Method for producing chemical products by using catalytic cracking gasoline
Technical Field
The invention belongs to the technical field of petrochemical industry, and particularly relates to a method for producing chemical products by using catalytic cracking gasoline.
Background
With the increasing weight of petroleum resources and the increasing influence of automobile exhaust emission on the atmospheric environment, the requirements on the quality of the motor gasoline are increasingly strict worldwide. The national VI automotive gasoline standard which is about to be implemented in 2019, 1 and 1 requires that the sulfur content of gasoline is lower than 10ppm, the olefin content is lower than 15v percent, and the octane number is maintained to be more than 93. Thus, the production of high quality gasoline is marked by an increase in octane number with a simultaneous decrease in the sulfur content and the olefin content.
At present, aiming at the production of low-sulfur, low-alkene and high-octane gasoline, developed countries mainly achieve corresponding quality standards by blending gasoline produced by various processes; generally, the catalytic cracking gasoline containing olefin is less than 1/3, the reformed gasoline containing aromatic hydrocarbon but no olefin is more than 1/3, and the other clean gasoline components such as alkylation, isomerization and etherification containing no olefin and no aromatic hydrocarbon are less than 1/3, and the blended gasoline has low sulfur content and olefin content and high octane number.
The catalytic cracking gasoline (i.e. FCC gasoline) is the main component of motor gasoline in China, and accounts for about 75% of gasoline pools. The catalytic cracking gasoline has the characteristics of high olefin content (30-45 v%), high sulfur content (150-1000ppm) and the like. As is known, 85-95 wt% of sulfur and 95 v% of olefin in the commodity gasoline in China come from catalytic cracking gasoline, which is the main reason that the gasoline for vehicles in China is difficult to meet the requirements that the sulfur content is lower than 10ppm and the olefin content is lower than 15-18 v%.
China mainly depends on a hydrodesulfurization technology aiming at the processing of catalytic cracking gasoline, the technology can meet the requirements that the sulfur content is lower than 10ppm and the olefin content is reduced, however, the olefin content is reduced in a mode of changing hydrogenation saturation into alkane, so that the octane number of a gasoline product is reduced, the aim of desulfurizing, reducing the olefin and protecting the octane number cannot be met, and in addition, the economic benefit of an enterprise is seriously influenced.
In the hydrodesulfurization process, octane number loss caused by olefin saturation cannot be effectively inhibited by optimizing a catalyst or process conditions of the hydrodesulfurization process, and olefin components are prevented from entering a hydrodesulfurization unit from a hydrogenation raw material, so that the method is a fundamental method for protecting olefins. At present, relevant research at home and abroad mainly focuses on distillation cutting of FCC gasoline and hydrotreatment of high-sulfur components, and the mode can prevent high-octane olefins from being subjected to hydrogenation saturation, but cannot convert the high-octane olefins into high-octane aromatics.
Meanwhile, the market demand for C5 components, ethylene feedstock, and chemical products such as benzene, toluene, and xylene (BTX for short) is extremely large. Wherein, the C5 component can be directly used for converting BTX products, isoprene formed by separating the C5 component can be used for synthesizing rubber and the like, and the C5 component can also be used as industrial raw materials in various chemical fields; in addition, the ethylene raw material and BTX can be directly used as chemical production raw materials, and have great market value. However, the existing research mainly focuses on producing gasoline blending components, and cannot be converted into various chemical products urgently needed by the market. Therefore, how to efficiently produce high-value chemical products by using the catalytic cracking gasoline becomes a problem to be solved urgently.
Disclosure of Invention
The invention provides a method for producing chemical products by using catalytic cracking gasoline, which can efficiently convert the catalytic cracking gasoline into high-value chemical products.
The invention provides a method for producing chemical products by using catalytic cracking gasoline, which comprises the following steps:
pre-hydrogenating the catalytic cracking gasoline to obtain pre-hydrogenated catalytic cracking gasoline;
cutting the prehydrogenated catalytically cracked gasoline into light, medium and heavy fractions;
performing solvent extraction on the middle distillate to obtain raffinate oil rich in olefin and extract oil rich in aromatic hydrocarbon;
carrying out mild aromatization on the raffinate oil to obtain an aromatization product;
recovering light olefin from the extracted oil to obtain light olefin and sulfur-rich oil;
carrying out selective hydrodesulfurization on the heavy fraction and the sulfur-rich oil to obtain a desulfurized heavy fraction;
and (3) performing aromatic extraction or extractive distillation on the aromatization product and the desulfurized heavy fraction.
In the invention, the catalytic cracking gasoline is pre-hydrogenated and is used for reacting light sulfides in the catalytic cracking gasoline with diolefins to form high-boiling sulfides, thereby avoiding the conditions of coking and the like in the subsequent process of the diolefins; during the pre-hydrogenation process, the olefins in the catalytically cracked gasoline are not saturated.
The prehydrogenation process is not critical to the present invention, and for example, the Prime-G + prehydrogenation process conventional in the art may be used.
In a particular embodiment of the invention, the prehydrogenation is carried out in the presence of a prehydrogenation catalyst; among them, the prehydrogenation catalyst may be a nickel-molybdenum bimetallic catalyst as is conventional in the art. Specifically, the composition of the nickel-molybdenum bimetallic catalyst may be, for example (% by mass): al (Al)2O390.5 percent of (carrier), 6 percent of nickel and 3.5 percent of molybdenum; the pre-hydrogenation process conditions are not strictly limited, for example, the pre-hydrogenation temperature can be controlled to be about 130 ℃, the hydrogen-oil ratio is about 5, and the volume space velocity is 3h-1Left and right.
In the invention, the cutting is to cut the prehydrogenation catalytic cracking gasoline into light, medium and heavy fractions according to the distillation range from low to high; the cleavage may be performed by a method conventional in the art, and for example, a distillation cleavage or the like may be used.
In the specific scheme of the invention, the cutting temperature of the light fraction and the middle fraction is 40-60 ℃, and the cutting temperature of the middle fraction and the heavy fraction is 100-160 ℃; namely, the distillation range of the middle distillate is 40-60 ℃ to 100-160 ℃.
The research shows that the hydrocarbon distribution in the catalytic cracking gasoline has the following characteristics: 1) the light fraction mainly contains low-carbon (such as C5) olefin and alkane, wherein the mass content of the olefin is more than 50%, and the light fraction can be directly used as a chemical raw material for comprehensive utilization; 2) the middle distillate mainly contains C6-C8 olefins, aromatic hydrocarbons, alkanes and the like, is the lowest part of octane number, and has the RON below 80 (a small part even below 70); 3) the heavy fraction mainly contains high-carbon aromatic hydrocarbon and polycyclic aromatic hydrocarbon above C9, the olefin content is lower, and the aromatic hydrocarbon and sulfur content is higher; 4) the octane number distribution of the narrow cut of catalytically cracked gasoline first decreases significantly to the lowest point with increasing carbon number or boiling range and then mostly rises back.
Based on the research results, the method combines distillation cutting, solvent extraction, mild aromatization, selective hydrodesulfurization and aromatic extraction or extractive rectification; wherein, the light and medium fractions formed by distillation and cutting can be well enriched with the olefin to be treated; the olefin and the aromatic hydrocarbon can be separated by adopting the solvent extraction to carry out directional separation on the middle distillate; the mild aromatization can efficiently convert the olefins in the middle distillate into aromatic hydrocarbons; the recovery and utilization of the light olefin can improve the yield of the C5 component and improve the separation effect of the solvent on the olefin and the aromatic hydrocarbon; the sulfur content of the heavy fraction can be reduced by carrying out selective hydrodesulfurization on the heavy fraction, and the octane number loss is reduced; after the selective hydrodesulfurization, the aromatic hydrocarbons are separated by adopting aromatic hydrocarbon extraction or extractive distillation according to the difference of the solubility or relative volatility between the aromatic hydrocarbons and other substances, so that the high-efficiency and directional separation of the aromatic hydrocarbons is realized, and thus chemical products such as ethylene raw materials/gasoline components and BTX are obtained.
In the invention, the ethylene raw material/gasoline component refers to the same component, which can be used as a chemical product ethylene raw material and a gasoline component and can be applied according to actual needs.
The combination mode of the invention can convert the catalytic cracking gasoline into high-value chemical products such as C5 components, ethylene raw materials/gasoline components, BTX and the like, thereby overcoming the defects that the chemical products cannot be prepared or obtained in the prior art and the like and realizing the aim of efficiently converting the catalytic cracking gasoline into the high-value chemical products.
In the invention, the solvent extraction mainly utilizes the solvent to realize the directional separation of the aromatic hydrocarbon from the olefin, the alkane and the cyclane, thereby being beneficial to the subsequent production of a series of chemical products. The solvent used for solvent extraction is not limited in the present invention, as long as the above directional separation can be achieved, and for example, one or a combination of two or more of diethylene glycol, triethylene glycol, tetraethylene glycol, dimethyl sulfoxide, sulfolane, N-formylmorpholine, N-methylpyrrolidone, polyethylene glycol, propylene carbonate, and the like can be used.
In a specific embodiment of the present invention, the solvent extraction is performed using a composite solvent of sulfolane and N-methylpyrrolidone (i.e., using the composite solvent as a solvent), wherein the volume content of N-methylpyrrolidone in the composite solvent is 5-40%, and further 5-20%.
The research shows that: when the composite solvent is adopted for solvent extraction, the mass content of olefin in raffinate oil is 40-45%, and the mass content of aromatic hydrocarbon in extract oil is 60-70%; when the sulfolane single solvent is adopted for solvent extraction, the mass content of olefin in raffinate oil is 35-40%, and the mass content of aromatic hydrocarbon in extract oil is 50-55%. The composite solvent has high selectivity, and when the composite solvent is used for solvent extraction, the mass content of olefin in raffinate oil is increased by about 5 percent, and the mass content of aromatic hydrocarbon in extract oil is increased by about 10-15 percent, so that the directional separation of the aromatic hydrocarbon and the olefin in middle distillate is facilitated.
In the present invention, solvent extraction may be carried out in a manner conventional in the art; specifically, the solvent extraction may include:
the middle distillate enters from the middle lower part of the extraction tower, the solvent enters from the upper part of the extraction tower, the temperature of the top of the extraction tower is controlled to be 55-100 ℃, the temperature of the bottom of the extraction tower is 40-80 ℃, the pressure (gauge pressure) of the top of the extraction tower is 0.2-0.7MPa, and the feeding ratio of the solvent to the middle distillate (namely the feeding volume ratio of the solvent to the middle distillate) is 1.0-5.0.
In the present invention, mild aromatization is aromatization carried out under relatively mild conditions (low temperature, normal pressure) and is mainly used for converting olefins in the raffinate oil into aromatic hydrocarbons.
Specifically, the mild aromatization of the present invention may be carried out in the presence of an aromatization catalyst; the aromatization catalyst is obtained by loading active ingredients on a carrier. Further, the carrier of the aromatization catalyst comprises a zeolite molecular sieve and pseudoboehmite, the active ingredients comprise a first ingredient, a second ingredient, a third ingredient and a fourth ingredient, the first ingredient is Na or K, the second ingredient is P, the third ingredient is Zn, the fourth ingredient is La, the loading amount of the first ingredient is 0.2-0.5%, the loading amount of the second ingredient is 1-3%, the loading amount of the third ingredient is 5-8%, and the loading amount of the fourth ingredient is 0-3%.
Further, the mass ratio of the zeolite molecular sieve to the pseudo-boehmite in the carrier of the aromatization catalyst may be (1-9): 1; the zeolite molecular sieve may be, for example, an HZSM-5 molecular sieve. Further, the supported amount of the fourth component is further 1.5 to 3%.
In the present invention, the above-mentioned method for preparing an aromatization catalyst comprises the following steps in order:
mixing a zeolite molecular sieve with pseudo-boehmite to obtain a catalyst precursor;
carrying out ion exchange modification on the catalyst precursor to enable the first component to be loaded on the catalyst precursor;
subjecting the catalyst precursor to a first modification treatment so that the second component or the fourth component is supported on the catalyst precursor;
carrying out active metal modification on the catalyst precursor to enable a third component to be loaded on the catalyst precursor;
the catalyst precursor is subjected to a second modification treatment to support a second component on the catalyst precursor.
Further, the ion exchange modification may include:
the catalyst precursor is subjected to ion exchange modification using a salt solution or an alkali solution containing sodium ions or potassium ions as the ion exchange solution, the ion exchange modification being controlled to last at 60-120 ℃ for at least 30 minutes, then dried at 60-280 ℃ for at least 3 hours, and finally calcined at 450-700 ℃ for at least 1 hour.
The first modification treatment, the active metal modification and the second modification treatment are not strictly limited, and conventional methods in the field, such as an isometric immersion method, can be adopted; further, the modification conditions may be conventional in the art.
In the invention, the temperature of the mild aromatization can be controlled to be 280-330 ℃, the pressure is normal pressure, and the volume space velocity is 1.0-2.0h-1. The mild aromatization can be carried out by adopting a fixed bed reactor, and can be carried out under the conditions of hydrogenation and non-hydrogenation; preferably under non-hydrogen conditions.
The mild aromatization of the invention is easy to realize by adopting the aromatization catalyst and aromatization conditions, so that the olefins in the raffinate oil are converted into the aromatic hydrocarbons.
The mild aromatization of the invention is carried out by adopting the aromatization catalyst, the aromatization catalyst has high aromatization activity, the aromatization reaction can be carried out under the mild conditions of temperature not higher than 400 ℃ (particularly not higher than 330 ℃) and normal pressure, the selectivity of olefin selective conversion into aromatic hydrocarbon is higher, the selectivity can reach more than 60% under the condition that the liquid yield is ensured to be higher than 98.5%, the generated aromatic hydrocarbon is mainly C7-C9 aromatic hydrocarbon, reaches about 90%, and the generation rate of benzene is lower; in addition, the aromatization catalyst has good anti-carbon deposition capability, so that the service life and the stability are longer, the single-pass activity is 8-10 days (the activity of olefin conversion rate is maintained to be more than 50 percent), and the liquid yield is maintained to be more than 98.5 percent, so that the oil loss is effectively avoided, and the long-period stable operation can be kept.
The mild aromatization of the invention not only can effectively reduce the olefin content in the aromatization product, obviously improve the aromatic hydrocarbon content, especially the C7-C9 aromatic hydrocarbon content and lower the benzene content, and achieve the effects of low olefin, high octane number and low benzene content, thereby being beneficial to obtaining high-quality gasoline meeting national VI standards, but also being beneficial to saving production energy consumption, having very good industrial amplification adaptability, and being really used in actual industrial production.
In the invention, the light olefin (light olefin for short) can be recovered from the extracted oil by adopting a conventional mode; wherein the recovered light olefins comprise primarily C5 olefins.
Specifically, the light olefin recovery may be carried out in a recovery column, wherein the overhead temperature of the recovery column may be controlled to be 80 to 95 ℃, the overhead pressure may be controlled to be 0.05 to 0.2MPa, the bottom temperature may be controlled to be 150-.
In the present invention, the recovered light olefins may be subjected to a subsequent treatment as required. In particular, at least a portion of the light olefins may be returned to the solvent extraction, i.e.: returning at least a portion of the light olefins to the system in which the solvent extraction is conducted for backwashing (stripping); at this point, another portion of the light olefins may be combined into the light fraction, thereby increasing the yield of C5 components. The amount of the light olefin returned to the solvent extraction is not strictly limited, and the amount of the light olefin returned to the solvent extraction may be 20 to 100% (by volume, 100% means that the whole is returned to the solvent extraction) of the amount of the light olefin recovered, and further may be 40 to 60%.
The research finds that: in the solvent extraction process, the smaller the carbon number of the same hydrocarbon is, the higher the solubility of the solvent to the solvent is, and the larger the carbon number is, the opposite is realized (namely, the lower olefin has higher solubility in the solvent); the method returns the light olefin to the solvent extraction step, and can separate and replace the high-carbon olefin into the raffinate oil, thereby obtaining the raffinate oil with higher olefin content and the extract oil with higher aromatic hydrocarbon content, and the purity of the output C5 olefin can be improved after repeated recovery for many times, thereby obviously improving the separation effect of the solvent on the olefin and the aromatic hydrocarbon, and simultaneously effectively protecting the olefin components.
The research shows that: after the light olefin is returned to be subjected to solvent extraction, the mass content of the olefin in the raffinate oil is 45-50%; and when the light olefin is not returned to carry out solvent extraction, the mass content of the olefin in the raffinate oil is 40-45 percent. The light olefin is returned to be subjected to solvent extraction, so that the mass content of the olefin in the raffinate oil can be improved by about 5 percent, and the separation effect of the olefin and the aromatic hydrocarbon is further improved.
In the present invention, selective hydrodesulfurization can be carried out in a manner conventionally realized in the prior art, for example, selective desulfurization techniques such as S-zorb, RSDS, OCT-M, Prime-G +, CODS, etc., or other selective deep desulfurization techniques can be adopted.
In a specific scheme of the invention, the selective hydrodesulfurization is carried out in the presence of a selective hydrodesulfurization catalyst, a carrier of the selective hydrodesulfurization catalyst is selected from at least one of a molecular sieve and a metal oxide, an active ingredient comprises Co and Mo, and the total mass content of the Co and the Mo in the selective hydrodesulfurization catalyst is 5-20%; the temperature of the selective hydrodesulfurization is 200--1The hydrogen to oil volume ratio (i.e., the ratio of the volume of hydrogen to the total volume of the heavy fraction and the sulfur-rich oil) is 300-600. Desulfurized heavy fraction, sulfur, obtained by said selective hydrodesulfurizationThe content is 10ppm or less.
In the invention, aromatic extraction and extractive distillation are used for separating aromatic hydrocarbon in aromatization products and desulfurized heavy fraction; when aromatic extraction and extractive distillation are carried out on the aromatic structured product and the desulfurized heavy fraction, the same solvent as the solvent extraction can be adopted; the extractive distillation is preferably carried out by adopting a composite solvent of sulfolane and N-methyl pyrrolidone, wherein the volume content of the N-methyl pyrrolidone in the composite solvent can be 5-40%, so that the separation of the aromatic hydrocarbon and the olefin is facilitated.
Specifically, the aromatic extraction may be carried out in a manner conventional in the art, and may be carried out, for example, by the method disclosed in publication No. CN 104694158A.
Further, the extractive distillation may include: enabling the aromatization product and the desulfurized heavy fraction to enter from the upper part of the extractive distillation tower, and enabling the solvent to enter from the lower part of the extractive distillation tower; wherein the theoretical plate number of the extractive distillation column is 20-25, the temperature of the column bottom is 140-160 ℃, the feeding ratio (solvent-oil ratio, namely the ratio of the volume of the solvent to the total volume of the aromatization product and the desulfurization heavy fraction) of the solvent to the aromatization product and the desulfurization heavy fraction is 1.0-5.0, and the reflux ratio is 0.1-4.0.
Further, the extract obtained by extractive distillation may be subjected to light olefin (mainly including C5 olefin) recovery, and the recovered light olefin is returned to the mild aromatization or to the extractive distillation; specifically, the yield of BTX can be increased by 2-4 percentage points when returning light olefins to the mild aromatization; when the light olefin is returned to the extractive distillation, the yield of the ethylene raw material can be improved by 1 to 2 percentage points.
In view of the limited dissolving capacity of the solvent, the light olefins are returned to be subjected to mild aromatization or extractive distillation, so that the high-carbon olefins dissolved in the solvent can be replaced into the light components at the top of the tower, and the purity and the yield of the ethylene raw material and BTX are further improved.
Specifically, the light olefin recovery may be carried out in a recovery column, and the overhead temperature of the recovery column may be controlled to be 80 to 95 ℃, the overhead pressure to be 0.05 to 0.2MPa, the bottom temperature to be 150-.
In the present invention, unless otherwise specified, the pressure means a gauge pressure; the content means mass content.
The invention divides the catalytic cracking gasoline into light, middle and heavy fractions by researching the distribution rules of hydrocarbon composition, sulfide, octane number of narrow fraction and the like in the catalytic cracking gasoline; the directional separation of the olefin and the aromatic hydrocarbon is realized by adopting a solvent extraction mode aiming at the distribution rule of the olefin, the aromatic hydrocarbon and the sulfide in the middle distillate, and simultaneously the recovery and the utilization of the extracted oil light olefin greatly improve the separation efficiency of the olefin and the yield of the C5 component; aiming at the raffinate oil rich in olefin, the raffinate oil can be converted into aromatic hydrocarbon with higher octane number through mild aromatization, the method not only can meet the requirement of reducing the olefin according to the national VI standard, but also can produce BTX products while keeping the octane number; in addition, according to the difference of solubility or relative volatility between the aromatic hydrocarbon and other substances, the aromatic hydrocarbon is separated by adopting aromatic hydrocarbon extraction or extractive distillation, so that the efficient and directional separation of the aromatic hydrocarbon is realized. The method of the invention utilizes the catalytic cracking gasoline to simultaneously produce high-value chemical products such as BTX, ethylene raw materials, C5 components and the like on the basis of sulfur reduction and alkene reduction, and has good application prospect.
Drawings
FIG. 1 is a flow diagram of one embodiment of a process for producing chemical products from catalytically cracked gasoline;
FIG. 2 is a flow chart of another embodiment of a process for producing chemical products from catalytically cracked gasoline;
FIG. 3 is a flow chart illustrating a process for producing chemical products from catalytically cracked gasoline according to still another embodiment.
Detailed Description
In order to make the objects, technical solutions and advantages of the present invention clearer, the technical solutions in the embodiments of the present invention will be clearly and completely described below with reference to the drawings and the embodiments of the present invention. All other embodiments, which can be derived by a person skilled in the art from the embodiments given herein without making any creative effort, shall fall within the protection scope of the present invention.
Example 1
1. Preparation of aromatization catalyst
1) Preparation of catalyst precursor
Under the room temperature environment, the ratio of silicon to aluminum is 25, the HZSM-5 molecular sieve and the pseudo-boehmite are mixed according to the ratio of 4: 1 to obtain the catalyst precursor.
2) Modification by ion exchange
The catalyst precursor is subjected to ion exchange treatment by adopting a constant-temperature water bath method, specifically, sodium hydroxide is dissolved in deionized water, is mixed with the catalyst precursor, is placed in a 90 ℃ water bath environment and is stirred for 2 hours, so that the loading capacity of sodium is about 0.2 wt%, and then is dried for about 8 hours at about 120 ℃ and is roasted for about 4 hours at about 540 ℃.
3) First modification treatment
Adopting an isometric impregnation method to carry out first modification treatment on the catalyst precursor subjected to ion exchange treatment, specifically dissolving ammonium dihydrogen phosphate in deionized water, then impregnating the catalyst precursor, and controlling the mass ratio of an aqueous solution of ammonium dihydrogen phosphate to the catalyst precursor to be (1.0 +/-0.2): 1, the loading amount of phosphorus is about 1 wt%; after the impregnation is completed, the mixture is aged at a temperature of about 20 ℃ for about 6 hours, dried at about 120 ℃ for about 8 hours, and calcined at about 540 ℃ for about 4 hours.
4) Reactive metal modification
The catalyst precursor subjected to the first modification treatment is subjected to hydrothermal treatment at a temperature of about 300 ℃ in a 100% steam atmosphere for about 6 hours, and then the catalyst precursor subjected to the hydrothermal treatment is subjected to active metal loading by an isometric immersion method: dissolving zinc nitrate in a citric acid solution with the concentration of about 0.1mol/L to obtain a steeping fluid; controlling the mass ratio of the impregnation liquid to the catalyst precursor to be (1.0 +/-0.2): 1. the dipping temperature is about 20 ℃, the dipping time is about 10 hours, and the loading amount of zinc is about 5 wt%; after the impregnation is completed, the mixture is aged at about 25 ℃ for 4 hours, dried at about 120 ℃ in an air atmosphere for about 10 hours, and baked at 540 ℃ for about 4 hours.
5) Second modification treatment
And (3) performing second modification treatment on the catalyst precursor subjected to active metal loading by adopting an isometric impregnation method, and referring to the step 3) to obtain the aromatization catalyst.
The aromatization catalyst comprises a zeolite molecular sieve (HZSM-5 molecular sieve) and pseudo-boehmite, wherein the mass ratio of the zeolite molecular sieve to the pseudo-boehmite is 4: 1; the active ingredients comprise Na, P and Zn, wherein the loading amount of Na is 0.2 wt%, the loading amount of P is 2 wt%, and the loading amount of Zn is 5 wt%.
2. Mild aromatization test
The C5 cut catalytic cracking gasoline (the family composition is shown in table 1) produced by the North China petrochemical through catalytic cracking is used as a raw material, and the aromatization catalyst is adopted to carry out a mild aromatization test under the non-hydrogenation condition.
In the presence of aromatization catalyst, the above-mentioned C5 cut stage catalytic cracking gasoline is undergone the process of mild aromatization in fixed bed, in which the temperature for controlling mild aromatization is 290 deg.C, pressure is normal pressure and volume space velocity is 1.5h-1(ii) a The mild aromatization results are shown in table 2.
TABLE 1C 5 cut fraction catalytically cracked gasoline family composition
TABLE 2C 5 cut segment moderated aromatization product family composition
The results show that:
the mild aromatization of the C5 cut catalytic cracking gasoline was performed in the manner of this example, with a liquid yield of 99.5%, an olefin conversion of 58%, and an aromatics content increase of 12%.
In addition, the aromatization catalyst has longer service life and stability, the single-pass activity is 8-10 days (the conversion rate of olefin is maintained at the activity of more than 50 percent), the liquid yield is maintained at more than 98.5 percent, the oil loss can be effectively avoided, and the long-period stable operation can be kept.
Example 2
1. Preparation of aromatization catalyst
1) Preparation of catalyst precursor
Under the room temperature environment, the HZSM-5 molecular sieve with the silica-alumina ratio of 25 and the nano-scale and the pseudo-boehmite are mixed according to the weight ratio of 9: 1 to obtain the catalyst precursor.
2) Modification by ion exchange
The catalyst precursor is subjected to ion exchange treatment by adopting a constant-temperature water bath method, specifically, sodium hydroxide is dissolved in deionized water, is mixed with the catalyst precursor, is placed in a 90 ℃ water bath environment and is stirred for 2 hours, so that the loading capacity of sodium is about 0.5 wt%, and then is dried for about 8 hours at about 120 ℃ and is roasted for about 4 hours at about 540 ℃.
3) First modification treatment
Adopting an isometric impregnation method to carry out first modification treatment on the catalyst precursor subjected to ion exchange treatment, specifically dissolving lanthanum nitrate in deionized water, and then impregnating the catalyst precursor to ensure that the loading capacity of lanthanum is 2 wt%; aging at about 23 deg.C for about 6 hr; then dried at about 120 ℃ for about 8 hours and finally calcined at about 540 ℃ for about 8 hours.
4) Reactive metal modification and second modification treatment
The catalyst precursor subjected to the first modification was subjected to hydrothermal treatment at a temperature of about 300 ℃ for about 6 hours in a 100% steam atmosphere.
Carrying out active metal loading and second modification treatment on the catalyst precursor after the hydrothermal treatment by adopting an isometric impregnation method: dissolving ammonium dihydrogen phosphate and zinc nitrate in a citric acid solution with the concentration of about 0.1mol/L to obtain a steeping fluid; controlling the mass ratio of the impregnation liquid to the catalyst precursor to be (1.0 +/-0.2): 1. the impregnation temperature is about 30 ℃, the impregnation time is about 15 hours, and the loading amount of phosphorus is about 1 wt%, and the loading amount of zinc is about 8 wt%; after the impregnation is finished, the aromatization catalyst is obtained by aging for 6 hours at about 28 ℃, drying for about 8 hours at about 120 ℃ in the air atmosphere and roasting for about 4 hours at 540 ℃.
The aromatization catalyst comprises a zeolite molecular sieve (HZSM-5 molecular sieve) and pseudo-boehmite, wherein the mass ratio of the zeolite molecular sieve to the pseudo-boehmite is 9: 1; the active ingredients comprise Na, La, Zn and P, wherein the loading amount of Na is 0.5 wt%, the loading amount of La is 2 wt%, the loading amount of Zn is 8 wt%, and the loading amount of P is 1 wt%.
2. Mild aromatization test
The C6/7 cut segment catalytic cracking gasoline (the family composition is shown in table 3) produced by the North China petrochemical through catalytic cracking is used as a raw material, and the aromatization catalyst is adopted to carry out a mild aromatization test under the non-hydrogenation condition.
In the presence of aromatization catalyst, the above-mentioned C5 cut stage catalytic cracking gasoline is undergone the process of mild aromatization in fixed bed, in which the temperature for controlling mild aromatization is 310 deg.C, pressure is normal pressure and volume space velocity is 1.0h-1(ii) a The mild aromatization results are shown in table 4.
TABLE 3C 6/7 cut fraction catalytically cracked gasoline family composition
TABLE 4C 6/7 cut segment moderated aromatization product family composition
The results show that:
the mild aromatization of the C6/7 cut segment catalytically cracked gasoline was carried out in the manner of this example with a liquid yield of 99.5%.
In addition, the aromatization catalyst has longer service life and stability, the single-pass activity is 8-10 days (the conversion rate of olefin is maintained at the activity of more than 50 percent), the liquid yield is maintained at more than 98.5 percent, the oil loss can be effectively avoided, and the long-period stable operation can be kept.
Example 3
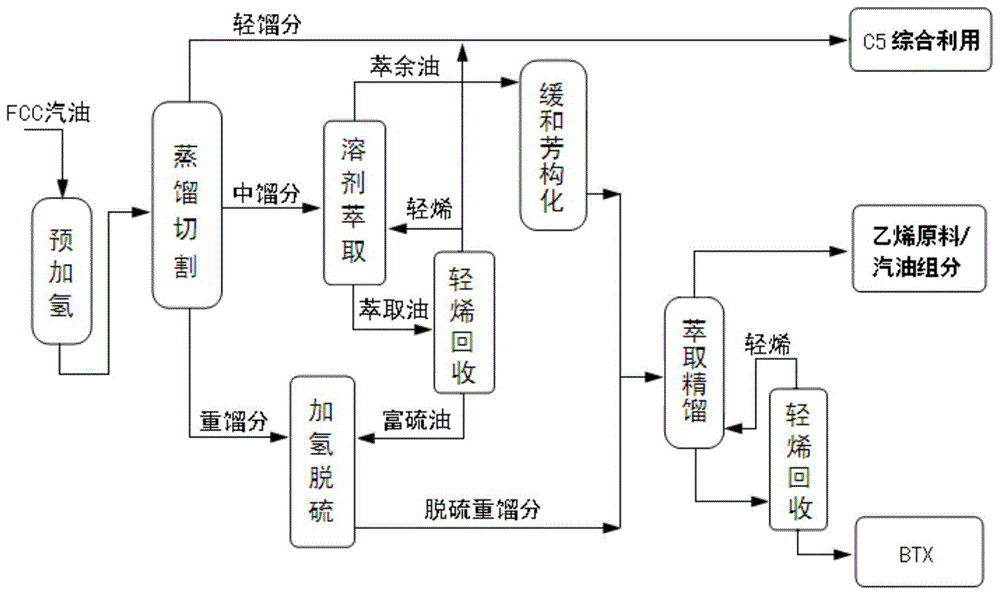
As shown in fig. 1, the method for producing chemical products by using catalytically cracked gasoline of the present embodiment includes the following steps:
1. prehydrogenation
The composition of the catalytically cracked gasoline feedstock of this example is shown in table 5.
TABLE 5 composition of catalytically cracked gasoline feedstock
Pre-hydrogenating the catalytic cracking gasoline in the presence of a pre-hydrogenation catalyst to obtain pre-hydrogenated catalytic cracking gasoline; wherein, the pre-hydrogenation catalyst is a nickel-molybdenum bimetallic catalyst, and the composition (mass content percent) thereof is as follows: al (Al)2O390.5 percent, Ni6 percent and Mo 3.5 percent; the pre-hydrogenation process conditions are as follows: the reaction temperature is controlled to be 130 ℃, the hydrogen-oil ratio is 5, and the volume space velocity is 3h-1。
After the pre-hydrogenation, light sulfides in the catalytically cracked gasoline react with diolefin to form sulfides with high boiling point, and the olefin is not saturated.
2. Cutting of
Cutting the pre-hydrocatalytic cracked gasoline into light fraction, medium fraction and heavy fraction, wherein the cutting temperature of the light fraction and the medium fraction is 40 ℃, and the cutting temperature of the medium fraction and the heavy fraction is 100 ℃, namely: the distillation range of the middle distillate is 40 ℃ to 100 ℃.
The light fraction mainly contains low-carbon (C5) olefin and alkane, and can be directly used as chemical raw materials for comprehensive utilization.
3. Solvent extraction and light olefin recovery
The solvent used for solvent extraction is a compound solvent of sulfolane and N-methyl pyrrolidone, wherein the volume content of the N-methyl pyrrolidone in the compound solvent is 10%.
And (3) allowing the middle distillate to enter from the middle-lower part of the extraction tower, allowing the composite solvent to enter from the upper part of the extraction tower, controlling the top temperature of the extraction tower to be 80 ℃, the bottom temperature of the extraction tower to be 60 ℃, the top pressure (gauge pressure) to be 0.3MPa, and controlling the feeding ratio of the composite solvent to the middle distillate to be 2.0, so as to obtain raffinate oil rich in olefin and extract oil rich in aromatic hydrocarbon.
And (3) delivering the extract oil into a recovery tower to recover light olefins (mainly comprising C5 olefins), wherein the temperature of the top of the recovery tower is controlled to be 88 ℃, the pressure of the top of the recovery tower is controlled to be 0.07MPa, the temperature of the bottom of the recovery tower is controlled to be 168 ℃, and the pressure of the bottom of the recovery tower is controlled to be 0.095MPa, so that the light olefins and sulfur-rich oil are obtained.
Of these, 40% of the light olefins are then returned to the extraction column for solvent extraction, and the remainder of the light olefins are combined into the above light fraction to form C5 components.
4. Mild aromatization
Mild aromatization was carried out without hydrogen using the aromatization catalyst of example 1.
Specifically, the raffinate oil was subjected to mild aromatization in the presence of the aromatization catalyst of example 1, wherein the temperature of the mild aromatization was controlled to 290 ℃, the pressure was normal pressure, and the volume space velocity was 1.5h-1The reaction time is about 200 hours, and an aromatization product is obtained.
The yield of the above-mentioned mild aromatization liquid was calculated to be 99.5%, and the selectivity (i.e., the ratio of selective conversion of olefins to aromatics) was 66.02%.
5. Selective hydrodesulfurization
Firstly adopting CoSO4Soaking ZSM-5 type molecular sieve (carrier) in the solution in the same volume, washing, drying, roasting, and then adopting (NH)4)6Mo7O24·4H2Aqueous solution of O to impregnated CoSO4Soaking the ZSM-5 type molecular sieve in the solution in the same volume, and washing, drying and roasting to prepare the selective hydrodesulfurization catalyst; the total specific surface area of the prepared selective hydrodesulfurization catalyst is 168m through detection2About/g, the total pore volume is about 0.378mL/g, the loading of Co on the carrier is about 7%, the loading of Mo on the carrier is about 10%, and the mass ratio of Co to Mo loaded on the carrier is 0.7: 1.
and (3) carrying out selective hydrodesulfurization on the heavy fraction and the sulfur-rich oil in the presence of the selective hydrodesulfurization catalyst, wherein the temperature of the selective hydrodesulfurization is controlled to be 260 ℃ and the pressure is 1.8MPa, and the volume space velocity of 3.0h-1And the volume ratio of the hydrogen to the oil (namely the volume ratio of the hydrogen to the total volume of the heavy fraction and the sulfur-rich oil) is 500, so as to obtain the desulfurized heavy fraction.
6. Aromatic extraction
The solvent adopted for aromatic extraction is a compound solvent of sulfolane and N-methyl pyrrolidone, wherein the volume content of the N-methyl pyrrolidone in the compound solvent is 10%.
The composite solvent is used as a solvent for aromatic extraction, and aromatic extraction is carried out on the aromatization product and the desulfurized heavy fraction by a method disclosed in Chinese patent with the publication number of CN104694158A to obtain an ethylene raw material/gasoline component and a BTX product.
The composition of the entire product system after the above treatment is shown in Table 6.
Example 4
As shown in fig. 2, the method for producing chemical products by using catalytically cracked gasoline of the present embodiment includes the following steps:
1. prehydrogenation
The composition of the catalytically cracked gasoline feedstock of this example was the same as in example 3.
Pre-hydrogenating the catalytic cracking gasoline in the presence of a pre-hydrogenation catalyst to obtain pre-hydrogenated catalytic cracking gasoline; wherein, the pre-hydrogenation catalyst is a nickel-molybdenum bimetallic catalyst, and the composition (mass content percent) thereof is as follows: al (Al)2O390.5 percent, Ni6 percent and Mo 3.5 percent; the pre-hydrogenation process conditions are as follows: the reaction temperature is controlled to be 130 ℃, the hydrogen-oil ratio is 5, and the volume space velocity is 3h-1。
After the pre-hydrogenation, light sulfides in the catalytically cracked gasoline react with diolefin to form sulfides with high boiling point, and the olefin is not saturated.
2. Cutting of
Cutting the pre-hydrocatalytic cracked gasoline into light fraction, medium fraction and heavy fraction, wherein the cutting temperature of the light fraction and the medium fraction is 40 ℃, and the cutting temperature of the medium fraction and the heavy fraction is 130 ℃, namely: the distillation range of the middle distillate is 40 ℃ to 130 ℃.
The light fraction mainly contains low-carbon (C5) olefin and alkane, and can be directly used as chemical raw materials for comprehensive utilization.
3. Solvent extraction and light olefin recovery
The solvent used for solvent extraction is a compound solvent of sulfolane and N-methyl pyrrolidone, wherein the volume content of the N-methyl pyrrolidone in the compound solvent is 20%.
And (3) allowing the middle distillate to enter from the middle-lower part of the extraction tower, allowing the composite solvent to enter from the upper part of the extraction tower, controlling the top temperature of the extraction tower to be 55 ℃, the bottom temperature of the extraction tower to be 40 ℃, the top pressure (gauge pressure) to be 0.7MPa, and controlling the feeding ratio of the composite solvent to the middle distillate to be 1.0, so as to obtain raffinate oil rich in olefin and extract oil rich in aromatic hydrocarbon.
And (3) delivering the extract oil into a recovery tower to recover light olefins (mainly C5 olefins), wherein the temperature of the top of the recovery tower is 88 ℃, the pressure of the top of the recovery tower is 0.07MPa, the temperature of the bottom of the recovery tower is 168 ℃, and the pressure of the bottom of the recovery tower is 0.095MPa, so as to obtain the light olefins and sulfur-rich oil.
Of these, 50% of the light olefins are then returned to the extraction column for solvent extraction, and the remainder of the light olefins are combined into the above light fraction to form C5 components.
4. Mild aromatization
Mild aromatization was carried out using the aromatization catalyst of example 2 in the absence of hydrogen.
Specifically, the raffinate oil is subjected to mild aromatization in the presence of an aromatization catalyst, wherein the temperature of the mild aromatization is controlled to be 330 ℃, the pressure is normal pressure, and the volume space velocity is 2.0h-1The reaction time is about 200 hours, and an aromatization product is obtained.
The yield of the solution for mild aromatization was calculated to be 99.3% and the selectivity was calculated to be 64.20%.
5. Selective hydrodesulfurization
A selective hydrodesulfurization catalyst was prepared according to the method of example 3, except that the loading of Co on the carrier was controlled to be about 4%, the loading of Mo on the carrier was controlled to be about 10%, and the mass ratio of Co to Mo loaded on the carrier was controlled to be 0.4: 1.
in the presence of a selective hydrodesulfurization catalystSelectively hydrodesulfurizing the heavy fraction and the sulfur-rich oil, wherein the temperature of the selective hydrodesulfurization is controlled to be 250 ℃, the pressure is 2.0MPa, and the volume space velocity is 3.0h-1And the volume ratio of the hydrogen to the oil (namely the volume ratio of the hydrogen to the total volume of the heavy fraction and the sulfur-rich oil) is 300, so as to obtain the desulfurized heavy fraction.
6. Extractive distillation and light olefin recovery
The solvent adopted by the extractive distillation is a compound solvent of sulfolane and N-methyl pyrrolidone, wherein the volume content of the N-methyl pyrrolidone in the compound solvent is 20%.
The number of theoretical plates of the extractive distillation tower is 21, so that the aromatization product and the desulfurized heavy fraction enter from the 18 th plate of the extractive distillation tower, the composite solvent enters from the 3 rd plate of the extractive distillation tower, the temperature of the tower bottom is controlled to be 150 ℃, the feeding ratio of the solvent to the aromatization product and the desulfurized heavy fraction (solvent-oil ratio, namely the ratio of the volume of the solvent to the total volume of the aromatization product and the desulfurized heavy fraction) is 1.0, and the reflux ratio is 1.0.
And (2) sending the extract obtained by the extractive distillation into a recovery tower for recovering light olefin (mainly C5 olefin), wherein the tower top temperature of the recovery tower is 88 ℃, the tower top pressure is 0.07MPa, the tower bottom temperature is 168 ℃, and the tower bottom pressure is 0.095MPa, and returning the obtained light olefin to the above extractive distillation to obtain the ethylene raw material/gasoline component and the BTX product.
The composition of the entire product system after the above treatment is shown in Table 6.
Example 5
As shown in fig. 3, the method for producing chemical products by using catalytically cracked gasoline of the present embodiment includes the following steps:
1. prehydrogenation
The composition of the catalytically cracked gasoline feedstock of this example was the same as in example 3.
Pre-hydrogenating the catalytic cracking gasoline in the presence of a pre-hydrogenation catalyst to obtain pre-hydrogenated catalytic cracking gasoline; wherein, the pre-hydrogenation catalyst is a nickel-molybdenum bimetallic catalyst, and the composition (mass content percent) thereof is as follows: al (Al)2O390.5 percent, Ni6 percent and Mo 3.5 percent; the pre-hydrogenation process conditions are as follows: the reaction temperature is controlled to be 130 ℃, the hydrogen-oil ratio is 5, and the volume space velocity is 3h-1。
After the pre-hydrogenation, light sulfides in the catalytically cracked gasoline react with diolefin to form sulfides with high boiling point, and the olefin is not saturated.
2. Cutting of
Cutting the pre-hydrocatalytic cracked gasoline into light fraction, medium fraction and heavy fraction, wherein the cutting temperature of the light fraction and the medium fraction is 40 ℃, and the cutting temperature of the medium fraction and the heavy fraction is 140 ℃, namely: the distillation range of the middle distillate is 40 ℃ to 140 ℃.
The light fraction mainly contains low-carbon (C5) olefin and alkane, and can be directly used as chemical raw materials for comprehensive utilization.
3. Solvent extraction and light olefin recovery
The solvent used for solvent extraction is a compound solvent of sulfolane and N-methyl pyrrolidone, wherein the volume content of the N-methyl pyrrolidone in the compound solvent is 5%.
And (3) allowing the middle distillate to enter from the middle-lower part of the extraction tower, allowing the composite solvent to enter from the upper part of the extraction tower, controlling the top temperature of the extraction tower to be 100 ℃, the bottom temperature of the extraction tower to be 70 ℃, the top pressure (gauge pressure) to be 0.2MPa, and controlling the feeding ratio of the composite solvent to the middle distillate to be 5.0, so as to obtain raffinate oil rich in olefin and extract oil rich in aromatic hydrocarbon.
And (3) delivering the extract oil into a recovery tower to recover light olefins (mainly C5 olefins), wherein the temperature of the top of the recovery tower is controlled to be 95 ℃, the pressure of the top of the recovery tower is controlled to be 0.05MPa, the temperature of the bottom of the recovery tower is controlled to be 80 ℃, and the pressure of the bottom of the recovery tower is controlled to be 0.07MPa, so that the light olefins and sulfur-rich oil are obtained.
Of these, 60% of the light olefins are then returned to the extraction column for solvent extraction, and the remainder of the light olefins are combined into the above light fraction to form C5 components.
4. Mild aromatization
Mild aromatization was carried out without hydrogen using the aromatization catalyst of example 1.
Specifically, the raffinate oil is subjected to mild aromatization in the presence of an aromatization catalyst, wherein the temperature of mild aromatization is controlledThe temperature is 280 ℃, the pressure is normal pressure, and the volume space velocity is 1.5h-1The reaction time is about 200 hours, and an aromatization product is obtained.
The yield of the solution for mild aromatization was calculated to be 99.4% and the selectivity was calculated to be 63.86%.
5. Selective hydrodesulfurization
Selective hydrodesulfurization was carried out using the selective hydrodesulfurization catalyst of example 3.
In the presence of a selective hydrodesulfurization catalyst, selectively hydrodesulfurization is carried out on the heavy fraction and the sulfur-rich oil, wherein the temperature of the selective hydrodesulfurization is controlled to be 300 ℃, the pressure is controlled to be 2.5MPa, and the volume space velocity is controlled to be 2.0h-1And the volume ratio of the hydrogen to the oil is 400, so that the desulfurized heavy fraction is obtained.
6. Extractive distillation and light olefin recovery
The solvent adopted by the extractive distillation is a compound solvent of sulfolane and N-methyl pyrrolidone, wherein the volume content of the N-methyl pyrrolidone in the compound solvent is 5%.
The number of theoretical plates of the extractive distillation tower is 21, so that the aromatization product and the desulfurized heavy fraction enter from the 18 th plate of the extractive distillation tower, the composite solvent enters from the 3 rd plate of the extractive distillation tower, the temperature of the tower bottom is controlled to be 160 ℃, the feeding ratio of the solvent to the aromatization product and the desulfurized heavy fraction (solvent-oil ratio, namely the ratio of the volume of the solvent to the total volume of the aromatization product and the desulfurized heavy fraction) is 2.0, and the reflux ratio is 2.0.
And (2) sending the extract obtained by the extractive distillation into a recovery tower for recovering light olefin (mainly C5 olefin), wherein the top temperature of the recovery tower is controlled to be 88 ℃, the top pressure is controlled to be 0.07MPa, the bottom temperature is controlled to be 168 ℃, and the bottom pressure is controlled to be 0.095MPa, and the obtained light olefin is returned for the mild aromatization to obtain the ethylene raw material/gasoline component and a BTX product.
The composition of the entire product system after the above treatment is shown in Table 6.
Comparative example 1
This control is the same as example 3 except that the mild aromatization of example 3 is replaced with conventional aromatization. Specifically, the preparation method of the aromatization catalyst used in this comparative example was substantially the same as that of example 1 except that: the catalyst precursor is not subjected to the ion exchange treatment of step 2), and is directly subjected to step 3), step 4), and step 5).
In the presence of the aromatization catalyst, performing conventional aromatization on raffinate oil, wherein the temperature of the conventional aromatization is 450 ℃, the pressure is normal pressure, and the volume space velocity is 1.0h-1The reaction time is about 200 hours, and an aromatization product is obtained.
Through calculation, the yield of the conventional aromatization solution is about 70 percent, and the selectivity is about 20 percent; the once-through activity is only 3-4 days (once-through activity is an activity in which the olefin conversion remains above 50%).
TABLE 6 composition of the product systems of the examples
Finally, it should be noted that: the above embodiments are only used to illustrate the technical solution of the present invention, and not to limit the same; while the invention has been described in detail and with reference to the foregoing embodiments, it will be understood by those skilled in the art that: the technical solutions described in the foregoing embodiments may still be modified, or some or all of the technical features may be equivalently replaced; and the modifications or the substitutions do not make the essence of the corresponding technical solutions depart from the scope of the technical solutions of the embodiments of the present invention.