EP0566323A2 - Developer for silver halide photographic light-sensitive material - Google Patents
Developer for silver halide photographic light-sensitive material Download PDFInfo
- Publication number
- EP0566323A2 EP0566323A2 EP93302781A EP93302781A EP0566323A2 EP 0566323 A2 EP0566323 A2 EP 0566323A2 EP 93302781 A EP93302781 A EP 93302781A EP 93302781 A EP93302781 A EP 93302781A EP 0566323 A2 EP0566323 A2 EP 0566323A2
- Authority
- EP
- European Patent Office
- Prior art keywords
- developer
- group
- carbon atoms
- compound
- mol
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
Classifications
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03C—PHOTOSENSITIVE MATERIALS FOR PHOTOGRAPHIC PURPOSES; PHOTOGRAPHIC PROCESSES, e.g. CINE, X-RAY, COLOUR, STEREO-PHOTOGRAPHIC PROCESSES; AUXILIARY PROCESSES IN PHOTOGRAPHY
- G03C5/00—Photographic processes or agents therefor; Regeneration of such processing agents
- G03C5/26—Processes using silver-salt-containing photosensitive materials or agents therefor
- G03C5/29—Development processes or agents therefor
- G03C5/305—Additives other than developers
Definitions
- the present invention relates to a photographic developer, more specifically a developer for a silver halide photographic light-sensitive material capable of forming an image of high sensitivity and high density without silver sludge formation therein even when processed at low replenishing rates.
- Sulfites such as sodium sulfite and potassium sulfite, used as antioxidants for photographic developers, have an ability dissolving silver halide.
- this silver sludge in suspension in the developer, adheres to the film and the rollers and belts of the automatic processing machine.
- Japanese Patent Publication Open to Public Inspection (hereinafter referred to as Japanese Patent O.P.I. Publication) No. 114035/1983, using a thiouracil, Japanese Patent O.P.I. Publication No. 2043/1988, using a mercaptobenzoic acid, Japanese Patent Examined Publication No. 14953/1972, using an aliphatic mercaptocarboxylic acid, Japanese Patent O.P.I. Publication No. 178959/1987, Japanese Patent O.P.I. Publication No. 51844/1991, using a disulfide, Japanese Patent O.P.I. Publication No. 26136/1971, using a sulfur-containing a-amino acid, and various mercaptoazoles, all of which are compounds likely to form a water-soluble silver salt.
- a developer for developing silver halide photographic light-sensitive material which contains a compound represented by the following formula 1 or 2: wherein R 1 and R 2 independently represent a hydrogen atom or an alkyl group having 1 to 3 carbon atoms. R 1 and R 2 are not hydrogen atoms at the same time.
- R 3 and R 4 independently represent a hydrogen atom or an alkyl group having 1 to 3 carbon atoms;
- R 5 represents a hydroxyl group, an amino group or an alkyl group having 1 to 3 carbon atoms.
- R 6 and R 7 independently represent a hydrogen atom, an alkyl group having 1 to 5 carbon atoms, an acyl group having up to 18 carbon atoms or a -COOM 2 group.
- R 6 and R 7 are not hydrogen atoms at the same time.
- M 1 represents a hydrogen atom, an alkali metal atom or an ammonium group, and m represents 0, 1 or 2.
- M 2 is a hydrogen atom, an alkyl group having 1 to 4 carbon atoms, an alkali metal atom, an aryl group or an aralkyl group having not more than 15 carbon atoms.
- Alkyl groups having 1 to 4 carbon atoms as described above include a methyl group, an ethyl group, a propyl group and a butyl group.
- acyl groups having up to 18 carbon atoms include an acetyl group and a benzoyl group.
- aralkyl groups having up to 15 carbon atoms include a benzyl group and a phenetyl group.
- Aryl groups include a phenyl group and a naphthyl group.
- alkali metal atoms for M 1 examples include sodium ion and potassium ion.
- Strecker's method of amino acid synthesis known to be an amino acid synthesis method, wherein amino acid acetylation is achieved by alternate addition of alkali and acetic anhydride in an aqueous solution.
- the above compounds of the present invention may be used singly or in combination. It is also acceptable to use in combination at least one kind of the compound of formula 1 and at least one kind of the compound of formula 2.
- the inventive compounds of formulas 1 and 2 are used in amounts of 1 x 10- s to 3 x 10- 2 mol, preferably 1 x 10- 4 to 1 x 10- 2 mol per liter of developer.
- the replenishing rate for the developer containing a compound of the present invention is normally 0.05 to 0.65 I, preferably 0.10 to 0.35 I per m 2 of light-sensitive material, though it varies depending on the kinds of light-sensitive material, automatic processing machine, and other factors.
- the developing through drying processes are completed within 90 seconds when an automatic processing machine capable of developing, fixing and washing or stabilizing is used to process a silver halide light-sensitive material relating to the present invention.
- the time from initiation of immersion of the tip of the light-sensitive material in the developer, via the various processes, to discharge of the same tip from the drying zone is preferably not longer than 90 seconds, more preferably not longer than 60 seconds.
- Fixing temperature and time are preferably about 20 to 50°C and 6 to 20 seconds, more preferably 30 to 40°C and 6 to 15 seconds.
- developing time is normally 5 to 45 seconds, preferably 8 to 30 seconds, and developing temperature is preferably 25 to 50°C, more preferably 30 to 40°C.
- Drying may be achieved by hot air blow at normally 35 to 100°C, preferably 40 to 80°C.
- a drying zone equipped with a far infrared heating means may be arranged in the automatic processing machine.
- the automatic processing machine may be equipped with a mechanism for adding eitherwateror an acidic rinsing solution having no fixing capability to the light-sensitive material, among the above developing, fixing and washing processes such described as in Japanese Patent O.P.I. Publication No. 264953/199.
- the automatic processing machine may also have therein equipment for preparing developers and fixers.
- the developer of the present invention preferably contains as a developing agent a 1,4-dihydroxybenzene compound or, if necessary, a p-aminophenol compound and/or a pyrazolidone compound.
- 1,4-dihydroxybenzenes include hydroquinone, chlorohydroquinone, bromohydroquinone, isopropylhydroquinone, methylhydroquinone, 2,3-dichlorohydroquinone, 2,5-dichlorohydroquinone, 2,3-dibromohydroquinone, 2,5-dimethylhydroquinone and hydroquinone monosulfonate, with preference given to hydroquinone.
- p-aminophenol-based developing agents include N-methyl-p-aminophenol, p-aminophenol, N-((3-hydroxyethyl)-p-aminophenol, N-(4-hydroxyphenyl)glycine, 2-methyl-p-aminophenol and p-benzylaminophenol, with preference given to N-methyl-p-aminophenol.
- the amount of 1,4-dihydroxybenzene added is 0.01 to 0.7 mol, preferably 0.1 to 0.5 mol per liter of developer.
- the amounts of p-aminophenol compound and pyrazolidone compound added are 0.0005 to 0.2 mol, preferably 0.001 to 0.1 mol per liter of developer.
- Examples of sulfites used in the developer of the present invention include sodium sulfite, potassium sulfite, lithium sulfite, ammonium sulfite, sodium bisulfite and potassium metasulfite.
- the amount ofthese sulfites used is 0.1 to 2.0 mol, preferably 0.1 to 1.0 mol per liter of developer. Also, the upper limit is preferably 3.0 mol per liter of developer for a concentrated developer.
- the developer may contain a chelating agent having an iron ion chelating stability constant of over 8.
- the iron ion mentioned herein is ferricion (Fe 3+ ).
- Chelating agents having an iron ion chelating stability constant of over 8 include organic carboxylic acid chelating agents, organic phosphoric acid chelating agents, inorganic phosphoric acid chelating agents and polyhydroxyl compounds.
- such chelating agents include ethylenediamine-di-o-hydroxyphenylacetic acid, triethylenete- tramineacetic acid, diaminopropanetetraacetic acid, nitrilotriacetic acid, hydroxyethylethylenediaminetriacetic acid, dihydroxyethylglycine, ethylenediaminediacetic acid, ethylenediaminedipropionic acid, iminodiacetic acid, diethylenetriaminepentaacetic acid, hydroxyethyliminodiacetic acid, 1,3-diamino-2-propanoltetraacetic acid, transcyclohexanediaminetetraacetic acid, ethylenediaminetetraacetic acid, glycol ether amine tetraacetic acid, ethylenediamine-N,N,N',N'-tetrakismethylenephosphonic acid, nitrilo-N,N,N-trimethylenephosphonic acid, 1-hydroxyethyi
- the developer of the present invention may contain a hardener which enhances the film physical properties by reacting with the gelatin in the light-sensitive material during the developing process.
- hardeners include glutaraldehyde, a-methylglutaraldehyde, (3-methylglutaraldehyde, maleic dialdehyde, succinic dialdehyde, methoxysuccinic dialdehyde, methylsuccinic dialdehyde, ⁇ -methoxy- ⁇ -ethoxyglutaraldehyde, an-butoxyglutaraldehyde, a,a-dimethoxysuccinic dialdehyde, ⁇ -isopropylsuccinic dialdehyde, a,a-diethylsuccinic dialdehyde, butylmaleic dialdehyde and bisulfite adducts thereof.
- the developer may further contain developing inhibitors such as sodium bromide and potassium iodide, organic solvents such as ethylene glycol, diethylene glycol, triethylene glycol, dimethylformamide, methyl cellosolve, hexylene glycol, ethanol and methanol, mercapto compounds such as 1-phenyl-5-mercaptotetrazole and sodium 2-mercaptobenzimidazole-5-sulfonate, and antifoggants such as 5-methylbenzotriazole and other benzotriazole compounds.
- organic solvents such as ethylene glycol, diethylene glycol, triethylene glycol, dimethylformamide, methyl cellosolve, hexylene glycol, ethanol and methanol
- mercapto compounds such as 1-phenyl-5-mercaptotetrazole and sodium 2-mercaptobenzimidazole-5-sulfonate
- antifoggants such as 5-methylbenzotriazole and other benzotriazole compounds.
- Other additives such as
- the pH of the developer is normally 9.0 to 12, preferably 9.0 to 11.5.
- alkali or buffer used for pH adjustment include pH regulators such as sodium hydroxide, potassium hydroxide, sodium carbonate, potassium carbonate, boric acid, sodium tertiary phosphate and potassium tertiary phosphate.
- a fixer for fixing the light sensitive material may contain a fixing agent such as sodium thiosulfate or ammonium thiosulfate, with preference given to ammonium thiosulfate from the viewpoint of fixing speed.
- fixing agents are usually used in amounts of about 0.1 to 6 mol/liter.
- the fixer may also contain a water-soluble aluminum salt as a hardener, such as aluminum chloride, aluminum sulfate or potassium alum.
- a water-soluble aluminum salt such as aluminum chloride, aluminum sulfate or potassium alum.
- the fixer may incorporate malic acid, tartaric acid, citric acid, gluconic acid and derivatives thereof, singly or in combination. These compounds are effective when contained at not less than 0.001 mol per liter of fixer, with a greater effect obtained at 0.005 to 0.03 mol per liter of fixer.
- Fixer pH is normally not lower than 3.8, preferably 4.2 to 7.0. In view of hardening in the fixer, sulfurous acid gas odor the pH is more preferably 4.3 to 4.8.
- Silver halide photographic light-sensitive material emulsions to which the present invention is applicable include medical radiographic materials, photographic materials for printing plate making and direct positive photographic materials.
- the silver halide photographic light-sensitive material to which the present invention is applied may be comprised a silver halide such as silver iodobromide, silver iodochloride or silver iodochlorobromide, with preference given to silver iodobromide from the viewpoint of high sensitivity.
- a silver halide such as silver iodobromide, silver iodochloride or silver iodochlorobromide, with preference given to silver iodobromide from the viewpoint of high sensitivity.
- Emulsions for the silver halide photographic light-sensitive material include monodispersed grains of high inner iodine content such as those disclosed in Japanese Patent O.P.I. Publication Nos. 177535/1984, 802237/1986, 132943/1986 and 49751/1988. Crystalline habit may be cubic, tetradecahedral or octahedral, and intermediate (111) and (100) planes may be present in any ratio.
- the emulsion may be of tabular grains having an aspect ratio of not less than 2. Such tabular grains have advantages such as improvements in spectral sensitizing efficiency, image granularity and sharpness, and are disclosed in British Patent No. 2,112,157, US Patent Nos. 4,439,520,4,433,048,4,414,310 and 4,434,226, and Japanese Patent O.P.I. Publication Nos. 113927/1983, 127921/1983, 138342/1988, 284272/1988 and 305343/1988.
- the emulsion can be prepared as described in these publications.
- Additives and other substances used in the silver halide photographic light-sensitive material include those described in Research Disclosure Nos. 17643 (December, 1978), 18716 (November, 1979) and 308119 (December, 1989) (hereinafter referred to as RD17643, RD18716 and RD308119, respectively). The following table shows where they are described.
- the resulting reaction mixture was desalinized at 40°C, using an aqueous solution of Demol-N (produced by Kao Atlas) and an aqueous solution of magnesium sulfate, after which it was re-dispersed in an aqueous gelatin solution, to yield a seed emulsion.
- grains were grown as follows: First, the seed emulsion was dispersed in an aqueous gelatin solution being kept at 40°C, and aqueous ammonia and acetic acid were added to obtain a pH of 9.7. An aqueous solution of ammoniacal silver nitrate and an aqueous solution of potassium bromide and potassium iodide were then added to the dispersion by the double jet method, while maintaining a pAg of 7.3 and a pH of 9.7, to yield a layer containing 35 mol% silver iodide. Next, another aqueous solution of ammoniacal silver nitrate and an aqueous solution of potassium bromide were added by the double jet method.
- the pAg was kept at 9.0, with the pH varied continuously over the range of 9.0 to 8.0. The pAg was then changed to 11.0, and while keeping the pH at 8.0, grains were grown up to the desired grain size. Subsequently, acetic acid was added to obtain a pH of 6.0, after which 400 mg of the anhydride of 5,5'-dichloro-9-ethyl-3,3'-di(3-sulfopropyl)oxacarbocyanine sodium salt was added per mol of silver halide. This mixture was then desalinized with the above aqueous solution of Demol-N and aqueous solution of magnesium sulfate, and then re-dispersed in a gelatin solution.
- Monodispersed silver iodobromide emulsions A, B and C comprising tetradecahedral grains with round tips having an average silver iodide content of 2.0 mol%, were thus prepared, which had average grain sizes of 0.40, 0.65 and 1.00 ⁇ m and variation coefficients of grain size distribution (a/ r ) of 0.17, 0.16 and 0.16, respectively, in which ⁇ is the standard deviation of grain size distribution and r is the average grain size.
- a hexagonal tabular seed emulsion was prepared as follows:
- solution A After stopping the addition of solutions Band C, the temperature of solution A was increased to 60°C over a period of 60 minutes, and solutions B and C were again added by the double jet method at a flow rate of 68.5 ml/min over a period of 50 minutes, while keeping the silver potential (determined using a silver ion selective electrode in combination with a saturated silver-silver chloride electrode as a reference electrode) at +6 mV using solution D.
- the silver potential was kept at +25 mV using solution D.
- the following spectral sensitizing dyes A and B were added in amounts of 300 mg and 15 mg, respectively, per mol of silver halide.
- a 200:1 (w/w) mixture of the above sensitizing dyes A and B was added at 975 mg, 600 mg, 390 mg and 500 mg per mol of silver halide, respectively.
- emulsion II Of the four ripened emulsions, A, B and C were mixed in a weight ratio of 15:65:20, to yield emulsion I, while emulsion D, designated as emulsion II, was used as such.
- aqueous solution a 9.3% aqueous solution of gelatin containing 1.35 kg of an anionic surfactant AS was prepared. This solution is designated as the aqueous solution.
- the oily solution and the aqueous solution were placed in a dispersing vessel and dispersed while keeping the liquid temperature at 40°C.
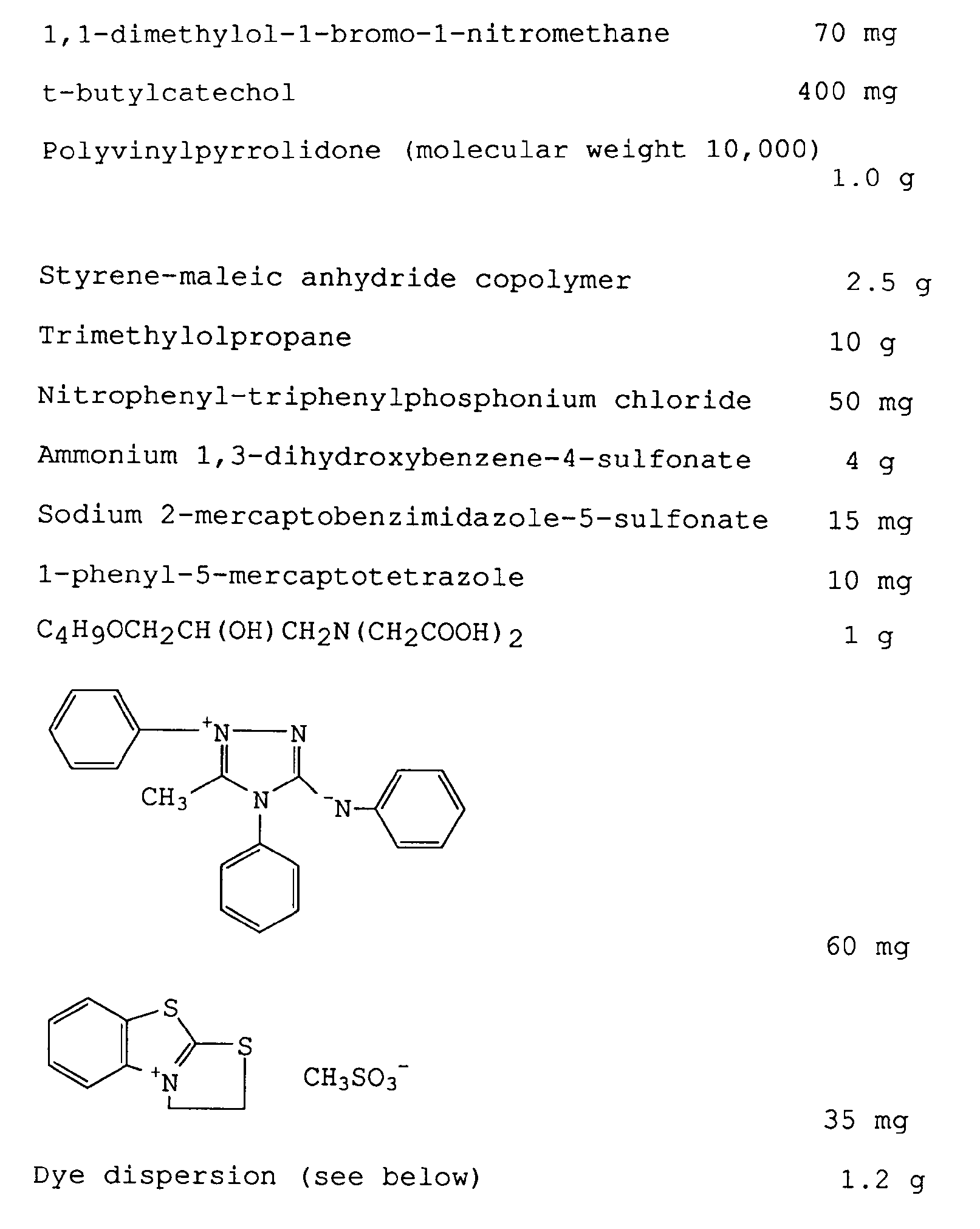
- To the resulting dispersion were added appropriate amounts of phenol and 1,1-dimethylol-bromo-1-nitromethane, and water was added to 240 kg.
- composition of the protective layer are as follows The amount of addition are shown per liter of coating solution.
- the resulting coating solution was coated and dried uniformly on a blue-colored subbed polyethylene terephthalate film base 180 ⁇ m thick.
- both the emulsion layer and the protective layer were coated simultaneously at a coating speed of 90 m per minute, to yield samples, wherein coating rates were 1.7 g/m 2 as of silver for the emulsion layer and 0.99 g/m 2 as of gelatin for the protective layer.
- developing sample 1 was obtained from emulsion I, and developing sample 2 obtained from emulsion II.
- compositions of the developer and fixer used in the present invention are as follows:
- fixer replenisher To prepare the fixer, parts A and B were added at the same time to about 5 liters of water, and while stirring and dissolving the mixture, water was added to 18 liters, and sulfuric acid or NaOH were added to obtain a pH of 4.4, to yield a fixer replenisher.
- Comparative Compounds 1 - 12 were used as comparative compounds.
- Processing temperatures were 35°C for developing, 33°C for fixation, 20°C for washing and 50°C for drying.
- the sample inserted between two sheets of fluorescent sensitizing paper KO-250, manufactured by Konica Corp., was subjected to exposure through an aluminum wedge of at a tube voltage of 80 kV, a tube amperage of 100 mAand an irradiation time of 50 msec, after which it was processed using the above automatic processing machines.
- the reciprocal of the exposure amount required to obtain a density of base density + fog density + 1.0 was calculated as a percent sensitivity relative to the sensitivity of sample No. 1 processed with the above developer and fixer of basic composition using automatic processing machine No. 2 (experiment No. 15).
- the density in the unexposed portion of the exposed sample was determined using Konica PDA-65 densitometer, and the base density was subtracted therefrom to obtain the fog density, and the maximum density was expressed as Dm.
- the developing samples used were the above sample Nos. 1 and 2.
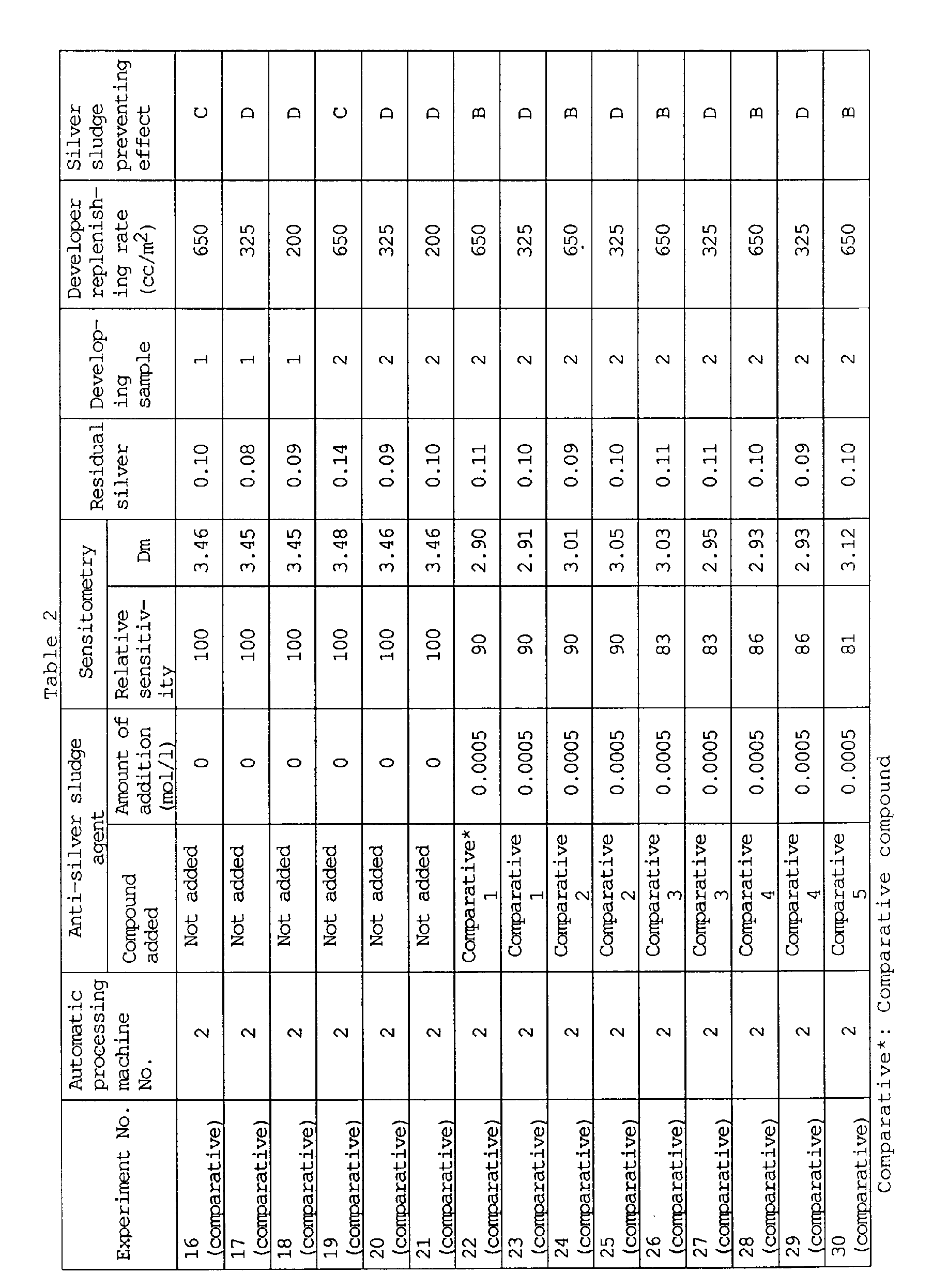
- Table 1 shows that the addition of a compound of the present invention to the developer did not lower the sensitivity or Dm (maximum density), while the addition of a comparative compound significantly lowered the sensitivity, though it had a slight preventive effect on silver sludge at high replenishing rates.
- Results of experiment Nos. 1 through 10 demonstrate that developers containing a compound of the present invention remain excellently effective without deterioration of the silver sludge preventing effect even when the replenishing rate is reduced to 200 ml/m 2 . Also, the results of experiment Nos. 12 and 14 demonstrate that there is no difference in silver sludge preventing effect among different automatic processing machines.
Abstract
wherein R1, R2 R3, and R4 are each a hydrogen atom or an alkyl group having 1 to 3 carbon atoms provided that both of R1 and R2 are not hydrogen atoms at the same time ; R5 is a hydroxy group, an amino group or an alkyl group having 1 to 3 carbon atoms ; R6 and R7 are each a hydrogen atom, an alkyl group having 1 to 5 carbon atoms, an acyl group having 1 to 18 carbon atoms or a -COOM2 group, provided that both of R1 and R2 are not hydrogen atoms at the same time, in the above M1 is a hydrogen atom, an alkali metal atom or an ammonium group ; m is an integer 0, 1 or 2 ; M2 is a hydrogen atom, an alkyl group having 1 to 4 carbon atoms, an alkali metal atom, an aryl group or an aralkyl group having not more than 15 carbon atoms. Formation of silver sludge in the developer is prevented.
Description
- The present invention relates to a photographic developer, more specifically a developer for a silver halide photographic light-sensitive material capable of forming an image of high sensitivity and high density without silver sludge formation therein even when processed at low replenishing rates.
- Sulfites such as sodium sulfite and potassium sulfite, used as antioxidants for photographic developers, have an ability dissolving silver halide.
- This results in the formation of a large amount of a sulfite-silver complex salt upon light-sensitive material processing, which complex salt elutes in the developer. The eluted silver complex is easily reduced by the developing agent, resulting in the accumulation of precipitated silver known as silver sludge.
- In continuous processing using an automatic processing machine, this silver sludge, in suspension in the developer, adheres to the film and the rollers and belts of the automatic processing machine.
- This can cause serious failures such as yellow-brown streak stains and flaws on the film being transported.
- In recent years, processing solution retention in developing machine tanks has increased as the photographic processing solution replenishing rate has been reduced to meet the requirements related to environmental conservation. Thus the amount of silver sludge accumulated has increased.
- Traditionally, there have been proposed a large number of arts for prevention of silver sludge, including Japanese Patent Publication Open to Public Inspection (hereinafter referred to as Japanese Patent O.P.I. Publication) No. 114035/1983, using a thiouracil, Japanese Patent O.P.I. Publication No. 2043/1988, using a mercaptobenzoic acid, Japanese Patent Examined Publication No. 14953/1972, using an aliphatic mercaptocarboxylic acid, Japanese Patent O.P.I. Publication No. 178959/1987, Japanese Patent O.P.I. Publication No. 51844/1991, using a disulfide, Japanese Patent O.P.I. Publication No. 26136/1971, using a sulfur-containing a-amino acid, and various mercaptoazoles, all of which are compounds likely to form a water-soluble silver salt.
- However, many of these compounds have the following drawbacks, and few are satisfactory from the viewpoint of practical use.
- 1) Air oxidation degrades the sludge preventing effect of the compounds in the developer.
- 2) Large amounts must be used to obtain the desired sludge preventing effect.
- 3) Use in large amounts deteriorates film sensitivity and gamma value.
- 4) Malodor is generated.
- 5) Expensive for processing solutions.
- There has been strong demand for the development of a new art free of these drawbacks.
- It is an object of the present invention to provide a developer which allows easy obtainment of an image of excellent finish quality without silver sludge formation, photographic developer stain and roller/belt stain even when used at low replenishing rates in continuous processing of a large amount of light-sensitive material using an automatic processing machine.
- It is another object of the present invention to provide a silver halide photographic light-sensitive material developer having a sludge-preventing effect without affecting the photographic performance. The other objects of the present invention will become obvious through the following description.
- The above objects of the present invention are accomplished by the present invention described as follows:
- The objects are accomplished by a developer for developing silver halide photographic light-sensitive material which contains a compound represented by the following formula 1 or 2:
wherein R1 and R2 independently represent a hydrogen atom or an alkyl group having 1 to 3 carbon atoms. R1 and R2 are not hydrogen atoms at the same time. R3 and R4 independently represent a hydrogen atom or an alkyl group having 1 to 3 carbon atoms; R5 represents a hydroxyl group, an amino group or an alkyl group having 1 to 3 carbon atoms. R6 and R7 independently represent a hydrogen atom, an alkyl group having 1 to 5 carbon atoms, an acyl group having up to 18 carbon atoms or a -COOM2 group. R6 and R7 are not hydrogen atoms at the same time. M1 represents a hydrogen atom, an alkali metal atom or an ammonium group, and m represents 0, 1 or 2. M2 is a hydrogen atom, an alkyl group having 1 to 4 carbon atoms, an alkali metal atom, an aryl group or an aralkyl group having not more than 15 carbon atoms. - The present invention is described in detail below. Alkyl groups having 1 to 4 carbon atoms as described above include a methyl group, an ethyl group, a propyl group and a butyl group.
- Examples of acyl groups having up to 18 carbon atoms include an acetyl group and a benzoyl group. Examples of aralkyl groups having up to 15 carbon atoms include a benzyl group and a phenetyl group. Aryl groups include a phenyl group and a naphthyl group.
- Examples of alkali metal atoms for M1 include sodium ion and potassium ion.
- Although various methods of synthesis can be used to synthesize the above compounds of the present invention, Strecker's method of amino acid synthesis, known to be an amino acid synthesis method, can be used, wherein amino acid acetylation is achieved by alternate addition of alkali and acetic anhydride in an aqueous solution.
-
- The above compounds of the present invention may be used singly or in combination. It is also acceptable to use in combination at least one kind of the compound of formula 1 and at least one kind of the compound of formula 2.
- The inventive compounds of formulas 1 and 2 are used in amounts of 1 x 10-s to 3 x 10-2 mol, preferably 1 x 10-4 to 1 x 10-2 mol per liter of developer. The replenishing rate for the developer containing a compound of the present invention is normally 0.05 to 0.65 I, preferably 0.10 to 0.35 I per m2 of light-sensitive material, though it varies depending on the kinds of light-sensitive material, automatic processing machine, and other factors.
- Preferably, the developing through drying processes are completed within 90 seconds when an automatic processing machine capable of developing, fixing and washing or stabilizing is used to process a silver halide light-sensitive material relating to the present invention.
- In otherwords, the time from initiation of immersion of the tip of the light-sensitive material in the developer, via the various processes, to discharge of the same tip from the drying zone (what is called "dry to dry time") is preferably not longer than 90 seconds, more preferably not longer than 60 seconds.
- Fixing temperature and time are preferably about 20 to 50°C and 6 to 20 seconds, more preferably 30 to 40°C and 6 to 15 seconds.
- When the developer of the present invention is used, developing time is normally 5 to 45 seconds, preferably 8 to 30 seconds, and developing temperature is preferably 25 to 50°C, more preferably 30 to 40°C.
- Drying may be achieved by hot air blow at normally 35 to 100°C, preferably 40 to 80°C. Alternatively, a drying zone equipped with a far infrared heating means may be arranged in the automatic processing machine.
- The automatic processing machine may be equipped with a mechanism for adding eitherwateror an acidic rinsing solution having no fixing capability to the light-sensitive material, among the above developing, fixing and washing processes such described as in Japanese Patent O.P.I. Publication No. 264953/199. The automatic processing machine may also have therein equipment for preparing developers and fixers.
- The developer of the present invention preferably contains as a developing agent a 1,4-dihydroxybenzene compound or, if necessary, a p-aminophenol compound and/or a pyrazolidone compound.
- 1,4-dihydroxybenzenes include hydroquinone, chlorohydroquinone, bromohydroquinone, isopropylhydroquinone, methylhydroquinone, 2,3-dichlorohydroquinone, 2,5-dichlorohydroquinone, 2,3-dibromohydroquinone, 2,5-dimethylhydroquinone and hydroquinone monosulfonate, with preference given to hydroquinone. p-aminophenol-based developing agents include N-methyl-p-aminophenol, p-aminophenol, N-((3-hydroxyethyl)-p-aminophenol, N-(4-hydroxyphenyl)glycine, 2-methyl-p-aminophenol and p-benzylaminophenol, with preference given to N-methyl-p-aminophenol.
- Examples of pyrazolidone compounds which can be used in the present invention include 1-phenyl-3-pyrazolidone, 1-phenyl-4,4-dimethyl-3-pyrazolidone, 1-phenyl-4-ethyl-3-pyrazolidone, 1-phenyl-5-methyl-3-pyrazolidone, 1-phenyl-4-methyl-3-pyrazolidone, 1-phenyl-4-methyl-4-hydroxymethyl-3-pyrazolidone, 1-phenyl-4,4-dihydroxymethyl-3-pyrazolidone, 1,5-diphenyl-3-pyrazolidone, 1-p-tolyl-3-pyrazolidone, 1-phenyl-2-acetyl-4,4-dimethyl-3-pyrazolidone, 1-p-hydroxyphenyl-4,4-dimethyl-3-pyrazolidone, 1-(2-benzothiazolyl)-3-pyrazolidone and 3-acetoxy-1-phenyl-3-pyrazolidone.
- The amount of 1,4-dihydroxybenzene added is 0.01 to 0.7 mol, preferably 0.1 to 0.5 mol per liter of developer.
- The amounts of p-aminophenol compound and pyrazolidone compound added are 0.0005 to 0.2 mol, preferably 0.001 to 0.1 mol per liter of developer.
- Examples of sulfites used in the developer of the present invention include sodium sulfite, potassium sulfite, lithium sulfite, ammonium sulfite, sodium bisulfite and potassium metasulfite. The amount ofthese sulfites used is 0.1 to 2.0 mol, preferably 0.1 to 1.0 mol per liter of developer. Also, the upper limit is preferably 3.0 mol per liter of developer for a concentrated developer.
- The developer may contain a chelating agent having an iron ion chelating stability constant of over 8. The iron ion mentioned herein is ferricion (Fe3+).
- Chelating agents having an iron ion chelating stability constant of over 8 include organic carboxylic acid chelating agents, organic phosphoric acid chelating agents, inorganic phosphoric acid chelating agents and polyhydroxyl compounds.
- Specifically, such chelating agents include ethylenediamine-di-o-hydroxyphenylacetic acid, triethylenete- tramineacetic acid, diaminopropanetetraacetic acid, nitrilotriacetic acid, hydroxyethylethylenediaminetriacetic acid, dihydroxyethylglycine, ethylenediaminediacetic acid, ethylenediaminedipropionic acid, iminodiacetic acid, diethylenetriaminepentaacetic acid, hydroxyethyliminodiacetic acid, 1,3-diamino-2-propanoltetraacetic acid, transcyclohexanediaminetetraacetic acid, ethylenediaminetetraacetic acid, glycol ether amine tetraacetic acid, ethylenediamine-N,N,N',N'-tetrakismethylenephosphonic acid, nitrilo-N,N,N-trimethylenephosphonic acid, 1-hydroxyethyiidene-1,1-diphosphonicacid, 1,1-diphosphonoethane-2-carboxylicacid, 2-phosphonobutane-1,2,4-tricarboxylic acid, 1-hydroxy-1-phosphonopropane-1,2,3-tricarboxylic acid, catechol-3,5-disulfonic acid, sodium pyrophosphate, sodium tetrapolyphosphate and sodium hexametaphosphate.
- The developer of the present invention may contain a hardener which enhances the film physical properties by reacting with the gelatin in the light-sensitive material during the developing process. Examples of hardeners include glutaraldehyde, a-methylglutaraldehyde, (3-methylglutaraldehyde, maleic dialdehyde, succinic dialdehyde, methoxysuccinic dialdehyde, methylsuccinic dialdehyde, α-methoxy-β-ethoxyglutaraldehyde, an-butoxyglutaraldehyde, a,a-dimethoxysuccinic dialdehyde, β-isopropylsuccinic dialdehyde, a,a-diethylsuccinic dialdehyde, butylmaleic dialdehyde and bisulfite adducts thereof.
- In addition to these components, the developer may further contain developing inhibitors such as sodium bromide and potassium iodide, organic solvents such as ethylene glycol, diethylene glycol, triethylene glycol, dimethylformamide, methyl cellosolve, hexylene glycol, ethanol and methanol, mercapto compounds such as 1-phenyl-5-mercaptotetrazole and sodium 2-mercaptobenzimidazole-5-sulfonate, and antifoggants such as 5-methylbenzotriazole and other benzotriazole compounds. Other additives such as toning agents, surfactants, defoaming agents may also be added as necessary.
- The pH of the developer is normally 9.0 to 12, preferably 9.0 to 11.5. Examples of the alkali or buffer used for pH adjustment include pH regulators such as sodium hydroxide, potassium hydroxide, sodium carbonate, potassium carbonate, boric acid, sodium tertiary phosphate and potassium tertiary phosphate.
- A fixer for fixing the light sensitive material may contain a fixing agent such as sodium thiosulfate or ammonium thiosulfate, with preference given to ammonium thiosulfate from the viewpoint of fixing speed. These fixing agents are usually used in amounts of about 0.1 to 6 mol/liter.
- The fixer may also contain a water-soluble aluminum salt as a hardener, such as aluminum chloride, aluminum sulfate or potassium alum.
- The fixer may incorporate malic acid, tartaric acid, citric acid, gluconic acid and derivatives thereof, singly or in combination. These compounds are effective when contained at not less than 0.001 mol per liter of fixer, with a greater effect obtained at 0.005 to 0.03 mol per liter of fixer.
- Fixer pH is normally not lower than 3.8, preferably 4.2 to 7.0. In view of hardening in the fixer, sulfurous acid gas odor the pH is more preferably 4.3 to 4.8.
- Silver halide photographic light-sensitive material emulsions to which the present invention is applicable include medical radiographic materials, photographic materials for printing plate making and direct positive photographic materials.
- The silver halide photographic light-sensitive material to which the present invention is applied may be comprised a silver halide such as silver iodobromide, silver iodochloride or silver iodochlorobromide, with preference given to silver iodobromide from the viewpoint of high sensitivity.
- Emulsions for the silver halide photographic light-sensitive material include monodispersed grains of high inner iodine content such as those disclosed in Japanese Patent O.P.I. Publication Nos. 177535/1984, 802237/1986, 132943/1986 and 49751/1988. Crystalline habit may be cubic, tetradecahedral or octahedral, and intermediate (111) and (100) planes may be present in any ratio.
- The emulsion may be of tabular grains having an aspect ratio of not less than 2. Such tabular grains have advantages such as improvements in spectral sensitizing efficiency, image granularity and sharpness, and are disclosed in British Patent No. 2,112,157, US Patent Nos. 4,439,520,4,433,048,4,414,310 and 4,434,226, and Japanese Patent O.P.I. Publication Nos. 113927/1983, 127921/1983, 138342/1988, 284272/1988 and 305343/1988. The emulsion can be prepared as described in these publications.
- Additives and other substances used in the silver halide photographic light-sensitive material include those described in Research Disclosure Nos. 17643 (December, 1978), 18716 (November, 1979) and 308119 (December, 1989) (hereinafter referred to as RD17643, RD18716 and RD308119, respectively). The following table shows where they are described.
- While maintaining a temperature of 60°C, a pAg of 8 and a pH of 2.0, monodispersed cubic grains of silver iodobromide having an average grain size of 0.3 µm and a silver iodide content of 2 mol% were prepared by the double jet method.
- The resulting reaction mixture was desalinized at 40°C, using an aqueous solution of Demol-N (produced by Kao Atlas) and an aqueous solution of magnesium sulfate, after which it was re-dispersed in an aqueous gelatin solution, to yield a seed emulsion.
- Using the above seed emulsion, grains were grown as follows: First, the seed emulsion was dispersed in an aqueous gelatin solution being kept at 40°C, and aqueous ammonia and acetic acid were added to obtain a pH of 9.7. An aqueous solution of ammoniacal silver nitrate and an aqueous solution of potassium bromide and potassium iodide were then added to the dispersion by the double jet method, while maintaining a pAg of 7.3 and a pH of 9.7, to yield a layer containing 35 mol% silver iodide. Next, another aqueous solution of ammoniacal silver nitrate and an aqueous solution of potassium bromide were added by the double jet method.
- Until 95% of the desired grain size was reached, the pAg was kept at 9.0, with the pH varied continuously over the range of 9.0 to 8.0. The pAg was then changed to 11.0, and while keeping the pH at 8.0, grains were grown up to the desired grain size. Subsequently, acetic acid was added to obtain a pH of 6.0, after which 400 mg of the anhydride of 5,5'-dichloro-9-ethyl-3,3'-di(3-sulfopropyl)oxacarbocyanine sodium salt was added per mol of silver halide. This mixture was then desalinized with the above aqueous solution of Demol-N and aqueous solution of magnesium sulfate, and then re-dispersed in a gelatin solution.
- Monodispersed silver iodobromide emulsions A, B and C, comprising tetradecahedral grains with round tips having an average silver iodide content of 2.0 mol%, were thus prepared, which had average grain sizes of 0.40, 0.65 and 1.00 µm and variation coefficients of grain size distribution (a/
r ) of 0.17, 0.16 and 0.16, respectively, in which σ is the standard deviation of grain size distribution andr is the average grain size. -
- After stopping the addition of solutions Band C, the temperature of solution A was increased to 60°C over a period of 60 minutes, and solutions B and C were again added by the double jet method at a flow rate of 68.5 ml/min over a period of 50 minutes, while keeping the silver potential (determined using a silver ion selective electrode in combination with a saturated silver-silver chloride electrode as a reference electrode) at +6 mV using solution D.
- After completion of the addition, 3% KOH solution was added to obtain a pH of 6, followed by immediate desalinization and washing. The resulting emulsion was designated as seed emulsion Em 0. Electron microscopy revealed that this emulsion comprised hexagonal tabular silver halide grains not less than 90% by projected area of which had a ratio of the largest edge length to the shortest edge length of 1.0 to 2.0 and which tabular grains had an average thickness of 0.07 µm and an average diameter of 0.5 µm as of circle diameter.
-
- Throughout this operation, the silver potential was kept at +25 mV using solution D. After completion of the addition, the following spectral sensitizing dyes A and B were added in amounts of 300 mg and 15 mg, respectively, per mol of silver halide.
- After the excess salts were removed by precipitation and desalinization with the same aqueous solutions of Demol-N and of magnesium sulfate as above, an aqueous solution of 92.2 g of ossein gelatin was added, and the mixture was stirred and re-dispersed.
- A tabular silver iodobromide emulsion D having an average silver iodide content of 1.5 mol%, a projected area diameter of 0.96 µm, a coefficient of variation of 0.25 and an aspect ratio of 4.0 was thus prepared.
- Sensitizing dye A: Anhydride of 5,5'-dichloro-9-ethyl-3,3'di-(3-sulfopropyl)oxacarboxycyanine sodium salt
- Sensitizing dye B: Anhydride of 5,5'-di-(butoxycarbonyl)1,1'-diethyl-3,3'-di-(4-sulfobutyl )benzimidazolocarbocyanine sodium salt
- To each of the thus-obtained emulsions A, B, C and D, a 200:1 (w/w) mixture of the above sensitizing dyes A and B was added at 975 mg, 600 mg, 390 mg and 500 mg per mol of silver halide, respectively.
- Ten minutes later, appropriate amounts of chloroauric acid, sodium thiosulfate and ammonium thiocyanate were added for optimum chemical sensitization. Fifteen minutes before completion of ripening, 200 mg of potassium iodide was added per mol of silver halide. Subsequently, 3 x 10-2 mol of 4-hydroxy-6-methyl-1,3,3a,7-tetrazaindene was added per mol of silver halide, and this mixture was dispersed in an aqueous solution of 70 g of gelatin.
- Of the four ripened emulsions, A, B and C were mixed in a weight ratio of 15:65:20, to yield emulsion I, while emulsion D, designated as emulsion II, was used as such.
-
- Ten Kilograms of the following dye was dissolved in a mixed solvent of 28 I of tricresyl phosphate and 85 I of ethyl acetate at 55°C. This solution is designated as the oily solution.
- Separately, 270 I of a 9.3% aqueous solution of gelatin containing 1.35 kg of an anionic surfactant AS was prepared. This solution is designated as the aqueous solution. Next, the oily solution and the aqueous solution were placed in a dispersing vessel and dispersed while keeping the liquid temperature at 40°C. To the resulting dispersion were added appropriate amounts of phenol and 1,1-dimethylol-bromo-1-nitromethane, and water was added to 240 kg.
-
- The resulting coating solution was coated and dried uniformly on a blue-colored subbed polyethylene terephthalate film base 180 µm thick.
- Using two slide hopper coaters, both the emulsion layer and the protective layer were coated simultaneously at a coating speed of 90 m per minute, to yield samples, wherein coating rates were 1.7 g/m2 as of silver for the emulsion layer and 0.99 g/m2 as of gelatin for the protective layer.
- In Table 1, developing sample 1 was obtained from emulsion I, and developing sample 2 obtained from emulsion II.
-
- To prepare the developer, parts A and B were added at the same time to about 5 liters of water, and while stirring and dissolving the mixture, water was added to 12 liters, and glacial acetic acid was added to obtain a pH of 10.40, to yield a developer replenisher.
- To this developer replenisher, the above starter was added at 20 ml/I, followed by pH adjustment to 10.26 before use.
- To prepare the fixer, parts A and B were added at the same time to about 5 liters of water, and while stirring and dissolving the mixture, water was added to 18 liters, and sulfuric acid or NaOH were added to obtain a pH of 4.4, to yield a fixer replenisher.
- Based on the above developer compounds of the present invention and comparative compounds were added as listed in Table 1 below to prepare developers, which were used for actual developing.
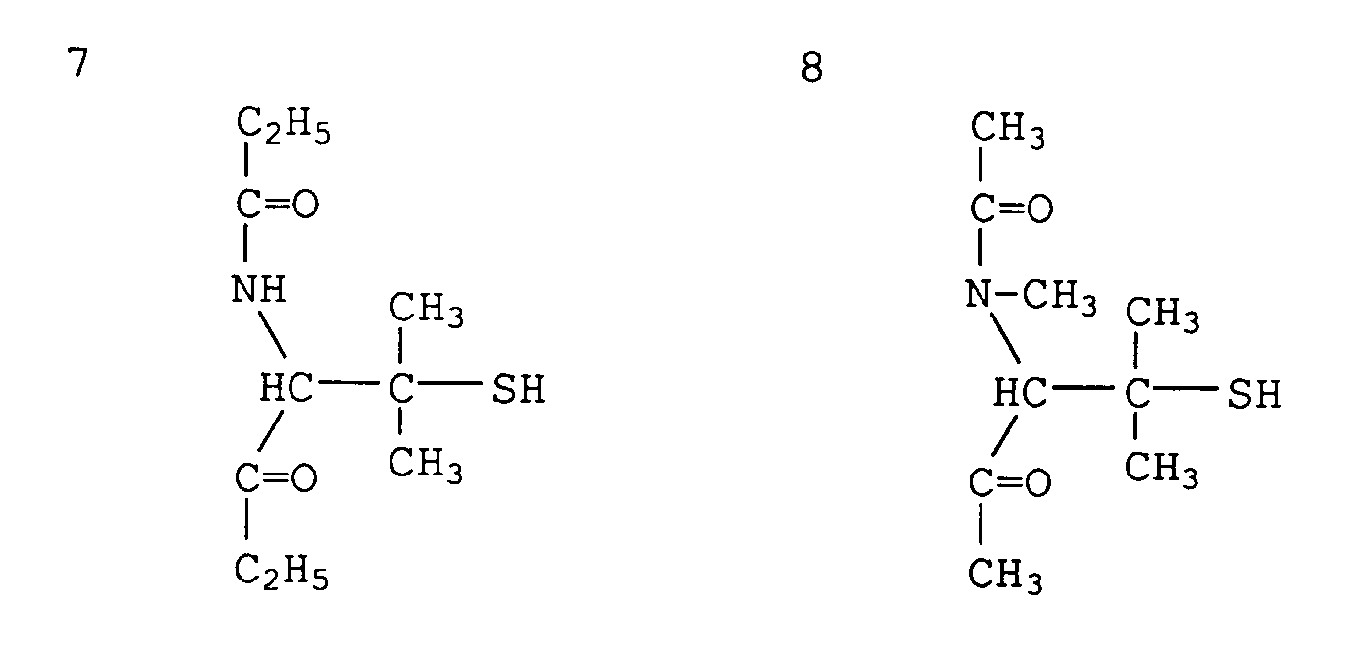
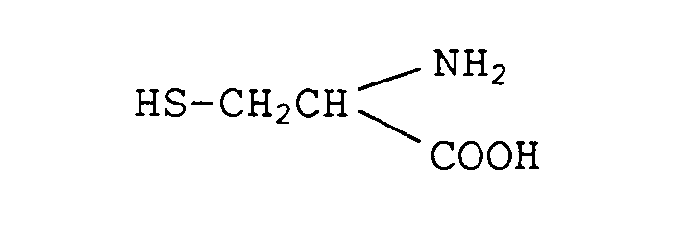
- The following Comparative Compounds 1 - 12 were used as comparative compounds.
- Comparative Compound 1: L-cysteine (free base)
- Comparative Compound 2: Cystine
- Comparative Compound 3: L-methionine
- Comparative Compound 4: DL-penicillamine
- Comparative Compound 5: Acetylmethionine
- Comparative Compound 6: DL-ethionine
- Comparative Compound 7: Mercaptoisobutyric acid
- Comparative Compound 8: β-phenyl-α-mercaptoacrylic acid
- Comparative Compound 9: y-isothioureidobutyric acid
- Comparative Compound 10: N-acetyl-L-cysteine
- Comparative Compound 11: Homocysteine
- Comparative Compound 12: Isoleucine
-
- Processing temperatures were 35°C for developing, 33°C for fixation, 20°C for washing and 50°C for drying.
- The samples were evaluated as follows:
- The sample, inserted between two sheets of fluorescent sensitizing paper KO-250, manufactured by Konica Corp., was subjected to exposure through an aluminum wedge of at a tube voltage of 80 kV, a tube amperage of 100 mAand an irradiation time of 50 msec, after which it was processed using the above automatic processing machines. With respect to the processed sample, the reciprocal of the exposure amount required to obtain a density of base density + fog density + 1.0 was calculated as a percent sensitivity relative to the sensitivity of sample No. 1 processed with the above developer and fixer of basic composition using automatic processing machine No. 2 (experiment No. 15).
- The density in the unexposed portion of the exposed sample was determined using Konica PDA-65 densitometer, and the base density was subtracted therefrom to obtain the fog density, and the maximum density was expressed as Dm.
- Next, developing samples, subjected to X-ray exposure to a density of 1.0 over the entire surface of the sample, were processed at a fixer replenishing rate of 300 cc/m2 at 70 sheets per day for 30 consecutive days, using the above-described automatic processing machine Nos. 1, and 3, wherein the developer replenishing rate and developing sample were changed as shown in Table 1.
- The developing samples used were the above sample Nos. 1 and 2.
- Upon processing, visual evaluation was made for possible stains on the developing rack, rollers and wall, and of the processed sample, in the following four grades:
- A: Almost no silver sludge seen, with no stain on the rollers or wall.
- B: Developer turbidity seen, with slight stain on the processing tank wall.
- C: Silver sludge seen, with developing rack stain difficult to wash down.
- D: Much silver sludge seen on the developer tank, causing image stain as a result of its adhesion to the film being processed.
- The above sample No. 2 was processed unexposed and evaluated for residual silver as follows:
- One drop of a 2.6 x 10-3 mol/I aqueous solution of sodium sulfide, as the residual silver evaluating solution, was dropped on a surface of the above residual silver evaluating film. Three minutes later, the solution was thoroughly wiped away, and the film was kept standing at normal temperature and normal humidity for 15 hours.
- Then, using a PDA-65 densitometer (produced by Konica Corporation), blue light transmission densities were determined for the portion where the residual silver evaluating solution was dropped and the other portion. The difference of the densities was used as the index of residual silver. The residual silver concentration in the processed film increases as this difference increases.
-
- As seen in these tables, according to the present invention, silver sludge is well prevented without photographic performance deterioration even at reduced developer replenishing rates.
- Specifically, Table 1 shows that the addition of a compound of the present invention to the developer did not lower the sensitivity or Dm (maximum density), while the addition of a comparative compound significantly lowered the sensitivity, though it had a slight preventive effect on silver sludge at high replenishing rates.
- Results of experiment Nos. 1 through 10 demonstrate that developers containing a compound of the present invention remain excellently effective without deterioration of the silver sludge preventing effect even when the replenishing rate is reduced to 200 ml/m2. Also, the results of experiment Nos. 12 and 14 demonstrate that there is no difference in silver sludge preventing effect among different automatic processing machines.
- An unexpected finding is that the present invention offers improved fixing performance with no residual silver.
Claims (10)
wherein R1 and R2 are each a hydrogen atom or an alkyl group having 1 to 3 carbon atoms provided that both of R1 and R2 are not hydrogen atoms at the same time; R3 and R4 are each a hydrogen atom or an alkyl group having 1 to 3 carbon atoms; R5 is a hydroxy group, an amino group or an alkyl group having 1 to 3 carbon atoms; R6 and R7 are each a hydrogen atom, an alkyl group having 1 to 5 carbon atoms, an acyl group having 1 to 18 carbon atoms or a -COOM2 group, provided that both of R6 and R7 are not hydrogen atoms at the same time, in the above M1 is a hydrogen atom, an alkali metal atom or an ammonium group; m is an integer 0, 1 or 2; M2 is a hydrogen atom, an alkyl group having 1 to 4 carbon atoms, an alkali metal atom, an aryl group or an aralkyl group having not more than 15 carbon atoms.
wherein R1 and R2 are each a hydrogen atom or an alkyl group having 1 to 3 carbon atoms provided that both of R1 and R2 are not hydrogen atoms at the same time; R3 and R4 are each a hydrogen atom or an alkyl group having 1 to 3 carbon atoms; R5 is a hydroxy group, an amino group or an alkyl group having 1 to 3 carbon atoms; R6 and R7 are each a hydrogen atom, an alkyl group having 1 to 5 carbon atoms, an acyl group having 1 to 18 carbon atoms or a -COOM2 group, provided that both of R6 and R7 are not hydrogen atoms at the same time, in the above M1 is a hydrogen atom, an alkali metal atom or an ammonium group; m is an integer 0, 1 or 2; M2 is a hydrogen atom, an alkyl group having 1 to 4 carbon atoms, an alkali metal atom, an aryl group or an aralkyl group having not more than 15 carbon atoms.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP92947/92 | 1992-04-13 | ||
| JP4092947A JP2955906B2 (en) | 1992-04-13 | 1992-04-13 | Developer for silver halide photographic materials |
Publications (3)
| Publication Number | Publication Date |
|---|---|
| EP0566323A2 true EP0566323A2 (en) | 1993-10-20 |
| EP0566323A3 EP0566323A3 (en) | 1994-03-23 |
| EP0566323B1 EP0566323B1 (en) | 1995-10-11 |
Family
ID=14068669
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP19930302781 Expired - Lifetime EP0566323B1 (en) | 1992-04-13 | 1993-04-08 | Developer for silver halide photographic light-sensitive material |
Country Status (3)
| Country | Link |
|---|---|
| EP (1) | EP0566323B1 (en) |
| JP (1) | JP2955906B2 (en) |
| DE (1) | DE69300613T2 (en) |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0664479A1 (en) * | 1993-12-06 | 1995-07-26 | Konica Corporation | Method of processing silver halide photographic lightsensitive material |
| US5510231A (en) * | 1993-04-27 | 1996-04-23 | Konica Corporation | Solid developing composition for silver halide photographic light-sensitive material and processing method using the same |
| US5824458A (en) * | 1994-02-28 | 1998-10-20 | Fuji Photo Film Co., Ltd. | Developer and fixing solution for silver halide photographic material and processing method using the same |
| EP1231504A2 (en) * | 2001-02-13 | 2002-08-14 | Eastman Kodak Company | Photographic developing composition and use thereof in the development of a photographic element |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE69516054T2 (en) | 1994-07-18 | 2000-10-26 | Konishiroku Photo Ind | Silver halide photographic element and its processing method |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4141734A (en) * | 1975-09-11 | 1979-02-27 | Ciba-Geiby Ag | Photographic developing process |
| US4254215A (en) * | 1978-03-31 | 1981-03-03 | Ciba-Geigy Ag | Process for the prevention of darkening and the formation of a sediment in photographic developer solutions |
-
1992
- 1992-04-13 JP JP4092947A patent/JP2955906B2/en not_active Expired - Fee Related
-
1993
- 1993-04-08 EP EP19930302781 patent/EP0566323B1/en not_active Expired - Lifetime
- 1993-04-08 DE DE1993600613 patent/DE69300613T2/en not_active Expired - Fee Related
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4141734A (en) * | 1975-09-11 | 1979-02-27 | Ciba-Geiby Ag | Photographic developing process |
| US4254215A (en) * | 1978-03-31 | 1981-03-03 | Ciba-Geigy Ag | Process for the prevention of darkening and the formation of a sediment in photographic developer solutions |
Cited By (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5510231A (en) * | 1993-04-27 | 1996-04-23 | Konica Corporation | Solid developing composition for silver halide photographic light-sensitive material and processing method using the same |
| EP0664479A1 (en) * | 1993-12-06 | 1995-07-26 | Konica Corporation | Method of processing silver halide photographic lightsensitive material |
| US5824458A (en) * | 1994-02-28 | 1998-10-20 | Fuji Photo Film Co., Ltd. | Developer and fixing solution for silver halide photographic material and processing method using the same |
| EP1231504A2 (en) * | 2001-02-13 | 2002-08-14 | Eastman Kodak Company | Photographic developing composition and use thereof in the development of a photographic element |
| EP1231504A3 (en) * | 2001-02-13 | 2003-05-21 | Eastman Kodak Company | Photographic developing composition and use thereof in the development of a photographic element |
| US6764814B2 (en) | 2001-02-13 | 2004-07-20 | Eastman Kodak Company | Photographic developing composition and use thereof in the development of a photographic element |
| US6927021B2 (en) | 2001-02-13 | 2005-08-09 | Eastman Kodak Company | Photographic developing composition and use thereof in the development of a photographic element |
Also Published As
| Publication number | Publication date |
|---|---|
| EP0566323B1 (en) | 1995-10-11 |
| JP2955906B2 (en) | 1999-10-04 |
| JPH05289255A (en) | 1993-11-05 |
| EP0566323A3 (en) | 1994-03-23 |
| DE69300613T2 (en) | 1996-04-04 |
| DE69300613D1 (en) | 1995-11-16 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US5474879A (en) | Radiographic film developers containing ascorbic acid and thioether development accelerators | |
| US4826757A (en) | Process for processing silver halide photographic materials | |
| US4777118A (en) | Process for the formation of high contrast negative images and silver halide photographic element | |
| US5364746A (en) | Developer for silver halide photographic light-sensitive material | |
| EP0566323B1 (en) | Developer for silver halide photographic light-sensitive material | |
| US5545508A (en) | Photographic silver halide developer compositions and process for forming photographic silver images | |
| US5508152A (en) | Method for processing a silver halide photographic material | |
| US5298372A (en) | Method for processing black-and-white silver halide photographic material | |
| JP3243676B2 (en) | Concentrate of fixer for silver halide photographic materials | |
| EP0430212A1 (en) | Method for development processing of silver halide photosensitive materials | |
| JP2890076B2 (en) | Developing method of silver halide photographic material | |
| JP3500200B2 (en) | Processing of silver halide photographic materials | |
| US5368983A (en) | Method for forming an image | |
| JP2840625B2 (en) | Processing method of black and white silver halide photographic material | |
| JPH06138591A (en) | Development processing method for silver halide photographic sensitive material | |
| JPH06138616A (en) | Method for processing silver halide photographic sensitive material | |
| JPH0193737A (en) | Developing method | |
| JPH06230526A (en) | Processing method of silver halide photographic sensitive material | |
| JPH07301874A (en) | Silver halide photographic sensitive material and its processing method | |
| EP0622670A1 (en) | Photographic silver halide developer compositions and process for forming photographic silver images | |
| JPH08286332A (en) | Processing method for silver halide photosensitive material | |
| JPH08201992A (en) | Method for processing silver halide photographic sensitive material | |
| JPH07209823A (en) | Method for processing silver halide photographic sensitive material | |
| JP2002072425A (en) | Ascorbic acid developing composition and method for using the same | |
| JPH03255437A (en) | Processing method for silver halide photographic sensitive material |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AK | Designated contracting states |
Kind code of ref document: A2 Designated state(s): DE FR GB IT |
|
| PUAL | Search report despatched |
Free format text: ORIGINAL CODE: 0009013 |
|
| AK | Designated contracting states |
Kind code of ref document: A3 Designated state(s): DE FR GB IT |
|
| 17P | Request for examination filed |
Effective date: 19940819 |
|
| 17Q | First examination report despatched |
Effective date: 19941012 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): DE FR GB IT |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IT Free format text: LAPSE BECAUSE OF FAILURE TO SUBMIT A TRANSLATION OF THE DESCRIPTION OR TO PAY THE FEE WITHIN THE PRE;WARNING: LAPSES OF ITALIAN PATENTS WITH EFFECTIVE DATE BEFORE 2007 MAY HAVE OCCURRED AT ANY TIME BEFORE 2007. THE CORRECT EFFECTIVE DATE MAY BE DIFFERENT FROM THE ONE RECORDED.SCRIBED TIME-LIMIT Effective date: 19951011 Ref country code: FR Effective date: 19951011 |
|
| REF | Corresponds to: |
Ref document number: 69300613 Country of ref document: DE Date of ref document: 19951116 |
|
| EN | Fr: translation not filed | ||
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| 26N | No opposition filed | ||
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 19980330 Year of fee payment: 6 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GB Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19990408 |
|
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 19990408 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: DE Payment date: 20060406 Year of fee payment: 14 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DE Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20071101 |