BACKGROUND OF THE INVENTION
1. Field of the Invention
The present invention relates to novel cyclic amine compounds which have inhibitory effects on both cell adhesion and cell infiltration and are useful as anti-asthmatic agents, anti-allergic agents, anti-rheumatic agents, anti-arteriosclerotic agents, anti-inflammatory agents, anti-Sjogren's syndrome agents or the like, and medicines containing such compounds.
2. Description of the Background Art
In various inflammatory diseases, infiltration of leukocytes into inflammatory sites is observed. For example, infiltration of eosinophils into the bronchus in asthma (Ohkawara, Y. et al., Am. J. Respir. Cell Mol. Biol., 12, 4-12 (1995)), infiltration of macrophages and T lymphocytes into the aorta in arteriosclerosis (Sakai, A. et al., Arterioscler Thromb. Vasc. Biol., 17, 310-316 (1997)), infiltration of T lymphocytes and eosinophils into the skin in atopic dermatitis (Wakita, H. et al., J. Cutan. Pathol., 21, 33-39 (1994)) or contact dermatitis (Satoh, T. et al., Eur. J. Immunol., 27, 85-91 (1997)), and infiltration of various leukocytes into rheumatoid synovial tissue (Tak, P P. et al., Clin. Immunol. Immunopathol., 77, 236-242 (1995)), have been reported. Sjogren's syndrome in humans is an organ-specific autoimmune disease characterized by lymphocytic infiltration into the salivary and lacrimal glands, resulting in symptoms of dry mouth and dry eye due to insufficient secretion (Fox R I et al.: “Sjogren's syndrome: proposed criteria for classification” Arthritis Rheum., 29, 577-585(1986)).
Infiltration of these leukocytes is elicited by cytokines, chemokines, lipids, and complements produced in inflammatory sites (Albelda, S M. et al., FASEB J., , 504-512 (1994)). Activated leukocytes adhere to vascular endothelial cells through an interaction called rolling or tethering with endothelial cells activated likewise. Thereafter, the leukocytes transmigrate through endothelium to infiltrate into the inflammatory sites (Springer, T A., Annu. Rev. Physiol., 57, 827-872 (1995)). In adhesion of leukocytes to the vascular endothelial cells in this process, various cell adhesion molecules such as an immunoglobulin superfamily (ICAM-1, VCAM-1 and the like), a selectin family (E-selectin and the like), an integrin family (LFA-1, VLA-4 and the like) and CD44, which are induced on the surfaces of the cells by stimulation by cytokines or the like, play important roles (“Rinsho Meneki (Clinical Immune)”, 30, Supple. 18 (1998)), and a relationship between the disorder state and aberrant expression of the cell adhesion molecules is noted.
Accordingly, an agent capable of inhibiting cell adhesion or cell infiltration can be useful as an agent for preventing and treating allergic diseases such as bronchial asthma, dermatitis, rhinitis and conjunctivitis; autoimmune diseases such as rheumatoid arthritis, nephritis, Sjogren's syndrome, inflammatory bowel diseases, diabetes and arteriosclerosis; and chronic inflammatory diseases. In fact, it has been reported that antibodies against adhesion molecules or leukocytes such as LFA-1, Mac-1 and VLA-4 on antibodies against ICAM-1, VCAM-1, P-selectin, E-selectin and the like on vascular endothelial cells, which become ligands thereof, inhibit infiltration of leukocytes into inflammatory sites in animal models. For example, neutralizing antibodies against VCAM-1 and VLA-4, which is a counter receptor thereof, can delay development of diabetes in an NOD mouse model which spontaneously causes the diabetes (Michie, S A. et al., Curr. Top. Microbiol. Immunol., 231, 65-83 (1998)). It has also been reported that an antibody against VLA-4 or ICAM-1 and its counter receptor, LFA-1, inhibits infiltration of eosinophils in a guinea pig and mouse allergic conjunctivitis model (Ebihara et al., Current Eye Res., 19, 20-25 (1999); Whitcup, S M et al., Clin. Immunol., 93, 107-113 (1999)), and a monoclonal antibody against VCAM-1 inhibits infiltration of leukocytes in a mouse DSS-induced colitis model to attenuate colitis (Soriano, A. et al., Lab. Invest., 80, 1541-1551 (2000)). Further, an anti-VLA-4 antibody and an anti-CD44 antibody reduce the incidence of disease symptoms in a mouse collagen arthritis model (Zeidler, A. et al., Autoimmunity, 21, 245-252 (1995)). Even in cell adhesion molecule deficient-mice, inhibition of infiltration of leukocytes into inflammatory tissues is observed likewise in inflammatory models (Bendjelloul, F. et al., Clin. Exp. Immunol., 119, 57-63 (2000); Wolyniec, W W. et al., Am. J. Respir. Cell Mol. Biol., 18, 777-785 (1998); Bullard, D C. et al., J. Immunol., 157, 3153-3158 (1996)).
However, it is difficult to develop antibody-based drugs because they are polypeptides and so oral administration is a problem. Moreover, the possible side effects due to antigenicity and allergic reactions are problems.
On the other hand, there have been various investigations of low-molecular weight compounds having an inhibitory effect on cell adhesion with a view toward permitting oral administration. These compounds include benzothiophene derivatives (Boschelli, D H. et al., J. Med. Chem., 38, 4597-4614 (1995)), naphthalene derivatives (Japanese Patent Application Laid-Open No. 10-147568), hydroxybenzoic acid derivatives (Japanese Patent Application Laid-Open No. 10-182550), lignans (Japanese Patent Application Laid-Open No. 10-67656), 2-substituted benzothiazole derivatives (Japanese Patent Application Laid-Open No. 2000-086641 through PCT route), condensed pyrazine compounds (Japanese Patent Application Laid-Open No. 2000-319277 through PCT route), 2,6-dialkyl-4-silylphenol (Japanese Patent Application Laid-Open No.2000-509070 through PCT route) and the like. However, the goal has not often been sufficiently achieved under the circumstances. Cyclic diamine compounds described in Japanese Patent Application Laid-Open Nos. 9-143075 and 11-92382 and Japanese Patent Application No.2000-271220 do not exhibit a sufficient inhibitory effect on cell adhesion, and so there is a demand for further improvement in activity.
SUMMARY OF THE INVENTION
An object of the present invention is to provide a substance having inhibitory effects on both cell adhesion and cell infiltration, plus excellent anti-asthmatic effects, anti-allergic effects, anti-rheumatic effects, anti-arteriosclerotic effects, anti-inflammatory effects and anti-Sjogren's syndrome effect.
With the foregoing circumstances in mind, the present inventors carried out an extensive investigation to find a substance which inhibits cell adhesion and cell infiltration. As a result, we found that compounds represented by the general formula (1) having phenyl-pyridyl or biphenyl groups at both ends of the cyclic amine, have excellent cell adhesion-inhibiting effects and cell infiltration-inhibiting effects and are useful as anti-allergic agents, anti-asthmatic agents, anti-rheumatic agents, anti-arteriosclerotic agents or anti-inflammatory agents or anti-Sjogren's syndrome agents.
The present invention provides a cyclic amine compound represented by the following general formula (1):
wherein,
-
- R1, R2 and R3 each independently represent a hydrogen atom, a halogen atom, or hydroxy, alkyl, halogen-substituted alkyl, alkoxy, alkylthio, carboxyl, alkoxycarbonyl or alkanoyl group;
- W1 and W2 each independently represent N or CH;
- X represents O, NR4, CONR4 or NR4CO;
- R4 represents a hydrogen atom, or an alkyl, alkenyl, alkynyl, substituted or unsubstituted aryl, substituted or unsubstituted heteroaryl, substituted or unsubstituted aralkyl, or substituted or unsubstituted heteroaralkyl group; and
- l, m and n each represents a number of 0 or 1.
According to the present invention, there is also provided a medicine comprising the above cyclic amine compound, a salt thereof, or a hydrate thereof as an active ingredient.
According to the present invention, there is further provided a pharmaceutical composition comprising the above cyclic amine compound, the salt thereof, or the hydrate thereof and a pharmaceutically acceptable carrier.
According to the present invention, there is still further provided a method for treating a disease caused by cell adhesion and/or cell infiltration, which comprises administering an effective amount of the above cyclic amine compound, a salt thereof, or a hydrate thereof to a patient who requires such treatment.
DETAILED DESCRIPTION OF THE PREFERRED EMBODIMENTS
The compound of the present invention is characterized in that the cyclic amine has two phenyl-pyridyl or biphenyl groups, or phenyl-pyridyl and biphenyl groups. It has not been known at all that compounds having such structure have both of excellent cell adhesion-inhibiting effects and cell infiltration-inhibiting effects.
In the general formula (1), the halogen atoms for R1, R2 and R3 include fluorine, chlorine, bromine and iodine atoms.
The alkyl group for R1, R2, R3 and R4 typically includes straight, branched or cyclic C1-C8 alkyl groups, such as straight or branched C1-C8 alkyl groups, for example, methyl, ethyl, propyl, butyl, pentyl, hexyl, heptyl and octyl groups, and C3-C8 cycloalkyl groups, for example, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cyclohexylmethyl and cyclohexylethyl groups. Among them, particularly preferred are C1-C6 alkyl groups such as methyl, ethyl, n-propyl, isopropyl, n-butyl and the like.
The halogen-substituted alkyl group for R1, R2 and R3 typically includes C1-C8 alkyl groups substituted with 1 to 3 halogen atoms. Among them, particularly preferred are C1-C6 alkyl groups substituted with 1 to 3 halogen atoms, such as trifluoromethyl, 2,2,2-trifluoroethyl, etc.
The alkoxy group typically includes straight, branched or cyclic C1-C8 alkoxy groups, such as straight or branched C1-C8 alkoxy groups, for example, methoxy, ethoxy, propoxy, butoxy, pentyloxy and hexyloxy groups; and C3-C8 cycloalkyloxy groups, for example, cyclopropyloxy, cyclobutyloxy, cyclopentyloxy, cyclohexyloxyl, cyclohexylmethyloxy and cyclohexylethyloxy groups. Among them, particularly preferred are C1-C6 alkoxy groups such as methoxy, ethoxy, n-propoxy, isopropoxy and n-butyloxy groups.
The alkylthio group typically includes C1-C8 alkylthio groups, and is preferably a C1-C6 alkylthio group such as, for example, methylthio, ethylthio, n-propylthio, isopropylthio or the like.
The alkoxycarbonyl group typically includes C1-C6 alkoxycarbonyl groups, and is preferably a C1-C6 alkoxycarbonyl group such as methoxycarbonyl, ethoxycarbonyl, tert-butoxycarbonyl or the like.
The alkanoyl group typically includes C1-C6 alkanoyl groups and is preferably a C1-C4 alkanoyl group such as acetyl, propionyl, butyryl, isobutyryl or the like.
The alkenyl group for R4 typically includes C3-C8 alkenyl groups and is preferably a C3-C6 alkenyl group such as 2-propenyl, 3-butenyl or the like. The alkynyl group typically includes C3-C8 alkynyl groups and is preferably a C3-C6 alkynyl group such as 2-propynyl, 3-butynyl or the like.
The aryl group for R4 typically includes C6-C14 aryl groups and is preferably phenyl, naphthyl, anthryl, indenyl, indanyl, 5,6,7,8-tetrahydronaphthyl or the like. The heteroaryl group for R4 typically includes heteroaryl groups of 5- or 6-membered ring containing 1 to 4 nitrogen atoms in the ring, and is preferably imidazolyl, pyridyl, pyrimidinyl or the like. The aralkyl group typically includes C6-C14 aryl-C1-C6 alkyl groups such as phenyl C1-C6 alkyl groups and naphthyl C1-C6 alkyl groups, for example, benzyl, naphthylmethyl, phenylethyl, phenylpropyl, etc. The heteroaralkyl group typically includes 5- or 6-membered ring heteroaryl containing 1 to 4 nitrogen atoms-C1-C6 alkyl groups, such as imidazolyl-C1-C6 alkyl, pyridyl-C1-C6 alkyl, pyrimidinyl-C1-C6 alkyl, etc.
The groups which can substitute the above-mentioned aryl, heteroaryl, aralkyl and heteroaralkyl include 1 to 3 groups or atoms selected from alkyl, alkoxy, halogen-substituted alkoxy, alkylthio, alkylsulfinyl, alkylsulfonyl, halogen, nitro, amino, acetylamino, trifluoromethyl and alkylenedioxy, wherein said alkyl, alkoxy and alkylthio include those illustrated for R1˜R3. The alkyl group of the alkylsulfinyl group and alkylsulfonyl group typically includes C1-C3 alkyl groups such as methyl, ethyl, n-propyl and isopropyl groups. The halogen-substituted alkoxy includes C1-C8 alkoxy groups substituted by 1 to 3 halogen atoms, and is preferably a C1-C6 alkoxy group substituted by 1 to 3 halogen atoms such as trifluoromethoxy or 2,2,2-trifluoroethoxy. The alkylenedioxy group typically includes C1-C3 alkylenedioxy groups such as methylenedioxy, ethylenedioxy and propylenedioxy groups.
Preferably, X represents NR4. More preferably, X represents NR4 and R4 represents a substituted or unsubstituted C6-C14 aryl group or a substituted or unsubstituted 5- or 6-membered ring heteroaryl group containing 1 to 4 nitrogen atoms in the ring. The compounds of the formula (1) wherein X represents NR4 have particularly strong cell adhesion-inhibiting action as shown later in Test Example 1.
Preferably, R1, R2 and R3 are attached to the phenyl group at the 3, 4 and 5-positions thereof. In this case, it is more preferable that R1 and R3 (at the 3- and 5-positions of the phenyl ring) are an alkoxy group or a halogen. It is also preferable that R2 (at the 4-position of the phenyl ring) is a hydrogen atom, a halogen atom, or a hydroxy, alkyl, halogen-substituted alkyl, alkoxy, alkylthio, carboxy, alkoxycarbonyl or alkanoyl group. l denotes 0 or 1, and is preferably 1.
Preferably, W1 represents N.
Preferably, W2 represents N.
Most preferable compounds include the compounds of the formula (1), wherein X represents NR4, and R4 represents a substituted or unsubstituted C6-C14 aryl group or a substituted or unsubstituted 5- or 6-membered ring heteroaryl group containing 1 to 4 nitrogen atoms in the ring. Particularly preferably, R4 represents a phenyl or pyridyl group which may be substituted with one or two groups or atoms selected from halogen, alkyl, alkoxy, alkylthio, trifluoromethyl and alkylenedioxy.
No particular limitation is imposed on the salts of the compounds (1) according to the invention as long as they are pharmaceutically acceptable salts. Examples include the acid-addition salts of mineral acids, such as hydrochlorides, hydrobromides, hydroiodides, sulfates and phosphates; and acid-addition salts of organic acids, such as benzoates, methanesulfonates, ethanesulfonates, benzenesulfonates, p-toluenesulfonates, oxalates, maleates, fumarates, tartrates, citrates and acetates.
The compounds of formula (1) may be present in the form of solvates typified by hydrates, and the solvates are embraced in the present invention.
The compounds of formula (1) can be prepared in accordance with the following processes A˜L:
- Process A: Preparation of the compound of the formula (1) wherein l=1, m=0, n=1 and X=CONR4
- wherein, W1, W2, R1, R2, R3 and R4 are as defined above, W3 has the same meaning as W1 or W2, and B denotes a leaving group such as a halogen atom, or methanesulfonyloxy or p-toluenesulfonyloxy group.
Compound (2) and an N-(2-nitro)benzenesulfonylamine derivative (3) are reacted to give compound (4). The resulting compound (4) is treated with thiophenol in the presence of a base such as potassium carbonate to eliminate the 2-nitrobenzenesulfonyl group, thereby giving amine compound (5). Alternatively, when R4 is H, it is possible to react compound (2) with potassium phthalimide and then treat the resulting phthalimide derivative (6) with hydrazine to give the corresponding amine compound (5).
On the other hand, compound (2) is reacted with ethyl isonipecotate (7) in a solvent such as acetonitrile, N,N-dimethylformamide (DMF), dimethyl sulfoxide (DMSO), tetrahydrofuran (THF), dioxane, toluene, benzene, etc. in the presence of a base such as potassium carbonate or the like at a temperature between 0° C. and a reflux temperature for several hours to several days, preferably at room temperature overnight, to give compound (8). The compound (8) is subjected to a usual alkaline hydrolysis to give the corresponding carboxylic acid compound (9).
The carboxylic acid compound (9) is reacted with the amine compound (5) using a dehydration condensing agent such as 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide hydrochloride (water-soluble carbodiimide), 2-(1H-benzotriazol-1-yl)-1,1,3,3-tetramethyluronium hexafluorophosphate (HBTU) or the like in a solvent such as chloroform, dichloroethane, THF, dioxane, acetonitrile, etc. at a temperature between 0° C. and a reflux temperature for several hours to several days, preferably at room temperature for 12 hours, to give an end product (1A).
- Process B: Preparation of the compound of the formula (1) wherein l=1, m=0, n=1 and X=O
- wherein, B, W1, W2, R1, R2 and R3 are as defined above, and J denotes a protecting group such as benzyloxycarbonyl, tert-butoxycarbonyl, acetyl, benzoyl or benzyl group. Incidentally, in the reaction schemes shown above and below, the expression “(W2→W1)” following the term “compound(2)” means that W2 in the formula representing compound (2) is changed to W1.
4-hydroxypiperidine compound (10) with a protected amino group is reacted with compound (2) in the presence of sodium hydride or potassium iodide in a solvent such as DMF, DMSO, etc. at a temperature between 0° C. and a reflux temperature for several hours to several days, preferably at room temperature for 2 days, to give compound (11). The protecting group in the compound (11) is removed in a known manner. The resulting compound (12) is reacted with compound (2) in the presence of a base such as potassium carbonate in a solvent such as acetonitrile, DMF, DMSO, THF, dioxane, etc. at a temperature between 0° C. and a reflux temperature for several hours to several days, preferably at room temperature for 4 hours, to give an end product (1B).
- Process C: Preparation of the compound of the formula (1) wherein l=1, m=0, n=0, X=NR4CO and R4=H or Me
- wherein, B, W1, W2,, R1, R2 and R3 are as defined above, and R4denotes a hydrogen atom or methyl group.
Isonipecotamide (13) is reacted with compound (2) in the presence of a base such as potassium carbonate, sodium carbonate or the like in a solvent such as acetonitrile, DMF, DMSO, THF, dioxane, etc. at a temperature between 0° C. and a reflux temperature for several hours to several days, preferably at room temperature for 4 hours, to give compound (14). The compound (14) is subjected to Hofmann rearrangement reaction to give amine compound (15).
On the other hand, by subjecting the compound (14) to Hofmann rearrangement reaction in ethanol, carbamate compound (16) is obtained. Then, by subjecting the compound (16) to a reduction reaction using lithium aluminum hydride, methylamine compound (17) is obtained.
By reacting carboxylic acid compound (18) with the amine compound (15) or methylamine compound (17) similarly to the condensation reaction in Process A, an end compound (1C) is obtained.
- Process D: Preparation of the compound of the formula (1) wherein l=1, m=0, n=1 and X=NR4
- wherein, B, W1, W2, R1, R2 and R3 are as defined above, and R4 denotes an alkyl, alkenyl, alkynyl, aralkyl or heteroaralkyl group.
The amine compound (15) mentioned in the above is reacted with 2-nitrobenzenesulfonyl chloride (19) according to a known manner to give compound (20). The compound (20) is reacted with compound (2) in the presence of a base such as potassium carbonate in a solvent such as acetonitrile, DMF, DMSO, THF, dioxane or the like at a temperature between 0° C. and a reflux temperature for several hours to several days, preferably at room temperature for 4 hours, to give compound (21). The benzenesulfonyl group of the compound (21) is removed similarly to the procedure for the compound (4) in Process A to give an end compound (1D) (R4=H). The compound (1D) is reacted with R4—B in the presence of a base such as sodium carbonate, sodium bicarbonate, potassium carbonate, cesium carbonate or the like in a solvent such as acetonitrile, THF, dioxane, chloroform, dichloromethane, DMF, DMSO or the like at a temperature between 0° C. and a reflux temperature for several hours to several days, preferably at 80° C. for 12 hours, to give compound (1D′).
On the other hand, the methylamine compound (17) is reacted compound (2) in the presence of a base such as potassium carbonate in a solvent such as acetonitrile, DMF, DMSO, THF, dioxane or the like at a temperature between 0° C. and a reflux temperature for several hours to several days, preferably at room temperature for 4 hours, to give an end compound (1D″) (R4=Me).
- Process E: Preparation of the compound of the formula (1) wherein l=1, m=0 or 1, n=1 and X=NR4,
- wherein, B, J, W1, W2, R1, R2 and R3 are as defined above, and R4 denotes an alkyl, alkenyl, alkynyl, aralkyl or heteroaralkyl group.
Aminopiperidine derivative (22) in which the amino group on the ring is protected is reacted with compound (2) in the presence of a base such as potassium carbonate in a solvent such as acetonitrile, DMF, DMSO, THF, dioxane or the like at a temperature between 0° C. and a reflux temperature for several hours to several days, preferably at room temperature for 4 hours, to give compound (23). The compound (23) is reacted with R4—B in the presence of a base such as sodium carbonate, sodium bicarbonate, potassium carbonate, cesium carbonate or the like in a solvent such as acetonitrile, THF, dioxane, chloroform, dichloroethane, DMF, DMSO or the like at a temperature between 0° C. and a reflux temperature for several hours to several days, preferably at 80° C. for 12 hours, to give compound (24). After removal of the protecting group, the compound (25) is reacted compound (2) in the presence of a base such as potassium carbonate in a solvent such as acetonitrile, DMF, DMSO, THF, dioxane or the like at a temperature between 0° C. and a reflux temperature for several hours to several days, preferably at room temperature for 4 hours, to give compound (1E).
- Process F: Preparation of the compound of the formula (1) wherein l=1, m=0, n=1 and X=NR4,
- wherein, B, W1, W2, R1, R2 and R3 are as defined above, and R4 denotes an alkyl, alkenyl, alkynyl, aralkyl, heteroaralkyl, aryl or heteroaryl group.
4-piperidone ethylene ketal (26) is reacted with compound (2) in the presence of a base such as potassium carbonate in a solvent such as acetonitrile, DMF, DMSO, THF, dioxane, etc. at a temperature between 0° C. and a reflux temperature for several hours to several days, preferably at room temperature for 4 hours, to give compound (27), which in turn is deketalized by using an acid to give ketone compound (28).
On the other hand, 4-piperidone (29) is reacted compound (2) in the presence of a base such as potassium carbonate in a solvent such as acetonitrile, DMF, DMSO, THF, dioxane or the like at a temperature between 0° C. and a reflux temperature for several hours to several days, preferably at room temperature for 4 hours, to give compound (28). Using the compound (28), amine compound (30) can be prepared according to either of the following two synthesis processes:
- Synthesis process 1: The compound (28) is reacted with an amine compound of the formula: R4—NH2 in the presence of molecular sieves in toluene or benzene at a temperature between 0° C. and a reflux temperature for several hours to several days, preferably at reflux temperature for 12 hours, followed by reaction with a reducing agent such as sodium borohydride or sodium cyanoborohydride at a temperature between 0° C. and a reflux temperature for several minutes to several days, preferably at room temperature for 1 hour, to give the amine compound (30).
- Synthesis process 2: The compound (28) is reacted with an amine compound of the formula: R4—NH2 in the presence of a reducing agent such as sodium triacetoxy boron hydride in a solvent such as dichloromethane, 1,2-dichloroethane, methanol, ethanol, etc. at a temperature between 0° C. and a reflux temperature for several minutes to several days, preferably at room temperature for 4 hours, to give the amine compound (30).
The resulting compound (30) is reacted compound (2) in a solvent such as acetonitrile, DMF, DMSO, THF, dioxane, etc. at a temperature between 0° C. and a reflux temperature for several hours to several days, preferably at room temperature for 4 hours, to give an end product (1F).
- Process G: Preparation of the compound of the formula (1) wherein l=1, m=0, n=1 and X=NR4
- wherein, B, J, W1, W2, R1, R2 and R3 are as defined above, and R4 denotes an alkyl, alkenyl, alkynyl, aralkyl, heteroaralkyl, aryl or heteroaryl group.
4-piperidone derivative (31) in which the amino group on the ring is protected is reacted with an amine compound R4—NH2 similarly to the procedure for preparation of compound (30) in Process F to give compound (32). The compound (32) is reacted with compound (2) in the presence of a base such as potassium carbonate in a solvent such as acetonitrile, DMF, DMSO, THF, dioxane, etc. at a temperature between 0° C. and a reflux temperature for several hours to several days, preferably at room temperature for 4 hours, to give compound (33). After removal of the protecting group from the compound (33), the resulting compound (34) is reacted with compound (2) in the presence of a base such as potassium carbonate in a solvent such as acetonitrile, DMF, DMSO, THF, dioxane, etc. at a temperature between 0° C. and a reflux temperature for several hours to several days, preferably at room temperature for 4 hours, to give an end product (1G).
- Process H: Preparation of the compound of the formula (1) wherein l=0, m=0, n=1 and X=NH
- wherein, B, J, W1, W2, R1, R2 and R3 are as defined above.
3-aminopyrrolidine derivative (35) with a protected amino group on the ring is reacted with 2-nitrobenzenesulfonyl chloride (19) under usual conditions to give a benzenesulfonyl derivative (36). The derivative (36) is reacted with compound (2) in the presence of a base such as potassium carbonate in a solvent such as acetonitrile, DMF, DMSO, THF, dioxane, etc. at a temperature between 0° C. and a reflux temperature for several hours to several days, preferably at room temperature for 4 hours, to give compound (37). The protecting group of the amino group is removed from the compound (37) to give compound (38), which in turn is reacted with compound (2) in the presence of a base such as potassium carbonate in a solvent such as acetonitrile, DMF, DMSO, THF, dioxane, etc. at a temperature between 0° C. and a reflux temperature for several hours to several days, preferably at room temperature for 4 hours, to give compound (39). By subjecting the compound (39) to a reaction similar to that in the preparation of compound (5) in Process A, an end product (1H) is obtained.
- Process I: Preparation of the compound of the formula (1) wherein l=0, m=0, n=1 and X=NR4
- wherein, B, J, W1, W2, R1, R2 and R3 are as defined above, and R4 denotes an alkyl, alkenyl, alkynyl or aralkyl group.
Compound (36) is reacted with R4—B in the presence of a base such as sodium carbonate, potassium carbonate, etc. in a solvent such as acetonitrile, THF, dioxane, chloroform, dichloroethane, DMF, DMSO, etc. at a temperature between 0° C. and a reflux temperature for several hours to several days, preferably at 80° C. for 12 hours, to give compound (40). The amino-protecting group is removed from the compound (40), and the resulting compound (41) is reacted with compound (2) in the presence of a base such as potassium carbonate in a solvent such as acetonitrile, DMF, DMSO, THF, dioxane, etc. at a temperature between 0° C. and a reflux temperature for several hours to several days, preferably at room temperature for 4 hours, to give compound (42). By subjecting the compound (42) to a reaction similar to that in the preparation of compound (5) in Process A, compound (43) is obtained. The compound (43) is reacted with compound (2) in the presence of a base such as potassium carbonate in a solvent such as acetonitrile, DMF, DMSO, THF, dioxane, etc. at a temperature between 0° C. and a reflux temperature for several hours to several days, preferably at room temperature for 4 hours, to give an end product (1I).
- Process J: Preparation of the compound of the formula (1) wherein R2=OH
- wherein, X, W1, W2, R1, R3, l, m and n have the same meanings as initially defined.
By reacting methoxy compound (1J) with iodotrimethylsilane in a solvent such as toluene, chloroform, dichloromethane, etc. at a temperature between −25° C. and a reflux temperature for several minutes to several days, preferably at 0° C. for 2 hours, there can be obtained an end product (1J′).
- Process K: Preparation of the compound of the formula (1) wherein l=1, m=0, n=0 and X=NR4CO
- wherein, B, J, W1, W2, R1, R2 and R3 are as defined above, and R4 denotes an alkyl, alkenyl, alkynyl, aralkyl, heteroaralkyl, aryl or heteroaryl group.
Compound (32), which is described in the Process G, is reacted with compound (18) in the similar procedure as described in the preparation of compound (1A) in Process A to give compound (44). After removal of the protecting group from the compound (44), the resulting compound (45) is reacted with compound (2) in the presence of a base such as potassium carbonate in a solvent such as acetonitrile, DMF, DMSO, THF, dioxane, etc. at a temperature between 0° C. and a reflux temperature for several hours to several days, preferably at room temperature for 4 hours, to give an end product (1K).
- Process L: Preparation of the compound of the formula (1) wherein l=1, m=O, n=1 and X=alkylsulfonylphenylamino group
- wherein, B, W1, W2, R1, R2 and R3 are as defined above.
Compound (34), which was prepared in the Process G (wherein X denotes alkylthiophenylamino group), is reacted with an oxdation agent such as 3-chloroperbenzoic acid, peracetic acid, hydrogen peroxide, etc. in the known manner to give an alkylsulfonyl derivative (46). Compound (46) is then reacted with compound (2) in the presence of a base such as potassium carbonate in a solvent such as acetonitrile, DMF, DMSO, THF, dioxane, etc. at a temperature between 0° C. and a reflux temperature for several hours to several days, preferably at 70° C. overnight, to give an end product (1L).
The compounds (1) according to the present invention are obtained by any of the above-described processes and may further be purified by using an ordinary purification means such as recrystallization or column chromatography as needed. As needed, the compounds may also be converted into the desired salts or solvates in a method known per se in the art. When the compounds (1) have an asymmetric carbon atom, the present invention includes any configurational isomers.
The compounds (1) according to the present invention, or salts or solvates thereof thus obtained have an excellent inhibitory effect on cell adhesion as demonstrated in the examples, which will be described subsequently, and are useful as medicines for treatment and prevention of diseases of animals including humans, caused by cell adhesion or cell infiltration, for example, asthma, allergy, rheumatism, arteriosclerosis, inflammation, Sjogren's syndrome, etc.
The medicine according to the present invention comprises a compound (1), a salt thereof, or a solvate thereof as an active ingredient. The form of administration may be suitably selected as necessary for the therapeutic application intended without any particular limitation, including oral preparations, injections, suppositories, ointments, inhalants, eye drops, nose drops and plasters. A composition suitable for use in these administration forms can be prepared by blending a pharmaceutically acceptable carrier in accordance with the conventional preparation method publicly known by those skilled in the art.
When an oral solid preparation is formulated, an excipient, and optionally, a binder, disintegrator, lubricant, colorant, a taste corrigent, a smell corrigent and the like are added to compound (1) and the resulting composition can be formulated into tablets, coated tablets, granules, powders, capsules, etc. in accordance with methods known in the art.
As such additives described above, any additives may be used which are generally used in the pharmaceutical field. Examples include excipients such as lactose, sucrose, sodium chloride, glucose, starch, calcium carbonate, kaolin, microcrystalline cellulose and silicic acid; binders such as water, ethanol, propanol, simple syrup, glucose solution, starch solution, gelatin solution, carboxymethyl cellulose, hydroxypropyl cellulose, hydroxypropyl starch, methyl cellulose, ethyl cellulose, shellac, calcium phosphate and polyvinyl pyrrolidone; disintegrators such as dry starch, sodium alginate, agar powder, sodium hydrogencarbonate, calcium carbonate, sodium lauryl sulfate, monoglyceryl stearate and lactose; lubricants such as purified talc, stearic acid salts, borax and polyethylene glycol; and taste corrigents such as sucrose, orange peel, citric acid and tartaric acid.
When an oral liquid preparation is formulated, a taste corrigent, buffer, stabilizer, smell corrigent and/or the like are added to compound (1) and the resulting composition can be formulated into internal liquid preparations, syrup preparations, elixirs, etc. in accordance with methods known in the art. In this case, vanillin as the taste corrigent, may be used. As the buffer, sodium citrate may be mentioned. As examples of the stabilizer, tragacanth, gum arabic and gelatin may be mentioned.
When an injection is formulated, a pH adjustor, buffer, stabilizer, isotonicity agent, local anesthetic and the like may be added to compound (1) according to the present invention, and the resultant composition can be formulated into subcutaneous, intramuscular and intravenous injections in accordance with methods known in the art. Examples of the pH adjustor and buffer in this case include sodium citrate, sodium acetate and sodium phosphate. Examples of the stabilizer include sodium pyrosulfite, EDTA, thioglycolic acid and thiolactic acid. Examples of the local anesthetic include procaine hydrochloride and lidocaine hydrochloride. Examples of the isotonicity agent include sodium chloride and glucose.
When a suppository is formulated, a carrier preparation known in the art, for example, polyethylene glycol, lanoline, cacao butter, fatty acid triglyceride or the like, and optionally, a surfactant such as Tween (trade mark) and the like are added to the compound (1), and the resultant composition can be formulated into suppositories in accordance with methods known in the art.
When an ointment is formulated, a base material, stabilizer, wetting agent, preservative and the like, which are generally used, are blended with compound (1) as needed, and the resulting blend is mixed and formulated into ointments in accordance with known method known methods. Examples of the base material include liquid paraffin, white vaseline, bleached beeswax, octyldodecyl alcohol and paraffin. Examples of the preservative include methyl p-hydroxybenzoate, ethyl p-hydroxybenzoate and propyl p-hydroxybenzoate.
Besides the above preparations, inhalants, eye drops and nose drops may also be formulated in accordance with known methods.
The dose of the medicine according to the present invention varies according to the age, weight and condition of the patient to be treated, the administration method, the number of times of administration, and the like. It is however preferred that the medicine is generally orally or parenterally administered at once or in several portions in a dose of 1 to 1,000 mg per day in terms of compound (1), for an adult.
The present invention will hereinafter be described in more detail by Examples. However, the present invention is not limited to these examples.
PREPARATION EXAMPLE 1
Synthesis of ethyl 2-(3,4,5-trimethoxyphenyl)isonicotinate
3,4,5-Trimethoxyphenylboronic acid (20.10 g) and ethyl 2-chloroisonicotinate (18.56 g) were suspended in a mixted solvent of toluene (200 mL) and THF(100 mL), and to the suspension 2 M sodium carbonate (200 mL) and tetrakis(triphenyl phosphine) palladium(0) (5.78 g) were added. The mixture was stirred at 90° C. overnight under an argon atmosphere. Ethyl acetate was added to the reaction mixture to separate an organic layer. The organic layer was washed with brine, dried over anhydrous sodium magnesium and concentrated under reduced pressure. The residue was purified by column chromatography on silica gel using hexane-ethyl acetate (5:1) to give the title compound.
Yield: 27.99 g (88%). 1H-NMR (400 MHz, CDCl3) δ: 1.45 (t, 3H, J=7.0 Hz), 3.92 (s, 3H), 3.99 (s, 6H), 4.46 (q, 2H, J=7.0 Hz), 7.30 (s, 2H), 7.76 (dd, 1H, J=5.1 Hz, 1.6 Hz), 8.24 (dd, 1H, J=1.6 Hz, 0.8 Hz), 8.81 (dd, 1H, J=5.1 Hz, 0.8 Hz).
PREPARATION EXAMPLE 2
Synthesis of 4-hydroxymethyl-2-(3,4,5-trimethoxyphenyl)pyridine
Ethyl 2-(3,4,5-trimethoxyphenyl)isonicotinate (24.57 g) was dissolved in dry THF (200 mL), and to the solution lithium aluminum hydride (2.94 g) was added at 0° C. under an argon atmosphere. The mixture was stirred at 0° C. for 1 hour as it is. A small amount of water and then sodium sulfate were added to the reaction mixture, and the reaction mixture was filtered through celite. The filtrate was evaporated, and the reultant crude crystals were recrystalized from ethyl acetate-hexane to give the title compound.
Yield: 17.53 g (82%). 1H-NMR (400 MHz, CDCl3) δ: 3.90 (s, 3H), 3.95 (s, 6H), 4.79 (s, 2H), 7.19 (d, 1H, J=5.1 Hz), 7.21 (s, 2H), 7.66 (s, 1H), 8.60 (d, 1H, J=5.1 Hz).
PREPARATION EXAMPLE 3
Synthesis of 4-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine
4-hydroxymethyl-2-(3,4,5-trimethoxyphenyl)pyridine(19.18 g) was dissolved in chloroform (100 mL), and to the solution thinly chloride (10.2 mL) was added at 0° C. After 30 minutes, the mixture was warmed to room temperature and stirred for 4 hours. The reaction mixture was washed with aqaueous saturated sodium hydrogendcarbonate and brine, dried over anhydrous sodium sulfate and evaporated. The residue was then recrystallized from ethyl acetate-hexane to give the title compound as pale yellow crystalline powder.
Yield: 18.24 g (89%). 1H-NMR (400 MHz, CDCl3) δ: 3.91 (s, 3H), 3.97 (s, 6H), 4.61 (s, 2H), 7.24 (s, 2H), 7.26 (d, 1H, J=5.1 Hz), 7.68 (s, 1H), 8.67 (d, 1H, J=5.1 Hz).
PREPARATION EXAMPLE 4
Synthesis of N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]phthalimide
To a solution of 4-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (881 mg) in chloroform (10 mL) was added potassium phthalimide (556 mg). The mixture was stirred at room temperature overnight and water was added. After separating the organic layer, the aqueous layer was extracted with chloroform. Organic layers were combined, dried over anhydrous magnesium sulfate and evaporated to give the title compound as white powder.
Yield: 1.16 g (96%).
PREPARATION EXAMPLE 5
Synthesis of 4-aminomethyl-2-(3,4,5-trimethoxyphenyl)pyridine
To a suspension of N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]phthalimide (1.16 g) in ethanol (30 mL) was added hydrazine monohydrate (1 mL). The mixture was refluxed for 3 hours. After cooling, the precipitates were filtered off. The filtrate was evaporated and the residue was dissolved in chloroform. The solution was washed with saturated aqueous sodium hydrogen carbonate and brine, dried over anhydrous magnesium sulfate and evaporated to give the title compound as pale yellow oil.
Yield: 418 mg (53%).
PREPARATION EXAMPLE 6
Synthesis of ethyl 1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine-4-carboxylate
To a solution of ethyl piperidine-4-carboxylate (514 mg) and 4-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (969 mg) in acetonitrile (20 mL) was added potassium carbonate (452 mg). The mixture was stirred at room temperature for 4 hours and evaporated. The residual oil was subjected to a column of silica gel and eluted using hexane-ethyl acetate (2:1) and then chloroform-methanol (40:1). Fractions containing the product were collected and evaporated to give the title compound as white prisms.
Yield: 1.20 g (88%). 1H-NMR (400 MHz, CDCl3) δ: 1.25 (t, 3H, J=7.0 Hz), 1.72-1.93 (m, 4H), 2.10 (t, 2H, J=9.8 Hz), 2.27-2.35 (m, 1H), 2.86 (d, 2H, J=11.3 Hz), 3.55 (s, 2H), 3.91 (s, 3H), 3.98 (s, 6H), 4.14 (q, 2H, J=7.0 Hz), 7.21 (d, 1H, J=4.9 Hz), 7.24 (s, 2H), 7.63 (s, 1H), 8.59 (d, 1H, J=5.1 Hz).
PREPARATION EXAMPLE 7
Synthesis of 1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine-4-carboxylic acid
To a solution of ethyl 1-[[2-(3,4,5-trimethoxyphenyl)prydine-4-yl]methyl]piperidine-4-carboxylate (760 mg) in ethanol (10 mL) was added 1 M sodium hydroxide (10 mL). The mixture was stirred at room temperature for 4 hours and evaporated. The residue was dissolved in water (20 mL) and 5% aqueous potassium hydrogen sulfate was added dropwise until pH of the solution became 7. Precipitates were collected and the product was used for the next steps without further purification.
Yield: 779 mg (theoretical amount).
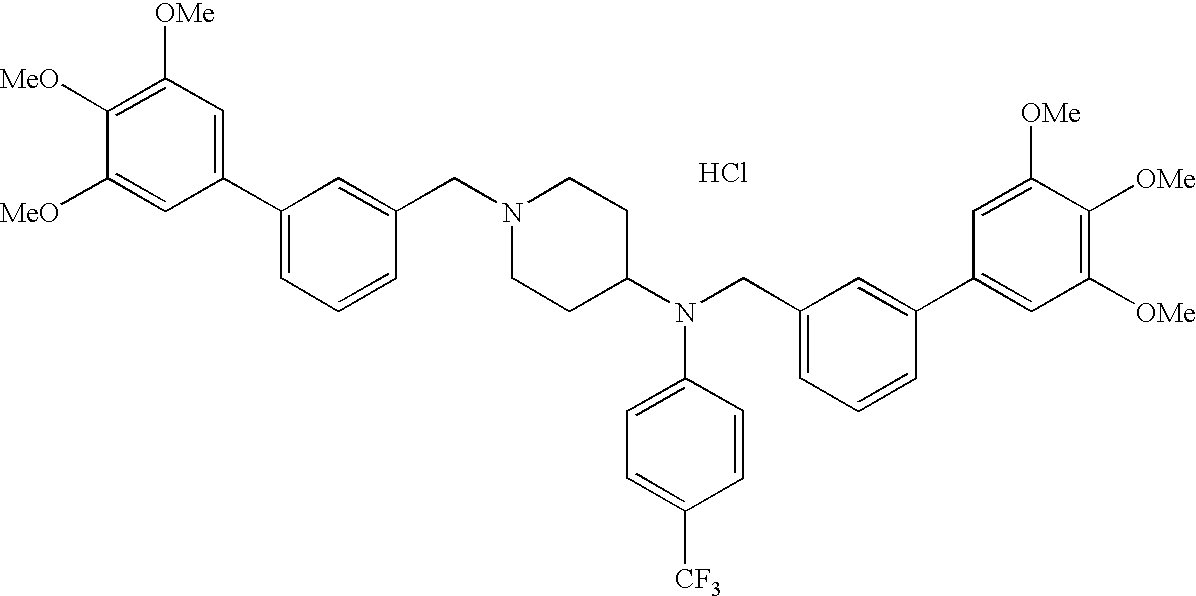
EXAMPLE 1
Synthesis of 1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]-4-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methylaminocarbonyl]piperidine maleate
To a solution of 1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine-4-caroxylic acid (97 mg) and 4-aminomethyl-2-(3,4,5-trimethoxyphenyl) pyridine (68 mg) in acetonitrile (5 mL) was added HBTU (95 mg). The mixture was stirred at room temperature for 12 hours and evaporated. The residual oil was dissolved in chloroform, washed with saturated aqueous sodium hydrogen carbonate and brine, dried over anhydrous magnesium sulfate and evaporated. Resulting residue was applied to a column of silica gel and eluted using chloroform-methanol (40:1) and then chloroform-methanol (20:1). Fractions containing the product were collected and evaporated. The free base of the product was then converted to a maleate by the usual method.
Yield: 93 mg (49%). 1H-NMR (400 MHz, measured as a maleate, DMSO-d6) δ: 1.87-2.01 (m, 4H), 2.48-2.56 (m, 1H), 2.78-2.86 (m, 2H), 3.26-3.31 (m, 2H), 3.78 (s, 3H), 3.79 (s, 3H), 3.87 (s, 6H), 3.90 (s, 6H), 4.15 (s, 2H), 4.39 (d, 2H, J=5.9 Hz), 6.16 (s, 2H), 7.16 (d, 1H, J=5.9 Hz), 7.35 (s, 2H), 7.39 (d, 1H, J=5.9 Hz), 7.39 (s, 2H), 7.73 (s, 1H), 7.95 (s, 1H), 8.15 (d, 1H, J=5.9 Hz), 8.54 (d, 1H, J=4.9 Hz), 8.68 (d, 1H, J=4.9 Hz).
PREPARATION EXAMPLE 8
Synthesis of 1-(benzyloxycarbonyl)-4-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyloxy]piperidine
To a solution of 1-(benzyloxycarbonyl)-4-hydroxypiperidine (1.00 g) in DMF (20 mL) was added sodium hydride (55%, dispersion in mineral oil, 222 mg). The mixture was stirred at room temperature for 1 hour and then, 4-chlolromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (1.37 g) and potassium iodide (755 mg) was added. The mixture was stirred at 70° C. overnight, poured into water and extracted with chloroform. The organic layer was washed with brine, dried over anhydrous sodium sulfate and evaporated. The residual oil was applied to a column of silica gel and column chromatography was performed using chloroform-methanol (99:1) as an eluent giving the title compound.
Yield: 213 mg (10%). 1H NMR (400 MHz, CDCl3) δ: 1.63 (br, 2H), 1.89 (br, 2H), 3.20-3.35 (m, 2H), 3.57-3.68 (m, 1H), 3.84-3.92 (m, 5H), 3.94 (s, 6H), 4.62 (s, 2H), 5.11 (s, 2H), 7.21-7.35 (m, 8H), 7.61 (s, 1H), 8.61 (d, 1H, J=5.0 Hz).
PREPARATION EXAMPLE 9
Synthesis of 4-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyloxy]piperidine
To a solution of 1-(benzyloxycarbonyl)-4-[[2-(3,4,5-trimethoxyphenyl) pyridin-4-yl]methyloxy]piperidine (213 mg) in methanol (10 mL) was added 40% aqueous potassium hydroxide (10 mL). The mixture was stirred at 100° C. for 3 hours and evaporated. Water was added to the residue and extracted with chloroform. The organic layer was washed with brine, dried over anhydrous sodium sulfate and evaporated. The residual oil was subjected to column chromatography of silica gel using chloroform-ammonia saturated methanol (20:1) to give the title compound.
Yield: 93 mg (60%). 1H NMR (400 MHz, CDCl3) δ: 1.55-1.68 (m, 2H), 2.01 (br, 2H), 2.67-2.72 (m, 2H), 3.13-3.18 (m, 2H), 3.50-3.60 (m, 1H), 3.91 (s, 3H), 3.97 (s, 6H), 4.64 (s, 2H, 7.22 (d, 1H, J=4.3 Hz), 7.24 (s, 2H), 7.64 (s, 1H), 8.63 (d, 1H, J=5.1 Hz).
EXAMPLE 2
Synthesis of 1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]-4-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyloxy]piperidine trihydrochloride
4-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyloxy]piperidine (70 mg), 4-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (22 mg), potassium carbonate (56 mg) and potassium iodide (40 mg) were suspended in acetonitrile (5 mL). The mixture was stirred at room temperature for 5 hr and evaporated. Chloroform and water were added to the residual oil and the organic layer was separated. Aqueous layer was then extracted with chloroform and the organic layers were combined, dried over anhydrous magnesium sulfate and evaporated. The residue was applied to a column of silica gel using chloroform-methanol (40:1) as an eluent. Fractions containing the product were collected and evaporated. The title compound was obtained by converting the free base to a trihydrochloride.
Yield: 42 mg (39%). 1H NMR (400 MHz, measured as a free base, CDCl3) δ: 1.53-2.42 (m, 6H), 2.80 (br, 2H), 3.57 (br, 3H), 3.88 (s, 6H), 3.94 (s, 6H), 3.95 (s, 6H), 4.60 (s, 2H), 7.18-7.24(m, 6H), 7.61 (s, 2H), 8.58-8.61 (m, 2H).
PREPARATION EXAMPLE 10
Synthesis of (3S)-1-(tert-butoxycarbonyl)-3-[(2-nitrobenzene)sulfonylamino]pyrrolidine
To an ice-cooled solution of (3S)-3-amino-1-(tert-butoxycarbonyl) pyrrolidine (404 mg) and triethylamine (220 mg) in THF (5 mL) was added 2-nitrobenzenesulfonyl chloride (481 mg). The mixture was stirred at room temperature for 30 minutes and evaporated. Ethyl acetate was added to the residue. The solution was washed with water and brine, dried over anhydrous sodium sulfate and evaporated. The residual oil was subjected to a column of silica gel and column chromatography was performed using chroloform-methanol (20:1) as an eluent. Fractions containing the product were collected and evaporated to give the title compound as pale yellow amorphous.
Yield: 597 mg (74%). 1H-NMR (400 MHz, CDCl3) δ: 1.44 (s, 9H), 1.80-2.12 (m, 2H), 3.14-3.44 (m, 4H), 4.02 (br, 1H), 5.48 (d, 1H, J=7.2 Hz), 7.77 (t, 2H, J=4.4 Hz), 7.87-7.90 (m, 1H), 8.17-8.19 (m, 1H).
PREPARATION EXAMPLE 11
Synthesis of (3S)-1-(tert-butoxycarbonyl)-3-[N-methyl-N-(2-nitrobenzene) sulfonylamino]pyrrolidine
To a suspension of (3S)-1-(tert-butoxycarbonyl)-3-[(2-nitrobenzene) sulfonylamino]pyrrolidine (371 mg) and potassium carbonate (141 mg) in acetonitrile (10 mL) was added methyl iodide (141 mg). The mixture was stirred at 60° C. for 2 hours and evaporated. Ethyl acetate was added to the mixture. The solution was washed with saturated aqueous sodium hydrogen carbonate and brine, dried over anhydrous sodium sulfate and evaporated. The residue was applied to a column of silica gel using hexane-ethyl acetate (2:1) as an eluent. Fractions containing the product were collected and evaporated to give the title compound as yellow syrup.
Yield: 365 mg (95%). 1H-NMR (400 MHz, CDCl3) δ: 1.44 (s, 9H), 1.95 (br, 1H), 2.09 (br, 1H), 2.87 (s, 3H), 3.20-3.31 (m, 2H), 3.53 (br, 2H), 4.58 (br, 1H), 7.65 (br, 1H), 7.71 (br, 2H), 8.04 (br, 1H).
PREPARATION EXAMPLE 12
Synthesis of (3S)-3-[N-methyl-N-(2-nitrobenzene)sulfonylamino]pyrrolidine
To an ice-cooled solution of (3S)-1-(tert-butoxycarbonyl)-3-[N-methyl-N-(2-nitrobenzenesulfonyl)amino]pyrrolidine (365 mg) in dichloromethane (25 mL) was added trifluoroacetic acid (1 mL). The mixture was stirred at room temperature for 3 hours and evaporated. The residue was dissolved in chloroform. The solution was washed with saturated aqueous sodium hydrogen carbonate and brine, dried over anhydrous sodium sulfate and evaporated to give the title compound as yellow syrup.
Yield: 135 mg (50%). 1H-NMR (400 MHz, CDCl3) δ: 1.69-1.74 (m, 1H), 1.87 (br, 1H), 1.95-2.02 (m, 1H), 2.80 (dd, 1H, J=11.7 Hz, 5.7 Hz), 2.84-2.91 (m, 4H), 2.96-3.05 (m, 1H), 3.10 (dd, 1H, J=11.7 Hz, 8.2 Hz), 4.48-4.56 (m, 1H), 7.61-7.63 (m, 1H), 7.66-7.73 (m, 2H), 8.01-8.04 (m, 1H).
PREPARATION EXAMPLE 13
Synthesis of (3S)-3-[N-methyl-N-(2-nitrobenzene)sulfonylamino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]pyrrolidine
(3S)-3-[N-methyl-N-(2-nitrobenzene)sulfonylamino]pyrrolidine (135 mg) and 4-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (139 mg) were coupled in the same manner as described in Example 2 to give the title compound as yellow amorphous.
Yield: 247 mg (96%). 1H-NMR (400 MHz, CDCl3) δ: 1.80-1.87 (m, 1H), 2.15-2.30 (m, 2H), 2.52 (dd, 1H, J=10.5 Hz, 8.2 Hz), 2.71 (dd, 1H, J=10.5 Hz, 8.2 Hz), 2.90 (dt, 1H, J=8.8 Hz, 2.9 Hz), 2.96 (s, 3H), 3.53 (d, 1H, J=13.9 Hz), 3.68 (d, 1H, J=13.9 Hz), 3.90 (s, 3H), 3.96 (s, 6H), 4.61-4.68 (m, 1H), 7.16 (dd, 1H, J=4.9 Hz, 1.2 Hz), 7.21 (s, 2H), 7.58-7.60 (m, 2H), 7.64-7.69 (m, 2H), 7.99-8.02 (m, 1H), 8.58 (d, 1H, J=4.9 Hz,).
PREPARATION EXAMPLE 14
Synthesis of (3S)-3-methylamino-1-[[2-(3,4,5-trimethoxyphenyl)-pyridin-4-yl]methyl]pyrrolidine
To a solution of (3S)-3-[N-methyl-N-(2-nitrobenzene)sulfonylamino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]pyrrolidine (242 mg) in acetonitrile (5 mL) was added potassium carbonate (94 mg) and thiophenol (75 mg). The mixture was stirred at 80° C. for 3 hours and evaporated. Ethyl acetate was added to the mixture, the solution was washed with saturated aqueous sodium hydrogen carbonate, water, and bine, dried over anhydrous sodium sulfate and evaporated. The residual oil was subjected to preparative TLC using chloroform-methanol (20:1) as a solvent system giving yellow syrup of the title compound.
Yield: 104 mg (64%). 1H-NMR (400 MHz, CDCl3) δ: 1.32 (br, 1H), 1.56-1.64 (m, 1H), 2.11-2.17 (m, 1H), 2.38 (s, 3H), 2.44 (dd, 1H, J=7.4 Hz, 4.5 Hz), 2.50-2.55 (m, 1H), 2.66-2.75 (m, 2H), 3.20-3.26 (m, 1H), 3.66 (s, 2H), 3.90 (s, 3H), 3.97 (s, 6H), 7.21 (d, 1H, J=4.1 Hz), 7.25 (s, 2H), 7.64 (s, 1H), 8.59 (d, 1H, J=4.9 Hz).
EXAMPLE 3
Synthesis of (3S)-3-[N-methyl-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]pirrolidine tertrahydrochloride
(3S)-3-methylamino-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]pyrrolidine (104 mg) and 4-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (85 mg) was condensed in the same manner as described in Example 2. Yellow syrup obtained was converted to a tetrahydrochloride by the usual method giving the title compound as yellow powder.
Yield: 151 mg (68%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.89-1.92 (m, 1H), 2.04-2.08 (m, 1H), 2.18 (s, 3H), 2.60-2.76 (m, 4H), 3.25-3.29 (m, 1H), 3.53 (d, 1H, J=14.3 Hz), 3.62 (d, 1H, J=14.3 Hz), 3.64 (d, 1H, J=13.9 Hz), 3.73 (d, 1H, J=13.9 Hz), 3.89 (s, 6H), 3.95 (s, 6H), 3.96 (s, 6H), 7.20-7.21 (m, 2H), 7.23 (s, 2H), 7.24 (s, 2H), 7.61 (s, 1H), 7.65 (s, 1H), 8.59 (d, 1H, J=5.7 Hz), 8.60 (d, 1H, J=5.3 Hz).
PREPARATION EXAMPLE 15
Synthesis of 1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine-4-carboxamide
Piperidine-4-carboxamide (385 mg) and 4-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (881 mg) were condensed by the same method as described in Example 2 to give the title compound as white needles.
Yield: 1.01 g (87%). 1H-NMR (400 MHz, CDCl3) δ: 1.70-1.88 (m, 4H), 2.01-2.23 (m, 3H), 2.95 (d, 2H, J=11.0 Hz), 3.56 (s, 2H), 3.90 (s, 3H), 3.98 (s, 6H), 5.46 (d, 2H, J=16.3 Hz), 7.21 (d, 1H, J=5.0 Hz), 7.24 (s, 2H), 7.64 (s, 1H), 8.59 (d, 1H, J=5.0 Hz).
PREPARATION EXAMPLE 16
Synthesis of 4-amino-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine trihydrochloride
To a solution of 1-[[2-(3,4,5-trimethoxypheyl)pyridin-4-yl]methyl]piperidine-4-carboxamide (192 mg) in a mixed solvent of water (50 mL) and acetonitrile (50 mL) was added [bis(trifluoroacetoxy)iodo]benzene (323 mg). The mixture was stirred at room temperature overnight and evaporated. Saturated aqueous sodium hydrogen carbonate was added to the residue and extracted with chloroform. The organic layer was washed with brine, dried over anhydrous magnesium sulfate and evaporated. Yellow syrup obtained was then converted to trihydrochloride which gave yellow powder. The title compound was used for next step without further purification.
Yield: 201 mg (theoretical amount).
PREPARATION EXAMPLE 17
Synthesis of 2-(3,4,5-trimethoxyphenyl)isonicotinic acid
To a solution of ethyl 2-(3,4,5-trimethoxyphenyl)isonicotinate (3.17 g) in ethanol (40 mL) was added 10% potassium hydroxide (2.42 g). The mixture was stirred at room temperature for 5 hours and evaporated. Water was added to the residue and pH was adjusted to 7. White precipitates of the title compound were collected by filtration and the compound was used for next step without further purification.
Yield: 2.60 g (90%).
EXAMPLE 4
Synthiesis of 4-[2-(3,4,5-trimethoxyphenyl)pyridin-4-carbonylamino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine maleate
2-(3,4,5-trimethoxyphenyl)isonicotinic acid (72 mg) and 4-amino-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine (117 mg) were condensed in the same manner as described in Example 1. The title compound was obtained as a maleate.
Yield: 173 mg (93%). 1H-NMR (400 MHz, measured as a maleate, DMSO-d6) δ: 1.82-1.94 (m, 2H), 2.03-2.08 (m, 2H), 2.77-2.83 (m, 2H), 3.20-3.27 (m, 2H), 3.79 (s, 6H), 3.90 (s, 12H), 4.00 (br, 1H), 4.06 (s, 2H), 6.15 (s, 2H), 7.36-7.38 (m, 1H), 7.39 (s, 2H), 7.41 (s, 2H), 7.61-7.63 (m, 1H), 7.90 (s, 1H), 8.12 (s, 1H), 8.27-8.32 (m, 1H), 8.67 (d, 1H, J=4.9 Hz), 8.74 (d, 1H, J=5.1 Hz).
PREPARATION EXAMPLE 18
Synthesis of 4-[(2-nitrobenzene)sulfonylamino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine
4-amino-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine (467 mg) and 2-nitrobenzenesulfonyl chloride (244 mg) were condensed in the same manner as described in Preparation Example 10 to give the title compound.
Yield: 494 mg (91%).
PREPARATION EXAMPLE 19
Synthesis of 4-[N-(2-nitrobenzene)sulfonyl-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]-1-[[2-(3,4,5-trimethoxyphneyl)pyridin-4-yl]methyl]piperidine
4-[(2-nitrobenzene)sulfonylamino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine (494 mg) and 4-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (267 mg) were condensed in the same manner as described in Example 2 to give the title compound.
Yield: 443 mg (61%).
EXAMPLE 5
Synthesis of 1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]-4-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methylamino]piperidine difumalate
4-[N-(2-nitrobenzene)sulfonyl-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]-1-[[2-(3,4,5-trimethoxyphneyl)pyridin-4-yl]methyl]piperidine (443 mg) was treated in the same manner as described in Preparation Example 14. The title compound was obtained after converting to a difumalate.
Yiled: 103 mg (24%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.44-1.53 (m, 2H), 1.87-1.91 (m, 2H), 2.15 (t, 2H, J=1.1 Hz), 2.57-2.64 (m, 1H), 2.82-2.85 (m, 2H), 3.59 (s, 2H), 3.78 (s, 6H), 3.89 (s, 12H), 3.90 (s, 2H), 6.63 (s, 4H), 7.24 (d, 1H, J=4.9 Hz), 7.29 (d, 1H, J=4.9 Hz), 7.35 (s, 2H), 7.37 (s, 2H), 7.76 (s, 1H), 7.85 (s, 1H), 8.53-8.56 (m, 2H).
PREPARATION EXAMPLE 20
Synthesis of 4-(ethoxycarbonylamino)-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine
To a solution of 1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine-4-carboxamide (528 mg) in a mixed solvent of ethanol (10 mL) and acetonitrile (10 mL) was added [bis(trifluoroacetoxy)iodo]benzene (884 mg). The mixture was stirred at room temperature overnight and evaporated. Saturated aqueous sodium hydrogen carbonate was added to the residue and extracted with chloroform. The organic layer was washed with brine, dried over anhydrous magnesium sulfate and evaporated. The residue was applied to a column of silica gel and purified using chloroform-methanol (20:1) as an eluent to give the title compound.
Yield: 566 mg (96%). 1H-NMR (400 MHz, CDCl3) δ: 1.21 (t, 3H, J=7.0 Hz), 1.40-1.51 (m, 2H), 1.92 (d, 2H, J=10.9 Hz), 2.15 (t, 2H, J=10.9 Hz), 2.78 (d, 2H, J=11.6 Hz), 3.52 (br, 3H), 3.87 (s, 3H), 3.94 (s, 6H), 4.07 (q, 2H, J=7.0 Hz), 4.56 (br, 1H), 7.17 (d, 1H, J=4.9 Hz), 7.21 (s, 2H), 7.59 (s, 1H), 8.56 (d, 1H, J=5.1 Hz).
PREPARATION EXAMPLE 21
Synthesis of 4-(methylamino)-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine
To a suspension of lithium aluminum hydride (100 mg) in dry THF (50 mL) was added a solution of 4-(ethoxycarbonylamino)-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine (566 mg) in dry THF (50 mL) under an argon atmosphere. The mixture was then refluxed overnight, then cooled down. Saturated aqueous ammonium chloride was added to the mixture and extracted with ethyl acetate. The organic layer was washed with brine, dried over anhydrous sodium sulfate and evaporated. The residue was subjected to silica gel column chromatography using chloroform-ammonia saturated methanol (9:1) to give the title compound as yellow oil.
Yiled: 379 mg (78%). 1H-NMR (400 MHz, CDCl3) δ: 1.36-1.46 (m, 2H), 1.89 (d, 2H, J=12.5 Hz), 2.10 (dt, 2H, J=11.5 Hz, 1.1 Hz), 2.35-2.43 (m, 1H), 2.43 (s, 3H), 2.86 (d, 2H, J=11.6 Hz), 3.56 (s, 2H), 3.90 (s, 3H), 3.97 (s, 6H), 7.21 (d, 1H, J=5.1 Hz), 7.24 (s, 2H), 7.64 (s, 1H), 8.59(d, 1H, J=4.9 Hz).
PREPARATION EXAMPLE 22
Synthesis of 1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]-4-piperidone ethylene ketal
4-Piperidone ethylene ketal (12.0 g) and 4-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (12.3 g) was condensed in the same manner as described in Example 2 to give the title compound.
Yield: 19.0 g (theoretical amount). 1H-NMR (400 MHz, CDCl3) δ: 1.68 (t, 4H, J=5.6 Hz), 2.48 (br, 4H), 3.50 (s, 2H), 3.82 (s, 3H), 3.86 (s, 4H), 3.88 (s, 6H), 7.13 (d, 1H, J=4.9 Hz), 7.17 (s, 2H), 7.57 (s, 1H), 8.51 (d, 1H, J=4.9 Hz).
PREPARATION EXAMPLE 23
Synthesis of 1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]-4-piperidone
To a solution of 1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]-4-piperidone ethylene ketal (19.0 g) in THF (200 mL) was added 1 M hydrochloric acid (200 mL). The mixture was stirred at 90° C. overnight, then neutralized with 2 M sodium hydroxide and extracted with ethyl acetate. The organic layer was washed with brine, dried over anhydrous sodium sulfate and evaporated. The residual oil was applied to a column of silica gel using chloroform-methanol (40:1) as an eluent. Fractions containing the product were collected and evaporated to give the title compound.
Yield: 15.0 g (75%). 1H-NMR (400 MHz, CDCl3) δ: 2.48 (t, 4H, J=6.1 Hz), 2.79 (t, 4H, J=6.0 Hz), 3.69 (s, 2H), 3.89 (s, 3H), 3.96 (s, 6H), 7.24 (s, 2H), 7.26 (d, 1H, J=4.9 Hz), 7.66 (s, 1H), 8.62 (d, 1H, J=4.9 Hz).
PREPARATION EXAMPLE 24
Synthesis of 1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]-4-piperidone
4-Piperidone hydrochloride monohydrate (3.07 g) and 4-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (2.94 g) were coupled by the same manner as described in Example 2 to give the title compound.
Yield: 3.55 g (99%).
PREPARATION EXAMPLE 25
Synthesis of 4-(methylamino)-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine
To a solution of 1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]-4-piperidone (1.00 g) in 1,2-dichloroethane (60 mL) was added 30% solution of methylamine in ethanol (750 mg) and sodium triacetoxyborohydride (1.66 g). The mixture was stirred at room temperature for 3 hours, then small amount of water was added and evaporated. Water was added to the residue and extracted with chloroform. The organic layer was washed with brine, dried over anhydrous sodium sulfate and evaporated. The residue was subjected to silica gel column chromatography using chloroform-methanol (40:1) to give the title compound.
Yield: 640 mg (62%).
PREPARATION EXAMPLE 26
Synthesis of ethyl 3-(3,4,5-trimethoxyphenyl)benzoate
3,4,5-Trimethoyphenylboronic acid (3.7 g) and ethyl 3-bromobenzoate (4.02 g) were condensed in the same manner as described in Preparation Example 1 to give the title compound.
Yield: 5.09 g (92%). 1H-NMR (400 MHz, CDCl3) δ: 1.42 (t, 3H, J=7.1 Hz), 3.90 (s, 3H), 3.94 (s, 6H), 4.41 (q, 2H, J=7.1 Hz), 6.79 (s, 2H), 7.50 (t, 1H, J=7.8 Hz), 7.73 (dt, 1H, J=7.1 Hz, 1.5 Hz), 8.01 (dt, 1H, J=7.8 Hz, 1.4 Hz), 8.23 (t, 1H, J=1.8 Hz).
PREPARATION EXAMPLE 27
Synthesis of 3-(3,4,5-trimethoxyphenyl)benzoic acid
Ethyl 3-(3,4,5-trimethoxyphenyl)benzoate (1.19 g) was treated in the same manner as described in Preparation Example 17 to give the title compound.
Yield: 986 mg (91%).
EXAMPLE 6
Synthesis of 4-[N-methyl-N-[3-(3,4,5-trimethoxyphenyl)]benzoylamino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine dihydrochloride
3-(3,4,5-trimethoxyphenyl)benzoic acid (1.03 g) and 4-(methylamino)-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine (1.32 g) were condensed in the same method as described in Example 1. The title compound was obtained after converting a free amine to a dihydrochloride.
Yield: 1.44 g (57%). 1H-NMR (400 MHz, measured as a dihydrochloride, DMSO-d6) δ: 1.89 (d, 2H, J=11.7 Hz), 2.54-2.62 (m, 2H), 2.89 (s, 3H), 3.09 (t, 2H, J=12.7 Hz), 3.43 (d, 2H, J=14.4 Hz), 3.76 (s, 3H), 3.78 (s, 3H), 3.88 (s, 6H), 3.91 (s, 6H), 4.34 (br, 3H), 6.91 (s, 2H), 7.33 (d, 1H, J=7.6 Hz), 7.47-7.51 (m, 2H), 7.54 (s, 2H), 7.60 (s, 1H), 7.71 (d, 1H, J=7.8 Hz), 8.55 (s, 1H), 8.68 (d, 1H, J=5.1 Hz).
EXAMPLE 7
Synthesis of 4-[N-methyl-N-[[5-(3,4,5-trimethoxyphenyl)pyridin-3-yl]methyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine difumarate
4-methylamino-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine (135 mg) and 3-chloromethyl-5-(3,4,5-trimethoxypyenyl)pyridine (107 mg) were condensed by the same method as described in Example 2. White powder of the title compound was obtained after converting a free base to a difumarate.
Yield: 180 mg (58%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.69-1.73 (m, 2H), 1.82-1.85 (m, 2H), 2.03-2.08 (m, 2H), 2.25 (s, 3H), 2.48-2.51 (m, 1H), 2.97-2.99 (m, 2H), 3.56 (s, 2H), 3.67 (s, 2H), 3.90 (s, 3H), 3.91 (s, 3H), 3.94 (s, 6H), 3.98 (s, 6H), 6.76 (s, 2H), 7.22 (d, 1H, J=5.1 Hz), 7.24 (s, 2H), 7.62 (s, 1H), 7.80 (s, 1H), 8.50 (d, 1H, J=2.0 Hz), 8.60 (d, 1H, J=4.3 Hz), 8.69 (d, 1H, J=5.1 Hz).
PREPARATION EXAMPLE 28
Synthesis of 1-bromo-4-chloro-3,5-dimethoxybenzene
A solution of sodium nitrite (97 mg) in water (2.0 mL) was added dropwise to an ice-cold suspension of 4-bromo-2,6-dimethoxyaniline (232 mg) in 6.0 M hydrochloric acid (2.5 mL). After stirring in ice for 30 minutes, a solution of cupric chloride (495 mg) in concentrated hydrochloric acid (2.0 mL) was added. The reaction mixture was stirred at room temperature for 30 minutes, then at 100° C. for 2 hours, and extracted with ethyl acetate. The organic layer was washed with a saturated aqueous sodium hydrogencarbonate and water, dried over anhydrous sodium sulfate, and evaporated. The residue was subjected to a column of silica gel using hexane-ethyl acetate (10:1) as an eluent to give the title compound as white powder.
Yield: 230 mg (92%).
PREPARATION EXAMPLE 29
Synthesis of 4-chloro-3,5-dimethoxyphenylboronic acid
Under an argon atomsphere, to dry THF (2 mL) stirred in a dry ice-methanol bath was gradually added a 1.57 M solution of n-butyllithium in hexane (0.8 mL), followed by the dropwise addition of a solution of 1-bromo-4-chloro-3,5-dimethoxybenzene (160 mg) in dry THF (2 mL). After the mixture was stirred for 20 minutes in the dry ice-methanol bath, triisopropyl borate (0.18 mL) was added and the mixture was additionally stirred for 20 minutes. The reaction mixture was then stirred at room temperature for 1 hour and pH of the mixture was adjusted at 3 using 4 M hydrochloric acid. The mixture was stirred at 0° C. for 1 hour and extracted with ethyl acetate. The organic layer was washed with brine, dried over anhydrous sodium sulfate and evaporated. The residue was recrystallized from ethyl acetate-hexane giving the title compound as white powder.
Yield: 90 mg (66%).
PREPARATION EXAMPLE 30
Synthesis of ethyl 2-(4-chloro-3,5-dimethoxyphenyl)isonicotinate
4-Chloro-3,5-dimethoxyphenylboronic acid (7.45 g) and ethyl 2-chloroisonicotinate (6.39 g) were condensedn in the same manner as described in Preparation Example 1 to give the title compound.
Yield: 8.55 g (77%). 1H-NMR (400 MHz, CDCl3) δ: 1.45 (t, 3H, J=7.3 Hz), 4.03 (s, 6H), 4.45 (q, 2H, J=7.3 Hz), 7.32 (s, 2H), 7.80 (d, 1H, J=5.1 Hz), 8.27 (s, 1H), 8.83 (d, 1H, J=5.0 Hz).
PREPARATION EXAMPLE 31
Synthesis of 2-(4-chloro-3,5-dimethoxyphenyl)isonicotinic acid
To a solution of ethyl 2-(4-chloro-3,5-dimethoxyphenyl)isonicotinate (8.55 g) in ethanol (80 mL) was added 2 M sodium hydroxide (100 mL). The mixture was refluxed for 30 min and evaporated. The aqueous layer was neutralized by 1 M hydrochloric acid and precipitates were dissolved in a mixed solvent of ethyl acetate-THF (3:1). After drying over anhydrous sodium sulfate, the solvent was evaporated to give the title compound.
Yield: 7.20 g (92%). 1H-NMR (400 MHz, CDCl3) δ: 4.02 (s, 6H), 7.34 (s, 2H), 7.83 (d, 1H, J=4.9 Hz), 7.84 (s, 1H), 8.82 (d, 1H, J=4.9 Hz).
PREPARATION EXAMPLE 32
Synthesis of 2-(4-chloro-3,5-dimethoxyphenyl)-4-hydroxymethylpyridine
To an ice-cooled solution of 2-(4-chloro-3,5-dimethoxyphenyl)isonicotinic acid (7.20 g) and triethylamine (5.6 mL) in THF (70 mL) was added ethyl chloroformate (2.8 mL). The mixture was stirred at room temperature for 1 hour and filtered. To the filtrate was then added a solution of sodium borohydride (1.25 g) in water (4 mL). The mixture was stirred at room temperature for another hour and evaporated. Water was added to the residue and extracted with chloroform. The organic layer was washed with brine, dried over anhydrous sodium sulfate and evaporated. The residue was subjected to silica gel column chromatography using chloroform-methanol (20:1) and then chloroform-methanol (15:1) to give the title compound.
Yield: 4.10 g (60%). 1H-NMR (400 MHz, CDCl3+DMSO-d6) δ: 4.01 (s, 6H), 4.76 (s, 2H), 7.20-7.35 (m, 3H), 7.78 (s, 1H), 8.62 (s,1H).
PREPARATION EXAMPLE 33
Synthesis of 2-(4-chloro-3,5-dimethoxyphenyl)-4-chloromethylpyridine
2-(4-Chloro-3,5-dimethoxyphenyl)-4-hydroxymethylpyridine (4.10 g) was treated in the same manner as described in Preparation Example 3 to give the title compound.
Yield: 4.20 g (96%). 1H-NMR (400 MHz, CDCl3) δ: 4.02 (s, 6H), 4.63 (s, 2H), 7.26 (s, 2H), 7.29 (d, 1H, J=4.9 Hz), 7.72 (s, 1H), 8.69 (d, 1H, J=4.9 Hz).
PREPARATION EXAMPLE 34
Synthesis of 1-[[2-(4-chloro-3,5-dimethoxyphenyl)pyridin-4-yl]methyl]piperidine-4-carboxamide
Piperidine-4-carboxamide (301 mg) and 2-(4-chloro-3,5-dimethoxyphenyl)-4-chloromethylpyridine (600 mg) were coupled in the same manner as described in Example 2 to give the title compound.
Yield: 743 mg (95%). 1H-NMR (400 MHz, CDCl3) δ: 1.75-1.90 (m, 4H), 2.07-2.25 (m, 3H), 2.94 (d, 2H, J=11.6 Hz), 3.57 (s, 2H), 4.02(s, 6H), 7.24-7.31 (m, 3H), 7.67 (s, 1H), 8.61 (d, 1H, J=5.1 Hz).
PREPARATION EXAMPLE 35
Synthesis of 1-[[2-(4-chloro-3,5-dimethoxyphenyl)pyridin-4-yl]methyl]-4-(ethoxycarbonylamino)piperidine
1-[[2-(4-chloro-3,5-dimethoxyphenyl)pyridin-4-yl]methyl]piperidine-4-carboxamide (743 mg) was treated in the same manner as described in Preparation Example 20 to give the title compound.
Yield: 887 mg (theoretical amount). 1H-NMR (400 MHz, CDCl3) δ: 1.24 (t, 3H, J=7.1 Hz), 1.43-1.59 (m, 2H), 1.96 (d, 2H, J=11.4 Hz), 2.19 (t, 2H, J=11.0 Hz), 2.82 (d, 2H, J=11.5 Hz), 3.56 (s, 2H), 4.02 (s, 6H), 4.10 (q, 2H, J=7.1 Hz), 7.26 (s, 2H), 7.66 (s, 1H), 7.71 (dd, 1H, J=5.6 Hz, 1.0 Hz), 8.6 (dd, 1H, J=4.9 Hz, 0.5 Hz).
PREPARATION EXAMPLE 36
Synthesis of 1-[[2-(4-chloro-3,5-dimethoxyphenyl)pyridin-4-yl]methyl]-4-methylaminopiperidine
1-[[2-(4-chloro-3,5-diemthoxyphenyl)pyridin-4-yl]methyl]-4-(ethoxy-carbonylamino)piperidine (887 mg) was treated in the same manner as described in Preparation Example 21 to give the title compound.
Yield: 195 mg (27%). 1H-NMR (400 MHz, CDCl3) δ: 1.35-1.49 (m, 2H), 1.89 (d, 2H, J=12.3 Hz), 2.11 (t, 2H, J=9.4 Hz), 2.38-2.45 (m, 1H), 2.44 (s, 3H), 2.87 (d, 2H, J=10.7 Hz), 3.57 (s, 2H), 4.02 (s, 6H), 7.23-7.29 (m, 3H), 7.68 (s, 1H), 8.61 (d, 1H, J=4.9 Hz).
EXAMPLE 8
Synthesis of 1-[[2-(4-chloro-3,5-dimethoxyphenyl)pyridin-4-yl]methyl]-4-[N-[[2-(4-chloro-3,5-dimethoxyphenyl)pyridin-4-yl]methyl]-N-methylamino]piperidine tetrahydrochloride
1-[[2-(4-chloro-3,5-dimethoxyphenyl)pyridin-4-yl]methyl]-4-methylaminopiperidine (195 mg) and 2-(4-chloro-3,5-dimethoxyphenyl)-4-chloromethylpyridine (152 mg) were condensed in the same manner as described in Example 2. A free base obtained was converted to a tetrahydrochloride giving yellow powder.
Yield: 300 mg (75%). 1H-NMR (400 MHz, CDCl3) δ: 1.60-1.90 (m, 4H), 2.06 (t, 2H, J=11.7 Hz), 2.26 (s, 3H), 2.45-2.55 (m, 1H), 2.97 (d, 2H, J=11.3 Hz), 3.57 (s, 2H), 3.67 (s, 2H), 4.01 (s, 6H), 4.02 (s, 6H), 7.24-7.28 (m, 6H), 7.65 (s, 1H), 7.67 (s, 1H), 8.61 (d, 1H, J=5.4 Hz), 8.62 (d, 1H, J=5.4 Hz).
PREPARATION EXAMPLE 37
Synthesis of 4-(p-anisidino)-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine
To a solution of 1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]-4-piperidone (2.17 g) in toluene (40 mL) was added p-anisidine (900 mg) and molecular sieves 4A (6.0 g). The mixture was refluxed overnight, then filtered and the filtrate was evaporated. The residual oil was dissolved in ethanol (40 mL) and sodium borohydride (276 mg) was added. The mixture was stirred at room temperature for 2 hours before concentration in vacuo. The residue was dissolved in ethyl acetate, washed with brine, dried over anhydrous sodium sulfate and evaporated. The residual oil was subjected to silica gel column chromatography using chloroform-methanol (50:1) to give the title compound as yellow amorphous.
Yield: 1.56 g (55%). 1H-NMR (400 MHz, CDCl3) δ: 1.48 (br, 2H), 2.05 (br, 2H), 2.20 (br, 2H), 2.86 (br, 2H), 3.23 (s, 1H), 3.58 (s, 2H), 3.74 (s, 3H), 3.91 (s, 3H), 3.97 (s, 6H), 6.58 (d, 2H, J=8.8 Hz), 6.77 (d, 2H, J=9.0 Hz), 7.22 (d, 1H, J=5.1 Hz), 7.26 (s, 2H), 7.64 (s, 1H), 8.59 (d, 1H, J=4.9 Hz).
PREPARATION EXAMPLE 38
Synthesis of ethyl 2-(3,4,5-trimethoxyphenyl)nicotinate
3,4,5-Trimethoxyphenylboronic acid (694 mg) and ethyl 2-chloronicotinate (608 mg) were reacted in the same manner as described in Preparation Example 1 to give the title compound.
Yield: 799 mg (77%). 1H-NMR (400 MHz, CDCl3) δ: 1.10 (t, 3H, J=7.2 Hz), 3.89 (s, 9H), 4.19 (q, 2H, J=7.2 Hz), 6.79 (s, 2H), 7.34 (dd, 1H, J=7.8 Hz, 4.8 Hz), 8.06 (dd, 1H, J=7.8 Hz, 1.7 Hz), 8.75 (dd, 1H, J=4.8 Hz, 1.7 Hz).
PREPARATION EXAMPLE 39
Synthesis of 3-hydroxymethyl-2-(3,4,5-trimethoxyphenyl)pyridine
Ethyl 2-(3,4,5-trimethoxyphenyl)nicotinate (468 mg) was treated in the same manner as described in Preparation Example 2 to give the title compound.
Yield: 293 mg (72%). 1H-NMR (400 MHz, CDCl3) δ: 3.90 (s, 9H), 4.72 (s, 2H), 6.83 (s, 2H), 7.32 (dd, 1H, J=7.9 Hz, 4.8 Hz), 7.92 (dd, 1H, J=7.9 Hz, 1.7 Hz), 8.62 (dd, 1H, J=4.8 Hz, 1.7 Hz).
PREPARATION EXAMPLE 40
Synthesis of 3-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine
3-Hydroxymethyl-2-(3,4,5-trimethoxyphenyl)pyridine (293 mg) was treated in the same manner as described in the Preparation Example 3 to give the title compound.
Yield: 311 mg (theoretical amount).
EXAMPLE 9
Synthesis of 4-[N-(4-methoxyphenyl)-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-3-yl]methyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine trihydrochloride
To a solution of 4-p-anisidino)-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine (139 mg) and 3-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (114 mg) in acetonitrile (5 ml) was added potassium carbonate (83 mg) and potassium iodide (63 mg). The mixture was stirred at 70° C. overnight and evaporated. The residue was dissolved in chloroform, washed with water and brine, dried over anhydrous magnesium sulfate and evaporated. The residual oil was applied to a column of silica gel using diethylether-metanol (20:1) as an eluent. A free base obtained was converted to a trihydrochloride to give the title compound as yellow powder.
Yield: 16 mg, (8%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.60 (br, 2H), 1.77 (br, 2H), 2.09 (br, 2H), 2.93 (br, 2H), 3.45 (br, 1H), 3.54 (s, 2H), 3.73 (s, 3H), 3.90 (s, 6H), 3.91 (s, 6H), 3.96 (s, 6H), 4.34 (s, 2H), 6.65 (d, 2H, J=9.0 Hz), 6.71 (s, 2H), 6.74 (d, 2H, J=9.0 Hz), 7.16-7.19 (m, 2H), 7.22 (s, 2H), 7.55 (s, 1H), 7.79 (d, 1H, J=7.0 Hz), 8.50 (br, 1H), 8.58 (d, 1H, J=4.9 Hz).
EXAMPLE 10
Synthesis of 4-[N-(4-methoxyphenyl)-N-[[2-(3,4,5-trimethoxypheny)pyridin-4-yl]methyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine trihydrochloride
4-p-Anisidino)-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine (1.56 g) and 4-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (1.08 g) were condensed by the same manner as described in Example 9. Yellow oil of a free base was converted to a trihydrochloride which gave the title compound as yellow powder.
Yield: 1.17 g (40%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.68-1.97 (m, 4H), 2.09-2.23 (m, 2H), 2.98 (br, 2H), 3.54-3.66 (m, 3H), 3.73 (s, 3H), 3.89 (s, 3H), 3.90 (s, 3H), 3.93 (s, 6H), 3.96 (s, 6H), 4.45 (s, 2H), 6.74 (d, 2H, J=9.2 Hz), 6.79 (d, 2H, J=9.2 Hz), 7.15 (s, 2H), 7.16-7.21 (m, 2H), 7.23 (s, 2H), 7.57 (s, 1H), 7.60 (s, 1H), 8.54 (d, 1H, J=5.1 Hz), 8.59 (d, 1H, J=4.9 Hz).
PREPARATION EXAMPLE 41
Synthesis of 3-(3,4,5-trimethoxyphenyl)benzyl alcohol
Ethyl 3-(3,4,5-trimethoxyphenyl)benzoate (5.09 g) was treated in the same manner as described in Preparation Example 2 to give the title compound.
Yield: 4.25 g (97%). 1H-NMR (400 MHz, CDCl3) δ: 1.87 (t, 1H, J=6.0 Hz), 3.89 (s, 3H), 3.92 (s, 6H), 4.76 (d, 1H, J=5.6 Hz), 6.77 (s, 2H), 7.34 (d, 1H, J=7.4 Hz), 7.42 (t, 1H, J=7.5 Hz), 7.48 (d, 1H, J=7.6 Hz), 7.55 (s, 1H).
PREPARATION EXAMPLE 42
Synthesis of 3-(3,4,5-trimethoxyphenyl)benzyl chloride
3-(3,4,5-Trimethoxyphenyl)benzyl alcohol (1.21 g) was treated in the same manner as described in Preparation Example 3 to give the title compound.
Yield: 893 mg (69%). 1H-NMR (400 MHz, CDCl3) δ: 3.87 (s, 3H), 3.90 (s, 6H), 4.62 (s, 2H), 6.75 (s, 2H), 7.33 (d, 1H, J=7.6 Hz), 7.39 (t, 1H, J=7.7 Hz), 7.48 (d, 1H, J=7.6 Hz), 7.54 (s, 1H).
EXAMPLE 11
Synthesis of 4-[N-(4-methoxyphenyl)-N-[3-(3,4,5-trimethoxyphenyl)benzyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine dihydrochloride
4-(p-Anisidino)-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine (139 mg) and 3-(3,4,5-trimethoxyphenyl)benzyl chloride (114 mg) were condensed by the same manner as described in Example 9. Yellow oil of a free base was converted to a dihydrochloride which gave the title compound as yellow powder.
Yield: 52 mg (22%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.77-1.92 (m, 5H), 2.14-2.20 (m, 2H), 2.95-3.00 (m, 2H), 3.58 (s, 2H), 3.72 (s, 3H), 3.88 (s, 3H), 3.89 (s, 6H), 3.90 (s, 3H), 3.96 (s, 6H), 4.47 (s, 2H), 6.70 (s, 2H), 6.74-6.83 (m, 4H), 7.20 (d, 1H, J=7.4 Hz), 7.23 (s, 2H), 7.25-7.27 (m, 1H), 7.33 (t, 1H, J=7.4 Hz), 7.38 (d, 1H, J=8.7 Hz), 7.43 (s, 1H), 7.62 (s, 1H), 8.59 (d, 1H, J=5.1 Hz).
PREPARATION EXAMPLE 43
Synthesis of ethyl 6-(3,4,5-trimethoxyphenyl)nicotinate
3,4,5-Trimethoxyphneylboronic acid (1.16 g) and ethyl 6-chloronitotinate (1.02 g) were coupled in the same manner as described in the Preparation Example 1 to give the title compound.
Yield: 1.42 g (82%). 1H-NMR (400 MHz, CDCl3) δ: 1.43 (t, 3H, J=7.2 Hz), 3.92 (s, 3H), 3.98 (s, 6H), 4.44 (q, 2H, J=7.2 Hz), 7.32 (s, 2H), 7.76 (d, 1H, J=8.3 Hz), 8.33 (dd, 1H, J=8.2 Hz, 2.2 Hz), 9.26 (d, 1H, J=2.2 Hz).
PREPARATION EXAMPLE 44
Synthesis of 5-hydroxymethyl-2-(3,4,5-trimethoxyphenyl)pyridine
Ethyl 6-(3,4,5-trimethoxyphenyl)nicotinate (658 mg) was treated in the same manner as described in Preparation Example 2 to give the title compound.
Yield: 482 mg (85%). 1H-NMR (400 MHz, CDCl3) δ: 3.91 (s, 3H), 3.97 (s, 6H), 4.76 (s, 2H), 7.23 (s, 2H), 7.68 (d, 1H, J=7.4 Hz), 7.78 (dd, 1H, J=7.4 Hz, 2.3 Hz), 8.63 (d, 1H, J=2.3 Hz).
PREPARATION EXAMPLE 45
Synthesis of 5-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine
5-Hydroxymethyl-2-(3,4,5-trimethoxyphenyl)pyridine (685 mg) was treated in the same manner as described in Preparation Example 3 to give the title compound.
Yield: 717 mg (theoretical amount).
EXAMPLE 12
Synthesis of 4-[N-(4-methoxyphenyl)-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine trihydrochloride
4-(p-Anisidino)-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine (139 mg) and 5-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (114 mg) were condensed by the same manner as described in Example 9. Yellow oil of a free base was converted to a trihydrochloride which gave the title compound as yellow powder.
Yield: 13 mg (5%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.76 (br, 2H), 1.88 (br, 2H), 2.14 (br, 2H), 2.97 (br, 2H), 3.51 (br, 1H), 3.57 (s, 2H), 3.73 (s, 3H), 3.89 (s, 3H), 3.90 (s, 3H), 3.94 (s, 6H), 3.96 (s, 6H), 4.42 (s, 2H), 6.78 (br, 4H), 7.20 (br, 3H), 7.23 (s, 2H), 7.57-7.70 (m, 3H), 8.58-8.60 (m, 2H).
PREPARATION EXAMPLE 46
Synthesis of ethyl 5-(3,4,5-trimethoxyphenyl)nicotinate
3,4,5-Trimethoxyphenylboronic acid (6.36 g) and ethyl 5-bromonicotinate (6.90 g) were reacted in the same manner as described in Preparation Example 1 to give the title compound.
Yield: 7.19 g (76%). 1H-NMR (400 MHz, CDCl3) δ: 1.44 (t, 3H, J=7.1 Hz), 3.91 (s, 3H), 3.95 (s, 6H), 4.46 (q, 2H, J=7.1 Hz), 6.79 (s, 2H), 8.44 (t, 1H, J=2.1 Hz), 8.96 (d, 1H, J=2.1 Hz), 9.18 (d, 1H, J=1.8 Hz).
PREPARATION EXAMPLE 47
Synthesis of 3-hydroxymethyl-5-(3,4,5-trimethoxyphenyl)pyridine
Ethyl 5-(3,4,5-trimethoxyphenyl)nicotinate (7.19 g) was treated in the same manner as described in the Preparation Example 2 to give the title compound.
Yield; 3.83 g (61%). 1H-NMR (400 MHz, CDCl3) δ: 3.88 (s, 3H), 3.89 (s, 6H), 4.39 (br, 1H), 4.80 (s, 2H), 6.72 (s, 2H), 7.89 (t, 1H, J=1.2 Hz), 8.47 (d, 1H, J=2.1 Hz), 8.63 (d, 1H, J=2.2 Hz).
PREPARATION EXAMPLE 48
Synthesis of 3-chloromethyl-5-(3,4,5-trimethoxyphenyl)pyridine
3-Hydroxymethyl-5-(3,4,5-trimethoxyphenyl)pyridine (2.85 g) was treated in the same manner as described in Preparation Example 3 to give the title compound.
Yield: 1.97 g (65%). 1H-NMR (400 MHz, CDCl3) δ: 3.90 (s, 3H), 3.94 (s, 6H), 4.67 (s, 2H), 6.75 (s, 2H), 7.87 (t, 1H, J=2.1 Hz), 8.59 (d, 1H, J=2.0 Hz), 8.76 (d, 1H, J=2.1 Hz).
EXAMPLE 13
Synthesis of 4-[N-(4-methoxyphenyl)-N-[[5-(3,4,5-trimethoxyphenyl)pyridin-3-yl]methyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine trihydrochloride
4-(p-Anisidino)-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine (139 mg) and 3-chloromethyl-5-(3,4,5-trimethoxyphenyl)pyridine (114 mg) were condensed by the same manner as described in Example 9. Yellow oil of a free base was converted to a trihydrochloride which gave the title compound as yellow powder.
Yield: 14 mg (5%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ; 1.73-1.75 (m, 2H), 1.88 (d, 2H, J=11.3 Hz), 2.13 (t, 2H, J=11.3 Hz), 2.96 (d, 2H, J=11.5 Hz), 3.50 (br, 1H), 3.55 (s, 2H), 3.72 (s, 3H), 3.88 (s, 3H), 3.89 (s, 9H), 3.96 (s, 6H), 4.45 (s, 2H), 6.65 (s, 2H), 6.76 (d, 2H, J=9.6 Hz), 6.80 (d, 2H, J=9.4 Hz), 7.20 (d, 1H, J=5.3 Hz), 7.22 (s, 2H), 7.59 (s, 1H), 7.67 (s, 1H), 8.50 (s, 1H), 8.59 (d, 1H, J=4.7 Hz), 8.62 (s, 1H).
PREPARATION EXAMPLE 49
Synthesis of 2,6-dimethoxy-4-iodophenol
To a solution of 5-iodo-1,2,3-trimethoxybenzene (3.2 g) in 1,2-dichloroethane (40 mL) was added aluminum chloride (1.6 g). The mixture was stirred at 60° C. for 4 hours and evaporated. The residue was dissolved in 1 M aqueous sodium hydroxide solution and washed with ether. The aqueous layer was then acidified and extracted with chloroform. The organic layer was washed with brine, dried over anhydrous magnesium sulfate and evaporated to give the title compound as white crystalline powder.
Yield: 1.0 g (31%)
PREPARATION EXAMPLE 50
Synthesis of 1,3-dimethoxy-5-iodo-2-isopropoxybenzene
To a suspension of 2,6-dimethoxy-4-iodophenol (1.0 g) and potassium carbonate (938 mg) in DMF (10 mL) was added isopropyl iodide (507 mL). The mixture was stirred at 60° C. for 3 hours and evaporated. Ethyl acetate and water were added to the residue, the organic layer was separated, washed with brine, dried over anhydrous sodium sulfate and evaporated. The residue was applied to a column of silica gel using hexane-ethyl acetate (5:1) as an eluent to give the title compound.
Yield: 788 mg (72%).
PREPARATION EXAMPLE 51
Synthesis of 3,5-dimethoxy-4-isopropoxyphenylboronic acid
1,3-Dimethoxy-5-iodo-2-isopropoxybenzene (2.25 g) was treated in the same manner as described in Preparation Example 27 to give the title compound.
Yield: 1.23 g (74%).
PREPARATION EXAMPLE 52
Synthesis of ethyl 2-(3,5-dimethoxy-4-isopropoxyphenyl)isonicotinate
To a solution of 3,5-dimethoxy-4-isopropoxyphenylboronic acid (1.23 g) and ethyl 2-chloroisonicotinate (0.95 g) were condensed in the same manner as described in Preparation Example 1 to give the title compound.
Yield: 1.57 g(89%). 1H-NMR (400 MHz, CDCl3) δ: 1.33 (d, 6H, J=4.9 Hz), 1.44 (t, 3H, J=7.1 Hz), 3.95 (s, 6H), 4.42-4.49 (m, 3H), 7.29 (s, 2H), 7.75 (dd, 1H, J=4.9 Hz, 1.4 Hz), 8.24 (s, 1H), 8.80 (d, 1H, J=4.9 Hz).
PREPARATION EXAMPLE 53
Synthesis of 2-(3,5-dimethoxy-4-isopropoxyphenyl)-4-hydroxymethylpyridine
Ethyl 2-(3,5-dimethoxy -4-isopropoxyphenyl)isonicotinate (1.57 g) was treated in the same manner as described in the Preparation Example 2 to give the title compound.
Yield: 1.27 g (92%). 1H-NMR (400 MHz, CDCl3) δ: 1.32 (d, 6H, J=6.1 Hz), 3.93 (s, 6H), 4.45 (quint, 1H, J=6.1 Hz), 4.81 (s, 2H), 7.20 (d, 1H, J=5.1 Hz), 7.23 (s, 2H), 7.68 (s, 1H), 8.62 (d, 1H, J=5.1 Hz).
PREPARATION EXAMPLE 54
Synthesis of 4-chloromethyl-2-(3,5-dimethoxy-4-isopropoxyphenyl)pyridine
2-(3,5-Dimethoxy-4-isopropoxyphenyl)-4-hydroxymethylpyridine (1.49 g) was treated in the same manner as described in Preparation Example 3 to give the title compound.
Yield: 1.33 g (84%). 1H-NMR (400 MHz, CDCl3) δ: 1.32 (d, 6H, J=6.2 Hz), 3.94 (s, 6H), 4.45 (quint, 1H, J=6.1 Hz), 4.61 (s, 2H), 7.23-7.26 (m, 3H), 7.69 (s, 1H), 8.66 (d, 1H, J=5.1 Hz).
PREPARATION EXAMPLE 55
Synthesis of 1-[[2-(3,5-dimethoxy-4-isopropoxyphenyl)pyridin-4-yl]methyl]-4-piperidone ethylene ketal
4-Chloromethyl-2-(3,5-dimethoxy-4-isopropoxyphenyl)pyridine (643 mg) and 4-piperidone ethylene ketal (287 mg) were coupled in the same manner as described in Example 2 to give the title compound.
Yield: 818 mg (95%). 1H-NMR (400 MHz, CDCl3) δ: 1.32 (d, 6H, J=6.1 Hz), 1.78 (t, 4H, J=5.7 Hz), 2,57 (br, 4H), 3.49 (s, 4H), 3.59 (s, 2H), 3.94 (s, 6H), 4.44 (quint, 1H, J=6.1 Hz), 7.21 (d, 1H, J=5.1 Hz), 7.23 (s, 2H), 7.65 (s, 1H), 8.59 (d, 1H, J=5.1 Hz).
PREPARATION EXAMPLE 56
Synthesis of 1-[[2-(3,5-dimethoxy-4-isopropoxyphenyl)pyridin-4-yl]methyl]-4-piperidone
1-[[2-(3,5-Dimethoxy-4-isopropoxyphenyl)pyridin-4-yl]methyl]-4-piperidone ethylene ketal (818 mg) was treated in the same manner as described in Preparation Example 23 to give the title compound.
Yield: 717 mg (98%). 1H-NMR (400 MHz, CDCl3) δ: 1.32 (d, 6H, J=6.2 Hz), 2.50 (t, 4H, J=6.1 Hz), 2.81 (t, 4H, J=6.1 Hz), 3.69 (s, 2H), 3.95 (s, 6H), 4.45 (quint, 1H, J=6.2 Hz), 7.24 (s, 2H), 7.25-7.27 (m, 1H), 7.68 (s, 1H), 8.63 (d, 1H, J=5.1 Hz).
PREPARATION EXAMPLE 57
Synthesis of 4-(p-anisidino)-1-[[2-(3,5-dimethoxy-4-isopropoxyphenyl)pyridin -4-yl]methyl]piperidine
1-[[2-(3,5-dimethoxy-4-isopropoxyphenyl)pyridin-4-yl]methyl]-4-piperidone (350 mg) and p-anisidine (123 mg) were condensed in the same manner as described in Preparation Example 37 to give the title compound.
Yield: 307 mg (69%). 1H-NMR (400 MHz, CDCl3) δ: 1.32 (d, 6H, J=6.3 Hz), 1.46-1.52 (m, 2H), 2.00-2.24 (m, 2H), 2.22 (t, 2H, J=11.1 Hz), 2.86 (d, 2H, J=12.1 Hz), 3.18-3.28 (m, 1H), 3.58 (s, 2H), 3.74 (s, 3H), 3.94 (s, 6H), 4.40 (quint, 1H, J=6.3 Hz), 6.58 (d, 2H, J=6.6 Hz), 6.78 (d, 2H, J=6.6 Hz), 7.20 (d, 1H, J=5.1 Hz), 7.24 (s, 2H), 7.64 (s, 1H), 8.59 (d, 1H, J=5.1 Hz).
EXAMPLE 14
Synthesis of 1-[[2-(3,5-dimethoxy-4-isopropoxyphenyl)pyridin-4-yl]methyl]-4-[N-[[2-(3,5-dimethoxy-4-isopropoxyphenyl)pyridin-4-yl]methyl]-N-(4-methoxyphenyl) amino]piperidine trihydrochloride
4-p-anisidino)-1-[[2-(3,5-dimethoxy-4-isopropoxyphenyl)pyridin-4-yl]methyl]piperidine (307 mg) and 4-chloromethyl-2-(3,5-dimethoxy-4-isopropoxyphenyl) pyridine (201 mg) were condensed in the same manner as described in Example 9. A free base obtained was converted to a trihydrochloride giving the title compound as yellow powder.
Yield: 230 mg (46%). 1H-NMR (400 MHz, CDCl3) δ: 1.31 (d, 6H, J=3.3 Hz), 1.32 (d, 6H, J=6.8 Hz), 1.70-1.92 (m, 4H), 2.10-2.20 (m, 2H), 2.92-3.01 (m, 2H), 3.56 (s, 2H), 3.73 (s, 3H), 3.85-3.95 (m, 1H), 3.90 (s, 6H), 3.93 (s, 6H), 4.39-4.49 (m, 4H), 6.73 (d, 2H, J=4.8 Hz), 6.78 (d, 2H, J=4.8 Hz), 7.14 (s, 2H), 7.15-7.20 (m, 2H), 7.23 (s, 2H), 7.58 (s, 1H), 7.60 (s, 1H), 8.53 (d, 1H, J=5.1 Hz), 8.58 (d, 1H, J=5.1 Hz).
PREPARATION EXAMPLE 58
Synthesis of 4-benzylamino-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]-methyl]piperidine
1-[[2-(3,4,5-Trimethoxyphenyl)pyridin-4-yl]methyl-4-piperidone (1.40 g) and benzylamine (0.51 g) was condensed in the same manner as described in Preparation Example 37 to give the title compound as yellow amorphous.
Yield: 1.20 g (68%). 1H-NMR (400 MHz, CDCl3) δ: 1.40-1.60 (m, 2H), 1.88-2.09 (m, 5H), 2.54 (br, 1H), 2.82-2.85 (m, 2H), 3.52 (s, 2H), 3.80 (s, 2H), 3.89 (s, 3H), 3.95 (s, 6H), 7.18-7.31 (m, 8H), 7.64 (s, 1H), 8.57 (d, 1H, J=5.1 Hz).
EXAMPLE 15
Synthesis of 4-[N-benzyl-N-[[2-(3,4,5-trimethoxypheny)pyridin-3-yl]-methyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine tetrahydrochloride
4-Benzylamino-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine (134 mg) and 3-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (114 mg) were condensed in the same manner as described in Example 9. A free base obtained was converted to a tetrahydrochloride to give the title compound as yellow powder.
Yield: 43 mg, (17%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.63 (br, 4H), 1.87 (br, 2H), 2.39 (br, 1H), 2.88 (br, 2H), 3.49 (s, 2H), 3.57 (s, 2H), 3.68 (s, 2H), 3.86 (s, 6H), 3.88 (s, 3H), 3.90 (s, 3H), 3.96 (s, 6H), 6.60 (s, 2H), 7.17 (d, 1H, J=5.1 Hz), 7.22-7.29 (m, 8H), 7.56 (s, 1H), 8.02 (d, 1H, J=8.0 Hz), 8.50 (d, 1H, J=6.4 Hz), 8.58 (d, 1H, J=5.1 Hz).
EXAMPLE 16
Synthesis of 4-[N-benzyl-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine tetrahydrochloride
4-Benzylamino-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine (230 mg) and 4-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (158 mg) were condensed by the same manner as described in Example 9. Yellow oil of a free base was converted to a tetrahydrochloride which gave the title compound as yellow powder.
Yield: 172 mg (47%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.69-1.85 (m, 4H), 1.93-1.99 (m, 2H), 2.56 (br, 1H), 2.93-3.00 (m, 2H), 3.51 (s, 2H), 3.71 (s, 2H), 3.74 (s, 2H), 3.90 (s, 6H), 3.96 (s, 6H), 7.18-7.32 (m, 9H), 7.38 (d, 2H, J=7.1 Hz), 7.59 (s, 1H), 7.68 (s, 1H), 8.56 (d, 1H, J=5.1 Hz), 8.60 (d, 1H, J=5.1 Hz).
EXAMPLE 17
Synthesis of 4-[N-benzyl-N-[3-(3,4,5-trimethoxyphenyl)benzyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine trihydrochloride
4-Benzylamino-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine (134 mg) and 3-(3,4,5-trimethoxyphenyl)benzyl chloride (114 mg) were condensed by the same manner as described in Example 9. Yellow oil of a free base was converted to a trihydrochloride which gave the title compound as yellow powder.
Yield: 47 mg (18%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.70-1.86 (m, 4H), 1.96 (br, 2H), 2.59 (br, 1H), 2.94 (br, 2H), 3.51 (s, 2H), 3.70 (s, 2H), 3.74 (s, 2H), 3.89 (s, 3H), 3.90 (s, 3H), 3.92 (s, 6H), 3.96 (s, 6H), 6.75 (s, 2H), 7.18-7.30 (m, 6H), 7.35-7.40 (m, 5H), 7.56 (s, 1H), 7.60 (s, 1H), 8.58 (d, 1H, J=5.1 Hz).
EXAMPLE 18
Synthesis of 4-[N-benzyl-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine tetrahydrochloride
4-Benzylamino-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine (134 mg) and 5-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (114 mg) were condensed by the same manner as described in Example 9. Yellow oil of a free base was converted to a tetrahydrochloride which gave the title compound as yellow powder.
Yield: 44 mg (17%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.81 (br, 4H), 1.96 (br, 2H), 2.55 (br, 1H), 2.96 (br, 2H), 3.52 (s, 2H), 3.69 (s, 4H), 3.89 (s, 6H), 3.95 (s, 6H), 3.96 (s, 6H), 7.19-7.32 (m, 8H), 7.36-7.38 (m, 2H), 7.61 (d, 2H, J=7.6 Hz), 7.69-7.73 (m, 1H), 8.59 (d, 1H, J=4.9 Hz), 8.63 (s, 1H).
EXAMPLE 19
Synthesis of 4-[N-benzyl-N-[[5-(3,4,5-trimethoxypheny)pyridin-3-yl]methyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine tetrahydrochloride
4-Benzylamino-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine (134 mg) and 3-chloromethyl-5-(3,4,5-trimethoxyphenyl)pyridine (114 mg) were condensed by the same manner as described in Example 9. Yellow oil of a free base was converted to a tetrahydrochloride which gave the title compound as yellow powder.
Yield: 26 mg (10%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.83 (br, 4H), 1.97 (br, 2H), 2.58 (br, 1H), 2.95 (br, 2H), 3.53 (s, 2H), 3.71 (s, 2H), 3.75 (s, 2H), 3.90 (s, 6H), 3.93 (s, 6H), 3.96 (s, 6H), 6.74 (s, 2H), 7.19-7.30 (m, 6H), 7.36 (d, 2H, J=6.8 Hz), 7.60 (s, 1H), 7.79 (s, 1H), 8.54 (s, 1H), 8.59 (d, 1H, J=5.1 Hz), 8.64 (s, 1H).
PREPARATION EXAMPLE 59
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-[[2-(3,4,5-trmethoxyphenyl)pyridin-4-yl]methyl]aminomethyl]piperidine
1-(tert-Butoxycarbonyl)-4-aminomethylpiperidine (200 mg) and 4-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (183 mg) were condensed in the same manner as described in Example 2 to give the title compound as yellow syrup.
Yield: 264 mg (90%). 1H-NMR (400 MHz, CDCl3) δ: 1.12-1.27 (m, 3H), 1.45 (s, 9H), 1.60 (br, 1H), 1.74 (d, 2H, J=12.9 Hz), 2.54 (d, 2H, J=6.6 Hz), 2.69 (br, 2H), 3.87 (s, 2H), 3.90 (s, 3H), 3.97 (s, 6H), 4.03-4.14 (m, 2H), 7.20 (d, 1H, J=3.9 Hz), 7.24 (s, 2H), 7.65 (s, 1H), 8.60 (d, 1H, J=4.9 Hz).
PREPARATION EXAMPLE 60
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-methyl-N-[[2-(3,4,5-trimethoxyphenyl) pyridin-4-yl]methyl]aminomethyl]piperidine
1-(tert-butoxycarbonyl)-4-[N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]aminomethyl]piperidine (264 mg) was treated in the same manner as described in Preparation Example 11 to give the title compound as yellow syrup.
Yield: 157 mg (58%). 1H-NMR (400 MHz, CDCl3) δ: 1.00-1.09 (m, 2H), 1.43 (s, 9H), 1.65-1.70 (m, 1H), 1.79 (d, 2H, J=12.7 Hz), 2.21 (d, 2H, J=7.4 Hz), 2.23 (s, 3H), 2.69 (br, 2H), 3.52 (s, 2H), 3.89 (s, 3H), 3.96 (s, 6H), 4.07-4.13 (m, 2H), 7.20 (d, 1H, J=4.9 Hz), 7.24 (s, 2H), 7.64 (s, 1H), 8.58 (d, 1H, J=5.1 Hz).
PREPARATION EXAMPLE 61
Synthesis of 4-[N-methyl-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]aminomethyl]piperidine
1-(tert-Butoxycarbonyl)-4-[N-methyl-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]aminomethyl]piperidine (152 mg) was treated in the same manner as described in Preparation Example 12 to give the title compound as yellow crystals.
Yield: 105 mg (88%). 1H-NMR (400 MHz, CDCl3) δ: 1.00-1.10 (m, 2H), 1.60-1.68 (m, 1H), 1.80 (d, 2H, J=12.5 Hz), 2.03 (br, 1H), 2.20 (d, 2H, J=8.4 Hz), 2.21 (s, 3H), 2.58 (dt, 2H, J=12.1 Hz, 2.1 Hz), 3.05 (d, 2H, J=12.1 Hz), 3.51 (s, 2H), 3.89 (s, 3H), 3.95 (s, 6H), 7.20 (d, 1H, J=5.1 Hz), 7.24 (s, 2H), 7.65 (s, 1H), 8.57 (d, 1H, J=5.9 Hz).
EXAMPLE 20
Synthesis of 4-[N-methyl-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]aminomethyl]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine dioxalate
4-[N-methyl-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino-methyl] piperidine (96 mg) and 4-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (73 mg) were condensed in the same manner as described in Example 2. The title compound was obtained as white powder after converting a free base to a dioxalate.
Yield: 109 mg (40%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.19-1.27 (m, 2H), 1.56 (br, 1H), 1.81 (d, 2H, J=11.1 Hz), 1.99-2.04 (m, 2H), 2.23 (s, 5H), 2.88 (d, 2H, J=11.1 Hz), 3.53 (s, 4H), 3.89 (s, 3H), 3.90 (s, 3H), 3.94 (s, 6H), 3.96 (s, 6H), 7.20 (br, 2H), 7.23 (s, 4H), 7.61 (s, 1H), 7.64 (s, 1H), 8.58 (d, 2H, J=4.9 Hz).
PREPARATION EXAMPLE 62
Synthesis of 4-(3,5-dimethoxyphenylamino)-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine
1-[[2-(3,4,5-Trimethoxyphenyl)pyridin-4-yl]methyl-4-piperidone (1.40 g) and 3,5-dimethoxyaniline (722 mg) were treated in the same manner as described in Preparation Example 37 to give the title compound.
Yield: 800 mg (41%). 1H-NMR (400 MHz, CDCl3) δ: 1.40-1.90 (m, 2H), 1.95-2.50 (m, 4H), 2.93 (br, 2H), 3.31 (br, 1H), 3.65 (br, 2H), 3.72 (s, 6H), 3.88 (s, 3H), 3.96 (s, 6H), 5.76 (s, 2H), 5.85 (s, 1H), 7.20-7.35 (m, 3H), 7.73 (br, 1H), 8.60 (d, 1H, J=4.9 Hz).
EXAMPLE 21
Synthesis of 4-[N-(3,5-dimethoxyphenyl)-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-3-yl]methyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine trihydrochloride
4-(3,5-Dimethoxyphenylamino)-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine (148 mg) and 3-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (114 mg) were condensed in the same manner as described in Example 9. Yellow syrup obtained was converted to a trihydrochloroide to give the title compound as yellow powder.
Yield: 29 mg, (11%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.60-1.63 (m, 2H), 1.79 (d, 2H, J=11.7 Hz), 2.13 (t, 2H, J=11.4 Hz), 2.94 (d, 2H, J=11.3 Hz), 3.54 (s, 2H), 3.71 (s, 6H), 3.78-3.84 (m, 1H), 3.90 (s, 3H), 3.91 (s, 6H), 3.92 (s, 3H), 3.96 (s, 6H), 4.41 (s, 2H), 5.84 (s, 2H), 6.72 (s, 2H), 7.09-7.24 (m, 5H), 7.53 (s, 1H), 7.71 (d, 1H, J=6.6 Hz), 8.51 (dd, 1H, J=4.7 Hz, 1.6 Hz), 8.59 (d, 1H, J=4.9 Hz).
PREPARATION EXAMPLE 63
Synthesis of ethyl 2-(3,4,5-trimethoxyphenyl)benzoate
3,4,5-Trimethoxyphenylboronic acid (639 mg) and ethyl 2-bromobenzoate (479 mg) were condensed in the same manner as described in Preparation Example 1 to give the title compound.
Yield: 655 mg (69%). 1H-NMR (400 MHz, CDCl3) δ: 1.04 (t, 3H, J=7.2 Hz), 3.86 (s, 6H), 3.89 (s, 3H), 4.12 (q, 2H, J=7.2 Hz), 6.54 (s, 2H), 7.40-7.42 (m, 2H), 7.51 (t, 1H, J=7.8 Hz), 7.77 (d, 1H, J=6.8 Hz).
PREPARATION EXAMPLE 64
Synthesis of 2-(3,4,5-trimethoxyphenyl)benzyl alcohol
Ethyl 2-(3,4,5-trimethoxyphenyl)benzoate (655 mg) was treated in the same manner as described in Preparation Example 2 to give the title compound.
Yield: 630 mg (theoretical amount). 1H-NMR (400 MHz, CDCl3) δ: 3.85 (s, 6H), 3.90 (s, 3H), 4.61 (s, 2H), 6.61 (s, 2H), 7.26-7.39 (m, 3H), 7.53 (d, 1H, J=6.8 Hz).
PREPARATION EXAMPLE 65
Synthesis of 2-(3,4,5-trimethoxyphneyl)benzyl chloride
2-(3,4,5-Trimethoxyphenyl)benzyl alcohol (630 mg) was treated in the same manner as described in Preparation Example 3 to give the title compound.
Yield: 615 mg (theoretical amount). 1H-NMR (400 MHz, CDCl3) δ: 3.87 (s, 6H), 3.90 (s, 3H), 4.53 (s, 2H), 6.66 (s, 2H), 7.29-7.32 (m, 1H), 7.34-7.39 (m, 2H), 7.50-7.52 (m, 1H).
EXAMPLE 22
Synthesis of 4-[N-(3,5-dimethoxyphenyl)-N-[2-(3,4,5-trimethoxyphenyl)benzyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine dihydrochloride
4-(3,5-Dimethoxyphenylamino)-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine (148 mg) and 2-(3,4,5-trimethoxyphenyl)benzyl chloride (114 mg) were condensed in the same manner as described in Example 9. A free base obtained was converted to a dihydrochloroide to give the title compound as yellow powder.
Yield: 20 mg, (8%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.50-1.90 (m, 4H), 2.05-2.20 (m, 2H), 2.92 (br, 2H), 3.52 (br, 3H), 3.68 (s, 6H), 3.85 (s, 6H), 3.88 (s, 3H), 3.89 (s, 3H), 3.94 (s, 6H), 4.31 (s, 2H), 5.85 (br, 3H), 6.52 (s, 2H), 7.05-7.27 (m, 6H), 7.34 (s, 1H), 7.51 (s, 1H), 8.56 (s, 1H).
EXAMPLE 23
Synthesis of 4-[N-(3,5-dimethoxyphenyl)-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine trihydrochloride
4-(3,5-Dimethoxyphenylamino)-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine (148 mg) and 4-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (114 mg) were condensed by the same manner as described in Example 9. Yellow oil of a free base was converted to a trihydrochloride which gave the title compound as yellow powder.
Yield: 40 mg (18%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.68-1.90 (m, 4H), 2.12-2.22 (m, 2H), 2.94-3.02 (m, 2H), 3.57 (s, 2H), 3.71 (s, 6H), 3.81-3.83 (m, 1H), 3.89 (s, 3H), 3.90 (s, 3H), 3.93 (s, 6H), 3.96 (s, 6H), 4.52 (s, 2H), 5.89-5.94 (m, 3H), 7.14 (d, 1H, J=5.3 Hz), 7.16 (s, 2H), 7.20 (d, 1H, J=3.7 Hz), 7.22 (s, 2H), 7.54-7.60 (m, 2H), 8.55 (d, 1H, J=5.1 Hz), 8.59 (d, 1H, J=5.1 Hz).
EXAMPLE 24
Synthesis of 4-[N-(3,5-dimethoxyphenyl)-N-[3-(3,4,5-trimethoxyphenyl)benzyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine dihydrochloride
4-(3,5-Dimethoxyphenylamino)-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine (148 mg) and 3-(3,4,5-trimethoxyphenyl)benzyl chloride (114 mg) were condensed by the same manner as described in Example 9. Yellow oil of a free base was converted to a dihydrochloride which gave the title compound as yellow powder.
Yield: 41 mg (16%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.78-1.88 (m, 4H), 2.16 (t, 2H, J=10.7 Hz), 2.96 (d, 2H, J=11.3 Hz), 3.56 (s, 2H), 3.70 (s, 6H), 3.73-3.84 (m, 1H), 3.87 (s, 3H), 3.89 (s, 6H), 3.90 (s, 3H), 3.95 (s, 6H), 4.54 (s, 2H), 5.95 (s, 2H), 6.71 (s, 2H), 7.19-7.26 (m, 4H), 7.31-7.39 (m, 3H), 7.42 (s, 1H), 7.59 (s, 1H), 8.58 (d, 1H, J=4.9 Hz).
EXAMPLE 25
Synthesis of 4-[N-(3,5-dimethoxyphenyl)-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine trihydrochloride
4-(3,5-Dimethoxyphenylamino)-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine (148 mg) and 5-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (114 mg) were condensed by the same manner as described in Example 9. Yellow oil of a free base was converted to a trihydrochloride which gave the title compound as yellow powder.
Yield: 23 mg (10%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.64 (br, 2H), 1.82 (br, 2H), 2.10 (br, 2H), 2.94 (br, 2H), 3.48-3.60 (m, 3H), 3.64 (s, 6H), 3.82 (s, 3H), 3.83 (s, 3H), 3.87 (s, 6H), 3.90 (s, 6H), 4.46 (s, 2H), 5.85 (br, 3H), 7.05-7.24 (m, 6H), 7.53-7.54 (m, 2H), 8.51 (s, 1H), 8.54 (br, 1H).
PREPARATION EXAMPLE 66
Synthesis of ethyl 4-(3,4,5-trimethoxyphenyl)benzoate
3,4,5-Trimethoxyphenylboronic acid (2.01 g) and ethyl 4-bromobenzoate (2.29 g) were condensed in the same manner as described in Preparation Example 1 to give the title compound.
Yield: 2.99 g (95%). 1H-NMR (400 MHz, CDCl3) δ: 1.42 (t, 3H, J=7.2 Hz), 3.90 (s, 3H), 3.94 (s, 6H), 4.38 (q, 2H, J=7.2 Hz), 6.81 (s, 2H), 7.62 (d, 2H, J=8.2 Hz), 8.10 (d, 2H, J=8.2 Hz).
PREPARATION EXAMPLE 67
Synthesis of 4-(3,4,5-trimethoxyphenyl)benzyl alcohol
Ethyl 4-(3,4,5-trimethoxyphenyl)benzoate (2.99 g) was treated in the same manner as described in Preparation Example 2 to give the title compound.
Yield: 1.83 g (71%)
PREPARATION EXAMPLE 68
Synthesis of 4-(3,4,5-trimethoxyphenyl)benzyl chloride
4-(3,4,5-Trimethoxyphenyl)benzyl alcohol (1.83 g) was treated in the same manner as describe in Preparation Example 3 to give the title compound.
Yield: 1.65 g (84%) 1H-NMR (400 MHz, CDCl3) δ: 3.90 (s, 3H), 3.93 (s, 6H), 4.65 (s, 2H), 6.77 (s, 2H), 7.46 (d, 2H, J=8.0 Hz), 7.55 (d, 2H, J=8.0 Hz).
EXAMPLE 26
Synthesis of 4-[N-(3,5-dimethoxyphenyl)-N-[4-(3,4,5-trimethoxypheny)benzyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine dihydrochloride
4-(3,5-Dimethoxyphenylamino)-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine (148 mg) and 4-(3,4,5-trimethoxyphenyl)benzyl chloride (114 mg) were condensed by the same manner as described in Example 9. Yellow oil of a free base was converted to a dihydrochloride which gave yellow powder of the title compound.
Yield: 35 mg (14%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ; 1.80-1.89 (m, 4H), 2.17 (br, 2H), 2.97 (d, 2H, J=10.5 Hz), 3.57 (s, 2H), 3.70 (s, 6H), 3.77-3.84 (m, 1H), 3.87 (s, 3H), 3.90 (s, 3H), 3.91 (s, 6H), 3.96 (s, 6H), 4.52 (s, 2H), 5.93 (s, 2H), 6.74 (s, 2H), 7.19-7.22 (m, 4H), 7.31 (d, 2H, J=8.2 Hz), 7.46 (d, 2H, J=8.2 Hz), 7.60 (s, 1H), 8.59 (d, 1H, J=5.1 Hz).
PREPARATION EXAMPLE 69
Synthesis of 4-(3,4-methylenedioxyphenylamino)-1-[[2-(3,4,5-trimethoxyphenyl) pyridin-4-yl]methyl]piperidine
1-[[2-(3,4,5-Trimethoxyphenyl)pyridin-4-yl]methyl-4-piperidone (1.40 g) and 3,4-methylenedioxyaniline (646 mg) were treated in the same manner as described in Preparation Example 29 to give the title compound.
Yield: 810 mg (43%). 1H-NMR (400 MHz, CDCl3) δ: 1.63 (br, 2H), 2.02-2.60 (m, 4H), 2.80-3.15 (m, 2H), 3.25 (br, 1H), 3.70 (br, 2H), 3.88 (s, 3H), 3.96 (s, 6H), 5.83 (s, 2H), 6.02 (d, 1H, J=8.3 Hz), 6.22 (s, 1H), 6.61 (d, 1H, J=8.3 Hz), 7.18-7.28 (m, 3H), 7.64 (br, 1H), 8.60 (d, 1H, J=4.9 Hz).
EXAMPLE 27
Synthesis of 4-[N-(3,4-methylenedioxyphenyl)-N-[[2-(3,4,5-trimethoxypheny) pyridin-3-yl]methyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine trihydrochloride
4-(3,4-Methylenedioxyphenylamino)-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine (119 mg) and 3-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (114 mg) were condensed in the same manner as described in Example 9. Yellow syrup obtained was converted to a trihydrochloroide to give the title compound as yellow powder.
Yield: 30 mg (14%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.45-2.25 (m, 6H), 2.90 (br, 2H), 3.40 (br, 1H), 3.55 (br, 2H), 3.87 (s, 3H), 3.88 (s, 9H), 3.93 (s, 6H), 4.28 (s, 2H), 5.82 (s, 2H), 6.10 (br, 1H), 6.28 (s, 1H), 6.58 (d, 1H, J=8.4 Hz), 6.67 (s, 2H), 7.12-7.30 (m, 4H), 7.52 (br, 1H), 7.75 (br, 1H), 8.51 (br, 1H), 8.57 (br, 1H).
EXAMPLE 28
Synthesis of 4-[N-(3,4-methylenedioxyphenyl)-N-[2-(3,4,5-trimethoxyphenyl) benzyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]metyl]piperidine dihydrochloride
4-(3,4-Methylenedioxyphenylamino)-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine (119 mg) and 2-(3,4,5-trimethoxyphenyl)benzyl chloride (114 mg) were condensed in the same manner as described in Example 9. A free base obtained was converted to a dihydrochloroide to give the title compound as yellow powder.
Yield: 13 mg (6%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.61 (br, 2H), 1.78 (br, 2H), 2.10 (br, 2H), 2.91 (br, 2H), 3.50-3.54 (m, 3H), 3.87 (s, 6H), 3.90 (s, 3H), 3.92 (s, 3H), 3.99 (s, 6H), 4.26 (s, 2H), 5.82 (s, 2H), 6.12 (d, 1H, J=8.6 Hz), 6.32 (s, 1H), 6.53 (s, 2H), 6.62 (d, 1H, J=8.6 Hz), 7.17-7.26 (m, 6H), 7.42 (br, 1H), 7.55 (s, 1H), 8.58 (d, 1H, J=4.9 Hz).
EXAMPLE 29
Synthesis of 4-[N-(3,4-methylenedioxyphenyl)-N-[[2-(3,4,5-trimethoxyphenyl) pyridin-4-yl]methyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine trihydrochloride
4-(3,4-Methylenedioxyphenylamino)-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine (119 mg) and 4-chloromethyl-2-(3,4,5-trimethoxyphenyl) pyridine (114 mg) were condensed by the same manner as described in Example 9. Yellow oil of a free base was converted to a trihydrochloride which gave the title compound as yellow powder.
Yield: 52 mg (25%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.60-1.95 (m, 4H), 2.20 (br, 2H), 3.00 (br, 2H), 3.58 (br, 3H), 3.86 (s, 3H), 3.87 (s, 3H), 3.91 (s, 6H), 3.94 (s, 6H), 4.41 (s, 2H), 5.82 (s, 2H), 6.17 (d, 1H, J=8.4 Hz), 6.39 (s, 1H), 6.62 (d, 1H, J=8.4 Hz), 7.12-7.13 (m, 3H), 7.18 (d, 1H, J=4.1 Hz), 7.23 (br, 2H), 7.54 (br, 2H), 8.51 (d, 1H, J=5.1 Hz), 8.57 (d, 1H, J=4.9 Hz).
EXAMPLE 30
Synthesis of 4-[N-(3,4-methylenedioxyphenyl)-N-[3-(3,4,5-trimethoxyphenyl) benzyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine dihydrochloride
4-(3,4-Methylenedioxyphenylamino)-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine (119 mg) and 3-(3,4,5-trimethoxyphenyl)benzyl chloride (114 mg) were condensed by the same manner as described in Example 9. Yellow oil of a free base was converted to a dihydrochloride which gave the title compound as yellow powder.
Yield: 58 mg (29%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.60-1.97 (m, 4H), 2.15 (br, 2H), 3.00 (br, 2H), 3.58 (br, 3H), 3.86 (s, 3H), 3.88 (s, 9H), 3.94 (s, 6H), 4.43 (s, 2H), 5.81 (s, 2H), 6.21 (br, 1H), 6.42 (s, 1H), 6.62 (d, 1H, J=8.4 Hz), 6.69 (s, 2H), 7.18 (d, 1H, J=4.9 Hz), 7.22-7.39 (m, 6H), 7.60 (br, 1H), 8.57 (d, 1H, J=4.9 Hz).
EXAMPLE 31
Synthesis of 4-[N-(3,4-methylenedioxyphenyl)-N-[[2-(3,4,5-trimethoxyphenyl) pyridin-5-yl]methyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine trihydrochloride
4-(3,4-Methylenedioxyphenylamino)-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine (119 mg) and 5-chloromethyl-2-(3,4,5-trimethoxyphenyl) pyridine (114 mg) were condensed by the same manner as described in Example 9. Yellow oil of a free base was converted to a trihydrochloride which gave the title compound as yellow powder.
Yield: 69 mg (27%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.71-1.88 (m, 4H), 2.14 (d, 2H, J=11.2 Hz), 2.97 (d, 2H, J=11.5 Hz), 3.45-3.52 (m, 1H), 3.56 (s, 2H), 3.89 (s, 3H), 3.90 (s, 3H), 3.94 (s, 6H), 3.96 (s, 6H), 4.12 (s, 2H), 5.85 (s, 2H), 6.24 (dd, 1H, J=8.5 Hz, 2.5 Hz), 6.45 (d, 1H, J=2.4 Hz), 6.64 (d, 1H, J=8.5 Hz), 7.20-7.21 (m, 1H), 7.21 (s, 2H), 7.23 (s, 2H), 7.58-7.65 (m, 3H), 8.57 (d, 1H, J=1.5 Hz), 8.59 (d, 1H, J=4.9 Hz).
EXAMPLE 32
Synthesis of 4-[N-(3,4-Methylenedioxyphenyl)-N-[4-(3,4,5-trimethoxyphenyl) benzyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine dihydrochloride
4-(3,4-Methylenedioxyphenylamino)-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine (119 mg) and 4-(3,4,5-trimethoxyphenyl)benzyl chloride (114 mg) were condensed by the same manner as described in Example 9. Yellow oil of a free base was converted to a dihydrochloride which gave the title compound as yellow powder.
Yield: 29 mg (14%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ; 1.62-2.00 (m, 4H), 2.20 (br, 2H), 2.99 (br, 2H), 3.58 (br, 3H), 3.86 (s, 3H), 3.87 (s, 3H), 3.88 (s, 6H), 3.89 (s, 6H), 4.41 (s, 2H), 5.82 (s, 2H), 6.19 (d, 1H, J=8.6 Hz), 6.39 (s, 1H), 6.63 (d, 1H, J=8.4 Hz), 6.72 (s, 2H), 7.18 (d, 1H, J=5.1 Hz), 7.23 (s, 2H), 7.29 (d, 2H, J=8.0 Hz), 7.43 (d, 2H, J=8.2 Hz), 7.60 (br, 1H), 8.57 (d, 1H, J=4.9 Hz).
PREPARATION EXAMPLE 70
Synthesis of 4-[N-methyl-N-[(2-nitrobenzene)sulfonyl]aminomethyl]-2-(3,4,5-trimethoxyphenyl)pyridine
4-Chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (232 mg), N-methyl-2-nitrobenzenesulfonamide (171 mg) and potassium carbonate (138 mg) were suspended in acetonitrile (10 mL). The mixture was stirred at room temperature overnight and evaporated. To the residue was added chloroform and water. The organic layer was separated, washed with saturated aqueous sodium hydrogencarbonate and brine, dried over anhydrous magnesium sulfate and evaporated to give the title compound.
Yield: 362 mg (97.0%).
PREPARATION EXAMPLE 71
Synthesis of 4-(methylaminomethyl)-2-(3,4,5-trimethoxyphenyl)pyridine
To a suspension of 4-[N-methyl-N-[(2-nitrobenzene)sulfonyl]aminomethyl]-2-(3,4,5-trimethoxyphenyl)pyridine (691 mg) and potassium carbonate (203 mg) in acetonitrile (20 mL) was added thiophenol (228 μL). The mixture was stirred at 50° C. overnight and evaporated. To the residue was added chloroform and water. The organic layer was separated, washed with saturated aqueous sodium hydrogencarbonate and brine, dried over anhydrous magnesium sulfate and evaporated. The residue was subjected to a column of silica gel using chloroform-methanol (40:1) and then chloroform-methanol (10:1) as eluents. Fractions containing the product were collected and evaporated to give the title compound.
Yield: 356 mg (84%).
EXAMPLE 33
Synthesis of 4-[N-methyl-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]aminocarbonyl]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine maleate
1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine-4-caroxylic acid (98 mg) and 4-(methylaminomethyl)-2-(3,4,5-trimethoxyphenyl)pyridine (73 mg) were condensed by the same manner as described in Example 1 giving a maleate of the title compound as white powder.
Yield: 145 mg (75%). 1H-NMR (400 MHz, measured as a maleate, DMSO-d6) δ: 1.89-1.97 (m, 4H), 2.75-2.96 (m, 3H), 3.03 (s, 3H), 3.27 (d, 2H, J=12.0 Hz), 3.78 (s, 3H), 3.79 (s, 3H), 3.87 (s, 6H), 3.90 (s, 6H), 4.09 (s, 2H), 4.64 (s, 2H), 6.14 (s, 2H), 7.09 (d, 1H, J=5.0 Hz), 7.33 (s, 2H), 7.37 (d, 1H, J=5.0 Hz), 7.38 (s, 2H), 7.65 (s, 1H), 7.90 (s, 1H), 8.57 (d, 1H, J=5.0 Hz), 8.67 (d, 1H, J=5.0 Hz).
PREPARATION EXAMPLE 72
Synthesis of (3S)-1-(tert-butoxycarbonyl)-3-[N-[(2-nitrobenzene)sulfonyl]-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]pyrrolidine
(3S)-1-(tert-Butoxycarbonyl)-3-[(2-nitrobenzene)sulfonylamino]pyrrolidine (72 mg) and 4-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (57 mg) were condensed in the same manner as described in Example 2 to give colorless amorphous of the title compound.
Yield:103 mg (85%).
PREPARATION EXAMPLE 73
Synthesis of (3S)-3-[N-[(2-nitrobenzene)sulfonyl]-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]pyrrolidine
(3S)-1-(tert-butoxycarbonyl)-3-[N-[(2-nitrobenzene)sulfonyl]-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]pyrrolidine (103 mg) was treated in the same manner as described in Preparation Example 12 to give yellow amorphous of the title compound.
Yield: 72 mg (84%). 1H-NMR (400 MHz, CDCl3) δ: 1.66-1.75 (m, 1H), 2.03-2.05 (m, 1H), 2.78-2.85 (m, 2H), 3.00-3.10 (m, 2H), 3.39 (br, 1H), 3.90 (s, 3H), 3.96 (s, 6H), 4.59-4.67 (m, 1H), 4.70 (s, 2H), 7.13-7.18 (m, 1H), 7.20 (s, 2H), 7.52-7.64 (m, 4H), 7.95 (dd, 1H, J=7.9 Hz, 1.1 Hz), 8.52 (d, 1H, J=5.1 Hz).
PREPARATION EXAMPLE 74
Synthesis of (3S)-3-[N-[(2-nitrobenzene)sulfonyl]-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]pyrrolidine
(3S)-3-[N-[(2-Nitrobenzene)sulfonyl]-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]pyrrolidine (72 mg) and 4-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (40 mg) were treated in the same manner as described in Example 2 to give a yellow amorphous of the title compound.
Yield: 97 mg (91%). 1H-NMR (400 MHz, CDCl3) δ: 1.59 (br, 1H), 1.80-1.90 (m, 1H), 2.20-2.30 (m, 2H), 2.55 (dd, 1H, J=10.5 Hz, 8.2 Hz), 2.78 (dd, 1H, J=10.6 Hz, 3.2 Hz), 2.87 (t, 1H, J=7.2 Hz), 3.50 (d, 1H, J=13.7 Hz), 3.64 (d, 1H, J=13.7 Hz), 3.89 (s, 3H), 3.90 (s, 3H), 3.92 (s, 6H), 3.93 (s, 6H), 4.83 (d, 2H, J=4.5 Hz), 7.07 (d, 1H, J=5.1 Hz), 7.10 (d, 1H, J=4.9 Hz), 7.15 (s, 2H), 7.17 (s, 2H), 7.41-7.45 (m, 1H), 7.50-7.55 (m, 3H), 7.61 (s, 1H), 7.81 (d, 1H, J=7.4 Hz), 8.45 (d, 1H, J=4.9 Hz), 8.51(d, 1H, J=5.1 Hz).
EXAMPLE 34
Synthesis of (3S)-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]-3-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methylamino]pyrrolidine trihydrochloride
(3S)-3-[N-[(2-nitrobenzene)sulfonyl]-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]pyrrolidine (97 mg) was treated in the same manner as described in Preparation Example 11 to give yellow amorphous of the title compound, which was converted to a trihydrochloride.
Yield: 80 mg (89%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.71(br, 2H), 2.19-2.21 (m, 1H), 2.52-2.55 (m, 2H), 2.73-2.77 (m, 2H), 3.39 (br, 1H), 3.66 (d, 1H, J=13.7 Hz), 3.71 (d, 1H, J=13.7 Hz), 3.82 (s, 2H), 3.90 (s, 6H), 3.95 (s, 12H), 7.18-7.21 (m, 2H), 7.23 (s, 2H), 7.24 (s, 2H), 7.63 (s, 2H), 8.59 (d, 1H, J=4.3 Hz), 8.60 (d, 1H, J=4.3 Hz).
EXAMPLE 35
Synthesis of 4-[3-(3,4,5-trimethoxyphenyl)benzoylamino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine maleate
3-(3,4,5-trimethoxyphenyl)benzoic acid (69 mg) and 4-amino-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine (114 mg) were condensed in the same manner as described in Example 1. The title compound was obtained after converting the product to a maleate.
Yield: 100 mg (56%). 1H-NMR (400 MHz, measured as a maleate, DMSO-d6) δ: 1.85-2.10 (m, 4H), 2.77-2.93 (m, 2H), 3.20-3.31 (m, 2H), 3.77 (s, 3H), 3.79(s, 3H), 3.89 (s, 6H), 3.91 (s, 6H), 3.984.07 (m, 1H), 4.13 (s, 2H), 6.15 (s, 2H), 6.94 (s, 2H), 7.40-7.52 (m, 4H), 7.73-7.80 (m, 2H), 8.02-8.10 (m, 3H), 8.67-8.68 (m, 1H).
EXAMPLE 36
Synthesis of 4-[N-methyl-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine tetrahydrochloride
4-(methylamino)-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine (2.67 g) and 4-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (2.12 g) were condensed in the same manner as described in Example 2. The title compound was obtained after converting a free base to a tetrahydrochloride.
Yield: 2.55 g (46%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.66-1.74 (m, 2H), 1.82 (d, 2H, J=10.7 Hz), 2.04 (t, 2H, J=11.0 Hz), 2.25 (s, 3H), 2.45-2.51 (m, 1H), 2.98 (d, 2H, J=11.7 Hz), 3.55 (s, 2H), 3.66 (s, 2H), 3.90 (s, 3H), 3.91 (s, 3H), 3.96 (s, 6H), 3.97 (s, 6H), 7.21-7.23 (m, 2H), 7.24 (s, 2H), 7.25 (s, 2H), 7.62 (s, 1H), 7.63 (s, 1H), 8.59 (d, 1H, J=5.1 Hz), 8.60 (d, 1H, J=5.3 Hz).
PREPARATION EXAMPLE 75
Synthesis of 1-(ethoxycarbonyl)-4-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methylamino]piperidine
4-Amino-1-(ethoxycarbonyl)piperidine (341 mg) and 4-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (300 mg) were condensed in the same manner as described in Example 2 to give the title compound.
Yield: 438 mg (theoretical yield). 1H-NMR (400 MHz, CDCl3) δ: 1.25 (t, 3H, J=7.1 Hz), 1.27-1.34 (m, 2H), 1.60 (br, 1H), 1.90 (d, 2H, J=10.9 Hz), 2.67-2.72 (m, 1H), 2.87 (t, 2H, J=11.5 Hz), 3.90 (s, 3H), 3.91 (br, 2H), 3.96 (s, 6H), 4.09 (br, 2H), 4.12 (q, 2H, J=7.0 Hz), 7.21 (d, 1H, J=3.5 Hz), 7.24 (s, 2H), 7.65 (s, 1H), 8.59 (d, 1H, J=4.9 Hz).
PREPARATION EXAMPLE 76
Synthesis of 1-(ethoxycarbonyl)-4-[N-methyl-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]piperidine
To a solution of 1-(ethoxycarbonyl)-4-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methylamino]piperidine (438 mg) was treated in the same manner as described in Preparation Example 11 to give the title compound as yellow syrup.
Yield: 235 mg (52%). 1H-NMR (400 MHz, CDCl3) δ: 1.26 (t, 3H, J=7.1 Hz), 1.42-1.57 (m, 2H), 1.82 (d, 2H, J=11.9 Hz), 2.24 (s, 3H), 2.59-2.65 (m, 1H), 2.75 (t, 2H, J=12.0 Hz), 3.65 (s, 2H), 3.90 (s, 3H), 3.97 (s, 6H), 4.13 (q, 2H, J=7.0 Hz), 4.23 (br, 2H), 7.22 (dd, 1H, J=5.0 Hz, 1.3 Hz), 7.24 (s, 2H), 7.63 (s, 1H), 8.59 (d, 1H, J=4.5 Hz).
PREPARATION EXAMPLE 77
Synthesis of 4-[N-methyl-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]piperidine
To a solution of 1-(ethoxycarbonyl)-4-[N-methyl-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]piperidine (100 mg) in ethanol (2 mL) was added 4 M sodium hydroxide (8 mL). The mixture was refluxed overnight and extracted with chloroform. The organic layer was washed with water and brine, dried over anhydrous sodium sulfate and evaporated. The residue was subjected to a column of silica gel and liquid chromatography was performed using chloroform-methanol (20:1) to give the title compound as yellow syrup.
Yield: 73 mg (88%). 1H-NMR (400 MHz, CDCl3) δ: 1.50-1.55 (m, 2H), 1.84 (d, 2H, J=12.0 Hz), 1.99 (br, 1H), 2.25 (s, 3H), 2.55-2.63 (m, 3H), 3.16 (d, 2H, J=12.2 Hz), 3.65 (s, 2H), 3.90 (s, 3H), 3.97 (s, 6H), 7.22 (d, 1H, J=6.1 Hz), 7.24 (s, 2H), 7.64 (s, 1H), 8.58 (d, 1H, J=5.1 Hz).
EXAMPLE 37
Synthesis of 4-[N-methyl-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine tetrahydrochloride
4-[N-methyl-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]piperidine (73 mg) and 4-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (58 mg) were condensed in the same manner as described in Example 2. The title compound was obtained after converting a free base to a tetrahydrochloride.
Yield: 126 mg (84%).
EXAMPLE 38
Synthesis of 4-[N-methyl-N-[3-(3,4,5-trimethoxyphenyl)benzyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine difumarate
4-(methylamino)-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine (111 mg) and 3-(3,4,5-trimethoxyphenyl)benzyl chloride (88 mg) were condensed in the same manner as described in Example 2. The title compound was obtained as white powder after converting a free base to a difumarate.
Yield: 59 mg (46%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.70-1.77 (m, 2H), 1.85-1.87 (m, 2H), 2.03-2.08 (m, 2H), 2.27 (s, 3H), 2.55-2.59 (m, 1H), 2.98 (d, 2H, J=11.3 Hz), 3.56 (s, 2H), 3.69 (s, 2H), 3.89 (s, 3H), 3.90 (s, 3H), 3.93 (s, 6H), 3.98 (s, 6H), 6.79 (s, 2H), 7.22 (d, 1H, J=4.9 Hz), 7.28 (s, 2H), 7.31 (d, 1H, J=7.6 Hz), 7.38 (t, 1H, J=7.4 Hz), 7.45 (d, 1H, J=7.6 Hz), 7.51 (s, 1H), 7.63 (s, 1H), 8.60 (d. 1H, J=5.1 Hz).
EXAMPLE 39
Synthesis of 4-[N-methyl-N-[[2-(3,5-dimethoxy-4-hydroxyphenyl)pyridin-4-yl]methyl]amino]-1-[[2-(3,5-dimethoxy-4-hydroxyphenyl)pyridin-4-yl]methyl]piperidine tetrahydrochloride
To an ice-cooled solution of 4-[N-methyl-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine (100 mg) in dichloromethane (5 mL) was added iodotrimethylsilane (173 μL). The mixture was stirred at 0° C. for 2 hours and then at room temperature overnight. A small amount of water, ethyl acetate and saturated aqueous sodium hydrogencarbonate were added to the mixture at 0° C. and the organic layer was separated. The organic layer was washed with brine, dried over anhydrous magnesium sulfate and evaporated. The residue was applied to a preparative TLC using chloroform-ammonia saturated methanol (15:1) to give a free base of the title compound which was converted to a tetrahydrochloride by the conventional method.
Yield: 50 mg (52.3%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.68-1.89 (m, 4H), 2.03-2.12 (m, 2H), 2.26 (s, 3H), 2.48-2.60 (m, 1H), 2.98-3.05 (m, 2H), 3.57 (s, 2H), 3.65 (s, 2H), 3.94 (s, 6H), 3.95 (s, 6H), 7.16-7.19 (m, 2H), 7.26 (s, 2H), 7.27 (s, 2H), 7.62-7.68 (m, 2H), 8.56 (d, 1H, J=5.3 Hz), 8.58 (d, 1H, J=5.2 Hz).
PREPARATION EXAMPLE 78
Synthesis of 1-(ethoxycarbonyl)-4-[N-ethyl-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]piperidine
To a solution of 1-(ethoxycarbonyl)-4-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methylamino]piperidine (400 mg) in acetonitrile (5 mL) was added potassium carbonate (13 mg) and iodoethane (145 mg). The mixture was placed in sealed vessel and stirred at 80° C. for 2 hours. After removing the solvent in vacuo, ethyl acetate was added, washed with water and brine, dried over anhydrous sodium sulfate and evaporated. The residue was subjected to a column of silica gel using chloroform-methanol (30:1) as an eluent. Fractions containing the product were collected and evaporated to give the title compound as yellow syrup.
Yield: 242 mg (57%). 1H-NMR (400 MHz, CDCl3) δ: 1.04 (t, 3H, J=7.1 Hz), 1.25 (t, 3H, J=7.1 Hz),1.43-1.52 (m, 2H), 1.79 (d, 2H, J=11.5 Hz), 2.60 (q, 2H, J=7.0 Hz), 2.66-2.76 (m, 3H), 3.70 (s, 2H), 3.90 (s, 3H), 3.97 (s, 6H), 4.12 (q, 2H, J=7.0 Hz), 4.20 (br, 2H), 7.23 (s, 2H), 7.26 (d, 1H, J=5.7 Hz), 7.67 (s, 1H), 8.58 (d, 1H, J =4.9 Hz).
PREPARATION EXAMPLE 79
Synthesis of 4-[N-ethyl-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]piperidine
1-(ethoxycarbonyl)-4-[N-ethyl-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]piperidine (242 mg) was treated in the same manner as described in Preparation Example 77 to give the title compound as yellow syrup.
Yield: 150 mg (74%). 1H-NMR (400 MHz, CDCl3) δ: 1.03 (t, 3H, J=7.0 Hz), 1.43-1.52 (m, 2H),1.70 (br, 1H), 1.79 (d, 2H, J=12.3 Hz), 2.53-2.67 (m, 5H), 3.13 (d, 2H, J=11.9 Hz), 3.71 (s, 2H), 3.90 (s, 3H), 3.97 (s, 6H), 7.24 (s, 2H), 7.27 (d, 1H, J=5.1 Hz), 7.68 (s, 1H), 8.57 (d, 1H, J=4.3 Hz).
EXAMPLE 40
Synthesis of 4-[N-ethyl-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine tetrahydrochloride
4-[N-ethyl-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]piperidine (65 mg) and 4-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (50 mg) were condensed in the same manner as described in Example 2. The title compound was obtained after converting a free base to a tetrahydrochloride.
Yield: 121 mg (90%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.03 (t, 3H, J=7.1 Hz), 1.641.69 (m, 2H), 1.77 (d, 2H, J=10.7 Hz), 2.01 (t, 2H, J=10.8 Hz), 2.55-2.64 (m, 3H), 2.95 (d, 2H, J=11.1 Hz), 3.53 (s, 2H), 3.71 (s, 2H), 3.90 (s, 6H), 3.97 (s, 12H), 7.20-7.27 (m, 6H), 7.60 (s, 1H), 7.68 (s, 1H), 8.57 (d, 1H, J=4.9 Hz), 8.59 (d, 1H, J=5.1 Hz).
PREPARATION EXAMPLE 80
Synthesis of 4-(cyclohexylamino)-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine
1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]-4-pieridone (400 mg) and cyclohexylamine (134 mg) were reacted in the same manner as described in Preparation Example 37 to give the title compound.
Yield: 342 mg (69%). 1H-NMR (400 MHz, CDCl3) δ: 1.05-1.30 (m, 6H), 1.38-1.52 (m, 2H), 1.53-1.80 (m, 3H), 1.87 (br, 4H), 2.07 (t, 2H, J=10.7 Hz), 2.59(br, 2H), 2.86 (br, 2H), 3.5 (s, 2H), 3.90 (s, 3H), 3.97 (s, 6H), 7.19 (d, 1H, J=4.9 Hz), 7.24 (s, 2H), 7.64 (s, 1H), 8.58 (d, 1H, J=4.9 Hz).
EXAMPLE 41
Synthesis of 4-[N-cyclohexyl-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine tetrahydrochloride
4-(Cyclohexylamino)-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine (342 mg) and 4-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (252 mg) were reacted in the same manner as described in Preparation Example 6. The title compound was obtained after converting the product to a tetrahydrochloride.
Yield: 55 mg (8%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.00-1.39 (m, 6H),1.58-1.88 (m, 8H), 2.07 (br, 2H), 2.61(br, 2H), 2.96 (br, 2H), 3.57 (br, 2H), 3.85 (s, 2H), 3.90 (s, 3H), 3.91 (s, 3H), 3.97 (s, 12H), 7.19-7.28 (m, 6H), 7.70 (br, 2H), 8.56 (d, 1H, J=5.1 Hz), 8.60 (d, 1H, J=5.1 Hz).
PREPARATION EXAMPLE 81
Synthesis of 4-anilino-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine
1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]-4-piperidone (1.1 g) and aniline (344 mg) were reacted in the same manner as described in Preparation Example 37 to give the title compound.
Yield: 1.09 g (81%). 1H-NMR (400 MHz, CDCl3) δ: 1.53 (br, 2H), 2.02-2.13 (m, 2H), 2.16-2.32 (m, 2H), 2.86 (br, 2H), 3.32 (br, 1H), 3.59 (s, 2H), 3.88 (s, 3H), 3.95 (s, 6H), 6.57 (d, 2H, J=8.6 Hz), 6.66 (t, 1H, J=7.3 Hz), 7.14 (t, 2H, =7.9 Hz), 7.20-7.24 (m, 5H), 7.65 (br, 1H), 8.59 (d, 1H, J=5.1 Hz).
EXAMPLE 42
Synthesis of 4-[N-phenyl-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine trihydrochloride
4-Anilino-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine (1.64 g) and 4-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (1.64 g) were reacted in the same manner as described in Preparation Example 9. The title compound was obtained after converting the product to a trihydrochloride.
Yield: 635 mg (20%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.60-2.00 (m, 4H), 2.10-2.35 (m, 2H), 2.99 (br, 2H), 3.58 (br, 3H), 3.86 (s, 3H), 3.88 (s, 3H), 3.90 (s, 6H), 3.94 (s, 6H), 4.52 (s, 2H), 6.66-6.78 (m, 3H), 7.13-7.28 (m, SH), 7.54 (br, 2H), 8.53 (d, 1H, J=5.1 Hz), 8.58 (d, 1H, J=4.9 Hz).
PREPARATION EXAMPLE 82
Synthesis of 1-[[2-(4-chloro-3,5-dimethoxy)phenylpyridin-4-yl]methyl]-4-piperidone ethylene ketal
4-Piperidone ethylene ketal (573 mg) and 2-(4-chloro-3,5-dimetoxyphenyl)-4-chloromethylpyridine (1.19 g) were condensed in the same manner as described in Example 2 to give the title compound.
Yield: 1.67 g (theoretical amount). 1H-NMR (400 MHz, CDCl3) δ: 1.78 (t, 4H, J=5.6 Hz), 2.58 (br, 4H), 3.61 (s, 2H), 3.67 (s, 4H), 4.02 (s, 6H), 7.25-7.29 (m, 3H), 7.68 (s, 1H), 8.61 (d, 1H, J=4.9 Hz).
PREPARATION EXAMPLE 83
Synthesis of 1-[[2-(4-chloro-3,5-dimethoxyphenyl)pyridin-4-yl]methyl]-4-piperidone
1-[[2-(4-Chloro-3,5-dimethoxyphenyl)pyridin-4-yl]methyl]-4-piperidone ethylene ketal (1.67 g) was treated in the same manner as described in Preparation Example 23 to give the title compound.
Yield: 1.29 g (89%). 1H-NMR (400 MHz, CDCl3) δ: 2.50 (t, 4H, J=5.8 Hz), 2.81 (t, 4H, J=5.8 Hz), 3.71 (s, 2H), 4.02 (s, 6H), 7.26 (s, 2H), 7.33 (d, 1H, J=4.3 Hz), 7.70 (s, 1H), 8.66 (d, 1H, J=4.9 Hz).
PREPARATION EXAMPLE 84
Synthesis of 4-anilino-1-[[2-(4-chloro-3,5-dimethoxyphenyl)pyridin-4-yl]methyl]piperidine
1-[[2-(4-Chloro-3,5-dimethoxyphenyl)pyridin-4-yl]methyl]-4-piperidone (600 mg) and aniline (0.18 mL) were reacted in the same manner as described in Preparation Example 37 to give the title compound.
Yield: 465 mg (63%). 1H-NMR (400 MHz, CDCl3) δ: 1.49-1.69 (m, 2H), 2.08 (d, 2H, J=7.8 Hz), 2.23 (t, 2H, J=9.3 Hz), 2.87 (d, 2H, J=7.8 Hz), 3.34 (br, 1H), 3.60 (s, 2H), 4.02 (s, 6H), 6.60 (d, 2H), J=7.6 Hz), 6.69 (t, 1H, J=7.3 Hz), 7.10-7.20 (m, 2H), 7.20-7.30 (m, 3H), 7.67 (s, 1H), 8.62 (d, 1H, J=5.2 Hz).
EXAMPLE 43
Synthesis of 1-[[2-(4-chloro-3,5-dimethoxyphenyl)pyridin-4-yl]methyl]-4-[N-[[2-(4-chloro-3,5-dimethoxyphenyl)pyridin-4-yl]methyl]-N-phenylamino]piperidine trihydrochloride
4-Anilino-1-[[2-(4-chloro-3,5-dimethoxyphenyl)pyridin-4-yl]methyl]piperidine (230 mg) and 2-(4-chloro-3,5-dimethoxyphenyl)-4-chloromethylpyridine (157 mg) were condensed in the same manner as described in Example 9. The title compound was obtained as yellow powder after converting a free base to a trihydrochloride.
Yield: 104 mg (24%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.70-1.85 (m, 4H), 2.20 (t, 2H, J=2.3 Hz), 3.00 (d, 2H, J=1.3 Hz), 3.59 (s, 2H), 3.96 (s, 6H), 4.00 (s, 6H), 4.56 (s, 2H), 6.65-6.78 (m, 3H), 7.16 (s, 2H), 7.18-7.28 (m, 6H), 7.59 (s, 1H), 7.62 (s, 1H), 8.57 (d, 1H), J=5.1 Hz), 8.57 (d, 1H, J=4.8 Hz).
PREPARATION EXAMPLE 85
Synthesis of 4-(p-anisidino)-1-[[2-(4-chloro-3,5-dimethoxyphenyl)pyridin-4-yl]methyl]piperidine
1-[[2-(4-Chloro-3,5-dimethoxyphenyl)pyridin-4-yl]methyl]-4-piperidone (690 mg) and p-anisidine (283 mg) were reacted in the same manner as described in Preparation Example 37 to give the title compound.
Yield: 646 mg (72%). 1H-NMR (400 MHz, CDCl3) δ: 1.45-1.55 (m, 2H), 2.05 (d, 2H, J=11.7 Hz), 2.20 (t, 2H, J=11.2 Hz), 2.87 (d, 2H, J=11.7 Hz), 3.20-3.35 (m, 1H), 3.59 (s, 2H), 3.74 (s, 3H), 4.02 (s, 6H), 6.58 (d, 2H, J=8.7 Hz), 6.77 (d, 2H, J=8.7 Hz), 7.25-7.28 (m, 3H), 7.67 (s, 1H), 8.62 (d, 1H, J=4.9 Hz).
EXAMPLE 44
Synthesis of 1-[[2-(4-chloro-3,5-dimethoxyphenyl)pyridin-4-yl]methyl]-4-[N-[[2-(4-chloro-3,5-dimethoxyphenyl)pyridin-4-yl]methyl]-N-(4-methoxyphenyl)amino]piperidine trihydrochloride
4-(p-Anisidino)-1-[[2-(4-chloro-3,5-dimethoxyphenyl)pyridin-4-yl]methyl]piperidine (271 mg) and 2-(4-chloro-3,5-dimethoxyphenyl)-4-chloromethylpyridine (173 mg) were condensed in the same manner as described in Example 9. The title compound was obtained as yellow powder after converting a free base to a trihydrochloride.
Yield: 324 mg (67%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.65-1.90 (m, 4H), 2.16 (t, 2H, J=10.4 Hz), 2.97 (d, 2H, J=7.5 Hz), 3.54-3.60 (m, 1H), 3.58 (s, 2H), 3.73 (s, 3H), 3.97 (s, 6H), 4.00 (s, 6H), 4.46 (s, 2H), 6.74 (d, 2H, J=9.4 Hz), 6.79 (d, 2H, J=9.4 Hz), 7.16 (s, 2H), 7.20-7.29 (m, 4H), 7.59 (s, 1H), 7.62 (s, 1H), 8.56 (d, 1H, J=4.8 Hz), 8.60 (d, 1H, J=4.8 Hz).
PREPARATION EXAMPLE 86
Synthesis of 4-(3-methylthioanilino)-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine
1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]-4-piperidone (1.40 g) and 3-methylthioaniline (655 mg) were reacted in the same manner as described in Preparation Example 37 to give the title compound.
Yield: 1.01 g (54%). 1H-NMR (400 MHz, CDCl3) δ: 1.44-1.60 (m, 2H), 1.98-2.10 (m, 2H), 2.23 (br, 2H), 2.42 (s, 3H), 2.88 (br, 2H), 3.30 (br, 1H), 3.59 (s, 2H), 3.88 (s, 3H), 3.95 (s, 6H), 6.35 (d, 1H), J=7.6 Hz), 6.47 (s, 1H), 6.55 (d, 1H, J=8.6 Hz), 7.05 (t, 1H, J=7.9 Hz), 7.20 (d, 1H, J=4.9 Hz), 7.24 (s, 2H), 7.68 (br, 1H), 8.58 (d, 1H, J=4.9 Hz).
EXAMPLE 45
Synthesis of 4-[N-(3-methylthiophenyl)-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-3-yl]methyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine trihydrochloride
4-(3-Methylthioanilino)-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine (143 mg) and 3-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (114 mg) were condensed in the same manner as described in Example 9. The title compound was obtained as yellow powder after converting a free base to a trihydrochloride.
Yield: 45 mg (18%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.58-1.71 (s, 2H),1.79 (d, 2H, J=10.7 Hz), 2.16 (t, 2H, J=11.2 Hz), 2.38 (s, 3H), 2.96 (d, 2H, J=11.2 Hz), 3.56 (s, 3H), 3.68-3.97 (m, 1H), 3.90 (s, 3H), 3.92 (s, 9H), 3.96 (s, 9H), 4.42 (s, 2H), 6.45 (d, 1H, J=8.3 Hz), 6.52 (s, 1H), 6.61 (d, 1H, J=7.3 Hz), 6.74 (s, 2H), 7.11 (t, 1H, J=8.1 Hz), 7.15-7.26 (m, 4H), 7.54 (s, 1H), 7.68 (d, 1H, J=7.8 Hz), 8.53 (d, 1H, J=3.2 Hz), 8.59 (d, 1H, J=4.8 Hz).
EXAMPLE 46
Synthesis of 4-[N-(3-methylthiophenyl)-N-[2-(3,4,5-trimethoxyphenyl)benzyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine dihydrochloride
4-(3-Methylthioanilino)-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine (143 mg) and 2-(3,4,5-trimethoxyphenyl)benzyl chloride (114 mg) were condensed in the same manner as described in Example 9. The title compound was obtained as yellow powder after converting a free base to a dihydrochloride.
Yield: 51 mg (23%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.56-1.73 (m, 2H), 1.78-1.87 (m, 2H), 2.10-2.20 (m, 2H), 2.38 (s, 3H), 2.91-2.98 (m, 2H), 3.55 (s, 2H), 3.70-3.80 (m, 1H), 3.88 (s, 6H), 3.90 (s, 3H), 3.92 (s, 3H), 3.96 (s, 6H), 4.35 (s, 2H), 6.47 (d, 1H, J=8.2 Hz), 6.53-6.62 (m, SH), 7.09 (t, 1H, J=8.0 Hz), 7.18-7.40 (m, 6H), 7.54 (s, 1H), 8.58 (d, 1H, J=4.7 Hz).
EXAMPLE 47
Synthesis of 4-[N-(3-methylthiophenyl)-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine fumarate
4-(3-Methylthioanilino)-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine (143 mg) and 4-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (114 mg) were condensed in the same manner as described in Example 9. The title compound was obtained as white powder after converting a free base to a fumarate.
Yield: 14 mg (5%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.76-1.86 (m, 5H), 2.17-2.23 (m, 2H), 2.39 (s, 3H), 2.97-3.00 (m, 2H), 3.58 (s, 2H), 3.89 (s, 3H), 3.90 (s, 3H), 3.93 (s, 6H), 3.96 (s, 6H), 4.54 (s, 2H), 6.47-6.50 (m, 1H), 6.63 (s, 1H), 6.64 (s, 1H), 7.10-7.15 (m, 2H), 7.15 (s, 2H), 7.20-7.21 (m, 1H), 7.22 (s, 2H), 7.55 (s, 1H), 7.59 (s, 1H), 8.56 (d, 1 H), J=5.1 Hz), 8.59 (d, 1 H, J=5.1 Hz).
EXAMPLE 48
Synthesis of 4-[N-(3-methylthiophenyl)-N-[3-(3,4,5-trimethoxyphenyl)benzyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine dihydrochloride
4-(3-Methylthioanilino)-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine (143 mg) and 3-(3,4,5-trimethoxyphenyl)benzyl chloride (114 mg) were condensed in the same manner as described in Example 9. The title compound was obtained as yellow powder after converting a free base to a dihydrochloride.
Yield: 60 mg (24%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.65-1.91 (m, 4H), 2.18 (t, 2H, J=10.5 Hz), 2.38 (s, 3H), 2.97 (d, 2H, J=10.9 Hz), 3.58 (s, 2H), 3.70-3.85 (m, 1H), 3.88 (s, 3H), 3.89 (s, 6H), 3.90 (s, 3H), 3.96 (s, 6H), 4.56 (s, 2H), 6.52 (d, 1H, J=8.4 Hz), 6.59 (d, 1H, J=7.6 Hz), 6.65 (s, 1H), 6.72 (s, 2H), 7.10 (t, 2H, J=8.0 Hz), 7.19-7.25 (m, 4H), 7.31-7.42 (m, 3H), 7.60 (s, 1H), 8.59 (d, 1H, J=7.8 Hz).
EXAMPLE 49
Synthesis of 4-[N-(3-methylthiophenyl)-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine trihydrochloride
4-(3-Methylthioanilino)-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine (143 mg) and 5-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (114 mg) were condensed in the same manner as described in Example 9. The title compound was obtained as yellow powder after converting a free base to a trihydrochloride.
Yield: 22 mg (9%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.50-2.05 (m, 4H), 2.20 (br, 2H), 2.37 (s, 3H), 3.05 (br, 2H), 3.50-3.70 (br, 3H), 3.86 (s, 3H), 3.87 (s, 3H), 3.92 (s, 6H), 3.95 (s, 6H), 4.52 (s, 2H), 6.49 (d, 1H, J=8.3 Hz), 6.62 (br, 2H), 7.09 (t, 1H, J=8.2 Hz), 7.18-7.30 (m, 6H), 7.58 (s, 2H), 8.54 (br 1H), 8.60 (br, 1H).
EXAMPLE 50
Synthesis of 4-[N-(3-methyithiophenyl)-N-[4-(3,4,5-trimethoxyphenyl)benzyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine dihydrochloride
4-(3-Methylthioanilino)-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine (143 mg) and 4-(3,4,5-trimethoxyphenyl)benzyl chloride (114 mg) were condensed in the same manner as described in Example 9. The title compound was obtained as yellow powder after converting a free base to a dihydrochloride.
Yield: 57 mg (22%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.58-1.83 (m, 4H), 2.20 (t, 2H, J=11.3 Hz), 2.39 (s, 3H), 2.98 (d, 2H, J=11.1 Hz), 3.58 (s, 2H), 3.88 (s, 3H), 3.90 (s, 3H), 3.91 (s, 6H), 3.96 (s, 6H), 4.53 (s, 2H), 6.51 (dd, 1H, J=8.4 Hz, 2.4 Hz), 6.60 (d, 1H, J=8.0 Hz), 6.64 (s, 1H), 6.75 (s, 2H), 7.10 (t, 1H, J=8.1 Hz), 7.24-7.33 (m, 4H), 7.47 (d, 2H, J=8.0 Hz), 7.61 (s, 1H), 8.59 (d, 1H, J=5.0 Hz).
PREPARATION EXAMPLE 87
Synthesis of 4-propargylamino-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine
1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]-4-piperidone (400 mg) and propargylamine (80 mg) were reacted in the same manner as described in Preparation Example 25 to give the title compound.
Yield: 227 mg (63%). 1H-NMR (400 MHz, CDCl3) δ: 1.38-1.51 (m, 2H), 1.83-1.86 (m, 3H), 2.10-2.15 (m, 2H), 2.21 (s, 1H), 2.74 (br, 1H), 2.83-2.87 (m, 2H), 3.45 (s, 2H), 3.56 (s, 2H), 3.89 (s, 3H), 3.96 (s, 6H), 7.19 (d, 1H, J=4.9 Hz), 7.24 (s, 2H), 7.65 (s, 1H), 8.58 (d, 1H, J=4.9 Hz).
EXAMPLE 51
Synthesis of 4-[N-propargyl-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine tetrahydrochloride
4-Propargylamino-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine (227 mg) and 4-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (226 mg) were condensed in the same manner as described in Example 9. The title compound was obtained as yellow powder after converting a free base to a tetrahydrochloride.
Yield: 128 mg (23%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.48-2.40 (m, 7H), 2.72 (br, 1H), 3.02 (br, 2H), 3.39 (s, 2H), 3.64 (br, 2H), 3.84 (s, 2H), 3.91 (s, 6H), 3.98 (s, 6H), 3.99 (s, 6H), 7.22-7.29 (m, 6H), 7.66 (br, 2H), 8.60 (d, 1H, J=4.9 Hz), 8.62 (d, 1H, J=4.9 Hz).
PREPARATION EXAMPLE 88
Synthesis of 4-(5-indanylamino)-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine
1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]-4-piperidone (1.40 g) and 5-aminoindan (680 mg) were reacted in the same manner as described in Preparation Example 37 to give the title compound.
Yield: 1.22 g (59%). 1H-NMR (400 MHz, CDCl3) δ: 1.40-1.57 (m, 2H), 2.00-2.15 (m, 5H), 2.19-2.25 (m, 2H), 2.77-2.93 (m, 6H), 3.30 (br, 1H), 3.58 (s, 2H), 3.91 (s, 3H), 3.97 (s, 6H), 6.41 (d, 1H, J=8.0 Hz), 6.52 (s. 1H), 7.01(d. 1H, J=8.0 Hz), 7.21-7.26 (m, 3H), 7.64 (s, 1H), 8.60 (d, 1H, J=4.9 Hz).
EXAMPLE 52
Synthesis of 4-[N-(indan-5-yl)-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-3-yl]methyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine trihydrochloride
4-(5-Indanylamino)-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine (142 mg) and 3-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (114 mg) were condensed in the same manner as described in Example 9. The title compound was obtained as yellow powder after converting a free base to a trihydrochloride.
Yield: 90 mg (41%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.54-1.67 (m, 2H), 1.74-1.83 (m, 2H), 1.98-2.07 (m, 2H), 2.09-2.98 (m, 2H), 3.55 (s, 2H), 3.64-3.74 (m, 1H), 3.90 (s, 3H), 3.91 (s, 6H), 3.92 (s, 3H), 3.96 (s, 6H), 4.41 (s, 2H), 6.49 (dd, 1H, J=8.2 Hz, 2.4 Hz), 6.59 (s, 1H), 6.74 (s, 2H), 7.04 (d, 1H, J=8.2 Hz), 7.15-7.20 (m, 2H), 7.22 (s, 2H), 7.54 (s, 1H), 7.77 (dd, 1H, J=7.8 Hz, 1.4 Hz), 8.52 (dd, 1H, J=4.7 Hz, 1.8 Hz), 8.59 (d, 1H, J=5.1 Hz).
EXAMPLE 53
Synthesis of 4-[N-(indan-5-yl)-N-[2-(3,4,5-trimethoxyphenyl)benzyl]amino]1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine dihydrochloride
4-(5-Indanylamino)-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine (142 mg) and 2-(3,4,5-trimethoxyphenyl)benzyl chloride (114 mg) were condensed in the same manner as described in Example 9. The title compound was obtained as yellow powder after converting a free base to a dibydrochloride.
Yield: 115 mg (47%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.56-1.66 (m, 2H), 1.80-1.83 (m, 2H), 2.00-2.05 (m, 2H), 2.11-2.18 (m, 2H), 2.77-2.83 (m, 4H), 2.92-2.95 (m, 2H), 3.55 (s, 2H), 3.72 (br, 1H), 3.87 (s, 6H), 3.90 (s, 3H), 3.92 (s, 3H), 3.96 (s, 6H), 4.34 (s, 2H), 6.49 (d, 1H, J=8.3 Hz), 6.56 (s, 2H), 6.60 (s, 1H), 7.02 (d, 1H, J=8.3 Hz), 7.17-7.27 (m, 5H), 7.42-7.45 (m, 1H), 7.54 (s, 1H), 8.58 (d, 1H, J=4.9 Hz).
EXAMPLE 54
Synthesis of 4-[N-(indan-5-yl)-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine trihydrochloride
4-(5-Indanylamino)-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine (142 mg) and 4-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (114 mg) were condensed in the same manner as described in Example 9. The title compound was obtained as white powder after converting a free base to a trihydrochloride.
Yield: 23 mg (9%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.60-1.95 (m, 4H), 2.00 (quint, 2H, J=7.3 Hz), 2.20 (br, 2H), 2.75-2.81 (m, 4H), 2.99 (br, 2H), 3.58 (br, 2H), 3.77 (s, 1H), 3.86 (s, 3H), 3.87 (s, 3H), 3.91 (s, 6H), 3.94 (s, 6H), 4.49 (s, 2H), 6.51 (d, 1H, J=8.3 Hz), 6.62 (s, 1H), 7.02 (d, 1H, J=8.0 Hz), 7.16 (s, 2H), 7.18-7.22 (m, 4H), 7.57 (br, 2H), 8.52 (d, 1H, J=4.9 Hz), 8.57 (d, 1H, J=4.9 Hz).
EXAMPLE 55
Synthesis of 4-[N-(indan-5-yl)-N-[3-(3,4,5-trimethoxyphenyl)benzyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine dihydrochloride
4-(5-Indanylamino)-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine (60 mg) and 3-(3,4,5-trimethoxyphenyl)benzyl chloride (114 mg) were condensed in the same manner as described in Example 9. The title compound was obtained as yellow powder after converting a free base to a dihydrochloride.
Yield: 18 mg (19%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.60-1.95 (m, 4H), 2.00 (quint, 2H, J=7.2 Hz), 2.20 (br, 2H), 2.75-2.81 (m, 4H), 2.95 (br, 2H), 3.60 (br, 2H), 3.85 (br, 1H), 3.86 (s, 3H), 3.87 (s, 6H), 3.88 (s, 3H), 3.94 (s, 6H), 4.51 (s, 2H), 6.54 (d, 1H, J=8.2 Hz), 6.66 (s, 1H), 6.70 (s, 2H), 7.01 (d, 1H, J=8.4 Hz), 7.19 (d, 1H, J=4.9 Hz), 7.19-7.42 (m, 6H), 7.60 (br, 1H), 8.59 (br, 1H).
EXAMPLE 56
Synthesis of 4-[N-(indan-5-yl)-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]piperidine trihydrochloride
4-(5-Indanylamino)-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine (143 mg) and 5-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (114 mg) were condensed in the same manner as described in Example 9. The title compound was obtained as yellow powder after converting a free base to a trihydrochloride.
Yield: 138 mg (63%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.71-1.91 (m, 4H), 1.98-2.06 (m, 2H), 2.13-2.22 (m, 2H), 2.76-2.84 (m, 4H), 2.94-3.05 (m, 2H), 3.57 (s, 2H), 3.69-3.78 (m, 1H), 3.89 (s, 3H), 3.90 (s, 3H), 3.94 (s, 6H), 3.96 (s, 6H), 4.50 (s, 2H), 6.57 (dd, 1H, J=8.2 Hz, 2.3 Hz), 6.67 (s, 1H), 7.04 (d, 1H, J=8.4 Hz), 7.20-7.22 (m, 1H), 7.22 (s, 2H), 7.23 (s, 2H), 7.57-7.62 (m, 1H), 7.60 (s, 1H), 7.65 (dd, 1H, J=8.2 Hz, 2.2 Hz), 8.58-8.62 (m, 2H).
EXAMPLE 57
Synthesis of 4-[N-(indan-5-yl)-N-[4-(3,4,5-trimethoxyphenyl)benzyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine dihydrochloride
4-(5-Indanylamino)-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine (143 mg) and 4-(3,4,5-trimethoxyphenyl)benzyl chloride (114 mg) were condensed in the same manner as described in Example 9. The title compound was obtained as yellow powder after converting a free base to a dihydrochloride.
Yield: 95 mg (39%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.74-1.90 (m, 4H), 2.01-2.06 (m, 2H), 2.16-2.22 (m, 2H), 2.78-2.84 (m, 4H), 2.96-2.99 (m, 2H), 3.58 (s, 2H), 3.72 (br, 1H), 3.88 (s, 3H), 3.90 (s, 3H), 3.91 (s, 6H), 3.96 (s, 6H), 4.51 (s, 2H), 6.55 (d, 1H, J=8.3 Hz), 6.67 (s, 1H), 6.72 (s, 2H), 7.04 (d, 1H, J=8.3 Hz), 7.20 (d, 1H, J=5.1 Hz), 7.23 (s, 2H), 7.35 (d, 2H, J=8.1 Hz), 7.47 (d, 2H, J=8.1 Hz), 7.61 (s, 1H), 8.59 (d, 1H, J=4.9 Hz).
PREPARATION EXAMPLE 89
Synthesis of 4-(4-butylanilino)-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine
1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]-4-piperidone (1.24 g) and 4-butylaniline (149 mg) were reacted in the same manner as described in Preparation Example 37 to give the title compound.
Yield: 1.23 g (72%). 1H-NMR (400 MHz, CDCl3) δ: 0.82 (t, 3H, J=7.3 Hz), 1.20-1.30 (m, 2H), 1.38-1.50 (m, 4H), 1.92-2.25 (m, 4H), 2.40 (t, 2H, J=7.7 Hz), 2.77 (br, 2H), 3.21(br, 1H), 3.50 (s, 2H), 3.82 (s, 3H), 3.89 (s, 6H), 6.45 (d, 2H, J=7.8 Hz), 6.89 (d, 2H, J=8.0 Hz), 7.13 (d, 1H, J=4.9 Hz), 7.18 (s, 2H), 7.58 (s, 1H), 8.52 (d, 1H, J=4.9 Hz).
EXAMPLE 58
Synthesis of 4-[N-(4-butylphenyl)-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-3-yl ]methyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine trihydrochloride
4-(4-Butylanilino)-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl ]methyl]piperidine (147 mg) and 3-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (114 mg) were condensed in the same manner as described in Example 9. The title compound was obtained as yellow powder after converting a free base to a trihydrochloride.
Yield: 58 mg (27%). 1NMR (400 MHz, measured as a free base, CDCl3) δ: 0.91 (t, 3H, J=7.3 Hz), 1.321.35 (m, 2H), 1.50-1.70 (m, 4H), 1.75 (br, 2H), 2.10-2.20 (m, 2H), 2.49 (t, 2H, J=7.6 Hz, 2.95 (br, 2H), 3.55 (s, 2H), 3.70 (br, 1H), 3.90 (s, 3H), 3.91 (s, 6H), 3.92 (s, 3H), 3.96 (s, 6H), 4.41 (s, 2H), 6.59 (d, 2H, J=8.8 Hz), 6.74 (s, 2H), 7.00 (d, 2H, J=8.6 Hz), 7.16-7.17 (m, 1H), 7.19 (d, 1H, J=4.9 Hz), 7.22 (s, 2H), 7.54 (s, 1H), 8.59 (d, 1H, J=7.5 Hz), 8.52 (br, 1H), 8.59 (d, 1H, J=4.9 Hz).
EXAMPLE 59
Synthesis of 4-[N-(4-butylphenyl)-N-[2-(3,4,5-trimethoxyphenyl)benzyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine dihydrochloride
4-(4-Butylanilino)-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine (147 mg) and 2-(3,4,5-trimethoxyphenyl)benzyl chloride (114 mg) were condensed in the same manner as described in Example 9. The title compound was obtained as yellow powder after converting a free base to a dihydrochloride.
Yield: 59 mg (24%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 0.90 (t, 3H, J=7.4 Hz), 1.251.41 (m, 2H),1.48-1.75 (m, 4H), 1.81 (d, 2H, J=11.7 Hz), 2.13 (t, 2H, J=11.2 Hz), 2.48 (t, 2H, J=7.5 Hz), 2.93 (d, 2H, J=11.2 Hz), 3.55 (s, 2H), 3.65-3.80 (m, 1H), 3.87 (s, 6H), 3.90 (s, 3H), 3.92 (s, 1H), 3.96 (s, 6H), 4.33 (s, 2H), 6.56 (s, 2H), 6.60 (d, 2H, J=8.5 Hz), 6.98 (d, 2H, J=8.5 Hz), 7.18 (d, 1H, J=4.9 Hz), 7.21 (s, 2H), 7.20-7.37 (m, 3H), 7.41 (br, 1H), 7.54 (s, 1H), 8.58 (d, 1H, J=4.9 Hz).
EXAMPLE 60
Synthesis of 4-[N-(4-buthylphenyl)-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine trihydrochloride
4-(4-Butylanilino)-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine (196 mg) and 4-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (129 mg) were condensed in the same manner as described in Example 9. The title compound was obtained as white powder after converting a free base to a trihydrochloride.
Yield: 20 mg (6%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 0.88 (t, 3H, J=1.3 Hz), 1.201.35 (m, 2H), 1.49-1.60 (m, 2H), 1.62-2.02 (m, 4H), 2.20 (br, 2H), 2.46 (t, 2H, J=7.3 Hz), 3.05 (br, 2H), 3.60 (br, 3H), 3.87 (s, 3H), 3.88 (s, 3H), 3.90 (s, 6H), 3.94 (s, 6H), 4.49 (s, 2H), 6.62 (d, 2H, J=8.3 Hz), 6.98 (d, 2H, J=8.3 Hz), 7.13 (s, 2H), 7.15-7.40 (m, 4H), 7.55 (br, 2H), 8.52 (d, 1H, J=4.9 Hz), 8.60 (br, 1H).
EXAMPLE 61
Synthesis of 4-[N-(4-butylphenyl)-N-[3-(3,4,5-trimethoxyphenyl)benzyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine dihydrochloride
4-(4-Butylanilino)-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine (147 mg) and 3-(3,4,5-trimethoxyphenyl)benzyl chloride (114 mg) were condensed in the same manner as described in Example 9. The title compound was obtained as yellow powder after converting a free base to a dihydrochloride.
Yield: 102 mg (42%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 0.90 (t, 3H, J=7.4 Hz), 1.30-1.36 (m, 2H), 1.48-1.56 (m, 2H), 1.76-1.89 (m, 4H), 2.19 (br, 2H), 2.48 (t, 2H, J=7.8 Hz), 2.97 (br, 2H), 3.58 (s, 2H), 3.86 (br, 1H), 3.88 (s, 3H), 3.90 (s, 3H), 3.95 (s, 6H), 4.54 (s, 2H), 6.68 (d, 2H, J=8.6 Hz), 6.72 (s, 2H), 7.00 (d, 2H, J=8.6 Hz), 7.20-7.27 (m, 2H), 7.23 (s, 2H), 7.32-7.40 (m, 2H), 7.44 (s, 1H), 7.62 (s, 1H), 8.59 (d, 1H, J=5.1 Hz).
EXAMPLE 62
Synthesis of 4-[N-(4-butylphenyl)-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine trihydrochloride
4-(4-Butylanilino)-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine (147 mg) and 5-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (114 mg) were condensed in the same manner as described in Example 9. The title compound was obtained as yellow powder after converting a free base to a trihydrochloride.
Yield: 65 mg (21%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 0.90 (t, 3H, J=7.3 Hz), 1.321.36 (m, 2H), 1.50-1.54 (m, 2H), 1.70-1.95 (m, 4H), 2.17 (br, 2H), 2.49 (t, 2H, J=7.7 Hz), 2.96 (br, 2H), 3.58 (s, 2H), 3.75 (br, 1H), 3.89 (s, 3H), 3.90 (s, 3H), 3.94 (s, 6H), 3.96 (s, 6H), 4.50 (s, 2H), 6.68 (d, 2H, J=8.6 Hz), 7.00 (d, 2H, J=8.6 Hz), 7.20-7.22 (m, 3H), 7.23 (s, 2H), 7.58-7.66 (m, 3H), 8.59 (br, 1H), 8.60 (br, 1H).
EXAMPLE 63
Synthesis of 4-[N-(4-butylphenyl)-N-[4-(3,4,5-trimethoxyphenyl)benzyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine dihydrochloride
4-(4-Butylanilino)-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine (147 mg) and 4-(3,4,5-trimethoxyphenyl)benzyl chloride (114 mg) were condensed in the same manner as described in Example 9. The title compound was obtained as yellow powder after converting a free base to a dihydrochloride.
Yield: 82 mg (33%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 0.90 (t, 3H, J=7.3 Hz), 1.301.36 (m, 2H), 1.51-1.55 (m, 2H), 1.79-1.90 (m, 4H), 2.18 (br, 2H), 2.48 (t, 2H, J=7.7 Hz), 2.98 (d, 2H, J=10.7 Hz), 3.57 (s, 2H), 3.72-3.85 (m, 1H), 3.88 (s, 3H), 3.90 (s, 3H), 3.91 (s, 6H), 3.96 (s, 6H), 4.50 (s, 2H), 6.66 (d, 2H, J=8.8 Hz), 6.75 (s, 2H), 7.00 (d, 2H, J=8.8 Hz), 7.20 (d, 1H, J=4.9 Hz), 7.22 (s, 2H), 7.33 (d, 2H, J=8.2 Hz), 7.47 (d, 2H, J=8.2 Hz), 7.61 (s, 1H), 8.59 (d, 1H, J=5.1 Hz).
PREPARATION EXAMPLE 90
Synthesis of 1-(4-pycolyl)-4-piperidone
4-piperidone hydrochloride monohydrate (922 mg) and 4-picolyl chloride hydrochloride (820 mg) were reacted in the same manner as described in Example 9 to give the title compound.
Yield: 870 mg (92%). 1H-NMR (400 MHz, CDCl3) δ: 2.46 (t, 4H, J=5.9 Hz), 2.74 (t, 4H, J=6.2 Hz), 3.61 (s, 2H), 7.29 (d, 2H, J=6.2 Hz), 8.55 (dd, 2H, J=6.2 Hz, 1.1 Hz).
PREPARATION EXAMPLE 91
Synthesis of 1-(4-pycolyl)-4-(4-pycolylamino)piperidine tetrahydrochloride
1-(4-pycolyl)-4-piperidone (870 mg) and 4-picolylamine (497 mg) were coupled in the same manner as described in Preparation Example 37. The title compound was obtained as pale brown powder after converting a free base to tetrahydrochloride.
Yield: 363 mg (19%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.37-1.51 (m, 2H), 1.82-1.90 (m, 21T), 2.04 (dt, 2H, J=11.6 Hz, 2.7 Hz), 2.44-2.55 (m, 1H), 2.76-2.82 (m, 2H), 3.47 (s, 2H), 3.82 (s, 2H), 7.23-7.26 (m, 4H), 8.50-8.53 (m, 4H).
PREPARATION EXAMPLE 92
Synthesis of 4-(p-anisidino)-1-(tert-butoxycarbonyl)piperidine
1-(tert-Butoxycarbonyl)-4-piperidone (116 g) and p-anisidine (68.3 g) were condensed in the same manner as described in Preparation Example 37 to give the title compound.
Yield: 125 g (74%). 1H-NMR (400 MHz, CDCl3) δ: 1.23-1.35 (m, 2H), 1.46 (s, 9H), 1.96-2.06 (m, 2H), 2.83-2.96 (m, 2H), 3.27-3.38 (m, 1H), 3.74 (s, 9H), 3.94-4.12 (m, 2H), 6.58 (d, 2H, J=9.0 Hz), 6.77 (d, 2H, J=9.0 Hz).
PREPARATION EXAMPLE 93
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-(4-methoxyphenyl)-N-[3-(3,4,5-trimethoxyphenyl)benzoyl]amino]piperidine
4-(p-Anisidino)-1-(tert-butoxycarbonyl)piperidine (613 mg) and 3-(3,4,5-trimethoxyphenyl)benzoic acid (577 mg) were condensed in the same manner as described in Example 1 to give the title compound.
Yield: 416 mg (36%).
PREPARATION EXAMPLE 94
Synthesis of 4-[N-(4-methoxyphenyl)-N-[3-(3,4,5-trimethoxyphenyl)benzoyl]amino]piperidine hydrochloride
To a solution of 1-(tert-butoxycarbonyl)-4-[N-(4-methoxyphenyl)-N-[3-(3,4,5-trimethoxyphenyl)benzoyl]amino]piperidine (416 mg) in ethyl acetate (5 mL) was added 4 M hydrogen chloride in ethyl acetate (5 mL). The mixture was stirred at room temperature for 4 hr, resulting precipitates were collected and washed with ethyl acetate on a funnel to give the title compound.
Yield: 315 mg (85%)
EXAMPLES 64 TO 66
These compounds were prepared by the condensation of 4-[N-(4-methoxyphenyl)-N-[3-(3,4,5-trimethoxyphenyl)benzoyl]amino]piperidine hydrochloride with chloride derivatives obtained in Preparation Examples 3, 42 and 48. Free bases obtained were then converted to the corresponding hydrochlorides.
Yields and NMR data of their free bases are listed below.
| |
| Ex- |
|
|
|
| am- |
|
|
NMR data (400 MHz, measured |
| ple |
Structure |
Yield |
as free bases, CDCl3) δ |
| |
| 64 |
|
68% |
1.53-1.55(m, 2H), 1.89(d, 2H, J=12.0Hz), 2.23(t, 2H, J=12.0 Hz), 2.91(d, 2H, J=11.0Hz), 3.51 (s, 2H), 3.70(s, 3H), 3.84(s, 3H), 3.87(s, 9H), 3.92(s, 6H), 4.78(br, 1H), 6.54(s, 2H), 6.72(d, 2H, J=8.5Hz), 6.94(d, 2H, J=8.5Hz), 7.13-7.20(m, 4H), 7.18(s, 2H), |
| # 7.32(d, 1H, J=5.3Hz), 7.45(s, 1H), 8.19(d, 1H, J=4.9Hz). |
| 65 |
|
52% |
1.66-1.89(m, 4H), 2.05-2.17(m, 2H), 2.97(d, 2H, J=10.3Hz), 3.43-3.60(m, 1H), 3.57(s, 2H), 3.86(s, 3H), 3.87(s, 6H), 3.91(s, 6H), 4.42(s, 2H), 6.63(s, 2H), 6.72-6.79(m, 6H), 7.64(s, 1H), 7.78(br, 1H), 8.46(d, 2H, J=1.6 Hz), 8.59(d, 1H, J=2.4Hz), 8.68 |
| # (d, 1H, J=2.2Hz). |
| 66 |
|
75% |
1.42-1.58(m, 2H), 1.85-1.92(m, 2H), 2.14-2.23(m, 2H), 2.93-3.03 (m, 2H), 3.56(s, 2H), 3.70(s, 3H), (m, 2H), 3.56(s, 2H), 3.70(s, 3H), 3.85(s, 3H), 3.87(s, 3H), 3.87(s, 6H), 3.90(s, 6H), 4.79(br, 1H), 6.54(s, 2H), 6.70(d, 2H, J=8.9 Hz), 6.74(s, 2H), 6.93(d, 2H, |
| # J=8.9Hz), 7.17-7.23(m, 3H), 7.31-7.43(m, 5H). |
| |
PREPARATION EXAMPLE 95
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-(4-methoxyphenyl)-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]piperidine
4-(p-Anisidino)-1-(tert-butoxycarbonyl)piperidine (2.21 g) and 4-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (2.12 g) were condensed in the same manner as described in Example 9 to give the title compound.
Yield: 3.76 g (93%). 1H-NMR (400 MHz, CDCl3) δ: 1.40-1.64 (m, 2H), 1.44 (s, 9H), 1.82-1.91 (m, 2H), 2.71-2.84 (m, 2H), 3.62-3.73 (m, 1H), 3.74 (s, 3H), 3.89 (s, 3H), 3.94 (s, 6H), 4.10-4.30 (m, 2H), 4.40 (s, 2H), 6.76 (d, 2H, J=9.4 Hz), 6.79 (d, 2H, J=9.8 Hz), 7.14-7.19 (m, 3H), 7.56 (s, 1H), 8.55 (d, 1H, J=5.1 Hz).
PREPARATION EXAMPLE 96
Synthesis of 4-[N-(4-methoxyphenyl)-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]piperidine dihydrochloride
1-(tert-Butoxycarbonyl)-4-[N-(4-methoxyphenyl)-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]piperidine (3.76 g) was treated in the same manner as described in Preparation Example 94 to give the title compound.
Yield: 3.77 g (theoretical yield).
PREPARATION EXAMPLE 97
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-(4-methoxyphenyl)-N-[[3-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]amino]piperidine
4-(p-anisidino)-1-(tert-butoxycarbonyl)piperidine (613 mg) and 5-chloromethyl-3-(3,4,5-trimethoxyphenyl)pyridine (588 mg) was treated in the same manner as described in Preparation Example 9 to give pale yellow amorphous of the title compound.
Yield: 159 mg (14%). 1H-NMR (400 MHz, CDCl3) δ: 1.44 (s, 9H), 1.50-1.65 (m, 2H), 1.83-1.91 (m, 2H), 2.70-2.84 (m, 2H), 3.53-3.62 (m, 1H), 3.73 (s, 3H), 3.89 (s, 3H), 3.91 (s, 6H), 4.10-4.29 (m, 2H), 4.41 (s, 2H), 6.66 (s, 2H), 6.76-6.84 (m, 4H), 7.70 (s, 1H), 8.49 (s, 1H), 8.63 (d, 1H, J=2.1 Hz).
PREPARATION EXAMPLE 98
Synthesis of 4-[N-(4-methoxyphenyl)-N-[[3-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]amino]piperidine dihydrochloride
1-(tert-Butoxycarbonyl)-4-[N-(4-methoxyphenyl)-N-[[3-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]amino]piperidine (159 mg) was treated in the same manner as described in Preparation Example 94 to give pale yellow powder of the title compound.
Yield: 142 mg (94%).
PREPARATION EXAMPLE 99
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-(4-methoxyphenyl)-N-[3-(3,4,5-trimethoxyphenyl)benzyl]amino]piperidine
4-(p-Anisidino)-1-(tert-butoxycarbonyl)piperidine (613 mg) and 3-(3,4,5-trimethoxyphenyl)benzyl chloride (586 mg) was treated in the same manner as described in Example 9 to give pale yellow amorphous of the title compound.
Yield: 1.12 g (90%). 1H-NMR (400 MHz, CDCl3) δ: 1.44 (s, 9H), 1.50-1.63 (m, 2H), 1.82-1.91 (m, 2H), 2.71-2.83 (m, 2H), 3.69 (tt, 1H, J=11.5 Hz, 3.5 Hz), 3.73 (s, 3H), 3.88 (s, 3H), 3.90 (s, 6H), 4.10-4.28 (m, 2H), 4.42 (s, 2H), 6.71 (s, 2H), 6.78 (s, 4H), 7.24-7.28 (m, 1H), 7.31-7.40 (m, 2H), 7.42 (s, 1H).
PREPARATION EXAMPLE 100
Synthesis of 4-[N-(4-methoxyphenyl)-N-[3-(3,4,5-trimethoxyphenyl)benzyl]amino]piperidine hydrochloride
1-(tert-Butoxycarbonyl)-4-[N-(4-methoxyphenyl)-N-[3-(3,4,5-trimethoxyphenyl)benzyl]amino]piperidine (1.12 g) was treated in the same manner as described in Preparation Example 94 to give pale yellow powder of the title compound.
Yield: 980 mg (99%).
EXAMPLES 67 TO 71
These compounds were obtained by the condensation of amines obtained in Preparation Examples 96, 98 and 100 with chloride derivatives obtained in Preparation Examples 42 and 48. Free bases obtained were then converted to the corresponding hydrochlorides. Yields and NMR data of their free bases are listed below.
| |
| Ex- |
|
|
|
| am- |
|
|
NMR data (400 MHz, measured |
| ple |
Structure |
Yield |
as free bases, CDCl3) δ |
| |
| 67 |
|
62% |
1.60-1.92(m, 4H), 2.08-2.22(m, 2H), 2.92-3.06(m, 2H), 3.54-3.64 (m, 3H), 3.73(s, 3H), 3.89(s, 3H), 3.90(s, 3H), 3.93(s, 12H), 4.43(s, 2H), 6.70-6.81(m, 6H), 7.12-7.17(m, 3H), 7.56(s, 1H), 7.76(s, 1H), 8.49(d, 1H, J=1.8 Hz), 8.53(d, 1H, J=5.1Hz), 8.70 |
| # (s, 1H). |
| 68 |
|
54% |
1.65-1.79(m, 2H), 1.81-1.90(m, 2H), 2.04-2.18(m, 2H), 2.94-3.06 (m, 2H), 3.52-3.66(m, 3H), 3.72 (s, 3H), 3.89(s, 6H), 3.92(s, 6H), 3.93(s, 6H), 4.44(s, 2H), 6.70- 6.80(m, 6H), 7.13-7.17(m, 3H), 7.24-7.50(m, 4H), 7.55(s, 1H), 8.53(d, 1H, J=4.9Hz). |
| 69 |
|
52% |
1.66-1.89(m, 4H), 2.05-2.17(m, 2H), 2.97(d, 2H, J=10.3Hz), 3.43-3.60(m, 1H), 3.57(s, 2H), 3.86(s, 3H), 3.87(s, 6H), 3.91(s, 6H), 4.42(s, 2H), 6.63(s, 2H), 6.72-6.79(m, 6H), 7.64(s, 1H), 7.78(br, 1H), 8.46(d, 2H, J=1.6 Hz), 8.59(d, 1H, J=2.4Hz), 8.68 |
| # (d, 1H, J=2.2Hz). |
| 70 |
|
69% |
1.55-1.97(m, 4H), 2.06-2.21(m, 2H), 2.92-3.07(m, 2H), 3.53-3.68 (m, 3H), 3.72(s, 3H), 3.87(s, 3H), 3.89(s, 6H), 3.94(s, 3H), 4.46(s, 2H), 6.69(s, 2H), 6.73- 6.82(m, 6H), 7.22-7.29(m, 1H), 7.32(t, 1H, J=7.4Hz), 7.36(d, 1H, J=7.8Hz), 7.41(s, 1H), |
| # 7.79(br, 1H), 8.48(s, 1H), 8.71(br, 1H). |
| 71 |
|
75% |
1.69-1.89(m, 4H), 2.06-2.15(m, 2H), 2.96-3.04(m, 2H), 3.56-3.66 (m, 1H), 3.57(s, 2H), 3.72(s, 3H), 3.87(s, 3H), 3.89(s, 9H), 3.92(s, 6H), 4.46(s, 2H), 6.70(s, 2H), 6.71-6.79(m, 6H), 7.23-7.47 (m, 8H). |
| |
PREPARATION EXAMPLE 101
Synthesis of 1-(tert-butoxycarbonyl)-4-(4-ethoxyphenylamino)piperidine
1-(tert-butoxycarbonyl)-4-piperidinone (5.00 g) and p-phenetidine (3.28 g) was treated in the same manner as described in Preparation Example 37 to give brown powder of the title compound.
Yield: 7.00 g (91%). 1H-NMR (400 MHz, CDCl3) δ: 1.21-1.31 (m, 2H), 1.37 (t, 3H, J=7.0 Hz), 1.46 (s, 9H), 1.97-2.05 (m, 2H), 2.84-2.95 (m, 2H), 3.28-3.37 (m, 1H), 3.96 (q, 2H, J=7.0 Hz), 3.99-4.10 (m, 2H), 6.57 (d, 2H, J=8.8 Hz), 6.77 (d, 2H, J=9.0 Hz).
PREPARATION EXAMPLE 102
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-(4-ethoxyphenyl)-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]piperidine
1-(tert-Butoxycarbonyl)-4-[(4-ethoxyphenyl)amino]piperidine (641 mg) and 4-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (588 mg) was treated in the same manner as described in Example 9 to give light yellow amorphous of the title compound.
Yield: 1.08 g (94%). 1H-NMR (400 MHz, CDCl3) δ: 1.36 (t, 3H, J=7.9 Hz), 1.44 (s, 9H), 1.49-1.58 (m, 2H), 1.82-1.92 (m, 2H), 2.70-2.85 (m, 2H), 3.62-3.72 (m, 1H), 3.89 (s, 3H), 3.94 (s, 6H), 4.12-4.29 (m, 2H), 4.39 (s, 2H), 6.75 (d, 2H, J=9.2 Hz), 6.78 (d, 2H, J=9.6 Hz), 7.14-7.18 (m, 3H), 7.55 (s, 1H), 8.54 (d, 1H, J=5.1 Hz).
PREPARATION EXAMPLE 103
Synthesis of 4-[N-(4-ethoxyphenyl)-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]piperidine dihydrochloride
1-(tert-Butoxycarbonyl)-4-[N-(4-ethoxyphenyl)-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]piperidine (1.08 g) was treated in the same manner as described in Preparation Example 94 to give light yellow powder of the title compound.
Yield: 1.01 g (98%).
PREPARATION EXAMPLE 104
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-(4-ethoxyphenyl)-N-[[3-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]amino]piperidine
1-(tert-Butoxycarbonyl)-4-[(4-ethoxyphenyl)amino]piperidine (641 mg) and 5-chloromethyl-3-(3,4,5-trimethoxyphenyl)pyridine (588 mg) was treated in the same manner as described in Example 9 to give light yellow amorphous of the title compound.
Yield: 452 mg (39%). 1H-NMR (400 MHz, CDCl3) δ: 1.36 (t, 3H, J=6.8 Hz), 1.44 (s, 9H), 1.50-1.60 (m, 2H), 1.82-1.90 (m, 1H), 2.68-2.82 (m, 2H), 3.52-3.61 (m, 1H), 3.88 (s, 3H), 3.90 (s, 6H), 3.94 (q, 2H, J=7.0 Hz), 4.10-4.25 (m, 2H), 4.40 (s, 2H), 6.66 (s, 2H), 6.77 (d, 2H, J=9.2 Hz), 6.81 (d, 2H, J=9.2 Hz), 7.67 (s, 1H), 8.49 (d, 1H, J=2.0 Hz), 8.62 (d, 1H, J=2.1 Hz).
PREPARATION EXAMPLE 105
Synthesis of 4-[N-(4-ethoxyphenyl)-N-[[3-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]amino]piperidine dihydrochloride
1-(tert-Butoxycarbonyl)-4-[N-(4-ethoxyphenyl)-N-[[3-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]amino]piperidine (452 mg) was treated in the same manner as described in Preparation Example 94 to give light yellow powder of the title compound.
Yield: 380 mg (88%).
PREPARATION EXAMPLE 106
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-(4-ethoxyphenyl)-N-[3-(3,4,5-trimethoxyphenyl)benzyl]amino]piperidine
1-(tert-Butoxycarbonyl)-4-[(4-ethoxyphenyl)amino]piperidine (641 mg) and 3-(3,4,5-trimethoxyphenyl)benzyl chloride (586 mg) was treated in the same manner as described in Example 9 to give light yellow amorphous of the title compound.
Yield: 1.06 g (92%). 1H-NMR (400 MHz, CDCl3) δ: 1.36 (t, 3H, J=7.0 Hz), 1.44 (s, 9H), 1.53-1.59 (m, 2H), 1.83-1.91 (m, 2H), 2.70-2.83 (m, 2H), 3.64-3.73 (m, 1H), 3.88 (s, 3H), 3.90 (s, 6H), 3.94 (q, 2H, J=7.0 Hz), 4.10-4.29 (m, 2H), 4.41 (s, 2H), 6.71 (s, 2H), 6.76 (s, 4H), 7.26 (d, 1H, J=7.9 Hz), 7.33 (dd, 1H, J=7.4 Hz, 7.4 Hz), 7.38 (d, 1H, J=7.6 Hz), 7.42 (s, 1H),
PREPARATION EXAMPLE 107
Synthesis of 4-[N-(4-ethoxyphenyl)-N-[3-(3,4,5-trimethoxyphenyl)benzyl]amino]piperidine hydrochloride
1-(tert-Butoxycarbonyl)-4-[N-(4-ethoxyphenyl)-N-[3-(3,4,5-trimethoxyphenyl)benzyl]amino]piperidine (1.06 g) was treated in the same manner as described in Preparation Example 94 to give light yellow powder of the title compound.
Yield: 913 mg (97%).
EXAMPLES 72 TO 79
These compounds were obtained by the condensation of amines obtained in Preparation Examples 103, 105 and 107 with chloride derivatives obtained in Preparation Examples 3, 42 and 48. Free bases obtained were then converted to the corresponding hydrochlorides. Yields and NMR data of their free bases are listed below.
| |
| Ex- |
|
|
|
| am- |
|
|
NMR data (400 MHz, measured as |
| ple |
Structure |
Yield |
free bases, CDCl3) δ |
| |
| 72 |
|
49% |
1.36(t, 3H, J=7.1Hz), 1.68-1.94 (m, 4H), 2.10-2.24(m, 2H), 2.93- 3.04(m, 2H), 3.54-3.65(m, 3H), 3.89(s, 3H), 3.90(s, 3H), 3.93(s, 6H), 3.96(s, 6H), 4.45(s, 2H), 6.72(d, 2H, J=9.2Hz), 6.78(d, 2H, J=9.3Hz), 7.15(s, 2H), 7.17 (d, 1H, J=6.1Hz), 7.20(dd, 1H, |
| # J=4.9Hz, 1.0Hz), 7.23(s, 2H), 7.57(s, 1H), 7.61(br, 1H), 8.54(d, 1H, J=5.2Hz), 8.59(d, 1H, J=4.9 Hz). |
| 73 |
|
63% |
1.36(t, 3H, J=7.0Hz), 1.56-1.74 (m, 2H), 1.80-1.90(m, 2H), 2.07- 2.19(m, 2H), 2.92-3.02(m, 2H), 3.58(s, 2H), 3.88-3.95(m, 2H), 3.89(s, 3H), 3.93(s, 12H), 4.43 (s, 2H), 6.69-6.79(m, 6H), 7.12- 7.17(m, 3H), 7.55(s, 1H), 7.76(s, 1H), 8.49(d, 1H, J=1.8Hz), 8.53 |
| # (d, 1H, J=5.1Hz), 8.69(s, 1H). |
| 74 |
|
65% |
1.36(t, 3H, J=7.0Hz), 1.58-1.78 (m, 2H), 1.80-1.89(m, 2H), 2.04- 2.16(m, 2H), 2.95-3.05(m, 2H), 3.52-3.66(m, 1H), 3.57(s, 1H), 3.85-3.97(m, 2H), 3.89(s, 6H), 3.92(s, 6H), 3.93(s, 6H), 4.44(s, 2H), 6.67-6.80(m, 6H), 7.13-7.18 (m, 3H), 7.25-7.31(m, 1H), 7.37 |
| # (dd, 1H, J=7.6Hz, 7.6Hz), 7.41- 7.48(m, 2H), 7.55(s, 1H), 8.53 (d, 1H, J=4.9Hz). |
| 75 |
|
42% |
1.36(t, 3H, J=7.0Hz), 1.74-2.34 (m, 6H), 2.96-3.10(m, 2H), 3.47- 3.73(m, 3H), 3.87-3.98(m, 2H), 3.88(s, 3H), 3.90(s, 9H), 3.97(s, 6H), 4.44(s, 2H), 6.65(s, 2H), 6.74-6.82(m, 4H), 7.18-7.32(m, 4H), 7.67(s, 1H), 8.49(d, 1H, J=1.6Hz), 8.57-8.65(m, 2H). |
| 76 |
|
43% |
1.36(t, 3H, J=6.8Hz), 1.63-1.96 (m, 4H), 2.00-2.26(m, 2H), 2.92- 3.03(m, 2H), 3.44-3.66(m, 3H), 3.86-3.96(m, 2H), 3.88(s, 3H), 3.89(s, 6H), 3.90(s, 3H), 3.93(s, 6H), 4.44(s, 2H), 6.65(s, 2H), 6.72-6.80(m, 6H), 7.67(s, 1H), 7.77(br, 1H), 8.47-8.53(m, 2H), |
| # 8.62(d, 1H, J=1.9Hz), 8.70(s, 1H). |
| 77 |
|
82% |
1.35(t, 3H, J=6.8Hz), 1.70-1.82 (m, 2H), 1.84-1.92(m, 2H), 2.10- 2.19(m, 2H), 2.92-3.00(m, 2H), 3.52-3.65(m, 3H), 3.88(s, 3H), 3.89(s, 6H), 3.90(s, 3H), 3.93(q, 2H, J=7.1Hz), 3.96(s, 6H), 4.47 (s, 2H), 6.70(s, 2H), 6.73(d, 2H, J=9.3Hz), 6.77(d, 2H, J=9.3Hz), |
| # 7.18-7.28(m, 4H), 7.33(dd, 1H, J=7.3Hz, 7.3Hz), 7.37(d, 1H, J=7.6Hz), 7.43(s, 1H), 7.59(s, 1H), 8.58(d, 1H, J=4.9Hz). |
| 78 |
|
61% |
1.35(t, 3H, J=6.9Hz), 1.58-1.80 (m, 2H), 1.82-1.91(m, 2H), 2.09- 2.18(m, 2H), 2.93-3.20(m, 2H), 3.56-3.65(m, 1H), 3.58(s, 2H), 3.87(s, 3H), 3.89(s, 6H), 3.89(s, 3H), 3.91-3.94(m, 2H), 3.93(s, 6H), 4.45(s, 2H), 6.69(s, 2H), 6.71-6.78(m, 6H), 7.23-7.28(m, |
| # 1H), 7.32(t, 1H, J=7.5Hz), 7.36 (d, 1H, J=7.6Hz), 7.42(s, 1H), 7.77(s, 1H), 8.49(d, 1H, J=1.8 Hz), 8.69(d, 1H, J=1.8Hz). |
| 79 |
|
73% |
1.35(t, 3H, J=6.8Hz), 1.68-1.80 (m, 2H), 1.81-1.89(m, 2H), 2.06- 2.14(m, 2H), 2.96-3.03(m, 2H), 3.57(s, 2H), 3.57-3.65(m, 1H), 3.87(s, 3H), 3.89(s, 9H), 3.91- 3.96(m, 2H), 3.92(s, 6H), 4.46(s, 2H), 6.69-6.79(m, 9H), 7.23-7.47 (m, 7H). |
| |
PREPARATION EXAMPLE 108
Synthesis of 1-(tert-butoxycarbonyl)-4-(4-butoxyphenylamino)piperidine
1-(tert-butoxycarbonyl)-4-piperidone (5.00 g) and 4-butoxyaniline (3.95 g) was treated in the same manner as described in Preparation Example 37 to give brown powder of the title compound.
Yield: 6.91 g (83%). 1H-NMR (400 MHz, CDCl3) δ: 0.96 (t, 3H, J=7.2 Hz), 1.23-1.35 (m, 2H), 1.42-1.53 (m, 2H), 1.46 (s, 9H), 1.68-1.76 (m, 2H), 1.97-2.05 (m, 2H), 2.84-2.95 (m, 2H), 3.28-3.37 (m, 1H), 3.88 (t, 2H, J=6.6 Hz), 3.96-4.12 (m, 2H), 6.57 (d, 2H, J=9.0 Hz), 6.77 (d, 2H, J=8.8 Hz).
PREPARATION EXAMPLE 109
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-(4-butoxyphenyl)-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]piperidine
1-(tert-Butoxycarbonyl)-4-[(4-butoxyphenyl)amino]piperidine (696 mg) and 4-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (588 mg) was treated in the same manner as described in Example 9 to give light yellow amorphous of the title compound.
Yield: 980 mg (81%). 1H-NMR (400 MHz, CDCl3) δ: 0.95 (t, 3H, J=7.4 Hz), 1.40-1.50 (m, 2H), 1.44 (s, 9H), 1.67-1.76 (m, 2H), 1.82-1.90 (m, 2H), 1.82-1.90 (m, 2H), 2.70-2.82 (m, 2H), 3.61-3.71 (m, 1H), 3.84-3.90 (m, 5H), 3.94 (s, 6H), 4.10-4.28 (m, 2H), 4.39 (s, 2H), 6.74 (d, 2H, J=9.4 Hz), 6.78 (d, 2H, J=9.4 Hz), 7.14-7.18 (m, 3H), 7.56 (s, 1H), 8.54 (d, 1H, J=5.1 Hz).
PREPARATION EXAMPLE 110
Synthesis of 4-[N-(4-butoxyphenyl)-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]piperidine dihydrochloride
1-(tert-Butoxycarbonyl)-4-[N-(4-butoxyphenyl)-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]piperidine (980 mg) was treated in the same manner as described in Preparation Example 94 to give light yellow powder of the title compound.
Yield: 926 mg (99%).
PREPARATION EXAMPLE 111
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-(4-butoxyphenyl)-N-[[3-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]amino]piperidine
1-(tert-Butoxycarbonyl)-4-[(4-butoxyphenyl)amino]piperidine (697 mg) and 5-chloromethyl-3-(3,4,5-trimethoxyphenyl)pyridine (588 mg) was treated in the same manner as described in Example 9 to give light yellow amorphous of the title compound.
Yield: 485 mg (40%). 1H-NMR (400 MHz, CDCl3) δ: 0.95 (t, 3H, J=7.4 Hz), 1.40-1.57 (m, 2H), 1.44 (s, 9H), 1.67-1.75 (m, 2H), 1.82-1.90 (m, 2H), 2.69-2.81 (m, 2H), 3.51-3.60 (m, 1H), 3.87 (q, 2H, J=6.6 Hz), 3.88 (s, 3H), 3.90 (s, 6H), 4.06-4.23 (m, 2H), 4.39 (s, 2H), 6.66 (s, 2H), 6.77 (d, 2H, J=9.2 Hz), 6.81 (d, 2H, J=9.2 Hz), 6.81 (d, 2H, J=9.4 Hz), 7.67 (s, 1H), 8.49 (d, 1H, J=1.8 Hz), 8.62 (d, 1H, J=2.2 Hz).
PREPARATION EXAMPLE 112
Synthesis of 4-[N-(4-butoxyphenyl)-N-[[3-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]amino]piperidine dihydrochloride
1-(tert-Butoxycarbonyl)-4-[N-(4-butoxyphenyl)-N-[[3-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]amino]piperidine (485 mg) was treated in the same manner as described in Preparation Example 94 to give light yellow powder of the title compound.
Yield: 456 mg (98%).
PREPARATION EXAMPLE 113
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-(4-butoxyphenyl)-N-[3-(3,4,5-trimethoxyphenyl)benzyl]amino]piperidine
1-(tert-Butoxycarbonyl)-4-[(4-butoxyphenyl)amino]piperidine (697 mg) and 3-(3,4,5-trimethoxyphenyl)benzyl chloride (586 mg) was treated in the same manner as described in Example 9 to give light yellow amorphous of the title compound.
Yield: 1.17 g (97%). 1H-NMR (400 MHz, CDCl3) δ: 0.95 (t, 3H, J=7.3 Hz), 1.40-1.61 (m, 4H), 1.44 (s, 9H), 1.67-1.75 (m, 2H), 1.83-1.90 (m, 2H), 2.70-2.83 (m, 2H), 3.63-3.72 (m, 2H), 3.87 (q, 2H, J=6.6 Hz), 3.88 (s, 3H), 3.90 (s, 6H), 4.09-4.28 (m, 2H), 4.41 (s, 2H), 6.70 (s, 2H), 6.76 (s, 4H), 7.26 (d, 2H, J=8.0 Hz), 7.33 (t, 1H, J=7.6 Hz), 7.38 (d, 1H, J=7.3 Hz), 7.42 (s, 1H).
PREPARATION EXAMPLE 114
Synthesis of 4-[N-(4-butoxyphenyl)-N-[3-(3,4,5-trimethoxyphenyl)benzyl]amino]piperidine hydrochloride
1-(tert-Butoxycarbonyl)-4-[N-(4-butoxyphenyl)-N-[3-(3,4,5-trimethoxyphenyl)benzyl]amino]piperidine (1.17 g) was treated in the same manner as described in Preparation Example 94 to give light yellow powder of the title compound.
Yield: 1.02 g (98%).
EXAMPLES 80 TO 87
These compounds were obtained by the condensation of amines obtained in Preparation Examples 110, 112 and 114 with chloride derivatives obtained in Preparation Examples 3, 42 and 48. Free bases obtained were then converted to the corresponding hydrochlorides. Yields and NMR data of their free bases are listed below.
| |
| Ex- |
|
|
|
| am- |
|
|
NMR data (400 MHz, measured as |
| ple |
Structure |
Yield |
free bases, CDCl3) δ |
| |
| 80 |
|
63% |
0.95(t, 3H, J=7.3Hz), 1.40-1.51 (m, 2H), 1.66-1.79(m, 2H), 1.83- 1.92(m, 2H), 2.10-2.21(m, 2H), 2.92-3.02(m, 2H), 3.53-3.63(m, 3H), 3.84-3.90(m, 2H), 3.89(s, 3H), 3.93(s, 6H), 3.96(s, 6H), 6.72(d, 2H, J=9.3Hz), 6.77(d, 2H, J=9.3Hz), 7.15(s, 2H), 7.17 |
| # (d, 1H, J=5.1Hz), 7.20(d, 1H, J=6.1Hz), 7.22(s, 2H), 7.57(s, 1H), 7.59(s, 1H), 8.54(d, 1H, J=4.9Hz), 8.59(d, 1H, J=5.1Hz). |
| 81 |
|
44% |
0.95(t, 3H, J=7.4Hz), 1.42-1.51 (m, 2H), 1.67-1.76(m, 4H), 1.80- 1.91(m, 2H), 2.08-2.20(m, 2H), 2.92-3.03(m, 2H), 3.84-3.96(m, 3H), 3.89(s, 3H), 3.90(s, 3H), 3.93(s, 12H), 4.43(s, 2H), 6.69- 6.79(m, 6H), 7.14(s, 2H), 7.16 (d, 1H, J=5.2Hz), 7.55(s, 1H), |
| # 7.76(s, 1H), 8.49(d, 1H, J=1.8 Hz), 8.53(d, 1H, J=5.0Hz), 8.69 (s, 1H). |
| 82 |
|
53% |
0.95(t, 3H, J=7.2Hz), 1.40-1.51 (m, 2H), 1.65-1.78(m, 4H), 1.81- (m, 2H), 2.05-2.18(m, 2H), 1.89(m, 2H), 2.05-2.18(m, 2H), 3.05-3.06(m, 2H), 3.54-3.65(m, 3H), 3.84-3.96(m, 20H), 4.44(s, 2H), 6.70(d, 2H, J=9.2Hz), 6.74- 6.80(m, 4H), 7.11-7.19(m, 3H), |
| # 7.22-7.32(m, 1H), 7.34-7.50(m, 3H), 7.55(s, 1H), 8.53(d, 1H, J=5.1Hz). |
| 83 |
|
42% |
0.95(t, 3H, 7.4Hz), 1.40-1.5 1(m, 2H), 1.67-1.86(m, 6H), 2.03-2.30 (m, 2H), 2.92-3.06(m, 2H), 3.46- 3.56(m, 1H), 3.60(s, 2H), 3.84- 3.91(m, 2H), 3.88(s, 3H), 3.89(s, 6H), 3.90(s, 3H), 3.96(s, 6H), 4.44(s, 2H), 6.65(s, 2H), 6.74- 6.81(m, 4H), 7.20(d, 1H, J=4.9 |
| # Hz), 7.25(s, 2H), 7.67(br, 2H), 8.50(d, 1H, J=1.6Hz), 8.60(d, 1H, J=5.6Hz). |
| 84 |
|
36% |
0.95(t, 3H, J=7.4Hz), 1.40-1.51 (m, 2H), 1.66-1.79(m, 4H), 1.82- 1.92(m, 2H), 2.00-2.22(m, 2H), 2.83-3.06(m, 2H), 3.44-3.67(m, 3H), 3.82-3.97(m, 2H), 3.88(s, 3H), 3.89(s, 6H), 3.90(s, 3H), 3.93(s, 6H), 4.44(s, 2H), 6.65(s, 2H), 6.72-6.80(m, 6H), 7.67(s, |
| # 1H), 7.76(br, 1H), 8.47-8.53(m, 2H), 8.62(d, 1H, J=2.2Hz), 8.70 (s, 1H). |
| 85 |
|
72% |
0.95(t, 3H, J=7.3Hz), 1.40-1.51 (m, 2H), 1.66-1.82(m, 4H), 1.84- (m, 2H), 2.10-2.20(m, 2H), 1.92(m, 2H), 2.10-2.20(m, 2H), 2.92-3.00(m, 2H), 3.53-3.66(m, 3H), 3.83-3.92(m, 2H), 3.88(s, 3H), 3.89(s, 6H), 3.90(s, 3H), 3.96(s, 6H), 4.47(s, 2H), 6.67(s, |
| # 2H), 6.73(d, 2H, J=9.2Hz), 6.77 (d, 2H, J=9.5Hz), 7.18-7.29(m, 4H), 7.33(dd, 1H, J=7.3Hz, 7.3 Hz), 7.37(d, 1H, J=7.6Hz), 7.43 (s, 1H), 7.60(s, 1H), 8.58(d, 1H, J=4.9Hz). |
| 86 |
|
24% |
0.94(t, 3H,J=7.4Hz), 1.41-1.51 (m, 2H), 1.61-1.80(m, 4H), 1.82- 1.92(m, 2H), 2.08-2.19(m, 2H), 2.92-3.02(m, 2H), 3.55-3.65(m, 1H), 3.57(s, 2H), 3.84-3.91(m, 2H), 3.87(s, 3H), 3.88(s, 6H), 3.89(s, 3H), 3.93(s, 6H), 4.45(s, 2H), 6.69(s, 2H), 6.71-6.78(m, |
| # 4H), 6.75(s, 2H), 7.23-7.28(m, 1H), 7.32(t, 1H, J=7.4Hz), 7.36 (d, 1H, J=7.6Hz), 7.42(s, 1H), 7.77(s, 1H), 8.49(d, 1H, J=1.6 Hz), 8.69(s, 1H). |
| 87 |
|
78% |
0.94(t, 3H, J=7.3Hz), 1.40-1.50 (m, 2H), 1.66-1.88(m, 4H), 1.82- 1.89(m, 2H), 2.04-2.16(m, 2H), 2.96-3.03(m, 2H), 3.55-3.65(m, 3H), 3.83-3.90(m, 2H), 3.87(s, 3H), 3.89(s, 9H), 3.92(s, 6H), 4.46(s, 2H), 6.69-6.79(m, 9H), 7.23-7.48(m, 7H). |
| |
PREPARATION EXAMPLE 115
Synthesis of 4-(m-anisidino)-1-(tert-butoxycarbonyl)piperidine
1-(tert-Butoxycarbonyl)-4-piperidone (4.78 g) and m-anisidine (2.96 g) were condensed in the same manner as described in Preparation Example 37 to give the title compound.
Yield: 4.83 g (66%). 1H-NMR (400 MHz, CDCl3) δ: 1.20-1.39 (m, 2H), 1.44 (s, 9H), 1.99-2.05 (m, 2H), 2.89 (dt, 2H, J=13.5 Hz, 2.2 Hz), 3.33-3.44 (m, 1H), 3.75 (s, 3H), 3.96-4.07 (m, 2H), 6.14 (t, 1H, J=2.2 Hz), 6.18-6.29 (m, 2H), 7.05 (t, 1H, J=8.1 Hz).
PREPARATION EXAMPLE 116
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-(3-methoxyphenyl)-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]piperidine
4-(m-Anisidino)-1-(tert-butoxycarbonyl)piperidine (613 mg) and 4-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (588 mg) was treated in the same manner as described in Example 9 to give light yellow amorphous of the title compound.
Yield: 789 mg (70%). 1H-NMR (400 MHz, CDCl3) δ: 1.45 (s, 9H), 1.50-1.67 (m, 2H), 1.82-1.91 (m, 2H), 2.74-2.87 (m, 2H), 3.74 (s, 3H), 3.88-3.98 (m, 1H), 3.89 (s, 3H), 3.94 (s, 6H), 4.14-4.32 (m, 2H), 4.48 (s, 2H), 6.28 (dd, 1H, J=2.2 Hz, 2.2 Hz), 6.31-6.37 (m, 2H), 7.10-7.15 (m, 2H), 7.16 (s, 2H), 7.55 (s, 1H), 8.56 (d, 1H, J=5.1 Hz).
PREPARATION EXAMPLE 117
Synthesis of 4-[N-(3-methoxyphenyl)-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]piperidine dihydrochloride
1-(tert-Butoxycarbonyl)-4-[N-(3-methoxyphenyl)-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]piperidine (789 mg) was treated in the same manner as described in Preparation Example 94 to give light yellow powder of the title compound.
Yield: 710 mg (95%).
PREPARATION EXAMPLE 118
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-(3-methoxyphenyl)-N-[[3-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]amino]piperidine
4-(m-Anisidino)-1-(tert-butoxycarbonyl)piperidine (613 mg) and 5-chloromethyl-3-(3,4,5-trimethoxyphenyl)pyridine (588 mg) was treated in the same manner as described in Example 9 to give light yellow amorphous of the title compound.
Yield: 396 mg (35%). 1H-NMR (400 MHz, CDCl3) δ: 1.45 (s, 9H), 1.54-1.66 (m, 2H), 1.81-1.91 (m, 2H), 2.73-2.87 (m, 2H), 3.74 (s, 3H), 3.87-3.93 (m, 1H), 3.88 (s, 3H), 3.90 (s, 6H), 4.14-4.29 (m, 2H), 4.51 (s, 2H), 6.30-6.35 (m, 2H), 6.38 (d, 1H, J=7.2 Hz), 6.68 (s, 2H), 7.12 (dd, 1H, J=8.8 Hz, 8.8 Hz), 7.66 (s, 1H), 8.49 (d, 1H, J=2.0 Hz), 8.66 (d, 1H, J=2.2 Hz).
PREPARATION EXAMPLE 119
Synthesis of 4-[N-(3-methoxyphenyl)-N-[[3-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]amino]piperidine dihydrochloride
1-(tert-Butoxycarbonyl)-4-[N-(3-methoxyphenyl)-N-[[3-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]amino]piperidine (396 mg) was treated in the same manner as described in Preparation Example 94 to give light yellow powder of the title compound.
Yield: 348 mg (92%).
PREPARATION EXAMPLE 120
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-(3-methoxyphenyl)-N-[3-(3,4,5-trimethoxyphenyl)benzyl]amino]piperidine
4-(m-Anisidino)-1-(tert-butoxycarbonyl)piperidine (613 mg) and 3-(3,4,5-trimethoxyphenyl)benzyl chloride (586 mg) was treated in the same manner as described in Preparation Example 9 to give light yellow amorphous of the title compound.
Yield: 1.01 g (90%). 1H-NMR (400 MHz, CDCl3) δ: 1.44 (s, 9H), 1.56-1.67 (m, 2H), 1.83-1.91 (m, 2H), 2.72-2.86 (m, 2H), 3.73 (s, 3H), 3.85-3.98 (m, 1H), 3.88 (s, 3H), 3.90 (s, 6H), 4.12-4.30 (m, 2H), 4.50 (s, 2H), 6.27-6.34 (m, 2H), 6.38 (dd, 1H, J=8.2 Hz, 2.4 Hz), 6.72 (s, 2H), 7.10 (dd, 1H, J=8.2 Hz, 8.2 Hz), 7.21-7.27 (m, 1H), 7.32-7.43 (m, 3H).
PREPARATION EXAMPLE 121
Synthesis of 4-[N-(3-methoxyphenyl)-N-[3-(3,4,5-trimethoxyphenyl)benzyl]amino]piperidine hydrochloride
1-(tert-Butoxycarbonyl)-4-[N-(3-methoxyphenyl)-N-[3-(3,4,5-trimethoxyphenyl)benzyl]amino]piperidine (1.01 g) was treated in the same manner as described in Preparation Example 94 to give light yellow powder of the title compound.
Yield: 820 mg (92%).
EXAMPLES 88 TO 95
These compounds were obtained by the condensation of amines obtained in Preparation Examples 117, 119 and 121 with chloride derivatives obtained in Preparation Examples 3, 42 and 48. Free bases obtained were then converted to the corresponding hydrochlorides. Yields and NMR data of their free bases are listed below.
| |
| Ex- |
|
|
|
| am- |
|
|
NMR data (400 MHz, measured as |
| ple |
Structure |
Yield |
free bases, CDCl3) δ |
| |
| 88 |
|
63% |
1.70-1.82(m, 2H), 1.83-1.90(m, 2H), 2.14-2.23(m, 21-1), 2.94- 3.01(m, 2H), 3.57(s, 2H), 3.73(s, 3H), 3.76-3.88(m, 1H), 3.89(s, 3H), 3.90(s, 3H), 3.93(s, 6H), 3.96 (s, 6H), 4.53(s, 2H), 6.26-6.35(m, 3H), 7.11(dd, 1H, J=8.3Hz, 8.3 Hz), 7.12-7.14(m, 1H), 7.15(s, |
| # 2H), 7.20(d, 1H, J=5.1Hz), 7.22 (s, 2H), 7.55(s, 1H), 7.58(s, 1H), 8.55(d, 1H, J=4.9Hz), 8.59(d, 1H, J=4.9Hz). |
| 89 |
|
72% |
1.67-1.90(m, 4H), 2.13-2.22(m, 2H), 2.94-3.04(m, 2H), 3.59(s, 2H), 3.74(s, 3H), 3.77-3.87(m, 1H), 3.89(s, 3H), 3.89(s, 3H), 3.92(s, 6H), 3.93(s, 6H), 4.52(s, 2H), 6.27(dd, 1H, J=2.4Hz, 2.4 Hz), 6.29-6.34(m, 2H), 6.75(s, 2H), 7.08-7.17(m, 4H), 7.54(s, |
| # 1H), 7.75(s, 1H), 8.50(d, 1H, J=1.8Hz), 8.54(d, 1H, J=5.1Hz), 8.69(d, 1H, J=2.0Hz). |
| 90 |
|
60% |
1.68-1.90(m, 4H), 2.09-2.19(m, 2H), 2.97-3.06(m, 2H), 3.58(s, 2H), 3.73(s, 3H), 3.76-3.87(m, 1H), 3.89(s, 6H), 3.92(s, 6H), 3.92(s, 6H), 4.52(s, 2H), 6.25- 6.35(m, 3H), 6.76(s, 2H), 6.78- 7.17(m, 4H), 7.25-7.32(m, 1H), 7.37(dd, 1H, J=7.4Hz, 7.4Hz), 7.41-7.47(m, 2H), 7.54(s, 1H), |
| # 8.54(d, 1H, J=5.1Hz). |
| 91 |
|
50% |
1.80-1.93(m, 4H), 2.13-2.32(m, 2H), 2.87-3.10(m, 2H), 3.60(s, 1H), 3.69-3.85(m, 1H), 3.73(s, 3H), 3.88(s, 3H), 3.89(s, 6H), 3.90(s, 3H), 3.96(s, 6H), 4.57(s, 2H), 6.29-6.34(m, 2H), 6.37(dd, 1H, J=8.2Hz, 8.1Hz), 6.67(s, 2H), 7.11(dd, 1H, J=8.6Hz, 8.6 |
| # Hz), 7.20-7.28(m, 3H), 7.58-7.72 (m, 1H), 7.68(s, 1H), 8.50(d, 1H, J=1.8Hz), 8.60(d, 1H, J=4.7Hz), 8.65(d, 1H, J=2.0Hz). |
| 92 |
|
35% |
1.70-1.90(m, 4H), 2.12-2.25(m, 2H), 2.95-3.03(m, 2H), 3.59(s, 2H), 3.72-3.97(m, 1H), 3.73(s, 3H), 3.88(s, 3H), 3.89(s, 6H), 3.90(s, 3H), 3.93(s, 6H), 4.54(s, 2H), 6.25-6.38(m, 2H), 6.36(d, 1H, J=8.4Hz, 8.4Hz), 6.67(s, 2H), 6.75(s, 2H), 7.11(dd, 1H, |
| # J=8.4Hz), 7.66(s, 1H), 8.49(s, 1H), 8.50(d, 1H, J=1.8Hz), 8.64 (d, 1H, J=2.0Hz), 8.70(d, 1H, J=1.9Hz). |
| 93 |
|
86% |
1.73-1.93(m, 4H), 2.13-2.23(m, 2H), 2.94-3.02(m, 2H), 3.57(s, 2H), 3.73(s, 3H), 3.77-3.87(m, 1H), 3.88(s, 3H), 3.88(s, 6H), 3.90(s, 3H), 3.96(s, 6H), 4.56 (s, 2H), 6.27(dd, 1H, J=8.0Hz, 2.2 Hz), 6.31(dd, 1H, J=2.2Hz, 2.2 Hz), 6.36(dd, 1H, J=8.2Hz, 2.2 |
| # Hz), 6.71(s, 2H), 7.09(dd, 1H, J=8.1Hz, 8.1Hz), 7.18-7.28(m, 4H), 7.34(dd, 1H, J=7.4Hz, 7.4 Hz), 7.38(d, 1H, J=7.6Hz), 7.42 (s, 1H), 7.59(s, 1H), 8.59(d, 1H, J=4.9Hz). |
| 94 |
|
56% |
1.72-1.92(m, 4H), 2.10-2.23(m, 2H), 2.92-3.60(m, 2H), 3.59(s, 2H), 3.72(s, 3H), 3.77-3.89(m, 1H), 3.87(s, 3H), 3.88(s, 6H), 3.93(s, 6H), 4.55(s, 2H), 6.27 (dd, 1H, J=8.0Hz, 2.2Hz), 6.31 (dd, 1H, J=2.1Hz, 2.1Hz), 6.36 (dd, 1H, J=8.4Hz, 2.4Hz), 6.70 |
| # (s, 2H), 6.75(s, 2H), 7.09(dd, 1H, J=8.2Hz, 8.2Hz), 7.22(d, 1H, J=7.4Hz), 7.33(dd, 1H, J=7.4Hz, 7.4Hz), 7.38(d, 1H, J=7.8Hz), 7.40(s, 1H), 7.77(s, 1H), 8.50(d, 1H, J=1.8Hz), 8.69(d, 1H, J=1.8 Hz). |
| 95 |
|
77% |
1.66-1.89(m, 4H), 2.08-2.18(m, 2H), 2.95-3.05(m, 2H), 3.58(s, 2H), 3.72(s, 3H), 3.75-3.84(m, 1H), 3.87(s, 3H), 3.88(s, 6H), 3.89(s, 3H), 3.92(s, 6H), 4.55(s, 2H), 6.26(dd, 1H, J=8.0Hz, 2.2 Hz), 6.30(dd, 1H, J=2.2Hz, 2.2 Hz), 6.36(dd, 1H, J=8.3Hz, 2.2 |
| # Hz), 6.70(s, 2H), 6.76(s, 2H), 7.08(dd, 1H, J=8.3Hz, 8.3Hz), 7.22(d, 1H, J=7.3Hz), 7.27-7.47 (m, 7H). |
| |
PREPARATION EXAMPLE 122
Synthesis of 4-(o-anisidino)-1-(tert-butoxycarbonyl)piperidine
1-(tert-Butoxycarbonyl)-4-piperidone (4.78 g) and o-anisidine (2.96 g) were condensed in the same manner as described in Preparation Example 37 to give the title compound.
Yield: 2.61 g (36%). 1H-NMR (400 MHz, CDCl3) δ: 1.31-1.41 (m, 2H), 1.47 (s, 9H), 2.00-2.08 (m, 2H), 2.90-3.01 (m, 2H), 3.38-3.47 (m, 1H), 3.83 (s, 3H), 4.00-4.21 (m, 2H), 6.60-6.69 (m, 2H), 6.76-6.89 (m, 2H).
PREPARATION EXAMPLE 123
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-(2-methoxyphenyl)-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]piperidine
4-(o-Anisidino)-1-(tert-butoxycarbonyl)piperidine (613 mg) and 4-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (588 mg) was treated in the same manner as described in Example 9 to give light yellow amorphous of the title compound.
Yield: 763 mg (68%). 1H-NMR (400 MHz, CDCl3) δ: 1.41-1.58 (m, 2H), 1.44 (s, 9H), 1.81-1.91 (m, 2H), 2.62-2.78 (m, 2H), 3.29 (tt, 1H, J=7.6 Hz, 3.7 Hz), 3.86 (s, 3H), 3.89 (s, 3H), 3.95 (s, 6H), 4.06-4.16 (m, 2H), 4.37 (s, 2H), 6.80 (ddd, 1H, J=7.6 Hz, 7.6 Hz, 1.2 Hz), 6.87 (dd, 1H, J=8.5 Hz, 1.0 Hz), 7.00-7.06 (m, 2H), 7.14 (s, 2H), 7.20 (dd, 1H, J=4.9 Hz, 1.0 Hz), 7.61 (s, 1H), 8.49 (d, 1H, J=4.9 Hz).
PREPARATION EXAMPLE 124
Synthesis of 4-[N-(2-methoxyphenyl)-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]piperidine dihydrochloride
1-(tert-Butoxycarbonyl)-4-[N-(2-methoxyphenyl)-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]piperidine (763 mg) was treated in the same manner as described in Preparation Example 94 to give light yellow powder of the title compound.
Yield: 701 mg (97%).
PREPARATION EXAMPLE 125
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-(2-methoxyphenyl)-N-[[3-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]amino]piperidine
4-(o-Anisidino)-1-(tert-butoxycarbonyl)piperidine (613 mg) and 5-chloromethyl-3-(3,4,5-trimethoxyphenyl)pyridine (588 mg) was treated in the same manner as described in Example 9 to give light yellow amorphous of the title compound.
Yield: 353 mg (31%). 1H-NMR (400 MHz, CDCl3) δ: 1.44 (s, 9H), 1.46-1.53 (m, 2H), 1.82-1.91 (m, 2H), 2.62-2.78 (m, 2H), 3.24-3.33 (m, 1H), 3.83 (s, 3H), 3.89 (s, 3H), 3.91 (s, 6H), 4.03-4.16 (m, 2H), 4.37 (s, 2H), 6.64 (s, 2H), 6.79 (ddd, 1H, J=7.6 Hz, 7.6 Hz, 1.2 Hz), 6.84 (dd, 1H, J=7.0 Hz, 1.2 Hz), 6.97-7.06 (m, 2H), 7.68 (dd, 1H, J=1.3 Hz, 1.3 Hz), 8.49 (d, 1H, J=2.0 Hz), 8.56 (d, 1H, J=2.2 Hz).
PREPARATION EXAMPLE 126
Synthesis of 4-[N-(2-methoxyphenyl)-N-[[3-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]amino]piperidine dihydrochloride
1-(tert-Butoxycarbonyl)-4-[N-(2-methoxyphenyl)-N-[[3-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]amino]piperidine (353 mg) was treated in the same manner as described in Preparation Example 94 to give light yellow powder of the title compound.
Yield: 312 mg (93%).
PREPARATION EXAMPLE 127
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-(2-methoxyphenyl)-N-[3-(3,4,5-trimethoxyphenyl)benzyl]amino]piperidine
4-(o-Anisidino)-1-(tert-butoxycarbonyl)piperidine (613 mg) and 3-(3,4,5-trimethoxyphenyl)benzyl chloride (586 mg) was treated in the same manner as described in Example 9 to give light yellow amorphous of the title compound.
Yield: 1.12 g (100%). 1H-NMR (400 MHz, CDCl3) δ: 1.43 (s, 9H), 1.46-1.57 (m, 2H), 1.81-1.90 (m, 2H), 2.62-2.76 (m, 2H), 3.31 (tt, 1H, J=11.1 Hz, 3.3 Hz), 3.84 (s, 3H), 3.88 (s, 3H), 3.91 (s, 6H), 4.00-4.16 (m, 2H), 4.36 (s, 2H), 6.67 (s, 2H), 6.78 (t, 1H, J=7.3 Hz), 6.85 (d, 1H, J=7.9 Hz), 6.96-7.03 (m, 2H), 7.24-7.34 (m, 3H), 7.43 (s, 1H).
PREPARATION EXAMPLE 128
Synthesis of 4-[N-(2-methoxyphenyl)-N-[3-(3,4,5-trimethoxyphenyl)benzyl]amino]piperidine hydrochloride
1-(tert-Butoxycarbonyl)-4-[N-(2-methoxyphenyl)-N-[3-(3,4,5-trimethoxyphenyl)benzyl]amino]piperidine (1.12 g) was treated in the same manner as described in Preparation Example 94 to give light yellow powder of the title compound.
Yield: 987 mg (99%).
EXAMPLES 96 TO 101
These compounds were obtained by the condensation of amines obtained in Preparation Examples 124, 126 and 128 with chloride derivatives obtained in Preparation Examples 3 and 48. Free bases obtained were then converted to the corresponding hydrochlorides. Yields and NMR data of their free bases are listed below.
| |
| Ex- |
|
|
|
| am- |
|
|
NMR data (400 MHz, measured as |
| ple |
Structure |
Yield |
free bases, CDCl3) δ |
| |
| 96 |
|
73% |
1.62-1.74(m, 2H), 1.82-1.90(m, 2H), 1.98-2.08(m, 2H), 2.86-2.94 (m, 2H), 3.13-3.22(m, 1H), 3.52 (s, 2H), 3.85(s, 3H), 3.89(s, 3H), 3.90(s, 3H), 3.94(s, 6H), 3.96(s, 6H), 4.40(s, 2H), 6.80(ddd, 1H, J=7.6Hz, 7.6Hz, 1.2Hz), 6.86 (dd, 1H, J=8.1Hz,1.2Hz), 6.98- |
| # 7.05(m, 1H), 7.14(s, 2H), 7.18 (dd, 1H, J=4.9Hz, 1.2Hz), 7.20- 7.24(m, 1H), 7.22(s, 2H), 7.58(s, 1H), 7.62(s, 1H), 8.49(d, 1H, J=4.9Hz , 8.57(d, 1H, J=5.2Hz). |
| 97 |
|
55% |
1.60-1.73(m, 4H), 1.82-1.93(m, 2H), 1.98-2.07(m, 2H), 2.87-2.97 (m, 2H), 3.12-3.22(m, 1H), 3.54 (s, 2H), 3.85(s, 3H), 3.89(s, 3H), 3.90(s, 3H), 3.93(s, 6H), 3.94(s, 6H), 4.39(s, 2H), 6.75(s, 2H), 6.79(dd, 1H, J=7.4Hz, 7.4Hz), 6.86(d, 1H, J=7.8Hz), 6.97-7.05 |
| # (m, 2H), 7.13(s, 2H), 7.20(d, 1H, J=4.7Hz), 7.61(s, 1H), 7.75(s, 1H), 8.46-8.50(m, 2H), 8.68(d, 1H, J=2.0Hz). |
| 98 |
|
29% |
1.64-1.82(m, 2H), 1.84-1.97(m, 2H), 2.00-2.15(m, 2H), 2.84-3.01 (m, 2H), 3.13-3.27(m, 1H), 3.56 (s, 2H), 3.82(s, 3H), 3.88(s, 3H), 3.90(s, 3H), 3.91(s, 6H), 3.96(s, 6H), 4.40(s, 2H), 6.63(s, 2H), 6.75-6.88(m, 2H), 6.97-7.04(m, 2H), 7.19(d, 1H, J=4.3Hz), 7.25 |
| # (s, 2H), 7.58-7.73(m, 2H), 8.50 (d, 1H, J=1.6Hz), 8.56(d, 1H, J=2.2Hz), 8.58(d, 1H, J=4.9Hz). |
| 99 |
|
30% |
1.62-1.75(m, 2H), 1.83-1.94(m, 2H), 1.95-2.11(m, 2H), 2.84-3.01 (m, 2H), 3.12-3.23(m, 1H), 3.55 (s, 2H), 3.82(s, 3H), 3.88(s, 3H), 3.90(s, 3H), 3.90(s, 6H), 3.93(s, 6H), 4.39(s, 2H), 6.63(s, 2H), 6.70-6.86(m, 4H), 6.94-7.06(m, 2H), 7.68(s, 1H), 7.76(s, 1H), |
| # 8.47(d, 1H, J=1.7Hz), 8.49(d, 1H, J=1.7Hz), 8.55(d, 1H, J=2.2 Hz), 8.69(s, 1H). |
| 100 |
|
67% |
1.64-1.79(m, 2H), 1.85-1.93(m, 2H), 1.99-2.09(m, 2H), 2.86-2.95 (m, 2H), 3.16-3.26(m, 1H), 3.52 (s, 2H), 3.84(s, 3H), 3.88(s, 3H), 3.90(s, 6H), 3.96(s, 6H), 4.40(s, 2H), 6.67(s, 2H), 6.78(dd, 1H, J=7.4Hz, 7.4Hz), 6.85(d, 1H, J=8.2Hz), 6.97(dd, 1H, J=7.8Hz, |
| # 7.8Hz), 7.02(dd, 1H, J=7.8, 1.6 Hz), 7.17-7.33(m, 6H), 7.44(s, 1H), 7.59(s, 1H), 8.57(d, 1H, J=5.1Hz). |
| 101 |
|
55% |
1.62-1.77(m, 2H), 1.82-1.94(m, 2H), 1.98-2.08(m, 2H), 2.86-2.96 (m, 2H), 3.16-3.26(m, 1H), 3.54 (s, 2H), 3.83(s, 3H), 3.87(s, 3H), 3.90(s, 9H), 3.93(s, 6H), 4.39(s, 2H), 6.66(s, 2H), 6.73-6.80(m, 3H), 6.84(d, 1H, J=7.8Hz), 6.97 (dd, 1H, J=7.8Hz, 7.8Hz), 7.01 |
| # (d, 1H, J=7.8Hz), 7.23-7.32(m, 3H), 7.43(s, 1H), 7.77(s, 1H), 8.47(d, 1H, J=1.4Hz), 8.68(d, 1H, J=1.8Hz). |
| |
PREPARATION EXAMPLE 129
Synthesis of 1-(tert-butoxycarbonyl)-4-[(2,3-dimethoxyphenyl)amino]piperidine
1-(tert-Butoxycarbonyl)-4-piperidone (4.78 g) and 2,3-dimethoxyaniline (3.68 g) were condensed in the same manner as described in Preparation Example 37 to give the title compound.
Yield: 3.18 g (39%). 1H-NMR (400 MHz, CDCl3) δ: 1.29-1.42 (m, 2H), 1.45 (s, 9H), 1.97-2.03 (m, 2H), 2.92 (dt, 2H, J=13.5 Hz, 2.2 Hz), 3.38 (dt, 1H, J=13.8 Hz, 4.1 Hz), 3.77 (s, 3H), 3.82 (s, 3H), 3.99-4.03 (m, 2H), 4.17 (m, 1H), 6.27-6.32 (m, 2H), 6.88 (t, 1H, J=8.4 Hz).
PREPARATION EXAMPLE 130
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-(2,3-dimethoxyphenyl)-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]piperidine
1-(tert-Butoxycarbonyl)-4-[(2,3-dimethoxyphenyl)amino]piperidine (673 mg) and 4-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (588 mg) was treated in the same manner as described in Example 9 to give light yellow amorphous of the title compound.
Yield: 613 mg (52%). 1H-NMR (400 MHz, CDCl3) δ: 1.45 (s, 9H), 1.56-1.70 (m, 2H), 1.84-1.91 (m, 2H), 2.62-2.76 (m, 2H), 3.58 (tt, 1H, J=11.8 Hz, 3.6 Hz), 3.83 (s, 3H), 3.89 (s, 6H), 3.93 (s, 6H), 4.08-4.25 (m, 2H), 4.35 (s, 2H), 6.56-6.63 (m, 2H), 6.86 (t, 1H, J=8.3 Hz), 7.14 (s, 2H), 7.17 (dd, 1H, J=5.1 Hz, 1.2 Hz), 7.62 (s, 1H), 8.50 (d, 1H, J=5.1 Hz).
PREPARATION EXAMPLE 131
Synthesis of 4-[N-(2,3-dimethoxyphenyl)-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]piperidine dihydrochloride
1-(tert-Butoxycarbonyl)-4-[N-(2,3-dimethoxyphenyl)-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]piperidine (613 mg) was treated in the same manner as described in Preparation Example 94 to give light yellow powder of the title compound.
Yield: 512 mg (88%).
EXAMPLE 102
Synthesis of 4-[N-(2,3-dimethoxyphenyl)-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine trihydrochloride
4-[N-(2,3-Dimethoxyphenyl)-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]piperidine dihydrochloride (113 mg) and 4-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (59 mg) were condensed in the same manner as described in Example 9. The title compound was obtained as light yellow powder after converting a free base to a trihydrochloride.
Yield: 21 mg (12%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.76-1.96 (m, 4H), 2.00-2.13 (m, 2H), 2.86-3.00 (m, 2H), 3.42-3.60 (m, 1H), 3.54 (s, 2H), 3.82 (s, 3H), 3.88 (s, 3H), 3.90 (s, 3H), 3.97 (s, 6H), 4.41 (s, 2H), 6.57 (d, 1H, J=8.0 Hz), 6.62 (d, 1H, J=8.2 Hz), 6.85 (dd, 1H, J=8.4 Hz, 8.4 Hz), 7.11-7.29 (m, 6H), 7.59 (s, 1H), 7.63 (s, 1H), 8.50 (d, 1H, J=4.9 Hz), 8.59 (d, 1H, J=4.9 Hz).
PREPARATION EXAMPLE 132
Synthesis of 1-(tert-butoxycarbonyl)-4-[[4-(trifluoromethoxy)phenyl]amino]piperidine
1-(tert-Butoxycarbonyl)-4-piperidone (5.00 g) and 4-(trifluoromethoxy)aniline (4.23 g) was treated in the same manner as described in Preparation Example 37 to give white powder of the title compound.
Yield: 5.22 g (60%). 1H-NMR (400 MHz, CDCl3) δ: 1.25-1.40 (m, 2H), 1.47 (s, 9H), 1.98-2.08 (m, 2H), 2.83-2.98 (m, 2H), 3.34-3.43 (m, 1H), 3.97-4.12 (m, 2H), 6.58 (d, 2H, J=8.8 Hz), 7.03 (d, 2H, J=8.8 Hz).
PREPARATION EXAMPLE 133
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-[4-(trifluoromethoxy)phenyl]-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]piperidine
1-(tert-Butoxycarbonyl)-4-[[4-(trifluoromethoxy)phenyl]amino]piperidine (721 mg) and 4-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (588 mg) was treated in the same manner as described in Example 9 to give light yellow amorphous of the title compound.
Yield: 543 mg (44%). 1H-NMR (400 MHz, CDCl3) δ: 1.45 (s, 9H), 1.52-1.66 (m, 2H), 1.81-1.91 (m, 2H), 2.73-2.88 (m, 2H), 3.88-3.99 (m, 1H), 3.89 (s, 3H), 3.93 (s, 6H), 4.15-4.34 (m, 2H), 4.48 (s, 2H), 6.68 (d, 2H, J=9.2 Hz), 7.07 (d, 2H, J=8.6 Hz), 7.12 (dd, 1H, J=5.2 Hz, 1.3 Hz), 7.15 (s, 2H), 7.52 (s, 1H), 8.58 (d, 1H, J=5.2 Hz).
PREPARATION EXAMPLE 134
Synthesis of 4-[N-[4-(trifluoromethoxy)phenyl]-N-[[2-(3,4,5-trimethoxyphenyl) pyridin-4-yl]methyl]amino]piperidine dihydrochloride
1-(tert-Butoxycarbonyl)-4-[N-[4-(trifluoromethoxy)phenyl]-N-[[2-(3,4,5-trime thoxyphenyl)pyridin-4-yl]methyl]amino]piperidine (543 mg) was treated in the same manner as described in Preparation Example 94 to give light yellow powder of the title compound.
Yield: 481 mg (93%).
PREPARATION EXAMPLE 135
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-[4-(trifluoromethoxy)phenyl]-N-[[3-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]amino]piperidine
1-(tert-Butoxycarbonyl)-4-[[4-(trifluoromethoxy)phenyl]amino]piperidine (721 mg) and 5-chloromethyl-3-(3,4,5-trimethoxyphenyl)pyridine (588 mg) was treated in the same manner as described in Example 9 to give light yellow amorphous of the title compound.
Yield: 201 mg (16%). 1H-NMR (400 MHz, CDCl3) δ: 1.45 (s, 9H), 1.54-1.67 (m, 2H), 1.82-1.90 (m, 2H), 2.74-2.86 (m, 2H), 3.84-3.91 (m, 1H), 3.88 (s, 3H), 3.89 (s, 6H), 4.16-4.30 (m, 2H), 4.52 (s, 2H), 6.67 (s, 2H), 6.72 (d, 2H, J=9.4 Hz), 7.06 (d, 2H, J=8.4 Hz), 7.64 (t, 1H, J=2.1 Hz), 8.49 (d, 1H, J=2.2 Hz), 8.68 (d, 1H, J=2.1 Hz).
PREPARATION EXAMPLE 136
Synthesis of 4-[N-[4-(trifluoromethoxy)phenyl]-N-[[3-(3,4,5-trimethoxyphenyl) pyridin-5-yl]methyl]amino]piperidine dihydrochloride
1-(tert-Butoxycarbonyl)-4-[N-[4-(trifluoromethoxy)phenyl]-N-[[3-(3,4,5-trime thoxyphenyl)pyridin-5-yl]methyl]amino]piperidine (201 mg) was treated in the same manner as described in Preparation Example 94 to give light yellow powder of the title compound.
Yield: 185 mg (96%).
PREPARATION EXAMPLE 137
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-[4-(trifluoromethoxy)phenyl]-N-[3-(3,4,5-trimethoxyphen yl)benzyl]amino)piperidine
1-(tert-Butoxycarbonyl)-4-[[4-(trifluoromethoxy)phenyl]amino]piperidine (721 mg) and 3-(3,4,5-trimethoxyphenyl)benzyl chloride (586 mg) was treated in the same manner as described in Example 9 to give light yellow amorphous of the title compound.
Yield: 1.06 mg (86%). 1H-NMR (400 MHz, CDCl3) δ: 1.45 (s, 9H), 1.56-1.68 (m, 2H), 1.83-1.90 (m, 2H), 2.71-2.86 (m, 2H), 3.87-3.90 (m, 1H), 3.88 (s, 3H), 3.89 (s, 6H), 4.16-4.29 (m, 2H), 4.51 (s, 2H), 6.70 (d, 2H, J=9.3 Hz), 6.70 (s, 2H), 7.04 (d, 2H, J=8.5 Hz), 7.22 (d, 1H, J=7.8 Hz), 7.34-7.44 (m, 3H).
PREPARATION EXAMPLE 138
Synthesis of 4-[N-[4-(trifluoromethoxy)phenyl]-N-[3-(3,4,5-trimethoxyphenyl)benzyl]amino]piperidine hydrochloride
1-(tert-Butoxycarbonyl)-4-[N-[4-(trifluoromethoxy)phenyl]-N-[3-(3,4,5-trimethoxyphenyl)benzyl]amino]piperidine (1.06 g) was treated in the same manner as described in Preparation Example 94 to give light yellow powder of the title compound.
Yield: 795 mg (84%).
EXAMPLES 103 TO 110
These compounds were obtained by the condensation of amines obtained in Preparation Examples 134, 136 and 138 with chloride derivatives obtained in Preparation Examples 3, 42 and 48. Free bases obtained were then converted to the corresponding hydrochlorides. Yields and NMR data of their free bases are listed below.
| |
| Ex- |
|
|
|
| am- |
|
|
NMR data (400 MHz, measured as |
| ple |
Structure |
Yield |
free bases, CDCl3) δ |
| |
| 103 |
|
70% |
1.71-1.90(m, 4H), 2.15-2.23(m, 2H), 2.95-3.02(m, 2H) 3.58(s, 2H), 3.76-3.85(m, 1H), 3.89(s, 3H), 3.90(s, 3H), 3.92(s, 6H), 3.96(s, 6H), 4.54(s, 2H), 6.66(d, 2H, J=9.3Hz), 7.05(d, 2H, J=8.5 Hz), 7.13(dd, 1H, J=5.1Hz, 1.2 Hz), 7.14(s, 2H), 7.20(dd, 1H, |
| # J=4.9Hz, 1.2Hz), 7.22(s, 2H), 7.53(s, 1H), 7.59(s, 1H), 8.57(d, 1H, J=4.9Hz), 8.59(d, 1H, J=5.2 Hz). |
| 104 |
|
48% |
1.68-1.92(m, 4H), 2.13-2.25(m, 2H), 2.95-3.06(m, 2H), 3.60(s, 2H), 3.75-3.87(m, 1H), 3.89(s, 3H), 3.90(s, 3H), 3.91(s, 6H), 3.93(s, 6H), 4.52(s, 2H), 6.65(d, 2H, J=9.4Hz), 6.75(s, 2H), 7.05 (d, 2H, J=9.2Hz), 7.12(d, 1H, J=5.1Hz), 7.14(s, 2H), 7.52(s, |
| # 1H), 7.76(s, 1H), 8.51(d, 1H, J=1.8Hz), 8.57(d, 1H, J=5.1Hz), 8.70(d, 1H, J=2.1Hz). |
| 105 |
|
69% |
1.70-1.89(m, 4H), 2.10-2.19(m, 2H), 2.98-3.08(m, 2H), 3.59(s, 2H), 3.72-3.84(m, 1H), 3.89(s, 6H), 3.92(s, 6H), 3.92(s, 6H), 4.52(s, 2H), 6.65(d, 2H, J=9.4 Hz), 6.76(s, 2H), 7.04(d, 2H, J=8.6Hz), 7.11(d, 1H, J=5.1Hz), 7.14(s, 2H), 7.25-7.33(m, 1H), |
| # 7.37(dd, 1H, J=7.4Hz, 7.4Hz), 7.41-7.48(m, 2H), 7.51(s, 1H), 8.56(d, 1H, J=5.1Hz). |
| 106 |
|
41% |
1.73-1.93(m, 4H), 2.12-2.26(m, 2H), 2.93-3.07(m, 2H), 3.53-3.65 (m, 2H), 3.74-3.84(m, 1H), 3.88 (s, 9H), 3.90(s, 3H), 3.96(s, 6H), 4.58(s, 2H), 6.66(s, 2H), 6.69(d, 2H, J=9.2Hz), 7.05(d, 2H, J=8.8 Hz), 7.18-7.29(m, 3H), 7.59(br, 1H), 7.64(s, 1H), 8.49(s, 1H), |
| # 8.60(d, 1H, J=5.3Hz), 8.67(d, 1H, J=2.0Hz). |
| 107 |
|
28% |
1.72-1.91(m, 4H), 2.12-2.28(m, 2H), 2.94-3.06(m, 2H), 3.60(s, 2H), 3.76-3.82(m, 1H), 3.88(s, 9H), 3.90(s, 3H), 3.93(s, 6H), 4.56(s, 2H), 6.65(s, 2H), 6.69(d, 2H, J=9.2Hz), 6.75(s, 2H), 7.05 (d, 2H, J=8.8Hz), 7.63(s, 1H), 7.76(s, 1H), 8.48(d, 1H, J=1.8 |
| # Hz), 8.51(d, 1H, J=1.8Hz), 8.66 (d, 1H, J=2.2Hz), 8.70(d, 1H, J=2.2Hz). |
| 108 |
|
78% |
1.76-1.91(m, 4H), 2.14-2.23(m, 2H), 2.94-3.03(m, 2H), 3.57(s, 2H), 3.75-3.84(m, 1H), 3.87(s, 9H), 3.90(s, 3H), 3.96(s, 6H), 4.56(s, 2H), 6.65-6.72(m, 4H), 7.03(d, 2H, J=8.8Hz), 7.18-7.24 (m, 4H), 7.33-7.43(m, 3H), 7.59 (s, 1H), 8.59(d, 1H, J=4.9Hz). |
| 109 |
|
5% |
1.72-1.90(m, 4H), 2.12-2.21(m, 2H), 2.94-3.03(m, 2H), 3.59(s, 2H), 3.73-3.86(m, 1H), 3.87(s, 9H), 3.90(s, 3H), 3.93(s, 6H), 4.54(s, 2H), 6.66-6.70(m, 4H), 6.75(s, 2H), 7.03(d, 2H, J=9.0 Hz), 7.21(d, 1H, J=7.2Hz), 7.32- 7.41(m, 3H), 7.76(s, 1H), 8.50 |
| # (d, 1H, J=1.6Hz), 8.69(d, 1H, J=1.6Hz). |
| 110 |
|
62% |
1.72-1.89(m, 4H), 2.08-2.20(m, 2H), 2.97-3.07(m, 2H), 3.59(s, 2H), 3.73-3.83(m, 1H), 3.87(s, 9H), 3.89(s, 3H), 3.92(s, 6H), 4.55(s, 2H), 6.67(d, 2H, J=9.3 Hz), 6.69(s, 2H), 6.76(s, 2H), 7.02(d, 2H, J=8.6Hz), 7.20(d, 1H, J=7.6 Hz), 7.25-7.47(m, 7H). |
| |
PREPARATION EXAMPLE 139
Synthesis of 1-(tert-butoxycarbonyl)-4-[[4-(methylthio)phenyl]amino]piperidine
1-(tert-Butoxycarbonyl)-4-piperidone (5.00 g) and 4-(methylthio)aniline (3.33 g) was treated in the same manner as described in Preparation Example 37 to give white powder of the title compound.
Yield: 3.80 g (49%). 1H-NMR (400 MHz, CDCl3) δ: 1.26-1.38 (m, 2H), 1.46 (s, 9H), 1.98-2.06 (m, 2H), 2.41 (s, 3H), 2.88-2.97 (m, 2H), 3.36-3.45 (m, 2H), 3.48-3.56 (br, 1H), 3.96-4.12 (m, 2H), 6.55 (d, 2H, J=8.8 Hz), 7.21 (d, 2H, J=8.8 Hz).
PREPARATION EXAMPLE 140
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-[4-(methylthio)phenyl]-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]piperidine
1-(tert-Butoxycarbonyl)-4-[[4-(methylthio)phenyl]amino]piperidine (644 mg) and 4-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (588 mg) was treated in the same manner as described in Example 9 to give light yellow amorphous of the title compound.
Yield: 671 mg (58%). 1H-NMR (400 MHz, CDCl3) δ: 1.45 (s, 9H), 1.50-1.66 (m, 2H), 1.81-1.89 (m, 2H), 2.40 (s, 3H), 2.74-2.87 (m, 2H), 3.88-3.94 (m, 1H), 3.90 (s, 3H), 3.94 (s, 6H), 4.15-4.29 (m, 2H), 4.48 (s, 2H), 6.67 (d, 2H, J=9.0 Hz), 7.11-7.18 (m, 1H), 7.16 (s, 2H), 7.22 (d, 2H, J=6.6 Hz), 7.54 (s, 1H), 8.57 (d, 1H, J=5.1 Hz).
PREPARATION EXAMPLE 141
Synthesis of 4-[N-[4-(methylthio)phenyl]-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]piperidine dihydrochloride
1-(tert-Butoxycarbonyl)-4-[N-[4-(methylthio)phenyl]-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]piperidine (671 mg) was treated in the same manner as described in Preparation Example 94 to give light yellow powder of the title compound.
Yield: 602 mg (94%).
PREPARATION EXAMPLE 142
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-[4-(methylthio)phenyl]-N-[[3-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]amino]piperidine
1-(tert-Butoxycarbonyl)-4-[[4-(methylthio)phenyl]amino]piperidine (645 mg) and 5-chloromethyl-3-(3,4,5-trimethoxyphenyl)pyridine (588 mg) was treated in the same manner as described in Example 9 to give light yellow amorphous of the title compound.
Yield: 312 mg (27%). 1H-NMR (400 MHz, CDCl3) δ: 1.45 (s, 9H), 1.53-1.63 (m, 2H), 1.83-1.89 (m, 2H), 2.40 (s, 3H), 2.73-2.85 (m, 2H), 3.87-3.91 (m, 1H), 3.88 (s, 3H), 3.90 (s, 6H), 4.16-4.30 (m, 2H), 4.50 (s, 2H), 6.67 (s, 2H), 6.71 (d, 2H, J=9.0 Hz), 7.21 (d, 2H, J=9.0 Hz), 7.64 (s, 1H), 8.48 (d, 1H, J=2.2 Hz), 8.66 (d, 1H, J=2.1 Hz).
PREPARATION EXAMPLE 143
Synthesis of 4-[N-[4-(methylthio)phenyl]-N-[[3-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]amino]piperidine dihydrochloride
1-(tert-Butoxycarbonyl)-4-[N-[4-(methylthio)phenyl]-N-[[3-(3,4,5-trimethoxy phenyl)pyridin-5-yl]methyl]amino]piperidine (312 mg) was treated in the same manner as described in Preparation Example 94 to give light yellow powder of the title compound.
Yield: 251 mg (84%).
PREPARATION EXAMPLE 144
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-[4-(methylthio)phenyl]-N-[3-(3,4,5-trimethoxyphenyl)benzyl]amino]piperidine
1-(tert-Butoxycarbonyl)-4-[[4-(methylthio)phenyl]amino]piperidine (645 mg) and 3-(3,4,5-trimethoxyphenyl)benzyl chloride (586 mg) was treated in the same manner as described in Example 9 to give light yellow amorphous of the title compound.
Yield: 1.10 g (95%). 1H-NMR (400 MHz, CDCl3) δ: 1.45 (s, 9H), 1.55-1.68 (m, 2H), 1.81-1.90 (m, 2H), 2.39 (s, 3H), 2.73-2.86 (m, 2H), 3.87-3.91 (m, 1H), 3.88 (s, 3H), 3.89 (s, 6H), 4.15-4.29 (m, 2H), 4.50 (s, 2H), 6.68-6.73 (m, 4H), 7.19-7.24 (m, 3H), 7.33-7.43 (m, 3H).
PREPARATION EXAMPLE 145
Synthesis of 4-[N-[4-(methylthio)phenyl]-N-[3-(3,4,5-trimethoxyphenyl)benzyl]amino]piperidine hydrochloride
1-(tert-Butoxycarbonyl)-4-[N-[4-(methylthio)phenyl]-N-[3-(3,4,5-trimethoxyphenyl)benzyl]amino]piperidine (1.10 g) was treated in the same manner as described in Preparation Example 94 to give light yellow powder of the title compound.
Yield: 866 mg (89%).
EXAMPLES 111 TO 118
These compounds were obtained by the condensation of amines obtained in Preparation Examples 141, 143 and 145 with chloride derivatives obtained in Preparation Examples 3, 42 and 48. Free bases obtained were then converted to the corresponding hydrochlorides. Yields and NMR data of their free bases are listed below.
| |
| Ex- |
|
|
|
| am- |
|
|
NMR data (400 MHz, measured |
| ple |
Structure |
Yield |
as free bases, CDCl3) δ |
| |
| 111 |
|
40% |
1.70-1.90(m, 4H), 2.14-2.26(m, 2H), 2.40(s, 3H), 2.94-3.04(m, 2H), 3.58(s, 2H), 3.76-3.88(m, 1H), 3.89(s, 3H), 3.90(s, 3H), 3.93(s, 6H), 3.96(s, 6H), 4.53(s, 2H), 6.66(d, 2H, J=9.0Hz), 7.11- 7.24(m, 8H), 7.54(s, 1H), 7.59(s, 1H), 8.56(d, 1H, J=5.1Hz), 8.59 |
| # (d, 1H, J=5.1Hz). |
| 112 |
|
53% |
1.66-1.90(m, 4H), 2.12-2.24(m, 2H), 2.40(s, 3H), 2.94-3.05(m, 2H), 3.59(s, 2H), 3.73-3.88(m, 1H), 3.89(s, 3H), 3.90(s, 3H), 3.92(s, 6H), 3.93(s, 6H), 4.51(s, 2H), 6.65(d, 2H, J=8.8Hz), 6.75 (s, 2H), 7.12(d, 1H, J=4.9Hz), 7.14(s, 2H), 7.21(d, 2H, J=8.8 |
| # Hz), 7.53(s, 1H), 7.76(s, 1H), 8.50(d, 1H, J=1.9Hz), 6.55(d, 1H, J=4.9Hz), 8.69(d, 1H, J=1.4 Hz). |
| 113 |
|
53% |
1.68-1.89(m, 4H), 2.10-2.20(m, 2H), 2.39(s, 3H), 2.98-3.07(m, 2H), 3.58(s, 2H), 3.75-3.87(m, 1H), 3.89(s, 6H), 3.92(s, 6H), 3.92(s, 6H), 4.51(s, 2H), 6.65(d, 2H, J=9.0Hz), 6.76(s, 2H), 7.11 (d, 1H, J=5.1Hz), 7.14(s, 2H), 7.21(d, 2H, J=8.8Hz), 7.29(d, |
| # 1H, J=7.4Hz), 7.37(dd, 1H, J=7.6 Hz, 7.6Hz), 7.42-7.49(m, 2H), 7.52(s, 1H), 8.54(d, 1H, J=4.9 Hz). |
| 114 |
|
50% |
1.57-2.00(m, 4H), 2.12-2.30(m, 2H), 2.39(s, 3H), 2.90-3.13(m, 2H), 3.50-3.74(m, 2H), 3.75-3.86 (m, 1H), 3.88(s, 3H), 3.89(s, 3H), 3.90(s, 6H), 3.97(s, 6H), 4.57(s, 2H), 6.66(s, 2H), 6.70(d, 2H, J=9.0Hz), 7.17-7.30(m, 5H), 7.66(br, 2H), 8.48(s, 1H), 8.58- |
| # 8.70(m, 2H). |
| 115 |
|
59% |
1.68-1.92(m, 4H), 2.12-2.27(m, 2H), 2.39(s, 3H), 2.94-3.08(m, 2H), 3.60(s, 2H), 3.74-3.83(m, 1H), 3.88(s, 3H), 3.89(s, 6H), 3.90(s, 3H), 3.93(s, 6H), 4.55(s, 2H), 6.66(s, 2H), 6.69(d, 2H, J=8.8Hz), 6.73-6.80(m, 2H), 7.20 (d, 2H, J=8.8Hz), 7.64(s, 1H), |
| # 7.77(br, 1H), 8.48(s, 1H), 8.50(s, 1H), 8.65(s, 1H), 8.71(s, 1H). |
| 116 |
|
85% |
1.76-1.93(m, 4H), 2.14-2.24(m, 2H), 2.39(s, 3H), 2.94-3.03(m, 2H), 3.57(s, 2H), 3.76-3.86(m, 1H), 3.88(s, 6H), 3.90(s, 3H), 3.96(s, 6H), 4.55(s, 2H), 6.67- 6.73(m, 4H), 7.18-7.29(m, 6H), 7.34(dd, 1H, J=7.6Hz, 7.6Hz), 7.37-7.44(m, 2H), 7.59(s, 1H), |
| # 8.59(d, 1H, J=4.9Hz). |
| 117 |
|
53% |
1.72-1.90(m, 4H), 2.12-2.22(m, 2H), 2.39(s, 3H), 2.95-3.05(m, 2H), 3.59(s, 2H), 3.74-3.85(m, 1H), 3.87(s, 3H), 3.88(s, 6H), 3.89(s, 3H), 3.93(s, 6H), 4.54(s, 2H), 6.67-6.70(m, 4H), 6.75(s, 2H), 7.19-7.23(m, 3H), 7.33(dd, 1H, J=7.4Hz, 7.4Hz), 7.36-7.40 |
| # (m, 2H), 7.76(s, 1H), 8.50(d, 1H, J=1.8Hz), 8.69(s, 1H). |
| 118 |
|
83% |
1.72-1.90(m, 4H), 2.09-2.20(m, 2H), 2.38(s, 3H), 2.97-3.06(m, 2H), 3.58(s, 2H), 3.73-3.84(m, 1H), 3.87(s, 3H), 3.88(s, 3H), 3.89(s, 6H), 3.92(s, 6H), 4.54(s, 2H), 6.66-6.71(m, 4H), 6.76(s, 2H), 7.18-7.24(m, 3H), 7.26-7.48 (m, 7H). |
| |
PREPARATION EXAMPLE 146
Synthesis of 1-(tert-butoxycarbonyl)-4-[(4-methylphenyl)amino]piperidine
1-(tert-Butoxycarbonyl)-4-piperidone (5.00 g) and p-toluidine (2.56 g) was treated in the same manner as described in Preparation Example 37 to give white powder of the title compound.
Yield: 5.79 g (83%). 1H-NMR (400 MHz, CDCl3) δ: 1.25-1.36 (m, 2H), 1.46 (s, 9H), 1.99-2.06 (m, 2H), 2.23 (s, 3H), 2.86-2.96 (m, 2H), 3.30-3.43 (m, 2H), 3.96-4.10 (m, 2H), 6.53 (d, 2H, J=8.4 Hz), 6.98 (d, 2H, J=8.0 Hz).
PREPARATION EXAMPLE 147
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-(4-methylphenyl)-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]piperidine
1-(tert-Butoxycarbonyl)-4-[(4-methylphenyl)amino]piperidine (581 mg) and 4-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (588 mg) was treated in the same manner as described in Example 9 to give light yellow amorphous of the title compound.
Yield: 1.00 g (91%). 1H-NMR (400 MHz, CDCl3) δ: 1.45 (s, 9H), 1.55-1.59 (m, 2H), 1.81-1.90 (m, 2H), 2.23 (s, 3H), 2.72-2.86 (m, 2H), 3.81-3.94 (m, 1H), 3.89 (s, 3H), 3.93 (s, 6H), 4.14-4.30 (m, 2H), 4.45 (s, 2H), 6.66 (d, 2H, J=8.6 Hz), 7.02 (d, 2H, J=8.2 Hz), 7.13-7.16 (m, 3H), 7.55 (s, 1H), 8.55 (d, 1H, J=8.1 Hz).
PREPARATION EXAMPLE 148
Synthesis of 4-[N-(4-methylphenyl)-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]piperidine dihydrochloride
1-(tert-Butoxycarbonyl)-4-[N-(4-methylphenyl)-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]piperidine (1.00 g) was treated in the same manner as described in Preparation Example 94 to give light yellow powder of the title compound.
Yield: 924 mg (97%).
PREPARATION EXAMPLE 149
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-(4-methylphenyl)-N-[[3-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]amino]piperidine
1-(tert-Butoxycarbonyl)-4-[(4-methylphenyl)amino]piperidine (581 mg) and 5-chloromethyl-3-(3,4,5-trimethoxyphenyl)pyridine (588 mg) was treated in the same manner as described in Example 9 to give light yellow amorphous of the title compound.
Yield: 426 mg (39%). 1H-NMR (400 MHz, CDCl3) δ: 1.45 (s, 9H), 1.52-1.70 (m, 2H), 1.82-1.90 (m, 2H), 2.23 (s, 3H), 2.72-2.86 (m, 2H), 3.77-3.86 (m, 1H), 3.88 (s, 3H), 3.90 (s, 6H), 4.10-4.28 (m, 2H), 4.47 (s, 2H), 6.67 (s, 2H), 6.70 (d, 2H, J=8.6 Hz), 7.01 (d, 2H, J=8.2 Hz), 7.67 (dd, 1H, J=2.1 Hz, 2.1 Hz), 8.50 (d, 1H, J=2.0 Hz), 8.64 (d, 1H, J=2.2 Hz).
PREPARATION EXAMPLE 150
Synthesis of 4-[N-(4-methylphenyl)-N-[[3-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]amino]piperidine dihydrochloride
1-(tert-Butoxycarbonyl)-4-[N-(4-methylphenyl)-N-[[3-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]amino]piperidine (426 mg) was treated in the same manner as described in Preparation Example 94 to give light yellow powder of the title compound.
Yield: 400 mg (99%).
PREPARATION EXAMPLE 151
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-(4-methylphenyl)-N-[3-(3,4,5-trimethoxyphenyl)benzyl]amino]piperidine
1-(tert-Butoxycarbonyl)-4-[(4-methylphenyl)amino]piperidine (581 mg) and 3-(3,4,5-trimethoxyphenyl)benzyl chloride (586 mg) was treated in the same manner as described in Example 9 to give light yellow amorphous of the title compound.
Yield: 1.03 g (94%). 1H-NMR (400 MHz, CDCl3) δ: 1.44 (s, 9H), 1.50-1.66 (m, 2H), 1.83-1.90 (m, 2H), 2.23 (s, 3H), 2.72-2.85 (m, 2H), 3.82-3.92 (m, 1H), 3.88 (s, 3H), 3.89 (s, 6H), 4.11-4.30 (m, 2H), 4.47 (s, 2H), 6.68 (d, 2H, J=8.6 Hz), 6.71 (s, 2H), 7.00 (d, 2H, J=8.8 Hz), 7.23-7.27 (m, 1H), 7.32-7.44 (m, 3H).
PREPARATION EXAMPLE 152
Synthesis of 4-[N-(4-methylphenyl)-N-[3-(3,4,5-trimethoxyphenyl)benzyl]amino]piperidine hydrochloride
1-(tert-Butoxycarbonyl)-4-[N-(4-methylphenyl)-N-[3-(3,4,5-trimethoxyphenyl)benzyl]amino]piperidine (1.03 g) was treated in the same manner as described in Preparation Example 94 to give light yellow powder of the title compound.
Yield: 882 mg (97%).
EXAMPLES 119 TO 126
These compounds were obtained by the condensation of amines obtained in Preparation Examples 148, 150 and 152 with chloride derivatives obtained in Preparation Examples 3, 42 and 48. Free bases obtained were then converted to the corresponding hydrochlorides. Yields and NMR data of their free bases are listed below.
| |
| Ex- |
|
|
|
| am- |
|
|
NMR data (400 MHz, measured |
| ple |
Structure |
Yield |
as free bases, CDCl3) δ |
| |
| 119 |
|
66% |
1.70-1.82(m, 2H), 1.83-1.91(m, 2H), 2.13-2.25(m, 2H), 2.23(s, 3H), 2.96-3.02(m, 2H), 3.57(s, 2H), 3.73-3.83(m, 1H), 3.89(s, 3H), 3.90(s, 3H), 3.93(s, 6H), 3.96(s, 6H), 4.50(s, 2H), 6.64 (d, 2H, J=8.8Hz), 7.01(d, 2H, J=8.5Hz), 7.13-7.17(m, 3H), |
| # 7.20(d, 1H, J=4.9Hz), 7.22(s, 2H), 7.56(s, 1H), 7.59(s, 1H), 8.54(d, 1H, J=5.1Hz), 8.59(d, 1H, J=4.9Hz). |
| 120 |
|
41% |
1.60-1.91(m, 4H), 2.12-2.24(m, 2H), 2.23(s, 3H), 2.95-3.05(m, 2H), 3.59(s, 2H), 3.73-3.83(m, 1H), 3.89(s, 3H), 3.89(s, 3H), 3.92(s, 6H), 3.93(s, 6H), 4.49 (s, 2H), 6.63(d, 2H, J=8.6Hz), 6.75(s, 2H), 7.00(d, 2H, J=8.6 Hz), 7.13-7.16(m, 3H), 7.55(s, |
| # 1H), 7.76(s, 1H), 8.50(d, 1H, J=1.8Hz), 8.53(d, 1H, J=5.1 Hz), 8.70(s, 1H). |
| 121 |
|
69% |
1.67-1.80(m, 2H), 1.81-1.89(m, 2H), 2.09-2.20(m, 2H), 2.22(s, 3H), 2.98-3.06(m, 2H), 3.58(s, 2H), 3.72-3.81(m, 1H), 3.88(s, 3H), 3.89(s, 3H), 3.92(s, 6H), 3.92(s, 6H), 4.49(s, 2H), 6.63 (d, 2H, J=8.4Hz), 6.76(s, 2H), 7.00(d, 2H, J=8.6Hz), 7.12- |
| # 7.15(m, 3H), 7.26-7.32(m, 1H), 7.37 (dd, 1H, J=7.6Hz, 7.6Hz), 7.41-7.48(m, 2H), 7.55(s, 1H), 8.53(d, 1H, J=5.0Hz). |
| 122 |
|
47% |
1.55-2.00(m, 4H), 2.12-2.31(m, 2H), 2.22(s, 3H), 2.93-3.10(m, 2H), 3.60(br, 2H), 3.69-3.80(m, 1H), 3.88(s, 3H), 3.89(s, 6H), 3.90(s, 3H), 3.96(s, 6H), 4.53 (s, 2H), 6.66(s, 2H), 6.69(d, 2H, J=8.6Hz), 7.00(d, 2H, J=8.6Hz), 7.19-7.27(m, 4H), |
| # 7.68(s, 1H), 8.50(s, 1H), 8.60 (d, 1H, J=4.9Hz), 8.64(d, 1H, J=2.2Hz). |
| 123 |
|
34% |
1.67-1.98(m, 4H), 2.10-2.38(m, 2H), 2.22(s, 3H), 2.85-3.10(m, 2H), 3.53-3.67(s, 2H), 3.67-3.79 (m, 1H), 3.88(s, 3H), 3.89(s, 6H), 3.90(s, 3H), 3.93(s, 6H), 4.51(s, 2H), 6.66(s, 2H), 6.68 (d, 2H, J=8.8Hz), 6.76(s, 2H), 7.00(d, 2H, J=8.2Hz), 7.67(s, |
| # 1H), 7.77(br, 1H), 8.47-8.53(m, 2H), 8.63(d, 1H, J=2.0Hz), 8.70(s, 1H). |
| 124 |
|
91% |
1.73-1.92(m, 4H), 2.12-2.26(m, 2H), 2.21(s, 3H), 2.92-3.02(m, 2H), 3.57(s, 2H), 3.72-3.82(m, 1H), 3.87(s, 3H), 3.88(s, 6H), 3.90(s, 3H), 3.95(s, 6H), 4.53 (s, 2H), 6.67(d, 2H, J=7.8Hz), 6.70(s, 2H), 6.99(d, 2H, J=8.0 Hz), 7.18-7.25(m, 4H), 7.33 |
| # (dd, 1H,J=7.4Hz, 7.4Hz), 7.38 (d, 1H, J=7.2Hz), 7.42(s, 1H), 7.59(s, 1H), 8.58(d, 1H, J=4.7 Hz). |
| 125 |
|
74% |
1.70-1.92(m, 4H), 2.10-2.28(m, 2H), 2.21(s, 3H), 2.92-3.06(m, 2H), 3.58(s, 2H), 3.72-3.82(m, 1H), 3.87(s, 3H), 3.88(s, 6H), 3.89(s, 3H), 3.93(s, 6H), 4.51 (s, 2H), 6.66(d, 2H, J=8.6Hz), 6.70(s, 2H), 6.75(s, 2H), 7.23 (d, 1H, J=7.0Hz), 7.32(dd, 1H, |
| # J=7.6Hz, 7.6Hz), 7.37(d, 1H, J=7.8Hz), 7.41(s, 1H), 7.77(s, 1H), 8.49(s, 1H), 8.69(s, 1H). |
| 126 |
|
84% |
1.71-1.88(m, 4H), 2.08-2.18(m, 2H), 2.21(s, 3H), 2.96-3.04(m, 2H), 3.58(s, 2H), 3.71-3.83(m, 1H), 3.87(s, 3H), 3.88(s, 6H), 3.89(s, 3H), 3.92(s, 6H), 4.52 (s, 2H), 6.66(d, 2H, J=8.6Hz), 6.70(s, 2H), 6.76(s, 2H), 6.98 (d, 2H, J=8.3Hz), 7.22-7.47(m, |
| # 8H). |
| |
PREPARATION EXAMPLE 153
Synthesis of 1-(tert-butoxycarbonyl)-4-[[4-(trifluoromethyl)phenyl]amino]piperidine
1-(tert-Butoxycarbonyl)-4-piperidone (5.00 g) and 4-(trifluoromethyl)aniline (3.85 g) was treated in the same manner as described in Preparation Example 37 to give white powder of the title compound.
Yield: 3.30 g (40%). 1H-NMR (400 MHz, CDCl3) δ: 1.30-1.41 (m, 2H), 1.47 (s, 9H), 2.00-2.07 (m, 2H), 2.88-2.99 (m, 2H), 3.32-3.52 (m, 1H), 3.83-3.89 (m, 1H), 4.00-4.14 (m, 2H), 6.59 (d, 2H, J=8.4 Hz), 7.39 (d, 2H, J=8.4 Hz).
PREPARATION EXAMPLE 154
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-[4-(trifluoromethyl)phenyl]-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]piperidine
1-(tert-Butoxycarbonyl)-4-[[4-(trifluoromethyl)phenyl]amino]piperidine (688 mg) and 4-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (588 mg) was treated in the same manner as described in Example 9 to give light yellow amorphous of the title compound.
Yield: 412 mg (34%). 1H-NMR (400 MHz, CDCl3) δ: 1.45 (s, 9H), 1.54-1.68 (m, 2H), 1.81-1.90 (m, 2H), 2.77-2.90 (m, 2H), 3.89 (s, 3H), 3.92 (s, 6H), 3.98-4.07 (m, 1H), 4.18-4.33(m, 2H), 4.55 (s, 2H), 6.73 (d, 2H, J=8.8 Hz), 7.09 (d, 1H, J=3.7 Hz), 7.13 (s, 2H), 7.44 (d, 2H, J=8.8 Hz), 7.49 (s, 1H), 8.58 (d, 1H, J=5.1 Hz).
PREPARATION EXAMPLE 155
Synthesis of 4-[N-[4-(trifluoromethyl)phenyl]-N-[[2-(3,4,5-trimethoxyphenyl) pyridin-4-yl]methyl]amino]piperidine dihydrochloride
1-(tert-Butoxycarbonyl)-4-[N-[4-(trifluoromethyl)phenyl]-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]piperidine (412 mg) was treated in the same manner as described in Preparation Example 94 to give light yellow powder of the title compound.
Yield: 359 mg (91%).
PREPARATION EXAMPLE 156
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-[4-(trifluoromethyl)phenyl]-N-[3-(3,4,5-trimethoxyphenyl)benzyl]amino]piperidine
1-(tert-Butoxycarbonyl)-4-[[4-(trifluoromethyl)phenyl]amino]piperidine (689 mg) and 3-(3,4,5-trimethoxyphenyl)benzyl chloride (586 mg) was treated in the same manner as described in Example 9 to give light yellow amorphous of the title compound.
Yield: 522 mg (44%). 1H-NMR (400 MHz, CDCl3) δ: 1.45 (s, 9H), 1.58-1.70 (m, 2H), 1.83-1.90 (m, 2H), 2.76-2.87 (m, 2H), 3.87 (s, 6H), 3.88 (s, 3H), 3.96-4.06 (m, 1H), 4.15-4.30 (m, 2H), 4.58 (s, 2H), 6.68 (s, 2H), 6.76 (d, 2H, J=8.8 Hz), 7.19 (s, 1H, J=7.4 Hz), 7.33-7.44 (m, 5H).
PREPARATION EXAMPLE 157
Synthesis of 4-[N-[4-(trifluoromethyl)phenyl]-N-[3-(3,4,5-trimethoxyphenyl)benzyl]amino]piperidine hydrochloride
1-(tert-Butoxycarbonyl)-4-[N-[4-(trifluoromethyl)phenyl]-N-[3-(3,4,5-trimethoxyphenyl)benzyl]amino]piperidine (522 mg) was treated in the same manner as described in Preparation Example 94 to give light yellow powder of the title compound.
Yield: 460 mg (99%).
EXAMPLES 127 TO 132
These compounds were obtained by the condensation of amines obtained in Preparation Examples 155 and 157 with chloride derivatives obtained in Preparation Examples 3, 42 and 48. Free bases obtained were then converted to the corresponding hydrochlorides. Yields and NMR data of their free bases are listed below.
| |
| Ex- |
|
|
|
| am- |
|
|
NMR data (400 MHz, measured as |
| ple |
Structure |
Yield |
free bases, CDCl3) δ |
| |
| 127 |
|
72% |
1.74-1.92(m, 4H), 2.17-2.26(m, 2H), 2.96-3.04(m, 2H), 3.59(s, 2H), 3.89(s, 3H), 3.90(s, 3H), 3.91(s, 6H), 3.96(s, 6H), 4.60(s, 2H), 6.72(d, 2H, J=8.8Hz), 7.10 (d, 1H, J=4.9Hz), 7.13(s, 2H), 7.20(d, 1H, J=5.1Hz), 7.43(d, 2H, J=8.8Hz), 7.50(s, 1H), 7.59 |
| # (s, 1H), 8.56(d, 1H, J=4.9Hz), 8.58(d, 1H, J=5.1Hz). |
| 128 |
|
51% |
1.70-1.90(m, 4H), 2.14-2.28(m, 2H), 2.96-3.08(m, 2H), 3.61(s 2H), 3.87-3.96(m, 1H), 3.89(s, 3H), 3.90(s, 3H), 3.91(s, 6H), 3.93(s, 6H), 4.59(s, 2H), 6.71(d, 2H, J=8.8Hz), 6.75(s, 2H), 7.07- 7.15(m, 3H), 7.43(d, 2H, J=8.8 Hz), 7.49(s, 1H), 7.76(s, 1H), |
| # 8.51(d, 1H, J=1.8Hz), 8.57(d, 1H, J=5.1Hz), 8.70(s, 1H). |
| 129 |
|
59% |
1.72-1.88(m, 4H), 2.11-2.24(m, 2H), 2.98-3.10(m, 2H), 3.59(s, 2H), 3.87-3.95(m, 1H), 3.88(s, 3H), 3.89(s, 3H), 3.90(s, 6H), 3.92(s, 6H), 4.59(s, 2H), 6.71(d, 2H, J=9.0Hz), 6.76(s, 2H), 7.08 (d, 1H, J=5.1Hz), 7.12(s, 2H), 7.29(d, 1H, J=7.4Hz), 7.37(dd, |
| # 1H, J=7.6Hz, 7.6Hz), 7.40-7.52 (m, 5H), 8.56(d, 1H, J=5.1Hz). |
| 130 |
|
81% |
1.78-1.94(m, 4H), 2.15-2.27(m, 2H), 2.94-3.08(m, 2H), 3.58(s, 2H), 3.86(s, 6H), 3.87(s, 3H), 3.90(s, 3H), 3.96(s, 6H), 4.63(s, 2H), 6.67(s, 2H), 6.74(d, 2H, J=8.8Hz), 7.17-7.24(m, 4H), 7.34-7.45(m, 5H), 7.59(s, 1H), 8.59(d, 1H, J=5.1Hz). |
| 131 |
|
54% |
1.75-1.90(m, 4H), 2.14-2.24(m, 2H), 2.95-3.04(m, 2H), 3.60(s, 2H), 3.84-3.88(m, 1H), 3.86(m, 1H), 3.87(s, 3H), 3.90(s, 3H), 3.93(s, 6H), 4.61(s, 2H), 6.67(s, 2H), 6.72-6.77(m, 4H), 7.18(d, 1H, J=7.4Hz), 7.33-7.43(m, 5H), 7.76(s, 1H), 8.50(d, 1H, J=1.9 |
| # Hz), 8.69(d, 1H, J=1.9Hz). |
| 132 |
|
67% |
1.76-1.88(m, 4H), 2.11-2.19(m, 2H), 2.98-3.06(m, 2H), 3.59(s, 2H), 3.86(s, 6H), 3.87(s, 3H), 3.89(s, 3H), 3.92(s, 6H), 4.61(s, 2H), 6.67(s, 2H), 6.73(d, 2H, J=8.8Hz), 6.76(s, 2H), 7.18(d, 1H, J=7.3Hz), 7.29(d, 1H, J=7.6 Hz), 7.32-7.47(m, 8H). |
| |
PREPARATION EXAMPLE 158
Synthesis of 4-(4-bromophenyl)amino-1-(tert-butoxycarbonyl)piperidine
1-(tert-Butoxycarbonyl)-4-piperidone (5.00 g) and 4-bromoaniline (4.11 g) was treated in the same manner as described in Example 37 to give white crystalline powder of the title compound.
Yield: 3.09 g (36%). 1H-NMR (400 MHz, CDCl3) δ: 1.25-1.37 (m, 2H), 1.46 (s, 9H), 1.97-2.05 (m, 2H), 2.86-2.96 (m, 2H), 3.33-3.42 (m, 2H), 3.47-3.57 (m, 1H), 3.96-4.12 (m, 2H), 6.47 (d, 2H, J=8.8 Hz), 7.24 (d, 2H, J=9.0 Hz).
PREPARATION EXAMPLE 159
Synthesis of 4-[N-(4-bromophenyl)-N-[[2-(3,4,5-trimethoxyphenyl) pyridin-4-yl]methyl]amino]-1-(tert-butoxycarbonyl)piperidine
4-(4-Bromophenyl)amino-1-(tert-butoxycarbonyl)piperidine (711 mg) and 4-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (588 mg) was treated in the same manner as described in Example 9 to give light yellow amorphous of the title compound.
Yield: 607 mg (50%). 1H-NMR (400 MHz, CDCl3) δ: 1.45 (s, 9H), 1.50-1.64 (m, 2H), 1.81-1.88 (m, 2H), 2.74-2.88 (m, 2H), 3.86-3.94 (m, 1H), 3.89 (s, 3H), 3.93 (s, 6H), 4.14-4.32 (m, 2H), 4.46 (s, 2H), 6.59 (d, 2H, J=9.1 Hz), 7.10 (d, 1H, J=5.2 Hz), 7.14 (s, 2H), 7.28 (d, 2H, J=9.1 Hz), 7.50 (s, 1H), 8.57 (d, 1H, J=5.0 Hz).
PREPARATION EXAMPLE 160
Synthesis of 4-[N-(4-bromophenyl)-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]piperidine dihydrochloride
4-[N-(4-Bromophenyl)-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]-1-(tert-butoxycarbonyl)piperidine (607 mg) was treated in the same manner as described in Preparation Example 94 to give light yellow powder of the title compound.
Yield: 541 mg (93%).
PREPARATION EXAMPLE 161
Synthesis of 4-[N-(4-bromophenyl)-N-[[3-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]amino]-1-(tert-butoxycarbonyl)piperidine
4-(4-Bromophenyl)amino-1-(tert-butoxycarbonyl)piperidine (711 mg) and 5-chloromethyl-3-(3,4,5-trimethoxyphenyl)pyridine (588 mg) was treated in the same manner as described in Example 9 to give light yellow amorphous of the title compound.
Yield: 347 mg (28%). 1H-NMR (400 MHz, CDCl3) δ: 1.45 (s, 9H), 1.52-1.67 (m, 2H), 1.80-1.89 (m, 2H), 2.72-2.87 (m, 2H), 3.82-3.92 (m, 1H), 3.89 (s, 3H), 3.90 (s, 6H), 4.14-4.33 (m, 2H), 4.50 (s, 2H), 6.63 (d, 2H, J=9.2 Hz), 6.65 (s, 2H), 7.28 (d, 2H, J=9.4 Hz), 7.61 (s, 1H), 8.47 (d, 1H, J=2.0 Hz), 8.67 (d, 1H, J=2.2 Hz).
PREPARATION EXAMPLE 162
Synthesis of 4-[N-(4-bromophenyl)-N-[[3-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]amino]piperidine dihydrochloride
4-[N-(4-Bromophenyl)-N-[[3-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]amino]-1-(tert-butoxycarbonyl)piperidine (347 mg) was treated in the same manner as described in Preparation Example 94 to give light yellow powder of the title compound.
Yield: 302 mg (91%).
PREPARATION EXAMPLE 163
Synthesis of 4-[N-(4-bromophenyl)-N-[3-(3,4,5-trimethoxyphenyl)benzyl]amino]-1-(tert-butoxycarbonyl)piperidine
4-(4-Bromophenyl)amino-1-(tert-butoxycarbonyl)piperidine (711 mg) and 3-(3,4,5-trimethoxyphenyl)benzyl chloride (586 mg) was treated in the same manner as described in Example 9 to give light yellow amorphous of the title compound.
Yield: 1.14 g (93%). 1H-NMR (400 MHz, CDCl3) δ: 1.45 (s, 9H), 1.52-1.67 (m, 2H), 1.80-1.89 (m, 2H), 2.72-2.86 (m, 2H), 3.84-3.91 (m, 1H), 3.88 (s, 3H), 3.89 (s, 6H), 4.11-4.32 (m, 2H), 4.49 (s, 2H), 6.62 (d, 2H, J=9.2 Hz), 6.69 (s, 2H), 7.19 (d, 1H, J=7.6 Hz), 7.25 (d, 2H, J=5.5 Hz), 7.32-7.42 (m, 3H).
PREPARATION EXAMPLE 164
Synthesis of 4-[N-(4-bromophenyl)-N-[3-(3,4,5-trimethoxyphenyl)benzyl]amino]piperidine hydrochloride
4-[N-(4-Bromophenyl)-N-[3-(3,4,5-trimethoxyphenyl)benzyl]amino]-1-(tert-butoxycarbonyl)piperidine (1.14 g) was treated in the same manner as described in Preparation Example 94 to give light yellow powder of the title compound.
Yield: 973 mg (84%).
EXAMPLES 133 TO 140
These compounds were obtained by the condensation of amines obtained in Preparation Examples 160, 162 and 164 with chloride derivatives obtained in Preparation Examples 3, 42 and 48. Free bases obtained were then converted to the corresponding hydrochlorides. Yields and NMR data of their free bases are listed below.
| |
| Ex- |
|
|
|
| am- |
|
|
NMR data (400 MHz, measured as |
| ple |
Structure |
Yield |
free bases, CDCl3) δ |
| |
| 133 |
|
52% |
1.70-1.90(m, 4H), 2.14-2.25(m, 2H), 2.94-3.04(m, 2H), 3.58(s, 2H), 3.73-3.84(m, 1H), 3.89(s, 3H), 3.90(s, 3H), 3.92(s, 6H), 3.96(s, 6H), 4.52(s, 2H), 6.57(d, 2H, J=8.8Hz), 7.10(d, 1H, J=4.9 Hz), 7.14(s, 2H), 7.20(d, 1H, J=4.9Hz), 7.22(s, 2H), 7.26(d, |
| # 2H, J=8.5Hz), 7.51(s, 1H), 7.59 (s, 1H), 8.56(d, 1H, J=4.9Hz), 8.59(d, 1H, J=4.9Hz). |
| 134 |
|
56% |
1.68-1.88(m, 4H), 2.12-2.24(m, 2H), 2.95-3.04(m, 2H), 3.59(s, 2H), 3.72-3.84(m, 1H), 3.89(s, 3H), 3.90(s, 3H), 3.92(s, 6H), 3.93(s, 6H), 4.50(s, 2H), 6.57(d, 2H, J=9.2Hz), 6.74(s, 2H), 7.09 (d, 1H, J=3.9Hz), 7.13(s, 2H), 7.26(d, 2H, J=8.8Hz), 7.50(s, |
| # 1H), 7.75(s, 1H), 8.50(d, 1H, J=2.0Hz), 8.55(d, 1H, J=5.0Hz), 8.69(d, 1H, J=2.0Hz). |
| 135 |
|
65% |
1.70-1.86(m, 4H), 2.10-2.20(m, 2H), 2.97-3.08(m, 2H), 3.59(s, 2H), 3.72-3.82(m, 1H), 3.89(s, 6H), 3.92(s, 6H), 3.92(s, 6H), 4.50(s, 2H), 6.56(d, 2H, J=9.2 Hz), 6.76(s, 2H), 7.09(d, 1H, J=5.1Hz), 7.13(s, 2H), 7.23-7.33 (m, 3H), 7.37(dd, 1H, J=7.4Hz), |
| # 7.41-7.48(m, 2H), 7.49(s, 1H), 8.54(d, 1H, J=5.1Hz). |
| 136 |
|
49% |
1.77-1.93(m, 4H), 2.12-2.30(m, 2H), 2.94-3.10(m, 2H), 3.60(s, 2H), 3.73-3.83(m, 1H), 3.88(s, 3H), 3.89(s, 6H), 3.90(s, 3H), 3.96(s, 6H), 4.55(s, 2H), 6.61(d, 2H, J=9.2Hz), 6.65(s, 2H), 7.19- 7.29(m, 5H), 7.62(br, 2H), 8.47 (d, 1H, J=1.6Hz), 8.60(d, 1H, |
| # J=4.9Hz), 8.66(d, 1H, J=2.0Hz). |
| 137 |
|
50% |
1.70-1.92(m, 4H), 2.12-2.27(m, 2H), 2.93-3.07(m, 2H), 3.60(s, 2H), 3.67-4.08(m, 1H), 3.88(s, 3H), 3.89(s, 6H), 3.90(s, 3H), 3.93(s, 6H), 4.54(s, 2H), 6.60(d, 2H, J=9.0Hz), 6.64(s, 2H), 6.73- 6.80(m, 2H), 7.25(s, 2H), 7.61(s, 1H), 7.77(br, 1H), 8.45(d, 1H, |
| # J=1.7Hz), 8.50(d, 1H, J=1.7Hz), 8.65(d, 1H, J=2.0Hz). |
| 138 |
|
81% |
1.75-1.90(m, 4H), 2.17-2.24(m, 2H), 2.94-3.02(m, 2H), 3.57(s, 2H), 3.72-3.83(m, 1H), 3.88(s, 3H), 3.88(s, 6H), 3.90(s, 3H), 3.95(s, 6H), 4.54(s, 2H), 6.60(d, 2H, J=9.2Hz), 6.69(s, 2H), 7.18- 7.27(m, 6H), 7.32-7.42(m, 3H), 7.60(s, 1H), 8.58(d, 1H, J=4.9 |
| # Hz). |
| 139 |
|
80% |
1.72-1.90(m, 4H), 2.13-2.21(m, 2H), 2.94-3.05(m, 2H), 3.59(s, 2H), 3.72-3.82(m, 1H), 3.87(s, 3H), 3.88(s, 6H), 3.89(s, 3H), 3.93(s, 6H), 4.53(s, 2H), 6.60(d, 2H, J=9.0Hz), 6.68(s, 2H), 6.75 (s, 2H), 7.19(d, 1H, J=7.2Hz), 7.24(d, 2H, 1=9.0Hz), 7.31-7.41 |
| # (m, 3H), 7.76(s, 1H), 8.50(d, 1H, J=1.8Hz), 8.70(s, 1H). |
| 140 |
|
78% |
1.72-1.88(m, 4H), 2.08-2.18(m, 2H), 2.97-3.06(m, 2H), 3.58(s, 2H), 3.71-3.82(m, 1H), 3.87(s, 3H), 3.88(s, 6H), 3.89(s, 3H), 3.92(s, 6H), 4.53(s, 2H), 6.59(d, 2H, J=9.3Hz), 6.68(s, 2H), 6.76 (s, 2H), 7.18(d, 1H, J=7.3Hz), 7.21-7.47(m, 9H) |
| |
PREPARATION EXAMPLE 165
Synthesis of 1-(tert-butoxycarbonyl)-4-[(4-chlorophenyl)amino]piperidine
1-(tert-Butoxycarbonyl)-4-piperidone (5.00 g) and 4-chloroaniline (3.05 g) was treated in the same manner as described in Preparation Example 37 to give white powder of the title compound.
Yield: 3.80 g (49%). 1H-NMR (400 MHz, CDCl3) δ: 1.24-1.38 (m, 2H), 1.46 (s, 9H), 1.97-2.05 (m, 2H), 2.86-2.96 (m, 2H), 3.32-3.42 (m, 2H), 3.51 (br, 1H), 6.52 (d, 2H, J=9.0 Hz), 7.11 (d, 2H, J=9.0 Hz).
PREPARATION EXAMPLE 166
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-(4-chlorophenyl)-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]piperidine
1-(tert-Butoxycarbonyl)-4-[(4-chlorophenyl)amino]piperidine (621 mg) and 4-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (588 mg) was treated in the same manner as described in Example 9 to give light yellow amorphous of the title compound.
Yield: 789 mg (69%). 1H-NMR (400 MHz, CDCl3) δ: 1.45 (s, 9H), 1.51-1.68 (m, 2H), 1.80-1.89 (m, 2H), 2.72-2.86 (m, 2H), 3.87-3.90 (m, 1H), 3.89 (s, 3H), 3.93 (s, 6H), 4.64 (s, 2H), 6.64 (d, 2H, J=9.0 Hz), 7.14 (d, 1H, J=5.3 Hz), 7.15 (d, 2H, J=9.0 Hz), 7.51 (s, 2H), 8.57 (d, 2H, J=5.1 Hz).
PREPARATION EXAMPLE 167
Synthesis of 4-[N-(4-chlorophenyl)-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]piperidine dihydrochloride
1-(tert-Butoxycarbonyl)-4-[N-(4-chlorophenyl)-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]piperidine 789 mg) was treated in the same manner as described in Preparation Example 94 to give light yellow powder of the title compound.
Yield: 673 mg (90%).
PREPARATION EXAMPLE 168
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-(4-chlorophenyl)-N-[[3-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]amino]piperidine
1-(tert-Butoxycarbonyl)-4-[(4-chlorophenyl)amino]piperidine (621 mg) and 5-chloromethyl-3-(3,4,5-trimethoxyphenyl)pyridine (588 mg) was treated in the same manner as described in Example 9 to give light yellow amorphous of the title compound.
Yield: 268 mg (24%). 1H-NMR (400 MHz, CDCl3) δ: 1.45 (s, 9H), 1.56-1.76 (m, 2H), 1.80-1.90 (m, 2H), 2.76-2.83 (m, 2H), 3.86-3.90 (m, 1H), 3.89 (s, 3H), 3.90 (s, 6H), 4.15-4.30 (m, 2H), 4.50 (s, 2H), 6.66 (s, 2H), 6.68 (d, 2H, J=9.2 Hz), 7.15 (d, 2H, J=9.0 Hz), 7.63 (s, 1H), 8.47 (d, 1H, J=2.0 Hz), 8.66 (d, 1H, J=2.0 Hz).
PREPARATION EXAMPLE 169
Synthesis of 4-[N-(4-chlorophenyl)-N-[[3-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]amino]piperidine dihydrochloride
1-(tert-Butoxycarbonyl)-4-[N-(4-chlorophenyl)-N-[[3-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]amino]piperidine (268 mg) was treated in the same manner as described in Preparation Example 94 to give light yellow powder of the title compound.
Yield: 233 mg (91%).
PREPARATION EXAMPLE 170
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-(4-chlorophenyl)-N-[3-(3,4,5-trimethoxyphenyl)benzyl]amino]piperidine
1-(tert-Butoxycarbonyl)-4-[4-(chlorophenyl)amino]piperidine (622 mg) and 3-(3,4,5-trimethoxyphenyl)benzyl chloride (586 mg) was treated in the same manner as described in Example 9 to give light yellow amorphous of the title compound.
Yield: 1.04 g (92%). 1H-NMR (400 MHz, CDCl3) δ: 1.45 (s, 9H), 1.58-1.67 (m, 2H), 1.82-1.91 (m, 2H), 2.74-2.86 (m, 2H), 3.85-3.92 (m, 1H), 3.88 (s, 3H), 3.89 (s, 6H), 4.35-4.31 (m, 2H), 4.49 (s, 2H), 6.66 (d, 2H, J=9.2 Hz), 6.70 (s, 2H), 7.12 (d, 2H, J=9.0 Hz), 7.20 (d, 2H, J=7.3 Hz), 7.33-7.43 (m, 3H).
PREPARATION EXAMPLE 171
Synthesis of 4-[N-(4-chlorophenyl)-N-[3-(3,4,5-trimethoxyphenyl)benzyl]amino]piperidine hydrochloride
1-(tert-Butoxycarbonyl)-4-[N-(4-chlorophenyl)-N-[3-(3,4,5-trimethoxyphenyl)benzyl]amino]piperidine (1.04 g) was treated in the same manner as described in Preparation Example 94 to give light yellow powder of the title compound.
Yield: 899 mg (97%).
EXAMPLES 141 TO 148
These compounds were obtained by the condensation of amines obtained in Preparation Examples 167, 169 and 171 with chloride derivatives obtained in Preparation Examples 3, 42 and 48. Free bases obtained were then converted to the corresponding hydrochlorides. Yields and NMR data of their free bases are listed below.
| |
| Ex- |
|
|
|
| am- |
|
|
NMR data (400 MHz, measured as |
| ple |
Structure |
Yield |
free bases, CDCl3) δ |
| |
| 141 |
|
66% |
1.71-1.90(m, 4H), 2.15-2.24(m, 2H), 2.95-3.05(m, 2H), 3.58(s, 2H), 3.73-3.84(m, 1H), 3.89(s, 3H), 3.90(s, 3H), 3.93(s, 6H), 3.96(s, 6H), 4.52(s, 2H), 6.62(d, 2H, J=9.0Hz), 7.10-7.16(m, 5H), 7.19-7.24(m, 3H), 7.52(s, 1H), 7.59(s, 1H), 8.56(d, 1H, J=4.9 |
| # Hz), 8.59(d, 1H, J=4.9Hz). |
| 142 |
|
67% |
1.69-1.90(m, 1H), 2.12-2.25(m, 2H), 2.93-3.06(m, 2H), 3.59(s, 2H), 3.72-3.83(m, 1H), 3.89(s, 3H), 3.90(s, 3H), 3.92(s, 6H), 3.93(s, 6H), 4.50(s, 2H), 6.62(d, 2H, J=9.2Hz), 6.75(s, 2H), 7.10 (d, 1H, J=5.3Hz), 7.13(s, 2H), 7.13(d, 2H, J=9.0Hz), 7.50(s, |
| # 1H), 7.76(s, 1H), 8.50(d, 1H, J=1.8Hz), 8.55(d, 1H, J=5.1Hz), 8.70(d, 1H, J=1.8Hz). |
| 143 |
|
70% |
1.65-1.88(m, 4H), 2.08-2.20(m, 2H), 2.97-3.07(m, 2H), 3.59(s, 2H), 3.71-3.82(m, 1H), 3.88(s, 3H), 3.89(s, 3H), 3.90-3.93(m, 3H), 4.50(s, 2H), 6.61(d, 2H, J=8.2Hz), 6.76(s, 2H), 7.07-7.14 (m, 5H), 7.28(d, 1H, J=6.6Hz), 7.37(dd, 1H, J=7.4Hz), 7.40-7.47 |
| # (m, 2H), 7.50(s, 1H), 8.54(d, 1H, J=5.1Hz). |
| 144 |
|
57% |
1.56-1.93(m, 4H), 2.12-2.30(m, 2H), 2.92-3.10(m, 2H), 3.53-3.68 (m, 2H), 3.70-3.82(m, 1H), 3.88 (s, 3H), 3.89(s, 6H), 3.90(s, 3H), 3.96(s, 6H), 4.56(s, 2H), 6.64- 6.70(m, 4H), 7.13(d, 2H, J=9.0 Hz), 7.20-7.30(m, 3H), 7.63(br, 2H), 8.48(s, 1H), 8.60(d, 1H, |
| # J=5.1Hz), 8.66(d, 1H, J=2.2Hz). |
| 145 |
|
70% |
1.71-1.92(m, 4H), 2.12-2.27(m, 2H), 2.94-3.07(m, 2H), 3.59(s, 2H), 3.69-3.81(s, 1H), 3.88(s, 3H), 3.89(s, 6H), 3.90(s, 3H), 3.93(s, 6H), 4.54(s, 2H), 6.63- 6.68(m, 4H), 6.75(s, 2H), 7.13(d, 2H, J=9.0Hz), 7.62(s, 1H), 7.76 (s, 1H), 8.47(d, 1H, J=1.8Hz), |
| # 8.50(d, 1H, J=1.8Hz), 8.65(d, 1H, J=2.0Hz), 8.70(s, 1H). |
| 146 |
|
78% |
1.75-1.91(m, 4H), 2.13-2.23(m, 2H), 2.94-3.02(m, 2H), 3.57(s, 2H), 3.73-3.82(m, 1H), 3.88(s, 3H), 3.88(s, 6H), 3.90(s, 3H), 3.96(s, 6H), 4.55(s, 2H), 6.65(d, 2H, J=9.0Hz), 6.68(s, 2H), 7.11 (d, 2H, J=8.5Hz), 7.18-7.24(m, 4H), 7.32-7.42(m, 3H), 7.59(s, |
| # 1H), 8.59(d, 1H, J=4.9Hz). |
| 147 |
|
63% |
1.72-1.89(m, 4H), 2.12-2.21(m, 2H), 2.94-3.03(m, 2H), 3.59(s, 2H), 3.72-3.82(m, 1H), 3.87(s, 3H), 3.88(s, 6H), 3.90(s, 3H), 3.93(s, 6H), 4.53(s, 2H), 6.64(d, 2H, J=9.2Hz), 6.68(s, 2H), 6.75 (s, 2H), 7.11(d, 2H), 7.19(d, 1H, J=7.6Hz), 7.32-7.40(m, 3H), 7.76 |
| # (s, 1H), 8.50(d, 1H, J=1.8Hz), 8.69(d, 1H, J=2.2Hz). |
| 148 |
|
68% |
1.72-1.87(m, 4H), 2.08-2.18(m, 2H), 2.97-3.05(m, 2H), 3.58(s, 2H), 3.71-3.82(m, 1H), 3.87(s, 3H), 3.88(s, 6H), 3.89(s, 3H), 3.92(s, 6H), 4.53(s, 2H), 6.64(dt, 2H, J=9.3Hz, 2.9Hz), 6.68(s, 2H), 6.76(s, 2H), 7.10(dt, 2H, J=9.0Hz, 2.8Hz), 7.19(d, 1H, |
| # J=7.6Hz), 7.24-7.47(m, 7H). |
| |
PREPARATION EXAMPLE 172
Synthesis of 1-(tert-butoxycarbonyl)-4-[(3,4-difluorophenyl)amino]piperidine
1-(tert-Butoxycarbonyl)-4-piperidone (5.00 g) and 3,4-difluoroaniline (3.09 g) was treated in the same manner as described in Preparation Example 37 to give light brown prism crystal of the title compound.
Yield: 4.66 g (62%). 1H-NMR (400 MHz, CDCl3) δ: 1.24-1.37 (m, 2H), 1.46 (s, 9H), 1.97-2.05 (m, 2H), 2.85-2.96 (m, 2H), 3.26-3.36 (m, 1H), 3.38-3.52 (m, 1H), 3.96-4.14 (m, 2H), 6.22-6.28 (m, 1H), 6.38 (ddd, 1H, J=12.7 Hz, 6.6 Hz, 2.9 Hz), 6.94 (dd, 1H, J=19.1 Hz, 9.0 Hz).
PREPARATION EXAMPLE 173
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-(3,4-difluorophenyl)-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]piperidine
1-(tert-Butoxycarbonyl)-4-[(3,4-difluorophenyl)amino]piperidine (625 mg) and 4-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (588 mg) was treated in the same manner as described in Example 9 to give light yellow amorphous of the title compound.
Yield: 534 mg (47%). 1H-NMR (400 MHz, CDCl3) δ: 1.45 (s, 9H), 1.50-1.70 (m, 2H), 1.82-1.90 (m, 2H), 2.73-2.88 (m, 2H), 3.90 (s, 3H), 3.94 (s, 6H), 4.15-4.30 (m, 2H), 4.43 (s, 2H), 6.33-6.39 (m, 1H), 6.52 (ddd, 1H, J=13.6 Hz, 6.4 Hz, 3.1 Hz), 6.98 (dd, 1H, J=19.1 Hz, 9.2 Hz). 7.11 (dd, 1H, J=5.0 Hz, 1.3 Hz), 7.16 (s, 2H), 7.51 (s, 1H), 8.58 (d, 1H, J=5.1 Hz).
PREPARATION EXAMPLE 174
Synthesis of 4-[N-(3,4-difluorophenyl)-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]piperidine dihydrochloride
1-(tert-Butoxycarbonyl)-4-[N-(3,4-difluorophenyl)-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]piperidine (534 mg) was treated in the same manner as described in Preparation Example 94 to give light yellow powder of the title compound.
Yield: 442 mg (87%).
PREPARATION EXAMPLE 175
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-(3,4-difluorophenyl)-N-[[3-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]amino]piperidine
1-(tert-Butoxycarbonyl)-4-[(3,4-difluorophenyl)amino]piperidine (625 mg) and 5-chloromethyl-3-(3,4,5-trimethoxyphenyl)pyridine (588 mg) was treated in the same manner as described in Example 9 to give light yellow amorphous of the title compound.
Yield: 350 mg (31%).
PREPARATION EXAMPLE 176
Synthesis of 4-[N-(3,4-difluorophenyl)-N-[[3-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]amino]piperidine dihydrochloride
1-(tert-Butoxycarbonyl)-4-[N-(3,4-difluorophenyl)-N-[[3-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]amino]piperidine (350 mg) was treated in the same manner as described in Preparation Example 94 to give light yellow powder of the title compound.
Yield: 305 mg (92%).
PREPARATION EXAMPLE 177
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-(3,4-difluorophenyl)-N-[3-(3,4,5-trimethoxyphenyl)benzyl]amino]piperidine
1-(tert-Butoxycarbonyl)-4-[(3,4-difluorophenyl)amino]piperidine (625 mg) and 3-(3,4,5-trimethoxyphenyl)benzyl chloride (586 mg) was treated in the same manner as described in Example 9 to give light yellow amorphous of the title compound.
Yield: 980 mg (86%). 1H-NMR (400 MHz, CDCl3) δ: 1.45 (s, 9H), 1.52-1.66 (m, 2H), 1.81-1.89 (m, 2H), 2.72-2.85 (m, 2H), 3.78 (tt, 1H, J=11.8 Hz, 3.8 Hz), 3.88 (s, 3H), 3.90 (s, 6H), 4.12-4.30 (m, 2H), 4.45 (s, 2H), 6.36-6.42 (m, 1H), 6.54 (ddd, 1H, J=13.9 Hz, 6.8 Hz, 2.9 Hz), 6.71 (s, 2H), 6.95 (dd, 1H, J=19.2 Hz, 9.2 Hz), 7.20 (d, 1H, J=7.4 Hz), 7.36-7.43 (m, 3H).
PREPARATION EXAMPLE 178
Synthesis of 4-[N-(3,4-difluorophenyl)-N-[3-(3,4,5-trimethoxyphenyl)benzyl]amino]piperidine hydrochloride
1-(tert-Butoxycarbonyl)-4-[N-(3,4-difluorophenyl)-N-[3-(3,4,5-trimethoxyphenyl)benzyl]amino]piperidine (980 mg) was treated in the same manner as described in Preparation Example 94 to give light yellow powder of the title compound.
Yield: 819 mg (94%).
EXAMPLES 149 TO 156
These compounds were obtained by the condensation of amines obtained in Preparation Examples 174, 176 and 178 with chloride derivatives obtained in Preparation Examples 3, 42 and 48. Free bases obtained were then converted to the corresponding hydrochlorides. Yields and NMR data of their free bases are listed below.
| |
| Ex- |
|
|
|
| am- |
|
|
NMR data(400 MHz, measured as |
| ple |
Structure |
Yield |
free bases, CDCl3) δ |
| |
| 149 |
|
67% |
1.70-1.90(m, 4H), 2.16-2.23(m, 2H), 2.95-3.03(m, 2H), 3.58(s, 2H), 3.64-3.74(m, 1H), 3.89(s, 3H), 3.90(s, 3H), 3.93(s, 6H), 3.96(s, 6H), 4.49(s, 2H), 6.31- 6.37(m, 1H), 6.51(ddd, 1H, J=13.9Hz, 6.6Hz, 3.1Hz), 6.96 (dd, 1H, J=19.2Hz, 9.8Hz), 7.11 |
| # (d, 1H, J=5.1Hz), 7.15(s, 2H), 7.20(d, 1H, J=5.1Hz), 7.22(s, 2H), 7.52(s, 1H), 7.59(s, 1H), 8.57(d, 1H, J=5.1Hz), 8.59(d, 1H, J=5.1Hz). |
| 150 |
|
47% |
1.67-1.79(m, 2H), 1.81-1.89(m, 2H), 2.13-2.20(m, 2H), 2.95-3.05 (m, 2H), 3.59(s, 2H), 3.63-3.75 (m, 1H), 3.89(s, 3H), 3.90(s, 3H), 3.93(s, 12H), 4.47(s, 2H), 6.30- 6.36(m, 1H), 6.50(ddd, 1H, J=13.9Hz, 6.6Hz, 3.1Hz), 6.75(s, 2H), 6.96(d, 1H, J=19.0Hz, 9.4 |
| # Hz), 7.10(d, 1H, J=4.1Hz), 7.15 (s, 2H), 7.51(s, 1H), 7.75(s, 1H), 8.50(d, 1H, J=1.8Hz), 8.56(d, 1H, J=5.1Hz), 8.70(s, 1H). |
| 151 |
|
53% |
1.68-1.87(m, 4H), 2.09-2.18(m, 2H), 2.98-3.06(m, 2H), 3.58(s, 2H), 3.63-3.72(m, 1H), 3.89(s, 3H), 3.89(s, 3H), 3.92(s, 6H), 3.93(s, 6H), 4.47(s, 2H), 6.33- 6.35(m, 1H), 6.50(ddd, 1H, J=13.9Hz, 6.4Hz, 2.9Hz), 6.76(s, 2H), 6.95(dd, 1H, J=19.2Hz, 9.4 |
| # Hz), 7.09(d, 1H, J=5.1Hz), 7.15 (s, 2H), 7.25-7.30(m, 1H), 7.37 (dd, 1H, J=7.3Hz, 7.3Hz), 7.42- 7.46(m, 2H), 7.50(s, 1H), 8.56(d, 1H, J=5.1Hz). |
| 152 |
|
50% |
1.72-1.96(m, 4H), 2.12-2.28(m, 2H), 2.94-3.08(m, 2H), 3.59(s, 2H), 3.62-3.72(m, 1H), 3.89(s, 3H), 3.90(s, 9H), 3.96(s, 6H), 4.52(s, 2H), 6.36-6.43(m, 1H), 6.55(ddd, 1H, J=13.7Hz, 6.6Hz, 2.9Hz), 6.67(s, 2H), 6.96(dd, 1H, J=19.1Hz, 9.2Hz), 7.21(dd, 1H, |
| # J=5.1Hz, 1.2Hz), 7.24(s, 2H), 7.61(br, 1H), 7.64(s, 1H), 8.47(d, 1H, J=2.0Hz), 8.60(d, 1H, J=4.9 Hz), 8.67(d, 1H, J=2.0Hz). |
| 153 |
|
61% |
1.71-1.90(m, 4H), 2.12-2.25(m, 2H), 2.95-3.05(m, 2H), 3.57-3.75 (m, 1H), 3.59(s, 2H), 3.88(s, 3H), 3.90(s, 9H), 3.93(s, 6H), 4.50(s, 2H), 6.32-6.43(m, 1H), 6.54(ddd, 1H, J=13.6Hz, 6.4Hz, 2.7Hz), 6.67(s, 2H), 6.73-6.78(m, 3H), 6.96(dd, 1H, J=18.9Hz, 9.6Hz), 7.63(s, 1H), 7.76(s, 1H), 8.46(s, |
| # 1H), 8.50(d, 1H, J=1.6Hz), 8.66 (d, 1H, J=1.8Hz), 8.70(d, 1H, J=2.0Hz). |
| 154 |
|
82% |
1.74-1.90(m, 4H), 2.13-2.22(m, 2H), 2.95-3.01(m, 2H), 3.57(s, 2H), 3.63-3.73(m, 1H), 3.88(s, 3H), 3.89(s, 6H), 3.90(s, 3H), 3.96(s, 6H), 4.51(s, 2H), 6.34- 6.40(m, 1H), 6.52(ddd, 1H, J=14.1Hz, 6.6Hz, 3.1Hz), 6.70(s, 2H), 6.94(dd, 1H, J=19.2Hz, 9.4 |
| # Hz), 7.17-7.26(m, 4H), 7.32-7.42 (m, 3H), 7.59(s, 1H), 8.59(d, 1H, J=5.1Hz). |
| 155 |
|
75% |
1.74-1.90(m, 4H), 2.13-2.21(m, 2H), 2.95-3.04(m, 2H), 3.59(s, 2H), 3.63-3.72(m, 1H), 3.88(s, 3H), 3.89(s, 6H), 3.89(s, 3H), 3.93(s, 6H), 4.49(s, 2H), 6.33- 6.39(m, 1H), 6.52(ddd, 1H, J=14.3Hz, 3.7Hz, 2.9Hz), 6.69(s, 2H), 6.75(s, 2H), 6.94(dd, 1H, |
| # J=19.1Hz, 9.8Hz), 7.19(d, 1H, J=7.8Hz), 7.32-7.41(m, 3H), 7.76 (s, 1H), 8.50(d, 1H, J=1.5Hz), 8.69(s, 1H). |
| 156 |
|
79% |
1.72-1.88(m, 4H), 2.08-2.18(m, 2H), 2.98-3.05(m, 2H), 3.58(s, 2H), 3.62-3.72(m, 1H), 3.88(s, 3H), 3.89(s, 9H), 3.92(s, 6H), 4.45(s, 2H), 6.33-6.39(m, 1H), 6.51(ddd, 1H, J=13.9Hz, 6.6Hz, 3.0Hz), 6.69(s, 2H), 6.76(s, 2H), 6.93(dd, 1H, J=19.3Hz, 9.5Hz), |
| # 7.19(d, 1H, J=7.6Hz), 7.25-7.47 (m, 7H). |
| |
PREPARATION EXAMPLE 179
Synthesis of 1-(tert-butoxycarbonyl)-4-[(4-fluorophenyl)amino]piperidine
1-(tert-Butoxycarbonyl)-4-piperidone (5.00 g) and 4-fluoroaniline (2.66 g) was treated in the same manner as described in Preparation Example 37 to give white crystalline powder of the title compound.
Yield: 4.99 g (71%). 1H-NMR (400 MHz, CDCl3) δ: 1.23-1.36 (m, 2H), 1.46 (s, 9H), 1.97-2.05 (m, 2H), 2.84-2.96 (m, 2H), 3.30-3.39 (m, 2H), 3.96-4.14 (m, 2H), 6.51-6.57 (m, 2H), 6.84-6.91 (m, 2H).
PREPARATION EXAMPLE 180
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-(4-fluorophenyl)-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]piperidine
1-(tert-Butoxycarbonyl)-4-[(4-fluorophenyl)amino]piperidine (589 mg) and 4-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (588 mg) was treated in the same manner as described in Example 9 to give light yellow amorphous of the title compound.
Yield: 702 mg (64%). 1H-NMR (400 MHz, CDCl3) δ: 1.45 (s, 9H), 1.48-1.64 (m, 2H), 1.81-1.90 (m, 2H), 2.72-2.85 (m, 2H), 3.69-3.98 (m, 1H), 3.89 (m, 3H), 3.94 (m, 6H), 4.16-4.28 (m, 2H), 4.43 (s, 2H), 6.66-6.73 (m, 2H), 6.91 (dd, 2H, J=9.2 Hz, 9.2 Hz), 7.12-7.16 (m, 3H), 7.53 (s, 1H).
PREPARATION EXAMPLE 181
Synthesis of 4-[N-(4-fluorophenyl)-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]piperidine dihydrochloride
1-(tert-Butoxycarbonyl)-4-[N-(4-fluorophenyl)-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]piperidine (702 mg) was treated in the same manner as described in Preparation Example 94 to give light yellow powder of the title compound.
Yield: 561 mg (84%).
PREPARATION EXAMPLE 182
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-(4-fluorophenyl)-N-[[3-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]amino]piperidine
1-(tert-Butoxycarbonyl)-4-[(4-fluorophenyl)amino]piperidine (589 mg) and 5-chloromethyl-3-(3,4,5-trimethoxyphenyl)pyridine (588 mg) was treated in the same manner as described in Example 9 to give light yellow amorphous of the title compound.
Yield: 190 mg (17%). 1H-NMR (400 MHz, CDCl3) δ: 1.45 (s, 9H), 1.50-1.73 (m, 2H), 1.82-1.90 (m, 2H), 2.71-2.85 (m, 2H), 3.71 (tt, 1H, J=11.7 Hz, 3.1 Hz), 3.89 (s, 3H), 3.90 (s, 6H), 4.12-4.30 (m, 2H), 4.45 (s, 2H), 6.66 (s, 2H), 6.73-6.78 (m, 2H), 6.91 (dd, 2H, J=9.2 Hz, 8.2 Hz), 7.65 (s, 1H), 8.49 (d, 1H, J=2.0 Hz), 8.65 (d, 1H, J=2.0 Hz).
PREPARATION EXAMPLE 183
Synthesis of 4-[N-(4-fluorophenyl)-N-[[3-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]amino]piperidine dihydrochloride
1-(tert-Butoxycarbonyl)-4-[N-(4-fluorophenyl)-N-[[3-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]amino]piperidine (190 mg) was treated in the same manner as described in Preparation Example 94 to give light yellow powder of the title compound.
Yield: 165 mg (91%).
PREPARATION EXAMPLE 184
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-(4-fluorophenyl)-N-[3-(3,4,5-trimethoxyphenyl)benzyl]amino]piperidine
1-(tert-Butoxycarbonyl)-4-[(4-fluorophenyl)amino]piperidine (589 mg) and 3-(3,4,5-trimethoxyphenyl)benzyl chloride (586 mg) was treated in the same manner as described in Example 9 to give light yellow amorphous of the title compound.
Yield: 1.01 g (92%). 1H-NMR (400 MHz, CDCl3) δ: 1.44 (s, 9H), 1.51-1.65 (m, 2H), 1.82-1.90 (m, 2H), 2.82-2.84 (m, 2H), 3.78 (tt, 1H, J=11.7 Hz, 3.5 Hz), 3.88 (s, 3H), 3.90 (s, 6H), 4.10-4.30 (m, 2H), 4.45 (s, 2H), 6.68-6.73 (m, 4H), 6.89 (dd, 2H, J=9.2 Hz, 8.2 Hz), 7.21-7.25 (m, 1H, 7.32-7.41 (m, 3H).
PREPARATION EXAMPLE 185
Synthesis of 4-[N-(4-fluorophenyl)-N-[3-(3,4,5-trimethoxyphenyl)benzyl]amino]piperidine hydrochloride
1-(tert-Butoxycarbonyl)-4-[N-(4-fluorophenyl)-N-[3-(3,4,5-trimethoxyphenyl)benzyl]amino]piperidine (1.01 g) was treated in the same manner as described in Preparation Example 94 to give light yellow powder of the title compound.
Yield: 790 mg (88%).
EXAMPLES 157 TO 164
These compounds were obtained by the condensation of amines obtained in Preparation Examples 181, 183 and 185 with chloride derivatives obtained in Preparation Examples 3, 42 and 48. Free bases obtained were then converted to the corresponding hydrochlorides. Yields and NMR data of their free bases are listed below.
| |
| Ex- |
|
|
|
| am- |
|
|
NMR data (400 MHz, measured as |
| ple |
Structure |
Yield |
free bases, CDCl3) δ |
| |
| 157 |
|
62% |
1.60-1.82(m, 2H), 1.83-1.91(m, 2H), 2.13-2.23(m, 2H), 2.95-3.03 (m, 2H), 3.57(s, 2H), 3.64-3.75 (m, 1H), 3.89(s, 3H), 3.90(s, 3H), 3.93(s, 6H), 3.96(s, 6H), 4.48(s, 2H), 6.65-6.70(m, 2H), 6.90(dd, 2H, J=8.8Hz, 8.8Hz), 7.13-7.16 (m, 3H), 7.20(d, 1H, J=5.1Hz), |
| # 7.22(s, 2H), 7.54(s, 1H), 7.59(s, 1H), 8.55(d, 1H, J=5.1Hz), 8.59 (d, 1H, J=4.9Hz). |
| 158 |
|
53% |
1.66-1.95(m, 4H), 2.12-2.24(m, 2H), 2.95-3.07(m, 2H), 3.60(s, 2H), 3.64-3.76(m, 1H), 3.89(s, 3H), 3.90(s, 3H), 3.92(s, 6H), 3.93(s, 6H), 4.47(s, 2H), 6.63- 6.70(m, 1H), 6.75(s, 2H), 6.90 (dd, 1H, J=9.2Hz, 9.2Hz), 7.11- 7.16(m, 3H), 7.53(s, 1H), 7.77(s, |
| # 1H), 8.50(d, 1H, J=2.0Hz), 8.55 (d, 1H, J=4.9Hz), 8.70(d, 1H, J=5.9Hz). |
| 159 |
|
51% |
1.64-1.90(m, 4H), 2.07-2.20(m, 4H), 2.97-3.08(m, 2H), 3.59(s, 2H), 3.64-3.76(m, 1H), 3.89(s, 6H), 3.92(s, 6H), 3.93(s, 6H), 4.47(s, 2H), 6.62-6.70(m, 2H), 6.77(s, 2H), 6.86-6.93(m, 2H), 7.11-7.16(m, 3H), 7.25-7.31(m, 3H), 7.37(dd, 1H, J=7.4Hz, 7.4 Hz), 7.42-7.49(m, 2H), 7.53(s, |
| # 1H), 8.54(d, 1H, J=5.1Hz). |
| 160 |
|
49% |
1.74-1.98(m, 4H), 2.10-2.30(m, 2H), 2.90-3.12(m, 2H), 3.53-3.73 (m, 3H), 3.88(s, 3H), 3.89(s, 6H), 3.90(s, 3H), 3.96(s, 6H), 4.50(s, 2H), 6.66(s, 2H), 6.70-6.76(m, 2H), 6.90(dd, 2H, J=8.8Hz, 8.8 Hz), 7.19-7.28(m, 3H), 7.65(br, 2H), 8.49(d, 1H, J=1.8Hz), 8.60 |
| # (d, 1H, J=4.9Hz), 8.64(d, 1H, J=2.2Hz). |
| 161 |
|
26% |
1.67-1.97(m, 4H), 2.10-2.27(m, 2H), 2.94-3.06(m, 2H), 3.56-3.68 (m, 3H), 3.88(s, 3H), 3.89(s, 6H), 3.90(s, 3H), 3.93(s, 6H), 4.49(s, 2H), 6.65(s, 2H), 6.69-6.80(m, 4H), 6.84-6.93(m, 2H), 7.64(s, 1H), 7.77(br, 1H), 8.48(d, 1H, J=1.7Hz), 8.50(d, 1H, J=1.7Hz), |
| # 8.64(d, 1H, J=1.9Hz), 8.70(s, 1H). |
| 162 |
|
83% |
1.72-1.92(m, 4H), 2.12-2.21(m, 2H), 2.94-3.02(m, 2H), 3.57(s, 2H), 3.64-3.74(m, 1H), 3.88(s, 3H), 3.89(s, 6H), 3.90(s, 3H), 3.96(s, 6H), 4.51(s, 1H), 6.66- 6.71(m, 4H), 6.88(dd, 2H, J=8.6 Hz, 8.6Hz), 7.18-7.27(m, 4H), 7.34(dd, 1H, J=7.4Hz, 7.4Hz), |
| # 7.39(d, 2H, J=5.4Hz), 7.59(s, 1H), 8.59(d, 1H, J=5.1Hz). |
| 163 |
|
68% |
1.68-1.87(m, 4H), 2.10-2.22(m, 2H), 2.94-3.04(m, 2H), 3.59(s, 2H), 3.65-3.74(m, 1H), 3.87(s, 3H), 3.88(s, 6H), 3.90(s, 3H), 3.93(s, 6H), 4.49(s, 2H), 6.66- 6.70(m, 6H), 6.88(dd, 2H, J=8.8 Hz, 8.8Hz), 7.19-7.40(m, 4H), 7.77(s, 1H), 8.49(d, 1H, J=1.8 |
| # Hz), 8.70(s, 1H). |
| 164 |
|
74% |
1.70-1.90(m, 4H), 2.08-2.18(m, 2H), 2.95-3.05(m, 2H), 3.58(s, 2H), 3.63-3.73(m, 1H), 3.87(s, 3H), 3.88(s, 6H), 3.89(s, 3H), 3.92(s, 6H), 4.50(s, 2H), 6.65- 6.72(m, 2H), 6.69(s, 2H), 6.76(s, 2H), 6.87(dd, 2H, J=9.0Hz, 9.0 Hz), 7.22(d, 1H, J=7.6Hz), 7.25- |
| # 7.48(m, 9H). |
| |
PREPARATION EXAMPLE 186
Synthesis of 1-(tert-butoxycarbonyl)-4-phenylaminopiperidine
1-(tert-Butoxycarbonyl)-4-piperidone (5.00 g) and aniline (2.23 g) was treated in the same manner as described in Preparation Example 37 to give white needles of the title compound.
Yield: 3.77 g (57%). 1H-NMR (400 MHz, CDCl3) δ: 1.25-1.38 (m, 2H), 1.47 (s, 9H), 2.00-2.07 (m, 2H), 2.87-2.97 (m, 2H), 3.38-3.53 (m, 2H), 3.96-4.14 (m, 2H), 6.57-6.52 (m, 2H), 6.70 (tt, 1H, J=6.2 Hz, 1.0 Hz), 7.17 (dd, 2H, J=8.6 Hz, 7.2 Hz).
PREPARATION EXAMPLE 187
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-phenyl-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]piperidine
1-(tert-Butoxycarbonyl)-4-phenylaminopiperidine (553 mg) and 4-chloromethyl-2-(3,4,5-trimethoxyphenyl)pyridine (588 mg) was treated in the same manner as described in Example 9 to give light yellow amorphous of the title compound.
Yield: 760 mg (71%). 1H-NMR (400 MHz, CDCl3) δ: 1.45 (s, 9H), 1.53-1.63 (m, 2H), 1.83-1.91 (m, 2H), 2.76-2.90 (m, 2H), 3.86-3.97 (m, 1H), 3.89 (s, 3H), 3.93 (s, 6H), 4.14-4.32 (m, 2H), 4.49 (s, 2H), 6.71-6.78 (m, 3H), 7.14 (s, 1H), 7.15 (s, 2H), 7.21 (dd, 2H, J=8.8 Hz, 7.4 Hz), 7.55 (s, 1H), 8.56 (d, 1H, J=5.1 Hz).
PREPARATION EXAMPLE 188
Synthesis of 4-[N-phenyl-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]piperidine dihydrochloride
1-(tert-Butoxycarbonyl)-4-[N-phenyl-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]piperidine (760 mg) was treated in the same manner as described in Preparation Example 94 to give light yellow powder of the title compound.
Yield: 652 mg (90%).
PREPARATION EXAMPLE 189
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-phenyl-N-[[3-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]amino]piperidine
1-(tert-Butoxycarbonyl)-4-phenylaminopiperidine (553 mg) and 5-chloromethyl-3-(3,4,5-trimethoxyphenyl)pyridine (588 mg) was treated in the same manner as described in Example 9 to give light yellow amorphous of the title compound.
Yield: 222 mg (21%). 1H-NMR (400 MHz, CDCl3) δ: 1.45 (s, 9H), 1.52-1.67 (m, 2H), 1.82-1.91 (m, 2H), 2.74-2.87 (m, 2H), 3.88-3.90 (m, 1H), 3.88 (s, 3H), 3.89 (s, 6H), 4.14-4.31 (m, 2H), 4.53 (s, 2H), 6.67 (s, 2H), 6.74-6.80 (m, 3H), 7.21 (dd, 2H, J=8.8 Hz, 7.2 Hz), 7.67 (s, 1H), 8.50 (d, 1H, J=5.3 Hz, 2.2 Hz), 8.66 (d, 1H, J=2.1 Hz).
PREPARATION EXAMPLE 190
Synthesis of 4-[N-phenyl-N-[[3-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]amino]piperidine dihydrochloride
1-(tert-Butoxycarbonyl)-4-[N-phenyl-N-[[3-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]amino]piperidine (222 mg) was treated in the same manner as described in Preparation Example 94 to give light yellow powder of the title compound.
Yield: 197 mg (94%).
PREPARATION EXAMPLE 191
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-phenyl-N-[3-(3,4,5-trimethoxyphenyl)benzyl]amino]piperidine
1-(tert-Butoxycarbonyl)-4-phenylaminopiperidine (553 mg) and 3-(3,4,5-trimethoxyphenyl)benzyl chloride (586 mg) was treated in the same manner as described in Example 9 to give light yellow amorphous of the title compound.
Yield: 1.06 g (100%). 1H-NMR (400 MHz, CDCl3) δ: 1.45 (s, 9H), 1.52-1.68 (m, 2H), 1.83-1.92 (m, 2H), 2.73-2.86 (m, 2H), 3.88 (s, 3H), 3.89 (s, 6H), 3.94 (tt, 1H, J=11.7 Hz, 3.3 Hz), 4.14-4.30 (m, 2H), 4.52 (s, 2H), 6.69-6.78 (m, 6H), 7.17-7.27 (m, 2H), 7.32-7.42 (m, 3H).
PREPARATION EXAMPLE 192
Synthesis of 4-[N-phenyl-N-[3-(3,4,5-trimethoxyphenyl)benzyl]amino]piperidine hydrochloride
1-(tert-Butoxycarbonyl)-4-[N-phenyl-N-[3-(3,4,5-trimethoxyphenyl)benzyl]amino]piperidine (1.06 g) was treated in the same manner as described in Preparation Example 94 to give light yellow powder of the title compound.
Yield: 909 mg (97%).
EXAMPLES 165 TO 169
These compounds were obtained by the condensation of amines obtained in Preparation Examples 188, 190 and 192 with chloride derivatives obtained in Preparation Examples 3 and 48. Free bases obtained were then converted to the corresponding hydrochlorides. Yields and NMR data of their free bases are listed below.
| |
| Ex- |
|
|
|
| am- |
|
|
NMR data (400 MHz, measured as |
| ple |
Structure |
Yield |
free bases, CDCl3) δ |
| |
| 165 |
|
53% |
1.63-1.81(m, 4H), 1.82-1.92(m, 2H), 2.14-2.24(m, 2H), 2.95-3.05 (m, 2H), 3.59(s, 2H), 3.80-4.02 (m, 1H), 3.89(s, 3H), 3.90(s, 3H), 3.92(s, 6H), 3.93(s, 6H), 4.53(s, 2H), 6.69-6.77(m, 5H), 7.13-7.17 (m, 3H), 7.20(dd, 2H, J=7.6Hz, 7.6Hz), 7.55(s, 1H), 7.76(s, 1H), |
| # 8.51(d, 1H, J=1.8Hz), 8.55(d, 1H, J=5.1Hz), 8.70(s, 1H). |
| 166 |
|
50% |
1.85-2.04(m, 4H), 2.20-2.40(m, 2H), 2.92-3.25(m, 2H), 3.60-3.77 (m, 3H), 3.88(s, 3H), 3.89(s, 6H), 3.90(s, 3H), 3.97(s, 6H), 4.59(s, 2H), 6.67(s, 2H), 6.72-6.81(m, 4H), 7.17-7.30(m, 4H), 7.68(s, 1H), 8.50(s, 1H), 8.62(d, 1H, J=4.9Hz , 8.65(d, 1H, J=2.0Hz). |
| 167 |
|
43% |
1.72-1.92(m, 4H), 2.13-2.26(m, 2H), 2.95-3.04(m, 2H), 3.59(s, 2H), 3.78-4.01(m, 1H), 3.88(s, 9H), 3.90(s, 3H), 3.93(s, 6H), 4.56(s, 2H), 6.66(s, 2H), 6.70- 6.78(m, 5H), 7.19(dd, 2H, J=8.2 Hz, 8.2Hz), 7.66(s, 1H), 7.77(s, 1H), 8.50(d, 1H, J=2.3Hz), 8.51 |
| # (d, 1H, J=2.2Hz), 8.65(d, 1H, J=1.9Hz), 8.70(d, 1H, J=2.2Hz). |
| 168 |
|
82% |
1.75-1.92(m, 4H), 2.14-2.23(m, 2H), 2.94-3.01(m, 2H), 3.57(s, 2H), 3.80-3.94(m, 1H), 3.87(s, 3H), 3.88(s, 6H), 3.90(s, 3H), 3.96(s, 6H), 4.57(s, 2H), 6.67- 6.77(m, 5H), 7.15-7.27(m, 5H), 7.34(dd, 1H, J=7.4Hz, 7.4Hz), 7.39(d, 1H, 7.6Hz), 7.42(s, 1H), |
| # 7.59(s, 1H), 8.59(d, 1H, J=5.1 Hz). |
| 169 |
|
65% |
1.72-1.91(m, 4H), 2.13-2.22(m, 2H), 2.95-3.03(m, 2H), 3.59(s, 2H), 3.79-4.00(m, 1H), 3.87(s, 3H), 3.87(s, 6H), 3.90(s, 3H), 3.93(s, 6H), 4.56(s, 2H), 6.66- 6.77(m, 7H), 7.18(dd, 2H, J=7.4 Hz, 7.4Hz), 7.24(d, 1H, J=7.4 Hz), 7.33(dd, 1H, J=7.4Hz, 7.4 |
| # Hz), 7.38(d, 1H, J=7.6Hz), 7.41 (s, 1H), 7.76(s, 1H), 8.50(d, 1H, J=1.6Hz), 8.69(d, 1H, J=2.2Hz). |
| |
PREPARATION EXAMPLES 193 TO 203
These compounds were prepared by the same procedure as described in Preparation Example from 1 to 3. Structures and NMR data are listed below.
| |
| Preparation |
|
|
| Example |
Structure |
NMR data (400 MHz, CDCl3) δ |
| |
| 193 |
|
4.61(s, 2H), 7.25(d, 1H, J=1.2Hz), 7.41-7.52 (m, 3H), 7.75(d, 1H, J=0.8Hz), 7.98-8.02(m, 2H), 8.69(d, 1H, J=4.9Hz). |
| 194 |
|
3.87(s, 3H), 4.60(s, 2H), 7.01(d, 1H, J=8.4Hz), 7.08(t, 1H, J=7.4Hz), 7.24(dd, 1H, J=5.1Hz, 1.4Hz), 7.38(dt, 1H, J=7.4Hz, 1.8Hz), 7.77 (dd, 1H, J=7.6Hz, 1.8Hz), 7.84(s, 1H), 8.69(d, 1H, J=5.1Hz) |
| 195 |
|
3.90(s, 3H), 4.60(s, 2H), 6.87-7.03(1H, m), 7.39(t, 1H, 7.8Hz), 7.50-7.66(m, 2H), 7.73(s, 1H), 8.68(d, 1H, J=5.1Hz) |
| 196 |
|
1.45(t, 3H, J=7.0Hz), 4.12(q, 2H, J=7.0Hz), 4.59(s, 2H), 6.99(d, 2H, J=8.8Hz), 7.18(d, 1H, J=5.1Hz), 7.20-7.29(m, 1H), 7.68(s, 1H), 7.95 (d, 2H, J=8.8Hz), 8.63(d, 1H, J=5.1Hz) |
| 197 |
|
3.95(s, 3H), 4.00(s, 3H), 4.60(s, 2H), 6.96(d, 1H, J=8.4Hz), 7.21(d, 1H, J=4.1Hz), 7.53(dd, 1H, J=8.4Hz, 2.0Hz), 7.67(d, 1H, J=2.0Hz), 7.70(s, 1H), 8.65(d, 1H, J=5.1Hz) |
| 198 |
|
4.61(s, 2H), 7.14-7.21(m, 1H), 7.21-7.23(m, 2H), 7.35-7.42(m, 1H), 7.80(s, 1H), 7.98(1H, dt, J=8.0Hz, 2.0Hz), 8.73(d, 1H, J=5.1Hz) |
| 199 |
|
4.61(s, 2H), 7.13(1H, dt, J=8.4Hz, 2.8Hz), 7.28 (1H, d, J=5.0Hz), 7.40-7.79(m, 1H), 7.70-7.79 (m, 3H), 8.69(d, 1H, J=5.0Hz) |
| 200 |
|
4.60(s, 2H), 7.13-7.20(m, 2H), 7.25(1H, d, J=5.1Hz), 7.70(s, 1H), 7.95-8.03(m, 2H), 8.66 (d, 1H, J=5.1Hz) |
| 201 |
|
4.61(s, 2H), 7.21-7.30(m, 2H), 7.69(s, 1H), 7.73-7.76(m, 1H), 7.85-7.92(m, 1H), 8.76(d, 1H, J=4.9Hz) |
| 202 |
|
4.61(s, 2H), 6.86-6.91(m, 1H), 7.31(1H, d, J=5.1Hz), 7.51-7.59(m, 2H), 7.71(s, 1H), 8.69 (d, 1H, J=5.1Hz) |
| 203 |
|
4.61(s, 2H), 7.26(d, 1H, J=4.9Hz), 7.45(d, 2H, J=8.4Hz), 7.72(s, 1H), 7.95(d, 2H, J=8.4Hz), 8.68(s, 1H, J=4.9Hz) |
| |
PREPARATION EXAMPLE 204
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-(4-methoxyphenyl)-N-[(2-phenylpyridin-4-yl)methyl]amino]piperidine
4-p-Anisidino)-1-(tert-butoxycarbonyl)piperidine (612 mg) and 4-chloromethyl -2-phenylpyridine (204 mg) were condensed in the same manner as described in Example 9 to give the title compound.
Yield: 407 mg (43%).
PREPARATION EXAMPLE 205
Synthesis of 4-[N-(4-methoxyphenyl)-N-[(2-phenylpyridin-4-yl)methyl]amino]piperidine dihydrochloride
1-(tert-Butoxycarbonyl)-4-[N-(4-methoxyphenyl)-N-[(2-phenylpyridin-4-yl)methyl]amino]piperidine (407 mg) was treated in the same manner as described in Preparation Example 94 to give the title compound.
Yield: 365 mg (95%).
PREPARATION EXAMPLE 206
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-(4-methoxyphenyl)-N-[[2-(2-methoxyphenyl)pyridin-4-yl]methyl]amino]piperidine
4-p-Anisidino)-1-(tert-butoxycarbonyl)piperidine (306 mg) and 4-chloromethyl -2-(2-methoxyphenyl)pyridine (234 mg) were condensed in the same manner as described in Example 9 to give the title compound.
Yield: 237 mg (72%).
PREPARATION EXAMPLE 207
Synthesis of 4-[N-(4-methoxyphenyl)-N-[[2-(2-methoxyphenyl)pyridin-4-yl]methyl]amino]piperidine dihydrochloride
1-(tert-Butoxycarbonyt)-4-[N-(4-methoxyphenyl)-N-[[2-(2-methoxyphenyl)pyridin-4-yl]methyl]amino]piperidine (360 mg) was treated in the same manner as described in Preparation Example 94 to give the title compound.
Yield: 365 mg (65%).
PREPARATION EXAMPLE 208
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-(4-methoxyphenyl)-N-[[2-(3-methoxyphenyl)pyridin-4-yl]methyl]amino]piperidine
4-p-Anisidino)-1-(tert-butoxycarbonyl)piperidine (306 mg) and 4-chloromethyl-2-(3-methoxyphenyl)pyridine (234 mg) were condensed in the same manner as described in Example 9 to give the title compound.
Yield: 550 mg (theoretical yield).
PREPARATION EXAMPLE 209
Synthesis of 4-[N-(4-methoxyphenyl)-N-[[2-(3-methoxyphenyl)pyridin-4-yl]methyl]amino]piperidine dihydrochloride
1-(tert-Butoxycarbonyl)-4-[N-(4-methoxyphenyl)-N-[[2-(3-methoxyphenyl)pyridin-4-yl]methyl]amino]piperidine (550 mg) was treated in the same manner as described in Preparation Example 94 to give the title compound.
Yield: 436 g (85%).
PREPARATION EXAMPLE 210
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-[2-(4-ethoxyphenyl)pyridin-4-yl]methyl-N-(4-methoxyphenyl)amino]piperidine
4-(p-Anisidino)-1-(tert-butoxycarbonyl)piperidine (306 mg) and 4-chloromethyl-2-(4-ethoxyphenyl)pyridine (248 mg) were condensed in the same manner as described in Example 9 to give the title compound.
Yield: 515 mg (99%).
PREPARATION EXAMPLE 211
Synthesis of 4-[N-[2-(4-ethoxyphenyl)pyridin-4-yl]methyl-N-(4-methoxyphenyl)amino]piperidine dihydrochloride
1-(tert-Butoxycarbonyl)-4-[N-[2-(4-ethoxyphenyl)pyridin-4-yl]methyl-N-(4-methoxyphenyl)amino]piperidine (515 mg) was treated in the same manner as described in Preparation Example 94 to give the title compound.
Yield: 418 mg (80%).
PREPARATION EXAMPLE 212
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-(4-methoxyphenyl)-N-[[2-(3,4-dimethoxyphenyl)pyridin-4-yl]methyl]amino]piperidine
4-(p-Anisidino)-1-(tert-butoxycarbonyl)piperidine (306 mg) and 4-chloromethyl-2-(3,4-dimethoxyphenyl)pyridine (264 mg) were condensed in the same manner as described in Example 9 to give the title compound.
Yield: 600 mg (theoretical yield).
PREPARATION EXAMPLE 213
Synthesis of 4-[N-[2-(3,4-dimethoxyphenyl)pyridin-4-yl]methyl-N-(4-methoxyphenyl)amino]piperidine dihydrochloride
1-(tert-Butoxycarbonyl)-4-[N-[2-(3,4-dimethoxyphenyl)pyridin-4-yl]methyl-N-(4-methoxyphenyl)amino]piperidine (600 mg) was treated in the same manner as described in Preparation Example 94 to give the title compound.
Yield: 416 mg (77%).
PREPARATION EXAMPLE 214
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-[2-(2-fluorophenyl)pyridin-4-yl]methyl-N-(4-methoxyphenyl)amino]piperidine
4-(p-Anisidino)-1-(tert-butoxycarbonyl)piperidine (306 mg) and 4-chloromethyl-2-(2-fluorophenyl)pyridine (222 mg) were condensed in the same manner as described in Example 9 to give the title compound.
Yield: 530 mg (theoretical yield).
PREPARATION EXAMPLE 215
Synthesis of 4-[N-[2-(2-fluorophenyl)pyridin-4-yl]methyl-N-(4-methoxyphenyl)amino]piperidine dihydrochloride
1-(tert-Butoxycarbonyl)-4-[N-[2-(2-fluorophenyl)pyridin-4-yl]methyl-N-(4-methoxyphenyl)amino]piperidine (530 mg) was treated in the same manner as described in Preparation Example 94 to give the title compound.
Yield: 423 mg (85%).
PREPARATION EXAMPLE 216
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-[2-(3-fluorophenyl)pyridin-4-yl]methyl-N-(4-methoxyphenyl)amino]piperidine
4-p-Anisidino)-1-(tert-butoxycarbonyl)piperidine (153 mg) and 4-chloromethyl-2-(3-fluorophenyl)pyridine (111 mg) were condensed in the same manner as described in Example 9 to give the title compound.
Yield: 270 mg (theoretical yield).
PREPARATION EXAMPLE 217
Synthesis of 4-[N-[2-(3-fluorophenyl)pyridin-4-yl]methyl-N-(4-methoxyphenyl)amino]piperidine dihydrochloride
1-(tert-Butoxycarbonyl)-4-[N-[2-(3-fluorophenyl)pyridin-4-yl]methyl-N-(4-methoxyphenyl)amino]piperidine (270 mg) was treated in the same manner as described in Preparation Example 94 to give the title compound.
Yield: 193 mg (70%).
PREPARATION EXAMPLE 218
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-[2-(4-fluorophenyl)pyridin-4-yl]methyl-N-(4-methoxyphenyl)amino]piperidine
4-(p-Anisidino)-1-(tert-butoxycarbonyl)piperidine (306 mg) and 4-chloromethyl-2-(4-fluorophenyl)pyridine (222 mg) were condensed in the same manner as described in Example 9 to give the title compound.
Yield: 550 mg (theoretical yield).
PREPARATION EXAMPLE 219
Synthesis of 4-[N-[2-(4-fluorophenyl)pyridin-4-yl]methyl-N-(4-methoxyphenyl)amino]piperidine dihydrochloride
1-(tert-Butoxycarbonyl)-4-[N-[2-(4-fluorophenyl)pyridin-4-yl]methyl-N-(4-m ethoxyphenyl)amino]piperidine (550 mg) was treated in the same manner as described in Preparation Example 94 to give the title compound.
Yield: 439 mg (88%).
PREPARATION EXAMPLE 220
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-[2-(3,4-difluorophenyl)pyridin-4-yl]methyl-N-(4-methoxyphenyl)amino]piperidine
4-(p-Anisidino)-1-(tert-butoxycarbonyl)piperidine (306 mg) and 4-chloromethyl-2-(3,4-difluorophenyl)pyridine (240 mg) were condensed in the same manner as described in Example 9 to give the title compound.
Yield: 590 mg (theoretical yield).
PREPARATION EXAMPLE 221
Synthesis of 4-[N-[2-(3,4-difluorophenyl)pyridin-4-yl]methyl-N-(4-methoxyphenyl)amino]piperidine dihydrochloride
1-(tert-Butoxycarbonyl)-4-[-N-[2-(3,4-difluorophenyl)pyridin-4-yl]methyl-N-(4-methoxyphenyl)amino]piperidine (590 mg) was treated in the same manner as described in Preparation Example 94 to give the title compound.
Yield: 483 mg (93%).
PREPARATION EXAMPLE 222
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-[2-(3,5-difluorophenyl)pyridin-4-yl]methyl-N-(4-methoxyphenyl)amino]piperidine
4-p-Anisidino)-1-(tert-butoxycarbonyl)piperidine (306 mg) and 4-chloromethyl-2-(3,5-difluorophenyl)pyridine (240 mg) were condensed in the same manner as described in Example 9 to give the title compound.
Yield: 530 mg (theoretical yield).
PREPARATION EXAMPLE 223
Synthesis of 4-[N-[2-(3,5-difluorophenyl)pyridin-4-yl]methyl-N-(4-methoxyphenyl)amino]piperidine dihydrochloride
1-(tert-Butoxycarbonyl)-4-[N-[2-(3,5-difluorophenyl)pyridin-4-yl]methyl-N-(4-methoxyphenyl)amino]piperidine: (530 mg) was treated in the same manner as described in Preparation Example 94 to give the title compound.
Yield: 418 mg (81%).
PREPARATION EXAMPLE 224
Synthesis of 1-(tert-butoxycarbonyl)-4-[N-[2-(4-chlorophenyl)pyridin-4-yl]methyl-N-(4-methoxyphenyl)amino]piperidine
4-(p-Anisidino)-1-(tert-butoxycarbonyl)piperidine (306 mg) and 4-chloromethyl-2-(4-chlorophenyl)pyridine (238 mg) were condensed in the same manner as described in Example 9 to give the title compound.
Yield: 600 mg (theoretical yield).
PREPARATION EXAMPLE 225
Synthesis of 4-[N-[2-(4-chlorophenyl)pyridin-4-yl]methyl-N-(4-methoxyphenyl)amino]piperidine dihydrochloride
1-(tert-Butoxycarbonyl)-4-[N-[2-(4-chlorophenyl)pyridin-4-yl]methyl-N-(4-methoxyphenyl)amino]piperidine: (600 mg) was treated in the same manner as described in Preparation Example 94 to give the title compound.
Yield: 447 mg (86%).
EXAMPLES 170 TO 202
These compounds were obtained by the condensation of amines obtained in Preparation Examples 96, 205, 207, 209, 211, 213, 215, 217, 219, 221, 223 and 225 with chloride derivatives obtained in Preparation Examples 3, 93, 94, 95, 96, 97, 98, 99, 100, 101, 102 and 103. Free bases obtained were then converted to the corresponding hydrochlorides. Yields and NMR data of their free bases are listed below.
| |
| Ex- |
|
|
|
| am- |
|
|
NMR data (400 MHz, measured as |
| ple |
Structure |
Yield |
free bases, CDCl3) δ |
| |
| 170 |
|
47% |
1.67-1.80(m, 2H), 1.83-1.91(m, 2H), 2.10-2.19(m, 2H), 2.93-3.00 (m, 2H), 3.54-3.65(m, 1H), 3.56 (s, 2H), 3.73(s, 3H), 3.89(s, 3H), 3.93(s, 6H), 4.45(s, 3H), 6.73(d, 2H, J=9.4Hz), 6.78(d, 2H, J=9.4 Hz), 7.14-7.21(m, 2H), 7.15(s, 2H), 7.38-7.49(m, 3H), 7.57(s, 1H), 7.68(s, 1H), 7.97(d, 1H, |
| # J=1.0Hz), 7.99(d, 1H, J=1.6Hz), 8.54(d, 1H, J=5.1Hz), 8.61(d, 1H, J=5.1Hz). |
| 171 |
|
55% |
1.62-1.80(m, 2H), 1.84-1.93(m, 2H), 2.10-2.20(m, 2H), 2.93-3.02 (m, 2H), 3.53-3.66(m, 1H), 3.56 (s, 2H), 3.73(s, 3H), 3.90(s, 3H), 3.96(s, 6H), 4.44(s, 2H), 6.65- 6.83(m, 4H), 7.14-7.30(m, 4H), 7.36-7.50(m, 3H), 7.59(s, 1H), 7.67(s, 1H), 7.93(d, 2H, J=7.0 Hz), 8.54-8.61(m, 2H). |
| 172 |
|
54% |
1.67-1.92(m, 4H), 2.08-2.20(m, 2H), 2.92-3.01(m, 2H), 3.52-3.65 (m, 1H), 3.55(s, 2H), 3.72(s, 3H), 4.38(s, 2H), 6.72(d, 2H, J=9.2 Hz), 6.78(d, 2H, J=9.0Hz), 7.18 (dd, 2H, J=4.9Hz, 4.9Hz), 7.36- 7.50(m, 6H), 7.67(s, 1H), 7.68(s, 1H), 7.93(dd, 2H, J=8.4Hz, 1.2 Hz), 7.98(dd, 2H, J=8.6Hz, 1.4 |
| # Hz), 8.57(d, 1H, J=5.1Hz), 8.60 (d, 1H, J=s,1Hz). |
| 173 |
|
100% |
1.66-1.79(m, 2H), 1.82-1.91(m, 2H), 2.09-2.20(m, 2H), 2.93-3.03 (m, 2H), 3.56(s, 2H), 3.56-3.59 (m, 1H), 3.73(s, 3H), 3.80(s, 3H), 3.89(s, 3H), 3.93(s, 6H), 4.45(s, 2H), 6.73(d, 2H, J=9.3Hz), 6.78 (d, 2H, J=9.3Hz), 6.98(d, 1H, J=8.5Hz), 7.07(t, 1H, J=7.6Hz), 7.15(s, 2H), 7.15-7.19(m, 2H), |
| # 7.33-7.38(m, 1H), 7.57(s, 1H), 7.66-7.74(m, 2H), 8.53(d, 1H, J=5.1Hz), 8.61(d, 1H, J=4.9Hz). |
| 174 |
|
94% |
1.70-1.80(m, 2H), 1.83-1.91(m, 2H), 2.11-2.18(m, 2H), 2.92-3.01 (m, 2H), 3.56(s, 2H), 3.57-3.65 (m, 1H), 3.73(s, 3H), 3.74(s, 3H), 3.90(s, 3H), 3.96(s, 6H), 4.44(s, 2H), 6.71(d, 2H, J=9.0Hz), 6.78 (d, 2H, J=9.0Hz), 6.96(d, 1H, J=8.3Hz), 7.05(dt, 1H, J=7.3Hz, 1.0Hz), 7.14(d, 1H, J=5.2Hz), |
| # 7.20(d, 1H, J=5.2Hz), 7.22(2H, s), 7.32-7.37(m, 1H), 7.59(s, 1H), 7.71-7.75(m, 2H), 8.56-8.60(m, 2H). |
| 175 |
|
98% |
1.67-1.80(m, 2H), 1.83-1.90(m, 2H), 2.10-2.19(m, 2H), 2.94-3.03 (m, 2H), 3.50-3.67(m, 1H), 3.56 (s, 2H), 3.73(s, 3H), 3.74(s, 3H), 3.79(s, 3H), 4.44(s, 2H), 6.70(d, 2H, J=9.3Hz), 6.78(d, 2H, J=9.3 Hz), 6.96(d, 1H, J=8.3Hz), 6.98 (d, 1H, J=8.8Hz), 7.04(dd, 1H), J=7.6Hz, 1.0Hz), 7.07 (dd, 1H, 7.6, J=1.0Hz), 7.12-7.19 |
| # (m, 2H), 7.32-7.39(m, 2H), 7.70- 7.75(m, 4H), 8.58(d, 1H, J= 5.1Hz), 8.61 (d, 1H, J=4.9Hz). |
| 176 |
|
100% |
1.68-1.79(m, 2H), 1.82-1.90(m, 2H), 2.10-2.19(m, 2H), 2.90-3.01 (m, 2H), 3.56(s, 2H), 3.56-3.58 (m, 1H), 3.73(s, 3H), 3.89(s, 3H), 3.91(s, 3H), 3.93(s, 6H), 4.45(s, 2H), 6.73(d, 2H, J=9.3Hz), 6.78 (d, 2H, J=9.3Hz), 6.93-6.99(m, 1H), 7.15(s, 2H), 7.16-7.20(m, 2H), 7.37(t, 1H, J=7.8Hz), 7.52- |
| # 7.59(m, 3H), 7.67(s, 1H), 8.54 (d, 1H, J=5.1Hz), 8.60(d, 1H, J=5.1Hz). |
| 177 |
|
100% |
1.68-1.79(m, 2H), 1.83-1.92(m, 2H), 2.11-2.16(m, 2H), 2.91-3.02 (m, 2H), 3.56(s, 2H), 3.55-3.65 (m, 1H), 3.73(s, 3H), 3.88(s, 3H), 3.90(s, 3H), 3.96(s, 6H), 4.43(s, 2H), 6.72(d, 2H, J=9.3Hz), 6.78 (d, 2H, J=9.3Hz), 6.95(dd, 1H. J=8.3Hz, 2.7Hz), 7.16-7.21(m, 2H), 7.22(s, 2H), 7.35(t, 1H, |
| # J=7.8Hz), 7.48(d, 1H, J=7.8Hz), 7.53(t, 1H, J=2.7Hz), 7.59(s, 1H), 7.65(s, 1H), 8.55-8.60(m, 2H). |
| 178 |
|
100% |
1.65-1.79(m, 2H), 1.82-1.90(m, 2H), 2.09-2.19(m, 2H), 2.92-3.00 (m, 2H), 3.50-3.66(m, 1H), 3.56 (s, 2H), 3.73(s, 3H), 3.73(s, 3H), 3.88(s, 3H), 3.89(s, 3H), 4.44(s, 2H), 6.72(d, 2H, J=9.3Hz), 6.78 (d, 2H, J=9.3Hz), 6.92-6.98(m, 2H), 7.16-7.21(m, 2H), 7.34(d, 1H, J=7.8Hz), 7.38(d, 1H, J=8.5 |
| # Hz), 7.46-7.59(m, 4H), 7.65(s, 1H), 7.67(s, 1H), 8.57(dd, 1H, J=5.1Hz, 0.7Hz), 8.60(d, 1H, J=5.1Hz). |
| 179 |
|
76% |
1.44(t, 3H, J=7.1Hz), 1.70-1.80 (m, 2H), 1.82-1.91(m, 2H), 2.10- 2.19(m, 2H), 2.90-3.02(m, 2H), 3.54(s, 2H), 3.73-3.78(m, 1H), 3.73(s, 3H), 3.88(s, 3H), 3.93(s, 6H), 4.09(q, 2H, J=7.1Hz), 4.45 (s, 2H), 6.73(d, 2H, J=9.2Hz), 6.78(d, 2H, J=9.2Hz), 6.97(d, 2H, J=8.8Hz), 7. 10-7.18(m, 2H), |
| # 7.15(s, 2H), 7.57(s, 1H), 7.61(s, 1H), 7.92(d, 2H, J=8.8Hz), 8.52- 8.58(m, 2H). |
| 180 |
|
93% |
1.43(t, 3H, J=6.8Hz), 1.68-1.80 (m, 2H), 1.82-1.92(m, 2H), 2.10- 2.19(m, 2H), 2.90-3.01(m, 2H), 3.56(s, 2H), 3.57-3.64(m, 1H), 3.73(s, 3H), 3.90(s, 3H), 3.96(s, 6H), 4.08(q, 2H, J=6.8Hz), 4.42 (s, 2H), 6.72(d, 2H, J=9.0Hz), 6.78(d, 2H, J=9.3Hz), 6.95(d, 2H, J=8.8Hz), 7.11(d, 1H, J=5.1 |
| # Hz), 7.20(d, 1H, J=5.1Hz), 7.22 (s, 2H), 7.58-7.62(m, 2H), 7.87 (d, 2H, J=8.8Hz), 8.52(d, 1H, J=5.1Hz), 8.58(d, 1H, J=5.1Hz). |
| 181 |
|
100% |
1.43(t, 3H, J=7.1Hz), 1.44(t, 3H, J=7.1Hz), 1.67-1.78(m, 2H), 1.82-1.90(m, 2H), 2.09-2.18(m, 2H), 2.92-3.00(m, 2H), 3.54(s, 2H), 3.55-3.65(m, 1H), 3.73(s, 3H), 4.08(q, 2H, J=7.1Hz), 4.09 (q, 2H, J=6.8Hz), 4.42(s, 2H), 6.71(d, 2H, J=9.0Hz), 6.78(d, 2H, J=9.0Hz), 6.93-7.00(m, 4H), |
| # 7.10-7.14(m, 2H), 7.60(s, 2H), 7.88(s, 2H), 7.88(d, 2H, J=8.8 Hz), 7.93(d, 2H, J=8.8Hz), 8.52 (d, 1H, J=5.1Hz), 8.56(d, 1H, J=4.9Hz). |
| 182 |
|
100% |
1.68-1.79(m, 2H), 1.82-1.90(m, 2H), 2.10-2.19(m, 2H), 2.90-3.01 (m, 2H), 3.55(s, 2H), 3.56-3.59 (m, 1H), 3.73(s, 3H), 3.89(s, 3H), 3.93(s, 6H), 3.94(s, 3H), 3.99(s, 3H), 4.45(s, 2H), 6.76(d, 2H, J=9.5Hz), 6.78(d, 2H, J=9.5Hz), 6.94(d, 1H, J=8.3Hz), 7.15(s, 2H), 7.16-7.19(m, 2H), 7.49-7.66 |
| # (m, 4H), 8.54(d, 1H, J=4.9Hz), 8.57(d, 1H, J=5.1Hz). |
| 183 |
|
100% |
1.68-1.78(m, 2H), 1.82-1.91(m, 2H), 2.10-2.18(m, 2H), 2.93-3.00 (m, 2H), 3.56(s, 2H), 3.56-3.62 (m, 1H), 3.73(s, 3H), 3.90(s, 3H), 3.93(s, 3H), 3.96(s, 6H), 3.97(S, 3H), 4.43(s, 2H), 6.72(d, 2H, J=9.3Hz), 6.78(d, 2H, J=9.3Hz), 6.92(d, 1H, J=8.3Hz), 7.12(d, 1H, J=5.1Hz), 7.20(d, 1H, J=5.1 |
| # Hz), 7.22(s, 2H), 7.42(d, 1H, J=8.5Hz, 2.2Hz), 7.58-7.63(m, 3H), 8.53(d, 1H, J=4.9Hz), 8.58 (d, 1H, J=5.1Hz). |
| 184 |
|
89% |
1.67-1.79(m, 2H), 1.84-1.90(m, 2H), 2.10-2.19(m, 2H), 2.93-3.01 (m, 2H), 3.50-3.65(m, 1H), 3.55 (s, 2H), 3.73(s, 3H), 3.94(s, 3H), 3.97(s, 3H), 3.99(s, 3H), 4.43(s, 2H), 6.72(d, 2H, J=9.3Hz), 6.78 (d, 2H, J=9.3Hz), 6.92(d, 1H, J=8.6Hz), 6.94(d, 1H, J=8.3Hz), 7.14(d, 1H, J=5.6Hz), 7.15(d, |
| # 1H, J=6.4Hz), 7.43(dd, 1H, J=8.6 Hz, 2.0Hz), 7.50(dd, 1H, J=8.3 Hz, 1.9Hz), 7.60-7.63(m, 3H), 7.66(d, 1H, J=2.2Hz), 8.53(d, 1H, J=5.1Hz), 8.57(d, 1H, J=4.9 Hz). |
| 185 |
|
100% |
1.68-1.79(m, 2H), 1.82-1.90(m, 2H), 2.10-2.20(m, 2H), 2.93-3.01 (m, 2H), 3.57(s, 2H), 3.57-3.65 (m, 1H), 3.73(s, 3H), 3.89(s, 3H), 3.93(s, 6H), 4.46(s, 2H), 6.73(d, 2H, J=7.3Hz), 6.78(d, 2H, J=7.3 Hz), 7.11-7.19(m, 2H), 7.15(s, 2H), 7.22-7.29(m, 2H), 7.34-7.40 (m, 1H), 7.58(s, 1H), 7.73(s, 1H), |
| # 7.94(t, 1H, J=8.3Hz), 8.54(d, 1H, J=5.1Hz), 8.64(d, 1H, J=4.9 Hz). |
| 186 |
|
88% |
1.68-1.79(m, 2H), 1.83-1.92(m, 2H), 2.09-2.16(m, 2H), 2.93-3.01 (m, 2H), 3.56(s, 2H), 3.56-3.62 (m, 1H), 3.73(s, 3H), 3.90(s, 3H), 3.96(s, 6H), 4.44(s, 2H), 6.71(d, 2H, J=9.3Hz), 6.77(d, 2H, J=9.3 Hz), 7.10-7.16(m, 1H), 7.17-7.26 (m, 3H), 7.22(s, 2H), 7.32-7.38 (m, 1H), 7.59(s, 1H), 7.73(s, |
| # 1H), 7.92(dt, 1H, J=8.0Hz, 2.0 Hz), 8.57-8.61(m, 2H). |
| 187 |
|
100% |
1.66-1.80(m, 2H), 1.83-1.93(m, 2H), 2.10-2.20(m, 2H), 2.92-3.02 (m, 2H), 3.53-3.65(m, 1H), 3.57 (s, 2H), 3.73(s, 3H), 4.44(s, 2H), 6.71(d, 2H, J=9.0Hz), 6.78(d, 2H, J=9.3Hz), 7.10-7.18(m, 2H), 7.19-7.29(m, 4H), 7.32-7.40(m, 2H), 7.73(s, 2H), 7.91(dd, 1H, J=8.1Hz, 1.4Hz), 7.95(dd, 1H, |
| # J=7.6Hz, 1.5Hz), 8.60(d, 1H, J=4.9Hz), 8.64(d, 1H, J=5.1Hz). |
| 188 |
|
96% |
1.67-1.80(m, 2H), 1.82-1.92(m, 2H), 2.10-2.20(m, 2H), 2.91-3.01 (m, 2H), 3.56(s, 2H), 3.56- 3.61(m, 1H), 3.73(s, 3H), 3.89(s, 3H), 3.93(s, 6H), 4.46(s, 2H), 6.73(d, 2H, J=9.3Hz), 6.78(d, 2H, J=9.3Hz), 7.06-7.19(m, 2H), 7.15(s, 2H), 7.20-7.26(m, 1H), 7.38-7.45(m, 1H), 7.56(s, 1H), |
| # 7.66-7.78(m, 3H), 8.54(d, 1H, J=5.1Hz), 8.61(d, 1H, J=4.9Hz). |
| 189 |
|
92% |
1.65-1.78(m, 2H), 1.79-1.92(m, 2H), 2.21-2.26(m, 2H), 2.90-3.01 (m, 2H), 3.56(s, 2H), 3.56-3.63 (m, 1H), 3.73(s, 3H), 3.90(s, 3H), 3.96(s, 6H), 4.44(s, 2H), 6.72(d, 2H, J=9.3Hz), 6.78(d, 2H, J=9.3 Hz), 7.08(dt, 1H, J=8.3Hz, 1.7 Hz), 7.18-7.40(m, 2H), 7.22(s, 2H), 7.37-7.43(m, 1H), 7.56-7.72 |
| # (m, 4H), 8.55-8.60(m, 2H). |
| 190 |
|
55% |
1.66-1.79(m, 2H), 1.80-1.91(m, 2H), 2.10-2.20(m, 2H), 2.88-3.01 (m, 2H), 3.50-3.66(m, 1H), 3.56 (s, 2H), 3.73(s, 3H), 4.45(s, 2H), 6.72(d, 2H, J=8.5Hz), 6.79(d, 2H, J=9.0Hz), 7.04-7.13(m, 2H), 7.19-7.25(m, 2H), 7.35-7.46(m, 2H), 7.62-7.79(m, 6H), 8.57(d, 1H, J=5.1Hz), 8.61(d, 1H, J=4.9 |
| # Hz). |
| 191 |
|
100% |
1.68-1.79(m, 2H), 1.82-1.91(m, 2H), 2.10-2.19(m, 2H), 2.92-3.00 (m, 2H), 3.55(s, 2H), 3.56-3.63 (m, 1H), 3.73(s, 3H), 3.89(s, 3H), 3.93(s, 6H), 4.45(s, 2H), 6.73(d, 2H, J=9.3Hz), 6.78(d, 2H, J=9.3 Hz), 7.11-7.19(m, 4H), 7.15(s, 2H), 7.57(s, 1H), 7.63(s, 1H), 7.92-8.01(m, 2H), 8.54(d, 1H, |
| # J=5.1Hz), 8.58(d, 1H, J=5.1Hz). |
| 192 |
|
100% |
1.68-1.79(m, 2H), 1.83-1.92(m, 2H), 2.11-2.19(m, 2H), 2.93-3.01 (m, 2H), 3.56(s, 2H), 3.57-3.62 (m, 1H), 3.73(s, 3H), 3.90(s, 3H), 3.96(s, 6H), 4.43(s, 2H), 6.72(d, 2H, J=9.3Hz), 6.78(d, 2H, J=9.3 Hz), 7.10-7.22(m, 4H), 7.22(s, 2H), 7.54-7.66(m, 2H), 7.88-7.94 (m, 2H), 8.55(d, 1H, J=4.9Hz), |
| # 8.58(d, 1H, J=4.9Hz). |
| 193 |
|
90% |
1.66-1.80(m, 2H), 1.83-1.91(m, 2H), 2.10-2.19(m, 2H), 2.92-3.00 (m, 2H), 3.50-3.66(m, 1H), 3.55 (s, 2H), 3.73(s, 3H), 4.44(s, 2H), 6.72(d, 2H, J=9.3Hz), 7.78(d, 2H, J=9.3Hz), 7.09-7.20(m, 6H), 7.62(s, 1H), 7.63(s, 1H), 7.89- 8.00(m, 4H), 8.55(d, 1H, J=5.1 Hz), 8.58(d, 1H, J=4.9Hz). |
| 194 |
|
36% |
1.68-1.80(m, 2H), 1.82-1.90(m, 2H), 2.11-2.19(m, 2H), 2.91-2.99 (m, 2H), 3.55(s, 2H), 3.56-3.62 (m, 1H), 3.73(s, 3H), 3.89(s, 3H), 3.93(s, 6H), 4.45(s, 2H), 6.73(d, 2H, J=9.3Hz), 6.78(d, 2H, J=9.3 Hz), 7.15(s, 2H), 7.16-7.26(m, 3H), 7.57(s, 1H), 7.62(s, 1H), 7.71(br, 1H), 7.80-7.90(m, 1H), |
| # 8.54(d, 1H, J=5.1Hz), 8.58(d, 1H, J=4.9Hz). |
| 195 |
|
100% |
1.60-1.80(m, 2H), 1.82-1.91(m, 2H), 2.12-2.19(m, 2H), 2.91-3.00 (m, 2H), 3.56(s, 2H), 3.56-3.64 (m, 1H), 3.73(s, 3H), 3.90(s, 3H), 3.96(s, 6H), 4.45(s, 2H), 6.72(d, 2H, J=9.0Hz), 6.78(d, 2H, J=9.0 Hz), 7.17-7.24(m, 4H), 7.25-7.27 (m, 1H), 7.60(s, 2H), 7.65(br, 1H), 7.77-7.84(m, 1H), 8.53-8.61 |
| # (m, 2H). |
| 196 |
|
100% |
1.66-1.79(m, 2H), 1.82-1.91(m, 2H), 2.09-2.20(m, 2H), 2.90-3.00 (m, 2H), 3.50-3.65(m, 1H), 3.55 (s, 2H), 3.73(s, 3H), 4.44(s, 2H), 6.72(d, 2H, J=9.3Hz), 6.79(d, 2H, J=9.3Hz), 7.18-7.28(m, 4H), 7.60(s, 1H), 7.62(s, 1H), 7.63- 7.68(m, 1H), 7.70-7.75(m, 1H), 7.77-7.89(m, 2H), 8.55(d, 1H, |
| # J=4.9Hz), 8.58(d, 1H, J=5.1Hz). |
| 197 |
|
100% |
1.68-1.80(m, 2H), 1.82-1.90(m, 2H), 2.10-2.21(m, 2H), 2.90-3.00 (m, 2H), 3.56(s, 2H), 3.56-3.63 (m, 1H), 3.73(s, 3H), 3.89(s, 3H), 3.93(s, 6H), 4.56(s, 2H), 6.73(d, 2H, J=9.3Hz), 6.78(d, 2H, J=9.3 Hz), 6.81-6.87(m, 1H), 7.15(s, 2H), 7.18(d, 1H, J=4.2Hz), 7.22- 7.26(m, 1H), 7.51-7.59(m, 3H), |
| # 7.65(s, 1H), 8.54(d, 1H, J=4.9 Hz), 8.59(d, 1H, J= 5.1Hz). |
| 198 |
|
100% |
1.65-1.79(m, 2H), 1.80-1.94(m, 2H), 2.22-2.25(m, 2H), 2.90-3.05 (m, 2H), 3.56(s, 2H), 3.56-3.65 (m, 1H), 3.73(s, 3H), 3.90(s, 3H), 3.96(s, 6H), 4.44(s, 2H), 6.72(d, 2H, J=9.2Hz), 6.78(d, 2H, J=9.2 Hz), 6.80-6.94(m, 2H), 7.22(s, 2H), 7.19-7.28(m, 1H), 7.45-7.51 (m, 2H), 7.59(s, 1H), 7.62(s, 1H), |
| # 8.56(d, 1H, J=4.9Hz), 8.59(d, 1H, J=5.1Hz). |
| 199 |
|
100% |
1.67-1.79(m, 2H), 1.82-1.92(m, 2H), 2.12-2.20(m, 2H), 2.92-2.99 (m, 2H), 3.50-3.65(m, 1H), 3.56 (s, 2H), 3.73(s, 3H), 4.45(s, 2H), 6.72(d, 2H, J=9.0Hz), 6.79(d, 2H, J=9.3Hz), 6.80-6.88(m, 2H), 7.23-7.27(m, 2H), 7.48(dd, 2H, J=8.8Hz, 2.2Hz), 7.55(dd, 2H, J=8.8Hz, 2.2Hz), 7.63(s, 1H), |
| # 7.65(s, 1H), 8.57(d, 1H, J=4.9 Hz), 8.60(d, 1H, J=4.9Hz). |
| 200 |
|
84% |
1.68-1.80(m, 2H), 1.83-1.92(m, 2H), 2.10-2.21(m, 2H), 2.91-3.00 (m, 2H), 3.56(s, 2H), 3.57-3.62 (m, 1H), 3.73(s, 3H), 3.89(s, 3H), 3.93(s, 6H), 4.45(s, 2H), 6.73(d, 2H, J=9.3Hz), 6.78(d, 2H, J=9.3 Hz), 7.15(s, 2H), 7.17(d, 1H, J=4.9Hz), 7.20(d, 1H, J=5.1Hz), 7.43(d, 2H, J=8.3Hz), 7.57(s, |
| # 1H), 7.65(s, 1H), 7.93(d, 2H, J=8.3Hz), 8.54(d, 1H, J=4.9Hz), 8.59(d, 1H, J=5.1Hz). |
| 201 |
|
72% |
1.65-1.78(m, 2H), 1.82-1.91(m, 2H), 2.10-2.16(m, 2H), 2.91-3.02 (m, 2H), 3.56(s, 2H), 3.56-3.64 (m, 1H), 3.73(s, 3H), 3.90(s, 3H), 3.96(s, 6H), 4.43(s, 2H), 6.72(d, 2H, J=9.3Hz), 6.78(d, 2H, J=9.3 Hz), 7.17-7.21(m, 1H), 7.22(2H, s), 7.41(d, 2H, J=8.7Hz), 7.48(d, 1H, J=7.8Hz), 7.59(s, 1H), 7.63 |
| # (s, 1H) 7.87(d, 2H, J=8.7Hz), 8.56(d, 1H, J=4.9Hz), 8.58(d, 1H, J=5.1Hz). |
| 202 |
|
94% |
1.67-1.88(m, 2H), 1.83-1.90(m, 2H), 2.10-2.17(m, 2H), 2.92-2.99 (m, 2H), 3.50-3.65(m, 1H), 3.55 (s, 2H), 3.73(s, 3H), 4.44(s, 2H), 6.72(d, 2H, J=9.OHz), 6.78(d, 2H, J=9.3Hz), 7.17-7.22(m, 2H), 7.39-7.45(m, 4H), 7.63(s, 1H), 7.65(s, 1H), 7.88(d, 2H, J=8.6 Hz), 7.93(d, 2H, J=8.5Hz), 8.56 |
| # (d, 1H, J=4.9Hz), 8.59(d, 1H, J=4.9Hz). |
| |
PREPARATION EXAMPLE 226
Synthesis of 4-[N-[3-(3,4,5-trimethoxyphenyl)benzyl]-N-[4-(methylsulfonyl)phenyl]amino]piperidine
To a solution of 4-[N-[3-(3,4,5-trimethoxyphenyl)benzyl]-N-[4-(methylthio)phenyl]amino]piperidine hydrochloride (52 mg, obtained in the Preparation Example 145) in dichloromethane (1 mL) was added 3-chloroperbenzoic acid (69 mg) at 0° C. The mixture was stirred at room temperature for 3 hours and saturated aqueous sodium hydrogen carbonate was added. After separating the organic layer, the aqueous layer was extracted with chloroform. Organic layers were combined, washed with brine, dried over anhydrous sodium sulfate and evaporated to give pale yellow oil of the title compound which was used for the next step without further purification.
EXAMPLE 203
Synthesis of 4-[[N-[4-(methylsulfonyl)phenyl]-N-[3-(3,4,5-trimethoxyphenyl)benzyl]amino]-1-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]piperidine dihydrochloride
Crude 4-[N-[3-(3,4,5-trimethoxyphenyl)benzyl]-N-[4-(methylsulfonyl)phenyl]amino]piperidine and 4-chloromethyl-2-(3,4,5-trimethoxyphenyl)pylidine (29 mg) were condensed in the same manner as described in Example 9. The title compound was obtained as pale yellow powder after converting a free base to a dihydrochloride.
Yield: 23 mg (26% in 2 steps). 1H-NMR (400 MHz, measured as a free base, CDCl3) 67 : 1.70-1.97 (m, 4H), 2.16-2.28 (m, 2H), 2.95-3.04 (m, 2H), 2.99 (s, 3H), 3.59 (s, 2H), 3.82 (s, 3H), 3.87-3.97 (m, 1H), 3.90 (s, 3H), 3.91 (s, 3H), 3.92 (s, 3H), 3.96 (s, 9H), 4.65 (s, 2H), 6.59 (s, 1H), 6.75 (d, 2H, J=9.3 Hz), 7.19-7.30 (m, 7H), 7.39 (dd, 1H, J=7.6, 7.6 Hz), 7.60 (s, 1H), 7.68 (d, 2H, J=9.0 Hz), 8.60 (d, 1H, J=4.9 Hz).
EXAMPLE 204
Synthesis of 4-[N-(4-metoxyphenyl)-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]-1-[[2-(3-methoxyphenyl)pyridin-4-yl]methyl]piperidine trihydrochloride
4-[N-(4-Methoxyphenyl)-N-[[3-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]amino]piperidine dihydrochloride (139 mg, obtained in the Preparation Example 98) and 4-chloromethyl-2-(3-methoxyphenyl)pyridine (70 mg, obtained in the Preparation Example 195) were condensed in the same manner described in the Example 9 to give the title compound as a trihydrochloride.
Yield: 131 mg (66%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.70-1.95 (m, 4H), 2.05-2.25 (m, 2H), 2.90-3.08 (m, 2H), 3.45-3.68 (m, 3H), 3.72 (s, 3H), 3.88 (s, 3H), 3.90 (s, 9H), 4.46 (s, 2H), 6.66 (s, 2H), 6.70-6.85 (m, 4H), 6.96 (d, 1H, J=8.3 Hz), 7.21 (br, 1H), 7.38 (t, 1H, J=7.8 Hz), 7.55 (t, 1H, J=7.8 Hz), 7.59 (s, 1H), 7.63-7.75 (m, 2H), 8.50 (s, 1H), 8.62 (m, 2H).
EXAMPLE 205
Synthesis of 4-[N-(4-metoxyphenyl)-N-[[2-(3,4,5-trimethoxyphenyl)pyridin-4-yl]methyl]amino]-1-[[2-(3,4-dimethoxyphenyl)pyridin-4-yl]methyl]piperidine trihydrochloride
4-[N-(4-Methoxyphenyl)-N-[[3-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]amino]piperidine dihydrochloride (139 mg, obtained in the Preparation Example 98) and 4-chloromethyl-2-(3,4-dimethoxyphenyl)pyridine (80 mg, obtained in the Preparation Example 197) were condensed in the same manner described in the Example 9 to give the title compound as a trihydrochloride.
Yield: 139 mg (67%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.70-1.95 (m, 4H), 2.05-2.20 (m, 2H), 2.90-3.05 (m, 2H), 3.45-3.60 (m, 3H), 3.73 (s, 3H), 3.88 (s, 3H), 3.89 (s, 6H), 3.94 (s, 3H), 4.00 (s, 3H), 4.46 (s, 2H), 6.65 (s, 2H), 6.74-6.82 (m, 4H), 6.94 (d, 1H, J=8.3 Hz), 7.15 (br, 1H), 7.52 (br, 1H), 7.58-7.71 (m, 3H), 8.50 (s, 1H), 8.57 (d, 1H, J=5.2 Hz), 8.62 (br, 1H).
EXAMPLE 206
Synthesis of 4-[N-(4-fluorophenyl)-N-[[3-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]amino]-1-[[2-(3-methoxyphenyl)pyridin-4-yl]methyl]piperidine trihydrochloride
4-[N-(4-Fluorophenyl)-N-[[3-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]amino]piperidine dihydrochloride (135 mg, obtained in the Preparation Example 183) and 4-chloromethyl-2-(3-methoxyphenyl)pyridine (70 mg, obtained in the Preparation Example 195) were condensed in the same manner described in the Example 9 to give the title compound as a trihydrochloride.
Yield: 178 mg (92%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.73-1.95 (m, 4H), 2.10-2.25 (m, 2H), 2.93-3.05 (m, 2H), 3.57 (s, 2H), 3.64 (br, 1H), 3.88 (s, 3H), 3.89 (s, 9H), 4.51 (s, 2H), 6.66 (s, 2H), 6.70-6.76 (m, 2H), 6.90 (t, 2H, J=8.3 Hz), 6.96 (d, 1H, J=8.3 Hz), 7.21 (br, 1H), 7.38 (t, 1H, J=8.0 Hz), 7.54 (d, 1H, J=7.8 Hz), 7.58 (s, 1H), 7.65 (s, 1H), 7.74 (br, 1H), 8.50 (s, 1H), 8.61 (d, 1H, J=5.1 Hz), 8.65 (br, 1H).
EXAMPLE 207
Synthesis of 4-[N-(4-fluorophenyl)-N-[[3-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]amino]-1-[[2-(3,4-dimethoxyphenyl)pyridin-4-yl]methyl]piperidine trihydrochloride
4-[N-(4-Fluorophenyl)-N-[[3-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]amino]piperidine dihydrochloride (135 mg, obtained in the Preparation Example 183) and 4-chloromethyl-2-(3,4-dimethoxyphenyl)pyridine (80 mg, obtained in the Preparation Example 197) were condensed in the same manner described in the Example 9 to give the title compound as a trihydrochloride.
Yield: 195 mg (96%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.70-1.95 (m, 4H), 2.10-2.24 (m, 2H), 2.94-3.09 (m, 2H), 3.57 (s, 2H), 3.64 (br, 1H), 3.88 (s, 3H), 3.89 (s, 6H), 3.94 (s, 3H), 4.00 (s, 3H), 4.51 (s, 2H), 6.65 (s, 2H), 6.69-6.78 (m, 2H), 6.86-6.97 (m, 3H), 7.16 (d, 1H, J=4.9 Hz), 7.51 (d, 1H, J=8.5 Hz), 7.60-7.70 (m, 3H), 8.50 (s, 1H), 8.58 (d, 1H, J=4.9 Hz), 8.65 (s, 1H).
EXAMPLE 208
Synthesis of 4-[N-(3,4-difluorophenyl)-N-[[3-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]amino]-1-[[2-(3-methoxyphenyl)pyridin-4-yl]methyl]piperidine trihydrochloride
4-[N-(3,4-Difluorophenyl)-N-[[3-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]amino]piperidine dihydrochloride (160 mg, obtained in the Preparation Example 176) and 4-chloromethyl-2-(3-methoxyphenyl)pyridine (80 mg, obtained in the Preparation Example 195) were condensed in the same manner described in the Example 9 to give the title compound as a trihydrochloride.
Yield: 130 mg (57%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.73-1.90 (m, 4H), 2.01-2.24 (m, 2H), 2.92-3.05 (m, 2H), 3.57 (s, 2H), 3.67 (br, 1H), 3.88 (s, 3H), 3.89 (s, 3H), 3.90 (s,6H), 4.52 (s, 2H), 6.36-6.42 (m, 1H), 6.50-6.58 (m, 1H), 6.67 (s, 2H), 6.93-7.01 (m, 2H), 7.20 (br, 1H), 7.38 (t, 1H, J=7.8 Hz), 7.52-7.62 (m, 2H), 7.62-7.72 (m, 2H), 8.48 (br, 1H), 8.61 (br, 1H), 8.66 (d, 1H, J=2.0 Hz).
EXAMPLE 209
Synthesis of 1-[[2-(3-methoxyphenyl)pyridin-4-yl]methyl]-4-[N-[[3-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]-N-(4-methylthiophenyl)amino]piperidine
4-[N-(4-Metythiophenyl)-N-[[3-(3,4,5-trimethoxyphenyl)pyridin-5-yl]methyl]amino]piperidine dihydrochloride (121 mg, obtained in the Preparation Example 143) and 4-chloromethyl-2-(4-methoxyphenyl)pyridine (55 mg, obtained in the Preparation Example 195) were condensed in the same manner described in the Example 9 to give the title compound.
Yield: 71 mg (44%). 1H-NMR (400 MHz, measured as a free base, CDCl3) δ: 1.72-1.83 (m, 4H), 2.12-2.20 (m, 2H), 2.37 (s, 3H), 2.97 (d, 2H, J=10.8 Hz), 3.56 (s, 2H), 3.75-3.81 (m, 1H), 3.86 (s, 3H), 3.87 (s, 6H), 4.54 (s, 2H), 6.64-6.69 (m, 3H), 6.94 (dd, 1H, J=7.8 Hz, 1.9 Hz), 7.17-7.26 (m, 4H), 7.35 (t, 1H, J=7.8 Hz), 7.51-7.66 (m, 4H), 8.47 (s, 1H), 8.59 (d, 1H, J=4.6 Hz), 8.63 (s, 1H).
Test Example 1
Inhibitory Effect on Cell Adhesion
The test was conducted by reference to the method by Ross et al. (J. Biol. Chem., 267, 8537-8543 (1992)). More specifically, after human umbilical venous endothelial cells (HUVEC) were cultured on a 48-well plate to confluent growth, TNFα was added thereto. Upon elapsed time of 5 hours after the addition, U937, which was a human monocytic/histocytic cell fluorescence-labeled with PKH2 (product of Sigma-Aldrich Co., Ltd.), was added in a proportion of 1×106 cells per well. After the plate was left at rest at room temperature for 1 hour, unadhered U937 was washed out and lysed in 1% Triton X-100 to measure a remaining fluorescence intensity (excitation wavelength: 485 nm; measuring wavelength: 530 nm). HUVEC and U937 were cultured in EGM-2 (product of Sanko Junyaku K. K.) and 10% FCS-containing RPMI1640, respectively. Each test agent was added to HUVEC upon the addition of TNFα and to U937 24 hours prior to the cell adhesion test. The inhibitory activity was calculated out as IC50 value after inhibitory ratio at each concentration of test compounds was determined. The inhibitory ratio was calculated according to the equation [(C−B)/(A−B)×100 (%)], wherein A is the number of U937 cells adhered to HUVEC stimulated by TNFα when no test agent was added, B is the number of U937 cells adhered to HUVEC not stimulated by TNFα when no test agent was added, and C is the number of U937 cells adhered to HUVEC stimulated by TNFα when the test agent was added. The results are shown in Table 1. As control compounds, Test Compound 1 described in Japanese Patent Application Laid-Open No. 9-143075 and dilazep described in Japanese Patent Application Laid-Open No. 11-92382 were simultaneously evaluated.
| |
TABLE 1 |
| |
|
| |
Example |
IC50 (μM) |
| |
|
| |
| |
3 |
0.3 |
| |
5 |
0.2 |
| |
7 |
0.3 |
| |
10 |
0.04 |
| |
13 |
0.03 |
| |
16 |
0.3 |
| |
19 |
0.3 |
| |
23 |
0.03 |
| |
29 |
0.03 |
| |
36 |
0.2 |
| |
40 |
0.2 |
| |
42 |
0.07 |
| |
67 |
0.09 |
| |
88 |
0.07 |
| |
89 |
0.09 |
| |
111 |
0.08 |
| |
157 |
0.09 |
| |
Test compound 1 |
10 |
| |
Dilazep |
>10 |
| |
|
As mentioned above, the compound of the present invention is characterized in that the cyclic amine has two phenyl-pyridyl or biphenyl groups, or phenyl-pyridyl and biphenyl groups. The inhibitory effect on cell adhesion of the cyclic amine compound having pyridyl groups at the both ends thereof obtained in Preparation Example 91 was examined similar to the above procedure. As the result, the compound was inactive even at a concentration as high as 10 μM.
Specific formulation examples will hereinafter be described.
EXAMPLE 210
Capsule Preparation
| |
|
| |
Compound obtained by Example 13 |
30 mg |
| |
Microcrystalline cellulose |
30 mg |
| |
Lactose |
57 mg |
| |
Magnesium stearate |
3 mg |
| |
Total amount |
120 mg. |
| |
|
The above ingredients were mixed in accordance with a method known per se in the art and then charged in capsules to obtain capsule preparations.
EXAMPLE 211
Tablet Preparation
| |
|
| |
Compound obtained by Example 13 |
30 mg |
| |
Starch |
44 mg |
| |
Starch (for glue) |
5.6 mg |
| |
Magnesium stearate |
0.4 mg |
| |
Calcium carboxymethyl cellulose |
20 mg |
| |
Total amount |
100 mg. |
| |
|
The above ingredients were mixed in accordance with a method known per se in the art to obtain tablet preparations.
EXAMPLE 212
Injection Preparation
Compound obtained by Example 13 (100 mg) and sodium chloride (900 mg) were dissolved in distilled water (about 80 mL) for injection, and distilled water for injection was added to the resultant solution to 100 mL in total. This diluted solution was sterilized by filtration and then subdivided and charged into 10 ampoules, and the ampoules were sealed to obtain injection preparations.
As described above, the compounds (1) according to the present invention have inhibitory effects on both cell adhesion and cell infiltration and are useful as medicines for prevention or treatment of allergy, asthma, rheumatism, arteriosclerosis, inflammatory, Sjogren's syndrome, etc.
Obviously, numerous modifications of the above teachings are apparent to those skilled in the art. Therefore, within the scope of the appended claims the invention may be practiced otherwise than as specifically described herein.