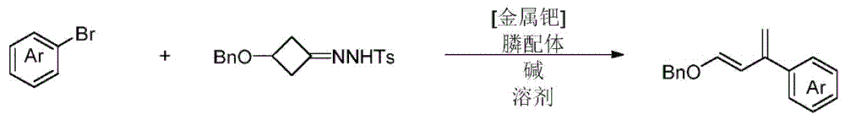Disclosure of Invention
Aiming at the problems in the prior art, the invention provides the electron-rich conjugated diene compound with better stereoselectivity, simple and convenient operation, the preparation method and the application.
The technical scheme adopted by the invention is as follows: an electron-rich conjugated diene compound has the following structure:
in the formula: ar is one of phenyl, 4-methylphenyl, 4-tert-butylphenyl, 4-methoxyphenyl, 4-phenyl, 4-fluorophenyl, 4-chlorobenzene, 4-trifluoromethylphenyl, 3-methoxyphenyl, 2-methylphenyl, 2-methoxyphenyl, 1, 2-methylenedioxyphenyl, 2-naphthyl and 3-thienyl.
The preparation method of the electron-rich conjugated diene compound comprises the following steps:
step 1: mixing N- (3- (benzyloxy) cyclobutyl) -4-methylbenzenesulfonylhydrazone, a base and a catalyst;
step 2: adding a phosphine ligand, aryl halide and a solvent, and fully reacting under magnetic stirring at 90 ℃ in a nitrogen atmosphere;
and step 3: cooling and purifying to obtain the required electron-rich conjugated diene compound.
The preparation method of the electron-rich conjugated diene compound comprises the following steps:
step 1: mixing 3- (benzyloxy) cyclobutane-1-ketone, p-toluenesulfonyl hydrazide and a solvent, and fully reacting under the conditions of magnetic stirring and 60 ℃;
step 2: after cooling, adding an alkali, metal palladium, a phosphine ligand, an aryl halide and a solvent into the reaction mixture obtained in the step 1; fully reacting under the condition of magnetic stirring at 90 ℃ in nitrogen atmosphere;
and step 3: cooling and purifying to obtain the required electron-rich conjugated diene compound.
Further, the alkali is cesium carbonate, the catalyst is metal palladium, the phosphine ligand is diphenyl cyclohexyl phosphine, and the solvent is 1,4 dioxane.
Further, the catalyst is one of palladium acetate, tris (dibenzylideneacetone) dipalladium, bis (acetonitrile) palladium (II) dichloride and allyl palladium (II) chloride dimer.
Further, the molar ratio of the aryl halide to the N- (3- (benzyloxy) cyclobutyl) -4-methyl benzenesulfonylhydrazone or the p-toluenesulfonylhydrazide is 1: 1-1.5.
Further, the molar ratio of the catalyst to the phosphine ligand is 1: 2-4, the molar ratio of the aromatic halide to the catalyst is 1:0.04, and the molar ratio of the aromatic halide to the base is 1: 2.5-3.
Further, the molar ratio of the 3- (benzyloxy) cyclobutane-1-one to the aryl halide is 6: 5.
An application of an electron-rich conjugated diene compound, wherein the electron-rich conjugated diene compound is used as a substrate for [4+2] cyclization and reacts with a dienophile to construct a six-membered ring compound.
The invention has the beneficial effects that:
(1) the preparation method has Z/E selectivity, and the obtained electron-rich conjugated diene is E type 1, 3-butadiene with a single structure;
(2) the electron-rich conjugated diene is synthesized in one step by the simple and easily-obtained bromobenzene and p-toluenesulfonylhydrazone derived from cyclobutanone, and the preparation method is simple and convenient to operate;
(3) the electron-rich conjugated diene obtained by the invention is a common substrate for [4+2] cyclization, and can construct a six-membered ring compound with a yield of more than 85%.
Detailed Description
The invention is further described with reference to the following figures and specific embodiments.
An electron-rich conjugated diene compound has the following structure:
in the formula: ar is one of phenyl, 4-methylphenyl, 4-tert-butylphenyl, 4-methoxyphenyl, 4-phenyl, 4-fluorophenyl, 4-chlorobenzene, 4-trifluoromethylphenyl, 3-methoxyphenyl, 2-methylphenyl, 2-methoxyphenyl, 1, 2-methylenedioxyphenyl, 2-naphthyl and 3-thienyl.
The preparation method of the electron-rich conjugated diene compound is shown in figure 1 and comprises the following steps:
step 1: mixing N- (3- (benzyloxy) cyclobutyl) -4-methylbenzenesulfonylhydrazone, a base and a catalyst;
step 2: adding a phosphine ligand, aryl halide and a solvent, and fully reacting under magnetic stirring at 90 ℃ in a nitrogen atmosphere;
and step 3: cooling and purifying to obtain the required electron-rich conjugated diene compound.
The preparation method of the electron-rich conjugated diene compound is shown in figure 2 and comprises the following steps:
step 1: mixing 3- (benzyloxy) cyclobutane-1-ketone, p-toluenesulfonyl hydrazide and a solvent, and fully reacting under the conditions of magnetic stirring and 60 ℃;
step 2: after cooling, adding an alkali, metal palladium, a phosphine ligand, an aryl halide and a solvent into the reaction mixture obtained in the step 1; fully reacting under the condition of magnetic stirring at 90 ℃ in nitrogen atmosphere;
and step 3: cooling and purifying to obtain the required electron-rich conjugated diene compound. Purification is carried out by using a silica gel column layer.
The alkali is cesium carbonate, the catalyst is metal palladium, the phosphine ligand is diphenyl cyclohexyl phosphine, and the solvent is 1,4 dioxane. The catalyst is one of palladium acetate, tris (dibenzylideneacetone) dipalladium, bis (acetonitrile) palladium dichloride (II) and allyl palladium chloride (II) dimer. The molar ratio of aryl halide to N- (3- (benzyloxy) cyclobutyl) -4-methylbenzenesulfonylhydrazone or p-toluenesulfonylhydrazide is 1:1 to 1.5. The molar ratio of the catalyst to the phosphine ligand is 1: 2-4, the molar ratio of the aromatic halide to the catalyst is 1:0.04, and the molar ratio of the aromatic halide to the base is 1: 2.5-3. The molar ratio of 3- (benzyloxy) cyclobutane-1-one to aryl halide is 6: 5.
The electron-rich conjugated diene compound is used as a substrate for cyclization of [4+2], and reacts with dienophile to construct a six-membered ring compound.
The aromatic halide is aryl bromide and is any compound in the following structure:
the electron-rich conjugated diene compound is any one of the following structures:
example 1
Electron rich 1,3 butadiene was prepared as follows:
step 1: to an 8mL vial equipped with a magnetic stir bar were added 0.5mmol cesium carbonate, 0.24mmol N- (3- (benzyloxy) cyclobutyl) -4-methylbenzenesulfonylhydrazone, 0.004mmol tris (dibenzylideneacetone) dipalladium, and the vial was sealed with a screw cap and transferred to a glove box.
Step 2: a vial was charged with 0.008mmol diphenylcyclohexylphosphine, 2.0mL 1, 4-dioxane, and 0.2mmol bromobenzene, capped with a screw cap and removed from the glove box. The mixture was stirred for 16 hours on a magnetic stirrer at 90 ℃.
And step 3: the reaction mixture was cooled to room temperature, filtered through celite, eluted with ethyl acetate, dried in vacuo, and transferred to a silica gel column, which was then eluted with an eluent (PE/EA 100:1) to obtain the desired product (Rf 0.5(PE EA 40:1)) as a colorless oil in 79% yield.
The structure is as follows:
the reaction process is as follows:
the NMR spectrum of the product is shown in FIG. 4:
1H NMR(400MHz,CDCl3)δ7.45-7.23(m,10H),6.55(d,J=12.6Hz,1H),5.90(d,J=12.6Hz,1H),5.09(d,J=1.7Hz,1H),4.91(d,J=1.8Hz,1H),4.80(s,2H)。
the nuclear magnetic resonance carbon spectrum of the product is shown in FIG. 5:
13C NMR(101MHz,CDCl3)150.4,145.4,140.8,136.7,128.5,128.1,128.0,128.0,127.5,127.4,111.9,108.8,71.9。
the high resolution mass spectrum of the product is shown in fig. 6:
the high resolution mass spectrum is as follows: HRMS (ESI, m/z): calcd for C17H17O[M+H]+237.1274,found 237.1274。
Example 2
Electron rich 1,3 butadiene was prepared as follows:
step 1: to an 8mL vial equipped with a magnetic stir bar were added 0.5mmol cesium carbonate, 0.24mmol N- (3- (benzyloxy) cyclobutyl) -4-methylbenzenesulfonylhydrazone, 0.004mmol tris (dibenzylideneacetone) dipalladium, and the vial was sealed with a screw cap and transferred to a glove box.
Step 2: a vial was charged with 0.008mmol diphenylcyclohexylphosphine, 2.0mL 1, 4-dioxane, and 0.2mmol aryl bromide (Ar is naphthyl), capped and removed from the glove box. The mixture was stirred for 16 hours on a magnetic stirrer at 90 ℃.
And step 3: the reaction mixture was cooled to room temperature, filtered through celite, eluted with ethyl acetate, dried in vacuo, and transferred to a silica gel column, which was then eluted with an eluent (PE/EA 100:1) to obtain the desired product (Rf 0.5(PE EA 40:1)) as a colorless oil with a yield of 60%.
The reaction process is as follows:
the nuclear magnetic resonance hydrogen spectrum of the product is as follows:1H NMR(400MHz,CDCl3)δ7.83-7.76(m,4H),7.50-7.44(m,3H),7.39-7.28(m,5H),6.58(d,J=12.7Hz,1H),5.98(d,J=12.7Hz,1H),5.18(s,1H),5.04(s,1H),4.81(s,2H)。
the nuclear magnetic resonance carbon spectrum of the product is as follows:13C NMR(101MHz,CDCl3)δ150.7,145.4,138.3,136.8,133.4,132.9,128.6,128.1,128.1,127.6,127.6,126.7,126.5,126.1,125.9,112.5,108.9,72.0.
the high resolution mass spectrum of the product was: HRMS (ESI, m/z) C21H19O[M+H]+287.1430,calcd for 287.1429。
Example 3
Electron rich 1,3 butadiene was prepared as follows:
step 1: to an 8mL vial equipped with a magnetic stir bar were added 0.5mmol cesium carbonate, 0.24mmol N- (3- (benzyloxy) cyclobutyl) -4-methylbenzenesulfonylhydrazone, 0.004mmol tris (dibenzylideneacetone) dipalladium, and the vial was sealed with a screw cap and transferred to a glove box.
Step 2: a vial was charged with 0.008mmol diphenylcyclohexylphosphine, 2.0mL 1, 4-dioxane, and 0.2mmol aryl bromide (Ar is thiophene), capped, and removed from the glove box. The mixture was stirred for 16 hours on a magnetic stirrer at 90 ℃.
And step 3: the reaction mixture was cooled to room temperature, filtered through celite, eluted with ethyl acetate, dried in vacuo, and transferred to a silica gel column, which was then eluted with an eluent (PE/EA 100:1) to obtain the desired product (Rf 0.5(PE EA 40:1)) as a colorless oil in 73% yield.
The nuclear magnetic resonance hydrogen spectrum of the product is as follows:1H NMR(400MHz,CDCl3)δ7.87-7.74(m,2H),7.38-7.23(m,8H),6.39(d,J=12.6Hz,1H),5.99(d,J=12.7Hz,1H),5.27(d,J=2.0Hz,1H),5.00(d,J=2.1Hz,1H),4.73(s,2H)。
the reaction process is as follows:
the nuclear magnetic resonance carbon spectrum of the product is as follows:
13C NMR(101MHz,CDCl3)δ150.8,140.0,139.5,138.4,136.6,136.4,128.5,128.0,127.4,124.2,124.0,123.8,123.5,122.6,114.1,109.0,72.0。
the high resolution mass spectrum is: HRMS (ESI, m/z): calcd for C19H17OS[M+H]+293.0995,found 293.0995。
Example 4
Electron rich 1,3 butadiene was prepared as follows:
step 1: to an 8mL vial equipped with a magnetic stir bar were added 0.24mmol of 3- (benzyloxy) cyclobutane-1-one, 0.24mmol of p-toluenesulfonylhydrazide, and 1.0mL of 1, 4-dioxane. The resulting solution was sealed and stirred on a magnetic stirrer at 60 ℃ for 1 hour.
Step 2: after the reaction solution was cooled to room temperature, 0.5mmol of cesium carbonate, 0.004mmol of tris (dibenzylideneacetone) dipalladium, 0.008mmol of diphenylcyclohexylphosphine, 1.0mL of 1, 4-dioxane, and 0.2mmol of bromobenzene were added to the reaction mixture. The screw cap was closed and removed from the glove box and placed on a magnetic stirrer at 90 ℃ for 16 hours.
And step 3: the reaction mixture was cooled to room temperature, filtered through celite, eluted with ethyl acetate, dried in vacuo, transferred to a silica gel column, and eluted with an eluent (PE/EA 100:1) to give the desired product (Rf 0.5(PE EA 40:1)) as a colorless oil in 67% yield.
The reaction process is as follows:
the [4+2] cyclization reaction of partial compounds is explored, wherein the conjugated diene rich in electrons is a common substrate for [4+2] cyclization, and the conjugated diene and a dienophile (olefin or alkyne) can be subjected to Diels-Alder reaction to construct a six-membered ring compound, so that the conjugated diene is one of the very important means for forming carbon-carbon bonds in organic chemical synthesis reaction. The description will be made by taking example 5 as an example.
Example 5
The method comprises the following steps:
0.2mmol of 1-phenyl-1H-pyrrole-2, 5-dione was charged in a 4mL vial equipped with a magnetic stir bar, and 0.6mL of toluene was added. Then, 0.2mmol of conjugated diene was added. The resulting reaction mixture was stirred at 80 ℃ for 15 hours under nitrogen atmosphere. After cooling to room temperature, the reaction mixture was concentrated in vacuo. The crude mixture was purified by column chromatography on silica gel (PE: EA ═ 5:1) to afford the product as a white solid in 88% yield. The product structure is as follows:
the reaction process is as follows:
the nmr spectrum of the product is shown in fig. 7:1H NMR(400MHz,CDCl3)δ7.48-7.25(m,12H),7.24(s,1H),7.20-7.12(m,2H),6.45(dd,J=5.5,2.3Hz,1H),4.77-4.70(m,1H),4.58(d,J=11.4Hz,1H),4.50(d,J=11.4Hz,1H),3.37(td,J=9.4,7.1Hz,1H),3.25(dd,J=9.8,4.5Hz,1H),3.19-3.04(m,2H)。
the nuclear magnetic resonance carbon spectrum of the product is shown in FIG. 8:13C NMR(101MHz,CDCl3)δ179.0,175.8,143.2,139.7,137.8,132.1,129.0,128.5,128.4,128.3,128.2,127.6,127.5,126.7,125.6,123.2,123.1,70.7,70.6,45.0,37.6,25.4。
the high resolution mass spectrum of the product is shown in fig. 9: HRMS (ESI, m/z): calcd for C27H23NNaO3[M+Na]+432.1570,found 432.1574。
Compared with the prior art, the preparation method of the electron-rich conjugated diene compound provided by the invention has the advantages that the electron-rich conjugated diene compound is mainly obtained through ring opening isomerization of a four-membered ring, the stereospecificity is good, and an E-type product can be selectively obtained. The 1, 3-butadiene obtained by the method is obtained by thermal ring-opening isomerization of four-membered rings, and the thermal ring-opening of the four-membered rings can obtain products of Z type or E type or a mixture of the two by inward and outward rotation. One factor affecting the thermal ring opening of the four-membered ring is the substituent at the 3-position of cyclobutene, which increases its outward preference for substituents in the ring-opening isomerized product as the pi-donor properties of its substituents increase.
The raw materials adopted by the invention are beneficial to generating the product with the E-type structure.
The invention has the advantages of simple raw material preparation, mild reaction conditions, few reaction steps and simple reaction operation. The preparation method of the electron-rich 1,3 butadiene has good substrate universality and good functional group compatibility, can substitute groups such as electron-deficient groups, electron-rich groups, heterocyclic groups, naphthalene rings and the like on aromatic rings, has mild reaction conditions, and obtains products which have single structures and are E-type 1,3 butadiene.