EP0672749A1 - Bleaching compositions - Google Patents
Bleaching compositions Download PDFInfo
- Publication number
- EP0672749A1 EP0672749A1 EP95301219A EP95301219A EP0672749A1 EP 0672749 A1 EP0672749 A1 EP 0672749A1 EP 95301219 A EP95301219 A EP 95301219A EP 95301219 A EP95301219 A EP 95301219A EP 0672749 A1 EP0672749 A1 EP 0672749A1
- Authority
- EP
- European Patent Office
- Prior art keywords
- acid
- compound
- precursor
- alkyl
- preferred
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- HOXMBUQLRDTYRV-JECWYVHBSA-N CCCC(OC1O[C@@H]1CNC)I Chemical compound CCCC(OC1O[C@@H]1CNC)I HOXMBUQLRDTYRV-JECWYVHBSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/16—Organic compounds
- C11D3/26—Organic compounds containing nitrogen
- C11D3/33—Amino carboxylic acids
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/0005—Other compounding ingredients characterised by their effect
- C11D3/0073—Anticorrosion compositions
-
- C—CHEMISTRY; METALLURGY
- C11—ANIMAL OR VEGETABLE OILS, FATS, FATTY SUBSTANCES OR WAXES; FATTY ACIDS THEREFROM; DETERGENTS; CANDLES
- C11D—DETERGENT COMPOSITIONS; USE OF SINGLE SUBSTANCES AS DETERGENTS; SOAP OR SOAP-MAKING; RESIN SOAPS; RECOVERY OF GLYCEROL
- C11D3/00—Other compounding ingredients of detergent compositions covered in group C11D1/00
- C11D3/39—Organic or inorganic per-compounds
- C11D3/3902—Organic or inorganic per-compounds combined with specific additives
- C11D3/3905—Bleach activators or bleach catalysts
- C11D3/3907—Organic compounds
Definitions
- the present invention relates to the inclusion of Mn(II) compounds in bleaching compositions, suitable for use in machine dishwashing.
- the compositions exhibit good bleachable stain removal and enhanced anti silver-tarnishing properties.
- compositions designed for use in automatic dishwasher machines are well known, and a consistent effort has been made by detergent manufacturers to improve the cleaning and/or rinsing efficiency of said compositions on dishes and glassware, as reflected by numerous patent publications.
- the present invention is concerned with the silver-tarnishing problem encountered when compositions which contain oxygen-bleaching species are employed in machine dishwashing methods.
- a problem encountered with the use of such oxygen bleaches is the tarnishing of any silverware components of the washload.
- the level of tarnishing observed can range from slight discolouration of the silverware to the formation of a dense black coating on the surface of the silverware.
- the formulator thus faces the dual challenge of formulating a product which maximises bleachable soil cleaning but minimises the occurrence of tarnishing of silverware components of the washload.
- the bleach system comprises a combination of an inorganic perhydrate bleach, especially a percarbonate bleach, and a peroxyacid bleach precursor selected from perbenzoic acid precursors, perbenzoic acid derivative precursors, N-acylated lactam precursors and cationic peroxyacid precursors.
- the total level of oxygen bleach in the composition is desirably optimized, and preferably the rate of release of the oxygen bleach is controlled.
- the rate of release of oxygen bleach is preferably rapid enough to provide satisfactory cleaning, but with minimised tarnishing.
- compositions suitable for use in machine dishwashing methods, having enhanced anti-silver tarnishing properties, as well as good cleaning performance, particularly bleachable soil removal performance.
- German patent DE-A-4,128,672 in the name of Henkel KgaA discloses strong alkaline, bleach-containing detergents described to have a reduced tendency to tarnish silverware.
- Compositions containing Mn(II) salts such as MnSO4 and MnCl2 in combination with inorganic perhydrate salts and certain bleach activators are disclosed.
- the combination of said Mn(II) salts or compounds with N-acylated lactam, perbenzoic acid, perbenzoic acid derivative or cationic peroxyacid precursors is not disclosed. Nor is there any recognition provided that such bleach precursors particularly give rise to the problem of silver tarnishing.
- Mn(II) salts are used as activators for peroxy compounds there is no protection of the silver, in direct contrast to the present invention which recognizes the ability of certain Mn(II) salts to ameliorate the problem of silver tarnishing.
- EP-A-0 530 870 in the name of Unilever discloses machine dishwashing compositions containing a dinuclear manganese complex in which the manganese is in the III or IV oxidation state. No recognition is provided is this document of the utility of Mn(II) salts in reducing silver tarnishing.
- said oxygen-releasing bleaching system is incorporated such that the level of available oxygen measured according to the method herein is from 0.3% to 2.5%, preferably 0.5% to 1.7% by weight.
- the rate of release of said available oxygen is such that the available oxygen is completely released from the composition in a time interval of from 3.5 minutes to 10.0 minutes, using the test protocol described in the present description.
- the bleaching composition contains no additional corrosion inhibitor component, particularly no benzotriazole.
- compositions are adapted for use in a machine dishwashing method, in particular being low foaming in character.
- compositions contain, at most, low levels of low foaming surfactant which is often nonionic in character.
- the Mn(II) compound is an Mn(II) salt or complex with an organic ligand, which may itself have heavy metal ion sequestering or crystal growth inhibiting properties.
- compositions contain as essential components an oxygen-releasing bleach system and an Mn(II) compound.
- the level of available oxygen is preferably controlled.
- the rate of release of available oxygen and of any organic peroxyacid bleaching species is also controlled.
- An essential feature of the invention is an oxygen-releasing bleaching system containing a hydrogen peroxide source and a peroxyacid bleach precursor compound.
- the production of the peroxyacid occurs by an in situ reaction of the precursor with a source of hydrogen peroxide.
- Suitable sources of hydrogen peroxide include inorganic perhydrate bleaches.
- the peroxyacid bleach precursor is most preferably selected from perbenzoic acid precursors, perbenzoic acid derivative precursors, N-acylated lactam bleach precursors and cationic peroxyacid precursors, or mixtures thereof.
- the peroxyacid bleach precursors are preferably incorporated at a level of from 0.5% to 20% by weight, more preferably from 12% to 15% by weight, most preferably from 1.5% to 10% by weight of the compositions.
- N-acylated precursor compounds of the lactam class are disclosed generally in GB-A-855735. Whilst the broadest aspect of the invention contemplates the use of any lactam useful as a peroxyacid precursor, preferred materials comprise the caprolactams and valerolactams.
- Suitable N-acylated lactam precursors have the formula: wherein n is from 0 to about 8, preferably from 0 to 2, and R6 is H, an alkyl, aryl, alkoxyaryl or alkaryl group containing from 1 to 12 carbons, or a substituted phenyl group containing from 6 to 18 carbon atoms
- Suitable caprolactam bleach precursors are of the formula: wherein R1 is H or an alkyl, aryl, alkoxyaryl or alkaryl group containing from 1 to 12 carbon atoms, preferably from 6 to 12 carbon atoms, most preferably R1 is phenyl.
- Suitable valero lactams have the formula: wherein R1 is H or an alkyl, aryl, alkoxyaryl or alkaryl group containing from 1 to 12 carbon atoms, preferably from 6 to 12 carbon atoms.
- R1 is selected from phenyl, heptyl, octyl, nonyl, 2,4,4-trimethylpentyl, decenyl and mixtures thereof.
- the most preferred materials are those which are normally solid at ⁇ 30°C, particularly the phenyl derivatives, ie. benzoyl valerolactam, benzoyl caprolactam and their substituted benzoyl analogues such as chloro, amino alkyl, alkyl, aryl and alkoxy derivatives.
- Precursor compounds wherein R1 comprises from 1 to 6 carbon atoms provide hydrophilic bleaching species which are particularly efficient for bleaching beverage stains.
- Mixtures of 'hydrophobic' and 'hydrophilic' caprolactams and valero lactams, typically at weight ratios of 1:5 to 5:1, preferably 1:1, can be used herein for mixed stain removal benefits.
- caprolactam and valerolactam precursors include benzoyl caprolactam, nonanoyl capro-lactam, benzoyl valerolactam, nonanoyl valerolactam, 3,5,5-trimethylhexanoyl caprolactam, 3,5,5-trimethylhexanoyl valerolactam, octanoyl caprolactam, octanoyl valerolactam, decanoyl caprolactam, decanoyl valerolactam, undecenoyl caprolactam, undecenoyl valerolactam, (6-octanamidocaproyl)oxybenzene-sulfonate, (6-nonanamidocaproyl)oxybenzenesulfonate, (6-decanamidocaproyl)oxybenzenesulfonate, and mixtures thereof.
- Examples of highly preferred substituted benzoyl lactams include methylbenzoyl caprolactam, methylbenzoyl valerolactam, ethylbenzoyl caprolactam, ethylbenzoyl valerolactam, propylbenzoyl caprolactam, propylbenzoyl valerolactam, isopropylbenzoyl caprolactam, isopropylbenzoyl valerolactam, butylbenzoyl caprolactam, butylbenzoyl valerolactam, tert-butylbenzoyl caprolactam, tert-butylbenzoyl valerolactam, pentylbenzoyl caprolactam, pentylbenzoyl valerolactam, hexylbenzoyl caprolactam, hexylbenzoyl valerolactam, ethoxybenzoyl caprolactam, ethoxybenzo
- any perbenzoic acid precursors are suitable herein, including those of the N-acylated lactam class, which are preferred.
- O-acylated perbenzoic acid precursor compounds include benzoyl oxybenzene sulfonate and monobenzoyltetraacetyl glucose benzoyl peroxide.
- Preferred perbenzoic acid precursor compounds of the imide type include N-benzoyl succinimide, tetrabenzoyl ethylene diamine and the N-benzoyl substituted ureas.
- Suitable imidazoles include N-benzoyl imidazole and N-benzoyl benzimidazole and other useful N-acyl group-containing peroxyacid precursors include N-benzoyl pyrrolidone, dibenzoyl taurine and benzoyl pyroglutamic acid.
- Preferred perbenzoic acid precursors include the benzoyl diacyl peroxides and benzoyl tetraacyl peroxides.
- R1 is an aryl group with from 1 to 14 carbon atoms
- R2 is an alkylene, arylene, and alkarylene group containing from 1 to 14 carbon atoms
- R5 is H or an alkyl, aryl, or alkaryl group containing 1 to 10 carbon atoms and L can be essentially any leaving group.
- R1 preferably contains from 6 to 12 carbon atoms.
- R2 preferably contains from 4 to 8 carbon atoms.
- R1 may be aryl, substituted aryl or alkylaryl containing branching, substitution, or both and may be sourced from either synthetic sources or natural sources including for example, tallow fat.
- R2 Analogous structural variations are permissible for R2.
- the substitution can include alkyl, aryl, halogen, nitrogen, sulphur and other typical substituent groups or organic compounds.
- R5 is preferably H or methyl.
- R1 and R5 should not contain more than 18 carbon atoms in total. Amide substituted bleach activator compounds of this type are described in EP-A-0170386.
- the L group must be sufficiently reactive for the reaction to occur within the optimum time frame (e.g., a wash cycle). However, if L is too reactive, this activator will be difficult to stabilize for use in a bleaching composition.
- pKa of the conjugate acid of the leaving group although exceptions to this convention are known. Ordinarily, leaving groups that exhibit such behavior are those in which their conjugate acid has a pKa in the range of from 4 to 13, preferably from 6 to 11 and most preferably from 8 to 11.
- Preferred bleach precursors are those wherein R1, R2 and R5 are as defined for the amide substituted compounds and L is selected from the group consisting of: and mixtures thereof, wherein R1 is an alkyl, aryl, or alkaryl group containing from 1 to 14 carbon atoms, R3 is an alkyl chain containing from 1 to 8 carbon atoms, R4 is H or R3, and Y is H or a solubilizing group.
- the preferred solubilizing groups are -SO3 ⁇ M+, -CO2 ⁇ M+, -SO4 ⁇ M+, -N+(R3)4X ⁇ and O ⁇ --N(R3)3 and most preferably -SO3 ⁇ M+ and -CO2 ⁇ M+ wherein R3 is an alkyl chain containing from 1 to 4 carbon atoms, M is a cation which provides solubility to the bleach activator and X is an anion which provides solubility to the bleach activator.
- M is an alkali metal, ammonium or substituted ammonium cation, with sodium and potassium being most preferred, and X is a halide, hydroxide, methylsulfate or acetate anion.
- bleach activators with a leaving group that does not contain a solubilizing groups should be well dispersed in the bleaching solution in order to assist in their dissolution.
- perbenzoic acid precursors include:
- Certain preformed peracids including:
- Suitable perbenzoic acid derivative precursors include any of the herein disclosed perbenzoic precursors in which the perbenzoic group is substituted by essentially any functional group including alkyl groups.
- Cationic peroxyacid precursor compounds are also suitable herein.
- cationic peroxyacid precursors are formed by substituting the peroxyacid part with an ammonium or alkyl ammmonium group, preferably an ethyl or methyl ammonium group.
- Cationic peroxyacid precursors are described in U.S. Patents 4,904,406; 4,751,015; 4,988,451; 4,397,757; 5,269,962; 5,127,852; 5,093,022; 5,106,528; U.K. 1,382,594; EP 475,512, 458,396 and 284,292; and in JP 87-318,332.
- Suitable cationic peroxyacid precursors include any of the ammonium or alkyl ammonium substituted alkyl or benzoyl oxybenzene sulfonates, N-acylated caprolactams, and monobenzoyltetraacetyl glucose benzoyl peroxides.
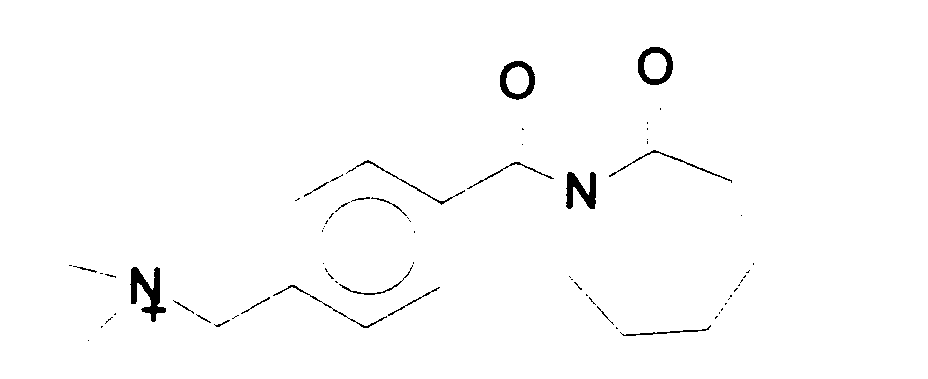
- a preferred cationically substituted benzoyl oxybenzene sulfonate is the 4-(trimethyl ammonium) methyl derivative of benzoyl oxybenzene sulfonate:
- a preferred cationically substituted alkyl oxybenzene sulfonate is the methyl ammonium derivative of 2,3,3-tri-methyl hexanoyloxybenzene sulfonate.
- Preferred cationic peroxyacid precursors of the N-acylated caprolactam class include the trialkyl ammonium methylene benzoyl caprolactams, particularly trimethyl ammonium methylene benzoyl caprolactam:
- Another preferred cationic peroxyacid precursor is 2-(N,N,N-trimethyl ammonium) ethyl sodium 4-sulphophenyl carbonate chloride.
- Betaine ester trimethyl ammonium meta chloro xylenol is also preferred:
- compositions in accord with the invention include a hydrogen peroxide source.
- Suitable hydrogen peroxide sources include the inorganic perhydrate salts.
- the inorganic perhydrate salts are normally incorporated in the form of the sodium salt at a level of from 1% to 40% by weight, more preferably from 2% to 30% by weight and most preferably from 5% to 25% by weight of the compositions.
- inorganic perhydrate salts include perborate, percarbonate, perphosphate, persulfate and persilicate salts.
- the inorganic perhydrate salts are normally the alkali metal salts.
- the inorganic perhydrate salt may be included as the crystalline solid without additional protection.
- the preferred executions of such granular compositions utilize a coated form of the material which provides better storage stability for the perhydrate salt in the granular product.
- Sodium perborate can be in the form of the monohydrate of nominal formula NaBO2H2O2 or the tetrahydrate NaBO2H2O2.3H2O.
- Sodium percarbonate which is a preferred perhydrate for inclusion in compositions in accordance with the invention, is an addition compound having a formula typically corresponding to 2Na2CO3.3H2O2, and is available commercially as a crystalline solid.
- the percarbonate is most preferably incorporated into such compositions in a coated form which provides in product stability.
- a suitable coating material providing in product stability comprises mixed salt of a water soluble alkali metal sulphate and carbonate.
- the weight ratio of the mixed salt coating material to percarbonate lies in the range from 1 : 200 to 1: 4, more preferably from 1: 99 to 1: 9, and most preferably from 1: 49 to 1 : 19.
- the mixed salt is of sodium sulphate and sodium carbonate which has the general formula Na2SO4.n.Na2CO3 wherein n is form 0.1 to 3, preferably n is from 0.3 to 1.0 and most preferably n is from 0.2 to 0.5.
- coatings which contain silicate (alone or with borate salts or boric acids or other inorganics), waxes, oils, fatty soaps can alos be used advantageously within the present invention.
- Potassium peroxymonopersulfate is another inorganic perhydrate salt of use in the detergent compositions herein.
- Suitable additional peroxyacid bleach precursors typically contain one or more N- or O- acyl groups, which precursors can be selected from a wide range of classes. Suitable classes include anhydrides, esters, imides and acylated derivatives of imidazoles and oximes, and examples of useful materials within these classes are disclosed in GB-A-1586789.
- Suitable esters are disclosed in GB-A-836988, 864798, 1147871,2143231 and EP-A-0170386.
- Useful N-acyl compounds are disclosed in GB-A-855735, 907356 and GB-A-1246338.
- Preferred precursor compounds of the imide type include the N-,N,N1N1 tetra acetylated alkylene diamines wherein the alkylene group contains from 1 to 6 carbon atoms, particularly those compounds in which the alkylene group contains 1, 2 and 6 carbon atoms. Tetraacetyl ethylene diamine (TAED) is particularly preferred.
- TAED Tetraacetyl ethylene diamine
- R1 is an alkyl group with from 1 to 14 carbon atoms
- R2 is an alkylene, arylene, and alkarylene group containing from 1 to 14 carbon atoms
- R5 is H or an alkyl, aryl, or alkaryl group containing 1 to 10 carbon atoms and L can be essentially any leaving group.
- R1 preferably contains from 6 to 12 carbon atoms.
- R2 preferably contains from 4 to 8 carbon atoms.
- R1 may be straight chain or branched alkyl containing branching, substitution, or both and may be sourced from either synthetic sources or natural sources including for example, tallow fat.
- R2 Analogous structural variations are permissible for R2.
- the substitution can include alkyl, aryl, halogen, nitrogen, sulphur and other typical substituent groups or organic compounds.
- R5 is preferably H or methyl.
- R1 and R5 should not contain more than 18 carbon atoms in total.
- L may be selected from any of the leaving groups descrobed hereinbefore for the analogues having R1 as an aryl or alkaryl group.
- Amide substituted bleach activator compounds of this type are described in EP-A-0170386.
- compositions may also contain organic peroxyacids, typically at a level of from 1% to 15% by weight, more preferably from 1% to 10% by weight of the composition.
- a preferred class of organic peroxyacid compounds are the amide substituted compounds of the following general formulae: wherein R1 is an alkyl, aryl or alkaryl group with from 1 to 14 carbon atoms, R2 is an alkylene, arylene, and alkarylene group containing from 1 to 14 carbon atoms, and R5 is H or an alkyl, aryl, or alkaryl group containing 1 to 10 carbon atoms. R1 preferably contains from 6 to 12 carbon atoms. R2 preferably contains from 4 to 8 carbon atoms.
- R1 may be straight chain or branched alkyl, substituted aryl or alkylaryl containing branching, substitution, or both and may be sourced from either synthetic sources or natural sources including for example, tallow fat. Analogous structural variations are permissible for R2. The substitution can include alkyl, aryl, halogen, nitrogen, sulphur and other typical substituent groups or organic compounds.
- R5 is preferably H or methyl. R1 and R5 should not contain more than 18 carbon atoms in total. Amide substituted organic peroxyacid compounds of this type are described in EP-A-0170386.
- organic peroxyacids include diperoxydodecanedioc acid, diperoxytetra decanedioc acid, diperoxyhexadecanedioc acid, mono- and diperazelaic acid, mono- and diperbrassylic acid, monoperoxy phthalic acid and its magnesium salt, perbenzoic acid, and their salts as disclosed in, for example, EP-A-0341 947.
- the level of available oxygen in the present compositions should preferably be carefully controlled; the level of available oxygen should hence preferably be in the range from 0.3% to 2.5%, preferably from 0.5% to 1.7%, more preferably from 0.6% to 1.5%, most preferably from 0.7% to 1.2%, measured according to the method described hereunder.
- the rate of release of available oxygen is preferably also controlled; the rate of release of available oxygen from the compositions herein preferably should be such that, when using the method described hereinafter, the available oxygen is not completely released from the composition until after 3.5 minutes, preferably the available oxygen is released in a time interval of from 3.5 minutes to 10.0 minutes, more preferably from 4.0 minutes to 9.0 minutes, most preferably from 5.0 minutes to 8.5 minutes.
- the level of AvO, measured in units of % available oxygen by weight, for the sample at each time interval corresponds to the amount of titre according to the following equation VolS2O3(ml) x Molarity (S2O3) x 8 Sample mass (g)
- AvO level is plotted versus time to determine the maximum level of AvO, and the rate of release of AvO
- the rate of release of any organic peroxyacid species is also preferably controlled.
- the kinetics of release to a wash solution are preferably such that in the T50 test method herein described the time to achieve a concentration that is 50% of the ultimate concentration of the peroxyacid bleach is from 180 seconds to 480 seconds, more preferably from 200 seconds to 400 seconds, most preferably from 240 seconds to 360 seconds.
- controlled release kinetics or organic peroxyacid herein are defined with respect to a 'TA test method' which measures the time to achieve A% of the ultimate concentration/level of the organic peroxyacid when a composition containing the organic peroxyacid is dissolved according to the standard conditions now set out.
- the standard conditions involve a 1 litre glass beaker filled with 1000 ml of distilled water at 20°C, to which 10g of composition is added. The contents of the beaker are agitated using a magnetic stirrer set at 100 rpm. The ultimate concentration/level is taken to be the concentration/level maximum level of organic peroxyacid attained after addition of the composition to the water-filled beaker.
- Suitable analytical methods are chosen to enable a reliable determination of the incidental, and ultimate in solution concentrations of the organic peroxyacid, subsequent to the addition of the composition to the water in the beaker.
- Such analytical methods can include those involving a continuous monitoring of the level of concentration of the component, including for example photometric and conductrimetric methods.
- methods involving removing titres from the solution at set time intervals, stopping the disssolution process by an appropriate means such as by rapidly reducing the temperature of the titre, and then determining the concentration of the component in the titre by any means such as chemical titrimetric methods, can be employed.
- Suitable graphical methods including curve fitting methods, can be employed, where appropriate, to enable calculation of the the TA value from raw analytical results.
- the particular analytical method selected for determining the concentration of the organic peroxyacid component will depend on the nature of that component, and of the nature of the composition containing that component.
- Controlled rate of release - means
- a means may be provided for controlling the rate of release of AvO, or of organic peroxyacid to the wash solution.
- Means for controlling the rate of release of any oxygen bleaching species may provide for controlled release of the oxygen bleaching species itself to the wash solution.
- the oxygen bleaching species comprises in combination a hydrogen peroxide source and an organic peroxyacid bleach precursor compound
- the means may comprise a means of inhibiting, or preventing the in situ perhydrolysis reaction which releases the peroxyacid into the solution.
- the means could, for example, include controlling release of the hydrogen peroxide source to the wash solution, by for example, controlling release of any inorganic perhydrate salt, acting as a hydrogen peroxide source, to the wash solution.
- Suitable controlled release means can include coating any suitable component with a coating designed to provide the controlled release.
- the coating may therefore, for example, comprise a poorly water soluble material, or be a coating of sufficient thickness that the kinetics of dissolution of the thick coating provide the controlled rate of release.
- the coating material may be applied using various methods. Any coating material is typically present at a weight ratio of coating material to bleach of from 1:99 to 1:2, preferably from 1:49 to 1:9.
- Suitable coating materials include triglycerides (e.g. partially) hydrogenated vegetable oil, soy bean oil, cotton seed oil) mono or diglycerides, microcrystalline waxes, gelatin, cellulose, fatty acids and any mixtures thereof.
- suitable coating materials can comprise the alkali and alkaline earth metal sulphates, silicates and carbonates, including calcium carbonate and silicas.
- a preferred coating material is sodium silicate of SiO2 : Na2O ratio from 1.6 : 1 to 3.4 : 1, preferably 2.2:1 to 2.8:1, applied as an aqueous solution to give a level of from 2% to 10%, (normally from 3% to 5%) of silicate solids by weight of the oxygen bleaching species.
- Magnesium silicate can also be included in the coating.
- Any inorganic salt coating materials may be combined with organic binder materials to provie composite inorganic salt/organic binder coatings.
- Suitable binders include the C10-C20 alcohol ethoxylates containing from 5 - 100 moles of ethylene oxide per mole of alcohol and more preferably the C15-C20 primary alcohol ethoxylates containing from 20 - 100 moles of ethylene oxide per mole of alcohol.
- binders include certain polymeric materials.
- Polyvinylpyrrolidones with an average molecular weight of from 12,000 to 700,000 and polyethylene glycols (PEG) with an average molecular weight of from 600 to 5 x 106 preferably 1000 to 400,000 most preferably 1000 to 10,000 are examples of such polymeric materials.
- Copolymers of maleic anhydride with ethylene, methylvinyl ether or methacrylic acid, the maleic anhydride constituting at least 20 mole percent of the polymer are further examples of polymeric materials useful as binder agents.
- polymeric materials may be used as such or in combination with solvents such as water, propylene glycol and the above mentioned C10-C20 alcohol ethoxylates containing from 5 - 100 moles of ethylene oxide per mole.
- solvents such as water, propylene glycol and the above mentioned C10-C20 alcohol ethoxylates containing from 5 - 100 moles of ethylene oxide per mole.
- binders include the C10-C20 mono- and diglycerol ethers and also the C10-C20 fatty acids.
- Cellulose derivatives such as methylcellulose, carboxymethylcellulose and hydroxyethylcellulose, and homo- or co-polymeric polycarboxylic acids or their salts are other examples of binders suitable for use herein.
- One method for applying the coating material involves agglomeration.
- Preferred agglomeration processes include the use of any of the organic binder materials described hereinabove. Any conventional agglomerator/mixer may be used including, but not limted to pan, rotary drum and vertical blender types. Molten coating compositions may also be applied either by being poured onto, or spray atomized onto a moving bed of bleaching agent.
- Suitable means of providing the required controlled release include mechanical means for altering the physical characteristics of the bleach to control its solubility and rate of release. Suitable protocols could include compaction, mechanical injection, manual injection, and adjustment of the solubility of the bleach compound by selection of particle size of any particulate component.
- particle size Whilst the choice of particle size will depend both on the composition of the particulate component, and the desire to meet the desired controlled release kinetics, it is desirable that the particle size should be more than 500 micrometers, preferably having an average particle diameter of from 800 to 1200 micrometers.
- Additional protocols for providing the means of controlled release include the suitable choice of any other components of the detergent composition matrix such that when the composition is introduced to the wash solution the ionic strength environment therein provided enables the required controlled release kinetics to be achieved.
- the compositions contain an Mn(II) compound to inhibit the tarnishing of silverware.
- the Mn(II) compound is preferably incorporated at a level of from 0.005% to 5% by weight, more preferably from 0.01% to 1%, most preferably from 0.02% to 0.4% by weight of the compositions.
- the Mn(II) compound is incorporated at a level to provide from 0.1 ppm to 250 ppm, more preferably from 0.5 ppm to 50 ppm, most preferably from 1 ppm to 20 ppm by weight of Mn(II) ions in any bleaching solution.
- the Mn (II) compound may be an inorganic salt in anhydrous, or any hydrated forms.
- Suitable salts include manganese sulphate, manganese carbonate, manganese phosphate, manganese nitrate, manganese acetate and manganese chloride.
- the Mn(II) compound may be a salt or complex of an organic fatty acid such as manganese acetate or manganese stearate.
- the Mn(II) compound may be a salt or complex of an organic ligand.
- any salts/complexes are water soluble.
- the organic ligand is a heavy metal ion sequestrant.
- the organic ligand is a crystal growth inhibitor compound.
- heavy metal ion sequestrant it is meant herein components which act to sequester (chelate) heavy metal ions. These components may also have calcium and magnesium chelation capacity, but preferentially they show selectivity to binding heavy metal ions such as iron, manganese and copper.
- Suitable heavy metal ion sequestrants include organic aminophosphonates, such as the amino alkylene poly (alkylene phosphonates), and nitrilo trimethylene phosphonates.
- Preferred among the above species are diethylene triamine penta (methylene phosphonate), ethylene diamine tri (methylene phosphonate) and hexamethylene diamine tetra (methylene phosphonate).
- Suitable heavy metal ion sequestrants include nitrilotriacetic acid and polyaminocarboxylic acids such as ethylenediaminetetracetic acid, ethylenetriamine pentacetic acid, ethylenediamine disuccinic acid, ethylenediamine diglutaric acid, 2-hydroxypropylenediamine disuccinic acid, and the partially or fully ionized forms thereof.
- Ethylenediamine disuccinic acid is preferred, most preferably in the form of its S,S isomer.
- Suitable heavy metal ion sequestrants for use herein are iminodiacetic acid derivatives such as 2-hydroxyethyl diacetic acid or glyceryl imino diacetic acid, described in EP-A-317,542 and EP-A-399,133.
- iminodiacetic acid-N-2-hydroxypropyl sulfonic acid and aspartic acid N-carboxymethyl N-2-hydroxypropyl-3-sulfonic acid sequestrants described in EP-A-516,102 are also suitable herein.
- EP-A-476,257 describes suitable amino based heavy metal ion sequestrants.
- EP-A-510,331 describes suitable sequestrants derived from collagen, keratin or casein.
- EP-A-528,859 describes a suitable alkyl iminodiacetic acid sequestrant. Dipicolinic acid and 2-phosphonobutane-1,2,4-tricarboxylic acid are also suitable.
- Glycinamide-N,N'-disuccinic acid (GADS) is also suitable.
- the organic ligand may be a crystal growth inhibitor component, particularly an organo diphosphonic acid component.
- organo diphosphonic acid it is meant herein an organo diphosphonic acid which does not contain nitrogen as part of its chemical structure. This definition therefore excludes the organo aminophosphonates, which however may be included in compositions of the invention as heavy metal ion sequestrant components.
- the organo diphosphonic acid is preferably a C1-C4 diphosphonic acid, more preferably a C2 diphosphonic acid, such as ethylene diphosphonic acid, or most preferably ethane 1-hydroxy-1,1-diphosphonic acid (HEDP) and may be present in the Mn(II) compound in partially or fully ionized form.
- a C1-C4 diphosphonic acid more preferably a C2 diphosphonic acid, such as ethylene diphosphonic acid, or most preferably ethane 1-hydroxy-1,1-diphosphonic acid (HEDP) and may be present in the Mn(II) compound in partially or fully ionized form.
- compositions of the invention may comprise additional ingredients, which are often quite desirable ones.
- compositions of the present invention may contain as a highly preferred component a water-soluble builder compound, typically present at a level of from 1% to 80% by weight, preferably from 10% to 70% by weight, most preferably from 20% to 60% by weight of the composition.
- Suitable water-soluble builder compounds include the water soluble monomeric polycarboxylates, or their acid forms, homo or copolymeric polycarboxylic acids or their salts in which the polycarboxylic acid comprises at least two carboxylic radicals separated from each other by not more that two carbon atoms, carbonates, bicarbonates, borates, phosphates, silicates and mixtures of any of the foregoing.
- the carboxylate or polycarboxylate builder can be momomeric or oligomeric in type although monomeric polycarboxylates are generally preferred for reasons of cost and performance.
- Suitable carboxylates containing one carboxy group include the water soluble salts of lactic acid, glycolic acid and ether derivatives thereof.
- Polycarboxylates containing two carboxy groups include the water-soluble salts of succinic acid, malonic acid, (ethylenedioxy) diacetic acid, maleic acid, diglycolic acid, tartaric acid, tartronic acid and fumaric acid, as well as the ether carboxylates and the sulfinyl carboxylates.
- Polycarboxylates containing three carboxy groups include, in particular, water-soluble citrates, aconitrates and citraconates as well as succinate derivatives such as the carboxymethyloxysuccinates described in British Patent No.
- Polycarboxylates containing four carboxy groups include oxydisuccinates disclosed in British Patent No. 1,261,829, 1,1,2,2-ethane tetracarboxylates, 1,1,3,3-propane tetracarboxylates and 1,1,2,3-propane tetracarboxylates.
- Polycarboxylates containing sulfo substituents include the sulfosuccinate derivatives disclosed in British Patent Nos. 1,398,421 and 1,398,422 and in U.S. Patent No. 3,936,448, and the sulfonated pyrolysed citrates described in British Patent No. 1,439,000.
- Alicyclic and heterocyclic polycarboxylates include cyclopentanecis,cis,cis-tetracarboxylates, cyclopentadienide pentacarboxylates, 2,3,4,5-tetrahydrofuran - cis, cis, cis-tetracarboxylates, 2,5-tetrahydrofuran - cisdicarboxylates, 2,2,5,5-tetrahydrofuran - tetracarboxylates, 1,2,3,4,5,6-hexane - hexacarboxylates and carboxymethyl derivatives of polyhydric alcohols such as sorbitol, mannitol and xylitol.
- Aromatic polycarboxylates include mellitic acid, pyromellitic acid and the phthalic acid derivatives disclosed in British Patent No. 1,425,343.
- the preferred polycarboxylates are hydroxycarboxylates containing up to three carboxy groups per molecule, more particularly citrates.
- the parent acids of the monomeric or oligomeric polycarboxylate chelating agents or mixtures thereof with their salts e.g. citric acid or citrate/citric acid mixtures are also contemplated as useful builder components.
- Borate builders, as well as builders containing borate-forming materials that can produce borate under detergent storage or wash conditions can also be used but are not preferred at wash conditions less that about 50°C, especially less than about 40°C.
- carbonate builders are the alkaline earth and alkali metal carbonates, including sodium carbonate and sesqui-carbonate and mixtures thereof with ultra-fine calcium carbonate as disclosed in German Patent Application No. 2,321,001 published on November 15, 1973.
- water-soluble phosphate builders are the alkali metal tripolyphosphates, sodium, potassium and ammonium pyrophosphate, sodium and potassium and ammonium pyrophosphate, sodium and potassium orthophosphate, sodium polymeta/phosphate in which the degree of polymerization ranges from about 6 to 21, and salts of phytic acid.
- Suitable silicates include the water soluble sodium silicates with an Si02: Na20 ratio of from 1.0 to 2.8, with ratios of from 1.6 to 2.4 being preferred, and 2.0 ratio being most preferred.
- the silicates may be in the form of either the anhydrous salt or a hydrated salt.
- Sodium silicate with an SiO2: Na20 ratio of 2.0 is the most preferred silicate.
- Silicates are preferably present in the detergent compositions in accord with the invention at a level of from 5% to 50% by weight of the composition, more preferably from 10% to 40% by weight.
- Partially soluble or insoluble builder compound Partially soluble or insoluble builder compound
- compositions of the present invention may contain a partially soluble or insoluble builder compound, typically present at a level of from 1% to 80% by weight, preferably from 10% to 70% by weight, most preferably from 20% to 60% weight of the composition.
- Examples of partially water soluble builders include the crystalline layered silicates.
- Examples of largely water insoluble builders include the sodium aluminosilicates.
- Crystalline layered sodium silicates have the general formula NaMSi x 0 2x+1 .yH20 wherein M is sodium or hydrogen, x is a number from 1.9 to 4 and y is a number from 0 to 20. Crystalline layered sodium silicates of this type are disclosed in EP-A-0164514 and methods for their preparation are disclosed in DE-A-3417649 and DE-A-3742043.
- x in the general formula above has a value of 2, 3 or 4 and is preferably 2.
- the most preferred material is ⁇ -Na2Si205, available from Hoechst AG as NaSKS-6.
- the crystalline layered sodium silicate material is preferably present in granular detergent compositions as a particulate in intimate admixture with a solid, water-soluble ionisable material.
- the solid, water-soluble ionisable material is selected from organic acids, organic and inorganic acid salts and mixtures thereof.
- Suitable aluminosilicate zeolites have the unit cell formula Na z [(AlO2) z (SiO2)y]. XH2O wherein z and y are at least 6; the molar ratio of z to y is from 1.0 to 0.5 and x is at least 5, preferably from 7.5 to 276, more preferably from 10 to 264.
- the aluminosilicate material are in hydrated form and are preferably crystalline, containing from 10% to 28%, more preferably from 18% to 22% water in bound form.
- the aluminosilicate ion exchange materials can be naturally occurring materials, but are preferably synthetically derived. Synthetic crystalline aluminosilicate ion exchange materials are available under the designations Zeolite A, Zeolite B, Zeolite P, Zeolite X, Zeoilte MAP, Zeolite HS and mixtures thereof. Zeolite A has the formula Na 12 [AlO2) 12 (SiO2)12]. xH2O wherein x is from 20 to 30, especially 27. Zeolite X has the formula Na86 [(AlO2)86(SiO2)106]. 276 H2O.
- a highly preferred component of the compositions of the invention is a surfactant system comprising surfactant selected from anionic, cationic, nonionic ampholytic and zwitterionic surfactants and mixtures thereof.
- the surfactant system comprises low foaming surfactant, especially low foaming nonionic surfactant.
- the surfactant system is preferably present at a level of from 0.25% to 30% by weight, more preferably 0.5% to 10% by weight, most preferably from 1% to 5% by weight of the compositions.
- Sulphonate and sulphate surfactants are useful herein.
- Sulphonates include alkyl benzene sulphonates having from 5 to 15 carbon atoms in the alkyl radical, and alpha-sulphonated methyl fatty acid testers in which the fatty acid is derived from a C6-C18 fatty source.
- Preferred sulphate surfactants are alkyl sulphates having from 6 to 16, preferably 6 to 10 carbon atoms in the alkyl radical.
- Useful surfactant system comprises a mixture of two alkyl sulphate materials whose respective mean chain lengths differ from each other.
- the cation in each instance is again an alkali metal, preferably sodium.
- the alkyl sulfate salts may be derived from natural or synthetic hydrocarbon sources.
- the C6-C16 alkyl ethoxysulfate salt comprises a primary alkyl ethoxysulfate which is derived from the condensation product of a C6-C16 alcohol condensed with an average of from one to seven ethylene oxide groups, per mole.
- alkali metal sarcosinates of formula R-CON (R1) CH2 COOM wherein R is a C5-C17 linear or branched alkyl or alkenyl group, R1 is a C1-C4 alkyl group and M is an alkali metal ion.
- R is a C5-C17 linear or branched alkyl or alkenyl group
- R1 is a C1-C4 alkyl group
- M is an alkali metal ion.
- Preferred examples are the lauroyl, Cocoyl (C12-C14), myristyl and oleyl methyl sarcosinates in the form of their sodium salts.
- alkyl ester sulfonate surfactants which include linear esters of C8-C20 carboxylic acids (i.e., fatty acids) which are sulfonated with gaseous SO3 according to "The Journal of the American Oil Chemists Society," 52 (1975), pp. 323-329.
- Suitable starting materials would include natural fatty substances as derived from tallow, palm oil, etc.
- the preferred alkyl ester sulfonate surfactants have the structural formula: wherein R3 is a C8-C20 hydrocarbyl, preferably an alkyl, or combination thereof, R4 is a C1-C6 hydrocarbyl, preferably an alkyl, or combination thereof, and M is a cation which forms a water soluble salt with the alkyl ester sulfonate.
- Suitable salt-forming cations include metals such as sodium, potassium, and lithium, and substituted or unsubstituted ammonium cations, such as monoethanolamine, diethanolamine, and triethanolamine.
- R3 is C10-C16 alkyl
- R4 is methyl, ethyl or isopropyl.
- the methyl ester sulfonates wherein R3 is C10-C16 alkyl.
- One preferred class of low foaming nonionic surfactants useful in the present invention comprises the water soluble ethoxylated C6-C16 fatty alcohols and C6-C16 mixed ethoxylated/propoxylated fatty alcohols and mixtures thereof.
- the ethoxylated fatty alcohols are the C10-C16 ethoxylated fatty alcohols with a degree of ethoxylation of from 3 to 50, most preferably these are the C12-C16 ethoxylated fatty alcohols with a degree of ethoxylation from 3 to 40.
- the mixed ethoxylated/propoxylated fatty alcohols have an alkyl chain length of from 10 to 16 carbon atoms, a degree of ethoxylation of from 3 to 30 and a degree of propoxylation of from 1 to 10.
- C6-C16 alcohol itself can be obtained from natural or synthetic sources.
- C6-C16 alcohols derived from natural fats, or Ziegler olefin build-up, or OXO synthesis can form suitable sources for the alkyl group.
- Examples of synthetically derived materials include Dobanol 25 (RTM) sold by Shell Chemicals (UK) Ltd which is a blend of C12-C15 alcohols, Ethyl 24 sold by the Ethyl Corporation which is a blend of C12-C15 alcohols, and a blend of C13-C15 alcohols in the ratio 67% C13, 33% C15 sold under the trade name Lutensol by BASF GmbH and Synperonic (RTM) by ICI Ltd., and Lial 125 sold by Liquichimica Italiana.
- Examples of naturally occuring materials from which the alcohols can be derived are coconut oil and palm kernel oil and the corresponding fatty acids.
- Nonionic surfactants comprises alkyl polyglucoside compounds of general formula RO (C n H 2n O) t Z x wherein Z is a moiety derived from glucose; R is a saturated hydrophobic alkyl group that contains from 6 to 16 carbon atoms preferably from 6 to 14 carbon atoms; t is from 0 to 10 and n is 2 or 3; x is from 1.1 to 4, the compounds including less than 10% unreacted fatty alcohol and less than 50% short chain alkyl polyglucosides.
- RO C n H 2n O
- t Z x alkyl polyglucoside compounds of general formula RO (C n H 2n O) t Z x wherein Z is a moiety derived from glucose; R is a saturated hydrophobic alkyl group that contains from 6 to 16 carbon atoms preferably from 6 to 14 carbon atoms; t is from 0 to 10 and n is 2 or 3; x is from 1.1 to 4, the compounds including less than 10% unreacted fatty
- Another preferred nonionic surfactant is a polyhydroxy fatty acid amide surfactant compound having the structural formula: wherein R1 is H, C1-C4 hydrocarbyl, 2-hydroxy ethyl, 2-hydroxy propyl, or a mixture thereof, preferably C1-C4 alkyl, more preferably C1 or C2 alkyl, most preferably C1 alkyl (ie., methyl); and R2 is a C5-C15 hydrocarbyl, preferably straight chain C5-C13 alkyl or alkenyl, more preferably straight chain C5-C11 alkyl or alkenyl, most preferably straight chain C5-C9 alkyl or alkenyl, or mixture thereof: and Z is a polyhydroxyhydrocarbyl having linear hydrocarbyl chain with at least 3 hydroxyls directly connected to the chain, or an alkoxlylated derivative (preferably ethoxylated or propoxylated) thereof.
- Z preferably will be derived from a reducing sugar in a reductive amination reaction; more preferably Z is a glycityl.
- Suitable reducing sugars include glucose, fructose, maltose, lactose, galactose, mannose, and xylose.
- high dextrose corn syrup, high fructose corn syrup, and high maltose corn syrup can be utilized as well as the individual sugars listed above. These corn syrups may yield a mix of sugar components for Z. It should be understood that it is by no means intended to exclude other suitable raw materials.
- Z preferably will be selected from the group consisting of -CH2-(CHOH) n -CH2OH, -CH(CH2OH)-(CHOH) n-1 -CH2OH, -CH2-(CHOH)2(CHOR')(CHOH)-CH2OH, where n is an integer from 3 to 5, inclusive, and R' is H or a cyclic or aliphatic monosaccharide, and alkoxylated derivatives thereof. Most preferred are glycityls wherein n is 4, particularly - CH2-(CHOH)4-CH2OH.
- R1 can be, for example, N-methyl, N-ethyl, N-propyl, N-isopropyl, N-butyl, N-2-hydroxy ethyl, or N-2-hydroxy propyl.
- R2-CO-N ⁇ can be, for example, cocamide, stearamide, oleamide, lauramide, myristamide, capricamide, palmitamide, or tallowamide.
- Z can be 1-deoxyglucityl, 2-deoxyfrucittyl, 1-deoxymaltityl, 1-deoxylactityl, 1-deoxygalactityl or 1-deoxymannityl, or 1-deoxymaltotriotityl.
- Preferred compounds are N-methyl N-1deoxyglucityl C14-C18 fatty acid amides.
- a further class of surfactants are the semi-polar surfactants such as amine oxides.
- Suitable amine oxides are selected from mono C6-C20, preferably C6-C10 N-alkyl or alkenyl amine oxides and propylene-1,3-diamine dioxides wherein the remaining N positions are substituted by methyl, hydroxyethyl or hydroxpropyl groups.
- Cationic surfactants can also be used in the detergent compositions herein and suitable quaternary ammonium surfactants are selected from mono C6-C16, preferably C6-C10 N-alkyl or alkenyl ammonium surfactants wherein remaining N positions are substituted by methyl, hydroxyethyl or hydroxypropyl groups.
- compositions may contain narrowly defined levels of a heavy metal ion sequestrant which is not in the form of a Mn(II) compound, such levels being in the range 0.005% to 3%, preferably 0.01 to 1%, most preferably 0.05% to 0.8%, by weight of the total composition.
- Heavy metal ion sequestrants which are acidic in nature, having for example phosphonic acid or carboxylic acid functionalities, may be present either in their acid form or as a complex/salt with a suitable non Mn(II) counter cation such as an alkali or alkaline metal ion, ammonium, or substituted ammonium ion, or any mixtures thereof.
- a suitable non Mn(II) counter cation such as an alkali or alkaline metal ion, ammonium, or substituted ammonium ion, or any mixtures thereof.
- the molar ratio of said counter cation to the heavy metal ion sequestrant is preferably at least 1:1.
- the heavy metal ion sequestrant may be any of those described as suitable heavy metal ion sequestrant-type organic ligands in the Mn(II) compounds section of this descrption.
- EDDS ethylenediamine-N,N'-disuccinic acid
- Preferred EDDS compounds are the free acid form and the sodium or magnesium salt or complex thereof.
- Examples of such preferred sodium salts of EDDS include Na2EDDS and Na3EDDS.
- Examples of such preferred magnesium complexes of EDDS include MgEDDS and Mg2EDDS.
- the heavy metal ion sequestrant herein can consist of a mixture of the above described species.
- Chlorine bleaches include the alkali metal hypochlorites and chlorinated cyanuric acid salts.
- the use of chlorine bleaches in the composition of the invention is optional and preferably minimized, and more preferably the present compositions contain no chlorine bleach.
- Another optional ingredient useful in the compositions is one or more additional enzymes.
- Preferred additional enzymatic materials include the commercially available lipases, amylases, neutral and alkaline proteases, esterases, cellulases, pectinases, lactases and peroxidases conventionally incorporated into detergent compositions. Suitable enzymes are discussed in US Patents 3,519,570 and 3,533,139.
- protease enzymes include those sold under the tradenames Alcalase, Savinase, Primase, Durazym, and Esperase by Novo Industries A/S (Denmark), those sold under the tradename Maxatase, Maxacal and Maxapem by Gist-Brocades, those sold by Genencor International, and those sold under the tradename Opticlean and Optimase by Solvay Enzymes.
- Protease enzyme may be incorporated into the compositions in accordance with the invention at a level of from 0.0001% to 4% active enzyme by weight of the composition.
- Preferred amylases include, for example, ⁇ -amylases obtained from a special strain of B licheniformis, described in more detail in GB-1,269,839 (Novo).
- Preferred commercially available amylases include for example, those sold under the tradename Rapidase by Gist-Brocades, and those sold under the tradename Termamyl and BAN by Novo Industries A/S.
- Amylase enzyme may be incorporated into the composition in accordance with the invention at a level of from 0.0001% to 2% active enzyme by weight of the composition.
- Lipolytic enzyme may be present at levels of active lipolytic enzyme of from 0.0001% to 2% by weight, preferably 0.001% to 1% by weight, most preferably from 0.001% to 0.5% by weight of the compositions.
- the lipase may be fungal or bacterial in origin being obtained, for example, from a lipase producing strain of Humicola sp., T hermomyces sp. or Pseudomonas sp. including Pseudomonas pseudoalcaligenes or Pseudomas fluorescens. Lipase from chemically or genetically modified mutants of these strains are also useful herein.
- a preferred lipase is derived from Pseudomonas pseudoalcaligenes , which is described in Granted European Patent, EP-B-0218272.
- Another preferred lipase herein is obtained by cloning the gene from Humicola lanuginosa and expressing the gene in Aspergillus oryza , as host, as described in European Patent Application, EP-A-0258 068, which is commercially available from Novo Industri A/S, Bagsvaerd, Denmark, under the trade name Lipolase. This lipase is also described in U.S. Patent 4,810,414, Huge-Jensen et al, issued March 7, 1989.
- Preferred enzyme-containing compositions herein may comprise from about 0.001% to about 10%, preferably from about 0.005% to about 8%,most preferably from about 0.01% to about 6%, by weight of an enzyme stabilizing system.
- the enzyme stabilizing system can be any stabilizing system which is compatible with the detersive enzyme.
- Such stabilizing systems can comprise calcium ion, boric acid, propylene glycol, short chain carboxylic acid, boronic acid, and mixtures thereof.
- Such stabilizing systems can also comprise reversible enzyme inhibitors, such as reversible protease inhibitors.
- compositions herein may further comprise from 0 to about 10%, preferably from about 0.01% to about 6% by weight, of chlorine bleach scavengers, added to prevent chlorine bleach species present in many water supplies from attacking and inactivating the enzymes, especially under alkaline conditions.
- chlorine bleach scavengers While chlorine levels in water may be small, typically in the range from about 0.5 ppm to about 1.75 ppm, the available chlorine in the total volume of water that comes in contact with the enzyme during washing is usually large; accordingly, enzyme stability in-use can be problematic.
- Suitable chlorine scavenger anions are widely available, and are illustrated by salts containing ammonium cations or sulfite, bisulfite, thiosulfite, thiosulfate, iodide, etc.
- Antioxidants such as carbamate, ascorbate, etc., organic amines such as ethylenediaminetetracetic acid (EDTA) or alkali metal salt thereof, monoethanolamine (MEA), and mixtures thereof can likewise be used.
- EDTA ethylenediaminetetracetic acid
- MEA monoethanolamine
- scavengers such as bisulfate, nitrate, chloride, sources of hydrogen peroxide such as sodium perborate tetrahydrate, sodium perborate monohydrate and sodium percarbonate, as well as phosphate, condensed phosphate, acetate, benzoate, citrate, formate, lactate, malate, tartrate, salicylate, etc. and mixtures thereof can be used if desired.
- Organic polymeric compounds are particularly preferred components of the compositions in accord with the invention.
- organic polymeric compound it is meant essentially any polymeric organic compound commonly used as dispersants, and anti-redeposition and soil suspension agents in detergent compositions.
- Organic polymeric compound is typically incorporated in the detergent compositions of the invention at a level of from 0.1% to 30%, preferably from 0.5% to 15%, most preferably from 1% to 10% by weight of the compositions.
- organic polymeric compounds include the water soluble organic homo- or co-polymeric polycarboxylic acids or their salts in which the polycarboxylic acid comprises at least two carboxyl radicals separated from each other by not more than two carbon atoms.
- Polymers of the latter type are disclosed in GB-A-1,596,756.
- salts are polyacrylates of molecular weight 2000-10000 and their copolymers with any suitable other monomer units including modified acrylic, fumaric, maleic, itaconic, aconitic, mesaconic, citraconic and methylenemalonic acid or their salts, maleic anhydride, acrylamide, alkylene, vinylmethyl ether, styrene and any mixtures thereof.
- Preferred are the copolymers of acrylic acid and maleic anhydride having a molecular weight of from 20,000 to 100,000.
- Preferred commercially available acrylic acid containing polymers having a molecular weight below 15,000 include those sold under the tradename Sokalan PA30, PA20, PA15, PA10 and Sokalan CP10 by BASF GmbH, and those sold under the tradename Acusol 45N by Rohm and Haas.
- Preferred acrylic acid containing copolymers include those which contain as monomer units: a) from 90% to 10%, preferably from 80% to 20% by weight acrylic acid or its salts and b) from 10% to 90%, preferably from 20% to 80% by weight of a substituted acrylic monomer or its salts having the general formula -[CR2-CR1(CO-O-R3)]- wherein at least one of the substituents R1, R2 or R3, preferably R1 or R2 is a 1 to 4 carbon alkyl or hydroxyalkyl group, R1 or R2 can be a hydrogen and R3 can be a hydrogen or alkali metal salt.
- R1 is methyl
- R2 is hydrogen (i.e. a methacrylic acid monomer).
- the most preferred copolymer of this type has a molecular weight of 3500 and contains 60% to 80% by weight of acrylic acid and 40% to 20% by weight of methacrylic acid.
- suitable polyacrylate/modified polyacrylate copolymers include those copolymers of unsaturated aliphatic carboxylic acids disclosed in U.S. Patents No.s 4,530,766, and 5,084,535 which have a molecular weight of less than 15,000.
- Suitable organic polymeric compounds include the copolymers of acrylamide and acrylate having a molecular weight of from 3,000 to 100,000, and the acrylate/fumarate copolymers having a molecular weight of from 2,000 to 80,000.
- polyamino compounds are useful herein including those derived from aspartic acid such as those disclosed in EP-A-305282, EP-A-305283 and EP-A-351629.
- organic polymeric compounds suitable for incorporation in the detergent compositions herein include cellulose derivatives such as methylcellulose, carboxymethylcellulose and hydroxyethylcellulose.
- organic polymeric compounds are the polyethylene glycols, particularly those of molecular weight 1000-10000, more particularly 2000 to 8000 and most preferably about 4000.
- compositions of the invention may contain a lime soap dispersant compound, which has a lime soap dispersing power (LSDP), as defined hereinafter of no more than 8, preferably no more than 7, most preferably no more than 6.
- LSDP lime soap dispersing power
- the lime soap dispersant compound is preferably present at a level of from 0.1% to 40% by weight, more preferably 1% to 20% by weight, most preferably from 2% to 10% by weight of the compositions.
- a lime soap dispersant is a material that prevents the precipitation of alkali metal, ammonium or amine salts of fatty acids by calcium or magnesium ions.
- a numerical measure of the effectiveness of a lime soap dispersant is given by the lime soap dispersing power (LSDP) which is determined using the lime soap dispersion test as described in an article by H.C. Borghetty and C.A. Bergman, J. Am. Oil. Chem. Soc., volume 27, pages 88-90, (1950).
- This lime soap dispersion test method is widely used by practitioners in this art field being referred to, for example, in the following review articles; W.N. Linfield, Surfactant Science Series, Volume 7, p3; W.N. Linfield, Tenside Surf.
- Surfactants having good lime soap dispersant capability will include certain amine oxides, betaines, sulfobetaines, alkyl ethoxysulfates and ethoxylated alcohols.
- Polymeric lime soap dispersants suitable for use herein are described in the article by M.K. Nagarajan and W.F. Masler, to be found in Cosmetics and Toiletries, Volume 104, pages 71-73, (1989).
- Examples of such polymeric lime soap dispersants include certain water-soluble salts of copolymers of acrylic acid, methacrylic acid or mixtures thereof, and an acrylamide or substituted acrylamide, where such polymers typically have a molecular weight of from 5,000 to 20,000.
- compositions of the invention when formulated for use in machine washing compositions, preferably comprise a suds suppressing system present at a level of from 0.01% to 15%, preferably from 0.05% to 10%, most preferably from 0.1% to 5% by weight of the composition.
- Suitable suds suppressing systems for use herein may comprise essentially any known antifoam compound, including, for example silicone antifoam compounds, 2-alkyl and alcanol antifoam compounds.
- antifoam compound any compound or mixtures of compounds which act such as to depress the foaming or sudsing produced by a solution of a detergent composition, particularly in the presence of agitation of that solution.
- Particularly preferred antifoam compounds for use herein are silicone antifoam compounds defined herein as any antifoam compound including a silicone component. Such silicone antifoam compounds also typically contain a silica component.
- silicone antifoam compounds as used herein, and in general throughout the industry, encompasses a variety of relatively high molecular weight polymers containing siloxane units and hydrocarbyl group of various types.
- Preferred silicone antifoam compounds are the siloxanes, particularly the polydimethylsiloxanes having trimethylsilyl end blocking units.
- Suitable antifoam compounds include the monocarboxylic fatty acids and soluble salts thereof. These materials are described in US Patent 2,954,347, issued September 27, 1960 to Wayne St. John.
- the monocarboxylic fatty acids, and salts thereof, for use as suds suppressor typically have hydrocarbyl chains of 10 to about 24 carbon atoms, preferably 12 to 18 carbon atoms.
- Suitable salts include the alkali metal salts such as sodium, potassium, and lithium salts, and ammonium and alkanolammonium salts.
- Suitable antifoam compounds include, for example, high molecular weight fatty esters (e.g. fatty acid triglycerides), fatty acid esters of monovalent alcohols, aliphatic C18-C40 ketones (e.g. stearone) N-alkylated amino triazines such as tri- to hexa-alkylmelamines or di- to tetra alkyldiamine chlortriazines formed as products of cyanuric chloride with two or three moles of a primary or secondary amine containing 1 to 24 carbon atoms, propylene oxide, bis stearic acid amide and monostearyl dialkali metal (e.g. sodium, potassium, lithium) phosphates and phosphate esters.
- high molecular weight fatty esters e.g. fatty acid triglycerides
- fatty acid esters of monovalent alcohols e.g. fatty acid esters of monovalent alcohols
- Copolymers of ethylene oxide and propylene oxide particularly the mixed ethoxylated/propoxylated fatty alcohols with an alkyl chain length of from 10 to 16 carbon atoms, a degree of ethoxylation of from 3 to 30 and a degree of propoxylation of from 1 to 10, are also suitable antifoam compounds for use herein.
- 2-alky-alcanols antifoam compounds for use herein have been described in DE 40 21 265.
- the 2-alkyl-alcanols suitable for use herein consist of a C6 to C16 alkyl chain carrying a terminal hydroxy group, and said alkyl chain is substituted in the a position by a C1 to C10 alkyl chain.
- Mixtures of 2-alkyl-alcanols can be used in the compositions according to the present invention.
- a preferred suds suppressing system comprises (a) antifoam compound, preferably silicone antifoam compound, most preferably a silicone antifoam compound comprising in combination
- a preferred particulate suds suppressor system useful herein comprises a mixture of an alkylated siloxane of the type hereinabove disclosed and solid silica.
- the solid silica can be a fumed silica, a precipitated silica or a silica, made by the gel formation technique.
- the silica particles suitable have an average particle size of from 0.1 to 50 micrometers, preferably from 1 to 20 micrometers and a surface area of at least 50m2/g.
- These silica particles can be rendered hydrophobic by treating them with dialkylsilyl groups and/or trialkylsilyl groups either bonded directly onto the silica or by means of a silicone resin. It is preferred to employ a silica the particles of which have been rendered hydrophobic with dimethyl and/or trimethyl silyl groups.
- a preferred particulate antifoam compound for inclusion in the detergent compositions in accordance with the invention suitably contain an amount of silica such that the weight ratio of silica to silicone lies in the range from 1:100 to 3:10, preferably from 1:50 to 1:7.
- Another suitable particulate suds suppressing system is represented by a hydrophobic silanated (most preferably trimethyl-silanated) silica having a particle size in the range from 10 nanometers to 20 nanometers and a specific surface area above 50m2/g, intimately admixed with dimethyl silicone fluid having a molecular weight in the range from about 500 to about 200,000 at a weight ratio of silicone to silanated silica of from about 1:1 to about 1:2.
- a hydrophobic silanated (most preferably trimethyl-silanated) silica having a particle size in the range from 10 nanometers to 20 nanometers and a specific surface area above 50m2/g, intimately admixed with dimethyl silicone fluid having a molecular weight in the range from about 500 to about 200,000 at a weight ratio of silicone to silanated silica of from about 1:1 to about 1:2.
- a highly preferred particulate suds suppressing system is described in EP-A-0210731 and comprises a silicone antifoam compound and an organic carrier material having a melting point in the range 50°C to 85°C, wherein the organic carrier material comprises a monoester of glycerol and a fatty acid having a carbon chain containing from 12 to 20 carbon atoms.
- EP-A-0210721 discloses other preferred particulate suds suppressing systems wherein the organic carrier material is a fatty acid or alcohol having a carbon chain containing from 12 to 20 carbon atoms, or a mixture thereof, with a melting point of from 45°C to 80°C.
- An exemplary particulate suds suppressing system for use herein is a particulate agglomerate component, made by an agglomeration process, comprising in combination
- compositions herein may also comprise from 0.01% to 10 %, preferably from 0.05% to 0.5% by weight of polymeric dye transfer inhibiting agents.
- the polymeric dye transfer inhibiting agents are preferably selected from polyamine N-oxide polymers, copolymers of N-vinylpyrrolidone and N-vinylimidazole, polyvinylpyrrolidonepolymers or combinations thereof.
- compositions may contain, but preferably do not contain, additional corrosion inhibitor inhibitors such as benzotriazole and paraffin.
- compositions of the invention can be formulated in any desirable form such as powders, granulates, pastes, liquids, gels and tablets, granular forms being preferred.
- the bulk density of the granular detergent compositions in accordance with the present invention is typically of at least 650 g/litre, more usually at least 700 g/litre and more preferably from 800 g/litre to 1200 g/litre.
- Bulk density is measured by means of a simple funnel and cup device consisting of a conical funnel moulded rigidly on a base and provided with a flap valve at its lower extremity to allow the contents of the funnel to be emptied into an axially aligned cylindrial cup disposed below the funnel.
- the funnel is 130 mm and 40 mm at its respective upper and lower extremities. It is mounted so that the lower extremity is 140 mm above the upper surface of the base.
- the cup has an overall height of 90 mm, an internal height of 87 mm and an internal diameter of 84 mm. Its nominal volume is 500 ml.
- the funnel is filled with powder by hand pouring, the flap valve is opened and powder allowed to overfill the cup.
- the filled cup is removed from the frame and excess powder removed from the cup by passing a straight edged implement e.g. a knife, across its upper edge.
- the filled cup is then weighed and the value obtained for the weight of powder doubled to provide the bulk density in g/litre. Replicate measurements are made as required.
- the particle size of the components of granular compositions in accordance with the invention should preferably be such that no more that 5% of particles are greater than 1.4mm in diameter and not more than 5% of particles are less than 0.15mm in diameter.
- the liquid should be thixotropic (ie; exhibit high viscosity when subjected to low stress and lower viscosity when subjected to high stress), or at least have very high viscosity, for example, of from 1,000 to 10,000,000 centipoise.
- a viscosity control agent or a thixotropic agent to provide a suitable liquid product form.
- Suitable thixotropic or viscosity control agents include methyl cellulose, carboxymethylcellulose, starch, polyvinyl, pyrrolidone, gelatin, colloidal silica, and natural or synthetic clay minerals.
- Pasty compositions in accordance with the invention generally have viscosities of about 5,000 centipoise and up to several hundred million centipoise.
- a small amount of a solvent or solubilizing agent or of a gel-forming agent can be included. Most commonly, water is used in this context and forms the continuous phase of a concentrated dispersion. Certain nonionic surfactants at high levels form a gel in the presence of small amount of water and other solvents. Such gelled compositions also envisaged in the present invention.
- the pH of a 1% solution of the present compositions is preferably less than 12, preferably from 9.8 to 11.5, most preferably from 10.0 to 11.0.
- the use is provided, in a method for washing soiled tableware, including soiled silverware, of an oxygen-releasing bleach system containing composition including Mn(II) compounds to inhibit the tarnishing of said silverware.
- Any suitable methods for washing or cleaning soiled tableware, including soiled silverware, are envisaged including machine dishwashing methods.
- a preferred machine dishwashing method comprises treating soiled articles selected from crockery, glassware, hollowware and cutlery and mixtures thereof, with an aqueous liquid having dissolved or dispensed therein an effective amount of a machine dishwashing composition in accord with the inevntion.
- an effective amount of the machine dishwashing composition it is meant from 8g to 60g of product dissolved or dispersed in a wash solution of volume from 3 to 10 litres, as are typical product dosages and wash solution volumes commonly employed in conventional machine dishwashing methods.
- the following base machine dishwashing detergent compositions were prepared (parts by weight).
- base composition A of Example I was compared for anti-silver tarnishing performance to reference Compositions G and H having the base Composition A but additionally containing 0.13% MnSO4 and 0.13% Mn(II)EDDS respectively.
- the testing involved machine testing, using a Bosch Siemens dishwasher, 20g product dosage, 65°C economy cycles, and 14 cycles.
- Results were as follows : (average of the 4 gradings from the panellists) Composition A Composition G Composition H 2.0 ⁇ 0.5 ⁇ 0.5
- machine dishwashing detergents according to the invention are prepared (parts by weight):
Landscapes
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Wood Science & Technology (AREA)
- Organic Chemistry (AREA)
- Inorganic Chemistry (AREA)
- Detergent Compositions (AREA)
Abstract
There is provided a bleaching composition providing enhanced anti-silver tarnishing properties containing an oxygen-releasing bleach system comprising a hydrogen peroxide source and a peroxyacid bleach precursor compound selected from the group consisting of
- (i) an N-acylated lactam bleach precursor
- (ii) perbenzoic acid precursor
- (iii) perbenzoic acid derivative precursor
- (iv) cationic peroxyacid precursor
and any combinations of (i) - (iv); and
(b) an Mn(II) compound.
Description
- The present invention relates to the inclusion of Mn(II) compounds in bleaching compositions, suitable for use in machine dishwashing. The compositions exhibit good bleachable stain removal and enhanced anti silver-tarnishing properties.
- Compositions designed for use in automatic dishwasher machines are well known, and a consistent effort has been made by detergent manufacturers to improve the cleaning and/or rinsing efficiency of said compositions on dishes and glassware, as reflected by numerous patent publications.
- The present invention is concerned with the silver-tarnishing problem encountered when compositions which contain oxygen-bleaching species are employed in machine dishwashing methods.
- The satisfactory removal of bleachable soils such as tea and coloured vegetable soils, such as carotenoid soils is a particular challenge to the formulator of a machine dishwashing detergent composition. Traditionally, the removal of such soils has been enabled by the use of bleach components such as oxygen and chlorine bleaches. Oxygen bleaching species are these days however, largely preferred over chlorine bleaches.
- A problem encountered with the use of such oxygen bleaches is the tarnishing of any silverware components of the washload. The level of tarnishing observed can range from slight discolouration of the silverware to the formation of a dense black coating on the surface of the silverware.
- The formulator thus faces the dual challenge of formulating a product which maximises bleachable soil cleaning but minimises the occurrence of tarnishing of silverware components of the washload.
- The Applicants have found that silver tarnishing can be a particular problem when the bleach system comprises a combination of an inorganic perhydrate bleach, especially a percarbonate bleach, and a peroxyacid bleach precursor selected from perbenzoic acid precursors, perbenzoic acid derivative precursors, N-acylated lactam precursors and cationic peroxyacid precursors.
- It has been found that enhanced anti-silver tarnishing as well as good cleaning performance can be achieved through the use of certain Mn(II) compounds, even in the absence of conventional corrosion inhibitors such as benzotriazole.
- To provide further reduction of silver tarnishing it has been found that the total level of oxygen bleach in the composition is desirably optimized, and preferably the rate of release of the oxygen bleach is controlled. The rate of release of oxygen bleach is preferably rapid enough to provide satisfactory cleaning, but with minimised tarnishing.
- It is an object of the present invention to provide compositions, suitable for use in machine dishwashing methods, having enhanced anti-silver tarnishing properties, as well as good cleaning performance, particularly bleachable soil removal performance.
- German patent DE-A-4,128,672 in the name of Henkel KgaA discloses strong alkaline, bleach-containing detergents described to have a reduced tendency to tarnish silverware. Compositions containing Mn(II) salts such as MnSO₄ and MnCl₂ in combination with inorganic perhydrate salts and certain bleach activators are disclosed. The combination of said Mn(II) salts or compounds with N-acylated lactam, perbenzoic acid, perbenzoic acid derivative or cationic peroxyacid precursors is not disclosed. Nor is there any recognition provided that such bleach precursors particularly give rise to the problem of silver tarnishing. Teaching is provided that where Mn(II) salts are used as activators for peroxy compounds there is no protection of the silver, in direct contrast to the present invention which recognizes the ability of certain Mn(II) salts to ameliorate the problem of silver tarnishing.
- European Patent Applications EP-A-0082563, EP-A-127,910, EP-A-141,470, EP-A-145,090 and EP-A-145,091 in the name of Unilever disclose detergent compositions for use in fabric washing containing various Mn(II) compounds. The combination of these various Mn(II) compounds with N-acylated lactam, perbenzoic acid, perbenzoic acid derivative or cationic peroxyacid precursors is not specifically disclosed. Problems specific to the art field of machine dishwashing, in particular to the tarnishing of silverware, are not addressed.
- European Patent Application EP-A-0 530 870 in the name of Unilever discloses machine dishwashing compositions containing a dinuclear manganese complex in which the manganese is in the III or IV oxidation state. No recognition is provided is this document of the utility of Mn(II) salts in reducing silver tarnishing.
- Applicant's copending US Patent Application No. 08/210186, from which this Application claims priority, discloses bleach compositions containing manganese ethylenediamine-N,N'-disuccinate.
- According to one aspect of the present invention there is provided a bleaching composition containing
- (a) an oxygen-releasing bleach system comprising a hydrogen peroxide source and a peroxyacid bleach precursor compound selected from the group consisting of
- (i) an N-acylated lactam bleach precursor of the formula:
- (ii) a perbenzoic acid precursor
- (iii) a perbenzoic acid derivative precursor
- (iv) a cationic peroxyacid precursor
and any combinations of (i) - (iv); and
- (i) an N-acylated lactam bleach precursor of the formula:
- (b) an Mn(II) compound
- Preferably, said oxygen-releasing bleaching system is incorporated such that the level of available oxygen measured according to the method herein is from 0.3% to 2.5%, preferably 0.5% to 1.7% by weight.
- Preferably, the rate of release of said available oxygen is such that the available oxygen is completely released from the composition in a time interval of from 3.5 minutes to 10.0 minutes, using the test protocol described in the present description.
- Preferably the bleaching composition contains no additional corrosion inhibitor component, particularly no benzotriazole.
- Preferably the compositions are adapted for use in a machine dishwashing method, in particular being low foaming in character. Typically the compositions contain, at most, low levels of low foaming surfactant which is often nonionic in character.
- According to another aspect of the invention there is provided the use, in a method for washing soiled tableware, including soiled silverware, of a composition containing an oxygen-releasing bleach system wherein said composition contains an Mn(II) compound to inhibit the tarnishing of said silverware.
- Preferably, the Mn(II) compound is an Mn(II) salt or complex with an organic ligand, which may itself have heavy metal ion sequestering or crystal growth inhibiting properties.
- The present compositions contain as essential components an oxygen-releasing bleach system and an Mn(II) compound. The level of available oxygen is preferably controlled. Preferably, the rate of release of available oxygen and of any organic peroxyacid bleaching species is also controlled.
- An essential feature of the invention is an oxygen-releasing bleaching system containing a hydrogen peroxide source and a peroxyacid bleach precursor compound. The production of the peroxyacid occurs by an in situ reaction of the precursor with a source of hydrogen peroxide. Suitable sources of hydrogen peroxide include inorganic perhydrate bleaches.
- The peroxyacid bleach precursor is most preferably selected from perbenzoic acid precursors, perbenzoic acid derivative precursors, N-acylated lactam bleach precursors and cationic peroxyacid precursors, or mixtures thereof.
- The peroxyacid bleach precursors are preferably incorporated at a level of from 0.5% to 20% by weight, more preferably from 12% to 15% by weight, most preferably from 1.5% to 10% by weight of the compositions.
- N-acylated precursor compounds of the lactam class are disclosed generally in GB-A-855735. Whilst the broadest aspect of the invention contemplates the use of any lactam useful as a peroxyacid precursor, preferred materials comprise the caprolactams and valerolactams.
-
-
- Suitable valero lactams have the formula:
wherein R¹ is H or an alkyl, aryl, alkoxyaryl or alkaryl group containing from 1 to 12 carbon atoms, preferably from 6 to 12 carbon atoms. In highly preferred embodiments, R¹ is selected from phenyl, heptyl, octyl, nonyl, 2,4,4-trimethylpentyl, decenyl and mixtures thereof. - The most preferred materials are those which are normally solid at <30°C, particularly the phenyl derivatives, ie. benzoyl valerolactam, benzoyl caprolactam and their substituted benzoyl analogues such as chloro, amino alkyl, alkyl, aryl and alkoxy derivatives.
- Caprolactam and valerolactam precursor materials wherein the R¹ moiety contains at least 6, preferably from 6 to 12, carbon atoms provide peroxyacids on perhydrolysis of a hydrophobic character which afford nucleophilic and body soil clean-up. Precursor compounds wherein R¹ comprises from 1 to 6 carbon atoms provide hydrophilic bleaching species which are particularly efficient for bleaching beverage stains. Mixtures of 'hydrophobic' and 'hydrophilic' caprolactams and valero lactams, typically at weight ratios of 1:5 to 5:1, preferably 1:1, can be used herein for mixed stain removal benefits.
- Highly preferred caprolactam and valerolactam precursors include benzoyl caprolactam, nonanoyl capro-lactam, benzoyl valerolactam, nonanoyl valerolactam, 3,5,5-trimethylhexanoyl caprolactam, 3,5,5-trimethylhexanoyl valerolactam, octanoyl caprolactam, octanoyl valerolactam, decanoyl caprolactam, decanoyl valerolactam, undecenoyl caprolactam, undecenoyl valerolactam, (6-octanamidocaproyl)oxybenzene-sulfonate, (6-nonanamidocaproyl)oxybenzenesulfonate, (6-decanamidocaproyl)oxybenzenesulfonate, and mixtures thereof. Examples of highly preferred substituted benzoyl lactams include methylbenzoyl caprolactam, methylbenzoyl valerolactam, ethylbenzoyl caprolactam, ethylbenzoyl valerolactam, propylbenzoyl caprolactam, propylbenzoyl valerolactam, isopropylbenzoyl caprolactam, isopropylbenzoyl valerolactam, butylbenzoyl caprolactam, butylbenzoyl valerolactam, tert-butylbenzoyl caprolactam, tert-butylbenzoyl valerolactam, pentylbenzoyl caprolactam, pentylbenzoyl valerolactam, hexylbenzoyl caprolactam, hexylbenzoyl valerolactam, ethoxybenzoyl caprolactam, ethoxybenzoyl valerolactam, propoxybenzoyl caprolactam, propoxybenzoyl valerolactam, isopropoxybenzoyl caprolactam, isopropoxybenzoyl valerolactam, butoxybenzoyl caprolactam, butoxybenzoyl valerolactam, tert-butoxybenzoyl caprolactam, tert-butoxybenzoyl valerolactam, pentoxybenzoyl caprolactam, pentoxybenzoyl valerolactam, hexoxybenzoyl caprolactam, hexoxybenzoyl valerolactam, 2,4,6-trichlorobenzoyl caprolactam, 2,4,6-trichlorobenzoyl valerolactam, pentafluorobenzoyl caprolactam, pentafluorobenzoyl valerolactam, dichlorobenzoyl caprolactam, dimethoxybenzoyl caprolactam, 4-chlorobenzoyl caprolactam, 2,4-dichlororbenzoyl caprolactam, terephthaloyl dicaprolactam, pentafluorobenzoyl caprolactam, pentafluorobenzoyl valerolactam, dichlorobenzoyl valerolactam, dimethoxybenzoyl valerolactam, 4-chlorobenzoyl valerolactam, 2,4-dichlororbenzoyl valerolactam, terephthaloyl divalerolactam, 4-nitrobenzoyl caprolactam, 4-nitrobenzoyl valerolactam, and mixtures thereof.
- Essentially any perbenzoic acid precursors are suitable herein, including those of the N-acylated lactam class, which are preferred.
- Specific O-acylated perbenzoic acid precursor compounds include benzoyl oxybenzene sulfonate and monobenzoyltetraacetyl glucose benzoyl peroxide.
- Preferred perbenzoic acid precursor compounds of the imide type include N-benzoyl succinimide, tetrabenzoyl ethylene diamine and the N-benzoyl substituted ureas.
- Suitable imidazoles include N-benzoyl imidazole and N-benzoyl benzimidazole and other useful N-acyl group-containing peroxyacid precursors include N-benzoyl pyrrolidone, dibenzoyl taurine and benzoyl pyroglutamic acid.
- Preferred perbenzoic acid precursors include the benzoyl diacyl peroxides and benzoyl tetraacyl peroxides.
- Another preferred class of perbenzoic acid precursor compounds are the amide substituted compounds of the following general formulae:
wherein R¹ is an aryl group with from 1 to 14 carbon atoms, R² is an alkylene, arylene, and alkarylene group containing from 1 to 14 carbon atoms, and R⁵ is H or an alkyl, aryl, or alkaryl group containing 1 to 10 carbon atoms and L can be essentially any leaving group. R¹ preferably contains from 6 to 12 carbon atoms. R² preferably contains from 4 to 8 carbon atoms. R¹ may be aryl, substituted aryl or alkylaryl containing branching, substitution, or both and may be sourced from either synthetic sources or natural sources including for example, tallow fat. Analogous structural variations are permissible for R². The substitution can include alkyl, aryl, halogen, nitrogen, sulphur and other typical substituent groups or organic compounds. R⁵ is preferably H or methyl. R¹ and R⁵ should not contain more than 18 carbon atoms in total. Amide substituted bleach activator compounds of this type are described in EP-A-0170386. - The L group must be sufficiently reactive for the reaction to occur within the optimum time frame (e.g., a wash cycle). However, if L is too reactive, this activator will be difficult to stabilize for use in a bleaching composition. These characteristics are generally paralleled by the pKa of the conjugate acid of the leaving group, although exceptions to this convention are known. Ordinarily, leaving groups that exhibit such behavior are those in which their conjugate acid has a pKa in the range of from 4 to 13, preferably from 6 to 11 and most preferably from 8 to 11.
- Preferred bleach precursors are those wherein R¹, R² and R⁵ are as defined for the amide substituted compounds and L is selected from the group consisting of:
and mixtures thereof, wherein R¹ is an alkyl, aryl, or alkaryl group containing from 1 to 14 carbon atoms, R³ is an alkyl chain containing from 1 to 8 carbon atoms, R⁴ is H or R³, and Y is H or a solubilizing group. - The preferred solubilizing groups are -SO₃⁻M⁺, -CO₂⁻M⁺, -SO₄⁻M⁺, -N⁺(R³)₄X⁻ and O<--N(R³)₃ and most preferably -SO₃⁻M⁺ and -CO₂⁻M⁺ wherein R³ is an alkyl chain containing from 1 to 4 carbon atoms, M is a cation which provides solubility to the bleach activator and X is an anion which provides solubility to the bleach activator. Preferably, M is an alkali metal, ammonium or substituted ammonium cation, with sodium and potassium being most preferred, and X is a halide, hydroxide, methylsulfate or acetate anion. It should be noted that bleach activators with a leaving group that does not contain a solubilizing groups should be well dispersed in the bleaching solution in order to assist in their dissolution.
- Other preferred perbenzoic acid precursors include:
- Benzoylation products of sorbitol, glucose, and all saccharides with benzoylating agents.
-
-
-
-
- Suitable perbenzoic acid derivative precursors include any of the herein disclosed perbenzoic precursors in which the perbenzoic group is substituted by essentially any functional group including alkyl groups.
- Cationic peroxyacid precursor compounds are also suitable herein. Typically such cationic peroxyacid precursors are formed by substituting the peroxyacid part with an ammonium or alkyl ammmonium group, preferably an ethyl or methyl ammonium group.
- Cationic peroxyacid precursors are described in U.S. Patents 4,904,406; 4,751,015; 4,988,451; 4,397,757; 5,269,962; 5,127,852; 5,093,022; 5,106,528; U.K. 1,382,594; EP 475,512, 458,396 and 284,292; and in JP 87-318,332.
- Examples of preferred cationic peroxyacid precursors are described in UK Patent Application No. 9407944.9 and copending US Patent Applications having the Procter & Gamble attorneys' docket No's 5410, 5411, 5412 and 5413.
- Suitable cationic peroxyacid precursors include any of the ammonium or alkyl ammonium substituted alkyl or benzoyl oxybenzene sulfonates, N-acylated caprolactams, and monobenzoyltetraacetyl glucose benzoyl peroxides.
-
- A preferred cationically substituted alkyl oxybenzene sulfonate is the methyl ammonium derivative of 2,3,3-tri-methyl hexanoyloxybenzene sulfonate.
-
- Another preferred cationic peroxyacid precursor is 2-(N,N,N-trimethyl ammonium) ethyl sodium 4-sulphophenyl carbonate chloride.
-
-
- The compositions in accord with the invention include a hydrogen peroxide source. Suitable hydrogen peroxide sources include the inorganic perhydrate salts.
- The inorganic perhydrate salts are normally incorporated in the form of the sodium salt at a level of from 1% to 40% by weight, more preferably from 2% to 30% by weight and most preferably from 5% to 25% by weight of the compositions.
- Examples of inorganic perhydrate salts include perborate, percarbonate, perphosphate, persulfate and persilicate salts. The inorganic perhydrate salts are normally the alkali metal salts. The inorganic perhydrate salt may be included as the crystalline solid without additional protection. For certain perhydrate salts however, the preferred executions of such granular compositions utilize a coated form of the material which provides better storage stability for the perhydrate salt in the granular product.
- Sodium perborate can be in the form of the monohydrate of nominal formula NaBO₂H₂O₂ or the tetrahydrate NaBO₂H₂O₂.3H₂O.
- Sodium percarbonate, which is a preferred perhydrate for inclusion in compositions in accordance with the invention, is an addition compound having a formula typically corresponding to 2Na₂CO₃.3H₂O₂, and is available commercially as a crystalline solid. The percarbonate is most preferably incorporated into such compositions in a coated form which provides in product stability.
- A suitable coating material providing in product stability comprises mixed salt of a water soluble alkali metal sulphate and carbonate. Such coatings together with coating processes have previously been described in GB-1,466,799, granted to Interox on 9th March 1977. The weight ratio of the mixed salt coating material to percarbonate lies in the range from 1 : 200 to 1: 4, more preferably from 1: 99 to 1: 9, and most preferably from 1: 49 to 1 : 19. Preferably, the mixed salt is of sodium sulphate and sodium carbonate which has the general formula Na₂SO₄.n.Na₂CO₃ wherein n is form 0.1 to 3, preferably n is from 0.3 to 1.0 and most preferably n is from 0.2 to 0.5.
- Other coatings which contain silicate (alone or with borate salts or boric acids or other inorganics), waxes, oils, fatty soaps can alos be used advantageously within the present invention.
- Potassium peroxymonopersulfate is another inorganic perhydrate salt of use in the detergent compositions herein.
- Suitable additional peroxyacid bleach precursors typically contain one or more N- or O- acyl groups, which precursors can be selected from a wide range of classes. Suitable classes include anhydrides, esters, imides and acylated derivatives of imidazoles and oximes, and examples of useful materials within these classes are disclosed in GB-A-1586789.
- Suitable esters are disclosed in GB-A-836988, 864798, 1147871,2143231 and EP-A-0170386.
- Useful N-acyl compounds are disclosed in GB-A-855735, 907356 and GB-A-1246338.
- Preferred precursor compounds of the imide type include the N-,N,N¹N¹ tetra acetylated alkylene diamines wherein the alkylene group contains from 1 to 6 carbon atoms, particularly those compounds in which the alkylene group contains 1, 2 and 6 carbon atoms. Tetraacetyl ethylene diamine (TAED) is particularly preferred.
- Another preferred class of peroxyacid bleach activator compounds are the amide substituted compounds of the following general formulae:
wherein R¹ is an alkyl group with from 1 to 14 carbon atoms, R² is an alkylene, arylene, and alkarylene group containing from 1 to 14 carbon atoms, and R⁵ is H or an alkyl, aryl, or alkaryl group containing 1 to 10 carbon atoms and L can be essentially any leaving group. R¹ preferably contains from 6 to 12 carbon atoms. R² preferably contains from 4 to 8 carbon atoms. R¹ may be straight chain or branched alkyl containing branching, substitution, or both and may be sourced from either synthetic sources or natural sources including for example, tallow fat. Analogous structural variations are permissible for R². The substitution can include alkyl, aryl, halogen, nitrogen, sulphur and other typical substituent groups or organic compounds. R⁵ is preferably H or methyl. R¹ and R⁵ should not contain more than 18 carbon atoms in total. L may be selected from any of the leaving groups descrobed hereinbefore for the analogues having R¹ as an aryl or alkaryl group. Amide substituted bleach activator compounds of this type are described in EP-A-0170386. - The compositions may also contain organic peroxyacids, typically at a level of from 1% to 15% by weight, more preferably from 1% to 10% by weight of the composition.
- A preferred class of organic peroxyacid compounds are the amide substituted compounds of the following general formulae:
wherein R¹ is an alkyl, aryl or alkaryl group with from 1 to 14 carbon atoms, R² is an alkylene, arylene, and alkarylene group containing from 1 to 14 carbon atoms, and R⁵ is H or an alkyl, aryl, or alkaryl group containing 1 to 10 carbon atoms. R¹ preferably contains from 6 to 12 carbon atoms. R² preferably contains from 4 to 8 carbon atoms. R¹ may be straight chain or branched alkyl, substituted aryl or alkylaryl containing branching, substitution, or both and may be sourced from either synthetic sources or natural sources including for example, tallow fat. Analogous structural variations are permissible for R². The substitution can include alkyl, aryl, halogen, nitrogen, sulphur and other typical substituent groups or organic compounds. R⁵ is preferably H or methyl. R¹ and R⁵ should not contain more than 18 carbon atoms in total. Amide substituted organic peroxyacid compounds of this type are described in EP-A-0170386. - Other organic peroxyacids include diperoxydodecanedioc acid, diperoxytetra decanedioc acid, diperoxyhexadecanedioc acid, mono- and diperazelaic acid, mono- and diperbrassylic acid, monoperoxy phthalic acid and its magnesium salt, perbenzoic acid, and their salts as disclosed in, for example, EP-A-0341 947.
- It has been found that, for optimal anti-silver tarnishing performance, the level of available oxygen in the present compositions, measured in units of % available oxygen by weight of the composition, should preferably be carefully controlled; the level of available oxygen should hence preferably be in the range from 0.3% to 2.5%, preferably from 0.5% to 1.7%, more preferably from 0.6% to 1.5%, most preferably from 0.7% to 1.2%, measured according to the method described hereunder.
- The rate of release of available oxygen is preferably also controlled; the rate of release of available oxygen from the compositions herein preferably should be such that, when using the method described hereinafter, the available oxygen is not completely released from the composition until after 3.5 minutes, preferably the available oxygen is released in a time interval of from 3.5 minutes to 10.0 minutes, more preferably from 4.0 minutes to 9.0 minutes, most preferably from 5.0 minutes to 8.5 minutes.
-
- 1. A beaker of water (typically 2L) is placed on a stirrer Hotplate, and the stirrer speed is selected to ensure that the product is evenly dispersed through the solution.
- 2. The detergent composition (typically 8g of product which has been sampled down from a bulk supply using a Pascal sampler), is added and simultaneously a stop clock is started.
- 3. The temperature control should be adjusted so as to maintain a constant temperature of 20°C throughout the experiment.
- 4. Samples are taken from the detergent solution at 2 minute time intervals for 20 mins, starting after 1 minute, and are titrated by the "titration procedure" described below to determine the level of available oxygen at each point.
-
- 1. An aliquot from the detergent solution (above) and 2ml sulphuric acid are added into a stirred beaker
- 2. Approximately 0.2g ammonium molybdate catalyst (tetra hydrate form) are added
- 3. 3mls of 10% sodium iodide solution are added
- 4. Titration with sodium thiosulphate is conducted until the end point. The end point can be seen using either of two procedures. First procedure consists simply in seeing the yellow iodine colour fading to clear. The second and preferred procedure consists of adding soluble starch when the yellow colour is becoming faint, turning the solution blue. More thiosulphate is added until the end point is reached (blue starch complex is decolourised).
-
- AvO level is plotted versus time to determine the maximum level of AvO, and the rate of release of AvO
- The rate of release of any organic peroxyacid species is also preferably controlled. The kinetics of release to a wash solution are preferably such that in the T50 test method herein described the time to achieve a concentration that is 50% of the ultimate concentration of the peroxyacid bleach is from 180 seconds to 480 seconds, more preferably from 200 seconds to 400 seconds, most preferably from 240 seconds to 360 seconds.
- The controlled release kinetics or organic peroxyacid herein are defined with respect to a 'TA test method' which measures the time to achieve A% of the ultimate concentration/level of the organic peroxyacid when a composition containing the organic peroxyacid is dissolved according to the standard conditions now set out.
- The standard conditions involve a 1 litre glass beaker filled with 1000 ml of distilled water at 20°C, to which 10g of composition is added. The contents of the beaker are agitated using a magnetic stirrer set at 100 rpm. The ultimate concentration/level is taken to be the concentration/level maximum level of organic peroxyacid attained after addition of the composition to the water-filled beaker.
- Suitable analytical methods are chosen to enable a reliable determination of the incidental, and ultimate in solution concentrations of the organic peroxyacid, subsequent to the addition of the composition to the water in the beaker.
- Such analytical methods can include those involving a continuous monitoring of the level of concentration of the component, including for example photometric and conductrimetric methods.
- Alternatively, methods involving removing titres from the solution at set time intervals, stopping the disssolution process by an appropriate means such as by rapidly reducing the temperature of the titre, and then determining the concentration of the component in the titre by any means such as chemical titrimetric methods, can be employed.
- Suitable graphical methods, including curve fitting methods, can be employed, where appropriate, to enable calculation of the the TA value from raw analytical results.
- The particular analytical method selected for determining the concentration of the organic peroxyacid component, will depend on the nature of that component, and of the nature of the composition containing that component.
- A means may be provided for controlling the rate of release of AvO, or of organic peroxyacid to the wash solution.
- Means for controlling the rate of release of any oxygen bleaching species may provide for controlled release of the oxygen bleaching species itself to the wash solution. Alternatively where the oxygen bleaching species comprises in combination a hydrogen peroxide source and an organic peroxyacid bleach precursor compound, the means may comprise a means of inhibiting, or preventing the in situ perhydrolysis reaction which releases the peroxyacid into the solution. The means could, for example, include controlling release of the hydrogen peroxide source to the wash solution, by for example, controlling release of any inorganic perhydrate salt, acting as a hydrogen peroxide source, to the wash solution.
- Suitable controlled release means can include coating any suitable component with a coating designed to provide the controlled release. The coating may therefore, for example, comprise a poorly water soluble material, or be a coating of sufficient thickness that the kinetics of dissolution of the thick coating provide the controlled rate of release.
- The coating material may be applied using various methods. Any coating material is typically present at a weight ratio of coating material to bleach of from 1:99 to 1:2, preferably from 1:49 to 1:9.
- Suitable coating materials include triglycerides (e.g. partially) hydrogenated vegetable oil, soy bean oil, cotton seed oil) mono or diglycerides, microcrystalline waxes, gelatin, cellulose, fatty acids and any mixtures thereof.
- Other suitable coating materials can comprise the alkali and alkaline earth metal sulphates, silicates and carbonates, including calcium carbonate and silicas.
- A preferred coating material is sodium silicate of SiO₂ : Na₂O ratio from 1.6 : 1 to 3.4 : 1, preferably 2.2:1 to 2.8:1, applied as an aqueous solution to give a level of from 2% to 10%, (normally from 3% to 5%) of silicate solids by weight of the oxygen bleaching species. Magnesium silicate can also be included in the coating.
- Any inorganic salt coating materials may be combined with organic binder materials to provie composite inorganic salt/organic binder coatings. Suitable binders include the C₁₀-C₂₀ alcohol ethoxylates containing from 5 - 100 moles of ethylene oxide per mole of alcohol and more preferably the C₁₅-C₂₀ primary alcohol ethoxylates containing from 20 - 100 moles of ethylene oxide per mole of alcohol.
- Other preferred binders include certain polymeric materials. Polyvinylpyrrolidones with an average molecular weight of from 12,000 to 700,000 and polyethylene glycols (PEG) with an average molecular weight of from 600 to 5 x 10⁶ preferably 1000 to 400,000 most preferably 1000 to 10,000 are examples of such polymeric materials. Copolymers of maleic anhydride with ethylene, methylvinyl ether or methacrylic acid, the maleic anhydride constituting at least 20 mole percent of the polymer are further examples of polymeric materials useful as binder agents. These polymeric materials may be used as such or in combination with solvents such as water, propylene glycol and the above mentioned C₁₀-C₂₀ alcohol ethoxylates containing from 5 - 100 moles of ethylene oxide per mole. Further examples of binders include the C₁₀-C₂₀ mono- and diglycerol ethers and also the C₁₀-C₂₀ fatty acids.
- Cellulose derivatives such as methylcellulose, carboxymethylcellulose and hydroxyethylcellulose, and homo- or co-polymeric polycarboxylic acids or their salts are other examples of binders suitable for use herein.
- One method for applying the coating material involves agglomeration. Preferred agglomeration processes include the use of any of the organic binder materials described hereinabove. Any conventional agglomerator/mixer may be used including, but not limted to pan, rotary drum and vertical blender types. Molten coating compositions may also be applied either by being poured onto, or spray atomized onto a moving bed of bleaching agent.
- Other means of providing the required controlled release include mechanical means for altering the physical characteristics of the bleach to control its solubility and rate of release. Suitable protocols could include compaction, mechanical injection, manual injection, and adjustment of the solubility of the bleach compound by selection of particle size of any particulate component.
- Whilst the choice of particle size will depend both on the composition of the particulate component, and the desire to meet the desired controlled release kinetics, it is desirable that the particle size should be more than 500 micrometers, preferably having an average particle diameter of from 800 to 1200 micrometers.
- Additional protocols for providing the means of controlled release include the suitable choice of any other components of the detergent composition matrix such that when the composition is introduced to the wash solution the ionic strength environment therein provided enables the required controlled release kinetics to be achieved.
- The compositions contain an Mn(II) compound to inhibit the tarnishing of silverware. The Mn(II) compound is preferably incorporated at a level of from 0.005% to 5% by weight, more preferably from 0.01% to 1%, most preferably from 0.02% to 0.4% by weight of the compositions. Preferably, the Mn(II) compound is incorporated at a level to provide from 0.1 ppm to 250 ppm, more preferably from 0.5 ppm to 50 ppm, most preferably from 1 ppm to 20 ppm by weight of Mn(II) ions in any bleaching solution.
- The Mn (II) compound may be an inorganic salt in anhydrous, or any hydrated forms. Suitable salts include manganese sulphate, manganese carbonate, manganese phosphate, manganese nitrate, manganese acetate and manganese chloride.
- The Mn(II) compound may be a salt or complex of an organic fatty acid such as manganese acetate or manganese stearate.
- The Mn(II) compound may be a salt or complex of an organic ligand. Preferably any salts/complexes are water soluble. In one preferred execution the organic ligand is a heavy metal ion sequestrant. In another preferred execution the organic ligand is a crystal growth inhibitor compound.
- By heavy metal ion sequestrant it is meant herein components which act to sequester (chelate) heavy metal ions. These components may also have calcium and magnesium chelation capacity, but preferentially they show selectivity to binding heavy metal ions such as iron, manganese and copper.
- Suitable heavy metal ion sequestrants include organic aminophosphonates, such as the amino alkylene poly (alkylene phosphonates), and nitrilo trimethylene phosphonates.
- Preferred among the above species are diethylene triamine penta (methylene phosphonate), ethylene diamine tri (methylene phosphonate) and hexamethylene diamine tetra (methylene phosphonate).
- Other suitable heavy metal ion sequestrants include nitrilotriacetic acid and polyaminocarboxylic acids such as ethylenediaminetetracetic acid, ethylenetriamine pentacetic acid, ethylenediamine disuccinic acid, ethylenediamine diglutaric acid, 2-hydroxypropylenediamine disuccinic acid, and the partially or fully ionized forms thereof. Ethylenediamine disuccinic acid is preferred, most preferably in the form of its S,S isomer.
- Other suitable heavy metal ion sequestrants for use herein are iminodiacetic acid derivatives such as 2-hydroxyethyl diacetic acid or glyceryl imino diacetic acid, described in EP-A-317,542 and EP-A-399,133. The iminodiacetic acid-N-2-hydroxypropyl sulfonic acid and aspartic acid N-carboxymethyl N-2-hydroxypropyl-3-sulfonic acid sequestrants described in EP-A-516,102 are also suitable herein. The β-alanine-N,N'-diacetic acid, aspartic acid-N,N'-diacetic acid, aspartic acid-N-monoacetic acid and iminodisuccinic acid sequestrants described in EP-A-509,382 are also suitable.
- EP-A-476,257 describes suitable amino based heavy metal ion sequestrants. EP-A-510,331 describes suitable sequestrants derived from collagen, keratin or casein. EP-A-528,859 describes a suitable alkyl iminodiacetic acid sequestrant. Dipicolinic acid and 2-phosphonobutane-1,2,4-tricarboxylic acid are also suitable. Glycinamide-N,N'-disuccinic acid (GADS) is also suitable.
- The organic ligand may be a crystal growth inhibitor component, particularly an organo diphosphonic acid component. By organo diphosphonic acid it is meant herein an organo diphosphonic acid which does not contain nitrogen as part of its chemical structure. This definition therefore excludes the organo aminophosphonates, which however may be included in compositions of the invention as heavy metal ion sequestrant components.
- The organo diphosphonic acid is preferably a C₁-C₄ diphosphonic acid, more preferably a C₂ diphosphonic acid, such as ethylene diphosphonic acid, or most preferably ethane 1-hydroxy-1,1-diphosphonic acid (HEDP) and may be present in the Mn(II) compound in partially or fully ionized form.
- In addition to the essential ingredients described hereinabove, the compositions of the invention may comprise additional ingredients, which are often quite desirable ones.
- The compositions of the present invention may contain as a highly preferred component a water-soluble builder compound, typically present at a level of from 1% to 80% by weight, preferably from 10% to 70% by weight, most preferably from 20% to 60% by weight of the composition.
- Suitable water-soluble builder compounds include the water soluble monomeric polycarboxylates, or their acid forms, homo or copolymeric polycarboxylic acids or their salts in which the polycarboxylic acid comprises at least two carboxylic radicals separated from each other by not more that two carbon atoms, carbonates, bicarbonates, borates, phosphates, silicates and mixtures of any of the foregoing.
- The carboxylate or polycarboxylate builder can be momomeric or oligomeric in type although monomeric polycarboxylates are generally preferred for reasons of cost and performance.
- Suitable carboxylates containing one carboxy group include the water soluble salts of lactic acid, glycolic acid and ether derivatives thereof. Polycarboxylates containing two carboxy groups include the water-soluble salts of succinic acid, malonic acid, (ethylenedioxy) diacetic acid, maleic acid, diglycolic acid, tartaric acid, tartronic acid and fumaric acid, as well as the ether carboxylates and the sulfinyl carboxylates. Polycarboxylates containing three carboxy groups include, in particular, water-soluble citrates, aconitrates and citraconates as well as succinate derivatives such as the carboxymethyloxysuccinates described in British Patent No. 1,379,241, lactoxysuccinates described in British Patent No. 1,389,732, and aminosuccinates described in Netherlands Application 7205873, and the oxypolycarboxylate materials such as 2-oxa-1,1,3-propane tricarboxylates described in British Patent No. 1,387,447.
- Polycarboxylates containing four carboxy groups include oxydisuccinates disclosed in British Patent No. 1,261,829, 1,1,2,2-ethane tetracarboxylates, 1,1,3,3-propane tetracarboxylates and 1,1,2,3-propane tetracarboxylates. Polycarboxylates containing sulfo substituents include the sulfosuccinate derivatives disclosed in British Patent Nos. 1,398,421 and 1,398,422 and in U.S. Patent No. 3,936,448, and the sulfonated pyrolysed citrates described in British Patent No. 1,439,000.
- Alicyclic and heterocyclic polycarboxylates include cyclopentanecis,cis,cis-tetracarboxylates, cyclopentadienide pentacarboxylates, 2,3,4,5-tetrahydrofuran - cis, cis, cis-tetracarboxylates, 2,5-tetrahydrofuran - cisdicarboxylates, 2,2,5,5-tetrahydrofuran - tetracarboxylates, 1,2,3,4,5,6-hexane - hexacarboxylates and carboxymethyl derivatives of polyhydric alcohols such as sorbitol, mannitol and xylitol. Aromatic polycarboxylates include mellitic acid, pyromellitic acid and the phthalic acid derivatives disclosed in British Patent No. 1,425,343.
- Of the above, the preferred polycarboxylates are hydroxycarboxylates containing up to three carboxy groups per molecule, more particularly citrates.
- The parent acids of the monomeric or oligomeric polycarboxylate chelating agents or mixtures thereof with their salts, e.g. citric acid or citrate/citric acid mixtures are also contemplated as useful builder components.
- Borate builders, as well as builders containing borate-forming materials that can produce borate under detergent storage or wash conditions can also be used but are not preferred at wash conditions less that about 50°C, especially less than about 40°C.
- Examples of carbonate builders are the alkaline earth and alkali metal carbonates, including sodium carbonate and sesqui-carbonate and mixtures thereof with ultra-fine calcium carbonate as disclosed in German Patent Application No. 2,321,001 published on November 15, 1973.
- Specific examples of water-soluble phosphate builders are the alkali metal tripolyphosphates, sodium, potassium and ammonium pyrophosphate, sodium and potassium and ammonium pyrophosphate, sodium and potassium orthophosphate, sodium polymeta/phosphate in which the degree of polymerization ranges from about 6 to 21, and salts of phytic acid.
- Suitable silicates include the water soluble sodium silicates with an Si0₂: Na₂0 ratio of from 1.0 to 2.8, with ratios of from 1.6 to 2.4 being preferred, and 2.0 ratio being most preferred. The silicates may be in the form of either the anhydrous salt or a hydrated salt. Sodium silicate with an SiO₂: Na₂0 ratio of 2.0 is the most preferred silicate.
- Silicates are preferably present in the detergent compositions in accord with the invention at a level of from 5% to 50% by weight of the composition, more preferably from 10% to 40% by weight.
- The compositions of the present invention may contain a partially soluble or insoluble builder compound, typically present at a level of from 1% to 80% by weight, preferably from 10% to 70% by weight, most preferably from 20% to 60% weight of the composition.
- Examples of partially water soluble builders include the crystalline layered silicates. Examples of largely water insoluble builders include the sodium aluminosilicates.
- Crystalline layered sodium silicates have the general formula
NaMSix02x+1.yH₂0
wherein M is sodium or hydrogen, x is a number from 1.9 to 4 and y is a number from 0 to 20. Crystalline layered sodium silicates of this type are disclosed in EP-A-0164514 and methods for their preparation are disclosed in DE-A-3417649 and DE-A-3742043. For the purpose of the present invention, x in the general formula above has a value of 2, 3 or 4 and is preferably 2. The most preferred material is δ-Na₂Si₂0₅, available from Hoechst AG as NaSKS-6. - The crystalline layered sodium silicate material is preferably present in granular detergent compositions as a particulate in intimate admixture with a solid, water-soluble ionisable material. The solid, water-soluble ionisable material is selected from organic acids, organic and inorganic acid salts and mixtures thereof.
- Suitable aluminosilicate zeolites have the unit cell formula Naz[(AlO₂)z(SiO₂)y]. XH₂O wherein z and y are at least 6; the molar ratio of z to y is from 1.0 to 0.5 and x is at least 5, preferably from 7.5 to 276, more preferably from 10 to 264. The aluminosilicate material are in hydrated form and are preferably crystalline, containing from 10% to 28%, more preferably from 18% to 22% water in bound form.
- The aluminosilicate ion exchange materials can be naturally occurring materials, but are preferably synthetically derived. Synthetic crystalline aluminosilicate ion exchange materials are available under the designations Zeolite A, Zeolite B, Zeolite P, Zeolite X, Zeoilte MAP, Zeolite HS and mixtures thereof. Zeolite A has the formula
Na ₁₂ [AlO₂) ₁₂ (SiO₂)₁₂]. xH₂O
wherein x is from 20 to 30, especially 27. Zeolite X has the formula Na₈₆ [(AlO₂)₈₆(SiO₂)₁₀₆]. 276 H₂O. - A highly preferred component of the compositions of the invention is a surfactant system comprising surfactant selected from anionic, cationic, nonionic ampholytic and zwitterionic surfactants and mixtures thereof. Most preferably the surfactant system comprises low foaming surfactant, especially low foaming nonionic surfactant. The surfactant system is preferably present at a level of from 0.25% to 30% by weight, more preferably 0.5% to 10% by weight, most preferably from 1% to 5% by weight of the compositions.
- A typical listing of anionic, nonionic, ampholytic and zwitterionic classes, and species of these surfactants, is given in U.S.P. 3,929,678 issued to Laughlin and Heuring on December, 30, 1975. A list of suitable cationic surfactants is given in U.S.P. 4,259,217 issued to Murphy on March 31,1981. A listing of surfactants typically included in automatic dishwashing detergent compositions is given in EP-A-0414 549.
- Sulphonate and sulphate surfactants are useful herein. Sulphonates include alkyl benzene sulphonates having from 5 to 15 carbon atoms in the alkyl radical, and alpha-sulphonated methyl fatty acid testers in which the fatty acid is derived from a C₆-C18 fatty source. Preferred sulphate surfactants are alkyl sulphates having from 6 to 16, preferably 6 to 10 carbon atoms in the alkyl radical.
- Useful surfactant system comprises a mixture of two alkyl sulphate materials whose respective mean chain lengths differ from each other. The cation in each instance is again an alkali metal, preferably sodium. The alkyl sulfate salts may be derived from natural or synthetic hydrocarbon sources.
- The C₆-C₁₆ alkyl ethoxysulfate salt comprises a primary alkyl ethoxysulfate which is derived from the condensation product of a C₆-C₁₆ alcohol condensed with an average of from one to seven ethylene oxide groups, per mole. Preferred are the C₆-C₁₀ alkyl ethoxysulfate salts with an average of from one to five ethoxy groups per mole.
- Other anionic surfactants suitable for the purposes of the invention are the alkali metal sarcosinates of formula
R-CON (R¹) CH₂ COOM
wherein R is a C₅-C₁₇ linear or branched alkyl or alkenyl group, R¹ is a C₁-C₄ alkyl group and M is an alkali metal ion. Preferred examples are the lauroyl, Cocoyl (C₁₂-C₁₄), myristyl and oleyl methyl sarcosinates in the form of their sodium salts. - Another class of anionic surfactants useful herein are the alkyl ester sulfonate surfactants which include linear esters of C₈-C₂₀ carboxylic acids (i.e., fatty acids) which are sulfonated with gaseous SO₃ according to "The Journal of the American Oil Chemists Society," 52 (1975), pp. 323-329. Suitable starting materials would include natural fatty substances as derived from tallow, palm oil, etc.
- The preferred alkyl ester sulfonate surfactants have the structural formula:
wherein R³ is a C₈-C₂₀ hydrocarbyl, preferably an alkyl, or combination thereof, R⁴ is a C₁-C₆ hydrocarbyl, preferably an alkyl, or combination thereof, and M is a cation which forms a water soluble salt with the alkyl ester sulfonate. Suitable salt-forming cations include metals such as sodium, potassium, and lithium, and substituted or unsubstituted ammonium cations, such as monoethanolamine, diethanolamine, and triethanolamine. Preferably, R³ is C₁₀-C₁₆ alkyl, and R⁴is methyl, ethyl or isopropyl. Especially preferred are the methyl ester sulfonates wherein R³ is C₁₀-C₁₆ alkyl. - One preferred class of low foaming nonionic surfactants useful in the present invention comprises the water soluble ethoxylated C₆-C₁₆ fatty alcohols and C₆-C₁₆ mixed ethoxylated/propoxylated fatty alcohols and mixtures thereof. Preferably the ethoxylated fatty alcohols are the C₁₀-C₁₆ ethoxylated fatty alcohols with a degree of ethoxylation of from 3 to 50, most preferably these are the C₁₂-C₁₆ ethoxylated fatty alcohols with a degree of ethoxylation from 3 to 40. Preferably the mixed ethoxylated/propoxylated fatty alcohols have an alkyl chain length of from 10 to 16 carbon atoms, a degree of ethoxylation of from 3 to 30 and a degree of propoxylation of from 1 to 10.
- Thus C6-C16 alcohol itself can be obtained from natural or synthetic sources. Thus, C6-C16 alcohols, derived from natural fats, or Ziegler olefin build-up, or OXO synthesis can form suitable sources for the alkyl group. Examples of synthetically derived materials include Dobanol 25 (RTM) sold by Shell Chemicals (UK) Ltd which is a blend of C₁₂-C₁₅ alcohols, Ethyl 24 sold by the Ethyl Corporation which is a blend of C₁₂-C₁₅ alcohols, and a blend of C₁₃-C₁₅ alcohols in the ratio 67% C₁₃, 33% C₁₅ sold under the trade name Lutensol by BASF GmbH and Synperonic (RTM) by ICI Ltd., and Lial 125 sold by Liquichimica Italiana. Examples of naturally occuring materials from which the alcohols can be derived are coconut oil and palm kernel oil and the corresponding fatty acids.
- Another class of nonionic surfactants comprises alkyl polyglucoside compounds of general formula
RO (CnH2nO)tZx
wherein Z is a moiety derived from glucose; R is a saturated hydrophobic alkyl group that contains from 6 to 16 carbon atoms preferably from 6 to 14 carbon atoms; t is from 0 to 10 and n is 2 or 3; x is from 1.1 to 4, the compounds including less than 10% unreacted fatty alcohol and less than 50% short chain alkyl polyglucosides. Compounds of this type and their use in detergent compositions are disclosed in EP-B 0070074, 0070077, 0075996 and 0094118. - Another preferred nonionic surfactant is a polyhydroxy fatty acid amide surfactant compound having the structural formula:
wherein R¹ is H, C₁-C₄ hydrocarbyl, 2-hydroxy ethyl, 2-hydroxy propyl, or a mixture thereof, preferably C₁-C₄ alkyl, more preferably C₁ or C₂ alkyl, most preferably C₁ alkyl (ie., methyl); and R² is a C₅-C₁₅ hydrocarbyl, preferably straight chain C₅-C₁₃ alkyl or alkenyl, more preferably straight chain C₅-C₁₁ alkyl or alkenyl, most preferably straight chain C₅-C₉ alkyl or alkenyl, or mixture thereof: and Z is a polyhydroxyhydrocarbyl having linear hydrocarbyl chain with at least 3 hydroxyls directly connected to the chain, or an alkoxlylated derivative (preferably ethoxylated or propoxylated) thereof. Z preferably will be derived from a reducing sugar in a reductive amination reaction; more preferably Z is a glycityl. Suitable reducing sugars include glucose, fructose, maltose, lactose, galactose, mannose, and xylose. As raw materials, high dextrose corn syrup, high fructose corn syrup, and high maltose corn syrup can be utilized as well as the individual sugars listed above. These corn syrups may yield a mix of sugar components for Z. It should be understood that it is by no means intended to exclude other suitable raw materials. Z preferably will be selected from the group consisting of -CH₂-(CHOH)n-CH₂OH, -CH(CH₂OH)-(CHOH)n-1-CH₂OH, -CH₂-(CHOH)₂(CHOR')(CHOH)-CH₂OH, where n is an integer from 3 to 5, inclusive, and R' is H or a cyclic or aliphatic monosaccharide, and alkoxylated derivatives thereof. Most preferred are glycityls wherein n is 4, particularly - CH₂-(CHOH)₄-CH₂OH. - In Formula (I), R¹ can be, for example, N-methyl, N-ethyl, N-propyl, N-isopropyl, N-butyl, N-2-hydroxy ethyl, or N-2-hydroxy propyl.
- R²-CO-N< can be, for example, cocamide, stearamide, oleamide, lauramide, myristamide, capricamide, palmitamide, or tallowamide.
- Z can be 1-deoxyglucityl, 2-deoxyfrucittyl, 1-deoxymaltityl, 1-deoxylactityl, 1-deoxygalactityl or 1-deoxymannityl, or 1-deoxymaltotriotityl. Preferred compounds are N-methyl N-1deoxyglucityl C₁₄-C₁₈ fatty acid amides.
- A further class of surfactants are the semi-polar surfactants such as amine oxides. Suitable amine oxides are selected from mono C₆-C₂₀, preferably C₆-C₁₀ N-alkyl or alkenyl amine oxides and propylene-1,3-diamine dioxides wherein the remaining N positions are substituted by methyl, hydroxyethyl or hydroxpropyl groups.
- Cationic surfactants can also be used in the detergent compositions herein and suitable quaternary ammonium surfactants are selected from mono C₆-C₁₆, preferably C₆-C₁₀ N-alkyl or alkenyl ammonium surfactants wherein remaining N positions are substituted by methyl, hydroxyethyl or hydroxypropyl groups.
- The compositions may contain narrowly defined levels of a heavy metal ion sequestrant which is not in the form of a Mn(II) compound, such levels being in the range 0.005% to 3%, preferably 0.01 to 1%, most preferably 0.05% to 0.8%, by weight of the total composition.
- Heavy metal ion sequestrants, which are acidic in nature, having for example phosphonic acid or carboxylic acid functionalities, may be present either in their acid form or as a complex/salt with a suitable non Mn(II) counter cation such as an alkali or alkaline metal ion, ammonium, or substituted ammonium ion, or any mixtures thereof. The molar ratio of said counter cation to the heavy metal ion sequestrant is preferably at least 1:1.
- The heavy metal ion sequestrant may be any of those described as suitable heavy metal ion sequestrant-type organic ligands in the Mn(II) compounds section of this descrption.
- Especially preferred is ethylenediamine-N,N'-disuccinic acid (EDDS) or the alkali metal, alkaline earth metal, ammonium, or substituted ammonium salts thereof, or mixtures thereof. Preferred EDDS compounds are the free acid form and the sodium or magnesium salt or complex thereof. Examples of such preferred sodium salts of EDDS include Na₂EDDS and Na₃EDDS. Examples of such preferred magnesium complexes of EDDS include MgEDDS and Mg₂EDDS.
- The heavy metal ion sequestrant herein can consist of a mixture of the above described species.
- Chlorine bleaches include the alkali metal hypochlorites and chlorinated cyanuric acid salts. The use of chlorine bleaches in the composition of the invention is optional and preferably minimized, and more preferably the present compositions contain no chlorine bleach.
- Another optional ingredient useful in the compositions is one or more additional enzymes.
- Preferred additional enzymatic materials include the commercially available lipases, amylases, neutral and alkaline proteases, esterases, cellulases, pectinases, lactases and peroxidases conventionally incorporated into detergent compositions. Suitable enzymes are discussed in US Patents 3,519,570 and 3,533,139.
- Preferred commercially available protease enzymes include those sold under the tradenames Alcalase, Savinase, Primase, Durazym, and Esperase by Novo Industries A/S (Denmark), those sold under the tradename Maxatase, Maxacal and Maxapem by Gist-Brocades, those sold by Genencor International, and those sold under the tradename Opticlean and Optimase by Solvay Enzymes. Protease enzyme may be incorporated into the compositions in accordance with the invention at a level of from 0.0001% to 4% active enzyme by weight of the composition.
- Preferred amylases include, for example, α-amylases obtained from a special strain of B licheniformis, described in more detail in GB-1,269,839 (Novo). Preferred commercially available amylases include for example, those sold under the tradename Rapidase by Gist-Brocades, and those sold under the tradename Termamyl and BAN by Novo Industries A/S. Amylase enzyme may be incorporated into the composition in accordance with the invention at a level of from 0.0001% to 2% active enzyme by weight of the composition.
- Lipolytic enzyme (lipase) may be present at levels of active lipolytic enzyme of from 0.0001% to 2% by weight, preferably 0.001% to 1% by weight, most preferably from 0.001% to 0.5% by weight of the compositions.
- The lipase may be fungal or bacterial in origin being obtained, for example, from a lipase producing strain of Humicola sp., Thermomyces sp. or Pseudomonas sp. including Pseudomonas pseudoalcaligenes or Pseudomas fluorescens. Lipase from chemically or genetically modified mutants of these strains are also useful herein.
- A preferred lipase is derived from Pseudomonas pseudoalcaligenes, which is described in Granted European Patent, EP-B-0218272.
- Another preferred lipase herein is obtained by cloning the gene from Humicola lanuginosa and expressing the gene in Aspergillus oryza, as host, as described in European Patent Application, EP-A-0258 068, which is commercially available from Novo Industri A/S, Bagsvaerd, Denmark, under the trade name Lipolase. This lipase is also described in U.S. Patent 4,810,414, Huge-Jensen et al, issued March 7, 1989.
- Preferred enzyme-containing compositions herein may comprise from about 0.001% to about 10%, preferably from about 0.005% to about 8%,most preferably from about 0.01% to about 6%, by weight of an enzyme stabilizing system. The enzyme stabilizing system can be any stabilizing system which is compatible with the detersive enzyme. Such stabilizing systems can comprise calcium ion, boric acid, propylene glycol, short chain carboxylic acid, boronic acid, and mixtures thereof. Such stabilizing systems can also comprise reversible enzyme inhibitors, such as reversible protease inhibitors.
- The compositions herein may further comprise from 0 to about 10%, preferably from about 0.01% to about 6% by weight, of chlorine bleach scavengers, added to prevent chlorine bleach species present in many water supplies from attacking and inactivating the enzymes, especially under alkaline conditions. While chlorine levels in water may be small, typically in the range from about 0.5 ppm to about 1.75 ppm, the available chlorine in the total volume of water that comes in contact with the enzyme during washing is usually large; accordingly, enzyme stability in-use can be problematic.
- Suitable chlorine scavenger anions are widely available, and are illustrated by salts containing ammonium cations or sulfite, bisulfite, thiosulfite, thiosulfate, iodide, etc. Antioxidants such as carbamate, ascorbate, etc., organic amines such as ethylenediaminetetracetic acid (EDTA) or alkali metal salt thereof, monoethanolamine (MEA), and mixtures thereof can likewise be used. Other conventional scavengers such as bisulfate, nitrate, chloride, sources of hydrogen peroxide such as sodium perborate tetrahydrate, sodium perborate monohydrate and sodium percarbonate, as well as phosphate, condensed phosphate, acetate, benzoate, citrate, formate, lactate, malate, tartrate, salicylate, etc. and mixtures thereof can be used if desired.
- Organic polymeric compounds are particularly preferred components of the compositions in accord with the invention. By organic polymeric compound it is meant essentially any polymeric organic compound commonly used as dispersants, and anti-redeposition and soil suspension agents in detergent compositions.
- Organic polymeric compound is typically incorporated in the detergent compositions of the invention at a level of from 0.1% to 30%, preferably from 0.5% to 15%, most preferably from 1% to 10% by weight of the compositions.
- Examples of organic polymeric compounds include the water soluble organic homo- or co-polymeric polycarboxylic acids or their salts in which the polycarboxylic acid comprises at least two carboxyl radicals separated from each other by not more than two carbon atoms. Polymers of the latter type are disclosed in GB-A-1,596,756. Examples of such salts are polyacrylates of molecular weight 2000-10000 and their copolymers with any suitable other monomer units including modified acrylic, fumaric, maleic, itaconic, aconitic, mesaconic, citraconic and methylenemalonic acid or their salts, maleic anhydride, acrylamide, alkylene, vinylmethyl ether, styrene and any mixtures thereof. Preferred are the copolymers of acrylic acid and maleic anhydride having a molecular weight of from 20,000 to 100,000.
- Preferred commercially available acrylic acid containing polymers having a molecular weight below 15,000 include those sold under the tradename Sokalan PA30, PA20, PA15, PA10 and Sokalan CP10 by BASF GmbH, and those sold under the tradename Acusol 45N by Rohm and Haas.
- Preferred acrylic acid containing copolymers include those which contain as monomer units: a) from 90% to 10%, preferably from 80% to 20% by weight acrylic acid or its salts and b) from 10% to 90%, preferably from 20% to 80% by weight of a substituted acrylic monomer or its salts having the general formula -[CR₂-CR₁(CO-O-R₃)]- wherein at least one of the substituents R₁, R₂ or R₃, preferably R₁ or R₂ is a 1 to 4 carbon alkyl or hydroxyalkyl group, R₁ or R₂ can be a hydrogen and R₃ can be a hydrogen or alkali metal salt. Most preferred is a substituted acrylic monomer wherein R₁ is methyl, R₂ is hydrogen (i.e. a methacrylic acid monomer). The most preferred copolymer of this type has a molecular weight of 3500 and contains 60% to 80% by weight of acrylic acid and 40% to 20% by weight of methacrylic acid.
- Other suitable polyacrylate/modified polyacrylate copolymers include those copolymers of unsaturated aliphatic carboxylic acids disclosed in U.S. Patents No.s 4,530,766, and 5,084,535 which have a molecular weight of less than 15,000.
- Other suitable organic polymeric compounds include the copolymers of acrylamide and acrylate having a molecular weight of from 3,000 to 100,000, and the acrylate/fumarate copolymers having a molecular weight of from 2,000 to 80,000.
- The polyamino compounds are useful herein including those derived from aspartic acid such as those disclosed in EP-A-305282, EP-A-305283 and EP-A-351629.
- Other organic polymeric compounds suitable for incorporation in the detergent compositions herein include cellulose derivatives such as methylcellulose, carboxymethylcellulose and hydroxyethylcellulose.
- Further useful organic polymeric compounds are the polyethylene glycols, particularly those of molecular weight 1000-10000, more particularly 2000 to 8000 and most preferably about 4000.
- The compositions of the invention may contain a lime soap dispersant compound, which has a lime soap dispersing power (LSDP), as defined hereinafter of no more than 8, preferably no more than 7, most preferably no more than 6. The lime soap dispersant compound is preferably present at a level of from 0.1% to 40% by weight, more preferably 1% to 20% by weight, most preferably from 2% to 10% by weight of the compositions.
- A lime soap dispersant is a material that prevents the precipitation of alkali metal, ammonium or amine salts of fatty acids by calcium or magnesium ions. A numerical measure of the effectiveness of a lime soap dispersant is given by the lime soap dispersing power (LSDP) which is determined using the lime soap dispersion test as described in an article by H.C. Borghetty and C.A. Bergman, J. Am. Oil. Chem. Soc., volume 27, pages 88-90, (1950). This lime soap dispersion test method is widely used by practitioners in this art field being referred to, for example, in the following review articles; W.N. Linfield, Surfactant Science Series, Volume 7, p3; W.N. Linfield, Tenside Surf. Det., Volume 27, pages 159-161, (1990); and M.K. Nagarajan, W.F. Masler, Cosmetics and Toiletries, Volume 104, pages 71-73, (1989). The LSDP is the % weight ratio of dispersing agent to sodium oleate required to disperse the lime soap deposits formed by 0.025g of sodium oleate in 30ml of water of 333ppm CaCO₃ (Ca:Mg=3:2) equivalent hardness.
- Surfactants having good lime soap dispersant capability will include certain amine oxides, betaines, sulfobetaines, alkyl ethoxysulfates and ethoxylated alcohols.
- Exemplary surfactants having a LSDP of no more than 8 for use in accord with the invention include C₁₆-C₁₈ dimethyl amine oxide, C₁₂-C₁₈ alkyl ethoxysulfates with an average degree of ethoxylation of from 1-5, particularly C₁₂-C₁₅ alkyl ethoxysulfate surfactant with a degree of ethoxylation of about 3 (LSDP=4), and the C₁₃-C₁₅ ethoxylated alcohols with an average degree of ethoxylation of either 12 (LSDP=6) or 30, sold under the trade names Lutensol A012 and Lutensol A030 respectively, by BASF GmbH.
- Polymeric lime soap dispersants suitable for use herein are described in the article by M.K. Nagarajan and W.F. Masler, to be found in Cosmetics and Toiletries, Volume 104, pages 71-73, (1989). Examples of such polymeric lime soap dispersants include certain water-soluble salts of copolymers of acrylic acid, methacrylic acid or mixtures thereof, and an acrylamide or substituted acrylamide, where such polymers typically have a molecular weight of from 5,000 to 20,000.
- The compositions of the invention, when formulated for use in machine washing compositions, preferably comprise a suds suppressing system present at a level of from 0.01% to 15%, preferably from 0.05% to 10%, most preferably from 0.1% to 5% by weight of the composition.
- Suitable suds suppressing systems for use herein may comprise essentially any known antifoam compound, including, for example silicone antifoam compounds, 2-alkyl and alcanol antifoam compounds.
- By antifoam compound it is meant herein any compound or mixtures of compounds which act such as to depress the foaming or sudsing produced by a solution of a detergent composition, particularly in the presence of agitation of that solution.
- Particularly preferred antifoam compounds for use herein are silicone antifoam compounds defined herein as any antifoam compound including a silicone component. Such silicone antifoam compounds also typically contain a silica component. The term "silicone" as used herein, and in general throughout the industry, encompasses a variety of relatively high molecular weight polymers containing siloxane units and hydrocarbyl group of various types. Preferred silicone antifoam compounds are the siloxanes, particularly the polydimethylsiloxanes having trimethylsilyl end blocking units.
- Other suitable antifoam compounds include the monocarboxylic fatty acids and soluble salts thereof. These materials are described in US Patent 2,954,347, issued September 27, 1960 to Wayne St. John. The monocarboxylic fatty acids, and salts thereof, for use as suds suppressor typically have hydrocarbyl chains of 10 to about 24 carbon atoms, preferably 12 to 18 carbon atoms. Suitable salts include the alkali metal salts such as sodium, potassium, and lithium salts, and ammonium and alkanolammonium salts.
- Other suitable antifoam compounds include, for example, high molecular weight fatty esters (e.g. fatty acid triglycerides), fatty acid esters of monovalent alcohols, aliphatic C₁₈-C₄₀ ketones (e.g. stearone) N-alkylated amino triazines such as tri- to hexa-alkylmelamines or di- to tetra alkyldiamine chlortriazines formed as products of cyanuric chloride with two or three moles of a primary or secondary amine containing 1 to 24 carbon atoms, propylene oxide, bis stearic acid amide and monostearyl dialkali metal (e.g. sodium, potassium, lithium) phosphates and phosphate esters.
- Copolymers of ethylene oxide and propylene oxide, particularly the mixed ethoxylated/propoxylated fatty alcohols with an alkyl chain length of from 10 to 16 carbon atoms, a degree of ethoxylation of from 3 to 30 and a degree of propoxylation of from 1 to 10, are also suitable antifoam compounds for use herein.
- Suitable 2-alky-alcanols antifoam compounds for use herein have been described in DE 40 21 265. The 2-alkyl-alcanols suitable for use herein consist of a C₆ to C₁₆ alkyl chain carrying a terminal hydroxy group, and said alkyl chain is substituted in the a position by a C₁ to C₁₀ alkyl chain. Mixtures of 2-alkyl-alcanols can be used in the compositions according to the present invention.
- A preferred suds suppressing system comprises
(a) antifoam compound, preferably silicone antifoam compound, most preferably a silicone antifoam compound comprising in combination - (i) polydimethyl siloxane, at a level of from 50% to 99%, preferably 75% to 95% by weight of the silicone antifoam compound; and
- (ii) silica, at a level of from 1% to 50%, preferably 5% to 25% by weight of the silicone/silica antifoam compound;
- (b) a dispersant compound, most preferably comprising a silicone glycol rake copolymer with a polyoxyalkylene content of 72-78% and an ethylene oxide to propylene oxide ratio of from 1:0.9 to 1:1.1, at a level of from 0.5% to 10%, preferably 1% to 10% by weight; a particularly preferred silicone glycol rake copolymer of this type is DCO544, commercially available from DOW Corning under the tradename DCO544;
- (c) an inert carrier fluid compound, most preferably comprising a C₁₆-C₁₈ ethoxylated alcohol with a degree of ethoxylation of from 5 to 50, preferably 8 to 15, at a level of from 5% to 80%, preferably 10% to 70%, by weight;
- A preferred particulate suds suppressor system useful herein comprises a mixture of an alkylated siloxane of the type hereinabove disclosed and solid silica.
- The solid silica can be a fumed silica, a precipitated silica or a silica, made by the gel formation technique. The silica particles suitable have an average particle size of from 0.1 to 50 micrometers, preferably from 1 to 20 micrometers and a surface area of at least 50m²/g. These silica particles can be rendered hydrophobic by treating them with dialkylsilyl groups and/or trialkylsilyl groups either bonded directly onto the silica or by means of a silicone resin. It is preferred to employ a silica the particles of which have been rendered hydrophobic with dimethyl and/or trimethyl silyl groups. A preferred particulate antifoam compound for inclusion in the detergent compositions in accordance with the invention suitably contain an amount of silica such that the weight ratio of silica to silicone lies in the range from 1:100 to 3:10, preferably from 1:50 to 1:7.
- Another suitable particulate suds suppressing system is represented by a hydrophobic silanated (most preferably trimethyl-silanated) silica having a particle size in the range from 10 nanometers to 20 nanometers and a specific surface area above 50m²/g, intimately admixed with dimethyl silicone fluid having a molecular weight in the range from about 500 to about 200,000 at a weight ratio of silicone to silanated silica of from about 1:1 to about 1:2.
- A highly preferred particulate suds suppressing system is described in EP-A-0210731 and comprises a silicone antifoam compound and an organic carrier material having a melting point in the range 50°C to 85°C, wherein the organic carrier material comprises a monoester of glycerol and a fatty acid having a carbon chain containing from 12 to 20 carbon atoms. EP-A-0210721 discloses other preferred particulate suds suppressing systems wherein the organic carrier material is a fatty acid or alcohol having a carbon chain containing from 12 to 20 carbon atoms, or a mixture thereof, with a melting point of from 45°C to 80°C.
- Other highly preferred particulate suds suppressing systems are described in copending European Application 91870007.1 in the name of the Procter and Gamble Company which systems comprise silicone antifoam compound, a carrier material, an organic coating material and glycerol at a weight ratio of glycerol : silicone antifoam compound of 1:2 to 3:1. Copending European Application 91201342.0 also discloses highly preferred particulate suds suppressing systems comprising silicone antifoam compound, a carrier material, an organic coating material and crystalline or amorphous aluminosilicate at a weight ratio of aluminosilicate : silicone antifoam compound of 1:3 to 3:1. The preferred carrrier material in both of the above described highly preferred granular suds controlling agents is starch.
- An exemplary particulate suds suppressing system for use herein is a particulate agglomerate component, made by an agglomeration process, comprising in combination
- (i) from 5% to 30%, preferably from 8% to 15% by weight of the component of silicone antifoam compound, preferably comprising in combination polydimethyl siloxane and silica;
- (ii) from 50% to 90%, preferably from 60% to 80% by weight of the component, of carrier material, preferably starch;
- (iii) from 5% to 30%, preferably from 10% to 20% by weight of the component of agglomerate binder compound, where herein such compound can be any compound, or mixtures thereof typically employed as binders for agglomerates, most preferably said agglomerate binder compound comprises a C₁₆-C₁₈ ethoxylated alcohol with a degree of ethoxylation of from 50 to 100; and
- (iv) from 2% to 15%, preferably from 3% to 10%, by weight of C₁₂-C₂₂ hydrogenated fatty acid.
- The compositions herein may also comprise from 0.01% to 10 %, preferably from 0.05% to 0.5% by weight of polymeric dye transfer inhibiting agents.
- The polymeric dye transfer inhibiting agents are preferably selected from polyamine N-oxide polymers, copolymers of N-vinylpyrrolidone and N-vinylimidazole, polyvinylpyrrolidonepolymers or combinations thereof.
- The compositions may contain, but preferably do not contain, additional corrosion inhibitor inhibitors such as benzotriazole and paraffin.
- The compositions of the invention can be formulated in any desirable form such as powders, granulates, pastes, liquids, gels and tablets, granular forms being preferred.
- The bulk density of the granular detergent compositions in accordance with the present invention is typically of at least 650 g/litre, more usually at least 700 g/litre and more preferably from 800 g/litre to 1200 g/litre.
- Bulk density is measured by means of a simple funnel and cup device consisting of a conical funnel moulded rigidly on a base and provided with a flap valve at its lower extremity to allow the contents of the funnel to be emptied into an axially aligned cylindrial cup disposed below the funnel.
- The funnel is 130 mm and 40 mm at its respective upper and lower extremities. It is mounted so that the lower extremity is 140 mm above the upper surface of the base. The cup has an overall height of 90 mm, an internal height of 87 mm and an internal diameter of 84 mm. Its nominal volume is 500 ml.
- To carry out a measurement, the funnel is filled with powder by hand pouring, the flap valve is opened and powder allowed to overfill the cup. The filled cup is removed from the frame and excess powder removed from the cup by passing a straight edged implement e.g. a knife, across its upper edge. The filled cup is then weighed and the value obtained for the weight of powder doubled to provide the bulk density in g/litre. Replicate measurements are made as required.
- The particle size of the components of granular compositions in accordance with the invention should preferably be such that no more that 5% of particles are greater than 1.4mm in diameter and not more than 5% of particles are less than 0.15mm in diameter.
- Generally, if the compositions are in liquid form the liquid should be thixotropic (ie; exhibit high viscosity when subjected to low stress and lower viscosity when subjected to high stress), or at least have very high viscosity, for example, of from 1,000 to 10,000,000 centipoise. In many cases it is desirable to include a viscosity control agent or a thixotropic agent to provide a suitable liquid product form. Suitable thixotropic or viscosity control agents include methyl cellulose, carboxymethylcellulose, starch, polyvinyl, pyrrolidone, gelatin, colloidal silica, and natural or synthetic clay minerals.
- Pasty compositions in accordance with the invention generally have viscosities of about 5,000 centipoise and up to several hundred million centipoise. In order to provide satisfactory pasty compositions a small amount of a solvent or solubilizing agent or of a gel-forming agent can be included. Most commonly, water is used in this context and forms the continuous phase of a concentrated dispersion. Certain nonionic surfactants at high levels form a gel in the presence of small amount of water and other solvents. Such gelled compositions also envisaged in the present invention.
- The pH of a 1% solution of the present compositions is preferably less than 12, preferably from 9.8 to 11.5, most preferably from 10.0 to 11.0.
- The use is provided, in a method for washing soiled tableware, including soiled silverware, of an oxygen-releasing bleach system containing composition including Mn(II) compounds to inhibit the tarnishing of said silverware.
- Any suitable methods for washing or cleaning soiled tableware, including soiled silverware, are envisaged including machine dishwashing methods.
- A preferred machine dishwashing method comprises treating soiled articles selected from crockery, glassware, hollowware and cutlery and mixtures thereof, with an aqueous liquid having dissolved or dispensed therein an effective amount of a machine dishwashing composition in accord with the inevntion. By an effective amount of the machine dishwashing composition it is meant from 8g to 60g of product dissolved or dispersed in a wash solution of volume from 3 to 10 litres, as are typical product dosages and wash solution volumes commonly employed in conventional machine dishwashing methods.
- In the detergent compositions, the abbreviated component identifications have the following meanings:
- XYEZS
- : C1X - C1Y sodium alkyl sulfate condensed with an average of Z moles of ethylene oxide per mole
- Nonionic
- : C₁₃-C₁₅ mixed ethoxylated/propoxylated fatty alcohol with an average degree of ethoxylation of 3.8 and an average degree of propoxylation of 4.5 sold under the tradename Plurafac LF404 by BASF Gmbh (low foaming)
- Silicate
- : Amorphous Sodium Silicate (SiO₂:Na₂O ratio = 2.0)
- Carbonate
- : Anhydrous sodium carbonate
- Phosphate
- : Sodium tripolyphosphate
- MA/AA
- : Copolymer of 1:4 maleic/acrylic acid, average molecular weight about 80,000
- AA
- : Homopolymer of polyacrylic acid having an average molecular weight of 4,500
- Polyacrylate
- : Polyacrylate homopolymer with an average molecular weight of 8,000 sold under the tradename PA30 by BASF GmbH
- Citrate
- : Tri-sodium citrate dihydrate
- PB4
- : Anhydrous sodium perborate tetrahydrate
- Percarbonate
- : Anhydrous sodium percarbonate bleach of empirical formula 2Na₂CO₃.3H₂O₂ coated with a mixed salt of formula Na₂SO₄.n.Na₂CO₃ where n is 0.29 and where the weight ratio of percarbonate to mixed salt is 39:1
- TAED
- : Tetraacetyl ethylene diamine
- Caprolactam
- : Benzoyl caprolactam
- Paraffin
- : Paraffin oil sold under the tradename Winog 70 by Wintershall.
- Protease
- : Proteolytic enzyme sold under the tradename Savinase by Novo Industries A/S (approx 2% enzyme activity).
- Amylase
- : Amylolytic enzyme sold under the tradename Termamyl 60T by Novo Industries A/S (approx 0.9% enzyme activity)
- Lipase
- : Lipolytic enzyme sold under the tradename Lipolase by Novo Industries A/S (approx 2% enzyme activity)
- DETPMP
- : Diethylene triamine penta (methylene phosphonic acid), marketed by Monsanto under the Trade name Dequest 2060
- HEDP
- : ethane 1-hydroxy-1,1 diphosphonate (sodium salt)
- EDDS
- : Ethylenediamine disuccinic acid(S,S isomer)
- Granular Suds Suppressor
- : 12% Silicone/silica, 18% stearyl alcohol,70% starch in granular form
- Sulphate
- : Anhydrous sodium sulphate.
- In the following examples all levels of enzyme quoted are expressed as % active enzyme by weight of the composition.
-
- To each of base compositions A to F the following Mn(II) compounds were added at the indicated levels to give compositions in accord with the invention.
Mn(II) compound Level of addition (by weight) Mn(II) SO₄ 0.1 MnCl₂ 0.1 Mn(II) HEDP 0.13 Mn(II) EDDS 0.13 Mn(II) DETPMP 0.13 - The following comparative testing was conducted; base composition A of Example I was compared for anti-silver tarnishing performance to reference Compositions G and H having the base Composition A but additionally containing 0.13% MnSO₄ and 0.13% Mn(II)EDDS respectively.
- The testing involved machine testing, using a Bosch Siemens dishwasher, 20g product dosage, 65°C economy cycles, and 14 cycles.
- Performance was graded by 4 expert panellists through visual inspection according to the following scale:
where - 0 = no tarnish (shiny silver)
- 1 = very slight tarnish
- 2 = tarnish
- 3 = very tarnished
- 4 = severe tarnish (black coverage)
- Results were as follows : (average of the 4 gradings from the panellists)
Composition A Composition G Composition H 2.0 <0.5 <0.5 -
wherein said silica/silicone antifoam compound is incorporated at a level of from 5% to 50%, preferably 10% to 40% by weight;
Claims (10)
- A bleaching composition containing(a) an oxygen-releasing bleach system comprising a hydrogen peroxide source and a peroxyacid bleach precursor compound selected from the group consisting of(i) an N-acylated lactam bleach precursor of the formula:
- A bleaching composition according to Claim 1 wherein said Mn(II) compound is an Mn(II) salt or complex with an organic ligand.
- A bleaching composition according to Claim 2 wherein said organic ligand has heavy metal ion sequestering properties.
- A bleaching composition according to Claim 3 wherein said organic ligand is selected from the group consisting of ethylenediamine disuccinic acid, ethylenediamine diglutaric acid, 2-hydroxypropylenediamine disuccinic acid and mixtures thereof.
- A bleaching composition according to Claim 2 wherein said organic ligand has crystal growth inhibiting properties.
- A bleaching composition according to Claim 5 wherein said organic ligand is an organo diphosphonic acid.
- A composition according to any of Claims 1-6 wherein the level of oxygen releasing bleach system is such that the level of available oxygen measured according to the method herein is from 0.3% to 2.5%.
- A composition according to any of Claims 1-7 wherein the rate of release of the available oxygen is such that the available oxygen is completely released from the composition in a time interval of from 3.5 minutes to 10 minutes, using the test protocol described in the present description.
- A composition according to any of Claims 1-8 wherein said hydrogen peroxide source is an inorganic perhydrate salt.
- The use, in a method for washing soiled tableware, including soiled silverware, of a composition containing an oxygen-releasing bleach system wherein said composition contains an Mn(II) compound to inhibit the tarnishing of said silverware.
Applications Claiming Priority (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US21018694A | 1994-03-17 | 1994-03-17 | |
| US210186 | 1994-03-17 | ||
| GB9410377A GB9410377D0 (en) | 1994-05-24 | 1994-05-24 | Detergent compositions |
| GB9410377 | 1994-05-24 | ||
| GB9418567 | 1994-09-15 | ||
| GB9418567A GB9418567D0 (en) | 1994-03-17 | 1994-09-15 | Bleaching compositions |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| EP0672749A1 true EP0672749A1 (en) | 1995-09-20 |
Family
ID=27267194
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP95301219A Ceased EP0672749A1 (en) | 1994-03-17 | 1995-02-24 | Bleaching compositions |
Country Status (2)
| Country | Link |
|---|---|
| EP (1) | EP0672749A1 (en) |
| CA (1) | CA2144103C (en) |
Cited By (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1995033043A1 (en) * | 1994-06-01 | 1995-12-07 | The Procter & Gamble Company | Bleach compositions comprising oleoyl sarcosinate surfactants |
| WO1996017920A1 (en) * | 1994-12-05 | 1996-06-13 | Henkel Kommanditgesellschaft Auf Aktien | Activator mixtures for inorganic per compounds |
| GB2307914A (en) * | 1995-12-06 | 1997-06-11 | Procter & Gamble | Detergent compositions |
| WO2005014768A1 (en) * | 2003-07-30 | 2005-02-17 | Degussa Ag | Bleaching-action dishwasher detergent with improved yellowing resistance and process for its production |
| US7435714B2 (en) | 2002-12-20 | 2008-10-14 | Evonik Degussa Gmbh | Liquid detergent and cleaning agent composition comprising a multi-coated bleach particle |
| US7588697B2 (en) | 2003-05-07 | 2009-09-15 | Evonik Degussa Gmbh | Coated sodium percarbonate granules with improved storage stability |
| US7718592B2 (en) | 2004-11-11 | 2010-05-18 | Degussa, Gmbh | Sodium percarbonate particles having a shell layer comprising thiosulfate |
| CN101469127B (en) * | 2007-12-24 | 2011-11-09 | 上海杰事杰新材料(集团)股份有限公司 | Preparation of polyolefin nylon in situ alloy |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0132860A1 (en) * | 1983-06-20 | 1985-02-13 | Unilever N.V. | Detergent bleach compositions |
| WO1994019445A1 (en) * | 1993-02-22 | 1994-09-01 | Unilever N.V. | Machine dishwashing composition |
| WO1994021605A1 (en) * | 1993-03-22 | 1994-09-29 | Unilever N.V. | Peroxyacids |
| WO1994028105A1 (en) * | 1993-05-20 | 1994-12-08 | The Procter & Gamble Company | Bleaching compounds comprising n-acyl caprolactam and alkanoyloxybenzene sulfonate bleach activators |
| WO1995000626A1 (en) * | 1993-06-24 | 1995-01-05 | The Procter & Gamble Company | Bleaching compounds comprising acyl valerolactam bleach activators |
-
1995
- 1995-02-24 EP EP95301219A patent/EP0672749A1/en not_active Ceased
- 1995-03-07 CA CA 2144103 patent/CA2144103C/en not_active Expired - Fee Related
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0132860A1 (en) * | 1983-06-20 | 1985-02-13 | Unilever N.V. | Detergent bleach compositions |
| WO1994019445A1 (en) * | 1993-02-22 | 1994-09-01 | Unilever N.V. | Machine dishwashing composition |
| WO1994021605A1 (en) * | 1993-03-22 | 1994-09-29 | Unilever N.V. | Peroxyacids |
| WO1994028105A1 (en) * | 1993-05-20 | 1994-12-08 | The Procter & Gamble Company | Bleaching compounds comprising n-acyl caprolactam and alkanoyloxybenzene sulfonate bleach activators |
| WO1995000626A1 (en) * | 1993-06-24 | 1995-01-05 | The Procter & Gamble Company | Bleaching compounds comprising acyl valerolactam bleach activators |
Cited By (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1995033043A1 (en) * | 1994-06-01 | 1995-12-07 | The Procter & Gamble Company | Bleach compositions comprising oleoyl sarcosinate surfactants |
| WO1996017920A1 (en) * | 1994-12-05 | 1996-06-13 | Henkel Kommanditgesellschaft Auf Aktien | Activator mixtures for inorganic per compounds |
| GB2307914A (en) * | 1995-12-06 | 1997-06-11 | Procter & Gamble | Detergent compositions |
| US7435714B2 (en) | 2002-12-20 | 2008-10-14 | Evonik Degussa Gmbh | Liquid detergent and cleaning agent composition comprising a multi-coated bleach particle |
| US7588697B2 (en) | 2003-05-07 | 2009-09-15 | Evonik Degussa Gmbh | Coated sodium percarbonate granules with improved storage stability |
| WO2005014768A1 (en) * | 2003-07-30 | 2005-02-17 | Degussa Ag | Bleaching-action dishwasher detergent with improved yellowing resistance and process for its production |
| US7718592B2 (en) | 2004-11-11 | 2010-05-18 | Degussa, Gmbh | Sodium percarbonate particles having a shell layer comprising thiosulfate |
| CN101469127B (en) * | 2007-12-24 | 2011-11-09 | 上海杰事杰新材料(集团)股份有限公司 | Preparation of polyolefin nylon in situ alloy |
Also Published As
| Publication number | Publication date |
|---|---|
| CA2144103C (en) | 2000-08-01 |
| CA2144103A1 (en) | 1995-09-18 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US5698504A (en) | Machine dishwashing composition containing oxygen bleach and paraffin oil and benzotriazole compound silver tarnishing inhibitors | |
| EP0706559B1 (en) | Machine dishwashing composition containing oxygen bleach and paraffin oil and benzotriazole compound silver tarnishing inhibitors | |
| US5695679A (en) | Detergent compositions containing an organic silver coating agent to minimize silver training in ADW washing methods | |
| US5965505A (en) | Detergents containing a heavy metal sequestrant and a delayed release peroxyacid bleach system | |
| EP0755429B1 (en) | Detergents containing a heavy metal sequestrant and a delayed release peroxyacid bleach system | |
| US5755992A (en) | Detergents containing a surfactant and a delayed release peroxyacid bleach system | |
| EP0755434B1 (en) | Detergents containing a builder and a delayed release enzyme | |
| CA2146051C (en) | Bleaching composition | |
| GB2283494A (en) | Machine dishwashing | |
| CA2144103C (en) | Bleaching compositions | |
| EP0755435A1 (en) | Detergent compositions | |
| US6559113B2 (en) | Detergents containing a builder and a delayed released enzyme | |
| WO1995028467A1 (en) | Detergents containing an enzyme and a delayed release peroxyacid bleaching system | |
| WO1995012652A1 (en) | Detergent compositions | |
| CA2187436C (en) | Detergents containing a surfactant and a delayed release peroxyacid bleach source | |
| CA2187306C (en) | Detergents containing a surfactant and a delayed release enzyme | |
| WO1997023593A1 (en) | Detergent composition comprising enzyme and delayed release mechanism | |
| EP0755431A1 (en) | Detergents containing a builder and a delayed release peroxyacid bleach source | |
| EP0679177A4 (en) | ||
| CA2151659C (en) | Detergent compositions | |
| EP0759056A1 (en) | Detergent compositions | |
| GB2295625A (en) | Bleaching composition for machine dishwashing | |
| EP0795004A1 (en) | Machine dishwashing detergent compositions containing silicate mixtures | |
| MXPA96004782A (en) | Detergent compositions |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): AT BE CH DE DK ES FR GB GR IE IT LI NL PT SE |
|
| 17P | Request for examination filed |
Effective date: 19960126 |
|
| 17Q | First examination report despatched |
Effective date: 19981203 |
|
| GRAG | Despatch of communication of intention to grant |
Free format text: ORIGINAL CODE: EPIDOS AGRA |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: THE APPLICATION HAS BEEN REFUSED |
|
| 18R | Application refused |
Effective date: 20010219 |