EP3363888A1 - Use of liquid laundry detergent composition to minimize adverse effects upon accidental exposure to contents of water-soluble unit dose articles - Google Patents
Use of liquid laundry detergent composition to minimize adverse effects upon accidental exposure to contents of water-soluble unit dose articles Download PDFInfo
- Publication number
- EP3363888A1 EP3363888A1 EP18157942.6A EP18157942A EP3363888A1 EP 3363888 A1 EP3363888 A1 EP 3363888A1 EP 18157942 A EP18157942 A EP 18157942A EP 3363888 A1 EP3363888 A1 EP 3363888A1
- Authority
- EP
- European Patent Office
- Prior art keywords
- detergent composition
- laundry detergent
- liquid laundry
- water
- unit dose
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Withdrawn
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D17/00—Detergent materials or soaps characterised by their shape or physical properties
- C11D17/04—Detergent materials or soaps characterised by their shape or physical properties combined with or containing other objects
- C11D17/041—Compositions releasably affixed on a substrate or incorporated into a dispensing means
- C11D17/042—Water soluble or water disintegrable containers or substrates containing cleaning compositions or additives for cleaning compositions
- C11D17/043—Liquid or thixotropic (gel) compositions
Definitions
- the present invention relates to the use of a liquid laundry detergent composition to minimize the adverse effects of rare accidental exposure of the detergent composition comprised with a liquid laundry detergent composition.
- Laundry water-soluble unit dose articles are liked by consumers as they are efficient and convenient to use. They offer simple unit dosing of laundry detergent composition to be used in the laundry wash operation removing the need for inconvenient and messy dosing.
- a liquid laundry detergent composition according to the present invention comprised within a water-soluble unit dose article provided a desired reduction in adverse effects from rare accidental exposures to the contents of the water-soluble unit dose article whilst providing laundry cleaning, especially greasy food stain cleaning, and freshness benefits and still maintaining a good whiteness profile.
- the present invention discloses the use of a liquid laundry detergent composition comprised in a water-soluble unit dose article to provide laundry cleaning benefits and to minimize potential adverse effects associated with accidental exposure to said liquid laundry detergent composition contained within the water-soluble unit dose article, wherein the liquid laundry detergent composition comprises between 10% and 35%, or even more preferably between 12% and 35%, most preferably between 15% and 30% or even between 15% and 25% by weight of the liquid laundry detergent composition of a surfactant.
- the present invention is to the use of a liquid laundry detergent composition comprised in a water-soluble unit dose article to provide laundry cleaning benefits and to minimize potential adverse effects associated with accidental exposure to said liquid laundry detergent composition contained within the water-soluble unit dose article.
- water-soluble unit dose articles contain a single dose of a laundry detergent composition enveloped in a water-soluble film.
- the consumer places the entire article into the drum of a washing machine or other wash receptacle together with fabrics to be washed.
- the water-soluble film dissolves releasing the laundry detergent composition contained within into the water and into contact with the fabrics.
- the concept is that the laundry detergent composition remains contained within the unit dose article until the unit dose article is contacted with water during use.
- Irritation is herein defined as the production of changes to the body following the application of test chemical to the anterior surface of the body, which are fully reversible within 21 days of application.
- eye irritation is defined as the production of changes in the eye following the application of test chemical to the anterior surface of the eye, which are fully reversible within 21 days of application.
- the term is interchangeable with "Reversible effects on the Eye” and with "UN GHS Category 2" (4), and can be assessed following the OECD Guidelines for Testing of Chemicals, Test No. 438: Isolated Chicken Eye Test Method for Identifying i) Chemicals Inducing Serious Eye Damage and ii) Chemicals Not Requiring Classification for Eye Irritation or Serious Eye Damage (Adopted version : 26 July 2013).
- the liquid laundry detergent composition minimizes potential adverse effects associated with accidental exposure to said liquid laundry detergent composition contained within the water-soluble unit dose article by minimizing potential adverse effects of the water-soluble unit dose article, preferably reducing skin irritation, eye irritation or a mixture thereof, most preferably eye irritation, of the water-soluble unit dose article.
- the liquid laundry detergent composition comprises between 10% and 35%, or even more preferably between 12% and 35%, most preferably between 15% and 30% or even between 15% and 25% by weight of the liquid laundry detergent composition of a surfactant.
- the liquid laundry detergent composition and surfactant and described in more detail below.
- the potential adverse effects associated with accidental exposure to said liquid laundry detergent composition contained within the water-soluble unit dose article is for example when the water-soluble unit dose article is accidentally prematurely ruptured and the skin, eye or mixture thereof of the user are contacted with the liquid laundry detergent composition.
- the water-soluble unit dose article comprises the water-soluble film shaped such that the unit-dose article comprises at least one internal compartment surrounded by the water-soluble film.
- the unit dose article may comprise a first water-soluble film and a second water-soluble film sealed to one another such to define the internal compartment.
- the water-soluble unit dose article is constructed such that the liquid laundry detergent composition does not leak out of the compartment during storage. However, upon addition of the water-soluble unit dose article to water, the water-soluble film dissolves and releases the contents of the internal compartment into the wash liquor.
- the compartment should be understood as meaning a closed internal space within the unit dose article, which holds the liquid laundry detergent composition.
- a first water-soluble film may be shaped to comprise an open compartment into which the liquid laundry detergent composition is added.
- a second water-soluble film is then laid over the first film in such an orientation as to close the opening of the compartment. The first and second films are then sealed together along a seal region.
- the water-soluble film is described in more detail below.
- the unit dose article may comprise more than one compartment, even at least two compartments, or even at least three compartments.
- the compartments may be arranged in superposed orientation, i.e. one positioned on top of the other. In such an orientation the unit dose article will comprise three films, top, middle and bottom.
- the compartments may be positioned in a side-by-side orientation, i.e. one orientated next to the other.
- the compartments may even be orientated in a 'tyre and rim' arrangement, i.e. a first compartment is positioned next to a second compartment, but the first compartment at least partially surrounds the second compartment, but does not completely enclose the second compartment.
- one compartment may be completely enclosed within another compartment.
- the unit dose article comprises at least two compartments, one of the compartments may be smaller than the other compartment.
- the unit dose article comprises at least three compartments, two of the compartments may be smaller than the third compartment, and preferably the smaller compartments are superposed on the larger compartment.
- the superposed compartments preferably are orientated side-by-side.
- the liquid laundry detergent composition according to the present invention may be comprised in at least one of the compartments. It may for example be comprised in just one compartment, or may be comprised in two compartments, or even in three compartments.
- Each compartment may comprise the same or different compositions.
- the different compositions could all be in the same form, or they may be in different forms.
- the water-soluble unit dose article may comprise at least two internal compartments, wherein the liquid laundry detergent composition is comprised in at least one of the compartments, preferably wherein the unit dose article comprises at least three compartments, wherein the detergent composition is comprised in at least one of the compartments.
- the water-soluble unit dose article comprises at least two compartments and the liquid laundry detergent composition is comprised in at least one compartment, preferably wherein the water-soluble unit dose article comprises at least two compartments of different sizes and the liquid laundry detergent composition is comprised in at least the larger compartment.
- the film of the present invention is soluble or dispersible in water.
- the water-soluble film preferably has a thickness of from 20 to 150 micron, preferably 35 to 125 micron, even more preferably 50 to 110 micron, most preferably about 76 micron.
- the film has a water-solubility of at least 50%, preferably at least 75% or even at least 95%, as measured by the method set out here after using a glass-filter with a maximum pore size of 20 microns:
- Preferred film materials are preferably polymeric materials.
- the film material can, for example, be obtained by casting, blow-moulding, extrusion or blown extrusion of the polymeric material, as known in the art.
- Preferred polymers, copolymers or derivatives thereof suitable for use as pouch material are selected from polyvinyl alcohols, polyvinyl pyrrolidone, polyalkylene oxides, acrylamide, acrylic acid, cellulose, cellulose ethers, cellulose esters, cellulose amides, polyvinyl acetates, polycarboxylic acids and salts, polyaminoacids or peptides, polyamides, polyacrylamide, copolymers of maleic/acrylic acids, polysaccharides including starch and gelatine, natural gums such as xanthum and carragum.
- More preferred polymers are selected from polyacrylates and water-soluble acrylate copolymers, methylcellulose, carboxymethylcellulose sodium, dextrin, ethylcellulose, hydroxyethyl cellulose, hydroxypropyl methylcellulose, maltodextrin, polymethacrylates, and most preferably selected from polyvinyl alcohols, polyvinyl alcohol copolymers and hydroxypropyl methyl cellulose (HPMC), and combinations thereof.
- the level of polymer in the pouch material for example a PVA polymer, is at least 60%.
- the polymer can have any weight average molecular weight, preferably from about 1000 to 1,000,000, more preferably from about 10,000 to 300,000 yet more preferably from about 20,000 to 150,000.
- polymers and/or copolymers can also be used as the pouch material, especially mixtures of polyvinylalcohol polymers and/or copolymers, especially mixtures of polyvinylalcohol homopolymers and/or anionic polyvinylalcohol copolymers preferably selected from sulphonated and carboxylated anionic polyvinylalcohol copolymers especially carboxylated anionic polyvinylalcohol copolymers.
- the water soluble film comprises a blend of a polyvinylalcohol homopolymer and a carboxylated anionic polyvinylalcohol copolymer.
- Preferred films exhibit good dissolution in cold water, meaning unheated distilled water.
- Preferably such films exhibit good dissolution at temperatures of 24°C, even more preferably at 10°C.
- good dissolution it is meant that the film exhibits water-solubility of at least 50%, preferably at least 75% or even at least 95%, as measured by the method set out here after using a glass-filter with a maximum pore size of 20 microns, described above.
- Preferred films are those supplied by Monosol under the trade references M8630, M8900, M8779, M8310.
- the film may be opaque, transparent or translucent.
- the film may comprise a printed area.
- the area of print may be achieved using standard techniques, such as flexographic printing or inkjet printing.
- the film may comprise an aversive agent, for example a bittering agent.
- Suitable bittering agents include, but are not limited to, naringin, sucrose octaacetate, quinine hydrochloride, denatonium benzoate, or mixtures thereof.
- Any suitable level of aversive agent may be used in the film. Suitable levels include, but are not limited to, 1 to 5000ppm, or even 100 to 2500ppm, or even 250 to 2000rpm.
- the water-soluble unit dose article comprises a water-soluble film, preferably wherein the water-soluble film comprises polyvinyl alcohol, preferably wherein the water-soluble film comprises polyvinyl alcohol polymer or copolymer, preferably a blend of polyvinylalcohol polymers and/or polyvinylalcohol copolymers, more preferably selected from sulphonated and carboxylated anionic polyvinylalcohol copolymers especially carboxylated anionic polyvinylalcohol copolymers, most preferably a blend of a polyvinylalcohol homopolymer and a carboxylated anionic polyvinylalcohol copolymer.
- liquid laundry detergent composition refers to any laundry detergent composition comprising a liquid capable of wetting and treating a fabric, and includes, but is not limited to, liquids, gels, pastes, dispersions and the like.
- the liquid composition can include solids or gases in suitably subdivided form, but the liquid composition excludes forms which are non-fluid overall, such as tablets or granules.
- the liquid laundry detergent composition comprises between 10% and 35%, or even more preferably between 12% and 35%, most preferably between 15% and 30% or even between 15% and 25% by weight of the liquid laundry detergent composition of a surfactant.
- the surfactant preferably comprises a fatty alcohol ethoxylate non-ionic surfactant, wherein the liquid laundry detergent composition preferably comprises less than 10%, preferably between 0% and 9.5%, preferably between 0.01% and 9%, more preferably between 0.1% and 7%, even more preferably between 1% and 5%, most preferably between 1 and 3% by weight of the liquid laundry detergent composition of the fatty alcohol ethoxylate non-ionic surfactant.
- Suitable alcohol ethoxylate nonionic surfactants include the condensation products of aliphatic alcohols with ethylene oxide.
- the alkyl chain of the aliphatic alcohol can either be straight or branched, substituted or unsubstituted.
- the starting alcohol can be naturally derived, e.g. starting from natural oils, or synthetically derived, e.g. alcohols obtained from for example oxo-, modified oxo- or Fischer-Tropsch processes.
- oxo-process derived alcohols include the Lial and Isalchem alcohols ex Sasol company and Lutensol alcohols ex BASF company.
- modified-oxo process derived alcohols include the Neodol alcohols ex Shell company.
- Fischer-Tropsch derived alcohols include Safol alcohols ex Sasol company.
- the alkoxylate chain of alcohol ethoxylates is made up solely of ethoxylate groups.
- the fatty alcohol ethoxylate has an average alkyl carbon chain length of between 5 and 30, preferably between 8 and 18, more preferably between 10 and 16, most preferably between 12 and 15.
- the fatty alcohol ethoxylate has an average degree of ethoxylation of between 0.5 and 20, preferably between 1 and 15, more preferably between 5 and 12, even more preferably between 6 and 10, most preferably between 7 and 8.
- Suitable for use herein are the ethoxylated alcohol of the formula R(OC 2 H 4 )n OH, wherein R is selected from the group consisting of aliphatic hydrocarbon radicals containing from about 8 to about 22 carbon atoms and the average value of n is from about 5 to about 22.
- particularly useful materials are condensation products of C 9 -C 16 alcohols with from about 5 to about 20 moles of ethylene oxide per mole of alcohol.
- particularly useful materials are condensation products of C 12 -C 16 alcohols with from about 6 to about 9 moles of ethylene oxide per mole of alcohol.
- nonionic surfactants may include: C12-C18 alkyl ethoxylates based on modified oxo alcohols, such as, NEODOL® nonionic surfactants from Shell; C12-C15 alkyl ethoxylates based on Fischer Tropsch Oxo alcohols, such as, SAFOL® nonionic surfactants from Sasol; C12-C18 alkyl ethoxylates based on natural or Ziegler alcohols, such as, Surfonic® nonionic surfactants from Huntsman; C14-C22 mid-chain branched alcohols ethoxylates, BAE x , wherein x is from 1 to 30.
- modified oxo alcohols such as, NEODOL® nonionic surfactants from Shell
- C12-C15 alkyl ethoxylates based on Fischer Tropsch Oxo alcohols such as, SAFOL® nonionic surfactants from Sasol
- the surfactant comprises a non-soap anionic surfactant, preferably between 10% and 35%, or even more preferably between 12% and 35%, most preferably between 15% and 30% or even more preferably between 15% and 25% by weight of the liquid laundry detergent composition of the non-soap anionic surfactant.
- the non-soap anionic surfactant may comprise a sulphate or a sulphonate anionic surfactant or a mixture thereof, preferably linear alkylbenzene sulphonate, alkyl sulphate, alkoxylated alkyl sulphate or a mixture thereof, more preferably a mixture of linear alkylbenzene sulphonate and alkoxylated alkyl sulphate.
- the ratio of linear alkylbenzene sulphonate to alkoxylated alkyl sulphate more preferably the ratio of linear alkylbenzene sulphonate to ethoxylated alkyl sulphate is from 1:2 to 20:1, preferably from 1.1:1 to 15:1, more preferably from 1.2:1 to 10:1, even more preferably from 1.3:1 to 5:1, most preferably from 1.4:1 to 3:1.
- the alkoxylated alkyl sulphate is an ethoxylated alkyl sulphate with an average degree of ethoxylation of between 0.5 and 7, preferably between 1 and 5, more preferably between 2 and 4, most preferably about 3.
- the non-soap surfactant comprises a mixture of one or more alkoxylated alkyl sulphates, preferably ethoxylated alkyl sulphates, and optionally an alkyl sulphate, the mixture having an average degree of ethoxylation of between 0.5 and 7, preferably between 1 and 5, more preferably between 2 and 4, most preferably about 3.
- the alkyl sulphate and/or alkoxylated alkyl sulphate preferably have an alkyl chain comprising on average between 8 and 18 carbon atoms, preferably between 10 and 16 carbons atoms, most preferably between 12 and 14 carbon atoms.
- the alkoxylated alkyl sulphate is an ethoxylated alkyl chain comprising on average between 12 and 14 carbon atoms in its alkyl chain and has an average degree of ethoxylation of about 3.
- the alkyl chain of the alkoxylated alkyl sulphate surfactant may be linear or branched or a mixture thereof.
- the linear alkylbenzene sulphonate may be a C 10 -C 16 linear alkylbenzene sulphonate or a C 11 -C 14 linear alkylbenzene sulphonate or a mixture thereof.
- Exemplary linear alkylbenzene sulphonates are C 10 -C 16 alkyl benzene sulfonic acids, or C 11 -C 14 alkyl benzene sulfonic acids.
- 'linear' we herein mean the alkyl group is linear.
- Alkyl benzene sulfonates are well known in the art.
- the weight ratio of non-soap anionic surfactant to fatty alcohol ethoxylate non-ionic surfactant is between 5:1 and 23:1 preferably between 7:1 and 23:1, more preferably between 8:1 and 23:1, most preferably between 9:1 and 20:1.
- the surfactant may comprise a fatty acid, neutralised fatty acid soap or a mixture thereof.
- the liquid laundry detergent composition may comprise less than 10%, preferably less than 8%, more preferably less than 5%, most preferably between 1% and 5% by weight of the liquid laundry detergent composition of fatty acid, neutralised fatty acid soap or a mixture thereof.
- the neutralised fatty acid soap may be alkali metal neutralised, amine neutralised or a mixture thereof.
- the alkali metal may be selected from sodium, potassium, magnesium or a mixture thereof, preferably sodium.
- the amine is preferably an alkanolamine, preferably selected from monoethanolamine, diethanolamine, triethanolamine or a mixture thereof, more preferably monoethanolamine.
- the fatty acid, neutralised fatty acid soap or mixture thereof may be selected from palm kernel fatty acid, coconut fatty acid, rapeseed fatty acid, neutralized palm kernel fatty acid, neutralized coconut fatty acid, neutralized rapeseed fatty acid, or mixture thereof, preferably neutralized palm kernel fatty acid.
- the liquid laundry detergent composition may comprise between 0.1% and 10%, preferably between 0.5% and 8%, more preferably between 1% and 7%, even more preferably between 2% and 6%, most preferably between 3% and 5% by weight of the liquid laundry detergent composition of a soil release polymer, wherein the soil release polymer is preferably selected from polyester terephthalates, polyethylene glycol containing soil release polymers and a mixture thereof.
- An example of a preferred polyethyleneglycol containing soil release polymer comprises a polyethylene glycol graft polymer comprising a polyethylene glycol backbone (Pluriol E6000) and hydrophobic vinyl acetate side chains, comprising 40% by weight of the polymer system of a polyethylene glycol backbone polymer and 60% by weight of the polymer system of the grafted vinyl acetate side chains.
- Polyester terephtalate soil release polymers are commercially available from Clariant under the Texcare SRN and SRA tradenames.
- One particularly preferred polyester terephtalate soil release polymer is Texcare SRA300.
- the liquid laundry detergent composition may comprise a polyethyleneimine, preferably an alkoxylated polyethyleneimine, more preferably an ethoxylated polyethyleneimine and wherein preferably the liquid laundry detergent composition comprises greater than 0.5%, preferably between 1% and 10%, more preferably between 2% and 7%, most preferably between 3% and 5% by weight of the liquid laundry detergent composition of the polyethyleneimine, preferably ethoxylated polyethyleneimine.
- An example of an ethoxylated polyethyleneimine includes an ethoxylated polyethyleneimine with a polyethyleneimine backbone with a molecular weight of about 600, and comprising on average about 20 ethoxy units per ethoxylation chain.
- the ratio of polyethyleneimine to soil release polymer is higher than 1:1, preferably between 1.1:1 and 5:1, more preferably between 1.2:1 and 3:1, most preferably between 1.3:1 and 2:1.
- the liquid laundry detergent composition may comprise a polysaccharide polymer, a modified polysaccharide polymer, a polysaccharide derived polymer, or a mixture thereof, preferably a cellulosic polymer, a modified cellulosic polymer or a mixture thereof, most preferably a cationically modified cellulosic polymer or a mixture thereof.
- the polysaccharide polymer is a cationically modified hydroethylcellulose ("Polyquaternium 10).
- the water-soluble unit dose article comprises up to 60% preferably up to 50%, more preferably up to 40% by weight of the liquid laundry detergent composition of a non-aqueous solvent, preferably selected from propylene glycol, dipropylene glycol, glycerol, sorbitol, ethanol or a mixture thereof.
- a non-aqueous solvent preferably selected from propylene glycol, dipropylene glycol, glycerol, sorbitol, ethanol or a mixture thereof.
- the liquid laundry detergent composition comprises less than 2%, preferably less than 1% by weight of the liquid laundry detergent composition of aminocarboxylate chelants, aminophosphonate chelants or a mixture thereof.
- the liquid laundry detergent composition has a pH from 6 to 10 preferably from 7 to 9, more preferably from 7 to 8.
- the liquid laundry detergent composition comprises a pH adjusting agent selected from alkanolamines, preferably monethanolamine, diethanolamine, triethanolamine or a mixture thereof, most preferably monoethanolamine.
- the liquid laundry detergent composition comprises an adjunct ingredient selected from hueing dyes, polymers, builders, dye transfer inhibiting agents, dispersants, enzymes, enzyme stabilizers, catalytic materials, bleach, bleach activators, polymeric dispersing agents, anti-redeposition agents, suds suppressors, aesthetic dyes, opacifiers, perfumes, perfume delivery systems, structurants, hydrotropes, processing aids, pigments, amphoteric surfactants, cyclic diamines, zwitterionic polyamines, anti-oxidants, preservatives and mixtures thereof.
- an adjunct ingredient selected from hueing dyes, polymers, builders, dye transfer inhibiting agents, dispersants, enzymes, enzyme stabilizers, catalytic materials, bleach, bleach activators, polymeric dispersing agents, anti-redeposition agents, suds suppressors, aesthetic dyes, opacifiers, perfumes, perfume delivery systems, structurants, hydrotropes, processing aids, pigments, amphoteric surfactants, cyclic di
- the water-soluble unit dose article comprises 15% or less by weight of the unit dose article of water, preferably the unit dose article comprises between 0.1% and 15%, more preferably between 1% and 12.5% by weight of the unit dose article of water.
- a first water-soluble film may be shaped to comprise an open compartment into which the detergent composition is added.
- a second water-soluble film is then laid over the first film in such an orientation as to close the opening of the compartment.
- the first and second films are then sealed together along a seal region using known sealing means such as solvent, heat or a mixture thereof.
- Test formulations were assessed for irritancy profile following the OECD Guidelines for Testing of Chemicals, Test No. 438: Isolated Chicken Eye Test Method for Identifying i) Chemicals Inducing Serious Eye Damage and ii) Chemicals Not Requiring Classification for Eye Irritation or Serious Eye Damage (Adopted version : 26 July 2013).
- Irritation index CS + 20 * CO + 20 * FR in which CS equals the mean % of corneal swelling, CO equals the maximum mean opacity score and FR equals the mean fluorescein retention score at 30 minutes after treatment.
- a higher irritation index reflects a worse irritancy profile.
- the below Figures 1 to 4 have been extracted from the resulting irritancy formulation model.
- the AES data reflect the irritancy index of an AES comprising paste (67.5% AES + 20% DTPK fatty acid + water/minors).
- Active surfactant content reflects the combined % level by weight of the detergent composition of HLAS, AES, FA and NI surfactant. Values in the X-axis reflect weight fraction of the specific compound relative to the total detergent formulation.
Landscapes
- Chemical & Material Sciences (AREA)
- Dispersion Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Wood Science & Technology (AREA)
- Organic Chemistry (AREA)
- Detergent Compositions (AREA)
Abstract
The use of a liquid laundry detergent composition to minimize the adverse effects of rare accidental exposure of the detergent composition contained within a water-soluble unit dose article.
Description
- The present invention relates to the use of a liquid laundry detergent composition to minimize the adverse effects of rare accidental exposure of the detergent composition comprised with a liquid laundry detergent composition.
- Laundry water-soluble unit dose articles are liked by consumers as they are efficient and convenient to use. They offer simple unit dosing of laundry detergent composition to be used in the laundry wash operation removing the need for inconvenient and messy dosing.
- All laundry detergent compositions have to meet stringent regulatory requirements and industry standards in order to be placed on the market. However, detergent products, due to their primary function of cleaning, all have intrinsic hazards and are labeled as such. On rare occasions accidental misuse or accidental rupture may occur resulting in the potential for consumers to be exposed to their contents. Given the nature of the cleaning compositions contained within the unit dose article, in a minority of instances adverse effects may be associated with these exposures including, but not limited to, eye irritation. While such unit dose products already meet existing safety standards and regulatory expectation, there is a desire within the industry to further reduce the severity or potential of adverse effects associated with these rare accidental exposures, whilst still providing cleaning performance and other benefits.
- It is known from the prior art, e.g.
WO2016155993 , that addition of soil release polymers can reduce the irritation of liquid laundry compositions. However, this approach requires addition of materials and due to the compacted nature of water-soluble unit dose articles, there is limited space for the addition of further ingredients or increasing the overall levels of ingredients. - Therefore, there is a need to provide alternative means to minimize the adverse effects of rare accidental exposure of the detergent composition, including reducing the irritation profile whilst still providing laundry cleaning benefits, especially greasy food stain cleaning, whiteness and freshness benefits. It is especially preferred to find ways to reduce the adverse effect from rare accidental exposure to the contents without the addition of further ingredients.
- It was surprisingly found that a liquid laundry detergent composition according to the present invention comprised within a water-soluble unit dose article provided a desired reduction in adverse effects from rare accidental exposures to the contents of the water-soluble unit dose article whilst providing laundry cleaning, especially greasy food stain cleaning, and freshness benefits and still maintaining a good whiteness profile.
- The present invention discloses the use of a liquid laundry detergent composition comprised in a water-soluble unit dose article to provide laundry cleaning benefits and to minimize potential adverse effects associated with accidental exposure to said liquid laundry detergent composition contained within the water-soluble unit dose article,
wherein the liquid laundry detergent composition comprises between 10% and 35%, or even more preferably between 12% and 35%, most preferably between 15% and 30% or even between 15% and 25% by weight of the liquid laundry detergent composition of a surfactant. -
-
FIG. 1 : Eye irritancy profile of a 30% active surfactant composition with HLAS as main anionic surfactant -
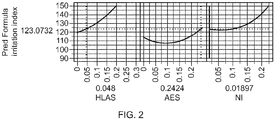
FIG. 2 : Eye irritancy profile of a 30% active surfactant composition with AES as main anionic surfactant -
FIG. 3 : Eye irritancy profile of a 40% active surfactant composition with HLAS as main anionic surfactant -
FIG. 4 : Eye irritancy profile of a 40% active surfactant composition with AES as main anionic surfactant - The present invention is to the use of a liquid laundry detergent composition comprised in a water-soluble unit dose article to provide laundry cleaning benefits and to minimize potential adverse effects associated with accidental exposure to said liquid laundry detergent composition contained within the water-soluble unit dose article.
- The water-soluble unit dose article is described in more detail below. Without wishing to be bound by theory, water-soluble unit dose articles contain a single dose of a laundry detergent composition enveloped in a water-soluble film. The consumer places the entire article into the drum of a washing machine or other wash receptacle together with fabrics to be washed. Upon application of water, the water-soluble film dissolves releasing the laundry detergent composition contained within into the water and into contact with the fabrics. The concept is that the laundry detergent composition remains contained within the unit dose article until the unit dose article is contacted with water during use.
- By 'accidental exposure' we herein mean unintentional exposure, for example from premature rupture of the unit dose article. Such accidental premature rupture is rare, but given the nature of the cleaning compositions contained within the unit dose article, in a minority of instances adverse effects may be associated with these exposures including, but not limited to, eye irritation.
- "Irritation" is herein defined as the production of changes to the body following the application of test chemical to the anterior surface of the body, which are fully reversible within 21 days of application. In particular eye irritation is defined as the production of changes in the eye following the application of test chemical to the anterior surface of the eye, which are fully reversible within 21 days of application. The term is interchangeable with "Reversible effects on the Eye" and with "UN GHS Category 2" (4), and can be assessed following the OECD Guidelines for Testing of Chemicals, Test No. 438: Isolated Chicken Eye Test Method for Identifying i) Chemicals Inducing Serious Eye Damage and ii) Chemicals Not Requiring Classification for Eye Irritation or Serious Eye Damage (Adopted version : 26 July 2013).
- Preferably, the liquid laundry detergent composition minimizes potential adverse effects associated with accidental exposure to said liquid laundry detergent composition contained within the water-soluble unit dose article by minimizing potential adverse effects of the water-soluble unit dose article, preferably reducing skin irritation, eye irritation or a mixture thereof, most preferably eye irritation, of the water-soluble unit dose article.
- The liquid laundry detergent composition comprises between 10% and 35%, or even more preferably between 12% and 35%, most preferably between 15% and 30% or even between 15% and 25% by weight of the liquid laundry detergent composition of a surfactant. The liquid laundry detergent composition and surfactant and described in more detail below.
- Without wishing to be bound by theory it is the specific use of the present liquid laundry detergent composition in a water-soluble unit dose article that minimises the adverse effects of accidental exposure to said laundry detergent composition.
- The potential adverse effects associated with accidental exposure to said liquid laundry detergent composition contained within the water-soluble unit dose article is for example when the water-soluble unit dose article is accidentally prematurely ruptured and the skin, eye or mixture thereof of the user are contacted with the liquid laundry detergent composition.
- The water-soluble unit dose article comprises the water-soluble film shaped such that the unit-dose article comprises at least one internal compartment surrounded by the water-soluble film. The unit dose article may comprise a first water-soluble film and a second water-soluble film sealed to one another such to define the internal compartment. The water-soluble unit dose article is constructed such that the liquid laundry detergent composition does not leak out of the compartment during storage. However, upon addition of the water-soluble unit dose article to water, the water-soluble film dissolves and releases the contents of the internal compartment into the wash liquor.
- The compartment should be understood as meaning a closed internal space within the unit dose article, which holds the liquid laundry detergent composition. During manufacture, a first water-soluble film may be shaped to comprise an open compartment into which the liquid laundry detergent composition is added. A second water-soluble film is then laid over the first film in such an orientation as to close the opening of the compartment. The first and second films are then sealed together along a seal region.
- The water-soluble film is described in more detail below.
- The unit dose article may comprise more than one compartment, even at least two compartments, or even at least three compartments. The compartments may be arranged in superposed orientation, i.e. one positioned on top of the other. In such an orientation the unit dose article will comprise three films, top, middle and bottom. Alternatively, the compartments may be positioned in a side-by-side orientation, i.e. one orientated next to the other. The compartments may even be orientated in a 'tyre and rim' arrangement, i.e. a first compartment is positioned next to a second compartment, but the first compartment at least partially surrounds the second compartment, but does not completely enclose the second compartment. Alternatively one compartment may be completely enclosed within another compartment.
- Wherein the unit dose article comprises at least two compartments, one of the compartments may be smaller than the other compartment. Wherein the unit dose article comprises at least three compartments, two of the compartments may be smaller than the third compartment, and preferably the smaller compartments are superposed on the larger compartment. The superposed compartments preferably are orientated side-by-side.
- In a multi-compartment orientation, the liquid laundry detergent composition according to the present invention may be comprised in at least one of the compartments. It may for example be comprised in just one compartment, or may be comprised in two compartments, or even in three compartments.
- Each compartment may comprise the same or different compositions. The different compositions could all be in the same form, or they may be in different forms.
- The water-soluble unit dose article may comprise at least two internal compartments, wherein the liquid laundry detergent composition is comprised in at least one of the compartments, preferably wherein the unit dose article comprises at least three compartments, wherein the detergent composition is comprised in at least one of the compartments.
- Preferably, the water-soluble unit dose article comprises at least two compartments and the liquid laundry detergent composition is comprised in at least one compartment, preferably wherein the water-soluble unit dose article comprises at least two compartments of different sizes and the liquid laundry detergent composition is comprised in at least the larger compartment.
- The film of the present invention is soluble or dispersible in water. The water-soluble film preferably has a thickness of from 20 to 150 micron, preferably 35 to 125 micron, even more preferably 50 to 110 micron, most preferably about 76 micron.
- Preferably, the film has a water-solubility of at least 50%, preferably at least 75% or even at least 95%, as measured by the method set out here after using a glass-filter with a maximum pore size of 20 microns:
- 5 grams ± 0.1 gram of film material is added in a pre-weighed 3L beaker and 2L ± 5ml of distilled water is added. This is stirred vigorously on a magnetic stirrer, Labline model No. 1250 or equivalent and 5 cm magnetic stirrer, set at 600 rpm, for 30 minutes at 30°C. Then, the mixture is filtered through a folded qualitative sintered-glass filter with a pore size as defined above (max. 20 micron). The water is dried off from the collected filtrate by any conventional method, and the weight of the remaining material is determined (which is the dissolved or dispersed fraction). Then, the percentage solubility or dispersability can be calculated.
- Preferred film materials are preferably polymeric materials. The film material can, for example, be obtained by casting, blow-moulding, extrusion or blown extrusion of the polymeric material, as known in the art.
- Preferred polymers, copolymers or derivatives thereof suitable for use as pouch material are selected from polyvinyl alcohols, polyvinyl pyrrolidone, polyalkylene oxides, acrylamide, acrylic acid, cellulose, cellulose ethers, cellulose esters, cellulose amides, polyvinyl acetates, polycarboxylic acids and salts, polyaminoacids or peptides, polyamides, polyacrylamide, copolymers of maleic/acrylic acids, polysaccharides including starch and gelatine, natural gums such as xanthum and carragum. More preferred polymers are selected from polyacrylates and water-soluble acrylate copolymers, methylcellulose, carboxymethylcellulose sodium, dextrin, ethylcellulose, hydroxyethyl cellulose, hydroxypropyl methylcellulose, maltodextrin, polymethacrylates, and most preferably selected from polyvinyl alcohols, polyvinyl alcohol copolymers and hydroxypropyl methyl cellulose (HPMC), and combinations thereof. Preferably, the level of polymer in the pouch material, for example a PVA polymer, is at least 60%. The polymer can have any weight average molecular weight, preferably from about 1000 to 1,000,000, more preferably from about 10,000 to 300,000 yet more preferably from about 20,000 to 150,000.
- Mixtures of polymers and/or copolymers can also be used as the pouch material, especially mixtures of polyvinylalcohol polymers and/or copolymers, especially mixtures of polyvinylalcohol homopolymers and/or anionic polyvinylalcohol copolymers preferably selected from sulphonated and carboxylated anionic polyvinylalcohol copolymers especially carboxylated anionic polyvinylalcohol copolymers. Most preferably the water soluble film comprises a blend of a polyvinylalcohol homopolymer and a carboxylated anionic polyvinylalcohol copolymer.
- Preferred films exhibit good dissolution in cold water, meaning unheated distilled water. Preferably such films exhibit good dissolution at temperatures of 24°C, even more preferably at 10°C. By good dissolution it is meant that the film exhibits water-solubility of at least 50%, preferably at least 75% or even at least 95%, as measured by the method set out here after using a glass-filter with a maximum pore size of 20 microns, described above.
- Preferred films are those supplied by Monosol under the trade references M8630, M8900, M8779, M8310.
- The film may be opaque, transparent or translucent. The film may comprise a printed area.
- The area of print may be achieved using standard techniques, such as flexographic printing or inkjet printing.
- The film may comprise an aversive agent, for example a bittering agent. Suitable bittering agents include, but are not limited to, naringin, sucrose octaacetate, quinine hydrochloride, denatonium benzoate, or mixtures thereof. Any suitable level of aversive agent may be used in the film. Suitable levels include, but are not limited to, 1 to 5000ppm, or even 100 to 2500ppm, or even 250 to 2000rpm.
- Preferably, the water-soluble unit dose article comprises a water-soluble film, preferably wherein the water-soluble film comprises polyvinyl alcohol, preferably wherein the water-soluble film comprises polyvinyl alcohol polymer or copolymer, preferably a blend of polyvinylalcohol polymers and/or polyvinylalcohol copolymers, more preferably selected from sulphonated and carboxylated anionic polyvinylalcohol copolymers especially carboxylated anionic polyvinylalcohol copolymers, most preferably a blend of a polyvinylalcohol homopolymer and a carboxylated anionic polyvinylalcohol copolymer.
- The term 'liquid laundry detergent composition' refers to any laundry detergent composition comprising a liquid capable of wetting and treating a fabric, and includes, but is not limited to, liquids, gels, pastes, dispersions and the like. The liquid composition can include solids or gases in suitably subdivided form, but the liquid composition excludes forms which are non-fluid overall, such as tablets or granules.
- The liquid laundry detergent composition comprises between 10% and 35%, or even more preferably between 12% and 35%, most preferably between 15% and 30% or even between 15% and 25% by weight of the liquid laundry detergent composition of a surfactant.
- The surfactant preferably comprises a fatty alcohol ethoxylate non-ionic surfactant, wherein the liquid laundry detergent composition preferably comprises less than 10%, preferably between 0% and 9.5%, preferably between 0.01% and 9%, more preferably between 0.1% and 7%, even more preferably between 1% and 5%, most preferably between 1 and 3% by weight of the liquid laundry detergent composition of the fatty alcohol ethoxylate non-ionic surfactant.
- Suitable alcohol ethoxylate nonionic surfactants include the condensation products of aliphatic alcohols with ethylene oxide. The alkyl chain of the aliphatic alcohol can either be straight or branched, substituted or unsubstituted. The starting alcohol can be naturally derived, e.g. starting from natural oils, or synthetically derived, e.g. alcohols obtained from for example oxo-, modified oxo- or Fischer-Tropsch processes. Examples of oxo-process derived alcohols include the Lial and Isalchem alcohols ex Sasol company and Lutensol alcohols ex BASF company. Examples of modified-oxo process derived alcohols include the Neodol alcohols ex Shell company. Fischer-Tropsch derived alcohols include Safol alcohols ex Sasol company. The alkoxylate chain of alcohol ethoxylates is made up solely of ethoxylate groups.
- Preferably, the fatty alcohol ethoxylate has an average alkyl carbon chain length of between 5 and 30, preferably between 8 and 18, more preferably between 10 and 16, most preferably between 12 and 15.
- Preferably, the fatty alcohol ethoxylate has an average degree of ethoxylation of between 0.5 and 20, preferably between 1 and 15, more preferably between 5 and 12, even more preferably between 6 and 10, most preferably between 7 and 8.
- Suitable for use herein are the ethoxylated alcohol of the formula R(OC2H4)n OH, wherein R is selected from the group consisting of aliphatic hydrocarbon radicals containing from about 8 to about 22 carbon atoms and the average value of n is from about 5 to about 22. In one aspect, particularly useful materials are condensation products of C9-C16 alcohols with from about 5 to about 20 moles of ethylene oxide per mole of alcohol. In another aspect, particularly useful materials are condensation products of C12-C16 alcohols with from about 6 to about 9 moles of ethylene oxide per mole of alcohol.
- Other non-limiting examples of nonionic surfactants may include: C12-C18 alkyl ethoxylates based on modified oxo alcohols, such as, NEODOL® nonionic surfactants from Shell; C12-C15 alkyl ethoxylates based on Fischer Tropsch Oxo alcohols, such as, SAFOL® nonionic surfactants from Sasol; C12-C18 alkyl ethoxylates based on natural or Ziegler alcohols, such as, Surfonic® nonionic surfactants from Huntsman; C14-C22 mid-chain branched alcohols ethoxylates, BAEx, wherein x is from 1 to 30.
- Preferably, the surfactant comprises a non-soap anionic surfactant, preferably between 10% and 35%, or even more preferably between 12% and 35%, most preferably between 15% and 30% or even more preferably between 15% and 25% by weight of the liquid laundry detergent composition of the non-soap anionic surfactant.
- The non-soap anionic surfactant may comprise a sulphate or a sulphonate anionic surfactant or a mixture thereof, preferably linear alkylbenzene sulphonate, alkyl sulphate, alkoxylated alkyl sulphate or a mixture thereof, more preferably a mixture of linear alkylbenzene sulphonate and alkoxylated alkyl sulphate. Preferably, the ratio of linear alkylbenzene sulphonate to alkoxylated alkyl sulphate more preferably the ratio of linear alkylbenzene sulphonate to ethoxylated alkyl sulphate is from 1:2 to 20:1, preferably from 1.1:1 to 15:1, more preferably from 1.2:1 to 10:1, even more preferably from 1.3:1 to 5:1, most preferably from 1.4:1 to 3:1.
- Preferably, the alkoxylated alkyl sulphate is an ethoxylated alkyl sulphate with an average degree of ethoxylation of between 0.5 and 7, preferably between 1 and 5, more preferably between 2 and 4, most preferably about 3. Alternatively, the non-soap surfactant comprises a mixture of one or more alkoxylated alkyl sulphates, preferably ethoxylated alkyl sulphates, and optionally an alkyl sulphate, the mixture having an average degree of ethoxylation of between 0.5 and 7, preferably between 1 and 5, more preferably between 2 and 4, most preferably about 3. The alkyl sulphate and/or alkoxylated alkyl sulphate preferably have an alkyl chain comprising on average between 8 and 18 carbon atoms, preferably between 10 and 16 carbons atoms, most preferably between 12 and 14 carbon atoms. Most preferably the alkoxylated alkyl sulphate is an ethoxylated alkyl chain comprising on average between 12 and 14 carbon atoms in its alkyl chain and has an average degree of ethoxylation of about 3. The alkyl chain of the alkoxylated alkyl sulphate surfactant may be linear or branched or a mixture thereof.
- The linear alkylbenzene sulphonate may be a C10-C16 linear alkylbenzene sulphonate or a C11-C14 linear alkylbenzene sulphonate or a mixture thereof.
- Exemplary linear alkylbenzene sulphonates are C10-C16 alkyl benzene sulfonic acids, or C11-C14 alkyl benzene sulfonic acids. By 'linear', we herein mean the alkyl group is linear. Alkyl benzene sulfonates are well known in the art.
- Preferably, the weight ratio of non-soap anionic surfactant to fatty alcohol ethoxylate non-ionic surfactant is between 5:1 and 23:1 preferably between 7:1 and 23:1, more preferably between 8:1 and 23:1, most preferably between 9:1 and 20:1.
- The surfactant may comprise a fatty acid, neutralised fatty acid soap or a mixture thereof. Preferably, the liquid laundry detergent composition may comprise less than 10%, preferably less than 8%, more preferably less than 5%, most preferably between 1% and 5% by weight of the liquid laundry detergent composition of fatty acid, neutralised fatty acid soap or a mixture thereof.
- The neutralised fatty acid soap may be alkali metal neutralised, amine neutralised or a mixture thereof. The alkali metal may be selected from sodium, potassium, magnesium or a mixture thereof, preferably sodium. The amine is preferably an alkanolamine, preferably selected from monoethanolamine, diethanolamine, triethanolamine or a mixture thereof, more preferably monoethanolamine.
- The fatty acid, neutralised fatty acid soap or mixture thereof may be selected from palm kernel fatty acid, coconut fatty acid, rapeseed fatty acid, neutralized palm kernel fatty acid, neutralized coconut fatty acid, neutralized rapeseed fatty acid, or mixture thereof, preferably neutralized palm kernel fatty acid.
- The liquid laundry detergent composition may comprise between 0.1% and 10%, preferably between 0.5% and 8%, more preferably between 1% and 7%, even more preferably between 2% and 6%, most preferably between 3% and 5% by weight of the liquid laundry detergent composition of a soil release polymer, wherein the soil release polymer is preferably selected from polyester terephthalates, polyethylene glycol containing soil release polymers and a mixture thereof. An example of a preferred polyethyleneglycol containing soil release polymer comprises a polyethylene glycol graft polymer comprising a polyethylene glycol backbone (Pluriol E6000) and hydrophobic vinyl acetate side chains, comprising 40% by weight of the polymer system of a polyethylene glycol backbone polymer and 60% by weight of the polymer system of the grafted vinyl acetate side chains. Polyester terephtalate soil release polymers are commercially available from Clariant under the Texcare SRN and SRA tradenames. One particularly preferred polyester terephtalate soil release polymer is Texcare SRA300.
- The liquid laundry detergent composition may comprise a polyethyleneimine, preferably an alkoxylated polyethyleneimine, more preferably an ethoxylated polyethyleneimine and wherein preferably the liquid laundry detergent composition comprises greater than 0.5%, preferably between 1% and 10%, more preferably between 2% and 7%, most preferably between 3% and 5% by weight of the liquid laundry detergent composition of the polyethyleneimine, preferably ethoxylated polyethyleneimine. An example of an ethoxylated polyethyleneimine includes an ethoxylated polyethyleneimine with a polyethyleneimine backbone with a molecular weight of about 600, and comprising on average about 20 ethoxy units per ethoxylation chain.
- Preferably, the ratio of polyethyleneimine to soil release polymer, more preferably the ratio of ethoxylated polyethyleneimine to soil release polymer is higher than 1:1, preferably between 1.1:1 and 5:1, more preferably between 1.2:1 and 3:1, most preferably between 1.3:1 and 2:1.
- The liquid laundry detergent composition may comprise a polysaccharide polymer, a modified polysaccharide polymer, a polysaccharide derived polymer, or a mixture thereof, preferably a cellulosic polymer, a modified cellulosic polymer or a mixture thereof, most preferably a cationically modified cellulosic polymer or a mixture thereof. Most preferably the polysaccharide polymer is a cationically modified hydroethylcellulose ("Polyquaternium 10).
- Preferably, the water-soluble unit dose article comprises up to 60% preferably up to 50%, more preferably up to 40% by weight of the liquid laundry detergent composition of a non-aqueous solvent, preferably selected from propylene glycol, dipropylene glycol, glycerol, sorbitol, ethanol or a mixture thereof.
- Preferably, the liquid laundry detergent composition comprises less than 2%, preferably less than 1% by weight of the liquid laundry detergent composition of aminocarboxylate chelants, aminophosphonate chelants or a mixture thereof.
- Preferably, the liquid laundry detergent composition has a pH from 6 to 10 preferably from 7 to 9, more preferably from 7 to 8. Preferably, the liquid laundry detergent composition comprises a pH adjusting agent selected from alkanolamines, preferably monethanolamine, diethanolamine, triethanolamine or a mixture thereof, most preferably monoethanolamine.
- Preferably, the liquid laundry detergent composition comprises an adjunct ingredient selected from hueing dyes, polymers, builders, dye transfer inhibiting agents, dispersants, enzymes, enzyme stabilizers, catalytic materials, bleach, bleach activators, polymeric dispersing agents, anti-redeposition agents, suds suppressors, aesthetic dyes, opacifiers, perfumes, perfume delivery systems, structurants, hydrotropes, processing aids, pigments, amphoteric surfactants, cyclic diamines, zwitterionic polyamines, anti-oxidants, preservatives and mixtures thereof.
- Preferably, the water-soluble unit dose article comprises 15% or less by weight of the unit dose article of water, preferably the unit dose article comprises between 0.1% and 15%, more preferably between 1% and 12.5% by weight of the unit dose article of water.
- Those skilled in the art will know how to make the unit dose article and laundry detergent composition of the present invention using known techniques in the art.
- During manufacture, a first water-soluble film may be shaped to comprise an open compartment into which the detergent composition is added. A second water-soluble film is then laid over the first film in such an orientation as to close the opening of the compartment. The first and second films are then sealed together along a seal region using known sealing means such as solvent, heat or a mixture thereof.
- A design of experiment modeling exercise has been conducted to assess the irritancy profile, of soluble unit dose laundry formulations. The below formulation space has been assessed, having formulated 106 compositions within this space, as dictated by the response surface experimental design. Formulations were prepared through simple mixing of the individual raw materials.
Ingredients in weight % active MIN MAX Linear alkyl benzene sulphonate anionic surfactant (LAS) 0.00 35.84 C1214 Alkyl sulphate anionic surfactant with an average degree of ethoxylation of 3 (AES) 0.00 24.60 C1215EO7 nonionic surfactant (NI) 1.90 22.76 DTPK Fatty Acid (FA) 0.00 16.85 Ethoxylated polyethyleneimine (PEI600E020)* 0.00 5.60 Polyethyleneglycol graft polymer** 0.00 4.19 HEDP chelant 0.00 2.50 Citric Acid 0.00 0.79 Water 6.40 11.50 MgCl2 0.18 0.66 pH 6.90 8.60 Constant ingredients Glycerol K2SO3 Hydrogenated Castor Oil Enzymes (Protease only) Perfume Perfume capsules Brightener *ethoxylate polyethyleneimine having an average degree of ethoxylation of 20 per EO chain and a polyethyleneimine backbone with MW of about 600
**polyethylene glycol graft polymer comprising a polyethylene glycol backbone (Pluriol E6000) and hydrophobic vinyl acetate side chains, comprising 40% by weight of the polymer system of a polyethylene glycol backbone polymer and 60% by weight of the polymer system of the grafted vinyl acetate side chains - (Although the FIGs refer to 'HLAS' (linear alkyl benzene sulphonic acid, in the tests, the HLAS was neutralized to the corresponding salt, linear alkyl benzene sulphonate)
- Test formulations were assessed for irritancy profile following the OECD Guidelines for Testing of Chemicals, Test No. 438: Isolated Chicken Eye Test Method for Identifying i) Chemicals Inducing Serious Eye Damage and ii) Chemicals Not Requiring Classification for Eye Irritation or Serious Eye Damage (Adopted version : 26 July 2013).
- An irritation index has been calculated according to formula I :
A higher irritation index reflects a worse irritancy profile. - Test results from experiments conducted were fed into a digital test design using commercially available JMP predictive software package. From this the irritancy formulation model then got calculated.
- The below
Figures 1 to 4 have been extracted from the resulting irritancy formulation model. The AES data reflect the irritancy index of an AES comprising paste (67.5% AES + 20% DTPK fatty acid + water/minors). Active surfactant content reflects the combined % level by weight of the detergent composition of HLAS, AES, FA and NI surfactant. Values in the X-axis reflect weight fraction of the specific compound relative to the total detergent formulation. - From the data the following conclusions can be drawn :
- Within both the main LAS and main AES formulations, lower active surfactant content formulations have shown a lower irritation index compared to higher active surfactant content formulations (single variable comparison), eg the irritation index ex
FIG 1 (30% active, LAS as main anionic surfactant) is lower (103.5 vs 123.1) compared toFIG 3 (40% active, LAS as main anionic surfactant), the irritation index exFIG 2 (30% active, AES as main anionic surfactant) is lower (118.8vs 140.7) compared toFIG 4 (40% active, AES as main anionic surfactant). - Across the four formulation spaces exemplified, i.e. 30% or 40% surfactant active content - LAS or AES as main anionic surfactant, increasing the nonionic surfactant content beyond 10% would exponentially increase the irritation index, e.g. the irritation index at a weight fraction of 0.2 (or 20 wt% in formula) is substantially higher than the irritation index at a weight fraction of 0.1 (or 10 wt% in formula) across the 4 FIGS.
- Within both the 30% and the 40% active surfactant content formulations, LAS is preferred as a main anionic surfactant compared to AES, i.e. LAS as main anionic surfactant formulations have shown a lower irritation index compared to equal total surfactant content formulations with AES as main anionic surfactant (single variable comparison), eg the irritation index ex
FIG 1 (30% active, LAS as main anionic surfactant) is lower (103.5 vs 123.1) compared toFIG 2 (30% active, AES as main anionic surfactant), the irritation index exFIG 3 (40% active, LAS as main anionic surfactant) is lower (117.2 vs 140.7) compared toFIG 4 (40% active, AES as main anionic surfactant). - The dimensions and values disclosed herein are not to be understood as being strictly limited to the exact numerical values recited. Instead, unless otherwise specified, each such dimension is intended to mean both the recited value and a functionally equivalent range surrounding that value. For example, a dimension disclosed as "40 mm" is intended to mean "about 40 mm."
Claims (20)
- The use of a liquid laundry detergent composition comprised in a water-soluble unit dose article to provide laundry cleaning benefits and to minimize potential adverse effects associated with accidental exposure to said liquid laundry detergent composition contained within the water-soluble unit dose article,
wherein the liquid laundry detergent composition comprises between 10% and 35%, or even more preferably between 12% and 35%, most preferably between 15% and 30% or even between 15% and 25% by weight of the liquid laundry detergent composition of a surfactant. - The use according to claim 1 wherein the liquid laundry detergent composition minimizes potential adverse effects associated with accidental exposure to said liquid laundry detergent composition contained within the water-soluble unit dose article by minimizing potential adverse effects of the water-soluble unit dose article, preferably reducing skin irritation, eye irritation or a mixture thereof, most preferably eye irritation, of the water-soluble unit dose article.
- The use according to any preceding claims, wherein the surfactant comprises a fatty alcohol ethoxylate non-ionic surfactant and wherein the liquid laundry detergent composition comprises between 0% and 9.5%, preferably between 0.01% and 9%, more preferably between 0.1% and 7%, even more preferably between 1% and 5%, most preferably between 1 and 3% by weight of the liquid laundry detergent composition of the fatty alcohol ethoxylate non-ionic surfactant.
- The use according to claim 3 wherein the fatty alcohol ethoxylate has an average alkyl carbon chain length of between 5 and 30, preferably between 8 and 18, more preferably between 10 and 16, most preferably between 12 and 15.
- The use according to claims 3 or 4 wherein the fatty alcohol ethoxylate has an average degree of ethoxylation of between 0.5 and 20, preferably between 1 and 15, more preferably between 5 and 12, even more preferably between 6 and 10, most preferably between 7 and 8.
- The use according to any preceding claims wherein the surfactant comprises a non-soap anionic surfactant, preferably between 10% and 35%, or even more preferably between 12% and 35%, most preferably between 15% and 30% or even more preferably between 15% and 25% by weight of the liquid laundry detergent composition of the non-soap anionic surfactant, wherein preferably the non-soap anionic surfactant comprises linear alkylbenzene sulphonate, alkoxylated alkyl sulphate or a mixture thereof, more preferably a mixture thereof wherein preferably the ratio of linear alkylbenzene sulphonate to alkoxylated alkyl sulphate more preferably the ratio of linear alkylbenzene sulphonate to ethoxylated alkyl sulphate is from 1:2 to 20:1, preferably from 1.1:1 to 15:1, more preferably from 1.2:1 to 10:1, even more preferably from 1.3:1 to 5:1, most preferably from 1.4:1 to 3:1.
- The use according to claims 3 to 6 wherein the weight ratio of non-soap anionic surfactant to fatty alcohol ethoxylate non-ionic surfactant is between 5:1 and 23:1 preferably between 7:1 and 23:1, more preferably between 8:1 and 23:1, most preferably between 9:1 and 20:1.
- The use according to any preceding claims wherein the surfactant comprises a fatty acid, neutralised fatty acid soap or a mixture thereof, preferably wherein the liquid laundry detergent composition comprises less than 10%, preferably less than 8%, more preferably less than 5%, most preferably between 1% and 5% by weight of the liquid laundry detergent composition of fatty acid, neutralised fatty acid soap or a mixture thereof.
- The use according to any preceding claims wherein the liquid laundry detergent composition comprises between 0.1% and 10%, preferably between 0.5% and 8%, more preferably between 1% and 7%, even more preferably between 2% and 6%, most preferably between 3% and 5% by weight of the liquid laundry detergent composition of a soil release polymer, wherein the soil release polymer is preferably selected from polyester terephthalates, polyethylene glycol containing soil release polymers and a mixture thereof.
- The use according to any preceding claims wherein the liquid laundry detergent composition comprises a polyethyleneimine, preferably an ethoxylated polyethyleneimine and wherein preferably the liquid laundry detergent composition comprises greater than 0.5%, preferably between 1% and 10%, more preferably between 2% and 7%, most preferably between 3% and 5% by weight of the liquid laundry detergent composition of the polyethyleneimine, preferably ethoxylated polyethyleneimine.
- The use according to any of claims 9-10 wherein the ratio of polyethyleneimine to soil release polymer, preferably the ratio of ethoxylated polyethyleneimine to soil release polymer is higher than 1:1, preferably between 1.1:1 and 5:1, more preferably between 1.2:1 and 3:1, most preferably between 1.3:1 and 2:1.
- The use according to any preceding claims wherein the liquid laundry detergent composition comprises a polysaccharide polymer, a modified polysaccharide polymer, a polysaccharide derived polymer, or a mixture thereof, preferably a cellulosic polymer, a modified cellulosic polymer or a mixture thereof, most preferably a cationically modified cellulosic polymer or a mixture thereof.
- The use according to any preceding claims wherein the water-soluble unit dose article comprises a water-soluble film, preferably wherein the water-soluble film comprises polyvinyl alcohol, preferably wherein the water-soluble film comprises polyvinyl alcohol polymer or copolymer, preferably a blend of polyvinylalcohol polymers and/or polyvinylalcohol copolymers, preferably selected from sulphonated and carboxylated anionic polyvinylalcohol copolymers especially carboxylated anionic polyvinylalcohol copolymers, most preferably a blend of a polyvinylalcohol homopolymer and a carboxylated anionic polyvinylalcohol copolymer.
- The use according to any preceding claims wherein the water-soluble unit dose article comprises up to 60% preferably up to 50%, more preferably up to 40% by weight of the liquid laundry detergent composition of a non-aqueous solvent, preferably selected from propylene glycol, dipropylene glycol, glycerol, sorbitol, ethanol or a mixture thereof.
- The use according to any preceding claims wherein the liquid laundry detergent composition comprises less than 2%, preferably less than 1% by weight of the liquid laundry detergent composition of aminocarboxylate chelants, aminophosphonate chelants, or a mixture thereof.
- The use according to any preceding claims wherein the liquid laundry detergent composition has a pH from 6 to 10 preferably from 7 to 9, more preferably from 7 to 8, preferably wherein the liquid laundry detergent composition comprises a pH adjusting agent selected from alkanolamines, preferably monethanolamine, diethanolamine, triethanolamine or a mixture thereof, most preferably monoethanolamine.
- The use according to any preceding claims wherein the liquid laundry detergent composition comprises an adjunct ingredient selected from hueing dyes, polymers, builders, dye transfer inhibiting agents, dispersants, enzymes, enzyme stabilizers, catalytic materials, bleach, bleach activators, polymeric dispersing agents, anti-redeposition agents, suds suppressors, aesthetic dyes, opacifiers, perfumes, perfume delivery systems, structurants, hydrotropes, processing aids, pigments, amphoteric surfactants, cyclic diamines, zwitterionic polyamines, anti-oxidants, preservatives and mixtures thereof.
- The use according to any preceding claims wherein the water-soluble unit dose article comprises 15% or less by weight of the unit dose article of water, preferably the unit dose article comprises between 0.1% and 15%, more preferably between 1% and 12.5% by weight of the unit dose article of water.
- The use according to any preceding claims wherein the potential adverse effects associated with accidental exposure to said liquid laundry detergent composition contained within the water-soluble unit dose article is when the water-soluble unit dose article is accidentally prematurely ruptured and the skin, eye or mixture thereof of the user is contacted with the liquid laundry detergent composition.
- The use according to any preceding claims wherein the water-soluble unit dose article comprises at least two compartments and the liquid laundry detergent composition is comprised in at least one compartment, preferably wherein the water-soluble unit dose article comprises at least two compartments of different sizes and the liquid laundry detergent composition is comprised in at least the larger compartment.
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201762461351P | 2017-02-21 | 2017-02-21 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| EP3363888A1 true EP3363888A1 (en) | 2018-08-22 |
Family
ID=61256723
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP18157942.6A Withdrawn EP3363888A1 (en) | 2017-02-21 | 2018-02-21 | Use of liquid laundry detergent composition to minimize adverse effects upon accidental exposure to contents of water-soluble unit dose articles |
Country Status (1)
| Country | Link |
|---|---|
| EP (1) | EP3363888A1 (en) |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2003052040A1 (en) * | 2001-12-14 | 2003-06-26 | Unilever N.V. | Water-soluble package with layered liquid laundry detergent |
| US20130072410A1 (en) * | 2011-09-20 | 2013-03-21 | The Sun Products Corporation | Cleaning Formulations with Improved Surfactant Solubility and Methods of Production and Use Thereof |
| WO2016155993A1 (en) | 2015-04-02 | 2016-10-06 | Unilever Plc | Composition |
-
2018
- 2018-02-21 EP EP18157942.6A patent/EP3363888A1/en not_active Withdrawn
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2003052040A1 (en) * | 2001-12-14 | 2003-06-26 | Unilever N.V. | Water-soluble package with layered liquid laundry detergent |
| US20130072410A1 (en) * | 2011-09-20 | 2013-03-21 | The Sun Products Corporation | Cleaning Formulations with Improved Surfactant Solubility and Methods of Production and Use Thereof |
| WO2016155993A1 (en) | 2015-04-02 | 2016-10-06 | Unilever Plc | Composition |
Non-Patent Citations (6)
| Title |
|---|
| CORNELIS M ET AL: "Prediction of eye irritancy potential of surfactants by cytotoxicity tests in vitro on cultures of human skin fibroblasts and keratinocytes", TOXICOLOGY IN VITRO, ELSEVIER SCIENCE, GB, vol. 6, no. 2, 1 March 1992 (1992-03-01), pages 119 - 128, XP025543233, ISSN: 0887-2333, [retrieved on 19920301], DOI: 10.1016/0887-2333(92)90004-B * |
| EFFENDY AND MAIBACH: "Surfactants and experimental irritant contact dermatitis", CONTACT DERMATITIS, vol. 33, 1 January 1995 (1995-01-01), pages 217 - 225, XP055127310, DOI: 10.1111/j.1600-0536.1995.tb00470.x * |
| H KORTING ET AL: "Discrimination of the irritancy potential of surfactants in vitro by two cytotoxicity assays using normal human keratinocytes, HaCaT cells and 3T3 mouse fibroblasts: Correlation with in vivo data from a soap chamber assay", JOURNAL OF DERMATOLOGICAL SCIENCE, vol. 7, no. 2, 1 April 1994 (1994-04-01), AMSTERDAM, NL, pages 119 - 129, XP055491103, ISSN: 0923-1811, DOI: 10.1016/0923-1811(94)90085-X * |
| I. CLAUDET ET AL: "Expositions des enfants aux lessives capsules, écodoses ou pods : plus toxiques que les lessives traditionnelles ?", ARCHIVES DE PEDIATRIE., vol. 21, no. 6, 1 June 2014 (2014-06-01), FR, pages 601 - 607, XP055465337, ISSN: 0929-693X, DOI: 10.1016/j.arcped.2014.03.020 * |
| L D RHEIN ET AL: "Surfactant structure effects on swelling of isolated human stratum corneum Presented at the Society of Cosmetic Chemists' Annual Meeting", J. SOC. COSMET. CHEM, vol. 37, 1 May 1986 (1986-05-01), pages 125 - 139, XP055491871 * |
| R. A. TUPKER ET AL: "Irritancy ranking of anionic detergents using one-time occlusive, repeated occlusive and repeated open tests", CONTACT DERMATITIS: ENVIRONMENTAL AND OCCUPATIONAL DERMATITIS, vol. 40, no. 6, 1 June 1999 (1999-06-01), US, pages 316 - 322, XP055491131, ISSN: 0105-1873, DOI: 10.1111/j.1600-0536.1999.tb06082.x * |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US11401489B2 (en) | Water-soluble multicompartment unit dose article | |
| EP3517596B1 (en) | Method of making an opaque liquid detergent composition | |
| RU2508395C2 (en) | Detergent composition containing surfactant-boosting polymers | |
| US10323220B2 (en) | Laundry detergent composition comprising a cyclic diamine and an amphoteric/anionic surfactant mixture | |
| US20190048298A1 (en) | Water-soluble unit dose article comprising an amphiphilic graft polymer and a polyester terephthalate | |
| EP3170882A1 (en) | Liquid laundry detergent composition comprising a polymer system | |
| US20230279317A1 (en) | Water-soluble unit dose article comprising an ethoxylated secondary alcohol non-ionic surfactant | |
| EP3363884A1 (en) | Use of liquid laundry detergent composition in water-soluble unit dose article to minimize adverse effects upon accidental exposure to contents of | |
| EP3495466A1 (en) | Use of a liquid laundry detergent composition | |
| EP3978590A1 (en) | Water-soluble unit dose article comprising a first non-ionic surfactant and a second non-ionic surfactant | |
| EP3363888A1 (en) | Use of liquid laundry detergent composition to minimize adverse effects upon accidental exposure to contents of water-soluble unit dose articles | |
| EP3363882A1 (en) | Use of liquid laundry detergent composition to minimize adverse effects upon accidental exposure to contents of water-soluble unit dose articles | |
| EP3363885A1 (en) | Use of liquid laundry detergent composition in water-soluble unit dose article to minimize emesis and/or narcosis effects upon accidental exposure to contents of | |
| EP3363887A1 (en) | Use of liquid laundry detergent composition to minimize adverse effects upon accidental exposure to contents of water-soluble unit dose articles | |
| EP3342848B1 (en) | Water-soluble unit dose article comprising zwitterionic polyamine | |
| EP3363889A1 (en) | Use of liquid laundry detergent composition to minimize adverse effects upon accidental exposure to contents of water-soluble unit dose articles | |
| EP3363883A1 (en) | Use of liquid laundry detergent composition to minimize adverse effects upon accidental exposure to contents of water-soluble unit dose articles | |
| EP3372664A2 (en) | Use of liquid laundry detergent composition to minimize adverse effects upon accidental exposure to contents of water-soluble unit dose articles | |
| EP3342850A1 (en) | Water-soluble unit dose article comprising zwitterionic polyamine | |
| EP3363886A1 (en) | Use of non-ionic surfactant to minimize adverse effects upon accidental exposure to detergent compositions | |
| CA3044533A1 (en) | Water-soluble unit dose article comprising zwitterionic polyamine | |
| US20230365888A1 (en) | Water-soluble unit dose article | |
| US20230279311A1 (en) | Water-soluble unit dose article comprising an ethoxylated alcohol non-ionic surfactant | |
| US20230279310A1 (en) | Water-soluble unit dose article comprising a narrow range ethoxylate alkyl alcohol non-ionic surfactant | |
| US20230279318A1 (en) | Water-soluble unit dose article comprising an ethoxylated secondary alcohol non-ionic surfactant |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): AL AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL PT RO RS SE SI SK SM TR |
|
| AX | Request for extension of the european patent |
Extension state: BA ME |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: THE APPLICATION IS DEEMED TO BE WITHDRAWN |
|
| 18D | Application deemed to be withdrawn |
Effective date: 20190223 |