JP6282265B2 - 再構成可能なステントグラフト送達システムおよび使用方法 - Google Patents
再構成可能なステントグラフト送達システムおよび使用方法 Download PDFInfo
- Publication number
- JP6282265B2 JP6282265B2 JP2015508949A JP2015508949A JP6282265B2 JP 6282265 B2 JP6282265 B2 JP 6282265B2 JP 2015508949 A JP2015508949 A JP 2015508949A JP 2015508949 A JP2015508949 A JP 2015508949A JP 6282265 B2 JP6282265 B2 JP 6282265B2
- Authority
- JP
- Japan
- Prior art keywords
- access port
- stent graft
- component
- port
- delivery system
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 238000000034 method Methods 0.000 title description 23
- 238000007789 sealing Methods 0.000 claims description 119
- 230000017531 blood circulation Effects 0.000 claims description 37
- 238000003780 insertion Methods 0.000 claims description 3
- 230000037431 insertion Effects 0.000 claims description 3
- 230000000903 blocking effect Effects 0.000 description 27
- 206010002329 Aneurysm Diseases 0.000 description 24
- 210000002254 renal artery Anatomy 0.000 description 22
- 210000001367 artery Anatomy 0.000 description 21
- 206010061307 Neck deformity Diseases 0.000 description 17
- 230000008878 coupling Effects 0.000 description 15
- 238000010168 coupling process Methods 0.000 description 15
- 238000005859 coupling reaction Methods 0.000 description 15
- 239000000463 material Substances 0.000 description 15
- 230000007246 mechanism Effects 0.000 description 13
- 210000001363 mesenteric artery superior Anatomy 0.000 description 12
- 238000011282 treatment Methods 0.000 description 12
- 230000002792 vascular Effects 0.000 description 12
- 210000004204 blood vessel Anatomy 0.000 description 10
- 230000006870 function Effects 0.000 description 9
- 210000000709 aorta Anatomy 0.000 description 8
- 239000007943 implant Substances 0.000 description 7
- 210000000702 aorta abdominal Anatomy 0.000 description 6
- 210000003090 iliac artery Anatomy 0.000 description 6
- 238000002627 tracheal intubation Methods 0.000 description 6
- 229920002614 Polyether block amide Polymers 0.000 description 4
- 238000013459 approach Methods 0.000 description 4
- 229910001000 nickel titanium Inorganic materials 0.000 description 4
- 229920000642 polymer Polymers 0.000 description 4
- 229920001296 polysiloxane Polymers 0.000 description 4
- 208000002223 abdominal aortic aneurysm Diseases 0.000 description 3
- 229920001577 copolymer Polymers 0.000 description 3
- 210000001105 femoral artery Anatomy 0.000 description 3
- 239000012530 fluid Substances 0.000 description 3
- HLXZNVUGXRDIFK-UHFFFAOYSA-N nickel titanium Chemical compound [Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ti].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni].[Ni] HLXZNVUGXRDIFK-UHFFFAOYSA-N 0.000 description 3
- KZVVGZKAVZUACK-BJILWQEISA-N rilpivirine hydrochloride Chemical compound Cl.CC1=CC(\C=C\C#N)=CC(C)=C1NC1=CC=NC(NC=2C=CC(=CC=2)C#N)=N1 KZVVGZKAVZUACK-BJILWQEISA-N 0.000 description 3
- 210000005166 vasculature Anatomy 0.000 description 3
- PXHVJJICTQNCMI-UHFFFAOYSA-N Nickel Chemical compound [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 description 2
- 229920000299 Nylon 12 Polymers 0.000 description 2
- 229920006099 Vestamid® Polymers 0.000 description 2
- 239000004676 acrylonitrile butadiene styrene Substances 0.000 description 2
- 208000007474 aortic aneurysm Diseases 0.000 description 2
- 230000004888 barrier function Effects 0.000 description 2
- 230000008901 benefit Effects 0.000 description 2
- MTAZNLWOLGHBHU-UHFFFAOYSA-N butadiene-styrene rubber Chemical compound C=CC=C.C=CC1=CC=CC=C1 MTAZNLWOLGHBHU-UHFFFAOYSA-N 0.000 description 2
- 238000013461 design Methods 0.000 description 2
- 239000004744 fabric Substances 0.000 description 2
- 238000002955 isolation Methods 0.000 description 2
- 229910052751 metal Inorganic materials 0.000 description 2
- 239000002184 metal Substances 0.000 description 2
- -1 polyethylene Polymers 0.000 description 2
- 229920002635 polyurethane Polymers 0.000 description 2
- 239000004814 polyurethane Substances 0.000 description 2
- 239000010935 stainless steel Substances 0.000 description 2
- 229910001220 stainless steel Inorganic materials 0.000 description 2
- 238000001356 surgical procedure Methods 0.000 description 2
- 238000013519 translation Methods 0.000 description 2
- 238000011144 upstream manufacturing Methods 0.000 description 2
- 231100000216 vascular lesion Toxicity 0.000 description 2
- 210000003462 vein Anatomy 0.000 description 2
- AILDTIZEPVHXBF-UHFFFAOYSA-N Argentine Natural products C1C(C2)C3=CC=CC(=O)N3CC1CN2C(=O)N1CC(C=2N(C(=O)C=CC=2)C2)CC2C1 AILDTIZEPVHXBF-UHFFFAOYSA-N 0.000 description 1
- VYZAMTAEIAYCRO-UHFFFAOYSA-N Chromium Chemical compound [Cr] VYZAMTAEIAYCRO-UHFFFAOYSA-N 0.000 description 1
- 229920004934 Dacron® Polymers 0.000 description 1
- 239000004698 Polyethylene Substances 0.000 description 1
- 244000308495 Potentilla anserina Species 0.000 description 1
- 235000016594 Potentilla anserina Nutrition 0.000 description 1
- 239000002174 Styrene-butadiene Substances 0.000 description 1
- 239000004699 Ultra-high molecular weight polyethylene Substances 0.000 description 1
- 210000001015 abdomen Anatomy 0.000 description 1
- 230000005856 abnormality Effects 0.000 description 1
- XECAHXYUAAWDEL-UHFFFAOYSA-N acrylonitrile butadiene styrene Chemical compound C=CC=C.C=CC#N.C=CC1=CC=CC=C1 XECAHXYUAAWDEL-UHFFFAOYSA-N 0.000 description 1
- 229920000122 acrylonitrile butadiene styrene Polymers 0.000 description 1
- 230000001154 acute effect Effects 0.000 description 1
- 210000002376 aorta thoracic Anatomy 0.000 description 1
- 239000010953 base metal Substances 0.000 description 1
- 239000008280 blood Substances 0.000 description 1
- 210000004369 blood Anatomy 0.000 description 1
- 210000001715 carotid artery Anatomy 0.000 description 1
- 229910052804 chromium Inorganic materials 0.000 description 1
- 239000011651 chromium Substances 0.000 description 1
- 238000004140 cleaning Methods 0.000 description 1
- 239000010941 cobalt Substances 0.000 description 1
- 229910017052 cobalt Inorganic materials 0.000 description 1
- GUTLYIVDDKVIGB-UHFFFAOYSA-N cobalt atom Chemical compound [Co] GUTLYIVDDKVIGB-UHFFFAOYSA-N 0.000 description 1
- 239000002131 composite material Substances 0.000 description 1
- 210000004351 coronary vessel Anatomy 0.000 description 1
- 238000011461 current therapy Methods 0.000 description 1
- 229940022769 d- lactic acid Drugs 0.000 description 1
- 229940084014 edurant Drugs 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 238000010438 heat treatment Methods 0.000 description 1
- 230000003993 interaction Effects 0.000 description 1
- 238000013152 interventional procedure Methods 0.000 description 1
- 230000003902 lesion Effects 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 229910001092 metal group alloy Inorganic materials 0.000 description 1
- 150000002739 metals Chemical class 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 239000002991 molded plastic Substances 0.000 description 1
- 210000003739 neck Anatomy 0.000 description 1
- 229910052759 nickel Inorganic materials 0.000 description 1
- 150000002825 nitriles Chemical class 0.000 description 1
- 238000002355 open surgical procedure Methods 0.000 description 1
- 229920000728 polyester Polymers 0.000 description 1
- 229920000573 polyethylene Polymers 0.000 description 1
- 239000005020 polyethylene terephthalate Substances 0.000 description 1
- 229920001195 polyisoprene Polymers 0.000 description 1
- 229920001343 polytetrafluoroethylene Polymers 0.000 description 1
- 239000004810 polytetrafluoroethylene Substances 0.000 description 1
- 238000011084 recovery Methods 0.000 description 1
- 230000000717 retained effect Effects 0.000 description 1
- 230000002441 reversible effect Effects 0.000 description 1
- 230000002966 stenotic effect Effects 0.000 description 1
- 239000011115 styrene butadiene Substances 0.000 description 1
- 229920003048 styrene butadiene rubber Polymers 0.000 description 1
- 229910000601 superalloy Inorganic materials 0.000 description 1
- 239000003356 suture material Substances 0.000 description 1
- 230000001960 triggered effect Effects 0.000 description 1
- 229920000785 ultra high molecular weight polyethylene Polymers 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/95—Instruments specially adapted for placement or removal of stents or stent-grafts
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/95—Instruments specially adapted for placement or removal of stents or stent-grafts
- A61F2/9517—Instruments specially adapted for placement or removal of stents or stent-grafts handle assemblies therefor
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M39/00—Tubes, tube connectors, tube couplings, valves, access sites or the like, specially adapted for medical use
- A61M39/02—Access sites
- A61M39/06—Haemostasis valves, i.e. gaskets sealing around a needle, catheter or the like, closing on removal thereof
- A61M2039/062—Haemostasis valves, i.e. gaskets sealing around a needle, catheter or the like, closing on removal thereof used with a catheter
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M39/00—Tubes, tube connectors, tube couplings, valves, access sites or the like, specially adapted for medical use
- A61M39/02—Access sites
- A61M39/06—Haemostasis valves, i.e. gaskets sealing around a needle, catheter or the like, closing on removal thereof
- A61M2039/0633—Haemostasis valves, i.e. gaskets sealing around a needle, catheter or the like, closing on removal thereof the seal being a passive seal made of a resilient material with or without an opening
- A61M2039/0646—Duckbill-valve
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M39/00—Tubes, tube connectors, tube couplings, valves, access sites or the like, specially adapted for medical use
- A61M39/08—Tubes; Storage means specially adapted therefor
- A61M2039/082—Multi-lumen tubes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61M—DEVICES FOR INTRODUCING MEDIA INTO, OR ONTO, THE BODY; DEVICES FOR TRANSDUCING BODY MEDIA OR FOR TAKING MEDIA FROM THE BODY; DEVICES FOR PRODUCING OR ENDING SLEEP OR STUPOR
- A61M39/00—Tubes, tube connectors, tube couplings, valves, access sites or the like, specially adapted for medical use
- A61M39/02—Access sites
- A61M39/06—Haemostasis valves, i.e. gaskets sealing around a needle, catheter or the like, closing on removal thereof
- A61M39/0606—Haemostasis valves, i.e. gaskets sealing around a needle, catheter or the like, closing on removal thereof without means for adjusting the seal opening or pressure
Description
Claims (20)
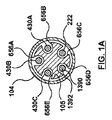
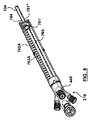
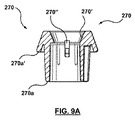
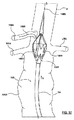
- ステントグラフト送達システムのための構成変更可能なポート構造体であって、
その中を通る内部アクセスポートを画成する内側密閉部品と、
前記内側密閉部品に着脱可能に連結され、その中を通る外部アクセスポートを画成する外側密閉部品と、を備え、
前記外部アクセスポートは、前記外側密閉部品および前記内側密閉部品が互いに対して連結されたとき、その中を通って延びるガイドワイヤに対する血流遮断を保持するよう構成され、
前記内部アクセスポートは、前記外側密閉部品が前記内側密閉部品から取り外されたとき、その中を通って延びるステントグラフト送達システムに対して血流遮断を保持するよう構成されている、
ことを特徴とするポート構造体。 - 前記外側密閉部品は、前記外部アクセスポートを画成する密閉部分と、前記外部アクセスポートから遠位方向に延びるその中を通るガイドワイヤ通路を画成する連結部分とを備えている、
請求項1に記載のポート構造体。 - 前記内側密閉部品の内部アクセスポートは、前記外側密閉部品の連結部分をその中に受容し、それにより前記内側密閉部品と前記外側密閉部品とを着脱可能に連結する寸法を有する、
請求項2に記載のポート構造体。 - 前記内側密閉部品は、前記外側密閉部品が前記内側密閉部品を介して前記ステントグラフト送達システムのハンドル部品に着脱可能に連結されるよう、前記ハンドル部品に連結される、
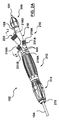
請求項1に記載のポート構造体。 - 長尺ガイドワイヤチューブの管腔がその中を通してガイドワイヤを受容するために前記外部アクセスポートから延びるよう、前記ガイドワイヤチューブが前記外側密閉部品に取り付けられる、
請求項1に記載のポート構造体。 - 前記ガイドワイヤチューブは、前記内側密閉部品および前記外側密閉部品が一緒に連結されたとき、前記内側密閉部品を通る、
請求項5に記載のポート構造体。 - 前記内側密閉部品は、取り付けられたガイドワイヤチューブを有する前記外側密閉部品が前記内側密閉部品を介して前記ステントグラフト送達システムのハンドル部品に着脱可能に連結されるよう、前記ハンドル部品に固定的に取り付けられる、
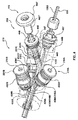
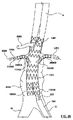
請求項6に記載のポート構造体。 - 前記外側密閉部品の前記外部アクセスポートに係合し且つ前記外部アクセスポートを拡張させるための着脱可能なアクセスポートダイレータであって、前記外部アクセスポートを通して挿入されるためにその中をガイドワイヤが誘導される管腔を画成する、アクセスポートダイレータをさらに備えている、
請求項1に記載のポート構造体。 - 前記アクセスポートダイレータは、前記外部アクセスポートを形成する成形済み穴を拡張させるために前記外部アクセスポート内で摺動する小径雄部分を備えている、
請求項8に記載のポート構造体。 - 前記内部アクセスポートは、前記外側密閉部品が前記内側密閉部品から取り外されたとき、13フレンチまでの医療装置をその中を通して受容するための寸法を有する、
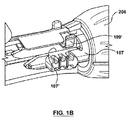
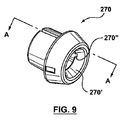
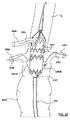
請求項1に記載のポート構造体。 - ステントグラフト送達システムのためのポート構造体であって、
その中を通る内部アクセスポートを画成する内側密閉部品と、
その中を通る外部アクセスポートを画成する密閉部分と、前記内側密閉部品の内部アクセスポート内で前記内側密閉部品にスナップ嵌合しそれにより前記内側密閉部品および前記外側密閉部品を着脱可能に一緒に連結する連結部分とを有する外側密閉部品を、を備え、
ことを特徴とするポート構造体。 - 前記外側密閉部品および前記内側密閉部品が互いに対して連結されたとき、前記外部アクセスポートは、その中を通って延びるガイドワイヤに対する血流遮断を保持するよう構成される、
請求項11に記載のポート構造体。 - 前記外側密閉部品が前記内側密閉部品から取り外されたとき、前記内部アクセスポートは、その中を通って延びるステントグラフト送達システムに対して血流遮断を保持するよう構成される、
請求項11に記載のポート構造体。 - 前記内部アクセスポートは、前記外側密閉部品が前記内側密閉部品から取り外されたとき、13フレンチまでの医療装置をその中を通して受容するための寸法を有する、
請求項11に記載のポート構造体。 - 長尺ガイドワイヤチューブの管腔がその中を通してガイドワイヤを受容するために前記外部アクセスポートから延びるよう、前記ガイドワイヤチューブが前記外側密閉部品に取り付けられる、
請求項1に記載のポート構造体。 - 前記ガイドワイヤチューブは、前記内側密閉部品および前記外側密閉部品が一緒に連結されたとき、前記内側密閉部品を通る、
請求項15に記載のポート構造体。 - 前記内側密閉部品は、取り付けられたガイドワイヤチューブを有する前記外側密閉部品が前記内側密閉部品を介して前記ステントグラフト送達システムのハンドル部品に着脱可能に連結されるよう、前記ハンドル部品に固定的に取り付けられる、
請求項16に記載のポート構造体。 - 前記内側密閉部品は、前記外側密閉部品が前記内側密閉部品を介して前記ステントグラフト送達システムのハンドル部品に着脱可能に連結されるよう、前記ハンドル部品に連結される、
請求項11に記載のポート構造体。 - 前記外側密閉部品の前記外部アクセスポートに係合し且つ前記外部アクセスポートを拡張させるための着脱可能なアクセスポートダイレータであって、前記外部アクセスポートを通して挿入されるためにその中をガイドワイヤが誘導される管腔を画成する、アクセスポートダイレータをさらに備えている、
請求項11に記載のポート構造体。 - 前記アクセスポートダイレータは、前記外部アクセスポートを形成する成形済み穴を拡張させるために前記外部アクセスポート内で摺動する小径雄部分を含む、
請求項19に記載のポート構造体。
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US13/457,544 US10098767B2 (en) | 2012-04-27 | 2012-04-27 | Reconfigurable stent-graft delivery system and method of use |
| US13/457,544 | 2012-04-27 | ||
| PCT/US2013/026739 WO2013162689A1 (en) | 2012-04-27 | 2013-02-19 | Reconfigurable stent-graft delivery system and method of use |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2015514548A JP2015514548A (ja) | 2015-05-21 |
| JP6282265B2 true JP6282265B2 (ja) | 2018-02-21 |
Family
ID=48087677
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2015508949A Active JP6282265B2 (ja) | 2012-04-27 | 2013-02-19 | 再構成可能なステントグラフト送達システムおよび使用方法 |
Country Status (5)
| Country | Link |
|---|---|
| US (1) | US10098767B2 (ja) |
| EP (1) | EP2841026B1 (ja) |
| JP (1) | JP6282265B2 (ja) |
| CN (1) | CN104254301B (ja) |
| WO (1) | WO2013162689A1 (ja) |
Families Citing this family (18)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20070198078A1 (en) | 2003-09-03 | 2007-08-23 | Bolton Medical, Inc. | Delivery system and method for self-centering a Proximal end of a stent graft |
| US11596537B2 (en) | 2003-09-03 | 2023-03-07 | Bolton Medical, Inc. | Delivery system and method for self-centering a proximal end of a stent graft |
| US8292943B2 (en) | 2003-09-03 | 2012-10-23 | Bolton Medical, Inc. | Stent graft with longitudinal support member |
| US11259945B2 (en) | 2003-09-03 | 2022-03-01 | Bolton Medical, Inc. | Dual capture device for stent graft delivery system and method for capturing a stent graft |
| WO2010005524A2 (en) | 2008-06-30 | 2010-01-14 | Bolton Medical, Inc. | Abdominal aortic aneurysms: systems and methods of use |
| EP3284447B1 (en) | 2009-03-13 | 2020-05-20 | Bolton Medical Inc. | System for deploying an endoluminal prosthesis at a surgical site |
| CN104363862B (zh) | 2012-04-12 | 2016-10-05 | 波顿医疗公司 | 血管假体输送装置及使用方法 |
| CN105209105B (zh) * | 2013-03-15 | 2019-01-04 | 波顿医疗公司 | 止血阀和输送系统 |
| US9439751B2 (en) | 2013-03-15 | 2016-09-13 | Bolton Medical, Inc. | Hemostasis valve and delivery systems |
| US9827126B2 (en) | 2013-08-27 | 2017-11-28 | Covidien Lp | Delivery of medical devices |
| EP3457948B1 (en) * | 2016-05-20 | 2020-07-01 | Vivasure Medical Limited | Vascular closure device |
| JP7194446B2 (ja) * | 2017-11-24 | 2022-12-22 | 有限会社Ptmc研究所 | 人工血管搬送装置 |
| WO2020030073A1 (zh) * | 2018-08-09 | 2020-02-13 | 杭州唯强医疗科技有限公司 | 控制支架分步释放的输送装置及输送系统 |
| CN109199659B (zh) * | 2018-09-12 | 2020-04-21 | 湖南埃普特医疗器械有限公司 | 一种支架输送系统 |
| WO2021176673A1 (ja) * | 2020-03-05 | 2021-09-10 | 日本ライフライン株式会社 | デリバリシャフトおよびデリバリシステム |
| CN111407463B (zh) * | 2020-03-19 | 2022-08-12 | 湖南埃普特医疗器械有限公司 | 一种覆膜支架系统 |
| US11357652B2 (en) * | 2020-03-24 | 2022-06-14 | Medtronic Vascular, Inc. | Branched stent graft delivery system |
| CN113877056B (zh) * | 2021-09-29 | 2023-03-31 | 复旦大学附属中山医院 | 一种桡动脉入路肝动脉灌注化疗体外导管收集器 |
Family Cites Families (52)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5195980A (en) * | 1992-01-03 | 1993-03-23 | Thomas Medical Products, Inc. | Hemostatic valve |
| US5425765A (en) | 1993-06-25 | 1995-06-20 | Tiefenbrun; Jonathan | Surgical bypass method |
| US5782904A (en) | 1993-09-30 | 1998-07-21 | Endogad Research Pty Limited | Intraluminal graft |
| US5899882A (en) * | 1994-10-27 | 1999-05-04 | Novoste Corporation | Catheter apparatus for radiation treatment of a desired area in the vascular system of a patient |
| WO1996036297A1 (fr) | 1995-05-19 | 1996-11-21 | Kanji Inoue | Instrument de transplantation, procede pour le courber et procede pour le transplanter |
| US6042605A (en) | 1995-12-14 | 2000-03-28 | Gore Enterprose Holdings, Inc. | Kink resistant stent-graft |
| EP0866677A4 (en) | 1995-12-14 | 1999-10-27 | Prograft Medical Inc | APPARATUS AND METHOD FOR DEPLOYING A GRAFT STENT |
| US5617878A (en) | 1996-05-31 | 1997-04-08 | Taheri; Syde A. | Stent and method for treatment of aortic occlusive disease |
| US6352561B1 (en) | 1996-12-23 | 2002-03-05 | W. L. Gore & Associates | Implant deployment apparatus |
| US6551350B1 (en) | 1996-12-23 | 2003-04-22 | Gore Enterprise Holdings, Inc. | Kink resistant bifurcated prosthesis |
| US5911710A (en) * | 1997-05-02 | 1999-06-15 | Schneider/Namic | Medical insertion device with hemostatic valve |
| AUPP083597A0 (en) | 1997-12-10 | 1998-01-08 | William A Cook Australia Pty Ltd | Endoluminal aortic stents |
| US6379365B1 (en) * | 1999-03-29 | 2002-04-30 | Alexis Diaz | Stent delivery catheter system having grooved shaft |
| JP3790474B2 (ja) * | 1999-09-07 | 2006-06-28 | メリット・メディカル・システムズ・インコーポレーテッド | 一体形導入子を有する止血弁装置 |
| US7131991B2 (en) | 2002-04-24 | 2006-11-07 | Medtronic Vascular, Inc. | Endoluminal prosthetic assembly and extension method |
| EP1596761B1 (en) * | 2003-02-14 | 2015-06-17 | Salviac Limited | Stent delivery and deployment system |
| US7537606B2 (en) | 2003-04-03 | 2009-05-26 | Cook Incorporated | Branch stent graft deployment and method |
| US7438721B2 (en) | 2003-04-25 | 2008-10-21 | Medtronic Vascular, Inc. | Universal modular stent graft assembly to accommodate flow to collateral branches |
| CA2540830C (en) | 2003-10-10 | 2012-08-14 | William A. Cook Australia Pty. Ltd. | Fenestrated stent grafts |
| US7347853B2 (en) * | 2004-05-12 | 2008-03-25 | C. R. Bard, Inc. | Catheter with removable extension |
| US7306623B2 (en) | 2005-01-13 | 2007-12-11 | Medtronic Vascular, Inc. | Branch vessel graft design and deployment method |
| US9320831B2 (en) | 2005-03-04 | 2016-04-26 | W. L. Gore & Associates, Inc. | Polymer shrink tubes and novel uses therefor |
| US7998134B2 (en) * | 2007-05-16 | 2011-08-16 | Icu Medical, Inc. | Medical connector |
| US8911491B2 (en) | 2005-09-02 | 2014-12-16 | Medtronic Vascular, Inc. | Methods and apparatus for treatment of aneurysms adjacent branch arteries including branch artery flow lumen alignment |
| US7955374B2 (en) | 2005-09-02 | 2011-06-07 | Medtronic Vascular, Inc. | Modular branch vessel stent-graft assembly |
| US20070083215A1 (en) | 2005-10-07 | 2007-04-12 | Hamer Rochelle M | Conduit for interventional procedures |
| CN2855365Y (zh) * | 2005-11-09 | 2007-01-10 | 王蓉珍 | 人工心脏支架瓣膜的输放装置 |
| US20070156224A1 (en) | 2006-01-04 | 2007-07-05 | Iulian Cioanta | Handle system for deploying a prosthetic implant |
| US20070208256A1 (en) | 2006-03-03 | 2007-09-06 | Medtronic Vascular, Inc. | Multiple Branch Tubular Prosthesis and Methods |
| US20070225797A1 (en) | 2006-03-24 | 2007-09-27 | Medtronic Vascular, Inc. | Prosthesis With Adjustable Opening for Side Branch Access |
| US9757260B2 (en) | 2006-03-30 | 2017-09-12 | Medtronic Vascular, Inc. | Prosthesis with guide lumen |
| US20070244547A1 (en) | 2006-04-18 | 2007-10-18 | Medtronic Vascular, Inc., A Delaware Corporation | Device and Method for Controlling the Positioning of a Stent Graft Fenestration |
| US7678141B2 (en) | 2006-04-18 | 2010-03-16 | Medtronic Vascular, Inc. | Stent graft having a flexible, articulable, and axially compressible branch graft |
| US8828074B2 (en) | 2006-04-21 | 2014-09-09 | Medtronic Vascular, Inc. | Stent graft having short tube graft for branch vessel |
| AU2007255001B2 (en) | 2006-06-02 | 2012-07-26 | Cook Incorporated | Multi-port delivery device |
| WO2008140796A1 (en) | 2007-05-11 | 2008-11-20 | William A. Cook Australia Pty. Ltd. | Stent grafts for the thoracic aorta |
| JP5290630B2 (ja) * | 2007-06-05 | 2013-09-18 | ニプロ株式会社 | 医療用コネクタおよびその製造方法 |
| GB2475494B (en) | 2009-11-18 | 2011-11-23 | Cook William Europ | Stent graft and introducer assembly |
| GB2476451A (en) | 2009-11-19 | 2011-06-29 | Cook William Europ | Stent Graft |
| US8163007B2 (en) | 2008-02-08 | 2012-04-24 | Cook Medical Technologies Llc | Stent designs for use with one or more trigger wires |
| WO2010005524A2 (en) * | 2008-06-30 | 2010-01-14 | Bolton Medical, Inc. | Abdominal aortic aneurysms: systems and methods of use |
| JP5134729B2 (ja) * | 2008-07-01 | 2013-01-30 | エンドロジックス、インク | カテーテルシステム |
| WO2010030370A1 (en) | 2008-09-12 | 2010-03-18 | William A. Cook Australia Pty. Ltd. | Radiopaque reinforcing member |
| US9301863B2 (en) * | 2009-03-10 | 2016-04-05 | Medtronic Vascular, Inc. | Prosthesis delivery apparatus and methods |
| EP2431009B1 (en) * | 2009-08-03 | 2016-09-14 | Terumo Kabushiki Kaisha | Stent delivery system |
| JP2011067558A (ja) * | 2009-09-28 | 2011-04-07 | Terumo Corp | イントロデューサシースおよびイントロデューサ組立体 |
| US9757263B2 (en) | 2009-11-18 | 2017-09-12 | Cook Medical Technologies Llc | Stent graft and introducer assembly |
| US9539413B2 (en) | 2010-05-11 | 2017-01-10 | Cathrx Ltd | Catheter handle |
| US8419783B2 (en) * | 2010-07-07 | 2013-04-16 | Cook Medical Technologies Llc | Graft deployment assist tool |
| AU2010210022B1 (en) | 2010-08-05 | 2011-09-08 | Cook Incorporated | Stent graft having a marker and a reinforcing and marker ring |
| CA2748206C (en) | 2010-08-21 | 2015-06-23 | Blayne A. Roeder | Prosthesis having pivoting fenestration |
| CN102247231B (zh) * | 2011-03-31 | 2014-06-04 | 上海微创医疗器械(集团)有限公司 | 改进的术中支架系统 |
-
2012
- 2012-04-27 US US13/457,544 patent/US10098767B2/en active Active
-
2013
- 2013-02-19 EP EP13715780.6A patent/EP2841026B1/en active Active
- 2013-02-19 JP JP2015508949A patent/JP6282265B2/ja active Active
- 2013-02-19 WO PCT/US2013/026739 patent/WO2013162689A1/en active Application Filing
- 2013-02-19 CN CN201380022018.5A patent/CN104254301B/zh active Active
Also Published As
| Publication number | Publication date |
|---|---|
| US20130289693A1 (en) | 2013-10-31 |
| CN104254301A (zh) | 2014-12-31 |
| WO2013162689A1 (en) | 2013-10-31 |
| CN104254301B (zh) | 2017-08-25 |
| US10098767B2 (en) | 2018-10-16 |
| EP2841026A1 (en) | 2015-03-04 |
| EP2841026B1 (en) | 2021-03-31 |
| JP2015514548A (ja) | 2015-05-21 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6282265B2 (ja) | 再構成可能なステントグラフト送達システムおよび使用方法 | |
| US9452069B2 (en) | Reconfigurable stent-graft delivery system and method of use | |
| JP6257588B2 (ja) | 再構成可能なステントグラフト送達システム | |
| US20210169670A1 (en) | Stent cannulation guiding device for bifurcated stent and method of use | |
| JP6282264B2 (ja) | 再構成可能ステントグラフト搬送システム | |
| JP5789867B2 (ja) | 分枝血管結合部のための内部密封カフを有する可動外部結合部 | |
| JP6825086B2 (ja) | ハイブリッド人工器官およびデリバリーシステム | |
| US9039752B2 (en) | Device and method for delivering a vascular device | |
| EP3138537B1 (en) | Prosthesis fixation system | |
| EP2944294B1 (en) | Stent cannulation guiding device |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20160218 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20161121 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20170104 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20170404 |
|
| RD04 | Notification of resignation of power of attorney |
Free format text: JAPANESE INTERMEDIATE CODE: A7424 Effective date: 20170414 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20170602 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20170704 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20171225 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20180123 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 6282265 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |