US6228551B1 - Toner for developing electrostatic image - Google Patents
Toner for developing electrostatic image Download PDFInfo
- Publication number
- US6228551B1 US6228551B1 US09/469,383 US46938399A US6228551B1 US 6228551 B1 US6228551 B1 US 6228551B1 US 46938399 A US46938399 A US 46938399A US 6228551 B1 US6228551 B1 US 6228551B1
- Authority
- US
- United States
- Prior art keywords
- toner
- molecular weight
- polypropylene
- particle
- particles
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Lifetime
Links
- 239000002245 particle Substances 0.000 claims abstract description 144
- -1 polypropylene Polymers 0.000 claims abstract description 122
- 239000004743 Polypropylene Substances 0.000 claims abstract description 114
- 229920001155 polypropylene Polymers 0.000 claims abstract description 114
- 238000000034 method Methods 0.000 claims abstract description 63
- 229920005989 resin Polymers 0.000 claims abstract description 45
- 239000011347 resin Substances 0.000 claims abstract description 45
- 239000003086 colorant Substances 0.000 claims abstract description 14
- 239000011230 binding agent Substances 0.000 claims abstract description 13
- 238000004062 sedimentation Methods 0.000 claims abstract description 12
- 239000000654 additive Substances 0.000 claims description 9
- 230000000996 additive effect Effects 0.000 claims description 9
- 239000000203 mixture Substances 0.000 description 22
- 239000007788 liquid Substances 0.000 description 14
- 239000011572 manganese Substances 0.000 description 14
- 230000008569 process Effects 0.000 description 14
- 238000000926 separation method Methods 0.000 description 14
- 230000000052 comparative effect Effects 0.000 description 13
- PPBRXRYQALVLMV-UHFFFAOYSA-N Styrene Chemical compound C=CC1=CC=CC=C1 PPBRXRYQALVLMV-UHFFFAOYSA-N 0.000 description 12
- 239000003795 chemical substances by application Substances 0.000 description 12
- 239000007771 core particle Substances 0.000 description 12
- 238000002156 mixing Methods 0.000 description 12
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 11
- 229910000859 α-Fe Inorganic materials 0.000 description 11
- 108091008695 photoreceptors Proteins 0.000 description 10
- 239000000725 suspension Substances 0.000 description 9
- 230000005291 magnetic effect Effects 0.000 description 8
- 229910052751 metal Inorganic materials 0.000 description 8
- 239000002184 metal Substances 0.000 description 8
- 239000000049 pigment Substances 0.000 description 8
- BQCIDUSAKPWEOX-UHFFFAOYSA-N 1,1-Difluoroethene Chemical compound FC(F)=C BQCIDUSAKPWEOX-UHFFFAOYSA-N 0.000 description 7
- 230000015572 biosynthetic process Effects 0.000 description 7
- 239000006229 carbon black Substances 0.000 description 7
- 238000009826 distribution Methods 0.000 description 7
- 239000000843 powder Substances 0.000 description 6
- 238000002360 preparation method Methods 0.000 description 6
- 239000000377 silicon dioxide Substances 0.000 description 6
- 239000004094 surface-active agent Substances 0.000 description 6
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 5
- 206010027146 Melanoderma Diseases 0.000 description 5
- 239000011248 coating agent Substances 0.000 description 5
- 238000000576 coating method Methods 0.000 description 5
- 238000011109 contamination Methods 0.000 description 5
- 229920001577 copolymer Polymers 0.000 description 5
- 230000002209 hydrophobic effect Effects 0.000 description 5
- SZVJSHCCFOBDDC-UHFFFAOYSA-N iron(II,III) oxide Inorganic materials O=[Fe]O[Fe]O[Fe]=O SZVJSHCCFOBDDC-UHFFFAOYSA-N 0.000 description 5
- 238000002844 melting Methods 0.000 description 5
- 230000008018 melting Effects 0.000 description 5
- 229920002050 silicone resin Polymers 0.000 description 5
- 238000003756 stirring Methods 0.000 description 5
- SOGAXMICEFXMKE-UHFFFAOYSA-N Butylmethacrylate Chemical compound CCCCOC(=O)C(C)=C SOGAXMICEFXMKE-UHFFFAOYSA-N 0.000 description 4
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 4
- BAPJBEWLBFYGME-UHFFFAOYSA-N Methyl acrylate Chemical compound COC(=O)C=C BAPJBEWLBFYGME-UHFFFAOYSA-N 0.000 description 4
- PXHVJJICTQNCMI-UHFFFAOYSA-N Nickel Chemical compound [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 description 4
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 description 4
- 229910052784 alkaline earth metal Inorganic materials 0.000 description 4
- 229910045601 alloy Inorganic materials 0.000 description 4
- 239000000956 alloy Substances 0.000 description 4
- 238000006243 chemical reaction Methods 0.000 description 4
- 230000005294 ferromagnetic effect Effects 0.000 description 4
- 239000010419 fine particle Substances 0.000 description 4
- 238000004898 kneading Methods 0.000 description 4
- 229920001225 polyester resin Polymers 0.000 description 4
- 239000004645 polyester resin Substances 0.000 description 4
- 239000000126 substance Substances 0.000 description 4
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 4
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 3
- 238000004140 cleaning Methods 0.000 description 3
- 150000001875 compounds Chemical class 0.000 description 3
- 239000007822 coupling agent Substances 0.000 description 3
- 238000011156 evaluation Methods 0.000 description 3
- 238000010438 heat treatment Methods 0.000 description 3
- JEIPFZHSYJVQDO-UHFFFAOYSA-N iron(III) oxide Inorganic materials O=[Fe]O[Fe]=O JEIPFZHSYJVQDO-UHFFFAOYSA-N 0.000 description 3
- 239000006249 magnetic particle Substances 0.000 description 3
- 229920002545 silicone oil Polymers 0.000 description 3
- BJQFWAQRPATHTR-UHFFFAOYSA-N 1,2-dichloro-4-ethenylbenzene Chemical compound ClC1=CC=C(C=C)C=C1Cl BJQFWAQRPATHTR-UHFFFAOYSA-N 0.000 description 2
- RFFLAFLAYFXFSW-UHFFFAOYSA-N 1,2-dichlorobenzene Chemical compound ClC1=CC=CC=C1Cl RFFLAFLAYFXFSW-UHFFFAOYSA-N 0.000 description 2
- KTZVZZJJVJQZHV-UHFFFAOYSA-N 1-chloro-4-ethenylbenzene Chemical compound ClC1=CC=C(C=C)C=C1 KTZVZZJJVJQZHV-UHFFFAOYSA-N 0.000 description 2
- DMADTXMQLFQQII-UHFFFAOYSA-N 1-decyl-4-ethenylbenzene Chemical compound CCCCCCCCCCC1=CC=C(C=C)C=C1 DMADTXMQLFQQII-UHFFFAOYSA-N 0.000 description 2
- WJNKJKGZKFOLOJ-UHFFFAOYSA-N 1-dodecyl-4-ethenylbenzene Chemical compound CCCCCCCCCCCCC1=CC=C(C=C)C=C1 WJNKJKGZKFOLOJ-UHFFFAOYSA-N 0.000 description 2
- OEVVKKAVYQFQNV-UHFFFAOYSA-N 1-ethenyl-2,4-dimethylbenzene Chemical compound CC1=CC=C(C=C)C(C)=C1 OEVVKKAVYQFQNV-UHFFFAOYSA-N 0.000 description 2
- NVZWEEGUWXZOKI-UHFFFAOYSA-N 1-ethenyl-2-methylbenzene Chemical compound CC1=CC=CC=C1C=C NVZWEEGUWXZOKI-UHFFFAOYSA-N 0.000 description 2
- JZHGRUMIRATHIU-UHFFFAOYSA-N 1-ethenyl-3-methylbenzene Chemical compound CC1=CC=CC(C=C)=C1 JZHGRUMIRATHIU-UHFFFAOYSA-N 0.000 description 2
- WHFHDVDXYKOSKI-UHFFFAOYSA-N 1-ethenyl-4-ethylbenzene Chemical compound CCC1=CC=C(C=C)C=C1 WHFHDVDXYKOSKI-UHFFFAOYSA-N 0.000 description 2
- LCNAQVGAHQVWIN-UHFFFAOYSA-N 1-ethenyl-4-hexylbenzene Chemical compound CCCCCCC1=CC=C(C=C)C=C1 LCNAQVGAHQVWIN-UHFFFAOYSA-N 0.000 description 2
- LUWBJDCKJAZYKZ-UHFFFAOYSA-N 1-ethenyl-4-nonylbenzene Chemical compound CCCCCCCCCC1=CC=C(C=C)C=C1 LUWBJDCKJAZYKZ-UHFFFAOYSA-N 0.000 description 2
- HLRQDIVVLOCZPH-UHFFFAOYSA-N 1-ethenyl-4-octylbenzene Chemical compound CCCCCCCCC1=CC=C(C=C)C=C1 HLRQDIVVLOCZPH-UHFFFAOYSA-N 0.000 description 2
- QEDJMOONZLUIMC-UHFFFAOYSA-N 1-tert-butyl-4-ethenylbenzene Chemical compound CC(C)(C)C1=CC=C(C=C)C=C1 QEDJMOONZLUIMC-UHFFFAOYSA-N 0.000 description 2
- SJIXRGNQPBQWMK-UHFFFAOYSA-N 2-(diethylamino)ethyl 2-methylprop-2-enoate Chemical compound CCN(CC)CCOC(=O)C(C)=C SJIXRGNQPBQWMK-UHFFFAOYSA-N 0.000 description 2
- QHVBLSNVXDSMEB-UHFFFAOYSA-N 2-(diethylamino)ethyl prop-2-enoate Chemical compound CCN(CC)CCOC(=O)C=C QHVBLSNVXDSMEB-UHFFFAOYSA-N 0.000 description 2
- DPBJAVGHACCNRL-UHFFFAOYSA-N 2-(dimethylamino)ethyl prop-2-enoate Chemical compound CN(C)CCOC(=O)C=C DPBJAVGHACCNRL-UHFFFAOYSA-N 0.000 description 2
- GOXQRTZXKQZDDN-UHFFFAOYSA-N 2-Ethylhexyl acrylate Chemical compound CCCCC(CC)COC(=O)C=C GOXQRTZXKQZDDN-UHFFFAOYSA-N 0.000 description 2
- WDQMWEYDKDCEHT-UHFFFAOYSA-N 2-ethylhexyl 2-methylprop-2-enoate Chemical compound CCCCC(CC)COC(=O)C(C)=C WDQMWEYDKDCEHT-UHFFFAOYSA-N 0.000 description 2
- RUMACXVDVNRZJZ-UHFFFAOYSA-N 2-methylpropyl 2-methylprop-2-enoate Chemical compound CC(C)COC(=O)C(C)=C RUMACXVDVNRZJZ-UHFFFAOYSA-N 0.000 description 2
- CFVWNXQPGQOHRJ-UHFFFAOYSA-N 2-methylpropyl prop-2-enoate Chemical compound CC(C)COC(=O)C=C CFVWNXQPGQOHRJ-UHFFFAOYSA-N 0.000 description 2
- JLBJTVDPSNHSKJ-UHFFFAOYSA-N 4-Methylstyrene Chemical compound CC1=CC=C(C=C)C=C1 JLBJTVDPSNHSKJ-UHFFFAOYSA-N 0.000 description 2
- JTHZUSWLNCPZLX-UHFFFAOYSA-N 6-fluoro-3-methyl-2h-indazole Chemical compound FC1=CC=C2C(C)=NNC2=C1 JTHZUSWLNCPZLX-UHFFFAOYSA-N 0.000 description 2
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 2
- JIGUQPWFLRLWPJ-UHFFFAOYSA-N Ethyl acrylate Chemical compound CCOC(=O)C=C JIGUQPWFLRLWPJ-UHFFFAOYSA-N 0.000 description 2
- VVQNEPGJFQJSBK-UHFFFAOYSA-N Methyl methacrylate Chemical compound COC(=O)C(C)=C VVQNEPGJFQJSBK-UHFFFAOYSA-N 0.000 description 2
- 239000006087 Silane Coupling Agent Substances 0.000 description 2
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 description 2
- XLOMVQKBTHCTTD-UHFFFAOYSA-N Zinc monoxide Chemical compound [Zn]=O XLOMVQKBTHCTTD-UHFFFAOYSA-N 0.000 description 2
- MCMNRKCIXSYSNV-UHFFFAOYSA-N Zirconium dioxide Chemical compound O=[Zr]=O MCMNRKCIXSYSNV-UHFFFAOYSA-N 0.000 description 2
- 125000005396 acrylic acid ester group Chemical group 0.000 description 2
- 239000000853 adhesive Substances 0.000 description 2
- 230000001070 adhesive effect Effects 0.000 description 2
- 229910052783 alkali metal Inorganic materials 0.000 description 2
- 150000001340 alkali metals Chemical class 0.000 description 2
- 125000000217 alkyl group Chemical group 0.000 description 2
- XYLMUPLGERFSHI-UHFFFAOYSA-N alpha-Methylstyrene Chemical compound CC(=C)C1=CC=CC=C1 XYLMUPLGERFSHI-UHFFFAOYSA-N 0.000 description 2
- CQEYYJKEWSMYFG-UHFFFAOYSA-N butyl acrylate Chemical compound CCCCOC(=O)C=C CQEYYJKEWSMYFG-UHFFFAOYSA-N 0.000 description 2
- 239000011247 coating layer Substances 0.000 description 2
- 238000009833 condensation Methods 0.000 description 2
- 230000005494 condensation Effects 0.000 description 2
- GMSCBRSQMRDRCD-UHFFFAOYSA-N dodecyl 2-methylprop-2-enoate Chemical compound CCCCCCCCCCCCOC(=O)C(C)=C GMSCBRSQMRDRCD-UHFFFAOYSA-N 0.000 description 2
- GVGUFUZHNYFZLC-UHFFFAOYSA-N dodecyl benzenesulfonate;sodium Chemical compound [Na].CCCCCCCCCCCCOS(=O)(=O)C1=CC=CC=C1 GVGUFUZHNYFZLC-UHFFFAOYSA-N 0.000 description 2
- SUPCQIBBMFXVTL-UHFFFAOYSA-N ethyl 2-methylprop-2-enoate Chemical compound CCOC(=O)C(C)=C SUPCQIBBMFXVTL-UHFFFAOYSA-N 0.000 description 2
- FFUAGWLWBBFQJT-UHFFFAOYSA-N hexamethyldisilazane Chemical compound C[Si](C)(C)N[Si](C)(C)C FFUAGWLWBBFQJT-UHFFFAOYSA-N 0.000 description 2
- AMWRITDGCCNYAT-UHFFFAOYSA-L hydroxy(oxo)manganese;manganese Chemical compound [Mn].O[Mn]=O.O[Mn]=O AMWRITDGCCNYAT-UHFFFAOYSA-L 0.000 description 2
- 229910052742 iron Inorganic materials 0.000 description 2
- PBOSTUDLECTMNL-UHFFFAOYSA-N lauryl acrylate Chemical compound CCCCCCCCCCCCOC(=O)C=C PBOSTUDLECTMNL-UHFFFAOYSA-N 0.000 description 2
- 239000010410 layer Substances 0.000 description 2
- 229910052749 magnesium Inorganic materials 0.000 description 2
- 239000011777 magnesium Substances 0.000 description 2
- 125000005397 methacrylic acid ester group Chemical group 0.000 description 2
- 229910052759 nickel Inorganic materials 0.000 description 2
- HMZGPNHSPWNGEP-UHFFFAOYSA-N octadecyl 2-methylprop-2-enoate Chemical compound CCCCCCCCCCCCCCCCCCOC(=O)C(C)=C HMZGPNHSPWNGEP-UHFFFAOYSA-N 0.000 description 2
- NZIDBRBFGPQCRY-UHFFFAOYSA-N octyl 2-methylprop-2-enoate Chemical compound CCCCCCCCOC(=O)C(C)=C NZIDBRBFGPQCRY-UHFFFAOYSA-N 0.000 description 2
- ANISOHQJBAQUQP-UHFFFAOYSA-N octyl prop-2-enoate Chemical compound CCCCCCCCOC(=O)C=C ANISOHQJBAQUQP-UHFFFAOYSA-N 0.000 description 2
- 239000003921 oil Substances 0.000 description 2
- HDBWAWNLGGMZRQ-UHFFFAOYSA-N p-Vinylbiphenyl Chemical compound C1=CC(C=C)=CC=C1C1=CC=CC=C1 HDBWAWNLGGMZRQ-UHFFFAOYSA-N 0.000 description 2
- PNJWIWWMYCMZRO-UHFFFAOYSA-N pent‐4‐en‐2‐one Natural products CC(=O)CC=C PNJWIWWMYCMZRO-UHFFFAOYSA-N 0.000 description 2
- QIWKUEJZZCOPFV-UHFFFAOYSA-N phenyl 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OC1=CC=CC=C1 QIWKUEJZZCOPFV-UHFFFAOYSA-N 0.000 description 2
- WRAQQYDMVSCOTE-UHFFFAOYSA-N phenyl prop-2-enoate Chemical compound C=CC(=O)OC1=CC=CC=C1 WRAQQYDMVSCOTE-UHFFFAOYSA-N 0.000 description 2
- 239000011164 primary particle Substances 0.000 description 2
- BOQSSGDQNWEFSX-UHFFFAOYSA-N propan-2-yl 2-methylprop-2-enoate Chemical compound CC(C)OC(=O)C(C)=C BOQSSGDQNWEFSX-UHFFFAOYSA-N 0.000 description 2
- LYBIZMNPXTXVMV-UHFFFAOYSA-N propan-2-yl prop-2-enoate Chemical compound CC(C)OC(=O)C=C LYBIZMNPXTXVMV-UHFFFAOYSA-N 0.000 description 2
- 229940080264 sodium dodecylbenzenesulfonate Drugs 0.000 description 2
- 239000002904 solvent Substances 0.000 description 2
- 125000001424 substituent group Chemical group 0.000 description 2
- SJMYWORNLPSJQO-UHFFFAOYSA-N tert-butyl 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OC(C)(C)C SJMYWORNLPSJQO-UHFFFAOYSA-N 0.000 description 2
- ISXSCDLOGDJUNJ-UHFFFAOYSA-N tert-butyl prop-2-enoate Chemical compound CC(C)(C)OC(=O)C=C ISXSCDLOGDJUNJ-UHFFFAOYSA-N 0.000 description 2
- 239000010936 titanium Substances 0.000 description 2
- 229910052719 titanium Inorganic materials 0.000 description 2
- MIZLGWKEZAPEFJ-UHFFFAOYSA-N 1,1,2-trifluoroethene Chemical group FC=C(F)F MIZLGWKEZAPEFJ-UHFFFAOYSA-N 0.000 description 1
- QOVCUELHTLHMEN-UHFFFAOYSA-N 1-butyl-4-ethenylbenzene Chemical compound CCCCC1=CC=C(C=C)C=C1 QOVCUELHTLHMEN-UHFFFAOYSA-N 0.000 description 1
- IEKHISJGRIEHRE-UHFFFAOYSA-N 16-methylheptadecanoic acid;propan-2-ol;titanium Chemical compound [Ti].CC(C)O.CC(C)CCCCCCCCCCCCCCC(O)=O.CC(C)CCCCCCCCCCCCCCC(O)=O.CC(C)CCCCCCCCCCCCCCC(O)=O IEKHISJGRIEHRE-UHFFFAOYSA-N 0.000 description 1
- QTKPMCIBUROOGY-UHFFFAOYSA-N 2,2,2-trifluoroethyl 2-methylprop-2-enoate Chemical compound CC(=C)C(=O)OCC(F)(F)F QTKPMCIBUROOGY-UHFFFAOYSA-N 0.000 description 1
- JKNCOURZONDCGV-UHFFFAOYSA-N 2-(dimethylamino)ethyl 2-methylprop-2-enoate Chemical compound CN(C)CCOC(=O)C(C)=C JKNCOURZONDCGV-UHFFFAOYSA-N 0.000 description 1
- GZWRMQNNGRSSNL-UHFFFAOYSA-N 3-trimethoxysilylpropan-1-amine;hydrochloride Chemical compound [Cl-].CO[Si](OC)(OC)CCC[NH3+] GZWRMQNNGRSSNL-UHFFFAOYSA-N 0.000 description 1
- XDLMVUHYZWKMMD-UHFFFAOYSA-N 3-trimethoxysilylpropyl 2-methylprop-2-enoate Chemical compound CO[Si](OC)(OC)CCCOC(=O)C(C)=C XDLMVUHYZWKMMD-UHFFFAOYSA-N 0.000 description 1
- IICCLYANAQEHCI-UHFFFAOYSA-N 4,5,6,7-tetrachloro-3',6'-dihydroxy-2',4',5',7'-tetraiodospiro[2-benzofuran-3,9'-xanthene]-1-one Chemical compound O1C(=O)C(C(=C(Cl)C(Cl)=C2Cl)Cl)=C2C21C1=CC(I)=C(O)C(I)=C1OC1=C(I)C(O)=C(I)C=C21 IICCLYANAQEHCI-UHFFFAOYSA-N 0.000 description 1
- QPQKUYVSJWQSDY-UHFFFAOYSA-N 4-phenyldiazenylaniline Chemical class C1=CC(N)=CC=C1N=NC1=CC=CC=C1 QPQKUYVSJWQSDY-UHFFFAOYSA-N 0.000 description 1
- KHLRJDNGHBXOSV-UHFFFAOYSA-N 5-trimethoxysilylpentane-1,3-diamine Chemical compound CO[Si](OC)(OC)CCC(N)CCN KHLRJDNGHBXOSV-UHFFFAOYSA-N 0.000 description 1
- 229910000505 Al2TiO5 Inorganic materials 0.000 description 1
- 229910052580 B4C Inorganic materials 0.000 description 1
- 229910052582 BN Inorganic materials 0.000 description 1
- PZNSFCLAULLKQX-UHFFFAOYSA-N Boron nitride Chemical compound N#B PZNSFCLAULLKQX-UHFFFAOYSA-N 0.000 description 1
- NLZUEZXRPGMBCV-UHFFFAOYSA-N Butylhydroxytoluene Chemical compound CC1=CC(C(C)(C)C)=C(O)C(C(C)(C)C)=C1 NLZUEZXRPGMBCV-UHFFFAOYSA-N 0.000 description 1
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 1
- KRHYYFGTRYWZRS-UHFFFAOYSA-M Fluoride anion Chemical compound [F-] KRHYYFGTRYWZRS-UHFFFAOYSA-M 0.000 description 1
- YCKRFDGAMUMZLT-UHFFFAOYSA-N Fluorine atom Chemical compound [F] YCKRFDGAMUMZLT-UHFFFAOYSA-N 0.000 description 1
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 1
- SQTSHRKDEDOZME-UHFFFAOYSA-N NCCC(CC[SiH](OC)OC)N Chemical compound NCCC(CC[SiH](OC)OC)N SQTSHRKDEDOZME-UHFFFAOYSA-N 0.000 description 1
- NRTOMJZYCJJWKI-UHFFFAOYSA-N Titanium nitride Chemical compound [Ti]#N NRTOMJZYCJJWKI-UHFFFAOYSA-N 0.000 description 1
- WGLPBDUCMAPZCE-UHFFFAOYSA-N Trioxochromium Chemical compound O=[Cr](=O)=O WGLPBDUCMAPZCE-UHFFFAOYSA-N 0.000 description 1
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical compound [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 description 1
- NWLCFADDJOPOQC-UHFFFAOYSA-N [Mn].[Cu].[Sn] Chemical compound [Mn].[Cu].[Sn] NWLCFADDJOPOQC-UHFFFAOYSA-N 0.000 description 1
- UMHKOAYRTRADAT-UHFFFAOYSA-N [hydroxy(octoxy)phosphoryl] octyl hydrogen phosphate Chemical compound CCCCCCCCOP(O)(=O)OP(O)(=O)OCCCCCCCC UMHKOAYRTRADAT-UHFFFAOYSA-N 0.000 description 1
- 230000001133 acceleration Effects 0.000 description 1
- 239000006230 acetylene black Substances 0.000 description 1
- 125000004423 acyloxy group Chemical group 0.000 description 1
- 238000004220 aggregation Methods 0.000 description 1
- 230000002776 aggregation Effects 0.000 description 1
- 125000003545 alkoxy group Chemical group 0.000 description 1
- PNEYBMLMFCGWSK-UHFFFAOYSA-N aluminium oxide Inorganic materials [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 description 1
- 229910000410 antimony oxide Inorganic materials 0.000 description 1
- IRERQBUNZFJFGC-UHFFFAOYSA-L azure blue Chemical compound [Na+].[Na+].[Na+].[Na+].[Na+].[Na+].[Na+].[Na+].[Al+3].[Al+3].[Al+3].[Al+3].[Al+3].[Al+3].[S-]S[S-].[O-][Si]([O-])([O-])[O-].[O-][Si]([O-])([O-])[O-].[O-][Si]([O-])([O-])[O-].[O-][Si]([O-])([O-])[O-].[O-][Si]([O-])([O-])[O-].[O-][Si]([O-])([O-])[O-] IRERQBUNZFJFGC-UHFFFAOYSA-L 0.000 description 1
- 229910052788 barium Inorganic materials 0.000 description 1
- JRPBQTZRNDNNOP-UHFFFAOYSA-N barium titanate Chemical compound [Ba+2].[Ba+2].[O-][Ti]([O-])([O-])[O-] JRPBQTZRNDNNOP-UHFFFAOYSA-N 0.000 description 1
- 229910002113 barium titanate Inorganic materials 0.000 description 1
- INAHAJYZKVIDIZ-UHFFFAOYSA-N boron carbide Chemical compound B12B3B4C32B41 INAHAJYZKVIDIZ-UHFFFAOYSA-N 0.000 description 1
- 150000001639 boron compounds Chemical class 0.000 description 1
- 229910052810 boron oxide Inorganic materials 0.000 description 1
- YHWCPXVTRSHPNY-UHFFFAOYSA-N butan-1-olate;titanium(4+) Chemical compound [Ti+4].CCCC[O-].CCCC[O-].CCCC[O-].CCCC[O-] YHWCPXVTRSHPNY-UHFFFAOYSA-N 0.000 description 1
- SXPLZNMUBFBFIA-UHFFFAOYSA-N butyl(trimethoxy)silane Chemical compound CCCC[Si](OC)(OC)OC SXPLZNMUBFBFIA-UHFFFAOYSA-N 0.000 description 1
- 229910052791 calcium Inorganic materials 0.000 description 1
- 239000004203 carnauba wax Substances 0.000 description 1
- 235000013869 carnauba wax Nutrition 0.000 description 1
- 229910000420 cerium oxide Inorganic materials 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- 239000006231 channel black Substances 0.000 description 1
- 229940090961 chromium dioxide Drugs 0.000 description 1
- 229910000423 chromium oxide Inorganic materials 0.000 description 1
- IAQWMWUKBQPOIY-UHFFFAOYSA-N chromium(4+);oxygen(2-) Chemical compound [O-2].[O-2].[Cr+4] IAQWMWUKBQPOIY-UHFFFAOYSA-N 0.000 description 1
- AYTAKQFHWFYBMA-UHFFFAOYSA-N chromium(IV) oxide Inorganic materials O=[Cr]=O AYTAKQFHWFYBMA-UHFFFAOYSA-N 0.000 description 1
- 239000010941 cobalt Substances 0.000 description 1
- 229910017052 cobalt Inorganic materials 0.000 description 1
- GUTLYIVDDKVIGB-UHFFFAOYSA-N cobalt atom Chemical compound [Co] GUTLYIVDDKVIGB-UHFFFAOYSA-N 0.000 description 1
- 238000006482 condensation reaction Methods 0.000 description 1
- 150000004696 coordination complex Chemical class 0.000 description 1
- 229910052802 copper Inorganic materials 0.000 description 1
- 239000010949 copper Substances 0.000 description 1
- XCJYREBRNVKWGJ-UHFFFAOYSA-N copper(II) phthalocyanine Chemical compound [Cu+2].C12=CC=CC=C2C(N=C2[N-]C(C3=CC=CC=C32)=N2)=NC1=NC([C]1C=CC=CC1=1)=NC=1N=C1[C]3C=CC=CC3=C2[N-]1 XCJYREBRNVKWGJ-UHFFFAOYSA-N 0.000 description 1
- 230000003247 decreasing effect Effects 0.000 description 1
- KQAHMVLQCSALSX-UHFFFAOYSA-N decyl(trimethoxy)silane Chemical compound CCCCCCCCCC[Si](OC)(OC)OC KQAHMVLQCSALSX-UHFFFAOYSA-N 0.000 description 1
- 230000007547 defect Effects 0.000 description 1
- 230000018044 dehydration Effects 0.000 description 1
- 238000006297 dehydration reaction Methods 0.000 description 1
- JKWMSGQKBLHBQQ-UHFFFAOYSA-N diboron trioxide Chemical compound O=BOB=O JKWMSGQKBLHBQQ-UHFFFAOYSA-N 0.000 description 1
- 230000000694 effects Effects 0.000 description 1
- 229910052731 fluorine Inorganic materials 0.000 description 1
- 239000011737 fluorine Substances 0.000 description 1
- 125000003709 fluoroalkyl group Chemical group 0.000 description 1
- 239000006232 furnace black Substances 0.000 description 1
- 229910052736 halogen Inorganic materials 0.000 description 1
- 150000002367 halogens Chemical class 0.000 description 1
- 229910001385 heavy metal Inorganic materials 0.000 description 1
- CZWLNMOIEMTDJY-UHFFFAOYSA-N hexyl(trimethoxy)silane Chemical compound CCCCCC[Si](OC)(OC)OC CZWLNMOIEMTDJY-UHFFFAOYSA-N 0.000 description 1
- 125000004435 hydrogen atom Chemical group [H]* 0.000 description 1
- 230000006872 improvement Effects 0.000 description 1
- 239000002440 industrial waste Substances 0.000 description 1
- 150000002484 inorganic compounds Chemical class 0.000 description 1
- 229910010272 inorganic material Inorganic materials 0.000 description 1
- 229910052809 inorganic oxide Inorganic materials 0.000 description 1
- MOUPNEIJQCETIW-UHFFFAOYSA-N lead chromate Chemical compound [Pb+2].[O-][Cr]([O-])(=O)=O MOUPNEIJQCETIW-UHFFFAOYSA-N 0.000 description 1
- 229910052744 lithium Inorganic materials 0.000 description 1
- 239000000696 magnetic material Substances 0.000 description 1
- 230000005415 magnetization Effects 0.000 description 1
- 229940002712 malachite green oxalate Drugs 0.000 description 1
- WPBNNNQJVZRUHP-UHFFFAOYSA-L manganese(2+);methyl n-[[2-(methoxycarbonylcarbamothioylamino)phenyl]carbamothioyl]carbamate;n-[2-(sulfidocarbothioylamino)ethyl]carbamodithioate Chemical compound [Mn+2].[S-]C(=S)NCCNC([S-])=S.COC(=O)NC(=S)NC1=CC=CC=C1NC(=S)NC(=O)OC WPBNNNQJVZRUHP-UHFFFAOYSA-L 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- 239000012968 metallocene catalyst Substances 0.000 description 1
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 description 1
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 1
- CXKWCBBOMKCUKX-UHFFFAOYSA-M methylene blue Chemical compound [Cl-].C1=CC(N(C)C)=CC2=[S+]C3=CC(N(C)C)=CC=C3N=C21 CXKWCBBOMKCUKX-UHFFFAOYSA-M 0.000 description 1
- BFXIKLCIZHOAAZ-UHFFFAOYSA-N methyltrimethoxysilane Chemical compound CO[Si](C)(OC)OC BFXIKLCIZHOAAZ-UHFFFAOYSA-N 0.000 description 1
- 239000000178 monomer Substances 0.000 description 1
- QGLKJKCYBOYXKC-UHFFFAOYSA-N nonaoxidotritungsten Chemical compound O=[W]1(=O)O[W](=O)(=O)O[W](=O)(=O)O1 QGLKJKCYBOYXKC-UHFFFAOYSA-N 0.000 description 1
- KSCKTBJJRVPGKM-UHFFFAOYSA-N octan-1-olate;titanium(4+) Chemical compound [Ti+4].CCCCCCCC[O-].CCCCCCCC[O-].CCCCCCCC[O-].CCCCCCCC[O-] KSCKTBJJRVPGKM-UHFFFAOYSA-N 0.000 description 1
- BMMGVYCKOGBVEV-UHFFFAOYSA-N oxo(oxoceriooxy)cerium Chemical compound [Ce]=O.O=[Ce]=O BMMGVYCKOGBVEV-UHFFFAOYSA-N 0.000 description 1
- VTRUBDSFZJNXHI-UHFFFAOYSA-N oxoantimony Chemical compound [Sb]=O VTRUBDSFZJNXHI-UHFFFAOYSA-N 0.000 description 1
- 125000004430 oxygen atom Chemical group O* 0.000 description 1
- 229920003216 poly(methylphenylsiloxane) Polymers 0.000 description 1
- 239000004848 polyfunctional curative Substances 0.000 description 1
- 229920000642 polymer Polymers 0.000 description 1
- 238000006116 polymerization reaction Methods 0.000 description 1
- 229920000098 polyolefin Polymers 0.000 description 1
- 229920001296 polysiloxane Polymers 0.000 description 1
- 238000001556 precipitation Methods 0.000 description 1
- 230000002035 prolonged effect Effects 0.000 description 1
- AABBHSMFGKYLKE-SNAWJCMRSA-N propan-2-yl (e)-but-2-enoate Chemical compound C\C=C\C(=O)OC(C)C AABBHSMFGKYLKE-SNAWJCMRSA-N 0.000 description 1
- QQONPFPTGQHPMA-UHFFFAOYSA-N propylene Natural products CC=C QQONPFPTGQHPMA-UHFFFAOYSA-N 0.000 description 1
- 125000004805 propylene group Chemical group [H]C([H])([H])C([H])([*:1])C([H])([H])[*:2] 0.000 description 1
- 150000003242 quaternary ammonium salts Chemical class 0.000 description 1
- 229940051201 quinoline yellow Drugs 0.000 description 1
- 235000012752 quinoline yellow Nutrition 0.000 description 1
- IZMJMCDDWKSTTK-UHFFFAOYSA-N quinoline yellow Chemical compound C1=CC=CC2=NC(C3C(C4=CC=CC=C4C3=O)=O)=CC=C21 IZMJMCDDWKSTTK-UHFFFAOYSA-N 0.000 description 1
- 239000004172 quinoline yellow Substances 0.000 description 1
- 239000002994 raw material Substances 0.000 description 1
- 238000010992 reflux Methods 0.000 description 1
- 229940081623 rose bengal Drugs 0.000 description 1
- 229930187593 rose bengal Natural products 0.000 description 1
- STRXNPAVPKGJQR-UHFFFAOYSA-N rose bengal A Natural products O1C(=O)C(C(=CC=C2Cl)Cl)=C2C21C1=CC(I)=C(O)C(I)=C1OC1=C(I)C(O)=C(I)C=C21 STRXNPAVPKGJQR-UHFFFAOYSA-N 0.000 description 1
- 229920006395 saturated elastomer Polymers 0.000 description 1
- HBMJWWWQQXIZIP-UHFFFAOYSA-N silicon carbide Chemical compound [Si+]#[C-] HBMJWWWQQXIZIP-UHFFFAOYSA-N 0.000 description 1
- 229910010271 silicon carbide Inorganic materials 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 125000006850 spacer group Chemical group 0.000 description 1
- 238000003860 storage Methods 0.000 description 1
- 229910052712 strontium Inorganic materials 0.000 description 1
- VEALVRVVWBQVSL-UHFFFAOYSA-N strontium titanate Chemical compound [Sr+2].[O-][Ti]([O-])=O VEALVRVVWBQVSL-UHFFFAOYSA-N 0.000 description 1
- 150000003440 styrenes Chemical class 0.000 description 1
- 229910052714 tellurium Inorganic materials 0.000 description 1
- PORWMNRCUJJQNO-UHFFFAOYSA-N tellurium atom Chemical compound [Te] PORWMNRCUJJQNO-UHFFFAOYSA-N 0.000 description 1
- BFKJFAAPBSQJPD-UHFFFAOYSA-N tetrafluoroethene Chemical group FC(F)=C(F)F BFKJFAAPBSQJPD-UHFFFAOYSA-N 0.000 description 1
- 239000006234 thermal black Substances 0.000 description 1
- XOLBLPGZBRYERU-UHFFFAOYSA-N tin dioxide Chemical compound O=[Sn]=O XOLBLPGZBRYERU-UHFFFAOYSA-N 0.000 description 1
- 229910001887 tin oxide Inorganic materials 0.000 description 1
- XYJRNCYWTVGEEG-UHFFFAOYSA-N trimethoxy(2-methylpropyl)silane Chemical compound CO[Si](OC)(OC)CC(C)C XYJRNCYWTVGEEG-UHFFFAOYSA-N 0.000 description 1
- NMEPHPOFYLLFTK-UHFFFAOYSA-N trimethoxy(octyl)silane Chemical compound CCCCCCCC[Si](OC)(OC)OC NMEPHPOFYLLFTK-UHFFFAOYSA-N 0.000 description 1
- ZNOCGWVLWPVKAO-UHFFFAOYSA-N trimethoxy(phenyl)silane Chemical compound CO[Si](OC)(OC)C1=CC=CC=C1 ZNOCGWVLWPVKAO-UHFFFAOYSA-N 0.000 description 1
- MWZATVIRTOMCCI-UHFFFAOYSA-N trimethoxy-(2-methylphenyl)silane Chemical compound CO[Si](OC)(OC)C1=CC=CC=C1C MWZATVIRTOMCCI-UHFFFAOYSA-N 0.000 description 1
- XQEGZYAXBCFSBS-UHFFFAOYSA-N trimethoxy-(4-methylphenyl)silane Chemical compound CO[Si](OC)(OC)C1=CC=C(C)C=C1 XQEGZYAXBCFSBS-UHFFFAOYSA-N 0.000 description 1
- MTPVUVINMAGMJL-UHFFFAOYSA-N trimethyl(1,1,2,2,2-pentafluoroethyl)silane Chemical compound C[Si](C)(C)C(F)(F)C(F)(F)F MTPVUVINMAGMJL-UHFFFAOYSA-N 0.000 description 1
- 229910001930 tungsten oxide Inorganic materials 0.000 description 1
- 235000013799 ultramarine blue Nutrition 0.000 description 1
- 230000000007 visual effect Effects 0.000 description 1
- XOSXWYQMOYSSKB-LDKJGXKFSA-L water blue Chemical compound CC1=CC(/C(\C(C=C2)=CC=C2NC(C=C2)=CC=C2S([O-])(=O)=O)=C(\C=C2)/C=C/C\2=N\C(C=C2)=CC=C2S([O-])(=O)=O)=CC(S(O)(=O)=O)=C1N.[Na+].[Na+] XOSXWYQMOYSSKB-LDKJGXKFSA-L 0.000 description 1
- 229910052725 zinc Inorganic materials 0.000 description 1
- 239000011701 zinc Substances 0.000 description 1
- 239000011787 zinc oxide Substances 0.000 description 1
Classifications
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03G—ELECTROGRAPHY; ELECTROPHOTOGRAPHY; MAGNETOGRAPHY
- G03G9/00—Developers
- G03G9/08—Developers with toner particles
- G03G9/12—Developers with toner particles in liquid developer mixtures
- G03G9/135—Developers with toner particles in liquid developer mixtures characterised by stabiliser or charge-controlling agents
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03G—ELECTROGRAPHY; ELECTROPHOTOGRAPHY; MAGNETOGRAPHY
- G03G9/00—Developers
- G03G9/08—Developers with toner particles
- G03G9/0802—Preparation methods
- G03G9/0804—Preparation methods whereby the components are brought together in a liquid dispersing medium
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03G—ELECTROGRAPHY; ELECTROPHOTOGRAPHY; MAGNETOGRAPHY
- G03G9/00—Developers
- G03G9/08—Developers with toner particles
- G03G9/12—Developers with toner particles in liquid developer mixtures
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03G—ELECTROGRAPHY; ELECTROPHOTOGRAPHY; MAGNETOGRAPHY
- G03G9/00—Developers
- G03G9/08—Developers with toner particles
- G03G9/12—Developers with toner particles in liquid developer mixtures
- G03G9/13—Developers with toner particles in liquid developer mixtures characterised by polymer components
- G03G9/131—Developers with toner particles in liquid developer mixtures characterised by polymer components obtained by reactions only involving carbon-to-carbon unsaturated bonds
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03G—ELECTROGRAPHY; ELECTROPHOTOGRAPHY; MAGNETOGRAPHY
- G03G9/00—Developers
- G03G9/08—Developers with toner particles
- G03G9/12—Developers with toner particles in liquid developer mixtures
- G03G9/13—Developers with toner particles in liquid developer mixtures characterised by polymer components
- G03G9/132—Developers with toner particles in liquid developer mixtures characterised by polymer components obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds
Definitions
- This invention relates to a toner for developing an electrostatic image, particularly relates to a toner comprising particles of low molecular weight polypropylene.
- an electrostatic image is formed on a photoreceptor by giving uniformly charge and imagewise exposure thereto.
- the electrostatic image is developed by a developer containing a toner to form a toner image, and the toner image is transferred to a paper and fixed to form a visual image.
- a cleaning member arranged on the surface of photoreceptor so as to be contacted with pressure to the surface removes the toner which is not transferred to the paper and remained on the photoreceptor.
- the fixing image is usually formed by the use of a heat-roller fixing device.
- a technique has been known to prevent the offset problem, in which a particle of low molecular weight polypropylene is contained in the colored particle for giving a mold-releasing ability of the toner.
- the low molecular weight polypropylene particle has a high mole-releasing ability itself and is able to be sharply fused when the particle is heated. Therefore, when the toner comprising such the colored particle is used, the low molecular weight polypropylene particle is rapidly fused at the fixing treatment by the heat-roller and the surface of the toner particle is covered by the fused low molecular weight polypropylene. As a result of that, the interface energy between the toner particle and the surface of heat-roller is lowered and the mold releasing ability of the toner is made higher.
- low molecular weight polypropylene particles can not be included in the colored particles since the miscibility of the low molecular weight polypropylene particle with the binder resin of the colored particle is not so high. Accordingly, low molecular weight polypropylene particles freely existing outside the colored particles, hereinafter referred to free polypropylene particle, are formed.
- the object of the invention is to provide a toner by which the offset problem and the toner film formation on the photoreceptor can be inhibited while maintaining a high transferring ability in the image forming process.
- the toner of the invention comprises a colored particle containing at least a binder resin, a colorant, and a low molecular weight polypropylene particle, which has a content of the low molecular weight polypropylene particle of from 0.3 to 8.0 percent by weight based on the toner, a free polypropylene index measured by a centrifugal method of from 0.1 to 0.7 and a free polypropylene index measured by a natural sedimentation method of not more than 0.1.
- the shape of the polypropylene particle separated by the centrifugal method is substantially ellipsoid. It is also preferable a number ratio of ellipsoid polypropylene particles each having an elliptical axis ratio of projection image of not more than 0.8 to the whole number of the separated polypropylene particles is not less than 50 percent.
- the ratio of the number of the particles each having a particle diameter of not more than 4 ⁇ m present in the polypropylene particles separated by the centrifugal method to the whole number of the polypropylene particles separated by the centrifugal method is not less than 50%, more preferably not less than 60%.
- the toner of the invention comprises colored particles and an external additive added according to necessity, and the colored particle comprises a binder resin, a colorant and a mold releasing agent, or low molecular weight polypropylene particle, in which an internal additive may be added according to necessity.
- a binder resin usually used in toner such as a styrene resin, an acryl resin, a styrene/acryl resin and a polyester resin, is usable for the binder resin of the toner of the invention.
- a colorant usually used in toner such as carbon black, a magnetic material, a dye and a pigment, is usable for the colorant of the toner.
- Examples of the carbon black usable as the colorant include channel black, furnace black, acetylene black, thermal black and lump black.
- the magnetic substance examples include a ferromagnetic metal such as iron, nickel and cobalt, an alloy containing such the metal, a compound of a ferromagnetic metal such as ferrite and magnetite, a ferromagnetic alloy by a heat treatment even though containing no ferromagnetic metal such as a kind of alloy called Heuslar alloy such as manganese-copper-aluminum and manganese-copper-tin, and chromium dioxide.
- a ferromagnetic metal such as iron, nickel and cobalt
- an alloy containing such the metal such as iron, nickel and cobalt
- a compound of a ferromagnetic metal such as ferrite and magnetite
- a ferromagnetic alloy by a heat treatment even though containing no ferromagnetic metal such as a kind of alloy called Heuslar alloy such as manganese-copper-aluminum and manganese-copper-tin
- chromium dioxide examples include a
- C.I. Solvent Red series of 1, 49, 52, 58, 63, 111, and 122 C.I. Solvent Yellow series of 19, 44, 77, 79, 81, 82, 93, 98, 103, 104, 112 and 162, and C.I. Solvent Blue series of 25, 36, 60, 70, 93 and 95, are usable. A mixture of them may also be used.
- Examples of the pigment include C.I. Pigment Red series of 5, 48:1, 53:1, 57:1, 122, 139, 144, 149, 166, 177, 178 and 222, C.I. Pigment Orange series of 31 and 43, C.I. Pigment Yellow series of 14, 17, 93, 94 and 138, C.I. Pigment Green 7, C.I. Pigment Blue series of 15:3 and 60. These pigments may be used singly or in combination.
- Examples of preferred colorant include carbon black, Nigrosine dye, aniline blue, chalcoil blue, chrome yellow, ultramarine blue, du Pont oil red, quinoline yellow, methylene blue chloride, phthalocyanine blue, malachite green oxalate and rose bengal.
- a low molecular weight polypropylene particle is used as the mold-releasing agent for the toner.
- a low molecular weight polypropylene particle synthesized by using a metallocene catalyst may be used as such the low molecular weight polypropylene particle.
- the molecular weight of the low molecular weight polypropylene particle is preferably from 1,500 to 10,000, particularly from 1,800 to 4,000, in number average molecular weight Mn.
- the number average molecular weight Mn is a number average molecular weight Mn measured by a high temperature GPC method.
- the number average molecular weight is determined by a procedure in which a sample is flowed out at 135° C. using o-dichlorobenzene containing 0.1% of ionol and the refractive index thereof is measured by a difference refractive index meter and converted by polypropylene absolute molecular weight conversion according to the universal compensation method.
- Heat energy necessary for fusing the toner can be lowered when the number average molecular weight Mn of the low molecular weight polypropylene particle is within the range of from 1,500 to 10,000, with the result that the toner fixable at a relative low temperature can be prepared.
- the toner containing the low molecular weight polypropylene particle is excellent in the storage ability and fixing ability since of the low molecular weight polypropylene particle has a narrow molecular weight distribution.
- the ratio of the weight average molecular weight Mw to the number average molecular weight Mn of the low molecular weight polypropylene particle Mw/Mn is preferably from 1.5 to 3, particularly from 2 to 3. When the ratio is within the range of from 1.5 to 3, the viscosity of the fused polymer is sufficiently lowered and the problem is effectively inhibited.
- the content of the low molecular weight polypropylene particles in the toner of the invention is within the range of from 0.3 to 8.0% by weight, preferably 1.3 to 5.7%, more preferably 1.9 to 3.8% by weight of the toner.
- Examples of internal additive to be added in the toner include a negative charge controlling agent such as an azo metal complex, a salicylic metal complex and a calixarene compound, a positive charge controlling agent such as a Nigrosine dye and a quaternary ammonium salt, a fixing ability improving agent such as a low molecular weight polyolefin and Carnauba wax, and a magnetic particles to be used in a magnetic toner.
- Examples of magnetic particle include a particle of ferrite and magnetite each having an average diameter of primary particles of from 0.1 to 2.0 ⁇ m. The content of the magnetic particles is from 20 to 70% by weight of the colored particle.
- an inorganic and organic fine particle may be used, and the inorganic fine particle is preferably used for giving a flowing ability to the colored particle.
- inorganic oxide compounds such as nitride compounds and boron compounds may be use for forming the inorganic fine particle.
- the inorganic compound include silica, alumina, titania, zirconia, barium titanate aluminum titanate, strontium titanate, magnesium titanate, zinc oxide, chromium oxide, cerium oxide, antimony oxide, tungsten oxide, tin oxide, tellurium oxide, manganese oxide, boron oxide, silicon carbide, titanium carbide, boron carbide, silica nitride, titanium nitride and boron nitride.
- the number average primary particle diameter is preferably from 5 to 200 nm, which is determined by analysis of the image observed by a transparency-type electronic microscope.
- the surface of such the inorganic fine particle is preferably subjected to a treatment to give hydrophobic property by a coupling agent such as a titanium coupling agent and silane coupling agent, or silicone oil.
- a coupling agent such as a titanium coupling agent and silane coupling agent, or silicone oil.
- titanium coupling agent tetrabutyl titanate, tetraoctyl titanate, isopropyltriisostearoyl titanate, isopropyltridecylbenzenesulfonyl titanate, and bis(dioctyl pyrophosphate) oxyacetate titanate are usable.
- silane coupling agent the followings are usable; ⁇ -(2-aminoethyl) aminopropyltrimethoxysilane, ⁇ -(2-aminoethyl) aminopropyldimethoxysilane, ⁇ -methacryloxypropyltrimethoxysilane, N- ⁇ -(N-vinylbenzylaminoethyl) ⁇ -aminopropyltrimethoxysilane hydrochloride, hexamethyldisilazane, methyltrimethoxysilane, butyltrimethoxysilane, isobutyltrimethoxysilane, hexyltrimethoxysilane, octyltrimethoxysilane, decyltrimethoxysilane, dodecytrimethoxysilane, phenyltrimethoxysilane, o-methylphenyltrimethoxysilane and p-methylphenyl
- silicone oil dimethylsilicone oil, methylphenylsilicone oil and amino-modified silicone oil are usable.
- the particle diameter of the colored particle of the toner of the invention is preferably within the range of from 2 to 10 ⁇ m, more preferably 3.0 to 9.0 ⁇ m, in a volume average diameter measured by Coulter Counter TA-11 or Coulter Multisizer.
- the diameter of the colored particle can be controlled by production conditions thereof.
- the colored particles contain the polypropylene having a diameter of 0.1 to 0.8 ⁇ m, more preferably 0.3 to 0.8 ⁇ m.
- the free polypropylene index measured by the centrifugal separation method is within the range of from 0.1 to 0.7, preferably 0.1 to 0.5, more preferably 0.1 to 0.4, and the free polypropylene index measured by the natural sedimentation method is not more than 0.1, preferably not more than 0.05.
- free polypropylene index measured by the centrifugal separation method is a light absorbency or turbidity of the top clean liquid obtained by centrifugal separation of a suspension of the toner.
- the light absorbency at 500 nm of the top clean liquid obtained by the following procedures (I) to (iv).
- a suitable amount of water is put in a measuring flask of 100 ml and 1 ml of a 12% solution of sodium dodecylbenzenesulfonate is added, then water is gently added to make the volume of the solution to 100 ml to prepare a surfactant solution.
- the suspension of toner is put into a 50 ml separation tube and the tube set in an angle rotor having a rotating radius of 70 mm. Then the suspension is subjected to the centrifugal separation for 20 minutes at 5000 rpm.
- the low molecular weight polypropylene particles adhered on the internal wall of the centrifuge tube are washed off from the wall by the top clean liquid using a pipette and the top clean liquid is sampled. If the precipitated toner particles are mixed in the top clean liquid, the liquid is stood for one day in the tube or subjected to the centrifugal separation once more.
- free polypropylene index measured by the natural sedimentation method is a light absorbency or turbidity of the top clean liquid obtained by natural sedimentation of a suspension of the toner.
- a suitable amount of water is put in a measuring flask of 100 ml and 1 ml of a 12% solution of sodium dodecylbenzenesulfonate is added, then water is gently added to make the volume of the solution to 100 ml to prepare a surfactant solution.
- the toner suspension is stood for one day and the top clean liquid is sampled by a pipette.
- the free polypropylene index measured by centrifugal separation method is not less than 0.1, a suitable amount of free polypropylene particles is fixed on the surface of the toner particle with a suitable adhesive force with the result that a high transferring ability of the toner can be obtained.
- the charging ability of the toner can be stabilized with the result that the formation of toner film on the photoreceptor can be inhibited.
- the shape of the free polypropylene particle separated by the centrifugal method is substantially ellipsoid and that the number of the free polypropylene particles having a elliptical ratio (shorter axis/longer axis) of the projection image of the ellipsoid of not more than 0.8 accounts not less than 50% of the number of the whole free polypropylene particles.
- the elliptical ratio of the free polypropylene particles separated by the centrifugal method can be determined by the following procedures: a white turbid part floating at the surface the top clean liquid is sampled after the centrifugal separation according to the above (5-1) (iii); the sampled portion is filtered to obtained a white powder; the white powder is subjected to IR measurement to confirm that the powder is polypropylene; the microscopic image of the powder is taken by a scanning electron micrometer T-330, (manufactured by Nihon Denshi Co., Ltd., acceleration potential: 20 kV, Spot size: 3 p.m. o'clock-set, in gamma intensifying mode); and the images of 2000 particles are subjected to image analyzing by an image analyzer SIA, manufactured by Nihon Denshi Co., Ltd.
- the charging ability of the toner can be stabilized with the result that the formation of toner film on the photoreceptor can be inhibited when the toner is used in the image forming process.
- the ratio of the free polypropylene particles separated by the centrifugal method having diameter of not more than 4 ⁇ m is not less than 50%, more preferably not less than 60%, in number of the whole polypropylene particles by the centrifugal method.
- the volume average diameter of the free polypropylene particles can be measured by subjecting the free polypropylene particles in the top clean liquid obtained by the procedure of the above (5-1) (iv) to measure by Coulter Multisizer manufactured by Coulter Co., Ltd.
- the transferring ability of the toner can be stabilized when the free polypropylene particle each having a diameter of not more than 4 ⁇ m are existed in a ratio of not less than 50% in number.
- the toner of the invention can be prepared by either a crushing method or a polymerization method.
- the crushing method is preferred.
- a raw composition of the toner containing raw materials such as the binder resin, the coloring agent, the mold releasing agent, or the low molecular weight polypropylene particles, and the charge controlling agent is molten, kneaded, crushed and classified to prepare the colored particles.
- the crushing and classifying processes may be repeated when it is necessary.
- the external additive is added to the colored particles to prepare the toner.
- the diameter of the low molecular weight polypropylene particle is preferably within the range of from 0.1 to 100 ⁇ m, more preferably from 0.3 to 6 ⁇ m.
- the shape of the low molecular weight polypropylene particles is preferably ellipsoid or spindle-shape.
- the low molecular weight polypropylene particles each having the above-mentioned diameter and shape can be produced by controlling the diameter and the shape by a solution sedimentation process and a heat treatment, respectively.
- the solution sedimentation process is a method in which the low molecular weight polypropylene particles are dissolved in toluene under reflux and the solution is cooled to 50° C. Then the solution is poured into cold acetone.
- the low molecular weight polypropylene particles each having a diameter within a specified range.
- the shape of the low molecular weight polypropylene particle can be made such as the spindle-shape by putting the particle in heated air blow.
- the temperature of the air blow is preferably from 100 to 140 C.
- the heat treatment may be carried out in a liquid.
- the free propylene index by centrifugal method is within the range of from 0.1 to 0.7 and the free polypropylene index by natural sedimentation method is not more than 0.1 in the toner of the invention, with the result that the occurrence of toner-filming can be inhibited while maintaining a sufficiently high transferring ability when the toner is used in the image forming process.
- the occurrence of toner-filming can be inhibited when the shape of the free polypropylene particle the centrifugal method is substantially ellipsoid and the ratio of the particles each having a elliptical ratio of the ellipsoid of not more than 0.8 account not less than 50% in number of the whole free polypropylene particles.
- a sufficiently high transferring ability in the image forming process can be obtained when the ratio of the number of the free polypropylene particles the centrifugal method having a diameter on not more than 4 ⁇ m to the whole number of free polypropylene particles is not less than 50%.
- the toner according to the invention can be used in any embodiment such as (1) singly use as a magnetic toner containing a magnetic substance, (2) singly use as a non-magnetic toner containing no magnetic substance and (3) use as a two-component developer together with a carrier. Among them the embodiment (3) is preferred.
- both of (1) a carrier composed of only a magnetic substance particle such as iron and ferrite, and (2) a resin coated carrier composed of a core particle covered with a resin may be used.
- the above-mentioned resin coated carrier (2) is preferred from the viewpoint of durability thereof.
- the volume average particle diameter of the carrier is preferably from 10 to 100 ⁇ m, more preferably 20 to 80 ⁇ m.
- the magnetic property of the carrier is preferably from 30 to 80 emu/g in the saturated magnetization.
- Iron powder, magnetite and various types of ferrite are usable for the core particle of the resin coated carrier. Among them, magnetite and ferrite are preferred.
- the light metal-containing ferrite is particularly preferred.
- the light metal-containing ferrite has the composition showing the following formula (1) or (2).
- M is an alkyl or an alkali-earth metal.
- Ones may be used, in which a part of M 2 O and/or Fe 2 O 3 is replaced by an alkali-earth metal.
- x is not more than 30 mole-%, preferably not more than 18 mole-%, and the replacing amount of alkali-earth metal and/or alkali metal is preferably from 1 to 15 mole-%, more preferably from 3 to 15 mole-%.
- the light metal-containing ferrite and magnetite are preferred since not only problems of pollution caused by the industrial waste can be reduced but the weight of the carrier can be reduced with result of that the stress on the toner can be decreased.
- the resin of the resin coated carrier is not specifically limited, and a resin such as a silicone resin, styrene-acryl resin, a vinylidene fluoride and a fluoride resin other than vinylidene fluoride resin are usable.
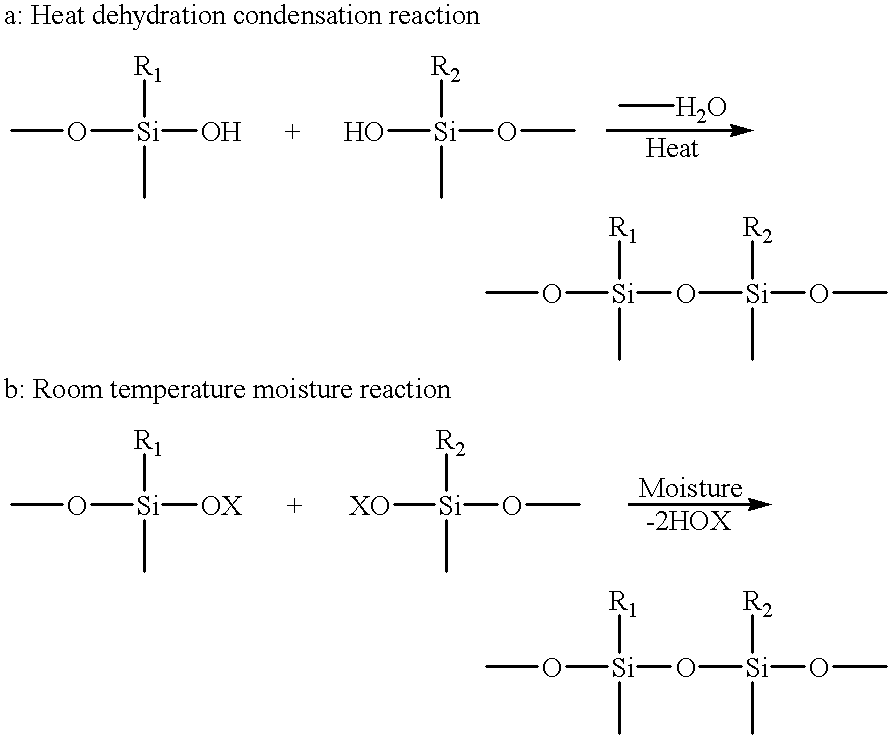
- a condensation reactive type silicone resin is preferably used, which is hardened by a heat dehydration condensation reaction or a room temperature moisture hardening reaction.
- a condensation reactive type silicone resin capable of hardening by the following reaction a or b is particularly preferable.
- R 1 and R 2 are each a substituent such as an alkyl group, and OX is an alkoxy group or an acyloxy group.
- silicone resin preferably usable in the invention examples include SR-2411 and SR-2710, manufactured by Toray Silicone co., Ltd., and KR-255 and KR-271, manufactured by Shin'etsu Kagaku Co., Ltd.
- styrene-acryl resin examples include copolymers of styrene or a styrene derivative such as styrene, o-methylstyrene, m-methylstyrene, p-methylstyrene, ⁇ -methylstyrene, p-chlorostyrene, 3,4-dichlorostyrene, p-phenylstyrene, p-ethylstyrene, 2,4-dimethylstyrene, p-t-butylstyrene, p-n-hexylstyrene, p-n-octylstyrene, p-n-nonylstyrene, p-n-decylstyrene and p-n-dodecylstyrene with a methacrylic acid ester derivative such as methyl methacrylate, ethacryl
- vinylidene fluoride resin a copolymer of vinylidene fluoride and tetrafluoroethylene, hexafluoropropylene, monochlorotrifluoroethylene, monofluoroethylene or trifluoroethylene and another copolymer component is preferred, and ones containing vinylidene fluoride in a ratio of not less than 20% by weight is particularly preferred.
- the copolymer component of the vinylidene fluoride other than the halogen-containing monomer includes copolymers of stylene and its derivative such as styrene, o-methylstyrene, m-methylstyrene, p-methylstyrene, ⁇ -methylstyrene, p-chlorostyrene, 3,4-dichlorostyrene, p-phenylstyrene, p-ethylstyrene, 2,4-dimethylstyrene, p-t-butylstyrene, p-n-butylstyrene, p-n-hexylstyrene, p-n-octylstyrene, p-n-nonylstyrene, p-n-decylstyrene and p-n-dodecylstyrene, with a me
- the fluorine-containing resin other than vinylidene fluoride includes a resin derived from a compound represented by the following Formula (3).
- R is a hydrogen atom or a methyl group
- X is an oxygen atom, a —COO group or a ⁇ CO group
- Rf is a fluoroalkyl group
- the following method can be applied for coating the resin on the core particle; (1) a solution of the resin is sprayed to the core particle and dried, (2) particles of the coating resin is electrostatically adhered to the core particle and mechanical energy is applied thereon to form a coating layer, (3) particles of the coating resin is electrostatically adhered to the core particle and the core particle is heated at a temperature higher than the melting point of the resin to form a coating resin layer, (4) the core particle is immersed in a solution of the resin, and (5) the resin containing a hardener is coated on the core particle and heated to harden the resin layer.
- the amount of the coated resin may be an amount sufficient for uniformly covering the core particle surface, and concretely from 0.1 to 5.0%, preferably from 0.5 to 3.0%, by weight of the core particles.
- the effect of the resin coating cannot be obtained when the coated amount is too low, and an image defect tends to be caused by the resin peeled from the core surface when the coated amount of resin is excessive.
- the toner remained on the photoreceptor is recovered by a cleaning device and the recovered toner is returned to a developing device or a toner supplying device to reuse the toner.
- a raw composition of carrier composed of 1000 parts by weight of ferrite particles, as the core particles, having a volume average diameter of 70 ⁇ m and 20 parts by weight of 1,1,1-trifluoroethyl methacrylate powder, as the resin powder, having a volume average diameter of 0.1 ⁇ m was put into a horizontal rotation impeller mixer. Then the mixture was heated to 70° C. while stirring with an impeller circumference speed of 4 m/sec. The mixture was further stirred for 20 minutes to prepare a carrier.
- a raw composition of toner composed of 1000 parts by weight of polyester resin, as the binder resin, 10 parts by weight of carbon black as the colorant, and 3 parts by weight of low molecular weight polypropylene as the mold releasing agent was preliminary mixed by a Henschel mixer for 5 minutes. The mixing conditions were adjusted so that the bulk density and the particle diameter distribution of the toner particles were made as shown in Table 1 and Table 2, respectively.
- the low molecular weight polypropylene had a shape of ellipsoid having an elliptical ratio of projection image of 0.66, a particle diameter of 0.7 ⁇ m, Mw of 6600, Mn of 3000, Mw/Mn of 2.20 and a melting point of 80° C.
- the mixture was molten and kneaded by a biaxial kneading extruder set at 170° C. Then the mixture was roughly crushed by a hammer mill and finely crushed by a jet crusher, and classified by a wind classification machine to prepare colored particles (A) having a volume average diameter of 10.0 ⁇ m were obtained.
- Toner (A) according to the invention was obtained, which has the properties shown in Table 3.
- the low molecular weight polypropylene had a shape of ellipsoid having an elliptical ratio of projection image of 0.31, a particle diameter of 0.8 ⁇ m, Mw of 13700, Mn of 7200, Mw/Mn of 1.90 and a melting point of 133° C.
- the mixture was molten and kneaded by a biaxial kneading extruder set at 170° C. Then the mixture was roughly crushed by a hammer mill and finely crushed by a jet crusher, and classified by as wind classification machine to prepare colored particles (B) having a volume average diameter of 9.0 ⁇ m were obtained.
- Toner (B) according to the invention was obtained, which has the properties shown in Table 3.
- the low molecular weight polypropylene had a shape of ellipsoid having an elliptical ratio of projection image of 0.55, a particle diameter of 1.0 pim, Mw of 13700, Mn of 7200, Mw/Mn of 1.90 and a melting point of 133° C.
- the mixture was molten and kneaded by a biaxial kneading extruder set at 170° C. Then the mixture was roughly crushed by a hammer mill and finely crushed by a jet crusher, and classified by as wind classification machine to prepare colored particles (C) having a volume average diameter of 8.0 ⁇ m were obtained.
- Toner (C) according to the invention was obtained, which has the properties shown in Table 3.
- the low molecular weight polypropylene had a sphere shape, a particle diameter of 100 pm, Mw of 8600, Mn of 2270, Mw/Mn of 3.79 and a melting point of 135° C.
- the mixture was molten and kneaded by a biaxial kneading extruder set at 170° C. Then the mixture was roughly crushed by a hammer mill and finely crushed by a jet crusher, and classified by as wind classification machine to prepare comparative colored particles (a) having a volume average diameter of 10.0 ⁇ m were obtained.
- Toner (a) according to the invention was obtained, which has the properties shown in Table 3.
- Comparative colored particles (b) and (c) were prepared in the same manner as in comparative toner (a) except that the mixing conditions were changed so that the bulk density shown in Table 1 and the particle diameter distribution shown in Table 2 were obtained.
- the comparative colored particles (b) and (c) each had a volume average diameter of 10.0 ⁇ m. Then comparative toners (b) and (c) having the properties shown in Table 3 were prepared using the comparative colored particles (b) and (c), respectively.
- Toners for evaluation were prepared by mixing the toner of the invention (A), (B) or (C), or the comparative toner (a), (b) or (c) was mixed with the carrier so that the ratio of the toner to the carrier was made to 5% by weight.
- the number of copied sheet was counted until a band-like contamination in the transporting direction of paper was occurred.
- the number of copied sheet was counted until 10 black spots contamination having a diameter 0.8 mm or more were formed on the white background area of the copy.
- the reflective density of the white background area of the copy sheet was measured by Sakura Densitometer, manufactured by Konica Corp., and the number of copied sheet was counted until the relative reflective density was exceed 0.01.
- the relative reflective density was determined based on the density of the paper it self of 0.
- a solid image having a density of 1.3 and the area of 20 mm ⁇ 50 mm was prepared, and the transferring ratio was calculated by the following equation.
- the off-set problem is prevented and formation of black spot can be inhibited by the toner according to the invention when the toner is used in the image forming process. Furthermore, formation of black spot contamination on the image can be prevented and the life of developer can be prolonged by the use of the toner of the invention.
Landscapes
- Physics & Mathematics (AREA)
- General Physics & Mathematics (AREA)
- Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Developing Agents For Electrophotography (AREA)
Abstract
A toner for developing an electrostatic image is disclosed. The toner comprises a colored particle containing a binder resin, a colorant, and a low molecular weight polypropylene particle, which has a content of the low molecular weight polypropylene particle of 0.3 to 8.0 percent by weight, a free polypropylene index measured by a centrifugal method of from 0.1 to 0.7 and a free polypropylene index measured by a natural sedimentation method of not more than 0.1.
Description
This invention relates to a toner for developing an electrostatic image, particularly relates to a toner comprising particles of low molecular weight polypropylene.
In an example of image formation process in the electrophotographic method, an electrostatic image is formed on a photoreceptor by giving uniformly charge and imagewise exposure thereto. The electrostatic image is developed by a developer containing a toner to form a toner image, and the toner image is transferred to a paper and fixed to form a visual image. On the other hand, a cleaning member arranged on the surface of photoreceptor so as to be contacted with pressure to the surface removes the toner which is not transferred to the paper and remained on the photoreceptor.
In the fixing process of such the image forming procedure, the fixing image is usually formed by the use of a heat-roller fixing device.
There is a drawback in the fixing by the heat-roller device that an offset problem tends to raise. The offset problem is caused by transferring a part of the molten toner to the surface of the heat-roller, the transferred toner is transferred back to the next paper so as to contaminate the image.
A technique has been known to prevent the offset problem, in which a particle of low molecular weight polypropylene is contained in the colored particle for giving a mold-releasing ability of the toner.
The low molecular weight polypropylene particle has a high mole-releasing ability itself and is able to be sharply fused when the particle is heated. Therefore, when the toner comprising such the colored particle is used, the low molecular weight polypropylene particle is rapidly fused at the fixing treatment by the heat-roller and the surface of the toner particle is covered by the fused low molecular weight polypropylene. As a result of that, the interface energy between the toner particle and the surface of heat-roller is lowered and the mold releasing ability of the toner is made higher.
However, a part of the low molecular weight polypropylene particles can not be included in the colored particles since the miscibility of the low molecular weight polypropylene particle with the binder resin of the colored particle is not so high. Accordingly, low molecular weight polypropylene particles freely existing outside the colored particles, hereinafter referred to free polypropylene particle, are formed.
When the toner containing the free polypropylene particles is used in the image forming process, problems are raised such as that the charging ability of the toner is made unstable and toner filming on the photoreceptor tends to be formed.
Moreover, a problem that a black spot like contamination is formed on the image when the diameter of the free polypropylene particle is large.
Furthermore, a problem is raised that the presence of the free polypropylene particles causes lowering in the flowing ability of the toner and the life of the developer is shortened.
On the other hand, when a toner with no free polypropylene particle or a wax-free toner is used in the image forming process, a sufficient transferring ability cannot be obtained since the particles are not present which fill the role of spacer for lowering the adhesive force between the toner particle and the photoreceptor.
The object of the invention is to provide a toner by which the offset problem and the toner film formation on the photoreceptor can be inhibited while maintaining a high transferring ability in the image forming process.
The toner of the invention comprises a colored particle containing at least a binder resin, a colorant, and a low molecular weight polypropylene particle, which has a content of the low molecular weight polypropylene particle of from 0.3 to 8.0 percent by weight based on the toner, a free polypropylene index measured by a centrifugal method of from 0.1 to 0.7 and a free polypropylene index measured by a natural sedimentation method of not more than 0.1.
It is preferable in the toner that the shape of the polypropylene particle separated by the centrifugal method is substantially ellipsoid. It is also preferable a number ratio of ellipsoid polypropylene particles each having an elliptical axis ratio of projection image of not more than 0.8 to the whole number of the separated polypropylene particles is not less than 50 percent.
It is preferable in the toner that the ratio of the number of the particles each having a particle diameter of not more than 4 μm present in the polypropylene particles separated by the centrifugal method to the whole number of the polypropylene particles separated by the centrifugal method is not less than 50%, more preferably not less than 60%.
The invention is described in detail bellow.
Toner
The toner of the invention comprises colored particles and an external additive added according to necessity, and the colored particle comprises a binder resin, a colorant and a mold releasing agent, or low molecular weight polypropylene particle, in which an internal additive may be added according to necessity.
(1) Binder resin
A binder resin usually used in toner, such as a styrene resin, an acryl resin, a styrene/acryl resin and a polyester resin, is usable for the binder resin of the toner of the invention.
(2) Colorant
A colorant usually used in toner, such as carbon black, a magnetic material, a dye and a pigment, is usable for the colorant of the toner.
Examples of the carbon black usable as the colorant include channel black, furnace black, acetylene black, thermal black and lump black.
Examples of the magnetic substance include a ferromagnetic metal such as iron, nickel and cobalt, an alloy containing such the metal, a compound of a ferromagnetic metal such as ferrite and magnetite, a ferromagnetic alloy by a heat treatment even though containing no ferromagnetic metal such as a kind of alloy called Heuslar alloy such as manganese-copper-aluminum and manganese-copper-tin, and chromium dioxide.
As the dye, for example, C.I. Solvent Red series of 1, 49, 52, 58, 63, 111, and 122, C.I. Solvent Yellow series of 19, 44, 77, 79, 81, 82, 93, 98, 103, 104, 112 and 162, and C.I. Solvent Blue series of 25, 36, 60, 70, 93 and 95, are usable. A mixture of them may also be used.
Examples of the pigment include C.I. Pigment Red series of 5, 48:1, 53:1, 57:1, 122, 139, 144, 149, 166, 177, 178 and 222, C.I. Pigment Orange series of 31 and 43, C.I. Pigment Yellow series of 14, 17, 93, 94 and 138, C.I. Pigment Green 7, C.I. Pigment Blue series of 15:3 and 60. These pigments may be used singly or in combination.
Examples of preferred colorant include carbon black, Nigrosine dye, aniline blue, chalcoil blue, chrome yellow, ultramarine blue, du Pont oil red, quinoline yellow, methylene blue chloride, phthalocyanine blue, malachite green oxalate and rose bengal.
(3) Mold releasing agent
A low molecular weight polypropylene particle is used as the mold-releasing agent for the toner. A low molecular weight polypropylene particle synthesized by using a metallocene catalyst may be used as such the low molecular weight polypropylene particle.
The molecular weight of the low molecular weight polypropylene particle is preferably from 1,500 to 10,000, particularly from 1,800 to 4,000, in number average molecular weight Mn.
The number average molecular weight Mn is a number average molecular weight Mn measured by a high temperature GPC method. In concrete, the number average molecular weight is determined by a procedure in which a sample is flowed out at 135° C. using o-dichlorobenzene containing 0.1% of ionol and the refractive index thereof is measured by a difference refractive index meter and converted by polypropylene absolute molecular weight conversion according to the universal compensation method.
Heat energy necessary for fusing the toner can be lowered when the number average molecular weight Mn of the low molecular weight polypropylene particle is within the range of from 1,500 to 10,000, with the result that the toner fixable at a relative low temperature can be prepared.
The toner containing the low molecular weight polypropylene particle is excellent in the storage ability and fixing ability since of the low molecular weight polypropylene particle has a narrow molecular weight distribution.
The ratio of the weight average molecular weight Mw to the number average molecular weight Mn of the low molecular weight polypropylene particle Mw/Mn is preferably from 1.5 to 3, particularly from 2 to 3. When the ratio is within the range of from 1.5 to 3, the viscosity of the fused polymer is sufficiently lowered and the problem is effectively inhibited.
The content of the low molecular weight polypropylene particles in the toner of the invention is within the range of from 0.3 to 8.0% by weight, preferably 1.3 to 5.7%, more preferably 1.9 to 3.8% by weight of the toner.
When the content of low molecular weight polypropylene particles is less than 0.3% by weight, the mold releasing ability of the toner is become insufficient and the offset tends to occur when the toner is used in the image forming process.
On the other hand, when the content of the low molecular weight polypropylene particles exceeds 8.0% by weight, a large number of free low molecular weight polypropylene particles are formed and the toner film tends to be formed on the surface of the photoreceptor when the toner is used in the image forming process.
(4) Additive
(a) Internal additive:
Examples of internal additive to be added in the toner include a negative charge controlling agent such as an azo metal complex, a salicylic metal complex and a calixarene compound, a positive charge controlling agent such as a Nigrosine dye and a quaternary ammonium salt, a fixing ability improving agent such as a low molecular weight polyolefin and Carnauba wax, and a magnetic particles to be used in a magnetic toner. Examples of magnetic particle include a particle of ferrite and magnetite each having an average diameter of primary particles of from 0.1 to 2.0 μm. The content of the magnetic particles is from 20 to 70% by weight of the colored particle.
(b) External additive:
As an external additive, or an optional component of the colored particle, contained in the toner of the invention, an inorganic and organic fine particle may be used, and the inorganic fine particle is preferably used for giving a flowing ability to the colored particle.
Various inorganic oxide compounds such as nitride compounds and boron compounds may be use for forming the inorganic fine particle. Concrete examples of the inorganic compound include silica, alumina, titania, zirconia, barium titanate aluminum titanate, strontium titanate, magnesium titanate, zinc oxide, chromium oxide, cerium oxide, antimony oxide, tungsten oxide, tin oxide, tellurium oxide, manganese oxide, boron oxide, silicon carbide, titanium carbide, boron carbide, silica nitride, titanium nitride and boron nitride.
The number average primary particle diameter is preferably from 5 to 200 nm, which is determined by analysis of the image observed by a transparency-type electronic microscope.
The surface of such the inorganic fine particle is preferably subjected to a treatment to give hydrophobic property by a coupling agent such as a titanium coupling agent and silane coupling agent, or silicone oil.
As the titanium coupling agent, tetrabutyl titanate, tetraoctyl titanate, isopropyltriisostearoyl titanate, isopropyltridecylbenzenesulfonyl titanate, and bis(dioctyl pyrophosphate) oxyacetate titanate are usable.
As the silane coupling agent, the followings are usable; γ-(2-aminoethyl) aminopropyltrimethoxysilane, γ-(2-aminoethyl) aminopropyldimethoxysilane, γ-methacryloxypropyltrimethoxysilane, N-β-(N-vinylbenzylaminoethyl) γ-aminopropyltrimethoxysilane hydrochloride, hexamethyldisilazane, methyltrimethoxysilane, butyltrimethoxysilane, isobutyltrimethoxysilane, hexyltrimethoxysilane, octyltrimethoxysilane, decyltrimethoxysilane, dodecytrimethoxysilane, phenyltrimethoxysilane, o-methylphenyltrimethoxysilane and p-methylphenyltrimethoxysilane.
As the silicone oil, dimethylsilicone oil, methylphenylsilicone oil and amino-modified silicone oil are usable.
The particle diameter of the colored particle of the toner of the invention is preferably within the range of from 2 to 10 μm, more preferably 3.0 to 9.0 μm, in a volume average diameter measured by Coulter Counter TA-11 or Coulter Multisizer. The diameter of the colored particle can be controlled by production conditions thereof. Preferably, the colored particles contain the polypropylene having a diameter of 0.1 to 0.8 μm, more preferably 0.3 to 0.8 μm.
(5) Free polypropylene index
In the toner of the invention, the free polypropylene index measured by the centrifugal separation method is within the range of from 0.1 to 0.7, preferably 0.1 to 0.5, more preferably 0.1 to 0.4, and the free polypropylene index measured by the natural sedimentation method is not more than 0.1, preferably not more than 0.05.
(5-1) Free polypropylene index measured by the centrifugal separation method
In the invention, “free polypropylene index measured by the centrifugal separation method” is a light absorbency or turbidity of the top clean liquid obtained by centrifugal separation of a suspension of the toner. In concrete, the light absorbency at 500 nm of the top clean liquid obtained by the following procedures (I) to (iv).
(i) Preparation of a surfactant solution:
A suitable amount of water is put in a measuring flask of 100 ml and 1 ml of a 12% solution of sodium dodecylbenzenesulfonate is added, then water is gently added to make the volume of the solution to 100 ml to prepare a surfactant solution.
(ii) Preparation of toner suspension
In a bottle of 50 ml with screw stopper, 15 g of toner is put, and 30 ml of the surfactant solution prepared in (i) is gently added. The bottle is closed by a stopper and the shook for 1 minute by hand to prepare a suspension of the toner.
(iii) Centrifugal separation
The suspension of toner is put into a 50 ml separation tube and the tube set in an angle rotor having a rotating radius of 70 mm. Then the suspension is subjected to the centrifugal separation for 20 minutes at 5000 rpm.
(iv) Separation of top clean liquid
The low molecular weight polypropylene particles adhered on the internal wall of the centrifuge tube are washed off from the wall by the top clean liquid using a pipette and the top clean liquid is sampled. If the precipitated toner particles are mixed in the top clean liquid, the liquid is stood for one day in the tube or subjected to the centrifugal separation once more.
(5-2) Free polypropylene index measured by the natural sedimentation method
In the invention, “free polypropylene index measured by the natural sedimentation method” is a light absorbency or turbidity of the top clean liquid obtained by natural sedimentation of a suspension of the toner. In concrete, the light absorbency at 500 nm of the top clean liquid obtained by the following procedures (I) to (iii).
(i) Preparation of a surfactant solution:
A suitable amount of water is put in a measuring flask of 100 ml and 1 ml of a 12% solution of sodium dodecylbenzenesulfonate is added, then water is gently added to make the volume of the solution to 100 ml to prepare a surfactant solution.
(ii) Preparation of toner suspension
In a bottle of 50 ml with screw stopper, 15 g of toner is put, and 30 ml of the surfactant solution prepared in (i) is gently added. The bottle is closed by the stopper and shook for 1 minute by hand, until the aggregation of toner can not be found by eyes view, as a guideline, to prepare a suspension of the toner.
(iii) Separation of top clean liquid
The toner suspension is stood for one day and the top clean liquid is sampled by a pipette.
When the free polypropylene index measured by centrifugal separation method is not less than 0.1, a suitable amount of free polypropylene particles is fixed on the surface of the toner particle with a suitable adhesive force with the result that a high transferring ability of the toner can be obtained.
Moreover, when the free polypropylene index measured by natural sedimentation method is not more than 0.1 and the free polypropylene index measured by centrifugal separation method is not more than 0.7, the charging ability of the toner can be stabilized with the result that the formation of toner film on the photoreceptor can be inhibited.
(6) Shape of the free polypropylene particle separated by the centrifugal method
It is preferred in the toner of the invention that the shape of the free polypropylene particle separated by the centrifugal method is substantially ellipsoid and that the number of the free polypropylene particles having a elliptical ratio (shorter axis/longer axis) of the projection image of the ellipsoid of not more than 0.8 accounts not less than 50% of the number of the whole free polypropylene particles.
The elliptical ratio of the free polypropylene particles separated by the centrifugal method can be determined by the following procedures: a white turbid part floating at the surface the top clean liquid is sampled after the centrifugal separation according to the above (5-1) (iii); the sampled portion is filtered to obtained a white powder; the white powder is subjected to IR measurement to confirm that the powder is polypropylene; the microscopic image of the powder is taken by a scanning electron micrometer T-330, (manufactured by Nihon Denshi Co., Ltd., acceleration potential: 20 kV, Spot size: 3 p.m. o'clock-set, in gamma intensifying mode); and the images of 2000 particles are subjected to image analyzing by an image analyzer SIA, manufactured by Nihon Denshi Co., Ltd.
When the number ratio of the free polypropylene particles each having an elliptical ratio of not more than 0.8 is not less than 50%, the charging ability of the toner can be stabilized with the result that the formation of toner film on the photoreceptor can be inhibited when the toner is used in the image forming process.
(7) Volume average diameter of the free polypropylene particles separated by the centrifugal method
In the toner of the invention, it is preferred that the ratio of the free polypropylene particles separated by the centrifugal method having diameter of not more than 4 μm is not less than 50%, more preferably not less than 60%, in number of the whole polypropylene particles by the centrifugal method.
The volume average diameter of the free polypropylene particles can be measured by subjecting the free polypropylene particles in the top clean liquid obtained by the procedure of the above (5-1) (iv) to measure by Coulter Multisizer manufactured by Coulter Co., Ltd.
The transferring ability of the toner can be stabilized when the free polypropylene particle each having a diameter of not more than 4 μm are existed in a ratio of not less than 50% in number.
(8) Producing method of the toner
The toner of the invention can be prepared by either a crushing method or a polymerization method. The crushing method is preferred.
In the crushing method, a raw composition of the toner containing raw materials such as the binder resin, the coloring agent, the mold releasing agent, or the low molecular weight polypropylene particles, and the charge controlling agent, is molten, kneaded, crushed and classified to prepare the colored particles. The crushing and classifying processes may be repeated when it is necessary. The external additive is added to the colored particles to prepare the toner.
In the above, the diameter of the low molecular weight polypropylene particle is preferably within the range of from 0.1 to 100 μm, more preferably from 0.3 to 6 μm.
The shape of the low molecular weight polypropylene particles is preferably ellipsoid or spindle-shape.
The low molecular weight polypropylene particles each having the above-mentioned diameter and shape can be produced by controlling the diameter and the shape by a solution sedimentation process and a heat treatment, respectively.
The solution sedimentation process is a method in which the low molecular weight polypropylene particles are dissolved in toluene under reflux and the solution is cooled to 50° C. Then the solution is poured into cold acetone. Thus the low molecular weight polypropylene particles each having a diameter within a specified range.
The shape of the low molecular weight polypropylene particle can be made such as the spindle-shape by putting the particle in heated air blow. In such the case, the temperature of the air blow is preferably from 100 to 140 C. When the temperature is excessively high, the shape of the low molecular weight polypropylene particles tends to be made not uniform. The heat treatment may be carried out in a liquid.
As is mentioned above, the free propylene index by centrifugal method is within the range of from 0.1 to 0.7 and the free polypropylene index by natural sedimentation method is not more than 0.1 in the toner of the invention, with the result that the occurrence of toner-filming can be inhibited while maintaining a sufficiently high transferring ability when the toner is used in the image forming process.
In the toner of the invention, the occurrence of toner-filming can be inhibited when the shape of the free polypropylene particle the centrifugal method is substantially ellipsoid and the ratio of the particles each having a elliptical ratio of the ellipsoid of not more than 0.8 account not less than 50% in number of the whole free polypropylene particles.
Moreover, a sufficiently high transferring ability in the image forming process can be obtained when the ratio of the number of the free polypropylene particles the centrifugal method having a diameter on not more than 4 μm to the whole number of free polypropylene particles is not less than 50%.
Developer
The toner according to the invention can be used in any embodiment such as (1) singly use as a magnetic toner containing a magnetic substance, (2) singly use as a non-magnetic toner containing no magnetic substance and (3) use as a two-component developer together with a carrier. Among them the embodiment (3) is preferred.
As the carrier to be mixed with the toner, both of (1) a carrier composed of only a magnetic substance particle such as iron and ferrite, and (2) a resin coated carrier composed of a core particle covered with a resin may be used. The above-mentioned resin coated carrier (2) is preferred from the viewpoint of durability thereof.
The volume average particle diameter of the carrier is preferably from 10 to 100 μm, more preferably 20 to 80 μm.
The magnetic property of the carrier is preferably from 30 to 80 emu/g in the saturated magnetization.
Iron powder, magnetite and various types of ferrite are usable for the core particle of the resin coated carrier. Among them, magnetite and ferrite are preferred.
A ferrite containing a heavy metal such as copper, zinc, nickel or manganese, and a ferrite containing a light metal such as an alkali metal, for example Li and Na, and/or alkali-earth metal, for example Mg, Ca, Sr and Ba, are preferably used. The light metal-containing ferrite is particularly preferred.
The light metal-containing ferrite has the composition showing the following formula (1) or (2).
(1) (M2O)x(Fe2O3)1−x
(2) (MO)x(Fe2O3)1−x
In Formula (1) or (2), M is an alkyl or an alkali-earth metal. Ones may be used, in which a part of M2O and/or Fe2O3 is replaced by an alkali-earth metal.
In the Formulas, x is not more than 30 mole-%, preferably not more than 18 mole-%, and the replacing amount of alkali-earth metal and/or alkali metal is preferably from 1 to 15 mole-%, more preferably from 3 to 15 mole-%.
The light metal-containing ferrite and magnetite are preferred since not only problems of pollution caused by the industrial waste can be reduced but the weight of the carrier can be reduced with result of that the stress on the toner can be decreased.
The resin of the resin coated carrier is not specifically limited, and a resin such as a silicone resin, styrene-acryl resin, a vinylidene fluoride and a fluoride resin other than vinylidene fluoride resin are usable.
As the silicone resin, a condensation reactive type silicone resin is preferably used, which is hardened by a heat dehydration condensation reaction or a room temperature moisture hardening reaction. A condensation reactive type silicone resin capable of hardening by the following reaction a or b is particularly preferable.
In the above reaction formulas, R1 and R2 are each a substituent such as an alkyl group, and OX is an alkoxy group or an acyloxy group.
Among them, ones in which the substituent is a methoxy group are suitable for producing a carrier having a high durability since a dense coating layer is formed thereby.
Examples of silicone resin preferably usable in the invention include SR-2411 and SR-2710, manufactured by Toray Silicone co., Ltd., and KR-255 and KR-271, manufactured by Shin'etsu Kagaku Co., Ltd.
Examples of the styrene-acryl resin include copolymers of styrene or a styrene derivative such as styrene, o-methylstyrene, m-methylstyrene, p-methylstyrene, α-methylstyrene, p-chlorostyrene, 3,4-dichlorostyrene, p-phenylstyrene, p-ethylstyrene, 2,4-dimethylstyrene, p-t-butylstyrene, p-n-hexylstyrene, p-n-octylstyrene, p-n-nonylstyrene, p-n-decylstyrene and p-n-dodecylstyrene with a methacrylic acid ester derivative such as methyl methacrylate, ethyl methacrylate, n-butyl methacrylate, isopropyl methacrylate, isobutyl methacrylate, t-butyl methacrylate, n-octyl methacrylate, 2-ethylhexyl methacrylate, stearyl methacrylate, lauryl methacrylate, phenyl methacrylate, and diethylaminoethyl methacrylate, or an acrylic acid ester derivative such as methyl acrylate, ethyl acrylate, isopropyl acrylate, n-butyl acrylate, t-butyl acrylate, isobutyl acrylate, n-octyl acrylate, 2-ethylhexyl acrylate, stearyl acrylate, lauryl acrylate, phenyl acrylate, dimethylaminoethyl acrylate and diethylaminoethyl acrylate, are usable. The content of styrene or its derivative is preferably from 20 to 90% by weight from the view point of the improvement in the durability.
As the vinylidene fluoride resin, a copolymer of vinylidene fluoride and tetrafluoroethylene, hexafluoropropylene, monochlorotrifluoroethylene, monofluoroethylene or trifluoroethylene and another copolymer component is preferred, and ones containing vinylidene fluoride in a ratio of not less than 20% by weight is particularly preferred.
The copolymer component of the vinylidene fluoride other than the halogen-containing monomer includes copolymers of stylene and its derivative such as styrene, o-methylstyrene, m-methylstyrene, p-methylstyrene, α-methylstyrene, p-chlorostyrene, 3,4-dichlorostyrene, p-phenylstyrene, p-ethylstyrene, 2,4-dimethylstyrene, p-t-butylstyrene, p-n-butylstyrene, p-n-hexylstyrene, p-n-octylstyrene, p-n-nonylstyrene, p-n-decylstyrene and p-n-dodecylstyrene, with a methacrylic acid ester derivative such as methyl methacrylate, ethyl methacrylate, n-butyl methacrylate, isopropyl methacrylate, isobutyl methacrylate, t-butyl methacrylate, n-octyl methacrylate, 2-ethylhexyl methacrylate, stearyl methacrylate, lauryl methacrylate, phenyl methacrylate, diethylaminoethyl methacrylate and dimethylaminoethyl methacrylate, and/or an acrylic acid ester derivative such as methyl acrylate, ethyl acrylate, isopropyl acrylate, n-butyl acrylate, t-butyl acrylate, isobutyl acrylate, n-octyl acrylate, 2-ethylhexyl acrylate, stearyl acrylate, lauryl acrylate, phenyl acrylate, dimethylaminoethyl acrylate and diethylaminoethyl acrylate.
The fluorine-containing resin other than vinylidene fluoride includes a resin derived from a compound represented by the following Formula (3).
In the formula, R is a hydrogen atom or a methyl group, X is an oxygen atom, a —COO group or a ═CO group, and Rf is a fluoroalkyl group.
The following method can be applied for coating the resin on the core particle; (1) a solution of the resin is sprayed to the core particle and dried, (2) particles of the coating resin is electrostatically adhered to the core particle and mechanical energy is applied thereon to form a coating layer, (3) particles of the coating resin is electrostatically adhered to the core particle and the core particle is heated at a temperature higher than the melting point of the resin to form a coating resin layer, (4) the core particle is immersed in a solution of the resin, and (5) the resin containing a hardener is coated on the core particle and heated to harden the resin layer.
The amount of the coated resin may be an amount sufficient for uniformly covering the core particle surface, and concretely from 0.1 to 5.0%, preferably from 0.5 to 3.0%, by weight of the core particles. The effect of the resin coating cannot be obtained when the coated amount is too low, and an image defect tends to be caused by the resin peeled from the core surface when the coated amount of resin is excessive.
Image Forming Method
In the image forming method using the toner of the invention, it is preferable that the toner remained on the photoreceptor is recovered by a cleaning device and the recovered toner is returned to a developing device or a toner supplying device to reuse the toner.
Examples of the invention are described below. Example of a carrier preparation
A raw composition of carrier composed of 1000 parts by weight of ferrite particles, as the core particles, having a volume average diameter of 70 μm and 20 parts by weight of 1,1,1-trifluoroethyl methacrylate powder, as the resin powder, having a volume average diameter of 0.1 μm was put into a horizontal rotation impeller mixer. Then the mixture was heated to 70° C. while stirring with an impeller circumference speed of 4 m/sec. The mixture was further stirred for 20 minutes to prepare a carrier.
A raw composition of toner composed of 1000 parts by weight of polyester resin, as the binder resin, 10 parts by weight of carbon black as the colorant, and 3 parts by weight of low molecular weight polypropylene as the mold releasing agent was preliminary mixed by a Henschel mixer for 5 minutes. The mixing conditions were adjusted so that the bulk density and the particle diameter distribution of the toner particles were made as shown in Table 1 and Table 2, respectively. The low molecular weight polypropylene had a shape of ellipsoid having an elliptical ratio of projection image of 0.66, a particle diameter of 0.7 μm, Mw of 6600, Mn of 3000, Mw/Mn of 2.20 and a melting point of 80° C. The mixture was molten and kneaded by a biaxial kneading extruder set at 170° C. Then the mixture was roughly crushed by a hammer mill and finely crushed by a jet crusher, and classified by a wind classification machine to prepare colored particles (A) having a volume average diameter of 10.0 μm were obtained.
To 100 parts by weight of the colored particles (A) thus obtained, 1 part by weight of hydrophobic silica was externally added and mix by a Henschel mixer with a stirring speed of 40 m/sec for 20 minutes at 37° C. Thus Toner (A) according to the invention was obtained, which has the properties shown in Table 3.
A raw composition of toner composed of 100 parts by weight of polyester resin, as the binder resin, 10 parts by weight of carbon black, as the colorant, and 4 parts by weight of low molecular weight polypropylene, as the mold releasing agent, was preliminary mixing by a Loedige mixer (crushing blade: ON) for 5 minutes. The mixing conditions were adjusted so that the toner has the bulk density shown in Table 1 and the particle diameter distribution shown in Table 2. The low molecular weight polypropylene had a shape of ellipsoid having an elliptical ratio of projection image of 0.31, a particle diameter of 0.8 μm, Mw of 13700, Mn of 7200, Mw/Mn of 1.90 and a melting point of 133° C. The mixture was molten and kneaded by a biaxial kneading extruder set at 170° C. Then the mixture was roughly crushed by a hammer mill and finely crushed by a jet crusher, and classified by as wind classification machine to prepare colored particles (B) having a volume average diameter of 9.0 μm were obtained.
To 100 parts by weight of the colored particles (B) thus obtained, 1 part by weight of hydrophobic silica was externally added and mix by a Henschel mixer with a stirring speed of 40 m/sec for 20 minutes at 37° C. Thus Toner (B) according to the invention was obtained, which has the properties shown in Table 3.
A raw composition of toner composed of 100 parts by weight of styrene-acryl resin, as the binder resin, 10 parts by weight of carbon black, as the colorant, and 4 parts by weight of low molecular weight polypropylene, as the mold releasing agent, was preliminary mixing by a Herschel mixer for 5 minutes. The mixing conditions were adjusted so that the toner has the bulk density shown in Table 1 and the particle diameter distribution shown in Table 2. The low molecular weight polypropylene had a shape of ellipsoid having an elliptical ratio of projection image of 0.55, a particle diameter of 1.0 pim, Mw of 13700, Mn of 7200, Mw/Mn of 1.90 and a melting point of 133° C. The mixture was molten and kneaded by a biaxial kneading extruder set at 170° C. Then the mixture was roughly crushed by a hammer mill and finely crushed by a jet crusher, and classified by as wind classification machine to prepare colored particles (C) having a volume average diameter of 8.0 μm were obtained.
To 100 parts by weight of the colored particles (B) thus obtained, 1 part by weight of hydrophobic silica was externally added and mix by a Henschel mixer with a stirring speed of 40 m/sec for 20 minutes at 37° C. Thus Toner (C) according to the invention was obtained, which has the properties shown in Table 3.
A raw composition of toner composed of 100 parts by weight of polyester resin, as the binder resin, 10 parts by weight of carbon black, as the colorant, and 4 parts by weight of low molecular weight polypropylene, as the mold releasing agent, was preliminary mixing by a Loedige mixer (crushing wing ON) for 5 minutes. The mixing conditions were adjusted so that the bulk density shown in Table 1 and the particle diameter distribution shown in Table 2 were obtained. The low molecular weight polypropylene had a sphere shape, a particle diameter of 100 pm, Mw of 8600, Mn of 2270, Mw/Mn of 3.79 and a melting point of 135° C. The mixture was molten and kneaded by a biaxial kneading extruder set at 170° C. Then the mixture was roughly crushed by a hammer mill and finely crushed by a jet crusher, and classified by as wind classification machine to prepare comparative colored particles (a) having a volume average diameter of 10.0 μm were obtained.
To 100 parts by weight of the comparative colored particles (a) thus obtained, 1 part by weight of hydrophobic silica was externally added and mix by a Henschel mixer with a stirring speed of 25 m/sec for 15 minutes at 17° C. Thus Toner (a) according to the invention was obtained, which has the properties shown in Table 3.
Comparative colored particles (b) and (c) were prepared in the same manner as in comparative toner (a) except that the mixing conditions were changed so that the bulk density shown in Table 1 and the particle diameter distribution shown in Table 2 were obtained. The comparative colored particles (b) and (c) each had a volume average diameter of 10.0 μm. Then comparative toners (b) and (c) having the properties shown in Table 3 were prepared using the comparative colored particles (b) and (c), respectively.
| TABLE 1 | ||
| Bulk density (g/cm3) | ||
| Mixing time | Changing ratio |
| 1 min. | 5 min. | (%) | ||
| Example 1 | 0.609 | 0.618 | 101.5 | ||
| Example 2 | 0.589 | 0.592 | 100.5 | ||
| Example 3 | 0.556 | 0.575 | 103.4 | ||
| Comparative | 0.651 | 0.650 | 99.8 | ||
| example 1 | |||||
| Comparative | 0.610 | 0.609 | 99.8 | ||
| example 2 | |||||
| Comparative | 0.596 | 0.594 | 99.7 | ||
| example 3 | |||||
| TABLE 2 |
| Particle diameter distribution (%) |
| Mesh | Mixing time; 1 min. | Mixing time: 5 min. |
| (μm) | 250 | 150 | 75 | 45 | 20 | 250 | 150 | 75 | 45 | 20 |
| Ex. 1 | 59.0 | 18.8 | 20.9 | 1.2 | 0.1 | 56.2 | 15.7 | 24.9 | 3.1 | 0 |
| Ex. 2 | 61.4 | 17.0 | 18.4 | 3.1 | 0.1 | 62.9 | 12.3 | 16.4 | 8.1 | 0.2 |
| Ex. 3 | 59.9 | 17.3 | 19.5 | 3.2 | 0.1 | 59.5 | 18.1 | 20.5 | 1.8 | 0 |
| Comp. | 68.8 | 14.5 | 12.4 | 4.1 | 0.1 | 70.9 | 14.5 | 9.8 | 4.5 | 0.3 |
| Ex. 1 | ||||||||||
| Comp. | 70.9 | 15.5 | 10.4 | 3.1 | 0.1 | 70.1 | 15.2 | 11.2 | 3.4 | 0.1 |
| Ex. 2 | ||||||||||
| Comp. | 69.6 | 16.5 | 11.1 | 2.7 | 0.1 | 69.5 | 16.5 | 11.1 | 2.8 | 0.1 |
| Ex. 3 | ||||||||||
| TABLE 3 | |||||||
| Polypropylene | Free polypropylene | Free polypropylene | Ratio of free polypropylene | Ratio of free polypropylene | |||
| content | index by | index by | particle of diameter | particle of elliptical ratio | |||
| Toner | (Parts by weight) | centrifugal separation | natural precipitation | of not more than 4 μm (number %) | of not more than 0.8 (number %) | ||
| Ex. 1 | A | 3 | 0.25 | 0.01 | 65 | 70 |
| Ex. 2 | B | 4 | 0.61 | 0.02 | 55 | 60 |
| Ex. 3 | C | 4 | 0.12 | 0.04 | 75 | 55 |
| Comp. | a | 4 | 0.89 | 0.12 | 45 | 45 |
| Ex. 1 | ||||||
| Comp. | b | 4 | 0.96 | 0.18 | 86 | 14 |
| Ex. 2 | ||||||
| Comp. | c | 4 | 1.80 | 0.26 | 37 | 43 |
| Ex. 3 | ||||||
Preparation Example of Developer
Toners for evaluation were prepared by mixing the toner of the invention (A), (B) or (C), or the comparative toner (a), (b) or (c) was mixed with the carrier so that the ratio of the toner to the carrier was made to 5% by weight.
Evaluation by actual photographing
An original picture having a blackened area ratio of 5% was copied by an electrophotographic copying machine Konica 2125 under conditions of high temperature at 33° C. and high moisture at relative humidity of 80%. Konica 2125 was previously modified so as to change the cleaning blade load to 18.6 g/cm. The test results were evaluated regarding the following items (1) to (4). Results of evaluation are shown in Table 4.
(1) Toner filming
The number of copied sheet was counted until a band-like contamination in the transporting direction of paper was occurred.
(2) Black spot contamination
The number of copied sheet was counted until 10 black spots contamination having a diameter 0.8 mm or more were formed on the white background area of the copy.
(3) Life of developer
The reflective density of the white background area of the copy sheet was measured by Sakura Densitometer, manufactured by Konica Corp., and the number of copied sheet was counted until the relative reflective density was exceed 0.01. The relative reflective density was determined based on the density of the paper it self of 0.
(4) Transferring ratio
A solid image having a density of 1.3 and the area of 20 mm×50 mm was prepared, and the transferring ratio was calculated by the following equation.
| TABLE 4 | ||||||
| Occurrence | ||||||
| of toner | Formation of | Life of | Transferring | |||
| Toner | filming | black spot | developer | ratio (%) | ||
| Ex. 1 | A | Not occurred | Not formed | 60,000 | 98 |
| until | until | sheets | |||
| 120,000th | 120,000th | ||||
| sheets | sheets | ||||
| Ex. 2 | B | Not occurred | Not formed | 50,000 | 96 |
| until | until | sheets | |||
| 100,000th | 100,000th | ||||
| sheets | sheets | ||||
| Ex. 3 | C | Not occurred | Not formed | 80,000 | 97 |
| until | until | sheets | |||
| 140,000th | 140,000th | ||||
| sheets | sheets | ||||
| Comp. | a | Occurred at | Formed at | 20,000 | 78 |
| Ex. 1 | 20,000th | 40,000th | sheets | ||
| sheets | sheets | ||||
| Comp. | b | Occurred at | Formed at | 20,000 | 79 |
| Ex. 2 | 20,000th | 80,000th | sheets | ||
| sheets | sheets | ||||
| Comp. | c | Occurred at | Formed at | 20,000 | 69 |
| Ex. 3 | 20,000th | 30,000th | sheets | ||
| sheets | sheets | ||||
As above-described, the off-set problem is prevented and formation of black spot can be inhibited by the toner according to the invention when the toner is used in the image forming process. Furthermore, formation of black spot contamination on the image can be prevented and the life of developer can be prolonged by the use of the toner of the invention.
Claims (18)
1. A toner for developing an electrostatic image comprising a colored particle containing a binder resin, a colorant, and a low molecular weight polypropylene particle, which has a content of the low molecular weight polypropylene particle of 0.3 to 8.0 percent by weight, a free polypropylene index measured by a centrifugal method of from 0.1 to 0.7 and a free polypropylene index measured by a natural sedimentation method of not more than 0.1.
2. The toner of claim 1 wherein shape of the polypropylene particle separated by the centrifugal method is substantially ellipsoid.
3. The toner of claim 2 wherein a number ratio of ellipsoid polypropylene particles each having an elliptical axis ratio of projection image of not more than 0.8 to the whole number of the separated polypropylene particles is not less than 50 percent.
4. The toner of claim 1 wherein ratio of number of the polypropylene particles separated by centrifugal method having a particle diameter of not more than 4 μm to the whole number of the polypropylene particles separated by the centrifugal method is not less than 50%.
5. The toner of claim 4 wherein ratio of number of the polypropylene particles having a particle diameter of not more than 4 μm to the whole number of the polypropylene particles separated by the centrifugal method is not less than 60%.
6. The toner of claim 1 wherein molecular weight of the low molecular weight polypropylene particle is 1,500 to 10,000, in number average molecular weight Mn.
7. The toner of claim 6 the wherein molecular weight of the low molecular weight polypropylene particle is 1,800 to 4,000, in number average molecular weight Mn.
8. The toner of claim 1 wherein ratio of weight average molecular weight Mw to number average molecular weight Mn of the low molecular weight polypropylene particle Mw/Mn is 1.5 to 3.
9. The toner of claim 8 wherein the ratio of the weight average molecular weight Mw to the number average molecular weight Mn of the low molecular weight polypropylene particle Mw/Mn 2 to 3.
10. The toner of claim 8 wherein content of the low molecular weight polypropylene particles in the toner is 1.3 to 5.7% by weight.
11. The toner of claim 3 wherein ratio of number of the polypropylene particles separated by centrifugal method having a particle diameter of not more than 4 μm to the whole number of the polypropylene particles separated by the centrifugal method is not less than 50%.
12. The toner of claim 3 wherein the molecular weight of polypropylene particle is 1,500 to 10,000, in number average molecular weight Mn, and ratio of weight average molecular weight Mw to Mn of the polypropylene particles is 1.5 to 3.
13. The toner of claim 1 wherein the colored particles has a volume average particle dimeter of 2 to 10 μm.
14. The toner of claim 12, further comprising an external additive having number average primary diameter of 5 to 20 nm.
15. The toner of claim 12, comprising a free polypropylene index measured by centrifugal method of 0.1 to 0.4.
16. The toner of claim 15, comprising a free polypropylene index measured by a natural sedimentation is not more than 0.05.
17. A developer for developing an electrostatic image comprising a toner of claim 1.
18. The developer of claim 17, further comprising a carrier.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP10-367640 | 1998-12-24 | ||
| JP36764098A JP2000187349A (en) | 1998-12-24 | 1998-12-24 | Toner |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| US6228551B1 true US6228551B1 (en) | 2001-05-08 |
Family
ID=18489825
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US09/469,383 Expired - Lifetime US6228551B1 (en) | 1998-12-24 | 1999-12-22 | Toner for developing electrostatic image |
Country Status (2)
| Country | Link |
|---|---|
| US (1) | US6228551B1 (en) |
| JP (1) | JP2000187349A (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20070056359A1 (en) * | 2005-08-19 | 2007-03-15 | Xerox Corporation | Direct method to determine particulate concentration in a dispersion |
Families Citing this family (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP3925260B2 (en) * | 2001-03-21 | 2007-06-06 | コニカミノルタホールディングス株式会社 | Toner for electrostatic image development and image forming method |
| JP2002365830A (en) * | 2001-06-04 | 2002-12-18 | Ricoh Co Ltd | Electrophotographic toner and image forming method |
| JP2005227325A (en) * | 2004-02-10 | 2005-08-25 | Fuji Xerox Co Ltd | Electrophotographic toner and method for manufacturing the same |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5424162A (en) * | 1992-11-24 | 1995-06-13 | Minolta Camera Kabushiki Kaisha | Toner for electrophotography containing wax-particles dispersed in binder resin |
| US5814428A (en) * | 1997-03-04 | 1998-09-29 | Minolta Co., Ltd. | Toner for developing electrostatic latent image |
| US5824446A (en) * | 1996-04-23 | 1998-10-20 | Minolta Co., Ltd. | Toners for developing electrostatically charged images |
| US5935751A (en) * | 1996-06-27 | 1999-08-10 | Fuji Xerox Co., Ltd. | Toner for developing electrostatic latent image, process for manufacturing the same, developer for electrostatic latent image, and image-forming method |
-
1998
- 1998-12-24 JP JP36764098A patent/JP2000187349A/en active Pending
-
1999
- 1999-12-22 US US09/469,383 patent/US6228551B1/en not_active Expired - Lifetime
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5424162A (en) * | 1992-11-24 | 1995-06-13 | Minolta Camera Kabushiki Kaisha | Toner for electrophotography containing wax-particles dispersed in binder resin |
| US5824446A (en) * | 1996-04-23 | 1998-10-20 | Minolta Co., Ltd. | Toners for developing electrostatically charged images |
| US5935751A (en) * | 1996-06-27 | 1999-08-10 | Fuji Xerox Co., Ltd. | Toner for developing electrostatic latent image, process for manufacturing the same, developer for electrostatic latent image, and image-forming method |
| US5814428A (en) * | 1997-03-04 | 1998-09-29 | Minolta Co., Ltd. | Toner for developing electrostatic latent image |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20070056359A1 (en) * | 2005-08-19 | 2007-03-15 | Xerox Corporation | Direct method to determine particulate concentration in a dispersion |
| US7334458B2 (en) * | 2005-08-19 | 2008-02-26 | Xerox Corporation | Direct method to determine particulate concentration in a dispersion |
Also Published As
| Publication number | Publication date |
|---|---|
| JP2000187349A (en) | 2000-07-04 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US8592114B2 (en) | Toner for developing electrostatic images | |
| JP4054494B2 (en) | Toner for electrostatic latent image development | |
| US20080233506A1 (en) | Toner, and oilless fixing method and process cartridge using the toner | |
| EP0627669B1 (en) | Toner for developing electrostatic image and process for production thereof | |
| JPS6214166A (en) | Magnetic toner | |
| US4882258A (en) | Toner for development of electrostatic image and electrostatic latent image developer | |
| US20100272471A1 (en) | Electrophotographic toner and electrophotographic image forming apparatus using the same | |
| US4960664A (en) | Developer composition for developing electrostatic image and toner image forming process | |
| US10254666B2 (en) | Toner for development electrostatic images | |
| US6821702B2 (en) | Method of forming images | |
| US5482807A (en) | Positively-chargeable toner | |
| US6228551B1 (en) | Toner for developing electrostatic image | |
| US7364826B2 (en) | Electrostatic latent image developing toner, and method for manufacturing electrostatic latent image developer and electrostatic latent image developing toner | |
| JP4140197B2 (en) | Toner for developing electrostatic latent image and image forming method using the same | |
| JP3752877B2 (en) | Toner for developing electrostatic image, method for producing the same, electrostatic image developer, and image forming method | |
| JP2625804B2 (en) | Electrostatic latent image developing toner and method of manufacturing the same | |
| US5624778A (en) | Carrier for electrophotography, and two-component type developer having the carrier | |
| JP3083023B2 (en) | Electrostatic image developing toner and method of manufacturing the same | |
| JPH0814723B2 (en) | toner | |
| JP3919348B2 (en) | Toner and image forming method | |
| US7399567B2 (en) | Toner for electrostatic latent image development, electrostatic latent image developer containing the toner, and image formation method using the developer | |
| JP2004109406A (en) | Electrophotographic toner | |
| JP2000221726A (en) | Electrophotographic toner | |
| JP4219504B2 (en) | Toner for developer and developer | |
| JPH01224776A (en) | Color toner and method for fixing same |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AS | Assignment |
Owner name: KONICA CORPORATION, JAPAN Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNORS:OHMURA, KEN;OSHIBA, TOMOMI;REEL/FRAME:010508/0249 Effective date: 19991125 |
|
| FEPP | Fee payment procedure |
Free format text: PAYOR NUMBER ASSIGNED (ORIGINAL EVENT CODE: ASPN); ENTITY STATUS OF PATENT OWNER: LARGE ENTITY |
|
| STCF | Information on status: patent grant |
Free format text: PATENTED CASE |
|
| FPAY | Fee payment |
Year of fee payment: 4 |
|
| CC | Certificate of correction | ||
| CC | Certificate of correction | ||
| FPAY | Fee payment |
Year of fee payment: 8 |
|
| FPAY | Fee payment |
Year of fee payment: 12 |