EP4397752A2 - Verfahren zur gewinnung von induzierten glatten muskelzellen - Google Patents
Verfahren zur gewinnung von induzierten glatten muskelzellen Download PDFInfo
- Publication number
- EP4397752A2 EP4397752A2 EP24175168.4A EP24175168A EP4397752A2 EP 4397752 A2 EP4397752 A2 EP 4397752A2 EP 24175168 A EP24175168 A EP 24175168A EP 4397752 A2 EP4397752 A2 EP 4397752A2
- Authority
- EP
- European Patent Office
- Prior art keywords
- cells
- ismcs
- smooth muscle
- skeletal muscle
- mpcs
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Withdrawn
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
- C12N5/0602—Vertebrate cells
- C12N5/0652—Cells of skeletal and connective tissues; Mesenchyme
- C12N5/0661—Smooth muscle cells
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K35/00—Medicinal preparations containing materials or reaction products thereof with undetermined constitution
- A61K35/12—Materials from mammals; Compositions comprising non-specified tissues or cells; Compositions comprising non-embryonic stem cells; Genetically modified cells
- A61K35/34—Muscles; Smooth muscle cells; Heart; Cardiac stem cells; Myoblasts; Myocytes; Cardiomyocytes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P21/00—Drugs for disorders of the muscular or neuromuscular system
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/10—Growth factors
- C12N2501/15—Transforming growth factor beta (TGF-β)
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/90—Polysaccharides
- C12N2501/91—Heparin
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2506/00—Differentiation of animal cells from one lineage to another; Differentiation of pluripotent cells
- C12N2506/13—Differentiation of animal cells from one lineage to another; Differentiation of pluripotent cells from connective tissue cells, from mesenchymal cells
- C12N2506/1323—Differentiation of animal cells from one lineage to another; Differentiation of pluripotent cells from connective tissue cells, from mesenchymal cells from skeletal muscle cells
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2513/00—3D culture
Definitions
- CD56+ cells which are CD49a- and CD49e+.
- CD56- cells which are CD146- and SMA- (Lecourt et al., 2010).
- injection site refers to a site within the human body, such as close to or being muscle-tissue providing for anal continence, at which the injection process is initiated.
- the injection site needs not to be identical with the site where the injection process ends.
- CD34- or CD34 negative refers to a cell not expressing the cell marker CD34.
- the terms “CD34-” or “CD34 negative” can also be used for a cell population comprising different cell types, if preferably less than 50% or at most 49, 40, 30, 20, 10, 5, 4, 3, 2, 1 or 0 percent of the cell population express the cell marker CD34.
- the term “CD34-” or “CD34 negative” can be also used for a cell population comprising different cells, if preferably at most 19, 10, 5, 4, 3, 2, 1 or 0 percent of the cell population express the cell marker CD34.
- CD146+ refers to a cell expressing the cell marker CD146.
- CD146+ or “CD146 positive” can also be used for a cell population comprising different cell types, if preferably at least 50, 60, 70, 80, 90, 95, 98 or 99 percent of the cell population express the cell marker CD 146.
- CD146- or CD146 negative refers to a cell not expressing the cell marker CD146.
- the terms “CD146-” or “CD146 negative” can also be used for a cell population comprising different cell types, if preferably less than 50% or at most 49, 40, 30, 20, 10, 5, 4, 3, 2, 1 or 0 percent of the cell population express the cell marker CD146.
- the term "desmin positive” or “desmin+” as used herein refers to a cell expressing the cell marker desmin.
- the term “desmin positive” can also be used for a cell population comprising different cell types, if preferably at least 50, 60, 70, 80, 90, 95, 98 or 99 percent of the cell population express the cell marker desmin.
- desmin negative refers to a cell not expressing the cell marker desmin.
- the term “desmin negative” can also be used for a cell population comprising different cell types, if preferably less than 50% or at most 49, 40, 30, 20, 10, 5, 4, 3, 2, 1 or 0 percent of the cell population express the cell marker desmin.
- smoothelin negative or “smoothelin-”as used herein refers to a cell not expressing the cell marker smoothelin.
- smoothelin negative can also be used for a cell population comprising different cell types, if preferably less than 50% or at most 49, 40, 30, 20, 10, 5, 4, 3, 2, 1 or 0 percent of the cell population express the cell marker smoothelin.
- non-fusion competent or “non-skeletal-myogenic” as used herein refers to cells not able to fuse to multinucleated myotubes with less than 50% or at most 49, 30, 20, 10 or 0 percent of nuclei within multinucleated myotubes following cultivation in skeletal muscle differentiation media for 5 - 7 days.
- smooth muscle differentiation media refers to cell culture media which induce transdifferentiation of cells to a smooth muscle phenotype. However, said term refers also to cell culture medium not comprising any substances necessary for the induction of transdifferentiation, in case cells are able to transdifferentiate without a respective induction.
- cell growth medium refers to any medium suitable for the incubation of mammalian cells such as SMDCs, which allows the attachment of said mammalian cells on the surface of an incubation container as well as their proliferation.
- non-contractile refers to collagen gel lattice contraction of less than 40 % from the initial gel size within 48 hours.
- TGF-beta is used for the transforming growth factor beta which is a multifunctional cytokine belonging to the transforming growth factor superfamily that includes three different isoforms (TGF- ⁇ 1, 2 and 3) and many other signaling proteins produced by all white blood cell lineages.
- TGF-beta is used synonymously with the terms "TGF- ⁇ ", “TGF-b”, “TGFb” and "TGFB”.
- AChE positive or “AChE+” as used herein refers to an acetylcholinesterase enzymatic activity of at least 1*10 3 mUrel per mg of cellular protein measured in cells that have been cultivated in smooth muscle differentiation medium as e.g. described in the examples herein.
- acetylcholinesterase enzymatic activity can be tested by any test known in the art as e.g. described in Thurner et al., 2018.
- AChE negative or “AChE-” as used herein refers to an acetylcholinesterase enzymatic activity of less than 1*10 3 mUrel per mg of cellular protein measured in cells that have been cultivated in smooth muscle differentiation medium as e.g. described in the examples herein.
- acetylcholinesterase enzymatic activity can be tested by any test known in the art as e.g. described in Thurner et al., 2018.
- CK positive or "CK+” as used herein refers to a creatine kinase activity of at least 1*10 2 mU rel per mg of cellular protein measured in cells that have been cultivated in smooth muscle differentiation medium as e.g. described in the examples herein.
- acetylcholinesterase enzymatic activity can be tested by any test known in the art as e.g. described in Thurner et al., 2018.
- CD49e+ or “CD49 positive” as used herein refers to a cell expressing the cell marker CD49e.
- the terms “CD49e+” or “CD49e positive” can also be used for a cell population comprising different cell types, if preferably at least 50, 60, 70, 80, 90, 95, 98 or 99 percent of the cell population express the cell marker CD49e.
- Pax-7+ or “Pax-7 positive” as used herein refers to a cell expressing the transcription factor Pax-7.
- the terms “Pax-7+” or “Pax-7 positive” can also be used for a cell population comprising different cell types, if preferably at least 50, 60, 70, 80, 90, 95, 98 or 99 percent of the cell population express the cell marker Pax-7.
- SSEA4+ refers to a cell expressing the cell surface marker SSEA4.
- SSEA4+ or “SSEA4 positive” can also be used for a cell population comprising different cell types, if preferably at least 50, 60, 70, 80, 90, 95, 98 or 99 percent of the cell population express the cell marker SSEA4.
- SSEA4- refers to a cell not expressing the cell surface marker SSEA4.
- SSEA4- refers to a cell not expressing the cell surface marker SSEA4.
- SSEA4- or “SSEA4 negative” can also be used for a cell population comprising different cell types, if preferably less than 50% or at most 49, 40, 30, 20, 10, 5, 4, 3, 2, 1 or 0 percent of the cell population express the cell marker SSEA4.
- MPC myogenic progenitor cells.
- MPC refers to myogenic progenitor cells which are characterized by a negative expression of aSMA, CD49a, and CD146.
- MPCs are skeletal-myogenic and not multipotent. MPCs may be further characterized by a positive expression of CD105, CD90, CD73, CD56, desmin, ACHE and/or CK and/or a negative expression of CD34. An example of properties of MPCs is shown in Fig. 15 .
- MSC mesenchymal stromal cells
- MSC mesenchymal stromal cells
- MSCs mesenchymal stromal cells which are characterized by a negative expression of aSMA, CD49a, and CD146.
- MSCs are non-fusion competent and non-skeletal-myogenic but multipotent.
- MSCs may be further characterized by a positive expression of CD105, CD90, CD73 and/or desmin and/or a negative expression of CD56, CD34, desmin, ACHE and/or CK.
- An example of properties of MSCs is shown in Fig. 15 .
- a first subject-matter of the present invention is directed to a method for obtaining induced smooth muscle cells (iSMCs), the method comprising the steps of: (a) obtaining skeletal muscle derived cells from a subject; (b) transdifferentiating skeletal muscle derived cells by cultivating the cells in a medium containing TGF-beta, in particular TGFb1, TGFb2 and/or TGFb3, and heparin to obtain iSMCs.
- the skeletal muscle derived cells are transdifferentiated in step (b) by cultivating the cells in a medium containing TGFb1 and/or TGFb3, more preferably TGFb1, and heparin to obtain iSMCs.
- Step (b) of the present invention is performed in vitro or ex vivo. Accordingly, the method according to the present invention is an in vitro or ex vivo.
- step (b) is conducted in a cell culture medium containing 1-10 ⁇ g/ml TGFb1 and 10-30 ⁇ g/ml Heparin or 1-6 U/ml Heparin.
- the iSMCs obtained according to a method of the present invention are characterized by the positive expression of aSMA, CD49a, and CD146.
- the skeletal muscle derived cells are myogenic progenitor cells (MPCs) characterized by the positive expression of CD56 and desmin, and the negative expression of CD34; alternatively, the skeletal muscle derived cells are mesenchymal stromal cells (MSCs) characterized by the positive expression of CD105, CD73, and the negative expression of CD34, and CD56.
- MPCs myogenic progenitor cells
- MSCs mesenchymal stromal cells
- the skeletal muscle derived cells are oligopotent MPCs.
- the skeletal muscle derived cells are MSCs characterized by the negative expression of desmin and/or the positive expression of CD90.
- the skeletal muscle derived cells are multipotent MSCs.
- the iSMCs obtained from MPCs in step (b) are characterized by the positive expression of aSMA, CD49a, desmin, CD56, and CD146, and the negative expression of CD34; and foresees that the iSMCs obtained from MSCs in step (b) are characterized by the positive expression of aSMA, CD49a and CD146, and the negative expression of CD56.
- the iSMCs obtained from MPCs in step (b) are further characterized by a positive expression of smoothelin.
- the iSMCs obtained from MSCs in step (b) are further characterized by a negative expression of desmin and/or CD34.
- CD105 is also known as Endoglin. It is a type I integral membrane homodimer protein with subunits of 90 kD found on vascular endothelial cells and syncytiotrophoblasts of placenta. CD105 is weakly expressed on stromal fibroblasts. It is also expressed on activated monocytes and tissue macrophages. Expression of CD105 is increased on activated endothelium in tissues undergoing angiogenesis, such as in tumors, or in cases of wound healing or dermal inflammation. CD105 is a component of the TGF- ⁇ receptor system in human umbilical vein endothelial cells and binds TGF- ⁇ 1 and ⁇ 3 with high affinity.
- CD105 is a multipotent mesenchymal stromal cell (MSC) marker according the minimal criteria for MSC as suggested by the international society for cellular therapy (ISCT)(Dominici et al., 2006).
- ISCT international society for cellular therapy
- CD105 expression due to its role as a TGFb co- receptor can be helpful for the success of the method described herein.
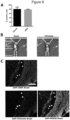
- the present invention thus, discloses isolation of MSCs or MPCs (Example 1) that are CD105 positive ( Figure 2 ) and therefore useful for isolation of iSMCs (Example 2).
- CD34 expression was described in muscle derived stem cells and quiescent satellite cells (Qu-Petersen et al., 2002). Further, CD34 positive skeletal muscle derived cells displayed enhanced regeneration of dystrophin in dystrophic skeletal muscle (Jankowski et al., 2002). State of the art is the use of CD34+ skeletal muscle derived cells for generation of smooth muscle cells in vitro and the use of CD34+ skeletal muscle derived cells for smooth muscle augmentation (Capelli et al., 2002).
- CD34 negative skeletal muscle derived cells are obtained in a first step and differentiated to CD34 negative iSMCs in a second step (Examples 1 and 2).
- CD146 is a surface protein and receptor of laminin alpha 4, which is present in the extracellular matrix of developing smooth muscle tissue (Iivanainen et al., 1995). Further, CD146 was shown to be expressed in bone marrow derived stem cells committed to the smooth muscle lineage (Espagnolle et al., 2014). As depicted in Figure 2 , bladder derived smooth muscle cells are positive for CD146. Summarizing CD146+ cells mark a population of smooth muscle committed cells as preferred for the use of smooth muscle regeneration and or tissue engineering. The present methods allow isolation of CD146+ iSMCs from skeletal muscle derived cells.
- Alpha smooth muscle actin (aSMA) is one of the first markers for smooth muscle commitment during development (McHugh, 1995). Its presence is essential for the function and contractility due to mechanotransduction in smooth muscle cells (J. Wang et al., 2006). Thus, the use of aSMA as a marker for iSMCs, intended for the regeneration of smooth muscle function, is essential.
- the present methods (Example 2) allow generation of aSMA+ cells from skeletal muscle derived cells ( Figure 3 ).
- Desmin is one of the earliest known myogenic markers present in all muscle types. Lack of desmin can result in muscle degeneration and malfunction (Capetanaki et al., 1997). Thus, the use of desmin+ cells for muscle regeneration is to be preferred. As it is shown in figure 3 , iSMCs derived from MPCs are desmin positive.
- smooth muscle tissue relies on the existence of highly differentiated smooth muscle cells expressing contractile proteins such as smoothelin (Niessen et al., 2005). Further, smoothelin is a well-known marker for fully differentiated smooth muscle cells and is the first marker to disappear when smooth muscle is compromised (van Eys et al., 2007). Thus, the use of smoothelin+ cells in regeneration of smooth muscle tissue might be favorable. Present methods (Example 2 and 5) allow isolation of smoothelin+ iSMCs from smoothelin- MPCs ( Figure 3 ).
- markers CD146, CD56, aSMA, CD34, desmin, CD49a, smoothelin are important for the identification and/or function of smooth muscle cells, combination of the markers might be favorable to identify iSMCs suitable for smooth muscle regeneration.
- the herein demonstrated methods allow isolation of iSMCs with combinations of the addressed markers (Example 2).
- the present invention provides a method to obtain iSMCs from skeletal muscle derived MPCs.
- the iSMCs obtained from MPCs are CD56+, aSMA+, CD49a+, desmin+, CD146+ and CD34-.
- the latter markers as described above are advantageous to identify smooth muscle cells, as for example hBd-SMCs are positive for CD146 and CD49a ( Figure 2 ) and thus are preferred for cells used as smooth muscle regenerating cells.
- the combination of CD56+, aSMA+, CD49a+, desmin+, CD146+ and CD34- marker expression is advantageous and novel for in vitro generated iSMCs from human skeletal muscle derived MPCs.
- the method comprises that after step (a) a step (a1) is conducted comprising proliferating the skeletal muscle derived cells, preferably to receive 20-40 ⁇ 10 6 cells.
- step (b) is conducted for one to six days. In a particular preferred embodiment of the present invention, step (b) is conducted three to six days.
- a further subject-matter of the present invention is directed to induced smooth muscle cells (iSMCs) obtained by a method according to the present invention.
- iSMCs induced smooth muscle cells
- the induced smooth muscle cells (iSMCs) obtained from MPCs are characterized by the positive expression of aSMA, CD49a, desmin, CD56, and CD146, and the negative expression of CD34.
- iSMCs from MPCs are advantageous due to pheno-copying the natural expression profile of smooth muscle cells by the methods described herein.
- the induced smooth muscle cells (iSMCs) obtained from MPCs are non-fusion competent.
- MSCs were rinsed with growth medium and centrifuged at 400*g for 10 minutes, supernatant was discarded and the pellet resuspended in growth medium.
- MSCs were purified by magnetic activated cell sorting (MACS). Therefore, human CD56 MicroBeads kit (MiltenyiBiotec GmbH, Bergisch Gladbach, Germany) was used.
- MACS-buffer magnetic activated cell sorting
- adipocytes chondrocytes and osteocytes visualized by oil red o (adipocytes), alcian blue (chondrocytes) and alizarin red s (osteocytes) staining, respectively.
- Quantification of oil red o, alcian blue and alizarin red s staining was performed on microscopic images of multiple individual experiments by image j software package. Therefore, images were loaded and color channels split. Red channels were used for oil red o and alizarin red s stainings, while blue channel was used for alcian blue stainings.
- Upregulated genes of the KEGG cluster or known smooth muscle marker genes in MPC-iSMCs compared to MPCs were PPP1R14A, KCNMB1, PLCB4, ACTG2, ITPR1, ADCY6, CALCRL, KCNMA1, GNA13, CNN1, ADCY2, KCNMB4, GUCY1A3, ARAF, ITGA1, PPP1R12A, MAPK1, CALD1, KCNMB2, PRKACB, ARHGEF11, PPP1R12C, ITPR2, PLCB1 and SMTN ( Figure 12 ).
- Example 2 Cells obtained by Example 2 were found neither to exhibit voltage sensitive inward nor outward currents of voltage dependent calcium or voltage dependent potassium channels, respectively.
- iSMCs derived from MPCs in Example 2 showed both, voltage sensitive inward and outward currents, as also found in hBd-SMCs ( Figure 6 ). Summarizing, the isolation of iSMCs by incubation with TGFb1 and Heparin results in functional maturation.
- Example 2 In order to measure the contractility of MSCs and MPCs obtained according to Example 1 as well as iSMCs derived thereof by Example 2 were seeded in collagen gel lattice and reduction of percent gel size was quantified.
- culture media from sub-confluent cells in standard cell culture vessels were removed and cells were washed twice with 1X PBS. Next, cells were covered with trypsin and incubated for 5 minutes at 37°C. Afterwards, cells were detached by tapping against the walls of the culture vessel and resuspended following addition of DMEM/Ham's F12 basal medium. Then, cells were centrifuged at 400*g for 10 minutes.
- each gel was covered with 500 ⁇ l DMEM/Ham'sF12 and released from the bottom of the 24 well plate to float on the surface by using a sterile pipette tip. Finally, the gels were incubated at 37°C, 5% CO 2 for 24 hours to allow gel contraction by included cells. To quantify gel contraction, stereomicroscopic pictures were taken and the area of the gel was calculated by applying FIJI (image J) software.
- MPCs and MSCs isolated according to step (a) of the present invention were found to be non-contractile in collagen gel lattice contraction assay.
- iSMCs isolated according to step (b) of the present invention from MPCs and MSCs did show significantly higher contractility than cells originating from (MPCs, MSCs).
- SMDCs transdifferentiated to iSMCs and isolated according to step (b) were found to be contractile compared to cells isolated in step (a) (Example 1; Figure 7 ).
- iSMCs derived from murine MPCs were injected into the pyloric sphincter of adult female SHO-Prkdc scid Hr hr mice.
- mice were first anesthetized by applying 100 mg/kg Ketamin; 10 mg/kg Xylazin and 3 mg/kg Acepromazin intraperitoneally. Eye protecting cream was applied during the procedure.
- Cryopreserved cells were freshly thawed, washed once with 1X PBS and centrifuged at 400*g for 10 minutes followed by resuspending the cells in 1X PBS to reach a final concentration of 40 000 000 cells/ml. Meanwhile the mice that receive cells were placed on a heating plate to maintain body temperature at 37°C. 25 ⁇ l of the cell suspension (containing 1 000 000 cells) was then mixed with 5 ⁇ l FluoSpheres ® polystyrene beads, 15 ⁇ m, yellow-green or blue (Thermo-Fisher Scientific, MA, USA), necessary to track the location of the injection after surgery.
- a median laparotomy was performed followed by localization of the pyloric sphincter region and application of 30 ⁇ l cell- fluosphere mixture using a 28G needle attached to a 1 ml syringe.
- the peritoneum and muscle-skin layer was closed separately by consecutive stitching with 6-0 ethicon PDS plus absorbable monofilaments.
- Postoperatively 200 mg Novalgin (Metamizol ® ) per kilogram bodyweight was applied subcutaneously for three days. 12 weeks after cell injection, mice were sacrificed by cervical dislocation to obtain and image pyloric sphincter muscles.
- Imaging of fresh isolated pyloric sphincter regions was performed with an IVIS Spectrum (PerkinElmer, MA, USA) by using Living Image ® software version 4.5.2 (PerkinElmer, MA, USA) according to manufacturer's instructions.
- IVIS Spectrum PerkinElmer, MA, USA
- pyloric sphincters of injected and control SHO-mice were placed on a glass petri dish and placed within the IVIS system. Fluorescence pictures at a height of 2 cm with automated exposure times for corresponding absorption and emission wavelengths of TdTomato and yellow Fluosphere beads were taken. Post hoc, signal intensities were adjusted in order to get rid of background signals by comparing with sphincter explants from control mice.
- Blocking and antibody dilution were performed using a PBS solution containing 1 % bovine serum albumin fraction (Sigma-Aldrich), 0.2 % fish skin gelatin (Sigma-Aldrich) and 0.1 % Tween-20 (Sigma- Aldrich).
- Primary antibodies against tdTomato (Sicgen) or aSMA (Thermo Scientific, MA, USA) were diluted 1:100 in blocking media following incubation was performed over night at 4°C. Secondary antibodies (Thermo scientific) were diluted 1:500 and applied at room temperature for 4 hours. Nuclei were stained with DAPI diluted to 0.5 ⁇ g/ml working concentration (Sigma-Aldrich). Slices were subsequently mounted using Prolong Gold Antifade (Life Technologies). Fluorescence images were acquired using a LSM 710 confocal microscope and ZEN 2011 Black Software (Carl Zeiss).
- iSMCs isolated according to Example 2 from TdTomato reporter protein expressing MPCs obtained according to Example 1 was detectable and co-localized with co-injected fluorescent beads at the site of the pyloric sphincter 12 weeks post implantation, suggesting the engraftment of iSMCs at the site of injection. Histological examination followed by fluorescence immunostaining for TdTomato and aSMA suggested that TdTomato positive iSMCs cells were found within the pyloric sphincter circular muscle as well as muscularis mucosa nearby co-injected fluorescent beads ( Figure 8 ).
- TdTomato positive iSMCs cells located within the smooth muscle layer of the pyloric sphincter also expressed aSMA protein, suggesting not only the engraftment into the smooth muscle tissue but also conserved phenotypic characteristics of smooth muscle cells after engraftment necessary for smooth muscle regeneration ( Figure 8 ).
- a 2% (w/v) agarose solution was prepared by weighing in 5 g low gelling agarose (Sigma-Aldrich, MA, USA) in a Schott ® flask filled up to 250 ml with Ham's F10 basal medium. Then, both the PDMS template and agarose solution was autoclaved at 121°C for 20 minutes. Afterwards, The PDMS template was filled with the agarose solution under sterile conditions followed by incubation at room temperature for 1 hour to let the agarose solidify.
- the solidified agarose was released from the PDMS template, cubes each containing one ring shaped template were cut out of the agarose and transferred to a 6-well plate filled with 2 ml Ham's F10 basal medium followed by storage at 37°C until further use.
- cells obtained in Example 1 were centrifuged at 400*g for 10 minutes and the cell pellet was resuspended in culture medium to reach a concentration of 5 ⁇ 10 6 cells/ml.
- 200 ⁇ l of the cell suspension was added into the agarose template followed by incubation at 37°C for 48 hours without further disturbances to allow ring formation of the cells.
- Example 1 medium outside the template within the 6-well plate was discarded and the 6- well plate was carefully filled with 5 ml medium as used in Example 1 to produce 3D cultured MPCs or MSCs or any of the latter as used in Example 2 to produce iSMCs 3D cultures.
- Cells were cultivated for 6 days to allow maturation at 37°C, 5% CO 2 until analysis. After 6 days of cultivation, cells were analyzed by standard light microscopy, cryo-sectioning followed by immunostaining (Example 12) or H&E staining, scanning electron microcopy or semi/ultra- thin sectioning followed by toluidine blue staining or transmission electron microcopy.
- MPCs derived according to Example 1 that were transdifferentiated towards iSMCs as described in Example 2 did form ring shaped sphincters employing the above described methods based on Gwyther et al., 2014 ( Figure 9 ).
- rings were carefully removed from the agarose template and using a sterile spoon, washed by transferring into a Eppendorf tube filled with 1XPBS and then fixed and permeabilized by submerging in -20°C pre-cooled MetOH for 5 minutes. Next, rings were washed by diluting MetOH three times with 1XPBS following removal of half of the solution. Bioengineered sphincters were cryo-fixed by plunging into liquid nitrogen cooled 2-methylbutane.
- Example 2 MPCs obtained according to Example 1 and transdifferentiated towards iSMCs as described in Example 2 during 3D cultivation (Example 2) appeared elongated and as an integral part of the tissue ring on the surface of the tissue rings that had formed in Example 10 ( Figure 9 ). Cells appeared intact on the surface of the ring and potentially viable ( Figure 9 ).
- rings were de-liquidized in increasing EtOH concentrations (70, 80, 90, 100 %), each 30 minutes and then incubated twice in fresh 100 % Aceton for each 20 minutes.
- specimens were embedded in 2:1 Aceton-Epon mixtures for 150 minutes followed by incubation in 1:2 Aceton-Epon mixture overnight.
- rings were incubated in pure Epon rotating on a rotator for 24 hours with a change of Epon after 8 hours.
- rings were incubated at 60°C for 24 hours.
- Epon embedded specimen were trimmed using an Ultratrim (Reichert) and ultra-thin sections, were obtained using an Ultracut S (Reichert). Ultrathin sections were viewed at 80 kV with a CM120 TEM (from Philips/FEI) equipped with a MORADA CCD-camera (from Olympus/SIS).
- Cell events were acquired by employing Guava InCyte TM v.2.3 software. Histograms were generated with a minimum of 3000 events with a sample flow rate of 1.8 ⁇ l/ml. The percentage of positive cells was obtained by comparison with isotype control set as 99% negative.
- Acetylcholine thioiodide (ATI, 5.76 mM) was prepared freshly for each AChE assay by weighing out 2 mg in 1.5 mL eppendorf tube. It was dissolved in 1.2 mL of distil water by vortexing for 1-2 minutes and then stored at 4°C until use.
- AChE mU rel was calculated based on OD412 values obtained from 60 min colorimetric measurement of cells by extrapolation of linear standard curves derived from AChE standard measurements (OD at 412nm after 7 or 8 minutes).
- AChE mU rel /mg protein values were then calculated by dividing AChE mU rel with mg of total protein (calculated according to Example 19) of corresponding cells cultivated in skeletal muscle differentiation medium.
- Lysis buffer was prepared by adding 10 ⁇ l of Triton-X-100 to 10 ml of dH 2 O (LC-MS-Ultrachromasol, Fluka).
- CK-NAC Thermo Scientific, MA, USA
- CK-NAC previously dissolved by adding 10 ml dH 2 O
- the reaction was analyzed in an Anthos Zenith 340rt microplate reader (Biochrom Ltd., Cambridge, UK) set to 30°C, by OD absorbance measurement at 340 nm. If not otherwise mentioned, OD340 nm values taken 21 minutes after addition of CK-NAC, were used for subsequent analysis.
- CK activity in mU rel was calculated according to manufacturer's instructions and if not otherwise mentioned normalized by dividing it by mg total protein of corresponding cells.
- the clear supernatant was transferred into a fresh Eppendorf tube and the protein concentration was determined using the Pierce BCA Protein Assay Kit (Thermo Scientific, MA, USA) according to the manufacturer's instructions by measuring the OD at 540 nm with an Anthos Zenyth 340rt microplate reader (Biochrom Ltd., Cambridge, UK).
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Biomedical Technology (AREA)
- Chemical & Material Sciences (AREA)
- Zoology (AREA)
- Biotechnology (AREA)
- Organic Chemistry (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Cell Biology (AREA)
- General Health & Medical Sciences (AREA)
- Wood Science & Technology (AREA)
- Genetics & Genomics (AREA)
- Developmental Biology & Embryology (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Medicinal Chemistry (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Rheumatology (AREA)
- Microbiology (AREA)
- Biochemistry (AREA)
- General Engineering & Computer Science (AREA)
- Cardiology (AREA)
- Immunology (AREA)
- Virology (AREA)
- Vascular Medicine (AREA)
- Epidemiology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Physical Education & Sports Medicine (AREA)
- Orthopedic Medicine & Surgery (AREA)
- Neurology (AREA)
- Medicines Containing Material From Animals Or Micro-Organisms (AREA)
- Micro-Organisms Or Cultivation Processes Thereof (AREA)
- Peptides Or Proteins (AREA)
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP19164574 | 2019-03-22 | ||
| PCT/EP2020/057940 WO2020193460A1 (en) | 2019-03-22 | 2020-03-23 | Methods for obtaining induced smooth muscle cells |
| EP20711954.6A EP3942020B1 (de) | 2019-03-22 | 2020-03-23 | Verfahren zur gewinnung von induzierten glatten muskelzellen |
Related Parent Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP20711954.6A Division EP3942020B1 (de) | 2019-03-22 | 2020-03-23 | Verfahren zur gewinnung von induzierten glatten muskelzellen |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP4397752A2 true EP4397752A2 (de) | 2024-07-10 |
| EP4397752A3 EP4397752A3 (de) | 2024-10-02 |
Family
ID=65904308
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP24175168.4A Withdrawn EP4397752A3 (de) | 2019-03-22 | 2020-03-23 | Verfahren zur gewinnung von induzierten glatten muskelzellen |
| EP20711954.6A Active EP3942020B1 (de) | 2019-03-22 | 2020-03-23 | Verfahren zur gewinnung von induzierten glatten muskelzellen |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP20711954.6A Active EP3942020B1 (de) | 2019-03-22 | 2020-03-23 | Verfahren zur gewinnung von induzierten glatten muskelzellen |
Country Status (14)
| Country | Link |
|---|---|
| US (2) | US12312600B2 (de) |
| EP (2) | EP4397752A3 (de) |
| JP (2) | JP7616665B2 (de) |
| KR (1) | KR20220002315A (de) |
| CN (2) | CN113614223B (de) |
| AU (1) | AU2020247211B2 (de) |
| BR (1) | BR112021016264A2 (de) |
| CL (1) | CL2021002169A1 (de) |
| DK (1) | DK3942020T3 (de) |
| ES (1) | ES2987152T3 (de) |
| IL (1) | IL286509A (de) |
| MX (1) | MX2021009862A (de) |
| WO (1) | WO2020193460A1 (de) |
| ZA (1) | ZA202104948B (de) |
Families Citing this family (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11617768B2 (en) | 2017-12-14 | 2023-04-04 | Innovacell Ag | Methods for obtaining muscle derived cells |
| JP7616665B2 (ja) | 2019-03-22 | 2025-01-17 | インノヴァセル ゲーエムベーハー | 誘導平滑筋細胞を得るための方法 |
| US20250114405A1 (en) | 2021-08-06 | 2025-04-10 | Innovacell Gmbh | Myogenic progenitor cells for use in an optimized method for the prevention and treatment of anal incontinence |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2001078754A2 (en) | 2000-04-14 | 2001-10-25 | University Of Pittsburgh | Soft tissue and bone augmentation and bulking utilizing muscle-derived progenitor cells, compositions and treatments thereof |
| EP2206774A1 (de) | 2008-12-23 | 2010-07-14 | Genzyme Corporation | Von Muskeln stammende Zellen mit Differenzierungsfähigkeiten |
Family Cites Families (20)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5709854A (en) * | 1993-04-30 | 1998-01-20 | Massachusetts Institute Of Technology | Tissue formation by injecting a cell-polymeric solution that gels in vivo |
| US6555374B1 (en) | 1999-08-19 | 2003-04-29 | Artecel Sciences, Inc. | Multiple mesodermal lineage differentiation potentials for adipose tissue-derived stromal cells and uses thereof |
| WO2003027281A2 (en) | 2001-09-20 | 2003-04-03 | Kyowa Hakko Kogyo Kk | Pluripotent stem cells originating in skeletal muscle intestinal tissue |
| CN1812800B (zh) | 2003-04-25 | 2013-01-16 | 匹兹堡大学 | 用于促进和增强神经修复和再生的肌肉来源的细胞(mdc) |
| US20080213231A1 (en) | 2005-07-15 | 2008-09-04 | Kyoto University | Pluripotent Stem Cell Cloned From Single Cell Derived From Skeletal Muscle Tissue |
| FR2890977A1 (fr) | 2005-09-19 | 2007-03-23 | Assist Publ Hopitaux De Paris | Procede d'obtention de cellules musculaires lisses humaines et leurs applications |
| US20100273231A1 (en) | 2005-09-20 | 2010-10-28 | Andreadis Stelios T | Multipotent mesenchymal stem cells from human hair follicles |
| TW200736399A (en) | 2006-01-10 | 2007-10-01 | Theradigm Inc | Expansion and differentiation of neural stem cells under low oxygen conditions |
| EP2097088B2 (de) | 2006-11-28 | 2024-06-12 | University of Pittsburgh - of the Commonwealth System of Higher Education | Zellen aus muskeln zur behandlung von herzerkrankungen sowie verfahren zu ihrer herstellung und verwendung |
| US20080208164A1 (en) | 2007-02-28 | 2008-08-28 | Innovacell Biotechnologie Gmbh | Methods for the treatment of anal incontinence |
| AU2008263134B2 (en) | 2007-05-29 | 2014-06-19 | University Of Pittsburgh-Of The Commonwealth System Of Higher Education | Bone augmentation utilizing muscle-derived progenitor compositions, and treatments thereof |
| US20140030236A1 (en) | 2010-02-25 | 2014-01-30 | The Johns Hopkins University | Derivation and maturation of synthetic and contractile vascular smooth muscle cells from human pluripotent stem cells |
| JP2013525077A (ja) * | 2010-05-03 | 2013-06-20 | テンジオン, インク. | 平滑筋細胞構築物 |
| US20150250826A1 (en) * | 2012-09-06 | 2015-09-10 | Joslin Diabetes Center, Inc. | Isolation and characterization of muscle regenerating cells |
| EP2898089A1 (de) * | 2012-09-24 | 2015-07-29 | Innovacell Biotechnologie AG | Stärketest für aus skelettmuskeln abgeleitete zellen |
| EP3056562A1 (de) * | 2015-02-11 | 2016-08-17 | University College Cork-National University of Ireland, Cork | Induzierte glatte muskelzellen, verfahren zur herstellung davon und verwendungen davon bei der zellbasierten geweberegenerierung und behandlung von gefässverengung |
| CN106350480B (zh) | 2016-08-26 | 2019-11-08 | 中国农业科学院北京畜牧兽医研究所 | 一种纯化牛胎儿骨骼肌组织来源成肌细胞的方法 |
| US11617768B2 (en) | 2017-12-14 | 2023-04-04 | Innovacell Ag | Methods for obtaining muscle derived cells |
| JP7616665B2 (ja) | 2019-03-22 | 2025-01-17 | インノヴァセル ゲーエムベーハー | 誘導平滑筋細胞を得るための方法 |
| US20250114405A1 (en) | 2021-08-06 | 2025-04-10 | Innovacell Gmbh | Myogenic progenitor cells for use in an optimized method for the prevention and treatment of anal incontinence |
-
2020
- 2020-03-23 JP JP2021552231A patent/JP7616665B2/ja active Active
- 2020-03-23 EP EP24175168.4A patent/EP4397752A3/de not_active Withdrawn
- 2020-03-23 EP EP20711954.6A patent/EP3942020B1/de active Active
- 2020-03-23 CN CN202080021489.4A patent/CN113614223B/zh active Active
- 2020-03-23 AU AU2020247211A patent/AU2020247211B2/en active Active
- 2020-03-23 KR KR1020217034375A patent/KR20220002315A/ko active Pending
- 2020-03-23 ES ES20711954T patent/ES2987152T3/es active Active
- 2020-03-23 DK DK20711954.6T patent/DK3942020T3/da active
- 2020-03-23 MX MX2021009862A patent/MX2021009862A/es unknown
- 2020-03-23 BR BR112021016264-0A patent/BR112021016264A2/pt unknown
- 2020-03-23 WO PCT/EP2020/057940 patent/WO2020193460A1/en not_active Ceased
- 2020-03-23 CN CN202510688777.XA patent/CN120738111A/zh active Pending
- 2020-03-23 US US17/441,690 patent/US12312600B2/en active Active
-
2021
- 2021-07-14 ZA ZA2021/04948A patent/ZA202104948B/en unknown
- 2021-08-16 CL CL2021002169A patent/CL2021002169A1/es unknown
- 2021-09-19 IL IL286509A patent/IL286509A/en unknown
-
2024
- 2024-10-11 JP JP2024179303A patent/JP2025011242A/ja active Pending
-
2025
- 2025-03-10 US US19/075,289 patent/US20250207097A1/en active Pending
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2001078754A2 (en) | 2000-04-14 | 2001-10-25 | University Of Pittsburgh | Soft tissue and bone augmentation and bulking utilizing muscle-derived progenitor cells, compositions and treatments thereof |
| EP2206774A1 (de) | 2008-12-23 | 2010-07-14 | Genzyme Corporation | Von Muskeln stammende Zellen mit Differenzierungsfähigkeiten |
Non-Patent Citations (44)
| Title |
|---|
| ABRAHAMSSON, H., GUT, vol. 56, no. 6, 2007, pages 877 - 883 |
| AL-ALI, S.BLYTH, P.BEATTY, S.DUANG, A.PARRY, B.BISSETT, I. P., JOURNAL OF ANATOMY, vol. 215, no. 2, 2009, pages 212 - 220 |
| BAJPAI, V. K., MISTRIOTIS, P., LOH, Y.-H., DALEY, G. Q., & ANDREADIS, S. T., CARDIOVASCULAR RESEARCH, vol. 96, no. 3, 2012, pages 391 - 400 |
| BELKIN, V. M.BELKIN, A. M.KOTELIANSKY, V. E., THE JOURNAL OF CELL BIOLOGY, vol. 111, no. 5, 1990, pages 2159 - 2170 |
| BOHL, J. L.ZAKHEM, E.BITAR, K. N., STEM CELLS TRANSLATIONAL MEDICINE, vol. 6, no. 9, 2017, pages 1795 - 1802 |
| CAPETANAKI, Y.MILNER, D. J.WEITZER, G., CELL STRUCTURE AND FUNCTION, vol. 22, no. 1, 1997, pages 103 - 116 |
| DASH, B. C.LEVI, K.SCHWAN, J.LUO, J.BARTULOS, O.WU, H.QIU, C.YI, T.REN, Y.CAMPBELL, S., STEM CELL REPORTS, vol. 7, no. 1, 2016, pages 19 - 28 |
| DOMINICI, M.LE BLANC, K.MUELLER, I.SLAPER-CORTENBACH, I.MARINI, F.KRAUSE, D.DEANS, R.KEATING, A.PROCKOP, D.HORWITZ, E., CYTOTHERAPY, vol. 8, no. 4, 2006, pages 315 - 317 |
| ESPAGNOLLE, N., GUILLOTON, F., DESCHASEAUX, F., GADELORGE, M., SENSEBE, L.,; BOURIN, P., JOURNAL OF CELLULAR AND MOLECULAR MEDICINE, vol. 18, no. 1, 2014, pages 104 - 114 |
| FRUDINGER, A.KOLLE, D.SCHWAIGER, W.PFEIFER, J.PAEDE, J.HALLIGAN, S., GUT, vol. 59, no. 1, 2010, pages 55 - 61 |
| FRUDINGER, ANDREA, MARKSTEINER, R., PFEIFER, J., MARGREITER, E., PAEDE, J.,; THURNER, M., STEM CELL RESEARCH & THERAPY, vol. 9, no. 1, 2018, pages 233 |
| GOODE, P. S., BURGIO, K. L., HALLI, A. D., JONES, R. W., RICHTER, H. E., REDDEN, D. T., BAKER, P. S.,ALLMAN, R. M., JOURNAL OF THE AMERICAN GERIATRICS SOCIETY, vol. 53, no. 4, 2005, pages 629 - 635 |
| HUARD, J., YOKOYAMA, T., PRUCHNIC, R., QU, Z., LI, Y., LEE, J. Y., SOMOGYI, G. T., DE GROAT, W. C.,CHANCELLOR, M. B., GENE THERAPY, vol. 9, no. 23, 2002, pages 1617 - 1626 |
| IIVANAINEN, A.SAINIO, K.SARIOLA, H.TRYGGVASON, K., FEBS LETTERS, vol. 365, no. 2-3, 1995, pages 183 - 188 |
| JUNG, Y.BAUER, G.NOLTA, J. A., STEM CELLS (DAYTON, OHIO), vol. 30, no. 1, 2012, pages 42 - 47 |
| KRAUSS, R. S.CHIHARA, D.ROMER, A. I., SKELETAL MUSCLE, vol. 6, 2016 |
| LECOURT, S., MAROLLEAU, J.-P., FROMIGU6, O., VAUCHEZ, K., ANDRIAMANALIJAONA, R., TERNAUX, B., LACASSAGNE, M.-N., ROBERT, I., BOUME, EXPERIMENTAL CELL RESEARCH, vol. 316, no. 15, 2010, pages 2513 - 2526 |
| LI, Y., WEN, Y., WANG, Z., WEI, Y., WANI, P., GREEN, M., SWAMINATHAN, G., RAMAMURTHI, A., PERA, R. R.,CHEN, B., STEM CELLS TRANSLATIONAL MEDICINE, vol. 5, no. 12, 2016, pages 1719 |
| LU, S.-H.LIN, A. T. L.CHEN, K.-K.CHIANG, H. S.CHANG, L. S., JOURNAL OF CELLULAR AND MOLECULAR MEDICINE, vol. 15, no. 3, 2011, pages 587 - 592 |
| MEYER, I.RICHTER, H. E., WOMEN'S HEALTH (LONDON, ENGLAND), vol. 11, no. 2, 2015, pages 225 - 238 |
| MIMURA, T.KAMINISHI, M.KAMM, M. A., DIGESTIVE SURGERY, vol. 21, no. 3, 2004, pages 235 - 241 |
| NIESSEN, P.RENSEN, S.DEURSEN, J. VANMAN, J. D.LAET, A. D.VANDERWINDEN, J.-M.WEDEL, T.BAKER, D.DOEVENDANS, P.HOFKER, M., GASTROENTEROLOGY, vol. 129, no. 5, 2005, pages 1592 - 1601 |
| PARK, W. S., HEO, S. C., JEON, E. S., HONG, D. H., SON, Y. K., KO, J.-H., KIM, H. K., LEE, S. Y., KIM, J. H.,HAN, J., AMERICAN JOURNAL OF PHYSIOLOGY. CELL PHYSIOLOGY, vol. 305, no. 4, 2013 |
| POPESCU, L. M.GHERGHICEANU, M.MANDACHE, E.CRETOIU, D., JOURNAL OF CELLULAR AND MOLECULAR MEDICINE, vol. 10, no. 4, 2006, pages 960 - 990 |
| QUANDER, C. R.MORRIS, M. C.MELSON, J.BIENIAS, J. L.EVANS, D. A., THE AMERICAN JOURNAL OF GASTROENTEROLOGY, vol. 100, no. 4, 2005, pages 905 - 909 |
| QU-PETERSEN, Z., DEASY, B., JANKOWSKI, R., IKEZAWA, M., CUMMINS, J., PRUCHNIC, R., MYTINGER, J., CAO, B., GATES, C., WERNIG, A.,HU, THE JOURNAL OF CELL BIOLOGY, vol. 157, no. 5, 2002, pages 851 - 864 |
| RAMKUMAR, D.,SCHULZE, K. S., JOURNAL OF THE EUROPEAN GASTROINTESTINAL MOTILITY SOCIETY, vol. 1, 2005, pages 22 - 30 |
| RAO, S. S. C., GASTROENTEROLOGY, vol. 126, no. 1, 2004, pages S14 - 22 |
| ROCHLIN, K.YU, S.ROY, S.BAYLIES, M. K., DEVELOPMENTAL BIOLOGY, vol. 341, no. 1, 2010, pages 66 - 83 |
| ROMANSKA, H. M., BISHOP, A. E., MOSCOSO, G., WALSH, F. S., SPITZ, L., BRERETON, R. J.,POLAK, J. M., JOURNAL OF PEDIATRIC GASTROENTEROLOGY AND NUTRITION, vol. 22, no. 4, 1996, pages 351 - 358 |
| SANDERS, K. M., NEUROGASTROENTEROLOGY AND MOTILITY: THE OFFICIAL JOURNAL OF THE, vol. 20, 2008, pages 39 - 53 |
| SANDISON, M.MCCARRON, J., THE FASEB JOURNAL, vol. 29, 2015, pages 418 |
| SKUK, D.GOULET, M.TREMBLAY, J. P, CELL TRANSPLANTATION, vol. 23, no. 1, 2014, pages 13 - 25 |
| THURNER, M.ASIM, F.GARCZARCZYK-ASIM, D.JANKE, K.DEUTSCH, M.MARGREITER, E.TROPPMAIR, J.MARKSTEINER, R., PLOS ONE, vol. 13, no. 3, 2018, pages e0194561 |
| VAIZEY, C. J.KAMM, M. A.BARTRAM, C. I., LANCET (LONDON, ENGLAND), vol. 349, no. 9052, 1997, pages 612 - 615 |
| VAN DE RIJN, M.HENDRICKSON, M. R.ROUSE, R. V., HUMAN PATHOLOGY, vol. 25, no. 8, 1994, pages 766 - 771 |
| VAN EYS, G. J.NIESSEN, P. M.RENSEN, S. S., TRENDS IN CARDIOVASCULAR MEDICINE, vol. 17, no. 1, 2007, pages 26 - 30 |
| WANG, G.JACQUET, L.KARAMARITI, E.XU, Q., THE JOURNAL OF PHYSIOLOGY, vol. 593, no. 14, 2015, pages 3013 - 3030 |
| WANG, J. Y.ABBAS, M. A., THE PERMANENTE JOURNAL, vol. 17, no. 3, 2013, pages 65 - 73 |
| WANG, J.ZOHAR, R.MCCULLOCH, C. A., EXPERIMENTAL CELL RESEARCH, vol. 312, no. 3, 2006, pages 205 - 214 |
| WANG, Y.HAN, Z.SONG, Y.HAN, Z. C., STEM CELLS INTERNATIONAL, 2012 |
| WEBB, R. C., ADVANCES IN PHYSIOLOGY EDUCATION, vol. 27, no. 4, 2003, pages 201 - 206 |
| WORL, J.BREUER, C.NEUHUBER, W. L., DEVELOPMENTAL DYNAMICS, vol. 238, no. 4, 2009, pages 864 - 874 |
| YIN, H.PRICE, F.RUDNICKI, M. A., PHYSIOLOGICAL REVIEWS, vol. 93, no. 1, 2013, pages 23 - 67 |
Also Published As
| Publication number | Publication date |
|---|---|
| US12312600B2 (en) | 2025-05-27 |
| ES2987152T3 (es) | 2024-11-14 |
| IL286509A (en) | 2021-10-31 |
| JP2022526229A (ja) | 2022-05-24 |
| MX2021009862A (es) | 2021-09-14 |
| NZ778000A (en) | 2025-07-25 |
| DK3942020T3 (da) | 2024-07-15 |
| JP2025011242A (ja) | 2025-01-23 |
| CN120738111A (zh) | 2025-10-03 |
| KR20220002315A (ko) | 2022-01-06 |
| US20250207097A1 (en) | 2025-06-26 |
| US20220145257A1 (en) | 2022-05-12 |
| CN113614223A (zh) | 2021-11-05 |
| ZA202104948B (en) | 2024-08-28 |
| CA3134213A1 (en) | 2020-10-01 |
| EP3942020B1 (de) | 2024-05-15 |
| EP4397752A3 (de) | 2024-10-02 |
| BR112021016264A2 (pt) | 2021-10-13 |
| CN113614223B (zh) | 2025-05-27 |
| CL2021002169A1 (es) | 2022-02-25 |
| WO2020193460A1 (en) | 2020-10-01 |
| AU2020247211A1 (en) | 2021-07-29 |
| JP7616665B2 (ja) | 2025-01-17 |
| EP3942020A1 (de) | 2022-01-26 |
| AU2020247211B2 (en) | 2024-07-04 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US20250207097A1 (en) | Methods for obtaining induced smooth muscle cells | |
| Tamaki et al. | Synchronized reconstitution of muscle fibers, peripheral nerves and blood vessels by murine skeletal muscle-derived CD34−/45− cells | |
| JP7494430B2 (ja) | 筋由来細胞を取得する方法 | |
| Oka et al. | Characterization and localization of side population cells in the lens | |
| CA3134213C (en) | Methods for obtaining induced smooth muscle cells | |
| HK40113062A (en) | Methods for obtaining induced smooth muscle cells | |
| WO2012115298A1 (ko) | 양수 유래 줄기세포를 함유하는 요실금 치료제 | |
| RU2815906C9 (ru) | Способы получения индуцированных клеток гладких мышц | |
| RU2815906C2 (ru) | Способы получения индуцированных клеток гладких мышц | |
| HK40064648B (en) | Methods for obtaining induced smooth muscle cells | |
| HK40064648A (en) | Methods for obtaining induced smooth muscle cells | |
| HK40129485A (zh) | 获得诱导性平滑肌细胞的方法 | |
| HK40063732B (zh) | 获得诱导性平滑肌细胞的方法 | |
| HK40063732A (en) | Methods for obtaining induced smooth muscle cells | |
| HK40098352A (en) | Methods for obtaining muscle derived cells | |
| BR112020011853B1 (pt) | Métodos para obtenção de células derivadas de músculos | |
| HK40033933A (en) | Methods for obtaining muscle derived cells |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: THE APPLICATION HAS BEEN PUBLISHED |
|
| AC | Divisional application: reference to earlier application |
Ref document number: 3942020 Country of ref document: EP Kind code of ref document: P |
|
| AK | Designated contracting states |
Kind code of ref document: A2 Designated state(s): AL AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL PT RO RS SE SI SK SM TR |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R079 Free format text: PREVIOUS MAIN CLASS: C12N0005074000 Ipc: C12N0005077000 |
|
| PUAL | Search report despatched |
Free format text: ORIGINAL CODE: 0009013 |
|
| AK | Designated contracting states |
Kind code of ref document: A3 Designated state(s): AL AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL PT RO RS SE SI SK SM TR |
|
| RIC1 | Information provided on ipc code assigned before grant |
Ipc: C12N 5/074 20100101ALI20240826BHEP Ipc: C12N 5/077 20100101AFI20240826BHEP |
|
| REG | Reference to a national code |
Ref country code: HK Ref legal event code: DE Ref document number: 40113062 Country of ref document: HK |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: THE APPLICATION IS DEEMED TO BE WITHDRAWN |
|
| 18D | Application deemed to be withdrawn |
Effective date: 20250403 |