CN111116632B - Amino film forming inhibitor for drilling fluid and preparation method thereof - Google Patents
Amino film forming inhibitor for drilling fluid and preparation method thereof Download PDFInfo
- Publication number
- CN111116632B CN111116632B CN201811293348.9A CN201811293348A CN111116632B CN 111116632 B CN111116632 B CN 111116632B CN 201811293348 A CN201811293348 A CN 201811293348A CN 111116632 B CN111116632 B CN 111116632B
- Authority
- CN
- China
- Prior art keywords
- amine
- inhibitor
- drilling fluid
- clay
- amine compound
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 239000003112 inhibitor Substances 0.000 title claims abstract description 83
- 238000005553 drilling Methods 0.000 title claims abstract description 39
- 239000012530 fluid Substances 0.000 title claims abstract description 39
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 title claims abstract description 31
- 238000002360 preparation method Methods 0.000 title abstract description 10
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 27
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 claims description 26
- -1 amine compound Chemical class 0.000 claims description 25
- 150000001412 amines Chemical class 0.000 claims description 22
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 claims description 21
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 claims description 18
- 238000006243 chemical reaction Methods 0.000 claims description 16
- 230000015572 biosynthetic process Effects 0.000 claims description 13
- 239000004593 Epoxy Substances 0.000 claims description 10
- 239000003795 chemical substances by application Substances 0.000 claims description 10
- 239000003054 catalyst Substances 0.000 claims description 9
- 238000001914 filtration Methods 0.000 claims description 9
- 229910052783 alkali metal Inorganic materials 0.000 claims description 8
- 150000001340 alkali metals Chemical class 0.000 claims description 8
- 238000002156 mixing Methods 0.000 claims description 8
- 239000002904 solvent Substances 0.000 claims description 8
- 150000003961 organosilicon compounds Chemical class 0.000 claims description 7
- 238000000034 method Methods 0.000 claims description 5
- 239000011261 inert gas Substances 0.000 claims description 3
- 239000002516 radical scavenger Substances 0.000 claims description 2
- 230000035484 reaction time Effects 0.000 claims description 2
- 238000004519 manufacturing process Methods 0.000 claims 4
- 239000004927 clay Substances 0.000 abstract description 48
- 230000036571 hydration Effects 0.000 abstract description 21
- 238000006703 hydration reaction Methods 0.000 abstract description 21
- 230000002401 inhibitory effect Effects 0.000 abstract description 17
- 239000006185 dispersion Substances 0.000 abstract description 7
- 238000001179 sorption measurement Methods 0.000 description 27
- 239000010410 layer Substances 0.000 description 12
- 230000000694 effects Effects 0.000 description 10
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 10
- 229920000768 polyamine Polymers 0.000 description 10
- 238000003756 stirring Methods 0.000 description 10
- 239000000126 substance Substances 0.000 description 10
- 239000011435 rock Substances 0.000 description 8
- 229910052710 silicon Inorganic materials 0.000 description 8
- WCUXLLCKKVVCTQ-UHFFFAOYSA-M Potassium chloride Chemical compound [Cl-].[K+] WCUXLLCKKVVCTQ-UHFFFAOYSA-M 0.000 description 7
- ONCZQWJXONKSMM-UHFFFAOYSA-N dialuminum;disodium;oxygen(2-);silicon(4+);hydrate Chemical compound O.[O-2].[O-2].[O-2].[O-2].[O-2].[O-2].[O-2].[O-2].[O-2].[O-2].[O-2].[O-2].[Na+].[Na+].[Al+3].[Al+3].[Si+4].[Si+4].[Si+4].[Si+4] ONCZQWJXONKSMM-UHFFFAOYSA-N 0.000 description 7
- LSHROXHEILXKHM-UHFFFAOYSA-N n'-[2-[2-[2-(2-aminoethylamino)ethylamino]ethylamino]ethyl]ethane-1,2-diamine Chemical compound NCCNCCNCCNCCNCCN LSHROXHEILXKHM-UHFFFAOYSA-N 0.000 description 7
- 239000001103 potassium chloride Substances 0.000 description 7
- 235000011164 potassium chloride Nutrition 0.000 description 7
- 238000005096 rolling process Methods 0.000 description 7
- 239000010703 silicon Substances 0.000 description 7
- 229940080314 sodium bentonite Drugs 0.000 description 7
- 229910000280 sodium bentonite Inorganic materials 0.000 description 7
- 238000012360 testing method Methods 0.000 description 7
- 238000001816 cooling Methods 0.000 description 6
- 238000007599 discharging Methods 0.000 description 6
- 238000010438 heat treatment Methods 0.000 description 6
- 239000007788 liquid Substances 0.000 description 6
- 238000011084 recovery Methods 0.000 description 6
- RTZKZFJDLAIYFH-UHFFFAOYSA-N ether Substances CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 5
- 230000005764 inhibitory process Effects 0.000 description 5
- 239000002002 slurry Substances 0.000 description 5
- BDLVNNJRAIUIMO-UHFFFAOYSA-N amino(hydroxy)silicon Chemical compound N[Si]O BDLVNNJRAIUIMO-UHFFFAOYSA-N 0.000 description 4
- 230000009286 beneficial effect Effects 0.000 description 4
- 238000010998 test method Methods 0.000 description 4
- 125000000217 alkyl group Chemical group 0.000 description 3
- 125000003368 amide group Chemical group 0.000 description 3
- 229920013822 aminosilicone Polymers 0.000 description 3
- 239000002245 particle Substances 0.000 description 3
- 238000006068 polycondensation reaction Methods 0.000 description 3
- 238000004537 pulping Methods 0.000 description 3
- 230000002829 reductive effect Effects 0.000 description 3
- 239000011734 sodium Substances 0.000 description 3
- 230000008961 swelling Effects 0.000 description 3
- 239000008399 tap water Substances 0.000 description 3
- 235000020679 tap water Nutrition 0.000 description 3
- 239000004721 Polyphenylene oxide Substances 0.000 description 2
- BLRPTPMANUNPDV-UHFFFAOYSA-N Silane Chemical compound [SiH4] BLRPTPMANUNPDV-UHFFFAOYSA-N 0.000 description 2
- 238000002441 X-ray diffraction Methods 0.000 description 2
- 230000032683 aging Effects 0.000 description 2
- 125000003277 amino group Chemical group 0.000 description 2
- 125000004429 atom Chemical group 0.000 description 2
- 229940092782 bentonite Drugs 0.000 description 2
- 229910000278 bentonite Inorganic materials 0.000 description 2
- 239000000440 bentonite Substances 0.000 description 2
- SVPXDRXYRYOSEX-UHFFFAOYSA-N bentoquatam Chemical compound O.O=[Si]=O.O=[Al]O[Al]=O SVPXDRXYRYOSEX-UHFFFAOYSA-N 0.000 description 2
- 150000001875 compounds Chemical class 0.000 description 2
- 230000007423 decrease Effects 0.000 description 2
- 238000011161 development Methods 0.000 description 2
- 230000018109 developmental process Effects 0.000 description 2
- KPUWHANPEXNPJT-UHFFFAOYSA-N disiloxane Chemical class [SiH3]O[SiH3] KPUWHANPEXNPJT-UHFFFAOYSA-N 0.000 description 2
- 238000011156 evaluation Methods 0.000 description 2
- 230000002349 favourable effect Effects 0.000 description 2
- 238000005098 hot rolling Methods 0.000 description 2
- 239000000203 mixture Substances 0.000 description 2
- 239000003921 oil Substances 0.000 description 2
- 229920002401 polyacrylamide Polymers 0.000 description 2
- 229920000570 polyether Polymers 0.000 description 2
- 150000003141 primary amines Chemical class 0.000 description 2
- 230000001681 protective effect Effects 0.000 description 2
- 238000007790 scraping Methods 0.000 description 2
- 229910000077 silane Inorganic materials 0.000 description 2
- 238000003786 synthesis reaction Methods 0.000 description 2
- NIQFAJBKEHPUAM-UHFFFAOYSA-N 2-[2-[2-(2-aminoethoxy)ethoxy]ethoxy]ethanamine Chemical compound NCCOCCOCCOCCN NIQFAJBKEHPUAM-UHFFFAOYSA-N 0.000 description 1
- RPNUMPOLZDHAAY-UHFFFAOYSA-N Diethylenetriamine Chemical compound NCCNCCN RPNUMPOLZDHAAY-UHFFFAOYSA-N 0.000 description 1
- IAYPIBMASNFSPL-UHFFFAOYSA-N Ethylene oxide Chemical compound C1CO1 IAYPIBMASNFSPL-UHFFFAOYSA-N 0.000 description 1
- PIICEJLVQHRZGT-UHFFFAOYSA-N Ethylenediamine Chemical compound NCCN PIICEJLVQHRZGT-UHFFFAOYSA-N 0.000 description 1
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 1
- 239000004698 Polyethylene Substances 0.000 description 1
- GOOHAUXETOMSMM-UHFFFAOYSA-N Propylene oxide Chemical compound CC1CO1 GOOHAUXETOMSMM-UHFFFAOYSA-N 0.000 description 1
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical group [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 description 1
- 150000003973 alkyl amines Chemical class 0.000 description 1
- 150000003863 ammonium salts Chemical class 0.000 description 1
- 230000000903 blocking effect Effects 0.000 description 1
- 238000012512 characterization method Methods 0.000 description 1
- 125000001309 chloro group Chemical group Cl* 0.000 description 1
- 238000003795 desorption Methods 0.000 description 1
- OTARVPUIYXHRRB-UHFFFAOYSA-N diethoxy-methyl-[3-(oxiran-2-ylmethoxy)propyl]silane Chemical compound CCO[Si](C)(OCC)CCCOCC1CO1 OTARVPUIYXHRRB-UHFFFAOYSA-N 0.000 description 1
- 238000001035 drying Methods 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 238000003912 environmental pollution Methods 0.000 description 1
- 125000003700 epoxy group Chemical group 0.000 description 1
- HHBOIIOOTUCYQD-UHFFFAOYSA-N ethoxy-dimethyl-[3-(oxiran-2-ylmethoxy)propyl]silane Chemical compound CCO[Si](C)(C)CCCOCC1CO1 HHBOIIOOTUCYQD-UHFFFAOYSA-N 0.000 description 1
- IWBOPFCKHIJFMS-UHFFFAOYSA-N ethylene glycol bis(2-aminoethyl) ether Chemical compound NCCOCCOCCN IWBOPFCKHIJFMS-UHFFFAOYSA-N 0.000 description 1
- 238000009775 high-speed stirring Methods 0.000 description 1
- 239000001257 hydrogen Substances 0.000 description 1
- 229910052739 hydrogen Inorganic materials 0.000 description 1
- 230000002209 hydrophobic effect Effects 0.000 description 1
- 239000011229 interlayer Substances 0.000 description 1
- 150000002500 ions Chemical class 0.000 description 1
- 239000007791 liquid phase Substances 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- 230000007935 neutral effect Effects 0.000 description 1
- 150000002894 organic compounds Chemical class 0.000 description 1
- 238000009304 pastoral farming Methods 0.000 description 1
- 239000003208 petroleum Substances 0.000 description 1
- 238000012643 polycondensation polymerization Methods 0.000 description 1
- 229920000573 polyethylene Polymers 0.000 description 1
- 229920000642 polymer Polymers 0.000 description 1
- XAEFZNCEHLXOMS-UHFFFAOYSA-M potassium benzoate Chemical compound [K+].[O-]C(=O)C1=CC=CC=C1 XAEFZNCEHLXOMS-UHFFFAOYSA-M 0.000 description 1
- 239000002994 raw material Substances 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 230000002441 reversible effect Effects 0.000 description 1
- 238000007151 ring opening polymerisation reaction Methods 0.000 description 1
- 150000003335 secondary amines Chemical class 0.000 description 1
- 239000002210 silicon-based material Substances 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 239000007858 starting material Substances 0.000 description 1
- 150000005846 sugar alcohols Polymers 0.000 description 1
- FAGUFWYHJQFNRV-UHFFFAOYSA-N tetraethylenepentamine Chemical compound NCCNCCNCCNCCN FAGUFWYHJQFNRV-UHFFFAOYSA-N 0.000 description 1
- JXUKBNICSRJFAP-UHFFFAOYSA-N triethoxy-[3-(oxiran-2-ylmethoxy)propyl]silane Chemical compound CCO[Si](OCC)(OCC)CCCOCC1CO1 JXUKBNICSRJFAP-UHFFFAOYSA-N 0.000 description 1
- BPSIOYPQMFLKFR-UHFFFAOYSA-N trimethoxy-[3-(oxiran-2-ylmethoxy)propyl]silane Chemical compound CO[Si](OC)(OC)CCCOCC1CO1 BPSIOYPQMFLKFR-UHFFFAOYSA-N 0.000 description 1
- MREDAUUVDJMZIU-UHFFFAOYSA-N trimethyl 5-(oxiran-2-ylmethoxy)pentyl silicate Chemical compound C(C1CO1)OCCCCCO[Si](OC)(OC)OC MREDAUUVDJMZIU-UHFFFAOYSA-N 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F7/00—Compounds containing elements of Groups 4 or 14 of the Periodic Table
- C07F7/02—Silicon compounds
- C07F7/08—Compounds having one or more C—Si linkages
- C07F7/18—Compounds having one or more C—Si linkages as well as one or more C—O—Si linkages
- C07F7/1804—Compounds having Si-O-C linkages
- C07F7/1872—Preparation; Treatments not provided for in C07F7/20
- C07F7/1892—Preparation; Treatments not provided for in C07F7/20 by reactions not provided for in C07F7/1876 - C07F7/1888
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K8/00—Compositions for drilling of boreholes or wells; Compositions for treating boreholes or wells, e.g. for completion or for remedial operations
- C09K8/02—Well-drilling compositions
- C09K8/03—Specific additives for general use in well-drilling compositions
- C09K8/035—Organic additives
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2208/00—Aspects relating to compositions of drilling or well treatment fluids
- C09K2208/12—Swell inhibition, i.e. using additives to drilling or well treatment fluids for inhibiting clay or shale swelling or disintegrating
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Solid-Sorbent Or Filter-Aiding Compositions (AREA)
Abstract
The invention discloses an amino film-forming inhibitor for drilling fluid and a preparation method thereof. The structural formula of the amino film forming inhibitor is one of the following two types:orThe inhibitor can effectively improve the inhibitive performance of the drilling fluid, including inhibiting clay slurrying and inhibiting clay hydration dispersion, and the temperature resistance can reach 150 ℃.
Description
Technical Field
The invention relates to the technical field of drilling, in particular to an amino film-forming inhibitor for drilling fluid and a preparation method thereof.
Background
Shale is a main stratum encountered in drilling engineering, and the inhibition of the slurrying of the shale stratum by improving the drilling fluid is the most important means for maintaining the stability of the well wall of the shale well section. Although the oil-based drilling fluid can inhibit slurry making of a shale stratum to a great extent and more effectively prevent collapse of a shale well section, due to the negative factors of high cost, environmental pollution and the like, the application range of the oil-based drilling fluid is limited, so that technical problems are emphasized by drilling fluid companies at home and abroad on an inhibitor for a water-based drilling fluid at present, namely, the inhibition of the drilling fluid is improved by adding a strong and effective inhibitor into the water-based drilling fluid, and the purpose of maintaining the stability of the well wall of the shale well section is achieved.
The synthesis, characterization and performance evaluation of the sodium polyamine serving as the drilling fluid inhibitor are provided in the literature structure and performance of the low polyamine shale inhibitor for the drilling fluid (drilling fluid and completion fluid 2015, 1 month). The treating agent takes amido as a main adsorption group and ether bond as a secondary adsorption group, so that molecules are flatly laid between clay layers, thereby being beneficial to reducing the formation of clay layer spacing.
The polyamine inhibitor SDPA provided in the literature, "development and application of novel polyamine shale hydration inhibitor" (journal of the university of Dian Petroleum, 2013, 3 months) is a polyetheramine inhibitor and belongs to conventional polyamine inhibitors.
The substance of the inhibitor disclosed in the above documents is that amine groups in molecules form ammonium ions under an alkaline condition (the liquid phase environment of the drilling fluid is alkaline), so that electrostatic adsorption occurs with negatively charged clay surfaces, or ether bonds or hydroxyl groups in the molecules form hydrogen bonds with hydroxyl groups on the clay particle surfaces, and the two types of adsorption belong to reversible physical adsorption, and are easy to desorb under a high temperature condition, so that the inhibition effect is reduced.
Chinese patent CN 103087691A discloses a polyamine strong inhibitor for drilling fluid and a preparation method thereof, the polyamine inhibitor provided by the patent (refuted) is obtained by reacting amine compounds (polyether amine, alkylamine, ether amine, tetraethylenepentamine, pentaethylenehexamine or polyethylene polyamine) with epoxy compounds (ethylene oxide or propylene oxide), the reaction mechanism is that the amine groups and epoxy groups are subjected to ring opening polymerization under alkaline conditions, and the obtained polymer molecular structure is similar to CN 104017208B and is polyether amine. The action mechanism is that amido, ether bond and hydroxyl distributed in a molecular chain are used as adsorption groups to be adsorbed with clay, so that the clay lamellar structure is bound, water molecules are prevented from invading between clay layers, and the hydration of the clay is inhibited.
At present, a polyamine film formation inhibitor obtained by reacting an amine compound with an epoxy compound, as proposed in CN 104017208B, is a polyamine inhibitor, which is mainly adsorbed on the surface of clay particles by taking amine and hydroxyl groups as adsorption groups to form a dense protective film to prevent water molecules from entering, thereby achieving the purpose of blocking hydration of water-sensitive clay, which is similar to the action mechanism of amino silanol. Currently, due to the outstanding inhibitory effect, amino inhibitors and their derivatives are gaining attention and are widely used. The amino silanol is an upgraded product of an amino inhibitor, and a strong adsorption group, namely silicon hydroxyl, is introduced on the basis of the amine inhibitor. The treating agent not only retains the molecular characteristics of the amido inhibitor, but also can lead the introduced silicon hydroxyl to have condensation polymerization reaction with the hydroxyl on the clay, so as to coat clay particles into a film, effectively prevent and slow down the hydration action of the clay surface, achieve the effect of inhibiting the hydration dispersion of the clay, and be beneficial to the stability of the well wall of the water-sensitive clay shale. The difference is that the amino polyalcohol not only depends on amino and hydroxyl as adsorption groups, and the adsorption effect of the introduced silicon hydroxyl is more favorable for improving the effect of the inhibitor.
Chinese patent CN 105670578A discloses an amino silanol inhibitor, which is a silane substance, wherein the silicon atom is directly connected with hydroxyl, primary amine and secondary amine are distributed on the long alkyl chain, and the key point of the preparation is step 3 in the reaction step, i.e. the reaction of silane with Cl atom and primary amine substance.
The literature, performance evaluation of novel aminosilicone alcohol inhibitor (petrochemical application, 2015, 12 months), provides a molecular structure of the aminosilicone alcohol inhibitor, explains the reaction mechanism, and does not provide a specific preparation method. The molecule has silicon hydroxyl as a chemical adsorption group and is provided with a long alkyl chain (R is presumed to be the long alkyl chain), so that the molecule forms a hydrophobic layer on the clay surface, and the hydration of the clay surface is prevented and slowed down.
In the prior amido silanol inhibitor, si atoms are directly connected with hydroxyl, and if the inhibitor is exposed in the air, intermolecular polycondensation reaction is easy to occur to influence the product quality, and the effective components are reduced. This is the case with the aminosilicone inhibitors referred to in CN 105670578A. In addition, CN 105670578A involves complicated synthesis steps of amino silanol inhibitors, involves expensive catalysts and is high in cost.
Therefore, the development of a high-efficiency inhibitor capable of preventing clay hydration dispersion and inhibiting hydration swelling is a technical problem to be solved at present.
Disclosure of Invention
In order to solve the problems in the prior art, the invention provides an amino film-forming inhibitor for drilling fluid and a preparation method thereof. Siloxane is introduced into molecules, and the sheet structure of the clay is firmly bound together by utilizing the chemical adsorption effect of silicon hydroxyl generated after the siloxane is hydrolyzed and the surface of the clay. Because the adsorption is chemical adsorption, the electrostatic adsorption on the clay surface is firmer than that of amino, thereby more effectively preventing water molecules from entering clay layers, and inhibiting hydration expansion while preventing hydration and dispersion of the clay. The inhibitive performance of the drilling fluid can be effectively improved, including inhibiting clay slurrying and inhibiting clay hydration and dispersion, and the temperature resistance can reach 150 ℃.
The invention aims to provide an amine-based film-forming inhibitor for drilling fluid.
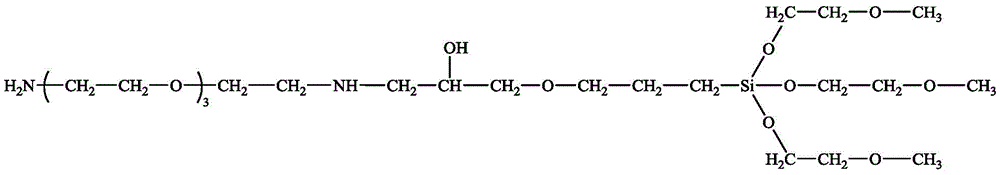
The structural formula of the amino film forming inhibitor is one of the following two types:
x is an integer of 0 to 5; preferably 0,1,2,3;
y is an integer of 1 to 10; preferably 1,2,3,4,5.
Wherein R is 1 、R 2 And R 3 Is selected from-CH 3 ,-O-CH 3 ,-O-CH 2 -CH 3 ,-O-CH 2 -CH 2 -O-CH 3 ,-O-CH 2 -CH 2 -O-CH 2 -CH 2 -O-CH 3 Is not simultaneously selected as-CH 3 。
The invention also aims to provide a preparation method of the amino film-forming inhibitor for the drilling fluid.
The method comprises the following steps:
fully mixing an amine compound, an alkali metal catalyst and a water removal agent in an alcohol solvent, then adding an epoxy organosilicon compound under the protection of inert gas, boosting the pressure to 0.3-0.5 MPa, reacting, filtering and distilling to obtain the amino film-forming inhibitor;
the molar ratio of the amine compound to the epoxy organosilicon organic compound is 1: (0.5-6); preferably 1: (1-3);
the mass of the alkali metal catalyst and the amine compound is 1: (500-800); preferably 1: (500-600);
the mass ratio of the water scavenger to the amine compound is 1: (300 to 500), preferably 1: (300-400).
The reaction temperature is 70-120 ℃; preferably 90 to 110 ℃; the reaction time is 1-5 h.
The alkali metal catalyst is preferably one of NaOH and KOH;
the alcohol solvent is preferably one of methanol and ethanol.
The water removal agent can adopt a conventional neutral water removal agent in the field, and in the invention, anhydrous CaCl can be preferably selected 2 。
The mass percentage concentration of the amine compound in the alcohol solvent is preferably 5.0-20%.
Wherein,
the structural formula of the amine compound is as follows:
x is an integer of 0 to 5, preferably 0,1,2,3;
y is an integer of 1 to 10, preferably 1,2,3,4,5.
The structural formula of the epoxy organosilicon compound is as follows:
wherein R is 1 、R 2 And R 3 Is selected from-CH 3 ,-O-CH 3 ,-O-CH 2 -CH 3 ,-O-CH 2 -CH 2 -O-CH 3 ,-O-CH 2 -CH 2 -O-CH 2 -CH 2 -O-CH 3 Is not simultaneously selected as-CH 3 。
The preparation method comprises the following steps:
fully mixing an amine compound, an alkali metal catalyst and a water removal agent in an alcohol solvent in a sealed reaction vessel, and heating to 70-120 ℃, preferably 90-110 ℃; then adding epoxy organosilicon compounds under the protection of inert gas, boosting the pressure to 0.3-0.5 MPa, and continuously reacting for 1-5 h; cooling, decompressing and discharging, filtering to remove insoluble matters, decompressing and distilling to remove the alcohol solvent to constant weight to obtain light yellow viscous liquid, namely the amino film-forming inhibitor.
The prepared amino film-forming inhibitor belongs to organosilicon inhibitors, and the amino, hydroxyl and ether bonds in the molecules can be beneficial to improving the adsorption capacity of inhibitor molecules in water-sensitive clay; in addition, si atoms in molecules are connected with methoxy, ethoxy or methoxyethoxy, and are hydrolyzed in alkaline drilling fluid to generate silicon hydroxyl which is then subjected to polycondensation reaction with hydroxyl on the surface of water-sensitive clay to generate firm chemical adsorption, so that a compact protective film is formed on the surface of the water-sensitive clay; in addition, the molecules entering the clay layers bind the clay lamellar structure together under the adsorption action of protonated ammonium positive ions, and silicon hydroxyl in the molecules can also perform polycondensation reaction with hydroxyl among the clay layers, so that the binding effect is firmer, water molecules are more effectively prevented from entering the clay layers, and the inhibition of hydration expansion is realized while the hydration dispersion of the clay is prevented.
Drawings
FIG. 1 is a graph of the test data of the slurry forming performance of the sodium bentonite inhibited by the inhibitor, clear water and KCl solution prepared in the example.
Detailed Description
The present invention is further illustrated by the following examples.
The raw material is from Nanjing Engineers organic silicon materials GmbH, jiangsu Nanjing; qufuchenguang chemical Co., ltd, shandong Jinning;
all starting materials are commercially available.
Example 1
In a sealed reaction vessel, 60.1g (1.0 mol) ethylenediamine (x = 0), 0.12g NaOH, and 0.17g anhydrous CaCl 2 Fully mixing the materials in 500mL of ethanol, and heating to 105 ℃; then in N 2 Under the protection of (2.0 mol), adding 472.68g (2.0 mol) of 3- (2, 3-epoxypropoxy) propyl trimethoxy silane, increasing the pressure to 0.35MPa, and continuously reacting for 2.5h; cooling, decompressing and discharging, filtering to remove insoluble substances, decompressing and distilling to remove ethanol to constant weight to obtain light yellow viscous liquid, namely the amino film-forming inhibitor. The structural formula is as follows:
example 2
In a sealed reaction vessel, 148.2g of EDR148 (3, 6-dioxaoctane-1, 8-diamine, 1.0 mol) (x = 2), 0.27g of KOH and 0.42g of anhydrous CaCl 2 Fully mixing the components in 1000mL of methanol, and heating to 90 ℃; then in N 2 Under the protection of (1.0) g of (3-glycidoxypropyl) dimethyl ethoxysilane, increasing the pressure to 0.5MPa, and continuously reacting for 2.0h; cooling, decompressing and discharging, filtering to remove insoluble matters, decompressing and distilling to remove methanol to constant weight to obtain light yellow viscous liquid, namely the amino film-forming inhibitor. The structural formula is as follows:
example 3
In a sealed reaction vessel, 192.26g (1.0 mol)3,6,9-Trioxaundecane-1, 11-diamine (x = 3), 0.35g NaOH and 0.6g anhydrous CaCl 2 Fully mixing in 2000mL of ethanol, and heating to 95 ℃; then in N 2 Under the protection of (2), 407.74g (1.106 mol) of 3- (2, 3-epoxypropoxy) propyl trimethoxy ethoxy silane is added, the pressure is increased to 0.43MPa, and the reaction is continued for 4.0h; cooling, decompressing and discharging, filtering to remove insoluble substances, decompressing and distilling to remove ethanol to constant weight to obtain light yellow viscous liquid, namely the amino film-forming inhibitor.
The structural formula is as follows:
example 4
In a sealed reaction vessel, a solution of the formula 103.17g (1.0 mol) diethylenetriamine (y = 1), 0.2g NaOH and 0.3g anhydrous CaCl 2 Fully mixing in 1800mL ethanol, and heating to 105 ℃; then in N 2 661.02g (2.66 mol) of (3-glycidoxypropyl) methyldiethoxysilane is added under the protection of (1), the pressure is increased to 0.5MPa, and the reaction is continued for 5.0h; cooling, decompressing and discharging, filtering to remove insoluble substances, decompressing and distilling to remove ethanol to constant weight to obtain light yellow viscous liquid, namely the amino film-forming inhibitor. The structural formula is as follows:
example 5
In a sealed reaction vessel, 232.37g (1.0 mol) of pentaethylenehexamine (y = 4), 0.4g NaOH and 0.63g anhydrous CaCl 2 Fully mixing in 2200mL of methanol, and heating to 105 ℃; then in N 2 Adding 325.77g (1.17 mol) of 3- (2, 3-epoxypropoxy) propyltriethoxysilane under the protection of the (1.17 mol), increasing the pressure to 0.3MPa, and continuously reacting for 5.0h; cooling, decompressing and discharging, filtering to remove insoluble matters, decompressing and distilling to remove methanol to constant weight to obtain light yellow viscous liquid, namely the amino film-forming inhibitor. The structural formula is as follows:
example 6
Test for inhibiting hydration dispersion performance of shale by amino film forming inhibitor
( The test method comes from: zhuqi, li Tao, zhang Tianxiao, etc. Jinshan aerofield strong inhibition anti-collapse drilling fluid research and application [ J ] scientific technology and engineering, 2014, 14 (22), 35-40. )
40.0g of shale rock debris with 6-10 meshes is respectively put into a high-temperature aging tank filled with clear water, 7.0 percent of KCl, 1.0 percent of polyacrylamide sylvite and 0.3 percent of the amino film forming inhibitor for drilling fluid prepared in the embodiments 1-5, and is taken out after rolling aging is carried out for 16.0h at 90 ℃, 120 ℃ and 150 ℃, the temperature is cooled to room temperature, the rock debris is recovered by passing through a 28-mesh sieve and a 40-mesh sieve in sequence, the rock debris is dried to constant weight at 105 ℃, the mass of the rock debris is weighed, and the rolling recovery rate is calculated, and the experimental results are shown in tables 1-3:
TABLE 1 Rolling recovery of rock debris (90 ℃ C.. Times.16 h) for different inhibitors
TABLE 2 rock debris rolling recovery of different inhibitors (120 ℃ C.. Times.16 h)
TABLE 3 Rolling recovery of rock debris (150 ℃ C.. Times.16 h) for different inhibitors
As can be seen from tables 1 to 3, the experimental slurry containing 5 different amine-based film-forming inhibitors in an amount of 0.3% was higher in the rolling recovery rate through a 28-mesh sieve and higher in the rolling recovery rate through a 40-mesh sieve than the clear water, the 7.0% KCl and the 1.0% polyacrylamide potassium salt, which indicates that the 5 different amine-based film-forming inhibitors prepared by the examples of the present invention can effectively ensure the integrity of rock debris and are more favorable for improving the stability of the well wall of the well.
Example 7
Test of performance of amino film forming inhibitor for inhibiting sodium bentonite pulping
( The test method comes from: zhuqi, li Tao, wang dong, etc. Longfeng mountain air field plugging type anti-collapse drilling fluid [ J ] the drilling fluid and completion fluid 2016, 33 (5), 35-40. )
Clean water, 7.0 percent of KCl, 0.3 percent of the amino film-forming inhibitor for the drilling fluid prepared in the examples 1 to 5 and 5.0 percent of sodium bentonite are quantitatively added into 400mL of tap water, the mixture is stirred at a high speed for 30min, the pH value of the system is adjusted to be more than or equal to 9.0 by using 0.1 percent of NaOH solution, the 3-turn reading of the experimental slurry after hot rolling for 16h at 90 ℃ (the 3-turn reading (the rotation speed of a rotor is 3 rmp) is the reading of a six-speed rotational viscometer, and the rheological property is characterized under the condition of low shear rate. The larger the reading, the more viscous the drilling fluid and the higher the slurrification rate ]. Then 5.0% sodium bentonite is added, high speed stirring is carried out for 30min, and the measurement is carried out after hot rolling under the same conditions. Repeating the steps until the degree can not be measured. The effect of the 3-turn reading of the experimental slurry at each stage was compared to the effect of the different inhibitors.
Under the same bentonite addition condition, the 3-turn reading of the amino film-forming inhibitor for the drilling fluid prepared in the examples 1 to 5 is equivalent and always maintained at a lower level, which shows that the ability of inhibiting the hydration of sodium bentonite is stronger than that of the conventional inhibitor KCl in inhibiting the hydration and pulping of bentonite, and is shown in figure 1. (the higher the pulping capacity, the greater the 3-revolution reading, although the 3-revolution reading for each sample increased with increasing sodium bentonite addition, the rate of rise of the 3-revolution reading for the sample with the amine-based film forming inhibitor added was significantly less than the rate of rise of the 3-revolution reading for the control (clear water and KCl)).
Example 8
Test for inhibiting hydration expansion of sodium bentonite by amino film forming inhibitor
( The test method comes from: structure and performance of oligoamine shale inhibitors for drilling fluids [ J ] 2015, 32 (1), 26-29. )
Adding 400mL of tap water into a high stirring cup, adding 32g of clay under a certain stirring speed, and then adding 1.6g of Na 2 CO 3 . Stirring for 20min, stopping stirring at least twice during the stirring, scraping off clay adhered to the wall of the container, and curing in a sealed container for 24h. Adding 0.3% of EDR148, 0.3% of pentaethylenehexamine and 0.3% of the amine film-forming inhibitor for drilling fluid prepared in examples 1 to 5, drying the mixture to constant weight after 2.0h, 4.0h and 6.0h respectively, testing XRD (X-ray diffraction) spectrograms of different samples by using an X-ray diffractometer to obtain grazing angles of the different samples, and calculating the clay layer spacing (the interlayer spacing of the untreated clay is 0.9675 nm) based on a Bragg equation. The calculation results are shown in table 4:
TABLE 4 Clay layer spacing (nm) after treatment with different inhibitors
As can be seen from table 4, water molecules continuously invade between clay layers to cause hydration swelling as time goes on. In contrast, the addition of an inhibitor is beneficial to hinder the occurrence of hydration swelling of the clay. Obviously, the effect of the amine filming inhibitor obtained in the embodiments 1 to 5 on inhibiting hydration expansion of clay is obviously better than that of EDR148 and pentaethylenehexamine, which indicates that the adsorption force between the amine filming inhibitor for drilling fluid and clay provided by the invention is stronger, and the effect of inhibiting water molecules from invading into clay layers is more obvious.
Example 9
Adsorption Capacity test of amine-based film formation inhibitor
( The test method comes from: method for testing adsorption performance of organic drilling fluid treating agent [ P ]. CN 105277675A, 2014.06.26 )
400mL of tap water was added to a high stirring cup, 32g of clay was added at a certain stirring speed, and 1.6g of Na was added 2 CO 3 . Stirring for 20min, stopping stirring at least twice during the stirring, scraping off clay adhered to the wall of the container, and curing in a sealed container for 24h. Adding 0.3% of EDR148, 0.3% of pentaethylenehexamine and0.3% of the amino film-forming inhibitor for drilling fluid prepared in the examples 1 to 5 was allowed to stand for 2 hours, and then the adsorption amounts of different samples at different temperatures were measured by a thermal filtration method. The calculation results are shown in table 5:
TABLE 5 adsorption amounts (mg/L) of different inhibitors
As can be seen from table 5, the adsorption amounts of the amine-based film formation inhibitors obtained in examples 1 to 5 are significantly higher than those of EDR148 and pentaethylenehexamine, the adsorption amount of the sample gradually decreases with the increase of the temperature, and the adsorption amounts of EDR148 and pentaethylenehexamine rapidly decrease when the temperature is higher than 140 ℃, which indicates that significant desorption occurs between the sample and clay under the temperature condition; when the temperature is 160 ℃, the adsorption amount of the amine-based film formation inhibitor obtained in examples 1 to 5 is remarkably reduced, which indicates that the temperature resistance of the amine-based film formation inhibitor can reach 150 ℃.
Claims (8)
1. An amino film-forming inhibitor for drilling fluid is characterized in that the structural formula of the amino film-forming inhibitor is one of the following two:
x is an integer of 1 to 5; y is an integer of 1 to 10;
wherein R is 1 、R 2 And R 3 Is selected from-CH 3 ,-O-CH 3 ,-O-CH 2 -CH 3 ,-O-CH 2 -CH 2 -O-CH 3 ,-O-CH 2 -CH 2 -O-CH 2 -CH 2 -O-CH 3 Is not simultaneously selected as-CH 3 。
2. An amine-based film-forming inhibitor for drilling fluids according to claim 1, wherein:
x is 1,2 or 3;
y is 1,2,3,4 or 5.
3. A method for preparing the amine-based film-forming inhibitor for drilling fluid according to claim 1 or 2, wherein the method comprises:
fully mixing an amine compound, an alkali metal catalyst and a water removal agent in an alcohol solvent, then adding an epoxy organosilicon compound under the protection of inert gas, boosting the pressure to 0.3-0.5 MPa, reacting, filtering and distilling to obtain the amino film-forming inhibitor; the reaction temperature is 70-120 ℃; the reaction time is 1-5 h;
the molar ratio of the amine compound to the epoxy organosilicon compound is 1: 0.5-6;
the mass ratio of the alkali metal catalyst to the amine compound is 1: 500-800;
the mass ratio of the water removing agent to the amine compound is 1: 300-500;
the structural formula of the amine compound is as follows:
x is an integer of 1 to 5, and y is an integer of 1 to 10;
the structural formula of the epoxy organosilicon compound is as follows:
wherein R is 1 、R 2 And R 3 Is selected from-CH 3 ,-O-CH 3 ,-O-CH 2 -CH 3 ,-O-CH 2 -CH 2 -O-CH 3 ,-O-CH 2 -CH 2 -O-CH 2 -CH 2 -O-CH 3 Is not simultaneously selected as-CH 3 。
4. The method for producing the amine-based film formation inhibitor according to claim 3, wherein:
the reaction temperature is 90 to 110 ℃.
5. The method for preparing the amine-based film formation inhibitor according to claim 3, wherein:
the alkali metal catalyst is one of NaOH and KOH;
the alcohol solvent is one of methanol and ethanol;
the water removing agent is anhydrous CaCl 2 。
6. The method for producing the amine-based film formation inhibitor according to claim 3, wherein:
the molar ratio of the amine compound to the epoxy organosilicon compound is 1: (1 to 3);
the mass of the alkali metal catalyst and the amine compound is 1: (500 to 600);
the mass ratio of the water scavenger to the amine compound is 1: (300 to 400).
7. The method for producing the amine-based film formation inhibitor according to claim 3, wherein:
the mass percentage concentration of the amine compound in the alcohol solvent is 5.0-20%.
8. The method for producing the amine-based film formation inhibitor according to claim 3, wherein:
x is 1,2 or 3;
y is 1,2,3,4 or 5.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201811293348.9A CN111116632B (en) | 2018-11-01 | 2018-11-01 | Amino film forming inhibitor for drilling fluid and preparation method thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201811293348.9A CN111116632B (en) | 2018-11-01 | 2018-11-01 | Amino film forming inhibitor for drilling fluid and preparation method thereof |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN111116632A CN111116632A (en) | 2020-05-08 |
| CN111116632B true CN111116632B (en) | 2022-11-29 |
Family
ID=70494689
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201811293348.9A Active CN111116632B (en) | 2018-11-01 | 2018-11-01 | Amino film forming inhibitor for drilling fluid and preparation method thereof |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN111116632B (en) |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN114315605B (en) * | 2020-10-09 | 2024-04-02 | 中石化南京化工研究院有限公司 | Amine intercalation inhibitor for water-based drilling fluid and preparation method thereof |
Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4513115A (en) * | 1982-02-17 | 1985-04-23 | General Electric Company | Low modulus one component RTV compositions processes |
| WO2008037468A2 (en) * | 2006-09-28 | 2008-04-03 | Huntsman Textile Effects (Germany) Gmbh | Dimeric and oligomeric silanes and reaction products thereof with organosiloxanes |
| CN103443109A (en) * | 2011-02-25 | 2013-12-11 | 纳尔科公司 | Reducing aluminosilicate scale in the Bayer process |
| CN103724368A (en) * | 2014-01-09 | 2014-04-16 | 嘉兴学院 | Preparation method of amino polyether-type silanization reagent |
| CN103965843A (en) * | 2014-05-04 | 2014-08-06 | 中国石油集团渤海钻探工程有限公司 | Multiple-effect inhibitor for water-based drilling fluid and preparing method thereof |
| CN105670578A (en) * | 2016-02-22 | 2016-06-15 | 中石化石油工程技术服务有限公司 | Silicon amino drilling fluid and preparation method thereof |
| CN106190062A (en) * | 2016-06-28 | 2016-12-07 | 长江大学 | A kind of preparation method of drilling fluid poly-organosilicon amine inhibitors |
| CN107619465A (en) * | 2017-08-21 | 2018-01-23 | 湖南七纬科技有限公司 | A kind of organic silicon modified aqueous isocyanate trimer curing agent and preparation method thereof |
-
2018
- 2018-11-01 CN CN201811293348.9A patent/CN111116632B/en active Active
Patent Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4513115A (en) * | 1982-02-17 | 1985-04-23 | General Electric Company | Low modulus one component RTV compositions processes |
| WO2008037468A2 (en) * | 2006-09-28 | 2008-04-03 | Huntsman Textile Effects (Germany) Gmbh | Dimeric and oligomeric silanes and reaction products thereof with organosiloxanes |
| CN103443109A (en) * | 2011-02-25 | 2013-12-11 | 纳尔科公司 | Reducing aluminosilicate scale in the Bayer process |
| CN103724368A (en) * | 2014-01-09 | 2014-04-16 | 嘉兴学院 | Preparation method of amino polyether-type silanization reagent |
| CN103965843A (en) * | 2014-05-04 | 2014-08-06 | 中国石油集团渤海钻探工程有限公司 | Multiple-effect inhibitor for water-based drilling fluid and preparing method thereof |
| CN105670578A (en) * | 2016-02-22 | 2016-06-15 | 中石化石油工程技术服务有限公司 | Silicon amino drilling fluid and preparation method thereof |
| CN106190062A (en) * | 2016-06-28 | 2016-12-07 | 长江大学 | A kind of preparation method of drilling fluid poly-organosilicon amine inhibitors |
| CN107619465A (en) * | 2017-08-21 | 2018-01-23 | 湖南七纬科技有限公司 | A kind of organic silicon modified aqueous isocyanate trimer curing agent and preparation method thereof |
Non-Patent Citations (2)
| Title |
|---|
| "新型胺基硅醇强抑制剂性能评价";邢希金等;《石油化工应用》;20151231;第34卷(第12期);第88-91页 * |
| "深水钻井液技术现状与发展趋势";邱正松等;《特种油气藏》;20130630;第20卷(第3期);第1-8页 * |
Also Published As
| Publication number | Publication date |
|---|---|
| CN111116632A (en) | 2020-05-08 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN111116631B (en) | Organic silicon inhibitor for drilling fluid and preparation method thereof | |
| CN105670578B (en) | A kind of silicon amine base drilling fluids and preparation method thereof | |
| Jiang et al. | A new inhibitor of P (AM‐DMDAAC)/PVA intermacromolecular complex for shale in drilling fluids | |
| CN102433110A (en) | Amine shale stabilizer for drilling fluid | |
| CN109762535B (en) | Shale intercalation inhibitor prepared from hyperbranched polyamine | |
| CN115991977B (en) | A raw material composition, a fluid loss reducer for drilling fluid, and a preparation method and application thereof | |
| CN109233762A (en) | A kind of drilling fluid, preparation method and application | |
| CN104357031A (en) | Silicate-cation alkyl glucoside drilling fluid and preparation method thereof | |
| CN117210208B (en) | Multiple double-effect collapse-preventing environment-friendly high-performance drilling fluid and preparation method thereof | |
| CN113861953B (en) | Drilling fluid and preparation method and application thereof | |
| CN111116632B (en) | Amino film forming inhibitor for drilling fluid and preparation method thereof | |
| CN112920783B (en) | Wetting reversal inhibitor for drilling fluid and preparation method thereof | |
| CN111057159B (en) | Anti-collapse modified starch for drilling fluid and preparation method thereof | |
| CN111057526B (en) | Environment-friendly anti-sloughing drilling fluid and preparation method thereof | |
| CN116462849B (en) | High temperature resistant inhibitor based on molecular nano particles and preparation method thereof | |
| CN117106180B (en) | Shale inhibitor for water-based drilling fluid and preparation method thereof | |
| CN116425979B (en) | A betaine type polysiloxane, preparation method and application thereof | |
| CN110373166B (en) | Environment-friendly drilling fluid and preparation method and application thereof | |
| CN109912545B (en) | Modified cationic compound, preparation method thereof and solid-phase chemical cleaning agent for drilling fluid | |
| CN115074095B (en) | Dendritic macromolecule coupling agent modified nano material and preparation method and application thereof | |
| CN111117580B (en) | Strong-adsorption amino inhibitor for drilling fluid and preparation method thereof | |
| CN111116633B (en) | Amino silanol inhibitor for drilling fluid and preparation method thereof | |
| CN120005581B (en) | Improved anti-collapse drilling fluid and preparation method thereof | |
| CN120118000B (en) | Water-based drilling fluid inhibitor, water-based drilling fluid and application thereof | |
| Zhou et al. | Nanoparticles and polymers complexes as a harmless shale plugging inhibitor for ocean water-based drilling fluids |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant |