CN102688184B - Preparation method of ephedrine hydrochloride injection for improving rat blood rheological obstacle - Google Patents
Preparation method of ephedrine hydrochloride injection for improving rat blood rheological obstacle Download PDFInfo
- Publication number
- CN102688184B CN102688184B CN 201210151403 CN201210151403A CN102688184B CN 102688184 B CN102688184 B CN 102688184B CN 201210151403 CN201210151403 CN 201210151403 CN 201210151403 A CN201210151403 A CN 201210151403A CN 102688184 B CN102688184 B CN 102688184B
- Authority
- CN
- China
- Prior art keywords
- purity
- preparation
- ephedrine
- ephedrine hydrochloride
- tween
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
Abstract
A preparation method of ephedrine hydrochloride injection for improving rat blood rheological obstacle belongs to the field of preparation of drugs for improving biological blood disorders. The preparation method is characterized in that active ingredients of sedanolideat least 98% in purity, gastrodin at least 98% in purity and ephedrine hydrochloride at least 98% in purity are proportionally mixed in 5% tween-80 aqueous solution to make the ephedrine hydrochloride injection, and a combination of the sedanolide, the gastrodin and the ephedrine hydrochloride is 5 mg/l in concentration in the 5% tween-80 aqueous solution. Compared with clinically common drug nimodipine, the ephedrine hydrochloride injection has evidently improved blood viscosity, maximum deformation index of erythrocytes, maximum aggregation index of the erythrocytes and aggregation rate of platelets.
Description
Technical field
Improve the preparation method of the injection that contains ephedrine that the hemorheology obstacle uses, belong to the manufacturing technology field that prevents and treats brain ischemia medicament.
Background technology
Application number is 200880001478.9, the Chinese invention patent of " a kind of pharmaceutical composition of preventing and treating the cerebral ischemia apoplexy and preparation method thereof " by name has been pointed out a kind of pharmaceutical composition for the control cerebral infarction, is together to form by sedanolide, ligustilide and gastrodine.Wherein, gastrodine is that purity is the above commercially available prod of 98 %, and the purity of sedanolide and ligustilide has correspondingly proposed purity and be 99.17% sedanolide and purity and be the preparation method of 98.32% ligustilide all more than 98 %.Its experimental result shows that the compatibility of these three kinds of compositions can reduce the degree of the scope of ischemic brain infarction (representing with cerebral infarction percent) and hemiplegia of limb (representing with the behavior symptom scoring) effectively.During namely as injection treatment administration, the rat cerebral infarction area percentage is between 13.03%-18.58 %; Behavior scoring is between 1.50 minutes-2.30 minutes; During as oral agents prevention administration, the rat cerebral infarction area percentage is between 21.36%-21.67%, and behavior scoring is between 3.36 minutes-4.33 minutes.Its drug effect is better than clinical application MAILUONING (injection) commonly used and ligustrazine (injection and oral agents).Improve the health microcirculation experimental data of (representing with hemorheology index) but lack.
In the document that retrieves at present, do not find to have in the pharmaceutical composition and adopt ephedrine, gastrodine and sedanolide to come compatibility.
Summary of the invention
The object of the invention is to extract and prepare a kind of injection preparation that contains ephedrine of using improving the hemorheology obstacle.
The present invention is a kind of preparation method that blood flow becomes the injection that contains ephedrine that obstacle uses of improving:
One of its feature, taking by weighing purity is at least 98% sedanolide 0.16g, purity and is at least 98% gastrodine 0.024g, purity and is at least 98% ephedrine 0.24g, be 5% Tween-81 85ml in percent by volume, stir, the compositions that makes described sedanolide, gastrodine and ephedrine is that concentration in 5% the Tween-81 aqueous solution is 5mg/ml in percent by volume.
Two of its feature, taking by weighing purity is at least 98% sedanolide 0.16g, purity and is at least 98% gastrodine 0.01g, purity and is at least 98% ephedrine 0.24g, the adding percent by volume is 5% Tween-81 82ml, stir, the compositions that makes described sedanolide, gastrodine and ephedrine is that concentration in 5% the Tween-81 aqueous solution is 5mg/ml in percent by volume.
Experiment showed, that above-mentioned two kinds of tested materials all can improve hemorheology index, thus the performance treating cerebral ischemia.Suitable with the clinical application nimotop.Wherein embodiment 1 is better than embodiment 2.
The specific embodiment
One, tested material preparation:
Embodiment 1: the sedanolide 0.16g that takes by weighing 98% purity, the gastrodine 0.024g of 98% purity, the ephedrine 0.24g of 98% purity, the adding percent by volume is 5% Tween-81 85ml, stir, make the compositions of described sedanolide, gastrodine and ephedrine in the 5%(volume ratio) the Tween-81 aqueous solution in concentration be 5mg/ml.
Embodiment 2: the sedanolide 0.16g that takes by weighing 98% purity, the gastrodine 0.01g of 98% purity, the ephedrine 0.24g of 98% purity, the adding percent by volume is 5% Tween-81 82ml, stir, make the compositions of described sedanolide, gastrodine and ephedrine in the 5%(volume ratio) the Tween-81 aqueous solution in concentration be 5mg/ml.
Described sedanolide is called the described method preparation of application for a patent for invention of a kind of pharmaceutical composition of preventing and treating the cerebral ischemia apoplexy and preparation method thereof by application number 200880001478.9, name, gastrodine and ephedrine are the commercially available prod.
Two, test method:
Experiment is finished by Xuanwu Hospital of Capital University of Medical Science.
Model manufacturing: experiment uses rat with 25% urethane, 1.5 g/kg intraperitoneal injection of anesthesia, at rat parietal bone of head position, the bregma center is 2mm forward, 2.5mm place to the right, make a call to the bone window of a diameter 2mm with dental burr, measurement electrode is inserted 2mm under the pia mater encephali, measure the front cerebral tissue blood flow (rCBF) of ligation, then ligation bilateral common carotid arteries, rCBF when measuring respectively after the ligation 10min, 60min and 120min.Blood flow is in ml/100g/min.And calculating relative blood flow amount.Detect principle according to Hydrogen Gas Clearance.
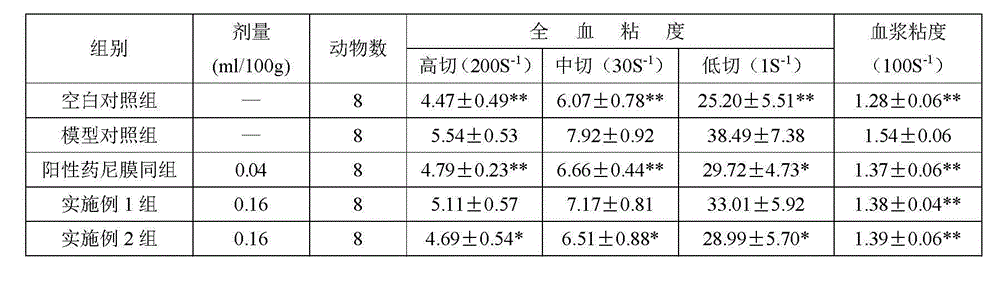
Before laboratory animal was put to death, through abdominal aortic blood 0.8ml, anticoagulant detected with blood viscosity instrument, cuts (200S with height
-1), in cut (30 S
-1) and low (5 S that cut
-1) time blood viscosity represent whole blood viscosity (seeing Table 1); Other gets anticoagulation and leaves heart 8min with 2000, and its supernatant is blood plasma, gets 0.8m1 blood plasma and tests 100 S with blood viscosity instrument
-1The time plasma viscosity (seeing Table 2).With erythrocyte maximum distortion index (MAXDI) expression red cell deformability (seeing Table 3); With the maximum aggregate index of erythrocyte (MAXD) expression erythrocyte aggregation (seeing Table 3).
The preparation of middle cerebral artery thrombus model: the method improvement by Tamura etc. forms.Laboratory animal adopts 350g ± 20g SD rat, with chloral hydrate 0.35g/kg intraperitoneal injection of anesthesia.The Rat Right lateral position is fixed, curved incision at paropia and external auditory meatus line mid point work one long 1.5cm exposes temporal bone, makes the bone window of a diameter 2.5mm to nasal side 1mm place at cheekbone and temporo squamosum joint with dental burr, expose middle cerebral artery, suction is had 10 μ, 1 50% FeCl
3The small pieces filter paper of liquid applies on middle cerebral artery, and operation is carried out under stereomicroscope.Filter paper is taken off in the middle cerebral artery blackening behind 30min, uses the normal saline flushing local organization, layer-by-layer suture.Sham operated rats is not applied FeCl
3, remaining same model group.
The experiment grouping: laboratory animal adopts the SD rat, and body weight is 350 ± 20g.Experiment is divided into 5 groups: blank group and model control group give normal saline, and dosage is the 0.04ml/100g body weight; Positive control drug is 0.02% sterile solution for injection of clinical treatment cerebral ischemia common medicine nimotop, is produced lot number CBWLL11B by Bayer A.G.Dosage is 8 μ g/100g, and dosage is the 0.04ml/100g body weight; 1 group of embodiment, 2 groups of embodiment, dosage is 0.16ml/100g body weight.
Medication: embodiment 1, nimotop 1h sublingual vein drug administration by injection 3 times altogether after 10 min, the modeling before modeling, after the modeling; The prevention administration is 3 before embodiment 2 arts, every day 1 time, and 0.5h modeling after the last administration, the postoperative medication is with embodiment 1.
Experimental data statistics employing mean ± standard deviation (
± SD) expression.Experimental result relatively, is carried out the t check through between group.The result is with the p value representation.Mean is the average between one group of data of expression.Standard deviation is the index of one group of data discrete degree of expression.The t check is that the significance of regression parameter is tested, and the p value is the declining indicator of credible result degree.P<0.05 is for having significant difference between group, and P<0.01 is for having significant differences between group.
Three, drug effect:
Two embodiment all can improve hemorheology index.For example, the red cell deformability that can raise reduces erythrocyte aggregation, reduces platelet aggregation rate.Can reduce whole blood viscosity and plasma viscosity.Prompting is one of mechanism of its treating cerebral ischemia to hemorheological improvement effect.Wherein embodiment 1 is better than embodiment 2.Show following (seeing table 1-3 for details):
1. after rat forms the middle cerebral artery thrombosis, can cause lower erythrocytes deformability, erythrocyte aggregation raises, and platelet aggregation rate raises, and (height is cut 200S to whole blood viscosity
-1, in cut 30 S
-1With low 5 S that cut
-1) and plasma viscosity (100 S
-1) all the compared with normal group increase to some extent, the variation that hemorheology is is sticking, dense, solidifying, gather state be described in the generating process of cerebral ischemia.
2. 1 pair of 24 hours middle cerebral artery thrombus model Mus blood viscosity of embodiment and hemorheological improvement effect are better than embodiment 2, and have significant difference in following index:
1 group of 24h whole blood reduced viscosity (height is cut): embodiment is starkly lower than 2 groups of embodiment, P<0.05; 1 group of 24h whole blood reduced viscosity (low cut): embodiment is starkly lower than 2 groups of embodiment, P<0.05.
Table 1. tested material on the impact of 24h middle cerebral artery thrombus model blood viscosity (
± SD)
Annotate: each group is compared * P<0.05 with model group; * P<0.01
Table 2. tested material on the impact of 48h middle cerebral artery thrombus model blood viscosity (
± SD)
Annotate: each group is compared * P<0.05 with model group; * P<0.01; Embodiment 1 compares with embodiment 2, # P<0.05
Table 3. tested material on the hemorheological impact of 24h middle cerebral artery thrombus model (
± SD)
Annotate: each group is all compared * P<0.05 with model group; * P<0.01; 1 group of embodiment compares # P<0.05 with 2 groups of embodiment.
Claims (2)
1. improve the preparation method that contains the ephedrine injection of rat blood rheology obstacle, it is characterized in that,
Taking by weighing purity is at least 98% sedanolide 0.16g, purity and is at least 98% gastrodine 0.024g, purity and is at least 98% ephedrine 0.24g, be added in percent by volume and be among 5% the Tween-81 85ml, stir, the compositions that makes described sedanolide, gastrodine and ephedrine is that concentration in 5% the Tween-81 aqueous solution is 5mg/ml in percent by volume.
2. that improves rat blood rheology obstacle contains the ephedrine injection preparation, it is characterized in that,
Taking by weighing purity is at least 98% sedanolide 0.16g, purity and is at least 98% gastrodine 0.01g, purity and is at least 98% ephedrine 0.24g, in percent by volume is 5% Tween-81 82ml, stir, the compositions that makes described sedanolide, gastrodine and ephedrine is that concentration in 5% the Tween-81 aqueous solution is 5mg/ml in percent by volume.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN 201210151403 CN102688184B (en) | 2012-05-15 | 2012-05-15 | Preparation method of ephedrine hydrochloride injection for improving rat blood rheological obstacle |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN 201210151403 CN102688184B (en) | 2012-05-15 | 2012-05-15 | Preparation method of ephedrine hydrochloride injection for improving rat blood rheological obstacle |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN102688184A CN102688184A (en) | 2012-09-26 |
| CN102688184B true CN102688184B (en) | 2013-10-16 |
Family
ID=46854033
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN 201210151403 Expired - Fee Related CN102688184B (en) | 2012-05-15 | 2012-05-15 | Preparation method of ephedrine hydrochloride injection for improving rat blood rheological obstacle |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN102688184B (en) |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10869845B1 (en) | 2020-01-22 | 2020-12-22 | Nevakar Inc. | Ephedrine compositions and methods |
Family Cites Families (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1843775A1 (en) * | 2005-02-04 | 2007-10-17 | DSMIP Assets B.V. | Novel composition comprising ligustilide and process for their manufacture |
| CN101677989B (en) * | 2008-02-14 | 2012-10-10 | 清华大学 | A drug composition for treatment and prevention of ischemic stroke and its preparation methods |
-
2012
- 2012-05-15 CN CN 201210151403 patent/CN102688184B/en not_active Expired - Fee Related
Also Published As
| Publication number | Publication date |
|---|---|
| CN102688184A (en) | 2012-09-26 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN101647776B (en) | Doxofylline venous injection with small volume as well as preparation method and quality control method thereof | |
| CN102688184B (en) | Preparation method of ephedrine hydrochloride injection for improving rat blood rheological obstacle | |
| CN104688784B (en) | Purposes of the ginkgolides in the drug for preparing blood pressure lowering | |
| CN100391506C (en) | Medicine for treating dementia and preparing method thereof | |
| CN102961517B (en) | Pharmaceutical composition for treating cardiovascular and cerebrovascular disease | |
| Taiwo et al. | Cardiovascular effects of Vernonia amygdalina in rats and the implications for treatment of hypertension in diabetes | |
| RU2600668C1 (en) | Method of infusion support for caesarean section under spinal anaesthesia | |
| CN104586866A (en) | A pharmaceutical composition for treating cerebrovascular diseases | |
| CN107296808B (en) | Application of compound epothilone B in preparation of medicine for repairing corneal nerve trauma | |
| CN103550702A (en) | Traditional Chinese medicine composition for cerebral arterial thrombosis | |
| JP4718443B2 (en) | Radixnotoginshen saponin family intravenous injection and method for its preparation | |
| Sinulingga et al. | Hemostatic effect of ethanol extract of Piper betle, Linn leaves to male mice | |
| CN102579568B (en) | Pure traditional Chinese medicine composition for treating various hemorrhage diseases and preparation method of same | |
| Dar et al. | Comparison of intrathecal hyperbaric bupivacaine and levobupivacaine for Cesarean section. | |
| CN102940657A (en) | Medicine composition for treating cardia-cerebrovascular diseases | |
| CN106619701A (en) | Western medicine composition for treating coronary heart disease and preparation method thereof | |
| Roberts | The CRASH-2 trial of an antifibrinolytic agent in traumatic haemorrhage: an international collaboration | |
| CN107737108A (en) | A kind of combination of oral medication for treating Pathogenesis of Post-infarction Ventricular Remodeling | |
| CN101919812B (en) | Novel formulation of piperazine ferulate and preparation method thereof | |
| RU2431488C1 (en) | Pharmaceutical composition of hydroxyethyl starch in hypertonic saline used as plasma volume replacement solution in low-volume resuscitation | |
| CN107281208B (en) | Pharmaceutical composition for preventing and treating cerebral apoplexy | |
| WO2021180087A1 (en) | Medical use of anyhdroicaritin | |
| Nannapaneni et al. | Managing a rivaroxaban bleed: understanding the difficulties in acute reversal of the new oral anticoagulants through a case report | |
| CN102697776B (en) | Pharmaceutical composition with function of treating cerebrovascular diseases | |
| CN1879637B (en) | A medicine for treating cardiovascular and cerebrovascular disease |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C14 | Grant of patent or utility model | ||
| GR01 | Patent grant | ||
| CF01 | Termination of patent right due to non-payment of annual fee |
Granted publication date: 20131016 Termination date: 20160515 |
|
| CF01 | Termination of patent right due to non-payment of annual fee |