CN101804318A - Preparation of lanthanum doping cerium dioxide porous microspheres and application thereof to Cr<6+> removal - Google Patents
Preparation of lanthanum doping cerium dioxide porous microspheres and application thereof to Cr<6+> removal Download PDFInfo
- Publication number
- CN101804318A CN101804318A CN 201010151035 CN201010151035A CN101804318A CN 101804318 A CN101804318 A CN 101804318A CN 201010151035 CN201010151035 CN 201010151035 CN 201010151035 A CN201010151035 A CN 201010151035A CN 101804318 A CN101804318 A CN 101804318A
- Authority
- CN
- China
- Prior art keywords
- cerium dioxide
- porous microspheres
- dioxide porous
- lanthanum doping
- doping cerium
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Images
Abstract
The invention provides preparation of lanthanum doping cerium dioxide porous microspheres and application thereof to Cr<6+> removal. The lanthanum doping cerium dioxide porous microspheres are prepared by using a coprecipitation method, so the lanthanum doping cerium dioxide porous microspheres have the characteristics of small size, high specific surface area and high activity of nanometer materials and the characteristics of rich hole passages, high specific surface area, high hole volume and the like of porous materials. The method of the invention has the following concrete steps: preparing solid Ce(NO3)3.6H2O and La(NO3)3.6H2O into a required solution according to the atomic ratio; stirring and heating the mixed solution; then, dripping a formic acid into the mixed solution; repeatedly washing and drying the materials after the centrifugation; then, placing the materials into a muffle furnace to be sintered; and obtaining the lanthanum doping cerium dioxide porous microspheres after cooling. The preparation method of the lanthanum doping cerium dioxide porous microspheres of the invention is simple, the synthesis temperature is low, the cost can be saved, and the preparation effect is good. The lanthanum doping cerium dioxide porous microspheres have high Cr<6+> removal capability in the aspect of sewage treatment, have certain application prospects in the aspect of pollution control, and belong to an ideal rare-earth absorbing agent.
Description
Technical field
The invention belongs to rare earth oxide material field or technical field of waste water processing, relate to a kind of preparation method and application thereof of lanthanum doping cerium dioxide porous microspheres, relate in particular to a kind of preparation of lanthanum doping cerium dioxide porous microspheres and as adsorbent to Cr in the aqueous solution
6+Removal use.
Background technology
Cerium oxide is a kind of cheapness and the extremely wide Multifunction material of purposes.Because its unique cubic system fluorite structure and high oxygen ionic conductivity, oxygen storage capacity, participate in redox reaction, characteristics such as specific area is big, make it extensively apply to heterogeneous absorption, SOFC, lambda sensor, the low temperature water gas shift reation, fields such as the polishing of oxygen permeable membrane system, luminescent material, UV absorption material, glass, radiation resistant glass, electronic ceramics.
Because the specific area of nanometer powder is big, active high, therefore along with the development of rare earth new material, the performance of rare earth nano powder in materials such as magnetic material, superconductor, fluorescent material, sensor and superhigh temperature heat-resisting alloy is existing significantly to be improved.Yet nano particle is because of the undersized shortcomings such as being easy to reunite, being difficult to recycling that often exists, and therefore in recent years, micron-sized nanometer assembled material has caused the more concerns of people.It is found that micron-sized nanometer assembling CeO
2Have new excellent properties and application, CeO
2Preparation, functional characteristic and the application study of base Nano/micron material have become a problem that presses for research.
Heavy metal chromium and compound thereof are widely used industrial, plating, process hides, pigment, chemical industry, pharmacy, chromium salt production, industry light industry textile industry, mining smelting etc.Chromium is often with trivalent (Cr in water
3+) and sexavalence ion (Cr
6+) the form existence.In the industrial wastewater, chromium mainly exists with the sexavalence form, Cr
6+Have very strong bio-toxicity, can pass through alimentary canal, skin and mucous membrane and invade health, cause nausea, vomiting, rhinitis, laryngitis, eczema etc., under the long term, can cause illnesss such as anaemia, pulmonary emphysema, bronchiectasis, this is with the serious threat human health.Processing method commonly used has electronation and the precipitation method, ion-exchange, the barium chromate precipitation method, electrolysis, absorption method etc., wherein absorption method have simple to operate, characteristics such as investment cost is few, and treatment effect is good thereby quite paid attention to.
Summary of the invention
At the deficiencies in the prior art, the purpose of this invention is to provide the lanthanum doping titanium dioxide porous microsphere that a kind of low temperature simple method for preparing acquisition cheaply tool nano material small size, high-specific surface area, high activity and porous material enrich features such as duct, high-specific surface area, high pore volume.
A kind of preparation method of lanthanum doping cerium dioxide porous microspheres is to utilize the coprecipitation preparation to have the lanthanum doping cerium dioxide porous microspheres powder that nano material small size, high-specific surface area, high activity and porous material enrich duct, high-specific surface area, high pore volume concurrently.
A kind of preparation method of lanthanum doping cerium dioxide porous microspheres may further comprise the steps:
Step 1: with La (NO
3)
36H
2O, Ce (NO
3)
36H
2O presses La
3+Mix the atom proportioning and be 10%~60% and be dissolved in and be mixed with reaction solution in the absolute ethyl alcohol, it is 0.1molL that GOLD FROM PLATING SOLUTION belongs to the ion concentration summation
-1, entire reaction course is that mixed solution is heated with stirring to 60~90 ℃, after, to La/Ce mixed solution and dripping formic acid solution, make that the concentration of H+ in reaction is 0.01~0.24molL
-1Between; Metal ion is fully precipitated, form white suspension.
Step 2: gained white suspension is centrifugal, use the ethanol cyclic washing, oven dry obtains presoma, and is standby.
Step 3: with presoma sintering in Muffle furnace of gained in the step 2, sintering temperature is 350~500 ℃, and sintering time 2~4h promptly obtains lanthanum doping cerium dioxide porous microspheres after the cooling.
Another object of the present invention be the lanthanum doping titanium dioxide porous microsphere that will prepare as rare-earth adsorbent to Cr in the aqueous solution
6+Remove.
The lanthanum doping cerium dioxide porous microspheres for preparing as stated above as rare-earth adsorbent to Cr in the aqueous solution
6+Removal use.Its method is:
The doping cerium dioxide porous microspheres of getting the multiple lanthanum content for preparing in right amount places Cr as adsorbent
6+Content is 0~60mgL
-1The aqueous solution in, the adsorbent use amount is 0.4~6gL
-1At room temperature, with 1molL
-1HCl and 1molL
-1NH
3H
2O conditioned reaction system pH is 1~6, and the reaction time is 1~1380min, Cr
6+Clearance is 15~95%.
The described aqueous solution is Cr
6+Content is 0~60mgL
-1The aqueous solution.
The described aqueous solution is with K
2Cr
2O
7The Cr that is mixed with for raw material
6+Content is 0~60mgL
-1The aqueous solution.
Lanthanum doping cerium dioxide porous microspheres as adsorbent to Cr in the aqueous solution
6+Removal use.Studied different lanthanum dopings, different pH values, different adsorbent consumption, different adsorption time are to the influence of experimental result.Its characterization result is as follows: lanthanum doping cerium dioxide porous micron ball largest specific surface area reaches 197m
2G
-1And under the room temperature, for Cr
6+Ion concentration is 20mgL
-1The aqueous solution, the optimal adsorption condition is for selecting for use lanthanum doping 20% porous micron ball as rare-earth adsorbent, consumption is 2gL
-1, pH value of solution is 3, adsorption time 5h, Cr
6+Clearance can reach 97%.
The invention has the advantages that: the preparation method of a kind of lanthanum doping cerium dioxide porous microspheres of the present invention is simple, and synthesis temperature is low; Investment cost is few, prepares advantages such as effective.Aspect sewage disposal, find that it has to remove Cr preferably
6+Ability, at the pollution controlling party mask of heavy metal ion certain application prospect is arranged, be a kind of more satisfactory rare earth based adsorbent.
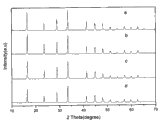
Description of drawings
The XRD figure spectrum of Fig. 1 lanthanum doping 10%, 20%, 40%, 60% presoma cesium formate;
Fig. 2 sintering temperature is 400 ℃, and sintering time is 10%, 20%, 40%, the 60% lanthanum doped Ce O that synthesizes under the condition of 3h
2XRD figure spectrum;
Fig. 3 sintering temperature is 400 ℃, and sintering time is the 20% lanthanum doped Ce O that synthesizes under the condition of 3h
2Sem photograph;
The sample of the different lanthanum dopings of Fig. 4 is to Cr
6+Absorption property.
The specific embodiment
Embodiment one
With 1.7369g Ce (NO
3)
36H
2O and 0.4330g La (NO
3)
36H
2The O mixing is dissolved in the 50mL absolute ethyl alcohol, and high degree of agitation 20min is heated to mixed solution 75 ℃ simultaneously, slowly drips formic acid solution then in mixed solution, and metal ion is fully precipitated.With gained white suspension in 9000rmin
-1Centrifugal, use the absolute ethyl alcohol cyclic washing, oven dry.With gained presoma 400 ℃ of roasting 3h in Muffle furnace, promptly obtain the pale yellow powder of 20% lanthanum doping cerium dioxide porous microspheres after the cooling, the surface area of this sample is 197m
2G
-1
Fig. 1 is the XRD figure spectrum of lanthanum doping 10%, 20%, 40%, 60% presoma cesium formate, as seen from the figure, along with improving constantly to 60% of lanthanum doping, still can synthesize pure cesium formate.Fig. 2 is that sintering temperature is 400 ℃, and sintering time is 10%, 20%, 40%, the 60% lanthanum doped Ce O that synthesizes under the condition of 3h
2XRD figure spectrum, as seen from the figure, along with improving constantly of lanthanum doping, the angle of diffraction constantly is offset to low angle.Fig. 3 is that sintering temperature is 400 ℃, and sintering time is the 20% lanthanum doped Ce O that synthesizes under the condition of 3h
2Sem photograph.
Embodiment two
With 1.95404g Ce (NO
3)
36H
2O and 0.21651g La (NO
3)
36H
2The O mixing is dissolved in the 50mL absolute ethyl alcohol, and high degree of agitation 20min is heated to mixed solution 75 ℃ simultaneously, slowly drips formic acid solution then in mixed solution, and metal ion is fully precipitated.With gained white suspension in 9000rmin
-1Centrifugal, use the absolute ethyl alcohol cyclic washing, oven dry.With gained presoma 400 ℃ of roasting 3h in Muffle furnace, promptly obtain the pale yellow powder of 10% lanthanum doping cerium dioxide porous microspheres after the cooling, the surface area of this sample is 147m
2G
-1
Embodiment three
Take by weighing the 20% cerium dioxide porous micron ball 0.05g of lanthanum doping for preparing, joining initial concentration is 10mgL
-1Cr
6+Among the solution 25mL, use 1molL
-1HCl and 1molL
-1NH
3H
2The pH value of O regulator solution is respectively between 2~6, and adsorption time 5h measures remaining Cr in the solution
6+Ion concentration.Through the characterization result analysis: the lanthanum doping is that 20% cerium dioxide porous micron ball is 10mgL at initial concentration
-1Cr
6+In the solution, the adsorption capacity under the acid situation is all about 95%.The sample of the different lanthanum dopings of Fig. 4 is to Cr
6+Absorption property.
Embodiment four
Take by weighing the 20% cerium dioxide porous micron ball 0.05g of lanthanum doping for preparing, joining initial concentration is 20mgL
-1Cr
6+Among the solution 25mL, use 1molL
-1HCl and 1molL
-1NH
3H
2The pH value of O regulator solution is respectively between 2~6, and adsorption time 5h measures remaining Cr in the solution
6+Ion concentration.Through the characterization result analysis: initial concentration is 20mgL
-1Cr
6+In the solution, solution reaches the optimal adsorption amount at pH=3.
Claims (8)
1. the preparation method of a lanthanum doping cerium dioxide porous microspheres is characterized in that, described preparation method may further comprise the steps:
Step 1: with La (NO
3)
36H
2O, Ce (NO
3)
36H
2O presses La
3+Mix the atom proportioning and be 10%~60% and be dissolved in and be mixed with reaction solution in the absolute ethyl alcohol, it is 0.1molL that GOLD FROM PLATING SOLUTION belongs to the ion concentration summation
-1, entire reaction course is after mixed solution is heated with stirring to 60~90 ℃, to La/Ce mixed solution and dripping formic acid solution, makes H
+Concentration in reaction is 0.01~0.24molL
-1Between; Metal ion is fully precipitated, form white suspension;
Step 2: gained white suspension is centrifugal, use the ethanol cyclic washing, oven dry obtains presoma, and is standby;
Step 3: with presoma sintering in Muffle furnace of gained in the step 2, sintering temperature is 350~500 ℃, and sintering time 2~4h promptly obtains lanthanum doping cerium dioxide porous microspheres after the cooling.
2. the preparation method of lanthanum doping cerium dioxide porous microspheres according to claim 1 is characterized in that: the speed that drips formic acid solution in the described step 1 is 0.5~2mLmin
-1
3. the preparation method of lanthanum doping cerium dioxide porous microspheres according to claim 1 is characterized in that: in the described step 1, and H
+Concentration in reaction is 0.04~0.18molL
-1Between.
4. the preparation method of lanthanum doping cerium dioxide porous microspheres according to claim 1 is characterized in that: entire reaction course is that mixed solution is stirred 10~20min in the described step 1, simultaneously mixed solution is heated between 70~80 ℃.
5. the preparation method of lanthanum doping cerium dioxide porous microspheres according to claim 1, it is characterized in that: the lanthanum doping cerium dioxide porous microspheres that makes has cube fluorite structure of ceria, and yardstick is 100nm~4 μ m, and specific area is 140~230m
2G
-1, average pore size is 2~10nm.
6. a lanthanum doping cerium dioxide porous microspheres is to Cr
6+Removal use, it is characterized in that: the lanthanum doping cerium dioxide porous microspheres that will prepare by described preparation method as adsorbent to Cr in the aqueous solution
6+Remove, the adsorbent consumption is 0.4~6gL
-1At room temperature, with 1molL
-1HCl and 1molL
-1NH
3H
2O conditioned reaction system pH is 1~6, and adsorption time is 1~1380min, Cr
6+Clearance is 15~95%.
7. lanthanum doping cerium dioxide porous microspheres according to claim 6 is to Cr
6+Removal use, it is characterized in that: the described aqueous solution is Cr
6+Content is 10~60mgL
-1The aqueous solution.
According to any described lanthanum doping cerium dioxide porous microspheres in claim 6 or 7 to Cr
6+Removal use, it is characterized in that:
The lanthanum doping of described lanthanum doping cerium dioxide porous micron ball is 20%, and specific area is 197m
2G
-1, for Cr
6+Ion concentration is 20mgL
-1The aqueous solution, the adsorbent addition is 2gL
-1, pH value of solution is 3, adsorption time 5h, Cr
6+Clearance is 97%.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN 201010151035 CN101804318A (en) | 2010-04-16 | 2010-04-16 | Preparation of lanthanum doping cerium dioxide porous microspheres and application thereof to Cr<6+> removal |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN 201010151035 CN101804318A (en) | 2010-04-16 | 2010-04-16 | Preparation of lanthanum doping cerium dioxide porous microspheres and application thereof to Cr<6+> removal |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN101804318A true CN101804318A (en) | 2010-08-18 |
Family
ID=42606372
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN 201010151035 Pending CN101804318A (en) | 2010-04-16 | 2010-04-16 | Preparation of lanthanum doping cerium dioxide porous microspheres and application thereof to Cr<6+> removal |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN101804318A (en) |
Cited By (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102078793A (en) * | 2010-11-25 | 2011-06-01 | 北京科技大学 | Hydrothermal preparation of oxalic acid system cerium oxide sphere and application of oxalate system cerium oxide sphere for removing Cr<6+> |
| CN103596886A (en) * | 2011-04-13 | 2014-02-19 | 莫利康普矿物有限责任公司 | Rare earth removal of hydrated and hydroxyl species |
| CN106622109A (en) * | 2016-12-15 | 2017-05-10 | 北京工业大学 | Preparation method of lanthanum modified red mud chromium removing adsorbent |
| CN107029794A (en) * | 2017-04-01 | 2017-08-11 | 滨州学院 | A kind of catalyst for handling esterification waste water |
| CN108408756A (en) * | 2018-03-02 | 2018-08-17 | 复旦大学 | Cerium dioxide micro-balloon and its preparation method and application with multistage nucleocapsid |
| US10577259B2 (en) | 2014-03-07 | 2020-03-03 | Secure Natural Resources Llc | Removal of arsenic from aqueous streams with cerium (IV) oxide compositions |
| CN114082396A (en) * | 2021-09-28 | 2022-02-25 | 淮阴师范学院 | Magnetic persimmon cake-shaped cerium ferrite/cerium dioxide composite adsorbent and preparation method thereof |
| CN115465879A (en) * | 2021-06-11 | 2022-12-13 | 四川大学 | Spherical CeO 2 Preparation method of (1) |
| CN115504500A (en) * | 2022-07-28 | 2022-12-23 | 中国矿业大学 | Preparation method of yttrium-doped cerium oxide and method for removing hexavalent chromium in sewage |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101070180A (en) * | 2006-12-12 | 2007-11-14 | 北京科技大学 | Method for preparing cerium oxide nano powder |
| CN101117233A (en) * | 2007-07-23 | 2008-02-06 | 北京科技大学 | Method for preparing high specific surface area meso-porous cerium dioxide micro-balloon |
-
2010
- 2010-04-16 CN CN 201010151035 patent/CN101804318A/en active Pending
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101070180A (en) * | 2006-12-12 | 2007-11-14 | 北京科技大学 | Method for preparing cerium oxide nano powder |
| CN101117233A (en) * | 2007-07-23 | 2008-02-06 | 北京科技大学 | Method for preparing high specific surface area meso-porous cerium dioxide micro-balloon |
Non-Patent Citations (2)
| Title |
|---|
| 《中国稀土学报》 20100410 曾俐等 掺杂二氧化铈多孔微球对水溶液中Cr6+去除作用 第252页左栏第2段,第253页左栏第4段至右栏最后1段以及图3 1-8 第28卷, 2 * |
| 《稀有金属材料与工程》 20091231 杨晓丹等 两步法制备镧掺杂氧化铈纳米粉体 第1067页右栏第2段,第1069页右栏第1段以及图5、6 1-5 第38卷, 2 * |
Cited By (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102078793A (en) * | 2010-11-25 | 2011-06-01 | 北京科技大学 | Hydrothermal preparation of oxalic acid system cerium oxide sphere and application of oxalate system cerium oxide sphere for removing Cr<6+> |
| CN102078793B (en) * | 2010-11-25 | 2012-09-19 | 北京科技大学 | Hydrothermal preparation of oxalic acid system cerium oxide sphere and application of oxalate system cerium oxide sphere for removing Cr<6+> |
| CN103596886A (en) * | 2011-04-13 | 2014-02-19 | 莫利康普矿物有限责任公司 | Rare earth removal of hydrated and hydroxyl species |
| US10577259B2 (en) | 2014-03-07 | 2020-03-03 | Secure Natural Resources Llc | Removal of arsenic from aqueous streams with cerium (IV) oxide compositions |
| CN106622109A (en) * | 2016-12-15 | 2017-05-10 | 北京工业大学 | Preparation method of lanthanum modified red mud chromium removing adsorbent |
| CN107029794B (en) * | 2017-04-01 | 2019-08-23 | 滨州学院 | A kind of catalyst handling esterification waste water |
| CN107029794A (en) * | 2017-04-01 | 2017-08-11 | 滨州学院 | A kind of catalyst for handling esterification waste water |
| CN108408756A (en) * | 2018-03-02 | 2018-08-17 | 复旦大学 | Cerium dioxide micro-balloon and its preparation method and application with multistage nucleocapsid |
| CN108408756B (en) * | 2018-03-02 | 2020-07-07 | 复旦大学 | Cerium dioxide microsphere with multistage core-shell structure and preparation method and application thereof |
| CN115465879A (en) * | 2021-06-11 | 2022-12-13 | 四川大学 | Spherical CeO 2 Preparation method of (1) |
| CN115465879B (en) * | 2021-06-11 | 2023-06-02 | 四川大学 | Spherical CeO 2 Is prepared by the preparation method of (2) |
| CN114082396A (en) * | 2021-09-28 | 2022-02-25 | 淮阴师范学院 | Magnetic persimmon cake-shaped cerium ferrite/cerium dioxide composite adsorbent and preparation method thereof |
| CN114082396B (en) * | 2021-09-28 | 2023-07-11 | 淮阴师范学院 | Magnetic persimmon cake-shaped cerium ferrite/cerium dioxide composite adsorbent and preparation method thereof |
| CN115504500A (en) * | 2022-07-28 | 2022-12-23 | 中国矿业大学 | Preparation method of yttrium-doped cerium oxide and method for removing hexavalent chromium in sewage |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN101804318A (en) | Preparation of lanthanum doping cerium dioxide porous microspheres and application thereof to Cr<6+> removal | |
| Zinatloo-Ajabshir et al. | Facile synthesis of Nd2Sn2O7-SnO2 nanostructures by novel and environment-friendly approach for the photodegradation and removal of organic pollutants in water | |
| Zinatloo-Ajabshir et al. | Green synthesis and characterization of Dy2Ce2O7 ceramic nanostructures with good photocatalytic properties under visible light for removal of organic dyes in water | |
| Xu et al. | Electrochemical synthesis of ammonia using a cell with a Nafion membrane and SmFe 0.7 Cu 0.3− x Ni x O 3 (x= 0− 0.3) cathode at atmospheric pressure and lower temperature | |
| CN101947454B (en) | Transitional metal-doped zinc oxide mesoporous material with visible light catalytic activity and preparation method thereof | |
| CN108383160B (en) | Preparation method and application of metal element doped BiOCl nanosheet material | |
| Zhang et al. | Surface characterization studies of CuO-CeO2-ZrO2 catalysts for selective catalytic reduction of NO with NH3 | |
| CN101584981B (en) | Low-temperature preparation method of ceria nano-crystalline microsphere of graduation structure | |
| Chen et al. | Magnetic recyclable lanthanum-nitrogen co-doped titania/strontium ferrite/diatomite heterojunction composite for enhanced visible-light-driven photocatalytic activity and recyclability | |
| CN111545192A (en) | MOFs-derived perovskite catalyst, preparation method thereof and application of MOFs-derived perovskite catalyst in catalytic degradation of organic pollutants | |
| Liu et al. | Proton conduction at intermediate temperature and its application in ammonia synthesis at atmospheric pressure of BaCe 1− x Ca x O 3− α | |
| CN102616861A (en) | Fe2O3 micro-nano porous sphere, preparation method thereof and uses thereof | |
| CN110743570A (en) | Preparation method of catalyst containing porous structure base material and method for decomposing formaldehyde by using catalyst | |
| CN109772465A (en) | A kind of preparation method of water solubility carbon dots modified perovskite type catalysis material | |
| CN105381797A (en) | Preparation of catalytic ozonation supported catalyst used for treating sewage and doped with dual rare earth elements | |
| Zhang et al. | Mesoporous delafossite CuCrO2 and spinel CuCr2O4: synthesis and catalysis | |
| CN103754953A (en) | Preparation method of multi-morphologynano-sized zinc ferrite | |
| CN107281999A (en) | A kind of ferriferous oxide/manganese dioxide nano-composite material and preparation method and application | |
| Du et al. | RE/ZrO2 (RE= Sm, Eu) composite oxide nano-materials: Synthesis and applications in photocatalysis | |
| CN104475094A (en) | Diatom ooze wall material applied to non-photo-catalysis purification of formaldehyde in air at room temperature | |
| Yin et al. | Synergistically enhanced photocatalytic degradation of tetracycline hydrochloride by Z-scheme heterojunction MT-BiVO4 microsphere/P-doped g-C3N4 nanosheet composite | |
| CN108714426A (en) | A kind of nanocube perovskite type catalyst and its preparation method and application | |
| CN107500336A (en) | A kind of method for preparing rare earth layered hydroxide using template method is sacrificed certainly | |
| CN103846090A (en) | Silicon dioxide compounded titanium dioxide catalyst for degrading coking wastewater as well as preparation method thereof | |
| CN100398195C (en) | Synthesis method of boron-doped nanometer oxide and use thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C02 | Deemed withdrawal of patent application after publication (patent law 2001) | ||
| WD01 | Invention patent application deemed withdrawn after publication |
Open date: 20100818 |