WO2000076495A1 - Il-8 receptor antagonists - Google Patents
Il-8 receptor antagonists Download PDFInfo
- Publication number
- WO2000076495A1 WO2000076495A1 PCT/US2000/016499 US0016499W WO0076495A1 WO 2000076495 A1 WO2000076495 A1 WO 2000076495A1 US 0016499 W US0016499 W US 0016499W WO 0076495 A1 WO0076495 A1 WO 0076495A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
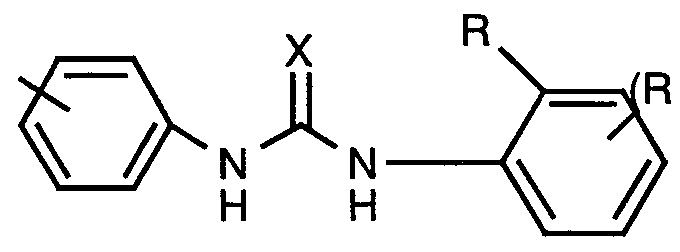
- urea
- hydroxy
- phenyl
- alkyl
- bromophenyl
- Prior art date
Links
- 108010038415 interleukin-8 receptors Proteins 0.000 title description 5
- 102000010681 interleukin-8 receptors Human genes 0.000 title description 4
- 239000002464 receptor antagonist Substances 0.000 title description 2
- 229940044551 receptor antagonist Drugs 0.000 title description 2
- 150000001875 compounds Chemical class 0.000 claims abstract description 165
- 238000000034 method Methods 0.000 claims abstract description 128
- 108090001007 Interleukin-8 Proteins 0.000 claims abstract description 63
- 201000010099 disease Diseases 0.000 claims abstract description 33
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 claims abstract description 33
- 102000019034 Chemokines Human genes 0.000 claims abstract description 32
- 108010012236 Chemokines Proteins 0.000 claims abstract description 32
- 150000003839 salts Chemical class 0.000 claims abstract description 24
- 230000001404 mediated effect Effects 0.000 claims abstract description 17
- 102000012740 beta Adrenergic Receptors Human genes 0.000 claims abstract description 13
- 108010079452 beta Adrenergic Receptors Proteins 0.000 claims abstract description 13
- 125000000217 alkyl group Chemical group 0.000 claims description 165
- 229910052739 hydrogen Inorganic materials 0.000 claims description 102
- 239000001257 hydrogen Substances 0.000 claims description 94
- 125000003545 alkoxy group Chemical group 0.000 claims description 79
- 125000000623 heterocyclic group Chemical group 0.000 claims description 76
- 125000001072 heteroaryl group Chemical group 0.000 claims description 72
- 125000003118 aryl group Chemical group 0.000 claims description 66
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 claims description 53
- 125000006374 C2-C10 alkenyl group Chemical group 0.000 claims description 51
- -1 methylene dioxy Chemical group 0.000 claims description 48
- 229910052736 halogen Inorganic materials 0.000 claims description 45
- 150000002367 halogens Chemical class 0.000 claims description 44
- 125000003107 substituted aryl group Chemical group 0.000 claims description 44
- 125000000475 sulfinyl group Chemical group [*:2]S([*:1])=O 0.000 claims description 42
- 150000002431 hydrogen Chemical class 0.000 claims description 33
- 125000003342 alkenyl group Chemical group 0.000 claims description 31
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims description 29
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 claims description 29
- 229910052757 nitrogen Inorganic materials 0.000 claims description 24
- 125000004093 cyano group Chemical group *C#N 0.000 claims description 23
- 125000004446 heteroarylalkyl group Chemical group 0.000 claims description 22
- IJGRMHOSHXDMSA-UHFFFAOYSA-N nitrogen Substances N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 claims description 18
- 229910052760 oxygen Inorganic materials 0.000 claims description 18
- 125000004104 aryloxy group Chemical group 0.000 claims description 17
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical group [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 claims description 16
- 150000001540 azides Chemical class 0.000 claims description 16
- 125000005842 heteroatom Chemical group 0.000 claims description 16
- 239000001301 oxygen Substances 0.000 claims description 16
- 229910052717 sulfur Chemical group 0.000 claims description 15
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical group [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 claims description 14
- 239000011593 sulfur Chemical group 0.000 claims description 14
- 241000124008 Mammalia Species 0.000 claims description 13
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 claims description 12
- 125000003710 aryl alkyl group Chemical group 0.000 claims description 11
- 125000002023 trifluoromethyl group Chemical group FC(F)(F)* 0.000 claims description 11
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims description 10
- QJGQUHMNIGDVPM-UHFFFAOYSA-N nitrogen group Chemical group [N] QJGQUHMNIGDVPM-UHFFFAOYSA-N 0.000 claims description 10
- JFLMWUAMXJXUAG-UHFFFAOYSA-N 1-(2-chlorophenyl)-3-(2-hydroxy-4-nitrophenyl)urea Chemical compound OC1=CC([N+]([O-])=O)=CC=C1NC(=O)NC1=CC=CC=C1Cl JFLMWUAMXJXUAG-UHFFFAOYSA-N 0.000 claims description 6
- PMMYTIINRJXKSU-UHFFFAOYSA-N 1-(2-hydroxy-4-nitrophenyl)-3-(2-iodophenyl)urea Chemical compound OC1=CC([N+]([O-])=O)=CC=C1NC(=O)NC1=CC=CC=C1I PMMYTIINRJXKSU-UHFFFAOYSA-N 0.000 claims description 6
- BNWWISXBCWURFH-UHFFFAOYSA-N 3-[(2-bromophenyl)carbamoylamino]-2-hydroxy-n-phenylbenzamide Chemical compound C1=CC=C(C(=O)NC=2C=CC=CC=2)C(O)=C1NC(=O)NC1=CC=CC=C1Br BNWWISXBCWURFH-UHFFFAOYSA-N 0.000 claims description 6
- WTBSKJMUSCALAE-UHFFFAOYSA-N 1-(2,3-dichlorophenyl)-3-(2-hydroxy-4-nitrophenyl)urea Chemical compound OC1=CC([N+]([O-])=O)=CC=C1NC(=O)NC1=CC=CC(Cl)=C1Cl WTBSKJMUSCALAE-UHFFFAOYSA-N 0.000 claims description 5
- UUDFKBXAWIEQJG-UHFFFAOYSA-N 1-(2-bromophenyl)-3-(2-hydroxy-3-nitrophenyl)urea Chemical compound C1=CC=C([N+]([O-])=O)C(O)=C1NC(=O)NC1=CC=CC=C1Br UUDFKBXAWIEQJG-UHFFFAOYSA-N 0.000 claims description 5
- MQBZVUNNWUIPMK-UHFFFAOYSA-N 1-(2-bromophenyl)-3-(2-hydroxy-4-nitrophenyl)urea Chemical compound OC1=CC([N+]([O-])=O)=CC=C1NC(=O)NC1=CC=CC=C1Br MQBZVUNNWUIPMK-UHFFFAOYSA-N 0.000 claims description 5
- DNYOWRQDSNGAQQ-UHFFFAOYSA-N 1-(2-bromophenyl)-3-(3-cyano-2-hydroxy-4-methylphenyl)urea Chemical compound OC1=C(C#N)C(C)=CC=C1NC(=O)NC1=CC=CC=C1Br DNYOWRQDSNGAQQ-UHFFFAOYSA-N 0.000 claims description 5
- KNPLCJZGNRGZAN-UHFFFAOYSA-N 1-(2-hydroxy-4-nitrophenyl)-3-(2-methoxyphenyl)urea Chemical compound COC1=CC=CC=C1NC(=O)NC1=CC=C([N+]([O-])=O)C=C1O KNPLCJZGNRGZAN-UHFFFAOYSA-N 0.000 claims description 5
- MHEWCJHSRKVNIF-UHFFFAOYSA-N 1-(2-hydroxy-4-nitrophenyl)-3-(2-phenoxyphenyl)urea Chemical compound OC1=CC([N+]([O-])=O)=CC=C1NC(=O)NC1=CC=CC=C1OC1=CC=CC=C1 MHEWCJHSRKVNIF-UHFFFAOYSA-N 0.000 claims description 5
- GTJXCKKJNGSTCK-UHFFFAOYSA-N 1-(3-chloro-2-methoxyphenyl)-3-(2-hydroxy-4-nitrophenyl)urea Chemical compound COC1=C(Cl)C=CC=C1NC(=O)NC1=CC=C([N+]([O-])=O)C=C1O GTJXCKKJNGSTCK-UHFFFAOYSA-N 0.000 claims description 5
- ZHFMJOHHDQGOTA-UHFFFAOYSA-N 1-(4-cyano-2-hydroxyphenyl)-3-(2-methoxyphenyl)urea Chemical compound COC1=CC=CC=C1NC(=O)NC1=CC=C(C#N)C=C1O ZHFMJOHHDQGOTA-UHFFFAOYSA-N 0.000 claims description 5
- 125000002950 monocyclic group Chemical group 0.000 claims description 5
- CBPGTUOXTFTWTE-UHFFFAOYSA-N 1-(1,3-benzodioxol-4-yl)-3-(2-hydroxy-4-nitrophenyl)urea Chemical compound OC1=CC([N+]([O-])=O)=CC=C1NC(=O)NC1=CC=CC2=C1OCO2 CBPGTUOXTFTWTE-UHFFFAOYSA-N 0.000 claims description 4
- GZOMTDYQJHIAGF-UHFFFAOYSA-N 1-(2,3-dichlorophenyl)-3-(2-hydroxy-5-nitrophenyl)urea Chemical compound OC1=CC=C([N+]([O-])=O)C=C1NC(=O)NC1=CC=CC(Cl)=C1Cl GZOMTDYQJHIAGF-UHFFFAOYSA-N 0.000 claims description 4
- JYYRLQNSIZPUPF-UHFFFAOYSA-N 1-(2-bromophenyl)-3-(2-hydroxy-4-nitrophenyl)thiourea Chemical compound OC1=CC([N+]([O-])=O)=CC=C1NC(=S)NC1=CC=CC=C1Br JYYRLQNSIZPUPF-UHFFFAOYSA-N 0.000 claims description 4
- MPBMQOWOKKDSFC-UHFFFAOYSA-N 1-(2-bromophenyl)-3-(3-chloro-2-hydroxyphenyl)urea Chemical compound OC1=C(Cl)C=CC=C1NC(=O)NC1=CC=CC=C1Br MPBMQOWOKKDSFC-UHFFFAOYSA-N 0.000 claims description 4
- XJGIXEVHERZBQV-UHFFFAOYSA-N 1-(2-bromophenyl)-3-(3-cyano-2-hydroxyphenyl)urea Chemical compound C1=CC=C(C#N)C(O)=C1NC(=O)NC1=CC=CC=C1Br XJGIXEVHERZBQV-UHFFFAOYSA-N 0.000 claims description 4
- VIKJECVSALYBJB-UHFFFAOYSA-N 1-(2-bromophenyl)-3-(4-cyano-2-hydroxyphenyl)urea Chemical compound OC1=CC(C#N)=CC=C1NC(=O)NC1=CC=CC=C1Br VIKJECVSALYBJB-UHFFFAOYSA-N 0.000 claims description 4
- UZWXETXUKFOJFK-UHFFFAOYSA-N 1-(2-hydroxy-4-nitrophenyl)-3-(2-methylsulfanylphenyl)urea Chemical compound CSC1=CC=CC=C1NC(=O)NC1=CC=C([N+]([O-])=O)C=C1O UZWXETXUKFOJFK-UHFFFAOYSA-N 0.000 claims description 4
- MPSVIJAQCVNHHQ-UHFFFAOYSA-N 1-(2-hydroxy-4-nitrophenyl)-3-(2-phenylphenyl)urea Chemical compound OC1=CC([N+]([O-])=O)=CC=C1NC(=O)NC1=CC=CC=C1C1=CC=CC=C1 MPSVIJAQCVNHHQ-UHFFFAOYSA-N 0.000 claims description 4
- WJGRUAQVAFBYOC-UHFFFAOYSA-N 1-(3,4-dichloro-2-hydroxyphenyl)-3-(2,3-dichlorophenyl)urea Chemical compound OC1=C(Cl)C(Cl)=CC=C1NC(=O)NC1=CC=CC(Cl)=C1Cl WJGRUAQVAFBYOC-UHFFFAOYSA-N 0.000 claims description 4
- PXEWTGQJHBTLOG-UHFFFAOYSA-N 1-(3,4-dichloro-2-hydroxyphenyl)-3-(2-methoxyphenyl)urea Chemical compound COC1=CC=CC=C1NC(=O)NC1=CC=C(Cl)C(Cl)=C1O PXEWTGQJHBTLOG-UHFFFAOYSA-N 0.000 claims description 4
- UROHGCKFAXVYKN-UHFFFAOYSA-N 1-(3,4-dichloro-2-hydroxyphenyl)-3-(2-phenylphenyl)urea Chemical compound OC1=C(Cl)C(Cl)=CC=C1NC(=O)NC1=CC=CC=C1C1=CC=CC=C1 UROHGCKFAXVYKN-UHFFFAOYSA-N 0.000 claims description 4
- IAIFWAOFGOIHNN-UHFFFAOYSA-N 1-(3-cyano-2-hydroxyphenyl)-3-(2,3-dichlorophenyl)urea Chemical compound C1=CC=C(C#N)C(O)=C1NC(=O)NC1=CC=CC(Cl)=C1Cl IAIFWAOFGOIHNN-UHFFFAOYSA-N 0.000 claims description 4
- WXLBSLQDYGMCGF-UHFFFAOYSA-N 1-(4-cyano-2-hydroxyphenyl)-3-(2-methylphenyl)urea Chemical compound CC1=CC=CC=C1NC(=O)NC1=CC=C(C#N)C=C1O WXLBSLQDYGMCGF-UHFFFAOYSA-N 0.000 claims description 4
- YZAZGBOPFBFDAC-UHFFFAOYSA-N 1-(4-cyano-2-hydroxyphenyl)-3-(2-phenylphenyl)urea Chemical compound OC1=CC(C#N)=CC=C1NC(=O)NC1=CC=CC=C1C1=CC=CC=C1 YZAZGBOPFBFDAC-UHFFFAOYSA-N 0.000 claims description 4
- PSODDICKDGEFJX-UHFFFAOYSA-N 1-(4-cyano-2-hydroxyphenyl)-3-[2-(trifluoromethyl)phenyl]urea Chemical compound OC1=CC(C#N)=CC=C1NC(=O)NC1=CC=CC=C1C(F)(F)F PSODDICKDGEFJX-UHFFFAOYSA-N 0.000 claims description 4
- 241000700605 Viruses Species 0.000 claims description 4
- 125000003831 tetrazolyl group Chemical group 0.000 claims description 4
- DSSYKIVIOFKYAU-XCBNKYQSSA-N (R)-camphor Chemical compound C1C[C@@]2(C)C(=O)C[C@@H]1C2(C)C DSSYKIVIOFKYAU-XCBNKYQSSA-N 0.000 claims description 3
- 241000723346 Cinnamomum camphora Species 0.000 claims description 3
- 230000033115 angiogenesis Effects 0.000 claims description 3
- 229960000846 camphor Drugs 0.000 claims description 3
- 229930008380 camphor Natural products 0.000 claims description 3
- BVKZGUZCCUSVTD-UHFFFAOYSA-N carbonic acid Chemical group OC(O)=O BVKZGUZCCUSVTD-UHFFFAOYSA-N 0.000 claims description 3
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 claims description 3
- 150000001732 carboxylic acid derivatives Chemical group 0.000 claims description 3
- 210000003958 hematopoietic stem cell Anatomy 0.000 claims description 3
- 125000004447 heteroarylalkenyl group Chemical group 0.000 claims description 3
- 201000004792 malaria Diseases 0.000 claims description 3
- 150000003573 thiols Chemical group 0.000 claims description 3
- PEBCQTKZVQCXJP-UHFFFAOYSA-N 1-(2-bromophenyl)-3-(3,4-dichloro-2-hydroxyphenyl)urea Chemical compound OC1=C(Cl)C(Cl)=CC=C1NC(=O)NC1=CC=CC=C1Br PEBCQTKZVQCXJP-UHFFFAOYSA-N 0.000 claims description 2
- IGNKQJCDASZUBP-UHFFFAOYSA-N 1-(2-bromophenyl)-3-[2-hydroxy-3-(trifluoromethyl)phenyl]urea Chemical compound C1=CC=C(C(F)(F)F)C(O)=C1NC(=O)NC1=CC=CC=C1Br IGNKQJCDASZUBP-UHFFFAOYSA-N 0.000 claims description 2
- 201000001320 Atherosclerosis Diseases 0.000 claims description 2
- 241000711549 Hepacivirus C Species 0.000 claims description 2
- 241000700721 Hepatitis B virus Species 0.000 claims description 2
- 208000001132 Osteoporosis Diseases 0.000 claims description 2
- 125000002102 aryl alkyloxo group Chemical group 0.000 claims description 2
- 208000007565 gingivitis Diseases 0.000 claims description 2
- 125000004438 haloalkoxy group Chemical group 0.000 claims description 2
- 125000005843 halogen group Chemical group 0.000 claims description 2
- 208000006454 hepatitis Diseases 0.000 claims description 2
- 231100000283 hepatitis Toxicity 0.000 claims description 2
- 208000002672 hepatitis B Diseases 0.000 claims description 2
- 125000002768 hydroxyalkyl group Chemical group 0.000 claims description 2
- 230000000241 respiratory effect Effects 0.000 claims description 2
- 125000005000 thioaryl group Chemical group 0.000 claims description 2
- 241001529453 unidentified herpesvirus Species 0.000 claims description 2
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 541
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 404
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 238
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 237
- 238000002360 preparation method Methods 0.000 description 235
- OKKJLVBELUTLKV-MZCSYVLQSA-N Deuterated methanol Chemical compound [2H]OC([2H])([2H])[2H] OKKJLVBELUTLKV-MZCSYVLQSA-N 0.000 description 202
- XSQUKJJJFZCRTK-UHFFFAOYSA-N Urea Chemical compound NC(N)=O XSQUKJJJFZCRTK-UHFFFAOYSA-N 0.000 description 178
- 239000000047 product Substances 0.000 description 177
- 238000007429 general method Methods 0.000 description 153
- CSNNHWWHGAXBCP-UHFFFAOYSA-L Magnesium sulfate Chemical compound [Mg+2].[O-][S+2]([O-])([O-])[O-] CSNNHWWHGAXBCP-UHFFFAOYSA-L 0.000 description 114
- 238000001556 precipitation Methods 0.000 description 113
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 112
- 238000002451 electron ionisation mass spectrometry Methods 0.000 description 109
- 238000001914 filtration Methods 0.000 description 108
- 239000007787 solid Substances 0.000 description 97
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 96
- 239000011541 reaction mixture Substances 0.000 description 94
- 235000013877 carbamide Nutrition 0.000 description 90
- 239000004202 carbamide Substances 0.000 description 89
- 238000005160 1H NMR spectroscopy Methods 0.000 description 88
- 239000000741 silica gel Substances 0.000 description 86
- 229910002027 silica gel Inorganic materials 0.000 description 86
- 235000019439 ethyl acetate Nutrition 0.000 description 82
- 230000015572 biosynthetic process Effects 0.000 description 78
- 238000003786 synthesis reaction Methods 0.000 description 78
- 239000000243 solution Substances 0.000 description 75
- 238000004587 chromatography analysis Methods 0.000 description 70
- 239000000203 mixture Substances 0.000 description 65
- 239000002904 solvent Substances 0.000 description 63
- ZGEGCLOFRBLKSE-UHFFFAOYSA-N methylene hexane Natural products CCCCCC=C ZGEGCLOFRBLKSE-UHFFFAOYSA-N 0.000 description 62
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 62
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 61
- XKRFYHLGVUSROY-UHFFFAOYSA-N Argon Chemical compound [Ar] XKRFYHLGVUSROY-UHFFFAOYSA-N 0.000 description 60
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 60
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 60
- 102000004890 Interleukin-8 Human genes 0.000 description 59
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 59
- XKTZWUACRZHVAN-VADRZIEHSA-N interleukin-8 Chemical compound C([C@H](NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CC=1C2=CC=CC=C2NC=1)NC(=O)[C@@H](NC(C)=O)CCSC)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC=1C=CC=CC=1)C(=O)N[C@@H]([C@@H](C)O)C(=O)NCC(=O)N[C@@H](CCSC)C(=O)N1[C@H](CCC1)C(=O)N1[C@H](CCC1)C(=O)N[C@@H](C)C(=O)N[C@H](CC(O)=O)C(=O)N[C@H](CCC(O)=O)C(=O)N[C@H](CC(O)=O)C(=O)N[C@H](CC=1C=CC(O)=CC=1)C(=O)N[C@H](CO)C(=O)N1[C@H](CCC1)C(N)=O)C1=CC=CC=C1 XKTZWUACRZHVAN-VADRZIEHSA-N 0.000 description 57
- 229940096397 interleukin-8 Drugs 0.000 description 57
- 229910052943 magnesium sulfate Inorganic materials 0.000 description 57
- PAYRUJLWNCNPSJ-UHFFFAOYSA-N N-phenyl amine Natural products NC1=CC=CC=C1 PAYRUJLWNCNPSJ-UHFFFAOYSA-N 0.000 description 55
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 52
- 238000005481 NMR spectroscopy Methods 0.000 description 45
- 238000010790 dilution Methods 0.000 description 45
- 239000012895 dilution Substances 0.000 description 45
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 41
- VWDWKYIASSYTQR-UHFFFAOYSA-N sodium nitrate Chemical compound [Na+].[O-][N+]([O-])=O VWDWKYIASSYTQR-UHFFFAOYSA-N 0.000 description 40
- LPXPTNMVRIOKMN-UHFFFAOYSA-M sodium nitrite Chemical compound [Na+].[O-]N=O LPXPTNMVRIOKMN-UHFFFAOYSA-M 0.000 description 38
- DOPJTDJKZNWLRB-UHFFFAOYSA-N 2-Amino-5-nitrophenol Chemical compound NC1=CC=C([N+]([O-])=O)C=C1O DOPJTDJKZNWLRB-UHFFFAOYSA-N 0.000 description 33
- 238000003756 stirring Methods 0.000 description 31
- QXEDXIJDCOADGG-UHFFFAOYSA-N (2-bromophenyl)urea Chemical compound NC(=O)NC1=CC=CC=C1Br QXEDXIJDCOADGG-UHFFFAOYSA-N 0.000 description 30
- 229910052786 argon Inorganic materials 0.000 description 30
- 239000012044 organic layer Substances 0.000 description 30
- 239000012074 organic phase Substances 0.000 description 30
- 239000007858 starting material Substances 0.000 description 26
- GOOVAYJIVMBWPP-UHFFFAOYSA-N 1-bromo-2-isocyanatobenzene Chemical compound BrC1=CC=CC=C1N=C=O GOOVAYJIVMBWPP-UHFFFAOYSA-N 0.000 description 23
- 239000012267 brine Substances 0.000 description 23
- 230000003197 catalytic effect Effects 0.000 description 23
- HPALAKNZSZLMCH-UHFFFAOYSA-M sodium;chloride;hydrate Chemical compound O.[Na+].[Cl-] HPALAKNZSZLMCH-UHFFFAOYSA-M 0.000 description 23
- 102000016950 Chemokine CXCL1 Human genes 0.000 description 22
- 229910021626 Tin(II) chloride Inorganic materials 0.000 description 22
- 230000002829 reductive effect Effects 0.000 description 22
- 125000006296 sulfonyl amino group Chemical group [H]N(*)S(*)(=O)=O 0.000 description 22
- AXZWODMDQAVCJE-UHFFFAOYSA-L tin(II) chloride (anhydrous) Chemical compound [Cl-].[Cl-].[Sn+2] AXZWODMDQAVCJE-UHFFFAOYSA-L 0.000 description 22
- 235000011150 stannous chloride Nutrition 0.000 description 21
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 20
- KDLHZDBZIXYQEI-UHFFFAOYSA-N palladium Substances [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 description 20
- 239000004317 sodium nitrate Substances 0.000 description 20
- 235000010344 sodium nitrate Nutrition 0.000 description 20
- 102100039398 C-X-C motif chemokine 2 Human genes 0.000 description 19
- 108010014419 Chemokine CXCL1 Proteins 0.000 description 19
- 101000973997 Homo sapiens Nucleosome assembly protein 1-like 4 Proteins 0.000 description 19
- 101000947178 Homo sapiens Platelet basic protein Proteins 0.000 description 19
- 102100036154 Platelet basic protein Human genes 0.000 description 19
- 235000010288 sodium nitrite Nutrition 0.000 description 19
- 101000889128 Homo sapiens C-X-C motif chemokine 2 Proteins 0.000 description 18
- 238000003818 flash chromatography Methods 0.000 description 18
- 229940086542 triethylamine Drugs 0.000 description 18
- 238000006243 chemical reaction Methods 0.000 description 16
- 239000012948 isocyanate Substances 0.000 description 16
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical compound [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 14
- 102000005962 receptors Human genes 0.000 description 14
- 108020003175 receptors Proteins 0.000 description 14
- GEYOCULIXLDCMW-UHFFFAOYSA-N 1,2-phenylenediamine Chemical compound NC1=CC=CC=C1N GEYOCULIXLDCMW-UHFFFAOYSA-N 0.000 description 13
- 150000002513 isocyanates Chemical class 0.000 description 13
- 210000000440 neutrophil Anatomy 0.000 description 13
- HKZPOGZBKIWMPO-UHFFFAOYSA-N 4-amino-3-hydroxybenzonitrile Chemical compound NC1=CC=C(C#N)C=C1O HKZPOGZBKIWMPO-UHFFFAOYSA-N 0.000 description 12
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 12
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 12
- 125000001424 substituent group Chemical group 0.000 description 12
- UMGDCJDMYOKAJW-UHFFFAOYSA-N thiourea Chemical compound NC(N)=S UMGDCJDMYOKAJW-UHFFFAOYSA-N 0.000 description 12
- 239000000284 extract Substances 0.000 description 11
- 229940124530 sulfonamide Drugs 0.000 description 11
- DGTNSSLYPYDJGL-UHFFFAOYSA-N phenyl isocyanate Chemical compound O=C=NC1=CC=CC=C1 DGTNSSLYPYDJGL-UHFFFAOYSA-N 0.000 description 10
- 150000003456 sulfonamides Chemical class 0.000 description 10
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 9
- JFDZBHWFFUWGJE-UHFFFAOYSA-N benzonitrile Chemical compound N#CC1=CC=CC=C1 JFDZBHWFFUWGJE-UHFFFAOYSA-N 0.000 description 9
- 239000010410 layer Substances 0.000 description 9
- 238000001953 recrystallisation Methods 0.000 description 9
- PXIPVTKHYLBLMZ-UHFFFAOYSA-N Sodium azide Chemical compound [Na+].[N-]=[N+]=[N-] PXIPVTKHYLBLMZ-UHFFFAOYSA-N 0.000 description 8
- 239000012298 atmosphere Substances 0.000 description 8
- 229910052799 carbon Inorganic materials 0.000 description 8
- 235000019341 magnesium sulphate Nutrition 0.000 description 8
- 125000006276 2-bromophenyl group Chemical group [H]C1=C([H])C(Br)=C(*)C([H])=C1[H] 0.000 description 7
- LBKNLCXXJZQFHU-UHFFFAOYSA-N 6-amino-2,3-dichlorophenol Chemical compound NC1=CC=C(Cl)C(Cl)=C1O LBKNLCXXJZQFHU-UHFFFAOYSA-N 0.000 description 7
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 7
- 239000000706 filtrate Substances 0.000 description 7
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 7
- 238000010992 reflux Methods 0.000 description 7
- VLZVIIYRNMWPSN-UHFFFAOYSA-N 2-Amino-4-nitrophenol Chemical compound NC1=CC([N+]([O-])=O)=CC=C1O VLZVIIYRNMWPSN-UHFFFAOYSA-N 0.000 description 6
- FMQVUWGTHZPXQU-UHFFFAOYSA-N 3-amino-2-hydroxybenzonitrile Chemical compound NC1=CC=CC(C#N)=C1O FMQVUWGTHZPXQU-UHFFFAOYSA-N 0.000 description 6
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical compound [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 6
- 206010040070 Septic Shock Diseases 0.000 description 6
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 6
- 229960000583 acetic acid Drugs 0.000 description 6
- 235000011054 acetic acid Nutrition 0.000 description 6
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 description 6
- 229910052794 bromium Inorganic materials 0.000 description 6
- 210000004027 cell Anatomy 0.000 description 6
- RAXXELZNTBOGNW-UHFFFAOYSA-N imidazole Natural products C1=CNC=N1 RAXXELZNTBOGNW-UHFFFAOYSA-N 0.000 description 6
- 238000004519 manufacturing process Methods 0.000 description 6
- 239000008194 pharmaceutical composition Substances 0.000 description 6
- 229910000030 sodium bicarbonate Inorganic materials 0.000 description 6
- 229910052938 sodium sulfate Inorganic materials 0.000 description 6
- 235000011152 sodium sulphate Nutrition 0.000 description 6
- MHNCVYSYIFOKDX-UHFFFAOYSA-N 1-(2,3-dichlorophenyl)-3-(2-hydroxy-3-phenylphenyl)urea Chemical compound C1=CC=C(C=2C=CC=CC=2)C(O)=C1NC(=O)NC1=CC=CC(Cl)=C1Cl MHNCVYSYIFOKDX-UHFFFAOYSA-N 0.000 description 5
- ZZQDGDJCUKXXBC-UHFFFAOYSA-N 1-(2-hydroxy-4-propan-2-ylphenyl)-3-[3-(trifluoromethyl)phenyl]urea Chemical compound OC1=CC(C(C)C)=CC=C1NC(=O)NC1=CC=CC(C(F)(F)F)=C1 ZZQDGDJCUKXXBC-UHFFFAOYSA-N 0.000 description 5
- PQAXIHNOJWBTGN-UHFFFAOYSA-N 1-(2-hydroxy-5-nitrophenyl)-3-(3-methoxythiophen-2-yl)urea Chemical compound C1=CSC(NC(=O)NC=2C(=CC=C(C=2)[N+]([O-])=O)O)=C1OC PQAXIHNOJWBTGN-UHFFFAOYSA-N 0.000 description 5
- ZYQPVOZZWSDVQB-UHFFFAOYSA-N 1-[2-hydroxy-4-(trifluoromethyl)phenyl]-3-phenylurea Chemical compound OC1=CC(C(F)(F)F)=CC=C1NC(=O)NC1=CC=CC=C1 ZYQPVOZZWSDVQB-UHFFFAOYSA-N 0.000 description 5
- IIDUNAVOCYMUFB-UHFFFAOYSA-N 2-amino-5-fluorophenol Chemical compound NC1=CC=C(F)C=C1O IIDUNAVOCYMUFB-UHFFFAOYSA-N 0.000 description 5
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 5
- 239000002585 base Substances 0.000 description 5
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 description 5
- DOBRDRYODQBAMW-UHFFFAOYSA-N copper(i) cyanide Chemical compound [Cu+].N#[C-] DOBRDRYODQBAMW-UHFFFAOYSA-N 0.000 description 5
- 239000003085 diluting agent Substances 0.000 description 5
- 230000002401 inhibitory effect Effects 0.000 description 5
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 5
- 239000003921 oil Substances 0.000 description 5
- 235000019198 oils Nutrition 0.000 description 5
- IDEIMXJJLMVTNN-UHFFFAOYSA-N 1,3-bis(2-hydroxy-4-nitrophenyl)urea Chemical compound OC1=CC([N+]([O-])=O)=CC=C1NC(=O)NC1=CC=C([N+]([O-])=O)C=C1O IDEIMXJJLMVTNN-UHFFFAOYSA-N 0.000 description 4
- YPUIHNGFAQOMLY-UHFFFAOYSA-N 1-(2-bromophenyl)-3-(2-hydroxy-3-phenylmethoxyphenyl)urea Chemical compound C1=CC=C(OCC=2C=CC=CC=2)C(O)=C1NC(=O)NC1=CC=CC=C1Br YPUIHNGFAQOMLY-UHFFFAOYSA-N 0.000 description 4
- SWZSLTLBOSGOGA-UHFFFAOYSA-N 1-(2-bromophenyl)-3-(2-hydroxy-4-phenylphenyl)urea Chemical compound OC1=CC(C=2C=CC=CC=2)=CC=C1NC(=O)NC1=CC=CC=C1Br SWZSLTLBOSGOGA-UHFFFAOYSA-N 0.000 description 4
- IFOKUNVJYFFYQT-UHFFFAOYSA-N 1-(2-bromophenyl)-3-(3,4-difluoro-2-hydroxyphenyl)urea Chemical compound OC1=C(F)C(F)=CC=C1NC(=O)NC1=CC=CC=C1Br IFOKUNVJYFFYQT-UHFFFAOYSA-N 0.000 description 4
- DHXOJNFEHNIGMB-UHFFFAOYSA-N 1-(2-bromophenyl)-3-(4-fluoro-2-hydroxyphenyl)urea Chemical compound OC1=CC(F)=CC=C1NC(=O)NC1=CC=CC=C1Br DHXOJNFEHNIGMB-UHFFFAOYSA-N 0.000 description 4
- MZFHJQMUHUVVLD-UHFFFAOYSA-N 1-(2-bromophenyl)-3-[2-hydroxy-5-(trifluoromethyl)phenyl]urea Chemical compound OC1=CC=C(C(F)(F)F)C=C1NC(=O)NC1=CC=CC=C1Br MZFHJQMUHUVVLD-UHFFFAOYSA-N 0.000 description 4
- QOMIBRVYULIIPL-UHFFFAOYSA-N 1-(4-azido-2-hydroxyphenyl)-3-(2-iodophenyl)urea Chemical compound OC1=CC(N=[N+]=[N-])=CC=C1NC(=O)NC1=CC=CC=C1I QOMIBRVYULIIPL-UHFFFAOYSA-N 0.000 description 4
- UFYKIHANBCVMKW-UHFFFAOYSA-N 1-(4-azido-2-hydroxyphenyl)-3-(2-methoxyphenyl)urea Chemical compound COC1=CC=CC=C1NC(=O)NC1=CC=C(N=[N+]=[N-])C=C1O UFYKIHANBCVMKW-UHFFFAOYSA-N 0.000 description 4
- CENOMWDZGNQIGT-UHFFFAOYSA-N 1-(5-bromo-3-fluoro-2-hydroxyphenyl)-3-(2-bromophenyl)urea Chemical compound OC1=C(F)C=C(Br)C=C1NC(=O)NC1=CC=CC=C1Br CENOMWDZGNQIGT-UHFFFAOYSA-N 0.000 description 4
- PICNBCJMPWKROU-UHFFFAOYSA-N 1-[2-(benzenesulfonamido)-4-nitrophenyl]-3-phenylurea Chemical compound C=1C=CC=CC=1S(=O)(=O)NC1=CC([N+](=O)[O-])=CC=C1NC(=O)NC1=CC=CC=C1 PICNBCJMPWKROU-UHFFFAOYSA-N 0.000 description 4
- WAFUAYGNTDBHDS-UHFFFAOYSA-N 2-[tert-butyl(dimethyl)silyl]oxy-4-nitroaniline Chemical compound CC(C)(C)[Si](C)(C)OC1=CC([N+]([O-])=O)=CC=C1N WAFUAYGNTDBHDS-UHFFFAOYSA-N 0.000 description 4
- IFCGVNYNIRTWNU-UHFFFAOYSA-N 2-amino-5-propan-2-ylphenol Chemical compound CC(C)C1=CC=C(N)C(O)=C1 IFCGVNYNIRTWNU-UHFFFAOYSA-N 0.000 description 4
- MPLDLWHRQIVKLC-UHFFFAOYSA-N 2-bromo-3-methyl-6-nitrophenol Chemical compound CC1=CC=C([N+]([O-])=O)C(O)=C1Br MPLDLWHRQIVKLC-UHFFFAOYSA-N 0.000 description 4
- CURLTUGMZLYLDI-UHFFFAOYSA-N Carbon dioxide Chemical compound O=C=O CURLTUGMZLYLDI-UHFFFAOYSA-N 0.000 description 4
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 4
- 102000000589 Interleukin-1 Human genes 0.000 description 4
- 108010002352 Interleukin-1 Proteins 0.000 description 4
- MZRVEZGGRBJDDB-UHFFFAOYSA-N N-Butyllithium Chemical compound [Li]CCCC MZRVEZGGRBJDDB-UHFFFAOYSA-N 0.000 description 4
- GRYLNZFGIOXLOG-UHFFFAOYSA-N Nitric acid Chemical compound O[N+]([O-])=O GRYLNZFGIOXLOG-UHFFFAOYSA-N 0.000 description 4
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical compound C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 4
- YTPLMLYBLZKORZ-UHFFFAOYSA-N Thiophene Chemical compound C=1C=CSC=1 YTPLMLYBLZKORZ-UHFFFAOYSA-N 0.000 description 4
- HEDRZPFGACZZDS-MICDWDOJSA-N Trichloro(2H)methane Chemical compound [2H]C(Cl)(Cl)Cl HEDRZPFGACZZDS-MICDWDOJSA-N 0.000 description 4
- 102100040247 Tumor necrosis factor Human genes 0.000 description 4
- 230000004913 activation Effects 0.000 description 4
- 150000001448 anilines Chemical class 0.000 description 4
- 125000000852 azido group Chemical group *N=[N+]=[N-] 0.000 description 4
- 230000035605 chemotaxis Effects 0.000 description 4
- 235000015165 citric acid Nutrition 0.000 description 4
- 238000004440 column chromatography Methods 0.000 description 4
- 125000000524 functional group Chemical group 0.000 description 4
- 239000007789 gas Substances 0.000 description 4
- 230000008595 infiltration Effects 0.000 description 4
- 238000001764 infiltration Methods 0.000 description 4
- 239000003112 inhibitor Substances 0.000 description 4
- DDUNMNVFNVRLFB-VQHVLOKHSA-N methyl (e)-3-[4-[(2-bromophenyl)carbamoylamino]-3-hydroxyphenyl]prop-2-enoate Chemical compound OC1=CC(/C=C/C(=O)OC)=CC=C1NC(=O)NC1=CC=CC=C1Br DDUNMNVFNVRLFB-VQHVLOKHSA-N 0.000 description 4
- 229910017604 nitric acid Inorganic materials 0.000 description 4
- 238000000746 purification Methods 0.000 description 4
- 239000000126 substance Substances 0.000 description 4
- 238000006467 substitution reaction Methods 0.000 description 4
- 125000005420 sulfonamido group Chemical group S(=O)(=O)(N*)* 0.000 description 4
- MGOABCWEOZZCDP-UHFFFAOYSA-N 1-(2,4-dibromophenyl)-3-(2-hydroxy-4-nitrophenyl)urea Chemical compound OC1=CC([N+]([O-])=O)=CC=C1NC(=O)NC1=CC=C(Br)C=C1Br MGOABCWEOZZCDP-UHFFFAOYSA-N 0.000 description 3
- CWCZXUGASXHLMK-UHFFFAOYSA-N 1-(2,4-dichlorophenyl)-3-(2-hydroxy-4-nitrophenyl)urea Chemical compound OC1=CC([N+]([O-])=O)=CC=C1NC(=O)NC1=CC=C(Cl)C=C1Cl CWCZXUGASXHLMK-UHFFFAOYSA-N 0.000 description 3
- MIUZBQLHNFEROV-UHFFFAOYSA-N 1-(2,6-difluorophenyl)-3-(2-hydroxy-4-nitrophenyl)urea Chemical compound OC1=CC([N+]([O-])=O)=CC=C1NC(=O)NC1=C(F)C=CC=C1F MIUZBQLHNFEROV-UHFFFAOYSA-N 0.000 description 3
- BWFIEDHJOPRIMJ-UHFFFAOYSA-N 1-(2-bromophenyl)-3-(2-hydroxy-3,4-diphenylphenyl)urea Chemical compound C1=CC(C=2C=CC=CC=2)=C(C=2C=CC=CC=2)C(O)=C1NC(=O)NC1=CC=CC=C1Br BWFIEDHJOPRIMJ-UHFFFAOYSA-N 0.000 description 3
- OQVARPSTLHEQDV-UHFFFAOYSA-N 1-(2-bromophenyl)-3-(2-hydroxy-3-iodophenyl)urea Chemical compound OC1=C(I)C=CC=C1NC(=O)NC1=CC=CC=C1Br OQVARPSTLHEQDV-UHFFFAOYSA-N 0.000 description 3
- QWLSUPKOFQHSRW-UHFFFAOYSA-N 1-(2-bromophenyl)-3-(4-bromo-2-sulfanylphenyl)urea Chemical compound SC1=CC(Br)=CC=C1NC(=O)NC1=CC=CC=C1Br QWLSUPKOFQHSRW-UHFFFAOYSA-N 0.000 description 3
- JGHIGXYVAWUVLN-UHFFFAOYSA-N 1-(2-bromophenyl)-3-[2-(2-phenylethylsulfonylamino)phenyl]urea Chemical compound BrC1=CC=CC=C1NC(=O)NC1=CC=CC=C1NS(=O)(=O)CCC1=CC=CC=C1 JGHIGXYVAWUVLN-UHFFFAOYSA-N 0.000 description 3
- YWKGOEOCYBFILN-UHFFFAOYSA-N 1-(2-bromophenyl)-3-[2-[[2-(trifluoromethyl)phenyl]sulfonylamino]phenyl]urea Chemical compound FC(F)(F)C1=CC=CC=C1S(=O)(=O)NC1=CC=CC=C1NC(=O)NC1=CC=CC=C1Br YWKGOEOCYBFILN-UHFFFAOYSA-N 0.000 description 3
- JGLOHKYHPQNEJY-UHFFFAOYSA-N 1-(2-bromophenyl)-3-[2-hydroxy-4-(trifluoromethyl)phenyl]urea Chemical compound OC1=CC(C(F)(F)F)=CC=C1NC(=O)NC1=CC=CC=C1Br JGLOHKYHPQNEJY-UHFFFAOYSA-N 0.000 description 3
- MWODCLJTSRCRGB-UHFFFAOYSA-N 1-(2-bromophenyl)-3-[3-fluoro-2-hydroxy-4-(trifluoromethyl)phenyl]urea Chemical compound OC1=C(F)C(C(F)(F)F)=CC=C1NC(=O)NC1=CC=CC=C1Br MWODCLJTSRCRGB-UHFFFAOYSA-N 0.000 description 3
- TVHIIIDDOHJFQT-UHFFFAOYSA-N 1-(2-hydroxy-4-nitrophenyl)-3-(2-nitrophenyl)urea Chemical compound OC1=CC([N+]([O-])=O)=CC=C1NC(=O)NC1=CC=CC=C1[N+]([O-])=O TVHIIIDDOHJFQT-UHFFFAOYSA-N 0.000 description 3
- QZOTURNBPCCOPX-UHFFFAOYSA-N 1-(2-hydroxy-4-nitrophenyl)-3-(3-methoxyphenyl)urea Chemical compound COC1=CC=CC(NC(=O)NC=2C(=CC(=CC=2)[N+]([O-])=O)O)=C1 QZOTURNBPCCOPX-UHFFFAOYSA-N 0.000 description 3
- USGOTVZCMDLNGF-UHFFFAOYSA-N 1-(2-hydroxy-4-nitrophenyl)-3-(3-methoxythiophen-2-yl)urea Chemical compound C1=CSC(NC(=O)NC=2C(=CC(=CC=2)[N+]([O-])=O)O)=C1OC USGOTVZCMDLNGF-UHFFFAOYSA-N 0.000 description 3
- ARLIDMNFLJKZDA-UHFFFAOYSA-N 1-(2-hydroxy-4-nitrophenyl)-3-[2-(trifluoromethyl)phenyl]urea Chemical compound OC1=CC([N+]([O-])=O)=CC=C1NC(=O)NC1=CC=CC=C1C(F)(F)F ARLIDMNFLJKZDA-UHFFFAOYSA-N 0.000 description 3
- SBPFCKQEIYYHBN-UHFFFAOYSA-N 1-(2-hydroxy-4-nitrophenyl)-3-naphthalen-1-ylurea Chemical compound OC1=CC([N+]([O-])=O)=CC=C1NC(=O)NC1=CC=CC2=CC=CC=C12 SBPFCKQEIYYHBN-UHFFFAOYSA-N 0.000 description 3
- MHNNUSRLYWTBJF-UHFFFAOYSA-N 1-(2-hydroxy-5-nitrophenyl)-3-phenylurea Chemical compound OC1=CC=C([N+]([O-])=O)C=C1NC(=O)NC1=CC=CC=C1 MHNNUSRLYWTBJF-UHFFFAOYSA-N 0.000 description 3
- LYGPWVRHLXLGNP-UHFFFAOYSA-N 1-(3-bromophenyl)-3-(2-hydroxy-4-nitrophenyl)urea Chemical compound OC1=CC([N+]([O-])=O)=CC=C1NC(=O)NC1=CC=CC(Br)=C1 LYGPWVRHLXLGNP-UHFFFAOYSA-N 0.000 description 3
- FLLYDYSIHJCMKD-UHFFFAOYSA-N 1-[2-[[3,5-bis(trifluoromethyl)phenyl]sulfonylamino]phenyl]-3-(2-bromophenyl)urea Chemical compound FC(F)(F)C1=CC(C(F)(F)F)=CC(S(=O)(=O)NC=2C(=CC=CC=2)NC(=O)NC=2C(=CC=CC=2)Br)=C1 FLLYDYSIHJCMKD-UHFFFAOYSA-N 0.000 description 3
- DVAWJTBZVQVMLL-UHFFFAOYSA-N 1-[2-[tert-butyl(dimethyl)silyl]oxy-4-nitrophenyl]-3-phenylthiourea Chemical compound CC(C)(C)[Si](C)(C)OC1=CC([N+]([O-])=O)=CC=C1NC(=S)NC1=CC=CC=C1 DVAWJTBZVQVMLL-UHFFFAOYSA-N 0.000 description 3
- SWIRMXTXRCDCJK-UHFFFAOYSA-N 1-[2-hydroxy-4-(trifluoromethyl)phenyl]-3-(2-phenylphenyl)urea Chemical compound OC1=CC(C(F)(F)F)=CC=C1NC(=O)NC1=CC=CC=C1C1=CC=CC=C1 SWIRMXTXRCDCJK-UHFFFAOYSA-N 0.000 description 3
- IHHUGFJSEJSCGE-UHFFFAOYSA-N 1-isocyanato-2-phenylbenzene Chemical compound O=C=NC1=CC=CC=C1C1=CC=CC=C1 IHHUGFJSEJSCGE-UHFFFAOYSA-N 0.000 description 3
- ILZRAAUVZAXXKZ-UHFFFAOYSA-N 2-amino-5-(trifluoromethyl)phenol Chemical compound NC1=CC=C(C(F)(F)F)C=C1O ILZRAAUVZAXXKZ-UHFFFAOYSA-N 0.000 description 3
- UDVRKKAWBVVSAM-UHFFFAOYSA-N 2-amino-6-phenylphenol Chemical compound NC1=CC=CC(C=2C=CC=CC=2)=C1O UDVRKKAWBVVSAM-UHFFFAOYSA-N 0.000 description 3
- VRVRGVPWCUEOGV-UHFFFAOYSA-N 2-aminothiophenol Chemical compound NC1=CC=CC=C1S VRVRGVPWCUEOGV-UHFFFAOYSA-N 0.000 description 3
- KKEQNHBOGAURSN-UHFFFAOYSA-N 2-hydroxy-1,2,3,4-tetrahydroinden-5-one Chemical compound C1=CC(=O)CC2=C1CC(O)C2 KKEQNHBOGAURSN-UHFFFAOYSA-N 0.000 description 3
- CHZCERSEMVWNHL-UHFFFAOYSA-N 2-hydroxybenzonitrile Chemical group OC1=CC=CC=C1C#N CHZCERSEMVWNHL-UHFFFAOYSA-N 0.000 description 3
- LSSMRBBQCHSDNS-UHFFFAOYSA-N 3-methoxythiophene-2-carboxylic acid Chemical compound COC=1C=CSC=1C(O)=O LSSMRBBQCHSDNS-UHFFFAOYSA-N 0.000 description 3
- QQURWFRNETXFTN-UHFFFAOYSA-N 5-fluoro-2-nitrophenol Chemical compound OC1=CC(F)=CC=C1[N+]([O-])=O QQURWFRNETXFTN-UHFFFAOYSA-N 0.000 description 3
- WFDIJRYMOXRFFG-UHFFFAOYSA-N Acetic anhydride Chemical compound CC(=O)OC(C)=O WFDIJRYMOXRFFG-UHFFFAOYSA-N 0.000 description 3
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 3
- 101001027327 Bos taurus Growth-regulated protein homolog alpha Proteins 0.000 description 3
- 101000611183 Homo sapiens Tumor necrosis factor Proteins 0.000 description 3
- 102000004889 Interleukin-6 Human genes 0.000 description 3
- 108090001005 Interleukin-6 Proteins 0.000 description 3
- WMFOQBRAJBCJND-UHFFFAOYSA-M Lithium hydroxide Chemical compound [Li+].[OH-] WMFOQBRAJBCJND-UHFFFAOYSA-M 0.000 description 3
- MUBZPKHOEPUJKR-UHFFFAOYSA-N Oxalic acid Chemical compound OC(=O)C(O)=O MUBZPKHOEPUJKR-UHFFFAOYSA-N 0.000 description 3
- 201000004681 Psoriasis Diseases 0.000 description 3
- RWRDLPDLKQPQOW-UHFFFAOYSA-N Pyrrolidine Chemical compound C1CCNC1 RWRDLPDLKQPQOW-UHFFFAOYSA-N 0.000 description 3
- 210000001744 T-lymphocyte Anatomy 0.000 description 3
- SORGEQQSQGNZFI-UHFFFAOYSA-N [azido(phenoxy)phosphoryl]oxybenzene Chemical compound C=1C=CC=CC=1OP(=O)(N=[N+]=[N-])OC1=CC=CC=C1 SORGEQQSQGNZFI-UHFFFAOYSA-N 0.000 description 3
- 239000002253 acid Substances 0.000 description 3
- 239000000010 aprotic solvent Substances 0.000 description 3
- 210000003651 basophil Anatomy 0.000 description 3
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid Chemical compound OC(=O)C1=CC=CC=C1 WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 description 3
- 229940061631 citric acid acetate Drugs 0.000 description 3
- MKRTXPORKIRPDG-UHFFFAOYSA-N diphenylphosphoryl azide Chemical compound C=1C=CC=CC=1P(=O)(N=[N+]=[N-])C1=CC=CC=C1 MKRTXPORKIRPDG-UHFFFAOYSA-N 0.000 description 3
- 210000002889 endothelial cell Anatomy 0.000 description 3
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 3
- 125000005343 heterocyclic alkyl group Chemical group 0.000 description 3
- 238000000338 in vitro Methods 0.000 description 3
- 230000002757 inflammatory effect Effects 0.000 description 3
- 229940100601 interleukin-6 Drugs 0.000 description 3
- 239000000543 intermediate Substances 0.000 description 3
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 description 3
- MOMPWLMXIZNJGK-UHFFFAOYSA-N methyl 3-hydroxy-4-(phenylcarbamoylamino)benzoate Chemical compound OC1=CC(C(=O)OC)=CC=C1NC(=O)NC1=CC=CC=C1 MOMPWLMXIZNJGK-UHFFFAOYSA-N 0.000 description 3
- HVSWRCHXFZIJMA-UHFFFAOYSA-N methyl 3-hydroxy-4-[(2-phenylphenyl)carbamoylamino]benzoate Chemical compound OC1=CC(C(=O)OC)=CC=C1NC(=O)NC1=CC=CC=C1C1=CC=CC=C1 HVSWRCHXFZIJMA-UHFFFAOYSA-N 0.000 description 3
- OCZXDVNSNDITBS-UHFFFAOYSA-N methyl 4-amino-3-hydroxybenzoate Chemical compound COC(=O)C1=CC=C(N)C(O)=C1 OCZXDVNSNDITBS-UHFFFAOYSA-N 0.000 description 3
- PSHKMPUSSFXUIA-UHFFFAOYSA-N n,n-dimethylpyridin-2-amine Chemical compound CN(C)C1=CC=CC=N1 PSHKMPUSSFXUIA-UHFFFAOYSA-N 0.000 description 3
- OTFMOOMWUNLLKQ-UHFFFAOYSA-N n-[4-[[2-[(2-bromophenyl)carbamoylamino]phenyl]sulfamoyl]phenyl]acetamide Chemical compound C1=CC(NC(=O)C)=CC=C1S(=O)(=O)NC1=CC=CC=C1NC(=O)NC1=CC=CC=C1Br OTFMOOMWUNLLKQ-UHFFFAOYSA-N 0.000 description 3
- NHDUIVOKCKQWEF-UHFFFAOYSA-N n-benzyl-4-[(2-bromophenyl)carbamoylamino]-3-hydroxybenzamide Chemical compound OC1=CC(C(=O)NCC=2C=CC=CC=2)=CC=C1NC(=O)NC1=CC=CC=C1Br NHDUIVOKCKQWEF-UHFFFAOYSA-N 0.000 description 3
- 230000008569 process Effects 0.000 description 3
- 229920006395 saturated elastomer Polymers 0.000 description 3
- HWLYIRABQGWLBV-UHFFFAOYSA-N (2-phenylphenyl)urea Chemical compound NC(=O)NC1=CC=CC=C1C1=CC=CC=C1 HWLYIRABQGWLBV-UHFFFAOYSA-N 0.000 description 2
- DRAKOGLGKMMYMG-UHFFFAOYSA-N (3-amino-2-hydroxyphenyl)-phenylmethanone Chemical compound NC1=CC=CC(C(=O)C=2C=CC=CC=2)=C1O DRAKOGLGKMMYMG-UHFFFAOYSA-N 0.000 description 2
- UIVHJEKZRQNSEB-UHFFFAOYSA-N (3-hydroxy-4-nitrophenyl)-phenylmethanone Chemical compound C1=C([N+]([O-])=O)C(O)=CC(C(=O)C=2C=CC=CC=2)=C1 UIVHJEKZRQNSEB-UHFFFAOYSA-N 0.000 description 2
- GTIBQOVZWUVLOF-UHFFFAOYSA-N (4-amino-3-hydroxyphenyl)-phenylmethanone Chemical compound C1=C(O)C(N)=CC=C1C(=O)C1=CC=CC=C1 GTIBQOVZWUVLOF-UHFFFAOYSA-N 0.000 description 2
- WTRTYLZMKRSHMF-CMDGGOBGSA-N (e)-3-[3-[(2-bromophenyl)carbamoylamino]-2-hydroxyphenyl]prop-2-enamide Chemical compound NC(=O)\C=C\C1=CC=CC(NC(=O)NC=2C(=CC=CC=2)Br)=C1O WTRTYLZMKRSHMF-CMDGGOBGSA-N 0.000 description 2
- XMKLTEGSALONPH-UHFFFAOYSA-N 1,2,4,5-tetrazinane-3,6-dione Chemical compound O=C1NNC(=O)NN1 XMKLTEGSALONPH-UHFFFAOYSA-N 0.000 description 2
- DBUAYOWCIUQXQW-UHFFFAOYSA-N 1,3-benzodioxole-4-carboxylic acid Chemical compound OC(=O)C1=CC=CC2=C1OCO2 DBUAYOWCIUQXQW-UHFFFAOYSA-N 0.000 description 2
- XCUDXBBFNPKYKN-UHFFFAOYSA-N 1-(2,3-dichlorophenyl)-3-(3-hydroxynaphthalen-2-yl)urea Chemical compound OC1=CC2=CC=CC=C2C=C1NC(=O)NC1=CC=CC(Cl)=C1Cl XCUDXBBFNPKYKN-UHFFFAOYSA-N 0.000 description 2
- UHOSHUNGLMWKCU-UHFFFAOYSA-N 1-(2-anilinophenyl)-3-(2-hydroxy-4-nitrophenyl)urea Chemical compound OC1=CC([N+]([O-])=O)=CC=C1NC(=O)NC1=CC=CC=C1NC1=CC=CC=C1 UHOSHUNGLMWKCU-UHFFFAOYSA-N 0.000 description 2
- CFFRUCPLXOUIGF-UHFFFAOYSA-N 1-(2-bromophenyl)-3-(2,3,5-trifluoro-6-hydroxyphenyl)urea Chemical compound OC1=C(F)C=C(F)C(F)=C1NC(=O)NC1=CC=CC=C1Br CFFRUCPLXOUIGF-UHFFFAOYSA-N 0.000 description 2
- ZDQFTSZMJHUTNA-UHFFFAOYSA-N 1-(2-bromophenyl)-3-(2-hydroxy-4-methylphenyl)urea Chemical compound OC1=CC(C)=CC=C1NC(=O)NC1=CC=CC=C1Br ZDQFTSZMJHUTNA-UHFFFAOYSA-N 0.000 description 2
- XDJSETLIIYOAOI-UHFFFAOYSA-N 1-(2-bromophenyl)-3-(2-hydroxyphenyl)urea Chemical compound OC1=CC=CC=C1NC(=O)NC1=CC=CC=C1Br XDJSETLIIYOAOI-UHFFFAOYSA-N 0.000 description 2
- XCDHYNNOCDUAGE-UHFFFAOYSA-N 1-(2-bromophenyl)-3-(3,5-dichloro-2-hydroxyphenyl)urea Chemical compound OC1=C(Cl)C=C(Cl)C=C1NC(=O)NC1=CC=CC=C1Br XCDHYNNOCDUAGE-UHFFFAOYSA-N 0.000 description 2
- ANTFJOTVFCVVAW-UHFFFAOYSA-N 1-(2-bromophenyl)-3-(3-hydroxynaphthalen-2-yl)urea Chemical compound OC1=CC2=CC=CC=C2C=C1NC(=O)NC1=CC=CC=C1Br ANTFJOTVFCVVAW-UHFFFAOYSA-N 0.000 description 2
- ZXNGXQUBIUOUHQ-UHFFFAOYSA-N 1-(2-bromophenyl)-3-(4-cyanophenyl)urea Chemical compound BrC1=CC=CC=C1NC(=O)NC1=CC=C(C#N)C=C1 ZXNGXQUBIUOUHQ-UHFFFAOYSA-N 0.000 description 2
- ZILKNQHGVMDEEV-UHFFFAOYSA-N 1-(2-bromophenyl)-3-(5-cyano-2-hydroxyphenyl)urea Chemical compound OC1=CC=C(C#N)C=C1NC(=O)NC1=CC=CC=C1Br ZILKNQHGVMDEEV-UHFFFAOYSA-N 0.000 description 2
- KPJZGZQNCRHCPT-UHFFFAOYSA-N 1-(2-bromophenyl)-3-(5-fluoro-2-hydroxyphenyl)urea Chemical compound OC1=CC=C(F)C=C1NC(=O)NC1=CC=CC=C1Br KPJZGZQNCRHCPT-UHFFFAOYSA-N 0.000 description 2
- KHTGUBPZTDJRNE-UHFFFAOYSA-N 1-(2-bromophenyl)-3-[2-(2-phenyl-1-sulfamoylethenyl)phenyl]urea Chemical compound C=1C=CC=C(NC(=O)NC=2C(=CC=CC=2)Br)C=1C(S(=O)(=O)N)=CC1=CC=CC=C1 KHTGUBPZTDJRNE-UHFFFAOYSA-N 0.000 description 2
- HJTAKPYPAXJAQZ-UHFFFAOYSA-N 1-(2-bromophenyl)-3-[2-(2-sulfamoylquinolin-8-yl)phenyl]urea Chemical compound C12=NC(S(=O)(=O)N)=CC=C2C=CC=C1C1=CC=CC=C1NC(=O)NC1=CC=CC=C1Br HJTAKPYPAXJAQZ-UHFFFAOYSA-N 0.000 description 2
- GLAXMZMFLGMTKJ-UHFFFAOYSA-N 1-(2-bromophenyl)-3-[2-(quinolin-8-ylsulfonylamino)phenyl]urea Chemical compound BrC1=CC=CC=C1NC(=O)NC1=CC=CC=C1NS(=O)(=O)C1=CC=CC2=CC=CN=C12 GLAXMZMFLGMTKJ-UHFFFAOYSA-N 0.000 description 2
- YIQZPEHGQARGRD-UHFFFAOYSA-N 1-(2-bromophenyl)-3-[2-[2-(sulfamoylamino)phenyl]phenyl]urea Chemical compound NS(=O)(=O)NC1=CC=CC=C1C1=CC=CC=C1NC(=O)NC1=CC=CC=C1Br YIQZPEHGQARGRD-UHFFFAOYSA-N 0.000 description 2
- KKBLAUFCLVADNC-UHFFFAOYSA-N 1-(2-bromophenyl)-3-[2-[phenyl(sulfamoyl)methyl]phenyl]urea Chemical compound C=1C=CC=C(NC(=O)NC=2C(=CC=CC=2)Br)C=1C(S(=O)(=O)N)C1=CC=CC=C1 KKBLAUFCLVADNC-UHFFFAOYSA-N 0.000 description 2
- ZGBWDCCQBAHGGC-UHFFFAOYSA-N 1-(2-chloro-6-methylphenyl)-3-(2-hydroxy-4-nitrophenyl)urea Chemical compound CC1=CC=CC(Cl)=C1NC(=O)NC1=CC=C([N+]([O-])=O)C=C1O ZGBWDCCQBAHGGC-UHFFFAOYSA-N 0.000 description 2
- XXMZFIZXCJRLQC-UHFFFAOYSA-N 1-(2-ethoxyphenyl)-3-(2-hydroxy-4-nitrophenyl)urea Chemical compound CCOC1=CC=CC=C1NC(=O)NC1=CC=C([N+]([O-])=O)C=C1O XXMZFIZXCJRLQC-UHFFFAOYSA-N 0.000 description 2
- PZBLZIXURGLVNI-UHFFFAOYSA-N 1-(2-ethylphenyl)-3-(2-hydroxy-4-nitrophenyl)urea Chemical compound CCC1=CC=CC=C1NC(=O)NC1=CC=C([N+]([O-])=O)C=C1O PZBLZIXURGLVNI-UHFFFAOYSA-N 0.000 description 2
- PPCPBEOXHRRPRQ-UHFFFAOYSA-N 1-(2-fluorophenyl)-3-(2-hydroxy-4-nitrophenyl)urea Chemical compound OC1=CC([N+]([O-])=O)=CC=C1NC(=O)NC1=CC=CC=C1F PPCPBEOXHRRPRQ-UHFFFAOYSA-N 0.000 description 2
- JDKDJPABXADLJT-UHFFFAOYSA-N 1-(2-hydroxy-4-nitrophenyl)-3-(2-methylphenyl)urea Chemical compound CC1=CC=CC=C1NC(=O)NC1=CC=C([N+]([O-])=O)C=C1O JDKDJPABXADLJT-UHFFFAOYSA-N 0.000 description 2
- HJGCROGLAWQCDZ-UHFFFAOYSA-N 1-(2-hydroxy-4-nitrophenyl)-3-[2-(trifluoromethoxy)phenyl]urea Chemical compound OC1=CC([N+]([O-])=O)=CC=C1NC(=O)NC1=CC=CC=C1OC(F)(F)F HJGCROGLAWQCDZ-UHFFFAOYSA-N 0.000 description 2
- OGTOIQDAKFXVIH-UHFFFAOYSA-N 1-(2-hydroxy-4-nitrophenyl)-3-[3-(trifluoromethyl)phenyl]urea Chemical compound OC1=CC([N+]([O-])=O)=CC=C1NC(=O)NC1=CC=CC(C(F)(F)F)=C1 OGTOIQDAKFXVIH-UHFFFAOYSA-N 0.000 description 2
- DAJJRPMUKJGYSD-UHFFFAOYSA-N 1-(2-hydroxy-4-nitrophenyl)-3-[4-(trifluoromethyl)phenyl]urea Chemical compound OC1=CC([N+]([O-])=O)=CC=C1NC(=O)NC1=CC=C(C(F)(F)F)C=C1 DAJJRPMUKJGYSD-UHFFFAOYSA-N 0.000 description 2
- KACUYPOBOHDUPW-UHFFFAOYSA-N 1-(2-hydroxy-4-nitrophenyl)-3-phenylthiourea Chemical compound OC1=CC([N+]([O-])=O)=CC=C1NC(=S)NC1=CC=CC=C1 KACUYPOBOHDUPW-UHFFFAOYSA-N 0.000 description 2
- SAUHQYBXEGARCC-UHFFFAOYSA-N 1-(2-hydroxy-4-nitrophenyl)-3-phenylurea Chemical compound OC1=CC([N+]([O-])=O)=CC=C1NC(=O)NC1=CC=CC=C1 SAUHQYBXEGARCC-UHFFFAOYSA-N 0.000 description 2
- YDCPBZQRMMLIFE-UHFFFAOYSA-N 1-(3-amino-2-hydroxyphenyl)-3-(2-bromophenyl)urea Chemical compound NC1=CC=CC(NC(=O)NC=2C(=CC=CC=2)Br)=C1O YDCPBZQRMMLIFE-UHFFFAOYSA-N 0.000 description 2
- IVTNMVXSFFOHJN-UHFFFAOYSA-N 1-(3-azido-2-hydroxyphenyl)-3-(2-bromophenyl)urea Chemical compound C1=CC=C(N=[N+]=[N-])C(O)=C1NC(=O)NC1=CC=CC=C1Br IVTNMVXSFFOHJN-UHFFFAOYSA-N 0.000 description 2
- YENXEPKNDWFMOD-UHFFFAOYSA-N 1-(3-cyano-2-hydroxyphenyl)-3-(2-phenylphenyl)urea Chemical compound C1=CC=C(C#N)C(O)=C1NC(=O)NC1=CC=CC=C1C1=CC=CC=C1 YENXEPKNDWFMOD-UHFFFAOYSA-N 0.000 description 2
- YHJMULJKVLDLHI-UHFFFAOYSA-N 1-(3-hydroxynaphthalen-2-yl)-3-[3-(trifluoromethyl)phenyl]urea Chemical compound OC1=CC2=CC=CC=C2C=C1NC(=O)NC1=CC=CC(C(F)(F)F)=C1 YHJMULJKVLDLHI-UHFFFAOYSA-N 0.000 description 2
- YRQTZTMQVYXFLH-UHFFFAOYSA-N 1-(4-amino-2-hydroxyphenyl)-3-(2-iodophenyl)urea Chemical compound OC1=CC(N)=CC=C1NC(=O)NC1=CC=CC=C1I YRQTZTMQVYXFLH-UHFFFAOYSA-N 0.000 description 2
- WWGPATOURAKKMY-UHFFFAOYSA-N 1-(4-amino-2-hydroxyphenyl)-3-(2-methoxyphenyl)urea Chemical compound COC1=CC=CC=C1NC(=O)NC1=CC=C(N)C=C1O WWGPATOURAKKMY-UHFFFAOYSA-N 0.000 description 2
- WRABKWBTESAPEL-UHFFFAOYSA-N 1-(4-bromophenyl)-3-(2-hydroxy-4-nitrophenyl)urea Chemical compound OC1=CC([N+]([O-])=O)=CC=C1NC(=O)NC1=CC=C(Br)C=C1 WRABKWBTESAPEL-UHFFFAOYSA-N 0.000 description 2
- DQRKQLTUZBFHPB-UHFFFAOYSA-N 1-(4-cyano-2-hydroxyphenyl)-3-(2,3-dichlorophenyl)urea Chemical compound OC1=CC(C#N)=CC=C1NC(=O)NC1=CC=CC(Cl)=C1Cl DQRKQLTUZBFHPB-UHFFFAOYSA-N 0.000 description 2
- OGRVRTFZEUPTGZ-UHFFFAOYSA-N 1-(4-cyano-2-hydroxyphenyl)-3-(3-methoxyphenyl)urea Chemical compound COC1=CC=CC(NC(=O)NC=2C(=CC(=CC=2)C#N)O)=C1 OGRVRTFZEUPTGZ-UHFFFAOYSA-N 0.000 description 2
- BIMKTPJMZSGZJT-UHFFFAOYSA-N 1-(4-cyano-2-hydroxyphenyl)-3-[4-(trifluoromethyl)phenyl]urea Chemical compound OC1=CC(C#N)=CC=C1NC(=O)NC1=CC=C(C(F)(F)F)C=C1 BIMKTPJMZSGZJT-UHFFFAOYSA-N 0.000 description 2
- YGAGRJGVAXCRFI-UHFFFAOYSA-N 1-(4-fluoro-2-hydroxyphenyl)-3-phenylurea Chemical compound OC1=CC(F)=CC=C1NC(=O)NC1=CC=CC=C1 YGAGRJGVAXCRFI-UHFFFAOYSA-N 0.000 description 2
- ASMCVDDUQQZBFK-UHFFFAOYSA-N 1-(5-chloro-2-hydroxy-4-nitrophenyl)-3-phenylurea Chemical compound OC1=CC([N+]([O-])=O)=C(Cl)C=C1NC(=O)NC1=CC=CC=C1 ASMCVDDUQQZBFK-UHFFFAOYSA-N 0.000 description 2
- WDUQWXBGIVITRM-UHFFFAOYSA-N 1-[2-(benzylsulfonylamino)phenyl]-3-(2-bromophenyl)urea Chemical compound BrC1=CC=CC=C1NC(=O)NC1=CC=CC=C1NS(=O)(=O)CC1=CC=CC=C1 WDUQWXBGIVITRM-UHFFFAOYSA-N 0.000 description 2
- ROVKFKQMANVQJQ-UHFFFAOYSA-N 1-[2-chloro-5-(trifluoromethyl)phenyl]-3-(2-hydroxy-4-propan-2-ylphenyl)urea Chemical compound OC1=CC(C(C)C)=CC=C1NC(=O)NC1=CC(C(F)(F)F)=CC=C1Cl ROVKFKQMANVQJQ-UHFFFAOYSA-N 0.000 description 2
- SJWMPHKDBJQBBJ-UHFFFAOYSA-N 2,3-dichloro-6-nitrophenol Chemical compound OC1=C(Cl)C(Cl)=CC=C1[N+]([O-])=O SJWMPHKDBJQBBJ-UHFFFAOYSA-N 0.000 description 2
- RQPVXOUSZLDZFI-UHFFFAOYSA-N 2-(phenylcarbamoylamino)benzoic acid Chemical compound OC(=O)C1=CC=CC=C1NC(=O)NC1=CC=CC=C1 RQPVXOUSZLDZFI-UHFFFAOYSA-N 0.000 description 2
- HYNQTSZBTIOFKH-UHFFFAOYSA-N 2-Amino-5-hydroxybenzoic acid Chemical compound NC1=CC=C(O)C=C1C(O)=O HYNQTSZBTIOFKH-UHFFFAOYSA-N 0.000 description 2
- PJIQDIIKISUDNK-UHFFFAOYSA-N 2-[(2-amino-5-bromophenyl)disulfanyl]-4-bromoaniline Chemical compound NC1=CC=C(Br)C=C1SSC1=CC(Br)=CC=C1N PJIQDIIKISUDNK-UHFFFAOYSA-N 0.000 description 2
- JSHJJLQJRLNBBA-UHFFFAOYSA-N 2-amino-3-chlorophenol Chemical compound NC1=C(O)C=CC=C1Cl JSHJJLQJRLNBBA-UHFFFAOYSA-N 0.000 description 2
- QOZLOYKAFDTQNU-UHFFFAOYSA-N 2-amino-3-fluorophenol Chemical compound NC1=C(O)C=CC=C1F QOZLOYKAFDTQNU-UHFFFAOYSA-N 0.000 description 2
- WASQBNCGNUTVNI-UHFFFAOYSA-N 2-amino-4,6-dichlorophenol Chemical compound NC1=CC(Cl)=CC(Cl)=C1O WASQBNCGNUTVNI-UHFFFAOYSA-N 0.000 description 2
- GOJUJUVQIVIZAV-UHFFFAOYSA-N 2-amino-4,6-dichloropyrimidine-5-carbaldehyde Chemical group NC1=NC(Cl)=C(C=O)C(Cl)=N1 GOJUJUVQIVIZAV-UHFFFAOYSA-N 0.000 description 2
- BHTKIYIEMXRHGL-UHFFFAOYSA-N 2-amino-4-(trifluoromethyl)phenol Chemical compound NC1=CC(C(F)(F)F)=CC=C1O BHTKIYIEMXRHGL-UHFFFAOYSA-N 0.000 description 2
- BXNQDUMAPNIZAG-UHFFFAOYSA-N 2-amino-4-bromo-6-fluorophenol Chemical compound NC1=CC(Br)=CC(F)=C1O BXNQDUMAPNIZAG-UHFFFAOYSA-N 0.000 description 2
- VBCFPAKCBOGUBW-UHFFFAOYSA-N 2-amino-5-ethylphenol Chemical compound CCC1=CC=C(N)C(O)=C1 VBCFPAKCBOGUBW-UHFFFAOYSA-N 0.000 description 2
- ILGJVPVZCNNBKI-UHFFFAOYSA-N 2-amino-5-phenylphenol Chemical compound C1=C(O)C(N)=CC=C1C1=CC=CC=C1 ILGJVPVZCNNBKI-UHFFFAOYSA-N 0.000 description 2
- XOXAZFFGKMIJKE-UHFFFAOYSA-N 2-amino-6-(trifluoromethyl)phenol Chemical compound NC1=CC=CC(C(F)(F)F)=C1O XOXAZFFGKMIJKE-UHFFFAOYSA-N 0.000 description 2
- ABKAJPLOOIISSW-UHFFFAOYSA-N 2-amino-6-propylphenol Chemical compound CCCC1=CC=CC(N)=C1O ABKAJPLOOIISSW-UHFFFAOYSA-N 0.000 description 2
- UTYJWCLETJNCCW-UHFFFAOYSA-N 2-amino-9h-fluoren-1-ol Chemical compound C1=CC=C2C3=CC=C(N)C(O)=C3CC2=C1 UTYJWCLETJNCCW-UHFFFAOYSA-N 0.000 description 2
- CDAWCLOXVUBKRW-UHFFFAOYSA-N 2-aminophenol Chemical compound NC1=CC=CC=C1O CDAWCLOXVUBKRW-UHFFFAOYSA-N 0.000 description 2
- LFAHYSRYSVNIET-UHFFFAOYSA-N 2-hydroxy-3-nitro-1,2,3,4-tetrahydroinden-5-one Chemical compound [O-][N+](=O)C1C(O)CC2=C1CC(=O)C=C2 LFAHYSRYSVNIET-UHFFFAOYSA-N 0.000 description 2
- ZWZZWLBNEPKBAR-UHFFFAOYSA-N 2-hydroxy-3-nitro-n-phenylbenzamide Chemical compound C1=CC=C([N+]([O-])=O)C(O)=C1C(=O)NC1=CC=CC=C1 ZWZZWLBNEPKBAR-UHFFFAOYSA-N 0.000 description 2
- ZUOCMGGILLTISV-UHFFFAOYSA-N 2-hydroxy-3-nitrobenzonitrile Chemical compound OC1=C(C#N)C=CC=C1[N+]([O-])=O ZUOCMGGILLTISV-UHFFFAOYSA-N 0.000 description 2
- HYPKGPVDQYUOSV-UHFFFAOYSA-N 2-nitro-5-phenylphenol Chemical compound C1=C([N+]([O-])=O)C(O)=CC(C=2C=CC=CC=2)=C1 HYPKGPVDQYUOSV-UHFFFAOYSA-N 0.000 description 2
- QJJGPZTVVISLNI-UHFFFAOYSA-N 2-nitro-5-propan-2-ylphenol Chemical compound CC(C)C1=CC=C([N+]([O-])=O)C(O)=C1 QJJGPZTVVISLNI-UHFFFAOYSA-N 0.000 description 2
- FMPKXGYPBUWNNG-UHFFFAOYSA-N 2-nitro-6-(trifluoromethyl)phenol Chemical compound OC1=C([N+]([O-])=O)C=CC=C1C(F)(F)F FMPKXGYPBUWNNG-UHFFFAOYSA-N 0.000 description 2
- OSIJREAKAWRMJQ-UHFFFAOYSA-N 2-nitro-6-phenylmethoxyphenol Chemical compound C1=CC=C([N+]([O-])=O)C(O)=C1OCC1=CC=CC=C1 OSIJREAKAWRMJQ-UHFFFAOYSA-N 0.000 description 2
- IIBOYMCHHLZIKC-UHFFFAOYSA-N 2-nitro-6-phenylphenol Chemical compound C1=CC=C([N+]([O-])=O)C(O)=C1C1=CC=CC=C1 IIBOYMCHHLZIKC-UHFFFAOYSA-N 0.000 description 2
- LJGHYPLBDBRCRZ-UHFFFAOYSA-N 3-(3-aminophenyl)sulfonylaniline Chemical group NC1=CC=CC(S(=O)(=O)C=2C=C(N)C=CC=2)=C1 LJGHYPLBDBRCRZ-UHFFFAOYSA-N 0.000 description 2
- HMNKTRSOROOSPP-UHFFFAOYSA-N 3-Ethylphenol Chemical compound CCC1=CC=CC(O)=C1 HMNKTRSOROOSPP-UHFFFAOYSA-N 0.000 description 2
- CAHLKWGNFWVZRJ-UHFFFAOYSA-N 3-[(2-bromophenyl)carbamoylamino]-2-hydroxybenzoic acid Chemical compound OC(=O)C1=CC=CC(NC(=O)NC=2C(=CC=CC=2)Br)=C1O CAHLKWGNFWVZRJ-UHFFFAOYSA-N 0.000 description 2
- CQFHTGIXWOGKOR-UHFFFAOYSA-N 3-[2-(hydroxymethyl)-3-nitrophenyl]prop-2-enoic acid Chemical compound OCC1=C(C=CC(O)=O)C=CC=C1[N+]([O-])=O CQFHTGIXWOGKOR-UHFFFAOYSA-N 0.000 description 2
- IUUXZDXFVTWINA-UHFFFAOYSA-N 3-[2-[(2-bromophenyl)carbamoylamino]phenyl]-2-hydroxybenzoic acid Chemical compound OC(=O)C1=CC=CC(C=2C(=CC=CC=2)NC(=O)NC=2C(=CC=CC=2)Br)=C1O IUUXZDXFVTWINA-UHFFFAOYSA-N 0.000 description 2
- JJIHEKRAOZPTLG-UHFFFAOYSA-N 3-[3-(hydroxymethyl)-4-nitrophenyl]prop-2-enoic acid Chemical compound OCC1=CC(C=CC(O)=O)=CC=C1[N+]([O-])=O JJIHEKRAOZPTLG-UHFFFAOYSA-N 0.000 description 2
- NGJXVRLGYMLJIW-UHFFFAOYSA-N 3-[3-amino-2-(hydroxymethyl)phenyl]prop-2-enoic acid Chemical compound NC1=CC=CC(C=CC(O)=O)=C1CO NGJXVRLGYMLJIW-UHFFFAOYSA-N 0.000 description 2
- CQXVCXOOWZJYNA-UHFFFAOYSA-N 3-[4-amino-3-(hydroxymethyl)phenyl]prop-2-enoic acid Chemical compound NC1=CC=C(C=CC(O)=O)C=C1CO CQXVCXOOWZJYNA-UHFFFAOYSA-N 0.000 description 2
- JPPJUPXPQUNYOH-UHFFFAOYSA-N 3-amino-2-hydroxy-1,2,3,4-tetrahydroinden-5-one Chemical compound C1C(=O)C=CC2=C1C(N)C(O)C2 JPPJUPXPQUNYOH-UHFFFAOYSA-N 0.000 description 2
- NTUMBWHJAMGXFO-UHFFFAOYSA-N 3-amino-2-hydroxy-6-methylbenzonitrile Chemical compound CC1=CC=C(N)C(O)=C1C#N NTUMBWHJAMGXFO-UHFFFAOYSA-N 0.000 description 2
- PTGSDRFHFSNCIP-UHFFFAOYSA-N 3-amino-2-hydroxy-n-phenylbenzamide Chemical compound NC1=CC=CC(C(=O)NC=2C=CC=CC=2)=C1O PTGSDRFHFSNCIP-UHFFFAOYSA-N 0.000 description 2
- ZEWCASRNRWXXSO-UHFFFAOYSA-N 3-amino-4-hydroxybenzonitrile Chemical compound NC1=CC(C#N)=CC=C1O ZEWCASRNRWXXSO-UHFFFAOYSA-N 0.000 description 2
- ZHVPTERSBUMMHK-UHFFFAOYSA-N 3-aminonaphthalen-2-ol Chemical compound C1=CC=C2C=C(O)C(N)=CC2=C1 ZHVPTERSBUMMHK-UHFFFAOYSA-N 0.000 description 2
- JHNIFYUIFUWEFO-UHFFFAOYSA-N 3-hydroxy-4-nitrobenzonitrile Chemical compound OC1=CC(C#N)=CC=C1[N+]([O-])=O JHNIFYUIFUWEFO-UHFFFAOYSA-N 0.000 description 2
- VLJSLTNSFSOYQR-UHFFFAOYSA-N 3-propan-2-ylphenol Chemical compound CC(C)C1=CC=CC(O)=C1 VLJSLTNSFSOYQR-UHFFFAOYSA-N 0.000 description 2
- YBAZINRZQSAIAY-UHFFFAOYSA-N 4-aminobenzonitrile Chemical compound NC1=CC=C(C#N)C=C1 YBAZINRZQSAIAY-UHFFFAOYSA-N 0.000 description 2
- 125000004801 4-cyanophenyl group Chemical group [H]C1=C([H])C(C#N)=C([H])C([H])=C1* 0.000 description 2
- NKJIFDNZPGLLSH-UHFFFAOYSA-N 4-nitrobenzonitrile Chemical compound [O-][N+](=O)C1=CC=C(C#N)C=C1 NKJIFDNZPGLLSH-UHFFFAOYSA-N 0.000 description 2
- YJIUQVPMLSGDMP-UHFFFAOYSA-N 5-ethyl-2-nitrophenol Chemical compound CCC1=CC=C([N+]([O-])=O)C(O)=C1 YJIUQVPMLSGDMP-UHFFFAOYSA-N 0.000 description 2
- KRURHZHJEDNBCM-UHFFFAOYSA-N 6-amino-2,3-difluorophenol Chemical compound NC1=CC=C(F)C(F)=C1O KRURHZHJEDNBCM-UHFFFAOYSA-N 0.000 description 2
- 206010001052 Acute respiratory distress syndrome Diseases 0.000 description 2
- 102000040717 Alpha family Human genes 0.000 description 2
- 108091071248 Alpha family Proteins 0.000 description 2
- 208000024827 Alzheimer disease Diseases 0.000 description 2
- QGZKDVFQNNGYKY-UHFFFAOYSA-N Ammonia Chemical compound N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 description 2
- GWEHVDNNLFDJLR-UHFFFAOYSA-N Carbanilide Natural products C=1C=CC=CC=1NC(=O)NC1=CC=CC=C1 GWEHVDNNLFDJLR-UHFFFAOYSA-N 0.000 description 2
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 2
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 2
- 208000006545 Chronic Obstructive Pulmonary Disease Diseases 0.000 description 2
- 206010009900 Colitis ulcerative Diseases 0.000 description 2
- 208000011231 Crohn disease Diseases 0.000 description 2
- 102000004127 Cytokines Human genes 0.000 description 2
- 108090000695 Cytokines Proteins 0.000 description 2
- 206010012438 Dermatitis atopic Diseases 0.000 description 2
- IAZDPXIOMUYVGZ-WFGJKAKNSA-N Dimethyl sulfoxide Chemical compound [2H]C([2H])([2H])S(=O)C([2H])([2H])[2H] IAZDPXIOMUYVGZ-WFGJKAKNSA-N 0.000 description 2
- 206010014824 Endotoxic shock Diseases 0.000 description 2
- 108090000790 Enzymes Proteins 0.000 description 2
- 102000004190 Enzymes Human genes 0.000 description 2
- PXGOKWXKJXAPGV-UHFFFAOYSA-N Fluorine Chemical compound FF PXGOKWXKJXAPGV-UHFFFAOYSA-N 0.000 description 2
- VZCYOOQTPOCHFL-OWOJBTEDSA-N Fumaric acid Chemical compound OC(=O)\C=C\C(O)=O VZCYOOQTPOCHFL-OWOJBTEDSA-N 0.000 description 2
- YLQBMQCUIZJEEH-UHFFFAOYSA-N Furan Chemical compound C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 2
- 206010018364 Glomerulonephritis Diseases 0.000 description 2
- NTYJJOPFIAHURM-UHFFFAOYSA-N Histamine Chemical compound NCCC1=CN=CN1 NTYJJOPFIAHURM-UHFFFAOYSA-N 0.000 description 2
- 101500025785 Homo sapiens IL-8(6-77) Proteins 0.000 description 2
- 102400001232 IL-8(6-77) Human genes 0.000 description 2
- 208000022559 Inflammatory bowel disease Diseases 0.000 description 2
- YNAVUWVOSKDBBP-UHFFFAOYSA-N Morpholine Chemical compound C1COCCN1 YNAVUWVOSKDBBP-UHFFFAOYSA-N 0.000 description 2
- 101001055218 Oryctolagus cuniculus Interleukin-8 Proteins 0.000 description 2
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 2
- GLUUGHFHXGJENI-UHFFFAOYSA-N Piperazine Chemical compound C1CNCCN1 GLUUGHFHXGJENI-UHFFFAOYSA-N 0.000 description 2
- NQRYJNQNLNOLGT-UHFFFAOYSA-N Piperidine Chemical compound C1CCNCC1 NQRYJNQNLNOLGT-UHFFFAOYSA-N 0.000 description 2
- KAESVJOAVNADME-UHFFFAOYSA-N Pyrrole Chemical compound C=1C=CNC=1 KAESVJOAVNADME-UHFFFAOYSA-N 0.000 description 2
- SMWDFEZZVXVKRB-UHFFFAOYSA-N Quinoline Chemical compound N1=CC=CC2=CC=CC=C21 SMWDFEZZVXVKRB-UHFFFAOYSA-N 0.000 description 2
- 206010063837 Reperfusion injury Diseases 0.000 description 2
- 208000013616 Respiratory Distress Syndrome Diseases 0.000 description 2
- 206010040047 Sepsis Diseases 0.000 description 2
- 208000006011 Stroke Diseases 0.000 description 2
- UCKMPCXJQFINFW-UHFFFAOYSA-N Sulphide Chemical compound [S-2] UCKMPCXJQFINFW-UHFFFAOYSA-N 0.000 description 2
- 208000007536 Thrombosis Diseases 0.000 description 2
- 206010044248 Toxic shock syndrome Diseases 0.000 description 2
- 231100000650 Toxic shock syndrome Toxicity 0.000 description 2
- 201000006704 Ulcerative Colitis Diseases 0.000 description 2
- ZUSWDTWYONAOPH-UHFFFAOYSA-N [2-(trifluoromethyl)phenyl]hydrazine;hydrochloride Chemical group [Cl-].[NH3+]NC1=CC=CC=C1C(F)(F)F ZUSWDTWYONAOPH-UHFFFAOYSA-N 0.000 description 2
- SLVJLNZLOZPQID-UHFFFAOYSA-N [2-[(2-hydroxy-4-nitrophenyl)carbamoylamino]phenyl]methyl hydrogen sulfate Chemical compound OC1=CC([N+]([O-])=O)=CC=C1NC(=O)NC1=CC=CC=C1COS(O)(=O)=O SLVJLNZLOZPQID-UHFFFAOYSA-N 0.000 description 2
- 208000011341 adult acute respiratory distress syndrome Diseases 0.000 description 2
- 201000000028 adult respiratory distress syndrome Diseases 0.000 description 2
- 206010003246 arthritis Diseases 0.000 description 2
- 208000006673 asthma Diseases 0.000 description 2
- 201000008937 atopic dermatitis Diseases 0.000 description 2
- CSKNSYBAZOQPLR-UHFFFAOYSA-N benzenesulfonyl chloride Chemical compound ClS(=O)(=O)C1=CC=CC=C1 CSKNSYBAZOQPLR-UHFFFAOYSA-N 0.000 description 2
- RROBIDXNTUAHFW-UHFFFAOYSA-N benzotriazol-1-yloxy-tris(dimethylamino)phosphanium Chemical compound C1=CC=C2N(O[P+](N(C)C)(N(C)C)N(C)C)N=NC2=C1 RROBIDXNTUAHFW-UHFFFAOYSA-N 0.000 description 2
- LLEMOWNGBBNAJR-UHFFFAOYSA-N biphenyl-2-ol Chemical compound OC1=CC=CC=C1C1=CC=CC=C1 LLEMOWNGBBNAJR-UHFFFAOYSA-N 0.000 description 2
- 125000001246 bromo group Chemical group Br* 0.000 description 2
- 125000004432 carbon atom Chemical group C* 0.000 description 2
- 235000011089 carbon dioxide Nutrition 0.000 description 2
- 125000002915 carbonyl group Chemical group [*:2]C([*:1])=O 0.000 description 2
- 230000000747 cardiac effect Effects 0.000 description 2
- 150000001768 cations Chemical class 0.000 description 2
- 230000008859 change Effects 0.000 description 2
- 239000003153 chemical reaction reagent Substances 0.000 description 2
- 239000002975 chemoattractant Substances 0.000 description 2
- 239000000460 chlorine Substances 0.000 description 2
- 229910052801 chlorine Inorganic materials 0.000 description 2
- 125000001309 chloro group Chemical group Cl* 0.000 description 2
- 125000004122 cyclic group Chemical group 0.000 description 2
- LPIQUOYDBNQMRZ-UHFFFAOYSA-N cyclopentene Chemical compound C1CC=CC1 LPIQUOYDBNQMRZ-UHFFFAOYSA-N 0.000 description 2
- 238000011161 development Methods 0.000 description 2
- 125000005442 diisocyanate group Chemical group 0.000 description 2
- 239000003814 drug Substances 0.000 description 2
- 238000002330 electrospray ionisation mass spectrometry Methods 0.000 description 2
- 125000001301 ethoxy group Chemical group [H]C([H])([H])C([H])([H])O* 0.000 description 2
- 229910052731 fluorine Inorganic materials 0.000 description 2
- 239000011737 fluorine Substances 0.000 description 2
- 230000006870 function Effects 0.000 description 2
- 230000005764 inhibitory process Effects 0.000 description 2
- 230000021995 interleukin-8 production Effects 0.000 description 2
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 2
- AWJUIBRHMBBTKR-UHFFFAOYSA-N isoquinoline Chemical compound C1=NC=CC2=CC=CC=C21 AWJUIBRHMBBTKR-UHFFFAOYSA-N 0.000 description 2
- JVTAAEKCZFNVCJ-UHFFFAOYSA-N lactic acid Chemical compound CC(O)C(O)=O JVTAAEKCZFNVCJ-UHFFFAOYSA-N 0.000 description 2
- 210000002540 macrophage Anatomy 0.000 description 2
- XVUHKSLMBFWCIU-MDZDMXLPSA-N methyl (e)-3-[3-[(2-bromophenyl)carbamoylamino]-2-hydroxyphenyl]prop-2-enoate Chemical compound COC(=O)\C=C\C1=CC=CC(NC(=O)NC=2C(=CC=CC=2)Br)=C1O XVUHKSLMBFWCIU-MDZDMXLPSA-N 0.000 description 2
- FGMISCVVYSYIAQ-UHFFFAOYSA-N methyl 4-[(2-bromophenyl)carbamoylamino]-3-hydroxybenzoate Chemical compound OC1=CC(C(=O)OC)=CC=C1NC(=O)NC1=CC=CC=C1Br FGMISCVVYSYIAQ-UHFFFAOYSA-N 0.000 description 2
- KUJANOFPCPKYLF-UHFFFAOYSA-N n-(2-amino-5-nitrophenyl)benzenesulfonamide Chemical compound NC1=CC=C([N+]([O-])=O)C=C1NS(=O)(=O)C1=CC=CC=C1 KUJANOFPCPKYLF-UHFFFAOYSA-N 0.000 description 2
- WMWPOLBAWLJWPR-UHFFFAOYSA-N n-(2-aminophenyl)-1-phenylmethanesulfonamide Chemical compound NC1=CC=CC=C1NS(=O)(=O)CC1=CC=CC=C1 WMWPOLBAWLJWPR-UHFFFAOYSA-N 0.000 description 2
- LRQSAKRTESGWED-UHFFFAOYSA-N n-(2-aminophenyl)-3,5-bis(trifluoromethyl)benzenesulfonamide Chemical compound NC1=CC=CC=C1NS(=O)(=O)C1=CC(C(F)(F)F)=CC(C(F)(F)F)=C1 LRQSAKRTESGWED-UHFFFAOYSA-N 0.000 description 2
- GOYLVLYWQRBVRD-UHFFFAOYSA-N n-(2-aminophenyl)-3-methylbenzenesulfonamide Chemical compound CC1=CC=CC(S(=O)(=O)NC=2C(=CC=CC=2)N)=C1 GOYLVLYWQRBVRD-UHFFFAOYSA-N 0.000 description 2
- NFPKLXASODMBIP-UHFFFAOYSA-N n-(2-aminophenyl)quinoline-8-sulfonamide Chemical compound NC1=CC=CC=C1NS(=O)(=O)C1=CC=CC2=CC=CN=C12 NFPKLXASODMBIP-UHFFFAOYSA-N 0.000 description 2
- YKWIWSQBYJZDJU-UHFFFAOYSA-N n-(2-anilinophenyl)-3,4-dimethoxybenzenesulfonamide Chemical compound C1=C(OC)C(OC)=CC=C1S(=O)(=O)NC1=CC=CC=C1NC1=CC=CC=C1 YKWIWSQBYJZDJU-UHFFFAOYSA-N 0.000 description 2
- WLHJKINWCGVNEU-UHFFFAOYSA-N n-[4-[(2-anilinophenyl)sulfamoyl]phenyl]acetamide Chemical compound C1=CC(NC(=O)C)=CC=C1S(=O)(=O)NC1=CC=CC=C1NC1=CC=CC=C1 WLHJKINWCGVNEU-UHFFFAOYSA-N 0.000 description 2
- AEURYAULWMMTBR-UHFFFAOYSA-N n-[5-[[2-[(2-bromophenyl)carbamoylamino]phenyl]sulfamoyl]-4-methyl-1,3-thiazol-2-yl]acetamide Chemical compound S1C(NC(=O)C)=NC(C)=C1S(=O)(=O)NC1=CC=CC=C1NC(=O)NC1=CC=CC=C1Br AEURYAULWMMTBR-UHFFFAOYSA-N 0.000 description 2
- 125000001624 naphthyl group Chemical group 0.000 description 2
- 125000004957 naphthylene group Chemical group 0.000 description 2
- 230000011242 neutrophil chemotaxis Effects 0.000 description 2
- 150000002828 nitro derivatives Chemical class 0.000 description 2
- 229910052763 palladium Inorganic materials 0.000 description 2
- WLJVXDMOQOGPHL-UHFFFAOYSA-N phenylacetic acid Chemical compound OC(=O)CC1=CC=CC=C1 WLJVXDMOQOGPHL-UHFFFAOYSA-N 0.000 description 2
- 230000001737 promoting effect Effects 0.000 description 2
- 125000006239 protecting group Chemical group 0.000 description 2
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 2
- 150000003254 radicals Chemical class 0.000 description 2
- 230000009467 reduction Effects 0.000 description 2
- 230000019254 respiratory burst Effects 0.000 description 2
- YGSDEFSMJLZEOE-UHFFFAOYSA-N salicylic acid Chemical compound OC(=O)C1=CC=CC=C1O YGSDEFSMJLZEOE-UHFFFAOYSA-N 0.000 description 2
- 230000036303 septic shock Effects 0.000 description 2
- 239000000377 silicon dioxide Substances 0.000 description 2
- 235000017557 sodium bicarbonate Nutrition 0.000 description 2
- 229910000104 sodium hydride Inorganic materials 0.000 description 2
- 230000003595 spectral effect Effects 0.000 description 2
- 125000000446 sulfanediyl group Chemical group *S* 0.000 description 2
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 2
- FPGGTKZVZWFYPV-UHFFFAOYSA-M tetrabutylammonium fluoride Chemical compound [F-].CCCC[N+](CCCC)(CCCC)CCCC FPGGTKZVZWFYPV-UHFFFAOYSA-M 0.000 description 2
- 150000003557 thiazoles Chemical class 0.000 description 2
- ZWZVWGITAAIFPS-UHFFFAOYSA-N thiophosgene Chemical compound ClC(Cl)=S ZWZVWGITAAIFPS-UHFFFAOYSA-N 0.000 description 2
- HPGGPRDJHPYFRM-UHFFFAOYSA-J tin(iv) chloride Chemical compound Cl[Sn](Cl)(Cl)Cl HPGGPRDJHPYFRM-UHFFFAOYSA-J 0.000 description 2
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 2
- VCGRFBXVSFAGGA-UHFFFAOYSA-N (1,1-dioxo-1,4-thiazinan-4-yl)-[6-[[3-(4-fluorophenyl)-5-methyl-1,2-oxazol-4-yl]methoxy]pyridin-3-yl]methanone Chemical compound CC=1ON=C(C=2C=CC(F)=CC=2)C=1COC(N=C1)=CC=C1C(=O)N1CCS(=O)(=O)CC1 VCGRFBXVSFAGGA-UHFFFAOYSA-N 0.000 description 1
- VATQKFJMBFFMBW-UHFFFAOYSA-N (2-anilinophenyl)urea Chemical compound NC(=O)NC1=CC=CC=C1NC1=CC=CC=C1 VATQKFJMBFFMBW-UHFFFAOYSA-N 0.000 description 1
- QIGMVYSPXPXCPN-UHFFFAOYSA-N (2-bromophenyl)thiourea Chemical compound NC(=S)NC1=CC=CC=C1Br QIGMVYSPXPXCPN-UHFFFAOYSA-N 0.000 description 1
- PAWVOCWEWJXILY-UHFFFAOYSA-N (2-fluorophenyl)urea Chemical compound NC(=O)NC1=CC=CC=C1F PAWVOCWEWJXILY-UHFFFAOYSA-N 0.000 description 1
- AKCUGQGKWDRTGL-UHFFFAOYSA-N (2-hydroxy-3-nitrophenyl)-phenylmethanone Chemical compound C1=CC=C([N+]([O-])=O)C(O)=C1C(=O)C1=CC=CC=C1 AKCUGQGKWDRTGL-UHFFFAOYSA-N 0.000 description 1
- OEBDKVHFNVETAN-UHFFFAOYSA-N (2-hydroxy-3-nitrophenyl)-phenylmethanone;(2-hydroxyphenyl)-phenylmethanone Chemical compound OC1=CC=CC=C1C(=O)C1=CC=CC=C1.C1=CC=C([N+]([O-])=O)C(O)=C1C(=O)C1=CC=CC=C1 OEBDKVHFNVETAN-UHFFFAOYSA-N 0.000 description 1
- QBYIENPQHBMVBV-HFEGYEGKSA-N (2R)-2-hydroxy-2-phenylacetic acid Chemical compound O[C@@H](C(O)=O)c1ccccc1.O[C@@H](C(O)=O)c1ccccc1 QBYIENPQHBMVBV-HFEGYEGKSA-N 0.000 description 1
- SHULEACXTONYPS-UHFFFAOYSA-N (3-hydroxyphenyl)-phenylmethanone Chemical compound OC1=CC=CC(C(=O)C=2C=CC=CC=2)=C1 SHULEACXTONYPS-UHFFFAOYSA-N 0.000 description 1
- 125000004178 (C1-C4) alkyl group Chemical group 0.000 description 1
- BJEPYKJPYRNKOW-REOHCLBHSA-N (S)-malic acid Chemical compound OC(=O)[C@@H](O)CC(O)=O BJEPYKJPYRNKOW-REOHCLBHSA-N 0.000 description 1
- ONWRSBMOCIQLRK-VOTSOKGWSA-N (e)-2-phenylethenesulfonyl chloride Chemical compound ClS(=O)(=O)\C=C\C1=CC=CC=C1 ONWRSBMOCIQLRK-VOTSOKGWSA-N 0.000 description 1
- HAXYXBBIILZWIZ-HRNDJLQDSA-N (e)-3-[3-[(2-bromophenyl)carbamoylamino]-2-hydroxyphenyl]prop-2-enamide;(2-bromophenyl)urea Chemical compound NC(=O)NC1=CC=CC=C1Br.NC(=O)\C=C\C1=CC=CC(NC(=O)NC=2C(=CC=CC=2)Br)=C1O HAXYXBBIILZWIZ-HRNDJLQDSA-N 0.000 description 1
- MOWXJLUYGFNTAL-DEOSSOPVSA-N (s)-[2-chloro-4-fluoro-5-(7-morpholin-4-ylquinazolin-4-yl)phenyl]-(6-methoxypyridazin-3-yl)methanol Chemical compound N1=NC(OC)=CC=C1[C@@H](O)C1=CC(C=2C3=CC=C(C=C3N=CN=2)N2CCOCC2)=C(F)C=C1Cl MOWXJLUYGFNTAL-DEOSSOPVSA-N 0.000 description 1
- FYWJWWMKCARWQG-UHFFFAOYSA-N 1,2-dichloro-3-isocyanatobenzene Chemical compound ClC1=CC=CC(N=C=O)=C1Cl FYWJWWMKCARWQG-UHFFFAOYSA-N 0.000 description 1
- OYLIHMYGWOJSRQ-UHFFFAOYSA-N 1,3-benzodioxol-4-ylurea Chemical compound NC(=O)NC1=CC=CC2=C1OCO2 OYLIHMYGWOJSRQ-UHFFFAOYSA-N 0.000 description 1
- FTNJQNQLEGKTGD-UHFFFAOYSA-N 1,3-benzodioxole Chemical compound C1=CC=C2OCOC2=C1 FTNJQNQLEGKTGD-UHFFFAOYSA-N 0.000 description 1
- YXHDLKWTPVMIOH-UHFFFAOYSA-N 1,3-difluoro-2-isocyanatobenzene Chemical compound FC1=CC=CC(F)=C1N=C=O YXHDLKWTPVMIOH-UHFFFAOYSA-N 0.000 description 1
- XPQRCZQIBHESRL-UHFFFAOYSA-N 1-(2,3-dichlorophenyl)-1-(2-hydroxy-4-propan-2-ylphenyl)urea Chemical compound OC1=C(C=CC(=C1)C(C)C)N(C(=O)N)C1=C(C(=CC=C1)Cl)Cl XPQRCZQIBHESRL-UHFFFAOYSA-N 0.000 description 1
- GANKNIPTHRSOST-UHFFFAOYSA-N 1-(2,3-dichlorophenyl)-3-(2-hydroxy-4-propan-2-ylphenyl)urea Chemical compound OC1=CC(C(C)C)=CC=C1NC(=O)NC1=CC=CC(Cl)=C1Cl GANKNIPTHRSOST-UHFFFAOYSA-N 0.000 description 1
- OXDGOLZNKIAUIL-UHFFFAOYSA-N 1-(2,3-dichlorophenyl)-3-(3-hydroxynaphthalen-2-yl)urea urea Chemical compound OC1=CC2=CC=CC=C2C=C1NC(=O)NC1=C(C(=CC=C1)Cl)Cl.NC(=O)N OXDGOLZNKIAUIL-UHFFFAOYSA-N 0.000 description 1
- NNURDMPNQHPAIX-UHFFFAOYSA-N 1-(2,3-dichlorophenyl)-3-(5-ethylsulfonyl-2-hydroxyphenyl)urea Chemical compound CCS(=O)(=O)C1=CC=C(O)C(NC(=O)NC=2C(=C(Cl)C=CC=2)Cl)=C1 NNURDMPNQHPAIX-UHFFFAOYSA-N 0.000 description 1
- GUOVHYMPDWCJRZ-UHFFFAOYSA-N 1-(2,3-dihydro-1,4-benzodioxin-5-yl)-3-(2-hydroxy-4-nitrophenyl)urea Chemical compound OC1=CC([N+]([O-])=O)=CC=C1NC(=O)NC1=CC=CC2=C1OCCO2 GUOVHYMPDWCJRZ-UHFFFAOYSA-N 0.000 description 1
- PDVMALXQVQNLMF-UHFFFAOYSA-N 1-(2-bromophenyl)-1-(2-hydroxy-4-methylphenyl)urea Chemical compound OC1=C(C=CC(=C1)C)N(C(=O)N)C1=C(C=CC=C1)Br PDVMALXQVQNLMF-UHFFFAOYSA-N 0.000 description 1
- COOHQYMORDKZIA-UHFFFAOYSA-N 1-(2-bromophenyl)-1-(3-cyano-2-hydroxy-4-methylphenyl)urea Chemical compound OC1=C(C#N)C(C)=CC=C1N(C(N)=O)C1=CC=CC=C1Br COOHQYMORDKZIA-UHFFFAOYSA-N 0.000 description 1
- ATKVMTPPFADENB-UHFFFAOYSA-N 1-(2-bromophenyl)-1-(3-fluoro-2-hydroxyphenyl)urea Chemical compound C=1C=CC(F)=C(O)C=1N(C(=O)N)C1=CC=CC=C1Br ATKVMTPPFADENB-UHFFFAOYSA-N 0.000 description 1
- RSZCNEGEBUDYSK-UHFFFAOYSA-N 1-(2-bromophenyl)-1-(4-ethyl-2-hydroxyphenyl)urea Chemical compound OC1=CC(CC)=CC=C1N(C(N)=O)C1=CC=CC=C1Br RSZCNEGEBUDYSK-UHFFFAOYSA-N 0.000 description 1
- SEBYEVXUHHITRG-UHFFFAOYSA-N 1-(2-bromophenyl)-3-(2-hydroxy-3-propylphenyl)urea Chemical compound CCCC1=CC=CC(NC(=O)NC=2C(=CC=CC=2)Br)=C1O SEBYEVXUHHITRG-UHFFFAOYSA-N 0.000 description 1
- YNISKJAWVRXTTD-UHFFFAOYSA-N 1-(2-bromophenyl)-3-(2-hydroxy-3-propylphenyl)urea Chemical compound CCCC1=CC=CC(NC(=O)NC=2C(=CC=CC=2)Br)=C1O.CCCC1=CC=CC(NC(=O)NC=2C(=CC=CC=2)Br)=C1O YNISKJAWVRXTTD-UHFFFAOYSA-N 0.000 description 1
- JLDCYZFMVUKXAN-UHFFFAOYSA-N 1-(2-bromophenyl)-3-(3,4-dichloro-2-hydroxyphenyl)urea Chemical compound OC1=C(Cl)C(Cl)=CC=C1NC(=O)NC1=CC=CC=C1Br.OC1=C(Cl)C(Cl)=CC=C1NC(=O)NC1=CC=CC=C1Br JLDCYZFMVUKXAN-UHFFFAOYSA-N 0.000 description 1
- BAJARMQMSIWURB-UHFFFAOYSA-N 1-(2-bromophenyl)-3-(3,5-dichloro-2-hydroxyphenyl)urea Chemical compound OC1=C(Cl)C=C(Cl)C=C1NC(=O)NC1=CC=CC=C1Br.OC1=C(Cl)C=C(Cl)C=C1NC(=O)NC1=CC=CC=C1Br BAJARMQMSIWURB-UHFFFAOYSA-N 0.000 description 1
- FWZQZACNPWDDLN-UHFFFAOYSA-N 1-(2-bromophenyl)-3-(3-chloro-2-hydroxyphenyl)urea Chemical compound OC1=C(Cl)C=CC=C1NC(=O)NC1=CC=CC=C1Br.OC1=C(Cl)C=CC=C1NC(=O)NC1=CC=CC=C1Br FWZQZACNPWDDLN-UHFFFAOYSA-N 0.000 description 1
- IXCFEOVJAXAJKK-UHFFFAOYSA-N 1-(2-bromophenyl)-3-(3-cyano-2-hydroxyphenyl)urea Chemical compound C1=CC=C(C#N)C(O)=C1NC(=O)NC1=CC=CC=C1Br.C1=CC=C(C#N)C(O)=C1NC(=O)NC1=CC=CC=C1Br IXCFEOVJAXAJKK-UHFFFAOYSA-N 0.000 description 1
- KYMGMLIJGMOCOG-UHFFFAOYSA-N 1-(2-bromophenyl)-3-(3-fluoro-2-hydroxyphenyl)urea Chemical compound OC1=C(F)C=CC=C1NC(=O)NC1=CC=CC=C1Br KYMGMLIJGMOCOG-UHFFFAOYSA-N 0.000 description 1
- QIVSQPCCOXYDOV-UHFFFAOYSA-N 1-(2-bromophenyl)-3-(3-fluoro-2-hydroxyphenyl)urea Chemical compound OC1=C(F)C=CC=C1NC(=O)NC1=CC=CC=C1Br.OC1=C(F)C=CC=C1NC(=O)NC1=CC=CC=C1Br QIVSQPCCOXYDOV-UHFFFAOYSA-N 0.000 description 1
- MGACCYGHGSFOQC-UHFFFAOYSA-N 1-(2-bromophenyl)-3-(3-hydroxy-9,10-dioxoanthracen-2-yl)urea Chemical compound OC1=CC=2C(=O)C3=CC=CC=C3C(=O)C=2C=C1NC(=O)NC1=CC=CC=C1Br MGACCYGHGSFOQC-UHFFFAOYSA-N 0.000 description 1
- NEWUZEKWAAQYFU-UHFFFAOYSA-N 1-(2-bromophenyl)-3-(4-cyano-2-hydroxyphenyl)urea Chemical compound OC1=CC(C#N)=CC=C1NC(=O)NC1=CC=CC=C1Br.OC1=CC(C#N)=CC=C1NC(=O)NC1=CC=CC=C1Br NEWUZEKWAAQYFU-UHFFFAOYSA-N 0.000 description 1
- QKEACRPDPSOQQG-UHFFFAOYSA-N 1-(2-bromophenyl)-3-(4-ethyl-2-hydroxyphenyl)urea Chemical compound OC1=CC(CC)=CC=C1NC(=O)NC1=CC=CC=C1Br.OC1=CC(CC)=CC=C1NC(=O)NC1=CC=CC=C1Br QKEACRPDPSOQQG-UHFFFAOYSA-N 0.000 description 1
- YUMCQMMDLZBCNR-UHFFFAOYSA-N 1-(2-bromophenyl)-3-(5-cyano-2-hydroxyphenyl)urea Chemical compound OC1=CC=C(C#N)C=C1NC(=O)NC1=CC=CC=C1Br.OC1=CC=C(C#N)C=C1NC(=O)NC1=CC=CC=C1Br YUMCQMMDLZBCNR-UHFFFAOYSA-N 0.000 description 1
- NVTONEJFDYCQFV-UHFFFAOYSA-N 1-(2-bromophenyl)-3-(5-fluoro-2-hydroxyphenyl)urea Chemical compound OC1=CC=C(F)C=C1NC(=O)NC1=CC=CC=C1Br.OC1=CC=C(F)C=C1NC(=O)NC1=CC=CC=C1Br NVTONEJFDYCQFV-UHFFFAOYSA-N 0.000 description 1
- MQKQFYVENRCNGH-UHFFFAOYSA-N 1-(2-bromophenyl)-3-[2-[(3-nitrophenyl)sulfonylamino]phenyl]urea Chemical compound [O-][N+](=O)C1=CC=CC(S(=O)(=O)NC=2C(=CC=CC=2)NC(=O)NC=2C(=CC=CC=2)Br)=C1 MQKQFYVENRCNGH-UHFFFAOYSA-N 0.000 description 1
- QDCRJFQIWPIXCV-UHFFFAOYSA-N 1-(2-bromophenyl)-3-[2-[(4-phenoxyphenyl)sulfonylamino]phenyl]urea Chemical compound BrC1=CC=CC=C1NC(=O)NC1=CC=CC=C1NS(=O)(=O)C(C=C1)=CC=C1OC1=CC=CC=C1 QDCRJFQIWPIXCV-UHFFFAOYSA-N 0.000 description 1
- RUBDEDUAMQGILI-UHFFFAOYSA-N 1-(2-ethylphenyl)-3-(2-hydroxy-4-nitrophenyl)urea Chemical compound CCC1=CC=CC=C1NC(=O)NC1=CC=C([N+]([O-])=O)C=C1O.CCC1=CC=CC=C1NC(=O)NC1=CC=C([N+]([O-])=O)C=C1O RUBDEDUAMQGILI-UHFFFAOYSA-N 0.000 description 1
- VJKCGAIAKWBLEZ-UHFFFAOYSA-N 1-(2-hydroxy-4-nitrophenyl)-3-(2-methoxyphenyl)urea Chemical compound COC1=CC=CC=C1NC(=O)NC1=CC=C([N+]([O-])=O)C=C1O.COC1=CC=CC=C1NC(=O)NC1=CC=C([N+]([O-])=O)C=C1O VJKCGAIAKWBLEZ-UHFFFAOYSA-N 0.000 description 1
- KJRHQZRZPHNIHJ-UHFFFAOYSA-N 1-(2-hydroxy-4-nitrophenyl)-3-(3-methoxyphenyl)urea Chemical compound COC1=CC=CC(NC(=O)NC=2C(=CC(=CC=2)[N+]([O-])=O)O)=C1.COC1=CC=CC(NC(=O)NC=2C(=CC(=CC=2)[N+]([O-])=O)O)=C1 KJRHQZRZPHNIHJ-UHFFFAOYSA-N 0.000 description 1
- CLRDVEMJNUPVTM-UHFFFAOYSA-N 1-(2-hydroxy-4-nitrophenyl)-3-(4-nitrophenyl)urea Chemical compound OC1=CC([N+]([O-])=O)=CC=C1NC(=O)NC1=CC=C([N+]([O-])=O)C=C1 CLRDVEMJNUPVTM-UHFFFAOYSA-N 0.000 description 1
- ZUYRHEBJDOSEEI-UHFFFAOYSA-N 1-(2-hydroxy-4-nitrophenyl)-3-[2-(trifluoromethoxy)phenyl]urea Chemical compound OC1=CC([N+]([O-])=O)=CC=C1NC(=O)NC1=CC=CC=C1OC(F)(F)F.OC1=CC([N+]([O-])=O)=CC=C1NC(=O)NC1=CC=CC=C1OC(F)(F)F ZUYRHEBJDOSEEI-UHFFFAOYSA-N 0.000 description 1
- YEYNQOFIQKNYSD-UHFFFAOYSA-N 1-(2-hydroxy-4-nitrophenyl)-3-[4-(trifluoromethyl)phenyl]urea Chemical compound OC1=CC([N+]([O-])=O)=CC=C1NC(=O)NC1=CC=C(C(F)(F)F)C=C1.OC1=CC([N+]([O-])=O)=CC=C1NC(=O)NC1=CC=C(C(F)(F)F)C=C1 YEYNQOFIQKNYSD-UHFFFAOYSA-N 0.000 description 1
- QOTXGIIIAWXWKX-UHFFFAOYSA-N 1-(2-hydroxy-4-nitrophenyl)-3-phenylthiourea Chemical compound OC1=CC([N+]([O-])=O)=CC=C1NC(=S)NC1=CC=CC=C1.OC1=CC([N+]([O-])=O)=CC=C1NC(=S)NC1=CC=CC=C1 QOTXGIIIAWXWKX-UHFFFAOYSA-N 0.000 description 1
- HSHAABFJOBPSBZ-UHFFFAOYSA-N 1-(2-hydroxy-5-nitrophenyl)-3-(2-methoxyphenyl)urea urea Chemical compound OC1=C(C=C(C=C1)[N+](=O)[O-])NC(=O)NC1=C(C=CC=C1)OC.NC(=O)N HSHAABFJOBPSBZ-UHFFFAOYSA-N 0.000 description 1
- SUVXETMUHQVVFC-UHFFFAOYSA-N 1-(2-hydroxy-5-nitrophenyl)-3-(2-phenylphenyl)urea Chemical compound OC1=CC=C([N+]([O-])=O)C=C1NC(=O)NC1=CC=CC=C1C1=CC=CC=C1 SUVXETMUHQVVFC-UHFFFAOYSA-N 0.000 description 1
- HKMPCIRBBJUVQZ-UHFFFAOYSA-N 1-(2-hydroxy-5-nitrophenyl)-3-(2-phenylphenyl)urea urea Chemical compound OC1=C(C=C(C=C1)[N+](=O)[O-])NC(=O)NC1=C(C=CC=C1)C1=CC=CC=C1.NC(=O)N HKMPCIRBBJUVQZ-UHFFFAOYSA-N 0.000 description 1
- ZSLIIGIYWQZBOQ-UHFFFAOYSA-N 1-(2-hydroxy-5-nitrophenyl)-3-[3-(trifluoromethyl)phenyl]urea Chemical compound OC1=CC=C([N+]([O-])=O)C=C1NC(=O)NC1=CC=CC(C(F)(F)F)=C1 ZSLIIGIYWQZBOQ-UHFFFAOYSA-N 0.000 description 1
- ZMFGYZRTZKXWGV-UHFFFAOYSA-N 1-(2-hydroxy-5-nitrophenyl)-3-[3-(trifluoromethyl)phenyl]urea urea Chemical compound OC1=C(C=C(C=C1)[N+](=O)[O-])NC(=O)NC1=CC(=CC=C1)C(F)(F)F.NC(=O)N ZMFGYZRTZKXWGV-UHFFFAOYSA-N 0.000 description 1
- CDTNLFCEAMKPDO-UHFFFAOYSA-N 1-(3,4-dichloro-2-hydroxyphenyl)-1-(4-methoxyphenyl)urea Chemical compound C1=CC(OC)=CC=C1N(C(N)=O)C1=CC=C(Cl)C(Cl)=C1O CDTNLFCEAMKPDO-UHFFFAOYSA-N 0.000 description 1
- MPHQBIQHINWCQF-UHFFFAOYSA-N 1-(3,4-dichloro-2-hydroxyphenyl)-3-(2,4-dimethoxyphenyl)urea Chemical compound COC1=CC(OC)=CC=C1NC(=O)NC1=CC=C(Cl)C(Cl)=C1O MPHQBIQHINWCQF-UHFFFAOYSA-N 0.000 description 1
- KSPGSKWILGYAFB-UHFFFAOYSA-N 1-(3,4-dichloro-2-hydroxyphenyl)-3-(4-methoxyphenyl)urea Chemical compound C1=CC(OC)=CC=C1NC(=O)NC1=CC=C(Cl)C(Cl)=C1O KSPGSKWILGYAFB-UHFFFAOYSA-N 0.000 description 1
- UAXAGWYQMOAGGL-UHFFFAOYSA-N 1-(3,4-dichloro-2-hydroxyphenyl)-3-[3-(trifluoromethyl)phenyl]urea Chemical compound OC1=C(Cl)C(Cl)=CC=C1NC(=O)NC1=CC=CC(C(F)(F)F)=C1 UAXAGWYQMOAGGL-UHFFFAOYSA-N 0.000 description 1
- WBNBOYLRUMMMIM-UHFFFAOYSA-N 1-(3,4-dichloro-2-hydroxyphenyl)-3-[3-(trifluoromethyl)phenyl]urea urea Chemical compound OC1=C(C=CC(=C1Cl)Cl)NC(=O)NC1=CC(=CC=C1)C(F)(F)F.NC(=O)N WBNBOYLRUMMMIM-UHFFFAOYSA-N 0.000 description 1
- AXZKKKPRCZGTBT-UHFFFAOYSA-N 1-(3-cyano-2-hydroxyphenyl)-3-(2-methoxyphenyl)urea urea Chemical compound OC1=C(C=CC=C1C#N)NC(=O)NC1=C(C=CC=C1)OC.NC(=O)N AXZKKKPRCZGTBT-UHFFFAOYSA-N 0.000 description 1
- UOUHGJIBGKXSHO-UHFFFAOYSA-N 1-(3-cyano-2-hydroxyphenyl)-3-(2-phenylphenyl)urea urea Chemical compound OC1=C(C=CC=C1C#N)NC(=O)NC1=C(C=CC=C1)C1=CC=CC=C1.NC(=O)N UOUHGJIBGKXSHO-UHFFFAOYSA-N 0.000 description 1
- ANAOKSOOHCYGTR-UHFFFAOYSA-N 1-(3-cyano-2-hydroxyphenyl)-3-[3-(trifluoromethyl)phenyl]urea Chemical compound C1=CC=C(C#N)C(O)=C1NC(=O)NC1=CC=CC(C(F)(F)F)=C1 ANAOKSOOHCYGTR-UHFFFAOYSA-N 0.000 description 1
- JHIQRZOXKYKKGJ-UHFFFAOYSA-N 1-(4-bromophenyl)-3-(2-hydroxy-4-nitrophenyl)urea Chemical compound OC1=CC([N+]([O-])=O)=CC=C1NC(=O)NC1=CC=C(Br)C=C1.OC1=CC([N+]([O-])=O)=CC=C1NC(=O)NC1=CC=C(Br)C=C1 JHIQRZOXKYKKGJ-UHFFFAOYSA-N 0.000 description 1
- ZZGNVSNBMGFDIE-UHFFFAOYSA-N 1-(4-cyano-2-hydroxyphenyl)-1-[3-(trifluoromethyl)phenyl]urea Chemical compound C=1C=C(C#N)C=C(O)C=1N(C(=O)N)C1=CC=CC(C(F)(F)F)=C1 ZZGNVSNBMGFDIE-UHFFFAOYSA-N 0.000 description 1
- KYVRTOOHPVNUPH-UHFFFAOYSA-N 1-(4-cyano-2-hydroxyphenyl)-3-(2,3-dichlorophenyl)urea urea Chemical compound OC1=C(C=CC(=C1)C#N)NC(=O)NC1=C(C(=CC=C1)Cl)Cl.NC(=O)N KYVRTOOHPVNUPH-UHFFFAOYSA-N 0.000 description 1
- FHYFXAPTPBQLDH-UHFFFAOYSA-N 1-(4-cyano-2-hydroxyphenyl)-3-(2-methylphenyl)urea urea Chemical compound OC1=C(C=CC(=C1)C#N)NC(=O)NC1=C(C=CC=C1)C.NC(=O)N FHYFXAPTPBQLDH-UHFFFAOYSA-N 0.000 description 1
- HVIZHGMWWZTGDS-UHFFFAOYSA-N 1-(4-cyano-2-hydroxyphenyl)-3-(2-phenylphenyl)urea urea Chemical compound OC1=C(C=CC(=C1)C#N)NC(=O)NC1=C(C=CC=C1)C1=CC=CC=C1.NC(=O)N HVIZHGMWWZTGDS-UHFFFAOYSA-N 0.000 description 1
- QIYGBMTTYPAFGZ-UHFFFAOYSA-N 1-(4-cyano-2-hydroxyphenyl)-3-(4-phenylphenyl)urea Chemical compound OC1=CC(C#N)=CC=C1NC(=O)NC1=CC=C(C=2C=CC=CC=2)C=C1 QIYGBMTTYPAFGZ-UHFFFAOYSA-N 0.000 description 1
- AAZOXZGQYYXRTC-UHFFFAOYSA-N 1-(4-cyano-2-hydroxyphenyl)-3-[2-(trifluoromethyl)phenyl]urea urea Chemical compound OC1=C(C=CC(=C1)C#N)NC(=O)NC1=C(C=CC=C1)C(F)(F)F.NC(=O)N AAZOXZGQYYXRTC-UHFFFAOYSA-N 0.000 description 1
- HGRXXAGRIICSQS-UHFFFAOYSA-N 1-(4-cyano-2-hydroxyphenyl)-3-[3-(trifluoromethyl)phenyl]urea Chemical compound OC1=CC(C#N)=CC=C1NC(=O)NC1=CC=CC(C(F)(F)F)=C1 HGRXXAGRIICSQS-UHFFFAOYSA-N 0.000 description 1
- QACGUVMJQUPIOD-UHFFFAOYSA-N 1-(4-fluoro-2-hydroxyphenyl)-1-phenylurea Chemical compound C=1C=C(F)C=C(O)C=1N(C(=O)N)C1=CC=CC=C1 QACGUVMJQUPIOD-UHFFFAOYSA-N 0.000 description 1
- HZWWPMXKFYNCEK-UHFFFAOYSA-N 1-(4-fluoro-2-hydroxyphenyl)-3-phenylurea Chemical compound OC1=CC(F)=CC=C1NC(=O)NC1=CC=CC=C1.OC1=CC(F)=CC=C1NC(=O)NC1=CC=CC=C1 HZWWPMXKFYNCEK-UHFFFAOYSA-N 0.000 description 1
- LUBJCRLGQSPQNN-UHFFFAOYSA-N 1-Phenylurea Chemical class NC(=O)NC1=CC=CC=C1 LUBJCRLGQSPQNN-UHFFFAOYSA-N 0.000 description 1
- ABDDQTDRAHXHOC-QMMMGPOBSA-N 1-[(7s)-5,7-dihydro-4h-thieno[2,3-c]pyran-7-yl]-n-methylmethanamine Chemical compound CNC[C@@H]1OCCC2=C1SC=C2 ABDDQTDRAHXHOC-QMMMGPOBSA-N 0.000 description 1
- JRMLXASCHVKDRW-UHFFFAOYSA-N 1-[2-chloro-5-(trifluoromethyl)phenyl]-1-(3,4-dichloro-2-hydroxyphenyl)urea Chemical compound C=1C(C(F)(F)F)=CC=C(Cl)C=1N(C(=O)N)C1=CC=C(Cl)C(Cl)=C1O JRMLXASCHVKDRW-UHFFFAOYSA-N 0.000 description 1
- QCPKCLJVLUZRHH-UHFFFAOYSA-N 1-[2-chloro-5-(trifluoromethyl)phenyl]-3-(3,4-dichloro-2-hydroxyphenyl)urea Chemical compound OC1=C(Cl)C(Cl)=CC=C1NC(=O)NC1=CC(C(F)(F)F)=CC=C1Cl QCPKCLJVLUZRHH-UHFFFAOYSA-N 0.000 description 1
- JOUCNJAIDGFKOE-UHFFFAOYSA-N 1-[3-amino-2-(2-sulfamoylphenyl)phenyl]-3-(2-bromophenyl)urea Chemical compound C=1C=CC=C(S(N)(=O)=O)C=1C=1C(N)=CC=CC=1NC(=O)NC1=CC=CC=C1Br JOUCNJAIDGFKOE-UHFFFAOYSA-N 0.000 description 1
- FHMMQQXRSYSWCM-UHFFFAOYSA-N 1-aminonaphthalen-2-ol Chemical compound C1=CC=C2C(N)=C(O)C=CC2=C1 FHMMQQXRSYSWCM-UHFFFAOYSA-N 0.000 description 1
- UMJGPXFGKALWKY-UHFFFAOYSA-N 1-benzyl-1-(3,4-dichloro-2-hydroxyphenyl)urea Chemical compound C=1C=C(Cl)C(Cl)=C(O)C=1N(C(=O)N)CC1=CC=CC=C1 UMJGPXFGKALWKY-UHFFFAOYSA-N 0.000 description 1
- NEFXLENDBNGLIG-UHFFFAOYSA-N 1-benzyl-3-(3-hydroxynaphthalen-2-yl)urea Chemical compound OC1=CC2=CC=CC=C2C=C1NC(=O)NCC1=CC=CC=C1 NEFXLENDBNGLIG-UHFFFAOYSA-N 0.000 description 1
- VQVBCZQTXSHJGF-UHFFFAOYSA-N 1-bromo-3-isocyanatobenzene Chemical compound BrC1=CC=CC(N=C=O)=C1 VQVBCZQTXSHJGF-UHFFFAOYSA-N 0.000 description 1
- CZQIJQFTRGDODI-UHFFFAOYSA-N 1-bromo-4-isocyanatobenzene Chemical compound BrC1=CC=C(N=C=O)C=C1 CZQIJQFTRGDODI-UHFFFAOYSA-N 0.000 description 1
- 125000004973 1-butenyl group Chemical group C(=CCC)* 0.000 description 1
- FBTQQNYGMICJQZ-UHFFFAOYSA-N 1-chloro-2-isocyanato-3-methylbenzene Chemical compound CC1=CC=CC(Cl)=C1N=C=O FBTQQNYGMICJQZ-UHFFFAOYSA-N 0.000 description 1
- NOHQUGRVHSJYMR-UHFFFAOYSA-N 1-chloro-2-isocyanatobenzene Chemical compound ClC1=CC=CC=C1N=C=O NOHQUGRVHSJYMR-UHFFFAOYSA-N 0.000 description 1
- BUIQXUQLYZPMLS-UHFFFAOYSA-N 1-ethoxy-2-isocyanatobenzene Chemical compound CCOC1=CC=CC=C1N=C=O BUIQXUQLYZPMLS-UHFFFAOYSA-N 0.000 description 1
- ZVFNUQWYLXXSJM-UHFFFAOYSA-N 1-ethyl-2-isocyanatobenzene Chemical compound CCC1=CC=CC=C1N=C=O ZVFNUQWYLXXSJM-UHFFFAOYSA-N 0.000 description 1
- VZNCSZQPNIEEMN-UHFFFAOYSA-N 1-fluoro-2-isocyanatobenzene Chemical compound FC1=CC=CC=C1N=C=O VZNCSZQPNIEEMN-UHFFFAOYSA-N 0.000 description 1
- LIKMAJRDDDTEIG-UHFFFAOYSA-N 1-hexene Chemical compound CCCCC=C LIKMAJRDDDTEIG-UHFFFAOYSA-N 0.000 description 1
- CTMGYQHKKIEXKF-UHFFFAOYSA-N 1-isocyanato-2-(trifluoromethoxy)benzene Chemical compound FC(F)(F)OC1=CC=CC=C1N=C=O CTMGYQHKKIEXKF-UHFFFAOYSA-N 0.000 description 1
- GZWGTVZRRFPVAS-UHFFFAOYSA-N 1-isocyanato-2-(trifluoromethyl)benzene Chemical compound FC(F)(F)C1=CC=CC=C1N=C=O GZWGTVZRRFPVAS-UHFFFAOYSA-N 0.000 description 1
- SUVCZZADQDCIEQ-UHFFFAOYSA-N 1-isocyanato-2-methoxybenzene Chemical compound COC1=CC=CC=C1N=C=O SUVCZZADQDCIEQ-UHFFFAOYSA-N 0.000 description 1
- WQXASSKJZYKJSI-UHFFFAOYSA-N 1-isocyanato-2-methylsulfanylbenzene Chemical compound CSC1=CC=CC=C1N=C=O WQXASSKJZYKJSI-UHFFFAOYSA-N 0.000 description 1
- JRVZITODZAQRQM-UHFFFAOYSA-N 1-isocyanato-2-nitrobenzene Chemical compound [O-][N+](=O)C1=CC=CC=C1N=C=O JRVZITODZAQRQM-UHFFFAOYSA-N 0.000 description 1
- SXJYSIBLFGQAND-UHFFFAOYSA-N 1-isocyanato-3-(trifluoromethyl)benzene Chemical compound FC(F)(F)C1=CC=CC(N=C=O)=C1 SXJYSIBLFGQAND-UHFFFAOYSA-N 0.000 description 1
- NPOVTGVGOBJZPY-UHFFFAOYSA-N 1-isocyanato-3-methoxybenzene Chemical compound COC1=CC=CC(N=C=O)=C1 NPOVTGVGOBJZPY-UHFFFAOYSA-N 0.000 description 1
- QZTWVDCKDWZCLV-UHFFFAOYSA-N 1-isocyanato-4-(trifluoromethyl)benzene Chemical compound FC(F)(F)C1=CC=C(N=C=O)C=C1 QZTWVDCKDWZCLV-UHFFFAOYSA-N 0.000 description 1
- BDQNKCYCTYYMAA-UHFFFAOYSA-N 1-isocyanatonaphthalene Chemical compound C1=CC=C2C(N=C=O)=CC=CC2=C1 BDQNKCYCTYYMAA-UHFFFAOYSA-N 0.000 description 1
- QAUYDNWQNRMCIU-UHFFFAOYSA-N 1-phenyl-3-(2-sulfanylphenyl)urea Chemical compound SC1=CC=CC=C1NC(=O)NC1=CC=CC=C1 QAUYDNWQNRMCIU-UHFFFAOYSA-N 0.000 description 1
- 125000006017 1-propenyl group Chemical group 0.000 description 1
- HYZJCKYKOHLVJF-UHFFFAOYSA-N 1H-benzimidazole Chemical compound C1=CC=C2NC=NC2=C1 HYZJCKYKOHLVJF-UHFFFAOYSA-N 0.000 description 1
- FEBNTTHBVXWGOJ-UHFFFAOYSA-N 2,3-bis(trifluoromethyl)benzenesulfonyl chloride Chemical compound FC(F)(F)C1=CC=CC(S(Cl)(=O)=O)=C1C(F)(F)F FEBNTTHBVXWGOJ-UHFFFAOYSA-N 0.000 description 1
- UMPSXRYVXUPCOS-UHFFFAOYSA-N 2,3-dichlorophenol Chemical compound OC1=CC=CC(Cl)=C1Cl UMPSXRYVXUPCOS-UHFFFAOYSA-N 0.000 description 1
- KEGOHDCHURMFKX-UHFFFAOYSA-N 2,3-difluoro-6-nitrophenol Chemical compound OC1=C(F)C(F)=CC=C1[N+]([O-])=O KEGOHDCHURMFKX-UHFFFAOYSA-N 0.000 description 1
- CGEZHWIPFKRZBF-UHFFFAOYSA-N 2,4-dibromo-1-isocyanatobenzene Chemical compound BrC1=CC=C(N=C=O)C(Br)=C1 CGEZHWIPFKRZBF-UHFFFAOYSA-N 0.000 description 1
- OLBJNSPBWLCTOT-UHFFFAOYSA-N 2,4-dichloro-1-isocyanatobenzene Chemical compound ClC1=CC=C(N=C=O)C(Cl)=C1 OLBJNSPBWLCTOT-UHFFFAOYSA-N 0.000 description 1
- ZIZGWNOAHUCACM-UHFFFAOYSA-N 2-(trifluoromethyl)benzenesulfonyl chloride Chemical compound FC(F)(F)C1=CC=CC=C1S(Cl)(=O)=O ZIZGWNOAHUCACM-UHFFFAOYSA-N 0.000 description 1
- ZOQOPXVJANRGJZ-UHFFFAOYSA-N 2-(trifluoromethyl)phenol Chemical compound OC1=CC=CC=C1C(F)(F)F ZOQOPXVJANRGJZ-UHFFFAOYSA-N 0.000 description 1
- HPIWUVVNMOAVQD-UHFFFAOYSA-N 2-[(2-methoxyphenyl)methylcarbamoylamino]benzoic acid Chemical compound COC1=CC=CC=C1CNC(=O)NC1=CC=CC=C1C(O)=O HPIWUVVNMOAVQD-UHFFFAOYSA-N 0.000 description 1
- IARYNIDODMTYSE-UHFFFAOYSA-N 2-[(3-fluorophenyl)carbamoylamino]benzoic acid Chemical compound OC(=O)C1=CC=CC=C1NC(=O)NC1=CC=CC(F)=C1 IARYNIDODMTYSE-UHFFFAOYSA-N 0.000 description 1
- GFSBFEFJWQVKFS-UHFFFAOYSA-N 2-[(4-chlorophenyl)carbamoylamino]benzoic acid Chemical compound OC(=O)C1=CC=CC=C1NC(=O)NC1=CC=C(Cl)C=C1 GFSBFEFJWQVKFS-UHFFFAOYSA-N 0.000 description 1
- NXGKPRKPUCSEIL-UHFFFAOYSA-N 2-acetamido-4-methyl-1,3-thiazole-5-sulfonyl chloride Chemical compound CC(=O)NC1=NC(C)=C(S(Cl)(=O)=O)S1 NXGKPRKPUCSEIL-UHFFFAOYSA-N 0.000 description 1
- BOBATFKNMFWLFG-UHFFFAOYSA-N 2-amino-2-cyano-n-methylacetamide Chemical compound CNC(=O)C(N)C#N BOBATFKNMFWLFG-UHFFFAOYSA-N 0.000 description 1
- NNJHZWICFMKUOE-UHFFFAOYSA-N 2-amino-3,4,6-trifluorophenol Chemical compound NC1=C(O)C(F)=CC(F)=C1F NNJHZWICFMKUOE-UHFFFAOYSA-N 0.000 description 1
- UPJVUFCLBYQKFH-UHFFFAOYSA-N 2-amino-4-ethylsulfonylphenol Chemical compound CCS(=O)(=O)C1=CC=C(O)C(N)=C1 UPJVUFCLBYQKFH-UHFFFAOYSA-N 0.000 description 1
- ULDFRPKVIZMKJG-UHFFFAOYSA-N 2-amino-4-fluorophenol Chemical compound NC1=CC(F)=CC=C1O ULDFRPKVIZMKJG-UHFFFAOYSA-N 0.000 description 1
- FZENJANBQJQMMF-UHFFFAOYSA-N 2-amino-5-(trifluoromethyl)phenol Chemical compound NC1=CC=C(C(F)(F)F)C=C1O.NC1=CC=C(C(F)(F)F)C=C1O FZENJANBQJQMMF-UHFFFAOYSA-N 0.000 description 1
- HCPJEHJGFKWRFM-UHFFFAOYSA-N 2-amino-5-methylphenol Chemical compound CC1=CC=C(N)C(O)=C1 HCPJEHJGFKWRFM-UHFFFAOYSA-N 0.000 description 1
- MDIAVSZFIQWYST-UHFFFAOYSA-N 2-amino-6-fluorophenol Chemical compound NC1=CC=CC(F)=C1O MDIAVSZFIQWYST-UHFFFAOYSA-N 0.000 description 1
- WKOJOGYKEWTXHX-UHFFFAOYSA-N 2-amino-6-iodophenol Chemical compound NC1=CC=CC(I)=C1O WKOJOGYKEWTXHX-UHFFFAOYSA-N 0.000 description 1
- AACMNEWXGKOJJK-UHFFFAOYSA-N 2-amino-6-nitrophenol Chemical compound NC1=CC=CC([N+]([O-])=O)=C1O AACMNEWXGKOJJK-UHFFFAOYSA-N 0.000 description 1
- OOHJKDQCYDJZJX-UHFFFAOYSA-N 2-amino-6-phenylmethoxyphenol Chemical compound NC1=CC=CC(OCC=2C=CC=CC=2)=C1O OOHJKDQCYDJZJX-UHFFFAOYSA-N 0.000 description 1
- UHGULLIUJBCTEF-UHFFFAOYSA-N 2-aminobenzothiazole Chemical compound C1=CC=C2SC(N)=NC2=C1 UHGULLIUJBCTEF-UHFFFAOYSA-N 0.000 description 1
- RMNCOQJSXAJRRL-UHFFFAOYSA-N 2-bromo-3-methyl-6-nitrophenol;2-hydroxy-6-methyl-3-nitrobenzonitrile Chemical compound CC1=CC=C([N+]([O-])=O)C(O)=C1Br.CC1=CC=C([N+]([O-])=O)C(O)=C1C#N RMNCOQJSXAJRRL-UHFFFAOYSA-N 0.000 description 1
- AOPBDRUWRLBSDB-UHFFFAOYSA-N 2-bromoaniline Chemical compound NC1=CC=CC=C1Br AOPBDRUWRLBSDB-UHFFFAOYSA-N 0.000 description 1
- 125000004974 2-butenyl group Chemical group C(C=CC)* 0.000 description 1
- YEDUAINPPJYDJZ-UHFFFAOYSA-N 2-hydroxybenzothiazole Chemical compound C1=CC=C2SC(O)=NC2=C1 YEDUAINPPJYDJZ-UHFFFAOYSA-N 0.000 description 1
- CJNZAXGUTKBIHP-UHFFFAOYSA-N 2-iodobenzoic acid Chemical compound OC(=O)C1=CC=CC=C1I CJNZAXGUTKBIHP-UHFFFAOYSA-N 0.000 description 1
- HDECRAPHCDXMIJ-UHFFFAOYSA-N 2-methylbenzenesulfonyl chloride Chemical compound CC1=CC=CC=C1S(Cl)(=O)=O HDECRAPHCDXMIJ-UHFFFAOYSA-N 0.000 description 1
- NFCPRRWCTNLGSN-UHFFFAOYSA-N 2-n-phenylbenzene-1,2-diamine Chemical compound NC1=CC=CC=C1NC1=CC=CC=C1 NFCPRRWCTNLGSN-UHFFFAOYSA-N 0.000 description 1
- ATXBGHLILIABGX-UHFFFAOYSA-N 2-nitro-4-(trifluoromethyl)aniline Chemical compound NC1=CC=C(C(F)(F)F)C=C1[N+]([O-])=O ATXBGHLILIABGX-UHFFFAOYSA-N 0.000 description 1
- XZEDEVRSUANQEM-UHFFFAOYSA-N 2-nitro-4-(trifluoromethyl)phenol Chemical compound OC1=CC=C(C(F)(F)F)C=C1[N+]([O-])=O XZEDEVRSUANQEM-UHFFFAOYSA-N 0.000 description 1
- ONMNPYAHERZCCC-UHFFFAOYSA-N 2-nitro-5-(trifluoromethyl)phenol Chemical compound OC1=CC(C(F)(F)F)=CC=C1[N+]([O-])=O.OC1=CC(C(F)(F)F)=CC=C1[N+]([O-])=O ONMNPYAHERZCCC-UHFFFAOYSA-N 0.000 description 1
- QMBIPOMGUFPDHY-UHFFFAOYSA-N 2-nitro-6-phenylmethoxyphenol;2-phenylmethoxyphenol Chemical compound OC1=CC=CC=C1OCC1=CC=CC=C1.C1=CC=C([N+]([O-])=O)C(O)=C1OCC1=CC=CC=C1 QMBIPOMGUFPDHY-UHFFFAOYSA-N 0.000 description 1
- SNIBPLNVBYUZKZ-UHFFFAOYSA-N 2-nitro-6-propylphenol Chemical compound CCCC1=CC=CC([N+]([O-])=O)=C1O SNIBPLNVBYUZKZ-UHFFFAOYSA-N 0.000 description 1
- PKRSYEPBQPFNRB-UHFFFAOYSA-N 2-phenoxybenzoic acid Chemical compound OC(=O)C1=CC=CC=C1OC1=CC=CC=C1 PKRSYEPBQPFNRB-UHFFFAOYSA-N 0.000 description 1
- 125000000094 2-phenylethyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])C([H])([H])* 0.000 description 1
- 229940061334 2-phenylphenol Drugs 0.000 description 1
- 125000003903 2-propenyl group Chemical group [H]C([*])([H])C([H])=C([H])[H] 0.000 description 1
- VAYMIYBJLRRIFR-UHFFFAOYSA-N 2-tolyl isocyanate Chemical compound CC1=CC=CC=C1N=C=O VAYMIYBJLRRIFR-UHFFFAOYSA-N 0.000 description 1
- RSJSYCZYQNJQPY-UHFFFAOYSA-N 3,4-dimethoxybenzenesulfonyl chloride Chemical compound COC1=CC=C(S(Cl)(=O)=O)C=C1OC RSJSYCZYQNJQPY-UHFFFAOYSA-N 0.000 description 1
- HCDMJFOHIXMBOV-UHFFFAOYSA-N 3-(2,6-difluoro-3,5-dimethoxyphenyl)-1-ethyl-8-(morpholin-4-ylmethyl)-4,7-dihydropyrrolo[4,5]pyrido[1,2-d]pyrimidin-2-one Chemical compound C=1C2=C3N(CC)C(=O)N(C=4C(=C(OC)C=C(OC)C=4F)F)CC3=CN=C2NC=1CN1CCOCC1 HCDMJFOHIXMBOV-UHFFFAOYSA-N 0.000 description 1
- GMLPYACMPUSEGC-UHFFFAOYSA-N 3-(2-hydroxy-3-nitrophenyl)prop-2-enoic acid;3-(2-hydroxyphenyl)prop-2-enoic acid Chemical compound OC(=O)C=CC1=CC=CC=C1O.OC(=O)C=CC1=CC=CC([N+]([O-])=O)=C1O GMLPYACMPUSEGC-UHFFFAOYSA-N 0.000 description 1
- RKMOQMFLVWHPHK-UHFFFAOYSA-N 3-(3-hydroxy-4-nitrophenyl)prop-2-enoic acid;3-(3-hydroxyphenyl)prop-2-enoic acid Chemical compound OC(=O)C=CC1=CC=CC(O)=C1.OC(=O)C=CC1=CC=C([N+]([O-])=O)C(O)=C1 RKMOQMFLVWHPHK-UHFFFAOYSA-N 0.000 description 1
- BYHQTRFJOGIQAO-GOSISDBHSA-N 3-(4-bromophenyl)-8-[(2R)-2-hydroxypropyl]-1-[(3-methoxyphenyl)methyl]-1,3,8-triazaspiro[4.5]decan-2-one Chemical compound C[C@H](CN1CCC2(CC1)CN(C(=O)N2CC3=CC(=CC=C3)OC)C4=CC=C(C=C4)Br)O BYHQTRFJOGIQAO-GOSISDBHSA-N 0.000 description 1
- CGZWSGLCVUWYAT-UHFFFAOYSA-N 3-(benzoylcarbamoylamino)-2-hydroxy-n-phenylbenzamide Chemical compound C1=CC=C(C(=O)NC=2C=CC=CC=2)C(O)=C1NC(=O)NC(=O)C1=CC=CC=C1 CGZWSGLCVUWYAT-UHFFFAOYSA-N 0.000 description 1
- UGEJOEBBMPOJMT-UHFFFAOYSA-N 3-(trifluoromethyl)phenol Chemical compound OC1=CC=CC(C(F)(F)F)=C1 UGEJOEBBMPOJMT-UHFFFAOYSA-N 0.000 description 1
- IQGMRVWUTCYCST-UHFFFAOYSA-N 3-Aminosalicylic acid Chemical compound NC1=CC=CC(C(O)=O)=C1O IQGMRVWUTCYCST-UHFFFAOYSA-N 0.000 description 1
- WNEODWDFDXWOLU-QHCPKHFHSA-N 3-[3-(hydroxymethyl)-4-[1-methyl-5-[[5-[(2s)-2-methyl-4-(oxetan-3-yl)piperazin-1-yl]pyridin-2-yl]amino]-6-oxopyridin-3-yl]pyridin-2-yl]-7,7-dimethyl-1,2,6,8-tetrahydrocyclopenta[3,4]pyrrolo[3,5-b]pyrazin-4-one Chemical compound C([C@@H](N(CC1)C=2C=NC(NC=3C(N(C)C=C(C=3)C=3C(=C(N4C(C5=CC=6CC(C)(C)CC=6N5CC4)=O)N=CC=3)CO)=O)=CC=2)C)N1C1COC1 WNEODWDFDXWOLU-QHCPKHFHSA-N 0.000 description 1
- WLKWVVMGNZMCQY-UHFFFAOYSA-N 3-[3-(hydroxymethyl)-4-nitrophenyl]prop-2-enoic acid 3-(3-hydroxy-4-nitrophenyl)prop-2-enoic acid Chemical compound [N+](=O)([O-])C1=C(C=C(C=CC(=O)O)C=C1)O.[N+](=O)([O-])C1=C(C=C(C=CC(=O)O)C=C1)CO WLKWVVMGNZMCQY-UHFFFAOYSA-N 0.000 description 1
- NJXPYZHXZZCTNI-UHFFFAOYSA-N 3-aminobenzonitrile Chemical compound NC1=CC=CC(C#N)=C1 NJXPYZHXZZCTNI-UHFFFAOYSA-N 0.000 description 1
- BMYNFMYTOJXKLE-UHFFFAOYSA-N 3-azaniumyl-2-hydroxypropanoate Chemical compound NCC(O)C(O)=O BMYNFMYTOJXKLE-UHFFFAOYSA-N 0.000 description 1
- DFMDAJMTLJGKFW-UHFFFAOYSA-N 3-chloro-2-nitrophenol Chemical compound OC1=CC=CC(Cl)=C1[N+]([O-])=O DFMDAJMTLJGKFW-UHFFFAOYSA-N 0.000 description 1
- GAWNBKUTBVLIPL-UHFFFAOYSA-N 3-fluoro-2-nitrophenol Chemical compound OC1=CC=CC(F)=C1[N+]([O-])=O GAWNBKUTBVLIPL-UHFFFAOYSA-N 0.000 description 1
- JELNXWUZFOQXGZ-UHFFFAOYSA-N 3-hydroxy-2-nitrobenzonitrile Chemical compound OC1=CC=CC(C#N)=C1[N+]([O-])=O JELNXWUZFOQXGZ-UHFFFAOYSA-N 0.000 description 1
- FWHZNBKCQVKRSZ-UHFFFAOYSA-N 3-hydroxy-4-(phenylcarbamoylamino)benzamide Chemical compound OC1=CC(C(=O)N)=CC=C1NC(=O)NC1=CC=CC=C1 FWHZNBKCQVKRSZ-UHFFFAOYSA-N 0.000 description 1
- MCSXGCZMEPXKIW-UHFFFAOYSA-N 3-hydroxy-4-[(4-methyl-2-nitrophenyl)diazenyl]-N-(3-nitrophenyl)naphthalene-2-carboxamide Chemical compound Cc1ccc(N=Nc2c(O)c(cc3ccccc23)C(=O)Nc2cccc(c2)[N+]([O-])=O)c(c1)[N+]([O-])=O MCSXGCZMEPXKIW-UHFFFAOYSA-N 0.000 description 1
- YMIKCZUFAPAVLQ-UHFFFAOYSA-N 3-hydroxybenzonitrile;3-hydroxy-4-nitrobenzonitrile Chemical compound OC1=CC=CC(C#N)=C1.OC1=CC(C#N)=CC=C1[N+]([O-])=O YMIKCZUFAPAVLQ-UHFFFAOYSA-N 0.000 description 1
- RFSKGCVUDQRZSD-UHFFFAOYSA-N 3-methoxythiophene Chemical compound COC=1C=CSC=1 RFSKGCVUDQRZSD-UHFFFAOYSA-N 0.000 description 1
- KFPMLWUKHQMEBU-UHFFFAOYSA-N 3-methylbenzenesulfonyl chloride Chemical compound CC1=CC=CC(S(Cl)(=O)=O)=C1 KFPMLWUKHQMEBU-UHFFFAOYSA-N 0.000 description 1
- MWWNNNAOGWPTQY-UHFFFAOYSA-N 3-nitrobenzenesulfonyl chloride Chemical compound [O-][N+](=O)C1=CC=CC(S(Cl)(=O)=O)=C1 MWWNNNAOGWPTQY-UHFFFAOYSA-N 0.000 description 1
- IVTWLTRKVRJPNG-UHFFFAOYSA-N 4,5-dichlorothiophene-2-sulfonyl chloride Chemical compound ClC=1C=C(S(Cl)(=O)=O)SC=1Cl IVTWLTRKVRJPNG-UHFFFAOYSA-N 0.000 description 1
- OZDCZHDOIBUGAJ-UHFFFAOYSA-N 4-(trifluoromethyl)benzenesulfonyl chloride Chemical compound FC(F)(F)C1=CC=C(S(Cl)(=O)=O)C=C1 OZDCZHDOIBUGAJ-UHFFFAOYSA-N 0.000 description 1
- SIPAGAIYBSRGJB-UHFFFAOYSA-N 4-[(2-bromophenyl)carbamoylamino]-3-hydroxybenzamide Chemical compound OC1=CC(C(=O)N)=CC=C1NC(=O)NC1=CC=CC=C1Br SIPAGAIYBSRGJB-UHFFFAOYSA-N 0.000 description 1
- HRQIIMWVCQJCIY-UHFFFAOYSA-N 4-[(2-bromophenyl)carbamoylamino]-3-hydroxybenzamide urea Chemical compound NC(=O)C1=CC(=C(C=C1)NC(=O)NC1=C(C=CC=C1)Br)O.NC(=O)N HRQIIMWVCQJCIY-UHFFFAOYSA-N 0.000 description 1
- MDQZQLIOAULXOM-UHFFFAOYSA-N 4-[(2-bromophenyl)carbamoylamino]-3-hydroxybenzamide;1-(2-bromophenyl)-3-(2,3,5-trifluoro-6-hydroxyphenyl)urea Chemical compound OC1=CC(C(=O)N)=CC=C1NC(=O)NC1=CC=CC=C1Br.OC1=C(F)C=C(F)C(F)=C1NC(=O)NC1=CC=CC=C1Br MDQZQLIOAULXOM-UHFFFAOYSA-N 0.000 description 1
- CKBWXMJWNJRKTI-UHFFFAOYSA-N 4-[3-[(2-bromophenyl)carbamoylamino]-2-hydroxyphenyl]benzoic acid Chemical compound C1=CC(C(=O)O)=CC=C1C1=CC=CC(NC(=O)NC=2C(=CC=CC=2)Br)=C1O.C1=CC(C(=O)O)=CC=C1C1=CC=CC(NC(=O)NC=2C(=CC=CC=2)Br)=C1O CKBWXMJWNJRKTI-UHFFFAOYSA-N 0.000 description 1
- GRDXCFKBQWDAJH-UHFFFAOYSA-N 4-acetamidobenzenesulfonyl chloride Chemical compound CC(=O)NC1=CC=C(S(Cl)(=O)=O)C=C1 GRDXCFKBQWDAJH-UHFFFAOYSA-N 0.000 description 1
- GLJFZIKVMDSXMY-UHFFFAOYSA-N 4-amino-3-hydroxybenzamide Chemical compound NC(=O)C1=CC=C(N)C(O)=C1 GLJFZIKVMDSXMY-UHFFFAOYSA-N 0.000 description 1
- NFPYJDZQOKCYIE-UHFFFAOYSA-N 4-amino-3-hydroxybenzoic acid Chemical compound NC1=CC=C(C(O)=O)C=C1O NFPYJDZQOKCYIE-UHFFFAOYSA-N 0.000 description 1
- KVCQTKNUUQOELD-UHFFFAOYSA-N 4-amino-n-[1-(3-chloro-2-fluoroanilino)-6-methylisoquinolin-5-yl]thieno[3,2-d]pyrimidine-7-carboxamide Chemical compound N=1C=CC2=C(NC(=O)C=3C4=NC=NC(N)=C4SC=3)C(C)=CC=C2C=1NC1=CC=CC(Cl)=C1F KVCQTKNUUQOELD-UHFFFAOYSA-N 0.000 description 1
- ACQVEWFMUBXEMR-UHFFFAOYSA-N 4-bromo-2-fluoro-6-nitrophenol Chemical compound OC1=C(F)C=C(Br)C=C1[N+]([O-])=O ACQVEWFMUBXEMR-UHFFFAOYSA-N 0.000 description 1
- DNYRLBZBDGVLHF-UHFFFAOYSA-N 4-bromo-5-methyl-2-nitrophenol Chemical compound CC1=CC(O)=C([N+]([O-])=O)C=C1Br DNYRLBZBDGVLHF-UHFFFAOYSA-N 0.000 description 1
- WDFQBORIUYODSI-IDEBNGHGSA-N 4-bromoaniline Chemical compound N[13C]1=[13CH][13CH]=[13C](Br)[13CH]=[13CH]1 WDFQBORIUYODSI-IDEBNGHGSA-N 0.000 description 1
- ZHRLVDHMIJDWSS-UHFFFAOYSA-N 4-fluoro-2-nitrophenol Chemical compound OC1=CC=C(F)C=C1[N+]([O-])=O ZHRLVDHMIJDWSS-UHFFFAOYSA-N 0.000 description 1
- INBLGVOPOSGVTA-UHFFFAOYSA-N 4-hydroxy-3-nitrobenzonitrile Chemical compound OC1=CC=C(C#N)C=C1[N+]([O-])=O INBLGVOPOSGVTA-UHFFFAOYSA-N 0.000 description 1
- RAUWPNXIALNKQM-UHFFFAOYSA-N 4-nitro-1,2-phenylenediamine Chemical compound NC1=CC=C([N+]([O-])=O)C=C1N RAUWPNXIALNKQM-UHFFFAOYSA-N 0.000 description 1
- QIZPONOMFWAPRR-UHFFFAOYSA-N 4-phenoxybenzenesulfonyl chloride Chemical compound C1=CC(S(=O)(=O)Cl)=CC=C1OC1=CC=CC=C1 QIZPONOMFWAPRR-UHFFFAOYSA-N 0.000 description 1
- FVKYLHATVWKUMW-UHFFFAOYSA-N 402-17-5 Chemical compound OC1=CC(C(F)(F)F)=CC=C1[N+]([O-])=O FVKYLHATVWKUMW-UHFFFAOYSA-N 0.000 description 1
- NWRLEWDBRMESLI-UHFFFAOYSA-N 5-hydroxy-2-(phenylcarbamoylamino)benzoic acid Chemical compound OC(=O)C1=CC(O)=CC=C1NC(=O)NC1=CC=CC=C1 NWRLEWDBRMESLI-UHFFFAOYSA-N 0.000 description 1
- KCBWAFJCKVKYHO-UHFFFAOYSA-N 6-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-1-[[4-[1-propan-2-yl-4-(trifluoromethyl)imidazol-2-yl]phenyl]methyl]pyrazolo[3,4-d]pyrimidine Chemical compound C1(CC1)C1=NC=NC(=C1C1=NC=C2C(=N1)N(N=C2)CC1=CC=C(C=C1)C=1N(C=C(N=1)C(F)(F)F)C(C)C)OC KCBWAFJCKVKYHO-UHFFFAOYSA-N 0.000 description 1
- JBMHSKGUMDJIIU-UHFFFAOYSA-N 6-amino-2,3-diphenylphenol Chemical compound C=1C=CC=CC=1C1=C(O)C(N)=CC=C1C1=CC=CC=C1 JBMHSKGUMDJIIU-UHFFFAOYSA-N 0.000 description 1
- LIHORUAALZXMLT-UHFFFAOYSA-N 6-amino-2-fluoro-3-(trifluoromethyl)phenol Chemical compound NC1=CC=C(C(F)(F)F)C(F)=C1O LIHORUAALZXMLT-UHFFFAOYSA-N 0.000 description 1
- MVGYYGCFVPMJAQ-UHFFFAOYSA-N 6-amino-7-hydroxynaphthalene-2-sulfonic acid Chemical compound C1=C(S(O)(=O)=O)C=C2C=C(O)C(N)=CC2=C1 MVGYYGCFVPMJAQ-UHFFFAOYSA-N 0.000 description 1
- LYPMXMBQPXPNIQ-UHFFFAOYSA-N 609-89-2 Chemical compound OC1=C(Cl)C=C(Cl)C=C1[N+]([O-])=O LYPMXMBQPXPNIQ-UHFFFAOYSA-N 0.000 description 1
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical compound [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 1
- CYJRNFFLTBEQSQ-UHFFFAOYSA-N 8-(3-methyl-1-benzothiophen-5-yl)-N-(4-methylsulfonylpyridin-3-yl)quinoxalin-6-amine Chemical compound CS(=O)(=O)C1=C(C=NC=C1)NC=1C=C2N=CC=NC2=C(C=1)C=1C=CC2=C(C(=CS2)C)C=1 CYJRNFFLTBEQSQ-UHFFFAOYSA-N 0.000 description 1
- NLXLAEXVIDQMFP-UHFFFAOYSA-N Ammonia chloride Chemical class [NH4+].[Cl-] NLXLAEXVIDQMFP-UHFFFAOYSA-N 0.000 description 1
- QGZKDVFQNNGYKY-UHFFFAOYSA-O Ammonium Chemical compound [NH4+] QGZKDVFQNNGYKY-UHFFFAOYSA-O 0.000 description 1
- USFZMSVCRYTOJT-UHFFFAOYSA-N Ammonium acetate Chemical class N.CC(O)=O USFZMSVCRYTOJT-UHFFFAOYSA-N 0.000 description 1
- 206010003645 Atopy Diseases 0.000 description 1
- 239000005711 Benzoic acid Substances 0.000 description 1
- LSNNMFCWUKXFEE-UHFFFAOYSA-M Bisulfite Chemical compound OS([O-])=O LSNNMFCWUKXFEE-UHFFFAOYSA-M 0.000 description 1
- 101001069913 Bos taurus Growth-regulated protein homolog beta Proteins 0.000 description 1
- 101001069912 Bos taurus Growth-regulated protein homolog gamma Proteins 0.000 description 1
- FUZKSCLFHRBHJG-UHFFFAOYSA-N BrC1=C(C=CC=C1)NC(N)=O.NC(=O)N Chemical compound BrC1=C(C=CC=C1)NC(N)=O.NC(=O)N FUZKSCLFHRBHJG-UHFFFAOYSA-N 0.000 description 1
- BRVONJGWHOVUHW-UHFFFAOYSA-N C(CC)C1=C(C=CC=C1)O.[N+](=O)([O-])C1=C(C(=CC=C1)CCC)O Chemical compound C(CC)C1=C(C=CC=C1)O.[N+](=O)([O-])C1=C(C(=CC=C1)CCC)O BRVONJGWHOVUHW-UHFFFAOYSA-N 0.000 description 1
- 102100036189 C-X-C motif chemokine 3 Human genes 0.000 description 1
- 108050006947 CXC Chemokine Proteins 0.000 description 1
- 102000019388 CXC chemokine Human genes 0.000 description 1
- 101100289995 Caenorhabditis elegans mac-1 gene Proteins 0.000 description 1
- 238000006969 Curtius rearrangement reaction Methods 0.000 description 1
- FEWJPZIEWOKRBE-JCYAYHJZSA-N Dextrotartaric acid Chemical compound OC(=O)[C@H](O)[C@@H](O)C(O)=O FEWJPZIEWOKRBE-JCYAYHJZSA-N 0.000 description 1
- 241000709661 Enterovirus Species 0.000 description 1
- 108091006027 G proteins Proteins 0.000 description 1
- 102000030782 GTP binding Human genes 0.000 description 1
- 108091000058 GTP-Binding Proteins 0.000 description 1
- 208000009889 Herpes Simplex Diseases 0.000 description 1
- 241000282412 Homo Species 0.000 description 1
- 101000947193 Homo sapiens C-X-C motif chemokine 3 Proteins 0.000 description 1
- 101000818522 Homo sapiens fMet-Leu-Phe receptor Proteins 0.000 description 1
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 1
- WRYCSMQKUKOKBP-UHFFFAOYSA-N Imidazolidine Chemical compound C1CNCN1 WRYCSMQKUKOKBP-UHFFFAOYSA-N 0.000 description 1
- 241001465754 Metazoa Species 0.000 description 1
- AFVFQIVMOAPDHO-UHFFFAOYSA-N Methanesulfonic acid Chemical compound CS(O)(=O)=O AFVFQIVMOAPDHO-UHFFFAOYSA-N 0.000 description 1
- AYCPARAPKDAOEN-LJQANCHMSA-N N-[(1S)-2-(dimethylamino)-1-phenylethyl]-6,6-dimethyl-3-[(2-methyl-4-thieno[3,2-d]pyrimidinyl)amino]-1,4-dihydropyrrolo[3,4-c]pyrazole-5-carboxamide Chemical compound C1([C@H](NC(=O)N2C(C=3NN=C(NC=4C=5SC=CC=5N=C(C)N=4)C=3C2)(C)C)CN(C)C)=CC=CC=C1 AYCPARAPKDAOEN-LJQANCHMSA-N 0.000 description 1
- 229910002651 NO3 Inorganic materials 0.000 description 1
- 229910003844 NSO2 Inorganic materials 0.000 description 1
- 206010028980 Neoplasm Diseases 0.000 description 1
- 206010029113 Neovascularisation Diseases 0.000 description 1
- FBDGAQGDJSFZSU-UHFFFAOYSA-N OC1=C(C=C(C=C1)S(=O)(=O)CC)NC(=O)NC1=C(C(=CC=C1)Cl)Cl.NC(=O)N Chemical compound OC1=C(C=C(C=C1)S(=O)(=O)CC)NC(=O)NC1=C(C(=CC=C1)Cl)Cl.NC(=O)N FBDGAQGDJSFZSU-UHFFFAOYSA-N 0.000 description 1
- DAULLLJPSWEKCG-UHFFFAOYSA-N OC1=C(C=C(C=C1)[N+](=O)[O-])NC(=O)NC1=C(C(=CC=C1)Cl)Cl.NC(=O)N Chemical compound OC1=C(C=C(C=C1)[N+](=O)[O-])NC(=O)NC1=C(C(=CC=C1)Cl)Cl.NC(=O)N DAULLLJPSWEKCG-UHFFFAOYSA-N 0.000 description 1
- QNCLNCQKWKPAHZ-UHFFFAOYSA-N OC1=C(C=CC(=C1)C#N)NC(=O)NC1=CC(=CC=C1)C(F)(F)F.NC(=O)N Chemical compound OC1=C(C=CC(=C1)C#N)NC(=O)NC1=CC(=CC=C1)C(F)(F)F.NC(=O)N QNCLNCQKWKPAHZ-UHFFFAOYSA-N 0.000 description 1
- PZHOEXPXKLTJHU-UHFFFAOYSA-N OC1=C(C=CC(=C1)C#N)NC(=O)NC1=CC(=CC=C1)OC.NC(=O)N Chemical compound OC1=C(C=CC(=C1)C#N)NC(=O)NC1=CC(=CC=C1)OC.NC(=O)N PZHOEXPXKLTJHU-UHFFFAOYSA-N 0.000 description 1
- RRFUVPCQVVSFCW-UHFFFAOYSA-N OC1=C(C=CC(=C1)C#N)NC(=O)NC1=CC=C(C=C1)C(F)(F)F.NC(=O)N Chemical compound OC1=C(C=CC(=C1)C#N)NC(=O)NC1=CC=C(C=C1)C(F)(F)F.NC(=O)N RRFUVPCQVVSFCW-UHFFFAOYSA-N 0.000 description 1
- QQASHPOWEWGVHT-UHFFFAOYSA-N OC1=C(C=CC(=C1)C#N)NC(=O)NC1=CC=C(C=C1)C1=CC=CC=C1.NC(=O)N Chemical compound OC1=C(C=CC(=C1)C#N)NC(=O)NC1=CC=C(C=C1)C1=CC=CC=C1.NC(=O)N QQASHPOWEWGVHT-UHFFFAOYSA-N 0.000 description 1
- JTSQDTDJWPDWDJ-UHFFFAOYSA-N OC1=C(C=CC(=C1)C(C)C)NC(=O)NC1=C(C(=CC=C1)Cl)Cl.NC(=O)N Chemical compound OC1=C(C=CC(=C1)C(C)C)NC(=O)NC1=C(C(=CC=C1)Cl)Cl.NC(=O)N JTSQDTDJWPDWDJ-UHFFFAOYSA-N 0.000 description 1
- YHBOBBKIYMJBQP-UHFFFAOYSA-N OC1=C(C=CC(=C1)C(C)C)NC(=O)NC1=C(C=CC(=C1)C(F)(F)F)Cl.NC(=O)N Chemical compound OC1=C(C=CC(=C1)C(C)C)NC(=O)NC1=C(C=CC(=C1)C(F)(F)F)Cl.NC(=O)N YHBOBBKIYMJBQP-UHFFFAOYSA-N 0.000 description 1
- JBZAVRFVWOEFNF-UHFFFAOYSA-N OC1=C(C=CC(=C1)[N+](=O)[O-])NC(=O)NC1=C(C=CC=C1)OCC.OC1=C(C=CC(=C1)[N+](=O)[O-])NC(=O)NC1=C(C=CC=C1)OCC Chemical compound OC1=C(C=CC(=C1)[N+](=O)[O-])NC(=O)NC1=C(C=CC=C1)OCC.OC1=C(C=CC(=C1)[N+](=O)[O-])NC(=O)NC1=C(C=CC=C1)OCC JBZAVRFVWOEFNF-UHFFFAOYSA-N 0.000 description 1
- YKSZIRMKYDFEGV-UHFFFAOYSA-N OC1=C(C=CC(=C1)[N+](=O)[O-])NC(=O)NC1=C(C=CC=C1C)Cl.OC1=C(C=CC(=C1)[N+](=O)[O-])NC(=O)NC1=C(C=CC=C1)SC Chemical compound OC1=C(C=CC(=C1)[N+](=O)[O-])NC(=O)NC1=C(C=CC=C1C)Cl.OC1=C(C=CC(=C1)[N+](=O)[O-])NC(=O)NC1=C(C=CC=C1)SC YKSZIRMKYDFEGV-UHFFFAOYSA-N 0.000 description 1
- NBZLIBJSOOCTKZ-UHFFFAOYSA-N OC1=C(C=CC(=C1Cl)Cl)NC(=O)NC1=C(C(=CC=C1)Cl)Cl.NC(=O)N Chemical compound OC1=C(C=CC(=C1Cl)Cl)NC(=O)NC1=C(C(=CC=C1)Cl)Cl.NC(=O)N NBZLIBJSOOCTKZ-UHFFFAOYSA-N 0.000 description 1
- CUOGGYMQKSIUNO-UHFFFAOYSA-N OC1=C(C=CC=C1C#N)NC(=O)NC1=C(C(=CC=C1)Cl)Cl.NC(=O)N Chemical compound OC1=C(C=CC=C1C#N)NC(=O)NC1=C(C(=CC=C1)Cl)Cl.NC(=O)N CUOGGYMQKSIUNO-UHFFFAOYSA-N 0.000 description 1
- FCYCSRITRRDGIN-UHFFFAOYSA-N OC1=C(C=CC=C1C#N)NC(=O)NC1=CC(=CC=C1)C(F)(F)F.NC(=O)N Chemical compound OC1=C(C=CC=C1C#N)NC(=O)NC1=CC(=CC=C1)C(F)(F)F.NC(=O)N FCYCSRITRRDGIN-UHFFFAOYSA-N 0.000 description 1
- SYUOLCIDOXSDBJ-UHFFFAOYSA-N OC1=C(C=CC=C1C(F)(F)F)NC(=O)NC1=C(C=CC=C1)Br.OC1=C(C=CC=C1C(F)(F)F)NC(=O)NC1=C(C=CC=C1)Br Chemical compound OC1=C(C=CC=C1C(F)(F)F)NC(=O)NC1=C(C=CC=C1)Br.OC1=C(C=CC=C1C(F)(F)F)NC(=O)NC1=C(C=CC=C1)Br SYUOLCIDOXSDBJ-UHFFFAOYSA-N 0.000 description 1
- ZCQWOFVYLHDMMC-UHFFFAOYSA-N Oxazole Chemical compound C1=COC=N1 ZCQWOFVYLHDMMC-UHFFFAOYSA-N 0.000 description 1
- 102000016387 Pancreatic elastase Human genes 0.000 description 1
- 108010067372 Pancreatic elastase Proteins 0.000 description 1
- YGYAWVDWMABLBF-UHFFFAOYSA-N Phosgene Chemical compound ClC(Cl)=O YGYAWVDWMABLBF-UHFFFAOYSA-N 0.000 description 1
- OFOBLEOULBTSOW-UHFFFAOYSA-N Propanedioic acid Natural products OC(=O)CC(O)=O OFOBLEOULBTSOW-UHFFFAOYSA-N 0.000 description 1
- WTKZEGDFNFYCGP-UHFFFAOYSA-N Pyrazole Chemical compound C=1C=NNC=1 WTKZEGDFNFYCGP-UHFFFAOYSA-N 0.000 description 1
- CZPWVGJYEJSRLH-UHFFFAOYSA-N Pyrimidine Chemical compound C1=CN=CN=C1 CZPWVGJYEJSRLH-UHFFFAOYSA-N 0.000 description 1
- IWYDHOAUDWTVEP-UHFFFAOYSA-N R-2-phenyl-2-hydroxyacetic acid Natural products OC(=O)C(O)C1=CC=CC=C1 IWYDHOAUDWTVEP-UHFFFAOYSA-N 0.000 description 1
- KEAYESYHFKHZAL-UHFFFAOYSA-N Sodium Chemical compound [Na] KEAYESYHFKHZAL-UHFFFAOYSA-N 0.000 description 1
- UIIMBOGNXHQVGW-DEQYMQKBSA-M Sodium bicarbonate-14C Chemical compound [Na+].O[14C]([O-])=O UIIMBOGNXHQVGW-DEQYMQKBSA-M 0.000 description 1
- KDYFGRWQOYBRFD-UHFFFAOYSA-N Succinic acid Natural products OC(=O)CCC(O)=O KDYFGRWQOYBRFD-UHFFFAOYSA-N 0.000 description 1
- OUUQCZGPVNCOIJ-UHFFFAOYSA-M Superoxide Chemical compound [O-][O] OUUQCZGPVNCOIJ-UHFFFAOYSA-M 0.000 description 1
- FEWJPZIEWOKRBE-UHFFFAOYSA-N Tartaric acid Natural products [H+].[H+].[O-]C(=O)C(O)C(O)C([O-])=O FEWJPZIEWOKRBE-UHFFFAOYSA-N 0.000 description 1
- DHXVGJBLRPWPCS-UHFFFAOYSA-N Tetrahydropyran Chemical compound C1CCOCC1 DHXVGJBLRPWPCS-UHFFFAOYSA-N 0.000 description 1
- FZWLAAWBMGSTSO-UHFFFAOYSA-N Thiazole Chemical compound C1=CSC=N1 FZWLAAWBMGSTSO-UHFFFAOYSA-N 0.000 description 1
- ZMZDMBWJUHKJPS-UHFFFAOYSA-M Thiocyanate anion Chemical compound [S-]C#N ZMZDMBWJUHKJPS-UHFFFAOYSA-M 0.000 description 1
- LXRZVMYMQHNYJB-UNXOBOICSA-N [(1R,2S,4R)-4-[[5-[4-[(1R)-7-chloro-1,2,3,4-tetrahydroisoquinolin-1-yl]-5-methylthiophene-2-carbonyl]pyrimidin-4-yl]amino]-2-hydroxycyclopentyl]methyl sulfamate Chemical compound CC1=C(C=C(S1)C(=O)C1=C(N[C@H]2C[C@H](O)[C@@H](COS(N)(=O)=O)C2)N=CN=C1)[C@@H]1NCCC2=C1C=C(Cl)C=C2 LXRZVMYMQHNYJB-UNXOBOICSA-N 0.000 description 1
- BGABKEVTHIJBIW-GMSGAONNSA-N [(1r,4s)-7,7-dimethyl-3-oxo-4-bicyclo[2.2.1]heptanyl]methanesulfonyl chloride Chemical compound C1C[C@@]2(CS(Cl)(=O)=O)C(=O)C[C@@H]1C2(C)C BGABKEVTHIJBIW-GMSGAONNSA-N 0.000 description 1
- BGABKEVTHIJBIW-XVKPBYJWSA-N [(1s,4r)-7,7-dimethyl-3-oxo-4-bicyclo[2.2.1]heptanyl]methanesulfonyl chloride Chemical compound C1C[C@]2(CS(Cl)(=O)=O)C(=O)C[C@H]1C2(C)C BGABKEVTHIJBIW-XVKPBYJWSA-N 0.000 description 1
- ANAPSEIYMQUBGO-UHFFFAOYSA-N [3-[(2-hydroxy-4-nitrophenyl)carbamoylamino]-2-methylphenyl] hydrogen sulfate Chemical compound C1=CC=C(OS(O)(=O)=O)C(C)=C1NC(=O)NC1=CC=C([N+]([O-])=O)C=C1O ANAPSEIYMQUBGO-UHFFFAOYSA-N 0.000 description 1
- OEYIVQGXJGCVAT-UHFFFAOYSA-N [N+](=O)([O-])C=1C(=C(C=CC(=O)O)C=CC1)O.[N+](=O)([O-])C=1C(=C(C=CC(=O)O)C=CC1)CO Chemical compound [N+](=O)([O-])C=1C(=C(C=CC(=O)O)C=CC1)O.[N+](=O)([O-])C=1C(=C(C=CC(=O)O)C=CC1)CO OEYIVQGXJGCVAT-UHFFFAOYSA-N 0.000 description 1
- 230000002159 abnormal effect Effects 0.000 description 1
- 238000009825 accumulation Methods 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 230000003213 activating effect Effects 0.000 description 1
- 230000001154 acute effect Effects 0.000 description 1
- 239000003513 alkali Substances 0.000 description 1
- 150000001447 alkali salts Chemical class 0.000 description 1
- BJEPYKJPYRNKOW-UHFFFAOYSA-N alpha-hydroxysuccinic acid Natural products OC(=O)C(O)CC(O)=O BJEPYKJPYRNKOW-UHFFFAOYSA-N 0.000 description 1
- 229910052782 aluminium Inorganic materials 0.000 description 1
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 1
- 150000001408 amides Chemical class 0.000 description 1
- 150000001413 amino acids Chemical class 0.000 description 1
- SOIFLUNRINLCBN-UHFFFAOYSA-N ammonium thiocyanate Chemical compound [NH4+].[S-]C#N SOIFLUNRINLCBN-UHFFFAOYSA-N 0.000 description 1
- ZZNCQTGLAPYNDN-UHFFFAOYSA-N anilino 4-[(2-bromophenyl)carbamoylamino]-3-hydroxybenzoate Chemical compound OC1=CC(C(=O)ONC=2C=CC=CC=2)=CC=C1NC(=O)NC1=CC=CC=C1Br ZZNCQTGLAPYNDN-UHFFFAOYSA-N 0.000 description 1
- WUVCQOUWKXAJHS-UHFFFAOYSA-N anilino 4-amino-3-hydroxybenzoate Chemical compound C1=C(O)C(N)=CC=C1C(=O)ONC1=CC=CC=C1 WUVCQOUWKXAJHS-UHFFFAOYSA-N 0.000 description 1
- 125000002490 anilino group Chemical group [H]N(*)C1=C([H])C([H])=C([H])C([H])=C1[H] 0.000 description 1
- 239000005557 antagonist Substances 0.000 description 1
- 229940121363 anti-inflammatory agent Drugs 0.000 description 1
- 239000002260 anti-inflammatory agent Substances 0.000 description 1
- 239000008346 aqueous phase Substances 0.000 description 1
- 239000012300 argon atmosphere Substances 0.000 description 1
- 125000005018 aryl alkenyl group Chemical group 0.000 description 1
- 150000001502 aryl halides Chemical class 0.000 description 1
- 230000008901 benefit Effects 0.000 description 1
- 235000010233 benzoic acid Nutrition 0.000 description 1
- 229960004365 benzoic acid Drugs 0.000 description 1
- WGQKYBSKWIADBV-UHFFFAOYSA-N benzylamine Chemical compound NCC1=CC=CC=C1 WGQKYBSKWIADBV-UHFFFAOYSA-N 0.000 description 1
- 125000000440 benzylamino group Chemical group [H]N(*)C([H])([H])C1=C([H])C([H])=C([H])C([H])=C1[H] 0.000 description 1
- 125000000051 benzyloxy group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])O* 0.000 description 1
- 230000002051 biphasic effect Effects 0.000 description 1
- UBXYXCRCOKCZIT-UHFFFAOYSA-N biphenyl-3-ol Chemical compound OC1=CC=CC(C=2C=CC=CC=2)=C1 UBXYXCRCOKCZIT-UHFFFAOYSA-N 0.000 description 1
- KDYFGRWQOYBRFD-NUQCWPJISA-N butanedioic acid Chemical compound O[14C](=O)CC[14C](O)=O KDYFGRWQOYBRFD-NUQCWPJISA-N 0.000 description 1
- 239000001569 carbon dioxide Substances 0.000 description 1
- 229910002092 carbon dioxide Inorganic materials 0.000 description 1
- HQCVEGNPQFRFPC-UHFFFAOYSA-N carboxy acetate Chemical compound CC(=O)OC(O)=O HQCVEGNPQFRFPC-UHFFFAOYSA-N 0.000 description 1
- 230000036978 cell physiology Effects 0.000 description 1
- 239000007795 chemical reaction product Substances 0.000 description 1
- 230000003399 chemotactic effect Effects 0.000 description 1
- 239000005482 chemotactic factor Substances 0.000 description 1
- 208000037976 chronic inflammation Diseases 0.000 description 1
- 208000037893 chronic inflammatory disorder Diseases 0.000 description 1
- 229960004106 citric acid Drugs 0.000 description 1
- 239000012230 colorless oil Substances 0.000 description 1
- 238000009833 condensation Methods 0.000 description 1
- 230000005494 condensation Effects 0.000 description 1
- 238000001816 cooling Methods 0.000 description 1
- 125000000392 cycloalkenyl group Chemical group 0.000 description 1
- 125000000753 cycloalkyl group Chemical group 0.000 description 1
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- 125000001559 cyclopropyl group Chemical group [H]C1([H])C([H])([H])C1([H])* 0.000 description 1
- 230000016396 cytokine production Effects 0.000 description 1
- NHADDZMCASKINP-HTRCEHHLSA-N decarboxydihydrocitrinin Natural products C1=C(O)C(C)=C2[C@H](C)[C@@H](C)OCC2=C1O NHADDZMCASKINP-HTRCEHHLSA-N 0.000 description 1
- 238000010511 deprotection reaction Methods 0.000 description 1
- SPWVRYZQLGQKGK-UHFFFAOYSA-N dichloromethane;hexane Chemical compound ClCCl.CCCCCC SPWVRYZQLGQKGK-UHFFFAOYSA-N 0.000 description 1
- IJKVHSBPTUYDLN-UHFFFAOYSA-N dihydroxy(oxo)silane Chemical compound O[Si](O)=O IJKVHSBPTUYDLN-UHFFFAOYSA-N 0.000 description 1
- DMBHHRLKUKUOEG-UHFFFAOYSA-N diphenylamine Chemical compound C=1C=CC=CC=1NC1=CC=CC=C1 DMBHHRLKUKUOEG-UHFFFAOYSA-N 0.000 description 1
- 230000008034 disappearance Effects 0.000 description 1
- 238000006073 displacement reaction Methods 0.000 description 1
- 229940079593 drug Drugs 0.000 description 1
- 230000009977 dual effect Effects 0.000 description 1
- 230000000694 effects Effects 0.000 description 1
- 230000003511 endothelial effect Effects 0.000 description 1
- 210000002919 epithelial cell Anatomy 0.000 description 1
- CCIVGXIOQKPBKL-UHFFFAOYSA-N ethanesulfonic acid Chemical compound CCS(O)(=O)=O CCIVGXIOQKPBKL-UHFFFAOYSA-N 0.000 description 1
- 238000001704 evaporation Methods 0.000 description 1
- 230000008020 evaporation Effects 0.000 description 1
- 230000003090 exacerbative effect Effects 0.000 description 1
- 102100021145 fMet-Leu-Phe receptor Human genes 0.000 description 1
- 238000010265 fast atom bombardment Methods 0.000 description 1
- 210000002950 fibroblast Anatomy 0.000 description 1
- 125000001153 fluoro group Chemical group F* 0.000 description 1
- 238000009472 formulation Methods 0.000 description 1
- 239000001530 fumaric acid Substances 0.000 description 1
- 229960002598 fumaric acid Drugs 0.000 description 1
- 235000011087 fumaric acid Nutrition 0.000 description 1
- 239000008187 granular material Substances 0.000 description 1
- 229940093915 gynecological organic acid Drugs 0.000 description 1
- 125000005553 heteroaryloxy group Chemical group 0.000 description 1
- 229960001340 histamine Drugs 0.000 description 1
- NPZTUJOABDZTLV-UHFFFAOYSA-N hydroxybenzotriazole Substances O=C1C=CC=C2NNN=C12 NPZTUJOABDZTLV-UHFFFAOYSA-N 0.000 description 1
- 239000011630 iodine Substances 0.000 description 1
- 229910052740 iodine Inorganic materials 0.000 description 1
- 125000002346 iodo group Chemical group I* 0.000 description 1
- NONOKGVFTBWRLD-UHFFFAOYSA-N isocyanatosulfanylimino(oxo)methane Chemical compound O=C=NSN=C=O NONOKGVFTBWRLD-UHFFFAOYSA-N 0.000 description 1
- 239000004310 lactic acid Substances 0.000 description 1
- 235000014655 lactic acid Nutrition 0.000 description 1
- 229960000448 lactic acid Drugs 0.000 description 1
- 108010019677 lymphotactin Proteins 0.000 description 1
- KKSDGJDHHZEWEP-UHFFFAOYSA-N m-hydroxycinnamic acid Natural products OC(=O)C=CC1=CC=CC(O)=C1 KKSDGJDHHZEWEP-UHFFFAOYSA-N 0.000 description 1
- 125000000040 m-tolyl group Chemical group [H]C1=C([H])C(*)=C([H])C(=C1[H])C([H])([H])[H] 0.000 description 1
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 description 1
- 239000011976 maleic acid Substances 0.000 description 1
- 229940098895 maleic acid Drugs 0.000 description 1
- 239000001630 malic acid Substances 0.000 description 1
- 235000011090 malic acid Nutrition 0.000 description 1
- 229940099690 malic acid Drugs 0.000 description 1
- 229960002510 mandelic acid Drugs 0.000 description 1
- 238000001819 mass spectrum Methods 0.000 description 1
- 201000001441 melanoma Diseases 0.000 description 1
- 125000004170 methylsulfonyl group Chemical group [H]C([H])([H])S(*)(=O)=O 0.000 description 1
- 150000007522 mineralic acids Chemical class 0.000 description 1
- 210000001616 monocyte Anatomy 0.000 description 1
- JFCHSQDLLFJHOA-UHFFFAOYSA-N n,n-dimethylsulfamoyl chloride Chemical compound CN(C)S(Cl)(=O)=O JFCHSQDLLFJHOA-UHFFFAOYSA-N 0.000 description 1
- DPMNELWTCXXOKN-UHFFFAOYSA-N n-(2-aminophenyl)-4-phenoxybenzenesulfonamide Chemical compound NC1=CC=CC=C1NS(=O)(=O)C(C=C1)=CC=C1OC1=CC=CC=C1 DPMNELWTCXXOKN-UHFFFAOYSA-N 0.000 description 1
- KKBCMONERKOXFP-UHFFFAOYSA-N n-(2-aminophenyl)thiophene-2-sulfonamide Chemical compound NC1=CC=CC=C1NS(=O)(=O)C1=CC=CS1 KKBCMONERKOXFP-UHFFFAOYSA-N 0.000 description 1
- APVPOHHVBBYQAV-UHFFFAOYSA-N n-(4-aminophenyl)sulfonyloctadecanamide Chemical compound CCCCCCCCCCCCCCCCCC(=O)NS(=O)(=O)C1=CC=C(N)C=C1 APVPOHHVBBYQAV-UHFFFAOYSA-N 0.000 description 1
- HJPVUCISGFMYLB-UHFFFAOYSA-N n-[[2-hydroxy-3-(trifluoromethyl)phenyl]carbamoyl]benzamide Chemical compound C1=CC=C(C(F)(F)F)C(O)=C1NC(=O)NC(=O)C1=CC=CC=C1 HJPVUCISGFMYLB-UHFFFAOYSA-N 0.000 description 1
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- KPKIPHUQBFYHKR-UHFFFAOYSA-N n-carbamoyl-n-(4-cyano-2-hydroxyphenyl)benzamide Chemical compound C=1C=C(C#N)C=C(O)C=1N(C(=O)N)C(=O)C1=CC=CC=C1 KPKIPHUQBFYHKR-UHFFFAOYSA-N 0.000 description 1
- 125000000740 n-pentyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 238000006396 nitration reaction Methods 0.000 description 1
- PMOWTIHVNWZYFI-UHFFFAOYSA-N o-Coumaric acid Natural products OC(=O)C=CC1=CC=CC=C1O PMOWTIHVNWZYFI-UHFFFAOYSA-N 0.000 description 1
- 150000007524 organic acids Chemical class 0.000 description 1
- 235000005985 organic acids Nutrition 0.000 description 1
- 235000010292 orthophenyl phenol Nutrition 0.000 description 1
- 229940116315 oxalic acid Drugs 0.000 description 1
- 235000006408 oxalic acid Nutrition 0.000 description 1
- 239000007800 oxidant agent Substances 0.000 description 1
- 230000001590 oxidative effect Effects 0.000 description 1
- FJKROLUGYXJWQN-UHFFFAOYSA-N papa-hydroxy-benzoic acid Natural products OC(=O)C1=CC=C(O)C=C1 FJKROLUGYXJWQN-UHFFFAOYSA-N 0.000 description 1
- RGSFGYAAUTVSQA-UHFFFAOYSA-N pentamethylene Natural products C1CCCC1 RGSFGYAAUTVSQA-UHFFFAOYSA-N 0.000 description 1
- 125000000951 phenoxy group Chemical group [H]C1=C([H])C([H])=C(O*)C([H])=C1[H] 0.000 description 1
- FCJSHPDYVMKCHI-UHFFFAOYSA-N phenyl benzoate Chemical compound C=1C=CC=CC=1C(=O)OC1=CC=CC=C1 FCJSHPDYVMKCHI-UHFFFAOYSA-N 0.000 description 1
- 229960003424 phenylacetic acid Drugs 0.000 description 1
- 239000003279 phenylacetic acid Substances 0.000 description 1
- OAHKWDDSKCRNFE-UHFFFAOYSA-N phenylmethanesulfonyl chloride Chemical compound ClS(=O)(=O)CC1=CC=CC=C1 OAHKWDDSKCRNFE-UHFFFAOYSA-N 0.000 description 1
- 230000035790 physiological processes and functions Effects 0.000 description 1
- 108090000765 processed proteins & peptides Proteins 0.000 description 1
- 230000000069 prophylactic effect Effects 0.000 description 1
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 238000001243 protein synthesis Methods 0.000 description 1
- 239000003586 protic polar solvent Substances 0.000 description 1
- 125000002294 quinazolinyl group Chemical group N1=C(N=CC2=CC=CC=C12)* 0.000 description 1
- JUYUYCIJACTHMK-UHFFFAOYSA-N quinoline-8-sulfonyl chloride Chemical compound C1=CN=C2C(S(=O)(=O)Cl)=CC=CC2=C1 JUYUYCIJACTHMK-UHFFFAOYSA-N 0.000 description 1
- 125000002943 quinolinyl group Chemical group N1=C(C=CC2=CC=CC=C12)* 0.000 description 1
- 239000000376 reactant Substances 0.000 description 1
- 238000001525 receptor binding assay Methods 0.000 description 1
- 230000001105 regulatory effect Effects 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 230000004044 response Effects 0.000 description 1
- 238000012552 review Methods 0.000 description 1
- 206010039073 rheumatoid arthritis Diseases 0.000 description 1
- WKEDVNSFRWHDNR-UHFFFAOYSA-N salicylanilide Chemical compound OC1=CC=CC=C1C(=O)NC1=CC=CC=C1 WKEDVNSFRWHDNR-UHFFFAOYSA-N 0.000 description 1
- 229960004889 salicylic acid Drugs 0.000 description 1
- 239000012047 saturated solution Substances 0.000 description 1
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 238000010898 silica gel chromatography Methods 0.000 description 1
- XGVXKJKTISMIOW-ZDUSSCGKSA-N simurosertib Chemical compound N1N=CC(C=2SC=3C(=O)NC(=NC=3C=2)[C@H]2N3CCC(CC3)C2)=C1C XGVXKJKTISMIOW-ZDUSSCGKSA-N 0.000 description 1
- 150000003384 small molecules Chemical class 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 239000012279 sodium borohydride Substances 0.000 description 1
- 229910000033 sodium borohydride Inorganic materials 0.000 description 1
- 239000012312 sodium hydride Substances 0.000 description 1
- OVYTZAASVAZITK-UHFFFAOYSA-M sodium;ethanol;hydroxide Chemical compound [OH-].[Na+].CCO OVYTZAASVAZITK-UHFFFAOYSA-M 0.000 description 1
- 239000012265 solid product Substances 0.000 description 1
- 241000894007 species Species 0.000 description 1
- 238000001228 spectrum Methods 0.000 description 1
- 238000010561 standard procedure Methods 0.000 description 1
- 230000004936 stimulating effect Effects 0.000 description 1
- 125000005504 styryl group Chemical group 0.000 description 1
- FDDDEECHVMSUSB-UHFFFAOYSA-N sulfanilamide Chemical compound NC1=CC=C(S(N)(=O)=O)C=C1 FDDDEECHVMSUSB-UHFFFAOYSA-N 0.000 description 1
- 125000000472 sulfonyl group Chemical group *S(*)(=O)=O 0.000 description 1
- YBBRCQOCSYXUOC-UHFFFAOYSA-N sulfuryl dichloride Chemical compound ClS(Cl)(=O)=O YBBRCQOCSYXUOC-UHFFFAOYSA-N 0.000 description 1
- 239000001117 sulphuric acid Substances 0.000 description 1
- 235000011149 sulphuric acid Nutrition 0.000 description 1
- 239000011975 tartaric acid Substances 0.000 description 1
- 229960001367 tartaric acid Drugs 0.000 description 1
- 235000002906 tartaric acid Nutrition 0.000 description 1
- YBRBMKDOPFTVDT-UHFFFAOYSA-N tert-butylamine Chemical compound CC(C)(C)N YBRBMKDOPFTVDT-UHFFFAOYSA-N 0.000 description 1
- BCNZYOJHNLTNEZ-UHFFFAOYSA-N tert-butyldimethylsilyl chloride Chemical compound CC(C)(C)[Si](C)(C)Cl BCNZYOJHNLTNEZ-UHFFFAOYSA-N 0.000 description 1
- 125000001981 tert-butyldimethylsilyl group Chemical group [H]C([H])([H])[Si]([H])(C([H])([H])[H])[*]C(C([H])([H])[H])(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- YJBKVPRVZAQTPY-UHFFFAOYSA-J tetrachlorostannane;dihydrate Chemical compound O.O.Cl[Sn](Cl)(Cl)Cl YJBKVPRVZAQTPY-UHFFFAOYSA-J 0.000 description 1
- 230000001225 therapeutic effect Effects 0.000 description 1
- 238000002560 therapeutic procedure Methods 0.000 description 1
- VLLMWSRANPNYQX-UHFFFAOYSA-N thiadiazole Chemical compound C1=CSN=N1.C1=CSN=N1 VLLMWSRANPNYQX-UHFFFAOYSA-N 0.000 description 1
- 125000001544 thienyl group Chemical group 0.000 description 1
- 238000004809 thin layer chromatography Methods 0.000 description 1
- 229930192474 thiophene Natural products 0.000 description 1
- QERYCTSHXKAMIS-UHFFFAOYSA-N thiophene-2-carboxylic acid Chemical compound OC(=O)C1=CC=CS1 QERYCTSHXKAMIS-UHFFFAOYSA-N 0.000 description 1
- VNNLHYZDXIBHKZ-UHFFFAOYSA-N thiophene-2-sulfonyl chloride Chemical compound ClS(=O)(=O)C1=CC=CS1 VNNLHYZDXIBHKZ-UHFFFAOYSA-N 0.000 description 1
- 210000001519 tissue Anatomy 0.000 description 1
- KKSDGJDHHZEWEP-SNAWJCMRSA-N trans-3-coumaric acid Chemical compound OC(=O)\C=C\C1=CC=CC(O)=C1 KKSDGJDHHZEWEP-SNAWJCMRSA-N 0.000 description 1
- 230000014616 translation Effects 0.000 description 1
- 150000003852 triazoles Chemical class 0.000 description 1
- NTJBWZHVSJNKAD-UHFFFAOYSA-N triethylazanium;fluoride Chemical compound [F-].CC[NH+](CC)CC NTJBWZHVSJNKAD-UHFFFAOYSA-N 0.000 description 1
- 125000000876 trifluoromethoxy group Chemical group FC(F)(F)O* 0.000 description 1
- UCPYLLCMEDAXFR-UHFFFAOYSA-N triphosgene Chemical compound ClC(Cl)(Cl)OC(=O)OC(Cl)(Cl)Cl UCPYLLCMEDAXFR-UHFFFAOYSA-N 0.000 description 1
- 241000712461 unidentified influenza virus Species 0.000 description 1
- 150000003672 ureas Chemical class 0.000 description 1
- 210000003556 vascular endothelial cell Anatomy 0.000 description 1
- 230000006444 vascular growth Effects 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/21—Esters, e.g. nitroglycerine, selenocyanates
- A61K31/27—Esters, e.g. nitroglycerine, selenocyanates of carbamic or thiocarbamic acids, meprobamate, carbachol, neostigmine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/16—Amides, e.g. hydroxamic acids
- A61K31/17—Amides, e.g. hydroxamic acids having the group >N—C(O)—N< or >N—C(S)—N<, e.g. urea, thiourea, carmustine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/02—Stomatological preparations, e.g. drugs for caries, aphtae, periodontitis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/08—Drugs for skeletal disorders for bone diseases, e.g. rachitism, Paget's disease
- A61P19/10—Drugs for skeletal disorders for bone diseases, e.g. rachitism, Paget's disease for osteoporosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/12—Antivirals
- A61P31/14—Antivirals for RNA viruses
- A61P31/16—Antivirals for RNA viruses for influenza or rhinoviruses
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/12—Antivirals
- A61P31/20—Antivirals for DNA viruses
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P33/00—Antiparasitic agents
- A61P33/02—Antiprotozoals, e.g. for leishmaniasis, trichomoniasis, toxoplasmosis
- A61P33/06—Antimalarials
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P7/00—Drugs for disorders of the blood or the extracellular fluid
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/10—Drugs for disorders of the cardiovascular system for treating ischaemic or atherosclerotic diseases, e.g. antianginal drugs, coronary vasodilators, drugs for myocardial infarction, retinopathy, cerebrovascula insufficiency, renal arteriosclerosis
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02A—TECHNOLOGIES FOR ADAPTATION TO CLIMATE CHANGE
- Y02A50/00—TECHNOLOGIES FOR ADAPTATION TO CLIMATE CHANGE in human health protection, e.g. against extreme weather
- Y02A50/30—Against vector-borne diseases, e.g. mosquito-borne, fly-borne, tick-borne or waterborne diseases whose impact is exacerbated by climate change
Definitions
- This invention relates to a novel group of phenyl urea compounds, processes for the preparation thereof, the use thereof in treating IL-8, GRO ⁇ , GRO ⁇ , GRO ⁇ and NAP-2 mediated diseases and pharmaceutical compositions for use in such therapy.
- Interleukin-8 such as neutrophil attractant/activation protein- 1 (NAP-1), monocyte derived neutrophil chemotactic factor (MDNCF), neutrophil activating factor (NAF), and T-cell lymphocyte chemotactic factor.
- Interleukin-8 is a chemoattractant for neutrophils, basophils, and a subset of T-cells.
- nucleated cells including macrophages, fibroblasts, endothelial and epithelial cells exposed to TNF, IL-l ⁇ , IL-l ⁇ or LPS, and by neutrophils themselves when exposed to LPS or chemotactic factors such as FMLP.
- GRO ⁇ , GRO ⁇ , GRO ⁇ and NAP-2 also belong to the chemokine ⁇ family. Like IL-8 these chemokines have also been referred to by different names. For instance
- GRO ⁇ , ⁇ , ⁇ have been referred to as MGSA ⁇ , ⁇ and ⁇ respectively (Melanoma Growth Stimulating Activity), see Richmond et al, J. Cell Physiology 129, 375 (1986) and Chang et al, J. Immunol 148, 451 (1992). All of the chemokines of the ⁇ -family which possess the ELR motif directly preceding the CXC motif bind to the IL-8 B receptor. IL-8, GRO ⁇ , GRO ⁇ , GRO ⁇ and NAP-2 stimulate a number of functions in vitro.
- IL-8 and GRO ⁇ have demonstrated T-lymphocytes, and basophiles chemotactic activity.
- IL-8 can induce histamine release from basophils from both normal and atopic individuals
- GRO- ⁇ and IL-8 can in addition, induce lysozomal enzyme release and respiratory burst from neutrophils.
- IL-8 has also been shown to increase the surface expression of Mac- 1 (CD l ib/CD 18) on neutrophils without de novo protein synthesis. This may contribute to increased adhesion of the neutrophils to vascular endothelial cells. Many known diseases are characterized by massive neutrophil infiltration.
- GRO ⁇ , GRO ⁇ , GRO ⁇ and NAP-2 promote the accumulation and activation of neutrophils
- these chemokines have been implicated in a wide range of acute and chronic inflammatory disorders including psoriasis and rheumatoid arthritis, Baggiolini et al, FEBS Lett. 307. 97 (1992); Miller et al, Crit. Rev. Immunol. 12. 17 (1992); Oppenheim et al, Annu. Rev. Immunol. 9. 617 (1991); Seitz et al., J. Clin. Invest. 87, 463 (1991); Miller et al., Am. Rev. Respir. Pis.
- ELR chemokines (those containing the amino acids ELR motif just prior to the CXC motif) have also been implicated in angiostasis. Strieter et al, Science 258, 1798 (1992).
- IL-8, GRO ⁇ , GRO ⁇ , GRO ⁇ and NAP-2 induce neutrophil shape change, chemotaxis, granule release, and respiratory burst, by binding to and activating receptors of the seven-transmembrane, G-protein-linked family, in particular by binding to BL-8 receptors, most notably the B-receptor. Thomas et al., J. Biol. Chem.
- the IL-8 receptor represents a promising target for the development of novel anti-inflammatory agents.
- IL-8R ⁇ which binds only IL-8 with high affinity
- IL-8R ⁇ which has high affinity for IL-8 as well as for GRO- ⁇ , GRO ⁇ , GRO ⁇ and NAP-2.
- This invention provides for a method of treating a chemokine mediated disease, wherein the chemokine is one which binds to an IL-8 ⁇ or ⁇ receptor and which method comprises administering an effective amount of a compound of Formula (I) or a pharmaceutically acceptable salt thereof.
- the chemokine is IL-8.
- This invention also relates to a method of inhibiting the binding of IL-8 to its receptors in a mammal in need thereof which comprises administering to said mammal an effective amount of a compound of Formula (I).
- X is oxygen or sulfur
- R is any functional moiety having an ionizable hydrogen and a pKa of 10 or less;
- Rl is independently selected from hydrogen; halogen; nitro; cyano; halosubstituted Ci-10 alkyl; Ci-io alkyl; C2-10 alkenyl; Ci-io alkoxy; halosubstituted Ci-io alkoxy; azide; S(O)tR4; hydroxy; hydroxy C ⁇ _4alkyl; aryl; aryl Ci-4 alkyl; aryloxy; aryl Ci-4 alkyloxy; heteroaryl; heteroarylalkyl; heterocyclic, heterocyclic Ci- 4alkyl; heteroaryl C ⁇ _4 alkyloxy; aryl C2-10 alkenyl; heteroaryl C2-10 alkenyl; heterocyclic C2-10 alkenyl; NR4R5; C2-10 alkenyl C(O)NR4R5; C(O)NR4R5; C(O)NR4R ⁇ o; S(O)3H; S(O) 3 R 8 ; C ⁇
- Y is independently selected from hydrogen; halogen; nitro; cyano; halosubstituted Ci-io alkyl; Ci-io alkyl; C2-10 alkenyl; Ci_io alkoxy; halosubstituted Ci-io alkoxy; azide; S(O) t R4; hydroxy; hydroxyC ⁇ _4alkyl; aryl; aryl Ci-4 alkyl; aryloxy; arylCi-4 alkyloxy; heteroaryl; heteroarylalkyl; heteroaryl Ci-4 alkyloxy; heterocyclic, heterocyclic Ci-4alkyl; aryl C2-10 alkenyl; heteroaryl C2-10 alkenyl; heterocyclic C2-10 alkenyl; NR4R5; C2-10 alkenyl C(O)NR- ⁇ R5; C(O)NR4R5; C(O)NR4Rl ⁇ ; S(O)3H; S(O) 3 R 8 ;
- R ⁇ is hydrogen or Ci-4 alkyl
- RlO is Ci-10 alkyl C(O)2R8;
- Rl 1 is hydrogen, Ci-4 alkyl, optionally substituted aryl, optionally substituted aryl
- Rl2 is hydrogen, Ci-io alkyl, optionally substituted aryl or optionally substituted arylalkyl; or a pharmaceutically acceptably salt thereof.
- Another aspect of the present invention is to a method of treating a chemokine mediated disease, wherein the chemokine is one which binds to an IL-8 ⁇ or ⁇ receptor and which method comprises administering an effective amount of a compound of Formula (II) or a pharmaceutically acceptable salt thereof, as defined herein.
- This invention also relates to a method of inhibiting the binding of IL-8 to its receptors in a mammal in need thereof which comprises administering to said mammal an effective amount of a compound of Formula (II), as defined herein.
- This invention also relates to the novel compounds of Formula (II), or a pharmaceutically acceptable salt thereof, as defined herein.
- Another aspect of the present invention is to a method of treating a chemokine mediated disease, wherein the chemokine is one which binds to an IL-8 ⁇ or ⁇ receptor and which method comprises administering an effective amount of a compound of Formula (UI) or a pharmaceutically acceptable salt thereof, as defined herein.
- a chemokine mediated disease wherein the chemokine is one which binds to an IL-8 ⁇ or ⁇ receptor and which method comprises administering an effective amount of a compound of Formula (UI) or a pharmaceutically acceptable salt thereof, as defined herein.
- UI compound of Formula
- This invention also relates to a method of inhibiting the binding of IL-8 to its receptors in a mammal in need thereof which comprises administering to said mammal an effective amount of a compound of Formula (III), as defined herein.
- This invention also relates to the novel compounds of Formula (IH), or a pharmaceutically acceptable salt thereof, as defined herein.
- the compounds of Formula (I) may also be used in association with the veterinary treatment of mammals, other than humans, in need of inhibition of IL-8 or other chemokines which bind to the IL-8 ⁇ and ⁇ receptors.
- Chemokine mediated diseases for treatment, therapeutically or prophylactically, in animals include disease states such as those noted herein in the Methods of Treatment section.
- R is suitably any functional moiety which provides an ionizable hydrogen having a pKa of 10 or less, preferably from about 3 to 9, more preferably from about 3 to 7.
- Such functional groups include, but are not limited to, hydroxy, carboxylic acid, thiol, -SR2 -OR2, -NH-C(O)R a , -C(O)NR6R7, a substituted sulfonamides of the formula -NHS(O)2Rb. -S(O)2NHR c , NHC(X2)NHRb, or a tetrazolyl; wherein X2 is oxygen or sulfur, preferably oxygen.
- the functional group is other than a sulfonic acid, either directly or as a substituent group on the aryl, heteroaryl, or heterocyclic moiety ring, such as in SR2 or OR2. More preferably R is OH, SH, or NHS(O)2Rb- Suitably, R2 is a substituted aryl, heteroaryl, or heterocyclic moiety which ring has the functional moiety providing the ionizable hydrogen having a pKa of 10 or less.
- R6 and R7 are independently hydrogen or a C 1-4 alkyl group, or R6 and R7 together with the nitrogen to which they are attached form a 5 to 7 member ring which ring may optionally contain an additional heteroatom which heteroatom is selected from oxygen, nitrogen or sulfur. This heteroring may be optionally substituted as defined herein.
- R a is an alkyl, aryl, arylCi_4alkyl, heteroaryl, heteroarylCi-4alkyl, heterocyclic, or a heterocyclic Ci-4alkyl moiety, all of which may be optionally substituted, as defined herein below.
- R is a NR6R7, alkyl, aryl, arylCi-4alkyl, arylC2-4alkenyl, heteroaryl, heteroarylCi-4alkyl, heteroarylC2-4 alkenyl, heterocyclic, or heterocyclic C ⁇ _4alkyl, or a heterocyclic C2-4alkenyl moiety, camphor, all of which may be optionally substituted one to three times independently by halogen; nitro; halosubstituted Ci-4 alkyl, such as CF3; Ci-4 alkyl, such as methyl; Ci-4 alkoxy, such as methoxy; NR9C(O)R a ; C(O)NR6R7, S(O)3H, or C(O)OCi-4 alkyl.
- Rb is preferably an optionally substituted phenyl, benzyl, or styryl.
- Rb is a heteroaryl preferably it is an optionally substituted thiazole, optionally substituted thienyl, or optionally substituted quinolinyl ring.
- R9 is hydrogen or a Ci-4 alkyl, preferably hydrogen, and suitably when the substituent group is NR9C(O)R a , then R a is preferably an alkyl group, such as methyl.
- R c is hydrogen, alkyl, aryl, arylCi-4alkyl, arylCi-4alkenyl, heteroaryl, heteroarylCi-4alkyl, heteroarylC ⁇ _4alkenyl, heterocyclic, or heterocyclic Ci-4alkyl, or a heterocyclic Ci-4alkenyl moiety, all of which may be optionally substituted one to three times independently by halogen, nitro, halosubstituted C1-.4 alkyl, Ci-4 alkyl, C ⁇ _ 4 alkoxy, NR9C(O)R a , C(O)NR6R7, S(O)3H, or C(O)OCi-4 alkyl, wherein R9 is hydrogen or a Ci-4 alkyl.
- R c is an optionally substituted phenyl.
- R is an OR2 or SR2 moiety it is recognized by one of skill in the art that the aryl ring must, therefore, contain the required ionizable hydrogen.
- the aryl ring may also be additionally substituted, independently, by one to three groups, which groups may also contain an additional ionizable group, and which include but are not limited to, halogen, nitro, halosubstituted Ci-4 alkyl, Ci-4 alkyl, C ⁇ _4 alkoxy, hydroxy, SH, -C(O)NR6R7, -NH-C(O)R a , -NHS(O)2Rb. S(O)2NR6R7, C(O)ORs, or a tetrazolyl ring.
- R is independently selected from hydrogen; halogen; nitro; cyano; halosubstituted Ci-io alkyl, such as CF3; Ci-io alkyl, such as methyl, ethyl, isopropyl, or n-propyl; C2-10 alkenyl; Ci-io alkoxy, such as methoxy, or ethoxy; halosubstituted Ci-io alkoxy, such as trifluoromethoxy; azide; S(O)tR4, wherein t is 0, 1 or 2; hydroxy; hydroxy C ⁇ _4alkyl, such as methanol or ethanol; aryl, such as phenyl or naphthyl; aryl C1-.4 alkyl, such as benzyl; aryloxy, such as phenoxy; aryl C1-.4 alkyloxy, such as benzyloxy; heteroaryl; heteroarylalkyl
- aryl, arylalkyl, arylalkenyl, heteroaryl, heteroarylalkyl, heteroarylalkenyl, heterocyclic, heterocyclicalkyl, and heterocyclicalkenyl moieties may all be optionally substituted as defined herein below.
- Rl is other than azido or S(O)3R8- When Ri forms a dioxybridge, s is preferably 1.
- Rl forms an additional unsaturated ring, it is preferably 6 membered resulting in a naphthylene ring system. This naphthylene ring may be substituted independently, 1 to 3 times by the other Ri moieties as defined above.
- R4 and R5 are independently hydrogen, optionally substituted Ci-4 alkyl, optionally substituted aryl, optionally substituted aryl C ⁇ _4alkyl, optionally substituted heteroaryl, optionally substituted heteroaryl Ci-4alkyl, heterocyclic, heterocyclicCi-4 alkyl, or R4 and R5 together with the nitrogen to which they are attached form a 5 to 7 member ring which may optionally comprise an additional heteroatom selected from O/N/S.
- Rio is suitably Ci-io alkyl C(O)2R8, such as CH2C(O)2H or CH2C(O)2CH3.
- Rl 1 is suitably hydrogen, C1-.4 alkyl, aryl, aryl Ci-4 alkyl, heteroaryl, heteroaryl Ci-4alkyl, heterocyclic, or heterocyclic C _4alkyl.
- Rl2 is suitably hydrogen, CI-10 alkyl, optionally substituted aryl or optionally substituted arylalkyl.
- Ri is halogen, cyano, nitro, CF3, C(O)NR4R5, alkenyl C(O)NR4R5,
- R4R10 alkenyl C(O)ORi2, heteroaryl, heteroarylalkyl , heteroaryl alkenyl, or S(O)NR4R5, and preferably R4 and R5 are both hydrogen or one is phenyl.
- a preferred ring substitution for R ⁇ is in the 4-position of the phenyl ring.
- R is OH, SH or NSO2Rb than Ri is preferably substituted in the 3- position, the 4- position or di substituted in the 3,4- position.
- the substituent group is suitably an electron withdrawing moiety.
- R is OH, SH or NSO2 b > than Ri is nitro, halogen, cyano, trifluoromethyl group, C(O)NR4R5.
- Ri is preferably hydrogen, or R is preferably substituted in the 4-position, more preferably substituted by trifluoromethyl or chloro.
- Y is independently selected from hydrogen; halogen; nitro; cyano; halosubstituted Ci-io alkyl; Ci-io alkyl; C2-10 alkenyl; Ci-io alkoxy; halosubstituted Ci-io alkoxy; azide; S(O)tR4; hydroxy; hydroxy Ci-4alkyl; aryl; aryl C1-.4 alkyl; aryloxy; arylCi-4 alkyloxy; aryl C2-10 alkenyl; heteroaryl; heteroarylalkyl; heteroaryl Ci-4 alkyloxy; heteroaryl C2-10 alkenyl; heterocyclic, heterocyclic Ci-4- ⁇ kyl; heterocyclicC2-10 alkenyl; NR4R5; C
- s is preferably 1.
- Y forms an additional unsaturated ring, it is preferably 6 membered resulting in a naphthylene ring system.
- This naphthylene ring may be substituted 1 to 3 times by other Y moieties as defined above.
- the aryl, heteroaryl and heterocyclic moieties noted above may all be optionally substituted as defined herein.
- Ri is other than azido or S(O)3R8-
- Y is preferably a halogen, Ci-4 alkoxy, optionally substituted aryl, optionally substituted aryloxy or arylalkoxy, methylene dioxy, NR4R5, thio C ⁇ _4alkyl, thioaryl, halosubstituted alkoxy, optionally substituted C -4 alkyl, or hydroxy alkyl.
- Y is more preferably mono-substituted halogen, disubstituted halogen, mono-substituted alkoxy, disubstituted alkoxy, methylenedioxy, aryl, or alkyl, more preferably these groups are mono or di-substituted in the 2 - position or 2'-, 3 -position.
- Y may be substituted in any of the 5 ring positions, preferably when R is OH, SH, or NSO2Rb.
- Y i preferably mono-substituted in the 2 -position or 3 - position, with the 4 - preferably being unsubstituted. If the ring is disubstituted, when R is OH, SH, or NSO2Rb > substituents are preferably in the 2' or 3' position of a monocyclic ring.
- Ri and Y can both be hydrogen, it is preferred that at least one of the rings be substituted, preferably both rings are substituted.
- X is suitably oxygen or sulfur, preferably oxygen.
- X is suitably oxygen or sulfur, preferably oxygen.
- Formula (I), (Ia-c), (II), or (HI) another aspect of this invention are the symmetrical bis compounds which are included for each structure.
- Formula (I) include:
- halogen such as fluorine, chlorine, bromine or iodine
- hydroxy hydroxy substituted Ci-ioalkyl
- C ⁇ _ ⁇ o alkoxy such as methoxy or ethoxy
- S(O)m' Ci- ⁇ o alkyl wherein m' is 0, 1 or 2, such as methyl thio, methyl sulfinyl or methyl sulfonyl
- Rl3 is suitably Ci-4 alkyl, aryl, aryl Ci-4alkyl, heteroaryl, heteroarylCi_4alkyl, heterocyclic, or heterocyclicC ⁇ _4alkyl.
- Another aspect of the present invention are the novel compounds of Formula (II), or a pharmaceutically acceptable salt thereof, as described below, which are also useful in inhibiting the binding of IL-8 to its receptors in a mammal in need thereof.
- This invention also relates to the pharmaceutical compositions comprising a compound of Formula (II) and a pharmaceutically acceptable diluent or carrier.
- Compounds of Formula (II) are also useful for treating a chemokine mediated disease, wherein the chemokine is one which binds to an IL-8 ⁇ or ⁇ receptor and which method comprises administering an effective amount of a compound of Formula (II) or a pharmaceutically acceptable salt thereof.
- Compounds of Formula (II) are represented by the structure:
- X is oxygen or sulfur
- R is any functional moiety having an ionizable hydrogen and a pKa of 10 or less;
- Rl is independently selected from hydrogen; halogen; nitro; cyano; halosubstituted Ci-io alkyl; Ci-io alkyl; C2-10 alkenyl; Ci-io alkoxy; halosubstituted Ci-io alkoxy; azide; S(O)tR4; hydroxy; hydroxyC ⁇ -4alkyl; aryl; aryl Ci-4 alkyl; aryloxy; arylCi-4 alkyloxy; heteroaryl; heteroarylalkyl; heterocyclic, heterocyclicCi-4alkyl; heteroarylCi-4 alkyloxy; aryl C2-10 alkenyl; heteroaryl C2-10 alkenyl; heterocyclicC2-10 alkenyl; NR4R5; C2-10 alkenyl C(O)NR4R5; C(O)NR4R5
- R4 and R5 are independently hydrogen, optionally substituted Ci-4 alkyl, optionally substituted aryl, optionally substituted aryl Ci-4alkyl, optionally substituted heteroaryl, optionally substituted heteroaryl C ⁇ _4alkyl, heterocyclic, heterocyclicCi-4 alkyl, or R4 and R5 together with the nitrogen to which they are attached form a 5 to 7 member ring which may optionally comprise an additional heteroatom selected from O/N/S; Y is independently selected from hydrogen; halogen; nitro; cyano; halosubstituted
- Ci-io alkyl Ci-io alkyl; C2-10 alkenyl; CMO alkoxy; halosubstituted Ci-io alkoxy; azide; S(O)tR4; hydroxy; hydroxy C ⁇ _4alkyl; aryl; aryl Ci-4 alkyl; aryloxy; aryl Ci-4 alkyloxy; heteroaryl; heteroarylalkyl; heteroarylCi-4 alkyloxy; heterocyclic, heterocyclic Ci-4alkyl; aryl C2-10 alkenyl; heteroaryl C2-10 alkenyl; heterocyclic C2-10 alkenyl; NR4R5; C2-10 alkenyl C(O)NR4R5; C(O)NR4R5;
- C2-10 alkenyl C(O)ORn; C(O)Rn; C(O)ORi2; OC(O) Rn; NR4C(O)Rn; or two Y moieties together may form O-(CH2)sO- or a 5 to 6 membered unsaturated ring; n is an integer having a value of 1 to 3; m is an integer having a value of 1 to 3; R8 is hydrogen or Ci-4 alkyl;
- RlO is CMO alkyl C(O)2R8.
- Rl 1 is hydrogen, Ci-4 alkyl, optionally substituted aryl, optionally substituted aryl
- Ci-4alkyl optionally substituted heteroaryl, optionally substituted heteroarylCi-4 alkyl, optionally substituted heterocyclic, or optionally substituted heterocyclicCi-4 alkyl
- Rl2 is hydrogen, Ci-io alkyl, optionally substituted aryl or optionally substituted arylalkyl
- E is optionally selected from
- the variables for Formula (II), such as X, R, Ri, R4 , R5, R6, R7, R8, R9, Y, R a , Rb, Rc> n > m > and s terms, etc. are as defined in Formula (I) above.
- the E ring denoted by its point of attachment through the asterix (*) may optionally be present. If it is not present the ring is a phenyl moiety which is substituted by the R and R terms as shown. At least one E ring is necessary.
- the E ring may be substituted by the Ri moiety in any ring, saturated or unsaturated, and is shown for purposes herein substituted only in the unsaturated ring(s).
- Exemplified compounds of Formula (HI) are: N- [2-hydroxy-5-indanone] -N '- [2-bromophenyl] urea; N-[l-hydroxyfluorene]-N'-[2-bromophenyl] urea;
- N-[3-hydroxy-9,10-anthraquinon-2-yl]-N'-[2-bromophenyl] urea Another aspect of the present invention are the novel compounds of Formula (IH), or a pharmaceutically acceptable salt thereof, as described below, which are also useful in inhibiting the binding of IL-8 to its receptors in a mammal in need thereof.
- This invention also relates to the pharmaceutical compositions comprising a compound of Formula (HI) and a pharmaceutically acceptable diluent or carrier.
- Compounds of Formula (El) are also useful for treating a chemokine mediated disease, wherein the chemokine is one which binds to an IL-8 ⁇ or ⁇ receptor and which method comprises administering an effective amount of a compound of Formula (HI) or a pharmaceutically acceptable salt thereof.
- Compounds of Formula (DI) are represented by the formula:
- X is oxygen or sulfur
- R is any functional moiety having an ionizable hydrogen and a pKa of 10 or less;
- Ri is independently selected from hydrogen; halogen; nitro; cyano; halosubstituted Ci-io alkyl; Ci-io alkyl; C2-10 alkenyl; CMO alkoxy; halosubstituted Ci-io alkoxy; azide; S(O)tR4; hydroxy; hydroxyC ⁇ _4alkyl; aryl; aryl Ci-4 alkyl; aryloxy; arylCi-4 alkyloxy; heteroaryl; heteroarylalkyl; heterocyclic, heterocyclicCi-4alkyl; heteroarylCi-4 alkyloxy; aryl C2-10 alkenyl; heteroaryl C2-10 alkenyl; heterocyclicC2-10 alkenyl; NR4R5; C2-10 alkenyl C(O)NR4R5; C(O)NR4Rs;
- CMO alkyl C(O)R ⁇ ; C 2 -10 alkenyl C(O)Rn; C2-IO alkenyl C(O)OR ⁇ ; C(O)Rn; C(O)ORi2; OC(O) Rn ; NR4C(0)Rn; or two Rl moieties together may form O-(CH2)sO- or a 5 to 6 membered unsaturated ring; t is 0, or an integer having a value of 1 or 2; s is an integer having a value of 1 to 3;
- R4 and R5 are independently hydrogen, optionally substituted Ci-4 alkyl, optionally substituted aryl, optionally substituted aryl C ⁇ _4alkyl, optionally substituted heteroaryl, optionally substituted heteroaryl Ci-4alkyl, heterocyclic, heterocyclicCi-4 alkyl, or R4 and R5 together with the nitrogen to which they are attached form a 5 to 7 member ring which may optionally comprise an additional heteroatom selected from O/N/S; Y is independently selected from hydrogen; halogen; nitro; cyano; halosubstituted
- C(O)NR4Rl ⁇ ; S(O) 3 H; S(O) 3 R8; Ci-io alkyl C(O)Rn; C 2 -10 alkenyl C(O)Rn; C2-10 alkenyl C(O)ORn; C(O)Rn; C(O)ORi2; OC(O) Rn; NR4C(O)Rn; or two Y moieties together may form O-(CH2)sO- or a 5 to 6 membered unsaturated ring; n is an integer having a value of 1 to 3; m is an integer having a value of 1 to 3; R8 is hydrogen or C 1-4 alkyl;
- RlO is Ci-10 alkyl C(O)2R ⁇ ;
- Rl 1 is hydrogen, Ci-4 alkyl, optionally substituted aryl, optionally substituted aryl C ⁇ _4alkyl, optionally substituted heteroaryl, optionally substituted heteroarylCi- 4alkyl, optionally substituted heterocyclic, or optionally substituted heterocyclicCi - 4alkyl;
- Rl2 is hydrogen, Ci-io alkyl, optionally substituted aryl or optionally substituted arylalkyl; or a pharmaceutically acceptably salt thereof.
- variables, etc. for Formula (II) are the same as those defined for Formula (I) above, such as for example the R variable.
- Exemplified compounds of Formula (HI) are N-(2-Hydroxy-4-nitrophenyl)-N'- (3-methoxy-2-thienyl)urea ; and N-(2-hydroxy-5-nitrophenyl)-N'-(3-methoxy-2- thienyl)urea.
- Another aspect of the present invention is the novel compounds of Formula (la), a subset of compounds of Formula (I) useful for treating a chemokine mediated disease as defined herein.
- This invention also relates to the pharmaceutical compositions comprising a compound of Formula (la) and a pharmaceutically acceptable diluent or carrier.
- the compounds of Formula (la) are represented by the structure:
- X is oxygen or sulfur
- R a is an alkyl, aryl, arylCi-4alkyl, heteroaryl, heteroaryl Ci-4alkyl, heterocyclic, or a heterocyclic Ci-4alkyl moiety, all of which may be optionally substituted;
- Rb is a NR6R7, alkyl, aryl, arylCi-4alkyl, aryl C2-4alkenyl, heteroaryl, heteroarylCi_4alkyl, heteroarylC2-4 alkenyl, heterocyclic, or heterocyclic
- Ci-4alkyl or a heterocyclic C2-4alkenyl moiety, camphor, all of which may be optionally substituted one to three times independently by halogen; nitro; halosubstituted Ci-4 alkyl; Ci-4 alkyl; Ci-4 alkoxy; NR9C(O)R a ; C(O)NR6R7, S(O)3H, or C(O)OCi_4 alkyl; R6 and R7 are independently hydrogen or a Ci-4 alkyl group, or R6 and R7 together with the nitrogen to which they are attached form a 5 to 7 member ring which ring may optionally contain an additional heteroatom which heteroatom is selected from oxygen, nitrogen or sulfur, which ring may be optionally substituted; R9 is hydrogen or a Ci-4 alkyl, preferably hydrogen; Ri is independently selected from hydrogen; halogen; nitro; cyano; halosubstituted Ci-io alkyl; Ci-io alkyl; C2-10 alkenyl;
- C(O)NR4Rl ⁇ ; S(O) 3 H; S(O) 3 R8; CMO alkyl C(O)R ⁇ ; C -10 alkenyl C(0)Rn; C2-IO alkenyl C(O)ORn; C(O)Rn; C(O)ORi2; OC(O) Rn ; NR4C(O)Rn; or two Ri moieties together may form O-(CH2)sO- or a 5 to 6 membered unsaturated ring; t is 0, or an integer having a value of 1 or 2; s is an integer having a value of 1 to 3;
- R4 and R5 are independently hydrogen, optionally substituted Ci-4 alkyl, optionally substituted aryl, optionally substituted aryl Ci-4alkyl, optionally substituted heteroaryl, optionally substituted heteroaryl C ⁇ _4alkyl, heterocyclic, heterocyclicCi -4 alkyl, or R4 and R5 together with the nitrogen to which they are attached form a 5 to 7 member ring which may optionally comprise an additional heteroatom selected from O/N/S; Y is independently selected from hydrogen; halogen; nitro; cyano; halosubstituted Ci-io alkyl; Ci-io alkyl; C2-10 alkenyl; Ci-io alkoxy; halosubstituted Ci-io alkoxy; azide; S(O)tR4; hydroxy; hydroxyC ⁇ _4alkyl; aryl; aryl Ci-4 alkyl; aryloxy; arylC -4 alkyloxy; heteroaryl; heteroaryl
- R8 is hydrogen or Ci-4 alkyl
- RlO is C O alkyl C(O)2R8;
- Rl l is hydrogen, Ci-4 alkyl, optionally substituted aryl, optionally substituted aryl Ci-4alkyl, optionally substituted heteroaryl, optionally substituted heteroarylCi- 4alkyl, optionally substituted heterocyclic, or optionally substituted heterocyclicCi .
- Rl2 is hydrogen, Ci_io alkyl, optionally substituted aryl or optionally substituted arylalkyl; or a pharmaceutically acceptably salt thereof.
- a preferred ring substitution for Ri variable is monosubstituted in the 3- position, or the 4- position, or di-substituted in the 3,4- position.
- the substituent group is suitably an electron withdrawing moiety.
- Ri is nitro, halogen, cyano, trifluoromethyl group, or C(O)NR4R5.
- Y may be substituted in any of the 5 ring positions, preferably the ring with the Y moiety is mono-substituted in the 2-position or 3- position, with the 4- preferably being unsubstituted. If the ring is di-substituted, substituents are preferably in the 2'-, 3 - positions of a monocyclic ring. While both Ri and Y can both be hydrogen, it is preferred that at least one of the rings be substituted, preferably both rings are at least mono-substituted, i.e. n amd m are each equal to 1 or more.
- Y is more preferably a mono-substituted halogen, disubstituted halogen, mono- substituted alkoxy, disubstituted alkoxy, methylenedioxy, aryl, or alkyl, preferably these groups are substituted in the 2 - position or 2'-, 3 -position.
- Exemplified compounds of Formula (la) are:
- Another aspect of the present invention is the novel compounds of Formula (lb), a subset of compounds of Formula (I) useful for treating a chemokine mediated disease.
- This invention also relates to the pharmaceutical compositions comprising a compound of Formula (lb) and a pharmaceutically acceptable diluent or carrier.
- the compounds of Formula (lb) are represented by the structure:
- X is oxygen or sulfur
- Xi is oxygen or sulfur
- Rl is independently selected from hydrogen; halogen; nitro; cyano; halosubstituted Ci-io alkyl; C MO alkyl; C2-10 alkenyl; Ci-io alkoxy; halosubstituted Ci-io alkoxy; azide; S(O) ⁇ R4; hydroxy; hydroxyC ⁇ _4alkyl; aryl; aryl Ci-4 alkyl; aryloxy; arylCi-4 alkyloxy; heteroaryl; heteroarylalkyl; heterocyclic, heterocyclic Ci-4alkyl; heteroaryl Ci-4 alkyloxy; aryl C2-10 alkenyl; heteroaryl C2-10 alkenyl; heterocyclicC2-10 alkenyl; NR4R5; C2-10 alkenyl C(O)NR4R5; C(O)NR4R5; C(O)NR4Rl ⁇ ; S(O) H; S(O) R8; CMO alkyl C(
- R2 is a substituted aryl, heteroaryl, or heterocyclic ring which ring has a functional moiety providing the ionizable hydrogen having a pKa of 10 or less;
- R4 and R5 are independently hydrogen, optionally substituted Ci-4 alkyl, optionally substituted aryl, optionally substituted aryl Ci-4alkyl, optionally substituted heteroaryl, optionally substituted heteroaryl Ci-4alkyl, heterocyclic, heterocyclicCi -.4 alkyl, or R4 and R5 together with the nitrogen to which they are attached form a 5 to 7 member ring which may optionally comprise an additional heteroatom selected from O/N/S;
- Y is independently selected from hydrogen; halogen; nitro; cyano; halosubstituted Ci-io alkyl; Ci-io alkyl; C2-10 alkenyl; Ci-io alkoxy; halosubstituted Ci-io alkoxy; azide; S(O)tR4; hydroxy; hydroxyCi-4alkyl; aryl; aryl Ci-4 alkyl; aryloxy; arylCi-4 alkyloxy; heteroaryl; heteroarylalkyl; heteroarylCi-4 alkyloxy; heterocyclic, heterocyclicCi -4alkyl; aryl C2-10 alkenyl; heteroaryl C2-10 alkenyl; heterocyclicC2-10 alkenyl; NR4R5; C2-IO alkenyl C(O)NR4R5; C(O)NR4R5; C(O)NR4R5; C(O)NR4Rl ⁇ ; S(O) 3 H;
- R8 is hydrogen or Ci-4 alkyl
- RlO is CMO alkyl C(O)2R8.
- Rl 1 is hydrogen, Ci-4 alkyl, optionally substituted aryl, optionally substituted aryl
- Ci-4alkyl optionally substituted heteroaryl, optionally substituted heteroarylCi-
- Rl2 is hydrogen, Ci-io alkyl, optionally substituted aryl or optionally substituted arylalkyl; or a pharmaceutically acceptable salt thereof.
- variable, etc. for Formula (lb) are the same as those defined for
- a preferred ring substitution for Ri is in the 3-position, the 4- position or is preferably di substituted in the 3,4- position.
- the substituent group is suitably an electron withdrawing moiety.
- Ri is nitro, halogen, cyano, trifluoromethyl group, or C(O)NR- ⁇ R5.
- Y may be substituted in any of the 5 ring positions, preferably the ring with the Y moiety is mono-substituted in the 2-position or 3- position, with the 4- preferably being unsubstituted. If the ring is disubstituted, substituents are preferably in the 2' or 3' position of a monocyclic ring. While both Ri and Y can both be hydrogen, it is preferred that at least one of the rings be substituted, preferably both rings are at least mono-substituted, i.e. n amd m are each equal to 1 or more.
- Y is more preferably disubstituted halogen, mono-substituted halogen, disubstituted alkoxy, mono-substituted alkoxy, methylenedioxy, aryl, or alkyl, preferably in the 2'position or 2 ',3 -position.
- Another aspect of the present invention is the novel compounds of Formula (Ic), a subset of compounds of Formula (I) useful for treating a chemokine mediated disease.
- This invention also relates to the pharmaceutical compositions comprising a compound of Formula (Ic) and a pharmaceutically acceptable diluent or carrier.
- the compounds of Formula (Ic) are represented by the structure:
- X is oxygen or sulfur
- Xi is oxygen or sulfur
- Rl is independently selected from hydrogen; halogen; nitro; cyano; halosubstituted Ci-io alkyl; Ci-io alkyl; C2-10 alkenyl; C MO alkoxy; halosubstituted Ci-io alkoxy; azide; S(O)tR4; hydroxy; hydroxyCi-4alkyl; aryl; aryl Ci-4 alkyl; aryloxy; aryl Ci-4 alkyloxy; heteroaryl; heteroarylalkyl; heterocyclic, heterocyclicCi .
- R4 and R5 are independently hydrogen, optionally substituted Ci-4 alkyl, optionally substituted aryl, optionally substituted aryl Ci-4alkyl, optionally substituted heteroaryl, optionally substituted heteroaryl Ci-4 alkyl, heterocyclic, heterocyclic Ci-4 alkyl, or R4 and R5 together with the nitrogen to which they are attached form a 5 to 7 member ring which may optionally comprise an additional heteroatom selected from O/N/S;
- Y is independently selected from halogen; nitro; cyano; halosubstituted Ci-io alkyl; Ci-io alkyl; C2-10 alkenyl; Ci-io alkoxy; halosubstituted Ci- o alkoxy; azide;
- C(O)OR ⁇ 1; C(O)R ⁇ 1; C(O)ORi2; OC(O) Ri 1; NR4C(O)R ⁇ 1; or two Y moieties together may form O-(CH2)sO- or a 5 to 6 membered unsaturated ring; n is an integer having a value of 1 to 3; m is an integer having a value of 1 to 3; R8 is hydrogen or C1.4 alkyl;
- RlO is Ci-10 alkyl C(O)2R8.
- Rl 1 is hydrogen, Ci-4 alkyl, optionally substituted aryl, optionally substituted aryl C ⁇ _4alkyl, optionally substituted heteroaryl, optionally substituted heteroarylCi- 4alkyl, optionally substituted heterocyclic, or optionally substituted heterocyclicCi . 4alkyl;
- a preferred ring substitution for Ri is in the 3-position, the 4- position or di substituted in the 3,4- position.
- Ri is other than hydrogen.
- the substituent group is suitably an electron withdrawing moiety.
- Ri is nitro, halogen, cyano, trifluoromethyl group, or C(O)NR4R5.
- Y may be substituted in any of the 5 ring positions, preferably the ring with the Y moiety is mono-substituted in the 2-position or 3- position, with the 4- preferably being unsubstituted. If the ring is disubstituted, substituents are preferably in the 2' or 3' position of a monocyclic ring. While both Ri and Y can both be hydrogen, it is preferred that at least one of the rings be substituted, preferably both rings are at least mono-substituted, i.e. n amd m are each equal to 1 or more.
- Y is more preferably a mono- substituted halogen, disubstituted halogen, mono-substituted alkoxy, disubstituted alkoxy, methylenedioxy, aryl, or alkyl, preferably with these groups in the 2'position or
- Exemplified compounds of Formula (Ic) are:
- N'-(2-bromophenyl)urea N-[2-Hydroxy-3-cyanophenyl]-N'-[2-methoxyphenyl] urea; N-[2-Hydroxy-3-cyanophenyl]-N'-[3-trifluoromethylphenyl] urea;
- Suitable pharmaceutically acceptable salts are well known to those skilled in the art and include basic salts of inorganic and organic acids, such as hydrochloric acid, hydrobromic acid, sulphuric acid, phosphoric acid, methane sulphonic acid, ethane sulphonic acid, acetic acid, malic acid, tartaric acid, citric acid, lactic acid, oxalic acid, succinic acid, fumaric acid, maleic acid, benzoic acid, salicylic acid, phenylacetic acid and mandelic acid.
- inorganic and organic acids such as hydrochloric acid, hydrobromic acid, sulphuric acid, phosphoric acid, methane sulphonic acid, ethane sulphonic acid, acetic acid, malic acid, tartaric acid, citric acid, lactic acid, oxalic acid, succinic acid, fumaric acid, maleic acid, benzoic acid, salicylic acid, phenylacetic acid and mandelic acid
- pharmaceutically acceptable salts of compounds of Formula (I) may also be formed with a pharmaceutically acceptable cation, for instance, if a substituent group comprises a carboxy moiety.
- Suitable pharmaceutically acceptable cations are well known to those skilled in the art and include alkaline, alkaline earth, ammonium and quaternary ammonium cations.
- halo all halogens, that is chloro, fluoro, bromo and iodo.
- CMO lkyi or “alkyl” - both straight and branched chain radicals of 1 to 10 carbon atoms, unless the chain length is otherwise limited, including, but not limited to, methyl, ethyl, «-propyl, -.o-propyl, n-butyl, sec-butyl, wo-butyl, tert-butyl, n-pentyl and the like.
- cycloalkyl is used herein to mean cyclic radicals, preferably of 3 to 8 carbons, including but not limited to cyclopropyl, cyclopentyl, cyclohexyl, and the like.
- alkenyl is used herein at all occurrences to mean straight or branched chain radical of 2-10 carbon atoms, unless the chain length is limited thereto, including, but not limited to ethenyl, 1-propenyl, 2-propenyl, 2-methyl-l-propenyl, 1- butenyl, 2-butenyl and the like.
- heteroaryl (on its own or in any combination, such as “heteroaryloxy”, or “heteroaryl alkyl”) - a 5-10 membered aromatic ring system in which one or more rings contain one or more heteroatoms selected from the group consisting of N, O or S, such as, but not limited, to pyrrole, pyrazole, furan, thiophene, quinoline, isoquinoline, quinazolinyl, pyridine, pyrimidine, oxazole, thiazole, thiadiazole, triazole, imidazole, or benzimidazole.
- heterocyclic (on its own or in any combination, such as “heterocyclicalkyl”) - a saturated or partially unsaturated 4-10 membered ring system in which one or more rings contain one or more heteroatoms selected from the group consisting of N, O, or S; such as, but not limited to, pyrrolidine, piperidine, piperazine, morpholine, tetrahydropyran, or imidazolidine.
- arylalkyl or “heteroarylalkyl” or “heterocyclicalkyl” is used herein to mean C ⁇ _ ⁇ o alkyl, as defined above, attached to an aryl, heteroaryl or heterocyclic moiety, as also defined herein, unless otherwise indicated.
- Ri moieties or two Y moieties may together form a 5 or 6 membered unsaturated ring
- a napthylene ring system or a phenyl moiety having attached a 6 membered partially unsaturated ring such as a C cycloalkenyl, i.e hexene, or a C5 cyloalkenyl moiety, cyclopentene.
- the compounds of Formula (I), (la), (lb), (Ic), (II) and (HI) may be obtained by applying synthetic procedures, some of which are illustrated in the Schemes below.
- the synthesis provided for in these Schemes is applicable for the producing compounds of Formula (I), (la), (II) and (HI) having a variety of different R, Rj, and Ar groups which are reacted, employing optional substituents which are suitably protected, to achieve compatibility with the reactions outlined herein. Subsequent deprotection, in those cases, then affords compounds of the nature generally disclosed.
- further compounds of these formulas may be prepared by applying standard techniques for functional group intercon version, well known in the art. While the schemes are shown with compounds only of Formula (I) this is merely for illustration purposes only.
- Ortho substituted phenyl ureas shown in 2-scheme 1 may be prepared by standard conditions involving the condensation of commercially available ortho substituted aniline(Aldrich Chemical Co., Milwaukee, Wi) with the commercially available optionally substituted aryl isocyanate (Aldrich Chemical Co., Milwaukee, Wi) in an aprotic solvent (DMF, toluene).
- DMF aprotic solvent
- the l-(RSO2NH)2-(NH2)Ph is not commercially available it can be made by treating the commercially available RSO2CI with the corresponding 2-phenylene diamine in the presence of an base like triethyl amine or NaH in an aprotic solvent (like methylene chloride or DMF).
- the desired 2-amino benzenethiol 8-scheme 3 is not commercially available it can be synthesized by reaction of the phenyl aniline with the thiocyanate anion in the presence of an oxidant(like bromine) to produce the 2-amino benzthiazole 7-scheme 3.
- This thiazole can then be hydrolyzed to the desired 2-amino benzenethiol 8-scheme 3 with a strong base like NaOH in a protic solvent (i.e., EtOH).
- a protic solvent i.e., EtOH
- the thiourea or urea 11 -scheme 4 may be prepared from the commercially available ortho substituted aniline.
- This compound is first protected with a protecting group (tert-butyl dimethyl silyl or benzyl ) by conditions well known in the art(see Greene, T Protecting Groups in Organic Synthesis, Wiley&Sons, New York, 1981).
- This protected aniline is then reacted, in the presence of a base(like triethyl amine or sodium bicarbonate), with either thiophosgene or a solution of phosgene in an aprotic solvent (ie.
- the urea can be formed using a Curtius rearrangement from the corresponding aromatic or thiophene carboxylic acid 12-scheme 5.
- the carboxylic acid is submitted to standard Curtius conditions ((PhO)2PON3, Et3N or C1COCOC1 followed by NaN3) and the intermediate isocyanate is trapped by an appropriately substituted aniline.
- compositions of Formula (I) may be obtained in known manner, for example by treatment thereof with an appropriate amount of acid or base in the presence of a suitable solvent.
- Another aspect of the present invention is the novel synthesis of cyano nitrophenol intermediates. Numerous conversions of aryl halides to aryl cyano derivatives with copper (I) cyanide have been published. However, no examples of an aryl ring with a hydroxy group present were mentioned. Several attempts to obtain a cyano phenol moiety with published results failed. Using known conditions of elevated temperatures, greater than 170°C, such as from 180 to 210° did not yield displacement of the halogen to a cyano moiety. Standard bases, such as DMF and pyridine further provided no desired product.
- Ri is as defined for Formula (I) above, which method comprises reacting a compound of the formula:
- X is halogen with copper (I) cyanide, dimethylformamide, triethylamine and a catalytic amount of dimethylamino pyridine.
- the process is run at reduced temperatures of about 60 to about 80°C.
- X is bromine.
- Example 1 Preparation of N-r2-Hydroxy-4-(methoxycarbonyl)phenvn-N'-phenyl urea N-[2- Hydroxy-4-(methoxycarbonyl)phenyl]-N'-phenyl urea was prepared from methyl-4- amino-3-hydroxybenzoate (200 mg, 1.19 mmol) and phenyl isocyanate (1.19 mmol) according to the procedure noted above in General Method A. The product was purified by precipitation from toluene, and filtering, to afford the titled compound (309 mg, 90%).
- Example 2 Preparation of N-[5-nitro-2-hvdroxyphenyl1 -N'-phenyl urea
- the N-[5-nitro-2- hydroxyphenyl] -N'-phenyl urea was prepared from the 5-nitro 2-hydroxy aniline and phenyl isocyanate according to the procedure in General Method A.
- the product was purified by precipitation from toluene and filtering to afford the titled compound (100 mg, 30%).
- Example 3 Preparation of 3-hvdroxy-4-( [(phenylamino)carbonyl]aminolbenzamide a)Preparation of 0.67 Molar (hereinafter "M") Stock Solutions of Aluminum Amide Reagents To a suspension of the appropriate hydrochloride (0.02 mole (hereinafter “mol")) in dry toluene (20 mL) at about 0°C, was slowly added a solution of (2M, 10 mL) of trimethyl aluminum in toluene. After the addition was complete, the reaction mixture was allowed to warm to room temperature and was stirred for about 1-2 hours until gas evolution has ceased.
- M Molar
- N-(2-hydroxy-4-fluorophenyl)-N'-phenyl urea N-(2-Hydroxy-4-fluorophenyl)-N'-phenyl urea was prepared from 2-amino-5- fluoro phenol (200 mg, 1.57 mmol) and phenyl isocyanate according to the procedure in General Method A. The product was purified by precipitation from toluene and filtering to afford the titled compound (352 mg, 91 %).
- N-(2-Carboxy-4-hydroxyphenyl)-N'-phenyl urea was prepared from 2-amino-5- hydroxy benzoic acid (1 g, 6.53 mmol) according to the procedure in General Method B.
- the reaction mixture was partitioned between ethyl acetate and water.
- the organic phase was washed with brine, dried over MgSO4 and filtered. Removal of solvent under reduced pressure and chromatography of the resulting solid on silica gel (hexane : ethyl acetate, 1:1 to 100% ethyl acetate) gave the titled compound (1.5 g, 84%). !
- N - [2 - Hydroxy - 4- (trifluoromethyl) phenyl] - N' - phenyl urea was prepared from 2-amino-5-trifluoromethylphenol (150 mg, 1.09 mmol) and phenyl isocyanate(1.09 mmol) according to the procedure in General method A. The product was purified by precipitation from methylene chloride and filtering to afford the titled compound ( 230 mg, 87% ).
- N-(2-tert-Butyldimethysilyloxy-4-nitrophenyl)-N'-phenyl-thiourea was prepared by treating a biphasic solution of 2-tert-butyldimethysilyloxy-4-nitroaniline(80 mg, 0.308 mmol) and NaHCO 3 in CHCl 3 :H 2 O(2.5: 1, 7mL) with thiophosgene at 0°C. The solution was allowed to warm to 23 °C and the reaction was continued overnight. The CHCI3 layer was separated and dried over sodium sulfate.
- N-(2-hydroxy-4-nitrophenyl)-N'-phenyl-thiourea was prepared by treating a solution of N-(2-tert-butyldimethysilyloxy-4-nitrophenyl)-N'-phenyl-thiourea (100 mg, 0.248 mmol) in CH3CN (1 mL) with Et 3 N » HF (lOOuL, 0.62 mmol) in acetonitrile for 10 minutes at 23°C.
- Example 10 Preparation of N-(4- nitro 2-(phenylsulfonylamino)phenyl)-N'-phenyl urea a) Preparation of 4-nitro 2-(phenylsulfonylamino) aniline A solution of 4-nitro 1 ,2-phenylene diamine(1.53 g, 10.0 mmol) in DMF was treated with phenyl sulfonyl chloride(1.76 g, 10.0 mmol) and triethyl amine(1.01 g) in DMF for 12 h at 23 °C. The reaction mixture was partitioned between saturated NH 4 C1 and methylene chloride.
- N-(4-Nitro 2-(phenylsulfonylamino)phenyl)-N'-phenyl urea was prepared from 4- nitro 2-(phenylsulfonylamino) aniline(82 mg) and phenyl isocyanate(33 mg) by method A.
- the reaction was cooled and then partitioned between saturated ammonium chloride and 9: 1 methylene chloride and methanol.
- the organic phase was dried over magnesium sulfate, filtered and concentrated in vacuo.
- the residue was purified by column chromatography (ethyl acetate/hexane) to afford desired(30.8 mg, 26%).
- Example 11 Preparation of N-(2-hydroxy-5-nitrophenyl)-N'-(3-methoxy-2-thienyl)urea a) Preparation of 3-methoxy-2-thienylcarboxlic acid To a solution of 3-methoxythiophene (4.81 g, 42.1 mmol) in ether (20 mL) at - 78°C, butyllithium (17 mL, 47.6 mmol) was added. The reaction mixture was stirred at -78°C for 1 hour, then it was warmed to 0 °C for 3 hours.
- N-(2-Hydroxy-4-nitrophenyl)-N'-(2-trifluoromethylphenyl)urea was prepared from 2- hydroxy 4-nitro aniline (154 mg, 1.0 mmol) and 2-trifluoromethyl phenyl isocyanate (1.0 mmol) according to the procedure in General Method B. The product was purified by dilution with methylene chloride and precipitation with hexane. Filtering afforded the title compound (180 mg, 52%). EI-MS m/z 342(M+H) +
- N-(2-hvdroxy-4-nitrophenyl)-N -(3-bromophenyl)urea N-(2-Hydroxy-4-nitrophenyl)-N'-(3-bromo phenyl)urea was prepared from 2-hydroxy 4-nitro aniline (500 mg, 3.24 mmol) and 3-bromo phenyl isocyanate (3.24 mmol)according to the procedure in General Method B. The product was purified by dilution with methylene chloride and precipitation with hexane. Filtering afforded the title compound(0.96g, 87%). EI-MS m/z 350(M-H) -
- Example 20 Preparation of N-(2-hydroxy-4-nitrophenyl)-N'-(4-bromophenyl)urea N-(2-Hydroxy-4-nitrophenyl)-N'-(4-bromo phenyl)urea was prepared from 2-hydroxy 4-nitro aniline (500 mg, 3.24 mmol) and 4-bromo phenyl isocyanate (3.24 mmol) according to the procedure in General Method B. The product was purified by dilution with methylene chloride and precipitation with hexane. Filtering afforded the title compound(0.41 g, 37%). EI-MS m/z 352(M+H) + Example 21
- N-(2-hvdroxy-4-nitrophenyl)-N'-(2-phenylphenyl)urea N-(2-Hydroxy-4-nitrophenyl)-N'-(2-phenylphenyl)urea was prepared from 2-hydroxy 4- nitro aniline (500 mg, 3.24 mmol) and 2-phenyl phenyl isocyanate (3.24 mmol) according to the procedure in General Method B. The product was purified by dilution with methylene chloride and precipitation with hexane. Filtering afforded the title compound(0.22 g, 19%). EI-MS m/z 350(M+H) +
- N-(2-Hydroxy-4-nitrophenyl)-N'-(l-naphthyl)urea was prepared from 2-hydroxy 4-nitro aniline (500 mg, 3.24 mmol) and 1-naphthyl isocyanate (3.24 mmol) according to the procedure in General Method B.
- the product precipitated from methylene chloride and filtered.
- the resulting solid was triturated with 1:3 triethyl amine: methylene chloride.
- the filtrate was concentrated in vacuo.
- the resulting residue was dissolved in methylene chloride and treated with IN HC1 in water.
- N-(2-Hydroxy-4-nitrophenyl)-N'-(2-fluorophenyl)urea was prepared from 2-hydroxy 4- nitro aniline (500 mg, 3.24 mmol) and 2-fluoro phenyl isocyanate (3.24 mmol) according to the procedure in General Method B. The product was purified by dilution with methylene chloride and precipitation with hexane. Filtering afforded the title compound(0.59 g, 31%). EI-MS m/z 292(M+H) +
- N-(2-hydroxy-4-nitrophenyl)-N '-(2-ethoxyphenyl)urea N-(2-Hydroxy-4-nitrophenyl)-N'-(2-ethoxyphenyl)urea was prepared from 2-hydroxy 4- nitro aniline (500 mg, 3.24 mmol) and 2-ethoxy phenyl isocyanate (3.24 mmol) according to the procedure in General Method B. The product was purified by dilution with methylene chloride and precipitation with hexane. Filtering afforded the title compound(0.84 g, 81%). EI-MS m/z 318(M+H) +
- N-(2-hydroxy-4-nitrophenyl)-N'-(2-ethylphenyl)urea N-(2-Hydroxy-4-nitrophenyl)-N'-(2-ethylphenyl)urea was prepared from 2-hydroxy 4- nitro aniline (500 mg, 3.24 mmol) and 2-ethyl phenyl isocyanate (3.24 mmol) according to the procedure in General Method B. The product was purified by dilution with methylene chloride and precipitation with hexane. Filtering afforded the title compound(0.44 g, 43%). EI-MS m/z 302(M+H) +
- Example 28 Preparation of N-(2-hydroxy-4-nitro phenyl)-N'-(2-trifluoromethoxyphenyl)urea N-(2-Hydroxy-4-nitrophenyl)-N'-(2-trifluoromethyloxyphenyl)urea was prepared from 2-hydroxy 4-nitro aniline (500 mg, 3.24 mmol) and 2-trifluoromethoxy phenyl isocyanate (3.24 mmol) according to the procedure in General Method B. The product was purified by dilution with methylene chloride and precipitation with hexane. Filtering afforded the title compound(0.69 g, 60%). EI-MS m/z 358(M+H) + Example 29
- the urea was prepared from 2-hydroxy 4-nitro aniline (500 mg , 3.24 mmol) and 2-methylthio phenyl isocyanate(3.24 mmol) by general Method B.
- the product was purified by dilution with methylene chloride and precipitation with hexane. Filtering afforded the title compound(0.63 g, 61 %).
- the urea was prepared from 2-hydroxy 4-nitro aniline (500 mg, 3.24 mmol) and 2-chloro 6-methyl phenyl isocyanate by general Method B. It was purified by dilution with methylene chloride and precipitation with hexane. Filtering afforded the desired compound(0.31 g, 29%). EI-MS m/z 322(M+H) +
- the urea was synthesized by treatment of N-( 2-hydroxy 4-nitro phenyl) N'-(2- methyl thio phenyl) urea(example 28, 100 mg) with sodium perorate(100 mg) in t- button/water for 12 hours at 23 °C. The product precipitated from the reaction mixture(30 mg, 29%). EI-MS m/z 336(M+H) +
- Example 32 Synthesis of N-(2-hydroxy 4-trifluoromethyl phenyl) N'-(2-bromo phenyl) urea
- the urea was prepared from 2-hydroxy 4-trifluoromethyl aniline(example 7a, 0.171g, 1 mmol) and 2-bromo phenyl isocyanate(l mmol) by general Method B. It was purified by dilution with methylene chloride and precipitation with hexane. Filtering afforded the desired compound(0.25 g, 54%). EI-MS m/z 375(M+H) +
- the urea was prepared from 2-hydroxy 4-carbomethoxy aniline(0.167 g, 1 mmol) and 2-bromo phenyl isocyanate(l mmol) by general Method B. It was purified by dilution with methylene chloride and precipitation with hexane. Filtering afforded the desired compound(0.12 g, 33%). EI-MS m/z 363(M-H) -
- the urea was prepared from 2-hydroxy 4-trifluoromethyl aniline(example 7a, 0.171 g, 1 mmol)) and 2-phenyl phenyl isocyanate by general Method B. It was purified by dilution with methylene chloride and precipitation with hexane. Filtering afforded the desired compound(0.24 g, 64%). EI-MS m/z 373(M+H) +
- the urea was prepared from 2-hydroxy 4-carbomethoxy aniline(0.167 g, 1 mmol) and 2-phenyl phenyl isocyanate(l mmol) by general Method B. It was purified by dilution with methylene chloride and precipitation with hexane. Filtering afforded the desired compound(0.185 g, 50%). EI-MS m/z 363(M-H) ⁇
- Example 36 Synthesis of N-(2-hydroxy 4-nitro phenyl) N'-(2.3-dichloro phenyl) urea
- the urea was prepared from 2-hydroxy 4-nitro aniline(308 mg, 2 mmol) and 2,3-dichloro phenyl isocyanate(2 mmol) by general Method B. It was purified by dilution with methylene chloride and precipitation with hexane. Filtering afforded the title compound(0.5 g, 73%).
- the urea was prepared from 2-hydroxy 4-nitro aniline(308 mg, 2 mmol) and 2,4-dichloro phenyl isocyanate(2 mmol) by general Method B. It was purified by dilution with methylene chloride and precipitation with hexane. Filtering afforded the title compound(0.26 g, 38%). EI-MS m/z 342 (M+H) + Example 38
- the urea was prepared from 4-nitro 2-hydroxy aniline(308 mg, 2 mmol) and 2- chloro phenyl isocyanate(2 mmol) by general Method B. It was purified by dilution with methylene chloride and precipitation with hexane. Filtering afforded the title compound(0.29 g, 47%). EI-MS m/z 308(M+H) +
- the urea was prepared from 4-nitro 2-hydroxy aniline(308 mg, 2 mmol) and 2,4-dibromo phenyl isocyanate(2 mmol) by general Method B. It was purified by dilution with methylene chloride and precipitation with hexane. Filtering afforded the title compound(0.34 g, 39%). EI-MS m/z 430(M+H) +
- Example 40 Synthesis of N-(2-hvdroxynapthyl) N' -(2-bromo phenyl) urea
- the urea was prepared from 1 -amino 2-hydroxy naphthalene(195 mg, 1 mmol) and 2-bromo phenyl isocyanate(l mmol) by general Method B. It was purified by dilution with methylene chloride and precipitation with hexane. Filtering afforded the title compound(0.030 g, 8%). EI-MS m/z 357(M+H) +
- the urea was prepared from 2-hydroxy 4-nitro aniline(308 mg, 2 mmol) and 2- methyl phenyl isocyanate(2 mmol) by general Method B. It was purified by dilution with methylene chloride and precipitation with hexane. Filtering afforded the title compound(0.38 g, 53%). EI-MS m/z 288(M+H) +
- Example 44 Synthesis of N(bis (2-hydroxy 4-nitro phenyl) N'-(dianisdine) diurea
- the urea was prepared from 2-hydroxy 4-nitro aniline(616 mg, 4 mmol) and dianidisdine diisocyanate(2 mmol) by general Method B (except 2 equiv. of 4-nitro 2- hydroxy aniline was used instead of 1 equiv.).
- the product was purified by dilution with methylene chloride and precipitation with hexane. Filtering afforded the title compound( 0.08 g, 6%).EI-MS m/z 605(M+H) +
- Example 45 Synthesis of 4-methylene bis(N-(2-chloro phenyl) N' -(2-hydroxy 4-nitro phenyl) urea)
- the urea was prepared from 2-hydroxy 4-nitro aniline(616 mg, 4 mmol) and 4- methylene bis(N-(2-chloro phenyl) diisocyanate(2 mmol) by general Method B (except 2 equiv. of 4-nitro 2-hydroxy aniline was used instead of lequiv.).
- the product was purified by dilution with methylene chloride and precipitation with hexane. Filtering afforded the title compound(0.10 g, 8%).
- Example 46 Example 46
- the urea was prepared from 2-hydroxy 3,4-difluoro aniline(0.290 g, 2 mmol) and 2-bromo phenyl isocyanate(0.4 g) by general Method B. It was purified by dilution with methylene chloride and precipitation with hexane(0.254 g, 37%). EI-MS m/z 343(M+H)+
- Example 50 Synthesis of N-(2-hvdroxy 3-napthyl) N'-(2-bromo phenyl) urea
- the urea was prepared from 3-amino 2-hydroxy napthalene(0.320 g, 2 mmol) and 2-bromo phenyl isocyanate(.40 g) by general Method B. It was purified by dilution of the with methylene chloride and precipitation with hexane(0.339, 47%).EI-MS m/z 357(M+H)+
- the urea was prepared from 2-hydroxy 4-phenyl aniline(0.185 g, 1 mmol) and 2-bromo phenyl isocyanate(0.198 g) by general Method B. It was purified by dilution of the DMF solution with methylene chloride and precipitation with hexane(215 mg, 56%).EI-MS m/z 383(M+H)+
- the urea was prepared from 2-hydroxy 4-methyl aniline(.274g, 2 mmol) and 2- bromo phenyl isocyanate(0.40 g, 2 mmol) by general Method B. It was purified by dilution of the DMF solution with methylene chloride and precipitation with hexane(249 mg, 39%). EI-MS m/z 319(M-H) "
- Example 54 Synthesis of N-(2-hydroxy 3-carboxylate phenyl) N'-(2-bromo phenyl) urea
- the urea was prepared from 2-hydroxy 3-amino benzoic acid(300 mg, 2 mmol) and 2-bromo phenyl isocyanate by general Method B. It was purified by dilution of the DMF solution with methylene chloride and precipitation with hexane(.287 g, 41%).
- the urea was synthesized by the treatment of 2-iodo benzoic acid(5 g, 20 mmol) with diphenyl phosphoryl azide(l equiv.) and triethyl amine (1 equiv.) in DMF at 80 °C after gas evolution ceased the 5-nitro 2-amino phenol (3 g, 1 equiv.) was added. The reaction was heated overnight at 80°C. The reaction mixture was purified by filtering through a plug of silica with methylene chloride. The desired product was then precipitated out with hexane. Filtering afforded the desired compound(1.08 g, 13%). EI-MS m/z 398(M-H) " Example 57
- the thiourea was synthesized by treatment of the 2-tert-butyldimethylsilyloxy 4-nitro phenyl thioisocyanate(see example 9a , 3.73 mmol) with 2-bromo aniline in toluene at 88°C over 36 h.
- the solution was concentrated and the residue was purified by flash chromatography(EtOAc/Hexane). The fraction slightly lower rf than starting material contained the desired compound. This fraction was concentrated and then treated with triethyl amine hydrofluoride in acetonitrile for 15 minutes at 23 °C.
- the urea was synthesized from 2-(phenylsulfamido) 4-amino benzonitrile(77 mg, 0.28 mmol) and 2-bromo phenyl isocyanate by general Method C. It was purified by column chromatography(ethyl acetate/hexane) to afford the title compound (30 mg, 22%). EI-MS m/z 469(M-H) "
- the sulfonamide was synthesized from styryl sulfonyl chloride(0.01 mol) and o- phenylene diamine(0.01 mol) by general Method C. It was purified by recrystallization from EtOH(1.2 g, 60%)EI-MS m/z 199(M+H)+. b) Synthesis of N-(2-(styryl sulfamido) phenyl) N'-(2-bromo phenyl) urea
- the urea was synthesized from 2-(styryl sulfamido) aniline( 1 mmol) and 2- bromo phenyl isocyanate(l mmol) by general Method B. It was purified by dilution with methylene chloride and precipitation with hexane. Filtering afforded the desired compound(0.309 g, 65%). EI-MS m/z 472(M+H)+ Example 61
- the urea was synthesized from 2-[(3,4-dimethoxyphenyl)sulfonyl amino]phenyl aniline(l mmol) and 2-bromo phenyl isocyanate by general Method B. It was purified by dilution with methylene chloride and precipitation with hexane. Filtering afforded the desired compound(0.062 g, 12%).EI-MS m/z 504(M-H) "
- the urea was synthesized from 2-[(4-acetamidophenyl)sulfonylamino]phenyl aniline(l mmol) and 2-bromo phenyl isocyanate(l mmol) by general Method B. It was purified by dilution with methylene chloride and precipitation with hexane. Filtering afforded the desired compound(0.12 g, 24%). EI-MS m/z 501(M-H) "
- the sulfonamide was synthesized from 2-thiophene sulfonyl chloride(0.01 mol) and o-phenylene diamine(0.01 mol) by general Method C. It was purified by recrystallization from EtOH(0.77 g, 30%). EI-MS m/z 255 (M+H)+ b) Synthesis of N-(2-(2-thiophene sulfonyl amino phenyl) N'-(2-bromo phenyl) urea
- the urea was synthesized from 2-( 2-thiophene sulfonyl amino) aniline( 1 mmol) and 2-bromo phenyl isocyanate(l mmol) by general Method B. It was purified by dilution with methylene chloride and precipitation with hexane. Filtering afforded the desired compound(0.29 g, 64%). EI-MS m/z 450(M-H) " Example 64
- the sulfonamide was synthesized from 3-tolyl sulfonyl chloride(0.01 mol) and o-phenylene diamine(0.01 mol) by general Method C. It was purified by recrystallization from EtOH(0.73g, 28%).EI-MS m/z 263 (M+H)+ b) Synthesis of N-(2-((3-tolyl sulfonyl amino) phenyl) N'-(2-bromo phenyl) urea
- the urea was synthesized from 2-(3-tolyl sulfonyl amino) aniline(l mmol) and 2-bromo phenyl isocyanate(l mmol) by general Method B. It was purified by dilution with methylene chloride and precipitation with hexane. It was recrysallized two times with EtOH(25 mg, 5%). EI-MS m/z 458(M-H) "
- Example 65 Synthesis of N-(2-(8-quinolinyl sulfonyl amino) phenyl) N'-(2-bromo phenyl) urea a) Synthesis of 2-(8-quinolinyl sulfonyl amino) aniline The sulfonamide was synthesized from 8-quinolinyl sulfonyl chloride(0.01 mol) and o-phenylene diamine(0.01 mol) by general Method C. It was purified by recrystallization from EtOH(0.82 g, 27%).EI-MS m/z 300 (M+H).
- the sulfonamide was synthesized from benzyl sulfonyl chloride(0.01 mol) and o-phenylene diamine(0.01 mol) by general Method C. It was purified by recrystallization from EtOH(0.87g, 33%). EI-MS m/z 263(M+H)+. b) Synthesis of N-(2-( benzyl sulfonyl amino) phenyl) N '-(2-bromo phenyl) urea
- the urea was synthesized from 2-( benzyl sulfonyl amino) aniline( 1 mmol) and 2-bromo phenyl isocyanate(lmmol) by general Method B. It was purified by dilution with methylene chloride and precipitation with hexane. Filtering afforded the desired compound(0.11 g, 23%). EI-MS m/z 460 (M+H)+
- Example 68 Preparation of N-r2-hvdroxy-5-cvanophenyl1-N'-[2-bromophenyl1 urea a) Preparation of 2-amino-4-cyanophenol To a solution of 2-nitro-4-cyanophenol(10g, 61 mmol) in methanol(250mL) was added 10% Pd/C (lg). The mixture was flushed with argon, then hydrogen was bubbled through the solution for 10 min. and a hydrogen atmosphere was maintained at balloon pressure overnight. The mixture was filtered through celite and the celite was washed with methanol.
- N-[2-hydroxy-3-fluorophenyl]-N'-[2-bromophenyl] urea was prepared from 2- amino-3-fluorophenol (254mg, 2.00 mmol) according to the procedure in General Method B. The product was purified by precipitation from methylene chloride/ hexane(l/20) and filtering. (500 mg, 77%).
- N-2-[l -hydroxyfluorene] -N - [2-bromophenyl] urea N-2-[l -hydroxyfluorene] -N - [2-bromo phenyl] urea was prepared from 2- amino-1- hydroxyfluorene (170mg, 0.86 mmol) according to the procedure in General Method B. The product was purified by chromatography of the resulting solid on silica gel (30%EtOAc/ Hexane) to give the desired product (300mg, 84.5%).
- N-3-[2-hvdroxy-9,10-anthraquinonvH-N'-F2-bromophenvI1 urea N-3- [2-Hydroxy-9,10-anthraquinonyl]-N'-[2-bromophenyl] urea was prepared from 2- hydroxy-3-aminoanthraquinone(480mg, 2.00 mmol) according to the procedure in General Method B. The product was purified by precipitation from methylene chloride/ hexane(l/20) and filtering. (610mg, 70%).
- N-[2-hydroxy-3-fluoro-5-bromophenyl]-N'-[2-bromophenyl] urea was prepared from 2-amino-6-fluoro-4-bromophenol (254mg, 2.00 mmol) according to the procedure in General Method B. The product was purified by precipitation from methylene chloride/ hexane(l/20) and filtering. (500 mg, 77%).
- N-[2-hydroxy-3- trifluoromethylphenyl]-N'-[2-bromophenyl] urea N-[2-hydroxy-3-trifluoromethylphenyl]-N'-[2-bromophenyl] urea was prepared from 2-amino-6-trifluoromethylphenol (280mg, 1.60 mmol) according to the procedure in General Method B. The product was purified by precipitation from methylene chloride/ hexane(l/20) and filtering. (390mg, 65%).
- N-[3,4 diphenyl-2-hydroxyphenyl]-N'-[2-bromophenyl] urea was prepared from 2-amino-5,6 diphenylphenol (50mg, 0.19 mmol) according to the procedure in General Method B. The product was purified by precipitation from methylene chloride/ hexane(l/20) and filtering (61mg, 69%).
- N-[2-hydroxy-3-glycinemethylestercarbonylphenyl]-N'-[2-bromophenyl] urea was prepared from 6-glycinemethylestercarbonyl-2-aminophenol (50mg, 0.22 mmol), purchased from the University of New Hampshire, according to the procedure in General Method B. The product was purified by precipitation from methylene chloride/ hexane(l/20) and filtering (65mg, 69%).
- Example 77 Preparation of N-[2-hvdroxy-3-glycinecarbonylphenyll-N'-[2-bromophenvn urea N- [2-Hydroxy-3-glycinecarbonylphenyl]-N'-[2-bromophenyl] urea was prepared from N- [2-hydroxy-3-glycinemethylestercarbonylphenyl]-N'-[2-bromophenyl] urea (50mg, 0.12 mmol) by stirring in a 3/1 ratio of methanol/water (10 mL). Addition of 1 equiv. of lithium hydroxide was added and stirring continued until the starting material had disappeared. (45mg, 92%).
- Example 78 Preparation of N-f2-hvdroxy-3,5-dichlorophenyl1-N'-[2-bromophenyl1 urea a) Preparation of 2-amino-4,6-dichlorophenol A mixture of 4,6-dichloro-2-nitrophenol( 1 g, 4.8mmol) and tin (II) chloride (3.2 g, 14.4mmol) in ethanol(50mL) was heated at 80°C under argon. After 2 hours, the starting material had disappeared and the solution was allowed to cool down and then poured into ice. The pH was made slightly basic (pH7-8), by addition of solid NaOH, before being extracted with ethyl acetate.
- Example 80 Preparation of N-[2-hydroxy-4-naphthalenesulfonic acid1-N'-[2-bromophenyl1 urea N-[2-hydroxy-4-naphthalenesulfonic acid] -N - [2-bromophenyl] urea was prepared from l-amino-2-hydroxy-4-naphthalensulfonic acid (0.48g, 2.0 mmol) according to the procedure in General Method B and the addition of lmL of triethylamine. The product was purified by precipitation from methylene chloride/ hexane(l/20) and filtering. (690 mg, 79%).
- N-3-[2-hydroxy-5-naphthalensulfonic acid] -N - [2-bromophenyl] urea was prepared from 2-amino-3-hydroxy-6-naphthalensulfonic acid (0.48g, 2.0 mmol) according to the procedure in General Method B and the addition of lmL of triethylamine. The product was purified by precipitation from methylene chloride/ hexane(l/20) and filtering. (715 mg, 82%).
- Example 84 Preparation of N-[2-hydroxy-4-cvanophenyl1-N'-[2-bromophenyl] urea a) Preparation of 2-nitro-5-cyanophenol 3-cyanophenol (2.38g, 20mmol) was dissolved in methylene chloride(40mL) followed by the addition of sodium nitrate (1.88g, 22mmol). The addition of sulfuric acid (20mL/ 3M) was then made, followed by addition of a catalytic amount of sodium nitrite. The mixture was allowed to stir. After 24 hours, the reaction mixture was diluted with methylene chloride and extracted with water. The organic layer was dried over MgSO 4 and filtered.
- N-[2-hydroxy-4-cvanophenyl]-N'-[2-phenylphenyl1 urea was prepared from 2-amino-5- cyanophenol (170 mg, 1.27 mmol) according to the procedure in General Method B. The product was purified by precipitation from methylene chloride/ hexane(l/20) and filtering. (150mg, 85%).
- N-[2-hvdroxy-4-cvanophenyl1-N'-[2-trifluoromethylphenyl] urea was prepared from 2- amino-5-cyanophenol (60mg, 0.45 mmol) according to the procedure in General Method B. The product was purified by precipitation from methylene chloride/ hexane(l/20) and filtering. (1 lOmg, 76%).
- N-[2-hvdroxy-4-cvanophenyl1-N'-[3-trifluoromethylphenvI1 urea was prepared from 2- amino-5-cyanophenol (60mg, 0.45 mmol) according to the procedure in General Method B. The product was purified by precipitation from methylene chloride/ hexane(l/20) and filtering. (114mg, 79%).
- N-[2-hvdroxy-4-cvanophenyll-N'-[4-trifluoromethylphenyl1 urea was prepared from 2- amino-5-cyanophenol (60mg, 0.45 mmol) according to the procedure in General Method B. The product was purified by precipitation from methylene chloride/ hexane(l/20) and filtering. (108mg, 75%).
- N-[2-hydroxy-4-ethylphenyl]-N'-[2-bromophenyl] urea N-[2-Hydroxy-4-ethylphenyl]-N'-[2-bromo phenyl] urea was prepared from 2- amino-5-ethylphenol (274mg, 2.00 mmol) according to the procedure in General Method B. The product was purified by precipitation from methylene chloride/ hexane(l/20) and filtering. (520 mg, 77%).
- 2-Phenylaminocarbonylphenol (5.00g, 23 mmol) was dissolved in methylene chloride(40mL) followed by the addition of sodium nitrate (2.20g, 25.5 mmol). The addition of sulfuric acid (30mL/ 3M) was then made, followed by addition of a catalytic amount of sodium nitrite. The mixture was allowed to stir. After 24 hours, the reaction mixture was diluted with methylene chloride and extracted with water.
- N-[2-hydroxy 3-Phenylaminocarbonyl phenyl]-N'-[2-bromo phenyl] urea was prepared from 2-amino-6-phenylaminocarbonylphenol (456mg, 2.00 mmol) according to the procedure in General Method B. The product was purified by precipitation from methylene chloride/ hexane(l/20) and filtering. (800mg,94%).
- N-[2-hydroxy 3-cyano 4-methyl phenyl]-N'-[2-bromophenyl] urea was prepared from 2-amino-5-methyl-6-cyano phenol (50mg, 0.34 mmol) according to the procedure in General Method B. The product was purified by precipitation from methylene chloride/ hexane(l/20) and filtering. (70mg, 60%).
- 3-Hydroxybenzophenone (3.00g, 15.1mmol) was dissolved in methylene chloride(40mL) followed by the addition of sodium nitrate (1.42g, 16.7mmol). The addition of sulfuric acid (25mI7 3M) was then made, followed by addition of a catalytic amount of sodium nitrite. The mixture was allowed to stir. After 24 hours, the reaction mixture was diluted with methylene chloride and extracted with water.
- N-[2-hydroxy 3-carboxyphenyl phenyl]-N'-[2-bromophenyl] urea was prepared from 3-amino-2-hydroxybenzophenone (250mg, 1.20 mmol) according to the procedure in General Method B. The product was purified by precipitation from methylene chloride/ hexane(l/20) and filtering. (200mg, 78%).
- N-[3-benzyloxy-2-hydroxyphenyl]-N'-[2-bromophenyl] urea was prepared from 2-nitro-6-benzyloxy phenol (430mg, 2.00 mmol) according to the procedure in General Method B. The product was purified by precipitation from methylene chloride/ hexane(l/20) and filtering. (630mg, 76%).
- Example 98 Preparation of N-3-[2-hydroxy-5-indanone1-N'-r2-bromophenyll urea a) Preparation of 2-hydroxy-3-nitro-5-indanone 2-Hydroxy-5-indanone(3.00g, 20.0mmol) was dissolved in methylene chloride(40mL) followed by the addition of sodium nitrate (1.95g, 21.0mmol). The addition of sulfuric acid (25mL/ 3M) was then made, followed by addition of a catalytic amount of sodium nitrite. The mixture was allowed to stir. After 24 hours, the reaction mixture was diluted with methylene chloride and extracted with water. The organic layer was dried over MgSO 4 and filtered.
- 2-Hydroxycinnamic acid (2.00g, 12.3 mmol) was dissolved in dimethyl formamide(lOmL) followed by the addition of benzotriazol-1-yloxy- tris(dimethylamino)phosphonium hexafluorophosphate (5.4g, 12.3 mmol) and triethylamine ( 1.7mL, 12.3mmol). Ammonia gas was bubbled into the reaction mixture for 30 minutes. The mixture was allowed to stir for 24 hours, the reaction mixture was diluted with methylene chloride and extracted with water. The organic layer was dried over MgSO 4 and filtered.
- 3-Hydroxycinnamic acid (2.00 g, 12.3 mmol) was dissolved in dimethyl formamide( 10 mL) followed by the addition of benzotriazol- 1 -yloxy- tris(dimethylamino)phosphonium hexafluorophosphate (5.4g, 12.3 mmol) and triethylamine ( 1.7 mL, 12.3mmol). Ammonia gas was bubbled into the reaction mixture for 30 minutes. The mixture was allowed to stir for 24 hours, the reaction mixture was diluted with methylene chloride and extracted with water. The organic layer was dried over MgSO 4 and filtered.
- N-[2-hydroxy 4-(phenyl amino carboxy) phenyl] -N '- [2-bromophenyl] urea was prepared from 5-(phenyl amino carboxy) 2-amino phenol (0.50 mmol) according to the procedure in General Method B. The product was purified by precipitation from methylene chloride/ hexane(l/20) and filtering. (150 mg, 70%).
- Example 104 N-[2-hydroxy 4-(phenyl amino carboxy) phenyl] -N '- [2-bromophenyl] urea was prepared from 5-(phenyl amino carboxy) 2-amino phenol (0.50 mmol) according to the procedure
- N-[4-aminocarbonyl-2-hvdroxyphenyl1-N'-[2-bromophenyl1 urea N- [4-Aminocarbonyl -2-hydroxyphenyl]-N'-[2-bromophenyl] urea was prepared from 5- aminocarbonyl-2-amino phenol (304 mg, 0.50 mmol) according to the procedure in General Method B. The product was purified by precipitation from methylene chloride/ hexane(l/20) and filtering. (440 mg, 62%).
- N-(2-Hydroxy-3-fluoro-4-trifluoromethylphenyl)-N'-(2-bromophenyl)urea was prepared from 4-trifluoromethyl-3-fluoro-2-hydroxyaniline (239 mg, 1.2 mmol) and 2- (bromophenyl)isocyanate (243 mg, 1.2 mmol) according to the procedure in General Method B. Removal of solvent under reduced pressure and chromatography of the resulting solid on silica gel (hexane:ethyl acetate) gave the title compound (20 mg, 4%). EI-MS m/z 391 (M-H) "
- N-(2-Hydroxy-3-iodophenyl)-N'-(2-bromophenyl)urea was prepared from 3- iodo-2-hydroxyaniline (200 mg, 0.85 mmol) and 2-(bromophenyl)isocyanate (169 mg, 0.85 mmol) according to the procedure in General Method B. Removal of solvent under reduced pressure and chromatography of the resulting solid on silica gel (hexane:ether) gave the title compound (40 mg, 11%).
- Example 111 Preparation of N-[2-[(2-acetamido-4-methylthiazol-5-yl)sulfonylamino1phenyl1-N'-(2- bromophenvPurea a) Preparation of [2-[(2-acetamido-4-methyl-5-thiazole)sulfonamido]aniline] The title compound was prepared using 2-acetamido-4-methyl-5- thiazolesulfonyl chloride (1 equiv.) according to General Method C. A solid precipitated from the reaction mixture and was filtered to give the desired aniline (1.68 g, 52%). ES-MS m/z 327 (M+H) + . b) Preparation of N-[2-[(2-acetamido-4-methylthiazol-5- yl)sulfonylamino]phenyl]-N'-(2-bromophenyl)urea
- the title compound was prepared from [2-[(2-acetamido-4-methyl-5- thiazole)sulfonamido] aniline] (1.68 g,5.14 mmol) and 2-(bromophenyl)isocyanate (1.02 g, 5.14 mmol) according to General Method B.
- the product was precipitated from ethyl acetate/hexane (220 mg, 8%). EI-MS m/z 524 (M+H) + .
- N-[2-hvdroxy-4-cvanophenyll-N'-F4-phenylphenyll urea N-[2- Hydroxy-4-cyanophenyl]-N'-[4-phenylphenyl] urea was prepared from 2-amino-5- cyanophenol (60mg, 0.45 mmol) according to the procedure in General Method B. The product was purified by precipitation from methylene chloride/ hexane(l/20) and filtering. (135 mg, 75%).
- N-[2-Hydroxy-4-cyanophenyl]-N'-[2-methoxyphenyl] urea was prepared from 2-amino-5-cyanophenol (60mg, 0.45 mmol) according to the procedure in General Method B. The product was purified by precipitation from methylene chloride/ hexane(l/20) and filtering (105mg, 83%).
- Example 115 Preparation of N-[2-hvdroxy-4-cyanophenyl1-N -[3-methoxyphenyll urea N-[2- Hydroxy-4-cyanophenyl]-N'-[3-methoxyphenyl] urea was prepared from 2-amino-5- cyanophenol (60mg, 0.45 mmol) according to the procedure in General Method B. The product was purified by precipitation from methylene chloride/ hexane(l/20) and filtering. (102mg, 80%). 1H NMR (CD 3 OD): ⁇ 8.25 (d, IH), 7.25-7.08 (m, 3H), 7.04 (s, IH), 6.90 (t, IH), 6.58 (d, IH).
- N-[2-hydroxy-5-trifluoromethylphenyl]-N'-[2-bromophenyl] urea was prepared from 2-amino-4-trifluoromethylphenol (354mg, 2.00 mmol) according to the procedure in General Method B. The product was purified by precipitation from methylene chloride/ hexane(lequiv./20equiv.) and filtering. (490mg, 65%).
- Example 118 Preparation of N-[2-hvdroxyphenyl]-N'-[2-bromophenyl] urea N-[2- hydroxyphenyl]-N'-[2-bromo phenyl] urea was prepared from 2- amino-phenol (141mg, 1.30 mmol) according to the procedure in General Method B. The product was purified by precipitation from methylene chloride/ hexane(l/20) and filtering. (300mg,75%). 1H NMR (CD 3 OD): ⁇ 8.05 (d, IH), 7.49 (d, IH), 7.25 (t, 2H), 6.96 (t, IH), 6.90 (t, 2H), 6.68 (t, IH).
- Trans-2-styrlphenol 500 mg, 2.55 mmol was dissolved in methylene chloride(40mL) followed by the addition of sodium nitrate (240 mg, 2.81mmol). The addition of sulfuric acid (3 mL of 3M) was then made, followed by addition of a catalytic amount of sodium nitrite. The mixture was allowed to stir. After 24 hours, the reaction mixture was diluted with methylene chloride and extracted with water. The organic layer was dried over MgSO and filtered. The solvent was evaporated and chromatography of the resulting solid on silica gel (4%MeOH7 CH 2 C1 2 ) gave the desired product (200 mg, 36 %).
- Example 120 Preparation of N-[2-hydroxy-3,4-dichlorophenyl1-N'-[2-methoxyphenyl] urea N-[2-hydroxy-3,4-dichlorophenyl]-N'-[2-methoxyphenyl] urea was prepared from 2-amino-5,6-dichlorophenol (80mg, 0.50 mmol, example 82b) according to the procedure in General Method B. The product was purified by precipitation from methylene chloride/ hexane(l/20) and filtering. (125mg,77%). 1H NMR (CD 3 OD): ⁇ 8.02 (d, IH), 7.79 (d, IH), 7.05-6.86 (m, 4H), 3.92 (s, 3H).
- Example 121 Example 121
- N-[2-hvdroxy-3,4-dichlorophenyl1-N , -[4-methoxyphenyl] urea was prepared from 2- amino-5,6-dichlorophenol (80mg, 0.50 mmol, example 82b) according to the procedure in General Method B. The product was purified by precipitation from methylene chloride/ hexane(lequiv./20equiv.) and filtering. (120mg, 74%).
- Example 122 Preparation of N-[2-hvdroxy-3,4-dichlorophenyn-N'-[3-trifluoromethylphenyl1 urea
- N-[2-hydroxy-3,4-dichlorophenyl]-N'-[3-trifluoromethylphenyl] urea was prepared from 2-amino-5,6-dichlorophenol (80mg, 0.50 mmol, example 82b) according to the procedure in General Method B. The product was purified by precipitation from methylene chloride/ hexane(lequiv./20equiv.) and filtering. (130mg, 71%).
- [2-hydroxy-3,4-dichlorophenyl]-N'-[2-phenylphenyl] urea was prepared from 2-amino- 5,6-dichlorophenol (80mg, 0.50 mmol, example 82b) according to the procedure in General Method B. The product was purified by precipitation from methylene chloride/ hexane(lequiv./20equiv.) and filtering. (1 lOmg, 59%).
- Example 124 Preparation of N-[2-hvdroxy-3,4-dichlorophenyll-N'-[2,3-dichlorophenyl1 urea
- N- [2-Hydroxy-3,4-dichlorophenyl]-N'-[2,3-dichlorophenyl] urea was prepared from 2- amino-5,6-dichlorophenol (80mg, 0.50 mmol, example 82b) according to the procedure in General Method B. The product was purified by precipitation from methylene chloride/ hexane(lequiv./20equiv.) and filtering. (130mg, 71%).
- Example 125 Example 125
- N-[2-hydroxy-4-isopropylphenyl]-N'-[3-trifluoromethylphenyl] urea was prepared from 2-amino-5-isopropylphenol (75mg, 0.50 mmol) according to the procedure in General Method B. The product was purified by precipitation from methylene chloride/ hexane(l equiv ./20equiv.) and filtering. (140mg, 83%).
- Example 126 Preparation of N-[2-hvdroxy-3-naphthyl]-N , -[2.3-dichlorophenyl1 urea N-[2- hydroxy-3-naphthyl]-N'-[2,3-dichlorophenyl] urea was prepared from 3-amino 2- naphthol (160mg, 1.00 mmol) according to the procedure in General Method B. The product was purified by precipitation from methylene chloride/ hexane(lequiv./20equiv.) and filtering. (285mg, 82%).
- Example 127 Preparation of N-[2-[(2,3-Dichlorothien-5-yl)]sulfonylamino]phenyl]-N -(2- bromophenvDurea a) Preparation of [2-[(2,3-Dichlorothien-5-yl)]sulfonylaminoaniline]
- the title compound was prepared according to General Method C using 2,3- dichlorothiophene-5-sulfonyl chloride ((1 eq).
- the product was purified by flash chromatography on silica gel (ethyl acetate/hexane 20/80-methylene chloride: methanol 90/10) (1.25 g, 39 %).
- the title compound was prepared from [2-[(2,3-dichlorothien-5- yl)]sulfonylaminoaniline (1.25 g,3.9 mmol) and 2-(bromophenyl)isocyanate (768 mg, 3.9 mmol) according to General Method B.
- the product was purified by flash chromatography on silica gel (ethyl acetate:hexane 30/70) (272 mg, 13 %) EI-MS m/z 520 (M-H) "
- the title compound was prepared from [2-(3,5-bistrifluoromethylphenyl) sulfonylaminoaniline (591 mg, 1.5 mmol) and 2-bromophenylisocyanate (305 mg, 1.5 mmol) according to General Method B.
- the product was purified by flash chromatography on silica gel (ethyl acetate: hexane 30/70) (10 mg, 1 %).EI-MS m/z 580 (M-H) "
- Example 129 Preparation of N-[2-f(2-Benzyl)sulfonylamino]-(5-trifluoromethyl)phenvn-N'-(2- bromophenvDurea a) Preparation of [(4-Benzylsulfonylamino)-(3 -nitro)-benzotrifluoride] 4-Amino-3-nitro-benzotrifluoride (1.0 g, 4.85 mmol) was mixed in DMF and the reaction mixture was cooled to 0°C. Sodium hydride (175 mg, 7.28 mmol) was added to the cold mixture and allowed to mix for ten minutes ( a deep red color was noted).
- the title compound was prepared from [(4-benzylsulfonylamino)-(3-amino)- benzotrifluoride (210 mg, 0.64 mmol) and 2-bromophenylisocyanate (126 mg, 0.64 mmol) according to the procedure in General Method B.
- the product was purified by flash chromatography on silica gel (ethyl acetate:hexane 30/70) (70 mg, 21%) EI-MS m/z 526 (M-H) "
- the title compound was prepared according to General Method C using 4- phenoxyphenylsulfonyl chloride (969 mg, 3.6 mmol) and o-phenylenediamine (300 mg, 2.77 mmol).
- the reaction mixture was partitioned between water (200 ml) and toluene: methylene chloride (1:3).
- the organic phase collected and the methylene chloride evaporated leaving the toluene. Hexane added and the product precipatated from solution.
- Example 132 Preparation of N-fF2-( 1 S)- 10-Camphorsulfonylamino]phenyl]-N'-(2-bromophenvI)urea a) Preparation of 2-((lS)-10-Camphorsulfonylamino)aniline The title compound was prepared according to General Method C using (1 S)(+)- 10-Camphorsulfonyl chloride ( 1.16 g, 4.6 mmol) and o-phenylenediamine
- the title compound was prepared from [2-(lS)-10- camphorsulfonylamino] aniline (130 mg, 0.4 mmol) and 2-(bromophenyl)isocyanate (80 mg, 0.4 mmol) according to the procedure in General Method B. The solvent was evaporated and product was precipitated from methylene chloride:hexane. (200 mg, 95
- Example 135 Preparation of N-(2-hydroxy-4-azidophenyl)-N'-(2-iodophenyl)urea a) Preparation of N-(2-hydroxy-4-aminophenyl)-N'-(2-iodophenyl)urea To a solution of N-(2-hydroxy-4-nitrophenyl)-N'-(2-iodophenyl)urea (220 mg, 0.55 mmol) in ethanol (15 mL), Tin chloride (522 mg, 2.75 mmol) was added. The reaction mixture was stirred at reflux for 16 hours then cooled to room temperature. The reaction mixture was basified to pH 8 with aq. NaHCO 3 then extracted with ethyl acetate (3x).
- Example 137 Preparation of N-F2-hvdroxy-3-cvanophenyl]-N '-[2-methoxyphenyll urea
- N-[2- hydroxy-3-cyanophenyl]-N'-[2-methoxyphenyl] urea was prepared from 2-amino-6- cyanophenol (134mg, 1.00 mmol) according to the procedure in General Method B. The product was purified by precipitation from methylene chloride/ hexane(lequiv./20equiv.) and filtering. (230 mg, 81%).
- Example 138 Preparation of N-[2-hvdroxy-3-cvanophenyl1-N'-[3-trifluoromethylphenyl1 urea
- N- [2-hydroxy-3-cyanophenyl]-N'-[3-trifluoromethylphenyl] urea was prepared from 2- amino-6-cyanophenol (134mg, 1.00 mmol, example 83a) according to the procedure in General Method B. The product was purified by precipitation from methylene chloride/ hexane(lequiv./20equiv.) and filtering. (280mg, 87%).
- Example 139 Preparation of N-r2-hydroxy-3-cyanophenyl1-N'-[2-phenylphenyl1 urea N-[2- hydroxy-3-cyanophenyl]-N'-[2-phenylphenyl] urea was prepared from 2-amino-6- cyanophenol (134mg, 1.00 mmol, example 83a) according to the procedure in General Method B. The product was purified by precipitation from methylene chloride/ hexane(lequiv./20equiv.) and filtering. (270mg, 82%). 1H NMR (CD 3 OD): ⁇ 7.81 (d, IH), 7.75 (d, IH), 7.56-7.15 (m, 9H), 6.91 (t, IH).
- Example 140 Preparation of N-[2-hydroxy-3-cvanophenyl]-N -[2,3-dichlorophenyll urea N-[2- hydroxy-3-cyanophenyl]-N'-[2,3 dichlorophenyl] urea was prepared from 2-amino-6- cyanophenol (134mg, 1.00 mmol, example 83a) according to the procedure in General Method B. The product was purified by precipitation from methylene chloride/ hexane(lequiv./20equiv.) and filtering. (300mg, 93%). 1H NMR (CD 3 OD): ⁇ 8.11 (d, IH), 8.01 (d, IH), 7.33-7.25 (m, 3H), 7.00 (t, IH).
- Example 141 Preparation of N-[2-hvdroxy-4-isopropylphenyl]-N'-[2.3-dichlorophenyl1 urea
- N- [2-hydroxy-4-isopropylphenyl]-N'-[2,3-dichlorophenyl] urea was prepared from 2- amino-5-isopropylphenol (150 mg, 1.00 mmol, example 128a) according to the procedure in General Method B. The product was purified by precipitation from methylene chloride/ hexane(lequiv./20equiv.) and filtering (285mg, 84%).
- Example 142 Preparation of N-[2-hvdroxy-4-isopropylphenvn-N'-[2-chloro-5-trifluoromethylphenyl1 urea N-[2-hydroxy-4-isopropylphenyl]-N'-[2-chloro-5-trifluoromethylphenyl] urea was prepared from 2-amino-5-isopropylphenol (150mg, 1.00 mmol, example 128a) according to the procedure in General Method B. The product was purified by precipitation from methylene chloride/ hexane(lequiv./20equiv.) and filtering. (275mg, 82%).
- N-[2-hydroxy-3-phenylphenyl]-N'-[2,3-dichlorophenyl] urea was prepared from 2- amino-6-phenylphenol (92.5mg, 0.50 mmol) according to the procedure in General Method B. The product was purified by precipitation from methylene chloride/ hexane(lequiv./20equiv.) and filtering. (150mg,81%).
- Example 144 Preparation of N-[2-hydroxy-5-nitrophenyl1-N'-[2-methoxyphenyl1 urea N-[2- hydroxy-5-nitrophenyl]-N'-[2-methoxyphenyl] urea was prepared from 2-amino-4- nitrophenol (154 mg, 1.00 mmol) according to the procedure in General Method B. The product was purified by precipitation from methylene chloride/ hexane( lequiv./20equiv.) and filtering. (270 mg, 89%). 1H NMR (CD 3 OD): ⁇ 9.10 (s, IH), 8.10 (d, IH), 7.85 (d, IH), 7.08-6.88 (m, 4H), 3.96 (s, 3H).
- Example 145 Preparation of N-[2-hvdroxy-5-nitrophenyl]-N'-[3-trifluoromethylphenyl1 urea
- N- [2-hydroxy-5-nitrophenyl]-N'-[3-trifluoromethylphenyl] urea was prepared from 2- amino-4-nitrophenol (154 mg, 1.00 mmol) according to the procedure in General Method B. The product was purified by precipitation from methylene chloride/ hexane(lequiv./20equiv.) and filtering. (290 mg, 85%).
- Example 146 Preparation of N-[2-hydroxy-5-nitrophenyl1-N'-[2-phenylphenyl1 urea N-[2- hydroxy-5-nitrophenyl]-N'-[2-phenylphenyl] urea was prepared from 2-amino-4- nitrophenol (154 mg, 1.00 mmol) according to the procedure in General Method B. The product was purified by precipitation from methylene chloride/ hexane(lequiv./20equiv.) and filtering. (285 mg, 81%). 1H NMR (CD 3 OD): ⁇ 8.09 (s, IH), 7.86 (d, IH), 7.58-7.20 (m, 9H), 6.95 (d, IH).
- Example 148 Preparation of N-[2-hvdroxy-5-ethylsulfonylphenyl]-N'-[2,3-dichlorophenyl1 urea
- N- [2-hydroxy-5-ethylsulfonylphenyl]-N'-[2,3-dichlorophenyl] urea was prepared from 2- amino-4-(ethylsulfonyl)phenol (185 mg, 1.00 mmol) according to the procedure in General Method B.
- the product was purified by precipitation from methylene chloride/ hexane(lequiv./20equiv.) and filtering. (310 mg, 84%).
- Example 149 N-[2-(2-Amino-(4-trifluoromethyl) phenyl) sulfonylamino] phenyl]- N'- (2- bromophenyl)urea EI-MS m/z 527 (M-H) " .
- Example 150 N-[2-(aminosulfonyl phenyl) 3-amino phenyl] N'-(2-bromo phenyl) ureaEI-MS m/z 426 (M+H) + .
- N-(5-Chloro-2-hydroxy-4-nitrophenyl)-N'-phenylurea The following compounds of Formula (I) may be prepared in accordance with the examples and schemes as described above, or as indicated by their respective citations in Chemical Abstracts: l-(m-Anisyl)-3-(2-carboxyphneyl)urea; l-(o-Anisyl)-3-(2-carboxyphenyl)urea ; l-(2-Carboxyphenyl)-3-(3,4-dichlorophenyl)urea; and l-(2-Carboxyphenyl)-3-(2,4-dichlorophenyl)urea.
- the compounds of Formula (I), (la), (II) and (III), or a pharmaceutically acceptable salt thereof can be used in the manufacture of a medicament for the prophylactic or therapeutic treatment of any disease state in a human, or other mammal, which is exacerbated or caused by excessive or unregulated IL-8 cytokine production by such mammal's cell, such as but not limited to monocytes and/or macrophages, or other chemokines which bind to the IL-8 ⁇ or ⁇ receptor, also referred to as the type I or type II receptor.
- the compounds of Formula (I), (la), (lb), (Ic), (II) and (DI) all have the same dosages, and dosage formulations as that of Formula (I) are used interchangeably.
- the present invention provides a method of treating a chemokine mediated disease, wherein the chemokine is one which binds to an IL-8 or ⁇ receptor and which method comprises administering an effective amount of a compound of Formula (I) or a pharmaceutically acceptable salt thereof.
- the chemokines ars IL-8, GRO ⁇ , GRO ⁇ , GRO ⁇ or NAP-2.
- the compounds of Formula (I) are administered in an amount sufficient to inhibit cytokine function, in particular IL-8,GRO ⁇ , GRO ⁇ , GRO ⁇ or NAP-2 , such that they are biologically regulated down to normal levels of physiological function, or in some case to subnormal levels, so as to ameliorate the disease state.
- Abnormal levels of IL-8, GRO ⁇ , GRO ⁇ , GRO ⁇ or NAP-2 for instance in the context of the present invention, constitute: (i) levels of free IL-8 greater than or equal to 1 picogram per mL; (ii) any cell associated IL-8, GRO ⁇ , GRO ⁇ , GRO ⁇ or NAP-2 above normal physiological levels; or (iii)the presence of IL-8, GRO ⁇ , GRO ⁇ , GRO ⁇ or NAP-2 above basal levels in cells or tissues in which IL-8, GRO ⁇ , GRO ⁇ , GRO ⁇ or NAP-2 respectively, is produced.
- Chemokine mediated diseases include psoriasis, atopic dermatitis, arthritis, asthma, chronic obstructive pulmonary disease, adult respiratory distress syndrome, inflammatory bowel disease, Crohn's disease, ulcerative colitis, stroke, septic shock, endotoxic shock, gram negative sepsis, toxic shock syndrome, cardiac and renal reperfusion injury, glomerulonephritis, thrombosis, graft vs. host reaction, alzheimers disease, allograft rejections, malaria, restinosis, angiogenesis or undesired hematopoietic stem cells release.
- IL-8, GRO ⁇ , GRO ⁇ , GRO ⁇ or NAP-2 has the unique property of promoting neutrophil chemotaxis, enzyme release including but not limited to elastase release as well as superoxide production and activation.
- the ⁇ -chemokines but particularly, GRO ⁇ , GRO ⁇ , GRO ⁇ or NAP-2, working through the IL-8 type I or II receptor can promote the neovascularization of tumors by promoting the directional growth of endothelial cells. Therefore, the inhibition of IL-8 induced chemotaxis or activation would lead to a direct reduction in the neutrophil infiltration.
- the compounds of Formula (I) are administered in an amount sufficient to inhibit IL-8, binding to the IL-8 alpha or beta receptors, from binding to these receptors, such as evidenced by a reduction in neutrophil chemotaxis and activation.
- the compounds of Formula (I) are inhibitors of IL-8 binding is based upon the effects of the compounds of Formulas (I) in the in vitro receptor binding assays which are described herein.
- the compounds of Formula (I) have been shown to be dual inhibitors of both recombinant type I and type II IL-8 receptors.
- the compounds are inhibitors of only one receptor, preferably Type H
- IL-8 mediated disease or disease state refers to any and all disease states in which IL-8, GRO ⁇ , GRO ⁇ , GRO ⁇ or NAP-2 plays a role, either by production of IL-8, GRO ⁇ , GRO ⁇ , GRO ⁇ or NAP-2 themselves, or by IL-8, GRO ⁇ , GRO ⁇ , GRO ⁇ or NAP-2 causing another monokine to be released, such as but not limited to IL-1, IL-6 or TNF.
- a disease state in which, for instance, IL-1 is a major component, and whose production or action, is exacerbated or secreted in response to IL-8, would therefore be considered a disease stated mediated by IL-8.
- Diseases for which the present compounds are useful include psoriasis, atopic dermatitis, arthritis, asthma, chronic obstructive pulmonary disease, adult respiratory distress syndrome, inflammatory bowel disease, Crohn's disease, ulcerative colitis, stroke, septic shock, endotoxic shock, gram negative sepsis, toxic shock syndrome, cardiac and renal reperfusion injury, glomerulonephritis, thrombosis, graft vs. host reaction, alzheimers disease, allograft rejections, malaria, restinosis, angiogenesis, atherosclerosis, osteoporosis, gingivitis or undesired hematopoietic stem cells release.
- the present compounds are also useful for the treatment of diseases caused by respiratory viruses, including but not limited to rhinovirus and influenza virus, herpesviruses, including but not limited to herpes simplex I and ⁇ , and hepatitis viruses, including but not limited to Hepatitis B and Hepatitis C virus.
- respiratory viruses including but not limited to rhinovirus and influenza virus
- herpesviruses including but not limited to herpes simplex I and ⁇
- hepatitis viruses including but not limited to Hepatitis B and Hepatitis C virus.
- chemokine mediated disease or disease state refers to any and all disease states in which a chemokine which binds to an IL-8 ⁇ or ⁇ receptor plays a role, such as but not limited to IL-8, GRO- ⁇ , GRO- ⁇ , GRO- ⁇ , or NAP-2. This would include a disease state in which, IL-8 plays a role, either by production of IL-8 itself, or by IL-8 causing another monokine to be released, such as but not limited to IL-1, IL-6 or TNF.
- cytokine refers to any secreted polypeptide that affects the functions of cells and is a molecule which modulates interactions between cells in the immune, inflammatory or hematopoietic response.
- a cytokine includes, but is not limited to, monokines and lymphokines, regardless of which cells produce them.
- a monokine is generally referred to as being produced and secreted by a mononuclear cell, such as a macrophage and/or monocyte.
- Lymphokines are generally referred to as being produced by lymphocyte cells.
- cytokines include, but are not limited to, Interleukin-1 (IL-1), Interleukin-6 (IL-6), Interleukin-8 (IL-8), Tumor Necrosis Factor-alpha (TNF- ⁇ ) and Tumor Necrosis Factor beta (TNF- ⁇ ).
- chemokine refers to any secreted polypeptide that affects the functions of cells and is a molecule which modulates interactions between cells in the immune, inflammatory or hematopoietic response, similar to the term
- cytokine above.
- a chemokine is primarily secreted through cell transmembranes and causes chemotaxis and activation of specific white blood cells and leukocytes, neutrophils, monocytes, macrophages, T-cells, B-cells, endothelial cells and smooth muscle cells.
- chemokines include, but are not limited to, IL-8, GRO- ⁇ , GRO- ⁇ , GRO- ⁇ , NAP-2, IP-10, MlP-l ⁇ , MlP- ⁇ , PF4, and MCP 1, 2, and 3.
- a compound of Formula (I) or a pharmaceutically acceptable salt thereof in therapy it will normally be formulated into a pharmaceutical composition in accordance with standard pharmaceutical practice.
- This invention also relates to a pharmaceutical composition comprising an effective, non-toxic amount of a compound of Formula (I) and a pharmaceutically acceptable carrier or diluent.
- Compounds of Formula (I), pharmaceutically acceptable salts thereof and pharmaceutical compositions incorporating such may conveniently be administered by any of the routes conventionally used for drug administration, for instance, orally, topically, parenterally or by inhalation.
- the compounds of Formula (I) may be administered in conventional dosage forms prepared by combining a compound of Formula (I) with standard pharmaceutical carriers according to conventional procedures.
- the compounds of Formula (I) may also be administered in conventional dosages in combination with a known, second therapeutically active compound. These procedures may involve mixing, granulating and compressing or dissolving the ingredients as appropriate to the desired preparation.
- the form and character of the pharmaceutically acceptable character or diluent is dictated by the amount of active ingredient with which it is to be combined, the route of administration and other well-known variables.
- the carrier(s) must be "acceptable” in the sense of being compatible with the other ingredients of the formulation and not deleterious to the recipient thereof.
- the pharmaceutical carrier employed may be, for example, either a solid or liquid.
- solid carriers are lactose, terra alba, sucrose, talc, gelatin, agar, pectin, acacia, magnesium stearate, stearic acid and the like.
- liquid carriers are syrup, peanut oil, olive oil, water and the like.
- the carrier or diluent may include time delay material well known to the art, such as glyceryl mono- stearate or glyceryl distearate alone or with a wax.
- the preparation can be tableted, placed in a hard gelatin capsule in powder or pellet form or in the form of a troche or lozenge.
- the amount of solid carrier will vary widely but preferably will be from about 25mg. to about lg.
- the preparation will be in the form of a syrup, emulsion, soft gelatin capsule, sterile injectable liquid such as an ampule or nonaqueous liquid suspension.
- Compounds of Formula (I) may be administered topically, that is by non- systemic administration.
- systemic administration refers to oral, intravenous, intraperitoneal and intramuscular administration.
- Formulations suitable for topical administration include liquid or semi-liquid preparations suitable for penetration through the skin to the site of inflammation such as liniments, lotions, creams, ointments or pastes, and drops suitable for administration to the eye, ear or nose.
- the active ingredient may comprise, for topical administration, from 0.001% to 10% w/w, for instance from 1% to 2% by weight of the Formulation. It may however comprise as much as 10% w/w but preferably will comprise less than 5% w/w, more preferably from 0.1% to 1% w/w of the Formulation.
- Lotions according to the present invention include those suitable for application to the skin or eye.
- An eye lotion may comprise a sterile aqueous solution optionally containing a bactericide and may be prepared by methods similar to those for the preparation of drops.
- Lotions or liniments for application to the skin may also include an agent to hasten drying and to cool the skin, such as an alcohol or acetone, and/or a moisturizer such as glycerol or an oil such as castor oil or arachis oil.
- Creams, ointments or pastes according to the present invention are semi-solid formulations of the active ingredient for external application. They may be made by mixing the active ingredient in finely-divided or powdered form, alone or in solution or suspension in an aqueous or non-aqueous fluid, with the aid of suitable machinery, with a greasy or non-greasy base.
- the base may comprise hydrocarbons such as hard, soft or liquid paraffin, glycerol, beeswax, a metallic soap; a mucilage; an oil of natural origin such as almond, corn, arachis, castor or olive oil; wool fat or its derivatives or a fatty acid such as steric or oleic acid together with an alcohol such as propylene glycol or a macrogel.
- the formulation may incorporate any suitable surface active agent such as an anionic, cationic or non-ionic surfactant such as a sorbitan ester or a polyoxyethylene derivative thereof.
- Suspending agents such as natural gums, cellulose derivatives or inorganic materials such as silicaceous silicas, and other ingredients such as lanolin, may also be included.
- Drops according to the present invention may comprise sterile aqueous or oily solutions or suspensions and may be prepared by dissolving the active ingredient in a suitable aqueous solution of a bactericidal and/or fungicidal agent and/or any other suitable preservative, and preferably including a surface active agent.
- the resulting solution may then be clarified by filtration, transferred to a suitable container which is then sealed and sterilized by autoclaving or maintaining at 98-100 °C. for half an hour.
- the solution may be sterilized by filtration and transferred to the container by an aseptic technique.
- bactericidal and fungicidal agents suitable for inclusion in the drops are phenylmercuric nitrate or acetate (0.002%), benzalkonium chloride (0.01%) and chlorhexidine acetate (0.01%).
- Suitable solvents for the preparation of an oily solution include glycerol, diluted alcohol and propylene glycol.
- Compounds of formula (I) may be administered parenteraly, that is by intravenous, intramuscular, subcutaneous intranasal, intrarectal, intravaginal or intraperitoneal administration. The subcutaneous and intramuscular forms of parenteral administration are generally preferred. Appropriate dosage forms for such administration may be prepared by conventional techniques.
- Compounds of Formula (I) may also be administered by inhalation, that is by intranasal and oral inhalation administration.
- Appropriate dosage forms for such administration such as an aerosol formulation or a metered dose inhaler, may be prepared by conventional techniques.
- the daily oral dosage regimen will preferably be from about 0.01 to about 80 mg/kg of total body weight.
- the daily parenteral dosage regimen about 0.001 to about 80 mg/kg of total body weight.
- the daily topical dosage regimen will preferably be from 0.1 mg to 150 mg, administered one to four, preferably two or three times daily.
- the daily inhalation dosage regimen will preferably be from about 0.01 mg/kg to about 1 mg/kg per day.
- the optimal quantity and spacing of individual dosages of a compound of Formula (I) or a pharmaceutically acceptable salt thereof will be determined by the nature and extent of the condition being treated, the form, route and site of administration, and the particular patient being treated, and that such optimums can be determined by conventional techniques. It will also be appreciated by one of skill in the art that the optimal course of treatment, i.e., the number of doses of a compound of Formula (I) or a pharmaceutically acceptable salt thereof given per day for a defined number of days, can be ascertained by those skilled in the art using conventional course of treatment determination tests.
- IL-8, and Gro- ⁇ chemokine inhibitory effects of compounds of the present invention were determined by the following in vitro assay: Receptor Binding Assays: [125JJ r ⁇ _8 (human recombinant) was obtained from Amersham Corp., Arlington Heights, IL, with specific activity 2000 Ci/mmol. Gro- ⁇ was obtained from NEN- New England Nuclear. All other chemicals were of analytical grade. High levels of recombinant human IL-8 type ⁇ and ⁇ receptors were individually expressed in Chinese hamster ovary cells as described previously (Holmes, et al, Science, 1991, 253, 1278).
- the Chinese hamster ovary membranes were homogenized according to a previously described protocol (Haour, et al., JBiol Chem., 249 pp 2195-2205 (1974)). Except that the homogenization buffer was changed to lOmM Tris-HCL, lmM MgS04, 0.5mM EDTA (ethylene-diaminetetra-acetic acid), lmMPMSF ( ⁇ -toluenesulphonyl fluoride), 0.5 mg/L Leupeptin, pH 7.5. Membrane protein concentration was determined using Pierce Co. micro-assay kit using bovine serum albumin as a standard. All assays were performed in a 96-well micro plate format.
- Each reaction mixture contained 125 I IL-8 (0.25 nM) or 125 I Gro- ⁇ and 0.5 ⁇ g/mL of IL-8R ⁇ or 1.0 ⁇ g/mL of IL-8R ⁇ membranes in 20 mM Bis-Trispropane and 0.4 mM Tris HC1 buffers, pH 8.0, containing 1.2 mM MgSO-i, 0.1 mM EDTA, 25 mM NaCI and 0.03% CHAPS.
- drug or compound of interest was added which had been pre-dissolved in DMSO so as to reach a final concentration of between O.OlnM and 100 uM.
- the assay was initiated by addition of 125j_]L_g After 1 hour at room temperature the plate was harvested using a Tomtec 96-well harvester onto a glass fiber filtermat blocked with 1% polyethylenimine/0.5% BSA and washed 3 times with 25 mM NaCI, 10 mM
- the filter was then dried and counted on the Betaplate liquid scintillation counter.
- the recombinant IL-8 R ⁇ , or Type I, receptor is also referred to herein as the non-permissive receptor and the recombinant IL-8 R ⁇ , or Type ⁇ , receptor is referred to as the permissive receptor. All of the exemplified compounds of Formulas (I) to (HI) noted herein in the
- N-(2-Hydroxy-4-nitrophenyl)-N'-(2-methoxy-4-nitrophenyl)urea N-(2-Hydroxy- 1 -napthyl)-N '-(2-phenylphenyl)urea N-(2-Hydroxy-5-ethylsulfonylphenyl)-N'-(2-bromophenyl)urea N-(2-hydroxy 3,4 dichlorophenyl )-N'-(4-phenylphenyl)urea N-(2-hydroxy-3-naphthyl)-N'-(2-methoxyphenyl)urea N-(2-hydroxy-3-naphthyl)-N'-(2-phenylphenyl)urea N-(2-Hydroxy-3-naphthyl)-N'-(4-methoxyphenyl)urea N-(2-Hydroxy-3-naphthyl)
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- General Health & Medical Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Pharmacology & Pharmacy (AREA)
- Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Organic Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Virology (AREA)
- Physical Education & Sports Medicine (AREA)
- Communicable Diseases (AREA)
- Oncology (AREA)
- Epidemiology (AREA)
- Rheumatology (AREA)
- Orthopedic Medicine & Surgery (AREA)
- Molecular Biology (AREA)
- Tropical Medicine & Parasitology (AREA)
- Vascular Medicine (AREA)
- Diabetes (AREA)
- Pulmonology (AREA)
- Urology & Nephrology (AREA)
- Biotechnology (AREA)
- Heart & Thoracic Surgery (AREA)
- Hematology (AREA)
- Cardiology (AREA)
- Emergency Medicine (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
- Heterocyclic Compounds That Contain Two Or More Ring Oxygen Atoms (AREA)
- Heterocyclic Compounds Containing Sulfur Atoms (AREA)
- Quinoline Compounds (AREA)
Abstract
Description
Claims
Priority Applications (11)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| IL14576100A IL145761A0 (en) | 1999-06-16 | 2000-06-15 | Il-8 receptor antagonists |
| EP00942843A EP1185261A4 (en) | 1999-06-16 | 2000-06-15 | Il-8 receptor antagonists |
| BR0010802-2A BR0010802A (en) | 1999-06-16 | 2000-06-15 | Il-8 receptor antagonists |
| JP2001502828A JP2003501459A (en) | 1999-06-16 | 2000-06-15 | IL-8 receptor antagonist |
| AU57413/00A AU766083B2 (en) | 1999-06-16 | 2000-06-15 | IL-8 receptor antagonists |
| MXPA01013004A MXPA01013004A (en) | 1999-06-16 | 2000-06-15 | Il-8 receptor antagonists. |
| CA002377341A CA2377341A1 (en) | 1999-06-16 | 2000-06-15 | Il-8 receptor antagonists |
| NZ514695A NZ514695A (en) | 1999-06-16 | 2000-06-15 | IL-8 receptor antagonists |
| KR1020017016142A KR20020010709A (en) | 1999-06-16 | 2000-06-15 | IL-8 Receptor Antagonists |
| NO20016053A NO20016053D0 (en) | 1999-06-16 | 2001-12-11 | IL-8 receptor antagonists |
| HK02106188.7A HK1044483A1 (en) | 1999-06-16 | 2002-08-22 | Il-8 receptor antagonists |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US13967599P | 1999-06-16 | 1999-06-16 | |
| US60/139,675 | 1999-06-16 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2000076495A1 true WO2000076495A1 (en) | 2000-12-21 |
Family
ID=22487790
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2000/016499 WO2000076495A1 (en) | 1999-06-16 | 2000-06-15 | Il-8 receptor antagonists |
Country Status (20)
| Country | Link |
|---|---|
| EP (1) | EP1185261A4 (en) |
| JP (1) | JP2003501459A (en) |
| KR (1) | KR20020010709A (en) |
| CN (1) | CN1355697A (en) |
| AR (1) | AR030391A1 (en) |
| AU (1) | AU766083B2 (en) |
| BR (1) | BR0010802A (en) |
| CA (1) | CA2377341A1 (en) |
| CO (1) | CO5200760A1 (en) |
| CZ (1) | CZ20014471A3 (en) |
| HK (1) | HK1044483A1 (en) |
| HU (1) | HUP0201571A3 (en) |
| IL (1) | IL145761A0 (en) |
| MX (1) | MXPA01013004A (en) |
| NO (1) | NO20016053D0 (en) |
| NZ (1) | NZ514695A (en) |
| PL (1) | PL352232A1 (en) |
| TR (1) | TR200103680T2 (en) |
| WO (1) | WO2000076495A1 (en) |
| ZA (1) | ZA200109479B (en) |
Cited By (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2004012733A2 (en) * | 2002-08-01 | 2004-02-12 | Neurosearch A/S | Compounds useful for the treatment of diseases responsive to antiangiogenetic therapy |
| WO2006083271A2 (en) * | 2004-05-18 | 2006-08-10 | Achillion Pharmaceuticals, Inc. | Substituted aryl acylthioureas and related compounds; inhibitors of viral replication |
| WO2006089871A2 (en) * | 2005-02-23 | 2006-08-31 | Neurosearch A/S | Diphenylurea derivatives useful as erg channel openers for the treatment of cardiac arrhythmias |
| WO2007002635A2 (en) | 2005-06-27 | 2007-01-04 | Bristol-Myers Squibb Company | C-linked cyclic antagonists of p2y1 receptor useful in the treatment of thrombotic conditions |
| US7521479B2 (en) * | 2001-04-16 | 2009-04-21 | Panacea Pharmaceuticals, Inc. | Methods of treating prion disease in mammals |
| US7550499B2 (en) | 2004-05-12 | 2009-06-23 | Bristol-Myers Squibb Company | Urea antagonists of P2Y1 receptor useful in the treatment of thrombotic conditions |
| US7582673B2 (en) | 2004-10-21 | 2009-09-01 | High Point Pharmaceuticals, Llc | Bissulfonamide compounds as agonists of GalR1, compositions, and methods of use |
| US7645778B2 (en) | 2005-01-19 | 2010-01-12 | Bristol-Myers Squibb Company | Heteroaryl compounds as P2Y1 receptor inhibitors |
| US7714002B2 (en) | 2005-06-27 | 2010-05-11 | Bristol-Myers Squibb Company | Carbocycle and heterocycle antagonists of P2Y1 receptor useful in the treatment of thrombotic conditions |
| US7728008B2 (en) | 2005-06-27 | 2010-06-01 | Bristol-Myers Squibb Company | N-linked heterocyclic antagonists of P2Y1 receptor useful in the treatment of thrombotic conditions |
| US7816382B2 (en) | 2005-06-27 | 2010-10-19 | Bristol-Myers Squibb Company | Linear urea mimics antagonists of P2Y1 receptor useful in the treatment of thrombotic condition |
| KR101030416B1 (en) * | 2002-01-28 | 2011-04-20 | 힉스-인스티튜티 오이 | Treatment of rheumatoid arthritis |
| US7960569B2 (en) | 2006-10-17 | 2011-06-14 | Bristol-Myers Squibb Company | Indole antagonists of P2Y1 receptor useful in the treatment of thrombotic conditions |
| US10093617B1 (en) | 2015-10-30 | 2018-10-09 | Sumitomo Chemical Company, Limited | Method for producing 2-amino-4-substituted phenol |
Families Citing this family (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| AU2005245399A1 (en) * | 2004-05-12 | 2005-12-01 | Schering Corporation | CXCR1 and CXCR2 chemokine antagonists |
| JP2008184403A (en) * | 2007-01-29 | 2008-08-14 | Japan Health Science Foundation | New hepatitis c virus inhibitor |
| KR100982661B1 (en) * | 2008-04-22 | 2010-09-17 | 전남대학교산학협력단 | Pharmaceutical composition for the prevention and treatment of malaria containing a compound that inhibits plasmincin II activity as an active ingredient and a method for treating malaria using the same |
| KR100970940B1 (en) * | 2010-05-03 | 2010-07-20 | 전남대학교산학협력단 | Pharmaceutical composition for preventing and treating malaria comprising compounds that inhibit Plasmepsin II activity and the method of treating malaria using thereof |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5900430A (en) * | 1991-02-19 | 1999-05-04 | Anormed, Inc. | Cytokine inhibitors |
Family Cites Families (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH11503110A (en) * | 1995-02-17 | 1999-03-23 | スミスクライン・ビーチャム・コーポレイション | IL-8 receptor antagonist |
| WO2000016768A1 (en) * | 1998-09-18 | 2000-03-30 | Smithkline Beecham Corporation | Cxcr2 inhibitors and pmn adhesion and t-cell chemotaxis |
-
2000
- 2000-06-13 CO CO00043844A patent/CO5200760A1/en not_active Application Discontinuation
- 2000-06-14 AR ARP000102923A patent/AR030391A1/en not_active Application Discontinuation
- 2000-06-15 IL IL14576100A patent/IL145761A0/en unknown
- 2000-06-15 PL PL00352232A patent/PL352232A1/en not_active Application Discontinuation
- 2000-06-15 NZ NZ514695A patent/NZ514695A/en unknown
- 2000-06-15 CA CA002377341A patent/CA2377341A1/en not_active Abandoned
- 2000-06-15 JP JP2001502828A patent/JP2003501459A/en not_active Withdrawn
- 2000-06-15 HU HU0201571A patent/HUP0201571A3/en unknown
- 2000-06-15 WO PCT/US2000/016499 patent/WO2000076495A1/en not_active Application Discontinuation
- 2000-06-15 CN CN00809045A patent/CN1355697A/en active Pending
- 2000-06-15 AU AU57413/00A patent/AU766083B2/en not_active Ceased
- 2000-06-15 TR TR2001/03680T patent/TR200103680T2/en unknown
- 2000-06-15 EP EP00942843A patent/EP1185261A4/en not_active Withdrawn
- 2000-06-15 CZ CZ20014471A patent/CZ20014471A3/en unknown
- 2000-06-15 KR KR1020017016142A patent/KR20020010709A/en not_active Application Discontinuation
- 2000-06-15 BR BR0010802-2A patent/BR0010802A/en not_active IP Right Cessation
- 2000-06-15 MX MXPA01013004A patent/MXPA01013004A/en unknown
-
2001
- 2001-11-16 ZA ZA200109479A patent/ZA200109479B/en unknown
- 2001-12-11 NO NO20016053A patent/NO20016053D0/en not_active Application Discontinuation
-
2002
- 2002-08-22 HK HK02106188.7A patent/HK1044483A1/en unknown
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5900430A (en) * | 1991-02-19 | 1999-05-04 | Anormed, Inc. | Cytokine inhibitors |
Cited By (21)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7521479B2 (en) * | 2001-04-16 | 2009-04-21 | Panacea Pharmaceuticals, Inc. | Methods of treating prion disease in mammals |
| KR101030416B1 (en) * | 2002-01-28 | 2011-04-20 | 힉스-인스티튜티 오이 | Treatment of rheumatoid arthritis |
| WO2004012733A3 (en) * | 2002-08-01 | 2004-03-18 | Neurosearch As | Compounds useful for the treatment of diseases responsive to antiangiogenetic therapy |
| WO2004012733A2 (en) * | 2002-08-01 | 2004-02-12 | Neurosearch A/S | Compounds useful for the treatment of diseases responsive to antiangiogenetic therapy |
| US7550499B2 (en) | 2004-05-12 | 2009-06-23 | Bristol-Myers Squibb Company | Urea antagonists of P2Y1 receptor useful in the treatment of thrombotic conditions |
| WO2006083271A2 (en) * | 2004-05-18 | 2006-08-10 | Achillion Pharmaceuticals, Inc. | Substituted aryl acylthioureas and related compounds; inhibitors of viral replication |
| WO2006083271A3 (en) * | 2004-05-18 | 2006-10-26 | Achillion Pharmaceuticals Inc | Substituted aryl acylthioureas and related compounds; inhibitors of viral replication |
| EA009882B1 (en) * | 2004-05-18 | 2008-04-28 | Ачиллион Фармасьютикалз, Инк. | Substituted aryl acylthioureas and related compounds; inhibitors of virus replication |
| US7582673B2 (en) | 2004-10-21 | 2009-09-01 | High Point Pharmaceuticals, Llc | Bissulfonamide compounds as agonists of GalR1, compositions, and methods of use |
| US7645778B2 (en) | 2005-01-19 | 2010-01-12 | Bristol-Myers Squibb Company | Heteroaryl compounds as P2Y1 receptor inhibitors |
| WO2006089871A2 (en) * | 2005-02-23 | 2006-08-31 | Neurosearch A/S | Diphenylurea derivatives useful as erg channel openers for the treatment of cardiac arrhythmias |
| WO2006089871A3 (en) * | 2005-02-23 | 2007-04-26 | Neurosearch As | Diphenylurea derivatives useful as erg channel openers for the treatment of cardiac arrhythmias |
| WO2007002635A2 (en) | 2005-06-27 | 2007-01-04 | Bristol-Myers Squibb Company | C-linked cyclic antagonists of p2y1 receptor useful in the treatment of thrombotic conditions |
| US7700620B2 (en) | 2005-06-27 | 2010-04-20 | Bristol-Myers Squibb Company | C-linked cyclic antagonists of P2Y1 receptor useful in the treatment of thrombotic conditions |
| US7714002B2 (en) | 2005-06-27 | 2010-05-11 | Bristol-Myers Squibb Company | Carbocycle and heterocycle antagonists of P2Y1 receptor useful in the treatment of thrombotic conditions |
| US7728008B2 (en) | 2005-06-27 | 2010-06-01 | Bristol-Myers Squibb Company | N-linked heterocyclic antagonists of P2Y1 receptor useful in the treatment of thrombotic conditions |
| US7816382B2 (en) | 2005-06-27 | 2010-10-19 | Bristol-Myers Squibb Company | Linear urea mimics antagonists of P2Y1 receptor useful in the treatment of thrombotic condition |
| WO2007002635A3 (en) * | 2005-06-27 | 2007-03-29 | Bristol Myers Squibb Co | C-linked cyclic antagonists of p2y1 receptor useful in the treatment of thrombotic conditions |
| US8329718B2 (en) | 2005-06-27 | 2012-12-11 | Bristol-Myers Squibb Company | N-linked heterocyclic antagonists of P2Y1 receptor useful in the treatment of thrombotic conditions |
| US7960569B2 (en) | 2006-10-17 | 2011-06-14 | Bristol-Myers Squibb Company | Indole antagonists of P2Y1 receptor useful in the treatment of thrombotic conditions |
| US10093617B1 (en) | 2015-10-30 | 2018-10-09 | Sumitomo Chemical Company, Limited | Method for producing 2-amino-4-substituted phenol |
Also Published As
| Publication number | Publication date |
|---|---|
| CO5200760A1 (en) | 2002-09-27 |
| PL352232A1 (en) | 2003-08-11 |
| CZ20014471A3 (en) | 2002-08-14 |
| BR0010802A (en) | 2002-02-19 |
| TR200103680T2 (en) | 2002-07-22 |
| KR20020010709A (en) | 2002-02-04 |
| JP2003501459A (en) | 2003-01-14 |
| AU5741300A (en) | 2001-01-02 |
| HK1044483A1 (en) | 2002-10-25 |
| CN1355697A (en) | 2002-06-26 |
| HUP0201571A2 (en) | 2002-08-28 |
| IL145761A0 (en) | 2002-07-25 |
| CA2377341A1 (en) | 2000-12-21 |
| MXPA01013004A (en) | 2002-07-30 |
| NZ514695A (en) | 2004-05-28 |
| NO20016053L (en) | 2001-12-11 |
| NO20016053D0 (en) | 2001-12-11 |
| EP1185261A1 (en) | 2002-03-13 |
| HUP0201571A3 (en) | 2002-11-28 |
| EP1185261A4 (en) | 2004-02-25 |
| AR030391A1 (en) | 2003-08-20 |
| AU766083B2 (en) | 2003-10-09 |
| ZA200109479B (en) | 2002-11-18 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| AU725456B2 (en) | IL-8 receptor antagonists | |
| US6262113B1 (en) | IL-8 receptor antagonists | |
| US6005008A (en) | IL-8 receptor antagonists | |
| US5780483A (en) | IL-8 receptor antagonists | |
| US6211373B1 (en) | Phenyl urea antagonists of the IL-8 receptor | |
| US6271261B1 (en) | IL-8 receptor antagonists | |
| AU766083B2 (en) | IL-8 receptor antagonists | |
| EP0907362A1 (en) | Il-8 receptor antagonists | |
| WO1998034929A9 (en) | Il-8 receptor antagonists | |
| EP0971908A1 (en) | Il-8 receptor antagonists | |
| EP0939634A1 (en) | Il-8 receptor antagonists | |
| WO1998005317A1 (en) | Il-8 receptor antagonists | |
| EP0915653A1 (en) | Il-8 receptor antagonists | |
| WO2000076457A2 (en) | Il-8 receptor antagonists | |
| IL141121A (en) | Process for preparing cyano-phenol derivatives | |
| CA2432662A1 (en) | Il-8 receptor antagonists | |
| WO2000076508A1 (en) | Il-8 receptor antagonists | |
| CA2377397A1 (en) | Il-8 receptor antagonists |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 00809045.9 Country of ref document: CN |
|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): AE AL AU BA BB BG BR CA CN CZ DZ EE GE GH GM HR HU ID IL IN IS JP KP KR LC LK LR LT LV MA MG MK MN MX NO NZ PL RO SG SI SK SL TR TT TZ UA US UZ VN YU ZA |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): GH GM KE LS MW MZ SD SL SZ TZ UG ZW AM AZ BY KG KZ MD RU TJ TM AT BE CH CY DE DK ES FI FR GB GR IE IT LU MC NL PT SE BF BJ CF CG CI CM GA GN GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 514695 Country of ref document: NZ Ref document number: IN/PCT/2001/01225/MU Country of ref document: IN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 57413/00 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2000942843 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2001/09479 Country of ref document: ZA Ref document number: 200109479 Country of ref document: ZA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 09980305 Country of ref document: US |
|
| WWE | Wipo information: entry into national phase |
Ref document number: PV2001-4471 Country of ref document: CZ |
|
| WWE | Wipo information: entry into national phase |
Ref document number: PA/a/2001/013004 Country of ref document: MX Ref document number: 2377341 Country of ref document: CA Ref document number: 2001/03680 Country of ref document: TR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1020017016142 Country of ref document: KR |
|
| ENP | Entry into the national phase |
Ref document number: 2001 502828 Country of ref document: JP Kind code of ref document: A |
|
| WWP | Wipo information: published in national office |
Ref document number: 1020017016142 Country of ref document: KR |
|
| WWP | Wipo information: published in national office |
Ref document number: 2000942843 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: PV2001-4471 Country of ref document: CZ |
|
| WWG | Wipo information: grant in national office |
Ref document number: 57413/00 Country of ref document: AU |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: 2000942843 Country of ref document: EP |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: 1020017016142 Country of ref document: KR |