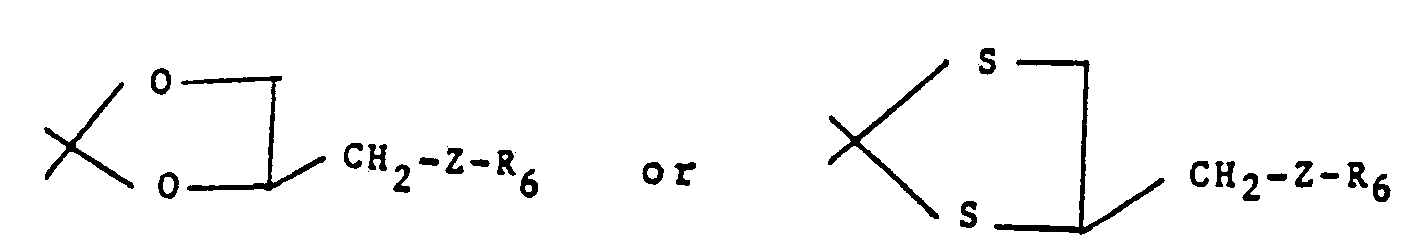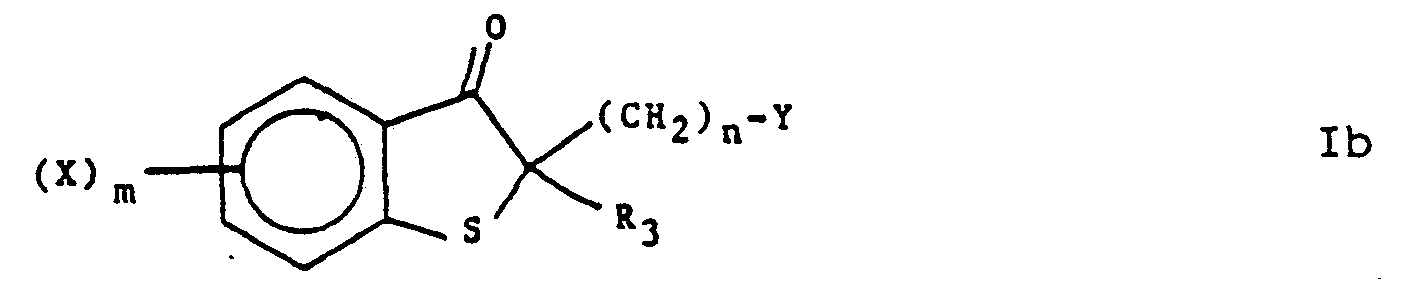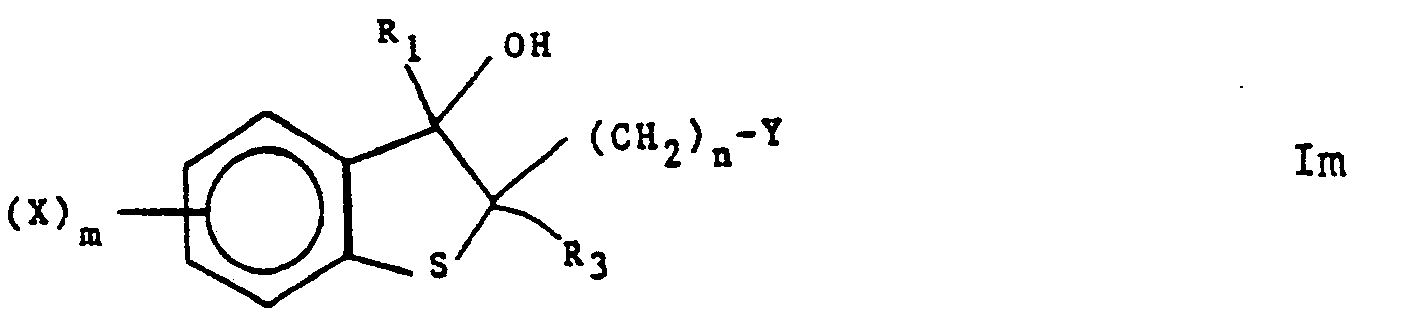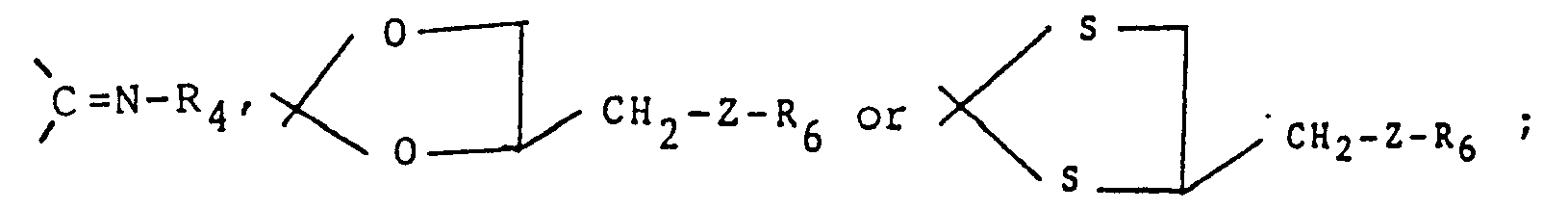EP0212428B1 - Dihydrobenzo/b /thiophenes and pharmaceutical compositions thereof useful as antifungals - Google Patents
Dihydrobenzo/b /thiophenes and pharmaceutical compositions thereof useful as antifungals Download PDFInfo
- Publication number
- EP0212428B1 EP0212428B1 EP86110821A EP86110821A EP0212428B1 EP 0212428 B1 EP0212428 B1 EP 0212428B1 EP 86110821 A EP86110821 A EP 86110821A EP 86110821 A EP86110821 A EP 86110821A EP 0212428 B1 EP0212428 B1 EP 0212428B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- compound
- formula
- alkyl
- substituted
- dihydro
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Lifetime
Links
- 0 CC1(CCC2SC(*)(*)CC2CC1)C1CCCCCCCC1 Chemical compound CC1(CCC2SC(*)(*)CC2CC1)C1CCCCCCCC1 0.000 description 14
- UAFDPKWGFZODRL-UHFFFAOYSA-N N=C=NC(CO)O Chemical compound N=C=NC(CO)O UAFDPKWGFZODRL-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D409/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms
- C07D409/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing two hetero rings
- C07D409/06—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing two hetero rings linked by a carbon chain containing only aliphatic carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D231/00—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings
- C07D231/02—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings not condensed with other rings
- C07D231/10—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members
- C07D231/12—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to ring carbon atoms
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/04—Antibacterial agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/10—Antimycotics
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D233/00—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, not condensed with other rings
- C07D233/54—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, not condensed with other rings having two double bonds between ring members or between ring members and non-ring members
- C07D233/56—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, not condensed with other rings having two double bonds between ring members or between ring members and non-ring members with only hydrogen atoms or radicals containing only hydrogen and carbon atoms, attached to ring carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D249/00—Heterocyclic compounds containing five-membered rings having three nitrogen atoms as the only ring hetero atoms
- C07D249/02—Heterocyclic compounds containing five-membered rings having three nitrogen atoms as the only ring hetero atoms not condensed with other rings
- C07D249/08—1,2,4-Triazoles; Hydrogenated 1,2,4-triazoles
Definitions
- This invention relates to 2-alkyl-, 2-alkenyl and 2-alkynyl-2,3-dihydro-2-[1H-azolyl(C 1 -C 2 )alkyl]-benzo[b]thiophenes and related derivatives which exhibit antifungal activity, pharmaceutical compositions thereof and methods for their use in treating fungal infections in a host including warm-blooded animals such as humans.
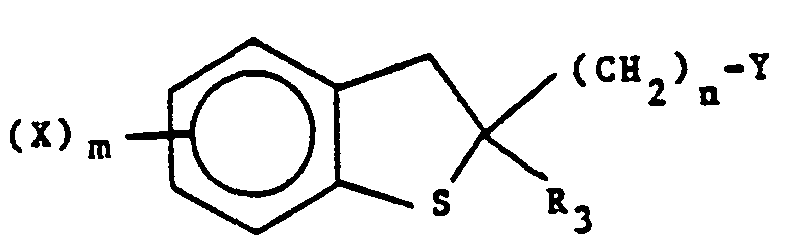
- the compounds of this invention are represented by the following formula I: in racemic or optically active form, or a pharmaceutically acceptable salt thereof wherein
- This invention also provides a pharmaceutical composition
- a pharmaceutical composition comprising an antifungally effective amount of a compound represented by formula I or a pharmaceutically acceptable acid salt thereof, together with a pharmaceutically acceptable carrier or diluent.
- This invention further provides a method of treating susceptible fungal infections which comprises administering to a host, e.g., warm-blooded animals including humans, in need of such treatment an antifungally effective amount of a compound represented by formula I or a pharmaceutical composition comprising such a compound and a pharmaceutically acceptable carrier or diluent.
- halogen means bromine, chlorine or fluorine with chlorine and fluorine being prefered; fluorine is most preferred.
- (lower)alkyl refers to straight and branched chain hydrocarbon groups of 1 to 6 carbon atoms, such as methyl, ethyl, n- and iso-propyl, n-, sec- and tert-butyl, n-, sec-, iso-, tert- and neo-pentyl, n-, sec-, iso- and tert-hexyl.
- (lower)alkanoyl refers to straight and branched chain alkanoyl groups having 2 to 8 carbon atoms such as acetyl, propanoyl, butanoyl, 2-methylpropanoyl, 3-methylpropanoyl, pentanoyl, 2-methylbutanoyl, 3-methylbutanoyl, 4-methylbutanoyl, hexanoyl, 2-methylpentanoyl, 3-methylpentanoyl, 4-methylpentanoyl, 5-methylpentanoyl, heptanoyl, 2-methylheptanoyl, octanoyl, 2-ethylhexanoyl and the like. Acetyl is preferred.
- heterocyclyl refers to five- and six-membered ring systems containing carbon and one to four heteroatoms chosen from N, O and S.
- suitable heterocyclyls include morpholino, piperazino, pyrrolidino, piperidino, imidazolyl, 1,2,4-triazolyl, furanyl, thienyl, thiadiazolyls (especially 1,2,3-thiadiazol-4-yl and 1,2,3-thiadiazol-5-yl), thiomorpholino, and pyridyls.
- the heterocyclyl may be attached via a carbon atom, e.g., N-methylpiperidin-4-yl and N-methylmorpholin-2-yl, or via the nitrogen atom, e.g., piperidin-1-yl (commonly called piperidino), morpholin-4-yl (commonly called morpholino), N-methylpiperazin-4-yl (commonly called N-methylpiperazino), 1H-1-imidazol-1-yl or 4H-1,2,4-triazol-4-yl.
- Azolyls especially 1 H-1-imidazolyl and 1 H-1,2,4-triazolyl
- pyridyls especially 2-pyridyl, and 2- or 3-thienyls are the preferred heterocyclyls.
- Substituted heterocyclyls include (lower)alkyl heterocyclyls especially N-(lower)alkylheterocyclyls such as N-methylmorpholin-4-yl, N-ethylpiperazino, but also 2-methylpyrrolidino, 4-methylpiperidino, 5-methyl-1H-1,2,4-triazol-3-yl, 3-methyl-1-phenyl-1H-1,2,4-triazol-5-yl, and 2-methylpyridyl; (lower)alkanoyl heterocyclyls such as 2-acetylthiophenyl, 2-acetylpyrrolidino; haloheterocyclys such as 2-halo-3-thienyl, 2,5-dihalo-3-thienyl, and 5-halo-2-thienyl; N-(lower)alkanoyl heterocyclyls such as N-acetylpiperazino and 4-acetylpiperidino; and ary
- (lower)alkynyl as used herein is meant straight and branched chain alkynyl radicals of 2 to 8 carbon atoms, such as 1-butynyl.
- phenylalkyl refers to phenyl(lower)alkyl, especially benzyl, a- and ⁇ -phenylethyl and a-, (3-and y-phenylpropyl.
- the preferred phenylalkyl is benzyl.
- Typical suitable aryl groups include phenyl, halo substituted phenyl such as 4-chlorophenyl or 4-fluorophenyl, 2,4-dichloro- or 2,4-difluorophenyl, 2,5-dichloro- or 2,5-difluorophenyl, 2,6-dichloro- or 2,6-difluorophenyl, 2,4,6-trichloro- or 2,4,6-trifluorophenyl and 2,3,4,6-tetrachloro- and 2,3,4,6-tetrafluorophenyl; (lower)alkanoyl substituted phenyl such as 4- acetylphenyl; phenyl substituted by-NR 4 R 5 such as 4-(N,N-dimethylamino)phenyl; phenyl substituted by two different groups such as 4-nitro-3-trifluoromethylphenyl or 3-nitro-4-trifluoromethylphenyl; (lower)al
- arylcarbonyl refers to a carbonyl group bonded to substituted or unsubstituted phenyl or substituted or unsubstituted phenyl(lower)alkyl wherein the substituents comprise one to four of one or more groups selected from halogen, nitro, -NR 4 R s where R 4 and R s are as defined previously, (lower)alkanoyl, (lower)alkyl, and halo(lower)alkyl.
- Preferred OR 4 groups include benzoyloxy, benzyloxy, substituted benzoyloxy and substituted benzyloxy, said benzoyloxy and benzyloxy substituents being one to four of one or more groups selected from halogen, (lower)alkyl, halo(lower)alkyl, nitro, -NR 5 R 6 where R s and R 6 are as defined previously or (lower)alkanoyl and 2- or 5-halo-3-thienyl, 3-halo-2-thienyl or 2,5-dihalo-3-thienyl.
- Typical suitable substituted benzyloxy groups include 4-chlorobenzyloxy, 4-fluorobenzyloxy, 2,4-dichloro- or 2,4-difluorobenzyloxy, 2-chloro-4-fluorobenzyloxy, 2,3-dichloro- or 2,3-difluorobenzyloxy, 2-chloro-3-fluorobenzyloxy, 2,5-dichloro- or 2,5-difluorobenzyloxy, 2-chloro-5-fluorobenzyloxy, 2,6-dichloro-or 2,6-difluorobenzyloxy, 2-chloro-6-fluorobenzyloxy, 2-, 3- or 4-trifluoromethylbenzyloxy, 2-, 3- or 4-methylbenzyloxy, and 2-, 3- or 4-acetylbenzyloxy.
- Halobenzyloxy groups are preferred; 2,6-difluorobenzyloxy and 2,4-difluorobenzyloxy are particularly
- Typical suitable R 3 groups include 2-propynyl, 2-propenyl, propyl, 1-methyl-2-propenyl, 2-methyl-2-propenyl, 1-methylpropyl, 2-butynyl, 2-butenyl, butyl, 1-methyl-2-butynyl, 1-methyl-2-butenyl, 2-methyl-2-butenyl, 2-ethyl-2-butenyl, 1-phenyl-2-propenyl, 2-phenyl-2-propenyl, 3-phenyl-2-propenyl, 3-phenyl-2-propynyl, 2-(4-fluorophenyl)-2-propenyl, 2-(4-chlorophenyl)-2-propenyl, 2-(2,4-difluorophenyl)-2-butenyl, 2-(2,4-dichlorophenyl)-butyl, 2-(4-nitro-3-trifluoromethyl)-2-propenyl, 2-piperazino-2-propen
- R 3 groups include propyl, 2-propenyl, 2-propynyl, 2-aryl-2-propenyl such as 2-(2,4-difluorophenyl)-2-propenyl and 2-heterocyclyl-2-propenyl such as 2-(N-acetylpiperazino)-2-propenyl.
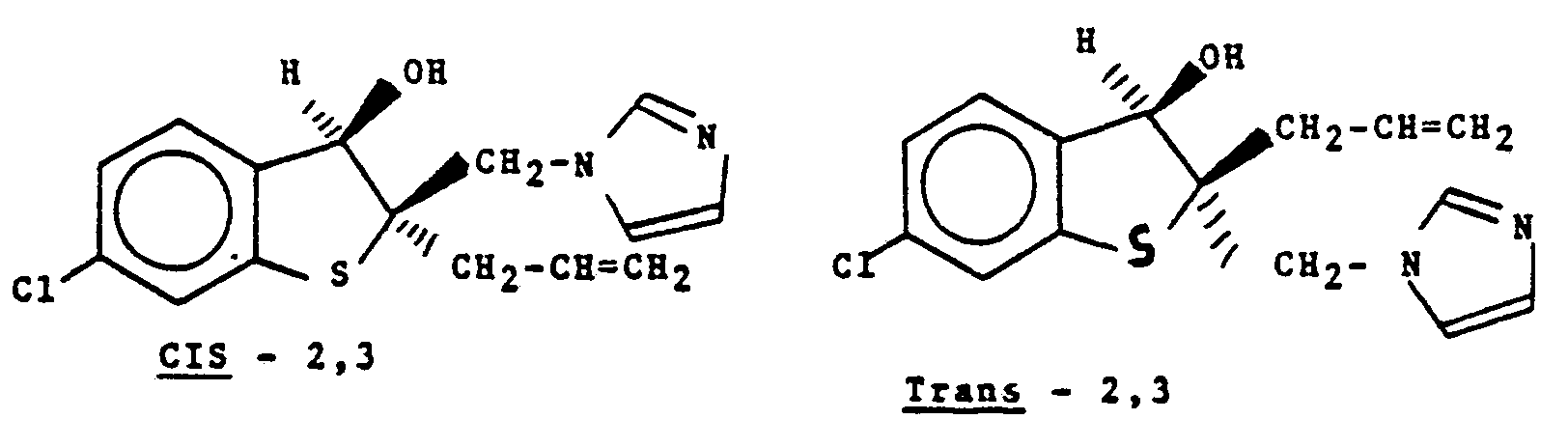
- cis-2,3 the hydroxy and 1 H-imidazolylmethyl groups are both positioned on the same side of the ring.
- the groups are positioned on opposite sides of the ring.
- Both forms are within the scope of the present invention, as are the individual optical isomers, e.g., ( ⁇ )-cis-2,3 and (-)-cis-2,3 which can be obtained by resolution of a racemic mixture [( ⁇ )-cis-2,3] by conventional means well known to those skilled in the art, such as fractional crystallization or chromatography.
- Preferred groups of compounds of this invention include those wherein:
- Preferred compounds include:
- the compounds of the present invention are made by the following process:
- Compounds of formula V may be formed by the following process.
- alkali metal base for example as alkali metal hydroxide especially NaOH or KOH or aqueous solutions thereof, alkali metal hydride, alkali metal amide or alkali metal alcoholate and an allyl compound of formula III, e.g., allyl bromide
- an aprotic organic solvent such as dimethylformamide, (DMF), dimethyl sulfoxide (DMSO), hexamethylphosphoric acid triamide (HMPTA), an ether such as tetrahydrofuran (THF), dioxane or dimethoxyethane, a lower alcohol, or a ketone, e.g. acetone
- PTC phase transfer catalyst
- the oxidation of IV is typically carried out by contacting compounds of formula IV with an oxidizing agent such as a peracid e.g., meta-chloroperbenzoic acid or peracetic acid, or hydrogen peroxide in an organic solvent such as halogenated alkanes, e.g., methylene chloride or chloroform, at about 0°-20°C, preferably 0°-5°C for about 1-4 hours.
- an oxidizing agent such as a peracid e.g., meta-chloroperbenzoic acid or peracetic acid, or hydrogen peroxide in an organic solvent such as halogenated alkanes, e.g., methylene chloride or chloroform, at about 0°-20°C, preferably 0°-5°C for about 1-4 hours.
- the substituted compounds of formula V are heated in a suitable solvent and normally in the presence of an alkanoic acid anhydride for a time sufficient to give the substituted ketone compounds of formula la.
- Typical suitable solvents include aromatic hydrocarbons such as benzene, xylenes, toluene, and halogenated alkanes such as chloroform.
- Typical suitable alkanoic acid anhydrides include acetic anhydride, perchloroacetic anhydride and perfluoroacetic acid anhydride. Temperatures of about 50° to about 100°C for about 1 to 2 hours are normally sufficient.
- the compounds of this invention exhibit broad spectrum antifungal activity, in conventional antifungal screening tests, against human and animal pathogens, such as the following: Aspergillus, Candida, Geotrichum, Microsporum, Monosporium, Rhodotorula, Saccharomyces, Torulopsis and Trichophyton.
- the compounds of this invention exhibit topical fungal activity in in vivo tests in animals that is superior to that for miconazole, a commercial product.
- Candida topical infection model is more potent than miconazole.
- the present invention also provides a pharmaceutical composition
- a pharmaceutical composition comprising an effective antifungal amount of a compound represented by formula I or a pharmaceutically acceptable salt thereof and a pharmaceutically acceptable carrier or diluent.
- the preferred pharmaceutically acceptable salts are nontoxic acid addition salts formed by adding to the compounds of the present invention about a stoichiometric amount of a mineral acid, such as HCI, HBr, H 2 S0 4 or H 3 PO 4 , or of an organic acid, such as acetic, propionic, valeric, oleic, palmitic, stearic, lauric, benzoic, lactic, para-toluene sulfonic, methane sulfonic, citric, maleic, fumaric, succinic and the like.
- a mineral acid such as HCI, HBr, H 2 S0 4 or H 3 PO 4
- an organic acid such as acetic, propionic, valeric, oleic, palmitic, stearic, lauric, benzoic, lactic, para-toluene sulfonic, methane sulfonic, citric, maleic, fumaric, succinic and the like.
- compositions of the present invention may be adapted for oral, parenteral or topical administration. They are formulated by combining the compound of this invention or an equivalent amount of a pharmaceutically acceptable salt thereof with any suitable, inert, pharmaceutically acceptable carrier or diluent.
- the preferred mode of administration is topical.
- compositions for oral administration such as tablets, capsules, pills, powders, granules, solutions, dragees, suspensions or emulsions. They may also be manufactured in the form of sterile solid compositions which can be dissolved in sterile water, physiological saline or some other sterile injectable medium immediately before use.
- Topical dosage forms may be prepared according to procedures well known in the art, and may contain a variety of ingredients, excipients and additives.
- the formulations for topical use include ointments, creams, lotions, powders, aerosols, pessaries and sprays.
- ointments, lotions and creams may contain water, oils, fats, waxes, polyesters, alcohols, or polyols, plus such other ingredients as fragrances, emulsifiers and preservatives.
- Powders are made by mixing the active ingredient with a readily available, inert, pulverous distributing agent, such as talcum, calcium carbonate, tricalcium phosphate, or boric acid. Aqueous suspensions of the above powders may also be made.
- Solutions or emulsions may also be prepared using inert solvents which are preferably nonflammable, odorless, colorless and nontoxic, for example, vegetable oils and isopropanol.
- aerosol or non-aerosol sprays may be prepared using solutions or suspensions in appropriate solvents, e.g., difluorodichloromethane for aerosols.
- Parenteral forms to be injected intravenously, intramuscularly, or subcutanteously are usually in the form of a sterile solution, and may contain salts or glucose to make the solution isotonic.
- the dosages of the compounds of the present invention employed to combat a given fungal infection in animals is generally somewhat less than the dosage requirements of present commercial products such as micoazole.
- the actual preferred dosages of the compounds of the present invention or pharmaceutically acceptable salts thereof will vary according to the particular composition formulated, the mode of application and the particular situs, host and disease being treated. Many factors that modify the action of the drug will be taken into account by the attending clinician, e.g., age, body weight, sex, diet, time of administration, rate of excretion, condition of the host, drug combinations, reaction sensitivities and severity of the disease. Administration can be carried out continuously or periodically within the maximum tolerated dose. Optimal application rates for a given set of conditions can be readily ascertained by the attending clinician using conventional dosage determination tests.
- topical dosage for humans ranges from about 50 mg per day to about 800 mg per day, in single or divided doses, with about 100 mg to about 200 mg per day being preferred. Each dose should be mixed with about 10 g of carrier.
- the oral dosage for human ranges from about 50 mg to about 800 mg per day, in single or divided doses, with about 100 mg to about 400 mg per day being preferred.
- parenteral dosage for humans ranges for about 5 mg per day to about 50 mg per day, in single or divided doses, with about 10 mg per day being preferred.
- Example 3C Following the procedure of Example 3C but substitute an equivalent quantity of the compound of Example 5B for the title compound of Examle 3C to give the corresponding benzo[b]thiophene-3-one which is thereafter treated in accordance with the procedure of Example 3D to provide the mixture of ( ⁇ )-cis and ( ⁇ )-trans-isomers of the title compound of this Example. Fractionally crystallize the mixture to obtain the title compounds of this Example, Part C.
- Example 12 which is similar to that of Example 3, but omit the step that is similar to Step D, Example 3 to yield the title compound of the Example, Part A.
- compositions containing as the active ingredient such as ( ⁇ )-cis-2-allyl-6-chloro-2,3-dihydro-3-hydroxy-2-(1H-1-imidazolylmethyl)benzo[b]thiophene or ( ⁇ )-cis-2-allyl-6-chloro-3-(2-chloro-6-fluorobenzyloxy)-2,3-dihydro-2-(1H-1-imidzolylmethyl)benzo[b]thiophene. It will be appreciated, however, that either of these compounds may be replaced by equally effective quantities of other compounds of this invention.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Communicable Diseases (AREA)
- Pharmacology & Pharmacy (AREA)
- Oncology (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Plural Heterocyclic Compounds (AREA)
Description
- This invention relates to 2-alkyl-, 2-alkenyl and 2-alkynyl-2,3-dihydro-2-[1H-azolyl(C1-C2)alkyl]-benzo[b]thiophenes and related derivatives which exhibit antifungal activity, pharmaceutical compositions thereof and methods for their use in treating fungal infections in a host including warm-blooded animals such as humans.
- Various antifungal 2,3-dihydro-2-(1H-imidazolylmethyl)benzo[b]thiophenes are known. For example U.S. Patent 4,431,816 discloses 3-hydroxy-2,3-dihydro-2-(1H-1-imidazolylmethyl)benzo[b]thiophenes and European Patent Application EP 54,233 discloses cis-6-chloro-3-allyloxy-2,3-dihydro-2-(l H-I-imidazolylmethyl)benzo[b]thiophene.
- However, none of the references are directed to the 2-alkyl, 2-alkenyl or 2-alkynyl compounds of this invention.
-
- X is one or more groups selected independently from halogen, Cl-6 alkyl, halo C1-C6 alkyl, cyano, nitro or substituted and unsubstituted phenyl, or substituted or unsubstituted phenyl Cl-6 alkyl wherein the substituents comprise one to four of one or more groups selected from halogen, nitro, -NR4R5, C2-8 alkanoyl, C1-6 alkyl, and halo C1-6 alkyl;
- Y is substituted or unsubstituted imidazolyl or substituted or unsubstituted 1,2,4-triazolyl, said substituents being one to three of one or more groups selected independently from C1_6 alkyl or substituted and unsubstituted phenyl, or substituted or unsubstituted phenyl C1_6 alkyl wherein the substituents comprise one to four of one or more groups selected from halogen, nitro, ―NR4R5, C2-8 alkanoyl, Ci-6 alkyl, and halo C1_6 alkyl;
- R1 and R2 are independently hydrogen, -CH2NR4R5, ―NR4R5, -OR4, -SRS, C1_6 alkyl, C2_8 alkynyl, halogen, or substituted and unsubstituted phenyl, or substituted or unsubstituted phenyl C1_6 alkyl wherein the substituents comprise one to four of one or more groups selected from halogen, nitro, ―NR4R5, C2_8 alkanoyl, C1_6 alkyl, and halo C1_6 alkyl or R1 and R2 together with the carbon atom to which they are attached form carbonyl, thiocarbonyl,
- R4 and R5 are independently hydrogen, C1_6 alkyl, N,N-diC1_6 alkyl carbamoyl, N,N-diC1_6 alkyl thiocarbamoyl, arylcarbonyl, or substituted or unsubstituted phenyl, or substituted or unsubstituted phenyl C1_6 alkyl wherein the substituents comprise one to four of one or more groups selected from halogen, nitro, ―NR4R5, C2_8 alkanoyl, C1_6 alkyl, and halo C1_6 alkyl, or R4 and R5 taken together with the nitrogen atom to which they are attached in -NR4R5 and -CH2NR4R5 form substituted or unsubstituted heterocyclyl as defined above;
- Z is 0, S or NR5 where where R5 is as defined previously;
- m is 1, 2, 3 or 4; and
- n is 1 or 2.
- This invention also provides a pharmaceutical composition comprising an antifungally effective amount of a compound represented by formula I or a pharmaceutically acceptable acid salt thereof, together with a pharmaceutically acceptable carrier or diluent.
- This invention further provides a method of treating susceptible fungal infections which comprises administering to a host, e.g., warm-blooded animals including humans, in need of such treatment an antifungally effective amount of a compound represented by formula I or a pharmaceutical composition comprising such a compound and a pharmaceutically acceptable carrier or diluent.
- As used in the specification and claims, the term "halogen" means bromine, chlorine or fluorine with chlorine and fluorine being prefered; fluorine is most preferred. The term "(lower)alkyl" refers to straight and branched chain hydrocarbon groups of 1 to 6 carbon atoms, such as methyl, ethyl, n- and iso-propyl, n-, sec- and tert-butyl, n-, sec-, iso-, tert- and neo-pentyl, n-, sec-, iso- and tert-hexyl. The term "halo(lower)alkyl" refers to "(lower)alkyl" groups having at least one halogen substituent, e.g., -CH2-CF3, -CF2-CH3 as well as perhalo groups such as -CF=CF3 or -CF3; trifluoromethyl is preferred. The term "(lower)alkanoyl" refers to straight and branched chain alkanoyl groups having 2 to 8 carbon atoms such as acetyl, propanoyl, butanoyl, 2-methylpropanoyl, 3-methylpropanoyl, pentanoyl, 2-methylbutanoyl, 3-methylbutanoyl, 4-methylbutanoyl, hexanoyl, 2-methylpentanoyl, 3-methylpentanoyl, 4-methylpentanoyl, 5-methylpentanoyl, heptanoyl, 2-methylheptanoyl, octanoyl, 2-ethylhexanoyl and the like. Acetyl is preferred.
- The term "heterocyclyl" refers to five- and six-membered ring systems containing carbon and one to four heteroatoms chosen from N, O and S. Typical suitable heterocyclyls include morpholino, piperazino, pyrrolidino, piperidino, imidazolyl, 1,2,4-triazolyl, furanyl, thienyl, thiadiazolyls (especially 1,2,3-thiadiazol-4-yl and 1,2,3-thiadiazol-5-yl), thiomorpholino, and pyridyls. The heterocyclyl may be attached via a carbon atom, e.g., N-methylpiperidin-4-yl and N-methylmorpholin-2-yl, or via the nitrogen atom, e.g., piperidin-1-yl (commonly called piperidino), morpholin-4-yl (commonly called morpholino), N-methylpiperazin-4-yl (commonly called N-methylpiperazino), 1H-1-imidazol-1-yl or 4H-1,2,4-triazol-4-yl. Azolyls, especially 1 H-1-imidazolyl and 1 H-1,2,4-triazolyl, and pyridyls, especially 2-pyridyl, and 2- or 3-thienyls are the preferred heterocyclyls.
- Substituted heterocyclyls include (lower)alkyl heterocyclyls especially N-(lower)alkylheterocyclyls such as N-methylmorpholin-4-yl, N-ethylpiperazino, but also 2-methylpyrrolidino, 4-methylpiperidino, 5-methyl-1H-1,2,4-triazol-3-yl, 3-methyl-1-phenyl-1H-1,2,4-triazol-5-yl, and 2-methylpyridyl; (lower)alkanoyl heterocyclyls such as 2-acetylthiophenyl, 2-acetylpyrrolidino; haloheterocyclys such as 2-halo-3-thienyl, 2,5-dihalo-3-thienyl, and 5-halo-2-thienyl; N-(lower)alkanoyl heterocyclyls such as N-acetylpiperazino and 4-acetylpiperidino; and aryl substituted heterocyclyls include heterocyclyls substituted by aryl as defined herein such as N-phenylpiperazino, N-(4-chlorophenyl)piperazino, 2-(4-trifluoromethylphenyl)piperazino, and the like. Substituted azolyls, thiadiazolyls, thienyls, and pyridyls are the preferred substituted heterocyclyls.
- By the term "(lower)alkynyl" as used herein is meant straight and branched chain alkynyl radicals of 2 to 8 carbon atoms, such as 1-butynyl.
- The term "phenylalkyl" refers to phenyl(lower)alkyl, especially benzyl, a- and β-phenylethyl and a-, (3-and y-phenylpropyl. The preferred phenylalkyl is benzyl. Typical suitable aryl groups include phenyl, halo substituted phenyl such as 4-chlorophenyl or 4-fluorophenyl, 2,4-dichloro- or 2,4-difluorophenyl, 2,5-dichloro- or 2,5-difluorophenyl, 2,6-dichloro- or 2,6-difluorophenyl, 2,4,6-trichloro- or 2,4,6-trifluorophenyl and 2,3,4,6-tetrachloro- and 2,3,4,6-tetrafluorophenyl; (lower)alkanoyl substituted phenyl such as 4- acetylphenyl; phenyl substituted by-NR4R5 such as 4-(N,N-dimethylamino)phenyl; phenyl substituted by two different groups such as 4-nitro-3-trifluoromethylphenyl or 3-nitro-4-trifluoromethylphenyl; (lower)alkyl substituted phenyl such as 4-methylphenyl, 2,4-dimethylphenyl, 2,4,6-trimethylphenyl or 2,3,4,6-tetramethylphenyl; halo(lower)alkyl substituted phenyl such as 4-trifluoromethylphenyl, 4-(1,1-difluoroethyl)phenyl and similarly substituted phenylalkyl groups, especially similarly substituted benzyl groups. Difluoro and trifluorophenyl are preferred aryl groups; 2,6-difluorophenyl is more preferred.
- The term "arylcarbonyl" refers to a carbonyl group bonded to substituted or unsubstituted phenyl or substituted or unsubstituted phenyl(lower)alkyl wherein the substituents comprise one to four of one or more groups selected from halogen, nitro, -NR4Rs where R4 and Rs are as defined previously, (lower)alkanoyl, (lower)alkyl, and halo(lower)alkyl.
- Preferred OR4 groups include benzoyloxy, benzyloxy, substituted benzoyloxy and substituted benzyloxy, said benzoyloxy and benzyloxy substituents being one to four of one or more groups selected from halogen, (lower)alkyl, halo(lower)alkyl, nitro, -NR5R6 where Rs and R6 are as defined previously or (lower)alkanoyl and 2- or 5-halo-3-thienyl, 3-halo-2-thienyl or 2,5-dihalo-3-thienyl.
- Typical suitable substituted benzyloxy groups include 4-chlorobenzyloxy, 4-fluorobenzyloxy, 2,4-dichloro- or 2,4-difluorobenzyloxy, 2-chloro-4-fluorobenzyloxy, 2,3-dichloro- or 2,3-difluorobenzyloxy, 2-chloro-3-fluorobenzyloxy, 2,5-dichloro- or 2,5-difluorobenzyloxy, 2-chloro-5-fluorobenzyloxy, 2,6-dichloro-or 2,6-difluorobenzyloxy, 2-chloro-6-fluorobenzyloxy, 2-, 3- or 4-trifluoromethylbenzyloxy, 2-, 3- or 4-methylbenzyloxy, and 2-, 3- or 4-acetylbenzyloxy. Halobenzyloxy groups are preferred; 2,6-difluorobenzyloxy and 2,4-difluorobenzyloxy are particularly preferred.
- Typical suitable R3 groups include 2-propynyl, 2-propenyl, propyl, 1-methyl-2-propenyl, 2-methyl-2-propenyl, 1-methylpropyl, 2-butynyl, 2-butenyl, butyl, 1-methyl-2-butynyl, 1-methyl-2-butenyl, 2-methyl-2-butenyl, 2-ethyl-2-butenyl, 1-phenyl-2-propenyl, 2-phenyl-2-propenyl, 3-phenyl-2-propenyl, 3-phenyl-2-propynyl, 2-(4-fluorophenyl)-2-propenyl, 2-(4-chlorophenyl)-2-propenyl, 2-(2,4-difluorophenyl)-2-butenyl, 2-(2,4-dichlorophenyl)-butyl, 2-(4-nitro-3-trifluoromethyl)-2-propenyl, 2-piperazino-2-propenyl, 2-(N-acetylpiperazino)-2-butyl, 2-(N-propyl piperazino)-2-pentenyl, 2-(4-acetylpiperidino)-2-butenyl, 2-[N-(4-trifluoromethylphenyl)]piperazino-2-propenyl and 2-benzyl-2-propenyl, 2-(2,4-difluorobenzyl)-2-butenyl and the like. The cis- and trans- isomers, e.g. cis- and trans-2-butenyl, are also contemplated. Preferred R3 groups include propyl, 2-propenyl, 2-propynyl, 2-aryl-2-propenyl such as 2-(2,4-difluorophenyl)-2-propenyl and 2-heterocyclyl-2-propenyl such as 2-(N-acetylpiperazino)-2-propenyl.
-
- In the formula labelled cis-2,3, the hydroxy and 1 H-imidazolylmethyl groups are both positioned on the same side of the ring. In the trans-2,3 formula, the groups are positioned on opposite sides of the ring. Both forms are within the scope of the present invention, as are the individual optical isomers, e.g., (±)-cis-2,3 and (-)-cis-2,3 which can be obtained by resolution of a racemic mixture [(±)-cis-2,3] by conventional means well known to those skilled in the art, such as fractional crystallization or chromatography.
- Preferred groups of compounds of this invention include those wherein:
- (1) one of R' and R2 is hydrogen and the other is hydroxyl, and/or
- (2) n is 1; and/or
- (3) Y is imidazol-1-yl, and/or
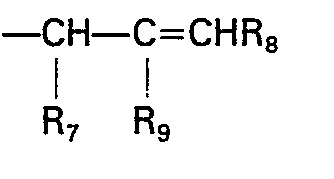
- (4) R3 is -CHR7-CR9=CHR8.
- Preferred compounds include:
- 2-allyl-6-chloro-2,3-dihydro-3-hydroxy-2-(1H-1-imidazolylmethyl)benzo[b]thiophene,
- 2-allyl-6-chloro-2,3-dihydro-3-fluoro-2-(1H-1-imidazolylmethyl)benzo[b]thiophene,
- 2-allyl-3-(2-chloro-6-fluorobenzyloxy)-2,3-dihydro-6-fluoro-2-(1H-1-imidazolylmethyl)benzo[b]thiophene,
- 2-allyl-3-(2,6-difluorobenzyloxy)-2,3-dihydro-6-fluoro-2-(1H-1-imidazolylmethyl)benzo[b]thiophene,
- 2-allyl-2,3-dihydro-6-chloro-2-(1H-1-imidazolylmethyl)benzo[b]thiophene,
- 2-allyl-2,3-dihydro-5-fluoro-3-hydroxy-2-(1,2,4-triazol-1-ylmethyl)benzo[b]thiophene,
- 6-chloro-2,3-dihydro-3-hydroxy-2-(1H-1-imidazolylmethyl)-2-[2-(4-chlorophenyl)-2-propenyl]benzo[b]-thiophene, and
- 6-chloro-2,3-dihydro-3-(1H-1-imidazolyl)-2-(1H-imidazol-1-ylmethyl)-2-allyl-benzo[b]thiophene.
- The compounds of the present invention are made by the following process:
-
- Compounds of formula V may be formed by the following process.
- Substituted hydroxy compounds of the formula II wherein X, Y, m and n have the meanings given above are reacted with an allyl compound of the formula III, i.e., e.g., allyl-LG wherein LG is a leaving group such as halide, e.g., bromide, tosylate, mesylate and wherein R7, Ra and R9 are as defined above, in the presence of an alkali metal base (MOH), a solvent or mixture of solvents for a time and temperature sufficient to give the substituted allyl ether of general formula IV to the compound of formula V as depicted in the following reaction scheme,
- The reaction of the compounds of formula II with an alkali metal base, for example as alkali metal hydroxide especially NaOH or KOH or aqueous solutions thereof, alkali metal hydride, alkali metal amide or alkali metal alcoholate and an allyl compound of formula III, e.g., allyl bromide, is carried out in an aprotic organic solvent such as dimethylformamide, (DMF), dimethyl sulfoxide (DMSO), hexamethylphosphoric acid triamide (HMPTA), an ether such as tetrahydrofuran (THF), dioxane or dimethoxyethane, a lower alcohol, or a ketone, e.g. acetone, and in the presence of a phase transfer catalyst (PTC) such as tricaprylylmethylammonium chloride at 0-60°C, especially 20°-40°C for 1-6 hours.
- Compounds of formula II are prepared in accordance with known methods, such as the procedures disclosed in U.S. Patent 4,352,808 at Col. 5, lines 13-43 and at Col. 11 line 1 to Col. 12 line 9 (Preparation No. 1). The preparation of many compounds of formula II is illustrated in the examples which follow.
- Compounds of formula IV are oxidized to give the substituted sulfoxide of formula V.
- The oxidation of IV is typically carried out by contacting compounds of formula IV with an oxidizing agent such as a peracid e.g., meta-chloroperbenzoic acid or peracetic acid, or hydrogen peroxide in an organic solvent such as halogenated alkanes, e.g., methylene chloride or chloroform, at about 0°-20°C, preferably 0°-5°C for about 1-4 hours.
-
- Typical suitable solvents include aromatic hydrocarbons such as benzene, xylenes, toluene, and halogenated alkanes such as chloroform.
- Typical suitable alkanoic acid anhydrides include acetic anhydride, perchloroacetic anhydride and perfluoroacetic acid anhydride. Temperatures of about 50° to about 100°C for about 1 to 2 hours are normally sufficient.
- The reaction of the compound of formula V to form a compound of formula la, which is in accordance with the invention, is followed if desired by one or more of sub-processes (i) to (xiii) listed below:
- (i) to produce a compound of formula I wherein R, and R2' together with the carbon atom to which they are attached, form the group
Compounds of the formulas Via and Vlb are standard chemical reagents such as disclosed in "Compendium of Organic Synthetic Methods" by I. T. Harrison and S. Harrison, Wiley-Interscience, pages 449-456, NY 1971 and "Steroid Reactions", C. Djerassi, Ed. Holden-Day Inc., pages 1-66 1963. For example, the ketals and thioketals of formula Ic and Ic' may be prepared by an exchange reaction of Ib with dioxolans and dithioxolans of formulas Via and Vlb, respectively,
Typical dioxolans of formula Via wherein Z is NH may be prepared by reductive amination of 2,2-dimethyl-4-formyl-1,3-dioxolans of the formula VII which are disclosed in Swiss Patent Appin. CH644,855A (8/31/84). - (ii) to produce a compound of formula I wherein R1and R2, together with the carbon atom to which they are attached form the group
- (iii) to produce a compound of formula I wherein R, and R2, together with the carbon atom to which they are attached, form a thiocarbonyl group, reacting a compound of the formula
- (iv) to form a compound of formula I wherein one of R1 is hydrogen and the other is hydroxyl, reducing a compound of the following formula with a metal boro hydride or aluminum hydride
- (v) To form a compound of formula I wherein one of R1 and R2 is hydrogen, and the other is -NR4R5, -OR4, or-SR5, reacting a compound of the formula
Especially suitable compounds of the formula L3-R4 and L5R5 are substituted benzyl or benzoyl halides which are commercially available or are made by synthetic procedures well known in the art. Typical suitable substituted benzyl halides are the benzyl chlorides or bromides substituted by groups described in reference to the substituted benzyloxy groups above. Typical suitable benzoyl halides include 2-, 3-, or 4- monohalo (e.g., chloro or fluoro) benzoyl chloride and 2,4-dihalo, 2,5-dihalo or 2,6-dihalo (e.g., 2,6-difluorobenzoyl chloride). Suitable bases include sodium and potassium hydroxide, etc. - (vi) To make a compound of formula I wherein R3 is-CHR7-CHR9-CH2R8, catalytically hydrogenating a compound of formula Ij
- (vii) To produce a compound of formula I wherein R3 is-CHR7-C=CR8, halogenating a compound of the formula
- (viii) To product a compound of formula I wherein one of R1 or R2 is halogen, reacting a compound of the following formula lm
- (ix) To produce a compound offormula I wherein both R1 and R2 are hydrogen, reacting a compound of the following formula
- (x) To produce a compound wherein R2 is substituted or unsubstituted heterocyclyl, said substituents being as previously defined, reacting a compound of the formula
- H-Het
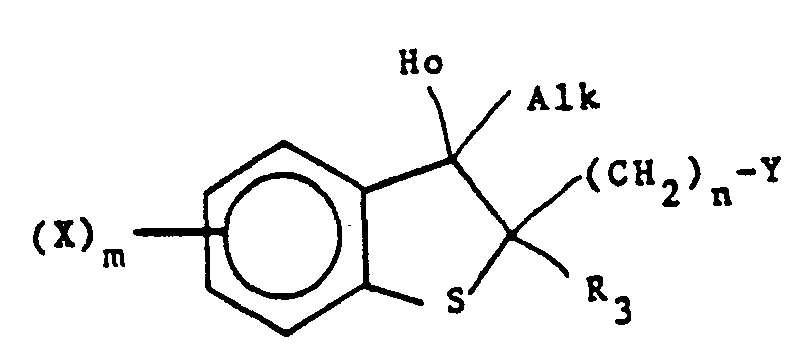
to form a compound of the formula - (xi) To produce a compound of formula I wherein one of R, and R2 is (lower)alkyl or (lower)alkynyl and the other is hydroxyl, reacting a compound of the formula
Typical compounds that introduce hydrogen and a (lower)alkynyl group are (lower)alkynes such as 3,3-dimethyl-1-butyne. The reaction takes place at room temperature or below in solvent, such as THF.
Typical compounds that introduce hydrogen and a (lower)alkyl group are Grignard reagents such as CH3MgBr, which are well known in the art. The reaction with the Grignard reagent takes place in solvent, such as THF, at room temperature or higher. - (xii) After the desired compound has been produced, it may be isolated from its optical isomers by chromatography or fractional crystallation.
- (xiii) The compounds may be converted to pharmaceutically acceptable salts by known methods.
- The compounds of this invention exhibit broad spectrum antifungal activity, in conventional antifungal screening tests, against human and animal pathogens, such as the following: Aspergillus, Candida, Geotrichum, Microsporum, Monosporium, Rhodotorula, Saccharomyces, Torulopsis and Trichophyton.
- The compounds of this invention exhibit topical fungal activity in in vivo tests in animals that is superior to that for miconazole, a commercial product. For example, (±)-cis-2-allyl-6-chloro-2,3-dihydro-3-hydroxy-2-(1H-1-imidazolylmethyl)-benzo[b]thiophene in a hamster vaginal Candida topical infection model is more potent than miconazole.
- The present invention also provides a pharmaceutical composition comprising an effective antifungal amount of a compound represented by formula I or a pharmaceutically acceptable salt thereof and a pharmaceutically acceptable carrier or diluent.
- The preferred pharmaceutically acceptable salts are nontoxic acid addition salts formed by adding to the compounds of the present invention about a stoichiometric amount of a mineral acid, such as HCI, HBr, H2S04 or H3PO4, or of an organic acid, such as acetic, propionic, valeric, oleic, palmitic, stearic, lauric, benzoic, lactic, para-toluene sulfonic, methane sulfonic, citric, maleic, fumaric, succinic and the like.
- The pharmaceutical compositions of the present invention may be adapted for oral, parenteral or topical administration. They are formulated by combining the compound of this invention or an equivalent amount of a pharmaceutically acceptable salt thereof with any suitable, inert, pharmaceutically acceptable carrier or diluent. The preferred mode of administration is topical.
- Examples of suitable compositions include solid or liquid compositions for oral administration such as tablets, capsules, pills, powders, granules, solutions, dragees, suspensions or emulsions. They may also be manufactured in the form of sterile solid compositions which can be dissolved in sterile water, physiological saline or some other sterile injectable medium immediately before use.
- Topical dosage forms may be prepared according to procedures well known in the art, and may contain a variety of ingredients, excipients and additives. The formulations for topical use include ointments, creams, lotions, powders, aerosols, pessaries and sprays. Of these, ointments, lotions and creams may contain water, oils, fats, waxes, polyesters, alcohols, or polyols, plus such other ingredients as fragrances, emulsifiers and preservatives. Powders are made by mixing the active ingredient with a readily available, inert, pulverous distributing agent, such as talcum, calcium carbonate, tricalcium phosphate, or boric acid. Aqueous suspensions of the above powders may also be made. Solutions or emulsions may also be prepared using inert solvents which are preferably nonflammable, odorless, colorless and nontoxic, for example, vegetable oils and isopropanol. Similarly, aerosol or non-aerosol sprays may be prepared using solutions or suspensions in appropriate solvents, e.g., difluorodichloromethane for aerosols.
- Parenteral forms to be injected intravenously, intramuscularly, or subcutanteously are usually in the form of a sterile solution, and may contain salts or glucose to make the solution isotonic.
- Based on the greater in vivo topical potency for the compounds of this invention compared to miconazole, the dosages of the compounds of the present invention employed to combat a given fungal infection in animals, e.g., mammals, including humans, is generally somewhat less than the dosage requirements of present commercial products such as micoazole.
- It will be appreciated that the actual preferred dosages of the compounds of the present invention or pharmaceutically acceptable salts thereof will vary according to the particular composition formulated, the mode of application and the particular situs, host and disease being treated. Many factors that modify the action of the drug will be taken into account by the attending clinician, e.g., age, body weight, sex, diet, time of administration, rate of excretion, condition of the host, drug combinations, reaction sensitivities and severity of the disease. Administration can be carried out continuously or periodically within the maximum tolerated dose. Optimal application rates for a given set of conditions can be readily ascertained by the attending clinician using conventional dosage determination tests.
- In general, the topical dosage for humans ranges from about 50 mg per day to about 800 mg per day, in single or divided doses, with about 100 mg to about 200 mg per day being preferred. Each dose should be mixed with about 10 g of carrier.
- In general, the oral dosage for human ranges from about 50 mg to about 800 mg per day, in single or divided doses, with about 100 mg to about 400 mg per day being preferred.
- In general, the parenteral dosage for humans ranges for about 5 mg per day to about 50 mg per day, in single or divided doses, with about 10 mg per day being preferred.
- The following Examples illustrate the invention.
- Dissolve 7-chlorothiochroman-4-one (10 g, 50.3 mmole) in chloroform (100 mL) and cool the solution to 0-5°C. Add bromine (2.60 mL, 50.3 mmole) dropwise over a 10-minute period. Stir the reaction mixture at room temperature for one hour, then add chloroform (100 mL) and extract with 10% aqueous sodium sulfite (100 mL) followed by water (200 mL). Dry the chloroform solution over anhydrous magnesium sulfate, filter and evaporate in vacuo to a residue. Recrystallize the residue from cyclohexane to give 3-bromo-7-chlorothiochroman-4-one, mp 109-110°C.
- Suspend 3-bromo-7-chlorothiochroman-4-one (59.6 g, 215 mmole) in methanol (500 mL), cool to 05°C, and with stirring add sodium borohydride (8.18 g, 215 mmole) in three portions. After stirring the reaction mixture at room temperature for three hours, pour it into ice water (4 liters) and extract with chloroform (2 liters). Dry the chloroform solution over anhydrous magnesium sulfate, filter and evaporate in vacuo to a residue. Triturate the residue with chloroform/hexane to give 3-bromo-7-chlorothiochroman-4-ol, mp 141-142°C.
- Add 3-bromo-7-chlorothiochroman-4-ol (5.27 g, 18.8 mmole) and imidzole (12.8 g, 188 mmole) to acetonitrile (100 mL), and reflux for 4 hours. Pour the reaction mixture into water (500 mL), and extract with chloroform (500 mL). Wash the organic layer with water (500 mL), dry it over anhydrous magnesium sulfate, filter and evaporated in vacuo. Triturate the residue with anhydrous ether, filter and recrystallize from acetonitrile to give (±)-cis-6-chloro-2,3-dihydro-3-hydroxy-2-(1H-1-imidazolylmethyl)benzo-[b]thiophene, m.p. 164-165°C.
-
- a) thiochroman-4-one,
- b) 6-chlorothiochroman-4-one,
- c) 8-chlorothiochroman-4-one,
- d) 5,7-dichlorothiochroman-4-one,
- e) 6,7-dichlorothiochroman-4-one,
- f) 6,8-dichlorothiochroman-4-one,
- g) 7-trifluoromethylthiochroman-4-one,
- h) 6-trifluoromethylthiochroman-4-one,
- i) 6-fluorothiochroman-4-one,
- j) 7-fluorothiochroman-4-one,
- k) 8-fluorothiochroman-4-one,
- I) 6,7-difluorothiochroman-4-one,
- m) 6-methylthiochroman-4-one,
- n) 7-isopropylthiochroman-4-one,
- o) 6-cyanothiochroman-4-one,
- p) 7-cyanothiochroman-4-one,
- q) 6-nitrothiochroman-4-one,
- r) 7-nitrothiochroman-4-one,
- s) 6-nitro-7-trifluoromethylthiochroman-4-one,
- t) 6-(2,6-difluorophenyl)-thiochroman-4-one,
- u) 7-(2,6-difluorophenyl)-thiochroman-4-one,
- v) 6-(2-chloro,6-fluorophenyl)-thiochroman-4-one,
- w) 7-(2,4,6-trifluorophenyl)-thiochroman-4-one,
- a) (±)-cis-2,3-dihydro-3-hydroxy-2-(1H-1-imidazolylmethyl)benzo[b]thiophene,
- b) (±)-cis-5-chloro-2,3-dihydro-3-hydroxy-2-(1H-1-(1H-1-imidazolylmethyl)benzo[b]thiophene,
- c) (±)-cis-7-chloro-2,3-dihydro-3-hydroxy-2-(1H-1-imidazolylmethyl)benzo[b]thiophene,
- d) (±)-cis-4,6-dichloro-2,3-dihydro-3-hydroxy-2-(1H-1-imidazolylmethyl)benzo[b]thiophene,
- e) (±)-cis-5,6-dichloro-2,3-dihydro-3-hydroxy-2-(1H-1-imidazolylmethyl)benzo[b]thiophene,
- f) (±)-cis-5,7-dichloro-2,3-dihydro-3-hydroxy-2-(1H-1-imidazolylmethyl)benzo[b]thiophene,
- g) (±)-cis-2,3-dihydro-3-hydroxy-2-(1H-1-imidazolylmethyl)-6-trifluoromethylbenzo[b]thiophene,
- h) (±)-cis-2,3-dihydro-3-hydroxy-2-(1H-1-imidazolylmethyl)-5-trifluoromethylbenzo[b]thiophene,
- i) (±)-cis-2,3-dihydro-5-fluoro-3-hydroxy-2-(1H-1-imidazolylmethyl)benzo[b]thiophene,
- j) (±)-cis-2,3-dihydro-6-fluoro-3-hydroxy-2-(1H-1-imidazolylmethyl)benzo[b)thiophene,
- k) (±)-cis-2,3-dihydro-7-fluoro-3-hydroxy-2-(1H-1-imidazolylmethyl)benzo[b]thiophene,
- I) (±)-cis-5,6-difluoro-2,3-dihydro-3-hydroxy-2-(1H-1-imidazolylmethyl)benzo[b]thiophene,
- m) (±)-cis-2,3-dihydro-3-hydroxy-2-(1H-1-imidazolylmethyl)-5-methylbenzo[b] thiophene,
- n) (±)-cis-2,3-dihydro-3-hydroxy-2-(1H-1-imidazolylmethyl)-6-isopropylbenzo[b]thiophene,
- o) (±)-cis-5-cyano-2,3-dihydro-3-hydroxy-2-(1H-1-imidazolylmethyl)benzo[b]thiophene,
- p) (±)-cis-6-cyano-2,3-dihydro-3-hydroxy-2-(1H-1-imidazolylmethyl)benzo[b]thiophene,
- q) (±)-cis-2,3-dihydro-3-hydroxy-2-(1H-1-imidazolylmethyl)-5-nitrobenzo[b]thiophene,
- r) (±)-cis-2,3-dihydro-3-hydroxy-2-(1H-1-imidazolylmethyl)-6-nitrobenzo[b]thiophene,
- s) (±)-cis-2,3-dihydro-3-hydroxy-2-(1H-1-imidazolylmethyl)-5-nitro-6-trifluoromethylbenzo[b]-thiophene,
- t) (±)-cis-5-(2,6-difluorophenyl)-2,3-dihydro-3-hydroxy-2-(1H-1-imidazolylmethyl)benzo[b]-thiophene,
- u) (±)-cis-6-(2,6-difluorophenyl)-2,3-dihydro-3-hydroxy-2-(1H-1-imidazolylmethyl)benzo[b] thiophene,
- v) (±)-cis-5-(2-chloro-6-fluorophenyl)-2,3-dihydro-3-hydroxy-2-(1H-1-imidazolylmethyl)benzo[b]-thiophene,
- w) (±)-cis-2,3-dihydro-3-hydroxy-2-(1H-1-imidazolylmethyl)-6-(2,4,6-trifluorophenyl)-benzo[b]-thiophene,
- Stir a mixture of 3.0 g (0.01 moles) of (±)-cis-6-chloro-2,3-dihydro-3-hydroxy-2-(1H-1-imidazolylmethyl)benzo[b]thiophene, 3.44 g (0.045 mole) of allyl chloride, 25 mL of 50 wt% sodium hydroxide and 3 drops of tricaprylylmethylammonium chloride in 50 mL of THF overnight. Dilute the reaction mixture with 300 mL of methylene chloride. Separate the organic layer and wash it with water, and dry over anhydrous magnesium sulfate. Evaporate the organic solvents to give 3.2 g of the title compound, m/e of the molecular ion (hereinafter "m/e") 306.
- Dissolve 21.7 g (70.7 mmole) of (±)-cis-3-allyloxy-6-chloro-2,3-dihydro-2-(1H-1-imidazolylmethyl)benzo[b]thiophene in 500 mL of CH2CI2 and cool the solution to 0-5°. Added 14.6 g (70.7 mmole) of m-chloroperoxybenzoic acid (tech. 80-85%) and stir the reaction mixture at 0-5° for 1 hour. Extract the reaction mixture with 500 mL of 5% aqueous NaHCO3 followed by 500 mL of H20. Dried the CH2CI2 layer over anhydrous MgSO4 and concentrate it to give (±)-cis-3-allyloxy-6-chloro-2,3-dihydro-2-(1H-1-imidazolylmethyl)benzo[b]thiophene-1-oxide, a gum, m/e 322.
- Fractionally crystallize the mixture of (±)-cis and (±)-trans isomers from acetonitrile to give the (±)-cis isomer of the title compound (mp 168°C; m/e 306) and a mother liquor which provides the (±)-trans isomer of the title compound (mp 13D-132°C, m/e 306).
- Dissolve 2.665 g (10 mmole) of the compound of Preparation 1 (A)(3) in 50 mL of THF and 25 mL of 50% aqueous NaOH. To the solution so formed, add, with stirring, 2.315 g (10 mmole) of a-bromomethyl-4-chlorostyrene and 3-5 drops of tricaprylylmethyl ammonium chloride. Continue to stir the reaction mixture overnight at room temperature and add 500 mL of CHCl3. Wash the organic layer with water until the pH of the aqueous layer is about 7. Dry the organic layer over anhydrous magnesium sulfate, filter and evaporate in vacuo to a residue. Chromatograph the residue on a silica gel column, eluting with CHCl3 to give the title compound, m/e 417.
- Add 1.61 g (9.32 mmole) of m-chloroperbenzoic acid to a solution of 2.50 g (5.7 mmole) of the compound of Example 3A in 100 mL of CHZCl2. Stir the reaction mixture at room temperature for 2 hours. Wash the reaction mixture with 5% aqueous sodium bicarbonate and water. Dry the organic layer over anhydrous magnesium sulfate, filter and evaporate in vacuo to give the title compound as a gum, m/e 433.
- Reflux a solution of 2.50 g (5.7 mmole) of the compound of Example 3B and 2.42 g (11.5 mmole) of trifluoroacetic anhydride in 100 mL of toluene for hour. Evaporate the solution to dryness and dissolve the residue with methylene chloride. Wash the solution so formed with water. Dry the solution over anhydrous magnesium sulfate, filter and evaporate to give a gum. Chromatograph the gum on a silica gel column, eluting with CHCl3 to give the title compound as an oil, m/e 416.
- Dissolve 0.700 g (1.69 mmole) of the compound of Example 3C in 25 mL of methanol and cool the solution so formed to 0°C. Add, with stirring, 0.064 g (1.69 mmole) of NaBH4. Continue stirring at 0°C for 2 hours. Evaporate the methanol and dissolve the residue in 500 mL of CHCl3. Wash the CHCI3 solution over anhydrous magnesium sulfate, filter and evaporate to give a residue. Purify the residue by flash chromatography on silica gel (TLC grade), eluting with ethyl acetate to give a solid. Crystallize the solid from ethyl acetate to give the title compound (a mixture of cis and trans isomers) as an off-white solid, mp 165--168°C, m/e 418.
- Dissolve 1.0 g (3.20 mmole) of the (±)-cis-isomer of Example 2 in 10 mL of dimethylformamide (DMF). Add 0.313 g (6.52 mmole) of 50% dispersion of sodium hydride in mineral oil and stir the reaction mixture for 30 minutes at room temperature. Add 0.83 mL (6.52 mmole) of 2-chloro-6-fluorobenzyl chloride (Aldrich Chemical Co.) and stir the reaction mixture for 1 hour at room temperature. Pour the mixture into 500 mL of diethyl ether and 500 mL of water. Stir for 5 minutes. Separate the layers and wash the organic layer with water. Dry the organic layer over anhydrous magnesium sulfate and evaporate in vacuo to give a gum. Chromatograph the gum on silica gel, eluting with CHCl3 to give the title compound as a gum, m/e 448.
- Follow the procedure of Example 3A but substitute equivalent quantities of (±)-cis-2,3-dihydro-6-fluoro-3-hydroxy-2-(1H-1-imidazolylmethyl)benzo[b]thiophene (Preparation 1(B)(j) and allyl bromide for the compound of Preparation 1(A)(3) and a-bromomethyl-4-chlorostyrene, respectively, to give the title compound of this Example, Part A.
- Follow the procedure of Example 3B but substitute an equivalent quantity of the title compound of Example 5A for (±)-cis-6-chloro-2,3-dihydro-2-(1H-1-imidazolylmethyl)-3-[2-(4-chlorophenyl)-2-propenyloxy]-benzo[b]thiophene to give the title compound of this Example, Part B.
- Follow the procedure of Example 3C but substitute an equivalent quantity of the compound of Example 5B for the title compound of Examle 3C to give the corresponding benzo[b]thiophene-3-one which is thereafter treated in accordance with the procedure of Example 3D to provide the mixture of (±)-cis and (±)-trans-isomers of the title compound of this Example. Fractionally crystallize the mixture to obtain the title compounds of this Example, Part C.
- Dissolve 2.0 g (6.89 mmoles) of the (±)-cis isomer of the title compound of Example 5C in 50 mL of DMF. Add, with stirring, 0.662 g (13.8 mmoles) of sodium hydride (a 50% dispersion in mineral oil) and continue stirring the slurry so formed at room temperature for 30 minutes. Add 1.31 mL (10.3 mmoles) of 2-chloro-6-fluorobenzyl chloride and continue stirring at room temperature for 1 hour. Pour the reaction mixture into a mixture of 1 liter of ethyl ether and 1 liter of water. Stir the mixture so formed for 10 minutes. Separate the layers and wash the organic layer with 1 liter of water. Dry the organic layer over anhydrous magnesium sulfate, filter and evaporate in vacuo to give a residue. Chromatograph the residue on silica gel, eluting with CHCI3 to give the title compound as a gum, m/e 432.
- Heat a solution of 2 g (6.5 mmoles) of the (±)-cis isomer of Example 2 and 3.8 g (32.6 mmoles) of thionylchloride in 50 mL of benzene at reflux for 2 hours. Evaporate the reaction mixture to dryness to give the title compound as a gum, m/e 324.
- Dissolve 1.0 g (3.25 mmoles) of the (±)-cis isomer of Example 2 in 50 mL of tetrahydrofuran and cool the solution to 0°-5°C. Add, with stirring, 1.06 g (6.50 mmoles) of diethylaminosulfur trifluoride ("DAST", obtained from Aldrich Chemical Co.) and continue to stir the reaction mixture for 1 hour. Evaporate the reaction mixture to give a residue. Triturate the residue with methylene chloride and wash the organic solution so formed with water. Dry the organic layer over anhydrous magnesium sulfate, filter and evaporate to give the title compound, m/e 308.
- Heat a solution of 2.0 g (6.1 mmoles) of the title compound of Example 6 and 0.9 g (13.2 mmoles) of imidazole in 50 mL of DMF at 100°C overnight. Evaporate the solution to a residue. Triturate the residue with CHCI3 and wash the CHCI3 solution with water. Dry the organic layer over anhydrous magnesium sulfate, filter and evaporate to give a residue. Chromatograph the residue on silica gel, eluting with 1:99 (v/ v) CH3OH:CHCl3 to give the cis and trans title compounds as gums, m/e 357 for each isomer.
- Heat a reaction mixture of 1.4 g (4.3 mmoles) of the title compound of Example 6 and 2.5 g (8.5 mmoles) of tri(n-butyl)tin hydride in 20 mL of toluene at reflux overnight. Evaporate to give a residue. Chromatograph the residue on silica gel, eluting with 1:1 (v/v) ethyl acetate:hexane to give the title compound as an oil, m/e 290.
- Dissolve 1.0 g (3.2 mmoles) of the (±)-cis isomer of the title compound of Example 2 in 25 mL of DMF. Add, with stirring, 0.086 g (3.58 mmoles) of sodium hydride, (as a 50% dispersion in mineral oil) and continue stirring at room temperature for 1 hour. Add to the stirred solution 0.6 g (4.85 mmoles) of dimethylthiocarbamoyl chloride (Aldrich Chemical Co.) and continue to stir the reaction mixture overnight. Evaporate the reaction mixture to a residue. Triturate the residue with methylene chloride. Wash the organic layer with water. Dry the organic layer over anhydrous magnesium sulfate, filter and evaporate to give an oil. Chromatograph the oil on silica gel, eluting with 1:99 (v/v) CH3OH:CHCl3 to give the title compound as a gum, m/e 394.
- Follow the procedure of Example 3A but substitute quantities of each of the (±)-cis-2,3-dihydro-3-hydroxy-2-(1H-1-imidazolylmethyl)benzo[b]thiophene compounds a)-v) obtained in Preparation 1B for the compound of Preparation 1(A)(3) and allyl chloride for a-bromomethyl-4-chlorostyrene. There are obtained the corresponding 3-allyloxy derivatives of each of the foregoing, which upon reaction with m-chloroperbenzoic acid according to the procedure of Example 3B provide the corresponding benzo[b]thiophene-1-oxide derivatives which are treated with trifluoroacetic anhydride and thereafter with NaBH4 in accordance with the procedures of Examples 3C and 3D to provide mixtures of (±)-cis and (±)-trans isomers which are separated into the (±)-cis isomers,
- a) (±)-cis-2-Allyl-2,3-dihydro-3-hydroxy-2-(1H-1-imidazolylmethyl)benzo[b]thiophene,
- b) (±)-cis-2-Allyl-5-chloro-2,3-dihydro-3-hydroxy-2-(1H-1-imidazolylmethyl)benzo[b]thiophene,
- c) (±)-cis-2-Allyl-7-chloro-2,3-dihydro-3-hydroxy-2-(1H-1-imidazolylmethyl)benzo[b]thiophene,
- d) (±)-cis-2-Allyl-4,6-dichloro-2,3-dihydro-3-hydroxy-2-(1H-1-imidazolylmethyl)benzo[b]thiophene,
- e) (±)-cis-2-Allyl-5,6-dichloro-2,3-dihydro-3-hydroxy-2-(1H-1-imidazolylmethyl)benzo[b]thiophene,
- f) (±)-cis-2-Allyl-5,7-dichloro-2,3-dihydro-3-hydroxy-2-(1H-1-imidazolylmethyl)benzo[b]thiophene,
- g) (±)-cis-2-Allyl-2,3-dihydro-3-hydroxy-2-(1H-1-imidazolylmethyl)-6-trifluoromethylbenzo[b]-thiophene,
- h) (±)-cis-2-Allyl-2,3-dihydro-3-hydroxy-2-(1H-1-imidzolylmethyl)-5-trifluoromethylbenzo[b]thiophene,
- i) (±)-cis-2-Allyl-2,3-dihdyro-5-fluoro-3-hydroxy-2-(1H-1-imidazolylmethyl)benzo[b]thiophene,
- j) (±)-cis-2-Allyl-2,3-dihydro-6-fluoro-3-hydroxy-2-(1H-1-imidazolylmethyl)benzo[b]thiophene,
- k) (±)-cis-2-Allyl-2,3-dihydro-7-fluoro-3-hydroxy-2-(1H-1-imidazolylmethyl)benzo[b]thiophene,
- I) (±)-cis-2-Allyl-5,6-difluoro-2,3-dihydro-3-hydroxy-2-(1H-1-imidazolylmethyl)benzo[b]thiophene,
- m) (±)-cis-2-Allyl-2,3-dihydro-3-hydroxy-2-(1H-1-imidazolylmethyl)-5-methylbenzo[b]thiophene,
- n) (±)-cis-2-Allyl-2,3-dihydro-3-hydroxy-2-(1H-1-imidazolylmethyl)-6-isopropylbenzo[b]thiophene,
- o) (±)-cis-2-Allyl-5-cyano-2,3-dihydro-3-hydroxy-2-(1H-1-imidazolylmethyl)benzo[b]thiophene,
- p) (±)-cis-2-Allyl-6-cyano-2,3-dihydro-3-hydroxy-2-(1H-1-imidazolylmethyl)benzo[b]thiophene,
- q) (±)-cis-2-Allyl-2,3-dihydro-3-hydroxy-2-(1H-1-imidazolylmethyl)-5-nitrobenzo[b]thiophene,
- r) (±)-cis-2-Allyl-2,3-dihydro-3-hydroxy-2-(1H-1-imidazolylmethyl)-6-nitrobenzo[b]thiophene,
- s) (±)-cis-2-Allyl-2,3-dihydro-3-hydroxy-2-(1H-1-imidazolylmethyl)-5-nitro-6-trifluoromethyl- benzo[b]thiophene,
- t) (±)-cis-2-Allyl-5-(2,6-difluorophenyl)-2,3-dihydro-3-hydroxy-2-(1H-1-imidazolylmethyl)-benzo[b]thiophene,
- u) (±)-cis-2-Allyl-6-(2,6-difluorophenyl)-2,3-dihydro-3-hydroxy-2-(1H-1-imidazolylmethyl)-benzo[b]thiophene,
- v) (±)-cis-2-Allyl-5-(2-chloro-6-fluorophenyl)-2,3-dihydro-3-hydroxy-2-(1H-1-imidazolylmethyl)-benzo[b]thiophene,
- w) (±)-cis-2-Allyl-2,3-dihydro-3-hydroxy-2-(1H-1-imidazolylmethyl)-6-(2,4,6-trifluorophenyl)-benzo[b]thiophene,
- Follow the procedure of Example 3A but substitute equivalent quantities of (±)-cis-2,3-dihydro-5-fluoro-3-hydroxy-2-(1H-1,2,4-triazol-1-ylmethyl)benzo[b]thiophene for the compound of Preparation 1 (A)(3) and of allyl chloride for a-bromoethyl-4-chlorostyrene. There is obtained the corresponding 3-allyloxy derivative which upon reaction with m-chloroperbenzoic acid according to the procedure of Example 3B provides the corresponding benzo[b]thiophene-1-oxide derivate which is treated with trifluoroacetic anhydride and thereafter with NaBH4 in accordance with the procedures of Example 3C and 3D to provide a mixture of the (±)-cis and (±)-trans-isomers which is separated by fractional crystallization into the title compounds, mp 149-150°C and 157-159°C, respectively, both white solids.
- Stir a mixture of 1 g (3.2 mmoles) of the title compounds of Example 2, 1.36 g (6.49 mmoles) of 2,4-dichlorobenzoyl chloride and 0.66 g (6.52 mmoles) of trimethylamine in 50 mL of methylene chloride at room temperature for 4 hours. Dilute the reaction mixture with water. Separate the organic phase and wash the organic phase with 5 wt% sodium bicarbonate and then with water. Dry over anhydrous magnesium sulfate and evaporate the organic solvent to give an oil. Chromatograph the oil on a silica gel column, eluting with chloroform to give the (±)-cis and (±)-trans title compound, each having m/e 479.
- Add 0.086 g (3.43 mmoles) of sodium hydride 50% oil dispersion to a solution of 1 g (3.2 mmoles) of the title compounds of Example 2 in 20 mL of DMSO. Stir the resulting slurry for 1 hour and add thereto 1.63 g (6.5 mmoles) of 1-(4-chlorophenoxy)-2-mesylethane. Heat the reaction mixture overnight. Extract the reaction mixture with methylene chloride. Wash the organic layer with water and dry it over anhydrous MgS04. Evaporate the solvent to give a gum. Chromatograph the gum on a silica gel column, eluting the chloroform to give 278 mg of the title compounds as an oil, m/e 461.
- Add 0.96 g (20 mmoles) of sodium hydride (50% oil dispersion) to a solution of 1.0 g (3.26 mmoles) of the title compounds of Example 2 in 10 ml of DMF. Stir for 30 minutes at RT. Add dropwise a solution of 2.43 g (10 mmoles) of 1-bromomethyl-1,2,4-triazole hydrogen bromide in 10 mL of DMF. Stir the reaction mixture overnight at RT. Pour the reaction mixture into 500 mL of CHCI3 and 500 mL of brine and stir for 10 minutes. Separate and dry the organic layer over anhydrous MgSO4. Remove the organic solvent in vacuo to give an oily residue. Chromatograph the oily residue on silica gel, eluting with 1:99 (v/v) MeOH:CHCl3 containing 1% (v/v) of conc. NH4OH to give 0.75 g of the title compounds, m/e 387.
- Add 0.32 mL (3.66 mmoles) of allyl bromide and two drops of tricaprylylmethylammonium chloride to a solution of 0.75 g (2.44 mmoles) of the title compounds of Example 2 and 10 mL of 50% of NaOH in 30 mL of THF. Stir the reaction mixture at RT for 3 hours. Pour the reaction mixture into 250 mL of CHCI3 and 250 mL of brine and stir for 5 minutes. Separate and dry the organic layer over anhydrous MgS04. Remove the organic solvent in vacuo to give an oily residue. Chromatograph the oily residue on a silica gel column, eluting with CHCI3 to give 0.83 g of the title compounds, m/e 346.
- Add 0.96 g 20 mmoles) of sodium hydride (50% oil dispersion) to a solution of 0.895 g (3.07 mmoles) of the title compounds of Example 12 in 10 mL of DMF and stir for 30 minutes at RT. Add dropwise thereto a solution of 2.43 g (1.0 mmoles) of 1-bromomethyl-1 H-1,2,4-triazole hydrogen bromide in 10 mL of DMF. Stir the reaction mixture for 1 hour at RT. Pour the reaction mixture into 500 mL of CHCI3 and 500 mL of brine. Separate and dry the organic layer over anhydrous MgSO4. Remove the organic solvents in vacuo to give an oily residue. Chromatograph the oily residue on a silica gel column, eluting with 1:99 (v/v) MeOH:CHCl3 containing 1 volume % of concentrated NH4OH to give 1.04 g of the title compounds, m/e 372.
- Follow the procedure of Example 12, which is similar to that of Example 3, but omit the step that is similar to Step D, Example 3 to yield the title compound of the Example, Part A.
- Add 12.0 mL (18.6 mmoles) of n-butyl lithium (1.55 M in hexane) to a solution of 2.0 mL (16.2 mmoles) of 3,3-dimehtyl-1-butyne in 50 mL of dry THF at a temperatrure of 0°-5°C. Stir for 30 minutes at 0°-5°C. Add thereto 2.0 g (6.92 mmoles) of the title compound of Part A. Stir the reaction mixture so formed overnight at RT. Pour the reaction mixture into one liter of CHCI3. Wash the organic layer with one liter of brine. Separate and dry the organic layer over anhydrous MgS04. Remove the organic solvents in vacuo to give a gum. Chromatograph the gum on a silica gel column, eluting with CHCI3 to give 0.429 g of the less polar title compounds, as a solid, mp 166°-167°C, m/e 369, and 0.541 g of the more polar title compound as a gum, m/e 369.
- Follow the procedure of Exmaple 11j, which is similar to that of Example 3, but omit the step that is similar to step D of Example 3 to yield the title compound of this Example, part A.
- Add 4.1 mL (11.6 mmoles) of 2.8M CH3MgBr to a suspension of 1.67 g (5.79 mmoles) of the compound of Part A in 25 mL of THF. Stir the reaction mixture for 1 hour at RT and then heat at reflux for 2 hours. Add 25 mL of 10% NHQCl and stir for 10 minutes. Pour into 500 mL of CHCI3 and 500 mL of brine. Remove the solvent in vacuo to give an oily residue. Chromatograph the oily residue on a silica gel column, eluting with 1:99 (v/v) MeOH:CHCl3 containing 1 vol% of conc. NH40H to give 0.281 g of the less polar title compound, m/e 304 and 0.303 g of the more polar isomer, m/e 304.
- The following are typical pharmaceutical formulations containing as the active ingredient (designated "Drug") the compound of this invention such as (±)-cis-2-allyl-6-chloro-2,3-dihydro-3-hydroxy-2-(1H-1-imidazolylmethyl)benzo[b]thiophene or (±)-cis-2-allyl-6-chloro-3-(2-chloro-6-fluorobenzyloxy)-2,3-dihydro-2-(1H-1-imidzolylmethyl)benzo[b]thiophene. It will be appreciated, however, that either of these compounds may be replaced by equally effective quantities of other compounds of this invention.
-
- Heat the polyethylene glycol 6000 to 70-80°C. Mix the drug, sodium lauryl sulfate, corn starch, and lactose into the liquid and allow the mixture to cool. Pass the solidified mixture through a mill. Blend granules with magnesium stearate and compress into tablets.
-
- Mix the first four ingredients in a suitable mixer for 10-15 minutes. Add the magnesium stearate and mix for 1-3 minutes. Fill the mixture into suitable two-piece hard gelatin capsules using an encapsulating machine.
to obtain, respectively, upon purification and separation into their respective cis and trans isomers,
and the trans isomers thereof.
and the trans isomers thereof.
Claims (13)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| AT86110821T ATE53390T1 (en) | 1985-08-12 | 1986-08-05 | DIHYDROBENZO(B>THIOPHENE AND PHARMACEUTICAL AGENT TO BE USED AS AN ANTIFUNCTION. |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US764289 | 1985-08-12 | ||
| US06/764,289 US4731364A (en) | 1985-08-12 | 1985-08-12 | Dihydrobenzo(B)thiophenes and pharmaceutical compositions thereof useful as antifungals |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP0212428A1 EP0212428A1 (en) | 1987-03-04 |
| EP0212428B1 true EP0212428B1 (en) | 1990-06-06 |
Family
ID=25070267
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP86110821A Expired - Lifetime EP0212428B1 (en) | 1985-08-12 | 1986-08-05 | Dihydrobenzo/b /thiophenes and pharmaceutical compositions thereof useful as antifungals |
Country Status (24)
| Country | Link |
|---|---|
| US (1) | US4731364A (en) |
| EP (1) | EP0212428B1 (en) |
| JP (1) | JPS6248683A (en) |
| KR (1) | KR900000673B1 (en) |
| CN (1) | CN1022105C (en) |
| AR (1) | AR244224A1 (en) |
| AT (1) | ATE53390T1 (en) |
| AU (1) | AU585381B2 (en) |
| CA (1) | CA1287634C (en) |
| DE (1) | DE3671748D1 (en) |
| DK (1) | DK370886A (en) |
| ES (1) | ES2001866A6 (en) |
| FI (1) | FI95251C (en) |
| GR (1) | GR862058B (en) |
| HU (1) | HU209302B (en) |
| IE (1) | IE59028B1 (en) |
| IL (1) | IL79598A0 (en) |
| MY (1) | MY101809A (en) |
| NO (1) | NO171066C (en) |
| NZ (1) | NZ217081A (en) |
| OA (1) | OA08374A (en) |
| PH (1) | PH23405A (en) |
| PT (1) | PT83151B (en) |
| ZA (1) | ZA865841B (en) |
Families Citing this family (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4695579A (en) * | 1984-12-21 | 1987-09-22 | Schering Corporation | Antifungal 2-azolylmethyl-3-difluorobenzyloxy-2,3-dihydrofluoro-benzo(B) thiophens |
| US4943311A (en) * | 1988-05-09 | 1990-07-24 | Uniroyal Chemical Company, Inc. | Azole derivatives of spiroheterocycles |
| DE4010797A1 (en) * | 1990-04-04 | 1991-10-10 | Hoechst Ag | SUBSTITUTED AZOLES, METHOD FOR THE PRODUCTION THEREOF, MEANS CONTAINING THEM AND THE USE THEREOF |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4352808A (en) * | 1980-12-12 | 1982-10-05 | Schering Corporation | 3-Aralkyloxy-2,3-dihydro-2-(imidazolylmethyl)benzo(b)thiophenes and related derivatives, their use as antimicrobials and pharmaceutical formulations useful therefore |
| US4468404A (en) * | 1980-12-12 | 1984-08-28 | Schering Corporation | 3-Aralkyloxy-2,3-dihydro-2-(triazolymethyl)benzo(b)thiophenes |
| US4431816A (en) * | 1980-12-12 | 1984-02-14 | Schering Corporation | 2,3-Dihydro-2-(imidazolylmethyl)benzo(b)thiophenes |
| ZA825413B (en) * | 1981-08-26 | 1983-06-29 | Pfizer | Thromboxane synthetase inhibitors, processes for their production, and pharmaceutical compositions comprising them |
| US4695579A (en) * | 1984-12-21 | 1987-09-22 | Schering Corporation | Antifungal 2-azolylmethyl-3-difluorobenzyloxy-2,3-dihydrofluoro-benzo(B) thiophens |
-
1985
- 1985-08-12 US US06/764,289 patent/US4731364A/en not_active Expired - Fee Related
-
1986
- 1986-08-04 ES ES8600837A patent/ES2001866A6/en not_active Expired
- 1986-08-04 AR AR86304795A patent/AR244224A1/en active
- 1986-08-04 IL IL79598A patent/IL79598A0/en not_active IP Right Cessation
- 1986-08-04 OA OA58915A patent/OA08374A/en unknown
- 1986-08-04 ZA ZA865841A patent/ZA865841B/en unknown
- 1986-08-04 AU AU60854/86A patent/AU585381B2/en not_active Ceased
- 1986-08-04 FI FI863168A patent/FI95251C/en not_active IP Right Cessation
- 1986-08-04 NO NO863140A patent/NO171066C/en unknown
- 1986-08-04 GR GR862058A patent/GR862058B/en unknown
- 1986-08-04 NZ NZ217081A patent/NZ217081A/en unknown
- 1986-08-04 PH PH34103A patent/PH23405A/en unknown
- 1986-08-04 DK DK370886A patent/DK370886A/en not_active Application Discontinuation
- 1986-08-05 DE DE8686110821T patent/DE3671748D1/en not_active Expired - Lifetime
- 1986-08-05 EP EP86110821A patent/EP0212428B1/en not_active Expired - Lifetime
- 1986-08-05 AT AT86110821T patent/ATE53390T1/en active
- 1986-08-06 PT PT83151A patent/PT83151B/en not_active IP Right Cessation
- 1986-08-06 JP JP61184999A patent/JPS6248683A/en active Pending
- 1986-08-06 KR KR1019860006486A patent/KR900000673B1/en not_active Expired
- 1986-08-06 CN CN86105964A patent/CN1022105C/en not_active Expired - Fee Related
- 1986-08-06 IE IE210486A patent/IE59028B1/en not_active IP Right Cessation
- 1986-08-07 HU HU863445A patent/HU209302B/en not_active IP Right Cessation
- 1986-08-07 CA CA000515527A patent/CA1287634C/en not_active Expired - Lifetime
-
1987
- 1987-08-10 MY MYPI87001252A patent/MY101809A/en unknown
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US4898954A (en) | Process for the preparation of oxiranes | |
| US4267179A (en) | Heterocyclic derivatives of (4-phenylpiperazin-1-yl-aryloxymethyl-1,3-dioxolan-2-yl)methyl-1H-imidazoles and 1H-1,2,4-triazoles | |
| CA1096873A (en) | Tertiary imidazolyl alcohols, a process for their production and their use as medicaments | |
| US5405861A (en) | Triazole compounds and antifungal compositions thereof | |
| EP0131302A2 (en) | Imidazolium salts, intermediates thereto and their use | |
| EP0212428B1 (en) | Dihydrobenzo/b /thiophenes and pharmaceutical compositions thereof useful as antifungals | |
| US4539325A (en) | 1-(2-Aryl-2-halo-1-ethenyl)-1H-azoles, and anticonvulsant use thereof | |
| EP0151477B1 (en) | 1h-imidazole derivatives, a process for preparing them and pharmaceutical compositions containing them | |
| US4465680A (en) | Combating fungi with azolylvinyldithioacetals | |
| US4737508A (en) | 1-aryl-1-(1H-azol-1-ylalkyl)-1,3-dihydroisobenzofurans, related derivatives and pharmaceutical compositions thereof useful as antifungals | |
| EP0185381B1 (en) | 2-azolylmethyl-3-difluorobenzyloxy-2,3-dihydrofluorobenzo(b)thiophenes, process for their preparation and compositions containing them | |
| US4640918A (en) | Substituted 2-phenyl-1,3-dioxolanes | |
| NO830655L (en) | DIAZOLYL ALKANOLS, THE PROCEDURE FOR THEIR PREPARATION AND THEIR USE AS AN ANTIMYCOTIC AGENT | |
| US4607045A (en) | Azolylmethylcycloacetals, their preparation and their use as drugs | |
| EP1044193B1 (en) | Process for preparing triazole antimycotic compounds | |
| FI65429C (en) | PROCEDURE FOR FRAMSTATION OF THERAPEUTIC ANVAENDBARA HETEROCYCLIC DERIVATIVES AV (4-PHENYL-PIPERAZIN-1-YL-ARYLOXIMETYL-1,3-DIOXOLAN-2-YL) -METHYL-1H-IMZERZERO-1H-IMIDAZER | |
| PL117988B1 (en) | Process for preparing novel heterocyclic derivatives of /4-phenylpiperazinyl-1-aryloxymethyl-1,3-dioxolanyl-2-/-methyl-1h-imidazoles and 1h-1,2,4-triazolesykh/4-fenilopiperazimilo-1-arilokimetilo-1,3-dioksolanilo-2/-metilo-1kha-imidazola i 1kha-1,2,4-triazola | |
| CS215362B1 (en) | 1-substituted imidazoles and triazoles and method of making the same | |
| SI7911478A8 (en) | Process for obtaining new heterocyclic derivatives of azole |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): AT BE CH DE FR GB IT LI LU NL SE |
|
| 17P | Request for examination filed |
Effective date: 19870606 |
|
| 17Q | First examination report despatched |
Effective date: 19881006 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AT BE CH DE FR GB IT LI LU NL SE |
|
| REF | Corresponds to: |
Ref document number: 53390 Country of ref document: AT Date of ref document: 19900615 Kind code of ref document: T |
|
| REF | Corresponds to: |
Ref document number: 3671748 Country of ref document: DE Date of ref document: 19900712 |
|
| ET | Fr: translation filed | ||
| ITF | It: translation for a ep patent filed | ||
| PLBE | No opposition filed within time limit |
Free format text: ORIGINAL CODE: 0009261 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: NO OPPOSITION FILED WITHIN TIME LIMIT |
|
| 26N | No opposition filed | ||
| ITTA | It: last paid annual fee | ||
| EPTA | Lu: last paid annual fee | ||
| EAL | Se: european patent in force in sweden |
Ref document number: 86110821.5 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: LU Payment date: 19950701 Year of fee payment: 10 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 19950710 Year of fee payment: 10 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: AT Payment date: 19950714 Year of fee payment: 10 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: SE Payment date: 19950717 Year of fee payment: 10 Ref country code: BE Payment date: 19950717 Year of fee payment: 10 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: GB Payment date: 19950718 Year of fee payment: 10 Ref country code: CH Payment date: 19950718 Year of fee payment: 10 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: NL Payment date: 19950721 Year of fee payment: 10 Ref country code: DE Payment date: 19950721 Year of fee payment: 10 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LU Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 19960805 Ref country code: GB Effective date: 19960805 Ref country code: AT Effective date: 19960805 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: SE Effective date: 19960806 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LI Effective date: 19960831 Ref country code: CH Effective date: 19960831 Ref country code: BE Effective date: 19960831 |
|
| BERE | Be: lapsed |
Owner name: SCHERING CORP. Effective date: 19960831 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: NL Effective date: 19970301 |
|
| GBPC | Gb: european patent ceased through non-payment of renewal fee |
Effective date: 19960805 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: FR Effective date: 19970430 |
|
| NLV4 | Nl: lapsed or anulled due to non-payment of the annual fee |
Effective date: 19970301 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DE Effective date: 19970501 |
|
| EUG | Se: european patent has lapsed |
Ref document number: 86110821.5 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: ST |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: IT Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20050805 |