CN102327444A - 一种治疗腹泻型褥疮的中药洗剂制备方法 - Google Patents
一种治疗腹泻型褥疮的中药洗剂制备方法 Download PDFInfo
- Publication number
- CN102327444A CN102327444A CN201110294763A CN201110294763A CN102327444A CN 102327444 A CN102327444 A CN 102327444A CN 201110294763 A CN201110294763 A CN 201110294763A CN 201110294763 A CN201110294763 A CN 201110294763A CN 102327444 A CN102327444 A CN 102327444A
- Authority
- CN
- China
- Prior art keywords
- grams
- herba
- gram
- radix
- traditional chinese
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
Abstract
Description
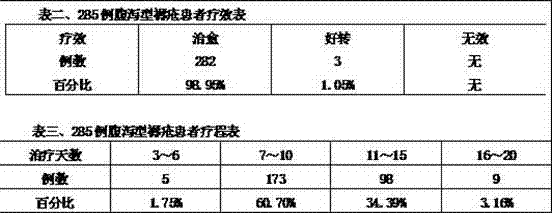
| 年龄(岁) | 17~20 | 21~30 | 31~40 | 41~55 |
| 例数 | 5 | 132 | 145 | 3 |
| 百分比 | 1.75% | 46.32% | 50.88% | 1.05% |
Claims (1)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201110294763A CN102327444B (zh) | 2011-09-28 | 2011-09-28 | 一种治疗腹泻型褥疮的中药洗剂制备方法 |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201110294763A CN102327444B (zh) | 2011-09-28 | 2011-09-28 | 一种治疗腹泻型褥疮的中药洗剂制备方法 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN102327444A true CN102327444A (zh) | 2012-01-25 |
| CN102327444B CN102327444B (zh) | 2012-10-24 |
Family
ID=45479575
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201110294763A Expired - Fee Related CN102327444B (zh) | 2011-09-28 | 2011-09-28 | 一种治疗腹泻型褥疮的中药洗剂制备方法 |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN102327444B (zh) |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102614415A (zh) * | 2012-02-27 | 2012-08-01 | 路维玲 | 一种治疗脓水型臁疮的中药洗剂制备方法 |
| CN102631592A (zh) * | 2012-04-02 | 2012-08-15 | 顾玉栋 | 一种治疗脓血型结肠炎的中药制备方法 |
| CN102743617A (zh) * | 2012-06-17 | 2012-10-24 | 邢霞 | 一种治疗腰痛型慢性子宫颈炎的中药冲洗剂制备方法 |
| CN103142718A (zh) * | 2013-03-11 | 2013-06-12 | 刘玉祥 | 一种治疗冻伤的药液 |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101658610A (zh) * | 2009-09-21 | 2010-03-03 | 石景洋 | 治疗褥疮的中药制剂及其制备方法 |
-
2011
- 2011-09-28 CN CN201110294763A patent/CN102327444B/zh not_active Expired - Fee Related
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101658610A (zh) * | 2009-09-21 | 2010-03-03 | 石景洋 | 治疗褥疮的中药制剂及其制备方法 |
Non-Patent Citations (1)
| Title |
|---|
| 马幸福: "褥疮的中药局部治疗概况", 《中医药临床杂志》 * |
Cited By (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102614415A (zh) * | 2012-02-27 | 2012-08-01 | 路维玲 | 一种治疗脓水型臁疮的中药洗剂制备方法 |
| CN102631592A (zh) * | 2012-04-02 | 2012-08-15 | 顾玉栋 | 一种治疗脓血型结肠炎的中药制备方法 |
| CN102631592B (zh) * | 2012-04-02 | 2013-09-04 | 顾玉栋 | 一种治疗脓血型结肠炎的中药制备方法 |
| CN102743617A (zh) * | 2012-06-17 | 2012-10-24 | 邢霞 | 一种治疗腰痛型慢性子宫颈炎的中药冲洗剂制备方法 |
| CN102743617B (zh) * | 2012-06-17 | 2013-11-27 | 张霞 | 一种治疗腰痛型慢性子宫颈炎的中药冲洗剂制备方法 |
| CN103142718A (zh) * | 2013-03-11 | 2013-06-12 | 刘玉祥 | 一种治疗冻伤的药液 |
| CN103142718B (zh) * | 2013-03-11 | 2014-11-12 | 刘玉祥 | 一种治疗冻伤的药液 |
Also Published As
| Publication number | Publication date |
|---|---|
| CN102327444B (zh) | 2012-10-24 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN102319372B (zh) | 一种治疗心经郁热型褥疮的中药洗剂制备方法 | |
| CN102366564B (zh) | 一种治疗溲赤型褥疮的中药洗剂制备方法 | |
| CN102366565B (zh) | 一种治疗热毒型褥疮的中药制备方法 | |
| CN102327515B (zh) | 一种治疗水肿型褥疮的中药洗剂制备方法 | |
| CN102631592B (zh) | 一种治疗脓血型结肠炎的中药制备方法 | |
| CN102327517B (zh) | 一种治疗风火型褥疮的中药洗剂制备方法 | |
| CN102327443B (zh) | 一种治疗瘙痒型褥疮的中药洗剂的制备方法 | |
| CN102327444B (zh) | 一种治疗腹泻型褥疮的中药洗剂制备方法 | |
| CN102327563A (zh) | 一种治疗疼痛型褥疮的中药洗剂制备方法 | |
| CN102319374B (zh) | 一种治疗咳嗽型褥疮的中药洗剂制备方法 | |
| CN102327442B (zh) | 一种治疗不寐型褥疮的中药洗剂制备方法 | |
| CN102366547B (zh) | 一种治疗便秘型褥疮的中药洗剂制备方法 | |
| CN102380041B (zh) | 一种治疗盗汗型褥疮的中药洗剂的制备方法 | |
| CN102380081B (zh) | 一种治疗麻木型褥疮的中药洗剂制备方法 | |
| CN102309741B (zh) | 一种治疗经久不敛型褥疮的中药洗剂制备方法 | |
| CN102631577B (zh) | 一种治疗溲赤型足底疔的中药洗剂制备方法 | |
| CN102319404B (zh) | 一种治疗酗酒型褥疮的中药洗剂制备方法 | |
| CN101934035B (zh) | 一种治疗瘀血红润期褥疮的中药制备方法 | |
| CN102327567B (zh) | 一种治疗暴饮暴食型褥疮的中药洗剂制备方法 | |
| CN102631576B (zh) | 一种治疗便秘型足底疔的中药洗剂制备方法 | |
| CN102631575B (zh) | 一种治疗热毒型足底疔的中药洗剂制备方法 | |
| CN102327564B (zh) | 一种治疗过食油腻型褥疮的中药洗剂制备方法 | |
| CN102327497B (zh) | 一种治疗惊厥型褥疮的中药洗剂制备方法 | |
| CN102366546B (zh) | 一种治疗血瘀型褥疮的中药制备方法 | |
| CN102327561B (zh) | 一种治疗肝郁型褥疮的中药洗剂制备方法 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| ASS | Succession or assignment of patent right |
Owner name: CENG ZHAOXIANG Free format text: FORMER OWNER: MA NIANFANG Effective date: 20120903 |
|
| C41 | Transfer of patent application or patent right or utility model | ||
| C53 | Correction of patent of invention or patent application | ||
| CB03 | Change of inventor or designer information |
Inventor after: Zeng Zhaoxiang Inventor after: Wei Yueling Inventor before: Ma Nianfang |
|
| COR | Change of bibliographic data |
Free format text: CORRECT: ADDRESS; FROM: 250013 JINAN, SHANDONG PROVINCE TO: 272051 JINING, SHANDONG PROVINCE Free format text: CORRECT: INVENTOR; FROM: MA NIANFANG TO: CENG ZHAOXIANG WEI YUELING |
|
| TA01 | Transfer of patent application right |
Effective date of registration: 20120903 Address after: 272051 Shandong province Jining City Ji Dai Road Jining city mental hospital Applicant after: Zeng Zhaoxiang Address before: 46 Shandong Chest Hospital, Lixia mountain road, Lixia District, Shandong, Ji'nan 250013, China Applicant before: Ma Nianfang |
|
| C14 | Grant of patent or utility model | ||
| GR01 | Patent grant | ||
| C17 | Cessation of patent right | ||
| CF01 | Termination of patent right due to non-payment of annual fee |
Granted publication date: 20121024 Termination date: 20130928 |