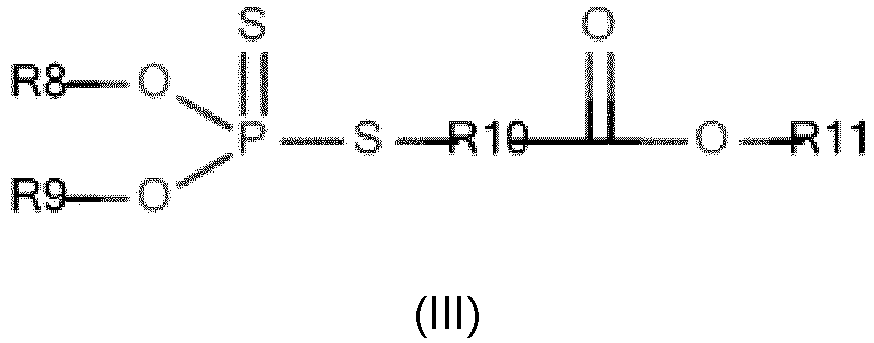EP3110929B1 - Lubricating composition based on metal nanoparticles - Google Patents
Lubricating composition based on metal nanoparticles Download PDFInfo
- Publication number
- EP3110929B1 EP3110929B1 EP15706812.3A EP15706812A EP3110929B1 EP 3110929 B1 EP3110929 B1 EP 3110929B1 EP 15706812 A EP15706812 A EP 15706812A EP 3110929 B1 EP3110929 B1 EP 3110929B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- lubricant composition
- group
- composition according
- compound
- dithiophosphate
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 239000000203 mixture Substances 0.000 title claims description 128
- 239000002082 metal nanoparticle Substances 0.000 title claims 8
- 230000001050 lubricating effect Effects 0.000 title description 96
- 150000001875 compounds Chemical class 0.000 claims description 49
- 125000004432 carbon atom Chemical group C* 0.000 claims description 32
- 229910052751 metal Inorganic materials 0.000 claims description 26
- 239000002199 base oil Substances 0.000 claims description 23
- 239000002184 metal Substances 0.000 claims description 23
- 125000000217 alkyl group Chemical group 0.000 claims description 21
- 239000000314 lubricant Substances 0.000 claims description 20
- 230000001603 reducing effect Effects 0.000 claims description 18
- 229910052723 transition metal Inorganic materials 0.000 claims description 16
- 150000003624 transition metals Chemical class 0.000 claims description 16
- 239000000654 additive Substances 0.000 claims description 14
- 239000002270 dispersing agent Substances 0.000 claims description 13
- 230000003647 oxidation Effects 0.000 claims description 12
- 238000007254 oxidation reaction Methods 0.000 claims description 12
- ZOKXTWBITQBERF-UHFFFAOYSA-N Molybdenum Chemical compound [Mo] ZOKXTWBITQBERF-UHFFFAOYSA-N 0.000 claims description 11
- 239000000446 fuel Substances 0.000 claims description 11
- 229910052750 molybdenum Inorganic materials 0.000 claims description 10
- 239000011733 molybdenum Substances 0.000 claims description 10
- 239000007787 solid Substances 0.000 claims description 10
- 150000001768 cations Chemical class 0.000 claims description 8
- 239000003607 modifier Substances 0.000 claims description 8
- WFKWXMTUELFFGS-UHFFFAOYSA-N tungsten Chemical compound [W] WFKWXMTUELFFGS-UHFFFAOYSA-N 0.000 claims description 8
- 229910052721 tungsten Inorganic materials 0.000 claims description 8
- 239000010937 tungsten Substances 0.000 claims description 8
- 229910052798 chalcogen Inorganic materials 0.000 claims description 7
- 150000001787 chalcogens Chemical class 0.000 claims description 7
- -1 MoSe2 Inorganic materials 0.000 claims description 6
- 150000001412 amines Chemical class 0.000 claims description 6
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Chemical compound [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 claims description 6
- 239000003963 antioxidant agent Substances 0.000 claims description 5
- 229920000642 polymer Polymers 0.000 claims description 5
- 230000000996 additive effect Effects 0.000 claims description 4
- 238000005260 corrosion Methods 0.000 claims description 4
- 150000002148 esters Chemical class 0.000 claims description 4
- 238000002173 high-resolution transmission electron microscopy Methods 0.000 claims description 4
- 238000005461 lubrication Methods 0.000 claims description 4
- CJSZLPWCFGHOPN-UHFFFAOYSA-N triazanium dioxido-sulfanylidene-sulfido-lambda5-phosphane Chemical class P([O-])([O-])(=S)[S-].[NH4+].[NH4+].[NH4+] CJSZLPWCFGHOPN-UHFFFAOYSA-N 0.000 claims description 4
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 claims description 3
- QCWXUUIWCKQGHC-UHFFFAOYSA-N Zirconium Chemical compound [Zr] QCWXUUIWCKQGHC-UHFFFAOYSA-N 0.000 claims description 3
- 229910052735 hafnium Inorganic materials 0.000 claims description 3
- VBJZVLUMGGDVMO-UHFFFAOYSA-N hafnium atom Chemical compound [Hf] VBJZVLUMGGDVMO-UHFFFAOYSA-N 0.000 claims description 3
- 229910052758 niobium Inorganic materials 0.000 claims description 3
- 239000010955 niobium Substances 0.000 claims description 3
- GUCVJGMIXFAOAE-UHFFFAOYSA-N niobium atom Chemical compound [Nb] GUCVJGMIXFAOAE-UHFFFAOYSA-N 0.000 claims description 3
- 229910052697 platinum Inorganic materials 0.000 claims description 3
- 229910052702 rhenium Inorganic materials 0.000 claims description 3
- WUAPFZMCVAUBPE-UHFFFAOYSA-N rhenium atom Chemical compound [Re] WUAPFZMCVAUBPE-UHFFFAOYSA-N 0.000 claims description 3
- 229910052715 tantalum Inorganic materials 0.000 claims description 3
- GUVRBAGPIYLISA-UHFFFAOYSA-N tantalum atom Chemical compound [Ta] GUVRBAGPIYLISA-UHFFFAOYSA-N 0.000 claims description 3
- 229910052719 titanium Inorganic materials 0.000 claims description 3
- 239000010936 titanium Substances 0.000 claims description 3
- 238000004627 transmission electron microscopy Methods 0.000 claims description 3
- 229910052726 zirconium Inorganic materials 0.000 claims description 3
- 229910052961 molybdenite Inorganic materials 0.000 claims description 2
- CWQXQMHSOZUFJS-UHFFFAOYSA-N molybdenum disulfide Chemical compound S=[Mo]=S CWQXQMHSOZUFJS-UHFFFAOYSA-N 0.000 claims description 2
- 229910052982 molybdenum disulfide Inorganic materials 0.000 claims description 2
- 229910016021 MoTe2 Inorganic materials 0.000 claims 1
- 229910020042 NbS2 Inorganic materials 0.000 claims 1
- 229910020039 NbSe2 Inorganic materials 0.000 claims 1
- 229910020046 NbTe2 Inorganic materials 0.000 claims 1
- 229910004211 TaS2 Inorganic materials 0.000 claims 1
- 229910004214 TaSe2 Inorganic materials 0.000 claims 1
- 229910003092 TiS2 Inorganic materials 0.000 claims 1
- 229910010322 TiS3 Inorganic materials 0.000 claims 1
- 229910003090 WSe2 Inorganic materials 0.000 claims 1
- 229910006247 ZrS2 Inorganic materials 0.000 claims 1
- 230000003247 decreasing effect Effects 0.000 claims 1
- 239000002105 nanoparticle Substances 0.000 description 61
- 150000002430 hydrocarbons Chemical group 0.000 description 35
- 230000005540 biological transmission Effects 0.000 description 27
- 239000003921 oil Substances 0.000 description 15
- 238000000034 method Methods 0.000 description 13
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 12
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 11
- 229910052717 sulfur Inorganic materials 0.000 description 10
- 239000011593 sulfur Substances 0.000 description 10
- 239000012141 concentrate Substances 0.000 description 9
- ITRNXVSDJBHYNJ-UHFFFAOYSA-N tungsten disulfide Chemical compound S=[W]=S ITRNXVSDJBHYNJ-UHFFFAOYSA-N 0.000 description 9
- 229920013639 polyalphaolefin Polymers 0.000 description 8
- 125000004429 atom Chemical group 0.000 description 7
- KZNICNPSHKQLFF-UHFFFAOYSA-N succinimide Chemical group O=C1CCC(=O)N1 KZNICNPSHKQLFF-UHFFFAOYSA-N 0.000 description 7
- 239000011701 zinc Substances 0.000 description 7
- 229910003472 fullerene Inorganic materials 0.000 description 6
- 239000010705 motor oil Substances 0.000 description 6
- 229910052757 nitrogen Inorganic materials 0.000 description 6
- 229910052760 oxygen Inorganic materials 0.000 description 6
- 239000001301 oxygen Substances 0.000 description 6
- 229910052725 zinc Inorganic materials 0.000 description 6
- XMWRBQBLMFGWIX-UHFFFAOYSA-N C60 fullerene Chemical compound C12=C3C(C4=C56)=C7C8=C5C5=C9C%10=C6C6=C4C1=C1C4=C6C6=C%10C%10=C9C9=C%11C5=C8C5=C8C7=C3C3=C7C2=C1C1=C2C4=C6C4=C%10C6=C9C9=C%11C5=C5C8=C3C3=C7C1=C1C2=C4C6=C2C9=C5C3=C12 XMWRBQBLMFGWIX-UHFFFAOYSA-N 0.000 description 5
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical compound [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 description 5
- 239000012208 gear oil Substances 0.000 description 5
- 239000004519 grease Substances 0.000 description 5
- 239000002245 particle Substances 0.000 description 5
- 229910052698 phosphorus Inorganic materials 0.000 description 5
- 230000008569 process Effects 0.000 description 5
- 230000009467 reduction Effects 0.000 description 5
- 229920000089 Cyclic olefin copolymer Polymers 0.000 description 4
- MYMOFIZGZYHOMD-UHFFFAOYSA-N Dioxygen Chemical compound O=O MYMOFIZGZYHOMD-UHFFFAOYSA-N 0.000 description 4
- PPBRXRYQALVLMV-UHFFFAOYSA-N Styrene Chemical compound C=CC1=CC=CC=C1 PPBRXRYQALVLMV-UHFFFAOYSA-N 0.000 description 4
- 125000003342 alkenyl group Chemical group 0.000 description 4
- 125000000304 alkynyl group Chemical group 0.000 description 4
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 description 4
- 229920001577 copolymer Polymers 0.000 description 4
- 229910052500 inorganic mineral Inorganic materials 0.000 description 4
- 239000011707 mineral Substances 0.000 description 4
- 239000002086 nanomaterial Substances 0.000 description 4
- 125000004430 oxygen atom Chemical group O* 0.000 description 4
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 4
- 125000004437 phosphorous atom Chemical group 0.000 description 4
- 238000012360 testing method Methods 0.000 description 4
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 3
- 229910000831 Steel Inorganic materials 0.000 description 3
- 150000004770 chalcogenides Chemical class 0.000 description 3
- 229910001092 metal group alloy Inorganic materials 0.000 description 3
- 150000002739 metals Chemical class 0.000 description 3
- 239000010959 steel Substances 0.000 description 3
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 2
- 229920002943 EPDM rubber Polymers 0.000 description 2
- 229910016001 MoSe Inorganic materials 0.000 description 2
- PXHVJJICTQNCMI-UHFFFAOYSA-N Nickel Chemical compound [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 description 2
- 241001080024 Telles Species 0.000 description 2
- 150000001336 alkenes Chemical class 0.000 description 2
- 239000007866 anti-wear additive Substances 0.000 description 2
- 229910052787 antimony Inorganic materials 0.000 description 2
- WATWJIUSRGPENY-UHFFFAOYSA-N antimony atom Chemical compound [Sb] WATWJIUSRGPENY-UHFFFAOYSA-N 0.000 description 2
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 2
- 230000015572 biosynthetic process Effects 0.000 description 2
- 235000010354 butylated hydroxytoluene Nutrition 0.000 description 2
- 229910052799 carbon Inorganic materials 0.000 description 2
- 229910052802 copper Inorganic materials 0.000 description 2
- 239000010949 copper Substances 0.000 description 2
- 235000014113 dietary fatty acids Nutrition 0.000 description 2
- 150000002118 epoxides Chemical class 0.000 description 2
- 230000001747 exhibiting effect Effects 0.000 description 2
- 229930195729 fatty acid Natural products 0.000 description 2
- 239000000194 fatty acid Substances 0.000 description 2
- 229910052742 iron Inorganic materials 0.000 description 2
- 238000012423 maintenance Methods 0.000 description 2
- 239000000463 material Substances 0.000 description 2
- 239000002480 mineral oil Substances 0.000 description 2
- JRZJOMJEPLMPRA-UHFFFAOYSA-N olefin Natural products CCCCCCCC=C JRZJOMJEPLMPRA-UHFFFAOYSA-N 0.000 description 2
- ISWSIDIOOBJBQZ-UHFFFAOYSA-N phenol group Chemical group C1(=CC=CC=C1)O ISWSIDIOOBJBQZ-UHFFFAOYSA-N 0.000 description 2
- 229920000058 polyacrylate Polymers 0.000 description 2
- 229920000768 polyamine Polymers 0.000 description 2
- 238000004901 spalling Methods 0.000 description 2
- 229910052714 tellurium Inorganic materials 0.000 description 2
- PORWMNRCUJJQNO-UHFFFAOYSA-N tellurium atom Chemical compound [Te] PORWMNRCUJJQNO-UHFFFAOYSA-N 0.000 description 2
- 229910052718 tin Inorganic materials 0.000 description 2
- 239000011135 tin Substances 0.000 description 2
- WMYJOZQKDZZHAC-UHFFFAOYSA-H trizinc;dioxido-sulfanylidene-sulfido-$l^{5}-phosphane Chemical compound [Zn+2].[Zn+2].[Zn+2].[O-]P([O-])([S-])=S.[O-]P([O-])([S-])=S WMYJOZQKDZZHAC-UHFFFAOYSA-H 0.000 description 2
- KWKAKUADMBZCLK-UHFFFAOYSA-N 1-octene Chemical compound CCCCCCC=C KWKAKUADMBZCLK-UHFFFAOYSA-N 0.000 description 1
- SPSPIUSUWPLVKD-UHFFFAOYSA-N 2,3-dibutyl-6-methylphenol Chemical compound CCCCC1=CC=C(C)C(O)=C1CCCC SPSPIUSUWPLVKD-UHFFFAOYSA-N 0.000 description 1
- NIXOWILDQLNWCW-UHFFFAOYSA-M Acrylate Chemical compound [O-]C(=O)C=C NIXOWILDQLNWCW-UHFFFAOYSA-M 0.000 description 1
- 241000234282 Allium Species 0.000 description 1
- 235000002732 Allium cepa var. cepa Nutrition 0.000 description 1
- BTBUEUYNUDRHOZ-UHFFFAOYSA-N Borate Chemical compound [O-]B([O-])[O-] BTBUEUYNUDRHOZ-UHFFFAOYSA-N 0.000 description 1
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 1
- VYZAMTAEIAYCRO-UHFFFAOYSA-N Chromium Chemical compound [Cr] VYZAMTAEIAYCRO-UHFFFAOYSA-N 0.000 description 1
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 1
- VGGSQFUCUMXWEO-UHFFFAOYSA-N Ethene Chemical compound C=C VGGSQFUCUMXWEO-UHFFFAOYSA-N 0.000 description 1
- 239000005977 Ethylene Substances 0.000 description 1
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 1
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 1
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 1
- PWHULOQIROXLJO-UHFFFAOYSA-N Manganese Chemical compound [Mn] PWHULOQIROXLJO-UHFFFAOYSA-N 0.000 description 1
- 241001465754 Metazoa Species 0.000 description 1
- 229910019142 PO4 Inorganic materials 0.000 description 1
- OAICVXFJPJFONN-UHFFFAOYSA-N Phosphorus Chemical compound [P] OAICVXFJPJFONN-UHFFFAOYSA-N 0.000 description 1
- 229920002367 Polyisobutene Polymers 0.000 description 1
- GLOYGJPNNKTDIG-UHFFFAOYSA-N SC=1N=NSC=1S Chemical class SC=1N=NSC=1S GLOYGJPNNKTDIG-UHFFFAOYSA-N 0.000 description 1
- BUGBHKTXTAQXES-UHFFFAOYSA-N Selenium Chemical compound [Se] BUGBHKTXTAQXES-UHFFFAOYSA-N 0.000 description 1
- BQCADISMDOOEFD-UHFFFAOYSA-N Silver Chemical compound [Ag] BQCADISMDOOEFD-UHFFFAOYSA-N 0.000 description 1
- 239000005864 Sulphur Substances 0.000 description 1
- 238000003917 TEM image Methods 0.000 description 1
- ATJFFYVFTNAWJD-UHFFFAOYSA-N Tin Chemical compound [Sn] ATJFFYVFTNAWJD-UHFFFAOYSA-N 0.000 description 1
- XYRMLECORMNZEY-UHFFFAOYSA-B [Mo+4].[Mo+4].[Mo+4].[O-]P([O-])([S-])=S.[O-]P([O-])([S-])=S.[O-]P([O-])([S-])=S.[O-]P([O-])([S-])=S Chemical class [Mo+4].[Mo+4].[Mo+4].[O-]P([O-])([S-])=S.[O-]P([O-])([S-])=S.[O-]P([O-])([S-])=S.[O-]P([O-])([S-])=S XYRMLECORMNZEY-UHFFFAOYSA-B 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 239000004480 active ingredient Substances 0.000 description 1
- 239000011149 active material Substances 0.000 description 1
- 239000008186 active pharmaceutical agent Substances 0.000 description 1
- 150000001298 alcohols Chemical class 0.000 description 1
- 229910052782 aluminium Inorganic materials 0.000 description 1
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 1
- 229910052785 arsenic Inorganic materials 0.000 description 1
- RQNWIZPPADIBDY-UHFFFAOYSA-N arsenic atom Chemical compound [As] RQNWIZPPADIBDY-UHFFFAOYSA-N 0.000 description 1
- SGQLUUBYYBASTD-UHFFFAOYSA-N azanium butoxy-butylsulfanyl-oxido-sulfanylidene-lambda5-phosphane Chemical class [NH4+].CCCCOP([O-])(=S)SCCCC SGQLUUBYYBASTD-UHFFFAOYSA-N 0.000 description 1
- RNGGCNUUKRUGCI-UHFFFAOYSA-N azanium;ethoxy-ethylsulfanyl-oxido-sulfanylidene-$l^{5}-phosphane Chemical class [NH4+].CCOP([O-])(=S)SCC RNGGCNUUKRUGCI-UHFFFAOYSA-N 0.000 description 1
- ATBPGPNOOVIFLN-UHFFFAOYSA-N azanium;methoxy-methylsulfanyl-oxido-sulfanylidene-$l^{5}-phosphane Chemical class [NH4+].COP([O-])(=S)SC ATBPGPNOOVIFLN-UHFFFAOYSA-N 0.000 description 1
- 239000010953 base metal Substances 0.000 description 1
- 229910052797 bismuth Inorganic materials 0.000 description 1
- JCXGWMGPZLAOME-UHFFFAOYSA-N bismuth atom Chemical compound [Bi] JCXGWMGPZLAOME-UHFFFAOYSA-N 0.000 description 1
- 229910052793 cadmium Inorganic materials 0.000 description 1
- BDOSMKKIYDKNTQ-UHFFFAOYSA-N cadmium atom Chemical compound [Cd] BDOSMKKIYDKNTQ-UHFFFAOYSA-N 0.000 description 1
- 229910052791 calcium Inorganic materials 0.000 description 1
- 239000011575 calcium Substances 0.000 description 1
- 239000003575 carbonaceous material Substances 0.000 description 1
- 150000001735 carboxylic acids Chemical class 0.000 description 1
- 238000004517 catalytic hydrocracking Methods 0.000 description 1
- 229910052804 chromium Inorganic materials 0.000 description 1
- 239000011651 chromium Substances 0.000 description 1
- 229910017052 cobalt Inorganic materials 0.000 description 1
- 239000010941 cobalt Substances 0.000 description 1
- GUTLYIVDDKVIGB-UHFFFAOYSA-N cobalt atom Chemical compound [Co] GUTLYIVDDKVIGB-UHFFFAOYSA-N 0.000 description 1
- 230000007797 corrosion Effects 0.000 description 1
- 239000010779 crude oil Substances 0.000 description 1
- 230000001627 detrimental effect Effects 0.000 description 1
- DMBHHRLKUKUOEG-UHFFFAOYSA-N diphenylamine Chemical class C=1C=CC=CC=1NC1=CC=CC=C1 DMBHHRLKUKUOEG-UHFFFAOYSA-N 0.000 description 1
- 239000006185 dispersion Substances 0.000 description 1
- 239000012990 dithiocarbamate Substances 0.000 description 1
- 150000004659 dithiocarbamates Chemical class 0.000 description 1
- 230000000694 effects Effects 0.000 description 1
- 238000001493 electron microscopy Methods 0.000 description 1
- 239000000839 emulsion Substances 0.000 description 1
- 239000003344 environmental pollutant Substances 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- 150000004665 fatty acids Chemical class 0.000 description 1
- 150000002191 fatty alcohols Chemical class 0.000 description 1
- 238000009472 formulation Methods 0.000 description 1
- 229910002804 graphite Inorganic materials 0.000 description 1
- 239000010439 graphite Substances 0.000 description 1
- 229910052739 hydrogen Inorganic materials 0.000 description 1
- 239000001257 hydrogen Substances 0.000 description 1
- 125000004435 hydrogen atom Chemical group [H]* 0.000 description 1
- 230000006872 improvement Effects 0.000 description 1
- 239000012535 impurity Substances 0.000 description 1
- 230000001939 inductive effect Effects 0.000 description 1
- 239000003112 inhibitor Substances 0.000 description 1
- 239000003446 ligand Substances 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- 239000011777 magnesium Substances 0.000 description 1
- 229910052748 manganese Inorganic materials 0.000 description 1
- 239000011572 manganese Substances 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- QSHDDOUJBYECFT-UHFFFAOYSA-N mercury Chemical compound [Hg] QSHDDOUJBYECFT-UHFFFAOYSA-N 0.000 description 1
- 229910052753 mercury Inorganic materials 0.000 description 1
- 239000013528 metallic particle Substances 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- 239000000178 monomer Substances 0.000 description 1
- AFFLGGQVNFXPEV-UHFFFAOYSA-N n-decene Natural products CCCCCCCCC=C AFFLGGQVNFXPEV-UHFFFAOYSA-N 0.000 description 1
- 230000007935 neutral effect Effects 0.000 description 1
- 229910052759 nickel Inorganic materials 0.000 description 1
- 150000002902 organometallic compounds Chemical class 0.000 description 1
- 239000002530 phenolic antioxidant Substances 0.000 description 1
- 235000021317 phosphate Nutrition 0.000 description 1
- 239000011574 phosphorus Substances 0.000 description 1
- 231100000719 pollutant Toxicity 0.000 description 1
- 229920001083 polybutene Polymers 0.000 description 1
- 229920000193 polymethacrylate Polymers 0.000 description 1
- 229920005862 polyol Polymers 0.000 description 1
- 229920000098 polyolefin Polymers 0.000 description 1
- 150000003077 polyols Chemical class 0.000 description 1
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 1
- 238000007670 refining Methods 0.000 description 1
- 239000013049 sediment Substances 0.000 description 1
- 229910052711 selenium Inorganic materials 0.000 description 1
- 239000011669 selenium Substances 0.000 description 1
- 229910052709 silver Inorganic materials 0.000 description 1
- 239000004332 silver Substances 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 239000002904 solvent Substances 0.000 description 1
- 238000000638 solvent extraction Methods 0.000 description 1
- 125000003011 styrenyl group Chemical group [H]\C(*)=C(/[H])C1=C([H])C([H])=C([H])C([H])=C1[H] 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 238000001308 synthesis method Methods 0.000 description 1
- 238000003786 synthesis reaction Methods 0.000 description 1
- 229910052716 thallium Inorganic materials 0.000 description 1
- BKVIYDNLLOSFOA-UHFFFAOYSA-N thallium Chemical compound [Tl] BKVIYDNLLOSFOA-UHFFFAOYSA-N 0.000 description 1
- 230000007704 transition Effects 0.000 description 1
- 238000005292 vacuum distillation Methods 0.000 description 1
- 235000013311 vegetables Nutrition 0.000 description 1
- 238000010792 warming Methods 0.000 description 1
- 239000004711 α-olefin Substances 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M141/00—Lubricating compositions characterised by the additive being a mixture of two or more compounds covered by more than one of the main groups C10M125/00 - C10M139/00, each of these compounds being essential
- C10M141/12—Lubricating compositions characterised by the additive being a mixture of two or more compounds covered by more than one of the main groups C10M125/00 - C10M139/00, each of these compounds being essential at least one of them being an organic compound containing atoms of elements not provided for in groups C10M141/02 - C10M141/10
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M171/00—Lubricating compositions characterised by purely physical criteria, e.g. containing as base-material, thickener or additive, ingredients which are characterised exclusively by their numerically specified physical properties, i.e. containing ingredients which are physically well-defined but for which the chemical nature is either unspecified or only very vaguely indicated
- C10M171/06—Particles of special shape or size
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M2201/00—Inorganic compounds or elements as ingredients in lubricant compositions
- C10M2201/06—Metal compounds
- C10M2201/065—Sulfides; Selenides; Tellurides
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M2201/00—Inorganic compounds or elements as ingredients in lubricant compositions
- C10M2201/06—Metal compounds
- C10M2201/065—Sulfides; Selenides; Tellurides
- C10M2201/066—Molybdenum sulfide
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M2203/00—Organic non-macromolecular hydrocarbon compounds and hydrocarbon fractions as ingredients in lubricant compositions
- C10M2203/02—Well-defined aliphatic compounds
- C10M2203/024—Well-defined aliphatic compounds unsaturated
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M2205/00—Organic macromolecular hydrocarbon compounds or fractions, whether or not modified by oxidation as ingredients in lubricant compositions
- C10M2205/02—Organic macromolecular hydrocarbon compounds or fractions, whether or not modified by oxidation as ingredients in lubricant compositions containing acyclic monomers
- C10M2205/028—Organic macromolecular hydrocarbon compounds or fractions, whether or not modified by oxidation as ingredients in lubricant compositions containing acyclic monomers containing aliphatic monomers having more than four carbon atoms
- C10M2205/0285—Organic macromolecular hydrocarbon compounds or fractions, whether or not modified by oxidation as ingredients in lubricant compositions containing acyclic monomers containing aliphatic monomers having more than four carbon atoms used as base material
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M2223/00—Organic non-macromolecular compounds containing phosphorus as ingredients in lubricant compositions
- C10M2223/02—Organic non-macromolecular compounds containing phosphorus as ingredients in lubricant compositions having no phosphorus-to-carbon bonds
- C10M2223/04—Phosphate esters
- C10M2223/043—Ammonium or amine salts thereof
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M2223/00—Organic non-macromolecular compounds containing phosphorus as ingredients in lubricant compositions
- C10M2223/02—Organic non-macromolecular compounds containing phosphorus as ingredients in lubricant compositions having no phosphorus-to-carbon bonds
- C10M2223/04—Phosphate esters
- C10M2223/045—Metal containing thio derivatives
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10M—LUBRICATING COMPOSITIONS; USE OF CHEMICAL SUBSTANCES EITHER ALONE OR AS LUBRICATING INGREDIENTS IN A LUBRICATING COMPOSITION
- C10M2223/00—Organic non-macromolecular compounds containing phosphorus as ingredients in lubricant compositions
- C10M2223/02—Organic non-macromolecular compounds containing phosphorus as ingredients in lubricant compositions having no phosphorus-to-carbon bonds
- C10M2223/04—Phosphate esters
- C10M2223/047—Thioderivatives not containing metallic elements
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10N—INDEXING SCHEME ASSOCIATED WITH SUBCLASS C10M RELATING TO LUBRICATING COMPOSITIONS
- C10N2020/00—Specified physical or chemical properties or characteristics, i.e. function, of component of lubricating compositions
- C10N2020/01—Physico-chemical properties
- C10N2020/055—Particles related characteristics
- C10N2020/06—Particles of special shape or size
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10N—INDEXING SCHEME ASSOCIATED WITH SUBCLASS C10M RELATING TO LUBRICATING COMPOSITIONS
- C10N2030/00—Specified physical or chemical properties which is improved by the additive characterising the lubricating composition, e.g. multifunctional additives
- C10N2030/02—Pour-point; Viscosity index
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10N—INDEXING SCHEME ASSOCIATED WITH SUBCLASS C10M RELATING TO LUBRICATING COMPOSITIONS
- C10N2030/00—Specified physical or chemical properties which is improved by the additive characterising the lubricating composition, e.g. multifunctional additives
- C10N2030/06—Oiliness; Film-strength; Anti-wear; Resistance to extreme pressure
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10N—INDEXING SCHEME ASSOCIATED WITH SUBCLASS C10M RELATING TO LUBRICATING COMPOSITIONS
- C10N2030/00—Specified physical or chemical properties which is improved by the additive characterising the lubricating composition, e.g. multifunctional additives
- C10N2030/10—Inhibition of oxidation, e.g. anti-oxidants
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10N—INDEXING SCHEME ASSOCIATED WITH SUBCLASS C10M RELATING TO LUBRICATING COMPOSITIONS
- C10N2030/00—Specified physical or chemical properties which is improved by the additive characterising the lubricating composition, e.g. multifunctional additives
- C10N2030/12—Inhibition of corrosion, e.g. anti-rust agents or anti-corrosives
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10N—INDEXING SCHEME ASSOCIATED WITH SUBCLASS C10M RELATING TO LUBRICATING COMPOSITIONS
- C10N2030/00—Specified physical or chemical properties which is improved by the additive characterising the lubricating composition, e.g. multifunctional additives
- C10N2030/54—Fuel economy
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10N—INDEXING SCHEME ASSOCIATED WITH SUBCLASS C10M RELATING TO LUBRICATING COMPOSITIONS
- C10N2040/00—Specified use or application for which the lubricating composition is intended
- C10N2040/04—Oil-bath; Gear-boxes; Automatic transmissions; Traction drives
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10N—INDEXING SCHEME ASSOCIATED WITH SUBCLASS C10M RELATING TO LUBRICATING COMPOSITIONS
- C10N2040/00—Specified use or application for which the lubricating composition is intended
- C10N2040/04—Oil-bath; Gear-boxes; Automatic transmissions; Traction drives
- C10N2040/044—Oil-bath; Gear-boxes; Automatic transmissions; Traction drives for manual transmissions
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10N—INDEXING SCHEME ASSOCIATED WITH SUBCLASS C10M RELATING TO LUBRICATING COMPOSITIONS
- C10N2050/00—Form in which the lubricant is applied to the material being lubricated
- C10N2050/015—Dispersions of solid lubricants
Definitions
- the present invention is applicable to the field of lubricants, and more particularly to the field of lubricants for motor vehicles.
- the invention relates to a lubricating composition comprising metallic nanoparticles. More particularly, the invention relates to a lubricating composition comprising an anti-wear additive and metallic nanoparticles.
- the lubricating composition according to the invention simultaneously exhibits good stability as well as good friction properties which last over time.
- additive concentrate type composition comprising an anti-wear additive and metallic nanoparticles.
- the level of friction can be adjusted by adding friction modifiers to these gear oils.
- the improvement in the energy performance of the lubricating compositions can be obtained in particular by mixing friction modifiers in the base oils.
- organometallic compounds comprising molybdenum are commonly used. In order to obtain good friction reduction properties, a sufficient quantity of molybdenum must be present within the lubricating composition.
- these compounds have the drawback of inducing the formation of sediments when the lubricating composition has too high a content of molybdenum element.
- the poor solubility of these compounds modifies or even deteriorates the properties of the lubricating composition, in particular its viscosity.
- a composition that is too viscous or not viscous enough is detrimental to the movement of moving parts, to the correct starting of an engine, to the protection of an engine when it has reached its operating temperature, and therefore ultimately causes in particular an increase in fuel consumption.
- these compounds contribute to an increase in the ash content, reducing their potential for use in a lubricating composition, in particular in Europe.
- compositions comprising organomolybdenum-type friction modifier compounds with organophosphorus and/or organosulfur and/or organophosphorus anti-wear and extreme pressure compounds, in particular to improve the anti-wear properties of these oils. engines or transmissions.
- the document CN 101691517 describes an engine oil comprising nanoparticles of tungsten disulphide, making it possible to improve engine life and reduce fuel consumption.
- the content of tungsten disulphide nanoparticles ranges from 15 to 34%, which can lead to risks of instability of the oil over time.
- FR 2 910 911 discloses a lubricant composition
- a lubricant composition comprising a base oil and metal-containing nanoparticles, the particles having an average diameter of 1 to 10 nm.
- a lubricating composition in particular for motor vehicles, which is not a grease while exhibiting good properties for reducing friction and retaining satisfactory anti-flaking properties.
- One objective of the present invention is to provide a lubricating composition which overcomes all or part of the aforementioned drawbacks.
- Another objective of the invention is to provide a lubricating composition which is stable and easy to implement.
- Another object of the present invention is to provide a method of lubrication which makes it possible in particular to reduce friction on the surface of mechanical parts, and more particularly of an engine or a transmission member of motor vehicles.
- the metallic nanoparticles have an average size, determined using images obtained by transmission electron micrography or by high-resolution transmission electron microscopy, of 50 to 200 nm and are concentric polyhedra with a multilayer or sheet structure.
- an anti-wear compound comprising a dithiophosphate group in a lubricating composition comprising at least one base oil and metallic nanoparticles as defined above makes it possible to confer on said composition very good friction reducing properties.
- an anti-wear compound comprising a dithiophosphate group and metallic nanoparticles as defined above in a lubricating composition makes it possible to maintain this reduction in friction over time.
- this maintenance over time of the effectiveness of friction reduction could be explained by the protection against the oxidation of the metallic nanoparticles by the anti-wear compound comprising a dithiophosphate group, thus extending the action of metallic nanoparticles on the surface of a mechanical part, and more particularly of a transmission member or motor vehicle engine.
- the present invention makes it possible to formulate stable lubricating compositions comprising a reduced content of metallic nanoparticles and yet exhibiting remarkable friction reduction properties.
- the lubricating compositions according to the invention have remarkable friction reduction properties which last over time.
- the lubricating compositions according to the invention have good oil stability and a viscosity which does not vary or varies very little.
- the lubricating compositions according to the invention have satisfactory anti-flaking properties.
- the lubricating compositions according to the invention have a reduced risk of oxidation.
- the lubricating compositions according to the invention have remarkable fuel saving properties
- the lubricating composition consists essentially of at least one base oil, at least one anti-wear compound comprising a dithiophosphate group and at least metallic nanoparticles as defined above in a content by weight ranging from 0 0.01 to 2% relative to the total weight of the lubricating composition.
- the application also describes an engine oil comprising a lubricating composition as defined above.
- the application also describes a transmission oil comprising a lubricating composition as defined above.
- the invention also relates to the use of a lubricating composition as defined above for lubricating a mechanical part, preferably a transmission member or a vehicle engine, advantageously motor vehicles.
- the invention also relates to the use of a lubricating composition as defined above for reducing friction on the surface of a mechanical part, preferably a transmission member or a vehicle engine, advantageously motor vehicles.
- the invention also relates to the use of a lubricating composition as defined above for reducing the fuel consumption of vehicles, in particular motor vehicles.
- the application also describes a process for lubricating a mechanical part, preferably a transmission member or a vehicle engine, advantageously motor vehicles, said process comprising at least one step of bringing the mechanical part into contact with a lubricating composition as defined above.
- the application also describes a method for reducing friction on the surface of a mechanical part, preferably of a transmission member or of a vehicle engine, advantageously of motor vehicles, comprising at least bringing the mechanical part into contact with a lubricating composition as defined above.
- the application also describes a method for reducing the fuel consumption of a vehicle, in particular of a motor vehicle, comprising at least one step of bringing a mechanical part of the engine of the vehicle into contact with a lubricating composition as defined above.
- the invention also relates to the use of an anti-wear compound comprising a dithiophosphate group to reduce the oxidation of a lubricating composition
- composition of the concentrate type of additives comprising at least at least one compound comprising a dithiophosphate group and tungsten disulphide nanoparticles.
- the lubricating composition according to the invention comprises metallic nanoparticles in a content by weight ranging from 0.01 to 2% relative to the total weight of the lubricating composition.
- metallic nanoparticles in particular metallic particles, generally solid, whose average size is less than or equal to 600 nm.
- the metallic nanoparticles consist of at least 80% by mass of at least one metal, or else of at least 80% by mass of at least one metal alloy or else of at least 80% by mass of at least one metal chalcogenide, in particular of transition metal, with respect to the total mass of the nanoparticle.
- the metallic nanoparticles consist of at least 90% by mass of at least one metal, or else of at least 90% by mass of at least one metal alloy or else of at least 90% by mass of at least one metal chalcogenide, in particular of transition metal, with respect to the total mass of the nanoparticle.
- the metallic nanoparticles consist of at least 99% by mass of at least one metal, or else of at least 99% by mass of at least one metal alloy or else of at least 99% by mass of at least one metal chalcogenide, in particular of transition metal, with respect to the total mass of the nanoparticle, the remaining 1% being constituted by impurities.
- the metal of which the metallic nanoparticle is made can be chosen from the group formed by tungsten, molybdenum, zirconium, hafnium, platinum, rhenium, titanium, tantalum and niobium, preferably molybdenum or tungsten, advantageously tungsten.
- M is chosen from the group formed by tungsten, molybdenum, zirconium, hafnium, platinum, rhenium, titanium, tantalum and niobium.
- M is selected from the group consisting of molybdenum and tungsten.
- M is tungsten
- X is chosen from the group formed by oxygen, sulfur, selenium and tellurium.
- X is chosen from sulfur or tellurium.
- X is sulfur
- the metallic nanoparticles according to the invention are chosen from the group formed by MoS2, MoSe 2 , MoTe 2 , WS 2 , WSe 2 , ZrS 2 , ZrSe 2 , HfS 2 , HfSe 2 , PtS 2 , ReS 2 , ReSe 2 , TiS 3 , ZrS 3 , ZrSe 3 , HfS 3 , HfSe 3 , TiS 2 , TaS 2 , TaSe 2 , NbS 2 , NbSe 2 and NbTe 2 .
- the metallic nanoparticles according to the invention are chosen from the group formed by WS 2 , WSe 2 , MoS 2 and MoSe 2 , preferentially WS 2 and MoS 2 , preferentially WS 2 .
- the nanoparticles according to the invention have a fullerene-type structure.
- fullerene designates a closed convex polyhedron nanostructure, composed of carbon atoms.
- Fullerenes are similar to graphite, consisting of sheets of bonded hexagonal rings, but they contain pentagonal, and sometimes heptagonal, rings that prevent the structure from being flat.
- fullerene-like structures are not limited to carbonaceous materials, but is likely to occur in all nanoparticles of materials in the form of sheets, especially for nanoparticles comprising chalcogens and metals of transition.
- These structures are analogous to that of carbon fullerenes and are called inorganic fullerenes or fullerene-like structures (in English terms “Inorganic Fullerene like materials”, also referred to as “IF”).
- Fullerene-like structures are described in particular by Tenne, R., Margulis, L., Genut M. Hodes, G. Nature 1992, 360, 444 .
- the document EP 0580 019 describes in particular these structures and their method of synthesis.
- the metallic nanoparticles are closed structures, of the spherical type, more or less perfect depending on the synthesis methods used.
- the nanoparticles according to the invention are concentric polyhedrons with a multilayer or sheet structure. We speak of an "onion” or “nested polyhedron” structure.
- the metallic nanoparticles are multilayer metallic nanoparticles comprising from 2 to 500 layers, preferably from 20 to 200 layers, advantageously from 20 to 100 layers.
- the average size of the metallic nanoparticles according to the invention ranges from 50 to 200 nm.
- the size of the metallic nanoparticles according to the invention can be determined using images obtained by transmission electron micrography or by electron microscopy. high resolution transmission.
- the average particle size can be determined from the measurement of the size of at least 50 solid particles visualized on transmission electron micrographs.
- the median value of the measured size distribution histogram of the solid particles is the average size of the solid particles used in the lubricating composition according to the invention.
- the content by weight of metallic nanoparticles ranges from 0.05 to 2%, preferably from 0.1 to 1%, advantageously from 0.1 to 0.5% relative to the total weight of the lubricating composition.
- NanoLub Gear Oil Concentrate marketed by the company Nanomaterials, which is in the form of a dispersion of multilayer nanoparticles of tungsten disulphide in a mineral or PAO type oil ( Poly Alfa Olefin).
- the lubricating composition according to the invention comprises at least one anti-wear compound comprising a dithiophosphate group.
- the anti-wear compound comprising a dithiophosphate group is called “dithiophosphate” in the rest of the present description.
- the dithiophosphate can be chosen from ammonium dithiophosphates, amine dithiophosphates, ester dithiophosphates and metal dithiophosphates, taken alone or as a mixture.
- the dithiophosphate is chosen from the ammonium dithiophosphates of formula (I): in which R1 and R2 represent, independently of each other, an optionally substituted hydrocarbon group comprising from 1 to 30 carbon atoms.
- R1 and R2 independently of each other represent a hydrocarbon group, optionally substituted, comprising from 2 to 24 carbon atoms, more preferentially from 3 to 18 carbon atoms, advantageously from 5 to 12 carbon atoms.
- R1 and R2 independently represent an unsubstituted hydrocarbon group, said hydrocarbon group possibly being an alkyl, alkenyl, alkynyl, phenyl or benzyl group.
- R1 and R2 independently represent a linear or branched alkyl hydrocarbon group, more preferably a linear alkyl hydrocarbon group.
- R1 and R2 independently represent a hydrocarbon group optionally substituted by at least one oxygen, nitrogen, sulfur and/or phosphorus atom, preferably by at least one oxygen atom.
- ammonium dithiophosphate mention may be made of ammonium dimethyl dithiophosphates, ammonium diethyl dithiophosphates and ammonium dibutyl dithiophosphates.
- R3 and R4 independently of each other represent a hydrocarbon group, optionally substituted, comprising from 2 to 24 carbon atoms, more preferentially from 3 to 18 carbon atoms, advantageously from 5 to 12 carbon atoms.
- R3 and R4 independently represent an unsubstituted hydrocarbon group, said hydrocarbon group possibly being an alkyl, alkenyl, alkynyl, phenyl or benzyl group.
- R3 and R4 independently represent a linear or branched alkyl hydrocarbon group, more preferably a linear alkyl hydrocarbon group.
- R3 and R4 independently represent a hydrocarbon group optionally substituted by at least one oxygen, nitrogen, sulfur and/or phosphorus atom, preferably by at least one oxygen atom.
- R5, R6 and R7 independently represent a hydrocarbon group comprising from 2 to 24 carbon atoms, more preferably from 3 to 18 carbon atoms, advantageously from 5 to 12 carbon atoms.
- R8 and R9 independently represent an optionally substituted hydrocarbon group comprising from 2 to 24 carbon atoms, more preferentially from 3 to 18 carbon atoms, advantageously from 5 to 12 carbon atoms.
- R8 and R9 independently represent an unsubstituted hydrocarbon group, said hydrocarbon group possibly being an alkyl, alkenyl, alkynyl, phenyl or benzyl group.
- R8 and R9 independently represent a linear or branched alkyl hydrocarbon group, more preferably a linear alkyl hydrocarbon group.
- R8 and R9 independently represent a hydrocarbon group optionally substituted by at least one oxygen, nitrogen, sulfur and/or phosphorus atom, preferably by at least one oxygen atom.
- R8 and R9 represent, independently of each other, a hydrocarbon group comprising from 2 to 6 carbon atoms.
- R10 and R11 independently represent a hydrocarbon group comprising from 2 to 6 carbon atoms.
- the metal is selected from the group consisting of zinc, aluminum, copper, iron, mercury, silver, cadmium, tin, lead, antimony, bismuth, thallium, chromium, molybdenum, cobalt, nickel, tungsten, sodium, calcium, magnesium, manganese and arsenic.
- Preferred metals are zinc, molybdenum, antimony, preferably zinc and molybdenum.
- the metal is zinc.
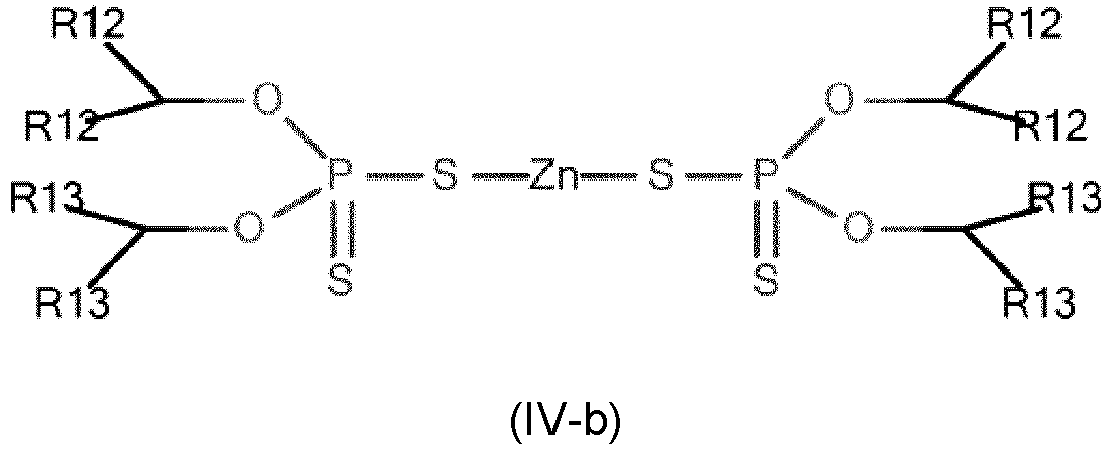
- Metal dithiophosphates are neutral as exemplified in formula (IV) or basic when a stoichiometric excess of metal is present.
- R12 and R13 independently represent an optionally substituted hydrocarbon group comprising from 2 to 24 carbon atoms, more preferentially from 3 to 18 carbon atoms, advantageously from 5 to 12 carbon atoms.
- R12 and R13 independently represent an unsubstituted hydrocarbon group, said hydrocarbon group possibly being an alkyl, alkenyl, alkynyl, phenyl or benzyl group.
- R12 and R13 independently represent a linear or branched alkyl hydrocarbon group, more preferably a linear alkyl hydrocarbon group.
- R12 and R13 independently represent a hydrocarbon group optionally substituted by at least one oxygen, nitrogen, sulfur and/or phosphorus atom, preferably by at least one oxygen atom.
- the dithiophosphate according to the invention is a zinc dithiophosphate of formula (IV-a) or of formula (IV-b): wherein R12 and R13 are as defined above.
- metal dithiophosphate As metal dithiophosphate according to the invention, one can quote for example Additin ® RC 3038, Additin ® RC 3045, Additin ® RC 3048, Additin ® RC 3058, Additin° RC 3080, Additin° RC 3180, Additin° RC 3212, Additin° RC 3580, Kikulube° Z112, Lubrizol ® 1371, Lubrizol ® 1375, Lubrizol ® 1395, Lubrizol ® 5179, Oloa ® 260, Oloa® 267.
- the content by weight of anti-wear compound comprising a dithiophosphate group ranges from 0.1 to 5%, relative to the total weight of the lubricating composition.
- the content by weight of compound comprising a dithiophosphate group ranges from 0.2 to 4%, more preferably from 0.5 to 2%, advantageously from 0.5 to 1.5% per relative to the total weight of the lubricating composition.
- the lubricating compositions according to the invention can contain any type of mineral, synthetic or natural, animal or vegetable lubricating base oil suitable for their use.
- the base oil(s) used in the lubricating compositions according to the present invention may be oils of mineral or synthetic origin of groups I to V according to the classes defined in the API classification (or their equivalents according to the ATIEL classification) as summarized below, singly or in combination.
- Table I Content Saturates content Sulfur content Viscosity index (VI) Group I Mineral Oils ⁇ 90% > 0.03% 80 ⁇ IV ⁇ 120 Group II Hydrocracked oils ⁇ 90% ⁇ 0.03% 80 ⁇ IV ⁇ 120 Group III Hydrocracked or hydroisomerized oils ⁇ 90% ⁇ 0.03% ⁇ 120 Group IV Polyalphaolefins (PAO) Group V Esters and other bases not included in bases groups I to IV
- the mineral base oils according to the invention include all types of bases obtained by atmospheric and vacuum distillation of crude oil, followed by refining operations such as solvent extraction, deasphalting, solvent dewaxing, hydrotreating, hydrocracking and hydroisomerization, hydrofinishing.
- the base oils of the lubricating compositions according to the invention can also be synthetic oils, such as certain esters of carboxylic acids and alcohols, or polyalphaolefins.
- the polyalphaolefins used as base oils are for example obtained from monomers having from 4 to 32 carbon atoms (for example octene, decene), and a viscosity at 100° C. of between 1.5 and 15 cSt measured according to the ASTM D445 standard. Their weight-average molecular weight is typically between 250 and 3000 measured according to standard ASTM D5296. Blends of synthetic and mineral oils can also be used.
- a lubricating base for producing the lubricating compositions according to the invention there is no limitation as to the use of such and such a lubricating base for producing the lubricating compositions according to the invention, except that they must have properties, in particular viscosity, viscosity index, sulphur, resistance to oxidation, suitable for use in a gearbox, in particular in a motor vehicle gearbox, in particular in a manual gearbox.
- the lubricating bases represent at least 70% by mass, relative to the total mass of the lubricating composition, Typically, they represent between 75 and 99.9% by mass, relative to the total mass of the lubricating compositions according to the invention.
- the lubricating composition according to the invention has a kinematic viscosity at 100° C. measured according to the ASTM D445 standard ranging from 4 to 50 cSt.
- the kinematic viscosity at 100° C. measured according to the ASTM D445 standard of the composition according to the invention ranges from 4 to 45 cSt, preferably from 4 to 30 cSt.
- the lubricating compositions comprise at least one Group IV base.
- the lubricating compositions have a viscosity index (VI) greater than 95 (ASTM 2270 standard).
- the lubricating compositions according to the invention may also contain any type of additive suitable for their use in the formulations of oils for transmissions, for example one or more additives chosen from polymers, antioxidants, anti-corrosion additives, modifiers different frictions of the metallic nanoparticles according to the invention and the dispersants, present at the usual levels required for the application.
- the additive is chosen from dispersants having a weight-average molecular mass greater than or equal to 2000 Daltons.
- the weight-average molecular mass of the dispersant is evaluated according to the ASTM D5296 standard.
- dispersant within the meaning of the present invention, is meant more particularly any compound which improves the maintenance in suspension of the metallic nanoparticles.
- the dispersant can be chosen from compounds comprising at least one succinimide group, polyolefins, olefin copolymers (OCP), copolymers comprising at least one styrene unit, polyacrylates or their derivatives. .
- derivatives any compound comprising at least one group or one polymeric chain as defined above.
- the dispersant according to the invention is chosen from compounds comprising at least one succinimide group.
- the dispersant is chosen from compounds comprising at least one substituted succinimide group or compounds comprising at least two substituted succinimide groups, the succinimide groups being linked at their top bearing a d atom. nitrogen by a polyamine group.
- substituted succinimide group within the meaning of the present invention, is meant a succinimide group of which at least one of the carbon vertices is substituted by a hydrocarbon group comprising from 8 to 400 carbon atoms.
- the dispersant is chosen from polyisobutylene succinimide-polyamine
- the dispersant according to the invention has a weight-average molecular mass ranging from 2000 to 15000 Daltons, preferably ranging from 2500 to 10000 Daltons, advantageously from 3000 to 7000 Daltons.
- the dispersant has a number molecular mass greater than or equal to 1000 Daltons, preferably ranging from 1000 to 5000 Daltons, more preferably from 1800 to 3500 Daltons, advantageously from 1800 to 3000 Daltons.
- the number molecular weight of the dispersant is evaluated according to the ASTM D5296 standard.
- the content by weight of dispersant having a weight average molecular mass greater than or equal to 2000 Daltons ranges from 0.1 to 10%, preferably from 0.1 to 5%, advantageously from 0.1 to 3% relative to the total weight of the lubricating composition.
- the polymers can be chosen from the group of shear-stable polymers, preferably from the group consisting of copolymers of ethylene and alpha-olefin, polyacrylates such as polymethacrylates, olefin copolymers (OCP), Ethylene Propylene Diene Monomers (EPDM), polybutenes, copolymers of styrene and olefin, hydrogenated or not, or copolymers of styrene and acrylate.
- polyacrylates such as polymethacrylates, olefin copolymers (OCP), Ethylene Propylene Diene Monomers (EPDM), polybutenes, copolymers of styrene and olefin, hydrogenated or not, or copolymers of styrene and acrylate.
- the antioxidants can be chosen from amino antioxidants, preferably diphenylamines, in particular dialkylphenylamines, such as octadiphenylamines, phenyl-alpha-naphthyl amines, phenolic antioxidants (dibutylhydroxytoluene BHT and derivatives) or sulfur antioxidants (sulphurized phenates) .
- amino antioxidants preferably diphenylamines, in particular dialkylphenylamines, such as octadiphenylamines, phenyl-alpha-naphthyl amines, phenolic antioxidants (dibutylhydroxytoluene BHT and derivatives) or sulfur antioxidants (sulphurized phenates) .
- the friction modifiers can be compounds providing metallic elements different from the metallic nanoparticles according to the invention or else an ashless compound.
- compounds providing metallic elements mention may be made of complexes of transition metals such as Mo, Sb, Sn, Fe, Cu, Zn, the ligands of which may be hydrocarbon compounds containing oxygen, nitrogen, sulfur or phosphorus, such as dithiocarbamates or molybdenum dithiophosphates.
- the ashless friction modifiers are of organic origin and can be chosen from monoesters of fatty acids and polyols, alkoxylated amines, fatty alkoxylated amines, amine phosphates, fatty alcohols, fatty epoxides, borate fatty epoxides, fatty amines or fatty acid glycerol esters.
- fat or “fat(s)” is meant within the meaning of the present invention a hydrocarbon group comprising from 8 to 24 carbon atoms.
- the anti-corrosion additives can be chosen from phenolic derivatives, in particular ethoxylated phenolic derivatives substituted by alkyl groups in the ortho position.
- the corrosion inhibitors may be derivatives of dimercaptothiadiazole.
- the lubricating composition is not an emulsion.
- the lubricating composition is anhydrous.
- the application also describes an engine oil comprising a lubricating composition according to the invention.
- the application also describes a transmission oil comprising a lubricating composition according to the invention.
- the lubricating composition according to the invention can lubricate at least one mechanical part or one mechanical component, in particular bearings, gears, universal joints, transmissions, the piston/segment/liner system, the camshafts, the clutch , manual or automatic gearboxes, axles, rocker arms, crankcases, etc.
- the lubricating composition according to the invention can lubricate a mechanical part or a metal component of the transmissions, of the clutch, of the axles, of manual or automatic, preferably manual, gearboxes.
- the invention also relates to the use of a lubricating composition as defined above for lubricating a mechanical part, preferably a transmission member or a vehicle engine, advantageously vehicles automobiles.
- the invention also relates to the use of a lubricating composition as defined above for reducing friction on the surface of a mechanical part, preferably a transmission member or a vehicle engine, advantageously of motor vehicles.
- a subject of the invention is also the use of a lubricating composition as defined above for reducing the fuel consumption of vehicles, in particular motor vehicles.
- a subject of the invention is also the use of a lubricating composition as defined above for reducing the spalling of a mechanical part, preferably of a transmission member or of a vehicle engine, advantageously of vehicles automobiles.
- the application also describes a process for lubricating a mechanical part, preferably a transmission member or a vehicle engine, advantageously motor vehicles, said process comprising at least one step of bringing the mechanical part into contact with a lubricating composition as defined above.
- the application also describes a method for reducing friction on the surface of a mechanical part, preferably of a transmission member or of a vehicle engine, advantageously of motor vehicles, comprising at least bringing the mechanical part into contact with a lubricating composition as defined above.
- the application also describes a method for reducing the fuel consumption of a vehicle, in particular of a motor vehicle comprising at least one step of in contact with a mechanical part of the vehicle engine with a lubricating composition as defined above.
- the application also describes a method for reducing the spalling of a mechanical part, preferably of a transmission member or of a vehicle engine, advantageously of motor vehicles, comprising at least bringing the mechanical part into contact with a lubricating composition as defined above.
- the application also describes a composition of the concentrate type of additives comprising at least one anti-wear compound comprising a dithiophosphate group and tungsten disulphide nanoparticles.
- composition of the concentrate type of additives can be added at least one base oil to obtain a lubricating composition according to the invention.
- the invention also relates to the use of an anti-wear compound comprising a dithiophosphate group to reduce the oxidation of a lubricating composition
- Lubricating compositions No. 1 to No. 4 are described in Table II; the percentages indicated are percentages by mass.
- Table II Lubricant composition #1 #2 #3 #4 base oil 100 99 99 98 Compound comprising a dithiophosphate group 1 1 Tungsten Disulfide Nanoparticles (NanoLub Gear Oil Concentrate) 1 1
- Test 1 evaluation of the friction properties of lubricating compositions
- Table III shows the average coefficient of friction of lubricating compositions No. 1 to No. 4; the average coefficient of friction representing the average of the values of the coefficient of friction obtained after 4 tests.
- Table III Composition #1 #2 #3 #4 Coefficient of friction 0.100 0.110 0.075 0.060
- the lubricating composition according to the invention No. 4 has improved friction properties, compared to a lubricating composition comprising a compound comprising a dithiophosphate group according to the invention but does not not comprising metallic nanoparticles (composition No. 2) and relative to a composition comprising metallic nanoparticles according to the invention but not comprising a compound comprising a dithiophosphate group (composition No. 3).
- lubricating composition No. 4 exhibits satisfactory stability.
Description
La présente invention est applicable au domaine des lubrifiants, et plus particulièrement au domaine des lubrifiants pour véhicules automobiles. L'invention concerne une composition lubrifiante comprenant des nanoparticules métalliques. Plus particulièrement, l'invention concerne une composition lubrifiante comprenant un additif anti-usure et des nanoparticules métalliques. La composition lubrifiante selon l'invention présente simultanément une bonne stabilité ainsi que de bonnes propriétés de frottements et qui perdurent dans le temps.The present invention is applicable to the field of lubricants, and more particularly to the field of lubricants for motor vehicles. The invention relates to a lubricating composition comprising metallic nanoparticles. More particularly, the invention relates to a lubricating composition comprising an anti-wear additive and metallic nanoparticles. The lubricating composition according to the invention simultaneously exhibits good stability as well as good friction properties which last over time.
Décrit est également un procédé de lubrification d'une pièce mécanique mettant en œuvre cette composition lubrifiante.Also described is a process for lubricating a mechanical part using this lubricating composition.
Décrite est également une composition type concentré d'additifs comprenant un additif anti-usure et des nanoparticules métalliques.Also described is an additive concentrate type composition comprising an anti-wear additive and metallic nanoparticles.
Les organes de transmissions des véhicules automobiles fonctionnent sous forte charge et vitesses élevées. Les huiles pour ces organes de transmissions doivent donc être particulièrement performantes dans la protection des pièces contre l'usure, et notamment présenter de bonnes propriétés de réduction des frottements à la surface des organes. Ainsi, si le niveau de frottements n'est pas adapté à la géométrie des pièces, il se produit une usure sur l'ensemble cône anneau.Motor vehicle transmission components operate under heavy load and high speeds. The oils for these transmission members must therefore be particularly effective in protecting the parts against wear, and in particular have good properties for reducing friction on the surface of the members. Thus, if the level of friction is not adapted to the geometry of the parts, wear occurs on the cone-ring assembly.
Le niveau de frottement peut être ajusté par l'ajout de modificateurs de frottement dans ces huiles pour boîtes de vitesses.The level of friction can be adjusted by adding friction modifiers to these gear oils.
Par ailleurs, la généralisation de l'automobile à l'échelle planétaire depuis la fin du siècle dernier pose des problèmes quant au réchauffement climatique, à la pollution, à la sécurité et à l'utilisation des ressources naturelles, en particulier à l'épuisement des réserves de pétrole.In addition, the generalization of the automobile on a planetary scale since the end of the last century poses problems with regard to global warming, pollution, safety and the use of natural resources, in particular the exhaustion oil reserves.
Suite à l'établissement du protocole de Kyoto, de nouvelles normes protégeant l'environnement imposent à la filière de l'automobile de construire des véhicules dont les émissions polluantes et les consommations de carburant sont réduites. Il en résulte que les moteurs de ces véhicules sont soumis à des contraintes techniques de plus en plus sévères : ils tournent notamment plus vite, à des températures de plus en plus élevées et doivent consommer de moins en moins de carburant.Following the establishment of the Kyoto Protocol, new standards protecting the environment require the automotive industry to build vehicles with reduced polluting emissions and fuel consumption. As a result, the engines of these vehicles are subject to increasingly severe technical constraints. In particular, they run faster, at increasingly high temperatures and need to consume less and less fuel.
La nature des lubrifiants moteurs pour automobiles a une influence sur l'émission de polluants et sur la consommation de carburant. Des lubrifiants moteurs pour automobiles dits économiseurs d'énergie ou « fuel-eco » (en terminologie anglo-saxonne), ont été développés pour satisfaire ces nouveaux besoins.The nature of automobile engine lubricants has an influence on the emission of pollutants and on fuel consumption. Engine lubricants for automobiles known as energy-saving or “fuel-eco” (in Anglo-Saxon terminology) have been developed to meet these new needs.
L'amélioration des performances énergétiques des compositions lubrifiantes peut être obtenue notamment en mélangeant dans des huiles de base des modificateurs de frottement.The improvement in the energy performance of the lubricating compositions can be obtained in particular by mixing friction modifiers in the base oils.
Parmi les modificateurs de frottement, les composés organométalliques comprenant du molybdène sont couramment utilisés. Afin d'obtenir de bonnes propriétés de réduction des frottements, une quantité suffisante de molybdène doit être présente au sein de la composition lubrifiante.Among the friction modifiers, organometallic compounds comprising molybdenum are commonly used. In order to obtain good friction reduction properties, a sufficient quantity of molybdenum must be present within the lubricating composition.
Cependant, ces composés présentent l'inconvénient d'induire la formation de sédiments lorsque la composition lubrifiante présente une trop forte teneur en élément molybdène. La mauvaise solubilité de ces composés modifie voire détériore les propriétés de la composition lubrifiante, notamment sa viscosité. Or, une composition trop ou pas assez visqueuse nuit au mouvement des pièces mobiles, au bon démarrage d'un moteur, à la protection d'un moteur lorsqu'il a atteint sa température de service, et donc in fine provoque notamment une augmentation de consommation de carburant.However, these compounds have the drawback of inducing the formation of sediments when the lubricating composition has too high a content of molybdenum element. The poor solubility of these compounds modifies or even deteriorates the properties of the lubricating composition, in particular its viscosity. However, a composition that is too viscous or not viscous enough is detrimental to the movement of moving parts, to the correct starting of an engine, to the protection of an engine when it has reached its operating temperature, and therefore ultimately causes in particular an increase in fuel consumption.
De plus, ces composés contribuent à augmenter le taux de cendre, réduisant leur potentiel d'utilisation dans une composition lubrifiante, notamment en Europe.In addition, these compounds contribute to an increase in the ash content, reducing their potential for use in a lubricating composition, in particular in Europe.
Il est également connu de formuler des compositions lubrifiantes comprenant des composés modificateurs de frottement de type organomolybdène avec des composés anti-usure et extrême pression organophosphorés et/ou organosoufrés, et/ou organophosphosoufrés, en particulier pour améliorer les propriétés anti-usure de ces huiles moteurs ou transmissions.It is also known to formulate lubricating compositions comprising organomolybdenum-type friction modifier compounds with organophosphorus and/or organosulfur and/or organophosphorus anti-wear and extreme pressure compounds, in particular to improve the anti-wear properties of these oils. engines or transmissions.
D'autres composés pour réduire les frottements ont été décrits comme pouvant présenter un intérêt dans la lubrification de pièces mécaniques, notamment des pièces d'un moteur.Other compounds for reducing friction have been described as being able to be of interest in the lubrication of mechanical parts, in particular parts of an engine.
Le document
Par ailleurs, la combinaison de nanoparticules et de composés anti-usure dans des compositions de graisse a été décrite, par exemple dans le document
Il serait donc souhaitable de disposer d'une composition lubrifiante, notamment pour véhicules automobiles, qui ne soit pas une graisse et qui soit à la fois stable tout en ayant de bonnes propriétés de réduction des frottements.It would therefore be desirable to have a lubricating composition, in particular for motor vehicles, which is not a grease and which is both stable while having good properties for reducing friction.
Il serait également souhaitable de disposer d'une composition lubrifiante, notamment pour véhicules automobiles, qui ne soit pas une graisse et dont les performances perdurent dans le temps.It would also be desirable to have a lubricating composition, in particular for motor vehicles, which is not a grease and whose performance lasts over time.
Il serait également souhaitable de disposer d'une composition lubrifiante, notamment pour véhicules automobiles, qui ne soit pas une graisse tout en présentant de bonnes propriétés de réduction des frottements et en conservant des propriétés anti-écaillage satisfaisantes.It would also be desirable to have a lubricating composition, in particular for motor vehicles, which is not a grease while exhibiting good properties for reducing friction and retaining satisfactory anti-flaking properties.
Un objectif de la présente invention est de fournir une composition lubrifiante palliant tout ou en partie les inconvénients précités.One objective of the present invention is to provide a lubricating composition which overcomes all or part of the aforementioned drawbacks.
Un autre objectif de l'invention est de fournir une composition lubrifiante stable et facile à mettre en œuvre.Another objective of the invention is to provide a lubricating composition which is stable and easy to implement.
Un autre objectif de la présente invention est de fournir un procédé de lubrification permettant notamment de réduire les frottements à la surface de pièces mécaniques, et plus particulièrement d'un moteur ou d'un organe de transmission de véhicules 30 automobiles.Another object of the present invention is to provide a method of lubrication which makes it possible in particular to reduce friction on the surface of mechanical parts, and more particularly of an engine or a transmission member of motor vehicles.
L'invention a ainsi pour objet une composition lubrifiante de viscosité cinématique à 100°C mesurée selon la norme ASTM D445 allant de 4 à 50 cSt et comprenant au moins 70% en poids d'au moins une huile de base, de 0,1 à 5% en poids d'au moins un composé anti-usure comprenant un groupement dithiophosphate et des nanoparticules métalliques solides ayant une structure de type fullerène représentées par la formule MXn dans laquelle M représente un métal de transition, X un chalcogène, avec n=2 ou n=3 en fonction de l'état d'oxydation du métal de transition en une teneur en poids allant de 0,01 à 2% par rapport au poids total de la composition lubrifiante. Les nanoparticules métalliques présentent une taille moyenne, déterminée à l'aide d'images obtenues par micrographie électronique à transmission ou par microscopie électronique à transmission à haute résolution, de 50 à 200 nm et sont des polyèdres concentriques avec une structure multicouche ou en feuillets.The subject of the invention is thus a lubricating composition with a kinematic viscosity at 100° C. measured according to the ASTM D445 standard ranging from 4 to 50 cSt and comprising at least 70% by weight of at least one base oil, from 0.1 to 5% by weight of at least one anti-wear compound comprising a dithiophosphate group and metallic nanoparticles solids having a fullerene-like structure represented by the formula MX n in which M represents a transition metal, X a chalcogen, with n=2 or n=3 depending on the oxidation state of the transition metal in a content by weight ranging from 0.01 to 2% relative to the total weight of the lubricating composition. The metallic nanoparticles have an average size, determined using images obtained by transmission electron micrography or by high-resolution transmission electron microscopy, of 50 to 200 nm and are concentric polyhedra with a multilayer or sheet structure.
De manière surprenante, la demanderesse a constaté que la présence d'un composé anti-usure comprenant un groupement dithiophosphate dans une composition lubrifiante comprenant au moins une huile de base et des nanoparticules métalliques telle que définies ci-dessus permet de conférer à ladite composition de très bonnes propriétés de réduction des frottements.Surprisingly, the Applicant has found that the presence of an anti-wear compound comprising a dithiophosphate group in a lubricating composition comprising at least one base oil and metallic nanoparticles as defined above makes it possible to confer on said composition very good friction reducing properties.
De plus, la demanderesse a constaté que l'association d'un composé anti-usure comprenant un groupement dithiophosphate et de nanoparticules métalliques telle que définies ci-dessus dans une composition lubrifiante permet de maintenir dans le temps cette réduction des frottements.In addition, the applicant has observed that the combination of an anti-wear compound comprising a dithiophosphate group and metallic nanoparticles as defined above in a lubricating composition makes it possible to maintain this reduction in friction over time.
Sans être lié par une théorie en particulier, ce maintien dans le temps de l'efficacité de réduction des frottements pourrait s'expliquer par la protection contre l'oxydation des nanoparticules métalliques par le composé anti-usure comprenant un groupement dithiophosphate, prolongeant ainsi l'action des nanoparticules métalliques à la surface d'une pièce mécanique, et plus particulièrement d'un organe de transmission ou d'un moteur de véhicules automobiles.Without being bound by a particular theory, this maintenance over time of the effectiveness of friction reduction could be explained by the protection against the oxidation of the metallic nanoparticles by the anti-wear compound comprising a dithiophosphate group, thus extending the action of metallic nanoparticles on the surface of a mechanical part, and more particularly of a transmission member or motor vehicle engine.
Ainsi, la présente invention permet de formuler des compositions lubrifiantes stables comprenant une teneur réduite en nanoparticules métalliques et présentant toutefois des propriétés de réduction des frottements remarquables.Thus, the present invention makes it possible to formulate stable lubricating compositions comprising a reduced content of metallic nanoparticles and yet exhibiting remarkable friction reduction properties.
Avantageusement, les compositions lubrifiantes selon l'invention présentent des propriétés de réduction des frottements remarquables et qui perdurent dans le temps. Avantageusement, les compositions lubrifiantes selon l'invention présentent une bonne stabilité au ainsi qu'une viscosité ne variant pas ou très peu.Advantageously, the lubricating compositions according to the invention have remarkable friction reduction properties which last over time. Advantageously, the lubricating compositions according to the invention have good oil stability and a viscosity which does not vary or varies very little.
Avantageusement, les compositions lubrifiantes selon l'invention présentent des propriétés anti-écaillages satisfaisantes.Advantageously, the lubricating compositions according to the invention have satisfactory anti-flaking properties.
Avantageusement, les compositions lubrifiantes selon l'invention présentent un risque d'oxydation réduit.Advantageously, the lubricating compositions according to the invention have a reduced risk of oxidation.
Avantageusement, les compositions lubrifiantes selon l'invention présentent des propriétés d'économies de carburant remarquablesAdvantageously, the lubricating compositions according to the invention have remarkable fuel saving properties
Dans un mode de réalisation, la composition lubrifiante consiste essentiellement en au moins une huile de base, au moins un composé anti-usure comprenant un groupement dithiophosphate et au moins des nanoparticules métalliques telles que définies ci-dessus en une teneur en poids allant de 0,01 à 2% par rapport au poids total de la composition lubrifiante.In one embodiment, the lubricating composition consists essentially of at least one base oil, at least one anti-wear compound comprising a dithiophosphate group and at least metallic nanoparticles as defined above in a content by weight ranging from 0 0.01 to 2% relative to the total weight of the lubricating composition.
La demande décrit également une huile moteur comprenant une composition lubrifiante telle que définie ci-dessus.The application also describes an engine oil comprising a lubricating composition as defined above.
La demande décrit également une huile transmission comprenant une composition lubrifiante telle que définie ci-dessus.The application also describes a transmission oil comprising a lubricating composition as defined above.
L'invention concerne également l'utilisation d'une composition lubrifiante telle que définie ci-dessus pour la lubrification d'une pièce mécanique, préférentiellement d'un organe de transmission ou d'un moteur de véhicules, avantageusement de véhicules automobiles.The invention also relates to the use of a lubricating composition as defined above for lubricating a mechanical part, preferably a transmission member or a vehicle engine, advantageously motor vehicles.
L'invention concerne également l'utilisation d'une composition lubrifiante telle que définie ci-dessus pour la réduction des frottements à la surface d'une pièce mécanique, préférentiellement d'un organe de transmission ou d'un moteur de véhicules, avantageusement de véhicules automobiles.The invention also relates to the use of a lubricating composition as defined above for reducing friction on the surface of a mechanical part, preferably a transmission member or a vehicle engine, advantageously motor vehicles.
L'invention concerne également l'utilisation d'une composition lubrifiante telle que définie ci-dessus pour réduire la consommation de carburant de véhicules, en particulier de véhicules automobiles.The invention also relates to the use of a lubricating composition as defined above for reducing the fuel consumption of vehicles, in particular motor vehicles.
La demande décrit également un procédé de lubrification d'une pièce mécanique, préférentiellement d'un organe de transmission ou d'un moteur de véhicules, avantageusement de véhicules automobiles, ledit procédé comprenant au moins une étape de mise en contact de la pièce mécanique avec une composition lubrifiante telle que définie ci-dessus.The application also describes a process for lubricating a mechanical part, preferably a transmission member or a vehicle engine, advantageously motor vehicles, said process comprising at least one step of bringing the mechanical part into contact with a lubricating composition as defined above.
La demande décrit également un procédé de réduction des frottements à la surface d'une pièce mécanique, préférentiellement d'un organe de transmission ou d'un moteur de véhicules, avantageusement de véhicules automobiles, comprenant au moins la mise en contact de la pièce mécanique avec une composition lubrifiante telle que définie ci-dessus.The application also describes a method for reducing friction on the surface of a mechanical part, preferably of a transmission member or of a vehicle engine, advantageously of motor vehicles, comprising at least bringing the mechanical part into contact with a lubricating composition as defined above.
La demande décrit également un procédé pour réduire la consommation de carburant d'un véhicule, en particulier d'un véhicule automobile, comprenant au moins une étape de mise en contact d'une pièce mécanique du moteur du véhicule avec une composition lubrifiante telle que définie ci-dessus.The application also describes a method for reducing the fuel consumption of a vehicle, in particular of a motor vehicle, comprising at least one step of bringing a mechanical part of the engine of the vehicle into contact with a lubricating composition as defined above.
L'invention concerne également l'utilisation d'un composé anti-usure comprenant un groupement dithiophosphate pour diminuer l'oxydation d'une composition lubrifiante comprenant au moins une huile de base et des nanoparticules métalliques solides ayant une structure de type fullerène représentées par la formule MXn dans laquelle M représente un métal de transition, X un chalcogène, avec n=2 ou n=3 en fonction de l'état d'oxydation du métal de transition, les nanoparticules métalliques présentant une taille moyenne, déterminée à l'aide d'images obtenues par micrographie électronique à transmission ou par microscopie électronique à transmission à haute résolution, de 50 à 200 nm et étant des polyèdres concentriques avec une structure multicouche ou en feuillets.The invention also relates to the use of an anti-wear compound comprising a dithiophosphate group to reduce the oxidation of a lubricating composition comprising at least one base oil and solid metallic nanoparticles having a fullerene-type structure represented by the formula MXn in which M represents a transition metal, X a chalcogen, with n=2 or n=3 depending on the oxidation state of the transition metal, the metallic nanoparticles having an average size, determined using images obtained by transmission electron micrography or by high resolution transmission electron microscopy, from 50 to 200 nm and being of concentric polyhedra with a multilayer or sheet structure.
La demande décrit également une composition de type concentré d'additifs comprenant au moins au moins un composé comprenant un groupement dithiophosphate et des nanoparticules de bisulfure de tungstène.The application also describes a composition of the concentrate type of additives comprising at least at least one compound comprising a dithiophosphate group and tungsten disulphide nanoparticles.
Les pourcentages indiqués ci-dessous correspondent à des pourcentages en masse de matière active.The percentages indicated below correspond to percentages by mass of active material.
La composition lubrifiante selon l'invention comprend des nanoparticules métalliques en une teneur en poids allant de 0,01 à 2% par rapport au poids total de la composition lubrifiante.The lubricating composition according to the invention comprises metallic nanoparticles in a content by weight ranging from 0.01 to 2% relative to the total weight of the lubricating composition.
Par nanoparticules métalliques, on entend notamment des particules métalliques, généralement solides, dont la taille moyenne est inférieure ou égale à 600 nm.By metallic nanoparticles is meant in particular metallic particles, generally solid, whose average size is less than or equal to 600 nm.
Avantageusement, les nanoparticules métalliques sont constituées d'au moins 80% en masse par au moins un métal, ou bien d'au moins 80% en masse d'au moins un alliage métallique ou bien d'au moins 80% en masse d'au moins un chalcogénure de métal, notamment de métal de transition, par rapport à la masse totale de la nanoparticule. Avantageusement, les nanoparticules métalliques sont constituées d'au moins 90% en masse par au moins un métal, ou bien d'au moins 90% en masse d'au moins un alliage métallique ou bien d'au moins 90% en masse d'au moins un chalcogénure de métal, notamment de métal de transition, par rapport à la masse totale de la nanoparticule.Advantageously, the metallic nanoparticles consist of at least 80% by mass of at least one metal, or else of at least 80% by mass of at least one metal alloy or else of at least 80% by mass of at least one metal chalcogenide, in particular of transition metal, with respect to the total mass of the nanoparticle. Advantageously, the metallic nanoparticles consist of at least 90% by mass of at least one metal, or else of at least 90% by mass of at least one metal alloy or else of at least 90% by mass of at least one metal chalcogenide, in particular of transition metal, with respect to the total mass of the nanoparticle.
Avantageusement, les nanoparticules métalliques sont constituées d'au moins 99% en masse par au moins un métal, ou bien d'au moins 99% en masse d'au moins un alliage métallique ou bien d'au moins 99% en masse d'au moins un chalcogénure de métal, notamment de métal de transition, par rapport à la masse totale de la nanoparticule, les 1% restants étant constitués d'impuretés.Advantageously, the metallic nanoparticles consist of at least 99% by mass of at least one metal, or else of at least 99% by mass of at least one metal alloy or else of at least 99% by mass of at least one metal chalcogenide, in particular of transition metal, with respect to the total mass of the nanoparticle, the remaining 1% being constituted by impurities.
Avantageusement, le métal dont est constitué la nanoparticule métallique peut être choisi parmi le groupe formé par le tungstène, le molybdène, le zirconium, l'hafnium, le platine, le rhénium, le titane, le tantale et le niobium, de préférence le molybdène ou le tungstène, avantageusement le tungstène.Advantageously, the metal of which the metallic nanoparticle is made can be chosen from the group formed by tungsten, molybdenum, zirconium, hafnium, platinum, rhenium, titanium, tantalum and niobium, preferably molybdenum or tungsten, advantageously tungsten.
Les nanoparticules métalliques utilisées dans les compositions selon l'invention sont des nanoparticules métalliques solides ayant une structure de type fullerène (en terme anglo-saxon fullerene-like) et sont représentées par la formule MXn dans laquelle M représente un métal de transition, X un chalcogène, avec n=2 ou n=3 en fonction de l'état d'oxydation du métal de transition M.The metallic nanoparticles used in the compositions according to the invention are solid metallic nanoparticles having a structure of the fullerene type (in English term fullerene-like) and are represented by the formula MX n in which M represents a transition metal, X a chalcogen, with n=2 or n=3 depending on the oxidation state of the transition metal M.
De manière préférée, M est choisi dans le groupe formé par le tungstène, le molybdène, le zirconium, l'hafnium, le platine, le rhénium, le titane, le tantale et le niobium.Preferably, M is chosen from the group formed by tungsten, molybdenum, zirconium, hafnium, platinum, rhenium, titanium, tantalum and niobium.
De manière plus préféré, M est choisi parmi le groupe formé par le molybdène et le 5 tungstène.More preferably M is selected from the group consisting of molybdenum and tungsten.
De manière encore plus préférée, M est le tungstène.Even more preferably, M is tungsten.
De manière préférée, X est choisi dans le groupe formé par l'oxygène, le soufre, le sélénium et le tellure.Preferably, X is chosen from the group formed by oxygen, sulfur, selenium and tellurium.
De manière préférée, X est choisi parmi le soufre ou le tellure.Preferably, X is chosen from sulfur or tellurium.
De manière encore plus préféré, X est le soufre.Even more preferably, X is sulfur.
Avantageusement, les nanoparticules métalliques selon l'invention sont choisies dans le groupe formé par MoS2, MoSe2, MoTe2, WS2, WSe2, ZrS2, ZrSe2, HfS2, HfSe2, PtS2, ReS2, ReSe2, TiS3, ZrS3, ZrSe3, HfS3, HfSe3, TiS2, TaS2, TaSe2, NbS2, NbSe2 et NbTe2.Advantageously, the metallic nanoparticles according to the invention are chosen from the group formed by MoS2, MoSe 2 , MoTe 2 , WS 2 , WSe 2 , ZrS 2 , ZrSe 2 , HfS 2 , HfSe 2 , PtS 2 , ReS 2 , ReSe 2 , TiS 3 , ZrS 3 , ZrSe 3 , HfS 3 , HfSe 3 , TiS 2 , TaS 2 , TaSe 2 , NbS 2 , NbSe 2 and NbTe 2 .
De manière préférée, les nanoparticules métalliques selon l'invention sont choisies dans le groupe formé par WS2, WSe2, MoS2 et MoSe2, préférentiellement WS2 et MoS2, préférentiellement WS2.Preferably, the metallic nanoparticles according to the invention are chosen from the group formed by WS 2 , WSe 2 , MoS 2 and MoSe 2 , preferentially WS 2 and MoS 2 , preferentially WS 2 .
Les nanoparticules selon l'invention présentent une structure de type fullèrene.The nanoparticles according to the invention have a fullerene-type structure.
Initialement, le terme fullerène désigne une nanostructure de polyèdre convexe fermé, composée d'atomes de carbone. Les fullerènes sont similaires au graphite, composé de feuilles d'anneaux hexagonaux liées, mais ils contiennent des anneaux pentagonaux, et parfois heptagonaux, qui empêchent la structure d'être plate.Initially, the term fullerene designates a closed convex polyhedron nanostructure, composed of carbon atoms. Fullerenes are similar to graphite, consisting of sheets of bonded hexagonal rings, but they contain pentagonal, and sometimes heptagonal, rings that prevent the structure from being flat.
Des études sur les structures de type fullerène ont montré que cette structure n'était pas limitée aux matériaux carbonés, mais était susceptible de se produire dans toutes les nanoparticules de matériaux sous forme de feuillets, notamment pour les nanoparticules comprenant des chalcogènes et des métaux de transition. Ces structures sont analogues à celle des fullerènes de carbone et sont nommées fullerènes inorganiques ou structure de type fullerène (en terme anglo-saxon « Inorganic Fullerene like materials », encore désignés par « IF »). Les structures de type fullerène sont décrites notamment par
Dans un mode de réalisation préféré de l'invention, les nanoparticules métalliques sont des structures fermées, de type sphérique, plus ou moins parfaites selon les procédés de synthèse utilisés. Les nanoparticules selon l'invention sont des polyèdres concentriques avec une structure multicouche ou en feuillets. On parle de structure en « oignons » ou de « polyèdre emboité ».In a preferred embodiment of the invention, the metallic nanoparticles are closed structures, of the spherical type, more or less perfect depending on the synthesis methods used. The nanoparticles according to the invention are concentric polyhedrons with a multilayer or sheet structure. We speak of an "onion" or "nested polyhedron" structure.
Dans un mode de réalisation de l'invention, les nanoparticules métalliques sont des nanoparticules métalliques multicouches comprenant de 2 à 500 couches, de préférence de 20 à 200 couches, avantageusement de 20 à 100 couches.In one embodiment of the invention, the metallic nanoparticles are multilayer metallic nanoparticles comprising from 2 to 500 layers, preferably from 20 to 200 layers, advantageously from 20 to 100 layers.
La taille moyenne des nanoparticules métalliques selon l'invention va de 50 à 200 nm. La taille des nanoparticules métalliques selon l'invention peut être déterminée à l'aide d'images obtenues par micrographie électronique à transmission ou par microscopie électronique à transmission à haute résolution. On peut déterminer la taille moyenne des particules à partir de la mesure de la taille d'au moins 50 particules solides visualisées sur des clichés de microscopie électronique à transmission. La valeur médiane de l'histogramme de distribution des tailles mesurée des particules solides est la taille moyenne des particules solides utilisées dans la composition lubrifiante selon l'invention.The average size of the metallic nanoparticles according to the invention ranges from 50 to 200 nm. The size of the metallic nanoparticles according to the invention can be determined using images obtained by transmission electron micrography or by electron microscopy. high resolution transmission. The average particle size can be determined from the measurement of the size of at least 50 solid particles visualized on transmission electron micrographs. The median value of the measured size distribution histogram of the solid particles is the average size of the solid particles used in the lubricating composition according to the invention.
De manière avantageuse, la teneur en poids de nanoparticules métalliques va de 0,05 à 2%, de préférence de 0,1 à 1%, avantageusement de 0,1 à 0,5% par rapport au poids total de la composition lubrifiante.Advantageously, the content by weight of metallic nanoparticles ranges from 0.05 to 2%, preferably from 0.1 to 1%, advantageously from 0.1 to 0.5% relative to the total weight of the lubricating composition.
Comme exemple de nanoparticules métalliques selon l'invention, on peut citer le produit NanoLub Gear Oil Concentrate commercialisé par la société Nanomaterials, se présentant sous la forme d'une dispersion de nanoparticules multicouches de bisulfure de tungstène dans une huile minérale ou de type PAO (Poly Alfa Oléfine).As an example of metallic nanoparticles according to the invention, mention may be made of the product NanoLub Gear Oil Concentrate marketed by the company Nanomaterials, which is in the form of a dispersion of multilayer nanoparticles of tungsten disulphide in a mineral or PAO type oil ( Poly Alfa Olefin).
La composition lubrifiante selon l'invention comprend au moins un composé anti-usure comprenant un groupement dithiophosphate.The lubricating composition according to the invention comprises at least one anti-wear compound comprising a dithiophosphate group.
Par soucis de simplification de la description, le composé anti-usure comprenant un groupement dithiophosphate est appelé « dithiophosphate » dans la suite de la présente description.For the sake of simplification of the description, the anti-wear compound comprising a dithiophosphate group is called “dithiophosphate” in the rest of the present description.
Le dithiophosphate, sans être limitatif, peut être choisi parmi les dithiophosphates d'ammonium, les dithiophosphates d'amine, les dithiophosphates d'ester et les dithiophosphates métalliques, pris seuls ou en mélange.The dithiophosphate, without being limiting, can be chosen from ammonium dithiophosphates, amine dithiophosphates, ester dithiophosphates and metal dithiophosphates, taken alone or as a mixture.
Dans un mode de réalisation de l'invention, le dithiophosphate est choisi parmi les dithiophosphates d'ammonium de formule (I) :
Dans un mode de réalisation préféré de l'invention, R1 et R2 représentent indépendamment l'un de l'autre un groupe hydrocarboné, éventuellement substitué, comprenant de 2 à 24 atomes de carbone, plus préférentiellement de 3 à 18 atomes de carbone, avantageusement de 5 à 12 atomes de carbone.In a preferred embodiment of the invention, R1 and R2 independently of each other represent a hydrocarbon group, optionally substituted, comprising from 2 to 24 carbon atoms, more preferentially from 3 to 18 carbon atoms, advantageously from 5 to 12 carbon atoms.
Dans un autre mode de réalisation préféré de l'invention, R1 et R2 représentent indépendamment l'un de l'autre un groupe hydrocarboné, non substitué, ledit groupe hydrocarboné pouvant être un groupe alkyle, alkényle, alkynyle, phényle ou benzyle.In another preferred embodiment of the invention, R1 and R2 independently represent an unsubstituted hydrocarbon group, said hydrocarbon group possibly being an alkyl, alkenyl, alkynyl, phenyl or benzyl group.
Dans un autre mode de réalisation préféré de l'invention, R1 et R2 représentent indépendamment l'un de l'autre un groupe hydrocarboné alkyle linéaire ou ramifié, plus préférentiellement un groupe hydrocarboné alkyle linéaire.In another preferred embodiment of the invention, R1 and R2 independently represent a linear or branched alkyl hydrocarbon group, more preferably a linear alkyl hydrocarbon group.
Dans un autre mode de réalisation préféré de l'invention, R1 et R2 représentent indépendamment l'un de l'autre un groupe hydrocarboné éventuellement substitué par au moins un atome d'oxygène, d'azote, de soufre et/ou de phosphore, de préférence par au moins un atome d'oxygène.In another preferred embodiment of the invention, R1 and R2 independently represent a hydrocarbon group optionally substituted by at least one oxygen, nitrogen, sulfur and/or phosphorus atom, preferably by at least one oxygen atom.
Comme exemples de dithiophosphate d'ammonium, on peut citer les diméthyl dithiophosphates d'ammonium, les diéthyl dithiophosphates d'ammonium et les dibutyl dithiophosphates d'ammonium.As examples of ammonium dithiophosphate, mention may be made of ammonium dimethyl dithiophosphates, ammonium diethyl dithiophosphates and ammonium dibutyl dithiophosphates.
Dans un autre mode de réalisation de l'invention, le dithiophosphate est choisi parmi les dithiophosphates d'amine de formule générale (II):
- R3 et R4 représentent indépendamment l'un de l'autre un groupe hydrocarboné, éventuellement substitué, comprenant de 1 à 30 atomes de carbone,
- R5, R6 et R7 représentent indépendamment l'un de l'autre un atome d'hydrogène ou un groupement hydrocarboné de 1 à 30 atomes de carbone, étant entendu qu'au moins un des groupes R5, R6 et R7 ne représente pas un atome d'hydrogène.
- R3 and R4 represent, independently of each other, an optionally substituted hydrocarbon group comprising from 1 to 30 carbon atoms,
- R5, R6 and R7 represent, independently of one another, a hydrogen atom or a hydrocarbon group of 1 to 30 carbon atoms, it being understood that at least one of the groups R5, R6 and R7 does not represent a of hydrogen.
Dans un mode de réalisation préféré de l'invention, R3 et R4 représentent indépendamment l'un de l'autre un groupe hydrocarboné, éventuellement substitué, comprenant de 2 à 24 atomes de carbone, plus préférentiellement de 3 à 18 atomes de carbone, avantageusement de 5 à 12 atomes de carbone.In a preferred embodiment of the invention, R3 and R4 independently of each other represent a hydrocarbon group, optionally substituted, comprising from 2 to 24 carbon atoms, more preferentially from 3 to 18 carbon atoms, advantageously from 5 to 12 carbon atoms.
Dans un autre mode de réalisation préféré de l'invention, R3 et R4 représentent indépendamment l'un de l'autre un groupe hydrocarboné, non substitué, ledit groupe hydrocarboné pouvant être un groupe alkyle, alkényle, alkynyle, phényle ou benzyle.In another preferred embodiment of the invention, R3 and R4 independently represent an unsubstituted hydrocarbon group, said hydrocarbon group possibly being an alkyl, alkenyl, alkynyl, phenyl or benzyl group.
Dans un autre mode de réalisation préféré de l'invention, R3 et R4 représentent indépendamment l'un de l'autre un groupe hydrocarboné alkyle linéaire ou ramifié, plus préférentiellement un groupe hydrocarboné alkyle linéaire.In another preferred embodiment of the invention, R3 and R4 independently represent a linear or branched alkyl hydrocarbon group, more preferably a linear alkyl hydrocarbon group.
Dans un autre mode de réalisation préféré de l'invention, R3 et R4 représentent indépendamment l'un de l'autre un groupe hydrocarboné éventuellement substitué par au moins un atome d'oxygène, d'azote, de soufre et/ou de phosphore, de préférence par au moins un atome d'oxygène.In another preferred embodiment of the invention, R3 and R4 independently represent a hydrocarbon group optionally substituted by at least one oxygen, nitrogen, sulfur and/or phosphorus atom, preferably by at least one oxygen atom.
Dans un autre mode de réalisation préféré de l'invention, R5, R6 et R7 représentent indépendamment l'un de l'autre un groupement hydrocarboné comprenant de 2 à 24 atomes de carbone, plus préférentiellement de 3 à 18 atomes de carbone, avantageusement de 5 à 12 atomes de carbone.In another preferred embodiment of the invention, R5, R6 and R7 independently represent a hydrocarbon group comprising from 2 to 24 carbon atoms, more preferably from 3 to 18 carbon atoms, advantageously from 5 to 12 carbon atoms.
Dans un autre mode de réalisation de l'invention, le dithiophosphate est choisi parmi les dithiophosphates d'ester de formule générale (III) :
- R8 et R9 représentent indépendamment l'un de l'autre un groupe hydrocarboné, éventuellement substitué, comprenant de 1 à 30 atomes de carbone,
- R10 et R11 représentent indépendamment l'un de l'autre un groupe hydrocarboné comprenant de 1 à 18 atomes de carbone.
- R8 and R9 represent, independently of each other, an optionally substituted hydrocarbon group comprising from 1 to 30 carbon atoms,
- R10 and R11 represent, independently of each other, a hydrocarbon group comprising from 1 to 18 carbon atoms.
Dans un mode de réalisation préféré de l'invention, R8 et R9 représentent indépendamment l'un de l'autre un groupe hydrocarboné, éventuellement substitué, comprenant de 2 à 24 atomes de carbone, plus préférentiellement de 3 à 18 atomes de carbone, avantageusement de 5 à 12 atomes de carbone.In a preferred embodiment of the invention, R8 and R9 independently represent an optionally substituted hydrocarbon group comprising from 2 to 24 carbon atoms, more preferentially from 3 to 18 carbon atoms, advantageously from 5 to 12 carbon atoms.
Dans un autre mode de réalisation préféré de l'invention, R8 et R9 représentent indépendamment l'un de l'autre un groupe hydrocarboné, non substitué, ledit groupe hydrocarboné pouvant être un groupe alkyle, alkényle, alkynyle, phényle ou benzyle.In another preferred embodiment of the invention, R8 and R9 independently represent an unsubstituted hydrocarbon group, said hydrocarbon group possibly being an alkyl, alkenyl, alkynyl, phenyl or benzyl group.
Dans un autre mode de réalisation préféré de l'invention, R8 et R9 représentent indépendamment l'un de l'autre un groupe hydrocarboné alkyle linéaire ou ramifié, plus préférentiellement un groupe hydrocarboné alkyle linéaire.In another preferred embodiment of the invention, R8 and R9 independently represent a linear or branched alkyl hydrocarbon group, more preferably a linear alkyl hydrocarbon group.
Dans un autre mode de réalisation préféré de l'invention, R8 et R9 représentent indépendamment l'un de l'autre un groupe hydrocarboné éventuellement substitué par au moins un atome d'oxygène, d'azote, de soufre et/ou de phosphore, de préférence par au moins un atome d'oxygène.In another preferred embodiment of the invention, R8 and R9 independently represent a hydrocarbon group optionally substituted by at least one oxygen, nitrogen, sulfur and/or phosphorus atom, preferably by at least one oxygen atom.
Dans un autre mode de réalisation préféré de l'invention, R8 et R9 représentent 20 indépendamment l'un de l'autre, un groupe hydrocarboné comprenant de 2 à 6 atomes de carbone.In another preferred embodiment of the invention, R8 and R9 represent, independently of each other, a hydrocarbon group comprising from 2 to 6 carbon atoms.
Dans un autre mode de réalisation préféré de l'invention, R10 et R11 représentent indépendamment l'un de l'autre un groupe hydrocarboné comprenant de 2 à 6 atomes de carbone.In another preferred embodiment of the invention, R10 and R11 independently represent a hydrocarbon group comprising from 2 to 6 carbon atoms.
Dans un autre mode de réalisation, le dithiophosphate est choisi parmi les dithiophosphates métalliques de formule générale (IV) :
- R12 représente un groupe alkyle linéaire ou ramifié, substitué ou non substitué comprenant de 1 à 30 atomes de carbone ;
- R13 représente un groupe alkyle linéaire ou ramifié, substitué ou non substitué comprenant de 1 à 30 atomes de carbone ;
- M représente un cation métallique, de préférence un cation Zn2+ ;
- n représente la valence du cation métallique.
- R12 represents a linear or branched, substituted or unsubstituted alkyl group comprising from 1 to 30 carbon atoms;
- R13 represents a linear or branched, substituted or unsubstituted alkyl group comprising from 1 to 30 carbon atoms;
- M represents a metal cation, preferably a Zn 2+ cation;
- n represents the valence of the metal cation.
Dans un mode de réalisation préféré de l'invention, le métal est choisi dans le groupe constitué par le zinc, l'aluminium, le cuivre, le fer, le mercure, l'argent, le cadmium, l'étain, le plomb, l'antimoine, le bismuth, le thallium, le chrome, le molybdène, le cobalt, le nickel, le tungstène, le sodium, le calcium, le magnésium, le manganèse et l'arsenic. Les métaux préférés sont le zinc, le molybdène, l'antimoine, de préférence le zinc et le molybdène.In a preferred embodiment of the invention, the metal is selected from the group consisting of zinc, aluminum, copper, iron, mercury, silver, cadmium, tin, lead, antimony, bismuth, thallium, chromium, molybdenum, cobalt, nickel, tungsten, sodium, calcium, magnesium, manganese and arsenic. Preferred metals are zinc, molybdenum, antimony, preferably zinc and molybdenum.
Dans un mode de réalisation préféré de l'invention, le métal est le zinc.In a preferred embodiment of the invention, the metal is zinc.
Des mélanges de métaux peuvent être utilisés. Les dithiophosphates métalliques 20 sont neutres comme exemplifiés dans la formule (IV) ou basiques quand un excès stœchiométrique de métal est présent.Mixtures of metals can be used. Metal dithiophosphates are neutral as exemplified in formula (IV) or basic when a stoichiometric excess of metal is present.
Dans un mode de réalisation préféré de l'invention, R12 et R13 représentent indépendamment l'un de l'autre un groupe hydrocarboné, éventuellement substitué, comprenant de 2 à 24 atomes de carbone, plus préférentiellement de 3 à 18 atomes de carbone, avantageusement de 5 à 12 atomes de carbone.In a preferred embodiment of the invention, R12 and R13 independently represent an optionally substituted hydrocarbon group comprising from 2 to 24 carbon atoms, more preferentially from 3 to 18 carbon atoms, advantageously from 5 to 12 carbon atoms.
Dans un autre mode de réalisation préféré de l'invention, R12 et R13 représentent indépendamment l'un de l'autre un groupe hydrocarboné, non substitué, ledit groupe hydrocarboné pouvant être un groupe alkyle, alkényle, alkynyle, phényle ou benzyle. Dans un autre mode de réalisation préféré de l'invention, R12 et R13 représentent indépendamment l'un de l'autre un groupe hydrocarboné alkyle linéaire ou ramifié, plus préférentiellement un groupe hydrocarboné alkyle linéaire.In another preferred embodiment of the invention, R12 and R13 independently represent an unsubstituted hydrocarbon group, said hydrocarbon group possibly being an alkyl, alkenyl, alkynyl, phenyl or benzyl group. In another preferred embodiment of the invention, R12 and R13 independently represent a linear or branched alkyl hydrocarbon group, more preferably a linear alkyl hydrocarbon group.
Dans un autre mode de réalisation préféré de l'invention, R12 et R13 représentent indépendamment l'un de l'autre un groupe hydrocarboné éventuellement substitué par au moins un atome d'oxygène, d'azote, de soufre et/ou de phosphore, de préférence par au moins un atome d'oxygène.In another preferred embodiment of the invention, R12 and R13 independently represent a hydrocarbon group optionally substituted by at least one oxygen, nitrogen, sulfur and/or phosphorus atom, preferably by at least one oxygen atom.
De manière avantageuse, le dithiophosphate selon l'invention est un dithiophosphate de zinc de formule (IV-a) ou de formule (IV-b) :
En tant que dithiophosphate métallique selon l'invention, on peut citer par exemple l'Additin® RC 3038, l'Additin® RC 3045, l'Additin® RC 3048, l'Additin® RC 3058, l'Additin° RC 3080, l'Additin° RC 3180, l'Additin° RC 3212, l'Additin° RC 3580, le Kikulube° Z112, le Lubrizol® 1371, le Lubrizol® 1375, le Lubrizol® 1395, le Lubrizol® 5179, l'Oloa® 260, l'Oloa® 267.As metal dithiophosphate according to the invention, one can quote for example Additin ® RC 3038, Additin ® RC 3045, Additin ® RC 3048, Additin ® RC 3058, Additin° RC 3080, Additin° RC 3180, Additin° RC 3212, Additin° RC 3580, Kikulube° Z112, Lubrizol ® 1371, Lubrizol ® 1375, Lubrizol ® 1395, Lubrizol ® 5179, Oloa ® 260, Oloa® 267.
La teneur en poids de composé anti-usure comprenant un groupement dithiophosphate va de 0,1 à 5%, par rapport au poids total de la composition lubrifiante.The content by weight of anti-wear compound comprising a dithiophosphate group ranges from 0.1 to 5%, relative to the total weight of the lubricating composition.
Dans un mode de réalisation de l'invention, la teneur en poids de composé comprenant un groupement dithiophosphate va de 0,2 à 4%, plus préférentiellement de 0,5 à 2%, avantageusement de 0,5 à 1,5% par rapport au poids total de la composition lubrifiante.In one embodiment of the invention, the content by weight of compound comprising a dithiophosphate group ranges from 0.2 to 4%, more preferably from 0.5 to 2%, advantageously from 0.5 to 1.5% per relative to the total weight of the lubricating composition.
Les compositions lubrifiantes selon l'invention peuvent contenir tout type d'huile de base lubrifiante minérale, synthétique ou naturelle, animale ou végétale adaptée(s) à leur utilisation.The lubricating compositions according to the invention can contain any type of mineral, synthetic or natural, animal or vegetable lubricating base oil suitable for their use.
La ou les huiles de base utilisées dans les compositions lubrifiantes selon la présente invention peuvent être des huiles d'origine minérales ou synthétiques des groupes I à V selon les classes définies dans la classification API (ou leurs équivalents selon la classification ATIEL) telle que résumée ci-dessous, seules ou en mélange.
Les huiles de base minérales selon l'invention incluent tous type de bases obtenues par distillation atmosphérique et sous vide du pétrole brut, suivies d'opérations de raffinage tels qu'extraction au solvant, désasphaltage, déparaffinage au solvant, hydrotraitement, hydrocraquage et hydroisomérisation, hydrofinition.The mineral base oils according to the invention include all types of bases obtained by atmospheric and vacuum distillation of crude oil, followed by refining operations such as solvent extraction, deasphalting, solvent dewaxing, hydrotreating, hydrocracking and hydroisomerization, hydrofinishing.
Les huiles de bases des compositions lubrifiantes selon l'invention peuvent également être des huiles synthétiques, tels certains esters d'acides carboxyliques et d'alcools, ou des polyalphaoléfines. Les polyalphaoléfines utilisées comme huiles de base, sont par exemple obtenues à partir de monomères ayant de 4 à 32 atomes de carbone (par exemple octène, decène), et une viscosité à 100°C comprise entre 1,5 et 15 cSt mesurée selon la norme ASTM D445. Leur masse moléculaire moyenne en poids est typiquement comprise entre 250 et 3000 mesurée selon la norme ASTM D5296. Des mélanges d'huiles synthétiques et minérales peuvent également être employés.The base oils of the lubricating compositions according to the invention can also be synthetic oils, such as certain esters of carboxylic acids and alcohols, or polyalphaolefins. The polyalphaolefins used as base oils are for example obtained from monomers having from 4 to 32 carbon atoms (for example octene, decene), and a viscosity at 100° C. of between 1.5 and 15 cSt measured according to the ASTM D445 standard. Their weight-average molecular weight is typically between 250 and 3000 measured according to standard ASTM D5296. Blends of synthetic and mineral oils can also be used.
Il n'existe aucune limitation quant à l'emploi de telle ou telle base lubrifiante pour réaliser les compositions lubrifiantes selon l'invention, si ce n'est qu'elles doivent avoir des propriétés, notamment de viscosité, indice de viscosité, teneur en soufre, résistance à l'oxydation, adaptées à une utilisation dans une boite de vitesses, en particulier dans une boite de vitesses de véhicules automobile, en particulier dans une boite de vitesses manuelle.There is no limitation as to the use of such and such a lubricating base for producing the lubricating compositions according to the invention, except that they must have properties, in particular viscosity, viscosity index, sulphur, resistance to oxidation, suitable for use in a gearbox, in particular in a motor vehicle gearbox, in particular in a manual gearbox.
Dans l'invention, les bases lubrifiantes représentent au moins 70% en masse, par rapport à la masse totale de la composition lubrifiante, Typiquement, elles représentent entre 75 et 99,9% en masse, par rapport à la masse totale des compositions lubrifiantes selon l'invention.In the invention, the lubricating bases represent at least 70% by mass, relative to the total mass of the lubricating composition, Typically, they represent between 75 and 99.9% by mass, relative to the total mass of the lubricating compositions according to the invention.
La composition lubrifiante selon l'invention a une viscosité cinématique à 100°C mesurée selon la norme ASTM D445 allant de 4 à 50 cSt.The lubricating composition according to the invention has a kinematic viscosity at 100° C. measured according to the ASTM D445 standard ranging from 4 to 50 cSt.
Dans un mode de réalisation, la viscosité cinématique à 100°C mesurée selon la norme ASTM D445 de la composition selon l'invention va de 4 à 45 cSt, de préférence de 4 à 30 cSt.In one embodiment, the kinematic viscosity at 100° C. measured according to the ASTM D445 standard of the composition according to the invention ranges from 4 to 45 cSt, preferably from 4 to 30 cSt.
Dans un mode de réalisation préféré de l'invention, les compositions lubrifiantes comprennent au moins une base de groupe IV.In a preferred embodiment of the invention, the lubricating compositions comprise at least one Group IV base.
Dans un autre mode de réalisation préféré de l'invention, les compositions lubrifiantes ont un indice de viscosité (VI) supérieur à 95 (norme ASTM 2270).In another preferred embodiment of the invention, the lubricating compositions have a viscosity index (VI) greater than 95 (ASTM 2270 standard).
Les compositions lubrifiantes selon l'invention peuvent également contenir tout type d'additif adapté pour leur utilisation dans les formulations d'huiles pour transmissions, par exemple un ou plusieurs additifs choisis parmi les polymères, les antioxydants, les additifs anti-corrosion, les modificateurs de frottements différents des nanoparticules métalliques selon l'invention et les dispersants, présents aux teneurs usuelles requises pour l'application. Dans un mode de réalisation de l'invention, l'additif est choisi parmi les dispersants 10 ayant une masse moléculaire moyenne en poids supérieure ou égale à 2000 Daltons.The lubricating compositions according to the invention may also contain any type of additive suitable for their use in the formulations of oils for transmissions, for example one or more additives chosen from polymers, antioxidants, anti-corrosion additives, modifiers different frictions of the metallic nanoparticles according to the invention and the dispersants, present at the usual levels required for the application. In one embodiment of the invention, the additive is chosen from dispersants having a weight-average molecular mass greater than or equal to 2000 Daltons.
Selon l'invention, la masse moléculaire moyenne en poids du dispersant est évaluée selon la norme ASTM D5296.According to the invention, the weight-average molecular mass of the dispersant is evaluated according to the ASTM D5296 standard.
Par dispersant au sens de la présente invention, on entend plus particulièrement tout composé qui améliore le maintien en suspension des nanoparticules métalliques.By dispersant within the meaning of the present invention, is meant more particularly any compound which improves the maintenance in suspension of the metallic nanoparticles.
Dans un mode de réalisation de l'invention, le dispersant peut être choisi parmi les composés comprenant au moins un groupement succinimide, les polyoléfines, les 20 copolymères oléfines (OCP), les copolymères comprenant au moins un motif styrène, les polyacrylates ou leurs dérivés.In one embodiment of the invention, the dispersant can be chosen from compounds comprising at least one succinimide group, polyolefins, olefin copolymers (OCP), copolymers comprising at least one styrene unit, polyacrylates or their derivatives. .
Par dérivés, on entend tout composé comprenant au moins un groupement ou une chaîne polymérique tels que définis ci-dessus.By derivatives is meant any compound comprising at least one group or one polymeric chain as defined above.
De manière avantageuse, le dispersant selon l'invention est choisi parmi les composés comprenant au moins un groupement succinimide.Advantageously, the dispersant according to the invention is chosen from compounds comprising at least one succinimide group.
Dans un mode de réalisation préféré de l'invention, le dispersant est choisi parmi les composés comprenant au moins un groupement succinimide substitué ou les composés comprenant au moins deux groupements succinimide substitués, les groupements succinimides étant reliés au niveau de leur sommet portant un atome d'azote par un groupement polyamine.In a preferred embodiment of the invention, the dispersant is chosen from compounds comprising at least one substituted succinimide group or compounds comprising at least two substituted succinimide groups, the succinimide groups being linked at their top bearing a d atom. nitrogen by a polyamine group.
Par groupement succinimide substitué au sens de la présente invention, on entend un groupement succinimide dont au moins un des sommets carboné est substitué par un groupement hydrocarboné comprenant de 8 à 400 atomes de carbone.By substituted succinimide group within the meaning of the present invention, is meant a succinimide group of which at least one of the carbon vertices is substituted by a hydrocarbon group comprising from 8 to 400 carbon atoms.
Dans un mode de réalisation préféré de l'invention, le dispersant est choisi parmi les polyisobutylène succinimide-polyamineIn a preferred embodiment of the invention, the dispersant is chosen from polyisobutylene succinimide-polyamine
De manière avantageuse, le dispersant selon l'invention a une masse moléculaire moyenne en poids allant de 2000 à 15000 Daltons, de préférence allant de 2500 à 10000 Daltons, avantageusement de 3000 à 7000 Daltons.Advantageously, the dispersant according to the invention has a weight-average molecular mass ranging from 2000 to 15000 Daltons, preferably ranging from 2500 to 10000 Daltons, advantageously from 3000 to 7000 Daltons.
De manière également avantageuse, le dispersant a une masse moléculaire en nombre supérieure ou égale à 1000 Daltons, de préférence allant de 1000 à 5000 Daltons, plus préférentiellement de 1800 à 3500 Daltons, avantageusement de 1800 à 3000 Daltons. Selon l'invention, la masse moléculaire en nombre du dispersant est évaluée selon la norme ASTM D5296.Also advantageously, the dispersant has a number molecular mass greater than or equal to 1000 Daltons, preferably ranging from 1000 to 5000 Daltons, more preferably from 1800 to 3500 Daltons, advantageously from 1800 to 3000 Daltons. According to the invention, the number molecular weight of the dispersant is evaluated according to the ASTM D5296 standard.
Dans un mode de réalisation préféré de l'invention, la teneur en poids de dispersant ayant une masse moléculaire moyenne en poids supérieure ou égale à 2000 Daltons 20 va de 0,1 à 10%, de préférence de 0,1 à 5%, avantageusement de 0,1 à 3% par rapport au poids total de la composition lubrifiante.In a preferred embodiment of the invention, the content by weight of dispersant having a weight average molecular mass greater than or equal to 2000 Daltons ranges from 0.1 to 10%, preferably from 0.1 to 5%, advantageously from 0.1 to 3% relative to the total weight of the lubricating composition.
Les polymères peuvent être choisis dans le groupe des polymères stables au cisaillement, de préférence dans le groupe constitué par les copolymères d'éthylène et d'alpha-oléfine, les polyacrylates tels que les polyméthacrylates, les oléfines copolymères (OCP), les Ethylène Propylène Diène Monomères (EPDM), les polybutènes, les copolymères de styrène et d'oléfine, hydrogénés ou non ou les copolymères de styrène et d'acrylate.The polymers can be chosen from the group of shear-stable polymers, preferably from the group consisting of copolymers of ethylene and alpha-olefin, polyacrylates such as polymethacrylates, olefin copolymers (OCP), Ethylene Propylene Diene Monomers (EPDM), polybutenes, copolymers of styrene and olefin, hydrogenated or not, or copolymers of styrene and acrylate.
Les antioxydants peuvent être choisis parmi les antioxydants aminés, de préférence les diphénylamines, en particulier des dialkylphénylamines, telles que les octadiphénylamines, les phényl-alpha-naphtyl aminés, les antioxydants phénoliques (dibutylhydroxytoluène BHT et dérivés) ou des antioxydants soufrés (phénates sulfurisés).The antioxidants can be chosen from amino antioxidants, preferably diphenylamines, in particular dialkylphenylamines, such as octadiphenylamines, phenyl-alpha-naphthyl amines, phenolic antioxidants (dibutylhydroxytoluene BHT and derivatives) or sulfur antioxidants (sulphurized phenates) .
Les modificateurs de frottement peuvent être des composés apportant des éléments métalliques différents des nanoparticules métalliques selon l'invention ou bien un composé sans cendres. Parmi les composés apportant des éléments métalliques, on peut citer les complexes de métaux de transition tels que Mo, Sb, Sn, Fe, Cu, Zn, dont les ligands peuvent être des composés hydrocarbonés contenant des atomes d'oxygène, azote, soufre ou phosphore, tels que les dithiocarbamates ou dithiophosphates de molybdène. Les modificateurs de frottement sans cendres sont d'origine organique et peuvent être choisis parmi les monoesters d'acides gras et de polyols, les amines alcoxylées, les amines alcoxylées grasses, les phosphates d'amine, les alcools gras, les époxydes gras, les époxydes gras de borate, les amines grasses ou les esters de glycérol d'acide gras. Par « gras » ou « grasse(s) » on entend au sens de la présente invention un groupement hydrocarboné comprenant de 8 à 24 atomes de carbone.The friction modifiers can be compounds providing metallic elements different from the metallic nanoparticles according to the invention or else an ashless compound. Among the compounds providing metallic elements, mention may be made of complexes of transition metals such as Mo, Sb, Sn, Fe, Cu, Zn, the ligands of which may be hydrocarbon compounds containing oxygen, nitrogen, sulfur or phosphorus, such as dithiocarbamates or molybdenum dithiophosphates. The ashless friction modifiers are of organic origin and can be chosen from monoesters of fatty acids and polyols, alkoxylated amines, fatty alkoxylated amines, amine phosphates, fatty alcohols, fatty epoxides, borate fatty epoxides, fatty amines or fatty acid glycerol esters. By “fat” or “fat(s)” is meant within the meaning of the present invention a hydrocarbon group comprising from 8 to 24 carbon atoms.
Les additifs anti-corrosion peuvent être choisis parmi les dérivés phénoliques, en particulier des dérivés phénoliques éthoxylés et substitués par des groupements alkyles en position ortho. Les inhibiteurs de corrosion pourront être des dérivés du dimercaptothiadiazole.The anti-corrosion additives can be chosen from phenolic derivatives, in particular ethoxylated phenolic derivatives substituted by alkyl groups in the ortho position. The corrosion inhibitors may be derivatives of dimercaptothiadiazole.
Dans un mode de réalisation de l'invention, la composition lubrifiante comprend :
- de 75 à 99,89% d'au moins une huile de base,
- de 0,01 à 2% de nanoparticules métalliques telles que définies ci-dessus,
- de 0,1 à 5% d'au moins un composé anti-usure comprenant un groupement dithiophosphate.
- from 75 to 99.89% of at least one base oil,
- from 0.01 to 2% of metallic nanoparticles as defined above,
- from 0.1 to 5% of at least one anti-wear compound comprising a dithiophosphate group.
Dans un autre mode de réalisation de l'invention, la composition lubrifiante consiste essentiellement en :
- 75 à 99,89% d'au moins une huile de base,
- 0,01 à 2% de nanoparticules métalliques telles que définies ci-dessus,
- 0,1 à 5% d'au moins un composé anti-usure comprenant un groupement dithiophosphate.
- 75 to 99.89% of at least one base oil,
- 0.01 to 2% of metallic nanoparticles as defined above,
- 0.1 to 5% of at least one anti-wear compound comprising a dithiophosphate group.
L'ensemble des caractéristiques et préférences présentées pour l'huile de base, les nanoparticules métalliques et le composé anti-usure comprenant un groupement dithiophosphate s'applique également aux compositions lubrifiantes ci-dessus.All of the characteristics and preferences presented for the base oil, the metallic nanoparticles and the anti-wear compound comprising a dithiophosphate group also apply to the lubricating compositions above.
Dans un mode de réalisation de l'invention, la composition lubrifiante n'est pas une émulsion.In one embodiment of the invention, the lubricating composition is not an emulsion.
Dans un autre mode de réalisation de l'invention, la composition lubrifiante est anhydre.In another embodiment of the invention, the lubricating composition is anhydrous.
La demande décrit également une huile moteur comprenant une composition lubrifiante selon l'invention.The application also describes an engine oil comprising a lubricating composition according to the invention.
La demande décrit également une huile transmission comprenant une composition lubrifiante selon l'invention.The application also describes a transmission oil comprising a lubricating composition according to the invention.
L'ensemble des caractéristiques et préférences présentées pour la composition lubrifiante s'applique également à l'huile moteur ou à l'huile transmission.All the characteristics and preferences presented for the lubricating composition also apply to engine oil or transmission oil.
La composition lubrifiante selon l'invention peut lubrifier au moins une pièce mécanique ou un organe mécanique, notamment des roulements, des engrenages, des joints de cardan, des transmissions, le système pistons/segments/chemises, les arbres à came, l'embrayage, les boîtes de vitesses manuelles ou automatiques, les ponts, les culbuteurs, les carters etc.The lubricating composition according to the invention can lubricate at least one mechanical part or one mechanical component, in particular bearings, gears, universal joints, transmissions, the piston/segment/liner system, the camshafts, the clutch , manual or automatic gearboxes, axles, rocker arms, crankcases, etc.
Dans un mode de réalisation préféré, la composition lubrifiante selon l'invention peut lubrifier une pièce mécanique ou un organe métallique des transmissions, de l'embrayage, des ponts, des boîtes de vitesses manuelles ou automatiques, de préférence manuelles.In a preferred embodiment, the lubricating composition according to the invention can lubricate a mechanical part or a metal component of the transmissions, of the clutch, of the axles, of manual or automatic, preferably manual, gearboxes.
Ainsi, l'invention a également pour objet l'utilisation d'une composition lubrifiante telle que définie ci-dessus pour la lubrification d'une pièce mécanique, préférentiellement d'un organe de transmission ou d'un moteur de véhicules, avantageusement de véhicules automobiles.Thus, the invention also relates to the use of a lubricating composition as defined above for lubricating a mechanical part, preferably a transmission member or a vehicle engine, advantageously vehicles automobiles.
L'invention a également pour l'utilisation d'une composition lubrifiante telle que définie ci-dessus pour la réduction des frottements à la surface d'une pièce mécanique, préférentiellement d'un organe de transmission ou d'un moteur de véhicules, avantageusement de véhicules automobiles.The invention also relates to the use of a lubricating composition as defined above for reducing friction on the surface of a mechanical part, preferably a transmission member or a vehicle engine, advantageously of motor vehicles.
L'invention a également pour objet l'utilisation d'une composition lubrifiante telle que définie ci-dessus pour réduire la consommation de carburant de véhicules, en particulier de véhicules automobiles.A subject of the invention is also the use of a lubricating composition as defined above for reducing the fuel consumption of vehicles, in particular motor vehicles.
L'invention a également pour objet l'utilisation d'une composition lubrifiante telle que définie ci-dessus pour réduire l'écaillage d'une pièce mécanique, préférentiellement d'un organe de transmission ou d'un moteur de véhicules, avantageusement de véhicules automobiles.A subject of the invention is also the use of a lubricating composition as defined above for reducing the spalling of a mechanical part, preferably of a transmission member or of a vehicle engine, advantageously of vehicles automobiles.
L'ensemble des caractéristiques et préférences présentées pour la composition lubrifiante s'applique également aux utilisations ci-dessus.All of the characteristics and preferences presented for the lubricating composition also apply to the above uses.
La demande décrit également un procédé de lubrification d'une pièce mécanique, préférentiellement d'un organe de transmission ou d'un moteur de véhicules, avantageusement de véhicules automobiles, ledit procédé comprenant au moins une étape de mise en contact de la pièce mécanique avec une composition lubrifiante telle que définie ci-dessus.The application also describes a process for lubricating a mechanical part, preferably a transmission member or a vehicle engine, advantageously motor vehicles, said process comprising at least one step of bringing the mechanical part into contact with a lubricating composition as defined above.
La demande décrit également un procédé de réduction des frottements à la surface d'une pièce mécanique, préférentiellement d'un organe de transmission ou d'un moteur de véhicules, avantageusement de véhicules automobiles, comprenant au moins la mise en contact de la pièce mécanique avec une composition lubrifiante telle que définie ci-dessus.The application also describes a method for reducing friction on the surface of a mechanical part, preferably of a transmission member or of a vehicle engine, advantageously of motor vehicles, comprising at least bringing the mechanical part into contact with a lubricating composition as defined above.
La demande décrit également un procédé pour réduire la consommation de carburant d'un véhicule, en particulier d'un véhicule automobile comprenant au moins une étape de mise en contact d'une pièce mécanique du moteur du véhicule avec une composition lubrifiante telle que définie ci-dessus.The application also describes a method for reducing the fuel consumption of a vehicle, in particular of a motor vehicle comprising at least one step of in contact with a mechanical part of the vehicle engine with a lubricating composition as defined above.
La demande décrit également un procédé de réduction de l'écaillage d'une pièce mécanique préférentiellement d'un organe de transmission ou d'un moteur de véhicules, avantageusement de véhicules automobiles, comprenant au moins la mise en contact de la pièce mécanique avec une composition lubrifiante telle que définie ci-dessus.The application also describes a method for reducing the spalling of a mechanical part, preferably of a transmission member or of a vehicle engine, advantageously of motor vehicles, comprising at least bringing the mechanical part into contact with a lubricating composition as defined above.
L'ensemble des caractéristiques et préférences présentées pour la composition lubrifiante s'applique également aux procédés ci-dessus.All of the characteristics and preferences presented for the lubricating composition also apply to the above methods.
La demande décrit également une composition de type concentré d'additifs comprenant au moins un composé anti-usure comprenant un groupement dithiophosphate et des nanoparticules de bisulfure de tungstène.The application also describes a composition of the concentrate type of additives comprising at least one anti-wear compound comprising a dithiophosphate group and tungsten disulphide nanoparticles.
L'ensemble des caractéristiques et préférences présentées pour les nanoparticules de bisulfure de tungstène et le composé anti-usure comprenant un groupement dithiophosphate s'applique également à la composition de type concentré d'additifs ci-dessus.All of the characteristics and preferences presented for the tungsten disulphide nanoparticles and the anti-wear compound comprising a dithiophosphate group also apply to the composition of the concentrate type of additives above.
Dans un mode de réalisation de l'invention, à la composition de type concentré d'additifs peut être ajoutée au moins une huile base pour obtenir une composition lubrifiante selon l'invention.In one embodiment of the invention, to the composition of the concentrate type of additives can be added at least one base oil to obtain a lubricating composition according to the invention.
L'ensemble des caractéristiques et préférences présentées pour l'huile de base s'applique également au mode de réalisation ci-dessus.All of the characteristics and preferences presented for the base oil also apply to the above embodiment.
L'invention a également pour objet l'utilisation d'un composé anti-usure comprenant un groupement dithiophosphate pour diminuer l'oxydation d'une composition lubrifiante comprenant au moins une huile de base et des nanoparticules métalliques solides ayant une structure de type fullerène représentées par la formule MXn dans laquelle M représente un métal de transition, X un chalcogène, avec n=2 ou n=3 en fonction de l'état d'oxydation du métal de transition, dans laquelle les nanoparticules métalliques présentent une taille moyenne, déterminée à l'aide d'images obtenues par micrographie électronique à transmission ou par microscopie électronique à transmission à haute résolution, de 50 à 200 nm et sont des polyèdres concentriques avec une structure multicouche ou en feuillets L'ensemble des caractéristiques et préférences présentées pour l'huile de base, les nanoparticules métalliques et le composé anti-usure comprenant un groupement dithiophosphate s'applique également à l'utilisation ci-dessusThe invention also relates to the use of an anti-wear compound comprising a dithiophosphate group to reduce the oxidation of a lubricating composition comprising at least one base oil and solid metallic nanoparticles having a fullerene-type structure represented by the formula MXn in which M represents a transition metal, X a chalcogen, with n=2 or n=3 depending on the oxidation state of the transition metal, in which the metallic nanoparticles have an average size, determined using images obtained by transmission electron micrography or by transmission electron microscopy at high resolution, from 50 to 200 nm and are concentric polyhedra with a multilayer or sheet structure All of the characteristics and preferences presented for the base oil, the metallic nanoparticles and the anti-wear compound comprising a dithiophosphate group also apply to the above use.
Les différents objets de la présente invention et leurs mises en œuvre seront mieux compris à la lecture des exemples qui suivent. Ces exemples sont donnés à titre indicatif, sans caractère limitatif.The various objects of the present invention and their implementations will be better understood on reading the examples which follow. These examples are given for information only, without limitation.
On a préparé les compositions lubrifiantes N°1 à N°4 à partir des composés suivants :
- une huile de base de type PAO (Poly Alpha Oléfine) de grade 6 (viscosité à 100°C aux environs de 6 cSt mesuré selon la norme ASTM D445),
- un mélange de nanoparticules de bisulfure de tungstène à 20% en matière active dans une huile (NanoLub Gear Oil Concentrate commercialisé par la société Nanomaterials),
- un composé comprenant un groupement dithiophosphate : dithiophosphate de zinc (Lz 1371 commercialisé par la société Lubrizol).
- a grade 6 PAO (Poly Alpha Olefin) type base oil (viscosity at 100°C around 6 cSt measured according to the ASTM D445 standard),
- a mixture of tungsten disulphide nanoparticles with 20% active ingredient in an oil (NanoLub Gear Oil Concentrate marketed by the company Nanomaterials),
- a compound comprising a dithiophosphate group: zinc dithiophosphate (Lz 1371 marketed by the company Lubrizol).
Les compositions lubrifiantes N°1 à N°4 sont décrites dans le tableau II ; les pourcentages indiqués sont des pourcentages massiques.
Il s'agit dévaluer les propriétés de frottement des compositions lubrifiantes N°1 à N°4 par la mesure du coefficient de frottement.This involves evaluating the friction properties of lubricating compositions No. 1 to No. 4 by measuring the coefficient of friction.
Le coefficient de frottement est évalué à l'aide d'un tribomètre linéaire pion/plan dans les conditions suivantes :
- nature de l'acier : AISI 52100 (dureté = 800 HV),
- rugosité du plan : 35 nm,
- température : 100°C,
- pression calculée de contact : 1,12 GPa,
- vitesse de glissement : 3 mm/s
- taux d'humidité : 35-45R (atmosphère ambiante),
- durée du test : 8h.
- type of steel: AISI 52100 (hardness = 800 HV),
- plane roughness: 35 nm,
- temperature: 100°C,
- calculated contact pressure: 1.12 GPa,
- sliding speed: 3 mm/s
- humidity: 35-45R (ambient atmosphere),
- duration of the test: 8 hours.
Le tableau III présente le coefficient de frottement moyen des compositions lubrifiantes N°1 à N°4 ; le coefficient de frottement moyen représentant la moyenne des valeurs du coefficient de frottement obtenu après 4 tests.
Ces résultats montrent que la composition lubrifiante selon l'invention N°4 présente des propriétés de frottement améliorées, par rapport à une composition lubrifiante comprenant un composé comprenant un groupement dithiophosphate selon l'invention mais ne comprenant pas de nanoparticules métalliques (composition N°2) et par rapport à une composition comprenant des nanoparticules métalliques selon l'invention mais ne comprenant pas de composé comprenant un groupement dithiophosphate (composition N°3).These results show that the lubricating composition according to the invention No. 4 has improved friction properties, compared to a lubricating composition comprising a compound comprising a dithiophosphate group according to the invention but does not not comprising metallic nanoparticles (composition No. 2) and relative to a composition comprising metallic nanoparticles according to the invention but not comprising a compound comprising a dithiophosphate group (composition No. 3).
Ces résultats montrent ainsi une synergie d'activité de la combinaison entre un composé comprenant un groupement dithiophosphate et des nanoparticules métalliques dans une composition lubrifiante pour réduire significativement le coefficient de frottement, notamment pour des contacts acier/acier.These results thus show a synergy of activity of the combination between a compound comprising a dithiophosphate group and metallic nanoparticles in a lubricating composition to significantly reduce the coefficient of friction, in particular for steel/steel contacts.
Ces résultats montrent également que l'efficacité de réduction des frottements est maintenue dans le temps par l'utilisation d'une composition lubrifiante selon l'invention.These results also show that the effectiveness of reducing friction is maintained over time by the use of a lubricating composition according to the invention.
Par ailleurs, la composition lubrifiante N°4 présente une stabilité satisfaisante.Furthermore, lubricating composition No. 4 exhibits satisfactory stability.
Claims (13)
- Lubricant composition with kinematic viscosity at 100°C measured according to standard ASTM D445 ranging from 4 to 50 cSt and comprising, with respect to the total weight of the lubricant composition, at least 70% by weight of at least one base oil, from 0.1 to 5% by weight of one at least one anti-wear compound comprising a dithiophosphate group and from 0.01 to 2% by weight of solid metal nanoparticles having a fullerene-type structure represented by the formula MXn in which M represents a transition metal, X a chalcogen, with n=2 or n=3 depending on the oxidation state of the transition metal M, the metal nanoparticles having an average size, determined using images obtained by transmission electron microscopy or by high resolution transmission electron microscopy, ranging from 50 to 200 nm and being concentric polyhedrons with a multilayer or sheet structure.
- Lubricant composition according to claim 1, wherein M is selected from the group constituted by tungsten, molybdenum, zirconium, hafnium, platinum, rhenium, titanium, tantalum and niobium.
- Lubricant composition according to claim 1 or 2, wherein the metal nanoparticles are selected from the group constituted by MoS2, MoSe2, MoTe2, WS2, WSe2, ZrS2, ZrSe2, HfS2, HfSe2, PtS2, ReS2, ReSe2, TiS3, ZrS3, ZrSe3, HfS3, HfSe3, TiS2, TaS2, TaSe2, NbS2, NbSe2 and NbTe2.
- Lubricant composition according to any one of the preceding claims, wherein the content by weight of metal nanoparticles ranges from 0.05 to 2%, with respect to the total weight of the lubricant composition.
- Lubricant composition according to any one of the preceding claims, wherein the the compound comprising a dithiophosphate group is selected from the group constituted by ammonium dithiophosphates, amine dithiophosphates, ester dithiophosphates and metal dithiophosphates, alone or in a mixture.
- Composition according to any one of the preceding claims, wherein the compound comprising a dithiophosphate group is a compound of formula (IV)
- Lubricant composition according to any one of the preceding claims, wherein the compound comprising a dithiophosphate group is a compound of formula (IV-a) or of formula (IV-b):
- Lubricant composition according to any one of the preceding claims, wherein the content by weight of the compound comprising a dithiophosphate group ranges from 0.2 to 4%, with respect to the total weight of the lubricant composition.
- Lubricant composition according to any one of the preceding claims, further comprising an additive selected from the shear-stable polymers, the antioxidants, the anti-corrosion additives, the friction modifiers different from metal nanoparticles, and the dispersants.
- Use of a lubricant composition according to any one of claims 1 to 9 for the lubrication of a mechanical part.
- Use of a lubricant composition according to the preceding claim for the lubrication of a mechanical part of motor vehicles.
- Use of a lubricant composition according to any one of claims 1 to 9 for reducing the fuel consumption of vehicles.
- Use of an anti-wear compound comprising a dithiophosphate group for decreasing the oxidation of a lubricant composition comprising at least one base oil and solid metal nanoparticles having a fullerene-type structure represented by the formula MXn in which M represents a transition metal, X a chalcogen, with n=2 or n=3 depending on the oxidation state of the transition metal M, the metal nanoparticles having an average size, determined using images obtained by transmission electron microscopy or by high resolution transmission electron microscopy, ranging from 50 to 200 nm and being concentric polyhedrons with a multilayer or sheet structure.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| FR1451648A FR3018079B1 (en) | 2014-02-28 | 2014-02-28 | LUBRICATING COMPOSITION BASED ON METALLIC NANOPARTICLES |
| PCT/EP2015/054099 WO2015128444A1 (en) | 2014-02-28 | 2015-02-26 | Lubricating composition based on metal nanoparticles |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP3110929A1 EP3110929A1 (en) | 2017-01-04 |
| EP3110929B1 true EP3110929B1 (en) | 2022-03-30 |
Family
ID=50639756
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP15706812.3A Active EP3110929B1 (en) | 2014-02-28 | 2015-02-26 | Lubricating composition based on metal nanoparticles |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US11015141B2 (en) |
| EP (1) | EP3110929B1 (en) |
| JP (1) | JP2017506694A (en) |
| KR (1) | KR102308393B1 (en) |
| CN (1) | CN106103670A (en) |
| FR (1) | FR3018079B1 (en) |
| WO (1) | WO2015128444A1 (en) |
Families Citing this family (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2015172846A1 (en) * | 2014-05-16 | 2015-11-19 | Ab Nanol Technologies Oy | Additive composition for lubricants |
| EP3400196B1 (en) * | 2016-01-05 | 2021-09-22 | Nanotech Industrial Solutions, Inc. | Water based nanoparticle dispersion |
| US11015140B2 (en) * | 2017-03-31 | 2021-05-25 | Uchicago Argonne, Llc | Catalytically active lubricants |
| CN111655827B (en) * | 2018-01-23 | 2022-07-26 | 赢创运营有限公司 | Polymer-inorganic nanoparticle compositions, methods of manufacture thereof, and use thereof as lubricant additives |
| KR101973490B1 (en) * | 2018-02-08 | 2019-09-02 | 이승우 | Lubricating additives composition, methode for preparing the same and lubricant oil comprising the same |
| KR102089942B1 (en) * | 2019-05-09 | 2020-03-18 | (주)에코시즌 | composition of oil for transmission |
| JP7434953B2 (en) | 2019-06-13 | 2024-02-21 | 株式会社ジェイテクト | Grease compositions and rolling bearings |
| EP3839022A1 (en) * | 2019-12-20 | 2021-06-23 | Total Marketing Services | Lubricating composition for improving fuel eco and reducing friction |
| CN111979016A (en) * | 2020-08-03 | 2020-11-24 | 容嘉和 | Engine oil additive |
| JP2023038798A (en) * | 2021-09-07 | 2023-03-17 | 三菱重工業株式会社 | Coating film forming method, and lubricant composition |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2910911A1 (en) * | 2007-01-03 | 2008-07-04 | Afton Chemical Corp | Reducing friction and wear on lubricated surfaces, especially moving parts of engines in vehicles, involves using base lubricating oil containing dispersed metal oxide or sulfide nano-particles, e.g. cerium oxide |
Family Cites Families (39)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4904401A (en) * | 1988-06-13 | 1990-02-27 | The Lubrizol Corporation | Lubricating oil compositions |
| ES2135427T3 (en) | 1992-07-08 | 1999-11-01 | Yeda Res & Dev | THIN AND FILM-ORIENTED FILMS OF CALGOGENURES OF A TRANSITIONAL METAL. |
| ES2329446T3 (en) | 2002-07-30 | 2009-11-26 | Chevron Oronite S.A. | ADDITIVE COMPOSITION FOR TRANSMISSION OIL CONTAINING A HYDRAATED ALKALINE METAL BORATE AND HEXAGONAL BORUS NITRIDE. |
| ATE420940T1 (en) | 2002-10-01 | 2009-01-15 | Lubrizol Corp | PRODUCTION OF LUBRICANTS FROM DEWATERED EMULSIONS CONTAINING METAL HYDROXIDE |
| FR2848668B1 (en) | 2002-12-16 | 2005-03-18 | Totalfinaelf France | METHOD AND DEVICE FOR THE CONTINUOUS DETERMINATION OF THE DEGRADATION OF THERMAL ENGINE EXHAUST GAS POST-TREATMENT SYSTEMS |
| EP1535987B1 (en) | 2003-11-28 | 2013-01-09 | Chevron Oronite SAS | Additive composition for transmission oil containing hexagonal boron nitride and a viscosity index improver |
| FR2864240B1 (en) | 2003-12-23 | 2006-03-17 | Total France | METHOD AND DEVICE FOR MONITORING THE DILUTION OF LUBRICATING OIL BY FUEL IN AN INTERNAL COMBUSTION ENGINE |
| FR2867564B1 (en) | 2004-03-11 | 2006-06-23 | Total France | METHOD AND DEVICE FOR REAL-TIME MEASUREMENT OF THE OIL CONSUMPTION OF THE ENGINE OIL SEPARATION SYSTEM |
| JP2009509027A (en) | 2005-09-20 | 2009-03-05 | ザ ルブリゾル コーポレイション | Method for lubricating an internal combustion engine |
| CA2634079A1 (en) | 2005-12-20 | 2007-10-25 | The Lubrizol Corporation | Method of preparing an overbased or neutral detergent |
| JP4886304B2 (en) | 2006-01-27 | 2012-02-29 | 昭和シェル石油株式会社 | Grease composition |
| ES2666595T3 (en) | 2006-10-24 | 2018-05-07 | Total Marketing Services | Use of a multifunctional lubricating fluid |
| US20080234149A1 (en) * | 2007-01-12 | 2008-09-25 | Malshe Ajay P | Nanoparticulate based lubricants |
| US20080182927A1 (en) | 2007-01-31 | 2008-07-31 | Air Products And Chemicals, Inc. | Polyisobutenyl containing dispersions and uses thereof |
| JP2009063154A (en) | 2007-09-10 | 2009-03-26 | Nsk Ltd | Rolling device |
| FR2924439B1 (en) | 2007-12-03 | 2010-10-22 | Total France | LUBRICATING COMPOSITION FOR FOUR-STROKE ENGINE WITH LOW ASH RATES |
| FR2932813B1 (en) | 2008-06-18 | 2010-09-03 | Total France | LUBRICANT CYLINDER FOR MARINE ENGINE TWO TIMES |
| FR2942627B1 (en) | 2009-02-27 | 2011-05-06 | Total Raffinage Marketing | FAT COMPOSITION |
| FR2945754A1 (en) | 2009-05-20 | 2010-11-26 | Total Raffinage Marketing | NEW ADDITIVES FOR TRANSMISSION OILS |
| FR2949786B1 (en) | 2009-09-10 | 2013-07-05 | Total Raffinage Marketing | GREASE COMPOSITION. |
| CN101691517B (en) * | 2009-09-29 | 2012-12-19 | 中南大学 | Vehicle engine oil additive and engine oil |
| PL218093B1 (en) | 2009-12-30 | 2014-10-31 | Inst Obróbki Plastycznej | Process for the preparation of nanocomposite graphene-like greases and unit for the preparation of nanocomposite graphene-like greases |
| FR2961823B1 (en) | 2010-06-25 | 2013-06-14 | Total Raffinage Marketing | LUBRICATING COMPOSITIONS FOR AUTOMOTIVE TRANSMISSIONS |
| FR2965274A1 (en) | 2010-09-28 | 2012-03-30 | Total Raffinage Marketing | LUBRICANT COMPOSITION |
| FR2968011B1 (en) | 2010-11-26 | 2014-02-21 | Total Raffinage Marketing | LUBRICATING COMPOSITION FOR ENGINE |
| FR2968670B1 (en) | 2010-12-13 | 2013-01-04 | Total Raffinage Marketing | FAT COMPOSITION |
| FR2968669B1 (en) * | 2010-12-13 | 2014-02-28 | Total Raffinage Marketing | FAT COMPOSITION |
| FR2980799B1 (en) | 2011-09-29 | 2013-10-04 | Total Raffinage Marketing | LUBRICATING COMPOSITION FOR MARINE ENGINE |
| PL2766427T3 (en) | 2011-10-13 | 2017-10-31 | Total Res & Technology Feluy | Nanocomposite |
| US9321905B2 (en) | 2011-10-13 | 2016-04-26 | Total Research & Technology Feluy | Nanocomposite |
| FR2984348B1 (en) | 2011-12-16 | 2015-02-27 | Total Raffinage Marketing | LUBRICATING COMPOSITIONS FOR TRANSMISSIONS |
| FR2986801B1 (en) * | 2012-02-15 | 2014-09-05 | Total Raffinage Marketing | LUBRICATING COMPOSITIONS FOR TRANSMISSIONS |
| FR2990213B1 (en) | 2012-05-04 | 2015-04-24 | Total Raffinage Marketing | LUBRICATING COMPOSITION FOR ENGINE |
| FR2998303B1 (en) | 2012-11-16 | 2015-04-10 | Total Raffinage Marketing | LUBRICANT COMPOSITION |
| US20140162915A1 (en) * | 2012-12-11 | 2014-06-12 | N1 Technologies Inc | Enhanced Lubricant Formulation |
| FR3002235B1 (en) | 2013-02-19 | 2015-02-27 | Total Raffinage Marketing | LUBRICANT COMPOSITION BASED ON AMINO COMPOUNDS |
| FR3004723B1 (en) | 2013-04-19 | 2016-04-15 | Total Raffinage Marketing | LUBRICATING COMPOSITION BASED ON METALLIC NANOPARTICLES |
| FR3005474B1 (en) | 2013-05-07 | 2016-09-09 | Total Raffinage Marketing | LUBRICANT FOR MARINE ENGINE |
| FR3009309B1 (en) | 2013-08-02 | 2016-10-07 | Total Marketing Services | LUBRICATING COMPOSITIONS FOR TRANSMISSIONS |
-
2014
- 2014-02-28 FR FR1451648A patent/FR3018079B1/en active Active
-
2015
- 2015-02-26 CN CN201580010884.1A patent/CN106103670A/en active Pending
- 2015-02-26 JP JP2016554406A patent/JP2017506694A/en active Pending
- 2015-02-26 WO PCT/EP2015/054099 patent/WO2015128444A1/en active Application Filing
- 2015-02-26 KR KR1020167025416A patent/KR102308393B1/en active IP Right Grant
- 2015-02-26 US US15/121,987 patent/US11015141B2/en active Active
- 2015-02-26 EP EP15706812.3A patent/EP3110929B1/en active Active
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| FR2910911A1 (en) * | 2007-01-03 | 2008-07-04 | Afton Chemical Corp | Reducing friction and wear on lubricated surfaces, especially moving parts of engines in vehicles, involves using base lubricating oil containing dispersed metal oxide or sulfide nano-particles, e.g. cerium oxide |
Non-Patent Citations (1)
| Title |
|---|
| JENEI ISTVAN ZOLTAN ET AL: "Correlation Studies ofWS2Fullerene-Like Nanoparticles Enhanced Tribofilms: A Scanning Electron Microscopy Analysis", TRIBOLOGY LETTERS, BALTZER SCIENCE PUBLISHIERS, NL, vol. 51, no. 3, 11 July 2013 (2013-07-11), pages 461 - 468, XP035350080, ISSN: 1023-8883, [retrieved on 20130711], DOI: 10.1007/S11249-013-0184-0 * |
Also Published As
| Publication number | Publication date |
|---|---|
| JP2017506694A (en) | 2017-03-09 |
| KR102308393B1 (en) | 2021-10-06 |
| US20170073612A1 (en) | 2017-03-16 |
| WO2015128444A1 (en) | 2015-09-03 |
| FR3018079B1 (en) | 2017-06-23 |
| EP3110929A1 (en) | 2017-01-04 |
| CN106103670A (en) | 2016-11-09 |
| FR3018079A1 (en) | 2015-09-04 |
| US11015141B2 (en) | 2021-05-25 |
| KR20160127034A (en) | 2016-11-02 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP3110929B1 (en) | Lubricating composition based on metal nanoparticles | |
| EP2986693B1 (en) | Lubricant composition based on metal nanoparticles | |
| EP2401349B1 (en) | Grease composition | |
| EP2652099B1 (en) | Grease composition | |
| EP2652100B1 (en) | Grease composition | |
| CA2771772C (en) | Fat composition | |
| EP3274432A1 (en) | Lubricating composition | |
| EP3027719A1 (en) | Lubricant compositions for transmissions | |
| EP2958980A1 (en) | Lubricating composition based on aminated compounds | |
| EP2935542A1 (en) | Lubricating composition made from polyglycerol ether | |
| EP3947610A1 (en) | Use of a lubricant composition for transmission | |
| EP3529341B1 (en) | Lubricant composition | |
| FR3065008A1 (en) | PROCESS FOR LUBRICATING MECHANICAL PARTS | |
| WO2020152137A1 (en) | Dinuclear molybdenum complex and use of same in lubricating compositions |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: THE INTERNATIONAL PUBLICATION HAS BEEN MADE |
|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: REQUEST FOR EXAMINATION WAS MADE |
|
| 17P | Request for examination filed |
Effective date: 20160906 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): AL AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL PT RO RS SE SI SK SM TR |
|
| AX | Request for extension of the european patent |
Extension state: BA ME |
|
| RIN1 | Information on inventor provided before grant (corrected) |
Inventor name: THIEBAUT, BENOIT Inventor name: DASSENOY, FABRICE Inventor name: USSA, PAULA |
|
| DAX | Request for extension of the european patent (deleted) | ||
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: EXAMINATION IS IN PROGRESS |
|
| 17Q | First examination report despatched |
Effective date: 20200214 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: EXAMINATION IS IN PROGRESS |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R079 Ref document number: 602015077841 Country of ref document: DE Free format text: PREVIOUS MAIN CLASS: C10M0141100000 Ipc: C10M0171060000 |
|
| RIC1 | Information provided on ipc code assigned before grant |
Ipc: C10N 50/00 20060101ALI20210901BHEP Ipc: C10N 40/04 20060101ALI20210901BHEP Ipc: C10N 30/00 20060101ALI20210901BHEP Ipc: C10N 30/10 20060101ALI20210901BHEP Ipc: C10N 30/06 20060101ALI20210901BHEP Ipc: C10N 30/02 20060101ALI20210901BHEP Ipc: C10N 20/06 20060101ALI20210901BHEP Ipc: C10M 141/12 20060101ALI20210901BHEP Ipc: C10M 171/06 20060101AFI20210901BHEP |
|
| GRAP | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOSNIGR1 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: GRANT OF PATENT IS INTENDED |
|
| INTG | Intention to grant announced |
Effective date: 20211018 |
|
| GRAS | Grant fee paid |
Free format text: ORIGINAL CODE: EPIDOSNIGR3 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: THE PATENT HAS BEEN GRANTED |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AL AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL PT RO RS SE SI SK SM TR |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: FG4D Free format text: NOT ENGLISH |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: EP |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R096 Ref document number: 602015077841 Country of ref document: DE |
|
| REG | Reference to a national code |
Ref country code: AT Ref legal event code: REF Ref document number: 1479202 Country of ref document: AT Kind code of ref document: T Effective date: 20220415 |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FG4D Free format text: LANGUAGE OF EP DOCUMENT: FRENCH |
|
| RAP2 | Party data changed (patent owner data changed or rights of a patent transferred) |
Owner name: TOTALENERGIES ONETECH |
|
| REG | Reference to a national code |
Ref country code: LT Ref legal event code: MG9D |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20220330 Ref country code: RS Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20220330 Ref country code: NO Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20220630 Ref country code: LT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20220330 Ref country code: HR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20220330 Ref country code: BG Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20220630 |
|
| REG | Reference to a national code |
Ref country code: NL Ref legal event code: MP Effective date: 20220330 |
|
| REG | Reference to a national code |
Ref country code: AT Ref legal event code: MK05 Ref document number: 1479202 Country of ref document: AT Kind code of ref document: T Effective date: 20220330 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LV Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20220330 Ref country code: GR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20220701 Ref country code: FI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20220330 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: NL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20220330 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SM Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20220330 Ref country code: SK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20220330 Ref country code: RO Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20220330 Ref country code: PT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20220801 Ref country code: ES Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20220330 Ref country code: EE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20220330 Ref country code: CZ Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20220330 Ref country code: AT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20220330 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: PL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20220330 Ref country code: IS Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20220730 Ref country code: AL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20220330 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R097 Ref document number: 602015077841 Country of ref document: DE |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20220330 |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| 26N | No opposition filed |
Effective date: 20230103 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 20230221 Year of fee payment: 9 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20220330 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 20230221 Year of fee payment: 9 Ref country code: DE Payment date: 20230216 Year of fee payment: 9 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20220330 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: MC Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20220330 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| REG | Reference to a national code |
Ref country code: BE Ref legal event code: MM Effective date: 20230228 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LU Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20230226 Ref country code: LI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20230228 Ref country code: CH Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20230228 |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: MM4A |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20230226 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: BE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20230228 |