JP2010512224A - 管腔のステント移植された領域における流れの阻害を最小化する装置および方法 - Google Patents
管腔のステント移植された領域における流れの阻害を最小化する装置および方法 Download PDFInfo
- Publication number
- JP2010512224A JP2010512224A JP2009541380A JP2009541380A JP2010512224A JP 2010512224 A JP2010512224 A JP 2010512224A JP 2009541380 A JP2009541380 A JP 2009541380A JP 2009541380 A JP2009541380 A JP 2009541380A JP 2010512224 A JP2010512224 A JP 2010512224A
- Authority
- JP
- Japan
- Prior art keywords
- stent
- flow
- scaffold
- flow surface
- longitudinal axis
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Withdrawn
Links
- 238000000034 method Methods 0.000 title claims description 18
- 239000003814 drug Substances 0.000 claims description 12
- 229940079593 drug Drugs 0.000 claims description 10
- 239000012530 fluid Substances 0.000 claims description 10
- 239000000463 material Substances 0.000 claims description 6
- 238000003698 laser cutting Methods 0.000 claims description 5
- 238000002595 magnetic resonance imaging Methods 0.000 claims description 2
- 239000010935 stainless steel Substances 0.000 claims description 2
- 229910001220 stainless steel Inorganic materials 0.000 claims description 2
- 229940124597 therapeutic agent Drugs 0.000 claims description 2
- 230000001154 acute effect Effects 0.000 claims 1
- 208000007536 Thrombosis Diseases 0.000 abstract description 13
- 238000013461 design Methods 0.000 abstract description 6
- 230000012010 growth Effects 0.000 abstract description 6
- 210000004204 blood vessel Anatomy 0.000 abstract description 5
- 208000031481 Pathologic Constriction Diseases 0.000 abstract description 4
- 230000015572 biosynthetic process Effects 0.000 abstract description 4
- 208000037804 stenosis Diseases 0.000 abstract description 4
- 230000036262 stenosis Effects 0.000 abstract description 4
- 238000012805 post-processing Methods 0.000 abstract 2
- 208000037803 restenosis Diseases 0.000 description 15
- 238000002513 implantation Methods 0.000 description 14
- 230000035755 proliferation Effects 0.000 description 11
- 239000003795 chemical substances by application Substances 0.000 description 6
- 230000007704 transition Effects 0.000 description 6
- 238000011161 development Methods 0.000 description 5
- 230000018109 developmental process Effects 0.000 description 5
- 238000009826 distribution Methods 0.000 description 5
- 230000002093 peripheral effect Effects 0.000 description 5
- 230000002792 vascular Effects 0.000 description 5
- 229910052751 metal Inorganic materials 0.000 description 4
- 239000002184 metal Substances 0.000 description 4
- 230000017531 blood circulation Effects 0.000 description 3
- 230000004663 cell proliferation Effects 0.000 description 3
- 230000008859 change Effects 0.000 description 3
- 230000006378 damage Effects 0.000 description 3
- 238000011112 process operation Methods 0.000 description 3
- 210000005166 vasculature Anatomy 0.000 description 3
- 206010028980 Neoplasm Diseases 0.000 description 2
- 239000003146 anticoagulant agent Substances 0.000 description 2
- 230000008901 benefit Effects 0.000 description 2
- 239000008280 blood Substances 0.000 description 2
- 210000004369 blood Anatomy 0.000 description 2
- 238000005520 cutting process Methods 0.000 description 2
- 230000002939 deleterious effect Effects 0.000 description 2
- 238000009760 electrical discharge machining Methods 0.000 description 2
- 238000005242 forging Methods 0.000 description 2
- 230000035876 healing Effects 0.000 description 2
- 230000000968 intestinal effect Effects 0.000 description 2
- 230000008569 process Effects 0.000 description 2
- 238000003466 welding Methods 0.000 description 2
- 206010051341 Bile duct stenosis Diseases 0.000 description 1
- 229910000684 Cobalt-chrome Inorganic materials 0.000 description 1
- 102000004127 Cytokines Human genes 0.000 description 1
- 108090000695 Cytokines Proteins 0.000 description 1
- 208000007217 Esophageal Stenosis Diseases 0.000 description 1
- 208000023281 Fallot tetralogy Diseases 0.000 description 1
- 206010061216 Infarction Diseases 0.000 description 1
- 206010022699 Intestinal stenosis Diseases 0.000 description 1
- 102000029749 Microtubule Human genes 0.000 description 1
- 108091022875 Microtubule Proteins 0.000 description 1
- 206010030194 Oesophageal stenosis Diseases 0.000 description 1
- 241000283973 Oryctolagus cuniculus Species 0.000 description 1
- 208000018262 Peripheral vascular disease Diseases 0.000 description 1
- 208000006396 Pulmonary artery stenosis Diseases 0.000 description 1
- 206010049171 Pulmonary vein stenosis Diseases 0.000 description 1
- 201000000660 Pyloric Stenosis Diseases 0.000 description 1
- 208000006011 Stroke Diseases 0.000 description 1
- 208000001122 Superior Vena Cava Syndrome Diseases 0.000 description 1
- 208000001106 Takayasu Arteritis Diseases 0.000 description 1
- 201000003005 Tetralogy of Fallot Diseases 0.000 description 1
- HZEWFHLRYVTOIW-UHFFFAOYSA-N [Ti].[Ni] Chemical compound [Ti].[Ni] HZEWFHLRYVTOIW-UHFFFAOYSA-N 0.000 description 1
- 230000002411 adverse Effects 0.000 description 1
- 230000004075 alteration Effects 0.000 description 1
- 238000002583 angiography Methods 0.000 description 1
- 230000000702 anti-platelet effect Effects 0.000 description 1
- 230000001028 anti-proliverative effect Effects 0.000 description 1
- 229940127219 anticoagulant drug Drugs 0.000 description 1
- 239000002246 antineoplastic agent Substances 0.000 description 1
- 206010002906 aortic stenosis Diseases 0.000 description 1
- 210000001367 artery Anatomy 0.000 description 1
- 230000004323 axial length Effects 0.000 description 1
- 210000000013 bile duct Anatomy 0.000 description 1
- 239000011230 binding agent Substances 0.000 description 1
- 239000000560 biocompatible material Substances 0.000 description 1
- 210000000748 cardiovascular system Anatomy 0.000 description 1
- 230000015556 catabolic process Effects 0.000 description 1
- 239000011248 coating agent Substances 0.000 description 1
- 238000000576 coating method Methods 0.000 description 1
- 239000010952 cobalt-chrome Substances 0.000 description 1
- 238000007887 coronary angioplasty Methods 0.000 description 1
- 208000029078 coronary artery disease Diseases 0.000 description 1
- 238000006731 degradation reaction Methods 0.000 description 1
- 230000001627 detrimental effect Effects 0.000 description 1
- 206010012601 diabetes mellitus Diseases 0.000 description 1
- 210000002249 digestive system Anatomy 0.000 description 1
- 210000000750 endocrine system Anatomy 0.000 description 1
- 210000002889 endothelial cell Anatomy 0.000 description 1
- 238000005530 etching Methods 0.000 description 1
- 239000003527 fibrinolytic agent Substances 0.000 description 1
- 230000006870 function Effects 0.000 description 1
- 210000001035 gastrointestinal tract Anatomy 0.000 description 1
- 229940125672 glycoprotein IIb/IIIa inhibitor Drugs 0.000 description 1
- 238000003384 imaging method Methods 0.000 description 1
- 229940125721 immunosuppressive agent Drugs 0.000 description 1
- 239000003018 immunosuppressive agent Substances 0.000 description 1
- 230000007574 infarction Effects 0.000 description 1
- 230000005764 inhibitory process Effects 0.000 description 1
- 230000003902 lesion Effects 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- 210000004688 microtubule Anatomy 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 210000004165 myocardium Anatomy 0.000 description 1
- 230000001338 necrotic effect Effects 0.000 description 1
- 229910001000 nickel titanium Inorganic materials 0.000 description 1
- 238000005457 optimization Methods 0.000 description 1
- 238000000206 photolithography Methods 0.000 description 1
- 229920000642 polymer Polymers 0.000 description 1
- 210000005227 renal system Anatomy 0.000 description 1
- 230000008439 repair process Effects 0.000 description 1
- 210000002345 respiratory system Anatomy 0.000 description 1
- 238000004904 shortening Methods 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- 239000013076 target substance Substances 0.000 description 1
- 238000002560 therapeutic procedure Methods 0.000 description 1
- 239000010409 thin film Substances 0.000 description 1
- 230000008467 tissue growth Effects 0.000 description 1
- 230000001052 transient effect Effects 0.000 description 1
- 230000006496 vascular abnormality Effects 0.000 description 1
- 208000019553 vascular disease Diseases 0.000 description 1
- 238000012800 visualization Methods 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/82—Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/86—Stents in a form characterised by the wire-like elements; Stents in the form characterised by a net-like or mesh-like structure
- A61F2/90—Stents in a form characterised by the wire-like elements; Stents in the form characterised by a net-like or mesh-like structure characterised by a net-like or mesh-like structure
- A61F2/91—Stents in a form characterised by the wire-like elements; Stents in the form characterised by a net-like or mesh-like structure characterised by a net-like or mesh-like structure made from perforated sheets or tubes, e.g. perforated by laser cuts or etched holes
- A61F2/915—Stents in a form characterised by the wire-like elements; Stents in the form characterised by a net-like or mesh-like structure characterised by a net-like or mesh-like structure made from perforated sheets or tubes, e.g. perforated by laser cuts or etched holes with bands having a meander structure, adjacent bands being connected to each other
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/82—Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/86—Stents in a form characterised by the wire-like elements; Stents in the form characterised by a net-like or mesh-like structure
- A61F2/90—Stents in a form characterised by the wire-like elements; Stents in the form characterised by a net-like or mesh-like structure characterised by a net-like or mesh-like structure
- A61F2/91—Stents in a form characterised by the wire-like elements; Stents in the form characterised by a net-like or mesh-like structure characterised by a net-like or mesh-like structure made from perforated sheets or tubes, e.g. perforated by laser cuts or etched holes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/02—Prostheses implantable into the body
- A61F2/30—Joints
- A61F2002/30001—Additional features of subject-matter classified in A61F2/28, A61F2/30 and subgroups thereof
- A61F2002/30108—Shapes
- A61F2002/3011—Cross-sections or two-dimensional shapes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2/00—Filters implantable into blood vessels; Prostheses, i.e. artificial substitutes or replacements for parts of the body; Appliances for connecting them with the body; Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/82—Devices providing patency to, or preventing collapsing of, tubular structures of the body, e.g. stents
- A61F2/86—Stents in a form characterised by the wire-like elements; Stents in the form characterised by a net-like or mesh-like structure
- A61F2/90—Stents in a form characterised by the wire-like elements; Stents in the form characterised by a net-like or mesh-like structure characterised by a net-like or mesh-like structure
- A61F2/91—Stents in a form characterised by the wire-like elements; Stents in the form characterised by a net-like or mesh-like structure characterised by a net-like or mesh-like structure made from perforated sheets or tubes, e.g. perforated by laser cuts or etched holes
- A61F2/915—Stents in a form characterised by the wire-like elements; Stents in the form characterised by a net-like or mesh-like structure characterised by a net-like or mesh-like structure made from perforated sheets or tubes, e.g. perforated by laser cuts or etched holes with bands having a meander structure, adjacent bands being connected to each other
- A61F2002/9155—Adjacent bands being connected to each other
- A61F2002/91558—Adjacent bands being connected to each other connected peak to peak
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2230/00—Geometry of prostheses classified in groups A61F2/00 - A61F2/26 or A61F2/82 or A61F9/00 or A61F11/00 or subgroups thereof
- A61F2230/0002—Two-dimensional shapes, e.g. cross-sections
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61F—FILTERS IMPLANTABLE INTO BLOOD VESSELS; PROSTHESES; DEVICES PROVIDING PATENCY TO, OR PREVENTING COLLAPSING OF, TUBULAR STRUCTURES OF THE BODY, e.g. STENTS; ORTHOPAEDIC, NURSING OR CONTRACEPTIVE DEVICES; FOMENTATION; TREATMENT OR PROTECTION OF EYES OR EARS; BANDAGES, DRESSINGS OR ABSORBENT PADS; FIRST-AID KITS
- A61F2230/00—Geometry of prostheses classified in groups A61F2/00 - A61F2/26 or A61F2/82 or A61F9/00 or A61F11/00 or subgroups thereof
- A61F2230/0002—Two-dimensional shapes, e.g. cross-sections
- A61F2230/0028—Shapes in the form of latin or greek characters
- A61F2230/0054—V-shaped
Landscapes
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Biomedical Technology (AREA)
- Heart & Thoracic Surgery (AREA)
- Life Sciences & Earth Sciences (AREA)
- Cardiology (AREA)
- Oral & Maxillofacial Surgery (AREA)
- Transplantation (AREA)
- Physics & Mathematics (AREA)
- Vascular Medicine (AREA)
- Optics & Photonics (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Prostheses (AREA)
- Media Introduction/Drainage Providing Device (AREA)
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US87442806P | 2006-12-12 | 2006-12-12 | |
| PCT/US2007/025517 WO2008073496A2 (en) | 2006-12-12 | 2007-12-12 | Apparatus and method for minimizing flow disturbances in a stented region of a lumen |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| JP2010512224A true JP2010512224A (ja) | 2010-04-22 |
Family
ID=39512343
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2009541380A Withdrawn JP2010512224A (ja) | 2006-12-12 | 2007-12-12 | 管腔のステント移植された領域における流れの阻害を最小化する装置および方法 |
Country Status (3)
| Country | Link |
|---|---|
| EP (1) | EP2094192A4 (de) |
| JP (1) | JP2010512224A (de) |
| WO (1) | WO2008073496A2 (de) |
Families Citing this family (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP6428050B2 (ja) * | 2014-02-25 | 2018-11-28 | ニプロ株式会社 | ステント |
| WO2015098629A1 (ja) * | 2013-12-24 | 2015-07-02 | 二プロ株式会社 | ステント |
| CN109998746B (zh) | 2013-12-24 | 2021-06-11 | 尼普洛株式会社 | 支架 |
| JP6380909B2 (ja) * | 2013-12-24 | 2018-08-29 | ニプロ株式会社 | ステント |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5902475A (en) * | 1997-04-08 | 1999-05-11 | Interventional Technologies, Inc. | Method for manufacturing a stent |
| US20030187498A1 (en) | 2002-03-28 | 2003-10-02 | Medtronic Ave, Inc. | Chamfered stent strut and method of making same |
| US6699275B1 (en) * | 2002-10-11 | 2004-03-02 | Enteromedics Inc. | Stent and delivery system |
| US7780721B2 (en) * | 2004-09-01 | 2010-08-24 | C. R. Bard, Inc. | Stent and method for manufacturing the stent |
| US7628807B2 (en) | 2004-11-04 | 2009-12-08 | Boston Scientific Scimed, Inc. | Stent for delivering a therapeutic agent having increased body tissue contact surface |
-
2007
- 2007-12-12 WO PCT/US2007/025517 patent/WO2008073496A2/en not_active Ceased
- 2007-12-12 EP EP07867742A patent/EP2094192A4/de not_active Withdrawn
- 2007-12-12 JP JP2009541380A patent/JP2010512224A/ja not_active Withdrawn
Also Published As
| Publication number | Publication date |
|---|---|
| WO2008073496A3 (en) | 2008-10-09 |
| EP2094192A2 (de) | 2009-09-02 |
| EP2094192A4 (de) | 2009-12-23 |
| WO2008073496A2 (en) | 2008-06-19 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US20230093376A1 (en) | Stent | |
| US11103371B2 (en) | Highly flexible stent and method of manufacture | |
| US8333798B2 (en) | Implantable medical devices with enhanced visibility, mechanical properties and biocompatability | |
| US8333799B2 (en) | Highly flexible stent and method of manufacture | |
| EP1189554B1 (de) | Stent mit variabler dicke | |
| US7540881B2 (en) | Bifurcation stent pattern | |
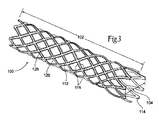
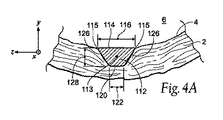
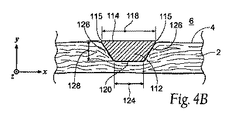
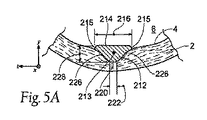
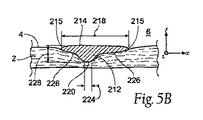
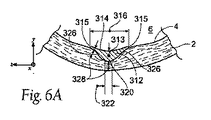
| US20080140179A1 (en) | Apparatus and method for minimizing flow disturbances in a stented region of a lumen | |
| US20110166641A1 (en) | Highly flexible stent and method of manufacture | |
| US9427302B2 (en) | Stent having a C-shaped body section for use in a bifurcation | |
| US20070213807A1 (en) | Non-foreshortening intraluminal prosthesis | |
| US20150039078A1 (en) | Highly flexible stent and method of manufacture | |
| US20130090721A1 (en) | Highly Flexible Stent and Method of Manufacture | |
| JP2009528886A (ja) | 均一な側枝突部を備えた分岐型ステント | |
| US8480728B2 (en) | Stent side branch deployment initiation geometry | |
| JP2009507561A (ja) | クラウンステントアセンブリ | |
| US20090259299A1 (en) | Side Branch Stent Having a Proximal Flexible Material Section | |
| US20070260304A1 (en) | Bifurcated stent with minimally circumferentially projected side branch | |
| JP2010512224A (ja) | 管腔のステント移植された領域における流れの阻害を最小化する装置および方法 | |
| WO2007103015A2 (en) | Stent with side aperture | |
| US9320628B2 (en) | Endoprosthesis devices including biostable and bioabsorable regions | |
| US20150374485A1 (en) | Targeted perforations in endovascular device |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A300 | Application deemed to be withdrawn because no request for examination was validly filed |
Free format text: JAPANESE INTERMEDIATE CODE: A300 Effective date: 20110301 |