyl, and Y is a heteroatom-containing substituent, including but not limited to OR 8 , NR 8 R 9 , NO 2 , SR 8 , SOR 8 , SO 2 R 8 , SO 2 NR 8 R 9 , NR 8 SO 2 R 9 , OCF 3 , CONR 8 R 9 , NR
- Compounds exemplifying embodiment 13 include compounds 1.019, 1.027, 1.028, 1.029, 1.035, 1.041, 1.042, 1.043, 1.057, 1.061, 1.099, 1.101, 1.103, 1.104, 1.105, 1.106, 1.107, 1.108, 1.109, 1.112, 1.114, 1.119, and 1.122, shown below in Table I.
- the invention is represented by Formula II in which Z is alkenyl, alkynyl, aryl, arylalkyl, arylalkenyl, arylalkynyl, heteroaryl, heteroarylalkyl, heteroarylalkenyl, heteroarylalkynyl, cycloalkyl, cycloalkenyl, cycloalkylalkyl, cycloalkylalkenyl, cycloalkylalkynyl, heterocycle, (heterocycle)alkyl, (heterocycle)alkenyl, or (heterocycle)alkynyl, and R 2 is 5-indazolyl (R 2 -1) or 5-isoquinolinyl (R 2 -2), optionally substituted.
- R 2 is 5-indazolyl (R 2 -1), optionally substituted by one or more alkyl, halo, amino, alkylamino, hydroxyl, or alkoxy substituents.
- R 2 is 5-isoquinolinyl (R 2 -2), optionally substituted by one or more alkyl, halo, amino, alkylamino, hydroxyl, or alkoxy substituents.
- R 2 is unsubstituted.
- Compounds exemplifying embodiment 14 include compounds 1.009, 1.010, 1.011, 1.012, 1.020, 1.021, 1.030, 1.034, 1.037, 1.044, 1.047, 1.076, 1.077, 1.083, 2.010, 2.011, 2.019, 2.020, 2.022, 2.023, and 2.031, shown below in Table I.
- the invention is represented by Formula II in which Z is absent, Y is a heteroatom-containing substituent, including but not limited to OR 8 , NR 8 R 9 , SR 8 , SOR 8 , SO 2 R 8 , SO 2 NR 8 R 9 , NR 8 SO 2 R 9 , CONR 8 R 9 , NR 8 C( ⁇ O)R 9 , NR 8 C( ⁇ O)OR 9 , OC( ⁇ O)NR 8 R 9 , or NR 8 C( ⁇ O)NR 9 R 10 , and R 2 is 5-indazolyl (R 2 -1) or 5-isoquinolinyl (R 2 -2), optionally substituted, with the proviso that if the substituent Y is acyclic and is connected to Ar by a carbon atom, then this substituent contains at least one nitrogen or sulfur atom, with the second proviso that if the substituent Y is acyclic and is connected to Ar by an oxygen or nitrogen atom, then this substituent
- R 2 is 5-indazolyl (R 2 -1), optionally substituted by one or more alkyl, halo, amino, alkylamino, hydroxyl, or alkoxy substituents.
- R 2 is 5-isoquinolinyl (R 2 -2), optionally substituted by one or more alkyl, halo, amino, alkylamino, hydroxyl, or alkoxy substituents.
- R 2 is unsubstituted.
- the heteroatom-containing substituent is connected to R 1 by an oxygen or nitrogen atom.
- the heteroatom-containing substituent is connected to R 1 by a sulfide linkage, “—S—”.
- Compounds exemplifying embodiment 15 include compounds 1.001, 1.002, 1.004, 1.005, 1.038, 1.048, 1.055, 1.056, 2.002, 2.003, 2.005, 2.007, 1.003, 1.006, 1.007, 1.018, 1.039, 1.051, 1.058, 1.060, 1.084, 1.085, 1.086, 1.087, 1.088, 1.090, 1.091, 1.092, 1.093, 1.094, 1.095, 1.096, 1.097, 1.098, 1.102, 1.111, 1.113, 1.115, 1.116, 1.117, 1.118, 1.120, 1.121, 1.123, 1.124, 1.125, 1.126, 1.127, 1.128, 1.129, 1.130, 2.004, 2.008, 2.032, 2.033, 2.034, 2.035, 2.036, 2.037, 2.038, 2.039, 2.040, 2.041, 2.042, 2.043, 2.044, 1.008, 1.017, 1.026, 1.0
- the invention is represented by Formula II in which Z is alkyl, alkenyl, alkynyl, aryl, arylalkyl, arylalkenyl, arylalkynyl, heteroaryl, heteroarylalkyl, heteroarylalkenyl, heteroarylalkynyl, cycloalkyl, cycloalkenyl, cycloalkylalkyl, cycloalkylalkenyl, cycloalkylalkynyl, heterocycle, (heterocycle)alkyl, (heterocycle)alkenyl, or (heterocycle)alkynyl, and Y is a heteroatom-containing substituent, including but not limited to OR 8 , NR 8 R 9 , NO 2 , SR 8 , SOR 8 , SO 2 R 8 , SO 2 NR 8 R 9 , NR 8 SO 2 R 9 , OCF 3 , CONR 8 R 9 , NR
- R 2 is 5-indazolyl (R 2 -1), optionally substituted by one or more alkyl, halo, amino, alkylamino, hydroxyl, or alkoxy substituents.
- R 2 is 5-isoquinolinyl (R 2 -2), optionally substituted by one or more alkyl, halo, amino, alkylamino, hydroxyl, or alkoxy substituents.
- R 2 is unsubstituted.
- Compounds exemplifying embodiment 16 include compounds 1.019, 1.027, 1.028, 1.029, 1.035, 1.041, 1.042, 1.043, 1.057, 1.061, 1.099, 1.101, 1.103, 1.104, 1.105, 1.106, 1.107, 1.108, 1.109, 1.112, 1.114, 1.119, and 1.122, shown below in Table I.
- the preferred Q is (CR 4 R 5 ) n3 , the more preferred Q is CH 2 , the preferred n 1 is 1 or 2, the preferred n 2 is 1, the preferred n 3 is 1 or 2, and the preferred R 3 is H.
- the present compounds are useful for ophthalmic use, particularly in reducing intraocular pressure or treating glaucoma.
- the compounds must have both adequate potency and proper pharmacokinetic properties such as good permeability across the ocular surface.
- compounds bearing polar functionality have preferred absorption properties and are particularly suitable for topical optical use.
- compounds bearing small lipophilic functional groups have good ROCK inhibitory potency.
- the inventors have discovered that the R 1 substitution in Formula I and X in Formula II are important factors for pharmacokinetic properties and ROCK inhibitory potency.
- the inventors have optimized and selected compounds that have improved ocular permeability and ROCK inhibitory potency.
- compounds bearing polar functionality especially those specified in the embodiments 11, 12, 13, 14, 15, and 16 in Formulae I and II, above, are particularly suitable for topical optical use with adequate ROCK inhibiting activity.
- Compounds bearing small lipophilic functional groups, as specified in the embodiments 11, 12, 13, 14, 15, and 16 in Formulae I and II, above, display ROCK inhibition with adequate ocular permeability.
- the present invention is additionally directed to procedures for preparing compounds of Formula I and Formula II.
- General approaches for preparations of the compounds of the Formulae are described in Scheme 1 and Scheme 2.
- Those having skill in the art will recognize that the starting materials can be varied and additional steps can be employed to produce compounds encompassed by the present invention.
- protection of certain reactive functionalities may be necessary to achieve some of the above transformations.
- the need for such protecting groups as well as the conditions necessary to attach and remove such groups will be apparent to those skilled in the art of organic synthesis.
- Typical example coupling reactions with 1.5 include reductive amination with an aldehyde, alkylation with an alkyl halide, and acylation with an acyl halide or sulfonyl halide. This coupling reaction provides compound 1.6, an example of the substances described by the Formulae.
- Appropriate protection of interfering function groups can be important for obtaining satisfactory reaction of 2.1 and 2.2 to give 2.3.
- protection of any unsubstituted indazole nitrogen is critical to the success of the reaction.
- Preferred protecting groups in this situation are p-methoxybenzyl (PMB) and 2-tetrahydropyranyl (THP), with THP being most preferred.
- PMB p-methoxybenzyl
- THP 2-tetrahydropyranyl
- Use of the THP protecting group provides high yields in the protection, coupling, and deprotection steps, and allows the protecting group to be removed without the need for scavenger reagents, which are otherwise needed for clean deprotection.
- the present invention also provides pharmaceutical compositions.
- the pharmaceutical compositions are pharmaceutically acceptable formulations comprising a pharmaceutically acceptable carrier and one or more compounds of Formula I and/or Formula II, pharmaceutically acceptable salts, solvates, and/or hydrates thereof.
- the pharmaceutically acceptable carrier can be selected by those skilled in the art using conventional criteria.
- Pharmaceutically acceptable carriers include, but are not limited to, aqueous- and non-aqueous based solutions, suspensions, emulsions, microemulsions, micellar solutions, gels, and ointments.
- the pharmaceutically active carriers may also contain ingredients that include, but are not limited to, saline and aqueous electrolyte solutions; ionic and nonionic osmotic agents such as sodium chloride, potassium chloride, glycerol, and dextrose; pH adjusters and buffers such as salts of hydroxide, hydronium, phosphate, citrate, acetate, borate, and tromethamine; antioxidants such as salts, acids and/or bases of bisulfite, sulfite, metabisulfite, thiosulfite, ascorbic acid, acetyl cysteine, cystein, glutathione, butylated hydroxyanisole, butylated hydroxytoluene, tocopherols, and ascorbyl palmitate; surfactants such as lecithin, phospholipids, including but not limited to phosphatidylcholine, phosphatidylethanolamine and phosphatidyl inositiol
- Such pharmaceutically acceptable carriers may be preserved against bacterial contamination using well-known preservatives, these include, but are not limited to, benzalkonium chloride, ethylene diamine tetra-acetic acid and its salts, benzethonium chloride, chlorhexidine, chlorobutanol, methylparaben, thimerosal, and phenylethyl alcohol, or may be formulated as a non-preserved formulation for either single or multiple use.
- preservatives include, but are not limited to, benzalkonium chloride, ethylene diamine tetra-acetic acid and its salts, benzethonium chloride, chlorhexidine, chlorobutanol, methylparaben, thimerosal, and phenylethyl alcohol, or may be formulated as a non-preserved formulation for either single or multiple use.
- the compositions are formulated as topical ophthalmic preparations, with a pH of about 3-9, preferably 4 to 8.
- the compounds of the invention are generally contained in these formulations in an amount of at least 0.001% by weight, for example, 0.001% to 5% by weight, preferably about 0.003% to about 2% by weight, with an amount of about 0.02% to about 1% by weight being most preferred.
- topical administration one to two drops of these formulations are delivered to the surface of the eye one to four times per day according to the routine discretion of a skilled clinician.
- compositions are formulated as aqueous pharmaceutical formulations comprising at least one compound of Formula I and/or Formula II in an amount of 0.001-2% w/v, and a tonicity agent to maintain a tonicity between 200-400 mOsm/kG, wherein the pH of the formulation is 3-9.
- the aqueous pharmaceutical formulation comprises at least one compound of Formula I and/or Formula II in an amount of 0.001-2% w/v, one or more complexing and/or solubilizing agents, 0.01-0.5% preservative, 0.01-1% chelating agent, and a tonicity agent to maintain a tonicity between 200-400 mOsm/kG, wherein the pH of the formulation is 4-8.
- the preferred amount of the compound is 0.01-1% w/v.
- the delivery of such ophthalmic preparations may be done using a single unit dose vial wherein the inclusion of a preservative may be precluded.
- the ophthalmic preparation may be contained in an ophthalmic dropper container intended for multi-use.
- the multi-use product container may or may not contain a preservative, especially in the event the formulation is self-preserving.
- the dropper container is designed to deliver a certain fixed volume of product preparation in each drop.
- the typical drop volume of such an ophthalmic preparation will range from 20-60 microliters, preferably 25-55 microliters, more preferably 30-50 microliters, with 35-50 microliters being most preferred.
- Glaucoma is an ophthalmic disease that leads to irreversible visual impairment.
- Primary open-angle glaucoma is characterized by abnormally high resistance to fluid (aqueous humor) drainage from the eye.
- Cellular contractility and changes in cell-cell and cell-trabeculae adhesion in the trabecular meshwork are major determinants of the resistance to flow.
- the compounds of the present invention cause a transient, pharmacological perturbation of both cell contractility and cell adhesions, mainly via disruption of the actomyosin-associated cytoskeletal structures and/or the modulation of their interactions with the membrane. Altering the contractility of trabecular meshwork cells leads to drainage-surface expansion.
- Loss of cell-cell, cell-trabeculae adhesion may influence paracellular fluid flow across Schlemm's canal or alter the fluid flow pathway through the juxtacanalicular tissue of the trabecular meshwork. Both mechanisms likely reduce the resistance of the trabecular meshwork to fluid flow and thereby reduce intraocular pressure in a therapeutically useful manner.
- the compounds of the present invention are useful for modulation of wound healing after trabeculectomy.
- the compounds in general are less toxic to both corneal epithelial and endothelial cells than the antimetabolites such as 5-fluorouracil or mitomycin C.
- the compounds inhibit actomyosin-driven contractility, leading to deterioration of the actin microfilament system and perturbation of its membrane anchorage, which weakens the cell-extracellular matrix adhesions. These properties inhibit wound healing and thereby reduce bleb failure following the surgery.
- PCO posterior capsule opacification
- Perturbation of the actin cytoskeleton and focal adhesions through rho kinase inhibition may facilitate surgical removal of all cells from the capsular bag and thereby reduce PCO.
- Angiogenesis is characterized by the development of new vasculature from pre-existing vessels and plays a central role in physiological processes such as embryogenesis, wound healing and female reproductive function, as well as pathophysiologic events including cancer, rheumatoid arthritis and diabetic retinopathy. The growth and metastasis of tumors is critically dependent upon angiogenesis.
- Angiogenesis is a multistep process involving the endothelial cell (EC) cytoskeleton in migration, proliferation, and barrier stabilization.
- EC endothelial cell
- Angiogenesis is also involved in several ocular diseases such as age-related macular degeneration, diabetic retinopathy, retinopathy of prematurity, corneal angiogenesis, choroidial neovascularization, neovascular, glaucoma, ocular tumorigenesis.
- Applicants believe that interactions between the cytoskeleton and apoptosis are involved in the intracellular pathways by which angiogenic tube formation occurs.
- the compounds of the present invention are useful in inhibiting angiogenesis and treating tumors and angiogenesis-associated ophthalmic diseases.
- the actin cytoskeleton is important in the modulation of fluid transport. Antimitotic drugs markedly interfere with antidiuretic response, strongly implying that cytoskeleton integrity is essential to this function. This role of the cytoskeleton in controlling the epithelial transport is a necessary step in the translocation of the water channel containing particle aggregates and in their delivery to the apical membrane. Osmolality-dependent reorganization of the cytoskeleton and expression of specific stress proteins are important components of the regulatory systems involved in the adaptation of medullary cells to osmotic stress.
- the compounds of the present invention are useful in directing epithelial function and modulating fluid transport, particularly modulating fluid transport on the ocular surface.
- Rho-associated protein kinase inhibitors due to their regulation of smooth muscle contractility, are useful in the treatment of vasospasm, specifically retinal vasospasm. Relaxation of retinal vasculature increases perfusion rates thereby providing a neuroprotective mechanism (decreased apoptosis and necrosis) in retinal diseases and retinopathies such as glaucoma, ocular hypertension, age-related macular degeneration or retinitis pigmentosa. Additionally, these kinase inhibitors regulate vascular endothelial permeability and as such can play a vasoprotective role to various atherogenic agents.
- the present invention provides a method of reducing intraocular pressure, including treating glaucoma such as primary open-angle glaucoma; a method of treating constriction of the visual field; a method of inhibiting wound healing after trabeculectomy; a method of treating posterior capsule opacification following extracapsular cataract extraction and intraocular lens implantation; a method of inhibiting angiogenesis; a method of modulating fluid transport on the ocular surface; a method of controlling vasospasm; a method of increasing tissue perfusion; a method of neuroprotection; and a method of vasoprotection to atherogenic agents.
- the method comprises the steps of identifying a subject in need of treatment, and administering to the subject a compound of Formula I or Formula II, in an amount effective to alter the actin cytoskeleton, such as by inhibiting actomyosin interactions.
- the pharmaceutical composition of the present invention is administered locally to the eye (e.g., topical, intracameral, intravitreal, subretinal, subconjunctival, retrobulbar or via an implant) in the form of ophthalmic formulations.
- the compounds of the invention can be combined with ophthalmologically acceptable preservatives, surfactants, viscosity enhancers, penetration enhancers, bioadhesives, antioxidants, buffers, sodium chloride, and water to form an aqueous or non-aqueous, sterile ophthalmic suspension, emulsion, microemulsion, gel, or solution to form the compositions of the invention.
- the active compounds disclosed herein can be administered to the eyes of a patient by any suitable means, but are preferably administered by administering a liquid or gel suspension of the active compound in the form of drops, spray or gel.
- the active compounds can be applied to the eye via liposomes.
- the active compounds can be infused into the tear film via a pump-catheter system.
- Another embodiment of the present invention involves the active compound contained within a continuous or selective-release device, for example, membranes such as, but not limited to, those employed in the OcusertTM System (Alza Corp., Palo Alto, Calif.).
- the active compounds can be contained within, carried by, or attached to contact lenses that are placed on the eye.
- Another embodiment of the present invention involves the active compound contained within a swab or sponge that can be applied to the ocular surface. Another embodiment of the present invention involves the active compound contained within a liquid spray that can be applied to the ocular surface. Another embodiment of the present invention involves an injection of the active compound directly into the lacrimal tissues or onto the eye surface.
- the compounds of the invention can be administered systematically by any methods known to a skilled person when used for the purposes described above.
- tert-butyl 3-oxopiperidine-1-carboxylate (14.2 g, 0.07 mol), 1,2-dichloroethane (300 mL), and 1-(5-amino-1H-indazol-1-yl)-2,2-dimethylpropan-1-one maleate salt (23.0 g, 0.07 mol).
- the vessel was purged with nitrogen and stirred at 20° C. for one hour.
- Sodium triacetoxyborohydride (19.0 g, 0.09 mol) was added, and the reaction was monitored by analytical TLC to completion.
- the reaction was diluted with 100 mL of saturated sodium bicarbonate.
- the organic phase was isolated, dried over MgSO 4 , filtered and evaporated to dryness to afford the title compound as a yellow solid (25.0 g, 91%).
- the salt was converted to its free base by partitioning between 75 mL of saturated sodium bicarbonate and ethyl acetate (300 mL). The organic phase was separated, dried over MgSO 4 , filtered and concentrated to give the title compound as an amorphous solid (15.5 g, 83%).
- the solution was cooled to room temperature and then filtered through a pad of celite, washing with additional toluene.
- the toluene solution was then loaded directly onto a silica gel column that had been packed with heptane.
- the column was flushed with 2 column volumes of heptane, and then eluted with 40/60-EtOAc/heptane to afford the title compound (1.00 g 82%).
- the title compound was prepared by reaction of 2,2-dimethyl-1-(5-(piperidin-3-ylamino)-1H-indazol-1-yl)propan-1-one with tert-butyl 4-oxoazepane-1-carboxylate using the method of Example 3, deprotection using the method of Example 4, reaction with benzaldehyde following the method of Example 8, and final deprotection using the method of Example 9.
- the title compound was prepared by reaction of 1-(5-amino-3-methyl-1H-indazol-1-yl)-2,2-dimethylpropan-1-one with tert-butyl 3-oxopiperidine-1-carboxylate using the method of Example 3, deprotection using the method of Example 4, reaction with benzaldehyde following the method of Example 8, and final deprotection using the method of Example 9.
- the title compound was prepared by reaction of isoquinolin-5-amine with tert-butyl 4-oxoazepane-1-carboxylate using the method of Example 3, followed by deprotection using the method of Example 4 and reaction with benzaldehyde following the method of Example 8.
- the flask containing the solids was then evacuated and back-filled with nitrogen three times to degas. Pyridine (200 mL) was then added, and the flask was again purged three times with vacuum and nitrogen. The dark greenish mixture was then stirred at 55° C. overnight under a nitrogen atmosphere. In the morning, the reaction was cooled to room temperature, diluted with 250 mL EtOAc, washed three times with 250 mL portions of 10% NaHSO 4 , and the organic phase was evaporated. The residue was dissolved in toluene and loaded onto a silica column that had been packed with heptane. The column was washed with 5 column volumes of heptane, after which the desired product was eluted with EtOAc/heptane, to provide the title compound as a pale yellow foam (2.4 g, 81%).
- ROCK2 kinase UpstateChemicon #14-451
- Flourescein tagged substrate peptide Fl-AKRRRLSSLRA (Molecular Devices product number R7184) was preincubated with test compound for 5 minutes in buffer containing 10 mM Tris-HCl pH 7.2, 10 mM MgCl 2 , and 0.1% BSA. Following the preincubation, 10 ⁇ M ATP was added to initiate the reaction. After 60 minutes at room temperature, Molecular Devices IMAPTM binding solution was added to bind phosphorylated substrate. After 30 minutes of incubation in the presence of the IMAPTM beads the fluorescence polarization was read and the ratio was reported as mP. IC 50 results were calculated using the Prism software from Graphpad.
- This assay demonstrates a compound's ability to inhibit ROCK2 in an in vitro setting using the isolated enzyme. Most of the compounds studied inhibited ROCK2 with an IC 50 below 10 ⁇ M, many of these inhibiting below 1 ⁇ M. The most potent compounds in this assay showed IC 50 values below 250 nM. Compounds having ROCK2 IC 50 values on the order of 2 ⁇ M or below have been shown to possess efficacy in numerous studies using in vivo models of the disease processes described in this application, specifically in models of elevated IOP and glaucoma. See Tian et al., Arch. Ophthalmol. 116: 633-643, 1998; Tian et al., Invest. Ophthalmol. Vis. Sci.
- NIH/3T3 cells were grown in DMEM-H containing glutamine and 10% Colorado Calf Serum. Cells were passaged regularly prior to reaching confluence. Eighteen to 24 hours prior to experimentation, the cells were plated onto Poly-L-Lysine-coated glass bottom 24-well plates. On the day of experimentation, the cell culture medium was removed and was replaced with the same medium containing from 10 nM to 25 ⁇ M of the test compound, and the cells were incubated for 60 minutes at 37° C. The culture medium was then removed and the cells were washed with warmed PBS and fixed for 10 minutes with warmed 4% paraformaldehyde.
- the cells were permeabilized with 0.5% Triton-X, stained with TRITC-conjugated phalloidin and imaged using a Nikon Eclipse E600 epifluorescent microscope to determine the degree of actin disruption. Results were expressed as a numerical score indicating the observed degree of disruption of the actin cytoskeleton at the test concentration, ranging from 0 (no effect) to 4 (complete disruption), and were the average of at least 2 determinations.
- Intraocular fluid aqueous humor
- aqueous humor Intraocular fluid (aqueous humor) was collected from New Zealand White rabbits to determine corneal and anterior chamber pharmacokinetics of formulations containing Compound 1.008, 1.039, and 1.051.
- Each animal was dose bilaterally with 2 ⁇ 10 ⁇ l of 25 mM of each test compound (in 10 mM acetate buffered saline, 0.01% benzalkonium chloride, 0.05% EDTA, pH 4.5) or with vehicle.
- the upper and lower eyelids were immobilized and the compound was administered to the superior aspect of the globe allowing it to flow across the ocular surface. Following instillation, blinking was prevented for 30 seconds.
- Aqueous humor was collected from 30 minutes to 8 hours following topical instillation using a 30-gauge needle inserted proximal to the corneal scleral limbus. Subsequently 30 ⁇ l of aqueous humor was aspirated using a 300 ⁇ l syringe. Aqueous humor samples were assayed for the concentration of the test compound using an LC/MS/MS assay system. All experiments were conducted in accordance with the ARVO Statement for the Use of Animals in Ophthalmic and Vision Research and in compliance with National Institutes of Health. The results of observed aqueous humor concentrations of the test compounds at 0.5, 2, and 4 hours after instillation in the animal eyes are shown in FIG. 1 .
- This pharmacokinetic assay shows that the compounds of the invention when dosed topically are able to penetrate the eye and achieve concentrations in the aqueous humor adequate to provide substantial ROCK inhibition at the sight of action, that is, concentrations at or above the ROCK IC 50 of the compound in question. Further, it shows that these compounds can show different pharmacokinetic profiles on topical ocular dosing, with some compounds showing a more prolonged presence, while others penetrate rapidly into the eye and are quickly cleared from the aqueous humor.
- a trained ophthalmologist performed a slit lamp examination to determine the integrity of the corneal epithelium and endothelium, presence of flare or cells in the AC, and clarity of the lens. All animals were free of ocular abnormalities when studied.
- IOP was measured every hour for 6 hours using a minified Goldmann applanation tonometer. Slit lamp exams were conducted at 3 and 6 hours. The intraocular pressure of animals after treatment with the test compounds or vehicle at day 1 and day 4 from hour 0 to hour 6 is shown in FIGS. 2-1 to 2 - 3 .
- This pharmacodynamic assay shows that the compounds of the invention are able to achieve meaningful reductions in intraocular pressure when dosed topically in normotensive primates. It further shows that these compounds are well-tolerated on the ocular surface when dosed in a therapeutically relevant fashion. Reduction in intraocular pressure in this way is the primary objective of current glaucoma therapy.
- the assay described here is the most widely accepted method for the preclinical evaluation of intraocular pressure lowering agents.
Abstract
Description
wherein:
R1 is aryl or heteroaryl, optionally substituted;
Q is C═O, SO2, or (CR4R5)n3;
n1 is 1, 2, or 3;
n2 is 1 or 2;
n3 is 0, 1, 2, or 3;
wherein the ring represented by
is optionally substituted by alkyl, halo, oxo, OR6, NR6R7, or SR6;
R2 is selected from the following heteroaryl systems, optionally substituted:
R3-R7 are independently H, alkyl, alkenyl, alkynyl, cycloalkyl, cycloalkenyl, cycloalkylalkyl, cycloalkylalkenyl, or cycloalkylalkynyl optionally substituted.
wherein:
Ar is a monocyclic or bicyclic aryl or heteroaryl ring, such as phenyl;
X is from 1 to 3 substituents on Ar, each independently in the form Y—Z, in which Z is attached to Ar;
Y is one or more substituents on Z, and each is chosen independently from H, halogen, or the heteroatom-containing substituents, including but not limited to OR8, NR8R9, NO2, SR8, SOR8, SO2R8, SO2NR8R9, NR8SO2R9, OCF3, CONR8R9, NR8C(═O)R9, NR8C(═O)OR9, OC(═O)NR8R9, or NR8C(═O)NR9R10;
Each instance of Z is chosen independently from alkyl, alkenyl, alkynyl, aryl, arylalkyl, arylalkenyl, arylalkynyl, cycloalkyl, cycloalkenyl, cycloalkylalkyl, cycloalkylalkenyl, cycloalkylalkynyl, heteroaryl, heteroarylalkyl, heteroarylalkenyl, heteroarylalkynyl, heterocycle, (heterocycle)alkyl, (heterocycle)alkenyl, (heterocycle)alkynyl, or is absent;
R8 is H, alkyl, alkenyl, alkynyl, aryl, arylalkyl, arylalkenyl, arylalkynyl, cycloalkyl, cycloalkenyl, cycloalkylalkyl, cycloalkylalkenyl, cycloalkylalkynyl, heteroaryl, heteroarylalkyl, heteroarylalkenyl, heteroarylalkynyl, (heterocycle)alkyl, (heterocycle)alkenyl, (heterocycle)alkynyl, or heterocycle; optionally substituted by one or more halogen or heteroatom-containing substituents, including but not limited to OR11, NR11R12, NO2, SR11, SOR11, SO2R11, SO2NR11R12, NR11SO2R12, OCF3, CONR11R12, NR11C(═O)R12, NR11C(═O)OR12, OC(═O)NR11R12, or NR11C(═O)NR12R13;
R9 and R10 are independently H, alkyl, alkenyl, alkynyl, aryl, arylalkyl, arylalkenyl, arylalkynyl, cycloalkyl, cycloalkenyl, cycloalkylalkyl, cycloalkylalkenyl, cycloalkylalkynyl, heteroaryl, heteroarylalkyl, heteroarylalkenyl, heteroarylalkynyl, (heterocycle)alkyl, (heterocycle)alkenyl, (heterocycle)alkynyl, or heterocycle; optionally substituted by one or more halogen or heteroatom-containing substituents, including but not limited to OR14, NR14R15, NO2, SR14, SOR14, SO2R14, SO2NR14R15, NR14SO2R15, OCF3, CONR14R15, NR14C(═O)R15, NR14C(═O)OR15, OC(═O)NR14R15, or NR14C(═O)NR15R16;
any two of the groups R8, R9 and R10 are optionally joined with a link selected from the group consisting of bond, —O—, —S—, —SO—, —SO2—, and —NR17— to form a ring;
R11-R17 are independently H, alkyl, alkenyl, alkynyl, aryl, arylalkyl, arylalkenyl, arylalkynyl, cycloalkyl, cycloalkenyl, cycloalkylalkyl, cycloalkylalkenyl, cycloalkylalkynyl, heteroaryl, heteroarylalkyl, heteroarylalkenyl, heteroarylalkynyl, (heterocycle)alkyl, (heterocycle)alkenyl, (heterocycle)alkynyl, or heterocycle.
| TABLE I |
| Example Compounds. |
| Compound | Structure | Embodiments |
| 1.001 | | 1c, 7, 8, 9, 10, 12, 15c |
| 1.002 | | 1c, 7, 8, 9, 10, 12, 15c |
| 1.003 | | 1c, 7, 8, 9, 10, 12a, 15c, 15d |
| 1.004 | | 1c, 7, 8, 9, 10, 12, 15c |
| 1.005 | | 1c, 7, 8, 9, 10, 12, 15c |
| 1.006 | | 1c, 7, 8, 9, 10, 12a, 15c, 15d |
| 1.007 | | 1c, 7, 8, 9, 10, 12a, 15c, 15d |
| 1.008 | | 1c, 7, 8, 9, 10, 12b, 15c, 15e |
| 1.009 | | 1c, 7, 8, 9, 10, 11, 14c |
| 1.010 | | 1c, 7, 8, 9, 10, 11, 14c |
| 1.011 | | 1c, 7, 8, 9, 10, 11, 14c |
| 1.012 | | 1c, 7, 8, 9, 10, 11, 14c |
| 1.013 | | 1c, 7, 8, 9, 10 |
| 1.014 | | 1c, 7, 8, 9, 10 |
| 1.015 | | 1c, 7, 8, 9, 10 |
| 1.016 | | 1c, 7, 8, 9, 10 |
| 1.017 | | 1c, 7, 8, 9, 10, 12b, 15c, 15e |
| 1.018 | | 1c, 7, 8, 9, 10, 12a, 15c, 15d |
| 1.019 | | 1c, 7, 8, 9, 10, 13, 16c |
| 1.020 | | 1c, 7, 8, 9, 10, 11, 14c |
| 1.021 | | 1c, 7, 8, 9, 10, 11, 14c |
| 1.022 | | 1c, 7, 8, 9, 10 |
| 1.023 | | 1c, 7, 8, 9, 10 |
| 1.024 | | 1c, 7, 8, 9, 10 |
| 1.025 | | 1c, 7, 8, 9, 10 |
| 1.026 | | 1c, 7, 8, 9, 10, 12b, 15c, 15e |
| 1.027 | | 1c, 7, 8, 9, 10, 13, 16c |
| 1.028 | | 1c, 7, 8, 9, 10, 13, 16c |
| 1.029 | | 1c, 7, 8, 9, 10, 13, 16c |
| 1.030 | | 1c, 7, 8, 9, 10, 11, 14c |
| 1.031 | | 1c, 7, 8, 9, 10 |
| 1.032 | | 1c, 7, 8, 9, 10 |
| 1.033 | | 1c, 7, 8, 9, 10 |
| 1.034 | | 1c, 7, 8, 9, 10, 11, 14c |
| 1.035 | | 1c, 7, 8, 9, 10, 13, 16c |
| 1.036 | | 1c, 7, 8, 9, 10 |
| 1.037 | | 1c, 7, 8, 9, 10, 11, 14c |
| 1.038 | | 1c, 7, 8, 9, 10, 12, 15c |
| 1.039 | | 1c, 7, 8, 9, 10, 12a, 15c, 15d |
| 1.040 | | 1c, 7, 8, 9, 10, 12b, 15c, 15e |
| 1.041 | | 1c, 7, 8, 9, 10, 13, 16c |
| 1.042 | | 1c, 7, 8, 9, 10, 13, 16c |
| 1.043 | | 1c, 7, 8, 9, 10, 13, 16c |
| 1.044 | | 1c, 7, 8, 9, 10, 11, 14c |
| 1.045 | | 1c, 7, 8, 9, 10 |
| 1.046 | | 1c, 7, 8, 9, 10 |
| 1.047 | | 1c, 7, 8, 9, 10, 11, 14c |
| 1.048 | | 1c, 7, 8, 9, 10, 12, 15c |
| 1.049 | | 1a, 6a, 8, 9, 10 |
| 1.050 | | 1b, 6b, 8, 9, 10 |
| 1.051 | | 1c, 7, 8, 9, 10, 12a, 15c, 15d |
| 1.052 | | 1c, 7, 8, 9, 10 |
| 1.053 | | 1c, 7, 8, 9, 10 |
| 1.054 | | 1 c, 7, 8, 9, 10 |
| 1.055 | | 1c, 7, 8, 9, 10, 12, 15c |
| 1.056 | | 1c, 7, 8, 9, 10, 12, 15c |
| 1.057 | | 1c, 7, 8, 9, 10, 13, 16c |
| 1.058 | | 1c, 7, 8, 9, 10, 12a, 15c, 15d |
| 1.059 | | 1c, 7, 8, 9, 10 |
| 1.060 | | 1c, 7, 8, 9, 10, 12a, 15c, 15d |
| 1.061 | | 1c, 7, 8, 9, 10, 13, 16c |
| 1.062 | | 1c, 7, 8, 9, 10 |
| 1.063 | | 1c, 7, 8, 9, 10 |
| 1.064 | | 1c, 7, 8, 9, 10 |
| 1.065 | | 1c, 7, 8, 9, 10 |
| 1.066 | | 1c, 7, 8, 9, 10 |
| 1.067 | | 1c, 7, 8, 9, 10 |
| 1.068 | | 1c, 7, 8, 9, 10 |
| 1.069 | | 1c, 7, 8, 9, 10 |
| 1.070 | | 1c, 7, 8, 9, 10 |
| 1.071 | | 1c, 7, 8, 9, 10 |
| 1.072 | | 1c, 7, 8, 9, 10 |
| 1.073 | | 1c, 7, 8, 9, 10 |
| 1.074 | | 1c, 7, 8, 9, 10, 12b, 15c, 15e |
| 1.075 | | 1c, 7, 8, 9, 10, 12b, 15c, 15e |
| 1.076 | | 1c, 7, 8, 9, 10, 11, 14c |
| 1.077 | | 1, 7, 8, 9, 10, 11, 14c |
| 1.078 | | 1c, 7, 8, 9, 10 |
| 1.079 | | 1c, 7, 8, 9, 10 |
| 1.080 | | 1c, 7, 8, 9, 10 |
| 1.081 | | 1c, 7, 8, 9, 10 |
| 1.082 | | 1c, 7, 8, 9, 10 |
| 1.083 | | 1c, 7, 8, 9, 10, 11, 14c |
| 1.084 | | 1c, 7, 8, 9, 10, 12a, 15c, 15d |
| 1.085 | | 1c, 7, 8, 9, 10, 12a, 15c, 15d |
| 1.086 | | 1c, 7, 8, 9, 10, 12a, 15c, 15d |
| 1.087 | | 1c, 7, 8, 9, 10, 12a, 15c, 15d |
| 1.088 | | 1c, 7, 8, 9, 10, 12a, 15c, 15d |
| 1.089 | | 1c, 7, 8, 9, 10 |
| 1.090 | | 1c, 7, 8, 9, 10, 12a, 15c, 15d |
| 1.091 | | 1c, 7, 8, 9, 10, 12a, 15c, 15d |
| 1.092 | | 1c, 7, 8, 9, 10, 12a, 15c, 15d |
| 1.093 | | 1c, 7, 8, 9, 10, 12a, 15c, 15d |
| 1.094 | | 1c, 7, 8, 9, 10, 12a, 15c, 15d |
| 1.095 | | 1c, 7, 8, 9, 10, 12a, 15c, 15d |
| 1.096 | | 1c, 7, 8, 9, 10, 12a, 15c, 15d |
| 1.097 | | 1c, 7, 8, 9, 10, 12a, 15c, 15d |
| 1.098 | | 1c, 7, 8, 9, 10, 12a, 15c, 15d |
| 1.099 | | 1c, 7, 8, 9, 10, 13, 16c |
| 1.100 | | 1c, 7, 8, 9, 10 |
| 1.101 | | 1c, 7, 8, 9, 10, 13, 16c |
| 1.102 | | 1c, 7, 8, 9, 10, 12a, 15c, 15d |
| 1.103 | | 1c, 7, 8, 9, 10, 13, 16c |
| 1.104 | | 1c, 7, 8, 9, 10, 13, 16c |
| 1.105 | | 1c, 7, 8, 9, 10, 13, 16c |
| 1.106 | | 1c, 7, 8, 9, 10, 13, 16c |
| 1.107 | | 1c, 7, 8, 9, 10, 13, 16c |
| 1.108 | | 1c, 7, 8, 9, 10, 13, 16c |
| 1.109 | | 1c, 7, 8, 9, 10, 13, 16c |
| 1.110 | | 1c, 7, 8, 9, 10 |
| 1.111 | | 1c, 7, 8, 9, 10, 12a, 15c, 15d |
| 1.112 | | 1c, 7, 8, 9, 10, 13, 16c |
| 1.113 | | 1c, 7, 8, 9, 10, 12a, 15c, 15d |
| 1.114 | | 1c, 7, 8, 9, 10, 13, 16c |
| 1.115 | | 1c, 7, 8, 9, 10, 12a, 15c, 15d |
| 1.116 | | 1c, 7, 8, 9, 10, 12a, 15c, 15d |
| 1.117 | | 1c, 7, 8, 9, 10, 12a, 15c, 15d |
| 1.118 | | 1c, 7, 8, 9, 10, 12a, 15c, 15d |
| 1.119 | | 1c, 7, 8, 9, 10, 13, 16c |
| 1.120 | | 1c, 7, 8, 9, 10, 12a, 15c, 15d |
| 1.121 | | 1c, 7, 8, 9, 10, 12a, 15c, 15d |
| 1.122 | | 1c, 7, 8, 9, 10, 13, 16c |
| 1.123 | | 1c, 7, 8, 9, 10, 12a, 15c, 15d |
| 1.124 | | 1c, 7, 8, 9, 10, 12a, 15c, 15d |
| 1.125 | | 1c, 7, 8, 9, 10, 12a, 15c, 15d |
| 1.126 | | 1c, 7, 8, 9, 10, 12a, 15c, 15d |
| 1.127 | | 1c, 7, 8, 9, 10, 12a, 15c, 15d |
| 1.128 | | 1c, 7, 8, 9, 10, 12a, 15c, 15d |
| 1.129 | | 1c, 7, 8, 9, 10, 12a, 15c, 15d |
| 1.130 | | 1c, 7, 8, 9, 10, 12a, 15c, 15d |
| 1.131 | | 1c, 7, 8, 9, 10 |
| 1.132 | | 1c, 7, 8, 9, 10 |
| 1.133 | | 1c, 7, 8, 9, 10 |
| 1.134 | | 1c, 7, 8, 9, 10 |
| 1.136 | | 1c, 7, 8, 9, 10 |
| 1.137 | | 1c, 7, 8, 9, 10 |
| 1.138 | | 1c, 7, 8, 9, 10 |
| 2.001 | | 2c, 7, 8, 9, 10 |
| 2.002 | | 2c, 7, 8, 9, 10, 12, 15c |
| 2.003 | | 2c, 7, 8, 9, 10, 12, 15c |
| 2.004 | | 2c, 7, 8, 9, 10, 12a, 15c, 15d |
| 2.005 | | 2c, 7, 8, 9, 10, 12, 15c |
| 2.006 | | 2c, 7, 8, 9, 10 |
| 2.007 | | 2c, 7, 8, 9, 10, 12, 15c |
| 2.008 | | 2c, 7, 8, 9, 10, 12a, 15c, 15d |
| 2.009 | | 2c, 7, 8, 9, 10, 12b, 15c, 15e |
| 2.010 | | 2c, 7, 8, 9, 10, 11, 14c |
| 2.011 | | 2c, 7, 8, 9, 10, 11, 14c |
| 2.012 | | 2c, 7, 8, 9, 10, 12b, 15c, 15e |
| 2.013 | | 2c, 7, 8, 9, 10 |
| 2.014 | | 2c, 7, 8, 9, 10 |
| 2.015 | | 2c, 7, 8, 9, 10 |
| 2.016 | | 2c, 7, 8, 9, 10 |
| 2.017 | | 2c, 7, 8, 9, 10 |
| 2.018 | | 2c, 7, 8, 9, 10 |
| 2.019 | | 2c, 7, 8, 9, 10, 11, 14c |
| 2.020 | | 2c, 7, 8, 9, 10, 11, 14c |
| 2.021 | | 2c, 7, 8, 9, 10, 12b, 15c, 15e |
| 2.022 | | 2c, 7, 8, 9, 10, 11, 14c |
| 2.023 | | 2c, 7, 8, 9, 10, 11, 14c |
| 2.024 | | 2c, 7, 8, 9, 10, 12b, 15c, 15e |
| 2.025 | | 2c, 7, 8, 9, 10 |
| 2.026 | | 2c, 7, 8, 9, 10, 12b, 15c, 15e |
| 2.027 | | 2c, 7, 8, 9, 10 |
| 2.028 | | 2c, 7, 8, 9, 10 |
| 2.029 | | 2c, 7, 8, 9, 10, 12b, 15c, 15e |
| 2.030 | | 2c, 7, 8, 9, 10 |
| 2.031 | | 2c, 7, 8, 9, 10, 11, 14c |
| 2.032 | | 2c, 7, 8, 9, 10, 12a, 15c, 15d |
| 2.033 | | 2c, 7, 8, 9, 10, 12a, 15c, 15d |
| 2.034 | | 2c, 7, 8, 9, 10, 12a, 15c, 15d |
| 2.035 | | 2c, 7, 8, 9, 10, 12a, 15c, 15d |
| 2.036 | | 2c, 7, 8, 9, 10, 12a, 15c, 15d |
| 2.037 | | 2c, 7, 8, 9, 10, 12a, 15c, 15d |
| 2.038 | | 2c, 7, 8, 9, 10, 12a, 15c, 15d |
| 2.039 | | 2c, 7, 8, 9, 10, 12a, 15c, 15d |
| 2.040 | | 2c, 7, 8, 9, 10, 12a, 15c, 15d |
| 2.041 | | 2c, 7, 8, 9, 10, 12a, 15c, 15d |
| 2.042 | | 2c, 7, 8, 9, 10, 12a, 15c, 15d |
| 2.043 | | 2c, 7, 8, 9, 10, 12a, 15c, 15d |
| 2.044 | | 2c, 7, 8, 9, 10, 12a, 15c, 15d |
| 3.001 | | 3c, 7, 8, 9, 10 |
| 3.002 | | 3c, 7, 8, 9, 10 |
| 4.001 | | 4c, 7, 8, 9, 10 |
| 4.002 | | 4c, 7, 8, 9, 10 |
| 5.001 | | 5a, 7, 8, 9, 10 |
| 5.002 | | 5a, 7, 8, 9, 10 |
Preparation of Compounds of Formula I and Formula II
| TABLE II |
| Rho Kinase Assay Data. |
| Compound | ROCK2 IC50, nM | ||
| 1.001 | 358 | ||
| 1.002 | 1,230 | ||
| 1.003 | 6,190 | ||
| 1.004 | 254 | ||
| 1.005 | 2,290 | ||
| 1.006 | 2,750 | ||
| 1.007 | 3,440 | ||
| 1.008 | 65.8 | ||
| 1.009 | 1,610 | ||
| 1.010 | 2,800 | ||
| 1.011 | 12,200 | ||
| 1.012 | 12,200 | ||
| 1.013 | 3,660 | ||
| 1.014 | 4,690 | ||
| 1.015 | 9,980 | ||
| 1.016 | 135 | ||
| 1.017 | 215 | ||
| 1.018 | 2,410 | ||
| 1.019 | 13,400 | ||
| 1.020 | 251 | ||
| 1.021 | 553 | ||
| 1.022 | 1,610 | ||
| 1.023 | 334 | ||
| 1.024 | 107 | ||
| 1.025 | 588 | ||
| 1.026 | 2,130 | ||
| 1.027 | 6,600 | ||
| 1.028 | 1,310 | ||
| 1.029 | 11,700 | ||
| 1.031 | 1,750 | ||
| 1.032 | 1,940 | ||
| 1.033 | 86.9 | ||
| 1.034 | 69.1 | ||
| 1.035 | 208 | ||
| 1.036 | 1,020 | ||
| 1.037 | 69.5 | ||
| 1.038 | 2,760 | ||
| 1.039 | 328 | ||
| 1.040 | 144 | ||
| 1.041 | 137 | ||
| 1.042 | 4,400 | ||
| 1.043 | 11,700 | ||
| 1.044 | 6,510 | ||
| 1.045 | 586 | ||
| 1.046 | 156 | ||
| 1.047 | 57.0 | ||
| 1.048 | 721 | ||
| 1.049 | 604 | ||
| 1.050 | 1,040 | ||
| 1.051 | 62.6 | ||
| 1.052 | 15.3 | ||
| 1.053 | 189 | ||
| 1.054 | 132 | ||
| 1.055 | 472 | ||
| 1.056 | 435 | ||
| 1.057 | 194 | ||
| 1.058 | 47.5 | ||
| 1.059 | 32.0 | ||
| 1.060 | 87.2 | ||
| 1.061 | 282 | ||
| 1.062 | 155 | ||
| 1.063 | 223 | ||
| 1.064 | 165 | ||
| 1.065 | 326 | ||
| 1.066 | 232 | ||
| 1.067 | 1,850 | ||
| 1.068 | 660 | ||
| 1.069 | 6,050 | ||
| 1.070 | 6,680 | ||
| 1.071 | 5,590 | ||
| 1.072 | 58.5 | ||
| 1.073 | 111 | ||
| 1.074 | 71.6 | ||
| 1.075 | 74.5 | ||
| 1.076 | 111 | ||
| 1.077 | 53.1 | ||
| 1.078 | 63.1 | ||
| 1.079 | 102 | ||
| 1.080 | 162 | ||
| 1.081 | 30.1 | ||
| 1.082 | 30.4 | ||
| 1.083 | 7,310 | ||
| 1.084 | 274 | ||
| 1.085 | 1,680 | ||
| 1.086 | 310 | ||
| 1.087 | 280 | ||
| 1.088 | 651 | ||
| 1.089 | 29.4 | ||
| 1.090 | 354 | ||
| 1.091 | 49.4 | ||
| 1.092 | 406 | ||
| 1.093 | 101 | ||
| 1.094 | 466 | ||
| 1.095 | 647 | ||
| 1.096 | 1,300 | ||
| 1.097 | 276 | ||
| 1.098 | 250 | ||
| 1.099 | 25.8 | ||
| 1.100 | 42.9 | ||
| 1.101 | 68.5 | ||
| 1.102 | 34.3 | ||
| 1.104 | 107 | ||
| 1.105 | 172 | ||
| 1.106 | 69.2 | ||
| 1.107 | 148 | ||
| 1.108 | 367 | ||
| 1.109 | 242 | ||
| 1.111 | 591 | ||
| 1.112 | 2,030 | ||
| 1.113 | 859 | ||
| 1.114 | 1,490 | ||
| 1.115 | 555 | ||
| 1.116 | 862 | ||
| 1.117 | 323 | ||
| 1.118 | 1,400 | ||
| 1.119 | 3,630 | ||
| 1.120 | 109 | ||
| 1.121 | 578 | ||
| 1.122 | 254 | ||
| 1.123 | 135 | ||
| 1.124 | 59.2 | ||
| 1.125 | 879 | ||
| 1.130 | 2,710 | ||
| 2.001 | 98.8 | ||
| 2.002 | 2,580 | ||
| 2.003 | 5,720 | ||
| 2.004 | 3,710 | ||
| 2.005 | 1,780 | ||
| 2.006 | 73.9 | ||
| 2.007 | 3,620 | ||
| 2.008 | 3,650 | ||
| 2.009 | 368 | ||
| 2.010 | 1,240 | ||
| 2.011 | 4,090 | ||
| 2.012 | 14,900 | ||
| 2.013 | 1,490 | ||
| 2.014 | 1,670 | ||
| 2.015 | 4,190 | ||
| 2.016 | 716 | ||
| 2.017 | 322 | ||
| 2.018 | 632 | ||
| 2.019 | 4,080 | ||
| 2.020 | 820 | ||
| 2.021 | 1,900 | ||
| 2.022 | 311 | ||
| 2.023 | 15,700 | ||
| 2.024 | 8,920 | ||
| 2.025 | 29.9 | ||
| 2.026 | 6,330 | ||
| 2.027 | 120 | ||
| 2.028 | 789 | ||
| 2.029 | 3,140 | ||
| 2.030 | 2,460 | ||
| 2.031 | 87.1 | ||
| 2.032 | 5,330 | ||
| 2.033 | 3,060 | ||
| 2.034 | 4,010 | ||
| 2.035 | 3,240 | ||
| 2.036 | 160 | ||
| 2.037 | 3,060 | ||
| 2.038 | 112 | ||
| 2.039 | 101 | ||
| 2.040 | 273 | ||
| 2.041 | 168 | ||
| 2.043 | 383 | ||
| 2.044 | 433 | ||
| TABLE III |
| Cell Morphology Assay Data. |
| Cell score at 1 | |||
| Compound | μM | ||
| 1.002 | 1.4 | ||
| 1.004 | 1.8 | ||
| 1.005 | 1.3 | ||
| 1.006 | 2 | ||
| 1.008 | 2 | ||
| 1.024 | 2.4 | ||
| 1.025 | 2 | ||
| 1.034 | 2 | ||
| 1.039 | 2 | ||
| 1.041 | 2.5 | ||
| 1.046 | 2.5 | ||
| 1.048 | 1.5 | ||
| 1.051 | 2.5 | ||
| 1.052 | 2.8 | ||
| 1.062 | 2.3 | ||
| 1.066 | 2 | ||
| 2.002 | 1.8 | ||
| 2.006 | 2.8 | ||
| 2.008 | 1 | ||
| 2.016 | 1.8 | ||
| 2.017 | 2 | ||
| 2.018 | 1.8 | ||
| 2.026 | 2 | ||
Claims (20)
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US13/273,043 US8604218B2 (en) | 2006-12-18 | 2011-10-13 | Cytoskeletal active rho kinase inhibitor compounds, composition and use |
| US13/614,641 US8604205B2 (en) | 2006-12-18 | 2012-09-13 | Cytoskeletal active rho kinase inhibitor compounds, composition and use |
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US87055506P | 2006-12-18 | 2006-12-18 | |
| US11/958,214 US8071779B2 (en) | 2006-12-18 | 2007-12-17 | Cytoskeletal active rho kinase inhibitor compounds, composition and use |
| US13/273,043 US8604218B2 (en) | 2006-12-18 | 2011-10-13 | Cytoskeletal active rho kinase inhibitor compounds, composition and use |
Related Parent Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US11/958,214 Division US8071779B2 (en) | 2006-12-18 | 2007-12-17 | Cytoskeletal active rho kinase inhibitor compounds, composition and use |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US13/614,641 Division US8604205B2 (en) | 2006-12-18 | 2012-09-13 | Cytoskeletal active rho kinase inhibitor compounds, composition and use |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| US20120046275A1 US20120046275A1 (en) | 2012-02-23 |
| US8604218B2 true US8604218B2 (en) | 2013-12-10 |
Family
ID=39537056
Family Applications (3)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US11/958,214 Active 2030-02-12 US8071779B2 (en) | 2006-12-18 | 2007-12-17 | Cytoskeletal active rho kinase inhibitor compounds, composition and use |
| US13/273,043 Active 2028-08-28 US8604218B2 (en) | 2006-12-18 | 2011-10-13 | Cytoskeletal active rho kinase inhibitor compounds, composition and use |
| US13/614,641 Active US8604205B2 (en) | 2006-12-18 | 2012-09-13 | Cytoskeletal active rho kinase inhibitor compounds, composition and use |
Family Applications Before (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US11/958,214 Active 2030-02-12 US8071779B2 (en) | 2006-12-18 | 2007-12-17 | Cytoskeletal active rho kinase inhibitor compounds, composition and use |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US13/614,641 Active US8604205B2 (en) | 2006-12-18 | 2012-09-13 | Cytoskeletal active rho kinase inhibitor compounds, composition and use |
Country Status (11)
| Country | Link |
|---|---|
| US (3) | US8071779B2 (en) |
| EP (1) | EP2099457A4 (en) |
| JP (1) | JP5511385B2 (en) |
| KR (1) | KR20090091767A (en) |
| CN (2) | CN101583361B (en) |
| AU (1) | AU2007333715B2 (en) |
| BR (1) | BRPI0720457A2 (en) |
| CA (1) | CA2672825A1 (en) |
| MX (1) | MX2009006493A (en) |
| RU (1) | RU2470928C2 (en) |
| WO (1) | WO2008077057A2 (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2016180918A1 (en) | 2015-05-12 | 2016-11-17 | Platod | Combination of pharmacological and microfluidic features for improved platelets production |
Families Citing this family (64)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1910297B1 (en) | 2005-07-11 | 2016-05-25 | Aerie Pharmaceuticals, Inc. | Isoquinoline compounds |
| PL1919290T3 (en) * | 2005-07-12 | 2014-06-30 | Ampio Pharmaceuticals Inc | Methods and products for treatment of diseases |
| WO2008036540A2 (en) | 2006-09-20 | 2008-03-27 | Boehringer Ingelheim International Gmbh | Rho kinase inhibitors |
| US20100022517A1 (en) * | 2006-12-18 | 2010-01-28 | Richards Lori A | Ophthalmic formulation of rho kinase inhibitor compound |
| US8455513B2 (en) | 2007-01-10 | 2013-06-04 | Aerie Pharmaceuticals, Inc. | 6-aminoisoquinoline compounds |
| CA2704199C (en) * | 2007-11-01 | 2016-01-19 | Acucela Inc. | Amine derivative compounds for treating ophthalmic diseases and disorders |
| US8455514B2 (en) | 2008-01-17 | 2013-06-04 | Aerie Pharmaceuticals, Inc. | 6-and 7-amino isoquinoline compounds and methods for making and using the same |
| CA2726611A1 (en) * | 2008-06-18 | 2009-12-23 | Inspire Pharmaceuticals, Inc. | Process for the preparation of rho-kinase inhibitor compounds |
| US8410147B2 (en) * | 2008-06-26 | 2013-04-02 | Inspire Pharmaceuticals, Inc. | Method for treating diseases associated with alterations in cellular integrity using Rho kinase inhibitor compounds |
| US20090325959A1 (en) * | 2008-06-26 | 2009-12-31 | Vittitow Jason L | Method for treating ophthalmic diseases using rho kinase inhibitor compounds |
| US20090325960A1 (en) * | 2008-06-26 | 2009-12-31 | Fulcher Emilee H | Method for treating inflammatory diseases using rho kinase inhibitor compounds |
| WO2009158587A1 (en) * | 2008-06-26 | 2009-12-30 | Inspire Pharmaceuticals, Inc. | Method for treating pulmonary diseases using rho kinase inhibitor compounds |
| US8207195B2 (en) * | 2008-06-26 | 2012-06-26 | Inspire Pharmaceuticals, Inc. | Method for treating neurological and neuropathic diseases using rho kinase inhibitor compounds |
| US20100008968A1 (en) * | 2008-06-26 | 2010-01-14 | Lampe John W | Method for treating cardiovascular diseases using rho kinase inhibitor compounds |
| US8450344B2 (en) | 2008-07-25 | 2013-05-28 | Aerie Pharmaceuticals, Inc. | Beta- and gamma-amino-isoquinoline amide compounds and substituted benzamide compounds |
| US20100204210A1 (en) * | 2008-12-04 | 2010-08-12 | Scott Sorensen | Method for treating pulmonary diseases using rho kinase inhibitor compounds |
| EP3828172A1 (en) | 2009-05-01 | 2021-06-02 | Aerie Pharmaceuticals, Inc. | Dual mechanism inhibitors for the treatment of disease |
| AU2015203100B2 (en) * | 2009-06-22 | 2017-03-16 | Ampio Pharmaceuticals, Inc. | Method for treatment of diseases |
| CA2765883A1 (en) * | 2009-06-22 | 2010-12-29 | Dmi Acquisition Corp. | Methods and products for treatment of diseases |
| DK2554173T3 (en) | 2009-06-22 | 2017-01-16 | Ampio Pharmaceuticals Inc | PROCEDURE FOR DISEASE CARE |
| CA2781390A1 (en) * | 2009-12-14 | 2011-06-23 | Inspire Pharmaceuticals, Inc. | Bridged bicyclic rho kinase inhibitor compounds, composition and use |
| BR112013001125A2 (en) * | 2010-07-19 | 2016-05-17 | Inspire Pharmaceuticals Inc | bifunctional rho kinase inhibiting compounds, composition and use |
| RU2013108641A (en) * | 2010-07-27 | 2014-09-10 | Инспайр Фармасьютикалз, Инк. | METHOD OF TREATMENT OF OPHTHALMIC DISEASES USING COMPOUNDS OF KINASE INHIBITORS IN DRUG FORMS |
| GB201107223D0 (en) * | 2011-04-29 | 2011-06-15 | Amakem Nv | Novel rock inhibitors |
| EP3517604A1 (en) * | 2011-12-06 | 2019-07-31 | Astellas Institute for Regenerative Medicine | Method of directed differentiation producing corneal endothelial cells, compositions thereof, and uses thereof |
| TWI519515B (en) | 2011-12-21 | 2016-02-01 | 諾維拉治療公司 | Hepatitis b antiviral agents |
| US20130168825A1 (en) * | 2011-12-30 | 2013-07-04 | Alliance For Sustainable Energy, Llc | Fabrication of ionic liquid electrodeposited cu-sn-zn-s-se thin films and method of making |
| BR112015004192B1 (en) | 2012-08-28 | 2021-02-09 | Janssen Sciences Ireland Uc | sulfamoyl-arylamides, pharmaceutical composition that comprises them and their use in the treatment of hepatitis b |
| KR102242280B1 (en) | 2012-10-31 | 2021-04-20 | 주식회사 피에이치파마 | Novel ROCK inhibitors |
| WO2014100352A1 (en) | 2012-12-19 | 2014-06-26 | Ampio Pharmaceuticals, Inc. | Method for treatment of diseases |
| DK2961732T3 (en) | 2013-02-28 | 2017-07-10 | Janssen Sciences Ireland Uc | SULFAMOYLARYLAMIDS AND USE THEREOF AS MEDICINES TO TREAT HEPATITIS B |
| EP3811943B1 (en) | 2013-03-15 | 2023-02-22 | Aerie Pharmaceuticals, Inc. | Compound for use in the treatment of ocular disorders |
| BR112015025052A2 (en) | 2013-04-03 | 2021-07-06 | Janssen Sciences Ireland Uc | n-phenyl carboxamide derivatives and their use as medicines for the treatment of hepatitis b |
| ES2695182T3 (en) | 2013-05-17 | 2019-01-02 | Janssen Sciences Ireland Uc | Derivatives of sulfamoyl thiophenamide and its use as medicines to treat hepatitis B |
| JO3603B1 (en) | 2013-05-17 | 2020-07-05 | Janssen Sciences Ireland Uc | Sulphamoylpyrrolamide derivatives and the use thereof as medicaments for the treatment of hepatitis b |
| SG10201805033XA (en) | 2013-07-25 | 2018-07-30 | Janssen Sciences Ireland Uc | Glyoxamide substituted pyrrolamide derivatives and the use thereof as medicaments for the treatment of hepatitis b |
| NZ717629A (en) | 2013-10-23 | 2021-12-24 | Janssen Sciences Ireland Uc | Carboxamide derivatives and the use thereof as medicaments for the treatment of hepatitis b |
| US9169212B2 (en) | 2014-01-16 | 2015-10-27 | Novira Therapeutics, Inc. | Azepane derivatives and methods of treating hepatitis B infections |
| US10392349B2 (en) | 2014-01-16 | 2019-08-27 | Novira Therapeutics, Inc. | Azepane derivatives and methods of treating hepatitis B infections |
| ES2792848T3 (en) | 2014-02-05 | 2020-11-12 | Novira Therapeutics Inc | Combination therapy for the treatment of HBV infections |
| CA2932551A1 (en) | 2014-02-06 | 2015-08-13 | Janssen Sciences Ireland Uc | Sulphamoylpyrrolamide derivatives and the use thereof as medicaments for the treatment of hepatitis b |
| WO2016057306A1 (en) | 2014-10-06 | 2016-04-14 | University Of Houston | Rho associated kinase (rock) inhibitors and their use in treating disease |
| CN107847762A (en) | 2015-03-19 | 2018-03-27 | 诺维拉治疗公司 | Azacyclooctane and azacyclo- nonane derivatives and the method for treating hepatitis B infection |
| US10875876B2 (en) | 2015-07-02 | 2020-12-29 | Janssen Sciences Ireland Uc | Cyclized sulfamoylarylamide derivatives and the use thereof as medicaments for the treatment of hepatitis B |
| AU2016330964B2 (en) | 2015-09-29 | 2021-04-01 | Novira Therapeutics, Inc. | Crystalline forms of a hepatitis B antiviral agent |
| JP6832946B2 (en) | 2015-11-17 | 2021-02-24 | アエリエ ファーマシューティカルズ インコーポレイテッド | How to prepare kinase inhibitors and their intermediates |
| US9643927B1 (en) | 2015-11-17 | 2017-05-09 | Aerie Pharmaceuticals, Inc. | Process for the preparation of kinase inhibitors and intermediates thereof |
| SG11201808949SA (en) | 2016-04-15 | 2018-11-29 | Novira Therapeutics Inc | Combinations and methods comprising a capsid assembly inhibitor |
| US11389441B2 (en) | 2016-08-31 | 2022-07-19 | Aerie Pharmaceuticals, Inc. | Ophthalmic compositions |
| CN110325535A (en) * | 2017-02-27 | 2019-10-11 | 詹森药业有限公司 | [1,2,4]-triazole [the 1,5-A]-pyrimidinyl derivatives as OGA inhibitor replaced through piperidines, morpholine or piperazine |
| MX2019011784A (en) | 2017-03-31 | 2019-11-18 | Aerie Pharmaceuticals Inc | Aryl cyclopropyl-amino-isoquinolinyl amide compounds. |
| JP2021502400A (en) * | 2017-11-03 | 2021-01-28 | アクラリス セラピューティクス,インコーポレイテッド | Substituted pyrolopyrimidine JAK inhibitor and method for making and using it |
| WO2019175657A1 (en) | 2018-03-14 | 2019-09-19 | Janssen Sciences Ireland Unlimited Company | Capsid assembly modulator dosing regimen |
| CN108929258A (en) * | 2018-07-09 | 2018-12-04 | 张立国 | A kind of Rho kinase inhibitor and its application in cancer treatment |
| CN108569990A (en) * | 2018-07-09 | 2018-09-25 | 张立国 | Polysubstituted pyrrole alkane derivatives and its application as Rho kinase inhibitors in cancer |
| MX2021001558A (en) | 2018-08-10 | 2021-04-28 | Aclaris Therapeutics Inc | Pyrrolopyrimidine itk inhibitors. |
| WO2020047229A1 (en) | 2018-08-29 | 2020-03-05 | University Of Massachusetts | Inhibition of protein kinases to treat friedreich ataxia |
| EP3849555A4 (en) | 2018-09-14 | 2022-05-04 | Aerie Pharmaceuticals, Inc. | Aryl cyclopropyl-amino-isoquinolinyl amide compounds |
| WO2020072456A1 (en) * | 2018-10-02 | 2020-04-09 | Case Western Reserve University | Compounds for treating myelin related disorders |
| KR20210130753A (en) | 2019-02-22 | 2021-11-01 | 얀센 사이언시즈 아일랜드 언리미티드 컴퍼니 | Amide derivatives useful for the treatment of HBV infection or HBV-induced disease |
| TW202108576A (en) | 2019-05-06 | 2021-03-01 | 愛爾蘭商健生科學愛爾蘭無限公司 | Amide derivatives useful in the treatment of hbv infection or hbv-induced diseases |
| CN110215519A (en) * | 2019-07-12 | 2019-09-10 | 温州医科大学附属眼视光医院 | Drug modification type intraocular lens and its preparation method and application |
| CN114133394B (en) * | 2020-08-12 | 2023-12-08 | 赛诺哈勃药业(成都)有限公司 | Compound selectively aiming at activity of cell cycle dependent kinase 12, preparation method and medical application |
| CN116891460A (en) * | 2023-07-12 | 2023-10-17 | 浙江大学 | Indazole derivative or pharmaceutically acceptable salt thereof and application thereof |
Citations (20)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2001056988A1 (en) | 2000-02-01 | 2001-08-09 | Kirin Beer Kabushiki Kaisha | Nitrogen-containing compounds having kinase inhibitory activity and drugs containing the same |
| WO2002068406A2 (en) | 2001-01-12 | 2002-09-06 | Amgen Inc. | Substituted amine derivatives and their use for the treatment of angiogenesis |
| US6555539B2 (en) | 2000-01-18 | 2003-04-29 | Agouron Pharmaceuticals | Indazole compounds, pharmaceutical compositions, and methods for mediating or inhibiting cell proliferation |
| WO2003042172A2 (en) | 2001-11-14 | 2003-05-22 | Bristol-Myers Squibb Company | C-5 modified indazolylpyrrolotriazines |
| EP1403255A1 (en) | 2001-06-12 | 2004-03-31 | Sumitomo Pharmaceuticals Company, Limited | Rho KINASE INHIBITORS |
| US20040106646A1 (en) | 2001-04-11 | 2004-06-03 | Yoshiko Takayama | Visual function disorder improving agents |
| US6831175B2 (en) | 2001-12-13 | 2004-12-14 | Abbott Laboratories | Kinase inhibitors |
| US6867221B2 (en) | 2001-08-30 | 2005-03-15 | Kowa Co., Ltd. | Cyclic amine compounds and pharmaceutical composition containing the same |
| US6906070B2 (en) | 1997-06-20 | 2005-06-14 | Bristol-Myers Squibb Pharma Company | Guanidine mimics as factor Xa inhibitors |
| EP1541151A1 (en) | 2002-08-29 | 2005-06-15 | Santen Pharmaceutical Co., Ltd. | REMEDY FOR GLAUCOMA COMPRISING Rho KINASE INHIBITOR AND PROSTAGLANDINS |
| EP1550660A1 (en) | 2002-09-12 | 2005-07-06 | Kirin Beer Kabushiki Kaisha | Isoquinoline derivatives having kinasae inhibitory activity and drugs containing the same |
| US20050148640A1 (en) | 2003-08-20 | 2005-07-07 | Come Jon H. | Aminofurazan compounds useful as protein kinase inhibitors |
| US6982274B2 (en) | 2001-04-16 | 2006-01-03 | Eisai Co., Ltd. | 1H-indazole compound |
| WO2006051311A1 (en) | 2004-11-12 | 2006-05-18 | Galapagos Nv | Nitrogen heteroaromatic compounds which bind to the active site of protein kinase enzymes |
| US20060183906A1 (en) | 2004-12-22 | 2006-08-17 | Rodgers James D | Pyrrolo[2,3-b]pyridin-4-yl-amines and pyrrolo[2,3-b]pyrimidin-4-yl-amines as janus kinase inhibitors |
| WO2006127587A1 (en) | 2005-05-20 | 2006-11-30 | Vertex Pharmaceuticals Incorporated | Pyrrolopyridines useful as inhibitors of protein kinase |
| WO2006135383A2 (en) | 2004-08-04 | 2006-12-21 | Myriad Genetics, Inc. | Indazoles |
| WO2007007919A2 (en) | 2005-07-14 | 2007-01-18 | Astellas Pharma Inc. | Heterocyclic janus kinase 3 inhibitors |
| US20070054916A1 (en) | 2004-10-01 | 2007-03-08 | Amgen Inc. | Aryl nitrogen-containing bicyclic compounds and methods of use |
| US20070135479A1 (en) | 2005-12-08 | 2007-06-14 | N.V. Organon | Isoquinoline derivatives |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4524816A (en) * | 1984-02-21 | 1985-06-25 | Dentsply Research & Development Corp. | Centrifugal casting furnace |
-
2007
- 2007-12-17 US US11/958,214 patent/US8071779B2/en active Active
- 2007-12-18 BR BRPI0720457-4A patent/BRPI0720457A2/en not_active IP Right Cessation
- 2007-12-18 CN CN2007800496081A patent/CN101583361B/en not_active Expired - Fee Related
- 2007-12-18 KR KR1020097012595A patent/KR20090091767A/en not_active Application Discontinuation
- 2007-12-18 CA CA002672825A patent/CA2672825A1/en not_active Abandoned
- 2007-12-18 WO PCT/US2007/087973 patent/WO2008077057A2/en active Search and Examination
- 2007-12-18 JP JP2009541644A patent/JP5511385B2/en not_active Expired - Fee Related
- 2007-12-18 MX MX2009006493A patent/MX2009006493A/en unknown
- 2007-12-18 CN CN2012105761391A patent/CN103254172A/en active Pending
- 2007-12-18 EP EP07869450A patent/EP2099457A4/en not_active Withdrawn
- 2007-12-18 AU AU2007333715A patent/AU2007333715B2/en not_active Ceased
- 2007-12-18 RU RU2009127798/04A patent/RU2470928C2/en not_active IP Right Cessation
-
2011
- 2011-10-13 US US13/273,043 patent/US8604218B2/en active Active
-
2012
- 2012-09-13 US US13/614,641 patent/US8604205B2/en active Active
Patent Citations (25)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6906070B2 (en) | 1997-06-20 | 2005-06-14 | Bristol-Myers Squibb Pharma Company | Guanidine mimics as factor Xa inhibitors |
| US6555539B2 (en) | 2000-01-18 | 2003-04-29 | Agouron Pharmaceuticals | Indazole compounds, pharmaceutical compositions, and methods for mediating or inhibiting cell proliferation |
| EP1256574A1 (en) | 2000-02-01 | 2002-11-13 | Kirin Beer Kabushiki Kaisha | Nitrogen-containing compounds having kinase inhibitory activity and drugs containing the same |
| US7217722B2 (en) | 2000-02-01 | 2007-05-15 | Kirin Beer Kabushiki Kaisha | Nitrogen-containing compounds having kinase inhibitory activity and drugs containing the same |
| WO2001056988A1 (en) | 2000-02-01 | 2001-08-09 | Kirin Beer Kabushiki Kaisha | Nitrogen-containing compounds having kinase inhibitory activity and drugs containing the same |
| WO2002068406A2 (en) | 2001-01-12 | 2002-09-06 | Amgen Inc. | Substituted amine derivatives and their use for the treatment of angiogenesis |
| US20040106646A1 (en) | 2001-04-11 | 2004-06-03 | Yoshiko Takayama | Visual function disorder improving agents |
| US6982274B2 (en) | 2001-04-16 | 2006-01-03 | Eisai Co., Ltd. | 1H-indazole compound |
| US20040138286A1 (en) | 2001-06-12 | 2004-07-15 | Naonori Imazaki | Rho kinase inhibitors |
| EP1403255A1 (en) | 2001-06-12 | 2004-03-31 | Sumitomo Pharmaceuticals Company, Limited | Rho KINASE INHIBITORS |
| US6867221B2 (en) | 2001-08-30 | 2005-03-15 | Kowa Co., Ltd. | Cyclic amine compounds and pharmaceutical composition containing the same |
| WO2003042172A2 (en) | 2001-11-14 | 2003-05-22 | Bristol-Myers Squibb Company | C-5 modified indazolylpyrrolotriazines |
| US6831175B2 (en) | 2001-12-13 | 2004-12-14 | Abbott Laboratories | Kinase inhibitors |
| EP1541151A1 (en) | 2002-08-29 | 2005-06-15 | Santen Pharmaceutical Co., Ltd. | REMEDY FOR GLAUCOMA COMPRISING Rho KINASE INHIBITOR AND PROSTAGLANDINS |
| EP1550660A1 (en) | 2002-09-12 | 2005-07-06 | Kirin Beer Kabushiki Kaisha | Isoquinoline derivatives having kinasae inhibitory activity and drugs containing the same |
| US20060167043A1 (en) | 2002-09-12 | 2006-07-27 | Kirin Beer Kabushiki Kaisha | Isoquinoline derivatives having kinasae inhibitory activity and drugs containing the same |
| US7615564B2 (en) | 2002-09-12 | 2009-11-10 | Kirin Beer Kabushiki Kaisha | Isoquinoline derivatives having kinasae inhibitory activity and drugs containing the same |
| US20050148640A1 (en) | 2003-08-20 | 2005-07-07 | Come Jon H. | Aminofurazan compounds useful as protein kinase inhibitors |
| WO2006135383A2 (en) | 2004-08-04 | 2006-12-21 | Myriad Genetics, Inc. | Indazoles |
| US20070054916A1 (en) | 2004-10-01 | 2007-03-08 | Amgen Inc. | Aryl nitrogen-containing bicyclic compounds and methods of use |
| WO2006051311A1 (en) | 2004-11-12 | 2006-05-18 | Galapagos Nv | Nitrogen heteroaromatic compounds which bind to the active site of protein kinase enzymes |
| US20060183906A1 (en) | 2004-12-22 | 2006-08-17 | Rodgers James D | Pyrrolo[2,3-b]pyridin-4-yl-amines and pyrrolo[2,3-b]pyrimidin-4-yl-amines as janus kinase inhibitors |
| WO2006127587A1 (en) | 2005-05-20 | 2006-11-30 | Vertex Pharmaceuticals Incorporated | Pyrrolopyridines useful as inhibitors of protein kinase |
| WO2007007919A2 (en) | 2005-07-14 | 2007-01-18 | Astellas Pharma Inc. | Heterocyclic janus kinase 3 inhibitors |
| US20070135479A1 (en) | 2005-12-08 | 2007-06-14 | N.V. Organon | Isoquinoline derivatives |
Non-Patent Citations (9)
| Title |
|---|
| Dorwald F. A. Side Reactions in Organic Synthesis, 2005, Wiley: VCH, Weinheim p. IX of Preface p. 1-15. * |
| International Search Report for PCT/US07/87973, mailed May 9, 2008. |
| Iwakubo et al. Bioorganic & Medicinal Chemistry 15 (2007) 350-364. * |
| Iwakubo, et al., "Design and synthesis of Rho kinase inhibitors (II)," Bioorganic & Medicinal Chem 15 (2007) 350-364. |
| Iwakubo, et al., "Design and synthesis of Rho kinase inhibitors (III)," Bioorganic & Medicinal Chem 15 (2007) 1022-1033. |
| Shimizu et al., "Parallel Coiled-coil Association of the RhoA-binding Domain in Rho-kinase," The Journal of Biological Chemistry, vol. 278, No. 46, Nov. 14, 2003, pp. 46046-46051. |
| Supplementary European Search Report, Jul. 14, 2011. |
| Takami, et al., "Design and synthesis of Rho kinase inhibitors (I)," Bioorganic & Medicinal Chem 12 (2004) 2115-2137. |
| Venkatesh et al., Role of the Development Scientist in Compound Lead Selection and Optimization J. Pharm. Sci. 89, 145-54 (2000). * |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2016180918A1 (en) | 2015-05-12 | 2016-11-17 | Platod | Combination of pharmacological and microfluidic features for improved platelets production |
Also Published As
| Publication number | Publication date |
|---|---|
| BRPI0720457A2 (en) | 2014-01-14 |
| CN103254172A (en) | 2013-08-21 |
| JP2010513319A (en) | 2010-04-30 |
| US20080214614A1 (en) | 2008-09-04 |
| CN101583361B (en) | 2013-01-23 |
| US20120046275A1 (en) | 2012-02-23 |
| AU2007333715B2 (en) | 2013-01-10 |
| EP2099457A2 (en) | 2009-09-16 |
| WO2008077057A3 (en) | 2008-08-21 |
| CN101583361A (en) | 2009-11-18 |
| CA2672825A1 (en) | 2008-06-26 |
| US8604205B2 (en) | 2013-12-10 |
| WO2008077057A2 (en) | 2008-06-26 |
| RU2009127798A (en) | 2011-01-27 |
| US20130012543A1 (en) | 2013-01-10 |
| EP2099457A4 (en) | 2011-08-17 |
| JP5511385B2 (en) | 2014-06-04 |
| MX2009006493A (en) | 2009-09-10 |
| RU2470928C2 (en) | 2012-12-27 |
| KR20090091767A (en) | 2009-08-28 |
| US8071779B2 (en) | 2011-12-06 |
| AU2007333715A1 (en) | 2008-06-26 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US8604218B2 (en) | Cytoskeletal active rho kinase inhibitor compounds, composition and use | |
| US20130131059A1 (en) | Method for treating ophthalmic diseases using kinase inhibitor compounds in prodrug forms | |
| US20130131106A1 (en) | Bifunctional rho kinase inhibitor compounds, composition and use | |
| JP5180834B2 (en) | Method of treatment | |
| JP7083073B2 (en) | Integrin antagonist | |
| US8039465B2 (en) | Cytoskeletal active compounds, composition and use | |
| US6960579B1 (en) | Serotonergic 5HT7 receptor compounds for treating ocular and CNS disorders |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| STCF | Information on status: patent grant |
Free format text: PATENTED CASE |
|
| AS | Assignment |
Owner name: MERCK SHARP & DOHME CORP., NEW JERSEY Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNOR:INSPIRE PHARMACEUTICALS, INC.;REEL/FRAME:034863/0372 Effective date: 20141023 |
|
| FPAY | Fee payment |
Year of fee payment: 4 |
|
| MAFP | Maintenance fee payment |
Free format text: PAYMENT OF MAINTENANCE FEE, 8TH YEAR, LARGE ENTITY (ORIGINAL EVENT CODE: M1552); ENTITY STATUS OF PATENT OWNER: LARGE ENTITY Year of fee payment: 8 |
|
| AS | Assignment |
Owner name: MERCK SHARP & DOHME LLC, NEW JERSEY Free format text: MERGER;ASSIGNOR:MERCK SHARP & DOHME CORP.;REEL/FRAME:061102/0145 Effective date: 20220407 |