EP3529416B1 - Security substrate - Google Patents
Security substrate Download PDFInfo
- Publication number
- EP3529416B1 EP3529416B1 EP17791021.3A EP17791021A EP3529416B1 EP 3529416 B1 EP3529416 B1 EP 3529416B1 EP 17791021 A EP17791021 A EP 17791021A EP 3529416 B1 EP3529416 B1 EP 3529416B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- metal oxide
- substrate according
- substrate
- doped
- post
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 239000000758 substrate Substances 0.000 title claims description 56
- 229910044991 metal oxide Inorganic materials 0.000 claims description 36
- 150000004706 metal oxides Chemical class 0.000 claims description 36
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 claims description 20
- 239000002245 particle Substances 0.000 claims description 20
- 239000000049 pigment Substances 0.000 claims description 19
- 239000000945 filler Substances 0.000 claims description 16
- 239000010445 mica Substances 0.000 claims description 12
- 229910052618 mica group Inorganic materials 0.000 claims description 12
- 238000010521 absorption reaction Methods 0.000 claims description 11
- XLOMVQKBTHCTTD-UHFFFAOYSA-N Zinc monoxide Chemical compound [Zn]=O XLOMVQKBTHCTTD-UHFFFAOYSA-N 0.000 claims description 8
- 239000000203 mixture Substances 0.000 claims description 7
- XOLBLPGZBRYERU-UHFFFAOYSA-N tin dioxide Chemical compound O=[Sn]=O XOLBLPGZBRYERU-UHFFFAOYSA-N 0.000 claims description 7
- VTYYLEPIZMXCLO-UHFFFAOYSA-L Calcium carbonate Chemical compound [Ca+2].[O-]C([O-])=O VTYYLEPIZMXCLO-UHFFFAOYSA-L 0.000 claims description 6
- 229910052782 aluminium Inorganic materials 0.000 claims description 5
- WATWJIUSRGPENY-UHFFFAOYSA-N antimony atom Chemical compound [Sb] WATWJIUSRGPENY-UHFFFAOYSA-N 0.000 claims description 5
- 230000005540 biological transmission Effects 0.000 claims description 5
- 229910052752 metalloid Inorganic materials 0.000 claims description 5
- 150000002738 metalloids Chemical class 0.000 claims description 5
- 229910001887 tin oxide Inorganic materials 0.000 claims description 5
- ATJFFYVFTNAWJD-UHFFFAOYSA-N Tin Chemical compound [Sn] ATJFFYVFTNAWJD-UHFFFAOYSA-N 0.000 claims description 4
- 229910052787 antimony Inorganic materials 0.000 claims description 4
- 239000011888 foil Substances 0.000 claims description 4
- 239000000454 talc Substances 0.000 claims description 4
- 229910052623 talc Inorganic materials 0.000 claims description 4
- 229910052718 tin Inorganic materials 0.000 claims description 4
- 239000005995 Aluminium silicate Substances 0.000 claims description 3
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 claims description 3
- 235000012211 aluminium silicate Nutrition 0.000 claims description 3
- 229910000019 calcium carbonate Inorganic materials 0.000 claims description 3
- 229910052733 gallium Inorganic materials 0.000 claims description 3
- NLYAJNPCOHFWQQ-UHFFFAOYSA-N kaolin Chemical compound O.O.O=[Al]O[Si](=O)O[Si](=O)O[Al]=O NLYAJNPCOHFWQQ-UHFFFAOYSA-N 0.000 claims description 3
- 150000005324 oxide salts Chemical class 0.000 claims description 3
- 229910052723 transition metal Inorganic materials 0.000 claims description 3
- 150000003624 transition metals Chemical class 0.000 claims description 3
- CETPSERCERDGAM-UHFFFAOYSA-N ceric oxide Chemical compound O=[Ce]=O CETPSERCERDGAM-UHFFFAOYSA-N 0.000 claims description 2
- 239000011258 core-shell material Substances 0.000 claims description 2
- 239000002019 doping agent Substances 0.000 claims description 2
- 229910052732 germanium Inorganic materials 0.000 claims description 2
- GNPVGFCGXDBREM-UHFFFAOYSA-N germanium atom Chemical compound [Ge] GNPVGFCGXDBREM-UHFFFAOYSA-N 0.000 claims description 2
- 229910052738 indium Inorganic materials 0.000 claims description 2
- APFVFJFRJDLVQX-UHFFFAOYSA-N indium atom Chemical compound [In] APFVFJFRJDLVQX-UHFFFAOYSA-N 0.000 claims description 2
- 150000003839 salts Chemical class 0.000 claims description 2
- 239000012798 spherical particle Substances 0.000 claims description 2
- 229910001848 post-transition metal Inorganic materials 0.000 claims 10
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 claims 8
- 229910052681 coesite Inorganic materials 0.000 claims 4
- 229910052906 cristobalite Inorganic materials 0.000 claims 4
- 239000000377 silicon dioxide Substances 0.000 claims 4
- 229910052682 stishovite Inorganic materials 0.000 claims 4
- 229910052905 tridymite Inorganic materials 0.000 claims 4
- MCMNRKCIXSYSNV-UHFFFAOYSA-N Zirconium dioxide Chemical compound O=[Zr]=O MCMNRKCIXSYSNV-UHFFFAOYSA-N 0.000 claims 2
- 239000011787 zinc oxide Substances 0.000 claims 2
- 239000004411 aluminium Substances 0.000 claims 1
- PNEYBMLMFCGWSK-UHFFFAOYSA-N aluminium oxide Inorganic materials [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 claims 1
- 229910000422 cerium(IV) oxide Inorganic materials 0.000 claims 1
- 229910052593 corundum Inorganic materials 0.000 claims 1
- 229910001845 yogo sapphire Inorganic materials 0.000 claims 1
- 229910052751 metal Inorganic materials 0.000 description 16
- 239000002184 metal Substances 0.000 description 16
- 238000012360 testing method Methods 0.000 description 16
- 229910010413 TiO 2 Inorganic materials 0.000 description 13
- 238000004519 manufacturing process Methods 0.000 description 11
- 229910004298 SiO 2 Inorganic materials 0.000 description 7
- 230000003287 optical effect Effects 0.000 description 7
- 239000000463 material Substances 0.000 description 6
- 239000004033 plastic Substances 0.000 description 6
- 229920003023 plastic Polymers 0.000 description 6
- 239000000835 fiber Substances 0.000 description 5
- 229920000742 Cotton Polymers 0.000 description 4
- 238000001514 detection method Methods 0.000 description 4
- 239000002250 absorbent Substances 0.000 description 3
- 238000005259 measurement Methods 0.000 description 3
- 150000002739 metals Chemical class 0.000 description 3
- -1 polyethylene terephthalate Polymers 0.000 description 3
- 230000003595 spectral effect Effects 0.000 description 3
- 229910018072 Al 2 O 3 Inorganic materials 0.000 description 2
- 239000004698 Polyethylene Substances 0.000 description 2
- 239000004743 Polypropylene Substances 0.000 description 2
- 230000002745 absorbent Effects 0.000 description 2
- 239000004927 clay Substances 0.000 description 2
- 238000003475 lamination Methods 0.000 description 2
- 230000014759 maintenance of location Effects 0.000 description 2
- 238000012986 modification Methods 0.000 description 2
- 230000004048 modification Effects 0.000 description 2
- 229920000573 polyethylene Polymers 0.000 description 2
- 229920000139 polyethylene terephthalate Polymers 0.000 description 2
- 239000005020 polyethylene terephthalate Substances 0.000 description 2
- 229920001155 polypropylene Polymers 0.000 description 2
- 239000000126 substance Substances 0.000 description 2
- 239000004408 titanium dioxide Substances 0.000 description 2
- GYHNNYVSQQEPJS-UHFFFAOYSA-N Gallium Chemical compound [Ga] GYHNNYVSQQEPJS-UHFFFAOYSA-N 0.000 description 1
- 241000208202 Linaceae Species 0.000 description 1
- 235000004431 Linum usitatissimum Nutrition 0.000 description 1
- 229910006404 SnO 2 Inorganic materials 0.000 description 1
- 238000000862 absorption spectrum Methods 0.000 description 1
- 239000003513 alkali Substances 0.000 description 1
- 229910052783 alkali metal Inorganic materials 0.000 description 1
- 150000001340 alkali metals Chemical class 0.000 description 1
- 229910052784 alkaline earth metal Inorganic materials 0.000 description 1
- 150000001342 alkaline earth metals Chemical class 0.000 description 1
- 229910000410 antimony oxide Inorganic materials 0.000 description 1
- 229910052797 bismuth Inorganic materials 0.000 description 1
- JCXGWMGPZLAOME-UHFFFAOYSA-N bismuth atom Chemical compound [Bi] JCXGWMGPZLAOME-UHFFFAOYSA-N 0.000 description 1
- 230000015556 catabolic process Effects 0.000 description 1
- 239000011248 coating agent Substances 0.000 description 1
- 238000000576 coating method Methods 0.000 description 1
- 238000004040 coloring Methods 0.000 description 1
- 150000001875 compounds Chemical class 0.000 description 1
- 238000006731 degradation reaction Methods 0.000 description 1
- 239000006185 dispersion Substances 0.000 description 1
- 230000001747 exhibiting effect Effects 0.000 description 1
- 238000010348 incorporation Methods 0.000 description 1
- 229910003437 indium oxide Inorganic materials 0.000 description 1
- PJXISJQVUVHSOJ-UHFFFAOYSA-N indium(iii) oxide Chemical compound [O-2].[O-2].[O-2].[In+3].[In+3] PJXISJQVUVHSOJ-UHFFFAOYSA-N 0.000 description 1
- 239000002105 nanoparticle Substances 0.000 description 1
- 229910052699 polonium Inorganic materials 0.000 description 1
- HZEBHPIOVYHPMT-UHFFFAOYSA-N polonium atom Chemical compound [Po] HZEBHPIOVYHPMT-UHFFFAOYSA-N 0.000 description 1
- 239000004417 polycarbonate Substances 0.000 description 1
- 229920000515 polycarbonate Polymers 0.000 description 1
- 239000002243 precursor Substances 0.000 description 1
- 238000003860 storage Methods 0.000 description 1
- 238000006467 substitution reaction Methods 0.000 description 1
- 239000012209 synthetic fiber Substances 0.000 description 1
- 229920002994 synthetic fiber Polymers 0.000 description 1
- 229910052716 thallium Inorganic materials 0.000 description 1
- BKVIYDNLLOSFOA-UHFFFAOYSA-N thallium Chemical compound [Tl] BKVIYDNLLOSFOA-UHFFFAOYSA-N 0.000 description 1
Images
Classifications
-
- D—TEXTILES; PAPER
- D21—PAPER-MAKING; PRODUCTION OF CELLULOSE
- D21H—PULP COMPOSITIONS; PREPARATION THEREOF NOT COVERED BY SUBCLASSES D21C OR D21D; IMPREGNATING OR COATING OF PAPER; TREATMENT OF FINISHED PAPER NOT COVERED BY CLASS B31 OR SUBCLASS D21G; PAPER NOT OTHERWISE PROVIDED FOR
- D21H21/00—Non-fibrous material added to the pulp, characterised by its function, form or properties; Paper-impregnating or coating material, characterised by its function, form or properties
- D21H21/14—Non-fibrous material added to the pulp, characterised by its function, form or properties; Paper-impregnating or coating material, characterised by its function, form or properties characterised by function or properties in or on the paper
- D21H21/40—Agents facilitating proof of genuineness or preventing fraudulent alteration, e.g. for security paper
- D21H21/42—Ribbons or strips
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B42—BOOKBINDING; ALBUMS; FILES; SPECIAL PRINTED MATTER
- B42D—BOOKS; BOOK COVERS; LOOSE LEAVES; PRINTED MATTER CHARACTERISED BY IDENTIFICATION OR SECURITY FEATURES; PRINTED MATTER OF SPECIAL FORMAT OR STYLE NOT OTHERWISE PROVIDED FOR; DEVICES FOR USE THEREWITH AND NOT OTHERWISE PROVIDED FOR; MOVABLE-STRIP WRITING OR READING APPARATUS
- B42D25/00—Information-bearing cards or sheet-like structures characterised by identification or security features; Manufacture thereof
- B42D25/20—Information-bearing cards or sheet-like structures characterised by identification or security features; Manufacture thereof characterised by a particular use or purpose
- B42D25/29—Securities; Bank notes
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B42—BOOKBINDING; ALBUMS; FILES; SPECIAL PRINTED MATTER
- B42D—BOOKS; BOOK COVERS; LOOSE LEAVES; PRINTED MATTER CHARACTERISED BY IDENTIFICATION OR SECURITY FEATURES; PRINTED MATTER OF SPECIAL FORMAT OR STYLE NOT OTHERWISE PROVIDED FOR; DEVICES FOR USE THEREWITH AND NOT OTHERWISE PROVIDED FOR; MOVABLE-STRIP WRITING OR READING APPARATUS
- B42D25/00—Information-bearing cards or sheet-like structures characterised by identification or security features; Manufacture thereof
- B42D25/30—Identification or security features, e.g. for preventing forgery
- B42D25/355—Security threads
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B42—BOOKBINDING; ALBUMS; FILES; SPECIAL PRINTED MATTER
- B42D—BOOKS; BOOK COVERS; LOOSE LEAVES; PRINTED MATTER CHARACTERISED BY IDENTIFICATION OR SECURITY FEATURES; PRINTED MATTER OF SPECIAL FORMAT OR STYLE NOT OTHERWISE PROVIDED FOR; DEVICES FOR USE THEREWITH AND NOT OTHERWISE PROVIDED FOR; MOVABLE-STRIP WRITING OR READING APPARATUS
- B42D25/00—Information-bearing cards or sheet-like structures characterised by identification or security features; Manufacture thereof
- B42D25/30—Identification or security features, e.g. for preventing forgery
- B42D25/36—Identification or security features, e.g. for preventing forgery comprising special materials
- B42D25/373—Metallic materials
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B42—BOOKBINDING; ALBUMS; FILES; SPECIAL PRINTED MATTER
- B42D—BOOKS; BOOK COVERS; LOOSE LEAVES; PRINTED MATTER CHARACTERISED BY IDENTIFICATION OR SECURITY FEATURES; PRINTED MATTER OF SPECIAL FORMAT OR STYLE NOT OTHERWISE PROVIDED FOR; DEVICES FOR USE THEREWITH AND NOT OTHERWISE PROVIDED FOR; MOVABLE-STRIP WRITING OR READING APPARATUS
- B42D25/00—Information-bearing cards or sheet-like structures characterised by identification or security features; Manufacture thereof
- B42D25/30—Identification or security features, e.g. for preventing forgery
- B42D25/36—Identification or security features, e.g. for preventing forgery comprising special materials
- B42D25/378—Special inks
- B42D25/382—Special inks absorbing or reflecting infrared light
-
- D—TEXTILES; PAPER
- D21—PAPER-MAKING; PRODUCTION OF CELLULOSE
- D21H—PULP COMPOSITIONS; PREPARATION THEREOF NOT COVERED BY SUBCLASSES D21C OR D21D; IMPREGNATING OR COATING OF PAPER; TREATMENT OF FINISHED PAPER NOT COVERED BY CLASS B31 OR SUBCLASS D21G; PAPER NOT OTHERWISE PROVIDED FOR
- D21H21/00—Non-fibrous material added to the pulp, characterised by its function, form or properties; Paper-impregnating or coating material, characterised by its function, form or properties
- D21H21/14—Non-fibrous material added to the pulp, characterised by its function, form or properties; Paper-impregnating or coating material, characterised by its function, form or properties characterised by function or properties in or on the paper
- D21H21/40—Agents facilitating proof of genuineness or preventing fraudulent alteration, e.g. for security paper
- D21H21/44—Latent security elements, i.e. detectable or becoming apparent only by use of special verification or tampering devices or methods
-
- D—TEXTILES; PAPER
- D21—PAPER-MAKING; PRODUCTION OF CELLULOSE
- D21H—PULP COMPOSITIONS; PREPARATION THEREOF NOT COVERED BY SUBCLASSES D21C OR D21D; IMPREGNATING OR COATING OF PAPER; TREATMENT OF FINISHED PAPER NOT COVERED BY CLASS B31 OR SUBCLASS D21G; PAPER NOT OTHERWISE PROVIDED FOR
- D21H21/00—Non-fibrous material added to the pulp, characterised by its function, form or properties; Paper-impregnating or coating material, characterised by its function, form or properties
- D21H21/14—Non-fibrous material added to the pulp, characterised by its function, form or properties; Paper-impregnating or coating material, characterised by its function, form or properties characterised by function or properties in or on the paper
- D21H21/40—Agents facilitating proof of genuineness or preventing fraudulent alteration, e.g. for security paper
- D21H21/44—Latent security elements, i.e. detectable or becoming apparent only by use of special verification or tampering devices or methods
- D21H21/48—Elements suited for physical verification, e.g. by irradiation
Definitions
- the present invention relates to security substrates and their manufacturing methods.
- a security substrate is a substrate used in the manufacture of secure documents, protected against counterfeiting by means that make it difficult for them to be reproduced or falsified by counterfeiters.
- the invention applies to paper substrates, for example intended for the manufacture of banknotes, as well as to plastic substrates, used in particular in the manufacture of banknotes, of security elements such as security threads, films or foils.
- the invention is more particularly interested in giving the substrate a specific signature in the infrared (IR), this signature making it possible to authenticate, or even identify, the substrate by comparison with a reference signature.
- IR infrared
- Requirement JP 2010084079 also discloses an IR-absorbent ink based on tin oxide doped with antimony and / or indium oxide doped with tin.
- tin oxide doped with antimony and / or indium oxide doped with tin The use of these substantially colorless materials allows better compatibility with the colored pigments used elsewhere to give the ink the desired color.
- the detection can be carried out by reflection.
- Requirement KR10-1131145 discloses a security paper comprising an IR absorbent substance. This application aims to overcome the coloring problems encountered during the use of certain known substances which absorb in the IR, and proposes materials of white or opaline color based on alkali, alkaline earth or transition metals.
- WO 2012/103578 discloses a security material comprising a transparent polymeric carrier medium having IR absorbing nanoparticles dispersed in the medium.
- WO 2007/132214 discloses a composition comprising an ink and an IR absorbent material.
- WO 00/32700 discloses a papermaking filler or coating derived from a hydrated clay having a metal oxide and / or a precursor of a metal oxide of a poor metal aggregated with the clay.
- the invention aims to further improve the security of the substrates in order in particular to confer a signature, preferably unitary, in the IR detectable by machine in transmission, while preserving the optical properties of whiteness and opacity of the substrate when the latter is a paper maker, and intended for example for the manufacture of banknotes.
- unitary signature should be understood to mean a signature associated in a one-to-one manner with a given substrate; for example the substrate used for the manufacture of all the banknotes corresponding to the same denomination has the same signature. In another example, all the banknotes issued by the same central bank have the same signature, regardless of the value of the denominations.
- the invention achieves this objective by virtue of a paper security substrate according to claim 1.
- the poor metal can be in the ionic (salt), molecular (oxide, in particular SnO 2 ) doped or not, or atomic (dopant, in particular Al or Ga) form.
- the term “poor metal” denotes a metal chosen from the group consisting of aluminum, bismuth, tin, gallium, indium, lead, polonium and thallium, the latter two metals however not being. favorite.
- Said oxide or metal oxide salt is preferably respectively an oxide or an oxide salt of a transition metal or of a metalloid.
- the feed comprises SiO 2 or TiO 2 as metal oxide and Sn as poor metal.
- the size of the particles of the filler is preferably between 0.2 and 5 ⁇ m, preferably being less than 1 ⁇ m and / or greater than 0.5 ⁇ m.
- the shape of the filler particles can be spherical, platelet or needle-like.
- the particles of the filler have a core made of TiO 2 or SiO 2 covered with tin oxide doped with antimony.
- the invention makes it possible, depending on the particulate system used, to have a spectral signature in the unitary IR, readable in transmission over the entire substrate, due to the mass incorporation during its manufacture.
- IR is meant the infrared in the spectral band 700nm - 1500nm.
- the invention also offers the advantage, through detection in transmission, of avoiding jamming the responses of other security elements detected in reflection.
- the detection can be carried out according to the invention in unprinted areas, which offers additional security compared to an ink absorbing in the IR and detected in reflection.
- the invention also makes it possible, if desired, to dispense with the use of certain conventional paper fillers such as titanium dioxide, kaolin or calcium carbonate, or to reduce the content thereof, while at the same time achieving the optical properties of whiteness and opacity usually desired in the manufacture of paper for banknotes in particular.
- the substrate can thus comprise less than 4% by dry weight of TiO 2 in the form of TiO 2 particles, relative to the total dry weight of the substrate, better still being completely devoid of TiO 2 in the form of TiO 2 particles.
- the substrate may comprise less than 4% by dry weight of kaolin or of calcium carbonate relative to the total dry weight of the substrate.
- Another advantage of the present invention is to provide electro-conductivity to the substrate, which can give it a specific electrical signature and help dissipate electrostatic charges in the event of the presence of large areas of plastic material, in particular during storage.
- a paper substrate of at least one plastic element such as a wide security thread, a lamination strip, a foil or a film, in particular in the case of a paper / plastic hybrid ticket.
- filler should be understood to mean a compound introduced in the particulate state into the rest of the substrate, and which remains in the particulate state.
- the particulate system can comprise or consist of a mixture of at least one filler consisting of at least one metal oxide or metal oxide salt, in particular a mica, and at least one lean metal in ionic, molecular form. , or atomic.
- the particulate system can also comprise or be constituted by a filler consisting of at least one metal oxide or metal oxide salt, in particular a mica, and at least one lean metal in ionic, molecular or atomic form.
- a filler of the core-shell type comprising a core consisting of said metal oxide or metal oxide salt and a shell consisting of said poor metal in ionic, molecular or atomic form, preferably in molecular form.
- it is a doped load, said metal oxide or metal oxide salt being doped with said lean metal in atomic form.
- the metal oxide is chosen from TiO 2 , ZnO, ZrO 2 , CeO 2 , SiO 2 , Al 2 O 3 , mica, talc and their mixtures.
- the metal oxide may in particular have a refractive index greater than 1.8 (TiO 2 , ZnO, etc.) or less than 1.8 (SiO 2 , Al 2 O 3 , etc.) allowing for example to have a colorless filler which does not modify the optical properties of a papermaking substrate.
- the doped poor metal metal oxide is doped with metalloids or poor metals, in particular with antimony, aluminum or germanium. Depending on the poor metals and metalloids chosen, the infrared absorption can be modified and thus obtain a different spectral response, to make it specific to each substrate if desired.
- the poor metal can be in the form of tin oxide.
- the doped poor metal metal oxide, on the one hand, and the metal oxide or the metal oxide salt, in particular a mica, on the other hand, can respectively constitute the shell and the core of a pigment to core bark structure.
- the heart can be hollow.
- the particulate system may consist of particles with a maximum size less than or equal to 100 ⁇ m, preferably less than 60 ⁇ m.
- the particulate system may consist of particles of number-average size D50 less than or equal to 10 ⁇ m, preferably 5 ⁇ m.
- the particulate system may consist of particles of number-average size D50 less than or equal to 1 ⁇ m, preferably 0.5 ⁇ m.
- the particulate system may consist of particles with a maximum size less than or equal to 5 ⁇ m, preferably less than 1 ⁇ m.
- the particulate system may consist of particles with a minimum size greater than or equal to 0.2 ⁇ m, preferably 0.5 ⁇ m.
- the particulate system can comprise and / or be made up of spherical particles.
- the particulate system can comprise and / or be made up of needle-like and / or platelet particles, which offers the advantage of increasing the specific surface.
- the particulate system may comprise a single doped metal oxide or, alternatively, several doped metal oxides.
- the particulate system can be substantially white and opaque to visible light, or alternatively be transparent.
- the substrate preferably comprises between 0.1 and 20% by mass, relative to the total mass of the finished dry substrate, of said particulate system, better between 0.2 and 5, even better between 0.5 and 2.5% .
- the amount of said particulate system is adjusted so as not to darken the substrate unduly, giving it a grayish appearance.
- the substrate can be paper.
- the substrate may in particular be suitable for the manufacture of banknotes.
- the substrate can be watermarked.
- the substrate can also be non-fibrous.
- the substrate when a papermaker, can comprise natural and / or synthetic fibers. In the case in particular of a substrate for banknotes, the latter may comprise flax fibers and / or cotton fibers, in particular cotton linters.
- the substrate can be made of plastic, that is to say polymeric material, in particular of polyethylene terephthalate (PET), polycarbonate (PC), polypropylene (PP) or polyethylene (PE).
- PET polyethylene terephthalate
- PC polycarbonate
- PP polypropylene
- PE polyethylene
- the plastic can be extruded or co-extruded with the particulate system.
- a further subject of the invention is a security document comprising a substrate according to the invention, in particular a bank note.
- a subject of the invention is also a thread, a film, a lamination strip or a security foil comprising a substrate according to the invention.
- Paper samples are made using a paper machine from a fiber composition 100% cotton linters.
- the particulate system for samples according to the invention is introduced into the fiber dispersion 100% cotton linters before papermaking.
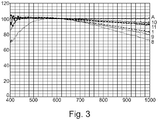
- IR absorption measurements (percentage absorption rate) are carried out in transmission between 400 nm and 1000 nm. The absorption spectra are then normalized (on the x-axis) at a wavelength of 500 nm to analyze the behavior of the pigment in IR.
- the tests show that the pigment used during test 1 exhibits a discriminating absorption from 2% by mass of the pigment. Pigments other than that of test 1, for example of different shape and size, such as those of tests 2, 3 and 4, also exhibit a discriminating response in the IR from a mass content of 2% .
- the pigments of tests 2 and 3 have a heart of titanium dioxide (TiO 2 ), covered with tin oxide doped with antimony.
- the pigments of tests 1 and 4 show a more intense response in the IR because they have a particle size greater than that of tests 2 and 3, which facilitates their retention within the structure of the paper during the manufacture of the paper. -this.
- the pigment of Test 4 exhibits a particle size which influences optical properties such as whiteness and opacity much more.
- Tests A and B demonstrate that the increase in weight does not significantly modify the IR absorption.
- the level of pigment used in test 7 makes it possible to have a very strong absorption in the IR but that this leads to marked modifications of the optical properties such as whiteness and opacity.
- the IR response of the pigments of tests 5 and 6 has a different appearance from the other tests, which makes it possible to distinguish the IR signatures from one another.
- the two pigments reduce the optical properties such as whiteness and opacity, which can be compensated for by adding conventional paper fillers.
- the pigments of tests 8 and 9 make it possible to have a very strong absorption in the IR but this causes marked modifications of the optical properties such as whiteness and opacity.
- the pigments of tests 10 and 11 have a low contribution to IR absorption because their small particle size ( ⁇ 0.2 ⁇ m) does not allow optimal retention within the paper during its manufacture.
- TO 1 2 3 4 Whiteness % 88.5 81.1 80.61 85.03 77.03 Opacity % 86.30 88.68 89.32 87.29 87.94 TO B 5 6 7 Whiteness % 88.5 89.28 83.67 82.64 75.8 Opacity % 86.30 86.34 80.81 82.18 93.63 TO 1 8 9 10 11 Whiteness % 88.5 81.1 72.8 77.8 86 85.5 Opacity % 86.30 88.68 89.16 87.7 77.7 79.3
- mixtures of fillers according to the invention can be used.
Description
La présente invention concerne les substrats de sécurité et leurs procédés de fabrication.The present invention relates to security substrates and their manufacturing methods.
Un substrat de sécurité est un substrat utilisé dans la fabrication de documents sécurisés, protégés contre la contrefaçon par des moyens rendant difficile leur reproduction ou leur falsification par des contrefacteurs.A security substrate is a substrate used in the manufacture of secure documents, protected against counterfeiting by means that make it difficult for them to be reproduced or falsified by counterfeiters.
L'invention s'applique à des substrats papetiers, destinés par exemple à la fabrication de billets de banque, ainsi qu'à des substrats en matière plastique, utilisés notamment dans la fabrication de billets de banque, d'éléments de sécurité tels que des fils de sécurité, des films ou des foils.The invention applies to paper substrates, for example intended for the manufacture of banknotes, as well as to plastic substrates, used in particular in the manufacture of banknotes, of security elements such as security threads, films or foils.
L'invention s'intéresse plus particulièrement à conférer au substrat une signature spécifique dans l'infrarouge (IR), cette signature permettant d'authentifier, voire d'identifier, le substrat par comparaison à une signature de référence.The invention is more particularly interested in giving the substrate a specific signature in the infrared (IR), this signature making it possible to authenticate, or even identify, the substrate by comparison with a reference signature.
Il est déjà bien connu d'utiliser des moyens de sécurité présentant une réponse spécifique dans l'IR.It is already well known to use security means exhibiting a specific response in the IR.
En particulier, il a été proposé dans la demande de brevet
La demande
La demande
La demande
La demande
L'invention vise à améliorer encore la sécurisation des substrats afin notamment de conférer une signature, de préférence unitaire, dans l'IR détectable par machine en transmission, tout en préservant les propriétés optiques de blancheur et d'opacité du substrat lorsque celui-ci est papetier, et destiné par exemple à la fabrication de billets de banque.The invention aims to further improve the security of the substrates in order in particular to confer a signature, preferably unitary, in the IR detectable by machine in transmission, while preserving the optical properties of whiteness and opacity of the substrate when the latter is a paper maker, and intended for example for the manufacture of banknotes.
Par « signature unitaire », il faut comprendre une signature associée de façon bijective à un substrat donné ; par exemple le substrat utilisé pour la fabrication de tous les billets correspondant à une même coupure présente la même signature. Dans un autre exemple, tous les billets issus par une même banque centrale présentent la même signature, indépendamment de la valeur des coupures.The term “unitary signature” should be understood to mean a signature associated in a one-to-one manner with a given substrate; for example the substrate used for the manufacture of all the banknotes corresponding to the same denomination has the same signature. In another example, all the banknotes issued by the same central bank have the same signature, regardless of the value of the denominations.
L'invention atteint cet objectif grâce à un substrat de sécurité papetier selon la revendication 1The invention achieves this objective by virtue of a paper security substrate according to
Le métal pauvre peut être sous la forme ionique (sel), moléculaire (oxyde, notamment SnO2) dopée ou non, ou atomique (dopant, notamment Al ou Ga). Par "métal pauvre", on désigne un métal choisi dans le groupe constitué par l'aluminium, le bismuth, l'étain, le gallium, l'indium, le plomb, le polonium et le thallium, ces deux derniers métaux étant toutefois non préférés.The poor metal can be in the ionic (salt), molecular (oxide, in particular SnO 2 ) doped or not, or atomic (dopant, in particular Al or Ga) form. The term “poor metal” denotes a metal chosen from the group consisting of aluminum, bismuth, tin, gallium, indium, lead, polonium and thallium, the latter two metals however not being. favorite.
Ledit oxyde ou sel d'oxyde métallique est de préférence respectivement un oxyde ou un sel d'oxyde d'un métal de transition ou d'un métalloïde.Said oxide or metal oxide salt is preferably respectively an oxide or an oxide salt of a transition metal or of a metalloid.
De façon préférée, la charge comporte comme oxyde métallique SiO2 ou TiO2 et comme métal pauvre Sn. La taille des particules de la charge est de préférence comprise entre 0,2 et 5 µm, étant de préférence inférieure à 1 µm et/ou supérieure à 0,5 µm. La forme des particules de la charge peut être sphérique, plaquettaire ou aciculaire.Preferably, the feed comprises SiO 2 or TiO 2 as metal oxide and Sn as poor metal. The size of the particles of the filler is preferably between 0.2 and 5 μm, preferably being less than 1 μm and / or greater than 0.5 μm. The shape of the filler particles can be spherical, platelet or needle-like.
De manière préférée, les particules de la charge ont un cœur en TiO2 ou SiO2 recouvert d'oxyde d'étain dopé à l'antimoine.Preferably, the particles of the filler have a core made of TiO 2 or SiO 2 covered with tin oxide doped with antimony.
L'invention permet, selon le système particulaire utilisé, d'avoir une signature spectrale dans l'IR unitaire, lisible en transmission sur la totalité du substrat, du fait de l'incorporation en masse durant sa fabrication. Par "IR", on désigne l'infra-rouge dans la bande spectrale 700nm - 1500nm. L'invention offre aussi l'avantage, de par la détection en transmission, d'éviter de brouiller les réponses d'autres éléments de sécurité détectés en réflexion. De plus, la détection peut s'effectuer selon l'invention dans des zones non imprimées, ce qui offre une sécurité complémentaire par rapport à une encre absorbant dans l'IR et détectée en réflexion.The invention makes it possible, depending on the particulate system used, to have a spectral signature in the unitary IR, readable in transmission over the entire substrate, due to the mass incorporation during its manufacture. By "IR" is meant the infrared in the spectral band 700nm - 1500nm. The invention also offers the advantage, through detection in transmission, of avoiding jamming the responses of other security elements detected in reflection. In addition, the detection can be carried out according to the invention in unprinted areas, which offers additional security compared to an ink absorbing in the IR and detected in reflection.
L'invention permet en outre, si on le souhaite, de s'affranchir de l'utilisation de certaines charges papetières conventionnelles telles que le dioxyde de titane, le kaolin ou le carbonate de calcium, ou d'en réduire la teneur, tout en atteignant les propriétés optiques de blancheur et d'opacité habituellement recherchées dans la fabrication d'un papier pour billet de banque notamment. Le substrat peut ainsi comporter moins de 4% en poids sec de TiO2 sous forme de particules de TiO2, par rapport au poids sec total du substrat, mieux étant totalement dépourvu de TiO2 sous forme de particules de TiO2. Le substrat peut comporter moins de 4% en poids sec de kaolin ou de carbonate de calcium par rapport au poids sec total du substrat.The invention also makes it possible, if desired, to dispense with the use of certain conventional paper fillers such as titanium dioxide, kaolin or calcium carbonate, or to reduce the content thereof, while at the same time achieving the optical properties of whiteness and opacity usually desired in the manufacture of paper for banknotes in particular. The substrate can thus comprise less than 4% by dry weight of TiO 2 in the form of TiO 2 particles, relative to the total dry weight of the substrate, better still being completely devoid of TiO 2 in the form of TiO 2 particles. The substrate may comprise less than 4% by dry weight of kaolin or of calcium carbonate relative to the total dry weight of the substrate.
Un autre avantage de la présente invention est d'apporter de l'électro-conductivité au substrat, qui peut lui conférer une signature électrique spécifique et contribuer à dissiper les charges électrostatiques en cas de présence de zones étendues en matière plastique, notamment lors de l'utilisation conjointe à un substrat papetier d'au moins un élément en matière plastique tel qu'un fil de sécurité large, une bande de lamination, un foil ou un film, notamment en cas de billet hybride papier/plastique.Another advantage of the present invention is to provide electro-conductivity to the substrate, which can give it a specific electrical signature and help dissipate electrostatic charges in the event of the presence of large areas of plastic material, in particular during storage. 'joint use with a paper substrate of at least one plastic element such as a wide security thread, a lamination strip, a foil or a film, in particular in the case of a paper / plastic hybrid ticket.
Par « charge » il faut comprendre un composé introduit à l'état particulaire dans le reste du substrat, et qui demeure à l'état particulaire.The term “filler” should be understood to mean a compound introduced in the particulate state into the rest of the substrate, and which remains in the particulate state.
Le système particulaire peut comprendre ou être constitué d'un mélange d'au moins une charge constituée d'au moins un oxyde métallique ou sel d'oxyde métallique, notamment un mica, et d'au moins un métal pauvre sous forme ionique, moléculaire, ou atomique.The particulate system can comprise or consist of a mixture of at least one filler consisting of at least one metal oxide or metal oxide salt, in particular a mica, and at least one lean metal in ionic, molecular form. , or atomic.
Le système particulaire peut encore comprendre ou être constitué par une charge constituée d'au moins un oxyde métallique ou sel d'oxyde métallique, notamment un mica, et d'au moins un métal pauvre sous forme ionique, moléculaire, ou atomique.The particulate system can also comprise or be constituted by a filler consisting of at least one metal oxide or metal oxide salt, in particular a mica, and at least one lean metal in ionic, molecular or atomic form.
Il s'agit en particulier d'une charge de type cœur-écorce (« core-shell » en anglais) comprenant un cœur constitué dudit oxyde métallique ou sel d'oxyde métallique et d'une écorce constituée dudit métal pauvre sous forme ionique, moléculaire ou atomique, de préférence sous forme moléculaire.It is in particular a filler of the core-shell type comprising a core consisting of said metal oxide or metal oxide salt and a shell consisting of said poor metal in ionic, molecular or atomic form, preferably in molecular form.
Selon une variante il s'agit d'une charge dopée, ledit oxyde métallique ou sel d'oxyde métallique étant dopé par ledit métal pauvre sous forme atomique.According to one variant, it is a doped load, said metal oxide or metal oxide salt being doped with said lean metal in atomic form.
L'oxyde métallique est choisi parmi TiO2, ZnO, ZrO2, CeO2, SiO2, Al2O3, le mica, le talc et leurs mélanges. L'oxyde métallique peut notamment présenter un indice de réfraction supérieur à 1,8 (TiO2, ZnO, ... ) ou inférieur à 1,8 (SiO2,Al2O3, ...) permettant par exemple d'avoir une charge incolore ne modifiant pas les propriétés optiques d'un substrat papetier.The metal oxide is chosen from TiO 2 , ZnO, ZrO 2 , CeO 2 , SiO 2 , Al 2 O 3 , mica, talc and their mixtures. The metal oxide may in particular have a refractive index greater than 1.8 (TiO 2 , ZnO, etc.) or less than 1.8 (SiO 2 , Al 2 O 3 , etc.) allowing for example to have a colorless filler which does not modify the optical properties of a papermaking substrate.
L'oxyde métallique de métal pauvre dopé est dopé avec des métalloïdes ou des métaux pauvres, notamment avec de l'antimoine, de l'aluminium ou du germanium. Selon les métaux pauvres et métalloïdes choisis, on peut modifier l'absorption infra-rouge et ainsi obtenir une réponse spectrale différente, pour la rendre spécifique à chaque substrat si on le souhaite.The doped poor metal metal oxide is doped with metalloids or poor metals, in particular with antimony, aluminum or germanium. Depending on the poor metals and metalloids chosen, the infrared absorption can be modified and thus obtain a different spectral response, to make it specific to each substrate if desired.
Le métal pauvre peut être sous forme d'oxyde d'étain.The poor metal can be in the form of tin oxide.
L'oxyde métallique de métal pauvre dopé, d'une part, et l'oxyde métallique ou le sel d'oxyde métallique, notamment un mica, d'autre part, peuvent constituer respectivement l'écorce et le cœur d'un pigment à structure cœur écorce. Le cœur peut être creux.The doped poor metal metal oxide, on the one hand, and the metal oxide or the metal oxide salt, in particular a mica, on the other hand, can respectively constitute the shell and the core of a pigment to core bark structure. The heart can be hollow.
Le système particulaire peut être constitué de particules de taille maximale inférieure ou égale à 100 µm, de préférence inférieure à 60 µm.The particulate system may consist of particles with a maximum size less than or equal to 100 μm, preferably less than 60 μm.
Le système particulaire peut être constitué de particules de taille moyenne en nombre D50 inférieure ou égale à 10 µm, de préférence à 5 µm.The particulate system may consist of particles of number-average size D50 less than or equal to 10 μm, preferably 5 μm.
Le système particulaire peut être constitué de particules de taille moyenne en nombre D50 inférieure ou égale à 1 µm, de préférence à 0,5 µm.The particulate system may consist of particles of number-average size D50 less than or equal to 1 μm, preferably 0.5 μm.
Le système particulaire peut être constitué de particules de taille maximale inférieure ou égale à 5 µm, de préférence inférieure à 1 µm. Le système particulaire peut être constitué de particules de taille minimale supérieure ou égale à 0,2 µm, de préférence à 0,5 µm.The particulate system may consist of particles with a maximum size less than or equal to 5 μm, preferably less than 1 μm. The particulate system may consist of particles with a minimum size greater than or equal to 0.2 μm, preferably 0.5 μm.
Le système particulaire peut comporter et/ou être constitué de particules sphériques. En variante, le système particulaire peut comporter et/ou être constitué de particules aciculaires et/ou plaquettaires, ce qui offre l'avantage d'accroître la surface spécifique.The particulate system can comprise and / or be made up of spherical particles. As a variant, the particulate system can comprise and / or be made up of needle-like and / or platelet particles, which offers the advantage of increasing the specific surface.
Le système particulaire peut comporter un seul oxyde métallique dopé ou en variante plusieurs oxydes métalliques dopés.The particulate system may comprise a single doped metal oxide or, alternatively, several doped metal oxides.
Le système particulaire peut être sensiblement blanc et opaque à la lumière visible, ou en variante être transparent.The particulate system can be substantially white and opaque to visible light, or alternatively be transparent.
Le substrat comporte de préférence entre 0,1 et 20 % en masse, par rapport à la masse totale du substrat fini à sec, dudit système particulaire, mieux entre 0,2 et 5, encore mieux entre 0,5 et 2,5%.The substrate preferably comprises between 0.1 and 20% by mass, relative to the total mass of the finished dry substrate, of said particulate system, better between 0.2 and 5, even better between 0.5 and 2.5% .
De préférence, la quantité dudit système particulaire est ajustée pour ne pas assombrir outre mesure le substrat en lui donnant un aspect grisâtre.Preferably, the amount of said particulate system is adjusted so as not to darken the substrate unduly, giving it a grayish appearance.
Le substrat peut être papetier. Le substrat peut notamment convenir à la fabrication de billets de banque. Le substrat peut être filigrané. Le substrat peut encore être non fibreux. Le substrat, lorsque papetier, peut comporter des fibres naturelles et/ou synthétiques. Dans le cas notamment d'un substrat pour billets de banque, celui-ci peut comporter des fibres de lin et/ou des fibres de coton, notamment des linters de coton.The substrate can be paper. The substrate may in particular be suitable for the manufacture of banknotes. The substrate can be watermarked. The substrate can also be non-fibrous. The substrate, when a papermaker, can comprise natural and / or synthetic fibers. In the case in particular of a substrate for banknotes, the latter may comprise flax fibers and / or cotton fibers, in particular cotton linters.
Le substrat peut être en matière plastique, c'est-à-dire polymérique, notamment en polyéthylène téréphtalate (PET), polycarbonate (PC), polypropylène (PP) ou polyéthylène (PE). La matière plastique peut être extrudée ou co-extrudée avec le système particulaire.The substrate can be made of plastic, that is to say polymeric material, in particular of polyethylene terephthalate (PET), polycarbonate (PC), polypropylene (PP) or polyethylene (PE). The plastic can be extruded or co-extruded with the particulate system.
L'invention a encore pour objet un document de sécurité comportant un substrat selon l'invention, notamment un billet de banque.A further subject of the invention is a security document comprising a substrate according to the invention, in particular a bank note.
L'invention a encore pour objet un fil, un film, une bande de lamination ou un foil de sécurité comportant un substrat selon l'invention.A subject of the invention is also a thread, a film, a lamination strip or a security foil comprising a substrate according to the invention.
Des échantillons de papier sont réalisés à l'aide d'une machine à papier à partir d'une composition fibreuse 100 % linters de coton.
Le système particulaire pour les échantillons selon l'invention est introduit dans la dispersion fibreuse 100% linters de coton avant la fabrication du papier.Paper samples are made using a paper machine from a
The particulate system for samples according to the invention is introduced into the
Des mesures d'absorption IR (taux d'absorption en pourcentage) sont effectuées en transmission entre 400 nm et 1000 nm. Les spectres d'absorption sont ensuite normalisés (en abscisses) à une longueur d'onde de 500 nm pour analyser le comportement du pigment dans l'IR.IR absorption measurements (percentage absorption rate) are carried out in transmission between 400 nm and 1000 nm. The absorption spectra are then normalized (on the x-axis) at a wavelength of 500 nm to analyze the behavior of the pigment in IR.
Les mesures d'opacité et de blancheur ont été réalisées selon respectivement les normes ISO 2471-1 et ISO 2470-2Opacity and whiteness measurements were carried out according to ISO 2471-1 and ISO 2470-2 respectively
Sur les graphiques : en ordonnée on a le pourcentage d'absorption et en abscisse la longueur d'onde.On the graphs: on the ordinate we have the absorption percentage and on the abscissa the wavelength.
Les essais montrent que le pigment utilisé lors de l'essai 1 présente une absorption discriminante à partir de 2% en masse du pigment. Des pigments autres que celui de l'essai 1, par exemple de forme et de taille différentes, tels que ceux des essais 2,3 et 4, présentent aussi une réponse discriminante dans l'IR à partir d'une teneur massique de 2%. Les pigments des essais 2 et 3 présentent un cœur en dioxyde de titane (TiO2), recouvert d'oxyde d'étain dopé à l'antimoine.The tests show that the pigment used during
Les pigments des essais 1 et 4 présentent une réponse dans l'IR plus intense car ils ont une taille de particule supérieure à celle des essais 2 et 3, ce qui facilite leur rétention au sein de la structure du papier lors de la fabrication de celui-ci.The pigments of
Des mesures dont les résultats sont reportés dans les tableaux ci-dessous ont permis de constater que les pigments des essais 1,2,3 ont pu être incorporés dans le papier en substitution d'une charge de référence de 5% de dioxyde de titane, sans dégradation outre mesure de la blancheur, qui reste acceptable, et en conservant l'opacité, voire en l'améliorant malgré le fait que la charge introduite ne présente plus que 2,5% ou 2% au lieu de 5%.Measurements, the results of which are reported in the tables below, have shown that the pigments of
Le pigment de l'essai 4 présente une taille de particule qui influence beaucoup plus les propriétés optiques telles que la blancheur et l'opacité.The pigment of Test 4 exhibits a particle size which influences optical properties such as whiteness and opacity much more.
Les essais A et B mettent en évidence que l'augmentation de grammage ne modifie pas de façon significative l'absorption IR.Tests A and B demonstrate that the increase in weight does not significantly modify the IR absorption.
On voit également que le taux de pigment utilisé dans l'essai 7 permet d'avoir une très forte absorption dans l'IR mais que cela entraine des modifications marquées des propriétés optiques telles que la blancheur et l'opacité.It can also be seen that the level of pigment used in
Par ailleurs, la réponse IR des pigments des essais 5 et 6 a une allure différente des autres essais, ce qui permet de discriminer les signatures IR entre elles. Enfin les deux pigments diminuent les propriétés optiques telles que la blancheur et l'opacité ce qui pourra être compensé par l'ajout de charges papetières conventionnelles.Furthermore, the IR response of the pigments of tests 5 and 6 has a different appearance from the other tests, which makes it possible to distinguish the IR signatures from one another. Finally, the two pigments reduce the optical properties such as whiteness and opacity, which can be compensated for by adding conventional paper fillers.
On voit que les pigments des essais 8 et 9 permettent d'avoir une très forte absorption dans l'IR mais cela entraine des modifications marquées des propriétés optiques telles que la blancheur et l'opacité.It can be seen that the pigments of
Les pigments des essais 10 et 11 ont une faible contribution sur l'absorption IR car leur faible taille de particules (< 0.2µm) ne permet pas d'avoir une rétention optimale au sein du papier lors de sa fabrication.
L'invention n'est pas limitée aux exemples qui viennent d'être donnés.The invention is not limited to the examples which have just been given.
En particulier, des mélanges de charges selon l'invention peuvent être utilisés.In particular, mixtures of fillers according to the invention can be used.
Claims (15)
- Paper security substrate comprising, dispersed in the bulk thereof, preferably between 0.1% and 20% by weight, relative to the total weight of the substrate, better still between 0.2% and 5%, even better still between 0.5% and 2.5%, of an IR-absorbing particulate system, the particulate system comprising:- a mixture of at least one filler consisting of at least one metal oxide or metal oxide salt, in particular a mica, and of at least one filler consisting of at least one post-transition metal in ionic, molecular or atomic form, and/or- at least one filler consisting of at least one metal oxide or metal oxide salt, in particular a mica, and of at least one post-transition metal in ionic, molecular or atomic form,said particulate system giving said substrate a specific response in transmission in the IR, preferably with at least one absorption peak in the IR, in particular between 700 and 1000 nm, better still with several absorption peaks in the IR, in particular between 700 and 1000 nm,
preferably, the particulate system comprising and/or consisting of spherical particles or the particulate system comprising and/or consisting of acicular and/or flakelike particles,
the metal oxide being chosen from TiO2, ZnO, ZrO2, CeO2, SiO2, Al2O3, mica, talc, and mixtures thereof, the post-transition metal being in the form of metal oxide of post-transition metal doped with metalloids or post-transition metals. - Substrate according to Claim 1, the post-transition metal being in ionic (salt), doped or undoped molecular (oxide, in particular SnO2), or atomic (dopant, in particular Al or Ga) form, preferably said metal oxide or metal oxide salt respectively being an oxide or an oxide salt of a transition metal or of a metalloid.
- Substrate according to either one of the preceding claims, the metal oxide being chosen from TiO2, SiO2, ZnO, mica or talc, better still TiO2 or SiO2.
- Substrate according to any one of the preceding claims, the metal oxide of post-transition metal that is doped being doped with antimony, or the metal oxide of post-transition metal that is doped being doped with aluminium or germanium.
- Substrate according to any one of the preceding claims, the metal oxide of post-transition metal that is doped being an oxide of tin or of indium, preferably of tin.
- Substrate according to any one of the preceding claims, the filler consisting of particles having an SiO2 or TiO2 core covered with antimony-doped tin oxide.
- Substrate according to any one of the preceding claims, the post-transition metal on the one hand and the metal oxide or the metal oxide salt, in particular a mica, on the other hand respectively constituting the shell and the core of a pigment with core-shell structure, preferably the core being hollow.
- Substrate according to any one of the preceding claims, comprising less than 4% by dry weight of TiO2 in the form of TiO2 particles, relative to the total dry weight of the substrate, better still being completely free of TiO2 in the form of TiO2 particles.
- Substrate according to any one of the preceding claims, comprising less than 4% by dry weight of kaolin or of calcium carbonate relative to the total dry weight of the substrate.
- Substrate according to any one of the preceding claims, the particulate system consisting of particles having a number-average size D50 of less than or equal to 10 µm, preferably less than 5 µm.
- Substrate according to any one of Claims 1 to 8, the particulate system consisting of particles having a number-average size D50 of less than or equal to 1 µm, preferably less than 0.5 µm.
- Substrate according to any one of the preceding claims, the particulate system comprising a single doped metal oxide or the particulate system comprising several doped metal oxides.
- Substrate according to any one of the preceding claims, the particulate system being substantially white and opaque to visible light or the particulate system being transparent.
- Security document comprising a substrate according to any one of the preceding claims, the substrate in particular being watermarked.
- Security thread, film or foil comprising a substrate according to any one of Claims 1 to 13.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| FR1660177A FR3057881B1 (en) | 2016-10-20 | 2016-10-20 | SECURITY SUBSTRATE |
| PCT/EP2017/076637 WO2018073320A1 (en) | 2016-10-20 | 2017-10-18 | Security substrate |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP3529416A1 EP3529416A1 (en) | 2019-08-28 |
| EP3529416B1 true EP3529416B1 (en) | 2021-08-04 |
Family
ID=57750207
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP17791021.3A Active EP3529416B1 (en) | 2016-10-20 | 2017-10-18 | Security substrate |
Country Status (3)
| Country | Link |
|---|---|
| EP (1) | EP3529416B1 (en) |
| FR (1) | FR3057881B1 (en) |
| WO (1) | WO2018073320A1 (en) |
Family Cites Families (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| AU1926600A (en) * | 1998-12-02 | 2000-06-19 | Engelhard Corporation | High opacity kaolin hybrid pigments |
| RS50766B2 (en) | 2005-11-25 | 2018-01-31 | Sicpa Holding Sa | Ir-absorbing intaglio ink |
| GB2438196B (en) * | 2006-05-13 | 2008-05-28 | Inovink Ltd | Improvements in or relating to printing |
| JP5489031B2 (en) | 2008-10-01 | 2014-05-14 | 共同印刷株式会社 | Infrared absorbing ink and printed matter for forgery prevention |
| KR101131145B1 (en) | 2010-02-02 | 2012-04-03 | 한국조폐공사 | Infrared Absorption Paper for counterfeit prevention |
| MX365504B (en) * | 2011-02-01 | 2019-06-05 | Australia Reserve Bank | Transparent infrared absorbing materials. |
-
2016
- 2016-10-20 FR FR1660177A patent/FR3057881B1/en not_active Expired - Fee Related
-
2017
- 2017-10-18 WO PCT/EP2017/076637 patent/WO2018073320A1/en unknown
- 2017-10-18 EP EP17791021.3A patent/EP3529416B1/en active Active
Non-Patent Citations (1)
| Title |
|---|
| None * |
Also Published As
| Publication number | Publication date |
|---|---|
| FR3057881A1 (en) | 2018-04-27 |
| EP3529416A1 (en) | 2019-08-28 |
| WO2018073320A1 (en) | 2018-04-26 |
| FR3057881B1 (en) | 2020-06-12 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP2984229B1 (en) | Security element comprising an interference pigment and a nanometric filler | |
| WO2013093848A1 (en) | Multilayer structure comprising at least one diffusing layer and method for manufacturing same | |
| EP2722172A1 (en) | Security sheet having a coextruded substrate | |
| FR3086202A1 (en) | Safety documents and safety devices including infrared absorbing compositions | |
| FR3014017A1 (en) | SECURITY STRUCTURE | |
| EP3060719B1 (en) | Paper especially for printing an electroconductive layer | |
| EP3529416B1 (en) | Security substrate | |
| EP3083258B1 (en) | Security item | |
| CA2729624A1 (en) | Security element having a variable optical effect and security sheet or document or article comprising it | |
| EP1825055B1 (en) | Glossy paper | |
| EP2984227B1 (en) | Security element comprising a masking structure containing a mixture of nanometric fillers | |
| FR2992255A1 (en) | SECURITY ELEMENT AND SECURE DOCUMENT. | |
| EP3194178A1 (en) | Security element | |
| CA2757155A1 (en) | Printable matt fine paper and method of preparing same | |
| EP3206885B1 (en) | Security element for a security document | |
| EP3023259B1 (en) | Security element | |
| EP3206884A1 (en) | Security element and security document | |
| EP3558692B1 (en) | Secure document | |
| WO2014002014A2 (en) | Sheet element capable of being engraved by a laser | |
| FR2978580A1 (en) | METHOD FOR CONTROLLING THE PHYSICAL STATE OF A DOCUMENT | |
| WO2013042061A1 (en) | Sheet-like medium for laser marking | |
| WO2015181686A1 (en) | Substrate for secure document | |
| FR2925534A1 (en) | SAFETY STRUCTURE COMPRISING NEMATIC LIQUID CRYSTALS |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: UNKNOWN |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: THE INTERNATIONAL PUBLICATION HAS BEEN MADE |
|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: REQUEST FOR EXAMINATION WAS MADE |
|
| 17P | Request for examination filed |
Effective date: 20190401 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): AL AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL PT RO RS SE SI SK SM TR |
|
| AX | Request for extension of the european patent |
Extension state: BA ME |
|
| DAV | Request for validation of the european patent (deleted) | ||
| DAX | Request for extension of the european patent (deleted) | ||
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: EXAMINATION IS IN PROGRESS |
|
| 17Q | First examination report despatched |
Effective date: 20200430 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: EXAMINATION IS IN PROGRESS |
|
| GRAP | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOSNIGR1 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: GRANT OF PATENT IS INTENDED |
|
| INTG | Intention to grant announced |
Effective date: 20210309 |
|
| GRAS | Grant fee paid |
Free format text: ORIGINAL CODE: EPIDOSNIGR3 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: THE PATENT HAS BEEN GRANTED |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AL AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL PT RO RS SE SI SK SM TR |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: FG4D Free format text: NOT ENGLISH |
|
| REG | Reference to a national code |
Ref country code: AT Ref legal event code: REF Ref document number: 1417110 Country of ref document: AT Kind code of ref document: T Effective date: 20210815 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: EP |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R096 Ref document number: 602017043451 Country of ref document: DE |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FG4D Free format text: LANGUAGE OF EP DOCUMENT: FRENCH |
|
| REG | Reference to a national code |
Ref country code: LT Ref legal event code: MG9D |
|
| REG | Reference to a national code |
Ref country code: NL Ref legal event code: MP Effective date: 20210804 |
|
| REG | Reference to a national code |
Ref country code: AT Ref legal event code: MK05 Ref document number: 1417110 Country of ref document: AT Kind code of ref document: T Effective date: 20210804 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20210804 Ref country code: BG Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20211104 Ref country code: AT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20210804 Ref country code: FI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20210804 Ref country code: ES Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20210804 Ref country code: NO Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20211104 Ref country code: PT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20211206 Ref country code: SE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20210804 Ref country code: RS Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20210804 Ref country code: HR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20210804 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: PL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20210804 Ref country code: LV Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20210804 Ref country code: GR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20211105 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: NL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20210804 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20210804 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R119 Ref document number: 602017043451 Country of ref document: DE |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SM Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20210804 Ref country code: SK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20210804 Ref country code: RO Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20210804 Ref country code: EE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20210804 Ref country code: CZ Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20210804 Ref country code: AL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20210804 |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| REG | Reference to a national code |
Ref country code: BE Ref legal event code: MM Effective date: 20211031 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: MC Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20210804 |
|
| 26N | No opposition filed |
Effective date: 20220506 |
|
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 20211104 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LU Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20211018 Ref country code: IT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20210804 Ref country code: DE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20220503 Ref country code: BE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20211031 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20210804 Ref country code: LI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20211031 Ref country code: CH Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20211031 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: FR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20211031 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20211018 Ref country code: GB Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20211104 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: CY Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20210804 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: HU Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT; INVALID AB INITIO Effective date: 20171018 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: MK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20210804 |