WO1997043267A1 - Pyrazines - Google Patents
Pyrazines Download PDFInfo
- Publication number
- WO1997043267A1 WO1997043267A1 PCT/GB1997/001227 GB9701227W WO9743267A1 WO 1997043267 A1 WO1997043267 A1 WO 1997043267A1 GB 9701227 W GB9701227 W GB 9701227W WO 9743267 A1 WO9743267 A1 WO 9743267A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- formula
- group
- pyrazine
- compound
- catalyst
- Prior art date
Links
- 0 **C(*C=C(*)*1)=C1C(*=*)=O Chemical compound **C(*C=C(*)*1)=C1C(*=*)=O 0.000 description 3
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D471/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00
- C07D471/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00 in which the condensed system contains two hetero rings
- C07D471/04—Ortho-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D241/00—Heterocyclic compounds containing 1,4-diazine or hydrogenated 1,4-diazine rings
- C07D241/02—Heterocyclic compounds containing 1,4-diazine or hydrogenated 1,4-diazine rings not condensed with other rings
- C07D241/10—Heterocyclic compounds containing 1,4-diazine or hydrogenated 1,4-diazine rings not condensed with other rings having three double bonds between ring members or between ring members and non-ring members
- C07D241/14—Heterocyclic compounds containing 1,4-diazine or hydrogenated 1,4-diazine rings not condensed with other rings having three double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D241/20—Nitrogen atoms
Definitions
- the present invention relates to novel pyrazines and to methods of their use in preparing further imidazopyrazine compounds.
- the luminescent chromophores such as Coelenterazine which can be isolated from Aequora Victoria, as described in the Article by Shimomura O, Johnson Fitt. and Morisett in Biochemistry 1974, 13,3278-3286, contain the imidazopyrazine ring system.
- Coelenterazine has been extensively studied because of the bioluminescent process which is triggered by calcium ions and has proved to be a sensitive method for the detection and quantification of calcium ions.
- Coelenterazine and analogues have been prepared by the "Kishi route", as described in the Article by Shimomura O, Musieki B. Kishi Y, Biochem J., 1989, 261,913-920.
- the aromatic groups Ar which can be used include groups such as phenyl, naphthyl and thiophenyl.
- the substituted aromatic groups include alkyl, aryl and substituted aryl, alkaryl, halides, halide substituted aryl groups and groups such as trifiuoromethyl, fluoro and alkoxy groups.
- the Ph group can be substituted by any of the substituents listed above.
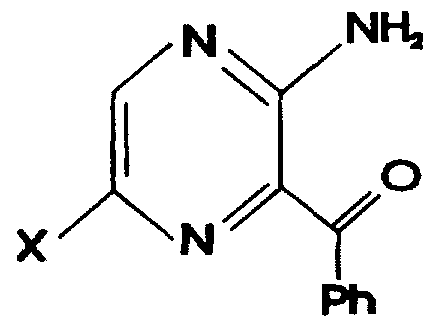
- the invention also provides pyrazines of the formula
- Ph is an aromatic or substituted aromatic group e.g.as Ar in (I).
- the invention further provides pyrazines of formula
- the compounds (I) of the present invention can be prepared by the reaction of the 5-halo substituted pyrazine:-
- the catalysts which can be used in the process of the present invention include, but are not limited to, platinum groups catalysts, particularly palladium compounds.
- the preferred palladium catalysts are Pd(II) complexes, particularly organic palladium compounds such as diphenyl phosphino complexes of Pd(II) e.g. [l ,4-bis(diphenylphosphino)butane]palladium(II) chloride and [1,1 bis(diphenylphosphino)ferrocene]palladium(II) acetate.
- reaction of compound B with the arylboronic acid preferably takes place in a solvent which will depend on the nature of the catalyst used.
- solvents which can be used are toluene, tetrahydrofuran, dimethyl formamide, optionally in the presence of co-solvents such as water or an amine such as triethylamine.
- the reaction can be illustrated as follows:
- Preferred Ar groups are 4-MeOC 6 H 4 , C 6 H 5 , naphthyl, 4-FC 6 H 4 , 4-CF 3 C 6 H 4 and 2-thienyl.
- Compound B can be prepared by bromination of the corresponding pyrazine:-
- the pyrazine C can be prepared by known methods, for example, as described in the Article by Turek A, Mojovie L, Queguiner G, Synthesis 1988, 881-884.
- 2-chloropyrazine was metallated using lithium tetramethylpiperidide and reacted with benzaldehyde to afford the alcohol (3) in 88% yield.
- Oxidation of (3) using freshly-prepared MnO 2 gave the ketone (4) in 93% yield.
- Introduction of the amino group was achieved by heating ketone (4) with a solution of ammonia in ethanol at 120°C overnight leading to aminopyrazine (5) in 63% yield.
- the preferred method for cleaving the methyl aryl ether is by reaction with ethanethiolate.
- the phenol (II) obtained can be converted to the phenol (III) by reduction.
- Any reduction which reduces the ketone group can be used such as using a metallic catalyst such as platinum or palladium or carbon, treatment with zinc and hydrochloric acid under usual Clemenson conditions (M Lucas and F Solano, Anal. Biochem, 1992 206,273).
- Reduction of the ketone to alcohol e.g. using NaBH 4 in methanol followed by treatment of the alcohol with Et 3 SiH and trifluoracetic acid.
- a preferred method is a Wolff-Kishner reduction by reaction with hydrazine hydrate and potassium hydroxide in ethylene glycol.
- the above compounds of the present invention can be used as intermediates to prepare compounds containing the imidazopyrazine ring system such as coelenterazine by reduction of the benzoyl substituent to benzyl, condensation with 4-methoxybenzylglyoxal and cleavage of the methyl ethers using pyridinium bromide or by reaction with a ketoaldehyde.
- compounds containing the imidazopyrazine ring system such as coelenterazine by reduction of the benzoyl substituent to benzyl, condensation with 4-methoxybenzylglyoxal and cleavage of the methyl ethers using pyridinium bromide or by reaction with a ketoaldehyde.
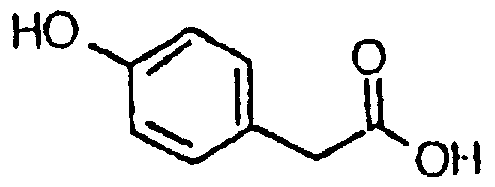
- the ⁇ -ketoaldehyde can be prepared by known methods, for example by the route described by S Inove, S Sugiura, H Kakoi and K Hasizume. Chem. Letters 1975, 141. In this route the phenol group in 4-hydroxy phenyl acetic acid is acylated, converted to the acid chloride (with SOCU) and reacted with diazomethane to give the ⁇ -diazoketone.
- the ⁇ -diazoketone was reacted with dry HBr to give the bromomethyl ketone which is reacted with silver nitrate in acetonitrile to give the nitrate ester which is reacted with sodium acetate to give the ⁇ -ketoaldehyde.
- the compound (V) is found to exist mainly in the enol form in solution in many solutions.
- Example 1 The invention is further described in the following Examples in which Examples 1 to 7 describe the preparation of the novel pyrazines of the invention and Example 8 illustrates the production of coelenterazine with the preparation of the 2-amino-3-benzoyl-5-(4'-methoxyphenyl)pyrazine used in Example 8 prepared as in Example 1.
- Example 1
- Phenyl and naphthylboronic acids reacted cleanly with (6) and gave the heterobiaryl products 7b and 7c in very high yields (entries 3 and 4).
- the introduction of electron-withdrawing substituents on the boronic acid moiety presented no problems and both the 4-fluoro and the 4-trifluoromethyl-substituted boronic acids reacted within a reasonable timescale to give the respective coupled products 7d and 7e in high yields.
- Diazald (7.4 g, 0.02 mol) in dry diethyl ether (82 ml) was added dropwise to a warm solution of aqueous potassium hydroxide (7.4 g, in 12 ml of H 2 O) and di(ethyleneglycol)ethyl ether (17 ml). Diazomethane was collected as a co-distillate with diethyl ether.
- Ethanethiol (0.6 ml. 8 mmol), dissolved in dry DMF (3 ml) was added to a suspension of sodium hydride (5.5 equiv) in dry DMF (2 ml) under an atmosphere or argon. The mixture was stirred for 5 mins before a solution of 2-amino-3 -benzoyl -5-(p-methoxyhenyl)pyrazine (520 mg, 1.7 mmol) in DMF (4 ml) was added. The solution was heated at 100°C for 8 hours. On cooling, the mixture was acidified with 10% aqueous HC1 and washed with EtOAc. The aqueous phase was made alkaline (10% aqueous NaOH) and extracted with EtOAc.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Plural Heterocyclic Compounds (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
Abstract
Description
Claims
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP97920835A EP0914318A1 (en) | 1996-05-11 | 1997-05-06 | Pyrazines |
| CA002253910A CA2253910A1 (en) | 1996-05-11 | 1997-05-06 | Pyrazines |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| GB9609972.6 | 1996-05-11 | ||
| GBGB9609972.6A GB9609972D0 (en) | 1996-05-11 | 1996-05-11 | Pyrazines |
| GB9624653.3 | 1996-11-27 | ||
| GBGB9624653.3A GB9624653D0 (en) | 1996-11-27 | 1996-11-27 | Synthesis of coelenterazine |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO1997043267A1 true WO1997043267A1 (en) | 1997-11-20 |
Family
ID=26309321
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/GB1997/001227 WO1997043267A1 (en) | 1996-05-11 | 1997-05-06 | Pyrazines |
Country Status (3)
| Country | Link |
|---|---|
| EP (1) | EP0914318A1 (en) |
| CA (1) | CA2253910A1 (en) |
| WO (1) | WO1997043267A1 (en) |
Cited By (30)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2003093297A3 (en) * | 2002-05-03 | 2004-07-01 | Exelixis Inc | Protein kinase modulators and methods of use |
| US7115621B2 (en) | 2001-04-25 | 2006-10-03 | Lilly Icos Llc | Chemical compounds |
| WO2009115517A3 (en) * | 2008-03-19 | 2010-01-07 | Novartis Ag | Pyridines and pyrazines as inhibitors of pi3k |
| US7704995B2 (en) | 2002-05-03 | 2010-04-27 | Exelixis, Inc. | Protein kinase modulators and methods of use |
| US20120035408A1 (en) * | 2010-05-12 | 2012-02-09 | Vertex Pharmaceuticals Incorporated | Compounds useful as inhibitors of atr kinase |
| CN102432614A (en) * | 2011-11-16 | 2012-05-02 | 泰州凯美迪生物医药技术有限公司 | Method for synthesizing coelenterazine |
| US8263585B2 (en) | 2007-05-04 | 2012-09-11 | Novartis Ag | Organic compounds |
| US8841308B2 (en) | 2008-12-19 | 2014-09-23 | Vertex Pharmaceuticals Incorporated | Pyrazin-2-amines useful as inhibitors of ATR kinase |
| US8846686B2 (en) | 2011-09-30 | 2014-09-30 | Vertex Pharmaceuticals Incorporated | Compounds useful as inhibitors of ATR kinase |
| US8846918B2 (en) | 2011-11-09 | 2014-09-30 | Vertex Pharmaceuticals Incorporated | Compounds useful as inhibitors of ATR kinase |
| US8853217B2 (en) | 2011-09-30 | 2014-10-07 | Vertex Pharmaceuticals Incorporated | Compounds useful as inhibitors of ATR kinase |
| US8912198B2 (en) | 2012-10-16 | 2014-12-16 | Vertex Pharmaceuticals Incorporated | Compounds useful as inhibitors of ATR kinase |
| US9035053B2 (en) | 2011-09-30 | 2015-05-19 | Vertex Pharmaceuticals Incorporated | Processes for making compounds useful as inhibitors of ATR kinase |
| US9096602B2 (en) | 2011-06-22 | 2015-08-04 | Vertex Pharmaceuticals Incorporated | Substituted pyrrolo[2,3-B]pyrazines as ATR kinase inhibitors |
| US9309250B2 (en) | 2011-06-22 | 2016-04-12 | Vertex Pharmaceuticals Incorporated | Substituted pyrrolo[2,3-b]pyrazines as ATR kinase inhibitors |
| US9334244B2 (en) | 2010-05-12 | 2016-05-10 | Vertex Pharmaceuticals Incorporated | Compounds useful as inhibitors of ATR kinase |
| US9340546B2 (en) | 2012-12-07 | 2016-05-17 | Vertex Pharmaceuticals Incorporated | Compounds useful as inhibitors of ATR kinase |
| CN105968114A (en) * | 2016-05-27 | 2016-09-28 | 山东大学 | Coelenterazine analog and preparing method and application thereof |
| US9630956B2 (en) | 2010-05-12 | 2017-04-25 | Vertex Pharmaceuticals Incorporated | Compounds useful as inhibitors of ATR kinase |
| US9663519B2 (en) | 2013-03-15 | 2017-05-30 | Vertex Pharmaceuticals Incorporated | Compounds useful as inhibitors of ATR kinase |
| US9670215B2 (en) | 2014-06-05 | 2017-06-06 | Vertex Pharmaceuticals Incorporated | Compounds useful as inhibitors of ATR kinase |
| US9791456B2 (en) | 2012-10-04 | 2017-10-17 | Vertex Pharmaceuticals Incorporated | Method for measuring ATR inhibition mediated increases in DNA damage |
| US10160760B2 (en) | 2013-12-06 | 2018-12-25 | Vertex Pharmaceuticals Incorporated | Compounds useful as inhibitors of ATR kinase |
| US10478430B2 (en) | 2012-04-05 | 2019-11-19 | Vertex Pharmaceuticals Incorporated | Compounds useful as inhibitors of ATR kinase and combination therapies thereof |
| US10813929B2 (en) | 2011-09-30 | 2020-10-27 | Vertex Pharmaceuticals Incorporated | Treating cancer with ATR inhibitors |
| US11046658B2 (en) | 2018-07-02 | 2021-06-29 | Incyte Corporation | Aminopyrazine derivatives as PI3K-γ inhibitors |
| US11179394B2 (en) | 2014-06-17 | 2021-11-23 | Vertex Pharmaceuticals Incorporated | Method for treating cancer using a combination of Chk1 and ATR inhibitors |
| US11464774B2 (en) | 2015-09-30 | 2022-10-11 | Vertex Pharmaceuticals Incorporated | Method for treating cancer using a combination of DNA damaging agents and ATR inhibitors |
| WO2023177233A1 (en) * | 2022-03-16 | 2023-09-21 | (주)아름테라퓨틱스 | Novel compound and use thereof for inhibiting checkpoint kinase 2 |
| US11926616B2 (en) | 2018-03-08 | 2024-03-12 | Incyte Corporation | Aminopyrazine diol compounds as PI3K-γ inhibitors |
-
1997
- 1997-05-06 WO PCT/GB1997/001227 patent/WO1997043267A1/en not_active Application Discontinuation
- 1997-05-06 EP EP97920835A patent/EP0914318A1/en not_active Withdrawn
- 1997-05-06 CA CA002253910A patent/CA2253910A1/en not_active Abandoned
Non-Patent Citations (1)
| Title |
|---|
| K.TERANISHI,T.GOTO: "SYNTHESIS AND CHEMILUMINESCENCE OF COELENTERAZINE.", BULLETIN OF THE CHEMICAL SOCIETY OF JAPAN, vol. 63, no. 11, 1990, TOKYO JP, pages 3132 - 3140, XP002037163 * |
Cited By (58)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7115621B2 (en) | 2001-04-25 | 2006-10-03 | Lilly Icos Llc | Chemical compounds |
| JP4901102B2 (en) * | 2002-05-03 | 2012-03-21 | エクセリクシス, インク. | Protein kinase modulator and method of use thereof |
| JP2005530760A (en) * | 2002-05-03 | 2005-10-13 | エクセリクシス, インク. | Protein kinase modulator and method of use thereof |
| AU2003234464B2 (en) * | 2002-05-03 | 2009-06-04 | Exelixis, Inc. | Protein kinase modulators and methods of use |
| WO2003093297A3 (en) * | 2002-05-03 | 2004-07-01 | Exelixis Inc | Protein kinase modulators and methods of use |
| US7704995B2 (en) | 2002-05-03 | 2010-04-27 | Exelixis, Inc. | Protein kinase modulators and methods of use |
| US8263585B2 (en) | 2007-05-04 | 2012-09-11 | Novartis Ag | Organic compounds |
| WO2009115517A3 (en) * | 2008-03-19 | 2010-01-07 | Novartis Ag | Pyridines and pyrazines as inhibitors of pi3k |
| JP2011515362A (en) * | 2008-03-19 | 2011-05-19 | ノバルティス アーゲー | Pyridines and pyrazines as PI3K inhibitors |
| US8268834B2 (en) | 2008-03-19 | 2012-09-18 | Novartis Ag | Pyrazine derivatives that inhibit phosphatidylinositol 3-kinase enzyme |
| EA018065B1 (en) * | 2008-03-19 | 2013-05-30 | Новартис Аг | Pyridines and pyrazines as pi3k inhibitors (phosphatidylinositol 3-kinase) |
| KR101284517B1 (en) | 2008-03-19 | 2013-07-23 | 노파르티스 아게 | Pyridines and pyrazines as inhibitors of pi3k |
| US8846687B2 (en) | 2008-03-19 | 2014-09-30 | Novartis Ag | Pyrazine compounds as PI3 kinase inhibitors |
| US10961232B2 (en) | 2008-12-19 | 2021-03-30 | Vertex Pharmaceuticals Incorporated | Substituted pyrazines as ATR kinase inhibitors |
| US10479784B2 (en) | 2008-12-19 | 2019-11-19 | Vertex Pharmaceuticals Incorporated | Substituted pyrazin-2-amines as inhibitors of ATR kinase |
| US9701674B2 (en) | 2008-12-19 | 2017-07-11 | Vertex Pharmaceuticals Incorporated | Substituted pyrazines as ATR kinase inhibitors |
| US8841308B2 (en) | 2008-12-19 | 2014-09-23 | Vertex Pharmaceuticals Incorporated | Pyrazin-2-amines useful as inhibitors of ATR kinase |
| US9365557B2 (en) | 2008-12-19 | 2016-06-14 | Vertex Pharmaceuticals Incorporated | Substituted pyrazin-2-amines as inhibitors of ATR kinase |
| US9630956B2 (en) | 2010-05-12 | 2017-04-25 | Vertex Pharmaceuticals Incorporated | Compounds useful as inhibitors of ATR kinase |
| US9334244B2 (en) | 2010-05-12 | 2016-05-10 | Vertex Pharmaceuticals Incorporated | Compounds useful as inhibitors of ATR kinase |
| JP2013529200A (en) * | 2010-05-12 | 2013-07-18 | バーテックス ファーマシューティカルズ インコーポレイテッド | Compounds useful as ATR kinase inhibitors |
| US20120035408A1 (en) * | 2010-05-12 | 2012-02-09 | Vertex Pharmaceuticals Incorporated | Compounds useful as inhibitors of atr kinase |
| US9062008B2 (en) | 2010-05-12 | 2015-06-23 | Vertex Pharmaceuticals Incorporated | Compounds useful as inhibitors of ATR kinase |
| US9096602B2 (en) | 2011-06-22 | 2015-08-04 | Vertex Pharmaceuticals Incorporated | Substituted pyrrolo[2,3-B]pyrazines as ATR kinase inhibitors |
| US9309250B2 (en) | 2011-06-22 | 2016-04-12 | Vertex Pharmaceuticals Incorporated | Substituted pyrrolo[2,3-b]pyrazines as ATR kinase inhibitors |
| US10813929B2 (en) | 2011-09-30 | 2020-10-27 | Vertex Pharmaceuticals Incorporated | Treating cancer with ATR inhibitors |
| US9035053B2 (en) | 2011-09-30 | 2015-05-19 | Vertex Pharmaceuticals Incorporated | Processes for making compounds useful as inhibitors of ATR kinase |
| US8846686B2 (en) | 2011-09-30 | 2014-09-30 | Vertex Pharmaceuticals Incorporated | Compounds useful as inhibitors of ATR kinase |
| US10822331B2 (en) | 2011-09-30 | 2020-11-03 | Vertex Pharmaceuticals Incorporated | Processes for preparing ATR inhibitors |
| US10208027B2 (en) | 2011-09-30 | 2019-02-19 | Vertex Pharmaceuticals Incorporated | Processes for preparing ATR inhibitors |
| US9862709B2 (en) | 2011-09-30 | 2018-01-09 | Vertex Pharmaceuticals Incorporated | Processes for making compounds useful as inhibitors of ATR kinase |
| US8853217B2 (en) | 2011-09-30 | 2014-10-07 | Vertex Pharmaceuticals Incorporated | Compounds useful as inhibitors of ATR kinase |
| US8846918B2 (en) | 2011-11-09 | 2014-09-30 | Vertex Pharmaceuticals Incorporated | Compounds useful as inhibitors of ATR kinase |
| CN102432614A (en) * | 2011-11-16 | 2012-05-02 | 泰州凯美迪生物医药技术有限公司 | Method for synthesizing coelenterazine |
| US11110086B2 (en) | 2012-04-05 | 2021-09-07 | Vertex Pharmaceuticals Incorporated | Compounds useful as inhibitors of ATR kinase and combination therapies thereof |
| US10478430B2 (en) | 2012-04-05 | 2019-11-19 | Vertex Pharmaceuticals Incorporated | Compounds useful as inhibitors of ATR kinase and combination therapies thereof |
| US9791456B2 (en) | 2012-10-04 | 2017-10-17 | Vertex Pharmaceuticals Incorporated | Method for measuring ATR inhibition mediated increases in DNA damage |
| US8912198B2 (en) | 2012-10-16 | 2014-12-16 | Vertex Pharmaceuticals Incorporated | Compounds useful as inhibitors of ATR kinase |
| US9340546B2 (en) | 2012-12-07 | 2016-05-17 | Vertex Pharmaceuticals Incorporated | Compounds useful as inhibitors of ATR kinase |
| US10392391B2 (en) | 2012-12-07 | 2019-08-27 | Vertex Pharmaceuticals Incorporated | Compounds useful as inhibitors of ATR kinase |
| US9718827B2 (en) | 2012-12-07 | 2017-08-01 | Vertex Pharmaceuticals Incorporated | Compounds useful as inhibitors of ATR kinase |
| US11370798B2 (en) | 2012-12-07 | 2022-06-28 | Vertex Pharmaceuticals Incorporated | Compounds useful as inhibitors of ATR kinase |
| US10787452B2 (en) | 2012-12-07 | 2020-09-29 | Vertex Pharmaceuticals Incorporated | Compounds useful as inhibitors of ATR kinase |
| US11117900B2 (en) | 2012-12-07 | 2021-09-14 | Vertex Pharmaceuticals Incorporated | Compounds useful as inhibitors of ATR kinase |
| US9650381B2 (en) | 2012-12-07 | 2017-05-16 | Vertex Pharmaceuticals Incorporated | Compounds useful as inhibitors of ATR kinase |
| US9663519B2 (en) | 2013-03-15 | 2017-05-30 | Vertex Pharmaceuticals Incorporated | Compounds useful as inhibitors of ATR kinase |
| US10160760B2 (en) | 2013-12-06 | 2018-12-25 | Vertex Pharmaceuticals Incorporated | Compounds useful as inhibitors of ATR kinase |
| US10815239B2 (en) | 2013-12-06 | 2020-10-27 | Vertex Pharmaceuticals Incorporated | Compounds useful as inhibitors of ATR kinase |
| US11485739B2 (en) | 2013-12-06 | 2022-11-01 | Vertex Pharmaceuticals Incorporated | Compounds useful as inhibitors of ATR kinase |
| US10093676B2 (en) | 2014-06-05 | 2018-10-09 | Vertex Pharmaceuticals Incorporated | Compounds useful as inhibitors of ATR kinase |
| US10800781B2 (en) | 2014-06-05 | 2020-10-13 | Vertex Pharmaceuticals Incorporated | Compounds useful as inhibitors of ATR kinase |
| US9670215B2 (en) | 2014-06-05 | 2017-06-06 | Vertex Pharmaceuticals Incorporated | Compounds useful as inhibitors of ATR kinase |
| US11179394B2 (en) | 2014-06-17 | 2021-11-23 | Vertex Pharmaceuticals Incorporated | Method for treating cancer using a combination of Chk1 and ATR inhibitors |
| US11464774B2 (en) | 2015-09-30 | 2022-10-11 | Vertex Pharmaceuticals Incorporated | Method for treating cancer using a combination of DNA damaging agents and ATR inhibitors |
| CN105968114A (en) * | 2016-05-27 | 2016-09-28 | 山东大学 | Coelenterazine analog and preparing method and application thereof |
| US11926616B2 (en) | 2018-03-08 | 2024-03-12 | Incyte Corporation | Aminopyrazine diol compounds as PI3K-γ inhibitors |
| US11046658B2 (en) | 2018-07-02 | 2021-06-29 | Incyte Corporation | Aminopyrazine derivatives as PI3K-γ inhibitors |
| WO2023177233A1 (en) * | 2022-03-16 | 2023-09-21 | (주)아름테라퓨틱스 | Novel compound and use thereof for inhibiting checkpoint kinase 2 |
Also Published As
| Publication number | Publication date |
|---|---|
| EP0914318A1 (en) | 1999-05-12 |
| CA2253910A1 (en) | 1997-11-20 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO1997043267A1 (en) | Pyrazines | |
| Gassman et al. | Generation of azasulfonium salts from halogen-sulfide complexes and anilines. Synthesis of indoles, oxindoles, and alkylated aromatic amines bearing cation stabilizing substituents | |
| KR100901402B1 (en) | Process for the preparation of a 14-hydroxynormorphinone derivative | |
| Ohta et al. | Syntheses and reactions of some 2, 5-disubstituted pyrazine monoxides | |
| CN111253395B (en) | Synthesis method of naphtho [1',2':4,5] imidazo [1,2-a ] pyridine-5, 6-diketone compound | |
| EP1045823B1 (en) | Process for making certain benzoic acid compounds | |
| KR20180116371A (en) | Process for producing 4-alkoxy-3-hydroxypicolic acid | |
| Morrow et al. | Intramolecular cyclization of 2'-olepinic side-chains on anodically oxidized 4-phenylphenols. The effect of olefin substituents on carbon—carbon bond formation | |
| Micouin et al. | Improvement of the Synthesis of Chiral Non-Racemic Bicyclic Lactams in the Piperidin-2-ones Series | |
| Anderson et al. | The reactions of 2-alkynylbenzaldehydes with hydrazides: a route to isoquinoline N-imines | |
| WO2000053554A1 (en) | Process for producing aldehydes | |
| Mladenova et al. | An Efficient Synthesis of Enediyne and Arenediyne Lactams | |
| US6235905B1 (en) | Process for preparing alkoxypyrazine derivatives | |
| US8168799B2 (en) | Process for the preparation of tetrazolytetrahydrocyclopentapyrazoles | |
| JP4399870B2 (en) | Process for producing 3-alkoxy-5-alkylpyrazin-2-amines | |
| WO2005021465A1 (en) | Method for producing aromatic unsaturated compound | |
| HU195659B (en) | Process for producing cotarnine | |
| Rogers et al. | Improved syntheses of 6‐hydroxy‐5‐methoxy‐and 5‐hydroxy‐6‐methoxyindoles and their O‐acetates, analogs of natural eumelanin precursors | |
| Cho et al. | Palladium‐catalyzed synthesis of 3‐oxo‐1, 3‐dihydro‐1‐isobenzofuranyl alkanoates from 2‐bromobenzaldehyde and sodium alkanoates | |
| WO2003082860A1 (en) | Processes for producing indole compound | |
| EP0008951B1 (en) | Steroid intermediate, method for production thereof, and method for producing another steroid intermediate therefrom | |
| Mohajer | A new catalyst for the synthesis of 2-substituted perimidines catalyzed by FePO4 [enter Paper Title] | |
| JPH09239275A (en) | Catalyst for synthesizing imine compound and production of imine compound using the same | |
| JPS584695B2 (en) | Method for producing cyclopentenone derivatives | |
| JPH0511110B2 (en) |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): CA JP US |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): AT BE CH DE DK ES FI FR GB GR IE IT LU MC NL PT SE |
|
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| ENP | Entry into the national phase |
Ref document number: 2253910 Country of ref document: CA Ref country code: CA Ref document number: 2253910 Kind code of ref document: A Format of ref document f/p: F |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1997920835 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: JP Ref document number: 97540617 Format of ref document f/p: F |
|
| WWP | Wipo information: published in national office |
Ref document number: 1997920835 Country of ref document: EP |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: 1997920835 Country of ref document: EP |