Detailed Description
The following description of the exemplary embodiments of the present application, taken in conjunction with the accompanying drawings, includes various details of the embodiments of the application for the understanding thereof, which are to be regarded as merely exemplary. Accordingly, those of ordinary skill in the art will recognize that various changes and modifications of the embodiments described herein can be made without departing from the scope and spirit of the present application. In the case where there is no conflict with a definition in the present specification, a term in the present specification has a meaning generally understood by those skilled in the art, but in the case of conflict, the definition in the present specification controls.
Potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate (PNAC for short), represented by formula (iii):
powder X-ray diffraction
X-ray powder diffraction (XRPD) is commonly used for the analysis of crystal structure. X-rays are electromagnetic waves that, when incident on a crystal, produce a periodically varying electromagnetic field in the crystal. Causing the vibration of electrons and atomic nuclei in atoms, and neglecting the vibration due to the large mass of the atomic nuclei. The vibrating electrons are the source of secondary X-rays and have the same wavelength and phase as the incident light. Due to the periodicity of the crystal structure, scattered waves of respective electrons in the crystal interfere with each other and are superimposed on each other, which is called diffraction. The directions in which the scattered wave cycles are consistent and mutually enhanced are called diffraction directions, and diffraction rays are generated.
The instrument model is as follows: PANALYTIC EMPyrean and X' Pert3 ray powder diffraction analyzer;
ray: monochromatic Cu — K α rays (λ ═ 1.5406);
the scanning mode is as follows: θ/2 θ, scan range: 2-40 °;
voltage: 40KV, current: 40 mA.
Thermogravimetric analysis
Thermogravimetric analysis (TGA) refers to a thermal analysis technique for measuring the relationship between the mass of a sample to be measured and the temperature variation at a programmed temperature, and is used for studying the thermal stability and composition of materials. TGA is a relatively common detection means in both development and quality control. Thermogravimetric analysis is often combined with other analysis methods in actual material analysis to perform comprehensive thermal analysis, so that materials are comprehensively and accurately analyzed. The curve recorded by the thermogravimetric analyzer is called TGA curve.
The instrument model is as follows: TAQ5000/Discovery 5500;
and (3) purging gas: nitrogen gas;
a heating mode: linearly heating;
temperature range: room temperature to 350 ℃.
Differential scanning calorimetry
Differential Scanning Calorimetry (DSC), a technique that measures the heat flow rate of a sample relative to a reference as a function of temperature or time under temperature program control. The curve recorded by the differential scanning calorimeter, called DSC curve, is generally characterized by W/g or mW/mg (i.e. power flowing per gram of sample) as ordinate and temperature T or time T as abscissa, and can measure various thermodynamic and kinetic parameters, such as specific heat capacity, heat of reaction, transition heat, phase diagram, reaction rate, crystallization rate, polymer crystallinity, sample purity, etc. The method has the advantages of wide application temperature range (-175-725 ℃), high resolution and small sample consumption. It is suitable for analyzing inorganic matter, organic compound and medicine.
The instrument model is as follows: TAQ2000/Discovery 2500;
and (3) purging gas: nitrogen gas;
a heating mode: linearly heating;
temperature range: 25 to 300 ℃.
The present application provides a crystalline polymorph of potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate in form I having a 2 Θ ° of: an X-ray powder diffraction pattern of characteristic peaks expressed by 7.83 + -0.2, 26.64 + -0.2 and 18.89 + -0.2.
In the present application, the form I has a 2 Θ ° of: an X-ray powder diffraction pattern of characteristic peaks expressed by 7.83 + -0.2, 26.64 + -0.2, 18.89 + -0.2 and 5.24 + -0.2.
In the present application, the form I has a 2 Θ ° of: an X-ray powder diffraction pattern of characteristic peaks expressed by 7.83 + -0.2, 26.64 + -0.2, 18.89 + -0.2 and 21.59 + -0.2.
In the present application, the form I has a 2 Θ ° of: an X-ray powder diffraction pattern of characteristic peaks expressed by 7.83 + -0.2, 26.64 + -0.2, 18.89 + -0.2, 5.24 + -0.2 and 21.59 + -0.2.
In the present application, the form I has a 2 Θ ° of: an X-ray powder diffraction pattern of characteristic peaks expressed by 7.83 + -0.2, 26.64 + -0.2, 18.89 + -0.2, 5.24 + -0.2, 21.59 + -0.2 and 24.29 + -0.2.
In the present application, the form I has a 2 Θ ° of: an X-ray powder diffraction pattern of characteristic peaks expressed by 7.83 + -0.2, 26.64 + -0.2, 18.89 + -0.2, 5.24 + -0.2, 21.59 + -0.2 and 13.02 + -0.2.
In the present application, the form I has a 2 Θ ° of: an X-ray powder diffraction pattern of characteristic peaks expressed by 7.83 + -0.2, 26.64 + -0.2, 18.89 + -0.2, 5.24 + -0.2, 21.59 + -0.2, 13.02 + -0.2 and 24.29 + -0.2.
In the present application, the form I has a 2 Θ ° of: an X-ray powder diffraction pattern of characteristic peaks expressed by 7.83 + -0.2, 26.64 + -0.2, 18.89 + -0.2, 5.24 + -0.2, 21.59 + -0.2, 13.02 + -0.2, 24.29 + -0.2 and 6.61 + -0.2.
In the present application, the form I has a 2 Θ ° of: an X-ray powder diffraction pattern of characteristic peaks expressed by 7.83 + -0.2, 26.64 + -0.2, 18.89 + -0.2, 5.24 + -0.2, 21.59 + -0.2, 13.02 + -0.2, 24.29 + -0.2, 6.61 + -0.2 and 10.43 + -0.2.
In the present application, the form I has a 2 Θ ° of: an X-ray powder diffraction pattern of characteristic peaks expressed by 7.83 + -0.2, 26.64 + -0.2, 18.89 + -0.2, 5.24 + -0.2, 21.59 + -0.2, 13.02 + -0.2, 24.29 + -0.2, 6.61 + -0.2, 10.43 + -0.2 and 31.63 + -0.2.
In the present application, the form I has a 2 Θ ° of: an X-ray powder diffraction pattern of characteristic peaks expressed by 7.83 + -0.2, 26.64 + -0.2, 18.89 + -0.2, 5.24 + -0.2, 21.59 + -0.2, 13.02 + -0.2, 24.29 + -0.2, 6.61 + -0.2, 10.43 + -0.2, 31.63 + -0.2 and 37.00 + -0.2.
In the present application, the X-ray powder diffraction pattern of said form I is as shown in figure 1.
In the present application, the melting point of form I is 163.1 ℃.
In the present application, the adsorbed water removal temperature for form I is 83.6 ℃.
In the present application, form I lost 3.0% weight at 140 ℃.
The present application provides a crystalline polymorph of potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate in form II having at least a 2 Θ ° of: x-ray powder diffraction pattern of characteristic peaks expressed by 24.76 + -0.2, 6.73 + -0.2 and 20.26 + -0.2.
In the present application, the crystalline form II has a 2 Θ ° of: 24.76. + -. 0.2, 6.73. + -. 0.2, 20.26. + -. 0.2, 14.68. + -. 0.2.
In the present application, the crystalline form II has a 2 Θ ° of: 24.76. + -. 0.2, 6.73. + -. 0.2, 20.26. + -. 0.2, 25.55. + -. 0.2.
In the present application, the crystalline form II has a 2 Θ ° of: 24.76. + -. 0.2, 6.73. + -. 0.2, 20.26. + -. 0.2, 14.68. + -. 0.2, 25.55. + -. 0.2.
In the present application, the crystalline form II has a 2 Θ ° of: an X-ray powder diffraction pattern of characteristic peaks expressed by 24.76 + -0.2, 6.73 + -0.2, 20.26 + -0.2, 14.68 + -0.2, 25.55 + -0.2 and 13.41 + -0.2.
In the present application, the crystalline form II has a 2 Θ ° of: an X-ray powder diffraction pattern of characteristic peaks expressed by 24.76 + -0.2, 6.73 + -0.2, 20.26 + -0.2, 14.68 + -0.2, 25.55 + -0.2 and 26.66 + -0.2.
In the present application, the crystalline form II has a 2 Θ ° of: an X-ray powder diffraction pattern of characteristic peaks expressed by 24.76 + -0.2, 6.73 + -0.2, 20.26 + -0.2, 14.68 + -0.2, 25.55 + -0.2, 13.41 + -0.2 and 26.66 + -0.2.
In the present application, the crystalline form II has a 2 Θ ° of: an X-ray powder diffraction pattern of characteristic peaks expressed by 24.76 + -0.2, 6.73 + -0.2, 20.26 + -0.2, 14.68 + -0.2, 25.55 + -0.2, 13.41 + -0.2, 26.66 + -0.2 and 21.08 + -0.2.
In the present application, the crystalline form II has a 2 Θ ° of: an X-ray powder diffraction pattern of characteristic peaks expressed by 24.76 + -0.2, 6.73 + -0.2, 20.26 + -0.2, 14.68 + -0.2, 25.55 + -0.2, 13.41 + -0.2, 26.66 + -0.2, 21.08 + -0.2 and 25.79 + -0.2.
In the present application, the crystalline form II has a 2 Θ ° of: an X-ray powder diffraction pattern of characteristic peaks expressed by 24.76 + -0.2, 6.73 + -0.2, 20.26 + -0.2, 14.68 + -0.2, 25.55 + -0.2, 13.41 + -0.2, 26.66 + -0.2, 21.08 + -0.2, 25.79 + -0.2 and 28.47 + -0.2.
In the present application, the crystalline form II has a 2 Θ ° of: 24.76. + -. 0.2, 6.73. + -. 0.2, 20.26. + -. 0.2, 14.68. + -. 0.2, 25.55. + -. 0.2, 13.41. + -. 0.2, 26.66. + -. 0.2, 21.08. + -. 0.2, 25.79. + -. 0.2, 28.47. + -. 0.2, 12.07. + -. 0.2.
In the present application, the crystalline form II has a 2 Θ ° of: 24.76. + -. 0.2, 6.73. + -. 0.2, 20.26. + -. 0.2, 14.68. + -. 0.2, 25.55. + -. 0.2, 13.41. + -. 0.2, 26.66. + -. 0.2, 21.08. + -. 0.2, 25.79. + -. 0.2, 28.47. + -. 0.2, 12.07. + -. 0.2, 15.38. + -. 0.2.
In the present application, the crystalline form II has a 2 Θ ° of: 24.76. + -. 0.2, 6.73. + -. 0.2, 20.26. + -. 0.2, 14.68. + -. 0.2, 25.55. + -. 0.2, 13.41. + -. 0.2, 26.66. + -. 0.2, 21.08. + -. 0.2, 25.79. + -. 0.2, 28.47. + -. 0.2, 12.07. + -. 0.2, 15.38. + -. 0.2, 23.38. + -. 0.2.
In the present application, the crystalline form II has a 2 Θ ° of: 24.76. + -. 0.2, 6.73. + -. 0.2, 20.26. + -. 0.2, 14.68. + -. 0.2, 25.55. + -. 0.2, 13.41. + -. 0.2, 26.66. + -. 0.2, 21.08. + -. 0.2, 25.79. + -. 0.2, 28.47. + -. 0.2, 12.07. + -. 0.2, 15.38. + -. 0.2, 23.38. + -. 0.2 and 29.48. + -. 0.2.
In the present application, the crystalline form II has a 2 Θ ° of: 24.76. + -. 0.2, 6.73. + -. 0.2, 20.26. + -. 0.2, 14.68. + -. 0.2, 25.55. + -. 0.2, 13.41. + -. 0.2, 26.66. + -. 0.2, 21.08. + -. 0.2, 25.79. + -. 0.2, 28.47. + -. 0.2, 12.07. + -. 0.2, 15.38. + -. 0.2, 23.38. + -. 0.2, 29.48. + -. 0.2, 22.55. + -. 0.2.
In the present application, the crystalline form II has a 2 Θ ° of: 24.76. + -. 0.2, 6.73. + -. 0.2, 20.26. + -. 0.2, 14.68. + -. 0.2, 25.55. + -. 0.2, 13.41. + -. 0.2, 26.66. + -. 0.2, 21.08. + -. 0.2, 25.79. + -. 0.2, 28.47. + -. 0.2, 12.07. + -. 0.2, 15.38. + -. 0.2, 23.38. + -. 0.2, 29.48. + -. 0.2, 22.55. + -. 0.2, 27.79. + -. 0.2.
In the present application, the crystalline form II has a 2 Θ ° of: 24.76. + -. 0.2, 6.73. + -. 0.2, 20.26. + -. 0.2, 14.68. + -. 0.2, 25.55. + -. 0.2, 13.41. + -. 0.2, 26.66. + -. 0.2, 21.08. + -. 0.2, 25.79. + -. 0.2, 28.47. + -. 0.2, 12.07. + -. 0.2, 15.38. + -. 0.2, 23.38. + -. 0.2, 29.48. + -. 0.2, 22.55. + -. 0.2, 27.79. + -. 0.2, 8.91. + -. 0.2.
In the present application, the X-ray powder diffraction pattern of said form II is as shown in figure 3.
In the present application, the melting point of form II is 162.5 ℃.
In the present application, the adsorbed water removal temperature for form II is 93 ℃.
In the present application, form II lost 5.6% weight at 140 ℃.
The present application provides a crystalline polymorph of potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate in form III having at least a 2 Θ ° of: 9.06. + -. 0.2, 23.30. + -. 0.2, 21.44. + -. 0.2.
In the present application, the form III has a 2 Θ ° of: 9.06. + -. 0.2, 23.30. + -. 0.2, 21.44. + -. 0.2, 24.75. + -. 0.2.
In the present application, the form III has a 2 Θ ° of: an X-ray powder diffraction pattern of characteristic peaks expressed by 9.06 + -0.2, 23.30 + -0.2, 21.44 + -0.2 and 6.03 + -0.2.
In the present application, the form III has a 2 Θ ° of: an X-ray powder diffraction pattern of characteristic peaks expressed by 9.06 + -0.2, 23.30 + -0.2, 21.44 + -0.2, 24.75 + -0.2 and 6.03 + -0.2.
In the present application, the form III has a 2 Θ ° of: an X-ray powder diffraction pattern of characteristic peaks expressed by 9.06 + -0.2, 23.30 + -0.2, 21.44 + -0.2, 24.75 + -0.2, 6.03 + -0.2 and 21.20 + -0.2.
In the present application, the form III has a 2 Θ ° of: an X-ray powder diffraction pattern of characteristic peaks expressed by 9.06 + -0.2, 23.30 + -0.2, 21.44 + -0.2, 24.75 + -0.2, 6.03 + -0.2 and 17.06 + -0.2.
In the present application, the form III has a 2 Θ ° of: an X-ray powder diffraction pattern of characteristic peaks expressed by 9.06 + -0.2, 23.30 + -0.2, 21.44 + -0.2, 24.75 + -0.2, 6.03 + -0.2, 21.20 + -0.2 and 17.06 + -0.2.
In the present application, the form III has a 2 Θ ° of: an X-ray powder diffraction pattern of characteristic peaks expressed by 9.06 + -0.2, 23.30 + -0.2, 21.44 + -0.2, 24.75 + -0.2, 6.03 + -0.2, 21.20 + -0.2, 17.06 + -0.2 and 21.75 + -0.2.
In the present application, the form III has a 2 Θ ° of: an X-ray powder diffraction pattern of characteristic peaks expressed by 9.06 + -0.2, 23.30 + -0.2, 21.44 + -0.2, 24.75 + -0.2, 6.03 + -0.2, 21.20 + -0.2, 17.06 + -0.2, 21.75 + -0.2 and 29.52 + -0.2.
In the present application, the form III has a 2 Θ ° of: an X-ray powder diffraction pattern of characteristic peaks expressed by 9.06 + -0.2, 23.30 + -0.2, 21.44 + -0.2, 24.75 + -0.2, 6.03 + -0.2, 21.20 + -0.2, 17.06 + -0.2, 21.75 + -0.2, 29.52 + -0.2 and 22.15 + -0.2.
In the present application, the form III has a 2 Θ ° of: 9.06 + -0.2, 23.30 + -0.2, 21.44 + -0.2, 24.75 + -0.2, 6.03 + -0.2, 21.20 + -0.2, 17.06 + -0.2, 21.75 + -0.2, 29.52 + -0.2, 22.15 + -0.2, 15.11 + -0.2.
In the present application, the form III has a 2 Θ ° of: 9.06 + -0.2, 23.30 + -0.2, 21.44 + -0.2, 24.75 + -0.2, 6.03 + -0.2, 21.20 + -0.2, 17.06 + -0.2, 21.75 + -0.2, 29.52 + -0.2, 22.15 + -0.2, 15.11 + -0.2, 28.47 + -0.2.
In the present application, the form III has a 2 Θ ° of: an X-ray powder diffraction pattern of characteristic peaks expressed by 9.06 + -0.2, 23.30 + -0.2, 21.44 + -0.2, 24.75 + -0.2, 6.03 + -0.2, 21.20 + -0.2, 17.06 + -0.2, 21.75 + -0.2, 29.52 + -0.2, 22.15 + -0.2, 15.11 + -0.2, 28.47 + -0.2 and 22.54 + -0.2.
In the present application, the form III has a 2 Θ ° of: 9.06 + -0.2, 23.30 + -0.2, 21.44 + -0.2, 24.75 + -0.2, 6.03 + -0.2, 21.20 + -0.2, 17.06 + -0.2, 21.75 + -0.2, 29.52 + -0.2, 22.15 + -0.2, 15.11 + -0.2, 28.47 + -0.2, 22.54 + -0.2, 30.71 + -0.2.
In the present application, the form III has a 2 Θ ° of: 9.06 + -0.2, 23.30 + -0.2, 21.44 + -0.2, 24.75 + -0.2, 6.03 + -0.2, 21.20 + -0.2, 17.06 + -0.2, 21.75 + -0.2, 29.52 + -0.2, 22.15 + -0.2, 15.11 + -0.2, 28.47 + -0.2, 22.54 + -0.2, 30.71 + -0.2, and 17.91 + -0.2.
In the present application, the form III has a 2 Θ ° of: 9.06 + -0.2, 23.30 + -0.2, 21.44 + -0.2, 24.75 + -0.2, 6.03 + -0.2, 21.20 + -0.2, 17.06 + -0.2, 21.75 + -0.2, 29.52 + -0.2, 22.15 + -0.2, 15.11 + -0.2, 28.47 + -0.2, 22.54 + -0.2, 30.71 + -0.2, 17.91 + -0.2, 15.64 + -0.2.
In the present application, the form III has a 2 Θ ° of: 9.06 + -0.2, 23.30 + -0.2, 21.44 + -0.2, 24.75 + -0.2, 6.03 + -0.2, 21.20 + -0.2, 17.06 + -0.2, 21.75 + -0.2, 29.52 + -0.2, 22.15 + -0.2, 15.11 + -0.2, 28.47 + -0.2, 22.54 + -0.2, 30.71 + -0.2, 17.91 + -0.2, 15.64 + -0.2, 26.49 + -0.2.
In the present application, the form III has an X-ray powder diffraction pattern as shown in fig. 5.
In the present application, the melting point of form III is 162.0 ℃.
In the present application, the adsorbed water removal temperature for form III is 94.5 ℃.
In the present application, form III lost 6.1% weight at 140 ℃.
The present application provides a crystalline polymorph of potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate in form IV having at least a 2 Θ ° of: 16.25. + -. 0.2, 6.8. + -. 0.2 and 22.08. + -. 0.2.
In the present application, the form IV has a 2 Θ ° of: 16.25. + -. 0.2, 6.8. + -. 0.2, 22.08. + -. 0.2 and 13.16. + -. 0.2.
In the present application, the form IV has a 2 Θ ° of: 16.25. + -. 0.2, 6.8. + -. 0.2, 22.08. + -. 0.2 and 19.39. + -. 0.2.
In the present application, the form IV has a 2 Θ ° of: 16.25. + -. 0.2, 6.8. + -. 0.2, 22.08. + -. 0.2, 13.16. + -. 0.2 and 19.39. + -. 0.2.
In the present application, the form IV has a 2 Θ ° of: 16.25. + -. 0.2, 6.8. + -. 0.2, 22.08. + -. 0.2, 13.16. + -. 0.2, 19.39. + -. 0.2 and 18.35. + -. 0.2.
In the present application, the form IV has a 2 Θ ° of: 16.25. + -. 0.2, 6.8. + -. 0.2, 22.08. + -. 0.2, 13.16. + -. 0.2, 19.39. + -. 0.2 and 9.68. + -. 0.2.
In the present application, the form IV has a 2 Θ ° of: 16.25. + -. 0.2, 6.8. + -. 0.2, 22.08. + -. 0.2, 13.16. + -. 0.2, 19.39. + -. 0.2, 18.35. + -. 0.2 and 9.68. + -. 0.2.
In the present application, the form IV has a 2 Θ ° of: 16.25. + -. 0.2, 6.8. + -. 0.2, 22.08. + -. 0.2, 13.16. + -. 0.2, 19.39. + -. 0.2, 18.35. + -. 0.2, 9.68. + -. 0.2, 15.92. + -. 0.2.
In the present application, the form IV has a 2 Θ ° of: 16.25. + -. 0.2, 6.8. + -. 0.2, 22.08. + -. 0.2, 13.16. + -. 0.2, 19.39. + -. 0.2, 18.35. + -. 0.2, 9.68. + -. 0.2, 15.92. + -. 0.2 and 11.71. + -. 0.2.
In the present application, the form IV has a 2 Θ ° of: 16.25. + -. 0.2, 6.8. + -. 0.2, 22.08. + -. 0.2, 13.16. + -. 0.2, 19.39. + -. 0.2, 18.35. + -. 0.2, 9.68. + -. 0.2, 15.92. + -. 0.2, 11.71. + -. 0.2, 29.91. + -. 0.2.
In the present application, the form IV has a 2 Θ ° of: 16.25. + -. 0.2, 6.8. + -. 0.2, 22.08. + -. 0.2, 13.16. + -. 0.2, 19.39. + -. 0.2, 18.35. + -. 0.2, 9.68. + -. 0.2, 15.92. + -. 0.2, 11.71. + -. 0.2, 29.91. + -. 0.2 and 23.04. + -. 0.2.
In the present application, the form IV has a 2 Θ ° of: 16.25. + -. 0.2, 6.8. + -. 0.2, 22.08. + -. 0.2, 13.16. + -. 0.2, 19.39. + -. 0.2, 18.35. + -. 0.2, 9.68. + -. 0.2, 15.92. + -. 0.2, 11.71. + -. 0.2, 29.91. + -. 0.2, 23.04. + -. 0.2, 16.56. + -. 0.2.
In the present application, the form IV has a 2 Θ ° of: 16.25. + -. 0.2, 6.8. + -. 0.2, 22.08. + -. 0.2, 13.16. + -. 0.2, 19.39. + -. 0.2, 18.35. + -. 0.2, 9.68. + -. 0.2, 15.92. + -. 0.2, 11.71. + -. 0.2, 29.91. + -. 0.2, 23.04. + -. 0.2, 16.56. + -. 0.2, 23.5. + -. 0.2.
In the present application, the form IV has a 2 Θ ° of: 16.25 + -0.2, 6.8 + -0.2, 22.08 + -0.2, 13.16 + -0.2, 19.39 + -0.2, 18.35 + -0.2, 9.68 + -0.2, 15.92 + -0.2, 11.71 + -0.2, 29.91 + -0.2, 23.04 + -0.2, 16.56 + -0.2, 23.5 + -0.2, and 27.31 + -0.2.
In the present application, the form IV has a 2 Θ ° of: 16.25 + -0.2, 6.8 + -0.2, 22.08 + -0.2, 13.16 + -0.2, 19.39 + -0.2, 18.35 + -0.2, 9.68 + -0.2, 15.92 + -0.2, 11.71 + -0.2, 29.91 + -0.2, 23.04 + -0.2, 16.56 + -0.2, 23.5 + -0.2, 27.31 + -0.2, and 19.74 + -0.2.
In the present application, the form IV has a 2 Θ ° of: 16.25 + -0.2, 6.8 + -0.2, 22.08 + -0.2, 13.16 + -0.2, 19.39 + -0.2, 18.35 + -0.2, 9.68 + -0.2, 15.92 + -0.2, 11.71 + -0.2, 29.91 + -0.2, 23.04 + -0.2, 16.56 + -0.2, 23.5 + -0.2, 27.31 + -0.2, 19.74 + -0.2, 34.34 + -0.2.
In the present application, the form IV has a 2 Θ ° of: 16.25 + -0.2, 6.8 + -0.2, 22.08 + -0.2, 13.16 + -0.2, 19.39 + -0.2, 18.35 + -0.2, 9.68 + -0.2, 15.92 + -0.2, 11.71 + -0.2, 29.91 + -0.2, 23.04 + -0.2, 16.56 + -0.2, 23.5 + -0.2, 27.31 + -0.2, 19.74 + -0.2, 34.34 + -0.2, 18.82 + -0.2.
In the present application, the form IV has an X-ray powder diffraction pattern as shown in figure 7.
In the present application, the melting point of form IV is 163.8 ℃.
In the present application, the adsorbed water removal temperature for form IV is 96.1 ℃.
In the present application, form IV lost 8.21% weight at 150 ℃.
The crystal forms I-IV of PNAC provided by the application have good solubility, bioavailability and solid stability, and particularly the bioavailability and solid stability of the crystal forms I and II are good.
The application provides a preparation method of a crystal form I of potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate, which comprises the following steps:
the method comprises the following steps: adding an organic solvent into a reaction container, stirring, then adding N- [8- (2-hydroxybenzoyl) amino ] caprylic acid, stirring uniformly, dropwise adding a potassium hydroxide solution, and concentrating to obtain a crude product after dropwise adding;
step two: adding an organic solvent into the crude product, pulping, filtering to obtain a filter cake, washing the filter cake, and drying in a drying oven at 60-100 ℃ for 30-40 h to obtain the crystal form I of the potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate.
The drying temperature may be 60 deg.C, 65 deg.C, 70 deg.C, 75 deg.C, 80 deg.C, 85 deg.C, 90 deg.C, 95 deg.C, 100 deg.C. The drying time can be 30h, 31h, 32h, 33h, 34h, 35h, 36h, 37h, 38h, 39h, 40 h.
Preferably, the drying is carried out in two steps, namely drying at 60 ℃ for 16h, flattening the system with nitrogen, and drying at 100 ℃ for 24h again.
In the present application, the organic solvent is isopropanol or acetone.
In the present application, the solution concentration of the potassium hydroxide is 40% to 90%, preferably 50%.
The concentration of the potassium hydroxide solution may be 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%.
In the application, after N- [8- (2-hydroxybenzoyl) amino ] caprylic acid is added, the temperature of the system is raised to be more than 48 ℃, then potassium hydroxide solution is dripped, and after the dripping is finished, the temperature of more than 48 ℃ is kept for reaction for 0.5h-2 h.
Preferably, after adding N- [8- (2-hydroxybenzoyl) amino ] caprylic acid, the system is heated to 48-52 ℃, then potassium hydroxide solution is added dropwise, and after the dropwise addition is finished, the temperature of 48-52 ℃ is kept for reaction for 1 h.
The system temperature can be 48 ℃, 49 ℃, 50 ℃, 51 ℃, 52 ℃, 53 ℃, 54 ℃ and 55 ℃.
The time of the heat preservation reaction can be 0.5h, 0.6h, 0.7h, 0.8h, 0.9h, 1h, 1.1h, 1.2h, 1.3h, 1.4h, 1.5h, 1.6h, 1.7h, 1.8h, 1.9h and 2 h.
In this application, the molar ratio of N- [8- (2-hydroxybenzoyl) amino ] octanoic acid to potassium hydroxide is 1: 1.
In the application, the organic solvent is added into the crude product for pulping for 0.5h-1.5h, preferably 1 h.
The time for adding the organic solvent into the crude product and pulping can be 0.5h, 0.6h, 0.7h, 0.8h, 0.9h, 1h, 1.1h, 1.2h, 1.3h, 1.4h, 1.5h, 1.6h, 1.7h, 1.8h, 1.9h and 2 h.
The present application provides a second process for preparing form I of potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate by heating a form of potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate other than form I to above at least 75 ℃ to form I.
In the application, the crystal form of the potassium N- [8- (2-hydroxybenzoyl) amino ] octoate except the crystal form I is at least one or more of crystal form II, crystal form III and crystal form IV.
The crystal form of the potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate except the crystal form I can be a crystal form II.
The crystal form of the potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate except the crystal form I can be a crystal form III.
The crystal form of the potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate except the crystal form I can be a crystal form IV.
The crystal forms of the potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate except the crystal form I can be a crystal form II and a crystal form III.
The crystal forms of the potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate except the crystal form I can be a crystal form II and a crystal form IV.
The crystal forms of the potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate except the crystal form I can be a crystal form III and a crystal form IV.
The crystal forms of the potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate except the crystal form I can be a crystal form II, a crystal form III and a crystal form IV.
In the application, crystal forms of potassium N- [8- (2-hydroxybenzoyl) amino ] octoate except crystal form I are heated to over 75 ℃ under the protection of nitrogen for 0-300 min to form the crystal form I of the potassium N- [8- (2-hydroxybenzoyl) amino ] octoate.
The heating temperature is preferably 110-140 ℃. The heating temperature may be 110 deg.C, 115 deg.C, 120 deg.C, 125 deg.C, 130 deg.C, 135 deg.C, 140 deg.C.
The heating time may be 10min, 15min, 20min, 25min, 30min, 35min, 40min, 45min, 50min, 55min, 60min, 65min, 70min, 75min, 80min, 85min, 90min, 95min, 100min, 105min, 110min, 115min, 120min, 125min, 130min, 135min, 140min, 145min, 150min, 155min, 160min, 165min, 170min, 175min, 180min, 185min, 190min, 195min, 200min, 205min, 210min, 220min, 225min, 230min, 235min, 240min, 245min, 250min, 255min, 260min, 265min, 270min, 275min, 280min, 285min, 290min, 295min, 300 min.
In this application, form II of potassium N- [8- (2-hydroxybenzoyl) amino ] octanoate is heated to 140 ℃ under nitrogen protection to form I of potassium N- [8- (2-hydroxybenzoyl) amino ] octanoate.
In this application, form IV of potassium N- [8- (2-hydroxybenzoyl) amino ] octanoate is heated to 110 ℃ under nitrogen protection to form I of potassium N- [8- (2-hydroxybenzoyl) amino ] octanoate.
The application provides a preparation method of a third potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate crystal form I, wherein crystal forms of the potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate except the crystal form I are lyophilized to form the crystal form I.
In the application, the crystal form of the potassium N- [8- (2-hydroxybenzoyl) amino ] octoate except the crystal form I is at least one or more of crystal form II, crystal form III and crystal form IV.
The application provides a preparation method of N- [8- (2-hydroxybenzoyl) amino ] potassium octoate crystal form III, which comprises the following steps:
the method comprises the following steps: adding an organic solvent into a reaction container, stirring, then adding N- [8- (2-hydroxybenzoyl) amino ] caprylic acid, stirring uniformly, dropwise adding a potassium hydroxide solution, and concentrating to obtain a crude product after dropwise adding;
step two: adding an organic solvent into the crude product, pulping, filtering to obtain a filter cake, preparing the filter cake into uniform particles, then putting the particles into the drying oven for drying, uniformly spreading the dried particles in a low-temperature environment of 2-8 ℃, and standing for 2 days with the relative humidity controlled at 50% to form the N- [8- (2-hydroxybenzoyl) amino ] potassium caprylate crystal form III.
In the application, the filter cake is sieved by a 20-24-mesh sieve to obtain uniform particles.
In the application, the drying temperature is 60-100 ℃, and the drying time is 30-40 h. Preferably, the drying temperature may be 60 ℃,65 ℃, 70 ℃, 75 ℃, 80 ℃, 85 ℃, 90 ℃, 95 ℃, 100 ℃. The drying time can be 30h, 31h, 32h, 33h, 34h, 35h, 36h, 37h, 38h, 39h, 40 h.
Preferably, the drying is carried out in two steps, namely drying at 60 ℃ for 16h, flattening the system with nitrogen, and drying at 100 ℃ for 24h again.
In the present application, the organic solvent is isopropanol or acetone.
In the present application, the solution concentration of the potassium hydroxide is 40% to 90%, preferably 50%.
The concentration of the potassium hydroxide solution may be 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%.
In the application, after N- [8- (2-hydroxybenzoyl) amino ] caprylic acid is added, the temperature of the system is raised to be more than 48 ℃, then potassium hydroxide solution is dripped, and after the dripping is finished, the temperature of more than 48 ℃ is kept for reaction for 0.5h-2 h.
Preferably, after adding N- [8- (2-hydroxybenzoyl) amino ] caprylic acid, the system is heated to 48-52 ℃, then potassium hydroxide solution is added dropwise, and after the dropwise addition is finished, the temperature of 48-52 ℃ is kept for reaction for 1 h.
The system temperature can be 48 ℃, 49 ℃, 50 ℃, 51 ℃, 52 ℃, 53 ℃, 54 ℃ and 55 ℃.
The time of the heat preservation reaction can be 0.5h, 0.6h, 0.7h, 0.8h, 0.9h, 1h, 1.1h, 1.2h, 1.3h, 1.4h, 1.5h, 1.6h, 1.7h, 1.8h, 1.9h and 2 h.
In this application, the molar ratio of N- [8- (2-hydroxybenzoyl) amino ] octanoic acid to potassium hydroxide is 1: 1.
In the application, the organic solvent is added into the crude product for pulping for 0.5h-1.5h, preferably 1 h.
The time for adding the organic solvent into the crude product and pulping can be 0.5h, 0.6h, 0.7h, 0.8h, 0.9h, 1h, 1.1h, 1.2h, 1.3h, 1.4h, 1.5h, 1.6h, 1.7h, 1.8h, 1.9h and 2 h.
The application provides a preparation method of a crystal form II of potassium N- [8- (2-hydroxybenzoyl) amino ] octoate, which comprises the step of exposing crystal forms of potassium N- [8- (2-hydroxybenzoyl) amino ] octoate except the crystal form II to an environment with 0-60% of relative humidity for more than 24 hours at room temperature so as to form the crystal form II of the potassium N- [8- (2-hydroxybenzoyl) amino ] octoate.
In the present application, the relative humidity is 20%, 30%, 40%, 50%, 60% of the environment.
In the present application, the time for forming the crystal form II of the potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate may be 24h, 25h, 26h, 27h, 28h, 29h, 30h, 31h, 32h, 33h, 34h, 35h, 36h, 37h, 38h, 39h, 40h, 41h, 42h, 43h, 44h, 45h, 46h, 47h, 48h, etc.
In the application, the crystal form of the potassium N- [8- (2-hydroxybenzoyl) amino ] octoate except the crystal form II is at least one or more of crystal form I, crystal form III and crystal form IV.
The crystal form of the potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate except the crystal form II can be a crystal form I.
The crystal form of the potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate except the crystal form II can be a crystal form III.
The crystal form of the potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate except the crystal form II can be a crystal form IV.
The crystal forms of the potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate except the crystal form II can be a crystal form I and a crystal form III.
The crystal forms of the potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate except the crystal form II can be a crystal form I and a crystal form IV.
The crystal forms of the potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate except the crystal form II can be a crystal form III and a crystal form IV.
The crystal forms of the potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate except the crystal form II can be a crystal form I, a crystal form III and a crystal form IV.
The application provides a preparation method of a potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate crystal form IV, which comprises the following steps:
forming a colloid substance from crystal forms of potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate except crystal form IV in an environment with relative humidity higher than 80%;
exposing the glue-like substance to an environment with relative humidity of 20-40% for more than 120h at room temperature to form a crystal form IV.
In the application, the crystal form of the potassium N- [8- (2-hydroxybenzoyl) amino ] octoate except the crystal form IV is at least one or more of crystal form I, crystal form II and crystal form III.
The crystal form of the potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate except the crystal form IV can be a crystal form I.
The crystal form of the potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate except the crystal form IV can be a crystal form II.
The crystal form of the potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate except the crystal form IV can be a crystal form III.
The crystal forms of the potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate except the crystal form IV can be a crystal form I and a crystal form III.
The crystal forms of the potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate except the crystal form IV can be a crystal form I and a crystal form II.
The crystal forms of the potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate except the crystal form IV can be a crystal form III and a crystal form II.
The crystal forms of the potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate except the crystal form IV can be a crystal form I, a crystal form II and a crystal form III.
In the present application, the gum-like substance is exposed to an environment having a relative humidity of 20%, 30% or 40%, preferably the gum-like substance is exposed to an environment having a relative humidity of 40%.
In the present application, the crystalline form other than form IV is allowed to stand in an environment of relative humidity higher than 80% for at least two days or more to form a gel-like substance. The standing time can be 48h, 50h, 55h, 60h, 65h, 72h and the like.
The glue-like substance is exposed to an environment with 20-40% relative humidity at room temperature, and the time for forming the crystal form IV can be 120h, 121h, 122h, 123h, 124h, 125h, 126h, 127h, 128h, 129h, 130h, 131h, 132h, 133h, 134h, 135h, 136h, 137h, 138h, 139h, 140h and the like.
The present application provides a pharmaceutical composition comprising a crystalline polymorph of potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate.
The pharmaceutical composition also comprises a preventive and/or therapeutic drug, wherein the preventive and/or therapeutic drug can be glucagon-like peptide-1 (GLP-1 for short), insulin, PYY, human amylin, heparin, human growth hormone, interferon, monoclonal antibody, protease inhibitor and thrombopoietin.
In the application, the polymorphic substance of the potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate crystal in the pharmaceutical composition is one or more than two of a crystal form I, a crystal form II, a crystal form III and a crystal form IV.
The pharmaceutical composition comprises a crystal form I of potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate.
The pharmaceutical composition comprises a crystal form II of potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate.
The pharmaceutical composition comprises a crystal form III of potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate.
The pharmaceutical composition comprises a crystal form IV of potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate.
The pharmaceutical composition comprises a crystal form I and a crystal form II of potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate.
The pharmaceutical composition comprises a crystal form I and a crystal form III of potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate.
The pharmaceutical composition comprises a crystal form I and a crystal form IV of potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate.
The pharmaceutical composition comprises a crystal form II and a crystal form III of potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate.
The pharmaceutical composition comprises a crystal form II and a crystal form IV of potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate.
The pharmaceutical composition comprises a crystal form III and a crystal form IV of potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate.
The pharmaceutical composition comprises a crystal form I, a crystal form II and a crystal form III of potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate.
The pharmaceutical composition comprises a crystal form II, a crystal form III and a crystal form IV of potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate.
The pharmaceutical composition comprises a crystal form I, a crystal form III and a crystal form IV of potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate.
The pharmaceutical composition comprises a crystal form I, a crystal form II, a crystal form III and a crystal form IV of potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate.
In the present application, the weight ratio of the crystalline polymorph of potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate to the prophylactic and/or therapeutic drug in the pharmaceutical composition may be (20-60): 1.
In the present application, the weight ratio of the crystalline polymorph of potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate to the prophylactic and/or therapeutic drug in the pharmaceutical composition may be 30: 1.
The weight ratio of the crystalline polymorph of potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate to the prophylactic and/or therapeutic drug in the pharmaceutical composition may be 20:1, 21:1, 22:1, 23:1, 24:1, 25:1, 26:1, 27:1, 28:1, 29:1, 30:1, 31:1, 32:1, 33:1, 34:1, 35:1, 36:1, 37:1, 38:1, 39:1, 40:1, 41:1, 42:1, 43:1, 44:1, 45:1, 46:1, 47:1, 48:1, 49:1, 50:1, 51:1, 52:1, 53:1, 54:1, 55:1, 56:1, 57:1, 58:1, 59:1, 60: 1.
The pharmaceutical composition may be a tablet.
The application provides an application of a potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate crystal polymorph or a pharmaceutical composition thereof in preparing preventive and/or therapeutic medicaments.
The application provides a use of a potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate crystal polymorph or a pharmaceutical composition thereof in promoting drug delivery.
The application provides an application of a potassium N- [8- (2-hydroxybenzoyl) amino ] caprylate crystal polymorph or a pharmaceutical composition thereof in preparing a medicament for preventing and/or treating diabetes or diabetic complications.
Prophylactic and/or therapeutic agent
Prophylactic and/or therapeutic agents refer to agents that are used for the purpose of avoiding, curing, alleviating, relieving, altering, treating, ameliorating, improving or affecting a condition (e.g., a disease), a symptom of a disease, or a susceptibility to a disease.
The prophylactic and/or therapeutic agent may be a protein; a polypeptide; a peptide; a hormone; polysaccharides, mucopolysaccharides and specific mixtures of mucopolysaccharides; a carbohydrate; a lipid; polar organic small molecules (i.e., polar organic molecules having a molecular weight of 500 daltons or less); other organic compounds; and specific compounds which are not themselves susceptible to cleavage by the gastrointestinal mucosa (only by part of the administered dose) and/or by the activity of acids and enzymes in the gastrointestinal tract; or any combination thereof.
Prophylactic and/or therapeutic agents may include (but are not limited to) the following: including their synthetic, natural or recombinant sources: growth hormones, including human growth hormone (hGH), recombinant human growth hormone (rhGH), bovine growth (hGH), bovine growth hormone, and porcine growth hormone; growth hormone releasing hormone; growth hormone releasing factor (e.g., GRF analog g); interferons, including α, β, and γ; interleukin 1; interleukin 2; insulin, including porcine, bovine, human and human recombinant insulin, optionally with counterions including zinc, sodium, calcium and ammonium; insulin-like growth factors including IGF-l; heparin, including unfractionated heparin, heparin analogs, dermatans, chondroitins, low molecular weight heparin, very low molecular weight heparin, and ultra low molecular weight heparin; calcitonin, including salmon, eel, pig and human calcitonin; erythropoietin; atrial natriuretic peptide; an antigen; a monoclonal antibody; a somatostatin; a protease inhibitor; corticotropin, gonadotropin releasing hormone; oxytocin; luteinizing hormone-releasing hormone; an egg cell stimulating hormone; glucocerebrosidase; thrombopoietin; filgrastim; prostaglandins; cyclosporine; a vasopressin; cromolyn sodium (sodium or disodium cromoglycate); vancomycin; desferrioxamine (DFO); bisphosphonates, including ibandronate, alendronate, tiludronate, etidronate, clodronate, pamidronate, olpadronate and incadronate and pharmaceutically acceptable salts thereof (e.g., ibandronate sodium); gallium salts (such as gallium nitrate, gallium nitrate nonahydrate, and gallium maltolate); acyclovir and pharmaceutically acceptable salts thereof (e.g., sodium acyclovir); parathyroid hormone (PTH), including fragments thereof; anti-migraine agents, such as BIBN-4096BS and other calcitonin gene-related protein antagonists; antimicrobial agents, including antibiotics (including bactericidal, lipopeptide and cyclopeptide antibiotics that act on gram-positive bacteria, including daptomycin), antibacterial and antifungal agents; a vitamin; analogs, fragments, mimetics, or polyethylene glycol (PEG) modified derivatives of these compounds; or any combination thereof.
The preferred drugs in the application are polypeptides and protein drugs which are difficult to be absorbed into intestinal tract successfully by oral administration, and common drugs are insulin, monoclonal antibodies, heparin, glucagon-like peptide, PYY, human amylin, human growth hormone, interferon, protease inhibitors, thrombopoietin and the like, including analogues, fragments, mimics and polyethylene glycol modified derivatives thereof.
Insulin
Insulin is a protein hormone secreted by the beta cells of the islets of langerhans in the pancreas stimulated by endogenous or exogenous substances such as glucose, lactose, ribose, arginine, glucagon, etc. Insulin is the only hormone in the body which reduces blood sugar and promotes the synthesis of glycogen, fat and protein. Exogenous insulin is mainly used for treating diabetes. All crystalline forms of insulin in the present application include, but are not limited to, naturally occurring insulin and synthetic insulin. Because oral administration is easily degraded, subcutaneous injection is still the main reason nowadays.
PYY
PYY is secreted by endocrine cells distal to the gastrointestinal tract after meals and acts on satiety for hypothalamic signaling. Recent studies have shown that fasting and postprandial PYY levels in obese subjects are low, which may be responsible for their high appetite and food consumption. When administered intravenously, it suppresses appetite and food intake in lean and obese subjects. Other peptides from the Pancreatic Peptide (PP) family, such as fragments of PYY (e.g., PYY [3-36]) and other PYY agonists, also suppress appetite. However, due to its low absorption and rapid degradation in the gastrointestinal tract, its oral activity is essentially negligible.
Glucagon-like peptide-1
Glucagon-like peptide-1 (GLP-1 for short) is a kind of brain intestine peptide secreted by ileum endocrine cell, and is mainly used as target point of medicine action for type 2 diabetes at present. GLP-1 can inhibit gastric emptying and reduce intestinal peristalsis, so that it is helpful for controlling food intake and reducing body weight. However, GLP-1 is polypeptide, so that oral administration is easy to degrade and difficult to reach the intestinal tract.
Human amylin
Human amylin (hIAPP) is a polypeptide hormone composed of 37 amino acids and synthesized and secreted by islet cells, and is used in conjunction with insulin and glucagon to regulate sugar homeostasis. The physiological and pharmacological functions of the hIAPP monomer are as follows: 1) affecting the secretion of insulin and glucagon; 2) the gastric emptying is delayed, and the postprandial blood sugar is reduced; 3) increase renin and angiotensin II, regulate kidney growth; 4) increasing aldosterone and reducing blood calcium; 5) adjusting bone density; 6) vasodilatation, regulating hemodynamics; 7) modulating immune effects. The hIAPP monomers induce the regulation of T cell differentiation, thereby regulating the inflammatory response and secretion of immune factors. The hIAPP has potential application prospect in the prevention and treatment process of obesity, diabetes, autoimmunity, osteoporosis and other diseases.
Pharmaceutical composition
In addition to the PNAC and prophylactic and/or therapeutic agents described herein, the pharmaceutical compositions may also include pharmaceutically acceptable excipients, which may be non-toxic fillers, stabilizers, diluents, carriers, solvents or other formulation excipients. For example, diluents, excipients, such as microcrystalline cellulose, mannitol, and the like; fillers, such as starch, sucrose, and the like; binders, such as starch, cellulose derivatives, alginates, gelatin and/or polyvinylpyrrolidone; disintegrants, such as calcium carbonate and/or sodium bicarbonate; absorption promoters, such as quaternary ammonium compounds; surfactants such as cetyl alcohol; carriers, solvents, such as water, physiological saline, kaolin, bentonite, etc.; lubricants, such as talc, calcium/magnesium stearate, polyethylene glycol, and the like. In addition, the pharmaceutical composition of the present application is preferably an injection.
The pharmaceutical compositions are preferably in solid form and they may be formulated into solid dosage forms. The solid dosage form may be a capsule, tablet or granule, such as a powder or sachet. The powder may be in a sachet that is mixed with the liquid and applied. Solid dosage forms may also be topical delivery systems, such as ointments, creams or semisolids. Contemplated solid dosage forms may include sustained or controlled release systems. Preferably the solid dosage form is a dosage form for oral administration.
The powders may be packaged in capsules or compressed into tablets, used in powder form or incorporated into ointments, creams or semisolids. Methods for forming solid dosage forms are well known in the art.
The PNAC in the present application may serve as a delivery agent in a pharmaceutical composition.
The amount of delivery agent in the solid dosage form is a delivery effective amount and can be determined by methods well known to those skilled in the art for any particular compound or biologically or chemically active agent.
Following administration, the active agent in the unit dosage form is absorbed into the circulation. The bioavailability of an active agent is readily assessed by measuring known pharmacological activity in the blood, such as increased clotting time caused by heparin or decreased circulating levels of calcium caused by calcitonin. Alternatively, the circulating level of the active agent itself may be measured directly.
Delivery system
The amount of prophylactic and/or therapeutic drug (which may be referred to as an active agent) used in the pharmaceutical compositions of the present application is an effective amount to achieve the purpose of the active agent for the target indication. The active agent is generally employed in the composition in a pharmacologically, biologically, therapeutically or chemically effective amount. However, the amount may be lower than when the composition is used in a unit dosage form, as the unit dosage form may contain multiple delivery agent compounds/active agent compositions or may contain fractionated pharmacologically, biologically, therapeutically or actively effective amounts. The total effective amount can be administered in cumulative units that collectively contain an effective amount of the active agent.
The total amount of active agent used can be determined by methods well known to those skilled in the art. However, because the compositions of the present application can deliver the active agent more effectively than other compositions or compositions containing the active agent alone, lower amounts of biologically or chemically active agent than are used in existing unit dosage forms or delivery systems can be administered to a subject while still achieving the same blood levels and/or therapeutic effect.
The delivery agents disclosed herein facilitate the delivery of biologically and chemically active agents, particularly oral, sublingual, buccal, intraduodenal, intracolonic, rectal, vaginal, mucosal, pulmonary, intranasal, and ocular systems.
The compounds and compositions of the present application are useful for administering biologically or chemically active agents to any animal, including (but not limited to): birds, such as chickens; mammals, such as rodents, cows, pigs, dogs, cats, primates, and particularly humans; and insects.
These compounds and compositions are particularly advantageous for the delivery of chemically or biologically active agents that might otherwise be destroyed or reduced in activity by the time the agent reaches the target area (i.e., the area from which the active agent of the delivery composition is released) and before it reaches the area encountered. In particular, the compounds and compositions of the present application are useful for oral administration of active agents, especially those that are not commonly delivered by oral administration or those for which enhanced delivery is desired.
Compositions comprising the compounds and active agents have utility in delivering the active agent to a selected biological system and increasing or enhancing the bioavailability of the active agent over delivery of the active agent without the use of a delivery agent. Delivery may be improved by delivering more active agent over a period of time or delivering the active agent over a particular period of time (such as faster acting or delayed delivery) or over a period of time (such as sustained delivery).
Examples
The experimental methods used in the following examples are all conventional methods, unless otherwise specified.
Materials, reagents and the like used in the following examples are commercially available unless otherwise specified.
Example 1
Preparation of NAC N- [8- (2-hydroxybenzoyl) amino ] octanoate by reference to the procedure of example 1 of International patent application WO2008/028859
Preparation of form I PNAC
To a 50L reactor was added isopropanol (22070.0ml, 4.0vol), with stirring on, and NAC (5518g, 1.0eq) was added. The system was warmed to 50 ℃ and the prepared 50% potassium hydroxide solution (1304.0g, 1.0eq) was added dropwise to the system. After the dripping is finished, the system becomes clear and transparent yellow solution, and the temperature is kept at 50 ℃ for reaction for 1 h. The reaction solution was concentrated in portions at 40 ℃ to give a crude product which was pale orange yellow in color.
The crude product is added with isopropanol (19310.0ml, 3.5vol) in batch for pulping for 1 h. The system was filtered with suction and the filter cake was rinsed with isopropanol (2760.0ml, 0.5 vol). And transferring the filter cake into a vacuum drying oven, flatly pressing the drying system with nitrogen, drying for 16h at the temperature of 60 ℃, and transferring into the vacuum drying oven again to dry for 24h at the temperature of 100 ℃. After the drying was completed, 4.52kg of the product was obtained in a yield of 72.8% and was in the form of a white powdery solid.
The product is determined to be the crystal form I, XRPD and TGA/DSC of the crystal form I are respectively shown in figures 1-2 b, and PNAC is the crystal form I, wherein characteristic XRPD peaks (characterized by degrees 2 theta) of the crystal form I are shown in the following table 1:
table 1 is the characteristic XRPD peak for form I (characterized by 2 θ.)
A DSC (figure 2a) graph shows that the melting endothermic peak of the crystal form I begins to appear at 195.5 ℃, and the peak value is 163.1 ℃; the combination of DSC (figure 2a) and TGA (figure 2b) patterns reveals that form I is an anhydrate.
Example 2
Preparation of PNAC Crystal form II
And (3) contacting the PNAC crystal form I prepared in the example 1 with an environment with relative humidity of 60% at room temperature, and standing for more than 24h to obtain the PNAC crystal form I.
The product was determined to be form II, which is shown in figures 3-4b for XRPD, TGA/DSC, respectively, wherein the characteristic XRPD peaks (characterized in ° 2 θ) for form II are given in table 2 below:
table 2 is the characteristic XRPD peak for form II (characterized by ° 2 θ)
A DSC (figure 4a) graph shows that the melting endothermic peak of the crystal form II begins to appear at 157.1 ℃, and the peak value is 162.5 ℃; the combination of DSC (fig. 4a) and TGA (fig. 4b) patterns may reveal form II as 1/3 hydrate.
Example 3
Preparation of NAC N- [8- (2-hydroxybenzoyl) amino ] octanoate by reference to the procedure of example 1 of International patent application WO2008/028859
Preparation of PNAC form III
To a 50L reactor was added isopropanol (22070.0ml, 4.0vol), with stirring on, and NAC (5518g, 1.0eq) was added. The system was warmed to 50 ℃ and the prepared 50% potassium hydroxide solution (1304.0g, 1.0eq) was added dropwise to the system. After the dripping is finished, the system becomes clear and transparent yellow solution, and the temperature is kept at 50 ℃ for reaction for 1 h. The reaction solution was concentrated in portions at 40 ℃ to give a crude product which was pale orange yellow in color.
The crude product is added with isopropanol (19310.0ml, 3.5vol) in batch for pulping for 1 h. And carrying out suction filtration on the system to obtain a filter cake, sieving the filter cake with a 24-mesh sieve to prepare uniform particles, then transferring the particles into a vacuum drying oven for drying, carrying out nitrogen flat pressure on the drying system, drying for 16h at 60 ℃, transferring into the vacuum drying oven again at 100 ℃, drying for 24h to obtain particles, uniformly spreading the particles in a low-temperature environment at 5 ℃, and standing for 2 days under the condition that the relative humidity is controlled to be 50% to obtain the product.
The product was determined to be form III having XRPD, TGA/DSC as shown in figures 5-6b, respectively, wherein the characteristic XRPD peaks (characterized in ° 2 θ) for form III are given in table 3 below:
table 3 is the characteristic XRPD peak for form III (characterized by ° 2 θ)
A DSC (figure 6a) shows that the melting endothermic peak of the crystal form III begins to appear at 156.6 ℃, and the peak value is 162.0 ℃; the combination of DSC (fig. 6a) and TGA (fig. 6b) patterns reveals form III as 1/2 hydrate.
Example 4
Preparation of PNAC form IV
The PNAC form I prepared in example 1 was left to stand at room temperature in an environment of 80% relative humidity for 3 days to form a gum-like material, which was exposed to an environment of 40% relative humidity for 5 days to form a product.
The product was determined to be form IV having XRPD, TGA/DSC as shown in figures 7-8b, respectively, wherein the characteristic XRPD peaks (characterized in ° 2 θ) for form IV are given in table 4 below:
table 4 is the characteristic XRPD peak for form IV (characterized by ° 2 θ)
A DSC profile (fig. 8a) indicates a melting endotherm peak for form IV of 163.8 ℃; the combination of DSC (figure 8a) and TGA (figure 8b) patterns reveals form IV as the 1 hydrate.
Examples of the experiments
SNAC:
Characterization of
SNAC in tablets (developed by norand nord, danish pharmaceutical company) was found to be identical to form I in patent WO 2005107462. Thus, the SNAC herein is prepared by referring to the preparation method of example 1SNAC crystal form I in patent WO 2005107462. The SNACs in the following examples were all prepared by this method.
Form I (PNAC-I) was prepared as in example 1.
Form II (PNAC-II) was prepared as in example 2.
Form III (PNAC-III) was prepared as in example 3.
Form IV (PNAC-IV) was prepared as in example 4.
Experimental example 1 solution stability experiment of different crystal forms of PNAC in aqueous solution
The PNAC crystal form I-IV samples prepared in the above examples and the SNAC crystal form I samples prepared in the above examples were dissolved in purified water, and were placed at 4 ℃ and room temperature, respectively, and the solution change conditions were observed, and the stability differences of different salt forms and different crystal forms in the aqueous solution at a high contrast ratio (500mg/mL) were observed, and the results are shown in table 5 (no difference occurred in the processes of dissolving the sample PNAC crystal form I-IV and placing the solution).
TABLE 5 solubility profiles of different crystal forms of PNAC under different dissolution conditions
As shown in Table 5, after the SNAC is dissolved at 4 ℃, the solution begins to generate floccules after 0.5h, the solution mobility gradually deteriorates along with the lapse of time, and larger white particles are separated out from the solution after 2 h; the SNAC solution is placed for 1h under the condition that the temperature is raised to room temperature, and floccules appear after 2h, and the floccules increase and become turbid. The PNAC I-IV dissolved solution prepared by the method can be placed at 4 ℃ for 1h, and a small amount of filamentous floccules can not appear at the bottom until the solution state changes little within 2 h; a small amount of floc appeared in the solution after standing at room temperature for 2 hours. This indicates that, under the same conditions, the PNAC I-IV of the present application has better solution stability than SNAC at high concentrations (500 mg/mL).
Experimental example 2 Effect of different crystal forms of PNAC on the bioavailability of GLP-1 drugs in beagle dogs
The preparation method of the GLP-1 drug M4 (M4 for short) is disclosed in the patent application WO 2019201328.
The preparation method of the combined oral tablet is shown in the patent WO2012080471 and the preparation method in the example 1. Wherein the delivery agents (SNAC, PNAC-I, PNAC-II) are each bound to M4. The combined oral tablet prepared by combining SNAC with M4 was SNAC + M4. The combined oral tablet prepared by combining PNAC-I and M4 is PNAC-I + M4. The combined oral tablet prepared by combining PNAC-II with M4 is PNAC-II + M4.
In a combination oral tablet: the content of M4 was 10mg, and the content of the delivery agent was 100 mg.
Prescription of intravenous M4: m4 was dissolved in 8mM phosphate buffer pH7.2 to give a final concentration of 1mg/mL M4.
28 beagle dogs, 9-12kg, males were selected and divided into 4 groups, of which the first group was given a single intravenous injection of M4(0.05mg/kg, N ═ 4), the second group was given a single oral administration of SNAC + M4(1 tablet, N ═ 8), the third group was given a single oral administration of PNAC-i + M4(1 tablet, N ═ 8), and the fourth group was given a single oral administration of PNAC-ii + M4(1 tablet, N ═ 8), and the effects of different forms of PNAC on the bioavailability of M4 in beagle dogs were compared.
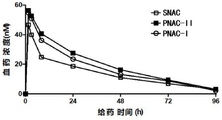
The beagle dogs were fasted without water prohibition one day before the oral administration of the administration group, the tablets were taken with 10ml of water (fixed amount) after the administration of the oral administration group, the dogs were returned to their food intake after 4 hours, blood sampling was performed according to the blood sampling schedule of Table 6 to measure blood concentrations at different time points and to calculate the exposure AUC in the animalslastThe specific results are shown in tables 7, 8 and 9 and FIGS. 9 and 10.
Table 6 shows the blood collection protocol
| Group of
|
Time point of blood sampling
|
| Intravenous M4 group
|
0 hours before administration, 0.5, 1, 3, 6, 12, 24, 48, 72, 96, 144 and 192 hours after administration;
|
| oral administration group
|
0 hour before administration, 1.5, 3, 8, 24, 48, 72 and 96 hours after administration |
Note: at least 500. mu.l plasma at each time point
Table 7 summarizes the blood concentrations at different time points in the group of intravenous administration
TABLE 8 summary of blood concentrations at different time points for each group administered orally
TABLE 9 in vivo Exposure (AUC) in animals for each group of drugslast) Comparison summary table
The absolute bioavailability of the single oral SNAC group was 0.88%, the absolute bioavailability of the single oral PNAC-ii group was 1.28%, the absolute bioavailability of the single oral PNAC-i group was 1.11%, and the bioavailability of the PNAC groups was significantly higher than that of the SNAC group, as calculated from the data in tables 7-9.
Compared with the single oral SNAC group, the absolute bioavailability of the single oral PNAC-II group is improved by 45.5 percent, and the absolute bioavailability of the single oral PNAC-I group is improved by 26.1 percent.
Experimental example 3 influence of different content and different crystal forms of PNAC on bioavailability of GLP-1 drug in beagle dog
The preparation method of the GLP-1 drug M4 (M4 for short) is disclosed in the patent application WO 2019201328.
The preparation method of the combined oral tablet is shown in the patent WO2012080471 and the preparation method in the example 1. Wherein the delivery agents (SNAC, PNAC-I, PNAC-II) are each bound to M4. The combined oral tablet prepared by combining SNAC with M4 was SNAC + M4. The combined oral tablet prepared by combining PNAC-I and M4 is PNAC-I + M4. The combined oral tablet prepared by combining PNAC-II with M4 is PNAC-II + M4.
In the combined oral tablet, each tablet ingredient: the content of M4 was 10mg, and the content of the delivery agent was 300mg and 450mg, respectively.
Prescription of intravenous M4: m4 was dissolved in 8mM phosphate buffer pH7.2 to give a final concentration of 1mg/mL M4.
28 beagle dogs, 9-15kg and males are selected and divided into 7 groups, 4 of each group, and the influence of different crystal forms of PNAC on the bioavailability of M4 in beagle dogs is compared. Wherein the grouping information is as follows in table 10.
The beagle dogs were fasted without water prohibition one day before the oral administration of the administration group, the tablets were taken with 10ml of water (fixed amount) after the administration of the oral administration group, the dogs were returned to their food intake after 4 hours, blood sampling was performed according to the blood sampling schedule of Table 11 to determine blood concentrations at different time points and to calculate the exposure AUC in the animalslastThe results are shown in tables 12 to 14 and FIGS. 19 and 20.
Table 10 is packet information
| Group of
|
Route of administration
|
Dosage to be administered
|
Number of animals (only)
|
| M4
|
i.v.
|
0.05mg/kg
|
4
|
| SNAC(300mg)+M4
|
p.o.
|
300+10mg tablet 1 tablet
|
4
|
| SNAC(450mg)+M4
|
p.o.
|
Tablet 1 tablet of 450+10mg
|
4
|
| PNAC-Ⅰ(300mg)+M4
|
p.o.
|
300+10mg tablet 1 tablet
|
4
|
| PNAC-Ⅰ(450mg)+M4
|
p.o.
|
Tablet 1 tablet of 450+10mg
|
4
|
| PNAC-IⅠ(300mg)+M4
|
p.o.
|
300+10mg tablet 1 tablet
|
4
|
| PNAC-IⅠ(450mg)+M4
|
p.o.
|
Tablet 1 tablet of 450+10mg
|
4 |
TABLE 11 blood sampling protocol
Note: at least 500. mu.l plasma was used at each time point.
TABLE 12 summary of blood concentrations at different time points for the intravenous administration group
TABLE 13 summary of blood concentrations at different time points for each group administered orally
TABLE 14 Exposure of each group of drugs in Animals (AUC)last) Comparison summary table
As can be seen from table 14, the absolute bioavailability of the SNAC (300mg) group for single oral administration is 0.70%, the absolute bioavailability of the SNAC (450mg) group for single oral administration is 0.80%, the absolute bioavailability of the PNAC-ii (300mg) group for single oral administration is 1.20%, the absolute bioavailability of the PNAC-ii (450mg) group for single oral administration is 1.92%, the absolute bioavailability of the PNAC-I (300mg) group for single oral administration is 1.01%, the absolute bioavailability of the PNAC-I (450mg) group for single oral administration is 1.72%, which are all significantly higher than SNAC, and the absolute bioavailability of PNAC increases with the increase of the PNAC content.
Compared with the single oral SNAC (300mg) group, the absolute bioavailability of the single oral PNAC-II (300mg) group is improved by 71 percent, and the absolute bioavailability of the single oral PNAC-I (300mg) group is improved by 44 percent; compared with the single oral SNAC (450mg) group, the absolute bioavailability of the single oral PNAC-II (450mg) group is improved by 140 percent, and the absolute bioavailability of the single oral PNAC-I (450mg) group is improved by 115 percent.
From the perspective of a single drug, compared with the single oral SNAC (450mg) group, the absolute bioavailability of the single oral SNAC (300mg) group is only improved by 14%, and the bioavailability between the two groups is not greatly different, which shows that the content of SNAC in the drug has no effect on the absolute bioavailability. Compared with the single oral PNAC-II (450mg) group, the absolute bioavailability of the single oral PNAC-II (300mg) group is improved by 60 percent; compared with the single oral PNAC-I (450mg) group, the absolute bioavailability of the single oral PNAC-I (300mg) group is improved by 70%. In combination with the absolute bioavailability of the single oral PNAC-ii and PNAC-I (100mg) groups of example 2, it was further verified that the PNAC of the present application showed a significant increase in absolute bioavailability and an unexpected qualitative increase in drug content.
Experimental example 4
Solid state stability experiment of PNAC Crystal form II
Form II prepared in example 2 was divided into three groups, the first group was left at 60 ℃ for 24 hours to obtain solid samples. The second group was left for two weeks at 40 ℃ and 75% relative humidity to obtain solid samples. The third group was left for 2 weeks at 25 ℃ with a relative humidity of 60% to give solid samples, which were then evaluated for physical and chemical stability by XRPD and HPLC tests, respectively.
Here, the HPLC test refers to using the crystalline form II obtained in example 2 as a reference sample, and the purity obtained by performing the HPLC purity test is set as 100 area%, and the relative purity is HPLC purity/reference sample purity. In the HPLC test method, a C18 reversed phase chromatographic column with the column length of 150mm is used, the mobile phase is an acetonitrile-water + 0.05% trifluoroacetic acid system, the flow rate is 1.2ml/min, the sample injection detection is carried out under the condition of the wavelength UV-215nm of a detector, and the purity of the main peak is calculated according to a normalization method.
After the test, the relative purities of HPLC (high performance liquid chromatography) of the reference sample after being placed in different environments for set time are all 100%. The XRPD overlay after the experiment is shown in figure 11, and is completely the same as the characteristic peak of the reference sample, and the crystal form is not changed in the solid state stability experiment.
The same procedure as described above was used to perform the PNAC form I solid state stability experiment and the results are shown in fig. 15.
Experimental example 5
Stability test in preparation Process
Form II obtained in example 2 was divided into three groups, the first group was subjected to manual milling for 10 minutes to obtain solid samples. The second set of tablets was compressed (3 kN pressure) against form II to give solid samples. And the third group performs air flow crushing on the crystal form II (the sample injection pressure is 0.4 MPa, and the crushing pressure is 0.2 MPa) to obtain a solid sample. And XRPD characterization was performed on three groups of solid samples to assess their physical stability. The results are shown in FIGS. 12-14.
From fig. 12-14, it can be seen that the crystal form of the sample did not change after grinding, tabletting and airflow pulverization of the crystal form II.
The stability experiments in the preparation process of the PNAC crystal form I, the PNAC crystal form II, the PNAC crystal form III and the PNAC crystal form IV are carried out by using the same method, and the results are shown in a table 15.
The XRPD characterization results of the PNAC crystal form I are shown in figures 16-18, and as can be seen in figures 16-18, the crystal form of the sample is not changed after grinding, tabletting and airflow crushing of the crystal form I.
Table 15 shows the stability test results of different crystal forms of PNAC
From the results in the above table, it can be seen that PNAC form I and form II are very stable, form I will only crystallize after long standing in an environment of over 60% RH, and form II is very stable under whatever conditions were tested.
While embodiments of the present application have been described above in connection with specific embodiments thereof, the present application is not limited to the above-described embodiments and fields of application, which are intended to be illustrative, instructive, and not restrictive. Those skilled in the art, having the benefit of this disclosure, may effect numerous modifications thereto and changes may be made without departing from the scope of the invention as defined by the appended claims.