JP4032115B2 - Exhaust gas flow rate measurement method - Google Patents
Exhaust gas flow rate measurement method Download PDFInfo
- Publication number
- JP4032115B2 JP4032115B2 JP2002295921A JP2002295921A JP4032115B2 JP 4032115 B2 JP4032115 B2 JP 4032115B2 JP 2002295921 A JP2002295921 A JP 2002295921A JP 2002295921 A JP2002295921 A JP 2002295921A JP 4032115 B2 JP4032115 B2 JP 4032115B2
- Authority
- JP
- Japan
- Prior art keywords
- exhaust gas
- flow rate
- catalyst
- gas flow
- pressure
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Lifetime
Links
- 238000000691 measurement method Methods 0.000 title description 6
- 239000003054 catalyst Substances 0.000 claims description 57
- 238000000034 method Methods 0.000 claims description 20
- 238000002485 combustion reaction Methods 0.000 claims description 8
- 239000007789 gas Substances 0.000 description 86
- 239000000446 fuel Substances 0.000 description 7
- 238000005259 measurement Methods 0.000 description 7
- 238000012937 correction Methods 0.000 description 6
- 238000012360 testing method Methods 0.000 description 6
- 239000000126 substance Substances 0.000 description 5
- 239000000383 hazardous chemical Substances 0.000 description 3
- 229910052751 metal Inorganic materials 0.000 description 3
- 239000002184 metal Substances 0.000 description 3
- 239000000203 mixture Substances 0.000 description 3
- 238000000746 purification Methods 0.000 description 3
- UGFAIRIUMAVXCW-UHFFFAOYSA-N Carbon monoxide Chemical compound [O+]#[C-] UGFAIRIUMAVXCW-UHFFFAOYSA-N 0.000 description 2
- 229910002091 carbon monoxide Inorganic materials 0.000 description 2
- 230000003197 catalytic effect Effects 0.000 description 2
- 238000010790 dilution Methods 0.000 description 2
- 239000012895 dilution Substances 0.000 description 2
- 230000000694 effects Effects 0.000 description 2
- 229910052760 oxygen Inorganic materials 0.000 description 2
- 229910052596 spinel Inorganic materials 0.000 description 2
- 239000011029 spinel Substances 0.000 description 2
- 238000010998 test method Methods 0.000 description 2
- 239000010409 thin film Substances 0.000 description 2
- 241001573881 Corolla Species 0.000 description 1
- 238000003915 air pollution Methods 0.000 description 1
- 238000003556 assay Methods 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- 239000000919 ceramic Substances 0.000 description 1
- 230000006866 deterioration Effects 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 238000007865 diluting Methods 0.000 description 1
- 238000006073 displacement reaction Methods 0.000 description 1
- 230000007613 environmental effect Effects 0.000 description 1
- 238000003912 environmental pollution Methods 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 239000003546 flue gas Substances 0.000 description 1
- 239000012530 fluid Substances 0.000 description 1
- 229910001385 heavy metal Inorganic materials 0.000 description 1
- 229930195733 hydrocarbon Natural products 0.000 description 1
- 150000002430 hydrocarbons Chemical class 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 238000010248 power generation Methods 0.000 description 1
- 230000010349 pulsation Effects 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 239000004576 sand Substances 0.000 description 1
- 238000005245 sintering Methods 0.000 description 1
Images
Classifications
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02A—TECHNOLOGIES FOR ADAPTATION TO CLIMATE CHANGE
- Y02A50/00—TECHNOLOGIES FOR ADAPTATION TO CLIMATE CHANGE in human health protection, e.g. against extreme weather
- Y02A50/20—Air quality improvement or preservation, e.g. vehicle emission control or emission reduction by using catalytic converters
Landscapes
- Measuring Volume Flow (AREA)
- Exhaust Gas After Treatment (AREA)
Description
【0001】
【発明の属する技術分野】
本発明は、ハニカム構造の触媒系を有する燃焼排ガス流、特にセラミックス又は薄膜金属で構成されたモノリス触媒コンバータを用いる自動車の排ガス流量を簡単に測定する方法に関するものである。
【0002】
【従来の技術】
近年、環境汚染の元凶として、自動車エンジンからの排気ガスをはじめとするボイラー、発電用内燃機関などについて、燃料排ガスの排出規制が一段と厳しくなり、排ガスの浄化が重要な問題となってきている。
【0003】
ところで、自動車の排ガスに含まれる有害物質、例えばNOx、炭化水素、一酸化炭素などを評価するには、被試験車を自動車排出ガス試験法に規定された走行モードに従って走行させ、その際排出される有害物質の質量(g/km)をCVS装置で測定することによって行われる(非特許文献1参照)。
【0004】
このCVS法は、試験中の自動車排ガスを大量の空気で希釈して、自動車の排ガス量と希釈空気量の和を一定に保ちながら大気中に排出し、この際の試験時間に大気中に排出される希釈排ガス全量(Q)をCVS装置の流量定数と試験時間の積として求めるとともに、この希釈排ガスの一部を試験期間中、連続的にプラスチックバッグに捕集し、試験終了後、バッグ内の有害物質ガス濃度(C)を測定し、前記のQと、このCの積に有害物質ガス中の各有害成分の密度を乗じ、試験中に走行した距離で除すことによって試験中に排出された有害物質の排出質量を求めるという原理に基づく方法である。
【0005】
そして、このCVS法は、1972年に米国で用いられるようになって以来、現在では全世界の自動車排出ガス試験法として採用されているが、度重なる自動車排出ガスの規制強化により、自動車の有害ガス濃度基準が大幅に低下した結果、最近では排ガス希釈用空気と希釈排ガスとの間の有害物質濃度の差が接近し、十分な測定精度が維持できなくなりつつある。
【0006】
また、CVS装置の最大の利点は、自動車の排出ガス量を測定することなく、有害物質の排出質量が求められることにあるが、大量の空気で排出ガスを希釈しなければならないため、大規模な希釈トンネル装置と高感度排ガス分析計が必要となり、全体として高価なものとなる。
【0007】
このほか、CVS法のように自動車排ガスを希釈することをせずに、試験車の瞬時排ガス量と瞬時ガス濃度の積から有害物質の質量(g/km)を求める直接測定法も知られているが(非特許文献2参照)、自動車排ガス規制が開始された初期において利用できる分析装置は、体積、質量、消費電力が大きく、車載が困難であり、実用に供することはできなかった。
また、排ガス流量をエンジンに吸入する空気量から推測することも試みられたが、大規模で高価な装置を必要とする上に、動力性能を変えずに車載することがむずかしいため、実現しなかった。
【0008】
最近に至り、排出質量測定精度を向上させることに対する要求や、様々な環境温度における実路走行状態での有害ガス瞬時排出量のデータに対する要求が高まった結果、かつての直接測定法が見なおされ、分析計を車載し、オンボードで排出量を測定することが試みられるようになったが(非特許文献3)、いずれも排出ガスの流量を直接測定することができず、現在に至るも有害ガスの瞬時排出量を求める方法は知られていない。
【0009】
しかしながら、瞬時排ガス量を吸入空気量に基づいて推計することは可能であるため、この原理を利用した多数の測定方法、例えば熱線式、ピトー管式、ラミナーフロー式、カルマン渦式などの方法が試みられているが(非特許文献4参照)、装置が大きすぎたり、高価な測定機器を必要とすることがネックとなり、車載用としては実用化されていない。
【0010】
他方、自動車エンジンからの排気ガスの浄化装置としては、各種メタロシリケート粉末をハニカム状に成形し、焼結して得た担体に重金属を担持させたモノリス触媒(特許文献1参照)、特定の構造をもつ排ガス管に接続される触媒容器とその容器内に収容されたモノリス触媒コンバータ(特許文献2参照)、スピネルで担体を焼成被覆したスピネルモノリス触媒(特許文献3参照)などモノリス触媒を用いたものが主流となっている。
また、最近では、昇温特性を向上させるため金属の薄膜を担体としたメタル触媒も用いられるようになってきている(非特許文献1及び非特許文献2参照)。
【0011】
このモノリス触媒は、一般にハニカム構造を有しており、この中を通過するガス流量を差圧から簡単に測定し得ると思われるが、現在に至るまでこの方式はまだ実現していない。
【0012】
【特許文献1】
特開平8−38905号公報(請求項1及び2)
【特許文献2】
特開2000−265830号公報(請求項1)
【特許文献3】
特開2001−149782号公報(請求項1及び3)
【非特許文献1】
「新型自動車審査関係基準集」、5次改訂版、交文社、平成10年3月20日、p376〜538
【非特許文献2】
「ジャーナル・オブ・エア・ポリューション・コントロール・アッセイ(J.Air Poll.Control Assy.)」、(米国)、1960年、第10巻、p60〜68
【非特許文献3】
矢長、「実走排出ガス測定用車載分析装置の開発」、自動車技術会講演集 20005569、2000年10月17日、ISSN 0919〜1364
【非特許文献4】
八田、外2名、「内燃機関計測ハンドブック」、初版第1刷、朝倉書店、1979年5月20日、p249〜254
【0013】
【発明が解決しようとする課題】
本発明は、排気系に何の影響も与えず、自動車排気ガス量を正確かつ迅速に測定することができる、CVS法に代わるべき新規な排ガス流量測定方法を提供することを目的としてなされたものである。
【0014】
【課題を解決するための手段】
本発明者らは、CVS法に代わるべき排ガス流量測定方法を開発するために鋭意研究を重ねた結果、排ガスがハニカム構造の触媒系を通過する前後の圧力差を利用すれば、瞬時に流量測定を行うことができ、経時的に正確な排ガス量を知ることができることを見出し、この知見に基づいて本発明をなすに至った。
【0015】
すなわち、本発明は、管径が直径1mm以下の複数本の流路が25.4mm平方あたり400本ないし600本の密度で集まって構成されたフロースルー型のハニカム構造の触媒に、内燃機関から排出される排ガスを導いて、該フロースルー型の触媒を通過する前後での排ガスの圧力を測定し、その圧力差に所定の比例係数を乗算することによって、排ガス流量を求める排ガス流量測定方法を提供するものである。
【0016】
【発明の実施の形態】
一般に自動車の排気系を流れるガスは脈動を含む乱流であり、その正確な流量を求めることは非常に困難であるが、これが層流であれば、流量は圧力損失に比例するから管路の差圧から流量を求めることができる。そして、自動車に装着された触媒は、ハニカム構造をもつモノリス触媒系がほとんどであるが、このものはきわめて細い流路、すなわちセルに分割されているので、このセルを毛細管とみれば、触媒系内を流れる排気ガスのレイノルズ数は十分に小さくなり、層流を形成しているとみなすことができる。
【0017】
したがって、このような触媒系においては、その前後の圧力を測定し、その差から排ガス流量を以下のようにして求めることができる。
すなわち、所定の管内を流れる流体が層流の場合、次のアーゲン・ポアジュイ(Hagen Poiseulle)の式から、流量Qは圧力低下Δpに比例することが分る。
Q=πd4n/128L ・1/μ ・ Δp (1)
ただし、d:ハニカム構造を構成する各毛細管の直径(mm)
L:同上の毛細管の長さ(mm)
n:同上の毛細管の数(本)
μ:排ガスの粘度係数
【0018】
普通の乗用車の場合、モノリス触媒を構成するセル数は25.4mm当り400〜600本であるから、毛細管の直径は1mm以下であり、触媒長すなわち毛細管の長さは通常200〜300mmの範囲である。また触媒系の断面積は6000mm2程度であるから毛細管の数nは1000〜2000本程度になる。そして、これらの数値のうちd、L、n、μはそれぞれの系について定数であるから、触媒の前後における圧力差Δpが分れば、流量Qを求めることができる。
【0019】
次に添付図面に従って、本発明を詳細に説明する。
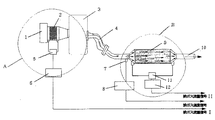
図1は、本発明方法の1例の流れ図であって、燃焼用空気は空気取入口1より吸入空気流量計2を通ってエンジン3に供給され、燃焼される。燃焼により発生する排気ガスは、排気マニホールド4を通って触媒系すなわち触媒キャニスター9に送られ、浄化後、排気口10から排出される。この際の吸入空気量を吸入空気流量計2で測定し、その圧力を圧力センサー5により、また触媒系の通過前後における排ガスの圧力差を第二圧力センサー11により測定し、それぞれ第一アンプ6及び第二アンプ12により増幅して、排ガス流量信号I及び排ガス流量信号IIとして出力する。一方において、排ガスの温度を排ガス温度センサー7で検知し、温度変換器8で変換して排ガス温度信号として出力する。
この図において、点線で囲まれた部分Bが、本発明方法を実施する部分である。
【0020】
この図1に示した流れ図に従って、シャシーダイナモに搭載した実験車の定常走行における触媒差圧と吸入空気量、排ガス温度を測定し、実際の触媒差圧と排ガス流量の関係を調べた結果をグラフとして図2に示す。この際の実験車両としては排気量1500ccの三元触媒系を搭載した乗用車(トヨタカローラNCV)を用い、層流型吸入空気流量計(司測砂LFE758)を吸気側へ取り付け、シャシーダイナモ上で、20、40、60、80及び100km/hの定常走行における吸入空気流量(リットル/sec)と触媒系の前後差圧とを比較した。この空気としては、温度20℃、圧力102kPaのものを用いた。
【0021】
実験の結果、吸入空気量に対する触媒差圧は次式のべき乗の関数の線に一致した。
y=6.95x 1.43 (2)
また、r2の回帰式では0.997という良好な相関が得られた。
以上の事実から実際の排気ガスについても、触媒差圧から吸入空気量を求めることができることが分る。
【0022】
ところで、実際に触媒通過前後の差圧に基づいて排ガスの体積流量を求める場合、触媒通過前後の差圧と排ガスの体積流量の関係は、ガスの組成温度、粘度、供給圧力によって変化するため、若干の補正が必要になる。
【0023】
しかしながら、自動車の空燃比は始動時を除き、常に一定に制御されているため、排ガスの組成は一定になっているし、走行中の排気管内のガスの圧力は全圧に対して多くても2%程度正に変動するだけであるから、流量に及ぼされる排ガス圧力の影響は少ない。そして、正確な測定を行う場合には、排気ガス圧の測定は常に行われているので、その測定値に基づいて容易に補正することができる。
【0024】
他方、排ガスはエンジン内の燃焼により生じるため、その体積は供給ガスの体積に比べ大幅に増加する。したがって触媒系を通過するガス流速が変わるので、標準状態の排ガス流量を求める場合には、温度について体積補正を行う必要がある。
【0025】
通常、自動車については排ガス浄化のために空燃比を正確に制御され、その数値は燃料質量に対して14.6倍の空気が消費されている。この値から吸入空気量に対する排出ガス量を算出すると、体積基準で排出ガス量は吸入空気量の1.065倍になり、これを体積補正係数として用いることができる。
【0026】
自動車の空燃比(A/F)はいずれの運転領域においても14.6であるので、組成は変らない。したがって、上記の体積補正係数はいずれの場合においても同様に用いることができる。
【0027】
次に、ガス温度は粘度係数にも影響を与え、例えば空気について温度が20℃の場合、1.81×104Paであるのが、600℃になると3.85×104Paとなり、粘度係数μは約2倍になる。この数値は、N2、H2O、CO2などのガスについてもほぼ同じであり、温度上昇に対する粘度の増加の傾向も同じである。
【0028】
したがって、ガス温度が20℃から触媒常用温度の600℃に上昇すると前記式(1)における触媒差圧Δpは1/2となるが、20℃から600℃に増加すると、シャルルの法則によりガス体積は約2倍に増加し、単位時間当りの質量流量は1/2となるので、結果的に両者は相殺され、図2のグラフに示されるように、触媒差圧と排出ガス流量とは直線関係を示すようになる。
このため、排ガス流量を吸入空気流量に基づいて校正する場合、触媒差圧Δpから真の排ガス流量を求める場合の補正係数は、ガス体積変化係数1.065のみとなる。
【0029】
図3は、25℃、102kPaの排ガスについて、ガス温度に対する補正を加えた場合の触媒差圧に対する排ガス流量の関係を示すグラフであるが、これから分るように差圧と排ガス流速との関係はほぼ完全な直線になっている。
【0030】
【発明の効果】
本発明は、以下に示す効果を奏する。
(1)既存の触媒を利用するため吸排気系に一切負荷を与えない。
(2)既存のモノリス触媒を搭載した自動車であれば、差圧を取り出すだけで容易に排ガス量の信号が得られる。
(3)触媒を搭載していない自動車でも、排気管に触媒又は触媒をコーティングしていないモノリスサポートを付加することにより排ガス流量が測定できる。
(4)自動車以外の高温ガス流路における排ガス量測定一般にも適用できる。(5)触媒の圧損増加から、触媒の事故、劣化が検知できる。
(6)排ガスのバイパスを設け、バイパス流量を触媒差圧とガス温度から最適流量になるよう制御することにより出力の向上が図れる。
(7)触媒差圧から得られる瞬時排ガス流量がエンジン吸入空気量と等しいとすれば、触媒差圧に比例した燃料を供給する新たな三元触媒空燃比制御システムが構築でき、従来の空燃比制御用エアーフローメータ、酸素センサーが不要となるが、このようにすると始動直後から空燃比制御が可能であり、排気対策が有利になる。
(8)直接触媒差圧でEGRバルブを駆動すれば、高精度でEGR量を負荷に比例して変えることができる。
【図面の簡単な説明】
【図1】 本発明方法の1例を示す流れ図。
【図2】 吸入空気流量と触媒差圧の関係を示すグラフ。
【図3】 ガス温度について、補正した場合の触媒差圧と排ガス流量との関係を示すグラフ。
【符号の説明】
1 空気取入口
2 吸入空気流量計
3 エンジン
4 排気マニホールド
5,11 圧力センサー
6,12 アンプ
7 温度センサー
8 温度変換器
9 触媒キャニスター
10 排気口[0001]
BACKGROUND OF THE INVENTION
The present invention relates to a method for simply measuring the exhaust gas flow of an automobile using a flue gas stream having a honeycomb structured catalyst system, in particular a monolith catalytic converter composed of ceramics or thin film metal.
[0002]
[Prior art]
In recent years, as a cause of environmental pollution, exhaust gas exhaust regulations from automobile engines and other internal combustion engines for power generation have become more stringent, and purification of exhaust gas has become an important issue.
[0003]
By the way, in order to evaluate harmful substances contained in the exhaust gas of automobiles, such as NOx, hydrocarbons, carbon monoxide, etc., the vehicle under test is driven in accordance with the driving mode defined in the automobile exhaust gas test method, and is emitted at that time. This is performed by measuring the mass (g / km) of harmful substances with a CVS device (see Non-Patent Document 1).
[0004]
In this CVS method, automobile exhaust gas under test is diluted with a large amount of air and discharged into the atmosphere while keeping the sum of the exhaust gas volume of the automobile and the diluted air volume constant. The total amount of diluted exhaust gas (Q) to be obtained is calculated as the product of the flow rate constant of the CVS device and the test time, and a part of this diluted exhaust gas is continuously collected in the plastic bag during the test period. Measure the hazardous substance gas concentration (C) of the product, multiply the product of Q and this C by the density of each harmful component in the hazardous substance gas, and divide by the distance traveled during the test. This is a method based on the principle of determining the emission mass of a hazardous substance.
[0005]
This CVS method has been adopted in the United States in 1972 and is now adopted as a vehicle emission test method worldwide. However, due to repeated regulations on vehicle emission, As a result of the significant reduction in gas concentration standards, recently, the difference in the concentration of harmful substances between the exhaust gas dilution air and the diluted exhaust gas has approached, and sufficient measurement accuracy cannot be maintained.
[0006]
The greatest advantage of the CVS device is that the emission mass of harmful substances is required without measuring the exhaust gas volume of automobiles. However, since the exhaust gas must be diluted with a large amount of air, it has a large scale. Neat dilution tunnel device and high-sensitivity exhaust gas analyzer are required, which is expensive as a whole.
[0007]
In addition, a direct measurement method for determining the mass (g / km) of harmful substances from the product of the instantaneous exhaust gas amount and the instantaneous gas concentration of the test vehicle without diluting the automobile exhaust gas as in the CVS method is also known. However (see Non-Patent Document 2), an analyzer that can be used in the early days when automobile exhaust gas regulations are started has a large volume, mass, and power consumption, is difficult to be mounted on a vehicle, and cannot be put to practical use.
Attempts have also been made to estimate the exhaust gas flow rate from the amount of air taken into the engine, but this is not possible because it requires a large and expensive device and it is difficult to mount the vehicle without changing the power performance. It was.
[0008]
As a result of recent demands for improving the accuracy of exhaust mass measurement and for data on instantaneous emission of harmful gases in actual road conditions at various environmental temperatures, the former direct measurement method has been considered. However, it has been tried to mount the analyzer on-board and measure the emission amount on-board (Non-patent Document 3), but none of them can directly measure the flow rate of the exhaust gas. There is no known method for determining the instantaneous emission of harmful gases.
[0009]
However, since it is possible to estimate the amount of instantaneous exhaust gas based on the amount of intake air, there are many measurement methods using this principle, such as a hot wire method, a Pitot tube method, a laminar flow method, and a Karman vortex method. Although an attempt has been made (see Non-Patent Document 4), the fact that the device is too large or requires an expensive measuring device has become a bottleneck, and it has not been put into practical use for in-vehicle use.
[0010]
On the other hand, as a device for purifying exhaust gas from an automobile engine, a monolith catalyst (refer to Patent Document 1) in which heavy metal is supported on a carrier obtained by forming various metallosilicate powders into a honeycomb shape and sintering them, and a specific structure. A monolithic catalyst such as a catalyst container connected to an exhaust pipe having a monolithic catalyst, a monolith catalytic converter housed in the container (see Patent Document 2), and a spinel monolith catalyst (see Patent Document 3) in which a carrier is calcined and coated with spinel were used. Things have become mainstream.
Recently, a metal catalyst using a metal thin film as a carrier has been used to improve temperature rise characteristics (see Non-Patent
[0011]
This monolithic catalyst generally has a honeycomb structure, and it seems that the gas flow rate passing through the monolithic catalyst can be easily measured from the differential pressure, but this method has not been realized until now.
[0012]
[Patent Document 1]
JP-A-8-38905 (
[Patent Document 2]
JP 2000-265830 A (Claim 1)
[Patent Document 3]
JP 2001-149784 A (
[Non-Patent Document 1]
“New Automobile Examination Standards Collection”, 5th revised edition, Kobunsha, March 20, 1998, p376-538
[Non-Patent Document 2]
“Journal of Air Pollution Control Assay (J. Air Poll. Control Assy.)” (USA), 1960, Vol. 10, p60-68.
[Non-Patent Document 3]
Yahagi, “Development of in-vehicle analyzer for actual exhaust gas measurement”, Proceedings of the Society of Automotive Engineers of Japan 2013569, October 17, 2000, ISSN 0919-1364
[Non-Patent Document 4]
Hatta, 2 others, “Internal combustion engine measurement handbook”, first edition, first edition, Asakura Shoten, May 20, 1979, p.
[0013]
[Problems to be solved by the invention]
The present invention has been made for the purpose of providing a novel exhaust gas flow rate measurement method that should replace the CVS method and that can accurately and quickly measure the amount of automobile exhaust gas without affecting the exhaust system. It is.
[0014]
[Means for Solving the Problems]
As a result of intensive research to develop an exhaust gas flow rate measurement method that should replace the CVS method, the present inventors measured the flow rate instantaneously if the pressure difference before and after the exhaust gas passed through the honeycomb catalyst system was used. It was found that the exhaust gas amount can be known over time, and the present invention has been made based on this finding.
[0015]
That is, the present invention relates to a flow-through type honeycomb structured catalyst in which a plurality of flow paths having a tube diameter of 1 mm or less are gathered at a density of 400 to 600 per 25.4 mm square from an internal combustion engine. leading the exhaust gas discharged by the pressure before later of the exhaust gas passing through the flow-through of the catalyst is measured and multiplied by a predetermined proportionality factor to the pressure difference, exhaust gas flow rate measuring method for determining the exhaust gas flow rate Is to provide.
[0016]
DETAILED DESCRIPTION OF THE INVENTION
In general, the gas flowing through the exhaust system of an automobile is a turbulent flow including pulsation, and it is very difficult to determine its exact flow rate. However, if this is a laminar flow, the flow rate is proportional to the pressure loss, so The flow rate can be obtained from the differential pressure. And most of the catalysts installed in automobiles are monolithic catalyst systems having a honeycomb structure, but these are divided into very narrow flow paths, that is, cells. It can be considered that the Reynolds number of the exhaust gas flowing inside is sufficiently small and a laminar flow is formed.
[0017]
Therefore, in such a catalyst system, the pressure before and after that is measured, and the exhaust gas flow rate can be obtained from the difference as follows.
That is, when the fluid flowing in the predetermined pipe is a laminar flow, the flow rate Q is proportional to the pressure drop Δp from the following Hagen Poisule equation.
Q = πd 4 n / 128L · 1 / μ · Δp (1)
Where d: diameter of each capillary constituting the honeycomb structure (mm)
L: Length of capillary tube as above (mm)
n: Number of capillaries as above
μ: Viscosity coefficient of exhaust gas [0018]
In the case of an ordinary passenger car, the number of cells constituting the monolith catalyst is 400 to 600 per 25.4 mm, so that the capillary diameter is 1 mm or less, and the catalyst length, that is, the capillary length is usually in the range of 200 to 300 mm. is there. Further, since the cross-sectional area of the catalyst system is about 6000 mm 2 , the number n of capillaries is about 1000 to 2000. Of these numerical values, d, L, n, and μ are constants for the respective systems. Therefore, if the pressure difference Δp before and after the catalyst is known, the flow rate Q can be obtained.
[0019]
Next, the present invention will be described in detail with reference to the accompanying drawings.
FIG. 1 is a flowchart of an example of the method of the present invention. Combustion air is supplied from an
In this figure, a portion B surrounded by a dotted line is a portion for carrying out the method of the present invention.
[0020]
In accordance with the flow chart shown in FIG. 1, the graph shows the results of measuring the catalyst differential pressure, the intake air amount, and the exhaust gas temperature during steady running of the experimental vehicle mounted on the chassis dynamo, and examining the relationship between the actual catalyst differential pressure and the exhaust gas flow rate. As shown in FIG. In this case, a passenger car (Toyota Corolla NCV) equipped with a three-way catalyst system with a displacement of 1500 cc was used as an experimental vehicle, and a laminar flow type intake air flow meter (Judgment Sand LFE758) was attached to the intake side. , 20, 40, 60, 80 and 100 km / h, the intake air flow rate (liter / sec) and the differential pressure across the catalyst system were compared. As the air, air having a temperature of 20 ° C. and a pressure of 10 2 kPa was used.
[0021]
As a result of the experiment, the catalyst differential pressure with respect to the intake air amount coincided with a power function line of the following equation.
y = 6.95x 1.43 (2)
Also, it was obtained a good correlation of 0.997 in regression r 2.
From the above facts, it is understood that the intake air amount can be obtained from the catalyst differential pressure also for the actual exhaust gas.
[0022]
By the way, when actually obtaining the volumetric flow rate of exhaust gas based on the differential pressure before and after passing through the catalyst, the relationship between the differential pressure before and after passing through the catalyst and the volumetric flow rate of exhaust gas changes depending on the gas composition temperature, viscosity, and supply pressure. Some correction is required.
[0023]
However, since the air-fuel ratio of the automobile is constantly controlled except at the time of starting, the composition of the exhaust gas is constant, and the pressure of the gas in the exhaust pipe during traveling is greater than the total pressure. Since it only fluctuates about 2% positively, the influence of the exhaust gas pressure on the flow rate is small. And when performing an accurate measurement, since the measurement of exhaust gas pressure is always performed, it can correct | amend easily based on the measured value.
[0024]
On the other hand, since the exhaust gas is generated by combustion in the engine, its volume increases significantly compared to the volume of the supply gas. Therefore, since the gas flow rate passing through the catalyst system changes, it is necessary to perform volume correction on the temperature when obtaining the exhaust gas flow rate in the standard state.
[0025]
In general, the air-fuel ratio of an automobile is accurately controlled for exhaust gas purification, and the numerical value consumes 14.6 times as much air as the fuel mass. If the exhaust gas amount relative to the intake air amount is calculated from this value, the exhaust gas amount is 1.065 times the intake air amount on a volume basis, and this can be used as a volume correction coefficient.
[0026]
Since the air-fuel ratio (A / F) of the automobile is 14.6 in any operating region, the composition does not change. Therefore, the above volume correction coefficient can be used in any case.
[0027]
Next, the gas temperature also affects the viscosity coefficient. For example, when the temperature of air is 20 ° C., it is 1.81 × 10 4 Pa, but when it reaches 600 ° C., it becomes 3.85 × 10 4 Pa, and the viscosity The coefficient μ is approximately doubled. This numerical value is substantially the same for gases such as N 2 , H 2 O, and CO 2 , and the tendency of increase in viscosity with temperature rise is the same.
[0028]
Therefore, when the gas temperature is increased from 20 ° C. to the catalyst normal temperature of 600 ° C., the catalyst differential pressure Δp in the above equation (1) becomes ½, but when it is increased from 20 ° C. to 600 ° C., the gas volume is determined by Charles' law. Increases approximately twice, and the mass flow rate per unit time is halved. As a result, both cancel each other, and the catalyst differential pressure and the exhaust gas flow rate are linear as shown in the graph of FIG. Shows the relationship.
For this reason, when the exhaust gas flow rate is calibrated based on the intake air flow rate, the correction coefficient for obtaining the true exhaust gas flow rate from the catalyst differential pressure Δp is only the gas volume change coefficient 1.065.
[0029]
FIG. 3 is a graph showing the relationship of the exhaust gas flow rate with respect to the catalyst differential pressure when the correction is made to the gas temperature for the exhaust gas at 25 ° C. and 10 2 kPa. The relationship is almost a complete straight line.
[0030]
【The invention's effect】
The present invention has the following effects.
(1) No load is applied to the intake / exhaust system because the existing catalyst is used.
(2) If the vehicle is equipped with an existing monolith catalyst, an exhaust gas amount signal can be easily obtained simply by taking out the differential pressure.
(3) Even in an automobile not equipped with a catalyst, the exhaust gas flow rate can be measured by adding a catalyst or a monolith support not coated with a catalyst to the exhaust pipe.
(4) Applicable to general exhaust gas measurement in high-temperature gas flow paths other than automobiles. (5) Accidents and deterioration of the catalyst can be detected from an increase in catalyst pressure loss.
(6) The output can be improved by providing a bypass for the exhaust gas and controlling the bypass flow rate so as to obtain an optimum flow rate from the catalyst differential pressure and the gas temperature.
(7) If the instantaneous exhaust gas flow rate obtained from the catalyst differential pressure is equal to the engine intake air amount, a new three-way catalyst air-fuel ratio control system that supplies fuel proportional to the catalyst differential pressure can be constructed. A control air flow meter and an oxygen sensor are not required, but if this is done, air-fuel ratio control can be performed immediately after startup, which is advantageous as a countermeasure against exhaust.
(8) If the EGR valve is directly driven by the catalyst differential pressure, the amount of EGR can be changed in proportion to the load with high accuracy.
[Brief description of the drawings]
FIG. 1 is a flowchart showing an example of the method of the present invention.
FIG. 2 is a graph showing the relationship between the intake air flow rate and the catalyst differential pressure.
FIG. 3 is a graph showing the relationship between the catalyst differential pressure and the exhaust gas flow rate when the gas temperature is corrected.
[Explanation of symbols]
DESCRIPTION OF
Claims (3)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2002295921A JP4032115B2 (en) | 2002-10-09 | 2002-10-09 | Exhaust gas flow rate measurement method |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2002295921A JP4032115B2 (en) | 2002-10-09 | 2002-10-09 | Exhaust gas flow rate measurement method |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| JP2004132212A JP2004132212A (en) | 2004-04-30 |
| JP2004132212A5 JP2004132212A5 (en) | 2005-05-19 |
| JP4032115B2 true JP4032115B2 (en) | 2008-01-16 |
Family
ID=32286033
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2002295921A Expired - Lifetime JP4032115B2 (en) | 2002-10-09 | 2002-10-09 | Exhaust gas flow rate measurement method |
Country Status (1)
| Country | Link |
|---|---|
| JP (1) | JP4032115B2 (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2012127864A (en) * | 2010-12-16 | 2012-07-05 | Toyota Motor Corp | Flow rate measurement method of pulsatile flow and gas flow rate measurement device |
Families Citing this family (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2007041870A (en) * | 2005-08-03 | 2007-02-15 | Horiba Stec Co Ltd | Gas flow rate control method |
| JP4539595B2 (en) * | 2006-03-28 | 2010-09-08 | 三菱自動車工業株式会社 | PM deposition amount estimation device for internal combustion engine |
| JP5669402B2 (en) * | 2010-01-08 | 2015-02-12 | 三菱重工業株式会社 | Heat pump and heat medium flow rate calculation method for heat pump |
| DE102015211169A1 (en) * | 2015-06-17 | 2016-12-22 | Mtu Friedrichshafen Gmbh | A method of operating an exhaust aftertreatment system, exhaust aftertreatment system, and internal combustion engine having an exhaust aftertreatment system |
-
2002
- 2002-10-09 JP JP2002295921A patent/JP4032115B2/en not_active Expired - Lifetime
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2012127864A (en) * | 2010-12-16 | 2012-07-05 | Toyota Motor Corp | Flow rate measurement method of pulsatile flow and gas flow rate measurement device |
Also Published As
| Publication number | Publication date |
|---|---|
| JP2004132212A (en) | 2004-04-30 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP4294863B2 (en) | Modular flow meter and exhaust notification system for vehicle exhaust gas during real-time driving | |
| KR101707759B1 (en) | Method for diagnosing an exhaust gas catalytic converter, diagnosis device and motor vehicle having such a device | |
| US8626451B2 (en) | Method and device for characterization and sensing of exhaust gas and control of engines and components for aftertreatment of exhaust gases | |
| GB2324157A (en) | Testing the conversion capacity of a catalytic converter | |
| IT8948609A1 (en) | PROCEDURE FOR DETERMINING THE STATE OF ACTIVITY OF A CATALYST IN EXHAUST SYSTEMS OF INTERNAL COMBUSTION ENGINES. | |
| Kihara et al. | Real-time on-board measurement of mass emission of NOx, fuel consumption, road load, and engine output for diesel vehicles | |
| CN110637148B (en) | System and method for controlling flow distribution in an aftertreatment system | |
| JP5563602B2 (en) | Method for operating an exhaust system | |
| JP3645756B2 (en) | Nitrogen oxide simple measurement method for traveling vehicles | |
| CN111305937A (en) | Three-way catalyst oxygen storage model | |
| US7452724B2 (en) | Method for operating a metering unit of a catalytic converter | |
| JP4032115B2 (en) | Exhaust gas flow rate measurement method | |
| US11939898B2 (en) | Exhaust system of an internal combustion engine | |
| JP7265449B2 (en) | Method for diagnosing catalyst deterioration | |
| US7624571B2 (en) | Exhaust gas purifying apparatus for internal combustion engine and method for estimating collected amount of exhaust particles | |
| JP2006284470A (en) | Exhaust gas measuring device and exhaust gas measuring method | |
| CN107269368A (en) | Method and exhaust aftertreatment device for monitoring methane oxidation catalyst converter | |
| JPH09318572A (en) | Exhaust gas component measuring method and device | |
| JP4607541B2 (en) | Three-way catalyst deterioration diagnosis method | |
| CN105443216A (en) | Vehicle catalyst oxygen storage capacity testing system | |
| JP4077968B2 (en) | Nitrogen oxide simple measurement method for traveling vehicles | |
| JP4049300B2 (en) | Deterioration diagnosis method and apparatus for exhaust gas countermeasure device | |
| Staab et al. | Measurement of automobile exhaust emissions under realistic road conditions | |
| JP2013029395A (en) | Gas analysis apparatus | |
| KR100210664B1 (en) | Apparatus for sampling automotive exhaust gas |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20040707 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20040707 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20070130 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20070208 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20070402 |
|
| RD01 | Notification of change of attorney |
Free format text: JAPANESE INTERMEDIATE CODE: A7421 Effective date: 20070402 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A821 Effective date: 20070402 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20070613 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20070402 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20070925 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 4032115 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| S111 | Request for change of ownership or part of ownership |
Free format text: JAPANESE INTERMEDIATE CODE: R313111 |
|
| R350 | Written notification of registration of transfer |
Free format text: JAPANESE INTERMEDIATE CODE: R350 |
|
| S531 | Written request for registration of change of domicile |
Free format text: JAPANESE INTERMEDIATE CODE: R313532 |
|
| R350 | Written notification of registration of transfer |
Free format text: JAPANESE INTERMEDIATE CODE: R350 |
|
| EXPY | Cancellation because of completion of term |