CN111574466A - Novel allyl benzoate compound containing triazole as well as preparation method and application thereof - Google Patents
Novel allyl benzoate compound containing triazole as well as preparation method and application thereof Download PDFInfo
- Publication number
- CN111574466A CN111574466A CN202010504490.4A CN202010504490A CN111574466A CN 111574466 A CN111574466 A CN 111574466A CN 202010504490 A CN202010504490 A CN 202010504490A CN 111574466 A CN111574466 A CN 111574466A
- Authority
- CN
- China
- Prior art keywords
- triazole
- formula
- arh
- phenyl
- reaction
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- -1 allyl benzoate compound Chemical class 0.000 title claims abstract description 141
- 238000002360 preparation method Methods 0.000 title claims abstract description 42
- 150000003852 triazoles Chemical class 0.000 title claims abstract description 38
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims abstract description 25
- 125000001424 substituent group Chemical group 0.000 claims abstract description 20
- 230000000844 anti-bacterial effect Effects 0.000 claims abstract description 13
- 230000005764 inhibitory process Effects 0.000 claims abstract description 12
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 claims abstract description 8
- 229910052739 hydrogen Inorganic materials 0.000 claims abstract description 7
- 239000001257 hydrogen Substances 0.000 claims abstract description 7
- 125000002023 trifluoromethyl group Chemical group FC(F)(F)* 0.000 claims abstract description 7
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 claims abstract description 6
- 125000004178 (C1-C4) alkyl group Chemical group 0.000 claims abstract description 5
- 241000221696 Sclerotinia sclerotiorum Species 0.000 claims abstract description 5
- 229910052736 halogen Inorganic materials 0.000 claims abstract description 5
- 150000002367 halogens Chemical class 0.000 claims abstract description 5
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 claims abstract description 5
- WPYMKLBDIGXBTP-UHFFFAOYSA-N Benzoic acid Natural products OC(=O)C1=CC=CC=C1 WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 claims description 56
- 238000006243 chemical reaction Methods 0.000 claims description 47
- 239000005711 Benzoic acid Substances 0.000 claims description 30
- 235000010233 benzoic acid Nutrition 0.000 claims description 30
- 238000000034 method Methods 0.000 claims description 26
- 239000002904 solvent Substances 0.000 claims description 25
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 claims description 24
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 22
- 239000003960 organic solvent Substances 0.000 claims description 20
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 claims description 18
- 238000001914 filtration Methods 0.000 claims description 14
- 238000005406 washing Methods 0.000 claims description 14
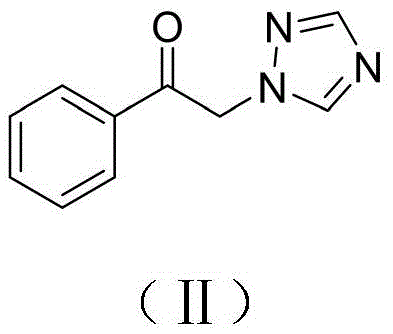
- RXGNQUOYJPSWRW-UHFFFAOYSA-N 1-phenyl-2-(1,2,4-triazol-1-yl)ethanone Chemical compound C=1C=CC=CC=1C(=O)CN1C=NC=N1 RXGNQUOYJPSWRW-UHFFFAOYSA-N 0.000 claims description 12
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 claims description 12
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 claims description 12
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 claims description 12
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 claims description 12
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 claims description 12
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 claims description 12
- 239000003153 chemical reaction reagent Substances 0.000 claims description 12
- 238000001035 drying Methods 0.000 claims description 12
- 239000000706 filtrate Substances 0.000 claims description 12
- 238000004440 column chromatography Methods 0.000 claims description 9
- VHYFNPMBLIVWCW-UHFFFAOYSA-N 4-Dimethylaminopyridine Chemical compound CN(C)C1=CC=NC=C1 VHYFNPMBLIVWCW-UHFFFAOYSA-N 0.000 claims description 8
- LYJHVEDILOKZCG-UHFFFAOYSA-N Allyl benzoate Chemical class C=CCOC(=O)C1=CC=CC=C1 LYJHVEDILOKZCG-UHFFFAOYSA-N 0.000 claims description 8
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 claims description 8
- NQRYJNQNLNOLGT-UHFFFAOYSA-N Piperidine Chemical compound C1CCNCC1 NQRYJNQNLNOLGT-UHFFFAOYSA-N 0.000 claims description 8
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical class [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 claims description 8
- 239000012074 organic phase Substances 0.000 claims description 8
- LIGACIXOYTUXAW-UHFFFAOYSA-N phenacyl bromide Chemical compound BrCC(=O)C1=CC=CC=C1 LIGACIXOYTUXAW-UHFFFAOYSA-N 0.000 claims description 8
- 238000000926 separation method Methods 0.000 claims description 8
- 238000003756 stirring Methods 0.000 claims description 8
- PEUPUKDBCPLDIH-UHFFFAOYSA-N 1,2,4-triazole Chemical group C1=NC=N[N]1 PEUPUKDBCPLDIH-UHFFFAOYSA-N 0.000 claims description 7
- NSPMIYGKQJPBQR-UHFFFAOYSA-N 4H-1,2,4-triazole Chemical compound C=1N=CNN=1 NSPMIYGKQJPBQR-UHFFFAOYSA-N 0.000 claims description 7
- 239000003899 bactericide agent Substances 0.000 claims description 7
- 238000001816 cooling Methods 0.000 claims description 7
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 claims description 6
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 claims description 6
- HUMNYLRZRPPJDN-UHFFFAOYSA-N benzaldehyde Chemical compound O=CC1=CC=CC=C1 HUMNYLRZRPPJDN-UHFFFAOYSA-N 0.000 claims description 6
- 239000012141 concentrate Substances 0.000 claims description 6
- 239000003208 petroleum Substances 0.000 claims description 6
- 229910000033 sodium borohydride Inorganic materials 0.000 claims description 6
- 239000012279 sodium borohydride Substances 0.000 claims description 6
- ILWRPSCZWQJDMK-UHFFFAOYSA-N triethylazanium;chloride Chemical compound Cl.CCN(CC)CC ILWRPSCZWQJDMK-UHFFFAOYSA-N 0.000 claims description 6
- AMSRANWZOYZEKJ-LFIBNONCSA-N (E)-1,3-diphenyl-2-(1,2,4-triazol-1-yl)prop-2-en-1-one Chemical compound C1=CC=C(C=C1)/C=C(\C(=O)C2=CC=CC=C2)/N3C=NC=N3 AMSRANWZOYZEKJ-LFIBNONCSA-N 0.000 claims description 5
- BEOBZEOPTQQELP-UHFFFAOYSA-N 4-(trifluoromethyl)benzaldehyde Chemical compound FC(F)(F)C1=CC=C(C=O)C=C1 BEOBZEOPTQQELP-UHFFFAOYSA-N 0.000 claims description 5
- 239000003480 eluent Substances 0.000 claims description 5
- 239000000417 fungicide Substances 0.000 claims description 5
- 239000000203 mixture Substances 0.000 claims description 5
- 238000000746 purification Methods 0.000 claims description 5
- 229960000549 4-dimethylaminophenol Drugs 0.000 claims description 4
- 229920002582 Polyethylene Glycol 600 Polymers 0.000 claims description 4
- 150000001558 benzoic acid derivatives Chemical class 0.000 claims description 4
- 239000012295 chemical reaction liquid Substances 0.000 claims description 4
- 238000010992 reflux Methods 0.000 claims description 4
- QNGNSVIICDLXHT-UHFFFAOYSA-N para-ethylbenzaldehyde Natural products CCC1=CC=C(C=O)C=C1 QNGNSVIICDLXHT-UHFFFAOYSA-N 0.000 claims description 3
- 125000004201 2,4-dichlorophenyl group Chemical group [H]C1=C([H])C(*)=C(Cl)C([H])=C1Cl 0.000 claims description 2
- 125000004182 2-chlorophenyl group Chemical group [H]C1=C([H])C(Cl)=C(*)C([H])=C1[H] 0.000 claims description 2
- 125000004198 2-fluorophenyl group Chemical group [H]C1=C([H])C(F)=C(*)C([H])=C1[H] 0.000 claims description 2
- 125000004179 3-chlorophenyl group Chemical group [H]C1=C([H])C(*)=C([H])C(Cl)=C1[H] 0.000 claims description 2
- 125000004180 3-fluorophenyl group Chemical group [H]C1=C([H])C(*)=C([H])C(F)=C1[H] 0.000 claims description 2
- 125000004207 3-methoxyphenyl group Chemical group [H]C1=C([H])C(*)=C([H])C(OC([H])([H])[H])=C1[H] 0.000 claims description 2
- 125000001255 4-fluorophenyl group Chemical group [H]C1=C([H])C(*)=C([H])C([H])=C1F 0.000 claims description 2
- 125000004172 4-methoxyphenyl group Chemical group [H]C1=C([H])C(OC([H])([H])[H])=C([H])C([H])=C1* 0.000 claims description 2
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 claims description 2
- 125000003158 alcohol group Chemical group 0.000 claims description 2
- 238000010438 heat treatment Methods 0.000 claims description 2
- 150000002576 ketones Chemical class 0.000 claims description 2
- 125000000040 m-tolyl group Chemical group [H]C1=C([H])C(*)=C([H])C(=C1[H])C([H])([H])[H] 0.000 claims description 2
- 125000003261 o-tolyl group Chemical group [H]C1=C([H])C(*)=C(C([H])=C1[H])C([H])([H])[H] 0.000 claims description 2
- 125000001037 p-tolyl group Chemical group [H]C1=C([H])C(=C([H])C([H])=C1*)C([H])([H])[H] 0.000 claims description 2
- 238000004519 manufacturing process Methods 0.000 claims 1
- 150000001875 compounds Chemical class 0.000 abstract description 45
- 239000000575 pesticide Substances 0.000 abstract description 3
- 241000894006 Bacteria Species 0.000 abstract description 2
- 238000012827 research and development Methods 0.000 abstract 1
- 125000003626 1,2,4-triazol-1-yl group Chemical group [*]N1N=C([H])N=C1[H] 0.000 description 55
- HEDRZPFGACZZDS-MICDWDOJSA-N Trichloro(2H)methane Chemical compound [2H]C(Cl)(Cl)Cl HEDRZPFGACZZDS-MICDWDOJSA-N 0.000 description 52
- 238000005160 1H NMR spectroscopy Methods 0.000 description 26
- 230000015572 biosynthetic process Effects 0.000 description 10
- 238000003786 synthesis reaction Methods 0.000 description 10
- ODSNCZIFTQVJLF-UHFFFAOYSA-N prop-2-enyl 3-nitrobenzoate Chemical compound [O-][N+](=O)C1=CC=CC(C(=O)OCC=C)=C1 ODSNCZIFTQVJLF-UHFFFAOYSA-N 0.000 description 9
- JUZUVYLOVZGXAN-UHFFFAOYSA-N prop-2-enyl 4-methoxybenzoate Chemical compound COC1=CC=C(C(=O)OCC=C)C=C1 JUZUVYLOVZGXAN-UHFFFAOYSA-N 0.000 description 8
- 229940045860 white wax Drugs 0.000 description 8
- 239000000243 solution Substances 0.000 description 7
- 230000001580 bacterial effect Effects 0.000 description 6
- 239000003814 drug Substances 0.000 description 6
- 239000000460 chlorine Substances 0.000 description 5
- ZJVOQVIQHLGYNO-LFIBNONCSA-N (E)-1,3-diphenyl-2-(1,2,4-triazol-1-yl)prop-2-en-1-ol Chemical compound C1=CC=C(C=C1)/C=C(\C(C2=CC=CC=C2)O)/N3C=NC=N3 ZJVOQVIQHLGYNO-LFIBNONCSA-N 0.000 description 4
- YTRZIGHLUAGZLF-MHWRWJLKSA-N (E)-1-phenyl-2-(1,2,4-triazol-1-yl)-3-[4-(trifluoromethyl)phenyl]prop-2-en-1-ol Chemical compound C1=CC=C(C=C1)C(/C(=C\C2=CC=C(C=C2)C(F)(F)F)/N3C=NC=N3)O YTRZIGHLUAGZLF-MHWRWJLKSA-N 0.000 description 4
- GRUJFKXNBSECHW-MHWRWJLKSA-N (E)-1-phenyl-2-(1,2,4-triazol-1-yl)-3-[4-(trifluoromethyl)phenyl]prop-2-en-1-one Chemical compound C1=CC=C(C=C1)C(=O)/C(=C\C2=CC=C(C=C2)C(F)(F)F)/N3C=NC=N3 GRUJFKXNBSECHW-MHWRWJLKSA-N 0.000 description 4
- ATCRIUVQKHMXSH-UHFFFAOYSA-N 2,4-dichlorobenzoic acid Chemical compound OC(=O)C1=CC=C(Cl)C=C1Cl ATCRIUVQKHMXSH-UHFFFAOYSA-N 0.000 description 4
- ZEYHEAKUIGZSGI-UHFFFAOYSA-N 4-methoxybenzoic acid Chemical compound COC1=CC=C(C(O)=O)C=C1 ZEYHEAKUIGZSGI-UHFFFAOYSA-N 0.000 description 4
- 230000012010 growth Effects 0.000 description 4
- LULAYUGMBFYYEX-UHFFFAOYSA-N metachloroperbenzoic acid Natural products OC(=O)C1=CC=CC(Cl)=C1 LULAYUGMBFYYEX-UHFFFAOYSA-N 0.000 description 4
- ZWLPBLYKEWSWPD-UHFFFAOYSA-N o-toluic acid Chemical compound CC1=CC=CC=C1C(O)=O ZWLPBLYKEWSWPD-UHFFFAOYSA-N 0.000 description 4
- YNWVFADWVLCOPU-MDWZMJQESA-N (1E)-1-(4-chlorophenyl)-4,4-dimethyl-2-(1H-1,2,4-triazol-1-yl)pent-1-en-3-ol Chemical group C1=NC=NN1/C(C(O)C(C)(C)C)=C/C1=CC=C(Cl)C=C1 YNWVFADWVLCOPU-MDWZMJQESA-N 0.000 description 3
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 3
- 239000005788 Fluxapyroxad Substances 0.000 description 3
- 241000233616 Phytophthora capsici Species 0.000 description 3
- 241000209140 Triticum Species 0.000 description 3
- 235000021307 Triticum Nutrition 0.000 description 3
- 230000000694 effects Effects 0.000 description 3
- SXSGXWCSHSVPGB-UHFFFAOYSA-N fluxapyroxad Chemical compound FC(F)C1=NN(C)C=C1C(=O)NC1=CC=CC=C1C1=CC(F)=C(F)C(F)=C1 SXSGXWCSHSVPGB-UHFFFAOYSA-N 0.000 description 3
- 239000007788 liquid Substances 0.000 description 3
- 230000002265 prevention Effects 0.000 description 3
- QAPRPPQBBCRDKG-UHFFFAOYSA-N prop-2-enyl 2-chlorobenzoate Chemical compound ClC1=CC=CC=C1C(=O)OCC=C QAPRPPQBBCRDKG-UHFFFAOYSA-N 0.000 description 3
- WJTFPKSSYSADRW-UHFFFAOYSA-N prop-2-enyl 3-methylbenzoate Chemical compound CC1=CC=CC(C(=O)OCC=C)=C1 WJTFPKSSYSADRW-UHFFFAOYSA-N 0.000 description 3
- GGJIQHHMXJIVPF-UHFFFAOYSA-N prop-2-enyl 4-methylbenzoate Chemical compound CC1=CC=C(C(=O)OCC=C)C=C1 GGJIQHHMXJIVPF-UHFFFAOYSA-N 0.000 description 3
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 3
- RMOGWMIKYWRTKW-UONOGXRCSA-N (S,S)-paclobutrazol Chemical compound C([C@@H]([C@@H](O)C(C)(C)C)N1N=CN=C1)C1=CC=C(Cl)C=C1 RMOGWMIKYWRTKW-UONOGXRCSA-N 0.000 description 2
- QAOJBHRZQQDFHA-UHFFFAOYSA-N 2,3-dichlorobenzoic acid Chemical compound OC(=O)C1=CC=CC(Cl)=C1Cl QAOJBHRZQQDFHA-UHFFFAOYSA-N 0.000 description 2
- NSTREUWFTAOOKS-UHFFFAOYSA-N 2-fluorobenzoic acid Chemical compound OC(=O)C1=CC=CC=C1F NSTREUWFTAOOKS-UHFFFAOYSA-N 0.000 description 2
- XHQZJYCNDZAGLW-UHFFFAOYSA-N 3-methoxybenzoic acid Chemical compound COC1=CC=CC(C(O)=O)=C1 XHQZJYCNDZAGLW-UHFFFAOYSA-N 0.000 description 2
- AFPHTEQTJZKQAQ-UHFFFAOYSA-N 3-nitrobenzoic acid Chemical compound OC(=O)C1=CC=CC([N+]([O-])=O)=C1 AFPHTEQTJZKQAQ-UHFFFAOYSA-N 0.000 description 2
- ATZHGRNFEFVDDJ-UHFFFAOYSA-N 4-propylbenzoic acid Chemical compound CCCC1=CC=C(C(O)=O)C=C1 ATZHGRNFEFVDDJ-UHFFFAOYSA-N 0.000 description 2
- KDVYCTOWXSLNNI-UHFFFAOYSA-N 4-t-Butylbenzoic acid Chemical compound CC(C)(C)C1=CC=C(C(O)=O)C=C1 KDVYCTOWXSLNNI-UHFFFAOYSA-N 0.000 description 2
- 241001530056 Athelia rolfsii Species 0.000 description 2
- 240000008067 Cucumis sativus Species 0.000 description 2
- 235000010799 Cucumis sativus var sativus Nutrition 0.000 description 2
- 241000223221 Fusarium oxysporum Species 0.000 description 2
- 240000007594 Oryza sativa Species 0.000 description 2
- 235000007164 Oryza sativa Nutrition 0.000 description 2
- 239000005985 Paclobutrazol Substances 0.000 description 2
- 241000233622 Phytophthora infestans Species 0.000 description 2
- 239000005822 Propiconazole Substances 0.000 description 2
- 241000813090 Rhizoctonia solani Species 0.000 description 2
- 241001558929 Sclerotium <basidiomycota> Species 0.000 description 2
- 230000004071 biological effect Effects 0.000 description 2
- 238000004364 calculation method Methods 0.000 description 2
- 239000003054 catalyst Substances 0.000 description 2
- 230000001276 controlling effect Effects 0.000 description 2
- 201000010099 disease Diseases 0.000 description 2
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 2
- 238000000605 extraction Methods 0.000 description 2
- 230000000855 fungicidal effect Effects 0.000 description 2
- 238000002386 leaching Methods 0.000 description 2
- 231100000053 low toxicity Toxicity 0.000 description 2
- GPSDUZXPYCFOSQ-UHFFFAOYSA-N m-toluic acid Chemical compound CC1=CC=CC(C(O)=O)=C1 GPSDUZXPYCFOSQ-UHFFFAOYSA-N 0.000 description 2
- LPNBBFKOUUSUDB-UHFFFAOYSA-N p-toluic acid Chemical compound CC1=CC=C(C(O)=O)C=C1 LPNBBFKOUUSUDB-UHFFFAOYSA-N 0.000 description 2
- STJLVHWMYQXCPB-UHFFFAOYSA-N propiconazole Chemical compound O1C(CCC)COC1(C=1C(=CC(Cl)=CC=1)Cl)CN1N=CN=C1 STJLVHWMYQXCPB-UHFFFAOYSA-N 0.000 description 2
- 235000009566 rice Nutrition 0.000 description 2
- HPALAKNZSZLMCH-UHFFFAOYSA-M sodium;chloride;hydrate Chemical class O.[Na+].[Cl-] HPALAKNZSZLMCH-UHFFFAOYSA-M 0.000 description 2
- WURBVZBTWMNKQT-UHFFFAOYSA-N 1-(4-chlorophenoxy)-3,3-dimethyl-1-(1,2,4-triazol-1-yl)butan-2-one Chemical compound C1=NC=NN1C(C(=O)C(C)(C)C)OC1=CC=C(Cl)C=C1 WURBVZBTWMNKQT-UHFFFAOYSA-N 0.000 description 1
- MRUDNSFOFOQZDA-UHFFFAOYSA-N 2,6-dichlorobenzoic acid Chemical compound OC(=O)C1=C(Cl)C=CC=C1Cl MRUDNSFOFOQZDA-UHFFFAOYSA-N 0.000 description 1
- IKCLCGXPQILATA-UHFFFAOYSA-N 2-chlorobenzoic acid Chemical compound OC(=O)C1=CC=CC=C1Cl IKCLCGXPQILATA-UHFFFAOYSA-N 0.000 description 1
- MXNBDFWNYRNIBH-UHFFFAOYSA-N 3-fluorobenzoic acid Chemical compound OC(=O)C1=CC=CC(F)=C1 MXNBDFWNYRNIBH-UHFFFAOYSA-N 0.000 description 1
- BBYDXOIZLAWGSL-UHFFFAOYSA-N 4-fluorobenzoic acid Chemical compound OC(=O)C1=CC=C(F)C=C1 BBYDXOIZLAWGSL-UHFFFAOYSA-N 0.000 description 1
- 241000213004 Alternaria solani Species 0.000 description 1
- 235000017060 Arachis glabrata Nutrition 0.000 description 1
- 244000105624 Arachis hypogaea Species 0.000 description 1
- 235000010777 Arachis hypogaea Nutrition 0.000 description 1
- 235000018262 Arachis monticola Nutrition 0.000 description 1
- 241000123650 Botrytis cinerea Species 0.000 description 1
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 1
- 229910020323 ClF3 Inorganic materials 0.000 description 1
- 241000233866 Fungi Species 0.000 description 1
- 241000223195 Fusarium graminearum Species 0.000 description 1
- 235000007688 Lycopersicon esculentum Nutrition 0.000 description 1
- 208000031888 Mycoses Diseases 0.000 description 1
- 241000315044 Passalora arachidicola Species 0.000 description 1
- 206010039509 Scab Diseases 0.000 description 1
- 240000003768 Solanum lycopersicum Species 0.000 description 1
- 239000005846 Triadimenol Substances 0.000 description 1
- 230000000843 anti-fungal effect Effects 0.000 description 1
- 229940121375 antifungal agent Drugs 0.000 description 1
- 125000003118 aryl group Chemical group 0.000 description 1
- 230000003385 bacteriostatic effect Effects 0.000 description 1
- 238000010009 beating Methods 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- 239000011203 carbon fibre reinforced carbon Chemical group 0.000 description 1
- 229910052801 chlorine Inorganic materials 0.000 description 1
- 229940079593 drug Drugs 0.000 description 1
- 230000008686 ergosterol biosynthesis Effects 0.000 description 1
- 125000004185 ester group Chemical group 0.000 description 1
- 239000001963 growth medium Substances 0.000 description 1
- 150000002431 hydrogen Chemical class 0.000 description 1
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 1
- 239000003112 inhibitor Substances 0.000 description 1
- 230000002401 inhibitory effect Effects 0.000 description 1
- 239000011159 matrix material Substances 0.000 description 1
- 239000011259 mixed solution Substances 0.000 description 1
- 239000012452 mother liquor Substances 0.000 description 1
- 229910017464 nitrogen compound Inorganic materials 0.000 description 1
- 235000020232 peanut Nutrition 0.000 description 1
- 238000003359 percent control normalization Methods 0.000 description 1
- 230000008635 plant growth Effects 0.000 description 1
- 229920000136 polysorbate Polymers 0.000 description 1
- 239000011541 reaction mixture Substances 0.000 description 1
- 230000001105 regulatory effect Effects 0.000 description 1
- 239000011550 stock solution Substances 0.000 description 1
- 238000010998 test method Methods 0.000 description 1
- BAZVSMNPJJMILC-UHFFFAOYSA-N triadimenol Chemical compound C1=NC=NN1C(C(O)C(C)(C)C)OC1=CC=C(Cl)C=C1 BAZVSMNPJJMILC-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D249/00—Heterocyclic compounds containing five-membered rings having three nitrogen atoms as the only ring hetero atoms
- C07D249/02—Heterocyclic compounds containing five-membered rings having three nitrogen atoms as the only ring hetero atoms not condensed with other rings
- C07D249/08—1,2,4-Triazoles; Hydrogenated 1,2,4-triazoles
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/64—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with three nitrogen atoms as the only ring hetero atoms
- A01N43/647—Triazoles; Hydrogenated triazoles
- A01N43/653—1,2,4-Triazoles; Hydrogenated 1,2,4-triazoles
Abstract
Description
Claims (9)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202010504490.4A CN111574466B (en) | 2020-06-05 | 2020-06-05 | Allyl benzoate compound containing triazole as well as preparation method and application thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202010504490.4A CN111574466B (en) | 2020-06-05 | 2020-06-05 | Allyl benzoate compound containing triazole as well as preparation method and application thereof |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN111574466A true CN111574466A (en) | 2020-08-25 |
| CN111574466B CN111574466B (en) | 2021-08-03 |
Family
ID=72122276
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202010504490.4A Active CN111574466B (en) | 2020-06-05 | 2020-06-05 | Allyl benzoate compound containing triazole as well as preparation method and application thereof |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN111574466B (en) |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS59186964A (en) * | 1983-04-05 | 1984-10-23 | Sumitomo Chem Co Ltd | Production of alcoholic compound |
| CN101445488A (en) * | 2007-11-27 | 2009-06-03 | 西南大学 | Triazoles compounds with antimicrobial activity and preparation method and pharmaceutical use thereof |
| CN104370892A (en) * | 2014-10-27 | 2015-02-25 | 湖南大学 | 1-(7-methoxybenzofuran-5-yl)-3-(2-methoxyphenyl)-2-(1,2,4-triazole-1-yl) allyl alcohol |
| CN104557888A (en) * | 2015-01-23 | 2015-04-29 | 邵阳学院 | Application of furan phenol allyl alcohol derivative used as herbicide |
-
2020
- 2020-06-05 CN CN202010504490.4A patent/CN111574466B/en active Active
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS59186964A (en) * | 1983-04-05 | 1984-10-23 | Sumitomo Chem Co Ltd | Production of alcoholic compound |
| CN101445488A (en) * | 2007-11-27 | 2009-06-03 | 西南大学 | Triazoles compounds with antimicrobial activity and preparation method and pharmaceutical use thereof |
| CN104370892A (en) * | 2014-10-27 | 2015-02-25 | 湖南大学 | 1-(7-methoxybenzofuran-5-yl)-3-(2-methoxyphenyl)-2-(1,2,4-triazole-1-yl) allyl alcohol |
| CN104557888A (en) * | 2015-01-23 | 2015-04-29 | 邵阳学院 | Application of furan phenol allyl alcohol derivative used as herbicide |
Non-Patent Citations (2)
| Title |
|---|
| YONG KI MIN,等: "New Lead Compounds for Brassinosteroid Biosynthesis Inhibitors", 《BIOORGANIC & MEDICINAL CHEMISTRY LETTERS》 * |
| 李春芳,等: "新型含氟三唑类化合物的合成及生物活性研究", 《青岛科技大学学报( 自然科学版)》 * |
Also Published As
| Publication number | Publication date |
|---|---|
| CN111574466B (en) | 2021-08-03 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CA3052898A1 (en) | Composition for controlling harmful microorganisms comprising 1 -(phenoxy-pyridinyl)-2-(1,2,4-triazol-1 -yl)-ethanol derivatives | |
| JPH0625140B2 (en) | Novel azole derivative, method for producing the same and agricultural / horticultural drug of the derivative | |
| DK157133B (en) | FUNGICID PLANT PROTECTANT CONTAINING AN N, N-DISUBSTITUTED 3-AMINO-TETRAHYDRO-2-FURANON DERIVATIVE | |
| WO2020108518A1 (en) | N-(1, 3, 4-oxadiazole-2-yl)aryl formamides or salt thereof, preparation method therefor, herbicidal composition and use thereof | |
| JP2010529971A (en) | New microbicide | |
| JPH0356466A (en) | Pyridazinones having insecticide and tickicide activity | |
| JP2704655B2 (en) | Tri-substituted phenylpyrazole derivatives or their salts and herbicides | |
| CN112979627A (en) | Pyrazole bi-1, 2, 4-oxadiazole substituted benzamide compounds and preparation method and application thereof | |
| JP2000226374A (en) | Pyrazolinone derivative | |
| WO2017050275A1 (en) | Oxazolines insecticides and acaricides as well as preparation method and application therefor | |
| JPS626548B2 (en) | ||
| CN110627692B (en) | Paeonol benzenesulfonylhydrazone and derivative thereof, preparation method of paeonol benzenesulfonylhydrazone and derivative thereof, botanical insecticide and application of botanical insecticide | |
| CN111574466B (en) | Allyl benzoate compound containing triazole as well as preparation method and application thereof | |
| CN108069915B (en) | Pyrazinamide compound, preparation method and application thereof, and bactericide | |
| CN110963973B (en) | Triketone compound containing quinazoline diketone fragment, preparation method and application thereof, and herbicide | |
| JP2613651B2 (en) | Oxa or thiazoline compound and insecticide and acaricide containing the same | |
| CN115260175A (en) | 5- (pyrazole-5-yl) -1,2,4-oxadiazole substituted benzamide compound and preparation method and application thereof | |
| CN108383790B (en) | Amide compound containing pyrazole ring, preparation method and application thereof | |
| CN112209894B (en) | 5-aryl substituted 2-aminobenzoxazole derivative, preparation method and application thereof | |
| WO1992009581A1 (en) | Benzohydroximoylazole derivative, production thereof and insecticide | |
| JP2782463B2 (en) | 4-Substituted phenyl-1,2,4-triazolin-5-one derivatives and fungicides for agricultural and horticultural use | |
| CN114213311A (en) | Substituted benzaldehyde oxime ester compound and preparation method and application thereof | |
| EP0101288A2 (en) | Aryl(aryloxy or arylthio)azolomethanes, their preparation and their use | |
| CN111620827B (en) | Allyl ether compound containing triazole and preparation method and application thereof | |
| CN113234066A (en) | Propiconazole-like compound and preparation method and application thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant | ||
| EE01 | Entry into force of recordation of patent licensing contract |
Application publication date: 20200825 Assignee: Wenzhou Jinhua International Trade Co.,Ltd. Assignor: JIANG University OF TECHNOLOGY Contract record no.: X2023330000144 Denomination of invention: An allyl benzoate compound containing triazole and its preparation method and application Granted publication date: 20210803 License type: Common License Record date: 20230311 |
|
| EE01 | Entry into force of recordation of patent licensing contract | ||
| EE01 | Entry into force of recordation of patent licensing contract | ||
| EE01 | Entry into force of recordation of patent licensing contract |
Application publication date: 20200825 Assignee: Hubei Zhiwei Electric Power Engineering Construction Co.,Ltd. Assignor: JIANG University OF TECHNOLOGY Contract record no.: X2023980035930 Denomination of invention: A triazole containing allyl group benzoate compound and its preparation method and application Granted publication date: 20210803 License type: Common License Record date: 20230525 |
|
| EE01 | Entry into force of recordation of patent licensing contract | ||
| EE01 | Entry into force of recordation of patent licensing contract |
Application publication date: 20200825 Assignee: Guangzhou Fangshao Technology Co.,Ltd. Assignor: JIANG University OF TECHNOLOGY Contract record no.: X2023980036218 Denomination of invention: A triazole containing Allyl group benzoate compound and its preparation method and application Granted publication date: 20210803 License type: Common License Record date: 20230602 |
|
| EE01 | Entry into force of recordation of patent licensing contract | ||
| EE01 | Entry into force of recordation of patent licensing contract |
Application publication date: 20200825 Assignee: Linhai Hongkang Lighting Co.,Ltd. Assignor: JIANG University OF TECHNOLOGY Contract record no.: X2023980047308 Denomination of invention: A triazole containing allyl benzoate compound and its preparation method and application Granted publication date: 20210803 License type: Common License Record date: 20231116 |