USRE39561E1 - Metallocenes containing ligands of 2-substituted indenyl derivatives, process for their preparation, and their use as catalysts - Google Patents
Metallocenes containing ligands of 2-substituted indenyl derivatives, process for their preparation, and their use as catalysts Download PDFInfo
- Publication number
- USRE39561E1 USRE39561E1 US08/895,950 US89595097A USRE39561E US RE39561 E1 USRE39561 E1 US RE39561E1 US 89595097 A US89595097 A US 89595097A US RE39561 E USRE39561 E US RE39561E
- Authority
- US
- United States
- Prior art keywords
- group
- different
- compound
- halogen atom
- alkyl group
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Lifetime
Links
- 239000003054 catalyst Substances 0.000 title claims abstract description 15
- -1 2-substituted indenyl Chemical class 0.000 title claims description 15
- 238000000034 method Methods 0.000 title claims description 14
- 238000002360 preparation method Methods 0.000 title abstract description 10
- 239000003446 ligand Substances 0.000 title description 9
- 125000004435 hydrogen atom Chemical class [H]* 0.000 claims abstract description 33
- 239000001257 hydrogen Substances 0.000 claims abstract description 19
- 229910052739 hydrogen Inorganic materials 0.000 claims abstract description 19
- 229920000098 polyolefin Polymers 0.000 claims abstract description 8
- 229910052735 hafnium Inorganic materials 0.000 claims abstract description 7
- 229910052726 zirconium Inorganic materials 0.000 claims abstract description 7
- 229910052736 halogen Inorganic materials 0.000 claims abstract description 4
- 150000002367 halogens Chemical class 0.000 claims abstract description 4
- 150000001875 compounds Chemical class 0.000 claims description 40
- 239000000203 mixture Substances 0.000 claims description 37
- 125000005843 halogen group Chemical group 0.000 claims description 30
- 125000000041 C6-C10 aryl group Chemical group 0.000 claims description 24
- 125000000008 (C1-C10) alkyl group Chemical group 0.000 claims description 23
- 239000000460 chlorine Substances 0.000 claims description 23
- 238000006116 polymerization reaction Methods 0.000 claims description 20
- VPGLGRNSAYHXPY-UHFFFAOYSA-L zirconium(2+);dichloride Chemical compound Cl[Zr]Cl VPGLGRNSAYHXPY-UHFFFAOYSA-L 0.000 claims description 17
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims description 15
- 229910052801 chlorine Inorganic materials 0.000 claims description 11
- 125000004178 (C1-C4) alkyl group Chemical group 0.000 claims description 9
- 125000006374 C2-C10 alkenyl group Chemical group 0.000 claims description 8
- 150000001336 alkenes Chemical class 0.000 claims description 8
- 229910052732 germanium Inorganic materials 0.000 claims description 8
- GNPVGFCGXDBREM-UHFFFAOYSA-N germanium atom Chemical compound [Ge] GNPVGFCGXDBREM-UHFFFAOYSA-N 0.000 claims description 8
- 229910052710 silicon Inorganic materials 0.000 claims description 8
- 239000010703 silicon Substances 0.000 claims description 8
- 125000000027 (C1-C10) alkoxy group Chemical group 0.000 claims description 7
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 claims description 7
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 claims description 7
- 125000003118 aryl group Chemical group 0.000 claims description 7
- 125000004429 atom Chemical group 0.000 claims description 7
- 229910052751 metal Inorganic materials 0.000 claims description 7
- 239000002184 metal Substances 0.000 claims description 7
- 125000002023 trifluoromethyl group Chemical group FC(F)(F)* 0.000 claims description 6
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical compound [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 claims description 5
- ATJFFYVFTNAWJD-UHFFFAOYSA-N Tin Chemical compound [Sn] ATJFFYVFTNAWJD-UHFFFAOYSA-N 0.000 claims description 5
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 claims description 5
- 230000000737 periodic effect Effects 0.000 claims description 5
- 229910052718 tin Inorganic materials 0.000 claims description 5
- 125000006273 (C1-C3) alkyl group Chemical group 0.000 claims description 4
- 125000006656 (C2-C4) alkenyl group Chemical group 0.000 claims description 4
- UGVVIPAIDGTTNN-UHFFFAOYSA-N C[Zr]C Chemical compound C[Zr]C UGVVIPAIDGTTNN-UHFFFAOYSA-N 0.000 claims description 4
- RTAQQCXQSZGOHL-UHFFFAOYSA-N Titanium Chemical compound [Ti] RTAQQCXQSZGOHL-UHFFFAOYSA-N 0.000 claims description 4
- QCWXUUIWCKQGHC-UHFFFAOYSA-N Zirconium Chemical compound [Zr] QCWXUUIWCKQGHC-UHFFFAOYSA-N 0.000 claims description 4
- 229910052731 fluorine Inorganic materials 0.000 claims description 4
- 239000011737 fluorine Substances 0.000 claims description 4
- VBJZVLUMGGDVMO-UHFFFAOYSA-N hafnium atom Chemical compound [Hf] VBJZVLUMGGDVMO-UHFFFAOYSA-N 0.000 claims description 4
- 239000000178 monomer Substances 0.000 claims description 4
- 125000001424 substituent group Chemical group 0.000 claims description 4
- 229910052719 titanium Inorganic materials 0.000 claims description 4
- 239000010936 titanium Substances 0.000 claims description 4
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical group [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 claims description 3
- JRZJOMJEPLMPRA-UHFFFAOYSA-N olefin Natural products CCCCCCCC=C JRZJOMJEPLMPRA-UHFFFAOYSA-N 0.000 claims description 3
- 125000006274 (C1-C3)alkoxy group Chemical group 0.000 claims description 2
- 125000000229 (C1-C4)alkoxy group Chemical group 0.000 claims description 2
- KZBUYRJDOAKODT-UHFFFAOYSA-N Chlorine Chemical compound ClCl KZBUYRJDOAKODT-UHFFFAOYSA-N 0.000 claims description 2
- PXGOKWXKJXAPGV-UHFFFAOYSA-N Fluorine Chemical compound FF PXGOKWXKJXAPGV-UHFFFAOYSA-N 0.000 claims description 2
- 125000001309 chloro group Chemical group Cl* 0.000 claims description 2
- 229910052758 niobium Inorganic materials 0.000 claims description 2
- 239000010955 niobium Substances 0.000 claims description 2
- GUCVJGMIXFAOAE-UHFFFAOYSA-N niobium atom Chemical compound [Nb] GUCVJGMIXFAOAE-UHFFFAOYSA-N 0.000 claims description 2
- 125000000538 pentafluorophenyl group Chemical group FC1=C(F)C(F)=C(*)C(F)=C1F 0.000 claims description 2
- 229910052715 tantalum Inorganic materials 0.000 claims description 2
- GUVRBAGPIYLISA-UHFFFAOYSA-N tantalum atom Chemical compound [Ta] GUVRBAGPIYLISA-UHFFFAOYSA-N 0.000 claims description 2
- 229910052720 vanadium Inorganic materials 0.000 claims description 2
- GPPXJZIENCGNKB-UHFFFAOYSA-N vanadium Chemical compound [V]#[V] GPPXJZIENCGNKB-UHFFFAOYSA-N 0.000 claims description 2
- 229920006395 saturated elastomer Polymers 0.000 claims 4
- 230000000379 polymerizing effect Effects 0.000 claims 2
- 238000002844 melting Methods 0.000 abstract description 12
- 230000008018 melting Effects 0.000 abstract description 12
- 125000000217 alkyl group Chemical group 0.000 abstract description 7
- 125000001188 haloalkyl group Chemical group 0.000 abstract 1
- 125000005842 heteroatom Chemical group 0.000 abstract 1
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 33
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 31
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 30
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 29
- 239000000243 solution Substances 0.000 description 23
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 20
- 0 CC.CC.[1*]C([2*])(C)C.[3*]c1c([5*])c(C[7*]Cc2c([6*])c([4*])c3c2CCCC3)c2c1CCCC2 Chemical compound CC.CC.[1*]C([2*])(C)C.[3*]c1c([5*])c(C[7*]Cc2c([6*])c([4*])c3c2CCCC3)c2c1CCCC2 0.000 description 17
- 230000015572 biosynthetic process Effects 0.000 description 15
- 238000003786 synthesis reaction Methods 0.000 description 15
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 15
- 239000002904 solvent Substances 0.000 description 14
- OFBQJSOFQDEBGM-UHFFFAOYSA-N Pentane Chemical compound CCCCC OFBQJSOFQDEBGM-UHFFFAOYSA-N 0.000 description 12
- 238000000921 elemental analysis Methods 0.000 description 11
- 150000003254 radicals Chemical class 0.000 description 11
- 239000005977 Ethylene Substances 0.000 description 10
- 238000006243 chemical reaction Methods 0.000 description 10
- 239000004743 Polypropylene Substances 0.000 description 9
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 9
- 239000000706 filtrate Substances 0.000 description 9
- 239000000047 product Substances 0.000 description 9
- 230000000694 effects Effects 0.000 description 7
- MZRVEZGGRBJDDB-UHFFFAOYSA-N N-Butyllithium Chemical compound [Li]CCCC MZRVEZGGRBJDDB-UHFFFAOYSA-N 0.000 description 6
- 229910019020 PtO2 Inorganic materials 0.000 description 6
- YKIOKAURTKXMSB-UHFFFAOYSA-N adams's catalyst Chemical compound O=[Pt]=O YKIOKAURTKXMSB-UHFFFAOYSA-N 0.000 description 6
- 239000000725 suspension Substances 0.000 description 6
- VGGSQFUCUMXWEO-UHFFFAOYSA-N Ethene Chemical compound C=C VGGSQFUCUMXWEO-UHFFFAOYSA-N 0.000 description 5
- 230000000052 comparative effect Effects 0.000 description 5
- 229920000642 polymer Polymers 0.000 description 5
- 238000000425 proton nuclear magnetic resonance spectrum Methods 0.000 description 5
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 5
- HEDRZPFGACZZDS-MICDWDOJSA-N Trichloro(2H)methane Chemical compound [2H]C(Cl)(Cl)Cl HEDRZPFGACZZDS-MICDWDOJSA-N 0.000 description 4
- 239000007795 chemical reaction product Substances 0.000 description 4
- 125000003454 indenyl group Chemical group C1(C=CC2=CC=CC=C12)* 0.000 description 4
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 4
- 239000007788 liquid Substances 0.000 description 4
- 239000000463 material Substances 0.000 description 4
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Chemical compound [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 description 4
- 239000000377 silicon dioxide Substances 0.000 description 4
- 239000007787 solid Substances 0.000 description 4
- YBYIRNPNPLQARY-UHFFFAOYSA-N 1H-indene Natural products C1=CC=C2CC=CC2=C1 YBYIRNPNPLQARY-UHFFFAOYSA-N 0.000 description 3
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 3
- 238000005481 NMR spectroscopy Methods 0.000 description 3
- 229910052681 coesite Inorganic materials 0.000 description 3
- 238000001816 cooling Methods 0.000 description 3
- 229910052906 cristobalite Inorganic materials 0.000 description 3
- 229930195733 hydrocarbon Natural products 0.000 description 3
- 150000002430 hydrocarbons Chemical class 0.000 description 3
- 238000005984 hydrogenation reaction Methods 0.000 description 3
- 239000012442 inert solvent Substances 0.000 description 3
- 239000002243 precursor Substances 0.000 description 3
- QQONPFPTGQHPMA-UHFFFAOYSA-N propylene Natural products CC=C QQONPFPTGQHPMA-UHFFFAOYSA-N 0.000 description 3
- 125000004805 propylene group Chemical group [H]C([H])([H])C([H])([*:1])C([H])([H])[*:2] 0.000 description 3
- 150000003839 salts Chemical class 0.000 description 3
- 229910052682 stishovite Inorganic materials 0.000 description 3
- 238000006467 substitution reaction Methods 0.000 description 3
- 229910052723 transition metal Inorganic materials 0.000 description 3
- 150000003624 transition metals Chemical class 0.000 description 3
- 229910052905 tridymite Inorganic materials 0.000 description 3
- LIKMAJRDDDTEIG-UHFFFAOYSA-N 1-hexene Chemical compound CCCCC=C LIKMAJRDDDTEIG-UHFFFAOYSA-N 0.000 description 2
- KWKAKUADMBZCLK-UHFFFAOYSA-N 1-octene Chemical compound CCCCCCC=C KWKAKUADMBZCLK-UHFFFAOYSA-N 0.000 description 2
- 238000001644 13C nuclear magnetic resonance spectroscopy Methods 0.000 description 2
- WSSSPWUEQFSQQG-UHFFFAOYSA-N 4-methyl-1-pentene Chemical compound CC(C)CC=C WSSSPWUEQFSQQG-UHFFFAOYSA-N 0.000 description 2
- NLXLAEXVIDQMFP-UHFFFAOYSA-N Ammonia chloride Chemical compound [NH4+].[Cl-] NLXLAEXVIDQMFP-UHFFFAOYSA-N 0.000 description 2
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 2
- 239000004215 Carbon black (E152) Substances 0.000 description 2
- LCGLNKUTAGEVQW-UHFFFAOYSA-N Dimethyl ether Chemical compound COC LCGLNKUTAGEVQW-UHFFFAOYSA-N 0.000 description 2
- YCKRFDGAMUMZLT-UHFFFAOYSA-N Fluorine atom Chemical compound [F] YCKRFDGAMUMZLT-UHFFFAOYSA-N 0.000 description 2
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 2
- CSNNHWWHGAXBCP-UHFFFAOYSA-L Magnesium sulfate Chemical compound [Mg+2].[O-][S+2]([O-])([O-])[O-] CSNNHWWHGAXBCP-UHFFFAOYSA-L 0.000 description 2
- IMNFDUFMRHMDMM-UHFFFAOYSA-N N-Heptane Chemical compound CCCCCCC IMNFDUFMRHMDMM-UHFFFAOYSA-N 0.000 description 2
- KDLHZDBZIXYQEI-UHFFFAOYSA-N Palladium Chemical compound [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 description 2
- 239000004698 Polyethylene Substances 0.000 description 2
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical compound [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 2
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 2
- 229910007932 ZrCl4 Inorganic materials 0.000 description 2
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 2
- 125000004432 carbon atom Chemical group C* 0.000 description 2
- 238000004440 column chromatography Methods 0.000 description 2
- YMWUJEATGCHHMB-DICFDUPASA-N dichloromethane-d2 Chemical compound [2H]C([2H])(Cl)Cl YMWUJEATGCHHMB-DICFDUPASA-N 0.000 description 2
- 239000007789 gas Substances 0.000 description 2
- 239000000499 gel Substances 0.000 description 2
- 150000002431 hydrogen Chemical class 0.000 description 2
- 150000002469 indenes Chemical class 0.000 description 2
- 230000010354 integration Effects 0.000 description 2
- DLEDOFVPSDKWEF-UHFFFAOYSA-N lithium butane Chemical compound [Li+].CCC[CH2-] DLEDOFVPSDKWEF-UHFFFAOYSA-N 0.000 description 2
- CPOFMOWDMVWCLF-UHFFFAOYSA-N methyl(oxo)alumane Chemical compound C[Al]=O CPOFMOWDMVWCLF-UHFFFAOYSA-N 0.000 description 2
- UAEPNZWRGJTJPN-UHFFFAOYSA-N methylcyclohexane Chemical compound CC1CCCCC1 UAEPNZWRGJTJPN-UHFFFAOYSA-N 0.000 description 2
- DVSDBMFJEQPWNO-UHFFFAOYSA-N methyllithium Chemical compound C[Li] DVSDBMFJEQPWNO-UHFFFAOYSA-N 0.000 description 2
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 2
- 230000037048 polymerization activity Effects 0.000 description 2
- 229920001155 polypropylene Polymers 0.000 description 2
- 230000008092 positive effect Effects 0.000 description 2
- 229910052701 rubidium Inorganic materials 0.000 description 2
- 239000012266 salt solution Substances 0.000 description 2
- JOXIMZWYDAKGHI-UHFFFAOYSA-N toluene-4-sulfonic acid Chemical compound CC1=CC=C(S(O)(=O)=O)C=C1 JOXIMZWYDAKGHI-UHFFFAOYSA-N 0.000 description 2
- DUNKXUFBGCUVQW-UHFFFAOYSA-J zirconium tetrachloride Chemical compound Cl[Zr](Cl)(Cl)Cl DUNKXUFBGCUVQW-UHFFFAOYSA-J 0.000 description 2
- 125000004169 (C1-C6) alkyl group Chemical group 0.000 description 1
- PAAZPARNPHGIKF-UHFFFAOYSA-N 1,2-dibromoethane Chemical compound BrCCBr PAAZPARNPHGIKF-UHFFFAOYSA-N 0.000 description 1
- 238000005160 1H NMR spectroscopy Methods 0.000 description 1
- JWVVFPXYZALZDT-UHFFFAOYSA-N 2-methyl-1,3-dihydroinden-2-ol Chemical compound C1=CC=C2CC(C)(O)CC2=C1 JWVVFPXYZALZDT-UHFFFAOYSA-N 0.000 description 1
- YSAXEHWHSLANOM-UHFFFAOYSA-N 2-methyl-1h-indene Chemical compound C1=CC=C2CC(C)=CC2=C1 YSAXEHWHSLANOM-UHFFFAOYSA-N 0.000 description 1
- PNEYBMLMFCGWSK-UHFFFAOYSA-N Alumina Chemical class [O-2].[O-2].[O-2].[Al+3].[Al+3] PNEYBMLMFCGWSK-UHFFFAOYSA-N 0.000 description 1
- VYZAMTAEIAYCRO-UHFFFAOYSA-N Chromium Chemical compound [Cr] VYZAMTAEIAYCRO-UHFFFAOYSA-N 0.000 description 1
- XDTMQSROBMDMFD-UHFFFAOYSA-N Cyclohexane Chemical compound C1CCCCC1 XDTMQSROBMDMFD-UHFFFAOYSA-N 0.000 description 1
- XTHFKEDIFFGKHM-UHFFFAOYSA-N Dimethoxyethane Chemical compound COCCOC XTHFKEDIFFGKHM-UHFFFAOYSA-N 0.000 description 1
- NHTMVDHEPJAVLT-UHFFFAOYSA-N Isooctane Chemical compound CC(C)CC(C)(C)C NHTMVDHEPJAVLT-UHFFFAOYSA-N 0.000 description 1
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 description 1
- ZOKXTWBITQBERF-UHFFFAOYSA-N Molybdenum Chemical compound [Mo] ZOKXTWBITQBERF-UHFFFAOYSA-N 0.000 description 1
- CTQNGGLPUBDAKN-UHFFFAOYSA-N O-Xylene Chemical compound CC1=CC=CC=C1C CTQNGGLPUBDAKN-UHFFFAOYSA-N 0.000 description 1
- 229910000831 Steel Inorganic materials 0.000 description 1
- 229910007926 ZrCl Inorganic materials 0.000 description 1
- 125000001931 aliphatic group Chemical group 0.000 description 1
- 150000001338 aliphatic hydrocarbons Chemical class 0.000 description 1
- 229910052783 alkali metal Inorganic materials 0.000 description 1
- 150000001340 alkali metals Chemical class 0.000 description 1
- 125000003342 alkenyl group Chemical group 0.000 description 1
- 229910052782 aluminium Inorganic materials 0.000 description 1
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 1
- 235000019270 ammonium chloride Nutrition 0.000 description 1
- 150000004945 aromatic hydrocarbons Chemical class 0.000 description 1
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 description 1
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 description 1
- 229910052794 bromium Inorganic materials 0.000 description 1
- 244000309464 bull Species 0.000 description 1
- 239000001273 butane Substances 0.000 description 1
- 239000013522 chelant Substances 0.000 description 1
- 238000004587 chromatography analysis Methods 0.000 description 1
- 229910052804 chromium Inorganic materials 0.000 description 1
- 239000011651 chromium Substances 0.000 description 1
- 238000007334 copolymerization reaction Methods 0.000 description 1
- 239000012043 crude product Substances 0.000 description 1
- 239000013078 crystal Substances 0.000 description 1
- 238000002425 crystallisation Methods 0.000 description 1
- 230000008025 crystallization Effects 0.000 description 1
- 125000004122 cyclic group Chemical group 0.000 description 1
- 230000005595 deprotonation Effects 0.000 description 1
- 238000010537 deprotonation reaction Methods 0.000 description 1
- 239000002283 diesel fuel Substances 0.000 description 1
- FZTMUIDAMRKQPP-UHFFFAOYSA-N dimethyl-(2-methyl-1H-inden-1-yl)silane Chemical compound C1=CC=C2C([SiH](C)C)C(C)=CC2=C1 FZTMUIDAMRKQPP-UHFFFAOYSA-N 0.000 description 1
- JVSWJIKNEAIKJW-UHFFFAOYSA-N dimethyl-hexane Natural products CCCCCC(C)C JVSWJIKNEAIKJW-UHFFFAOYSA-N 0.000 description 1
- LIKFHECYJZWXFJ-UHFFFAOYSA-N dimethyldichlorosilane Chemical compound C[Si](C)(Cl)Cl LIKFHECYJZWXFJ-UHFFFAOYSA-N 0.000 description 1
- 238000001035 drying Methods 0.000 description 1
- 230000008030 elimination Effects 0.000 description 1
- 238000003379 elimination reaction Methods 0.000 description 1
- NLFBCYMMUAKCPC-KQQUZDAGSA-N ethyl (e)-3-[3-amino-2-cyano-1-[(e)-3-ethoxy-3-oxoprop-1-enyl]sulfanyl-3-oxoprop-1-enyl]sulfanylprop-2-enoate Chemical compound CCOC(=O)\C=C\SC(=C(C#N)C(N)=O)S\C=C\C(=O)OCC NLFBCYMMUAKCPC-KQQUZDAGSA-N 0.000 description 1
- 238000001704 evaporation Methods 0.000 description 1
- 230000008020 evaporation Effects 0.000 description 1
- 239000000284 extract Substances 0.000 description 1
- 238000010528 free radical solution polymerization reaction Methods 0.000 description 1
- 238000010438 heat treatment Methods 0.000 description 1
- UMJJFEIKYGFCAT-UHFFFAOYSA-N indan-2-one Chemical compound C1=CC=C2CC(=O)CC2=C1 UMJJFEIKYGFCAT-UHFFFAOYSA-N 0.000 description 1
- PNDPGZBMCMUPRI-UHFFFAOYSA-N iodine Chemical compound II PNDPGZBMCMUPRI-UHFFFAOYSA-N 0.000 description 1
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 229920001580 isotactic polymer Polymers 0.000 description 1
- 229910052744 lithium Inorganic materials 0.000 description 1
- 229910052943 magnesium sulfate Inorganic materials 0.000 description 1
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 description 1
- GYNNXHKOJHMOHS-UHFFFAOYSA-N methyl-cycloheptane Natural products CC1CCCCCC1 GYNNXHKOJHMOHS-UHFFFAOYSA-N 0.000 description 1
- 229910052750 molybdenum Inorganic materials 0.000 description 1
- 239000011733 molybdenum Substances 0.000 description 1
- 239000012452 mother liquor Substances 0.000 description 1
- 238000000465 moulding Methods 0.000 description 1
- TVMXDCGIABBOFY-UHFFFAOYSA-N n-Octanol Natural products CCCCCCCC TVMXDCGIABBOFY-UHFFFAOYSA-N 0.000 description 1
- IJDNQMDRQITEOD-UHFFFAOYSA-N n-butane Chemical compound CCCC IJDNQMDRQITEOD-UHFFFAOYSA-N 0.000 description 1
- 229910052757 nitrogen Inorganic materials 0.000 description 1
- SJYNFBVQFBRSIB-UHFFFAOYSA-N norbornadiene Chemical compound C1=CC2C=CC1C2 SJYNFBVQFBRSIB-UHFFFAOYSA-N 0.000 description 1
- JFNLZVQOOSMTJK-KNVOCYPGSA-N norbornene Chemical compound C1[C@@H]2CC[C@H]1C=C2 JFNLZVQOOSMTJK-KNVOCYPGSA-N 0.000 description 1
- 238000006384 oligomerization reaction Methods 0.000 description 1
- 239000012074 organic phase Substances 0.000 description 1
- MUMZUERVLWJKNR-UHFFFAOYSA-N oxoplatinum Chemical compound [Pt]=O MUMZUERVLWJKNR-UHFFFAOYSA-N 0.000 description 1
- 229910052763 palladium Inorganic materials 0.000 description 1
- 239000002245 particle Substances 0.000 description 1
- 239000003208 petroleum Substances 0.000 description 1
- 239000012071 phase Substances 0.000 description 1
- 229910052697 platinum Inorganic materials 0.000 description 1
- 229910003446 platinum oxide Inorganic materials 0.000 description 1
- 229920000573 polyethylene Polymers 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 239000002244 precipitate Substances 0.000 description 1
- XOJVVFBFDXDTEG-UHFFFAOYSA-N pristane Chemical compound CC(C)CCCC(C)CCCC(C)CCCC(C)C XOJVVFBFDXDTEG-UHFFFAOYSA-N 0.000 description 1
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 230000001681 protective effect Effects 0.000 description 1
- 238000010992 reflux Methods 0.000 description 1
- 239000000741 silica gel Substances 0.000 description 1
- 229910002027 silica gel Inorganic materials 0.000 description 1
- 229910000030 sodium bicarbonate Inorganic materials 0.000 description 1
- 239000011780 sodium chloride Substances 0.000 description 1
- 238000010561 standard procedure Methods 0.000 description 1
- 239000007858 starting material Substances 0.000 description 1
- 239000010959 steel Substances 0.000 description 1
- 230000000707 stereoselective effect Effects 0.000 description 1
- 238000003756 stirring Methods 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- 238000010557 suspension polymerization reaction Methods 0.000 description 1
- 230000036962 time dependent Effects 0.000 description 1
- 239000012485 toluene extract Substances 0.000 description 1
- 150000003623 transition metal compounds Chemical class 0.000 description 1
- WFKWXMTUELFFGS-UHFFFAOYSA-N tungsten Chemical compound [W] WFKWXMTUELFFGS-UHFFFAOYSA-N 0.000 description 1
- 229910052721 tungsten Inorganic materials 0.000 description 1
- 239000010937 tungsten Substances 0.000 description 1
- 230000002747 voluntary effect Effects 0.000 description 1
- 239000008096 xylene Substances 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F17/00—Metallocenes
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F7/00—Compounds containing elements of Groups 4 or 14 of the Periodic Table
- C07F7/02—Silicon compounds
- C07F7/08—Compounds having one or more C—Si linkages
- C07F7/0803—Compounds with Si-C or Si-Si linkages
- C07F7/0805—Compounds with Si-C or Si-Si linkages comprising only Si, C or H atoms
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F10/00—Homopolymers and copolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F110/00—Homopolymers of unsaturated aliphatic hydrocarbons having only one carbon-to-carbon double bond
- C08F110/04—Monomers containing three or four carbon atoms
- C08F110/06—Propene
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F4/00—Polymerisation catalysts
- C08F4/42—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors
- C08F4/44—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors selected from light metals, zinc, cadmium, mercury, copper, silver, gold, boron, gallium, indium, thallium, rare earths or actinides
- C08F4/60—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors selected from light metals, zinc, cadmium, mercury, copper, silver, gold, boron, gallium, indium, thallium, rare earths or actinides together with refractory metals, iron group metals, platinum group metals, manganese, rhenium technetium or compounds thereof
- C08F4/62—Refractory metals or compounds thereof
- C08F4/639—Component covered by group C08F4/62 containing a transition metal-carbon bond
- C08F4/63912—Component covered by group C08F4/62 containing a transition metal-carbon bond in combination with an organoaluminium compound
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F4/00—Polymerisation catalysts
- C08F4/42—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors
- C08F4/44—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors selected from light metals, zinc, cadmium, mercury, copper, silver, gold, boron, gallium, indium, thallium, rare earths or actinides
- C08F4/60—Metals; Metal hydrides; Metallo-organic compounds; Use thereof as catalyst precursors selected from light metals, zinc, cadmium, mercury, copper, silver, gold, boron, gallium, indium, thallium, rare earths or actinides together with refractory metals, iron group metals, platinum group metals, manganese, rhenium technetium or compounds thereof
- C08F4/62—Refractory metals or compounds thereof
- C08F4/639—Component covered by group C08F4/62 containing a transition metal-carbon bond
- C08F4/6392—Component covered by group C08F4/62 containing a transition metal-carbon bond containing at least one cyclopentadienyl ring, condensed or not, e.g. an indenyl or a fluorenyl ring
- C08F4/63922—Component covered by group C08F4/62 containing a transition metal-carbon bond containing at least one cyclopentadienyl ring, condensed or not, e.g. an indenyl or a fluorenyl ring containing at least two cyclopentadienyl rings, fused or not
- C08F4/63927—Component covered by group C08F4/62 containing a transition metal-carbon bond containing at least one cyclopentadienyl ring, condensed or not, e.g. an indenyl or a fluorenyl ring containing at least two cyclopentadienyl rings, fused or not two cyclopentadienyl rings being mutually bridged
Definitions
- the present invention relates to novel metallocenes which contain ligands of 2-substituted indenyl derivatives and can very advantageously be used as catalysts in the preparation of polyolefins of high melting point (high isotacticity).
- Polyolefins of relatively high melting point and thus relatively high crystallinity and relatively high hardness are particularly important as engineering materials (for example large hollow articles, tubes and moldings).
- Chiral metallocenes are, in combination with aluminoxanes, active, stereospecific catalysts for the preparation of polyolefins (U.S. Pat. No. 4,769,510). These metallocenes also include substituted indene compounds.
- aluminoxanes active, stereospecific catalysts for the preparation of polyolefins
- These metallocenes also include substituted indene compounds.
- the use of the ethylenebis(4,5,6,7-tetrahydro-1-indenyl)zirconium dichloride/aluminoxane catalyst system is known for the preparation of isotactic polypropylene; cf. EP-A 185 918). Both this and numerous other polymerization processes coming under the prior art have, in particular, the disadvantage that, at industrially interesting polymerization temperatures, only polymers of relatively low melting points are obtained. Their crystallinity and thus their hardness are too low for use as engineering materials.
- metallocenes which contain, as ligands, certain 2-substituted indenyl derivatives are suitable catalysts for the preparation of polyolefins of high isotacticity (melting point) and narrow molecular weight distribution.
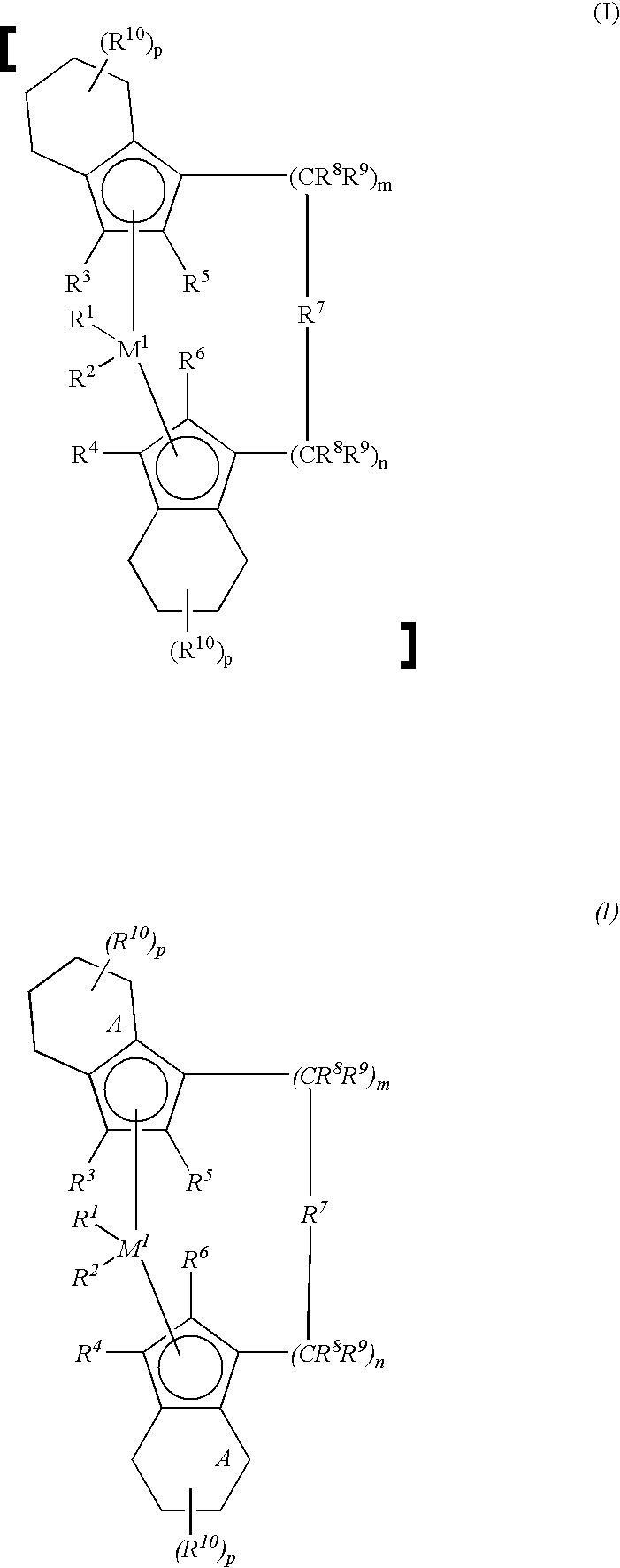
- M 1 is a metal from group IVb, Vb or VIb of the Periodic Table, for example titanium, zirconium, hafnium, vanadium, niobium, tantalum, chromium, molybdenum or tungsten, preferably zirconium, hafnium or titanium.
- the radicals R 10 are identical or different and are as defined for R 11 , R 12 and R 13 .
- the radicals R 10 are preferably hydrogen atoms or C 1 -C 10 -, preferably C 1 -C 4 -alkyl groups.
- the particularly preferred metallocenes are thus those in which, in the formula I, M 1 is Zr or Hf, R 1 and R 2 are identical or different and are methyl or chlorine, R 3 and R 4 are hydrogen, R 5 and R 6 are identical or different and are methyl, ethyl or trifluoromethyl, R 7 is a radical, n plus m is zero or 1, and R 10 is hydrogen; in particular the compounds I listed in the working examples.
- rac-dimethylsilyl(2-methyl-4,5,6,7-tetrahydro-1-indenyl) 2 zirconium dichloride rac-ethylene(2-methyl-4,5,6,7-tetrahydro-1-indenyl) 2 zirconium dichloride, racdimethylsilyl(2-methyl-4,5,6,7-tetrahydro-1-indenyl) 2 dimethylzirconium and rac-ethylene(2-methyl-4,5,6,7-tetrahydro-1-indenyl) 2 dimethylzirconium are particularly important.
- the chiral metallocenes are employed as racemates for the preparation of highly isotactic poly-1-olefins. However, it is also possible to use the pure R- or S-form. These pure stereoisomeric forms allow the preparation of an optically active polymer. However, the meso form of the metallocenes should be separated off since the polymerization-active center (the metal atom) in these compounds is no longer chiral due to mirror symmetry at the central metal, and it is therefore not possible to produce a highly isotactic polymer.
- the present invention furthermore provides a process for the preparation of the metallocenes I, which comprises
- the synthesis is carried out under a protective gas and in anhydrous solvents.
- the dried salt of the formula II/IIa is added to a suspension of the compound of the formula III in a solvent such as toluene, n-hexane, dichloromethane, ether, THF, n-pentane or benzene, preferably dichloromethane or toluene.
- the reaction temperature is from ⁇ 78° C. to 30° C., preferably from ⁇ 40° C. to 10° C.
- the reaction duration is from 0.25 to 24 hours, preferably from 1 to 4 hours.
- a further embodiment of the process according to the invention comprises replacing the compound III, M 1 X 4 , by a compound of the formula IIIa, M 1 X 4 L 2 .
- L is a donor ligand.
- suitable donor ligands are tetrahydrofuran, diethyl ether, dimethyl ether, inter alia, preferably tetrahydrofuran (THF).
- a solution of the salt of the formula II/IIa in one of the abovementioned solvents is added to a solution or suspension of a compound of the formula IIIa in a solvent such as toluene, xylene, ether or THF, preferably THF.
- a solvent such as toluene, xylene, ether or THF, preferably THF.
- an alternative procedure is to simultaneously add both components dropwise to a solvent. This is the preferred procedure.
- the reaction temperature is from ⁇ 40° C. to 100° C., preferably from 0° C. to 50° C., in particular from 10° C. to 35° C.
- the reaction duration is from 0.25 hour to 48 hours, preferably from 1 hour to 24 hours, in particular from 2 hours to 9 hours.
- the hydrogenation is carried out in a dry, anhydrous solvent such as H 2 CCl 2 or glyme.
- the reaction temperature is 20° to 70° C., preferably from ambient temperature to 50° C.
- the pressure is from 5 to 200 bar, preferably from 20 to 120 bar, in particular from 35 to 100 bar
- the reaction duration is from 0.25 to 24 hours, preferably from 0.5 to 18 hours, in particular from 1 to 12 hours.
- Hydrogenation reactors which can be used are steel autoclaves.
- the hydrogenation catalyst used is platinum, platinum oxide, palladium or another conventional transition-metal catalyst.
- halogen derivatives obtained in this way can be converted into the alkyl, aryl or alkenyl complexes by known standard methods.
- the compounds of the formulae II and IIa are synthesized by deprotonation. This reaction is known; cf. J. Am. Chem. Soc., 112 (1990) 2030-2031, ibid. 110 (1988) 6255-6256, ibid. 109 (1987), 6544-6545, J. Organomet. Chem., 322 (1987) 65-70, New. J. Chem. 14 (1990) 499-503 and the working examples.
- the metallocenes I can thus in principle be prepared in accordance with the reaction scheme below:
- the cocatalyst used according to the invention in the polymerization of olefins is an aluminoxane of the formula (IV) for the linear type and/or of the formula (V) for the cyclic type, where, in the formulae (IV) and (V), the radicals R may be identical or different and are a C 1 -C 6 -alkyl group, a C 6 -C 18 -aryl group or hydrogen, and p is an integer from 2 to 50, preferably from 10 to 35.
- the radicals R are preferably identical and are methyl, isobutyl, phenyl or benzyl, particularly preferably methyl.
- radicals R are different, they are preferably methyl and hydrogen or alternatively methyl and isobutyl, preferably from 0.01 to 40% (of the number of radicals R) being hydrogen or isobutyl.
- the aluminoxane can be prepared in different ways by known processes.
- One of the methods is, for example, the reaction of an aluminium-hydrocarbon compound and/or a hydridoaluminum-hydrocarbon compound with water (gaseous, solid, liquid or bound—for example as water of crystallization) in an inert solvent (such as, for example, toluene).
- an inert solvent such as, for example, toluene.
- two different trialkylaluminum compounds AlR 3 +AlR′ 3
- water cf. S. Pasynkiewicz, Polyhedron 9 (1990) 429 and EP-A 302 424).
- the preactivation of the transition-metal compound is carried out in solution.
- the metallocene is preferably dissolved in a solution of the aluminoxane in an inert hydrocarbon.
- Suitable inert hydrocarbons are aliphatic or aromatic hydrocarbons. Toluene is preferred.
- the concentration of the aluminoxane in the solution is in the range from about 1% by weight up to the saturation limit, preferably from 5 to 30% by weight, in each case based on the entire solution.
- the metallocene can be employed in the same concentration, but is preferably employed in an amount of from 10 ⁇ 4 ⁇ 1 mol per mole of aluminoxane.
- the preactivation time is from 5 minutes to 60 hours, preferably from 5 to 60 minutes.
- the preactivation temperature is from ⁇ 78° C. to 100° C., preferably from 0 to 70° C.
- the metallocene can also be prepolymerized or applied to a support.
- the prepolymerization is preferably carried out using the olefin (or one of the olefins) employed in the polymerization.
- suitable supports are silica gels, aluminum oxides, solid aluminoxane or other inorganic support materials.
- Another suitable support material is a polyolefin powder in finely divided form.
- a further possible variation of the process comprises using a salt-like compound of the formula R x NH 4-x BR′ 4 or of the formula R 3 PHBR′ 4 as cocatalyst instead of or in addition to an aluminoxane.
- x here is 1, 2 or 3, the R radicals are identical or different and are alkyl or aryl, and R′ is aryl, which may also be fluorinated or partially fluorinated.
- the catalyst comprises the product of the reaction of a metallocene with one of said compounds (cf. EP-A 277 004).
- the polymerization or copolymerization is carried out in a known manner in solution, in suspension or in the gas phase, continuously or batchwise, in one or more steps, at a temperature of from 0° to 150° C., preferably from 30° to 80° C.
- Olefins of the formula R a —CH ⁇ CH—R b are polymerized or copolymerized.
- R a and R b are identical or different and are a hydrogen atom or an alkyl radical having 1 to 14 carbon atoms.
- R a and R b together with the carbon atoms connecting them, may also form a ring.
- olefins are ethylene, propylene, 1-butene, 1-hexene, 4-methyl-1-pentene, 1-octene, norbornene or norbornadiene.
- propylene and ethylene are polymerized.
- the molecular weight regulator added is hydrogen.
- the overall pressure in the polymerization system is from 0.5 to 100 bar.
- the polymerization is preferably carried out in the industrially particularly interesting pressure range of from 5 to 64 bar.
- the metallocene is used in a concentration, based on the transition metal, of from 10 ⁇ 3 to 10 ⁇ 8 , preferably from 10 ⁇ 4 to 10 ⁇ 7 mol of transition metal per dm 3 of solvent or per dm 3 of reactor volume.
- the aluminoxane is used in a concentration of from 10 ⁇ 5 to 10 ⁇ 1 mol, preferably from 10 ⁇ 4 to 10 ⁇ 2 mol, per dm 3 of solvent or per dm 3 of reactor volume. In principle, however, higher concentrations are also possible.
- the polymerization is carried out as a suspension or solution polymerization, an inert solvent which is customary for the Ziegler low-pressure process is used.
- the polymerization is carried out in an aliphatic or cycloaliphatic hydrocarbon; examples of these which may be mentioned are butane, pentane, hexane, heptane, isooctane, cyclohexane and methylcyclohexane.
- the polymerization is preferably carried out in the liquid monomer.
- the monomers are metered in in gaseous or liquid form.
- the polymerization can have any desired duration since the catalyst system to be used according to the invention exhibits only a low time-dependent drop in polymerization activity.
- the process is distinguished by the fact that the metallocenes according to the invention give, in the industrially interesting temperature range of between 30° and 80° C, polymers of high molecular weight, high stereospecificity, narrow molecular weight dispersity and, in particular, high melting point, which is to say high crystallinity and high hardness.
- VN viscosity number in cm 3 /g determined
- M w weight average molecular weight by gel perme- M w /M
- the melting points and heats of melting ⁇ H melt were determined using DSC (heating and cooling rate 20° C./min).
- the 1 H-NMR spectrum exhibits the signals expected for an isomer mixture with respect to shift and integration ratio.
- the 1 H-NMR spectrum corresponds to expectations for an isomer mixture in signal shift and integration.
- the dilithio salt obtained in this way was added at ⁇ 78° C. to a suspension of 1.24 g (5.32 mmol) of ZrCl in 50 cm 3 of CH 2 Cl 2 , and the mixture was stirred at this temperature for 3 hours. The mixture was then warmed to room temperature overnight and evaporated.
- the 1 H-NMR spectrum showed, in addition to the presence of some ZrCl 4 (thf) 2 , a rac/meso mixture. After stirring with n-pentane and drying, the solid, yellow residue was suspended in THF, filtered off and examined by NMR spectroscopy. These three working steps were repeated a number of times; finally, 0.35 g (0.73 mmol/14%) of product was obtained in which the rac form, according to 1 H-NMR, was enriched to more than 17:1.
- ⁇ 0.90 (s, 6H, Me-Si); 1.43-1.93 (m, 8H, indenyl-H); 2.10 (s, 6H, 2-Me); 2.44-3.37 (m, 8H, indenyl-H); 6.05 (s, 2H, 3-H-Ind).
- Ind indenyl
- THF tetrahydrofuran
- PP polypropylene
- PE polyethylene
- Metallocenes I as catalysts for the polymerization of olefins.
- the activity of the metallocene was 50.3 kg of PP/g of metallocene ⁇ h.
- Example 1 was repeated, but 19.5 mg (0.04 mmol) of the metallocene were employed, and the polymerization temperature was 50° C.
- the activity of the metallocene was 18.8 kg of PP/g of metallocene ⁇ h.
- Example 1 was repeated, but 58.0 mg (0.12 mmol) of the metallocene were used and the polymerization temperature was 30° C.
- the activity of the metallocene was 9.7 kg of PP/g of metallocene ⁇ h.
- Examples 1 to 3 were repeated, but the metallocenes dimethylsilyl(2-Me-1-indenyl) 2 zirconium dichloride (metallocene 1 ), dimethylsilyl(4,5,6,7-tetrahydro-1-indenyl) 2 zirconium dichloride (metallocene 2) and dimethylsilyl(1-indenyl) 2 zirconium dichloride (metallocene 3) were used.
- Comparative Examples F/G with D/E confirms the positive effect of the 4,5,6,7-tetrahydroindenyl ligand compared with indenyl
- Comparative Examples F/G/H compared with A/B/C show the positive effect of the substitution in the 2-position of the indenyl ligand.
- Example 1 was repeated, but 6.8 mg (0.015 mmol) of ethylene(2-Me-4,5,6,7-tetrahydro-1-indenyl) 2 zirconium dichloride were employed.
- the metallocene activity was 72.5 kg of PP/g of metallocene ⁇ h.
- Example 4 was repeated, but 28.1 mg (0.062 mmol) of the metallocene were used and the polymerization temperature was 50° C.
- the metallocene activity was 28.5 kg of PP/g of metallocene ⁇ h.
- Example 4 was repeated, but 50 mg (0.110 mmol) of the metallocene were used and the polymerization temperature was 30° C.
- the metallocene activity was 10.9 kg of PP/g of metallocene ⁇ h.
- Examples 4 to 6 were repeated, but the metallocenes ethylene(1-indenyl) 2 zirconium dichloride (metallocene 4) and ethylene(2-Me-1-indenyl) 2 zirconium dichloride (metallocene 5) were used.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Transition And Organic Metals Composition Catalysts For Addition Polymerization (AREA)
Abstract
in which, preferably, M1 is Zr or Hf, R1 and R2 are alkyl or halogen, R3 and R4 are hydrogen, R5 and R6 are alkyl or haloalkyl, —(CR8R9)m—R7—(CR8R9)n— is a single- or multi-membered chain in which R7 may also be a (substituted) hetero atom, m+n is zero or 1, and R10 is hydrogen, form, together with aluminoxanes as cocatalysts, a very effective catalyst system for the preparation of polyolefins of high stereospecificity and high melting point.
Description
- M1 is a metal from group IVb, Vb or VIb of the Periodic Table,
- R1 and R2 are identical or different and are a hydrogen atom, a C1-C10-alkyl group, a C1-C10-alkoxy group, a C6-C10-aryl group, a C6-C10-aryloxy group, a C2-C10-alkenyl group, a C7-C40-arylalkyl group, a C7-C40-alkylaryl group, a C8-C40-arylalkenyl group or a halogen atom,
- R3 and R4 are identical or different and are a hydrogen atom, a halogen atom, a C1-C10-alkyl group, which may be halogenated, a C6-C10-aryl group, an —NR2 15, —SR15, —OSiR3 15, —SiR3 15 or —PR2 15 radical in which R15 is a halogen atom, a C1-C10-alkyl group or a C6-C10-aryl group,
- R5 and R6 are identical or different and are as defined for R3 and R4, with the proviso that R5 and R6 are not hydrogen,
- R7 is
where - R11, R12 and R13 are identical or different and are a hydrogen atom, a halogen atom, a C1-C10-alkyl group, a C1-C10-fluoroalkyl group, a C6-C10-aryl group, a C6-C10-fluoroaryl group, a C1-C10-alkoxy group, a C2-C10-alkenyl group, a C7-C40-arylalkyl group, a C8-C40-arylalkenyl group or a C7-C40-alkylaryl group, or R11 and R12 or R11 and R13, in each case with the atoms connecting them, form a ring,
- M2 is silicon, germanium or tin,
- R8 and R9 are identical or different and are as defined for R11,
- m and n are identical or different and are zero, 1 or 2, m plus n being zero, 1 or 2, and,
- the radicals R10 are identical or different and are as defined for R11, R12 and R13.
- Alkyl is straight-chain or branched alkyl. Halogen (halogenated) is fluorine, chlorine, bromine or iodine, preferably fluorine or chlorine.
- R1 and R2 are identical or different and are a hydrogen atom, a C1-C10-, preferably C1-C3-alkyl group, a C1-C10-, preferably C1-C3-alkoxy group, a C6-C10-, preferably C6-C8-aryl group, a C6-C10-, preferably C6-C8-aryloxy group, a C2-C10-, preferably C2-C4-alkenyl group, a C7-C40-, preferably C7-C10-arylalkyl group, a C7-C40-, preferably C7-C12-alkylaryl group, a C8-C40-, preferably C8-C12-arylalkenyl group or a halogen atom, preferably chlorine.
- R3 and R4 are identical or different and are a hydrogen atom, a halogen atom, preferably a fluorine, chlorine or bromine atom, a C1-C10-, preferably C1-C4-alkyl group, which may be halogenated, a C6-C10-, preferably C6-C8-aryl group, an —NR2 15, —SR15, —O—SiR3 15, —SiR3 15 or —PR2 15 radical in which R15 is a halogen atom, preferably a chlorine atom, or a C1-C10-, preferably C1-C3-alkyl group or a C6-C10-, preferably C6-C8-aryl group. R3 and R4 are particularly preferably hydrogen.
- R5 and R6 are identical or different, preferably identical, and are as defined for R3 and R4, with the proviso that R5 and R6 cannot be hydrogen. R5 and R6 are preferably (C1-C4)-alkyl, which may be halogenated, such as methyl, ethyl, propyl, isopropyl, butyl, isobutyl or trifluoromethyl, in particular methyl.
- R7 is
═BR11, ═AlR11, —Ge—, —Sn—, —O—, —S—, ═SO, ═SO2, ═NR11, ═CO, ═PR11 or ═P(O)R11, where R11, R12 and R13 are identical or different and are a hydrogen atom, a halogen atom, a C1-C10-, preferably C1-C4-alkyl group, in particular a methyl group, a C1-C10-fluoroalkyl group, preferably a CF3 group, a C6-C10-, preferably C6-C8-aryl group, a C6-C10-fluoroaryl group, preferably a pentafluorophenyl group, a C1-C10-, preferably C1-C4-alkoxy group, in particular a methoxy group, a C2-C10-, preferably C2-C4-alkenyl group, a C7-C40-, preferably C7-C10-arylalkyl group, a C8-C40-, preferably C8-C12-arylalkenyl group or a C7-C40-, preferably C7-C12-alkylaryl group, or R11 and R12 or R11 and R13, in each case together with the atoms connecting them, form a ring. - M2 is silicon, germanium or tin, preferably silicon or germanium.
- R7 is preferably ═CR11R12, ═SiR11R12, ═GeR11R12, —O—, —S—, ═SO, ═PR11 or ═P(O)R11.
- R8 and R9 are identical or different and are as define as for R11.
- m and n are identical or different and are zero, 1 or 2, preferably zero or 1, where m plus n is zero, 1 or 2, preferably zero or 1.
radical, n plus m is zero or 1, and R10 is hydrogen; in particular the compounds I listed in the working examples.
- a) reacting a compound of the formula II
in which R3-R10, m and n are defined in the formula I and M3 is an alkali metal, preferably lithium, with a compound of the formula III
M1X4 (III)
in which M1 is a defined in the formula I, and X is a halogen atom, preferably chlorine, and catalytically hydrogenating the reaction product, or - b) reacting a compound of the formula IIa
with a compound of the formula III
M1X4 (III)
in which all the substituents are as defined under a), and, if desired, derivatizing the reaction product obtained under a) or b).
for the linear type and/or of the formula (V)
for the cyclic type, where, in the formulae (IV) and (V), the radicals R may be identical or different and are a C1-C6-alkyl group, a C6-C18-aryl group or hydrogen, and p is an integer from 2 to 50, preferably from 10 to 35.
| VN = | viscosity number in cm3/g | ||
| determined | |||
| Mw = | weight average molecular weight | by gel perme- | |
| Mw/Mn = | molecular weight dispersity | ation chroma- | |
| tography | |||
| II = | isotactic index (II = mm + 1/8 ms), determined by |
| 13C-NMR spectroscopy | |
| niso = | length of the isotactic blocks (in propylene units) |
| (niso = 1 + 2 mm/mr), determined by 13C-NMR | |
| spectroscopy | |
| Comp. | Polym. | M.p. | ΔHmeh | ||
| Ex. | Metallocene | temp. [° C.] | niso | [° C. ] | [J/g] |
| A | 1 | 70 | 38 | 145 | 86.6 |
| B | 1 | 50 | 48 | 148 | 88.1 |
| C | 1 | 30 | 48 | 152 | 90.2 |
| D | 2 | 70 | 34 | 141 | — |
| E | 2 | 50 | 38 | 143 | — |
| F | 3 | 70 | 32 | 140 | — |
| G | 3 | 50 | 34 | 142 | — |
| H | 3 | 30 | 37 | 145 | — |
| Comp. | Polym. | M.p. | ΔHmeh | ||
| Ex. | Metallocene | temp. [° C.] | niso | [° C. ] | [J/g] |
| I | 4 | 70 | 23 | 132 | 64.9 |
| K | 4 | 50 | 30 | 138 | 78.1 |
| L | 4 | 30 | 29 | 137 | 78.6 |
| M | 5 | 70 | 25 | 134 | 77.0 |
| N | 5 | 50 | 30 | 138 | 78.9 |
| O | 5 | 30 | 32 | 138 | 78.6 |
Claims (23)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US08/895,950 USRE39561E1 (en) | 1990-11-12 | 1997-07-17 | Metallocenes containing ligands of 2-substituted indenyl derivatives, process for their preparation, and their use as catalysts |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE4035884 | 1990-11-12 | ||
| US07/789,361 US5276208A (en) | 1990-11-12 | 1991-11-08 | Metallocenes containing ligands of 2-substituted idenyl derivatives, process for their preparation, and their use as catalysts |
| US08/324,260 USRE37208E1 (en) | 1990-11-12 | 1994-10-17 | Polyolefins prepared with metallocene catalysts having 2-substituted indenyl type ligands |
| US08/895,950 USRE39561E1 (en) | 1990-11-12 | 1997-07-17 | Metallocenes containing ligands of 2-substituted indenyl derivatives, process for their preparation, and their use as catalysts |
Related Parent Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US07/789,361 Reissue US5276208A (en) | 1990-11-12 | 1991-11-08 | Metallocenes containing ligands of 2-substituted idenyl derivatives, process for their preparation, and their use as catalysts |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| USRE39561E1 true USRE39561E1 (en) | 2007-04-10 |
Family
ID=27201879
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US08/895,909 Expired - Lifetime USRE39532E1 (en) | 1990-11-12 | 1997-07-17 | Metallocenes containing ligands of 2-substituted indenyl derivatives, process for their preparation, and their use as catalysts |
| US08/895,950 Expired - Lifetime USRE39561E1 (en) | 1990-11-12 | 1997-07-17 | Metallocenes containing ligands of 2-substituted indenyl derivatives, process for their preparation, and their use as catalysts |
Family Applications Before (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US08/895,909 Expired - Lifetime USRE39532E1 (en) | 1990-11-12 | 1997-07-17 | Metallocenes containing ligands of 2-substituted indenyl derivatives, process for their preparation, and their use as catalysts |
Country Status (1)
| Country | Link |
|---|---|
| US (2) | USRE39532E1 (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20080200708A1 (en) * | 2002-10-25 | 2008-08-21 | Basell Polyolefine Gmbh | Preparation of Partially Hydrogenated Rac-Ansa-Metallocene Complexes |
Citations (29)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE284708C (en) | ||||
| EP0129368A1 (en) | 1983-06-06 | 1984-12-27 | Exxon Research And Engineering Company | Process and catalyst for polyolefin density and molecular weight control |
| EP0185918A2 (en) | 1984-11-27 | 1986-07-02 | Hoechst Aktiengesellschaft | Process for preparing polyolefins |
| JPS62121707A (en) * | 1985-11-22 | 1987-06-03 | Mitsui Petrochem Ind Ltd | Production of alpha-olefin random copolymer |
| DE3726067A1 (en) | 1987-08-06 | 1989-02-16 | Hoechst Ag | METHOD FOR PRODUCING 1-OLEFIN POLYMERS |
| EP0316155A2 (en) | 1987-11-09 | 1989-05-17 | Chisso Corporation | A transition-metal compound having a substituted, bridged bis-cyclopentadienyl ligand |
| EP0320762A2 (en) | 1987-12-18 | 1989-06-21 | Hoechst Aktiengesellschaft | Process for the preparation of a chiral, stereorigid metallocen compound |
| US4871705A (en) * | 1988-06-16 | 1989-10-03 | Exxon Chemical Patents Inc. | Process for production of a high molecular weight ethylene a-olefin elastomer with a metallocene alumoxane catalyst |
| EP0336128A2 (en) | 1988-03-12 | 1989-10-11 | Hoechst Aktiengesellschaft | Process for preparing an alpha-olefin polymer |
| EP0344887A2 (en) | 1988-03-21 | 1989-12-06 | Exxon Chemical Patents Inc. | Silicon bridged transition metal compounds |
| EP0347129A1 (en) | 1988-06-16 | 1989-12-20 | Exxon Chemical Patents Inc. | Process for production of high molecular weight EPDM elastomers using a metallocene-alumoxane catalyst system |
| US4892851A (en) | 1988-07-15 | 1990-01-09 | Fina Technology, Inc. | Process and catalyst for producing syndiotactic polyolefins |
| DE3826075A1 (en) | 1988-07-30 | 1990-02-01 | Hoechst Ag | 1-OLEFIN ISO BLOCK POLYMER AND METHOD FOR THE PRODUCTION THEREOF |
| EP0355289A1 (en) | 1988-07-01 | 1990-02-28 | ATOCHEM NORTH AMERICA, INC. (a Pennsylvania corp.) | Accelerometer |
| EP0366290A2 (en) | 1988-10-24 | 1990-05-02 | Chisso Corporation | Process for producing olefin polymers |
| EP0407870A2 (en) | 1989-07-08 | 1991-01-16 | Hoechst Aktiengesellschaft | Process for preparing cycloolefin polymers |
| EP0426643A1 (en) | 1989-10-30 | 1991-05-08 | Fina Research S.A. | Process for the preparation of metallocenes |
| US5017714A (en) | 1988-03-21 | 1991-05-21 | Exxon Chemical Patents Inc. | Silicon-bridged transition metal compounds |
| EP0433990A2 (en) | 1989-12-21 | 1991-06-26 | Hoechst Aktiengesellschaft | Process for preparing polypropylene molding compositions |
| EP0442725A2 (en) | 1990-02-13 | 1991-08-21 | Mitsui Petrochemical Industries, Ltd. | Olefin polymerization solid catalysts and process for the polymerization of olefins |
| EP0485823A1 (en) | 1990-11-12 | 1992-05-20 | Hoechst Aktiengesellschaft | 2-Substituted bisindenyl-metallocenes, process for their preparation and their use as catalysts for the polymerization of olefins |
| EP0485821A1 (en) | 1990-11-12 | 1992-05-20 | Hoechst Aktiengesellschaft | Metallocenes with 2-substituted indenyl-derivates as ligands, process for their preparation and their use as catalysts |
| EP0500005A1 (en) | 1991-02-18 | 1992-08-26 | Hoechst Aktiengesellschaft | Process for the preparation of substituted indenes |
| EP0529908A1 (en) | 1991-08-20 | 1993-03-03 | Mitsubishi Chemical Corporation | Catalyst useful for the polymerization of olefin |
| US5243001A (en) | 1990-11-12 | 1993-09-07 | Hoechst Aktiengesellschaft | Process for the preparation of a high molecular weight olefin polymer |
| US5278264A (en) | 1991-08-26 | 1994-01-11 | Hoechst Ag | Process for the preparation of an olefin polymer |
| US5296434A (en) | 1991-06-18 | 1994-03-22 | Basf Aktiengesellschaft | Soluble catalyst systems for the preparation of polyalk-1-enes having high molecular weights |
| US5324800A (en) | 1983-06-06 | 1994-06-28 | Exxon Chemical Patents Inc. | Process and catalyst for polyolefin density and molecular weight control |
| US5455365A (en) | 1992-08-03 | 1995-10-03 | Hoechst Ag | Process for the preparation of an olefin polymer using metallocenes containing specifically substituted indenyl ligands |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3192276A (en) * | 1962-09-10 | 1965-06-29 | Union Carbide Corp | Alkylation of indene |
-
1997
- 1997-07-17 US US08/895,909 patent/USRE39532E1/en not_active Expired - Lifetime
- 1997-07-17 US US08/895,950 patent/USRE39561E1/en not_active Expired - Lifetime
Patent Citations (39)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE284708C (en) | ||||
| EP0129368A1 (en) | 1983-06-06 | 1984-12-27 | Exxon Research And Engineering Company | Process and catalyst for polyolefin density and molecular weight control |
| US5324800A (en) | 1983-06-06 | 1994-06-28 | Exxon Chemical Patents Inc. | Process and catalyst for polyolefin density and molecular weight control |
| EP0185918A2 (en) | 1984-11-27 | 1986-07-02 | Hoechst Aktiengesellschaft | Process for preparing polyolefins |
| US4769510A (en) * | 1984-11-27 | 1988-09-06 | Hoechst Aktiengesellschaft | Process for the preparation of polyolefins |
| JPS62121707A (en) * | 1985-11-22 | 1987-06-03 | Mitsui Petrochem Ind Ltd | Production of alpha-olefin random copolymer |
| DE3726067A1 (en) | 1987-08-06 | 1989-02-16 | Hoechst Ag | METHOD FOR PRODUCING 1-OLEFIN POLYMERS |
| US4931417A (en) | 1987-11-09 | 1990-06-05 | Chisso Corporation | Transition-metal compound having a bis-substituted-cyclopentadienyl ligand of bridged structure |
| EP0316155A2 (en) | 1987-11-09 | 1989-05-17 | Chisso Corporation | A transition-metal compound having a substituted, bridged bis-cyclopentadienyl ligand |
| EP0320762A2 (en) | 1987-12-18 | 1989-06-21 | Hoechst Aktiengesellschaft | Process for the preparation of a chiral, stereorigid metallocen compound |
| US5103030A (en) | 1987-12-18 | 1992-04-07 | Hoechst Aktiengesellschaft | Process for the preparation of a chiral stereorigid metallocene |
| EP0336128A2 (en) | 1988-03-12 | 1989-10-11 | Hoechst Aktiengesellschaft | Process for preparing an alpha-olefin polymer |
| EP0344887A2 (en) | 1988-03-21 | 1989-12-06 | Exxon Chemical Patents Inc. | Silicon bridged transition metal compounds |
| US5017714A (en) | 1988-03-21 | 1991-05-21 | Exxon Chemical Patents Inc. | Silicon-bridged transition metal compounds |
| EP0347129A1 (en) | 1988-06-16 | 1989-12-20 | Exxon Chemical Patents Inc. | Process for production of high molecular weight EPDM elastomers using a metallocene-alumoxane catalyst system |
| US4871705A (en) * | 1988-06-16 | 1989-10-03 | Exxon Chemical Patents Inc. | Process for production of a high molecular weight ethylene a-olefin elastomer with a metallocene alumoxane catalyst |
| EP0355289A1 (en) | 1988-07-01 | 1990-02-28 | ATOCHEM NORTH AMERICA, INC. (a Pennsylvania corp.) | Accelerometer |
| EP0351392A2 (en) | 1988-07-15 | 1990-01-17 | Fina Technology, Inc. | Process and catalyst for producing syndiotactic polyolefins |
| US4892851A (en) | 1988-07-15 | 1990-01-09 | Fina Technology, Inc. | Process and catalyst for producing syndiotactic polyolefins |
| DE3826075A1 (en) | 1988-07-30 | 1990-02-01 | Hoechst Ag | 1-OLEFIN ISO BLOCK POLYMER AND METHOD FOR THE PRODUCTION THEREOF |
| EP0366290A2 (en) | 1988-10-24 | 1990-05-02 | Chisso Corporation | Process for producing olefin polymers |
| EP0407870A2 (en) | 1989-07-08 | 1991-01-16 | Hoechst Aktiengesellschaft | Process for preparing cycloolefin polymers |
| US5087677A (en) | 1989-07-08 | 1992-02-11 | Hoechst Aktiengesellschaft | Process for the preparation of cycloolefin polymers |
| EP0426643A1 (en) | 1989-10-30 | 1991-05-08 | Fina Research S.A. | Process for the preparation of metallocenes |
| EP0433990A2 (en) | 1989-12-21 | 1991-06-26 | Hoechst Aktiengesellschaft | Process for preparing polypropylene molding compositions |
| EP0442725A2 (en) | 1990-02-13 | 1991-08-21 | Mitsui Petrochemical Industries, Ltd. | Olefin polymerization solid catalysts and process for the polymerization of olefins |
| EP0485821A1 (en) | 1990-11-12 | 1992-05-20 | Hoechst Aktiengesellschaft | Metallocenes with 2-substituted indenyl-derivates as ligands, process for their preparation and their use as catalysts |
| US5145819A (en) | 1990-11-12 | 1992-09-08 | Hoechst Aktiengesellschaft | 2-substituted disindenylmetallocenes, process for their preparation, and their use as catalysts in the polymerization of olefins |
| US5243001A (en) | 1990-11-12 | 1993-09-07 | Hoechst Aktiengesellschaft | Process for the preparation of a high molecular weight olefin polymer |
| US5276208A (en) | 1990-11-12 | 1994-01-04 | Hoechst Aktiengesellschaft | Metallocenes containing ligands of 2-substituted idenyl derivatives, process for their preparation, and their use as catalysts |
| EP0485823A1 (en) | 1990-11-12 | 1992-05-20 | Hoechst Aktiengesellschaft | 2-Substituted bisindenyl-metallocenes, process for their preparation and their use as catalysts for the polymerization of olefins |
| EP0500005A1 (en) | 1991-02-18 | 1992-08-26 | Hoechst Aktiengesellschaft | Process for the preparation of substituted indenes |
| US5296434A (en) | 1991-06-18 | 1994-03-22 | Basf Aktiengesellschaft | Soluble catalyst systems for the preparation of polyalk-1-enes having high molecular weights |
| EP0529908A1 (en) | 1991-08-20 | 1993-03-03 | Mitsubishi Chemical Corporation | Catalyst useful for the polymerization of olefin |
| US5561093A (en) | 1991-08-20 | 1996-10-01 | Mitsubishi Chemical Corporation | Catalyst useful for the polymerization of olefins |
| US6143911A (en) | 1991-08-20 | 2000-11-07 | Mitsubishi Petrochemical Company Limited | Catalyst useful for the polymerization of olefins |
| US5278264A (en) | 1991-08-26 | 1994-01-11 | Hoechst Ag | Process for the preparation of an olefin polymer |
| US5329033A (en) | 1991-08-26 | 1994-07-12 | Hoechst Aktiengesellschaft | Process for the preparation of an olefin polymer |
| US5455365A (en) | 1992-08-03 | 1995-10-03 | Hoechst Ag | Process for the preparation of an olefin polymer using metallocenes containing specifically substituted indenyl ligands |
Non-Patent Citations (20)
| Title |
|---|
| Adcock et al., Austr. J. Chem., vol. 29, "Substituent Effects by <SUP>19</SUP>F Nuclear Magnetic Resonance: Polar and pi-Electron Effects", pp. 2571-2581. |
| BULLETIN DE LA SOCIETE CHIMIQUE DE FRANCE, "Etude de monomeres halogenes et de leur polymerisation cationique", No. 11, pp. 3092-3095, (1973). |
| Chemical Abstracts 90:567 103691p; (1978). |
| Criegee et al., Chem. Ber., vol. 94, "Uber den Nickelkomplex C<SUB>18</SUB>H<SUB>22</SUB>Ni und den daraus gewonnenen Kohlenwasserstoff C<SUB>13</SUB>H<SUB>18</SUB>", pp. 3461-3468 (1964). |
| Esperas, S., ACTA CHEMICA SCANDINAVICA, "The Crystal and Molecular Structure of Cyano(methylisocyanide)gold(I)", A 30, No. 7, pp. 527-530 (1976). |
| Ewen, J.A., et al, J. Am. Chem. Soc., Crystal Structures and Sterospecific Propylene Polymerizations with Chiral Hafnium Metallocene Catalysts, vol. 109, 1987, pp. 6544-6545. |
| Hart et al., NOTES, J. Am. Chem. Soc., vol. 72, "Acylation-Akylation Studies", pp. 3286-3287 (1950). |
| J. ORG. CHEM., "Friedel-Crafts Chemistry, A Mechanistic Study of the Reaction of 3-Chloro-4'-fluoro-2-methylpropiophenone with AlCl<SUB>3</SUB>and AlCl<SUB>3</SUB>-CH<SUB>3</SUB>NO<SUB>2</SUB>", vol. 43, No. 16, pp. 3126-3131 (1978). |
| J. ORG. CHEM., "Friedel-Crafts Reactions of Ethyl Cyclopropanecarboxylate", vol. 46, pp. 3758-3760 (1981). |
| JACS (1967) 89(23) 5868-5876, Nov. 1967. * |
| Katz, Thomas J., J. Am. Chem. Soc., "Asymmetric Synthesis of Helical Metallocenes", vol. 108, 1986, pp. 179-181. |
| Marechal et al., Bull. Soc. Chim. Fr. 6, "Homopolymerisation cationlique des dimethyl-4,7,dimethyl-4,6 et dimethyl-5,6 indenes", No. 348, pp. 1981-2039, (1969). |
| Miyamota, T.K., et al., CHEMISTRY LETTERS, The Chemical Society of Japan, "A Bulky Ligand and its Organometallic Compound: Synthesis of Heptamethylidene and a Ferrocene-Type Complex, Fe(n<SUP>5</SUP>-C<SUB>9</SUB>Me<SUB>7</SUB>)<SUB>2</SUB>", pp. 729-730 (1981). |
| Piccoliovazzi, N. et al., ORGANOMETALLICS, "Electronic Effects in Homogeneous Indenylzirconium Ziegler-Natta Catalysts", vol. 9, pp. 3098-3105 (1990). |
| Röll, V.W., et al., ANGEW. CHEM., "Stereo- und Regioselektivitat von chiralen, alkylsubstituierten ansa-Zirconocen-Katalysatoren bei der Methylalumoxan-aktivierten Propen-Polymerization", vol. 102, No. 3, pp. 339-341 (1990). |
| Soga, K. et al., MACROMOLECULES, "Perfect Conversion of Aspecific Sites into Isopecific Sites in Ziegler-Natta Catalysts", vol. 22, pp. 3824-3826 (1989). |
| Spaleci et al., New J. Chem., "Stereorigid Metallocenes: Correlations Between Structure and Behavior in Homopolymerizations of Propylene", vol. 14, pp. 499-503, 1990. * |
| Spaleck et al., NEW J. CHEM., "Stereorigid Metallocenes: Correlations Between Structure and Behaviour in Homopolymerizations of Propylene", vol. 14, pp. 499-503 (1990). |
| Winter v. Fujita, 53 U.S.P.Q.2d 1234 (Bd of Appeals 1999). |
| Winter v. Fujita, 53 U.S.P.Q.2d 1478 (Bd of Appeals 2000). |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20080200708A1 (en) * | 2002-10-25 | 2008-08-21 | Basell Polyolefine Gmbh | Preparation of Partially Hydrogenated Rac-Ansa-Metallocene Complexes |
| US7619106B2 (en) * | 2002-10-25 | 2009-11-17 | Basell Polyolefine Gmbh | Preparation of partially hydrogenated rac-ansa-metallocene complexes |
Also Published As
| Publication number | Publication date |
|---|---|
| USRE39532E1 (en) | 2007-03-27 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| USRE37208E1 (en) | Polyolefins prepared with metallocene catalysts having 2-substituted indenyl type ligands | |
| USRE37384E1 (en) | 2-substituted disindenylmetallocenes, process for their preparation, and their use as catalysts in the polymerization of olefins | |
| US5239022A (en) | Process for the preparation of a syndiotactic polyolefin | |
| US5243001A (en) | Process for the preparation of a high molecular weight olefin polymer | |
| JP3143174B2 (en) | Method for producing olefin polymer | |
| AU640286B2 (en) | Process for the preparation of a high molecular weight olefin polymer | |
| JP3234876B2 (en) | Bridged chiral metallocenes, process for their preparation and their use as catalysts | |
| JP3394997B2 (en) | Process for producing olefin polymers using metallocenes having specifically substituted indenyl ligands | |
| JP3371118B2 (en) | Metallocenes having benzene-fused indenyl derivatives as ligands, their preparation and their use as catalysts | |
| KR100261362B1 (en) | Process for preparing high molecular weight polyolefins | |
| US5504232A (en) | Process for the preparation of an olefin polymer using specific metallocenes | |
| US6268518B1 (en) | Metallocene compounds and their use in catalysts for the polymerization of olefins | |
| US5268495A (en) | Metallocenes having bicyclic cyclopentadiene derivatives as ligands, processes for their preparation and their use as catalysts | |
| US5830821A (en) | Process for olefin preparation using metallocenes having benzo-fused indenyl derivatives as ligands | |
| US5932669A (en) | Metallocenes having benzo-fused indenyl derivatives as ligands, processes for their preparation and their use as catalysts | |
| USRE39561E1 (en) | Metallocenes containing ligands of 2-substituted indenyl derivatives, process for their preparation, and their use as catalysts | |
| US20110306741A1 (en) | Synthesis of substituted tetrahydroindenyl complexes | |
| USRE37573E1 (en) | Process for the preparation of an olefin polymer using metallocenes containing specifically substituted indenyl ligands | |
| JP2001519436A (en) | Ferrocenyl-substituted bridged metallocenes used in olefin polymerization |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AS | Assignment |
Owner name: CITIBANK, N.A., AS COLLATERAL AGENT,NEW YORK Free format text: GRANT OF SECURITY INTEREST IN UNITED STATES PATENTS AND PATENT APPLICATIONS;ASSIGNORS:BASELL POLYOLEFINE GMBH;ARCO CHEMICAL TECHNOLOGY L.P.;ARCO CHEMICAL TECHNOLOGY, INC.;AND OTHERS;REEL/FRAME:020704/0562 Effective date: 20071220 Owner name: CITIBANK, N.A., AS COLLATERAL AGENT, NEW YORK Free format text: GRANT OF SECURITY INTEREST IN UNITED STATES PATENTS AND PATENT APPLICATIONS;ASSIGNORS:BASELL POLYOLEFINE GMBH;ARCO CHEMICAL TECHNOLOGY L.P.;ARCO CHEMICAL TECHNOLOGY, INC.;AND OTHERS;REEL/FRAME:020704/0562 Effective date: 20071220 |
|
| AS | Assignment |
Owner name: CITIBANK, N.A., AS COLLATERAL AGENT,NEW YORK Free format text: SECURITY AGREEMENT;ASSIGNORS:BASELL POLYOLEFINE GMBH;ARCO CHEMICAL TECHNOLOGY L.P.;ARCO CHEMICAL TECHNOLOGY, INC.;AND OTHERS;REEL/FRAME:021354/0708 Effective date: 20071220 Owner name: CITIBANK, N.A., AS COLLATERAL AGENT, NEW YORK Free format text: SECURITY AGREEMENT;ASSIGNORS:BASELL POLYOLEFINE GMBH;ARCO CHEMICAL TECHNOLOGY L.P.;ARCO CHEMICAL TECHNOLOGY, INC.;AND OTHERS;REEL/FRAME:021354/0708 Effective date: 20071220 |
|
| CC | Certificate of correction | ||
| AS | Assignment |
Owner name: EQUISTAR CHEMICALS, LP, TEXAS Free format text: RELEASE BY SECURED PARTY;ASSIGNOR:CITIBANK, N.A., AS COLLATERAL AGENT;REEL/FRAME:024337/0856 Effective date: 20100430 Owner name: LYONDELL CHEMICAL TECHNOLOGY, L.P., DELAWARE Free format text: RELEASE BY SECURED PARTY;ASSIGNOR:CITIBANK, N.A., AS COLLATERAL AGENT;REEL/FRAME:024337/0856 Effective date: 20100430 Owner name: EQUISTAR CHEMICALS, LP, TEXAS Free format text: RELEASE BY SECURED PARTY;ASSIGNOR:CITIBANK, N.A., AS COLLATERAL AGENT;REEL/FRAME:024337/0705 Effective date: 20100430 Owner name: LYONDELL CHEMICAL TECHNOLOGY, L.P., DELAWARE Free format text: RELEASE BY SECURED PARTY;ASSIGNOR:CITIBANK, N.A., AS COLLATERAL AGENT;REEL/FRAME:024337/0705 Effective date: 20100430 Owner name: EQUISTAR CHEMICALS, LP,TEXAS Free format text: RELEASE BY SECURED PARTY;ASSIGNOR:CITIBANK, N.A., AS COLLATERAL AGENT;REEL/FRAME:024337/0856 Effective date: 20100430 Owner name: LYONDELL CHEMICAL TECHNOLOGY, L.P.,DELAWARE Free format text: RELEASE BY SECURED PARTY;ASSIGNOR:CITIBANK, N.A., AS COLLATERAL AGENT;REEL/FRAME:024337/0856 Effective date: 20100430 Owner name: EQUISTAR CHEMICALS, LP,TEXAS Free format text: RELEASE BY SECURED PARTY;ASSIGNOR:CITIBANK, N.A., AS COLLATERAL AGENT;REEL/FRAME:024337/0705 Effective date: 20100430 Owner name: LYONDELL CHEMICAL TECHNOLOGY, L.P.,DELAWARE Free format text: RELEASE BY SECURED PARTY;ASSIGNOR:CITIBANK, N.A., AS COLLATERAL AGENT;REEL/FRAME:024337/0705 Effective date: 20100430 |