EP2668977B1 - Golf ball - Google Patents
Golf ball Download PDFInfo
- Publication number
- EP2668977B1 EP2668977B1 EP13168981.2A EP13168981A EP2668977B1 EP 2668977 B1 EP2668977 B1 EP 2668977B1 EP 13168981 A EP13168981 A EP 13168981A EP 2668977 B1 EP2668977 B1 EP 2668977B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- hardness
- core
- acid
- jis
- salt
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 235000019589 hardness Nutrition 0.000 claims description 198
- 229920001971 elastomer Polymers 0.000 claims description 107
- 239000005060 rubber Substances 0.000 claims description 105
- 239000000203 mixture Substances 0.000 claims description 98
- 150000003839 salts Chemical class 0.000 claims description 89
- 229910052751 metal Inorganic materials 0.000 claims description 41
- 239000002184 metal Substances 0.000 claims description 41
- 150000007934 α,β-unsaturated carboxylic acids Chemical class 0.000 claims description 41
- 239000003431 cross linking reagent Substances 0.000 claims description 36
- 238000004132 cross linking Methods 0.000 claims description 23
- 125000004432 carbon atom Chemical group C* 0.000 claims description 16
- 239000003999 initiator Substances 0.000 claims description 12
- 238000000034 method Methods 0.000 claims description 10
- 150000001735 carboxylic acids Chemical class 0.000 claims description 7
- 150000003751 zinc Chemical class 0.000 claims description 7
- RMVRSNDYEFQCLF-UHFFFAOYSA-N thiophenol Chemical compound SC1=CC=CC=C1 RMVRSNDYEFQCLF-UHFFFAOYSA-N 0.000 description 71
- 229920005989 resin Polymers 0.000 description 68
- 239000011347 resin Substances 0.000 description 68
- 229920000554 ionomer Polymers 0.000 description 57
- PPBRXRYQALVLMV-UHFFFAOYSA-N Styrene Chemical compound C=CC1=CC=CC=C1 PPBRXRYQALVLMV-UHFFFAOYSA-N 0.000 description 48
- 150000002898 organic sulfur compounds Chemical class 0.000 description 46
- -1 aluminum ion Chemical class 0.000 description 43
- TZCXTZWJZNENPQ-UHFFFAOYSA-L barium sulfate Chemical compound [Ba+2].[O-]S([O-])(=O)=O TZCXTZWJZNENPQ-UHFFFAOYSA-L 0.000 description 40
- 239000000126 substance Substances 0.000 description 35
- XLOMVQKBTHCTTD-UHFFFAOYSA-N Zinc monoxide Chemical compound [Zn]=O XLOMVQKBTHCTTD-UHFFFAOYSA-N 0.000 description 34
- 238000009826 distribution Methods 0.000 description 32
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 30
- 150000001875 compounds Chemical class 0.000 description 26
- 125000001424 substituent group Chemical group 0.000 description 25
- 229920003182 Surlyn® Polymers 0.000 description 24
- 239000011342 resin composition Substances 0.000 description 24
- 229920002725 thermoplastic elastomer Polymers 0.000 description 22
- 239000002253 acid Substances 0.000 description 21
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 description 20
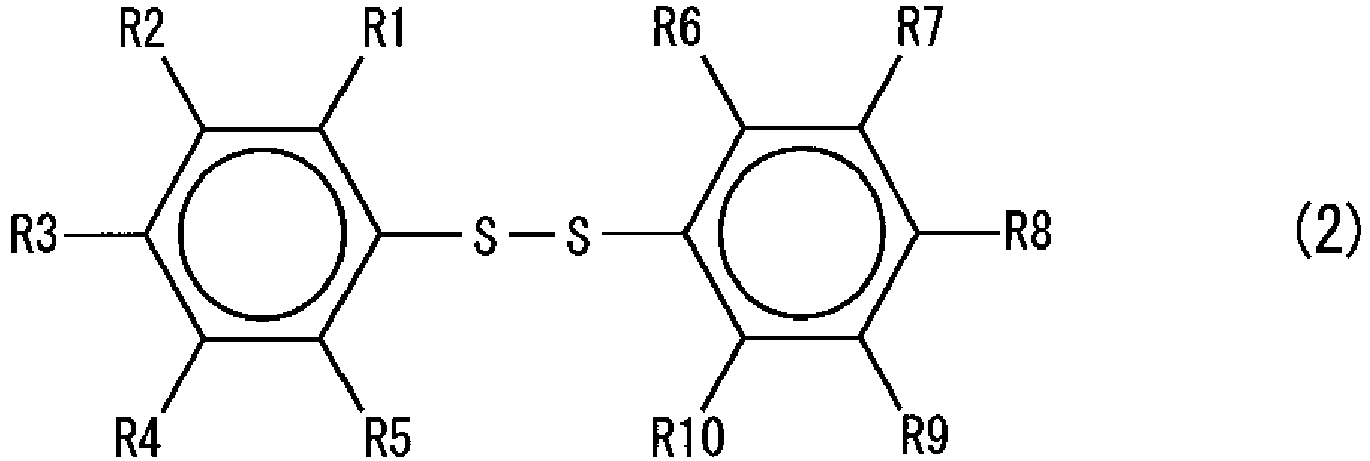
- GUUVPOWQJOLRAS-UHFFFAOYSA-N Diphenyl disulfide Chemical group C=1C=CC=CC=1SSC1=CC=CC=C1 GUUVPOWQJOLRAS-UHFFFAOYSA-N 0.000 description 19
- 229920001577 copolymer Polymers 0.000 description 19
- 150000001732 carboxylic acid derivatives Chemical class 0.000 description 17
- 229910021645 metal ion Inorganic materials 0.000 description 17
- 239000011787 zinc oxide Substances 0.000 description 17
- 229910052742 iron Inorganic materials 0.000 description 16
- 229920005601 base polymer Polymers 0.000 description 14
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 14
- 238000005259 measurement Methods 0.000 description 14
- 150000002736 metal compounds Chemical class 0.000 description 14
- 229920002857 polybutadiene Polymers 0.000 description 14
- 239000005062 Polybutadiene Substances 0.000 description 12
- 239000000945 filler Substances 0.000 description 12
- XOOUIPVCVHRTMJ-UHFFFAOYSA-L zinc stearate Chemical compound [Zn+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O XOOUIPVCVHRTMJ-UHFFFAOYSA-L 0.000 description 12
- XKMZOFXGLBYJLS-UHFFFAOYSA-L zinc;prop-2-enoate Chemical compound [Zn+2].[O-]C(=O)C=C.[O-]C(=O)C=C XKMZOFXGLBYJLS-UHFFFAOYSA-L 0.000 description 11
- XMNIXWIUMCBBBL-UHFFFAOYSA-N 2-(2-phenylpropan-2-ylperoxy)propan-2-ylbenzene Chemical compound C=1C=CC=CC=1C(C)(C)OOC(C)(C)C1=CC=CC=C1 XMNIXWIUMCBBBL-UHFFFAOYSA-N 0.000 description 10
- RFCQDOVPMUSZMN-UHFFFAOYSA-N 2-Naphthalenethiol Chemical compound C1=CC=CC2=CC(S)=CC=C21 RFCQDOVPMUSZMN-UHFFFAOYSA-N 0.000 description 10
- IPCSVZSSVZVIGE-UHFFFAOYSA-N hexadecanoic acid Chemical compound CCCCCCCCCCCCCCCC(O)=O IPCSVZSSVZVIGE-UHFFFAOYSA-N 0.000 description 10
- 238000004898 kneading Methods 0.000 description 10
- 229920000346 polystyrene-polyisoprene block-polystyrene Polymers 0.000 description 10
- 229920006027 ternary co-polymer Polymers 0.000 description 10
- 239000004408 titanium dioxide Substances 0.000 description 10
- 239000004711 α-olefin Substances 0.000 description 10
- CERQOIWHTDAKMF-UHFFFAOYSA-N Methacrylic acid Chemical compound CC(=C)C(O)=O CERQOIWHTDAKMF-UHFFFAOYSA-N 0.000 description 9
- 235000021355 Stearic acid Nutrition 0.000 description 9
- 238000011156 evaluation Methods 0.000 description 9
- QIQXTHQIDYTFRH-UHFFFAOYSA-N octadecanoic acid Chemical compound CCCCCCCCCCCCCCCCCC(O)=O QIQXTHQIDYTFRH-UHFFFAOYSA-N 0.000 description 9
- OQCDKBAXFALNLD-UHFFFAOYSA-N octadecanoic acid Natural products CCCCCCCC(C)CCCCCCCCC(O)=O OQCDKBAXFALNLD-UHFFFAOYSA-N 0.000 description 9
- 239000008117 stearic acid Substances 0.000 description 9
- 229920002126 Acrylic acid copolymer Polymers 0.000 description 8
- KAKZBPTYRLMSJV-UHFFFAOYSA-N Butadiene Chemical compound C=CC=C KAKZBPTYRLMSJV-UHFFFAOYSA-N 0.000 description 8
- VGGSQFUCUMXWEO-UHFFFAOYSA-N Ethene Chemical compound C=C VGGSQFUCUMXWEO-UHFFFAOYSA-N 0.000 description 8
- 239000005977 Ethylene Substances 0.000 description 8
- RRHGJUQNOFWUDK-UHFFFAOYSA-N Isoprene Chemical compound CC(=C)C=C RRHGJUQNOFWUDK-UHFFFAOYSA-N 0.000 description 8
- 150000001336 alkenes Chemical class 0.000 description 8
- 239000000956 alloy Substances 0.000 description 8
- 229910045601 alloy Inorganic materials 0.000 description 8
- UKMSUNONTOPOIO-UHFFFAOYSA-N docosanoic acid Chemical compound CCCCCCCCCCCCCCCCCCCCCC(O)=O UKMSUNONTOPOIO-UHFFFAOYSA-N 0.000 description 8
- POULHZVOKOAJMA-UHFFFAOYSA-N dodecanoic acid Chemical compound CCCCCCCCCCCC(O)=O POULHZVOKOAJMA-UHFFFAOYSA-N 0.000 description 8
- 230000005484 gravity Effects 0.000 description 8
- 238000001746 injection moulding Methods 0.000 description 8
- 239000000463 material Substances 0.000 description 8
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 description 8
- 229920006132 styrene block copolymer Polymers 0.000 description 8
- JLVVSXFLKOJNIY-UHFFFAOYSA-N Magnesium ion Chemical compound [Mg+2] JLVVSXFLKOJNIY-UHFFFAOYSA-N 0.000 description 7
- PTFCDOFLOPIGGS-UHFFFAOYSA-N Zinc dication Chemical compound [Zn+2] PTFCDOFLOPIGGS-UHFFFAOYSA-N 0.000 description 7
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid Chemical compound OC(=O)C1=CC=CC=C1 WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 description 7
- 150000007942 carboxylates Chemical class 0.000 description 7
- 235000014113 dietary fatty acids Nutrition 0.000 description 7
- 239000000194 fatty acid Substances 0.000 description 7
- 229930195729 fatty acid Natural products 0.000 description 7
- 229910001425 magnesium ion Inorganic materials 0.000 description 7
- WQEPLUUGTLDZJY-UHFFFAOYSA-N n-Pentadecanoic acid Natural products CCCCCCCCCCCCCCC(O)=O WQEPLUUGTLDZJY-UHFFFAOYSA-N 0.000 description 7
- WWZKQHOCKIZLMA-UHFFFAOYSA-N octanoic acid Chemical compound CCCCCCCC(O)=O WWZKQHOCKIZLMA-UHFFFAOYSA-N 0.000 description 7
- YXIWHUQXZSMYRE-UHFFFAOYSA-N 1,3-benzothiazole-2-thiol Chemical compound C1=CC=C2SC(S)=NC2=C1 YXIWHUQXZSMYRE-UHFFFAOYSA-N 0.000 description 6
- LQJBNNIYVWPHFW-UHFFFAOYSA-N 20:1omega9c fatty acid Natural products CCCCCCCCCCC=CCCCCCCCC(O)=O LQJBNNIYVWPHFW-UHFFFAOYSA-N 0.000 description 6
- VTYYLEPIZMXCLO-UHFFFAOYSA-L Calcium carbonate Chemical compound [Ca+2].[O-]C([O-])=O VTYYLEPIZMXCLO-UHFFFAOYSA-L 0.000 description 6
- HBBGRARXTFLTSG-UHFFFAOYSA-N Lithium ion Chemical compound [Li+] HBBGRARXTFLTSG-UHFFFAOYSA-N 0.000 description 6
- 229920003298 Nucrel® Polymers 0.000 description 6
- FKNQFGJONOIPTF-UHFFFAOYSA-N Sodium cation Chemical compound [Na+] FKNQFGJONOIPTF-UHFFFAOYSA-N 0.000 description 6
- 206010000496 acne Diseases 0.000 description 6
- 125000003545 alkoxy group Chemical group 0.000 description 6
- 239000003086 colorant Substances 0.000 description 6
- 230000000052 comparative effect Effects 0.000 description 6
- 125000004093 cyano group Chemical group *C#N 0.000 description 6
- 239000002270 dispersing agent Substances 0.000 description 6
- 150000004665 fatty acids Chemical class 0.000 description 6
- 238000010438 heat treatment Methods 0.000 description 6
- 229910001416 lithium ion Inorganic materials 0.000 description 6
- 239000011572 manganese Substances 0.000 description 6
- JRZJOMJEPLMPRA-UHFFFAOYSA-N olefin Natural products CCCCCCCC=C JRZJOMJEPLMPRA-UHFFFAOYSA-N 0.000 description 6
- 229920001021 polysulfide Polymers 0.000 description 6
- 239000005077 polysulfide Substances 0.000 description 6
- 150000008117 polysulfides Polymers 0.000 description 6
- 239000000843 powder Substances 0.000 description 6
- 229910001415 sodium ion Inorganic materials 0.000 description 6
- 229920000468 styrene butadiene styrene block copolymer Polymers 0.000 description 6
- 125000000020 sulfo group Chemical group O=S(=O)([*])O[H] 0.000 description 6
- 239000006097 ultraviolet radiation absorber Substances 0.000 description 6
- 229920002554 vinyl polymer Polymers 0.000 description 6
- 229940098697 zinc laurate Drugs 0.000 description 6
- 229940105125 zinc myristate Drugs 0.000 description 6
- CHJMFFKHPHCQIJ-UHFFFAOYSA-L zinc;octanoate Chemical compound [Zn+2].CCCCCCCC([O-])=O.CCCCCCCC([O-])=O CHJMFFKHPHCQIJ-UHFFFAOYSA-L 0.000 description 6
- SMZOUWXMTYCWNB-UHFFFAOYSA-N 2-(2-methoxy-5-methylphenyl)ethanamine Chemical compound COC1=CC=C(C)C=C1CCN SMZOUWXMTYCWNB-UHFFFAOYSA-N 0.000 description 5
- NIXOWILDQLNWCW-UHFFFAOYSA-N 2-Propenoic acid Natural products OC(=O)C=C NIXOWILDQLNWCW-UHFFFAOYSA-N 0.000 description 5
- BHPQYMZQTOCNFJ-UHFFFAOYSA-N Calcium cation Chemical compound [Ca+2] BHPQYMZQTOCNFJ-UHFFFAOYSA-N 0.000 description 5
- ZQPPMHVWECSIRJ-UHFFFAOYSA-N Oleic acid Natural products CCCCCCCCC=CCCCCCCCC(O)=O ZQPPMHVWECSIRJ-UHFFFAOYSA-N 0.000 description 5
- 235000021314 Palmitic acid Nutrition 0.000 description 5
- 125000002252 acyl group Chemical group 0.000 description 5
- 125000000217 alkyl group Chemical group 0.000 description 5
- 125000002521 alkyl halide group Chemical group 0.000 description 5
- 125000004644 alkyl sulfinyl group Chemical group 0.000 description 5
- 229910001424 calcium ion Inorganic materials 0.000 description 5
- 125000003917 carbamoyl group Chemical group [H]N([H])C(*)=O 0.000 description 5
- 125000002915 carbonyl group Chemical group [*:2]C([*:1])=O 0.000 description 5
- 150000001768 cations Chemical class 0.000 description 5
- 238000006243 chemical reaction Methods 0.000 description 5
- 150000001993 dienes Chemical class 0.000 description 5
- ZQPPMHVWECSIRJ-MDZDMXLPSA-N elaidic acid Chemical compound CCCCCCCC\C=C\CCCCCCCC(O)=O ZQPPMHVWECSIRJ-MDZDMXLPSA-N 0.000 description 5
- 125000002485 formyl group Chemical group [H]C(*)=O 0.000 description 5
- 150000004820 halides Chemical group 0.000 description 5
- 125000005843 halogen group Chemical group 0.000 description 5
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 5
- QXJSBBXBKPUZAA-UHFFFAOYSA-N isooleic acid Natural products CCCCCCCC=CCCCCCCCCC(O)=O QXJSBBXBKPUZAA-UHFFFAOYSA-N 0.000 description 5
- 125000000213 sulfino group Chemical group [H]OS(*)=O 0.000 description 5
- 125000002128 sulfonyl halide group Chemical group 0.000 description 5
- 230000001629 suppression Effects 0.000 description 5
- WRIDQFICGBMAFQ-UHFFFAOYSA-N (E)-8-Octadecenoic acid Natural products CCCCCCCCCC=CCCCCCCC(O)=O WRIDQFICGBMAFQ-UHFFFAOYSA-N 0.000 description 4
- SDJHPPZKZZWAKF-UHFFFAOYSA-N 2,3-dimethylbuta-1,3-diene Chemical compound CC(=C)C(C)=C SDJHPPZKZZWAKF-UHFFFAOYSA-N 0.000 description 4
- KVZUCOGWKYOPID-UHFFFAOYSA-N 2,4,5-Trimethoxybenzoic acid Chemical compound COC1=CC(OC)=C(C(O)=O)C=C1OC KVZUCOGWKYOPID-UHFFFAOYSA-N 0.000 description 4
- CJBDUOMQLFKVQC-UHFFFAOYSA-N 3-(2-hydroxyphenyl)propanoic acid Chemical compound OC(=O)CCC1=CC=CC=C1O CJBDUOMQLFKVQC-UHFFFAOYSA-N 0.000 description 4
- LBKFGYZQBSGRHY-UHFFFAOYSA-N 3-hydroxy-4-methoxybenzoic acid Chemical compound COC1=CC=C(C(O)=O)C=C1O LBKFGYZQBSGRHY-UHFFFAOYSA-N 0.000 description 4
- CNGYZEMWVAWWOB-VAWYXSNFSA-N 5-[[4-anilino-6-[bis(2-hydroxyethyl)amino]-1,3,5-triazin-2-yl]amino]-2-[(e)-2-[4-[[4-anilino-6-[bis(2-hydroxyethyl)amino]-1,3,5-triazin-2-yl]amino]-2-sulfophenyl]ethenyl]benzenesulfonic acid Chemical compound N=1C(NC=2C=C(C(\C=C\C=3C(=CC(NC=4N=C(N=C(NC=5C=CC=CC=5)N=4)N(CCO)CCO)=CC=3)S(O)(=O)=O)=CC=2)S(O)(=O)=O)=NC(N(CCO)CCO)=NC=1NC1=CC=CC=C1 CNGYZEMWVAWWOB-VAWYXSNFSA-N 0.000 description 4
- QSBYPNXLFMSGKH-UHFFFAOYSA-N 9-Heptadecensaeure Natural products CCCCCCCC=CCCCCCCCC(O)=O QSBYPNXLFMSGKH-UHFFFAOYSA-N 0.000 description 4
- 235000021357 Behenic acid Nutrition 0.000 description 4
- 239000005063 High cis polybutadiene Substances 0.000 description 4
- 239000005639 Lauric acid Substances 0.000 description 4
- 239000005642 Oleic acid Substances 0.000 description 4
- NPYPAHLBTDXSSS-UHFFFAOYSA-N Potassium ion Chemical compound [K+] NPYPAHLBTDXSSS-UHFFFAOYSA-N 0.000 description 4
- 239000012190 activator Substances 0.000 description 4
- 125000004453 alkoxycarbonyl group Chemical group 0.000 description 4
- 125000005138 alkoxysulfonyl group Chemical group 0.000 description 4
- 229910052782 aluminium Inorganic materials 0.000 description 4
- 229940063655 aluminum stearate Drugs 0.000 description 4
- 125000003277 amino group Chemical group 0.000 description 4
- 239000003963 antioxidant agent Substances 0.000 description 4
- 230000003078 antioxidant effect Effects 0.000 description 4
- 229940116226 behenic acid Drugs 0.000 description 4
- UJMDYLWCYJJYMO-UHFFFAOYSA-N benzene-1,2,3-tricarboxylic acid Chemical compound OC(=O)C1=CC=CC(C(O)=O)=C1C(O)=O UJMDYLWCYJJYMO-UHFFFAOYSA-N 0.000 description 4
- XITRBUPOXXBIJN-UHFFFAOYSA-N bis(2,2,6,6-tetramethylpiperidin-4-yl) decanedioate Chemical compound C1C(C)(C)NC(C)(C)CC1OC(=O)CCCCCCCCC(=O)OC1CC(C)(C)NC(C)(C)C1 XITRBUPOXXBIJN-UHFFFAOYSA-N 0.000 description 4
- 238000000748 compression moulding Methods 0.000 description 4
- 150000001879 copper Chemical class 0.000 description 4
- LNTHITQWFMADLM-UHFFFAOYSA-N gallic acid Chemical compound OC(=O)C1=CC(O)=C(O)C(O)=C1 LNTHITQWFMADLM-UHFFFAOYSA-N 0.000 description 4
- 238000007542 hardness measurement Methods 0.000 description 4
- 239000004611 light stabiliser Substances 0.000 description 4
- YDSWCNNOKPMOTP-UHFFFAOYSA-N mellitic acid Chemical compound OC(=O)C1=C(C(O)=O)C(C(O)=O)=C(C(O)=O)C(C(O)=O)=C1C(O)=O YDSWCNNOKPMOTP-UHFFFAOYSA-N 0.000 description 4
- 238000006386 neutralization reaction Methods 0.000 description 4
- 150000002892 organic cations Chemical class 0.000 description 4
- 150000002894 organic compounds Chemical class 0.000 description 4
- 150000001451 organic peroxides Chemical class 0.000 description 4
- 239000003973 paint Substances 0.000 description 4
- 125000003356 phenylsulfanyl group Chemical group [*]SC1=C([H])C([H])=C([H])C([H])=C1[H] 0.000 description 4
- NMHMNPHRMNGLLB-UHFFFAOYSA-N phloretic acid Chemical compound OC(=O)CCC1=CC=C(O)C=C1 NMHMNPHRMNGLLB-UHFFFAOYSA-N 0.000 description 4
- 239000000049 pigment Substances 0.000 description 4
- 229920002742 polystyrene-block-poly(ethylene/propylene) -block-polystyrene Polymers 0.000 description 4
- 229920002743 polystyrene-poly(ethylene-ethylene/propylene) block-polystyrene Polymers 0.000 description 4
- 229910001414 potassium ion Inorganic materials 0.000 description 4
- 238000003825 pressing Methods 0.000 description 4
- QQONPFPTGQHPMA-UHFFFAOYSA-N propylene Natural products CC=C QQONPFPTGQHPMA-UHFFFAOYSA-N 0.000 description 4
- 125000004805 propylene group Chemical group [H]C([H])([H])C([H])([*:1])C([H])([H])[*:2] 0.000 description 4
- CYIDZMCFTVVTJO-UHFFFAOYSA-N pyromellitic acid Chemical compound OC(=O)C1=CC(C(O)=O)=C(C(O)=O)C=C1C(O)=O CYIDZMCFTVVTJO-UHFFFAOYSA-N 0.000 description 4
- 150000004671 saturated fatty acids Chemical class 0.000 description 4
- 229920001935 styrene-ethylene-butadiene-styrene Polymers 0.000 description 4
- JMSVCTWVEWCHDZ-UHFFFAOYSA-N syringic acid Chemical compound COC1=CC(C(O)=O)=CC(OC)=C1O JMSVCTWVEWCHDZ-UHFFFAOYSA-N 0.000 description 4
- QENJZWZWAWWESF-UHFFFAOYSA-N tri-methylbenzoic acid Natural products CC1=CC(C)=C(C(O)=O)C=C1C QENJZWZWAWWESF-UHFFFAOYSA-N 0.000 description 4
- ARCGXLSVLAOJQL-UHFFFAOYSA-N trimellitic acid Chemical compound OC(=O)C1=CC=C(C(O)=O)C(C(O)=O)=C1 ARCGXLSVLAOJQL-UHFFFAOYSA-N 0.000 description 4
- GPYYEEJOMCKTPR-UHFFFAOYSA-L zinc;dodecanoate Chemical compound [Zn+2].CCCCCCCCCCCC([O-])=O.CCCCCCCCCCCC([O-])=O GPYYEEJOMCKTPR-UHFFFAOYSA-L 0.000 description 4
- GBFLQPIIIRJQLU-UHFFFAOYSA-L zinc;tetradecanoate Chemical compound [Zn+2].CCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCC([O-])=O GBFLQPIIIRJQLU-UHFFFAOYSA-L 0.000 description 4
- ZEYHEAKUIGZSGI-UHFFFAOYSA-N 4-methoxybenzoic acid Chemical compound COC1=CC=C(C(O)=O)C=C1 ZEYHEAKUIGZSGI-UHFFFAOYSA-N 0.000 description 3
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 3
- WMFOQBRAJBCJND-UHFFFAOYSA-M Lithium hydroxide Chemical compound [Li+].[OH-] WMFOQBRAJBCJND-UHFFFAOYSA-M 0.000 description 3
- 239000004793 Polystyrene Substances 0.000 description 3
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 description 3
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 3
- OBETXYAYXDNJHR-UHFFFAOYSA-N alpha-ethylcaproic acid Natural products CCCCC(CC)C(O)=O OBETXYAYXDNJHR-UHFFFAOYSA-N 0.000 description 3
- 229910001422 barium ion Inorganic materials 0.000 description 3
- GCAIEATUVJFSMC-UHFFFAOYSA-N benzene-1,2,3,4-tetracarboxylic acid Chemical compound OC(=O)C1=CC=C(C(O)=O)C(C(O)=O)=C1C(O)=O GCAIEATUVJFSMC-UHFFFAOYSA-N 0.000 description 3
- WLZRMCYVCSSEQC-UHFFFAOYSA-N cadmium(2+) Chemical compound [Cd+2] WLZRMCYVCSSEQC-UHFFFAOYSA-N 0.000 description 3
- 229910000019 calcium carbonate Inorganic materials 0.000 description 3
- QRMZSPFSDQBLIX-UHFFFAOYSA-N homovanillic acid Chemical compound COC1=CC(CC(O)=O)=CC=C1O QRMZSPFSDQBLIX-UHFFFAOYSA-N 0.000 description 3
- ZLNQQNXFFQJAID-UHFFFAOYSA-L magnesium carbonate Chemical compound [Mg+2].[O-]C([O-])=O ZLNQQNXFFQJAID-UHFFFAOYSA-L 0.000 description 3
- 239000001095 magnesium carbonate Substances 0.000 description 3
- 229910000021 magnesium carbonate Inorganic materials 0.000 description 3
- 239000000178 monomer Substances 0.000 description 3
- QEFYFXOXNSNQGX-UHFFFAOYSA-N neodymium atom Chemical compound [Nd] QEFYFXOXNSNQGX-UHFFFAOYSA-N 0.000 description 3
- AMKYESDOVDKZKV-UHFFFAOYSA-N o-orsellinic acid Chemical compound CC1=CC(O)=CC(O)=C1C(O)=O AMKYESDOVDKZKV-UHFFFAOYSA-N 0.000 description 3
- ZWLPBLYKEWSWPD-UHFFFAOYSA-N o-toluic acid Chemical compound CC1=CC=CC=C1C(O)=O ZWLPBLYKEWSWPD-UHFFFAOYSA-N 0.000 description 3
- 229960002446 octanoic acid Drugs 0.000 description 3
- 235000021313 oleic acid Nutrition 0.000 description 3
- CKMXAIVXVKGGFM-UHFFFAOYSA-N p-cumic acid Chemical compound CC(C)C1=CC=C(C(O)=O)C=C1 CKMXAIVXVKGGFM-UHFFFAOYSA-N 0.000 description 3
- 229920002223 polystyrene Polymers 0.000 description 3
- PCMORTLOPMLEFB-ONEGZZNKSA-N sinapic acid Chemical compound COC1=CC(\C=C\C(O)=O)=CC(OC)=C1O PCMORTLOPMLEFB-ONEGZZNKSA-N 0.000 description 3
- 125000004434 sulfur atom Chemical group 0.000 description 3
- TUNFSRHWOTWDNC-HKGQFRNVSA-N tetradecanoic acid Chemical compound CCCCCCCCCCCCC[14C](O)=O TUNFSRHWOTWDNC-HKGQFRNVSA-N 0.000 description 3
- QAIPRVGONGVQAS-DUXPYHPUSA-N trans-caffeic acid Chemical compound OC(=O)\C=C\C1=CC=C(O)C(O)=C1 QAIPRVGONGVQAS-DUXPYHPUSA-N 0.000 description 3
- 229910052725 zinc Inorganic materials 0.000 description 3
- YWWVWXASSLXJHU-AATRIKPKSA-N (9E)-tetradecenoic acid Chemical compound CCCC\C=C\CCCCCCCC(O)=O YWWVWXASSLXJHU-AATRIKPKSA-N 0.000 description 2
- PMJHHCWVYXUKFD-SNAWJCMRSA-N (E)-1,3-pentadiene Chemical compound C\C=C\C=C PMJHHCWVYXUKFD-SNAWJCMRSA-N 0.000 description 2
- ULQISTXYYBZJSJ-UHFFFAOYSA-N 12-hydroxyoctadecanoic acid Chemical compound CCCCCCC(O)CCCCCCCCCCC(O)=O ULQISTXYYBZJSJ-UHFFFAOYSA-N 0.000 description 2
- HGEFWFBFQKWVMY-DUXPYHPUSA-N 2,4-dihydroxy-trans cinnamic acid Chemical compound OC(=O)\C=C\C1=CC=C(O)C=C1O HGEFWFBFQKWVMY-DUXPYHPUSA-N 0.000 description 2
- TWJNQYPJQDRXPH-UHFFFAOYSA-N 2-cyanobenzohydrazide Chemical compound NNC(=O)C1=CC=CC=C1C#N TWJNQYPJQDRXPH-UHFFFAOYSA-N 0.000 description 2
- LFLOMAIEONDOLV-UHFFFAOYSA-N 2-oxalobenzoic acid Chemical compound OC(=O)C(=O)C1=CC=CC=C1C(O)=O LFLOMAIEONDOLV-UHFFFAOYSA-N 0.000 description 2
- DAUAQNGYDSHRET-UHFFFAOYSA-N 3,4-dimethoxybenzoic acid Chemical compound COC1=CC=C(C(O)=O)C=C1OC DAUAQNGYDSHRET-UHFFFAOYSA-N 0.000 description 2
- DOBIZWYVJFIYOV-UHFFFAOYSA-N 7-hydroxynaphthalene-1,3-disulfonic acid Chemical compound C1=C(S(O)(=O)=O)C=C(S(O)(=O)=O)C2=CC(O)=CC=C21 DOBIZWYVJFIYOV-UHFFFAOYSA-N 0.000 description 2
- GGZZISOUXJHYOY-UHFFFAOYSA-N 8-amino-4-hydroxynaphthalene-2-sulfonic acid Chemical compound C1=C(S(O)(=O)=O)C=C2C(N)=CC=CC2=C1O GGZZISOUXJHYOY-UHFFFAOYSA-N 0.000 description 2
- FERIUCNNQQJTOY-UHFFFAOYSA-N Butyric acid Chemical compound CCCC(O)=O FERIUCNNQQJTOY-UHFFFAOYSA-N 0.000 description 2
- 229920003307 DuPont™ Surlyn® 8150 Polymers 0.000 description 2
- VZCYOOQTPOCHFL-OWOJBTEDSA-N Fumaric acid Chemical compound OC(=O)\C=C\C(O)=O VZCYOOQTPOCHFL-OWOJBTEDSA-N 0.000 description 2
- WAEMQWOKJMHJLA-UHFFFAOYSA-N Manganese(2+) Chemical compound [Mn+2] WAEMQWOKJMHJLA-UHFFFAOYSA-N 0.000 description 2
- 235000021360 Myristic acid Nutrition 0.000 description 2
- TUNFSRHWOTWDNC-UHFFFAOYSA-N Myristic acid Natural products CCCCCCCCCCCCCC(O)=O TUNFSRHWOTWDNC-UHFFFAOYSA-N 0.000 description 2
- QGMRQYFBGABWDR-UHFFFAOYSA-M Pentobarbital sodium Chemical compound [Na+].CCCC(C)C1(CC)C(=O)NC(=O)[N-]C1=O QGMRQYFBGABWDR-UHFFFAOYSA-M 0.000 description 2
- IWYDHOAUDWTVEP-UHFFFAOYSA-N R-2-phenyl-2-hydroxyacetic acid Natural products OC(=O)C(O)C1=CC=CC=C1 IWYDHOAUDWTVEP-UHFFFAOYSA-N 0.000 description 2
- FOIXSVOLVBLSDH-UHFFFAOYSA-N Silver ion Chemical compound [Ag+] FOIXSVOLVBLSDH-UHFFFAOYSA-N 0.000 description 2
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical compound [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 description 2
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 2
- KKEYFWRCBNTPAC-UHFFFAOYSA-N Terephthalic acid Chemical compound OC(=O)C1=CC=C(C(O)=O)C=C1 KKEYFWRCBNTPAC-UHFFFAOYSA-N 0.000 description 2
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 2
- CGQCWMIAEPEHNQ-UHFFFAOYSA-N Vanillylmandelic acid Chemical compound COC1=CC(C(O)C(O)=O)=CC=C1O CGQCWMIAEPEHNQ-UHFFFAOYSA-N 0.000 description 2
- GPVDHNVGGIAOQT-UHFFFAOYSA-N Veratric acid Natural products COC1=CC=C(C(O)=O)C(OC)=C1 GPVDHNVGGIAOQT-UHFFFAOYSA-N 0.000 description 2
- 150000007933 aliphatic carboxylic acids Chemical class 0.000 description 2
- AZDRQVAHHNSJOQ-UHFFFAOYSA-N alumane Chemical class [AlH3] AZDRQVAHHNSJOQ-UHFFFAOYSA-N 0.000 description 2
- CEGOLXSVJUTHNZ-UHFFFAOYSA-K aluminium tristearate Chemical compound [Al+3].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O CEGOLXSVJUTHNZ-UHFFFAOYSA-K 0.000 description 2
- 230000003712 anti-aging effect Effects 0.000 description 2
- YZXBAPSDXZZRGB-DOFZRALJSA-N arachidonic acid Chemical compound CCCCC\C=C/C\C=C/C\C=C/C\C=C/CCCC(O)=O YZXBAPSDXZZRGB-DOFZRALJSA-N 0.000 description 2
- 125000004429 atom Chemical group 0.000 description 2
- NWCHELUCVWSRRS-UHFFFAOYSA-N atrolactic acid Chemical compound OC(=O)C(O)(C)C1=CC=CC=C1 NWCHELUCVWSRRS-UHFFFAOYSA-N 0.000 description 2
- IOJUPLGTWVMSFF-UHFFFAOYSA-N benzothiazole Chemical compound C1=CC=C2SC=NC2=C1 IOJUPLGTWVMSFF-UHFFFAOYSA-N 0.000 description 2
- GJKZSOHUVOQISW-UHFFFAOYSA-N buta-1,3-diene;2-methylbuta-1,3-diene;styrene Chemical compound C=CC=C.CC(=C)C=C.C=CC1=CC=CC=C1.C=CC1=CC=CC=C1 GJKZSOHUVOQISW-UHFFFAOYSA-N 0.000 description 2
- 229910052799 carbon Inorganic materials 0.000 description 2
- 239000003054 catalyst Substances 0.000 description 2
- 239000003795 chemical substances by application Substances 0.000 description 2
- 239000011248 coating agent Substances 0.000 description 2
- 238000000576 coating method Methods 0.000 description 2
- 150000001868 cobalt Chemical class 0.000 description 2
- XLJKHNWPARRRJB-UHFFFAOYSA-N cobalt(2+) Chemical compound [Co+2] XLJKHNWPARRRJB-UHFFFAOYSA-N 0.000 description 2
- 238000007334 copolymerization reaction Methods 0.000 description 2
- GHVNFZFCNZKVNT-UHFFFAOYSA-N decanoic acid Chemical compound CCCCCCCCCC(O)=O GHVNFZFCNZKVNT-UHFFFAOYSA-N 0.000 description 2
- 230000007423 decrease Effects 0.000 description 2
- AFZSMODLJJCVPP-UHFFFAOYSA-N dibenzothiazol-2-yl disulfide Chemical compound C1=CC=C2SC(SSC=3SC4=CC=CC=C4N=3)=NC2=C1 AFZSMODLJJCVPP-UHFFFAOYSA-N 0.000 description 2
- LMBWSYZSUOEYSN-UHFFFAOYSA-N diethyldithiocarbamic acid Chemical compound CCN(CC)C(S)=S LMBWSYZSUOEYSN-UHFFFAOYSA-N 0.000 description 2
- GWZCCUDJHOGOSO-UHFFFAOYSA-N diphenic acid Chemical compound OC(=O)C1=CC=CC=C1C1=CC=CC=C1C(O)=O GWZCCUDJHOGOSO-UHFFFAOYSA-N 0.000 description 2
- AUZONCFQVSMFAP-UHFFFAOYSA-N disulfiram Chemical compound CCN(CC)C(=S)SSC(=S)N(CC)CC AUZONCFQVSMFAP-UHFFFAOYSA-N 0.000 description 2
- 239000012990 dithiocarbamate Substances 0.000 description 2
- 150000004659 dithiocarbamates Chemical class 0.000 description 2
- 239000000806 elastomer Substances 0.000 description 2
- 150000002148 esters Chemical class 0.000 description 2
- 238000002474 experimental method Methods 0.000 description 2
- KSEBMYQBYZTDHS-HWKANZROSA-N ferulic acid Chemical compound COC1=CC(\C=C\C(O)=O)=CC=C1O KSEBMYQBYZTDHS-HWKANZROSA-N 0.000 description 2
- KSEBMYQBYZTDHS-UHFFFAOYSA-N ferulic acid Natural products COC1=CC(C=CC(O)=O)=CC=C1O KSEBMYQBYZTDHS-UHFFFAOYSA-N 0.000 description 2
- 125000000524 functional group Chemical group 0.000 description 2
- KEMQGTRYUADPNZ-UHFFFAOYSA-N heptadecanoic acid Chemical compound CCCCCCCCCCCCCCCCC(O)=O KEMQGTRYUADPNZ-UHFFFAOYSA-N 0.000 description 2
- MNWFXJYAOYHMED-UHFFFAOYSA-N heptanoic acid Chemical compound CCCCCCC(O)=O MNWFXJYAOYHMED-UHFFFAOYSA-N 0.000 description 2
- XMHIUKTWLZUKEX-UHFFFAOYSA-N hexacosanoic acid Chemical compound CCCCCCCCCCCCCCCCCCCCCCCCCC(O)=O XMHIUKTWLZUKEX-UHFFFAOYSA-N 0.000 description 2
- FUZZWVXGSFPDMH-UHFFFAOYSA-N hexanoic acid Chemical compound CCCCCC(O)=O FUZZWVXGSFPDMH-UHFFFAOYSA-N 0.000 description 2
- YPGCWEMNNLXISK-UHFFFAOYSA-N hydratropic acid Chemical compound OC(=O)C(C)C1=CC=CC=C1 YPGCWEMNNLXISK-UHFFFAOYSA-N 0.000 description 2
- VKOBVWXKNCXXDE-UHFFFAOYSA-N icosanoic acid Chemical compound CCCCCCCCCCCCCCCCCCCC(O)=O VKOBVWXKNCXXDE-UHFFFAOYSA-N 0.000 description 2
- 150000002500 ions Chemical class 0.000 description 2
- 150000002505 iron Chemical class 0.000 description 2
- BWXLCOBSWMQCGP-UHFFFAOYSA-N isohomovanillic acid Chemical compound COC1=CC=C(CC(O)=O)C=C1O BWXLCOBSWMQCGP-UHFFFAOYSA-N 0.000 description 2
- QQVIHTHCMHWDBS-UHFFFAOYSA-N isophthalic acid Chemical compound OC(=O)C1=CC=CC(C(O)=O)=C1 QQVIHTHCMHWDBS-UHFFFAOYSA-N 0.000 description 2
- 159000000003 magnesium salts Chemical class 0.000 description 2
- 238000000691 measurement method Methods 0.000 description 2
- 238000013508 migration Methods 0.000 description 2
- 230000005012 migration Effects 0.000 description 2
- 238000012986 modification Methods 0.000 description 2
- 230000004048 modification Effects 0.000 description 2
- SEXOVMIIVBKGGM-UHFFFAOYSA-N naphthalene-1-thiol Chemical compound C1=CC=C2C(S)=CC=CC2=C1 SEXOVMIIVBKGGM-UHFFFAOYSA-N 0.000 description 2
- 150000002815 nickel Chemical class 0.000 description 2
- FBUKVWPVBMHYJY-UHFFFAOYSA-N nonanoic acid Chemical compound CCCCCCCCC(O)=O FBUKVWPVBMHYJY-UHFFFAOYSA-N 0.000 description 2
- UTOPWMOLSKOLTQ-UHFFFAOYSA-N octacosanoic acid Chemical compound CCCCCCCCCCCCCCCCCCCCCCCCCCCC(O)=O UTOPWMOLSKOLTQ-UHFFFAOYSA-N 0.000 description 2
- SECPZKHBENQXJG-FPLPWBNLSA-N palmitoleic acid Chemical compound CCCCCC\C=C/CCCCCCCC(O)=O SECPZKHBENQXJG-FPLPWBNLSA-N 0.000 description 2
- YWAKXRMUMFPDSH-UHFFFAOYSA-N pentene Chemical compound CCCC=C YWAKXRMUMFPDSH-UHFFFAOYSA-N 0.000 description 2
- XNGIFLGASWRNHJ-UHFFFAOYSA-N phthalic acid Chemical compound OC(=O)C1=CC=CC=C1C(O)=O XNGIFLGASWRNHJ-UHFFFAOYSA-N 0.000 description 2
- PMJHHCWVYXUKFD-UHFFFAOYSA-N piperylene Natural products CC=CC=C PMJHHCWVYXUKFD-UHFFFAOYSA-N 0.000 description 2
- 239000004014 plasticizer Substances 0.000 description 2
- 229920000642 polymer Polymers 0.000 description 2
- 229920002635 polyurethane Polymers 0.000 description 2
- 239000004814 polyurethane Substances 0.000 description 2
- XAEFZNCEHLXOMS-UHFFFAOYSA-M potassium benzoate Chemical compound [K+].[O-]C(=O)C1=CC=CC=C1 XAEFZNCEHLXOMS-UHFFFAOYSA-M 0.000 description 2
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 2
- 229910052761 rare earth metal Inorganic materials 0.000 description 2
- RZWQDAUIUBVCDD-UHFFFAOYSA-M sodium;benzenethiolate Chemical compound [Na+].[S-]C1=CC=CC=C1 RZWQDAUIUBVCDD-UHFFFAOYSA-M 0.000 description 2
- LMCILKQUBHEOPB-UHFFFAOYSA-M sodium;ethene;prop-2-enoate Chemical compound [Na+].C=C.[O-]C(=O)C=C LMCILKQUBHEOPB-UHFFFAOYSA-M 0.000 description 2
- 229910052717 sulfur Inorganic materials 0.000 description 2
- 239000011593 sulfur Substances 0.000 description 2
- 229920003002 synthetic resin Polymers 0.000 description 2
- 239000000057 synthetic resin Substances 0.000 description 2
- 229920002397 thermoplastic olefin Polymers 0.000 description 2
- 229920006345 thermoplastic polyamide Polymers 0.000 description 2
- 229920006346 thermoplastic polyester elastomer Polymers 0.000 description 2
- 125000003396 thiol group Chemical group [H]S* 0.000 description 2
- KUAZQDVKQLNFPE-UHFFFAOYSA-N thiram Chemical compound CN(C)C(=S)SSC(=S)N(C)C KUAZQDVKQLNFPE-UHFFFAOYSA-N 0.000 description 2
- 229960002447 thiram Drugs 0.000 description 2
- 229910001432 tin ion Inorganic materials 0.000 description 2
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 2
- VHOCUJPBKOZGJD-UHFFFAOYSA-N triacontanoic acid Chemical compound CCCCCCCCCCCCCCCCCCCCCCCCCCCCCC(O)=O VHOCUJPBKOZGJD-UHFFFAOYSA-N 0.000 description 2
- JACRWUWPXAESPB-UHFFFAOYSA-N tropic acid Chemical compound OCC(C(O)=O)C1=CC=CC=C1 JACRWUWPXAESPB-UHFFFAOYSA-N 0.000 description 2
- 235000021122 unsaturated fatty acids Nutrition 0.000 description 2
- 150000004670 unsaturated fatty acids Chemical class 0.000 description 2
- NQPDZGIKBAWPEJ-UHFFFAOYSA-N valeric acid Chemical compound CCCCC(O)=O NQPDZGIKBAWPEJ-UHFFFAOYSA-N 0.000 description 2
- WKOLLVMJNQIZCI-UHFFFAOYSA-N vanillic acid Chemical compound COC1=CC(C(O)=O)=CC=C1O WKOLLVMJNQIZCI-UHFFFAOYSA-N 0.000 description 2
- TUUBOHWZSQXCSW-UHFFFAOYSA-N vanillic acid Natural products COC1=CC(O)=CC(C(O)=O)=C1 TUUBOHWZSQXCSW-UHFFFAOYSA-N 0.000 description 2
- 238000004073 vulcanization Methods 0.000 description 2
- 229940057977 zinc stearate Drugs 0.000 description 2
- GBNDTYKAOXLLID-UHFFFAOYSA-N zirconium(4+) ion Chemical compound [Zr+4] GBNDTYKAOXLLID-UHFFFAOYSA-N 0.000 description 2
- BITHHVVYSMSWAG-KTKRTIGZSA-N (11Z)-icos-11-enoic acid Chemical compound CCCCCCCC\C=C/CCCCCCCCCC(O)=O BITHHVVYSMSWAG-KTKRTIGZSA-N 0.000 description 1
- GWHCXVQVJPWHRF-KTKRTIGZSA-N (15Z)-tetracosenoic acid Chemical compound CCCCCCCC\C=C/CCCCCCCCCCCCCC(O)=O GWHCXVQVJPWHRF-KTKRTIGZSA-N 0.000 description 1
- QBYIENPQHBMVBV-HFEGYEGKSA-N (2R)-2-hydroxy-2-phenylacetic acid Chemical compound O[C@@H](C(O)=O)c1ccccc1.O[C@@H](C(O)=O)c1ccccc1 QBYIENPQHBMVBV-HFEGYEGKSA-N 0.000 description 1
- ACEAELOMUCBPJP-UHFFFAOYSA-N (E)-3,4,5-trihydroxycinnamic acid Natural products OC(=O)C=CC1=CC(O)=C(O)C(O)=C1 ACEAELOMUCBPJP-UHFFFAOYSA-N 0.000 description 1
- KSEBMYQBYZTDHS-HWKANZROSA-M (E)-Ferulic acid Natural products COC1=CC(\C=C\C([O-])=O)=CC=C1O KSEBMYQBYZTDHS-HWKANZROSA-M 0.000 description 1
- NALFRYPTRXKZPN-UHFFFAOYSA-N 1,1-bis(tert-butylperoxy)-3,3,5-trimethylcyclohexane Chemical compound CC1CC(C)(C)CC(OOC(C)(C)C)(OOC(C)(C)C)C1 NALFRYPTRXKZPN-UHFFFAOYSA-N 0.000 description 1
- LGXISKQYIKXYTC-UHFFFAOYSA-N 1,2,3,4,5-pentabromo-6-[(2,3,4,5,6-pentabromophenyl)disulfanyl]benzene Chemical group BrC1=C(Br)C(Br)=C(Br)C(Br)=C1SSC1=C(Br)C(Br)=C(Br)C(Br)=C1Br LGXISKQYIKXYTC-UHFFFAOYSA-N 0.000 description 1
- LSVXAQMPXJUTBV-UHFFFAOYSA-N 1,2,3,4,5-pentachloro-6-[(2,3,4,5,6-pentachlorophenyl)disulfanyl]benzene Chemical group ClC1=C(Cl)C(Cl)=C(Cl)C(Cl)=C1SSC1=C(Cl)C(Cl)=C(Cl)C(Cl)=C1Cl LSVXAQMPXJUTBV-UHFFFAOYSA-N 0.000 description 1
- DVDJHJDHPBSYTN-UHFFFAOYSA-N 1,2,3,4,5-pentafluoro-6-[(2,3,4,5,6-pentafluorophenyl)disulfanyl]benzene Chemical group FC1=C(F)C(F)=C(F)C(F)=C1SSC1=C(F)C(F)=C(F)C(F)=C1F DVDJHJDHPBSYTN-UHFFFAOYSA-N 0.000 description 1
- DALSLJFWXBCJLI-UHFFFAOYSA-N 1,2,3,4,5-pentaiodo-6-[(2,3,4,5,6-pentaiodophenyl)disulfanyl]benzene Chemical group IC1=C(I)C(I)=C(I)C(I)=C1SSC1=C(I)C(I)=C(I)C(I)=C1I DALSLJFWXBCJLI-UHFFFAOYSA-N 0.000 description 1
- LUKRQVSOYHSWQV-UHFFFAOYSA-N 1,2,3,4,5-pentakis(methylsulfinyl)-6-[[2,3,4,5,6-pentakis(methylsulfinyl)phenyl]disulfanyl]benzene Chemical group CS(=O)C1=C(S(C)=O)C(S(C)=O)=C(S(C)=O)C(S(=O)C)=C1SSC1=C(S(C)=O)C(S(C)=O)=C(S(C)=O)C(S(C)=O)=C1S(C)=O LUKRQVSOYHSWQV-UHFFFAOYSA-N 0.000 description 1
- NGVNZFSOWOVOKE-UHFFFAOYSA-N 1,2,3,4,5-pentamethoxy-6-[(2,3,4,5,6-pentamethoxyphenyl)disulfanyl]benzene Chemical compound COC1=C(OC)C(OC)=C(OC)C(OC)=C1SSC1=C(OC)C(OC)=C(OC)C(OC)=C1OC NGVNZFSOWOVOKE-UHFFFAOYSA-N 0.000 description 1
- MFFNBOPWXQHZSB-UHFFFAOYSA-N 1,2,3,4,5-pentamethyl-6-[(2,3,4,5,6-pentamethylphenyl)disulfanyl]benzene Chemical group CC1=C(C)C(C)=C(C)C(C)=C1SSC1=C(C)C(C)=C(C)C(C)=C1C MFFNBOPWXQHZSB-UHFFFAOYSA-N 0.000 description 1
- QLMQQROHXJNREC-UHFFFAOYSA-N 1,2,3,4,5-pentatert-butyl-6-[(2,3,4,5,6-pentatert-butylphenyl)disulfanyl]benzene Chemical group CC(C)(C)C1=C(C(C)(C)C)C(C(C)(C)C)=C(C(C)(C)C)C(C(C)(C)C)=C1SSC1=C(C(C)(C)C)C(C(C)(C)C)=C(C(C)(C)C)C(C(C)(C)C)=C1C(C)(C)C QLMQQROHXJNREC-UHFFFAOYSA-N 0.000 description 1
- IJFNNTSLKFFMPS-UHFFFAOYSA-N 1,2,3,5-tetrabromo-4-[(2,3,4,6-tetrabromophenyl)disulfanyl]benzene Chemical group BrC1=C(Br)C(Br)=CC(Br)=C1SSC1=C(Br)C=C(Br)C(Br)=C1Br IJFNNTSLKFFMPS-UHFFFAOYSA-N 0.000 description 1
- QHFLLDLTUKTUGF-UHFFFAOYSA-N 1,2,3,5-tetrachloro-4-[(2,3,4,6-tetrachlorophenyl)disulfanyl]benzene Chemical group ClC1=C(Cl)C(Cl)=CC(Cl)=C1SSC1=C(Cl)C=C(Cl)C(Cl)=C1Cl QHFLLDLTUKTUGF-UHFFFAOYSA-N 0.000 description 1
- BPPGGSFJQMKSME-UHFFFAOYSA-N 1,2,3,5-tetrafluoro-4-[(2,3,4,6-tetrafluorophenyl)disulfanyl]benzene Chemical group FC1=C(F)C(F)=CC(F)=C1SSC1=C(F)C=C(F)C(F)=C1F BPPGGSFJQMKSME-UHFFFAOYSA-N 0.000 description 1
- NRZLUSCAQJBOSJ-UHFFFAOYSA-N 1,2,3,5-tetraiodo-4-[(2,3,4,6-tetraiodophenyl)disulfanyl]benzene Chemical group IC1=C(I)C(I)=CC(I)=C1SSC1=C(I)C=C(I)C(I)=C1I NRZLUSCAQJBOSJ-UHFFFAOYSA-N 0.000 description 1
- JJEQUOLCTTUGIA-UHFFFAOYSA-N 1,2,4-tribromo-5-[(2,4,5-tribromophenyl)disulfanyl]benzene Chemical group C1=C(Br)C(Br)=CC(Br)=C1SSC1=CC(Br)=C(Br)C=C1Br JJEQUOLCTTUGIA-UHFFFAOYSA-N 0.000 description 1
- ZUVJVEOHQKNMPN-UHFFFAOYSA-N 1,2,4-trichloro-5-[(2,4,5-trichlorophenyl)disulfanyl]benzene Chemical group C1=C(Cl)C(Cl)=CC(Cl)=C1SSC1=CC(Cl)=C(Cl)C=C1Cl ZUVJVEOHQKNMPN-UHFFFAOYSA-N 0.000 description 1
- JCBJXOVCIBDTRN-UHFFFAOYSA-N 1,2,4-trifluoro-5-[(2,4,5-trifluorophenyl)disulfanyl]benzene Chemical group C1=C(F)C(F)=CC(F)=C1SSC1=CC(F)=C(F)C=C1F JCBJXOVCIBDTRN-UHFFFAOYSA-N 0.000 description 1
- RVGZKFOSOBGOPS-UHFFFAOYSA-N 1,2,4-triiodo-5-[(2,4,5-triiodophenyl)disulfanyl]benzene Chemical group C1=C(I)C(I)=CC(I)=C1SSC1=CC(I)=C(I)C=C1I RVGZKFOSOBGOPS-UHFFFAOYSA-N 0.000 description 1
- PJVOHXVORUADMA-UHFFFAOYSA-N 1,2,4-trimethyl-5-[(2,4,5-trimethylphenyl)disulfanyl]benzene Chemical group C1=C(C)C(C)=CC(C)=C1SSC1=CC(C)=C(C)C=C1C PJVOHXVORUADMA-UHFFFAOYSA-N 0.000 description 1
- KWLRADHHMBFZDQ-UHFFFAOYSA-N 1,2,4-tritert-butyl-5-[(2,4,5-tritert-butylphenyl)disulfanyl]benzene Chemical group CC(C)(C)C1=CC(C(C)(C)C)=C(C(C)(C)C)C=C1SSC1=CC(C(C)(C)C)=C(C(C)(C)C)C=C1C(C)(C)C KWLRADHHMBFZDQ-UHFFFAOYSA-N 0.000 description 1
- ISMODUSOBFKDMJ-UHFFFAOYSA-N 1,3,5-trimethoxy-2-[(2,4,6-trimethoxyphenyl)disulfanyl]benzene Chemical compound COC1=CC(OC)=CC(OC)=C1SSC1=C(OC)C=C(OC)C=C1OC ISMODUSOBFKDMJ-UHFFFAOYSA-N 0.000 description 1
- JZQMASJKJLKGRV-UHFFFAOYSA-N 1,3,5-tris(methylsulfinyl)-2-[[2,4,6-tris(methylsulfinyl)phenyl]disulfanyl]benzene Chemical group CS(=O)C1=CC(S(=O)C)=CC(S(C)=O)=C1SSC1=C(S(C)=O)C=C(S(C)=O)C=C1S(C)=O JZQMASJKJLKGRV-UHFFFAOYSA-N 0.000 description 1
- XQQNXKWGGSWCNE-UHFFFAOYSA-N 1,3,5-tris(trichloromethyl)-2-[[2,4,6-tris(trichloromethyl)phenyl]disulfanyl]benzene Chemical group ClC(Cl)(Cl)C1=CC(C(Cl)(Cl)Cl)=CC(C(Cl)(Cl)Cl)=C1SSC1=C(C(Cl)(Cl)Cl)C=C(C(Cl)(Cl)Cl)C=C1C(Cl)(Cl)Cl XQQNXKWGGSWCNE-UHFFFAOYSA-N 0.000 description 1
- RSZIPSKIWRSQAU-UHFFFAOYSA-N 1,4-dibromo-2-[(2,5-dibromophenyl)disulfanyl]benzene Chemical group BrC1=CC=C(Br)C(SSC=2C(=CC=C(Br)C=2)Br)=C1 RSZIPSKIWRSQAU-UHFFFAOYSA-N 0.000 description 1
- QGKFQPPIXYPVIE-UHFFFAOYSA-N 1,4-dichloro-2-[(2,5-dichlorophenyl)disulfanyl]benzene Chemical group ClC1=CC=C(Cl)C(SSC=2C(=CC=C(Cl)C=2)Cl)=C1 QGKFQPPIXYPVIE-UHFFFAOYSA-N 0.000 description 1
- FWIFXCARKJCTGL-UHFFFAOYSA-N 1,7-dimethylindole-3-carbaldehyde Chemical compound CC1=CC=CC2=C1N(C)C=C2C=O FWIFXCARKJCTGL-UHFFFAOYSA-N 0.000 description 1
- XIZZZHSGWRGMMV-UHFFFAOYSA-N 1-(2,3,5,6-tetraacetyl-4-sulfanylphenyl)ethanone Chemical compound CC(=O)C1=C(S)C(C(C)=O)=C(C(C)=O)C(C(C)=O)=C1C(C)=O XIZZZHSGWRGMMV-UHFFFAOYSA-N 0.000 description 1
- XTTRISHCQXMLJM-UHFFFAOYSA-N 1-(2,3,5,6-tetraacetyl-4-sulfanylphenyl)ethanone;zinc Chemical compound [Zn].CC(=O)C1=C(S)C(C(C)=O)=C(C(C)=O)C(C(C)=O)=C1C(C)=O XTTRISHCQXMLJM-UHFFFAOYSA-N 0.000 description 1
- ULJSOFOANORMGZ-UHFFFAOYSA-N 1-(3,5-diacetyl-4-sulfanylphenyl)ethanone Chemical compound CC(=O)C1=CC(C(C)=O)=C(S)C(C(C)=O)=C1 ULJSOFOANORMGZ-UHFFFAOYSA-N 0.000 description 1
- DTUOPSJPXLJWOH-UHFFFAOYSA-N 1-(3,5-diacetyl-4-sulfanylphenyl)ethanone;sodium Chemical compound [Na].CC(=O)C1=CC(C(C)=O)=C(S)C(C(C)=O)=C1 DTUOPSJPXLJWOH-UHFFFAOYSA-N 0.000 description 1
- DCOQXEZHJAAKSJ-UHFFFAOYSA-N 1-(3,5-diacetyl-4-sulfanylphenyl)ethanone;zinc Chemical compound [Zn].CC(=O)C1=CC(C(C)=O)=C(S)C(C(C)=O)=C1 DCOQXEZHJAAKSJ-UHFFFAOYSA-N 0.000 description 1
- JNWHTINWIJHHDJ-UHFFFAOYSA-N 1-(3-amino-4-sulfanylphenyl)ethanone Chemical compound CC(=O)C1=CC=C(S)C(N)=C1 JNWHTINWIJHHDJ-UHFFFAOYSA-N 0.000 description 1
- GSJFCOXMLCDIHU-UHFFFAOYSA-N 1-(3-amino-4-sulfanylphenyl)ethanone;sodium Chemical compound [Na].CC(=O)C1=CC=C(S)C(N)=C1 GSJFCOXMLCDIHU-UHFFFAOYSA-N 0.000 description 1
- KCEQABNTVAFCSZ-UHFFFAOYSA-N 1-(3-amino-4-sulfanylphenyl)ethanone;zinc Chemical compound [Zn].CC(=O)C1=CC=C(S)C(N)=C1 KCEQABNTVAFCSZ-UHFFFAOYSA-N 0.000 description 1
- FXMMKPVMYJWDEB-UHFFFAOYSA-N 1-(3-chloro-4-sulfanylphenyl)ethanone Chemical compound CC(=O)C1=CC=C(S)C(Cl)=C1 FXMMKPVMYJWDEB-UHFFFAOYSA-N 0.000 description 1
- XCYVLVHGEGCSKI-UHFFFAOYSA-N 1-(3-chloro-4-sulfanylphenyl)ethanone;sodium Chemical compound [Na].CC(=O)C1=CC=C(S)C(Cl)=C1 XCYVLVHGEGCSKI-UHFFFAOYSA-N 0.000 description 1
- IDFODVHVKZFGMV-UHFFFAOYSA-N 1-(3-chloro-4-sulfanylphenyl)ethanone;zinc Chemical compound [Zn].CC(=O)C1=CC=C(S)C(Cl)=C1 IDFODVHVKZFGMV-UHFFFAOYSA-N 0.000 description 1
- NQEBFQGUGIISNJ-UHFFFAOYSA-N 1-(3-hydroxy-4-sulfanylphenyl)ethanone Chemical compound CC(=O)C1=CC=C(S)C(O)=C1 NQEBFQGUGIISNJ-UHFFFAOYSA-N 0.000 description 1
- DJQBUSYSBZWPBF-UHFFFAOYSA-N 1-(3-hydroxy-4-sulfanylphenyl)ethanone;sodium Chemical compound [Na].CC(=O)C1=CC=C(S)C(O)=C1 DJQBUSYSBZWPBF-UHFFFAOYSA-N 0.000 description 1
- VBDFKODVTAWPLR-UHFFFAOYSA-N 1-(3-hydroxy-4-sulfanylphenyl)ethanone;zinc Chemical compound [Zn].CC(=O)C1=CC=C(S)C(O)=C1 VBDFKODVTAWPLR-UHFFFAOYSA-N 0.000 description 1
- JSWOOQQBQYKDEW-UHFFFAOYSA-N 1-(3-methoxy-4-sulfanylphenyl)ethanone Chemical compound COC1=CC(C(C)=O)=CC=C1S JSWOOQQBQYKDEW-UHFFFAOYSA-N 0.000 description 1
- GVBILKFAIWKPQD-UHFFFAOYSA-N 1-(3-methoxy-4-sulfanylphenyl)ethanone;sodium Chemical compound [Na].COC1=CC(C(C)=O)=CC=C1S GVBILKFAIWKPQD-UHFFFAOYSA-N 0.000 description 1
- NOXZFXQTTZPGMR-UHFFFAOYSA-N 1-(3-methoxy-4-sulfanylphenyl)ethanone;zinc Chemical compound [Zn].COC1=CC(C(C)=O)=CC=C1S NOXZFXQTTZPGMR-UHFFFAOYSA-N 0.000 description 1
- JJWURSKHYARVBM-UHFFFAOYSA-N 1-(3-methyl-4-sulfanylphenyl)ethanone Chemical compound CC(=O)C1=CC=C(S)C(C)=C1 JJWURSKHYARVBM-UHFFFAOYSA-N 0.000 description 1
- YUIAIZWPYUTANI-UHFFFAOYSA-N 1-(3-methyl-4-sulfanylphenyl)ethanone;sodium Chemical compound [Na].CC(=O)C1=CC=C(S)C(C)=C1 YUIAIZWPYUTANI-UHFFFAOYSA-N 0.000 description 1
- RPCLINMXJORUGV-UHFFFAOYSA-N 1-(3-methyl-4-sulfanylphenyl)ethanone;zinc Chemical compound [Zn].CC(=O)C1=CC=C(S)C(C)=C1 RPCLINMXJORUGV-UHFFFAOYSA-N 0.000 description 1
- YFYBYHDKWLGNKM-UHFFFAOYSA-N 1-(3-methylsulfinyl-4-sulfanylphenyl)ethanone Chemical compound CC(=O)C1=CC=C(S)C(S(C)=O)=C1 YFYBYHDKWLGNKM-UHFFFAOYSA-N 0.000 description 1
- HRMWTYDHVURIDQ-UHFFFAOYSA-N 1-(3-methylsulfinyl-4-sulfanylphenyl)ethanone;sodium Chemical compound [Na].CC(=O)C1=CC=C(S)C(S(C)=O)=C1 HRMWTYDHVURIDQ-UHFFFAOYSA-N 0.000 description 1
- YXBDQLHKQWIPTE-UHFFFAOYSA-N 1-(3-methylsulfinyl-4-sulfanylphenyl)ethanone;zinc Chemical compound [Zn].CC(=O)C1=CC=C(S)C(S(C)=O)=C1 YXBDQLHKQWIPTE-UHFFFAOYSA-N 0.000 description 1
- PBLVWSWMHATILR-UHFFFAOYSA-N 1-(3-nitro-4-sulfanylphenyl)ethanone Chemical compound CC(=O)C1=CC=C(S)C([N+]([O-])=O)=C1 PBLVWSWMHATILR-UHFFFAOYSA-N 0.000 description 1
- XBEFHJAUHUFYQF-UHFFFAOYSA-N 1-(3-nitro-4-sulfanylphenyl)ethanone;sodium Chemical compound [Na].CC(=O)C1=CC=C(S)C([N+]([O-])=O)=C1 XBEFHJAUHUFYQF-UHFFFAOYSA-N 0.000 description 1
- WCUFIXKAXLOEHU-UHFFFAOYSA-N 1-(3-nitro-4-sulfanylphenyl)ethanone;zinc Chemical compound [Zn].CC(=O)C1=CC=C(S)C([N+]([O-])=O)=C1 WCUFIXKAXLOEHU-UHFFFAOYSA-N 0.000 description 1
- PGOTXOAWBWTJEY-UHFFFAOYSA-N 1-(3-phenylsulfanyl-4-sulfanylphenyl)ethanone Chemical compound CC(=O)C1=CC=C(S)C(SC=2C=CC=CC=2)=C1 PGOTXOAWBWTJEY-UHFFFAOYSA-N 0.000 description 1
- NUSXKJIDFXFLTA-UHFFFAOYSA-N 1-(3-phenylsulfanyl-4-sulfanylphenyl)ethanone;sodium Chemical compound [Na].CC(=O)C1=CC=C(S)C(SC=2C=CC=CC=2)=C1 NUSXKJIDFXFLTA-UHFFFAOYSA-N 0.000 description 1
- TYTRCAZVUJXNOT-UHFFFAOYSA-N 1-(3-phenylsulfanyl-4-sulfanylphenyl)ethanone;zinc Chemical compound [Zn].CC(=O)C1=CC=C(S)C(SC=2C=CC=CC=2)=C1 TYTRCAZVUJXNOT-UHFFFAOYSA-N 0.000 description 1
- QNGBRPMOFJSFMF-UHFFFAOYSA-N 1-(4-sulfanylphenyl)ethanone Chemical compound CC(=O)C1=CC=C(S)C=C1 QNGBRPMOFJSFMF-UHFFFAOYSA-N 0.000 description 1
- QOZVQEFGFCYCQN-UHFFFAOYSA-N 1-(4-sulfanylphenyl)ethanone;zinc Chemical compound [Zn].CC(=O)C1=CC=C(S)C=C1 QOZVQEFGFCYCQN-UHFFFAOYSA-N 0.000 description 1
- HVUJKYBSQWTDOI-UHFFFAOYSA-N 1-(trichloromethyl)-4-[[4-(trichloromethyl)phenyl]disulfanyl]benzene Chemical group C1=CC(C(Cl)(Cl)Cl)=CC=C1SSC1=CC=C(C(Cl)(Cl)Cl)C=C1 HVUJKYBSQWTDOI-UHFFFAOYSA-N 0.000 description 1
- NVPBPCGLTIWQRG-UHFFFAOYSA-N 1-[2,3,5,6-tetraacetyl-4-[(2,3,4,5,6-pentaacetylphenyl)disulfanyl]phenyl]ethanone Chemical group CC(=O)C1=C(C(C)=O)C(C(C)=O)=C(C(C)=O)C(C(=O)C)=C1SSC1=C(C(C)=O)C(C(C)=O)=C(C(C)=O)C(C(C)=O)=C1C(C)=O NVPBPCGLTIWQRG-UHFFFAOYSA-N 0.000 description 1
- FOTOFSXRCHWQHH-UHFFFAOYSA-N 1-[3,5-diacetyl-4-[(2,4,6-triacetylphenyl)disulfanyl]phenyl]ethanone Chemical group CC(=O)C1=CC(C(=O)C)=CC(C(C)=O)=C1SSC1=C(C(C)=O)C=C(C(C)=O)C=C1C(C)=O FOTOFSXRCHWQHH-UHFFFAOYSA-N 0.000 description 1
- CIUOHXUMPRZDDT-UHFFFAOYSA-N 1-[4-[(4-acetyl-2-aminophenyl)disulfanyl]-3-aminophenyl]ethanone Chemical compound NC1=CC(C(=O)C)=CC=C1SSC1=CC=C(C(C)=O)C=C1N CIUOHXUMPRZDDT-UHFFFAOYSA-N 0.000 description 1
- CHYIQHUZLDAXRV-UHFFFAOYSA-N 1-[4-[(4-acetyl-2-chlorophenyl)disulfanyl]-3-chlorophenyl]ethanone Chemical compound ClC1=CC(C(=O)C)=CC=C1SSC1=CC=C(C(C)=O)C=C1Cl CHYIQHUZLDAXRV-UHFFFAOYSA-N 0.000 description 1
- CBXSTRBNOUYYDL-UHFFFAOYSA-N 1-[4-[(4-acetyl-2-hydroxyphenyl)disulfanyl]-3-hydroxyphenyl]ethanone Chemical compound OC1=CC(C(=O)C)=CC=C1SSC1=CC=C(C(C)=O)C=C1O CBXSTRBNOUYYDL-UHFFFAOYSA-N 0.000 description 1
- RJDYMWVFCWIANA-UHFFFAOYSA-N 1-[4-[(4-acetyl-2-methoxyphenyl)disulfanyl]-3-methoxyphenyl]ethanone Chemical compound COC1=CC(C(C)=O)=CC=C1SSC1=CC=C(C(C)=O)C=C1OC RJDYMWVFCWIANA-UHFFFAOYSA-N 0.000 description 1
- RZULWONZSWUMCA-UHFFFAOYSA-N 1-[4-[(4-acetyl-2-methylphenyl)disulfanyl]-3-methylphenyl]ethanone Chemical compound CC1=CC(C(=O)C)=CC=C1SSC1=CC=C(C(C)=O)C=C1C RZULWONZSWUMCA-UHFFFAOYSA-N 0.000 description 1
- KHUIGFYOCYWGOV-UHFFFAOYSA-N 1-[4-[(4-acetyl-2-methylsulfinylphenyl)disulfanyl]-3-methylsulfinylphenyl]ethanone Chemical compound CS(=O)C1=CC(C(=O)C)=CC=C1SSC1=CC=C(C(C)=O)C=C1S(C)=O KHUIGFYOCYWGOV-UHFFFAOYSA-N 0.000 description 1
- QYJHGWFGUKJFPT-UHFFFAOYSA-N 1-[4-[(4-acetyl-2-nitrophenyl)disulfanyl]-3-nitrophenyl]ethanone Chemical compound [O-][N+](=O)C1=CC(C(=O)C)=CC=C1SSC1=CC=C(C(C)=O)C=C1[N+]([O-])=O QYJHGWFGUKJFPT-UHFFFAOYSA-N 0.000 description 1
- RRJHCMPJUMFLNP-UHFFFAOYSA-N 1-[4-[(4-acetyl-2-phenylsulfanylphenyl)disulfanyl]-3-phenylsulfanylphenyl]ethanone Chemical compound C=1C=CC=CC=1SC1=CC(C(=O)C)=CC=C1SSC1=CC=C(C(C)=O)C=C1SC1=CC=CC=C1 RRJHCMPJUMFLNP-UHFFFAOYSA-N 0.000 description 1
- KIRIOGATKMSJIW-UHFFFAOYSA-N 1-[4-[(4-acetylphenyl)disulfanyl]phenyl]ethanone Chemical group C1=CC(C(=O)C)=CC=C1SSC1=CC=C(C(C)=O)C=C1 KIRIOGATKMSJIW-UHFFFAOYSA-N 0.000 description 1
- MHLSHEVPZJCCPR-UHFFFAOYSA-N 1-[4-[[4-acetyl-2-(trichloromethyl)phenyl]disulfanyl]-3-(trichloromethyl)phenyl]ethanone Chemical compound ClC(Cl)(Cl)C1=CC(C(=O)C)=CC=C1SSC1=CC=C(C(C)=O)C=C1C(Cl)(Cl)Cl MHLSHEVPZJCCPR-UHFFFAOYSA-N 0.000 description 1
- SPJJRUNDNDXMEO-UHFFFAOYSA-N 1-[4-sulfanyl-3-(trichloromethyl)phenyl]ethanone Chemical compound CC(=O)C1=CC=C(S)C(C(Cl)(Cl)Cl)=C1 SPJJRUNDNDXMEO-UHFFFAOYSA-N 0.000 description 1
- FZZVVFSNRUMXNR-UHFFFAOYSA-N 1-[4-sulfanyl-3-(trichloromethyl)phenyl]ethanone;zinc Chemical compound [Zn].CC(=O)C1=CC=C(S)C(C(Cl)(Cl)Cl)=C1 FZZVVFSNRUMXNR-UHFFFAOYSA-N 0.000 description 1
- GJKRYLGFYSBCNW-UHFFFAOYSA-N 1-[[2,3,4,5,6-pentakis(trichloromethyl)phenyl]disulfanyl]-2,3,4,5,6-pentakis(trichloromethyl)benzene Chemical group ClC(Cl)(Cl)C1=C(C(Cl)(Cl)Cl)C(C(Cl)(Cl)Cl)=C(C(Cl)(Cl)Cl)C(C(Cl)(Cl)Cl)=C1SSC1=C(C(Cl)(Cl)Cl)C(C(Cl)(Cl)Cl)=C(C(Cl)(Cl)Cl)C(C(Cl)(Cl)Cl)=C1C(Cl)(Cl)Cl GJKRYLGFYSBCNW-UHFFFAOYSA-N 0.000 description 1
- VZQVHIINDXJOQK-UHFFFAOYSA-N 1-bromo-4-[(4-bromophenyl)disulfanyl]benzene Chemical group C1=CC(Br)=CC=C1SSC1=CC=C(Br)C=C1 VZQVHIINDXJOQK-UHFFFAOYSA-N 0.000 description 1
- ZIXXRXGPBFMPFD-UHFFFAOYSA-N 1-chloro-4-[(4-chlorophenyl)disulfanyl]benzene Chemical group C1=CC(Cl)=CC=C1SSC1=CC=C(Cl)C=C1 ZIXXRXGPBFMPFD-UHFFFAOYSA-N 0.000 description 1
- SLXZJIKDNHDPKL-UHFFFAOYSA-N 1-fluoro-4-[(4-fluorophenyl)disulfanyl]benzene Chemical group C1=CC(F)=CC=C1SSC1=CC=C(F)C=C1 SLXZJIKDNHDPKL-UHFFFAOYSA-N 0.000 description 1
- VBSTXRUAXCTZBQ-UHFFFAOYSA-N 1-hexyl-4-phenylpiperazine Chemical compound C1CN(CCCCCC)CCN1C1=CC=CC=C1 VBSTXRUAXCTZBQ-UHFFFAOYSA-N 0.000 description 1
- HABDEXOINBJHPA-UHFFFAOYSA-N 1-iodo-4-[(4-iodophenyl)disulfanyl]benzene Chemical group C1=CC(I)=CC=C1SSC1=CC=C(I)C=C1 HABDEXOINBJHPA-UHFFFAOYSA-N 0.000 description 1
- PZQGLCGLPMWYBT-UHFFFAOYSA-N 1-methoxy-4-[(4-methoxyphenyl)disulfanyl]benzene Chemical compound C1=CC(OC)=CC=C1SSC1=CC=C(OC)C=C1 PZQGLCGLPMWYBT-UHFFFAOYSA-N 0.000 description 1
- TZOVOULUMXXLOJ-UHFFFAOYSA-N 1-methyl-4-[(4-methylphenyl)disulfanyl]benzene Chemical group C1=CC(C)=CC=C1SSC1=CC=C(C)C=C1 TZOVOULUMXXLOJ-UHFFFAOYSA-N 0.000 description 1
- IYPSPAHCKHHDME-UHFFFAOYSA-N 1-methylsulfinyl-4-[(4-methylsulfinylphenyl)disulfanyl]benzene Chemical group C1=CC(S(=O)C)=CC=C1SSC1=CC=C(S(C)=O)C=C1 IYPSPAHCKHHDME-UHFFFAOYSA-N 0.000 description 1
- JHJKMOPTKMUFOG-UHFFFAOYSA-N 1-tert-butyl-4-[(4-tert-butylphenyl)disulfanyl]benzene Chemical group C1=CC(C(C)(C)C)=CC=C1SSC1=CC=C(C(C)(C)C)C=C1 JHJKMOPTKMUFOG-UHFFFAOYSA-N 0.000 description 1
- 229940114072 12-hydroxystearic acid Drugs 0.000 description 1
- UTLUYJULFYZZTK-UHFFFAOYSA-N 2,3,4,5,6-pentabromobenzenethiol Chemical compound SC1=C(Br)C(Br)=C(Br)C(Br)=C1Br UTLUYJULFYZZTK-UHFFFAOYSA-N 0.000 description 1
- DJXXRGIOQUPUFT-UHFFFAOYSA-N 2,3,4,5,6-pentabromobenzenethiol;sodium Chemical compound [Na].SC1=C(Br)C(Br)=C(Br)C(Br)=C1Br DJXXRGIOQUPUFT-UHFFFAOYSA-N 0.000 description 1
- YQXUITOUDPPOFS-UHFFFAOYSA-N 2,3,4,5,6-pentabromobenzenethiol;zinc Chemical compound [Zn].SC1=C(Br)C(Br)=C(Br)C(Br)=C1Br YQXUITOUDPPOFS-UHFFFAOYSA-N 0.000 description 1
- LLMLGZUZTFMXSA-UHFFFAOYSA-N 2,3,4,5,6-pentachlorobenzenethiol Chemical compound SC1=C(Cl)C(Cl)=C(Cl)C(Cl)=C1Cl LLMLGZUZTFMXSA-UHFFFAOYSA-N 0.000 description 1
- CHBIPFPNKXVVJN-UHFFFAOYSA-N 2,3,4,5,6-pentachlorobenzenethiol;sodium Chemical compound [Na].SC1=C(Cl)C(Cl)=C(Cl)C(Cl)=C1Cl CHBIPFPNKXVVJN-UHFFFAOYSA-N 0.000 description 1
- XCPAVPRPNMYCDD-UHFFFAOYSA-N 2,3,4,5,6-pentachlorobenzenethiol;zinc Chemical compound [Zn].SC1=C(Cl)C(Cl)=C(Cl)C(Cl)=C1Cl XCPAVPRPNMYCDD-UHFFFAOYSA-N 0.000 description 1
- UVAMFBJPMUMURT-UHFFFAOYSA-N 2,3,4,5,6-pentafluorobenzenethiol Chemical compound FC1=C(F)C(F)=C(S)C(F)=C1F UVAMFBJPMUMURT-UHFFFAOYSA-N 0.000 description 1
- VVJWWPYXDZEGEV-UHFFFAOYSA-N 2,3,4,5,6-pentafluorobenzenethiol;sodium Chemical compound [Na].FC1=C(F)C(F)=C(S)C(F)=C1F VVJWWPYXDZEGEV-UHFFFAOYSA-N 0.000 description 1
- LGHBUCIVKPTXER-UHFFFAOYSA-N 2,3,4,5,6-pentaiodobenzenethiol Chemical compound SC1=C(I)C(I)=C(I)C(I)=C1I LGHBUCIVKPTXER-UHFFFAOYSA-N 0.000 description 1
- VTDAPQPMISMQHE-UHFFFAOYSA-N 2,3,4,5,6-pentaiodobenzenethiol;sodium Chemical compound [Na].SC1=C(I)C(I)=C(I)C(I)=C1I VTDAPQPMISMQHE-UHFFFAOYSA-N 0.000 description 1
- RDVSWVYMULRAHH-UHFFFAOYSA-N 2,3,4,5,6-pentaiodobenzenethiol;zinc Chemical compound [Zn].SC1=C(I)C(I)=C(I)C(I)=C1I RDVSWVYMULRAHH-UHFFFAOYSA-N 0.000 description 1
- CPHFXOPRBDPPHJ-UHFFFAOYSA-N 2,3,4,5,6-pentakis(methylsulfinyl)benzenethiol;zinc Chemical compound [Zn].CS(=O)C1=C(S)C(S(C)=O)=C(S(C)=O)C(S(C)=O)=C1S(C)=O CPHFXOPRBDPPHJ-UHFFFAOYSA-N 0.000 description 1
- LXFFDXPAHCPDFG-UHFFFAOYSA-N 2,3,4,5,6-pentakis(trichloromethyl)benzenethiol Chemical compound SC1=C(C(Cl)(Cl)Cl)C(C(Cl)(Cl)Cl)=C(C(Cl)(Cl)Cl)C(C(Cl)(Cl)Cl)=C1C(Cl)(Cl)Cl LXFFDXPAHCPDFG-UHFFFAOYSA-N 0.000 description 1
- QWGUDXHIEHKIEM-UHFFFAOYSA-N 2,3,4,5,6-pentakis(trichloromethyl)benzenethiol;sodium Chemical compound [Na].SC1=C(C(Cl)(Cl)Cl)C(C(Cl)(Cl)Cl)=C(C(Cl)(Cl)Cl)C(C(Cl)(Cl)Cl)=C1C(Cl)(Cl)Cl QWGUDXHIEHKIEM-UHFFFAOYSA-N 0.000 description 1
- CZSPCIRSFJFSOA-UHFFFAOYSA-N 2,3,4,5,6-pentakis(trichloromethyl)benzenethiol;zinc Chemical compound [Zn].SC1=C(C(Cl)(Cl)Cl)C(C(Cl)(Cl)Cl)=C(C(Cl)(Cl)Cl)C(C(Cl)(Cl)Cl)=C1C(Cl)(Cl)Cl CZSPCIRSFJFSOA-UHFFFAOYSA-N 0.000 description 1
- JCLUHBSZTZLWPJ-UHFFFAOYSA-N 2,3,4,5,6-pentamethoxybenzenethiol Chemical compound COC1=C(S)C(OC)=C(OC)C(OC)=C1OC JCLUHBSZTZLWPJ-UHFFFAOYSA-N 0.000 description 1
- WJWKMIVXAUMDBF-UHFFFAOYSA-N 2,3,4,5,6-pentamethoxybenzenethiol;sodium Chemical compound [Na].COC1=C(S)C(OC)=C(OC)C(OC)=C1OC WJWKMIVXAUMDBF-UHFFFAOYSA-N 0.000 description 1
- KFBUDPOFIGIPOG-UHFFFAOYSA-N 2,3,4,5,6-pentamethoxybenzenethiol;zinc Chemical compound [Zn].COC1=C(S)C(OC)=C(OC)C(OC)=C1OC KFBUDPOFIGIPOG-UHFFFAOYSA-N 0.000 description 1
- TZUPAWCZQMMBGH-UHFFFAOYSA-N 2,3,4,5,6-pentamethylbenzenethiol Chemical compound CC1=C(C)C(C)=C(S)C(C)=C1C TZUPAWCZQMMBGH-UHFFFAOYSA-N 0.000 description 1
- XFEIPPPLGZLXOO-UHFFFAOYSA-N 2,3,4,5,6-pentamethylbenzenethiol;sodium Chemical compound [Na].CC1=C(C)C(C)=C(S)C(C)=C1C XFEIPPPLGZLXOO-UHFFFAOYSA-N 0.000 description 1
- OYOCVDPKJHPRKV-UHFFFAOYSA-N 2,3,4,5,6-pentamethylbenzenethiol;zinc Chemical compound [Zn].CC1=C(C)C(C)=C(S)C(C)=C1C OYOCVDPKJHPRKV-UHFFFAOYSA-N 0.000 description 1
- WZJSOPXGVJAHJP-UHFFFAOYSA-N 2,3,4,5,6-pentatert-butylbenzenethiol Chemical compound CC(C)(C)C1=C(S)C(C(C)(C)C)=C(C(C)(C)C)C(C(C)(C)C)=C1C(C)(C)C WZJSOPXGVJAHJP-UHFFFAOYSA-N 0.000 description 1
- BQCBMYSESKIIIL-UHFFFAOYSA-N 2,3,4,5,6-pentatert-butylbenzenethiol;sodium Chemical compound [Na].CC(C)(C)C1=C(S)C(C(C)(C)C)=C(C(C)(C)C)C(C(C)(C)C)=C1C(C)(C)C BQCBMYSESKIIIL-UHFFFAOYSA-N 0.000 description 1
- GXKOMLNEOQUFBM-UHFFFAOYSA-N 2,3,4,5,6-pentatert-butylbenzenethiol;zinc Chemical compound [Zn].CC(C)(C)C1=C(S)C(C(C)(C)C)=C(C(C)(C)C)C(C(C)(C)C)=C1C(C)(C)C GXKOMLNEOQUFBM-UHFFFAOYSA-N 0.000 description 1
- FDMIQHRXURRUEC-UHFFFAOYSA-N 2,3,4,6-tetrabromobenzenethiol Chemical compound SC1=C(Br)C=C(Br)C(Br)=C1Br FDMIQHRXURRUEC-UHFFFAOYSA-N 0.000 description 1
- YXDWNVHLJGWDFH-UHFFFAOYSA-N 2,3,4,6-tetrabromobenzenethiol;zinc Chemical compound [Zn].SC1=C(Br)C=C(Br)C(Br)=C1Br YXDWNVHLJGWDFH-UHFFFAOYSA-N 0.000 description 1
- DKRBSULGQNLLNQ-UHFFFAOYSA-N 2,3,4,6-tetrachlorobenzenethiol Chemical compound SC1=C(Cl)C=C(Cl)C(Cl)=C1Cl DKRBSULGQNLLNQ-UHFFFAOYSA-N 0.000 description 1
- WSKUGTOZIZBMAW-UHFFFAOYSA-N 2,3,4,6-tetrachlorobenzenethiol;zinc Chemical compound [Zn].SC1=C(Cl)C=C(Cl)C(Cl)=C1Cl WSKUGTOZIZBMAW-UHFFFAOYSA-N 0.000 description 1
- VKRDINAGHCFZLB-UHFFFAOYSA-N 2,3,4,6-tetrafluorobenzenethiol Chemical compound FC1=CC(F)=C(S)C(F)=C1F VKRDINAGHCFZLB-UHFFFAOYSA-N 0.000 description 1
- ZXPLOIYRWZNJHI-UHFFFAOYSA-N 2,3,4,6-tetrafluorobenzenethiol;zinc Chemical compound [Zn].FC1=CC(F)=C(S)C(F)=C1F ZXPLOIYRWZNJHI-UHFFFAOYSA-N 0.000 description 1
- INKQDIYIUXDCRW-UHFFFAOYSA-N 2,3,4,6-tetraiodobenzenethiol Chemical compound SC1=C(I)C=C(I)C(I)=C1I INKQDIYIUXDCRW-UHFFFAOYSA-N 0.000 description 1
- HQYJQJILCDWSMA-UHFFFAOYSA-N 2,3,4,6-tetraiodobenzenethiol;zinc Chemical compound [Zn].SC1=C(I)C=C(I)C(I)=C1I HQYJQJILCDWSMA-UHFFFAOYSA-N 0.000 description 1
- HDIJZFORGDBEKL-UHFFFAOYSA-N 2,3,4-trimethylbenzoic acid Chemical compound CC1=CC=C(C(O)=O)C(C)=C1C HDIJZFORGDBEKL-UHFFFAOYSA-N 0.000 description 1
- KDUZDSXRZJLZNA-UHFFFAOYSA-N 2,3-dimethoxybenzoic acid Chemical compound C(C1=C(OC)C(OC)=CC=C1)(=O)O.COC1=C(C(=O)O)C=CC=C1OC KDUZDSXRZJLZNA-UHFFFAOYSA-N 0.000 description 1
- RIZUCYSQUWMQLX-UHFFFAOYSA-N 2,3-dimethylbenzoic acid Chemical compound CC1=CC=CC(C(O)=O)=C1C RIZUCYSQUWMQLX-UHFFFAOYSA-N 0.000 description 1
- WFIJMPSSNFCYEN-UHFFFAOYSA-N 2,4,5-tribromobenzenethiol Chemical compound SC1=CC(Br)=C(Br)C=C1Br WFIJMPSSNFCYEN-UHFFFAOYSA-N 0.000 description 1
- LXGPJIDOUYPBEE-UHFFFAOYSA-N 2,4,5-tribromobenzenethiol;zinc Chemical compound [Zn].SC1=CC(Br)=C(Br)C=C1Br LXGPJIDOUYPBEE-UHFFFAOYSA-N 0.000 description 1
- JARIALSGFXECCH-UHFFFAOYSA-N 2,4,5-trichlorobenzenethiol Chemical compound SC1=CC(Cl)=C(Cl)C=C1Cl JARIALSGFXECCH-UHFFFAOYSA-N 0.000 description 1
- JFDFCMASTJJMIX-UHFFFAOYSA-N 2,4,5-trichlorobenzenethiol;zinc Chemical compound [Zn].SC1=CC(Cl)=C(Cl)C=C1Cl JFDFCMASTJJMIX-UHFFFAOYSA-N 0.000 description 1
- ODVDATKZUNPWNS-UHFFFAOYSA-N 2,4,5-trifluorobenzenethiol Chemical compound FC1=CC(F)=C(S)C=C1F ODVDATKZUNPWNS-UHFFFAOYSA-N 0.000 description 1
- POAYHFYRJJIPHV-UHFFFAOYSA-N 2,4,5-trifluorobenzenethiol;zinc Chemical compound [Zn].FC1=CC(F)=C(S)C=C1F POAYHFYRJJIPHV-UHFFFAOYSA-N 0.000 description 1
- RIJUWWHCFCDBLC-UHFFFAOYSA-N 2,4,5-triiodobenzenethiol Chemical compound SC1=CC(I)=C(I)C=C1I RIJUWWHCFCDBLC-UHFFFAOYSA-N 0.000 description 1
- UOEJGYJOPXJVRD-UHFFFAOYSA-N 2,4,5-triiodobenzenethiol;zinc Chemical compound [Zn].SC1=CC(I)=C(I)C=C1I UOEJGYJOPXJVRD-UHFFFAOYSA-N 0.000 description 1
- JSYBJWAZPHAASJ-UHFFFAOYSA-N 2,4,5-trimethylbenzenethiol Chemical compound CC1=CC(C)=C(S)C=C1C JSYBJWAZPHAASJ-UHFFFAOYSA-N 0.000 description 1
- ZNFOAVMRTCZOPW-UHFFFAOYSA-N 2,4,5-trimethylbenzenethiol;zinc Chemical compound [Zn].CC1=CC(C)=C(S)C=C1C ZNFOAVMRTCZOPW-UHFFFAOYSA-N 0.000 description 1
- OCNKCJJJHJKXCE-UHFFFAOYSA-N 2,4,5-tritert-butylbenzenethiol Chemical compound CC(C)(C)C1=CC(C(C)(C)C)=C(C(C)(C)C)C=C1S OCNKCJJJHJKXCE-UHFFFAOYSA-N 0.000 description 1
- UTFXNBHETHXNQJ-UHFFFAOYSA-N 2,4,5-tritert-butylbenzenethiol;zinc Chemical compound [Zn].CC(C)(C)C1=CC(C(C)(C)C)=C(C(C)(C)C)C=C1S UTFXNBHETHXNQJ-UHFFFAOYSA-N 0.000 description 1
- DZWLIENCQXGZFF-UHFFFAOYSA-N 2,4,6-trimethoxybenzenethiol Chemical compound COC1=CC(OC)=C(S)C(OC)=C1 DZWLIENCQXGZFF-UHFFFAOYSA-N 0.000 description 1
- IWBKNHHXBSNIQF-UHFFFAOYSA-N 2,4,6-trimethoxybenzenethiol;zinc Chemical compound [Zn].COC1=CC(OC)=C(S)C(OC)=C1 IWBKNHHXBSNIQF-UHFFFAOYSA-N 0.000 description 1
- UAKLTVDHTCGWJL-UHFFFAOYSA-N 2,4,6-trimethylbenzoic acid Chemical compound CC1=CC(C)=C(C(O)=O)C(C)=C1.CC1=CC(C)=C(C(O)=O)C(C)=C1 UAKLTVDHTCGWJL-UHFFFAOYSA-N 0.000 description 1
- HQRKJCGLBJXICR-UHFFFAOYSA-N 2,4,6-tris(methylsulfinyl)benzenethiol Chemical compound CS(=O)C1=CC(S(C)=O)=C(S)C(S(C)=O)=C1 HQRKJCGLBJXICR-UHFFFAOYSA-N 0.000 description 1
- NMWAZBMYQIPEPF-UHFFFAOYSA-N 2,4,6-tris(methylsulfinyl)benzenethiol;zinc Chemical compound [Zn].CS(=O)C1=CC(S(C)=O)=C(S)C(S(C)=O)=C1 NMWAZBMYQIPEPF-UHFFFAOYSA-N 0.000 description 1
- PMQYMQKSUIZCFM-UHFFFAOYSA-N 2,4,6-tris(trichloromethyl)benzenethiol Chemical compound SC1=C(C(Cl)(Cl)Cl)C=C(C(Cl)(Cl)Cl)C=C1C(Cl)(Cl)Cl PMQYMQKSUIZCFM-UHFFFAOYSA-N 0.000 description 1
- ONKFTXCAVUDCAZ-UHFFFAOYSA-N 2,4,6-tris(trichloromethyl)benzenethiol;zinc Chemical compound [Zn].SC1=C(C(Cl)(Cl)Cl)C=C(C(Cl)(Cl)Cl)C=C1C(Cl)(Cl)Cl ONKFTXCAVUDCAZ-UHFFFAOYSA-N 0.000 description 1
- FGBVJFREPSJSNG-UHFFFAOYSA-N 2,4-dichlorobenzenethiol Chemical compound SC1=CC=C(Cl)C=C1Cl FGBVJFREPSJSNG-UHFFFAOYSA-N 0.000 description 1
- DMWVYCCGCQPJEA-UHFFFAOYSA-N 2,5-bis(tert-butylperoxy)-2,5-dimethylhexane Chemical compound CC(C)(C)OOC(C)(C)CCC(C)(C)OOC(C)(C)C DMWVYCCGCQPJEA-UHFFFAOYSA-N 0.000 description 1
- ZUAMDMWZDFEYFG-UHFFFAOYSA-N 2,5-dibromobenzenethiol Chemical compound SC1=CC(Br)=CC=C1Br ZUAMDMWZDFEYFG-UHFFFAOYSA-N 0.000 description 1
- FRXGYGPYPRAIPO-UHFFFAOYSA-N 2,5-dibromobenzenethiol;sodium Chemical compound [Na].SC1=CC(Br)=CC=C1Br FRXGYGPYPRAIPO-UHFFFAOYSA-N 0.000 description 1
- SPXUBWWVBCGANM-UHFFFAOYSA-N 2,5-dibromobenzenethiol;zinc Chemical compound [Zn].SC1=CC(Br)=CC=C1Br SPXUBWWVBCGANM-UHFFFAOYSA-N 0.000 description 1
- QIULLHZMZMGGFH-UHFFFAOYSA-N 2,5-dichlorobenzenethiol Chemical compound SC1=CC(Cl)=CC=C1Cl QIULLHZMZMGGFH-UHFFFAOYSA-N 0.000 description 1
- SLWPINCUBCBQHP-UHFFFAOYSA-N 2,5-dichlorobenzenethiol;sodium Chemical compound [Na].SC1=CC(Cl)=CC=C1Cl SLWPINCUBCBQHP-UHFFFAOYSA-N 0.000 description 1
- PZWCYHUBFBXIOH-UHFFFAOYSA-N 2,5-dichlorobenzenethiol;zinc Chemical compound [Zn].SC1=CC(Cl)=CC=C1Cl PZWCYHUBFBXIOH-UHFFFAOYSA-N 0.000 description 1
- PQRVQUXEBQKVEQ-UHFFFAOYSA-N 2,5-difluorobenzenethiol Chemical compound FC1=CC=C(F)C(S)=C1 PQRVQUXEBQKVEQ-UHFFFAOYSA-N 0.000 description 1
- IILQWOODNUQYQD-UHFFFAOYSA-N 2,5-difluorobenzenethiol;sodium Chemical compound [Na].FC1=CC=C(F)C(S)=C1 IILQWOODNUQYQD-UHFFFAOYSA-N 0.000 description 1
- HTRWCXLZZJNPCH-UHFFFAOYSA-N 2,5-difluorobenzenethiol;zinc Chemical compound [Zn].FC1=CC=C(F)C(S)=C1 HTRWCXLZZJNPCH-UHFFFAOYSA-N 0.000 description 1
- YTWHOCPMYGUYHD-UHFFFAOYSA-N 2,5-diiodobenzenethiol Chemical compound SC1=CC(I)=CC=C1I YTWHOCPMYGUYHD-UHFFFAOYSA-N 0.000 description 1
- BXFYSBZPGUEMBC-UHFFFAOYSA-N 2,5-diiodobenzenethiol;sodium Chemical compound [Na].SC1=CC(I)=CC=C1I BXFYSBZPGUEMBC-UHFFFAOYSA-N 0.000 description 1
- SSHRHHWAYFBOJB-UHFFFAOYSA-N 2,5-diiodobenzenethiol;zinc Chemical compound [Zn].SC1=CC(I)=CC=C1I SSHRHHWAYFBOJB-UHFFFAOYSA-N 0.000 description 1
- VVEMXQMSJUUTFO-UHFFFAOYSA-N 2,6-dihydroxybenzoic acid Chemical compound OC(=O)C1=C(O)C=CC=C1O.OC(=O)C1=C(O)C=CC=C1O VVEMXQMSJUUTFO-UHFFFAOYSA-N 0.000 description 1
- GSFSVEDCYBDIGW-UHFFFAOYSA-N 2-(1,3-benzothiazol-2-yl)-6-chlorophenol Chemical compound OC1=C(Cl)C=CC=C1C1=NC2=CC=CC=C2S1 GSFSVEDCYBDIGW-UHFFFAOYSA-N 0.000 description 1
- JDICEKWSLNPYSN-UHFFFAOYSA-N 2-(2,4-dinitrophenyl)-1,3-benzothiazole-4-thiol Chemical compound [O-][N+](=O)C1=CC([N+](=O)[O-])=CC=C1C1=NC2=C(S)C=CC=C2S1 JDICEKWSLNPYSN-UHFFFAOYSA-N 0.000 description 1
- MNWUYMNVJNYHLO-UHFFFAOYSA-N 2-(2,5-dihydroxyphenyl)acetic acid Chemical compound OC(=O)CC1=CC(O)=CC=C1O.OC(=O)CC1=CC(O)=CC=C1O MNWUYMNVJNYHLO-UHFFFAOYSA-N 0.000 description 1
- DSIWIQXUFAAPJH-UHFFFAOYSA-N 2-(carboxymethyl)benzoic acid Chemical compound OC(=O)CC1=CC=CC=C1C(O)=O.OC(=O)CC1=CC=CC=C1C(O)=O DSIWIQXUFAAPJH-UHFFFAOYSA-N 0.000 description 1
- RDKPBLFKPVFDOJ-UHFFFAOYSA-N 2-[(2,4,6-tricarbamoylphenyl)disulfanyl]benzene-1,3,5-tricarboxamide Chemical group NC(=O)C1=CC(C(=O)N)=CC(C(N)=O)=C1SSC1=C(C(N)=O)C=C(C(N)=O)C=C1C(N)=O RDKPBLFKPVFDOJ-UHFFFAOYSA-N 0.000 description 1
- FHUGKCNOKBVBFA-UHFFFAOYSA-N 2-[(2,4,6-tricarbonochloridoylphenyl)disulfanyl]benzene-1,3,5-tricarbonyl chloride Chemical group ClC(=O)C1=CC(C(=O)Cl)=CC(C(Cl)=O)=C1SSC1=C(C(Cl)=O)C=C(C(Cl)=O)C=C1C(Cl)=O FHUGKCNOKBVBFA-UHFFFAOYSA-N 0.000 description 1
- FSYWNRKDQWZRCD-UHFFFAOYSA-N 2-[(2,4,6-tricarboxyphenyl)disulfanyl]benzene-1,3,5-tricarboxylic acid Chemical group OC(=O)C1=CC(C(=O)O)=CC(C(O)=O)=C1SSC1=C(C(O)=O)C=C(C(O)=O)C=C1C(O)=O FSYWNRKDQWZRCD-UHFFFAOYSA-N 0.000 description 1
- UARWHEMYCIUGHC-UHFFFAOYSA-N 2-[(2,4,6-tricyanophenyl)disulfanyl]benzene-1,3,5-tricarbonitrile Chemical compound N#CC1=CC(C#N)=CC(C#N)=C1SSC1=C(C#N)C=C(C#N)C=C1C#N UARWHEMYCIUGHC-UHFFFAOYSA-N 0.000 description 1
- RZQYEMLMOMOHOG-UHFFFAOYSA-N 2-[(2,4,6-triformylphenyl)disulfanyl]benzene-1,3,5-tricarbaldehyde Chemical group O=CC1=CC(C=O)=CC(C=O)=C1SSC1=C(C=O)C=C(C=O)C=C1C=O RZQYEMLMOMOHOG-UHFFFAOYSA-N 0.000 description 1
- CJYIJSRAOKBNNZ-UHFFFAOYSA-N 2-[(2,4,6-trisulfinophenyl)disulfanyl]benzene-1,3,5-trisulfinic acid Chemical group OS(=O)C1=CC(S(=O)O)=CC(S(O)=O)=C1SSC1=C(S(O)=O)C=C(S(O)=O)C=C1S(O)=O CJYIJSRAOKBNNZ-UHFFFAOYSA-N 0.000 description 1
- FZZWFLLDFYYGEK-UHFFFAOYSA-N 2-[(2,4,6-trisulfophenyl)disulfanyl]benzene-1,3,5-trisulfonic acid Chemical group OS(=O)(=O)C1=CC(S(=O)(=O)O)=CC(S(O)(=O)=O)=C1SSC1=C(S(O)(=O)=O)C=C(S(O)(=O)=O)C=C1S(O)(=O)=O FZZWFLLDFYYGEK-UHFFFAOYSA-N 0.000 description 1
- QFBSAAGMHNOCOB-UHFFFAOYSA-N 2-[(2,5-difluorophenyl)disulfanyl]-1,4-difluorobenzene Chemical group FC1=CC=C(F)C(SSC=2C(=CC=C(F)C=2)F)=C1 QFBSAAGMHNOCOB-UHFFFAOYSA-N 0.000 description 1
- XOVRAXDVGGXDAN-UHFFFAOYSA-N 2-[(2,5-diiodophenyl)disulfanyl]-1,4-diiodobenzene Chemical group IC1=CC=C(I)C(SSC=2C(=CC=C(I)C=2)I)=C1 XOVRAXDVGGXDAN-UHFFFAOYSA-N 0.000 description 1
- XMIKJNSWZZMXKK-UHFFFAOYSA-N 2-[(2-amino-4-chlorophenyl)disulfanyl]-5-chloroaniline Chemical compound NC1=CC(Cl)=CC=C1SSC1=CC=C(Cl)C=C1N XMIKJNSWZZMXKK-UHFFFAOYSA-N 0.000 description 1
- ZOKDXRSDLYAUQB-UHFFFAOYSA-N 2-[(2-amino-4-methoxyphenyl)disulfanyl]-5-methoxyaniline Chemical compound NC1=CC(OC)=CC=C1SSC1=CC=C(OC)C=C1N ZOKDXRSDLYAUQB-UHFFFAOYSA-N 0.000 description 1
- WLVKUHMLYPMLJY-UHFFFAOYSA-N 2-[(2-amino-4-methylphenyl)disulfanyl]-5-methylaniline Chemical compound NC1=CC(C)=CC=C1SSC1=CC=C(C)C=C1N WLVKUHMLYPMLJY-UHFFFAOYSA-N 0.000 description 1
- GJRHTEVHZPTSGJ-UHFFFAOYSA-N 2-[(2-amino-4-methylsulfinylphenyl)disulfanyl]-5-methylsulfinylaniline Chemical compound NC1=CC(S(=O)C)=CC=C1SSC1=CC=C(S(C)=O)C=C1N GJRHTEVHZPTSGJ-UHFFFAOYSA-N 0.000 description 1
- OVCBBKKFDUAATR-UHFFFAOYSA-N 2-[(2-hydroxy-4-methoxyphenyl)disulfanyl]-5-methoxyphenol Chemical compound OC1=CC(OC)=CC=C1SSC1=CC=C(OC)C=C1O OVCBBKKFDUAATR-UHFFFAOYSA-N 0.000 description 1
- VJSGBVNTNBMOJT-UHFFFAOYSA-N 2-[(2-hydroxy-4-methylphenyl)disulfanyl]-5-methylphenol Chemical compound OC1=CC(C)=CC=C1SSC1=CC=C(C)C=C1O VJSGBVNTNBMOJT-UHFFFAOYSA-N 0.000 description 1
- XKPGGVAJIYNCOO-UHFFFAOYSA-N 2-[(2-hydroxy-4-methylsulfinylphenyl)disulfanyl]-5-methylsulfinylphenol Chemical compound OC1=CC(S(=O)C)=CC=C1SSC1=CC=C(S(C)=O)C=C1O XKPGGVAJIYNCOO-UHFFFAOYSA-N 0.000 description 1
- XFDCIPJRGGPGDG-UHFFFAOYSA-N 2-[[2,4,6-tris(chlorosulfonyl)phenyl]disulfanyl]benzene-1,3,5-trisulfonyl chloride Chemical group ClS(=O)(=O)C1=CC(S(=O)(=O)Cl)=CC(S(Cl)(=O)=O)=C1SSC1=C(S(Cl)(=O)=O)C=C(S(Cl)(=O)=O)C=C1S(Cl)(=O)=O XFDCIPJRGGPGDG-UHFFFAOYSA-N 0.000 description 1
- GVPHVJIQRJPWGH-UHFFFAOYSA-N 2-[[2-amino-4-(trichloromethyl)phenyl]disulfanyl]-5-(trichloromethyl)aniline Chemical compound NC1=CC(C(Cl)(Cl)Cl)=CC=C1SSC1=CC=C(C(Cl)(Cl)Cl)C=C1N GVPHVJIQRJPWGH-UHFFFAOYSA-N 0.000 description 1
- NGOCKDUQBTZLEU-UHFFFAOYSA-N 2-[[2-hydroxy-4-(trichloromethyl)phenyl]disulfanyl]-5-(trichloromethyl)phenol Chemical compound OC1=CC(C(Cl)(Cl)Cl)=CC=C1SSC1=CC=C(C(Cl)(Cl)Cl)C=C1O NGOCKDUQBTZLEU-UHFFFAOYSA-N 0.000 description 1
- PTHLFBHWHFSRSL-UHFFFAOYSA-N 2-amino-4-(trichloromethyl)benzenethiol Chemical compound NC1=CC(C(Cl)(Cl)Cl)=CC=C1S PTHLFBHWHFSRSL-UHFFFAOYSA-N 0.000 description 1
- JFNKEWGSSKQRKL-UHFFFAOYSA-N 2-amino-4-(trichloromethyl)benzenethiol;sodium Chemical compound [Na].NC1=CC(C(Cl)(Cl)Cl)=CC=C1S JFNKEWGSSKQRKL-UHFFFAOYSA-N 0.000 description 1
- QYYDIDVILCOOHB-UHFFFAOYSA-N 2-amino-4-(trichloromethyl)benzenethiol;zinc Chemical compound [Zn].NC1=CC(C(Cl)(Cl)Cl)=CC=C1S QYYDIDVILCOOHB-UHFFFAOYSA-N 0.000 description 1
- NGIRMPARLVGMPX-UHFFFAOYSA-N 2-amino-4-chlorobenzenethiol Chemical compound NC1=CC(Cl)=CC=C1S NGIRMPARLVGMPX-UHFFFAOYSA-N 0.000 description 1
- ITWZHYKEXDWMTD-UHFFFAOYSA-N 2-amino-4-chlorobenzenethiol;sodium Chemical compound [Na].NC1=CC(Cl)=CC=C1S ITWZHYKEXDWMTD-UHFFFAOYSA-N 0.000 description 1
- LEHHDHAIMYASQW-UHFFFAOYSA-N 2-amino-4-chlorobenzenethiol;zinc Chemical compound [Zn].NC1=CC(Cl)=CC=C1S LEHHDHAIMYASQW-UHFFFAOYSA-N 0.000 description 1
- KIDLBEYNOGVICH-UHFFFAOYSA-N 2-amino-4-methoxybenzenethiol Chemical compound COC1=CC=C(S)C(N)=C1 KIDLBEYNOGVICH-UHFFFAOYSA-N 0.000 description 1
- VNZDOSYDQXIZDV-UHFFFAOYSA-N 2-amino-4-methoxybenzenethiol;sodium Chemical compound [Na].COC1=CC=C(S)C(N)=C1 VNZDOSYDQXIZDV-UHFFFAOYSA-N 0.000 description 1
- GNBGXZLSTOMZDN-UHFFFAOYSA-N 2-amino-4-methoxybenzenethiol;zinc Chemical compound [Zn].COC1=CC=C(S)C(N)=C1 GNBGXZLSTOMZDN-UHFFFAOYSA-N 0.000 description 1
- QCLMTLDABHUUBC-UHFFFAOYSA-N 2-amino-4-methylbenzenethiol Chemical compound CC1=CC=C(S)C(N)=C1 QCLMTLDABHUUBC-UHFFFAOYSA-N 0.000 description 1
- UTUZNVFVQSWEHC-UHFFFAOYSA-N 2-amino-4-methylbenzenethiol;sodium Chemical compound [Na].CC1=CC=C(S)C(N)=C1 UTUZNVFVQSWEHC-UHFFFAOYSA-N 0.000 description 1
- YVONXSNDLPDWPM-UHFFFAOYSA-N 2-amino-4-methylbenzenethiol;zinc Chemical compound [Zn].CC1=CC=C(S)C(N)=C1 YVONXSNDLPDWPM-UHFFFAOYSA-N 0.000 description 1
- YEHGEZRBMZQMEK-UHFFFAOYSA-N 2-amino-4-methylsulfinylbenzenethiol Chemical compound CS(=O)C1=CC=C(S)C(N)=C1 YEHGEZRBMZQMEK-UHFFFAOYSA-N 0.000 description 1
- ZEOGSCSFTKFXMR-UHFFFAOYSA-N 2-amino-4-methylsulfinylbenzenethiol;sodium Chemical compound [Na].CS(=O)C1=CC=C(S)C(N)=C1 ZEOGSCSFTKFXMR-UHFFFAOYSA-N 0.000 description 1
- RWLJNBQNKXVFTE-UHFFFAOYSA-N 2-amino-4-methylsulfinylbenzenethiol;zinc Chemical compound [Zn].CS(=O)C1=CC=C(S)C(N)=C1 RWLJNBQNKXVFTE-UHFFFAOYSA-N 0.000 description 1
- PWOBDMNCYMQTCE-UHFFFAOYSA-N 2-chlorobenzenethiol Chemical compound SC1=CC=CC=C1Cl PWOBDMNCYMQTCE-UHFFFAOYSA-N 0.000 description 1
- JXCYBKCGNUBYPW-UHFFFAOYSA-N 2-hydroxy-2,2-diphenylacetic acid Chemical compound C=1C=CC=CC=1C(O)(C(=O)O)C1=CC=CC=C1.C=1C=CC=CC=1C(O)(C(=O)O)C1=CC=CC=C1 JXCYBKCGNUBYPW-UHFFFAOYSA-N 0.000 description 1
- XWMRMJOORZIBKJ-UHFFFAOYSA-N 2-hydroxy-3-methylbenzoic acid Chemical compound CC1=CC=CC(C(O)=O)=C1O.CC1=CC=CC(C(O)=O)=C1O XWMRMJOORZIBKJ-UHFFFAOYSA-N 0.000 description 1
- UXMQLRNDJOZAPS-UHFFFAOYSA-N 2-hydroxy-5-methylbenzoic acid Chemical compound CC1=CC=C(O)C(C(O)=O)=C1.CC1=CC=C(O)C(C(O)=O)=C1 UXMQLRNDJOZAPS-UHFFFAOYSA-N 0.000 description 1
- VHBSECWYEFJRNV-UHFFFAOYSA-N 2-hydroxybenzoic acid Chemical compound OC(=O)C1=CC=CC=C1O.OC(=O)C1=CC=CC=C1O VHBSECWYEFJRNV-UHFFFAOYSA-N 0.000 description 1
- FVPAXFPIVPPMKJ-UHFFFAOYSA-N 2-methylsulfinylbenzenethiol Chemical compound CS(=O)C1=CC=CC=C1S FVPAXFPIVPPMKJ-UHFFFAOYSA-N 0.000 description 1
- UIZSCIWRUISTDZ-UHFFFAOYSA-N 2-nitro-1-[[2-nitro-4-(trichloromethyl)phenyl]disulfanyl]-4-(trichloromethyl)benzene Chemical compound [O-][N+](=O)C1=CC(C(Cl)(Cl)Cl)=CC=C1SSC1=CC=C(C(Cl)(Cl)Cl)C=C1[N+]([O-])=O UIZSCIWRUISTDZ-UHFFFAOYSA-N 0.000 description 1
- WUZOBYBVVNVFEQ-UHFFFAOYSA-N 2-nitro-4-(trichloromethyl)benzenethiol Chemical compound [O-][N+](=O)C1=CC(C(Cl)(Cl)Cl)=CC=C1S WUZOBYBVVNVFEQ-UHFFFAOYSA-N 0.000 description 1
- IRPBMMFOWMMGCD-UHFFFAOYSA-N 2-nitro-4-(trichloromethyl)benzenethiol;sodium Chemical compound [Na].[O-][N+](=O)C1=CC(C(Cl)(Cl)Cl)=CC=C1S IRPBMMFOWMMGCD-UHFFFAOYSA-N 0.000 description 1
- YIOZRGDQSNQASD-UHFFFAOYSA-N 2-nitro-4-(trichloromethyl)benzenethiol;zinc Chemical compound [Zn].[O-][N+](=O)C1=CC(C(Cl)(Cl)Cl)=CC=C1S YIOZRGDQSNQASD-UHFFFAOYSA-N 0.000 description 1
- XTILJCALGBRMPR-UHFFFAOYSA-N 2-phenylacetic acid Chemical compound OC(=O)CC1=CC=CC=C1.OC(=O)CC1=CC=CC=C1 XTILJCALGBRMPR-UHFFFAOYSA-N 0.000 description 1
- CMPJKTQBBPRKQU-UHFFFAOYSA-N 2-phenylsulfanyl-1-[[2-phenylsulfanyl-4-(trichloromethyl)phenyl]disulfanyl]-4-(trichloromethyl)benzene Chemical compound C=1C=CC=CC=1SC1=CC(C(Cl)(Cl)Cl)=CC=C1SSC1=CC=C(C(Cl)(Cl)Cl)C=C1SC1=CC=CC=C1 CMPJKTQBBPRKQU-UHFFFAOYSA-N 0.000 description 1
- YPISMTSXYJJJRZ-UHFFFAOYSA-N 2-phenylsulfanyl-4-(trichloromethyl)benzenethiol Chemical compound SC1=CC=C(C(Cl)(Cl)Cl)C=C1SC1=CC=CC=C1 YPISMTSXYJJJRZ-UHFFFAOYSA-N 0.000 description 1
- NMWCJPSJJREKKF-UHFFFAOYSA-N 2-phenylsulfanyl-4-(trichloromethyl)benzenethiol;sodium Chemical compound [Na].SC1=CC=C(C(Cl)(Cl)Cl)C=C1SC1=CC=CC=C1 NMWCJPSJJREKKF-UHFFFAOYSA-N 0.000 description 1
- AEXDULLISCUIGG-UHFFFAOYSA-N 2-phenylsulfanyl-4-(trichloromethyl)benzenethiol;zinc Chemical compound [Zn].SC1=CC=C(C(Cl)(Cl)Cl)C=C1SC1=CC=CC=C1 AEXDULLISCUIGG-UHFFFAOYSA-N 0.000 description 1
- DVTINHHRFGEPEX-UHFFFAOYSA-N 2-sulfanyl-5-(trichloromethyl)phenol Chemical compound OC1=CC(C(Cl)(Cl)Cl)=CC=C1S DVTINHHRFGEPEX-UHFFFAOYSA-N 0.000 description 1
- HZOYCNPHFYNTFX-UHFFFAOYSA-N 2-sulfanyl-5-(trichloromethyl)phenol;zinc Chemical compound [Zn].OC1=CC(C(Cl)(Cl)Cl)=CC=C1S HZOYCNPHFYNTFX-UHFFFAOYSA-N 0.000 description 1
- BIBPNOKWKYKSQV-UHFFFAOYSA-N 2-sulfanylbenzene-1,3,5-tricarbaldehyde Chemical compound SC1=C(C=O)C=C(C=O)C=C1C=O BIBPNOKWKYKSQV-UHFFFAOYSA-N 0.000 description 1
- BGLLRSPCZGFXJT-UHFFFAOYSA-N 2-sulfanylbenzene-1,3,5-tricarbaldehyde;zinc Chemical compound [Zn].SC1=C(C=O)C=C(C=O)C=C1C=O BGLLRSPCZGFXJT-UHFFFAOYSA-N 0.000 description 1
- WPCCQPZWKCHMGT-UHFFFAOYSA-N 2-sulfanylbenzene-1,3,5-tricarbonitrile Chemical compound SC1=C(C#N)C=C(C#N)C=C1C#N WPCCQPZWKCHMGT-UHFFFAOYSA-N 0.000 description 1
- YYTSQCMDIVZAPV-UHFFFAOYSA-N 2-sulfanylbenzene-1,3,5-tricarbonitrile;zinc Chemical compound [Zn].SC1=C(C#N)C=C(C#N)C=C1C#N YYTSQCMDIVZAPV-UHFFFAOYSA-N 0.000 description 1
- VYYSYFCXFXGTAU-UHFFFAOYSA-N 2-sulfanylbenzene-1,3,5-tricarbonyl chloride Chemical compound SC1=C(C(Cl)=O)C=C(C(Cl)=O)C=C1C(Cl)=O VYYSYFCXFXGTAU-UHFFFAOYSA-N 0.000 description 1
- SZCDYSXBARLVEU-UHFFFAOYSA-N 2-sulfanylbenzene-1,3,5-tricarbonyl chloride;zinc Chemical compound [Zn].SC1=C(C(Cl)=O)C=C(C(Cl)=O)C=C1C(Cl)=O SZCDYSXBARLVEU-UHFFFAOYSA-N 0.000 description 1
- LPLCFMXJTOYGCQ-UHFFFAOYSA-N 2-sulfanylbenzene-1,3,5-tricarboxamide Chemical compound NC(=O)C1=CC(C(N)=O)=C(S)C(C(N)=O)=C1 LPLCFMXJTOYGCQ-UHFFFAOYSA-N 0.000 description 1
- PUPWQFPUPDJOOK-UHFFFAOYSA-N 2-sulfanylbenzene-1,3,5-tricarboxamide;zinc Chemical compound [Zn].NC(=O)C1=CC(C(N)=O)=C(S)C(C(N)=O)=C1 PUPWQFPUPDJOOK-UHFFFAOYSA-N 0.000 description 1
- UQGUJROUFSTNPR-UHFFFAOYSA-N 2-sulfanylbenzene-1,3,5-tricarboxylic acid Chemical compound OC(=O)C1=CC(C(O)=O)=C(S)C(C(O)=O)=C1 UQGUJROUFSTNPR-UHFFFAOYSA-N 0.000 description 1
- FSQXGSFHMFMESC-UHFFFAOYSA-N 2-sulfanylbenzene-1,3,5-trisulfinic acid Chemical compound OS(=O)C1=CC(S(O)=O)=C(S)C(S(O)=O)=C1 FSQXGSFHMFMESC-UHFFFAOYSA-N 0.000 description 1
- UZZPUPKFLJNETC-UHFFFAOYSA-N 2-sulfanylbenzene-1,3,5-trisulfonic acid Chemical compound OS(=O)(=O)C1=CC(S(O)(=O)=O)=C(S)C(S(O)(=O)=O)=C1 UZZPUPKFLJNETC-UHFFFAOYSA-N 0.000 description 1
- UVPKAKRKIXBABC-UHFFFAOYSA-N 2-sulfanylbenzene-1,3,5-trisulfonyl chloride Chemical compound SC1=C(S(Cl)(=O)=O)C=C(S(Cl)(=O)=O)C=C1S(Cl)(=O)=O UVPKAKRKIXBABC-UHFFFAOYSA-N 0.000 description 1
- XDZOHXFGSUIYHP-UHFFFAOYSA-N 2-sulfanylbenzene-1,3,5-trisulfonyl chloride;zinc Chemical compound [Zn].SC1=C(S(Cl)(=O)=O)C=C(S(Cl)(=O)=O)C=C1S(Cl)(=O)=O XDZOHXFGSUIYHP-UHFFFAOYSA-N 0.000 description 1
- UDPPDGRNIXGNQJ-UHFFFAOYSA-N 2-sulfanylbenzenesulfonyl chloride Chemical compound SC1=CC=CC=C1S(Cl)(=O)=O UDPPDGRNIXGNQJ-UHFFFAOYSA-N 0.000 description 1
- UYEMGAFJOZZIFP-UHFFFAOYSA-N 3,5-dihydroxybenzoic acid Chemical compound OC(=O)C1=CC(O)=CC(O)=C1 UYEMGAFJOZZIFP-UHFFFAOYSA-N 0.000 description 1
- FCRAWHRICODQTR-UHFFFAOYSA-N 3-(3,4-dihydroxyphenyl)propanoic acid Chemical compound OC(=O)CCC1=CC=C(O)C(O)=C1.OC(=O)CCC1=CC=C(O)C(O)=C1 FCRAWHRICODQTR-UHFFFAOYSA-N 0.000 description 1
- VKEFJODALIKKMK-UHFFFAOYSA-N 3-(3-hydroxy-4-methoxyphenyl)propanoic acid Chemical compound OC=1C=C(C=CC1OC)CCC(=O)O.C(CCC1=CC(O)=C(OC)C=C1)(=O)O VKEFJODALIKKMK-UHFFFAOYSA-N 0.000 description 1
- WAOUCKBSGSTKQV-UHFFFAOYSA-N 3-(4-hydroxy-3-methoxyphenyl)propanoic acid Chemical compound OC1=C(C=C(C=C1)CCC(=O)O)OC.C(CCC1=CC(OC)=C(O)C=C1)(=O)O WAOUCKBSGSTKQV-UHFFFAOYSA-N 0.000 description 1
- CPGFMWPQXUXQRX-UHFFFAOYSA-N 3-amino-3-(4-fluorophenyl)propanoic acid Chemical compound OC(=O)CC(N)C1=CC=C(F)C=C1 CPGFMWPQXUXQRX-UHFFFAOYSA-N 0.000 description 1
- SWBGWQJLGDMGNS-UHFFFAOYSA-N 3-amino-4-[(2-amino-4-carbamoylphenyl)disulfanyl]benzamide Chemical compound NC1=CC(C(=O)N)=CC=C1SSC1=CC=C(C(N)=O)C=C1N SWBGWQJLGDMGNS-UHFFFAOYSA-N 0.000 description 1
- KDZIQGNNKQMBOR-UHFFFAOYSA-N 3-amino-4-[(2-amino-4-carbonochloridoylphenyl)disulfanyl]benzoyl chloride Chemical compound NC1=CC(C(Cl)=O)=CC=C1SSC1=CC=C(C(Cl)=O)C=C1N KDZIQGNNKQMBOR-UHFFFAOYSA-N 0.000 description 1
- KQFIYYRMDLHHIH-UHFFFAOYSA-N 3-amino-4-[(2-amino-4-carboxyphenyl)disulfanyl]benzoic acid Chemical compound NC1=CC(C(O)=O)=CC=C1SSC1=CC=C(C(O)=O)C=C1N KQFIYYRMDLHHIH-UHFFFAOYSA-N 0.000 description 1
- LBCYCXWFJDXTEL-UHFFFAOYSA-N 3-amino-4-[(2-amino-4-chlorosulfonylphenyl)disulfanyl]benzenesulfonyl chloride Chemical compound NC1=CC(S(Cl)(=O)=O)=CC=C1SSC1=CC=C(S(Cl)(=O)=O)C=C1N LBCYCXWFJDXTEL-UHFFFAOYSA-N 0.000 description 1
- LGWXSDIGTQAFON-UHFFFAOYSA-N 3-amino-4-[(2-amino-4-cyanophenyl)disulfanyl]benzonitrile Chemical compound NC1=CC(C#N)=CC=C1SSC1=CC=C(C#N)C=C1N LGWXSDIGTQAFON-UHFFFAOYSA-N 0.000 description 1
- AQGAGZJBXFEQCP-UHFFFAOYSA-N 3-amino-4-[(2-amino-4-formylphenyl)disulfanyl]benzaldehyde Chemical compound NC1=CC(C=O)=CC=C1SSC1=CC=C(C=O)C=C1N AQGAGZJBXFEQCP-UHFFFAOYSA-N 0.000 description 1
- FKWRMMIWTDDSRQ-UHFFFAOYSA-N 3-amino-4-[(2-amino-4-sulfinophenyl)disulfanyl]benzenesulfinic acid Chemical compound NC1=CC(S(O)=O)=CC=C1SSC1=CC=C(S(O)=O)C=C1N FKWRMMIWTDDSRQ-UHFFFAOYSA-N 0.000 description 1
- FIXIBUUHTYXLJD-UHFFFAOYSA-N 3-amino-4-[(2-amino-4-sulfophenyl)disulfanyl]benzenesulfonic acid Chemical compound NC1=CC(S(O)(=O)=O)=CC=C1SSC1=CC=C(S(O)(=O)=O)C=C1N FIXIBUUHTYXLJD-UHFFFAOYSA-N 0.000 description 1
- HDDSZPGQRRVXCR-UHFFFAOYSA-N 3-amino-4-sulfanylbenzaldehyde Chemical compound NC1=CC(C=O)=CC=C1S HDDSZPGQRRVXCR-UHFFFAOYSA-N 0.000 description 1
- YEJFGXYHIYUTSI-UHFFFAOYSA-N 3-amino-4-sulfanylbenzaldehyde;sodium Chemical compound [Na].NC1=CC(C=O)=CC=C1S YEJFGXYHIYUTSI-UHFFFAOYSA-N 0.000 description 1
- NYJFFRQWWFLVBZ-UHFFFAOYSA-N 3-amino-4-sulfanylbenzaldehyde;zinc Chemical compound [Zn].NC1=CC(C=O)=CC=C1S NYJFFRQWWFLVBZ-UHFFFAOYSA-N 0.000 description 1
- FWNAHKCMVDJMAT-UHFFFAOYSA-N 3-amino-4-sulfanylbenzamide Chemical compound NC(=O)C1=CC=C(S)C(N)=C1 FWNAHKCMVDJMAT-UHFFFAOYSA-N 0.000 description 1
- XDNRBFPVFYHOJM-UHFFFAOYSA-N 3-amino-4-sulfanylbenzamide;sodium Chemical compound [Na].NC(=O)C1=CC=C(S)C(N)=C1 XDNRBFPVFYHOJM-UHFFFAOYSA-N 0.000 description 1
- LQUKLKYVJPGLMV-UHFFFAOYSA-N 3-amino-4-sulfanylbenzamide;zinc Chemical compound [Zn].NC(=O)C1=CC=C(S)C(N)=C1 LQUKLKYVJPGLMV-UHFFFAOYSA-N 0.000 description 1
- FVQFGHRZEWCWLL-UHFFFAOYSA-N 3-amino-4-sulfanylbenzenesulfinic acid Chemical compound NC1=CC(S(O)=O)=CC=C1S FVQFGHRZEWCWLL-UHFFFAOYSA-N 0.000 description 1
- OQOSNFWADUEBEP-UHFFFAOYSA-N 3-amino-4-sulfanylbenzenesulfonic acid Chemical compound NC1=CC(S(O)(=O)=O)=CC=C1S OQOSNFWADUEBEP-UHFFFAOYSA-N 0.000 description 1
- HZIOYKXBNRJZGQ-UHFFFAOYSA-N 3-amino-4-sulfanylbenzenesulfonyl chloride Chemical compound NC1=CC(S(Cl)(=O)=O)=CC=C1S HZIOYKXBNRJZGQ-UHFFFAOYSA-N 0.000 description 1
- MJCVCKHKLCFEMB-UHFFFAOYSA-N 3-amino-4-sulfanylbenzenesulfonyl chloride;sodium Chemical compound [Na].NC1=CC(S(Cl)(=O)=O)=CC=C1S MJCVCKHKLCFEMB-UHFFFAOYSA-N 0.000 description 1
- NTNQYHVTPYCFFU-UHFFFAOYSA-N 3-amino-4-sulfanylbenzenesulfonyl chloride;zinc Chemical compound [Zn].NC1=CC(S(Cl)(=O)=O)=CC=C1S NTNQYHVTPYCFFU-UHFFFAOYSA-N 0.000 description 1
- XTYZKVGSYFKJAR-UHFFFAOYSA-N 3-amino-4-sulfanylbenzoic acid Chemical compound NC1=CC(C(O)=O)=CC=C1S XTYZKVGSYFKJAR-UHFFFAOYSA-N 0.000 description 1
- CJEDABGJILXBLL-UHFFFAOYSA-N 3-amino-4-sulfanylbenzonitrile Chemical compound NC1=CC(C#N)=CC=C1S CJEDABGJILXBLL-UHFFFAOYSA-N 0.000 description 1
- CNUALXKMVDWSDY-UHFFFAOYSA-N 3-amino-4-sulfanylbenzonitrile;sodium Chemical compound [Na].NC1=CC(C#N)=CC=C1S CNUALXKMVDWSDY-UHFFFAOYSA-N 0.000 description 1
- LAAUDZYSQQSYOI-UHFFFAOYSA-N 3-amino-4-sulfanylbenzonitrile;zinc Chemical compound [Zn].NC1=CC(C#N)=CC=C1S LAAUDZYSQQSYOI-UHFFFAOYSA-N 0.000 description 1
- ABIWECFOEZXCAR-UHFFFAOYSA-N 3-amino-4-sulfanylbenzoyl chloride Chemical compound NC1=CC(C(Cl)=O)=CC=C1S ABIWECFOEZXCAR-UHFFFAOYSA-N 0.000 description 1
- BSCOWJGPZQKUKT-UHFFFAOYSA-N 3-amino-4-sulfanylbenzoyl chloride;sodium Chemical compound [Na].NC1=CC(C(Cl)=O)=CC=C1S BSCOWJGPZQKUKT-UHFFFAOYSA-N 0.000 description 1
- ZFUQAVYVEWWGIU-UHFFFAOYSA-N 3-amino-4-sulfanylbenzoyl chloride;zinc Chemical compound [Zn].NC1=CC(C(Cl)=O)=CC=C1S ZFUQAVYVEWWGIU-UHFFFAOYSA-N 0.000 description 1
- BUZICZZQJDLXJN-UHFFFAOYSA-N 3-azaniumyl-4-hydroxybutanoate Chemical compound OCC(N)CC(O)=O BUZICZZQJDLXJN-UHFFFAOYSA-N 0.000 description 1
- RIERSGULWXEJKL-UHFFFAOYSA-N 3-hydroxy-2-methylbenzoic acid Chemical compound CC1=C(O)C=CC=C1C(O)=O RIERSGULWXEJKL-UHFFFAOYSA-N 0.000 description 1
- OKRMDBDWOXCVDY-UHFFFAOYSA-N 3-hydroxy-4-[(2-hydroxy-4-sulfinophenyl)disulfanyl]benzenesulfinic acid Chemical compound OC1=CC(S(O)=O)=CC=C1SSC1=CC=C(S(O)=O)C=C1O OKRMDBDWOXCVDY-UHFFFAOYSA-N 0.000 description 1
- LHXDVHGACNZQGV-UHFFFAOYSA-N 3-hydroxy-4-[(2-hydroxy-4-sulfophenyl)disulfanyl]benzenesulfonic acid Chemical compound OC1=CC(S(O)(=O)=O)=CC=C1SSC1=CC=C(S(O)(=O)=O)C=C1O LHXDVHGACNZQGV-UHFFFAOYSA-N 0.000 description 1
- QYQUCKSOUOFGJL-UHFFFAOYSA-N 3-hydroxy-4-sulfanylbenzaldehyde Chemical compound OC1=CC(C=O)=CC=C1S QYQUCKSOUOFGJL-UHFFFAOYSA-N 0.000 description 1
- CUCSNSMDWGWQMQ-UHFFFAOYSA-N 3-hydroxy-4-sulfanylbenzaldehyde;sodium Chemical compound [Na].OC1=CC(C=O)=CC=C1S CUCSNSMDWGWQMQ-UHFFFAOYSA-N 0.000 description 1
- BVBAJDXFTXLGMP-UHFFFAOYSA-N 3-hydroxy-4-sulfanylbenzaldehyde;zinc Chemical compound [Zn].OC1=CC(C=O)=CC=C1S BVBAJDXFTXLGMP-UHFFFAOYSA-N 0.000 description 1
- BEQAARAEDCCWFG-UHFFFAOYSA-N 3-hydroxy-4-sulfanylbenzamide Chemical compound NC(=O)C1=CC=C(S)C(O)=C1 BEQAARAEDCCWFG-UHFFFAOYSA-N 0.000 description 1
- LNURRAUFEWYJCB-UHFFFAOYSA-N 3-hydroxy-4-sulfanylbenzamide;sodium Chemical compound [Na].NC(=O)C1=CC=C(S)C(O)=C1 LNURRAUFEWYJCB-UHFFFAOYSA-N 0.000 description 1
- ZCGVJZKEUZRNHW-UHFFFAOYSA-N 3-hydroxy-4-sulfanylbenzamide;zinc Chemical compound [Zn].NC(=O)C1=CC=C(S)C(O)=C1 ZCGVJZKEUZRNHW-UHFFFAOYSA-N 0.000 description 1
- PGCQRZAWBVIWDS-UHFFFAOYSA-N 3-hydroxy-4-sulfanylbenzenesulfinic acid Chemical compound OC1=CC(S(O)=O)=CC=C1S PGCQRZAWBVIWDS-UHFFFAOYSA-N 0.000 description 1
- MDJIJICPGQLRCY-UHFFFAOYSA-N 3-hydroxy-4-sulfanylbenzenesulfonic acid Chemical compound OC1=CC(S(O)(=O)=O)=CC=C1S MDJIJICPGQLRCY-UHFFFAOYSA-N 0.000 description 1
- AULRIRBTQBRAOO-UHFFFAOYSA-N 3-hydroxy-4-sulfanylbenzenesulfonyl chloride Chemical compound OC1=CC(S(Cl)(=O)=O)=CC=C1S AULRIRBTQBRAOO-UHFFFAOYSA-N 0.000 description 1
- YMYBRXLMFPICQM-UHFFFAOYSA-N 3-hydroxy-4-sulfanylbenzenesulfonyl chloride;sodium Chemical compound [Na].OC1=CC(S(Cl)(=O)=O)=CC=C1S YMYBRXLMFPICQM-UHFFFAOYSA-N 0.000 description 1
- QZNQOPLETRORDZ-UHFFFAOYSA-N 3-hydroxy-4-sulfanylbenzenesulfonyl chloride;zinc Chemical compound [Zn].OC1=CC(S(Cl)(=O)=O)=CC=C1S QZNQOPLETRORDZ-UHFFFAOYSA-N 0.000 description 1
- SCGLKJCYQVSCHS-UHFFFAOYSA-N 3-hydroxy-4-sulfanylbenzoic acid Chemical compound OC(=O)C1=CC=C(S)C(O)=C1 SCGLKJCYQVSCHS-UHFFFAOYSA-N 0.000 description 1
- MDRQHDWOVPYQLN-UHFFFAOYSA-N 3-hydroxy-4-sulfanylbenzonitrile Chemical compound OC1=CC(C#N)=CC=C1S MDRQHDWOVPYQLN-UHFFFAOYSA-N 0.000 description 1
- GSTNNHQSGIAZCN-UHFFFAOYSA-N 3-hydroxy-4-sulfanylbenzonitrile;sodium Chemical compound [Na].OC1=CC(C#N)=CC=C1S GSTNNHQSGIAZCN-UHFFFAOYSA-N 0.000 description 1
- DOKKYNWWSYJCCO-UHFFFAOYSA-N 3-hydroxy-4-sulfanylbenzonitrile;zinc Chemical compound [Zn].OC1=CC(C#N)=CC=C1S DOKKYNWWSYJCCO-UHFFFAOYSA-N 0.000 description 1
- VDHIATXBQMRYTF-UHFFFAOYSA-N 3-hydroxy-4-sulfanylbenzoyl chloride Chemical compound OC1=CC(C(Cl)=O)=CC=C1S VDHIATXBQMRYTF-UHFFFAOYSA-N 0.000 description 1
- NJTXCUXFBALYOH-UHFFFAOYSA-N 3-hydroxy-4-sulfanylbenzoyl chloride;sodium Chemical compound [Na].OC1=CC(C(Cl)=O)=CC=C1S NJTXCUXFBALYOH-UHFFFAOYSA-N 0.000 description 1
- LSIXUCVHQUUVIY-UHFFFAOYSA-N 3-hydroxy-4-sulfanylbenzoyl chloride;zinc Chemical compound [Zn].OC1=CC(C(Cl)=O)=CC=C1S LSIXUCVHQUUVIY-UHFFFAOYSA-N 0.000 description 1
- WHSXTWFYRGOBGO-UHFFFAOYSA-N 3-methylsalicylic acid Chemical compound CC1=CC=CC(C(O)=O)=C1O WHSXTWFYRGOBGO-UHFFFAOYSA-N 0.000 description 1
- NUNIMBIEGAAWBN-UHFFFAOYSA-N 3-nitro-4-[(2-nitro-4-sulfinophenyl)disulfanyl]benzenesulfinic acid Chemical compound [O-][N+](=O)C1=CC(S(=O)O)=CC=C1SSC1=CC=C(S(O)=O)C=C1[N+]([O-])=O NUNIMBIEGAAWBN-UHFFFAOYSA-N 0.000 description 1
- QHXOBEBKRZVVGO-UHFFFAOYSA-N 3-nitro-4-[(2-nitro-4-sulfophenyl)disulfanyl]benzenesulfonic acid Chemical compound [O-][N+](=O)C1=CC(S(=O)(=O)O)=CC=C1SSC1=CC=C(S(O)(=O)=O)C=C1[N+]([O-])=O QHXOBEBKRZVVGO-UHFFFAOYSA-N 0.000 description 1
- XPSQNYBBAPODJJ-UHFFFAOYSA-N 3-nitro-4-sulfanylbenzaldehyde Chemical compound [O-][N+](=O)C1=CC(C=O)=CC=C1S XPSQNYBBAPODJJ-UHFFFAOYSA-N 0.000 description 1
- JYKNYBALROHOHH-UHFFFAOYSA-N 3-nitro-4-sulfanylbenzaldehyde;sodium Chemical compound [Na].[O-][N+](=O)C1=CC(C=O)=CC=C1S JYKNYBALROHOHH-UHFFFAOYSA-N 0.000 description 1
- OORFRUGGPDUUKL-UHFFFAOYSA-N 3-nitro-4-sulfanylbenzaldehyde;zinc Chemical compound [Zn].[O-][N+](=O)C1=CC(C=O)=CC=C1S OORFRUGGPDUUKL-UHFFFAOYSA-N 0.000 description 1
- YUGQWTWLPCLHDA-UHFFFAOYSA-N 3-nitro-4-sulfanylbenzamide Chemical compound NC(=O)C1=CC=C(S)C([N+]([O-])=O)=C1 YUGQWTWLPCLHDA-UHFFFAOYSA-N 0.000 description 1
- LFEJNKWEFXGFIE-UHFFFAOYSA-N 3-nitro-4-sulfanylbenzamide;sodium Chemical compound [Na].NC(=O)C1=CC=C(S)C([N+]([O-])=O)=C1 LFEJNKWEFXGFIE-UHFFFAOYSA-N 0.000 description 1
- SBKLJURNUKSQFN-UHFFFAOYSA-N 3-nitro-4-sulfanylbenzamide;zinc Chemical compound [Zn].NC(=O)C1=CC=C(S)C([N+]([O-])=O)=C1 SBKLJURNUKSQFN-UHFFFAOYSA-N 0.000 description 1
- IJIALRQKXIIQHB-UHFFFAOYSA-N 3-nitro-4-sulfanylbenzenesulfinic acid Chemical compound OS(=O)C1=CC=C(S)C([N+]([O-])=O)=C1 IJIALRQKXIIQHB-UHFFFAOYSA-N 0.000 description 1
- QFRADYUMNINKDC-UHFFFAOYSA-N 3-nitro-4-sulfanylbenzenesulfonic acid Chemical compound OS(=O)(=O)C1=CC=C(S)C([N+]([O-])=O)=C1 QFRADYUMNINKDC-UHFFFAOYSA-N 0.000 description 1
- NQCXOCCOZAXFBU-UHFFFAOYSA-N 3-nitro-4-sulfanylbenzenesulfonyl chloride Chemical compound [O-][N+](=O)C1=CC(S(Cl)(=O)=O)=CC=C1S NQCXOCCOZAXFBU-UHFFFAOYSA-N 0.000 description 1
- FZOCMUDESVYLLT-UHFFFAOYSA-N 3-nitro-4-sulfanylbenzenesulfonyl chloride;sodium Chemical compound [Na].[O-][N+](=O)C1=CC(S(Cl)(=O)=O)=CC=C1S FZOCMUDESVYLLT-UHFFFAOYSA-N 0.000 description 1
- UCTGBEHQQHGDEX-UHFFFAOYSA-N 3-nitro-4-sulfanylbenzenesulfonyl chloride;zinc Chemical compound [Zn].[O-][N+](=O)C1=CC(S(Cl)(=O)=O)=CC=C1S UCTGBEHQQHGDEX-UHFFFAOYSA-N 0.000 description 1
- RLHGSIUQINRDSS-UHFFFAOYSA-N 3-nitro-4-sulfanylbenzoic acid Chemical compound OC(=O)C1=CC=C(S)C([N+]([O-])=O)=C1 RLHGSIUQINRDSS-UHFFFAOYSA-N 0.000 description 1
- IGZILJAEWSBUFW-UHFFFAOYSA-N 3-nitro-4-sulfanylbenzonitrile Chemical compound [O-][N+](=O)C1=CC(C#N)=CC=C1S IGZILJAEWSBUFW-UHFFFAOYSA-N 0.000 description 1
- SZAJNTQWTBADAP-UHFFFAOYSA-N 3-nitro-4-sulfanylbenzonitrile;sodium Chemical compound [Na].[O-][N+](=O)C1=CC(C#N)=CC=C1S SZAJNTQWTBADAP-UHFFFAOYSA-N 0.000 description 1
- XZXYCTLKDCPLPT-UHFFFAOYSA-N 3-nitro-4-sulfanylbenzonitrile;zinc Chemical compound [Zn].[O-][N+](=O)C1=CC(C#N)=CC=C1S XZXYCTLKDCPLPT-UHFFFAOYSA-N 0.000 description 1
- URDZPTVHVSPEMV-UHFFFAOYSA-N 3-nitro-4-sulfanylbenzoyl chloride Chemical compound [O-][N+](=O)C1=CC(C(Cl)=O)=CC=C1S URDZPTVHVSPEMV-UHFFFAOYSA-N 0.000 description 1
- RUWVOAITTBPTJU-UHFFFAOYSA-N 3-nitro-4-sulfanylbenzoyl chloride;sodium Chemical compound [Na].[O-][N+](=O)C1=CC(C(Cl)=O)=CC=C1S RUWVOAITTBPTJU-UHFFFAOYSA-N 0.000 description 1
- JGLAOSWTFVLJTG-UHFFFAOYSA-N 3-nitro-4-sulfanylbenzoyl chloride;zinc Chemical compound [Zn].[O-][N+](=O)C1=CC(C(Cl)=O)=CC=C1S JGLAOSWTFVLJTG-UHFFFAOYSA-N 0.000 description 1
- LZBBEECOKDJNLB-UHFFFAOYSA-N 3-phenylpropanoic acid Chemical compound OC(=O)CCC1=CC=CC=C1.OC(=O)CCC1=CC=CC=C1 LZBBEECOKDJNLB-UHFFFAOYSA-N 0.000 description 1
- UXCHAGJZXQWUCA-UHFFFAOYSA-N 3-phenylsulfanyl-4-[(2-phenylsulfanyl-4-sulfinophenyl)disulfanyl]benzenesulfinic acid Chemical compound C=1C=CC=CC=1SC1=CC(S(=O)O)=CC=C1SSC1=CC=C(S(O)=O)C=C1SC1=CC=CC=C1 UXCHAGJZXQWUCA-UHFFFAOYSA-N 0.000 description 1
- WOQNZWUQZKIDHR-UHFFFAOYSA-N 3-phenylsulfanyl-4-[(2-phenylsulfanyl-4-sulfophenyl)disulfanyl]benzenesulfonic acid Chemical compound C=1C=CC=CC=1SC1=CC(S(=O)(=O)O)=CC=C1SSC1=CC=C(S(O)(=O)=O)C=C1SC1=CC=CC=C1 WOQNZWUQZKIDHR-UHFFFAOYSA-N 0.000 description 1
- VALXHBWSRDMRCJ-UHFFFAOYSA-N 3-phenylsulfanyl-4-sulfanylbenzaldehyde Chemical compound SC1=CC=C(C=O)C=C1SC1=CC=CC=C1 VALXHBWSRDMRCJ-UHFFFAOYSA-N 0.000 description 1
- TULIEBUEXFADAB-UHFFFAOYSA-N 3-phenylsulfanyl-4-sulfanylbenzaldehyde;sodium Chemical compound [Na].SC1=CC=C(C=O)C=C1SC1=CC=CC=C1 TULIEBUEXFADAB-UHFFFAOYSA-N 0.000 description 1
- DURWKKVHNTVWOK-UHFFFAOYSA-N 3-phenylsulfanyl-4-sulfanylbenzaldehyde;zinc Chemical compound [Zn].SC1=CC=C(C=O)C=C1SC1=CC=CC=C1 DURWKKVHNTVWOK-UHFFFAOYSA-N 0.000 description 1
- FCUCUEXVAQBPKK-UHFFFAOYSA-N 3-phenylsulfanyl-4-sulfanylbenzamide Chemical compound NC(=O)C1=CC=C(S)C(SC=2C=CC=CC=2)=C1 FCUCUEXVAQBPKK-UHFFFAOYSA-N 0.000 description 1
- YENIDRAFUXKBQV-UHFFFAOYSA-N 3-phenylsulfanyl-4-sulfanylbenzamide;sodium Chemical compound [Na].NC(=O)C1=CC=C(S)C(SC=2C=CC=CC=2)=C1 YENIDRAFUXKBQV-UHFFFAOYSA-N 0.000 description 1
- RFCHWGCMPKDHBZ-UHFFFAOYSA-N 3-phenylsulfanyl-4-sulfanylbenzamide;zinc Chemical compound [Zn].NC(=O)C1=CC=C(S)C(SC=2C=CC=CC=2)=C1 RFCHWGCMPKDHBZ-UHFFFAOYSA-N 0.000 description 1
- STFVWOJGIADLNL-UHFFFAOYSA-N 3-phenylsulfanyl-4-sulfanylbenzenesulfinic acid Chemical compound OS(=O)C1=CC=C(S)C(SC=2C=CC=CC=2)=C1 STFVWOJGIADLNL-UHFFFAOYSA-N 0.000 description 1
- IBDUGXYKVOKOIG-UHFFFAOYSA-N 3-phenylsulfanyl-4-sulfanylbenzenesulfonic acid Chemical compound OS(=O)(=O)C1=CC=C(S)C(SC=2C=CC=CC=2)=C1 IBDUGXYKVOKOIG-UHFFFAOYSA-N 0.000 description 1
- JWCKAAXXGUFFTE-UHFFFAOYSA-N 3-phenylsulfanyl-4-sulfanylbenzenesulfonyl chloride Chemical compound SC1=CC=C(S(Cl)(=O)=O)C=C1SC1=CC=CC=C1 JWCKAAXXGUFFTE-UHFFFAOYSA-N 0.000 description 1
- FBEFOFHNZWUNBM-UHFFFAOYSA-N 3-phenylsulfanyl-4-sulfanylbenzenesulfonyl chloride;sodium Chemical compound [Na].SC1=CC=C(S(Cl)(=O)=O)C=C1SC1=CC=CC=C1 FBEFOFHNZWUNBM-UHFFFAOYSA-N 0.000 description 1
- PYISTXJFZTZJJN-UHFFFAOYSA-N 3-phenylsulfanyl-4-sulfanylbenzenesulfonyl chloride;zinc Chemical compound [Zn].SC1=CC=C(S(Cl)(=O)=O)C=C1SC1=CC=CC=C1 PYISTXJFZTZJJN-UHFFFAOYSA-N 0.000 description 1
- FEQGMCNHRDTMGZ-UHFFFAOYSA-N 3-phenylsulfanyl-4-sulfanylbenzoic acid Chemical compound OC(=O)C1=CC=C(S)C(SC=2C=CC=CC=2)=C1 FEQGMCNHRDTMGZ-UHFFFAOYSA-N 0.000 description 1
- HCYAQKLXGYVDJF-UHFFFAOYSA-N 3-phenylsulfanyl-4-sulfanylbenzonitrile Chemical compound SC1=CC=C(C#N)C=C1SC1=CC=CC=C1 HCYAQKLXGYVDJF-UHFFFAOYSA-N 0.000 description 1
- XBIIJIJJWBPAQE-UHFFFAOYSA-N 3-phenylsulfanyl-4-sulfanylbenzonitrile;sodium Chemical compound [Na].SC1=CC=C(C#N)C=C1SC1=CC=CC=C1 XBIIJIJJWBPAQE-UHFFFAOYSA-N 0.000 description 1
- PVWRMSXTNJNRAY-UHFFFAOYSA-N 3-phenylsulfanyl-4-sulfanylbenzonitrile;zinc Chemical compound [Zn].SC1=CC=C(C#N)C=C1SC1=CC=CC=C1 PVWRMSXTNJNRAY-UHFFFAOYSA-N 0.000 description 1
- YDBWNWJQZOGPAN-UHFFFAOYSA-N 3-phenylsulfanyl-4-sulfanylbenzoyl chloride Chemical compound SC1=CC=C(C(Cl)=O)C=C1SC1=CC=CC=C1 YDBWNWJQZOGPAN-UHFFFAOYSA-N 0.000 description 1
- VCBGBKBIRSNISP-UHFFFAOYSA-N 3-phenylsulfanyl-4-sulfanylbenzoyl chloride;sodium Chemical compound [Na].SC1=CC=C(C(Cl)=O)C=C1SC1=CC=CC=C1 VCBGBKBIRSNISP-UHFFFAOYSA-N 0.000 description 1
- KMDZCPBEWDCOSV-UHFFFAOYSA-N 3-phenylsulfanyl-4-sulfanylbenzoyl chloride;zinc Chemical compound [Zn].SC1=CC=C(C(Cl)=O)C=C1SC1=CC=CC=C1 KMDZCPBEWDCOSV-UHFFFAOYSA-N 0.000 description 1
- MHKLKWCYGIBEQF-UHFFFAOYSA-N 4-(1,3-benzothiazol-2-ylsulfanyl)morpholine Chemical compound C1COCCN1SC1=NC2=CC=CC=C2S1 MHKLKWCYGIBEQF-UHFFFAOYSA-N 0.000 description 1
- HYFIZWVNOLEFDH-UHFFFAOYSA-N 4-(carboxymethyl)benzoic acid Chemical compound C(=O)(O)CC1=CC=C(C(=O)O)C=C1.C(CC1=CC=C(C(=O)O)C=C1)(=O)O HYFIZWVNOLEFDH-UHFFFAOYSA-N 0.000 description 1
- FVOHGLILKFQSLT-UHFFFAOYSA-N 4-(trichloromethyl)benzenethiol Chemical compound SC1=CC=C(C(Cl)(Cl)Cl)C=C1 FVOHGLILKFQSLT-UHFFFAOYSA-N 0.000 description 1
- MLBJINAZDMJNGJ-UHFFFAOYSA-N 4-(trichloromethyl)benzenethiol;zinc Chemical compound [Zn].SC1=CC=C(C(Cl)(Cl)Cl)C=C1 MLBJINAZDMJNGJ-UHFFFAOYSA-N 0.000 description 1
- OKIHXNKYYGUVTE-UHFFFAOYSA-N 4-Fluorothiophenol Chemical compound FC1=CC=C(S)C=C1 OKIHXNKYYGUVTE-UHFFFAOYSA-N 0.000 description 1
- FTAHAAJKCSKTHU-UHFFFAOYSA-N 4-[(4-carbamoyl-2-hydroxyphenyl)disulfanyl]-3-hydroxybenzamide Chemical compound OC1=CC(C(=O)N)=CC=C1SSC1=CC=C(C(N)=O)C=C1O FTAHAAJKCSKTHU-UHFFFAOYSA-N 0.000 description 1
- FPCBRBRLXUBHLM-UHFFFAOYSA-N 4-[(4-carbamoyl-2-nitrophenyl)disulfanyl]-3-nitrobenzamide Chemical compound [O-][N+](=O)C1=CC(C(=O)N)=CC=C1SSC1=CC=C(C(N)=O)C=C1[N+]([O-])=O FPCBRBRLXUBHLM-UHFFFAOYSA-N 0.000 description 1
- PEQAUYNBJQOIBZ-UHFFFAOYSA-N 4-[(4-carbamoyl-2-phenylsulfanylphenyl)disulfanyl]-3-phenylsulfanylbenzamide Chemical compound C=1C=CC=CC=1SC1=CC(C(=O)N)=CC=C1SSC1=CC=C(C(N)=O)C=C1SC1=CC=CC=C1 PEQAUYNBJQOIBZ-UHFFFAOYSA-N 0.000 description 1
- QHGRZHKKJGYARF-UHFFFAOYSA-N 4-[(4-carbamoylphenyl)disulfanyl]benzamide Chemical group C1=CC(C(=O)N)=CC=C1SSC1=CC=C(C(N)=O)C=C1 QHGRZHKKJGYARF-UHFFFAOYSA-N 0.000 description 1
- ULUZUTHJZCQXBL-UHFFFAOYSA-N 4-[(4-carbonochloridoyl-2-hydroxyphenyl)disulfanyl]-3-hydroxybenzoyl chloride Chemical compound OC1=CC(C(Cl)=O)=CC=C1SSC1=CC=C(C(Cl)=O)C=C1O ULUZUTHJZCQXBL-UHFFFAOYSA-N 0.000 description 1
- FJKSUXIIYGXWEC-UHFFFAOYSA-N 4-[(4-carbonochloridoyl-2-nitrophenyl)disulfanyl]-3-nitrobenzoyl chloride Chemical compound [O-][N+](=O)C1=CC(C(Cl)=O)=CC=C1SSC1=CC=C(C(Cl)=O)C=C1[N+]([O-])=O FJKSUXIIYGXWEC-UHFFFAOYSA-N 0.000 description 1
- BLRSVONUYVIZHB-UHFFFAOYSA-N 4-[(4-carbonochloridoyl-2-phenylsulfanylphenyl)disulfanyl]-3-phenylsulfanylbenzoyl chloride Chemical compound C=1C=CC=CC=1SC1=CC(C(=O)Cl)=CC=C1SSC1=CC=C(C(Cl)=O)C=C1SC1=CC=CC=C1 BLRSVONUYVIZHB-UHFFFAOYSA-N 0.000 description 1
- LZQMZNSHRMJRKK-UHFFFAOYSA-N 4-[(4-carbonochloridoylphenyl)disulfanyl]benzoyl chloride Chemical group C1=CC(C(=O)Cl)=CC=C1SSC1=CC=C(C(Cl)=O)C=C1 LZQMZNSHRMJRKK-UHFFFAOYSA-N 0.000 description 1
- KVVFTNTWTIGWNA-UHFFFAOYSA-N 4-[(4-carboxy-2-hydroxyphenyl)disulfanyl]-3-hydroxybenzoic acid Chemical compound OC1=CC(C(=O)O)=CC=C1SSC1=CC=C(C(O)=O)C=C1O KVVFTNTWTIGWNA-UHFFFAOYSA-N 0.000 description 1
- AZVMFHXFQAGELP-UHFFFAOYSA-N 4-[(4-carboxy-2-nitrophenyl)disulfanyl]-3-nitrobenzoic acid Chemical compound [O-][N+](=O)C1=CC(C(=O)O)=CC=C1SSC1=CC=C(C(O)=O)C=C1[N+]([O-])=O AZVMFHXFQAGELP-UHFFFAOYSA-N 0.000 description 1
- MRPSYTHWBBZUTF-UHFFFAOYSA-N 4-[(4-carboxy-2-phenylsulfanylphenyl)disulfanyl]-3-phenylsulfanylbenzoic acid Chemical compound C=1C=CC=CC=1SC1=CC(C(=O)O)=CC=C1SSC1=CC=C(C(O)=O)C=C1SC1=CC=CC=C1 MRPSYTHWBBZUTF-UHFFFAOYSA-N 0.000 description 1
- GAMSSMZJKUMFEY-UHFFFAOYSA-N 4-[(4-carboxyphenyl)disulfanyl]benzoic acid Chemical group C1=CC(C(=O)O)=CC=C1SSC1=CC=C(C(O)=O)C=C1 GAMSSMZJKUMFEY-UHFFFAOYSA-N 0.000 description 1
- YYQWQSJSFIGFMU-UHFFFAOYSA-N 4-[(4-chlorosulfonyl-2-hydroxyphenyl)disulfanyl]-3-hydroxybenzenesulfonyl chloride Chemical compound OC1=CC(S(Cl)(=O)=O)=CC=C1SSC1=CC=C(S(Cl)(=O)=O)C=C1O YYQWQSJSFIGFMU-UHFFFAOYSA-N 0.000 description 1
- VWBNTJXAVZJLQI-UHFFFAOYSA-N 4-[(4-chlorosulfonyl-2-nitrophenyl)disulfanyl]-3-nitrobenzenesulfonyl chloride Chemical compound [O-][N+](=O)C1=CC(S(Cl)(=O)=O)=CC=C1SSC1=CC=C(S(Cl)(=O)=O)C=C1[N+]([O-])=O VWBNTJXAVZJLQI-UHFFFAOYSA-N 0.000 description 1
- ZCIFHTHQCXCGKP-UHFFFAOYSA-N 4-[(4-chlorosulfonyl-2-phenylsulfanylphenyl)disulfanyl]-3-phenylsulfanylbenzenesulfonyl chloride Chemical compound C=1C=CC=CC=1SC1=CC(S(=O)(=O)Cl)=CC=C1SSC1=CC=C(S(Cl)(=O)=O)C=C1SC1=CC=CC=C1 ZCIFHTHQCXCGKP-UHFFFAOYSA-N 0.000 description 1
- VUKZJNCBRXLCAA-UHFFFAOYSA-N 4-[(4-chlorosulfonylphenyl)disulfanyl]benzenesulfonyl chloride Chemical group C1=CC(S(=O)(=O)Cl)=CC=C1SSC1=CC=C(S(Cl)(=O)=O)C=C1 VUKZJNCBRXLCAA-UHFFFAOYSA-N 0.000 description 1
- PZBMRRZQRVSHKN-UHFFFAOYSA-N 4-[(4-cyano-2-hydroxyphenyl)disulfanyl]-3-hydroxybenzonitrile Chemical compound OC1=CC(C#N)=CC=C1SSC1=CC=C(C#N)C=C1O PZBMRRZQRVSHKN-UHFFFAOYSA-N 0.000 description 1
- PFCCJXRAXQCDMK-UHFFFAOYSA-N 4-[(4-cyano-2-nitrophenyl)disulfanyl]-3-nitrobenzonitrile Chemical compound [O-][N+](=O)C1=CC(C#N)=CC=C1SSC1=CC=C(C#N)C=C1[N+]([O-])=O PFCCJXRAXQCDMK-UHFFFAOYSA-N 0.000 description 1
- GPNWTFDOAXIYMV-UHFFFAOYSA-N 4-[(4-cyano-2-phenylsulfanylphenyl)disulfanyl]-3-phenylsulfanylbenzonitrile Chemical compound C=1C=CC=CC=1SC1=CC(C#N)=CC=C1SSC1=CC=C(C#N)C=C1SC1=CC=CC=C1 GPNWTFDOAXIYMV-UHFFFAOYSA-N 0.000 description 1
- BCUVQZBVGSXWCG-UHFFFAOYSA-N 4-[(4-cyanophenyl)disulfanyl]benzonitrile Chemical compound C1=CC(C#N)=CC=C1SSC1=CC=C(C#N)C=C1 BCUVQZBVGSXWCG-UHFFFAOYSA-N 0.000 description 1
- XIBJJZBBXVCTPR-UHFFFAOYSA-N 4-[(4-formyl-2-hydroxyphenyl)disulfanyl]-3-hydroxybenzaldehyde Chemical compound OC1=CC(C=O)=CC=C1SSC1=CC=C(C=O)C=C1O XIBJJZBBXVCTPR-UHFFFAOYSA-N 0.000 description 1
- JALWVTKNYNANRB-UHFFFAOYSA-N 4-[(4-formyl-2-nitrophenyl)disulfanyl]-3-nitrobenzaldehyde Chemical compound [O-][N+](=O)C1=CC(C=O)=CC=C1SSC1=CC=C(C=O)C=C1[N+]([O-])=O JALWVTKNYNANRB-UHFFFAOYSA-N 0.000 description 1
- XVKUOOVERWVUSB-UHFFFAOYSA-N 4-[(4-formyl-2-phenylsulfanylphenyl)disulfanyl]-3-phenylsulfanylbenzaldehyde Chemical compound C=1C=CC=CC=1SC1=CC(C=O)=CC=C1SSC1=CC=C(C=O)C=C1SC1=CC=CC=C1 XVKUOOVERWVUSB-UHFFFAOYSA-N 0.000 description 1
- GSPKFDVCAFADTI-UHFFFAOYSA-N 4-[(4-formylphenyl)disulfanyl]benzaldehyde Chemical group C1=CC(C=O)=CC=C1SSC1=CC=C(C=O)C=C1 GSPKFDVCAFADTI-UHFFFAOYSA-N 0.000 description 1
- YBCCVBQPJXNJLL-UHFFFAOYSA-N 4-[(4-sulfinophenyl)disulfanyl]benzenesulfinic acid Chemical group C1=CC(S(=O)O)=CC=C1SSC1=CC=C(S(O)=O)C=C1 YBCCVBQPJXNJLL-UHFFFAOYSA-N 0.000 description 1
- LUENVHHLGFLMFJ-UHFFFAOYSA-N 4-[(4-sulfophenyl)disulfanyl]benzenesulfonic acid Chemical group C1=CC(S(=O)(=O)O)=CC=C1SSC1=CC=C(S(O)(=O)=O)C=C1 LUENVHHLGFLMFJ-UHFFFAOYSA-N 0.000 description 1
- FTBCOQFMQSTCQQ-UHFFFAOYSA-N 4-bromobenzenethiol Chemical compound SC1=CC=C(Br)C=C1 FTBCOQFMQSTCQQ-UHFFFAOYSA-N 0.000 description 1
- YDLIDTMQRMRQKU-UHFFFAOYSA-N 4-bromobenzenethiol;sodium Chemical compound [Na].SC1=CC=C(Br)C=C1 YDLIDTMQRMRQKU-UHFFFAOYSA-N 0.000 description 1
- CRAFCSNAOKYCRU-UHFFFAOYSA-N 4-bromobenzenethiol;zinc Chemical compound [Zn].SC1=CC=C(Br)C=C1 CRAFCSNAOKYCRU-UHFFFAOYSA-N 0.000 description 1
- DESADCWXGJLRSR-UHFFFAOYSA-N 4-chloro-1-[(4-chloro-2-nitrophenyl)disulfanyl]-2-nitrobenzene Chemical compound [O-][N+](=O)C1=CC(Cl)=CC=C1SSC1=CC=C(Cl)C=C1[N+]([O-])=O DESADCWXGJLRSR-UHFFFAOYSA-N 0.000 description 1
- XMEPVRKUDORHTL-UHFFFAOYSA-N 4-chloro-1-[(4-chloro-2-phenylsulfanylphenyl)disulfanyl]-2-phenylsulfanylbenzene Chemical compound C=1C=CC=CC=1SC1=CC(Cl)=CC=C1SSC1=CC=C(Cl)C=C1SC1=CC=CC=C1 XMEPVRKUDORHTL-UHFFFAOYSA-N 0.000 description 1
- YAESYRWEEVNLOA-UHFFFAOYSA-N 4-chloro-2-nitrobenzenethiol Chemical compound [O-][N+](=O)C1=CC(Cl)=CC=C1S YAESYRWEEVNLOA-UHFFFAOYSA-N 0.000 description 1
- MPRIRVKZIPSLGN-UHFFFAOYSA-N 4-chloro-2-nitrobenzenethiol;sodium Chemical compound [Na].[O-][N+](=O)C1=CC(Cl)=CC=C1S MPRIRVKZIPSLGN-UHFFFAOYSA-N 0.000 description 1
- RETUUCYTZXUSQJ-UHFFFAOYSA-N 4-chloro-2-nitrobenzenethiol;zinc Chemical compound [Zn].[O-][N+](=O)C1=CC(Cl)=CC=C1S RETUUCYTZXUSQJ-UHFFFAOYSA-N 0.000 description 1
- FURSEZKDUOBNGA-UHFFFAOYSA-N 4-chloro-2-phenylsulfanylbenzenethiol Chemical compound SC1=CC=C(Cl)C=C1SC1=CC=CC=C1 FURSEZKDUOBNGA-UHFFFAOYSA-N 0.000 description 1
- HPDUIALUGLJTKA-UHFFFAOYSA-N 4-chloro-2-phenylsulfanylbenzenethiol;sodium Chemical compound [Na].SC1=CC=C(Cl)C=C1SC1=CC=CC=C1 HPDUIALUGLJTKA-UHFFFAOYSA-N 0.000 description 1
- WFYLNZXJPTYQCC-UHFFFAOYSA-N 4-chloro-2-phenylsulfanylbenzenethiol;zinc Chemical compound [Zn].SC1=CC=C(Cl)C=C1SC1=CC=CC=C1 WFYLNZXJPTYQCC-UHFFFAOYSA-N 0.000 description 1
- VZXOZSQDJJNBRC-UHFFFAOYSA-N 4-chlorobenzenethiol Chemical compound SC1=CC=C(Cl)C=C1 VZXOZSQDJJNBRC-UHFFFAOYSA-N 0.000 description 1
- JKYJNJJOOAKMLB-UHFFFAOYSA-N 4-chlorobenzenethiol;sodium Chemical compound [Na].SC1=CC=C(Cl)C=C1 JKYJNJJOOAKMLB-UHFFFAOYSA-N 0.000 description 1
- ARMKTTCTVPCUTA-UHFFFAOYSA-N 4-chlorobenzenethiol;zinc Chemical compound [Zn].SC1=CC=C(Cl)C=C1 ARMKTTCTVPCUTA-UHFFFAOYSA-N 0.000 description 1
- AQLVOIGFVDPUCK-UHFFFAOYSA-N 4-fluorobenzenethiol;sodium Chemical compound [Na].FC1=CC=C(S)C=C1 AQLVOIGFVDPUCK-UHFFFAOYSA-N 0.000 description 1
- YMPOALZXIPGKIX-UHFFFAOYSA-N 4-fluorobenzenethiol;zinc Chemical compound [Zn].FC1=CC=C(S)C=C1 YMPOALZXIPGKIX-UHFFFAOYSA-N 0.000 description 1
- IKZUTVQEBGHQJA-UHFFFAOYSA-N 4-iodobenzenethiol Chemical compound SC1=CC=C(I)C=C1 IKZUTVQEBGHQJA-UHFFFAOYSA-N 0.000 description 1
- LTWGNKUGRGIAEQ-UHFFFAOYSA-N 4-iodobenzenethiol;sodium Chemical compound [Na].SC1=CC=C(I)C=C1 LTWGNKUGRGIAEQ-UHFFFAOYSA-N 0.000 description 1
- QUDWVRGEEKSPLO-UHFFFAOYSA-N 4-iodobenzenethiol;zinc Chemical compound [Zn].SC1=CC=C(I)C=C1 QUDWVRGEEKSPLO-UHFFFAOYSA-N 0.000 description 1
- GDPGXDPBJUYOJU-UHFFFAOYSA-N 4-methoxy-1-[(4-methoxy-2-nitrophenyl)disulfanyl]-2-nitrobenzene Chemical compound [O-][N+](=O)C1=CC(OC)=CC=C1SSC1=CC=C(OC)C=C1[N+]([O-])=O GDPGXDPBJUYOJU-UHFFFAOYSA-N 0.000 description 1
- BYUWQARTKOCNQW-UHFFFAOYSA-N 4-methoxy-1-[(4-methoxy-2-phenylsulfanylphenyl)disulfanyl]-2-phenylsulfanylbenzene Chemical compound C=1C=CC=CC=1SC1=CC(OC)=CC=C1SSC1=CC=C(OC)C=C1SC1=CC=CC=C1 BYUWQARTKOCNQW-UHFFFAOYSA-N 0.000 description 1
- POWGCXLBEOGHNG-UHFFFAOYSA-N 4-methoxy-2-nitrobenzenethiol Chemical compound COC1=CC=C(S)C([N+]([O-])=O)=C1 POWGCXLBEOGHNG-UHFFFAOYSA-N 0.000 description 1
- WMMGLBNUXHVZGU-UHFFFAOYSA-N 4-methoxy-2-nitrobenzenethiol;sodium Chemical compound [Na].COC1=CC=C(S)C([N+]([O-])=O)=C1 WMMGLBNUXHVZGU-UHFFFAOYSA-N 0.000 description 1
- ZZZGVFPVUDIBDK-UHFFFAOYSA-N 4-methoxy-2-nitrobenzenethiol;zinc Chemical compound [Zn].COC1=CC=C(S)C([N+]([O-])=O)=C1 ZZZGVFPVUDIBDK-UHFFFAOYSA-N 0.000 description 1
- REOKAKWTQDXTSH-UHFFFAOYSA-N 4-methoxy-2-phenylsulfanylbenzenethiol Chemical compound COC1=CC=C(S)C(SC=2C=CC=CC=2)=C1 REOKAKWTQDXTSH-UHFFFAOYSA-N 0.000 description 1
- GDDMPQNKOIDJBD-UHFFFAOYSA-N 4-methoxy-2-phenylsulfanylbenzenethiol;sodium Chemical compound [Na].COC1=CC=C(S)C(SC=2C=CC=CC=2)=C1 GDDMPQNKOIDJBD-UHFFFAOYSA-N 0.000 description 1
- NNHPUHYRSXIBCS-UHFFFAOYSA-N 4-methoxy-2-phenylsulfanylbenzenethiol;zinc Chemical compound [Zn].COC1=CC=C(S)C(SC=2C=CC=CC=2)=C1 NNHPUHYRSXIBCS-UHFFFAOYSA-N 0.000 description 1
- NIFAOMSJMGEFTQ-UHFFFAOYSA-N 4-methoxybenzenethiol Chemical compound COC1=CC=C(S)C=C1 NIFAOMSJMGEFTQ-UHFFFAOYSA-N 0.000 description 1
- AAMIZCAOMORXMU-UHFFFAOYSA-N 4-methoxybenzenethiol;sodium Chemical compound [Na].COC1=CC=C(S)C=C1 AAMIZCAOMORXMU-UHFFFAOYSA-N 0.000 description 1
- CJYRMNCJUZEHCF-UHFFFAOYSA-N 4-methoxybenzenethiol;zinc Chemical compound [Zn].COC1=CC=C(S)C=C1 CJYRMNCJUZEHCF-UHFFFAOYSA-N 0.000 description 1
- MBTLEECRJWKPIP-UHFFFAOYSA-N 4-methyl-1-[(4-methyl-2-nitrophenyl)disulfanyl]-2-nitrobenzene Chemical compound [O-][N+](=O)C1=CC(C)=CC=C1SSC1=CC=C(C)C=C1[N+]([O-])=O MBTLEECRJWKPIP-UHFFFAOYSA-N 0.000 description 1
- INCKURQVNDWSBP-UHFFFAOYSA-N 4-methyl-1-[(4-methyl-2-phenylsulfanylphenyl)disulfanyl]-2-phenylsulfanylbenzene Chemical compound C=1C=CC=CC=1SC1=CC(C)=CC=C1SSC1=CC=C(C)C=C1SC1=CC=CC=C1 INCKURQVNDWSBP-UHFFFAOYSA-N 0.000 description 1
- MEEVQELVDKDHLB-UHFFFAOYSA-N 4-methyl-2-nitrobenzenethiol Chemical compound CC1=CC=C(S)C([N+]([O-])=O)=C1 MEEVQELVDKDHLB-UHFFFAOYSA-N 0.000 description 1
- LGIUBZRDAGLYPG-UHFFFAOYSA-N 4-methyl-2-nitrobenzenethiol;sodium Chemical compound [Na].CC1=CC=C(S)C([N+]([O-])=O)=C1 LGIUBZRDAGLYPG-UHFFFAOYSA-N 0.000 description 1
- KYHBKWQIXRLEJA-UHFFFAOYSA-N 4-methyl-2-nitrobenzenethiol;zinc Chemical compound [Zn].CC1=CC=C(S)C([N+]([O-])=O)=C1 KYHBKWQIXRLEJA-UHFFFAOYSA-N 0.000 description 1
- YLFNWCRGYBXOLX-UHFFFAOYSA-N 4-methyl-2-phenylsulfanylbenzenethiol Chemical compound CC1=CC=C(S)C(SC=2C=CC=CC=2)=C1 YLFNWCRGYBXOLX-UHFFFAOYSA-N 0.000 description 1
- AKYVXRRONVHCFM-UHFFFAOYSA-N 4-methyl-2-phenylsulfanylbenzenethiol;sodium Chemical compound [Na].CC1=CC=C(S)C(SC=2C=CC=CC=2)=C1 AKYVXRRONVHCFM-UHFFFAOYSA-N 0.000 description 1
- SUARCLQLQVSKQX-UHFFFAOYSA-N 4-methyl-2-phenylsulfanylbenzenethiol;zinc Chemical compound [Zn].CC1=CC=C(S)C(SC=2C=CC=CC=2)=C1 SUARCLQLQVSKQX-UHFFFAOYSA-N 0.000 description 1
- WLHCBQAPPJAULW-UHFFFAOYSA-N 4-methylbenzenethiol Chemical compound CC1=CC=C(S)C=C1 WLHCBQAPPJAULW-UHFFFAOYSA-N 0.000 description 1
- MIPHPIQGSPIJAE-UHFFFAOYSA-N 4-methylbenzenethiol;sodium Chemical compound [Na].CC1=CC=C(S)C=C1 MIPHPIQGSPIJAE-UHFFFAOYSA-N 0.000 description 1
- ZKCKVQQWQIYOFQ-UHFFFAOYSA-N 4-methylbenzenethiol;zinc Chemical compound [Zn].CC1=CC=C(S)C=C1 ZKCKVQQWQIYOFQ-UHFFFAOYSA-N 0.000 description 1
- XDQJSXWPDAMVOT-UHFFFAOYSA-N 4-methylsulfinyl-1-[(4-methylsulfinyl-2-nitrophenyl)disulfanyl]-2-nitrobenzene Chemical compound [O-][N+](=O)C1=CC(S(=O)C)=CC=C1SSC1=CC=C(S(C)=O)C=C1[N+]([O-])=O XDQJSXWPDAMVOT-UHFFFAOYSA-N 0.000 description 1
- CRPFFSCCMQMRHV-UHFFFAOYSA-N 4-methylsulfinyl-1-[(4-methylsulfinyl-2-phenylsulfanylphenyl)disulfanyl]-2-phenylsulfanylbenzene Chemical compound C=1C=CC=CC=1SC1=CC(S(=O)C)=CC=C1SSC1=CC=C(S(C)=O)C=C1SC1=CC=CC=C1 CRPFFSCCMQMRHV-UHFFFAOYSA-N 0.000 description 1
- RVHTXXSUWJXYER-UHFFFAOYSA-N 4-methylsulfinyl-2-nitrobenzenethiol Chemical compound CS(=O)C1=CC=C(S)C([N+]([O-])=O)=C1 RVHTXXSUWJXYER-UHFFFAOYSA-N 0.000 description 1
- QEOCRAQFFOXEMH-UHFFFAOYSA-N 4-methylsulfinyl-2-nitrobenzenethiol;sodium Chemical compound [Na].CS(=O)C1=CC=C(S)C([N+]([O-])=O)=C1 QEOCRAQFFOXEMH-UHFFFAOYSA-N 0.000 description 1
- QRRNULWQOYQMCX-UHFFFAOYSA-N 4-methylsulfinyl-2-nitrobenzenethiol;zinc Chemical compound [Zn].CS(=O)C1=CC=C(S)C([N+]([O-])=O)=C1 QRRNULWQOYQMCX-UHFFFAOYSA-N 0.000 description 1
- KGXHHLGPJBCGLZ-UHFFFAOYSA-N 4-methylsulfinyl-2-phenylsulfanylbenzenethiol Chemical compound CS(=O)C1=CC=C(S)C(SC=2C=CC=CC=2)=C1 KGXHHLGPJBCGLZ-UHFFFAOYSA-N 0.000 description 1
- FKYHTJSBNCIWCX-UHFFFAOYSA-N 4-methylsulfinyl-2-phenylsulfanylbenzenethiol;sodium Chemical compound [Na].CS(=O)C1=CC=C(S)C(SC=2C=CC=CC=2)=C1 FKYHTJSBNCIWCX-UHFFFAOYSA-N 0.000 description 1
- IXPVIZJZNXUEJG-UHFFFAOYSA-N 4-methylsulfinyl-2-phenylsulfanylbenzenethiol;zinc Chemical compound [Zn].CS(=O)C1=CC=C(S)C(SC=2C=CC=CC=2)=C1 IXPVIZJZNXUEJG-UHFFFAOYSA-N 0.000 description 1
- HSQAADZXGOHMNQ-UHFFFAOYSA-N 4-methylsulfinylbenzenethiol Chemical compound CS(=O)C1=CC=C(S)C=C1 HSQAADZXGOHMNQ-UHFFFAOYSA-N 0.000 description 1
- ZADQZSVXXXMSBM-UHFFFAOYSA-N 4-methylsulfinylbenzenethiol;sodium Chemical compound [Na].CS(=O)C1=CC=C(S)C=C1 ZADQZSVXXXMSBM-UHFFFAOYSA-N 0.000 description 1
- SCYXJPHLQSOXSM-UHFFFAOYSA-N 4-methylsulfinylbenzenethiol;zinc Chemical compound [Zn].CS(=O)C1=CC=C(S)C=C1 SCYXJPHLQSOXSM-UHFFFAOYSA-N 0.000 description 1
- HZUNZWYHHZWZHB-UHFFFAOYSA-N 4-oxalobenzoic acid Chemical compound OC(=O)C(=O)C1=CC=C(C(O)=O)C=C1 HZUNZWYHHZWZHB-UHFFFAOYSA-N 0.000 description 1
- NRLPPGBADQVXSJ-UHFFFAOYSA-N 4-sulfanylbenzaldehyde Chemical compound SC1=CC=C(C=O)C=C1 NRLPPGBADQVXSJ-UHFFFAOYSA-N 0.000 description 1
- WDMPQJBJCAIFQI-UHFFFAOYSA-N 4-sulfanylbenzaldehyde;zinc Chemical compound [Zn].SC1=CC=C(C=O)C=C1 WDMPQJBJCAIFQI-UHFFFAOYSA-N 0.000 description 1
- CTOUXQDUYJORQK-UHFFFAOYSA-N 4-sulfanylbenzamide Chemical compound NC(=O)C1=CC=C(S)C=C1 CTOUXQDUYJORQK-UHFFFAOYSA-N 0.000 description 1
- PJXZGOLSDFYALX-UHFFFAOYSA-N 4-sulfanylbenzamide;zinc Chemical compound [Zn].NC(=O)C1=CC=C(S)C=C1 PJXZGOLSDFYALX-UHFFFAOYSA-N 0.000 description 1
- QFSXSKUPBOIUCV-UHFFFAOYSA-N 4-sulfanylbenzenesulfinic acid Chemical compound OS(=O)C1=CC=C(S)C=C1 QFSXSKUPBOIUCV-UHFFFAOYSA-N 0.000 description 1
- ULBNVDPBWRTOPN-UHFFFAOYSA-N 4-sulfanylbenzenesulfonic acid Chemical compound OS(=O)(=O)C1=CC=C(S)C=C1 ULBNVDPBWRTOPN-UHFFFAOYSA-N 0.000 description 1
- MXVGIZYLVMLDPP-UHFFFAOYSA-N 4-sulfanylbenzenesulfonyl chloride Chemical compound SC1=CC=C(S(Cl)(=O)=O)C=C1 MXVGIZYLVMLDPP-UHFFFAOYSA-N 0.000 description 1
- UBKAJUVADLDOKA-UHFFFAOYSA-N 4-sulfanylbenzenesulfonyl chloride;zinc Chemical compound [Zn].SC1=CC=C(S(Cl)(=O)=O)C=C1 UBKAJUVADLDOKA-UHFFFAOYSA-N 0.000 description 1
- LMJXSOYPAOSIPZ-UHFFFAOYSA-N 4-sulfanylbenzoic acid Chemical compound OC(=O)C1=CC=C(S)C=C1 LMJXSOYPAOSIPZ-UHFFFAOYSA-N 0.000 description 1
- MVPUXVBBHWUOFS-UHFFFAOYSA-N 4-sulfanylbenzonitrile Chemical compound SC1=CC=C(C#N)C=C1 MVPUXVBBHWUOFS-UHFFFAOYSA-N 0.000 description 1
- YKLXHOVRMYCEIY-UHFFFAOYSA-N 4-sulfanylbenzonitrile;zinc Chemical compound [Zn].SC1=CC=C(C#N)C=C1 YKLXHOVRMYCEIY-UHFFFAOYSA-N 0.000 description 1
- PIGPGBJMESGOTG-UHFFFAOYSA-N 4-sulfanylbenzoyl chloride Chemical compound SC1=CC=C(C(Cl)=O)C=C1 PIGPGBJMESGOTG-UHFFFAOYSA-N 0.000 description 1
- MWCSZMXXTJHRHL-UHFFFAOYSA-N 4-sulfanylbenzoyl chloride;zinc Chemical compound [Zn].SC1=CC=C(C(Cl)=O)C=C1 MWCSZMXXTJHRHL-UHFFFAOYSA-N 0.000 description 1
- GNXBFFHXJDZGEK-UHFFFAOYSA-N 4-tert-butylbenzenethiol Chemical compound CC(C)(C)C1=CC=C(S)C=C1 GNXBFFHXJDZGEK-UHFFFAOYSA-N 0.000 description 1
- ZYVISVFANLTAQA-UHFFFAOYSA-N 4-tert-butylbenzenethiol;sodium Chemical compound [Na].CC(C)(C)C1=CC=C(S)C=C1 ZYVISVFANLTAQA-UHFFFAOYSA-N 0.000 description 1
- NRGPZHZHPDCRDX-UHFFFAOYSA-N 4-tert-butylbenzenethiol;zinc Chemical compound [Zn].CC(C)(C)C1=CC=C(S)C=C1 NRGPZHZHPDCRDX-UHFFFAOYSA-N 0.000 description 1
- TZUOTVOJJUHDKB-UHFFFAOYSA-N 5-acetyl-2-[(4-acetyl-2-carbamoylphenyl)disulfanyl]benzamide Chemical compound NC(=O)C1=CC(C(=O)C)=CC=C1SSC1=CC=C(C(C)=O)C=C1C(N)=O TZUOTVOJJUHDKB-UHFFFAOYSA-N 0.000 description 1
- KNMYRVGSBBUIRJ-UHFFFAOYSA-N 5-acetyl-2-[(4-acetyl-2-carbonochloridoylphenyl)disulfanyl]benzoyl chloride Chemical compound ClC(=O)C1=CC(C(=O)C)=CC=C1SSC1=CC=C(C(C)=O)C=C1C(Cl)=O KNMYRVGSBBUIRJ-UHFFFAOYSA-N 0.000 description 1
- BOODKCOSYZTYSL-UHFFFAOYSA-N 5-acetyl-2-[(4-acetyl-2-carboxyphenyl)disulfanyl]benzoic acid Chemical compound OC(=O)C1=CC(C(=O)C)=CC=C1SSC1=CC=C(C(C)=O)C=C1C(O)=O BOODKCOSYZTYSL-UHFFFAOYSA-N 0.000 description 1
- AGUNIACFFFEFIC-UHFFFAOYSA-N 5-acetyl-2-[(4-acetyl-2-chlorosulfonylphenyl)disulfanyl]benzenesulfonyl chloride Chemical compound ClS(=O)(=O)C1=CC(C(=O)C)=CC=C1SSC1=CC=C(C(C)=O)C=C1S(Cl)(=O)=O AGUNIACFFFEFIC-UHFFFAOYSA-N 0.000 description 1
- SWFXPCPUMGQPNY-UHFFFAOYSA-N 5-acetyl-2-[(4-acetyl-2-cyanophenyl)disulfanyl]benzonitrile Chemical compound N#CC1=CC(C(=O)C)=CC=C1SSC1=CC=C(C(C)=O)C=C1C#N SWFXPCPUMGQPNY-UHFFFAOYSA-N 0.000 description 1
- IFWCLSWXZNZPLI-UHFFFAOYSA-N 5-acetyl-2-[(4-acetyl-2-formylphenyl)disulfanyl]benzaldehyde Chemical compound O=CC1=CC(C(=O)C)=CC=C1SSC1=CC=C(C(C)=O)C=C1C=O IFWCLSWXZNZPLI-UHFFFAOYSA-N 0.000 description 1
- GSXZOJDGWHBHPP-UHFFFAOYSA-N 5-acetyl-2-[(4-acetyl-2-sulfinophenyl)disulfanyl]benzenesulfinic acid Chemical compound OS(=O)C1=CC(C(=O)C)=CC=C1SSC1=CC=C(C(C)=O)C=C1S(O)=O GSXZOJDGWHBHPP-UHFFFAOYSA-N 0.000 description 1
- SPJPTMTWFIXHAT-UHFFFAOYSA-N 5-acetyl-2-[(4-acetyl-2-sulfophenyl)disulfanyl]benzenesulfonic acid Chemical compound OS(=O)(=O)C1=CC(C(=O)C)=CC=C1SSC1=CC=C(C(C)=O)C=C1S(O)(=O)=O SPJPTMTWFIXHAT-UHFFFAOYSA-N 0.000 description 1
- BRBYWPUWYSRHSW-UHFFFAOYSA-N 5-acetyl-2-sulfanylbenzaldehyde Chemical compound CC(=O)C1=CC=C(S)C(C=O)=C1 BRBYWPUWYSRHSW-UHFFFAOYSA-N 0.000 description 1
- XRRYIDLIBSAZKB-UHFFFAOYSA-N 5-acetyl-2-sulfanylbenzaldehyde;sodium Chemical compound [Na].CC(=O)C1=CC=C(S)C(C=O)=C1 XRRYIDLIBSAZKB-UHFFFAOYSA-N 0.000 description 1
- OJNRYBYHSMVBSN-UHFFFAOYSA-N 5-acetyl-2-sulfanylbenzaldehyde;zinc Chemical compound [Zn].CC(=O)C1=CC=C(S)C(C=O)=C1 OJNRYBYHSMVBSN-UHFFFAOYSA-N 0.000 description 1
- MSOSTCBAMDIHNW-UHFFFAOYSA-N 5-acetyl-2-sulfanylbenzamide Chemical compound CC(=O)C1=CC=C(S)C(C(N)=O)=C1 MSOSTCBAMDIHNW-UHFFFAOYSA-N 0.000 description 1
- HKFBBDDEBDCBEQ-UHFFFAOYSA-N 5-acetyl-2-sulfanylbenzamide;sodium Chemical compound [Na].CC(=O)C1=CC=C(S)C(C(N)=O)=C1 HKFBBDDEBDCBEQ-UHFFFAOYSA-N 0.000 description 1
- NTNWZYVKYODVQB-UHFFFAOYSA-N 5-acetyl-2-sulfanylbenzamide;zinc Chemical compound [Zn].CC(=O)C1=CC=C(S)C(C(N)=O)=C1 NTNWZYVKYODVQB-UHFFFAOYSA-N 0.000 description 1
- MRKNXOHBOZFUMB-UHFFFAOYSA-N 5-acetyl-2-sulfanylbenzenesulfinic acid Chemical compound CC(=O)C1=CC=C(S)C(S(O)=O)=C1 MRKNXOHBOZFUMB-UHFFFAOYSA-N 0.000 description 1
- QAQYFESUHBWCJP-UHFFFAOYSA-N 5-acetyl-2-sulfanylbenzenesulfonic acid Chemical compound CC(=O)C1=CC=C(S)C(S(O)(=O)=O)=C1 QAQYFESUHBWCJP-UHFFFAOYSA-N 0.000 description 1
- ZTFYANZYMOUUOO-UHFFFAOYSA-N 5-acetyl-2-sulfanylbenzenesulfonyl chloride Chemical compound CC(=O)C1=CC=C(S)C(S(Cl)(=O)=O)=C1 ZTFYANZYMOUUOO-UHFFFAOYSA-N 0.000 description 1
- QVYMUHNVFYCDDC-UHFFFAOYSA-N 5-acetyl-2-sulfanylbenzenesulfonyl chloride;sodium Chemical compound [Na].CC(=O)C1=CC=C(S)C(S(Cl)(=O)=O)=C1 QVYMUHNVFYCDDC-UHFFFAOYSA-N 0.000 description 1
- CHUZNAHXNDENMH-UHFFFAOYSA-N 5-acetyl-2-sulfanylbenzenesulfonyl chloride;zinc Chemical compound [Zn].CC(=O)C1=CC=C(S)C(S(Cl)(=O)=O)=C1 CHUZNAHXNDENMH-UHFFFAOYSA-N 0.000 description 1
- DBXHFAFECCYHSL-UHFFFAOYSA-N 5-acetyl-2-sulfanylbenzoic acid Chemical compound CC(=O)C1=CC=C(S)C(C(O)=O)=C1 DBXHFAFECCYHSL-UHFFFAOYSA-N 0.000 description 1
- QUEQOMZJWZTAJR-UHFFFAOYSA-N 5-acetyl-2-sulfanylbenzonitrile Chemical compound CC(=O)C1=CC=C(S)C(C#N)=C1 QUEQOMZJWZTAJR-UHFFFAOYSA-N 0.000 description 1
- ADMPZYGQWKQTSI-UHFFFAOYSA-N 5-acetyl-2-sulfanylbenzonitrile;sodium Chemical compound [Na].CC(=O)C1=CC=C(S)C(C#N)=C1 ADMPZYGQWKQTSI-UHFFFAOYSA-N 0.000 description 1
- NRCFIMHCMSOLFZ-UHFFFAOYSA-N 5-acetyl-2-sulfanylbenzonitrile;zinc Chemical compound [Zn].CC(=O)C1=CC=C(S)C(C#N)=C1 NRCFIMHCMSOLFZ-UHFFFAOYSA-N 0.000 description 1
- LWGWJEXZRYWBFY-UHFFFAOYSA-N 5-acetyl-2-sulfanylbenzoyl chloride Chemical compound CC(=O)C1=CC=C(S)C(C(Cl)=O)=C1 LWGWJEXZRYWBFY-UHFFFAOYSA-N 0.000 description 1
- AIPPKKGCQMHMRU-UHFFFAOYSA-N 5-acetyl-2-sulfanylbenzoyl chloride;sodium Chemical compound [Na].CC(=O)C1=CC=C(S)C(C(Cl)=O)=C1 AIPPKKGCQMHMRU-UHFFFAOYSA-N 0.000 description 1
- QNQZLQZLJNNCIO-UHFFFAOYSA-N 5-acetyl-2-sulfanylbenzoyl chloride;zinc Chemical compound [Zn].CC(=O)C1=CC=C(S)C(C(Cl)=O)=C1 QNQZLQZLJNNCIO-UHFFFAOYSA-N 0.000 description 1
- OMGWQHDPPXQNCH-UHFFFAOYSA-N 5-chloro-2-[(4-chloro-2-hydroxyphenyl)disulfanyl]phenol Chemical compound OC1=CC(Cl)=CC=C1SSC1=CC=C(Cl)C=C1O OMGWQHDPPXQNCH-UHFFFAOYSA-N 0.000 description 1
- KGCLPWUBYGOUMV-UHFFFAOYSA-N 5-chloro-2-sulfanylphenol Chemical compound OC1=CC(Cl)=CC=C1S KGCLPWUBYGOUMV-UHFFFAOYSA-N 0.000 description 1
- RMOQZBZNGUTLOA-UHFFFAOYSA-N 5-chloro-2-sulfanylphenol;sodium Chemical compound [Na].OC1=CC(Cl)=CC=C1S RMOQZBZNGUTLOA-UHFFFAOYSA-N 0.000 description 1
- GRQJJYDTFSWJAI-UHFFFAOYSA-N 5-chloro-2-sulfanylphenol;zinc Chemical compound [Zn].OC1=CC(Cl)=CC=C1S GRQJJYDTFSWJAI-UHFFFAOYSA-N 0.000 description 1
- PTKWENDYGQZVBT-UHFFFAOYSA-N 5-methoxy-2-sulfanylphenol Chemical compound COC1=CC=C(S)C(O)=C1 PTKWENDYGQZVBT-UHFFFAOYSA-N 0.000 description 1
- UHHDNGHUCVVZET-UHFFFAOYSA-N 5-methoxy-2-sulfanylphenol;sodium Chemical compound [Na].COC1=CC=C(S)C(O)=C1 UHHDNGHUCVVZET-UHFFFAOYSA-N 0.000 description 1
- GXUMZMQSOBZWDA-UHFFFAOYSA-N 5-methoxy-2-sulfanylphenol;zinc Chemical compound [Zn].COC1=CC=C(S)C(O)=C1 GXUMZMQSOBZWDA-UHFFFAOYSA-N 0.000 description 1
- SXLBJFAJTHUSQF-UHFFFAOYSA-N 5-methyl-2-sulfanylphenol Chemical compound CC1=CC=C(S)C(O)=C1 SXLBJFAJTHUSQF-UHFFFAOYSA-N 0.000 description 1
- NRADSYMFUFLAEM-UHFFFAOYSA-N 5-methyl-2-sulfanylphenol;sodium Chemical compound [Na].CC1=CC=C(S)C(O)=C1 NRADSYMFUFLAEM-UHFFFAOYSA-N 0.000 description 1
- JBQDGQWVSYPCHX-UHFFFAOYSA-N 5-methyl-2-sulfanylphenol;zinc Chemical compound [Zn].CC1=CC=C(S)C(O)=C1 JBQDGQWVSYPCHX-UHFFFAOYSA-N 0.000 description 1
- PMZBHPUNQNKBOA-UHFFFAOYSA-N 5-methylbenzene-1,3-dicarboxylic acid Chemical compound CC1=CC(C(O)=O)=CC(C(O)=O)=C1 PMZBHPUNQNKBOA-UHFFFAOYSA-N 0.000 description 1
- JGJSLSFWCUOXST-UHFFFAOYSA-N 5-methylsulfinyl-2-sulfanylphenol Chemical compound CS(=O)C1=CC=C(S)C(O)=C1 JGJSLSFWCUOXST-UHFFFAOYSA-N 0.000 description 1
- RPEXGWOXSLWJOF-UHFFFAOYSA-N 5-methylsulfinyl-2-sulfanylphenol;sodium Chemical compound [Na].CS(=O)C1=CC=C(S)C(O)=C1 RPEXGWOXSLWJOF-UHFFFAOYSA-N 0.000 description 1
- AMCXZGQJJOAVAX-UHFFFAOYSA-N 5-methylsulfinyl-2-sulfanylphenol;zinc Chemical compound [Zn].CS(=O)C1=CC=C(S)C(O)=C1 AMCXZGQJJOAVAX-UHFFFAOYSA-N 0.000 description 1
- MRDCTEVWKXFECQ-UHFFFAOYSA-N 6-[(2,3,4,5,6-pentacarbamoylphenyl)disulfanyl]benzene-1,2,3,4,5-pentacarboxamide Chemical group NC(=O)C1=C(C(N)=O)C(C(N)=O)=C(C(N)=O)C(C(=O)N)=C1SSC1=C(C(N)=O)C(C(N)=O)=C(C(N)=O)C(C(N)=O)=C1C(N)=O MRDCTEVWKXFECQ-UHFFFAOYSA-N 0.000 description 1
- SUFVSMZGKXXTDJ-UHFFFAOYSA-N 6-[(2,3,4,5,6-pentacarbonochloridoylphenyl)disulfanyl]benzene-1,2,3,4,5-pentacarbonyl chloride Chemical group ClC(=O)C1=C(C(Cl)=O)C(C(Cl)=O)=C(C(Cl)=O)C(C(=O)Cl)=C1SSC1=C(C(Cl)=O)C(C(Cl)=O)=C(C(Cl)=O)C(C(Cl)=O)=C1C(Cl)=O SUFVSMZGKXXTDJ-UHFFFAOYSA-N 0.000 description 1
- IFIPCXNVWVMXLE-UHFFFAOYSA-N 6-[(2,3,4,5,6-pentacarboxyphenyl)disulfanyl]benzene-1,2,3,4,5-pentacarboxylic acid Chemical group OC(=O)C1=C(C(O)=O)C(C(O)=O)=C(C(O)=O)C(C(=O)O)=C1SSC1=C(C(O)=O)C(C(O)=O)=C(C(O)=O)C(C(O)=O)=C1C(O)=O IFIPCXNVWVMXLE-UHFFFAOYSA-N 0.000 description 1
- UAUNCHRPXFMQIX-UHFFFAOYSA-N 6-[(2,3,4,5,6-pentacyanophenyl)disulfanyl]benzene-1,2,3,4,5-pentacarbonitrile Chemical compound N#CC1=C(C#N)C(C#N)=C(C#N)C(C#N)=C1SSC1=C(C#N)C(C#N)=C(C#N)C(C#N)=C1C#N UAUNCHRPXFMQIX-UHFFFAOYSA-N 0.000 description 1
- NLFZITCVCYRXLZ-UHFFFAOYSA-N 6-[(2,3,4,5,6-pentaformylphenyl)disulfanyl]benzene-1,2,3,4,5-pentacarbaldehyde Chemical group O=CC1=C(C=O)C(C=O)=C(C=O)C(C=O)=C1SSC1=C(C=O)C(C=O)=C(C=O)C(C=O)=C1C=O NLFZITCVCYRXLZ-UHFFFAOYSA-N 0.000 description 1
- PFEWZUBZJKZLEN-UHFFFAOYSA-N 6-[(2,3,4,5,6-pentasulfinophenyl)disulfanyl]benzene-1,2,3,4,5-pentasulfinic acid Chemical group OS(=O)C1=C(S(O)=O)C(S(O)=O)=C(S(O)=O)C(S(=O)O)=C1SSC1=C(S(O)=O)C(S(O)=O)=C(S(O)=O)C(S(O)=O)=C1S(O)=O PFEWZUBZJKZLEN-UHFFFAOYSA-N 0.000 description 1
- NJJZPLLVHQOCBG-UHFFFAOYSA-N 6-[(2,3,4,5,6-pentasulfophenyl)disulfanyl]benzene-1,2,3,4,5-pentasulfonic acid Chemical group OS(=O)(=O)C1=C(S(O)(=O)=O)C(S(O)(=O)=O)=C(S(O)(=O)=O)C(S(=O)(=O)O)=C1SSC1=C(S(O)(=O)=O)C(S(O)(=O)=O)=C(S(O)(=O)=O)C(S(O)(=O)=O)=C1S(O)(=O)=O NJJZPLLVHQOCBG-UHFFFAOYSA-N 0.000 description 1
- PHTMGFVJDOOOHZ-UHFFFAOYSA-N 6-[[2,3,4,5,6-pentakis(chlorosulfonyl)phenyl]disulfanyl]benzene-1,2,3,4,5-pentasulfonyl chloride Chemical group ClS(=O)(=O)C1=C(S(Cl)(=O)=O)C(S(Cl)(=O)=O)=C(S(Cl)(=O)=O)C(S(=O)(=O)Cl)=C1SSC1=C(S(Cl)(=O)=O)C(S(Cl)(=O)=O)=C(S(Cl)(=O)=O)C(S(Cl)(=O)=O)=C1S(Cl)(=O)=O PHTMGFVJDOOOHZ-UHFFFAOYSA-N 0.000 description 1
- BSFGCJHXMFCDDN-UHFFFAOYSA-N 6-sulfanylbenzene-1,2,3,4,5-pentacarbaldehyde Chemical compound SC1=C(C=O)C(C=O)=C(C=O)C(C=O)=C1C=O BSFGCJHXMFCDDN-UHFFFAOYSA-N 0.000 description 1
- GWHFITYLWCAUSH-UHFFFAOYSA-N 6-sulfanylbenzene-1,2,3,4,5-pentacarbaldehyde;zinc Chemical compound [Zn].SC1=C(C=O)C(C=O)=C(C=O)C(C=O)=C1C=O GWHFITYLWCAUSH-UHFFFAOYSA-N 0.000 description 1
- JLCVSIXQTSVBTB-UHFFFAOYSA-N 6-sulfanylbenzene-1,2,3,4,5-pentacarbonitrile Chemical compound SC1=C(C#N)C(C#N)=C(C#N)C(C#N)=C1C#N JLCVSIXQTSVBTB-UHFFFAOYSA-N 0.000 description 1
- XTOZUGGZVFZUCA-UHFFFAOYSA-N 6-sulfanylbenzene-1,2,3,4,5-pentacarbonitrile;zinc Chemical compound [Zn].SC1=C(C#N)C(C#N)=C(C#N)C(C#N)=C1C#N XTOZUGGZVFZUCA-UHFFFAOYSA-N 0.000 description 1
- WGRLZAAEBSGXNF-UHFFFAOYSA-N 6-sulfanylbenzene-1,2,3,4,5-pentacarbonyl chloride Chemical compound SC1=C(C(Cl)=O)C(C(Cl)=O)=C(C(Cl)=O)C(C(Cl)=O)=C1C(Cl)=O WGRLZAAEBSGXNF-UHFFFAOYSA-N 0.000 description 1
- ULRAGKHDVZFUJK-UHFFFAOYSA-N 6-sulfanylbenzene-1,2,3,4,5-pentacarbonyl chloride;zinc Chemical compound [Zn].SC1=C(C(Cl)=O)C(C(Cl)=O)=C(C(Cl)=O)C(C(Cl)=O)=C1C(Cl)=O ULRAGKHDVZFUJK-UHFFFAOYSA-N 0.000 description 1
- KYSQCLPBTSZQDR-UHFFFAOYSA-N 6-sulfanylbenzene-1,2,3,4,5-pentacarboxamide Chemical compound NC(=O)C1=C(S)C(C(N)=O)=C(C(N)=O)C(C(N)=O)=C1C(N)=O KYSQCLPBTSZQDR-UHFFFAOYSA-N 0.000 description 1
- WWXREJVZRIQWFK-UHFFFAOYSA-N 6-sulfanylbenzene-1,2,3,4,5-pentacarboxamide;zinc Chemical compound [Zn].NC(=O)C1=C(S)C(C(N)=O)=C(C(N)=O)C(C(N)=O)=C1C(N)=O WWXREJVZRIQWFK-UHFFFAOYSA-N 0.000 description 1
- AGFMICFENYRAMP-UHFFFAOYSA-N 6-sulfanylbenzene-1,2,3,4,5-pentacarboxylic acid Chemical compound OC(=O)C1=C(S)C(C(O)=O)=C(C(O)=O)C(C(O)=O)=C1C(O)=O AGFMICFENYRAMP-UHFFFAOYSA-N 0.000 description 1
- YBWIZBBFDKLMNU-UHFFFAOYSA-N 6-sulfanylbenzene-1,2,3,4,5-pentasulfinic acid Chemical compound OS(=O)C1=C(S)C(S(O)=O)=C(S(O)=O)C(S(O)=O)=C1S(O)=O YBWIZBBFDKLMNU-UHFFFAOYSA-N 0.000 description 1
- SYZNNQHXQIEALB-UHFFFAOYSA-N 6-sulfanylbenzene-1,2,3,4,5-pentasulfonic acid Chemical compound OS(=O)(=O)C1=C(S)C(S(O)(=O)=O)=C(S(O)(=O)=O)C(S(O)(=O)=O)=C1S(O)(=O)=O SYZNNQHXQIEALB-UHFFFAOYSA-N 0.000 description 1
- OXJHTSVVFOAACW-UHFFFAOYSA-N 6-sulfanylbenzene-1,2,3,4,5-pentasulfonyl chloride;zinc Chemical compound [Zn].SC1=C(S(Cl)(=O)=O)C(S(Cl)(=O)=O)=C(S(Cl)(=O)=O)C(S(Cl)(=O)=O)=C1S(Cl)(=O)=O OXJHTSVVFOAACW-UHFFFAOYSA-N 0.000 description 1
- YWWVWXASSLXJHU-UHFFFAOYSA-N 9E-tetradecenoic acid Natural products CCCCC=CCCCCCCCC(O)=O YWWVWXASSLXJHU-UHFFFAOYSA-N 0.000 description 1
- 239000005711 Benzoic acid Substances 0.000 description 1
- DPUOLQHDNGRHBS-UHFFFAOYSA-N Brassidinsaeure Natural products CCCCCCCCC=CCCCCCCCCCCCC(O)=O DPUOLQHDNGRHBS-UHFFFAOYSA-N 0.000 description 1
- XXQJEMXIBUTPMZ-UHFFFAOYSA-N C(=O)(O)C(=O)C=1C=C(C(=O)O)C=CC1.C(C(=O)C1=CC(C(=O)O)=CC=C1)(=O)O Chemical compound C(=O)(O)C(=O)C=1C=C(C(=O)O)C=CC1.C(C(=O)C1=CC(C(=O)O)=CC=C1)(=O)O XXQJEMXIBUTPMZ-UHFFFAOYSA-N 0.000 description 1
- NEJGZUZSXNYPDL-UHFFFAOYSA-N C(=O)(O)CC=1C=C(C(=O)O)C=CC1.C(CC1=CC(C(=O)O)=CC=C1)(=O)O Chemical compound C(=O)(O)CC=1C=C(C(=O)O)C=CC1.C(CC1=CC(C(=O)O)=CC=C1)(=O)O NEJGZUZSXNYPDL-UHFFFAOYSA-N 0.000 description 1
- BPMMUODPPJXZHI-UHFFFAOYSA-N C(CC1=CC(OC)=C(OC)C=C1)(=O)O.COC=1C=C(C=CC1OC)CC(=O)O Chemical compound C(CC1=CC(OC)=C(OC)C=C1)(=O)O.COC=1C=C(C=CC1OC)CC(=O)O BPMMUODPPJXZHI-UHFFFAOYSA-N 0.000 description 1
- FVYUAHFFHLFLDC-UHFFFAOYSA-N C1(=C(C(=CC(=C1)C(=O)O)C(=O)O)C(=O)O)C(=O)O.C(C1=C(C(=O)O)C(C(=O)O)=CC(C(=O)O)=C1)(=O)O Chemical compound C1(=C(C(=CC(=C1)C(=O)O)C(=O)O)C(=O)O)C(=O)O.C(C1=C(C(=O)O)C(C(=O)O)=CC(C(=O)O)=C1)(=O)O FVYUAHFFHLFLDC-UHFFFAOYSA-N 0.000 description 1
- NDIVWQXCPCHGPH-UHFFFAOYSA-N CC1=C(C(=O)O)C=C(C=C1C)C.C(C1=C(C)C(C)=CC(C)=C1)(=O)O Chemical compound CC1=C(C(=O)O)C=C(C=C1C)C.C(C1=C(C)C(C)=CC(C)=C1)(=O)O NDIVWQXCPCHGPH-UHFFFAOYSA-N 0.000 description 1
- IZANKHTWYLVBAI-UHFFFAOYSA-N CC1=CC=C(C(O)=O)C(O)=C1.CC1=CC=C(C(O)=O)C(O)=C1 Chemical compound CC1=CC=C(C(O)=O)C(O)=C1.CC1=CC=C(C(O)=O)C(O)=C1 IZANKHTWYLVBAI-UHFFFAOYSA-N 0.000 description 1
- DHXXBHKDZOKZMN-UHFFFAOYSA-N CC=1C=C(C(=O)O)C=C(C1C)C.C(C1=CC(C)=C(C)C(C)=C1)(=O)O Chemical compound CC=1C=C(C(=O)O)C=C(C1C)C.C(C1=CC(C)=C(C)C(C)=C1)(=O)O DHXXBHKDZOKZMN-UHFFFAOYSA-N 0.000 description 1
- OFQAMONEKGFYFG-UHFFFAOYSA-N COC1=C(C=CC=C1OC)CC(=O)O.C(CC1=C(OC)C(OC)=CC=C1)(=O)O Chemical compound COC1=C(C=CC=C1OC)CC(=O)O.C(CC1=C(OC)C(OC)=CC=C1)(=O)O OFQAMONEKGFYFG-UHFFFAOYSA-N 0.000 description 1
- ZILVNTITQOYDGD-UHFFFAOYSA-N COC1=CC=C(CC(O)=O)C=C1.COC1=CC=C(CC(O)=O)C=C1 Chemical compound COC1=CC=C(CC(O)=O)C=C1.COC1=CC=C(CC(O)=O)C=C1 ZILVNTITQOYDGD-UHFFFAOYSA-N 0.000 description 1
- RLACXPZOJHROHB-UHFFFAOYSA-N COC=1C=C(C(C(=O)O)=CC1OC)C(=O)O.C(C=1C(C(=O)O)=CC(OC)=C(OC)C1)(=O)O Chemical compound COC=1C=C(C(C(=O)O)=CC1OC)C(=O)O.C(C=1C(C(=O)O)=CC(OC)=C(OC)C1)(=O)O RLACXPZOJHROHB-UHFFFAOYSA-N 0.000 description 1
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 1
- 239000005632 Capric acid (CAS 334-48-5) Substances 0.000 description 1
- JJLJMEJHUUYSSY-UHFFFAOYSA-L Copper hydroxide Chemical compound [OH-].[OH-].[Cu+2] JJLJMEJHUUYSSY-UHFFFAOYSA-L 0.000 description 1
- 239000005750 Copper hydroxide Substances 0.000 description 1
- QPLDLSVMHZLSFG-UHFFFAOYSA-N Copper oxide Chemical compound [Cu]=O QPLDLSVMHZLSFG-UHFFFAOYSA-N 0.000 description 1
- 239000005751 Copper oxide Substances 0.000 description 1
- VMQMZMRVKUZKQL-UHFFFAOYSA-N Cu+ Chemical compound [Cu+] VMQMZMRVKUZKQL-UHFFFAOYSA-N 0.000 description 1
- JPVYNHNXODAKFH-UHFFFAOYSA-N Cu2+ Chemical compound [Cu+2] JPVYNHNXODAKFH-UHFFFAOYSA-N 0.000 description 1
- URXZXNYJPAJJOQ-UHFFFAOYSA-N Erucic acid Natural products CCCCCCC=CCCCCCCCCCCCC(O)=O URXZXNYJPAJJOQ-UHFFFAOYSA-N 0.000 description 1
- CWYNVVGOOAEACU-UHFFFAOYSA-N Fe2+ Chemical compound [Fe+2] CWYNVVGOOAEACU-UHFFFAOYSA-N 0.000 description 1
- 244000043261 Hevea brasiliensis Species 0.000 description 1
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 1
- 235000021353 Lignoceric acid Nutrition 0.000 description 1
- CQXMAMUUWHYSIY-UHFFFAOYSA-N Lignoceric acid Natural products CCCCCCCCCCCCCCCCCCCCCCCC(=O)OCCC1=CC=C(O)C=C1 CQXMAMUUWHYSIY-UHFFFAOYSA-N 0.000 description 1
- OYHQOLUKZRVURQ-HZJYTTRNSA-N Linoleic acid Chemical compound CCCCC\C=C/C\C=C/CCCCCCCC(O)=O OYHQOLUKZRVURQ-HZJYTTRNSA-N 0.000 description 1
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 description 1
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 1
- 229910052779 Neodymium Inorganic materials 0.000 description 1
- XJXROGWVRIJYMO-SJDLZYGOSA-N Nervonic acid Natural products O=C(O)[C@@H](/C=C/CCCCCCCC)CCCCCCCCCCCC XJXROGWVRIJYMO-SJDLZYGOSA-N 0.000 description 1
- PXHVJJICTQNCMI-UHFFFAOYSA-N Nickel Chemical compound [Ni] PXHVJJICTQNCMI-UHFFFAOYSA-N 0.000 description 1
- VEQPNABPJHWNSG-UHFFFAOYSA-N Nickel(2+) Chemical compound [Ni+2] VEQPNABPJHWNSG-UHFFFAOYSA-N 0.000 description 1
- RINSJDWDXSWUOK-UHFFFAOYSA-N OC(=O)C1=CC=C(O)C(O)=C1.OC(=O)C1=CC=C(O)C(O)=C1 Chemical compound OC(=O)C1=CC=C(O)C(O)=C1.OC(=O)C1=CC=C(O)C(O)=C1 RINSJDWDXSWUOK-UHFFFAOYSA-N 0.000 description 1
- QKTAFNAZTCSZGF-UHFFFAOYSA-N OC(=O)C1=CC=C(O)C=C1O.OC(=O)C1=CC=C(O)C=C1O Chemical compound OC(=O)C1=CC=C(O)C=C1O.OC(=O)C1=CC=C(O)C=C1O QKTAFNAZTCSZGF-UHFFFAOYSA-N 0.000 description 1
- UTXFGSMATYQXBX-UHFFFAOYSA-N OC(=O)C1=CC=CC(O)=C1O.OC(=O)C1=CC=CC(O)=C1O Chemical compound OC(=O)C1=CC=CC(O)=C1O.OC(=O)C1=CC=CC(O)=C1O UTXFGSMATYQXBX-UHFFFAOYSA-N 0.000 description 1
- MMOPREBWKCZVPO-ZIKNSQGESA-N OC(=O)C=CC1=CC=C(O)C=C1.OC(=O)\C=C\C1=CC=C(O)C=C1 Chemical compound OC(=O)C=CC1=CC=C(O)C=C1.OC(=O)\C=C\C1=CC=C(O)C=C1 MMOPREBWKCZVPO-ZIKNSQGESA-N 0.000 description 1
- GTCZNPJCKDRXJR-UHFFFAOYSA-N OC(=O)CC1=CC=C(O)C(O)=C1.OC(=O)CC1=CC=C(O)C(O)=C1 Chemical compound OC(=O)CC1=CC=C(O)C(O)=C1.OC(=O)CC1=CC=C(O)C(O)=C1 GTCZNPJCKDRXJR-UHFFFAOYSA-N 0.000 description 1
- GYQGALAEKFCZNA-UHFFFAOYSA-N OC=1C=C(C=CC1OC)C=CC(=O)O.C(C=CC1=CC(O)=C(OC)C=C1)(=O)O Chemical compound OC=1C=C(C=CC1OC)C=CC(=O)O.C(C=CC1=CC(O)=C(OC)C=C1)(=O)O GYQGALAEKFCZNA-UHFFFAOYSA-N 0.000 description 1
- 235000021319 Palmitoleic acid Nutrition 0.000 description 1
- 239000005643 Pelargonic acid Substances 0.000 description 1
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 1
- OFOBLEOULBTSOW-UHFFFAOYSA-N Propanedioic acid Natural products OC(=O)CC(O)=O OFOBLEOULBTSOW-UHFFFAOYSA-N 0.000 description 1
- 229910006080 SO2X Inorganic materials 0.000 description 1
- 229910006069 SO3H Inorganic materials 0.000 description 1
- FZWLAAWBMGSTSO-UHFFFAOYSA-N Thiazole Chemical compound C1=CSC=N1 FZWLAAWBMGSTSO-UHFFFAOYSA-N 0.000 description 1
- ATJFFYVFTNAWJD-UHFFFAOYSA-N Tin Chemical compound [Sn] ATJFFYVFTNAWJD-UHFFFAOYSA-N 0.000 description 1
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 description 1
- LCKIEQZJEYYRIY-UHFFFAOYSA-N Titanium ion Chemical compound [Ti+4] LCKIEQZJEYYRIY-UHFFFAOYSA-N 0.000 description 1
- JACRWUWPXAESPB-QMMMGPOBSA-N Tropic acid Natural products OC[C@H](C(O)=O)C1=CC=CC=C1 JACRWUWPXAESPB-QMMMGPOBSA-N 0.000 description 1
- 235000021322 Vaccenic acid Nutrition 0.000 description 1
- UWHZIFQPPBDJPM-FPLPWBNLSA-M Vaccenic acid Natural products CCCCCC\C=C/CCCCCCCCCC([O-])=O UWHZIFQPPBDJPM-FPLPWBNLSA-M 0.000 description 1
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical compound [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 description 1
- FMRLDPWIRHBCCC-UHFFFAOYSA-L Zinc carbonate Chemical compound [Zn+2].[O-]C([O-])=O FMRLDPWIRHBCCC-UHFFFAOYSA-L 0.000 description 1
- QCWXUUIWCKQGHC-UHFFFAOYSA-N Zirconium Chemical compound [Zr] QCWXUUIWCKQGHC-UHFFFAOYSA-N 0.000 description 1
- NBXJFTVWDVBWHJ-UHFFFAOYSA-I [Na+].[Na+].[Na+].[Na+].[Na+].[O-]C(=O)c1c(S)c(C([O-])=O)c(C([O-])=O)c(C([O-])=O)c1C([O-])=O Chemical compound [Na+].[Na+].[Na+].[Na+].[Na+].[O-]C(=O)c1c(S)c(C([O-])=O)c(C([O-])=O)c(C([O-])=O)c1C([O-])=O NBXJFTVWDVBWHJ-UHFFFAOYSA-I 0.000 description 1
- FXRDKIFQJPGWIW-UHFFFAOYSA-K [Na+].[Na+].[Na+].[O-]S(=O)(=O)c1cc(c(S)c(c1)S([O-])(=O)=O)S([O-])(=O)=O Chemical compound [Na+].[Na+].[Na+].[O-]S(=O)(=O)c1cc(c(S)c(c1)S([O-])(=O)=O)S([O-])(=O)=O FXRDKIFQJPGWIW-UHFFFAOYSA-K 0.000 description 1
- GDDLDAOOOIUCNV-UHFFFAOYSA-N [Na].ClS(=O)(=O)C1=C(C=CC=C1)S Chemical compound [Na].ClS(=O)(=O)C1=C(C=CC=C1)S GDDLDAOOOIUCNV-UHFFFAOYSA-N 0.000 description 1
- CRIAGMLCPNTWPI-UHFFFAOYSA-N [Na].Sc1ccccc1C(Cl)=O Chemical compound [Na].Sc1ccccc1C(Cl)=O CRIAGMLCPNTWPI-UHFFFAOYSA-N 0.000 description 1
- TUMSDSTZVRGYST-UHFFFAOYSA-D [Zn++].[Zn++].[Zn++].[Zn++].[Zn++].[O-]C(=O)c1c(S)c(C([O-])=O)c(C([O-])=O)c(C([O-])=O)c1C([O-])=O.[O-]C(=O)c1c(S)c(C([O-])=O)c(C([O-])=O)c(C([O-])=O)c1C([O-])=O Chemical compound [Zn++].[Zn++].[Zn++].[Zn++].[Zn++].[O-]C(=O)c1c(S)c(C([O-])=O)c(C([O-])=O)c(C([O-])=O)c1C([O-])=O.[O-]C(=O)c1c(S)c(C([O-])=O)c(C([O-])=O)c(C([O-])=O)c1C([O-])=O TUMSDSTZVRGYST-UHFFFAOYSA-D 0.000 description 1
- UQWLKSSDZSOEDW-UHFFFAOYSA-D [Zn+2].S(=O)(=O)([O-])C1=C(C(=C(C(=C1S)S(=O)(=O)[O-])S(=O)(=O)[O-])S(=O)(=O)[O-])S(=O)(=O)[O-].S(=O)(=O)([O-])C1=C(C(=C(C(=C1S)S(=O)(=O)[O-])S(=O)(=O)[O-])S(=O)(=O)[O-])S(=O)(=O)[O-].[Zn+2].[Zn+2].[Zn+2].[Zn+2] Chemical compound [Zn+2].S(=O)(=O)([O-])C1=C(C(=C(C(=C1S)S(=O)(=O)[O-])S(=O)(=O)[O-])S(=O)(=O)[O-])S(=O)(=O)[O-].S(=O)(=O)([O-])C1=C(C(=C(C(=C1S)S(=O)(=O)[O-])S(=O)(=O)[O-])S(=O)(=O)[O-])S(=O)(=O)[O-].[Zn+2].[Zn+2].[Zn+2].[Zn+2] UQWLKSSDZSOEDW-UHFFFAOYSA-D 0.000 description 1
- 150000001334 alicyclic compounds Chemical class 0.000 description 1
- 125000002723 alicyclic group Chemical group 0.000 description 1
- 150000007824 aliphatic compounds Chemical class 0.000 description 1
- 125000001931 aliphatic group Chemical group 0.000 description 1
- DTOSIQBPPRVQHS-PDBXOOCHSA-N alpha-linolenic acid Chemical compound CC\C=C/C\C=C/C\C=C/CCCCCCCC(O)=O DTOSIQBPPRVQHS-PDBXOOCHSA-N 0.000 description 1
- 235000020661 alpha-linolenic acid Nutrition 0.000 description 1
- 229940114079 arachidonic acid Drugs 0.000 description 1
- 235000021342 arachidonic acid Nutrition 0.000 description 1
- 150000001491 aromatic compounds Chemical class 0.000 description 1
- 125000003118 aryl group Chemical group 0.000 description 1
- 229910052788 barium Inorganic materials 0.000 description 1
- DSAJWYNOEDNPEQ-UHFFFAOYSA-N barium atom Chemical compound [Ba] DSAJWYNOEDNPEQ-UHFFFAOYSA-N 0.000 description 1
- ZOQOMVWXXWHKGT-UHFFFAOYSA-N benzene-1,3,5-tricarboxylic acid Chemical compound OC(=O)C1=CC(C(O)=O)=CC(C(O)=O)=C1.OC(=O)C1=CC(C(O)=O)=CC(C(O)=O)=C1 ZOQOMVWXXWHKGT-UHFFFAOYSA-N 0.000 description 1
- 235000010233 benzoic acid Nutrition 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 229910052794 bromium Inorganic materials 0.000 description 1
- 235000004883 caffeic acid Nutrition 0.000 description 1
- 229940074360 caffeic acid Drugs 0.000 description 1
- 229910052791 calcium Inorganic materials 0.000 description 1
- 239000011575 calcium Substances 0.000 description 1
- AXCZMVOFGPJBDE-UHFFFAOYSA-L calcium dihydroxide Chemical compound [OH-].[OH-].[Ca+2] AXCZMVOFGPJBDE-UHFFFAOYSA-L 0.000 description 1
- 239000000920 calcium hydroxide Substances 0.000 description 1
- 229910001861 calcium hydroxide Inorganic materials 0.000 description 1
- BRPQOXSCLDDYGP-UHFFFAOYSA-N calcium oxide Chemical compound [O-2].[Ca+2] BRPQOXSCLDDYGP-UHFFFAOYSA-N 0.000 description 1
- 239000000292 calcium oxide Substances 0.000 description 1
- ODINCKMPIJJUCX-UHFFFAOYSA-N calcium oxide Inorganic materials [Ca]=O ODINCKMPIJJUCX-UHFFFAOYSA-N 0.000 description 1
- 150000004649 carbonic acid derivatives Chemical class 0.000 description 1
- 125000002091 cationic group Chemical group 0.000 description 1
- 229910052801 chlorine Inorganic materials 0.000 description 1
- QAIPRVGONGVQAS-UHFFFAOYSA-N cis-caffeic acid Natural products OC(=O)C=CC1=CC=C(O)C(O)=C1 QAIPRVGONGVQAS-UHFFFAOYSA-N 0.000 description 1
- SECPZKHBENQXJG-UHFFFAOYSA-N cis-palmitoleic acid Natural products CCCCCCC=CCCCCCCCC(O)=O SECPZKHBENQXJG-UHFFFAOYSA-N 0.000 description 1
- GWHCXVQVJPWHRF-UHFFFAOYSA-N cis-tetracosenoic acid Natural products CCCCCCCCC=CCCCCCCCCCCCCCC(O)=O GWHCXVQVJPWHRF-UHFFFAOYSA-N 0.000 description 1
- 229910001429 cobalt ion Inorganic materials 0.000 description 1
- 229910001956 copper hydroxide Inorganic materials 0.000 description 1
- 229910001431 copper ion Inorganic materials 0.000 description 1
- 229910000431 copper oxide Inorganic materials 0.000 description 1
- ZOUQIAGHKFLHIA-UHFFFAOYSA-L copper;n,n-dimethylcarbamodithioate Chemical compound [Cu+2].CN(C)C([S-])=S.CN(C)C([S-])=S ZOUQIAGHKFLHIA-UHFFFAOYSA-L 0.000 description 1
- LDHQCZJRKDOVOX-NSCUHMNNSA-N crotonic acid Chemical compound C\C=C\C(O)=O LDHQCZJRKDOVOX-NSCUHMNNSA-N 0.000 description 1
- 150000003946 cyclohexylamines Chemical class 0.000 description 1
- LSXWFXONGKSEMY-UHFFFAOYSA-N di-tert-butyl peroxide Chemical compound CC(C)(C)OOC(C)(C)C LSXWFXONGKSEMY-UHFFFAOYSA-N 0.000 description 1
- PGAXJQVAHDTGBB-UHFFFAOYSA-N dibutylcarbamothioylsulfanyl n,n-dibutylcarbamodithioate Chemical compound CCCCN(CCCC)C(=S)SSC(=S)N(CCCC)CCCC PGAXJQVAHDTGBB-UHFFFAOYSA-N 0.000 description 1
- RLULIUSIDLLCSW-UHFFFAOYSA-N diethylcarbamothioylsulfanylselanyl n,n-diethylcarbamodithioate Chemical compound CCN(CC)C(=S)S[Se]SC(=S)N(CC)CC RLULIUSIDLLCSW-UHFFFAOYSA-N 0.000 description 1
- 229940116901 diethyldithiocarbamate Drugs 0.000 description 1
- DZAUWHJDUNRCTF-UHFFFAOYSA-N dihydrocaffeic acid Natural products OC(=O)CCC1=CC=C(O)C(O)=C1 DZAUWHJDUNRCTF-UHFFFAOYSA-N 0.000 description 1
- 150000002023 dithiocarboxylic acids Chemical class 0.000 description 1
- 229950004394 ditiocarb Drugs 0.000 description 1
- 229940108623 eicosenoic acid Drugs 0.000 description 1
- BITHHVVYSMSWAG-UHFFFAOYSA-N eicosenoic acid Natural products CCCCCCCCC=CCCCCCCCCCC(O)=O BITHHVVYSMSWAG-UHFFFAOYSA-N 0.000 description 1
- DPUOLQHDNGRHBS-KTKRTIGZSA-N erucic acid Chemical compound CCCCCCCC\C=C/CCCCCCCCCCCC(O)=O DPUOLQHDNGRHBS-KTKRTIGZSA-N 0.000 description 1
- FARYTWBWLZAXNK-WAYWQWQTSA-N ethyl (z)-3-(methylamino)but-2-enoate Chemical compound CCOC(=O)\C=C(\C)NC FARYTWBWLZAXNK-WAYWQWQTSA-N 0.000 description 1
- 230000002349 favourable effect Effects 0.000 description 1
- WHDGWKAJBYRJJL-UHFFFAOYSA-K ferbam Chemical compound [Fe+3].CN(C)C([S-])=S.CN(C)C([S-])=S.CN(C)C([S-])=S WHDGWKAJBYRJJL-UHFFFAOYSA-K 0.000 description 1
- 235000001785 ferulic acid Nutrition 0.000 description 1
- 229940114124 ferulic acid Drugs 0.000 description 1
- 229910052731 fluorine Inorganic materials 0.000 description 1
- 239000001530 fumaric acid Substances 0.000 description 1
- LQJBNNIYVWPHFW-QXMHVHEDSA-N gadoleic acid Chemical compound CCCCCCCCCC\C=C/CCCCCCCC(O)=O LQJBNNIYVWPHFW-QXMHVHEDSA-N 0.000 description 1
- 229940074391 gallic acid Drugs 0.000 description 1
- 235000004515 gallic acid Nutrition 0.000 description 1
- 238000005227 gel permeation chromatography Methods 0.000 description 1
- 150000002391 heterocyclic compounds Chemical class 0.000 description 1
- 229910052740 iodine Inorganic materials 0.000 description 1
- 229960004232 linoleic acid Drugs 0.000 description 1
- 229960004488 linolenic acid Drugs 0.000 description 1
- KQQKGWQCNNTQJW-UHFFFAOYSA-N linolenic acid Natural products CC=CCCC=CCC=CCCCCCCCC(O)=O KQQKGWQCNNTQJW-UHFFFAOYSA-N 0.000 description 1
- 229910052744 lithium Inorganic materials 0.000 description 1
- XGZVUEUWXADBQD-UHFFFAOYSA-L lithium carbonate Chemical compound [Li+].[Li+].[O-]C([O-])=O XGZVUEUWXADBQD-UHFFFAOYSA-L 0.000 description 1
- 229910052808 lithium carbonate Inorganic materials 0.000 description 1
- 229910052749 magnesium Inorganic materials 0.000 description 1
- 239000011777 magnesium Substances 0.000 description 1
- VTHJTEIRLNZDEV-UHFFFAOYSA-L magnesium dihydroxide Chemical compound [OH-].[OH-].[Mg+2] VTHJTEIRLNZDEV-UHFFFAOYSA-L 0.000 description 1
- 239000000347 magnesium hydroxide Substances 0.000 description 1
- 229910001862 magnesium hydroxide Inorganic materials 0.000 description 1
- 239000000395 magnesium oxide Substances 0.000 description 1
- CPLXHLVBOLITMK-UHFFFAOYSA-N magnesium oxide Inorganic materials [Mg]=O CPLXHLVBOLITMK-UHFFFAOYSA-N 0.000 description 1
- AXZKOIWUVFPNLO-UHFFFAOYSA-N magnesium;oxygen(2-) Chemical compound [O-2].[Mg+2] AXZKOIWUVFPNLO-UHFFFAOYSA-N 0.000 description 1
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 description 1
- 239000011976 maleic acid Substances 0.000 description 1
- 229960002510 mandelic acid Drugs 0.000 description 1
- 229910001437 manganese ion Inorganic materials 0.000 description 1
- 229910000000 metal hydroxide Inorganic materials 0.000 description 1
- 150000004692 metal hydroxides Chemical class 0.000 description 1
- 229910044991 metal oxide Inorganic materials 0.000 description 1
- 150000004706 metal oxides Chemical class 0.000 description 1
- KWBUESBCUXDZMO-UHFFFAOYSA-N methyl 3-amino-4-[(2-amino-4-methoxycarbonylphenyl)disulfanyl]benzoate Chemical compound NC1=CC(C(=O)OC)=CC=C1SSC1=CC=C(C(=O)OC)C=C1N KWBUESBCUXDZMO-UHFFFAOYSA-N 0.000 description 1
- PEGINHQWTXGQSG-UHFFFAOYSA-N methyl 3-amino-4-[(2-amino-4-methoxysulfonylphenyl)disulfanyl]benzenesulfonate Chemical compound NC1=CC(S(=O)(=O)OC)=CC=C1SSC1=CC=C(S(=O)(=O)OC)C=C1N PEGINHQWTXGQSG-UHFFFAOYSA-N 0.000 description 1
- SYXYKQBUJIDNFM-UHFFFAOYSA-N methyl 3-amino-4-sulfanylbenzenesulfonate Chemical compound COS(=O)(=O)C1=CC=C(S)C(N)=C1 SYXYKQBUJIDNFM-UHFFFAOYSA-N 0.000 description 1
- UQLSATXYXXVFKB-UHFFFAOYSA-N methyl 3-amino-4-sulfanylbenzenesulfonate;sodium Chemical compound [Na].COS(=O)(=O)C1=CC=C(S)C(N)=C1 UQLSATXYXXVFKB-UHFFFAOYSA-N 0.000 description 1
- ZYENXZKYFUMRNC-UHFFFAOYSA-N methyl 3-amino-4-sulfanylbenzenesulfonate;zinc Chemical compound [Zn].COS(=O)(=O)C1=CC=C(S)C(N)=C1 ZYENXZKYFUMRNC-UHFFFAOYSA-N 0.000 description 1
- ZCKPTTBRWYHPOT-UHFFFAOYSA-N methyl 3-amino-4-sulfanylbenzoate Chemical compound COC(=O)C1=CC=C(S)C(N)=C1 ZCKPTTBRWYHPOT-UHFFFAOYSA-N 0.000 description 1
- MTENGGQWYCLEOD-UHFFFAOYSA-N methyl 3-amino-4-sulfanylbenzoate;sodium Chemical compound [Na].COC(=O)C1=CC=C(S)C(N)=C1 MTENGGQWYCLEOD-UHFFFAOYSA-N 0.000 description 1
- ZBGHIROYSFKLRP-UHFFFAOYSA-N methyl 3-amino-4-sulfanylbenzoate;zinc Chemical compound [Zn].COC(=O)C1=CC=C(S)C(N)=C1 ZBGHIROYSFKLRP-UHFFFAOYSA-N 0.000 description 1
- CLCCIBBXEKSWKP-UHFFFAOYSA-N methyl 3-hydroxy-4-[(2-hydroxy-4-methoxycarbonylphenyl)disulfanyl]benzoate Chemical compound OC1=CC(C(=O)OC)=CC=C1SSC1=CC=C(C(=O)OC)C=C1O CLCCIBBXEKSWKP-UHFFFAOYSA-N 0.000 description 1
- HLOQJRAVUVNCNK-UHFFFAOYSA-N methyl 3-hydroxy-4-[(2-hydroxy-4-methoxysulfonylphenyl)disulfanyl]benzenesulfonate Chemical compound OC1=CC(S(=O)(=O)OC)=CC=C1SSC1=CC=C(S(=O)(=O)OC)C=C1O HLOQJRAVUVNCNK-UHFFFAOYSA-N 0.000 description 1
- CXYRBYFUDGMNSE-UHFFFAOYSA-N methyl 3-hydroxy-4-sulfanylbenzenesulfonate Chemical compound COS(=O)(=O)C1=CC=C(S)C(O)=C1 CXYRBYFUDGMNSE-UHFFFAOYSA-N 0.000 description 1
- NXJMOBSYBJIFCF-UHFFFAOYSA-N methyl 3-hydroxy-4-sulfanylbenzenesulfonate;sodium Chemical compound [Na].COS(=O)(=O)C1=CC=C(S)C(O)=C1 NXJMOBSYBJIFCF-UHFFFAOYSA-N 0.000 description 1
- JKJZUNMWHVGBOK-UHFFFAOYSA-N methyl 3-hydroxy-4-sulfanylbenzenesulfonate;zinc Chemical compound [Zn].COS(=O)(=O)C1=CC=C(S)C(O)=C1 JKJZUNMWHVGBOK-UHFFFAOYSA-N 0.000 description 1
- KPMLVEPVDPOSIO-UHFFFAOYSA-N methyl 3-hydroxy-4-sulfanylbenzoate Chemical compound COC(=O)C1=CC=C(S)C(O)=C1 KPMLVEPVDPOSIO-UHFFFAOYSA-N 0.000 description 1
- BHJPQLUVWCCPQH-UHFFFAOYSA-N methyl 3-hydroxy-4-sulfanylbenzoate;sodium Chemical compound [Na].COC(=O)C1=CC=C(S)C(O)=C1 BHJPQLUVWCCPQH-UHFFFAOYSA-N 0.000 description 1
- GPKITENCWTZZRH-UHFFFAOYSA-N methyl 3-hydroxy-4-sulfanylbenzoate;zinc Chemical compound [Zn].COC(=O)C1=CC=C(S)C(O)=C1 GPKITENCWTZZRH-UHFFFAOYSA-N 0.000 description 1
- LAQGDMTYADYTBP-UHFFFAOYSA-N methyl 3-nitro-4-sulfanylbenzenesulfonate Chemical compound COS(=O)(=O)C1=CC=C(S)C([N+]([O-])=O)=C1 LAQGDMTYADYTBP-UHFFFAOYSA-N 0.000 description 1
- LRSXVXSTQDXNHW-UHFFFAOYSA-N methyl 3-nitro-4-sulfanylbenzenesulfonate;sodium Chemical compound [Na].COS(=O)(=O)C1=CC=C(S)C([N+]([O-])=O)=C1 LRSXVXSTQDXNHW-UHFFFAOYSA-N 0.000 description 1
- RVEISEAIIOAGTK-UHFFFAOYSA-N methyl 3-nitro-4-sulfanylbenzenesulfonate;zinc Chemical compound [Zn].COS(=O)(=O)C1=CC=C(S)C([N+]([O-])=O)=C1 RVEISEAIIOAGTK-UHFFFAOYSA-N 0.000 description 1
- JPDSHWJXMMBSTD-UHFFFAOYSA-N methyl 3-nitro-4-sulfanylbenzoate Chemical compound COC(=O)C1=CC=C(S)C([N+]([O-])=O)=C1 JPDSHWJXMMBSTD-UHFFFAOYSA-N 0.000 description 1
- CLUKEEOSFIRSJK-UHFFFAOYSA-N methyl 3-nitro-4-sulfanylbenzoate;sodium Chemical compound [Na].COC(=O)C1=CC=C(S)C([N+]([O-])=O)=C1 CLUKEEOSFIRSJK-UHFFFAOYSA-N 0.000 description 1
- RCPKZPRNJFCDJR-UHFFFAOYSA-N methyl 3-nitro-4-sulfanylbenzoate;zinc Chemical compound [Zn].COC(=O)C1=CC=C(S)C([N+]([O-])=O)=C1 RCPKZPRNJFCDJR-UHFFFAOYSA-N 0.000 description 1
- RSVYCAVGJWRYNO-UHFFFAOYSA-N methyl 3-phenylsulfanyl-4-sulfanylbenzenesulfonate Chemical compound COS(=O)(=O)C1=CC=C(S)C(SC=2C=CC=CC=2)=C1 RSVYCAVGJWRYNO-UHFFFAOYSA-N 0.000 description 1
- UVDMTLNLOADPES-UHFFFAOYSA-N methyl 3-phenylsulfanyl-4-sulfanylbenzenesulfonate;sodium Chemical compound [Na].COS(=O)(=O)C1=CC=C(S)C(SC=2C=CC=CC=2)=C1 UVDMTLNLOADPES-UHFFFAOYSA-N 0.000 description 1
- QDGCLHXEVMXGHU-UHFFFAOYSA-N methyl 3-phenylsulfanyl-4-sulfanylbenzenesulfonate;zinc Chemical compound [Zn].COS(=O)(=O)C1=CC=C(S)C(SC=2C=CC=CC=2)=C1 QDGCLHXEVMXGHU-UHFFFAOYSA-N 0.000 description 1
- RQJHACAIDWFERS-UHFFFAOYSA-N methyl 3-phenylsulfanyl-4-sulfanylbenzoate Chemical compound COC(=O)C1=CC=C(S)C(SC=2C=CC=CC=2)=C1 RQJHACAIDWFERS-UHFFFAOYSA-N 0.000 description 1
- SXGPYJDSFIJUBT-UHFFFAOYSA-N methyl 3-phenylsulfanyl-4-sulfanylbenzoate;sodium Chemical compound [Na].COC(=O)C1=CC=C(S)C(SC=2C=CC=CC=2)=C1 SXGPYJDSFIJUBT-UHFFFAOYSA-N 0.000 description 1
- FDBGLAQZNSSRPF-UHFFFAOYSA-N methyl 3-phenylsulfanyl-4-sulfanylbenzoate;zinc Chemical compound [Zn].COC(=O)C1=CC=C(S)C(SC=2C=CC=CC=2)=C1 FDBGLAQZNSSRPF-UHFFFAOYSA-N 0.000 description 1
- UYYXQLZKGJWXIQ-UHFFFAOYSA-N methyl 4-[(4-methoxycarbonyl-2-nitrophenyl)disulfanyl]-3-nitrobenzoate Chemical compound [O-][N+](=O)C1=CC(C(=O)OC)=CC=C1SSC1=CC=C(C(=O)OC)C=C1[N+]([O-])=O UYYXQLZKGJWXIQ-UHFFFAOYSA-N 0.000 description 1
- XOYNQYXKFLLVBT-UHFFFAOYSA-N methyl 4-[(4-methoxycarbonyl-2-phenylsulfanylphenyl)disulfanyl]-3-phenylsulfanylbenzoate Chemical compound C=1C=CC=CC=1SC1=CC(C(=O)OC)=CC=C1SSC1=CC=C(C(=O)OC)C=C1SC1=CC=CC=C1 XOYNQYXKFLLVBT-UHFFFAOYSA-N 0.000 description 1
- VGCZYLQMVYJBNY-UHFFFAOYSA-N methyl 4-[(4-methoxycarbonylphenyl)disulfanyl]benzoate Chemical group C1=CC(C(=O)OC)=CC=C1SSC1=CC=C(C(=O)OC)C=C1 VGCZYLQMVYJBNY-UHFFFAOYSA-N 0.000 description 1
- IKASTZLULPJBCM-UHFFFAOYSA-N methyl 4-[(4-methoxysulfonyl-2-nitrophenyl)disulfanyl]-3-nitrobenzenesulfonate Chemical compound [O-][N+](=O)C1=CC(S(=O)(=O)OC)=CC=C1SSC1=CC=C(S(=O)(=O)OC)C=C1[N+]([O-])=O IKASTZLULPJBCM-UHFFFAOYSA-N 0.000 description 1
- WSZRIGPYHURCMA-UHFFFAOYSA-N methyl 4-[(4-methoxysulfonyl-2-phenylsulfanylphenyl)disulfanyl]-3-phenylsulfanylbenzenesulfonate Chemical compound C=1C=CC=CC=1SC1=CC(S(=O)(=O)OC)=CC=C1SSC1=CC=C(S(=O)(=O)OC)C=C1SC1=CC=CC=C1 WSZRIGPYHURCMA-UHFFFAOYSA-N 0.000 description 1
- GMXOHOVQWSZIJK-UHFFFAOYSA-N methyl 4-[(4-methoxysulfonylphenyl)disulfanyl]benzenesulfonate Chemical group C1=CC(S(=O)(=O)OC)=CC=C1SSC1=CC=C(S(=O)(=O)OC)C=C1 GMXOHOVQWSZIJK-UHFFFAOYSA-N 0.000 description 1
- WEIPRCBTQPXBTJ-UHFFFAOYSA-N methyl 4-sulfanylbenzenesulfonate Chemical compound COS(=O)(=O)C1=CC=C(S)C=C1 WEIPRCBTQPXBTJ-UHFFFAOYSA-N 0.000 description 1
- FJYSXJNBEHCFTN-UHFFFAOYSA-N methyl 4-sulfanylbenzenesulfonate;sodium Chemical compound [Na].COS(=O)(=O)C1=CC=C(S)C=C1 FJYSXJNBEHCFTN-UHFFFAOYSA-N 0.000 description 1
- SLWJNWACJRQYKC-UHFFFAOYSA-N methyl 4-sulfanylbenzenesulfonate;zinc Chemical compound [Zn].COS(=O)(=O)C1=CC=C(S)C=C1 SLWJNWACJRQYKC-UHFFFAOYSA-N 0.000 description 1
- BTZOMWXSWVOOHG-UHFFFAOYSA-N methyl 4-sulfanylbenzoate Chemical compound COC(=O)C1=CC=C(S)C=C1 BTZOMWXSWVOOHG-UHFFFAOYSA-N 0.000 description 1
- KOOTVGNVUUDNCB-UHFFFAOYSA-N methyl 4-sulfanylbenzoate;sodium Chemical compound [Na].COC(=O)C1=CC=C(S)C=C1 KOOTVGNVUUDNCB-UHFFFAOYSA-N 0.000 description 1
- RLWJMJKAHQFUNE-UHFFFAOYSA-N methyl 4-sulfanylbenzoate;zinc Chemical compound [Zn].COC(=O)C1=CC=C(S)C=C1 RLWJMJKAHQFUNE-UHFFFAOYSA-N 0.000 description 1
- OXLSBJSNTAOWCI-UHFFFAOYSA-N methyl 5-acetyl-2-[(4-acetyl-2-methoxycarbonylphenyl)disulfanyl]benzoate Chemical compound COC(=O)C1=CC(C(C)=O)=CC=C1SSC1=CC=C(C(C)=O)C=C1C(=O)OC OXLSBJSNTAOWCI-UHFFFAOYSA-N 0.000 description 1
- SITMGAXHAJSFQD-UHFFFAOYSA-N methyl 5-acetyl-2-[(4-acetyl-2-methoxysulfonylphenyl)disulfanyl]benzenesulfonate Chemical compound COS(=O)(=O)C1=CC(C(C)=O)=CC=C1SSC1=CC=C(C(C)=O)C=C1S(=O)(=O)OC SITMGAXHAJSFQD-UHFFFAOYSA-N 0.000 description 1
- FQYGOZKPUNBGPK-UHFFFAOYSA-N methyl 5-acetyl-2-sulfanylbenzenesulfonate Chemical compound COS(=O)(=O)C1=CC(C(C)=O)=CC=C1S FQYGOZKPUNBGPK-UHFFFAOYSA-N 0.000 description 1
- VTOHSHPGIBDGHL-UHFFFAOYSA-N methyl 5-acetyl-2-sulfanylbenzenesulfonate;sodium Chemical compound [Na].COS(=O)(=O)C1=CC(C(C)=O)=CC=C1S VTOHSHPGIBDGHL-UHFFFAOYSA-N 0.000 description 1
- AXYLJWKBUUKEPF-UHFFFAOYSA-N methyl 5-acetyl-2-sulfanylbenzenesulfonate;zinc Chemical compound [Zn].COS(=O)(=O)C1=CC(C(C)=O)=CC=C1S AXYLJWKBUUKEPF-UHFFFAOYSA-N 0.000 description 1
- JGSGPXHQDKWINW-UHFFFAOYSA-N methyl 5-acetyl-2-sulfanylbenzoate Chemical compound COC(=O)C1=CC(C(C)=O)=CC=C1S JGSGPXHQDKWINW-UHFFFAOYSA-N 0.000 description 1
- XDUZMBKMBRKVNK-UHFFFAOYSA-N methyl 5-acetyl-2-sulfanylbenzoate;sodium Chemical compound [Na].COC(=O)C1=CC(C(C)=O)=CC=C1S XDUZMBKMBRKVNK-UHFFFAOYSA-N 0.000 description 1
- ZFDDTTGBLQBBKD-UHFFFAOYSA-N methyl 5-acetyl-2-sulfanylbenzoate;zinc Chemical compound [Zn].COC(=O)C1=CC(C(C)=O)=CC=C1S ZFDDTTGBLQBBKD-UHFFFAOYSA-N 0.000 description 1
- 150000002762 monocarboxylic acid derivatives Chemical class 0.000 description 1
- IUJLOAKJZQBENM-UHFFFAOYSA-N n-(1,3-benzothiazol-2-ylsulfanyl)-2-methylpropan-2-amine Chemical compound C1=CC=C2SC(SNC(C)(C)C)=NC2=C1 IUJLOAKJZQBENM-UHFFFAOYSA-N 0.000 description 1
- DEQZTKGFXNUBJL-UHFFFAOYSA-N n-(1,3-benzothiazol-2-ylsulfanyl)cyclohexanamine Chemical compound C1CCCCC1NSC1=NC2=CC=CC=C2S1 DEQZTKGFXNUBJL-UHFFFAOYSA-N 0.000 description 1
- MJCJUDJQDGGKOX-UHFFFAOYSA-N n-dodecyldodecan-1-amine Chemical compound CCCCCCCCCCCCNCCCCCCCCCCCC MJCJUDJQDGGKOX-UHFFFAOYSA-N 0.000 description 1
- HKUFIYBZNQSHQS-UHFFFAOYSA-N n-octadecyloctadecan-1-amine Chemical compound CCCCCCCCCCCCCCCCCCNCCCCCCCCCCCCCCCCCC HKUFIYBZNQSHQS-UHFFFAOYSA-N 0.000 description 1
- 229940032017 n-oxydiethylene-2-benzothiazole sulfenamide Drugs 0.000 description 1
- 229920003052 natural elastomer Polymers 0.000 description 1
- 229920001194 natural rubber Polymers 0.000 description 1
- 229910001453 nickel ion Inorganic materials 0.000 description 1
- ILUJQPXNXACGAN-UHFFFAOYSA-N ortho-methoxybenzoic acid Natural products COC1=CC=CC=C1C(O)=O ILUJQPXNXACGAN-UHFFFAOYSA-N 0.000 description 1
- 125000004043 oxo group Chemical group O=* 0.000 description 1
- CBVLLBOOKRSBMN-UHFFFAOYSA-N pentamethyl 6-[[2,3,4,5,6-pentakis(methoxycarbonyl)phenyl]disulfanyl]benzene-1,2,3,4,5-pentacarboxylate Chemical group COC(=O)C1=C(C(=O)OC)C(C(=O)OC)=C(C(=O)OC)C(C(=O)OC)=C1SSC1=C(C(=O)OC)C(C(=O)OC)=C(C(=O)OC)C(C(=O)OC)=C1C(=O)OC CBVLLBOOKRSBMN-UHFFFAOYSA-N 0.000 description 1
- SLGMHPUEIZPKRN-UHFFFAOYSA-N pentamethyl 6-[[2,3,4,5,6-pentakis(methoxysulfonyl)phenyl]disulfanyl]benzene-1,2,3,4,5-pentasulfonate Chemical group COS(=O)(=O)C1=C(S(=O)(=O)OC)C(S(=O)(=O)OC)=C(S(=O)(=O)OC)C(S(=O)(=O)OC)=C1SSC1=C(S(=O)(=O)OC)C(S(=O)(=O)OC)=C(S(=O)(=O)OC)C(S(=O)(=O)OC)=C1S(=O)(=O)OC SLGMHPUEIZPKRN-UHFFFAOYSA-N 0.000 description 1
- FLQGTNONDXLWGV-UHFFFAOYSA-N pentamethyl 6-sulfanylbenzene-1,2,3,4,5-pentacarboxylate Chemical compound COC(=O)C1=C(S)C(C(=O)OC)=C(C(=O)OC)C(C(=O)OC)=C1C(=O)OC FLQGTNONDXLWGV-UHFFFAOYSA-N 0.000 description 1
- OIWIASOJHMHIKJ-UHFFFAOYSA-N pentamethyl 6-sulfanylbenzene-1,2,3,4,5-pentacarboxylate;sodium Chemical compound [Na].COC(=O)C1=C(S)C(C(=O)OC)=C(C(=O)OC)C(C(=O)OC)=C1C(=O)OC OIWIASOJHMHIKJ-UHFFFAOYSA-N 0.000 description 1
- PAXYSHUSVCZBPY-UHFFFAOYSA-N pentamethyl 6-sulfanylbenzene-1,2,3,4,5-pentacarboxylate;zinc Chemical compound [Zn].COC(=O)C1=C(S)C(C(=O)OC)=C(C(=O)OC)C(C(=O)OC)=C1C(=O)OC PAXYSHUSVCZBPY-UHFFFAOYSA-N 0.000 description 1
- ZILDISZKQRRDHH-UHFFFAOYSA-N pentamethyl 6-sulfanylbenzene-1,2,3,4,5-pentasulfonate Chemical compound COS(=O)(=O)C1=C(S)C(S(=O)(=O)OC)=C(S(=O)(=O)OC)C(S(=O)(=O)OC)=C1S(=O)(=O)OC ZILDISZKQRRDHH-UHFFFAOYSA-N 0.000 description 1
- PUWMLLHGVOZLMG-UHFFFAOYSA-N pentamethyl 6-sulfanylbenzene-1,2,3,4,5-pentasulfonate;sodium Chemical compound [Na].COS(=O)(=O)C1=C(S)C(S(=O)(=O)OC)=C(S(=O)(=O)OC)C(S(=O)(=O)OC)=C1S(=O)(=O)OC PUWMLLHGVOZLMG-UHFFFAOYSA-N 0.000 description 1
- YDZLJCCAZDHEPN-UHFFFAOYSA-N pentamethyl 6-sulfanylbenzene-1,2,3,4,5-pentasulfonate;zinc Chemical compound [Zn].COS(=O)(=O)C1=C(S)C(S(=O)(=O)OC)=C(S(=O)(=O)OC)C(S(=O)(=O)OC)=C1S(=O)(=O)OC YDZLJCCAZDHEPN-UHFFFAOYSA-N 0.000 description 1
- UEVMAGDKIVIDRS-UHFFFAOYSA-I pentasodium 6-sulfanylbenzene-1,2,3,4,5-pentasulfinate Chemical compound [Na+].[Na+].[Na+].[Na+].[Na+].[O-]S(=O)c1c(S)c(c(c(c1S([O-])=O)S([O-])=O)S([O-])=O)S([O-])=O UEVMAGDKIVIDRS-UHFFFAOYSA-I 0.000 description 1
- ICVWCARRWPRIDF-UHFFFAOYSA-I pentasodium 6-sulfanylbenzene-1,2,3,4,5-pentasulfonate Chemical compound [Na+].S(=O)(=O)([O-])C1=C(C(=C(C(=C1S)S(=O)(=O)[O-])S(=O)(=O)[O-])S(=O)(=O)[O-])S(=O)(=O)[O-].[Na+].[Na+].[Na+].[Na+] ICVWCARRWPRIDF-UHFFFAOYSA-I 0.000 description 1
- ZTMCIQXYVCHSHH-UHFFFAOYSA-D pentazinc 6-sulfanylbenzene-1,2,3,4,5-pentasulfinate Chemical compound [Zn++].[Zn++].[Zn++].[Zn++].[Zn++].[O-]S(=O)c1c(S)c(c(c(c1S([O-])=O)S([O-])=O)S([O-])=O)S([O-])=O.[O-]S(=O)c1c(S)c(c(c(c1S([O-])=O)S([O-])=O)S([O-])=O)S([O-])=O ZTMCIQXYVCHSHH-UHFFFAOYSA-D 0.000 description 1
- 229920001195 polyisoprene Polymers 0.000 description 1
- 230000037048 polymerization activity Effects 0.000 description 1
- 239000011591 potassium Substances 0.000 description 1
- 229910052700 potassium Inorganic materials 0.000 description 1
- 229910000027 potassium carbonate Inorganic materials 0.000 description 1
- YQUVCSBJEUQKSH-UHFFFAOYSA-N protochatechuic acid Natural products OC(=O)C1=CC=C(O)C(O)=C1 YQUVCSBJEUQKSH-UHFFFAOYSA-N 0.000 description 1
- 150000003254 radicals Chemical class 0.000 description 1
- PCMORTLOPMLEFB-UHFFFAOYSA-N sinapinic acid Natural products COC1=CC(C=CC(O)=O)=CC(OC)=C1O PCMORTLOPMLEFB-UHFFFAOYSA-N 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 229910000029 sodium carbonate Inorganic materials 0.000 description 1
- 159000000000 sodium salts Chemical class 0.000 description 1
- VPABLRQBUTXQIK-UHFFFAOYSA-N sodium;1-(2,3,5,6-tetraacetyl-4-sulfanylphenyl)ethanone Chemical compound [Na].CC(=O)C1=C(S)C(C(C)=O)=C(C(C)=O)C(C(C)=O)=C1C(C)=O VPABLRQBUTXQIK-UHFFFAOYSA-N 0.000 description 1
- DKGGBLVWXRWRSG-UHFFFAOYSA-N sodium;1-(4-sulfanylphenyl)ethanone Chemical compound [Na].CC(=O)C1=CC=C(S)C=C1 DKGGBLVWXRWRSG-UHFFFAOYSA-N 0.000 description 1
- CVWGXHYIYUJHDD-UHFFFAOYSA-N sodium;1-[4-sulfanyl-3-(trichloromethyl)phenyl]ethanone Chemical compound [Na].CC(=O)C1=CC=C(S)C(C(Cl)(Cl)Cl)=C1 CVWGXHYIYUJHDD-UHFFFAOYSA-N 0.000 description 1
- QZCCFJVIQOWFHN-UHFFFAOYSA-N sodium;2,3,4,6-tetrabromobenzenethiol Chemical compound [Na].SC1=C(Br)C=C(Br)C(Br)=C1Br QZCCFJVIQOWFHN-UHFFFAOYSA-N 0.000 description 1
- QJAZPZXICQKPKA-UHFFFAOYSA-N sodium;2,3,4,6-tetrachlorobenzenethiol Chemical compound [Na].SC1=C(Cl)C=C(Cl)C(Cl)=C1Cl QJAZPZXICQKPKA-UHFFFAOYSA-N 0.000 description 1
- UJJOYFQSCKBVLZ-UHFFFAOYSA-N sodium;2,3,4,6-tetrafluorobenzenethiol Chemical compound [Na].FC1=CC(F)=C(S)C(F)=C1F UJJOYFQSCKBVLZ-UHFFFAOYSA-N 0.000 description 1
- RIBXYPHSINBTNP-UHFFFAOYSA-N sodium;2,3,4,6-tetraiodobenzenethiol Chemical compound [Na].SC1=C(I)C=C(I)C(I)=C1I RIBXYPHSINBTNP-UHFFFAOYSA-N 0.000 description 1
- FVRZWTOGMBGIIH-UHFFFAOYSA-N sodium;2,4,5-tribromobenzenethiol Chemical compound [Na].SC1=CC(Br)=C(Br)C=C1Br FVRZWTOGMBGIIH-UHFFFAOYSA-N 0.000 description 1
- KVLDESBEYFESPZ-UHFFFAOYSA-N sodium;2,4,5-trichlorobenzenethiol Chemical compound [Na].SC1=CC(Cl)=C(Cl)C=C1Cl KVLDESBEYFESPZ-UHFFFAOYSA-N 0.000 description 1
- JMBKDXOSXIMLCO-UHFFFAOYSA-N sodium;2,4,5-trifluorobenzenethiol Chemical compound [Na].FC1=CC(F)=C(S)C=C1F JMBKDXOSXIMLCO-UHFFFAOYSA-N 0.000 description 1
- XKHUMLOGAXYASV-UHFFFAOYSA-N sodium;2,4,5-triiodobenzenethiol Chemical compound [Na].SC1=CC(I)=C(I)C=C1I XKHUMLOGAXYASV-UHFFFAOYSA-N 0.000 description 1
- QMEOTHHUIAMMHX-UHFFFAOYSA-N sodium;2,4,5-trimethylbenzenethiol Chemical compound [Na].CC1=CC(C)=C(S)C=C1C QMEOTHHUIAMMHX-UHFFFAOYSA-N 0.000 description 1
- RHFLUQJEPFMUHG-UHFFFAOYSA-N sodium;2,4,5-tritert-butylbenzenethiol Chemical compound [Na].CC(C)(C)C1=CC(C(C)(C)C)=C(C(C)(C)C)C=C1S RHFLUQJEPFMUHG-UHFFFAOYSA-N 0.000 description 1
- RQYNNPKALCYURE-UHFFFAOYSA-N sodium;2,4,6-trimethoxybenzenethiol Chemical compound [Na].COC1=CC(OC)=C(S)C(OC)=C1 RQYNNPKALCYURE-UHFFFAOYSA-N 0.000 description 1
- YRUQFGMTXLCXLR-UHFFFAOYSA-N sodium;2,4,6-tris(methylsulfinyl)benzenethiol Chemical compound [Na].CS(=O)C1=CC(S(C)=O)=C(S)C(S(C)=O)=C1 YRUQFGMTXLCXLR-UHFFFAOYSA-N 0.000 description 1
- UILZFFQVEPHGDC-UHFFFAOYSA-N sodium;2,4,6-tris(trichloromethyl)benzenethiol Chemical compound [Na].SC1=C(C(Cl)(Cl)Cl)C=C(C(Cl)(Cl)Cl)C=C1C(Cl)(Cl)Cl UILZFFQVEPHGDC-UHFFFAOYSA-N 0.000 description 1
- PAULKJAEXBJCMU-UHFFFAOYSA-N sodium;2-sulfanyl-5-(trichloromethyl)phenol Chemical compound [Na].OC1=CC(C(Cl)(Cl)Cl)=CC=C1S PAULKJAEXBJCMU-UHFFFAOYSA-N 0.000 description 1
- QELGYKKQLSXAHM-UHFFFAOYSA-N sodium;2-sulfanylbenzene-1,3,5-tricarbaldehyde Chemical compound [Na].SC1=C(C=O)C=C(C=O)C=C1C=O QELGYKKQLSXAHM-UHFFFAOYSA-N 0.000 description 1
- GFXGBWIBMCJMDA-UHFFFAOYSA-N sodium;2-sulfanylbenzene-1,3,5-tricarbonitrile Chemical compound [Na].SC1=C(C#N)C=C(C#N)C=C1C#N GFXGBWIBMCJMDA-UHFFFAOYSA-N 0.000 description 1
- RXMIUKMEKAIHMU-UHFFFAOYSA-N sodium;2-sulfanylbenzene-1,3,5-tricarbonyl chloride Chemical compound [Na].SC1=C(C(Cl)=O)C=C(C(Cl)=O)C=C1C(Cl)=O RXMIUKMEKAIHMU-UHFFFAOYSA-N 0.000 description 1
- DQYFSIRNCPTDSV-UHFFFAOYSA-N sodium;2-sulfanylbenzene-1,3,5-tricarboxamide Chemical compound [Na].NC(=O)C1=CC(C(N)=O)=C(S)C(C(N)=O)=C1 DQYFSIRNCPTDSV-UHFFFAOYSA-N 0.000 description 1
- OVDOKVAFOSCJFQ-UHFFFAOYSA-N sodium;2-sulfanylbenzene-1,3,5-trisulfonyl chloride Chemical compound [Na].SC1=C(S(Cl)(=O)=O)C=C(S(Cl)(=O)=O)C=C1S(Cl)(=O)=O OVDOKVAFOSCJFQ-UHFFFAOYSA-N 0.000 description 1
- UUVCVNBLBWFONY-UHFFFAOYSA-M sodium;3-amino-4-sulfanylbenzenesulfinate Chemical compound [Na+].NC1=CC(S([O-])=O)=CC=C1S UUVCVNBLBWFONY-UHFFFAOYSA-M 0.000 description 1
- NQIJLVSTAAEBTB-UHFFFAOYSA-M sodium;3-amino-4-sulfanylbenzenesulfonate Chemical compound [Na+].NC1=CC(S([O-])(=O)=O)=CC=C1S NQIJLVSTAAEBTB-UHFFFAOYSA-M 0.000 description 1
- QSKOEOJCLCIOCE-UHFFFAOYSA-M sodium;3-amino-4-sulfanylbenzoate Chemical compound [Na+].NC1=CC(C([O-])=O)=CC=C1S QSKOEOJCLCIOCE-UHFFFAOYSA-M 0.000 description 1
- DVJLZIKMFIQCFA-UHFFFAOYSA-M sodium;3-hydroxy-4-sulfanylbenzenesulfinate Chemical compound [Na+].OC1=CC(S([O-])=O)=CC=C1S DVJLZIKMFIQCFA-UHFFFAOYSA-M 0.000 description 1
- HVTGUAYDTLZWKL-UHFFFAOYSA-M sodium;3-hydroxy-4-sulfanylbenzenesulfonate Chemical compound [Na+].OC1=CC(S([O-])(=O)=O)=CC=C1S HVTGUAYDTLZWKL-UHFFFAOYSA-M 0.000 description 1
- YSLKRZMCZVVHJF-UHFFFAOYSA-M sodium;3-hydroxy-4-sulfanylbenzoate Chemical compound [Na+].OC1=CC(C([O-])=O)=CC=C1S YSLKRZMCZVVHJF-UHFFFAOYSA-M 0.000 description 1
- GJGKAOSAJOXCPP-UHFFFAOYSA-M sodium;3-nitro-4-sulfanylbenzenesulfinate Chemical compound [Na+].[O-][N+](=O)C1=CC(S([O-])=O)=CC=C1S GJGKAOSAJOXCPP-UHFFFAOYSA-M 0.000 description 1
- DGDQCBDWPJPOBQ-UHFFFAOYSA-M sodium;3-nitro-4-sulfanylbenzenesulfonate Chemical compound [Na+].[O-][N+](=O)C1=CC(S([O-])(=O)=O)=CC=C1S DGDQCBDWPJPOBQ-UHFFFAOYSA-M 0.000 description 1
- DTJFVJAEHGWSNW-UHFFFAOYSA-M sodium;3-nitro-4-sulfanylbenzoate Chemical compound [Na+].[O-]C(=O)C1=CC=C(S)C([N+]([O-])=O)=C1 DTJFVJAEHGWSNW-UHFFFAOYSA-M 0.000 description 1
- QQIILAAFCRLFNI-UHFFFAOYSA-M sodium;3-phenylsulfanyl-4-sulfanylbenzenesulfinate Chemical compound [Na+].[O-]S(=O)C1=CC=C(S)C(SC=2C=CC=CC=2)=C1 QQIILAAFCRLFNI-UHFFFAOYSA-M 0.000 description 1
- JITZRBMQISWWOT-UHFFFAOYSA-M sodium;3-phenylsulfanyl-4-sulfanylbenzenesulfonate Chemical compound [Na+].[O-]S(=O)(=O)C1=CC=C(S)C(SC=2C=CC=CC=2)=C1 JITZRBMQISWWOT-UHFFFAOYSA-M 0.000 description 1
- PCHFGZFDMFKGSM-UHFFFAOYSA-M sodium;3-phenylsulfanyl-4-sulfanylbenzoate Chemical compound [Na+].[O-]C(=O)C1=CC=C(S)C(SC=2C=CC=CC=2)=C1 PCHFGZFDMFKGSM-UHFFFAOYSA-M 0.000 description 1
- PKKBZLJSMALSIQ-UHFFFAOYSA-N sodium;4-(trichloromethyl)benzenethiol Chemical compound [Na].SC1=CC=C(C(Cl)(Cl)Cl)C=C1 PKKBZLJSMALSIQ-UHFFFAOYSA-N 0.000 description 1
- VGQXKEVPYSJUHY-UHFFFAOYSA-M sodium;4-carboxybenzenethiolate Chemical compound [Na+].[O-]C(=O)C1=CC=C(S)C=C1 VGQXKEVPYSJUHY-UHFFFAOYSA-M 0.000 description 1
- JVPAIVHUZIKWTC-UHFFFAOYSA-N sodium;4-sulfanylbenzaldehyde Chemical compound [Na].SC1=CC=C(C=O)C=C1 JVPAIVHUZIKWTC-UHFFFAOYSA-N 0.000 description 1
- FFGCGWZYWRCUHZ-UHFFFAOYSA-N sodium;4-sulfanylbenzamide Chemical compound [Na].NC(=O)C1=CC=C(S)C=C1 FFGCGWZYWRCUHZ-UHFFFAOYSA-N 0.000 description 1
- RLNNEJGCXDRAKF-UHFFFAOYSA-M sodium;4-sulfanylbenzenesulfinate Chemical compound [Na+].[O-]S(=O)C1=CC=C(S)C=C1 RLNNEJGCXDRAKF-UHFFFAOYSA-M 0.000 description 1
- FBZVOYZHSCHMLJ-UHFFFAOYSA-M sodium;4-sulfanylbenzenesulfonate Chemical compound [Na+].[O-]S(=O)(=O)C1=CC=C(S)C=C1 FBZVOYZHSCHMLJ-UHFFFAOYSA-M 0.000 description 1
- JLZVWIJPPDKNET-UHFFFAOYSA-N sodium;4-sulfanylbenzenesulfonyl chloride Chemical compound [Na].SC1=CC=C(S(Cl)(=O)=O)C=C1 JLZVWIJPPDKNET-UHFFFAOYSA-N 0.000 description 1
- JRFPOSLTFJQEGB-UHFFFAOYSA-N sodium;4-sulfanylbenzonitrile Chemical compound [Na].SC1=CC=C(C#N)C=C1 JRFPOSLTFJQEGB-UHFFFAOYSA-N 0.000 description 1
- PZDZTMAUAIXJAR-UHFFFAOYSA-N sodium;4-sulfanylbenzoyl chloride Chemical compound [Na].SC1=CC=C(C(Cl)=O)C=C1 PZDZTMAUAIXJAR-UHFFFAOYSA-N 0.000 description 1
- OLKRQBLTMJDISC-UHFFFAOYSA-M sodium;5-acetyl-2-sulfanylbenzenesulfinate Chemical compound [Na+].CC(=O)C1=CC=C(S)C(S([O-])=O)=C1 OLKRQBLTMJDISC-UHFFFAOYSA-M 0.000 description 1
- RXPUGEFMSFCGGD-UHFFFAOYSA-M sodium;5-acetyl-2-sulfanylbenzenesulfonate Chemical compound [Na+].CC(=O)C1=CC=C(S)C(S([O-])(=O)=O)=C1 RXPUGEFMSFCGGD-UHFFFAOYSA-M 0.000 description 1
- UTIALFMWPZMKQS-UHFFFAOYSA-M sodium;5-acetyl-2-sulfanylbenzoate Chemical compound [Na+].CC(=O)C1=CC=C(S)C(C([O-])=O)=C1 UTIALFMWPZMKQS-UHFFFAOYSA-M 0.000 description 1
- PFWNKXNNUDSLFE-UHFFFAOYSA-N sodium;6-sulfanylbenzene-1,2,3,4,5-pentacarbaldehyde Chemical compound [Na].SC1=C(C=O)C(C=O)=C(C=O)C(C=O)=C1C=O PFWNKXNNUDSLFE-UHFFFAOYSA-N 0.000 description 1
- VZJYZZVYCWFAIN-UHFFFAOYSA-N sodium;6-sulfanylbenzene-1,2,3,4,5-pentacarbonitrile Chemical compound [Na].SC1=C(C#N)C(C#N)=C(C#N)C(C#N)=C1C#N VZJYZZVYCWFAIN-UHFFFAOYSA-N 0.000 description 1
- TWJIQGXVZVIRMB-UHFFFAOYSA-N sodium;6-sulfanylbenzene-1,2,3,4,5-pentacarboxamide Chemical compound [Na].NC(=O)C1=C(S)C(C(N)=O)=C(C(N)=O)C(C(N)=O)=C1C(N)=O TWJIQGXVZVIRMB-UHFFFAOYSA-N 0.000 description 1
- QQDXSZZWUMJOKO-UHFFFAOYSA-N sodium;trimethyl 2-sulfanylbenzene-1,3,5-tricarboxylate Chemical compound [Na].COC(=O)C1=CC(C(=O)OC)=C(S)C(C(=O)OC)=C1 QQDXSZZWUMJOKO-UHFFFAOYSA-N 0.000 description 1
- RPLBQCNDQFGMJF-UHFFFAOYSA-N sodium;trimethyl 2-sulfanylbenzene-1,3,5-trisulfonate Chemical compound [Na].COS(=O)(=O)C1=CC(S(=O)(=O)OC)=C(S)C(S(=O)(=O)OC)=C1 RPLBQCNDQFGMJF-UHFFFAOYSA-N 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 229910052712 strontium Inorganic materials 0.000 description 1
- CIOAGBVUUVVLOB-UHFFFAOYSA-N strontium atom Chemical compound [Sr] CIOAGBVUUVVLOB-UHFFFAOYSA-N 0.000 description 1
- 229920003048 styrene butadiene rubber Polymers 0.000 description 1
- QAZLUNIWYYOJPC-UHFFFAOYSA-M sulfenamide Chemical compound [Cl-].COC1=C(C)C=[N+]2C3=NC4=CC=C(OC)C=C4N3SCC2=C1C QAZLUNIWYYOJPC-UHFFFAOYSA-M 0.000 description 1
- 238000003786 synthesis reaction Methods 0.000 description 1
- YIBXWXOYFGZLRU-UHFFFAOYSA-N syringic aldehyde Natural products CC12CCC(C3(CCC(=O)C(C)(C)C3CC=3)C)C=3C1(C)CCC2C1COC(C)(C)C(O)C(O)C1 YIBXWXOYFGZLRU-UHFFFAOYSA-N 0.000 description 1
- 229910052714 tellurium Inorganic materials 0.000 description 1
- PORWMNRCUJJQNO-UHFFFAOYSA-N tellurium atom Chemical compound [Te] PORWMNRCUJJQNO-UHFFFAOYSA-N 0.000 description 1
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 1
- 150000003557 thiazoles Chemical class 0.000 description 1
- 150000003566 thiocarboxylic acids Chemical class 0.000 description 1
- 239000010936 titanium Substances 0.000 description 1
- 229910052719 titanium Inorganic materials 0.000 description 1
- LDHQCZJRKDOVOX-UHFFFAOYSA-N trans-crotonic acid Natural products CC=CC(O)=O LDHQCZJRKDOVOX-UHFFFAOYSA-N 0.000 description 1
- QURCVMIEKCOAJU-UHFFFAOYSA-N trans-isoferulic acid Natural products COC1=CC=C(C=CC(O)=O)C=C1O QURCVMIEKCOAJU-UHFFFAOYSA-N 0.000 description 1
- UWHZIFQPPBDJPM-BQYQJAHWSA-N trans-vaccenic acid Chemical compound CCCCCC\C=C\CCCCCCCCCC(O)=O UWHZIFQPPBDJPM-BQYQJAHWSA-N 0.000 description 1
- HYHHYDMKAPPOOS-UHFFFAOYSA-N trimethyl 2-[[2,4,6-tris(methoxycarbonyl)phenyl]disulfanyl]benzene-1,3,5-tricarboxylate Chemical group COC(=O)C1=CC(C(=O)OC)=CC(C(=O)OC)=C1SSC1=C(C(=O)OC)C=C(C(=O)OC)C=C1C(=O)OC HYHHYDMKAPPOOS-UHFFFAOYSA-N 0.000 description 1
- PNSLNROJZVCCKN-UHFFFAOYSA-N trimethyl 2-[[2,4,6-tris(methoxysulfonyl)phenyl]disulfanyl]benzene-1,3,5-trisulfonate Chemical group COS(=O)(=O)C1=CC(S(=O)(=O)OC)=CC(S(=O)(=O)OC)=C1SSC1=C(S(=O)(=O)OC)C=C(S(=O)(=O)OC)C=C1S(=O)(=O)OC PNSLNROJZVCCKN-UHFFFAOYSA-N 0.000 description 1
- WFURELALFVNZSX-UHFFFAOYSA-N trimethyl 2-sulfanylbenzene-1,3,5-tricarboxylate Chemical compound COC(=O)C1=CC(C(=O)OC)=C(S)C(C(=O)OC)=C1 WFURELALFVNZSX-UHFFFAOYSA-N 0.000 description 1
- JXTVFNOZNNGTFE-UHFFFAOYSA-N trimethyl 2-sulfanylbenzene-1,3,5-tricarboxylate;zinc Chemical compound [Zn].COC(=O)C1=CC(C(=O)OC)=C(S)C(C(=O)OC)=C1 JXTVFNOZNNGTFE-UHFFFAOYSA-N 0.000 description 1
- LYCNKONNEXHXRZ-UHFFFAOYSA-N trimethyl 2-sulfanylbenzene-1,3,5-trisulfonate Chemical compound COS(=O)(=O)C1=CC(S(=O)(=O)OC)=C(S)C(S(=O)(=O)OC)=C1 LYCNKONNEXHXRZ-UHFFFAOYSA-N 0.000 description 1
- AOYKINDBYLKSNY-UHFFFAOYSA-N trimethyl 2-sulfanylbenzene-1,3,5-trisulfonate;zinc Chemical compound [Zn].COS(=O)(=O)C1=CC(S(=O)(=O)OC)=C(S)C(S(=O)(=O)OC)=C1 AOYKINDBYLKSNY-UHFFFAOYSA-N 0.000 description 1
- IJYNHVFWHDZASZ-UHFFFAOYSA-K trisodium;2-sulfanylbenzene-1,3,5-tricarboxylate Chemical compound [Na+].[Na+].[Na+].[O-]C(=O)C1=CC(C([O-])=O)=C(S)C(C([O-])=O)=C1 IJYNHVFWHDZASZ-UHFFFAOYSA-K 0.000 description 1
- HAORCFQJRPVBFD-UHFFFAOYSA-K trisodium;2-sulfanylbenzene-1,3,5-trisulfinate Chemical compound [Na+].[Na+].[Na+].[O-]S(=O)C1=CC(S([O-])=O)=C(S)C(S([O-])=O)=C1 HAORCFQJRPVBFD-UHFFFAOYSA-K 0.000 description 1
- WBTZUCAMSWVTTN-UHFFFAOYSA-H trizinc 2-sulfanylbenzene-1,3,5-trisulfonate Chemical compound [Zn++].[Zn++].[Zn++].[O-]S(=O)(=O)c1cc(c(S)c(c1)S([O-])(=O)=O)S([O-])(=O)=O.[O-]S(=O)(=O)c1cc(c(S)c(c1)S([O-])(=O)=O)S([O-])(=O)=O WBTZUCAMSWVTTN-UHFFFAOYSA-H 0.000 description 1
- PAHFNKBBQGOFLK-UHFFFAOYSA-H trizinc;2-sulfanylbenzene-1,3,5-tricarboxylate Chemical compound [Zn+2].[Zn+2].[Zn+2].[O-]C(=O)C1=CC(C([O-])=O)=C(S)C(C([O-])=O)=C1.[O-]C(=O)C1=CC(C([O-])=O)=C(S)C(C([O-])=O)=C1 PAHFNKBBQGOFLK-UHFFFAOYSA-H 0.000 description 1
- JYPDFDJLVWOUEB-UHFFFAOYSA-H trizinc;2-sulfanylbenzene-1,3,5-trisulfinate Chemical compound [Zn+2].[Zn+2].[Zn+2].[O-]S(=O)C1=CC(S([O-])=O)=C(S)C(S([O-])=O)=C1.[O-]S(=O)C1=CC(S([O-])=O)=C(S)C(S([O-])=O)=C1 JYPDFDJLVWOUEB-UHFFFAOYSA-H 0.000 description 1
- 229940005605 valeric acid Drugs 0.000 description 1
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 description 1
- 239000011701 zinc Substances 0.000 description 1
- 239000011667 zinc carbonate Substances 0.000 description 1
- 229910000010 zinc carbonate Inorganic materials 0.000 description 1
- 235000004416 zinc carbonate Nutrition 0.000 description 1
- 150000003752 zinc compounds Chemical class 0.000 description 1
- BOXSVZNGTQTENJ-UHFFFAOYSA-L zinc dibutyldithiocarbamate Chemical compound [Zn+2].CCCCN(C([S-])=S)CCCC.CCCCN(C([S-])=S)CCCC BOXSVZNGTQTENJ-UHFFFAOYSA-L 0.000 description 1
- RKQOSDAEEGPRER-UHFFFAOYSA-L zinc diethyldithiocarbamate Chemical compound [Zn+2].CCN(CC)C([S-])=S.CCN(CC)C([S-])=S RKQOSDAEEGPRER-UHFFFAOYSA-L 0.000 description 1
- UGZADUVQMDAIAO-UHFFFAOYSA-L zinc hydroxide Chemical compound [OH-].[OH-].[Zn+2] UGZADUVQMDAIAO-UHFFFAOYSA-L 0.000 description 1
- 229910021511 zinc hydroxide Inorganic materials 0.000 description 1
- 229940007718 zinc hydroxide Drugs 0.000 description 1
- AYTGKLVTBOQQRP-UHFFFAOYSA-L zinc;3-amino-4-sulfanylbenzenesulfinate Chemical compound [Zn+2].NC1=CC(S([O-])=O)=CC=C1S.NC1=CC(S([O-])=O)=CC=C1S AYTGKLVTBOQQRP-UHFFFAOYSA-L 0.000 description 1
- CQXRJEZSOOQZMQ-UHFFFAOYSA-L zinc;3-amino-4-sulfanylbenzenesulfonate Chemical compound [Zn+2].NC1=CC(S([O-])(=O)=O)=CC=C1S.NC1=CC(S([O-])(=O)=O)=CC=C1S CQXRJEZSOOQZMQ-UHFFFAOYSA-L 0.000 description 1
- OMYUEEMBBMBXKR-UHFFFAOYSA-L zinc;3-amino-4-sulfanylbenzoate Chemical compound [Zn+2].NC1=CC(C([O-])=O)=CC=C1S.NC1=CC(C([O-])=O)=CC=C1S OMYUEEMBBMBXKR-UHFFFAOYSA-L 0.000 description 1
- ZGRSPLFUBUEOIM-UHFFFAOYSA-L zinc;3-hydroxy-4-sulfanylbenzenesulfinate Chemical compound [Zn+2].OC1=CC(S([O-])=O)=CC=C1S.OC1=CC(S([O-])=O)=CC=C1S ZGRSPLFUBUEOIM-UHFFFAOYSA-L 0.000 description 1
- VCDDJHZSISWGQI-UHFFFAOYSA-L zinc;3-hydroxy-4-sulfanylbenzenesulfonate Chemical compound [Zn+2].OC1=CC(S([O-])(=O)=O)=CC=C1S.OC1=CC(S([O-])(=O)=O)=CC=C1S VCDDJHZSISWGQI-UHFFFAOYSA-L 0.000 description 1
- GBFNJTVNAFXDBH-UHFFFAOYSA-L zinc;3-nitro-4-sulfanylbenzenesulfinate Chemical compound [Zn+2].[O-][N+](=O)C1=CC(S([O-])=O)=CC=C1S.[O-][N+](=O)C1=CC(S([O-])=O)=CC=C1S GBFNJTVNAFXDBH-UHFFFAOYSA-L 0.000 description 1
- GWHCKOGWHBHMEO-UHFFFAOYSA-L zinc;3-nitro-4-sulfanylbenzenesulfonate Chemical compound [Zn+2].[O-][N+](=O)C1=CC(S([O-])(=O)=O)=CC=C1S.[O-][N+](=O)C1=CC(S([O-])(=O)=O)=CC=C1S GWHCKOGWHBHMEO-UHFFFAOYSA-L 0.000 description 1
- AOACDIWKNVIYKZ-UHFFFAOYSA-L zinc;3-nitro-4-sulfanylbenzoate Chemical compound [Zn+2].[O-]C(=O)C1=CC=C(S)C([N+]([O-])=O)=C1.[O-]C(=O)C1=CC=C(S)C([N+]([O-])=O)=C1 AOACDIWKNVIYKZ-UHFFFAOYSA-L 0.000 description 1
- ZGHAMBCSXGLSMJ-UHFFFAOYSA-L zinc;3-phenylsulfanyl-4-sulfanylbenzenesulfinate Chemical compound [Zn+2].[O-]S(=O)C1=CC=C(S)C(SC=2C=CC=CC=2)=C1.[O-]S(=O)C1=CC=C(S)C(SC=2C=CC=CC=2)=C1 ZGHAMBCSXGLSMJ-UHFFFAOYSA-L 0.000 description 1
- BHDHLQKIQRLTAM-UHFFFAOYSA-L zinc;3-phenylsulfanyl-4-sulfanylbenzenesulfonate Chemical compound [Zn+2].[O-]S(=O)(=O)C1=CC=C(S)C(SC=2C=CC=CC=2)=C1.[O-]S(=O)(=O)C1=CC=C(S)C(SC=2C=CC=CC=2)=C1 BHDHLQKIQRLTAM-UHFFFAOYSA-L 0.000 description 1
- YADNTLMOAFSWTH-UHFFFAOYSA-L zinc;3-phenylsulfanyl-4-sulfanylbenzoate Chemical compound [Zn+2].[O-]C(=O)C1=CC=C(S)C(SC=2C=CC=CC=2)=C1.[O-]C(=O)C1=CC=C(S)C(SC=2C=CC=CC=2)=C1 YADNTLMOAFSWTH-UHFFFAOYSA-L 0.000 description 1
- CTBSOSCEZSHKMH-UHFFFAOYSA-L zinc;4-sulfanylbenzenesulfinate Chemical compound [Zn+2].[O-]S(=O)C1=CC=C(S)C=C1.[O-]S(=O)C1=CC=C(S)C=C1 CTBSOSCEZSHKMH-UHFFFAOYSA-L 0.000 description 1
- BBAXJINBIJFGIT-UHFFFAOYSA-L zinc;4-sulfanylbenzenesulfonate Chemical compound [Zn+2].[O-]S(=O)(=O)C1=CC=C(S)C=C1.[O-]S(=O)(=O)C1=CC=C(S)C=C1 BBAXJINBIJFGIT-UHFFFAOYSA-L 0.000 description 1
- RVLPSXJARBGHOK-UHFFFAOYSA-L zinc;4-sulfanylbenzoate Chemical compound [Zn+2].[O-]C(=O)C1=CC=C(S)C=C1.[O-]C(=O)C1=CC=C(S)C=C1 RVLPSXJARBGHOK-UHFFFAOYSA-L 0.000 description 1
- IEDFVWMHKJYPJU-UHFFFAOYSA-L zinc;5-acetyl-2-sulfanylbenzenesulfinate Chemical compound [Zn+2].CC(=O)C1=CC=C(S)C(S([O-])=O)=C1.CC(=O)C1=CC=C(S)C(S([O-])=O)=C1 IEDFVWMHKJYPJU-UHFFFAOYSA-L 0.000 description 1
- OGHFRQCZEMWIIJ-UHFFFAOYSA-L zinc;5-acetyl-2-sulfanylbenzenesulfonate Chemical compound [Zn+2].CC(=O)C1=CC=C(S)C(S([O-])(=O)=O)=C1.CC(=O)C1=CC=C(S)C(S([O-])(=O)=O)=C1 OGHFRQCZEMWIIJ-UHFFFAOYSA-L 0.000 description 1
- RMFSVRHVUKCUBP-UHFFFAOYSA-L zinc;5-acetyl-2-sulfanylbenzoate Chemical compound [Zn+2].CC(=O)C1=CC=C(S)C(C([O-])=O)=C1.CC(=O)C1=CC=C(S)C(C([O-])=O)=C1 RMFSVRHVUKCUBP-UHFFFAOYSA-L 0.000 description 1
- SUUHKTAHEUYQOU-UHFFFAOYSA-L zinc;5-carboxy-2-sulfanylphenolate Chemical compound [Zn+2].OC(=O)C1=CC=C(S)C([O-])=C1.OC(=O)C1=CC=C(S)C([O-])=C1 SUUHKTAHEUYQOU-UHFFFAOYSA-L 0.000 description 1
- KMNUDJAXRXUZQS-UHFFFAOYSA-L zinc;n-ethyl-n-phenylcarbamodithioate Chemical compound [Zn+2].CCN(C([S-])=S)C1=CC=CC=C1.CCN(C([S-])=S)C1=CC=CC=C1 KMNUDJAXRXUZQS-UHFFFAOYSA-L 0.000 description 1
- DUBNHZYBDBBJHD-UHFFFAOYSA-L ziram Chemical compound [Zn+2].CN(C)C([S-])=S.CN(C)C([S-])=S DUBNHZYBDBBJHD-UHFFFAOYSA-L 0.000 description 1
- 229910052726 zirconium Inorganic materials 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A63—SPORTS; GAMES; AMUSEMENTS
- A63B—APPARATUS FOR PHYSICAL TRAINING, GYMNASTICS, SWIMMING, CLIMBING, OR FENCING; BALL GAMES; TRAINING EQUIPMENT
- A63B37/00—Solid balls; Rigid hollow balls; Marbles
- A63B37/0003—Golf balls
- A63B37/005—Cores
- A63B37/0051—Materials other than polybutadienes; Constructional details
- A63B37/0054—Substantially rigid, e.g. metal
-
- A—HUMAN NECESSITIES
- A63—SPORTS; GAMES; AMUSEMENTS
- A63B—APPARATUS FOR PHYSICAL TRAINING, GYMNASTICS, SWIMMING, CLIMBING, OR FENCING; BALL GAMES; TRAINING EQUIPMENT
- A63B37/00—Solid balls; Rigid hollow balls; Marbles
-
- A—HUMAN NECESSITIES
- A63—SPORTS; GAMES; AMUSEMENTS
- A63B—APPARATUS FOR PHYSICAL TRAINING, GYMNASTICS, SWIMMING, CLIMBING, OR FENCING; BALL GAMES; TRAINING EQUIPMENT
- A63B37/00—Solid balls; Rigid hollow balls; Marbles
- A63B37/0003—Golf balls
-
- A—HUMAN NECESSITIES
- A63—SPORTS; GAMES; AMUSEMENTS
- A63B—APPARATUS FOR PHYSICAL TRAINING, GYMNASTICS, SWIMMING, CLIMBING, OR FENCING; BALL GAMES; TRAINING EQUIPMENT
- A63B37/00—Solid balls; Rigid hollow balls; Marbles
- A63B37/0003—Golf balls
- A63B37/0023—Covers
- A63B37/0029—Physical properties
- A63B37/0031—Hardness
-
- A—HUMAN NECESSITIES
- A63—SPORTS; GAMES; AMUSEMENTS
- A63B—APPARATUS FOR PHYSICAL TRAINING, GYMNASTICS, SWIMMING, CLIMBING, OR FENCING; BALL GAMES; TRAINING EQUIPMENT
- A63B37/00—Solid balls; Rigid hollow balls; Marbles
- A63B37/0003—Golf balls
- A63B37/0023—Covers
- A63B37/0029—Physical properties
- A63B37/0031—Hardness
- A63B37/0032—Hardness gradient
-
- A—HUMAN NECESSITIES
- A63—SPORTS; GAMES; AMUSEMENTS
- A63B—APPARATUS FOR PHYSICAL TRAINING, GYMNASTICS, SWIMMING, CLIMBING, OR FENCING; BALL GAMES; TRAINING EQUIPMENT
- A63B37/00—Solid balls; Rigid hollow balls; Marbles
- A63B37/0003—Golf balls
- A63B37/0038—Intermediate layers, e.g. inner cover, outer core, mantle
-
- A—HUMAN NECESSITIES
- A63—SPORTS; GAMES; AMUSEMENTS
- A63B—APPARATUS FOR PHYSICAL TRAINING, GYMNASTICS, SWIMMING, CLIMBING, OR FENCING; BALL GAMES; TRAINING EQUIPMENT
- A63B37/00—Solid balls; Rigid hollow balls; Marbles
- A63B37/0003—Golf balls
- A63B37/0038—Intermediate layers, e.g. inner cover, outer core, mantle
- A63B37/004—Physical properties
- A63B37/0043—Hardness
- A63B37/0044—Hardness gradient
-
- A—HUMAN NECESSITIES
- A63—SPORTS; GAMES; AMUSEMENTS
- A63B—APPARATUS FOR PHYSICAL TRAINING, GYMNASTICS, SWIMMING, CLIMBING, OR FENCING; BALL GAMES; TRAINING EQUIPMENT
- A63B37/00—Solid balls; Rigid hollow balls; Marbles
- A63B37/0003—Golf balls
- A63B37/005—Cores
- A63B37/0051—Materials other than polybutadienes; Constructional details
-
- A—HUMAN NECESSITIES
- A63—SPORTS; GAMES; AMUSEMENTS
- A63B—APPARATUS FOR PHYSICAL TRAINING, GYMNASTICS, SWIMMING, CLIMBING, OR FENCING; BALL GAMES; TRAINING EQUIPMENT
- A63B37/00—Solid balls; Rigid hollow balls; Marbles
- A63B37/0003—Golf balls
- A63B37/005—Cores
- A63B37/006—Physical properties
- A63B37/0062—Hardness
- A63B37/00622—Surface hardness
-
- A—HUMAN NECESSITIES
- A63—SPORTS; GAMES; AMUSEMENTS
- A63B—APPARATUS FOR PHYSICAL TRAINING, GYMNASTICS, SWIMMING, CLIMBING, OR FENCING; BALL GAMES; TRAINING EQUIPMENT
- A63B37/00—Solid balls; Rigid hollow balls; Marbles
- A63B37/0003—Golf balls
- A63B37/005—Cores
- A63B37/006—Physical properties
- A63B37/0062—Hardness
- A63B37/0063—Hardness gradient
-
- A—HUMAN NECESSITIES
- A63—SPORTS; GAMES; AMUSEMENTS
- A63B—APPARATUS FOR PHYSICAL TRAINING, GYMNASTICS, SWIMMING, CLIMBING, OR FENCING; BALL GAMES; TRAINING EQUIPMENT
- A63B37/00—Solid balls; Rigid hollow balls; Marbles
- A63B37/0003—Golf balls
- A63B37/007—Characteristics of the ball as a whole
- A63B37/0072—Characteristics of the ball as a whole with a specified number of layers
- A63B37/0076—Multi-piece balls, i.e. having two or more intermediate layers
-
- A—HUMAN NECESSITIES
- A63—SPORTS; GAMES; AMUSEMENTS
- A63B—APPARATUS FOR PHYSICAL TRAINING, GYMNASTICS, SWIMMING, CLIMBING, OR FENCING; BALL GAMES; TRAINING EQUIPMENT
- A63B37/00—Solid balls; Rigid hollow balls; Marbles
- A63B37/0003—Golf balls
- A63B37/007—Characteristics of the ball as a whole
- A63B37/0077—Physical properties
- A63B37/0092—Hardness distribution amongst different ball layers
- A63B37/00922—Hardness distribution amongst different ball layers whereby hardness of the cover is lower than hardness of the intermediate layers
-
- A—HUMAN NECESSITIES
- A63—SPORTS; GAMES; AMUSEMENTS
- A63B—APPARATUS FOR PHYSICAL TRAINING, GYMNASTICS, SWIMMING, CLIMBING, OR FENCING; BALL GAMES; TRAINING EQUIPMENT
- A63B37/00—Solid balls; Rigid hollow balls; Marbles
- A63B37/02—Special cores
- A63B37/06—Elastic cores
Definitions
- the present invention relates to golf balls. Specifically, the present invention relates to golf balls that include a solid core and a cover including two or more layers.
- Flight performance In particular, golf players place importance on flight performance upon shots with a driver and a long iron. Flight performance correlates with the resilience performance of a golf ball. When a golf ball having excellent resilience performance is hit, the golf ball flies at a high speed, thereby achieving a large flight distance. Golf balls that include a core having excellent resilience performance are disclosed in JP61-37178 , JP2008-212681 ( US2008/0214324 ), JP2008-523952 ( US2006/0135287 and US2007/0173607 ), and JP2009-119256 ( US2009/0124757 ).
- JP61-37178 is obtained from a rubber composition that includes a co-crosslinking agent and a crosslinking activator.
- This publication discloses palmitic acid, stearic acid, and myristic acid as the crosslinking activator.
- the core disclosed in JP2008-212681 is obtained from a rubber composition that includes an organic peroxide, a metal salt of an ⁇ , ⁇ -unsaturated carboxylic acid, and a copper salt of a fatty acid.
- the core disclosed in JP2008-523952 is obtained from a rubber composition that includes a metal salt of an unsaturated monocarboxylic acid, a free radical initiator, and a non-conjugated diene monomer.
- the core disclosed in JP2009-119256 is obtained from a rubber composition that includes a polybutadiene whose vinyl content is equal to or less than 2%, whose cis 1,4-blond content is equal to or greater than 80%, and which has an active end modified with an alkoxysilane compound.
- An appropriate trajectory height is required in order to achieve a large flight distance.
- a trajectory height depends on a spin rate and a launch angle. With a golf ball that achieves a high trajectory by a high spin rate, a flight distance is insufficient. With a golf ball that achieves a high trajectory by a high launch angle, a large flight distance is obtained.
- Use of an outer-hard/inner-soft structure in a golf ball can achieve a low spin rate and a high launch angle.
- JP6-154357 (USP 5,403,010 ), JP2008-194471 (USP 7,344,455 , US2008/0194358 , US2008/0194359 , and US2008/0214325 ), and JP2008-194473 ( US2008/0194357 and US2008/0312008 ).
- a JIS-C hardness H1 at the central point of the core is 58 to 73
- a JIS-C hardness H2 in a region that extends over a distance range from equal to or greater than 5 mm to equal to or less than 10 mm from the central point is equal to or greater than 65 but equal to or less than 75
- a JIS-C hardness H3 at a point located at a distance of 15 mm from the central point is equal to or greater than 74 but equal to or less than 82
- a JIS-C hardness H4 at the surface of the core is equal to or greater than 76 but equal to or less than 84.
- the hardness H2 is greater than the hardness H1
- the hardness H3 is greater than the hardness H2
- the hardness H4 is equal to or greater than the hardness H3.
- a Shore D hardness at the central point of the core is equal to or greater than 30 but equal to or less than 48, a Shore D hardness at a point located at a distance of 4 mm from the central point is equal to or greater than 34 but equal to or less than 52, a Shore D hardness at a point located at a distance of 8 mm from the central point is equal to or greater than 40 but equal to or less than 58, a Shore D hardness at a point located at a distance of 12 mm from the central point is equal to or greater than 43 but equal to or less than 61, a Shore D hardness in a region that extends over a distance range from equal to or greater than 2 mm to equal to or less than 3 mm from the surface of the core is equal to or greater than 36 but equal to or less than 54, and a Shore D hardness at the surface is equal to or greater than 41 but equal to or less than 59.
- a Shore D hardness at the central point of the core is equal to or greater than 25 but equal to or less than 45
- a Shore D hardness in a region that extends over a distance range from equal to or greater than 5 mm to equal to or less than 10 mm from the central point is equal to or greater than 39 but equal to or less than 58
- a Shore D hardness at a point located at a distance of 15 mm from the central point is equal to or greater than 36 but equal to or less than 55
- a Shore D hardness at the surface of the core is equal to or greater than 55 but equal to or less than 75.
- JP2010-253268 ( US2010/0273575 ) discloses a golf ball that includes a core, an envelope layer, a mid layer, and a cover.
- the hardness gradually increases from the central point of the core to the surface of the core.
- the difference between a JIS-C hardness at the surface and a JIS-C hardness at the central point is equal to or greater than 15.
- the hardness of the cover is greater than the hardness of the mid layer, and the hardness of the mid layer is greater than the hardness of the envelope layer.
- US 2011/306443 A1 discloses a golf ball comprising a core, a mid layer positioned outside the core and a cover positioned outside the mid layer, wherein the core comprises a center and an envelope layer.
- the core of the golf ball may be composed of a rubber composition including a base resin, a crosslinking initiator, a co-crosslinking agent, zinc oxide and barium sulfate.
- US 2003/109332 A1 relates to a golf ball having a core, an inner cover and an outer cover, wherein the JIS-C hardness of the inner cover is higher than the JIS-C hardness at the surface of the core by less than 15.
- the core of the golf ball may be composed of a rubber composition including a base resin, a crosslinking initiator, a co-crosslinking agent, zinc oxide and barium sulfate.
- An object of the present invention is to provide a golf ball that exerts excellent flight performance upon a shot with a middle iron.
- Another object of the present invention is to provide a golf ball that achieves a large flight distance and excellent feel at impact upon a shot with a driver.
- the present invention relates to a golf ball, which consists of a spherical core and a cover covering the core and including two or more layers, wherein when distances (%) from a central point of the core to nine points and JIS-C hardnesses at the nine points, which nine points are obtained by dividing a region from the central point of the core to a surface of the core at intervals of 12.5% of a radius of the core, are plotted in a graph, R2 of a linear approximation curve obtained by a least-square method is equal to or greater than 0.95, wherein R2 is calculated by squaring a correlation coefficient R, wherein the correlation coefficient R is calculated by dividing the covariance of the distance (%) from the central point and the hardness (JIS-C) by the standard deviation of the distance (%) from the central point and the standard deviation of the hardness (JIS-C), the JIS-C hardness Hi of the innermost layer of the cover is greater than the JIS-C hardness Hs at the surface of
- the JIS-C hardness Hi of an innermost layer of the cover is greater than a JIS-C hardness Hs at the surface of the core.
- the hardness distribution of the golf ball is appropriate.
- the energy loss is low when the golf ball is hit with a middle iron.
- the spin rate is low. The low spin rate achieves a large flight distance.
- the JIS-C hardness Hi of the innermost layer of the cover is equal to or less than the JIS-C hardness Hs at the surface of the core.
- the hardness distribution is appropriate.
- the energy loss is low when the golf ball is hit with a driver.
- the spin rate is low.
- the low spin rate achieves a large flight distance.
- the innermost layer achieves soft feel at impact.
- the golf ball of this embodiment has both excellent flight performance and excellent feel at impact.
- a golf ball 2 shown in FIG. 1 includes a spherical core 4 and a cover 6 covering the core 4.
- the cover 6 includes an inner cover 8 and an outer cover 10 positioned outside the inner cover 8.
- the inner cover 8 is an innermost layer of the cover 6.
- the outer cover 10 is an outermost layer of the cover 6.
- the cover 6 may include another one or more layers between the inner cover 8 and the outer cover 10.
- On the surface of the outer cover 10, a large number of dimples 12 are formed.
- a part other than the dimples 12 is a land 14.
- the golf ball 2 includes a paint layer and a mark layer on the external side of the outer cover 10, but these layers are not shown in the drawing.
- the golf ball 2 has a diameter of 40 mm or greater but 45 mm or less. From the standpoint of conformity to the rules established by the United States Golf Association (USGA), the diameter is preferably equal to or greater than 42.67 mm. In light of suppression of air resistance, the diameter is preferably equal to or less than 44 mm and more preferably equal to or less than 42.80 mm.
- the golf ball 2 has a weight of 40 g or greater but 50 g or less. In light of attainment of great inertia, the weight is preferably equal to or greater than 44 g and more preferably equal to or greater than 45.00 g. From the standpoint of conformity to the rules established by the USGA, the weight is preferably equal to or less than 45.93 g.
- FIG. 2 is a line graph showing a hardness distribution of the core 4 of the golf ball 2 in FIG. 1 .
- the horizontal axis of the graph indicates the ratio (%) of a distance from the central point of the core 4 to the radius of the core 4.
- the vertical axis of the graph indicates a JIS-C hardness.
- FIG. 2 also shows a linear approximation curve obtained by a least-square method on the basis of the distances and the hardnesses of the nine measuring points.
- the broken line does not greatly deviate from the linear approximation curve.
- the broken line has a shape close to the linear approximation curve.
- the hardness linearly increases from its central point toward its surface.
- R 2 of the linear approximation curve obtained by the least-square method is equal to or greater than 0.95.
- R 2 is an index indicating the linearity of the broken line.
- the shape of the broken line of the hardness distribution is close to a straight line.
- the core 4 for which R 2 is equal to or greater than 0.95 has excellent resilience performance.
- R 2 is more preferably equal to or greater than 0.96 and particularly preferably equal to or greater than 0.97.
- R 2 is calculated by squaring a correlation coefficient R.
- the correlation coefficient R is calculated by dividing the covariance of the distance (%) from the central point and the hardness (JIS-C) by the standard deviation of the distance (%) from the central point and the standard deviation of the hardness (JIS-C).
- the core 4 is obtained by crosslinking a rubber composition.
- the rubber composition includes:
- the base rubber (a) is crosslinked by the co-crosslinking agent (b).
- the heat of the crosslinking reaction remains near the central point of the core 4.
- the temperature at the central portion is high.
- the temperature gradually decreases from the central point toward the surface.
- the acid reacts with a metal salt of the co-crosslinking agent (b) to bond to cation.
- the salt reacts with the metal salt of the co-crosslinking agent (b) to exchange cation. By the bonding and the exchange, metallic bonding is broken.
- Examples of the base rubber (a) of the core 4 include polybutadienes, polyisoprenes, styrene-butadiene copolymers, ethylene-propylene-diene copolymers, and natural rubbers. In light of resilience performance, polybutadienes are preferred. When a polybutadiene and another rubber are used in combination, it is preferred that the polybutadiene is included as a principal component. Specifically, the proportion of the polybutadiene to the entire base rubber is preferably equal to or greater than 50% by weight and more preferably equal to or greater than 80% by weight. The proportion of cis-1,4 bonds in the polybutadiene is preferably equal to or greater than 40% by weight and more preferably equal to or greater than 80% by weight.
- a polybutadiene in which the proportion of 1,2-vinyl bonds is equal to or less than 2.0% by weight is preferred.
- the polybutadiene can contribute to the resilience performance of the core 4.
- the proportion of 1,2-vinyl bonds is preferably equal to or less than 1.7% by weight and particularly preferably equal to or less than 1.5% by weight.
- a rare-earth-element-containing catalyst is preferably used for synthesis of a polybutadiene.
- a polybutadiene synthesized with a catalyst containing neodymium, which is a lanthanum-series rare earth element compound, is preferred.
- the polybutadiene has a Mooney viscosity (ML 1+4 (100°C)) of preferably 30 or greater, more preferably 32 or greater, and particularly preferably 35 or greater.
- the Mooney viscosity (ML 1+4 (100°C)) is preferably equal to or less than 140, more preferably equal to or less than 120, even more preferably equal to or less than 100, and particularly preferably equal to or less than 80.
- the Mooney viscosity (ML 1+4 (100°C)) is measured according to the standards of "JIS K6300". The measurement conditions are as follows.
- the polybutadiene has a molecular weight distribution (Mw/Mn) of preferably 2.0 or greater, more preferably 2.2 or greater, even more preferably 2.4 or greater, and particularly preferably 2.6 or greater.
- Mw/Mn molecular weight distribution
- the molecular weight distribution (Mw/Mn) is preferably equal to or less than 6.0, more preferably equal to or less than 5.0, even more preferably equal to or less than 4.0, and particularly preferably equal to or less than 3.4.
- the molecular weight distribution (Mw/Mn) is calculated by dividing the weight average molecular weight Mw by the number average molecular weight Mn.
- the molecular weight distribution is measured by gel permeation chromatography ("HLC-8120GPC" manufactured by Tosoh Corporation).
- the measurement conditions are as follows.
- the co-crosslinking agent (b) is:
- the rubber composition may include only the ⁇ , ⁇ -unsaturated carboxylic acid (b1) or only the metal salt (b2) of the ⁇ , ⁇ -unsaturated carboxylic acid as the co-crosslinking agent (b).
- the rubber composition may include both the ⁇ , ⁇ -unsaturated carboxylic acid (b1) and the metal salt (b2) of the ⁇ , ⁇ -unsaturated carboxylic acid as the co-crosslinking agent (b).
- the metal salt (b2) of the ⁇ , ⁇ -unsaturated carboxylic acid graft-polymerizes with the molecular chain of the base rubber, thereby crosslinking the rubber molecules.
- the rubber composition preferably further includes a metal compound (f).
- the metal compound (f) reacts with the ⁇ , ⁇ -unsaturated carboxylic acid (b1) in the rubber composition.
- a salt obtained by this reaction graft-polymerizes with the molecular chain of the base rubber.
- Examples of the metal compound (f) include metal hydroxides such as magnesium hydroxide, zinc hydroxide, calcium hydroxide, sodium hydroxide, lithium hydroxide, potassium hydroxide, and copper hydroxide; metal oxides such as magnesium oxide, calcium oxide, zinc oxide, and copper oxide; and metal carbonates such as magnesium carbonate, zinc carbonate, calcium carbonate, sodium carbonate, lithium carbonate, and potassium carbonate.
- metal hydroxides such as magnesium hydroxide, zinc hydroxide, calcium hydroxide, sodium hydroxide, lithium hydroxide, potassium hydroxide, and copper hydroxide
- metal oxides such as magnesium oxide, calcium oxide, zinc oxide, and copper oxide
- metal carbonates such as magnesium carbonate, zinc carbonate, calcium carbonate, sodium carbonate, lithium carbonate, and potassium carbonate.
- a compound that includes a bivalent metal is preferred.
- the compound that includes the bivalent metal reacts with the co-crosslinking agent (b) to form metal crosslinks.
- the metal compound (f) is particularly preferably a zinc compound
- Examples of the ⁇ , ⁇ -unsaturated carboxylic acids include acrylic acid, methacrylic acid, fumaric acid, maleic acid, and crotonic acid.
- Examples of the metal component in the metal salt (b2) of the ⁇ , ⁇ -unsaturated carboxylic acid include sodium ion, potassium ion, lithium ion, magnesium ion, calcium ion, zinc ion, barium ion, cadmium ion, aluminum ion, tin ion, and zirconium ion.
- the metal salt (b2) of the ⁇ , ⁇ -unsaturated carboxylic acid may include two or more types of ions.
- bivalent metal ions such as magnesium ion, calcium ion, zinc ion, barium ion, and cadmium ion are preferred.
- the metal salt (b2) of the ⁇ , ⁇ -unsaturated carboxylic acid is particularly preferably zinc acrylate.
- the amount of the co-crosslinking agent (b) is preferably equal to or greater than 15 parts by weight and particularly preferably equal to or greater than 20 parts by weight, per 100 parts by weight of the base rubber. In light of feel at impact, the amount is preferably equal to or less than 50 parts by weight, more preferably equal to or less than 45 parts by weight, and particularly preferably equal to or less than 40 parts by weight, per 100 parts by weight of the base rubber.
- the crosslinking initiator (c) is preferably an organic peroxide.
- the organic peroxide contributes to the resilience performance of the golf ball 2.
- examples of preferable organic peroxides include dicumyl peroxide, 1,1-bis(t-butylperoxy)-3,3,5-trimethylcyclohexane, 2,5-dimethyl-2,5-di(t-butylperoxy)hexane, and di-t-butyl peroxide. In light of versatility, dicumyl peroxide is preferred.
- the amount of the crosslinking initiator (c) is preferably equal to or greater than 0.2 parts by weight and particularly preferably equal to or greater than 0.5 parts by weight, per 100 parts by weight of the base rubber. In light of feel at impact and durability of the golf ball 2, the amount is preferably equal to or less than 5.0 parts by weight and particularly preferably equal to or less than 2.5 parts by weight, per 100 parts by weight of the base rubber.
- the co-crosslinking agent (b) is not included in the concept of the acid and/or the salt (d) . It is inferred that as described above, the acid and/or the salt (d) breaks the metal crosslinks by the co-crosslinking agent (b) in the central portion of the core 4 during heating and forming of the core 4.
- the acid and/or the salt (d) is a carboxylic acid and/or a salt thereof (d1). Carboxylates are particularly preferred.
- the carboxylic acid component of the carboxylic acid and/or the salt thereof (d1) has a carboxyl group.
- the carbon number of the carboxylic acid component of the carboxylic acid and/or the salt thereof (d1) is preferably equal to or greater than 1 but equal to or less than 30, more preferably equal to or greater than 3 but equal to or less than 30, and even more preferably equal to or greater than 5 but equal to or less than 28.
- Examples of the carboxylic acid include aliphatic carboxylic acids (fatty acids) and aromatic carboxylic acids. Fatty acids and salts thereof are preferred.
- the rubber composition may include a saturated fatty acid or a salt thereof, or may include an unsaturated fatty acid or a salt thereof.
- the saturated fatty acid and the salt thereof are preferred.
- fatty acids examples include butyric acid (C4), valeric acid (C5), caproic acid (C6), enanthic acid (C7), caprylic acid (octanoic acid) (C8), pelargonic acid (C9), capric acid (C10), lauric acid (C12), myristic acid (C14), myristoleic acid (C14), pentadecylic acid (C15), palmitic acid (C16), palmitoleic acid (C16), margaric acid (C17), stearic acid (C18), elaidic acid (C18), vaccenic acid (C18), oleic acid (C18), linolic acid (C18), linolenic acid (C18), 12-hydroxystearic acid (C18), arachidic acid (C20), gadoleic acid (C20), arachidonic acid (C20), eicosenoic acid (C20), behenic acid (C22), erucic
- aromatic carboxylic acid has an aromatic ring and a carboxyl group.
- aromatic carboxylic acids include benzoic acid, phthalic acid, isophthalic acid, terephthalic acid, hemimellitic acid (benzene-1,2,3-tricarboxylic acid), trimellitic acid (benzene-1,2,4-tricarboxylic acid), trimesic acid (benzene-1,3,5-tricarboxylic acid), mellophanic acid (benzene-1,2,3,4-tetracarboxylic acid), prehnitic acid (benzene-1,2,3,5-tetracarboxylic acid), pyromellitic acid (benzene-1,2,4,5-tetracarboxylic acid), mellitic acid (benzene hexacarboxylic acid), diphenic acid (biphenyl-2,2'-dicarboxylic acid), toluic acid (methylbenzoic acid), xylic acid, prehnitylic acid (2,3,
- the rubber composition may include a salt of an aromatic carboxylic acid substituted with a hydroxyl group, an alkoxy group, or an oxo group.
- this carboxylic acid can include salicylic acid (2-hydroxybenzoic acid), anisic acid (methoxybenzoic acid), cresotinic acid (hydroxy(methyl) benzoic acid), o-homosalicylic acid (2-hydroxy-3-methylbenzoic acid), m-homosalicylic acid (2-hydroxy-4-methylbenzoic acid), p-homosalicylic acid (2-hydroxy-5-methylbenzoic acid), o-pyrocatechuic acid (2,3-dihydroxybenzoic acid), ⁇ -resorcylic acid (2,4-dihydroxybenzoic acid), ⁇ -resorcylic acid (2,6-dihydroxybenzoic acid), protocatechuic acid (3,4-dihydroxybenzoic acid), ⁇ -resorcylic acid (3,5-dihydroxybenzo
- the cationic component of the carboxylate is a metal ion or an organic cation.
- the metal ion include sodium ion, potassium ion, lithium ion, silver ion, magnesium ion, calcium ion, zinc ion, barium ion, cadmium ion, copper ion, cobalt ion, nickel ion, manganese ion, aluminum ion, iron ion, tin ion, zirconium ion, and titanium ion. Two or more types of ions may be used in combination.
- the organic cation is a cation having a carbon chain.
- Examples of the organic cation include organic ammonium ions.
- Examples of organic ammonium ions include primary ammonium ions such as stearylammonium ion, hexylammonium ion, octylammonium ion, and 2-ethylhexylammonium ion; secondary ammonium ions such as dodecyl (lauryl) ammonium ion, and octadecyl (stearyl) ammonium ion; tertiary ammonium ions such as trioctylammonium ion; and quaternary ammonium ions such as dioctyldimethylammonium ion, and distearyldimethylammonium ion. Two or more types of organic cations may be used in combination.
- preferable carboxylates include a potassium salt, a magnesium salt, an aluminum salt, a zinc salt, an iron salt, a copper salt, a nickel salt, or a cobalt salt of octanoic acid, lauric acid, myristic acid, palmitic acid, stearic acid, oleic acid, or behenic acid.
- Zinc salts of carboxylic acids are particularly preferred.
- Specific examples of preferable carboxylates include zinc octoate, zinc laurate, zinc myristate, and zinc stearate.
- the amount of the carboxylic acid and/or the salt (d1) is preferably equal to or greater than 0.5 parts by weight, more preferably equal to or greater than 1.0 parts by weight, even more preferably equal to or greater than 1.5 parts by weight, and particularly preferably equal to or greater than 2.0 parts by weight, per 100 parts by weight of the base rubber.
- the amount is preferably equal to or less than 40 parts by weight, more preferably less than 40 parts by weight, even more preferably equal to or less than 30 parts by weight, and particularly preferably equal to or less than 20 parts by weight, per 100 parts by weight of the base rubber.
- the weight ratio of the co-crosslinking agent (b) and the carboxylic acid and/or the salt (d1) in the rubber composition is preferably equal to or greater than 3/7 but equal to or less than 9/1, and is particularly preferably equal to or greater than 4/6 but equal to or less than 8/2. From the rubber composition in which this weight ratio is within the above range, the core 4 whose hardness linearly increases from its central point toward its surface can be obtained.
- zinc acrylate is preferably used as the co-crosslinking agent (b).
- Zinc acrylate whose surface is coated with stearic acid or zinc stearate for the purpose of improving dispersibility to rubber is present.
- this coating material is not included in the concept of the acid and/or the salt (d).
- the rubber composition preferably further includes an organic sulfur compound (e).
- the organic sulfur compound (e) can contribute to control of: the linearity of the hardness distribution of the core 4; and the degree of the outer-hard/inner-soft structure.
- An example of the organic sulfur compound (e) is an organic compound having a thiol group or a polysulfide linkage having 2 to 4 sulfur atoms. A metal salt of this organic compound is also included in the organic sulfur compound (e).
- organic sulfur compound (e) examples include aliphatic compounds such as aliphatic thiols, aliphatic thiocarboxylic acids, aliphatic dithiocarboxylic acids, and aliphatic polysulfides; heterocyclic compounds; alicyclic compounds such as alicyclic thiols, alicyclic thiocarboxylic acids, alicyclic dithiocarboxylic acids, and alicyclic polysulfides; and aromatic compounds.
- aliphatic compounds such as aliphatic thiols, aliphatic thiocarboxylic acids, aliphatic dithiocarboxylic acids, and aliphatic polysulfides
- heterocyclic compounds such as alicyclic thiols, alicyclic thiocarboxylic acids, alicyclic dithiocarboxylic acids, and alicyclic polysulfides
- aromatic compounds such as aliphatic thiols, aliphatic
- organic sulfur compound (e) examples include thiophenols, thionaphthols, polysulfides, thiocarboxylic acids, dithiocarboxylic acids, sulfenamides, thiurams, dithiocarbamates, and thiazoles.
- Preferable organic sulfur compounds (e) are thiophenols, polysulfides having 2 to 4 sulfur atoms, thionaphthols, thiurams, and metal salts thereof.
- organic sulfur compound (e) are represented by the following chemical formulas (1) to (4).
- R1 to R5 each represent H or a substituent.
- R1 to R10 each represent H or a substituent.
- R1 to R5 each represent H or a substituent, and M1 represents a monovalent metal atom.
- R1 to R10 each represent H or a substituent
- M2 represents a bivalent metal atom.
- each substituent is at least one group selected from the group consisting of a halogen group (F, Cl, Br, I), an alkyl group, a carboxyl group (-COOH), an ester (-COOR)of a carboxyl group, a formyl group (-CHO), an acyl group (-COR), a carbonyl halide group (-COX), a sulfo group (-SO 3 H), an ester (-SO 3 R) of a sulfo group, a sulfonyl halide group (-SO 2 X), a sulfino group (-SO 2 H), an alkylsulfinyl group (-SOR), a carbamoyl group (-CONH 2 ), an alkyl halide group, a cyano group (-CN), and an alkoxy group (-OR).
- a halogen group F, Cl, Br, I
- an alkyl group a carboxyl group (
- Examples of the organic sulfur compound represented by the chemical formula (1) include thiophenol; thiophenols substituted with halogen groups, such as 4-fluorothiophenol, 2,5-difluorothiophenol, 2,4,5-trifluorothiophenol, 2,4,5,6-tetrafluorothiophenol, pentafluorothiophenol, 2-chlorothiophenol, 4-chlorothiophenol, 2,4-dichlorothiophenol, 2,5-dichlorothiophenol, 2,4,5-trichlorothiophenol, 2,4,5,6-tetrachlorothiophenol, pentachlorothiophenol, 4-bromothiophenol, 2,5-dibromothiophenol, 2,4,5-tribromothiophenol, 2,4,5,6-tetrabromothiophenol, pentabromothiophenol, 4-iodothiophenol, 2,5-diiodothiophenol, 2,4,5-triiodothiophenol, 2,4,5,6
- organic sulfur compound represented by the chemical formula (1) is a compound substituted with at least one type of the above substituents and another substituent.
- substituents include a nitro group (-NO 2 ), an amino group (-NH 2 ), a hydroxyl group (-OH), and a phenylthio group (-SPh).
- the compound include 4-chloro-2-nitrothiophenol, 4-chloro-2-aminothiophenol, 4-chloro-2-hydroxythiophenol, 4-chloro-2-phenylthiothiophenol, 4-methyl-2-nitrothiophenol, 4-methyl-2-aminothiophenol, 4-methyl-2-hydroxythiophenol, 4-methyl-2-phenylthiothiophenol, 4-carboxy-2-nitrothiophenol, 4-carboxy-2-aminothiophenol, 4-carboxy-2-hydroxythiophenol, 4-carboxy-2-phenylthiothiophenol, 4-methoxycarbonyl-2-nitrothiophenol, 4-methoxycarbonyl-2-aminothiophenol, 4-methoxycarbonyl-2-hydroxythiophenol, 4-methoxycarbonyl-2-phenylthiothiophenol, 4-formyl-2-nitrothiophenol, 4-formyl-2-aminothiophenol, 4-formyl-2-hydroxythiophenol, 4-formyl-2-phenylthiophenol
- Still another example of the organic sulfur compound represented by the chemical formula (1) is a compound substituted with two or more types of substituents.
- the compound include 4-acetyl-2-chlorothiophenol, 4-acetyl-2-methylthiophenol, 4-acetyl-2-carboxythiophenol, 4-acetyl-2-methoxycarbonylthiophenol, 4-acetyl-2-formylthiophenol, 4-acetyl-2-chlorocarbonylthiophenol, 4-acetyl-2-sulfothiophenol, 4-acetyl-2-methoxysulfonylthiophenol, 4-acetyl-2-chlorosulfonylthiophenol, 4-acetyl-2-sulfinothiophenol, 4-acetyl-2-methylsulfinylthiophenol, 4-acetyl-2-carbamoylthiophenol, 4-acetyl-2-trichloromethylthiophenol, 4-acetyl-2-cyano
- Examples of the organic sulfur compound represented by the chemical formula (2) include diphenyl disulfide; diphenyl disulfides substituted with halogen groups, such as bis(4-fluorophenyl)disulfide, bis(2,5-difluorophenyl)disulfide, bis(2,4,5-trifluorophenyl)disulfide, bis(2,4,5,6-tetrafluorophenyl)disulfide, bis(pentafluorophenyl)disulfide, bis(4-chlorophenyl)disulfide, bis(2,5-dichlorophenyl)disulfide, bis(2,4,5-trichlorophenyl)disulfide, bis(2,4,5,6-tetrachlorophenyl)disulfide, bis(pentachlorophenyl)disulfide, bis(4-bromophenyl)disulfide, bis(2,5-
- organic sulfur compound represented by the chemical formula (2) is a compound substituted with at least one type of the above substituents and another substituent.
- substituents include a nitro group (-NO 2 ), an amino group (-NH 2 ), a hydroxyl group (-OH), and a phenylthio group (-SPh).
- the compound include bis(4-chloro-2-nitrophenyl)disulfide, bis(4-chloro-2-aminophenyl)disulfide, bis(4-chloro-2-hydroxyphenyl)disulfide, bis(4-chloro-2-phenylthiophenyl)disulfide, bis(4-methyl-2-nitrophenyl)disulfide, bis(4-methyl-2-aminophenyl)disulfide, bis(4-methyl-2-hydroxyphenyl)disulfide, bis(4-methyl-2-phenylthiophenyl)disulfide, bis(4-carboxy-2-nitrophenyl)disulfide, bis(4-carboxy-2-aminophenyl)disulfide, bis(4-carboxy-2-hydroxyphenyl)disulfide, bis(4-carboxy-2-phenylthiophenyl)disulfide, bis
- Still another example of the organic sulfur compound represented by the chemical formula (2) is a compound substituted with two or more types of substituents.
- the compound include bis(4-acetyl-2-chlorophenyl)disulfide, bis(4-acetyl-2-methylphenyl)disulfide, bis(4-acetyl-2-carboxyphenyl)disulfide, bis(4-acetyl-2-methoxycarbonylphenyl)disulfide, bis(4-acetyl-2-formylphenyl)disulfide, bis(4-acetyl-2-chlorocarbonylphenyl)disulfide, bis(4-acetyl-2-sulfophenyl)disulfide, bis(4-acetyl-2-methoxysulfonylphenyl)disulfide, bis(4-acetyl-2-chlorosulfonylphenyl)d
- Examples of the organic sulfur compound represented by the chemical formula (3) include thiophenol sodium salt; thiophenol sodium salts substituted with halogen groups, such as 4-fluorothiophenol sodium salt, 2,5-difluorothiophenol sodium salt, 2,4,5-trifluorothiophenol sodium salt, 2,4,5,6-tetrafluorothiophenol sodium salt, pentafluorothiophenol sodium salt, 4-chlorothiophenol sodium salt, 2,5-dichlorothiophenol sodium salt, 2,4,5-trichlorothiophenol sodium salt, 2,4,5,6-tetrachlorothiophenol sodium salt, pentachlorothiophenol sodium salt, 4-bromothiophenol sodium salt, 2,5-dibromothiophenol sodium salt, 2,4,5-tribromothiophenol sodium salt, 2,4,5,6-tetrabromothiophenol sodium salt, pentabromothiophenol sodium salt, 4-iodothiophenol sodium salt, 2,5-diiodothiophenol
- organic sulfur compound represented by the chemical formula (3) is a compound substituted with at least one type of the above substituents and another substituent.
- substituents include a nitro group (-NO 2 ), an amino group (-NH 2 ), a hydroxyl group (-OH), and a phenylthio group (-SPh).
- the compound include 4-chloro-2-nitrothiophenol sodium salt, 4-chloro-2-aminothiophenol sodium salt, 4-chloro-2-hydroxythiophenol sodium salt, 4-chloro-2-phenylthiothiophenol sodium salt, 4-methyl-2-nitrothiophenol sodium salt, 4-methyl-2-aminothiophenol sodium salt, 4-methyl-2-hydroxythiophenol sodium salt, 4-methyl-2-phenylthiothiophenol sodium salt, 4-carboxy-2-nitrothiophenol sodium salt, 4-carboxy-2-aminothiophenol sodium salt, 4-carboxy-2-hydroxythiophenol sodium salt, 4-carboxy-2-phenylthiothiophenol sodium salt, 4-methoxycarbonyl-2-nitrothiophenol sodium salt, 4-methoxycarbonyl-2-aminothiophenol sodium salt, 4-methoxycarbonyl-2-hydroxythiophenol sodium salt, 4-methoxycarbonyl-2-phenylthiothiophenol sodium salt, 4-formyl-2-nitrothiophenol sodium salt,
- Still another example of the organic sulfur compound represented by the chemical formula (3) is a compound substituted with two or more types of substituents.
- the compound include 4-acetyl-2-chlorothiophenol sodium salt, 4-acetyl-2-methylthiophenol sodium salt, 4-acetyl-2-carboxythiophenol sodium salt, 4-acetyl-2-methoxycarbonylthiophenol sodium salt, 4-acetyl-2-formylthiophenol sodium salt, 4-acetyl-2-chlorocarbonylthiophenol sodium salt, 4-acetyl-2-sulfothiophenol sodium salt, 4-acetyl-2-methoxysulfonylthiophenol sodium salt, 4-acetyl-2-chlorosulfonylthiophenol sodium salt, 4-acetyl-2-sulfinothiophenol sodium salt, 4-acetyl-2-methylsulfinylthiophenol sodium salt, 4-acetyl-2-carbamoylthiophenol sodium salt, 4-
- Examples of the organic sulfur compound represented by the chemical formula (4) include thiophenol zinc salt; thiophenol zinc salts substituted with halogen groups, such as 4-fluorothiophenol zinc salt, 2,5-difluorothiophenol zinc salt, 2,4,5-trifluorothiophenol zinc salt, 2,4,5,6-tetrafluorothiophenol zinc salt, pentafluorothiophenolzinc salt, 4-chlorothiophenol zinc salt, 2,5-dichlorothiophenol zinc salt, 2,4,5-trichlorothiophenol zinc salt, 2,4,5,6-tetrachlorothiophenol zinc salt, pentachlorothiophenol zinc salt, 4-bromothiophenol zinc salt, 2,5-dibromothiophenol zinc salt, 2,4,5-tribromothiophenol zinc salt, 2,4,5,6-tetrabromothiophenol zinc salt, pentabromothiophenol zinc salt, 4-iodothiophenol zinc salt, 2,5-diiodothi
- organic sulfur compound represented by the chemical formula (4) is a compound substituted with at least one type of the above substituents and another substituent.
- substituents include a nitro group (-NO 2 ), an amino group (-NH 2 ), a hydroxyl group (-OH), and a phenylthio group (-SPh).
- the compound include 4-chloro-2-nitrothiophenol zinc salt, 4-chloro-2-aminothiophenol zinc salt, 4-chloro-2-hydroxythiophenol zinc salt, 4-chloro-2-phenylthiothiophenol zinc salt, 4-methyl-2-nitrothiophenol zinc salt, 4-methyl-2-aminothiophenol zinc salt, 4-methyl-2-hydroxythiophenol zinc salt, 4-methyl-2-phenylthiothiophenol zinc salt, 4-carboxy-2-nitrothiophenol zinc salt, 4-carboxy-2-aminothiophenol zinc salt, 4-carboxy-2-hydroxythiophenol zinc salt, 4-carboxy-2-phenylthiothiophenol zinc salt, 4-methoxycarbonyl-2-nitrothiophenol zinc salt, 4-methoxycarbonyl-2-aminothiophenol zinc salt, 4-methoxycarbonyl-2-hydroxythiophenol zinc salt, 4-methoxycarbonyl-2-phenylthiothiophenol zinc salt, 4-formyl-2-nitrothiophenol zinc salt,
- Still another example of the organic sulfur compound represented by the chemical formula (4) is a compound substituted with two or more types of substituents.
- the compound include 4-acetyl-2-chlorothiophenol zinc salt, 4-acetyl-2-methylthiophenol zinc salt, 4-acetyl-2-carboxythiophenol zinc salt, 4-acetyl-2-methoxycarbonylthiophenol zinc salt, 4-acetyl-2-formylthiophenol zinc salt, 4-acetyl-2-chlorocarbonylthiophenol zinc salt, 4-acetyl-2-sulfothiophenol zinc salt, 4-acetyl-2-methoxysulfonylthiophenol zinc salt, 4-acetyl-2-chlorosulfonylthiophenol zinc salt, 4-acetyl-2-sulfinothiophenol zinc salt, 4-acetyl-2-methylsulfinylthiophenol zinc salt, 4-acetyl-2-carbamoylthiophenol zinc salt,
- Examples of the bivalent metal represented by M2 in the chemical formula (4) include zinc, magnesium, calcium, strontium, barium, titanium (II), manganese (II), iron (II), cobalt (II), nickel (II), zirconium (II), and tin (II).
- thionaphthols examples include 2-thionaphthol, 1-thionaphthol, 2-chloro-1-thionaphthol, 2-bromo-1-thionaphthol, 2-fluoro-1-thionaphthol, 2-cyano-1-thionaphthol, 2-acetyl-1-thionaphthol, 1-chloro-2-thionaphthol, 1-bromo-2-thionaphthol, 1-fluoro-2-thionaphthol, 1-cyano-2-thionaphthol, 1-acetyl-2-thionaphthol, and metal salts thereof.
- 1-thionaphthol, 2-thionaphthol, and zinc salts thereof are preferred.
- Examples of sulfenamide type organic sulfur compounds include N-cyclohexyl-2-benzothiazole sulfenamide, N-oxydiethylene-2-benzothiazole sulfenamide, and N-t-butyl-2-benzothiazole sulfenamide.
- Examples of thiuram type organic sulfur compounds include tetramethylthiuram monosulfide, tetramethylthiuram disulfide, tetraethylthiuram disulfide, tetrabutylthiuram disulfide, and dipentamethylenethiuram tetrasulfide.
- dithiocarbamates include zinc dimethyldithiocarbamate, zinc diethyldithiocarbamate, zinc dibutyldithiocarbamate, zinc ethylphenyldithiocarbamate, sodium dimethyldithiocarbamate, sodium diethyldithiocarbamate, copper (II) dimethyldithiocarbamate, iron (III) dimethyldithiocarbamate, selenium diethyldithiocarbamate, and tellurium diethyldithiocarbamate.
- thiazole type organic sulfur compounds examples include 2-mercaptobenzothiazole (MBT); dibenzothiazyl disulfide (MBTS); a sodium salt, a zinc salt, a copper salt, or a cyclohexylamine salt of 2-mercaptobenzothiazole; 2-(2,4-dinitrophenyl)mercaptobenzothiazole; and 2-(2,6-diethyl-4-morpholinothio)benzothiazole.
- MBT 2-mercaptobenzothiazole
- MBTS dibenzothiazyl disulfide
- sodium salt a zinc salt, a copper salt, or a cyclohexylamine salt of 2-mercaptobenzothiazole
- 2-(2,4-dinitrophenyl)mercaptobenzothiazole 2-(2,6-diethyl-4-morpholinothio)benzothiazole.
- the amount of the organic sulfur compound (e) is preferably equal to or greater than 0.05 parts by weight and particularly preferably equal to or greater than 0.1 parts by weight, per 100 parts by weight of the base rubber. In light of resilience performance, the amount is preferably equal to or less than 5.0 parts by weight and particularly preferably equal to or less than 2.0 parts by weight, per 100 parts by weight of the base rubber.
- a filler may be included in the core 4.
- suitable fillers include zinc oxide, barium sulfate, calcium carbonate, and magnesium carbonate.
- the amount of the filler is determined as appropriate so that the intended specific gravity of the core 4 is accomplished.
- a particularly preferable filler is zinc oxide. Zinc oxide serves not only as a specific gravity adjuster but also as a crosslinking activator.
- an anti-aging agent a coloring agent, a plasticizer, a dispersant, sulfur, a vulcanization accelerator, and the like are added to the rubber composition of the core 4.
- Crosslinked rubber powder or synthetic resin powder may also be dispersed in the rubber composition.
- the difference (Hs-H(0)) between the surface hardness Hs and the central hardness H(0) is preferably equal to or greater than 15.
- the difference is great.
- the core 4 has an outer-hard/inner-soft structure.
- the recoil (torsional return) is great, and thus spin is suppressed.
- the core 4 contributes to the flight performance of the golf ball 2.
- the difference (Hs-H(0)) is more preferably equal to or greater than 20 and particularly preferably equal to or greater than 25. From the standpoint that the core 4 can easily be formed, the difference (Hs-H(0)) is preferably equal to or less than 50.
- the hardness H(0) at the central point of the core 4 is preferably equal to or greater than 40.0 but equal to or less than 70.0.
- the golf ball 2 having a hardness H(0) of 40.0 or greater has excellent resilience performance.
- the hardness H(0) is more preferably equal to or greater than 45.0 and particularly preferably equal to or greater than 50.0.
- the core 4 having a hardness H(0) of 70.0 or less can achieve an outer-hard/inner-soft structure.
- spin can be suppressed.
- the hardness H(0) is more preferably equal to or less than 68.0 and particularly preferably equal to or less than 66.0.
- the hardness Hs at the surface of the core 4 is preferably equal to or greater than 78.0 but equal to or less than 95.0.
- the core 4 having a hardness Hs of 78.0 or greater an outer-hard/inner-soft structure can be achieved.
- spin can be suppressed.
- the hardness Hs is more preferably equal to or greater than 80.0 and particularly preferably equal to or greater than 82.0.
- the golf ball 2 having a hardness Hs of 95.0 or less has excellent durability.
- the hardness Hs is more preferably equal to or less than 93.0 and particularly preferably equal to or less than 90.0.
- the core 4 preferably has a diameter of 38.0 mm or greater but 42.0 mm or less.
- the core 4 having a diameter of 38.0 mm or greater can achieve excellent resilience performance of the golf ball 2.
- the diameter is more preferably equal to or greater than 38.5 mm and particularly preferably equal to or greater than 39.0 mm.
- the inner cover 8 and the outer cover 10 can have sufficient thicknesses.
- the golf ball 2 that includes the inner cover 8 and the outer cover 10 which have large thicknesses has excellent durability.
- the diameter is more preferably equal to or less than 41.0 mm and particularly preferably equal to or less than 40.0 mm.
- the core 4 has an amount of compressive deformation Dc of preferably 3.0 mm or greater and particularly preferably 3.3 mm or greater.
- the amount of compressive deformation Dc is preferably equal to or less than 4.6 mm and particularly preferably equal to or less than 4.3 mm.
- a resin composition is suitably used.
- the base polymer of the resin composition include ionomer resins, styrene block-containing thermoplastic elastomers, thermoplastic polyester elastomers, thermoplastic polyamide elastomers, and thermoplastic polyolefin elastomers.
- Particularly preferable base polymers are ionomer resins.
- the golf ball 2 that includes the inner cover 8 including an ionomer resin has excellent resilience performance.
- An ionomer resin and another resin may be used in combination for the inner cover 8.
- the principal component of the base polymer is preferably the ionomer resin.
- the proportion of the ionomer resin to the entire base polymer is preferably equal to or greater than 50% by weight, more preferably equal to or greater than 60% by weight, and particularly preferably equal to or greater than 70% by weight.
- preferable ionomer resins include binary copolymers formed with an ⁇ -olefin and an ⁇ , ⁇ -unsaturated carboxylic acid having 3 to 8 carbon atoms.
- a preferable binary copolymer includes 80% by weight or greater but 90% by weight or less of an ⁇ -olefin, and 10% by weight or greater but 20% by weight or less of an ⁇ , ⁇ -unsaturated carboxylic acid.
- the binary copolymer has excellent resilience performance.
- Examples of other preferable ionomer resins include ternary copolymers formed with: an ⁇ -olefin; an ⁇ , ⁇ -unsaturated carboxylic acid having 3 to 8 carbon atoms; and an ⁇ , ⁇ -unsaturated carboxylate ester having 2 to 22 carbon atoms.
- a preferable ternary copolymer includes 70% by weight or greater but 85% by weight or less of an ⁇ -olefin, 5% by weight or greater but 30% by weight or less of an ⁇ , ⁇ -unsaturated carboxylic acid, and 1% by weight or greater but 25% by weight or less of an ⁇ , ⁇ -unsaturated carboxylate ester.
- the ternary copolymer has excellent resilience performance.
- ⁇ -olefins are ethylene and propylene, while preferable ⁇ , ⁇ -unsaturated carboxylic acids are acrylic acid and methacrylic acid.
- a particularly preferable ionomer resin is a copolymer formed with ethylene and acrylic acid or methacrylic acid.
- some of the carboxyl groups are neutralized with metal ions.
- metal ions for use in neutralization include sodium ion, potassium ion, lithium ion, zinc ion, calcium ion, magnesium ion, aluminum ion, and neodymium ion.
- the neutralization may be carried out with two or more types of metal ions.
- Particularly suitable metal ions in light of resilience performance and durability of the golf ball 2 are sodium ion, zinc ion, lithium ion, and magnesium ion.
- ionomer resins include trade names "Himilan 1555”, “Himilan 1557”, “Himilan 1605", “Himilan 1706", “Himilan 1707”, “Himilan 1856", “Himilan 1855”, “Himilan AM7311”, “Himilan AM7315”, “Himilan AM7317”,”Himilan AM7318”, “Himilan AM7329”, “Himilan AM7337”, “Himilan MK7320”, and “Himilan MK7329”, manufactured by Du Pont-MITSUI POLYCHEMICALS Co., Ltd.; trade names “Surlyn 6120”, “Surlyn 6910", “Surlyn 7930”, “Surlyn 7940", “Surlyn 8140”, “Surlyn 8150”, “Surlyn 8940", “Surlyn 8945”, “Surlyn 9120”, “Surlyn 9150”, “Surlyn 9910", “Surlyn 9945”, "Surlyn
- Two or more ionomer resins may be used in combination for the inner cover 8.
- An ionomer resin neutralized with a monovalent metal ion, and an ionomer resin neutralized with a bivalent metal ion may be used in combination.
- a preferable resin that can be used in combination with an ionomer resin is a styrene block-containing thermoplastic elastomer.
- the styrene block-containing thermoplastic elastomer has excellent compatibility with ionomer resins.
- a resin composition including the styrene block-containing thermoplastic elastomer has excellent fluidity.
- the styrene block-containing thermoplastic elastomer includes a polystyrene block as a hard segment, and a soft segment.
- a typical soft segment is a diene block.
- Examples of compounds for the diene block include butadiene, isoprene, 1, 3-pentadiene, and 2,3-dimethyl-1,3-butadiene. Butadiene and isoprene are preferred. Two or more compounds may be used in combination.
- styrene block-containing thermoplastic elastomers examples include styrene-butadiene-styrene block copolymers (SBS), styrene-isoprene-styrene block copolymers (SIS), styrene-isoprene-butadiene-styrene block copolymers (SIBS), hydrogenated SBS, hydrogenated SIS, and hydrogenated SIBS.
- hydrogenated SBS include styrene-ethylene-butylene-styrene block copolymers (SEBS).
- hydrogenated SIS examples include styrene-ethylene-propylene-styrene block copolymers (SEPS).
- SIBS styrene-ethylene-ethylene-propylene-styrene block copolymers
- the content of the styrene component in the styrene block-containing thermoplastic elastomer is preferably equal to or greater than 10% by weight, more preferably equal to or greater than 12% by weight, and particularly preferably equal to or greater than 15% by weight.
- the content is preferably equal to or less than 50% by weight, more preferably equal to or less than 47% by weight, and particularly preferably equal to or less than 45% by weight.
- styrene block-containing thermoplastic elastomers include an alloy of an olefin and one or more members selected from the group consisting of SBS, SIS, SIBS, SEBS, SEPS, SEEPS, and hydrogenated products thereof.
- the olefin component in the alloy is presumed to contribute to improvement of compatibility with ionomer resins. Use of this alloy improves the resilience performance of the golf ball 2.
- An olefin having 2 to 10 carbon atoms is preferably used. Examples of suitable olefins include ethylene, propylene, butene, and pentene. Ethylene and propylene are particularly preferred.
- polymer alloys include trade names “Rabalon T3221C”, “Rabalon T3339C”, “Rabalon SJ4400N”, “Rabalon SJ5400N”, “Rabalon SJ6400N”, “Rabalon SJ7400N”, “Rabalon SJ8400N”, “Rabalon SJ9400N”, and “Rabalon SR04", manufactured by Mitsubishi Chemical Corporation.
- Other specific examples of styrene block-containing thermoplastic elastomers include trade name “Epofriend A1010” manufactured by Daicel Chemical Industries, Ltd., and trade name “Septon HG-252" manufactured by Kuraray Co., Ltd.
- a coloring agent such as titanium dioxide and a fluorescent pigment
- a filler such as barium sulfate, a dispersant, an antioxidant, an ultraviolet absorber, a light stabilizer, a fluorescent material, a fluorescent brightener, and the like are included in the resin composition of the inner cover 8 in an adequate amount.
- the inner cover 8 may include powder of a metal with a high specific gravity.
- the inner cover 8 has a hardness Hi of preferably 80 or greater, more preferably 83 or greater, and particularlypreferably 85 or greater.
- the hardness Hi is preferably equal to or less than 95 and particularly preferably equal to or less than 90.
- the hardness Hi is measured with a JIS-C type hardness scale mounted to an automated rubber hardness measurement machine (trade name "P1", manufactured by Kobunshi Keiki Co., Ltd.). For the measurement, a slab that is formed by hot press and that has a thickness of about 2 mm is used. A slab kept at 23°C for two weeks is used for the measurement. At the measurement, three slabs are stacked. A slab formed from the same resin composition as the resin composition of the inner cover 8 is used.
- the hardness Hi of the inner cover 8 is preferably greater than the surface hardness Hs of the core 4.
- the difference (Hi-Hs) between the hardness Hi and the hardness Hs is preferably equal to or greater than 1 and particularly preferably equal to or greater than 2.
- the difference (Hi-Hs) is preferably equal to or less than 5.
- the hardness linearly increases from its central point toward its surface.
- the energy loss is low when the golf ball 2 is hit with a middle iron.
- the inner cover 8 preferably has a thickness of 0.2 mm or greater but 2.0 mm or less.
- an outer-hard/inner-soft structure can be achieved.
- the thickness is more preferably equal to or greater than 0.5 mm and particularly preferably equal to or greater than 0.8 mm.
- the golf ball 2 that includes the inner cover 8 having a thickness of 2.0 mm or less has excellent resilience performance.
- the thickness is more preferably equal to or less than 1.6 mm and particularly preferably equal to or less than 1.3 mm.
- the sphere 16 consisting of the core 4 and the inner cover 8 has an amount of compressive deformation Di of preferably 3.2 mm or greater and particularly preferably 3.4 mm or greater.
- the amount of compressive deformation Di is preferably equal to or less than 3.8 mm and particularly preferably equal to or less than 3.6 mm.
- the inner cover 8 For forming the inner cover 8, known methods such as injection molding, compression molding, and the like can be used.
- a resin composition is suitably used for the outer cover 10.
- a preferable base polymer of the resin composition is an ionomer resin.
- the golf ball 2 that includes the outer cover 10 including the ionomer resin has excellent resilience performance.
- the ionomer resin described above for the inner cover 8 can be used for the outer cover 10.
- an ionomer resin and another resin may be used in combination.
- the ionomer resin is included as the principal component of the base polymer.
- the proportion of the ionomer resin to the entire base polymer is preferably equal to or greater than 50% by weight, more preferably equal to or greater than 60% by weight, and particularly preferably equal to or greater than 70% by weight.
- a preferable resin that can be used in combination with an ionomer resin is an ethylene-(meth)acrylic acid copolymer.
- the copolymer is obtained by a copolymerization reaction of a monomer composition that contains ethylene and (meth)acrylic acid. In the copolymer, some of the carboxyl groups are neutralized with metal ions.
- the copolymer includes 3% by weight or greater but 25% by weight or less of a (meth)acrylic acid component.
- An ethylene-(meth)acrylic acid copolymer having a polar functional group is particularly preferred.
- a specific example of ethylene- (meth) acrylic acid copolymers is trade name "NUCREL" manufactured by Du Pont-MITSUI POLYCHEMICALS Co., Ltd.
- Another preferable resin that can be used in combination with an ionomer resin is a styrene block-containing thermoplastic elastomer.
- the styrene block-containing thermoplastic elastomer described above for the inner cover 8 can be used for the outer cover 10.
- a coloring agent such as titanium dioxide and a fluorescent pigment
- a filler such as barium sulfate, a dispersant, an antioxidant, an ultraviolet absorber, a light stabilizer, a fluorescent material, a fluorescent brightener, and the like are included in the outer cover 10 in an adequate amount.
- the outer cover 10 preferably has a JIS-C hardness Ho of 83 or greater but 96 or less.
- a hardness Ho of 83 or greater an outer-hard/inner-soft structure can be achieved.
- spin is suppressed.
- the golf ball 2 has excellent flight performance.
- the hardness Ho is more preferably equal to or greater than 84 and particularly preferably equal to or greater than 85.
- the golf ball 2 that includes the outer cover 10 having a hardness Ho of 96 or less has excellent feel at impact.
- the hardness Ho is more preferably equal to or less than 95 and particularly preferably equal to or less than 93.
- the hardness Ho is measured by the same measurement method as that for the hardness Hi.
- the outer cover 10 preferably has a thickness of 0.2 mm or greater but 1.5 mm or less.
- the outer cover 10 having a thickness of 0.2 mm or greater can easily be formed.
- the thickness is more preferably equal to or greater than 0.4 mm and particularly preferably equal to or greater than 0.6 mm.
- spin is suppressed.
- the thickness is more preferably equal to or less than 1.3 mm and particularly preferably equal to or less than 1.1 mm.
- the outer cover 10 For forming the outer cover 10, known methods such as injection molding, compression molding, and the like can be used. When forming the outer cover 10, the dimples 12 are formed by pimples formed on the cavity face of a mold.
- the cover 6 preferably has a total thickness of 2.5 mm or less.
- the golf ball 2 that includes the cover 6 having a total thickness of 2.5 mm or less has excellent feel at impact.
- the total thickness is more preferably equal to or less than 2.3 mm and particularly preferably equal to or less than 2.1 mm.
- the total thickness is preferably equal to or greater than 0.3 mm, more preferably equal to or greater than 0.5 mm, and particularly preferably equal to or greater than 0.8 mm.
- the JIS-C hardness Ho of the outer cover 10 is greater than the JIS-C hardness Hi of the inner cover 8.
- the outer cover 10 can achieve an outer-hard/inner-soft structure of the golf ball 2.
- the golf ball 2 has excellent flight performance and excellent feel at impact.
- the difference (Ho-Hi) is preferably equal to or greater than 2, more preferably equal to or greater than 4, and particularly preferably equal to or greater than 6. In light of suppression of energy loss when the golf ball 2 is hit, the difference (Ho-Hi) is preferably equal to or less than 10.
- the golf ball 2 has an amount of compressive deformation Db of preferably 2.8 mm or greater, more preferably 2.9 mm or greater, and particularly preferably 3.0 mm or greater.
- the amount of compressive deformation Db is preferably equal to or less than 3.6 mm, more preferably equal to or less than 3.5 mm, and particularly preferably equal to or less than 3.4 mm.
- a sphere such as the core 4, the golf ball 2, or the like is placed on a hard plate made of metal.
- a cylinder made of metal gradually descends toward the sphere.
- a migration distance of the cylinder starting from the state in which an initial load of 98 N is applied to the sphere up to the state in which a final load of 1274 N is applied thereto, is measured.
- a golf ball 102 shown in FIG. 3 includes a spherical core 104 and a cover 106 covering the core 104.
- the cover 106 includes an inner cover 108 and an outer cover 110 positioned outside the inner cover 108.
- the inner cover 108 is an innermost layer of the cover 106.
- the outer cover 110 is an outermost layer of the cover 106.
- the cover 106 may include another one or more layers between the inner cover 108 and the outer cover 110.
- On the surface of the outer cover 110 a large number of dimples 112 are formed. Of the surface of the golf ball 102, a part other than the dimples 112 is a land 114.
- the golf ball 102 includes a paint layer and a mark layer on the external side of the outer cover 110, but these layers are not shown in the drawing.
- the golf ball 102 has a diameter of 40 mm or greater but 45 mm or less. From the standpoint of conformity to the rules established by the United States Golf Association (USGA), the diameter is preferably equal to or greater than 42.67 mm. In light of suppression of air resistance, the diameter is preferably equal to or less than 44 mm and more preferably equal to or less than 42.80 mm.
- the golf ball 102 has a weight of 40 g or greater but 50 g or less. In light of attainment of great inertia, the weight is preferably equal to or greater than 44 g and more preferably equal to or greater than 45.00 g. From the standpoint of conformity to the rules established by the USGA, the weight is preferably equal to or less than 45.93 g.
- FIG. 4 is a line graph showing a hardness distribution of the core 104 of the golf ball 102 in FIG. 3 .
- the horizontal axis of the graph indicates the ratio (%) of a distance from the central point of the core 104 to the radius of the core 104.
- the vertical axis of the graph indicates a JIS-C hardness.
- Nine measuring points obtained by dividing a region from the central point of the core 104 to the surface of the core 104 at intervals of 12.5% of the radius of the core 104 are plotted in the graph.
- the ratio of the distance from the central point of the core 104 to each of these measuring points to the radius of the core 104 is as follows.
- FIG. 4 also shows a linear approximation curve obtained by a least-square method on the basis of the distances and the hardnesses of the nine measuring points.
- the broken line does not greatly deviate from the linear approximation curve.
- the broken line has a shape close to the linear approximation curve.
- the hardness linearly increases from its central point toward its surface.
- the core 104 has excellent resilience performance.
- Inthegolfball 102 that includes the core 104 spin is suppressed. When the golf ball 102 is hit with a driver, the flight distance is large.
- R 2 of the linear approximation curve obtained by the least-square method is equal to or greater than 0.95.
- R 2 is an index indicating the linearity of the broken line.
- the shape of the broken line of the hardness distribution is close to a straight line.
- the core 104 for which R 2 is equal to or greater than 0.95 has excellent resilience performance.
- R 2 is more preferably equal to or greater than 0.96 and particularly preferably equal to or greater than 0.97.
- R 2 is calculated by squaring a correlation coefficient R.
- the correlation coefficient R is calculated by dividing the covariance of the distance (%) from the central point and the hardness (JIS-C) by the standard deviation of the distance (%) from the central point and the standard deviation of the hardness (JIS-C).
- the core 104 is obtained by crosslinking a rubber composition.
- the rubber composition includes:
- the base rubber (a) is crosslinked by the co-crosslinking agent (b).
- the heat of the crosslinking reaction remains near the central point of the core 104.
- the temperature at the central portion is high.
- the temperature gradually decreases from the central point toward the surface.
- the acid reacts with a metal salt of the co-crosslinking agent (b) to bond to cation.
- the salt reacts with the metal salt of the co-crosslinking agent (b) to exchange cation. By the bonding and the exchange, metallic bonding is broken.
- This breaking is likely to occur in the central portion of the core 104 where the temperature is high, and is unlikely to occur near the surface of the core 104.
- the crosslinking density of the core 104 increases from its central point toward its surface.
- an outer-hard/inner-soft structure can be achieved.
- the rubber composition includes an organic sulfur compound (e) together with the acid and/or the salt (d)
- the gradient of the hardness distribution can be controlled, and the degree of the outer-hard/inner-soft structure of the core 104 can be increased.
- the spin rate is low.
- excellent flight performance is achieved upon a shot with a driver.
- the rubber composition of the core 104 can include, as the base rubber (a), the base rubber (a) described above for the rubber composition of the core 4 according to the first embodiment.
- the rubber composition of the core 104 can include, as the co-crosslinking agent (b), the co-crosslinking agent (b) described above for the rubber composition of the core 4 according to the first embodiment.
- the co-crosslinking agent (b) is:
- the rubber composition may include only the ⁇ , ⁇ -unsaturated carboxylic acid (b1) or only the metal salt (b2) of the ⁇ , ⁇ -unsaturated carboxylic acid as the co-crosslinking agent (b).
- the rubber composition may include both the ⁇ , ⁇ -unsaturated carboxylic acid (b1) and the metal salt (b2) of the ⁇ , ⁇ -unsaturated carboxylic acid as the co-crosslinking agent (b).
- the metal salt (b2) of the ⁇ , ⁇ -unsaturated carboxylic acid graft-polymerizes with the molecular chain of the base rubber, thereby crosslinking the rubber molecules.
- the rubber composition preferably further includes a metal compound (f).
- the metal compound (f) reacts with the ⁇ , ⁇ -unsaturated carboxylic acid (b1) in the rubber composition.
- a salt obtained by this reaction graft-polymerizes with the molecular chain of the base rubber.
- the rubber composition of the core 104 can include, as the metal compound (f), the metal compound (f) described above for the rubber composition of the core 4 according to the first embodiment.
- the amount of the co-crosslinking agent (b) is preferably equal to or greater than 15 parts by weight and particularly preferably equal to or greater than 20 parts by weight, per 100 parts by weight of the base rubber. In light of feel at impact, the amount is preferably equal to or less than 50 parts by weight, more preferably equal to or less than 45 parts by weight, and particularly preferably equal to or less than 40 parts by weight, per 100 parts by weight of the base rubber.
- the rubber composition of the core 104 can include, as the crosslinking initiator (c), the crosslinking initiator (c) described above for the rubber composition of the core 4 according to the first embodiment.
- the amount of the crosslinking initiator (c) is preferably equal to or greater than 0.2 parts by weight and particularly preferably equal to or greater than 0.5 parts by weight, per 100 parts by weight of the base rubber. In light of feel at impact and durability of the golf ball 102, the amount is preferably equal to or less than 5. 0 parts by weight and particularly preferably equal to or less than 2.5 parts by weight, per 100 parts by weight of the base rubber.
- the rubber composition of the core 104 includes, as the acid and/or the salt (d) the carboxylic acid and/or the salt (d1) described above for the rubber composition of the core 4 according to the first embodiment.
- the co-crosslinking agent (b) is not included in the concept of the acid and/or the salt (d). It is inferred that as described above, the acid and/or the salt (d) breaks the metal crosslinks by the co-crosslinking agent (b) in the central portion of the core 104 during heating and forming of the core 104. Carboxylates are particularly preferred.
- the carboxylic acid component of the carboxylic acid and/or the salt thereof (d1) has a carboxyl group.
- the carbon number of the carboxylic acid component of the carboxylic acid and/or the salt thereof (d1) is preferably equal to or greater than 1 but equal to or less than 30, more preferably equal to or greater than 3 but equal to or less than 30, and even more preferably equal to or greater than 5 but equal to or less than 28.
- Examples of the carboxylic acid include aliphatic carboxylic acids (fatty acids) and aromatic carboxylic acids. Fatty acids and salts thereof are preferred.
- the rubber composition may include a saturated fatty acid or a salt thereof, or may include an unsaturated fatty acid or a salt thereof.
- the saturated fatty acid and the salt thereof are preferred.
- preferable carboxylates include a potassium salt, a magnesium salt, an aluminum salt, a zinc salt, an iron salt, a copper salt, a nickel salt, or a cobalt salt of octanoic acid, lauric acid, myristic acid, palmitic acid, stearic acid, oleic acid, or behenic acid.
- Zinc salts of carboxylic acids are particularly preferred.
- Specific examples of preferable carboxylates include zinc octoate, zinc laurate, zinc myristate, and zinc stearate.
- the amount of the acid and/or the salt (d) is preferably equal to or greater than 0.5 parts by weight, more preferably equal to or greater than 1.0 parts by weight, even more preferably equal to or greater than 1.5 parts by weight, and particularly preferably equal to or greater than 2.0 parts by weight, per 100 parts by weight of the base rubber.
- the amount is preferably equal to or less than 40 parts by weight, more preferably less than 40 parts by weight, even more preferably equal to or less than 30 parts by weight, and particularly preferably equal to or less than 20 parts by weight, per 100 parts by weight of the base rubber.
- the weight ratio of the co-crosslinking agent (b) and the acid and/or the salt (d) in the rubber composition is preferably equal to or greater than 3/7 but equal to or less than 9/1, and is particularly preferably equal to or greater than 4/6 but equal to or less than 8/2. From the rubber composition in which this weight ratio is within the above range, the core 104 whose hardness linearly increases from its central point toward its surface can be obtained.
- zinc acrylate is preferably used as the co-crosslinking agent (b).
- Zinc acrylate whose surface is coated with stearic acid or zinc stearate for the purpose of improving dispersibility to rubber is present.
- this coating material is not included in the concept of the acid and/or the salt (d).
- the rubber composition preferably further includes an organic sulfur compound (e).
- the organic sulfur compound (e) can contribute to control of: the linearity of the hardness distribution of the core 104; and the degree of the outer-hard/inner-soft structure.
- An example of the organic sulfur compound (e) is an organic compound having a thiol group or a polysulfide linkage having 2 to 4 sulfur atoms.
- a metal salt of this organic compound is also included in the organic sulfur compound (e).
- the rubber composition of the core 104 can include, as the organic sulfur compound (e), the organic sulfur compound (e) described above for the rubber composition of the core 4 according to the first embodiment.
- the amount of the organic sulfur compound (e) is preferably equal to or greater than 0.05 parts by weight and particularly preferably equal to or greater than 0.1 parts by weight, per 100 parts by weight of the base rubber. In light of resilience performance, the amount is preferably equal to or less than 5.0 parts by weight and particularly preferably equal to or less than 2.0 parts by weight, per 100 parts by weight of the base rubber.
- a filler may be included in the core 104.
- suitable fillers include zinc oxide, barium sulfate, calcium carbonate, and magnesium carbonate.
- the amount of the filler is determined as appropriate so that the intended specific gravity of the core 104 is accomplished.
- a particularly preferable filler is zinc oxide. Zinc oxide serves not only as a specific gravity adjuster but also as a crosslinking activator.
- an anti-aging agent a coloring agent, a plasticizer, a dispersant, sulfur, a vulcanization accelerator, and the like are added to the rubber composition of the core 104.
- Crosslinked rubber powder or synthetic resin powder may also be dispersed in the rubber composition.
- the difference (Hs-H(0)) between the surface hardness Hs and the central hardness H (0) is preferably equal to or greater than 15.
- the difference is great.
- the core 104 has an outer-hard/inner-soft structure.
- the recoil (torsional return) is great, and thus spin is suppressed.
- the core 104 contributes to the flight performance of the golf ball 102.
- the difference (Hs-H(0)) is more preferably equal to or greater than 20 and particularly preferably equal to or greater than 25. From the standpoint that the core 104 can easily be formed, the difference (Hs-H(0)) is preferably equal to or less than 50.
- the hardness H(0) at the central point of the core 104 is preferably equal to or greater than 40.0 but equal to or less than 70.0.
- the golf ball 102 having a hardness H(0) of 40.0 or greater has excellent resilience performance.
- the hardness H (0) is more preferably equal to or greater than 45.0 and particularly preferably equal to or greater than 50.0.
- the core 104 having a hardness H(0) of 70.0 or less can achieve an outer-hard/inner-soft structure. In the golf ball 102 that includes the core 104, spin can be suppressed.
- the hardness H(0) is more preferably equal to or less than 68.0 and particularly preferably equal to or less than 66.0.
- the hardness Hs at the surface of the core 104 is preferably equal to or greater than 78.0 but equal to or less than 95.0.
- the core 104 having a hardness Hs of 78.0 or greater an outer-hard/inner-soft structure can be achieved.
- the golf ball 102 that includes the core 104 spin can be suppressed.
- the hardness Hs is more preferably equal to or greater than 80.0 and particularly preferably equal to or greater than 82.0.
- the golf ball 102 having a hardness Hs of 95.0 or less has excellent durability.
- the hardness Hs is more preferably equal to or less than 93.0 and particularly preferably equal to or less than 90.0.
- the core 104 preferably has a diameter of 38.0 mm or greater but 42.0 mm or less.
- the core 104 having a diameter of 38.0 mm or greater can achieve excellent resilience performance of the golf ball 102.
- the diameter is more preferably equal to or greater than 38.5 mm and particularly preferably equal to or greater than 39.0 mm.
- the inner cover 108 and the outer cover 110 can have sufficient thicknesses.
- the golf ball 102 that includes the inner cover 108 and the outer cover 110 which have large thicknesses has excellent durability.
- the diameter is more preferably equal to or less than 41.0 mm and particularly preferably equal to or less than 40.0 mm.
- the core 104 has an amount of compressive deformation Dc of preferably 3.0 mm or greater and particularly preferably 3.3 mm or greater.
- the amount of compressive deformation Dc is preferably equal to or less than 4.6 mm and particularly preferably equal to or less than 4.3 mm.
- a resin composition is suitably used.
- the base polymer of the resin composition include ionomer resins, styrene block-containing thermoplastic elastomers, thermoplastic polyester elastomers, thermoplastic polyamide elastomers, and thermoplastic polyolefin elastomers.
- Particularly preferable base polymers are ionomer resins.
- the golf ball 102 that includes the inner cover 108 including an ionomer resin has excellent resilience performance.
- An ionomer resin and another resin may be used in combination for the inner cover 108.
- the principal component of the base polymer is preferably the ionomer resin.
- the proportion of the ionomer resin to the entire base polymer is preferably equal to or greater than 50% by weight, more preferably equal to or greater than 60% by weight, and particularly preferably equal to or greater than 70% by weight.
- preferable ionomer resins include binary copolymers formed with an ⁇ -olefin and an ⁇ , ⁇ -unsaturated carboxylic acid having 3 to 8 carbon atoms.
- a preferable binary copolymer includes 80% by weight or greater but 90% by weight or less of an ⁇ -olefin, and 10% by weight or greater but 20% by weight or less of an ⁇ , ⁇ -unsaturated carboxylic acid.
- the binary copolymer has excellent resilience performance.
- Examples of other preferable ionomer resins include ternary copolymers formed with: an ⁇ -olefin; an ⁇ , ⁇ -unsaturated carboxylic acid having 3 to 8 carbon atoms; and an ⁇ , ⁇ -unsaturated carboxylate ester having 2 to 22 carbon atoms.
- Apreferable ternary copolymer includes 70% by weight or greater but 85% by weight or less of an ⁇ -olefin, 5% by weight or greater but 30% by weight or less of an ⁇ , ⁇ -unsaturated carboxylic acid, and 1% by weight or greater but 25% by weight or less of an ⁇ , ⁇ -unsaturated carboxylate ester.
- the ternary copolymer has excellent resilience performance.
- ⁇ -olefins are ethylene and propylene, while preferable ⁇ , ⁇ -unsaturated carboxylic acids are acrylic acid and methacrylic acid.
- a particularly preferable ionomer resin is a copolymer formed with ethylene and acrylic acid or methacrylic acid.
- some of the carboxyl groups are neutralized with metal ions.
- metal ions for use in neutralization include sodium ion, potassium ion, lithium ion, zinc ion, calcium ion, magnesium ion, aluminum ion, and neodymium ion.
- the neutralization may be carried out with two or more types of metal ions.
- Particularly suitable metal ions in light of resilience performance and durability of the golf ball 2 are sodium ion, zinc ion, lithium ion, and magnesium ion.
- ionomer resins include trade names "Himilan 1555”, “Himilan 1557”, “Himilan 1605", “Himilan 1706", “Himilan 1707”, “Himilan 1856", “Himilan 1855”, “Himilan AM7311”, “Himilan AM7315”, “Himilan AM7317”,”Himilan AM7318”, “Himilan AM7329”, “Himilan AM7337”, “Himilan MK7320”, and “Himilan MK7329”, manufactured by Du Pont-MITSUI POLYCHEMICALS Co., Ltd.; trade names “Surlyn 6120”, “Surlyn 6910", “Surlyn 7930”, “Surlyn 7940", “Surlyn 8140”, “Surlyn 8150”, “Surlyn 8940", “Surlyn 8945”, “Surlyn 9120”, “Surlyn 9150”, “Surlyn 9910", “Surlyn 9945”, "Surlyn
- Two or more ionomer resins may be used in combination for the inner cover 108.
- An ionomer resin neutralized with a monovalent metal ion, and an ionomer resin neutralized with a bivalent metal ion may be used in combination.
- a preferable resin that can be used in combination with an ionomer resin is a styrene block-containing thermoplastic elastomer.
- the styrene block-containing thermoplastic elastomer has excellent compatibility with ionomer resins.
- a resin composition including the styrene block-containing thermoplastic elastomer has excellent fluidity.
- the styrene block-containing thermoplastic elastomer includes a polystyrene block as a hard segment, and a soft segment.
- a typical soft segment is a diene block.
- Examples of compounds for the diene block include butadiene, isoprene, 1,3-pentadiene, and 2,3-dimethyl-1,3-butadiene. Butadiene and isoprene are preferred. Two or more compounds may be used in combination.
- styrene block-containing thermoplastic elastomers examples include styrene-butadiene-styrene block copolymers (SBS), styrene-isoprene-styrene block copolymers (SIS), styrene-isoprene-butadiene-styrene block copolymers (SIBS), hydrogenated SBS, hydrogenated SIS, and hydrogenated SIBS.
- hydrogenated SBS include styrene-ethylene-butylene-styrene block copolymers (SEBS).
- hydrogenated SIS examples include styrene-ethylene-propylene-styrene block copolymers (SEPS).
- SIBS styrene-ethylene-ethylene-propylene-styrene block copolymers
- the content of the styrene component in the styrene block-containing thermoplastic elastomer is preferably equal to or greater than 10% by weight, more preferably equal to or greater than 12% by weight, and particularly preferably equal to or greater than 15% by weight.
- the content is preferably equal to or less than 50% by weight, more preferably equal to or less than 47% by weight, and particularly preferably equal to or less than 45% by weight.
- styrene block-containing thermoplastic elastomers include an alloy of an olefin and one or more members selected from the group consisting of SBS, SIS, SIBS, SEBS, SEPS, SEEPS, and hydrogenated products thereof.
- the olefin component in the alloy is presumed to contribute to improvement of compatibility with ionomer resins. Use of this alloy improves the resilience performance of the golf ball 102.
- An olefin having 2 to 10 carbon atoms is preferably used. Examples of suitable olefins include ethylene, propylene, butene, and pentene. Ethylene and propylene are particularly preferred.
- polymer alloys include trade names “Rabalon T3221C”, “Rabalon T3339C”, “Rabalon SJ4400N”, “Rabalon SJ5400N”, “Rabalon SJ6400N”, “Rabalon SJ7400N”, “Rabalon SJ8400N”, “Rabalon SJ9400N”, and “Rabalon SR04", manufactured by Mitsubishi Chemical Corporation.
- Other specific examples of styrene block-containing thermoplastic elastomers include trade name “Epofriend A1010” manufactured by Daicel Chemical Industries, Ltd., and trade name “Septon HG-252" manufactured by Kuraray Co., Ltd.
- a coloring agent such as titanium dioxide and a fluorescent pigment
- a filler such as barium sulfate, a dispersant, an antioxidant, an ultraviolet absorber, a light stabilizer, a fluorescent material, a fluorescent brightener, and the like are included in the resin composition of the inner cover 108 in an adequate amount.
- the inner cover 108 may include powder of a metal with a high specific gravity.
- the inner cover 108 has a hardness Hi which is equal to or less than the JIS-C hardness Hs at the surface of the core 104.
- the inner cover 108 achieves soft feel at impact.
- the difference (Hi-Hs) is preferably equal to or greater than 1, more preferably equal to or greater than 2, and particularly preferably equal to or greater than 7.
- the difference (Hi-Hs) is preferably equal to or less than 20, more preferably equal to or less than 18, and particularly preferably equal to or less than 12.
- the hardness Hi of the inner cover 108 is preferably equal to or less than 90, more preferably equal to or less than 85, and particularly preferably equal to or less than 83. In light of light feel at impact, the hardness Hi is preferably equal to or greater than 60, more preferably equal to or greater than 65, and particularly preferably equal to or greater than 71.
- the hardness Hi is measured with a JIS-C type hardness scale mounted to an automated rubber hardness measurement machine (trade name "P1", manufactured by Kobunshi Keiki Co., Ltd.).
- a slab that is formed by hot press and that has a thickness of about 2 mm is used.
- a slab kept at 23°C for two weeks is used for the measurement.
- three slabs are stacked.
- a slab formed fromthe same resin composition as the resin composition of the inner cover 108 is used.
- the inner cover 108 preferably has a thickness of 0.2 mm or greater but 2.0 mm or less.
- the golf ball 102 that includes the inner cover 108 having a thickness of 0.2 mm or greater has excellent feel at impact.
- the thickness of the inner cover 108 is more preferably equal to or greater than 0.5 mm and particularly preferably equal to or greater than 0. 8 mm.
- the golf ball 102 that includes the inner cover 108 having a thickness of 2.0 mm or less has excellent resilience performance.
- the thickness is more preferably equal to or less than 1.5 mm and particularly preferably equal to or less than 1.2 mm.
- a sphere 116 consisting of the core 104 and the inner cover 108 has an amount of compressive deformation Di of preferably 3.2 mm or greater and particularly preferably 3.4 mm or greater.
- the amount of compressive deformation Di is preferably equal to or less than 3.8 mm and particularly preferably equal to or less than 3.6 mm.
- the inner cover 108 For forming the inner cover 108, known methods such as injection molding, compression molding, and the like can be used.
- a resin composition is suitably used for the outer cover 110.
- a preferable base polymer of the resin composition is an ionomer resin.
- the golf ball 102 that includes the outer cover 110 including the ionomer resin has excellent resilience performance.
- the ionomer resin described above for the inner cover 108 can be used for the outer cover 110.
- an ionomer resin and another resin may be used in combination.
- the ionomer resin is included as the principal component of the base polymer.
- the proportion of the ionomer resin to the entire base polymer is preferably equal to or greater than 50% by weight, more preferably equal to or greater than 60% by weight, and particularly preferably equal to or greater than 70% by weight.
- a preferable resin that can be used in combination with an ionomer resin is an ethylene-(meth)acrylic acid copolymer.
- the copolymer is obtained by a copolymerization reaction of a monomer composition that contains ethylene and (meth)acrylic acid. In the copolymer, some of the carboxyl groups are neutralized with metal ions.
- the copolymer includes 3% by weight or greater but 25% by weight or less of a (meth)acrylic acid component.
- An ethylene-(meth)acrylic acid copolymer having a polar functional group is particularly preferred.
- a specific example of ethylene- (meth) acrylic acid copolymers is trade name "NUCREL" manufactured by Du Pont-MITSUI POLYCHEMICALS Co., Ltd.
- a coloring agent such as titanium dioxide and a fluorescent pigment
- a filler such as barium sulfate, a dispersant, an antioxidant, an ultraviolet absorber, a light stabilizer, a fluorescent material, a fluorescent brightener, and the like are included in the outer cover 110 in an adequate amount.
- the outer cover 110 preferably has a JIS-C hardness Ho of 83 or greater but 96 or less.
- a hardness Ho of 83 or greater an outer-hard/inner-soft structure can be achieved.
- spin is suppressed.
- the golf ball 102 has excellent flight performance.
- the hardness Ho is more preferably equal to or greater than 84 and particularly preferably equal to or greater than 85.
- the golf ball 102 that includes the outer cover 110 having a hardness Ho of 96 or less has excellent feel at impact.
- the hardness Ho is more preferably equal to or less than 95 and particularly preferably equal to or less than 93.
- the hardness Ho is measured by the same measurement method as that for the hardness Hi.
- the outer cover 110 preferably has a thickness of 0.2 mm or greater but 1.5 mm or less.
- the outer cover 110 having a thickness of 0.2 mm or greater can easily be formed.
- the thickness is more preferably equal to or greater than 0.4 mm and particularly preferably equal to or greater than 0.6 mm.
- the golf ball 102 that includes the outer cover 110 having a thickness of 1. 5 mm or less has excellent feel at impact.
- the thickness is more preferably equal to or less than 1.3 mm and particularly preferably equal to or less than 1.1 mm.
- the outer cover 110 For forming the outer cover 110 , known methods such as injection molding, compression molding, and the like can be used. When forming the outer cover 110, the dimples 112 are formed by pimples formed on the cavity face of a mold.
- the cover 106 preferably has a total thickness of 2.5 mm or less.
- the golf ball 102 that includes the cover 106 having a total thickness of 2.5 mm or less has excellent feel at impact.
- the total thickness is more preferably equal to or less than 2.3 mm and particularly preferably equal to or less than 2.1 mm.
- the total thickness is preferably equal to or greater than 0.3 mm, more preferably equal to or greater than 0.5 mm, and particularly preferably equal to or greater than 0.8 mm.
- the JIS-C hardness Ho of the outer cover 110 is greater than the JIS-C hardness Hi of the inner cover 108.
- the outer cover 110 can achieve an outer-hard/inner-soft structure of the golf ball 102 .
- the golf ball 102 has excellent flight performance and excellent feel at impact.
- the difference (Ho-Hi) is preferably equal to or greater than 5, more preferably equal to or greater than 9, and particularly preferably equal to or greater than 16. In light of suppression of energy loss when the golf ball 102 is hit, the difference (Ho-Hi) is preferably equal to or less than 27 and particularly preferably equal to or less than 21.
- the hardness of the outer cover 110 is the greatest. In the golf ball 102, spin is suppressed.
- the golf ball 102 has an amount of compressive deformation Db of preferably 2.8 mm or greater, more preferably 2.9 mm or greater, and particularly preferably 3.0 mm or greater.
- the amount of compressive deformation Db is preferably equal to or less than 3.6 mm, more preferably equal to or less than 3.5 mm, and particularly preferably equal to or less than 3.4 mm.
- a sphere such as the core 104, the golf ball 102, or the like is placed on a hard plate made of metal.
- a cylinder made of metal gradually descends toward the sphere.
- the sphere squeezed between the bottom face of the cylinder and the hard plate, becomes deformed.
- a migration distance of the cylinder starting from the state in which an initial load of 98 N is applied to the sphere up to the state in which a final load of 1274 N is applied thereto, is measured.
- a rubber composition was obtained by kneading 100 parts by weight of a high-cis polybutadiene (trade name "BR-730", manufactured by JSR Corporation), 26 parts by weight of zinc diacrylate (trade name "Sanceler SR”, manufactured by SANSHIN CHEMICAL INDUSTRY CO., LTD.), 5 parts by weight of zinc oxide, an appropriate amount of barium sulfate, 0.2 parts by weight of 2-thionaphthol, 10 parts by weight of zinc stearate, and 0.75 parts by weight of dicumyl peroxide.
- BR-730 high-cis polybutadiene
- zinc diacrylate trade name "Sanceler SR” manufactured by SANSHIN CHEMICAL INDUSTRY CO., LTD.
- This rubber composition was placed into a mold including upper and lower mold halves each having a hemispherical cavity, and heated at 170°C for 25 minutes to obtain a core with a diameter of 39.1 mm.
- the amount of barium sulfate was adjusted such that the weight of a golf ball is 45.4 g.
- a resin composition was obtained by kneading 40 parts by weight of an ionomer resin (the aforementioned "Himilan AM7337”), 40 parts by weight of another ionomer resin (the aforementioned "Himilan AM7329”), 20 parts by weight of a styrene block-containing thermoplastic elastomer (the aforementioned "Rabalon T3221C”), and 6 parts by weight of titanium dioxide with a twin-screw kneading extruder.
- the core was placed into a mold.
- the resin composition was injected around the core by injection molding to form an inner cover with a thickness of 1.0 mm.
- a resin composition was obtained by kneading 5 parts by weight of an ionomer resin (the aforementioned "Himilan AM7337”), 10 parts by weight of another ionomer resin (the aforementioned "Himilan 1555"), 55 parts by weight of still another ionomer resin (the aforementioned "Himilan AM7329”), 30 parts by weight of an ethylene- (meth) acrylic acid copolymer (trade name "NUCREL N1050H", manufactured by Du Pont-MITSUI POLYCHEMICALS Co., Ltd.), 3 parts by weight of titanium dioxide, and 0.2 parts by weight of an ultraviolet absorber (trade name "TINUVIN 770", manufactured by Ciba Japan K.K.) with a twin-screw kneading extruder.
- an ionomer resin the aforementioned "Himilan AM7337
- 10 parts by weight of another ionomer resin the aforementioned "Himilan 1555"
- the sphere consisting of the core and the inner cover was placed into a final mold having a large number of pimples on its cavity face.
- the resin composition was injected around the sphere by injection molding to form an outer cover with a thickness of 0.8 mm. Dimples having a shape that is the inverted shape of the pimples were formed on the outer cover.
- a clear paint including a two-component curing type polyurethane as a base material was applied to the outer cover to obtain a golf ball of Example I-1 with a diameter of 42.7 mm.
- Example I-2 to I-14 and Comparative Examples I-1 to I-5 were obtained in the same manner as Example I-1, except the specifications of the core, the inner cover, and the outer cover were as shown in Tables I-7 to I-9 below.
- the composition of the core is shown in detail in Tables I-1 and I-2 below.
- the compositions of the inner cover and the outer cover are shown in detail in Table I-3 below.
- a hardness distribution of the core is shown in Tables I-4 to I-6 below.
- the golf ball according to Comparative Example I-3 does not have an inner cover.
- a 5-iron (trade name "XXIO”, manufactured by SRI Sports Limited, shaft hardness: R, loft angle: 24°) was attached to a swing machine manufactured by Golf Laboratories, Inc.
- a golf ball was hit under the condition of a head speed of 35 m/sec.
- the spin rate was measured immediately after the hit.
- the distance from the launch point to the stop point was measured.
- the average value of data obtained by 12 measurements is shown in Tables I-7 to I-9 below.
- Table I-1 Composition of Core (parts by weight) A C D E F M BR-730 100 100 100 100 100 100 100 100 Sanceler SR 27.0 26.0 27.5 29.5 31.5 25.0 Zinc oxide 5 5 5 5 5 5 Barium sulfate Appropriate amount 2-thionaphthol 0.2 0.2 0.2 0.2 0.2 0.2 Zinc stearate 0 10 20 30 40 - Aluminum stearate - - - - - 10 Dicumyl peroxide 0.75 0.75 0.75 0.75 0.75 0.75 0.75 0.75 0.75 0.75 0.75 Amount of compressive deformation Dc (mm) 3.86 3.85 3.86 3.85 3.86 3.83 Table I-2 Composition of Core (parts by weight) G H I J K L BR-730 100 100 100 100 100 100 100 100 100 100 100 100 Sanceler SR 26.5 25.5 25.0 25.5 26.0 25.5 Zinc oxide 5 5 5 5 5 5 Barium sulfate Appropriate amount 2-thionaphthol
- the golf balls according to Examples have excellent flight performance upon a shot with a middle iron. From the results of evaluation, advantages of the present invention are clear.
- a rubber composition was obtained by kneading 100 parts by weight of a high-cis polybutadiene (trade name "BR-730", manufactured by JSR Corporation), 26 parts by weight of zinc diacrylate (trade name "Sanceler SR”, manufactured by SANSHIN CHEMICAL INDUSTRY CO., LTD.), 5 parts by weight of zinc oxide, an appropriate amount of barium sulfate, 0.2 parts by weight of 2- thionaphthol, 10 parts by weight of zinc stearate, and 0.75 parts by weight of dicumyl peroxide.
- BR-730 high-cis polybutadiene
- zinc diacrylate trade name "Sanceler SR” manufactured by SANSHIN CHEMICAL INDUSTRY CO., LTD.
- This rubber composition was placed into a mold including upper and lower mold halves each having a hemispherical cavity, and heated at 170°C for 25 minutes to obtain a core with a diameter of 39.1 mm.
- the amount of barium sulfate was adjusted such that the weight of a golf ball is 45.4 g.
- a resin composition was obtained by kneading 24 parts by weight of an ionomer resin (the aforementioned "Himilan AM7337”), 50 parts by weight of another ionomer resin (the aforementioned "Himilan AM7329”), 26 parts by weight of a styrene block-containing thermoplastic elastomer (the aforementioned "Rabalon T3221C”), and 6 parts by weight of titanium dioxide with a twin-screw kneading extruder.
- the core was placed into a mold.
- the resin composition was injected around the core by injection molding to form an inner cover with a thickness of 1.0 mm.
- a resin composition was obtained by kneading 5 parts by weight of an ionomer resin (the aforementioned "Himilan AM7337”), 10 parts by weight of another ionomer resin (the aforementioned "Himilan 1555"), 55 parts by weight of still another ionomer resin (the aforementioned "Himilan AM7329”), 30 parts by weight of an ethylene- (meth) acrylic acid copolymer (trade name "NUCREL N1050H", manufactured by Du Pont-MITSUI POLYCHEMICALS Co., Ltd.), 3 parts by weight of titanium dioxide, and 0.2 parts by weight of an ultraviolet absorber (trade name "TINUVIN 770", manufactured by Ciba Japan K.K.) with a twin-screw kneading extruder.
- an ionomer resin the aforementioned "Himilan AM7337
- 10 parts by weight of another ionomer resin the aforementioned "Himilan 1555"
- the sphere consisting of the core and the inner cover was placed into a final mold having a large number of pimples on its cavity face.
- the resin composition was injected around the sphere by injection molding to form an outer cover with a thickness of 0.8 mm. Dimples having a shape that is the inverted shape of the pimples were formed on the outer cover.
- a clear paint including a two-component curing type polyurethane as a base material was applied to the outer cover to obtain a golf ball of Example II-1 with a diameter of 42.7 mm.
- Examples II-2 to II-14 and Comparative Examples II-1 to II-5 were obtained in the same manner as Example II-1, except the specifications of the core, the inner cover, and the outer cover were as shown in Tables II-7 to II-9 below.
- the composition of the core is shown in detail in Tables II-1 and II-2 below.
- the compositions of the inner cover and the outer cover are shown in detail in Table II-3 below.
- a hardness distribution of the core is shown in Tables II-4 to II-6 below.
- the golf ball according to Comparative Example II-2 does not have an inner cover.
- a driver (trade name "XXIO”, manufactured by SRI Sports Limited, shaft hardness: R, loft angle: 10.5°) was attached to a swing machine manufactured by Golf Laboratories, Inc. A golf ball was hit under the condition of a head speed of 40 m/sec. The spin rate was measured immediately after the hit. Furthermore, the distance from the launch point to the stop point was measured. The average value of data obtained by 12 measurements is shown in Tables II-7 to II-9 below.
- the golf balls according to Examples have excellent flight performance and excellent feel at impact upon a shot with a driver. From the results of evaluation, advantages of the present invention are clear.
- the golf ball according to the present invention can be used for playing golf on golf courses and practicing at driving ranges.
- the above descriptions are merely for illustrative examples, and various modifications can be made without departing from the principles of the present invention.
Landscapes
- Health & Medical Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Physical Education & Sports Medicine (AREA)
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Compositions Of Macromolecular Compounds (AREA)
Description
- The present invention relates to golf balls. Specifically, the present invention relates to golf balls that include a solid core and a cover including two or more layers.
- Golf players' foremost requirement for golf balls is flight performance. In particular, golf players place importance on flight performance upon shots with a driver and a long iron. Flight performance correlates with the resilience performance of a golf ball. When a golf ball having excellent resilience performance is hit, the golf ball flies at a high speed, thereby achieving a large flight distance. Golf balls that include a core having excellent resilience performance are disclosed in
JP61-37178 JP2008-212681 US2008/0214324 ),JP2008-523952 US2006/0135287 andUS2007/0173607 ), andJP2009-119256 US2009/0124757 ). - The core disclosed in
JP61-37178 - The core disclosed in
JP2008-212681 - The core disclosed in
JP2008-523952 - The core disclosed in
JP2009-119256 - An appropriate trajectory height is required in order to achieve a large flight distance. A trajectory height depends on a spin rate and a launch angle. With a golf ball that achieves a high trajectory by a high spin rate, a flight distance is insufficient. With a golf ball that achieves a high trajectory by a high launch angle, a large flight distance is obtained. Use of an outer-hard/inner-soft structure in a golf ball can achieve a low spin rate and a high launch angle. Modifications regarding a hardness distribution of a core are disclosed in
JP6-154357 5,403,010 ),JP2008-194471 7,344,455 ,US2008/0194358 ,US2008/0194359 , andUS2008/0214325 ), andJP2008-194473 US2008/0194357 andUS2008/0312008 ). - In the core disclosed in
JP6-154357 - In the core disclosed in
JP2008-194471 - In the core disclosed in
JP2008-194473 -
JP2010-253268 US2010/0273575 ) discloses a golf ball that includes a core, an envelope layer, a mid layer, and a cover. In the core, the hardness gradually increases from the central point of the core to the surface of the core. The difference between a JIS-C hardness at the surface and a JIS-C hardness at the central point is equal to or greater than 15. The hardness of the cover is greater than the hardness of the mid layer, and the hardness of the mid layer is greater than the hardness of the envelope layer. -
US 2011/306443 A1 discloses a golf ball comprising a core, a mid layer positioned outside the core and a cover positioned outside the mid layer, wherein the core comprises a center and an envelope layer. The core of the golf ball may be composed of a rubber composition including a base resin, a crosslinking initiator, a co-crosslinking agent, zinc oxide and barium sulfate. -
US 2003/109332 A1 relates to a golf ball having a core, an inner cover and an outer cover, wherein the JIS-C hardness of the inner cover is higher than the JIS-C hardness at the surface of the core by less than 15. The core of the golf ball may be composed of a rubber composition including a base resin, a crosslinking initiator, a co-crosslinking agent, zinc oxide and barium sulfate. - For a tee shot on a par-three hole and a second shot on a par-four hole, a middle iron is frequently used. Golf players also desire a large flight distance upon a shot with a middle iron. An object of the present invention is to provide a golf ball that exerts excellent flight performance upon a shot with a middle iron.
- Golf players' requirements for flight distance have been escalated more than ever. Golf players further desire golf balls having excellent feel at impact. Golf balls that satisfy in terms of flight distance tend to have inferior feel at impact.
- Another object of the present invention is to provide a golf ball that achieves a large flight distance and excellent feel at impact upon a shot with a driver.
- The present invention relates to a golf ball, which consists of a spherical core and a cover covering the core and including two or more layers, wherein
when distances (%) from a central point of the core to nine points and JIS-C hardnesses at the nine points, which nine points are obtained by dividing a region from the central point of the core to a surface of the core at intervals of 12.5% of a radius of the core, are plotted in a graph, R2 of a linear approximation curve obtained by a least-square method is equal to or greater than 0.95, wherein R2 is calculated by squaring a correlation coefficient R, wherein the correlation coefficient R is calculated by dividing the covariance of the distance (%) from the central point and the hardness (JIS-C) by the standard deviation of the distance (%) from the central point and the standard deviation of the hardness (JIS-C),
the JIS-C hardness Hi of the innermost layer of the cover is greater than the JIS-C hardness Hs at the surface of the core or the JIS-C hardness Hi of the innermost layer of the cover is equal to or less than the JIS-C hardness Hs at the surface of the core, and
the core is formed by a rubber composition being crosslinked, wherein the rubber composition includes: - (a) a base rubber,
- (b) a co-crosslinking agent,
- (c) a crosslinking initiator and
- (d1) a carboxylic acid and/or a salt thereof, wherein the co-crosslinking agent (b) is:
- (b1) an α,β-unsaturated carboxylic acid having 3 to 8 carbon atoms; or
- (b2) a metal salt of an α,β-unsaturated carboxylic acid having 3 to 8 carbon atoms, wherein the carboxylic acid and/or salt thereof (d1) is different from the co-crosslinking agent (b).
- According to one embodiment, the JIS-C hardness Hi of an innermost layer of the cover is greater than a JIS-C hardness Hs at the surface of the core. In this embodiment, the hardness distribution of the golf ball is appropriate. In the golf ball of this embodiment, the energy loss is low when the golf ball is hit with a middle iron. When the golf ball is hit with a middle iron, the spin rate is low. The low spin rate achieves a large flight distance.
- According to another embodiment, the JIS-C hardness Hi of the innermost layer of the cover is equal to or less than the JIS-C hardness Hs at the surface of the core. In the golf ball according to this embodiment, the hardness distribution is appropriate. In the golf ball of this embodiment, the energy loss is low when the golf ball is hit with a driver. When the golf ball is hit with a driver, the spin rate is low. The low spin rate achieves a large flight distance. In the golf ball of this embodiment, the innermost layer achieves soft feel at impact. The golf ball of this embodiment has both excellent flight performance and excellent feel at impact.
-
-
FIG. 1 is a partially cutaway cross-sectional view of a golf ball according to a first embodiment of the present invention; -
FIG. 2 is a line graph showing a hardness distribution of a core of the golf ball inFIG. 1 ; -
FIG. 3 is a partially cutaway cross-sectional view of a golf ball according to a second embodiment of the present invention; and -
FIG. 4 is a line graph showing a hardness distribution of a core of the golf ball inFIG. 3 . - The following will describe in detail the present invention, based on preferred embodiments with reference to the accompanying drawings.
- A
golf ball 2 shown inFIG. 1 includes aspherical core 4 and acover 6 covering thecore 4. Thecover 6 includes an inner cover 8 and anouter cover 10 positioned outside the inner cover 8. The inner cover 8 is an innermost layer of thecover 6. Theouter cover 10 is an outermost layer of thecover 6. Thecover 6 may include another one or more layers between the inner cover 8 and theouter cover 10. On the surface of theouter cover 10, a large number ofdimples 12 are formed. Of the surface of thegolf ball 2, a part other than thedimples 12 is aland 14. Thegolf ball 2 includes a paint layer and a mark layer on the external side of theouter cover 10, but these layers are not shown in the drawing. - The
golf ball 2 has a diameter of 40 mm or greater but 45 mm or less. From the standpoint of conformity to the rules established by the United States Golf Association (USGA), the diameter is preferably equal to or greater than 42.67 mm. In light of suppression of air resistance, the diameter is preferably equal to or less than 44 mm and more preferably equal to or less than 42.80 mm. Thegolf ball 2 has a weight of 40 g or greater but 50 g or less. In light of attainment of great inertia, the weight is preferably equal to or greater than 44 g and more preferably equal to or greater than 45.00 g. From the standpoint of conformity to the rules established by the USGA, the weight is preferably equal to or less than 45.93 g. -
FIG. 2 is a line graph showing a hardness distribution of thecore 4 of thegolf ball 2 inFIG. 1 . The horizontal axis of the graph indicates the ratio (%) of a distance from the central point of thecore 4 to the radius of thecore 4. The vertical axis of the graph indicates a JIS-C hardness. Nine measuring points obtained by dividing a region from the central point of thecore 4 to the surface of thecore 4 at intervals of 12.5% of the radius of thecore 4 are plotted in the graph. The ratio of the distance from the central point of thecore 4 to each of these measuring points to the radius of thecore 4 is as follows. - First point: 0.0% (central point)
- Second point: 12.5%
- Third point: 25.0%
- Fourth point: 37.5%
- Fifth point: 50.0%
- Sixth point: 62.5%
- Seventh point: 75.0%
- Eighth point: 87.5%
- Ninth point: 100.0% (surface)
-
FIG. 2 also shows a linear approximation curve obtained by a least-square method on the basis of the distances and the hardnesses of the nine measuring points. As is clear fromFIG. 2 , the broken line does not greatly deviate from the linear approximation curve. In other words, the broken line has a shape close to the linear approximation curve. In thecore 4, the hardness linearly increases from its central point toward its surface. When thecore 4 is hit with a middle iron, the energy loss is low. Thecore 4 has excellent resilience performance. When thegolf ball 2 is hit with a middle iron, the flight distance is large. - In the
core 4, R2 of the linear approximation curve obtained by the least-square method is equal to or greater than 0.95. R2 is an index indicating the linearity of the broken line. For thecore 4 for which R2 is equal to or greater than 0.95, the shape of the broken line of the hardness distribution is close to a straight line. Thecore 4 for which R2 is equal to or greater than 0.95 has excellent resilience performance. R2 is more preferably equal to or greater than 0.96 and particularly preferably equal to or greater than 0.97. R2 is calculated by squaring a correlation coefficient R. The correlation coefficient R is calculated by dividing the covariance of the distance (%) from the central point and the hardness (JIS-C) by the standard deviation of the distance (%) from the central point and the standard deviation of the hardness (JIS-C). - The
core 4 is obtained by crosslinking a rubber composition. The rubber composition includes: - (a) a base rubber;
- (b) a co-crosslinking agent;
- (c) a crosslinking initiator; and
- (d) an acid and/or a salt.
- During heating and forming of the
core 4, the base rubber (a) is crosslinked by the co-crosslinking agent (b). The heat of the crosslinking reaction remains near the central point of thecore 4. Thus, during heating and forming of thecore 4, the temperature at the central portion is high. The temperature gradually decreases from the central point toward the surface. It is inferred that in the rubber composition, the acid reacts with a metal salt of the co-crosslinking agent (b) to bond to cation. It is inferred that in the rubber composition, the salt reacts with the metal salt of the co-crosslinking agent (b) to exchange cation. By the bonding and the exchange, metallic bonding is broken. This breaking is likely to occur in the central portion of thecore 4 where the temperature is high, and is unlikely to occur near the surface of thecore 4. As a result, the crosslinking density of thecore 4 increases from its central point toward its surface. In thecore 4, an outer-hard/inner-soft structure can be achieved. Furthermore, when the rubber composition includes an organic sulfur compound (e) together with the acid and/or the salt (d), the gradient of the hardness distribution can be controlled, and the degree of the outer-hard/inner-soft structure of thecore 4 can be increased. When thegolf ball 2 that includes thecore 4 is hit with a middle iron, the spin rate is low. In thegolf ball 2, excellent flight performance is achieved upon a shot with a middle iron. - Examples of the base rubber (a) of the
core 4 include polybutadienes, polyisoprenes, styrene-butadiene copolymers, ethylene-propylene-diene copolymers, and natural rubbers. In light of resilience performance, polybutadienes are preferred. When a polybutadiene and another rubber are used in combination, it is preferred that the polybutadiene is included as a principal component. Specifically, the proportion of the polybutadiene to the entire base rubber is preferably equal to or greater than 50% by weight and more preferably equal to or greater than 80% by weight. The proportion of cis-1,4 bonds in the polybutadiene is preferably equal to or greater than 40% by weight and more preferably equal to or greater than 80% by weight. - A polybutadiene in which the proportion of 1,2-vinyl bonds is equal to or less than 2.0% by weight is preferred. The polybutadiene can contribute to the resilience performance of the
core 4. In this respect, the proportion of 1,2-vinyl bonds is preferably equal to or less than 1.7% by weight and particularly preferably equal to or less than 1.5% by weight. - From the standpoint that a polybutadiene having a low proportion of 1,2-vinyl bonds and excellent polymerization activity is obtained, a rare-earth-element-containing catalyst is preferably used for synthesis of a polybutadiene. In particular, a polybutadiene synthesized with a catalyst containing neodymium, which is a lanthanum-series rare earth element compound, is preferred.
- The polybutadiene has a Mooney viscosity (ML1+4(100°C)) of preferably 30 or greater, more preferably 32 or greater, and particularly preferably 35 or greater. The Mooney viscosity (ML1+4(100°C)) is preferably equal to or less than 140, more preferably equal to or less than 120, even more preferably equal to or less than 100, and particularly preferably equal to or less than 80. The Mooney viscosity (ML1+4 (100°C)) is measured according to the standards of "JIS K6300". The measurement conditions are as follows.
- Rotor: L rotor
- Preheating time: 1 minute
- Rotating time of rotor: 4 minutes
- Temperature: 100°C
- In light of workability, the polybutadiene has a molecular weight distribution (Mw/Mn) of preferably 2.0 or greater, more preferably 2.2 or greater, even more preferably 2.4 or greater, and particularly preferably 2.6 or greater. In light of resilience performance, the molecular weight distribution (Mw/Mn) is preferably equal to or less than 6.0, more preferably equal to or less than 5.0, even more preferably equal to or less than 4.0, and particularly preferably equal to or less than 3.4. The molecular weight distribution (Mw/Mn) is calculated by dividing the weight average molecular weight Mw by the number average molecular weight Mn.
- The molecular weight distribution is measured by gel permeation chromatography ("HLC-8120GPC" manufactured by Tosoh Corporation). The measurement conditions are as follows.
- Detector: differential refractometer
- Column: GMHHXL (manufactured by Tosoh Corporation)
- Column temperature: 40°C
- Mobile phase: tetrahydrofuran
- The co-crosslinking agent (b) is:
- (b1) an α,β-unsaturated carboxylic acid having 3 to 8 carbon atoms; or
- (b2) a metal salt of an α,β-unsaturated carboxylic acid having 3 to 8 carbon atoms.
- The rubber composition may include only the α,β-unsaturated carboxylic acid (b1) or only the metal salt (b2) of the α,β-unsaturated carboxylic acid as the co-crosslinking agent (b). The rubber composition may include both the α,β-unsaturated carboxylic acid (b1) and the metal salt (b2) of the α,β-unsaturated carboxylic acid as the co-crosslinking agent (b).
- The metal salt (b2) of the α,β-unsaturated carboxylic acid graft-polymerizes with the molecular chain of the base rubber, thereby crosslinking the rubber molecules. When the rubber composition includes the α,β-unsaturated carboxylic acid (b1), the rubber composition preferably further includes a metal compound (f). The metal compound (f) reacts with the α,β-unsaturated carboxylic acid (b1) in the rubber composition. A salt obtained by this reaction graft-polymerizes with the molecular chain of the base rubber.
- Examples of the metal compound (f) include metal hydroxides such as magnesium hydroxide, zinc hydroxide, calcium hydroxide, sodium hydroxide, lithium hydroxide, potassium hydroxide, and copper hydroxide; metal oxides such as magnesium oxide, calcium oxide, zinc oxide, and copper oxide; and metal carbonates such as magnesium carbonate, zinc carbonate, calcium carbonate, sodium carbonate, lithium carbonate, and potassium carbonate. A compound that includes a bivalent metal is preferred. The compound that includes the bivalent metal reacts with the co-crosslinking agent (b) to form metal crosslinks. The metal compound (f) is particularly preferably a zinc compound. Two or more metal compounds may be used in combination.
- Examples of the α,β-unsaturated carboxylic acids include acrylic acid, methacrylic acid, fumaric acid, maleic acid, and crotonic acid. Examples of the metal component in the metal salt (b2) of the α,β-unsaturated carboxylic acid include sodium ion, potassium ion, lithium ion, magnesium ion, calcium ion, zinc ion, barium ion, cadmium ion, aluminum ion, tin ion, and zirconium ion. The metal salt (b2) of the α,β-unsaturated carboxylic acid may include two or more types of ions. From the standpoint that metal crosslinks are likely to occur between the rubber molecules, bivalent metal ions such as magnesium ion, calcium ion, zinc ion, barium ion, and cadmium ion are preferred. The metal salt (b2) of the α,β-unsaturated carboxylic acid is particularly preferably zinc acrylate.
- In light of resilience performance of the
golf ball 2, the amount of the co-crosslinking agent (b) is preferably equal to or greater than 15 parts by weight and particularly preferably equal to or greater than 20 parts by weight, per 100 parts by weight of the base rubber. In light of feel at impact, the amount is preferably equal to or less than 50 parts by weight, more preferably equal to or less than 45 parts by weight, and particularly preferably equal to or less than 40 parts by weight, per 100 parts by weight of the base rubber. - The crosslinking initiator (c) is preferably an organic peroxide. The organic peroxide contributes to the resilience performance of the
golf ball 2. Examples of preferable organic peroxides include dicumyl peroxide, 1,1-bis(t-butylperoxy)-3,3,5-trimethylcyclohexane, 2,5-dimethyl-2,5-di(t-butylperoxy)hexane, and di-t-butyl peroxide. In light of versatility, dicumyl peroxide is preferred. - In light of resilience performance of the
golf ball 2, the amount of the crosslinking initiator (c) is preferably equal to or greater than 0.2 parts by weight and particularly preferably equal to or greater than 0.5 parts by weight, per 100 parts by weight of the base rubber. In light of feel at impact and durability of thegolf ball 2, the amount is preferably equal to or less than 5.0 parts by weight and particularly preferably equal to or less than 2.5 parts by weight, per 100 parts by weight of the base rubber. - In the present invention, the co-crosslinking agent (b) is not included in the concept of the acid and/or the salt (d) . It is inferred that as described above, the acid and/or the salt (d) breaks the metal crosslinks by the co-crosslinking agent (b) in the central portion of the
core 4 during heating and forming of thecore 4. According to the present invention, the acid and/or the salt (d) is a carboxylic acid and/or a salt thereof (d1). Carboxylates are particularly preferred. - The carboxylic acid component of the carboxylic acid and/or the salt thereof (d1) has a carboxyl group. The carbon number of the carboxylic acid component of the carboxylic acid and/or the salt thereof (d1) is preferably equal to or greater than 1 but equal to or less than 30, more preferably equal to or greater than 3 but equal to or less than 30, and even more preferably equal to or greater than 5 but equal to or less than 28. Examples of the carboxylic acid include aliphatic carboxylic acids (fatty acids) and aromatic carboxylic acids. Fatty acids and salts thereof are preferred.
- The rubber composition may include a saturated fatty acid or a salt thereof, or may include an unsaturated fatty acid or a salt thereof. The saturated fatty acid and the salt thereof are preferred.
- Examples of fatty acids include butyric acid (C4), valeric acid (C5), caproic acid (C6), enanthic acid (C7), caprylic acid (octanoic acid) (C8), pelargonic acid (C9), capric acid (C10), lauric acid (C12), myristic acid (C14), myristoleic acid (C14), pentadecylic acid (C15), palmitic acid (C16), palmitoleic acid (C16), margaric acid (C17), stearic acid (C18), elaidic acid (C18), vaccenic acid (C18), oleic acid (C18), linolic acid (C18), linolenic acid (C18), 12-hydroxystearic acid (C18), arachidic acid (C20), gadoleic acid (C20), arachidonic acid (C20), eicosenoic acid (C20), behenic acid (C22), erucic acid (C22), lignoceric acid (C24), nervonic acid (C24), cerotic acid (C26), montanic acid (C28), and melissic acid (C30). Two or more fatty acid salts may be used in combination. Octanoic acid, lauric acid, myristic acid, palmitic acid, stearic acid, oleic acid, and behenic acid are preferred.
- An aromatic carboxylic acid has an aromatic ring and a carboxyl group. Examples of aromatic carboxylic acids include benzoic acid, phthalic acid, isophthalic acid, terephthalic acid, hemimellitic acid (benzene-1,2,3-tricarboxylic acid), trimellitic acid (benzene-1,2,4-tricarboxylic acid), trimesic acid (benzene-1,3,5-tricarboxylic acid), mellophanic acid (benzene-1,2,3,4-tetracarboxylic acid), prehnitic acid (benzene-1,2,3,5-tetracarboxylic acid), pyromellitic acid (benzene-1,2,4,5-tetracarboxylic acid), mellitic acid (benzene hexacarboxylic acid), diphenic acid (biphenyl-2,2'-dicarboxylic acid), toluic acid (methylbenzoic acid), xylic acid, prehnitylic acid (2,3,4-trimethylbenzoic acid), γ-isodurylic acid (2,3,5-trimethylbenzoic acid), durylic acid (2,4,5-trimethylbenzoic acid), β-isodurylic acid (2,4,6-trimethylbenzoic acid), α-isodurylic acid (3,4,5-trimethylbenzoic acid), cuminic acid (4-isopropylbenzoic acid), uvitic acid (5-methylisophthalic acid), α-toluic acid (phenylacetic acid), hydratropic acid (2-phenylpropanoic acid), and hydrocinnamic acid (3-phenylpropanoic acid).
- The rubber composition may include a salt of an aromatic carboxylic acid substituted with a hydroxyl group, an alkoxy group, or an oxo group. Examples of this carboxylic acid can include salicylic acid (2-hydroxybenzoic acid), anisic acid (methoxybenzoic acid), cresotinic acid (hydroxy(methyl) benzoic acid), o-homosalicylic acid (2-hydroxy-3-methylbenzoic acid), m-homosalicylic acid (2-hydroxy-4-methylbenzoic acid), p-homosalicylic acid (2-hydroxy-5-methylbenzoic acid), o-pyrocatechuic acid (2,3-dihydroxybenzoic acid), β-resorcylic acid (2,4-dihydroxybenzoic acid), γ-resorcylic acid (2,6-dihydroxybenzoic acid), protocatechuic acid (3,4-dihydroxybenzoic acid), α-resorcylic acid (3,5-dihydroxybenzoic acid), vanillic acid (4-hydroxy-3-methoxybenzoic acid), isovanillic acid (3-hydroxy-4-methoxybenzoic acid), veratric acid (3,4-dimethoxybenzoic acid), o-veratric acid (2,3-dimethoxybenzoic acid), orsellinic acid (2,4-dihydroxy-6-methylbenzoic acid), m-hemipinic acid (4,5-dimethoxyphthalic acid), gallic acid (3,4,5-trihydroxybenzoic acid), syringic acid (4-hydroxy-3,5-dimethoxybenzoic acid), asaronic acid (2,4,5-trimethoxybenzoic acid), mandelic acid (hydroxy(phenyl)acetic acid), vanillylmandelic acid (hydroxy(4-hydroxy-3-methoxyphenyl)acetic acid), homoanisic acid ((4-methoxyphenyl)acetic acid), homogentisic acid ((2,5-dihydroxyphenyl)acetic acid), homoprotocatechuic acid ((3,4-dihydroxyphenyl)acetic acid), homovanillic acid ((4-hydroxy-3-methoxyphenyl)acetic acid), homoisovanillic acid ((3-hydroxy-4-methoxyphenyl)acetic acid), homoveratric acid ((3,4-dimethoxyphenyl)acetic acid), o-homoveratric acid ((2,3-dimethoxyphenyl)acetic acid), homophthalic acid (2-(carboxymethyl)benzoic acid), homoisophthalic acid (3-(carboxymethyl)benzoic acid), homoterephthalic acid (4-(carboxymethyl)benzoic acid), phthalonic acid (2-(carboxycarbonyl)benzoic acid), isophthalonic acid (3-(carboxycarbonyl)benzoic acid), terephthalonic acid (4-(carboxycarbonyl)benzoic acid), benzilic acid (hydroxydiphenylacetic acid), atrolactic acid (2-hydroxy-2-phenylpropanoic acid), tropic acid (3-hydroxy-2-phenylpropanoic acid), melilotic acid (3-(2-hydroxyphenyl)propanoic acid), phloretic acid (3-(4-hydroxyphenyl)propanoic acid), hydrocaffeic acid (3-(3,4-dihydroxyphenyl)propanoic acid), hydroferulic acid (3-(4-hydroxy-3-methoxyphenyl)propanoic acid), hydroisoferulic acid (3-(3-hydroxy-4-methoxyphenyl)propanoic acid), p-coumaric acid (3-(4-hydroxyphenyl)acrylic acid), umbellic acid (3-(2,4-dihydroxyphenyl)acrylic acid), caffeic acid (3-(3,4-dihydroxyphenyl)acrylic acid), ferulic acid (3-(4-hydroxy-3-methoxyphenyl)acrylic acid), isoferulic acid (3- (3-hydroxy-4-methoxyphenyl) acrylic acid), and sinapic acid (3-(4-hydroxy-3,5-dimethoxyphenyl)acrylic acid).
- The cationic component of the carboxylate is a metal ion or an organic cation. Examples of the metal ion include sodium ion, potassium ion, lithium ion, silver ion, magnesium ion, calcium ion, zinc ion, barium ion, cadmium ion, copper ion, cobalt ion, nickel ion, manganese ion, aluminum ion, iron ion, tin ion, zirconium ion, and titanium ion. Two or more types of ions may be used in combination.
- The organic cation is a cation having a carbon chain. Examples of the organic cation include organic ammonium ions. Examples of organic ammonium ions include primary ammonium ions such as stearylammonium ion, hexylammonium ion, octylammonium ion, and 2-ethylhexylammonium ion; secondary ammonium ions such as dodecyl (lauryl) ammonium ion, and octadecyl (stearyl) ammonium ion; tertiary ammonium ions such as trioctylammonium ion; and quaternary ammonium ions such as dioctyldimethylammonium ion, and distearyldimethylammonium ion. Two or more types of organic cations may be used in combination.
- Examples of preferable carboxylates include a potassium salt, a magnesium salt, an aluminum salt, a zinc salt, an iron salt, a copper salt, a nickel salt, or a cobalt salt of octanoic acid, lauric acid, myristic acid, palmitic acid, stearic acid, oleic acid, or behenic acid. Zinc salts of carboxylic acids are particularly preferred. Specific examples of preferable carboxylates include zinc octoate, zinc laurate, zinc myristate, and zinc stearate.
- In light of linearity of the hardness distribution of the
core 4, the amount of the carboxylic acid and/or the salt (d1) is preferably equal to or greater than 0.5 parts by weight, more preferably equal to or greater than 1.0 parts by weight, even more preferably equal to or greater than 1.5 parts by weight, and particularly preferably equal to or greater than 2.0 parts by weight, per 100 parts by weight of the base rubber. In light of resilience performance, the amount is preferably equal to or less than 40 parts by weight, more preferably less than 40 parts by weight, even more preferably equal to or less than 30 parts by weight, and particularly preferably equal to or less than 20 parts by weight, per 100 parts by weight of the base rubber. - The weight ratio of the co-crosslinking agent (b) and the carboxylic acid and/or the salt (d1) in the rubber composition is preferably equal to or greater than 3/7 but equal to or less than 9/1, and is particularly preferably equal to or greater than 4/6 but equal to or less than 8/2. From the rubber composition in which this weight ratio is within the above range, the
core 4 whose hardness linearly increases from its central point toward its surface can be obtained. - As the co-crosslinking agent (b), zinc acrylate is preferably used. Zinc acrylate whose surface is coated with stearic acid or zinc stearate for the purpose of improving dispersibility to rubber is present. In the present invention, when the rubber composition includes this zinc acrylate, this coating material is not included in the concept of the acid and/or the salt (d).
- The rubber composition preferably further includes an organic sulfur compound (e). The organic sulfur compound (e) can contribute to control of: the linearity of the hardness distribution of the
core 4; and the degree of the outer-hard/inner-soft structure. An example of the organic sulfur compound (e) is an organic compound having a thiol group or a polysulfide linkage having 2 to 4 sulfur atoms. A metal salt of this organic compound is also included in the organic sulfur compound (e). Examples of the organic sulfur compound (e) include aliphatic compounds such as aliphatic thiols, aliphatic thiocarboxylic acids, aliphatic dithiocarboxylic acids, and aliphatic polysulfides; heterocyclic compounds; alicyclic compounds such as alicyclic thiols, alicyclic thiocarboxylic acids, alicyclic dithiocarboxylic acids, and alicyclic polysulfides; and aromatic compounds. Specific examples of the organic sulfur compound (e) include thiophenols, thionaphthols, polysulfides, thiocarboxylic acids, dithiocarboxylic acids, sulfenamides, thiurams, dithiocarbamates, and thiazoles. Preferable organic sulfur compounds (e) are thiophenols, polysulfides having 2 to 4 sulfur atoms, thionaphthols, thiurams, and metal salts thereof. -
-
-
-
- In the chemical formula (4), R1 to R10 each represent H or a substituent, and M2 represents a bivalent metal atom.
- In the formulas (1) to (4), each substituent is at least one group selected from the group consisting of a halogen group (F, Cl, Br, I), an alkyl group, a carboxyl group (-COOH), an ester (-COOR)of a carboxyl group, a formyl group (-CHO), an acyl group (-COR), a carbonyl halide group (-COX), a sulfo group (-SO3H), an ester (-SO3R) of a sulfo group, a sulfonyl halide group (-SO2X), a sulfino group (-SO2H), an alkylsulfinyl group (-SOR), a carbamoyl group (-CONH2), an alkyl halide group, a cyano group (-CN), and an alkoxy group (-OR).
- Examples of the organic sulfur compound represented by the chemical formula (1) include thiophenol; thiophenols substituted with halogen groups, such as 4-fluorothiophenol, 2,5-difluorothiophenol, 2,4,5-trifluorothiophenol, 2,4,5,6-tetrafluorothiophenol, pentafluorothiophenol, 2-chlorothiophenol, 4-chlorothiophenol, 2,4-dichlorothiophenol, 2,5-dichlorothiophenol, 2,4,5-trichlorothiophenol, 2,4,5,6-tetrachlorothiophenol, pentachlorothiophenol, 4-bromothiophenol, 2,5-dibromothiophenol, 2,4,5-tribromothiophenol, 2,4,5,6-tetrabromothiophenol, pentabromothiophenol, 4-iodothiophenol, 2,5-diiodothiophenol, 2,4,5-triiodothiophenol, 2,4,5,6-tetraiodothiophenol, and pentaiodothiophenol; thiophenols substituted with alkyl groups, such as 4-methylthiophenol, 2,4,5-trimethylthiophenol, pentamethylthiophenol, 4-t-butylthiophenol, 2,4,5-tri-t-butylthiophenol, and penta-t-butylthiophenol; thiophenols substituted with carboxyl groups, such as 4-carboxythiophenol, 2,4,6-tricarboxythiophenol, and pentacarboxythiophenol; thiophenols substituted with alkoxycarbonyl groups, such as 4-methoxycarbonylthiophenol, 2,4,6-trimethoxycarbonylthiophenol, and pentamethoxycarbonylthiophenol; thiophenols substituted with formyl groups, such as 4-formylthiophenol, 2,4,6-triformylthiophenol, and pentaformylthiophenol; thiophenols substituted with acyl groups, such as 4-acetylthiophenol, 2,4,6-triacetylthiophenol, and pentaacetylthiophenol; thiophenols substituted with carbonyl halide groups, such as 4-chlorocarbonylthiophenol, 2,4,6-tri(chlorocarbonyl)thiophenol, and penta (chlorocarbonyl) thiophenol; thiophenols substituted with sulfo groups, such as 4-sulfothiophenol, 2,4,6-trisulfothiophenol, and pentasulfothiophenol; thiophenols substituted with alkoxysulfonyl groups, such as 4-methoxysulfonylthiophenol, 2,4,6-trimethoxysulfonylthiophenol, and pentamethoxysulfonylthiophenol; thiophenols substituted with sulfonyl halide groups, such as 4-chlorosulfonylthiophenol, 2,4,6-tri(chlorosulfonyl)thiophenol, and pent a (chlorosulfonyl) thiophenol; thiophenols substituted with sulfino groups, such as 4-sulfinothiophenol, 2,4,6-trisulfinothiophenol, and pentasulfinothiophenol; thiophenols substituted with alkylsulfinyl groups, such as 4-methylsulfinylthiophenol, 2,4,6-tri(methylsulfinyl)thiophenol, and pent a (methylsulfinyl) thiophenol; thiophenols substituted with carbamoyl groups, such as 4-carbamoylthiophenol, 2,4,6-tricarbamoylthiophenol, and pentacarbamoylthiophenol; thiophenols substituted with alkyl halide groups, such as 4-trichloromethylthiophenol, 2,4,6-tri(trichloromethyl)thiophenol, and penta(trichloromethyl)thiophenol; thiophenols substituted with cyano groups, such as 4-cyanothiophenol, 2,4,6-tricyanothiophenol, and pentacyanothiophenol; and thiophenols substituted with alkoxy groups, such as 4-methoxythiophenol, 2,4,6-trimethoxythiophenol, and pentamethoxythiophenol. Each of these thiophenols is substituted with one type of substituent.
- Another example of the organic sulfur compound represented by the chemical formula (1) is a compound substituted with at least one type of the above substituents and another substituent. Examples of the other substituent include a nitro group (-NO2), an amino group (-NH2), a hydroxyl group (-OH), and a phenylthio group (-SPh). Specific examples of the compound include 4-chloro-2-nitrothiophenol, 4-chloro-2-aminothiophenol, 4-chloro-2-hydroxythiophenol, 4-chloro-2-phenylthiothiophenol, 4-methyl-2-nitrothiophenol, 4-methyl-2-aminothiophenol, 4-methyl-2-hydroxythiophenol, 4-methyl-2-phenylthiothiophenol, 4-carboxy-2-nitrothiophenol, 4-carboxy-2-aminothiophenol, 4-carboxy-2-hydroxythiophenol, 4-carboxy-2-phenylthiothiophenol, 4-methoxycarbonyl-2-nitrothiophenol, 4-methoxycarbonyl-2-aminothiophenol, 4-methoxycarbonyl-2-hydroxythiophenol, 4-methoxycarbonyl-2-phenylthiothiophenol, 4-formyl-2-nitrothiophenol, 4-formyl-2-aminothiophenol, 4-formyl-2-hydroxythiophenol, 4-formyl-2-phenylthiothiophenol, 4-acetyl-2-nitrothiophenol, 4-acetyl-2-aminothiophenol, 4-acetyl-2-hydroxythiophenol, 4-acetyl-2-phenylthiothiophenol, 4-chlorocarbonyl-2-nitrothiophenol, 4-chlorocarbonyl-2-aminothiophenol, 4-chlorocarbonyl-2-hydroxythiophenol, 4-chlorocarbonyl-2-phenylthiothiophenol, 4-sulfo-2-nitrothiophenol, 4-sulfo-2-aminothiophenol, 4-sulfo-2-hydroxythiophenol, 4-sulfo-2-phenylthiothiophenol, 4-methoxysulfonyl-2-nitrothiophenol, 4-methoxysulfonyl-2-aminothiophenol, 4-methoxysulfonyl-2-hydroxythiophenol, 4-methoxysulfonyl-2-phenylthiothiophenol, 4-chlorosulfonyl-2-nitrothiophenol, 4-chlorosulfonyl-2-aminothiophenol, 4-chlorosulfonyl-2-hydroxythiophenol, 4-chlorosulfonyl-2-phenylthiothiophenol, 4-sulfino-2-nitrothiophenol, 4-sulfino-2-aminothiophenol, 4-sulfino-2-hydroxythiophenol, 4-sulfino-2-phenylthiothiophenol, 4-methylsulfinyl-2-nitrothiophenol, 4-methylsulfinyl-2-aminothiophenol, 4-methylsulfinyl-2-hydroxythiophenol, 4-methylsulfinyl-2-phenylthiothiophenol, 4-carbamoyl-2-nitrothiophenol, 4-carbamoyl-2-aminothiophenol, 4-carbamoyl-2-hydroxythiophenol, 4-carbamoyl-2-phenylthiothiophenol, 4-trichloromethyl-2-nitrothiophenol, 4-trichloromethyl-2-aminothiophenol, 4-trichloromethyl-2-hydroxythiophenol, 4-trichloromethyl-2-phenylthiothiophenol, 4-cyano-2-nitrothiophenol, 4-cyano-2-aminothiophenol, 4-cyano-2-hydroxythiophenol, 4-cyano-2-phenylthiothiophenol, 4-methoxy-2-nitrothiophenol, 4-methoxy-2-aminothiophenol, 4-methoxy-2-hydroxythiophenol, and 4-methoxy-2-phenylthiothiophenol.
- Still another example of the organic sulfur compound represented by the chemical formula (1) is a compound substituted with two or more types of substituents. Specific examples of the compound include 4-acetyl-2-chlorothiophenol, 4-acetyl-2-methylthiophenol, 4-acetyl-2-carboxythiophenol, 4-acetyl-2-methoxycarbonylthiophenol, 4-acetyl-2-formylthiophenol, 4-acetyl-2-chlorocarbonylthiophenol, 4-acetyl-2-sulfothiophenol, 4-acetyl-2-methoxysulfonylthiophenol, 4-acetyl-2-chlorosulfonylthiophenol, 4-acetyl-2-sulfinothiophenol, 4-acetyl-2-methylsulfinylthiophenol, 4-acetyl-2-carbamoylthiophenol, 4-acetyl-2-trichloromethylthiophenol, 4-acetyl-2-cyanothiophenol, and 4-acetyl-2-methoxythiophenol.
- Examples of the organic sulfur compound represented by the chemical formula (2) include diphenyl disulfide; diphenyl disulfides substituted with halogen groups, such as bis(4-fluorophenyl)disulfide, bis(2,5-difluorophenyl)disulfide, bis(2,4,5-trifluorophenyl)disulfide, bis(2,4,5,6-tetrafluorophenyl)disulfide, bis(pentafluorophenyl)disulfide, bis(4-chlorophenyl)disulfide, bis(2,5-dichlorophenyl)disulfide, bis(2,4,5-trichlorophenyl)disulfide, bis(2,4,5,6-tetrachlorophenyl)disulfide, bis(pentachlorophenyl)disulfide, bis(4-bromophenyl)disulfide, bis(2,5-dibromophenyl)disulfide, bis(2,4,5-tribromophenyl)disulfide, bis(2,4,5,6-tetrabromophenyl)disulfide, bis(pentabromophenyl)disulfide, bis(4-iodophenyl)disulfide, bis(2,5-diiodophenyl)disulfide, bis(2,4,5-triiodophenyl)disulfide, bis(2,4,5,6-tetraiodophenyl)disulfide, and bis(pentaiodophenyl)disulfide; diphenyl disulfides substituted with alkyl groups, such as bis(4-methylphenyl)disulfide, bis(2,4,5-trimethylphenyl)disulfide, bis(pentamethylphenyl)disulfide, bis(4-t-butylphenyl)disulfide, bis(2,4,5-tri-t-butylphenyl)disulfide, and bis(penta-t-butylphenyl)disulfide; diphenyl disulfides substituted with carboxyl groups, such as bis(4-carboxyphenyl)disulfide, bis(2,4,6-tricarboxyphenyl)disulfide, and bis(pentacarboxyphenyl)disulfide; diphenyl disulfides substituted with alkoxycarbonyl groups, such as bis(4-methoxycarbonylphenyl)disulfide, bis(2,4,6-trimethoxycarbonylphenyl)disulfide, and bis(pentamethoxycarbonylphenyl)disulfide; diphenyl disulfides substituted with formyl groups, such as bis(4-formylphenyl)disulfide, bis(2,4,6-triformylphenyl)disulfide, and bis(pentaformylphenyl)disulfide; diphenyl disulfides substituted with acyl groups, such as bis(4-acetylphenyl)disulfide, bis(2,4,6-triacetylphenyl)disulfide, and bis(pentaacetylphenyl)disulfide; diphenyl disulfides substituted with carbonyl halide groups, such as bis(4-chlorocarbonylphenyl)disulfide, bis(2,4,6-tri(chlorocarbonyl)phenyl)disulfide, and bis (penta (chlorocarbonyl) phenyl) disulfide; diphenyl disulfides substituted with sulfo groups, such as bis(4-sulfophenyl)disulfide, bis(2,4,6-trisulfophenyl)disulfide, and bis(pentasulfophenyl)disulfide; diphenyl disulfides substituted with alkoxysulfonyl groups, such as bis(4-methoxysulfonylphenyl)disulfide, bis(2,4,6-trimethoxysulfonylphenyl)disulfide, and bis(pentamethoxysulfonylphenyl)disulfide; diphenyl disulfides substituted with sulfonyl halide groups, such as bis(4-chlorosulfonylphenyl)disulfide, bis (2,4,6-tri (chlorosulfonyl) phenyl) disulfide, and bis (penta (chlorosulfonyl) phenyl) disulfide; diphenyl disulfides substituted with sulfino groups, such as bis(4-sulfinophenyl)disulfide, bis(2,4,6-trisulfinophenyl)disulfide, and bis(pentasulfinophenyl)disulfide; diphenyl disulfides substituted with alkylsulfinyl groups, such as bis(4-methylsulfinylphenyl)disulfide, bis(2,4,6-tri(methylsulfinyl)phenyl)disulfide, and bis (penta (methylsulfinyl) phenyl) disulfide; diphenyl disulfides substituted with carbamoyl groups, such as bis(4-carbamoylphenyl)disulfide, bis(2,4,6-tricarbamoylphenyl)disulfide, and bis(pentacarbamoylphenyl)disulfide; diphenyl disulfides substituted with alkyl halide groups, such as bis(4-trichloromethylphenyl)disulfide, bis(2,4,6-tri(trichloromethyl)phenyl)disulfide, and bis(penta(trichloromethyl)phenyl)disulfide; diphenyl disulfides substituted with cyano groups, such as bis(4-cyanophenyl)disulfide, bis(2,4,6-tricyanophenyl)disulfide, and bis(pentacyanophenyl)disulfide; and diphenyl disulfides substituted with alkoxy groups, such as bis(4-methoxyphenyl)disulfide, bis(2,4,6-trimethoxyphenyl)disulfide, and bis(pentamethoxyphenyl)disulfide. Each of these diphenyl disulfides is substituted with one type of substituent.
- Another example of the organic sulfur compound represented by the chemical formula (2) is a compound substituted with at least one type of the above substituents and another substituent. Examples of the other substituent include a nitro group (-NO2), an amino group (-NH2), a hydroxyl group (-OH), and a phenylthio group (-SPh). Specific examples of the compound include bis(4-chloro-2-nitrophenyl)disulfide, bis(4-chloro-2-aminophenyl)disulfide, bis(4-chloro-2-hydroxyphenyl)disulfide, bis(4-chloro-2-phenylthiophenyl)disulfide, bis(4-methyl-2-nitrophenyl)disulfide, bis(4-methyl-2-aminophenyl)disulfide, bis(4-methyl-2-hydroxyphenyl)disulfide, bis(4-methyl-2-phenylthiophenyl)disulfide, bis(4-carboxy-2-nitrophenyl)disulfide, bis(4-carboxy-2-aminophenyl)disulfide, bis(4-carboxy-2-hydroxyphenyl)disulfide, bis(4-carboxy-2-phenylthiophenyl)disulfide, bis(4-methoxycarbonyl-2-nitrophenyl)disulfide, bis(4-methoxycarbonyl-2-aminophenyl)disulfide, bis(4-methoxycarbonyl-2-hydroxyphenyl)disulfide, bis(4-methoxycarbonyl-2-phenylthiophenyl)disulfide, bis(4-formyl-2-nitrophenyl)disulfide, bis(4-formyl-2-aminophenyl)disulfide, bis(4-formyl-2-hydroxyphenyl)disulfide, bis(4-formyl-2-phenylthiophenyl)disulfide, bis(4-acetyl-2-nitrophenyl)disulfide, bis(4-acetyl-2-aminophenyl)disulfide, bis(4-acetyl-2-hydroxyphenyl)disulfide, bis(4-acetyl-2-phenylthiophenyl)disulfide, bis(4-chlorocarbonyl-2-nitrophenyl)disulfide, bis(4-chlorocarbonyl-2-aminophenyl)disulfide, bis(4-chlorocarbonyl-2-hydroxyphenyl)disulfide, bis(4-chlorocarbonyl-2-phenylthiophenyl)disulfide, bis(4-sulfo-2-nitrophenyl)disulfide, bis(4-sulfo-2-aminophenyl)disulfide, bis(4-sulfo-2-hydroxyphenyl)disulfide, bis(4-sulfo-2-phenylthiophenyl)disulfide, bis(4-methoxysulfonyl-2-nitrophenyl)disulfide, bis(4-methoxysulfonyl-2-aminophenyl)disulfide, bis(4-methoxysulfonyl-2-hydroxyphenyl)disulfide, bis(4-methoxysulfonyl-2-phenylthiophenyl)disulfide, bis(4-chlorosulfonyl-2-nitrophenyl)disulfide, bis(4-chlorosulfonyl-2-aminophenyl)disulfide, bis(4-chlorosulfonyl-2-hydroxyphenyl)disulfide, bis(4-chlorosulfonyl-2-phenylthiophenyl)disulfide, bis(4-sulfino-2-nitrophenyl)disulfide, bis(4-sulfino-2-aminophenyl)disulfide, bis(4-sulfino-2-hydroxyphenyl)disulfide, bis(4-sulfino-2-phenylthiophenyl)disulfide, bis(4-methylsulfinyl-2-nitrophenyl)disulfide, bis(4-methylsulfinyl-2-aminophenyl)disulfide, bis(4-methylsulfinyl-2-hydroxyphenyl)disulfide, bis(4-methylsulfinyl-2-phenylthiophenyl)disulfide, bis(4-carbamoyl-2-nitrophenyl)disulfide, bis(4-carbamoyl-2-aminophenyl)disulfide, bis(4-carbamoyl-2-hydroxyphenyl)disulfide, bis(4-carbamoyl-2-phenylthiophenyl)disulfide, bis(4-trichloromethyl-2-nitrophenyl)disulfide, bis(4-trichloromethyl-2-aminophenyl)disulfide, bis(4-trichloromethyl-2-hydroxyphenyl)disulfide, bis(4-trichloromethyl-2-phenylthiophenyl)disulfide, bis(4-cyano-2-nitrophenyl)disulfide, bis(4-cyano-2-aminophenyl)disulfide, bis(4-cyano-2-hydroxyphenyl)disulfide, bis(4-cyano-2-phenylthiophenyl)disulfide, bis(4-methoxy-2-nitrophenyl)disulfide, bis(4-methoxy-2-aminophenyl)disulfide, bis(4-methoxy-2-hydroxyphenyl)disulfide, and bis(4-methoxy-2-phenylthiophenyl)disulfide.
- Still another example of the organic sulfur compound represented by the chemical formula (2) is a compound substituted with two or more types of substituents. Specific examples of the compound include bis(4-acetyl-2-chlorophenyl)disulfide, bis(4-acetyl-2-methylphenyl)disulfide, bis(4-acetyl-2-carboxyphenyl)disulfide, bis(4-acetyl-2-methoxycarbonylphenyl)disulfide, bis(4-acetyl-2-formylphenyl)disulfide, bis(4-acetyl-2-chlorocarbonylphenyl)disulfide, bis(4-acetyl-2-sulfophenyl)disulfide, bis(4-acetyl-2-methoxysulfonylphenyl)disulfide, bis(4-acetyl-2-chlorosulfonylphenyl)disulfide, bis(4-acetyl-2-sulfinophenyl)disulfide, bis(4-acetyl-2-methylsulfinylphenyl)disulfide, bis(4-acetyl-2-carbamoylphenyl)disulfide, bis(4-acetyl-2-trichloromethylphenyl)disulfide, bis(4-acetyl-2-cyanophenyl)disulfide, and bis(4-acetyl-2-methoxyphenyl)disulfide.
- Examples of the organic sulfur compound represented by the chemical formula (3) include thiophenol sodium salt; thiophenol sodium salts substituted with halogen groups, such as 4-fluorothiophenol sodium salt, 2,5-difluorothiophenol sodium salt, 2,4,5-trifluorothiophenol sodium salt, 2,4,5,6-tetrafluorothiophenol sodium salt, pentafluorothiophenol sodium salt, 4-chlorothiophenol sodium salt, 2,5-dichlorothiophenol sodium salt, 2,4,5-trichlorothiophenol sodium salt, 2,4,5,6-tetrachlorothiophenol sodium salt, pentachlorothiophenol sodium salt, 4-bromothiophenol sodium salt, 2,5-dibromothiophenol sodium salt, 2,4,5-tribromothiophenol sodium salt, 2,4,5,6-tetrabromothiophenol sodium salt, pentabromothiophenol sodium salt, 4-iodothiophenol sodium salt, 2,5-diiodothiophenol sodium salt, 2,4,5-triiodothiophenol sodium salt, 2,4,5,6-tetraiodothiophenol sodium salt, and pentaiodothiophenol sodium salt; thiophenol sodium salts substituted with alkyl groups, such as 4-methylthiophenol sodium salt, 2,4,5-trimethylthiophenol sodium salt, pentamethylthiophenol sodium salt, 4-t-butylthiophenol sodium salt, 2,4,5-tri-t-butylthiophenol sodium salt, and penta(t-butyl)thiophenol sodium salt; thiophenol sodium salts substituted with carboxyl groups, such as 4-carboxythiophenol sodium salt, 2,4,6-tricarboxythiophenol sodium salt, and pentacarboxythiophenol sodium salt; thiophenol sodium salts substituted with alkoxycarbonyl groups, such as 4-methoxycarbonylthiophenol sodium salt, 2,4,6-trimethoxycarbonylthiophenol sodium salt, and pentamethoxycarbonylthiophenol sodium salt; thiophenol sodium salts substituted with formyl groups, such as 4-formylthiophenol sodium salt, 2,4,6-triformylthiophenol sodium salt, and pentaformylthiophenol sodium salt; thiophenol sodium salts substituted with acyl groups, such as 4-acetylthiophenol sodium salt, 2,4,6-triacetylthiophenol sodium salt, and pentaacetylthiophenol sodium salt; thiophenol sodium salts substituted with carbonyl halide groups, such as 4-chlorocarbonylthiophenol sodium salt, 2,4,6-tri(chlorocarbonyl)thiophenol sodium salt, and pent a (chlorocarbonyl) thiophenol sodium salt; thiophenol sodium salts substituted with sulfo groups, such as 4-sulfothiophenol sodium salt, 2,4,6-trisulfothiophenol sodium salt, and pentasulfothiophenol sodium salt; thiophenol sodium salts substituted with alkoxysulfonyl groups, such as 4-methoxysulfonylthiophenol sodium salt, 2,4,6-trimethoxysulfonylthiophenol sodium salt, and pentamethoxysulfonylthiophenol sodium salt; thiophenol sodium salts substituted with sulfonyl halide groups, such as 4-chlorosulfonylthiophenol sodium salt, 2,4,6-tri(chlorosulfonyl)thiophenol sodium salt, and pent a (chlorosulfonyl) thiophenol sodium salt; thiophenol sodium salts substituted with sulfino groups, such as 4-sulfinothiophenol sodium salt, 2,4,6-trisulfinothiophenol sodium salt, and pentasulfinothiophenol sodium salt; thiophenol sodium salts substituted with alkylsulfinyl groups, such as 4-methylsulfinylthiophenol sodium salt, 2,4,6-tri(methylsulfinyl)thiophenol sodium salt, and penta(methylsulfinyl)thiophenolsodiumsalt; thiophenolsodium salts substituted with carbamoyl groups, such as 4-carbamoylthiophenol sodium salt, 2,4,6-tricarbamoylthiophenol sodium salt, and pentacarbamoylthiophenol sodium salt; thiophenol sodium salts substituted with alkyl halide groups, such as 4-trichloromethylthiophenol sodium salt, 2,4,6-tri(trichloromethyl)thiophenol sodium salt, and penta(trichloromethyl)thiophenol sodium salt; thiophenol sodium salts substituted with cyano groups, such as 4-cyanothiophenol sodium salt, 2,4,6-tricyanothiophenol sodium salt, and pentacyanothiophenol sodium salt; and thiophenol sodium salts substituted with alkoxy groups, such as 4-methoxythiophenol sodium salt, 2,4,6-trimethoxythiophenol sodium salt, and pentamethoxythiophenol sodium salt. Each of these thiophenol sodium salts is substituted with one type of substituent.
- Another example of the organic sulfur compound represented by the chemical formula (3) is a compound substituted with at least one type of the above substituents and another substituent. Examples of the other substituent include a nitro group (-NO2), an amino group (-NH2), a hydroxyl group (-OH), and a phenylthio group (-SPh). Specific examples of the compound include 4-chloro-2-nitrothiophenol sodium salt, 4-chloro-2-aminothiophenol sodium salt, 4-chloro-2-hydroxythiophenol sodium salt, 4-chloro-2-phenylthiothiophenol sodium salt, 4-methyl-2-nitrothiophenol sodium salt, 4-methyl-2-aminothiophenol sodium salt, 4-methyl-2-hydroxythiophenol sodium salt, 4-methyl-2-phenylthiothiophenol sodium salt, 4-carboxy-2-nitrothiophenol sodium salt, 4-carboxy-2-aminothiophenol sodium salt, 4-carboxy-2-hydroxythiophenol sodium salt, 4-carboxy-2-phenylthiothiophenol sodium salt, 4-methoxycarbonyl-2-nitrothiophenol sodium salt, 4-methoxycarbonyl-2-aminothiophenol sodium salt, 4-methoxycarbonyl-2-hydroxythiophenol sodium salt, 4-methoxycarbonyl-2-phenylthiothiophenol sodium salt, 4-formyl-2-nitrothiophenol sodium salt, 4-formyl-2-aminothiophenol sodium salt, 4-formyl-2-hydroxythiophenol sodium salt, 4-formyl-2-phenylthiothiophenol sodium salt, 4-acetyl-2-nitrothiophenol sodium salt, 4-acetyl-2-aminothiophenol sodium salt, 4-acetyl-2-hydroxythiophenol sodium salt, 4-acetyl-2-phenylthiothiophenol sodium salt, 4-chlorocarbonyl-2-nitrothiophenol sodium salt, 4-chlorocarbonyl-2-aminothiophenol sodium salt, 4-chlorocarbonyl-2-hydroxythiophenol sodium salt, 4-chlorocarbonyl-2-phenylthiothiophenol sodium salt, 4-sulfo-2-nitrothiophenol sodium salt, 4-sulfo-2-aminothiophenol sodium salt, 4-sulfo-2-hydroxythiophenol sodium salt, 4-sulfo-2-phenylthiothiophenol sodium salt, 4-methoxysulfonyl-2-nitrothiophenol sodium salt, 4-methoxysulfonyl-2-aminothiophenol sodium salt, 4-methoxysulfonyl-2-hydroxythiophenol sodium salt, 4-methoxysulfonyl-2-phenylthiothiophenol sodium salt, 4-chlorosulfonyl-2-nitrothiophenol sodium salt, 4-chlorosulfonyl-2-aminothiophenol sodium salt, 4-chlorosulfonyl-2-hydroxythiophenol sodium salt, 4-chlorosulfonyl-2-phenylthiothiophenol sodium salt, 4-sulfino-2-nitrothiophenol sodium salt, 4-sulfino-2-aminothiophenol sodium salt, 4-sulfino-2-hydroxythiophenol sodium salt, 4-sulfino-2-phenylthiothiophenol sodium salt, 4-methylsulfinyl-2-nitrothiophenol sodium salt, 4-methylsulfinyl-2-aminothiophenol sodium salt, 4-methylsulfinyl-2-hydroxythiophenol sodium salt, 4-methylsulfinyl-2-phenylthiothiophenol sodium salt, 4-carbamoyl-2-nitrothiophenol sodium salt, 4-carbamoyl-2-aminothiophenol sodium salt, 4-carbamoyl-2-hydroxythiophenol sodium salt, 4-carbamoyl-2-phenylthiothiophenol sodium salt, 4-trichloromethyl-2-nitrothiophenol sodium salt, 4-trichloromethyl-2-aminothiophenol sodium salt, 4-trichloromethyl-2-hydroxythiophenol sodium salt, 4-trichloromethyl-2-phenylthiothiophenol sodium salt, 4-cyano-2-nitrothiophenol sodium salt, 4-cyano-2-aminothiophenol sodium salt, 4-cyano-2-hydroxythiophenol sodium salt, 4-cyano-2-phenylthiothiophenol sodium salt, 4-methoxy-2-nitrothiophenol sodium salt, 4-methoxy-2-aminothiophenol sodium salt, 4-methoxy-2-hydroxythiophenol sodium salt, and 4-methoxy-2-phenylthiothiophenol sodium salt.
- Still another example of the organic sulfur compound represented by the chemical formula (3) is a compound substituted with two or more types of substituents. Specific examples of the compound include 4-acetyl-2-chlorothiophenol sodium salt, 4-acetyl-2-methylthiophenol sodium salt, 4-acetyl-2-carboxythiophenol sodium salt, 4-acetyl-2-methoxycarbonylthiophenol sodium salt, 4-acetyl-2-formylthiophenol sodium salt, 4-acetyl-2-chlorocarbonylthiophenol sodium salt, 4-acetyl-2-sulfothiophenol sodium salt, 4-acetyl-2-methoxysulfonylthiophenol sodium salt, 4-acetyl-2-chlorosulfonylthiophenol sodium salt, 4-acetyl-2-sulfinothiophenol sodium salt, 4-acetyl-2-methylsulfinylthiophenol sodium salt, 4-acetyl-2-carbamoylthiophenol sodium salt, 4-acetyl-2-trichloromethylthiophenol sodium salt, 4-acetyl-2-cyanothiophenol sodium salt, and 4-acetyl-2-methoxythiophenol sodium salt. Examples of the monovalent metal represented by M1 in the chemical formula (3) include sodium, lithium, potassium, copper (I), and silver (I).
- Examples of the organic sulfur compound represented by the chemical formula (4) include thiophenol zinc salt; thiophenol zinc salts substituted with halogen groups, such as 4-fluorothiophenol zinc salt, 2,5-difluorothiophenol zinc salt, 2,4,5-trifluorothiophenol zinc salt, 2,4,5,6-tetrafluorothiophenol zinc salt, pentafluorothiophenolzinc salt, 4-chlorothiophenol zinc salt, 2,5-dichlorothiophenol zinc salt, 2,4,5-trichlorothiophenol zinc salt, 2,4,5,6-tetrachlorothiophenol zinc salt, pentachlorothiophenol zinc salt, 4-bromothiophenol zinc salt, 2,5-dibromothiophenol zinc salt, 2,4,5-tribromothiophenol zinc salt, 2,4,5,6-tetrabromothiophenol zinc salt, pentabromothiophenol zinc salt, 4-iodothiophenol zinc salt, 2,5-diiodothiophenol zinc salt, 2,4,5-triiodothiophenol zinc salt, 2,4,5,6-tetraiodothiophenol zinc salt, and pentaiodothiophenol zinc salt; thiophenol zinc salts substituted with alkyl groups, such as 4-methylthiophenol zinc salt, 2,4,5-trimethylthiophenol zinc salt, pentamethylthiophenol zinc salt, 4-t-butylthiophenol zinc salt, 2,4,5-tri-t-butylthiophenol zinc salt, and penta-t-butylthiophenol zinc salt; thiophenol zinc salts substituted with carboxyl groups, such as 4-carboxythiophenol zinc salt, 2,4,6-tricarboxythiophenol zinc salt, and pentacarboxythiophenol zinc salt; thiophenol zinc salts substituted with alkoxycarbonyl groups, such as 4-methoxycarbonylthiophenol zinc salt, 2,4,6-trimethoxycarbonylthiophenol zinc salt, and pentamethoxycarbonylthiophenol zinc salt; thiophenol zinc salts substituted with formyl groups, such as 4-formylthiophenol zinc salt, 2,4,6-triformylthiophenol zinc salt, and pentaformylthiophenol zinc salt; thiophenol zinc salts substituted with acyl groups, such as 4-acetylthiophenol zinc salt, 2,4,6-triacetylthiophenol zinc salt, and pentaacetylthiophenol zinc salt; thiophenol zinc salts substituted with carbonyl halide groups, such as 4-chlorocarbonylthiophenol zinc salt, 2,4,6-tri(chlorocarbonyl)thiophenol zinc salt, and penta(chlorocarbonyl)thiophenol zinc salt; thiophenol zinc salts substituted with sulfo groups, such as 4-sulfothiophenol zinc salt, 2,4,6-trisulfothiophenol zinc salt, and pentasulfothiophenol zinc salt; thiophenol zinc salts substituted with alkoxysulfonyl groups, such as 4-methoxysulfonylthiophenol zinc salt, 2,4,6-trimethoxysulfonylthiophenol zinc salt, and pentamethoxysulfonylthiophenol zinc salt; thiophenol zinc salts substituted with sulfonyl halide groups, such as 4-chlorosulfonylthiophenol zinc salt, 2,4,6-tri(chlorosulfonyl)thiophenol zinc salt, and penta(chlorosulfonyl)thiophenol zinc salt; thiophenol zinc salts substituted with sulfino groups, such as 4-sulfinothiophenol zinc salt, 2,4,6-trisulfinothiophenol zinc salt, and pentasulfinothiophenol zinc salt; thiophenol zinc salts substituted with alkylsulfinyl groups, such as 4-methylsulfinylthiophenol zinc salt, 2,4,6-tri(methylsulfinyl)thiophenol zinc salt, and penta(methylsulfinyl)thiophenol zinc salt; thiophenol zinc salts substituted with carbamoyl groups, such as 4-carbamoylthiophenol zinc salt, 2,4,6-tricarbamoylthiophenol zinc salt, and pentacarbamoylthiophenol zinc salt; thiophenol zinc salts substituted with alkyl halide groups, such as 4-trichloromethylthiophenol zinc salt, 2,4,6-tri(trichloromethyl)thiophenol zinc salt, and penta(trichloromethyl)thiophenol zinc salt; thiophenol zinc salts substituted with cyano groups, such as 4-cyanothiophenol zinc salt, 2,4,6-tricyanothiophenol zinc salt, and pentacyanothiophenol zinc salt; and thiophenol zinc salts substituted with alkoxy groups, such as 4-methoxythiophenol zinc salt, 2,4,6-trimethoxythiophenol zinc salt, and pentamethoxythiophenol zinc salt. Each of these thiophenol zinc salts is substituted with one type of substituent.
- Another example of the organic sulfur compound represented by the chemical formula (4) is a compound substituted with at least one type of the above substituents and another substituent. Examples of the other substituent include a nitro group (-NO2), an amino group (-NH2), a hydroxyl group (-OH), and a phenylthio group (-SPh). Specific examples of the compound include 4-chloro-2-nitrothiophenol zinc salt, 4-chloro-2-aminothiophenol zinc salt, 4-chloro-2-hydroxythiophenol zinc salt, 4-chloro-2-phenylthiothiophenol zinc salt, 4-methyl-2-nitrothiophenol zinc salt, 4-methyl-2-aminothiophenol zinc salt, 4-methyl-2-hydroxythiophenol zinc salt, 4-methyl-2-phenylthiothiophenol zinc salt, 4-carboxy-2-nitrothiophenol zinc salt, 4-carboxy-2-aminothiophenol zinc salt, 4-carboxy-2-hydroxythiophenol zinc salt, 4-carboxy-2-phenylthiothiophenol zinc salt, 4-methoxycarbonyl-2-nitrothiophenol zinc salt, 4-methoxycarbonyl-2-aminothiophenol zinc salt, 4-methoxycarbonyl-2-hydroxythiophenol zinc salt, 4-methoxycarbonyl-2-phenylthiothiophenol zinc salt, 4-formyl-2-nitrothiophenol zinc salt, 4-formyl-2-aminothiophenol zinc salt, 4-formyl-2-hydroxythiophenol zinc salt, 4-formyl-2-phenylthiothiophenol zinc salt, 4-acetyl-2-nitrothiophenol zinc salt, 4-acetyl-2-aminothiophenol zinc salt, 4-acetyl-2-hydroxythiophenol zinc salt, 4-acetyl-2-phenylthiothiophenol zinc salt, 4-chlorocarbonyl-2-nitrothiophenol zinc salt, 4-chlorocarbonyl-2-aminothiophenol zinc salt, 4-chlorocarbonyl-2-hydroxythiophenol zinc salt, 4-chlorocarbonyl-2-phenylthiothiophenol zinc salt, 4-sulfo-2-nitrothiophenol zinc salt, 4-sulfo-2-aminothiophenol zinc salt, 4-sulfo-2-hydroxythiophenol zinc salt, 4-sulfo-2-phenylthiothiophenol zinc salt, 4-methoxysulfonyl-2-nitrothiophenol zinc salt, 4-methoxysulfonyl-2-aminothiophenol zinc salt, 4-methoxysulfonyl-2-hydroxythiophenol zinc salt, 4-methoxysulfonyl-2-phenylthiothiophenol zinc salt, 4-chlorosulfonyl-2-nitrothiophenol zinc salt, 4-chlorosulfonyl-2-aminothiophenol zinc salt, 4-chlorosulfonyl-2-hydroxythiophenol zinc salt, 4-chlorosulfonyl-2-phenylthiothiophenol zinc salt, 4-sulfino-2-nitrothiophenol zinc salt, 4-sulfino-2-aminothiophenol zinc salt, 4-sulfino-2-hydroxythiophenol zinc salt, 4-sulfino-2-phenylthiothiophenol zinc salt, 4-methylsulfinyl-2-nitrothiophenol zinc salt, 4-methylsulfinyl-2-aminothiophenol zinc salt, 4-methylsulfinyl-2-hydroxythiophenol zinc salt, 4-methylsulfinyl-2-phenylthiothiophenol zinc salt, 4-carbamoyl-2-nitrothiophenol zinc salt, 4-carbamoyl-2-aminothiophenol zinc salt, 4-carbamoyl-2-hydroxythiophenol zinc salt, 4-carbamoyl-2-phenylthiothiophenol zinc salt, 4-trichloromethyl-2-nitrothiophenol zinc salt, 4-trichloromethyl-2-aminothiophenol zinc salt, 4-trichloromethyl-2-hydroxythiophenol zinc salt, 4-trichloromethyl-2-phenylthiothiophenol zinc salt, 4-cyano-2-nitrothiophenol zinc salt, 4-cyano-2-aminothiophenol zinc salt, 4-cyano-2-hydroxythiophenol zinc salt, 4-cyano-2-phenylthiothiophenol zinc salt, 4-methoxy-2-nitrothiophenol zinc salt, 4-methoxy-2-aminothiophenol zinc salt, 4-methoxy-2-hydroxythiophenol zinc salt, and 4-methoxy-2-phenylthiothiophenol zinc salt.
- Still another example of the organic sulfur compound represented by the chemical formula (4) is a compound substituted with two or more types of substituents. Specific examples of the compound include 4-acetyl-2-chlorothiophenol zinc salt, 4-acetyl-2-methylthiophenol zinc salt, 4-acetyl-2-carboxythiophenol zinc salt, 4-acetyl-2-methoxycarbonylthiophenol zinc salt, 4-acetyl-2-formylthiophenol zinc salt, 4-acetyl-2-chlorocarbonylthiophenol zinc salt, 4-acetyl-2-sulfothiophenol zinc salt, 4-acetyl-2-methoxysulfonylthiophenol zinc salt, 4-acetyl-2-chlorosulfonylthiophenol zinc salt, 4-acetyl-2-sulfinothiophenol zinc salt, 4-acetyl-2-methylsulfinylthiophenol zinc salt, 4-acetyl-2-carbamoylthiophenol zinc salt, 4-acetyl-2-trichloromethylthiophenol zinc salt, 4-acetyl-2-cyanothiophenol zinc salt, and 4-acetyl-2-methoxythiophenol zinc salt. Examples of the bivalent metal represented by M2 in the chemical formula (4) include zinc, magnesium, calcium, strontium, barium, titanium (II), manganese (II), iron (II), cobalt (II), nickel (II), zirconium (II), and tin (II).
- Examples of thionaphthols include 2-thionaphthol, 1-thionaphthol, 2-chloro-1-thionaphthol, 2-bromo-1-thionaphthol, 2-fluoro-1-thionaphthol, 2-cyano-1-thionaphthol, 2-acetyl-1-thionaphthol, 1-chloro-2-thionaphthol, 1-bromo-2-thionaphthol, 1-fluoro-2-thionaphthol, 1-cyano-2-thionaphthol, 1-acetyl-2-thionaphthol, and metal salts thereof. 1-thionaphthol, 2-thionaphthol, and zinc salts thereof are preferred.
- Examples of sulfenamide type organic sulfur compounds include N-cyclohexyl-2-benzothiazole sulfenamide, N-oxydiethylene-2-benzothiazole sulfenamide, and N-t-butyl-2-benzothiazole sulfenamide. Examples of thiuram type organic sulfur compounds include tetramethylthiuram monosulfide, tetramethylthiuram disulfide, tetraethylthiuram disulfide, tetrabutylthiuram disulfide, and dipentamethylenethiuram tetrasulfide. Examples of dithiocarbamates include zinc dimethyldithiocarbamate, zinc diethyldithiocarbamate, zinc dibutyldithiocarbamate, zinc ethylphenyldithiocarbamate, sodium dimethyldithiocarbamate, sodium diethyldithiocarbamate, copper (II) dimethyldithiocarbamate, iron (III) dimethyldithiocarbamate, selenium diethyldithiocarbamate, and tellurium diethyldithiocarbamate. Examples of thiazole type organic sulfur compounds include 2-mercaptobenzothiazole (MBT); dibenzothiazyl disulfide (MBTS); a sodium salt, a zinc salt, a copper salt, or a cyclohexylamine salt of 2-mercaptobenzothiazole; 2-(2,4-dinitrophenyl)mercaptobenzothiazole; and 2-(2,6-diethyl-4-morpholinothio)benzothiazole.
- In light of resilience performance, the amount of the organic sulfur compound (e) is preferably equal to or greater than 0.05 parts by weight and particularly preferably equal to or greater than 0.1 parts by weight, per 100 parts by weight of the base rubber. In light of resilience performance, the amount is preferably equal to or less than 5.0 parts by weight and particularly preferably equal to or less than 2.0 parts by weight, per 100 parts by weight of the base rubber.
- For the purpose of adjusting specific gravity and the like, a filler may be included in the
core 4. Examples of suitable fillers include zinc oxide, barium sulfate, calcium carbonate, and magnesium carbonate. The amount of the filler is determined as appropriate so that the intended specific gravity of thecore 4 is accomplished. A particularly preferable filler is zinc oxide. Zinc oxide serves not only as a specific gravity adjuster but also as a crosslinking activator. - According to need, an anti-aging agent, a coloring agent, a plasticizer, a dispersant, sulfur, a vulcanization accelerator, and the like are added to the rubber composition of the
core 4. Crosslinked rubber powder or synthetic resin powder may also be dispersed in the rubber composition. - In the
core 4, the difference (Hs-H(0)) between the surface hardness Hs and the central hardness H(0) is preferably equal to or greater than 15. The difference is great. In other words, thecore 4 has an outer-hard/inner-soft structure. When thecore 4 is hit with a middle iron, the recoil (torsional return) is great, and thus spin is suppressed. Thecore 4 contributes to the flight performance of thegolf ball 2. In light of flight performance, the difference (Hs-H(0)) is more preferably equal to or greater than 20 and particularly preferably equal to or greater than 25. From the standpoint that thecore 4 can easily be formed, the difference (Hs-H(0)) is preferably equal to or less than 50. - The hardness H(0) at the central point of the
core 4 is preferably equal to or greater than 40.0 but equal to or less than 70.0. Thegolf ball 2 having a hardness H(0) of 40.0 or greater has excellent resilience performance. In this respect, the hardness H(0) is more preferably equal to or greater than 45.0 and particularly preferably equal to or greater than 50.0. Thecore 4 having a hardness H(0) of 70.0 or less can achieve an outer-hard/inner-soft structure. In thegolf ball 2 that includes thecore 4, spin can be suppressed. In this respect, the hardness H(0) is more preferably equal to or less than 68.0 and particularly preferably equal to or less than 66.0. - The hardness Hs at the surface of the
core 4 is preferably equal to or greater than 78.0 but equal to or less than 95.0. In thecore 4 having a hardness Hs of 78.0 or greater, an outer-hard/inner-soft structure can be achieved. In thegolf ball 2 that includes thecore 4, spin can be suppressed. In this respect, the hardness Hs is more preferably equal to or greater than 80.0 and particularly preferably equal to or greater than 82.0. Thegolf ball 2 having a hardness Hs of 95.0 or less has excellent durability. In this respect, the hardness Hs is more preferably equal to or less than 93.0 and particularly preferably equal to or less than 90.0. - The
core 4 preferably has a diameter of 38.0 mm or greater but 42.0 mm or less. Thecore 4 having a diameter of 38.0 mm or greater can achieve excellent resilience performance of thegolf ball 2. In this respect, the diameter is more preferably equal to or greater than 38.5 mm and particularly preferably equal to or greater than 39.0 mm. In thegolf ball 2 that includes thecore 4 having a diameter of 42.0 mm or less, the inner cover 8 and theouter cover 10 can have sufficient thicknesses. Thegolf ball 2 that includes the inner cover 8 and theouter cover 10 which have large thicknesses has excellent durability. In this respect, the diameter is more preferably equal to or less than 41.0 mm and particularly preferably equal to or less than 40.0 mm. - In light of feel at impact, the
core 4 has an amount of compressive deformation Dc of preferably 3.0 mm or greater and particularly preferably 3.3 mm or greater. In light of resilience performance, the amount of compressive deformation Dc is preferably equal to or less than 4.6 mm and particularly preferably equal to or less than 4.3 mm. - For the inner cover 8, a resin composition is suitably used. Examples of the base polymer of the resin composition include ionomer resins, styrene block-containing thermoplastic elastomers, thermoplastic polyester elastomers, thermoplastic polyamide elastomers, and thermoplastic polyolefin elastomers.
- Particularly preferable base polymers are ionomer resins. The
golf ball 2 that includes the inner cover 8 including an ionomer resin has excellent resilience performance. An ionomer resin and another resin may be used in combination for the inner cover 8. In this case, the principal component of the base polymer is preferably the ionomer resin. Specifically, the proportion of the ionomer resin to the entire base polymer is preferably equal to or greater than 50% by weight, more preferably equal to or greater than 60% by weight, and particularly preferably equal to or greater than 70% by weight. - Examples of preferable ionomer resins include binary copolymers formed with an α-olefin and an α,β-unsaturated carboxylic acid having 3 to 8 carbon atoms. A preferable binary copolymer includes 80% by weight or greater but 90% by weight or less of an α-olefin, and 10% by weight or greater but 20% by weight or less of an α,β-unsaturated carboxylic acid. The binary copolymer has excellent resilience performance. Examples of other preferable ionomer resins include ternary copolymers formed with: an α-olefin; an α,β-unsaturated carboxylic acid having 3 to 8 carbon atoms; and an α,β-unsaturated carboxylate ester having 2 to 22 carbon atoms. A preferable ternary copolymer includes 70% by weight or greater but 85% by weight or less of an α-olefin, 5% by weight or greater but 30% by weight or less of an α,β-unsaturated carboxylic acid, and 1% by weight or greater but 25% by weight or less of an α,β-unsaturated carboxylate ester. The ternary copolymer has excellent resilience performance. For the binary copolymers and the ternary copolymers, preferable α-olefins are ethylene and propylene, while preferable α,β-unsaturated carboxylic acids are acrylic acid and methacrylic acid. A particularly preferable ionomer resin is a copolymer formed with ethylene and acrylic acid or methacrylic acid.
- In the binary copolymers and the ternary copolymers, some of the carboxyl groups are neutralized with metal ions. Examples of metal ions for use in neutralization include sodium ion, potassium ion, lithium ion, zinc ion, calcium ion, magnesium ion, aluminum ion, and neodymium ion. The neutralization may be carried out with two or more types of metal ions. Particularly suitable metal ions in light of resilience performance and durability of the
golf ball 2 are sodium ion, zinc ion, lithium ion, and magnesium ion. - Specific examples of ionomer resins include trade names "Himilan 1555", "Himilan 1557", "Himilan 1605", "Himilan 1706", "Himilan 1707", "Himilan 1856", "Himilan 1855", "Himilan AM7311", "Himilan AM7315", "Himilan AM7317","Himilan AM7318", "Himilan AM7329", "Himilan AM7337", "Himilan MK7320", and "Himilan MK7329", manufactured by Du Pont-MITSUI POLYCHEMICALS Co., Ltd.; trade names "Surlyn 6120", "Surlyn 6910", "Surlyn 7930", "Surlyn 7940", "Surlyn 8140", "Surlyn 8150", "Surlyn 8940", "Surlyn 8945", "Surlyn 9120", "Surlyn 9150", "Surlyn 9910", "Surlyn 9945", "Surlyn AD8546", "HPF1000", and "HPF2000", manufactured by E. I. du Pont de Nemours and Company; and trade names "IOTEK 7010", "IOTEK 7030", "IOTEK 7510", "IOTEK 7520", "IOTEK 8000", and "IOTEK 8030", manufactured by ExxonMobil Chemical Company.
- Two or more ionomer resins may be used in combination for the inner cover 8. An ionomer resin neutralized with a monovalent metal ion, and an ionomer resin neutralized with a bivalent metal ion may be used in combination.
- A preferable resin that can be used in combination with an ionomer resin is a styrene block-containing thermoplastic elastomer. The styrene block-containing thermoplastic elastomer has excellent compatibility with ionomer resins. A resin composition including the styrene block-containing thermoplastic elastomer has excellent fluidity.
- The styrene block-containing thermoplastic elastomer includes a polystyrene block as a hard segment, and a soft segment. A typical soft segment is a diene block. Examples of compounds for the diene block include butadiene, isoprene, 1, 3-pentadiene, and 2,3-dimethyl-1,3-butadiene. Butadiene and isoprene are preferred. Two or more compounds may be used in combination.
- Examples of styrene block-containing thermoplastic elastomers include styrene-butadiene-styrene block copolymers (SBS), styrene-isoprene-styrene block copolymers (SIS), styrene-isoprene-butadiene-styrene block copolymers (SIBS), hydrogenated SBS, hydrogenated SIS, and hydrogenated SIBS. Examples of hydrogenated SBS include styrene-ethylene-butylene-styrene block copolymers (SEBS). Examples of hydrogenated SIS include styrene-ethylene-propylene-styrene block copolymers (SEPS). Examples of hydrogenated SIBS include styrene-ethylene-ethylene-propylene-styrene block copolymers (SEEPS).
- In light of resilience performance of the
golf ball 2, the content of the styrene component in the styrene block-containing thermoplastic elastomer is preferably equal to or greater than 10% by weight, more preferably equal to or greater than 12% by weight, and particularly preferably equal to or greater than 15% by weight. In light of feel at impact of thegolf ball 2, the content is preferably equal to or less than 50% by weight, more preferably equal to or less than 47% by weight, and particularly preferably equal to or less than 45% by weight. - In the present invention, styrene block-containing thermoplastic elastomers include an alloy of an olefin and one or more members selected from the group consisting of SBS, SIS, SIBS, SEBS, SEPS, SEEPS, and hydrogenated products thereof. The olefin component in the alloy is presumed to contribute to improvement of compatibility with ionomer resins. Use of this alloy improves the resilience performance of the
golf ball 2. An olefin having 2 to 10 carbon atoms is preferably used. Examples of suitable olefins include ethylene, propylene, butene, and pentene. Ethylene and propylene are particularly preferred. - Specific examples of polymer alloys include trade names "Rabalon T3221C", "Rabalon T3339C", "Rabalon SJ4400N", "Rabalon SJ5400N", "Rabalon SJ6400N", "Rabalon SJ7400N", "Rabalon SJ8400N", "Rabalon SJ9400N", and "Rabalon SR04", manufactured by Mitsubishi Chemical Corporation. Other specific examples of styrene block-containing thermoplastic elastomers include trade name "Epofriend A1010" manufactured by Daicel Chemical Industries, Ltd., and trade name "Septon HG-252" manufactured by Kuraray Co., Ltd.
- According to need, a coloring agent such as titanium dioxide and a fluorescent pigment, a filler such as barium sulfate, a dispersant, an antioxidant, an ultraviolet absorber, a light stabilizer, a fluorescent material, a fluorescent brightener, and the like are included in the resin composition of the inner cover 8 in an adequate amount. The inner cover 8 may include powder of a metal with a high specific gravity.
- From the standpoint that an outer-hard/inner-soft structure can be achieved in a
sphere 16 consisting of thecore 4 and the inner cover 8, the inner cover 8 has a hardness Hi of preferably 80 or greater, more preferably 83 or greater, and particularlypreferably 85 or greater. In light of feel at impact of thegolf ball 2, the hardness Hi is preferably equal to or less than 95 and particularly preferably equal to or less than 90. The hardness Hi is measured with a JIS-C type hardness scale mounted to an automated rubber hardness measurement machine (trade name "P1", manufactured by Kobunshi Keiki Co., Ltd.). For the measurement, a slab that is formed by hot press and that has a thickness of about 2 mm is used. A slab kept at 23°C for two weeks is used for the measurement. At the measurement, three slabs are stacked. A slab formed from the same resin composition as the resin composition of the inner cover 8 is used. - From the standpoint that an outer-hard/inner-soft structure is achieved in the
sphere 16 and spin of thegolf ball 2 is suppressed, the hardness Hi of the inner cover 8 is preferably greater than the surface hardness Hs of thecore 4. In light of suppression of spin, the difference (Hi-Hs) between the hardness Hi and the hardness Hs is preferably equal to or greater than 1 and particularly preferably equal to or greater than 2. The difference (Hi-Hs) is preferably equal to or less than 5. In thesphere 16 in which the difference (Hi-Hs) is equal to or less than 5, the hardness linearly increases from its central point toward its surface. In thesphere 16 whose hardness linearly increases, the energy loss is low when thegolf ball 2 is hit with a middle iron. - The inner cover 8 preferably has a thickness of 0.2 mm or greater but 2.0 mm or less. In the
sphere 16 that includes the inner cover 8 having a thickness of 0.2 mm or greater, an outer-hard/inner-soft structure can be achieved. In this respect, the thickness is more preferably equal to or greater than 0.5 mm and particularly preferably equal to or greater than 0.8 mm. Thegolf ball 2 that includes the inner cover 8 having a thickness of 2.0 mm or less has excellent resilience performance. In this respect, the thickness is more preferably equal to or less than 1.6 mm and particularly preferably equal to or less than 1.3 mm. - In light of feel at impact, the
sphere 16 consisting of thecore 4 and the inner cover 8 has an amount of compressive deformation Di of preferably 3.2 mm or greater and particularly preferably 3.4 mm or greater. In light of resilience performance, the amount of compressive deformation Di is preferably equal to or less than 3.8 mm and particularly preferably equal to or less than 3.6 mm. - For forming the inner cover 8, known methods such as injection molding, compression molding, and the like can be used.
- For the
outer cover 10, a resin composition is suitably used. A preferable base polymer of the resin composition is an ionomer resin. Thegolf ball 2 that includes theouter cover 10 including the ionomer resin has excellent resilience performance. The ionomer resin described above for the inner cover 8 can be used for theouter cover 10. - An ionomer resin and another resin may be used in combination. In this case, in light of resilience performance, the ionomer resin is included as the principal component of the base polymer. The proportion of the ionomer resin to the entire base polymer is preferably equal to or greater than 50% by weight, more preferably equal to or greater than 60% by weight, and particularly preferably equal to or greater than 70% by weight.
- A preferable resin that can be used in combination with an ionomer resin is an ethylene-(meth)acrylic acid copolymer. The copolymer is obtained by a copolymerization reaction of a monomer composition that contains ethylene and (meth)acrylic acid. In the copolymer, some of the carboxyl groups are neutralized with metal ions. The copolymer includes 3% by weight or greater but 25% by weight or less of a (meth)acrylic acid component. An ethylene-(meth)acrylic acid copolymer having a polar functional group is particularly preferred. A specific example of ethylene- (meth) acrylic acid copolymers is trade name "NUCREL" manufactured by Du Pont-MITSUI POLYCHEMICALS Co., Ltd.
- Another preferable resin that can be used in combination with an ionomer resin is a styrene block-containing thermoplastic elastomer. The styrene block-containing thermoplastic elastomer described above for the inner cover 8 can be used for the
outer cover 10. - According to need, a coloring agent such as titanium dioxide and a fluorescent pigment, a filler such as barium sulfate, a dispersant, an antioxidant, an ultraviolet absorber, a light stabilizer, a fluorescent material, a fluorescent brightener, and the like are included in the
outer cover 10 in an adequate amount. - The
outer cover 10 preferably has a JIS-C hardness Ho of 83 or greater but 96 or less. In thegolf ball 2 that includes theouter cover 10 having a hardness Ho of 83 or greater, an outer-hard/inner-soft structure can be achieved. In thegolf ball 2 that has the outer-hard/inner-soft structure, spin is suppressed. Thegolf ball 2 has excellent flight performance. In this respect, the hardness Ho is more preferably equal to or greater than 84 and particularly preferably equal to or greater than 85. Thegolf ball 2 that includes theouter cover 10 having a hardness Ho of 96 or less has excellent feel at impact. In this respect, the hardness Ho is more preferably equal to or less than 95 and particularly preferably equal to or less than 93. The hardness Ho is measured by the same measurement method as that for the hardness Hi. - The
outer cover 10 preferably has a thickness of 0.2 mm or greater but 1.5 mm or less. Theouter cover 10 having a thickness of 0.2 mm or greater can easily be formed. In this respect, the thickness is more preferably equal to or greater than 0.4 mm and particularly preferably equal to or greater than 0.6 mm. In thegolf ball 2 that includes theouter cover 10 having a thickness of 1.5 mm or less, spin is suppressed. In this respect, the thickness is more preferably equal to or less than 1.3 mm and particularly preferably equal to or less than 1.1 mm. - For forming the
outer cover 10, known methods such as injection molding, compression molding, and the like can be used. When forming theouter cover 10, thedimples 12 are formed by pimples formed on the cavity face of a mold. - The
cover 6 preferably has a total thickness of 2.5 mm or less. Thegolf ball 2 that includes thecover 6 having a total thickness of 2.5 mm or less has excellent feel at impact. In this respect, the total thickness is more preferably equal to or less than 2.3 mm and particularly preferably equal to or less than 2.1 mm. In light of durability of thegolf ball 2, the total thickness is preferably equal to or greater than 0.3 mm, more preferably equal to or greater than 0.5 mm, and particularly preferably equal to or greater than 0.8 mm. - The JIS-C hardness Ho of the
outer cover 10 is greater than the JIS-C hardness Hi of the inner cover 8. Theouter cover 10 can achieve an outer-hard/inner-soft structure of thegolf ball 2. Thegolf ball 2 has excellent flight performance and excellent feel at impact. The difference (Ho-Hi) is preferably equal to or greater than 2, more preferably equal to or greater than 4, and particularly preferably equal to or greater than 6. In light of suppression of energy loss when thegolf ball 2 is hit, the difference (Ho-Hi) is preferably equal to or less than 10. - In a hardness distribution curve of the
golf ball 2 from the central point of thecore 4 to theouter cover 10, the hardness of theouter cover 10 is the greatest. In thegolf ball 2, spin is suppressed. - In light of feel at impact, the
golf ball 2 has an amount of compressive deformation Db of preferably 2.8 mm or greater, more preferably 2.9 mm or greater, and particularly preferably 3.0 mm or greater. In light of resilience performance, the amount of compressive deformation Db is preferably equal to or less than 3.6 mm, more preferably equal to or less than 3.5 mm, and particularly preferably equal to or less than 3.4 mm. - At measurement of the amount of compressive deformation, first, a sphere such as the
core 4, thegolf ball 2, or the like is placed on a hard plate made of metal. Next, a cylinder made of metal gradually descends toward the sphere. The sphere, squeezed between the bottom face of the cylinder and the hard plate, becomes deformed. A migration distance of the cylinder, starting from the state in which an initial load of 98 N is applied to the sphere up to the state in which a final load of 1274 N is applied thereto, is measured. - A
golf ball 102 shown inFIG. 3 includes aspherical core 104 and acover 106 covering thecore 104. Thecover 106 includes aninner cover 108 and anouter cover 110 positioned outside theinner cover 108. Theinner cover 108 is an innermost layer of thecover 106. Theouter cover 110 is an outermost layer of thecover 106. Thecover 106 may include another one or more layers between theinner cover 108 and theouter cover 110. On the surface of theouter cover 110, a large number ofdimples 112 are formed. Of the surface of thegolf ball 102, a part other than thedimples 112 is aland 114. Thegolf ball 102 includes a paint layer and a mark layer on the external side of theouter cover 110, but these layers are not shown in the drawing. - The
golf ball 102 has a diameter of 40 mm or greater but 45 mm or less. From the standpoint of conformity to the rules established by the United States Golf Association (USGA), the diameter is preferably equal to or greater than 42.67 mm. In light of suppression of air resistance, the diameter is preferably equal to or less than 44 mm and more preferably equal to or less than 42.80 mm. Thegolf ball 102 has a weight of 40 g or greater but 50 g or less. In light of attainment of great inertia, the weight is preferably equal to or greater than 44 g and more preferably equal to or greater than 45.00 g. From the standpoint of conformity to the rules established by the USGA, the weight is preferably equal to or less than 45.93 g. -
FIG. 4 is a line graph showing a hardness distribution of thecore 104 of thegolf ball 102 inFIG. 3 . The horizontal axis of the graph indicates the ratio (%) of a distance from the central point of the core 104 to the radius of thecore 104. The vertical axis of the graph indicates a JIS-C hardness. Nine measuring points obtained by dividing a region from the central point of the core 104 to the surface of the core 104 at intervals of 12.5% of the radius of thecore 104 are plotted in the graph. The ratio of the distance from the central point of the core 104 to each of these measuring points to the radius of thecore 104 is as follows. - First point: 0.0% (central point)
- Second point: 12.5%
- Third point: 25.0%
- Fourth point: 37.5%
- Fifth point: 50.0%
- Sixth point: 62.5%
- Seventh point: 75.0%
- Eighth point: 87.5%
- Ninth point: 100.0% (surface)
-
FIG. 4 also shows a linear approximation curve obtained by a least-square method on the basis of the distances and the hardnesses of the nine measuring points. As is clear fromFIG. 4 , the broken line does not greatly deviate from the linear approximation curve. In other words, the broken line has a shape close to the linear approximation curve. In thecore 104, the hardness linearly increases from its central point toward its surface. When thecore 104 is hit with a driver, the energy loss is low. Thecore 104 has excellent resilience performance.Inthegolfball 102 that includes thecore 104, spin is suppressed. When thegolf ball 102 is hit with a driver, the flight distance is large. - In the
core 104, R2 of the linear approximation curve obtained by the least-square method is equal to or greater than 0.95. R2 is an index indicating the linearity of the broken line. For thecore 104 for which R2 is equal to or greater than 0.95, the shape of the broken line of the hardness distribution is close to a straight line. Thecore 104 for which R2 is equal to or greater than 0.95 has excellent resilience performance. R2 is more preferably equal to or greater than 0.96 and particularly preferably equal to or greater than 0.97. R2 is calculated by squaring a correlation coefficient R. The correlation coefficient R is calculated by dividing the covariance of the distance (%) from the central point and the hardness (JIS-C) by the standard deviation of the distance (%) from the central point and the standard deviation of the hardness (JIS-C). - The
core 104 is obtained by crosslinking a rubber composition. The rubber composition includes: - (a) a base rubber;
- (b) a co-crosslinking agent;
- (c) a crosslinking initiator; and
- (d) an acid and/or a salt.
- During heating and forming of the
core 104, the base rubber (a) is crosslinked by the co-crosslinking agent (b). The heat of the crosslinking reaction remains near the central point of thecore 104. Thus, during heating and forming of thecore 104, the temperature at the central portion is high. The temperature gradually decreases from the central point toward the surface. It is inferred that in the rubber composition, the acid reacts with a metal salt of the co-crosslinking agent (b) to bond to cation. It is inferred that in the rubber composition, the salt reacts with the metal salt of the co-crosslinking agent (b) to exchange cation. By the bonding and the exchange, metallic bonding is broken. This breaking is likely to occur in the central portion of thecore 104 where the temperature is high, and is unlikely to occur near the surface of thecore 104. As a result, the crosslinking density of the core 104 increases from its central point toward its surface. In thecore 104, an outer-hard/inner-soft structure can be achieved. Furthermore, when the rubber composition includes an organic sulfur compound (e) together with the acid and/or the salt (d), the gradient of the hardness distribution can be controlled, and the degree of the outer-hard/inner-soft structure of the core 104 can be increased. When thegolf ball 102 that includes thecore 104 is hit with a driver, the spin rate is low. In thegolf ball 102, excellent flight performance is achieved upon a shot with a driver. - The rubber composition of the core 104 can include, as the base rubber (a), the base rubber (a) described above for the rubber composition of the
core 4 according to the first embodiment. - The rubber composition of the core 104 can include, as the co-crosslinking agent (b), the co-crosslinking agent (b) described above for the rubber composition of the
core 4 according to the first embodiment. The co-crosslinking agent (b) is: - (b1) an α,β-unsaturated carboxylic acid having 3 to 8 carbon atoms; or
- (b2) a metal salt of an α,β-unsaturated carboxylic acid having 3 to 8 carbon atoms.
- The rubber composition may include only the α,β-unsaturated carboxylic acid (b1) or only the metal salt (b2) of the α,β-unsaturated carboxylic acid as the co-crosslinking agent (b). The rubber composition may include both the α,β-unsaturated carboxylic acid (b1) and the metal salt (b2) of the α,β-unsaturated carboxylic acid as the co-crosslinking agent (b).
- The metal salt (b2) of the α,β-unsaturated carboxylic acid graft-polymerizes with the molecular chain of the base rubber, thereby crosslinking the rubber molecules. When the rubber composition includes the α,β-unsaturated carboxylic acid (b1), the rubber composition preferably further includes a metal compound (f). The metal compound (f) reacts with the α,β-unsaturated carboxylic acid (b1) in the rubber composition. A salt obtained by this reaction graft-polymerizes with the molecular chain of the base rubber.
- The rubber composition of the core 104 can include, as the metal compound (f), the metal compound (f) described above for the rubber composition of the
core 4 according to the first embodiment. - In light of resilience performance of the
golf ball 102, the amount of the co-crosslinking agent (b) is preferably equal to or greater than 15 parts by weight and particularly preferably equal to or greater than 20 parts by weight, per 100 parts by weight of the base rubber. In light of feel at impact, the amount is preferably equal to or less than 50 parts by weight, more preferably equal to or less than 45 parts by weight, and particularly preferably equal to or less than 40 parts by weight, per 100 parts by weight of the base rubber. - The rubber composition of the core 104 can include, as the crosslinking initiator (c), the crosslinking initiator (c) described above for the rubber composition of the
core 4 according to the first embodiment. - In light of resilience performance of the
golf ball 102, the amount of the crosslinking initiator (c) is preferably equal to or greater than 0.2 parts by weight and particularly preferably equal to or greater than 0.5 parts by weight, per 100 parts by weight of the base rubber. In light of feel at impact and durability of thegolf ball 102, the amount is preferably equal to or less than 5. 0 parts by weight and particularly preferably equal to or less than 2.5 parts by weight, per 100 parts by weight of the base rubber. - The rubber composition of the
core 104 includes, as the acid and/or the salt (d) the carboxylic acid and/or the salt (d1) described above for the rubber composition of thecore 4 according to the first embodiment. In the present invention, the co-crosslinking agent (b) is not included in the concept of the acid and/or the salt (d). It is inferred that as described above, the acid and/or the salt (d) breaks the metal crosslinks by the co-crosslinking agent (b) in the central portion of the core 104 during heating and forming of thecore 104. Carboxylates are particularly preferred. - The carboxylic acid component of the carboxylic acid and/or the salt thereof (d1) has a carboxyl group. The carbon number of the carboxylic acid component of the carboxylic acid and/or the salt thereof (d1) is preferably equal to or greater than 1 but equal to or less than 30, more preferably equal to or greater than 3 but equal to or less than 30, and even more preferably equal to or greater than 5 but equal to or less than 28. Examples of the carboxylic acid include aliphatic carboxylic acids (fatty acids) and aromatic carboxylic acids. Fatty acids and salts thereof are preferred.
- The rubber composition may include a saturated fatty acid or a salt thereof, or may include an unsaturated fatty acid or a salt thereof. The saturated fatty acid and the salt thereof are preferred.
- Examples of preferable carboxylates include a potassium salt, a magnesium salt, an aluminum salt, a zinc salt, an iron salt, a copper salt, a nickel salt, or a cobalt salt of octanoic acid, lauric acid, myristic acid, palmitic acid, stearic acid, oleic acid, or behenic acid. Zinc salts of carboxylic acids are particularly preferred. Specific examples of preferable carboxylates include zinc octoate, zinc laurate, zinc myristate, and zinc stearate.
- In light of linearity of the hardness distribution of the
core 104, the amount of the acid and/or the salt (d) is preferably equal to or greater than 0.5 parts by weight, more preferably equal to or greater than 1.0 parts by weight, even more preferably equal to or greater than 1.5 parts by weight, and particularly preferably equal to or greater than 2.0 parts by weight, per 100 parts by weight of the base rubber. In light of resilience performance, the amount is preferably equal to or less than 40 parts by weight, more preferably less than 40 parts by weight, even more preferably equal to or less than 30 parts by weight, and particularly preferably equal to or less than 20 parts by weight, per 100 parts by weight of the base rubber. - The weight ratio of the co-crosslinking agent (b) and the acid and/or the salt (d) in the rubber composition is preferably equal to or greater than 3/7 but equal to or less than 9/1, and is particularly preferably equal to or greater than 4/6 but equal to or less than 8/2. From the rubber composition in which this weight ratio is within the above range, the
core 104 whose hardness linearly increases from its central point toward its surface can be obtained. - As the co-crosslinking agent (b), zinc acrylate is preferably used. Zinc acrylate whose surface is coated with stearic acid or zinc stearate for the purpose of improving dispersibility to rubber is present. In the present invention, when the rubber composition includes this zinc acrylate, this coating material is not included in the concept of the acid and/or the salt (d).
- The rubber composition preferably further includes an organic sulfur compound (e). The organic sulfur compound (e) can contribute to control of: the linearity of the hardness distribution of the
core 104; and the degree of the outer-hard/inner-soft structure. An example of the organic sulfur compound (e) is an organic compound having a thiol group or a polysulfide linkage having 2 to 4 sulfur atoms. A metal salt of this organic compound is also included in the organic sulfur compound (e). The rubber composition of the core 104 can include, as the organic sulfur compound (e), the organic sulfur compound (e) described above for the rubber composition of thecore 4 according to the first embodiment. - In light of resilience performance, the amount of the organic sulfur compound (e) is preferably equal to or greater than 0.05 parts by weight and particularly preferably equal to or greater than 0.1 parts by weight, per 100 parts by weight of the base rubber. In light of resilience performance, the amount is preferably equal to or less than 5.0 parts by weight and particularly preferably equal to or less than 2.0 parts by weight, per 100 parts by weight of the base rubber.
- For the purpose of adjusting specific gravity and the like, a filler may be included in the
core 104. Examples of suitable fillers include zinc oxide, barium sulfate, calcium carbonate, and magnesium carbonate. The amount of the filler is determined as appropriate so that the intended specific gravity of thecore 104 is accomplished. A particularly preferable filler is zinc oxide. Zinc oxide serves not only as a specific gravity adjuster but also as a crosslinking activator. - According to need, an anti-aging agent, a coloring agent, a plasticizer, a dispersant, sulfur, a vulcanization accelerator, and the like are added to the rubber composition of the
core 104. Crosslinked rubber powder or synthetic resin powder may also be dispersed in the rubber composition. - In the
core 104, the difference (Hs-H(0)) between the surface hardness Hs and the central hardness H (0) is preferably equal to or greater than 15. The difference is great. In other words, thecore 104 has an outer-hard/inner-soft structure. When thecore 104 is hit with a driver, the recoil (torsional return) is great, and thus spin is suppressed. Thecore 104 contributes to the flight performance of thegolf ball 102. In light of flight performance, the difference (Hs-H(0)) is more preferably equal to or greater than 20 and particularly preferably equal to or greater than 25. From the standpoint that thecore 104 can easily be formed, the difference (Hs-H(0)) is preferably equal to or less than 50. - The hardness H(0) at the central point of the
core 104 is preferably equal to or greater than 40.0 but equal to or less than 70.0. Thegolf ball 102 having a hardness H(0) of 40.0 or greater has excellent resilience performance. In this respect, the hardness H (0) is more preferably equal to or greater than 45.0 and particularly preferably equal to or greater than 50.0. Thecore 104 having a hardness H(0) of 70.0 or less can achieve an outer-hard/inner-soft structure. In thegolf ball 102 that includes thecore 104, spin can be suppressed. In this respect, the hardness H(0) is more preferably equal to or less than 68.0 and particularly preferably equal to or less than 66.0. - The hardness Hs at the surface of the
core 104 is preferably equal to or greater than 78.0 but equal to or less than 95.0. In thecore 104 having a hardness Hs of 78.0 or greater, an outer-hard/inner-soft structure can be achieved. In thegolf ball 102 that includes thecore 104, spin can be suppressed. In this respect, the hardness Hs is more preferably equal to or greater than 80.0 and particularly preferably equal to or greater than 82.0. Thegolf ball 102 having a hardness Hs of 95.0 or less has excellent durability. In this respect, the hardness Hs is more preferably equal to or less than 93.0 and particularly preferably equal to or less than 90.0. - The
core 104 preferably has a diameter of 38.0 mm or greater but 42.0 mm or less. Thecore 104 having a diameter of 38.0 mm or greater can achieve excellent resilience performance of thegolf ball 102. In this respect, the diameter is more preferably equal to or greater than 38.5 mm and particularly preferably equal to or greater than 39.0 mm. In thegolf ball 102 that includes thecore 104 having a diameter of 42.0 mm or less, theinner cover 108 and theouter cover 110 can have sufficient thicknesses. Thegolf ball 102 that includes theinner cover 108 and theouter cover 110 which have large thicknesses has excellent durability. In this respect, the diameter is more preferably equal to or less than 41.0 mm and particularly preferably equal to or less than 40.0 mm. - In light of feel at impact, the
core 104 has an amount of compressive deformation Dc of preferably 3.0 mm or greater and particularly preferably 3.3 mm or greater. In light of resilience performance, the amount of compressive deformation Dc is preferably equal to or less than 4.6 mm and particularly preferably equal to or less than 4.3 mm. - For the
inner cover 108, a resin composition is suitably used. Examples of the base polymer of the resin composition include ionomer resins, styrene block-containing thermoplastic elastomers, thermoplastic polyester elastomers, thermoplastic polyamide elastomers, and thermoplastic polyolefin elastomers. - Particularly preferable base polymers are ionomer resins. The
golf ball 102 that includes theinner cover 108 including an ionomer resin has excellent resilience performance. An ionomer resin and another resin may be used in combination for theinner cover 108. In this case, the principal component of the base polymer is preferably the ionomer resin. Specifically, the proportion of the ionomer resin to the entire base polymer is preferably equal to or greater than 50% by weight, more preferably equal to or greater than 60% by weight, and particularly preferably equal to or greater than 70% by weight. - Examples of preferable ionomer resins include binary copolymers formed with an α-olefin and an α,β-unsaturated carboxylic acid having 3 to 8 carbon atoms. A preferable binary copolymer includes 80% by weight or greater but 90% by weight or less of an α-olefin, and 10% by weight or greater but 20% by weight or less of an α,β-unsaturated carboxylic acid. The binary copolymer has excellent resilience performance. Examples of other preferable ionomer resins include ternary copolymers formed with: an α-olefin; an α,β-unsaturated carboxylic acid having 3 to 8 carbon atoms; and an α,β-unsaturated carboxylate ester having 2 to 22 carbon atoms. Apreferable ternary copolymer includes 70% by weight or greater but 85% by weight or less of an α-olefin, 5% by weight or greater but 30% by weight or less of an α,β-unsaturated carboxylic acid, and 1% by weight or greater but 25% by weight or less of an α,β-unsaturated carboxylate ester. The ternary copolymer has excellent resilience performance. For the binary copolymers and the ternary copolymers, preferable α-olefins are ethylene and propylene, while preferable α,β-unsaturated carboxylic acids are acrylic acid and methacrylic acid. A particularly preferable ionomer resin is a copolymer formed with ethylene and acrylic acid or methacrylic acid.
- In the binary copolymers and the ternary copolymers, some of the carboxyl groups are neutralized with metal ions. Examples of metal ions for use in neutralization include sodium ion, potassium ion, lithium ion, zinc ion, calcium ion, magnesium ion, aluminum ion, and neodymium ion. The neutralization may be carried out with two or more types of metal ions. Particularly suitable metal ions in light of resilience performance and durability of the
golf ball 2 are sodium ion, zinc ion, lithium ion, and magnesium ion. - Specific examples of ionomer resins include trade names "Himilan 1555", "Himilan 1557", "Himilan 1605", "Himilan 1706", "Himilan 1707", "Himilan 1856", "Himilan 1855", "Himilan AM7311", "Himilan AM7315", "Himilan AM7317","Himilan AM7318", "Himilan AM7329", "Himilan AM7337", "Himilan MK7320", and "Himilan MK7329", manufactured by Du Pont-MITSUI POLYCHEMICALS Co., Ltd.; trade names "Surlyn 6120", "Surlyn 6910", "Surlyn 7930", "Surlyn 7940", "Surlyn 8140", "Surlyn 8150", "Surlyn 8940", "Surlyn 8945", "Surlyn 9120", "Surlyn 9150", "Surlyn 9910", "Surlyn 9945", "Surlyn AD8546", "HPF1000", and "HPF2000", manufactured by E. I. du Pont de Nemours and Company; and trade names "IOTEK 7010", "IOTEK 7030", "IOTEK 7510", "IOTEK 7520", "IOTEK 8000", and "IOTEK 8030", manufactured by ExxonMobil Chemical Company.
- Two or more ionomer resins may be used in combination for the
inner cover 108. An ionomer resin neutralized with a monovalent metal ion, and an ionomer resin neutralized with a bivalent metal ion may be used in combination. - A preferable resin that can be used in combination with an ionomer resin is a styrene block-containing thermoplastic elastomer. The styrene block-containing thermoplastic elastomer has excellent compatibility with ionomer resins. A resin composition including the styrene block-containing thermoplastic elastomer has excellent fluidity.
- The styrene block-containing thermoplastic elastomer includes a polystyrene block as a hard segment, and a soft segment. A typical soft segment is a diene block. Examples of compounds for the diene block include butadiene, isoprene, 1,3-pentadiene, and 2,3-dimethyl-1,3-butadiene. Butadiene and isoprene are preferred. Two or more compounds may be used in combination.
- Examples of styrene block-containing thermoplastic elastomers include styrene-butadiene-styrene block copolymers (SBS), styrene-isoprene-styrene block copolymers (SIS), styrene-isoprene-butadiene-styrene block copolymers (SIBS), hydrogenated SBS, hydrogenated SIS, and hydrogenated SIBS. Examples of hydrogenated SBS include styrene-ethylene-butylene-styrene block copolymers (SEBS). Examples of hydrogenated SIS include styrene-ethylene-propylene-styrene block copolymers (SEPS). Examples of hydrogenated SIBS include styrene-ethylene-ethylene-propylene-styrene block copolymers (SEEPS).
- In light of resilience performance of the
golf ball 102, the content of the styrene component in the styrene block-containing thermoplastic elastomer is preferably equal to or greater than 10% by weight, more preferably equal to or greater than 12% by weight, and particularly preferably equal to or greater than 15% by weight. In light of feel at impact of thegolf ball 102, the content is preferably equal to or less than 50% by weight, more preferably equal to or less than 47% by weight, and particularly preferably equal to or less than 45% by weight. - In the present invention, styrene block-containing thermoplastic elastomers include an alloy of an olefin and one or more members selected from the group consisting of SBS, SIS, SIBS, SEBS, SEPS, SEEPS, and hydrogenated products thereof. The olefin component in the alloy is presumed to contribute to improvement of compatibility with ionomer resins. Use of this alloy improves the resilience performance of the
golf ball 102. An olefin having 2 to 10 carbon atoms is preferably used. Examples of suitable olefins include ethylene, propylene, butene, and pentene. Ethylene and propylene are particularly preferred. - Specific examples of polymer alloys include trade names "Rabalon T3221C", "Rabalon T3339C", "Rabalon SJ4400N", "Rabalon SJ5400N", "Rabalon SJ6400N", "Rabalon SJ7400N", "Rabalon SJ8400N", "Rabalon SJ9400N", and "Rabalon SR04", manufactured by Mitsubishi Chemical Corporation. Other specific examples of styrene block-containing thermoplastic elastomers include trade name "Epofriend A1010" manufactured by Daicel Chemical Industries, Ltd., and trade name "Septon HG-252" manufactured by Kuraray Co., Ltd.
- According to need, a coloring agent such as titanium dioxide and a fluorescent pigment, a filler such as barium sulfate, a dispersant, an antioxidant, an ultraviolet absorber, a light stabilizer, a fluorescent material, a fluorescent brightener, and the like are included in the resin composition of the
inner cover 108 in an adequate amount. Theinner cover 108 may include powder of a metal with a high specific gravity. - In the
golf ball 102, theinner cover 108 has a hardness Hi which is equal to or less than the JIS-C hardness Hs at the surface of thecore 104. When thegolf ball 102 is hit with a driver, theinner cover 108 achieves soft feel at impact. In light of feel at impact, the difference (Hi-Hs) is preferably equal to or greater than 1, more preferably equal to or greater than 2, and particularly preferably equal to or greater than 7. When the difference (Hi-Hs) is not excessively great, light feel at impact is obtained. In this respect, the difference (Hi-Hs) is preferably equal to or less than 20, more preferably equal to or less than 18, and particularly preferably equal to or less than 12. - In light of soft feel at impact, the hardness Hi of the
inner cover 108 is preferably equal to or less than 90, more preferably equal to or less than 85, and particularly preferably equal to or less than 83. In light of light feel at impact, the hardness Hi is preferably equal to or greater than 60, more preferably equal to or greater than 65, and particularly preferably equal to or greater than 71. - The hardness Hi is measured with a JIS-C type hardness scale mounted to an automated rubber hardness measurement machine (trade name "P1", manufactured by Kobunshi Keiki Co., Ltd.). For the measurement, a slab that is formed by hot press and that has a thickness of about 2 mm is used. A slab kept at 23°C for two weeks is used for the measurement. At the measurement, three slabs are stacked. A slab formed fromthe same resin composition as the resin composition of the
inner cover 108 is used. Theinner cover 108 preferably has a thickness of 0.2 mm or greater but 2.0 mm or less. Thegolf ball 102 that includes theinner cover 108 having a thickness of 0.2 mm or greater has excellent feel at impact. In this respect, the thickness of theinner cover 108 is more preferably equal to or greater than 0.5 mm and particularly preferably equal to or greater than 0. 8 mm. Thegolf ball 102 that includes theinner cover 108 having a thickness of 2.0 mm or less has excellent resilience performance. In this respect, the thickness is more preferably equal to or less than 1.5 mm and particularly preferably equal to or less than 1.2 mm. - In light of feel at impact, a
sphere 116 consisting of thecore 104 and theinner cover 108 has an amount of compressive deformation Di of preferably 3.2 mm or greater and particularly preferably 3.4 mm or greater. In light of resilience performance, the amount of compressive deformation Di is preferably equal to or less than 3.8 mm and particularly preferably equal to or less than 3.6 mm. - For forming the
inner cover 108, known methods such as injection molding, compression molding, and the like can be used. - For the
outer cover 110, a resin composition is suitably used. A preferable base polymer of the resin composition is an ionomer resin. Thegolf ball 102 that includes theouter cover 110 including the ionomer resin has excellent resilience performance. The ionomer resin described above for theinner cover 108 can be used for theouter cover 110. - An ionomer resin and another resin may be used in combination. In this case, in light of resilience performance, the ionomer resin is included as the principal component of the base polymer. The proportion of the ionomer resin to the entire base polymer is preferably equal to or greater than 50% by weight, more preferably equal to or greater than 60% by weight, and particularly preferably equal to or greater than 70% by weight.
- A preferable resin that can be used in combination with an ionomer resin is an ethylene-(meth)acrylic acid copolymer. The copolymer is obtained by a copolymerization reaction of a monomer composition that contains ethylene and (meth)acrylic acid. In the copolymer, some of the carboxyl groups are neutralized with metal ions. The copolymer includes 3% by weight or greater but 25% by weight or less of a (meth)acrylic acid component. An ethylene-(meth)acrylic acid copolymer having a polar functional group is particularly preferred. A specific example of ethylene- (meth) acrylic acid copolymers is trade name "NUCREL" manufactured by Du Pont-MITSUI POLYCHEMICALS Co., Ltd.
- According to need, a coloring agent such as titanium dioxide and a fluorescent pigment, a filler such as barium sulfate, a dispersant, an antioxidant, an ultraviolet absorber, a light stabilizer, a fluorescent material, a fluorescent brightener, and the like are included in the
outer cover 110 in an adequate amount. - The
outer cover 110 preferably has a JIS-C hardness Ho of 83 or greater but 96 or less. In thegolf ball 102 that includes theouter cover 110 having a hardness Ho of 83 or greater, an outer-hard/inner-soft structure can be achieved. In thegolf ball 102 that has the outer-hard/inner-soft structure, spin is suppressed. Thegolf ball 102 has excellent flight performance. In this respect, the hardness Ho is more preferably equal to or greater than 84 and particularly preferably equal to or greater than 85. Thegolf ball 102 that includes theouter cover 110 having a hardness Ho of 96 or less has excellent feel at impact. In this respect, the hardness Ho is more preferably equal to or less than 95 and particularly preferably equal to or less than 93. The hardness Ho is measured by the same measurement method as that for the hardness Hi. - The
outer cover 110 preferably has a thickness of 0.2 mm or greater but 1.5 mm or less. Theouter cover 110 having a thickness of 0.2 mm or greater can easily be formed. In this respect, the thickness is more preferably equal to or greater than 0.4 mm and particularly preferably equal to or greater than 0.6 mm. Thegolf ball 102 that includes theouter cover 110 having a thickness of 1. 5 mm or less has excellent feel at impact. In this respect, the thickness is more preferably equal to or less than 1.3 mm and particularly preferably equal to or less than 1.1 mm. - For forming the
outer cover 110, known methods such as injection molding, compression molding, and the like can be used. When forming theouter cover 110, thedimples 112 are formed by pimples formed on the cavity face of a mold. - The
cover 106 preferably has a total thickness of 2.5 mm or less. Thegolf ball 102 that includes thecover 106 having a total thickness of 2.5 mm or less has excellent feel at impact. In this respect, the total thickness is more preferably equal to or less than 2.3 mm and particularly preferably equal to or less than 2.1 mm. In light of durability of thegolf ball 102, the total thickness is preferably equal to or greater than 0.3 mm, more preferably equal to or greater than 0.5 mm, and particularly preferably equal to or greater than 0.8 mm. - The JIS-C hardness Ho of the
outer cover 110 is greater than the JIS-C hardness Hi of theinner cover 108. Theouter cover 110 can achieve an outer-hard/inner-soft structure of thegolf ball 102 . Thegolf ball 102 has excellent flight performance and excellent feel at impact. The difference (Ho-Hi) is preferably equal to or greater than 5, more preferably equal to or greater than 9, and particularly preferably equal to or greater than 16. In light of suppression of energy loss when thegolf ball 102 is hit, the difference (Ho-Hi) is preferably equal to or less than 27 and particularly preferably equal to or less than 21. - In a hardness distribution curve of the
golf ball 102 from the central point of the core 104 to theouter cover 110, the hardness of theouter cover 110 is the greatest. In thegolf ball 102, spin is suppressed. - In light of feel at impact, the
golf ball 102 has an amount of compressive deformation Db of preferably 2.8 mm or greater, more preferably 2.9 mm or greater, and particularly preferably 3.0 mm or greater. In light of resilience performance, the amount of compressive deformation Db is preferably equal to or less than 3.6 mm, more preferably equal to or less than 3.5 mm, and particularly preferably equal to or less than 3.4 mm. - At measurement of the amount of compressive deformation, first, a sphere such as the
core 104, thegolf ball 102, or the like is placed on a hard plate made of metal. Next, a cylinder made of metal gradually descends toward the sphere. The sphere, squeezed between the bottom face of the cylinder and the hard plate, becomes deformed. A migration distance of the cylinder, starting from the state in which an initial load of 98 N is applied to the sphere up to the state in which a final load of 1274 N is applied thereto, is measured. - A rubber composition was obtained by kneading 100 parts by weight of a high-cis polybutadiene (trade name "BR-730", manufactured by JSR Corporation), 26 parts by weight of zinc diacrylate (trade name "Sanceler SR", manufactured by SANSHIN CHEMICAL INDUSTRY CO., LTD.), 5 parts by weight of zinc oxide, an appropriate amount of barium sulfate, 0.2 parts by weight of 2-thionaphthol, 10 parts by weight of zinc stearate, and 0.75 parts by weight of dicumyl peroxide. This rubber composition was placed into a mold including upper and lower mold halves each having a hemispherical cavity, and heated at 170°C for 25 minutes to obtain a core with a diameter of 39.1 mm. The amount of barium sulfate was adjusted such that the weight of a golf ball is 45.4 g.
- A resin composition was obtained by kneading 40 parts by weight of an ionomer resin (the aforementioned "Himilan AM7337"), 40 parts by weight of another ionomer resin (the aforementioned "Himilan AM7329"), 20 parts by weight of a styrene block-containing thermoplastic elastomer (the aforementioned "Rabalon T3221C"), and 6 parts by weight of titanium dioxide with a twin-screw kneading extruder. The core was placed into a mold. The resin composition was injected around the core by injection molding to form an inner cover with a thickness of 1.0 mm.
- A resin composition was obtained by kneading 5 parts by weight of an ionomer resin (the aforementioned "Himilan AM7337"), 10 parts by weight of another ionomer resin (the aforementioned "Himilan 1555"), 55 parts by weight of still another ionomer resin (the aforementioned "Himilan AM7329"), 30 parts by weight of an ethylene- (meth) acrylic acid copolymer (trade name "NUCREL N1050H", manufactured by Du Pont-MITSUI POLYCHEMICALS Co., Ltd.), 3 parts by weight of titanium dioxide, and 0.2 parts by weight of an ultraviolet absorber (trade name "TINUVIN 770", manufactured by Ciba Japan K.K.) with a twin-screw kneading extruder. The sphere consisting of the core and the inner cover was placed into a final mold having a large number of pimples on its cavity face. The resin composition was injected around the sphere by injection molding to form an outer cover with a thickness of 0.8 mm. Dimples having a shape that is the inverted shape of the pimples were formed on the outer cover. A clear paint including a two-component curing type polyurethane as a base material was applied to the outer cover to obtain a golf ball of Example I-1 with a diameter of 42.7 mm.
- Golf balls of Examples I-2 to I-14 and Comparative Examples I-1 to I-5 were obtained in the same manner as Example I-1, except the specifications of the core, the inner cover, and the outer cover were as shown in Tables I-7 to I-9 below. The composition of the core is shown in detail in Tables I-1 and I-2 below. The compositions of the inner cover and the outer cover are shown in detail in Table I-3 below. A hardness distribution of the core is shown in Tables I-4 to I-6 below. The golf ball according to Comparative Example I-3 does not have an inner cover.
- A 5-iron (trade name "XXIO", manufactured by SRI Sports Limited, shaft hardness: R, loft angle: 24°) was attached to a swing machine manufactured by Golf Laboratories, Inc. A golf ball was hit under the condition of a head speed of 35 m/sec. The spin rate was measured immediately after the hit. Furthermore, the distance from the launch point to the stop point was measured. The average value of data obtained by 12 measurements is shown in Tables I-7 to I-9 below.
Table I-1 Composition of Core (parts by weight) A C D E F M BR-730 100 100 100 100 100 100 Sanceler SR 27.0 26.0 27.5 29.5 31.5 25.0 Zinc oxide 5 5 5 5 5 5 Barium sulfate Appropriate amount 2-thionaphthol 0.2 0.2 0.2 0.2 0.2 0.2 Zinc stearate 0 10 20 30 40 - Aluminum stearate - - - - - 10 Dicumyl peroxide 0.75 0.75 0.75 0.75 0.75 0.75 Amount of compressive deformation Dc (mm) 3.86 3.85 3.86 3.85 3.86 3.83 Table I-2 Composition of Core (parts by weight) G H I J K L BR-730 100 100 100 100 100 100 Sanceler SR 26.5 25.5 25.0 25.5 26.0 25.5 Zinc oxide 5 5 5 5 5 5 Barium sulfate Appropriate amount 2-thionaphthol 0.2 0.2 0.2 0.2 0.2 0.2 Zinc octoate - 2.5 5 - - - Zinc laurate - - - 10 - - Zinc myristate - - - - 5 10 Zinc stearate 0.5 - - - - - Dicumyl peroxide 0.75 0.75 0.75 0.75 0.75 0.75 Amount of compressive deformation Dc (mm) 3.86 3.87 3.83 3.85 3.86 3.84 - The details of the compounds listed in Tables I-1 and I-2 are as follows.
- BR-730: a high-cis polybutadiene manufactured by JSR Corporation (cis-1,4-bond content: 96% by weight, 1,2-vinyl bond content: 1.3% by weight, Mooney viscosity (ML1+4 (100 °C)) : 55, molecular weight distribution (Mw/Mn): 3)
- Sanceler SR: zinc diacrylate manufactured by SANSHIN CHEMICAL INDUSTRY CO., LTD. (a product coated with 10% by weight of stearic acid)
- Zinc oxide: trade name "Ginrei R" manufactured by Toho Zinc Co., Ltd.
- Barium sulfate : trade name "Barium Sulfate BD" manufactured by Sakai Chemical Industry Co., Ltd.
- 2-thionaphthol: a product of Tokyo Chemical Industry Co. , Ltd.
- Zinc stearate: a product of Wako Pure Chemical Industries, Ltd.
- Aluminum stearate : a product of Mitsuwa Chemicals Co., Ltd. Dicumyl peroxide: trade name "Percumyl D" manufactured by NOF Corporation
- Zinc octoate: a product of Mitsuwa Chemicals Co., Ltd.
- Zinc laurate: a product of Mitsuwa Chemicals Co., Ltd.
- Zinc myristate: a product of NOF Corporation
- As shown in Tables I-7 to I-9, the golf balls according to Examples have excellent flight performance upon a shot with a middle iron. From the results of evaluation, advantages of the present invention are clear.
- A rubber composition was obtained by kneading 100 parts by weight of a high-cis polybutadiene (trade name "BR-730", manufactured by JSR Corporation), 26 parts by weight of zinc diacrylate (trade name "Sanceler SR", manufactured by SANSHIN CHEMICAL INDUSTRY CO., LTD.), 5 parts by weight of zinc oxide, an appropriate amount of barium sulfate, 0.2 parts by weight of 2- thionaphthol, 10 parts by weight of zinc stearate, and 0.75 parts by weight of dicumyl peroxide. This rubber composition was placed into a mold including upper and lower mold halves each having a hemispherical cavity, and heated at 170°C for 25 minutes to obtain a core with a diameter of 39.1 mm. The amount of barium sulfate was adjusted such that the weight of a golf ball is 45.4 g.
- A resin composition was obtained by kneading 24 parts by weight of an ionomer resin (the aforementioned "Himilan AM7337"), 50 parts by weight of another ionomer resin (the aforementioned "Himilan AM7329"), 26 parts by weight of a styrene block-containing thermoplastic elastomer (the aforementioned "Rabalon T3221C"), and 6 parts by weight of titanium dioxide with a twin-screw kneading extruder. The core was placed into a mold. The resin composition was injected around the core by injection molding to form an inner cover with a thickness of 1.0 mm.
- A resin composition was obtained by kneading 5 parts by weight of an ionomer resin (the aforementioned "Himilan AM7337"), 10 parts by weight of another ionomer resin (the aforementioned "Himilan 1555"), 55 parts by weight of still another ionomer resin (the aforementioned "Himilan AM7329"), 30 parts by weight of an ethylene- (meth) acrylic acid copolymer (trade name "NUCREL N1050H", manufactured by Du Pont-MITSUI POLYCHEMICALS Co., Ltd.), 3 parts by weight of titanium dioxide, and 0.2 parts by weight of an ultraviolet absorber (trade name "TINUVIN 770", manufactured by Ciba Japan K.K.) with a twin-screw kneading extruder. The sphere consisting of the core and the inner cover was placed into a final mold having a large number of pimples on its cavity face. The resin composition was injected around the sphere by injection molding to form an outer cover with a thickness of 0.8 mm. Dimples having a shape that is the inverted shape of the pimples were formed on the outer cover. A clear paint including a two-component curing type polyurethane as a base material was applied to the outer cover to obtain a golf ball of Example II-1 with a diameter of 42.7 mm.
- Golf balls of Examples II-2 to II-14 and Comparative Examples II-1 to II-5 were obtained in the same manner as Example II-1, except the specifications of the core, the inner cover, and the outer cover were as shown in Tables II-7 to II-9 below. The composition of the core is shown in detail in Tables II-1 and II-2 below. The compositions of the inner cover and the outer cover are shown in detail in Table II-3 below. A hardness distribution of the core is shown in Tables II-4 to II-6 below. The golf ball according to Comparative Example II-2 does not have an inner cover.
- A driver (trade name "XXIO", manufactured by SRI Sports Limited, shaft hardness: R, loft angle: 10.5°) was attached to a swing machine manufactured by Golf Laboratories, Inc. A golf ball was hit under the condition of a head speed of 40 m/sec. The spin rate was measured immediately after the hit. Furthermore, the distance from the launch point to the stop point was measured. The average value of data obtained by 12 measurements is shown in Tables II-7 to II-9 below.
- Ten golf players hit golf balls with drivers and were asked about feel at impact. The evaluation was categorized as follows on the basis of the number of golf players who answered, "the feel at impact was favorable".
- A: 8 or more
- B: 6 to 7
- C: 4 to 5
- D: 3 or less
- The details of the compounds listed in Tables II-1 and II-2 are as follows.
- BR-730: a high-cis polybutadiene manufactured by JSR Corporation (cis-1,4-bond content: 96% by weight, 1,2-vinyl bond content: 1.3% by weight, Mooney viscosity (ML1+4 (100°C)): 55, molecular weight distribution (Mw/Mn): 3)
- Sanceler SR: zinc diacrylate manufactured by SANSHIN CHEMICAL INDUSTRY CO., LTD. (a product coated with 10% by weight of stearic acid)
- Zinc oxide: trade name "Ginrei R" manufactured by Toho Zinc Co., Ltd.
- Barium sulfate : trade name "Barium Sulfate BD" manufactured by Sakai Chemical Industry Co., Ltd.
- 2-thionaphthol: a product of Tokyo Chemical Industry Co. , Ltd.
- Zinc stearate: a product of Wako Pure Chemical Industries, Ltd.
- Aluminum stearate : a product of Mitsuwa Chemicals Co., Ltd.
- Dicumyl peroxide: trade name "Percumyl D" manufactured by NOF Corporation
- Zinc octoate: a product of Mitsuwa Chemicals Co., Ltd.
- Zinc laurate: a product of Mitsuwa Chemicals Co., Ltd.
- Zinc myristate: a product of NOF Corporation
- As shown in Tables II-7 to II-9, the golf balls according to Examples have excellent flight performance and excellent feel at impact upon a shot with a driver. From the results of evaluation, advantages of the present invention are clear.
- The golf ball according to the present invention can be used for playing golf on golf courses and practicing at driving ranges. The above descriptions are merely for illustrative examples, and various modifications can be made without departing from the principles of the present invention.
| (parts by weight) | ||||||
| C1 | I-M1 | I-M2 | I-M3 | I-M4 | I-M5 | |
| Himilan AM7337 | 5 | 51 | 45 | 40 | 24 | 26 |
| Himilan 1555 | 10 | - | - | - | - | - |
| Himilan AM7329 | 55 | 40 | 40 | 40 | 50 | 40 |
| NUCREL N1050H | 30 | - | - | - | - | - |
| Rabalon T3221C | - | 9 | 15 | 20 | 26 | 34 |
| Titanium dioxide (A220) | 3 | 6 | 6 | 6 | 6 | 6 |
| TINUVIN 770 | 0.2 | - | - | - | - | - |
| Hardness (JIS C) | 92 | 89 | 87 | 85 | 83 | 76 |
| Comp. Ex. | Comp. Ex. | Ex. | Ex. | Ex. | Comp. Ex. | Ex. | |
| I-1 | I-2 | I-1 | I-2 | I-3 | I-3 | I-13 | |
| Composition of core | C | C | C | C | C | C | M |
| H (0) | 54.0 | 54.0 | 54.0 | 54.0 | 54.0 | 54.0 | 55.6 |
| H (12.5) | 59.8 | 59.8 | 59.8 | 59.8 | 59.8 | 59.8 | 60.2 |
| H (25) | 63.0 | 63.0 | 63.0 | 63.0 | 63.0 | 63.0 | 63.9 |
| H (37.5) | 64.6 | 64.6 | 64.6 | 64.6 | 64.6 | 64.6 | 65.4 |
| H (50) | 67.0 | 67.0 | 67.0 | 67.0 | 67.0 | 67.0 | 67.1 |
| H (62.5) | 71.8 | 71.8 | 71.8 | 71.8 | 71.8 | 71.8 | 70.9 |
| H (75) | 76.0 | 76.0 | 76.0 | 76.0 | 76.0 | 76.0 | 74.8 |
| H (87.5) | 79.5 | 79.5 | 79.5 | 79.5 | 79.5 | 79.5 | 77.7 |
| Hs | 83.0 | 83.0 | 83.0 | 83.0 | 83.0 | 83.0 | 82.3 |
| R2 | 0.99 | 0.99 | 0.99 | 0.99 | 0.99 | 0.99 | 0.99 |
| Ex. | Ex. | Ex. | Ex. | Comp. Ex. | Comp. Ex. | |
| I-4 | I-5 | I-6 | I-7 | I-4 | I-5 | |
| Composition of core | C | C | D | E | F | A |
| H (0) | 54.0 | 54.0 | 56.5 | 59.2 | 61.9 | 59.0 |
| H (12.5) | 59.8 | 59.8 | 59.7 | 61.5 | 63.2 | 64.5 |
| H (25) | 63.0 | 63.0 | 62.0 | 63.2 | 64.3 | 67.1 |
| H (37.5) | 64.6 | 64.6 | 62.8 | 64.0 | 64.3 | 67.7 |
| H (50) | 67.0 | 67.0 | 66.6 | 66.8 | 67.0 | 68.6 |
| H (62.5) | 71.8 | 71.8 | 73.7 | 71.0 | 70.4 | 70.6 |
| H (75) | 76.0 | 76.0 | 75.4 | 72.1 | 70.5 | 74.1 |
| H (87.5) | 79.5 | 79.5 | 78.2 | 73.0 | 68.5 | 79.0 |
| Hs | 83.0 | 83.0 | 81.6 | 79.1 | 70.7 | 83.0 |
| R2 | 0.99 | 0.99 | 0.98 | 0.96 | 0.86 | 0.94 |
| Ex. | Ex. | Ex. | Ex. | Ex. | Ex. | |
| I-8 | I-9 | I-10 | I-11 | I-12 | I-14 | |
| Composition of core | H | I | J | K | L | G |
| H (0) | 53.6 | 51.4 | 54.2 | 54.9 | 53.3 | 57.5 |
| H (12.5) | 58.3 | 57.6 | 58.2 | 59.0 | 58.4 | 63.5 |
| H (25) | 61.7 | 61.2 | 62.1 | 63.6 | 62.6 | 66.6 |
| H (37.5) | 65.2 | 63.8 | 64.4 | 67.0 | 65.5 | 68.8 |
| H (50) | 67.4 | 67.9 | 66.4 | 68.5 | 67.4 | 70.0 |
| H (62.5) | 71.0 | 73.8 | 71.0 | 70.1 | 71.8 | 71.2 |
| H (75) | 75.3 | 77.8 | 77.0 | 76.7 | 77.5 | 74.8 |
| H (87.5) | 80.6 | 82.0 | 80.7 | 80.5 | 81.3 | 78.8 |
| Hs | 84.1 | 84.9 | 83.3 | 83.4 | 84.5 | 82.9 |
| R2 | 0.99 | 0.99 | 0.99 | 0.98 | 0.99 | 0.96 |
| Comp. Ex. | Comp. Ex. | Ex. | Ex. | Ex. | Comp. Ex. | Ex. | |
| I-1 | I-2 | I-1 | I-2 | I-3 | I-3 | I-13 | |
| Core | |||||||
| Composition | C | C | C | C | C | C | M |
| Acid and/or salt (PHR) | 10.0 | 10.0 | 10.0 | 10.0 | 10.0 | 10.0 | 10.0 |
| Diameter (mm) | 39.1 | 39.1 | 39.1 | 39.1 | 39.1 | 39.1 | 39.1 |
| Hs - H(0) | 29.0 | 29.0 | 29.0 | 29.0 | 29.0 | 29.0 | 26.7 |
| R2 | 0.99 | 0.99 | 0.99 | 0.99 | 0.99 | 0.99 | 0.99 |
| Amount of compressive deformation Dc (mm) | 3.85 | 3.85 | 3.85 | 3.85 | 3.85 | 3.85 | 3.83 |
| Inner cover | |||||||
| Composition | I-M5 | I-M4 | I-M3 | I-M2 | I-M1 | - | I-M2 |
| Thickness (mm) | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | - | 1.0 |
| Hardness Hi (JIS C) | 76.0 | 83.0 | 85.0 | 87.0 | 89.0 | - | 87.0 |
| Amount of compressive deformation Di (mm) | 3.57 | 3.55 | 3.55 | 3.55 | 3.55 | - | 3.53 |
| Outer cover | |||||||
| Composition | C1 | C1 | C1 | C1 | C1 | C1 | C1 |
| Thickness (mm) | 0.8 | 0.8 | 0.8 | 0.8 | 0.8 | 1.8 | 0.8 |
| Hardness Ho (JIS C) | 92.0 | 92.0 | 92.0 | 92.0 | 92.0 | 92.0 | 92.0 |
| Amount of compressive deformation Db (mm) | 3.22 | 3.20 | 3.20 | 3.20 | 3.20 | 3.25 | 3.18 |
| Cover total thickness (mm) | 1.8 | 1.8 | 1.8 | 1.8 | 1.8 | 1.8 | 1.8 |
| Hi - Hs | -7.0 | 0.0 | 2.0 | 4.0 | 6.0 | - | 4.7 |
| Ho - Hi | 16.0 | 9.0 | 7.0 | 5.0 | 3.0 | - | 5.0 |
| Spin (rpm) | 3,850 | 3,820 | 3,790 | 3,770 | 3,750 | 3,830 | 3,785 |
| Difference from Comp. Ex. 5 | -50 | -80 | -110 | -130 | -150 | -70 | -115 |
| Flight distance (m) | 150.2 | 150.4 | 151.4 | 151.6 | 151.8 | 150.3 | 151.2 |
| Difference from Comp. Ex. 5 | 0.2 | 0.4 | 1.4 | 1.6 | 1.8 | 0.3 | 1.2 |
| Ex. | Ex. | Ex. | Ex. | Comp. Ex. | Comp. Ex. | |
| I-4 | I-5 | I-6 | I-7 | I-4 | I-5 | |
| Core | ||||||
| Composition | C | C | D | E | F | A |
| Acid and/or salt (PHR) | 10.0 | 10.0 | 20.0 | 30.0 | 40.0 | 0.0 |
| Diameter (mm) | 38.5 | 37.9 | 39.1 | 39.1 | 39.1 | 39.1 |
| Hs - H(0) | 28.5 | 28.1 | 25.1 | 19.9 | 8.8 | 24.0 |
| R2 | 0.99 | 0.99 | 0.98 | 0.96 | 0.86 | 0.94 |
| Amount of compressive deformation Dc (mm) | 3.85 | 3.85 | 3.86 | 3.85 | 3.86 | 3.86 |
| Inner cover | ||||||
| Composition | I-M3 | I-M3 | I-M3 | I-M3 | I-M3 | I-M3 |
| Thickness (mm) | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 |
| Hardness Hi (JIS C) | 85.0 | 85.0 | 85.0 | 85.0 | 85.0 | 85.0 |
| Amount of compressive deformation Di (mm) | 3.55 | 3.55 | 3.56 | 3.60 | 3.66 | 3.56 |
| Outer cover | ||||||
| Composition | C1 | C1 | C1 | C1 | C1 | C1 |
| Thickness (mm) | 1.1 | 1.4 | 0.8 | 0.8 | 0.8 | 0.8 |
| Hardness Ho (JIS C) | 92.0 | 92.0 | 92.0 | 92.0 | 92.0 | 92.0 |
| Amount of compressive deformation Db (mm) | 3.18 | 3.16 | 3.21 | 3.25 | 3.31 | 3.21 |
| Cover total thickness (mm) | 2.1 | 2.4 | 1.8 | 1.8 | 1.8 | 1.8 |
| Hi - Hs | 1.5 | 2.9 | 3.4 | 5.9 | 14.3 | 2.0 |
| Ho - Hi | 7.0 | 7.0 | 7.0 | 7.0 | 7.0 | 7.0 |
| Spin (rpm) | 3, 795 | 3,775 | 3,780 | 3,800 | 4,000 | 3,900 |
| Difference from Comp. Ex. 5 | -105 | -125 | -120 | -100 | 100 | - |
| Flight distance (m) | 151.1 | 151.3 | 151.2 | 151.0 | 149.4 | 150.0 |
| Difference from Comp. Ex. 5 | 1.1 | 1.3 | 1.2 | 1.0 | -0.6 | - |
| Ex. | Ex. | Ex. | Ex. | Ex. | Ex. | |
| I-8 | I-9 | I-10 | I-11 | I-12 | I-14 | |
| Core | ||||||
| Composition | H | I | J | K | L | G |
| Acid and/or salt (PHR) | 2.5 | 5.0 | 10.0 | 5.0 | 10.0 | 0.5 |
| Diameter (mm) | 39.1 | 39.1 | 39.1 | 39.1 | 39.1 | 39.1 |
| Hs - H(0) | 30.5 | 33.5 | 29.1 | 28.5 | 31.2 | 25.4 |
| R2 | 0.99 | 0.99 | 0.99 | 0.98 | 0.99 | 0.96 |
| Amount of compressive deformation Dc (mm) | 3.87 | 3.83 | 3.85 | 3.86 | 3.84 | 3.86 |
| Inner cover | ||||||
| Composition | I-M1 | I-M1 | I-M1 | I-M1 | I-M1 | I-M1 |
| Thickness (mm) | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 |
| Hardness Hi (JIS C) | 89.0 | 89.0 | 89.0 | 89.0 | 89.0 | 89.0 |
| Amount of compressive deformation Di (mm) | 3.57 | 3.53 | 3.55 | 3.56 | 3.54 | 3.56 |
| Outer cover | ||||||
| Composition | C1 | C1 | C1 | C1 | C1 | C1 |
| Thickness (mm) | 0.8 | 0.8 | 0.8 | 0.8 | 0.8 | 0.8 |
| Hardness Ho (JIS C) | 92.0 | 92.0 | 92.0 | 92.0 | 92.0 | 92.0 |
| Amount of compressive deformation Db (mm) | 3.22 | 3.18 | 3.20 | 3.21 | 3.19 | 3.21 |
| Cover total thickness (mm) | 1.8 | 1.8 | 1.8 | 1.8 | 1.8 | 1.8 |
| Hi - Hs | 4.9 | 4.1 | 5.7 | 5.6 | 4.5 | 6.1 |
| Ho - Hi | 3.0 | 3.0 | 3.0 | 3.0 | 3.0 | 3.0 |
| Spin (rpm) | 3735 | 3710 | 3745 | 3765 | 3740 | 3850 |
| Difference from Comp. Ex. 5 | -165 | -190 | -155 | -135 | -160 | -50 |
| Flight distance (m) | 151.9 | 152.2 | 151.7 | 151.5 | 152.0 | 150.5 |
| Difference from Comp. Ex. 5 | 1.9 | 2.2 | 1.7 | 1.5 | 2.0 | 0.5 |
| (parts by weight) | ||||||
| A | C | D | E | F | M | |
| BR-730 | 100 | 100 | 100 | 100 | 100 | 100 |
| Sanceler SR | 27.0 | 26.0 | 27.5 | 29.5 | 31.5 | 25.0 |
| Zinc oxide | 5 | 5 | 5 | 5 | 5 | 5 |
| Barium sulfate | Appropriate amount | |||||
| 2-thionaphthol | 0.2 | 0.2 | 0.2 | 0.2 | 0.2 | 0.2 |
| | 0 | 10 | 20 | 30 | 40 | - |
| Aluminum stearate | - | - | - | - | - | 10 |
| Dicumyl peroxide | 0.75 | 0.75 | 0.75 | 0.75 | 0.75 | 0.75 |
| Amount of compressive deformation Dc (mm) | 3.86 | 3.85 | 3.86 | 3.85 | 3.86 | 3.83 |
| G | H | I | J | K | L | |
| BR-730 | 100 | 100 | 100 | 100 | 100 | 100 |
| Sanceler SR | 26.5 | 25.5 | 25.0 | 25.5 | 26.0 | 25.5 |
| Zinc oxide | 5 | 5 | 5 | 5 | 5 | 5 |
| Barium sulfate | Appropriate amount | |||||
| 2-thionaphthol | 0.2 | 0.2 | 0.2 | 0.2 | 0.2 | 0.2 |
| Zinc octoate | - | 2.5 | 5 | - | - | - |
| Zinc laurate | - | - | - | 10 | - | - |
| Zinc myristate | - | - | - | - | 5 | 10 |
| Zinc stearate | 0.5 | - | - | - | - | - |
| Dicumyl peroxide | 0.75 | 0.75 | 0.75 | 0.75 | 0.75 | 0.75 |
| Amount of compressive deformation Dc (mm) | 3.86 | 3.87 | 3.83 | 3.85 | 3.86 | 3.84 |
| (parts by weight) | ||||||
| C1 | II-M1 | II-M2 | II-M3 | II-M4 | II-M5 | |
| Himilan AM7337 | 5 | 45 | 24 | 26 | 30 | 26 |
| Himilan 1555 | 10 | - | - | - | - | - |
| Himilan AM7329 | 55 | 40 | 50 | 40 | 30 | 26 |
| NUCREL N1050H | 30 | - | - | - | - | - |
| Rabalon T3221C | - | 15 | 26 | 34 | 40 | 48 |
| Titanium dioxide (A220) | 3 | 6 | 6 | 6 | 6 | 6 |
| TINUVIN 770 | 0.2 | - | - | - | - | - |
| Hardness (JIS C) | 92 | 87 | 83 | 76 | 71 | 65 |
| Ex. | Ex. | Ex. | Ex. | Comp. Ex. | Comp. Ex. | Ex. | |
| II-1 | II-2 | II-3 | II-4 | II-1 | II-2 | II-14 | |
| Composition of core | C | C | C | C | C | C | M |
| H (0) | 54.0 | 54.0 | 54.0 | 54.0 | 54.0 | 54.0 | 55.6 |
| H (12.5) | 59.8 | 59.8 | 59.8 | 59.8 | 59.8 | 59.8 | 60.2 |
| H (25) | 63.0 | 63.0 | 63.0 | 63.0 | 63.0 | 63.0 | 63.9 |
| H (37.5) | 64.6 | 64.6 | 64.6 | 64.6 | 64.6 | 64.6 | 65.4 |
| H (50) | 67.0 | 67.0 | 67.0 | 67.0 | 67.0 | 67.0 | 67.1 |
| H (62.5) | 71.8 | 71.8 | 71.8 | 71.8 | 71.8 | 71.8 | 70.9 |
| H (75) | 76.0 | 76.0 | 76.0 | 76.0 | 76.0 | 76.0 | 74.8 |
| H (87.5) | 79.5 | 79.5 | 79.5 | 79.5 | 79.5 | 79.5 | 77.7 |
| Hs | 83.0 | 83.0 | 83.0 | 83.0 | 83.0 | 83.0 | 82.3 |
| R2 | 0.99 | 0.99 | 0.99 | 0.99 | 0.99 | 0.99 | 0.99 |
| Ex. | Ex. | Ex. | Ex. | Comp. Ex. | Comp. Ex. | |
| II-5 | II-6 | II-7 | II-8 | II-3 | II-4 | |
| Composition of core | C | C | D | E | F | A |
| H (0) | 54.0 | 54.0 | 56.5 | 59.2 | 61.9 | 59.0 |
| H (12.5) | 59.8 | 59.8 | 59.7 | 61.5 | 63.2 | 64.5 |
| H (25) | 63.0 | 63.0 | 62.0 | 63.2 | 64.3 | 67.1 |
| H (37.5) | 64.6 | 64.6 | 62.8 | 64.0 | 64.3 | 67.7 |
| H (50) | 67.0 | 67.0 | 66.6 | 66.8 | 67.0 | 68.6 |
| H (62.5) | 71.8 | 71.8 | 73.7 | 71.0 | 70.4 | 70.6 |
| H (75) | 76.0 | 76.0 | 75.4 | 72.1 | 70.5 | 74.1 |
| H (87.5) | 79.5 | 79.5 | 78.2 | 73.0 | 68.5 | 79.0 |
| Hs | 83.0 | 83.0 | 81.6 | 79.1 | 70.7 | 83.0 |
| R2 | 0.99 | 0.99 | 0.98 | 0.96 | 0.86 | 0.94 |
| Ex. | Ex. | Ex. | Ex. | Ex. | Comp. Ex. | |
| II-9 | II-10 | II-11 | II-12 | II-13 | II-5 | |
| Composition of core | H | I | J | K | L | G |
| H (0) | 53.6 | 51.4 | 54.2 | 54.9 | 53.3 | 57.5 |
| H (12.5) | 58.3 | 57.6 | 58.2 | 59.0 | 58.4 | 63.5 |
| H (25) | 61.7 | 61.2 | 62.1 | 63.6 | 62.6 | 66.6 |
| H (37.5) | 65.2 | 63.8 | 64.4 | 67.0 | 65.5 | 68.8 |
| H (50) | 67.4 | 67.9 | 66.4 | 68.5 | 67.4 | 70.0 |
| H (62.5) | 71.0 | 73.8 | 71.0 | 70.1 | 71.8 | 71.2 |
| H (75) | 75.3 | 77.8 | 77.0 | 76.7 | 77.5 | 74.8 |
| H (87.5) | 80.6 | 82.0 | 80.7 | 80.5 | 81.3 | 78.8 |
| Hs | 84.1 | 84.9 | 83.3 | 83.4 | 84.5 | 82.9 |
| R2 | 0.99 | 0.99 | 0.99 | 0.98 | 0.99 | 0.96 |
| Comp. Ex. | Ex. | Ex. | Ex. | Ex. | Comp. Ex. | Ex. | |
| II-1 | II-1 | II-2 | II-3 | II-4 | II-2 | II-14 | |
| Core | |||||||
| Composition | C | C | C | C | C | C | M |
| Acid and/or salt (PHR) | 10.0 | 10.0 | 10.0 | 10.0 | 10.0 | 10.0 | 10.0 |
| Diameter (mm) | 39.1 | 39.1 | 39.1 | 39.1 | 39.1 | 39.1 | 39.1 |
| Hs - H(0) | 29.0 | 29.0 | 29.0 | 29.0 | 29.0 | 29.0 | 26.7 |
| R2 | 0.99 | 0.99 | 0.99 | 0.99 | 0.99 | 0.99 | 0.99 |
| Amount of compressive deformation Dc (mm) | 3.85 | 3.85 | 3.85 | 3.85 | 3.85 | 3.85 | 3.83 |
| Inner cover | |||||||
| Composition | II-M1 | II-M2 | II-M3 | II-M4 | II-M5 | - | II-M4 |
| Thickness (mm) | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | - | 1.0 |
| Hardness Hi (JIS C) | 87.0 | 83.0 | 76.0 | 71.0 | 65.0 | - | 71.0 |
| Amount of compressive deformation Di (mm) | 3.50 | 3.55 | 3.57 | 3.58 | 3.60 | - | 3.56 |
| Outer cover | |||||||
| Composition | C1 | C1 | C1 | C1 | C1 | C1 | C1 |
| Thickness (mm) | 0.8 | 0.8 | 0.8 | 0.8 | 0.8 | 1.8 | 0.8 |
| Hardness Ho (JIS C) | 92.0 | 92.0 | 92.0 | 92.0 | 92.0 | 92.0 | 92.0 |
| Amount of compressive deformation Db (mm) | 3.15 | 3.20 | 3.22 | 3.23 | 3.25 | 3.25 | 3.21 |
| Cover total thickness (mm) | 1.8 | 1.8 | 1.8 | 1.8 | 1.8 | 1.8 | 1.8 |
| Hs - Hi | -4.0 | 0.0 | 7.0 | 12.0 | 18.0 | - | 11.3 |
| Ho - Hi | 5.0 | 9.0 | 16.0 | 21.0 | 27.0 | - | 21.0 |
| Spin (rpm) | 2,455 | 2,430 | 2,420 | 2,410 | 2,400 | 2,460 | 2,430 |
| Difference from Comp. Ex. 4 | -45 | -70 | -80 | -90 | -100 | -40 | -70 |
| Flight distance (m) | 200.7 | 201.7 | 201.9 | 202.1 | 202.5 | 200.4 | 201.6 |
| Difference from Comp. Ex. 4 | 0.7 | 1.7 | 1.9 | 2.1 | 2.5 | 0.4 | 1.6 |
| Feel at impact | D | B | A | A | C | D | B |
| Ex. | Ex. | Ex. | Ex. | Comp. Ex. | Comp. Ex. | |
| II-5 | II-6 | II-7 | II-8 | II-3 | II-4 | |
| Core | ||||||
| Composition | C | C | D | E | F | A |
| Acid and/or salt (PHR) | 10.0 | 10.0 | 20.0 | 30.0 | 40.0 | 0.0 |
| Diameter (mm) | 38.5 | 37.9 | 39.1 | 39.1 | 39.1 | 39.1 |
| Hs - H(0) | 28.5 | 28.1 | 25.1 | 19.9 | 8.8 | 24.0 |
| R2 | 0.99 | 0.99 | 0.98 | 0.96 | 0.86 | 0.94 |
| Amount of compressive deformation Dc (mm) | 3.85 | 3.85 | 3.86 | 3.85 | 3.86 | 3.86 |
| Inner cover | ||||||
| Composition | II-M3 | II-M3 | II-M4 | II-M4 | II-M2 | II-M2 |
| Thickness (mm) | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 |
| Hardness Hi (JIS C) | 76.0 | 76.0 | 71.0 | 71.0 | 83.0 | 83.0 |
| Amount of compressive deformation Di (mm) | 3.57 | 3.57 | 3.59 | 3.58 | 3.66 | 3.56 |
| Outer cover | ||||||
| Composition | C1 | C1 | C1 | C1 | C1 | C1 |
| Thickness (mm) | 1.1 | 1.4 | 0.8 | 0.8 | 0.8 | 0.8 |
| Hardness Ho (JIS C) | 92.0 | 92.0 | 92.0 | 92.0 | 92.0 | 92.0 |
| Amount of compressive deformation Db (mm) | 3.20 | 3.18 | 3.24 | 3.23 | 3.31 | 3.21 |
| Cover total thickness (mm) | 2.1 | 2.4 | 1.8 | 1.8 | 1.8 | 1.8 |
| Hs - Hi | 6.5 | 6.1 | 10.6 | 8.1 | -12.3 | 0.0 |
| Ho - Hi | 16.0 | 16.0 | 21.0 | 21.0 | 9.0 | 9.0 |
| Spin (rpm) | 2,435 | 2,445 | 2,440 | 2,450 | 2, 560 | 2,500 |
| Difference from Comp. Ex. 4 | -65 | -55 | -60 | -50 | 60 | - |
| Flight distance (m) | 201.5 | 201.3 | 201.4 | 201.1 | 198.9 | 200.0 |
| Difference from Comp. Ex. 4 | 1.5 | 1.3 | 1.4 | 1.1 | -1.1 | - |
| Feel at impact | B | C | B | C | D | D |
| Ex. | Ex. | Ex. | Ex. | Ex. | Comp. Ex. | |
| II-9 | II-10 | II-11 | II-12 | II-13 | II-5 | |
| Core | ||||||
| Composition | H | I | J | K | L | G |
| Acid and/or salt (PHR) | 2.5 | 5.0 | 10.0 | 5.0 | 10.0 | 0.5 |
| Diameter (mm) | 39.1 | 39.1 | 39.1 | 39.1 | 39.1 | 39.1 |
| Hs - H (0) | 30.5 | 33.5 | 29.1 | 28.5 | 31.2 | 25.4 |
| R2 | 0.99 | 0.99 | 0.99 | 0.98 | 0.99 | 0.96 |
| Amount of compressive deformation Dc (mm) | 3.87 | 3.83 | 3.85 | 3.86 | 3.84 | 3.86 |
| Inner cover | ||||||
| Composition | II-M2 | II-M2 | II-M2 | II-M2 | II-M2 | II-M2 |
| Thickness (mm) | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 |
| Hardness Hi (JIS C) | 83.0 | 83.0 | 83.0 | 83.0 | 83.0 | 83.0 |
| Amount of compressive deformation Di (mm) | 3.57 | 3.53 | 3.55 | 3.56 | 3.54 | 3.56 |
| Outer cover | ||||||
| Composition | C1 | C1 | C1 | C1 | C1 | C1 |
| Thickness (mm) | 0.8 | 0.8 | 0.8 | 0.8 | 0.8 | 0.8 |
| Hardness Ho (JIS C) | 92.0 | 92.0 | 92.0 | 92.0 | 92.0 | 92.0 |
| Amount of compressive deformation Db (mm) | 3.22 | 3.18 | 3.20 | 3.21 | 3.19 | 3.21 |
| Cover total thickness (mm) | 1.8 | 1.8 | 1.8 | 1.8 | 1.8 | 1.8 |
| Hs - Hi | 1.1 | 1.9 | 0.3 | 0.4 | 1.5 | -0.1 |
| Ho - Hi | 9.0 | 9.0 | 9.0 | 9.0 | 9.0 | 9.0 |
| Spin (rpm) | 2415 | 2380 | 2405 | 2425 | 2385 | 2480 |
| Difference from Comp. Ex. 4 | -85 | -120 | -95 | -75 | -115 | -20 |
| Flight distance (m) | 202.0 | 202.7 | 202.3 | 201.8 | 202.6 | 200.6 |
| Difference from Comp. Ex. 4 | 2.0 | 2.7 | 2.3 | 1.8 | 2.6 | 0.6 |
| Feel at impact | B | A | A | B | A | D |
Claims (9)
- A golf ball (2, 102) consists of a spherical core (4, 104) and a cover (6, 106) covering the core (4, 104) and including two or more layers, whereinwhen distances (%) from a central point of the core (4, 104) to nine points and JIS-C hardnesses at the nine points, which nine points are obtained by dividing a region from the central point of the core (4, 104) to a surface of the core (4, 104) at intervals of 12.5% of a radius of the core (4, 104), are plotted in a graph, R2 of a linear approximation curve obtained by a least-square method is equal to or greater than 0.95, wherein R2 is calculated by squaring a correlation coefficient R, wherein the correlation coefficient R is calculated by dividing the covariance of the distance (%) from the central point and the hardness (JIS-C) by the standard deviation of the distance (%) from the central point and the standard deviation of the hardness (JIS-C),the JIS-C hardness Hi of the innermost layer of the cover (6, 106) is greater than the JIS-C hardness Hs at the surface of the core (4, 104) or the JIS-C hardness Hi of the innermost layer of the cover (6, 104) is equal to or less than the JIS-C hardness Hs at the surface of the core (4, 104), andthe core (4, 104) is formed by a rubber composition being crosslinked, wherein the rubber composition includes:(a) a base rubber,(b) a co-crosslinking agent, wherein the co-crosslinking agent (b) is:(b1) an α,β-unsaturated carboxylic acid having 3 to 8 carbon atoms; or(b2) a metal salt of an α,β-unsaturated carboxylic acid having 3 to 8 carbon atoms,(c) a crosslinking initiator,
characterized in that the rubber composition further includes:(d1) a carboxylic acid and/or a salt thereof, wherein the carboxylic acid and/or salt thereof (d1) is different from the co-crosslinking agent (b). - The golf ball (2) according to claim 1, wherein the JIS-C hardness Hi of the innermost layer of the cover (6) is greater than the JIS-C hardness Hs at the surface of the core (4) and the difference (Hs-H(0)) between the hardness Hs and the JIS-C hardness H(0) at the central point of the core (4, 104) is equal to or greater than 15.
- The golf ball (2) according to claim 1 or 2, wherein the JIS-C hardness Hi of the innermost layer of the cover (6, 106) is greater than the JIS-C hardness Hs at the surface of the core (4, 104) and the difference (Hi-Hs) between the hardness Hi and the hardness Hs is equal to or greater than 1 but equal to or less than 5.
- The golf ball (2) according to any of claims 1 to 3, wherein the JIS-C hardness Hi of the innermost layer of the cover (6, 106) is greater than the JIS-C hardness Hs at the surface of the core (4, 104) and the difference (Ho-Hi) between the JIS-C hardness Ho of the outermost layer of the cover (6, 106) and the JIS-C hardness Hi of the innermost layer of the cover (6, 106) is equal to or greater than 2 but equal to or less than 10.
- The golf ball (2) according to claim 1, wherein the amount of the carboxylic acid and/or the salt (d) is equal to or greater than 0.5 parts by weight but equal to or less than 40 parts by weight, per 100 parts by weight of the base rubber (a).
- The golf ball (2, 102) according to claim 1, wherein the carboxylic acid and/or the salt thereof (d1) is a zinc salt of a carboxylic acid.
- The golf ball (2, 102) according to claim 1, wherein the JIS-C hardness Hi of the innermost layer of the cover (6, 106) is equal to or less than the JIS-C hardness Hs at the surface of the core (4) and the difference (Hs-H(0)) between the hardness Hs and the JIS-C hardness H(0) at the central point of the core (4, 104) is equal to or greater than 15.
- The golf ball (2, 102) according to claim 1 or 7, wherein the JIS-C hardness Hi of the innermost layer of the cover (6, 106) is equal to or less than the JIS-C hardness Hs at the surface of the core (4) and the difference (Hs-Hi) between the hardness Hs and the hardness Hi is equal to or greater than 1 but equal to or less than 20.
- The golf ball (2) according to any of claims 1, 7 or 8, wherein the JIS-C hardness Hi of the innermost layer of the cover (6, 106) is equal to or less than the JIS-C hardness Hs at the surface of the core (4, 104) and the difference (Ho-Hi) between the JIS-C hardness Ho of the outermost layer of the cover (6, 106) and the JIS-C hardness Hi of the innermost layer of the cover (6, 106) is equal to or greater than 5 but equal to or less than 30.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2012126211A JP5502146B2 (en) | 2012-06-01 | 2012-06-01 | Golf ball |
| JP2012126149A JP5502145B2 (en) | 2012-06-01 | 2012-06-01 | Golf ball |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| EP2668977A2 EP2668977A2 (en) | 2013-12-04 |
| EP2668977A3 EP2668977A3 (en) | 2014-09-03 |
| EP2668977B1 true EP2668977B1 (en) | 2017-02-01 |
Family
ID=48470804
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP13168981.2A Active EP2668977B1 (en) | 2012-06-01 | 2013-05-23 | Golf ball |
Country Status (4)
| Country | Link |
|---|---|
| US (1) | US20130324321A1 (en) |
| EP (1) | EP2668977B1 (en) |
| KR (1) | KR101522086B1 (en) |
| CN (1) | CN103446721B (en) |
Families Citing this family (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20150196809A1 (en) * | 2014-01-10 | 2015-07-16 | Acushent Company | Golf balls having particle-based foam center |
| US9839814B2 (en) * | 2014-12-30 | 2017-12-12 | Acushnet Company | Multi-layer golf ball |
| USD823956S1 (en) * | 2017-05-19 | 2018-07-24 | Nexen Corporation | Golf ball |
| JP7047591B2 (en) * | 2018-05-16 | 2022-04-05 | ブリヂストンスポーツ株式会社 | Multi-piece solid golf ball |
Family Cites Families (33)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS6137178A (en) | 1984-07-28 | 1986-02-22 | 住友ゴム工業株式会社 | Solid golf ball |
| JPS6154357A (en) | 1984-08-23 | 1986-03-18 | 株式会社東芝 | Train overrun preventive device |
| US4688801A (en) * | 1985-09-23 | 1987-08-25 | Pony Ind Inc | Production of homogeneous molded golf balls |
| US5387637A (en) * | 1992-08-20 | 1995-02-07 | Lisco, Inc. | Golf ball core compositions |
| JP3153362B2 (en) | 1992-11-26 | 2001-04-09 | 住友ゴム工業株式会社 | Two piece golf ball |
| AU667267B2 (en) * | 1993-05-10 | 1996-03-14 | Wilson Sporting Goods Company | Golf ball with cover formed from hard and soft ionomer resins |
| JP3505922B2 (en) * | 1996-03-29 | 2004-03-15 | ブリヂストンスポーツ株式会社 | Three piece solid golf ball |
| JP3772252B2 (en) * | 2000-02-10 | 2006-05-10 | ブリヂストンスポーツ株式会社 | Multi-piece golf ball manufacturing method |
| JP2002000764A (en) * | 2000-06-26 | 2002-01-08 | Bridgestone Sports Co Ltd | Golf ball |
| JP2003000762A (en) * | 2001-06-21 | 2003-01-07 | Bridgestone Sports Co Ltd | Golf ball |
| JP4708610B2 (en) * | 2001-07-06 | 2011-06-22 | Sriスポーツ株式会社 | Multi-piece solid golf ball |
| JP4092097B2 (en) * | 2001-10-31 | 2008-05-28 | Sriスポーツ株式会社 | Multi-piece solid golf ball |
| JP2003180872A (en) * | 2001-12-17 | 2003-07-02 | Bridgestone Sports Co Ltd | Multipiece golf ball |
| US7066836B2 (en) * | 2004-02-04 | 2006-06-27 | Bridgestone Sports Co., Ltd. | Golf ball |
| US7199192B2 (en) | 2004-12-21 | 2007-04-03 | Callaway Golf Company | Golf ball |
| US7850548B2 (en) * | 2006-05-31 | 2010-12-14 | Bridgestone Sports Co., Ltd. | Multi-piece solid golf ball |
| US8764584B2 (en) * | 2006-09-12 | 2014-07-01 | Bridgestone Sports Co., Ltd. | Multi-piece solid golf ball |
| US7695379B2 (en) | 2007-02-13 | 2010-04-13 | Bridgestone Sports Co., Ltd. | Solid golf ball |
| US7510488B2 (en) | 2007-02-13 | 2009-03-31 | Bridgestone Sports Co., Ltd. | Solid golf ball |
| US7344455B1 (en) | 2007-02-13 | 2008-03-18 | Bridgestone Sports Co., Ltd | Solid golf ball |
| US7727085B2 (en) | 2007-02-13 | 2010-06-01 | Bridgestone Sports Co., Ltd. | Solid golf ball |
| US7481722B2 (en) | 2007-02-13 | 2009-01-27 | Bridgestone Sports Co., Ltd. | Solid golf ball |
| US7625301B2 (en) * | 2007-03-02 | 2009-12-01 | Bridgestone Sports Co., Ltd. | Golf ball |
| US7635311B2 (en) | 2007-03-02 | 2009-12-22 | Bridgestone Sports Co., Ltd. | Golf ball |
| US8039529B2 (en) | 2007-11-09 | 2011-10-18 | Bridgestone Sports Co., Ltd. | Method of manufacturing a golf ball |
| US8133136B2 (en) * | 2009-01-28 | 2012-03-13 | Bridgestone Sports Co., Ltd. | Multi-piece solid golf ball |
| US8545346B2 (en) * | 2009-01-28 | 2013-10-01 | Bridgestone Sports Co., Ltd. | Multi-piece solid golf ball |
| US8123630B2 (en) | 2009-04-27 | 2012-02-28 | Bridgestone Sports Co., Ltd. | Multi-piece solid golf ball |
| US8216090B2 (en) * | 2009-05-21 | 2012-07-10 | Bridgestone Sports Co., Ltd. | Golf ball |
| US8393978B2 (en) * | 2009-12-10 | 2013-03-12 | Bridgestone Sports Co., Ltd. | Multi-piece solid golf ball |
| JP5394327B2 (en) * | 2010-06-09 | 2014-01-22 | ダンロップスポーツ株式会社 | Golf ball |
| JP5331756B2 (en) * | 2010-06-29 | 2013-10-30 | ダンロップスポーツ株式会社 | Golf ball |
| US8764580B2 (en) * | 2011-09-30 | 2014-07-01 | Nike, Inc. | Golf ball having relationships among the densities of various layers |
-
2013
- 2013-05-23 EP EP13168981.2A patent/EP2668977B1/en active Active
- 2013-05-28 KR KR1020130060115A patent/KR101522086B1/en active IP Right Grant
- 2013-05-30 CN CN201310207820.3A patent/CN103446721B/en active Active
- 2013-05-31 US US13/906,839 patent/US20130324321A1/en not_active Abandoned
Non-Patent Citations (1)
| Title |
|---|
| None * |
Also Published As
| Publication number | Publication date |
|---|---|
| KR101522086B1 (en) | 2015-05-20 |
| CN103446721A (en) | 2013-12-18 |
| CN103446721B (en) | 2016-02-17 |
| KR20130135754A (en) | 2013-12-11 |
| EP2668977A3 (en) | 2014-09-03 |
| EP2668977A2 (en) | 2013-12-04 |
| US20130324321A1 (en) | 2013-12-05 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US9345934B2 (en) | Golf ball | |
| EP2666521B1 (en) | Golf ball | |
| EP2666522B1 (en) | Golf ball | |
| US9283441B2 (en) | Golf ball | |
| JP6068002B2 (en) | Golf ball | |
| JP5502146B2 (en) | Golf ball | |
| EP2668977B1 (en) | Golf ball | |
| JP5499085B2 (en) | Golf ball | |
| JP5502145B2 (en) | Golf ball | |
| EP2669328B1 (en) | Golf ball | |
| EP2669326B1 (en) | Golf ball | |
| JP6068004B2 (en) | Golf ball | |
| JP5499086B2 (en) | Golf ball | |
| JP5499084B2 (en) | Golf ball | |
| JP6068007B2 (en) | Golf ball | |
| JP5499082B2 (en) | Golf ball | |
| JP5499083B2 (en) | Golf ball |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AK | Designated contracting states |
Kind code of ref document: A2 Designated state(s): AL AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL PT RO RS SE SI SK SM TR |
|
| AX | Request for extension of the european patent |
Extension state: BA ME |
|
| PUAL | Search report despatched |
Free format text: ORIGINAL CODE: 0009013 |
|
| AK | Designated contracting states |
Kind code of ref document: A3 Designated state(s): AL AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL PT RO RS SE SI SK SM TR |
|
| AX | Request for extension of the european patent |
Extension state: BA ME |
|
| RIC1 | Information provided on ipc code assigned before grant |
Ipc: A63B 37/00 20060101AFI20140728BHEP |
|
| 17P | Request for examination filed |
Effective date: 20150227 |
|
| RBV | Designated contracting states (corrected) |
Designated state(s): AL AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL PT RO RS SE SI SK SM TR |
|
| 17Q | First examination report despatched |
Effective date: 20151002 |
|
| GRAP | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOSNIGR1 |
|
| INTG | Intention to grant announced |
Effective date: 20160721 |
|
| GRAJ | Information related to disapproval of communication of intention to grant by the applicant or resumption of examination proceedings by the epo deleted |
Free format text: ORIGINAL CODE: EPIDOSDIGR1 |
|
| GRAR | Information related to intention to grant a patent recorded |
Free format text: ORIGINAL CODE: EPIDOSNIGR71 |
|
| GRAS | Grant fee paid |
Free format text: ORIGINAL CODE: EPIDOSNIGR3 |
|
| INTC | Intention to grant announced (deleted) | ||
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| INTG | Intention to grant announced |
Effective date: 20161222 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AL AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL PT RO RS SE SI SK SM TR |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: FG4D |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: EP Ref country code: AT Ref legal event code: REF Ref document number: 865048 Country of ref document: AT Kind code of ref document: T Effective date: 20170215 |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FG4D |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R096 Ref document number: 602013016918 Country of ref document: DE |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: PLFP Year of fee payment: 5 |
|
| REG | Reference to a national code |
Ref country code: NL Ref legal event code: MP Effective date: 20170201 |
|
| REG | Reference to a national code |
Ref country code: LT Ref legal event code: MG4D |
|
| REG | Reference to a national code |
Ref country code: AT Ref legal event code: MK05 Ref document number: 865048 Country of ref document: AT Kind code of ref document: T Effective date: 20170201 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IS Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20170601 Ref country code: GR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20170502 Ref country code: FI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20170201 Ref country code: LT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20170201 Ref country code: NO Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20170501 Ref country code: HR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20170201 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: BG Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20170501 Ref country code: PT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20170601 Ref country code: ES Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20170201 Ref country code: LV Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20170201 Ref country code: AT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20170201 Ref country code: RS Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20170201 Ref country code: PL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20170201 Ref country code: SE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20170201 Ref country code: LU Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20170531 Ref country code: NL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20170201 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20170201 Ref country code: SK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20170201 Ref country code: EE Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20170201 Ref country code: CZ Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20170201 Ref country code: RO Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20170201 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R097 Ref document number: 602013016918 Country of ref document: DE |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SM Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20170201 Ref country code: DK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20170201 |
|
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| 26N | No opposition filed |
Effective date: 20171103 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: MC Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20170201 |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: MM4A |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LI Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20170531 Ref country code: SI Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20170201 Ref country code: CH Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20170531 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LU Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20170523 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: PLFP Year of fee payment: 6 |
|
| REG | Reference to a national code |
Ref country code: BE Ref legal event code: MM Effective date: 20170531 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20170523 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R082 Ref document number: 602013016918 Country of ref document: DE Representative=s name: MANITZ FINSTERWALD PATENT- UND RECHTSANWALTSPA, DE Ref country code: DE Ref legal event code: R082 Ref document number: 602013016918 Country of ref document: DE Representative=s name: MANITZ FINSTERWALD PATENTANWAELTE PARTMBB, DE Ref country code: DE Ref legal event code: R081 Ref document number: 602013016918 Country of ref document: DE Owner name: SUMITOMO RUBBER INDUSTRIES LTD., KOBE-SHI, JP Free format text: FORMER OWNER: DUNLOP SPORTS CO., LTD., KOBE-SHI, HYOGO, JP |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: 732E Free format text: REGISTERED BETWEEN 20180607 AND 20180613 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: BE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20170531 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: MT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20170523 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: TP Owner name: SUMITOMO RUBBER INDUSTRIES, LTD., JP Effective date: 20180914 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: HU Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT; INVALID AB INITIO Effective date: 20130523 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: CY Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20170201 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: MK Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20170201 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: TR Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20170201 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: AL Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRESCRIBED TIME-LIMIT Effective date: 20170201 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: PLFP Year of fee payment: 11 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 20230330 Year of fee payment: 11 |
|
| P01 | Opt-out of the competence of the unified patent court (upc) registered |
Effective date: 20230510 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: DE Payment date: 20230331 Year of fee payment: 11 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 20240328 Year of fee payment: 12 |