US20030191066A1 - Enone cancer therapeutics - Google Patents
Enone cancer therapeutics Download PDFInfo
- Publication number
- US20030191066A1 US20030191066A1 US10/098,834 US9883402A US2003191066A1 US 20030191066 A1 US20030191066 A1 US 20030191066A1 US 9883402 A US9883402 A US 9883402A US 2003191066 A1 US2003191066 A1 US 2003191066A1
- Authority
- US
- United States
- Prior art keywords
- enone
- cancer therapeutic
- therapeutic
- sex hormone
- cancer
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Abandoned
Links
- 239000012830 cancer therapeutic Substances 0.000 title claims abstract description 33
- 150000001875 compounds Chemical class 0.000 claims abstract description 9
- RWSXRVCMGQZWBV-WDSKDSINSA-N glutathione Chemical compound OC(=O)[C@@H](N)CCC(=O)N[C@@H](CS)C(=O)NCC(O)=O RWSXRVCMGQZWBV-WDSKDSINSA-N 0.000 claims description 80
- 229960003180 glutathione Drugs 0.000 claims description 35
- 238000000034 method Methods 0.000 claims description 27
- MUMGGOZAMZWBJJ-DYKIIFRCSA-N Testostosterone Chemical compound O=C1CC[C@]2(C)[C@H]3CC[C@](C)([C@H](CC4)O)[C@@H]4[C@@H]3CCC2=C1 MUMGGOZAMZWBJJ-DYKIIFRCSA-N 0.000 claims description 26
- 206010028980 Neoplasm Diseases 0.000 claims description 21
- 239000003163 gonadal steroid hormone Substances 0.000 claims description 18
- 102000004357 Transferases Human genes 0.000 claims description 17
- 108090000992 Transferases Proteins 0.000 claims description 17
- 150000003431 steroids Chemical class 0.000 claims description 17
- 108010024636 Glutathione Proteins 0.000 claims description 16
- 229920001577 copolymer Polymers 0.000 claims description 16
- -1 glutathionyl Chemical group 0.000 claims description 16
- 229960003604 testosterone Drugs 0.000 claims description 14
- 239000000203 mixture Substances 0.000 claims description 9
- 201000011510 cancer Diseases 0.000 claims description 7
- 229960005309 estradiol Drugs 0.000 claims description 7
- 239000000126 substance Substances 0.000 claims description 7
- VOXZDWNPVJITMN-ZBRFXRBCSA-N 17β-estradiol Chemical compound OC1=CC=C2[C@H]3CC[C@](C)([C@H](CC4)O)[C@@H]4[C@@H]3CCC2=C1 VOXZDWNPVJITMN-ZBRFXRBCSA-N 0.000 claims description 6
- 230000009471 action Effects 0.000 claims description 6
- 229930182833 estradiol Natural products 0.000 claims description 6
- 239000000262 estrogen Substances 0.000 claims description 5
- 229940011871 estrogen Drugs 0.000 claims description 5
- 125000000219 ethylidene group Chemical group [H]C(=[*])C([H])([H])[H] 0.000 claims description 5
- 102000004169 proteins and genes Human genes 0.000 claims description 5
- 108090000623 proteins and genes Proteins 0.000 claims description 5
- 239000003795 chemical substances by application Substances 0.000 claims description 2
- 238000001990 intravenous administration Methods 0.000 claims description 2
- 230000001225 therapeutic effect Effects 0.000 claims 8
- 230000000269 nucleophilic effect Effects 0.000 claims 4
- 102000039446 nucleic acids Human genes 0.000 claims 2
- 108020004707 nucleic acids Proteins 0.000 claims 2
- 150000007523 nucleic acids Chemical class 0.000 claims 2
- 238000005935 nucleophilic addition reaction Methods 0.000 claims 2
- 239000008194 pharmaceutical composition Substances 0.000 claims 2
- 238000009472 formulation Methods 0.000 claims 1
- 238000004519 manufacturing process Methods 0.000 claims 1
- 0 *[C@@H]1C=C(CC)C(=O)[C@H](*)[C@@H]1*.*[C@@H]1C=C(COCOC=CC)C(=O)[C@H](*)[C@@H]1* Chemical compound *[C@@H]1C=C(CC)C(=O)[C@H](*)[C@@H]1*.*[C@@H]1C=C(COCOC=CC)C(=O)[C@H](*)[C@@H]1* 0.000 description 18
- 102000004190 Enzymes Human genes 0.000 description 15
- 108090000790 Enzymes Proteins 0.000 description 15
- 230000000259 anti-tumor effect Effects 0.000 description 14
- 230000015572 biosynthetic process Effects 0.000 description 11
- 238000003786 synthesis reaction Methods 0.000 description 10
- 238000006243 chemical reaction Methods 0.000 description 9
- 229940002612 prodrug Drugs 0.000 description 9
- 239000000651 prodrug Substances 0.000 description 9
- 210000001519 tissue Anatomy 0.000 description 9
- AIJULSRZWUXGPQ-UHFFFAOYSA-N Methylglyoxal Chemical compound CC(=O)C=O AIJULSRZWUXGPQ-UHFFFAOYSA-N 0.000 description 8
- 230000000694 effects Effects 0.000 description 8
- 102100030943 Glutathione S-transferase P Human genes 0.000 description 7
- 101001010139 Homo sapiens Glutathione S-transferase P Proteins 0.000 description 7
- 239000002246 antineoplastic agent Substances 0.000 description 7
- 239000003112 inhibitor Substances 0.000 description 7
- IQAJMALOOLNRLN-GORDUTHDSA-N C/C=C/C(=O)OCC1=CCCCC1=O Chemical compound C/C=C/C(=O)OCC1=CCCCC1=O IQAJMALOOLNRLN-GORDUTHDSA-N 0.000 description 6
- PSJQCAMBOYBQEU-KHSNLZPESA-N [(3r,4r,5r)-3,4,5-trihydroxy-6-oxocyclohexen-1-yl]methyl (e)-but-2-enoate Chemical compound C\C=C\C(=O)OCC1=C[C@@H](O)[C@@H](O)[C@@H](O)C1=O PSJQCAMBOYBQEU-KHSNLZPESA-N 0.000 description 6
- XUJNEKJLAYXESH-UHFFFAOYSA-N cysteine Natural products SCC(N)C(O)=O XUJNEKJLAYXESH-UHFFFAOYSA-N 0.000 description 6
- 235000018417 cysteine Nutrition 0.000 description 6
- 238000011534 incubation Methods 0.000 description 6
- 230000003389 potentiating effect Effects 0.000 description 6
- GTLOUKSKXJYPCX-UHFFFAOYSA-N C#CC(=O)OCC1=CCCCC1=O Chemical compound C#CC(=O)OCC1=CCCCC1=O GTLOUKSKXJYPCX-UHFFFAOYSA-N 0.000 description 5
- PCJVCNUNDKLWNR-UHFFFAOYSA-N C#CCCC(=O)OCC1=CCCCC1=O Chemical compound C#CCCC(=O)OCC1=CCCCC1=O PCJVCNUNDKLWNR-UHFFFAOYSA-N 0.000 description 5
- JTIFAOAWVCLNBR-UHFFFAOYSA-N CC#CC(=O)OCC1=CCCCC1=O Chemical compound CC#CC(=O)OCC1=CCCCC1=O JTIFAOAWVCLNBR-UHFFFAOYSA-N 0.000 description 5
- JRZJKWGQFNTSRN-UHFFFAOYSA-N Geldanamycin Natural products C1C(C)CC(OC)C(O)C(C)C=C(C)C(OC(N)=O)C(OC)CCC=C(C)C(=O)NC2=CC(=O)C(OC)=C1C2=O JRZJKWGQFNTSRN-UHFFFAOYSA-N 0.000 description 5
- 229940116851 Glyoxalase I inhibitor Drugs 0.000 description 5
- QTQAWLPCGQOSGP-GBTDJJJQSA-N geldanamycin Chemical compound N1C(=O)\C(C)=C/C=C\[C@@H](OC)[C@H](OC(N)=O)\C(C)=C/[C@@H](C)[C@@H](O)[C@H](OC)C[C@@H](C)CC2=C(OC)C(=O)C=C1C2=O QTQAWLPCGQOSGP-GBTDJJJQSA-N 0.000 description 5
- 230000007246 mechanism Effects 0.000 description 5
- 239000000758 substrate Substances 0.000 description 5
- 102000014017 Lactoylglutathione lyase Human genes 0.000 description 4
- 108010050765 Lactoylglutathione lyase Proteins 0.000 description 4
- FWFSEYBSWVRWGL-UHFFFAOYSA-N cyclohex-2-enone Chemical compound O=C1CCCC=C1 FWFSEYBSWVRWGL-UHFFFAOYSA-N 0.000 description 4
- 239000003814 drug Substances 0.000 description 4
- 229940079593 drug Drugs 0.000 description 4
- 238000000655 nuclear magnetic resonance spectrum Methods 0.000 description 4
- 150000003573 thiols Chemical class 0.000 description 4
- 206010006187 Breast cancer Diseases 0.000 description 3
- 208000026310 Breast neoplasm Diseases 0.000 description 3
- 108020004414 DNA Proteins 0.000 description 3
- WSFSSNUMVMOOMR-UHFFFAOYSA-N Formaldehyde Chemical compound O=C WSFSSNUMVMOOMR-UHFFFAOYSA-N 0.000 description 3
- PSJQCAMBOYBQEU-UHFFFAOYSA-N Glyoxalase I Natural products CC=CC(=O)OCC1=CC(O)C(O)C(O)C1=O PSJQCAMBOYBQEU-UHFFFAOYSA-N 0.000 description 3
- 102100035328 Transmembrane protein 258 Human genes 0.000 description 3
- 230000001093 anti-cancer Effects 0.000 description 3
- 229940041181 antineoplastic drug Drugs 0.000 description 3
- 210000004027 cell Anatomy 0.000 description 3
- 230000002860 competitive effect Effects 0.000 description 3
- 229940127089 cytotoxic agent Drugs 0.000 description 3
- 238000010494 dissociation reaction Methods 0.000 description 3
- 230000005593 dissociations Effects 0.000 description 3
- 210000003743 erythrocyte Anatomy 0.000 description 3
- 239000000543 intermediate Substances 0.000 description 3
- 230000003834 intracellular effect Effects 0.000 description 3
- 235000018102 proteins Nutrition 0.000 description 3
- 238000004007 reversed phase HPLC Methods 0.000 description 3
- QMDIIXACBPZLCK-UHFFFAOYSA-N 2-(hydroxymethyl)cyclohex-2-en-1-one Chemical compound OCC1=CCCCC1=O QMDIIXACBPZLCK-UHFFFAOYSA-N 0.000 description 2
- 102100032187 Androgen receptor Human genes 0.000 description 2
- 238000005712 Baylis-Hillman reaction Methods 0.000 description 2
- XUJNEKJLAYXESH-REOHCLBHSA-N L-Cysteine Chemical compound SC[C@H](N)C(O)=O XUJNEKJLAYXESH-REOHCLBHSA-N 0.000 description 2
- 241001529936 Murinae Species 0.000 description 2
- 108010080146 androgen receptors Proteins 0.000 description 2
- 230000008901 benefit Effects 0.000 description 2
- 230000015556 catabolic process Effects 0.000 description 2
- 230000003833 cell viability Effects 0.000 description 2
- 239000005515 coenzyme Substances 0.000 description 2
- 231100000433 cytotoxic Toxicity 0.000 description 2
- 230000001472 cytotoxic effect Effects 0.000 description 2
- 231100000135 cytotoxicity Toxicity 0.000 description 2
- 230000003013 cytotoxicity Effects 0.000 description 2
- 238000006731 degradation reaction Methods 0.000 description 2
- RGLYKWWBQGJZGM-ISLYRVAYSA-N diethylstilbestrol Chemical compound C=1C=C(O)C=CC=1C(/CC)=C(\CC)C1=CC=C(O)C=C1 RGLYKWWBQGJZGM-ISLYRVAYSA-N 0.000 description 2
- 229960000452 diethylstilbestrol Drugs 0.000 description 2
- 238000009792 diffusion process Methods 0.000 description 2
- 238000009510 drug design Methods 0.000 description 2
- 150000002083 enediols Chemical class 0.000 description 2
- 239000002532 enzyme inhibitor Substances 0.000 description 2
- 102000015694 estrogen receptors Human genes 0.000 description 2
- 108010038795 estrogen receptors Proteins 0.000 description 2
- 238000011156 evaluation Methods 0.000 description 2
- 238000004128 high performance liquid chromatography Methods 0.000 description 2
- 238000000338 in vitro Methods 0.000 description 2
- 230000005764 inhibitory process Effects 0.000 description 2
- 230000014759 maintenance of location Effects 0.000 description 2
- 125000003729 nucleotide group Chemical group 0.000 description 2
- 230000035699 permeability Effects 0.000 description 2
- 239000008363 phosphate buffer Substances 0.000 description 2
- 229920000642 polymer Polymers 0.000 description 2
- 239000011541 reaction mixture Substances 0.000 description 2
- 239000012048 reactive intermediate Substances 0.000 description 2
- 238000011160 research Methods 0.000 description 2
- 150000003839 salts Chemical class 0.000 description 2
- 210000002966 serum Anatomy 0.000 description 2
- 239000000243 solution Substances 0.000 description 2
- 238000001228 spectrum Methods 0.000 description 2
- 231100000419 toxicity Toxicity 0.000 description 2
- 230000001988 toxicity Effects 0.000 description 2
- 239000002023 wood Substances 0.000 description 2
- VHSHLMUCYSAUQU-UHFFFAOYSA-N 2-hydroxypropyl methacrylate Chemical compound CC(O)COC(=O)C(C)=C VHSHLMUCYSAUQU-UHFFFAOYSA-N 0.000 description 1
- VYTPDQSCOXJDJM-UHFFFAOYSA-N 2-methylidenecyclohexan-1-one Chemical class C=C1CCCCC1=O VYTPDQSCOXJDJM-UHFFFAOYSA-N 0.000 description 1
- KYPPJHVEENIEGO-UHFFFAOYSA-N 4-[2-(trifluoromethyl)imidazo[1,2-a]pyridin-3-yl]-n-[4-(trifluoromethyl)phenyl]-1,3-thiazol-2-amine Chemical compound FC(F)(F)C=1N=C2C=CC=CN2C=1C(N=1)=CSC=1NC1=CC=C(C(F)(F)F)C=C1 KYPPJHVEENIEGO-UHFFFAOYSA-N 0.000 description 1
- GCHOCCOURFPPPY-UHFFFAOYSA-N C=C1C(=O)CCCC1C.CC=COCOCC1=CCCCC1=O Chemical compound C=C1C(=O)CCCC1C.CC=COCOCC1=CCCCC1=O GCHOCCOURFPPPY-UHFFFAOYSA-N 0.000 description 1
- FDPXHEJETQGZDK-UHFFFAOYSA-N CCC(C)(CC(C)(C)C(=O)NCC(C)O)C(=O)OCC1=CCCCC1=O Chemical compound CCC(C)(CC(C)(C)C(=O)NCC(C)O)C(=O)OCC1=CCCCC1=O FDPXHEJETQGZDK-UHFFFAOYSA-N 0.000 description 1
- 230000007018 DNA scission Effects 0.000 description 1
- VYZAHLCBVHPDDF-UHFFFAOYSA-N Dinitrochlorobenzene Chemical compound [O-][N+](=O)C1=CC=C(Cl)C([N+]([O-])=O)=C1 VYZAHLCBVHPDDF-UHFFFAOYSA-N 0.000 description 1
- KCXVZYZYPLLWCC-UHFFFAOYSA-N EDTA Chemical compound OC(=O)CN(CC(O)=O)CCN(CC(O)=O)CC(O)=O KCXVZYZYPLLWCC-UHFFFAOYSA-N 0.000 description 1
- 229940122880 Estrogen receptor agonist Drugs 0.000 description 1
- 102000005720 Glutathione transferase Human genes 0.000 description 1
- 108010070675 Glutathione transferase Proteins 0.000 description 1
- 229930189936 Glyoxalase Natural products 0.000 description 1
- 101001016865 Homo sapiens Heat shock protein HSP 90-alpha Proteins 0.000 description 1
- 101001012157 Homo sapiens Receptor tyrosine-protein kinase erbB-2 Proteins 0.000 description 1
- 102000014150 Interferons Human genes 0.000 description 1
- 108010050904 Interferons Proteins 0.000 description 1
- 102000015696 Interleukins Human genes 0.000 description 1
- 108010063738 Interleukins Proteins 0.000 description 1
- 238000006845 Michael addition reaction Methods 0.000 description 1
- 241000699666 Mus <mouse, genus> Species 0.000 description 1
- 241000699670 Mus sp. Species 0.000 description 1
- LZESAMJQHYMINL-UHFFFAOYSA-N O=C1C=CCCC1.O=C1CCCC=C1CO Chemical compound O=C1C=CCCC1.O=C1CCCC=C1CO LZESAMJQHYMINL-UHFFFAOYSA-N 0.000 description 1
- 108091000080 Phosphotransferase Proteins 0.000 description 1
- 206010060862 Prostate cancer Diseases 0.000 description 1
- 208000000236 Prostatic Neoplasms Diseases 0.000 description 1
- 102000001708 Protein Isoforms Human genes 0.000 description 1
- 108010029485 Protein Isoforms Proteins 0.000 description 1
- 102100030086 Receptor tyrosine-protein kinase erbB-2 Human genes 0.000 description 1
- 241000187747 Streptomyces Species 0.000 description 1
- 241000187393 Streptomyces griseosporeus Species 0.000 description 1
- 108060008682 Tumor Necrosis Factor Proteins 0.000 description 1
- 238000009825 accumulation Methods 0.000 description 1
- 239000000556 agonist Substances 0.000 description 1
- 150000001345 alkine derivatives Chemical class 0.000 description 1
- 230000029936 alkylation Effects 0.000 description 1
- 238000005804 alkylation reaction Methods 0.000 description 1
- 235000001014 amino acid Nutrition 0.000 description 1
- 150000001413 amino acids Chemical class 0.000 description 1
- 239000012736 aqueous medium Substances 0.000 description 1
- 238000003556 assay Methods 0.000 description 1
- 230000007321 biological mechanism Effects 0.000 description 1
- 239000008366 buffered solution Substances 0.000 description 1
- 230000023852 carbohydrate metabolic process Effects 0.000 description 1
- 235000021256 carbohydrate metabolism Nutrition 0.000 description 1
- 230000001364 causal effect Effects 0.000 description 1
- 230000003915 cell function Effects 0.000 description 1
- 238000002512 chemotherapy Methods 0.000 description 1
- 230000000052 comparative effect Effects 0.000 description 1
- 238000013270 controlled release Methods 0.000 description 1
- LDHQCZJRKDOVOX-NSCUHMNNSA-N crotonic acid Chemical compound C\C=C\C(O)=O LDHQCZJRKDOVOX-NSCUHMNNSA-N 0.000 description 1
- 239000002254 cytotoxic agent Substances 0.000 description 1
- 231100000599 cytotoxic agent Toxicity 0.000 description 1
- 238000007405 data analysis Methods 0.000 description 1
- 230000001419 dependent effect Effects 0.000 description 1
- 238000001784 detoxification Methods 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 239000003937 drug carrier Substances 0.000 description 1
- 230000008030 elimination Effects 0.000 description 1
- 238000003379 elimination reaction Methods 0.000 description 1
- 229940125532 enzyme inhibitor Drugs 0.000 description 1
- 150000002148 esters Chemical class 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 238000005194 fractionation Methods 0.000 description 1
- 230000006870 function Effects 0.000 description 1
- 230000002538 fungal effect Effects 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 239000013628 high molecular weight specie Substances 0.000 description 1
- 229940088597 hormone Drugs 0.000 description 1
- 239000005556 hormone Substances 0.000 description 1
- 238000007031 hydroxymethylation reaction Methods 0.000 description 1
- 230000006872 improvement Effects 0.000 description 1
- 230000005918 in vitro anti-tumor Effects 0.000 description 1
- 229940047124 interferons Drugs 0.000 description 1
- 229940047122 interleukins Drugs 0.000 description 1
- 238000004989 laser desorption mass spectroscopy Methods 0.000 description 1
- 150000002611 lead compounds Chemical class 0.000 description 1
- 229920002521 macromolecule Polymers 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 238000001869 matrix assisted laser desorption--ionisation mass spectrum Methods 0.000 description 1
- 239000011159 matrix material Substances 0.000 description 1
- 238000000816 matrix-assisted laser desorption--ionisation Methods 0.000 description 1
- 201000001441 melanoma Diseases 0.000 description 1
- 230000000684 melanotic effect Effects 0.000 description 1
- 239000002207 metabolite Substances 0.000 description 1
- OKPYIWASQZGASP-UHFFFAOYSA-N n-(2-hydroxypropyl)-2-methylprop-2-enamide Chemical compound CC(O)CNC(=O)C(C)=C OKPYIWASQZGASP-UHFFFAOYSA-N 0.000 description 1
- JHJNPOSPVGRIAN-SFHVURJKSA-N n-[3-[(1s)-1-[[6-(3,4-dimethoxyphenyl)pyrazin-2-yl]amino]ethyl]phenyl]-5-methylpyridine-3-carboxamide Chemical compound C1=C(OC)C(OC)=CC=C1C1=CN=CC(N[C@@H](C)C=2C=C(NC(=O)C=3C=C(C)C=NC=3)C=CC=2)=N1 JHJNPOSPVGRIAN-SFHVURJKSA-N 0.000 description 1
- 229930014626 natural product Natural products 0.000 description 1
- 239000002773 nucleotide Substances 0.000 description 1
- 230000035515 penetration Effects 0.000 description 1
- 230000004526 pharmaceutical effect Effects 0.000 description 1
- 102000020233 phosphotransferase Human genes 0.000 description 1
- 230000003169 placental effect Effects 0.000 description 1
- 230000008569 process Effects 0.000 description 1
- 208000023958 prostate neoplasm Diseases 0.000 description 1
- 230000009257 reactivity Effects 0.000 description 1
- 238000012552 review Methods 0.000 description 1
- JXOHGGNKMLTUBP-HSUXUTPPSA-N shikimic acid Chemical class O[C@@H]1CC(C(O)=O)=C[C@@H](O)[C@H]1O JXOHGGNKMLTUBP-HSUXUTPPSA-N 0.000 description 1
- 102000034285 signal transducing proteins Human genes 0.000 description 1
- 108091006024 signal transducing proteins Proteins 0.000 description 1
- 150000003384 small molecules Chemical class 0.000 description 1
- 239000012064 sodium phosphate buffer Substances 0.000 description 1
- 241000894007 species Species 0.000 description 1
- 230000002194 synthesizing effect Effects 0.000 description 1
- 238000012360 testing method Methods 0.000 description 1
- 238000002560 therapeutic procedure Methods 0.000 description 1
- 231100000331 toxic Toxicity 0.000 description 1
- 230000002588 toxic effect Effects 0.000 description 1
- LDHQCZJRKDOVOX-UHFFFAOYSA-N trans-crotonic acid Natural products CC=CC(O)=O LDHQCZJRKDOVOX-UHFFFAOYSA-N 0.000 description 1
- 230000001052 transient effect Effects 0.000 description 1
- 230000007704 transition Effects 0.000 description 1
- 230000001960 triggered effect Effects 0.000 description 1
- 102000003390 tumor necrosis factor Human genes 0.000 description 1
- 230000002476 tumorcidal effect Effects 0.000 description 1
- 238000000108 ultra-filtration Methods 0.000 description 1
- 210000005166 vasculature Anatomy 0.000 description 1
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 description 1
- 239000003643 water by type Substances 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/56—Compounds containing cyclopenta[a]hydrophenanthrene ring systems; Derivatives thereof, e.g. steroids
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K5/00—Peptides containing up to four amino acids in a fully defined sequence; Derivatives thereof
- C07K5/02—Peptides containing up to four amino acids in a fully defined sequence; Derivatives thereof containing at least one abnormal peptide link
- C07K5/0215—Peptides containing up to four amino acids in a fully defined sequence; Derivatives thereof containing at least one abnormal peptide link containing natural amino acids, forming a peptide bond via their side chain functional group, e.g. epsilon-Lys, gamma-Glu
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
Definitions
- This invention relates to novel enones that can be used in enone-containing prodrugs for use as cancer therapeutics. It also relates to methods for synthesizing these novel enones and enone-containing prodrugs.
- Gl ⁇ l plays a pivotal role in detoxifying intracellular methylglyoxal, which is formed during normal carbohydrate metabolism (Vander Jagt, D. L. The Glyoxalase System. In Coenzymes and Cofactors: Glutathione ; Dolphin, D.; Poulson, R.; Avramovic, O., Eds.; John Wiley and Sons: New York, 1989; Vol. 3 (part A), pp 597-641.; Creighton, D. J. and Pourmotabbed, T. in Molecular Structure and Energetics: Principles of Enzyme Activity , Liebman, J. F. and Greenberg, A., Eds., VCH Publishers, New York 1988, Vol. 9.
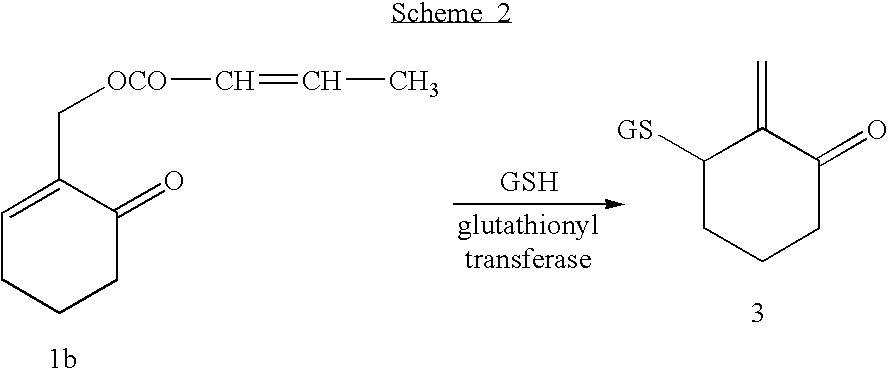
- the endocyclic enone, COMC is a substrate for glutathionyl transferase, an enzyme that is widely distributed in mammalian tissue. This enzyme catalyzes the addition of glutathione to COMC, forming a reactive intermediate that can, in turn, react with, and covalently modify, finctionality on proteins and DNA that may be critical to cell viability.
- An efficient synthesis of COMC has been developed that is superior to the earlier reported seven-step procedure.
- the preferred embodiments of this invention include several new families of endocyclic enones which can be conjugated to various moieties, including sex hormone steroid derivatives, that target cancer cells, thus making these conjugates effective anticancer prodrugs. These new endocyclic enones can also be bound to water-soluble copolymers to modify drug solubility and pharmacokinetics during their delivery to treat cancer.
- FIG. 1 shows the reciprocal plot of the velocity of the glyoxalase I reaction ( ⁇ OD 240 ) versus the concentration of GSH-methylglyoxal-thiohemiacetal (S) in the absence and presence of different concentrations of 2b.
- K diss 2.2 mM.
- FIG. 3 shows the fractionation of product mixtures in the absence and presence of GSTP1-by reverse phase HPLC (Waters, ⁇ -Bondapak C 18 , 0.78 ⁇ 30 cm) after 30 min incubation of reaction mixtures initially composed of 1b (0.1 MM), cysteine (0.5 mm), GSH (0.5 mM), 0.1 M KP i (pH 6.5) and EDTA (0.05 mM) at 25° C.
- FIG. 4 shows the 600 MHz 1 NMR spectrum of the putative exocyclic enone 3.
- the resonance at 7.16 ppm is due to contaminating adduct 2b.
- the other resonances in the spectrum are those expected for S-substituted GSH derivatives.
- FIG. 5 shows the MALDI spectrum of a two hour incubation mixture composed of ApA, GSH (0.5 mM), 1b (0.1 mM) and GST (0.5 units), phosphate buffer (50 mM, pH 7), 25° C.
- COMC is a substrate for glutathionyl transferase, an enzyme that is widely distributed in mammalian tissue.
- Glutathionyl transferase catalyzes a conjugate addition of GSH to 1b with concomitant elimination of crotonic acid leading to 3-glutathionyl-2-exomethylenecyclohexanone 3 (Scheme 2), a reactive intermediate that has now been isolated and characterized for the first time.
- Scheme 2 3-glutathionyl-2-exomethylenecyclohexanone
- the GSH conjugate 2b was prepared from 1b by adapting the procedure for making 2a from 1a (Huntley, C. F. M.; Wood, H. B.; Ganem, B. A New Synthesis of the Glyoxalase-I Inhibitor COTC. Tetrahedron Lett. 2000, 41, 2031-2034).
- the NMR spectrum of 2b featured the expected glutathionyl resonances and the downfield resonance ( ⁇ 7.12, triplet) characteristic of H3 in ⁇ , gamma-unsubstituted 2-cyclohexenones.
- Adduct 2b (0.5 mM) was stable under the reaction conditions, undergoing less than 1% conversion to 4 in the presence of cysteine (2.5 mM) over 27 h, as determined by HPLC.
- cytotoxicity could result from alkylation of DNA and/or proteins critical to cell function.
- the dinucleotides ApA, GpA and CpC were separately incubated with 0.5 mM GSH, 0.1 mM 1b and 0.5 units GSTP1-1. After two hours, the incubation mixtures were analyzed by matrix assisted laser desorption mass spectrometry (MALDI, FIG. 5). In all cases, significant concentrations of nucleotide adducts as well as GSH adducts were detected.
- 1a and 1b are enzyme-activated prodrugs in which the crotonate ester serves as a leaving group, in a process triggered by glutathionyl transferase.
- the preferred embodiments of the invention include several new families of endocyclic enones 1, 6, 7, 8, 9, and 10 which are antitumor prodrugs, and substrates for the enzyme glutathionyl transferase, an enzyme that is widely distributed in mammalian tissue.
- the preferred embodiments also include the derived glutathione adducts 3, 6b, 7b, 8b, 9b, and 10b, which are previously unknown compositions of matter, and which can be prepared, inter alia, using glutathionyl transferase.
- a key novel feature of the invention is the opportunity to take advantage of the prodrug properties of COMC to selectively target certain tumor tissues.
- Preferred embodiments of the invention that demonstrate this feature include several COMC analogs that use hormones to direct cytotoxic agents to targeted tissues.
- GDM Kuduk et al. covalently linked geldanamycin
- the resulting conjugates selectively inhibited prostate cancer cells containing the androgen receptor (Kuduk, S. D., Harris, C., Zheng, F. F., Sepp-Lorenzino, L., Ouerfelli, Q., Rosen, N., and Danishefsky, S. J.
- GDM exerts its toxic effects by binding to the Hsp90 chaperone protein resulting in the degradation of several key signaling proteins.
- testosterone conjugates built on frameworks 1 and 6-10 would also be expected to exhibit selective toxicity towards prostate tumors.
- Kuduk et al. also synthesized conjugates of GDM with estradiol and reported that the hybrid compounds were more selective and active than GDM in causing degradation of the estrogen receptor and HER2, a transmembrane kinase linked to a significant number of breast cancers (Kuduk, S. D., Zheng, F. F., Sepp-Lorenzino, L., Rosen, N., and Danishefsky, S. J. “Synthesis and Evaluation of Geldanamycin-Estradiol Hybrids,” Bioorg. Med. Chem. Lett. 1999, 9, 1233-1238).
- Preferred embodiments of the present invention include agent 11, which hybridizes COMC with diethylstilbestrol, a powerful estrogen receptor agonist.
- Compounds 12-14 are estradiol conjugates that also target against breast cancer.
- the preferred embodiments also include the derived glutathione adducts of these female sex hormone conjugates, which are previously unknown compositions of matter, and which can be prepared, inter alia, using glutathionyl transferase.
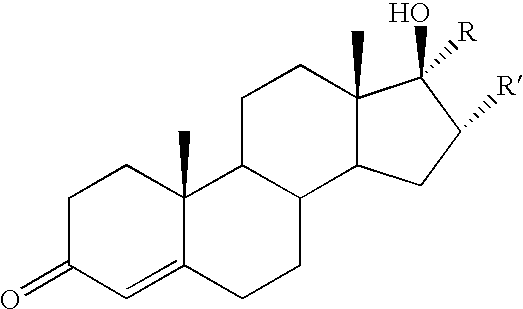
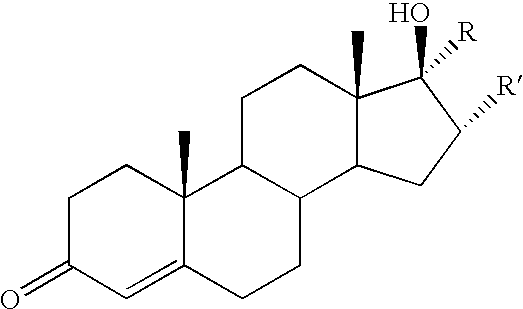
- Another set of preferred embodiments of the invention includes the testosterone-linked conjugates 15-18. These embodiments were based on work of Kuduk et al. indicating that the C17- ⁇ -hydroxyl group of testosterone is required for strong binding to the androgen receptor. Furthermore, conjugates based on C17 ⁇ -linked alkyne tethers displayed the most potent activities.
- Another preferred embodiment of the invention comprises polymeric substances in which COMC molecules are bound to water-soluble N-(2-hydroxypropyl) methacrylamide (HPMA) copolymers.
- HPMA copolymers have been used as drug carriers to modify the solubility and pharmacokinetics of the drug (Lu, Z.-R.; Shiah, J. -G.; Sakuma, S.; Kopeckova, P.; Kopecek, J. Journal of Controlled Release 2002, 78, 165-173).
- the derived copolymer-drug conjugates have been shown to accumulate efficiently in tumor tissue because of the tumor's leaky vasculature and enhanced permeability.
- EPR enhanced permeability and retention
- the conjugate of COMC with the HPMA copolymer shown in structure 19 is subject to the EPR effect, and can release active antitumor agent 3 from the polymer by the action of glutathionyl transferase, which is widely distributed throughout mammalian tissue types.
- These new cancer therapeutics will be manufactured as standard physiologically acceptable salts and derivatives formulated with the appropriate pharmaceutical materials.
- the enones developed can be delivered either alone or in combination with other cancer therapeutics that may enhance their pharmaceutical effects. Methods for delivering the enones and any additional cancer therapeutics combined with it will likely be either oral or intravenous, but other methods of delivery are also possible.
- the components of any combination can be delivered by the same or separate methods and they can be administered simultaneously or separately in time.
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Medicinal Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Genetics & Genomics (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Biochemistry (AREA)
- Biophysics (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Molecular Biology (AREA)
- Epidemiology (AREA)
- Pharmacology & Pharmacy (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
- Medicinal Preparation (AREA)
Abstract
Description
- [0001] This work was supported by National Institutes of Health Grant GM 24054 and by the U.S. Army Medical Research and Materiel Command.
- This invention relates to novel enones that can be used in enone-containing prodrugs for use as cancer therapeutics. It also relates to methods for synthesizing these novel enones and enone-containing prodrugs.
- The search for improved chemotherapeutic agents with novel mechanisms of action is driven by several, important, unmet clinical needs. First and foremost, almost all anticancer drugs cause extreme secondary side effects. Moreover, solid tumors, which are subject to abnormally high interstitial pressure, tend to be resistant to drug penetration by simple diffusion. Since diffusion rates correlate closely with molecular weights, the utility of anticancer strategies that rely on high molecular weight species such as monoclonal antibodies, tumor necrosis factor, interleukins, interferons, and other macromolecules for tumor therapy may ultimately be limited. For that reason, the development of new chemotherapeutic agents and strategies based on small molecules that can be targeted selectively to diseased tissues continues to be an important undertaking. Research over the past several decades indicates that some of the best new lead compounds for cancer chemotherapy have emerged from a better understanding of natural compounds possessing antitumor or anticancer activity.
- In 1975, the fungal metabolite 2-crotonyloxymethyl-(4R, 5R, 6R)-4,5,6-trihydroxy-2-cyclohexenone (COTC, 1a, Scheme 1) was found to display potent antitumor activity in vitro (Takeuchi, T.; Chimura, H.; Hamada, M.; Umezawa, H.; Yoshka, H.; Oguchi, N.; Takahashi, Y.; Matsuda, A. A Glyoxalase I Inhibitor of a New Structural Type Produced by Streptomyces. J. Antibiot. 1975, 28, 737-742). Besides that antitumor activity, it was noted that COTC reacted with reduced glutathione (GSH) to form a new product that inhibited glyoxalase I, a key enzyme in the detoxification of methylglyoxal. In 1975, Chimura et al attributed the antitumor activity of la to its putative glutathione (GSH) adduct 2a, which was proposed to inhibit the enzyme glyoxalase I (Gl×I) (Chimura, H.; Nakamura, H.; Takita, T.; Takeuchi, T.; Umezawa, M.; Kato, K.; Saito, S.; Tomisawa, T.; litaka, Y. The Structure of a Glyoxalase I Inhibitor and Its Chemical Reactivity with SH Compounds. J. Antibiot. 1975, 28, 743-748). In the 25 years since those findings were reported, the inhibition of Gl×I by 2a has come to be associated with its antitumor activity, although no direct evidence supports such a causal relationship, and the hypothesis has never been tested.
- Recently, it has been observed that the simpler COTC analog 1b (COMC) also displayed potent antitumor activity against a range of different murine and human tumors in culture, with 1b being somewhat more potent (IC 50=0.5-19 μM) than 1a (IC50=3-44 μM) (Aghil, O.; Bibby, M. C.; Carrington, S. J.; Doubic, J.; Douglas, K. T.; Phillips, R. M.; Shing, T. K. M. Synthesis and Cytotoxicity of Shikimate Analogues. Structure:Activity Studies Based on 2-Crotonyloxymethyl-3R,4R,5R-trihydroxycyclohex-2-enone. Anti-Cancer Drug Design 1992, 7, 67-82). The endocyclic enone function in these compounds was required for antitumor activity. The antitumor activities of 1b were also thought to result from inhibition of Gl×I by the corresponding GSH adduct 2b. Neither structure 2a nor 2b had been isolated or characterized, and the structures proposed for 2a and 2b were assigned indirectly, on the basis of model experiments with simple thiols.
- Gl×l plays a pivotal role in detoxifying intracellular methylglyoxal, which is formed during normal carbohydrate metabolism (Vander Jagt, D. L. The Glyoxalase System. In Coenzymes and Cofactors: Glutathione; Dolphin, D.; Poulson, R.; Avramovic, O., Eds.; John Wiley and Sons: New York, 1989; Vol. 3 (part A), pp 597-641.; Creighton, D. J. and Pourmotabbed, T. in Molecular Structure and Energetics: Principles of Enzyme Activity, Liebman, J. F. and Greenberg, A., Eds., VCH Publishers, New York 1988, Vol. 9. pp 353-386). Certain inhibitors of human Gl×I, the most potent of which are enediol transition state analogue inhibitors, have been shown to retard the growth of both murine and human tumors in culture and in tumor-bearing mice by causing the accumulation of intracellular methylglyoxal (Kavarana, M. J.; Kovaleva, E. G.; Creighton, D. J.; Wollman, M. B.; Eiseman, J. L. J. Med. Chem. 1999, 42, 221-228; Sharkey, E. M.; O'Neill, H. B.; Kavarana, M. J.; Wang, H.; Creighton, D. J.; Sentz, D. L.; Eiseman, J. L. Cancer Chemother. and Pharmacol. 2000, 46, 156-166).
- While a priori plausible, the hypothesis of Takeuchi et al. for the antitumor action of COTC and COMC has never been tested. Recently, authentic samples of both 2a and 2b were prepared by independent synthesis, and shown to be only modest inhibitors of Gl×I (Huntley, C. F.; Hamilton, D. S.; Creighton, D. J.; Ganem, B. Org. Lett. 2000, 2, 3143-3144; Hamilton, D. S.; Ding, Z.; Ganem, B.; Creighton, D. J. Glutathionyl Transferase Catalyzed Addition of Glutathione to COMC: A New Hypothesis for Antitumor Activity. Org. Lett. accepted and in press) (FIG. 1).
- The endocyclic enone, COMC, is a substrate for glutathionyl transferase, an enzyme that is widely distributed in mammalian tissue. This enzyme catalyzes the addition of glutathione to COMC, forming a reactive intermediate that can, in turn, react with, and covalently modify, finctionality on proteins and DNA that may be critical to cell viability. An efficient synthesis of COMC has been developed that is superior to the earlier reported seven-step procedure. The preferred embodiments of this invention include several new families of endocyclic enones which can be conjugated to various moieties, including sex hormone steroid derivatives, that target cancer cells, thus making these conjugates effective anticancer prodrugs. These new endocyclic enones can also be bound to water-soluble copolymers to modify drug solubility and pharmacokinetics during their delivery to treat cancer.
- Other objects, features, and advantages of the present invention will be apparent from the following Detailed Description of the Preferred Embodiments taken in conjunction with the accompanying drawings in which:
- FIG. 1 shows the reciprocal plot of the velocity of the glyoxalase I reaction (ΔOD 240) versus the concentration of GSH-methylglyoxal-thiohemiacetal (S) in the absence and presence of different concentrations of 2b. In each kinetic run, the concentration of free GSH was maintained at 0.2 mM by varying the total concentration of GSH and methylglyoxal on the basis of the dissociation constant of the hemithioacetal (Kdiss=2.2 mM). Conditions: 50 mM phosphate buffer, pH 7.0, 25° C.
- FIG. 2 shows the spectrophotometrically determined rates of reaction of GSH (1.03 MM) with 1b (0.05 mM) (A) in the absence GST (k=0.070±0.0002 min −) (B) in the presence 1.8 units of GSTP1-1 (k1=0.882±0.055 min−1; k2=0.633±0.053 min−1),and (C) in the presence of 2.4 units of GSTP1-1(k1=1.33±0.037 min−1; k2=0.685±0.024 min−1).
- FIG. 3 shows the fractionation of product mixtures in the absence and presence of GSTP1-by reverse phase HPLC (Waters, μ-Bondapak C 18, 0.78×30 cm) after 30 min incubation of reaction mixtures initially composed of 1b (0.1 MM), cysteine (0.5 mm), GSH (0.5 mM), 0.1 M KPi (pH 6.5) and EDTA (0.05 mM) at 25° C. Based on the integrated intensities of the well-resolved peaks corresponding to 4 (˜9.5 min) and 2b (˜11.5 min), the product ratio in the presence of GSTP1-1 was identical to that obtained from a nonenzymatic incubation of 1b with the same concentrations of cysteine and GSH (FIG. 3). That finding indicated that 3 dissociated from the enzyme prior to reacting with the free thiols in solution, and was consistent with the mechanism in Scheme 4. Adduct 2b (0.5 mM) was stable under the reaction conditions, undergoing less than 1% conversion to 4 in the presence of cysteine (2.5 mM) over 27 h, as determined by HPLC.
- FIG. 4 shows the 600 MHz 1NMR spectrum of the putative exocyclic enone 3. The resonance at 7.16 ppm is due to contaminating adduct 2b. The other resonances in the spectrum are those expected for S-substituted GSH derivatives.
- FIG. 5 shows the MALDI spectrum of a two hour incubation mixture composed of ApA, GSH (0.5 mM), 1b (0.1 mM) and GST (0.5 units), phosphate buffer (50 mM, pH 7), 25° C.
- In order to establish an alternative mode of tumor toxicity for COTC and COMC, we have now demonstrated that COMC is a substrate for glutathionyl transferase, an enzyme that is widely distributed in mammalian tissue. Glutathionyl transferase catalyzes a conjugate addition of GSH to 1b with concomitant elimination of crotonic acid leading to 3-glutathionyl-2-exomethylenecyclohexanone 3 (Scheme 2), a reactive intermediate that has now been isolated and characterized for the first time. Trapping studies with amino acids and nucleotides support the hypothesis that 3 can react with, and covalently modify, functionality on proteins and DNA that may be critical to cell viability.
- In the course of this work, a short and efficient synthesis of 1b was developed that is superior to the earlier reported seven-step procedure (Hamilton, D. S.; Ding, Z.; Ganem, B.; Creighton, D. J.; Org. Lett. accepted and in press). The improved synthesis is depicted in Scheme 3. Baylis-Hillman reaction of 2-cyclohexenone 4 with formaldehyde afforded 2-hydroxymethyl-2-cyclohexenone 5, (Rezgui, F.; El Gaied, M. M. DMAP-Catalyzed Hydroxymethylation of 2-Cyclohexenones in Aqueous Medium Through Baylis Hillman Reaction. Tetrahedron Lett. 1998, 39, 5965-5966) which was then crotonylated to 1b following the literature procedure. (Mirza, S.; Molleyres, L. -P.; Vasella, A. Synthesis of a Glyoxalase I Inhibitor from Streptomyces griseosporeus Niida et Ogasawara. Helv. Chim. Acta 1985, 68, 988-996).
- The GSH conjugate 2b was prepared from 1b by adapting the procedure for making 2a from 1a (Huntley, C. F. M.; Wood, H. B.; Ganem, B. A New Synthesis of the Glyoxalase-I Inhibitor COTC. Tetrahedron Lett. 2000, 41, 2031-2034). The NMR spectrum of 2b featured the expected glutathionyl resonances and the downfield resonance (δ 7.12, triplet) characteristic of H3 in β, gamma-unsubstituted 2-cyclohexenones.
- Kinetic studies with human erythrocyte Gl×I (sodium phosphate buffer, pH 7.0, 25° C., FIG. 1) indicated that 2b was a competitive inhibitor of the enzyme, with a dissociation constant K i=107±0.1 μM. It has previously been reported that 2a competitively inhibited human erythrocyte Gl×I, with a dissociation constant Ki=183±6 μM (Huntley, C. F. M.; Wood, H. B.; Ganem, B. A New Synthesis of the Glyoxalase-I Inhibitor COTC. Tetrahedron Lett. 2000, 41, 2031-2034). To investigate the mechanism of formation of 2b, the nonenzymatic reaction of 1b with GSH was monitored spectrophotometrically, and followed a simple first-order decay (FIG. 2, trace A). No intermediate species was detectable. However, in the presence of human placental glutathione transferase (GSTP1-1) (Predominantly the pi or P1-1 isoform, purchased from Sigma Chemical Company. Salts and free GSH were removed by ultrafiltration. Units of transferase activity were determined using 1-chloro-2,4-dinitrobenzene as substrate (Mannervik, B.; Danielson, U. H. CRC Critical Reviews in Biochemistry 1988, 23, 283-337). The reaction rate profile conformed to a double exponential decay, composed of a rapid, enzyme-dependent, initial phase involving 1b followed by a slower enzyme-independent first-order phase (FIG. 2, traces B and C).
-
- When 1b (0.1 mM) was incubated with cysteine (0.5 mM) and GSH (0.5 mM) in the presence of GSTP 1-1 (1.5 units) for 30 min and the reaction mixture fractionated by reverse phase HPLC, thiol adducts 2b and 4 were isolated. Based on the integrated intensities of the well-resolved peaks corresponding to 4 (˜9.5 min) and 2b (˜11.5 min), the product ratio in the presence of GSTP1-1 was identical to that obtained from a nonenzymatic incubation of 1b with the same concentrations of cysteine and GSH (FIG. 3). That finding indicated that 3 dissociated from the enzyme prior to reacting with the free thiols in solution, and was consistent with the mechanism in Scheme 4. Adduct 2b (0.5 mM) was stable under the reaction conditions, undergoing less than 1% conversion to 4 in the presence of cysteine (2.5 mM) over 27 h, as determined by HPLC.
- Initial rates of reaction of 1a and 1b with hGSTP1-1 were determined from reciprocal plots of initial velocities (ΔOD 235/min), versus [substrate] in buffered solution at pH 6.5, with [GSH]=1 mM (25° C.). Under those conditions, the enzyme-catalyzed GSH addition became rate determining (<0.01 units of transferase in the assay cuvettes). For 1b, kcat=1.2±0.2 s−1 and Km=52±10 μM. For 1a, the individual kinetic constants could not be accurately determined, although kcat/Km was estimated to be 8.3-fold lower than that of 1b.
- Brief incubation of a mixture of 1b, GSH, and GSTP1-1 gave rise to a transient intermediate that could be isolated by reverse-phase HPLC, with a retention time close to that of synthetic 2b. The 600 MHz 1NMR spectrum of the intermediate was consistent with the structure of 3 (FIG. 4). The vinyl proton resonances at 5.76 and 5.29 ppm were characteristic of geminal vinylic hydrogens, and consistent with published NMR spectra of several closely related 2-methylenecyclohexanones (Tamura, R., Watabe, K., Ono, N., Yamamoto, Y. J. Org. Chem. 1992, 57, 4895-4903.) Other resonances in the spectrum corresponded to those expected for the tripeptide moiety (Rabenstein, D. L.; Keire, D. A. in Coenzymes and Cofactors: Glutathione; Dolphin, D.; Poulson, R.; Avramovic, O., Eds.; John Wiley, New York, 1989; Vol 3, Part A, pp.67-101). The resonance at δ 7.12 indicated the presence of 2b, which was formed in the transferase-independent addition of GSH to 3.
- Thus, comparative data analysis suggested that the potent antitumor activities of COTC (1a) and COMC (1b) cannot be rationalized by the action of 2a and 2b, respectively, as weak competitive inhibitors of human erythrocyte Gl×I. Earlier reported enediol analogue inhibitors of Gl×I that inhibited the growth of L1210 and B16 melanotic melanoma in vitro in ester prodrug form exhibited K i values in the submicromolar range (Kavarana, M. J.; Kovaleva, E. G.; Creighton, D. J.; Wollman, M. B.; Eiseman, J. L. Mechanism-Based Competitive Inhibitors of Glyoxalase I: Intracellular Delivery, In Vitro Antitumor Activities, and Stabilities in Human Serum and Mouse Serum. J. Med. Chem. 1999, 42, 221-228). The IC50 values of the prodrugs were approximately proportional to the Ki values of the enzyme inhibitors, such that the weakest enzyme inhibitor (Ki=0.16 μM) gave an IC50 value >100 μM. On this basis, 2b should exhibit very poor antitumor potency, in contrast with the observed antitumor activity of 1b (IC50=0.5-19 μM) (Aghil, O.; Bibby, M. C.; Carrington, S. J.; Double, J.; Douglas, K. T.; Phillips, R. M.; Shing, T. K. M. Anti-Cancer Drug Design 1992, 7, 67-82).
- In principle, cytotoxicity could result from alkylation of DNA and/or proteins critical to cell function. Using COMC to test this hypothesis, the dinucleotides ApA, GpA and CpC were separately incubated with 0.5 mM GSH, 0.1 mM 1b and 0.5 units GSTP1-1. After two hours, the incubation mixtures were analyzed by matrix assisted laser desorption mass spectrometry (MALDI, FIG. 5). In all cases, significant concentrations of nucleotide adducts as well as GSH adducts were detected.
- As indicated above, an alternative biological mechanism has now been established for the tumoricidal activity of both COTC and COMC. It may now be hypothesized that 1a and 1b are enzyme-activated prodrugs in which the crotonate ester serves as a leaving group, in a process triggered by glutathionyl transferase.
- The preferred embodiments of the invention, shown in Scheme 5, include several new families of
endocyclic enones - i, R=OCOCH=CHCH 3; ii, R=OCOCH3; iii, R=OCOC6H5; iv, R=OCONHCH3, v, R=glutathionyl
- A key novel feature of the invention is the opportunity to take advantage of the prodrug properties of COMC to selectively target certain tumor tissues. Preferred embodiments of the invention that demonstrate this feature include several COMC analogs that use hormones to direct cytotoxic agents to targeted tissues. For example, Kuduk et al. covalently linked geldanamycin (GDM) to testosterone, and demonstrated that the resulting conjugates selectively inhibited prostate cancer cells containing the androgen receptor (Kuduk, S. D., Harris, C., Zheng, F. F., Sepp-Lorenzino, L., Ouerfelli, Q., Rosen, N., and Danishefsky, S. J. “Synthesis and Evaluation of Geldanamycin-Testosterone Hybrids,” Bioorg. Med. Chem. Lett. 2000, 10, 1303-1306). GDM exerts its toxic effects by binding to the Hsp90 chaperone protein resulting in the degradation of several key signaling proteins. By analogy, testosterone conjugates built on
frameworks 1 and 6-10 would also be expected to exhibit selective toxicity towards prostate tumors. - Kuduk et al. also synthesized conjugates of GDM with estradiol and reported that the hybrid compounds were more selective and active than GDM in causing degradation of the estrogen receptor and HER2, a transmembrane kinase linked to a significant number of breast cancers (Kuduk, S. D., Zheng, F. F., Sepp-Lorenzino, L., Rosen, N., and Danishefsky, S. J. “Synthesis and Evaluation of Geldanamycin-Estradiol Hybrids,” Bioorg. Med. Chem. Lett. 1999, 9, 1233-1238). In related work, conjugates of enediyne antitumor agents with diethylstilbestrol, a powerful agonist of the estrogen receptor, have been prepared by Jones et al. (Jones, Graham B.; Huber, Robert S.; Matthews, Jude E.; Li, A. “Target Directed Enediyne Prodrugs: Cytotoxic Estrogen Conjugates,” Tetrahedron Lett. 1996, 37, 3643-3646). These conjugates causes DNA strand scission at 10−3 M, and are cytotoxic against the MCF-7 human breast cancer cell line.
- Preferred embodiments of the present invention include agent 11, which hybridizes COMC with diethylstilbestrol, a powerful estrogen receptor agonist. Compounds 12-14 are estradiol conjugates that also target against breast cancer. The preferred embodiments also include the derived glutathione adducts of these female sex hormone conjugates, which are previously unknown compositions of matter, and which can be prepared, inter alia, using glutathionyl transferase.
- Another set of preferred embodiments of the invention includes the testosterone-linked conjugates 15-18. These embodiments were based on work of Kuduk et al. indicating that the C17-β-hydroxyl group of testosterone is required for strong binding to the androgen receptor. Furthermore, conjugates based on C17 α-linked alkyne tethers displayed the most potent activities. The preferred embodiments also include the derived glutathione adducts of these testosterone conjugates, which are previously unknown compositions of matter, and which can be prepared, inter alia, using glutathionyl transferase.
15 R′ = H, R = 16 R′ = H, R = 17 R = H, R′ = 18 R = H, R′ = - Another preferred embodiment of the invention comprises polymeric substances in which COMC molecules are bound to water-soluble N-(2-hydroxypropyl) methacrylamide (HPMA) copolymers. Such HPMA copolymers have been used as drug carriers to modify the solubility and pharmacokinetics of the drug (Lu, Z.-R.; Shiah, J. -G.; Sakuma, S.; Kopeckova, P.; Kopecek, J. Journal of Controlled Release 2002, 78, 165-173). The derived copolymer-drug conjugates have been shown to accumulate efficiently in tumor tissue because of the tumor's leaky vasculature and enhanced permeability. That effect, known as the enhanced permeability and retention (EPR) effect (Maeda, H.; Seymour, L. M.; Miyamoto, Y. Bioconjugate Chem. 1992, 3, 351-362.), makes possible the efficient treatment of solid tumors, provided the surrounding tumor tissue contains enzymes that can release the active antitumor agent from the polymer.
-
- These new cancer therapeutics will be manufactured as standard physiologically acceptable salts and derivatives formulated with the appropriate pharmaceutical materials. The enones developed can be delivered either alone or in combination with other cancer therapeutics that may enhance their pharmaceutical effects. Methods for delivering the enones and any additional cancer therapeutics combined with it will likely be either oral or intravenous, but other methods of delivery are also possible. The components of any combination can be delivered by the same or separate methods and they can be administered simultaneously or separately in time.
- The present invention is illustrated by the previous examples. However, it should be understood that the invention is not limited to the specific details of these examples. It will now be apparent to those skilled in the art that other embodiments, improvements, details, and uses can be made that are consistent with the letter and spirit of the foregoing disclosure and within the scope of this patent and the appended claims.
Claims (47)
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US10/098,834 US20030191066A1 (en) | 2002-03-15 | 2002-03-15 | Enone cancer therapeutics |
| US11/006,132 US7569711B2 (en) | 2002-03-15 | 2004-12-07 | Enone cancer therapeutics |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US10/098,834 US20030191066A1 (en) | 2002-03-15 | 2002-03-15 | Enone cancer therapeutics |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US11/006,132 Continuation US7569711B2 (en) | 2002-03-15 | 2004-12-07 | Enone cancer therapeutics |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| US20030191066A1 true US20030191066A1 (en) | 2003-10-09 |
Family
ID=28673493
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US10/098,834 Abandoned US20030191066A1 (en) | 2002-03-15 | 2002-03-15 | Enone cancer therapeutics |
| US11/006,132 Expired - Fee Related US7569711B2 (en) | 2002-03-15 | 2004-12-07 | Enone cancer therapeutics |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US11/006,132 Expired - Fee Related US7569711B2 (en) | 2002-03-15 | 2004-12-07 | Enone cancer therapeutics |
Country Status (1)
| Country | Link |
|---|---|
| US (2) | US20030191066A1 (en) |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2007081966A2 (en) * | 2006-01-09 | 2007-07-19 | University Of Southern California | Small molecules for treating cancer and abnormal cell proliferation disorders |
-
2002
- 2002-03-15 US US10/098,834 patent/US20030191066A1/en not_active Abandoned
-
2004
- 2004-12-07 US US11/006,132 patent/US7569711B2/en not_active Expired - Fee Related
Also Published As
| Publication number | Publication date |
|---|---|
| US7569711B2 (en) | 2009-08-04 |
| US20050233975A1 (en) | 2005-10-20 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP3991754A1 (en) | Antibody-drug conjugate, intermediate thereof, preparation method therefor and application thereof | |
| EP3380124B1 (en) | Conjugates comprising self-immolative groups and methods related thereto | |
| US8394816B2 (en) | Methods of using [3.2.0] heterocyclic compounds and analogs thereof in treating Waldenstrom's Macroglobulinemia | |
| EP4434549A1 (en) | Antibody-drug conjugate and use thereof | |
| US20080070969A1 (en) | [3.2.0] heterocyclic compounds and methods of using the same | |
| CN111529716B (en) | A kind of polypeptide-paclitaxel conjugate and its application | |
| US20070167353A1 (en) | Prodrug composition | |
| EP4563163A1 (en) | Antibody-drug conjugate and use thereof | |
| EP4458823A1 (en) | Menin inhibitor and use thereof | |
| AU612348B2 (en) | Deuterated aryl aldehydes and derivatives thereof | |
| US7569711B2 (en) | Enone cancer therapeutics | |
| Hartley-Asp et al. | Anti-tumour, toxicological and pharmacokinetic properties of a novel taurine-based nitrosourea (TCNU) | |
| US8586626B2 (en) | Metabolites of wortmannin analogs and methods of using the same | |
| Ottenheijm et al. | 6 Chemical and Biological Aspects of Sparsomycin, an Antibiotic, from Streptomyces | |
| AU722595B2 (en) | Urethane mediated, GST specific molecular release systems | |
| EP3995480A1 (en) | Akr1c3 inhibitor and medical use | |
| Yesair et al. | Pharmacokinetics and metabolism of adriamycin and daunomycin | |
| Kotthaus et al. | Synthesis and biological evaluation of L-valine-amidoximeesters as double prodrugs of amidines | |
| EP4659764A1 (en) | Linker drug, and preparation method and use of antibody-drug conjugate thereof | |
| Karthikeyan et al. | Cytotoxicity of boron containing dipeptide analogs | |
| KR20030019371A (en) | Use of substituted acryloyl distamycin derivatives in the treatment of tumors associated with high levels of glutathione | |
| CN1154499C (en) | 13-Deoxyanthracycline derivatives and processes for preparing them | |
| JP3398255B2 (en) | Novel peptide and therapeutic agent for bone disease containing the same as active ingredient | |
| Pratesi et al. | Antitumor activity of N-trifluoroacetyladriamycin-14-valerate | |
| EP3964236A1 (en) | Ligand-drug conjugate including linker having tris structure |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AS | Assignment |
Owner name: CORNELL RESEARCH FOUNDATION, INC., NEW YORK Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNORS:GANEM, BRUCE;DING, ZHEBO;REEL/FRAME:013046/0931 Effective date: 20020524 |
|
| AS | Assignment |
Owner name: UNITED STATES GOVERNMENT, THE, MARYLAND Free format text: CONFIRMATORY LICENSE;ASSIGNOR:CORNELL UNIVERSITY;REEL/FRAME:013074/0095 Effective date: 20020510 |
|
| STCB | Information on status: application discontinuation |
Free format text: EXPRESSLY ABANDONED -- DURING EXAMINATION |
|
| AS | Assignment |
Owner name: NATIONAL INSTITUTES OF HEALTH (NIH), U.S. DEPT. OF Free format text: EXECUTIVE ORDER 9424, CONFIRMATORY LICENSE;ASSIGNOR:CORNELL UNIVERSITY;REEL/FRAME:021433/0670 Effective date: 20020510 |