JP6532854B2 - レーザ水晶体嚢硝子体切開術 - Google Patents
レーザ水晶体嚢硝子体切開術 Download PDFInfo
- Publication number
- JP6532854B2 JP6532854B2 JP2016500433A JP2016500433A JP6532854B2 JP 6532854 B2 JP6532854 B2 JP 6532854B2 JP 2016500433 A JP2016500433 A JP 2016500433A JP 2016500433 A JP2016500433 A JP 2016500433A JP 6532854 B2 JP6532854 B2 JP 6532854B2
- Authority
- JP
- Japan
- Prior art keywords
- lens
- laser
- eye
- oct
- capsule
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
- 239000002775 capsule Substances 0.000 claims description 65
- 238000003384 imaging method Methods 0.000 claims description 27
- 230000003287 optical effect Effects 0.000 claims description 23
- 238000001356 surgical procedure Methods 0.000 claims description 19
- 238000011282 treatment Methods 0.000 claims description 15
- 238000000926 separation method Methods 0.000 claims description 10
- 230000015572 biosynthetic process Effects 0.000 claims description 9
- 230000006378 damage Effects 0.000 claims description 4
- 238000012014 optical coherence tomography Methods 0.000 description 60
- 238000000034 method Methods 0.000 description 42
- 208000002177 Cataract Diseases 0.000 description 16
- 230000010287 polarization Effects 0.000 description 13
- 230000008901 benefit Effects 0.000 description 10
- 230000033001 locomotion Effects 0.000 description 10
- 230000003750 conditioning effect Effects 0.000 description 8
- 238000001514 detection method Methods 0.000 description 8
- 239000012528 membrane Substances 0.000 description 8
- 210000002159 anterior chamber Anatomy 0.000 description 7
- 230000006870 function Effects 0.000 description 6
- 239000011521 glass Substances 0.000 description 5
- 238000005286 illumination Methods 0.000 description 5
- 238000002604 ultrasonography Methods 0.000 description 5
- 230000004075 alteration Effects 0.000 description 4
- 230000008859 change Effects 0.000 description 4
- 238000002059 diagnostic imaging Methods 0.000 description 4
- 238000000605 extraction Methods 0.000 description 3
- 239000000835 fiber Substances 0.000 description 3
- 238000003780 insertion Methods 0.000 description 3
- 230000037431 insertion Effects 0.000 description 3
- 239000007788 liquid Substances 0.000 description 3
- 230000004048 modification Effects 0.000 description 3
- 238000012986 modification Methods 0.000 description 3
- 230000008569 process Effects 0.000 description 3
- 238000011269 treatment regimen Methods 0.000 description 3
- 201000004569 Blindness Diseases 0.000 description 2
- 238000013459 approach Methods 0.000 description 2
- 201000009310 astigmatism Diseases 0.000 description 2
- 230000004888 barrier function Effects 0.000 description 2
- 230000015556 catabolic process Effects 0.000 description 2
- 238000009792 diffusion process Methods 0.000 description 2
- 239000006185 dispersion Substances 0.000 description 2
- 238000006073 displacement reaction Methods 0.000 description 2
- 238000004945 emulsification Methods 0.000 description 2
- 238000005516 engineering process Methods 0.000 description 2
- 238000000799 fluorescence microscopy Methods 0.000 description 2
- 239000007789 gas Substances 0.000 description 2
- CPBQJMYROZQQJC-UHFFFAOYSA-N helium neon Chemical compound [He].[Ne] CPBQJMYROZQQJC-UHFFFAOYSA-N 0.000 description 2
- 238000002513 implantation Methods 0.000 description 2
- 229920002521 macromolecule Polymers 0.000 description 2
- 238000003032 molecular docking Methods 0.000 description 2
- 238000000059 patterning Methods 0.000 description 2
- 102000004169 proteins and genes Human genes 0.000 description 2
- 108090000623 proteins and genes Proteins 0.000 description 2
- 230000000717 retained effect Effects 0.000 description 2
- 210000001525 retina Anatomy 0.000 description 2
- KIUKXJAPPMFGSW-DNGZLQJQSA-N (2S,3S,4S,5R,6R)-6-[(2S,3R,4R,5S,6R)-3-Acetamido-2-[(2S,3S,4R,5R,6R)-6-[(2R,3R,4R,5S,6R)-3-acetamido-2,5-dihydroxy-6-(hydroxymethyl)oxan-4-yl]oxy-2-carboxy-4,5-dihydroxyoxan-3-yl]oxy-5-hydroxy-6-(hydroxymethyl)oxan-4-yl]oxy-3,4,5-trihydroxyoxane-2-carboxylic acid Chemical compound CC(=O)N[C@H]1[C@H](O)O[C@H](CO)[C@@H](O)[C@@H]1O[C@H]1[C@H](O)[C@@H](O)[C@H](O[C@H]2[C@@H]([C@@H](O[C@H]3[C@@H]([C@@H](O)[C@H](O)[C@H](O3)C(O)=O)O)[C@H](O)[C@@H](CO)O2)NC(C)=O)[C@@H](C(O)=O)O1 KIUKXJAPPMFGSW-DNGZLQJQSA-N 0.000 description 1
- 206010002945 Aphakia Diseases 0.000 description 1
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical compound [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 description 1
- 238000002679 ablation Methods 0.000 description 1
- 238000010521 absorption reaction Methods 0.000 description 1
- 230000004308 accommodation Effects 0.000 description 1
- 238000004458 analytical method Methods 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- 230000005540 biological transmission Effects 0.000 description 1
- 230000004397 blinking Effects 0.000 description 1
- 150000001875 compounds Chemical class 0.000 description 1
- 238000004590 computer program Methods 0.000 description 1
- 210000004087 cornea Anatomy 0.000 description 1
- 230000008878 coupling Effects 0.000 description 1
- 238000010168 coupling process Methods 0.000 description 1
- 238000005859 coupling reaction Methods 0.000 description 1
- 238000010586 diagram Methods 0.000 description 1
- 238000002224 dissection Methods 0.000 description 1
- 239000003814 drug Substances 0.000 description 1
- 229940079593 drug Drugs 0.000 description 1
- 230000009977 dual effect Effects 0.000 description 1
- 230000000694 effects Effects 0.000 description 1
- 238000001914 filtration Methods 0.000 description 1
- 239000012530 fluid Substances 0.000 description 1
- 229920002674 hyaluronan Polymers 0.000 description 1
- 229960003160 hyaluronic acid Drugs 0.000 description 1
- 238000007654 immersion Methods 0.000 description 1
- 239000007943 implant Substances 0.000 description 1
- 230000001678 irradiating effect Effects 0.000 description 1
- 230000002262 irrigation Effects 0.000 description 1
- 238000003973 irrigation Methods 0.000 description 1
- 208000018769 loss of vision Diseases 0.000 description 1
- 231100000864 loss of vision Toxicity 0.000 description 1
- 238000012423 maintenance Methods 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- 230000001404 mediated effect Effects 0.000 description 1
- 238000000386 microscopy Methods 0.000 description 1
- 238000012544 monitoring process Methods 0.000 description 1
- 208000001491 myopia Diseases 0.000 description 1
- 230000004379 myopia Effects 0.000 description 1
- 239000013307 optical fiber Substances 0.000 description 1
- 238000012634 optical imaging Methods 0.000 description 1
- 238000000399 optical microscopy Methods 0.000 description 1
- 230000008447 perception Effects 0.000 description 1
- 229920000642 polymer Polymers 0.000 description 1
- 230000003252 repetitive effect Effects 0.000 description 1
- 230000002207 retinal effect Effects 0.000 description 1
- 230000000630 rising effect Effects 0.000 description 1
- 230000011218 segmentation Effects 0.000 description 1
- 239000004065 semiconductor Substances 0.000 description 1
- 229910052710 silicon Inorganic materials 0.000 description 1
- 239000010703 silicon Substances 0.000 description 1
- 239000003381 stabilizer Substances 0.000 description 1
- 238000006467 substitution reaction Methods 0.000 description 1
- 230000008685 targeting Effects 0.000 description 1
- 238000012546 transfer Methods 0.000 description 1
- 230000004393 visual impairment Effects 0.000 description 1
- 210000004127 vitreous body Anatomy 0.000 description 1
- 238000004383 yellowing Methods 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F9/00—Methods or devices for treatment of the eyes; Devices for putting-in contact lenses; Devices to correct squinting; Apparatus to guide the blind; Protective devices for the eyes, carried on the body or in the hand
- A61F9/007—Methods or devices for eye surgery
- A61F9/008—Methods or devices for eye surgery using laser
- A61F9/00825—Methods or devices for eye surgery using laser for photodisruption
- A61F9/00834—Inlays; Onlays; Intraocular lenses [IOL]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F9/00—Methods or devices for treatment of the eyes; Devices for putting-in contact lenses; Devices to correct squinting; Apparatus to guide the blind; Protective devices for the eyes, carried on the body or in the hand
- A61F9/007—Methods or devices for eye surgery
- A61F9/008—Methods or devices for eye surgery using laser
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F9/00—Methods or devices for treatment of the eyes; Devices for putting-in contact lenses; Devices to correct squinting; Apparatus to guide the blind; Protective devices for the eyes, carried on the body or in the hand
- A61F9/007—Methods or devices for eye surgery
- A61F9/008—Methods or devices for eye surgery using laser
- A61F9/00825—Methods or devices for eye surgery using laser for photodisruption
- A61F9/00836—Flap cutting
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F9/00—Methods or devices for treatment of the eyes; Devices for putting-in contact lenses; Devices to correct squinting; Apparatus to guide the blind; Protective devices for the eyes, carried on the body or in the hand
- A61F9/007—Methods or devices for eye surgery
- A61F9/008—Methods or devices for eye surgery using laser
- A61F2009/00861—Methods or devices for eye surgery using laser adapted for treatment at a particular location
- A61F2009/0087—Lens
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F9/00—Methods or devices for treatment of the eyes; Devices for putting-in contact lenses; Devices to correct squinting; Apparatus to guide the blind; Protective devices for the eyes, carried on the body or in the hand
- A61F9/007—Methods or devices for eye surgery
- A61F9/008—Methods or devices for eye surgery using laser
- A61F2009/00885—Methods or devices for eye surgery using laser for treating a particular disease
- A61F2009/00887—Cataract
- A61F2009/00889—Capsulotomy
Landscapes
- Health & Medical Sciences (AREA)
- Ophthalmology & Optometry (AREA)
- Heart & Thoracic Surgery (AREA)
- Vascular Medicine (AREA)
- Optics & Photonics (AREA)
- Surgery (AREA)
- Engineering & Computer Science (AREA)
- Biomedical Technology (AREA)
- Physics & Mathematics (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Laser Surgery Devices (AREA)
- Prostheses (AREA)
Description
本願は、2013年3月14日に出願された米国特許出願第61/785,672号の優先権主張出願であり、この米国特許出願を参照により引用し、全ての目的に関してその記載内容を本明細書の一部とする。ここにパリ条約上の優先権全体を明示的に保持する。
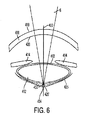
無傷の前硝子体表面の場合であっても、後嚢中の割れ目が水晶体嚢内摘出後の無水晶体眼のセグメントと同様な眼の前セグメントと後セグメントとの間の物理的バリヤを破断させる。このバリヤがないと、前眼房内への、ヒアルロン酸、硝子体ゲルの安定化剤の拡散が容易になる場合があり、この状況は、硝子体ゲルの潰れとして臨床的に顕在化する場合がある。
Claims (3)
- 水晶体嚢、前硝子体表面、及び、前記水晶体嚢の後方部分と前記前硝子体表面との間に位置するベルゲル腔を有する眼に対してレーザ補助手術を実施するシステムであって、
複数個のレーザパルスを含む治療ビームを生じさせるよう構成されたレーザ源と、
画像化組立体を含む一体形光学系と、
前記一体形光学系を制御するように構成された制御部と、
を備え、
前記画像化組立体及び治療レーザ送出組立体が、少なくとも1つの共通光学素子を共有し、
前記一体形光学系は、1つ又は2つ以上の標的組織構造体に関する画像情報を収集して前記治療ビームを3次元パターンで方向付けて前記標的組織構造体のうちの少なくとも1つの破壊を生じさせる、というように構成されており、
前記制御部は、前記一体形光学系を制御して前記ベルゲル腔内に複数個の気泡を形成し、それにより前記水晶体嚢の前記後方部分と前記前硝子体表面との間の離隔距離を増大させる、というように構成されている、システム。 - 前記制御部は、前記ベルゲル腔内への前記複数個の気泡の形成の次に、前記一体形光学系を制御して前記水晶体嚢の前記後方部分を切開させるように構成されている、請求項1記載のシステム。
- 前記制御部は、前記一体形光学系を制御して前記水晶体嚢の前記後方部分に対して水晶体嚢切開術を実施させるように構成されている、請求項2記載のシステム。
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201361785672P | 2013-03-14 | 2013-03-14 | |
| US61/785,672 | 2013-03-14 | ||
| PCT/US2014/018710 WO2014143555A1 (en) | 2013-03-14 | 2014-02-26 | Laser capsulovitreotomy |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2019095072A Division JP6698916B2 (ja) | 2013-03-14 | 2019-05-21 | レーザ水晶体嚢硝子体切開術 |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| JP2016510660A JP2016510660A (ja) | 2016-04-11 |
| JP2016510660A5 JP2016510660A5 (ja) | 2017-02-16 |
| JP6532854B2 true JP6532854B2 (ja) | 2019-06-19 |
Family
ID=50241574
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2016500433A Expired - Fee Related JP6532854B2 (ja) | 2013-03-14 | 2014-02-26 | レーザ水晶体嚢硝子体切開術 |
| JP2019095072A Active JP6698916B2 (ja) | 2013-03-14 | 2019-05-21 | レーザ水晶体嚢硝子体切開術 |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2019095072A Active JP6698916B2 (ja) | 2013-03-14 | 2019-05-21 | レーザ水晶体嚢硝子体切開術 |
Country Status (6)
| Country | Link |
|---|---|
| US (2) | US10022270B2 (ja) |
| EP (2) | EP3195838B1 (ja) |
| JP (2) | JP6532854B2 (ja) |
| AU (2) | AU2014228568B2 (ja) |
| CA (1) | CA2905321C (ja) |
| WO (1) | WO2014143555A1 (ja) |
Families Citing this family (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10022270B2 (en) * | 2013-03-14 | 2018-07-17 | Optimedica Corporation | Laser capsulovitreotomy |
| RU2704469C1 (ru) * | 2019-05-22 | 2019-10-28 | Федеральное государственное автономное учреждение "Национальный медицинский исследовательский центр "Межотраслевой научно-технический комплекс "Микрохирургия глаза" имени академика С.Н. Федорова Министерства здравоохранения Российской Федерации | Способ снижения риска интраоперационных геморрагических осложнений у детей с врожденной катарактой в сочетании с синдромом первичного персистирующего гиперпластического стекловидного тела |
| DE102020204622A1 (de) * | 2020-04-09 | 2021-10-14 | Trumpf Laser Gmbh | Verfahren und Bearbeitungsmaschine zur Werkstücklageerfassung mittels OCT |
| AU2022272129A1 (en) * | 2021-05-10 | 2023-10-19 | Alcon Inc. | Laser pulse selection using motorized shutter |
| WO2023062650A1 (en) | 2021-10-14 | 2023-04-20 | Appasamy Associates Pvt Ltd | SINGLE AND/OR DUAL (532 AND/OR 577) nm LASER DELIVERY SYSTEM ATTACHED TO AN OPHTHALMIC MICROSCOPE |
Family Cites Families (47)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4174389A (en) * | 1977-05-13 | 1979-11-13 | Cope Louise A | Ophthalmic use of collagenase |
| FR2442622A1 (fr) * | 1978-06-08 | 1980-06-27 | Aron Rosa Daniele | Appareil de chirurgie ophtalmologique |
| US4560383A (en) * | 1983-10-27 | 1985-12-24 | Leiske Larry G | Anterior chamber intraocular lens |
| US6111645A (en) | 1991-04-29 | 2000-08-29 | Massachusetts Institute Of Technology | Grating based phase control optical delay line |
| EP0581871B2 (en) | 1991-04-29 | 2009-08-12 | Massachusetts Institute Of Technology | Apparatus for optical imaging and measurement |
| US5984916A (en) | 1993-04-20 | 1999-11-16 | Lai; Shui T. | Ophthalmic surgical laser and method |
| JP3261244B2 (ja) | 1993-12-17 | 2002-02-25 | ブラザー工業株式会社 | 走査光学装置 |
| US5743902A (en) | 1995-01-23 | 1998-04-28 | Coherent, Inc. | Hand-held laser scanner |
| US6454761B1 (en) | 1995-01-30 | 2002-09-24 | Philip D. Freedman | Laser surgery device and method |
| JPH0955843A (ja) | 1995-08-10 | 1997-02-25 | Nec Corp | 画像データ送受信システム |
| US5720894A (en) | 1996-01-11 | 1998-02-24 | The Regents Of The University Of California | Ultrashort pulse high repetition rate laser system for biological tissue processing |
| US7655002B2 (en) | 1996-03-21 | 2010-02-02 | Second Sight Laser Technologies, Inc. | Lenticular refractive surgery of presbyopia, other refractive errors, and cataract retardation |
| US6019472A (en) | 1997-05-12 | 2000-02-01 | Koester; Charles J. | Contact lens element for examination or treatment of ocular tissues |
| US6027531A (en) * | 1997-10-14 | 2000-02-22 | Tassignon; Marie-Joseb. R. | Intraocular lens and method for preventing secondary opacification |
| US6053613A (en) | 1998-05-15 | 2000-04-25 | Carl Zeiss, Inc. | Optical coherence tomography with new interferometer |
| WO2002009619A2 (en) * | 2000-07-28 | 2002-02-07 | Kiyoshi Okada | Intraocular lenses with reduced complications |
| WO2002016951A2 (en) * | 2000-08-21 | 2002-02-28 | The General Hospital Corporation | Methods for diagnosing a neurodegenerative condition |
| DE10202036A1 (de) * | 2002-01-18 | 2003-07-31 | Zeiss Carl Meditec Ag | Femtosekunden Lasersystem zur präzisen Bearbeitung von Material und Gewebe |
| US8186357B2 (en) * | 2004-01-23 | 2012-05-29 | Rowiak Gmbh | Control device for a surgical laser |
| US8394084B2 (en) * | 2005-01-10 | 2013-03-12 | Optimedica Corporation | Apparatus for patterned plasma-mediated laser trephination of the lens capsule and three dimensional phaco-segmentation |
| US8262646B2 (en) | 2006-01-20 | 2012-09-11 | Lensar, Inc. | System and method for providing the shaped structural weakening of the human lens with a laser |
| US20110319875A1 (en) | 2007-01-19 | 2011-12-29 | Frieder Loesel | Apparatus and Method for Morphing a Three-Dimensional Target Surface into a Two-Dimensional Image for Use in Guiding a Laser Beam in Ocular Surgery |
| AU2008226828B2 (en) * | 2007-03-13 | 2012-08-16 | Amo Development, Llc | Apparatus for creating incisions to improve intraocular lens placement |
| DE102007028042B3 (de) | 2007-06-14 | 2008-08-07 | Universität Zu Lübeck | Verfahren zur Laserbearbeitung transparenter Materialien |
| US20090149841A1 (en) * | 2007-09-10 | 2009-06-11 | Kurtz Ronald M | Effective Laser Photodisruptive Surgery in a Gravity Field |
| DE112008002511T5 (de) * | 2007-09-18 | 2010-07-15 | LenSx Lasers, Inc., Aliso Viejo | Verfahren und Vorrichtungen für integrierte Kataraktchirurgie |
| US20100324543A1 (en) * | 2007-09-18 | 2010-12-23 | Kurtz Ronald M | Method And Apparatus For Integrating Cataract Surgery With Glaucoma Or Astigmatism Surgery |
| US7717907B2 (en) | 2007-12-17 | 2010-05-18 | Technolas Perfect Vision Gmbh | Method for intrastromal refractive surgery |
| US20100022996A1 (en) | 2008-07-25 | 2010-01-28 | Frey Rudolph W | Method and system for creating a bubble shield for laser lens procedures |
| US8382745B2 (en) | 2009-07-24 | 2013-02-26 | Lensar, Inc. | Laser system and method for astigmatic corrections in association with cataract treatment |
| US8506559B2 (en) * | 2009-11-16 | 2013-08-13 | Alcon Lensx, Inc. | Variable stage optical system for ophthalmic surgical laser |
| US20110184395A1 (en) * | 2009-12-23 | 2011-07-28 | Optimedica Corporation | Method for laser capsulotomy and lens conditioning |
| EP3138475B1 (en) * | 2010-01-22 | 2023-10-25 | AMO Development, LLC | Apparatus for automated placement of scanned laser capsulorhexis incisions |
| US9278028B2 (en) * | 2010-02-08 | 2016-03-08 | Optimedica Corporation | System and method for plasma-mediated modification of tissue |
| US8414564B2 (en) | 2010-02-18 | 2013-04-09 | Alcon Lensx, Inc. | Optical coherence tomographic system for ophthalmic surgery |
| US8403918B2 (en) * | 2010-04-01 | 2013-03-26 | John Taboada | Automated non-invasive capsulectomy and anterior segment surgical apparatus and method |
| WO2011151064A1 (de) * | 2010-06-03 | 2011-12-08 | Carl Zeiss Meditec Ag | Vorrichtung und verfahren zur glaskörperchirurgie |
| US8845624B2 (en) | 2010-06-25 | 2014-09-30 | Alcon LexSx, Inc. | Adaptive patient interface |
| US20120089134A1 (en) * | 2010-10-11 | 2012-04-12 | Christopher Horvath | Contactless Photodisruptive Laser assisted Cataract Surgery |
| US9622913B2 (en) * | 2011-05-18 | 2017-04-18 | Alcon Lensx, Inc. | Imaging-controlled laser surgical system |
| TW201406707A (zh) * | 2012-05-04 | 2014-02-16 | Acucela Inc | 用以治療糖尿病性視網膜病變及其他眼部疾病之方法 |
| CN104349751B (zh) * | 2012-05-30 | 2017-08-15 | 艾利克斯研发私人有限公司 | 反射同轴照明器 |
| CA2884235C (en) * | 2012-09-07 | 2021-05-25 | Optimedica Corporation | Methods and systems for performing a posterior capsulotomy and for laser eye surgery with a penetrated cornea |
| CA2904894C (en) * | 2013-03-13 | 2021-07-27 | Optimedica Corporation | Free floating support for laser eye surgery system |
| US10022270B2 (en) * | 2013-03-14 | 2018-07-17 | Optimedica Corporation | Laser capsulovitreotomy |
| EP3054907B1 (en) * | 2013-10-08 | 2019-04-03 | Optimedica Corporation | Laser eye surgery system calibration |
| US10363173B2 (en) * | 2014-02-04 | 2019-07-30 | Optimedica Corporation | Confocal detection to minimize capsulotomy overcut while dynamically running on the capsular surface |
-
2014
- 2014-02-26 US US14/190,915 patent/US10022270B2/en active Active
- 2014-02-26 AU AU2014228568A patent/AU2014228568B2/en not_active Ceased
- 2014-02-26 WO PCT/US2014/018710 patent/WO2014143555A1/en active Application Filing
- 2014-02-26 CA CA2905321A patent/CA2905321C/en active Active
- 2014-02-26 EP EP17152041.4A patent/EP3195838B1/en active Active
- 2014-02-26 EP EP14709516.0A patent/EP2967999B1/en active Active
- 2014-02-26 JP JP2016500433A patent/JP6532854B2/ja not_active Expired - Fee Related
-
2018
- 2018-07-13 US US16/035,507 patent/US10610410B2/en active Active
- 2018-08-29 AU AU2018222939A patent/AU2018222939B2/en not_active Ceased
-
2019
- 2019-05-21 JP JP2019095072A patent/JP6698916B2/ja active Active
Also Published As
| Publication number | Publication date |
|---|---|
| AU2018222939A1 (en) | 2018-09-20 |
| JP2019134978A (ja) | 2019-08-15 |
| EP2967999B1 (en) | 2017-04-19 |
| AU2018222939B2 (en) | 2020-09-17 |
| AU2014228568A1 (en) | 2015-09-24 |
| US20190008682A1 (en) | 2019-01-10 |
| US10022270B2 (en) | 2018-07-17 |
| EP3195838A1 (en) | 2017-07-26 |
| CA2905321A1 (en) | 2014-09-18 |
| JP2016510660A (ja) | 2016-04-11 |
| US20140276676A1 (en) | 2014-09-18 |
| CA2905321C (en) | 2021-10-12 |
| EP3195838B1 (en) | 2018-08-22 |
| WO2014143555A1 (en) | 2014-09-18 |
| US10610410B2 (en) | 2020-04-07 |
| JP6698916B2 (ja) | 2020-05-27 |
| EP2967999A1 (en) | 2016-01-20 |
| AU2014228568B2 (en) | 2018-06-14 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6487023B2 (ja) | 目組織および人工水晶体の変更システム | |
| US20220331155A1 (en) | Methods and systems for performing a posterior capsulotomy and for laser eye surgery with a penetrated cornea | |
| JP6161140B2 (ja) | 眼内レンズ | |
| US20110184395A1 (en) | Method for laser capsulotomy and lens conditioning | |
| JP6698916B2 (ja) | レーザ水晶体嚢硝子体切開術 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20170111 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20170111 |
|
| RD04 | Notification of resignation of power of attorney |
Free format text: JAPANESE INTERMEDIATE CODE: A7424 Effective date: 20170420 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20171023 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20171116 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A821 Effective date: 20180330 |
|
| RD02 | Notification of acceptance of power of attorney |
Free format text: JAPANESE INTERMEDIATE CODE: A7422 Effective date: 20180330 |
|
| RD04 | Notification of resignation of power of attorney |
Free format text: JAPANESE INTERMEDIATE CODE: A7424 Effective date: 20180405 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20180522 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20180822 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20181005 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20190326 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20190425 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20190522 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 6532854 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| LAPS | Cancellation because of no payment of annual fees |