US8058387B2 - Soluble polythiophene derivatives - Google Patents
Soluble polythiophene derivatives Download PDFInfo
- Publication number
- US8058387B2 US8058387B2 US12/412,651 US41265109A US8058387B2 US 8058387 B2 US8058387 B2 US 8058387B2 US 41265109 A US41265109 A US 41265109A US 8058387 B2 US8058387 B2 US 8058387B2
- Authority
- US
- United States
- Prior art keywords
- polythiophene derivative
- soluble polythiophene
- polymer
- soluble
- compound
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related, expires
Links
- 229920000123 polythiophene Polymers 0.000 title claims abstract description 41
- 230000005693 optoelectronics Effects 0.000 claims abstract description 15
- 239000010409 thin film Substances 0.000 claims abstract description 11
- XSCHRSMBECNVNS-UHFFFAOYSA-N quinoxaline Chemical compound N1=CC=NC2=CC=CC=C21 XSCHRSMBECNVNS-UHFFFAOYSA-N 0.000 claims description 12
- 125000003118 aryl group Chemical group 0.000 claims description 7
- GQPLMRYTRLFLPF-UHFFFAOYSA-N nitrous oxide Inorganic materials [O-][N+]#N GQPLMRYTRLFLPF-UHFFFAOYSA-N 0.000 claims description 6
- 125000000732 arylene group Chemical group 0.000 claims description 4
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 4
- FYNROBRQIVCIQF-UHFFFAOYSA-N pyrrolo[3,2-b]pyrrole-5,6-dione Chemical compound C1=CN=C2C(=O)C(=O)N=C21 FYNROBRQIVCIQF-UHFFFAOYSA-N 0.000 claims description 4
- DTDZVQXOCHUQLZ-UHFFFAOYSA-N thiadiazolo[5,4-f]quinoxaline Chemical compound C1=CC2=NC=CN=C2C2=C1N=NS2 DTDZVQXOCHUQLZ-UHFFFAOYSA-N 0.000 claims description 4
- YJSKZIATOGOJEB-UHFFFAOYSA-N thieno[2,3-b]pyrazine Chemical compound C1=CN=C2SC=CC2=N1 YJSKZIATOGOJEB-UHFFFAOYSA-N 0.000 claims description 4
- XFXPMWWXUTWYJX-UHFFFAOYSA-N Cyanide Chemical compound N#[C-] XFXPMWWXUTWYJX-UHFFFAOYSA-N 0.000 claims description 3
- 125000000217 alkyl group Chemical group 0.000 claims description 3
- 235000010290 biphenyl Nutrition 0.000 claims description 3
- 239000004305 biphenyl Substances 0.000 claims description 3
- 125000006267 biphenyl group Chemical group 0.000 claims description 3
- 125000004093 cyano group Chemical group *C#N 0.000 claims description 3
- NIHNNTQXNPWCJQ-UHFFFAOYSA-N fluorene Chemical compound C1=CC=C2CC3=CC=CC=C3C2=C1 NIHNNTQXNPWCJQ-UHFFFAOYSA-N 0.000 claims description 3
- 229910052736 halogen Inorganic materials 0.000 claims description 3
- 150000002367 halogens Chemical group 0.000 claims description 3
- 229910052739 hydrogen Inorganic materials 0.000 claims description 3
- 239000001257 hydrogen Substances 0.000 claims description 3
- 150000002431 hydrogen Chemical group 0.000 claims description 3
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims description 3
- ZUOUZKKEUPVFJK-UHFFFAOYSA-N phenylbenzene Natural products C1=CC=CC=C1C1=CC=CC=C1 ZUOUZKKEUPVFJK-UHFFFAOYSA-N 0.000 claims description 3
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 claims description 3
- SLLFVLKNXABYGI-UHFFFAOYSA-N 1,2,3-benzoxadiazole Chemical compound C1=CC=C2ON=NC2=C1 SLLFVLKNXABYGI-UHFFFAOYSA-N 0.000 claims description 2
- 125000005037 alkyl phenyl group Chemical group 0.000 claims description 2
- 125000005428 anthryl group Chemical group [H]C1=C([H])C([H])=C2C([H])=C3C(*)=C([H])C([H])=C([H])C3=C([H])C2=C1[H] 0.000 claims description 2
- 125000001624 naphthyl group Chemical group 0.000 claims description 2
- RWXCVESEMJNNMF-UHFFFAOYSA-N oxadiazolo[5,4-b]pyridine Chemical class C1=CN=C2ON=NC2=C1 RWXCVESEMJNNMF-UHFFFAOYSA-N 0.000 claims description 2
- 125000005561 phenanthryl group Chemical group 0.000 claims description 2
- 125000001725 pyrenyl group Chemical group 0.000 claims description 2
- YEYHFKBVNARCNE-UHFFFAOYSA-N pyrido[2,3-b]pyrazine Chemical compound N1=CC=NC2=CC=CN=C21 YEYHFKBVNARCNE-UHFFFAOYSA-N 0.000 claims description 2
- MABNMNVCOAICNO-UHFFFAOYSA-N selenophene Chemical compound C=1C=C[se]C=1 MABNMNVCOAICNO-UHFFFAOYSA-N 0.000 claims description 2
- QKTRRACPJVYJNU-UHFFFAOYSA-N thiadiazolo[5,4-b]pyridine Chemical compound C1=CN=C2SN=NC2=C1 QKTRRACPJVYJNU-UHFFFAOYSA-N 0.000 claims description 2
- 230000003993 interaction Effects 0.000 abstract description 7
- 230000021615 conjugation Effects 0.000 abstract description 3
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 75
- 229920000642 polymer Polymers 0.000 description 61
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 37
- 238000003786 synthesis reaction Methods 0.000 description 37
- 230000015572 biosynthetic process Effects 0.000 description 36
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 36
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 28
- 150000001875 compounds Chemical class 0.000 description 27
- MVPPADPHJFYWMZ-UHFFFAOYSA-N chlorobenzene Chemical compound ClC1=CC=CC=C1 MVPPADPHJFYWMZ-UHFFFAOYSA-N 0.000 description 22
- COIOYMYWGDAQPM-UHFFFAOYSA-N tris(2-methylphenyl)phosphane Chemical compound CC1=CC=CC=C1P(C=1C(=CC=CC=1)C)C1=CC=CC=C1C COIOYMYWGDAQPM-UHFFFAOYSA-N 0.000 description 22
- MCEWYIDBDVPMES-UHFFFAOYSA-N [60]pcbm Chemical compound C123C(C4=C5C6=C7C8=C9C%10=C%11C%12=C%13C%14=C%15C%16=C%17C%18=C(C=%19C=%20C%18=C%18C%16=C%13C%13=C%11C9=C9C7=C(C=%20C9=C%13%18)C(C7=%19)=C96)C6=C%11C%17=C%15C%13=C%15C%14=C%12C%12=C%10C%10=C85)=C9C7=C6C2=C%11C%13=C2C%15=C%12C%10=C4C23C1(CCCC(=O)OC)C1=CC=CC=C1 MCEWYIDBDVPMES-UHFFFAOYSA-N 0.000 description 20
- 239000011521 glass Substances 0.000 description 19
- 239000000243 solution Substances 0.000 description 19
- 238000006243 chemical reaction Methods 0.000 description 18
- 239000000463 material Substances 0.000 description 15
- 229920000301 poly(3-hexylthiophene-2,5-diyl) polymer Polymers 0.000 description 14
- -1 poly(3-hexylthiophene) Polymers 0.000 description 13
- HEDRZPFGACZZDS-MICDWDOJSA-N Trichloro(2H)methane Chemical compound [2H]C(Cl)(Cl)Cl HEDRZPFGACZZDS-MICDWDOJSA-N 0.000 description 12
- 238000001556 precipitation Methods 0.000 description 12
- 238000000605 extraction Methods 0.000 description 11
- 239000010408 film Substances 0.000 description 11
- 238000006116 polymerization reaction Methods 0.000 description 11
- CSNNHWWHGAXBCP-UHFFFAOYSA-L Magnesium sulfate Chemical compound [Mg+2].[O-][S+2]([O-])([O-])[O-] CSNNHWWHGAXBCP-UHFFFAOYSA-L 0.000 description 10
- 238000006392 deoxygenation reaction Methods 0.000 description 10
- 239000011541 reaction mixture Substances 0.000 description 9
- 239000003054 catalyst Substances 0.000 description 8
- 239000000203 mixture Substances 0.000 description 8
- CYPYTURSJDMMMP-WVCUSYJESA-N (1e,4e)-1,5-diphenylpenta-1,4-dien-3-one;palladium Chemical compound [Pd].[Pd].C=1C=CC=CC=1\C=C\C(=O)\C=C\C1=CC=CC=C1.C=1C=CC=CC=1\C=C\C(=O)\C=C\C1=CC=CC=C1.C=1C=CC=CC=1\C=C\C(=O)\C=C\C1=CC=CC=C1 CYPYTURSJDMMMP-WVCUSYJESA-N 0.000 description 7
- 239000000178 monomer Substances 0.000 description 7
- 239000004065 semiconductor Substances 0.000 description 7
- 239000007787 solid Substances 0.000 description 7
- KKRPPVXJVZKJON-UHFFFAOYSA-N trimethyl-(5-trimethylstannylthiophen-2-yl)stannane Chemical compound C[Sn](C)(C)C1=CC=C([Sn](C)(C)C)S1 KKRPPVXJVZKJON-UHFFFAOYSA-N 0.000 description 7
- 238000005160 1H NMR spectroscopy Methods 0.000 description 6
- YMSYWBKJXDMGNG-UHFFFAOYSA-N 3-hexyl-2-phenylthiophene Chemical compound C1=CSC(C=2C=CC=CC=2)=C1CCCCCC YMSYWBKJXDMGNG-UHFFFAOYSA-N 0.000 description 6
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 6
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 6
- 238000005481 NMR spectroscopy Methods 0.000 description 6
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 6
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 6
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Chemical compound O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 6
- 0 *C1(*)C2=CC3=C(C=C2C2=C1C=C(C)S2)C1=C(C=C([Ar]C)S1)C3(*)*.*C1(*)C2=CC3=C(C=C2C2=C1C=C([Ar]C)S2)C(*)(*)C1=C3SC(C)=C1 Chemical compound *C1(*)C2=CC3=C(C=C2C2=C1C=C(C)S2)C1=C(C=C([Ar]C)S1)C3(*)*.*C1(*)C2=CC3=C(C=C2C2=C1C=C([Ar]C)S2)C(*)(*)C1=C3SC(C)=C1 0.000 description 5
- YLQBMQCUIZJEEH-UHFFFAOYSA-N Furan Chemical compound C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 5
- PCLIMKBDDGJMGD-UHFFFAOYSA-N N-bromosuccinimide Chemical compound BrN1C(=O)CCC1=O PCLIMKBDDGJMGD-UHFFFAOYSA-N 0.000 description 5
- LNUFLCYMSVYYNW-ZPJMAFJPSA-N [(2r,3r,4s,5r,6r)-2-[(2r,3r,4s,5r,6r)-6-[(2r,3r,4s,5r,6r)-6-[(2r,3r,4s,5r,6r)-6-[[(3s,5s,8r,9s,10s,13r,14s,17r)-10,13-dimethyl-17-[(2r)-6-methylheptan-2-yl]-2,3,4,5,6,7,8,9,11,12,14,15,16,17-tetradecahydro-1h-cyclopenta[a]phenanthren-3-yl]oxy]-4,5-disulfo Chemical compound O([C@@H]1[C@@H](COS(O)(=O)=O)O[C@@H]([C@@H]([C@H]1OS(O)(=O)=O)OS(O)(=O)=O)O[C@@H]1[C@@H](COS(O)(=O)=O)O[C@@H]([C@@H]([C@H]1OS(O)(=O)=O)OS(O)(=O)=O)O[C@@H]1[C@@H](COS(O)(=O)=O)O[C@H]([C@@H]([C@H]1OS(O)(=O)=O)OS(O)(=O)=O)O[C@@H]1C[C@@H]2CC[C@H]3[C@@H]4CC[C@@H]([C@]4(CC[C@@H]3[C@@]2(C)CC1)C)[C@H](C)CCCC(C)C)[C@H]1O[C@H](COS(O)(=O)=O)[C@@H](OS(O)(=O)=O)[C@H](OS(O)(=O)=O)[C@H]1OS(O)(=O)=O LNUFLCYMSVYYNW-ZPJMAFJPSA-N 0.000 description 5
- 230000000052 comparative effect Effects 0.000 description 5
- 229920000547 conjugated polymer Polymers 0.000 description 5
- 239000000706 filtrate Substances 0.000 description 5
- 239000011888 foil Substances 0.000 description 5
- 229910052943 magnesium sulfate Inorganic materials 0.000 description 5
- 239000012074 organic phase Substances 0.000 description 5
- 239000004912 1,5-cyclooctadiene Substances 0.000 description 4
- 229920000144 PEDOT:PSS Polymers 0.000 description 4
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 4
- YTPLMLYBLZKORZ-UHFFFAOYSA-N Thiophene Chemical compound C=1C=CSC=1 YTPLMLYBLZKORZ-UHFFFAOYSA-N 0.000 description 4
- KGNDCEVUMONOKF-UGPLYTSKSA-N benzyl n-[(2r)-1-[(2s,4r)-2-[[(2s)-6-amino-1-(1,3-benzoxazol-2-yl)-1,1-dihydroxyhexan-2-yl]carbamoyl]-4-[(4-methylphenyl)methoxy]pyrrolidin-1-yl]-1-oxo-4-phenylbutan-2-yl]carbamate Chemical compound C1=CC(C)=CC=C1CO[C@H]1CN(C(=O)[C@@H](CCC=2C=CC=CC=2)NC(=O)OCC=2C=CC=CC=2)[C@H](C(=O)N[C@@H](CCCCN)C(O)(O)C=2OC3=CC=CC=C3N=2)C1 KGNDCEVUMONOKF-UGPLYTSKSA-N 0.000 description 4
- 229940125833 compound 23 Drugs 0.000 description 4
- 125000001072 heteroaryl group Chemical group 0.000 description 4
- 125000005549 heteroarylene group Chemical group 0.000 description 4
- 238000000034 method Methods 0.000 description 4
- 239000012299 nitrogen atmosphere Substances 0.000 description 4
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical class [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 3
- 229920001519 homopolymer Polymers 0.000 description 3
- RAXXELZNTBOGNW-UHFFFAOYSA-N imidazole Natural products C1=CNC=N1 RAXXELZNTBOGNW-UHFFFAOYSA-N 0.000 description 3
- 238000002360 preparation method Methods 0.000 description 3
- 239000000376 reactant Substances 0.000 description 3
- 238000010992 reflux Methods 0.000 description 3
- 238000010189 synthetic method Methods 0.000 description 3
- 150000003577 thiophenes Chemical class 0.000 description 3
- AOSZTAHDEDLTLQ-AZKQZHLXSA-N (1S,2S,4R,8S,9S,11S,12R,13S,19S)-6-[(3-chlorophenyl)methyl]-12,19-difluoro-11-hydroxy-8-(2-hydroxyacetyl)-9,13-dimethyl-6-azapentacyclo[10.8.0.02,9.04,8.013,18]icosa-14,17-dien-16-one Chemical compound C([C@@H]1C[C@H]2[C@H]3[C@]([C@]4(C=CC(=O)C=C4[C@@H](F)C3)C)(F)[C@@H](O)C[C@@]2([C@@]1(C1)C(=O)CO)C)N1CC1=CC=CC(Cl)=C1 AOSZTAHDEDLTLQ-AZKQZHLXSA-N 0.000 description 2
- SZUVGFMDDVSKSI-WIFOCOSTSA-N (1s,2s,3s,5r)-1-(carboxymethyl)-3,5-bis[(4-phenoxyphenyl)methyl-propylcarbamoyl]cyclopentane-1,2-dicarboxylic acid Chemical compound O=C([C@@H]1[C@@H]([C@](CC(O)=O)([C@H](C(=O)N(CCC)CC=2C=CC(OC=3C=CC=CC=3)=CC=2)C1)C(O)=O)C(O)=O)N(CCC)CC(C=C1)=CC=C1OC1=CC=CC=C1 SZUVGFMDDVSKSI-WIFOCOSTSA-N 0.000 description 2
- VYXHVRARDIDEHS-UHFFFAOYSA-N 1,5-cyclooctadiene Chemical compound C1CC=CCCC=C1 VYXHVRARDIDEHS-UHFFFAOYSA-N 0.000 description 2
- FCEHBMOGCRZNNI-UHFFFAOYSA-N 1-benzothiophene Chemical compound C1=CC=C2SC=CC2=C1 FCEHBMOGCRZNNI-UHFFFAOYSA-N 0.000 description 2
- UAFWSHOCWNNSKQ-UHFFFAOYSA-N 4-bromohexylbenzene Chemical compound CCC(Br)CCCC1=CC=CC=C1 UAFWSHOCWNNSKQ-UHFFFAOYSA-N 0.000 description 2
- NUKVKUBFUWBAEN-UHFFFAOYSA-N 5,7-dibromo-2,3-diphenylthieno[3,4-b]pyrazine Chemical compound C=1C=CC=CC=1C1=NC2=C(Br)SC(Br)=C2N=C1C1=CC=CC=C1 NUKVKUBFUWBAEN-UHFFFAOYSA-N 0.000 description 2
- ROFVEXUMMXZLPA-UHFFFAOYSA-N Bipyridyl Chemical group N1=CC=CC=C1C1=CC=CC=N1 ROFVEXUMMXZLPA-UHFFFAOYSA-N 0.000 description 2
- 229940126657 Compound 17 Drugs 0.000 description 2
- SIKJAQJRHWYJAI-UHFFFAOYSA-N Indole Chemical compound C1=CC=C2NC=CC2=C1 SIKJAQJRHWYJAI-UHFFFAOYSA-N 0.000 description 2
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 2
- KYQCOXFCLRTKLS-UHFFFAOYSA-N Pyrazine Chemical compound C1=CN=CC=N1 KYQCOXFCLRTKLS-UHFFFAOYSA-N 0.000 description 2
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical compound C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 2
- SMWDFEZZVXVKRB-UHFFFAOYSA-N Quinoline Chemical compound N1=CC=CC2=CC=CC=C21 SMWDFEZZVXVKRB-UHFFFAOYSA-N 0.000 description 2
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 2
- IOJUPLGTWVMSFF-UHFFFAOYSA-N benzothiazole Chemical compound C1=CC=C2SC=NC2=C1 IOJUPLGTWVMSFF-UHFFFAOYSA-N 0.000 description 2
- 229940126543 compound 14 Drugs 0.000 description 2
- 229920001577 copolymer Polymers 0.000 description 2
- 238000011161 development Methods 0.000 description 2
- 238000001914 filtration Methods 0.000 description 2
- 150000002500 ions Chemical class 0.000 description 2
- AWJUIBRHMBBTKR-UHFFFAOYSA-N isoquinoline Chemical compound C1=NC=CC2=CC=CC=C21 AWJUIBRHMBBTKR-UHFFFAOYSA-N 0.000 description 2
- 239000011777 magnesium Substances 0.000 description 2
- 229910052749 magnesium Inorganic materials 0.000 description 2
- 239000011259 mixed solution Substances 0.000 description 2
- 238000012986 modification Methods 0.000 description 2
- 230000004048 modification Effects 0.000 description 2
- 238000000746 purification Methods 0.000 description 2
- 150000003254 radicals Chemical class 0.000 description 2
- 239000000377 silicon dioxide Substances 0.000 description 2
- 239000002904 solvent Substances 0.000 description 2
- CXWXQJXEFPUFDZ-UHFFFAOYSA-N tetralin Chemical compound C1=CC=C2CCCCC2=C1 CXWXQJXEFPUFDZ-UHFFFAOYSA-N 0.000 description 2
- 229930192474 thiophene Natural products 0.000 description 2
- BCMCBBGGLRIHSE-UHFFFAOYSA-N 1,3-benzoxazole Chemical compound C1=CC=C2OC=NC2=C1 BCMCBBGGLRIHSE-UHFFFAOYSA-N 0.000 description 1
- JVVGLKKTAZYUQU-UHFFFAOYSA-N 1,4-bis(5-bromothiophen-2-yl)-2,5-bis(2-ethylhexyl)pyrrolo[3,4-c]pyrrole-3,6-dione Chemical compound CCCCC(CC)CN1C(=O)C2=C(C=3SC(Br)=CC=3)N(CC(CC)CCCC)C(=O)C2=C1C1=CC=C(Br)S1 JVVGLKKTAZYUQU-UHFFFAOYSA-N 0.000 description 1
- UNILWMWFPHPYOR-KXEYIPSPSA-M 1-[6-[2-[3-[3-[3-[2-[2-[3-[[2-[2-[[(2r)-1-[[2-[[(2r)-1-[3-[2-[2-[3-[[2-(2-amino-2-oxoethoxy)acetyl]amino]propoxy]ethoxy]ethoxy]propylamino]-3-hydroxy-1-oxopropan-2-yl]amino]-2-oxoethyl]amino]-3-[(2r)-2,3-di(hexadecanoyloxy)propyl]sulfanyl-1-oxopropan-2-yl Chemical compound O=C1C(SCCC(=O)NCCCOCCOCCOCCCNC(=O)COCC(=O)N[C@@H](CSC[C@@H](COC(=O)CCCCCCCCCCCCCCC)OC(=O)CCCCCCCCCCCCCCC)C(=O)NCC(=O)N[C@H](CO)C(=O)NCCCOCCOCCOCCCNC(=O)COCC(N)=O)CC(=O)N1CCNC(=O)CCCCCN\1C2=CC=C(S([O-])(=O)=O)C=C2CC/1=C/C=C/C=C/C1=[N+](CC)C2=CC=C(S([O-])(=O)=O)C=C2C1 UNILWMWFPHPYOR-KXEYIPSPSA-M 0.000 description 1
- QWENRTYMTSOGBR-UHFFFAOYSA-N 1H-1,2,3-Triazole Chemical compound C=1C=NNN=1 QWENRTYMTSOGBR-UHFFFAOYSA-N 0.000 description 1
- RSNQVABHABAKEZ-UHFFFAOYSA-N 2,3-diphenylquinoxaline Chemical compound C1=CC=CC=C1C1=NC2=CC=CC=C2N=C1C1=CC=CC=C1 RSNQVABHABAKEZ-UHFFFAOYSA-N 0.000 description 1
- KMVPSPDLRGMLEA-UHFFFAOYSA-N 2,3-diphenylthieno[3,4-b]pyrazine Chemical compound C=1C=CC=CC=1C1=NC2=CSC=C2N=C1C1=CC=CC=C1 KMVPSPDLRGMLEA-UHFFFAOYSA-N 0.000 description 1
- UUNIOFWUJYBVGQ-UHFFFAOYSA-N 2-amino-4-(3,4-dimethoxyphenyl)-10-fluoro-4,5,6,7-tetrahydrobenzo[1,2]cyclohepta[6,7-d]pyran-3-carbonitrile Chemical compound C1=C(OC)C(OC)=CC=C1C1C(C#N)=C(N)OC2=C1CCCC1=CC=C(F)C=C12 UUNIOFWUJYBVGQ-UHFFFAOYSA-N 0.000 description 1
- RSEBUVRVKCANEP-UHFFFAOYSA-N 2-pyrroline Chemical compound C1CC=CN1 RSEBUVRVKCANEP-UHFFFAOYSA-N 0.000 description 1
- DWWZELSEBSRUMM-UHFFFAOYSA-N 4-dodecyl-2-(4-dodecylthiophen-2-yl)thiophene Chemical compound CCCCCCCCCCCCC1=CSC(C=2SC=C(CCCCCCCCCCCC)C=2)=C1 DWWZELSEBSRUMM-UHFFFAOYSA-N 0.000 description 1
- NSPMIYGKQJPBQR-UHFFFAOYSA-N 4H-1,2,4-triazole Chemical compound C=1N=CNN=1 NSPMIYGKQJPBQR-UHFFFAOYSA-N 0.000 description 1
- LELMSURPXIAXOW-UHFFFAOYSA-N 5,8-dibromo-2,3-diphenylquinoxaline Chemical compound C=1C=CC=CC=1C=1N=C2C(Br)=CC=C(Br)C2=NC=1C1=CC=CC=C1 LELMSURPXIAXOW-UHFFFAOYSA-N 0.000 description 1
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 description 1
- YMDQTYHNHWGKLN-UHFFFAOYSA-L B=NS.BrC1=CC=CS1.C.CC1=CC(Br)=C(C)C=C1Br.CC1=CC=C(C2(C3=CC=C(C)C=C3)C3=CC4=C(C=C3C3=C2C=CS3)C(C2=CC=C(C)C=C2)(C2=CC=C(C)C=C2)C2=C4S/C=C\2)C=C1.CC1=CC=C([Mg]Br)C=C1.CCOC(=O)C1=CC(Br)=C(C(=O)OCC)C=C1Br.CCOC(=O)C1=CC(C2=CC=CS2)=C(C(=O)OCC)C=C1C1=CC=CS1.Cl[Zn]C1=CC=CS1.O=C(O)C1=CC(Br)=C(C(=O)O)C=C1Br Chemical compound B=NS.BrC1=CC=CS1.C.CC1=CC(Br)=C(C)C=C1Br.CC1=CC=C(C2(C3=CC=C(C)C=C3)C3=CC4=C(C=C3C3=C2C=CS3)C(C2=CC=C(C)C=C2)(C2=CC=C(C)C=C2)C2=C4S/C=C\2)C=C1.CC1=CC=C([Mg]Br)C=C1.CCOC(=O)C1=CC(Br)=C(C(=O)OCC)C=C1Br.CCOC(=O)C1=CC(C2=CC=CS2)=C(C(=O)OCC)C=C1C1=CC=CS1.Cl[Zn]C1=CC=CS1.O=C(O)C1=CC(Br)=C(C(=O)O)C=C1Br YMDQTYHNHWGKLN-UHFFFAOYSA-L 0.000 description 1
- UOMZWBZHPUOKKJ-UHFFFAOYSA-L B=NS.BrC1=CC=CS1.CC1=CC(C)=C(Br)C=C1Br.CC1=CC=C(C2(C3=CC=C(C)C=C3)C3=CC4=C(C=C3C3=C2C=C(Br)S3)C2=C(C=C(Br)S2)C4(C2=CC=C(C)C=C2)C2=CC=C(C)C=C2)C=C1.CC1=CC=C(C2(C3=CC=C(C)C=C3)C3=CC4=C(C=C3C3=C2C=CS3)C2=C(C=CS2)C4(C2=CC=C(C)C=C2)C2=CC=C(C)C=C2)C=C1.CC1=CC=C([Mg]Br)C=C1.CC1=CC=CC(C)=C1.CCO.CCOC(=O)C1=CC(C(=O)OCC)=C(Br)C=C1Br.CCOC(=O)C1=CC(C(=O)OCC)=C(C2=CC=CS2)C=C1C1=CC=CS1.Cl[Zn]C1=CC=CS1.O=C(O)C1=CC(C(=O)O)=C(Br)C=C1Br.S=S=S Chemical compound B=NS.BrC1=CC=CS1.CC1=CC(C)=C(Br)C=C1Br.CC1=CC=C(C2(C3=CC=C(C)C=C3)C3=CC4=C(C=C3C3=C2C=C(Br)S3)C2=C(C=C(Br)S2)C4(C2=CC=C(C)C=C2)C2=CC=C(C)C=C2)C=C1.CC1=CC=C(C2(C3=CC=C(C)C=C3)C3=CC4=C(C=C3C3=C2C=CS3)C2=C(C=CS2)C4(C2=CC=C(C)C=C2)C2=CC=C(C)C=C2)C=C1.CC1=CC=C([Mg]Br)C=C1.CC1=CC=CC(C)=C1.CCO.CCOC(=O)C1=CC(C(=O)OCC)=C(Br)C=C1Br.CCOC(=O)C1=CC(C(=O)OCC)=C(C2=CC=CS2)C=C1C1=CC=CS1.Cl[Zn]C1=CC=CS1.O=C(O)C1=CC(C(=O)O)=C(Br)C=C1Br.S=S=S UOMZWBZHPUOKKJ-UHFFFAOYSA-L 0.000 description 1
- ULBWSVDGDVRPSW-UHFFFAOYSA-N B=NS.CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(Br)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(Br)=C2)C=C1.CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=CS3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC=C2)C=C1.S=S=S=S=S Chemical compound B=NS.CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(Br)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(Br)=C2)C=C1.CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=CS3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC=C2)C=C1.S=S=S=S=S ULBWSVDGDVRPSW-UHFFFAOYSA-N 0.000 description 1
- FLIUSVPHJJMIOV-UHFFFAOYSA-N BC1=CC2=C(S1)C1=CC3=C(C=C1C2(C1=CC=C(CCCCCC)C=C1)C1=CC=C(CCCCCC)C=C1)C(C1=CC=C(CCCCCC)C=C1)(C1=CC=C(CCCCCC)C=C1)C1=C3SC(Br)=C1.CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=CS3)C2=C(C=CS2)C4(C2=CC=C(CCCCCC)C=C2)C2=CC=C(CCCCCC)C=C2)C=C1.S=S=S=S=S=S Chemical compound BC1=CC2=C(S1)C1=CC3=C(C=C1C2(C1=CC=C(CCCCCC)C=C1)C1=CC=C(CCCCCC)C=C1)C(C1=CC=C(CCCCCC)C=C1)(C1=CC=C(CCCCCC)C=C1)C1=C3SC(Br)=C1.CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=CS3)C2=C(C=CS2)C4(C2=CC=C(CCCCCC)C=C2)C2=CC=C(CCCCCC)C=C2)C=C1.S=S=S=S=S=S FLIUSVPHJJMIOV-UHFFFAOYSA-N 0.000 description 1
- FGEZNZGVBFJLNN-UHFFFAOYSA-N BrC1=C2N=C(C3=CC=CC=C3)C(C3=CC=CC=C3)=NC2=C(Br)C=C1.CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(Br)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(Br)=C2)C=C1.CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(C)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(C3=CC=C(C4=C5N=C(C6=CC=CC=C6)C(C6=CC=CC=C6)=NC5=C(C5=CC=C(C)S5)C=C4)S3)=C2)C=C1.C[Sn](C)(C)C1=CC=C([Sn](C)(C)C)S1 Chemical compound BrC1=C2N=C(C3=CC=CC=C3)C(C3=CC=CC=C3)=NC2=C(Br)C=C1.CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(Br)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(Br)=C2)C=C1.CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(C)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(C3=CC=C(C4=C5N=C(C6=CC=CC=C6)C(C6=CC=CC=C6)=NC5=C(C5=CC=C(C)S5)C=C4)S3)=C2)C=C1.C[Sn](C)(C)C1=CC=C([Sn](C)(C)C)S1 FGEZNZGVBFJLNN-UHFFFAOYSA-N 0.000 description 1
- NWFNVGWVGRHBST-UHFFFAOYSA-N BrC1=C2N=C(C3=CC=CC=C3)C(C3=CC=CC=C3)=NC2=C(Br)S1.CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(Br)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(Br)=C2)C=C1.CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(C)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(C3=CC=C(C4=C5N=C(C6=CC=CC=C6)C(C6=CC=CC=C6)=NC5=C(C5=CC=C(C)S5)S4)S3)=C2)C=C1.C[Sn](C)(C)C1=CC=C([Sn](C)(C)C)S1 Chemical compound BrC1=C2N=C(C3=CC=CC=C3)C(C3=CC=CC=C3)=NC2=C(Br)S1.CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(Br)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(Br)=C2)C=C1.CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(C)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(C3=CC=C(C4=C5N=C(C6=CC=CC=C6)C(C6=CC=CC=C6)=NC5=C(C5=CC=C(C)S5)S4)S3)=C2)C=C1.C[Sn](C)(C)C1=CC=C([Sn](C)(C)C)S1 NWFNVGWVGRHBST-UHFFFAOYSA-N 0.000 description 1
- RNVNMDXVXTXHRR-UHFFFAOYSA-N BrC1=CC=C(Br)C2=NSN=C12.CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(Br)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(Br)=C2)C=C1.CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(C2=CC=C(C5=CC=C(C6=CC=C(C)S6)C6=NSN=C56)S2)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(C)=C2)C=C1.C[Sn](C)(C)C1=CC=C([Sn](C)(C)C)S1 Chemical compound BrC1=CC=C(Br)C2=NSN=C12.CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(Br)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(Br)=C2)C=C1.CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(C2=CC=C(C5=CC=C(C6=CC=C(C)S6)C6=NSN=C56)S2)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(C)=C2)C=C1.C[Sn](C)(C)C1=CC=C([Sn](C)(C)C)S1 RNVNMDXVXTXHRR-UHFFFAOYSA-N 0.000 description 1
- WDHUHICJNUWGFG-UHFFFAOYSA-N BrC1=CC=C(Br)C2=NSN=C12.CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(Br)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(Br)=C2)C=C1.CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(C2=CC=C(C5=CC=C(C6=CC=C(C7=CC=C(C8=CC=C(C)S8)S7)C7=NSN=C67)S5)S2)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(C)=C2)C=C1.C[Sn](C)(C)C1=CC=C(C2=CC=C([Sn](C)(C)C)S2)S1 Chemical compound BrC1=CC=C(Br)C2=NSN=C12.CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(Br)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(Br)=C2)C=C1.CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(C2=CC=C(C5=CC=C(C6=CC=C(C7=CC=C(C8=CC=C(C)S8)S7)C7=NSN=C67)S5)S2)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(C)=C2)C=C1.C[Sn](C)(C)C1=CC=C(C2=CC=C([Sn](C)(C)C)S2)S1 WDHUHICJNUWGFG-UHFFFAOYSA-N 0.000 description 1
- UXJJGOFYHNJEQV-UHFFFAOYSA-M C1CCOC1.CCCCCCC1=CC=C(Br)C=C1.CCCCCCC1=CC=C(C(O)(C2=CC=C(CCCCCC)C=C2)C2=CC(C(O)(C3=CC=C(CCCCCC)C=C3)C3=CC=C(CCCCCC)C=C3)=C(C3=CC=CS3)C=C2C2=CC=CS2)C=C1.CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=CS3)C2=C(C=CS2)C4(C2=CC=C(CCCCCC)C=C2)C2=CC=C(CCCCCC)C=C2)C=C1.CCCCCCC1=CC=C([Mg]Br)C=C1.CCOC(=O)C1=CC(C(=O)OCC)=C(C2=CC=CS2)C=C1C1=CC=CS1 Chemical compound C1CCOC1.CCCCCCC1=CC=C(Br)C=C1.CCCCCCC1=CC=C(C(O)(C2=CC=C(CCCCCC)C=C2)C2=CC(C(O)(C3=CC=C(CCCCCC)C=C3)C3=CC=C(CCCCCC)C=C3)=C(C3=CC=CS3)C=C2C2=CC=CS2)C=C1.CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=CS3)C2=C(C=CS2)C4(C2=CC=C(CCCCCC)C=C2)C2=CC=C(CCCCCC)C=C2)C=C1.CCCCCCC1=CC=C([Mg]Br)C=C1.CCOC(=O)C1=CC(C(=O)OCC)=C(C2=CC=CS2)C=C1C1=CC=CS1 UXJJGOFYHNJEQV-UHFFFAOYSA-M 0.000 description 1
- CPDIWUTWCNDCAE-UHFFFAOYSA-M C1CCOC1.CCCCCCC1=CC=C(Br)C=C1.CCCCCCC1=CC=C(C(O)(C2=CC=C(CCCCCC)C=C2)C2=CC(C3=CC=CS3)=C(C(O)(C3=CC=C(CCCCCC)C=C3)C3=CC=C(CCCCCC)C=C3)C=C2C2=CC=CS2)C=C1.CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=CS3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC=C2)C=C1.CCCCCCC1=CC=C([Mg]Br)C=C1.CCOC(=O)C1=CC(C2=CC=CS2)=C(C(=O)OCC)C=C1C1=CC=CS1 Chemical compound C1CCOC1.CCCCCCC1=CC=C(Br)C=C1.CCCCCCC1=CC=C(C(O)(C2=CC=C(CCCCCC)C=C2)C2=CC(C3=CC=CS3)=C(C(O)(C3=CC=C(CCCCCC)C=C3)C3=CC=C(CCCCCC)C=C3)C=C2C2=CC=CS2)C=C1.CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=CS3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC=C2)C=C1.CCCCCCC1=CC=C([Mg]Br)C=C1.CCOC(=O)C1=CC(C2=CC=CS2)=C(C(=O)OCC)C=C1C1=CC=CS1 CPDIWUTWCNDCAE-UHFFFAOYSA-M 0.000 description 1
- XMWRBQBLMFGWIX-UHFFFAOYSA-N C60 fullerene Chemical compound C12=C3C(C4=C56)=C7C8=C5C5=C9C%10=C6C6=C4C1=C1C4=C6C6=C%10C%10=C9C9=C%11C5=C8C5=C8C7=C3C3=C7C2=C1C1=C2C4=C6C4=C%10C6=C9C9=C%11C5=C5C8=C3C3=C7C1=C1C2=C4C6=C2C9=C5C3=C12 XMWRBQBLMFGWIX-UHFFFAOYSA-N 0.000 description 1
- GJNSOHADHHMTKM-UHFFFAOYSA-N CC1=CC=C(C2(C3=CC=C(C)C=C3)C3=CC4=C(C=C3C3=C2C=C(Br)S3)C(C2=CC=C(C)C=C2)(C2=CC=C(C)C=C2)C2=C4S/C(Br)=C\2)C=C1.S=S Chemical compound CC1=CC=C(C2(C3=CC=C(C)C=C3)C3=CC4=C(C=C3C3=C2C=C(Br)S3)C(C2=CC=C(C)C=C2)(C2=CC=C(C)C=C2)C2=C4S/C(Br)=C\2)C=C1.S=S GJNSOHADHHMTKM-UHFFFAOYSA-N 0.000 description 1
- QNQXJHDDGDYPPU-UHFFFAOYSA-N CC1=CC=C(C2(C3=CC=C(C)C=C3)C3=CC4=C(C=C3C3=C2C=C(Br)S3)C(C2=CC=C(C)C=C2)(C2=CC=C(C)C=C2)C2=C4SC(Br)=C2)C=C1.CCCCCCCCCCCCC1=C(C)SC(C2=CC(CCCCCCCCCCCC)=C(C3=CC4=C(S3)C3=CC5=C(C=C3C4(C3=CC=C(C)C=C3)C3=CC=C(C)C=C3)C3=C(C=C(C)S3)C5(C3=CC=C(C)C=C3)C3=CC=C(C)C=C3)S2)=C1.CCCCCCCCCCCCC1=C([Sn](C)(C)C)SC(C2=CC(CCCCCCCCCCCC)=C([Sn](C)(C)C)S2)=C1.PP(P)P(P)P Chemical compound CC1=CC=C(C2(C3=CC=C(C)C=C3)C3=CC4=C(C=C3C3=C2C=C(Br)S3)C(C2=CC=C(C)C=C2)(C2=CC=C(C)C=C2)C2=C4SC(Br)=C2)C=C1.CCCCCCCCCCCCC1=C(C)SC(C2=CC(CCCCCCCCCCCC)=C(C3=CC4=C(S3)C3=CC5=C(C=C3C4(C3=CC=C(C)C=C3)C3=CC=C(C)C=C3)C3=C(C=C(C)S3)C5(C3=CC=C(C)C=C3)C3=CC=C(C)C=C3)S2)=C1.CCCCCCCCCCCCC1=C([Sn](C)(C)C)SC(C2=CC(CCCCCCCCCCCC)=C([Sn](C)(C)C)S2)=C1.PP(P)P(P)P QNQXJHDDGDYPPU-UHFFFAOYSA-N 0.000 description 1
- MKHQMSCTPGJODT-UHFFFAOYSA-N CC1=CC=C(C2(C3=CC=C(C)C=C3)C3=CC4=C(C=C3C3=C2C=C(Br)S3)C2=C(C=C(Br)S2)C4(C2=CC=C(C)C=C2)C2=CC=C(C)C=C2)C=C1.CCCCCCCCCCCCC1=C(C)SC(C2=CC(CCCCCCCCCCCC)=C(C3=CC4=C(S3)C3=C(C=C5C(=C3)C3=C(C=C(C)S3)C5(C3=CC=C(C)C=C3)C3=CC=C(C)C=C3)C4(C3=CC=C(C)C=C3)C3=CC=C(C)C=C3)S2)=C1.CCCCCCCCCCCCC1=C([Sn](C)(C)C)SC(C2=CC(CCCCCCCCCCCC)=C([Sn](C)(C)C)S2)=C1.PP(P)P(P)P(P)P Chemical compound CC1=CC=C(C2(C3=CC=C(C)C=C3)C3=CC4=C(C=C3C3=C2C=C(Br)S3)C2=C(C=C(Br)S2)C4(C2=CC=C(C)C=C2)C2=CC=C(C)C=C2)C=C1.CCCCCCCCCCCCC1=C(C)SC(C2=CC(CCCCCCCCCCCC)=C(C3=CC4=C(S3)C3=C(C=C5C(=C3)C3=C(C=C(C)S3)C5(C3=CC=C(C)C=C3)C3=CC=C(C)C=C3)C4(C3=CC=C(C)C=C3)C3=CC=C(C)C=C3)S2)=C1.CCCCCCCCCCCCC1=C([Sn](C)(C)C)SC(C2=CC(CCCCCCCCCCCC)=C([Sn](C)(C)C)S2)=C1.PP(P)P(P)P(P)P MKHQMSCTPGJODT-UHFFFAOYSA-N 0.000 description 1
- AUAYHINGFMNLPB-UHFFFAOYSA-N CCCCC(CC)CN1C(=O)C2=C(C3=CC=C(Br)S3)N(CC(CC)CCCC)C(=O)C2=C1C1=CC=C(Br)S1.CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(Br)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(Br)=C2)C=C1.CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(C)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(C3=CC=C(C4=CC=C(C5=C6C(=O)N(CC(CC)CCC)C(C7=CC=C(C8=CC=C(C)S8)S7)=C6C(=O)N5CC(CC)CCCC)S4)S3)=C2)C=C1.C[Sn](C)(C)C1=CC=C([Sn](C)(C)C)S1 Chemical compound CCCCC(CC)CN1C(=O)C2=C(C3=CC=C(Br)S3)N(CC(CC)CCCC)C(=O)C2=C1C1=CC=C(Br)S1.CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(Br)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(Br)=C2)C=C1.CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(C)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(C3=CC=C(C4=CC=C(C5=C6C(=O)N(CC(CC)CCC)C(C7=CC=C(C8=CC=C(C)S8)S7)=C6C(=O)N5CC(CC)CCCC)S4)S3)=C2)C=C1.C[Sn](C)(C)C1=CC=C([Sn](C)(C)C)S1 AUAYHINGFMNLPB-UHFFFAOYSA-N 0.000 description 1
- PZQNMCAYMJPPPN-QCDAUUDISA-N CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(Br)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(Br)=C2)C=C1.CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(C)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(C)=C2)C=C1.PP(P)P(P(P)P)P(P)P.[2H]C(=O)[Ni]C([2H])=O Chemical compound CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(Br)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(Br)=C2)C=C1.CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(C)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(C)=C2)C=C1.PP(P)P(P(P)P)P(P)P.[2H]C(=O)[Ni]C([2H])=O PZQNMCAYMJPPPN-QCDAUUDISA-N 0.000 description 1
- YNMDRJGSAOSTFB-UHFFFAOYSA-N CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(Br)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(Br)=C2)C=C1.CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(C)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(C3=CC=C(C)S3)=C2)C=C1.C[Sn](C)(C)C1=CC=C([Sn](C)(C)C)S1.PP(P)P(P)P(P(P)P)P(P)P Chemical compound CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(Br)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(Br)=C2)C=C1.CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(C)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(C3=CC=C(C)S3)=C2)C=C1.C[Sn](C)(C)C1=CC=C([Sn](C)(C)C)S1.PP(P)P(P)P(P(P)P)P(P)P YNMDRJGSAOSTFB-UHFFFAOYSA-N 0.000 description 1
- YGGNAZARAGQKDX-UHFFFAOYSA-N CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(Br)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(Br)=C2)C=C1.CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(C)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(C3=CC=C(C4=C5N=C(CCCCCC)C(CCCCCC)=NC5=C(C5=CC=C(C)S5)C5=NSN=C54)S3)=C2)C=C1.CCCCCCC1=NC2=C(Br)C3=NSN=C3C(Br)=C2N=C1CCCCCC.C[Sn](C)(C)C1=CC=C([Sn](C)(C)C)S1 Chemical compound CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(Br)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(Br)=C2)C=C1.CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(C)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(C3=CC=C(C4=C5N=C(CCCCCC)C(CCCCCC)=NC5=C(C5=CC=C(C)S5)C5=NSN=C54)S3)=C2)C=C1.CCCCCCC1=NC2=C(Br)C3=NSN=C3C(Br)=C2N=C1CCCCCC.C[Sn](C)(C)C1=CC=C([Sn](C)(C)C)S1 YGGNAZARAGQKDX-UHFFFAOYSA-N 0.000 description 1
- XOIQHAFYPARDBN-UHFFFAOYSA-N CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(Br)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(Br)=C2)C=C1.CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(C)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(C3=CC=C(C4=CC=C(C)S4)S3)=C2)C=C1.C[Sn](C)(C)C1=CC=C([Sn](C)(C)C)S1.PP(P)P(P(P)P)P(P(P)P)P(P)P Chemical compound CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(Br)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(Br)=C2)C=C1.CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(C)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(C3=CC=C(C4=CC=C(C)S4)S3)=C2)C=C1.C[Sn](C)(C)C1=CC=C([Sn](C)(C)C)S1.PP(P)P(P(P)P)P(P(P)P)P(P)P XOIQHAFYPARDBN-UHFFFAOYSA-N 0.000 description 1
- RNVPCSKIPQYETB-UHFFFAOYSA-N CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(C)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(C)=C2)C=C1.PP(P)P(P(P)P)P(P)P Chemical compound CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(C)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(C)=C2)C=C1.PP(P)P(P(P)P)P(P)P RNVPCSKIPQYETB-UHFFFAOYSA-N 0.000 description 1
- DHLGDCLUGXAUGB-UHFFFAOYSA-N CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(C)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(C3=CC=C(C)S3)=C2)C=C1.PP(P)P(P)P(P(P)P)P(P)P Chemical compound CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(C)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(C3=CC=C(C)S3)=C2)C=C1.PP(P)P(P)P(P(P)P)P(P)P DHLGDCLUGXAUGB-UHFFFAOYSA-N 0.000 description 1
- YZUDXBKMJZLWDT-UHFFFAOYSA-N CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(C)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(C3=CC=C(C4=C5N=C(C6=CC=CC=C6)C(C6=CC=CC=C6)=NC5=C(C5=CC=C(C)S5)S4)S3)=C2)C=C1.PPP(P)P(P(P(P)P)P(P)P)P(P(P(P)P)P(P)P)P(P(P)P)P(P)P Chemical compound CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(C)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(C3=CC=C(C4=C5N=C(C6=CC=CC=C6)C(C6=CC=CC=C6)=NC5=C(C5=CC=C(C)S5)S4)S3)=C2)C=C1.PPP(P)P(P(P(P)P)P(P)P)P(P(P(P)P)P(P)P)P(P(P)P)P(P)P YZUDXBKMJZLWDT-UHFFFAOYSA-N 0.000 description 1
- LOBAWKOVOGNXQO-UHFFFAOYSA-N CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(C)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(C3=CC=C(C4=C5N=C(CCCCCC)C(CCCCCC)=NC5=C(C5=CC=C(C)S5)C5=NSN=C54)S3)=C2)C=C1.PPP(P(P)P)P(P(P)P)P(P(P(P)P)P(P)P)P(P(P)P)P(P)P Chemical compound CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(C)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(C3=CC=C(C4=C5N=C(CCCCCC)C(CCCCCC)=NC5=C(C5=CC=C(C)S5)C5=NSN=C54)S3)=C2)C=C1.PPP(P(P)P)P(P(P)P)P(P(P(P)P)P(P)P)P(P(P)P)P(P)P LOBAWKOVOGNXQO-UHFFFAOYSA-N 0.000 description 1
- DGDPZQFBMKTAFF-UHFFFAOYSA-N CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(C)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(C3=CC=C(C4=CC=C(C)S4)S3)=C2)C=C1.PP(P)P(P(P)P)P(P(P)P)P(P)P Chemical compound CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(C)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(C3=CC=C(C4=CC=C(C)S4)S3)=C2)C=C1.PP(P)P(P(P)P)P(P(P)P)P(P)P DGDPZQFBMKTAFF-UHFFFAOYSA-N 0.000 description 1
- KNNYRJIYSNWNSO-UHFFFAOYSA-N CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(C)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(C3=CC=C(C4=CC=C(C5=C6C(=O)N(CC(CC)CCCC)C(C7=CC=C(C8=CC=C(C)S8)S7)=C6C(=O)N5CC(CC)CCCC)S4)S3)=C2)C=C1.PPP(P)P(P(P)P)P(P(P(P)P)P(P)P)P(P(P(P)P)P(P)P)P(P(P)P)P(P)P Chemical compound CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(C)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(C3=CC=C(C4=CC=C(C5=C6C(=O)N(CC(CC)CCCC)C(C7=CC=C(C8=CC=C(C)S8)S7)=C6C(=O)N5CC(CC)CCCC)S4)S3)=C2)C=C1.PPP(P)P(P(P)P)P(P(P(P)P)P(P)P)P(P(P(P)P)P(P)P)P(P(P)P)P(P)P KNNYRJIYSNWNSO-UHFFFAOYSA-N 0.000 description 1
- LQFXRTMYSLTBJR-UHFFFAOYSA-N CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(C)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(C3=CC=C(C4=CC=C(C5=CC=C(C)S5)C5=C4N=C(C4=CC=CC=C4)C(C4=CC=CC=C4)=N5)S3)=C2)C=C1.PPP(P(P)P)P(P(P(P)P)P(P)P)P(P(P(P)P)P(P)P)P(P(P)P)P(P)P Chemical compound CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(C)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(C3=CC=C(C4=CC=C(C5=CC=C(C)S5)C5=C4N=C(C4=CC=CC=C4)C(C4=CC=CC=C4)=N5)S3)=C2)C=C1.PPP(P(P)P)P(P(P(P)P)P(P)P)P(P(P(P)P)P(P)P)P(P(P)P)P(P)P LQFXRTMYSLTBJR-UHFFFAOYSA-N 0.000 description 1
- QMPIPCZTZUMSPA-UHFFFAOYSA-N CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(C2=CC=C(C5=CC=C(C6=CC=C(C)S6)C6=NSN=C56)S2)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(C)=C2)C=C1.PP(P)P(P(P)P)P(P(P)P)P(P(P)P)P(P)P Chemical compound CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(C2=CC=C(C5=CC=C(C6=CC=C(C)S6)C6=NSN=C56)S2)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(C)=C2)C=C1.PP(P)P(P(P)P)P(P(P)P)P(P(P)P)P(P)P QMPIPCZTZUMSPA-UHFFFAOYSA-N 0.000 description 1
- UAYPJMCYKGXWOO-UHFFFAOYSA-N CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(C2=CC=C(C5=CC=C(C6=CC=C(C7=CC=C(C8=CC=C(C)S8)S7)C7=NSN=C67)S5)S2)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(C)=C2)C=C1.PP(P)P(P)P(P(P)P)P(P(P)P)P(P)P Chemical compound CCCCCCC1=CC=C(C2(C3=CC=C(CCCCCC)C=C3)C3=CC4=C(C=C3C3=C2C=C(C2=CC=C(C5=CC=C(C6=CC=C(C7=CC=C(C8=CC=C(C)S8)S7)C7=NSN=C67)S5)S2)S3)C(C2=CC=C(CCCCCC)C=C2)(C2=CC=C(CCCCCC)C=C2)C2=C4SC(C)=C2)C=C1.PP(P)P(P)P(P(P)P)P(P(P)P)P(P)P UAYPJMCYKGXWOO-UHFFFAOYSA-N 0.000 description 1
- KWPXJJYRWOWMAL-UHFFFAOYSA-N CCCCCCCCCCCCC1=C(C)SC(C2=CC(CCCCCCCCCCCC)=C(C3=CC4=C(S3)C3=C(C=C5C(=C3)C3=C(C=C(C)S3)C5(C3=CC=C(C)C=C3)C3=CC=C(C)C=C3)C4(C3=CC=C(C)C=C3)C3=CC=C(C)C=C3)S2)=C1.PP(P)P(P)P(P)P Chemical compound CCCCCCCCCCCCC1=C(C)SC(C2=CC(CCCCCCCCCCCC)=C(C3=CC4=C(S3)C3=C(C=C5C(=C3)C3=C(C=C(C)S3)C5(C3=CC=C(C)C=C3)C3=CC=C(C)C=C3)C4(C3=CC=C(C)C=C3)C3=CC=C(C)C=C3)S2)=C1.PP(P)P(P)P(P)P KWPXJJYRWOWMAL-UHFFFAOYSA-N 0.000 description 1
- IZXOKUJBWHXVTB-UHFFFAOYSA-N CCCCCCCCCCCCC1=C(C)SC(C2=CC(CCCCCCCCCCCC)=C(C3=CC4=C(S3)C3=CC5=C(C=C3C4(C3=CC=C(C)C=C3)C3=CC=C(C)C=C3)C3=C(/C=C(/C)S3)C5(C3=CC=C(C)C=C3)C3=CC=C(C)C=C3)S2)=C1.PP(P)P(P)P Chemical compound CCCCCCCCCCCCC1=C(C)SC(C2=CC(CCCCCCCCCCCC)=C(C3=CC4=C(S3)C3=CC5=C(C=C3C4(C3=CC=C(C)C=C3)C3=CC=C(C)C=C3)C3=C(/C=C(/C)S3)C5(C3=CC=C(C)C=C3)C3=CC=C(C)C=C3)S2)=C1.PP(P)P(P)P IZXOKUJBWHXVTB-UHFFFAOYSA-N 0.000 description 1
- 206010008583 Chloroma Diseases 0.000 description 1
- QXNVGIXVLWOKEQ-UHFFFAOYSA-N Disodium Chemical compound [Na][Na] QXNVGIXVLWOKEQ-UHFFFAOYSA-N 0.000 description 1
- ZCQWOFVYLHDMMC-UHFFFAOYSA-N Oxazole Chemical compound C1=COC=N1 ZCQWOFVYLHDMMC-UHFFFAOYSA-N 0.000 description 1
- PCNDJXKNXGMECE-UHFFFAOYSA-N Phenazine Natural products C1=CC=CC2=NC3=CC=CC=C3N=C21 PCNDJXKNXGMECE-UHFFFAOYSA-N 0.000 description 1
- 229920001609 Poly(3,4-ethylenedioxythiophene) Polymers 0.000 description 1
- WTKZEGDFNFYCGP-UHFFFAOYSA-N Pyrazole Chemical compound C=1C=NNC=1 WTKZEGDFNFYCGP-UHFFFAOYSA-N 0.000 description 1
- CZPWVGJYEJSRLH-UHFFFAOYSA-N Pyrimidine Chemical compound C1=CN=CN=C1 CZPWVGJYEJSRLH-UHFFFAOYSA-N 0.000 description 1
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical compound [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 description 1
- 238000006619 Stille reaction Methods 0.000 description 1
- FZWLAAWBMGSTSO-UHFFFAOYSA-N Thiazole Chemical compound C1=CSC=N1 FZWLAAWBMGSTSO-UHFFFAOYSA-N 0.000 description 1
- DGEZNRSVGBDHLK-UHFFFAOYSA-N [1,10]phenanthroline Chemical compound C1=CN=C2C3=NC=CC=C3C=CC2=C1 DGEZNRSVGBDHLK-UHFFFAOYSA-N 0.000 description 1
- CWQJBIYNELJGBE-UHFFFAOYSA-N [3-dodecyl-5-(4-dodecyl-5-trimethylstannylthiophen-2-yl)thiophen-2-yl]-trimethylstannane Chemical compound S1C([Sn](C)(C)C)=C(CCCCCCCCCCCC)C=C1C1=CC(CCCCCCCCCCCC)=C([Sn](C)(C)C)S1 CWQJBIYNELJGBE-UHFFFAOYSA-N 0.000 description 1
- 239000002253 acid Substances 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- RFRXIWQYSOIBDI-UHFFFAOYSA-N benzarone Chemical compound CCC=1OC2=CC=CC=C2C=1C(=O)C1=CC=C(O)C=C1 RFRXIWQYSOIBDI-UHFFFAOYSA-N 0.000 description 1
- 229910052681 coesite Inorganic materials 0.000 description 1
- 238000001816 cooling Methods 0.000 description 1
- 229910052906 cristobalite Inorganic materials 0.000 description 1
- 229950000178 cyclopentobarbital Drugs 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 238000001704 evaporation Methods 0.000 description 1
- 230000008020 evaporation Effects 0.000 description 1
- RMBPEFMHABBEKP-UHFFFAOYSA-N fluorene Chemical compound C1=CC=C2C3=C[CH]C=CC3=CC2=C1 RMBPEFMHABBEKP-UHFFFAOYSA-N 0.000 description 1
- 229910003472 fullerene Inorganic materials 0.000 description 1
- 150000002390 heteroarenes Chemical class 0.000 description 1
- PZOUSPYUWWUPPK-UHFFFAOYSA-N indole Natural products CC1=CC=CC2=C1C=CN2 PZOUSPYUWWUPPK-UHFFFAOYSA-N 0.000 description 1
- RKJUIXBNRJVNHR-UHFFFAOYSA-N indolenine Natural products C1=CC=C2CC=NC2=C1 RKJUIXBNRJVNHR-UHFFFAOYSA-N 0.000 description 1
- ZLTPDFXIESTBQG-UHFFFAOYSA-N isothiazole Chemical compound C=1C=NSC=1 ZLTPDFXIESTBQG-UHFFFAOYSA-N 0.000 description 1
- CTAPFRYPJLPFDF-UHFFFAOYSA-N isoxazole Chemical compound C=1C=NOC=1 CTAPFRYPJLPFDF-UHFFFAOYSA-N 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- 229910052751 metal Inorganic materials 0.000 description 1
- 239000002184 metal Substances 0.000 description 1
- 229910052760 oxygen Inorganic materials 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- 229920001467 poly(styrenesulfonates) Polymers 0.000 description 1
- 239000002861 polymer material Substances 0.000 description 1
- 229920006389 polyphenyl polymer Polymers 0.000 description 1
- 238000007639 printing Methods 0.000 description 1
- 230000008569 process Effects 0.000 description 1
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 1
- ZVJHJDDKYZXRJI-UHFFFAOYSA-N pyrroline Natural products C1CC=NC1 ZVJHJDDKYZXRJI-UHFFFAOYSA-N 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 238000010898 silica gel chromatography Methods 0.000 description 1
- 229910052710 silicon Inorganic materials 0.000 description 1
- 239000010703 silicon Substances 0.000 description 1
- 235000012239 silicon dioxide Nutrition 0.000 description 1
- 239000000344 soap Substances 0.000 description 1
- 238000004528 spin coating Methods 0.000 description 1
- 238000012430 stability testing Methods 0.000 description 1
- 239000007858 starting material Substances 0.000 description 1
- 229910052682 stishovite Inorganic materials 0.000 description 1
- 229910052717 sulfur Inorganic materials 0.000 description 1
- 125000004434 sulfur atom Chemical group 0.000 description 1
- 229910052905 tridymite Inorganic materials 0.000 description 1
- DOIRPCDOGSNNCS-UHFFFAOYSA-N trimethyl-[5-(5-trimethylstannylthiophen-2-yl)thiophen-2-yl]stannane Chemical compound S1C([Sn](C)(C)C)=CC=C1C1=CC=C([Sn](C)(C)C)S1 DOIRPCDOGSNNCS-UHFFFAOYSA-N 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G61/00—Macromolecular compounds obtained by reactions forming a carbon-to-carbon link in the main chain of the macromolecule
- C08G61/12—Macromolecular compounds containing atoms other than carbon in the main chain of the macromolecule
- C08G61/122—Macromolecular compounds containing atoms other than carbon in the main chain of the macromolecule derived from five- or six-membered heterocyclic compounds, other than imides
- C08G61/123—Macromolecular compounds containing atoms other than carbon in the main chain of the macromolecule derived from five- or six-membered heterocyclic compounds, other than imides derived from five-membered heterocyclic compounds
- C08G61/126—Macromolecular compounds containing atoms other than carbon in the main chain of the macromolecule derived from five- or six-membered heterocyclic compounds, other than imides derived from five-membered heterocyclic compounds with a five-membered ring containing one sulfur atom in the ring
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/10—Organic polymers or oligomers
- H10K85/111—Organic polymers or oligomers comprising aromatic, heteroaromatic, or aryl chains, e.g. polyaniline, polyphenylene or polyphenylene vinylene
- H10K85/113—Heteroaromatic compounds comprising sulfur or selene, e.g. polythiophene
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/10—Organic polymers or oligomers
- H10K85/151—Copolymers
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G2261/00—Macromolecular compounds obtained by reactions forming a carbon-to-carbon link in the main chain of the macromolecule
- C08G2261/10—Definition of the polymer structure
- C08G2261/14—Side-groups
- C08G2261/148—Side-chains having aromatic units
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G2261/00—Macromolecular compounds obtained by reactions forming a carbon-to-carbon link in the main chain of the macromolecule
- C08G2261/30—Monomer units or repeat units incorporating structural elements in the main chain
- C08G2261/32—Monomer units or repeat units incorporating structural elements in the main chain incorporating heteroaromatic structural elements in the main chain
- C08G2261/322—Monomer units or repeat units incorporating structural elements in the main chain incorporating heteroaromatic structural elements in the main chain non-condensed
- C08G2261/3223—Monomer units or repeat units incorporating structural elements in the main chain incorporating heteroaromatic structural elements in the main chain non-condensed containing one or more sulfur atoms as the only heteroatom, e.g. thiophene
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G2261/00—Macromolecular compounds obtained by reactions forming a carbon-to-carbon link in the main chain of the macromolecule
- C08G2261/30—Monomer units or repeat units incorporating structural elements in the main chain
- C08G2261/32—Monomer units or repeat units incorporating structural elements in the main chain incorporating heteroaromatic structural elements in the main chain
- C08G2261/324—Monomer units or repeat units incorporating structural elements in the main chain incorporating heteroaromatic structural elements in the main chain condensed
- C08G2261/3241—Monomer units or repeat units incorporating structural elements in the main chain incorporating heteroaromatic structural elements in the main chain condensed containing one or more nitrogen atoms as the only heteroatom, e.g. carbazole
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G2261/00—Macromolecular compounds obtained by reactions forming a carbon-to-carbon link in the main chain of the macromolecule
- C08G2261/30—Monomer units or repeat units incorporating structural elements in the main chain
- C08G2261/32—Monomer units or repeat units incorporating structural elements in the main chain incorporating heteroaromatic structural elements in the main chain
- C08G2261/324—Monomer units or repeat units incorporating structural elements in the main chain incorporating heteroaromatic structural elements in the main chain condensed
- C08G2261/3243—Monomer units or repeat units incorporating structural elements in the main chain incorporating heteroaromatic structural elements in the main chain condensed containing one or more sulfur atoms as the only heteroatom, e.g. benzothiophene
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G2261/00—Macromolecular compounds obtained by reactions forming a carbon-to-carbon link in the main chain of the macromolecule
- C08G2261/30—Monomer units or repeat units incorporating structural elements in the main chain
- C08G2261/32—Monomer units or repeat units incorporating structural elements in the main chain incorporating heteroaromatic structural elements in the main chain
- C08G2261/324—Monomer units or repeat units incorporating structural elements in the main chain incorporating heteroaromatic structural elements in the main chain condensed
- C08G2261/3246—Monomer units or repeat units incorporating structural elements in the main chain incorporating heteroaromatic structural elements in the main chain condensed containing nitrogen and sulfur as heteroatoms
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G2261/00—Macromolecular compounds obtained by reactions forming a carbon-to-carbon link in the main chain of the macromolecule
- C08G2261/40—Polymerisation processes
- C08G2261/41—Organometallic coupling reactions
- C08G2261/414—Stille reactions
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G2261/00—Macromolecular compounds obtained by reactions forming a carbon-to-carbon link in the main chain of the macromolecule
- C08G2261/90—Applications
- C08G2261/91—Photovoltaic applications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G2261/00—Macromolecular compounds obtained by reactions forming a carbon-to-carbon link in the main chain of the macromolecule
- C08G2261/90—Applications
- C08G2261/92—TFT applications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G2261/00—Macromolecular compounds obtained by reactions forming a carbon-to-carbon link in the main chain of the macromolecule
- C08G2261/90—Applications
- C08G2261/95—Use in organic luminescent diodes
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K10/00—Organic devices specially adapted for rectifying, amplifying, oscillating or switching; Organic capacitors or resistors having potential barriers
- H10K10/40—Organic transistors
- H10K10/46—Field-effect transistors, e.g. organic thin-film transistors [OTFT]
- H10K10/462—Insulated gate field-effect transistors [IGFETs]
- H10K10/466—Lateral bottom-gate IGFETs comprising only a single gate
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K10/00—Organic devices specially adapted for rectifying, amplifying, oscillating or switching; Organic capacitors or resistors having potential barriers
- H10K10/40—Organic transistors
- H10K10/46—Field-effect transistors, e.g. organic thin-film transistors [OTFT]
- H10K10/462—Insulated gate field-effect transistors [IGFETs]
- H10K10/484—Insulated gate field-effect transistors [IGFETs] characterised by the channel regions
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K2102/00—Constructional details relating to the organic devices covered by this subclass
- H10K2102/10—Transparent electrodes, e.g. using graphene
- H10K2102/101—Transparent electrodes, e.g. using graphene comprising transparent conductive oxides [TCO]
- H10K2102/103—Transparent electrodes, e.g. using graphene comprising transparent conductive oxides [TCO] comprising indium oxides, e.g. ITO
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K30/00—Organic devices sensitive to infrared radiation, light, electromagnetic radiation of shorter wavelength or corpuscular radiation
- H10K30/30—Organic devices sensitive to infrared radiation, light, electromagnetic radiation of shorter wavelength or corpuscular radiation comprising bulk heterojunctions, e.g. interpenetrating networks of donor and acceptor material domains
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K30/00—Organic devices sensitive to infrared radiation, light, electromagnetic radiation of shorter wavelength or corpuscular radiation
- H10K30/50—Photovoltaic [PV] devices
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/10—Organic polymers or oligomers
- H10K85/111—Organic polymers or oligomers comprising aromatic, heteroaromatic, or aryl chains, e.g. polyaniline, polyphenylene or polyphenylene vinylene
- H10K85/113—Heteroaromatic compounds comprising sulfur or selene, e.g. polythiophene
- H10K85/1135—Polyethylene dioxythiophene [PEDOT]; Derivatives thereof
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E10/00—Energy generation through renewable energy sources
- Y02E10/50—Photovoltaic [PV] energy
- Y02E10/549—Organic PV cells
Definitions
- the present invention relates to a soluble polythiophene derivative containing highly coplanar repeating units, and in particular relates to using the soluble thiophene derivative in optoelectronic devices.
- the material has attracted considerable research interest because of the potential for being used in optoelectronic devices such as organic thin film transistors (OTFTs), organic light-emitting diodes (OLEDs), and organic solar cells (OSCs) and being mass produced.
- OFTs organic thin film transistors
- OLEDs organic light-emitting diodes
- OSCs organic solar cells
- the active layer of the optoelectronic device being fabricated by printing technology, material having polymer properties can produce a flexible optoelectronic device with advantages of light-weight, low cost and large area fabrication.
- polythiophene with carrier mobility of 10 ⁇ 5 cm 2 /Vs is first used as an active layer.
- P3HT poly(3-hexylthiophene)
- P8T2 poly(9,9-dioctylfluoreneco-bithiophene)
- the active layer For application of polymer semiconducting material by organic solar cells (OSCs), the active layer must be made of high mobility materials that can effectively transport holes and electrons, respectively. Otherwise, if the electrons and holes recombine, the carrier function will disappear, thus reducing power conversion efficiency of the OSCs.
- OSCs organic solar cells
- a solution having hetero-junction structure including an electron donor and an electron acceptor was proposed, which sparked development of OSCs. At the time, initial power conversion efficiency was about 1%.
- C60 material was used as an electron acceptor material, such as poly[2-methoxy-5-(2′-ethylhexyloxy)-1,4-phenylenevinylene] (MEH-PPV) with C60, P3HT with PCBM, thus improving power conversion efficiency in the range of 4 to 5%.
- MEH-PPV poly[2-methoxy-5-(2′-ethylhexyloxy)-1,4-phenylenevinylene]
- PCPDTBT poly[2,6]-(4,4-bis-(2-ethylhexyl)-4H-cyclopental[2,1-b;3,4-b′]dithiophene)-alt-4,7-(2,1,3-benzothiadiazole) polymer with a low bandgap, which improved power conversion efficiency of the polymer with PC 71 BM to 3.2%.
- PCPDTBT poly[2,6]-(4,4-bis-(2-ethylhexyl)-4H-cyclopental[2,1-b;3,4-b′]dithiophene)-alt-4,7-(2,1,3-benzothiadiazole
- the above-mentioned polymers such as P3HT F8T2 and PCPDTBT are polymerized by thiophene derivatives.
- the main chain is based on a planar conjugated thiophene derivative. Because of interaction between the side-chain molecules, the intermolecules ⁇ - ⁇ interaction and carrier mobility are improved.
- An object of the present invention is to provide a soluble polythiophene derivative containing highly coplanar repeating units.
- the coplanar characteristic of the TPT (thiophene-phenylene-thiophene) units improves the degree of intramolecular conjugation and intermolecular ⁇ - ⁇ interaction, further increasing carrier mobility.
- the present invention provides a soluble polythiophene derivative containing coplanar repeating units, having the structure of formula (I) or (II):
- R is hydrogen, alkyl, hydroxy, halogen, cyano (—CN), nitrous (—NO 2 ), amino, substituted or unsubstituted aryl, or substituted or unsubstituted heteroaryl
- Ar is substituted or unsubstituted arylene or heteroarylene
- m and n are the numbers of repeating units, where m is an integer between 2 and 1000, and n is an integer between 0 and 100.
- FIG. 1 is a bottom-contact organic thin-film transistor device.
- FIG. 2 is the carrier mobility and stability in air of the P14 and known P3HT
- FIG. 3 is the device of the organic solar cell.
- FIG. 4 is the HOMO (highest occupied molecular orbit) and LUMO (lowest unoccupied molecular orbit) energy level of the present invention and known P3HT and PCBM.
- FIG. 6 is the stability in air of the polymer of the present invention and other cells.
- the present invention provides a TPT or a long side-chain TPT as a monomer, and a suitable polymerization method to produce a coplanar polythiophene.
- the present invention provides a soluble polythiophene derivative containing coplanar repeating units, having the structure of formula (I) or (II):
- R is hydrogen, alkyl, hydroxy, halogen, cyano (—CN), nitrous (—NO 2 ), amino, substituted or unsubstituted aryl, or substituted or unsubstituted heteroaryl
- Ar is substituted or unsubstituted arylene or heteroarylene
- m and n are the numbers of repeating units, where m is an integer between 2 and 1000, n is an integer between 0 and 100.
- the aryl comprises phenyl, naphthyl, diphenyl, anthryl, pyrenyl, phenanthryl or fluorene.
- the aryl may be other forms of polyphenyl diphenyl.
- the heteroaryl comprises pyrane, pyrroline, furan, benzofuran, thiophene, benzothiophene, benzothiodiazole, pyridine, quinoline, isoquinoline, pyrazine, pyrimidine, pyrazole, imidazole, indole, thiazole, isothiazole, oxazole, isoxazole, benzothiazole, benzoxazole, 1,2,4-triazole, 1,2,3-triazole, phenanthroline, oxadiazolopyridine, pyridopyrazine, benzooxadiazole, thiadiazolopyridine, selenophene, thiadiazoloquinoxaline, thienopyrazine, quinoxaline or diketopyrrolopyrrole.
- the heteraryl may be other forms of heteroaromatic compound.
- the arylene is divalent radical of the above aryl groups
- the heteroarylene is divalent radical of the above heteroaryl groups.
- Ar is heteroarylene with sulfur atom, such as thiophene, dithiophene, benzothiodiazole (BT), thiadiazoloquinoxaline, thienopyrazine, quinoxaline or diketopyrrolopyrrole.
- the table shows several representative compounds of the polythiophene
- the molecular weight of the soluble polythiophene derivative is between 1000 and 1,000,000.
- the p-TPT or m-TPT was used as a reactant, Ni(COD), 1,5 cyclooctadiene and 2,2′-bipyridyl were used as a catalyst, and N,N-dimethylformamide and toluene were used as a solvent.
- the reaction was undertaken in N 2 , and the preferable temperature was about 60 ⁇ 150° C. After filtering and purification, the homopolymer was obtained.
- the p-TPT or m-TPT was used as a reactant and tris(dibenzylideneacetone)-dipalladium and tri(o-tolyl)phosphine were used as a catalyst.
- the mixture was dissolved in chlorobenzene.
- the polymer was synthesized by a Stille coupling reaction. After the degassed process, the mixture was put in a microwave reactor to polymerize. After filtering and purification, the copolymer was obtained.
- the soluble polythiophene derivative is suitable to be used in optoelectronic devices, such as organic thin film transistors (OTFTs), organic light-emitting diodes (OLEDs), or organic solar cells (OSCs).
- OFTs organic thin film transistors
- OLEDs organic light-emitting diodes
- OSCs organic solar cells
- the soluble polythiophene derivative is suitable to be used as an active layer of organic thin film transistors (OTFTs).
- OTFTs organic thin film transistors
- the soluble polythiophene derivative with carrier mobility was about 10 ⁇ 6 ⁇ 10 ⁇ 1 when used in an OTFT.
- the best carrier mobility was up to 3.02 ⁇ 10 ⁇ 3 .
- the carrier mobility was attributable to the TPT polymer being almost coplanar, thus consequently improving intramolecular conjugation, intermolecular ⁇ - ⁇ interaction and carrier mobility.
- the soluble polythiophene derivative was used as an active layer of organic light-emitting diodes (OLEDs).
- OLEDs organic light-emitting diodes
- the soluble polythiophene derivative was used as an active layer of organic solar cells (OSCs).
- OSCs organic solar cells
- the performances of the OSCs were seen as follows: the power conversion efficiency was about 0.5% ⁇ 3.3%, the open-circuit voltage was about 0.7 ⁇ 0.8 V, the short-circuit current was about 3 ⁇ 8 mA/cm 2 , and the fill factor was about 0.4 ⁇ 0.6.
- Scheme1 depicts the synthesis of compound S2 (p-TPT).
- Compound 8 was prepared by the synthetic method described in Org. Lett. 2006, 8, 5033-5036. 626 mg (1 mmol) of Compound 8 and 392 mg (2.2 mmol) of N-bromosuccinimide (NBS) were dissolved in 20 mL of chloroform in a 100 mL two-necked bottle. The bottle was wrapped by Al foil and kept under N 2 atmosphere overnight. The organic phase was extracted by a chloroform and saturated sodium chloride solution. Next, the organic phase was dried over anhydrous MgSO 4 and filtered. The filtrate was concentrated on a rotary evaporator. A pale yellow solid S2 was obtained after precipitation by methanol.
- NBS N-bromosuccinimide
- Scheme 2 depicts the synthesis of compound S3 (m-TPT).
- Compound 14 was prepared by the synthetic method described in Org. Lett. 2006, 8, 5033-5036.
- a pale yellow solid S3 was obtained following the same procedure for preparing S2, except that compound 14 (626 mg) was used as a starting material. (690 mg, 88%)
- Scheme 3 depicts the synthesis of compound 18.
- Compound 7 was prepared by the synthetic method described in Org. Lett. 2006, 8, 5033-5036. 1.2 g of magnesium flakes and a small amount of I 2 were placed in a 250 mL two-necked bottle, equipped with a charging funnel and a reflux condenser. 50 ⁇ L of anhydrous tetrahydrofuran and 10.2 mL of 4-bromo-hexylbenzene were added to the charging funnel. Several droplets of the tetrahydrofuran and 4-bromo-hexylbenzene were first added to initiate the reaction, and then the remaining amount was added dropwise. After the addition was complete, the resulting mixture was heated to reflux.
- Scheme 4 depicts the synthesis of compound S5 (para-alkyl-thiophene-phenylene -thiophene, p-ATPT).
- 907 mg (1 mmol) of Compound 18 and 392 mg (2.2 mmol) of NBS were placed in a 100 mL two-necked bottle. 30 mL of chrolorm was added to the bottle. Then, the whole bottle was wrapped by Al foil and immersed into an ice bath. The mixed solution was under N 2 atmosphere overnight. The organic phase was extracted by a chloroform and saturated sodium chloride solution and then was dried over anhydrous MgSO 4 and filtered. The filtrate was concentrated on a rotary evaporator to obtain a pale yellow solid S5 (980 mg, yield: 92%) by methanol.
- Scheme 5 depicts the synthesis of compound 23.
- a pale yellow solid 23 (435 mg) was obtained following the same procedure for preparing compound 18, except that compound 13 was used as a reactant. (435 mg, 48%)
- Scheme 6 depicts the synthesis of compound S6 (meta-alkyl-thiophene-phenylene -thiophene, m-ATPT).
- 907 mg (1 mmol) of Compound 23 and 392 mg (2.2 mmol) of NBS was placed in a 100 mL two-necked bottle. 30 mL of chrolorm was added to the bottle. Then, the whole bottle was wrapped by Al foil and immersed into an ice bath. The mixed solution was under N 2 atmosphere overnight. The organic phase was extracted by a chloroform and saturated sodium chloride solution and then was dried over anhydrous MgSO 4 and filtered. The filtrate was concentrated on a rotary evaporator to obtain a pale yellow solid S6 (586 mg, 55%) by methanol.
- Scheme 7 depicts the synthesis of compound P6.
- 0.3 mmol of p-TPT 5.5 mg (2 mol %) of 5,5′-bis-trimethylstanny 4,4′-bis(dodecyl)-2,2′-bithiophene, 14.6 mg (16 mol %) of tri(o-tolyl)phosphine and 5 mL of chlorobenzene were placed in a glass flask. After deoxygenation, the flask was placed in a microwave reactor (640 W, 30 min) for polymerization. After cooled to room temperature, the reaction mixture was added to methanol for precipitation.
- Scheme 8 depicts the synthesis of compound P8.
- 0.3 mmol of m-TPT 5.5 mg (2 mol %) of 5,5′-bis-trimethylstannyl-4,4′-bis(dodecyl)-2,2′-bithiophene, 14.6 mg (16 mol %) of tri(o-tolyl)phosphine and 5 mL of chlorobenzene were placed in a glass flask. After deoxygenation, the flask was placed in a microwave reactor (640 W, 30 min) for polymerization. After cooled to room temperature, the reaction mixture was added to methanol for precipitation.
- Scheme 9 depicts the synthesis of compound P10.
- 1.6 mmol of p-ATPT, 1.9 mmol of Ni(COD), 0.35 mL of 1,5-cyclooctadiene, 1.9 mol of 2,2′-bipyridyl, 15 mL of N,N-dimethylformamide and 15 mL of toluene were placed in a 100 mL glass flask.
- the mixture was under N 2 atmosphere at 60° C. for 48 hours. After cooled to room temperature, the mixture was washed with a disodium ethylenediamineteraacetate solution twice and DI water twice, respectively. The mixture was added to methanol for precipitation.
- Scheme 10 depicts the synthesis of compound P12.
- Scheme 11 depicts the synthesis of compound P14.
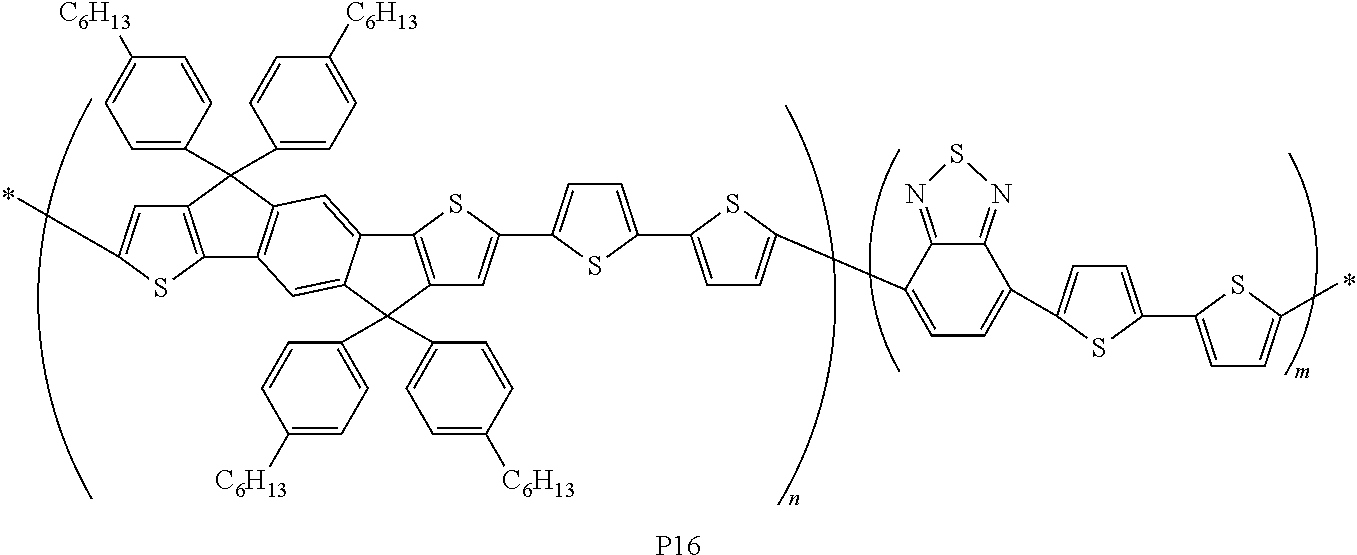
- Scheme 12 depicts the synthesis of compound P16.
- 0.15 mmol of p-ATPT, 0.3 mmol of 5,5′-bis-trimethylstannyl-2,2′-bithiophene, and 0.15 mmole of 4,7 bibromo-2,1,3 benzothiadiazole (BT) were as monomer and placed in a glass flask.
- 5.5 mg (2 mol %) of tris(dibenzylideneacetone)dipalladium, 14.6 mg (16 mol %) of tri(o-tolyl)phosphine and 5 mL of chlorobenzene were as catalyst.
- the flask was placed in a microwave reactor (640 W, 30 min) for polymerization.
- Scheme 13 depicts the synthesis of compound P18.
- 0.15 mmol of p-ATPT, 0.3 mmol of 2,5-bis-trimethylstannylthiophene, and 0.15 mmole of 4,7 bibromo-2,1,3 benzothiadiazole (BT) were as monomer and placed in a glass flask.
- 5.5 mg (2 mol %) of tris(dibenzylideneacetone)dipalladium, 14.6 mg (16 mol %) of tri(o-tolyl)phosphine and 5 mL of chlorobenzene were as catalyst.
- the flask was placed in a microwave reactor (640 W, 30 min) for polymerization.
- Scheme 14 depicts the synthesis of compound P25.
- 0.15 mmol of p-ATPT, 0.3 mmol of 2,5-bis-trimethylstannylthiophene, and 0.15 mmole of 6,7-dihexyl-4,9-dibromo[2,1,5]thiadiazolo[3,4g]quinoxaline(TQ) were as monomer and placed in a glass flask.
- 5.5 mg (2 mol %) of tris(dibenzylideneacetone)dipalladium, 14.6 mg (16 mol %) of tri(o-tolyl)phosphine and 10 mL of chlorobenzene were as catalyst.
- Scheme 15 depicts the synthesis of compound P27.
- 0.15 mmol of p-ATPT, 0.3 mmol of 2,5-bis-trimethylstannylthiophene, and 0.15 mmole of 5,7-dibromo-2,3-diphenylthieno[3,4-b]pyrazine (TP) were as monomer and placed in a glass flask.
- 5.5 mg (2 mol %) of tris(dibenzylideneacetone)dipalladium, 14.6 mg (16 mol %) of tri(o-tolyl)phosphine and 10 mL of chlorobenzene were as catalyst.
- the flask was placed in a microwave reactor (640 W, 30 min) for polymerization.
- Scheme 16 depicts the synthesis of compound P29.
- 0.15 mmol of p-ATPT, mmol of 2,5-bis-trimethylstannylthiophene, and 0.15 mmole of 5,8-dibromo-2,3-diphenylquinoxaline were as monomer and placed in a glass flask.
- 5.5 mg (2 mol %) of tris(dibenzylideneacetone)dipalladium, 14.6 mg (16 mol %) of tri(o-tolyl)phosphine and 10 mL of chlorobenzene were as catalyst.
- the flask was placed in a microwave reactor (640 W, 30 min) for polymerization.
- Scheme 17 depicts the synthesis of compound P31.
- 0.15 mmol of p-ATPT, 0.3 mmol of 2,5-bis-trimethylstannylthiophene, and 0.15 mmole of 3,6-bis -(5-bromo-thiophen-2-yl)-2,5-bis-(2-ethyl-hexyl)pyrrolo[3,4-c]pyrrole-1,4-dione were as monomer and placed in a glass flask.
- 5.5 mg (2 mol %) of tris(dibenzylideneacetone)dipalladium, 14.6 mg (16 mol %) of tri(o-tolyl)phosphine and 10 mL of chlorobenzene were as catalyst.
- FIG. 1 is a bottom-contact organic thin-film transistor device 10 , which comprise: an N + type silicon wafer as a gate electrode 11 ; a silicon dioxide insulating layer 12 , formed on the said gate electrode; a Pt wire as a source 13 and a drain electrode 14 , formed on opposite sides of the insulating layer; an active layer 15 , including the dissolved polythiophene derivative of the present invention, formed on the insulating layer.
- the width of the device was 1000 ⁇ m, the channel length between the source electrode and the drain electrode was 10 ⁇ m.
- the device was prepared as follows:
- the patterned metal of the organic thin-film transistor 10 was cleaned as follows: sonicated with soap water for 30 minutes, cleaned with DI water, sonicated with DI water for 30 minutes, sonicated with acetone for 30 minutes, sonicated with isopropane for 30 minutes, dried over by N 2 gun, and reserved in desiccator in vacuum.
- step 3 The transistor of step 1 was placed in a spin-coator.
- the solution of step 2 was filtered by a 0.45 ⁇ m filter plate.
- the solution of step 2 was uniformly coated on the transistor at a spin rate of 3000 rpm for 1 minute.
- the coated transistor was taken out and vacuumed at 150° C. for 1 hour in a dark room. After cooled to room temperature, the transistor was wrapped by Al foil and reserved in a desiccator in vacuum for the next step.
- the transistor was repeated measured for the electrical properties after time periods and observed for air-stability testing.
- the transistor was wrapped by Al foil and placed in a desiccator each time after the measurement.
- Table 1 is experimental data of OTFTs containing the polymer of the present invention, including carrier mobility and on/off ratio.
- FIG. 2 is the carrier mobility and stability in air of the P14 and known P3HT.
- the initial carrier mobility of P3HT was 5.2 ⁇ 10 ⁇ 3 , and the on/off ratio was 1.4 ⁇ 10 3 , after 16 days, the carrier mobility of P3HT became 1.7 ⁇ 10 ⁇ 3 , and the on/off ratio became 2.1 ⁇ 10 2 .
- the initial carrier mobility of P14 was 3.0 ⁇ 10 ⁇ 3 , and the on/off ratio was 1.2 ⁇ 10 5 , after 16 days, the carrier mobility of P14 became 8.1 ⁇ 10 ⁇ 4 , and the on/off ratio became 4.1 ⁇ 10 4 . Comparing the data, the loss of the carrier mobility of polymer P14 was close to P3HT, and the I on /I off in air was still larger than 10 4 , thus showing that the stability of P14 was better than P3HT.
- FIG. 3 is an organic solar cell device, which comprise: an ITO anode 21 ; a PEDOT:PSS (Poly(3,4-ethylenedioxythiophene):poly(styrene-sulfonate)) layer as hole transporting layer 22 , formed on the anode; an active layer 23 , including the dissolved polythiophene derivatives of the present invention, formed on the hole transporting layer; and a Ca/Al as a cathode 24 .
- PEDOT:PSS Poly(3,4-ethylenedioxythiophene):poly(styrene-sulfonate)
- an active layer 23 including the dissolved polythiophene derivatives of the present invention, formed on the hole transporting layer
- Ca/Al as a cathode 24 .
- the above-mentioned active layer was composed by the polymer of the present invention and [6,6]-phenyl-C-butyric acid methyl ester (PCBM).
- PCBM [6,6]-phenyl-C-butyric acid methyl ester
- the polymer and PCBM was mixed by the ratio of 1:3.
- the fullerene cluster in PCBM can be C61 or C71.
- the cell was measured under AM1.5G.
- the device was prepared as follows:
- the ITO glass was sonicated with acetone and isopropane for 15 minutes, respectively. Then, the ITO glass was dried by N 2 and placed in hot plate for 5 minutes.
- the ITO glass was placed in oxygen plasma for 5 minutes.
- PEDOT:PSS (Baytron P-VP AI4083) was coated on the ITO glass by spin coating with the rate of 3000 rpm/30 sec. Then, the ITO glass was baked in a dry box at 150° C. for one hour.
- the ITO glass was then coated on Polymer/PCBM(1:3, w:w) in a dry box with the rate of 1000 rpm/60 sec.
- the ITO glass was annealed on a hot plate at 140° C. for 20 minutes and left standing for cooling.
- a mask was placed on the ITO glass and a Ca/Al electrode was deposited by evaporation.
- Table 2 shows the power conversion efficiency, short-circuit current, open-circuit voltage and fill factor of polymer P6, P12, and P14.
- the power conversion efficiency of the P12/PC 71 BM and P14/PC 71 BM were 3.28% and 2.69%, respectively.
- the P12/PC 71 BM had the best performance with a short-circuit current of 7.48 mA/cm 2 and an open-circuit voltage of 0.80 V.
- the fill factor for the others was about 0.5.
- FIG. 4 is the HOMO (highest occupied molecular orbit) and LUMO (lowest unoccupied molecular orbit) energy level of the present invention and known P3HT and PCBM.
- the energy band gap of the polymer of the present invention was about 2.08 ⁇ 2.11 V and the HOMO and LUMO of polymers matched with those of PCBM, which indicated that the polymers were suitable as electron donor materials.
- the right side of the figure is the energy level of PCBM, which is the known electron acceptor material.
- FIG. 5 is the current density vs. voltage of polymer of the present invention with PCBM.
- the P12C70 had a highest current density of 7.48 mA/cm 2
- the P6C60 had a lowest current density of 3.49 mA/cm 2 .
- An organic solar cell as in Example 9 was prepared by using P3HT:PCBM as the active layer and PEDOT:PSS/AI4083 (IIC Stack) as the hole transporting material. The power conversion efficiency was measured on a daily basis.
- An organic solar cell as in Example 9 was prepared by using P3HT:PCBM as an active layer and PEDOT:PSS/Bytron P (HC Stack) as the hole transporting material. The power conversion efficiency was measured on a daily basis.
- FIG. 6 is the air stability of the polymer of the present invention and the Comparative Examples. After 29 days, the loss of power conversion efficiency of the P6:PCBM was about 17%, the loss of power conversion efficiency of the Comparative Example 1 was about 56%, and the loss of power conversion efficiency of Comparative Example 2 was about 35%, showing that the stability of the polymer of the present invention was better.
Landscapes
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Organic Chemistry (AREA)
- Polyoxymethylene Polymers And Polymers With Carbon-To-Carbon Bonds (AREA)
- Photovoltaic Devices (AREA)
Abstract
Description
| No. | R | Ar | Structure | |
| P6 | methyl- |
4,4′-bis(dodecyl)- 2,2′-bithiophene |
|
|
| P8 | methyl- |
4,4′-bis(dodecyl)- 2,2′-bithiophene |
|
|
| P10 | hexyl- phenyl | none |
|
|
| P12 | hexyl- phenyl | thiophene |
|
|
| P14 | hexyl- phenyl | dithiophene |
|
|
| P16 | hexyl- phenyl | dithiophene, benzothiodiazole |
|
|
| P18 | hexyl- phenyl | thiophene, benzothiodiazole |
|
|
| P25 | hexyl- phenyl | thiophene, 6,7-dihexyl [2,1,5]thiadiazolo[3, 4 g] quinoxaline |
|
|
| P27 | hexyl- phenyl | thiophene, 2,3- diphenylthieno[3,4- b] pyrazine) |
|
|
| P29 | hexyl- phenyl | thiophene, 2,3- diphenylquinoxaline |
|
|
| P31 | hexyl- | thiophene | 3,6-Bis-(thiophen-2- yl)-2,5-bis-(2-ethyl- hexyl) pyrrrolo[3,4- c]pyrrole-1,4-dione |
|
| polymer | μh(cm2/Vs) | Ion/Ioff | ||
| P6 | 3.72 × 10−4 | 5.15 × 103 | ||
| P8 | 5.83 × 10−6 | 1.59 × 103 | ||
| p10 | 1.45 × 10−4 | 1.41 × 104 | ||
| P12 | 8.26 × 10−4 | 9.10 × 104 | ||
| P14 | 3.02 × 10−3 | 1.25 × 106 | ||
| TABLE 2 | ||||
| power | short-circuit | open- | ||
| conversion | current | circuit | ||
| active layer | efficiency(%) | (mA/cm2) | voltage (V) | fill factor |
| P6/PC61BM | 1.08 | 3.49 | 0.792 | 0.392 |
| P10/PC61BM | 0.58 | 1.40 | 0.760 | 0.540 |
| P12/PC61BM | 2.16 | 5.28 | 0.766 | 0.533 |
| P12/PC71BM | 3.28 | 7.48 | 0.800 | 0.548 |
| P14/PC61BM | 1.67 | 4.55 | 0.759 | 0.485 |
| P14/PC71BM | 2.69 | 6.97 | 0.790 | 0.485 |
Claims (13)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US12/412,651 US8058387B2 (en) | 2008-05-30 | 2009-03-27 | Soluble polythiophene derivatives |
Applications Claiming Priority (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US12/130,917 US7754847B2 (en) | 2007-12-27 | 2008-05-30 | Soluble polythiophene derivatives |
| TW097150001A TWI375691B (en) | 2007-12-27 | 2008-12-22 | Soluble polythiophene derivatives |
| TW97150001A | 2008-12-22 | ||
| TW97150001 | 2008-12-22 | ||
| US12/412,651 US8058387B2 (en) | 2008-05-30 | 2009-03-27 | Soluble polythiophene derivatives |
Related Parent Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US12/130,917 Continuation-In-Part US7754847B2 (en) | 2007-12-27 | 2008-05-30 | Soluble polythiophene derivatives |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| US20090299029A1 US20090299029A1 (en) | 2009-12-03 |
| US8058387B2 true US8058387B2 (en) | 2011-11-15 |
Family
ID=41380618
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US12/412,651 Expired - Fee Related US8058387B2 (en) | 2008-05-30 | 2009-03-27 | Soluble polythiophene derivatives |
Country Status (1)
| Country | Link |
|---|---|
| US (1) | US8058387B2 (en) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20120142872A1 (en) * | 2009-05-27 | 2012-06-07 | Basf Se | Process for polymerising (hetero)aromatic compounds |
| US9428603B2 (en) | 2014-03-18 | 2016-08-30 | Council Of Scientific & Industrial Research | Polymeric dispersion of thiophene copolymers and a process for preparation thereof |
| US10991893B2 (en) | 2016-10-05 | 2021-04-27 | Raynergy Tek Incorporation | Organic semiconducting compounds |
Families Citing this family (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9017577B2 (en) * | 2008-08-18 | 2015-04-28 | Merck Patent Gmbh | Indacenodithiophene and indacenodiselenophene polymers and their use as organic semiconductors |
| US8691931B2 (en) | 2009-09-04 | 2014-04-08 | Plextronics, Inc. | Organic electronic devices and polymers, including photovoltaic cells and diketone-based and diketopyrrolopyrrole-based polymers |
| US8895693B2 (en) | 2010-06-25 | 2014-11-25 | Samsung Electronics Co., Ltd. | Electron-donating polymers and organic solar cells including the same |
| US8802812B2 (en) * | 2010-12-30 | 2014-08-12 | Ocean's King Lighting Science & Technology Co., Ltd. | Photovoltaic polymer material, preparation method and use thereof |
| CN102643284B (en) * | 2011-02-17 | 2014-11-26 | 兰州大成科技股份有限公司 | Preparation method and application of 4,9-bisalkylene indene thieno[3,2-b]thiophene derivative and conjugated polymer thereof |
| CN106025099B (en) * | 2011-04-12 | 2018-09-07 | 精工爱普生株式会社 | Light-emitting component, light-emitting device, authentication device and electronic equipment |
| KR101853395B1 (en) | 2011-05-23 | 2018-04-30 | 삼성전자주식회사 | Electron donating polymer and solar cell including the same |
| JP5935261B2 (en) * | 2011-08-09 | 2016-06-15 | セイコーエプソン株式会社 | Thiadiazole compounds, compounds for light emitting devices, light emitting devices, light emitting devices, authentication devices, and electronic devices |
| KR101777326B1 (en) | 2011-10-05 | 2017-09-12 | 삼성전자주식회사 | Electron donating polymer and organic solar cell including the same |
| CN104039864A (en) * | 2012-03-12 | 2014-09-10 | 海洋王照明科技股份有限公司 | Polymer containing thiophene-benzene-thiophene unit, preparation method therefor and solar cell device |
| US20140366947A1 (en) * | 2012-03-12 | 2014-12-18 | Ocean's King Lighting Science & Technology Co., Ltd. | Polyer containing thiophene-benzene-thiophene unit, preparation method therefor and solar cell device |
| CN103848961A (en) * | 2012-11-28 | 2014-06-11 | 海洋王照明科技股份有限公司 | Polymer containing thiophene-benzene-thiophene unit and preparation method thereof, and solar cell device |
| CN103833989A (en) * | 2012-11-28 | 2014-06-04 | 海洋王照明科技股份有限公司 | Polymer containing thiophene-benzene-thiophene unit and preparation method thereof, and solar cell device |
| CN108148073B (en) | 2016-12-06 | 2023-12-08 | 天光材料科技股份有限公司 | Organic semiconductor compound |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2006130717A2 (en) | 2005-06-02 | 2006-12-07 | The Regents Of The University Of California | Effective organic solar cells based on triplet materials |
-
2009
- 2009-03-27 US US12/412,651 patent/US8058387B2/en not_active Expired - Fee Related
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2006130717A2 (en) | 2005-06-02 | 2006-12-07 | The Regents Of The University Of California | Effective organic solar cells based on triplet materials |
Non-Patent Citations (8)
| Title |
|---|
| Chan et al., "Synthesis, Characterization, and Photovoltaic Properties of Novel Semiconducting Polymers with Thiophene-Phenylene-Thiophene (TPT) as Coplanar Units", Macromolecules 2008, 41, 5519-5526, American Chemical Society. |
| Forster et al., "Conjugated Ladder Polymers Containing Thienylene Units", Macromolecules, vol. 32, No. 9, Apr. 10, 1999, pp. 3159-3162, ACS, Washington, DC, US. |
| Hong et al. (Polymer Preprints 2000, 41(1), 189). * |
| Muhlbacher et al., High Photovoltaic Performance of a Low-Bandgap Polymer, Adv. Mater. 2006, vol. 18, 2884-2889, Wiley-VCH Verlag GmbH, Weinheim. |
| Second Examination Report and Citations issued by the Intellectual Property Office of the People's Repulic of China, on Dec. 14, 2010, for the counterpart application in the People's Republic of China (Appl. No. 200910001233.2). |
| Sirringhaus et al., High-resolution inkjet printing of all-polymer transistor circuits, Science, Dec. 15, 2000, vol. 290, 2123-2126. |
| Wong et al., "Syntheses and Structures of Novel Heteroarene-Fused Coplanar pi-Conjuated Chromophores", Organic Letters, vol. 8 , No. 22, Sep. 28, 2006, pp. 5033-5036, ACS, Washington, DC, US. |
| Wong et al., Syntheses and Structures of Novel Heteroarene-Fused Coplanar pi-Conjugated Chromophores, Organic Letters, 2006, vol. 8, No. 22, 5033-5036. |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20120142872A1 (en) * | 2009-05-27 | 2012-06-07 | Basf Se | Process for polymerising (hetero)aromatic compounds |
| US8835579B2 (en) * | 2009-05-27 | 2014-09-16 | Basf Se | Process for polymerising (hetero)aromatic compounds |
| US9428603B2 (en) | 2014-03-18 | 2016-08-30 | Council Of Scientific & Industrial Research | Polymeric dispersion of thiophene copolymers and a process for preparation thereof |
| US10991893B2 (en) | 2016-10-05 | 2021-04-27 | Raynergy Tek Incorporation | Organic semiconducting compounds |
Also Published As
| Publication number | Publication date |
|---|---|
| US20090299029A1 (en) | 2009-12-03 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP2075274B1 (en) | Soluble polythiophene derivatives | |
| US8058387B2 (en) | Soluble polythiophene derivatives | |
| US7754847B2 (en) | Soluble polythiophene derivatives | |
| Yuan et al. | Design of benzodithiophene-diketopyrrolopyrrole based donor–acceptor copolymers for efficient organic field effect transistors and polymer solar cells | |
| US8816035B2 (en) | Photoactive polymers | |
| US8871884B2 (en) | Copolymer semiconductors comprising thiazolothiazole or benzobisthiazole, or benzobisoxazole electron acceptor subunits, and electron donor subunits, and their uses in transistors and solar cells | |
| US8835598B2 (en) | Conjugated polymers and their use in optoelectronic devices | |
| US10263190B2 (en) | Difluorobithiophene-based donor-acceptor polymers for electronic and photonic applications | |
| US8748739B2 (en) | Conjugated polymers and their use in optoelectronic devices | |
| JP5179415B2 (en) | Soluble polythiophene derivatives | |
| KR101719379B1 (en) | Semiconductor materials prepared from dithienylvinylene copolymers | |
| US10516111B2 (en) | Polymer/fullerene formations and their use in electronic/photonic devices | |
| CN101492529B (en) | Soluble polythiophene derivative and application thereof in photoelectric component | |
| Patil et al. | Synthesis and photovoltaic properties of narrow band gap copolymers of dithieno [3, 2-b: 2′, 3′-d] thiophene and diketopyrrolopyrrole | |
| US7999062B2 (en) | Soluble polythiophene derivative | |
| Li et al. | Benzodifuran‐containing Well‐defined π‐conjugated Polymers for Photovoltaic Cells | |
| Jung et al. | A low band-gap copolymer composed of thienyl substituted anthracene and diketopyrrolopyrrole compatible with multiple electron acceptors for high efficiency polymer solar cells | |
| US20130035464A1 (en) | Semiconducting Compounds and Devices Incorporating Same | |
| Kim et al. | Synthesis and characterization of indeno [1, 2-b] fluorene-based low bandgap copolymers for photovoltaic cells | |
| WO2014031750A1 (en) | Acenaphthylene imide-derived semiconductors | |
| Cui et al. | Metallated conjugation in small-sized-molecular donors for solution-processed organic solar cells | |
| KR100907752B1 (en) | New fullerene derivative and organic solar cell device using same | |
| KR101553806B1 (en) | Organic semiconductor compounds Containing Posphine oxide and Solar Cell Device Using This Material | |
| US8907108B2 (en) | P-type organic semiconductor material and optoelectronic device utilizing the same | |
| KR20130085860A (en) | Organic electronic device |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AS | Assignment |
Owner name: INDUSTRIAL TECHNOLOGY RESEARCH INSTITUTE, TAIWAN Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNORS:CHAN, SHU-HUA;CHAO, TENG-CHIH;KO, BAO-TSAN;AND OTHERS;SIGNING DATES FROM 20090209 TO 20090216;REEL/FRAME:022462/0329 |
|
| ZAAA | Notice of allowance and fees due |
Free format text: ORIGINAL CODE: NOA |
|
| ZAAB | Notice of allowance mailed |
Free format text: ORIGINAL CODE: MN/=. |
|
| ZAAA | Notice of allowance and fees due |
Free format text: ORIGINAL CODE: NOA |
|
| STCF | Information on status: patent grant |
Free format text: PATENTED CASE |
|
| FPAY | Fee payment |
Year of fee payment: 4 |
|
| MAFP | Maintenance fee payment |
Free format text: PAYMENT OF MAINTENANCE FEE, 8TH YEAR, LARGE ENTITY (ORIGINAL EVENT CODE: M1552); ENTITY STATUS OF PATENT OWNER: LARGE ENTITY Year of fee payment: 8 |
|
| FEPP | Fee payment procedure |
Free format text: MAINTENANCE FEE REMINDER MAILED (ORIGINAL EVENT CODE: REM.); ENTITY STATUS OF PATENT OWNER: LARGE ENTITY |
|
| LAPS | Lapse for failure to pay maintenance fees |
Free format text: PATENT EXPIRED FOR FAILURE TO PAY MAINTENANCE FEES (ORIGINAL EVENT CODE: EXP.); ENTITY STATUS OF PATENT OWNER: LARGE ENTITY |
|
| STCH | Information on status: patent discontinuation |
Free format text: PATENT EXPIRED DUE TO NONPAYMENT OF MAINTENANCE FEES UNDER 37 CFR 1.362 |
|
| FP | Lapsed due to failure to pay maintenance fee |
Effective date: 20231115 |