CN111663168A - Anode production process for improving anode activity by electrochemical electrode sintering method - Google Patents
Anode production process for improving anode activity by electrochemical electrode sintering method Download PDFInfo
- Publication number
- CN111663168A CN111663168A CN202010687239.6A CN202010687239A CN111663168A CN 111663168 A CN111663168 A CN 111663168A CN 202010687239 A CN202010687239 A CN 202010687239A CN 111663168 A CN111663168 A CN 111663168A
- Authority
- CN
- China
- Prior art keywords
- anode
- titanium plate
- production process
- plate
- activity
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C25—ELECTROLYTIC OR ELECTROPHORETIC PROCESSES; APPARATUS THEREFOR
- C25D—PROCESSES FOR THE ELECTROLYTIC OR ELECTROPHORETIC PRODUCTION OF COATINGS; ELECTROFORMING; APPARATUS THEREFOR
- C25D17/00—Constructional parts, or assemblies thereof, of cells for electrolytic coating
- C25D17/10—Electrodes, e.g. composition, counter electrode
-
- C—CHEMISTRY; METALLURGY
- C02—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F—TREATMENT OF WATER, WASTE WATER, SEWAGE, OR SLUDGE
- C02F1/00—Treatment of water, waste water, or sewage
- C02F1/46—Treatment of water, waste water, or sewage by electrochemical methods
- C02F1/461—Treatment of water, waste water, or sewage by electrochemical methods by electrolysis
- C02F1/467—Treatment of water, waste water, or sewage by electrochemical methods by electrolysis by electrochemical disinfection; by electrooxydation or by electroreduction
-
- C—CHEMISTRY; METALLURGY
- C25—ELECTROLYTIC OR ELECTROPHORETIC PROCESSES; APPARATUS THEREFOR
- C25D—PROCESSES FOR THE ELECTROLYTIC OR ELECTROPHORETIC PRODUCTION OF COATINGS; ELECTROFORMING; APPARATUS THEREFOR
- C25D9/00—Electrolytic coating other than with metals
- C25D9/04—Electrolytic coating other than with metals with inorganic materials
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Electrochemistry (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Metallurgy (AREA)
- Inorganic Chemistry (AREA)
- General Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Hydrology & Water Resources (AREA)
- Environmental & Geological Engineering (AREA)
- Water Supply & Treatment (AREA)
- Electrodes For Compound Or Non-Metal Manufacture (AREA)
Abstract
The invention discloses an anode production process for improving anode activity by an electrochemical electrode sintering method, wherein an anode comprises a titanium plate and an anode coating, and the anode production process comprises the following steps: the method comprises the following steps: taking a complete titanium plate with proper thickness, and cutting the titanium plate by using a cutting device to ensure that the size of the titanium plate meets the requirement; step two: polishing the cut titanium plate, and removing the polished impurities; step three: carrying out degreasing and deoiling treatment on the polished titanium plate; step four: carrying out acid cleaning treatment on the titanium plate after the degreasing and deoiling treatment; step five: and preparing the anode coating solution. The anode production process for improving the anode activity by the electrochemical electrode sintering method has the advantages that the titanium plate is processed more finely, the post-processing of the titanium plate is not influenced, the quality of the produced anode material is better, the activity of the anode material can be improved, and the using effect of the produced anode material is better.
Description
Technical Field
The invention relates to the technical field related to anode production, in particular to an anode production process for improving anode activity by an electrochemical electrode sintering method.
Background
The anode material is a very important material in the electrochemical field, and as a material in electrochemical reaction, the anode material can realize the fields of sterilization, disinfection, electroplating and the like of water, and certain defects still exist in the production process of the anode, such as:
the general anode production process is relatively rough in titanium plate treatment, so that the post-treatment of the titanium plate is influenced, the quality of the produced anode material is poor, the activity of the anode material cannot be improved by the existing anode production process, and the use effect of the produced anode material is poor, so that the anode production process for improving the anode activity by the electrochemical electrode sintering method is provided, and the problems provided in the above are solved.
Disclosure of Invention
The invention aims to provide an anode production process for improving the anode activity by an electrochemical electrode sintering method, which aims to solve the problems that most anode production processes proposed in the background art have rough treatment on a titanium plate, so that the post-treatment of the titanium plate is influenced, the quality of a produced anode material is poor, the activity of the anode material cannot be improved, and the using effect of the produced anode material is poor.
In order to achieve the purpose, the invention provides the following technical scheme: an anode production process for improving anode activity by an electrochemical electrode sintering method, wherein the anode comprises a titanium plate and an anode coating, and the anode production process comprises the following steps:
the method comprises the following steps: selecting a complete titanium plate with proper thickness, and cutting the titanium plate by using a cutting device to ensure that the size of the titanium plate meets the requirement;
step two: polishing the cut titanium plate, and removing the polished impurities;
step three: carrying out degreasing and deoiling treatment on the polished titanium plate;
step four: carrying out acid cleaning treatment on the titanium plate after the degreasing and deoiling treatment;
step five: preparing an anode coating solution;
step six: uniformly coating the anode coating solution on the surface of the titanium plate to form an anode plate;
step seven: placing the anode plate into a drying box, and drying the surface of the anode plate;
step eight: performing film covering treatment on the anode plate film;
step nine: sintering the dried anode plate;
step ten: and cooling the sintered anode plate to obtain a finished anode plate.
Preferably, the polishing mode in the second step includes rough polishing, fine polishing, rough polishing and fine polishing, and the rough polishing, the fine polishing, the rough polishing and the fine polishing are respectively completed by using an abrasive belt, a nylon wheel, a linen wheel matched with green wax, and a cloth wheel matched with white wax.
Preferably, the degreasing and deoiling treatment in the third step mainly uses a mixed solution for soaking and washing, wherein the mixed solution refers to a solution formed by mixing particles prepared from an anionic surfactant, sodium alkyl benzene sulfonate, a small amount of a nonionic surfactant, a plurality of auxiliaries, phosphate, silicate, anhydrous sodium sulphate, a fluorescent agent, an enzyme and the like with water, and distilled water is used for washing the residual solution on the surface of the titanium plate after soaking and washing.
Preferably, the solution used in the acid washing treatment in the fourth step is an oxalic acid solution, the concentration of the oxalic acid solution is 10%, the solution is treated in a digestion mode for 2 hours, and the oxalic acid solution residue on the surface of the titanium plate is removed by using distilled water after the acid washing treatment.
Preferably, the anode coating solution preparation in the fifth step comprises the following steps:
(a) weighing 50-100ml of ethanol, 5-10g of ethyl acetate, 1-10g of chloroiridic acid, 1-10g of ruthenium trichloride, 1-10g of tantalum pentachloride and 1-10g of chloroplatinic acid;
(b) uniformly mixing 50-100ml of ethanol and 5-10g of ethyl acetate, sequentially adding 1-10g of chloroiridic acid, 1-10g of ruthenium trichloride, 1-10g of tantalum pentachloride and 1-10g of chloroplatinic acid, and uniformly stirring to obtain the anode coating solution.
Preferably, in the sixth step, the brush is used for uniformly coating the anode coating solution on the surface of the titanium plate in a small amount of times, and the anode coating solution is continuously coated on the surface of the titanium plate after the upper layer is shaped.
Preferably, the film coating treatment in the step eight includes:
(I): mixing 50-100ml of absolute ethyl alcohol, 50-100ml of water and 1-10ml of precursor, adding HCL to adjust the pH value to 2.0-6.0, and fully stirring;
(II): the anode plate is treated by adopting an electroplating method, and then is dried at the temperature of 40-150 ℃, so that the surface of the anode plate is covered with the silicon dioxide film.
Preferably, the silica membrane has a nanoporous structure.
Preferably, the sintering treatment is performed in the ninth step by means of resistance furnace sintering, wherein the temperature of the resistance furnace is controlled at 500 ℃, and the sintering time is 1-1.5 hours.
Compared with the prior art, the invention has the beneficial effects that: the anode production process for improving the anode activity by the electrochemical electrode sintering method has the advantages that the titanium plate is processed more finely, the post-processing of the titanium plate is not influenced, the quality of the produced anode material is better, the activity of the anode material can be improved, and the using effect of the produced anode material is better;
the silicon dioxide film is arranged on the outer surface of the anode plate and has a nano porous structure, so that the porous structure and the electrochemical activity of the anode coating solution coating which is coated subsequently can be improved.
Drawings
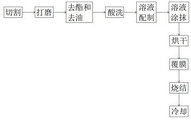
FIG. 1 is a schematic view of the working process of the present invention.
Detailed Description
The technical solutions in the embodiments of the present invention will be clearly and completely described below with reference to the drawings in the embodiments of the present invention, and it is obvious that the described embodiments are only a part of the embodiments of the present invention, and not all embodiments, and all other embodiments obtained by a person of ordinary skill in the art without creative efforts based on the embodiments of the present invention belong to the protection scope of the present invention.
Referring to fig. 1, the present invention provides a technical solution: an anode production process for improving anode activity by an electrochemical electrode sintering method comprises the following steps:
the method comprises the following steps: selecting a complete titanium plate with proper thickness, and cutting the titanium plate by using a cutting device to ensure that the size of the titanium plate meets the requirement;
step two: polishing the cut titanium plate, and removing the polished impurities;
step three: carrying out degreasing and deoiling treatment on the polished titanium plate;
step four: carrying out acid cleaning treatment on the titanium plate after the degreasing and deoiling treatment;
step five: preparing an anode coating solution;
step six: uniformly coating the anode coating solution on the surface of the titanium plate to form an anode plate;
step seven: placing the anode plate into a drying box, and drying the surface of the anode plate;
step eight: performing film covering treatment on the anode plate film;
step nine: sintering the dried anode plate;
step ten: and cooling the sintered anode plate to obtain a finished anode plate.
Furthermore, the polishing mode in the second step comprises rough polishing, fine polishing, rough polishing and fine polishing, and the rough polishing, the fine polishing, the rough polishing and the fine polishing are respectively completed by using an abrasive belt, a nylon wheel, a linen wheel matched with green wax and a cloth wheel matched with white wax.
Furthermore, in the third step, degreasing and deoiling treatment is mainly carried out by soaking and washing with a mixed solution, wherein the mixed solution refers to a solution formed by mixing particles prepared from an anionic surfactant, sodium alkyl benzene sulfonate, a small amount of a nonionic surfactant, a plurality of auxiliaries, phosphate, silicate, anhydrous sodium sulphate, a fluorescent agent, an enzyme and the like with water, and washing with distilled water to remove the residue of the solution on the surface of the titanium plate after soaking and washing.
Furthermore, the solution used in the acid washing treatment in the fourth step is oxalic acid solution, the concentration of the oxalic acid solution is 10%, the solution is treated in a soaking and boiling mode for 2 hours, and the oxalic acid solution residue on the surface of the titanium plate is removed by using distilled water after the acid washing treatment.
Furthermore, the anode coating solution preparation in the fifth step comprises the following steps:
(a) weighing 50-100ml of ethanol, 5-10g of ethyl acetate, 1-10g of chloroiridic acid, 1-10g of ruthenium trichloride, 1-10g of tantalum pentachloride and 1-10g of chloroplatinic acid;
(b) uniformly mixing 50-100ml of ethanol and 5-10g of ethyl acetate, sequentially adding 1-10g of chloroiridic acid, 1-10g of ruthenium trichloride, 1-10g of tantalum pentachloride and 1-10g of chloroplatinic acid, and uniformly stirring to obtain the anode coating solution.
Furthermore, in the sixth step, the anode coating solution is uniformly coated on the surface of the titanium plate by using a brush in a small quantity and multiple times, and the next layer is continuously coated after the former layer is shaped.
Further, the film coating treatment in the step eight includes:
(I): mixing 50-100ml of absolute ethyl alcohol, 50-100ml of water and 1-10ml of precursor, adding HCL to adjust the pH value to 2.0-6.0, and fully stirring;
(II): the anode plate is treated by adopting an electroplating method, and then is dried at the temperature of 40-150 ℃, so that the surface of the anode plate is covered with the silicon dioxide film.
Further, the silica membrane has a nanoporous structure.
Furthermore, in the ninth step, the sintering treatment is carried out by means of resistance furnace sintering, wherein the temperature of the resistance furnace is controlled at 500 ℃, and the sintering time is 1-1.5 hours.
In the embodiments provided in the present application, it should be understood that the disclosed apparatus and method can be implemented in other ways. The apparatus embodiments described above are merely illustrative, and for example, the flowchart and block diagrams in the figures illustrate the architecture, functionality, and operation of possible implementations of apparatus, methods and computer program products according to various embodiments of the present application. In this regard, each block in the flowchart or block diagrams may represent a module, segment, or portion of code, which comprises one or more executable instructions for implementing the specified logical function(s). It should also be noted that, in some alternative implementations, the functions noted in the block may occur out of the order noted in the figures. For example, two blocks shown in succession may, in fact, be executed substantially concurrently, or the blocks may sometimes be executed in the reverse order, depending upon the functionality involved. It will also be noted that each block of the block diagrams and/or flowchart illustration, and combinations of blocks in the block diagrams and/or flowchart illustration, can be implemented by special purpose hardware-based systems which perform the specified functions or acts, or combinations of special purpose hardware and computer instructions.
In addition, functional modules in the embodiments of the present application may be integrated together to form an independent part, or each module may exist separately, or two or more modules may be integrated to form an independent part.
The functions, if implemented in the form of software functional modules and sold or used as a stand-alone product, may be stored in a computer readable storage medium. Based on such understanding, the technical solution of the present application or portions thereof that substantially contribute to the prior art may be embodied in the form of a software product stored in a storage medium and including instructions for causing a computer device (which may be a personal computer, a server, or a network device) to execute all or part of the steps of the method according to the embodiments of the present application. And the aforementioned storage medium includes: various media capable of storing program codes, such as a usb disk, a removable hard disk, a Read-only memory (ROM), a Random Access Memory (RAM), a magnetic disk, or an optical disk. It is noted that, herein, relational terms such as first and second, and the like may be used solely to distinguish one entity or action from another entity or action without necessarily requiring or implying any actual such relationship or order between such entities or actions. Also, the terms "comprises," "comprising," or any other variation thereof, are intended to cover a non-exclusive inclusion, such that a process, method, article, or apparatus that comprises a list of elements does not include only those elements but may include other elements not expressly listed or inherent to such process, method, article, or apparatus. Without further limitation, an element defined by the phrase "comprising an … …" does not exclude the presence of other identical elements in a process, method, article, or apparatus that comprises the element.
The above description is only a preferred embodiment of the present application and is not intended to limit the present application, and various modifications and changes may be made by those skilled in the art. Any modification, equivalent replacement, improvement and the like made within the spirit and principle of the present application shall be included in the protection scope of the present application. It should be noted that: like reference numbers and letters refer to like items in the following figures, and thus, once an item is defined in one figure, it need not be further defined and explained in subsequent figures.
The above description is only for the specific embodiments of the present application, but the scope of the present application is not limited thereto, and any person skilled in the art can easily conceive of the changes or substitutions within the technical scope of the present application, and all the changes or substitutions should be covered by the scope of the present application. Therefore, the protection scope of the present application shall be subject to the protection scope of the claims.
Claims (9)
1. An anode production process for improving anode activity by an electrochemical electrode sintering method is characterized in that: the anode comprises a titanium plate and an anode coating, and the anode production process comprises the following steps:
the method comprises the following steps: selecting a complete titanium plate with proper thickness, and cutting the titanium plate by using a cutting device to ensure that the size of the titanium plate meets the requirement;
step two: polishing the cut titanium plate, and removing the polished impurities;
step three: carrying out degreasing and deoiling treatment on the polished titanium plate;
step four: carrying out acid cleaning treatment on the titanium plate after the degreasing and deoiling treatment;
step five: preparing an anode coating solution;
step six: uniformly coating the anode coating solution on the surface of the titanium plate to form an anode plate;
step seven: placing the anode plate into a drying box, and drying the surface of the anode plate;
step eight: performing film covering treatment on the anode plate film;
step nine: sintering the dried anode plate;
step ten: and cooling the sintered anode plate to obtain a finished anode plate.
2. The anode production process for improving the anode activity by the electrochemical electrode sintering method according to claim 1, is characterized in that: and the grinding mode in the second step comprises rough grinding, fine grinding, rough polishing and fine polishing, and the rough grinding, the fine grinding, the rough polishing and the fine polishing are respectively completed by using an abrasive belt, a nylon wheel, a linen wheel matched with green wax and a cloth wheel matched with white wax.
3. The anode production process for improving the anode activity by the electrochemical electrode sintering method according to claim 1, is characterized in that: in the third step, the degreasing and deoiling treatment is mainly carried out by soaking and washing by using a mixed solution, wherein the mixed solution refers to a solution formed by mixing particles prepared from an anionic surfactant, sodium alkyl benzene sulfonate, a small amount of nonionic surfactant, some additives, phosphate, silicate, anhydrous sodium sulphate, fluorescer, enzyme and the like with water, and the residual of the solution on the surface of the titanium plate is washed by using distilled water after soaking and washing.
4. The anode production process for improving the anode activity by the electrochemical electrode sintering method according to claim 1, is characterized in that: and in the fourth step, the solution used in the acid washing treatment is oxalic acid solution, the concentration of the oxalic acid solution is 10%, the solution is treated for 2 hours in a soaking and boiling mode, and the oxalic acid solution residue on the surface of the titanium plate is removed by using distilled water after the acid washing treatment.
5. The anode production process for improving the anode activity by the electrochemical electrode sintering method according to claim 1, is characterized in that: the preparation of the anode coating solution in the fifth step comprises the following steps:
(a) weighing 50-100ml of ethanol, 5-10g of ethyl acetate, 1-10g of chloroiridic acid, 1-10g of ruthenium trichloride, 1-10g of tantalum pentachloride and 1-10g of chloroplatinic acid;
(b) uniformly mixing 50-100ml of ethanol and 5-10g of ethyl acetate, sequentially adding 1-10g of chloroiridic acid, 1-10g of ruthenium trichloride, 1-10g of tantalum pentachloride and 1-10g of chloroplatinic acid, and uniformly stirring to obtain the anode coating solution.
6. The anode production process for improving the anode activity by the electrochemical electrode sintering method according to claim 1, is characterized in that: and in the sixth step, the anode coating solution is uniformly coated on the surface of the titanium plate by using a brush in a small quantity and multiple times, and the next layer is continuously coated after the former layer is shaped.
7. The anode production process for improving the anode activity by the electrochemical electrode sintering method according to claim 1, is characterized in that: the film covering treatment in the step eight comprises the following steps:
(I): mixing 50-100ml of absolute ethyl alcohol, 50-100ml of water and 1-10ml of precursor, adding HCL to adjust the pH value to 2.0-6.0, and fully stirring;
(II): the anode plate is treated by adopting an electroplating method, and then is dried at the temperature of 40-150 ℃, so that the surface of the anode plate is covered with the silicon dioxide film.
8. The anode production process for improving the anode activity by the electrochemical electrode sintering method according to claim 7, wherein the anode production process comprises the following steps: the silica membrane has a nanoporous structure.
9. The anode production process for improving the anode activity by the electrochemical electrode sintering method according to claim 1, is characterized in that: and in the ninth step, sintering treatment is carried out in a resistance furnace sintering mode, wherein the temperature of the resistance furnace is controlled at 500 ℃, and the sintering time is 1-1.5 hours.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202010687239.6A CN111663168A (en) | 2020-07-16 | 2020-07-16 | Anode production process for improving anode activity by electrochemical electrode sintering method |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202010687239.6A CN111663168A (en) | 2020-07-16 | 2020-07-16 | Anode production process for improving anode activity by electrochemical electrode sintering method |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN111663168A true CN111663168A (en) | 2020-09-15 |
Family
ID=72392503
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202010687239.6A Pending CN111663168A (en) | 2020-07-16 | 2020-07-16 | Anode production process for improving anode activity by electrochemical electrode sintering method |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN111663168A (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2023150386A (en) * | 2022-03-31 | 2023-10-16 | Toto株式会社 | Hypochlorite generation electrode |
| WO2024176956A1 (en) * | 2023-02-24 | 2024-08-29 | パナソニックIpマネジメント株式会社 | Electrolysis electrode, and electrolysis method using said electrode |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102677092A (en) * | 2012-05-30 | 2012-09-19 | 浙江大学 | Preparation method of titanium anode |
| CN106521404A (en) * | 2016-11-02 | 2017-03-22 | 苏州云瑞环境科技有限公司 | Process for manufacturing titanium anode by once oxidation |
| CN106929177A (en) * | 2015-12-31 | 2017-07-07 | 孙良云 | A kind of washing powder for removing obstinate greasy dirt |
| CN107892998A (en) * | 2017-10-29 | 2018-04-10 | 安徽安庆南风日化有限责任公司 | A kind of washing powder of strong dirt-removing stain |
| CN109706513A (en) * | 2019-03-12 | 2019-05-03 | 江阴安诺电极有限公司 | The preparation method of coated anode plate |
| CN109989075A (en) * | 2019-05-10 | 2019-07-09 | 建滔(连州)铜箔有限公司 | A kind of back coating technique producing electrolytic copper foil titanium anode plate |
-
2020
- 2020-07-16 CN CN202010687239.6A patent/CN111663168A/en active Pending
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102677092A (en) * | 2012-05-30 | 2012-09-19 | 浙江大学 | Preparation method of titanium anode |
| CN106929177A (en) * | 2015-12-31 | 2017-07-07 | 孙良云 | A kind of washing powder for removing obstinate greasy dirt |
| CN106521404A (en) * | 2016-11-02 | 2017-03-22 | 苏州云瑞环境科技有限公司 | Process for manufacturing titanium anode by once oxidation |
| CN107892998A (en) * | 2017-10-29 | 2018-04-10 | 安徽安庆南风日化有限责任公司 | A kind of washing powder of strong dirt-removing stain |
| CN109706513A (en) * | 2019-03-12 | 2019-05-03 | 江阴安诺电极有限公司 | The preparation method of coated anode plate |
| CN109989075A (en) * | 2019-05-10 | 2019-07-09 | 建滔(连州)铜箔有限公司 | A kind of back coating technique producing electrolytic copper foil titanium anode plate |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2023150386A (en) * | 2022-03-31 | 2023-10-16 | Toto株式会社 | Hypochlorite generation electrode |
| JP7396391B2 (en) | 2022-03-31 | 2023-12-12 | Toto株式会社 | Hypochlorous acid generation electrode |
| US12188136B2 (en) | 2022-03-31 | 2025-01-07 | Toto Ltd. | Hypochlorous acid generating electrode |
| WO2024176956A1 (en) * | 2023-02-24 | 2024-08-29 | パナソニックIpマネジメント株式会社 | Electrolysis electrode, and electrolysis method using said electrode |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN101235513B (en) | Coating titanium anode | |
| TWI433963B (en) | Electrode for electrolysis cell | |
| DE602005002661T2 (en) | Method for producing a conductive diamond electrode | |
| CN111663168A (en) | Anode production process for improving anode activity by electrochemical electrode sintering method | |
| CN103088363A (en) | A kind of manufacturing method of stainless steel electrolytic electrode with titanium dioxide coating | |
| CA2958328C (en) | Electrode for electrochlorination processes and method of manufacturing thereof | |
| CN102953114A (en) | Stainless steel product and preparation method thereof | |
| CN106563176A (en) | Atomic layer deposition-based preparation method for zinc oxide/carbon nanotube nano-antibacterial coating | |
| CN107195728A (en) | A kind of solar cell is done over again the processing method of piece | |
| KR101131146B1 (en) | Dismutting composition and method for dismutting aluminum alloy using same | |
| US20080203352A1 (en) | Formulation of grinding coolant | |
| JP4990737B2 (en) | Manufacturing method of fine structure | |
| CN110106545B (en) | Nickel-titanium alloy electrochemical polishing solution, surface treatment method and left atrial appendage occluder | |
| JPH01118489A (en) | Method of manufacturing aluminum support for print | |
| KR101073279B1 (en) | Method for pickling molybdenum added ferritic stainless steel | |
| JPS6315349B2 (en) | ||
| CN117552076A (en) | Preparation method of magnesium-aluminum alloy surface porous structure | |
| CN113186589B (en) | Electrochemical surface treatment method for selectively laser melting AlSi10Mg alloy heat treatment product | |
| CN108179452B (en) | Aluminum material without pit defects on surface and preparation method thereof | |
| US2461035A (en) | Electrolytic stainless steel polishing | |
| JP4804657B2 (en) | A descaling method for austenitic stainless steel cold-rolled annealed steel sheets | |
| CN108949033B (en) | Polishing fluid and preparation method thereof | |
| KR20010109413A (en) | Electrolytic Anode of Electrolytic Cell and Method for the Same | |
| CN115595584B (en) | Bright water for polishing zipper, metal natural-color bright zipper and preparation method thereof | |
| JP2007229694A (en) | Surface treatment method of hoden stone-made stone ware |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| RJ01 | Rejection of invention patent application after publication |
Application publication date: 20200915 |
|
| RJ01 | Rejection of invention patent application after publication |