US20080255141A1 - 2-(3-aminoaryl) amino-4-aryl-thiazoles and their use as c-kit inhibitors - Google Patents
2-(3-aminoaryl) amino-4-aryl-thiazoles and their use as c-kit inhibitors Download PDFInfo
- Publication number
- US20080255141A1 US20080255141A1 US11/779,633 US77963307A US2008255141A1 US 20080255141 A1 US20080255141 A1 US 20080255141A1 US 77963307 A US77963307 A US 77963307A US 2008255141 A1 US2008255141 A1 US 2008255141A1
- Authority
- US
- United States
- Prior art keywords
- methyl
- phenyl
- thiazol
- ylamino
- pyridin
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Abandoned
Links
- 0 [1*]NC1=C([2*])C([3*])=C([4*])C(NC2=NC([6*])=C([7*])S2)=C1[5*] Chemical compound [1*]NC1=C([2*])C([3*])=C([4*])C(NC2=NC([6*])=C([7*])S2)=C1[5*] 0.000 description 29
- WJEOLQLKVOPQFV-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC=C(CN3CCN(C)CC3)C=C2)C=C1NC1=NC(C2=CN=CC=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC=C(CN3CCN(C)CC3)C=C2)C=C1NC1=NC(C2=CN=CC=C2)=CS1 WJEOLQLKVOPQFV-UHFFFAOYSA-N 0.000 description 4
- SKKZYPTWKSZJFL-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC=C(F)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC=C(F)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 SKKZYPTWKSZJFL-UHFFFAOYSA-N 0.000 description 3
- WXKDKFRAROPZKE-UHFFFAOYSA-N CC1=CC=C(CCN(C)C)C=C1.CC1=CC=C(CN(C)CCN(C)C)C=C1.CC1=CC=C(CN2CCN(C)CC2)C=C1.CCCCN(C)C.CCN(C)C.CCN1CCN(C)CC1 Chemical compound CC1=CC=C(CCN(C)C)C=C1.CC1=CC=C(CN(C)CCN(C)C)C=C1.CC1=CC=C(CN2CCN(C)CC2)C=C1.CCCCN(C)C.CCN(C)C.CCN1CCN(C)CC1 WXKDKFRAROPZKE-UHFFFAOYSA-N 0.000 description 2
- PPFQKCQGZBORSW-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=C(I)C=CC=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=C(I)C=CC=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 PPFQKCQGZBORSW-UHFFFAOYSA-N 0.000 description 2
- GMZJFDCZOMZJPJ-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC(F)=CC=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC(F)=CC=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 GMZJFDCZOMZJPJ-UHFFFAOYSA-N 0.000 description 2
- UDPCWSWVTRQVGM-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC=C(C#N)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC=C(C#N)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 UDPCWSWVTRQVGM-UHFFFAOYSA-N 0.000 description 2
- XCZQXHMVCNDPLG-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC=C(C3=CC=NC=C3)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC=C(C3=CC=NC=C3)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 XCZQXHMVCNDPLG-UHFFFAOYSA-N 0.000 description 2
- MXBYDCGTUCCYJR-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC=C(CN3CCN(C)CC3)C(F)=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC=C(CN3CCN(C)CC3)C(F)=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 MXBYDCGTUCCYJR-UHFFFAOYSA-N 0.000 description 2
- FLMMVGPIWPSVBQ-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC=C(N)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC=C(N)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 FLMMVGPIWPSVBQ-UHFFFAOYSA-N 0.000 description 2
- HPUOTTOGUYFWDO-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC=C(NC(=O)NC3=CC=CC=C3I)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC=C(NC(=O)NC3=CC=CC=C3I)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 HPUOTTOGUYFWDO-UHFFFAOYSA-N 0.000 description 2
- QRVUANQWDFOXLZ-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC=CC(C(F)(F)F)=C2F)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC=CC(C(F)(F)F)=C2F)C=C1NC1=NC(C2=CC=CN=C2)=CS1 QRVUANQWDFOXLZ-UHFFFAOYSA-N 0.000 description 2
- SCEVYEARSJSGFG-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2CCCCC2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2CCCCC2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 SCEVYEARSJSGFG-UHFFFAOYSA-N 0.000 description 2
- SCBIHBPBPQSHSH-UHFFFAOYSA-N CC1=CC=C(NC(=O)NC2=C(I)C=CC=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)NC2=C(I)C=CC=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 SCBIHBPBPQSHSH-UHFFFAOYSA-N 0.000 description 2
- VAKIMNBHVOIRBC-UHFFFAOYSA-N CC1=CC=C(NC(=O)NC2=CC=C(F)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)NC2=CC=C(F)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 VAKIMNBHVOIRBC-UHFFFAOYSA-N 0.000 description 2
- HLVZNOCHLJEKPQ-UHFFFAOYSA-N CC1=CC=C(NC(=O)OC(C)(C)C)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)OC(C)(C)C)C=C1NC1=NC(C2=CC=CN=C2)=CS1 HLVZNOCHLJEKPQ-UHFFFAOYSA-N 0.000 description 2
- OOAJRPGSJCCFNW-UHFFFAOYSA-N COC1=CC=C(NC(=O)NC2=CC=C(C)C(NC3=NC(C4=CC=CN=C4)=CS3)=C2)C=C1 Chemical compound COC1=CC=C(NC(=O)NC2=CC=C(C)C(NC3=NC(C4=CC=CN=C4)=CS3)=C2)C=C1 OOAJRPGSJCCFNW-UHFFFAOYSA-N 0.000 description 2
- JSZNVYZXJOMDSY-UHFFFAOYSA-N COC1=CC=CC(C2=CSC(NC3=CC(NC(=O)C4=CC=C(CN5CCN(C)CC5)C=C4)=CC=C3C)=N2)=C1 Chemical compound COC1=CC=CC(C2=CSC(NC3=CC(NC(=O)C4=CC=C(CN5CCN(C)CC5)C=C4)=CC=C3C)=N2)=C1 JSZNVYZXJOMDSY-UHFFFAOYSA-N 0.000 description 2
- IQMGXSROJBYCLS-UHFFFAOYSA-N Br.O=C(CBr)C1=CC=CN=C1 Chemical compound Br.O=C(CBr)C1=CC=CN=C1 IQMGXSROJBYCLS-UHFFFAOYSA-N 0.000 description 1
- QMCCCOUTBLKSRD-UHFFFAOYSA-N C#CC(=O)NC1=CC=C(C)C(NC2=NC(C3=CC=CN=C3)=CS2)=C1.CC1=CC=CC=C1 Chemical compound C#CC(=O)NC1=CC=C(C)C(NC2=NC(C3=CC=CN=C3)=CS2)=C1.CC1=CC=CC=C1 QMCCCOUTBLKSRD-UHFFFAOYSA-N 0.000 description 1
- HOVDUPXBRBXFBP-UHFFFAOYSA-N CC1=C(N)C=C(NC(=O)OC(C)(C)C)C=C1 Chemical compound CC1=C(N)C=C(NC(=O)OC(C)(C)C)C=C1 HOVDUPXBRBXFBP-UHFFFAOYSA-N 0.000 description 1
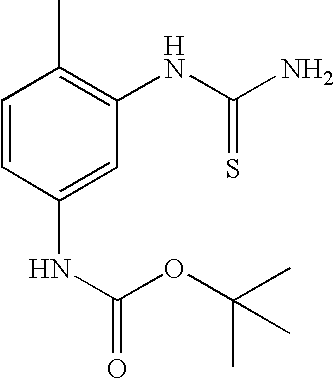
- BMJDMLUSJAPEKA-UHFFFAOYSA-N CC1=C(NC(N)=S)C=C(NC(=O)OC(C)(C)C)C=C1 Chemical compound CC1=C(NC(N)=S)C=C(NC(=O)OC(C)(C)C)C=C1 BMJDMLUSJAPEKA-UHFFFAOYSA-N 0.000 description 1
- BXSXHHGNJLSHQZ-UHFFFAOYSA-N CC1=CC(C2=CSC(NC3=CC(NC(=O)C4=CC=C(CN5CCN(C)CC5)C=C4)=CC=C3C)=N2)=C(C)C=C1 Chemical compound CC1=CC(C2=CSC(NC3=CC(NC(=O)C4=CC=C(CN5CCN(C)CC5)C=C4)=CC=C3C)=N2)=C(C)C=C1 BXSXHHGNJLSHQZ-UHFFFAOYSA-N 0.000 description 1
- MBDMUIBXKUVRIU-UHFFFAOYSA-N CC1=CC=C(F)C(C(=O)NC2=CC=C(C)C(NC3=NC(C4=CC=CN=C4)=CS3)=C2)=C1 Chemical compound CC1=CC=C(F)C(C(=O)NC2=CC=C(C)C(NC3=NC(C4=CC=CN=C4)=CS3)=C2)=C1 MBDMUIBXKUVRIU-UHFFFAOYSA-N 0.000 description 1
- LPQVLIARVYYSMD-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2(C)CCCCC2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2(C)CCCCC2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 LPQVLIARVYYSMD-UHFFFAOYSA-N 0.000 description 1
- CBORQCOAGKATDT-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=C(F)C(F)=C(CN3CCN(C)CC3)C(F)=C2F)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=C(F)C(F)=C(CN3CCN(C)CC3)C(F)=C2F)C=C1NC1=NC(C2=CC=CN=C2)=CS1 CBORQCOAGKATDT-UHFFFAOYSA-N 0.000 description 1
- VFHKPFDSVRRPNP-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC(Br)=C(CN3CCCCC3)C(Br)=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC(Br)=C(CN3CCCCC3)C(Br)=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 VFHKPFDSVRRPNP-UHFFFAOYSA-N 0.000 description 1
- SRHVBGWFEZUGDK-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC(Br)=C(CN3CCN(C)CC3)C(Br)=C2)C=C1NC1=NC(C2=CN=CC=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC(Br)=C(CN3CCN(C)CC3)C(Br)=C2)C=C1NC1=NC(C2=CN=CC=C2)=CS1 SRHVBGWFEZUGDK-UHFFFAOYSA-N 0.000 description 1
- HXPBQWLMFNCAMP-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC(Br)=C(CNCCCN3CCOCC3)C(Br)=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC(Br)=C(CNCCCN3CCOCC3)C(Br)=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 HXPBQWLMFNCAMP-UHFFFAOYSA-N 0.000 description 1
- YMTPDXSAOVXXPZ-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC(Br)=CC=C2)C=C1NC1=[SH]C(C)=C(C2=CC=C(Cl)C=C2)N1 Chemical compound CC1=CC=C(NC(=O)C2=CC(Br)=CC=C2)C=C1NC1=[SH]C(C)=C(C2=CC=C(Cl)C=C2)N1 YMTPDXSAOVXXPZ-UHFFFAOYSA-N 0.000 description 1
- MMQACRDUBAQOKX-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC(C(F)(F)F)=C(CN3CCN(C)CC3)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC(C(F)(F)F)=C(CN3CCN(C)CC3)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 MMQACRDUBAQOKX-UHFFFAOYSA-N 0.000 description 1
- MNFCKLABHYPVRO-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC(C(F)(F)F)=CC=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC(C(F)(F)F)=CC=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 MNFCKLABHYPVRO-UHFFFAOYSA-N 0.000 description 1
- YZDMBZHHYMNACR-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC(C3=CC=CC=C3)=CC=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC(C3=CC=CC=C3)=CC=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 YZDMBZHHYMNACR-UHFFFAOYSA-N 0.000 description 1
- SAFFSYZTDHGWFA-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC(Cl)=NC(Cl)=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC(Cl)=NC(Cl)=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 SAFFSYZTDHGWFA-UHFFFAOYSA-N 0.000 description 1
- SDQGEBMTALJSBZ-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC(N(C)C)=CC=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC(N(C)C)=CC=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 SDQGEBMTALJSBZ-UHFFFAOYSA-N 0.000 description 1
- VPUCPFOMKBFXJE-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC(N3CCN(C)CC3)=CC=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC(N3CCN(C)CC3)=CC=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 VPUCPFOMKBFXJE-UHFFFAOYSA-N 0.000 description 1
- QMNDIYFMJWMOPR-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC(N3CCOCC3)=CC=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC(N3CCOCC3)=CC=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 QMNDIYFMJWMOPR-UHFFFAOYSA-N 0.000 description 1
- MQQHLANQICCXGE-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC(OCCN3CCOCC3)=CC=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC(OCCN3CCOCC3)=CC=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 MQQHLANQICCXGE-UHFFFAOYSA-N 0.000 description 1
- SGGGYYFKGQLBFH-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC3=C(C=C2)OCO3)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC3=C(C=C2)OCO3)C=C1NC1=NC(C2=CC=CN=C2)=CS1 SGGGYYFKGQLBFH-UHFFFAOYSA-N 0.000 description 1
- RRJOLTDSXUHJOI-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC=C(C(C)(C)C)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC=C(C(C)(C)C)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 RRJOLTDSXUHJOI-UHFFFAOYSA-N 0.000 description 1
- ANWXGEHFDFQSAW-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC=C(C(C)N3CCN(C)CC3)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC=C(C(C)N3CCN(C)CC3)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 ANWXGEHFDFQSAW-UHFFFAOYSA-N 0.000 description 1
- JJTJOCVOUHFZFY-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC=C(CN(C(C)C)C(C)C)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC=C(CN(C(C)C)C(C)C)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 JJTJOCVOUHFZFY-UHFFFAOYSA-N 0.000 description 1
- AONQAAHQGPUOMH-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC=C(CN3CCCC3)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC=C(CN3CCCC3)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 AONQAAHQGPUOMH-UHFFFAOYSA-N 0.000 description 1
- KJVAAKIAQQUAHR-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC=C(CN3CCCCC3)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC=C(CN3CCCCC3)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 KJVAAKIAQQUAHR-UHFFFAOYSA-N 0.000 description 1
- YHGMOUIATBLANW-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC=C(CN3CCN(C)CC3)C(Br)=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC=C(CN3CCN(C)CC3)C(Br)=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 YHGMOUIATBLANW-UHFFFAOYSA-N 0.000 description 1
- SBWXEKIDMSQTJT-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC=C(CN3CCN(C)CC3)C(Cl)=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC=C(CN3CCN(C)CC3)C(Cl)=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 SBWXEKIDMSQTJT-UHFFFAOYSA-N 0.000 description 1
- RIGYPINMXOVQDY-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC=C(CN3CCN(C)CC3)C(I)=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC=C(CN3CCN(C)CC3)C(I)=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 RIGYPINMXOVQDY-UHFFFAOYSA-N 0.000 description 1
- JJKOMBJWKRKQPH-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC=C(CN3CCN(C)CC3)C=C2)C=C1NC1=NC(C2=CC([N+](=O)[O-])=CC=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC=C(CN3CCN(C)CC3)C=C2)C=C1NC1=NC(C2=CC([N+](=O)[O-])=CC=C2)=CS1 JJKOMBJWKRKQPH-UHFFFAOYSA-N 0.000 description 1
- BEYXMCLESTZBST-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC=C(CN3CCN(C)CC3)C=C2)C=C1NC1=NC(C2=CC=C(Cl)C=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC=C(CN3CCN(C)CC3)C=C2)C=C1NC1=NC(C2=CC=C(Cl)C=C2)=CS1 BEYXMCLESTZBST-UHFFFAOYSA-N 0.000 description 1
- YGWVJCXHJDGJCQ-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC=C(CN3CCN(C)CC3)C=C2)C=C1NC1=NC(C2=CC=C(F)C=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC=C(CN3CCN(C)CC3)C=C2)C=C1NC1=NC(C2=CC=C(F)C=C2)=CS1 YGWVJCXHJDGJCQ-UHFFFAOYSA-N 0.000 description 1
- MZEWHRCJOHCWLP-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC=C(CN3CCN(C)CC3)C=C2)C=C1NC1=NC(C2=CC=CC(F)=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC=C(CN3CCN(C)CC3)C=C2)C=C1NC1=NC(C2=CC=CC(F)=C2)=CS1 MZEWHRCJOHCWLP-UHFFFAOYSA-N 0.000 description 1
- WALVOEAGQTUGQN-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC=C(CN3CCN(C)CC3)C=C2)C=C1NC1=NC(C2=CC=CC=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC=C(CN3CCN(C)CC3)C=C2)C=C1NC1=NC(C2=CC=CC=C2)=CS1 WALVOEAGQTUGQN-UHFFFAOYSA-N 0.000 description 1
- AKQVJIWVBVJXCV-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC=C(CN3CCN(C)CC3)C=C2)C=C1NC1=NC(C2=CC=CC=C2F)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC=C(CN3CCN(C)CC3)C=C2)C=C1NC1=NC(C2=CC=CC=C2F)=CS1 AKQVJIWVBVJXCV-UHFFFAOYSA-N 0.000 description 1
- DPMDFBZMFUUHEC-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC=C(CN3CCN(C)CC3)C=C2)C=C1NC1=NC(C2=CC=CC=N2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC=C(CN3CCN(C)CC3)C=C2)C=C1NC1=NC(C2=CC=CC=N2)=CS1 DPMDFBZMFUUHEC-UHFFFAOYSA-N 0.000 description 1
- WGZKHTJHINOTGC-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC=C(CN3CCN(C)CC3)C=C2)C=C1NC1=NC(C2=CC=NC=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC=C(CN3CCN(C)CC3)C=C2)C=C1NC1=NC(C2=CC=NC=C2)=CS1 WGZKHTJHINOTGC-UHFFFAOYSA-N 0.000 description 1
- DUJUPCOUCGRXOT-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC=C(CN3CCN(C)CC3)C=C2)C=C1NC1=NC(C2=NC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC=C(CN3CCN(C)CC3)C=C2)C=C1NC1=NC(C2=NC=CN=C2)=CS1 DUJUPCOUCGRXOT-UHFFFAOYSA-N 0.000 description 1
- ZMACZMJSTAKFRT-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC=C(CN3CCN(C)CC3)C=C2)C=C1NC1=NC(C2=NC=CS2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC=C(CN3CCN(C)CC3)C=C2)C=C1NC1=NC(C2=NC=CS2)=CS1 ZMACZMJSTAKFRT-UHFFFAOYSA-N 0.000 description 1
- CGBAVWJYAUGAPT-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC=C(CN3CCOCC3)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC=C(CN3CCOCC3)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 CGBAVWJYAUGAPT-UHFFFAOYSA-N 0.000 description 1
- WZTAMPUODNTKPD-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC=C(CNC(=O)NC3=CC=C(C(F)(F)F)C=C3)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC=C(CNC(=O)NC3=CC=C(C(F)(F)F)C=C3)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 WZTAMPUODNTKPD-UHFFFAOYSA-N 0.000 description 1
- OQSRVMCZQXQZDF-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC=C(CNC(=O)OC(C)(C)C)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC=C(CNC(=O)OC(C)(C)C)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 OQSRVMCZQXQZDF-UHFFFAOYSA-N 0.000 description 1
- BWEAXAZSOMDUOY-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC=C(CO)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC=C(CO)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 BWEAXAZSOMDUOY-UHFFFAOYSA-N 0.000 description 1
- MCERIWKEZIJGRU-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC=C(I)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC=C(I)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 MCERIWKEZIJGRU-UHFFFAOYSA-N 0.000 description 1
- IADKBUOKLRYUFQ-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC=C(N(C)C)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC=C(N(C)C)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 IADKBUOKLRYUFQ-UHFFFAOYSA-N 0.000 description 1
- NWBVQVOAXGWQRI-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC=C(N3CCN(C)CC3)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC=C(N3CCN(C)CC3)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 NWBVQVOAXGWQRI-UHFFFAOYSA-N 0.000 description 1
- XGGSESMWJLIUFE-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC=C(NC(=O)NC3=C(C)ON=C3C)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC=C(NC(=O)NC3=C(C)ON=C3C)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 XGGSESMWJLIUFE-UHFFFAOYSA-N 0.000 description 1
- KVFINDHDAJOQAX-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC=C(NC(=O)NC3=CC=C(Br)C=C3)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC=C(NC(=O)NC3=CC=C(Br)C=C3)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 KVFINDHDAJOQAX-UHFFFAOYSA-N 0.000 description 1
- RATFTZXNYNJHSB-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC=C(NC(=O)NC3=CC=C(C(F)(F)F)C=C3)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC=C(NC(=O)NC3=CC=C(C(F)(F)F)C=C3)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 RATFTZXNYNJHSB-UHFFFAOYSA-N 0.000 description 1
- WHZLXYJMUNOZLF-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC=C(NC(=O)NC3=CC=C(F)C=C3)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC=C(NC(=O)NC3=CC=C(F)C=C3)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 WHZLXYJMUNOZLF-UHFFFAOYSA-N 0.000 description 1
- TWIHOYUAFXRKOM-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC=C(NC(=O)NC3=CC=C(OC(F)(F)F)C=C3)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC=C(NC(=O)NC3=CC=C(OC(F)(F)F)C=C3)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 TWIHOYUAFXRKOM-UHFFFAOYSA-N 0.000 description 1
- KJGHAUAVPDADDA-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC=C(NC(=O)NC3=CC=CS3)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC=C(NC(=O)NC3=CC=CS3)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 KJGHAUAVPDADDA-UHFFFAOYSA-N 0.000 description 1
- HBQUUXYOEONPFJ-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC=C(NS(=O)(=O)C3=CC=CS3)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC=C(NS(=O)(=O)C3=CC=CS3)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 HBQUUXYOEONPFJ-UHFFFAOYSA-N 0.000 description 1
- JLSZOQLVXHONSY-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC=C(O)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC=C(O)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 JLSZOQLVXHONSY-UHFFFAOYSA-N 0.000 description 1
- XNKQVBBQSPTBCZ-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC=C(OC(C)C)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC=C(OC(C)C)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 XNKQVBBQSPTBCZ-UHFFFAOYSA-N 0.000 description 1
- QRIDQZNRZUDWNI-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC=C(OS(=O)(=O)C3=CC=C(I)C=C3)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC=C(OS(=O)(=O)C3=CC=C(I)C=C3)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 QRIDQZNRZUDWNI-UHFFFAOYSA-N 0.000 description 1
- YWZLWVCIFZRJPM-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC=C(OS(=O)(=O)C3=CC=CC(F)=C3)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC=C(OS(=O)(=O)C3=CC=CC(F)=C3)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 YWZLWVCIFZRJPM-UHFFFAOYSA-N 0.000 description 1
- UTHRZYFTNFGDRP-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC=C(OS(=O)(=O)C3=CC=CC=C3F)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC=C(OS(=O)(=O)C3=CC=CC=C3F)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 UTHRZYFTNFGDRP-UHFFFAOYSA-N 0.000 description 1
- RRSCKLGUDQTBIK-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC=C(OS(=O)(=O)C3=CC=CS3)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC=C(OS(=O)(=O)C3=CC=CS3)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 RRSCKLGUDQTBIK-UHFFFAOYSA-N 0.000 description 1
- KHFNEWHLCQZGSA-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC=CC(Br)=C2)C=C1NC1=NC(C(=O)NCCN(C)C)=C(C2=CC=C(Cl)C=C2)S1 Chemical compound CC1=CC=C(NC(=O)C2=CC=CC(Br)=C2)C=C1NC1=NC(C(=O)NCCN(C)C)=C(C2=CC=C(Cl)C=C2)S1 KHFNEWHLCQZGSA-UHFFFAOYSA-N 0.000 description 1
- HTRNILAWCRVURF-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC=CC(Br)=C2)C=C1NC1=NC(C2=CN=CC=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC=CC(Br)=C2)C=C1NC1=NC(C2=CN=CC=C2)=CS1 HTRNILAWCRVURF-UHFFFAOYSA-N 0.000 description 1
- APUQHEUVLUFGOT-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC=CC(I)=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC=CC(I)=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 APUQHEUVLUFGOT-UHFFFAOYSA-N 0.000 description 1
- ULYVULMXRBPHAW-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2=CC=NC=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2=CC=NC=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 ULYVULMXRBPHAW-UHFFFAOYSA-N 0.000 description 1
- CZKBLNIWCQYTSJ-UHFFFAOYSA-N CC1=CC=C(NC(=O)C2CCC(C(C)(C)C)CC2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)C2CCC(C(C)(C)C)CC2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 CZKBLNIWCQYTSJ-UHFFFAOYSA-N 0.000 description 1
- LPBRBJBRSSHHJB-UHFFFAOYSA-N CC1=CC=C(NC(=O)CCCN2CCOCC2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)CCCN2CCOCC2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 LPBRBJBRSSHHJB-UHFFFAOYSA-N 0.000 description 1
- NCQGHLSMCXYVRT-UHFFFAOYSA-N CC1=CC=C(NC(=O)N(C(=O)NC2=C(I)C=CC=C2)C2=CC=CC=C2I)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)N(C(=O)NC2=C(I)C=CC=C2)C2=CC=CC=C2I)C=C1NC1=NC(C2=CC=CN=C2)=CS1 NCQGHLSMCXYVRT-UHFFFAOYSA-N 0.000 description 1
- GXRJFTLDAJPVML-UHFFFAOYSA-N CC1=CC=C(NC(=O)N(C(=O)NC2CCCCC2)C2CCCCC2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)N(C(=O)NC2CCCCC2)C2CCCCC2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 GXRJFTLDAJPVML-UHFFFAOYSA-N 0.000 description 1
- QPLMNSIZECOHPY-UHFFFAOYSA-N CC1=CC=C(NC(=O)NC2=C(C)ON=C2C)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)NC2=C(C)ON=C2C)C=C1NC1=NC(C2=CC=CN=C2)=CS1 QPLMNSIZECOHPY-UHFFFAOYSA-N 0.000 description 1
- UDRPEFNRPWYJAX-UHFFFAOYSA-N CC1=CC=C(NC(=O)NC2=CC=C(Br)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)NC2=CC=C(Br)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 UDRPEFNRPWYJAX-UHFFFAOYSA-N 0.000 description 1
- SXDISTILPFYOAS-UHFFFAOYSA-N CC1=CC=C(NC(=O)NC2=CC=C(C(F)(F)F)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)NC2=CC=C(C(F)(F)F)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 SXDISTILPFYOAS-UHFFFAOYSA-N 0.000 description 1
- FBLFKZGXBDMGCC-UHFFFAOYSA-N CC1=CC=C(NC(=O)NC2=CC=C(C)C(NC3=NC(C4=CC=CN=C4)=CS3)=C2)C=C1 Chemical compound CC1=CC=C(NC(=O)NC2=CC=C(C)C(NC3=NC(C4=CC=CN=C4)=CS3)=C2)C=C1 FBLFKZGXBDMGCC-UHFFFAOYSA-N 0.000 description 1
- YKLFZDACGSCZEZ-UHFFFAOYSA-N CC1=CC=C(NC(=O)NC2=CC=C(N(C)C)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)NC2=CC=C(N(C)C)C=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 YKLFZDACGSCZEZ-UHFFFAOYSA-N 0.000 description 1
- XEYQUTMMFCBYIF-UHFFFAOYSA-N CC1=CC=C(NC(=O)NC2=CC=CC(F)=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)NC2=CC=CC(F)=C2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 XEYQUTMMFCBYIF-UHFFFAOYSA-N 0.000 description 1
- LCJWBJVZGABBNT-UHFFFAOYSA-N CC1=CC=C(NC(=O)NC2=CC=CC=C2Cl)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)NC2=CC=CC=C2Cl)C=C1NC1=NC(C2=CC=CN=C2)=CS1 LCJWBJVZGABBNT-UHFFFAOYSA-N 0.000 description 1
- SBJPKQHAMUMAMS-UHFFFAOYSA-N CC1=CC=C(NC(=O)NC2=CC=CC=C2F)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)NC2=CC=CC=C2F)C=C1NC1=NC(C2=CC=CN=C2)=CS1 SBJPKQHAMUMAMS-UHFFFAOYSA-N 0.000 description 1
- UZRCOVVKVNUSFE-UHFFFAOYSA-N CC1=CC=C(NC(=O)NC2=CC=CS2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)NC2=CC=CS2)C=C1NC1=NC(C2=CC=CN=C2)=CS1 UZRCOVVKVNUSFE-UHFFFAOYSA-N 0.000 description 1
- LVDHDJUQIYZLHU-UHFFFAOYSA-N CC1=CC=C(NC(=O)OCC(C)C)C=C1NC1=NC(C2=CC=C(Cl)C=C2)=C(C)S1 Chemical compound CC1=CC=C(NC(=O)OCC(C)C)C=C1NC1=NC(C2=CC=C(Cl)C=C2)=C(C)S1 LVDHDJUQIYZLHU-UHFFFAOYSA-N 0.000 description 1
- BFVQTFUEPYFFGF-UHFFFAOYSA-N CC1=CC=C(NC(=O)OCC(C)C)C=C1NC1=NC(C2=CC=CN=C2)=CS1 Chemical compound CC1=CC=C(NC(=O)OCC(C)C)C=C1NC1=NC(C2=CC=CN=C2)=CS1 BFVQTFUEPYFFGF-UHFFFAOYSA-N 0.000 description 1
- KCGBOXHPFHEYPO-UHFFFAOYSA-N CC1=CC=CC(C(=O)NC2=CC=C(C)C(NC3=NC(C4=CC=CN=C4)=CS3)=C2)=C1 Chemical compound CC1=CC=CC(C(=O)NC2=CC=C(C)C(NC3=NC(C4=CC=CN=C4)=CS3)=C2)=C1 KCGBOXHPFHEYPO-UHFFFAOYSA-N 0.000 description 1
- DBYDMPQYTIUVOM-UHFFFAOYSA-N CC1=CC=CC(C2=CSC(NC3=CC(NC(=O)C4=CC=C(CN5CCN(C)CC5)C=C4)=CC=C3C)=N2)=C1 Chemical compound CC1=CC=CC(C2=CSC(NC3=CC(NC(=O)C4=CC=C(CN5CCN(C)CC5)C=C4)=CC=C3C)=N2)=C1 DBYDMPQYTIUVOM-UHFFFAOYSA-N 0.000 description 1
- SVSQGEGDCXDOOC-UHFFFAOYSA-N CC1=CC=CN=C1.CC1=CC=NC=C1 Chemical compound CC1=CC=CN=C1.CC1=CC=NC=C1 SVSQGEGDCXDOOC-UHFFFAOYSA-N 0.000 description 1
- VNMDYAXPMVGCHS-UHFFFAOYSA-N CC1=CN=CC(C2=CSC(NC3=CC(NC(=O)C4=CC=C(CN5CCN(C)CC5)C=C4)=CC=C3C)=N2)=C1 Chemical compound CC1=CN=CC(C2=CSC(NC3=CC(NC(=O)C4=CC=C(CN5CCN(C)CC5)C=C4)=CC=C3C)=N2)=C1 VNMDYAXPMVGCHS-UHFFFAOYSA-N 0.000 description 1
- OUHLELYQKLFGOV-UHFFFAOYSA-N CCCN(CCC)CC1=CC=C(C(=O)NC2=CC=C(C)C(NC3=NC(C4=CC=CN=C4)=CS3)=C2)C=C1 Chemical compound CCCN(CCC)CC1=CC=C(C(=O)NC2=CC=C(C)C(NC3=NC(C4=CC=CN=C4)=CS3)=C2)C=C1 OUHLELYQKLFGOV-UHFFFAOYSA-N 0.000 description 1
- DCEXZPYFLTXAGD-UHFFFAOYSA-N CCN(CC)CC1=CC=C(C(=O)NC2=CC=C(C)C(NC3=NC(C4=CC=CN=C4)=CS3)=C2)C=C1 Chemical compound CCN(CC)CC1=CC=C(C(=O)NC2=CC=C(C)C(NC3=NC(C4=CC=CN=C4)=CS3)=C2)C=C1 DCEXZPYFLTXAGD-UHFFFAOYSA-N 0.000 description 1
- XICKQDNHUQPULN-UHFFFAOYSA-N CCOC(=O)C1=C(C2=CC=C(Cl)C=C2)SC(NC2=C(C)C=CC(NC(=O)C3=CC(Br)=CC=C3)=C2)=N1 Chemical compound CCOC(=O)C1=C(C2=CC=C(Cl)C=C2)SC(NC2=C(C)C=CC(NC(=O)C3=CC(Br)=CC=C3)=C2)=N1 XICKQDNHUQPULN-UHFFFAOYSA-N 0.000 description 1
- ABVULGCWBWSKRG-UHFFFAOYSA-N CCOC(=O)C1=CC=C(NC(=O)NC2=CC=C(C(=O)NC3=CC=C(C)C(NC4=NC(C5=CC=CN=C5)=CS4)=C3)C=C2)C=C1 Chemical compound CCOC(=O)C1=CC=C(NC(=O)NC2=CC=C(C(=O)NC3=CC=C(C)C(NC4=NC(C5=CC=CN=C5)=CS4)=C3)C=C2)C=C1 ABVULGCWBWSKRG-UHFFFAOYSA-N 0.000 description 1
- FDIVAWZPIRPGLP-UHFFFAOYSA-N CCOC(=O)C1=CC=C(NC(=O)NC2=CC=C(C)C(NC3=NC(C4=CC=CN=C4)=CS3)=C2)C=C1 Chemical compound CCOC(=O)C1=CC=C(NC(=O)NC2=CC=C(C)C(NC3=NC(C4=CC=CN=C4)=CS3)=C2)C=C1 FDIVAWZPIRPGLP-UHFFFAOYSA-N 0.000 description 1
- RVRMMYRDJOVSJP-UHFFFAOYSA-N CCOC(=O)C1=CSC(NC2=C(C)C=CC(NC(=O)C3=CC=C(CN4CCN(C)CC4)C=C3)=C2)=N1 Chemical compound CCOC(=O)C1=CSC(NC2=C(C)C=CC(NC(=O)C3=CC=C(CN4CCN(C)CC4)C=C3)=C2)=N1 RVRMMYRDJOVSJP-UHFFFAOYSA-N 0.000 description 1
- NSAUAEORYOTRRQ-UHFFFAOYSA-N CCOC(=O)C1=CSC(NC2=C(C)C=CC(NC(=O)C3=CC=CC(I)=C3)=C2)=N1 Chemical compound CCOC(=O)C1=CSC(NC2=C(C)C=CC(NC(=O)C3=CC=CC(I)=C3)=C2)=N1 NSAUAEORYOTRRQ-UHFFFAOYSA-N 0.000 description 1
- NTZRKVXHQNZFKC-UHFFFAOYSA-N CN1CCN(CC2=CC=C(C(=O)NC3=CC=C(Cl)C(NC4=NC(C5=CC=CN=C5)=CS4)=C3)C=C2)CC1 Chemical compound CN1CCN(CC2=CC=C(C(=O)NC3=CC=C(Cl)C(NC4=NC(C5=CC=CN=C5)=CS4)=C3)C=C2)CC1 NTZRKVXHQNZFKC-UHFFFAOYSA-N 0.000 description 1
- YMTDWEYABFIRDQ-UHFFFAOYSA-N CN1CCN(CC2=CC=C(C(=O)NC3=CC=CC(NC4=NC(C5=CN=CC=C5)=CS4)=C3)C=C2)CC1 Chemical compound CN1CCN(CC2=CC=C(C(=O)NC3=CC=CC(NC4=NC(C5=CN=CC=C5)=CS4)=C3)C=C2)CC1 YMTDWEYABFIRDQ-UHFFFAOYSA-N 0.000 description 1
- MEYCYFIWDAQKQK-UHFFFAOYSA-N COC(=O)C1=CC=C(CN2CCN(C)CC2)C=C1 Chemical compound COC(=O)C1=CC=C(CN2CCN(C)CC2)C=C1 MEYCYFIWDAQKQK-UHFFFAOYSA-N 0.000 description 1
- BPDUQAIZDDBUFF-UHFFFAOYSA-N COC(=O)C1=CC=C(CNC2=CC=C(C)C(NC3=NC(C4=CC=CN=C4)=CS3)=C2)C=C1 Chemical compound COC(=O)C1=CC=C(CNC2=CC=C(C)C(NC3=NC(C4=CC=CN=C4)=CS3)=C2)C=C1 BPDUQAIZDDBUFF-UHFFFAOYSA-N 0.000 description 1
- BEHMNXSLMVYWSS-UHFFFAOYSA-N COC(C)C1=CC=C(C(=O)NC2=CC=C(C)C(NC3=NC(C4=CC=CN=C4)=CS3)=C2)C=C1 Chemical compound COC(C)C1=CC=C(C(=O)NC2=CC=C(C)C(NC3=NC(C4=CC=CN=C4)=CS3)=C2)C=C1 BEHMNXSLMVYWSS-UHFFFAOYSA-N 0.000 description 1
- NOTQASNRSMEJPJ-UHFFFAOYSA-N COC1=CC(NC(=O)NC2=CC=C(C)C(NC3=NC(C4=CC=CN=C4)=CS3)=C2)=CC(OC)=C1OC Chemical compound COC1=CC(NC(=O)NC2=CC=C(C)C(NC3=NC(C4=CC=CN=C4)=CS3)=C2)=CC(OC)=C1OC NOTQASNRSMEJPJ-UHFFFAOYSA-N 0.000 description 1
- DAKNZRAHYDFGKR-UHFFFAOYSA-N COC1=CC(OC)=C(NC(=O)NC2=CC=C(C)C(NC3=NC(C4=CC=CN=C4)=CS3)=C2)C=C1 Chemical compound COC1=CC(OC)=C(NC(=O)NC2=CC=C(C)C(NC3=NC(C4=CC=CN=C4)=CS3)=C2)C=C1 DAKNZRAHYDFGKR-UHFFFAOYSA-N 0.000 description 1
- DYHOZMNSKZNIBC-UHFFFAOYSA-N COC1=CC=C(C2=CSC(NC3=CC(NC(=O)C4=CC=C(CN5CCN(C)CC5)C=C4)=CC=C3C)=N2)C=C1 Chemical compound COC1=CC=C(C2=CSC(NC3=CC(NC(=O)C4=CC=C(CN5CCN(C)CC5)C=C4)=CC=C3C)=N2)C=C1 DYHOZMNSKZNIBC-UHFFFAOYSA-N 0.000 description 1
- LFHZGTMLDVLPKD-UHFFFAOYSA-N COC1=CC=C(NC(=O)NC2=CC=C(C(=O)NC3=CC=C(C)C(NC4=NC(C5=CC=CN=C5)=CS4)=C3)C=C2)C(OC)=C1 Chemical compound COC1=CC=C(NC(=O)NC2=CC=C(C(=O)NC3=CC=C(C)C(NC4=NC(C5=CC=CN=C5)=CS4)=C3)C=C2)C(OC)=C1 LFHZGTMLDVLPKD-UHFFFAOYSA-N 0.000 description 1
- ALSGXGJVKXBFAV-UHFFFAOYSA-N COC1=CC=C(NC(=O)NC2=CC=C(C(=O)NC3=CC=C(C)C(NC4=NC(C5=CC=CN=C5)=CS4)=C3)C=C2)C=C1 Chemical compound COC1=CC=C(NC(=O)NC2=CC=C(C(=O)NC3=CC=C(C)C(NC4=NC(C5=CC=CN=C5)=CS4)=C3)C=C2)C=C1 ALSGXGJVKXBFAV-UHFFFAOYSA-N 0.000 description 1
- OMKGWHVIYKGSHL-UHFFFAOYSA-N COC1=CC=CC(C(=O)NC2=CC=C(C)C(NC3=NC(C4=CC=CN=C4)=CS3)=C2)=C1 Chemical compound COC1=CC=CC(C(=O)NC2=CC=C(C)C(NC3=NC(C4=CC=CN=C4)=CS3)=C2)=C1 OMKGWHVIYKGSHL-UHFFFAOYSA-N 0.000 description 1
- VFENKHXXKSIVTR-UHFFFAOYSA-N [C-]#[N+]C1=CC=C(C2=CSC(NC3=CC(NC(=O)C4=CC=C(CN5CCN(C)CC5)C=C4)=CC=C3C)=N2)C=C1 Chemical compound [C-]#[N+]C1=CC=C(C2=CSC(NC3=CC(NC(=O)C4=CC=C(CN5CCN(C)CC5)C=C4)=CC=C3C)=N2)C=C1 VFENKHXXKSIVTR-UHFFFAOYSA-N 0.000 description 1
- HORJEKSGJRCUKV-UHFFFAOYSA-N [H]C(F)(F)OC1=CC=C(NC(=O)NC2=CC=C(C)C(NC3=NC(C4=CC=CN=C4)=CS3)=C2)C=C1 Chemical compound [H]C(F)(F)OC1=CC=C(NC(=O)NC2=CC=C(C)C(NC3=NC(C4=CC=CN=C4)=CS3)=C2)C=C1 HORJEKSGJRCUKV-UHFFFAOYSA-N 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D417/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00
- C07D417/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing two hetero rings
- C07D417/04—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing two hetero rings directly linked by a ring-member-to-ring-member bond
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/04—Drugs for disorders of the alimentary tract or the digestive system for ulcers, gastritis or reflux esophagitis, e.g. antacids, inhibitors of acid secretion, mucosal protectants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
- A61P11/02—Nasal agents, e.g. decongestants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
- A61P11/06—Antiasthmatics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P13/00—Drugs for disorders of the urinary system
- A61P13/12—Drugs for disorders of the urinary system of the kidneys
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P15/00—Drugs for genital or sexual disorders; Contraceptives
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/04—Antipruritics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/06—Antipsoriatics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/02—Drugs for skeletal disorders for joint disorders, e.g. arthritis, arthrosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/08—Drugs for skeletal disorders for bone diseases, e.g. rachitism, Paget's disease
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P21/00—Drugs for disorders of the muscular or neuromuscular system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/06—Antimigraine agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/08—Antiepileptics; Anticonvulsants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/14—Drugs for disorders of the nervous system for treating abnormal movements, e.g. chorea, dyskinesia
- A61P25/16—Anti-Parkinson drugs
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/18—Antipsychotics, i.e. neuroleptics; Drugs for mania or schizophrenia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/22—Anxiolytics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/24—Antidepressants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/28—Drugs for disorders of the nervous system for treating neurodegenerative disorders of the central nervous system, e.g. nootropic agents, cognition enhancers, drugs for treating Alzheimer's disease or other forms of dementia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/30—Drugs for disorders of the nervous system for treating abuse or dependence
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P27/00—Drugs for disorders of the senses
- A61P27/02—Ophthalmic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/04—Anorexiants; Antiobesity agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/06—Antihyperlipidemics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/08—Drugs for disorders of the metabolism for glucose homeostasis
- A61P3/10—Drugs for disorders of the metabolism for glucose homeostasis for hyperglycaemia, e.g. antidiabetics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/04—Antibacterial agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/12—Antivirals
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P33/00—Antiparasitic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
- A61P35/02—Antineoplastic agents specific for leukemia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/02—Immunomodulators
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/02—Immunomodulators
- A61P37/06—Immunosuppressants, e.g. drugs for graft rejection
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/08—Antiallergic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P41/00—Drugs used in surgical methods, e.g. surgery adjuvants for preventing adhesion or for vitreum substitution
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P7/00—Drugs for disorders of the blood or the extracellular fluid
- A61P7/04—Antihaemorrhagics; Procoagulants; Haemostatic agents; Antifibrinolytic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/06—Antiarrhythmics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/10—Drugs for disorders of the cardiovascular system for treating ischaemic or atherosclerotic diseases, e.g. antianginal drugs, coronary vasodilators, drugs for myocardial infarction, retinopathy, cerebrovascula insufficiency, renal arteriosclerosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/12—Antihypertensives
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D277/00—Heterocyclic compounds containing 1,3-thiazole or hydrogenated 1,3-thiazole rings
- C07D277/02—Heterocyclic compounds containing 1,3-thiazole or hydrogenated 1,3-thiazole rings not condensed with other rings
- C07D277/20—Heterocyclic compounds containing 1,3-thiazole or hydrogenated 1,3-thiazole rings not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members
- C07D277/32—Heterocyclic compounds containing 1,3-thiazole or hydrogenated 1,3-thiazole rings not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D277/38—Nitrogen atoms
- C07D277/42—Amino or imino radicals substituted by hydrocarbon or substituted hydrocarbon radicals
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D277/00—Heterocyclic compounds containing 1,3-thiazole or hydrogenated 1,3-thiazole rings
- C07D277/02—Heterocyclic compounds containing 1,3-thiazole or hydrogenated 1,3-thiazole rings not condensed with other rings
- C07D277/20—Heterocyclic compounds containing 1,3-thiazole or hydrogenated 1,3-thiazole rings not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members
- C07D277/32—Heterocyclic compounds containing 1,3-thiazole or hydrogenated 1,3-thiazole rings not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D277/56—Carbon atoms having three bonds to hetero atoms with at the most one bond to halogen
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D417/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00
- C07D417/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing three or more hetero rings
Definitions
- the present invention relates to novel compounds selected from 2-(3-aminoaryl)amino-4-aryl-thiazoles that selectively modulate, regulate, and/or inhibit signal transduction mediated by certain native and/or mutant tyrosine kinases implicated in a variety of human and animal diseases such as cell proliferative, metabolic, allergic, and degenerative disorders. More particularly, these compounds are potent and selective c-kit inhibitors.
- Tyrosine kinases are receptor type or non-receptor type proteins, which transfer the terminal phosphate of ATP to tyrosine residues of proteins thereby activating or inactivating signal transduction pathways. These proteins are known to be involved in many cellular mechanisms, which in case of disruption, lead to disorders such as abnormal cell proliferation and migration as well as inflammation.
- tyrosine kinases As of today, there are about 58 known receptor tyrosine kinases. Other tyrosine kinases are the well-known VEGF receptors (Kim et al., Nature 362, pp. 841-844, 1993), PDGF receptors, c-kit and the FLK family. These receptors can transmit signals to other tyrosine kinases including Src, Raf, Frk, Btk, Csk, Abl, Fes/Fps, Fak, Jak, Ack. etc.
- c-kit is of special interest. Indeed, c-kit is a key receptor activating mast cells, which have proved to be directly or indirectly implicated in numerous pathologies for which the Applicant filed WO 03/004007, WO 03/004006, WO 03/003006, WO 03/003004, WO 03/002114, WO 03/002109, WO 03/002108, WO 03/002107, WO 03/002106, WO 03/002105, WO 03/039550, WO 03/035050, WO 03/035049, U.S. 60/359,652 and U.S. 60/359,651.
- mast cells present in tissues of patients are implicated in or contribute to the genesis of diseases such as autoimmune diseases (rheumatoid arthritis, inflammatory bowel diseases (IBD)) allergic diseases, tumor angiogenesis, inflammatory diseases, and interstitial cystitis.
- autoimmune diseases rheumatoid arthritis, inflammatory bowel diseases (IBD)
- IBD inflammatory bowel diseases
- mast cells participate in the destruction of tissues by releasing a cocktail of different proteases and mediators such as histamine, neutral proteases, lipid-derived mediators (prostaglandins, thromboxanes and leucotrienes), and various cytokines (IL-1, IL-2, IL-3, IL-4, IL-5, IL-6, IL-8, TNF- ⁇ , GM-CSF, MIP-1a, MIP-1b, MIP-2 and IFN- ⁇ ).
- proteases and mediators such as histamine, neutral proteases, lipid-derived mediators (prostaglandins, thromboxanes and leucotrienes), and various cytokines (IL-1, IL-2, IL-3, IL-4, IL-5, IL-6, IL-8, TNF- ⁇ , GM-CSF, MIP-1a, MIP-1b, MIP-2 and IFN- ⁇ ).
- the c-kit receptor also can be constitutively activated by mutations leading to abnormal cell proliferation and development of diseases such as mastocytosis and various cancers.
- the main objective underlying the present invention is therefore to find potent and selective compounds capable of inhibiting wild type and/or mutated c-kit.
- tyrosine kinase inhibitors for example, bis monocyclic, bicyclic or heterocyclic aryl compounds (WO 92/20642), vinylene-azaindole derivatives (WO 94/14808) and 1-cycloproppyl-4-pyridyl-quinolones (U.S. Pat. No. 5,330,992), styryl compounds (U.S. Pat. No. 5,217,999), styryl-substituted pyridyl compounds (U.S. Pat. No.
- the present invention relates to compounds belonging to the 2-(3-amino)arylamino-4-aryl-thiazoles. These compounds are capable of selectively inhibiting signal transduction involving the tyrosine phosphokinase c-kit and mutant forms thereof.
- the invention is aimed at compounds of formula I, which may represent either free base forms of the substances or pharmaceutically acceptable salts thereof:
- R 1 is: a) a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality; b) an aryl or heteroaryl group optionally substituted by an alkyl or aryl group optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; c) a —CO—NH—R, —CO—R, —CO—OR or a —CO—NRR′ group, wherein R and R′ are independently chosen from H or an aryl, heteroaryl, alkyl and cycloalkyl group optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality; R 2 is hydrogen, halogen or a linear or branched alkyl
- H a halogen selected from I, F, Cl or Br
- NH2, NO2 or SO2-R wherein R is a linear or branched alkyl group containing one or more group such as 1 to 10 carbon atoms, and optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality;
- R is H or an organic group that can be selected for example from a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted by an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F and/or bearing a pendant basic nitrogen functionality.
- R is H or an organic group that can be selected for example from a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F and/or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group optionally substituted with a cycloalkyl, an aryl or heteroaryl group optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F and/or bearing a pendant basic nitrogen functionality; a —SO2-R group wherein R is an alkyl, cycloalkyl, aryl or heteroaryl optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F and/or bearing a pendant basic nitrogen functionality
- R is H or an organic group that can be selected for example from a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; or an alkyl, cycloalkyl, aryl or heteroaryl group substituted by a alkyl, cycloalkyl, aryl or heteroaryl group optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; a —SO2-R group wherein R is an alkyl, cycloalkyl, aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected
- R is H or an organic group that can be selected for example from a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, aryl or heteroaryl group optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F and/or bearing a pendant basic nitrogen functionality; or an alkyl, cycloalkyl, aryl or heteroaryl group substituted by a alkyl, cycloalkyl, aryl or heteroaryl group optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F and/or bearing a pendant basic nitrogen functionality; a —SO2-R group wherein R is an alkyl, cycloalkyl, aryl or heteroaryl group optionally substituted with an heteroatom, notably
- R is H or an organic group that can be selected for example from a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom (for example a halogen) and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group substituted by an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality.
- N-Aminoalkyl-N′-thiazol-2-yl-benzene-1,3-diamine compounds of the following formula:
- Y is a linear or branched alkyl group containing from 1 to 10 carbon atoms; wherein Z represents an aryl or heteroaryl group, optionally substituted at one or more ring position with any permutation of the following groups:
- X is R or NRR′ and wherein R and R′ are independently chosen from H, an aryl, a heteroaryl, an alkyl, or a cycloalkyl group optionally substituted with at least one heteroatom, such as for example a halogen chosen from F, I, Cl and Br and optionally bearing a pendant basic nitrogen functionality; or an aryl, a heteroaryl, an alkyl or a cycloalkyl group substituted with an aryl, a heteroaryl, an alkyl or a cycloalkyl group optionally substituted with at least one heteroatom, such as for example a halogen chosen from F, I, Cl and Br and optionally bearing a pendant basic nitrogen functionality, R 2 is hydrogen, halogen or a linear or branched alkyl group containing from 1 to 10 carbon atoms, trifluoromethyl or alkoxy; R 3 is hydrogen, halogen or a linear or branched alkyl group containing from 1 to 10 carbon atoms,
- H a halogen selected from I, F, Cl or Br
- NH2, NO2 or SO2-R wherein R is a linear or branched alkyl group containing one or more group such as 1 to 10 carbon atoms, and optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality.
- substituent R6 which in the formula II is connected to position 4 of the thiazole ring, may instead occupy position 5 of the thiazole ring.
- the invention is directed to compounds in which X is a substituted alkyl, aryl or heteroaryl group bearing a pendant basic nitrogen functionality represented for example by the structures a to f shown below, wherein the wavy line corresponds to the point of attachment to core structure of formula II:
- X (see formula II) is preferentially group d.
- the invention concerns the compounds in which R 2 and R 3 are hydrogen.
- R 4 is a methyl group and R 5 is H.
- R 6 is preferentially a 3—pyridyl group (cf. structure g below), or a 4-pyridyl group (cf. structure h below).
- the wavy line in structure g and h correspond to the point of attachment to the core structure of formula I or II.
- X is R or NRR′ and wherein R and R′ are independently chosen from H or an organic group that can be selected for example from a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group optionally substituted with a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; a —SO2-R group wherein R is an alkyl, cycloalkyl, aryl or heteroaryl optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Cl
- R 4 is hydrogen, halogen or a linear or branched alkyl group containing from 1 to 10 carbon atoms, trifluoromethyl or alkoxy;
- R 6 is one of the following: (i) an aryl group such as phenyl or a substituted variant thereof bearing any combination, at any one ring position, of one or more substituents such as halogen, alkyl groups containing from 1 to 10 carbon atoms, trifluoromethyl, and alkoxy; (ii) a heteroaryl group such as a 2, 3, or 4-pyridyl group, which may additionally bear any combination of one or more substituents such as halogen, alkyl groups containing from 1 to 10 carbon atoms, trifluoromethyl and alkoxy; (iii) a five-membered ring aromatic heterocyclic group such as for example 2-thienyl, 3-thienyl, 2-thiazolyl, 4-thiazolyl, 5-thiazolyl, which may additionally bear any combination of one or more substituents such as halogen, an alkyl group containing from 1 to 10 carbon atoms, trifluoromethyl, and alkoxy.
- substituents such as
- H a halogen selected from I, F, Cl or Br
- NH2, NO2 or SO2-R wherein R is a linear or branched alkyl group containing one or more group such as 1 to 10 carbon atoms, and optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality.
- substituent R6 which in the formula II is connected to position 4 of the thiazole ring, may instead occupy position 5 of the thiazole ring.
- the invention is particularly embodied by the compounds wherein X is a urea group, a —CO—NRR′ group, corresponding to the [3-(thiazol-2-ylamino)-phenyl]-urea family and the following formula II-2
- Ra, Rb are independently chosen from H or an organic group that can be selected for example from a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group optionally substituted with a cycloalkyl, an aryl or heteroaryl group optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; a —SO2-R group wherein R is an alkyl, cycloalkyl, aryl or heteroaryl optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic
- R 4 is hydrogen, halogen or a linear or branched alkyl group containing from 1 to 10 carbon atoms, trifluoromethyl or alkoxy
- R 6 is one of the following: (i) an aryl group such as phenyl or a substituted variant thereof bearing any combination, at any one ring position, of one or more substituents such as halogen, alkyl groups containing from 1 to 10 carbon atoms, trifluoromethyl, and alkoxy; (ii) a heteroaryl group such as a 2, 3, or 4-pyridyl group, which may additionally bear any combination of one or more substituents such as halogen, alkyl groups containing from 1 to 10 carbon atoms, trifluoromethyl and alkoxy; (iii) a five-membered ring aromatic heterocyclic group such as for example 2-thienyl, 3-thienyl, 2-thiazolyl, 4-thiazolyl, 5-thiazolyl, which may additionally bear any combination of one or more substitu
- H a halogen selected from I, F, Cl or Br
- NH2, NO2 or SO2-R wherein R is a linear or branched alkyl group containing one or more group such as 1 to 10 carbon atoms, and optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality.
- 011 1-[4-Methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-3-(4-trifluoromethyl-phenyl)-urea
- the invention is particularly embodied by the compounds wherein X is a -substituted Aryl group, corresponding to the N-[3-(Thiazol-2-ylamino)-phenyl]-amide family and the following formula II-3
- Ra, Rb, Rc, Rd, Re are independently chosen from H or an organic group that can be selected for example from a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group optionally substituted with a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; a —SO2-R group wherein R is an alkyl, cycloalkyl, aryl or heteroaryl optionally substituted with a heteroatom, notably a halogen selected from I, Cl, R
- a halogen such as I, Cl, Br and F
- R and R′ are H or a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group optionally substituted with a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality;
- R is H or a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group optionally substituted with a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; a —SO2-R′ group wherein R′ is an alkyl, cycloalkyl, aryl or heteroaryl optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality;
- Ra and Rb are H or a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group optionally substituted with a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality;
- Ra and Rb are H or a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group optionally substituted with a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality;
- R is a linear or branched alkyl group containing from 1 to 10 carbon atoms atoms optionally substituted with at least one heteroatom (for example a halogen) and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group substituted by an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality;
- Ra and Rb are a hydrogen or a linear or branched alkyl group containing from 1 to 10 carbon atoms atoms optionally substituted with at least one heteroatom (for example a halogen) and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group substituted by an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality;
- R is a linear or branched alkyl group containing from 1 to 10 carbon atoms atoms optionally substituted with at least one heteroatom (for example a halogen) and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group substituted by an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality;
- Ra and Rb are a linear or branched alkyl group containing from 1 to 10 carbon atoms atoms optionally substituted with at least one heteroatom (for example a halogen) and/or bearing a pendant basic nitrogen functionality;
- Ra can also be a hydrogen; a cycloalkyl, an aryl or heteroaryl group optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group substituted by an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality;
- R 4 is hydrogen, halogen or a linear or branched alkyl group containing from 1 to 10 carbon atoms, trifluoromethyl or alkoxy;
- R 6 is one of the following: (i) an aryl group such as phenyl or a substituted variant thereof bearing any combination, at any one ring position, of one or more substituents such as halogen, alkyl groups containing from 1 to 10 carbon atoms, trifluoromethyl, and alkoxy; (ii) a heteroaryl group such as a 2, 3, or 4-pyridyl group, which may additionally bear any combination of one or more substituents such as halogen, alkyl groups containing from 1 to 10 carbon atoms, trifluoromethyl and alkoxy; (iii) a five-membered ring aromatic heterocyclic group such as for example 2-thienyl, 3-thienyl, 2-thiazolyl, 4-thiazolyl, 5-thiazolyl, which may additionally bear any combination of one or more substituents such as halogen, an alkyl group containing from 1 to 10 carbon atoms, trifluoromethyl, and alkoxy; iv) H,
- the invention is particularly embodied by the compounds wherein X is a -substituted-aryl group, corresponding to the 4-(4-substituted-1-ylmethyl)-N-[3-(thiazol-2-ylamino)-phenyl]-benzamide family and the following formula II-4:
- X is a heteroatom, such as O or N
- Ra, Rb, Rd, Re, Rf, Rg, Rh are independently chosen from H or an organic group that can be selected for example from a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group optionally substituted with a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality;
- R and R′ are H or a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group optionally substituted with a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality;
- R is H or a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group optionally substituted with a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; a —SO2-R′ group wherein R′ is an alkyl, cycloalkyl, aryl or heteroaryl optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality;
- Ra and Rb are H or a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group optionally substituted with a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality;
- Ra and Rb are H or a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group optionally substituted with a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality;
- R is a linear or branched alkyl group containing from 1 to 10 carbon atoms atoms optionally substituted with at least one heteroatom (for example a halogen) and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group substituted by an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality;
- Ra and Rb are a hydrogen or a linear or branched alkyl group containing from 1 to 10 carbon atoms atoms optionally substituted with at least one heteroatom (for example a halogen) and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group substituted by an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality;
- R is a linear or branched alkyl group containing from 1 to 10 carbon atoms atoms optionally substituted with at least one heteroatom (for example a halogen) and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group substituted by an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality;
- R is a linear or branched alkyl group containing from 1 to 10 carbon atoms atoms optionally substituted with at least one heteroatom (for example a halogen) and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group substituted by an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality;
- Ra and Rb are a linear or branched alkyl group containing from 1 to 10 carbon atoms atoms optionally substituted with at least one heteroatom (for example a halogen) and/or bearing a pendant basic nitrogen functionality;
- Ra can also be a hydrogen; a cycloalkyl, an aryl or heteroaryl group optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group substituted by an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality;
- R is an alkyl, cycloalkyl, aryl or heteroaryl optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; or a —CO—R or a —CO—NRR′ group, wherein R and R′ are independently chosen from H, an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with at least one heteroatom, notably selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality.
- Ra, Rb, Rd, Re can also be halogen such as Cl, F, Br, I or trifluoromethyl;
- R 4 is hydrogen, halogen or a linear or branched alkyl group containing from 1 to 10 carbon atoms, trifluoromethyl or alkoxy
- R 6 is one of the following: (i) an aryl group such as phenyl or a substituted variant thereof bearing any combination, at any one ring position, of one or more substituents such as halogen, alkyl groups containing from 1 to 10 carbon atoms, trifluoromethyl, and alkoxy; (ii) a heteroaryl group such as a 2, 3, or 4-pyridyl group, which may additionally bear any combination of one or more substituents such as halogen, alkyl groups containing from 1 to 10 carbon atoms, trifluoromethyl and alkoxy; (iii) a five-membered ring aromatic heterocyclic group such as for example 2-thienyl, 3-thienyl, 2-thiazolyl, 4-thiazolyl, 5-thiazolyl, which may additionally bear any combination of one or more substitu
- the invention is particularly embodied by the compounds wherein X is a -aryl-substituted group, corresponding to the 3-Disubstituted-amino-N-[3-(thiazol-2-ylamino)-phenyl]-benzamide family and the following formula II-5:
- Ra, Rb, Rc, Re, Rf, Rg are independently chosen from H or an organic group that can be selected for example from a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group optionally substituted with a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality;
- R and R′ are H or a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group optionally substituted with a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality;
- R is H or a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group optionally substituted with a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; a —SO2-R′ group wherein R′ is an alkyl, cycloalkyl, aryl or heteroaryl optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality;
- Ra and Rb are H or a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group optionally substituted with a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality;
- Ra and Rb are H or a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group optionally substituted with a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality;
- R is a linear or branched alkyl group containing from 1 to 10 carbon atoms atoms optionally substituted with at least one heteroatom (for example a halogen) and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group substituted by an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality;
- Ra and Rb are a hydrogen or a linear or branched alkyl group containing from 1 to 10 carbon atoms atoms optionally substituted with at least one heteroatom (for example a halogen) and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group substituted by an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality;
- R is a linear or branched alkyl group containing from 1 to 10 carbon atoms atoms optionally substituted with at least one heteroatom (for example a halogen) and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group substituted by an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality;
- R is a linear or branched alkyl group containing from 1 to 10 carbon atoms atoms optionally substituted with at least one heteroatom (for example a halogen) and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group substituted by an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality;
- Ra and Rb are a linear or branched alkyl group containing from 1 to 10 carbon atoms atoms optionally substituted with at least one heteroatom (for example a halogen) and/or bearing a pendant basic nitrogen functionality;
- Ra can also be a hydrogen; a cycloalkyl, an aryl or heteroaryl group optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group substituted by an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality;
- R is an alkyl, cycloalkyl, aryl or heteroaryl optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; or a —CO—R or a —CO—NRR′ group, wherein R and R′ are independently chosen from H, an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with at least one heteroatom, notably selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality.
- Ra, Rb, Rc, Re can also be halogen such as Cl, F, Br, I or trifluoromethyl;
- R 4 is hydrogen, halogen or a linear or branched alkyl group containing from 1 to 10 carbon atoms, trifluoromethyl or alkoxy
- R 6 is one of the following: (i) an aryl group such as phenyl or a substituted variant thereof bearing any combination, at any one ring position, of one or more substituents such as halogen, alkyl groups containing from 1 to 10 carbon atoms, trifluoromethyl, and alkoxy; (ii) a heteroaryl group such as a 2, 3, or 4-pyridyl group, which may additionally bear any combination of one or more substituents such as halogen, alkyl groups containing from 1 to 10 carbon atoms, trifluoromethyl and alkoxy; (iii) a five-membered ring aromatic heterocyclic group such as for example 2-thienyl, 3-thienyl, 2-thiazolyl, 4-thiazolyl, 5-thiazolyl, which may additionally bear any combination of one or more substitu
- the invention is particularly embodied by the compounds wherein X is a —OR group, corresponding to the family [3-(Thiazol-2-ylamino)-phenyl]-carbamate and the following formula II-6
- R is independently chosen from an organic group that can be selected for example from a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F and/or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group optionally substituted with a cycloalkyl, an aryl or heteroaryl group optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F and/or bearing a pendant basic nitrogen functionality; R 4 is hydrogen, halogen or a linear or branched alkyl group containing from 1 to 10 carbon atoms, trifluoromethyl or alkoxy; R 6 is one of the following: (i) an aryl group such as phenyl
- the invention is directed to a process for manufacturing a compound of formula I depicted above. This entails the condensation of a substrate of general formula 10 with a thiourea of the type 11.
- Substituent “L” in formula 10 is a nucleofugal leaving group in nucleophilic substitution reactions (for example, L can be selected from chloro, bromo, iodo, toluenesulfonyloxy, methanesulfonyloxy, trifluoromethanesulfonyloxy, etc., with L being preferentially a bromo group).
- Group R1 in formula 11a corresponds to group R1 as described in formula I.
- Group “PG” in formula 11c is a suitable protecting group of a type commonly utilized by the person skilled in the art.
- Formula 12a is the same as formula I. Therefore, R1 in 12a corresponds to R1 in formula I.
- R1 the nature of which is as described on page 3 for the general formula I, is achieved by the use of standard reactions that are well known to the person skilled in the art, such as alkylation, acylation, sulfonylation, formation of ureas, etc.
- Formula 12c describes an N-protected variant of compound 12b.
- Group “PG” in formula 12c represents a protecting group of the type commonly utilized by the person skilled in the art. Therefore, in a second phase of the synthesis, group PG is cleaved to transform compound 12c into compound 12b. Compound 12b is subsequently advanced to structures of formula I as detailed above.
- Formula 12d describes a nitro analogue of compound 12b.
- the nitro group of compound 12d is reduced by any of the several methods utilized by the person skilled in the art to produce the corresponding amino group, namely compound 12b.
- Compound 12b thus obtained is subsequently advanced to structures of formula I as detailed above.
- aqueous phase was then basified (pH>12) by addition of 2.5N aqueous sodium hydroxyde solution.
- the crude product was extracted with ethyl acetate (4 ⁇ 30 mL). The combined organic layers were dried over MgSO 4 and concentrated under reduced pressure to afford a slightly yellow oil which became colorless after purification by Kugelrohr distillation (190° C.) in 68% yield.
- Benzoyl chloride (5.64 g, 80 mmol) was added dropwise to a well-stirred solution of ammonium thiocyanate (3.54 g, 88 mmol) in acetone (50 mL). The mixture was refluxed for 15 min, then, the hydrobromide salt of 2-methyl-5-tert-butoxycarbonylamino-aniline (8.4 g, 80 mmol) was added slowly portionwise. After 1 h, the reaction mixture was poured into ice-water (350 mL) and the bright yellow precipitate was isolated by filtration. This crude solid was then refluxed for 45 min in 70 mL of 2.5 N sodium hydroxide solution. The mixture was cooled down and basified with ammonium hydroxide.
- the invention relates to a pharmaceutical composition
- a pharmaceutical composition comprising a compound as depicted above.
- Such medicament can take the form of a pharmaceutical composition adapted for oral administration, which can be formulated using pharmaceutically acceptable carriers well known in the art in suitable dosages.
- pharmaceutically acceptable carriers well known in the art in suitable dosages.
- Such carriers enable the pharmaceutical compositions to be formulated as tablets, pills, dragees, capsules, liquids, gels, syrups, slurries, suspensions, and the like, for ingestion by the patient.
- these pharmaceutical compositions may contain suitable pharmaceutically-acceptable carriers comprising excipients and auxiliaries which facilitate processing of the active compounds into preparations which can be used pharmaceutically. Further details on techniques for formulation and administration may be found in the latest edition of Remington's Pharmaceutical Sciences (Maack Publishing Co., Easton, Pa.).
- composition of the invention can also take the form of a pharmaceutical or cosmetic composition for topical administration.
- compositions may be presented in the form of a gel, paste, ointment, cream, lotion, liquid suspension aqueous, aqueous-alcoholic or, oily solutions, or dispersions of the lotion or serum type, or anhydrous or lipophilic gels, or emulsions of liquid or semi-solid consistency of the milk type, obtained by dispersing a fatty phase in an aqueous phase or vice versa, or of suspensions or emulsions of soft, semi-solid consistency of the cream or gel type, or alternatively of microemulsions, of microcapsules, of microparticles or of vesicular dispersions to the ionic and/or nonionic type.
- These compositions are prepared according to standard methods.
- composition according to the invention comprises any ingredient commonly used in dermatology and cosmetic. It may comprise at least one ingredient selected from hydrophilic or lipophilic gelling agents, hydrophilic or lipophilic active agents, preservatives, emollients, viscosity enhancing polymers, humectants, surfactants, preservatives, antioxidants, solvents, and fillers, antioxidants, solvents, perfumes, fillers, screening agents, bactericides, odor absorbers and coloring matter.
- oils which can be used in the invention mineral oils (liquid paraffin), vegetable oils (liquid fraction of shea butter, sunflower oil), animal oils, synthetic oils, silicone oils (cyclomethicone) and fluorinated oils may be mentioned.
- Fatty alcohols, fatty acids (stearic acid) and waxes (paraffin, carnauba, beeswax) may also be used as fatty substances.
- glycerol stearate As emulsifiers which can be used in the invention, glycerol stearate, polysorbate 60 and the PEG-6/PEG-32/glycol stearate mixture are contemplated.
- hydrophilic gelling agents carboxyvinyl polymers (carbomer), acrylic copolymers such as acrylate/alkylacrylate copolymers, polyacrylamides, polysaccharides such as hydroxypropylcellulose, clays and natural gums may be mentioned, and as lipophilic gelling agents, modified clays such as bentones, metal salts of fatty acids such as aluminum stearates and hydrophobic silica, or alternatively ethylcellulose and polyethylene may be mentioned.
- hydrophilic active agents proteins or protein hydrolysates, amino acids, polyols, urea, allantoin, sugars and sugar derivatives, vitamins, starch and plant extracts, in particular those of Aloe vera may be used.
- agents As lipophilic active, agents, retinol (vitamin A) and its derivatives, tocopherol (vitamin E) and its derivatives, essential fatty acids, ceramides and essential oils may be used. These agents add extra moisturizing or skin softening features when utilized.
- a surfactant can be included in the composition so as to provide deeper penetration of the compound capable of depleting mast cells, such as a tyrosine kinase inhibitor, preferably a c-kit inhibitor.
- the invention embraces penetration enhancing agents selected for example from the group consisting of mineral oil, water, ethanol, triacetin, glycerin and propylene glycol; cohesion agents selected for example from the group consisting of polyisobutylene, polyvinyl acetate and polyvinyl alcohol, and thickening agents.
- compounds with penetration enhancing properties include sodium lauryl sulfate (Dugard, P. H. and Sheuplein, R. J., “Effects of Ionic Surfactants on the Permeability of Human Epidermis: An Electrometric Study,” J. Ivest. Dermatol., V. 60, pp. 263-69, 1973), lauryl amine oxide (Johnson et. al., U.S. Pat. No. 4,411,893), azone (Rajadhyaksha, U.S. Pat. Nos. 4,405,616 and 3,989,816) and decylmethyl sulfoxide (Sekura, D. L.
- a second class of chemical enhancers are generally referred to as co-solvents. These materials are absorbed topically relatively easily, and, by a variety of mechanisms, achieve permeation enhancement for some drugs.
- Ethanol (Gale et. al., U.S. Pat. No. 4,615,699 and Campbell et. al., U.S. Pat. Nos. 4,460,372 and 4,379,454)
- dimethyl sulfoxide U.S. Pat. Nos. 3,740,420 and 3,743,727, and U.S. Pat. No. 4,575,515)
- glycerine derivatives U.S. Pat. No. 4,322,433 are a few examples of compounds which have shown an ability to enhance the absorption of various compounds.
- compositions of the invention can also be intended for administration with aerosolized formulation to target areas of a patient's respiratory tract.
- Formulations are preferably solutions, e.g. aqueous solutions, ethanoic solutions, aqueous/ethanoic solutions, saline solutions, colloidal suspensions and microcrystalline suspensions.
- aerosolized particles comprise the active ingredient mentioned above and a carrier, (e.g., a pharmaceutically active respiratory drug and carrier) which are formed upon forcing the formulation through a nozzle which nozzle is preferably in the form of a flexible porous membrane.
- the particles have a size which is sufficiently small such that when the particles are formed they remain suspended in the air for a sufficient amount of time such that the patient can inhale the particles into the patient's lungs.
- liquid gas systems a liquefied gas is used as propellent gas (e.g. low-boiling FCHC or propane, butane) in a pressure container,
- propellent gas e.g. low-boiling FCHC or propane, butane
- suspension aerosol the active substance particles are suspended in solid form in the liquid propellent phase
- pressurized gas system a compressed gas such as nitrogen, carbon dioxide, dinitrogen monoxide, air is used.
- the pharmaceutical preparation is made in that the active substance is dissolved or dispersed in a suitable nontoxic medium and said solution or dispersion atomized to an aerosol, i.e. distributed extremely finely in a carrier gas.
- aerosol i.e. distributed extremely finely in a carrier gas.
- the invention is also directed to aerosol devices comprising the compound as defined above and such a formulation, preferably with metered dose valves.
- compositions of the invention can also be intended for intranasal administration.
- pharmaceutically acceptable carriers for administering the compound to the nasal mucosal surfaces will be readily appreciated by the ordinary artisan. These carriers are described in the Remington's Pharmaceutical Sciences” 16th edition, 1980, Ed. By Arthur Osol, the disclosure of which is incorporated herein by reference.
- the composition can be formulated into a solution, e.g., water or isotonic saline, buffered or unbuffered, or as a suspension, for intranasal administration as drops or as a spray.
- a solution e.g., water or isotonic saline, buffered or unbuffered, or as a suspension
- such solutions or suspensions are isotonic relative to nasal secretions and of about the same pH, ranging e.g., from about pH 4.0 to about pH 7.4 or, from pH 6.0 to pH 7.0.
- Buffers should be physiologically compatible and include, simply by way of example, phosphate buffers.
- a representative nasal decongestant is described as being buffered to a pH of about 6.2 (Remington's, Id. at page 1445).
- a suitable saline content and pH for an innocuous aqueous carrier for nasal and/or upper respiratory administration is described as being buffered to a pH of about 6.2 (Remington's, Id. at page 1445).
- the ordinary artisan can readily determine a suitable saline content and pH for an innocuous aqueous carrier for nasal and/or upper respiratory administration.
- Common intranasal carriers include nasal gels, creams, pastes or ointments with a viscosity of, e.g., from about 10 to about 3000 cps, or from about 2500 to 6500 cps, or greater, may also be used to provide a more sustained contact with the nasal mucosal surfaces.
- Such carrier viscous formulations may be based upon, simply by way of example, alkylcelluloses and/or other biocompatible carriers of high viscosity well known to the art (see e.g., Remington's, cited supra.
- a preferred alkylcellulose is, e.g., methylcellulose in a concentration ranging from about 5 to about 1000 or more mg per 100 ml of carrier.
- a more preferred concentration of methyl cellulose is, simply by way of example, from about 25 to about mg per 100 ml of carrier.
- ingredients such as art known preservatives, colorants, lubricating or viscous mineral or vegetable oils, perfumes, natural or synthetic plant extracts such as aromatic oils, and humectants and viscosity enhancers such as, e.g., glycerol, can also be included to provide additional viscosity, moisture retention and a pleasant texture and odor for the formulation.
- various devices are available in the art for the generation of drops, droplets and sprays.
- a premeasured unit dosage dispenser including a dropper or spray device containing a solution or suspension for delivery as drops or as a spray is prepared containing one or more doses of the drug to be administered and is another object of the invention.
- the invention also includes a kit containing one or more unit dehydrated doses of the compound, together with any required salts and/or buffer agents, preservatives, colorants and the like, ready for preparation of a solution or suspension by the addition of a suitable amount of water.
- Another aspect of the invention is directed to the use of said compound to manufacture a medicament.
- the invention embraces a method for treating a disease related to unregulated c-kit transduction comprising administering an effective amount of a compound as defined above to a mammal in need of such treatment.
- the invention is aimed at a method for treating a disease selected from autoimmune diseases, allergic diseases, bone loss, cancers such as leukemia and GIST, tumor angiogenesis, inflammatory diseases, inflammatory bowel diseases (IBD), interstitial cystitis, mastocytosis, infections diseases, metabolic disorders, fibrosis, diabetes and CNS disorders comprising administering an effective amount a compound depicted above to a mammal in need of such treatment.
- a disease selected from autoimmune diseases, allergic diseases, bone loss, cancers such as leukemia and GIST, tumor angiogenesis, inflammatory diseases, inflammatory bowel diseases (IBD), interstitial cystitis, mastocytosis, infections diseases, metabolic disorders, fibrosis, diabetes and CNS disorders
- the above described compounds are useful for manufacturing a medicament for the treatment of diseases related to unregulated c-kit transduction, including, but not limited to:
- the invention contemplates the use of the compounds as defined above for treating the different categories which can be classified as follows:
- the category I is composed by two sub-categories (IA and IB).
- Category IA is made by diseases in which mast cell infiltration is strictly localized to the skin. This category represents the most frequent form of the disease and includes: i) urticaria pigmentosa, the most common form of cutaneous mastocytosis, particularly encountered in children, ii) diffuse cutaneous mastocytosis, iii) solitary mastocytoma and iv) some rare subtypes like bullous, erythrodermic and teleangiectatic mastocytosis. These forms are characterized by their excellent prognosis with spontaneous remissions in children and a very indolent course in adults.
- SM systemic disease
- the category II includes mastocytosis with an associated hematological disorder, such as a myeloproliferative or myelodysplastic syndrome, or acute leukemia. These malignant mastocytosis does not usually involve the skin. The progression of the disease depends generally on the type of associated hematological disorder that conditions the prognosis.
- an associated hematological disorder such as a myeloproliferative or myelodysplastic syndrome, or acute leukemia.
- the category III is represented by aggressive systemic mastocytosis in which massive infiltration of multiple organs by abnormal mast cells is common. In patients who pursue this kind of aggressive clinical course, peripheral blood features suggestive of a myeloproliferative disorder are more prominent. The progression of the disease can be very rapid, similar to acute leukemia, or some patients can show a longer survival time.
- mast cell leukemia characterized by the presence of circulating mast cells and mast cell progenitors representing more than 10% of the white blood cells. This entity represents probably the rarest type of leukemia in humans, and has a very poor prognosis, similar to the rapidly progressing variant of malignant mastocytosis. Mast cell leukemia can occur either de novo or as the terminal phase of urticaria pigmentosa or systemic mastocytosis.
- the invention also contemplates the method as depicted for the treatment of recurrent bacterial infections, resurging infections after asymptomatic periods such as bacterial cystitis. More particularly, the invention can be practiced for treating FimH expressing bacteria infections such as Gram-negative enterobacteria including E. coli, Klebsiella pneumoniae, Serratia marcescens, Citrobactor freudii and Salmonella typhimurium.
- FimH expressing bacteria infections such as Gram-negative enterobacteria including E. coli, Klebsiella pneumoniae, Serratia marcescens, Citrobactor freudii and Salmonella typhimurium.
- the invention is directed to a method for treating neoplastic diseases such as mastocytosis, canine mastocytoma, human gastrointestinal stromal tumor (“GIST”), small cell lung cancer, non-small cell lung cancer, acute myelocytic leukemia, acute lymphocytic leukemia, myelodysplastic syndrome, chronic myelogenous leukemia, colorectal carcinomas, gastric carcinomas, gastrointestinal stromal tumors, testicular cancers, glioblastomas, and astrocytomas comprising administering a compound as defined herein to a human or mammal, especially dogs and cats, in need of such treatment.
- GIST human gastrointestinal stromal tumor
- the invention is directed to a method for treating allergic diseases such as asthma, allergic rhinitis, allergic sinusitis, anaphylactic syndrome, urticaria, angioedema, atopic dermatitis, allergic contact dermatitis, erythema nodosum, erythema multiforme, cutaneous necrotizing venulitis and insect bite skin inflammation and blood sucking parasitic infestation
- allergic diseases such as asthma, allergic rhinitis, allergic sinusitis, anaphylactic syndrome, urticaria, angioedema, atopic dermatitis, allergic contact dermatitis, erythema nodosum, erythema multiforme, cutaneous necrotizing venulitis and insect bite skin inflammation and blood sucking parasitic infestation
- administering a compound as defined herein to a human or mammal, especially dogs and cats, in need of such treatment.
- the invention is directed to a method for treating inflammatory diseases such as rheumatoid arthritis, conjunctivitis, rheumatoid spondylitis, osteoarthritis, gouty arthritis and other arthritic conditions comprising administering a compound as defined herein to a human in need of such treatment.
- inflammatory diseases such as rheumatoid arthritis, conjunctivitis, rheumatoid spondylitis, osteoarthritis, gouty arthritis and other arthritic conditions
- the invention is directed to a method for treating autoimmune diseases such as multiple sclerosis, psoriasis, intestine inflammatory disease, ulcerative colitis, Crohn's disease, rheumatoid arthritis and polyarthritis, local and systemic scleroderma, systemic lupus erythematosus, discoid lupus erythematosus, cutaneous lupus, dermatomyositis, polymyositis, Sjogren's syndrome, nodular panarteritis, autoimmune enteropathy, as well as proliferative glomerulonephritis comprising administering a compound as defined herein to a human in need of such treatment.
- autoimmune diseases such as multiple sclerosis, psoriasis, intestine inflammatory disease, ulcerative colitis, Crohn's disease, rheumatoid arthritis and polyarthritis
- local and systemic scleroderma systemic lupus ery
- the invention is directed to a method for treating graft-versus-host disease or graft rejection in any organ transplantation including kidney, pancreas, liver, heart, lung, and bone marrow comprising administering a compound as defined herein to a human in need of such treatment.
- Table 1 shows the potent inhibitory action of the catalytic activity of c-kit with an IC50 ⁇ 10 ⁇ M. Further experiments (not shown) indicates that at least one compound acts as perfect competitive inhibitors of ATP.
- Ba/F3 murine kit and human kit, Ba/F3 mkit ⁇ 27 are derived from the murine IL-3 dependent Ba/F3 proB lymphoid cells.
- the FMA3 and P815 cell lines are mastocytoma cells expressing endogenous mutated forms of Kit, i.e., frame deletion in the murine juxtamembrane coding region of the receptor-codons 573 to 579.
- the human leukaemic MC line HMC-1 expresses mutations JM-V560G;
- the membrane was then incubated either with HRP-conjugated goat anti mouse IgG antibody or with HRP-conjugated goat anti rabbit IgG antibody (Immunotech), Proteins of interest were then visualized by incubation with ECL reagent (Amersham).
- Ba/F3 cells were transfected by c-kit gene having A27 mutation (GIST model). Ba/F3 expressing the mutated c-kit gene readily proliferate in the absence of IL3 or SCF and are tumorigenic in nude mice.
- mice were irradiated at J-1 (5Gy) Tumor cells (10 6 ) were subcutaneously grafted at Jo Tumor size were daily measured from J14 Number of survival mice were daily estimated In this experimental model, the tumor size at J14 is about 20 mm 3 Treated mice received per os twice a day a dose of 100 mg/kg of one compound of formula II-3 during 5 days (from J26 to J30).
- mice were pretreated with the compound of formula II-3 (2 ⁇ , 12.5 mg/kg) for two days (day ⁇ 2, day ⁇ 1) before induction of arthritis.
- Arthritis was induced by ip injection of 150-ul serums at days 0 and 2.
- the treatment with the compound (2 ⁇ , 12.5 mg/kg) was continued for 14 days.
- the control mice were injected with, 1% PBS before the induction of arthritis and during the course of the disease.
- Ankle thickness and arthritis score was evaluated for 15 days.
- Table 3A and Table 3B show the number of mice used in this study. Two sets of experiments were done with different number of mice, one with 4 mices the other with 8 mices.
- Treated mice displays significant decrease of tumor size at J30 and J33 compared to control.
- one tested compound of the formula II-3 displays a significant antitumor activity against tumors cells expressing c-kit A27.
- a compound of the formula II-3 has demonstrated significant activity in the in vivo mouse model of arthritis. Results are shown on FIGS. 1 , 2 , 3 , 4 .
- FIG. 1 Effect of the compound in serum transfer experiments, Protocol, ip daily treatment with the compound (2 ⁇ 12.5 mg/kg) and on days ⁇ 2 and ⁇ 1, set of experiment with 4 mices (T: treated, C: control)
- FIG. 2 Effect of the compound in serum transfer experiments, Protocol, ip daily treatment with the compound (2 ⁇ 12.5 mg/kg) and on days ⁇ 2 and ⁇ 1, set of experiment with 4 mices (T: treated, C: control)
- FIG. 3 Effect of the compound in serum transfer experiments, Protocol, ip daily treatment with the compound (2 ⁇ 12.5 mg/kg) and on days ⁇ 2 and ⁇ 1, set of experiment with 8 mices (T: treated, C: control)
- FIG. 4 Effect of the compound in serum transfer experiments, Protocol, ip daily treatment with the compound (2 ⁇ 12.5 mg/kg) and on days ⁇ 2 and ⁇ 1, set of experiment with 8 mices (T: treated, C: control)
Abstract
Novel compounds selected from 2-(3-aminoaryl)amino-4-aryl-thiazoles of formula (I) that selectively modulate, regulate, and/or inhibit signal transductions mediated by certain native and/or mutant tyrosine kinases implicated in a variety of human and animal diseases such as cell proliferation metabolic, allergic and degenerative disorders. More particularly, these compounds are potent and selective c-kit inhibitors.
Description
- This application is a continuation of U.S. patent application Ser. No. 10/523,018 filed on Feb. 2, 2005, which is a national stage of PCT/IB03/03685 filed under 35 U.S.C. § 371 on Jul. 31, 2003, which claims the benefit of U.S. Provisional Application No. 60/400,064 filed on Aug. 2, 2002, the complete disclosures of which are incorporated into this application by reference. This application also claims the benefit of U.S. patent application Ser. No. 10/632,101 filed Aug. 1, 2003, which claims the benefit of U.S. Provisional Application No. 60/400,064 filed on Aug. 2, 2002.
- The present invention relates to novel compounds selected from 2-(3-aminoaryl)amino-4-aryl-thiazoles that selectively modulate, regulate, and/or inhibit signal transduction mediated by certain native and/or mutant tyrosine kinases implicated in a variety of human and animal diseases such as cell proliferative, metabolic, allergic, and degenerative disorders. More particularly, these compounds are potent and selective c-kit inhibitors.
- Tyrosine kinases are receptor type or non-receptor type proteins, which transfer the terminal phosphate of ATP to tyrosine residues of proteins thereby activating or inactivating signal transduction pathways. These proteins are known to be involved in many cellular mechanisms, which in case of disruption, lead to disorders such as abnormal cell proliferation and migration as well as inflammation.
- As of today, there are about 58 known receptor tyrosine kinases. Other tyrosine kinases are the well-known VEGF receptors (Kim et al., Nature 362, pp. 841-844, 1993), PDGF receptors, c-kit and the FLK family. These receptors can transmit signals to other tyrosine kinases including Src, Raf, Frk, Btk, Csk, Abl, Fes/Fps, Fak, Jak, Ack. etc.
- Among tyrosine kinase receptors, c-kit is of special interest. Indeed, c-kit is a key receptor activating mast cells, which have proved to be directly or indirectly implicated in numerous pathologies for which the Applicant filed WO 03/004007, WO 03/004006, WO 03/003006, WO 03/003004, WO 03/002114, WO 03/002109, WO 03/002108, WO 03/002107, WO 03/002106, WO 03/002105, WO 03/039550, WO 03/035050, WO 03/035049, U.S. 60/359,652 and U.S. 60/359,651.
- It was found that mast cells present in tissues of patients are implicated in or contribute to the genesis of diseases such as autoimmune diseases (rheumatoid arthritis, inflammatory bowel diseases (IBD)) allergic diseases, tumor angiogenesis, inflammatory diseases, and interstitial cystitis. In these diseases, it has been shown that mast cells participate in the destruction of tissues by releasing a cocktail of different proteases and mediators such as histamine, neutral proteases, lipid-derived mediators (prostaglandins, thromboxanes and leucotrienes), and various cytokines (IL-1, IL-2, IL-3, IL-4, IL-5, IL-6, IL-8, TNF-α, GM-CSF, MIP-1a, MIP-1b, MIP-2 and IFN-γ).
- The c-kit receptor also can be constitutively activated by mutations leading to abnormal cell proliferation and development of diseases such as mastocytosis and various cancers.
- For this reason, it has been proposed to target c-kit to deplete the mast cells responsible for these disorders.
- The main objective underlying the present invention is therefore to find potent and selective compounds capable of inhibiting wild type and/or mutated c-kit.
- Many different compounds have been described as tyrosine kinase inhibitors, for example, bis monocyclic, bicyclic or heterocyclic aryl compounds (WO 92/20642), vinylene-azaindole derivatives (WO 94/14808) and 1-cycloproppyl-4-pyridyl-quinolones (U.S. Pat. No. 5,330,992), styryl compounds (U.S. Pat. No. 5,217,999), styryl-substituted pyridyl compounds (U.S. Pat. No. 5,302,606), selenoindoles and selenides (WO 94/03427), tricyclic polyhydroxylic compounds (WO 92/21660) and benzylphosphonic acid compounds (WO 91/15495), pyrimidine derivatives (U.S. Pat. No. 5,521,184 and WO 99/03854), indolinone derivatives and pyrrole-substituted indolinones (U.S. Pat. No. 5,792,783, EP 934 931, U.S. Pat. No. 5,834,504, U.S. Pat. No. 5,883,116, U.S. Pat. No. 5,883,113, U.S. Pat. No. 5,886,020, WO 96/40116 and WO 00/38519), as well as bis monocyclic, bicyclic aryl and heteroaryl compounds (EP 584 222, U.S. Pat. No. 5,656,643 and WO 92/20642), quinazoline derivatives (EP 602 851, EP 520 722, U.S. Pat. No. 3,772,295 and U.S. Pat. No. 4,343,940) and aryl and heteroaryl quinazoline (U.S. Pat. No. 5,721,237, U.S. Pat. No. 5,714,493, U.S. Pat. No. 5,710,158 and WO 95/15758).
- However, none of these compounds have been described as potent and selective inhibitors of c-kit or of the c-kit pathway.
- In connection with the present invention, we have found that compounds corresponding to the 2-(3-aminoaryl)amino-4-aryl-thiazoles are potent and selective inhibitors of c-kit or c-kit pathway. These compounds are good candidates for treating diseases such as autoimmunes diseases, inflammatory diseases, cancer and mastocytosis.
- Therefore, the present invention relates to compounds belonging to the 2-(3-amino)arylamino-4-aryl-thiazoles. These compounds are capable of selectively inhibiting signal transduction involving the tyrosine phosphokinase c-kit and mutant forms thereof.
- In a first embodiment, the invention is aimed at compounds of formula I, which may represent either free base forms of the substances or pharmaceutically acceptable salts thereof:
- and wherein R1 is:
a) a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality;
b) an aryl or heteroaryl group optionally substituted by an alkyl or aryl group optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality;
c) a —CO—NH—R, —CO—R, —CO—OR or a —CO—NRR′ group, wherein R and R′ are independently chosen from H or an aryl, heteroaryl, alkyl and cycloalkyl group optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality;
R2 is hydrogen, halogen or a linear or branched alkyl group containing from 1 to 10 carbon atoms, trifluoromethyl or alkoxy;
R3 is hydrogen, halogen or a linear or branched alkyl group containing from 1 to 10 carbon atoms, trifluoromethyl or alkoxy;
R4 is hydrogen, halogen or a linear or branched alkyl group containing from 1 to 10 carbon atoms, trifluoromethyl or alkoxy;
R5 is hydrogen, halogen or a linear or branched alkyl group containing from 1 to 10 carbon atoms, trifluoromethyl or alkoxy;
R6 is one of the following:
(i) an aryl group such as phenyl or a substituted variant thereof bearing any combination, at any one ring position, of one or more substituents such as halogen, alkyl groups containing from 1 to 10 carbon atoms, trifluoromethyl, and alkoxy;
(ii) a heteroaryl group such as a 2, 3, or 4-pyridyl group, which may additionally bear any combination of one or more substituents such as halogen, alkyl groups containing from 1 to 10 carbon atoms, trifluoromethyl and alkoxy;
(iii) a five-membered ring aromatic heterocyclic group such as for example 2-thienyl, 3-thienyl, 2-thiazolyl, 4-thiazolyl, 5-thiazolyl, which may additionally bear any combination of one or more substituents such as halogen, an alkyl group containing from 1 to 10 carbon atoms, trifluoromethyl, and alkoxy,
iv) H, a halogen selected from I, F, Cl or Br; NH2, NO2 or SO2-R, wherein R is a linear or branched alkyl group containing one or more group such as 1 to 10 carbon atoms, and optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality; and R7 is one of the following:
(i) an aryl group such as phenyl or a substituted variant thereof bearing any combination, at any one ring position, of one or more substituents such as halogen, alkyl groups containing from 1 to 10 carbon atoms, trifluoromethyl, and alkoxy;
(ii) a heteroaryl group such as a 2, 3, or 4-pyridyl group, which may additionally bear any combination of one or more substituents such as halogen, alkyl groups containing from 1 to 10 carbon atoms, trifluoromethyl and alkoxy;
(iii) a five-membered ring aromatic heterocyclic group such as for example 2-thienyl, 3-thienyl, 2-thiazolyl, 4-thiazolyl, 5-thiazolyl, which may additionally bear any combination of one or more substituents such as halogen, an alkyl group containing from 1 to 10 carbon atoms, trifluoromethyl, and alkoxy.
iv) H, a halogen selected from I, F, Cl or Br; NH2, NO2 or SO2-R, wherein R is a linear or branched alkyl group containing one or more group such as 1 to 10 carbon atoms, and optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality; - In another preferred embodiment, when R1 has the meaning depicted in c) above, the invention is directed to compounds of the following formula:
- wherein R is H or an organic group that can be selected for example from a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted by an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F and/or bearing a pendant basic nitrogen functionality.
- Among the particular compounds in which R1 has the meaning as depicted in c) above, the invention is directed to amide-aniline compounds of the following formula:
- wherein R is H or an organic group that can be selected for example from a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F and/or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group optionally substituted with a cycloalkyl, an aryl or heteroaryl group optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F and/or bearing a pendant basic nitrogen functionality;
a —SO2-R group wherein R is an alkyl, cycloalkyl, aryl or heteroaryl optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F and/or bearing a pendant basic nitrogen functionality; or a —CO—R or a —CO—NRR′ group, wherein R and R′ are independently chosen from H, an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with at least one heteroatom, notably selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality. - Among the particular compounds in which R1 has the meaning as depicted in c) above, the invention is directed to amide-benzylamine compounds of the following formula:
- wherein R is H or an organic group that can be selected for example from a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; or an alkyl, cycloalkyl, aryl or heteroaryl group substituted by a alkyl, cycloalkyl, aryl or heteroaryl group optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality;
a —SO2-R group wherein R is an alkyl, cycloalkyl, aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; or a —CO—R or a —CO—NRR′ group, wherein R and R′ are independently chosen from H or an aryl heteroaryl, alkyl and cycloalkyl group optionally substituted with at least one heteroatom and/or bearing a pendant basic nitrogen functionality. - Among the particular compounds in which R1 has the meaning as depicted in c) above, the invention is directed to amide-phenol compounds of the following formula:
- wherein R is H or an organic group that can be selected for example from a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality;
a cycloalkyl, aryl or heteroaryl group optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F and/or bearing a pendant basic nitrogen functionality; or an alkyl, cycloalkyl, aryl or heteroaryl group substituted by a alkyl, cycloalkyl, aryl or heteroaryl group optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F and/or bearing a pendant basic nitrogen functionality;
a —SO2-R group wherein R is an alkyl, cycloalkyl, aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F and/or bearing a pendant basic nitrogen functionality; or a —CO—R or a —CO—NRR′ group, wherein R and R′ are independently chosen from H or an aryl, heteroaryl, alkyl and cycloalkyl group optionally substituted with at least one heteroatom and/or bearing a pendant basic nitrogen functionality. - Among the particular compounds in which R1 has the meaning as depicted in c) above, the invention is directed to urea compounds of the following formula:
- wherein R is H or an organic group that can be selected for example from a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom (for example a halogen) and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group substituted by an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality.
- Among the particular compounds in which R1 has the meaning as depicted in a) and b) above, the invention is directed to N-Aminoalkyl-N′-thiazol-2-yl-benzene-1,3-diamine compounds of the following formula:
- wherein Y is a linear or branched alkyl group containing from 1 to 10 carbon atoms;
wherein Z represents an aryl or heteroaryl group, optionally substituted at one or more ring position with any permutation of the following groups: -
- a halogen such as F, Cl, Br, I;
- a linear or branched alkyl group containing from 1 to 10 carbon atoms atoms optionally substituted with at least one heteroatom (for example a halogen) and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group substituted by an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality;
- an O—R, where R is a linear or branched alkyl group containing from 1 to 10 carbon atoms atoms optionally substituted with at least one heteroatom (for example a halogen) and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group substituted by an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality;
- an NRaRb, where Ra and Rb represents a hydrogen, or a linear or branched alkyl group containing from 1 to 10 carbon atoms atoms optionally substituted with at least one heteroatom (for example a halogen) and/or bearing a pendant basic nitrogen functionality or a cycle; a cycloalkyl, an aryl or heteroaryl group optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group substituted by an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality;
- a COOR, where R is a linear or branched alkyl group containing from 1 to 10 carbon atoms atoms optionally substituted with at least one heteroatom (for example a halogen) and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group substituted by an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality;
- a CONRaRb, where Ra and Rb are a hydrogen or a linear or branched alkyl group containing from 1 to 10 carbon atoms atoms optionally substituted with at least one heteroatom (for example a halogen) and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group substituted by an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality;
- an NHCOR, where R is a linear or branched alkyl group containing from 1 to 10 carbon atoms atoms optionally substituted with at least one heteroatom (for example a halogen) and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group substituted by an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality;
- an NHCOOR, where R is a linear or branched alkyl group containing from 1 to 10 carbon atoms atoms optionally substituted with at least one heteroatom (for example a halogen) and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group substituted by an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality;
- an NHCONRaRb, where Ra and Rb are a hydrogen or a linear or branched alkyl group containing from 1 to 10 carbon atoms atoms optionally substituted with at least one heteroatom (for example a halogen) and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group substituted by an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality;
- an OSO2R, where R is a linear or branched alkyl group containing from 1 to 10 carbon atoms atoms optionally substituted with at least one heteroatom (for example a halogen) and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group substituted by an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality;
- an NRaOSO2Rb, where Ra and Rb are a linear or branched alkyl group containing from 1 to 10 carbon atoms atoms optionally substituted with at least one heteroatom (for example a halogen) and/or bearing a pendant basic nitrogen functionality; Ra can also be a hydrogen; a cycloalkyl, an aryl or heteroaryl group optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group substituted by an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality;
R2 is hydrogen, halogen or a linear or branched alkyl group containing from 1 to 10 carbon atoms, trifluoromethyl or alkoxy;
R3 is hydrogen, halogen or a linear or branched alkyl group containing from 1 to 10 carbon atoms, trifluoromethyl or alkoxy;
R4 is hydrogen, halogen or a linear or branched alkyl group containing from 1 to 10 carbon atoms, trifluoromethyl or alkoxy;
R5 is hydrogen, halogen or a linear or branched alkyl group containing from 1 to 10 carbon atoms, trifluoromethyl or alkoxy;
R6 is one of the following:
(i) an aryl group such as phenyl or a substituted variant thereof bearing any combination, at any one ring position, of one or more substituents such as halogen, alkyl groups containing from 1 to 10 carbon atoms, trifluoromethyl, and alkoxy;
(ii) a heteroaryl group such as a 2, 3, or 4-pyridyl group, which may additionally bear any combination of one or more substituents such as halogen, alkyl groups containing from 1 to 10 carbon atoms, trifluoromethyl and alkoxy;
(iii) a five-membered ring aromatic heterocyclic group such as for example 2-thienyl, 3-thienyl, 2-thiazolyl, 4-thiazolyl, 5-thiazolyl, which may additionally bear any combination of one or more substituents such as halogen, an alkyl group containing from 1 to 10 carbon atoms, trifluoromethyl, and alkoxy.
iv) H, a halogen selected from I, F, Cl or Br; NH2, NO2 or SO2-R, wherein R is a linear or branched alkyl group containing one or more group such as 1 to 10 carbon atoms, and optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality; and R7 is one of the following:
(i) an aryl group such as phenyl or a substituted variant thereof bearing any combination, at any one ring position, of one or more substituents such as halogen, alkyl groups containing from 1 to 10 carbon atoms, trifluoromethyl, and alkoxy;
(ii) a heteroaryl group such as a 2, 3, or 4-pyridyl group, which may additionally bear any combination of one or more substituents such as halogen, alkyl groups containing from 1 to 10 carbon atoms, trifluoromethyl and alkoxy;
(iii) a five-membered ring aromatic heterocyclic group such as for example 2-thienyl, 3-thienyl, 2-thiazolyl, 4-thiazolyl, 5-thiazolyl, which may additionally bear any combination of one or more substituents such as halogen, an alkyl group containing from 1 to 10 carbon atoms, trifluoromethyl, and alkoxy.
iv) H, an halogen selected from I, F, Cl or Br; NH2, NO2 or SO2-R, wherein R is a linear or branched alkyl group containing one or more group such as 1 to 10 carbon atoms, and optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality.
- An example of preferred compounds of the above formula is depicted below:
-
- Among the compounds of formula I, the invention is particularly embodied by the compounds of the following formula II:
- wherein X is R or NRR′ and wherein R and R′ are independently chosen from H, an aryl, a heteroaryl, an alkyl, or a cycloalkyl group optionally substituted with at least one heteroatom, such as for example a halogen chosen from F, I, Cl and Br and optionally bearing a pendant basic nitrogen functionality; or an aryl, a heteroaryl, an alkyl or a cycloalkyl group substituted with an aryl, a heteroaryl, an alkyl or a cycloalkyl group optionally substituted with at least one heteroatom, such as for example a halogen chosen from F, I, Cl and Br and optionally bearing a pendant basic nitrogen functionality,
R2 is hydrogen, halogen or a linear or branched alkyl group containing from 1 to 10 carbon atoms, trifluoromethyl or alkoxy;
R3 is hydrogen, halogen or a linear or branched alkyl group containing from 1 to 10 carbon atoms, trifluoromethyl or alkoxy;
R4 is hydrogen, halogen or a linear or branched alkyl group containing from 1 to 10 carbon atoms, trifluoromethyl or alkoxy;
R5 is hydrogen, halogen or a linear or branched alkyl group containing from 1 to 10 carbon atoms, trifluoromethyl or alkoxy;
R6 is one of the following:
(i) an aryl group such as phenyl or a substituted variant thereof bearing any combination, at any one ring position, of one or more substituents such as halogen, alkyl groups containing from 1 to 10 carbon atoms, trifluoromethyl, and alkoxy;
(ii) a heteroaryl group such as a 2, 3, or 4-pyridyl group, which may additionally bear any combination of one or more substituents such as halogen, alkyl groups containing from 1 to 10 carbon atoms, trifluoromethyl and alkoxy;
(iii) a five-membered ring aromatic heterocyclic group such as for example 2-thienyl, 3-thienyl, 2-thiazolyl, 4-thiazolyl, 5-thiazolyl, which may additionally bear any combination of one or more substituents such as halogen, an alkyl group containing from 1 to 10 carbon atoms, trifluoromethyl, and alkoxy.
iv) H, a halogen selected from I, F, Cl or Br; NH2, NO2 or SO2-R, wherein R is a linear or branched alkyl group containing one or more group such as 1 to 10 carbon atoms, and optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality. - In another alternative, substituent R6, which in the formula II is connected to position 4 of the thiazole ring, may instead occupy position 5 of the thiazole ring.
- Among the preferred compounds corresponding formula II, the invention is directed to compounds in which X is a substituted alkyl, aryl or heteroaryl group bearing a pendant basic nitrogen functionality represented for example by the structures a to f shown below, wherein the wavy line corresponds to the point of attachment to core structure of formula II:
- Among group a to f, X (see formula II) is preferentially group d.
- Furthermore, among the preferred compounds of formula I or II, the invention concerns the compounds in which R2 and R3 are hydrogen. Preferentially, R4 is a methyl group and R5 is H. In addition, R6 is preferentially a 3—pyridyl group (cf. structure g below), or a 4-pyridyl group (cf. structure h below). The wavy line in structure g and h correspond to the point of attachment to the core structure of formula I or II.
- Thus, the invention contemplates:
-
- 1—A compound of formula II as depicted above, wherein X is group d and R6 is a 3-pyridyl group.
- 2—A compound of formula II as depicted above, wherein X is group d and R4 is a methyl group.
- 3—A compound of formula I or II as depicted above, wherein R1 is group d and R2 is H.
- 4—A compound of formula I or II as depicted above, wherein R1 is group d and R3 is H.
- 5—A compound of formula I or II as depicted above, wherein R1 is group d and R2 and/or R3 and/or R5 is H.
- 6—A compound of formula I or II as depicted above, wherein R6 is a 3-pyridyl group and R3 is a methyl group.
- 7—A compound of formula I or II as depicted above, wherein R6 is a 3-pyridyl group and R2 is H.
- 8—A compound of formula I or II as depicted above, wherein R2 and/or R3 and/or R is H and R4 is a methyl group.
- 9—A compound of formula I or II as depicted above wherein R2 and/or R3 and/or R5 is H, R4 is a methyl group and R6 is a 3-pyridyl group.
- Among the compounds of formula II, the invention is particularly embodied by the compounds wherein R2, R3, R5 are hydrogen, corresponding to the following formula II-1:
- wherein X is R or NRR′ and wherein R and R′ are independently chosen from H or an organic group that can be selected for example from a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group optionally substituted with a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality;
a —SO2-R group wherein R is an alkyl, cycloalkyl, aryl or heteroaryl optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; or a —CO—R or a —CO—NRR′ group, wherein R and R′ are independently chosen from H, an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with at least one heteroatom, notably selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality. - R4 is hydrogen, halogen or a linear or branched alkyl group containing from 1 to 10 carbon atoms, trifluoromethyl or alkoxy;
- R6 is one of the following:
(i) an aryl group such as phenyl or a substituted variant thereof bearing any combination, at any one ring position, of one or more substituents such as halogen, alkyl groups containing from 1 to 10 carbon atoms, trifluoromethyl, and alkoxy;
(ii) a heteroaryl group such as a 2, 3, or 4-pyridyl group, which may additionally bear any combination of one or more substituents such as halogen, alkyl groups containing from 1 to 10 carbon atoms, trifluoromethyl and alkoxy;
(iii) a five-membered ring aromatic heterocyclic group such as for example 2-thienyl, 3-thienyl, 2-thiazolyl, 4-thiazolyl, 5-thiazolyl, which may additionally bear any combination of one or more substituents such as halogen, an alkyl group containing from 1 to 10 carbon atoms, trifluoromethyl, and alkoxy.
(iv) H, a halogen selected from I, F, Cl or Br; NH2, NO2 or SO2-R, wherein R is a linear or branched alkyl group containing one or more group such as 1 to 10 carbon atoms, and optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality. - In another alternative, substituent R6, which in the formula II is connected to position 4 of the thiazole ring, may instead occupy position 5 of the thiazole ring.
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
- beige powder mp: 116-120° C.
- 1H RMN (DMSO-d6) δ=1.80-2.00 (m, 2H); 2.29 (s, 3H); 2.30-2.45 (m, 6H); 3.55-3.65 (m, 6H); 7.15-7.25 (m, 2H); 7.46-7.50 (m, 2H); 7.52 (s, 1H); 8.35 (d, J=6.2 Hz, 1H); 8.55 (dd, J=1.5 Hz, J=4.7 Hz, 2H); 9.22 (s, 1H); 9.45 (s, 1H); 9.93 (s, 1H)
- Among the compounds of formula II, the invention is particularly embodied by the compounds wherein X is a urea group, a —CO—NRR′ group, corresponding to the [3-(thiazol-2-ylamino)-phenyl]-urea family and the following formula II-2
- wherein Ra, Rb are independently chosen from H or an organic group that can be selected for example from a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group optionally substituted with a cycloalkyl, an aryl or heteroaryl group optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality;
a —SO2-R group wherein R is an alkyl, cycloalkyl, aryl or heteroaryl optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; or a —CO—R or a —CO—NRR′ group, wherein R and R′ are independently chosen from H, an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with at least one heteroatom, notably selected from I, Cl, Br and F, or bearing a pendant basic nitrogen functionality.
R4 is hydrogen, halogen or a linear or branched alkyl group containing from 1 to 10 carbon atoms, trifluoromethyl or alkoxy;
R6 is one of the following:
(i) an aryl group such as phenyl or a substituted variant thereof bearing any combination, at any one ring position, of one or more substituents such as halogen, alkyl groups containing from 1 to 10 carbon atoms, trifluoromethyl, and alkoxy;
(ii) a heteroaryl group such as a 2, 3, or 4-pyridyl group, which may additionally bear any combination of one or more substituents such as halogen, alkyl groups containing from 1 to 10 carbon atoms, trifluoromethyl and alkoxy;
(iii) a five-membered ring aromatic heterocyclic group such as for example 2-thienyl, 3-thienyl, 2-thiazolyl, 4-thiazolyl, 5-thiazolyl, which may additionally bear any combination of one or more substituents such as halogen, an alkyl group containing from 1 to 10 carbon atoms, trifluoromethyl, and alkoxy.
iv) H, a halogen selected from I, F, Cl or Br; NH2, NO2 or SO2-R, wherein R is a linear or branched alkyl group containing one or more group such as 1 to 10 carbon atoms, and optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality. -
-
-
-
-
-
-
-
-
-
-
-
-
-
-
- light brown powder mp: 203-206° C.
- 1H NMR (DMSO-d6): δ=2.24 (s, 3H); 6.98-7.00 (m, 2H); 7.10-7.23 (m, 3H); 7.40 (m, 1H); 7.48 (s, 1H); 8.25 (m, 1H); 8.37 (d, J=7.8 Hz, 1H); 8.51 (m, 3H); 9.03 (s, 1H); 9.19 (s, 1H); 9.39 (s, 1H)
-
-
- white powder mp: 210-215° C.
- 1H NMR (DMSO-d6): δ=2.24 (s, 3H); 6.79 (t, J=6.3 Hz, 1H); 6.99 (m, 1H); 7.09-7.14 (m, 2H); 7.30 (m, 1H); 7.41 (t, J=4.7 Hz, 1H); 7.48 (s, 1H); 7.56 (d, J=1.2 Hz, 1H); 8.39 (d, J=8.0 Hz, 1H); 8.49-8.52 (m, 2H); 8.71 (s, 1H); 8.87 (s, 1H); 9.18 (s, 1H); 9.38 (s, 1H)
-
- white powder mp: 238-240° C.
- 1H RMN (DMSO-d6) δ=2.29 (s, 3H); 2.31 (s, 3H); 7.05 (d, J=6.2 Hz, 1H); 7.10-1.16 (m, 3H); 7.42-7.49 (m, 3H); 7.53 (s, 1H); 8.35-8.62 (m, 5H); 9.22 (d, J=1.6 Hz, 1H); 9.43 (s, 1H)
- Among the compounds of formula II, the invention is particularly embodied by the compounds wherein X is a -substituted Aryl group, corresponding to the N-[3-(Thiazol-2-ylamino)-phenyl]-amide family and the following formula II-3
- wherein Ra, Rb, Rc, Rd, Re are independently chosen from H or an organic group that can be selected for example from a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group optionally substituted with a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; a —SO2-R group wherein R is an alkyl, cycloalkyl, aryl or heteroaryl optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; or a —CO—R or a —CO—NRR′ group, wherein R and R′ are independently chosen from H, an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with at least one heteroatom, notably selected from I, Cl, Br and F, and or bearing a pendant basic nitrogen functionality; Ra, Rb, Rc, Rd, Re may also be
- a halogen such as I, Cl, Br and F
- a NRR′ group where R and R′ are H or a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group optionally substituted with a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality;
- an OR group where R is H or a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group optionally substituted with a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; a —SO2-R′ group wherein R′ is an alkyl, cycloalkyl, aryl or heteroaryl optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality;
- a NRaCORb group where Ra and Rb are H or a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group optionally substituted with a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality;
- a NRCONRbRc group where Ra and Rb are H or a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group optionally substituted with a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality;
- a COOR, where R is a linear or branched alkyl group containing from 1 to 10 carbon atoms atoms optionally substituted with at least one heteroatom (for example a halogen) and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group substituted by an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality;
- a CONRaRb, where Ra and Rb are a hydrogen or a linear or branched alkyl group containing from 1 to 10 carbon atoms atoms optionally substituted with at least one heteroatom (for example a halogen) and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group substituted by an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality;
-
- an NHCOOR, where R is a linear or branched alkyl group containing from 1 to 10 carbon atoms atoms optionally substituted with at least one heteroatom (for example a halogen) and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group substituted by an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality;
- an OSO2R, where R is a linear or branched alkyl group containing from 1 to 10 carbon atoms atoms optionally substituted with at least one heteroatom (for example a halogen) and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group substituted by an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality;
- an NRaOSO2Rb, where Ra and Rb are a linear or branched alkyl group containing from 1 to 10 carbon atoms atoms optionally substituted with at least one heteroatom (for example a halogen) and/or bearing a pendant basic nitrogen functionality; Ra can also be a hydrogen; a cycloalkyl, an aryl or heteroaryl group optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group substituted by an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality;
- a CN group
- a trifluoromethyl group
- R4 is hydrogen, halogen or a linear or branched alkyl group containing from 1 to 10 carbon atoms, trifluoromethyl or alkoxy;
- R6 is one of the following:
(i) an aryl group such as phenyl or a substituted variant thereof bearing any combination, at any one ring position, of one or more substituents such as halogen, alkyl groups containing from 1 to 10 carbon atoms, trifluoromethyl, and alkoxy;
(ii) a heteroaryl group such as a 2, 3, or 4-pyridyl group, which may additionally bear any combination of one or more substituents such as halogen, alkyl groups containing from 1 to 10 carbon atoms, trifluoromethyl and alkoxy;
(iii) a five-membered ring aromatic heterocyclic group such as for example 2-thienyl, 3-thienyl, 2-thiazolyl, 4-thiazolyl, 5-thiazolyl, which may additionally bear any combination of one or more substituents such as halogen, an alkyl group containing from 1 to 10 carbon atoms, trifluoromethyl, and alkoxy;
iv) H, a halogen selected from I, F, Cl or Br; NH2, NO2 or SO2-R, wherein R is a linear or branched alkyl group containing one or more group such as 1 to 10 carbon atoms, and optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality. -
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
-
- brown powder mp: 230-233° C.
- 1H NMR (DMSO-d6) δ=2.29 (s, 3H); 7.15-7.18 (m, 2H); 7.22-7.32 (m, 3H); 7.48 (m, 2H); 7.67 (dd, J=1.3 Hz, J=3.7 Hz, 1H); 7.90-7.96 (m, 3H); 8.38-8.42 (m, 1H); 8.51 (m, 1H); 8.57 (d, J=1.9 Hz, 1H); 9.17 (d, J=1.7 Hz, 1H); 9.44 (s, 1H); 10.12 (s, 1H); 10.82 (s, 1H)
-
- off-white foam mp: 184-186° C.
- 1H NMR (CD3OD-d4): δ=2.23 (s, 3H); 7.12-7.14 (m, 2H); 7.20-7.23 (m, 2H); 7.30 (m, 1H); 7.43 (m, 1H); 7.50 (m, 1H); 7.66 (d, J=1.0 Hz, 1H); 8.23 (m, 1H); 8.33 (m, 1H) 8.38 (s, 1H); 8.98 (s, 1H)
-
- yellow powder mp: 254-256° C.
- 1H NMR (DMSO-d6): δ 2.34 (s, 3H); 7.28 (d, J=8.0 Hz, 1H); 7.45-7.49 (m, 2H); 7.54 (s, 1H); 7.78 (t, J=7.6 Hz, 1H); 7.89-7.91 (m, 2H); 8.10 (t, J=7.8 Hz, 2H); 8.37-8.42 (m, 2H); 8.55 (d, J=4.7 Hz, 1H); 8.73-8.77 (m, 3H); 9.24 (s, 1H); 9.52 (s, 1H); 10.43 (s, 1H)
-
- beige powder mp: 147-150° C.
- 1H NMR (DMSO-d6): δ 2.25 (s, 3H); 2.99 (s, 6H); 6.76 (d, J=8.9 Hz, 2H); 7.16 (d, J=8.3 Hz, 1H); 7.35 (d, J=2.0 Hz, 1H); 7.44-7.47 (m, 2H); 7.86-7.89 (m, 2H); 8.34-8.36 (m, 1H); 8.48-8.50 (m, 1H); 8.56-8.57 (m, 1H); 9.16 (s, 1H); 9.44 (s, 1H); 9.85 (s, 1H)
-
- brown orange powder mp: 103-106° C.
- 1H RMN (DMSO-d6) δ=2.26 (s, 3H); 2.35 (s, 3H); 7.17-7.47 (m, 7H); 8.29 (dd, J=1.6 Hz, J=7.9 Hz, 1H); 8.47 (d, J=3.5 Hz, 1H); 8.57 (s, 1H); 9.15 (d, J=2.0 Hz, 1H); 9.44 (s, 1H); 10.33 (s, 1H)
-
- brown powder mp: 145-150° C.
- 1H RMN (DMSO-d6) δ=1.32 (s, 9H); 2.04 (s, 3H); 7.18 (d, J=8.4 Hz, 1H); 7.35-7.44 (m, 2H); 7.46 (s, 1H); 7.55 (d, J=8.5 Hz, 1H); 7.90 (d, J=8.5 Hz, 1H); 8.32 (d, J=7.9 Hz, 1H); 8.47 (dd, J=1.5 Hz, J=4.7 Hz, 1H); 8.60 (d, J=2.0 Hz, 1H); 9.15 (d, J=1.7 Hz, 1H); 9.43 (s, 1H); 10.15 (s, 1H)
-
- brown powder mp: 154-155° C.
- 1H RMN (DMSO-d6) δ=1.34 (d, J=5.9 Hz, 6H); 4.72 (hept, J=5.9 Hz, 1H); 7.01 (d, J=7.0 Hz, 2H); 7.18 (d, J=8.5 Hz, 1H); 7.35-7.44 (m, 2H); 7.46 (s, 1H); 7.94 (dd, J=2.0 Hz, J=6.7 Hz, 2H); 8.32 (d, J=8.3 Hz, 1H); 8.48 (dd, J=3.3 Hz, J=4.8 Hz, 1H); 8.58 (d, J=2.0 Hz, 1H); 9.15 (d, J=1.8 Hz, 1H); 9.43 (s, 1H); 10.4 (s, 1H)
-
- brown orange powder mp: 130-132° C.
- 1H RMN (DMSO-d6) δ=2.23 (s, 3H); 6.10 (s, 2H); 7.03 (d, J=8.1 Hz, 1H); 7.15 (d, J=8.3 Hz, 1H); 7.25-7.55 (m, 6H); 8.26 (s, 1H); 8.45 (dd, J=1.5 Hz, J=4.7, 1H); 8.55 (d, J=2.0 Hz, 1H); 9.12 (d, J=1.7 Hz, 1H); 9.40 (s, 1H); 10.01 (s, 1H)
-
- beige yellow powder mp: 75-80° C.
- 1H RMN (DMSO-d6) δ=2.10-2.25 (m, 4H); 2.50-2.60 (m, 2H); 3.19 (s, 3H); 3.41-3.48 (m, 4H); 4.00-4.06 (m, 2H); 7.00-7.11 (m, 2H); 7.22-7.35 (m, 6H), 8.18 (d, J=8.0 Hz, 1H) 8.33 (d, J=0.9 Hz, 1H); 8.49 (d, J=1.7 Hz, 1H); 9.03 (s, 1H); 9.31 (s, 1H); 10.05 (s, 1H)
-
- brown powder mp: dec. 250° C.
- 1H RMN (DMSO-d6) δ=2.28 (s, 3H); 7.21 (d, J=7.9 Hz, 1H); 7.30-7.50 (m, 3H); 7.81 (d, J=4.7 Hz, 1H); 7.98 (d, J=7.5 Hz, 2H); 8.13 (d, J=7.9 Hz, 2H); 8.32 (d, J=7.7 Hz, 1H); 8.48 (d, J=4.9 Hz, 1H); 8.62-8.69 (m, 3H); 9.16 (s, 1H); 9.45 (s, 1H); 10.34 (s, 1H)
-
-
-
-
-
-
- white powder mp: 76-79° C.
- 1H RMN (DMSO-d6) δ=2.32 (s, 3H); 3.89 (s, 3H); 7.22-7.25 (m, 2H), 7.44-7.58 (m, 4H), 8.28-8.35 (m, 1H); 8.52 (dd, J=1.6 Hz, J=4.7 Hz, 1H); 8.66 (d, J=2.0 Hz, 1H); 9.20 (d, J=1.4 Hz, 1H); 9.50 (s, 1H); 10.25 (s, 1H)
-
- beige brown powder mp: 128-130° C.
- 1H RMN (DMSO-d6) δ=2.15 (s, 3H); 2.18 (s, 3H); 2.35-2.41 (m, 4H); 3.18-3.3.24 (m, 4H); 6.94 (d, J=8.9 Hz, 2H); 7.09 (d, J=8.4 Hz, 1H); 7.28-7.38 (m, 3H); 7.81 (d, J=8.9 Hz, 2H), 8.20-8.25 (m, 1H); 8.40 (dd, J=1.6 Hz, J=4.7, 1H); 8.48 (d, J=1.9 Hz, 1H); 9.07 (d, J=1.5 Hz, 1H); 9.35 (s, 1H); 9.84 (s, 1H)
-
-
-
-
-
-
-
-
-
-
- Among compounds of formula II, the invention is particularly embodied by the compounds wherein X is a -substituted-aryl group, corresponding to the 4-(4-substituted-1-ylmethyl)-N-[3-(thiazol-2-ylamino)-phenyl]-benzamide family and the following formula II-4:
- wherein X is a heteroatom, such as O or N
wherein Ra, Rb, Rd, Re, Rf, Rg, Rh are independently chosen from H or an organic group that can be selected for example from a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group optionally substituted with a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; - or a NRR′ group where R and R′ are H or a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group optionally substituted with a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality;
- or an OR group where R is H or a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group optionally substituted with a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; a —SO2-R′ group wherein R′ is an alkyl, cycloalkyl, aryl or heteroaryl optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality;
- or a NRaCORb group where Ra and Rb are H or a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group optionally substituted with a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality;
- or a NRaCONRbRc group where Ra and Rb are H or a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group optionally substituted with a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality;
- or a COOR, where R is a linear or branched alkyl group containing from 1 to 10 carbon atoms atoms optionally substituted with at least one heteroatom (for example a halogen) and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group substituted by an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality;
- or a CONRaRb, where Ra and Rb are a hydrogen or a linear or branched alkyl group containing from 1 to 10 carbon atoms atoms optionally substituted with at least one heteroatom (for example a halogen) and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group substituted by an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality;
- or an NHCOOR, where R is a linear or branched alkyl group containing from 1 to 10 carbon atoms atoms optionally substituted with at least one heteroatom (for example a halogen) and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group substituted by an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality;
- an OSO2R, where R is a linear or branched alkyl group containing from 1 to 10 carbon atoms atoms optionally substituted with at least one heteroatom (for example a halogen) and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group substituted by an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality;
- or an NRaOSO2Rb, where Ra and Rb are a linear or branched alkyl group containing from 1 to 10 carbon atoms atoms optionally substituted with at least one heteroatom (for example a halogen) and/or bearing a pendant basic nitrogen functionality; Ra can also be a hydrogen; a cycloalkyl, an aryl or heteroaryl group optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group substituted by an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality;
- or a —SO2-R group wherein R is an alkyl, cycloalkyl, aryl or heteroaryl optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; or a —CO—R or a —CO—NRR′ group, wherein R and R′ are independently chosen from H, an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with at least one heteroatom, notably selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality.
- Ra, Rb, Rd, Re can also be halogen such as Cl, F, Br, I or trifluoromethyl;
- R4 is hydrogen, halogen or a linear or branched alkyl group containing from 1 to 10 carbon atoms, trifluoromethyl or alkoxy;
R6 is one of the following:
(i) an aryl group such as phenyl or a substituted variant thereof bearing any combination, at any one ring position, of one or more substituents such as halogen, alkyl groups containing from 1 to 10 carbon atoms, trifluoromethyl, and alkoxy;
(ii) a heteroaryl group such as a 2, 3, or 4-pyridyl group, which may additionally bear any combination of one or more substituents such as halogen, alkyl groups containing from 1 to 10 carbon atoms, trifluoromethyl and alkoxy;
(iii) a five-membered ring aromatic heterocyclic group such as for example 2-thienyl, 3-thienyl, 2-thiazolyl, 4-thiazolyl, 5-thiazolyl, which may additionally bear any combination of one or more substituents such as halogen, an alkyl group containing from 1 to 10 carbon atoms, trifluoromethyl, and alkoxy;
iv) H, a halogen selected from I, F, Cl or Br; NH2, NO2 or SO2-R, wherein R is a linear or branched alkyl group containing one or more group such as 1 to 10 carbon atoms, and optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality. -
-
-
-
-
-
-
-
-
-
- yellow crystals mp: 118-120° C.
- 1H RMN (DMSO-d6) δ=2.22 (s, 3H); 2.33 (s, 3H); 2.34-2.50 (m, 8H); 3.74 (s, 2H); 7.26 (d, J=8.3 Hz, 1H); 7.41-7.49 (m, 2H); 7.53 (s, 1H); 7.99 (d, J=8.0 Hz, 1H); 8.28-8.31 (m, 2H); 8.38 (d, J=7.9 Hz, 1H); 8.53 (dd, J=1.3 Hz, J=4.7 Hz, 1H); 8.68 (d, J=1.9 Hz, 1H); 9.21 (d, J=2.0 Hz, 1H); 9.53 (s, 1H); 10.49 (s, 1H)
-
-
-
-
-
-
-
- beige powder mp: 153-155° C.
- 1H RMN (DMSO-d6) δ=1.29 (d, J=6.6 Hz, 3H); 2.15 (s, 3H); 2.26 (s, 3H); 3.15-3.25 (m, 9H); 7.18 (d, J=8.4 Hz, 1H); 7.35-7.47 (m, 5H); 7.91 (d, J=8.2 Hz, 2H); 8.31 (d, J=8.0 Hz, 1H); 8.47 (dd, J=1.6 Hz, J=4.7 Hz, 1H); 8.60 (d, J=2.0, 1H); 9.15 (d, J=0.6, 1H) 9.45 (s, 1H); 10.18 (s, 1H)
-
-
-
-
-
-
-
-
- 118: N-{3-[4-(2-Fluoro-phenyl)-thiazol-2-ylamino]-4-methyl-phenyl}-4-(4-methyl-piperazin-1-ylmethyl)-benzamides
- Among compounds of formula II, the invention is particularly embodied by the compounds wherein X is a -aryl-substituted group, corresponding to the 3-Disubstituted-amino-N-[3-(thiazol-2-ylamino)-phenyl]-benzamide family and the following formula II-5:
- wherein Ra, Rb, Rc, Re, Rf, Rg are independently chosen from H or an organic group that can be selected for example from a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group optionally substituted with a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality;
- or a NRR′ group where R and R′ are H or a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group optionally substituted with a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality;
- or an OR group where R is H or a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group optionally substituted with a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; a —SO2-R′ group wherein R′ is an alkyl, cycloalkyl, aryl or heteroaryl optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality;
- or a NRaCORb group where Ra and Rb are H or a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group optionally substituted with a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality;
- or a NRaCONRbRc group where Ra and Rb are H or a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group optionally substituted with a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality;
- or a COOR, where R is a linear or branched alkyl group containing from 1 to 10 carbon atoms atoms optionally substituted with at least one heteroatom (for example a halogen) and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group substituted by an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality;
- or a CONRaRb, where Ra and Rb are a hydrogen or a linear or branched alkyl group containing from 1 to 10 carbon atoms atoms optionally substituted with at least one heteroatom (for example a halogen) and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group substituted by an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality;
- or an NHCOOR, where R is a linear or branched alkyl group containing from 1 to 10 carbon atoms atoms optionally substituted with at least one heteroatom (for example a halogen) and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group substituted by an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality;
- an OSO2R, where R is a linear or branched alkyl group containing from 1 to 10 carbon atoms atoms optionally substituted with at least one heteroatom (for example a halogen) and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group substituted by an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality;
- or an NRaOSO2Rb, where Ra and Rb are a linear or branched alkyl group containing from 1 to 10 carbon atoms atoms optionally substituted with at least one heteroatom (for example a halogen) and/or bearing a pendant basic nitrogen functionality; Ra can also be a hydrogen; a cycloalkyl, an aryl or heteroaryl group optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group substituted by an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality;
- or a —SO2—R group wherein R is an alkyl, cycloalkyl, aryl or heteroaryl optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; or a —CO—R or a —CO—NRR′ group, wherein R and R′ are independently chosen from H, an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with at least one heteroatom, notably selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality.
- Ra, Rb, Rc, Re can also be halogen such as Cl, F, Br, I or trifluoromethyl;
- R4 is hydrogen, halogen or a linear or branched alkyl group containing from 1 to 10 carbon atoms, trifluoromethyl or alkoxy;
R6 is one of the following:
(i) an aryl group such as phenyl or a substituted variant thereof bearing any combination, at any one ring position, of one or more substituents such as halogen, alkyl groups containing from 1 to 10 carbon atoms, trifluoromethyl, and alkoxy;
(ii) a heteroaryl group such as a 2, 3, or 4-pyridyl group, which may additionally bear any combination of one or more substituents such as halogen, alkyl groups containing from 1 to 10 carbon atoms, trifluoromethyl and alkoxy;
(iii) a five-membered ring aromatic heterocyclic group such as for example 2-thienyl, 3-thienyl, 2-thiazolyl, 4-thiazolyl, 5-thiazolyl, which may additionally bear any combination of one or more substituents such as halogen, an alkyl group containing from 1 to 10 carbon atoms, trifluoromethyl, and alkoxy;
iv) H, a halogen selected from I, F, Cl or Br; NH2, NO2 or SO2-R, wherein R is a linear or branched alkyl group containing one or more group such as 1 to 10 carbon atoms, and optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality. -
- beige powder mp: 197-198° C.
- 1H NMR (DMSO-d6): δ=2.32 (s, 3H); 3.03 (s, 6H); 6.97 (d, J=6.4 Hz, 1H); 7.23-7.56 (m, 7H); 8.37 (d, J=7.3 Hz, 1H); 8.53 (d, J=4.7 Hz, 1H); 8.63 (s, 1H); 9.20 (s, 1H); 9.48 (s, 1H); 10.15 (s, 1H)
-
- beige powder mp: 274-246° C.
- 1H RMN (DMSO-d6) δ=2.23 (s, 3H); 2.24-2.30 (m, 4H); 3.22-3.27 (m, 4H); 7.07-7.20 (m, 2H); 7.36-7.53 (m, 6H); 8.31 (d, J=7.5 Hz, 1H); 8.47 (d, J=3.7 Hz, 1H); 8.58 (s, 1H) 9.12 (d, J=7.8 Hz, 1H); 9.44 (s, 1H); 10.12 (s, 1H)
-
- beige powder mp: 247-248° C.
- 1H RMN (CDCl3) δ=1.50 (s, 3H); 3.15-3.18 (m, 4H); 3.79-3.82 (m, 3H); 6.85 (s, 1H) 7.00-7.30 (m, 7H); 7.41 (s, 1H); 7.75 (s, 1H); 8.08 (d, J=7.9 Hz, 1H); 8.22 (d, J=1.7 Hz, 1H); 8.46 (dd, J=1.3 Hz, J=4.7 Hz, 1H); 9.01 (d, J=1.6 Hz, 1H)
- Among the compounds of formula II, the invention is particularly embodied by the compounds wherein X is a —OR group, corresponding to the family [3-(Thiazol-2-ylamino)-phenyl]-carbamate and the following formula II-6
- wherein R is independently chosen from an organic group that can be selected for example from a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom and/or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F and/or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group optionally substituted with a cycloalkyl, an aryl or heteroaryl group optionally substituted with a heteroatom, notably a halogen selected from I, Cl, Br and F and/or bearing a pendant basic nitrogen functionality;
R4 is hydrogen, halogen or a linear or branched alkyl group containing from 1 to 10 carbon atoms, trifluoromethyl or alkoxy;
R6 is one of the following:
(i) an aryl group such as phenyl or a substituted variant thereof bearing any combination, at any one ring position, of one or more substituents such as halogen, alkyl groups containing from 1 to 10 carbon atoms, trifluoromethyl, and alkoxy;
(ii) a heteroaryl group such as a 2, 3, or 4-pyridyl group, which may additionally bear any combination of one or more substituents such as halogen, alkyl groups containing from 1 to 10 carbon atoms, trifluoromethyl and alkoxy;
(iii) a five-membered ring aromatic heterocyclic group such as for example 2-thienyl, 3-thienyl, 2-thiazolyl, 4-thiazolyl, 5-thiazolyl, which may additionally bear any combination of one or more substituents such as halogen, an alkyl group containing from 1 to 10 carbon atoms, trifluoromethyl, and alkoxys;
iv) H, a halogen selected from I, F, Cl or Br; NH2, NO2 or SO2-R, wherein R is a linear or branched alkyl group containing one or more group such as 1 to 10 carbon atoms, and optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality. -
-
- In a second embodiment, the invention is directed to a process for manufacturing a compound of formula I depicted above. This entails the condensation of a substrate of
general formula 10 with a thiourea of the type 11. - Substituent “L” in
formula 10 is a nucleofugal leaving group in nucleophilic substitution reactions (for example, L can be selected from chloro, bromo, iodo, toluenesulfonyloxy, methanesulfonyloxy, trifluoromethanesulfonyloxy, etc., with L being preferentially a bromo group). - Group R1 in formula 11a corresponds to group R1 as described in formula I.
- Group “PG” in formula 11c is a suitable protecting group of a type commonly utilized by the person skilled in the art.
- The reaction of 10 with 1 a-d leads to a thiozole-type product of formula 12a-d.
- Formula 12a is the same as formula I. Therefore, R1 in 12a corresponds to R1 in formula I.
- Formula 12b describes a precursor to compounds of formula I which lack substituent R1. Therefore, in a second phase of the synthesis, substituent R1 is connected to the free amine group in 12b, leading to the complete structure embodied by formula I:
-
12b+“R1”→I - The introduction of R1, the nature of which is as described on page 3 for the general formula I, is achieved by the use of standard reactions that are well known to the person skilled in the art, such as alkylation, acylation, sulfonylation, formation of ureas, etc.
- Formula 12c describes an N-protected variant of compound 12b. Group “PG” in formula 12c represents a protecting group of the type commonly utilized by the person skilled in the art. Therefore, in a second phase of the synthesis, group PG is cleaved to transform compound 12c into compound 12b. Compound 12b is subsequently advanced to structures of formula I as detailed above.
- Formula 12d describes a nitro analogue of compound 12b. In a second phase of the synthesis, the nitro group of compound 12d is reduced by any of the several methods utilized by the person skilled in the art to produce the corresponding amino group, namely compound 12b. Compound 12b thus obtained is subsequently advanced to structures of formula I as detailed above.
- General: All chemicals used were commercial reagent grade products. Dimethylformamide (DMF), methanol (MeOH) were of anhydrous commercial grade and were used without further purification. Dichloromethane and tetrahydrofuran (THF) were freshly distilled under a stream of argon before use. The progress of the reactions was monitored by thin layer chromatography using precoated silica gel 60F 254, Fluka TLC plates, which were visualized under UV light. Multiplicities in 1H NMR spectra are indicated as singlet (s), broad singlet (br s), doublet (d), triplet (t), quadruplet (q), and multiplet (m) and the NMR spectrum were realized on a 300 MHz Bruker spectrometer.
-
- Dibromine (17.2 g, 108 mmol) was added dropwise to a cold (0° C.) solution of 3-acetyl-pyridine (12 g, 99 mmol) in acetic acid containing 33% of HBr (165 mL) under vigorous stirring. The vigorously stirred mixture was warmed to 40° C. for 2 h and then to 75° C. After 2 h at 75° C., the mixture was cooled and diluted with ether (400 mL) to precipitate the product. which was recovered by filtration and washed with ether and acetone to give white crystals (100%). This material may be recrystallised from methanol and ether.
- IR (neat): 3108, 2047, 2982, 2559, 1709, 1603, 1221, 1035, 798 cm−1—1H NMR (DMSO-d6) δ=5.09 (s, 2H, CH2Br); 7.88 (m, 1H, pyridyl-H); 8.63 (m, 1H, pyridyl-H); 8.96 (m, 1H, pyridyl-H); 9.29 (m, 1H, pyridyl-H).
-
- To methyl-4-formyl benzoate (4.92 g, 30 mmol) and N-methyl-piperazine (3.6 mL, 32 mmol) in acetonitrile (100 mL) was added dropwise 2.5 mL of trifluoroacetic acid. The reaction mixture was stirred at room temperature for 1 h. After slow addition of sodium cyanoborohydride (2 g, 32 mmol), the solution was left stirring overnight at room temperature. Water (10 mL) was then added to the mixture, which was further acidified with 1N HCl to pH=6-7. The acetonitrile was removed under reduced pressure and the residual aqueous solution was extracted with diethyl ether (4×30 mL). These extracts were discarded. The aqueous phase was then basified (pH>12) by addition of 2.5N aqueous sodium hydroxyde solution. The crude product was extracted with ethyl acetate (4×30 mL). The combined organic layers were dried over MgSO4 and concentrated under reduced pressure to afford a slightly yellow oil which became colorless after purification by Kugelrohr distillation (190° C.) in 68% yield.
- IR (neat): 3322, 2944, 2802, 1721, 1612, 1457, 1281, 1122, 1012—1H NMR (CDCl3) δ=2.27 (s, 3H, NCH3); 2.44 (m, 8H, 2×NCH2CH2N); 3.53 (s, 2H, ArCH2N); 3.88 (s, 3H, OCH3); 7.40 (d, 2H, J=8.3 Hz, 2×ArH); 7.91 (d, 2H, J=8.3 Hz, 2×ArH)—13C NMR (CDCl3) δ=45.8 (NCH3); 51.8 (OCH3); 52.9 (2×CH2N); 54.9 (2×CH2N); 62.4 (ArCH2N); 128.7 (2×ArC); 129.3 (2×ArC); 143.7 (ArC); 166.7 (ArCO2CH3)— MS CI (m/z) (%): 249 (M+1, 100%).
-
- A solution of di-tert-butyldicarbonate (70 g, 320 mmol) in methanol (200 mL) was added over 2 h to a cold (−10° C.) solution of 2,4-diaminotoluene (30 g, 245 mmol) and triethylamine (30 mL) in methanol (15 mL). The reaction was followed by thin layer chromatography (hexane/ethyl acetate, 3: 1) and stopped after 4 h by adding 50 mL of water. The mixture was concentrated in vacuo and the residue was dissolved in 500 mL of ethyl acetate. This organic phase was washed with water (1×150 mL) and brine (2×150 mL), dried over MgSO4, and concentrated under reduced pressure. The resulting light brown solid was washed with small amounts of diethyl ether to give off-white crystals of 2-methyl-5-tert-butoxycarbonylamino-aniline in 67% yield.
- IR (neat): 3359; 3246; 2970; 1719; 1609; 1557; 1173; 1050 cm−1—1H NMR (CDCl3): δ=1.50 (s, 9H, tBu); 2.10 (s, 3H, ArCH3); 3.61 (br s, 2H, NH2); 6.36 (br s, 1H, NH); 6.51 (dd, 1H, J=7.9 Hz, 2.3 Hz, ArH); 6.92 (d, 1H, J=7.9 Hz, ArH); 6.95 (s, 1H, ArH)—13C NMR (CDCl3) δ=16.6 (ArCH3); 28.3 (C(CH3)3); 80.0 (C(CH3)3); 105.2 (ArC); 108.6 (ArC); 116.9 (ArC); 130.4 (ArC-CH3); 137.2 (ArC—NH); 145.0 (ArC—NH2); 152.8 (COOtBu)
- MS ESI (m/z) (%): 223 (M+1), 167 (55, 100%).
-
- Benzoyl chloride (5.64 g, 80 mmol) was added dropwise to a well-stirred solution of ammonium thiocyanate (3.54 g, 88 mmol) in acetone (50 mL). The mixture was refluxed for 15 min, then, the hydrobromide salt of 2-methyl-5-tert-butoxycarbonylamino-aniline (8.4 g, 80 mmol) was added slowly portionwise. After 1 h, the reaction mixture was poured into ice-water (350 mL) and the bright yellow precipitate was isolated by filtration. This crude solid was then refluxed for 45 min in 70 mL of 2.5 N sodium hydroxide solution. The mixture was cooled down and basified with ammonium hydroxide. The precipitate of crude thiourea was recovered by filtration and dissolved in 150 mL of ethyl acetate. The organic phase was washed with brine, dried over Na2SO4, and concentrated under reduced pressure. The residue was purified by column chromatography (hexane/ethyl acetate, 1:1) to afford 63% of N-(2-methyl-5-tert-butoxycarbonylamino)phenyl-thiourea as a white solid.
- IR (neat): 3437, 3292, 3175, 2983, 1724, 1616, 1522, 1161, 1053 cm−1—1H NMR (DMSO-d6) δ=1.46 (s, 9H, tBu); 2.10 (s, 3H, ArCH3); 3.60 (br s, 2H, NH2); 7.10 (d, 1H, J=8.29 Hz, ArH); 7.25 (d, 1H, J=2.23 Hz, ArH); 7.28 (d, 1H, J=2.63 Hz, ArH); 9.20 (s, 1H, ArNH); 9.31 (s, 1H, ArNH)—13C NMR (DMSO-d6) δ=25.1 (ArCH3); 28.1 (C(CH3)3); 78.9 (C(CH3)3); 116.6 (ArC); 117.5 (ArC); 128.0 (ArC); 130.4 (ArC-CH3); 136.5 (ArC—NH); 137.9 (ArC—NH); 152.7 (COOtBu); 181.4 (C═S)—MS CI (m/z): 282 (M+1, 100%); 248 (33); 226 (55); 182 (99); 148 (133); 93 (188).
-
- A mixture of 3-bromoacetyl-pyridine, HBr salt (0.81 g, 2.85 mmol), N-(2-methyl-5-tert-butoxycarbonylamino)phenyl-thiourea (0.8 g, 2.85 mmol) and KHCO3 (˜0.4 g) in ethanol (40 mL) was heated at 75° C. for 20 h. The mixture was cooled, filtered (removal of KHCO3) and evaporated under reduced pressure. The residue was dissolved in CHCl3 (40 mL) and washed with saturated aqueous sodium hydrogen carbonate solution and with water. The organic layer was dried over Na2SO4 and concentrated. Column chromatographic purification of the residue (hexane/ethyl acetate, 1:1) gave the desired thiazole in 70% yield as an orange solid
- IR (neat): 3380, 2985, 2942, 1748, 1447, 1374, 1239, 1047, 938—1H NMR (CDCl3) δ=1.53 (s, 9H, tBu); 2.28 (s, 3H, ArCH3); 6.65 (s, 1H, thiazole-H); 6.89 (s, 1H); 6.99 (dd, 1H, J=8.3 Hz, 2.3 Hz); 7.12 (d, 2H, J=8.3 Hz); 7.35 (dd, 1H, J=2.6 Hz, 4.9 Hz); 8.03 (s, 1H); 8.19 (dt, 1H, J=1.9 Hz, 7.9 Hz); 8.54 (br s, 1H, NH); 9.09 (s, 1H, NH)—13C NMR (CDCl3) δ=18.02 (ArCH3); 29.2 (C(CH3)3); 81.3 (C(CH3)3); 104.2 (thiazole-C); 111.6; 115.2; 123.9; 124.3; 131.4; 132.1; 134.4; 139.5; 148.2; 149.1; 149.3; 153.6; 167.3 (C═O)— MS CI (m/z) (%): 383 (M+1, 100%); 339 (43); 327 (55); 309 (73); 283 (99); 71 (311).
-
- 2-(2-methyl-5-tert-butoxycarbonylamino)phenyl-4-(3-pyridyl)-thiazole (0.40 g, 1.2 mmol) was dissolved in 10 mL of 20% TFA/CH2Cl2. The solution was stirred at room temperature for 2 h, then it was evaporated under reduced pressure. The residue was dissolved in ethyl acetate. The organic layer was washed with aqueous 1N sodium hydroxide solution, dried over MgSO4, and concentrated to afford 2-(2-methyl-5-amino)phenyl-4-(3-pyridyl)-thiazole as a yellow-orange solid in 95% yield. This crude product was used directly in the next step.
- A 2M solution of trimethyl aluminium in toluene (2.75 mL) was added dropwise to a cold (0° C.) solution of 2-(2-methyl-5-amino)phenyl-4-(3-pyridyl)-thiazole (0.42 g, 1.5 mmol) in anhydrous dichloromethane (10 mL) under argon atmosphere. The mixture was warmed to room temperature and stirred at room temperature for 30 min. A solution of methyl-4-(1-N-methyl-piperazino)-methyl benzoate (0.45 g, 1.8 mmol) in anhydrous dichloromethane (1 mL) and added slowly, and the resulting mixture was heated at reflux for 5 h. The mixture was cooled to 0° C. and quenched by dropwise addition of a 4N aqueous sodium hydroxide solution (3 mL). The mixture was extracted with dichloromethane (3×20 mL). The combined organic layers were washed with brine (3×20 mL) and dried over anhydrous MgSO4. (2-(2-methyl-5-amino)phenyl-4-(3-pyridyl)-thiazole) is obtained in 72% after purification by column chromatography (dichloromethane/methanol, 3:1)
- IR (neat): 3318, 2926, 1647, 1610, 1535, 1492, 1282, 1207, 1160, 1011, 843—1H NMR (CDCl3) δ=2.31 (br s, 6H, ArCH3+NCH3); 2.50 (br s, 8H, 2×NCH2CH2N); 3.56 (s, 2H, ArCH2N); 6.89 (s, 1H, thiazole H); 7.21-7.38 (m, 4H); 7.45 (m, 2H); 7.85 (d, 2H, J=8.3 Hz); 8.03 (s, 1H); 8.13 (s, 1H); 8.27 (s, 1H); 8.52 (br s, 1H); 9.09 (s, 1H, NH)—13C NMR (CDCl3) δ=17.8 (ArCH3); 46.2 (NCH3); 53.3 (NCH2); 55.3 (NCH2); 62.8 (ArCH2N); 99.9 (thiazole-C); 112.5; 123.9; 125.2; 127.5; 129.6; 131.6; 133.7; 134.0; 137.6; 139.3; 142.9; 148.8; 149.1; 166.2 (C═O); 166.7 (thiazole C—NH)— MS CT (m/z) (%): 499 (M+H, 100%); 455 (43); 430 (68); 401 (97); 374 (124); 309 (189); 283 (215); 235 (263); 121 (377); 99 (399).
- In a third embodiment, the invention relates to a pharmaceutical composition comprising a compound as depicted above.
- Such medicament can take the form of a pharmaceutical composition adapted for oral administration, which can be formulated using pharmaceutically acceptable carriers well known in the art in suitable dosages. Such carriers enable the pharmaceutical compositions to be formulated as tablets, pills, dragees, capsules, liquids, gels, syrups, slurries, suspensions, and the like, for ingestion by the patient. In addition to the active ingredients, these pharmaceutical compositions may contain suitable pharmaceutically-acceptable carriers comprising excipients and auxiliaries which facilitate processing of the active compounds into preparations which can be used pharmaceutically. Further details on techniques for formulation and administration may be found in the latest edition of Remington's Pharmaceutical Sciences (Maack Publishing Co., Easton, Pa.).
- The composition of the invention can also take the form of a pharmaceutical or cosmetic composition for topical administration.
- Such compositions may be presented in the form of a gel, paste, ointment, cream, lotion, liquid suspension aqueous, aqueous-alcoholic or, oily solutions, or dispersions of the lotion or serum type, or anhydrous or lipophilic gels, or emulsions of liquid or semi-solid consistency of the milk type, obtained by dispersing a fatty phase in an aqueous phase or vice versa, or of suspensions or emulsions of soft, semi-solid consistency of the cream or gel type, or alternatively of microemulsions, of microcapsules, of microparticles or of vesicular dispersions to the ionic and/or nonionic type. These compositions are prepared according to standard methods.
- The composition according to the invention comprises any ingredient commonly used in dermatology and cosmetic. It may comprise at least one ingredient selected from hydrophilic or lipophilic gelling agents, hydrophilic or lipophilic active agents, preservatives, emollients, viscosity enhancing polymers, humectants, surfactants, preservatives, antioxidants, solvents, and fillers, antioxidants, solvents, perfumes, fillers, screening agents, bactericides, odor absorbers and coloring matter.
- As oils which can be used in the invention, mineral oils (liquid paraffin), vegetable oils (liquid fraction of shea butter, sunflower oil), animal oils, synthetic oils, silicone oils (cyclomethicone) and fluorinated oils may be mentioned. Fatty alcohols, fatty acids (stearic acid) and waxes (paraffin, carnauba, beeswax) may also be used as fatty substances.
- As emulsifiers which can be used in the invention, glycerol stearate, polysorbate 60 and the PEG-6/PEG-32/glycol stearate mixture are contemplated.
- As hydrophilic gelling agents, carboxyvinyl polymers (carbomer), acrylic copolymers such as acrylate/alkylacrylate copolymers, polyacrylamides, polysaccharides such as hydroxypropylcellulose, clays and natural gums may be mentioned, and as lipophilic gelling agents, modified clays such as bentones, metal salts of fatty acids such as aluminum stearates and hydrophobic silica, or alternatively ethylcellulose and polyethylene may be mentioned.
- As hydrophilic active agents, proteins or protein hydrolysates, amino acids, polyols, urea, allantoin, sugars and sugar derivatives, vitamins, starch and plant extracts, in particular those of Aloe vera may be used.
- As lipophilic active, agents, retinol (vitamin A) and its derivatives, tocopherol (vitamin E) and its derivatives, essential fatty acids, ceramides and essential oils may be used. These agents add extra moisturizing or skin softening features when utilized.
- In addition, a surfactant can be included in the composition so as to provide deeper penetration of the compound capable of depleting mast cells, such as a tyrosine kinase inhibitor, preferably a c-kit inhibitor.
- Among the contemplated ingredients, the invention embraces penetration enhancing agents selected for example from the group consisting of mineral oil, water, ethanol, triacetin, glycerin and propylene glycol; cohesion agents selected for example from the group consisting of polyisobutylene, polyvinyl acetate and polyvinyl alcohol, and thickening agents.
- Chemical methods of enhancing topical absorption of drugs are well known in the art. For example, compounds with penetration enhancing properties include sodium lauryl sulfate (Dugard, P. H. and Sheuplein, R. J., “Effects of Ionic Surfactants on the Permeability of Human Epidermis: An Electrometric Study,” J. Ivest. Dermatol., V. 60, pp. 263-69, 1973), lauryl amine oxide (Johnson et. al., U.S. Pat. No. 4,411,893), azone (Rajadhyaksha, U.S. Pat. Nos. 4,405,616 and 3,989,816) and decylmethyl sulfoxide (Sekura, D. L. and Scala, J., “The Percutaneous Absorption of Alkylmethyl Sulfides,” Pharmacology of the Skin, Advances In Biology of Skin, (Appleton-Century Craft) V. 12, pp. 257-69, 1972). It has been observed that increasing the polarity of the head group in amphoteric molecules increases their penetration-enhancing properties but at the expense of increasing their skin irritating properties (Cooper, E. R. and Berner, B., “Interaction of Surfactants with Epidermal Tissues: Physiochemical Aspects,” Surfactant Science Series, V. 16, Reiger, M. M. ed. (Marcel Dekker, Inc.) pp. 195-210, 1987).
- A second class of chemical enhancers are generally referred to as co-solvents. These materials are absorbed topically relatively easily, and, by a variety of mechanisms, achieve permeation enhancement for some drugs. Ethanol (Gale et. al., U.S. Pat. No. 4,615,699 and Campbell et. al., U.S. Pat. Nos. 4,460,372 and 4,379,454), dimethyl sulfoxide (U.S. Pat. Nos. 3,740,420 and 3,743,727, and U.S. Pat. No. 4,575,515), and glycerine derivatives (U.S. Pat. No. 4,322,433) are a few examples of compounds which have shown an ability to enhance the absorption of various compounds.
- The pharmaceutical compositions of the invention can also be intended for administration with aerosolized formulation to target areas of a patient's respiratory tract.
- Devices and methodologies for delivering aerosolized bursts of a formulation of a drug is disclosed in U.S. Pat. No. 5,906,202. Formulations are preferably solutions, e.g. aqueous solutions, ethanoic solutions, aqueous/ethanoic solutions, saline solutions, colloidal suspensions and microcrystalline suspensions. For example aerosolized particles comprise the active ingredient mentioned above and a carrier, (e.g., a pharmaceutically active respiratory drug and carrier) which are formed upon forcing the formulation through a nozzle which nozzle is preferably in the form of a flexible porous membrane. The particles have a size which is sufficiently small such that when the particles are formed they remain suspended in the air for a sufficient amount of time such that the patient can inhale the particles into the patient's lungs.
- The invention encompasses the systems described in U.S. Pat. No. 5,556,611:
- liquid gas systems (a liquefied gas is used as propellent gas (e.g. low-boiling FCHC or propane, butane) in a pressure container,
- suspension aerosol (the active substance particles are suspended in solid form in the liquid propellent phase),
- pressurized gas system (a compressed gas such as nitrogen, carbon dioxide, dinitrogen monoxide, air is used.
- Thus, according to the invention the pharmaceutical preparation is made in that the active substance is dissolved or dispersed in a suitable nontoxic medium and said solution or dispersion atomized to an aerosol, i.e. distributed extremely finely in a carrier gas. This is technically possible for example in the form of aerosol propellent gas packs, pump aerosols or other devices known per se for liquid misting and solid atomizing which in particular permit an exact individual dosage.
- Therefore, the invention is also directed to aerosol devices comprising the compound as defined above and such a formulation, preferably with metered dose valves.
- The pharmaceutical compositions of the invention can also be intended for intranasal administration.
- In this regard, pharmaceutically acceptable carriers for administering the compound to the nasal mucosal surfaces will be readily appreciated by the ordinary artisan. These carriers are described in the Remington's Pharmaceutical Sciences” 16th edition, 1980, Ed. By Arthur Osol, the disclosure of which is incorporated herein by reference.
- The selection of appropriate carriers depends upon the particular type of administration that is contemplated. For administration via the upper respiratory tract, the composition can be formulated into a solution, e.g., water or isotonic saline, buffered or unbuffered, or as a suspension, for intranasal administration as drops or as a spray. Preferably, such solutions or suspensions are isotonic relative to nasal secretions and of about the same pH, ranging e.g., from about pH 4.0 to about pH 7.4 or, from pH 6.0 to pH 7.0. Buffers should be physiologically compatible and include, simply by way of example, phosphate buffers. For example, a representative nasal decongestant is described as being buffered to a pH of about 6.2 (Remington's, Id. at page 1445). Of course, the ordinary artisan can readily determine a suitable saline content and pH for an innocuous aqueous carrier for nasal and/or upper respiratory administration.
- Common intranasal carriers include nasal gels, creams, pastes or ointments with a viscosity of, e.g., from about 10 to about 3000 cps, or from about 2500 to 6500 cps, or greater, may also be used to provide a more sustained contact with the nasal mucosal surfaces. Such carrier viscous formulations may be based upon, simply by way of example, alkylcelluloses and/or other biocompatible carriers of high viscosity well known to the art (see e.g., Remington's, cited supra. A preferred alkylcellulose is, e.g., methylcellulose in a concentration ranging from about 5 to about 1000 or more mg per 100 ml of carrier. A more preferred concentration of methyl cellulose is, simply by way of example, from about 25 to about mg per 100 ml of carrier.
- Other ingredients, such as art known preservatives, colorants, lubricating or viscous mineral or vegetable oils, perfumes, natural or synthetic plant extracts such as aromatic oils, and humectants and viscosity enhancers such as, e.g., glycerol, can also be included to provide additional viscosity, moisture retention and a pleasant texture and odor for the formulation. For nasal administration of solutions or suspensions according to the invention, various devices are available in the art for the generation of drops, droplets and sprays.
- A premeasured unit dosage dispenser including a dropper or spray device containing a solution or suspension for delivery as drops or as a spray is prepared containing one or more doses of the drug to be administered and is another object of the invention. The invention also includes a kit containing one or more unit dehydrated doses of the compound, together with any required salts and/or buffer agents, preservatives, colorants and the like, ready for preparation of a solution or suspension by the addition of a suitable amount of water.
- Another aspect of the invention is directed to the use of said compound to manufacture a medicament. In other words, the invention embraces a method for treating a disease related to unregulated c-kit transduction comprising administering an effective amount of a compound as defined above to a mammal in need of such treatment.
- More particularly, the invention is aimed at a method for treating a disease selected from autoimmune diseases, allergic diseases, bone loss, cancers such as leukemia and GIST, tumor angiogenesis, inflammatory diseases, inflammatory bowel diseases (IBD), interstitial cystitis, mastocytosis, infections diseases, metabolic disorders, fibrosis, diabetes and CNS disorders comprising administering an effective amount a compound depicted above to a mammal in need of such treatment.
- The above described compounds are useful for manufacturing a medicament for the treatment of diseases related to unregulated c-kit transduction, including, but not limited to:
-
- neoplastic diseases such as mastocytosis, canine mastocytoma, human gastrointestinal stromal tumor (“GIST”), small cell lung cancer, non-small cell lung cancer, acute myelocytic leukemia, acute lymphocytic leukemia, myelodysplastic syndrome, chronic myelogenous leukemia, colorectal carcinomas, gastric carcinomas, gastrointestinal stromal tumors, testicular cancers, glioblastomas, solid tumors and astrocytomas.
- tumor angiogenesis.
- metabolic diseases such as diabetes mellitus and its chronic complications; obesity; diabete type II; hyperlipidemias and dyslipidemias; atherosclerosis; hypertension; and cardiovascular disease.
- allergic diseases such as asthma, allergic rhinitis, allergic sinusitis, anaphylactic syndrome, urticaria, angioedema, atopic dermatitis, allergic contact dermatitis, erythema nodosum, erythema multiforme, cutaneous necrotizing venulitis and insect bite skin inflammation and blood sucking parasitic infestation.
- interstitial cystitis.
- bone loss (osteoporosis).
- inflammatory diseases such as rheumatoid arthritis, conjunctivitis, rheumatoid spondylitis, osteoarthritis, gouty arthritis and other arthritic conditions.
- autoimmune diseases such as multiple sclerosis, psoriasis, intestine inflammatory disease, ulcerative colitis, Crohn's disease, rheumatoid arthritis and polyarthritis, local and systemic scleroderma, systemic lupus erythematosus, discoid lupus erythematosus, cutaneous lupus, dermatomyositis, polymyositis, Sjogren's syndrome, nodular panarteritis, autoimmune enteropathy, as well as proliferative glomerulonephritis.
- graft-versus-host disease or graft rejection in any organ transplantation including kidney, pancreas, liver, heart, lung, and bone marrow.
- Other autoimmune diseases embraced by the invention active chronic hepatitis and chronic fatigue syndrome.
- subepidermal blistering disorders such as pemphigus.
- Vasculitis.
- melanocyte dysfunction associated diseases such as hypermelanosis resulting from melanocyte dysfunction and including lentigines, solar and senile lentigo, Dubreuilh melanosis, moles as well as malignant melanomas. In this regard, the invention embraces the use of the compounds defined above to manufacture a medicament or a cosmetic composition for whitening human skin.
- CNS disorders such as psychiatric disorders, migraine, pain, memory loss and nerve cells degeneracy. More particularly, the method according to the invention is useful for the treatment of the following disorders: Depression including dysthymic disorder, cyclothymic disorder, bipolar depression, severe or “melancholic” depression, atypical depression, refractory depression, seasonal depression, anorexia, bulimia, premenstrual syndrome, post-menopause syndrome, other syndromes such as mental slowing and loss of concentration, pessimistic worry, agitation, self-deprecation, decreased libido, pain including, acute pain, postoperative pain, chronic pain, nociceptive pain, cancer pain, neuropathic pain, psychogenic pain syndromes, anxiety disorders including anxiety associated with hyperventilation and cardiac arrhythmias, phobic disorders, obsessive-compulsive disorder, posttraumatic stress disorder, acute stress disorder, generalized anxiety disorder, psychiatric emergencies such as panic attacks, including psychosis, delusional disorders, conversion disorders, phobias, mania, delirium, dissociative episodes including dissociative amnesia, dissociative fugue and dissociative identity disorder, depersonalization, catatonia, seizures, severe psychiatric emergencies including suicidal behaviour, self-neglect, violent or aggressive behaviour, trauma, borderline personality, and acute psychosis, schizophrenia including paranoid schizophrenia, disorganized schizophrenia, catatonic schizophrenia, and undifferentiated schizophrenia,
- neurodegenerative diseases including Alzheimer's disease, Parkinson's disease, Huntington's disease, the prion diseases, Motor Neurone Disease (MND), and Amyotrophic Lateral Sclerosis (ALS).
- substance use disorders as referred herein include but are not limited to drug addiction, drug abuse, drug habituation, drug dependence, withdrawal syndrome and overdose.
- Cerebral ischemia
- Fibrosis
- Duchenne muscular dystrophy
- Regarding mastocytosis, the invention contemplates the use of the compounds as defined above for treating the different categories which can be classified as follows:
- The category I is composed by two sub-categories (IA and IB). Category IA is made by diseases in which mast cell infiltration is strictly localized to the skin. This category represents the most frequent form of the disease and includes: i) urticaria pigmentosa, the most common form of cutaneous mastocytosis, particularly encountered in children, ii) diffuse cutaneous mastocytosis, iii) solitary mastocytoma and iv) some rare subtypes like bullous, erythrodermic and teleangiectatic mastocytosis. These forms are characterized by their excellent prognosis with spontaneous remissions in children and a very indolent course in adults. Long term survival of this form of disease is generally comparable to that of the normal population and the translation into another form of mastocytosis is rare. Category IB is represented by indolent systemic disease (SM) with or without cutaneous involvement. These forms are much more usual in adults than in children. The course of the disease is often indolent, but sometimes signs of aggressive or malignant mastocytosis can occur, leading to progressive impaired organ function.
- The category II includes mastocytosis with an associated hematological disorder, such as a myeloproliferative or myelodysplastic syndrome, or acute leukemia. These malignant mastocytosis does not usually involve the skin. The progression of the disease depends generally on the type of associated hematological disorder that conditions the prognosis.
- The category III is represented by aggressive systemic mastocytosis in which massive infiltration of multiple organs by abnormal mast cells is common. In patients who pursue this kind of aggressive clinical course, peripheral blood features suggestive of a myeloproliferative disorder are more prominent. The progression of the disease can be very rapid, similar to acute leukemia, or some patients can show a longer survival time.
- Finally, the category IV of mastocytosis includes the mast cell leukemia, characterized by the presence of circulating mast cells and mast cell progenitors representing more than 10% of the white blood cells. This entity represents probably the rarest type of leukemia in humans, and has a very poor prognosis, similar to the rapidly progressing variant of malignant mastocytosis. Mast cell leukemia can occur either de novo or as the terminal phase of urticaria pigmentosa or systemic mastocytosis.
- The invention also contemplates the method as depicted for the treatment of recurrent bacterial infections, resurging infections after asymptomatic periods such as bacterial cystitis. More particularly, the invention can be practiced for treating FimH expressing bacteria infections such as Gram-negative enterobacteria including E. coli, Klebsiella pneumoniae, Serratia marcescens, Citrobactor freudii and Salmonella typhimurium.
- In this method for treating bacterial infection, separate, sequential or concomitant administration of at least one antibiotic selected bacitracin, the cephalosporins, the penicillins, the aminoglycosides, the tetracyclines, the streptomycins and the macrolide antibiotics such as erythromycin; the fluoroquinolones, actinomycin, the sulfonamides and trimethoprim, is of interest.
- In one preferred embodiment, the invention is directed to a method for treating neoplastic diseases such as mastocytosis, canine mastocytoma, human gastrointestinal stromal tumor (“GIST”), small cell lung cancer, non-small cell lung cancer, acute myelocytic leukemia, acute lymphocytic leukemia, myelodysplastic syndrome, chronic myelogenous leukemia, colorectal carcinomas, gastric carcinomas, gastrointestinal stromal tumors, testicular cancers, glioblastomas, and astrocytomas comprising administering a compound as defined herein to a human or mammal, especially dogs and cats, in need of such treatment.
- In one other preferred embodiment, the invention is directed to a method for treating allergic diseases such as asthma, allergic rhinitis, allergic sinusitis, anaphylactic syndrome, urticaria, angioedema, atopic dermatitis, allergic contact dermatitis, erythema nodosum, erythema multiforme, cutaneous necrotizing venulitis and insect bite skin inflammation and blood sucking parasitic infestation comprising administering a compound as defined herein to a human or mammal, especially dogs and cats, in need of such treatment.
- In still another preferred embodiment, the invention is directed to a method for treating inflammatory diseases such as rheumatoid arthritis, conjunctivitis, rheumatoid spondylitis, osteoarthritis, gouty arthritis and other arthritic conditions comprising administering a compound as defined herein to a human in need of such treatment.
- In still another preferred embodiment, the invention is directed to a method for treating autoimmune diseases such as multiple sclerosis, psoriasis, intestine inflammatory disease, ulcerative colitis, Crohn's disease, rheumatoid arthritis and polyarthritis, local and systemic scleroderma, systemic lupus erythematosus, discoid lupus erythematosus, cutaneous lupus, dermatomyositis, polymyositis, Sjogren's syndrome, nodular panarteritis, autoimmune enteropathy, as well as proliferative glomerulonephritis comprising administering a compound as defined herein to a human in need of such treatment.
- In still another preferred embodiment, the invention is directed to a method for treating graft-versus-host disease or graft rejection in any organ transplantation including kidney, pancreas, liver, heart, lung, and bone marrow comprising administering a compound as defined herein to a human in need of such treatment.
- Procedure
- Experiments were performed using purified intracellular domain of c-kit expressed in baculovirus. Estimation of the kinase activity was assessed by the phosphorylation of tyrosine containing target peptide estimated by established ELISA assay.
- Experimental Results on Tested Compounds
- Result in Table 1 shows the potent inhibitory action of the catalytic activity of c-kit with an IC50 <10 μM. Further experiments (not shown) indicates that at least one compound acts as perfect competitive inhibitors of ATP.
-
TABLE 1 In vitro Inhibition assay results c-kit Compounds IC50 (μM) 066; 074; 078; 084; 012; 016; 073; 021; 088; <10 μM 023; 025; 047; 048; 055; 049; 026; 087; 075; 089; 051; 082; 090; 060; 085; 052; 053; 096 - Procedures
- C-Kit WT and Mutated C-Kit (JM) Assay
- Cells were washed two times in PBS before plating at 5×104 cells per well of 96-well plates in triplicate and stimulated either with hematopoietic growth factors (HGF) or without. After 2 days of culture, 37 Bq (1.78 Tbq/mmol) of [3H] thymidine (Amersham Life Science, UK) was added for 6 hours. Cells were harvested and filtered through glass fiber filters and [3H] thymidine incorporation was measured in a scintillation counter. For proliferation assay, all drugs were prepared as 20 mM stock solutions in DMSO and conserved at −80° C. Fresh dilutions in PBS were made before each experiment. DMSO dissolved drugs were added at the beginning of the culture. Control cultures were done with corresponding DMSO dilutions. Results are represented in percentage by taking the proliferation without inhibitor as 100%.
- Cells
- Ba/F3 murine kit and human kit, Ba/F3 mkitΔ27 (juxtamembrane deletion) are derived from the murine IL-3 dependent Ba/F3 proB lymphoid cells. The FMA3 and P815 cell lines are mastocytoma cells expressing endogenous mutated forms of Kit, i.e., frame deletion in the murine juxtamembrane coding region of the receptor-codons 573 to 579. The human leukaemic MC line HMC-1 expresses mutations JM-V560G;
- Immunoprecipitation Assays and Western Blotting Analysis
- For each assay, 5.106 Ba/F3 cells and Ba/F3-derived cells with various c-kit mutations were lysed and immunoprecipitated as described (Beslu et al., 1996), excepted that cells were stimulated with 250 ng/ml of rmKL. Cell lysates were immunoprecipitated with a rabbit immunserum anti murine KIT, directed against the KIT cytoplasmic domain (Rottapel et al., 1991). Western blot was hybridized either with the 4G10 anti-phosphotyrosine antibody (UBI) or with the rabbit immunserum anti-murine KIT or with different antibodies (described in antibodies paragraph). The membrane was then incubated either with HRP-conjugated goat anti mouse IgG antibody or with HRP-conjugated goat anti rabbit IgG antibody (Immunotech), Proteins of interest were then visualized by incubation with ECL reagent (Amersham).
- Experimental Results
- The experimental results for various compounds according to the invention using above-described protocols are set forth at Table 2:
-
TABLE 2 Target IC50 (μM) Compounds c-Kit WT IC50 < 10 μM 002; 005; 006; 007; 008; 009; 010; 012; 017; 019; 020; 021; 023; 024; 025; 026; 028; 029; 030; 032; 042; 043; 045; 047; 048; 049; 050; 051; 052; 053; 054; 055; 056; 057; 059; 060; 061; 062; 063; 064; 065; 066; 067; 072; 073; 074; 075; 077; 078; 079; 080; 081; 082; 083; 084; 085; 086; 087; 088; 089; 090; 092; 093; 094; 095; 096; 097; 106; 105; 104; 103; 128; 129; 130; 131; 117; 110; 116; 124; 108; 122; 111; 113; 118; 107; c-Kit JM Δ27 IC50 < 1 μM 028; 074; 029; 009; 012; 073; 020; 042; 061; 065; 088; 025; 048; 049; 050; 089; 051; 082; 090; 083; 059; 052; 053; 066; 103; 067; 104; 078; 079; 105; 081; 084; 030; 010; 021; 043; 054; 062; 106; 023; 024; 064; 047; 055; 026; 087; 075; 085; 005; 077; 092; 060; 032; 017; 063; 093; 094; 095; 086; 093; 096; 108; 117; 122; 008; 080; 111; 118; 113; 007; 072; 019; 056; 057; 107; 097; - Procedures
- GIST
- cells: Ba/F3 cells were transfected by c-kit gene having A27 mutation (GIST model). Ba/F3 expressing the mutated c-kit gene readily proliferate in the absence of IL3 or SCF and are tumorigenic in nude mice.
- Protocol:
- Mice were irradiated at J-1 (5Gy)
Tumor cells (106) were subcutaneously grafted at Jo
Tumor size were daily measured from J14
Number of survival mice were daily estimated
In this experimental model, the tumor size at J14 is about 20 mm3
Treated mice received per os twice a day a dose of 100 mg/kg of one compound of formula II-3 during 5 days (from J26 to J30). - Rhumatoid Arthritis
- The mice were pretreated with the compound of formula II-3 (2×, 12.5 mg/kg) for two days (day −2, day −1) before induction of arthritis. Arthritis was induced by ip injection of 150-ul serums at
days -
TABLE 3 A Treated Mice C57B1/6 2x, 12.5 mg/ Kg 6 -
TABLE 3 B Controls C57B1/6 2X, 1 % PBS 6 - At the end of the experiment the hind limbs were collected. The skin of the limb was removed and the limbs were subsequently fixed in 2% Para formaldehyde.
- Experimental Results
- GIST
- Treated mice (with one compound of formula II-3) displays significant decrease of tumor size at J30 and J33 compared to control.
- When administrated per os, one tested compound of the formula II-3 displays a significant antitumor activity against tumors cells expressing c-kit A27.
- RA
- A compound of the formula II-3 has demonstrated significant activity in the in vivo mouse model of arthritis. Results are shown on
FIGS. 1 , 2, 3, 4. -
FIG. 1 : Effect of the compound in serum transfer experiments, Protocol, ip daily treatment with the compound (2×12.5 mg/kg) and on days −2 and −1, set of experiment with 4 mices (T: treated, C: control) -
FIG. 2 : Effect of the compound in serum transfer experiments, Protocol, ip daily treatment with the compound (2×12.5 mg/kg) and on days −2 and −1, set of experiment with 4 mices (T: treated, C: control) -
FIG. 3 : Effect of the compound in serum transfer experiments, Protocol, ip daily treatment with the compound (2×12.5 mg/kg) and on days −2 and −1, set of experiment with 8 mices (T: treated, C: control) -
FIG. 4 : Effect of the compound in serum transfer experiments, Protocol, ip daily treatment with the compound (2×12.5 mg/kg) and on days −2 and −1, set of experiment with 8 mices (T: treated, C: control)
Claims (28)
1. A compound of formula I:
wherein R1 is:
a) a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or or bearing a pendant basic nitrogen functionality;
b) an aryl or heteroaryl group optionally substituted by an alkyl or aryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F and/or bearing a pendant basic nitrogen functionality;
c) a —CO—NH—R, —CO—R, —CO—OR or a —CO—NRR′ group, wherein R and R′ are independently chosen from H or an aryl, heteroaryl, alkyl and cycloalkyl group optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, or bearing a pendant basic nitrogen functionality;
R2 is hydrogen, halogen or a linear or branched alkyl group containing from 1 to 10 carbon atoms, trifluoromethyl or alkoxy;
R3 is hydrogen, halogen or a linear or branched alkyl group containing from 1 to 10 carbon atoms, trifluoromethyl or alkoxy;
R4 is hydrogen, halogen or a linear or branched alkyl group containing from 1 to 10 carbon atoms, trifluoromethyl or alkoxy;
R5 is hydrogen, halogen or a linear or branched alkyl group containing from 1 to 10 carbon atoms, trifluoromethyl or alkoxy;
R6 is one of the following:
(i) an aryl group such as phenyl or a substituted variant thereof bearing any combination, at any one ring position, of one or more substituents such as halogen, alkyl groups containing from 1 to 10 carbon atoms, trifluoromethyl, and alkoxy;
(ii) a heteroaryl group such as a 2, 3, or 4-pyridyl group, which may additionally bear any combination of one or more substituents such as halogen, alkyl groups containing from 1 to 10 carbon atoms, trifluoromethyl and alkoxy;
(iii) a five-membered ring aromatic heterocyclic group such as for example 2-thienyl, 3-thienyl, 2-thiazolyl, 4-thiazolyl, 5-thiazolyl, which may additionally bear any combination of one or more substituents such as halogen, an alkyl group containing from 1 to 10 carbon atoms, trifluoromethyl, and alkoxy,
iv) H, a halogen selected from I, F, Cl or Br; NH2, NO2 or SO2-R, wherein R is a linear or branched alkyl group containing one or more group such as 1 to 10 carbon atoms, and optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality;
and R7 is one of the following:
(i) an aryl group such as phenyl or a substituted variant thereof bearing any combination, at any one ring position, of one or more substituents such as halogen, alkyl groups containing from 1 to 10 carbon atoms, trifluoromethyl, and alkoxy;
(ii) a heteroaryl group such as a 2, 3, or 4-pyridyl group, which may additionally bear any combination of one or more substituents such as halogen, alkyl groups containing from 1 to 10 carbon atoms, trifluoromethyl and alkoxy;
(iii) a five-membered ring aromatic heterocyclic group such as for example 2-thienyl, 3-thienyl, 2-thiazolyl, 4-thiazolyl, 5-thiazolyl, which may additionally bear any combination of one or more substituents such as halogen, an alkyl group containing from 1 to 10 carbon atoms, trifluoromethyl, and alkoxy.
iv) H, a halogen selected from I, F, Cl or Br; NH2, NO2 or SO2-R, wherein R is a linear or branched alkyl group containing one or more group such as 1 to 10 carbon atoms, and optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, and/or bearing a pendant basic nitrogen functionality.
2. A compound according to claim 1 selected from:
4-Diethylaminomethyl-N-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-1-benzamide,
N-[4-Methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-4-morpholin-4-ylmethyl-benzamide,
4-Dipropylaminomethyl-N-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-benzamide,
N-[4-Methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-4-piperidin-1-ylmethyl-benzamide,
3-Iodo-N-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-benzamide,
4-Hydroxymethyl-N-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-benzamide,
4-{[4-Methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenylamino]-methyl}-benzoic acid methyl ester,
3-Phenyl-propynoic acid [4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-amide,
4-Amino-N-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-benzamide,
2-Iodo-N-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-benzamide,
4-Iodo-N-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-benzamide,
4-(3-{4-[4-Methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenylcarbamoyl]-phenyl}-ureido)-benzoic acid ethyl ester,
N-[4-Methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-4-[3-(4-trifluoromethyl-phenyl)-ureido]-benzamide,
4-[3-(4-Bromo-phenyl)-ureido]-N-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-benzamide,
{4-[4-Methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenylcarbamoyl]-benzyl}-carbamic acid tert-butyl ester,
4-Hydroxy-N-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-benzamide
4-[(Diisopropylamino)-methyl]-N-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-benzamide,
N-[4-Methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-4-(3-thiophen-2-yl-ureido)-benzamide,
4-[3-(3,5-Dimethyl-isoxazol-4-yl)-ureido]-N-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-benzamide,
4-[3-(4-Methoxy-phenyl)-ureido]-N-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-benzamide,
4-[3-(4-Difluoromethoxy-phenyl)-ureido]-N-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-benzamide,
Thiophene-2-sulfonic acid 4-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenylcarbamoyl]-phenyl ester,
4-Iodo-benzenesulfonic acid 4-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenylcarbamoyl]-phenyl ester,
N-[4-Methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-4-pyrrolidin-1-ylmethyl-benzamide,
3-Methyl-N-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-benzamide,
N-[4-Methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-3-trifluoromethyl-benzamide,
4-[3-(2,4-Dimethoxy-phenyl)-ureido]-N-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-benzamide,
N-[4-Methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-4-[3-(4-trifluoromethyl-phenyl)-ureidomethyl]-benzamide,
N-[4-Methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-4-[3-(3,4,5-trimethoxy-phenyl)-ureido]-benzamide,
4-[3-(2-Iodo-phenyl)-ureido]-N-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-benzamide,
4-[3-(4-Fluoro-phenyl)-ureido]-N-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-benzamide,
2-Fluoro-benzenesulfonic acid 4-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenylcarbamoyl]-phenyl ester,
3-Fluoro-benzenesulfonic acid 4-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenylcarbamoyl]-phenyl ester,
2-(2-methyl-5-tert-butoxycarbonylamino)phenyl-4-(3-pyridyl)-thiazole,
2-(2-methyl-5-amino)phenyl-4-(3-pyridyl)-thiazole
4-(4-Methyl-piperazin-1-ylmethyl)-N-[3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-benzamide
N-[4-Methyl-3-(4-phenyl-thiazol-2-ylamino)-phenyl]-4-(4-methyl-piperazin-1-ylmethyl)-benzamide
N-[3-([2,4′]Bithiazolyl-2′-ylamino)-4-methyl-phenyl]-4-(4-methyl-piperazin-1-ylmethyl)-benzamide
4-(4-Methyl-piperazin-1-ylmethyl)-N-[4-methyl-3-(4-pyrazin-2-yl-thiazol-2-ylamino)-phenyl]-benzamide
2-[5-(3-Iodo-benzoylamino)-2-methyl-phenylamino]-thiazole-4-carboxylic acid ethyl ester
2-{2-Methyl-5-[4-(4-methyl-piperazin-1-ylmethyl)-benzoylamino]-phenylamino}-thiazole-4-carboxylic acid ethyl ester
N-[4-Chloro-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-4-(4-methyl-piperazin-1-ylmethyl)-benzamide
3-Bromo-N-{3-[4-(4-chloro-phenyl)-5-methyl-thiazol-2-ylamino]-4-methyl-phenyl}-benzamide
{3-[4-(4-Chloro-phenyl)-5-methyl-thiazol-2-ylamino]-4-methyl-phenyl}-carbamic acid isobutyl ester
2-[5-(3-Bromo-benzoylamino)-2-methyl-phenylamino]-5-(4-chloro-phenyl)-thiazole-4-carboxylic acid ethyl ester
2-[5-(3-Bromo-benzoylamino)-2-methyl-phenylamino]-5-(4-chloro-phenyl)-thiazole-4-carboxylic acid (2-dimethylamino-ethyl)-amide
N-{3-[4-(4-Methoxy-phenyl)-thiazol-2-ylamino]-4-methyl-phenyl}-4-(4-methyl-piperazin-1-ylmethyl)-benzamide
4-(4-Methyl-piperazin-1-ylmethyl)-N-{4-methyl-3-[4-(3-trifluoromethyl-phenyl)-thiazol-2-ylamino]-phenyl}-benzamide
N-{4-Methyl-3-[4-(3-nitro-phenyl)-thiazol-2-ylamino]-phenyl}-4-(4-methyl-piperazin-1-ylmethyl)-benzamide
N-{3-[4-(2,5-Dimethyl-phenyl)-thiazol-2-ylamino]-4-methyl-phenyl}-4-(4-methyl-piperazin-1-ylmethyl)-benzamide
N-{3-[4-(4-Chloro-phenyl)-thiazol-2-ylamino]-4-methyl-phenyl}-4-(4-methyl-piperazin-1-ylmethyl)-benzamide
3-Bromo-4-methyl-N-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-benzamide
4-Fluoro-N-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-benzamide
3,5-Dibromo-N-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-4-piperidin-1-ylmethyl-benzamide
N-{3-[4-(3-Fluoro-phenyl)-thiazol-2-ylamino]-4-methyl-phenyl}-4-(4-methyl-piperazin-1-ylmethyl)-benzamide
N-{3-[4-(3-Methoxy-phenyl)-thiazol-2-ylamino]-4-methyl-phenyl}-4-(4-methyl-piperazin-1-ylmethyl)-benzamide
N-{3-[4-(2-Fluoro-phenyl)-thiazol-2-ylamino]-4-methyl-phenyl}-4-(4-methyl-piperazin-1-ylmethyl)-benzamide
4-(4-Methyl-piperazin-1-ylmethyl)-N-[4-methyl-3-(4-pyridin-2-yl-thiazol-2-ylamino)-phenyl]-benzamide
4-Cyano-N-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-benzamide
4-Fluoro-N-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-benzamide
1-(2-Fluoro-phenyl)-3-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-urea
1-(2-Chloro-phenyl)-3-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-urea
1-(3-Fluoro-phenyl)-3-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-urea
1-[4-Methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-3-p-tolyl-urea
3-Bromo-N-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-benzamide
N-[4-Methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-4-(thiophene-2-sulfonylamino)-benzamide
3-Fluoro-N-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-benzamide
N-[4-Methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-4-pyridin-4-yl-benzamide
4-Dimethylamino-N-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-benzamide
2-Fluoro-5-methyl-N-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-benzamide
4-tert-Butyl-N-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-benzamide
4-Isopropoxy-N-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylmethyl)-phenyl]-benzamide
Benzo[1,3]dioxole-5-carboxylic acid [4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylmethyl)-phenyl]-amide
N-[4-Methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-3-(2-morpholin-4-yl-ethoxy)-benzamide
N-[4-Methyl-3-(4-pyridin-3-yl-thiazol-2-ylmethyl)-phenyl]-4-pyridin-4-yl-benzamide
3-Cyano-N-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-benzamide
2-Fluoro-N-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-3-trifluoromethyl-benzamide
4-Aminomethyl-N-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-benzamide
3-Methoxy-N-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylmethyl)-phenyl]-benzamide
4-(4-Methyl-piperazin-1-yl)-N-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylmethyl)-phenyl]-benzamide
Biphenyl-3-carboxylic acid [4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-amide
N-[4-Methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-isonicotinamide
2,6-Dichloro-N-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-isonicotinamide
3,5-Dibromo-4-(4-methyl-piperazin-1-ylmethyl)-N-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-benzamide
3-Fluoro-4-(4-methyl-piperazin-1-ylmethyl)-N-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-benzamide
4-(4-Methyl-piperazin-1-ylmethyl)-N-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylmethyl)-phenyl]-3-trifluoromethyl-benzamide
2,3,5,6-Tetrafluoro-4-(4-methyl-piperazin-1-ylmethyl)-N-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-benzamide
N-{3-[4-(4-Fluoro-phenyl)-thiazol-2-ylamino]-4-methyl-phenyl}-4-(4-methyl-piperazin-1-ylmethyl)-benzamide
3-Bromo-4-(4-methyl-piperazin-1-ylmethyl)-N-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-benzamide
3-Chloro-4-(4-methyl-piperazin-1-ylmethyl)-N-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-benzamide
4-(4-Methyl-piperazin-1-ylmethyl)-N-[4-methyl-3-(4-pyridin-4-yl-thiazol-2-ylamino)-phenyl]-benzamide
N-{3-[4-(4-Cyano-phenyl)-thiazol-2-ylamino]-4-methyl-phenyl}-4-(4-methyl-piperazin-1-ylmethyl)-benzamide
4-[1-(4-Methyl-piperazin-1-yl)-ethyl]-N-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylmethyl)-phenyl]-benzamide
4-(1-Methoxy-ethyl)-N-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylmethyl)-phenyl]-benzamide
N-{4-Methyl-3-[4-(5-methyl-pyridin-3-yl)-thiazol-2-ylamino]-phenyl}-4-(4-methyl-piperazin-1-ylmethyl)-benzamide
3-Iodo-4-(4-methyl-piperazin-1-ylmethyl)-N-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylmethyl)-phenyl]-benzamide
3,5-Dibromo-N-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-4-[(3-morpholin-4-yl-propylamino)-methyl]-benzamide
3-Dimethylamino-N-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-benzamide
3-(4-Methyl-piperazin-1-yl)-N-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-benzamide
N-[4-Methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-3-morpholin-4-yl-benzamide
Cyclohexanecarboxylic acid [4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylmethyl)-phenyl]-amide
5-[4-Methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenylcarbamoyl]-pentanoic acid ethyl ester
1-Methyl-cyclohexanecarboxylic acid [4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylmethyl)-phenyl]-amide
4-tert-Butyl-cyclohexanecarboxylic acid [4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-amide
N-[4-Methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-4-morpholin-4-yl-butyramide
[4-Methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-carbamic acid isobutyl ester
2-(2-methyl-5-tert-butoxycarbonylamino)phenyl-4-(3-pyridyl)-thiazole
3. A compound according to claim 1 of the following formula:
wherein R is H or an organic group that can be selected for example from a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted by an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality.
4. A compound according to claim 1 of the following formula:
wherein R is H or an organic group that can be selected for example from a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group optionally substituted with a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality;
a sulfonyl or a —SO2-R group wherein R is H, an alkyl, cycloalkyl, aryl or heteroaryl optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; or a —CO—R or a —CO—NRR′ group, wherein R and R′ are independently chosen from H, an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with at least one heteroatom, notably selected from I, Cl, Br and F, or bearing a pendant basic nitrogen functionality.
5. A compound according to claim 1 of the following formula:
wherein R is H or an organic group that can be selected for example from a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, or bearing a pendant basic nitrogen functionality; a cycloalkyl, aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; or an alkyl, cycloalkyl, aryl or heteroaryl group substituted by a alkyl, cycloalkyl, aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality;
a sulfonyl or a —SO2-R group wherein R is H or an alkyl, cycloalkyl, aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality;
or a —CO—R or a —CO—NRR′ group, wherein R and R′ are independently chosen from H or an aryl heteroaryl, alkyl and cycloalkyl group optionally substituted with at least one heteroatom or bearing a pendant basic nitrogen functionality.
6. A compound according to claim 1 of the following formula:
wherein R is H or an organic group that can be selected for example from a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F, or bearing a pendant basic nitrogen functionality;
a cycloalkyl, aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; or an alkyl, cycloalkyl, aryl or heteroaryl group substituted by a alkyl, cycloalkyl, aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality;
a sulfonyl or a —SO2-R group wherein R is H or an alkyl, cycloalkyl, aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; or a —CO—R or a —CO—NRR′ group, wherein R and R′ are independently chosen from H or an aryl heteroaryl, alkyl and cycloalkyl group optionally substituted with at least one heteroatom or bearing a pendant basic nitrogen functionality.
7. A compound according to claim 1 of the following formula:
wherein R is H or an organic group that can be selected for example from a linear or branched alkyl group containing from 1 to 10 carbon atoms optionally substituted with at least one heteroatom (for example an halogen) or bearing a pendant basic nitrogen functionality; a cycloalkyl, an aryl or heteroaryl group optionally substituted with at least one heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality; or a cycloalkyl, an aryl or heteroaryl group substituted by an alkyl, a cycloalkyl, an aryl or heteroaryl group optionally substituted with an heteroatom, notably a halogen selected from I, Cl, Br and F or bearing a pendant basic nitrogen functionality.
8. A compound according to claim 1 of formula II:
wherein X is R or NRR′ and wherein R and R′ are independently chosen from H, an aryl, an heteroaryl, an alkyl and a cycloalkyl group optionally substituted with at least one heteroatom, such as for example a halogen chosen from F, I, Cl and Br and optionally bearing a pendant basic nitrogen functionality; or an aryl, an heteroaryl, an alkyl and a cycloalkyl group substituted with an aryl, an heteroaryl, an alkyl and a cycloalkyl group optionally substituted with at least one heteroatom, such as for example a halogen chosen from F, I, Cl and Br and optionally bearing a pendant basic nitrogen functionality, R is hydrogen, halogen or a linear or branched alkyl group containing from 1 to 10 carbon atoms, trifluoromethyl or alkoxy;
R3 is hydrogen, halogen or a linear or branched alkyl group containing from 1 to 10 carbon atoms, trifluoromethyl or alkoxy;
R4 is hydrogen, halogen or a linear or branched alkyl group containing from 1 to 10 carbon atoms, trifluoromethyl or alkoxy;
R5 is hydrogen, halogen or a linear or branched alkyl group containing from 1 to 10 carbon atoms, trifluoromethyl or alkoxy;
R6 is one of the following:
(i) an aryl group such as phenyl or a substituted variant thereof bearing any combination, at any one ring position, of one or more substituents such as halogen, alkyl groups containing from 1 to 10 carbon atoms, trifluoromethyl, and alkoxy;
(ii) a heteroaryl group such as a 2, 3, or 4-pyridyl group, which may additionally bear any combination of one or more substituents such as halogen, alkyl groups containing from 1 to 10 carbon atoms, trifluoromethyl and alkoxy;
(iii) a five-membered ring aromatic heterocyclic group such as for example 2-thienyl, 3-thienyl, 2-thiazolyl, 4-thiazolyl, 5-thiazolyl, which may additionally bear any combination of one or more substituents such as halogen, an alkyl group containing from 1 to 10 carbon atoms, trifluoromethyl, and alkoxy.
9. A compound according to claim 8 selected from:
1-(4-Methoxy-phenyl)-3-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-urea,
1-(4-Bromo-phenyl)-3-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-urea,
1-[4-Methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-3-(4-trifluoromethyl-phenyl)-urea,
1-(4-Fluoro-phenyl)-3-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-urea,
1-[4-Methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-3-(3,4,5-trimethoxy-phenyl)-urea,
4-{3-[4-Methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-ureido}-benzoic acid ethyl ester,
1-[4-Methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-3-thiophen-2-yl-urea,
1-Cyclohexyl-1-(N-Cyclohexyl-formamide)-3-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-urea,
1-(2,4-Dimethoxy-phenyl)-3-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-urea,
1-(2-Iodo-phenyl)-1-(N-(2-Iodo-phenyl)-formamide)-3-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-urea,
1-(3,5-Dimethyl-isoxazol-4-yl)-3-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-urea,
1-(2-Iodo-phenyl)-3-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-urea,
1-(4-Difluoromethoxy-phenyl)-3-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-urea,
and 1-(4-Dimethylamino-phenyl)-3-[4-methyl-3-(4-pyridin-3-yl-thiazol-2-ylamino)-phenyl]-urea.
11. A compound according to claim 8 , wherein X is group d and R6 is a 3-pyridyl group.
12. A compound according to claim 8 , wherein X is group d and R4 is a methyl group.
13. A compound according to claim 8 , wherein X is group d and R2 and/or R3 and/or R5 is H.
14. A compound according to claim 1 , wherein R6 is a 3-pyridyl group and R4 is a methyl group.
15. A compound according to claim 1 , wherein R6 is a 3-pyridyl group and R2 and/or R3 and/or R5 is H.
16. A compound according to claim 1 , wherein R2 and/or R3 and/or R5 is H and R4 is a methyl group.
17. A compound according to claim 1 , wherein R2 and/or R3 and/or R5 is H, R4 is a methyl group and R6 is a 3-pyridyl group.
18. A compound according to claim 8 , which is the 2-(2-methyl-5-amino)phenyl-4-(3-pyridyl)-thiazole.
19. A pharmaceutical composition comprising a compound according to claim 1 .
20. A pharmaceutical composition according to claim 19 further comprising a pharmaceutically acceptable carrier.
21. A pharmaceutical composition according to claim 20 formulated as tablets, pills, dragees, capsules, liquids, gels, syrups, slurries, and suspensions.
22. A cosmetic composition for topical administration comprising a compound according to claim 1 .
23. Use of a compound according to claim 1 to manufacture a medicament.
24. Use of a compound according to claim 1 to manufacture a medicament for treating neoplastic diseases such as mastocytosis, canine mastocytoma, human gastrointestinal stromal tumor (“GIST”), small cell lung cancer, non-small cell lung cancer, acute myelocytic leukemia, acute lymphocytic leukemia, myelodysplastic syndrome, chronic myelogenous leukemia, colorectal carcinomas, gastric carcinomas, gastrointestinal stromal tumors, testicular cancers, glioblastomas, and astrocytomas.
25. Use of a compound according to claim 1 to manufacture a medicament for treating allergic diseases such as asthma, allergic rhinitis, allergic sinusitis, anaphylactic syndrome, urticaria, angioedema, atopic dermatitis, allergic contact dermatitis, erythema nodosum, erythema multiforme, cutaneous necrotizing venulitis and insect bite skin inflammation and blood sucking parasitic infestation.
26. Use of a compound according to claim 1 to manufacture a medicament for treating inflammatory diseases such as rheumatoid arthritis, conjunctivitis, rheumatoid spondylitis, osteoarthritis, gouty arthritis and other arthritic conditions.
27. Use of a compound according to claim 1 to manufacture a medicament for treating autoimmune diseases such as multiple sclerosis, psoriasis, intestine inflammatory disease, ulcerative colitis, Crohn's disease, rheumatoid arthritis and polyarthritis, local and systemic scleroderma, systemic lupus erythematosus, discoid lupus erythematosus, cutaneous lupus, dermatomyositis, polymyositis, Sjogren's syndrome, nodular panarteritis, autoimmune enteropathy, as well as proliferative glomerulonephritis.
28. Use of a compound according to claim 1 to manufacture a medicament for treating graft-versus-host disease or graft rejection in any organ transplantation including kidney, pancreas, liver, heart, lung, and bone marrow.
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US11/779,633 US20080255141A1 (en) | 2002-08-02 | 2007-07-18 | 2-(3-aminoaryl) amino-4-aryl-thiazoles and their use as c-kit inhibitors |
| US13/015,664 US8835435B2 (en) | 2002-08-02 | 2011-01-28 | 2-(3-aminoaryl) amino-4-aryl-thiazoles and their use as c-kit inhibitors |
| US13/016,100 US8450302B2 (en) | 2002-08-02 | 2011-01-28 | 2-(3-aminoaryl) amino-4-aryl-thiazoles and their use as c-kit inhibitors |
| US14/089,946 US8993573B2 (en) | 2002-08-02 | 2013-11-26 | 2-(3-aminoaryl) amino-4-aryl-thiazoles and their use as c-kit inhibitors |
Applications Claiming Priority (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US40006402P | 2002-08-02 | 2002-08-02 | |
| US10/523,018 US20050239852A1 (en) | 2002-08-02 | 2003-07-31 | 2-(3-aminoaryl)amino-4-aryl-thiazoles and their use as c-kit inhibitors |
| PCT/IB2003/003685 WO2004014903A1 (en) | 2002-08-02 | 2003-07-31 | 2-(3-aminoaryl)amino-4-aryl-thiazoles and their use as c-kit inhibitors |
| US10/632,101 US7423055B2 (en) | 2002-08-02 | 2003-08-01 | 2-(3-Aminoaryl)amino-4-aryl-thiazoles for the treatment of diseases |
| US11/779,633 US20080255141A1 (en) | 2002-08-02 | 2007-07-18 | 2-(3-aminoaryl) amino-4-aryl-thiazoles and their use as c-kit inhibitors |
Related Parent Applications (4)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US10/523,018 Continuation US20050239852A1 (en) | 2002-08-02 | 2003-07-31 | 2-(3-aminoaryl)amino-4-aryl-thiazoles and their use as c-kit inhibitors |
| PCT/IB2003/003685 Continuation WO2004014903A1 (en) | 2002-08-02 | 2003-07-31 | 2-(3-aminoaryl)amino-4-aryl-thiazoles and their use as c-kit inhibitors |
| US10523018 Continuation | 2003-07-31 | ||
| US13/015,664 Continuation US8835435B2 (en) | 2002-08-02 | 2011-01-28 | 2-(3-aminoaryl) amino-4-aryl-thiazoles and their use as c-kit inhibitors |
Related Child Applications (3)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US10/632,101 Continuation-In-Part US7423055B2 (en) | 2002-08-02 | 2003-08-01 | 2-(3-Aminoaryl)amino-4-aryl-thiazoles for the treatment of diseases |
| US13/016,100 Continuation-In-Part US8450302B2 (en) | 2002-08-02 | 2011-01-28 | 2-(3-aminoaryl) amino-4-aryl-thiazoles and their use as c-kit inhibitors |
| US13/015,664 Continuation US8835435B2 (en) | 2002-08-02 | 2011-01-28 | 2-(3-aminoaryl) amino-4-aryl-thiazoles and their use as c-kit inhibitors |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| US20080255141A1 true US20080255141A1 (en) | 2008-10-16 |
Family
ID=31715693
Family Applications (5)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US10/523,018 Abandoned US20050239852A1 (en) | 2002-08-02 | 2003-07-31 | 2-(3-aminoaryl)amino-4-aryl-thiazoles and their use as c-kit inhibitors |
| US10/632,101 Expired - Lifetime US7423055B2 (en) | 2002-08-02 | 2003-08-01 | 2-(3-Aminoaryl)amino-4-aryl-thiazoles for the treatment of diseases |
| US11/779,633 Abandoned US20080255141A1 (en) | 2002-08-02 | 2007-07-18 | 2-(3-aminoaryl) amino-4-aryl-thiazoles and their use as c-kit inhibitors |
| US13/015,664 Expired - Lifetime US8835435B2 (en) | 2002-08-02 | 2011-01-28 | 2-(3-aminoaryl) amino-4-aryl-thiazoles and their use as c-kit inhibitors |
| US14/089,946 Expired - Lifetime US8993573B2 (en) | 2002-08-02 | 2013-11-26 | 2-(3-aminoaryl) amino-4-aryl-thiazoles and their use as c-kit inhibitors |
Family Applications Before (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US10/523,018 Abandoned US20050239852A1 (en) | 2002-08-02 | 2003-07-31 | 2-(3-aminoaryl)amino-4-aryl-thiazoles and their use as c-kit inhibitors |
| US10/632,101 Expired - Lifetime US7423055B2 (en) | 2002-08-02 | 2003-08-01 | 2-(3-Aminoaryl)amino-4-aryl-thiazoles for the treatment of diseases |
Family Applications After (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US13/015,664 Expired - Lifetime US8835435B2 (en) | 2002-08-02 | 2011-01-28 | 2-(3-aminoaryl) amino-4-aryl-thiazoles and their use as c-kit inhibitors |
| US14/089,946 Expired - Lifetime US8993573B2 (en) | 2002-08-02 | 2013-11-26 | 2-(3-aminoaryl) amino-4-aryl-thiazoles and their use as c-kit inhibitors |
Country Status (20)
| Country | Link |
|---|---|
| US (5) | US20050239852A1 (en) |
| EP (1) | EP1525200B1 (en) |
| JP (1) | JP4726486B2 (en) |
| KR (1) | KR101036866B1 (en) |
| CN (1) | CN100491374C (en) |
| AT (1) | ATE375342T1 (en) |
| BR (1) | BRPI0313165B8 (en) |
| CA (1) | CA2494695C (en) |
| CY (1) | CY1107036T1 (en) |
| DE (1) | DE60316810T2 (en) |
| DK (1) | DK1525200T3 (en) |
| ES (1) | ES2294344T3 (en) |
| HK (1) | HK1084382A1 (en) |
| IL (1) | IL166528A0 (en) |
| MX (1) | MXPA05001277A (en) |
| NO (1) | NO330608B1 (en) |
| NZ (1) | NZ538490A (en) |
| PT (1) | PT1525200E (en) |
| WO (1) | WO2004014903A1 (en) |
| ZA (1) | ZA200501331B (en) |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20090221565A1 (en) * | 2008-02-29 | 2009-09-03 | Vetoquinol Sa | Novel 7-substituted 3-carboxy-oxadiazino-quinolone derivatives, their preparation and their application as anti-bacterials |
| US20100009980A1 (en) * | 2008-07-09 | 2010-01-14 | Vetoquinol Sa | Novel 9-substituted-5-carboxy-oxadiazino-quinolone derivatives, their preparation and their application as anti-bacterials |
| US8920785B2 (en) | 2010-03-23 | 2014-12-30 | Beiersdorf Ag | Cosmetic or dermatological preparations with a content of one or more thiazole derivates |
| US9168245B2 (en) | 2011-07-27 | 2015-10-27 | Ab Science | Selective protein kinase inhibitors |
Families Citing this family (89)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR100902171B1 (en) | 2001-03-08 | 2009-06-10 | 더 트러스티스 오브 더 유니버시티 오브 펜실베니아 | Facially amphiphilic polymers as anti-infective agents |
| JP2004537537A (en) | 2001-06-29 | 2004-12-16 | アブ サイエンス | Uses of tyrosine kinase inhibitors to treat inflammatory diseases |
| WO2003002106A2 (en) | 2001-06-29 | 2003-01-09 | Ab Science | Use of tyrosine kinase inhibitions for treating allergic diseases |
| US7727731B2 (en) * | 2001-06-29 | 2010-06-01 | Ab Science | Potent, selective and non toxic c-kit inhibitors |
| JP3836019B2 (en) * | 2001-11-21 | 2006-10-18 | 松下電器産業株式会社 | Reception device, transmission device, and transmission method |
| US8450302B2 (en) * | 2002-08-02 | 2013-05-28 | Ab Science | 2-(3-aminoaryl) amino-4-aryl-thiazoles and their use as c-kit inhibitors |
| IL166528A0 (en) * | 2002-08-02 | 2006-01-15 | Ab Science | 2-(3-aminoaryl)amino-4-aryl-thiazoles and their use as c-kit inhibitors |
| US20050282814A1 (en) * | 2002-10-03 | 2005-12-22 | Targegen, Inc. | Vasculostatic agents and methods of use thereof |
| US20080025916A1 (en) * | 2003-02-27 | 2008-01-31 | Ab Science | Tailored Treatment Suitable for Different Forms of Mastocytosis |
| CA2519023C (en) | 2003-03-17 | 2014-05-20 | The Trustees Of The University Of Pennsylvania | Facially amphiphilic polymers and oligomers and uses thereof |
| US20070191267A1 (en) * | 2003-04-28 | 2007-08-16 | Ab Science | Use of tyrosine kinase inhibitors for treating cerebral ischemia |
| WO2005016323A2 (en) * | 2003-08-15 | 2005-02-24 | Ab Science | Use of c-kit inhibitors for treating type ii diabetes |
| PT1702917T (en) * | 2003-12-25 | 2017-11-14 | Nippon Shinyaku Co Ltd | Amide derivative and medicine |
| KR20070001959A (en) | 2004-01-23 | 2007-01-04 | 더 트러스티스 오브 더 유니버시티 오브 펜실베니아 | Facially amphiphilic polyaryl and polyarylalkynyl polymers and olygomers and uses thereof |
| KR20060129413A (en) * | 2004-01-30 | 2006-12-15 | 에이비 사이언스 | 2-(3-substituted-aryl)amino-4-aryl-thiazoles as tyrosine kinase inhibitors |
| AU2005231507B2 (en) * | 2004-04-08 | 2012-03-01 | Targegen, Inc. | Benzotriazine inhibitors of kinases |
| WO2005102325A1 (en) * | 2004-04-20 | 2005-11-03 | Ab Science | Use of c-kit inhibitors for treating inflammatory muscle disorders including myositis and muscular dystrophy |
| WO2005102318A1 (en) * | 2004-04-20 | 2005-11-03 | Ab Science | Use of c-kit inhibitors for treating hiv related diseases |
| EP1742633A2 (en) * | 2004-04-23 | 2007-01-17 | AB Science | Use of c-kit inhibitors for treating fibrosis |
| WO2005102326A2 (en) * | 2004-04-23 | 2005-11-03 | Ab Science | Use of c-kit inhibitors for treating renal diseases |
| AU2005243384A1 (en) * | 2004-05-17 | 2005-11-24 | Otsuka Pharmaceutical Co., Ltd. | Thiazole compound and use thereof |
| CA2566104A1 (en) * | 2004-05-18 | 2005-12-01 | Ab Science | Use of mast cells inhibitors for treating patients exposed to chemical or biological weapons |
| WO2005115304A2 (en) * | 2004-05-24 | 2005-12-08 | Ab Science | Use of c-kit inhibitors for treating fibrodysplasia |
| WO2005115385A1 (en) * | 2004-05-24 | 2005-12-08 | Ab Science | Use of c-kit inhibitors for treating acne |
| NZ588896A (en) | 2004-08-25 | 2012-05-25 | Targegen Inc | Heterocyclic compounds and methods of use |
| EP1855704A4 (en) * | 2005-02-25 | 2009-12-02 | Univ Pennsylvania | Facially amphiphilic polymers and oligomers, compositions thereof, and use thereof in methods of treating cancer |
| EP1879575A2 (en) | 2005-05-09 | 2008-01-23 | Achillion Pharmaceuticals, Inc. | Thiazole compounds and methods of use |
| WO2007037898A2 (en) * | 2005-08-29 | 2007-04-05 | Housey Pharmaceuticals Inc | Theramutein modulators |
| JP2008543775A (en) * | 2005-06-08 | 2008-12-04 | ターゲジェン インコーポレーティッド | Methods and compositions for treating ocular disorders |
| GB0512091D0 (en) * | 2005-06-14 | 2005-07-20 | Novartis Ag | Organic compounds |
| EP1904065A2 (en) * | 2005-07-14 | 2008-04-02 | AB Science | Use of dual c-kit/fgfr3 inhibitors for treating multiple myeloma |
| US8133900B2 (en) * | 2005-11-01 | 2012-03-13 | Targegen, Inc. | Use of bi-aryl meta-pyrimidine inhibitors of kinases |
| KR100716891B1 (en) * | 2005-11-01 | 2007-05-09 | (주)아모레퍼시픽 | Pyridine thiazole carboxamide derivatives, the preparation method thereof, and the composition for skin whitening containing the same |
| MY167260A (en) * | 2005-11-01 | 2018-08-14 | Targegen Inc | Bi-aryl meta-pyrimidine inhibitors of kinases |
| US8604042B2 (en) * | 2005-11-01 | 2013-12-10 | Targegen, Inc. | Bi-aryl meta-pyrimidine inhibitors of kinases |
| WO2007056075A2 (en) * | 2005-11-02 | 2007-05-18 | Targegen, Inc. | Six membered heteroaromatic inhibitors targeting resistant kinase mutations |
| US20070135368A1 (en) * | 2005-12-09 | 2007-06-14 | Knapp Pamela E | Cell-to-cell transmission of siRNA induced gene silencing in mammalian cells |
| WO2007076460A2 (en) * | 2005-12-23 | 2007-07-05 | Kalypsys, Inc. | Substituted thiazole ureas useful as inhibitors of protein kinases |
| DE102005062990A1 (en) | 2005-12-28 | 2007-07-05 | Grünenthal GmbH | New N-thiazolyl-alkyl substituted propiolamide derivatives are inhibitors of the mGluR5 receptor useful for treatment and prevention of e.g. pain, anxiety and panic attacks |
| DE102005062986A1 (en) | 2005-12-28 | 2007-07-05 | Grünenthal GmbH | New substituted propiolic acid amides, useful for treatment and prevention of e.g. pain, anxiety and panic attacks, are inhibitors of the mGluR5 receptor |
| ES2389678T3 (en) * | 2006-04-20 | 2012-10-30 | Janssen Pharmaceutica Nv | A C-Kit kinase inhibitor for use in the treatment of gastrointestinal stromal tumors or mastocytosis |
| WO2008008234A1 (en) | 2006-07-07 | 2008-01-17 | Targegen, Inc. | 2-amino-5-substituted pyrimidine inhibitors |
| WO2008084103A1 (en) * | 2007-01-12 | 2008-07-17 | Ab Science | Combination treatment of solid cancers with antimetabolites and tyrosine kinase inhibitors |
| US9187485B2 (en) | 2007-02-02 | 2015-11-17 | Baylor College Of Medicine | Methods and compositions for the treatment of cancer and related hyperproliferative disorders |
| MX2009008665A (en) * | 2007-02-13 | 2009-08-21 | Ab Science | Process for the synthesis of 2-aminothiazole compounds as kinase inhibitors. |
| EP1964848A1 (en) * | 2007-03-01 | 2008-09-03 | Bayer Schering Pharma Aktiengesellschaft | Radiofluorination methods |
| JP2010528019A (en) | 2007-05-22 | 2010-08-19 | アキリオン ファーマシューティカルズ,インコーポレーテッド | Heteroaryl substituted thiazole |
| KR100885692B1 (en) * | 2007-06-12 | 2009-02-26 | 한국화학연구원 | 2-3-chloro-4-methoxyanilino-4-arylthiazole derivatives or pharmaceutically acceptable salts thereof, preparation method thereof, and phrmaceutical composition for the prevention and treatment of cancers containing the same as an active ingredient |
| JP5592265B2 (en) * | 2007-11-01 | 2014-09-17 | アキュセラ インコーポレイテッド | Amine derivative compounds for the treatment of eye diseases and disorders |
| US8106209B2 (en) | 2008-06-06 | 2012-01-31 | Achillion Pharmaceuticals, Inc. | Substituted aminothiazole prodrugs of compounds with anti-HCV activity |
| EP2417972B1 (en) * | 2009-04-06 | 2017-06-14 | Neurotech Pharmaceuticals Co., Ltd. | Pharmaceutical composition for treating or preventing burn injuries |
| WO2011092273A1 (en) * | 2010-01-28 | 2011-08-04 | Ab Science | Treatment of gist with masitinib |
| TW201130830A (en) | 2010-02-01 | 2011-09-16 | Ab Science | Combined treatment of pancreatic cancer with gemcitabine and masitinib |
| AR080380A1 (en) * | 2010-03-09 | 2012-04-04 | Ab Science | TREATMENT OF DEMENCE TYPE ALZHEIMER WITH MASITINIB |
| AR080933A1 (en) | 2010-04-20 | 2012-05-16 | Ab Science | TREATMENT OF MULTIPLE SCLEROSIS WITH MASITINIB |
| AR081772A1 (en) | 2010-06-02 | 2012-10-17 | Ab Science | TREATMENT OF REUMATOID ARTHRITIS WITH MASITINIB |
| WO2012059526A1 (en) | 2010-11-05 | 2012-05-10 | Ab Science | Treatment of mastocytosis with masitinib |
| US10045978B2 (en) | 2010-11-05 | 2018-08-14 | Ab Science | Treatment of mastocytosis with masitinib |
| CA2816957A1 (en) | 2010-11-07 | 2012-05-10 | Targegen, Inc. | Compositions and methods for treating myelofibrosis |
| WO2012104402A1 (en) * | 2011-02-04 | 2012-08-09 | Ab Science | Treatment of severe persitent asthma with masitinib |
| KR101821226B1 (en) * | 2011-03-01 | 2018-01-23 | 엔파마콘, 엘엘씨 | Use of n-(4-methoxyphenyl)-1-phenyl-1h-pyrazol-3-amine and related compounds |
| WO2012136732A1 (en) | 2011-04-08 | 2012-10-11 | Ab Science | Treatment of multiple myeloma with masitinib |
| CN103130791B (en) * | 2011-11-30 | 2016-05-04 | 正大天晴药业集团股份有限公司 | A kind of new benzamides compounds |
| CN103130792B (en) * | 2011-11-30 | 2016-05-04 | 正大天晴药业集团股份有限公司 | A kind of thiazolamine compounds |
| EP2903616B8 (en) | 2012-10-04 | 2018-02-07 | AB Science | Use of masitinib in combination with gemcitabine for treating a subgroup of patients suffering from pancreatic cancer |
| TW201534597A (en) | 2013-06-20 | 2015-09-16 | Ab Science | Benzimidazole derivatives as selective proteine kinase inhibitors |
| USD704961S1 (en) | 2013-07-03 | 2014-05-20 | Wearwell | Studded molded mat |
| WO2015082496A1 (en) | 2013-12-02 | 2015-06-11 | Ab Science | Use of masitinib for treatment of colorectal cancer |
| EP2886543A1 (en) | 2013-12-18 | 2015-06-24 | Sandoz Ag | Crystalline form of mastinib mesylate |
| WO2015185958A1 (en) | 2014-06-02 | 2015-12-10 | Ab Science | Use of an inhibitor of kinase activity, particularly masitinib, for treatment of prostate cancer |
| CN104045632B (en) * | 2014-06-03 | 2016-09-07 | 辽宁大学 | Antineoplastic benzodihydropyran (thiapyran) amides compound and pharmaceutically acceptable salt thereof and preparation method and application |
| US20160175302A1 (en) * | 2014-12-17 | 2016-06-23 | Ab Science | Masitinib for treating gastric cancer |
| ES2796276T3 (en) * | 2015-02-05 | 2020-11-26 | Ab Science | Compounds with antitumor activity |
| WO2017016512A1 (en) * | 2015-07-29 | 2017-02-02 | 苏州晶云药物科技有限公司 | New crystalline form of masitinib mesylate and preparation method therefor |
| WO2017060308A1 (en) | 2015-10-05 | 2017-04-13 | Ab Science | Treatment of severe systemic mastocytosis with masitinib |
| EA038531B1 (en) | 2016-03-25 | 2021-09-10 | Аб Сьянс | Method for treatment of an amyotrophic lateral sclerosis, use of a pharmaceutical composition comprising masitinib |
| US10961235B2 (en) | 2017-03-31 | 2021-03-30 | Sandoz Ag | Crystalline form of masitinib |
| CN108721283A (en) * | 2017-04-20 | 2018-11-02 | 华东理工大学 | Application of the thiazole in treating non-lymphocytic leukemia |
| CA3129665A1 (en) | 2019-03-21 | 2020-09-24 | Onxeo | A dbait molecule in combination with kinase inhibitor for the treatment of cancer |
| EP4054579A1 (en) | 2019-11-08 | 2022-09-14 | Institut National de la Santé et de la Recherche Médicale (INSERM) | Methods for the treatment of cancers that have acquired resistance to kinase inhibitors |
| WO2021099616A1 (en) | 2019-11-22 | 2021-05-27 | Ab Science | Masitinib for the treatment of sickle cell disease |
| EP3831384A1 (en) | 2019-12-02 | 2021-06-09 | AB Science | Use of masitinib for the treatment of eosinophilic asthma |
| WO2021148581A1 (en) | 2020-01-22 | 2021-07-29 | Onxeo | Novel dbait molecule and its use |
| AU2021223710A1 (en) | 2020-02-20 | 2022-07-28 | Ab Science | Masitinib for the treatment of a multiple sclerosis patient subpopulation |
| WO2021205029A1 (en) | 2020-04-10 | 2021-10-14 | Ab Science | Use of masitinib for the treatment of coronavirus disease 2019 (covid-19) |
| JP2023554354A (en) | 2020-12-16 | 2023-12-27 | エービー サイエンス | masitinib for the treatment of Alzheimer's disease |
| CN117337177A (en) | 2021-05-17 | 2024-01-02 | Ab科学有限公司 | Mosatinib for the treatment of castration resistant prostate cancer |
| WO2023081923A1 (en) | 2021-11-08 | 2023-05-11 | Frequency Therapeutics, Inc. | Platelet-derived growth factor receptor (pdgfr) alpha inhibitors and uses thereof |
| CN114644582B (en) * | 2022-04-11 | 2024-03-29 | 中原工学院 | Preparation method of phenyl dithiourea compound |
Citations (31)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US590622A (en) * | 1897-09-28 | Edward mcconnell | ||
| US3192225A (en) * | 1961-04-24 | 1965-06-29 | Geigy Chem Corp | 2-substituted aminothiazoles |
| US3467666A (en) * | 1966-11-07 | 1969-09-16 | Geigy Chem Corp | 2-substituted aminothiazoles |
| US3740420A (en) * | 1967-11-28 | 1973-06-19 | Crown Zellerbach Corp | Pharmaceutical compositions with dimethyl sulfoxide |
| US3743727A (en) * | 1970-11-16 | 1973-07-03 | Crown Zellerbach Corp | Enhancing tissue penetration of certain antimicrobial agents with dimethyl sulfoxide |
| US3772295A (en) * | 1970-02-16 | 1973-11-13 | Innothera Lab Sa | Quinazoline derivatives |
| US3989816A (en) * | 1975-06-19 | 1976-11-02 | Nelson Research & Development Company | Vehicle composition containing 1-substituted azacycloheptan-2-ones |
| US4322433A (en) * | 1979-11-06 | 1982-03-30 | Euroceltique, S.A. | Percutaneous administration of nitroglycerine |
| US4343940A (en) * | 1979-02-13 | 1982-08-10 | Mead Johnson & Company | Anti-tumor quinazoline compounds |
| US4379454A (en) * | 1981-02-17 | 1983-04-12 | Alza Corporation | Dosage for coadministering drug and percutaneous absorption enhancer |
| US4405616A (en) * | 1975-06-19 | 1983-09-20 | Nelson Research & Development Company | Penetration enhancers for transdermal drug delivery of systemic agents |
| US4411893A (en) * | 1981-08-14 | 1983-10-25 | Minnesota Mining And Manufacturing Company | Topical medicament preparations |
| US4460372A (en) * | 1981-02-17 | 1984-07-17 | Alza Corporation | Percutaneous absorption enhancer dispenser for use in coadministering drug and percutaneous absorption enhancer |
| US4575515A (en) * | 1984-05-14 | 1986-03-11 | Clark Pharmaceutical Laboratories Ltd. | Pharmaceutical solutions comprising dimethyl sulfoxide |
| US4615699A (en) * | 1985-05-03 | 1986-10-07 | Alza Corporation | Transdermal delivery system for delivering nitroglycerin at high transdermal fluxes |
| US5217999A (en) * | 1987-12-24 | 1993-06-08 | Yissum Research Development Company Of The Hebrew University Of Jerusalem | Styryl compounds which inhibit EGF receptor protein tyrosine kinase |
| US5302606A (en) * | 1990-04-16 | 1994-04-12 | Rhone-Poulenc Rorer Pharmaceuticals Inc. | Styryl-substituted pyridyl compounds which inhibit EGF receptor tyrosine kinase |
| US5330992A (en) * | 1992-10-23 | 1994-07-19 | Sterling Winthrop Inc. | 1-cyclopropyl-4-pyridyl-quinolinones |
| US5521184A (en) * | 1992-04-03 | 1996-05-28 | Ciba-Geigy Corporation | Pyrimidine derivatives and processes for the preparation thereof |
| US5556611A (en) * | 1988-05-04 | 1996-09-17 | Hermes Fabrik Pharmazeutischer Praparate | Vitamin A aerosol-inhalant preparations and method |
| US5656643A (en) * | 1993-11-08 | 1997-08-12 | Rhone-Poulenc Rorer Pharmaceuticals Inc. | Bis mono-and bicyclic aryl and heteroaryl compounds which inhibit EGF and/or PDGF receptor tyrosine kinase |
| US5710158A (en) * | 1991-05-10 | 1998-01-20 | Rhone-Poulenc Rorer Pharmaceuticals Inc. | Aryl and heteroaryl quinazoline compounds which inhibit EGF and/or PDGF receptor tyrosine kinase |
| US5714493A (en) * | 1991-05-10 | 1998-02-03 | Rhone-Poulenc Rorer Pharmaceuticals, Inc. | Aryl and heteroaryl quinazoline compounds which inhibit CSF-1R receptor tyrosine kinase |
| US5721237A (en) * | 1991-05-10 | 1998-02-24 | Rhone-Poulenc Rorer Pharmaceuticals Inc. | Protein tyrosine kinase aryl and heteroaryl quinazoline compounds having selective inhibition of HER-2 autophosphorylation properties |
| US5776020A (en) * | 1997-05-19 | 1998-07-07 | Barone; Michael A. | Tensioning device for sporting racquets |
| US5792783A (en) * | 1995-06-07 | 1998-08-11 | Sugen, Inc. | 3-heteroaryl-2-indolinone compounds for the treatment of disease |
| US5883166A (en) * | 1997-06-26 | 1999-03-16 | Jennings; Thomas C. | Liquid internal mold release agents for unsaturated polyester thermosetting molding compounds |
| US6291514B1 (en) * | 1998-02-09 | 2001-09-18 | 3-Dimensional Pharmaceuticals, Inc. | Heteroaryl amidines, methylamidines and guanidines, preparation thereof, and use thereof as protease inhibitors |
| US20010044545A1 (en) * | 2000-04-03 | 2001-11-22 | 3-Dimensional Pharmaceuticals, Inc. | Substituted thiazoles and the use thereof as inhibitors of plasminogen activator inhibitor-1 |
| US20030158199A1 (en) * | 2002-01-25 | 2003-08-21 | Kylix, B.V. | Novel compounds for inhibition of Tie-2 |
| US20040110801A1 (en) * | 2001-02-08 | 2004-06-10 | Franscesco Makovec | Novel benzamidine derivatives having anti-inflammatory and immunosuppressive |
Family Cites Families (44)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS5968336U (en) | 1982-10-28 | 1984-05-09 | シャープ株式会社 | Document cover opening/closing detection device |
| JPS6328447A (en) | 1986-07-21 | 1988-02-06 | Toshiba Corp | Accelerating tube |
| JP2752361B2 (en) | 1986-11-03 | 1998-05-18 | エバレディー、バッテリー、カンパニー、インコーポレーテッド | Sealed non-aqueous battery with positive terminal pin and perchlorate electrolyte |
| GB9413975D0 (en) * | 1994-07-11 | 1994-08-31 | Fujisawa Pharmaceutical Co | New heterobicyclic derivatives |
| DE69105495T2 (en) | 1990-04-02 | 1995-04-06 | Pfizer | BENZYLPHOSPHONIC ACID TYROSINKINAS INHIBITORS. |
| CA2102780C (en) | 1991-05-10 | 2007-01-09 | Alfred P. Spada | Bis mono-and bicyclic aryl and heteroaryl compounds which inhibit egf and/or pdgf receptor tyrosine kinase |
| JP2549583B2 (en) | 1991-05-10 | 1996-10-30 | 明成化学工業株式会社 | Method for preventing fading of unfixed dye in reactive dyeing of cellulose fiber |
| WO1992021660A1 (en) | 1991-05-29 | 1992-12-10 | Pfizer, Inc. | Tricyclic polyhydroxylic tyrosine kinase inhibitors |
| NZ243082A (en) | 1991-06-28 | 1995-02-24 | Ici Plc | 4-anilino-quinazoline derivatives; pharmaceutical compositions, preparatory processes, and use thereof |
| TW225528B (en) * | 1992-04-03 | 1994-06-21 | Ciba Geigy Ag | |
| SK283413B6 (en) | 1992-08-06 | 2003-07-01 | Warner-Lambert Company | 2-Thioindoles, 2-indolinethiones and polysulfides, their analogues (2-indolineselenone) (selenide), and pharmaceutical compositions based on them |
| GB9323290D0 (en) | 1992-12-10 | 1994-01-05 | Zeneca Ltd | Quinazoline derivatives |
| GB9226855D0 (en) | 1992-12-23 | 1993-02-17 | Erba Carlo Spa | Vinylene-azaindole derivatives and process for their preparation |
| US6239172B1 (en) * | 1997-04-10 | 2001-05-29 | Nitrosystems, Inc. | Formulations for treating disease and methods of using same |
| JP3728770B2 (en) | 1995-06-01 | 2005-12-21 | ブラザー工業株式会社 | Document reader |
| US5906202A (en) | 1996-11-21 | 1999-05-25 | Aradigm Corporation | Device and method for directing aerosolized mist to a specific area of the respiratory tract |
| CO4940418A1 (en) | 1997-07-18 | 2000-07-24 | Novartis Ag | MODIFICATION OF A CRYSTAL OF A DERIVATIVE OF N-PHENYL-2-PIRIMIDINAMINE, PROCESSES FOR ITS MANUFACTURE AND USE |
| JP4333676B2 (en) | 1997-11-21 | 2009-09-16 | オムロン株式会社 | Program control apparatus, program control method, and program recording medium |
| JP2002531504A (en) * | 1998-12-07 | 2002-09-24 | スミスクライン・ビーチャム・コーポレイション | MYT1 kinase inhibitor |
| EP1139754A4 (en) | 1998-12-31 | 2002-12-18 | Sugen Inc | 3-heteroarylidenyl-2-indolinone compounds for modulating protein kinase activity and for use in cancer chemotherapy |
| UA71971C2 (en) * | 1999-06-04 | 2005-01-17 | Agoron Pharmaceuticals Inc | Diaminothiazoles, composition based thereon, a method for modulation of protein kinases activity, a method for the treatment of diseases mediated by protein kinases |
| ATE414079T1 (en) | 2000-03-01 | 2008-11-15 | Janssen Pharmaceutica Nv | 2,4-DISUBSTITUTED THIAZOLYL DERIVATIVES |
| US7087608B2 (en) | 2000-03-03 | 2006-08-08 | Robert Charles Atkins | Use of PDGF receptor tyrosine kinase inhibitors for the treatment of diabetic nephropathy |
| US6892245B1 (en) | 2000-09-22 | 2005-05-10 | Nortel Networks Limited | Management information base for a multi-domain network address translator |
| JP2002185796A (en) | 2000-12-11 | 2002-06-28 | Canon Inc | Image read processing system and image read processing method, and recording medium |
| GB0108606D0 (en) | 2001-04-05 | 2001-05-23 | Novartis Ag | Organic compounds |
| EP1260876B1 (en) | 2001-05-25 | 2013-05-22 | Ricoh Company, Ltd. | A platen cover, image scanning device and image forming apparatus with the cover |
| US7727731B2 (en) | 2001-06-29 | 2010-06-01 | Ab Science | Potent, selective and non toxic c-kit inhibitors |
| DE60227709D1 (en) | 2001-06-29 | 2008-08-28 | Ab Science | THE USE OF C-CRITERIA FOR THE TREATMENT OF AUTOIMMUNE DISEASES |
| CA2452390A1 (en) | 2001-06-29 | 2003-01-09 | Ab Science | Use of tyrosine kinase inhibitors for treating bone loss |
| ATE343415T1 (en) | 2001-06-29 | 2006-11-15 | Ab Science | THE USE OF C-KIT INHIBITORS FOR THE TREATMENT OF INFLAMMATORY BOWEL DISEASE |
| CA2452167A1 (en) | 2001-06-29 | 2003-01-09 | Ab Science | Use of tyrosine kinase inhibitors for treating multiple sclerosis (ms) |
| US20030091974A1 (en) | 2001-06-29 | 2003-05-15 | Alain Moussy | Method for screening compounds capable of depleting mast cells |
| WO2003002106A2 (en) | 2001-06-29 | 2003-01-09 | Ab Science | Use of tyrosine kinase inhibitions for treating allergic diseases |
| JP2004537537A (en) | 2001-06-29 | 2004-12-16 | アブ サイエンス | Uses of tyrosine kinase inhibitors to treat inflammatory diseases |
| US20040266797A1 (en) | 2001-06-29 | 2004-12-30 | Alain Moussy | Use of potent,selective and non toxic c-kit inhibitors for treating tumor angiogensis |
| JP2005503361A (en) | 2001-06-29 | 2005-02-03 | アブ サイエンス | Use of potent, selective and non-toxic c-kit inhibitors for the treatment of mastocytosis |
| ATE388711T1 (en) | 2001-09-20 | 2008-03-15 | Ab Science | C-KIT INHIBITORS FOR THE TREATMENT OF BACTERIAL INFECTIONS |
| CA2461183A1 (en) | 2001-09-20 | 2003-05-15 | Ab Science | Use of tyrosine kinase inhibitors for whitening human skin and treating melanocyte dysfunction associated diseases |
| JP2005507917A (en) | 2001-09-20 | 2005-03-24 | アブ サイエンス | Methods of using tyrosine kinase inhibitors to promote hair growth |
| JP2003134307A (en) | 2001-10-26 | 2003-05-09 | Canon Inc | Image reading apparatus |
| WO2003062215A1 (en) | 2002-01-25 | 2003-07-31 | Kylix Pharmaceuticals B.V. | 4(hetero-) aryl substituted (thia-/oxa-/pyra) zoles for inhibition of tie-2 |
| IL166528A0 (en) * | 2002-08-02 | 2006-01-15 | Ab Science | 2-(3-aminoaryl)amino-4-aryl-thiazoles and their use as c-kit inhibitors |
| MX2009008665A (en) | 2007-02-13 | 2009-08-21 | Ab Science | Process for the synthesis of 2-aminothiazole compounds as kinase inhibitors. |
-
2003
- 2003-07-31 IL IL16652803A patent/IL166528A0/en active IP Right Grant
- 2003-07-31 JP JP2004527232A patent/JP4726486B2/en not_active Expired - Lifetime
- 2003-07-31 DE DE60316810T patent/DE60316810T2/en not_active Expired - Lifetime
- 2003-07-31 MX MXPA05001277A patent/MXPA05001277A/en active IP Right Grant
- 2003-07-31 CN CNB03823145XA patent/CN100491374C/en not_active Expired - Lifetime
- 2003-07-31 NZ NZ538490A patent/NZ538490A/en not_active IP Right Cessation
- 2003-07-31 PT PT03784419T patent/PT1525200E/en unknown
- 2003-07-31 DK DK03784419T patent/DK1525200T3/en active
- 2003-07-31 CA CA2494695A patent/CA2494695C/en not_active Expired - Lifetime
- 2003-07-31 AT AT03784419T patent/ATE375342T1/en active
- 2003-07-31 KR KR1020057001919A patent/KR101036866B1/en active IP Right Grant
- 2003-07-31 ES ES03784419T patent/ES2294344T3/en not_active Expired - Lifetime
- 2003-07-31 US US10/523,018 patent/US20050239852A1/en not_active Abandoned
- 2003-07-31 EP EP03784419A patent/EP1525200B1/en not_active Expired - Lifetime
- 2003-07-31 BR BRPI0313165A patent/BRPI0313165B8/en active IP Right Grant
- 2003-07-31 WO PCT/IB2003/003685 patent/WO2004014903A1/en active IP Right Grant
- 2003-08-01 US US10/632,101 patent/US7423055B2/en not_active Expired - Lifetime
-
2005
- 2005-02-15 ZA ZA2005/01331A patent/ZA200501331B/en unknown
- 2005-03-02 NO NO20051115A patent/NO330608B1/en not_active IP Right Cessation
-
2006
- 2006-03-07 HK HK06102936.7A patent/HK1084382A1/en not_active IP Right Cessation
-
2007
- 2007-07-18 US US11/779,633 patent/US20080255141A1/en not_active Abandoned
- 2007-11-27 CY CY20071101517T patent/CY1107036T1/en unknown
-
2011
- 2011-01-28 US US13/015,664 patent/US8835435B2/en not_active Expired - Lifetime
-
2013
- 2013-11-26 US US14/089,946 patent/US8993573B2/en not_active Expired - Lifetime
Patent Citations (34)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US590622A (en) * | 1897-09-28 | Edward mcconnell | ||
| US3192225A (en) * | 1961-04-24 | 1965-06-29 | Geigy Chem Corp | 2-substituted aminothiazoles |
| US3201409A (en) * | 1961-04-24 | 1965-08-17 | John D Spivack | 2-substituted aminothiazoles |
| US3467666A (en) * | 1966-11-07 | 1969-09-16 | Geigy Chem Corp | 2-substituted aminothiazoles |
| US3740420A (en) * | 1967-11-28 | 1973-06-19 | Crown Zellerbach Corp | Pharmaceutical compositions with dimethyl sulfoxide |
| US3772295A (en) * | 1970-02-16 | 1973-11-13 | Innothera Lab Sa | Quinazoline derivatives |
| US3743727A (en) * | 1970-11-16 | 1973-07-03 | Crown Zellerbach Corp | Enhancing tissue penetration of certain antimicrobial agents with dimethyl sulfoxide |
| US4405616A (en) * | 1975-06-19 | 1983-09-20 | Nelson Research & Development Company | Penetration enhancers for transdermal drug delivery of systemic agents |
| US3989816A (en) * | 1975-06-19 | 1976-11-02 | Nelson Research & Development Company | Vehicle composition containing 1-substituted azacycloheptan-2-ones |
| US4343940A (en) * | 1979-02-13 | 1982-08-10 | Mead Johnson & Company | Anti-tumor quinazoline compounds |
| US4322433A (en) * | 1979-11-06 | 1982-03-30 | Euroceltique, S.A. | Percutaneous administration of nitroglycerine |
| US4379454A (en) * | 1981-02-17 | 1983-04-12 | Alza Corporation | Dosage for coadministering drug and percutaneous absorption enhancer |
| US4460372A (en) * | 1981-02-17 | 1984-07-17 | Alza Corporation | Percutaneous absorption enhancer dispenser for use in coadministering drug and percutaneous absorption enhancer |
| US4411893A (en) * | 1981-08-14 | 1983-10-25 | Minnesota Mining And Manufacturing Company | Topical medicament preparations |
| US4575515A (en) * | 1984-05-14 | 1986-03-11 | Clark Pharmaceutical Laboratories Ltd. | Pharmaceutical solutions comprising dimethyl sulfoxide |
| US4615699A (en) * | 1985-05-03 | 1986-10-07 | Alza Corporation | Transdermal delivery system for delivering nitroglycerin at high transdermal fluxes |
| US5217999A (en) * | 1987-12-24 | 1993-06-08 | Yissum Research Development Company Of The Hebrew University Of Jerusalem | Styryl compounds which inhibit EGF receptor protein tyrosine kinase |
| US5556611A (en) * | 1988-05-04 | 1996-09-17 | Hermes Fabrik Pharmazeutischer Praparate | Vitamin A aerosol-inhalant preparations and method |
| US5302606A (en) * | 1990-04-16 | 1994-04-12 | Rhone-Poulenc Rorer Pharmaceuticals Inc. | Styryl-substituted pyridyl compounds which inhibit EGF receptor tyrosine kinase |
| US5710158A (en) * | 1991-05-10 | 1998-01-20 | Rhone-Poulenc Rorer Pharmaceuticals Inc. | Aryl and heteroaryl quinazoline compounds which inhibit EGF and/or PDGF receptor tyrosine kinase |
| US5714493A (en) * | 1991-05-10 | 1998-02-03 | Rhone-Poulenc Rorer Pharmaceuticals, Inc. | Aryl and heteroaryl quinazoline compounds which inhibit CSF-1R receptor tyrosine kinase |
| US5721237A (en) * | 1991-05-10 | 1998-02-24 | Rhone-Poulenc Rorer Pharmaceuticals Inc. | Protein tyrosine kinase aryl and heteroaryl quinazoline compounds having selective inhibition of HER-2 autophosphorylation properties |
| US5521184A (en) * | 1992-04-03 | 1996-05-28 | Ciba-Geigy Corporation | Pyrimidine derivatives and processes for the preparation thereof |
| US5330992A (en) * | 1992-10-23 | 1994-07-19 | Sterling Winthrop Inc. | 1-cyclopropyl-4-pyridyl-quinolinones |
| US5656643A (en) * | 1993-11-08 | 1997-08-12 | Rhone-Poulenc Rorer Pharmaceuticals Inc. | Bis mono-and bicyclic aryl and heteroaryl compounds which inhibit EGF and/or PDGF receptor tyrosine kinase |
| US5883113A (en) * | 1995-06-07 | 1999-03-16 | Sugen, Inc. | 3-(4'-Bromobenzylindenyl)-2-indolinone and analogues thereof for the treatment of disease |
| US5792783A (en) * | 1995-06-07 | 1998-08-11 | Sugen, Inc. | 3-heteroaryl-2-indolinone compounds for the treatment of disease |
| US5834504A (en) * | 1995-06-07 | 1998-11-10 | Sugen, Inc. | 3-(2'-halobenzylidenyl)-2-indolinone compounds for the treatment of disease |
| US5776020A (en) * | 1997-05-19 | 1998-07-07 | Barone; Michael A. | Tensioning device for sporting racquets |
| US5883166A (en) * | 1997-06-26 | 1999-03-16 | Jennings; Thomas C. | Liquid internal mold release agents for unsaturated polyester thermosetting molding compounds |
| US6291514B1 (en) * | 1998-02-09 | 2001-09-18 | 3-Dimensional Pharmaceuticals, Inc. | Heteroaryl amidines, methylamidines and guanidines, preparation thereof, and use thereof as protease inhibitors |
| US20010044545A1 (en) * | 2000-04-03 | 2001-11-22 | 3-Dimensional Pharmaceuticals, Inc. | Substituted thiazoles and the use thereof as inhibitors of plasminogen activator inhibitor-1 |
| US20040110801A1 (en) * | 2001-02-08 | 2004-06-10 | Franscesco Makovec | Novel benzamidine derivatives having anti-inflammatory and immunosuppressive |
| US20030158199A1 (en) * | 2002-01-25 | 2003-08-21 | Kylix, B.V. | Novel compounds for inhibition of Tie-2 |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20090221565A1 (en) * | 2008-02-29 | 2009-09-03 | Vetoquinol Sa | Novel 7-substituted 3-carboxy-oxadiazino-quinolone derivatives, their preparation and their application as anti-bacterials |
| US7713965B2 (en) | 2008-02-29 | 2010-05-11 | Vetoquinol Sa | 7-substituted 3-carboxy-oxadiazino-quinolone derivatives, their preparation and their application as anti-bacterials |
| US20100009980A1 (en) * | 2008-07-09 | 2010-01-14 | Vetoquinol Sa | Novel 9-substituted-5-carboxy-oxadiazino-quinolone derivatives, their preparation and their application as anti-bacterials |
| US8071590B2 (en) * | 2008-07-09 | 2011-12-06 | Vetoquinol Sa | 9-substituted-5-carboxy-oxadiazino-quinolone derivatives, their preparation and their application as anti-bacterials |
| US8920785B2 (en) | 2010-03-23 | 2014-12-30 | Beiersdorf Ag | Cosmetic or dermatological preparations with a content of one or more thiazole derivates |
| US9168245B2 (en) | 2011-07-27 | 2015-10-27 | Ab Science | Selective protein kinase inhibitors |
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US8993573B2 (en) | 2-(3-aminoaryl) amino-4-aryl-thiazoles and their use as c-kit inhibitors | |
| US8110591B2 (en) | 2-aminoaryloxazole compounds as tyrosine kinase inhibitors | |
| US8658659B2 (en) | Substituted oxazole derivatives and their use as tyrosine kinase inhibitors | |
| US8450302B2 (en) | 2-(3-aminoaryl) amino-4-aryl-thiazoles and their use as c-kit inhibitors | |
| NZ548884A (en) | 2-(3-substituted-aryl)amino-4-aryl-thiazoles as tyrosine kinase inhibitors | |
| WO2006064375A9 (en) | Aminoaryl substituted five-membered ring heterocyclic compounds for the treatment of diseases | |
| US20080039466A1 (en) | 2-(3-Substituted-Aryl) Amino-4-Aryl-Thiazoles As Tyrosine Kinase Inhibitors | |
| AU2003253195B2 (en) | 2-(3-aminoaryl)amino-4-aryl-thiazoles and their use as c-kit inhibitors | |
| MXPA06008597A (en) | 2-(3-substituted-aryl)amino-4-aryl-thiazoles as tyrosine kinase inhibitors |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| STCB | Information on status: application discontinuation |
Free format text: ABANDONED -- FAILURE TO RESPOND TO AN OFFICE ACTION |