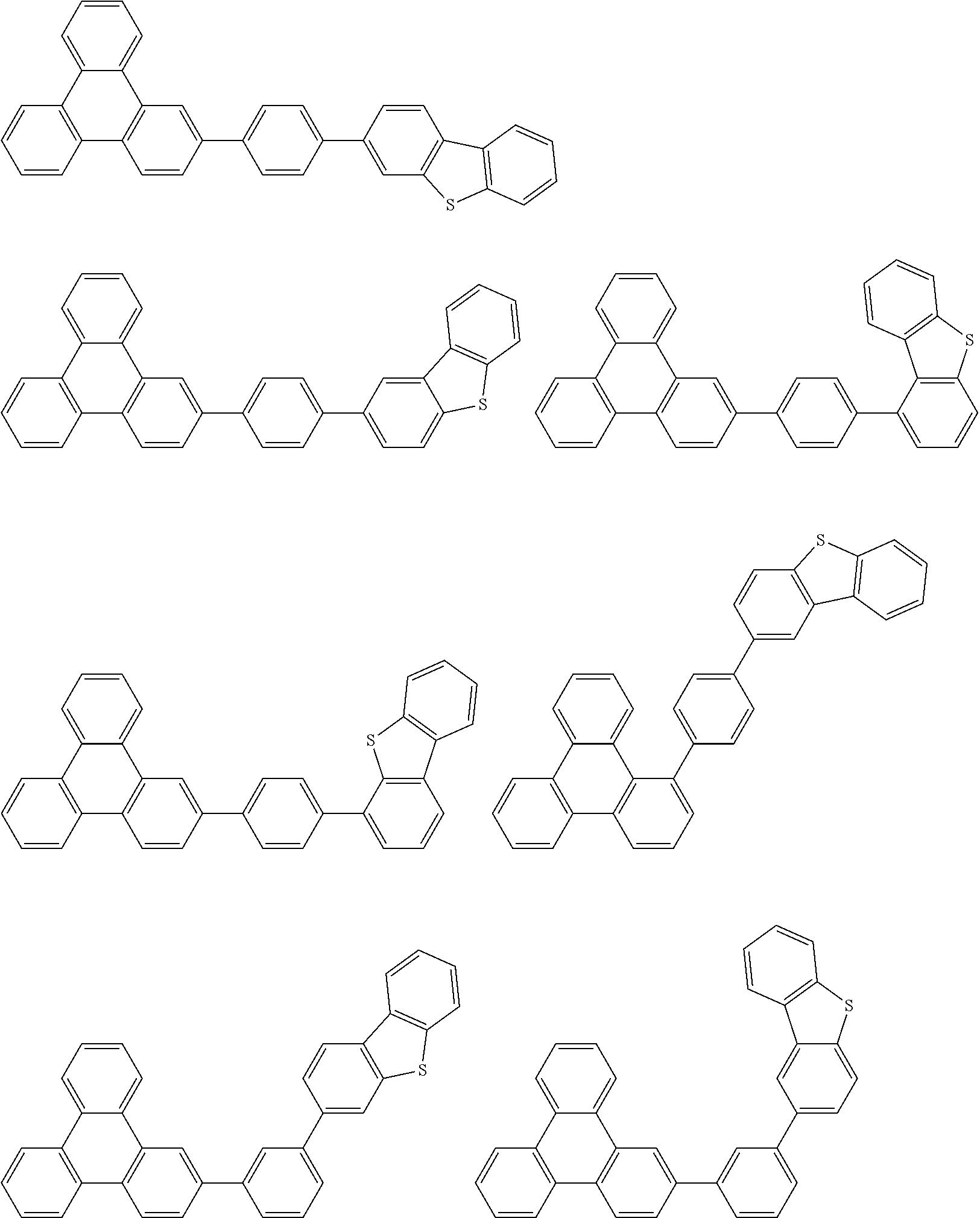US20180358564A1 - Organic light-emitting device - Google Patents
Organic light-emitting device Download PDFInfo
- Publication number
- US20180358564A1 US20180358564A1 US16/105,931 US201816105931A US2018358564A1 US 20180358564 A1 US20180358564 A1 US 20180358564A1 US 201816105931 A US201816105931 A US 201816105931A US 2018358564 A1 US2018358564 A1 US 2018358564A1
- Authority
- US
- United States
- Prior art keywords
- group
- substituted
- fluorenyl
- salt
- spiro
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Abandoned
Links
Images
Classifications
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/615—Polycyclic condensed aromatic hydrocarbons, e.g. anthracene
- H10K85/622—Polycyclic condensed aromatic hydrocarbons, e.g. anthracene containing four rings, e.g. pyrene
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/11—OLEDs or polymer light-emitting diodes [PLED] characterised by the electroluminescent [EL] layers
-
- H01L51/0072—
-
- H01L27/3248—
-
- H01L51/0052—
-
- H01L51/0054—
-
- H01L51/0058—
-
- H01L51/0059—
-
- H01L51/006—
-
- H01L51/0061—
-
- H01L51/0067—
-
- H01L51/0074—
-
- H01L51/0085—
-
- H01L51/5028—
-
- H01L51/5056—
-
- H01L51/5072—
-
- H01L51/5088—
-
- H01L51/5092—
-
- H01L51/5206—
-
- H01L51/5221—
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K30/00—Organic devices sensitive to infrared radiation, light, electromagnetic radiation of shorter wavelength or corpuscular radiation
- H10K30/80—Constructional details
- H10K30/865—Intermediate layers comprising a mixture of materials of the adjoining active layers
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/11—OLEDs or polymer light-emitting diodes [PLED] characterised by the electroluminescent [EL] layers
- H10K50/12—OLEDs or polymer light-emitting diodes [PLED] characterised by the electroluminescent [EL] layers comprising dopants
- H10K50/121—OLEDs or polymer light-emitting diodes [PLED] characterised by the electroluminescent [EL] layers comprising dopants for assisting energy transfer, e.g. sensitization
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/14—Carrier transporting layers
- H10K50/15—Hole transporting layers
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/14—Carrier transporting layers
- H10K50/16—Electron transporting layers
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/17—Carrier injection layers
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/17—Carrier injection layers
- H10K50/171—Electron injection layers
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K59/00—Integrated devices, or assemblies of multiple devices, comprising at least one organic light-emitting element covered by group H10K50/00
- H10K59/10—OLED displays
- H10K59/12—Active-matrix OLED [AMOLED] displays
- H10K59/123—Connection of the pixel electrodes to the thin film transistors [TFT]
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/30—Coordination compounds
- H10K85/341—Transition metal complexes, e.g. Ru(II)polypyridine complexes
- H10K85/342—Transition metal complexes, e.g. Ru(II)polypyridine complexes comprising iridium
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/615—Polycyclic condensed aromatic hydrocarbons, e.g. anthracene
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/615—Polycyclic condensed aromatic hydrocarbons, e.g. anthracene
- H10K85/626—Polycyclic condensed aromatic hydrocarbons, e.g. anthracene containing more than one polycyclic condensed aromatic rings, e.g. bis-anthracene
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/631—Amine compounds having at least two aryl rest on at least one amine-nitrogen atom, e.g. triphenylamine
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/631—Amine compounds having at least two aryl rest on at least one amine-nitrogen atom, e.g. triphenylamine
- H10K85/633—Amine compounds having at least two aryl rest on at least one amine-nitrogen atom, e.g. triphenylamine comprising polycyclic condensed aromatic hydrocarbons as substituents on the nitrogen atom
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/631—Amine compounds having at least two aryl rest on at least one amine-nitrogen atom, e.g. triphenylamine
- H10K85/636—Amine compounds having at least two aryl rest on at least one amine-nitrogen atom, e.g. triphenylamine comprising heteroaromatic hydrocarbons as substituents on the nitrogen atom
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/649—Aromatic compounds comprising a hetero atom
- H10K85/654—Aromatic compounds comprising a hetero atom comprising only nitrogen as heteroatom
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/649—Aromatic compounds comprising a hetero atom
- H10K85/657—Polycyclic condensed heteroaromatic hydrocarbons
- H10K85/6572—Polycyclic condensed heteroaromatic hydrocarbons comprising only nitrogen in the heteroaromatic polycondensed ring system, e.g. phenanthroline or carbazole
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/649—Aromatic compounds comprising a hetero atom
- H10K85/657—Polycyclic condensed heteroaromatic hydrocarbons
- H10K85/6576—Polycyclic condensed heteroaromatic hydrocarbons comprising only sulfur in the heteroaromatic polycondensed ring system, e.g. benzothiophene
-
- H01L2251/5384—
-
- H01L2251/552—
-
- H01L27/3244—
-
- H01L51/0081—
-
- H01L51/5004—
-
- H01L51/5008—
-
- H01L51/5016—
-
- H01L51/506—
-
- H01L51/5096—
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K2101/00—Properties of the organic materials covered by group H10K85/00
- H10K2101/10—Triplet emission
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K2101/00—Properties of the organic materials covered by group H10K85/00
- H10K2101/30—Highest occupied molecular orbital [HOMO], lowest unoccupied molecular orbital [LUMO] or Fermi energy values
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K2101/00—Properties of the organic materials covered by group H10K85/00
- H10K2101/40—Interrelation of parameters between multiple constituent active layers or sublayers, e.g. HOMO values in adjacent layers
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K2101/00—Properties of the organic materials covered by group H10K85/00
- H10K2101/90—Multiple hosts in the emissive layer
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/14—Carrier transporting layers
- H10K50/15—Hole transporting layers
- H10K50/155—Hole transporting layers comprising dopants
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/18—Carrier blocking layers
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/80—Constructional details
- H10K50/805—Electrodes
- H10K50/81—Anodes
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/80—Constructional details
- H10K50/805—Electrodes
- H10K50/82—Cathodes
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K59/00—Integrated devices, or assemblies of multiple devices, comprising at least one organic light-emitting element covered by group H10K50/00
- H10K59/10—OLED displays
- H10K59/12—Active-matrix OLED [AMOLED] displays
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/30—Coordination compounds
- H10K85/321—Metal complexes comprising a group IIIA element, e.g. Tris (8-hydroxyquinoline) gallium [Gaq3]
- H10K85/324—Metal complexes comprising a group IIIA element, e.g. Tris (8-hydroxyquinoline) gallium [Gaq3] comprising aluminium, e.g. Alq3
Definitions
- One or more embodiments of the present invention relate to an organic light-emitting device.
- OLEDs Organic light-emitting devices
- advantages such as wide viewing angles, good contrast, quick response, high brightness, low driving voltage characteristics, and can provide multicolored images.
- a typical organic light-emitting device has a structure including a first electrode, a hole transport region, an emission layer, an electron transport region, and a second electrode that are sequentially stacked on a substrate. Holes injected from the first electrode move to the emission layer via the hole transport region, and electrons injected from the second electrode move to the emission layer via the electron transport region. Carriers (i.e. the holes and electrons) recombine in the emission layer to generate excitons. When the excitons drop from an excited state to a ground state, light is emitted.
- One or more aspects of embodiments of the present invention are directed to a novel organic light-emitting device.
- an organic light-emitting device includes an anode, a cathode, and an organic layer that includes an emission layer and is positioned between the anode and the cathode.
- the organic layer further includes i) a hole transport region between the anode and the emission layer and including at least one selected from a hole injection layer, a hole transport layer, a buffer layer, and an electron blocking layer; and ii) an electron transport region between the emission layer and the cathode and including at least one selected from a hole blocking layer, an electron transport layer, and an electron injection layer;
- a mixed organic layer is positioned between the emission layer and the electron transport region.
- the mixed organic layer includes at least two different compounds, and at least one compound selected from the at least two different compounds has a triplet energy of 2.2 eV or higher.
- the drawing is a schematic view of a structure of an organic light-emitting device according to an embodiment of the present invention.
- an organic light-emitting device includes an anode, a cathode, and an organic layer between the anode and the cathode and including an emission layer (EML).
- EML emission layer
- the organic layer further includes i) a hole transport region between the anode and the EML and including at least one selected from a hole injection layer (HIL), a hole transport layer (HTL), a buffer layer, and an electron blocking layer (EBL); and ii) an electron transport region between the EML and the cathode and including at least one selected from a hole blocking layer (HBL), an electron transport layer (ETL), and an electron injection layer (EIL).
- HIL hole injection layer
- HTL hole transport layer
- EBL electron blocking layer
- EIL electron injection layer
- a mixed organic layer is positioned between the EML and the electron transport region.
- the mixed organic layer includes at least two different compounds, and at least one compound selected from the at least two different compounds has a triplet energy of 2.2 eV or higher.
- Deterioration in performance of an OLED device such as, for example, increase in driving voltage, may be caused by accumulation of holes due to introduction of an additional layer between the EML and the ETL.
- recombination of the holes and electrons may predominantly occur at the side of the EML closer to the anode, where the electrons are accumulated, and as a result, light-emitting lifespan of the organic light-emitting device may deteriorate.
- a compound included in the organic layer of an organic light-emitting device includes an electron withdrawing group (EWG) capable of electron transfer and a hydrocarbon-based ring on a side of an anode from the EML.
- EWG electron withdrawing group
- the organic light-emitting device according to an embodiment of the present invention includes a mixed organic layer including at least two different compounds, and at least one compound selected from the at least two different compounds has a triplet energy of 2.2 eV or greater.
- a triplet energy of the at least one compound may be 2.2 eV or greater, for example, from about 2.2 eV to about 4.0 eV, or from about 2.2 eV to about 3.8 eV.
- the organic light-emitting device according to an embodiment of the present invention may have a low driving voltage, a high efficiency, and a long lifespan.
- the EML may be a phosphorescent EML.
- the EML may be a fluorescent EML.
- the mixed organic layer contacts the EML, and the triplet energy of the at least one compound in the mixed organic layer may be greater than a triplet energy of a dopant of the EML.
- the at least one compound may include an electron-transporting material or a hole-transporting material.
- the at least two different compounds include a hole-transporting compound and an electron-transporting compound, respectively, and a weight ratio of the hole-transporting compound to the electron-transporting compound in the mixed organic layer may be in the range of about 0.1:1 to about 10:1.
- the organic light-emitting device may have a low driving voltage, a high efficiency, and a long lifespan.
- the at least two different compounds may include a hole-transporting compound and an electron-transporting compound, respectively, and an electron affinity (EA1) of the hole-transporting compound may be less than an electron affinity (EA2) of the electron-transporting compound (EA1 ⁇ EA2).
- the electrons injected from the anode and transported to the cathode may mainly pass through an electron-transporting material having a relatively large electron affinity, with some of the electrons being blocked by the hole-transporting material, which is additionally included in the cathode.
- electrons function as main carriers, and thus electon leakage may occur.
- the hole-transporting material may block some of the electrons in the mixed organic layer, thus contributing to balancing out the charges in the organic light-emitting device.
- the at least two different compounds may include at least two different.
- the EML may be a phosphorescent EML and may include an Ir, Pt, Cu, or Os-complex as a dopant.
- a thickness of the mixed organic layer may be about 5 ⁇ to about 400 ⁇ .
- a thickness of the mixed organic layer may be about 5 ⁇ to about 40 ⁇ .
- the triplet energy of a mixed organic layer material responsible for the blocking function of the mixed organic layer is mainly determined by a backbone structure of the mixed organic layer material.
- the triplet energy is transferred to whichever structure in each part of the backbone of the mixed organic layer material has the lowest triplet energy.
- the triplet energy of the mixed organic layer material needs to be high, and may be about 2.2 eV or greater.
- An example of the mixed organic layer material having a backbone structure with a triplet energy of about 2.2 eV or greater may be benzene (3.66 ev), phenathrene (2.70 ev), naphthalene (2.63 ev), chrysene (2.48 ev), fluorene (2.94 ev), triphenylene (2.90 ev), fluoranthene (2.30 ev), carbazole (3.18 ev), dibenzofuran (2.97 ev), dibenzothiophene (2.99 ev), phenanthroline (2.75 ev), or benzoimidazole (3.31 ev).
- the mixed organic layer material according to an embodiment of the present invention may be a compound with a high triplet energy or a compound with a high triplet energy backbone structure.
- a high triplet energy (T1) backbone structure may be one of structures below, but is not limited thereto:
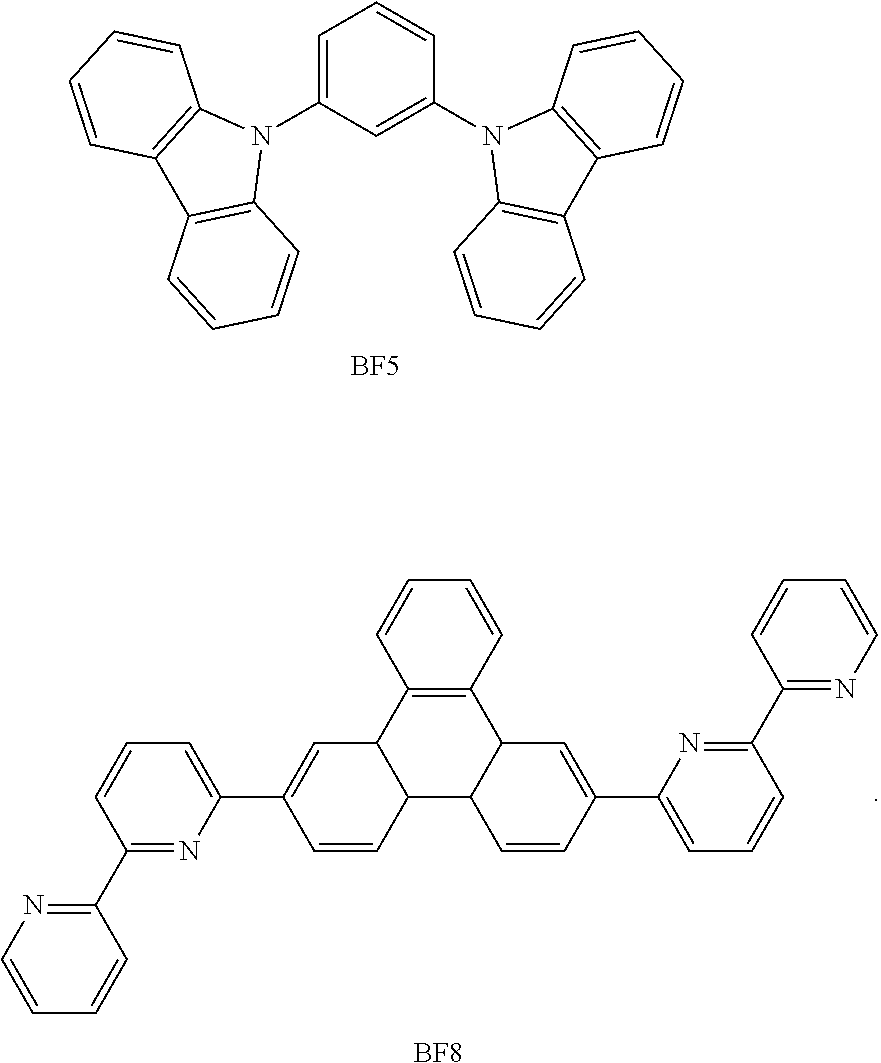
- the mixed organic layer may include at least two different compounds selected from compounds below:
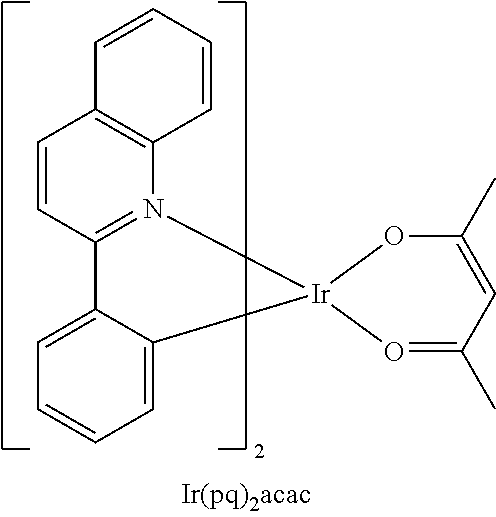
- the EML may include BD, Ir(ppy) 3 , or Ir(pq) 2 acac as a dopant, but the dopant is not limited thereto:
- the EML may include at least one compound selected from compounds below as a host, but the host is not limited thereto:
- a C 1 -C 60 alkyl group may refer to a monovalent linear or branched aliphatic hydrocarbon group.
- Non-limiting examples of the C 1 -C 60 alkyl group include a methyl group, an ethyl group, a propyl group, an isobutyl group, a sec-butyl group, a tert-butyl group, a pentyl group, an iso-amyl group, and a hexyl group.
- a C 1 -C 60 alkylene group may refer to a divalent group that has the same structure as the C 1 -C 60 alkyl group.
- a C 1 -C 60 alkoxy group may refer to a monovalent group having a formula of —OA 101 (where A 101 is the C 1 -C 60 alkyl group).
- a 101 is the C 1 -C 60 alkyl group.
- Non-limiting examples of the C 1 -C 60 alkoxy group include a methoxy group, an ethoxy group, and an isopropyloxy group.
- a C 2 -C 60 alkenyl group may refer to a hydrocarbon chain having at least one carbon-carbon double bond at one or more positions along a carbon chain of the C 2 -C 60 alkyl group.
- the C 2 -C 60 alkenyl group may include a terminal alkene and/or an internal alkene (e.g. in the middle or at an end of the C 2 -C 60 alkyl group).
- Non-limiting examples of the C 2 -C 60 alkenyl group include an ethenyl group, a propenyl group, and a butenyl group.
- a C 2 -C 60 alkenylene group may refer to a divalent group that has the same structure as the C 2 -C 60 alkenyl group.
- a C 2 -C 60 alkynyl group may refer to a hydrocarbon chain having at least one carbon-carbon triple bond at one or more positions along a carbon chain of the C 2 -C 60 alkyl group.
- the C 2 -C 60 alkynyl group may include a terminal alkyne and/or an internal alkyne (e.g. in the middle or at an end of the C 2 -C 60 alkyl group).
- Non-limiting examples of the C 2 -C 60 alkynyl group include an ethynyl group and a propynyl group.
- a C 2 -C 60 alkynylene group may refer to a divalent group that has the same structure as the C 2 -C 60 alkynyl group.
- a C 3 -C 10 cycloalkyl group may refer to a C 3 -C 10 monovalent saturated hydrocarbon monocyclic group.
- Non-limiting examples of the C 3 -C 10 cycloalkyl group include a cyclopropyl group, a cyclobutyl group, a cyclopentyl group, a cyclohexyl group, and a cycloheptyl group.
- a C 3 -C 10 cycloalkylene group may refer to a divalent group that has the same structure as the C 3 -C 10 cycloalkyl group.
- the C 2 -C 10 heterocycloalkyl group may refer to a C 2 -C 10 monovalent monocyclic group including at least one hetero atom selected from N, O, P, and S as a ring-forming atom, and carbon atoms as remaining ring-forming atoms.
- Non-limiting examples of the C 2 -C 10 heterocycloalkyl group include a tetrahydrofuranyl group and a tetrahydrothiophenyl group.
- a C 2 -C 10 heterocycloalkylene group may refer to a divalent group that has the same structure as the C 2 -C 10 heterocycloalkyl group.
- a C 3 -C 10 cycloalkenyl group may refer to a C 3 -C 10 monovalent monocyclic group that has at least one double bond in the ring, but does not have aromaticity.
- Non-limiting examples of the C 3 -C 10 cycloalkenyl group include a cyclopentyl group, a cyclohexenyl group, and a cycloheptenyl group.
- a C 3 -C 10 cycloalkenylene group may refer to a divalent group that has the same structure as the C 3 -C 10 cycloalkenyl group.
- a C 2 -C 10 heterocycloalkenyl group may refer to a C 2 -C 10 monovalent monocyclic group including at least one hetero atom selected from N, O, P, and S as a ring-forming atom and at least one double bond in the ring.
- Non-limiting examples of the C 2 -C 10 heterocycloalkenyl group include a 2,3-hydrofuranyl group and a 2,3-hydrothiophenyl group.
- a C 2 -C 10 heterocycloalkenylene group may refer to a divalent group that has the same structure as the C 2 -C 10 heterocycloalkenyl group.
- a C 6 -C 60 aryl group may refer to a monovalent group having a C 6 -C 60 carbocyclic aromatic system
- a C 6 -C 60 arylene group may refer to a divalent group that has a C 6 -C 60 carbocyclic aromatic system.
- Non-limiting examples of the C 6 -C 60 aryl group include a phenyl group, a naphthyl group, an anthracenyl group, a phenanthrenyl group, a pyrenyl group, and a chrysenyl group.
- the C 6 -C 60 aryl group and/or the C 6 -C 60 arylene group include at least two rings, the rings may be fused to each other.
- a C 2 -C 60 heteroaryl group may refer to a monovalent group having a C 2 -C 60 carbocyclic aromatic system and including at least one heteroatom selected from N, O, P, and S as a ring-forming atom, and carbon atoms as the remaining ring-forming atoms
- a C 2 -C 60 heteroarylene group may refer to a divalent group having a C 2 -C 60 carbocyclic aromatic system and including at least one heteroatom selected from N, O, P, and S as a ring-forming atom, and carbon atoms as the remaining ring-forming atoms.
- Non-limiting examples of the C 2 -C 60 heteroaryl group include a pyridinyl group, a pyrimidinyl group, a pyrazinyl group, a pyridazinyl group, a triazinyl group, a quinolinyl group, and an isoquinolinyl group.
- the C 2 -C 60 heteroaryl group and/or a C 2 -C 60 heteroarylene group include at least two rings, the rings may be fused to each other.
- a C 6 -C 60 aryloxy group may refer to a —OA 102 group (where A 102 is the C 6 -C 60 aryl group), and a C 6 -C 60 arylthio group may refer to a —SA 103 group (where A 103 is the C 6 -C 60 aryl group).
- a monovalent non-aromatic condensed polycyclic group may refer to a monovalent group that has at least two rings that are condensed to each other, each ring including only carbon atoms as ring-forming atoms (e.g., 8 to 60 carbon atoms), and does not have overall aromaticity.
- Non-limiting examples of the non-aromatic condensed polycyclic group include a fluorenyl group.
- a divalent non-aromatic condensed polycyclic group may refer to a divalent group that has the same structure as the monovalent non-aromatic condensed polycyclic group.
- a monovalent non-aromatic heterocondensed polycyclic group may refer to a monovalent group that has at least two rings that are condensed to each other, each ring including a heteroatom selected from N, O, P, and S as a ring-forming atom and carbon atoms as remaining ring-forming atoms (e.g., 2 to 60 carbon atoms), and does not have overall aromaticity.
- Non-limiting examples of the non-aromatic heterocondensed polycyclic group include a carbazolyl group.
- a divalent non-aromatic heterocondensed polycyclic group may refer to a divalent group that has the same structure as the monovalent non-aromatic heterocondensed polycyclic group.
- a deuterium —F, —Cl, —Br, —I, a hydroxyl group, a cyano group, a nitro group, an amino group, an amidino group, a hydrazine group, a hydrazone group, a carboxylic acid or a salt thereof, a sulfonic acid or a salt thereof, a phosphoric acid or a salt thereof, a C 1 -C 60 alkyl group, a C 2 -C 60 alkenyl group, a C 2 -C 60 alkynyl group, and/or a C 1 -C 60 alkoxy group;
- Q 11 to Q 17 , Q 21 to Q 27 , and Q 31 to Q 37 are each independently selected from a hydrogen, —F, —Cl, —Br, —I, a hydroxyl group, a cyano group, a nitro group, an amino group, an amidino group, a hydrazine group, a hydrazone group, a carboxylic acid or a salt thereof, a sulfonic acid or a salt thereof, a phosphoric acid or a salt thereof, a C 1 -C 60 alkyl group, a C 2 -C 60 alkenyl group, a C 2 -C 60 alkynyl group, a C 1 -C 60 alkoxy group, a C 3 -C 10 cycloalkyl group, a C 2 -C 10 heterocycloalkyl group, a C 3 -C 10 cycloalkenyl group, a C 2 -C 10 heterocycloalkenyl group,
- a deuterium —F, —Cl, —Br, —I, a hydroxyl group, a cyano group, a nitro group, an amino group, an amidino group, a hydrazine group, a hydrazone group, a carboxylic acid or a salt thereof, a sulfonic acid or a salt thereof, a phosphoric acid or a salt thereof, a C 1 -C 60 alkyl group, a C 2 -C 60 alkenyl group, a C 2 -C 60 alkynyl group, and/or a C 1 -C 60 alkoxy group;
- a cyclopentyl group a cyclohexyl group, a cycloheptyl group, a cyclopentenyl group, a cycloheptenyl group, a cyclohexenyl group, a phenyl group, a pentalenyl group, an indenyl group, a naphthyl group, an azulenyl group, a heptalenyl group, an indacenyl group, an acenaphthyl group, a fluorenyl group, a spiro-fluorenyl group, a benzofluorenyl group, a dibenzofluorenyl group, a phenalenyl group, a phenanthrenyl group, an anthracenyl group, a fluoranthenyl group, a triphenylenyl group, a pyrenyl group, a chrysenyl
- a cyclopentyl group a cyclohexyl group, a cycloheptyl group, a cyclopentenyl group, a cycloheptenyl group, a cyclohexenyl group, a phenyl group, a pentalenyl group, an indenyl group, a naphthyl group, an azulenyl group, a heptalenyl group, an indacenyl group, an acenaphthyl group, a fluorenyl group, a spiro-fluorenyl group, a benzofluorenyl group, a dibenzofluorenyl group, a phenalenyl group, a phenanthrenyl group, an anthracenyl group, a fluoranthenyl group, a triphenylenyl group, a pyrenyl group, a chrysenyl
- Q 11 to Q 17 , Q 21 to Q 27 , and Q 31 to Q 37 are each independently selected from a hydrogen, a deuterium, —F, —Cl, —Br, —I, a hydroxyl group, a cyano group, a nitro group, an amino group, an amidino group, a hydrazine group, a hydrazone group, a carboxylic acid or a salt thereof, a sulfonic acid or a salt thereof, a phosphoric acid or a salt thereof, a C 1 -C 60 alkyl group, a C 2 -C 60 alkenyl group, a C 2 -C 60 alkynyl group, a C 1 -C 60 alkoxy group, a cyclopentyl group, a cyclohexyl group, a cycloheptyl group, a cyclopentenyl group, a cycloheptenyl group, a cyclohex
- Ph refers to a phenyl group
- Me refers to a methyl group
- Et refers to an ethyl group
- ter-Bu or “But” used herein refers to a tert-butyl group.
- an organic layer includes at least one compound of Formula X” used herein may refer to an organic layer) including one compound of Formula X, or two or more different compounds of Formula X.
- organic layer used herein may refer to a single layer and/or a plurality of layers between the first electrode and the second electrode in the organic light-emitting device.
- a material included in the organic layer is not limited to an organic material.
- the drawing schematically illustrates a cross-sectional view of an organic light-emitting device 10 according to an embodiment of the present invention.
- the organic light-emitting device 10 includes a first electrode 110 , an organic layer 150 , and a second electrode 190 .
- a substrate may be positioned on a first side (e.g. lower side) of the first electrode 110 or a second side (e.g. upper side) of the second electrode 190 .
- the substrate may be a glass substrate or a transparent plastic substrate having good mechanical strength, thermal stability, transparency, surface smoothness, ease of handling, and water resistance.
- the first electrode 110 may be formed by applying a first electrode material on the substrate by, for example, deposition or sputtering.
- the first electrode material may be selected from materials having a high work function and capable of easily injecting the holes.
- the first electrode 110 may be a reflective electrode, a semi-transparent electrode, or a transparent electrode.
- Non-limiting examples of the first electrode material may include indium-tin oxide (ITO), indium-zinc-oxide (IZO), tin oxide (SnO 2 ), and zinc oxide (ZnO).
- the first electrode 110 is a semi-transparent electrode or a reflective electrode
- at least one selected from magnesium (Mg), aluminum (Al), aluminum-lithium (Al—Li), a Calcium (Ca), magnesium-indium (Mg—In), and magnesium-silver (Mg—Ag) may be selected as the first electrode material.
- the first electrode 110 may have a single-layered structure or a multi-layered structure including at least two layers. In one embodiment, the first electrode 110 may have a three-layered structure of ITO/Ag/ITO, but the structure of the first electrode 110 is not limited thereto.
- the organic layer 150 is positioned on the first electrode 110 and includes an EML.
- the organic layer 150 may further include a hole transport region between the first electrode 110 and the EML, an electron transport region between the EML and the second electrode 190 , and a mixed organic layer between the EML and the electron transport region.
- the hole transport region may include at least one selected from an HIL, an HTL, a buffer layer, and an EBL
- the electron transport region may include at least one selected from an HBL, an ETL, and an EIL, but the hole transport region and the electron transport region are not limited thereto.
- the structure of the hole transport region may include a single layer structure formed of one material, a single layer structure formed of multiple different materials, or multiple layers structure formed of multiple different materials.
- the hole transport region may have a single layer structure formed of multiple different materials, for example, HIL/HTL, HIL/HTL/buffer layer, HIL/buffer layer, HTL/buffer layer, or HIL/HTL/EBL sequentially stacked on the first electrode 110 , but the structure of the hole transport region is not limited thereto.
- the HIL may be formed on the first electrode 110 using (utilizing) various methods such as, for example, vacuum deposition, spin coating, casting, Langmuir-Blodgett (LB) deposition, inkjet printing, laser printing, or laser induced thermal imaging (LITI).
- various methods such as, for example, vacuum deposition, spin coating, casting, Langmuir-Blodgett (LB) deposition, inkjet printing, laser printing, or laser induced thermal imaging (LITI).
- the deposition temperature may be about 100 to about 500° C.
- the degree of vacuum may be about 10 ⁇ 8 to about 10 ⁇ 3 torr
- the deposition speed may be about 0.01 to about 100 ⁇ /sec, depending on the kind of compound for forming the HIL and the desired structure of the HIL.
- the coating speed may be about 2,000 rpm to about 5,000 rpm and the heat treatment temperature may be about 80° C. to about 200° C., depending on the kind of compound for forming the HIL and the desired structure of the HIL.
- the HTL may be formed on the first electrode 110 or on the HIL using (utilizing) various methods such as, for example, vacuum deposition, spin coating, casting, LB deposition, inkjet printing, laser printing, or LITI.
- various methods such as, for example, vacuum deposition, spin coating, casting, LB deposition, inkjet printing, laser printing, or LITI.
- the deposition conditions and the coating conditions for forming the HTL may be similar to the deposition conditions and the coating conditions for forming the HIL.
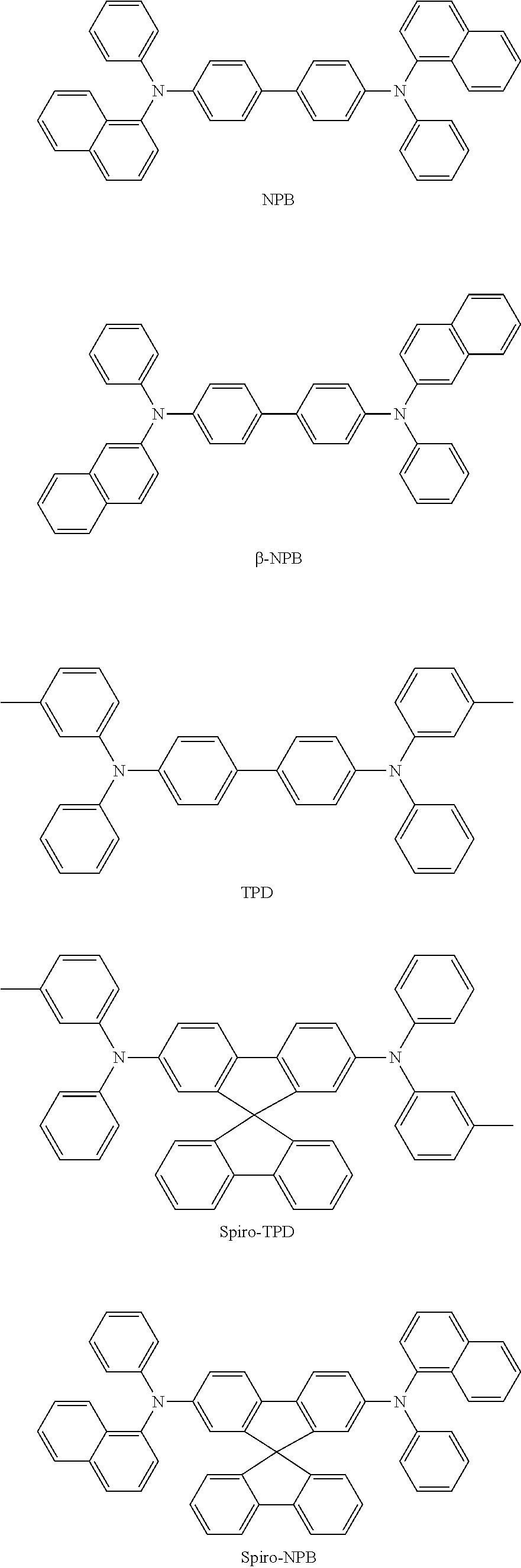
- the hole transport region may include at least one of m-MTDATA, TDATA, 2-TNATA, NPB, ⁇ -NPB, TPD, Spiro-TPD, Spiro-NPB, methylated NPB, TAPC, HMTPD, 4,4′,4′′-tris(N-carbazolyl)triphenylamine) (TCTA), polyaniline/Dodecylbenzenesulfonic acid (Pani/DBSA), poly(3,4-ethylenedioxythiophene)/poly(4-styrenesulfonate) (PEDOT/PSS), polyaniline/camphor sulfonic acid (Pani/CSA), polyaniline)/poly(4-styrenesulfonate (PANI/PSS), a compound represented by Formula 201, and a compound represented by Formula 202:
- L 201 to L 205 may each independently be the same as the definition of L 1 as described in the present specification;
- xa1 to xa4 are each independently selected from 0, 1, 2, and 3;
- xa5 is selected from 1, 2, 3, 4, and 5;
- R 201 to R 204 may be each independently selected from a substituted or unsubstituted C 3 -C 10 cycloalkyl group, a substituted or unsubstituted C 2 -C 10 heterocycloalkyl group, a substituted or unsubstituted C 3 -C 10 cycloalkenyl group, a substituted or unsubstituted C 2 -C 10 heterocycloalkenyl group, a substituted or unsubstituted C 6 -C 60 aryl group, a substituted or unsubstituted C 6 -C 60 aryloxy group, a substituted or unsubstituted C 6 -C 60 arylthio group, a substituted or unsubstituted C 2 -C 60 heteroaryl group, a substituted or unsubstituted monovalent non-aromatic condensed polycyclic group, and/or a substituted or unsubstituted monovalent non-aromatic hetero
- L 201 to L 205 are each independently selected from
- xa1 to xa4 are each independently selected from 0, 1, and 2;
- xa5 is selected from 1, 2, and 3;
- R 201 to R 204 are each independently selected from, but are not limited to,
- the compound represented by Formula 201 may be represented by Formula 201A below, but is not limited thereto:
- the compound represented by Formula 201 may be represented by Formula 201A-1:
- the compound represented by Formula 202 may be represented by Formula 202A, but is not limited thereto:
- L 201 to L 203 , xa1 to xa3, xa5, and R 202 to R 204 may be as defined in the present specification
- definition of R 211 and R 212 may be the same as the definition of R 203
- R 213 to R 216 may be each independently selected from, but are not limited to, a hydrogen, a deuterium, —F, —Cl, —Br, —I, a hydroxyl group, a cyano group, a nitro group, an amino group, an amidino group, a hydrazine group, a hydrazone group, a carboxylic acid or a salt thereof, a sulfonic acid or a salt thereof, a phosphoric acid or a salt thereof, a C 1 -C 60 alkyl group, a C 2 -C 60 alkenyl group, a C 2 -C 60 alkynyl group, a C 1 -C 60 alkoxy group, a C 3
- L 201 to L 203 are each independently selected from
- R 203 , R 211 , and R 212 are each independently selected from
- R 213 and R 214 are each independently selected from
- R 215 and R 216 are each independently selected from
- xa5 is selected from 1 and 2.
- R 213 and R 214 may be linked to each other to form a saturated or unsaturated ring.
- the compound represented by Formula 201 and the compound represented by Formula 202 may each independently include at least one of Compounds HT1 to HT20 below, but the compound represented by Formula 201 and the compound represented by Formula 202 are not limited thereto:
- a thickness of the hole transport region may be about 100 ⁇ to about 10,000 ⁇ , for example, about 100 ⁇ to about 1,000 ⁇ .
- a thickness of the HIL may be about 100 ⁇ to about 9,950 ⁇ , for example, about 100 ⁇ to about 950 ⁇
- a thickness of the HTL may be about 50 ⁇ to about 2,000 ⁇ , for example, about 100 ⁇ to about 1,500 ⁇ .
- the organic light-emitting device may have satisfactory hole transporting properties without a substantial increase in driving voltage.
- the hole transport region may further include a charge-generating material, in addition to the materials described above, to improve conductivity.
- the charge-generating material may be homogenously or inhomogenously dispersed in the hole transport region.
- the charge-generating material may be, for example, a p-dopant.
- the p-dopant may be one of a quinone derivative, a metal oxide, and/or a cyano group-containing compound, but the p-dopant is not limited thereto.
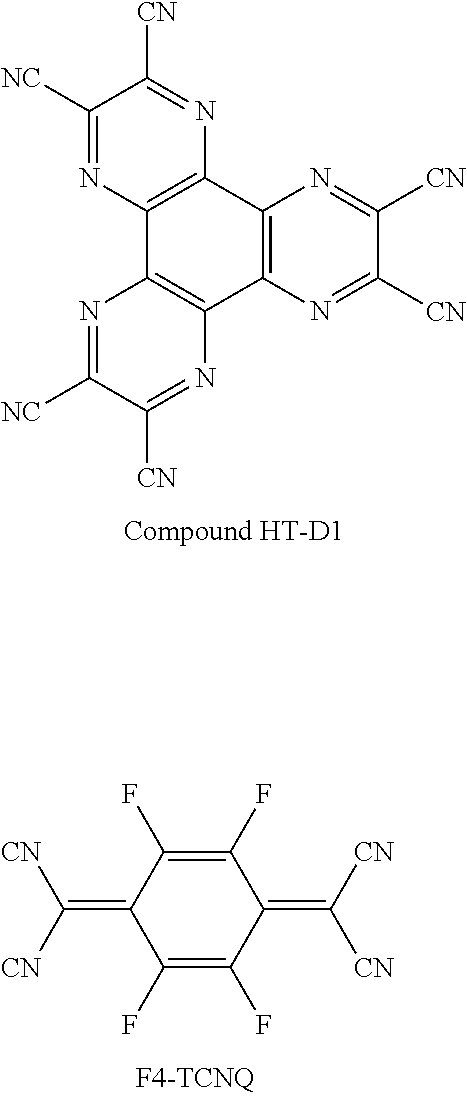
- Non-limiting examples of the p-dopant may include a quinone derivative, such as tetracyanoquinonedimethane (TCNQ) or 2,3,5,6-tetrafluoro-tetracyano-1,4-benzoquinondimethane (F4-TCNQ); a metal oxide, such as a tungsten oxide or a molybden oxide; and Compound HT-D1 below:
- the hole transport region may further include at least one of a buffer layer and an EBL, in addition to the HIL and the HTL.
- the buffer layer may increase light-emitting efficiency by compensating an optical resonance distance according to the wavelength of light emitted from the EML.
- the buffer layer may include a material included in the hole transport region.
- the EBL may block injection of electrons from the electron transport region.
- the HTL may include a first hole transport layer and a second hole transport layer, and the first hole transport layer and the second hole transport layer may be formed of the same material or of different from each other materials.
- the EML may be formed on the first electrode 110 or on the hole transport region using (utilizing) various methods such as, for example, vacuum deposition, spin coating, casting, LB deposition, inkjet printing, laser printing, or LITI.
- various methods such as, for example, vacuum deposition, spin coating, casting, LB deposition, inkjet printing, laser printing, or LITI.
- the deposition conditions and the coating conditions for forming the EML may be similar to the deposition conditions and the coating conditions for forming the HIL.
- the EML may be patterned into individual sub-pixels, such as a red EML, a green EML, and a blue EML.
- the EML may have a stacked structure of the red EML, the green EML, and the blue EML, or a single layer structure including a red light-emitting material, a green light-emitting material, and a blue light-emitting material formed as a single layer and capable of emitting white light.
- the EML may include a host and a dopant.
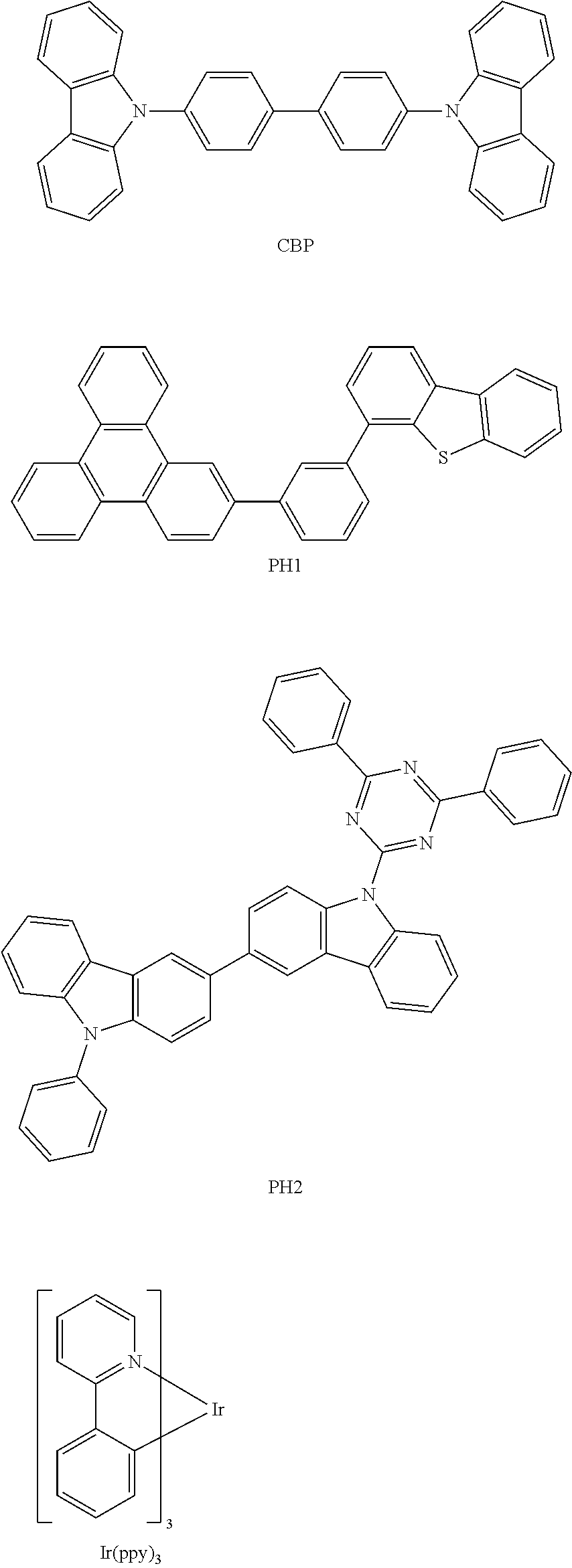
- Non-limiting examples of the host may include at least one of TPBi, TBADN, ADN (herein, also referred to as “DNA”), CBP, CDBP, and TCP:
- the host may include a compound represented by Formula 301:
- Ar 301 is selected from
- L 301 may be the same as the definition of L 201 as described in the present specification;
- R 301 is selected from
- xb1 is selected from 0, 1, 2, and 3;
- xb2 is selected from 1, 2, 3, and 4.
- L 301 is selected from
- a phenylene group a naphthylene group, a fluorenylene group, a spiro-fluorenylene group, a benzofluorenylene group, a dibenzofluorenylene group, a phenanthrenylene group, an anthracenylene group, a pyrenylene group, and/or a chrysenylene group; and/or
- R 301 is selected from
- a phenyl group a naphthyl group, a fluorenyl group, a spiro-fluorenyl group, a benzofluorenyl group, a dibenzofluorenyl group, a phenanthrenyl group, an anthracenyl group, a pyrenyl group, and/or a chrysenyl group; and/or
- the host may include a compound represented by Formula 301A:
- the compound represented by Formula 301A may include at least one of Compounds H1 to H42, but the compound represented by Formula 301A is not limited thereto:
- the host may include at least one of Compounds H43 to H49, but the host is not limited thereto:
- the dopant may include at least one of a fluorescent dopant and a phosphorescent dopant.
- the phosphorescent dopant may include an organic metal complex represented by Formula 401:
- M is selected from iridium (Ir), platinum (Pt), osmium (Os), titanium (Ti), zirconium (Zr), hafnium (Hf), europium (Eu), terbium (Tb), and/or thulium (Tm);
- X 401 to X 404 are each independently a nitrogen atom or a carbon atom;
- rings A 401 and A 402 are each independently selected from a substituted or unsubstituted benzene, a substituted or unsubstituted naphthalene, a substituted or unsubstituted fluorene, a substituted or unsubstituted spiro-fluorene, a substituted or unsubstituted indene, a substituted or unsubstituted pyrrole, a substituted or unsubstituted thiophene, a substituted or unsubstituted furan, a substituted or unsubstituted imidazole, a substituted or unsubstituted pyrazole, a substituted or unsubstituted thiazole, a substituted or unsubstituted isothiazole, a substituted or unsubstituted oxazole, a substituted or unsubstituted isoxazole, a substituted or unsubstit
- substituted benzene substituted naphthalene, substituted fluorene, substituted spiro-fluorene, substituted indene, substituted pyrrole, substituted thiophene, substituted furan, substituted imidazole, substituted pyrazole, substituted thiazole, substituted isothiazole, substituted oxazole, substituted isoxazole, substituted pyridine, substituted pyrazine, substituted pyrimidine, substituted pyridazine, substituted quinoline, substituted isoquinoline, substituted benzoquinoline, substituted quinoxaline, substituted quinazoline, substituted carbazole, substituted benzoimidazole, substituted benzofuran, substituted benzothiophene, substituted isobenzothiophene, substituted benzoxazole, substituted isobenzoxazole, substituted triazole, substituted oxadiazole, substituted
- a deuterium —F, —Cl, —Br, —I, a hydroxyl group, a cyano group, a nitro group, an amino group, an amidino group, a hydrazine group, a hydrazone group, a carboxylic acid or a salt thereof, a sulfonic acid or a salt thereof, a phosphoric acid or a salt thereof, a C 1 -C 60 alkyl group, a C 2 -C 60 alkenyl group, a C 2 -C 60 alkynyl group, and/or a alkoxy group;
- Q 401 to Q 407 , Q 411 to Q 417 , and Q 421 to Q 427 are defined as Q 11 to Q 17 , Q 21 to Q 27 , and Q 31 to Q 37 above;
- L 401 is an organic ligand
- xc1 is selected from 1, 2, and 3;
- xc2 is selected from 0, 1, 2, and 3.
- L 401 is a monovalent, divalent, or trivalent organic ligand.
- L 401 may be selected from a halogen ligand, such as Cl or F, a diketone ligand, such as acetylacetonate, 1,3-diphenyl-1,3-propanedionate, 2,2,6,6-tetramethyl-3,5-heptanedionate, or hexafluoroacetonate, a carboxylic acid ligand, such as picolinate, dimethyl-3-pyrazolecarboxylate, or benzoate, a carbon monoxide ligand, an isonitrile ligand, a cyano ligand, and/or a phosphorus ligand, such as phosphine or phosphite, but L 401 is not limited thereto.
- a halogen ligand such as Cl or F
- a diketone ligand such as acetylacetonate, 1,3
- the at least two substituents of A 401 may be linked to each other to form a saturated or unsaturated ring.
- the at least two substituents of A 402 may be linked to each to and form a saturated or unsaturated ring.
- a 401 and A 402 of one ligand may be linked to A 401 and A 402 of an adjacent ligand, respectively, directly (e.g. via a single bond)or via a connection group (e.g., a C 1 -C 5 alkylene group, —N(R′)— (where, R′ is C 1 -C 10 alkyl group or a C 6 -C 20 aryl group), or —C( ⁇ O)—).
- a connection group e.g., a C 1 -C 5 alkylene group, —N(R′)— (where, R′ is C 1 -C 10 alkyl group or a C 6 -C 20 aryl group), or —C( ⁇ O)—).
- the phosphorescent dopant may be selected from Compounds PD1 to PD74 below, but is not limited thereto:
- the phosphorescent dopant may include PtOEP illustrated below:
- the fluorescent dopant may include at least one of DPVBi, BDAVBi, TBPe, DCM, DCJTB, Coumarin 6, and C545T below:
- the fluorescent dopant may include a compound represented by Formula 501 below:
- Ar 501 is selected from
- definitions of L 501 to L 503 may be each independently the same as the definition of L 201 ;
- R 501 and R 502 are each independently selected from
- xd1 to xd3 are each independently selected from 0, 1, 2, and 3;
- xb4 is selected from 1, 2, 3, and 4.
- the fluorescent host may include at least one of Compounds FD1 to FD8, but is not limited thereto:
- the dopant may be present in the EML in an amount of about 0.01 part to about 15 parts by weight, based on about 100 parts by weight of the host, but the amount of the dopant is not limited thereto.
- a thickness of the EML may be about 100 ⁇ to about 1,000 ⁇ , for example, about 200 ⁇ to about 600 ⁇ . When the thickness of the EML is within any of these ranges, light-emitting properties of the organic light-emitting device may be improved, without a substantial increase in driving voltage.
- the mixed organic layer may be on the EML.
- the mixed organic layer may be formed on the EML using (utilizing) various methods such as, for example, vacuum deposition, spin coating, casting, LB deposition, inkjet printing, laser printing, or LITI.
- various methods such as, for example, vacuum deposition, spin coating, casting, LB deposition, inkjet printing, laser printing, or LITI.
- the deposition conditions and the coating conditions for forming the mixed organic layer may be similar to the deposition conditions and the coating conditions for forming the HIL.
- a compound for forming the mixed organic layer may be as described above.
- a thickness of the mixed organic layer may be about 5 ⁇ to about 400 ⁇ , for example, about 10 ⁇ to about 40 ⁇ . When the thickness of the mixed organic layer is within any of these ranges, light-emitting properties of the organic light-emitting device may be improved, without a substantial increase in driving voltage.
- a weight ratio of the hole-transporting compound to the electron-transporting compound in the mixed organic layer may be in the range of about 0.1:1 to about 10:1, but the respective amounts of the hole-transporting compound and the electron-transporting compound in the mixed organic layer are not limited thereto.
- An electron transport region may be positioned on the mixed organic layer.
- the electron transport region may include at least one selected from an HBL, an ETL, and an EIL, but is not limited thereto.
- the electron transport region may have a structure of ETL/EIL or EBL/ETL/EIL, sequentially stacked on the EML or on the mixed organic layer, but the structure of the electron transport region is not limited thereto.
- the organic layer 150 of the organic light-emitting device 10 includes an electron transport region between the EML and the second electrode 190 .
- the electron transport region may include at least one of an ETL and an EIL.
- the ETL may include at least one selected from bathocuproine (BCP), bathophenanthroline (Bphen), Alq 3 , Balq, TAZ, and NTAZ below:
- the ETL may include at least one compound selected from a group of compounds represented by Formula 601 and Formula 602:
- Ar 601 is selected from
- L 601 may be the same as the definition of L 201 above;
- E 601 is selected from
- xe1 is selected from 0, 1, 2, and 3;
- xe2 is selected from 1, 2, 3, and 4.
- X 611 is N or C-(L 611 ) xe611 -R 611
- X 612 is N or C-(L 612 ) xe612 -R 612
- X 613 is N or C-(L 613 ) xe613 -R 613
- at least one of X 611 to X 613 is N;
- each of L 611 to L 616 may be the same as the definition of L 201 as described in the present specification;
- R 611 to R 616 are each independently selected from
- xe611 to xe616 are each independently selected from 0, 1, 2, and 3.
- the compound represented by Formula 601 and the compound represented by Formula 602 may be each independently selected from compounds ET1 to ET15:
- a thickness of the ETL may be about 100 ⁇ to about 1,000 ⁇ , for example, about 150 ⁇ to about 500 ⁇ . When the thickness of the ETL is within any of these ranges, electron transporting properties of the organic light-emitting device may be improved, without a substantial increase in driving voltage.
- the ETL may further include a metal-containing material, in addition to the materials described above.
- the metal-containing material may include a Li-complex.
- the Li-complex may include, for example, compound ET-D1 (lithium quinolate (LiQ)) or ET-D2:
- the electron transport region may include an HBL.
- the HBL may prevent triplet excitons or holes from diffusing into the ETL.
- the HBL may be formed on the EML using (utilizing) various methods such as, for example, vacuum deposition, spin coating, casting, LB deposition, inkjet printing, laser printing, or LITI.
- various methods such as, for example, vacuum deposition, spin coating, casting, LB deposition, inkjet printing, laser printing, or LITI.
- the deposition conditions and the coating conditions for forming the HBL may be similar to the deposition conditions and the coating conditions for forming the HIL.
- the HBL may include, for example, at least one of BCP and Bphen below, but is not limited thereto:
- a thickness of the HBL may be about 20 ⁇ to about 1,000 ⁇ , for example, about 30 ⁇ to about 300 ⁇ . When the thickness of the HBL is within any of these ranges, the organic light-emitting device may exhibit good hole blocking properties, without a substantial increase in driving voltage.
- the electron transport region may include an ETL.
- the ETL may be formed on the EML or the HBL using (utilizing) various methods such as, for example, vacuum deposition, spin coating, casting, LB deposition, inkjet printing, laser printing, or LITI.
- the deposition conditions and the coating conditions for forming the ETL may be similar to the deposition conditions and the coating conditions for forming the HIL.
- the electron transport region may include an EIL that may facilitate the injection of electrons from the second electrode 190 .
- the EIL may be formed on the ETL using (utilizing) various methods such as, for example, vacuum deposition, spin coating, casting, LB deposition, inkjet printing, laser printing, or LITI.
- various methods such as, for example, vacuum deposition, spin coating, casting, LB deposition, inkjet printing, laser printing, or LITI.
- the deposition conditions and the coating conditions for forming the EIL may be similar to the deposition conditions and the coating conditions for forming the HIL.
- the EIL may include at least one selected from LiF, NaCl, CsF, Li 2 O, BaO, and LiQ, but is not limited thereto.
- a thickness of the EIL may be about 1 ⁇ to about 100 ⁇ , for example, about 3 ⁇ to about 90 ⁇ . When the thickness of the EIL is within any of these ranges, the organic light-emitting device may exhibit good electron injecting properties, without a substantial increase in driving voltage.
- the second electrode 190 is on the organic layer 150 .
- the second electrode 190 may be a cathode, which is an electron injection electrode.
- a material for forming the second electrode 190 may include a metal, an alloy, an electric conducting compound, all having a low work function, and/or a mixture thereof.
- the second electrode 190 may be a thin film formed of lithium (Li), magnesium (Mg), aluminum (Al), aluminum-lithium (Al—Li), calcium (Ca), magnesium-indium (Mg—In), and/or magnesium-silver (Mg—Ag), but the second electrode 190 is not limited thereto.
- ITO and/or IZO may be used (utilized) as a material for forming the second electrode 190 .
- the second electrode 190 may be a reflective electrode, a semitransparent electrode, or a transparent electrode.
- the organic layer of the organic light-emitting device according to embodiments of the present invention may be formed by a deposition method of the compound according to embodiments of the present invention, or by a wet method in which the organic light-emitting device is coated with the compound according to embodiments of the present invention that is first prepared as a solution.
- the organic light-emitting device may be included in various types (kinds) of flat panel displays, for example, a passive matrix organic light-emitting display apparatus and/or an active matrix organic light-emitting display apparatus.
- the first electrode located on the side of the substrate is a pixel electrode and may be electrically connected to a source electrode or a drain electrode of a thin film transistor.
- the organic light-emitting device may be included in a flat panel display that may display images on both surfaces.
- the organic light-emitting device has been described with reference to the drawing, the organic light-emitting device of embodiments of the present invention is not limited thereto.
- a transparent electrode of an indium tin oxide (ITO) having a thickness of 120 nm was formed on a glass substrate to prepare a cathode. Then, ultrasonic cleaning and pretreatment (UV-0 3 treatment and heat-treatment) were performed on the resulting cathode.
- ITO indium tin oxide
- Compound HTM (illustrated below) was deposited at a thickness of about 120 nm as an HTL on the pretreated cathode. Then, compound MADN (illustrated below) as a host, and compound BD as a dopant material, were co-deposited on the HTL in a total amount of 5% to form an EML having a thickness of about 30 nm. Compound BF1 (illustrated below) and compound BF9 (illustrated below) were deposited on the EML at a ratio of 1:1 to form a buffer having a thickness of about 20 nm, and then Alq was deposited on the buffer as an ETL having a thickness of about 20 nm.
- lithium fluoride was deposited on the ETL to form an EIL having a thickness of about 1 nm, and subsequently, aluminum was deposited at a thickness of about 200 nm on the EIL, thereby manufacturing an organic light-emitting device.
- IP Ionization Potential
- Electron affinities were calculated using the following equation:
- IP is the ionization potential calculated in (1) and Eg is the energy gap calculated in (2).
- ⁇ edg denotes a wavelength value at a point of intersection of a tangent and a horizontal axis, where the tangent is according to a slope of a short wavelength side of a phosphorescent spectrum.
- Organic light-emitting devices were manufactured as in Example 1-1, except that the EML, the host, the dopant, and the buffer layer were formed as shown in Table 3, and the dopant material Ir(ppy) 3 was deposited at a concentration of 10%, instead of 5%.
- the host is formed of two different compounds, a weight ratio of the compounds is 1:1.
- Organic light-emitting devices were manufactured as in Example 1-1, except that the EML, the host, the dopant, and the buffer layer were formed as shown in Table 4, and the dopant material Ir(pq)2acac was deposited at a concentration of 5%.
- the organic light-emitting devices prepared in Examples 1-1 to 9-3 showed improved efficiency and lifespan characteristics compared to those of the organic light-emitting devices prepared in Comparative Examples 1 to 9.
- the organic light-emitting device may have a low driving voltage, a high efficiency, and a long lifespan.
Landscapes
- Physics & Mathematics (AREA)
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Spectroscopy & Molecular Physics (AREA)
- Optics & Photonics (AREA)
- Crystallography & Structural Chemistry (AREA)
- Inorganic Chemistry (AREA)
- Electromagnetism (AREA)
- Microelectronics & Electronic Packaging (AREA)
- Electroluminescent Light Sources (AREA)
Abstract
Description
- This application is a continuation of U.S. application Ser. No. 15/425,324, filed on Feb. 6, 2017, which is a continuation of U.S. application Ser. No. 14/497,267, filed on Sep. 25, 2014, now U.S. Pat. No. 9,564,598, which claims priority to and the benefit of Korean Patent Application No. 10-2014-0053616, filed on May 2, 2014, the entire content of all of which is incorporated herein by reference.
- One or more embodiments of the present invention relate to an organic light-emitting device.
- Organic light-emitting devices (OLEDs) are self-emitting devices that have advantages such as wide viewing angles, good contrast, quick response, high brightness, low driving voltage characteristics, and can provide multicolored images.
- A typical organic light-emitting device has a structure including a first electrode, a hole transport region, an emission layer, an electron transport region, and a second electrode that are sequentially stacked on a substrate. Holes injected from the first electrode move to the emission layer via the hole transport region, and electrons injected from the second electrode move to the emission layer via the electron transport region. Carriers (i.e. the holes and electrons) recombine in the emission layer to generate excitons. When the excitons drop from an excited state to a ground state, light is emitted.
- One or more aspects of embodiments of the present invention are directed to a novel organic light-emitting device.
- Additional aspects will be set forth in part in the description which follows and, in part, will be apparent from the description, or may be learned by practice of the presented embodiments.
- According to one or more embodiments of the present invention, an organic light-emitting device includes an anode, a cathode, and an organic layer that includes an emission layer and is positioned between the anode and the cathode. The organic layer further includes i) a hole transport region between the anode and the emission layer and including at least one selected from a hole injection layer, a hole transport layer, a buffer layer, and an electron blocking layer; and ii) an electron transport region between the emission layer and the cathode and including at least one selected from a hole blocking layer, an electron transport layer, and an electron injection layer;
- In one embodiment, a mixed organic layer is positioned between the emission layer and the electron transport region. The mixed organic layer includes at least two different compounds, and at least one compound selected from the at least two different compounds has a triplet energy of 2.2 eV or higher.
- These and/or other aspects will become apparent and more readily appreciated from the following description of the embodiments, taken in conjunction with the accompanying drawings in which:
- The drawing is a schematic view of a structure of an organic light-emitting device according to an embodiment of the present invention.
- Reference will now be made to embodiments, examples of which are illustrated in the accompanying drawings, wherein like reference numerals refer to like elements throughout the specification. In this regard, the present embodiments may have different forms and should not be construed as being limited to the descriptions set forth herein. Accordingly, the embodiments are merely described below, by referring to the figures, to explain aspects of the present description. As used herein, the term “and/or” includes any and all combinations of one or more of the associated listed items. Expressions such as “at least one selected from,” when preceding a list of elements, modify the entire list of elements and do not modify the individual elements of the list. Further, the use of “may” when describing embodiments of the present invention refers to “one or more embodiments of the present invention.”
- According to an embodiment of the present invention, an organic light-emitting device includes an anode, a cathode, and an organic layer between the anode and the cathode and including an emission layer (EML).
- The organic layer further includes i) a hole transport region between the anode and the EML and including at least one selected from a hole injection layer (HIL), a hole transport layer (HTL), a buffer layer, and an electron blocking layer (EBL); and ii) an electron transport region between the EML and the cathode and including at least one selected from a hole blocking layer (HBL), an electron transport layer (ETL), and an electron injection layer (EIL).
- In one embodiment, a mixed organic layer is positioned between the EML and the electron transport region. The mixed organic layer includes at least two different compounds, and at least one compound selected from the at least two different compounds has a triplet energy of 2.2 eV or higher.
- Deterioration in performance of an OLED device, such as, for example, increase in driving voltage, may be caused by accumulation of holes due to introduction of an additional layer between the EML and the ETL. In addition, recombination of the holes and electrons may predominantly occur at the side of the EML closer to the anode, where the electrons are accumulated, and as a result, light-emitting lifespan of the organic light-emitting device may deteriorate.
- In general, a compound included in the organic layer of an organic light-emitting device includes an electron withdrawing group (EWG) capable of electron transfer and a hydrocarbon-based ring on a side of an anode from the EML. In contrast, the organic light-emitting device according to an embodiment of the present invention includes a mixed organic layer including at least two different compounds, and at least one compound selected from the at least two different compounds has a triplet energy of 2.2 eV or greater.
- A triplet energy of the at least one compound may be 2.2 eV or greater, for example, from about 2.2 eV to about 4.0 eV, or from about 2.2 eV to about 3.8 eV. When the triplet energy of the at least one compound is within any of these ranges, the organic light-emitting device according to an embodiment of the present invention may have a low driving voltage, a high efficiency, and a long lifespan.
- In one embodiment, the EML may be a phosphorescent EML.
- In one embodiment, the EML may be a fluorescent EML.
- In one embodiment, the mixed organic layer contacts the EML, and the triplet energy of the at least one compound in the mixed organic layer may be greater than a triplet energy of a dopant of the EML.
- In one embodiment, the at least one compound may include an electron-transporting material or a hole-transporting material.
- In one embodiment, the at least two different compounds include a hole-transporting compound and an electron-transporting compound, respectively, and a weight ratio of the hole-transporting compound to the electron-transporting compound in the mixed organic layer may be in the range of about 0.1:1 to about 10:1. When the weight ratio of the hole-transporting compound to the electron-transporting compound is within this range, the organic light-emitting device may have a low driving voltage, a high efficiency, and a long lifespan.
- In one embodiment, the at least two different compounds may include a hole-transporting compound and an electron-transporting compound, respectively, and an electron affinity (EA1) of the hole-transporting compound may be less than an electron affinity (EA2) of the electron-transporting compound (EA1<EA2).
- When the electron affinity (EA1) of the hole-transporting compound is less than the electron affinity (EA2) of the electron-transporting compound, the electrons injected from the anode and transported to the cathode may mainly pass through an electron-transporting material having a relatively large electron affinity, with some of the electrons being blocked by the hole-transporting material, which is additionally included in the cathode.
- In an organic light-emitting device, electrons function as main carriers, and thus electon leakage may occur. However, when a hole-transporting material that blocks electrons is introduced between the EML and the ETL, the hole-transporting material may block some of the electrons in the mixed organic layer, thus contributing to balancing out the charges in the organic light-emitting device.
- In one embodiment, the at least two different compounds may include at least two different.
- In one embodiment, the EML may be a phosphorescent EML and may include an Ir, Pt, Cu, or Os-complex as a dopant.
- In one embodiment, a thickness of the mixed organic layer may be about 5 Å to about 400 Å. For example, a thickness of the mixed organic layer may be about 5 Å to about 40 Å.
- According to an embodiment of the present invention, the triplet energy of a mixed organic layer material responsible for the blocking function of the mixed organic layer is mainly determined by a backbone structure of the mixed organic layer material. When the mixed organic layer is adjacent to the triplet excitons generated in the EML, the triplet energy is transferred to whichever structure in each part of the backbone of the mixed organic layer material has the lowest triplet energy. Thus, in order to confine the generated excitons in the EML, the triplet energy of the mixed organic layer material needs to be high, and may be about 2.2 eV or greater.
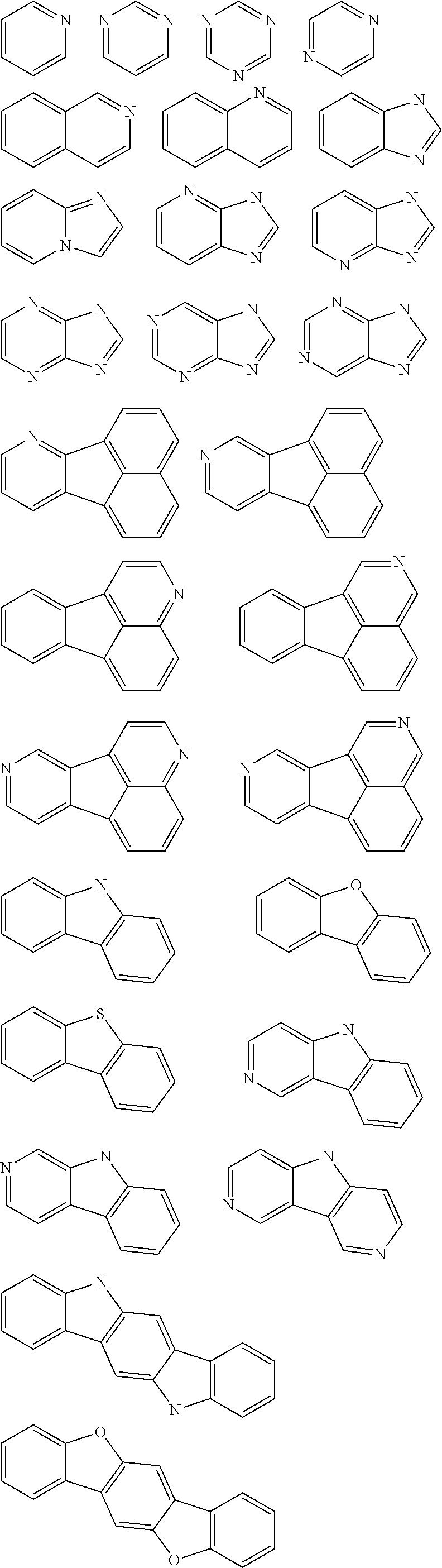
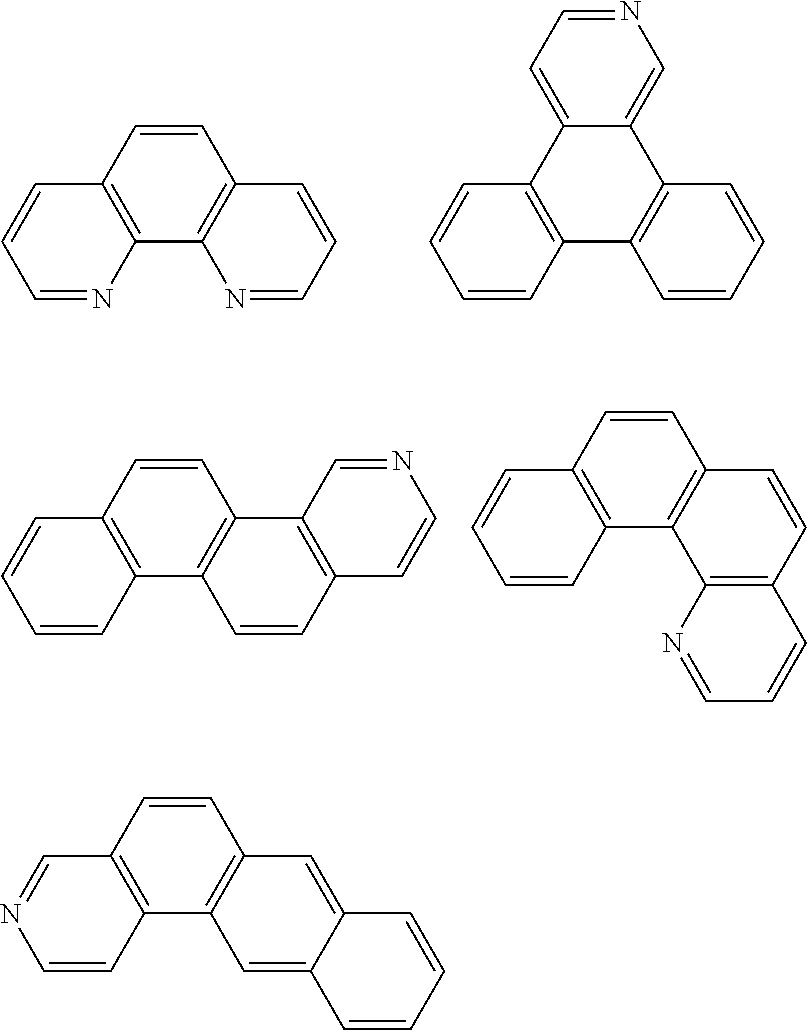
- An example of the mixed organic layer material having a backbone structure with a triplet energy of about 2.2 eV or greater may be benzene (3.66 ev), phenathrene (2.70 ev), naphthalene (2.63 ev), chrysene (2.48 ev), fluorene (2.94 ev), triphenylene (2.90 ev), fluoranthene (2.30 ev), carbazole (3.18 ev), dibenzofuran (2.97 ev), dibenzothiophene (2.99 ev), phenanthroline (2.75 ev), or benzoimidazole (3.31 ev).
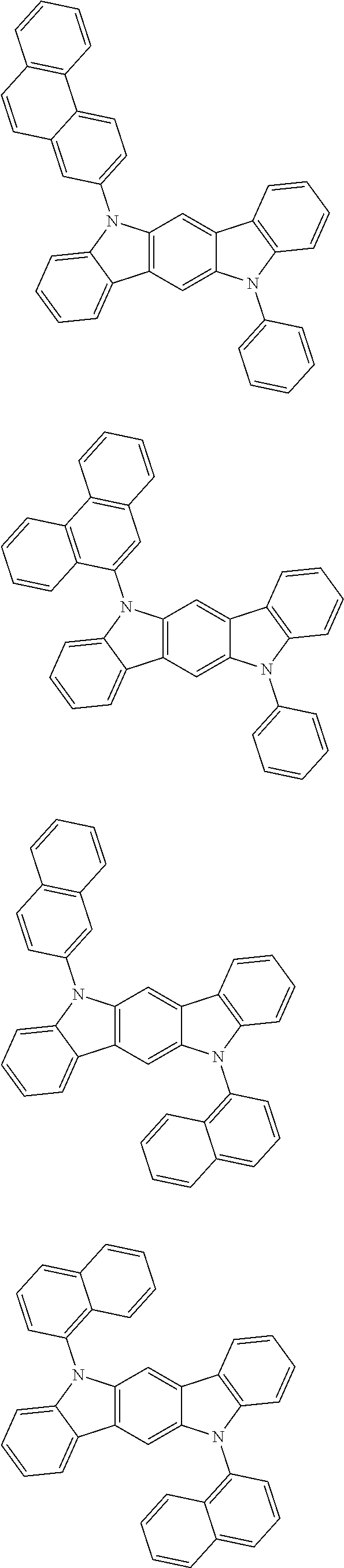
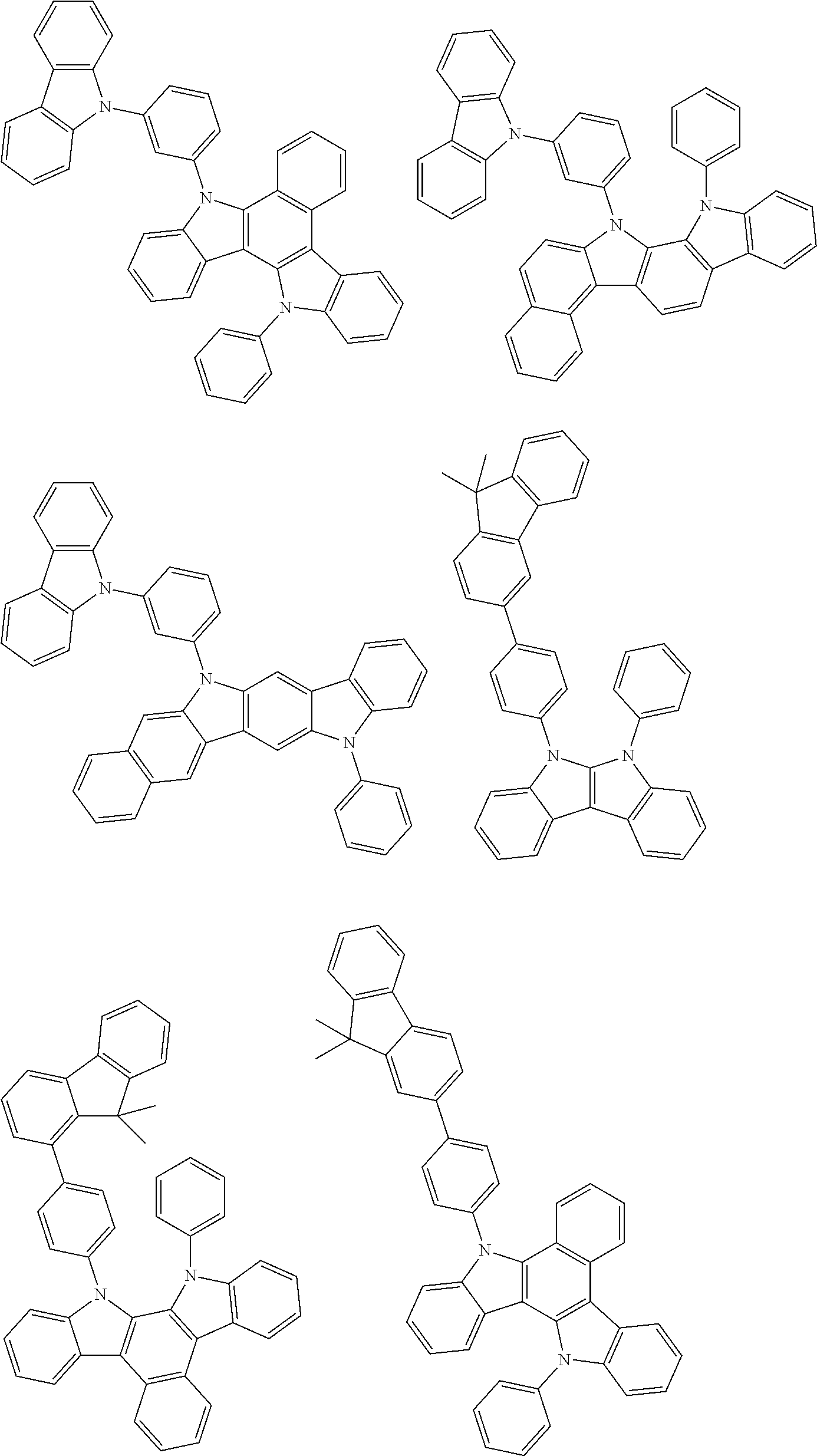
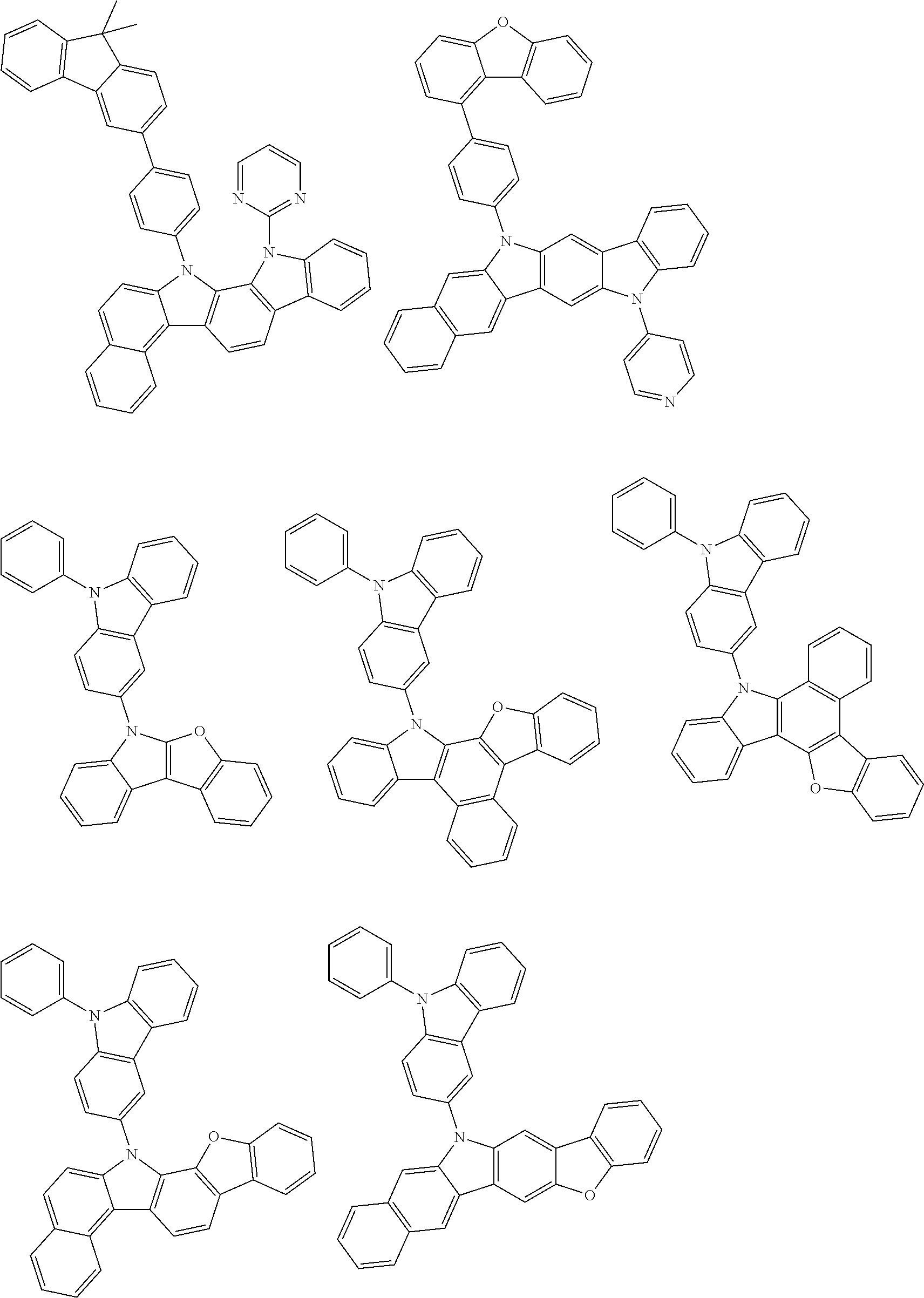
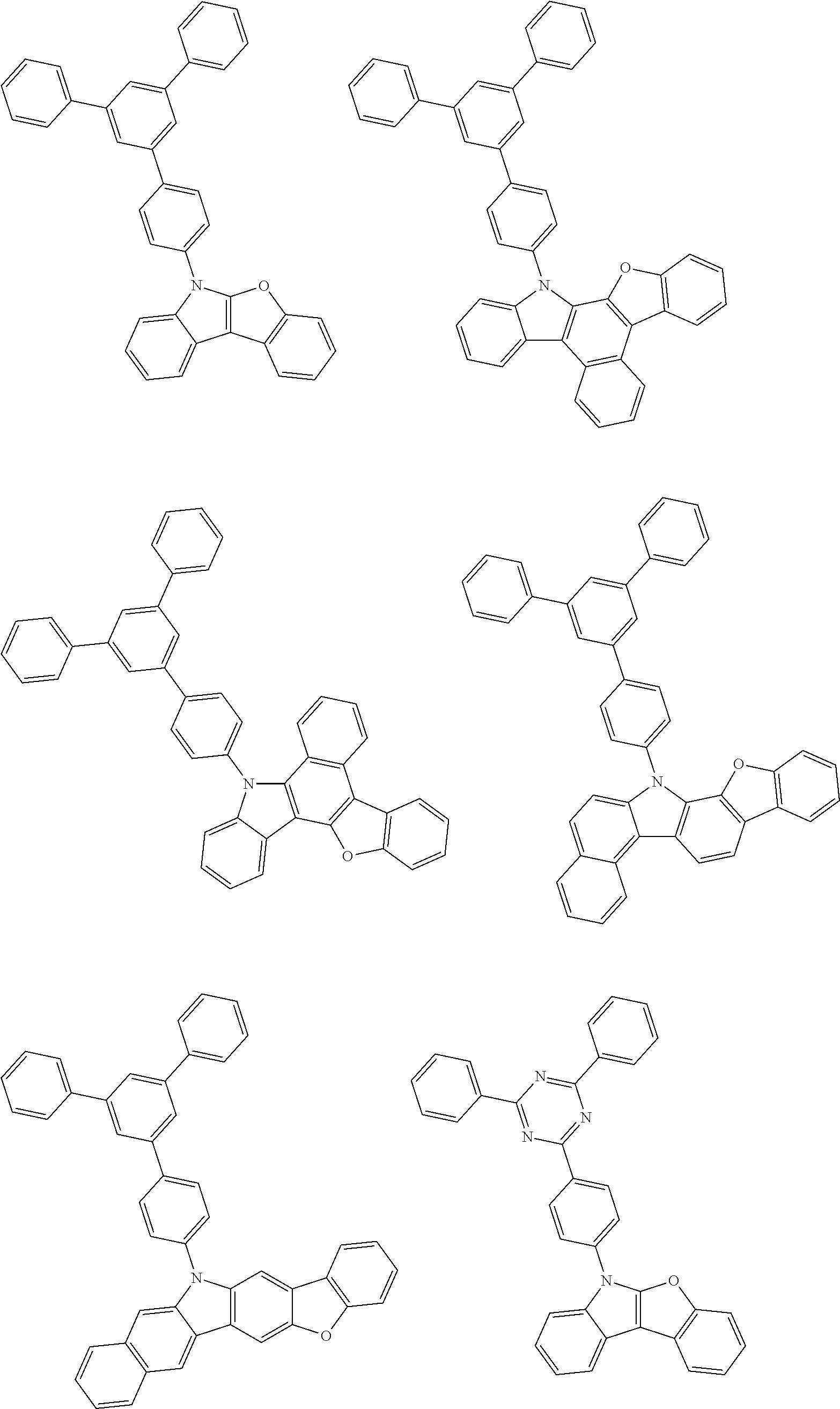
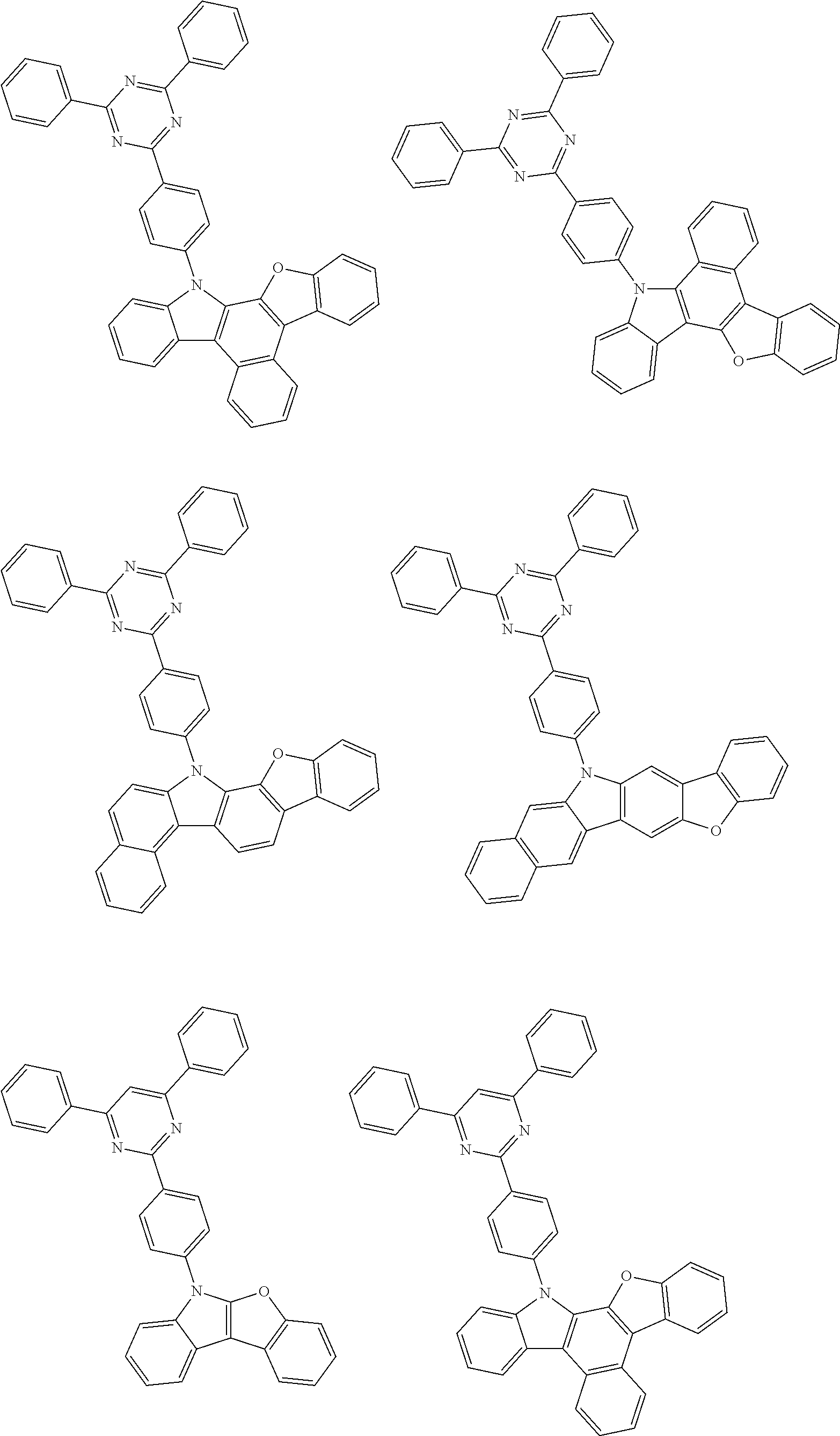
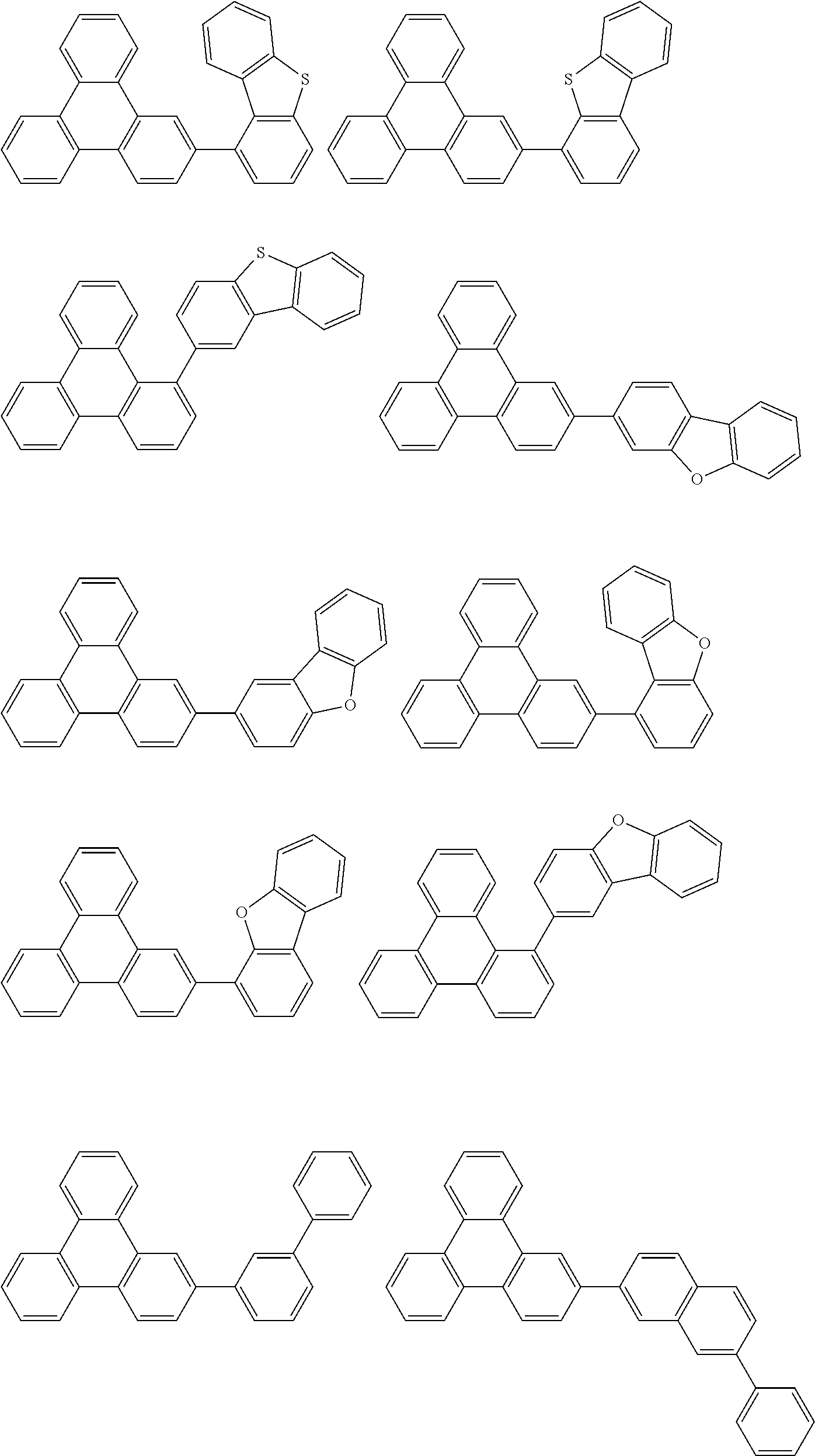
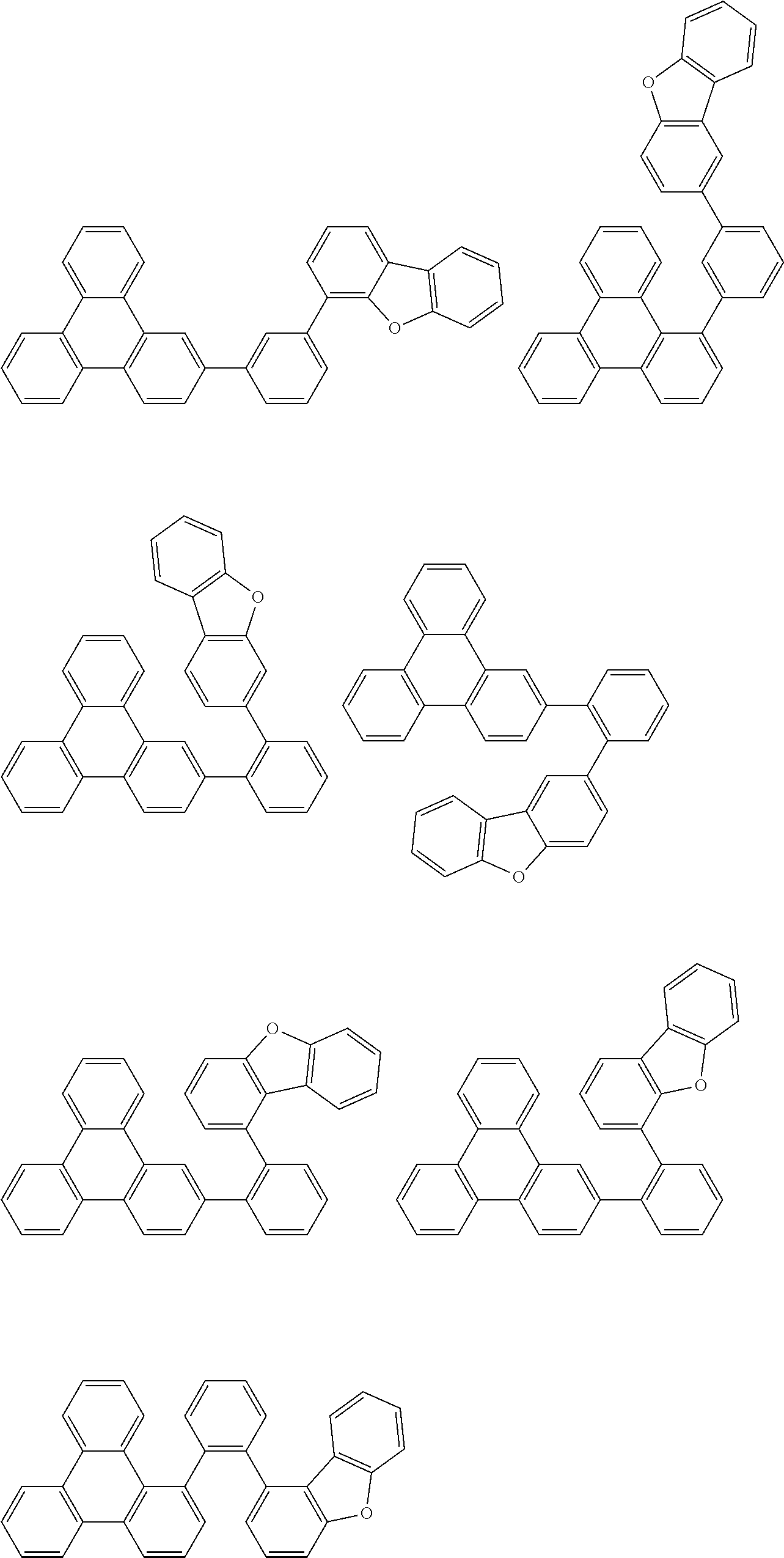
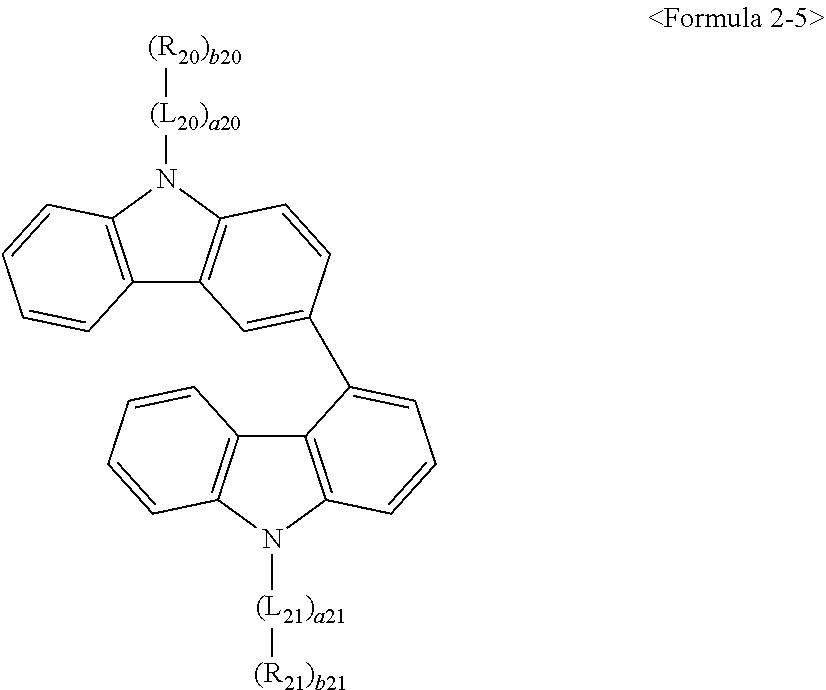
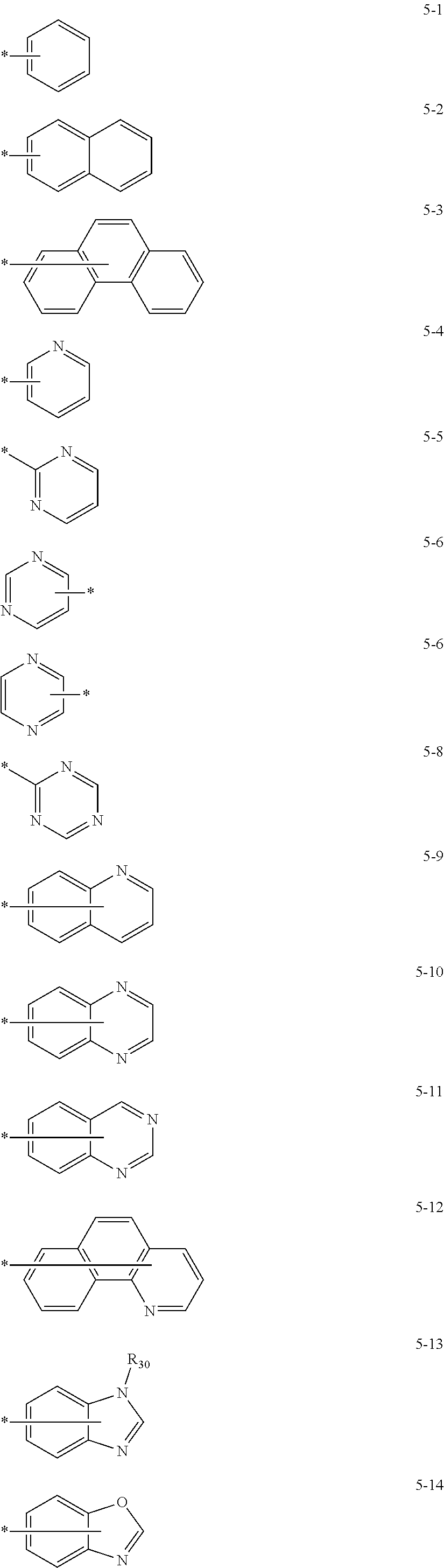
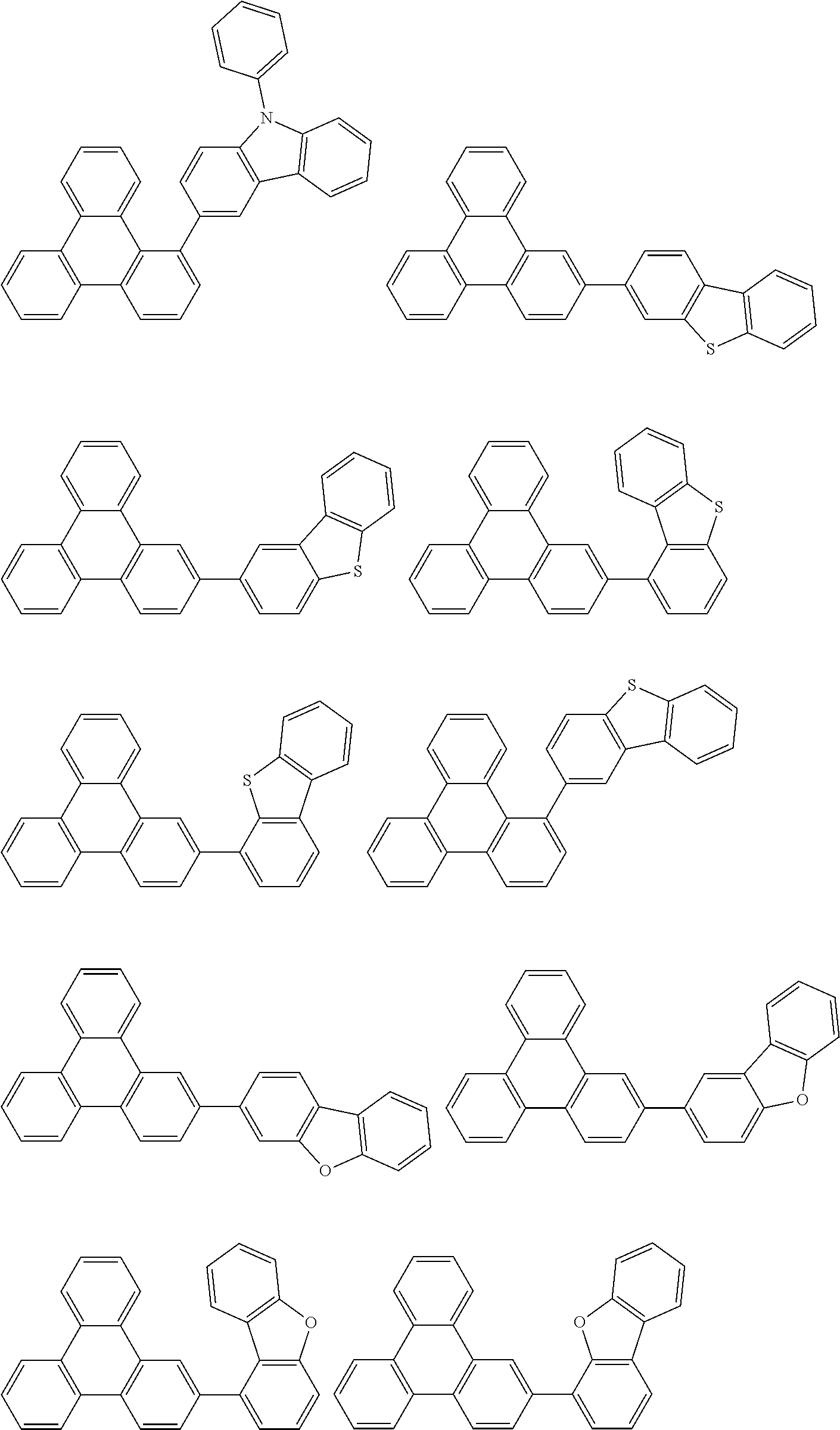
- Thus, the mixed organic layer material according to an embodiment of the present invention may be a compound with a high triplet energy or a compound with a high triplet energy backbone structure. A high triplet energy (T1) backbone structure may be one of structures below, but is not limited thereto:
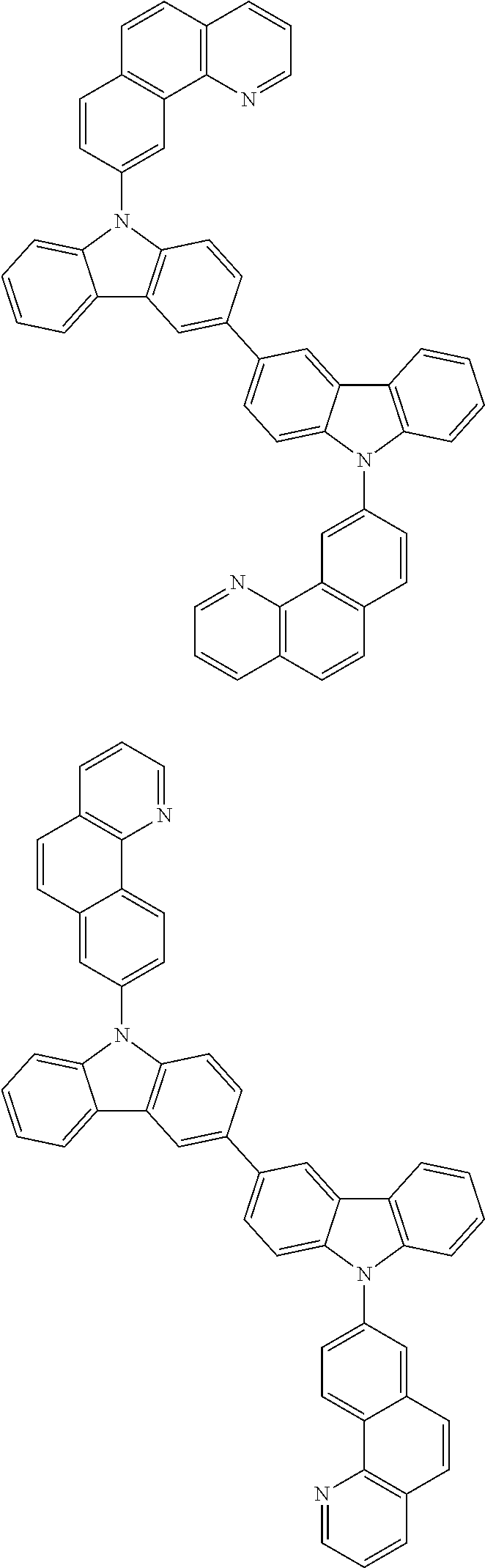
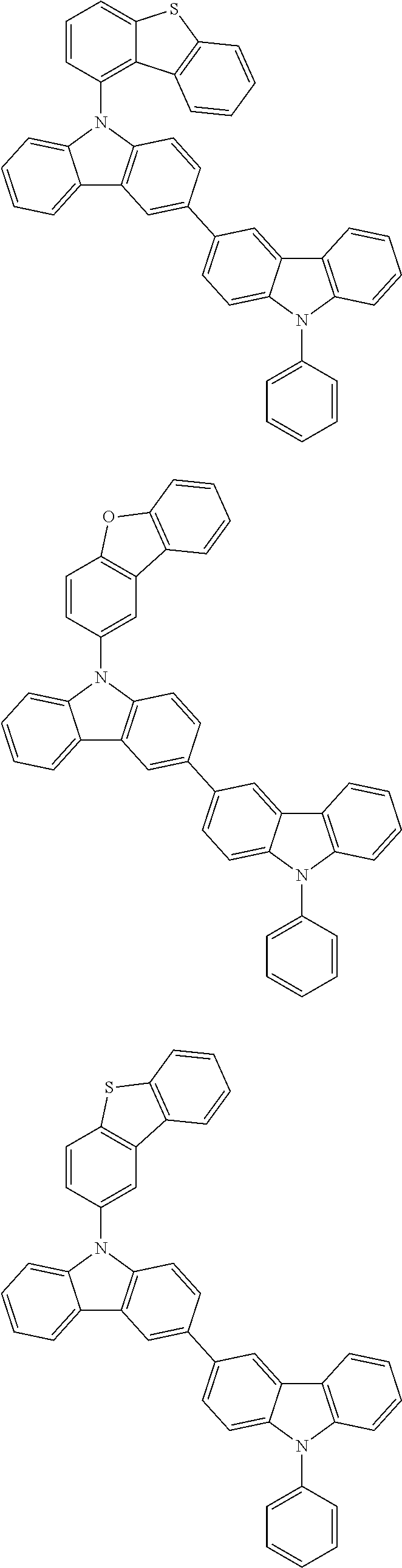
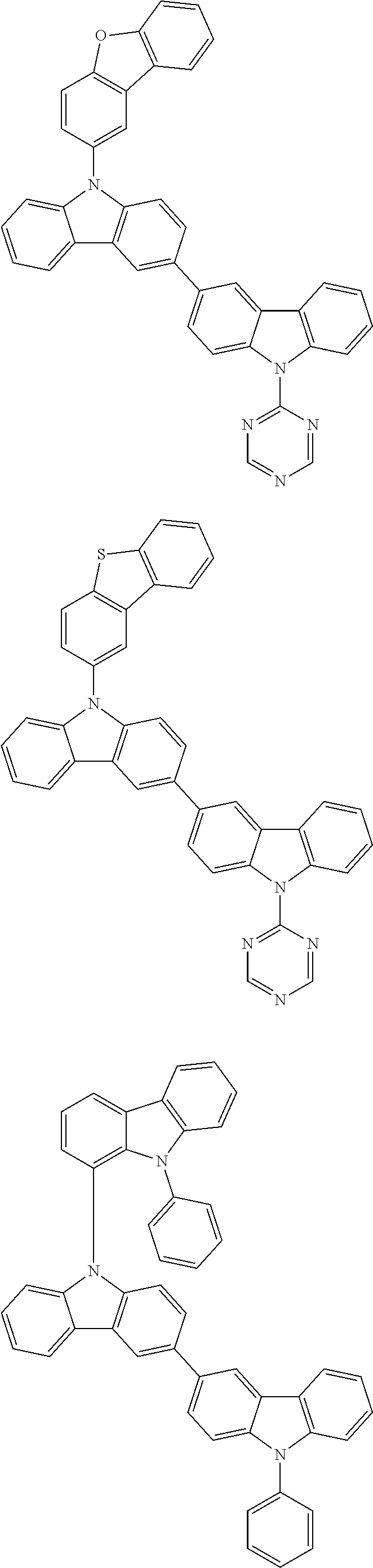
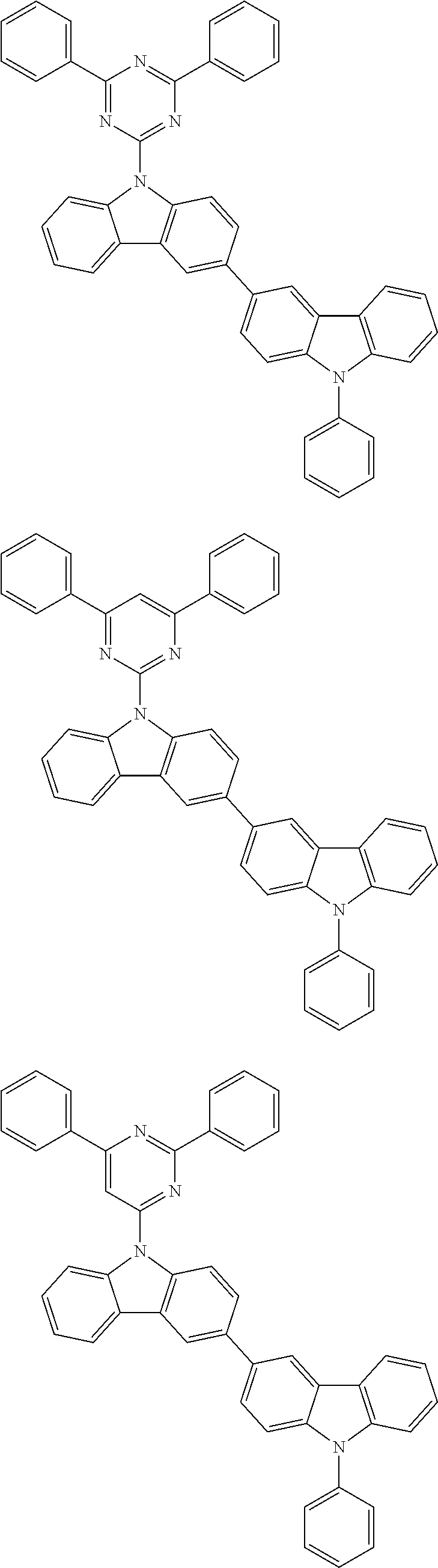
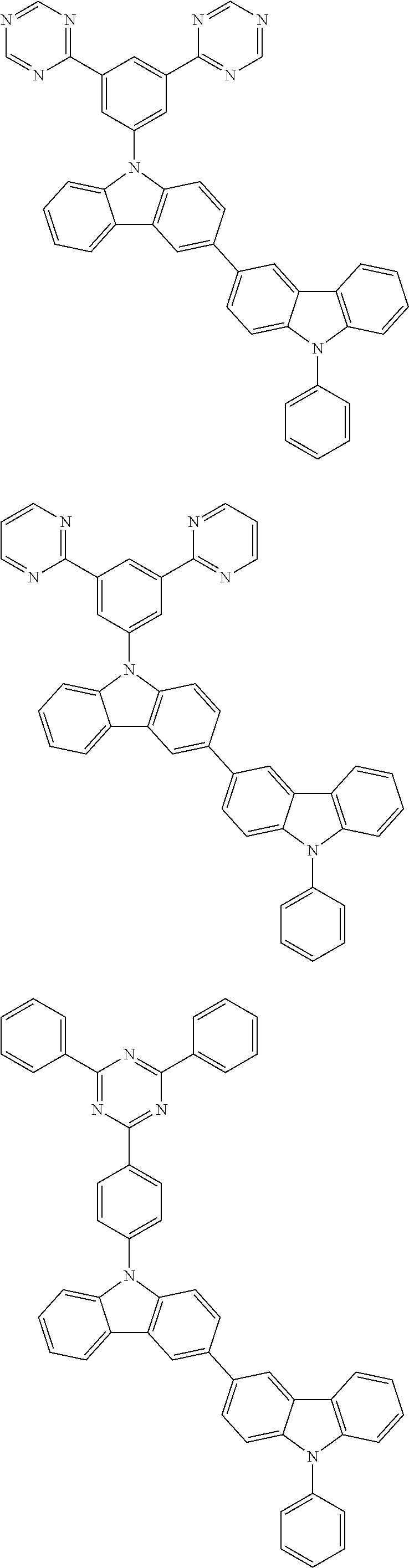
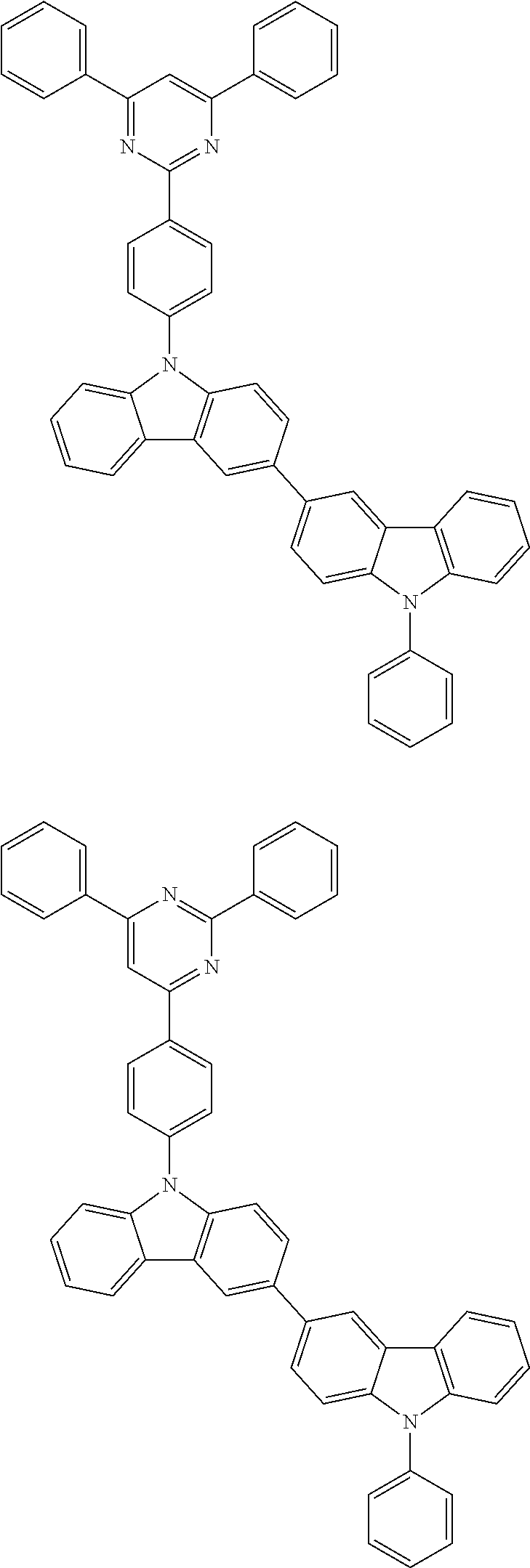
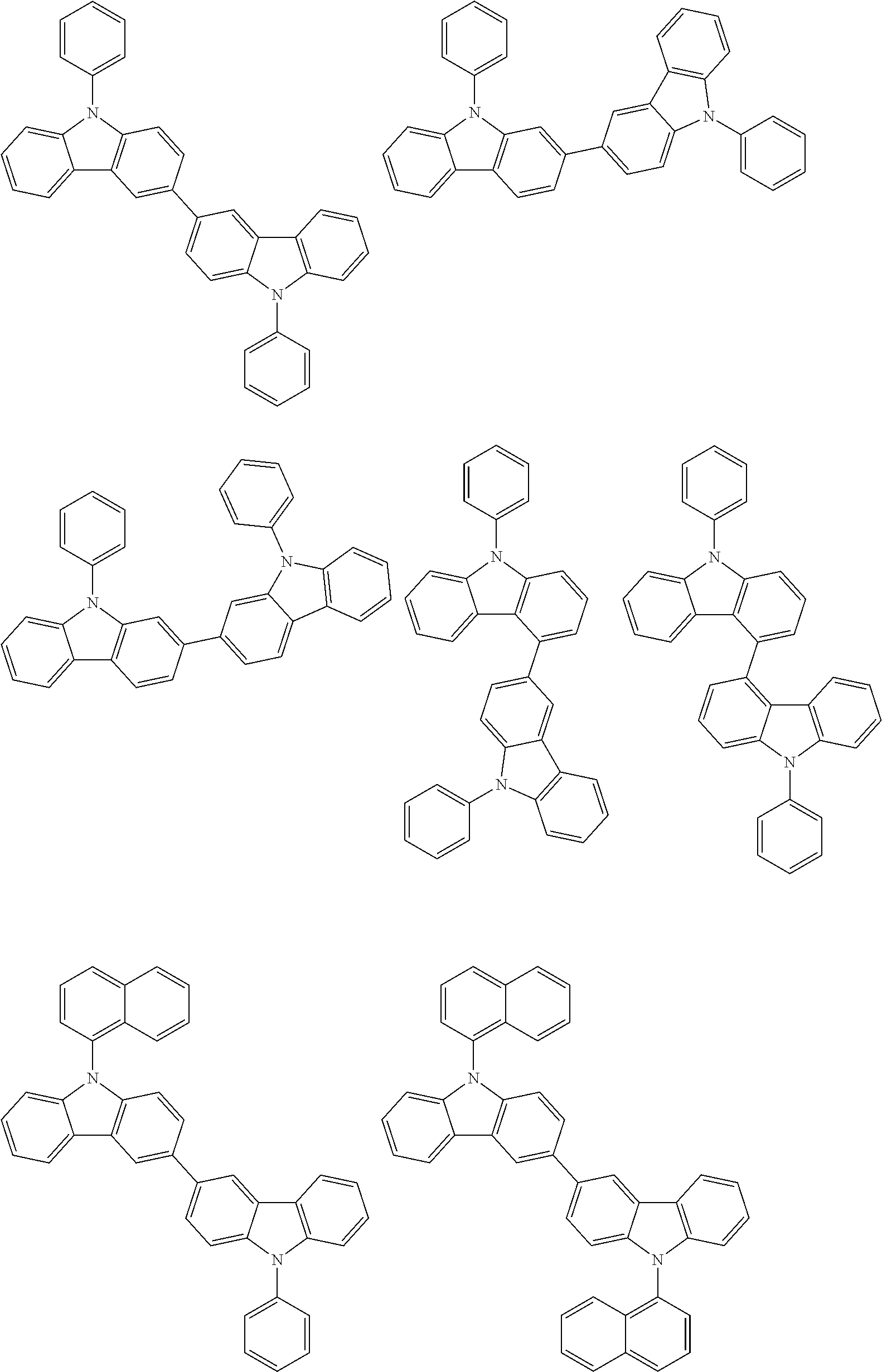
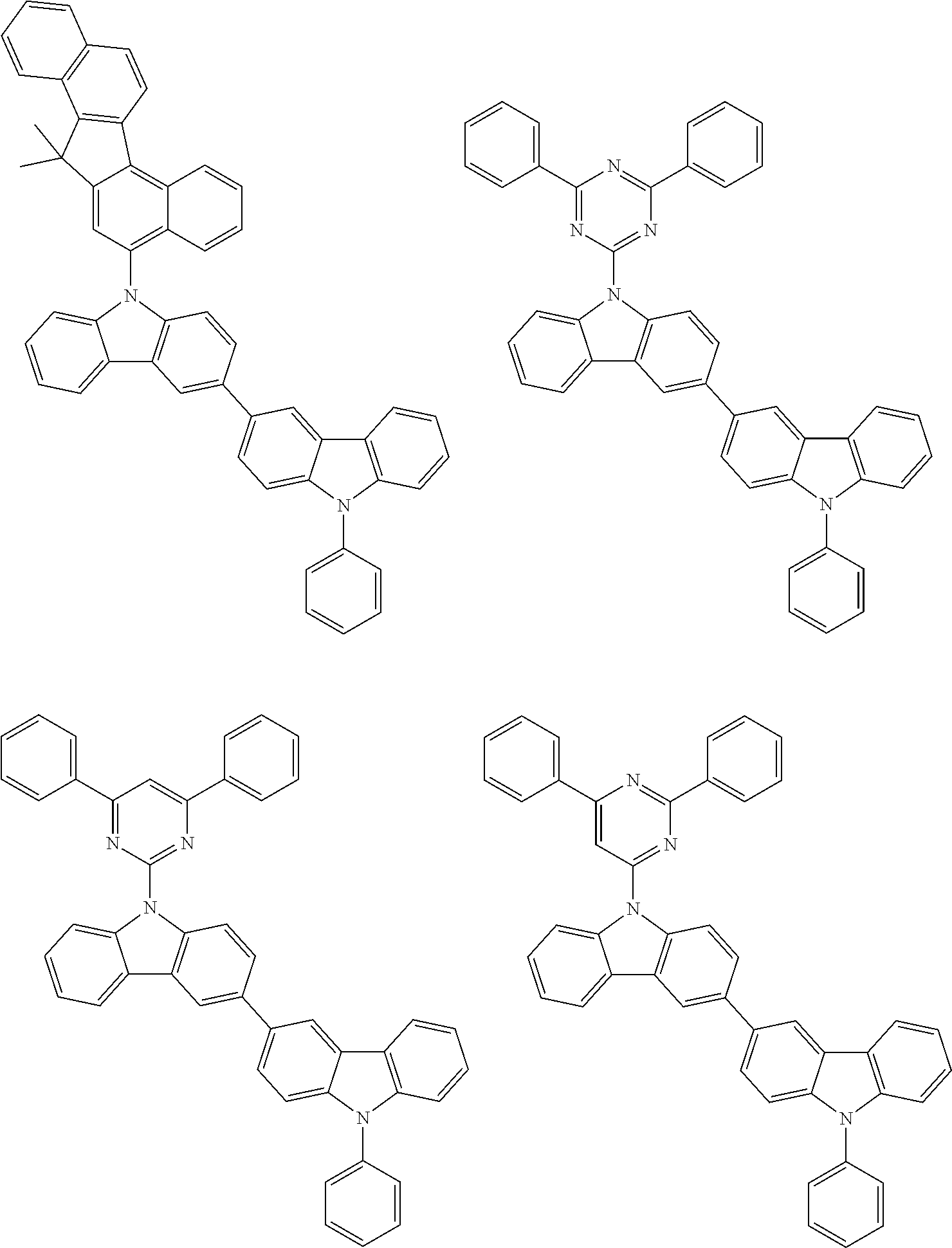
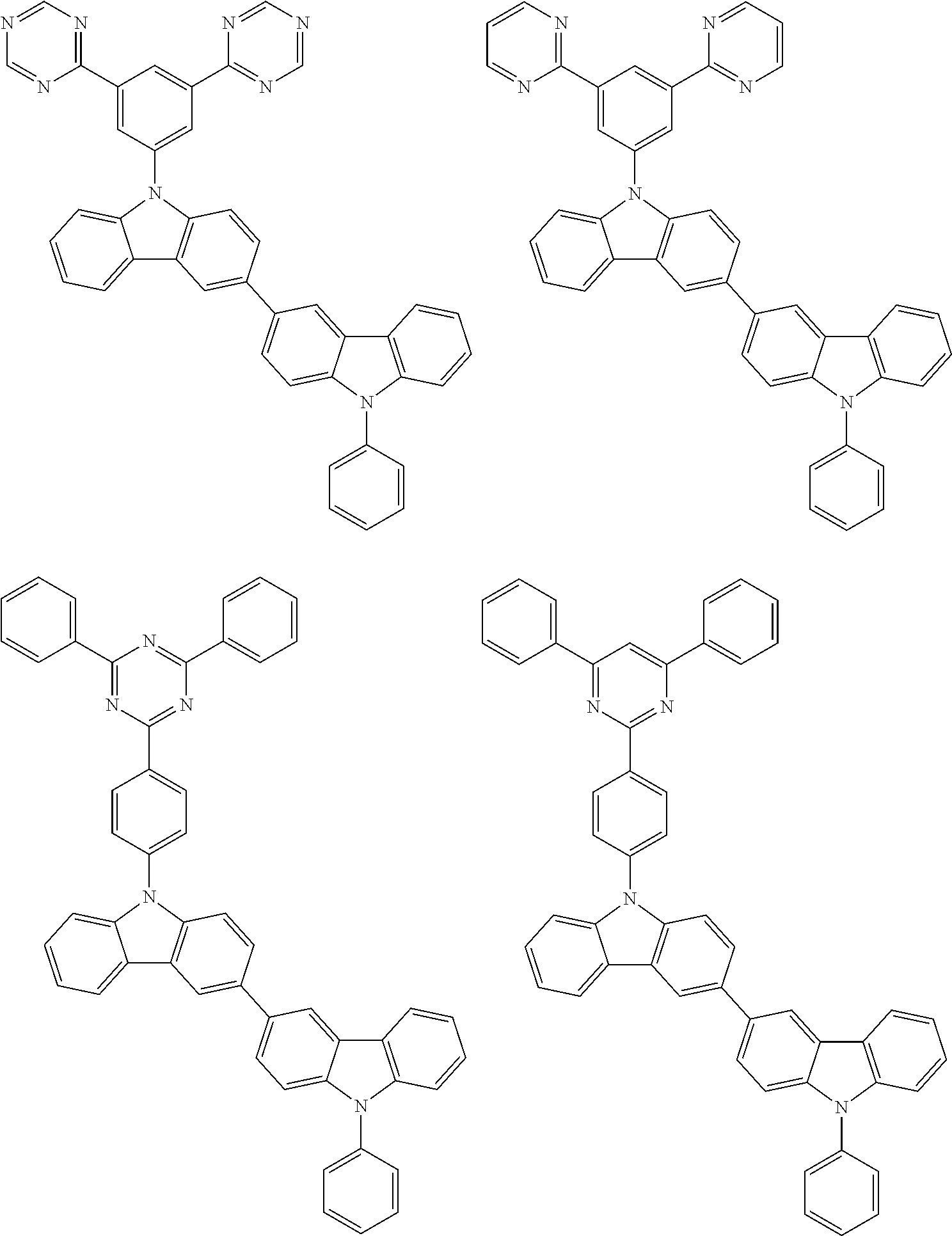
- In one embodiment, the mixed organic layer may include at least two different compounds selected from compounds below:
- In one embodiment, the EML may include BD, Ir(ppy)3, or Ir(pq)2acac as a dopant, but the dopant is not limited thereto:
- In one embodiment, the EML may include at least one compound selected from compounds below as a host, but the host is not limited thereto:
- Hereinafter, the substituents as used herein will be described with respect to certain representative groups. The provided number of carbon atoms is not intended to limit the properties of the substituents. The substituents that are not defined in the present specification should be apparent to those of ordinary skill in the art based on the general definition of the substituents as provided herein.
- As used herein, a C1-C60 alkyl group may refer to a monovalent linear or branched aliphatic hydrocarbon group. Non-limiting examples of the C1-C60 alkyl group include a methyl group, an ethyl group, a propyl group, an isobutyl group, a sec-butyl group, a tert-butyl group, a pentyl group, an iso-amyl group, and a hexyl group. As used herein, a C1-C60 alkylene group may refer to a divalent group that has the same structure as the C1-C60 alkyl group.
- As used herein, a C1-C60 alkoxy group may refer to a monovalent group having a formula of —OA101 (where A101 is the C1-C60 alkyl group). Non-limiting examples of the C1-C60 alkoxy group include a methoxy group, an ethoxy group, and an isopropyloxy group.
- As used herein, a C2-C60 alkenyl group may refer to a hydrocarbon chain having at least one carbon-carbon double bond at one or more positions along a carbon chain of the C2-C60 alkyl group. For example, the C2-C60 alkenyl group may include a terminal alkene and/or an internal alkene (e.g. in the middle or at an end of the C2-C60 alkyl group). Non-limiting examples of the C2-C60 alkenyl group include an ethenyl group, a propenyl group, and a butenyl group. As used herein, a C2-C60 alkenylene group may refer to a divalent group that has the same structure as the C2-C60 alkenyl group.
- As used herein, a C2-C60 alkynyl group may refer to a hydrocarbon chain having at least one carbon-carbon triple bond at one or more positions along a carbon chain of the C2-C60 alkyl group. For example, the C2-C60 alkynyl group may include a terminal alkyne and/or an internal alkyne (e.g. in the middle or at an end of the C2-C60 alkyl group). Non-limiting examples of the C2-C60 alkynyl group include an ethynyl group and a propynyl group. As used herein, a C2-C60 alkynylene group may refer to a divalent group that has the same structure as the C2-C60 alkynyl group.
- As used herein, a C3-C10 cycloalkyl group may refer to a C3-C10 monovalent saturated hydrocarbon monocyclic group. Non-limiting examples of the C3-C10 cycloalkyl group include a cyclopropyl group, a cyclobutyl group, a cyclopentyl group, a cyclohexyl group, and a cycloheptyl group. As used herein, a C3-C10 cycloalkylene group may refer to a divalent group that has the same structure as the C3-C10 cycloalkyl group.
- As used herein, the C2-C10 heterocycloalkyl group may refer to a C2-C10 monovalent monocyclic group including at least one hetero atom selected from N, O, P, and S as a ring-forming atom, and carbon atoms as remaining ring-forming atoms. Non-limiting examples of the C2-C10 heterocycloalkyl group include a tetrahydrofuranyl group and a tetrahydrothiophenyl group. As used herein, a C2-C10 heterocycloalkylene group may refer to a divalent group that has the same structure as the C2-C10 heterocycloalkyl group.
- As used herein, a C3-C10 cycloalkenyl group may refer to a C3-C10 monovalent monocyclic group that has at least one double bond in the ring, but does not have aromaticity. Non-limiting examples of the C3-C10 cycloalkenyl group include a cyclopentyl group, a cyclohexenyl group, and a cycloheptenyl group. As used herein, a C3-C10 cycloalkenylene group may refer to a divalent group that has the same structure as the C3-C10 cycloalkenyl group.
- As used herein, a C2-C10 heterocycloalkenyl group may refer to a C2-C10 monovalent monocyclic group including at least one hetero atom selected from N, O, P, and S as a ring-forming atom and at least one double bond in the ring. Non-limiting examples of the C2-C10 heterocycloalkenyl group include a 2,3-hydrofuranyl group and a 2,3-hydrothiophenyl group. As used herein, a C2-C10 heterocycloalkenylene group may refer to a divalent group that has the same structure as the C2-C10 heterocycloalkenyl group.
- As used herein, a C6-C60 aryl group may refer to a monovalent group having a C6-C60 carbocyclic aromatic system, and a C6-C60 arylene group may refer to a divalent group that has a C6-C60 carbocyclic aromatic system. Non-limiting examples of the C6-C60 aryl group include a phenyl group, a naphthyl group, an anthracenyl group, a phenanthrenyl group, a pyrenyl group, and a chrysenyl group. When the C6-C60 aryl group and/or the C6-C60 arylene group include at least two rings, the rings may be fused to each other.
- As used herein, a C2-C60 heteroaryl group may refer to a monovalent group having a C2-C60 carbocyclic aromatic system and including at least one heteroatom selected from N, O, P, and S as a ring-forming atom, and carbon atoms as the remaining ring-forming atoms, and a C2-C60 heteroarylene group may refer to a divalent group having a C2-C60 carbocyclic aromatic system and including at least one heteroatom selected from N, O, P, and S as a ring-forming atom, and carbon atoms as the remaining ring-forming atoms. Non-limiting examples of the C2-C60 heteroaryl group include a pyridinyl group, a pyrimidinyl group, a pyrazinyl group, a pyridazinyl group, a triazinyl group, a quinolinyl group, and an isoquinolinyl group. When the C2-C60 heteroaryl group and/or a C2-C60 heteroarylene group include at least two rings, the rings may be fused to each other.
- As used herein, a C6-C60 aryloxy group may refer to a —OA102 group (where A102 is the C6-C60 aryl group), and a C6-C60 arylthio group may refer to a —SA103 group (where A103 is the C6-C60 aryl group).
- As used herein, a monovalent non-aromatic condensed polycyclic group may refer to a monovalent group that has at least two rings that are condensed to each other, each ring including only carbon atoms as ring-forming atoms (e.g., 8 to 60 carbon atoms), and does not have overall aromaticity. Non-limiting examples of the non-aromatic condensed polycyclic group include a fluorenyl group. As used herein, a divalent non-aromatic condensed polycyclic group may refer to a divalent group that has the same structure as the monovalent non-aromatic condensed polycyclic group.
- As used herein, a monovalent non-aromatic heterocondensed polycyclic group may refer to a monovalent group that has at least two rings that are condensed to each other, each ring including a heteroatom selected from N, O, P, and S as a ring-forming atom and carbon atoms as remaining ring-forming atoms (e.g., 2 to 60 carbon atoms), and does not have overall aromaticity. Non-limiting examples of the non-aromatic heterocondensed polycyclic group include a carbazolyl group. As used herein, a divalent non-aromatic heterocondensed polycyclic group may refer to a divalent group that has the same structure as the monovalent non-aromatic heterocondensed polycyclic group.
- As used herein, at least one substituent of the substituted C3-C10 cycloalkylene group, substituted C2-C10 heterocycloalkylene group, substituted C3-C10 cycloalkenylene group, substituted C2-C10 heterocycloalkenylene group, substituted C6-C60 arylene group, substituted C2-C60 heteroarylene group, substituted divalent non-aromatic condensed polycyclic group, substituted divalent non-aromatic heterocondensed polycyclic group, substituted C1-C60 alkyl group, substituted C2-C60 alkenyl group, substituted C2-C60 alkynyl group, substituted C1-C60 alkoxy group, substituted C3-C10 cycloalkyl group, substituted C2-C10 heterocycloalkyl group, substituted C3-C10 cycloalkenyl group, substituted C2-C10 heterocycloalkenyl group, substituted C6-C60 aryl group, substituted C6-C60 aryloxy group, substituted C6-C60 arylthio group, substituted C2-C60 heteroaryl group, substituted monovalent non-aromatic condensed polycyclic group, and/or substituted monovalent non-aromatic heterocondensed polycyclic group is selected from
- a deuterium, —F, —Cl, —Br, —I, a hydroxyl group, a cyano group, a nitro group, an amino group, an amidino group, a hydrazine group, a hydrazone group, a carboxylic acid or a salt thereof, a sulfonic acid or a salt thereof, a phosphoric acid or a salt thereof, a C1-C60 alkyl group, a C2-C60 alkenyl group, a C2-C60 alkynyl group, and/or a C1-C60 alkoxy group;
- a C1-C60 alkyl group, a C2-C60 alkenyl group, a C2-C60 alkynyl group, and/or a C1-C60 alkoxy group, each substituted with at least one selected from a deuterium, —F, —Cl, —Br, —I, a hydroxyl group, a cyano group, a nitro group, an amino group, an amidino group, a hydrazine group, a hydrazone group, a carboxylic acid or a salt thereof, a sulfonic acid or a salt thereof, a phosphoric acid or a salt thereof, a C3-C10 cycloalkyl group, a C2-C10 heterocycloalkyl group, a C3-C10 cycloalkenyl group, a C2-C10 heterocycloalkenyl group, a C6-C60 aryl group, a C6-C60 aryloxy, a C6-C60 arylthio, a C2-C60 heteroaryl group, a monovalent non-aromatic condensed polycyclic group, a monovalent non-aromatic heterocondensed polycyclic group, —N(Q11)(Q12), —Si(Q13)(Q14)(Q15), and —B(Q16)(Q17);
- a C3-C10 cycloalkyl group, a C2-C10 heterocycloalkyl group, a C3-C10 cycloalkenyl group, a C2-C10 heterocycloalkenyl group, a C6-C60 aryl group, a C6-C60 aryloxy, a C6-C60 arylthio, a C2-C60 heteroaryl group, a monovalent non-aromatic condensed polycyclic group, and/or a monovalent non-aromatic heterocondensed polycyclic group;
- a C3-C10 cycloalkyl group, a C2-C10 heterocycloalkyl group, a C3-C10 cycloalkenyl group, a C2-C10 heterocycloalkenyl group, a C6-C60 aryl group, a C6-C60 aryloxy, a C6-C60 arylthio, a C2-C60 heteroaryl group, a monovalent non-aromatic condensed polycyclic group, and/or a monovalent non-aromatic heterocondensed polycyclic group, each substituted with at least one selected from a deuterium, —F, —Cl, —Br, —I, a hydroxyl group, a cyano group, a nitro group, an amino group, an amidino group, a hydrazine group, a hydrazone group, a carboxylic acid or a salt thereof, a sulfonic acid or a salt thereof, a phosphoric acid or a salt thereof, a C1-C60 alkyl group, a C2-C60 alkenyl group, a C2-C60 alkynyl group, a C1-C60 alkoxy group a C3-C10 cycloalkyl group, a C2-C10 heterocycloalkyl group, a C3-C10 cycloalkenyl group, a C2-C10 heterocycloalkenyl group, a C6-C60 aryl group, a C6-C60 aryloxy, a C6-C60 arylthio, a C2-C60 heteroaryl group, a monovalent non-aromatic condensed polycyclic group, a monovalent non-aromatic heterocondensed polycyclic group, N(Q21)(Q22), —Si(Q23)(Q24)(Q25), and —B(Q26)(Q27); and/or
- —N(Q31)(Q32), —Si(Q33)(Q34)(Q35), and/or —B(Q36)(Q37),
- Q11 to Q17, Q21 to Q27, and Q31 to Q37 are each independently selected from a hydrogen, —F, —Cl, —Br, —I, a hydroxyl group, a cyano group, a nitro group, an amino group, an amidino group, a hydrazine group, a hydrazone group, a carboxylic acid or a salt thereof, a sulfonic acid or a salt thereof, a phosphoric acid or a salt thereof, a C1-C60 alkyl group, a C2-C60 alkenyl group, a C2-C60 alkynyl group, a C1-C60 alkoxy group, a C3-C10 cycloalkyl group, a C2-C10 heterocycloalkyl group, a C3-C10 cycloalkenyl group, a C2-C10 heterocycloalkenyl group, a C6-C60 aryl group, a C2-C60 heteroaryl group, a monovalent non-aromatic condensed polycyclic group, and/or a monovalent non-aromatic heterocondensed polycyclic group.
- In one embodiment, at least one substituent of the substituted C3-C10 cycloalkylene group, substituted C2-C10 heterocycloalkylene group, substituted C3-C10 cycloalkenylene group, substituted C2-C10 heterocycloalkenylene group, substituted C6-C60 arylene group, substituted C2-C60 heteroarylene group, substituted divalent non-aromatic condensed polycyclic group, substituted divalent non-aromatic heterocondensed polycyclic group, substituted C1-C60 alkyl group, substituted C2-C60 alkenyl group, substituted C2-C60 alkynyl group, substituted C1-C60 alkoxy group, substituted C3-C10 cycloalkyl group, substituted C2-C10 heterocycloalkyl group, substituted C3-C10 cycloalkenyl group, substituted C2-C10 heterocycloalkenyl group, substituted C6-C60 aryl group, substituted C6-C60 aryloxy group, substituted C6-C60 arylthio group, substituted C2-C60 heteroaryl group, substituted monovalent non-aromatic condensed polycyclic group, and substituted monovalent non-aromatic heterocondensed polycyclic group is selected from
- a deuterium, —F, —Cl, —Br, —I, a hydroxyl group, a cyano group, a nitro group, an amino group, an amidino group, a hydrazine group, a hydrazone group, a carboxylic acid or a salt thereof, a sulfonic acid or a salt thereof, a phosphoric acid or a salt thereof, a C1-C60 alkyl group, a C2-C60 alkenyl group, a C2-C60 alkynyl group, and/or a C1-C60 alkoxy group;
- a C1-C60 alkyl group, a C2-C60 alkenyl group, a C2-C60 alkynyl group, and/or a C1-C60 alkoxy group, each substituted with at least one selected from a deuterium, —F, —Cl, —Br, —I, a hydroxyl group, a cyano group, a nitro group, an amino group, an amidino group, a hydrazine group, a hydrazone group, a carboxylic acid or a salt thereof, a sulfonic acid or a salt thereof, a phosphoric acid or a salt thereof, a cyclopentyl group, a cyclohexyl group, a cycloheptyl group, a cyclopentenyl group, a cycloheptenyl group, a cyclohexenyl group, a phenyl group, a pentalenyl group, an indenyl group, a naphthyl group, an azulenyl group, a heptalenyl group, an indacenyl group, an acenaphthyl group, a fluorenyl group, a spiro-fluorenyl group, a benzofluorenyl group, a dibenzofluorenyl group, a phenalenyl group, a phenanthrenyl group, an anthracenyl group, a fluoranthenyl group, a triphenylenyl group, a pyrenyl group, a chrysenyl group, a naphthacenyl group, a picenyl group, a perylenyl group, a pentaphenyl group, a hexacenyl group, a pentacenyl group, a rubicenyl group, a coroneryl group, an ovalenyl group, a pyrrolyl group, a thiophenyl group, a furanyl group, an imidazolyl group, a pyrazolyl group, a thiazolyl group, an isothiazolyl group, an oxazolyl group, an isoxazolyl group, a pyridinyl group, a pyrazinyl group, a pyrimidinyl group, a pyridazinyl group, an isoindolyl group, an indolyl group, an indazolyl group, a purinyl group, a quinolinyl group, an isoquinolinyl group, a benzoquinolinyl group, a phthalazinyl group, a naphthyridinyl group, a quinoxalinyl group, a quinazolinyl group, a cinnolinyl group, a carbazolyl group, a phenanthridinyl group, an acridinyl group, a phenanthrolinyl group, a phenazinyl group, a benzoimidazolyl group, a benzofuranyl group, a benzothiophenyl group, an isobenzothiazolyl group, a benzoxazolyl group, an isobenzoxazolyl group, a triazolyl group, a tetrazolyl group, an oxadiazolyl group, a triazinyl group, a dibenzofuranyl group, a dibenzothiophenyl group, a benzocarbazolyl group, a dibenzocarbazolyl group, a thiadiazolyl group, an imidazopyridinyl group, an imidazopyrimidinyl group, —N(Q11)(Q12), —Si(Q13)(Q14)(Q15), and —B(Q16)(Q17);
- a cyclopentyl group, a cyclohexyl group, a cycloheptyl group, a cyclopentenyl group, a cycloheptenyl group, a cyclohexenyl group, a phenyl group, a pentalenyl group, an indenyl group, a naphthyl group, an azulenyl group, a heptalenyl group, an indacenyl group, an acenaphthyl group, a fluorenyl group, a spiro-fluorenyl group, a benzofluorenyl group, a dibenzofluorenyl group, a phenalenyl group, a phenanthrenyl group, an anthracenyl group, a fluoranthenyl group, a triphenylenyl group, a pyrenyl group, a chrysenyl group, a naphthacenyl group, a picenyl group, a perylenyl group, a pentaphenyl group, a hexacenyl group, a pentacenyl group, a rubicenyl group, a coroneryl group, an ovalenyl group, a pyrrolyl group, a thiophenyl group, a furanyl group, an imidazolyl group, a pyrazolyl group, a thiazolyl group, an isothiazolyl group, an oxazolyl group, an isoxazolyl group, a pyridinyl group, a pyrazinyl group, a pyrimidinyl group, a pyridazinyl group, an isoindolyl group, an indolyl group, an indazolyl group, a purinyl group, a quinolinyl group, an isoquinolinyl group, a benzoquinolinyl group, a phthalazinyl group, a naphthyridinyl group, a quinoxalinyl group, a quinazolinyl group, a cinnolinyl group, a carbazolyl group, a phenanthridinyl group, an acridinyl group, a phenanthrolinyl group, a phenazinyl group, a benzoimidazolyl group, a benzofuranyl group, a benzothiophenyl group, an isobenzothiazolyl group, a benzoxazolyl group, an isobenzoxazolyl group, a triazolyl group, a tetrazolyl group, an oxadiazolyl group, a triazinyl group, a dibenzofuranyl group, a dibenzothiophenyl group, a benzocarbazolyl group, a dibenzocarbazolyl group, a thiadiazolyl group, an imidazopyridinyl group, and/or imidazopyrimidinyl group;
- a cyclopentyl group, a cyclohexyl group, a cycloheptyl group, a cyclopentenyl group, a cycloheptenyl group, a cyclohexenyl group, a phenyl group, a pentalenyl group, an indenyl group, a naphthyl group, an azulenyl group, a heptalenyl group, an indacenyl group, an acenaphthyl group, a fluorenyl group, a spiro-fluorenyl group, a benzofluorenyl group, a dibenzofluorenyl group, a phenalenyl group, a phenanthrenyl group, an anthracenyl group, a fluoranthenyl group, a triphenylenyl group, a pyrenyl group, a chrysenyl group, a naphthacenyl group, a picenyl group, a perylenyl group, a pentaphenyl group, a hexacenyl group, a pentacenyl group, a rubicenyl group, a coroneryl group, an ovalenyl group, a pyrrolyl group, a thiophenyl group, a furanyl group, an imidazolyl group, a pyrazolyl group, a thiazolyl group, an isothiazolyl group, an oxazolyl group, an isoxazolyl group, a pyridinyl group, a pyrazinyl group, a pyrimidinyl group, a pyridazinyl group, an isoindolyl group, an indolyl group, an indazolyl group, a purinyl group, a quinolinyl group, an isoquinolinyl group, a benzoquinolinyl group, a phthalazinyl group, a naphthyridinyl group, a quinoxalinyl group, a quinazolinyl group, a cinnolinyl group, a carbazolyl group, a phenanthridinyl group, an acridinyl group, a phenanthrolinyl group, a phenazinyl group, a benzoimidazolyl group, a benzofuranyl group, a benzothiophenyl group, an isobenzothiazolyl group, a benzoxazolyl group, an isobenzoxazolyl group, a triazolyl group, a tetrazolyl group, an oxadiazolyl group, a triazinyl group, a dibenzofuranyl group, a dibenzothiophenyl group, a benzocarbazolyl group, a dibenzocarbazolyl group, a thiadiazolyl group, an imidazopyridinyl group, and/or an imidazopyrimidinyl group, each substituted with at least one selected from a deuterium, —F, —Cl, —Br, —I, a hydroxyl group, a cyano group, a nitro group, an amino group, an amidino group, a hydrazine group, a hydrazone group, a carboxylic acid or a salt thereof, a sulfonic acid or a salt thereof, a phosphoric acid or a salt thereof, a C1-C60 alkyl group, a C2-C60 alkenyl group, a C2-C60 alkynyl group, a C1-C60 alkoxy group, a cyclopentyl group, a cyclohexyl group, a cycloheptyl group, a cyclopentenyl group, a cycloheptenyl group, a cyclohexenyl group, a phenyl group, a pentalenyl group, an indenyl group, a naphthyl group, an azulenyl group, a heptalenyl group, an indacenyl group, an acenaphthyl group, a fluorenyl group, a spiro-fluorenyl group, a benzofluorenyl group, a dibenzofluorenyl group, a phenalenyl group, a phenanthrenyl group, an anthracenyl group, a fluoranthenyl group, a triphenylenyl group, a pyrenyl group, a chrysenyl group, a naphthacenyl group, a picenyl group, a perylenyl group, a pentaphenyl group, a hexacenyl group, a pentacenyl group, a rubicenyl group, a coroneryl group, an ovalenyl group, a pyrrolyl group, a thiophenyl group, a furanyl group, an imidazolyl group, a pyrazolyl group, a thiazolyl group, an isothiazolyl group, an oxazolyl group, an isoxazolyl group, a pyridinyl group, a pyrazinyl group, a pyrimidinyl group, a pyridazinyl group, an isoindolyl group, an indolyl group, an indazolyl group, a purinyl group, a quinolinyl group, an isoquinolinyl group, a benzoquinolinyl group, a phthalazinyl group, a naphthyridinyl group, a quinoxalinyl group, a quinazolinyl group, a cinnolinyl group, a carbazolyl group, a phenanthridinyl group, an acridinyl group, a phenanthrolinyl group, a phenazinyl group, a benzoimidazolyl group, a benzofuranyl group, a benzothiophenyl group, an isobenzothiazolyl group, a benzoxazolyl group, an isobenzoxazolyl group, a triazolyl group, a tetrazolyl group, an oxadiazolyl group, a triazinyl group, a dibenzofuranyl group, a dibenzothiophenyl group, a benzocarbazolyl group, a dibenzocarbazolyl group, a thiadiazolyl group, an imidazopyridinyl group, an imidazopyrimidinyl group, —N(Q21)(Q22), —Si(Q23)(Q24)(Q25), and —B(Q26)(Q27); and/or
- —N(Q31)(Q32), —Si(Q33)(Q34)(Q35), and/or —B(Q36)(Q37),
- Q11 to Q17, Q21 to Q27, and Q31 to Q37 are each independently selected from a hydrogen, a deuterium, —F, —Cl, —Br, —I, a hydroxyl group, a cyano group, a nitro group, an amino group, an amidino group, a hydrazine group, a hydrazone group, a carboxylic acid or a salt thereof, a sulfonic acid or a salt thereof, a phosphoric acid or a salt thereof, a C1-C60 alkyl group, a C2-C60 alkenyl group, a C2-C60 alkynyl group, a C1-C60 alkoxy group, a cyclopentyl group, a cyclohexyl group, a cycloheptyl group, a cyclopentenyl group, a cycloheptenyl group, a cyclohexenyl group, a phenyl group, a pentalenyl group, an indenyl group, a naphthyl group, an azulenyl group, a heptalenyl group, an indacenyl group, an acenaphthyl group, a fluorenyl group, a spiro-fluorenyl group, a benzofluorenyl group, a dibenzofluorenyl group, a phenalenyl group, a phenanthrenyl group, an anthracenyl group, a fluoranthenyl group, a triphenylenyl group, a pyrenyl group, a chrysenyl group, a naphthacenyl group, a picenyl group, a perylenyl group, a pentaphenyl group, a hexacenyl group, a pentacenyl group, a rubicenyl group, a coroneryl group, an ovalenyl group, a pyrrolyl group, a thiophenyl group, a furanyl group, an imidazolyl group, a pyrazolyl group, a thiazolyl group, an isothiazolyl group, an oxazolyl group, an isoxazolyl group, a pyridinyl group, a pyrazinyl group, a pyrimidinyl group, a pyridazinyl group, an isoindolyl group, an indolyl group, an indazolyl group, a purinyl group, a quinolinyl group, an isoquinolinyl group, a benzoquinolinyl group, a phthalazinyl group, a naphthyridinyl group, a quinoxalinyl group, a quinazolinyl group, a cinnolinyl group, a carbazolyl group, a phenanthridinyl group, an acridinyl group, a phenanthrolinyl group, a phenazinyl group, a benzoimidazolyl group, a benzofuranyl group, a benzothiophenyl group, an isobenzothiazolyl group, a benzoxazolyl group, an isobenzoxazolyl group, a triazolyl group, a tetrazolyl group, an oxadiazolyl group, a triazinyl group, a dibenzofuranyl group, a dibenzothiophenyl group, a benzocarbazolyl group, a dibenzocarbazolyl group, a thiadiazolyl group, an imidazopyridinyl group, and/or an imidazopyrimidinyl group.
- The term “Ph” used herein refers to a phenyl group, the term “Me” used herein refers to a methyl group, the term “Et” used herein refers to an ethyl group, and the term “ter-Bu” or “But” used herein refers to a tert-butyl group.
- The expression “an organic layer includes at least one compound of Formula X” used herein may refer to an organic layer) including one compound of Formula X, or two or more different compounds of Formula X.
- The term “organic layer” used herein may refer to a single layer and/or a plurality of layers between the first electrode and the second electrode in the organic light-emitting device. A material included in the organic layer is not limited to an organic material.
- The drawing schematically illustrates a cross-sectional view of an organic light-emitting
device 10 according to an embodiment of the present invention. The organic light-emittingdevice 10 includes afirst electrode 110, anorganic layer 150, and asecond electrode 190. - Hereinafter, a structure and a preparation method of an organic light-emitting device are described by referring to the drawing.
- In the organic light-emitting
device 10 shown in the drawing, a substrate may be positioned on a first side (e.g. lower side) of thefirst electrode 110 or a second side (e.g. upper side) of thesecond electrode 190. The substrate may be a glass substrate or a transparent plastic substrate having good mechanical strength, thermal stability, transparency, surface smoothness, ease of handling, and water resistance. - The
first electrode 110 may be formed by applying a first electrode material on the substrate by, for example, deposition or sputtering. When thefirst electrode 110 is an anode, the first electrode material may be selected from materials having a high work function and capable of easily injecting the holes. Thefirst electrode 110 may be a reflective electrode, a semi-transparent electrode, or a transparent electrode. Non-limiting examples of the first electrode material may include indium-tin oxide (ITO), indium-zinc-oxide (IZO), tin oxide (SnO2), and zinc oxide (ZnO). In embodiments where thefirst electrode 110 is a semi-transparent electrode or a reflective electrode, at least one selected from magnesium (Mg), aluminum (Al), aluminum-lithium (Al—Li), a Calcium (Ca), magnesium-indium (Mg—In), and magnesium-silver (Mg—Ag) may be selected as the first electrode material. - The
first electrode 110 may have a single-layered structure or a multi-layered structure including at least two layers. In one embodiment, thefirst electrode 110 may have a three-layered structure of ITO/Ag/ITO, but the structure of thefirst electrode 110 is not limited thereto. - In one embodiment, the
organic layer 150 is positioned on thefirst electrode 110 and includes an EML. - The
organic layer 150 may further include a hole transport region between thefirst electrode 110 and the EML, an electron transport region between the EML and thesecond electrode 190, and a mixed organic layer between the EML and the electron transport region. - The hole transport region may include at least one selected from an HIL, an HTL, a buffer layer, and an EBL, and the electron transport region may include at least one selected from an HBL, an ETL, and an EIL, but the hole transport region and the electron transport region are not limited thereto.
- The structure of the hole transport region may include a single layer structure formed of one material, a single layer structure formed of multiple different materials, or multiple layers structure formed of multiple different materials.
- In one embodiment, the hole transport region may have a single layer structure formed of multiple different materials, for example, HIL/HTL, HIL/HTL/buffer layer, HIL/buffer layer, HTL/buffer layer, or HIL/HTL/EBL sequentially stacked on the
first electrode 110, but the structure of the hole transport region is not limited thereto. - When the hole transport region includes an HIL, the HIL may be formed on the
first electrode 110 using (utilizing) various methods such as, for example, vacuum deposition, spin coating, casting, Langmuir-Blodgett (LB) deposition, inkjet printing, laser printing, or laser induced thermal imaging (LITI). - When the HIL is formed by vacuum deposition, the deposition temperature may be about 100 to about 500° C., the degree of vacuum may be about 10−8 to about 10−3 torr, and the deposition speed may be about 0.01 to about 100 Å/sec, depending on the kind of compound for forming the HIL and the desired structure of the HIL.
- When the HIL is formed by spin coating, the the coating speed may be about 2,000 rpm to about 5,000 rpm and the heat treatment temperature may be about 80° C. to about 200° C., depending on the kind of compound for forming the HIL and the desired structure of the HIL.
- When the hole transport region includes an HTL, the HTL may be formed on the
first electrode 110 or on the HIL using (utilizing) various methods such as, for example, vacuum deposition, spin coating, casting, LB deposition, inkjet printing, laser printing, or LITI. When the HTL is formed by vacuum deposition or spin coating, the deposition conditions and the coating conditions for forming the HTL may be similar to the deposition conditions and the coating conditions for forming the HIL. - The hole transport region may include at least one of m-MTDATA, TDATA, 2-TNATA, NPB, β-NPB, TPD, Spiro-TPD, Spiro-NPB, methylated NPB, TAPC, HMTPD, 4,4′,4″-tris(N-carbazolyl)triphenylamine) (TCTA), polyaniline/Dodecylbenzenesulfonic acid (Pani/DBSA), poly(3,4-ethylenedioxythiophene)/poly(4-styrenesulfonate) (PEDOT/PSS), polyaniline/camphor sulfonic acid (Pani/CSA), polyaniline)/poly(4-styrenesulfonate (PANI/PSS), a compound represented by Formula 201, and a compound represented by Formula 202:
- In Formulae 201 and 202,
- definitions of L201 to L205 may each independently be the same as the definition of L1 as described in the present specification;
- xa1 to xa4 are each independently selected from 0, 1, 2, and 3;
- xa5 is selected from 1, 2, 3, 4, and 5; and
- R201 to R204 may be each independently selected from a substituted or unsubstituted C3-C10 cycloalkyl group, a substituted or unsubstituted C2-C10 heterocycloalkyl group, a substituted or unsubstituted C3-C10 cycloalkenyl group, a substituted or unsubstituted C2-C10 heterocycloalkenyl group, a substituted or unsubstituted C6-C60 aryl group, a substituted or unsubstituted C6-C60 aryloxy group, a substituted or unsubstituted C6-C60 arylthio group, a substituted or unsubstituted C2-C60 heteroaryl group, a substituted or unsubstituted monovalent non-aromatic condensed polycyclic group, and/or a substituted or unsubstituted monovalent non-aromatic heterocondensed polycyclic group.
- In one embodiment, in Formulae 201 and 202,
- L201 to L205 are each independently selected from
- a phenylene group, a naphthylene group, a fluorenylene group, a spiro-fluorenylene group, a benzofluorenylene group, a dibenzofluorenylene group, a phenanthrenylene group, an anthracenylene group, a pyrenylene group, a chrysenylene group, a pyridinylene group, a pyrazinylene group, a pyrimidinylene group, a pyridazinylene group, a quinolinylene group, an isoquinolinylene group, a quinoxalinylene group, a quinazolinylene group, a carbazolylene group, and/or a triazinylene group; and/or
- a phenylene group, a naphthylene group, a fluorenylene group, a spiro-fluorenylene group, a benzofluorenylene group, a dibenzofluorenylene group, a phenanthrenylene group, an anthracenylene group, a pyrenylene group, a chrysenylene group, a pyridinylene group, a pyrazinylene group, a pyrimidinylene group, a pyridazinylene group, a quinolinylene group, an isoquinolinylene group, a quinoxalinylene group, a quinazolinylene group, a carbazolylene group, and/or a triazinylene group, each substituted with at least one selected from a deuterium, —F, —Cl, —Br, —I, a hydroxyl group, a cyano group, a nitro group, an amino group, an am idino group, a hydrazine group, a hydrazone group, a carboxylic acid or a salt thereof, a sulfonic acid or a salt thereof, a phosphoric acid or a salt thereof, a C1-C20 alkyl group, a C1-C20 alkoxy group, a phenyl group, a naphthyl group, a fluorenyl group, a spiro-fluorenyl group, a benzofluorenyl group, a dibenzofluorenyl group, a phenanthrenyl group, an anthracenyl group, a pyrenyl group, a chrysenyl group, a pyridinyl group, a pyrazinyl group, a pyrimidinyl group, a pyridazinyl group, an isoindolyl group, a quinolinyl group, an isoquinolinyl group, a quinoxalinyl group, a quinazolinyl group, a carbazolyl group, and a triazinyl group;
- xa1 to xa4 are each independently selected from 0, 1, and 2;
- xa5 is selected from 1, 2, and 3;
- R201 to R204 are each independently selected from, but are not limited to,
- a phenyl group, a naphthyl group, a fluorenyl group, a spiro-fluorenyl group, a benzofluorenyl group, a dibenzofluorenyl group, a phenanthrenyl group, an anthracenyl group, a pyrenyl group, a chrysenyl group, a pyridinyl group, a pyrazinyl group, a pyrimidinyl group, a pyridazinyl group, a quinolinyl group, an isoquinolinyl group, a quinoxalinyl group, a quinazolinyl group, a carbazolyl group, and/or a triazinyl group; and/or
- a phenyl group, a naphthyl group, a fluorenyl group, a spiro-fluorenyl group, a benzofluorenyl group, a dibenzofluorenyl group, a phenanthrenyl group, an anthracenyl group, a pyrenyl group, a chrysenyl group, a pyridinyl group, a pyrazinyl group, a pyrimidinyl group, a pyridazinyl group, a quinolinyl group, an isoquinolinyl group, a quinoxalinyl group, a quinazolinyl group, a carbazolyl group, and/or a triazinyl group, each substituted with at least one selected from a deuterium, —F, —Cl, —Br, —I, a hydroxyl group, a cyano group, a nitro group, an amino group, an amidino group, a hydrazine group, a hydrazone group, a carboxylic acid or a salt thereof, a sulfonic acid or a salt thereof, a phosphoric acid or a salt thereof, a C1-C20 alkyl group, a C1-C20 alkoxy group, a phenyl group, a naphthyl group, an azulenyl group, a fluorenyl group, a spiro-fluorenyl group, a benzofluorenyl group, a dibenzofluorenyl group, a phenanthrenyl group, an anthracenyl group, a pyrenyl group, a chrysenyl group, a pyridinyl group, a pyrazinyl group, a pyrimidinyl group, a pyridazinyl group, a quinolinyl group, an isoquinolinyl group, a quinoxalinyl group, a quinazolinyl group, a carbazolyl group, and a triazinyl group.
- The compound represented by Formula 201 may be represented by Formula 201A below, but is not limited thereto:
- In one embodiment, the compound represented by Formula 201 may be represented by Formula 201A-1:
- The compound represented by Formula 202 may be represented by Formula 202A, but is not limited thereto:
- In Formulae 201A, 201A-1, and 202A,
- L201 to L203, xa1 to xa3, xa5, and R202 to R204 may be as defined in the present specification, definition of R211 and R212 may be the same as the definition of R203, and R213 to R216 may be each independently selected from, but are not limited to, a hydrogen, a deuterium, —F, —Cl, —Br, —I, a hydroxyl group, a cyano group, a nitro group, an amino group, an amidino group, a hydrazine group, a hydrazone group, a carboxylic acid or a salt thereof, a sulfonic acid or a salt thereof, a phosphoric acid or a salt thereof, a C1-C60 alkyl group, a C2-C60 alkenyl group, a C2-C60 alkynyl group, a C1-C60 alkoxy group, a C3-C10 cycloalkyl group, a C2-C10 heterocycloalkyl group, a C3-C10 cycloalkenyl group, a C2-C10 heterocycloalkenyl group, a C6-C60 aryl group, a C6-C60 aryloxy group, a C6-C60 arylthio group, a C2-C60 heteroaryl group, a monovalent non-aromatic condensed polycyclic group, and/or a monovalent non-aromatic heterocondensed polycyclic group.
- In one embodiment, in Formula 201A, 201A-1, and 202A,
- L201 to L203 are each independently selected from
- a phenylene group, a naphthylene group, a fluorenylene group, a spiro-fluorenylene group, a benzofluorenylene group, a dibenzofluorenylene group, a phenanthrenylene group, an anthracenylene group, a pyrenylene group, a chrysenylene group, a pyridinylene group, a pyrazinylene group, a pyrimidinylene group, a pyridazinylene group, a quinolinylene group, an isoquinolinylene group, a quinoxalinylene group, a quinazolinylene group, a carbazolylene group, and/or a triazinylene group; and/or
- a phenylene group, a naphthylene group, a fluorenylene group, a spiro-fluorenylene group, a benzofluorenylene group, a dibenzofluorenylene group, a phenanthrenylene group, an anthracenylene group, a pyrenylene group, a chrysenylene group, a pyridinylene group, a pyrazinylene group, a pyrimidinylene group, a pyridazinylene group, a quinolinylene group, an isoquinolinylene group, a quinoxalinylene group, a quinazolinylene group, a carbazolylene group, and/or a triazinylene group, each substituted with at least one selected from a deuterium, —F, —Cl, —Br, —I, a hydroxyl group, a cyano group, a nitro group, an amino group, an am idino group, a hydrazine group, a hydrazone group, a carboxylic acid or a salt thereof, a sulfonic acid or a salt thereof, a phosphoric acid or a salt thereof, a C1-C20 alkyl group, a C1-C20 alkoxy group, a phenyl group, a naphthyl group, a fluorenyl group, a spiro-fluorenyl group, a benzofluorenyl group, a dibenzofluorenyl group, a phenanthrenyl group, an anthracenyl group, a pyrenyl group, a chrysenyl group, a pyridinyl group, a pyrazinyl group, a pyrimidinyl group, a pyridazinyl group, a quinolinyl group, an isoquinolinyl group, a quinoxalinyl group, a quinazolinyl group, a carbazolyl group, and a triazinyl group;
- xa1 to xa3 are each independently selected from 0 and 1;
- R203, R211, and R212 are each independently selected from
- a phenyl group, a naphthyl group, a fluorenyl group, a spiro-fluorenyl group, a benzofluorenyl group, a dibenzofluorenyl group, a phenanthrenyl group, an anthracenyl group, a pyrenyl group, a chrysenyl group, a pyridinyl group, a pyrazinyl group, a pyrimidinyl group, a pyridazinyl group, a quinolinyl group, an isoquinolinyl group, a quinoxalinyl group, a quinazolinyl group, a carbazolyl group, and/or a triazinyl group; and/or
- a phenyl group, a naphthyl group, a fluorenyl group, a spiro-fluorenyl group, a benzofluorenyl group, a dibenzofluorenyl group, a phenanthrenyl group, an anthracenyl group, a pyrenyl group, a chrysenyl group, a phenanthrenyl group, an anthracenyl group, a pyrenyl group, a chrysenyl group, a pyridinyl group, a pyrazinyl group, a pyrimidinyl group, a pyridazinyl group, a quinolinyl group, an isoquinolinyl group, a quinoxalinyl group, a quinazolinyl group, a carbazolyl group, and/or a triazinyl group, each substituted with at least one selected from a deuterium, —F, —Cl, —Br, —I, a hydroxyl group, a cyano group, a nitro group, an amino group, an amidino group, a hydrazine group, a hydrazone group, a carboxylic acid or a salt thereof, a sulfonic acid or a salt thereof, a phosphoric acid or a salt thereof, a C1-C20 alkyl group, a C1-C20 alkoxy group, a phenyl group, a naphthyl group, a fluorenyl group, a spiro-fluorenyl group, a benzofluorenyl group, a dibenzofluorenyl group, a phenanthrenyl group, an anthracenyl group, a pyrenyl group, a chrysenyl group, a pyridinyl group, a pyrazinyl group, a pyrimidinyl group, a pyridazinyl group, a quinolinyl group, an isoquinolinyl group, a quinoxalinyl group, a quinazolinyl group, a carbazolyl group, and a triazinyl group;
- R213 and R214 are each independently selected from
- a C1-C20 alkyl group and/or a C1-C20 alkoxy group;
- a C1-C20 alkyl group and/or a C1-C20 alkoxy group, each substituted with at least one selected from a deuterium, —F, —Cl, —Br, —I, a hydroxyl group, a cyano group, a nitro group, an amino group, an amidino group, a hydrazine group, a hydrazone group, a carboxylic acid or a salt thereof, a sulfonic acid or a salt thereof, a phosphoric acid or a salt thereof, a phenyl group, a naphthyl group, a fluorenyl group, a spiro-fluorenyl group, a benzofluorenyl group, a dibenzofluorenyl group, a phenanthrenyl group, an anthracenyl group, a pyrenyl group, a chrysenyl group, a pyridinyl group, a pyrazinyl group, a pyrimidinyl group, a pyridazinyl group, a quinolinyl group, an isoquinolinyl group, a quinoxalinyl group, a quinazolinyl group, a carbazolyl group, and a triazinyl group;
- a phenyl group, a naphthyl group, a fluorenyl group, a spiro-fluorenyl group, a benzofluorenyl group, a dibenzofluorenyl group, a phenanthrenyl group, an anthracenyl group, a pyrenyl group, a chrysenyl group, a pyridinyl group, a pyrazinyl group, a pyrimidinyl group, a pyridazinyl group, a quinolinyl group, an isoquinolinyl group, a quinoxalinyl group, a quinazolinyl group, a carbazolyl group, and/or a triazinyl group; and/or
- a phenyl group, a naphthyl group, a fluorenyl group, a spiro-fluorenyl group, a benzofluorenyl group, a dibenzofluorenyl group, a phenanthrenyl group, an anthracenyl group, a pyrenyl group, a chrysenyl group, a pyridinyl group, a pyrazinyl group, a pyrimidinyl group, a pyridazinyl group, a quinolinyl group, an isoquinolinyl group, a quinoxalinyl group, a quinazolinyl group, a carbazolyl group, and/or a triazinyl group, each substituted with at least one selected from a deuterium, —F, —Cl, —Br, —I, a hydroxyl group, a cyano group, a nitro group, an amino group, an amidino group, a hydrazine group, a hydrazone group, a carboxylic acid or a salt thereof, a sulfonic acid or a salt thereof, a phosphoric acid or a salt thereof, a C1-C20 alkyl group, a C1-C20 alkoxy group, a phenyl group, a naphthyl group, a fluorenyl group, a spiro-fluorenyl group, a benzofluorenyl group, a dibenzofluorenyl group, a phenanthrenyl group, an anthracenyl group, a pyrenyl group, a chrysenyl group, a pyridinyl group, a pyrazinyl group, a pyrimidinyl group, a pyridazinyl group, a quinolinyl group, an isoquinolinyl group, a quinoxalinyl group, a quinazolinyl group, a carbazolyl group, and a triazinyl group;
- R215 and R216 are each independently selected from
- a hydrogen, a deuterium, —F, —Cl, —Br, —I, a hydroxyl group, a cyano group, a nitro group, an amino group, an amidino group, a hydrazine group, a hydrazone group, a carboxylic acid or a salt thereof, a sulfonic acid or a salt thereof, a phosphoric acid or a salt thereof, a C1-C20 alkyl group, and/or a C1-C20 alkoxy group;
- a C1-C20 alkyl group and/or a C1-C20 alkoxy group, each substituted with at least one selected from a deuterium, —F, —Cl, —Br, —I, a hydroxyl group, a cyano group, a nitro group, an amino group, an amidino group, a hydrazine group, a hydrazone group, a carboxylic acid or a salt thereof, a sulfonic acid or a salt thereof, a phosphoric acid or a salt thereof, a phenyl group, a naphthyl group, a fluorenyl group, a spiro-fluorenyl group, a benzofluorenyl group, a dibenzofluorenyl group, a phenanthrenyl group, an anthracenyl group, a pyrenyl group, a chrysenyl group, a pyridinyl group, a pyrazinyl group, a pyrimidinyl group, a pyridazinyl group, a quinolinyl group, an isoquinolinyl group, a quinoxalinyl group, a quinazolinyl group, a carbazolyl group, and a triazinyl group;
- a phenyl group, a naphthyl group, a fluorenyl group, a spiro-fluorenyl group, a benzofluorenyl group, a dibenzofluorenyl group, a phenanthrenyl group, an anthracenyl group, a pyrenyl group, a chrysenyl group, a pyridinyl group, a pyrazinyl group, a pyrimidinyl group, a pyridazinyl group, a quinolinyl group, an isoquinolinyl group, a quinoxalinyl group, a quinazolinyl group, and/or a triazinyl group; and/or
- a phenyl group, a naphthyl group, a fluorenyl group, a spiro-fluorenyl group, a benzofluorenyl group, a dibenzofluorenyl group, a phenanthrenyl group, an anthracenyl group, a pyrenyl group, a chrysenyl group, a pyridinyl group, a pyrazinyl group, a pyrimidinyl group, a pyridazinyl group, a quinolinyl group, an isoquinolinyl group, a quinoxalinyl group, a quinazolinyl group, a carbazolyl group, and/or a triazinyl group, each substituted with at least one selected from a deuterium, —F, —Cl, —Br, —I, a hydroxyl group, a cyano group, a nitro group, an amino group, an amidino group, a hydrazine group, a hydrazone group, a carboxylic acid or a salt thereof, a sulfonic acid or a salt thereof, a phosphoric acid or a salt thereof, a C1-C20 alkyl group, a C1-C20 alkoxy group, a phenyl group, a naphthyl group, a fluorenyl group, a spiro-fluorenyl group, a benzofluorenyl group, a dibenzofluorenyl group, a phenanthrenyl group, an anthracenyl group, a pyrenyl group, a chrysenyl group, a pyridinyl group, a pyrazinyl group, a pyrimidinyl group, a pyridazinyl group, a quinolinyl group, an isoquinolinyl group, a quinoxalinyl group, a quinazolinyl group, a carbazolyl group, and a triazinyl group; and
- xa5 is selected from 1 and 2.
- In Formulae 201A and 201A-1, R213 and R214 may be linked to each other to form a saturated or unsaturated ring.
- The compound represented by Formula 201 and the compound represented by Formula 202 may each independently include at least one of Compounds HT1 to HT20 below, but the compound represented by Formula 201 and the compound represented by Formula 202 are not limited thereto:
- A thickness of the hole transport region may be about 100 Å to about 10,000 Å, for example, about 100 Å to about 1,000 Å. When the hole transport region includes both the HIL and the HTL, a thickness of the HIL may be about 100 Å to about 9,950 Å, for example, about 100 Å to about 950 Å, and a thickness of the HTL may be about 50 Å to about 2,000 Å, for example, about 100 Å to about 1,500 Å. When thicknesses of the hole transport region, the HIL, and the HTL are within any of these ranges, the organic light-emitting device may have satisfactory hole transporting properties without a substantial increase in driving voltage.
- The hole transport region may further include a charge-generating material, in addition to the materials described above, to improve conductivity. The charge-generating material may be homogenously or inhomogenously dispersed in the hole transport region.
- The charge-generating material may be, for example, a p-dopant. The p-dopant may be one of a quinone derivative, a metal oxide, and/or a cyano group-containing compound, but the p-dopant is not limited thereto. Non-limiting examples of the p-dopant may include a quinone derivative, such as tetracyanoquinonedimethane (TCNQ) or 2,3,5,6-tetrafluoro-tetracyano-1,4-benzoquinondimethane (F4-TCNQ); a metal oxide, such as a tungsten oxide or a molybden oxide; and Compound HT-D1 below:
- The hole transport region may further include at least one of a buffer layer and an EBL, in addition to the HIL and the HTL. The buffer layer may increase light-emitting efficiency by compensating an optical resonance distance according to the wavelength of light emitted from the EML. The buffer layer may include a material included in the hole transport region. The EBL may block injection of electrons from the electron transport region.
- The HTL may include a first hole transport layer and a second hole transport layer, and the first hole transport layer and the second hole transport layer may be formed of the same material or of different from each other materials.
- The EML may be formed on the
first electrode 110 or on the hole transport region using (utilizing) various methods such as, for example, vacuum deposition, spin coating, casting, LB deposition, inkjet printing, laser printing, or LITI. When the EML is formed by vacuum deposition or spin coating, the deposition conditions and the coating conditions for forming the EML may be similar to the deposition conditions and the coating conditions for forming the HIL. - When the organic light-emitting
device 10 is a full-color organic light-emitting device, the EML may be patterned into individual sub-pixels, such as a red EML, a green EML, and a blue EML. Alternatively, the EML may have a stacked structure of the red EML, the green EML, and the blue EML, or a single layer structure including a red light-emitting material, a green light-emitting material, and a blue light-emitting material formed as a single layer and capable of emitting white light. - The EML may include a host and a dopant.
- Non-limiting examples of the host may include at least one of TPBi, TBADN, ADN (herein, also referred to as “DNA”), CBP, CDBP, and TCP:
- The host may include a compound represented by Formula 301:
-
Ar301-[(L301)xb1-R301]xb2 Formula 301 - In Formula 301, Ar301 is selected from
- a naphthalene, a heptalene, a fluorene, a spiro-fluorene, a benzofluorene, a dibenzofluorene, a phenalene, a phenanthrene, an anthracene, a fluoranthene, a triphenylene, a pyrene, a chrysene, a naphthacene, a picene, a perylene, a pentaphene, and/or an indenoanthracene;
- a naphthalene, a heptalene, a fluorene, a spiro-fluorene, a benzofluorene, a dibenzofluorene, a phenalene, a phenanthrene, an anthracene, a fluoranthene, a triphenylene, a pyrene, a chrysene, a naphthacene, a picene, a perylene, a pentaphene, and/or an indenoanthracene, each substituted with at least one selected from a deuterium, —F, —Cl, —Br, —I, a hydroxyl group, a cyano group, a nitro group, an amino group, an amidino group, a hydrazine group, a hydrazone group, a carboxylic acid or a salt thereof, a sulfonic acid or a salt thereof, a phosphoric acid or a salt thereof, a C1-C60 alkyl group, a C2-C60 alkenyl group, a C2-C60 alkynyl group, a C1-C60 alkoxy group, a C3-C10 cycloalkyl group, a C2-C10 heterocycloalkyl group, a C3-C10 cycloalkenyl group, a C2-C10 heterocycloalkenyl group, a C6-C60 aryl group, a C6-C60 aryloxy group, a C6-C60 arylthio group, a C2-C60 heteroaryl group, a monovalent non-aromatic condensed polycyclic group, a monovalent non-aromatic heterocondensed polycyclic group, and —Si(Q301)(Q302)(Q303) (where Q301 to Q303 are each independently selected from a hydrogen, a C1-C60 alkyl group, a C2-C60 alkenyl group, a C6-C60 aryl group, and/or a C2-C60 heteroaryl group);
- definition of L301 may be the same as the definition of L201 as described in the present specification;
- R301 is selected from
- a C1-C20 alkyl group and/or a C1-C20 alkoxy group;
- a C1-C20 alkyl group and/or a C1-C20 alkoxy group, each substituted with at least one selected from a deuterium, —F, —Cl, —Br, —I, a hydroxyl group, a cyano group, a nitro group, an amino group, an amidino group, a hydrazine group, a hydrazone group, a carboxylic acid or a salt thereof, a sulfonic acid or a salt thereof, a phosphoric acid or a salt thereof, a phenyl group, a naphthyl group, a fluorenyl group, a spiro-fluorenyl group, a benzofluorenyl group, a dibenzofluorenyl group, a phenanthrenyl group, an anthracenyl group, a pyrenyl group, a chrysenyl group, a pyridinyl group, a pyrazinyl group, a pyrimidinyl group, a pyridazinyl group, a quinolinyl group, an isoquinolinyl group, a quinoxalinyl group, a quinazolinyl group, a carbazolyl group, and a triazinyl group;
- a phenyl group, a naphthyl group, a fluorenyl group, a spiro-fluorenyl group, a benzofluorenyl group, a dibenzofluorenyl group, a phenanthrenyl group, an anthracenyl group, a pyrenyl group, a chrysenyl group, a pyridinyl group, a pyrazinyl group, a pyrimidinyl group, a pyridazinyl group, a quinolinyl group, an isoquinolinyl group, a quinoxalinyl group, a quinazolinyl group, a carbazolyl group, and/or a triazinyl group; and/or
- a phenyl group, a naphthyl group, a fluorenyl group, a spiro-fluorenyl group, a benzofluorenyl group, a dibenzofluorenyl group, a phenanthrenyl group, an anthracenyl group, a pyrenyl group, a chrysenyl group, a pyridinyl group, a pyrazinyl group, a pyrimidinyl group, a pyridazinyl group, a quinolinyl group, an isoquinolinyl group, a quinoxalinyl group, a quinazolinyl group, a carbazolyl group, and/or a triazinyl group, each substituted with at least one selected from a deuterium, —F, —Cl, —Br, —I, a hydroxyl group, a cyano group, a nitro group, an amino group, an amidino group, a hydrazine group, a hydrazone group, a carboxylic acid or a salt thereof, a sulfonic acid or a salt thereof, a phosphoric acid or a salt thereof, a C1-C20 alkyl group, a C1-C20 alkoxy group, a phenyl group, a naphthyl group, a fluorenyl group, a spiro-fluorenyl group, a benzofluorenyl group, a dibenzofluorenyl group, a phenanthrenyl group, an anthracenyl group, a pyrenyl group, a chrysenyl group, a pyridinyl group, a pyrazinyl group, a pyrimidinyl group, a pyridazinyl group, a quinolinyl group, an isoquinolinyl group, a quinoxalinyl group, a quinazolinyl group, a carbazolyl group, and a triazinyl group;
- xb1 is selected from 0, 1, 2, and 3; and
- xb2 is selected from 1, 2, 3, and 4.
- In one embodiment, in Formula 301,
- L301 is selected from
- a phenylene group, a naphthylene group, a fluorenylene group, a spiro-fluorenylene group, a benzofluorenylene group, a dibenzofluorenylene group, a phenanthrenylene group, an anthracenylene group, a pyrenylene group, and/or a chrysenylene group; and/or
- a phenylene group, a naphthylene group, a fluorenylene group, a spiro-fluorenylene group, a benzofluorenylene group, a dibenzofluorenylene group, a phenanthrenylene group, an anthracenylene group, a pyrenylene group, and/or a chrysenylene group, each substituted with at least one selected from a deuterium, —F, —Cl, —Br, —I, a hydroxyl group, a cyano group, a nitro group, an amino group, an amidino group, a hydrazine group, a hydrazone group, a carboxylic acid or a salt thereof, a sulfonic acid or a salt thereof, a phosphoric acid or a salt thereof, a C1-C20 alkyl group, a C1-C20 alkoxy group, a phenyl group, a naphthyl group, a fluorenyl group, a spiro-fluorenyl group, a benzofluorenyl group, a dibenzofluorenyl group, a phenanthrenyl group, an anthracenyl group, a pyrenyl group, and a chrysenyl group;
- R301 is selected from
- a C1-C20 alkyl group and/or a C1-C20 alkoxy group;
- a C1-C20 alkyl group and/or a C1-C20 alkoxy group, each substituted with at least one selected from a deuterium, —F, —Cl, —Br, —I, a hydroxyl group, a cyano group, a nitro group, an amino group, an amidino group, a hydrazine group, a hydrazone group, a carboxylic acid or a salt thereof, a sulfonic acid or a salt thereof, a phosphoric acid or a salt thereof, a phenyl group, a naphthyl group, a fluorenyl group, a spiro-fluorenyl group, a benzofluorenyl group, a dibenzofluorenyl group, a phenanthrenyl group, an anthracenyl group, a pyrenyl group, and a chrysenyl group;
- a phenyl group, a naphthyl group, a fluorenyl group, a spiro-fluorenyl group, a benzofluorenyl group, a dibenzofluorenyl group, a phenanthrenyl group, an anthracenyl group, a pyrenyl group, and/or a chrysenyl group; and/or
- a phenyl group, a naphthyl group, a fluorenyl group, a spiro-fluorenyl group, a benzofluorenyl group, a dibenzofluorenyl group, a phenanthrenyl group, an anthracenyl group, a pyrenyl group, and/or a chrysenyl group, each substituted with at least one selected from a deuterium, —F, —Cl, —Br, —I, a hydroxyl group, a cyano group, a nitro group, an amino group, an am idino group, a hydrazine group, a hydrazone group, a carboxylic acid or a salt thereof, a sulfonic acid or a salt thereof, a phosphoric acid or a salt thereof, a C1-C20 alkyl group, a C1-C20 alkoxy group, a phenyl group, a naphthyl group, a fluorenyl group, a spiro-fluorenyl group, a benzofluorenyl group, a dibenzofluorenyl group, a phenanthrenyl group, an anthracenyl group, a pyrenyl group, and a chrysenyl group, but R301 is not limited thereto.
- For example, the host may include a compound represented by Formula 301A:
- In Formula 301A, definitions of the substituents may be as described in the present specification.
- The compound represented by Formula 301A may include at least one of Compounds H1 to H42, but the compound represented by Formula 301A is not limited thereto:
- Additionally, the host may include at least one of Compounds H43 to H49, but the host is not limited thereto:
- The dopant may include at least one of a fluorescent dopant and a phosphorescent dopant.
- The phosphorescent dopant may include an organic metal complex represented by Formula 401:
- In Formula 401,
- M is selected from iridium (Ir), platinum (Pt), osmium (Os), titanium (Ti), zirconium (Zr), hafnium (Hf), europium (Eu), terbium (Tb), and/or thulium (Tm);
- X401 to X404 are each independently a nitrogen atom or a carbon atom;
- rings A401 and A402 are each independently selected from a substituted or unsubstituted benzene, a substituted or unsubstituted naphthalene, a substituted or unsubstituted fluorene, a substituted or unsubstituted spiro-fluorene, a substituted or unsubstituted indene, a substituted or unsubstituted pyrrole, a substituted or unsubstituted thiophene, a substituted or unsubstituted furan, a substituted or unsubstituted imidazole, a substituted or unsubstituted pyrazole, a substituted or unsubstituted thiazole, a substituted or unsubstituted isothiazole, a substituted or unsubstituted oxazole, a substituted or unsubstituted isoxazole, a substituted or unsubstituted pyridine, a substituted or unsubstituted pyrazine, a substituted or unsubstituted pyrimidine, a substituted or unsubstituted pyridazine, a substituted or unsubstituted quinoline, a substituted or unsubstituted isoquinoline, a substituted or unsubstituted benzoquinoline, a substituted or unsubstituted quinoxaline, a substituted or unsubstituted quinazoline, a substituted or unsubstituted carbazole, a substituted or unsubstituted benzoimidazole, a substituted or unsubstituted benzofuran, a substituted or unsubstituted benzothiophene, a substituted or unsubstituted isobenzothiophene, a substituted or unsubstituted benzoxazole, a substituted or unsubstituted isobenzoxazole, a substituted or unsubstituted triazole, a substituted or unsubstituted oxadiazole, a substituted or unsubstituted triazine, a substituted or unsubstituted dibenzofuran, and a substituted or unsubstituted dibenzothiophene;
- at least one substituent of the substituted benzene, substituted naphthalene, substituted fluorene, substituted spiro-fluorene, substituted indene, substituted pyrrole, substituted thiophene, substituted furan, substituted imidazole, substituted pyrazole, substituted thiazole, substituted isothiazole, substituted oxazole, substituted isoxazole, substituted pyridine, substituted pyrazine, substituted pyrimidine, substituted pyridazine, substituted quinoline, substituted isoquinoline, substituted benzoquinoline, substituted quinoxaline, substituted quinazoline, substituted carbazole, substituted benzoimidazole, substituted benzofuran, substituted benzothiophene, substituted isobenzothiophene, substituted benzoxazole, substituted isobenzoxazole, substituted triazole, substituted oxadiazole, substituted triazine, substituted dibenzofuran, and/or substituted dibenzothiophene is selected from
- a deuterium, —F, —Cl, —Br, —I, a hydroxyl group, a cyano group, a nitro group, an amino group, an amidino group, a hydrazine group, a hydrazone group, a carboxylic acid or a salt thereof, a sulfonic acid or a salt thereof, a phosphoric acid or a salt thereof, a C1-C60 alkyl group, a C2-C60 alkenyl group, a C2-C60 alkynyl group, and/or a alkoxy group;
- a C1-C60 alkyl group, a C2-C60 alkenyl group, a C2-C60 alkynyl group, and/or a C1-C60 alkoxy group, each substituted with at least one of a deuterium, —F, —Cl, —Br, —I, a hydroxyl group, a cyano group, a nitro group, an amino group, an amidino group, a hydrazine group, a hydrazone group, a carboxylic acid or a salt thereof, a sulfonic acid or a salt thereof, a phosphoric acid or a salt thereof, a C3-C10 cycloalkyl group, a C2-C10 heterocycloalkyl group, a C3-C10 cycloalkenyl group, a C2-C10 heterocycloalkenyl group, a C6-C60 aryl group, a C6-C60 aryloxy group, a C6-C60 arylthio group, a C2-C60 heteroaryl group, a monovalent non-aromatic condensed polycyclic group, a monovalent non-aromatic heterocondensed polycyclic group, —N(Q401)(Q402), —Si(Q403)(Q404)(Q405), and —B(Q406)(Q407);
- a C3-C10 cycloalkyl group, a C2-C10 heterocycloalkyl group, a C3-C10 cycloalkenyl group, a C2-C10 heterocycloalkenyl group, a C6-C60 aryl group, a C6-C60 aryloxy group, a C6-C60 arylthio group, a C2-C60 heteroaryl group, a monovalent non-aromatic condensed polycyclic group and/or a monovalent non-aromatic heterocondensed polycyclic group;
- a C3-C10 cycloalkyl group, a C2-C10 heterocycloalkyl group, a C3-C10 cycloalkenyl group, a C2-C10 heterocycloalkenyl group, a C6-C60 aryl group, a C6-C60 aryloxy group, a C6-C60 arylthio group, a C2-C60 heteroaryl group, a monovalent non-aromatic condensed polycyclic group, and/or a monovalent non-aromatic heterocondensed polycyclic group, each substituted with at least one of a deuterium, —F, —Cl, —Br, —I, a hydroxyl group, a cyano group, a nitro group, an amino group, an am idino group, a hydrazine group, a hydrazone group, a carboxylic acid or a salt thereof, a sulfonic acid or a salt thereof, a phosphoric acid or a salt thereof, a C1-C60 alkyl group, a C2-C60 alkenyl group, a C2-C60 alkynyl group, a C1-C60 alkoxy group, a C3-C10 cycloalkyl group, a C2-C10 heterocycloalkyl group, a C3-C10 cycloalkenyl group, a C2-C10 heterocycloalkenyl group, a C6-C60 aryl group, a C6-C60 aryloxy group, a C6-C60 arylthio group, a C2-C60 heteroaryl group, a monovalent non-aromatic condensed polycyclic group, a monovalent non-aromatic heterocondensed polycyclic group, —N(Q411)(Q412), —Si(Q413)(Q414)(Q415), and —B(Q416)(Q417); and/or
- —N(Q421)(Q422), —Si(Q423)(Q424)(Q425), and/or —B(Q426)(Q427), where Q401 to Q407, Q411 to Q417, and Q421 to Q427 are defined as Q11 to Q17, Q21 to Q27, and Q31 to Q37 above;
- L401 is an organic ligand;
- xc1 is selected from 1, 2, and 3; and
- xc2 is selected from 0, 1, 2, and 3.
- In one embodiment, L401 is a monovalent, divalent, or trivalent organic ligand. For example, L401 may be selected from a halogen ligand, such as Cl or F, a diketone ligand, such as acetylacetonate, 1,3-diphenyl-1,3-propanedionate, 2,2,6,6-tetramethyl-3,5-heptanedionate, or hexafluoroacetonate, a carboxylic acid ligand, such as picolinate, dimethyl-3-pyrazolecarboxylate, or benzoate, a carbon monoxide ligand, an isonitrile ligand, a cyano ligand, and/or a phosphorus ligand, such as phosphine or phosphite, but L401 is not limited thereto.
- In Formula 401, when A401 has at least two substituents, the at least two substituents of A401 may be linked to each other to form a saturated or unsaturated ring.
- In Formula 401, when A402 has at least two substituents, the at least two substituents of A402 may be linked to each to and form a saturated or unsaturated ring.
- In Formula 401, when xc1 is 2 or greater, a plurality of ligands,
- may be identical to or different from each other, and A401 and A402 of one ligand may be linked to A401 and A402 of an adjacent ligand, respectively, directly (e.g. via a single bond)or via a connection group (e.g., a C1-C5 alkylene group, —N(R′)— (where, R′ is C1-C10 alkyl group or a C6-C20 aryl group), or —C(═O)—).
- In one embodiment, the phosphorescent dopant may be selected from Compounds PD1 to PD74 below, but is not limited thereto:
- Additionally, the phosphorescent dopant may include PtOEP illustrated below:
- The fluorescent dopant may include at least one of DPVBi, BDAVBi, TBPe, DCM, DCJTB, Coumarin 6, and C545T below:
- Additionally, the fluorescent dopant may include a compound represented by Formula 501 below:
- In Formula 501, Ar501 is selected from
- a naphthalene, a heptalene, a fluorene, a spiro-fluorene, a benzofluorene, a dibenzofluorene, a phenalene, a phenanthrene, an anthracene, a fluoranthene, a triphenylene, a pyrene, a chrysene, a naphthacene, a picene, a perylene group, a pentaphene, and/or an indenoanthracene;
- a naphthalene, a heptalene, a fluorene, a spiro-fluorene, a benzofluorene, a dibenzofluorene, a phenalene, a phenanthrene, an anthracene, a fluoranthene, a triphenylene, a pyrene, a chrysene, a naphthacene, a picene, a perylene, a pentaphene, and/or an indenoanthracene, each substituted with at least one selected from a deuterium, —F, —Cl, —Br, —I, a hydroxyl group, a cyano group, a nitro group, an amino group, an am idino group, a hydrazine group, a hydrazone group, a carboxylic acid or a salt thereof, a sulfonic acid or a salt thereof, a phosphoric acid or a salt thereof, a C1-C60 alkyl group, a C2-C60 alkenyl group, a C2-C60 alkynyl group, a C1-C60 alkoxy group, a C3-C10 cycloalkyl group, a C2-C10 heterocycloalkyl group, a C3-C10 cycloalkenyl group, a C2-C10 heterocycloalkenyl group, a C6-C60 aryl group, a C6-C60 aryloxy group, a C6-C60 arylthio group, a C2-C60 heteroaryl group, a monovalent non-aromatic condensed polycyclic group, a monovalent non-aromatic heterocondensed polycyclic group, and —Si(Q501)(Q502)(Q503) (where, Q501 to Q503 are each independently selected from a hydrogen, a C1-C60 alkyl group, a C2-C60 alkenyl group, a C6-C60 aryl group, and/or a C2-C60 heteroaryl group);
- definitions of L501 to L503 may be each independently the same as the definition of L201;
- R501 and R502 are each independently selected from
- a phenyl group, a naphthyl group, a fluorenyl group, a spiro-fluorenyl group, a benzofluorenyl group, a dibenzofluorenyl group, a phenanthrenyl group, an anthracenyl group, a pyrenyl group, a chrysenyl group, a pyridinyl group, a pyrazinyl group, a pyrimidinyl group, a pyridazinyl group, a quinolinyl group, an isoquinolinyl group, a quinoxalinyl group, a quinazolinyl group, a carbazolyl group, a triazinyl group, a dibenzofuranyl group, and/or a dibenzothiophenyl group; and/or
- a phenyl group, a naphthyl group, a fluorenyl group, a spiro-fluorenyl group, a benzofluorenyl group, a dibenzofluorenyl group, a phenanthrenyl group, an anthracenyl group, a pyrenyl group, a chrysenyl group, a pyridinyl group, a pyrazinyl group, a pyrimidinyl group, a pyridazinyl group, a quinolinyl group, an isoquinolinyl group, a quinoxalinyl group, a quinazolinyl group, a carbazolyl group, a triazinyl group, a dibenzofuranyl group, and/or a dibenzothiophenyl group, each substituted with at least one selected from a deuterium, —F, —Cl, —Br, —I, a hydroxyl group, a cyano group, a nitro group, an amino group, an amidino group, a hydrazine group, a hydrazone group, a carboxylic acid or a salt thereof, a sulfonic acid or a salt thereof, a phosphoric acid or a salt thereof, a C1-C20 alkyl group, a C1-C20 alkoxy group, a phenyl group, a naphthyl group, a fluorenyl group, a spiro-fluorenyl group, a benzofluorenyl group, a dibenzofluorenyl group, a phenanthrenyl group, an anthracenyl group, a pyrenyl group, a chrysenyl group, a pyridinyl group, a pyrazinyl group, a pyrimidinyl group, a pyridazinyl group, a quinolinyl group, an isoquinolinyl group, a quinoxalinyl group, a quinazolinyl group, a carbazolyl group, a triazinyl group, a dibenzofuranyl group, and a dibenzothiophenyl group;
- xd1 to xd3 are each independently selected from 0, 1, 2, and 3;
- xb4 is selected from 1, 2, 3, and 4.
- The fluorescent host may include at least one of Compounds FD1 to FD8, but is not limited thereto:
- The dopant may be present in the EML in an amount of about 0.01 part to about 15 parts by weight, based on about 100 parts by weight of the host, but the amount of the dopant is not limited thereto.
- A thickness of the EML may be about 100 Å to about 1,000 Å, for example, about 200 Å to about 600 Å. When the thickness of the EML is within any of these ranges, light-emitting properties of the organic light-emitting device may be improved, without a substantial increase in driving voltage.
- The mixed organic layer may be on the EML.
- The mixed organic layer may be formed on the EML using (utilizing) various methods such as, for example, vacuum deposition, spin coating, casting, LB deposition, inkjet printing, laser printing, or LITI. When the mixed organic layer is formed by methods such as vacuum deposition or spin coating, the deposition conditions and the coating conditions for forming the mixed organic layer may be similar to the deposition conditions and the coating conditions for forming the HIL.
- A compound for forming the mixed organic layer may be as described above.
- A thickness of the mixed organic layer may be about 5 Å to about 400 Å, for example, about 10 Å to about 40 Å. When the thickness of the mixed organic layer is within any of these ranges, light-emitting properties of the organic light-emitting device may be improved, without a substantial increase in driving voltage.
- A weight ratio of the hole-transporting compound to the electron-transporting compound in the mixed organic layer may be in the range of about 0.1:1 to about 10:1, but the respective amounts of the hole-transporting compound and the electron-transporting compound in the mixed organic layer are not limited thereto.
- An electron transport region may be positioned on the mixed organic layer.
- The electron transport region may include at least one selected from an HBL, an ETL, and an EIL, but is not limited thereto.
- For example, the electron transport region may have a structure of ETL/EIL or EBL/ETL/EIL, sequentially stacked on the EML or on the mixed organic layer, but the structure of the electron transport region is not limited thereto.
- In one embodiment, the
organic layer 150 of the organic light-emittingdevice 10 includes an electron transport region between the EML and thesecond electrode 190. The electron transport region may include at least one of an ETL and an EIL. - The ETL may include at least one selected from bathocuproine (BCP), bathophenanthroline (Bphen), Alq3, Balq, TAZ, and NTAZ below:
- Additionally, the ETL may include at least one compound selected from a group of compounds represented by Formula 601 and Formula 602:
-
Ar601-[(L601)xe1-E601]xe2 Formula 601 - In Formula 601, Ar601 is selected from
- a naphthalene, a heptalene, a fluorene, a spiro-fluorene, a benzofluorene, a dibenzofluorene, a phenalene, a phenanthrene, an anthracene, a fluoranthene, a triphenylene, a pyrene, a chrysene, a naphthacene, a picene, a perylene, a pentaphene, and/or an indenoanthracene;
- a naphthalene, a heptalene, a fluorene, a spiro-fluorene, a benzofluorene, a dibenzofluorene, a phenalene, a phenanthrene, an anthracene, a fluoranthene, a triphenylene, a pyrene, a chrysene, a naphthacene, a picene, a perylene, a pentaphene, and/or an indenoanthracene, each substituted with at least one selected from a deuterium, —F, —Cl, —Br, —I, a hydroxyl group, a cyano group, a nitro group, an amino group, an am idino group, a hydrazine group, a hydrazone group, a carboxylic acid or a salt thereof, a sulfonic acid or a salt thereof, a phosphoric acid or a salt thereof, a C1-C60 alkyl group, a C2-C60 alkenyl group, a C2-C60 alkynyl group, a C1-C60 alkoxy group, a C3-C10 cycloalkyl group, a C3-C10 heterocycloalkyl group, a C3-C10 cycloalkenyl group, a C3-C10 heterocycloalkenyl group, a C6-C60 aryl group, a C6-C60 aryloxy group, a C6-C60 arylthio group, a C2-C60 heteroaryl group, a monovalent non-aromatic condensed polycyclic group, a monovalent non-aromatic heterocondensed polycyclic group, and —Si(Q301)(Q302)(Q303) (where, Q301 to Q303 are each independently selected from a hydrogen, a C1-C60 alkyl group, a C2-C60 alkenyl group, a C6-C60 aryl group, and/or a C2-C60 heteroaryl group);
- definition of L601 may be the same as the definition of L201 above;
- E601 is selected from
- a pyrrolyl group, a thiophenyl group, a furanyl group, an imidazolyl group, a pyrazolyl group, a thiazolyl group, an isothiazolyl group, an oxazolyl group, an isoxazolyl group, a pyridinyl group, a pyrazinyl group, a pyrimidinyl group, a pyridazinyl group, an isoindolyl group, an indolyl group, an indazolyl group, a purinyl group, a quinolinyl group, an isoquinolinyl group, a benzoquinolinyl group, a phthalazinyl group, a naphthyridinyl group, a quinoxalinyl group, a quinazolinyl group, a cinnolinyl group, a carbazolyl group, a phenanthridinyl group, an acridinyl group, a phenanthrolinyl group, a phenazinyl group, a benzoimidazolyl group, a benzofuranyl group, a benzothiophenyl group, an isobenzothiazolyl group, a benzoxazolyl group, an isobenzoxazolyl group, a triazolyl group, a tetrazolyl group, an oxadiazolyl group, a triazinyl group, a dibenzofuranyl group, a dibenzothiophenyl group, a benzocarbazolyl group, a dibenzocarbazolyl group, a thiadiazolyl group, an imidazopyridinyl group, and/or an imidazopyrimidinyl group; and/or
- a pyrrolyl group, a thiophenyl group, a furanyl group, an imidazolyl group, a pyrazolyl group, a thiazolyl group, an isothiazolyl group, an oxazolyl group, an isoxazolyl group, a pyridinyl group, a pyrazinyl group, a pyrimidinyl group, a pyridazinyl group, an isoindolyl group, an indolyl group, an indazolyl group, a purinyl group, a quinolinyl group, an isoquinolinyl group, a benzoquinolinyl group, a phthalazinyl group, a naphthyridinyl group, a quinoxalinyl group, a quinazolinyl group, a cinnolinyl group, a carbazolyl group, a phenanthridinyl group, an acridinyl group, a phenanthrolinyl group, a phenazinyl group, a benzoimidazolyl group, a benzofuranyl group, a benzothiophenyl group, an isobenzothiazolyl group, a benzoxazolyl group, an isobenzoxazolyl group, a triazolyl group, a tetrazolyl group, an oxadiazolyl group, a triazinyl group, a dibenzofuranyl group, a dibenzothiophenyl group, a benzocarbazolyl group, a dibenzocarbazolyl group, a thiadiazolyl group, an imidazopyridinyl group, and/or an imidazopyrimidinyl group, each substituted with at least one selected from a deuterium, —F, —Cl, —Br, —I, a hydroxyl group, a cyano group, a nitro group, an amino group, an am idino group, a hydrazine group, a hydrazone group, a carboxylic acid or a salt thereof, a sulfonic acid or a salt thereof, a phosphoric acid or a salt thereof, a C1-C20 alkyl group, a C1-C20 alkoxy group, a cyclopentyl group, a cyclohexyl group, a cycloheptyl group, a cyclopentenyl group, a cyclohexenyl group, a phenyl group, a pentalenyl group, an indenyl group, a naphthyl group, an azulenyl group, a heptalenyl group, an indacenyl group, an acenaphthyl group, a fluorenyl group, a spiro-fluorenyl group, a benzofluorenyl group, a dibenzofluorenyl group, a phenalenyl group, a phenanthrenyl group, an anthracenyl group, a fluoranthenyl group, a triphenylenyl group, a pyrenyl group, a chrysenyl group, a naphthacenyl group, a picenyl group, a perylenyl group, a pentaphenyl group, a hexacenyl group, a pentacenyl group, a rubicenyl group, a coroneryl group, an ovalenyl group, a pyrrolyl group, a thiophenyl group, a furanyl group, an imidazolyl group, a pyrazolyl group, a thiazolyl group, an isothiazolyl group, an oxazolyl group, an isoxazolyl group, a pyridinyl group, a pyrazinyl group, a pyrimidinyl group, a pyridazinyl group, an isoindolyl group, an indolyl group, an indazolyl group, a purinyl group, a quinolinyl group, an isoquinolinyl group, a benzoquinolinyl group, a phthalazinyl group, a naphthyridinyl group, a quinoxalinyl group, a quinazolinyl group, a cinnolinyl group, a carbazolyl group, a phenanthridinyl group, an acridinyl group, a phenanthrolinyl group, a phenazinyl group, a benzoimidazolyl group, a benzofuranyl group, a benzothiophenyl group, an isobenzothiazolyl group, a benzoxazolyl group, an isobenzoxazolyl group, a triazolyl group, a tetrazolyl group, an oxadiazolyl group, a triazinyl group, a dibenzofuranyl group, a dibenzothiophenyl group, a benzocarbazolyl group, a dibenzocarbazolyl group, a thiadiazolyl group, an imidazopyridinyl group, and an imidazopyrimidinyl group;
- xe1 is selected from 0, 1, 2, and 3;
- xe2 is selected from 1, 2, 3, and 4.
- In Formula 602, X611 is N or C-(L611)xe611-R611, X612 is N or C-(L612)xe612-R612, X613 is N or C-(L613)xe613-R613, and at least one of X611 to X613 is N;
- definitions of each of L611 to L616 may be the same as the definition of L201 as described in the present specification;
- R611 to R616 are each independently selected from
- a phenyl group, a naphthyl group, a fluorenyl group, a spiro-fluorenyl group, a benzofluorenyl group, a dibenzofluorenyl group, a phenanthrenyl group, an anthracenyl group, a pyrenyl group, a chrysenyl group, a pyridinyl group, a pyrazinyl group, a pyrimidinyl group, a pyridazinyl group, a quinolinyl group, an isoquinolinyl group, a quinoxalinyl group, a quinazolinyl group, a carbazolyl group, and/or a triazinyl group; and/or
- a phenyl group, a naphthyl group, a fluorenyl group, a spiro-fluorenyl group, a benzofluorenyl group, a dibenzofluorenyl group, a phenanthrenyl group, an anthracenyl group, a pyrenyl group, a chrysenyl group, a pyridinyl group, a pyrazinyl group, a pyrimidinyl group, a pyridazinyl group, a quinolinyl group, an isoquinolinyl group, a quinoxalinyl group, a quinazolinyl group, a carbazolyl group, and/or a triazinyl group, each substituted with at least one selected from a deuterium, —F, —Cl, —Br, —I, a hydroxyl group, a cyano group, a nitro group, an amino group, an amidino group, a hydrazine group, a hydrazone group, a carboxylic acid or a salt thereof, a sulfonic acid or a salt thereof, a phosphoric acid or a salt thereof, a C1-C20 alkyl group, a C1-C20 alkoxy group, a phenyl group, a naphthyl group, a fluorenyl group, a spiro-fluorenyl group, a benzofluorenyl group, a dibenzofluorenyl group, a phenanthrenyl group, an anthracenyl group, a pyrenyl group, a chrysenyl group, a pyridinyl group, a pyrazinyl group, a pyrimidinyl group, a pyridazinyl group, a quinolinyl group, an isoquinolinyl group, a quinoxalinyl group, a quinazolinyl group, a carbazolyl group, and a triazinyl group;
- xe611 to xe616 are each independently selected from 0, 1, 2, and 3.
- The compound represented by Formula 601 and the compound represented by Formula 602 may be each independently selected from compounds ET1 to ET15:
- A thickness of the ETL may be about 100 Å to about 1,000 Å, for example, about 150 Å to about 500 Å. When the thickness of the ETL is within any of these ranges, electron transporting properties of the organic light-emitting device may be improved, without a substantial increase in driving voltage.
- The ETL may further include a metal-containing material, in addition to the materials described above.
- The metal-containing material may include a Li-complex. The Li-complex may include, for example, compound ET-D1 (lithium quinolate (LiQ)) or ET-D2:
- The electron transport region may include an HBL. When the EML includes a phosphorescent dopant, the HBL may prevent triplet excitons or holes from diffusing into the ETL.
- When the electron transport region includes the HBL, the HBL may be formed on the EML using (utilizing) various methods such as, for example, vacuum deposition, spin coating, casting, LB deposition, inkjet printing, laser printing, or LITI. When the HBL is formed by methods such as vacuum deposition or spin coating, the deposition conditions and the coating conditions for forming the HBL may be similar to the deposition conditions and the coating conditions for forming the HIL.
- The HBL may include, for example, at least one of BCP and Bphen below, but is not limited thereto:
- A thickness of the HBL may be about 20 Å to about 1,000 Å, for example, about 30 Å to about 300 Å. When the thickness of the HBL is within any of these ranges, the organic light-emitting device may exhibit good hole blocking properties, without a substantial increase in driving voltage.
- The electron transport region may include an ETL. The ETL may be formed on the EML or the HBL using (utilizing) various methods such as, for example, vacuum deposition, spin coating, casting, LB deposition, inkjet printing, laser printing, or LITI. When the ETL is formed by methods such as vacuum deposition or spin coating, the deposition conditions and the coating conditions for forming the ETL may be similar to the deposition conditions and the coating conditions for forming the HIL.
- The electron transport region may include an EIL that may facilitate the injection of electrons from the
second electrode 190. - The EIL may be formed on the ETL using (utilizing) various methods such as, for example, vacuum deposition, spin coating, casting, LB deposition, inkjet printing, laser printing, or LITI. When the EIL is formed by vacuum deposition or spin coating, the deposition conditions and the coating conditions for forming the EIL may be similar to the deposition conditions and the coating conditions for forming the HIL.
- The EIL may include at least one selected from LiF, NaCl, CsF, Li2O, BaO, and LiQ, but is not limited thereto.
- A thickness of the EIL may be about 1 Å to about 100 Å, for example, about 3 Å to about 90 Å. When the thickness of the EIL is within any of these ranges, the organic light-emitting device may exhibit good electron injecting properties, without a substantial increase in driving voltage.
- In one embodiment, the
second electrode 190 is on theorganic layer 150. Thesecond electrode 190 may be a cathode, which is an electron injection electrode. When thesecond electrode 190 is a cathode, a material for forming thesecond electrode 190 may include a metal, an alloy, an electric conducting compound, all having a low work function, and/or a mixture thereof. For example, thesecond electrode 190 may be a thin film formed of lithium (Li), magnesium (Mg), aluminum (Al), aluminum-lithium (Al—Li), calcium (Ca), magnesium-indium (Mg—In), and/or magnesium-silver (Mg—Ag), but thesecond electrode 190 is not limited thereto. Also, ITO and/or IZO may be used (utilized) as a material for forming thesecond electrode 190. Thesecond electrode 190 may be a reflective electrode, a semitransparent electrode, or a transparent electrode. - The organic layer of the organic light-emitting device according to embodiments of the present invention may be formed by a deposition method of the compound according to embodiments of the present invention, or by a wet method in which the organic light-emitting device is coated with the compound according to embodiments of the present invention that is first prepared as a solution.
- The organic light-emitting device according to embodiments of the present invention may be included in various types (kinds) of flat panel displays, for example, a passive matrix organic light-emitting display apparatus and/or an active matrix organic light-emitting display apparatus. When the organic light-emitting device is included in an active matrix organic light-emitting display apparatus, the first electrode located on the side of the substrate is a pixel electrode and may be electrically connected to a source electrode or a drain electrode of a thin film transistor. In one embodiment, the organic light-emitting device may be included in a flat panel display that may display images on both surfaces.
- Although the organic light-emitting device has been described with reference to the drawing, the organic light-emitting device of embodiments of the present invention is not limited thereto.
- Hereinafter, embodiments are illustrated with reference to certain examples. However, these examples are provided for illustrative purposes only, and should not in any sense be interpreted as limiting the scope of the present disclosure (the compounds used in the following examples can be obtained by one skilled in the art).
- ITO/HTM (120)/Host+ BD 5%(30)/Buffer (20)/Alq3 (20)/LiF (1)/Al (200)
- A transparent electrode of an indium tin oxide (ITO) having a thickness of 120 nm was formed on a glass substrate to prepare a cathode. Then, ultrasonic cleaning and pretreatment (UV-03 treatment and heat-treatment) were performed on the resulting cathode.
- Compound HTM (illustrated below) was deposited at a thickness of about 120 nm as an HTL on the pretreated cathode. Then, compound MADN (illustrated below) as a host, and compound BD as a dopant material, were co-deposited on the HTL in a total amount of 5% to form an EML having a thickness of about 30 nm. Compound BF1 (illustrated below) and compound BF9 (illustrated below) were deposited on the EML at a ratio of 1:1 to form a buffer having a thickness of about 20 nm, and then Alq was deposited on the buffer as an ETL having a thickness of about 20 nm. Next, lithium fluoride was deposited on the ETL to form an EIL having a thickness of about 1 nm, and subsequently, aluminum was deposited at a thickness of about 200 nm on the EIL, thereby manufacturing an organic light-emitting device.
- Material Properties of the buffer layer were measured using (utilizing) the following methods, and the results are shown in Table 1.
- An organic material was irradiated with light, and the amount of electrons generated by charge separation was measured.
- Energy gaps were measured from edges of the UV absorption spectra of the materials.
- Electron affinities were calculated using the following equation:
-
EA(eV)=IP−Eg, - where IP is the ionization potential calculated in (1) and Eg is the energy gap calculated in (2).
- A conversion equation for calculating triplet energy (ET) is as follows:
-
ET(eV)=1239.85/λedge, - where λedg denotes a wavelength value at a point of intersection of a tangent and a horizontal axis, where the tangent is according to a slope of a short wavelength side of a phosphorescent spectrum.
-
TABLE 1 Material EA (eV) IP (eV) Eg (eV) ET (eV) BF1 3 6.1 3.1 2.67 BF2 2.21 5.84 3.63 2.64 BF3 2.1 5.5 3.4 2.9 BF4 2.35 5.67 3.32 2.87 BF5 2.4 5.9 3.5 3 BF6 2 5.5 3.5 2.9 BF7 2.8 5.9 3.1 1.8 BF8 2.5 5.7 3.2 2.6 BF9 2.77 5.49 2.72 2.67 BF10 2.9 6.4 3.5 2.5 BF11 2.7 6.3 3.6 2.7 BF12 3 6.12 3.12 2.27 - ITO/HTM (120)/Host+ BD 5%(30)/Buffer (20)/Alq3 (20)/LiF (1)/Al (200)
- Organic light-emitting devices for each of Examples 1-2 to 1-14 and Comparative Examples 1 to 3 were manufactured as in Example 1-1, except that the buffer layers were formed as shown in Table 2.
-
TABLE 2 Effi- Driving ciency voltage T90 EML Buffer (cd/A) (V) (hr) Example 1-1 MADN + BD BF1 + BF9 5.5 4.5 98 Example 1-2 MADN + BD BF2 + BF9 5.9 4.5 110 Example 1-3 MADN + BD BF3 + BF9 5.7 4.6 85 Example 1-4 MADN + BD BF4 + BF9 5.8 4.5 106 Example 1-5 MADN + BD BF5 + BF9 5.5 4.6 94 Example 1-6 MADN + BD BF6 + BF9 5.6 4.5 86 Example 1-7 MADN + BD BF4 + BF7 5.6 4.3 81 Example 1-8 MADN + BD BF5 + BF8 5.8 4.4 103 Example 1-9 MADN + BD BF6 + BF8 5.5 4.4 98 Example 1-10 MADN + BD BF4 + BF10 5.7 4.4 92 Example 1-11 MADN + BD BF4 + BF11 5.4 4.3 78 Example 1-12 MADN + BD BF4 + BF12 5.6 4.6 96 Example 1-13 MADN + BD BF7 + BF9 5.4 4.2 82 Example 1-14 MADN + BD BF8 + BF9 5.6 4.3 98 Comparative MADN + BD Alq3 4.5 4.8 35 Example 1 Comparative MADN + BD BF7 4.8 4.7 48 Example 2 Comparative MADN + BD BF4 4.8 5.0 29 Example 3 - Efficiencies (cd/A), driving voltages (V), and lifespans (hour) of the organic light-emitting devices prepared in Examples 1-2 to 1-14 and Comparative Examples 1 to 3 were each evaluated, and the results are shown in Table 2.
- ITO/HTM (120)/Host+ Ir(ppy)3_10% (30)/Buffer (20)/Alq3 (20)/LiF (1)/Al (200)
- Organic light-emitting devices were manufactured as in Example 1-1, except that the EML, the host, the dopant, and the buffer layer were formed as shown in Table 3, and the dopant material Ir(ppy)3 was deposited at a concentration of 10%, instead of 5%. When the host is formed of two different compounds, a weight ratio of the compounds is 1:1.
-
TABLE 3 Effi- Driving ciency voltage T90 EML Buffer (cd/A) (V) (hr) Example 2-1 CBP + Ir(ppy)3 BF4 + BF7 55 4.8 165 Example 2-2 CBP + Ir(ppy)3 BF5 + BF8 57 5.0 138 Example 2-3 CBP + Ir(ppy)3 BF6 + BF8 55 4.8 151 Example 3-1 PH1 + Ir(ppy)3 BF4 + BF7 57 5.2 181 Example 3-2 PH1 + Ir(ppy)3 BF5 + BF8 58 5.1 144 Example 3-3 PH1 + Ir(ppy)3 BF6 + BF8 55 5.1 160 Example 4-1 PH2 + Ir(ppy)3 BF4 + BF7 61 4.5 120 Example 4-2 PH2 + Ir(ppy)3 BF5 + BF8 63 4.8 137 Example 4-3 PH2 + Ir(ppy)3 BF6 + BF8 60 4.7 118 Example 5-1 CBP + PH1 + BF4 + BF7 68 4.5 177 Ir(ppy)3 Example 5-2 CBP + PH1 + BF5 + BF8 66 4.5 201 Ir(ppy)3 Example 5-3 CBP + PH1 + BF6 + BF8 65 4.6 165 Ir(ppy)3 Example 6 BF5 + BF8 + BF5 + BF8 63 4.3 173 Ir(ppy)3 Comparative CBP + Ir(ppy)3 Alq3 44 5.7 49 Example 4 Comparative CBP + Ir(ppy)3 BF7 48 5.3 66 Example 5 Comparative CBP + Ir(ppy)3 BF4 52 6.1 87 Example 6 - Efficiencies (cd/A), driving voltages (V), and lifespans (hour) of the organic light-emitting devices prepared in Examples 2-1 to 2-6 and Comparative Examples 4 to 6 were each evaluated, and the results are shown in Table 3.
- ITO/HTM (120)/Host+ Ir(pq)2acac_5% (30)/Buffer (20)/Alq3 (20)/LiF (1)/Al (200)
- Organic light-emitting devices were manufactured as in Example 1-1, except that the EML, the host, the dopant, and the buffer layer were formed as shown in Table 4, and the dopant material Ir(pq)2acac was deposited at a concentration of 5%.
-
TABLE 4 Effi- Driving ciency voltage T90 EML Buffer (cd/A) (V) (hr) Example 7-1 CBP + BF4 + BF7 23.1 5.3 151 Ir(pq)2acac Example 7-2 CBP + BF5 + BF8 22.5 5.4 163 Ir(pq)2acac Example 7-3 CBP + BF6 + BF8 24.3 5.3 170 Ir(pq)2acac Example 8-1 PH1 + BF4 + BF7 23.3 5.4 225 Ir(pq)2acac Example 8-2 PH1 + BF5 + BF8 21.8 5.5 166 Ir(pq)2acac Example 8-3 PH1 + BF6 + BF8 24.0 5.5 191 Ir(pq)2acac Example 9-1 PH2 + BF4 + BF7 25.1 5.1 243 Ir(pq)2acac Example 9-2 PH2 + BF5 + BF8 24.8 5.0 288 Ir(pq)2acac Example 9-3 PH2 + BF6 + BF8 23.5 4.9 260 Ir(pq)2acac Comparative CBP + Alq3 15.3 5.9 118 Example 7 Ir(pq)2acac Comparative CBP + BF7 19.8 5.3 95 Example 8 Ir(pq)2acac Comparative CBP + BF4 18.0 6.5 76 Example 9 Ir(pq)2acac - Efficiencies (cd/A), driving voltages (V), and lifespans (hour) of the organic light-emitting devices prepared in Examples 7-1 to 9-3 and Comparative Examples 7 to 9 were each evaluated, and the results are shown in Table 4.
- Referring to the results shown in Tables 2 to 4, the organic light-emitting devices prepared in Examples 1-1 to 9-3 showed improved efficiency and lifespan characteristics compared to those of the organic light-emitting devices prepared in Comparative Examples 1 to 9.
- As described above, the organic light-emitting device according to one or more embodiments of the present invention may have a low driving voltage, a high efficiency, and a long lifespan.
- It should be understood that the exemplary embodiments described therein should be considered in a descriptive sense only and not for purposes of limitation. Descriptions of features or aspects within each embodiment should typically be considered as available for other similar features or aspects in other embodiments.
- While one or more embodiments of the present invention have been described with reference to the figures, it will be understood by those of ordinary skill in the art that various changes in form and details may be made therein without departing from the spirit and scope of the present disclosure as defined by the following claims and equivalents thereof.
Claims (19)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US16/105,931 US20180358564A1 (en) | 2014-05-02 | 2018-08-20 | Organic light-emitting device |
Applications Claiming Priority (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR10-2014-0053616 | 2014-05-02 | ||
| KR1020140053616A KR102244071B1 (en) | 2014-05-02 | 2014-05-02 | Organic light emitting device |
| US14/497,267 US9564598B2 (en) | 2014-05-02 | 2014-09-25 | Organic light-emitting device |
| US15/425,324 US10056562B2 (en) | 2014-05-02 | 2017-02-06 | Organic light-emitting device |
| US16/105,931 US20180358564A1 (en) | 2014-05-02 | 2018-08-20 | Organic light-emitting device |
Related Parent Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US15/425,324 Continuation US10056562B2 (en) | 2014-05-02 | 2017-02-06 | Organic light-emitting device |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| US20180358564A1 true US20180358564A1 (en) | 2018-12-13 |
Family
ID=54355870
Family Applications (3)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US14/497,267 Active US9564598B2 (en) | 2014-05-02 | 2014-09-25 | Organic light-emitting device |
| US15/425,324 Active US10056562B2 (en) | 2014-05-02 | 2017-02-06 | Organic light-emitting device |
| US16/105,931 Abandoned US20180358564A1 (en) | 2014-05-02 | 2018-08-20 | Organic light-emitting device |
Family Applications Before (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US14/497,267 Active US9564598B2 (en) | 2014-05-02 | 2014-09-25 | Organic light-emitting device |
| US15/425,324 Active US10056562B2 (en) | 2014-05-02 | 2017-02-06 | Organic light-emitting device |
Country Status (2)
| Country | Link |
|---|---|
| US (3) | US9564598B2 (en) |
| KR (1) | KR102244071B1 (en) |
Families Citing this family (63)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR20120100709A (en) | 2010-01-15 | 2012-09-12 | 이데미쓰 고산 가부시키가이샤 | Organic electroluminescent element |
| KR102120894B1 (en) * | 2013-05-03 | 2020-06-10 | 삼성디스플레이 주식회사 | Organic light emitting device |
| KR102244071B1 (en) * | 2014-05-02 | 2021-04-26 | 삼성디스플레이 주식회사 | Organic light emitting device |
| KR101709379B1 (en) * | 2014-10-01 | 2017-02-23 | 주식회사 엘지화학 | Organic light emitting device |
| KR102363259B1 (en) * | 2014-12-02 | 2022-02-16 | 삼성디스플레이 주식회사 | Organic light emitting device |
| WO2016186321A1 (en) | 2015-05-19 | 2016-11-24 | Rohm And Haas Electronic Materials Korea Ltd. | Phosphorous host material and organic electroluminescent device comprising the same |
| KR20160136211A (en) * | 2015-05-19 | 2016-11-29 | 롬엔드하스전자재료코리아유한회사 | Phosphorous Host Material and Organic Electroluminescent Device Comprising the Same |
| KR20170001552A (en) * | 2015-06-26 | 2017-01-04 | 롬엔드하스전자재료코리아유한회사 | Multi-component host material and organic electroluminescent device comprising the same |
| WO2017078403A1 (en) * | 2015-11-03 | 2017-05-11 | Rohm And Haas Electronic Materials Korea Ltd. | A plurality of host materials and organic electroluminescent device comprising the same |
| KR102744947B1 (en) * | 2015-11-03 | 2024-12-23 | 듀폰스페셜티머터리얼스코리아 유한회사 | A plurality of host materials and organic electroluminescent device comprising the same |
| KR101764908B1 (en) | 2015-11-11 | 2017-08-07 | 주식회사 스킨앤스킨 | 2-phenanthrene carbazole derivative compound and organic electroluminescent device including the same |
| CN108368088A (en) * | 2015-12-08 | 2018-08-03 | 喜星素材株式会社 | Heterocyclic compound and organic light emitting device using same |
| KR20170069342A (en) | 2015-12-10 | 2017-06-21 | 삼성디스플레이 주식회사 | Organic light-emitting device |
| KR102419178B1 (en) * | 2015-12-29 | 2022-07-11 | 삼성디스플레이 주식회사 | Organic light-emitting device |
| JP6716138B2 (en) * | 2016-01-29 | 2020-07-01 | 国立大学法人山形大学 | Terpyridine derivative, light emitting material comprising the same, and organic EL device using the same |
| KR101999709B1 (en) * | 2016-03-21 | 2019-07-12 | 주식회사 엘지화학 | Organic light emitting device |
| KR102643972B1 (en) * | 2016-03-30 | 2024-03-07 | 솔루스첨단소재 주식회사 | Organic light-emitting compound and organic electroluminescent device using the same |
| KR102044057B1 (en) * | 2016-04-28 | 2019-11-12 | 주식회사 엘지화학 | Organic light emitting device |
| KR102120517B1 (en) * | 2016-04-28 | 2020-06-08 | 주식회사 엘지화학 | Organic light emitting device |
| CN106632262B (en) * | 2016-11-28 | 2019-05-31 | 江苏师范大学 | A class of triarylpyridine derivatives containing carbazole skeleton and preparation method and application thereof |
| CN106967021A (en) * | 2017-03-29 | 2017-07-21 | 江苏三月光电科技有限公司 | A kind of organic compound and its application using equal benzene as core |
| CN106977527B (en) * | 2017-04-25 | 2019-11-08 | 江西冠能光电材料有限公司 | A kind of organic semiconductor compound and the organic electroluminescence device using the compound |
| KR102536248B1 (en) | 2017-06-21 | 2023-05-25 | 삼성디스플레이 주식회사 | Heterocyclic compound and organic light emitting device comprising the same |
| CN114702480B (en) * | 2017-07-14 | 2024-10-18 | 三星显示有限公司 | Organic molecules, especially for optoelectronic devices |
| KR102415376B1 (en) | 2017-08-04 | 2022-07-01 | 삼성디스플레이 주식회사 | Condensed-cyclic compound and organic light emitting device comprising the same |
| KR102414108B1 (en) * | 2017-08-08 | 2022-06-29 | 삼성디스플레이 주식회사 | Heterocyclic compound and organic light-emitting device comprising the same |
| CN110520417B (en) * | 2017-08-28 | 2022-04-26 | 株式会社Lg化学 | Heterocyclic compound and organic light-emitting element using same |
| KR20190029422A (en) * | 2017-09-11 | 2019-03-20 | 롬엔드하스전자재료코리아유한회사 | Organic electroluminescent compound and organic electroluminescent device comprising the same |
| CN118027042A (en) * | 2017-09-11 | 2024-05-14 | 罗门哈斯电子材料韩国有限公司 | Organic electroluminescent compound and organic electroluminescent device comprising the same |
| WO2019121108A1 (en) * | 2017-12-20 | 2019-06-27 | Cynora Gmbh | Organic molecules for use in optoelectronic devices |
| US11706977B2 (en) | 2018-01-11 | 2023-07-18 | Samsung Electronics Co., Ltd. | Heterocyclic compound, composition including the same, and organic light-emitting device including the heterocyclic compound |
| KR102536246B1 (en) | 2018-03-23 | 2023-05-25 | 삼성디스플레이 주식회사 | Heterocyclic compound and organic light emitting device comprising the same |
| CN108863918B (en) * | 2018-06-19 | 2022-02-11 | 长春海谱润斯科技股份有限公司 | Arylamine derivative and organic electroluminescent device thereof |
| KR102209928B1 (en) * | 2018-07-09 | 2021-02-01 | 주식회사 엘지화학 | Compound and organic light emitting device comprising the same |
| CN110713486A (en) * | 2018-07-12 | 2020-01-21 | 苏州大学 | A kind of pyrimidine derivative with self-assembly property, preparation method and application thereof |
| KR102777743B1 (en) | 2018-08-10 | 2025-03-11 | 삼성디스플레이 주식회사 | Organic electroluminescence device and condensed cyclic compound for organic electroluminescence device |
| KR102628848B1 (en) | 2018-08-10 | 2024-01-25 | 삼성디스플레이 주식회사 | Condensed compound and organic light-emitting device including the same |
| KR20200018275A (en) * | 2018-08-10 | 2020-02-19 | 롬엔드하스전자재료코리아유한회사 | Organic electroluminescent compound and organic electroluminescent device comprising the same |
| CN109053739A (en) * | 2018-09-19 | 2018-12-21 | 西安瑞联新材料股份有限公司 | A kind of pyrrolotriazine derivatives and its application in OLED device |
| US12501828B2 (en) | 2018-12-28 | 2025-12-16 | Samsung Electronics Co., Ltd. | Organic light-emitting device and apparatus including the same |
| CN109456256A (en) * | 2018-12-28 | 2019-03-12 | 武汉天马微电子有限公司 | Compound, display panel and display device |
| JP7299020B2 (en) * | 2018-12-28 | 2023-06-27 | 三星電子株式会社 | Organic electroluminescence device and manufacturing method thereof |
| CN109879857A (en) * | 2019-03-29 | 2019-06-14 | 武汉华星光电半导体显示技术有限公司 | Hot activation delayed fluorescence material and preparation method thereof, electroluminescent device |
| CN112300171B (en) * | 2019-07-30 | 2023-08-15 | 江苏三月科技股份有限公司 | Organic compound based on carbazolo ring structure and application of organic compound to OLED |
| KR102798042B1 (en) * | 2019-09-06 | 2025-04-22 | 삼성디스플레이 주식회사 | Organic light-emitting device and apparatus including the same |
| EP3798213B1 (en) * | 2019-09-26 | 2024-01-10 | Novaled GmbH | Organic semiconductor layer, organic electronic device comprising the same and compounds therefor |
| KR102869108B1 (en) * | 2019-10-24 | 2025-10-14 | 듀폰스페셜티머터리얼스코리아 유한회사 | A plurality of host materials and organic electroluminescent device comprising the same |
| KR102720072B1 (en) * | 2019-11-05 | 2024-10-23 | 삼성디스플레이 주식회사 | Organic light emitting device and method for manufacturing the same |
| KR102857112B1 (en) * | 2019-12-12 | 2025-09-09 | 삼성디스플레이 주식회사 | Organic light-emitting device and apparatus including the same |
| KR102776384B1 (en) * | 2020-05-06 | 2025-03-10 | 삼성디스플레이 주식회사 | Llight emitting device and electronic apparatus comprising same |
| US20230165140A1 (en) * | 2020-06-03 | 2023-05-25 | Lg Chem, Ltd. | Compound and organic light-emitting device comprising same |
| CN112442023B (en) * | 2020-11-30 | 2021-08-24 | 长春海谱润斯科技股份有限公司 | Heterocyclic derivative and organic electroluminescent device thereof |
| CN112661714B (en) * | 2020-12-22 | 2022-01-07 | 长春海谱润斯科技股份有限公司 | Heterocyclic compound and organic electroluminescent device containing same |
| KR102841162B1 (en) * | 2020-12-23 | 2025-07-31 | 삼성디스플레이 주식회사 | Organic light-emitting device and apparatus including the same |
| CN114981274A (en) * | 2020-12-25 | 2022-08-30 | 京东方科技集团股份有限公司 | Organic electroluminescent device, display panel, display apparatus, and light-emitting apparatus |
| CN113024566B (en) * | 2021-01-28 | 2021-11-30 | 陕西莱特光电材料股份有限公司 | Nitrogen-containing compound, electronic element comprising same and electronic device |
| KR20220118591A (en) * | 2021-02-18 | 2022-08-26 | 삼성디스플레이 주식회사 | Nitrogen containing compound and light emitting device including the same |
| US20220310934A1 (en) * | 2021-02-18 | 2022-09-29 | Samsung Display Co., Ltd. | Compound including nitrogen emitting device including the same |
| CN113336744B (en) * | 2021-06-23 | 2024-04-26 | 长春海谱润斯科技股份有限公司 | Heterocyclic derivative and organic electroluminescent device thereof |
| CN113717093B (en) * | 2021-07-12 | 2024-04-05 | 阜阳欣奕华材料科技有限公司 | Compound and organic electroluminescent device, display device |
| CN113845512B (en) * | 2021-09-18 | 2023-05-05 | 长春海谱润斯科技股份有限公司 | Compound containing heterocycle and organic electroluminescent device thereof |
| KR20230066199A (en) * | 2021-11-05 | 2023-05-15 | 삼성디스플레이 주식회사 | Light emitting device and electronic apparatus comprising same |
| CN114105891B (en) * | 2021-12-02 | 2024-01-26 | 长春海谱润斯科技股份有限公司 | Fluorene derivative and organic electroluminescent device thereof |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20040137270A1 (en) * | 2002-12-24 | 2004-07-15 | Lg Electronics Inc. | Organic electroluminescent device |
| US20050116291A1 (en) * | 2003-11-29 | 2005-06-02 | Jae-Bon Koo | Organic electroluminescence display device and method for fabricating the same |
| WO2009072587A1 (en) * | 2007-12-03 | 2009-06-11 | Semiconductor Energy Laboratory Co., Ltd. | Carbazole derivative, and light-emitting element, light-emitting device, and electronic device using carbazole derivative |
| CN102757300A (en) * | 2012-05-31 | 2012-10-31 | 吉林奥来德光电材料股份有限公司 | Benzophenanthrene derivative, preparation method and light emitting device made thereof |
| US10431766B2 (en) * | 2014-05-02 | 2019-10-01 | Samsung Display Co., Ltd. | Organic light-emitting device |
Family Cites Families (25)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| SG115435A1 (en) * | 2000-12-28 | 2005-10-28 | Semiconductor Energy Lab | Luminescent device |
| KR100713989B1 (en) * | 2005-07-15 | 2007-05-04 | 삼성에스디아이 주식회사 | White organic light emitting device and its manufacturing method |
| CN101258622A (en) | 2005-09-08 | 2008-09-03 | 出光兴产株式会社 | organic electroluminescent element |
| JP5417702B2 (en) * | 2006-11-02 | 2014-02-19 | 三菱化学株式会社 | Organic electroluminescent device, organic electroluminescent layer coating solution, color display device |
| US20080284318A1 (en) * | 2007-05-17 | 2008-11-20 | Deaton Joseph C | Hybrid fluorescent/phosphorescent oleds |
| JP5530608B2 (en) * | 2007-09-13 | 2014-06-25 | 株式会社半導体エネルギー研究所 | Light emitting element and light emitting device |
| US20090191427A1 (en) * | 2008-01-30 | 2009-07-30 | Liang-Sheng Liao | Phosphorescent oled having double hole-blocking layers |
| KR20090098588A (en) * | 2008-03-14 | 2009-09-17 | 삼성전자주식회사 | Novel organic compound and organic light emitting device using same |
| KR101620091B1 (en) * | 2008-07-24 | 2016-05-13 | 삼성디스플레이 주식회사 | An aromatic heterocyclic compound and an organic light emitting diode comprising an organic layer comprising the same |
| KR101420327B1 (en) * | 2008-07-24 | 2014-08-14 | 삼성디스플레이 주식회사 | A method for preparing an organic light emitting device and am organic light emitting device |
| KR20120100709A (en) | 2010-01-15 | 2012-09-12 | 이데미쓰 고산 가부시키가이샤 | Organic electroluminescent element |
| JP5735241B2 (en) * | 2010-09-08 | 2015-06-17 | ユー・ディー・シー アイルランド リミテッド | Organic electroluminescent device and charge transport material |
| JP5405653B1 (en) | 2010-11-22 | 2014-02-05 | 出光興産株式会社 | Organic electroluminescence device |
| US20120126205A1 (en) | 2010-11-22 | 2012-05-24 | Idemitsu Kosan Co., Ltd. | Organic electroluminescence device |
| WO2012176818A1 (en) | 2011-06-24 | 2012-12-27 | 出光興産株式会社 | Organic electroluminescent element |
| KR101830790B1 (en) | 2011-06-30 | 2018-04-05 | 삼성디스플레이 주식회사 | Organic light-emitting diode and flat display device comprising the same |
| KR101358784B1 (en) | 2012-02-14 | 2014-02-10 | 삼성디스플레이 주식회사 | Organic light-emitting device having improved efficiency characterisitics and organic light-emitting display apparatus including the same |
| KR102025834B1 (en) * | 2012-06-29 | 2019-09-27 | 삼성디스플레이 주식회사 | Novel organic emitting compound, and organic light emitting diode comprising the same |
| KR101363544B1 (en) | 2012-08-23 | 2014-02-17 | 서강대학교산학협력단 | Method for formating porous membrane for generation of multiple chemical gradients in microchannel using spatially controlled self-assembly of particles and microchannel device |
| CN103915470B (en) * | 2012-12-31 | 2016-12-07 | 乐金显示有限公司 | Organic light-emitting display device |
| KR102014721B1 (en) | 2013-01-29 | 2019-10-21 | 삼성전자 주식회사 | Programmable intelligent storage architecture based on application and business requirements |
| KR102147839B1 (en) * | 2013-03-05 | 2020-08-26 | 삼성디스플레이 주식회사 | Compound and organic light emitting device comprising the same |
| KR102116496B1 (en) | 2013-08-26 | 2020-05-29 | 삼성디스플레이 주식회사 | Organic light emitting device |
| KR102065366B1 (en) * | 2013-08-30 | 2020-01-13 | 엘지디스플레이 주식회사 | Organic light emitting device |
| KR102244071B1 (en) * | 2014-05-02 | 2021-04-26 | 삼성디스플레이 주식회사 | Organic light emitting device |
-
2014
- 2014-05-02 KR KR1020140053616A patent/KR102244071B1/en active Active
- 2014-09-25 US US14/497,267 patent/US9564598B2/en active Active
-
2017
- 2017-02-06 US US15/425,324 patent/US10056562B2/en active Active
-
2018
- 2018-08-20 US US16/105,931 patent/US20180358564A1/en not_active Abandoned
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20040137270A1 (en) * | 2002-12-24 | 2004-07-15 | Lg Electronics Inc. | Organic electroluminescent device |
| US20050116291A1 (en) * | 2003-11-29 | 2005-06-02 | Jae-Bon Koo | Organic electroluminescence display device and method for fabricating the same |
| WO2009072587A1 (en) * | 2007-12-03 | 2009-06-11 | Semiconductor Energy Laboratory Co., Ltd. | Carbazole derivative, and light-emitting element, light-emitting device, and electronic device using carbazole derivative |
| CN102757300A (en) * | 2012-05-31 | 2012-10-31 | 吉林奥来德光电材料股份有限公司 | Benzophenanthrene derivative, preparation method and light emitting device made thereof |
| US10431766B2 (en) * | 2014-05-02 | 2019-10-01 | Samsung Display Co., Ltd. | Organic light-emitting device |
| US11316124B2 (en) * | 2014-05-02 | 2022-04-26 | Samsung Display Co., Ltd. | Organic light-emitting device |
Non-Patent Citations (2)
| Title |
|---|
| CN-102757300-A machine translation (Year: 2012) * |
| Li et al. Adv.Funct.Mater.2005,15, No. 3, March (Year: 2005) * |
Also Published As
| Publication number | Publication date |
|---|---|
| KR102244071B1 (en) | 2021-04-26 |
| KR20150126527A (en) | 2015-11-12 |
| US20170148999A1 (en) | 2017-05-25 |
| US9564598B2 (en) | 2017-02-07 |
| US10056562B2 (en) | 2018-08-21 |
| US20150318510A1 (en) | 2015-11-05 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US12262578B2 (en) | Organic light-emitting device | |
| US10056562B2 (en) | Organic light-emitting device | |
| US9728729B2 (en) | Organic light-emitting device | |
| US9978955B2 (en) | Organic light-emitting device | |
| US20160005979A1 (en) | Organic light-emitting device | |
| US10727417B2 (en) | Organic light-emitting device | |
| US20150318508A1 (en) | Organic light-emitting device | |
| US20150318486A1 (en) | Organic light-emitting device | |
| US20150325798A1 (en) | Organic light-emitting devices | |
| US12268044B2 (en) | Organic light-emitting device including a first exciplex and a second exciplex in an emission layer | |
| US20150333266A1 (en) | Organic light-emitting device | |
| US9711734B2 (en) | Organic light-emitting device | |
| US9847491B2 (en) | Organic light-emitting device | |
| US20160190481A1 (en) | Compound and organic light-emitting device comprising the compound | |
| US9825240B2 (en) | Organic light-emitting device | |
| US20170012211A1 (en) | Organic light-emitting device | |
| US9515271B2 (en) | Organic light-emitting device | |
| US10236453B2 (en) | Dibenzoborole-based compound and organic light emitting device including the same | |
| US11114625B2 (en) | Organic light-emitting device | |
| US10249829B2 (en) | Compound and organic light-emitting device including the same | |
| US20200185619A1 (en) | Organic light-emitting device | |
| US9887365B2 (en) | Organic light-emitting device | |
| US12075693B2 (en) | Organometallic compound and organic light-emitting device including the same | |
| US10181562B2 (en) | Compound and organic light-emitting device comprising the same | |
| KR20210046614A (en) | Organic light emitting device |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: DOCKETED NEW CASE - READY FOR EXAMINATION |
|
| AS | Assignment |
Owner name: SAMSUNG DISPLAY CO., LTD., KOREA, REPUBLIC OF Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNORS:ITO, NAOYUKI;KIM, SEUL-ONG;KIM, YOUN-SUN;AND OTHERS;REEL/FRAME:049189/0320 Effective date: 20140829 |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: NON FINAL ACTION MAILED |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: RESPONSE TO NON-FINAL OFFICE ACTION ENTERED AND FORWARDED TO EXAMINER |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: FINAL REJECTION MAILED |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: RESPONSE AFTER FINAL ACTION FORWARDED TO EXAMINER |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: ADVISORY ACTION MAILED |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: DOCKETED NEW CASE - READY FOR EXAMINATION |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: NON FINAL ACTION MAILED |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: FINAL REJECTION MAILED |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: ADVISORY ACTION MAILED |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: DOCKETED NEW CASE - READY FOR EXAMINATION |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: NON FINAL ACTION MAILED |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: RESPONSE TO NON-FINAL OFFICE ACTION ENTERED AND FORWARDED TO EXAMINER |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: FINAL REJECTION MAILED |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: ADVISORY ACTION MAILED |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: DOCKETED NEW CASE - READY FOR EXAMINATION |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: NON FINAL ACTION MAILED |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: RESPONSE TO NON-FINAL OFFICE ACTION ENTERED AND FORWARDED TO EXAMINER |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: FINAL REJECTION MAILED |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: RESPONSE AFTER FINAL ACTION FORWARDED TO EXAMINER |
|
| STPP | Information on status: patent application and granting procedure in general |
Free format text: ADVISORY ACTION MAILED |
|
| STCB | Information on status: application discontinuation |
Free format text: ABANDONED -- FAILURE TO RESPOND TO AN OFFICE ACTION |