KR20180098252A - 압축 수소를 생산하는 방법 및 장치 - Google Patents
압축 수소를 생산하는 방법 및 장치 Download PDFInfo
- Publication number
- KR20180098252A KR20180098252A KR1020187017202A KR20187017202A KR20180098252A KR 20180098252 A KR20180098252 A KR 20180098252A KR 1020187017202 A KR1020187017202 A KR 1020187017202A KR 20187017202 A KR20187017202 A KR 20187017202A KR 20180098252 A KR20180098252 A KR 20180098252A
- Authority
- KR
- South Korea
- Prior art keywords
- hydrogen
- fuel cell
- solid oxide
- oxide fuel
- stream
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Images
Classifications
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M8/00—Fuel cells; Manufacture thereof
- H01M8/06—Combination of fuel cells with means for production of reactants or for treatment of residues
- H01M8/0606—Combination of fuel cells with means for production of reactants or for treatment of residues with means for production of gaseous reactants
- H01M8/0612—Combination of fuel cells with means for production of reactants or for treatment of residues with means for production of gaseous reactants from carbon-containing material
- H01M8/0637—Direct internal reforming at the anode of the fuel cell
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01B—NON-METALLIC ELEMENTS; COMPOUNDS THEREOF; METALLOIDS OR COMPOUNDS THEREOF NOT COVERED BY SUBCLASS C01C
- C01B3/00—Hydrogen; Gaseous mixtures containing hydrogen; Separation of hydrogen from mixtures containing it; Purification of hydrogen; Reversible storage of hydrogen
- C01B3/02—Production of hydrogen; Production of gaseous mixtures containing hydrogen
- C01B3/32—Production of hydrogen; Production of gaseous mixtures containing hydrogen by reaction of gaseous or liquid organic compounds with gasifying agents, e.g. water, carbon dioxide or air
- C01B3/34—Production of hydrogen; Production of gaseous mixtures containing hydrogen by reaction of gaseous or liquid organic compounds with gasifying agents, e.g. water, carbon dioxide or air by reaction of hydrocarbons with gasifying agents
- C01B3/38—Production of hydrogen; Production of gaseous mixtures containing hydrogen by reaction of gaseous or liquid organic compounds with gasifying agents, e.g. water, carbon dioxide or air by reaction of hydrocarbons with gasifying agents using catalysts
-
- C—CHEMISTRY; METALLURGY
- C25—ELECTROLYTIC OR ELECTROPHORETIC PROCESSES; APPARATUS THEREFOR
- C25B—ELECTROLYTIC OR ELECTROPHORETIC PROCESSES FOR THE PRODUCTION OF COMPOUNDS OR NON-METALS; APPARATUS THEREFOR
- C25B1/00—Electrolytic production of inorganic compounds or non-metals
- C25B1/01—Products
- C25B1/02—Hydrogen or oxygen
- C25B1/04—Hydrogen or oxygen by electrolysis of water
-
- C—CHEMISTRY; METALLURGY
- C25—ELECTROLYTIC OR ELECTROPHORETIC PROCESSES; APPARATUS THEREFOR
- C25B—ELECTROLYTIC OR ELECTROPHORETIC PROCESSES FOR THE PRODUCTION OF COMPOUNDS OR NON-METALS; APPARATUS THEREFOR
- C25B5/00—Electrogenerative processes, i.e. processes for producing compounds in which electricity is generated simultaneously
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M16/00—Structural combinations of different types of electrochemical generators
- H01M16/003—Structural combinations of different types of electrochemical generators of fuel cells with other electrochemical devices, e.g. capacitors, electrolysers
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M8/00—Fuel cells; Manufacture thereof
- H01M8/04—Auxiliary arrangements, e.g. for control of pressure or for circulation of fluids
- H01M8/04298—Processes for controlling fuel cells or fuel cell systems
- H01M8/04694—Processes for controlling fuel cells or fuel cell systems characterised by variables to be controlled
- H01M8/04746—Pressure; Flow
- H01M8/04753—Pressure; Flow of fuel cell reactants
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M8/00—Fuel cells; Manufacture thereof
- H01M8/06—Combination of fuel cells with means for production of reactants or for treatment of residues
- H01M8/0606—Combination of fuel cells with means for production of reactants or for treatment of residues with means for production of gaseous reactants
- H01M8/0656—Combination of fuel cells with means for production of reactants or for treatment of residues with means for production of gaseous reactants by electrochemical means
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M8/00—Fuel cells; Manufacture thereof
- H01M8/06—Combination of fuel cells with means for production of reactants or for treatment of residues
- H01M8/0662—Treatment of gaseous reactants or gaseous residues, e.g. cleaning
- H01M8/0681—Reactant purification by the use of electrochemical cells
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M8/00—Fuel cells; Manufacture thereof
- H01M8/10—Fuel cells with solid electrolytes
- H01M8/12—Fuel cells with solid electrolytes operating at high temperature, e.g. with stabilised ZrO2 electrolyte
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01B—NON-METALLIC ELEMENTS; COMPOUNDS THEREOF; METALLOIDS OR COMPOUNDS THEREOF NOT COVERED BY SUBCLASS C01C
- C01B2203/00—Integrated processes for the production of hydrogen or synthesis gas
- C01B2203/02—Processes for making hydrogen or synthesis gas
- C01B2203/0205—Processes for making hydrogen or synthesis gas containing a reforming step
- C01B2203/0227—Processes for making hydrogen or synthesis gas containing a reforming step containing a catalytic reforming step
- C01B2203/0233—Processes for making hydrogen or synthesis gas containing a reforming step containing a catalytic reforming step the reforming step being a steam reforming step
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01B—NON-METALLIC ELEMENTS; COMPOUNDS THEREOF; METALLOIDS OR COMPOUNDS THEREOF NOT COVERED BY SUBCLASS C01C
- C01B2203/00—Integrated processes for the production of hydrogen or synthesis gas
- C01B2203/06—Integration with other chemical processes
- C01B2203/066—Integration with other chemical processes with fuel cells
- C01B2203/067—Integration with other chemical processes with fuel cells the reforming process taking place in the fuel cell
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01B—NON-METALLIC ELEMENTS; COMPOUNDS THEREOF; METALLOIDS OR COMPOUNDS THEREOF NOT COVERED BY SUBCLASS C01C
- C01B2203/00—Integrated processes for the production of hydrogen or synthesis gas
- C01B2203/12—Feeding the process for making hydrogen or synthesis gas
- C01B2203/1205—Composition of the feed
- C01B2203/1211—Organic compounds or organic mixtures used in the process for making hydrogen or synthesis gas
- C01B2203/1235—Hydrocarbons
- C01B2203/1241—Natural gas or methane
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M8/00—Fuel cells; Manufacture thereof
- H01M8/10—Fuel cells with solid electrolytes
- H01M8/12—Fuel cells with solid electrolytes operating at high temperature, e.g. with stabilised ZrO2 electrolyte
- H01M2008/1293—Fuel cells with solid oxide electrolytes
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E60/00—Enabling technologies; Technologies with a potential or indirect contribution to GHG emissions mitigation
- Y02E60/30—Hydrogen technology
- Y02E60/50—Fuel cells
Landscapes
- Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Engineering & Computer Science (AREA)
- Electrochemistry (AREA)
- General Chemical & Material Sciences (AREA)
- Sustainable Energy (AREA)
- Sustainable Development (AREA)
- Life Sciences & Earth Sciences (AREA)
- Manufacturing & Machinery (AREA)
- Organic Chemistry (AREA)
- Inorganic Chemistry (AREA)
- Combustion & Propulsion (AREA)
- General Health & Medical Sciences (AREA)
- Health & Medical Sciences (AREA)
- Materials Engineering (AREA)
- Metallurgy (AREA)
- Electrolytic Production Of Non-Metals, Compounds, Apparatuses Therefor (AREA)
- Fuel Cell (AREA)
- Hydrogen, Water And Hydrids (AREA)
Abstract
Description
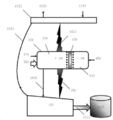
도 2는 본 발명에 따른 방법의 일 실시양태를 수행하는 본 발명에 따른 장치의 일 실시양태의 단순 구성도를 나타낸다.
Claims (8)
- (a) 메탄을 포함하는 공급스트림(feedstream) 및 증기 스트림(steam stream)을 고체 산화물 연료 전지에 공급하는 단계로서, 상기 고체 산화물 연료 전지의 애노드(anode)에서 개질 촉매 조성물(reforming catalyst composition)이 수소를 포함하는 생성 혼합물의 발생을 촉매화하는, 상기 공급하는 단계;
(b) 상기 고체 산화물 연료 전지를 이용하여, 수소를 포함하는 생성물 혼합물의 적어도 일부로부터 전기를 발생시키는 단계;
(c) 기상 수소를 추출하고 압축할 수 있는 전기화학소자를 적어도 부분적으로 가동(power)시키기 위해, 상기 고체 산화물 연료 전지에 의해 발생된 상기 전기의 적어도 일부를 공급하는 단계;
(d) 상기 전기화학소자가 압축 수소 스트림을 생산하도록, 상기 수소를 포함하는 생성물 혼합물의 적어도 일부를 상기 전기화학소자에 공급하는 단계; 및
(e) 상기 압축 수소 스트림의 적어도 일부를 연속적으로 또는 간헐적으로 적어도 하나의 수소 최종-사용자 및/또는 적어도 하나의 저장 유닛에 공급하는 단계
를 포함하는, 메탄을 포함하는 공급스트림으로부터 압축 수소 가스를 생산하는 방법.
- 제1항에 있어서,
상기 전기분해 장치를 적어도 부분적으로 가동시키기 위해, 상기 고체 산화물 연료 전지에 의해 발생된 전기의 적어도 또 다른 일부가 상기 전기분해 장치에 공급되는, 방법.
- 제2항에 있어서,
상기 전기분해 장치가 물 및 상기 고체 산화물 연료 전지에 의해 생산된 전기의 적어도 일부를 채용(intake)함으로써 수소를 포함하는 배출 스트림을 생산하는, 방법.
- 제1항 내지 제3항 중 어느 한 항에 있어서,
상기 전기화학소자가 상기 압축 수소 스트림을 생산하도록, 상기 수소를 포함하는 생성물 혼합물이 상기 전기화학소자에 연속적으로 또는 간헐적으로 공급되는, 방법.
- (a) (i) 일 측부가 고체 산화물 연료 전지의 애노드 챔버에 노출되어 있고, 타 측부가 전해질에 부착되어 있는 애노드; 및 (ii) 일 측부가 상기 고체 산화물 연료 전지의 캐소드(cathode) 챔버에 부착되어 있고, 타 측부가 상기 전해질에 부착되어 있는 캐소드를 갖는 상기 고체 산화물 연료 전지; 및
(b) 기상 수소를 추출하고 압축시켜 압축 수소 스트림을 생산할 수 있는 전기화학소자를 포함하되,
상기 애노드는 원소주기율표의 8, 9, 10 및 11족으로부터의 적어도 하나의 원소를 포함하는 개질 촉매 조성물을 포함하고;
상기 고체 산화물 연료 전지는 메탄을 포함하는 공급스트림과 증기 스트림을 상기 애노드 챔버 내로 받기 위한 적어도 하나의 주입구(inlet), 및 산소-함유 유입 스트림을 상기 캐소드 챔버 내로 받기 위한 또 다른 적어도 하나의 주입구를 가지며;
상기 애노드는 상기 메탄을 포함하는 공급스트림과 상기 증기 스트림으로부터 수소를 포함하는 생성물 혼합물을 발생시킬 수 있고;
상기 고체 산화물 연료 전지는 상기 수소를 포함하는 생성 혼합물의 적어도 일부로부터 전기를 발생시킬 수 있으며;
상기 고체 산화물 연료 전지는, 상기 고체 산화물 연료 전지에 의해 생산되는 상기 전기의 적어도 일부에 의해 상기 전기화학소자를 적어도 부분적으로 가동시키기 위해 제1 전기적 연결부에 의해 상기 전기화학소자에 연결되고;
제1 라인이, 상기 수소를 포함하는 생성물 혼합물의 적어도 일부를 상기 고체 산화물 연료 전지로부터 상기 전기화학소자에 공급하며;
상기 전기화학소자는 상기 압축 수소 스트림을 일시적으로 또는 연속적으로 적어도 하나의 수소 최종-사용자 및/또는 적어도 하나의 저장 유닛으로 배출하기 위해 제2 라인에 연결된 배출구(outlet)를 갖는,
압축 수소를 생산하는 장치.
- 제5항에 있어서,
상기 고체 산화물 연료 전지는, 상기 고체 산화물 연료 전지에 의해 생산되는 상기 전기의 적어도 일부에 의해 전기분해 장치를 적어도 부분적으로 가동시키기 위해 제2 전기적 연결부에 의해 상기 전기분해 장치에 연결되는, 장치.
- 제6항에 있어서,
상기 전기분해 장치가 물 및 상기 고체 산화물 연료 전지에 의해 생산되는 전기의 적어도 일부를 채용함으로써, 수소를 포함하는 배출 스트림을 생산할 수 있는, 장치.
- 제6항 또는 제7항에 있어서,
상기 전기분해 장치가 제3 라인에 의해 상기 전기화학소자에 연결된 배출부를 가지며, 상기 라인은 상기 수소를 포함하는 배출 스트림의 적어도 일부를 연속적으로 또는 간헐적으로 상기 전기화학소자에 공급하여 압축 수소 스트림을 생산할 수 있는, 장치.
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP15202758.7 | 2015-12-24 | ||
| EP15202758 | 2015-12-24 | ||
| PCT/EP2016/082368 WO2017109065A1 (en) | 2015-12-24 | 2016-12-22 | Process and an apparatus for the production of compressed hydrogen |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| KR20180098252A true KR20180098252A (ko) | 2018-09-03 |
Family
ID=55069741
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020187017202A Ceased KR20180098252A (ko) | 2015-12-24 | 2016-12-22 | 압축 수소를 생산하는 방법 및 장치 |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US20190006690A1 (ko) |
| EP (1) | EP3394922B1 (ko) |
| KR (1) | KR20180098252A (ko) |
| ES (1) | ES2962359T3 (ko) |
| PL (1) | PL3394922T3 (ko) |
| WO (1) | WO2017109065A1 (ko) |
Families Citing this family (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE112017005364T5 (de) * | 2016-10-24 | 2019-07-18 | Precision Combustion, Inc. | Regenerativer festoxidstapel |
| US20200227764A1 (en) * | 2019-01-14 | 2020-07-16 | W. Grover Coors | Combined Hydrogen and Electrical Power Generation System and Method |
| DE102019128934A1 (de) * | 2019-10-25 | 2021-04-29 | Forschungszentrum Jülich GmbH | Herstellung eines Synthesegases umfassend Kohlenmonoxid und Wasserstoff |
| CN117480275A (zh) * | 2021-07-08 | 2024-01-30 | 环球公用事业公司 | 集成氢气产生方法和系统 |
Family Cites Families (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2001297779A (ja) * | 2000-04-13 | 2001-10-26 | Matsushita Electric Ind Co Ltd | 燃料電池システム |
| JP2003282122A (ja) * | 2002-02-19 | 2003-10-03 | Proton Energy Systems Inc | エネルギ蓄積および回収システムならびにその使用方法 |
| US7482078B2 (en) * | 2003-04-09 | 2009-01-27 | Bloom Energy Corporation | Co-production of hydrogen and electricity in a high temperature electrochemical system |
| US7955491B2 (en) * | 2004-09-14 | 2011-06-07 | Honda Motor Co., Ltd. | Methods, devices, and infrastructure systems for separating, removing, compressing, and generating hydrogen |
| EP2064766A4 (en) * | 2006-09-06 | 2010-09-29 | Bloom Energy Corp | FLEXIBLE FUEL CELL SYSTEM CONFIGURATION FOR HANDLING OF MULTIPLE FUELS |
| US20080292921A1 (en) * | 2007-05-22 | 2008-11-27 | Balasubramanian Lakshmanan | Recovery of inert gas from a fuel cell exhaust stream |
| US20100266923A1 (en) * | 2009-04-15 | 2010-10-21 | Bloom Energy Corporation | Fuel cell system with electrochemical hydrogen pump and method of operating same |
-
2016
- 2016-12-22 ES ES16822168T patent/ES2962359T3/es active Active
- 2016-12-22 US US16/064,508 patent/US20190006690A1/en not_active Abandoned
- 2016-12-22 EP EP16822168.7A patent/EP3394922B1/en active Active
- 2016-12-22 KR KR1020187017202A patent/KR20180098252A/ko not_active Ceased
- 2016-12-22 PL PL16822168.7T patent/PL3394922T3/pl unknown
- 2016-12-22 WO PCT/EP2016/082368 patent/WO2017109065A1/en not_active Ceased
Also Published As
| Publication number | Publication date |
|---|---|
| EP3394922B1 (en) | 2023-10-11 |
| EP3394922A1 (en) | 2018-10-31 |
| US20190006690A1 (en) | 2019-01-03 |
| EP3394922C0 (en) | 2023-10-11 |
| ES2962359T3 (es) | 2024-03-18 |
| WO2017109065A1 (en) | 2017-06-29 |
| PL3394922T3 (pl) | 2024-01-22 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Chisholm et al. | Hydrogen from water electrolysis | |
| Pan et al. | High-yield electrochemical upgrading of CO2 into CH4 using large-area protonic ceramic electrolysis cells | |
| Ivanova et al. | Technological pathways to produce compressed and highly pure hydrogen from solar power | |
| DK3077574T3 (en) | PROCEDURE FOR OPERATING A SOEC-TYPE STACT REACTOR TO MAKE METHAN IN THE EVENT OF AVAILABLE ELECTRICITY | |
| CA3028495C (en) | Process for starting mode or stand-by mode operation of a power-to-gas unit comprising a plurality of high-temperature electrolysis (soec) or co-electrolysis reactors | |
| CA3013415C (en) | Carbon monoxide production process optimized by soec | |
| CA3005639A1 (en) | Energy storage using an rep with an engine | |
| EP3394922B1 (en) | Process and an apparatus for the production of compressed hydrogen | |
| Fujiwara et al. | Power-to-gas systems utilizing methanation reaction in solid oxide electrolysis cell cathodes: a model-based study | |
| JP4761195B2 (ja) | 水素製造装置 | |
| Reichelt et al. | Co-electrolysis | |
| JP2021017468A (ja) | エネルギ変換システム | |
| US20260028735A1 (en) | Methods and systems for power-to-fuel applications using tail gas from fuel synthesis and/or methane in high temperature electrolyzer | |
| US20260028737A1 (en) | Methods and systems for power-to-fuel applications using tail gas from fuel synthesis and/or methane in high temperature electrolyzer | |
| US20230078714A1 (en) | Hydrogen system and method for operating hydrogen system | |
| US12331417B1 (en) | Methods and systems for the conversion of carbon dioxide to chemicals and/or fuels utilizing steam electrolysis | |
| US20250197328A1 (en) | Methods and systems for the conversion of carbon dioxide to chemicals and/or fuels utilizing steam electrolysis | |
| Kiros et al. | Hydrogen through water electrolysis and biomass gasification for application in fuel cells | |
| Yildiz et al. | Fuel cells | |
| YILDIZ et al. | Department of Chemistry Hacettepe University | |
| Ismail | 1 nrnnrr11 DC) Based | |
| Fellet et al. | Power-to-gas plants use renewable energy to make sustainable fuel | |
| Garche | Electrochemical hydrogen compressor | |
| Lamy | Operation of fuel cells with |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PA0105 | International application |
Patent event date: 20180618 Patent event code: PA01051R01D Comment text: International Patent Application |
|
| PG1501 | Laying open of application | ||
| A201 | Request for examination | ||
| PA0201 | Request for examination |
Patent event code: PA02012R01D Patent event date: 20211214 Comment text: Request for Examination of Application |
|
| E902 | Notification of reason for refusal | ||
| PE0902 | Notice of grounds for rejection |
Comment text: Notification of reason for refusal Patent event date: 20240619 Patent event code: PE09021S01D |
|
| E601 | Decision to refuse application | ||
| PE0601 | Decision on rejection of patent |
Patent event date: 20250205 Comment text: Decision to Refuse Application Patent event code: PE06012S01D |