Summary of the invention
The object of the invention is to by optimum combination and optimum ratio to various liquid-crystal compositions, provide a kind of and have high clearing point, high index, good low temperature storage stability and be fit to high optically anisotropic liquid-crystal composition, described liquid-crystal composition can not show the shortcoming of prior art material or at least only show above-mentioned shortcoming on significantly less degree.
In order to complete the foregoing invention purpose, the invention provides a kind of liquid-crystal composition, it comprises four kinds of compounds that meet following general structure I, II, III, IV, wherein:
(1) account for the compound of the logical formula I of described liquid-crystal composition gross weight 5%-35%
(2) account for the compound of the logical formula II of described liquid-crystal composition gross weight 1%-35%
(3) account for the compound of the general formula (III) of described liquid-crystal composition gross weight 40%-85%
(4) account for the compound of the logical formula IV of described liquid-crystal composition gross weight 1-35%
Wherein,
R
1, R
2Can be identical or different, be independently selected from respectively by H, halogen, have the halo of 1-7 carbon atom or not by the alkyl of halo or alkoxyl group, the group that has the halo of 2-7 carbon atom or do not formed by the alkane thiazolinyl of halo or alcoxyl thiazolinyl, wherein, at described R
1And R
2In one or more-CH
2-group can be independently of one another by-CH=CH-,-O-,-CH=CF-,-CF=CH-,-CF=CF-,-CO-O-or-O-CO-substitutes, its prerequisite is that Sauerstoffatom directly is not connected to each other;
R
3Select free H, halogen, have the halo of 1-10 carbon atom or not by the alkyl of halo or alkoxyl group and the group that has the halo of 2-10 carbon atom or do not formed by the alkane thiazolinyl of halo or alcoxyl thiazolinyl, wherein, at described R
3In one or more-CH
2-group can be independently of one another by-CH=CH-,-O-,-CH=CF-,-CF=CH-,-CF=CF-,-CO-O-or-O-CO-substitutes, its prerequisite is that Sauerstoffatom directly is not connected to each other;
R
4Select the halo of a free 2-10 carbon atom or do not formed by the alkane thiazolinyl of halo or alcoxyl thiazolinyl;
R
5, R
6Can be identical or different, be independently selected from respectively by H, halogen, have the halo of 1-10 carbon atom or not by the alkyl of halo or alkoxyl group and the group that has the halo of 2-10 carbon atom or do not formed by the alkane thiazolinyl of halo, wherein, at described R
5And R
6In one or more-CH
2-group can be independently of one another by-CH=CH-,-O-,-CH=CF-,-CF=CH-,-CF=CF-,-CO-O-or-O-CO-substitutes, its prerequisite is that Sauerstoffatom directly is not connected to each other;
R
7, R
8Can be identical or different, be independently selected from respectively by H, halogen, have the halo of 1-10 carbon atom or not by the alkyl of halo or alkoxyl group and have the halo of 2-10 carbon atom or not by halo alkane thiazolinyl or the group that forms of alcoxyl thiazolinyl, wherein, at described R
7And R
8In one or more-CH
2-group can be independently of one another by-CH=CH-,-O-,-CH=CF-,-CF=CH-,-CF=CF-,-CO-O-or-O-CO-substitutes, its prerequisite is that Sauerstoffatom directly is not connected to each other;
Z
1, Z
2, Z
3, Z
4, Z
5, Z
6Can be identical or different, be independently selected from respectively by singly-bound ,-O-,-COO-,-OCO-,-C
2H
4-,-CH
2O-,-CH
2CH
2-,-CH=CH-,-CF=CF-,-CF
2O-and-group that C ≡ C-forms;
Y
1Be H, CH independently
3Or F;
L
1, L
2, L
3, L
4, L
5, L
6Can be identical or different, be H or F independently respectively;
Can be identical or different, be independently selected from respectively by
With
The group that forms;
Can be identical or different, be independently respectively
Or
Wherein said
In one or two non-conterminous-CH
2-can be replaced by O, described
Upper one or more H can be replaced by F independently of one another;
M, n are identical or different, are 0,1 or 2 independently of one another, and 0≤m+n≤3;
A, b, c, d are identical or different, are 0,1 or 2 independently of one another, and 1≤a+b+c+d≤7;
Q is 0 or 1.
In embodiments of the invention, one or more compounds in the group of the freely following compound composition of the compound of logical formula I choosing:
Wherein,
R
1Be independently selected from by the fluoro with 1-5 carbon atom or not by the alkyl of fluoro or alkoxyl group and the group that has the fluoro of 2-5 carbon atom or do not formed by the alkane thiazolinyl of fluoro or alcoxyl thiazolinyl;
R
2Be independently selected from by H, F, have the fluoro of 1-8 carbon atom or not by the alkyl of fluoro or alkoxyl group and the group that has the fluoro of 2-8 carbon atom or do not formed by the alkane thiazolinyl of fluoro or alcoxyl thiazolinyl, wherein, at described R1, R
2In one or more-CH
2-group can be independently of one another by-CH=CH-,-O-,-CH=CF-,-CF=CH-,-CF=CF-,-CO-O-or-O-CO-substitutes, its prerequisite is that Sauerstoffatom directly is not connected to each other.
In embodiments of the invention, one or more compounds in the group of the freely following compound composition of the compound of logical formula II choosing:
Wherein,
R
3Choosing freely has the fluoro of 1-8 carbon atom or not by the alkyl of fluoro or alkoxyl group with have the fluorine replacement of 2-8 carbon atom or the alkane thiazolinyl that do not replaced by fluorine or the group of alcoxyl thiazolinyl composition.
In embodiments of the invention, one or more compounds in the group of the freely following compound composition of the compound of logical formula III choosing:
Wherein,
R
5Be independently selected from by the fluoro with 2-8 carbon atom or not by the alkyl of fluoro or alkoxyl group and the group that has the fluoro of 2-5 carbon atom or do not formed by the alkane thiazolinyl of fluoro or alcoxyl thiazolinyl, wherein, at described R
5In one or more-CH
2-group can be independently of one another by-CH=CH-,-O-,-CH=CF-,-CF=CH-,-CF=CF-,-CO-O-or-O-CO-substitutes, its prerequisite is that Sauerstoffatom directly is not connected to each other;
R
6Be independently selected from by H, F and have the fluoro of 1-8 carbon atom or not by the alkyl of fluoro or alkoxyl group and the group that has the fluoro of 2-5 carbon atom or do not formed by the alkane thiazolinyl of fluoro or alcoxyl thiazolinyl, wherein, at described R
6In one or more-CH
2-group can be independently of one another by-CH=CH-,-O-,-CH=CF-,-CF=CH-,-CF=CF-,-CO-O-or-O-CO-substitutes, its prerequisite is that Sauerstoffatom directly is not connected to each other.
In embodiments of the invention, one or more compounds in the group of the freely following compound composition of the compound of logical formula IV choosing:
And
Wherein,
R
7Be independently selected from by the fluoro with 1-8 carbon atom or not by the alkyl of fluoro or alkoxyl group and the group that has the fluoro of 2-8 carbon atom or do not formed by the alkane thiazolinyl of fluoro or alcoxyl thiazolinyl;
R
8Be independently selected from by H ,-CH
2F ,-CH
2CH
2F ,-CH
2CH
2CH
2F ,-CH
2CH
2CH
2CH
2F ,-OCH
2F ,-CH
2OCH
2F ,-CH
2CH
2OCH
2F and-CH=CF
2The group that forms.
In embodiments of the invention, the compound of preferred formula (I) accounts for the 5%-30% of described composition total weight; The compound of logical formula II accounts for the 5%-30% of described composition total weight; The compound of logical formula III accounts for the 45%-80% of described composition total weight; The compound of logical formula IV accounts for the 10%-25% of described composition total weight.
Another aspect of the present invention provides the application of liquid-crystal composition in making the electrooptics device.
Another aspect of the present invention provides a kind of electrooptics liquid-crystal display, and described liquid-crystal display comprises liquid-crystal composition of the present invention.
The present invention is by carrying out combination experiment to above-claimed cpd, by with the contrast comparison, determined to comprise the liquid crystal media of above-mentioned liquid-crystal composition, had high clearing point, higher specific refractory power, good low temperature storage stability and be fit to high optically anisotropic performance.
As mentioned above, liquid-crystal composition of the present invention is used for liquid crystal cell, and its image display effect is good, without motion blur phenomenon.
In the present invention if no special instructions, described ratio is weight ratio, and all temperature are degree celsius temperature, and the box that the test of described response time data is selected is thick is 7 μ m.
Embodiment
Below with reference to specific embodiments, the present invention is described.Need to prove, the following examples are example of the present invention, only are used for illustrating the present invention, and are not used for limiting the present invention.In the situation that do not depart from purport of the present invention or scope, can carry out the present invention and conceive interior other combination and various improvement.
Below the liquid-crystal display that adopts of each embodiment be the TN-TFT liquid crystal display, the thick d=7 μ of box m partly is made of polarizer (polaroid), electrode base board etc.This display equipment is normal white mode, and when namely not having voltage difference to put between the row and column electrode, the viewer observes the pixel color of white.Polarizer axes up and down on substrate is 90 degrees to each other the angle.Space between two substrates is full of optical liquid crystal material.
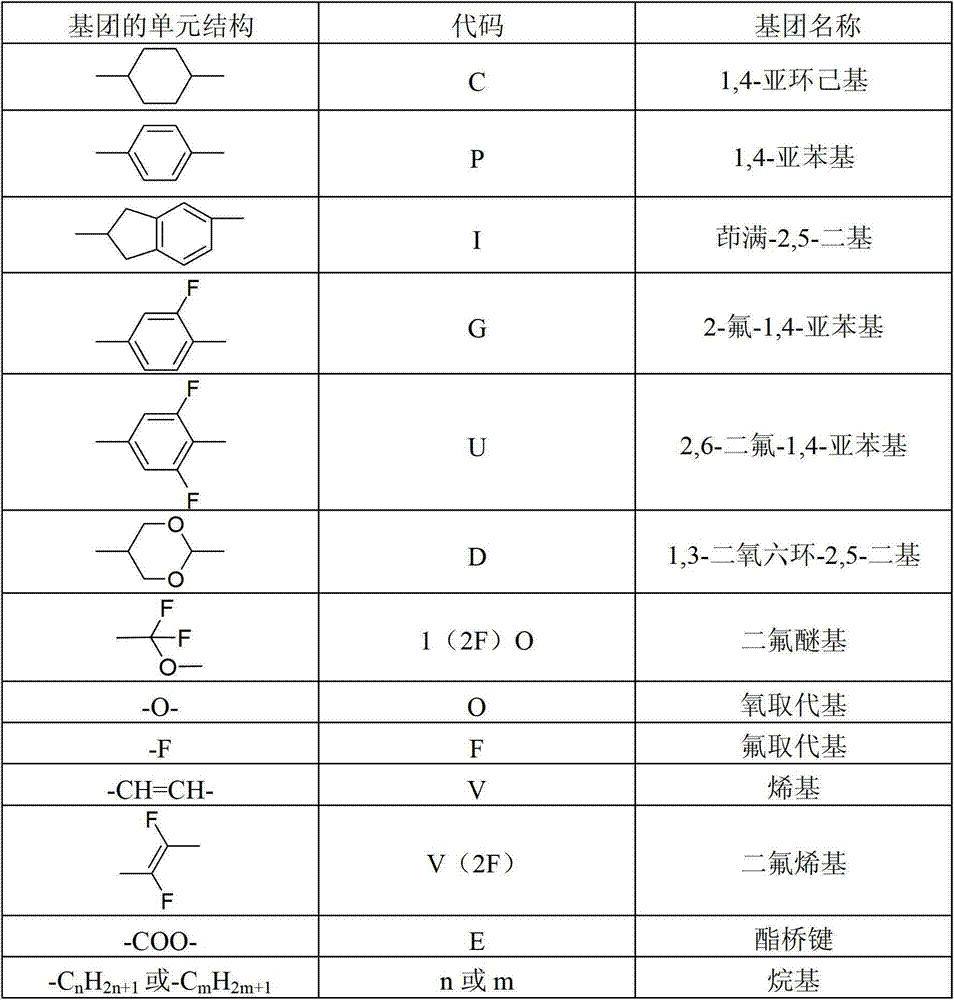
For ease of expressing, below in each embodiment, the unit structure of liquid crystalline cpd is with the listed coded representation of table 1:
The unit structure code of table 1 liquid crystalline cpd
Take following structure as example:
This structure is with the coded representation in table 1: can be expressed as 3PTG1(2F) OP3, and for example:
Can be expressed as nCPTPOm, the n in code represents the C atomicity of left end alkyl, and for example n is " 3 ", represents that namely this alkyl is-C
3H
7C in code represents cyclohexyl; O represention oxygen atom in code; P in code represents phenylene; M in code represents the C atomicity of right-hand member alkyl, and for example m is " 1 ", represents that namely the alkyl of right-hand member is-CH
3
In embodiment, the code name of writing a Chinese character in simplified form of each test event is expressed as respectively:
Each composition that adopts in following embodiment, except the compound of logical formula I, all the other components all can be synthesized by known method, perhaps obtain by commercial sources.These synthetic technologys are conventional, and resulting each liquid crystalline cpd meets electrical type compound standard after tested.
The preparation of the compound of general formula (I)
1) in solvents tetrahydrofurane, under room temperature, with the compound of general formula 1
Compound with formula 2
In nitrogen protection, there are lower reaction 16 hours in the catalyzer tetrakis triphenylphosphine palladium, obtains the compound of general formula 3
(general formula 3);
2) in solvents tetrahydrofurane, under nitrogen protection, under-100 ℃ ~-50 ℃, with the compound of general formula 4
React with nBuLi the lithium reagent that obtains general formula 5,
With the compound reaction of described lithium reagent and described general formula 3, obtain compound shown in general formula (I) again.
R in above-mentioned general formula (general formula 1, general formula 3, general formula 4 and general formula 5)
1, R
2With the R in general formula (I)
1, R
2Scope consistent, R
1, R
2Select independently of one another free H, halogen, have halo or unsubstituted alkyl or the alkoxyl group of 1-7 carbon atom and have the group of halo or unsubstituted alkane thiazolinyl or the alcoxyl thiazolinyl composition of 2-7 carbon atom, wherein, at described R
1And R
2In one or more-CH
2-group can be independently of one another by-CH=CH-,-O-,-CH=CF-,-CF=CH-,-CF=CF-,-CO-O-or-O-CO-substitutes, its prerequisite is that Sauerstoffatom directly is not connected to each other;
With
Identical or different, choosing independently of one another freely
With
The group that forms; Z
1, Z
2, Z
3And Z
4Identical or different, be independently from each other separately by-CO-O-,-O-CO-,-CF
2O-,-OCF
2-,-CH
2O-,-OCH
2-,-CH
2CH
2-,-(CH
2)
4-,-C
2F
4-,-CH
2CF
2-,-CF=CF-,-group that CH=CH-and singly-bound form; M, n, p and t are identical or different, are 0,1 or 2 separately independently of one another, and m+n+p+t≤3.
Preparation example
The synthetic concrete technology step of the Compound I-8-2 of preparation is as follows:
1) synthetic diisopropylamine lithium (LDA)
Add the 115mL Diisopropylamine in the 1000mL there-necked flask, 100mL tetrahydrofuran (THF) (THF), nitrogen protection, 0 ℃ ~-20 ℃ n-Butyl Lithiums (n-BuLi, 2.4 mol/L) that drip 320 mL of temperature control drip and finish, and 0 ~-20 ℃ was stirred 1 hour, and made LDA.
2) synthetic trifluorovinyl zinc chloride
Add the 53g Zinc Chloride Anhydrous in the 1000mL there-necked flask, 100 mL THF, nitrogen protection is cooled to-70 ℃; pass into 54g1,1,1,2-Tetrafluoroethane gas; temperature control-50 ℃~-70 ℃ of needle tubings pass into and slowly inject LDA under liquid level, add rear stirring 2 hours, make trifluorovinyl zinc chloride.
3) compound of synthesis type 6
Compound (our company's self-control, 45 g) and the tetrakis triphenylphosphine palladium (2 g) of formula 7 are joined in the reaction solution of the trifluorovinyl zinc chloride for preparing above, nitrogen protection, room temperature reaction spends the night.
With dilute hydrochloric acid frozen water solution cancellation reaction, ethyl acetate extraction merges organic layer, the saturated common salt washing, and anhydrous sodium sulfate drying is spin-dried for solvent, and resistates is purified through underpressure distillation, obtains 22.3 g light green transparent liquids, is the compound of formula 6.
4) compound of synthesis type 11
(formula 11)
The 100ml there-necked flask adds 2.9g3.5-difluoro bromobenzene, 50ml tetrahydrofuran (THF), and nitrogen protection is cooled to-78 ℃, slowly drips the 7.5ml2.4mol/L n-Butyl Lithium, reacts 2h below temperature control-70 ℃, makes a fluorobenzene lithium reagent.
Below the compound (14g) of the formula 6 that makes with 20mL tetrahydrofuran (THF) dissolving, temperature control-70 ℃, with its splash into above-mentioned make between in the fluorobenzene lithium reagent, room temperature reaction spends the night.
After reacting completely, rare cryosel sour water cancellation reaction, ethyl acetate extraction, washing, anhydrous sodium sulfate drying is spin-dried for solvent, obtains weak yellow liquid 3.4g take sherwood oil as the eluent column chromatography.Carry out with silica gel the compound that column chromatography gets 1.1g formula 11 take sherwood oil as eluent.MS:m/z:334.13
5) compound of synthesis type 12
The 100ml there-necked flask adds magnetic stir bar, the compound of 1.1g formula 11,20ml THF; nitrogen protection is cooled to-80 ℃, slowly drips n-BuLi1.2ml, and reaction color begins to be become by redness blackish green; become at last blueness, temperature control-80 ℃ stirring reaction 2h drips CF
2Br
2(2g), the reaction solution color becomes yellow, temperature control-80 ℃ stirring reaction 1h.
With the cancellation of 20ml frozen water, the salt acid for adjusting pH value is to 6 left and right, and ethyl acetate extraction merges organic layer, the washing organic layer, the saturated common salt water washing, drying is spin-dried for solvent, after column chromatography, the ethanol making beating obtains the 1g white solid, is the compound of formula 12, and productive rate is 62.5%.MS:m/z:462.04。
6) synthetic compound I-8-2
100ml single port bottle adds compound, the 0.35g3 of 1g formula 12, the salt of wormwood of 4,5-trifluoromethyl phenol, 0.6g, 0.03g potassiumiodide (KI) and 30mlN, and dinethylformamide (DMF) stirs, nitrogen protection, 80 ℃ of reaction 3h.
Reaction solution is poured into water, and ethyl acetate extraction merges organic layer, the washing organic layer, and drying, decompression is spin-dried for solvent.Carry out column chromatography take sherwood oil as eluent with silica gel.The ethanol making beating obtains white solid (I-8-2) 0.5g, and productive rate is 43.5%.MS:m/z:530.2。
The liquid crystal property of Compound I-8-2:
Δn:0.176 Δε:16.2 Cp:73.8℃
According to the proportioning of each liquid-crystal composition of following examples regulations, preparation liquid-crystal composition.The preparation of described liquid-crystal composition is to carry out according to the ordinary method of this area, as take the modes such as heating, ultrasonic wave, suspension according to the rules ratio mix and to make.
Prepare and study the liquid-crystal composition that provides in the following example.The below has shown composition and its performance parameter test result of each liquid-crystal composition.
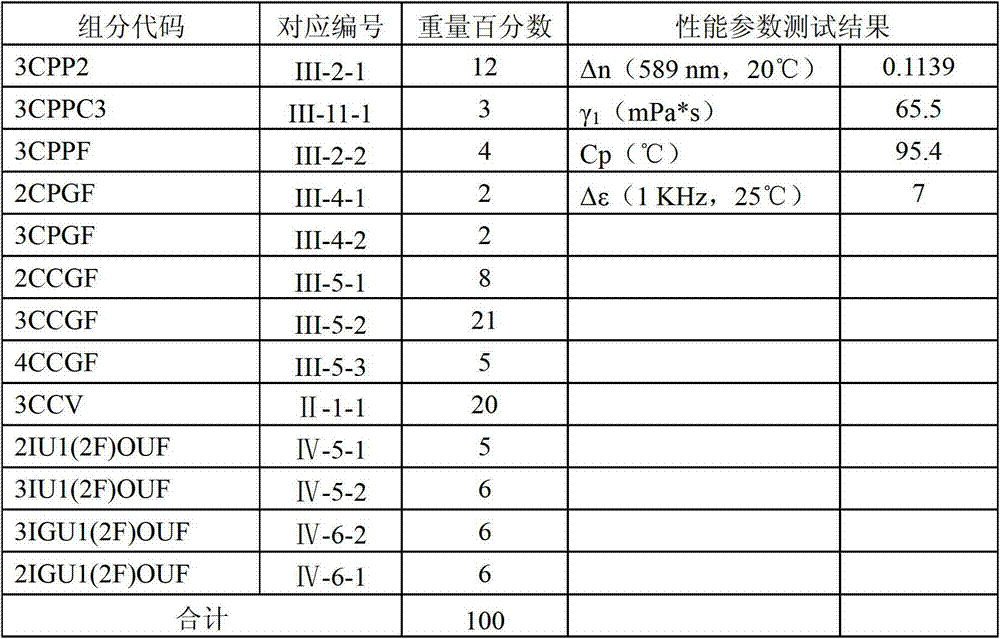
Table 2 and table 4 are listed to be composition, the proportioning of reference examples liquid-crystal composition and to be filled in the test result of carrying out performance test between the liquid-crystal display two substrates, so that carry out performance comparison with explanation liquid-crystal composition of the present invention.
Reference examples 1
By the liquid-crystal composition that each compound listed in table 2 and weight percentage are mixed with reference examples, it is filled in carries out performance test between the liquid-crystal display two substrates, test data is as shown in the table:
Table 2 liquid-crystal composition formula and test performance thereof
Embodiment 1
Be mixed with liquid-crystal composition of the present invention by each compound listed in table 3 and weight percentage, it is filled in and carries out performance test between the liquid-crystal display two substrates, and test data is as shown in the table:
Table 3 liquid-crystal composition formula and test performance thereof
Can find out by comparing embodiment 1 and Comparative Examples 1, composition provided by the invention has higher clearing point, thereby can be used in high temperature service, has simultaneously high optics anisotropic and suitable high dielectric anisotropy.
Reference examples 2
By the liquid-crystal composition that each compound listed in table 4 and weight percentage are mixed with reference examples, it is filled in carries out performance test between the liquid-crystal display two substrates, test data is as shown in the table:
Table 4 liquid-crystal composition formula and test performance thereof
Embodiment 2
Be mixed with liquid-crystal composition of the present invention by each compound listed in table 5 and weight percentage, it is filled in and carries out performance test between the liquid-crystal display two substrates, and test data is as shown in the table:
Table 5 liquid-crystal composition formula and test performance thereof
Can find out by comparing embodiment 2 and Comparative Examples 2, composition provided by the invention has higher clearing point, thereby can be used in high temperature service, has simultaneously high optics anisotropic and suitable high dielectric anisotropy.
Embodiment 3
Be mixed with liquid-crystal composition of the present invention by each compound listed in table 6 and weight percentage, it is filled in and carries out performance test between the liquid-crystal display two substrates, and test data is as shown in the table:
Table 6 liquid-crystal composition formula and test performance thereof
The characteristics of said composition are high clearing point, favourable optics anisotropic, suitably high dielectric anisotropy and response speed faster.
Embodiment 4
Be mixed with liquid-crystal composition of the present invention by each compound listed in table 7 and weight percentage, it is filled in and carries out performance test between the liquid-crystal display two substrates, and test data is as shown in the table:
Table 7 liquid-crystal composition formula and test performance thereof
The characteristics of said composition are high clearing point, favourable optics anisotropic, suitably high dielectric anisotropy and response speed faster.
Embodiment 5
Be mixed with liquid-crystal composition of the present invention by each compound listed in table 8 and weight percentage, it is filled in and carries out performance test between the liquid-crystal display two substrates, and test data is as shown in the table:
Table 8 liquid-crystal composition formula and test performance thereof
The characteristics of said composition are very high clearing point, favourable optics anisotropic, suitably high dielectric anisotropy and response speed faster.
Embodiment 6
Be mixed with liquid-crystal composition of the present invention by each compound listed in table 9 and weight percentage, it is filled in and carries out performance test between the liquid-crystal display two substrates, and test data is as shown in the table:
Table 9 liquid-crystal composition formula and test performance thereof
The characteristics of said composition are high clearing point, favourable optics anisotropic, higher specific refractory power, suitably high dielectric anisotropy and response speed faster.