EP0551999A1 - Mass spectrometry systems - Google Patents
Mass spectrometry systems Download PDFInfo
- Publication number
- EP0551999A1 EP0551999A1 EP93300131A EP93300131A EP0551999A1 EP 0551999 A1 EP0551999 A1 EP 0551999A1 EP 93300131 A EP93300131 A EP 93300131A EP 93300131 A EP93300131 A EP 93300131A EP 0551999 A1 EP0551999 A1 EP 0551999A1
- Authority
- EP
- European Patent Office
- Prior art keywords
- electrode
- ions
- mass
- ion
- mass spectrometry
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Withdrawn
Links
Images
Classifications
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01J—ELECTRIC DISCHARGE TUBES OR DISCHARGE LAMPS
- H01J49/00—Particle spectrometers or separator tubes
- H01J49/004—Combinations of spectrometers, tandem spectrometers, e.g. MS/MS, MSn
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01J—ELECTRIC DISCHARGE TUBES OR DISCHARGE LAMPS
- H01J49/00—Particle spectrometers or separator tubes
- H01J49/26—Mass spectrometers or separator tubes
- H01J49/28—Static spectrometers
- H01J49/30—Static spectrometers using magnetic analysers, e.g. Dempster spectrometer
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01J—ELECTRIC DISCHARGE TUBES OR DISCHARGE LAMPS
- H01J49/00—Particle spectrometers or separator tubes
- H01J49/26—Mass spectrometers or separator tubes
- H01J49/34—Dynamic spectrometers
- H01J49/40—Time-of-flight spectrometers
- H01J49/405—Time-of-flight spectrometers characterised by the reflectron, e.g. curved field, electrode shapes
Definitions
- This invention relates to mass spectrometry systems.
- tandem mass spectrometers have been employed, an example being a four-sector mass spectrometer used in combination with an array detector.
- tandem mass spectrometers have been employed, an example being a four-sector mass spectrometer used in combination with an array detector.
- the instrument needs to be repeatedly re-tuned in order to detect the entire daughter ion mass spectrum, and this is both inconvenient and time consuming.
- such arrangements are complex and expensive, and large magnets are needed to detect ions of higher mass.
- a mass spectrometry system comprising, an ion source, a magnetic sector mass analyser for selecting source ions having a predetermined mass-to-charge ratio, pulse-forming means for forming source ions into a pulse of ions, means for dissociating ions in the pulse whereby to generate daughter ions from source ions selected by the magnetic sector mass analyser, and a time-of-flight mass analyser for analysing the daughter ions as a function of their mass-to-charge ratios, wherein the time-of-flight mass analyser comprises an ion mirror for subjecting ions to an electrostatic reflecting field in the form of an electrostatic quadrupole field.
- the ion source may be a pulsed ion source, and the pulseforming means may comprise an ion buncher for compressing a pulse of the source ions, into an ion pulse of shorter duration.
- the ion mirror and the ion buncher in combination, enable a much improved mass resolving power to be achieved compared with that attained by known tandem mass spectrometry systems. Furthermore, use of the ion mirror enables the whole of the mass spectrum (including contributions from both the daughter ions and undissociated parent ions) to be detected without the need for any re-tuning, and the mass spectrum can be easily calibrated against mass with absolute precision.
- the flight times of the ions through the ion mirror to an associated detector depend on their mass-to-charge ratios, and are entirely independent of their energies, and so a high degree of mass resolution can be attained, even though the ions are subject to a substantial spreading of their energies, due to the effect of the ion buncher on the parent ions and to the fact that the ions entering the ion mirror will have a range of different masses.
- the ion mirror may comprise a monopole electrode structure operating at a d.c. voltage, and may comprise a first electrode having an electrode surface of substantially V-shaped transverse cross-section and a second electrode having an electrode surface of curvilinear transverse cross-section facing the electrode surface of the first electrode, wherein the second electrode is maintained, in operation, at a d.c. retarding voltage with respect to the first electrode, and the first electrode is adapted to allow ions to enter and exit the electrostatic reflecting field between the electrode surfaces of the first and second electrodes.
- the monopole electrode structure may comprise an electrically conductive member having a substantially V-shaped transverse cross-section and an electrically resistive member having a substantially V-shaped transverse cross-section, wherein the apex of the electrically resistive member is maintained, in operation, at a d.c. retarding voltage with respect to the electrically conductive member and the electrically conductive member is adapted to allow ions to enter and exit the electrostatic reflecting field bounded by the electrically conductive and the electrically resistive members.
- the monopole electrode structure may comprise an electrically conductive member having a substantially V-shaped transverse cross-section, electrode means which faces the electrically conductive member and is maintained in operation at a d.c. retarding voltage with respect to the electrically conductive member and electrically insulating side walls, wherein the electrically insulating side walls bear a plurality of electrodes along respective lines of intersection with selected equipotentials in the electrostatic quadrupole field and each said electrode is maintained at a respective voltage.
- the monopole electrode structures may have rotational symmetry about a longitudinal axis intersecting the or each V-shaped electrode.
- the electrostatic reflecting field may reflect each ion about a plane and may include means for controlling the angle of incidence of the ion relative to that plane whereby to control the spatial separation of mass-resolved ions exiting the electrostatic reflecting field.
- the ion buncher may comprise electrostatic means defining a buncher space having an entrance, by which a pulse of ions selected by the magnetic sector mass analyser enters the buncher space, and an exit by which the pulse exits the buncher space, the electrostatic means being operable to apply an electrostatic accelerating (or decelerating) force to ions in a pulse that has entered the buncher space whereby to accelerate (or decelerate) the ions to higher (or lower) energies in proportion to their separation from the exit of the buncher space.
- a mass spectrometry system comprising the serial arrangement of an ion source, a magnetic sector mass analyser and a time-of-flight mass analyser, wherein the time-of-flight mass analyser includes an ion mirror and a detector, and has the property that the flight times of ions through the ion mirror to the detector are related to the mass-to-charge ratios of the ions, and is independent of their energies.
- the mass spectrometry system to be described is used to analyse the mass spectrum of daughter ions derived, by dissociation, from parent ions having a selected mass-to-charge ratio.
- the mass spectrometry system involves the use of a magnetic sector (MS) mass analyser and a time-of-flight (TOF) mass analyser arranged in tandem.
- MS magnetic sector
- TOF time-of-flight
- the mass spectrometry system comprises the serial arrangement of a pulsed ion source 10, a MS mass analyser 20, an ion lens L and a first set of deflector plates D1, an ion buncher 30, a collision cell 40, a second set of deflector plates D2 and finally a TOF mass analyser 50.
- Parent ions produced by the ion source 10 are admitted to the MS mass analyser 20 in short pulses, typically of 300-500 nsec duration.
- a sample under investigation could be ionised using either a laser beam or an ion beam, both of which can be generated in a pulsed mode.
- the pulses forming the ionising beam may be of relatively short duration, the resulting ions being extracted from the source, for admittance to the MS mass analyser 20, using a static extraction field.
- longer ionising pulses could be used, in which case the extraction field would need to be pulsed.
- a pulsed extraction field can be problematical since it tends to give rise to a spreading of ion energies causing an undesirable spreading of flight times through the instrument.
- the parent ions produced by the ion source will have a range of different mass-to-charge ratios, and the MS mass analyser is tuned to separate out, for further analysis, only those parent ions having a single, preselected mass-to-charge ratio.
- the MS mass analyser 20 is of the well known, double-focussing sector kind which combines magnetic and electric sectors.
- Figure 1 shows a 'BE' double-focussing sector mass analyser; however, an 'EB' double-focussing sector mass analyser could alternatively be used.
- This form of MS mass analyser is particularly advantageous in the context of the present mass spectrometry system in that it subjects the parent ion pulses to relatively little time broadening as they travel through the analyser, and this assists in the attainment of a high mass resolving power.
- other known forms of MS mass analyser could alternatively be used.
- the beam of mass-selected parent ions converges on the resolving slit of the MS mass analyser 20 and diverges as the beam travels beyond the slit.
- a typical divergence angle is of the order of ⁇ 20 mradians (i.e. ⁇ 1°) and for a MS mass analyser having a mass resolving power of 3000, say, the width of the ion beam at the resolving slit would be about 50 ⁇ m.
- the TOF mass analyser 50 In order to improve the overall sensitivity of the instrument, it is important that the TOF mass analyser 50 should collect as large a proportion as possible of the parent ions (and their daughers) exiting the MS mass analyser and, to that end, the ion lens L is designed to refocus the diverging ion beam inside the TOF mass analyser 50.
- the lens has a magnification factor of about 10, giving a beam width of about 0.5 mm, and a convergence angle of about ⁇ 2m radians, at the entrance to the TOF mass analyser.
- the sensitivity of the TOF mass analyser 50 depends on the angles of incidence at which ions having different mass-to-charge ratios enter the flight path of analyser 50.
- the two sets of electrostatic deflector plates D1 and D2 are used to control these incidence angles with the aim of optimising sensitivity.
- the mass resolving power R can be improved if the time width ⁇ T of the ion pulses is as short as possible.
- ions entering the flight path of the TOF mass analyser 50 may typically have energies of the order of 8 keV and if the flight path is 1 m, a resolution of 5000 at mass 5000 can only be achieved if the pulse width ⁇ T is of the order of 14nsec or less.
- this pulse width is much smaller than that provided by the pulsed ion source 10 at the exit of the MS mass analyser - typically 300-500 nsec.
- An ion buncher 30 is therefore provided between the ion lens L and the TOF mass analyser 50 in order to compress the ion pulses produced by source 10 into pulses of much shorter duration.
- the ion buncher 30 comprises a pair of parallel electrode plates P1,P2 which are normally maintained at ground potential.
- the upstream plate P1 that is to say the plate nearer the MS mass analyser 20
- V B for positive ions
- This voltage subjects each ion in the pulse to an electrostatic accelerating force in the direction of the TOF mass analyser 50, and accelerates the ion to a higher energy by an amount proportional to its separation from the grounded, downstream electrode plate P2.
- the downstream ions in the pulse which entered the buncher space first and are closer to plate P2, spend less time in the accelerating field than do the upstream ions which entered the buncher space later. Accordingly, the upstream ions tend to catch up with the downstream ions.
- the distance s separating the two electrode plates, the distance d separating the downstream plate and the entrance to the TOF mass analyser 50, and the voltage V B applied to electrode plate P1 are chosen so that (in theory) the ions in a pulse all arrive at the entrance to the TOF mass analyser at substantially the same time.
- bunching could be achieved using a retarding field which is de-energised once the pulse lies wholly within the buncher space S B between the electrode plates.
- the upstream ions are retarded less than the downstream ions, and as before tend to catch up the downstream ions.
- a consequence of subjecting the mass-selected parent ions in each pulse to the accelerating voltage V B is to introduce a significant spreading of their energies. If, for example, the ion pulses produced by ion source 10 are spread out in space over 50 mm so that they just span the electrode plates P1,P2 of the ion buncher, the energies of parent ions arriving at the TOF mass analyser 50 would range from 8keV (the energy of the leading ion in the pulse which receives no energy from the accelerating field) and 12keV (the energy of the trailing ion in the pulse which receives the maximum energy of 4 keV from the accelerating field).
- the compressed ion pulse (which may typically have a time width of the order of 10nsec) passes through the collision cell 40 positioned at the entrance to the TOF mass analyser 50.
- a laser pulse to dissociate the mass-selected parent ions forming the compressed pulse, and since the compressed ion pulse is well defined in both time and space, the laser pulse can be synchronised to coincide with the arrival of each ion pulse at the time focal point.
- the daughter ions produced by dissociation of the mass-selected parent ions, continue on the same trajectory as the parent ions, with very nearly the same velocity, and so there is little effect on the position of the time focal point.
- undissociated parent ions and daughter ions introduced into the TOF mass analyser 50 will have a substantial energy spread. This is due to the effect of the ion buncher, as described hereinbefore, and to the fact that the parent ions and the daughter ions will have a range of different masses (each daughter ion of mass M D , say, will have a fraction M D /M P of the energy of the parent ion, of mass M P , from which it is derived).
- the TOF mass analyser 50 enables a high mass resolving power to be attained even though the ions introduced into the flight path of the analyser have different energies.
- the TOF mass analyser 50 comprises a special form of ion mirror, described in our copending European patent application, Publication No. 408,288A1.
- This form of ion mirror has the property that the flight time of an ion through the ion mirror depends on its mass-to-charge ratio, but is entirely independent of its energy.
- Figure 3 illustrates diagrammatically how the ion mirror affects the motion of an ion I as it moves in the X-Z plane along a path T inclined at an angle of incidence ⁇ to the longitudinal axis X - as mentioned hereinbefore, the angle of incidence ⁇ of the ion can be controlled by the deflector plates D1,D2.
- the ion mirror establishes an electrostatic field region E bounded by the broken lines F1,F2 and that the ion I of mass-to-charge ratio (m/q), say, moving on path T enters the field region at a point 1, undergoes a reflection at a point 2, returns on path T' and finally exits the field region at a point 3.
- paths T,T' lie in the X-Z plane and the ion I is reflected about the X-Y plane, normal to the page.
- the ion is subjected to an electrostatic reflecting force F which increases linearly as a function of the depth of penetration of the ion into the field region.
- This force acts in the direction of arrow A in Figure 3 and has a magnitude directly proportional to the separation x of the ion from the line joining the exit and entry points 1,3.
- the equation of motion of the ion in the field region is akin to that associated with damped simple harmonic motion, and it can be shown that the time interval t during which the ion travels from the point of entry 1 to the point of reflection 2 is given by the expression
- the ion occupies the field region for a total time interval t' given by
- the ion occupies the field region E for a time interval which depends only on its mass-to-charge ratio (m/q), and this enables ions to be distinguished from one another as a function of their mass-to-charge ratios, even if, as in the present case, they have different energies.
- Figure 4 shows, by way of example, the flight paths followed by undissociated parent ions I P and by two daughter ions I D (1),I D (2) having masses M D (1), M D (2) respectively, wherein M D (1) M D (2) - it will be assumed, in this example, that the ions all have the same charge.
- the undissociated parent ions I P being the heaviest, have the longest flight time through the field region and they move along the outermost path, whereas the lighter daughter ions I D (2) have the shortest flight time and because they have lower energy they follow the innermost path.
- the ions are detected separately, using a multichannel plate detector, for example, whereby to produce a mass spectrum of all the ions and since, in general, the undissociated parent ions will be much more energetic than the daughter ions the spatial spread of ions exiting the field region could be considerable.
- the two sets of deflector plates D1,D2 are used to control the angle of incidence ⁇ of the ions entering the TOF mass analyser, and it is the particular function of the second set of deflector plates D2 to reduce the spatial spread of ions at the detector, enabling all ions to be detected.
- the deflector plates D2 subject all the ions to an electrostatic deflecting force (in the downwards Z-direction in Figure 3) just before they enter the field region of the ion mirror.
- the relatively light daughter ions have lower energies than the heavier, undissociated parent ions and so suffer a comparatively large deflection, increasing their angles of incidence relative to that of the parent ions and so reducing the spatial spread of the ions received at the detector. In principle, therefore, it is possible to collect all the undissociated parent ions and the daughter ions that constitute the entire mass spectrum.
- An electrostatic field of this form has four-fold symmetry about the Z-axis and could be generated by a quadrupole electrode structure (which provides field in all four quadrants) or a monopole electrode structure (which provides field on only one of the quadrants).
- Quadrupole and monopole electrode structures are, of course, known in mass analysis spectrometry; however, in contrast to the present arrangement, such known electrode structures operate at radio frequencies.
- a quadrupole electrode arrangement is shown in Figure 5 and this drawing also shows the hyperbolic equipotential lines H in the electrostatic quadrupole field.
- Figures 6a and 6b show a monopole electrode structure.
- the monopole electrode structure 50 shown in Figures 6a and 6b, comprises two elongate electrodes 51,52 which extend parallel to the longitudinal Z-axis of the electrode structure, and are spaced apart from each other along the transverse X-axis.
- the two electrodes have inwardly facing electrode surfaces which are disposed symmetrically with respect to the X-Z plane and define an intermediate field region E.
- Electrode 51 has a substantially V-shaped transverse cross-section (subtending an angle of 90°) whereas electrode 52 is in the form of a rod and has a hyperbolic or, alternatively, a circular transverse cross-section.
- electrode 51 has an elongate window 53 by which the ions may enter the field region for reflection in the X-Z plane, one of the electrodes being maintained at a fixed d.c. voltage with respect to the other electrode. If, for example, electrode 52 is maintained at a positive d.c. voltage with respect to electrode 51, the electrostatic field created in the field region would be such as to reflect positively-charged ions. Conversely, if electrode 52 is maintained at a negative d.c. voltage with respect to electrode 51, the electrostatic field would be such as to reflect negatively-charged ions.
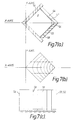
- FIG. 7a shows a transverse cross-sectional view through an alternative monopole electrode structure.
- This electrode structure has a pair of orthogonally inclined side walls 54,55 made from an electrically insulating material, such as glass.
- the side walls abut the electrode 51, as shown, to form a boundary structure enclosing a field region E of square cross-section.
- An electrode 56 positioned at the apex of the side walls, is maintained at an appropriate d.c. retarding voltage with respect to the electrode 51, and the side walls bear respective coatings 57,58 of an electrically resistive material inter-connecting the electrode 56 and the electrode 51.
- the structure may also have coated end walls (not shown) which serve to terminate electrostatic field lines extending in the Z-axis direction and so, in effect, simulate a structure having infinite length in that direction.
- the quadrupole electrostatic field created by this electrode structure has hyperbolic equipotential lines in the transverse (X-Y) plane, as defined by equation 5 above. These equipotential lines are illustrated in Figure 7b.
- the voltage varies linearly along the side walls, in the transverse direction, from the voltage value at electrode 56 to the voltage value at electrode 51.
- the coatings 57,58 should, therefore, ideally be of uniform thickness. However, such coatings may be difficult to deposit in practice.
- the coatings are replaced by discrete electrodes 59 provided on the side and/or end walls along the lines of intersection with selected equipotentials.
- Each such electrode 59 is maintained at a respective voltage intermediate that at electrode 56 and that at the electrode 51. Since the voltage must vary linearly along each side wall, the electrodes provided thereon may lie on parallel, equally-spaced lines, as shown in Figure 7c, and the required voltages may then be generated by connecting the electrodes together in series between electrode 51 and electrode 56 by means of resistors having equal resistance values.
- Figure 8a shows a transverse cross-sectional view through another monopole electrode structure.
- This structure has a pair of parallel, electrically-insulating side walls 60,61 giving a more compact structure in the transverse (Y-axis) direction.
- the quadrupole field may have rotational symmetry about an axis, the X axis say.
- Such a field could be generated by an electrode structure comprising one electrode having a conical electrode surface and a second electrode having a hyperbolic or spherical electrode surface facing the conical electrode surface. The second electrode would be maintained at a retarding voltage with respect to the first electrode.
- the flight times of ions through the ion mirror are substantially independent of angular deviation in the X-Y plane over a relatively small angular range (typically ⁇ 1° relative to the X-axis direction), provided that the ions are detected by a large area, flat-plate detector extending through the vertex of the V-shaped (or conical) electrode, normal to the X-axis.
- the ions are permitted to pass through the V-shaped (or conical) electrode 51 (for example, if part of the electrode is in the form of a mesh), and travel through a field-free region F before striking a large-area detector D positioned as described, time focussing is preserved. That is to say, the transmission of the device can be increased significantly without degrading resolution. In practice, small adjustments of the detector position in the direction of the X-axis may be necessary to compensate for non-idealities.
- a tandem mass spectrometry system as described finds particular application in the structural analysis of large molecules, for example biological and biochemical samples.
- the ion mirror used in the described system enables the whole of the daughter ion mass spectrum to be detected without the need for re-tuning, and the spectrum can be readily calibrated as a function of mass.
- the flight times of ions through the ion mirror depend on their mass-to-charge ratios, and are entirely independent of their energies, a relatively high mass resolution can be attained, even though the ions are subject to a substantial spreading of their energies due to the effect of the ion buncher on the parent ions and to the fact that the ions entering the ion mirror have a range of different masses.
- the MS mass analyser subjects the parent ions to relatively little time broadening as they travel through the analyser and this also assists in the attainment of a high mass resolving power.
Abstract
A mass spectrometry system comprises a magnetic sector (MS) mass analyser (20) and a time-of-flight (TOF) mass analyser (50) arranged in tandem. The TOF mass analyser (50) comprises an ion mirror having the property that the flight time of an ion through the ion mirror depends on the mass-to-charge ratio of the ion, but is entirely independent of its energy. The mass spectrometry system can be used to analyse the mass spectrum of daughter ions derived from a parent ion of preselected mass.
Description
- This invention relates to mass spectrometry systems.
- There has been an increasing need over recent years to provide mass spectrometry systems capable of analysing greater mass ranges with improved sensitivity. Hitherto, so-called tandem mass spectrometers have been employed, an example being a four-sector mass spectrometer used in combination with an array detector. However, in order to maximise the sensitivity attainable with such arrangements, it has only been feasible to detect a relatively small proportion (typically 10-20%) of the total daughter ion mass spectrum at any given time. Accordingly, the instrument needs to be repeatedly re-tuned in order to detect the entire daughter ion mass spectrum, and this is both inconvenient and time consuming. Furthermore, such arrangements are complex and expensive, and large magnets are needed to detect ions of higher mass.
- A paper by D.H. Russell entitled "A High Sensitivity/High Resolution Tandem Mass Spectrometer for High Mass Biomolecules" presented at the 37th ASMS Conference on Mass Spectrometry and Allied Topics, May 21-26 1989, Miami Beach, Florida discloses a tandem mass spectrometer comprising the combination of a magnetic sector mass analyser and a time-of-flight mass analyser. However, in practice, it is found that an instrument of the kind described in the paper has rather poor mass resolving power.
- It is an object of this invention to provide a mass spectrometry system which at least alleviates the afore-mentioned problems.
- According to the invention, there is provided a mass spectrometry system comprising, an ion source, a magnetic sector mass analyser for selecting source ions having a predetermined mass-to-charge ratio, pulse-forming means for forming source ions into a pulse of ions, means for dissociating ions in the pulse whereby to generate daughter ions from source ions selected by the magnetic sector mass analyser, and a time-of-flight mass analyser for analysing the daughter ions as a function of their mass-to-charge ratios, wherein the time-of-flight mass analyser comprises an ion mirror for subjecting ions to an electrostatic reflecting field in the form of an electrostatic quadrupole field.
- The ion source may be a pulsed ion source, and the pulseforming means may comprise an ion buncher for compressing a pulse of the source ions, into an ion pulse of shorter duration.
- The ion mirror and the ion buncher, in combination, enable a much improved mass resolving power to be achieved compared with that attained by known tandem mass spectrometry systems. Furthermore, use of the ion mirror enables the whole of the mass spectrum (including contributions from both the daughter ions and undissociated parent ions) to be detected without the need for any re-tuning, and the mass spectrum can be easily calibrated against mass with absolute precision.
- The flight times of the ions through the ion mirror to an associated detector depend on their mass-to-charge ratios, and are entirely independent of their energies, and so a high degree of mass resolution can be attained, even though the ions are subject to a substantial spreading of their energies, due to the effect of the ion buncher on the parent ions and to the fact that the ions entering the ion mirror will have a range of different masses.
- The ion mirror may comprise a monopole electrode structure operating at a d.c. voltage, and may comprise a first electrode having an electrode surface of substantially V-shaped transverse cross-section and a second electrode having an electrode surface of curvilinear transverse cross-section facing the electrode surface of the first electrode, wherein the second electrode is maintained, in operation, at a d.c. retarding voltage with respect to the first electrode, and the first electrode is adapted to allow ions to enter and exit the electrostatic reflecting field between the electrode surfaces of the first and second electrodes.
- Alternatively, the monopole electrode structure may comprise an electrically conductive member having a substantially V-shaped transverse cross-section and an electrically resistive member having a substantially V-shaped transverse cross-section, wherein the apex of the electrically resistive member is maintained, in operation, at a d.c. retarding voltage with respect to the electrically conductive member and the electrically conductive member is adapted to allow ions to enter and exit the electrostatic reflecting field bounded by the electrically conductive and the electrically resistive members.
- In another embodiment, the monopole electrode structure may comprise an electrically conductive member having a substantially V-shaped transverse cross-section, electrode means which faces the electrically conductive member and is maintained in operation at a d.c. retarding voltage with respect to the electrically conductive member and electrically insulating side walls, wherein the electrically insulating side walls bear a plurality of electrodes along respective lines of intersection with selected equipotentials in the electrostatic quadrupole field and each said electrode is maintained at a respective voltage.
- The monopole electrode structures may have rotational symmetry about a longitudinal axis intersecting the or each V-shaped electrode.
- The electrostatic reflecting field may reflect each ion about a plane and may include means for controlling the angle of incidence of the ion relative to that plane whereby to control the spatial separation of mass-resolved ions exiting the electrostatic reflecting field.
- The ion buncher may comprise electrostatic means defining a buncher space having an entrance, by which a pulse of ions selected by the magnetic sector mass analyser enters the buncher space, and an exit by which the pulse exits the buncher space, the electrostatic means being operable to apply an electrostatic accelerating (or decelerating) force to ions in a pulse that has entered the buncher space whereby to accelerate (or decelerate) the ions to higher (or lower) energies in proportion to their separation from the exit of the buncher space.
- According to a further aspect of the invention there is provided a mass spectrometry system comprising the serial arrangement of an ion source, a magnetic sector mass analyser and a time-of-flight mass analyser, wherein the time-of-flight mass analyser includes an ion mirror and a detector, and has the property that the flight times of ions through the ion mirror to the detector are related to the mass-to-charge ratios of the ions, and is independent of their energies.
- Mass spectrometry systems in accordance with the invention are now described, by way of example only, with reference to the accompanying drawings, of which:
- Figure 1 is a diagrammatic illustration of a mass spectrometry system in accordance with the invention;
- Figure 2 illustrates an ion buncher used in the mass spectrometry system of Figure 1;
- Figure 3 is a diagrammatic illustration of an ion mirror used in the mass spectrometry system of Figure 1;
- Figure 4 illustrates the flight paths, through the ion mirror of Figure 3, of undissociated parent ions and two daughter ions;
- Figure 5 shows a transverse, cross-sectional view through an ion mirror having a quadrupole electrode structure;
- Figures 6a and 6b show a transverse, cross-sectional view and a perspective view respectively of an ion mirror having a monopole electrode structure;
- Figure 7a shows a transverse, cross-sectional view through an ion mirror having a different monopole electrode structure;
- Figure 7b shows the equipotential lines generated by the monopole electrode structure of Figure 7a;
- Figure 7c shows a side elevational view of a side wall of the monopole electrode structure of Figure 7a;
- Figure 8a shows a transverse, cross-sectional view through a yet further ion mirror having a monopole electrode structure;
- Figure 8b shows a side elevational view of a side wall of the monopole electrode structure of Figure 8a, and
- Figure 9 shows a transverse cross-sectional view through an ion mirror having a yet further monopole electrode structure.
- The mass spectrometry system to be described is used to analyse the mass spectrum of daughter ions derived, by dissociation, from parent ions having a selected mass-to-charge ratio.
- The mass spectrometry system involves the use of a magnetic sector (MS) mass analyser and a time-of-flight (TOF) mass analyser arranged in tandem.
- Referring now to Figure 1 of the drawings, the mass spectrometry system comprises the serial arrangement of a
pulsed ion source 10, aMS mass analyser 20, an ion lens L and a first set of deflector plates D₁, anion buncher 30, acollision cell 40, a second set of deflector plates D₂ and finally aTOF mass analyser 50. - Parent ions produced by the
ion source 10 are admitted to theMS mass analyser 20 in short pulses, typically of 300-500 nsec duration. - A sample under investigation could be ionised using either a laser beam or an ion beam, both of which can be generated in a pulsed mode. The pulses forming the ionising beam may be of relatively short duration, the resulting ions being extracted from the source, for admittance to the
MS mass analyser 20, using a static extraction field. Alternatively, longer ionising pulses could be used, in which case the extraction field would need to be pulsed. However, a pulsed extraction field can be problematical since it tends to give rise to a spreading of ion energies causing an undesirable spreading of flight times through the instrument. - The parent ions produced by the ion source will have a range of different mass-to-charge ratios, and the MS mass analyser is tuned to separate out, for further analysis, only those parent ions having a single, preselected mass-to-charge ratio.
- In this embodiment, the
MS mass analyser 20 is of the well known, double-focussing sector kind which combines magnetic and electric sectors. Figure 1 shows a 'BE' double-focussing sector mass analyser; however, an 'EB' double-focussing sector mass analyser could alternatively be used. This form of MS mass analyser is particularly advantageous in the context of the present mass spectrometry system in that it subjects the parent ion pulses to relatively little time broadening as they travel through the analyser, and this assists in the attainment of a high mass resolving power. However, it is envisaged that other known forms of MS mass analyser could alternatively be used. - The beam of mass-selected parent ions converges on the resolving slit of the
MS mass analyser 20 and diverges as the beam travels beyond the slit. - A typical divergence angle is of the order of ±20 mradians (i.e. ±1°) and for a MS mass analyser having a mass resolving power of 3000, say, the width of the ion beam at the resolving slit would be about 50 µm.
- In order to improve the overall sensitivity of the instrument, it is important that the
TOF mass analyser 50 should collect as large a proportion as possible of the parent ions (and their daughers) exiting the MS mass analyser and, to that end, the ion lens L is designed to refocus the diverging ion beam inside theTOF mass analyser 50. In this example, the lens has a magnification factor of about 10, giving a beam width of about 0.5 mm, and a convergence angle of about ±2m radians, at the entrance to the TOF mass analyser. - Furthermore, as will be explained in greater detail hereinafter, the sensitivity of the
TOF mass analyser 50 depends on the angles of incidence at which ions having different mass-to-charge ratios enter the flight path ofanalyser 50. The two sets of electrostatic deflector plates D₁ and D₂ are used to control these incidence angles with the aim of optimising sensitivity. -
- Accordingly, the mass resolving power R can be improved if the time width ΔT of the ion pulses is as short as possible. For example, ions entering the flight path of the
TOF mass analyser 50 may typically have energies of the order of 8 keV and if the flight path is 1 m, a resolution of 5000 at mass 5000 can only be achieved if the pulse width ΔT is of the order of 14nsec or less. Clearly, this pulse width is much smaller than that provided by thepulsed ion source 10 at the exit of the MS mass analyser - typically 300-500 nsec. - An
ion buncher 30 is therefore provided between the ion lens L and theTOF mass analyser 50 in order to compress the ion pulses produced bysource 10 into pulses of much shorter duration. - As shown in Figure 2, the
ion buncher 30 comprises a pair of parallel electrode plates P₁,P₂ which are normally maintained at ground potential. In order to compress a pulse of the mass-selected parent ions into a much shorter pulse, the upstream plate P₁, that is to say the plate nearer theMS mass analyser 20, is ramped up rapidly to a positive voltage VB (for positive ions) when the pulse lies wholly within the buncher space SB between the electrode plates. - This voltage subjects each ion in the pulse to an electrostatic accelerating force in the direction of the
TOF mass analyser 50, and accelerates the ion to a higher energy by an amount proportional to its separation from the grounded, downstream electrode plate P₂. The downstream ions in the pulse, which entered the buncher space first and are closer to plate P₂, spend less time in the accelerating field than do the upstream ions which entered the buncher space later. Accordingly, the upstream ions tend to catch up with the downstream ions. The distance s separating the two electrode plates, the distance d separating the downstream plate and the entrance to theTOF mass analyser 50, and the voltage VB applied to electrode plate P₁ are chosen so that (in theory) the ions in a pulse all arrive at the entrance to the TOF mass analyser at substantially the same time. - Alternatively, bunching could be achieved using a retarding field which is de-energised once the pulse lies wholly within the buncher space SB between the electrode plates. In this case, the upstream ions are retarded less than the downstream ions, and as before tend to catch up the downstream ions.
- A consequence of subjecting the mass-selected parent ions in each pulse to the accelerating voltage VB is to introduce a significant spreading of their energies. If, for example, the ion pulses produced by
ion source 10 are spread out in space over 50 mm so that they just span the electrode plates P₁,P₂ of the ion buncher, the energies of parent ions arriving at theTOF mass analyser 50 would range from 8keV (the energy of the leading ion in the pulse which receives no energy from the accelerating field) and 12keV (the energy of the trailing ion in the pulse which receives the maximum energy of 4 keV from the accelerating field). - The compressed ion pulse (which may typically have a time width of the order of 10nsec) passes through the
collision cell 40 positioned at the entrance to theTOF mass analyser 50. As an alternative to gas collision it is convenient to use a laser pulse to dissociate the mass-selected parent ions forming the compressed pulse, and since the compressed ion pulse is well defined in both time and space, the laser pulse can be synchronised to coincide with the arrival of each ion pulse at the time focal point. - The daughter ions, produced by dissociation of the mass-selected parent ions, continue on the same trajectory as the parent ions, with very nearly the same velocity, and so there is little effect on the position of the time focal point. However, undissociated parent ions and daughter ions introduced into the
TOF mass analyser 50 will have a substantial energy spread. This is due to the effect of the ion buncher, as described hereinbefore, and to the fact that the parent ions and the daughter ions will have a range of different masses (each daughter ion of mass MD, say, will have a fraction MD/MP of the energy of the parent ion, of mass MP, from which it is derived). - As will now be explained, the
TOF mass analyser 50 enables a high mass resolving power to be attained even though the ions introduced into the flight path of the analyser have different energies. - The
TOF mass analyser 50 comprises a special form of ion mirror, described in our copending European patent application, Publication No. 408,288A1. This form of ion mirror has the property that the flight time of an ion through the ion mirror depends on its mass-to-charge ratio, but is entirely independent of its energy. - Figure 3 illustrates diagrammatically how the ion mirror affects the motion of an ion I as it moves in the X-Z plane along a path T inclined at an angle of incidence α to the longitudinal axis X - as mentioned hereinbefore, the angle of incidence α of the ion can be controlled by the deflector plates D₁,D₂.
- It will be assumed, for clarity of illustration, that the ion mirror establishes an electrostatic field region E bounded by the broken lines F₁,F₂ and that the ion I of mass-to-charge ratio (m/q), say, moving on path T enters the field region at a
point 1, undergoes a reflection at apoint 2, returns on path T' and finally exits the field region at apoint 3. In this illustration, paths T,T' lie in the X-Z plane and the ion I is reflected about the X-Y plane, normal to the page. - The ion is subjected to an electrostatic reflecting force F which increases linearly as a function of the depth of penetration of the ion into the field region. This force acts in the direction of arrow A in Figure 3 and has a magnitude directly proportional to the separation x of the ion from the line joining the exit and
entry points -
- The equation of motion of the ion in the field region is akin to that associated with damped simple harmonic motion, and it can be shown that the time interval t during which the ion travels from the point of
entry 1 to the point ofreflection 2 is given by the expression
Thus, the ion occupies the field region for a total time interval t' given by
As this result shows, the ion occupies the field region E for a time interval which depends only on its mass-to-charge ratio (m/q), and this enables ions to be distinguished from one another as a function of their mass-to-charge ratios, even if, as in the present case, they have different energies. - Figure 4 shows, by way of example, the flight paths followed by undissociated parent ions IP and by two daughter ions ID(1),ID(2) having masses MD(1), MD(2) respectively, wherein MD(1) MD(2) - it will be assumed, in this example, that the ions all have the same charge.
- The undissociated parent ions IP, being the heaviest, have the longest flight time through the field region and they move along the outermost path, whereas the lighter daughter ions ID(2) have the shortest flight time and because they have lower energy they follow the innermost path.
- As will be clear from this illustration, ions having different masses exit the field region at different positions. The ions are detected separately, using a multichannel plate detector, for example, whereby to produce a mass spectrum of all the ions and since, in general, the undissociated parent ions will be much more energetic than the daughter ions the spatial spread of ions exiting the field region could be considerable.
- As explained hereinbefore, the two sets of deflector plates D₁,D₂ are used to control the angle of incidence α of the ions entering the TOF mass analyser, and it is the particular function of the second set of deflector plates D₂ to reduce the spatial spread of ions at the detector, enabling all ions to be detected. To that end, the deflector plates D₂ subject all the ions to an electrostatic deflecting force (in the downwards Z-direction in Figure 3) just before they enter the field region of the ion mirror. However, the relatively light daughter ions have lower energies than the heavier, undissociated parent ions and so suffer a comparatively large deflection, increasing their angles of incidence relative to that of the parent ions and so reducing the spatial spread of the ions received at the detector. In principle, therefore, it is possible to collect all the undissociated parent ions and the daughter ions that constitute the entire mass spectrum.
- Furthermore, a consequence of using this form of energy independent ion mirror is that the
collision cell 40 can be maintained at ground potential, obviating the need for any energy dependent extraction optics. - An ion mirror, as described, uses an electrostatic reflecting field having the form of an electrostatic quadrupole field. Adopting the Cartesian co-ordinate system of Figure 3, the distribution of electrostatic potential V(x,y) in an electrostatic quadrupole field satisfies the condition
where Vo is a constant and x,y are the X,Y position co-ordinates in the field region. - An electrostatic field of this form has four-fold symmetry about the Z-axis and could be generated by a quadrupole electrode structure (which provides field in all four quadrants) or a monopole electrode structure (which provides field on only one of the quadrants).
- Quadrupole and monopole electrode structures are, of course, known in mass analysis spectrometry; however, in contrast to the present arrangement, such known electrode structures operate at radio frequencies.
- A quadrupole electrode arrangement is shown in Figure 5 and this drawing also shows the hyperbolic equipotential lines H in the electrostatic quadrupole field.
- Figures 6a and 6b show a monopole electrode structure.
- The
monopole electrode structure 50, shown in Figures 6a and 6b, comprises twoelongate electrodes - The two electrodes have inwardly facing electrode surfaces which are disposed symmetrically with respect to the X-Z plane and define an intermediate field region E.
-
Electrode 51 has a substantially V-shaped transverse cross-section (subtending an angle of 90°) whereaselectrode 52 is in the form of a rod and has a hyperbolic or, alternatively, a circular transverse cross-section. - As shown in Figures 6b,
electrode 51 has anelongate window 53 by which the ions may enter the field region for reflection in the X-Z plane, one of the electrodes being maintained at a fixed d.c. voltage with respect to the other electrode. If, for example,electrode 52 is maintained at a positive d.c. voltage with respect toelectrode 51, the electrostatic field created in the field region would be such as to reflect positively-charged ions. Conversely, ifelectrode 52 is maintained at a negative d.c. voltage with respect toelectrode 51, the electrostatic field would be such as to reflect negatively-charged ions. - Figure 7a shows a transverse cross-sectional view through an alternative monopole electrode structure. This electrode structure has a pair of orthogonally
inclined side walls electrode 51, as shown, to form a boundary structure enclosing a field region E of square cross-section. Anelectrode 56, positioned at the apex of the side walls, is maintained at an appropriate d.c. retarding voltage with respect to theelectrode 51, and the side walls bearrespective coatings electrode 56 and theelectrode 51. The structure may also have coated end walls (not shown) which serve to terminate electrostatic field lines extending in the Z-axis direction and so, in effect, simulate a structure having infinite length in that direction. - The quadrupole electrostatic field created by this electrode structure has hyperbolic equipotential lines in the transverse (X-Y) plane, as defined by equation 5 above. These equipotential lines are illustrated in Figure 7b. The voltage varies linearly along the side walls, in the transverse direction, from the voltage value at
electrode 56 to the voltage value atelectrode 51. Thecoatings - In an alternative embodiment, the coatings are replaced by
discrete electrodes 59 provided on the side and/or end walls along the lines of intersection with selected equipotentials. Eachsuch electrode 59 is maintained at a respective voltage intermediate that atelectrode 56 and that at theelectrode 51. Since the voltage must vary linearly along each side wall, the electrodes provided thereon may lie on parallel, equally-spaced lines, as shown in Figure 7c, and the required voltages may then be generated by connecting the electrodes together in series betweenelectrode 51 andelectrode 56 by means of resistors having equal resistance values. - The corresponding electrodes on the end walls would lie on hyperbolic lines, as illustrated in Figure 7b.
- Figure 8a shows a transverse cross-sectional view through another monopole electrode structure. This structure has a pair of parallel, electrically-insulating
side walls - The positions of the side walls are represented by the parallel broken lines in Figure 7b. It will be clear from that Figure that the voltage varies in a non-linear fashion along each side wall and, as shown in Figure 8b, the electrodes 59' applied to the side walls are spaced progressively closer together in the
direction approaching electrode 56. - In a yet further embodiment, the quadrupole field may have rotational symmetry about an axis, the X axis say. Such a field could be generated by an electrode structure comprising one electrode having a conical electrode surface and a second electrode having a hyperbolic or spherical electrode surface facing the conical electrode surface. The second electrode would be maintained at a retarding voltage with respect to the first electrode.
- It has been found that the flight times of ions through the ion mirror are substantially independent of angular deviation in the X-Y plane over a relatively small angular range (typically ±1° relative to the X-axis direction), provided that the ions are detected by a large area, flat-plate detector extending through the vertex of the V-shaped (or conical) electrode, normal to the X-axis.
- As illustrated in Figure 9, if the ions are permitted to pass through the V-shaped (or conical) electrode 51 (for example, if part of the electrode is in the form of a mesh), and travel through a field-free region F before striking a large-area detector D positioned as described, time focussing is preserved. That is to say, the transmission of the device can be increased significantly without degrading resolution. In practice, small adjustments of the detector position in the direction of the X-axis may be necessary to compensate for non-idealities.
- A tandem mass spectrometry system as described finds particular application in the structural analysis of large molecules, for example biological and biochemical samples. Furthermore, the ion mirror used in the described system enables the whole of the daughter ion mass spectrum to be detected without the need for re-tuning, and the spectrum can be readily calibrated as a function of mass. Also, because the flight times of ions through the ion mirror depend on their mass-to-charge ratios, and are entirely independent of their energies, a relatively high mass resolution can be attained, even though the ions are subject to a substantial spreading of their energies due to the effect of the ion buncher on the parent ions and to the fact that the ions entering the ion mirror have a range of different masses.
- Furthermore, the MS mass analyser subjects the parent ions to relatively little time broadening as they travel through the analyser and this also assists in the attainment of a high mass resolving power.
Claims (18)
- A mass spectrometry system comprising,
an ion source (10),
a magnetic sector mass analyser (20) for selecting source ions having a predetermined mass-to-charge ratio,
pulse-forming means (30) for forming source ions into a pulse of ions,
means (40) for dissociating ions in the pulse whereby to generate daughter ions from source ions selected by the magnetic sector mass analyser (20), and
a time-of-flight mass analyser (50) for analysing the daughter ions as a function of their mass-to-charge ratios, wherein the time-of-flight mass analyser (50) comprises an ion mirror for subjecting ions to an electrostatic reflecting field in the form of an electrostatic quadrupole field. - A mass spectrometer as claimed in claim 1, wherein the ion source (10) is a pulsed ion source, and the pulseforming means (30) comprises an ion buncher for compressing a pulse of the source ions into an ion pulse of shorter duration.
- A mass spectrometry system as claimed in claim 1, wherein the electrostatic reflecting field reflects the daughter ions about a plane and including means (D₁,D₂) for controlling the angle of incidence (α) of the ions relative to the plane whereby to control the spatial separation of the mass-resolved ions exiting the electrostatic reflecting field.
- A mass spectrometry system as claimed in any one of claims 1 to 3, wherein the ion mirror comprises a monopole electrode structure (51,52; 51,56) operating at a d.c. voltage.
- A mass spectrometry system as claimed in claim 4, wherein the monopole electrode structure (51,56) comprises a first electrode (51) having a substantially V-shaped transverse cross-section, and a second electrode (56) facing the first electrode (51), the second electrode (56) being maintained at a d.c. retarding voltage with respect to the first electrode (51), and including a large-area detector (D) extending through the vertex of first electrode (51), normal to the longitudinal axis thereof.
- A mass spectrometry system as claimed in claim 5, wherein the first electrode (51) has a mesh construction for enabling ions to exit the ion mirror to impinge on the large-area detector (D).
- A mass spectrometry system as claimed in claim 4, wherein the monopole electrode structure (51,52) comprises a first electrode (51) having an electrode surface of substantially V-shaped transverse cross-section and a second electrode (52) having an electrode surface of curvilinear transverse cross-section facing the electrode surface of the first electrode, wherein the second electrode (52) is maintained, in operation, at a d.c. retarding voltage with respect to the first electrode (51) and the first electrode (51) is adapted to allow ions to enter and exit the electrostatic reflecting field between the electrode surfaces of the first and second electrodes (51,52).
- A mass spectrometry system as claimed in claim 4, wherein the monopole electrode structure (51), comprises an electrically conductive member (51) having a substantially V-shaped transverse cross-section and an electrically resistive member (54,55) having a substantially V-shaped transverse cross-section, wherein the apex (56) of the electrically resistive member is maintained, in operation, at a d.c. retarding voltage with respect to the electrically conductive member (51) and the electrically conductive member is adapted to allow ions to enter and exit the electrostatic reflecting field bounded by the electrically conductive and the electrically resistive members (51; 54,55).
- A mass spectrometry system as claimed in claim 8, wherein the monopole electrode structure also has electrically resistive end walls.
- A mass spectrometry system as claimed in claim 4, wherein the monopole electrode structure comprises an electrically conductive member (51) having a substantially V-shaped transverse cross-section, electrode means (56) which faces the electrically conductive member and is maintained in operation at a d.c. retarding voltage with respect to the electrically conductive member and electrically insulating side walls (54,55), wherein the electrically insulating side walls (54,55) bear a plurality of electrodes (59) along respective lines of intersection with selected equipotentials in the electrostatic quadrupole field and each said electrode (59) is maintained at a respective voltage.
- A mass spectrometry system as claimed in claim 10, wherein the electrically insulating side walls (54,55) are formed by an electrically insulating member having a substantially V-shaped transverse cross-section and wherein the electrically conductive member and the electrically insulating member define a closed structure for the electrostatic quadrupole field (E) and said electrode means (56) is located at the apex of the electrically insulating member (54,55).
- A mass spectrometry system as claimed in claim 11, wherein the side walls are parallel.
- A mass spectrometry system as claimed in any one of claims 10 to 12, wherein the monopole electrode structure has electrically insulating end walls bearing a plurality of electrodes along respective lines of intersection with selected equipotentials in the electrostatic quadrupole field, each electrode on the end walls being maintained at a respective voltage.
- A mass spectrometry system as claimed in any one of claims 7, 8, 10 and 11, wherein the monopole electrode structure (51,56) has rotational symmetry about a longitudinal axis intersecting the apex of the or each V-shaped electrode.
- A mass spectrometry system as claimed in any one of claims 2 to 14, wherein the ion buncher (30) comprises electrostatic means (P₁,P₂) defining a buncher space (SB) having an entrance, by which a pulse of ions selected by the magnetic sector mass analyser (20) enters the buncher space (SB), and an exit by which the pulse exits the buncher space, the electrostatic means being operable to apply an electrostatic accelerating (P₁,P₂) (or decelerating) force to ions in a pulse that has entered the buncher space (SB) whereby to accelerate (or decelerate) the ions to higher (or lower) energies in proportion to their separation from the exit of the buncher space.
- A mass spectrometry system as claimed in claim 15, wherein the electrostatic means of the ion buncher comprises respective electrode plates (P₁,P₂) at the entrance to, and the exit from, the buncher space (SB).
- A mass spectrometry system comprising the serial arrangement of an ion source (10), a magnetic sector mass analyser (20) and a time-of-flight mass analyser (50), wherein the time-of-flight mass analyser (50) includes an ion mirror and a detector and has the property that the flight times of ions through the ion mirror to the detector are related to the mass-to-charge ratios of the ions, and is independent of their energies.
- A mass spectrometry system as claimed in claim 15 including means (40) to dissociate the ions before they enter the ion mirror.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| GB929200901A GB9200901D0 (en) | 1992-01-16 | 1992-01-16 | Mass spectrometry systems |
| GB9200901 | 1992-01-16 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| EP0551999A1 true EP0551999A1 (en) | 1993-07-21 |
Family
ID=10708700
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP93300131A Withdrawn EP0551999A1 (en) | 1992-01-16 | 1993-01-08 | Mass spectrometry systems |
Country Status (2)
| Country | Link |
|---|---|
| EP (1) | EP0551999A1 (en) |
| GB (1) | GB9200901D0 (en) |
Cited By (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5464985A (en) * | 1993-10-01 | 1995-11-07 | The Johns Hopkins University | Non-linear field reflectron |
| EP0704879A1 (en) * | 1994-09-30 | 1996-04-03 | Hewlett-Packard Company | Charged particle mirror |
| US5814813A (en) * | 1996-07-08 | 1998-09-29 | The Johns Hopkins University | End cap reflection for a time-of-flight mass spectrometer and method of using the same |
| US6107623A (en) * | 1997-08-22 | 2000-08-22 | Micromass Limited | Methods and apparatus for tandem mass spectrometry |
| GB2402545A (en) * | 2003-05-16 | 2004-12-08 | Micromass Ltd | Reflectron for a mass spectrometer |
| US7427752B2 (en) | 2002-11-15 | 2008-09-23 | Micromass Uk Limited | Mass spectrometer |
| DE10351010B4 (en) * | 2002-11-15 | 2009-09-24 | Micromass Uk Ltd. | mass spectrometry |
| CN104007163A (en) * | 2013-02-26 | 2014-08-27 | 株式会社岛津制作所 | Tandem mass spectrometer and mass spectrometric method |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1983004187A1 (en) * | 1982-06-04 | 1983-12-08 | Research Corporation | Combination of time resolution and mass dispersive techniques in mass spectrometry |
| DE4106796A1 (en) * | 1991-03-04 | 1991-11-07 | Wollnik Hermann | A FLIGHT-TIME MASS SPECTROMETER AS SECOND LEVEL OF AN MS-MS SYSTEM |
| EP0456517A2 (en) * | 1990-05-11 | 1991-11-13 | Kratos Analytical Limited | Time-of-flight mass spectrometer |
| US5073713A (en) * | 1990-05-29 | 1991-12-17 | Battelle Memorial Institute | Detection method for dissociation of multiple-charged ions |
-
1992
- 1992-01-16 GB GB929200901A patent/GB9200901D0/en active Pending
-
1993
- 1993-01-08 EP EP93300131A patent/EP0551999A1/en not_active Withdrawn
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1983004187A1 (en) * | 1982-06-04 | 1983-12-08 | Research Corporation | Combination of time resolution and mass dispersive techniques in mass spectrometry |
| EP0456517A2 (en) * | 1990-05-11 | 1991-11-13 | Kratos Analytical Limited | Time-of-flight mass spectrometer |
| US5073713A (en) * | 1990-05-29 | 1991-12-17 | Battelle Memorial Institute | Detection method for dissociation of multiple-charged ions |
| DE4106796A1 (en) * | 1991-03-04 | 1991-11-07 | Wollnik Hermann | A FLIGHT-TIME MASS SPECTROMETER AS SECOND LEVEL OF AN MS-MS SYSTEM |
Non-Patent Citations (2)
| Title |
|---|
| 37TH ASMS CONFERENCE ON MASS SPECTROMETRY AND ALLIED TOPICS 26 May 1989, FLORIDA pages 1021 - 1022 D. H. RUSSEL 'A HIGH SENSITIVITY / HIGH RESOLUTION TANDEM MASS SPECTROMETER FOR HIGH MASS BIOMOLECULES' * |
| PATENT ABSTRACTS OF JAPAN vol. 9, no. 251 (E-348)8 October 1985 & JP-A-60 101 851 ( ISHIHARA MORIO ) 5 June 1985 * |
Cited By (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5464985A (en) * | 1993-10-01 | 1995-11-07 | The Johns Hopkins University | Non-linear field reflectron |
| EP0704879A1 (en) * | 1994-09-30 | 1996-04-03 | Hewlett-Packard Company | Charged particle mirror |
| US5661300A (en) * | 1994-09-30 | 1997-08-26 | Hewlett-Packard | Charged particle mirror |
| US5814813A (en) * | 1996-07-08 | 1998-09-29 | The Johns Hopkins University | End cap reflection for a time-of-flight mass spectrometer and method of using the same |
| US6107623A (en) * | 1997-08-22 | 2000-08-22 | Micromass Limited | Methods and apparatus for tandem mass spectrometry |
| US7427752B2 (en) | 2002-11-15 | 2008-09-23 | Micromass Uk Limited | Mass spectrometer |
| DE10351010B4 (en) * | 2002-11-15 | 2009-09-24 | Micromass Uk Ltd. | mass spectrometry |
| GB2402545A (en) * | 2003-05-16 | 2004-12-08 | Micromass Ltd | Reflectron for a mass spectrometer |
| GB2402545B (en) * | 2003-05-16 | 2005-11-16 | Micromass Ltd | Mass spectrometer |
| CN104007163A (en) * | 2013-02-26 | 2014-08-27 | 株式会社岛津制作所 | Tandem mass spectrometer and mass spectrometric method |
| CN104007163B (en) * | 2013-02-26 | 2016-09-28 | 株式会社岛津制作所 | Tandem mass spectrometer and mass spectrography |
Also Published As
| Publication number | Publication date |
|---|---|
| GB9200901D0 (en) | 1992-03-11 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP1060502B1 (en) | A tandem time-of-flight mass spectrometer with delayed extraction and method for use | |
| EP0456517B1 (en) | Time-of-flight mass spectrometer | |
| US5464985A (en) | Non-linear field reflectron | |
| US6489610B1 (en) | Tandem time-of-flight mass spectrometer | |
| EP0917728B1 (en) | Ion storage time-of-flight mass spectrometer | |
| EP0587707B1 (en) | Time-of-flight mass spectrometer with an aperture enabling tradeoff of transmission efficiency and resolution | |
| US6717132B2 (en) | Gridless time-of-flight mass spectrometer for orthogonal ion injection | |
| US5077472A (en) | Ion mirror for a time-of-flight mass spectrometer | |
| CN112514029A (en) | Multi-pass mass spectrometer with high duty cycle | |
| WO1995033279A1 (en) | Tandem mass spectrometry apparatus | |
| CN101523548A (en) | Multi-reflecting time-of-flight mass analyser and a time-of-flight mass spectrometer including the mass analyser | |
| GB2274197A (en) | Time-of-flight mass spectrometer | |
| US7075065B2 (en) | Time of flight mass spectrometry apparatus | |
| US7223966B2 (en) | Time-of-flight mass spectrometers with orthogonal ion injection | |
| JP4398370B2 (en) | Quadrupole mass spectrometer with spatial dispersion | |
| EP1310982B1 (en) | Time-of-flight mass spectrometer | |
| EP0551999A1 (en) | Mass spectrometry systems | |
| US5120958A (en) | Ion storage device | |
| WO2001027971A1 (en) | Momentum acceleration orthogonal time of flight mass spectrometer | |
| GB2361806A (en) | Time of flight mass spectrometry apparatus |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): DE FR GB |
|
| 17P | Request for examination filed |
Effective date: 19931101 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: THE APPLICATION HAS BEEN WITHDRAWN |
|
| 18W | Application withdrawn |
Withdrawal date: 19940129 |