CN110721663A - Modified cellulose particle and preparation method and application thereof - Google Patents
Modified cellulose particle and preparation method and application thereof Download PDFInfo
- Publication number
- CN110721663A CN110721663A CN201911077821.4A CN201911077821A CN110721663A CN 110721663 A CN110721663 A CN 110721663A CN 201911077821 A CN201911077821 A CN 201911077821A CN 110721663 A CN110721663 A CN 110721663A
- Authority
- CN
- China
- Prior art keywords
- modified cellulose
- particle
- cellulose particles
- cellulose particle
- reaction
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- 239000001913 cellulose Substances 0.000 title claims abstract description 53
- 229920002678 cellulose Polymers 0.000 title claims abstract description 53
- 239000002245 particle Substances 0.000 title claims abstract description 53
- 238000002360 preparation method Methods 0.000 title abstract description 8
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 claims abstract description 28
- 238000006243 chemical reaction Methods 0.000 claims abstract description 17
- PFKFTWBEEFSNDU-UHFFFAOYSA-N carbonyldiimidazole Chemical compound C1=CN=CN1C(=O)N1C=CN=C1 PFKFTWBEEFSNDU-UHFFFAOYSA-N 0.000 claims abstract description 14
- NWZSZGALRFJKBT-KNIFDHDWSA-N (2s)-2,6-diaminohexanoic acid;(2s)-2-hydroxybutanedioic acid Chemical compound OC(=O)[C@@H](O)CC(O)=O.NCCCC[C@H](N)C(O)=O NWZSZGALRFJKBT-KNIFDHDWSA-N 0.000 claims abstract description 9
- IKDUDTNKRLTJSI-UHFFFAOYSA-N hydrazine monohydrate Substances O.NN IKDUDTNKRLTJSI-UHFFFAOYSA-N 0.000 claims abstract description 9
- 238000000502 dialysis Methods 0.000 claims abstract description 8
- 238000003756 stirring Methods 0.000 claims abstract description 6
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Chemical compound O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims abstract description 6
- 238000007865 diluting Methods 0.000 claims abstract description 4
- 239000008367 deionised water Substances 0.000 claims abstract 2
- 229910021641 deionized water Inorganic materials 0.000 claims abstract 2
- 108091006003 carbonylated proteins Proteins 0.000 claims description 7
- 238000000034 method Methods 0.000 claims description 7
- 230000008081 blood perfusion Effects 0.000 claims description 6
- 238000001035 drying Methods 0.000 claims 1
- 230000008014 freezing Effects 0.000 claims 1
- 238000007710 freezing Methods 0.000 claims 1
- 238000004108 freeze drying Methods 0.000 abstract description 5
- 108090000623 proteins and genes Proteins 0.000 description 16
- 102000004169 proteins and genes Human genes 0.000 description 16
- 230000006315 carbonylation Effects 0.000 description 10
- 238000005810 carbonylation reaction Methods 0.000 description 10
- 125000002915 carbonyl group Chemical group [*:2]C([*:1])=O 0.000 description 9
- OAKJQQAXSVQMHS-UHFFFAOYSA-N hydrazine group Chemical group NN OAKJQQAXSVQMHS-UHFFFAOYSA-N 0.000 description 8
- 230000032683 aging Effects 0.000 description 5
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 5
- 230000000694 effects Effects 0.000 description 5
- 239000004005 microsphere Substances 0.000 description 5
- 230000004048 modification Effects 0.000 description 5
- 238000012986 modification Methods 0.000 description 5
- 239000000243 solution Substances 0.000 description 5
- 201000010099 disease Diseases 0.000 description 4
- 125000000468 ketone group Chemical group 0.000 description 4
- 230000035882 stress Effects 0.000 description 4
- 238000006467 substitution reaction Methods 0.000 description 4
- 108091003079 Bovine Serum Albumin Proteins 0.000 description 3
- 229940098773 bovine serum albumin Drugs 0.000 description 3
- HGINCPLSRVDWNT-UHFFFAOYSA-N Acrolein Chemical compound C=CC=O HGINCPLSRVDWNT-UHFFFAOYSA-N 0.000 description 2
- 125000003172 aldehyde group Chemical group 0.000 description 2
- 150000001299 aldehydes Chemical class 0.000 description 2
- 239000000945 filler Substances 0.000 description 2
- 229920002521 macromolecule Polymers 0.000 description 2
- 239000011148 porous material Substances 0.000 description 2
- 239000012460 protein solution Substances 0.000 description 2
- 230000009758 senescence Effects 0.000 description 2
- NHJVRSWLHSJWIN-UHFFFAOYSA-N 2,4,6-trinitrobenzenesulfonic acid Chemical compound OS(=O)(=O)C1=C([N+]([O-])=O)C=C([N+]([O-])=O)C=C1[N+]([O-])=O NHJVRSWLHSJWIN-UHFFFAOYSA-N 0.000 description 1
- HWTDMFJYBAURQR-UHFFFAOYSA-N 80-82-0 Chemical compound OS(=O)(=O)C1=CC=CC=C1[N+]([O-])=O HWTDMFJYBAURQR-UHFFFAOYSA-N 0.000 description 1
- 208000024827 Alzheimer disease Diseases 0.000 description 1
- 102000015081 Blood Coagulation Factors Human genes 0.000 description 1
- 108010039209 Blood Coagulation Factors Proteins 0.000 description 1
- 208000024172 Cardiovascular disease Diseases 0.000 description 1
- 208000006545 Chronic Obstructive Pulmonary Disease Diseases 0.000 description 1
- 108090000790 Enzymes Proteins 0.000 description 1
- 102000004190 Enzymes Human genes 0.000 description 1
- 206010028980 Neoplasm Diseases 0.000 description 1
- 208000018737 Parkinson disease Diseases 0.000 description 1
- 208000001647 Renal Insufficiency Diseases 0.000 description 1
- 241000700605 Viruses Species 0.000 description 1
- 238000009098 adjuvant therapy Methods 0.000 description 1
- 239000003463 adsorbent Substances 0.000 description 1
- 206010003246 arthritis Diseases 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- 230000000975 bioactive effect Effects 0.000 description 1
- 230000003851 biochemical process Effects 0.000 description 1
- 239000008280 blood Substances 0.000 description 1
- 210000004369 blood Anatomy 0.000 description 1
- 239000003114 blood coagulation factor Substances 0.000 description 1
- 229940019700 blood coagulation factors Drugs 0.000 description 1
- 201000011510 cancer Diseases 0.000 description 1
- 150000001728 carbonyl compounds Chemical class 0.000 description 1
- 238000004132 cross linking Methods 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 230000018109 developmental process Effects 0.000 description 1
- 206010012601 diabetes mellitus Diseases 0.000 description 1
- 229940079593 drug Drugs 0.000 description 1
- 239000003814 drug Substances 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 1
- 238000001727 in vivo Methods 0.000 description 1
- 230000000977 initiatory effect Effects 0.000 description 1
- 238000005184 irreversible process Methods 0.000 description 1
- 201000006370 kidney failure Diseases 0.000 description 1
- 230000000670 limiting effect Effects 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 239000012567 medical material Substances 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- 244000052769 pathogen Species 0.000 description 1
- 231100000915 pathological change Toxicity 0.000 description 1
- 230000036285 pathological change Effects 0.000 description 1
- 230000001575 pathological effect Effects 0.000 description 1
- 102000004196 processed proteins & peptides Human genes 0.000 description 1
- 108090000765 processed proteins & peptides Proteins 0.000 description 1
- 238000000746 purification Methods 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 230000000717 retained effect Effects 0.000 description 1
- 230000002441 reversible effect Effects 0.000 description 1
- 230000002000 scavenging effect Effects 0.000 description 1
- 238000000926 separation method Methods 0.000 description 1
- 238000001179 sorption measurement Methods 0.000 description 1
- 241000894007 species Species 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 208000024891 symptom Diseases 0.000 description 1
- 238000012360 testing method Methods 0.000 description 1
- 239000003053 toxin Substances 0.000 description 1
- 231100000765 toxin Toxicity 0.000 description 1
- 108700012359 toxins Proteins 0.000 description 1
Classifications
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J20/00—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof
- B01J20/22—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof comprising organic material
- B01J20/26—Synthetic macromolecular compounds
Abstract
A modified cellulose particle is characterized in that the modified cellulose particle is a hydrazide functional cellulose particle, and the preparation method comprises the following steps of suspending the cellulose particle in a proper amount of dimethyl sulfoxide, adding carbonyldiimidazole with the mass of 0.8 ~ 1.5.5 times of the weight of the cellulose particle, stirring at room temperature for 2 ~ 4 hours, dropwise adding excessive hydrazine hydrate solution, continuing to react for 12 ~ 24 hours, after the reaction is terminated, diluting the reaction system with deionized water, dialyzing with a dialysis bag for 2 ~ 4 days, and storing after freeze drying.
Description
Technical Field
The invention belongs to the field of medical materials, and particularly relates to modified cellulose particles.
Background
With the advent of aging society, the incidence of age-and aging-related diseases is increasing. The existing research shows that the active carbonyl compounds are involved in the initiation and development process of various age-related diseases (such as arthritis, renal failure, cardiovascular diseases, Parkinson's disease, Alzheimer's disease, cancer, diabetes, chronic obstructive pulmonary disease and the like) and stress, and even directly participate in and initiate the aging process. Thus, the theory of carbonyl stress senescence suggests that carbonyl stress is one of the core biochemical processes of biological senescence. Carbonyl stress means that the generation of active carbonyl substances in a biological system exceeds the clearing capacity, so that the carbonylation modification of biological macromolecules such as protein and the like is caused, the biological macromolecules are subjected to structural change and functional loss, the functional disorder of cells and tissues is caused, and finally the pathological and physiological change of organisms and the aging process are caused.
The current strategies to solve this problem, mostly to eliminate reactive carbonyl species in vivo or to block the protein carbonylation process, are less concerned with how to solve the already formed carbonylated proteins. Protein carbonylation is an irreversible process, and when the carbonylation protein is accumulated in the body beyond a certain amount, a series of physiological and pathological changes can be caused.
Cellulose is widely used in the medical field due to its excellent properties and safety, and can be used for drug separation and purification, proteins, peptides, blood coagulation factors, enzymes, viruses and other bioactive media, as well as immunoadsorbent materials, and also in blood perfusion device fillers. The main mechanism of blood perfusion is the adsorption effect of the filler, which can directly remove pathogens or toxins in the blood of patients and adjust the stability of the microenvironment of human bodies, thereby achieving the purposes of relieving symptoms and treating diseases, and having obvious effect.
The Chinese patent with application number 201210334217.7 discloses a porous cellulose microsphere adsorbent for blood perfusion and a preparation method thereof, the particle size of the prepared porous cellulose microsphere is 0.1 ~ 2mm, the size of the internal pores of the cellulose microsphere is mainly distributed at 5nm ~ 40nm, the specific surface area is 400 ~ 1000m2/g and can resist the operation pressure of 7 ~ 15bar, and the preparation method is characterized in that a reverse suspension system is utilized to obtain the porous spherical microsphere, and the porous cellulose microsphere with the operation pressure of 7 ~ 15bar can be obtained through crosslinking.
Disclosure of Invention
The invention aims to provide cellulose particles containing a scavenging carbonylation protein and a preparation method thereof.
In order to solve the technical problems, the technical scheme of the invention is as follows:
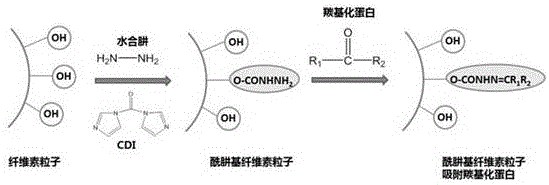
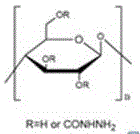
a modified cellulose particle which is a hydrazide-functionalized cellulose particle having the molecular schematic formula:
the synthetic route of the modified cellulose particle is as follows:
the effect of modified cellulose particles on modification and carbonylation of protein is shown as follows:
the preparation method comprises the following steps:
suspending cellulose particles in proper amount of dimethyl sulfoxide (DMSO), adding Carbonyldiimidazole (CDI) which is 0.8 ~ 1.5.5 times of the mass of the cellulose particles, stirring at room temperature for 2 ~ 4 hours, dropwise adding excess hydrazine hydrate solution, continuing the reaction for 12 ~ 24 hours, stopping the reaction, diluting the reaction system with water, dialyzing with a dialysis bag for 2 ~ 4 days, and storing after freeze drying.
Further, the concentration of the hydrazine hydrate solution is 78-82%.
Preferably, the molecular weight of the dialysis bag is 2000-4000 kD.
Furthermore, the purity of the dimethyl sulfoxide is more than or equal to 99.9 percent, and the purity of the carbonyldiimidazole is more than or equal to 97.0 percent.
A modified cellulose particle for use in blood perfusion apparatus to remove carbonylated proteins.
Compared with the prior art, the invention has the following beneficial effects:
1. the invention utilizes hydrazine hydrate to react with hydroxyl on cellulose particles, connects hydrazide groups on the cellulose particles to obtain hydrazide-functionalized cellulose particles, the particle diameter is 50 ~ 70 μm, the cellulose particles are circular, the surface is rough, the uniform pore diameter is provided, the cellulose particles are decomposed at about 340 ℃, compared with unfunctionalized cellulose particles, the product performance is compared, the particle diameter and the shape of the hydrazide-functionalized cellulose particles are not obviously changed, and the thermal stability is not obviously changed.
2. The excellent properties of the cellulose particles are retained, based on the fact that the hydrazide groups can bind to aldehyde or ketone groups, while the carbonylated proteins have aldehyde or ketone groups, and the function of adsorbing the carbonylated proteins is increased.
3. The invention introduces hydrazide group into the traditional cellulose particle to obtain hydrazide cellulose particle, and proves that the hydrazide cellulose particle has a clearing effect on carbonylation protein, and has wider application prospect for the adjuvant therapy of the aging diseases.
4. The preparation method of the modified cellulose particles is simple and controllable in particle size.
Detailed Description
The present invention will be described in detail with reference to specific embodiments, which are illustrative of the invention and are not to be construed as limiting the invention.
Example 1
Suspending 2g cellulose particles (CB) in 20ml dimethyl sulfoxide (DMSO), adding 1.6g Carbonyl Diimidazole (CDI), stirring at room temperature for 2 ~ 4 hours, adding 8 ml hydrazine hydrate solution drop by drop, continuing the reaction for 12 hours, after stopping the reaction, diluting the reaction system with water, dialyzing with dialysis bag (2000 and 4000 kD) for 2 ~ 4 days, and storing after freeze drying, thus obtaining the modified cellulose particles.
Example 2
After 4g of cellulose particles (CB) were suspended in 40 ml of dimethyl sulfoxide (DMSO), 6g of Carbonyldiimidazole (CDI) was added, and after stirring at room temperature for 3 hours, 15 ml of hydrazine hydrate solution was added dropwise, and the reaction was continued for 18 hours, after the reaction was terminated, the reaction system was diluted with water and dialyzed with a dialysis bag (2000 + 4000 kD) for 2 ~ 4 days, and stored after freeze-drying.
Example 3
5g of cellulose particles (CB) were suspended in 50 ml of dimethyl sulfoxide (DMSO), 5g of Carbonyldiimidazole (CDI) was added, and after stirring at room temperature for 4 hours, 18 ml of a hydrazine hydrate solution was added dropwise, and the reaction was continued for 24 hours, after the reaction was terminated, the reaction system was diluted with water and dialyzed with a dialysis bag (2000 + 4000 kD) for 2 ~ 4 days, and stored after freeze-drying.
In the above examples, the purity of dimethyl sulfoxide is not less than 99.9%, and the purity of carbonyldiimidazole is not less than 97.0%.
Examples of the experiments
The modified cellulose particles obtained in example 1 ~ 3 and unmodified cellulose particles (control) were subjected to,
the substitution rate refers to the content of hydrazine groups in hydrazine group modification of cellulose particles.
(1) And (3) measuring the substitution rate:
the amount of hydrazine groups was measured by the TNBS (nitrobenzenesulfonic acid) method and the results are shown in Table I, wherein the substitution rate refers to the content of hydrazine groups in the hydrazine modification of the cellulose particles.
Table one, content of hydrazine groups:
| sample (I) | Control group | Example 1 | Example 2 | Example 3 |
| Substitution Rate (%) | 0 | 0.2339 | 0.3730 | 1.0697 |
(2) Determination of carbonyl protein clearance:
mixing Bovine Serum Albumin (BSA) with acrolein overnight to obtain carbonylated BSA, immersing cellulose particles of control group and example 1 ~ 3 in carbonylated protein solution for 24 hr, and detecting carbonylated protein solution by UV-visible spectrumCalculating the amount of protein adsorbed by the cellulose particles;
| sample (I) | Control group | Example 1 | Example 2 | Example 3 |
| Amount of carbonyl protein eliminated (mg) for 100mg of sample | 0.002 | 0.065 | 0.161 | 0.264 |
As shown in Table 1, example 1 ~ 3 can effectively connect hydrazine group, and based on the hydrazide group can combine aldehyde group or ketone group, and the carbonylation protein has aldehyde group or ketone group, so as to increase the function of adsorbable carbonylation protein, Table 2 is the carbonyl protein clearance test, the technical scheme of the invention can have a certain clearance effect on carbonyl protein, thus, the modified cellulose particle can be applied to blood perfusion device, and clearance carbonylation protein.
The foregoing embodiments are merely illustrative of the principles and utilities of the present invention and are not intended to limit the invention. Any person skilled in the art can modify or change the above-mentioned embodiments without departing from the spirit and scope of the present invention. Accordingly, it is intended that all equivalent modifications or changes which can be made by those skilled in the art without departing from the spirit and technical spirit of the present invention be covered by the claims of the present invention.
Claims (7)
2. a modified cellulose particle according to claim 1, characterized in that the particle diameter is 50 ~ 70 μm.
3. A method for preparing modified cellulose particles, comprising the steps of:
suspending cellulose particles in proper amount of dimethyl sulfoxide (DMSO), adding Carbonyldiimidazole (CDI) which is 0.8 ~ 1.5.5 times of the mass of the cellulose particles, stirring at room temperature for 2 ~ 4 hours, dropwise adding excess hydrazine hydrate solution, continuing the reaction for 12 ~ 24 hours, stopping the reaction, diluting the reaction system with deionized water, dialyzing for 2 ~ 4 days by a dialysis bag, freezing and drying, and storing.
4. The method for preparing modified cellulose particles according to claim 3, wherein the concentration of the hydrazine hydrate solution is 78 to 82%.
5. The method for preparing modified cellulose particles as claimed in claim 3, wherein the molecular weight of the dialysis bag is 2000-4000 kD.
6. The method of claim 3, wherein the purity of dimethyl sulfoxide is not less than 99.9%, and the purity of carbonyldiimidazole is not less than 97.0%.
7. A modified cellulose particle for use in blood perfusion apparatus to remove carbonylated proteins.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201911077821.4A CN110721663A (en) | 2019-11-06 | 2019-11-06 | Modified cellulose particle and preparation method and application thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201911077821.4A CN110721663A (en) | 2019-11-06 | 2019-11-06 | Modified cellulose particle and preparation method and application thereof |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN110721663A true CN110721663A (en) | 2020-01-24 |
Family
ID=69224898
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201911077821.4A Pending CN110721663A (en) | 2019-11-06 | 2019-11-06 | Modified cellulose particle and preparation method and application thereof |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN110721663A (en) |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2008061427A1 (en) * | 2006-11-24 | 2008-05-29 | Bioregen Biomedical (Shanghai) Co., Ltd. | Dihydrazide compounds, preparation and uses thereof |
| US20090269407A1 (en) * | 2008-04-28 | 2009-10-29 | Surmodics, Inc. | Poly-alpha(1-4)glucopyranose-based matrices with hydrazide crosslinking |
| CN103442735A (en) * | 2011-01-31 | 2013-12-11 | 蒂姆·鲍登 | Active principle for mitigating undesired medical conditions in technical field |
-
2019
- 2019-11-06 CN CN201911077821.4A patent/CN110721663A/en active Pending
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2008061427A1 (en) * | 2006-11-24 | 2008-05-29 | Bioregen Biomedical (Shanghai) Co., Ltd. | Dihydrazide compounds, preparation and uses thereof |
| US20090269407A1 (en) * | 2008-04-28 | 2009-10-29 | Surmodics, Inc. | Poly-alpha(1-4)glucopyranose-based matrices with hydrazide crosslinking |
| CN103442735A (en) * | 2011-01-31 | 2013-12-11 | 蒂姆·鲍登 | Active principle for mitigating undesired medical conditions in technical field |
Non-Patent Citations (1)
| Title |
|---|
| VENUKUMAR VEMULA ET AL: "Fluorescence labeling of carbonylated lipids and proteins in cells using coumarin-hydrazide", 《REDOXBIOLOGY》 * |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| AU648655B2 (en) | Adsorbed cellular fibronectin and process for separating and purifying fibronectins | |
| Bereli et al. | Protein recognition via ion-coordinated molecularly imprinted supermacroporous cryogels | |
| EP1519721B1 (en) | Polymer affinity matrix, a method for the production and use thereof | |
| CN109224889B (en) | Blood purification membrane with anticoagulation performance and preparation method thereof | |
| JPS6030291B2 (en) | HGI glycoprotein that promotes differentiation and proliferation of human granulocytes, method for producing HGI glycoprotein, and therapeutic agent for leukopenia containing HGI glycoprotein | |
| US5136032A (en) | Method for separating phosphopolyol compounds using a separating agent | |
| AU2008265838A1 (en) | Device and method for restoration of the condition of blood | |
| BR112013016088B1 (en) | METHOD FOR ISOLATING AND PURIFYING HUMAN SERUM ALBUMIN FROM TRANSGENIC RICE GRAIN | |
| WO2022147847A1 (en) | Adsorbent for removing protein-bound uremic toxins by means of blood perfusion and preparation method therefor | |
| Odabaşı et al. | Pathogenic antibody removal using magnetically stabilized fluidized bed | |
| JP5908496B2 (en) | Method for extracting human serum albumin from the grain of transgenic rice | |
| CN108079974A (en) | A kind of preparation method and adsorbent equipment of western blot polymeric sorbent | |
| CN110721663A (en) | Modified cellulose particle and preparation method and application thereof | |
| CN115490867B (en) | Adsorption resin and preparation method and application thereof | |
| CN109248668B (en) | Adsorbent for removing LDL (low-density lipoprotein) by blood extracorporeal circulation, preparation method thereof and perfusion apparatus | |
| CN108755112B (en) | Antibacterial modification method of high polymer material | |
| Uzun et al. | Poly (hydroxyethyl methacrylate) based affinity membranes for in vitro removal of anti-dsDNA antibodies from SLE plasma | |
| CN113509919B (en) | Adsorbent for removing endotoxin and inflammatory factor in blood of sepsis patient and preparation method thereof | |
| CN114887495A (en) | Preparation method and application of modified dialysis membrane with alkylated composite molybdenum disulfide as modifier and trichosanthes kirilowii pulp starch as coating thickener | |
| JPS5836624B2 (en) | Adsorbent for blood processing | |
| Luo et al. | Study of the allergenic benzypenicilloyl–HSA and its specific separation from human plasma by a pre-designed hybrid imprinted membrane | |
| JPS6087854A (en) | Adsorbent for purifying blood | |
| CN115845822B (en) | Adsorption material for removing blood cytokines and preparation method thereof | |
| CN112755973B (en) | Composite adsorption material applied to blood purification field and preparation method thereof | |
| JP4329421B2 (en) | Materials for microbial adsorption |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| WD01 | Invention patent application deemed withdrawn after publication | ||
| WD01 | Invention patent application deemed withdrawn after publication |
Application publication date: 20200124 |