CN109599552B - Preparation method of silicon-carbon negative electrode material - Google Patents
Preparation method of silicon-carbon negative electrode material Download PDFInfo
- Publication number
- CN109599552B CN109599552B CN201811316861.5A CN201811316861A CN109599552B CN 109599552 B CN109599552 B CN 109599552B CN 201811316861 A CN201811316861 A CN 201811316861A CN 109599552 B CN109599552 B CN 109599552B
- Authority
- CN
- China
- Prior art keywords
- argon
- precursor
- solid
- fusion device
- silicon
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 238000002360 preparation method Methods 0.000 title claims abstract description 22
- HMDDXIMCDZRSNE-UHFFFAOYSA-N [C].[Si] Chemical compound [C].[Si] HMDDXIMCDZRSNE-UHFFFAOYSA-N 0.000 title claims abstract description 17
- 239000007773 negative electrode material Substances 0.000 title claims description 7
- XKRFYHLGVUSROY-UHFFFAOYSA-N Argon Chemical compound [Ar] XKRFYHLGVUSROY-UHFFFAOYSA-N 0.000 claims abstract description 98
- 230000004927 fusion Effects 0.000 claims abstract description 56
- 229910052786 argon Inorganic materials 0.000 claims abstract description 49
- 239000007789 gas Substances 0.000 claims abstract description 41
- 239000000463 material Substances 0.000 claims abstract description 37
- 239000002243 precursor Substances 0.000 claims abstract description 36
- 239000005543 nano-size silicon particle Substances 0.000 claims abstract description 34
- 238000010438 heat treatment Methods 0.000 claims abstract description 27
- 239000011300 coal pitch Substances 0.000 claims abstract description 23
- 239000007790 solid phase Substances 0.000 claims abstract description 23
- HSFWRNGVRCDJHI-UHFFFAOYSA-N alpha-acetylene Natural products C#C HSFWRNGVRCDJHI-UHFFFAOYSA-N 0.000 claims abstract description 22
- 125000002534 ethynyl group Chemical group [H]C#C* 0.000 claims abstract description 22
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 claims abstract description 20
- 239000012071 phase Substances 0.000 claims abstract description 18
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical compound [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 claims abstract description 17
- 238000000034 method Methods 0.000 claims abstract description 11
- 238000001354 calcination Methods 0.000 claims abstract description 10
- 229910052757 nitrogen Inorganic materials 0.000 claims abstract description 10
- 238000001816 cooling Methods 0.000 claims abstract description 9
- 238000007740 vapor deposition Methods 0.000 claims abstract description 9
- 239000010405 anode material Substances 0.000 claims abstract description 8
- 239000007787 solid Substances 0.000 claims description 10
- 238000005303 weighing Methods 0.000 claims description 8
- 238000000576 coating method Methods 0.000 abstract description 25
- 239000011248 coating agent Substances 0.000 abstract description 13
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 abstract description 12
- 229910052799 carbon Inorganic materials 0.000 abstract description 12
- 239000002131 composite material Substances 0.000 abstract description 3
- 239000010410 layer Substances 0.000 description 6
- 238000012986 modification Methods 0.000 description 3
- 230000004048 modification Effects 0.000 description 3
- 239000010406 cathode material Substances 0.000 description 2
- 239000011247 coating layer Substances 0.000 description 2
- 230000000052 comparative effect Effects 0.000 description 2
- 238000010532 solid phase synthesis reaction Methods 0.000 description 2
- 239000011294 coal tar pitch Substances 0.000 description 1
- 150000001875 compounds Chemical class 0.000 description 1
- 239000007791 liquid phase Substances 0.000 description 1
- 230000001105 regulatory effect Effects 0.000 description 1
- 239000012808 vapor phase Substances 0.000 description 1
Classifications
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/62—Selection of inactive substances as ingredients for active masses, e.g. binders, fillers
- H01M4/624—Electric conductive fillers
- H01M4/625—Carbon or graphite
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01M—PROCESSES OR MEANS, e.g. BATTERIES, FOR THE DIRECT CONVERSION OF CHEMICAL ENERGY INTO ELECTRICAL ENERGY
- H01M4/00—Electrodes
- H01M4/02—Electrodes composed of, or comprising, active material
- H01M4/36—Selection of substances as active materials, active masses, active liquids
- H01M4/38—Selection of substances as active materials, active masses, active liquids of elements or alloys
- H01M4/386—Silicon or alloys based on silicon
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E60/00—Enabling technologies; Technologies with a potential or indirect contribution to GHG emissions mitigation
- Y02E60/10—Energy storage using batteries
Landscapes
- Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Electrochemistry (AREA)
- General Chemical & Material Sciences (AREA)
- Battery Electrode And Active Subsutance (AREA)
- Silicon Compounds (AREA)
Abstract
The invention provides a preparation method of a silicon-carbon anode material, which comprises the following steps: 1) adding nano silicon powder into a vapor deposition tube furnace, heating the tube furnace under the protection of argon, closing the argon, introducing acetylene/argon mixed gas, stopping heating after continuous introduction, continuing introducing argon after the mixed gas is closed, and naturally cooling to room temperature to obtain a precursor; 2) adding the precursor and the high-temperature coal pitch into a material fusion device for low-speed fusion; 3) fusing the precursor and the high-temperature coal pitch at high speed in a material fusion device to obtain a solid-phase coated product; 4) and (4) putting the solid-phase coating product into a tubular furnace, and calcining under the protection of nitrogen. The method has the advantages of both a gas phase coating method and a solid phase coating method, can obtain a composite material with a uniform surface carbon layer, and improves the electrochemical performance of the material; but also can reduce the processing cost and is easy to realize industrialization.
Description
Technical Field
The invention relates to the field of battery materials, in particular to a preparation method of a silicon-carbon negative electrode material.
Background
In the current silicon-carbon cathode materials, carbon coating is generally carried out on nano silicon by a gas phase method, a liquid phase method or a solid phase method. The material coating layer obtained by the gas phase method has the highest uniformity and consistency, and the compound has the best performance, but the gas phase method has higher requirements on equipment and technology, so the cost is higher; the solid phase method is easy to operate and low in cost, but the coating layer is low in uniformity and poor in material performance.
Disclosure of Invention
In view of the above, the present invention provides a method for preparing a silicon-carbon negative electrode material having advantages of both a vapor phase coating method and a solid phase coating method.
Therefore, the invention provides a preparation method of a silicon-carbon negative electrode material, which comprises the following preparation steps:
1) weighing nano silicon powder with required mass, adding the nano silicon powder into a vapor deposition tube furnace, heating the tube furnace to 600-1000 ℃ at the speed of 2-10 ℃/min under the protection of argon, then closing the argon to introduce acetylene/argon mixed gas, adjusting the flow rate of the mixed gas to be 50-100 ml/min, then continuously introducing the gas for 2-10 hours, stopping heating, closing the acetylene/argon mixed gas, continuing introducing the argon, and naturally cooling to room temperature to obtain a precursor;
2) adding the precursor and the high-temperature coal pitch into a material fusion device according to the proportion of 50: 50-99: 1, adjusting the rotating speed of the material fusion device to 100-500 r/min, and fusing the precursor and the high-temperature coal pitch in the material fusion device at a low speed for 20-60 min;
3) starting heating, adjusting the temperature of the material fusion device to 90-120 ℃, adjusting the rotating speed of the material fusion device to 500-;
4) and putting the solid phase coating product into a tubular furnace, and calcining at the temperature of 600-1000 ℃ under the protection of nitrogen to obtain the gas-solid two-phase coated nano-silicon.
Further, the preparation method of the silicon-carbon anode material comprises the following preparation steps:
1) weighing nano silicon powder with required mass, adding the nano silicon powder into a vapor deposition tube furnace, heating the tube furnace to 700-1000 ℃ at the speed of 3-9 ℃/min under the protection of argon, then closing the argon to introduce acetylene/argon mixed gas, adjusting the flow rate of the mixed gas to 70-100 ml/min, then continuously introducing the gas for 2-8 hours, stopping heating, closing the acetylene/argon mixed gas, continuing introducing the argon, and naturally cooling to room temperature to obtain a precursor;
2) adding the precursor and the high-temperature coal pitch into a material fusion device according to the proportion of 60: 40-90: 10, adjusting the rotating speed of the material fusion device to 200-400 r/min, and fusing the precursor and the high-temperature coal pitch in the material fusion device at a low speed for 20-50 min;
3) starting heating, adjusting the temperature of the material fusion device to 90-110 ℃, adjusting the rotating speed of the material fusion device to 800-;
4) and putting the solid phase coating product into a tubular furnace, and calcining at the temperature of 800-1000 ℃ under the protection of nitrogen to obtain the gas-solid two-phase coated nano-silicon.
Further, the preparation method of the silicon-carbon anode material comprises the following preparation steps:
1) weighing nano silicon powder with required mass, adding the nano silicon powder into a vapor deposition tube furnace, heating the tube furnace to 800-1000 ℃ at the speed of 4-7 ℃/min under the protection of argon, then closing the argon to introduce acetylene/argon mixed gas, adjusting the flow rate of the mixed gas to 90-100 ml/min, then continuously introducing the gas for 2-4 hours, stopping heating, closing the acetylene/argon mixed gas, continuing introducing the argon, and naturally cooling to room temperature to obtain a precursor;
2) adding the precursor and the high-temperature coal pitch into a material fusion device according to the proportion of 70: 30-85: 15, adjusting the rotating speed of the material fusion device to 250-350 r/min, and fusing the precursor and the high-temperature coal pitch in the material fusion device at a low speed for 20-40 min;
3) starting heating, adjusting the temperature of the material fusion device to 90-100 ℃, adjusting the rotating speed of the material fusion device to 900-;
4) and putting the solid phase coating product into a tubular furnace, and calcining at the temperature of 900-1000 ℃ under the protection of nitrogen to obtain the gas-solid two-phase coated nano-silicon.
Further, the preparation method of the silicon-carbon anode material comprises the following preparation steps:
1) weighing nano silicon powder with required mass, adding the nano silicon powder into a vapor deposition tube furnace, heating the tube furnace to 900 ℃ at the speed of 5 ℃/min under the protection of argon, then closing the argon and introducing acetylene/argon mixed gas, adjusting the flow rate of the mixed gas to be 100 ml/min, then continuously introducing the gas for 2 hours, stopping heating, closing the acetylene/argon mixed gas, continuing introducing the argon, and naturally cooling to room temperature to obtain a precursor;
2) adding the precursor and the high-temperature coal pitch into a material fusion device according to the ratio of 80: 20, adjusting the rotating speed of the material fusion device to 300 revolutions per minute, and fusing the precursor and the high-temperature coal pitch in the material fusion device at a low speed for 20 minutes;
3) starting heating, adjusting the temperature of the material fusion device to 90 ℃, adjusting the rotating speed of the material fusion device to 1000 revolutions per minute, and carrying out high-speed fusion on the precursor and the high-temperature coal pitch in the material fusion device for 20 minutes to obtain a solid-phase coated product;
4) and putting the solid-phase coating product into a tubular furnace, and calcining at 1000 ℃ under the protection of nitrogen to obtain the gas-solid two-phase coated nano-silicon.
Furthermore, in the acetylene/argon mixed gas, the ratio of acetylene to argon is 5: 5-1: 9.
Further, the material fusing device is a mechanical fusing machine.
According to the preparation method of the silicon-carbon cathode material, provided by the invention, carbon coating is carried out on nano silicon by a gas-solid two-phase combined method, and a first carbon layer is coated on the surface of the nano silicon by a gas-phase coating method to obtain a precursor; then coating a second carbon layer on the surface of the precursor by using a solid-phase coating method to complete carbon coating of the nano silicon; the method has the advantages of both a gas phase coating method and a solid phase coating method, can obtain a composite material with a uniform surface carbon layer, and improves the electrochemical performance of the material; but also can reduce the processing cost and is easy to realize industrialization.
Detailed Description
Exemplary embodiments of the present disclosure will be described in more detail below. While exemplary embodiments of the present disclosure have been shown, it should be understood that the present disclosure may be embodied in various forms and should not be limited by the embodiments set forth herein. Rather, these embodiments are provided so that this disclosure will be thorough and complete, and will fully convey the scope of the disclosure to those skilled in the art.
Embodiment one (the most preferred embodiment):
the preparation method of the silicon-carbon anode material provided by the embodiment comprises the following preparation steps:
1) weighing 10g of nano silicon powder, adding the nano silicon powder into a vapor deposition tube furnace, heating the tube furnace to 900 ℃ at the speed of 5 ℃/min under the protection of argon, then closing the argon and introducing acetylene/argon mixed gas, adjusting the flow rate of the mixed gas to be 100 ml/min, then continuously introducing the gas for 2 hours, stopping heating, closing the acetylene/argon mixed gas, continuing introducing the argon, and naturally cooling to room temperature to obtain a precursor A; wherein the ratio of acetylene to argon is 1: 9.
2) Adding the precursor A and the high-temperature coal pitch into a mechanical fusion machine according to the ratio of 80: 20, adjusting the rotating speed of the mechanical fusion machine to 300 revolutions per minute, and fusing the precursor A and the high-temperature coal pitch in the mechanical fusion machine at a low speed for 20 minutes;
3) starting heating, adjusting the temperature of the mechanical fusion machine to 90 ℃, enabling the rotating speed of the mechanical fusion machine to be 1000 r/min, and fusing the precursor A and the high-temperature coal pitch in the mechanical fusion machine at a high speed for 20min to obtain a solid-phase coated product B;
4) and putting the solid-phase coated product B into a tubular furnace, and calcining at 1000 ℃ under the protection of nitrogen to obtain the gas-solid two-phase coated nano-silicon.
Example two:
the preparation method of the silicon-carbon anode material provided by the embodiment comprises the following preparation steps:
1) weighing 10g of nano silicon powder, adding the nano silicon powder into a vapor deposition tube furnace, heating the tube furnace to 600 ℃ at the speed of 2 ℃/min under the protection of argon, then closing the argon and introducing acetylene/argon mixed gas, adjusting the flow rate of the mixed gas to be 50 ml/min, then continuously introducing the gas for 10 hours, stopping heating, closing the acetylene/argon mixed gas, continuing introducing the argon, and naturally cooling to room temperature to obtain a precursor A; wherein the ratio of acetylene to argon is 5: 5.
2) Adding the precursor A and the high-temperature coal pitch into a mechanical fusion machine according to the proportion of 50: 50, adjusting the rotating speed of the mechanical fusion machine to 100 revolutions per minute, and fusing the precursor A and the high-temperature coal pitch in the mechanical fusion machine at a low speed for 60 minutes;
3) starting heating, adjusting the temperature of the mechanical fusion machine to 120 ℃, enabling the rotating speed of the mechanical fusion machine to be 500 r/min, and fusing the precursor A and the high-temperature coal pitch in the mechanical fusion machine at a high speed for 15 min to obtain a solid-phase coated product B;
4) and (3) putting the solid-phase coated product B into a tubular furnace, and calcining at 600 ℃ under the protection of nitrogen to obtain the gas-solid two-phase coated nano-silicon.
Example three:
the preparation method of the silicon-carbon anode material provided by the embodiment comprises the following preparation steps:
1) weighing 10g of nano silicon powder, adding the nano silicon powder into a vapor deposition tube furnace, heating the tube furnace to 800 ℃ at the speed of 7 ℃/min under the protection of argon, then closing the argon and introducing acetylene/argon mixed gas, adjusting the flow rate of the mixed gas to 70 ml/min, then continuously introducing the gas for 8 hours, stopping heating, closing the acetylene/argon mixed gas, continuing introducing the argon, and naturally cooling to room temperature to obtain a precursor A; wherein the ratio of acetylene to argon is 8: 2.
2) Adding the precursor A and the high-temperature coal pitch into a mechanical fusion machine according to the ratio of 99: 1, adjusting the rotating speed of the mechanical fusion machine to 400 revolutions per minute, and fusing the precursor A and the high-temperature coal pitch in the mechanical fusion machine at a low speed for 40 minutes;
3) starting heating, adjusting the temperature of the mechanical fusion machine to 100 ℃, enabling the rotating speed of the mechanical fusion machine to reach 900 revolutions per minute, and fusing the precursor A and the high-temperature coal pitch in the mechanical fusion machine at a high speed for 25 minutes to obtain a solid-phase coated product B;
4) and putting the solid-phase coating product B into a tubular furnace, and calcining at 800 ℃ under the protection of nitrogen to obtain the gas-solid two-phase coated nano-silicon.
Comparative example (prior art):
1) adding the nano silicon and the high-temperature coal tar pitch into a mechanical fusion machine according to the ratio of 80: 20, and regulating the rotating speed of the fusion machine to be 300 revolutions per minute for low-speed fusion for 20 minutes.
2) And starting heating, adjusting the temperature of the fusion machine to 90 ℃, adjusting the rotating speed of the fusion machine to 1000 revolutions per minute, and performing high-speed fusion for 20min to obtain a solid-phase coated product.
3) Putting the product obtained in the step 2) into a tubular furnace, and calcining at 1000 ℃ under the protection of nitrogen to obtain the solid-phase coated nano-silicon.
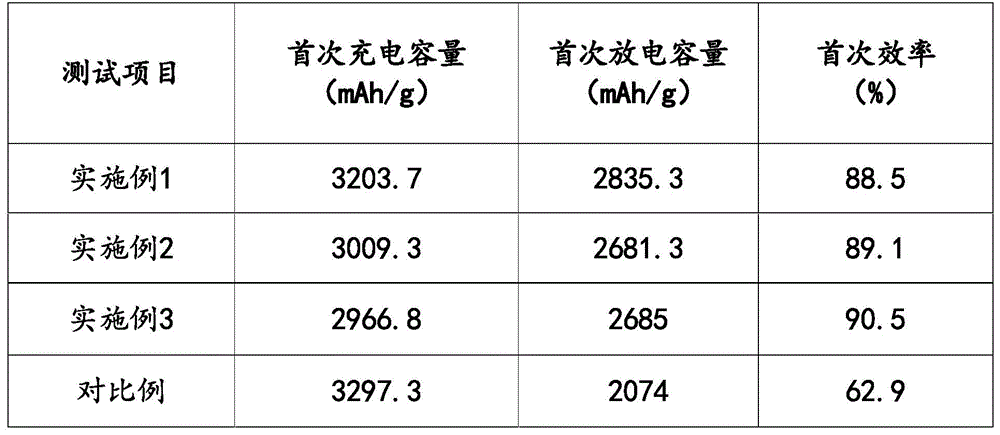
The experimental data of the first to third examples are compared with the comparative examples as follows:
therefore, in the preparation method of the silicon-carbon negative electrode material provided by the embodiment, carbon coating is performed on nano silicon by a gas-solid two-phase combination method, and a first carbon layer is coated on the surface of the nano silicon by a gas-phase coating method to obtain a precursor; then coating a second carbon layer on the surface of the precursor by using a solid-phase coating method to complete carbon coating of the nano silicon; the method has the advantages of both a gas phase coating method and a solid phase coating method, can obtain a composite material with a uniform surface carbon layer, and improves the electrochemical performance of the material; but also can reduce the processing cost and is easy to realize industrialization.
It will be apparent to those skilled in the art that various changes and modifications may be made in the present invention without departing from the spirit and scope of the invention. Thus, if such modifications and variations of the present invention fall within the scope of the claims of the present invention and their equivalents, the present invention is also intended to include such modifications and variations.
Claims (2)
1. The preparation method of the silicon-carbon negative electrode material is characterized by comprising the following preparation steps of:
1) weighing nano silicon powder with required mass, adding the nano silicon powder into a vapor deposition tube furnace, heating the tube furnace to 900 ℃ at the speed of 5 ℃/min under the protection of argon, then closing the argon and introducing acetylene/argon mixed gas, adjusting the flow rate of the mixed gas to be 100 ml/min, then continuously introducing the gas for 2 hours, stopping heating, closing the acetylene/argon mixed gas, continuing introducing the argon, and naturally cooling to room temperature to obtain a precursor A;
2) adding the precursor A and the high-temperature coal pitch into a material fusion device according to the ratio of 80: 20, adjusting the rotating speed of the material fusion device to 300 revolutions per minute, and fusing the precursor A and the high-temperature coal pitch in the material fusion device at a low speed for 20 minutes;
3) starting heating, adjusting the temperature of the material fusion device to 90 ℃, adjusting the rotating speed of the material fusion device to 1000 revolutions per minute, and performing high-speed fusion on the precursor A and the high-temperature coal pitch in the material fusion device for 20 minutes to obtain a solid-phase coated product B;
4) and putting the solid-phase coated product B into a tubular furnace, and calcining at 1000 ℃ under the protection of nitrogen to obtain the gas-solid two-phase coated nano-silicon.
2. The method for preparing the silicon-carbon anode material according to claim 1, wherein the material fusing device is a mechanical fusing machine.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201811316861.5A CN109599552B (en) | 2018-11-07 | 2018-11-07 | Preparation method of silicon-carbon negative electrode material |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201811316861.5A CN109599552B (en) | 2018-11-07 | 2018-11-07 | Preparation method of silicon-carbon negative electrode material |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN109599552A CN109599552A (en) | 2019-04-09 |
| CN109599552B true CN109599552B (en) | 2022-02-18 |
Family
ID=65958578
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201811316861.5A Active CN109599552B (en) | 2018-11-07 | 2018-11-07 | Preparation method of silicon-carbon negative electrode material |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN109599552B (en) |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN112421002B (en) * | 2020-11-10 | 2022-03-29 | 成都爱敏特新能源技术有限公司 | High-capacity silicon-carbon material and preparation method thereof |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102891297A (en) * | 2012-11-10 | 2013-01-23 | 江西正拓新能源科技有限公司 | Silicon-carbon composite material for lithium ion battery and preparation method thereof |
| CN103199254A (en) * | 2013-04-03 | 2013-07-10 | 深圳市贝特瑞新能源材料股份有限公司 | Graphite negative material of lithium-ion battery and preparation method of negative material |
| CN103647056A (en) * | 2013-11-29 | 2014-03-19 | 深圳市贝特瑞新能源材料股份有限公司 | SiOx based composite negative electrode material, preparation method and battery |
| CN107768626A (en) * | 2017-09-30 | 2018-03-06 | 深圳市贝特瑞新能源材料股份有限公司 | A kind of high power capacity rate C-base composte material, its preparation method and the purposes in lithium ion battery |
| CN108736007A (en) * | 2018-08-20 | 2018-11-02 | 赣州市瑞富特科技有限公司 | A kind of preparation method of high compacted density lithium ion battery silicon-carbon cathode material |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN103985867B (en) * | 2014-05-19 | 2016-08-24 | 电子科技大学 | A kind of method preparing carbon cladding ferrosilite lithium composite material |
-
2018
- 2018-11-07 CN CN201811316861.5A patent/CN109599552B/en active Active
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102891297A (en) * | 2012-11-10 | 2013-01-23 | 江西正拓新能源科技有限公司 | Silicon-carbon composite material for lithium ion battery and preparation method thereof |
| CN103199254A (en) * | 2013-04-03 | 2013-07-10 | 深圳市贝特瑞新能源材料股份有限公司 | Graphite negative material of lithium-ion battery and preparation method of negative material |
| CN103647056A (en) * | 2013-11-29 | 2014-03-19 | 深圳市贝特瑞新能源材料股份有限公司 | SiOx based composite negative electrode material, preparation method and battery |
| CN107768626A (en) * | 2017-09-30 | 2018-03-06 | 深圳市贝特瑞新能源材料股份有限公司 | A kind of high power capacity rate C-base composte material, its preparation method and the purposes in lithium ion battery |
| CN108736007A (en) * | 2018-08-20 | 2018-11-02 | 赣州市瑞富特科技有限公司 | A kind of preparation method of high compacted density lithium ion battery silicon-carbon cathode material |
Also Published As
| Publication number | Publication date |
|---|---|
| CN109599552A (en) | 2019-04-09 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US11851332B2 (en) | Silicon-carbon composite material and preparation method thereof | |
| Sun et al. | A review on recent advances for boosting initial coulombic efficiency of silicon anodic lithium ion batteries | |
| KR102798668B1 (en) | Cathode and cathode materials for lithium sulfur batteries | |
| US10566613B2 (en) | Negative electrode material for lithium-ion secondary battery, negative electrode for lithium-ion secondary battery, lithium-ion secondary battery, and method of producing negative electrode material for lithium-ion secondary battery | |
| Abdul Ahad et al. | Cu Current Collector with Binder‐Free Lithiophilic Nanowire Coating for High Energy Density Lithium Metal Batteries | |
| CN110589793B (en) | Metal-doped and Mxene-coated double-modified lithium iron phosphate composite material, and preparation method and application thereof | |
| CN106374081A (en) | High-capacity lithium ion battery negative electrode plate and preparation method thereof | |
| Li et al. | High interfacial‐energy and lithiophilic janus interphase enables stable lithium metal anodes | |
| WO2022236985A1 (en) | Uniformly modified silicon monoxide negative electrode material, and preparation method therefor and use thereof | |
| JP2024515911A (en) | Carbon-coated composite materials, their manufacture and use | |
| Zhang et al. | Orchestrating a Controllable Engineering of Dual‐Model Carbon Structure in Si/C Anodes | |
| CN109390548A (en) | Secondary battery negative pole, preparation method and secondary cell | |
| Li et al. | Coal tar electrode pitch modified rice husk ash as anode for lithium ion batteries | |
| CN109599552B (en) | Preparation method of silicon-carbon negative electrode material | |
| CN117199511A (en) | Solid electrolyte with core-shell structure, preparation method thereof and solid battery | |
| CN119517975A (en) | A pre-lithiation silicon-carbon negative electrode material and its preparation method and application | |
| CN110048092B (en) | Lithium battery silicon-carbon composite material and preparation method thereof | |
| CN111933919A (en) | Nano silicon powder, silicon-based negative electrode, lithium ion battery containing silicon-based negative electrode and manufacturing method of lithium ion battery | |
| JP6079651B2 (en) | Method for producing negative electrode material for non-aqueous electrolyte secondary battery | |
| CN119361631A (en) | Negative electrode material and preparation method and application thereof | |
| Li et al. | Si anode with high initial Coulombic efficiency, long cycle life, and superior rate capability by integrated utilization of graphene and pitch-based carbon | |
| CN115483380B (en) | Hybrid phenolic resin-based amorphous carbon@C composites and their preparation and application in sodium secondary batteries | |
| Fujikake et al. | Amorphous Ge/LiAlGePO Composite Anodes with a Multistacking Structure Developed via Co‐Sputtering for High‐Capacity Li+‐Ion Batteries | |
| CN109301216A (en) | A kind of preparation method of carbon-boron composite ball-coated lithium iron phosphate electrode | |
| JP2018206594A (en) | Method for producing negative electrode active material for non-aqueous electrolyte secondary battery |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant |