KR20190077714A - Lactobacillus plantarum KC28 having anti-obesity effect and uses thereof - Google Patents
Lactobacillus plantarum KC28 having anti-obesity effect and uses thereof Download PDFInfo
- Publication number
- KR20190077714A KR20190077714A KR1020170179135A KR20170179135A KR20190077714A KR 20190077714 A KR20190077714 A KR 20190077714A KR 1020170179135 A KR1020170179135 A KR 1020170179135A KR 20170179135 A KR20170179135 A KR 20170179135A KR 20190077714 A KR20190077714 A KR 20190077714A
- Authority
- KR
- South Korea
- Prior art keywords
- strain
- culture
- lactobacillus plantarum
- obesity
- present
- Prior art date
Links
- 240000006024 Lactobacillus plantarum Species 0.000 title claims abstract description 42
- 235000013965 Lactobacillus plantarum Nutrition 0.000 title claims abstract description 41
- 229940072205 lactobacillus plantarum Drugs 0.000 title claims abstract description 41
- 230000000694 effects Effects 0.000 title claims abstract description 24
- 230000003579 anti-obesity Effects 0.000 title claims abstract description 10
- 239000000203 mixture Substances 0.000 claims description 19
- 239000004480 active ingredient Substances 0.000 claims description 18
- 239000012141 concentrate Substances 0.000 claims description 14
- 239000000047 product Substances 0.000 claims description 14
- 238000000034 method Methods 0.000 claims description 9
- 239000008194 pharmaceutical composition Substances 0.000 claims description 9
- 239000006041 probiotic Substances 0.000 claims description 8
- 235000018291 probiotics Nutrition 0.000 claims description 8
- 230000036541 health Effects 0.000 claims description 7
- 230000000529 probiotic effect Effects 0.000 claims description 7
- 235000013376 functional food Nutrition 0.000 claims description 6
- 238000002360 preparation method Methods 0.000 claims description 6
- 150000001413 amino acids Chemical class 0.000 claims description 5
- 235000013361 beverage Nutrition 0.000 claims description 5
- 239000002243 precursor Substances 0.000 claims description 5
- AHLPHDHHMVZTML-BYPYZUCNSA-N L-Ornithine Chemical compound NCCC[C@H](N)C(O)=O AHLPHDHHMVZTML-BYPYZUCNSA-N 0.000 claims description 3
- HNDVDQJCIGZPNO-YFKPBYRVSA-N L-histidine Chemical compound OC(=O)[C@@H](N)CC1=CN=CN1 HNDVDQJCIGZPNO-YFKPBYRVSA-N 0.000 claims description 3
- KDXKERNSBIXSRK-YFKPBYRVSA-N L-lysine Chemical compound NCCCC[C@H](N)C(O)=O KDXKERNSBIXSRK-YFKPBYRVSA-N 0.000 claims description 3
- OUYCCCASQSFEME-QMMMGPOBSA-N L-tyrosine Chemical compound OC(=O)[C@@H](N)CC1=CC=C(O)C=C1 OUYCCCASQSFEME-QMMMGPOBSA-N 0.000 claims description 3
- KDXKERNSBIXSRK-UHFFFAOYSA-N Lysine Natural products NCCCCC(N)C(O)=O KDXKERNSBIXSRK-UHFFFAOYSA-N 0.000 claims description 3
- 239000004472 Lysine Substances 0.000 claims description 3
- AHLPHDHHMVZTML-UHFFFAOYSA-N Orn-delta-NH2 Natural products NCCCC(N)C(O)=O AHLPHDHHMVZTML-UHFFFAOYSA-N 0.000 claims description 3
- UTJLXEIPEHZYQJ-UHFFFAOYSA-N Ornithine Natural products OC(=O)C(C)CCCN UTJLXEIPEHZYQJ-UHFFFAOYSA-N 0.000 claims description 3
- 150000001412 amines Chemical class 0.000 claims description 3
- HNDVDQJCIGZPNO-UHFFFAOYSA-N histidine Natural products OC(=O)C(N)CC1=CN=CN1 HNDVDQJCIGZPNO-UHFFFAOYSA-N 0.000 claims description 3
- 229960003104 ornithine Drugs 0.000 claims description 3
- OUYCCCASQSFEME-UHFFFAOYSA-N tyrosine Natural products OC(=O)C(N)CC1=CC=C(O)C=C1 OUYCCCASQSFEME-UHFFFAOYSA-N 0.000 claims description 3
- 235000015872 dietary supplement Nutrition 0.000 claims description 2
- 208000008589 Obesity Diseases 0.000 abstract description 22
- 235000020824 obesity Nutrition 0.000 abstract description 22
- 208000030159 metabolic disease Diseases 0.000 abstract description 5
- 238000002474 experimental method Methods 0.000 description 15
- 235000005911 diet Nutrition 0.000 description 13
- 230000037213 diet Effects 0.000 description 13
- 235000013305 food Nutrition 0.000 description 12
- 235000008504 concentrate Nutrition 0.000 description 9
- 102000004190 Enzymes Human genes 0.000 description 8
- 108090000790 Enzymes Proteins 0.000 description 8
- 210000004027 cell Anatomy 0.000 description 8
- 229940088598 enzyme Drugs 0.000 description 8
- 239000002609 medium Substances 0.000 description 8
- 239000000796 flavoring agent Substances 0.000 description 7
- 238000011282 treatment Methods 0.000 description 7
- NTYJJOPFIAHURM-UHFFFAOYSA-N Histamine Chemical compound NCCC1=CN=CN1 NTYJJOPFIAHURM-UHFFFAOYSA-N 0.000 description 6
- 102000004139 alpha-Amylases Human genes 0.000 description 6
- 108090000637 alpha-Amylases Proteins 0.000 description 6
- 235000019634 flavors Nutrition 0.000 description 6
- 230000005764 inhibitory process Effects 0.000 description 6
- JVTAAEKCZFNVCJ-UHFFFAOYSA-N lactic acid Chemical compound CC(O)C(O)=O JVTAAEKCZFNVCJ-UHFFFAOYSA-N 0.000 description 6
- AHLBNYSZXLDEJQ-FWEHEUNISA-N orlistat Chemical compound CCCCCCCCCCC[C@H](OC(=O)[C@H](CC(C)C)NC=O)C[C@@H]1OC(=O)[C@H]1CCCCCC AHLBNYSZXLDEJQ-FWEHEUNISA-N 0.000 description 6
- 239000000126 substance Substances 0.000 description 6
- 238000012360 testing method Methods 0.000 description 6
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 5
- 241000186660 Lactobacillus Species 0.000 description 5
- 241000699666 Mus <mouse, genus> Species 0.000 description 5
- 229940024171 alpha-amylase Drugs 0.000 description 5
- 230000000390 anti-adipogenic effect Effects 0.000 description 5
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 5
- 235000014633 carbohydrates Nutrition 0.000 description 5
- 229940079593 drug Drugs 0.000 description 5
- 239000003814 drug Substances 0.000 description 5
- 229960001031 glucose Drugs 0.000 description 5
- 230000002401 inhibitory effect Effects 0.000 description 5
- 229940039696 lactobacillus Drugs 0.000 description 5
- 239000007788 liquid Substances 0.000 description 5
- 244000005700 microbiome Species 0.000 description 5
- 239000000546 pharmaceutical excipient Substances 0.000 description 5
- 241000894006 Bacteria Species 0.000 description 4
- 102100024295 Maltase-glucoamylase Human genes 0.000 description 4
- 238000010521 absorption reaction Methods 0.000 description 4
- 210000001789 adipocyte Anatomy 0.000 description 4
- 108010028144 alpha-Glucosidases Proteins 0.000 description 4
- 210000000941 bile Anatomy 0.000 description 4
- 230000015572 biosynthetic process Effects 0.000 description 4
- VHRGRCVQAFMJIZ-UHFFFAOYSA-N cadaverine Chemical compound NCCCCCN VHRGRCVQAFMJIZ-UHFFFAOYSA-N 0.000 description 4
- 150000001720 carbohydrates Chemical class 0.000 description 4
- 239000003085 diluting agent Substances 0.000 description 4
- 201000010099 disease Diseases 0.000 description 4
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 4
- 238000009472 formulation Methods 0.000 description 4
- 210000001035 gastrointestinal tract Anatomy 0.000 description 4
- 239000008103 glucose Substances 0.000 description 4
- 239000008187 granular material Substances 0.000 description 4
- 230000003870 intestinal permeability Effects 0.000 description 4
- 210000000936 intestine Anatomy 0.000 description 4
- 238000010150 least significant difference test Methods 0.000 description 4
- 239000006872 mrs medium Substances 0.000 description 4
- 229960001243 orlistat Drugs 0.000 description 4
- 239000000843 powder Substances 0.000 description 4
- KIDHWZJUCRJVML-UHFFFAOYSA-N putrescine Chemical compound NCCCCN KIDHWZJUCRJVML-UHFFFAOYSA-N 0.000 description 4
- 239000003826 tablet Substances 0.000 description 4
- DZGWFCGJZKJUFP-UHFFFAOYSA-N tyramine Chemical compound NCCC1=CC=C(O)C=C1 DZGWFCGJZKJUFP-UHFFFAOYSA-N 0.000 description 4
- 102000019280 Pancreatic lipases Human genes 0.000 description 3
- 108050006759 Pancreatic lipases Proteins 0.000 description 3
- 230000011759 adipose tissue development Effects 0.000 description 3
- 239000002775 capsule Substances 0.000 description 3
- 239000003795 chemical substances by application Substances 0.000 description 3
- 235000020940 control diet Nutrition 0.000 description 3
- 235000013365 dairy product Nutrition 0.000 description 3
- 238000011161 development Methods 0.000 description 3
- 230000018109 developmental process Effects 0.000 description 3
- 206010012601 diabetes mellitus Diseases 0.000 description 3
- 229960001340 histamine Drugs 0.000 description 3
- 230000000968 intestinal effect Effects 0.000 description 3
- 239000004310 lactic acid Substances 0.000 description 3
- 235000014655 lactic acid Nutrition 0.000 description 3
- 238000004519 manufacturing process Methods 0.000 description 3
- 229940116369 pancreatic lipase Drugs 0.000 description 3
- 230000035699 permeability Effects 0.000 description 3
- 210000000813 small intestine Anatomy 0.000 description 3
- QGZKDVFQNNGYKY-UHFFFAOYSA-N Ammonia Chemical compound N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 description 2
- VTYYLEPIZMXCLO-UHFFFAOYSA-L Calcium carbonate Chemical compound [Ca+2].[O-]C([O-])=O VTYYLEPIZMXCLO-UHFFFAOYSA-L 0.000 description 2
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 2
- FBPFZTCFMRRESA-JGWLITMVSA-N D-glucitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-JGWLITMVSA-N 0.000 description 2
- 229920002307 Dextran Polymers 0.000 description 2
- 239000004386 Erythritol Substances 0.000 description 2
- UNXHWFMMPAWVPI-UHFFFAOYSA-N Erythritol Natural products OCC(O)C(O)CO UNXHWFMMPAWVPI-UHFFFAOYSA-N 0.000 description 2
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 2
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 2
- 206010020751 Hypersensitivity Diseases 0.000 description 2
- XUJNEKJLAYXESH-REOHCLBHSA-N L-Cysteine Chemical compound SC[C@H](N)C(O)=O XUJNEKJLAYXESH-REOHCLBHSA-N 0.000 description 2
- CSNNHWWHGAXBCP-UHFFFAOYSA-L Magnesium sulfate Chemical compound [Mg+2].[O-][S+2]([O-])([O-])[O-] CSNNHWWHGAXBCP-UHFFFAOYSA-L 0.000 description 2
- AHLBNYSZXLDEJQ-UHFFFAOYSA-N N-formyl-L-leucylester Natural products CCCCCCCCCCCC(OC(=O)C(CC(C)C)NC=O)CC1OC(=O)C1CCCCCC AHLBNYSZXLDEJQ-UHFFFAOYSA-N 0.000 description 2
- 108010016731 PPAR gamma Proteins 0.000 description 2
- JGSARLDLIJGVTE-MBNYWOFBSA-N Penicillin G Chemical compound N([C@H]1[C@H]2SC([C@@H](N2C1=O)C(O)=O)(C)C)C(=O)CC1=CC=CC=C1 JGSARLDLIJGVTE-MBNYWOFBSA-N 0.000 description 2
- 102100038825 Peroxisome proliferator-activated receptor gamma Human genes 0.000 description 2
- 239000005700 Putrescine Substances 0.000 description 2
- PXIPVTKHYLBLMZ-UHFFFAOYSA-N Sodium azide Chemical compound [Na+].[N-]=[N+]=[N-] PXIPVTKHYLBLMZ-UHFFFAOYSA-N 0.000 description 2
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 2
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 2
- 229930006000 Sucrose Natural products 0.000 description 2
- TVXBFESIOXBWNM-UHFFFAOYSA-N Xylitol Natural products OCCC(O)C(O)C(O)CCO TVXBFESIOXBWNM-UHFFFAOYSA-N 0.000 description 2
- 210000000577 adipose tissue Anatomy 0.000 description 2
- 239000003242 anti bacterial agent Substances 0.000 description 2
- 230000000844 anti-bacterial effect Effects 0.000 description 2
- 229940088710 antibiotic agent Drugs 0.000 description 2
- 230000001580 bacterial effect Effects 0.000 description 2
- 230000008901 benefit Effects 0.000 description 2
- 239000011230 binding agent Substances 0.000 description 2
- 230000003115 biocidal effect Effects 0.000 description 2
- 239000008280 blood Substances 0.000 description 2
- 210000004369 blood Anatomy 0.000 description 2
- 239000000969 carrier Substances 0.000 description 2
- 235000019219 chocolate Nutrition 0.000 description 2
- MYSWGUAQZAJSOK-UHFFFAOYSA-N ciprofloxacin Chemical compound C12=CC(N3CCNCC3)=C(F)C=C2C(=O)C(C(=O)O)=CN1C1CC1 MYSWGUAQZAJSOK-UHFFFAOYSA-N 0.000 description 2
- 235000009508 confectionery Nutrition 0.000 description 2
- 238000007796 conventional method Methods 0.000 description 2
- 230000004069 differentiation Effects 0.000 description 2
- 230000029087 digestion Effects 0.000 description 2
- 150000002016 disaccharides Chemical class 0.000 description 2
- 239000007884 disintegrant Substances 0.000 description 2
- UNXHWFMMPAWVPI-ZXZARUISSA-N erythritol Chemical compound OC[C@H](O)[C@H](O)CO UNXHWFMMPAWVPI-ZXZARUISSA-N 0.000 description 2
- 235000019414 erythritol Nutrition 0.000 description 2
- 229940009714 erythritol Drugs 0.000 description 2
- XJRPTMORGOIMMI-UHFFFAOYSA-N ethyl 2-amino-4-(trifluoromethyl)-1,3-thiazole-5-carboxylate Chemical compound CCOC(=O)C=1SC(N)=NC=1C(F)(F)F XJRPTMORGOIMMI-UHFFFAOYSA-N 0.000 description 2
- 235000021107 fermented food Nutrition 0.000 description 2
- MHMNJMPURVTYEJ-UHFFFAOYSA-N fluorescein-5-isothiocyanate Chemical compound O1C(=O)C2=CC(N=C=S)=CC=C2C21C1=CC=C(O)C=C1OC1=CC(O)=CC=C21 MHMNJMPURVTYEJ-UHFFFAOYSA-N 0.000 description 2
- 235000015203 fruit juice Nutrition 0.000 description 2
- 210000004051 gastric juice Anatomy 0.000 description 2
- 150000004676 glycans Chemical class 0.000 description 2
- 201000001421 hyperglycemia Diseases 0.000 description 2
- 239000004615 ingredient Substances 0.000 description 2
- NOESYZHRGYRDHS-UHFFFAOYSA-N insulin Chemical compound N1C(=O)C(NC(=O)C(CCC(N)=O)NC(=O)C(CCC(O)=O)NC(=O)C(C(C)C)NC(=O)C(NC(=O)CN)C(C)CC)CSSCC(C(NC(CO)C(=O)NC(CC(C)C)C(=O)NC(CC=2C=CC(O)=CC=2)C(=O)NC(CCC(N)=O)C(=O)NC(CC(C)C)C(=O)NC(CCC(O)=O)C(=O)NC(CC(N)=O)C(=O)NC(CC=2C=CC(O)=CC=2)C(=O)NC(CSSCC(NC(=O)C(C(C)C)NC(=O)C(CC(C)C)NC(=O)C(CC=2C=CC(O)=CC=2)NC(=O)C(CC(C)C)NC(=O)C(C)NC(=O)C(CCC(O)=O)NC(=O)C(C(C)C)NC(=O)C(CC(C)C)NC(=O)C(CC=2NC=NC=2)NC(=O)C(CO)NC(=O)CNC2=O)C(=O)NCC(=O)NC(CCC(O)=O)C(=O)NC(CCCNC(N)=N)C(=O)NCC(=O)NC(CC=3C=CC=CC=3)C(=O)NC(CC=3C=CC=CC=3)C(=O)NC(CC=3C=CC(O)=CC=3)C(=O)NC(C(C)O)C(=O)N3C(CCC3)C(=O)NC(CCCCN)C(=O)NC(C)C(O)=O)C(=O)NC(CC(N)=O)C(O)=O)=O)NC(=O)C(C(C)CC)NC(=O)C(CO)NC(=O)C(C(C)O)NC(=O)C1CSSCC2NC(=O)C(CC(C)C)NC(=O)C(NC(=O)C(CCC(N)=O)NC(=O)C(CC(N)=O)NC(=O)C(NC(=O)C(N)CC=1C=CC=CC=1)C(C)C)CC1=CN=CN1 NOESYZHRGYRDHS-UHFFFAOYSA-N 0.000 description 2
- 210000004347 intestinal mucosa Anatomy 0.000 description 2
- 210000001596 intra-abdominal fat Anatomy 0.000 description 2
- 235000021109 kimchi Nutrition 0.000 description 2
- 150000002632 lipids Chemical class 0.000 description 2
- 230000002366 lipolytic effect Effects 0.000 description 2
- 230000007774 longterm Effects 0.000 description 2
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 description 2
- 239000000463 material Substances 0.000 description 2
- 238000005259 measurement Methods 0.000 description 2
- HEBKCHPVOIAQTA-UHFFFAOYSA-N meso ribitol Natural products OCC(O)C(O)C(O)CO HEBKCHPVOIAQTA-UHFFFAOYSA-N 0.000 description 2
- 235000015097 nutrients Nutrition 0.000 description 2
- 229920001282 polysaccharide Polymers 0.000 description 2
- 239000005017 polysaccharide Substances 0.000 description 2
- 230000002265 prevention Effects 0.000 description 2
- 230000008569 process Effects 0.000 description 2
- 108090000623 proteins and genes Proteins 0.000 description 2
- 230000004044 response Effects 0.000 description 2
- 150000003839 salts Chemical class 0.000 description 2
- 239000007787 solid Substances 0.000 description 2
- 235000010356 sorbitol Nutrition 0.000 description 2
- 239000000600 sorbitol Substances 0.000 description 2
- 229960002920 sorbitol Drugs 0.000 description 2
- 239000003381 stabilizer Substances 0.000 description 2
- UCSJYZPVAKXKNQ-HZYVHMACSA-N streptomycin Chemical compound CN[C@H]1[C@H](O)[C@@H](O)[C@H](CO)O[C@H]1O[C@@H]1[C@](C=O)(O)[C@H](C)O[C@H]1O[C@@H]1[C@@H](NC(N)=N)[C@H](O)[C@@H](NC(N)=N)[C@H](O)[C@H]1O UCSJYZPVAKXKNQ-HZYVHMACSA-N 0.000 description 2
- 210000004003 subcutaneous fat Anatomy 0.000 description 2
- 239000005720 sucrose Substances 0.000 description 2
- 235000000346 sugar Nutrition 0.000 description 2
- 150000008163 sugars Chemical class 0.000 description 2
- 230000004083 survival effect Effects 0.000 description 2
- 239000000725 suspension Substances 0.000 description 2
- 235000020357 syrup Nutrition 0.000 description 2
- 239000006188 syrup Substances 0.000 description 2
- 210000001550 testis Anatomy 0.000 description 2
- 229960003732 tyramine Drugs 0.000 description 2
- 235000013343 vitamin Nutrition 0.000 description 2
- 239000011782 vitamin Substances 0.000 description 2
- 229940088594 vitamin Drugs 0.000 description 2
- 229930003231 vitamin Natural products 0.000 description 2
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Chemical compound O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 2
- 229940002552 xenical Drugs 0.000 description 2
- 239000000811 xylitol Substances 0.000 description 2
- 235000010447 xylitol Nutrition 0.000 description 2
- 229960002675 xylitol Drugs 0.000 description 2
- HEBKCHPVOIAQTA-SCDXWVJYSA-N xylitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)CO HEBKCHPVOIAQTA-SCDXWVJYSA-N 0.000 description 2
- YVOOPGWEIRIUOX-BXRBKJIMSA-N (2r)-2-azanyl-3-sulfanyl-propanoic acid Chemical compound SC[C@H](N)C(O)=O.SC[C@H](N)C(O)=O YVOOPGWEIRIUOX-BXRBKJIMSA-N 0.000 description 1
- HSINOMROUCMIEA-FGVHQWLLSA-N (2s,4r)-4-[(3r,5s,6r,7r,8s,9s,10s,13r,14s,17r)-6-ethyl-3,7-dihydroxy-10,13-dimethyl-2,3,4,5,6,7,8,9,11,12,14,15,16,17-tetradecahydro-1h-cyclopenta[a]phenanthren-17-yl]-2-methylpentanoic acid Chemical compound C([C@@]12C)C[C@@H](O)C[C@H]1[C@@H](CC)[C@@H](O)[C@@H]1[C@@H]2CC[C@]2(C)[C@@H]([C@H](C)C[C@H](C)C(O)=O)CC[C@H]21 HSINOMROUCMIEA-FGVHQWLLSA-N 0.000 description 1
- LDVVTQMJQSCDMK-UHFFFAOYSA-N 1,3-dihydroxypropan-2-yl formate Chemical compound OCC(CO)OC=O LDVVTQMJQSCDMK-UHFFFAOYSA-N 0.000 description 1
- OWEGMIWEEQEYGQ-UHFFFAOYSA-N 100676-05-9 Natural products OC1C(O)C(O)C(CO)OC1OCC1C(O)C(O)C(O)C(OC2C(OC(O)C(O)C2O)CO)O1 OWEGMIWEEQEYGQ-UHFFFAOYSA-N 0.000 description 1
- FHVDTGUDJYJELY-UHFFFAOYSA-N 6-{[2-carboxy-4,5-dihydroxy-6-(phosphanyloxy)oxan-3-yl]oxy}-4,5-dihydroxy-3-phosphanyloxane-2-carboxylic acid Chemical compound O1C(C(O)=O)C(P)C(O)C(O)C1OC1C(C(O)=O)OC(OP)C(O)C1O FHVDTGUDJYJELY-UHFFFAOYSA-N 0.000 description 1
- GUBGYTABKSRVRQ-XLOQQCSPSA-N Alpha-Lactose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-XLOQQCSPSA-N 0.000 description 1
- 239000004475 Arginine Substances 0.000 description 1
- 241000193830 Bacillus <bacterium> Species 0.000 description 1
- 206010006187 Breast cancer Diseases 0.000 description 1
- 208000026310 Breast neoplasm Diseases 0.000 description 1
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 1
- 208000024172 Cardiovascular disease Diseases 0.000 description 1
- 102100035882 Catalase Human genes 0.000 description 1
- 108010053835 Catalase Proteins 0.000 description 1
- 206010009944 Colon cancer Diseases 0.000 description 1
- 241000195493 Cryptophyta Species 0.000 description 1
- 229920000858 Cyclodextrin Polymers 0.000 description 1
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 description 1
- 229920001353 Dextrin Polymers 0.000 description 1
- 239000004375 Dextrin Substances 0.000 description 1
- 208000037487 Endotoxemia Diseases 0.000 description 1
- 241000588724 Escherichia coli Species 0.000 description 1
- 102000008946 Fibrinogen Human genes 0.000 description 1
- 108010049003 Fibrinogen Proteins 0.000 description 1
- 239000004606 Fillers/Extenders Substances 0.000 description 1
- 206010016952 Food poisoning Diseases 0.000 description 1
- 208000019331 Foodborne disease Diseases 0.000 description 1
- 229930091371 Fructose Natural products 0.000 description 1
- 239000005715 Fructose Substances 0.000 description 1
- RFSUNEUAIZKAJO-ARQDHWQXSA-N Fructose Chemical compound OC[C@H]1O[C@](O)(CO)[C@@H](O)[C@@H]1O RFSUNEUAIZKAJO-ARQDHWQXSA-N 0.000 description 1
- 241000233866 Fungi Species 0.000 description 1
- 208000018522 Gastrointestinal disease Diseases 0.000 description 1
- 108010010803 Gelatin Proteins 0.000 description 1
- CEAZRRDELHUEMR-URQXQFDESA-N Gentamicin Chemical compound O1[C@H](C(C)NC)CC[C@@H](N)[C@H]1O[C@H]1[C@H](O)[C@@H](O[C@@H]2[C@@H]([C@@H](NC)[C@@](C)(O)CO2)O)[C@H](N)C[C@@H]1N CEAZRRDELHUEMR-URQXQFDESA-N 0.000 description 1
- 229930182566 Gentamicin Natural products 0.000 description 1
- 229920001503 Glucan Polymers 0.000 description 1
- 102000004877 Insulin Human genes 0.000 description 1
- 108090001061 Insulin Proteins 0.000 description 1
- 206010022489 Insulin Resistance Diseases 0.000 description 1
- 239000004201 L-cysteine Substances 0.000 description 1
- 235000013878 L-cysteine Nutrition 0.000 description 1
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 1
- 240000007472 Leucaena leucocephala Species 0.000 description 1
- 235000010643 Leucaena leucocephala Nutrition 0.000 description 1
- 229940127470 Lipase Inhibitors Drugs 0.000 description 1
- 229940086609 Lipase inhibitor Drugs 0.000 description 1
- 241000186779 Listeria monocytogenes Species 0.000 description 1
- GUBGYTABKSRVRQ-PICCSMPSSA-N Maltose Natural products O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@@H]1O[C@@H]1[C@@H](CO)OC(O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-PICCSMPSSA-N 0.000 description 1
- 244000070406 Malus silvestris Species 0.000 description 1
- 241000124008 Mammalia Species 0.000 description 1
- 229930195725 Mannitol Natural products 0.000 description 1
- 229920000168 Microcrystalline cellulose Polymers 0.000 description 1
- 241000699670 Mus sp. Species 0.000 description 1
- 206010028980 Neoplasm Diseases 0.000 description 1
- NPGIHFRTRXVWOY-UHFFFAOYSA-N Oil red O Chemical compound Cc1ccc(C)c(c1)N=Nc1cc(C)c(cc1C)N=Nc1c(O)ccc2ccccc12 NPGIHFRTRXVWOY-UHFFFAOYSA-N 0.000 description 1
- 240000007594 Oryza sativa Species 0.000 description 1
- 235000007164 Oryza sativa Nutrition 0.000 description 1
- 206010033307 Overweight Diseases 0.000 description 1
- 229920002230 Pectic acid Polymers 0.000 description 1
- 239000001888 Peptone Substances 0.000 description 1
- 108010080698 Peptones Proteins 0.000 description 1
- 241000293869 Salmonella enterica subsp. enterica serovar Typhimurium Species 0.000 description 1
- 241000191967 Staphylococcus aureus Species 0.000 description 1
- 229920002472 Starch Polymers 0.000 description 1
- 241000187747 Streptomyces Species 0.000 description 1
- 208000006011 Stroke Diseases 0.000 description 1
- 239000004098 Tetracycline Substances 0.000 description 1
- 244000269722 Thea sinensis Species 0.000 description 1
- JZRWCGZRTZMZEH-UHFFFAOYSA-N Thiamine Natural products CC1=C(CCO)SC=[N+]1CC1=CN=C(C)N=C1N JZRWCGZRTZMZEH-UHFFFAOYSA-N 0.000 description 1
- 241000700605 Viruses Species 0.000 description 1
- 238000009825 accumulation Methods 0.000 description 1
- 239000000654 additive Substances 0.000 description 1
- 230000002293 adipogenic effect Effects 0.000 description 1
- 239000000443 aerosol Substances 0.000 description 1
- 150000001298 alcohols Chemical class 0.000 description 1
- 229940072056 alginate Drugs 0.000 description 1
- 235000010443 alginic acid Nutrition 0.000 description 1
- 229920000615 alginic acid Polymers 0.000 description 1
- 208000026935 allergic disease Diseases 0.000 description 1
- 229910021529 ammonia Inorganic materials 0.000 description 1
- 229960000723 ampicillin Drugs 0.000 description 1
- AVKUERGKIZMTKX-NJBDSQKTSA-N ampicillin Chemical compound C1([C@@H](N)C(=O)N[C@H]2[C@H]3SC([C@@H](N3C2=O)C(O)=O)(C)C)=CC=CC=C1 AVKUERGKIZMTKX-NJBDSQKTSA-N 0.000 description 1
- 239000003392 amylase inhibitor Substances 0.000 description 1
- 230000000845 anti-microbial effect Effects 0.000 description 1
- ODKSFYDXXFIFQN-UHFFFAOYSA-N arginine Natural products OC(=O)C(N)CCCNC(N)=N ODKSFYDXXFIFQN-UHFFFAOYSA-N 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- 230000004888 barrier function Effects 0.000 description 1
- GUBGYTABKSRVRQ-QUYVBRFLSA-N beta-maltose Chemical compound OC[C@H]1O[C@H](O[C@H]2[C@H](O)[C@@H](O)[C@H](O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@@H]1O GUBGYTABKSRVRQ-QUYVBRFLSA-N 0.000 description 1
- 239000003613 bile acid Substances 0.000 description 1
- 230000010234 biliary secretion Effects 0.000 description 1
- 230000000035 biogenic effect Effects 0.000 description 1
- 235000015895 biscuits Nutrition 0.000 description 1
- 235000008429 bread Nutrition 0.000 description 1
- 210000000481 breast Anatomy 0.000 description 1
- ABIUHPWEYMSGSR-UHFFFAOYSA-N bromocresol purple Chemical compound BrC1=C(O)C(C)=CC(C2(C3=CC=CC=C3S(=O)(=O)O2)C=2C=C(Br)C(O)=C(C)C=2)=C1 ABIUHPWEYMSGSR-UHFFFAOYSA-N 0.000 description 1
- 235000012970 cakes Nutrition 0.000 description 1
- 235000001465 calcium Nutrition 0.000 description 1
- 239000011575 calcium Substances 0.000 description 1
- 229910052791 calcium Inorganic materials 0.000 description 1
- 229910000019 calcium carbonate Inorganic materials 0.000 description 1
- 229940041514 candida albicans extract Drugs 0.000 description 1
- 230000023852 carbohydrate metabolic process Effects 0.000 description 1
- 235000021256 carbohydrate metabolism Nutrition 0.000 description 1
- 235000014171 carbonated beverage Nutrition 0.000 description 1
- 210000002421 cell wall Anatomy 0.000 description 1
- 239000001913 cellulose Substances 0.000 description 1
- 235000010980 cellulose Nutrition 0.000 description 1
- 229920002678 cellulose Polymers 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- 235000013351 cheese Nutrition 0.000 description 1
- 238000006243 chemical reaction Methods 0.000 description 1
- 229960005091 chloramphenicol Drugs 0.000 description 1
- WIIZWVCIJKGZOK-RKDXNWHRSA-N chloramphenicol Chemical compound ClC(Cl)C(=O)N[C@H](CO)[C@H](O)C1=CC=C([N+]([O-])=O)C=C1 WIIZWVCIJKGZOK-RKDXNWHRSA-N 0.000 description 1
- 229960003405 ciprofloxacin Drugs 0.000 description 1
- 239000000084 colloidal system Substances 0.000 description 1
- 208000029742 colonic neoplasm Diseases 0.000 description 1
- 239000003086 colorant Substances 0.000 description 1
- 238000004040 coloring Methods 0.000 description 1
- 238000012258 culturing Methods 0.000 description 1
- 230000007123 defense Effects 0.000 description 1
- 230000003111 delayed effect Effects 0.000 description 1
- 230000001627 detrimental effect Effects 0.000 description 1
- 235000019425 dextrin Nutrition 0.000 description 1
- 239000008121 dextrose Substances 0.000 description 1
- 235000014113 dietary fatty acids Nutrition 0.000 description 1
- 235000018823 dietary intake Nutrition 0.000 description 1
- 238000007865 diluting Methods 0.000 description 1
- -1 disaccharide carbohydrates Chemical class 0.000 description 1
- 239000002270 dispersing agent Substances 0.000 description 1
- 239000012153 distilled water Substances 0.000 description 1
- 235000006694 eating habits Nutrition 0.000 description 1
- 230000000062 effect on obesity Effects 0.000 description 1
- 229920001971 elastomer Polymers 0.000 description 1
- 239000003792 electrolyte Substances 0.000 description 1
- 239000000839 emulsion Substances 0.000 description 1
- 239000002158 endotoxin Substances 0.000 description 1
- 239000003623 enhancer Substances 0.000 description 1
- 239000002532 enzyme inhibitor Substances 0.000 description 1
- 210000000981 epithelium Anatomy 0.000 description 1
- 230000029142 excretion Effects 0.000 description 1
- 239000000284 extract Substances 0.000 description 1
- 239000000194 fatty acid Substances 0.000 description 1
- 229930195729 fatty acid Natural products 0.000 description 1
- 150000004665 fatty acids Chemical class 0.000 description 1
- 238000000855 fermentation Methods 0.000 description 1
- 230000004151 fermentation Effects 0.000 description 1
- 235000021001 fermented dairy product Nutrition 0.000 description 1
- 229940012952 fibrinogen Drugs 0.000 description 1
- 239000000945 filler Substances 0.000 description 1
- 235000012041 food component Nutrition 0.000 description 1
- 235000013355 food flavoring agent Nutrition 0.000 description 1
- 239000005417 food ingredient Substances 0.000 description 1
- 230000037406 food intake Effects 0.000 description 1
- 235000012631 food intake Nutrition 0.000 description 1
- 239000003205 fragrance Substances 0.000 description 1
- 239000007789 gas Substances 0.000 description 1
- 229920000159 gelatin Polymers 0.000 description 1
- 239000008273 gelatin Substances 0.000 description 1
- 235000019322 gelatine Nutrition 0.000 description 1
- 235000011852 gelatine desserts Nutrition 0.000 description 1
- 235000011187 glycerol Nutrition 0.000 description 1
- 239000001963 growth medium Substances 0.000 description 1
- 230000008821 health effect Effects 0.000 description 1
- 235000013402 health food Nutrition 0.000 description 1
- 230000005802 health problem Effects 0.000 description 1
- 239000003906 humectant Substances 0.000 description 1
- 230000009610 hypersensitivity Effects 0.000 description 1
- 235000015243 ice cream Nutrition 0.000 description 1
- 230000028993 immune response Effects 0.000 description 1
- 210000000987 immune system Anatomy 0.000 description 1
- 230000006872 improvement Effects 0.000 description 1
- 230000002779 inactivation Effects 0.000 description 1
- 230000006698 induction Effects 0.000 description 1
- 230000001939 inductive effect Effects 0.000 description 1
- 230000002757 inflammatory effect Effects 0.000 description 1
- 239000003112 inhibitor Substances 0.000 description 1
- 229910052500 inorganic mineral Inorganic materials 0.000 description 1
- 235000020344 instant tea Nutrition 0.000 description 1
- 229940125396 insulin Drugs 0.000 description 1
- 230000007413 intestinal health Effects 0.000 description 1
- 238000002955 isolation Methods 0.000 description 1
- 239000008101 lactose Substances 0.000 description 1
- 229960001375 lactose Drugs 0.000 description 1
- OQMAKWGYQLJJIA-CUOOPAIESA-N lipstatin Chemical class CCCCCC[C@H]1[C@H](C[C@H](C\C=C/C\C=C/CCCCC)OC(=O)[C@H](CC(C)C)NC=O)OC1=O OQMAKWGYQLJJIA-CUOOPAIESA-N 0.000 description 1
- 239000006193 liquid solution Substances 0.000 description 1
- 208000019423 liver disease Diseases 0.000 description 1
- 210000005228 liver tissue Anatomy 0.000 description 1
- 235000020845 low-calorie diet Nutrition 0.000 description 1
- 239000000314 lubricant Substances 0.000 description 1
- 235000019359 magnesium stearate Nutrition 0.000 description 1
- 229910052943 magnesium sulfate Inorganic materials 0.000 description 1
- 235000019341 magnesium sulphate Nutrition 0.000 description 1
- 239000000845 maltitol Substances 0.000 description 1
- 235000010449 maltitol Nutrition 0.000 description 1
- VQHSOMBJVWLPSR-WUJBLJFYSA-N maltitol Chemical compound OC[C@H](O)[C@@H](O)[C@@H]([C@H](O)CO)O[C@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1O VQHSOMBJVWLPSR-WUJBLJFYSA-N 0.000 description 1
- 229940035436 maltitol Drugs 0.000 description 1
- 229940099596 manganese sulfate Drugs 0.000 description 1
- 239000011702 manganese sulphate Substances 0.000 description 1
- 235000007079 manganese sulphate Nutrition 0.000 description 1
- SQQMAOCOWKFBNP-UHFFFAOYSA-L manganese(II) sulfate Chemical compound [Mn+2].[O-]S([O-])(=O)=O SQQMAOCOWKFBNP-UHFFFAOYSA-L 0.000 description 1
- 239000000594 mannitol Substances 0.000 description 1
- 235000010355 mannitol Nutrition 0.000 description 1
- 229960001855 mannitol Drugs 0.000 description 1
- 235000013372 meat Nutrition 0.000 description 1
- 210000004379 membrane Anatomy 0.000 description 1
- 239000012528 membrane Substances 0.000 description 1
- 230000004630 mental health Effects 0.000 description 1
- 229920000609 methyl cellulose Polymers 0.000 description 1
- 239000001923 methylcellulose Substances 0.000 description 1
- 235000010981 methylcellulose Nutrition 0.000 description 1
- LXCFILQKKLGQFO-UHFFFAOYSA-N methylparaben Chemical compound COC(=O)C1=CC=C(O)C=C1 LXCFILQKKLGQFO-UHFFFAOYSA-N 0.000 description 1
- 239000008108 microcrystalline cellulose Substances 0.000 description 1
- 235000019813 microcrystalline cellulose Nutrition 0.000 description 1
- 229940016286 microcrystalline cellulose Drugs 0.000 description 1
- 210000000110 microvilli Anatomy 0.000 description 1
- 239000011707 mineral Substances 0.000 description 1
- 235000010755 mineral Nutrition 0.000 description 1
- 239000002480 mineral oil Substances 0.000 description 1
- 235000010446 mineral oil Nutrition 0.000 description 1
- 150000002772 monosaccharides Chemical class 0.000 description 1
- 230000004899 motility Effects 0.000 description 1
- 210000004400 mucous membrane Anatomy 0.000 description 1
- 229930014626 natural product Natural products 0.000 description 1
- 230000004770 neurodegeneration Effects 0.000 description 1
- 208000015122 neurodegenerative disease Diseases 0.000 description 1
- 235000012149 noodles Nutrition 0.000 description 1
- 210000000056 organ Anatomy 0.000 description 1
- 150000007524 organic acids Chemical class 0.000 description 1
- 235000005985 organic acids Nutrition 0.000 description 1
- 230000008520 organization Effects 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- 229910052760 oxygen Inorganic materials 0.000 description 1
- 239000003002 pH adjusting agent Substances 0.000 description 1
- 210000000496 pancreas Anatomy 0.000 description 1
- 244000052769 pathogen Species 0.000 description 1
- 230000001717 pathogenic effect Effects 0.000 description 1
- LCLHHZYHLXDRQG-ZNKJPWOQSA-N pectic acid Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)O[C@H](C(O)=O)[C@@H]1OC1[C@H](O)[C@@H](O)[C@@H](OC2[C@@H]([C@@H](O)[C@@H](O)[C@H](O2)C(O)=O)O)[C@@H](C(O)=O)O1 LCLHHZYHLXDRQG-ZNKJPWOQSA-N 0.000 description 1
- 229940056360 penicillin g Drugs 0.000 description 1
- 235000019319 peptone Nutrition 0.000 description 1
- 239000000825 pharmaceutical preparation Substances 0.000 description 1
- 229940127557 pharmaceutical product Drugs 0.000 description 1
- 235000013550 pizza Nutrition 0.000 description 1
- 239000010318 polygalacturonic acid Substances 0.000 description 1
- 235000010482 polyoxyethylene sorbitan monooleate Nutrition 0.000 description 1
- 229920000053 polysorbate 80 Polymers 0.000 description 1
- 239000001267 polyvinylpyrrolidone Substances 0.000 description 1
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 description 1
- 229920000036 polyvinylpyrrolidone Polymers 0.000 description 1
- 239000003755 preservative agent Substances 0.000 description 1
- QELSKZZBTMNZEB-UHFFFAOYSA-N propylparaben Chemical compound CCCOC(=O)C1=CC=C(O)C=C1 QELSKZZBTMNZEB-UHFFFAOYSA-N 0.000 description 1
- 229960003415 propylparaben Drugs 0.000 description 1
- 230000001681 protective effect Effects 0.000 description 1
- 102000004169 proteins and genes Human genes 0.000 description 1
- NGVDGCNFYWLIFO-UHFFFAOYSA-N pyridoxal 5'-phosphate Chemical compound CC1=NC=C(COP(O)(O)=O)C(C=O)=C1O NGVDGCNFYWLIFO-UHFFFAOYSA-N 0.000 description 1
- 235000007682 pyridoxal 5'-phosphate Nutrition 0.000 description 1
- 239000011589 pyridoxal 5'-phosphate Substances 0.000 description 1
- 229960001327 pyridoxal phosphate Drugs 0.000 description 1
- 208000023504 respiratory system disease Diseases 0.000 description 1
- 235000009566 rice Nutrition 0.000 description 1
- 235000013580 sausages Nutrition 0.000 description 1
- HFHDHCJBZVLPGP-UHFFFAOYSA-N schardinger α-dextrin Chemical compound O1C(C(C2O)O)C(CO)OC2OC(C(C2O)O)C(CO)OC2OC(C(C2O)O)C(CO)OC2OC(C(O)C2O)C(CO)OC2OC(C(C2O)O)C(CO)OC2OC2C(O)C(O)C1OC2CO HFHDHCJBZVLPGP-UHFFFAOYSA-N 0.000 description 1
- 238000012216 screening Methods 0.000 description 1
- 230000035945 sensitivity Effects 0.000 description 1
- 235000011888 snacks Nutrition 0.000 description 1
- 239000011780 sodium chloride Substances 0.000 description 1
- 239000007901 soft capsule Substances 0.000 description 1
- 239000000243 solution Substances 0.000 description 1
- 239000002904 solvent Substances 0.000 description 1
- 235000014347 soups Nutrition 0.000 description 1
- 241000894007 species Species 0.000 description 1
- 238000010186 staining Methods 0.000 description 1
- 235000019698 starch Nutrition 0.000 description 1
- 239000008107 starch Substances 0.000 description 1
- 229940032147 starch Drugs 0.000 description 1
- 238000003860 storage Methods 0.000 description 1
- 229960005322 streptomycin Drugs 0.000 description 1
- 229960004793 sucrose Drugs 0.000 description 1
- 150000005846 sugar alcohols Chemical class 0.000 description 1
- 239000004094 surface-active agent Substances 0.000 description 1
- 239000000375 suspending agent Substances 0.000 description 1
- 230000002459 sustained effect Effects 0.000 description 1
- 208000011580 syndromic disease Diseases 0.000 description 1
- 230000002195 synergetic effect Effects 0.000 description 1
- 238000003786 synthesis reaction Methods 0.000 description 1
- 239000000454 talc Substances 0.000 description 1
- 229910052623 talc Inorganic materials 0.000 description 1
- 235000012222 talc Nutrition 0.000 description 1
- 235000013616 tea Nutrition 0.000 description 1
- 229960002180 tetracycline Drugs 0.000 description 1
- 229930101283 tetracycline Natural products 0.000 description 1
- 235000019364 tetracycline Nutrition 0.000 description 1
- 150000003522 tetracyclines Chemical class 0.000 description 1
- 235000019157 thiamine Nutrition 0.000 description 1
- KYMBYSLLVAOCFI-UHFFFAOYSA-N thiamine Chemical compound CC1=C(CCO)SCN1CC1=CN=C(C)N=C1N KYMBYSLLVAOCFI-UHFFFAOYSA-N 0.000 description 1
- 229960003495 thiamine Drugs 0.000 description 1
- 239000011721 thiamine Substances 0.000 description 1
- 239000002562 thickening agent Substances 0.000 description 1
- 210000001578 tight junction Anatomy 0.000 description 1
- 210000001519 tissue Anatomy 0.000 description 1
- UFTFJSFQGQCHQW-UHFFFAOYSA-N triformin Chemical compound O=COCC(OC=O)COC=O UFTFJSFQGQCHQW-UHFFFAOYSA-N 0.000 description 1
- 239000012137 tryptone Substances 0.000 description 1
- 208000001072 type 2 diabetes mellitus Diseases 0.000 description 1
- 235000013311 vegetables Nutrition 0.000 description 1
- 230000035899 viability Effects 0.000 description 1
- 150000003722 vitamin derivatives Chemical class 0.000 description 1
- 230000004580 weight loss Effects 0.000 description 1
- 239000012138 yeast extract Substances 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N1/00—Microorganisms, e.g. protozoa; Compositions thereof; Processes of propagating, maintaining or preserving microorganisms or compositions thereof; Processes of preparing or isolating a composition containing a microorganism; Culture media therefor
- C12N1/20—Bacteria; Culture media therefor
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L2/00—Non-alcoholic beverages; Dry compositions or concentrates therefor; Their preparation
- A23L2/38—Other non-alcoholic beverages
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L2/00—Non-alcoholic beverages; Dry compositions or concentrates therefor; Their preparation
- A23L2/52—Adding ingredients
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS, OR NON-ALCOHOLIC BEVERAGES, NOT COVERED BY SUBCLASSES A21D OR A23B-A23J; THEIR PREPARATION OR TREATMENT, e.g. COOKING, MODIFICATION OF NUTRITIVE QUALITIES, PHYSICAL TREATMENT; PRESERVATION OF FOODS OR FOODSTUFFS, IN GENERAL
- A23L33/00—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof
- A23L33/10—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof using additives
- A23L33/135—Bacteria or derivatives thereof, e.g. probiotics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K35/00—Medicinal preparations containing materials or reaction products thereof with undetermined constitution
- A61K35/66—Microorganisms or materials therefrom
- A61K35/74—Bacteria
- A61K35/741—Probiotics
- A61K35/744—Lactic acid bacteria, e.g. enterococci, pediococci, lactococci, streptococci or leuconostocs
- A61K35/747—Lactobacilli, e.g. L. acidophilus or L. brevis
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23V—INDEXING SCHEME RELATING TO FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES AND LACTIC OR PROPIONIC ACID BACTERIA USED IN FOODSTUFFS OR FOOD PREPARATION
- A23V2002/00—Food compositions, function of food ingredients or processes for food or foodstuffs
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23V—INDEXING SCHEME RELATING TO FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES AND LACTIC OR PROPIONIC ACID BACTERIA USED IN FOODSTUFFS OR FOOD PREPARATION
- A23V2200/00—Function of food ingredients
- A23V2200/30—Foods, ingredients or supplements having a functional effect on health
- A23V2200/32—Foods, ingredients or supplements having a functional effect on health having an effect on the health of the digestive tract
- A23V2200/3204—Probiotics, living bacteria to be ingested for action in the digestive tract
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23V—INDEXING SCHEME RELATING TO FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES AND LACTIC OR PROPIONIC ACID BACTERIA USED IN FOODSTUFFS OR FOOD PREPARATION
- A23V2400/00—Lactic or propionic acid bacteria
- A23V2400/11—Lactobacillus
- A23V2400/169—Plantarum
-
- A23Y2220/67—
-
- C12R1/25—
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12R—INDEXING SCHEME ASSOCIATED WITH SUBCLASSES C12C - C12Q, RELATING TO MICROORGANISMS
- C12R2001/00—Microorganisms ; Processes using microorganisms
- C12R2001/01—Bacteria or Actinomycetales ; using bacteria or Actinomycetales
- C12R2001/225—Lactobacillus
- C12R2001/25—Lactobacillus plantarum
Abstract
Description
본 발명은 항비만 효과를 갖는 락토바실러스 플란타룸 균주 및 이의 용도에 관한 것으로, 보다 상세하게 항비만(Anti-Obesity) 효과를 갖는 락토바실러스 플란타룸 KC28 균주, 이를 포함하는 프로바이오틱스 제제, 건강기능식품 조성물 및 약학적 조성물에 관한 것이다.The present invention relates to a Lactobacillus plantarum strain having an anti-obesity effect and a use thereof, and more particularly to a Lactobacillus plantarum KC28 strain having an anti-obesity effect, a probiotic preparation containing the same, Food compositions and pharmaceutical compositions.
비만은 대부분의 국가에서 주요한 보건 상의 문제가 되고 있다. 세계보건 기구(WHO)는 전 세계적으로 10억 명 이상의 성인은 과체중이고, 이들 중 3억 명은 임상적으로 비만인 것으로 추정하고 있다(Hotamisligil, 2006). 이러한 비만은 심혈관 질환, 뇌졸중, 인슐린 내성, 당뇨병, 간 질환, 신경퇴행성 질환, 호흡기 질환 및 기타 중증 질환 등에 대한 주요한 위험 인자(Risk factor)이고, 유방암 및 결장암을 포함한 일부 암에 대한 위험인자인 것으로 확인되었다. 또한, 신체 건강에 대한 영향 이외에, 비만은 우울증, 식이장애 및 낮은 자긍심을 초래하여 삶의 질 및 정신 건강에 유해한 영향을 미친다.Obesity is a major health problem in most countries. The World Health Organization (WHO) estimates that over one billion adults worldwide are overweight, and 300 million of them are clinically obese (Hotamisligil, 2006). Such obesity is a major risk factor for cardiovascular disease, stroke, insulin resistance, diabetes, liver disease, neurodegenerative diseases, respiratory diseases and other serious diseases, and is a risk factor for some cancers including breast and colon cancer . In addition, in addition to physical health effects, obesity causes depression, dietary habits, and low self esteem, which have a detrimental effect on quality of life and mental health.
이러한 가운데, 높은 에너지 부하를 갖는 식품 섭취의 꾸준한 증가 및 적은 운동과 연관된 사회적 변화들은 비만 및 비만으로 야기된 대사성 질환의 발생을 증가시키고 있다. 그러나, 저칼로리 다이어트와 운동에 기초한 전통적인 치료는 비만 제어에 있어서 그다지 효과를 나타내지 못하고, 일시적인 체중 손실을 초래할 뿐이다.Among these, the steady increase in food intake with high energy loads and the social changes associated with less exercise increase the incidence of obesity and obesity-induced metabolic diseases. However, conventional treatments based on low-calorie diets and exercises have little effect on obesity control and result in temporary weight loss.
따라서, 항비만 효과를 갖는 약물의 개발이 절실한 실정이다.Therefore, the development of a drug having an anti-obesity effect is urgently needed.
본 발명은 상기와 같은 요구에 의해 도출된 것으로서, 항비만 효과를 가짐으로써, 현대인들의 비만 및 비만으로 야기된 대사성 질환의 발생을 억제하는데 사용될 수 있는 락토바실러스 플란타룸 KC28 균주를 제공한다.The present invention provides a Lactobacillus plantarum KC28 strain which can be used to inhibit the occurrence of metabolic diseases caused by obesity and obesity by having an anti-obesity effect derived from the above-mentioned demand.
여기서, KC28 균주는 김치로부터 분리될 수 있다.Here, the KC28 strain can be isolated from the kimchi.
본 발명의 일 실시예에 따른 락토바실러스 플란타룸 KC28 균주(기탁번호: KCTC13377BP)는 항비만 효과를 갖는 것을 특징으로 한다.The Lactobacillus plantarum KC28 strain (Accession No .: KCTC13377BP) according to an embodiment of the present invention is characterized by having an anti-obesity effect.
상기 균주는 티로신, 히스티딘, 오르니틴 및 라이신으로 이루어진 군에서 선택되는 하나 이상의 아미노산 전구체로부터 생체 아민을 생성하지 않는 것을 특징으로 한다.The strain is characterized in that it does not produce a biological amine from one or more amino acid precursors selected from the group consisting of tyrosine, histidine, ornithine and lysine.
본 발명의 실시예는, 상기 균주, 상기 균주의 배양물, 상기 배양물의 농축액 및 건조물로 이루어지는 군에서 선택되는 하나 이상을 유효성분으로 포함하는 프로바이오틱스 제제를 제공한다.An embodiment of the present invention provides a probiotic preparation comprising, as an active ingredient, at least one selected from the group consisting of the strain, the culture of the strain, the concentrate of the culture, and the dried product.
본 발명의 실시예는, 상기 균주, 상기 균주의 배양물, 상기 배양물의 농축액 및 건조물로 이루어지는 군에서 선택되는 하나 이상을 유효성분으로 포함하는 건강기능식품 조성물을 제공한다.An embodiment of the present invention provides a health functional food composition comprising, as an active ingredient, at least one selected from the group consisting of the strain, the culture of the strain, the concentrate of the culture, and the dried product.
본 발명의 실시예는, 상기 균주, 상기 균주의 배양물, 상기 배양물의 농축액 및 건조물로 이루어지는 군에서 선택되는 하나 이상을 유효성분으로 포함하는 약학적 조성물을 제공한다.An embodiment of the present invention provides a pharmaceutical composition comprising, as an active ingredient, at least one selected from the group consisting of the strain, the culture of the strain, the concentrate of the culture, and the dried material.
본 발명의 실시예는, 균주, 상기 균주의 배양물 및 상기 배양물의 농축액 또는 건조물 중 적어도 하나를 유효 성분으로 포함하는 음료를 제공한다.An embodiment of the present invention provides a beverage comprising at least one of a strain, a culture of the strain and a concentrate or a dried product of the culture as an active ingredient.
본 발명의 실시예는, 상기 균주, 상기 균주의 배양물 및 상기 배양물의 농축액 또는 건조물 중 적어도 하나를 유효 성분으로 포함하는 건강 보조 식품을 제공한다. An embodiment of the present invention provides a health supplement containing, as an active ingredient, at least one of the strain, a culture of the strain, and a concentrate or a dried product of the culture.
본 발명의 실시예들에 따르면, 락토바실러스 플란타룸 KC28 균주는 항비만 효과를 가짐으로써, 현대인들의 비만 및 비만으로 야기된 대사성 질환의 발생을 억제하는데 사용될 수 있다.According to the embodiments of the present invention, Lactobacillus plantarum KC28 strain has an anti-obesity effect, so that it can be used to suppress the occurrence of metabolic diseases caused by obesity and obesity in modern humans.
도 1은 본 발명의 락토바실러스 플란타룸 KC28 균주에 대한 Anti-adipogenic activity 결과를 나타낸 도면이다.
도 2는 본 발명의 락토바실러스 플란타룸 KC28 균주에 대한 담즙내성 실험 결과를 나타낸 도면이다.
도 3은 본 발명의 락토바실러스 플란타룸 KC28 균주에 대한 pH 내성 실험 결과를 나타낸 도면이다.
도 4는 본 발명의 락토바실러스 플란타룸 KC28 균주에 대한 장 정착능 실험 결과를 나타낸 도면이다.
도 5는 비만 유도 마우스의 12 주간의 무게에 대한 실험 결과를 나타낸 도면이다.
도 6은 마우스 실험 완료 후 적출한 정소지방 (A), 피하지방 (B), 및 내장지방 (C)의 무게 차이를 나타낸 도면이다.
도 7은 Fluorescein isothiocyanate conjugated dextran (FITC-dextran) (Sigma-Aldrich)를 이용하여 장 투과성을 측정한 결과를 나타낸 도면이다.FIG. 1 is a graph showing the anti-adipogenic activity of the Lactobacillus plantarum KC28 strain of the present invention.
FIG. 2 is a graph showing the results of an experiment of bile resistance against Lactobacillus plantarum KC28 strain of the present invention. FIG.
FIG. 3 is a graph showing the results of the pH resistance test on the Lactobacillus plantarum KC28 strain of the present invention.
FIG. 4 is a graph showing the results of an experiment for the long-term fixation of Lactobacillus plantarum KC28 strain of the present invention.
5 is a graph showing the results of an experiment on the weight of the obesity-induced mouse for 12 weeks.
FIG. 6 is a graph showing the weight difference between the testis fat (A), subcutaneous fat (B), and visceral fat (C) extracted after completion of the mouse experiment.
FIG. 7 is a graph showing the results of measurement of intestinal permeability using Fluorescein isothiocyanate conjugated dextran (FITC-dextran) (Sigma-Aldrich).
이하, 본 발명에 따른 실시예들을 첨부된 도면을 참조하여 상세하게 설명한다. 그러나 본 발명이 실시예들에 의해 제한되거나 한정되는 것은 아니며, 당업자는 이러한 실시예들을 용이하게 변경할 수 있다. 또한, 각 도면에 제시된 동일한 참조 부호는 동일한 부재를 나타낸다.Hereinafter, embodiments according to the present invention will be described in detail with reference to the accompanying drawings. However, the present invention is not limited or limited by the embodiments, and those skilled in the art can easily modify these embodiments. In addition, the same reference numerals shown in the drawings denote the same members.
또한, 본 명세서에서 사용되는 용어(terminology)들은 본 발명의 바람직한 실시예를 적절히 표현하기 위해 사용된 용어들로서, 이는 시청자, 운용자의 의도 또는 본 발명이 속하는 분야의 관례 등에 따라 달라질 수 있다. 따라서, 본 용어들에 대한 정의는 본 명세서 전반에 걸친 내용을 토대로 내려져야 할 것이다.Also, terminologies used herein are terms used to properly represent preferred embodiments of the present invention, which may vary depending on the viewer, the intention of the operator, or the custom in the field to which the present invention belongs. Therefore, the definitions of these terms should be based on the contents throughout this specification.
본 발명의 락토바실러스 플란타룸 KC28 균주에 대한 실험에 대해 간략하게 설명하면 다음과 같다.Experiments on the Lactobacillus plantarum KC28 strain of the present invention will be briefly described as follows.
Alpha-amylase 저해 활성 실험Alpha-amylase inhibitory activity experiment
최근에는 비만치료제 개발을 위한 많은 연구가 진행되고 있으며, 그 중 하나가 α-amylase 저해 실험이다 (α-amylase inhibition test). α-amylase는 탄수화물의 α-D-(1,4)-glucan 결합을 분해하는 효소이고, 탄수화물 대사에 있어서 중요한 효소이다. 현재 과혈당증, 당뇨병 및 인슐린 관련 대사 질환 연구를 위해서는 당의 소화 및 흡수를 제어할 필요가 있고, 이때 α-amylase 효소에 대한 활성 저해에 관한 연구가 많이 수행되고 있다. Recently, many studies for the development of obesity treatment drugs have been conducted, one of which is the α-amylase inhibition test. α-amylase is an enzyme that degrades the α-D- (1,4) -glucan bond of carbohydrates and is an important enzyme in carbohydrate metabolism. Currently, studies on hyperglycemia, diabetes, and insulin-related metabolic diseases require control of digestion and absorption of sugars, and studies on the inhibition of α-amylase enzyme activity have been conducted.
α-α- GlucosidaseGlucosidase 저해 활성 실험 Inhibition activity experiment
α-Glucosidase는 소장 상피세포에 존재하는 효소로서, 다당류 및 이당류 탄수화물의 소화 및 흡수를 위해 단당류로 분해하는 역할을 한다. α-amylase와 비슷하게 α-glucosidase 활성을 저해하면 혈당 상승이 억제될 수 있으며, 이러한 원리를 이용하여 과혈당증 및 당뇨병에 대한 활성을 측정할 수 있다. α-Glucosidase is an enzyme present in the small intestine epithelium, which acts as a disaccharide for the digestion and absorption of polysaccharides and disaccharide carbohydrates. Similar to α-amylase, inhibition of α-glucosidase activity may inhibit blood glucose elevation, and the activity against hyperglycemia and diabetes can be measured using this principle.
Pancreatic lipase 저해 활성 실험Pancreatic lipase inhibitory activity experiment
췌장 지방분해효소(pancreatic lipase)는 트리글리세라이드(triglyceride)를 2-모노아실글리세롤(2-monoacylglycerol)과 지방산(fatty acid)으로 분해하는 핵심 효소로 작용한다[Bitou, N., M. Nimomiya, T. Tsjita and H. Okuda. 1999. Screening of lipase inhibitors from marine algae. Lipids 34, 441-445]. 대표적인 췌장 지방분해소 저해제는 Streptomyces toxitricini로부터 유래된 lipstatin의 유도체인 tetrahydrolipstatin (Orlistat)으로서 섭취된 지방의 약 30%를 저해할 정도로 효능이 우수한 것으로 알려져 있으며[Drent, M. L., et al., 1995. Orlistat (RO 18-0647), a lipase inhibitor, in the treatment of human obesity: a multiple dose study. Int. J. Obes. 19, 221 -226.], 현재 의약품으로도 시판중이다. 그러나 이와 같은 효능에도 불구하고 tetrahydrolipstatin은 위장장애, 과민증, 담즙분비장애, 지용성 비타민 흡수억제등의 부작용이 있는 것으로 알려져 있다. 따라서 최근에는 부작용이 없는 식품 및 천연물로부터 췌장 지방분해효소 저해제의 개발을 위한 연구가 진행되고 있다.Pancreatic lipase acts as a key enzyme that breaks down triglyceride into 2-monoacylglycerol and fatty acid [Bitou, N., M. Nimomiya, T. Tsjita and H. Okuda. 1999. Screening of lipase inhibitors from marine algae. Lipids 34, 441-445]. Representative pancreatic lipolytic inhibitors are tetrahydrolipstatin (Orlistat), a derivative of lipstatin derived from Streptomyces toxitricini , which is known to be effective in inhibiting about 30% of the fat ingested [Drent, ML, et al., 1995. Orlistat RO 18-0647), a lipase inhibitor, in the treatment of human obesity: a multiple dose study. Int. J. Obes. 19, 221 -226.], And is currently also being marketed as a pharmaceutical product. Despite these benefits, however, tetrahydrolipstatin has been known to have side effects such as gastrointestinal disorders, hypersensitivity, biliary secretion, and lipid-soluble vitamin absorption inhibition. Recently, studies for the development of pancreatic lipolytic enzyme inhibitors from foods and natural products without side effects have been carried out.
Anti-Anti- adipogenicadipogenic activity 실험 activity experiment
고지방 식이 섭취로 인한 불균형이 초래하는 비만은 체내 지방의 과잉축적이 가장 큰 특징이고 지방 세포의 형성(adipogenesis)이 이뤄진다. 지방세포 형성 과정인 adipogenesis에 핵심적 역할을 하는 것이 peroxisome proliferator activated receptor gamma (PPARγ)는 섭취되는 지방에 반응해 간이나 지방조직에서 발현이 되는데, 이는 지방세포로 분화되는데 필요한 유전자 및 지방세포의 지질 합성 및 저장에 요구되는 단백질들로 발현되는 유전자들을 작동시키는 역할을 한다. 즉, anti-adipogenic activity는 균 대상으로 지방세포 분화 활성을 측정하는데, Oil Red O relative staining 기법으로 지방세포가 얼만큼 분해가 되었는지 측정하는 방법이다. Obesity caused by imbalance caused by high fat dietary intake is characterized by excessive accumulation of body fat and adipogenesis. The peroxisome proliferator activated receptor gamma (PPARγ), which plays a key role in the adipogenesis process of adipogenesis, is expressed in liver and adipose tissue in response to the ingested fat, which is necessary for differentiation into adipocytes and lipid synthesis And the genes that are expressed as proteins required for storage. In other words, anti-adipogenic activity is a method to measure the adipocyte differentiation activity as a fungus, and to measure how much fat cells are degraded by Oil Red O relative staining technique.
담즙 내성 및 pH 내성 실험Bile tolerance and pH resistance test
위액과 췌장이서 분비되는 담즙산과 pH가 미생물의 생존 저해에 영향을 미치는 아주 중요한 factor이다. 따라서 미생물이 생존 저해 환경에 대하여 얼마나 내성이 있는지 측정하는 방법이다. 인공위액과 담즙에 대한 내성을 조사하여 probiotics로서의 가능성을 검토하고, 이들 중 활성이 우수한 그리고 내성이 강한 균주를 선발 및 동정하는 과정이다. Bile acid and pH, which are secreted from gastric juice and pancreas, are very important factors affecting the viability of microorganisms. Therefore, it is a method to measure how tolerant a microorganism is to the survival inhibition environment. We investigated the possibility of probiotics by investigating the resistance to artificial gastric juice and bile, and selecting and isolating strains with high activity and resistance.
항균력 실험Experiment of antibacterial activity
항균력 실험은 에스케리키아 콜리 (Escherichia coli), 살모넬라 타이피머리움 (Salmonella typhimurium), 스테피로코커스 오레우스 (Staphylococcus aureus), 리스테리아 모노사이토제네스 (Listeria monocytogenes)에 대하여 억제력이 얼만큼 있는지 확인하는 실험이다. 유해균주에 대한 억제력이 좋으면 좋을수록 좋다. The antimicrobial activity was determined by Escherichia coli , Salmonella typhimurium , Staphylococcus aureus , Listeria monocytogenes , and the like. In the experiment, The better the deterrent against harmful strains, the better.
장 누수 실험 (Gut permeability test)Gut permeability test
우리가 섭취하는 음식물들은 장내 미생물 및 여러 효소에 의해 분해되어 소장에서 영양소를 소화 및 흡수한다. 지금까지 장은 단순히 소화 및 흡수하는 기관으로 여겼지만, 사실은 장은 외부에서 들어오는 유해물질로부터 1차 방어막 역할을 하고 있으며, 각종 논문에서 우리 몸의 70%-80% 정도로 큰 면역기관이라는 사실이 알려지면서 장 건강의 중요성이 부각되었다. 정상적인 장은 유해균 및 유해물질이 몸 안으로 들어오지 못하게 점막으로 1차 방어막 구실을 한다. 그러나 장 누수 증후군 (Leaky gut)은 장관 세포가 손상을 입고, 소장 내벽에는 미세 융모가 있는데, 그 세포들의 결합 부위에 틈이 생겨, 여러 종류의 유해물질이 그 사이로 통과되고, 혈액 내로 직접 운반되어 여러가지 질병의 원인이 된다. The foods we eat are digested by the intestinal microorganisms and enzymes, digesting and absorbing the nutrients in the small intestine. Until now, the intestines have been regarded as simply digesting and absorbing organs, but in fact, it is known that the intestines are the primary defense against harmful substances coming in from the outside, and that in various articles, the immune system is 70% to 80% The importance of intestinal health emerged. The normal intestine serves as a primary barrier to the mucous membranes, preventing harmful bacteria and harmful substances from entering the body. However, the Leaky gut is damaged by intestinal cells, and there is a microvilli on the inner wall of the small intestine. There are gaps in the binding sites of the cells, various kinds of harmful substances pass through them, and they are transported directly into the blood. It causes various diseases.
본 발명의 락토바실러스 플란타룸 KC28 균주에 대한 미생물 분리 및 동정 과정은, 본 발명자들이 각각 지역과 종류가 다른 김치를 수거하여 사용하였으며, 펩톤(peptone) 희석액으로 희석하여 브롬크레졸 퍼플(Bromcresol purple)과 소듐아자이드(sodium azide)를 첨가한 변형 MRS 배지에 0.1 ㎖ 씩 평면도말법으로 접종한 후 37에서 48시간 배양하고 균락을 변형 MRS 배지에서 순수 분리한 다음 노란색으로 변한 균락을 잠정적 젖산균으로 선발하였다. 선발된 균주는 변형 MRS 배지에 도말한 후, 호기 배양하여 순수분리 하였다. 순수 분리된 균주를 동정한 결과, 균주는 그람 양성의 간균이며, 산소유무와 상관없이 잘 생장하고 카탈라아제와 운동성에 대해서는 음성으로 확인되었다. 또한, 15와 45에서 생장하지 않았으며, 글루코오스(glucose)로부터 가스와 알기닌으로부터 암모니아를 생성하지 않아 락토바실러스(Lactobacillus) 속(genus)에 속하는 것으로 확인하였다.The microorganism isolation and identification process for the Lactobacillus plantarum KC28 strain of the present invention was performed by collecting and using kimchi having different types and regions from each of the present inventors and diluting it with a peptone diluent to obtain Bromcresol purple, And sodium azide were added to the transformed MRS medium. The culture broth was inoculated with 0.1 ml of each culture medium at 37 ° C for 48 hours. The transformant broth was purified from the transformed MRS medium and then transformed into yellow . The selected strains were plated on transformed MRS medium and then cultured aerobically to isolate pure strains. As a result of the identification of pure isolates, the strain was Gram positive bacillus and showed good growth regardless of presence or absence of oxygen and negative for catalase and motility. In addition, it did not grow at 15 and 45, and it was confirmed that it does not produce ammonia from gas and arginine from glucose and belongs to the genus of Lactobacillus .
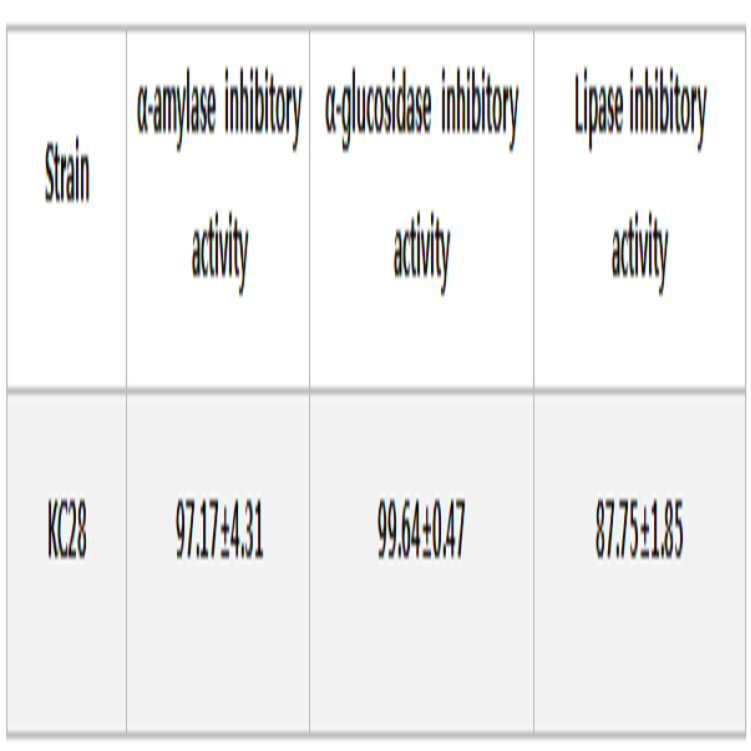
본 발명의 락토바실러스 플란타룸 KC28 균주에 대한 α-amylase, α-glucosidase, pancreatic lipase inhibition 결과는 아래 <표 1>과 같다.The results of α-amylase, α-glucosidase, and pancreatic lipase inhibition on Lactobacillus plantarum KC28 strain of the present invention are shown in Table 1 below.
도 1은 본 발명의 락토바실러스 플란타룸 KC28 균주에 대한 Anti-adipogenic activity 결과를 나타낸 도면이며, 아래 표 2는 락토바실러스 플란타룸 KC28 균주에 대한 Anti-adipogenic activity 결과 표를 나타낸 것이다.FIG. 1 is a graph showing the results of anti-adipogenic activity against Lactobacillus plantarum KC28 strain of the present invention, and Table 2 below shows the results of anti-adipogenic activity against Lactobacillus plantarum KC28 strain.
도 1과 표 2에 도시된 바와 같이, 락토바실러스 플란타룸 KC28 균주가 지방전구세포의 지방세포 분화 활성(Anti-adipogenic activity)을 36.99% 억제하는 것을 알 수 있다. 즉, 락토바실러스 플란타룸 KC28 균주의 지방세포 억제 활성이 우수한 것을 알 수 있다.As shown in Fig. 1 and Table 2, it was found that the Lactobacillus plantarum KC28 strain inhibited the adipogenic activity of fat precursor cells by 36.99%. In other words, it can be seen that the Lactobacillus plantarum KC28 strain has an excellent adipocyte inhibitory activity.
지방세포 분화 억제능을 Oil Red O staining을 통해 분석한 것으로, 모든 수치 또는 값은 Fisher's LSD test 사후 검정 후 p<0.05 일 때 *로 표기하고, p<0.01 일 때 **로 표기하며, p<0.001 일 때 ***를 표기한다.All values or values were expressed as * when p <0.05 after Fisher's LSD test, ** when p <0.01, p <0.001 When *** is marked.
도 2는 본 발명의 락토바실러스 플란타룸 KC28 균주에 대한 담즙내성 실험 결과를 나타낸 도면이다.FIG. 2 is a graph showing the results of an experiment of bile resistance against Lactobacillus plantarum KC28 strain of the present invention. FIG.
보다 상세하게는, 락토바실러스 플란타룸 KC28 균주는 0.03%의 담즙(oxgall)과 0.05%의 엘-시스테인(L-cysteine)을 포함하는 MRS 배지(with oxgall)와 0.05%의 엘-시스테인(L-cysteine)을 포함하는 MRS 배지(without oxgall)에서 성장되었으며, 모든 수치(또는 값)는 3회 반복의 평균±표준편차이며, *는 with oxgall와 without oxgall 간 p<0.05인 경우를 표기한 것이다.More specifically, the Lactobacillus plantarum KC28 strain contains an MRS medium (with oxgall) containing 0.03% of oxgall and 0.05% of L-cysteine and 0.05% of L-cysteine -cysteine), all values (or values) were the mean ± standard deviation of three replicates, and * indicates the case of p <0.05 between with oxgall and without oxgall .
도 3은 본 발명의 락토바실러스 플란타룸 KC28 균주에 대한 pH 내성 실험 결과를 나타낸 도면이다.FIG. 3 is a graph showing the results of the pH resistance test on the Lactobacillus plantarum KC28 strain of the present invention.
보다 상세하게는, pH가 2.0, 3.0, 4.0 및 6.4인 염산 용역에서 3시간 후 락토바실러스 플란타룸 KC28 균주의 생존률을 나타낸 것으로, 시작 시점(또는 시작 시간)과 비교하여 *은 p<0.05인 경우를 표기한 것이고, **은 p<0.01인 경우를 표기한 것이며, ***은 p<0.001을 표기한 것이다.More specifically, the survival rate of the Lactobacillus plantarum KC28 strain after three hours in hydrochloric acid at pH 2.0, 3.0, 4.0, and 6.4 is shown, and * is p <0.05 compared to the starting point (or start time) ** indicates the case of p <0.01, and *** indicates the case of p <0.001.
아래 표 3은 락토바실러스 플란타룸 KC28 균주에 대한 항균력 실험 결과를 나타낸 것이며, 락토바실러스 플란타룸 KC28 균주의 초기 균수는 6.23±0.40 × 106 CFU/mL이며, 37에서 6시간 후에 대한 결과를 나타낸 것으로, 모든 수치(또는 값)는 3회 반복의 평균±표준편차이다.Table 3 below shows the results of the antibacterial activity test for Lactobacillus plantarum KC28 strain. The initial number of bacteria of Lactobacillus plantarum KC28 strain was 6.23 ± 0.40 × 10 6 CFU / mL, and the result after 6 hours at 37 All values (or values) are mean ± standard deviation of three replicates.
아래 표 4는 락토바실러스 플란타룸 K10 균주에 대한 효소 활성(Enzyme) 패턴을 나타낸 것으로, 표준 컬러에는 0부터 2까지의 값이 할당되는데, 0은 음수를 나타내고 5는 최대 강도의 반응을 나타낸다. 1부터 4의 값은 강도 수준에 따른 중간 반응을 나타내고, 대략적인 활동은 컬러 강도에서 추정 할 수 있다. 1은 5 나노 몰, 2는 10 나노 몰 이하, 3은 20 나노 몰 이하, 4는 30 나노 몰 이하 그리고 5는 40 나노 몰 이하 또는 그 이상의 유리(liberation)에 해당한다.Table 4 below shows the enzyme activity pattern for Lactobacillus plantarum K10 strain. In the standard color, a value of 0 to 2 is assigned, where 0 represents a negative number and 5 represents a maximum intensity reaction. The values from 1 to 4 indicate the intermediate response according to the intensity level, and the approximate activity can be estimated from the color intensity. 1 corresponds to 5 nanomolar, 2 corresponds to 10 nanomolar or less, 3 corresponds to 20 nanomolar or less, 4 corresponds to 30 nanomolar or less and 5 corresponds to 40 nanomolar or less liberation.
아래 표 5는 락토바실러스 플란타룸 KC28 균주에 대한 항생제내성(Antibiotics susceptibility)을 나타낸 것으로, 젠타마이신(gentamycin), 암피실린(ampicillin), 테트라사이클린(tetracycline), 클로람페니콜(chloramphenicol), 스트렙토마이신(streptomycin), 시프로플록사신(ciprofloxacin) 및 페니실린 지(penicillin G) 등에 대한 상기 분리한 균주의 항생제 내성을 확인하였다.Table 5 below shows the antibiotic susceptibility of Lactobacillus plantarum KC28 strain, and it is shown that gentamycin, ampicillin, tetracycline, chloramphenicol, streptomycin, , Ciprofloxacin, penicillin G, and the like were confirmed to be resistant to antibiotics.
구체적으로, 각각의 항생제를 계단 희석(1:2)하고, 0.025 ~ 64 ㎍/ml의 농도로 MRS 고체배지에 혼합하였다. 18시간 배양한 락토바실러스 플란타룸 KC28 균주를 1 x 105 CFU가 되도록 상기 항생제가 섞인 고체배지에 접종한 후, 37℃에서 24시간 동안 배양하여 최소저해농도(MIC)를 측정하였다.Specifically, each antibiotic was stair-diluted (1: 2) and mixed in MRS solid medium at a concentration of 0.025 to 64 / / ml. The minimum inhibitory concentration (MIC) was measured by inoculating Lactobacillus planta KC28 strain, which had been cultivated for 18 hours, into a solid medium containing the above antibiotics at a concentration of 1 × 10 5 CFU, followed by culturing at 37 ° C. for 24 hours.
생체 아민(biogenic amines)은 식품의 발효에 의해 생성되고, 미생물 종류 또는 화학적, 물리적 조건에 따라 다양할 수 있다. 발효 식품에 생성된 생체 아민은 식중독 또는 알러지 반응을 일으킬 수 있기 때문에 식품 공학적으로 안전한 균주를 선택하기 위한 중요한 기준이 된다.Biogenic amines are produced by the fermentation of foodstuffs and may vary depending on the type of microorganism or chemical and physical conditions. Biomolecules produced in fermented foods are an important criterion for choosing a food-engineered strain because it can cause food poisoning or allergic reactions.
이에 본 발명의 균주의 생체 아민 형성 여부를 확인하기 위해 MRS 액체 배지, 37 조건에서 16시간 동안 자란 균주를 Bover-Cid 및 Holzapfel(1999)에 따른 특수 배지로 옮기고 37에서, 48시간 동안 배양하였다.In order to confirm the formation of biomolecules of the strain of the present invention, the strain grown for 16 hours in the MRS liquid medium, 37, was transferred to a special medium according to Bover-Cid and Holzapfel (1999) and cultured at 37 for 48 hours.
티로신(tyrosine), 히스티딘(histidine), 오르니틴(ornithine) 및 라이신(lysine) 각각의 아미노산 전구체를 첨가한 MRS 액체 배지를 제조하고, 각각의 배지에서 균주에 의해 생체 아민(티라민(tyramine), 히스타민(histamine), 푸트레신(putrescine) 및 카다베린(cadaverine))이 생성되는지 확인하였다. 구체적으로 상기 아미노산 전구체 0.1%를 첨가한 MRS 액체 배지에 분리한 락토바실러스 플란타룸 균주를 1%씩 접종한 뒤, 5 ~ 10번 계대 배양하여, 탈탄산효소(decarboxylase)를 활성화시켰다. 탈탄산효소 배지[트립톤 0.5%, 효모 추출물 0.5%, 고치 추출물 0.5%, 염화나트륨 0.5%, 글루코스 0.25%, 트윈-80 0.05%, 황산마그네슘 0.02%, 황산망간 0.005%, 황산철 0.004%, 시트르산염 0.2%, 티아민 0.001%, K2PO4 0.2%, 탄산칼슘 0.01%, 피로독살-5-포스페이트(pyridoxal-5-phosphate) 0.005%, 아미노산 1%, 브로모크레솔 퍼플(bromocresol purple) 0.006% 및 한천 2%를 증류수에 섞은 후 pH를 5.3으로 맞추어 사용]에 효소 활성화된 균을 도말한 후 37에서 24 ~ 48시간 배양하여 보라색으로 색이 변하는지를 확인함으로써 생체 아민 생성능을 판별하였다.MRS liquid medium supplemented with amino acid precursors of each of tyrosine, histidine, ornithine and lysine was prepared and the biomolecules (tyramine, histamine (histamine), putrescine and cadaverine) were produced. Specifically, 1% of the isolated Lactobacillus plantarum strain was inoculated into the MRS liquid medium supplemented with 0.1% of the amino acid precursor, and then cultured for 5-10 times to activate the decarboxylase. The concentration of dextrinase enzyme medium [tryptone 0.5%, yeast extract 0.5%, cocoon extract 0.5%, sodium chloride 0.5%, glucose 0.25%, tween-80 0.05%, magnesium sulfate 0.02%, manganese sulfate 0.005% 0.2% of salt, 0.001% of thiamine, 0.2% of K2PO4, 0.01% of calcium carbonate, 0.005% of pyridoxal-5-phosphate, 1% of amino acid, 0.006% of bromocresol purple, 2% was mixed with distilled water and adjusted to pH of 5.3], and then incubated at 37 for 24 to 48 hours to determine the color change to purple.
탈탄산효소 배지 속에 있는 브로모크레솔 퍼플은 pH 5.2에서 노란색을 띄지만 pH가 6.8로 올라갈수록 보라색으로 변한다. 따라서 생체 아민 생성으로 인해 pH가 올라갈 때 보라색으로 변하는 것을 이용하여 생체 아민 생성을 확인할 수 있다.The bromocresol purple in the decarbonated enzyme medium is yellow at pH 5.2 but turns purple as the pH rises to 6.8. Therefore, the production of bio-amines can be confirmed by using a substance that turns purple when the pH rises due to the formation of bio-amines.
아래 표 6은 락토바실러스 플란타룸 KC28 균주에 대한 생체 아민 생성능을 분석한 결과를 나타낸 것으로, 표 6을 통해 알 수 있듯이, 푸트레신(putrescine), 티라민(tyramine), 히스타민(histamine) 및 카다베린(cadaverine)에 대하여 모두 음성으로 확인되었다. 이로부터 본 발명의 균주는 과민성 면역반응을 유발할 수 있는 생체 아민 생성능이 없음을 확인할 수 있었다.As shown in Table 6, putrescine, tyramine, histamine, and Kade were found to be effective for the biosynthesis of Lactobacillus plantarum KC28. All of the cadaverine was confirmed by negative. From these results, it was confirmed that the strain of the present invention has no bio-amine generating ability capable of inducing an irritable immune response.
도 4는 본 발명의 락토바실러스 플란타룸 KC28 균주에 대한 장 정착능 실험 결과를 나타낸 도면이다.FIG. 4 is a graph showing the results of an experiment for the long-term fixation of Lactobacillus plantarum KC28 strain of the present invention.
보다 상세하게는, HT-29 세포에 대한 정착능(adhesion ability)을 나타낸 것으로, 락토바실러스 람노서스 GG프로바이오틱스 균주를 함께 나타낸 것이며, 모든 수치(또는 값)는 3회 반복의 평균±표준편차이고, **는 대조군(control)과 비교하여P<0.05인 경우를 표기하였다.More specifically, it shows the adhesion ability to HT-29 cells together with Lactobacillus lambusus GG probiotic strain, and all values (or values) are mean ± standard deviation of three replicates, ** indicates the case of P < 0.05 compared with the control (control).
도 5는 비만 유도 마우스의 12 주간의 무게에 대한 실험 결과를 나타낸 도면으로, 다른 유산균 균주들을 함께 나타낸 것이다.FIG. 5 is a graph showing the results of an experiment on the weight of the obesity-induced mouse for 12 weeks, together with other lactic acid bacterial strains.
보다 상세하게는, ND는 일반 chow fat 식이 투여그룹이고, HFD는 IF(intermediate fat) 식이 투여그룹이며, Xen는 IF + xenical 투여 그룹이고, LGG는 IF + LGG 투여그룹이며, 299v는 IF + 299v 투여그룹이고, KC3는 IF + KC3 투여그룹이며, KC4는 IF + KC4 투여그룹이고, K10는 IF + K10 투여그룹이며, K50는 IF + K50 투여그룹이고, KC28는 IF + KC28 투여그룹이며, K259는 IF + K259 투여그룹이고, LR86는 IF + LR86 투여그룹이며, KID7는 IF + KID7 투여그룹이다. 여기서, 값은 평균과 표준편차로 나타내었으며, 일반식이와 혼합식이 투여그룹은 IF 식이에 대하여 Fisher's LSD test 사후 검정 후 p<0.05 일 때 *로 표기하고, p<0.01 일 때 **로 표기하며, p<0.001 일 때 ***를 표기하였다. More specifically, ND is a normal chow fat diet group, HFD is an IF (intermediate fat) diet group, Xen is IF + xenical administration group, LGG is IF + LGG administration group, 299v is IF + KC3 is an IF + KC4 administration group, KC4 is an IF + KC4 administration group, K10 is an IF + K10 administration group, K50 is an IF + K50 administration group, KC28 is an IF + KC28 administration group, LR86 is the IF + LR86 administration group, and KID7 is the IF + KID7 administration group. The values were expressed as mean and standard deviation. For the IF diet, the control diet and mixed diet groups were marked with * when p <0.05 after Fisher's LSD test, and ** when p <0.01 , and *** when p <0.001.
도 6은 마우스 실험 완료 후 적출한 정소지방 (A), 피하지방 (B), 및 내장지방 (C)의 무게 차이를 나타낸 도면으로, 다른 유산균 균주들을 함께 나타낸 것이다.FIG. 6 is a graph showing the weight difference between the testis fat (A), subcutaneous fat (B) and visceral fat (C) extracted after completion of the mouse experiment, together with other lactic acid bacterial strains.
보다 상세하게는, ND는 일반 chow fat 식이 투여그룹이고, HFD는 IF(intermediate fat) 식이 투여그룹이며, Xen는 IF + xenical 투여 그룹이고, LGG는 IF + LGG 투여그룹이며, 299v는 IF + 299v 투여그룹이고, KC3는 IF + KC3 투여그룹이며, KC4는 IF + KC4 투여그룹이고, K10는 IF + K10 투여그룹이며, K50는 IF + K50 투여그룹이고, KC28는 IF + KC28 투여그룹이며, K259는 IF + K259 투여그룹이고, LR86는 IF + LR86 투여그룹이며, KID7는 IF + KID7 투여그룹이다. 여기서, 값은 평균과 표준편차로 나타내었으며, 일반식이와 혼합식이 투여그룹은 IF 식이에 대하여 Fisher's LSD test 사후 검정 후 p<0.05 일 때 *로 표기하고, p<0.01 일 때 **로 표기하며, p<0.001 일 때 ***를 표기하였다. More specifically, ND is a normal chow fat diet group, HFD is an IF (intermediate fat) diet group, Xen is IF + xenical administration group, LGG is IF + LGG administration group, 299v is IF + KC3 is an IF + KC4 administration group, KC4 is an IF + KC4 administration group, K10 is an IF + K10 administration group, K50 is an IF + K50 administration group, KC28 is an IF + KC28 administration group, LR86 is the IF + LR86 administration group, and KID7 is the IF + KID7 administration group. The values were expressed as mean and standard deviation. For the IF diet, the control diet and mixed diet groups were marked with * when p <0.05 after Fisher's LSD test, and ** when p <0.01 , and *** when p <0.001.
도 7은 Fluorescein isothiocyanate conjugated dextran (FITC-dextran) (Sigma-Aldrich)를 이용하여 장 투과성을 측정한 결과를 나타낸 도면으로, 다른 락토바실러스 플란타룸 균주들을 함께 나타낸 것이다.FIG. 7 is a graph showing the results of measurement of intestinal permeability using Fluorescein isothiocyanate conjugated dextran (FITC-dextran) (Sigma-Aldrich), together with other Lactobacillus plantarum strains.
보다 상세하게는, ND는 일반 chow fat 식이 투여그룹이고, HFD는 IF(intermediate fat) 식이 투여그룹이며, LGG는 IF + LGG 투여그룹이며, 299v는 IF + 299v 투여그룹이고, K10는 IF + K10 투여그룹이며, K50는 IF + K50 투여그룹이고, KC28는 IF + KC28 투여그룹이며, K259는 IF + K259 투여그룹이다. 여기서, 값은 평균과 표준편차로 나타내었으며, 일반식이와 혼합식이 투여그룹은 IF 식이에 대하여 Fisher's LSD test 사후 검정 후 p<0.05 일 때 *로 표기하고, p<0.01 일 때 **로 표기하며, p<0.001 일 때 ***를 표기하였다.More specifically, ND is a normal chow fat diet administration group, HFD is an IF (intermediate fat) diet administration group, LGG is IF + LGG administration group, 299v is IF + 299v administration group, K10 is IF + K10 K50 is an IF + K50 administration group, KC28 is an IF + KC28 administration group, and K259 is an IF + K259 administration group. The values were expressed as mean and standard deviation. For the IF diet, the control diet and mixed diet groups were marked with * when p <0.05 after Fisher's LSD test, and ** when p <0.01 , and *** when p <0.001.
구체적으로, 장의 점막세포는 단일 세포층으로서 세포 사이는 치밀결합 (tight junction)으로 서로 붙어있으나, 비만 유도 및 장균총 조성 변화로 인해 이 세포의 간격이 느슨해지거나 손상을 입어 장벽의 기능을 제대로 못하여 혈액 내의 고분자 물질이 장관 내로 누수 되거나 장관 내의 고분자 물질이 직접 혈액으로 들어가는 현상으로 인해 다양한 질환이 생기는 것이 "새는 장 증후군 (Leaky gut syndrome)"이다. 어떤 이유로든 투과성이 증가해 장이 샌다면, 병원균, 내독소(그람음성 균주의 부서진 세포벽 조직), 염증성 분자 등이 혈류로 유입되어 장관내독소혈증이 일어난다. 그러나 유산균은 장 투과성을 감소시키는 효과도 있으며, 장내 점막과 결합하여 군집성 방어막을 형성하고, 유해균이 장 점막에 손상을 주는 것을 막는다. 따라서, Bio-protocol 기준으로 15주간 혼합 식이를 먹은 마우스 (N=6~9)에게 FITC-dextran을 (44mg/100g 쥐 무게) 먹인 후 혈청을 얻어 확인한 장 투과성 차이는 도 7과 같다. 도 7을 통해 알 수 있듯이, 락토바실러스 플란타룸 KC28 균주는 strain specific하게 대조군(HFD)에 비해 장 투과성이 유의적으로 낮은 것을 알 수 있으며, 락토바실러스 람노서스 GG프로바이오틱스 균주와 비교해도 유의적으로 장 투과성이 낮은 결과를 확인하였다.Specifically, the mucosal cells of the intestine are a single cell layer, but the cells are attached to each other with tight junctions. However, due to the induction of obesity and changes in the fibrinogen composition, the gap between these cells is loosened or damaged, Leaky gut syndrome is the result of the leakage of polymeric materials into the intestinal tract or the introduction of polymeric substances directly into the bloodstream. If the permeability increases for any reason, the pathogen, endotoxin (broken cell wall tissue of gram negative strain), and inflammatory molecules enter the bloodstream and intestinal endotoxemia occurs. However, lactic acid bacteria also have the effect of reducing the intestinal permeability, and combine with intestinal mucosa to form a clustered membrane, preventing harmful bacteria from damaging the intestinal mucosa. Therefore, FITC-dextran (44 mg / 100 g mouse weight) was fed to mice (N = 6-9) fed with a 15-week mixed diet on the basis of bio-protocol, As can be seen from Fig. 7, the Lactobacillus plantarum KC28 strain was strain-specific and the intestinal permeability was significantly lower than that of the control (HFD), and it was significantly higher than that of Lactobacillus lambosus GG probiotic strain And the low permeability was confirmed.
또한, 본 발명은 상기 락토바실러스 플란타룸 KC28 균주, 상기 균주의 배양물, 상기 배양물의 농축액 및 건조물로 이루어지는 군에서 선택된 1종 이상을 유효성분으로 포함하는 프로바이오틱스 제제를 제공할 수도 있다.In addition, the present invention may provide a probiotic preparation comprising at least one member selected from the group consisting of the Lactobacillus plantarum KC28 strain, the culture of the strain, the concentrate of the culture and the dried product as an active ingredient.
상기 프로바이오틱스 제제는 당업계에 공지된 방법에 따라 다양한 제형과 방법으로 제조 및 투여될 수 있다. 예를 들어, 본 발명의 락토바실러스 플란타룸 KC28 균주, 이의 배양액, 상기 배양액의 농축액 또는 그의 건조물은 약제학적 분야에서 통상적으로 사용되는 담체와 혼합하여 산제(powder), 액제(liquids and solutions), 정제(tablet), 캡슐(capsule), 시럽(syrup), 현탁제(suspension) 또는 과립제(granule) 등의 형태로 제조되어 투여될 수 있다. 상기 담체로는 예를 들어, 결합제, 활탁제, 붕해제, 부형제, 가용화제, 분산제, 안정화제, 현탁화제, 색소 및 향료 등일 수 있으나, 이에 제한되지 않는다. 또한, 투여 용량은 체내에서의 활성성분의 흡수도, 불활성률, 배설속도, 피투여자의 연령, 성별, 축종, 상태 및 질병의 중증 정도 등에 따라 적절히 선택할 수 있다.The probiotic agent can be prepared and administered in various formulations and methods according to methods known in the art. For example, the Lactobacillus plantarum KC28 strain of the present invention, a culture thereof, a concentrate of the culture broth or a dried product thereof may be mixed with a carrier commonly used in the pharmaceutical field to prepare powders, liquids and solutions, May be formulated and administered in the form of tablets, capsules, syrups, suspensions or granules, and the like. Examples of the carrier include, but are not limited to, binders, lubricants, disintegrants, excipients, solubilizers, dispersants, stabilizers, suspending agents, coloring matters and fragrances. The administration dose can be appropriately selected depending on the degree of absorption of the active ingredient in the body, the inactivation rate, the excretion rate, the age, sex, strain, condition and severity of the disease.
또한, 본 발명은 상기 락토바실러스 플란타룸 KC28 균주, 상기 균주의 배양물, 상기 배양물의 농축액 및 건조물로 이루어지는 군에서 선택된 1종 이상을 유효성분으로 함유하는 비만 예방 또는 개선용 건강기능식품 조성물을 제공한다.The present invention also provides a health functional food composition for preventing or ameliorating obesity comprising at least one member selected from the group consisting of Lactobacillus plantarum KC28, a culture of the strain, a concentrate of the culture, and a dried product as an active ingredient to provide.
본 발명의 건강기능식품 조성물은 정제, 과립, 분말, 캅셀, 액상의 용액 및 환으로 이루어진 군으로부터 선택된 어느 하나의 제형으로 제조된 것일 수 있다. 본 발명에 따른 건강기능식품 조성물은 상기 락토바실러스 플란타룸 KC28 균주 등을 유효성분으로 포함시켜 분말제, 액제, 정제, 연질캅셀제, 과립제, 티백차, 인스턴트 차 또는 드링크제 등의 형태로 제형화될 수 있다. 유효 성분으로서의 락토바실러스 플란타룸 KC28 균주의 함량은 그 사용 목적(예방 또는 개선용)에 따라 적합하게 결정될 수 있다. 또한, 본 발명에 따른 식품 조성물은 락토바실러스 플란타룸 KC28 균주 이외에 본 발명이 목적으로 하는 주효과를 손상시키지 않는 범위 내에서 바람직하게는 주효과에 상승효과를 줄 수 있는 다른 성분을 함유하는 것도 무방하다. The health functional food composition of the present invention may be prepared in any one form selected from the group consisting of tablets, granules, powders, capsules, liquid solutions and rings. The health functional food composition according to the present invention may be formulated in the form of a powder, a liquid, a tablet, a soft capsule, a granule, a tea bag, an instant tea or a drink by incorporating the Lactobacillus plantarum KC28 strain or the like as an active ingredient . The content of Lactobacillus plantarum KC28 strain as an active ingredient can be suitably determined according to the intended use (for prevention or improvement). In addition, the food composition according to the present invention may contain, in addition to the Lactobacillus plantarum KC28 strain, other components that can give rise to a synergistic effect on the main effect, within the range not impairing the intended main effect of the present invention It is acceptable.
상기와 같은 형태로 제형화된 식품 조성물은 식품에 그대로 첨가하거나 다른 식품 또는 식품 성분과 함께 사용될 수 있고, 통상적인 방법에 따라 적절하게 사용될 수 있다. 식품의 예로는 드링크제, 육류, 소시지, 빵, 비스킷, 떡, 초콜릿, 캔디류, 스낵류, 과자류, 피자, 라면, 기타 면류, 껌류, 아이스크림류를 포함한 낙농제품, 각종 스프, 음료수, 알코올 음료 및 비타민 복합제, 유제품 및 유가공 제품 등이 있으며, 통상적인 의미에서의 건강기능 식품은 모두 포함한다. The food composition formulated in this form can be added directly to the food or used together with other food or food ingredients, and can be suitably used according to conventional methods. Examples of food products include dairy products including drinks, meat, sausage, bread, biscuits, rice cakes, chocolate, candies, snacks, confectionery, pizza, ramen, other noodles, gums, ice cream, soups, , Dairy products and dairy products, and all health functional foods in the ordinary sense are included.
본 발명의 식품 조성물이 음료인 경우는 지시된 비율로 필수성분으로서 락토바실러스 플란타룸 KC28 균주를 함유하며, 그 밖의 음료 제조를 목적으로 사용되는 다른 성분에는 특별한 제한이 없으며 통상의 음료와 같이 여러 가지 향미제 또는 천연 탄수화물 등을 추가 성분으로서 함유할 수 있다. 상기한 천연 탄수화물의 예는 모노사카라이드, 예를 들어, 포도당, 과당 등; 디사카라이드, 예를 들어 말토스, 슈크로스 등; 및 폴리사카라이드, 예를 들어 덱스트린, 시클로덱스트린 등과 같은 통상적인 당, 및 자일리톨, 소르비톨, 에리트리톨 등의 당알콜이다. 상술한 것 이외의 향미제로서 천연 향미제 및 합성 향미제 등을 사용할 수 있다. 상기 천연 탄수화물의 비율은 본 발명의 조성물 100 ㎖당 일반적으로 약 1 내지 20 g, 바람직하게는 약 5 내지 12 g이다. When the food composition of the present invention is a beverage, it contains Lactobacillus planta KC28 strain as an essential ingredient at the indicated ratio, and there are no particular restrictions on other ingredients used for the purpose of producing other beverages. Flavoring agents, natural carbohydrates and the like as additional components. Examples of such natural carbohydrates include monosaccharides such as glucose, fructose, and the like; Disaccharides such as maltose, sucrose and the like; And polysaccharides, for example, conventional sugars such as dextrin, cyclodextrin and the like, and sugar alcohols such as xylitol, sorbitol and erythritol. Natural flavors and synthetic flavors and the like can be used as the flavors other than those described above. The ratio of the natural carbohydrate is generally about 1 to 20 g, preferably about 5 to 12 g per 100 ml of the composition of the present invention.
또한 본 발명의 식품 조성물은 여러 가지 영양제, 비타민, 광물(전해질), 합성 풍미제 및 천연 풍미제 등의 풍 미제, 착색제 및 증진제(치즈, 초콜릿 등), 펙트산 및 그의 염, 알긴산 및 그의 염, 유기산, 보호성 콜로이드 증점제, pH 조절제, 안정화제, 방부제, 글리세린, 알코올, 탄산음료에 사용되는 탄산화제 등을 함유할 수 있다. 그 밖에도 본 발명의 본 발명의 식품 조성물은 천연 과일 쥬스 및 과일 쥬스 음료 및 야채 음료의 제조를 위한 과육을 함유할 수 있다. 이러한 성분은 독립적으로 또는 조합하여 사용할 수 있다. 이러한 첨가제의 비율은 그렇게 중요하진 않지만 본 발명의 락토바실러스 플란타룸 KC28 균주 100 중량부 당 0.1 내지 약 20 중량부의 범위에서 선택되는 것이 일반적이다.In addition, the food composition of the present invention can be used as a food composition containing various nutrients, vitamins, minerals (electrolytes), synthetic flavors and flavors such as natural flavors, colorants and enhancers (cheese, chocolate, etc.), pectic acid and its salts, , Organic acids, protective colloid thickeners, pH adjusting agents, stabilizers, preservatives, glycerin, alcohols, carbonating agents used in carbonated drinks, and the like. In addition, the inventive food composition of the present invention may contain natural fruit juice and pulp for the production of fruit juice drinks and vegetable drinks. These components may be used independently or in combination. Although the ratio of such additives is not so important, it is generally selected in the range of 0.1 to about 20 parts by weight per 100 parts by weight of Lactobacillus plantarum KC28 strain of the present invention.
또한, 본 발명은 상기 락토바실러스 플란타룸 KC28 균주, 상기 균주의 배양물, 상기 배양물의 농축액 및 건조물로 이루어지는 군에서 선택된 1종 이상을 유효성분으로 함유하는 비만 예방 또는 치료용 약학적 조성물을 제공한다.Further, the present invention provides a pharmaceutical composition for preventing or treating obesity, which comprises at least one selected from the group consisting of the above-mentioned Lactobacillus plantarum KC28 strain, the culture of the strain, the concentrate of the culture and the dried product as an active ingredient do.
본 발명에서 사용되는 비만 예방 또는 치료용 약학적 조성물의 처리량은 약학적으로 유효한 양이어야 한다. 본 발명에서 사용되는 용어, "약학적으로 유효한 양"은 의학적 치료에 적용 가능한 합리적인 수혜/위험 비율로 질환을 치료하기에 충분한 양을 의미하며, 유효 용량 수준은 개체 종류 및 중증도, 연령, 성별, 감염된 바이러스 종류, 약물의 활성, 약물에 대한 민감도, 투여 시간, 투여 경로 및 배출 비율, 치료 기간, 동시 사용되는 약물을 포함한 요소 및 기타 의학 분야에 잘 알려진 요소에 따라 결정될 수 있다. 유효량은 당업자에게 인식되어 있듯이 처리의 경로, 부형제의 사용 및 다른 약제와 함께 사용할 수 있는 가능성에 따라 변할 수 있다.The throughput of the pharmaceutical composition for preventing or treating obesity used in the present invention should be a pharmaceutically effective amount. As used herein, the term "pharmaceutically effective amount" means an amount sufficient to treat a disease at a reasonable benefit / risk ratio applicable to medical treatment and the effective dose level will vary depending on the species and severity, The type of virus being infected, the activity of the drug, the sensitivity to the drug, the time of administration, the route of administration and rate of release, the duration of the treatment, factors including co-administered drugs and other well known factors in the medical field. Effective amounts may vary depending on the route of treatment, the use of excipients, and the likelihood of use with other agents, as will be appreciated by those skilled in the art.
본 발명의 비만 예방 또는 치료용 약학적 조성물은 포유동물에 투여된 후 활성 성분의 신속, 지속 또는 지연된 방출을 제공할 수 있도록 당업계에 잘 알려진 방법을 사용하여 약학적 제형으로 제조될 수 있다. 제형의 제조에 있어서, 활성 성분을 담체와 함께 혼합 또는 희석하거나, 용기 형태의 담체 내에 봉입시키는 것이 바람직하다. The pharmaceutical composition for the prevention or treatment of obesity of the present invention may be manufactured into pharmaceutical formulations using methods well known in the art so as to provide rapid, sustained or delayed release of the active ingredient after administration to the mammal. In the preparation of the formulations, it is preferred that the active ingredient is mixed with or diluted with the carrier, or enclosed in a carrier in the form of a container.
따라서, 본 발명의 비만 예방 또는 치료용 약학적 조성물은 통상의 방법에 따라 산제, 과립제, 정제, 캡슐제, 현탁액, 에멀젼, 시럽, 에어로졸 등의 경구형 제형, 외용제 및 패치의 형태로 제형화하여 사용될 수 있고, 조성물의 제조에 통상적으로 사용하는 적절한 담체, 부형제 또는 희석제를 추가로 포함할 수 있다. Therefore, the pharmaceutical composition for preventing or treating obesity of the present invention can be formulated into oral formulations, external preparations and patches such as powders, granules, tablets, capsules, suspensions, emulsions, syrups and aerosols according to a conventional method And may further comprise suitable carriers, excipients or diluents conventionally used in the manufacture of the compositions.
예를 들어, 본 발명의 약학 조성물에 포함될 수 있는 담체, 부형제 및 희석제로는 락토즈, 덱스트로즈, 수크로스, 솔비톨, 만니톨, 자일리톨, 에리스리톨, 말티톨, 전분, 아카시아 고무, 알지네이트, 젤라틴, 칼슘 포스페이트, 칼슘 실리케이트, 셀룰로즈, 메틸 셀룰로즈, 미정질 셀룰로스, 폴리비닐 피롤리돈, 물, 메틸히드록시벤조에이트, 프로필히드록시벤조에이트, 탈크, 마그네슘 스테아레이트 및 광물유를 들 수 있다. 제제화할 경우에는 보통 사용하는 충진제, 증량제, 결합제, 습윤제, 붕해제, 계면활성제 등의 희석제 또는 부형제를 사용하여 조제될 수 있다.Examples of carriers, excipients and diluents that can be included in the pharmaceutical composition of the present invention include lactose, dextrose, sucrose, sorbitol, mannitol, xylitol, erythritol, maltitol, starch, acacia rubber, alginate, gelatin, calcium Cellulose, methylcellulose, microcrystalline cellulose, polyvinylpyrrolidone, water, methylhydroxybenzoate, propylhydroxybenzoate, talc, magnesium stearate and mineral oil. In the case of formulation, it can be prepared using diluents or excipients such as fillers, extenders, binders, humectants, disintegrants, surfactants and the like which are usually used.
또한, 본 명세서에 기재된 락토바실러스 락토바실러스 플란타룸 KC28 균주는 건강보조식품, 음료, 발효된 유제품을 포함하는 발효 식품 등과 같이 다양한 형태의 제품들에 함유될 수도 있다.In addition, the Lactobacillus lactobacillus plantarum KC28 strain described herein may be contained in various types of products such as health food, beverages, fermented foods including fermented dairy products, and the like.
Claims (7)
Lactobacillus plantarum KC28 strain (accession number: KCTC13377BP) having an anti-obesity effect.
상기 균주는 티로신, 히스티딘, 오르니틴 및 라이신으로 이루어진 군에서 선택되는 하나 이상의 아미노산 전구체로부터 생체 아민을 생성하지 않는 것을 특징으로 하는 락토바실러스 플란타룸 KC28 균주.
The method according to claim 1,
Wherein the strain does not produce a biological amine from one or more amino acid precursors selected from the group consisting of tyrosine, histidine, ornithine, and lysine.
A probiotic preparation comprising at least one member selected from the group consisting of a strain according to any one of claims 1 to 2, a culture of the strain, a concentrate of the culture and a dried product as an active ingredient.
A health functional food composition comprising, as an active ingredient, at least one selected from the group consisting of a strain of any one of claims 1 to 2, a culture of the strain, a concentrate of the culture, and a dried product.
A pharmaceutical composition comprising, as an active ingredient, at least one selected from the group consisting of a strain of any one of claims 1 to 2, a culture of the strain, a concentrate of the culture, and a dried product.
A beverage comprising at least one of the strains of any one of claims 1 to 2, a culture of the strain and a concentrate or a dried product of the culture as an active ingredient.
A health supplement containing at least one of the strains of any one of claims 1 to 2, a culture of the strain and a concentrate or a dried product of the culture as an active ingredient.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020170179135A KR20190077714A (en) | 2017-12-26 | 2017-12-26 | Lactobacillus plantarum KC28 having anti-obesity effect and uses thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020170179135A KR20190077714A (en) | 2017-12-26 | 2017-12-26 | Lactobacillus plantarum KC28 having anti-obesity effect and uses thereof |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| KR20190077714A true KR20190077714A (en) | 2019-07-04 |
Family
ID=67259035
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020170179135A KR20190077714A (en) | 2017-12-26 | 2017-12-26 | Lactobacillus plantarum KC28 having anti-obesity effect and uses thereof |
Country Status (1)
| Country | Link |
|---|---|
| KR (1) | KR20190077714A (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR20210062801A (en) * | 2019-11-22 | 2021-06-01 | 이진영 | Lactobacillus Plantarum YG1102 Strain Having Antimicrobial and Anti-Obesity Activity and Uses thereof |
| KR102506366B1 (en) * | 2022-08-16 | 2023-03-09 | (주)지에이치바이오 | Lactobacillus plantarum KU15120 strain or its use |
-
2017
- 2017-12-26 KR KR1020170179135A patent/KR20190077714A/en not_active Application Discontinuation
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR20210062801A (en) * | 2019-11-22 | 2021-06-01 | 이진영 | Lactobacillus Plantarum YG1102 Strain Having Antimicrobial and Anti-Obesity Activity and Uses thereof |
| KR102506366B1 (en) * | 2022-08-16 | 2023-03-09 | (주)지에이치바이오 | Lactobacillus plantarum KU15120 strain or its use |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR102011883B1 (en) | Lactobacillus plantarum KC3 having anti-obesity effect and uses thereof | |
| KR102011881B1 (en) | Lactobacillus plantarum K10 having anti-obesity effect and uses thereof | |
| KR101825836B1 (en) | Novel Lactobacillus plantarum Lb41 strain and compositions for the prevention and treatment of diabetes or insulin resistance syndrome containing the same | |
| CN107075455B (en) | Lactobacillus plantarum HAC01 strain with anti-inflammatory and metabolic disease improvement effects and application thereof | |
| AU2017264267A1 (en) | A composition comprising a lactic acid bacteria for preventing and treating vaginosis and the use thereof | |
| US11273189B2 (en) | Lactobacillus plantarum TCI378 and its uses in losing fat and improving gastrointestinal functions | |
| KR101680014B1 (en) | Lactic acid bacterium isolated from Kimchi for treating inflammatory bowel disease and uses thereof | |
| KR20160132050A (en) | Novel lactobacillus paracasei strain | |
| KR101825837B1 (en) | Novel Lactobacillus plantarum Ln4 strain and compositions for the prevention and treatment of diabetes or insulin resistance syndrome containing the same | |
| KR20200018532A (en) | Lactobacillus sp. strain having inhibitory effect against microorganisms causing vaginosis uses thereof | |
| KR102294437B1 (en) | Infant and child origin lactic acid bacteria Lactobacillus plantarum MG4553 and composition comprising the lactic acid bacteria for enhancing intestine activity, anti-oxidant and anti-obesity | |
| KR102294442B1 (en) | Infant and child origin lactic acid bacteria Lactobacillus plantarum MG4555 and composition comprising the lactic acid bacteria for enhancing intestine activity, anti-oxidant and anti-obesity | |
| KR20190077714A (en) | Lactobacillus plantarum KC28 having anti-obesity effect and uses thereof | |
| KR102011882B1 (en) | Lactobacillus plantarum K50 having anti-obesity effect and uses thereof | |
| KR20190067527A (en) | A Bacillus probiotics mixture for preventing or treating obesity, diabetes and fatty liver and uses thereof | |
| KR102190239B1 (en) | Novel Lactobacillus plantarum Lb41 strain and compositions for the prevention and treatment of obesity containing the same | |
| KR102479732B1 (en) | Limosilactobacillus reuteri MG5458 strain and composition for preventing, improving or treating alcoholic fatty liver comprising the same | |
| KR102037108B1 (en) | Composition for Preventing or Treating Obesity Comprising Heat-killed Lactic Acid Bacteria or Yeast from Kefir | |
| KR20190102498A (en) | Lactobacillus sp. strain having inhibitory effect against microorganisms causing vaginosis uses thereof | |
| KR20180040894A (en) | Novel Lactobacillus plantarum Lb41 strain and compositions for the prevention and treatment of obesity containing the same | |
| KR20190077715A (en) | Lactobacillus plantarum K259 having anti-obesity effect and uses thereof | |
| KR20180040899A (en) | Novel Lactobacillus plantarum Ln4 strain and compositions for the prevention and treatment of obesity containing the same | |
| KR20120119555A (en) | COMPOSITION FOR α-GLUCOSIDASE INHIBITORY ACTIVITY | |
| KR102506366B1 (en) | Lactobacillus plantarum KU15120 strain or its use | |
| KR101376629B1 (en) | New Lactobacillus arizonensis BCNU 9200 and probiotics composition comprising the same |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A201 | Request for examination | ||
| E902 | Notification of reason for refusal | ||
| E601 | Decision to refuse application |