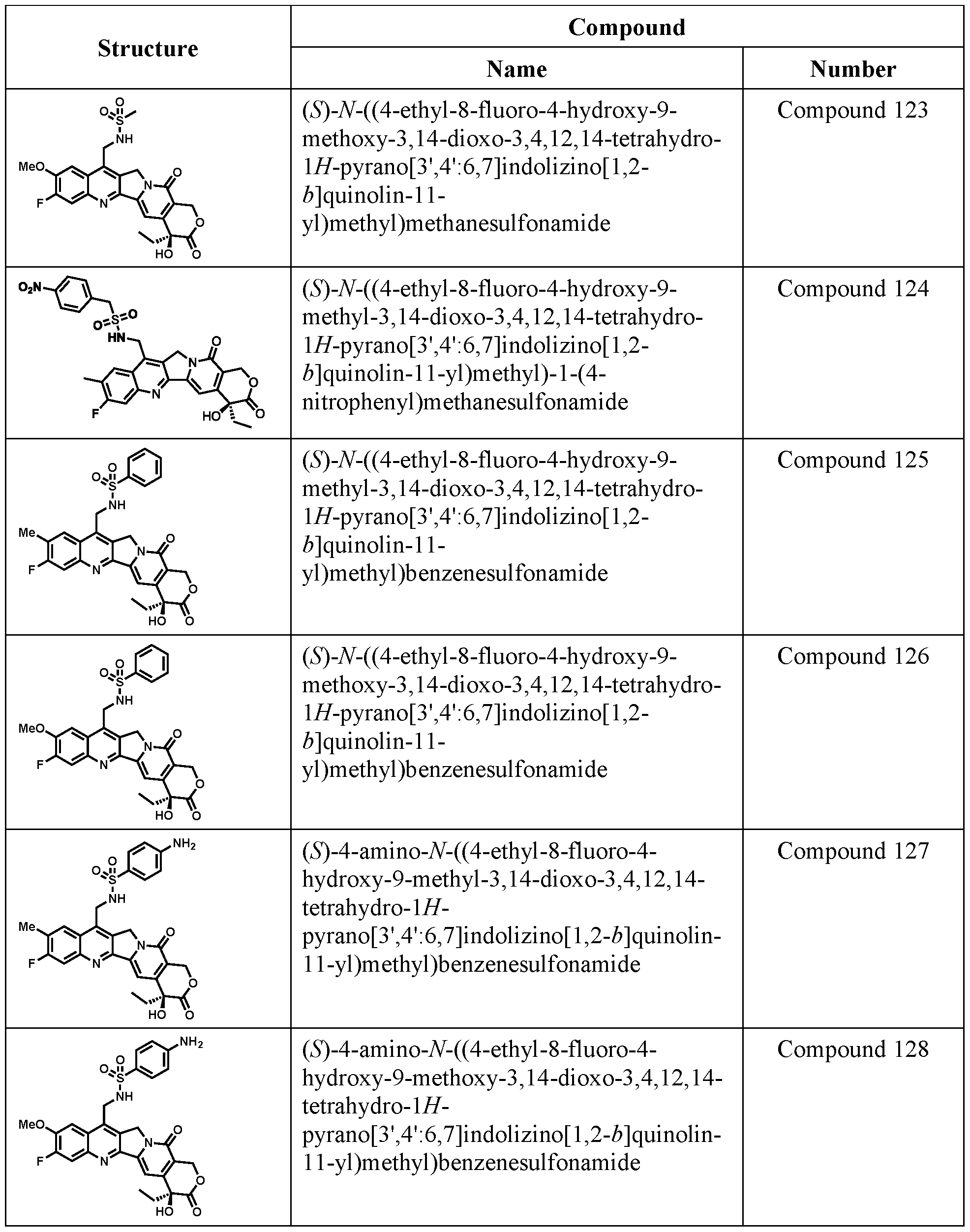
WO2022246576A1 - Camptothecin analogues, conjugates and methods of use - Google Patents
Camptothecin analogues, conjugates and methods of use Download PDFInfo
- Publication number
- WO2022246576A1 WO2022246576A1 PCT/CA2022/050864 CA2022050864W WO2022246576A1 WO 2022246576 A1 WO2022246576 A1 WO 2022246576A1 CA 2022050864 W CA2022050864 W CA 2022050864W WO 2022246576 A1 WO2022246576 A1 WO 2022246576A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- alkyl
- aryl
- cycloalkyl
- heteroaryl
- formula
- Prior art date
Links
- 238000000034 method Methods 0.000 title claims description 179
- VSJKWCGYPAHWDS-FQEVSTJZSA-N camptothecin Chemical class C1=CC=C2C=C(CN3C4=CC5=C(C3=O)COC(=O)[C@]5(O)CC)C4=NC2=C1 VSJKWCGYPAHWDS-FQEVSTJZSA-N 0.000 title abstract description 72
- 206010028980 Neoplasm Diseases 0.000 claims abstract description 63
- 201000011510 cancer Diseases 0.000 claims abstract description 39
- 239000003814 drug Substances 0.000 claims abstract description 17
- 208000023275 Autoimmune disease Diseases 0.000 claims abstract description 12
- 208000036142 Viral infection Diseases 0.000 claims abstract description 11
- 230000009385 viral infection Effects 0.000 claims abstract description 11
- 150000001875 compounds Chemical class 0.000 claims description 869
- 125000004169 (C1-C6) alkyl group Chemical group 0.000 claims description 421
- -1 -CHF2 Chemical group 0.000 claims description 143
- 125000000592 heterocycloalkyl group Chemical group 0.000 claims description 129
- 230000008685 targeting Effects 0.000 claims description 70
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims description 65
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 claims description 55
- 229910052717 sulfur Inorganic materials 0.000 claims description 52
- 125000000876 trifluoromethoxy group Chemical group FC(F)(F)O* 0.000 claims description 51
- 125000002023 trifluoromethyl group Chemical group FC(F)(F)* 0.000 claims description 50
- 229910052760 oxygen Inorganic materials 0.000 claims description 44
- 229910052736 halogen Inorganic materials 0.000 claims description 36
- 125000003118 aryl group Chemical group 0.000 claims description 34
- 125000001424 substituent group Chemical group 0.000 claims description 34
- 125000001309 chloro group Chemical group Cl* 0.000 claims description 33
- 125000000217 alkyl group Chemical group 0.000 claims description 32
- 150000002367 halogens Chemical class 0.000 claims description 28
- 125000000753 cycloalkyl group Chemical group 0.000 claims description 22
- 125000001072 heteroaryl group Chemical group 0.000 claims description 22
- 150000003839 salts Chemical class 0.000 claims description 19
- 239000008194 pharmaceutical composition Substances 0.000 claims description 18
- 125000004433 nitrogen atom Chemical group N* 0.000 claims description 17
- 239000000427 antigen Substances 0.000 claims description 14
- 108091007433 antigens Proteins 0.000 claims description 14
- 102000036639 antigens Human genes 0.000 claims description 14
- 125000004093 cyano group Chemical group *C#N 0.000 claims description 12
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 claims description 12
- 108091005804 Peptidases Proteins 0.000 claims description 11
- 125000002252 acyl group Chemical group 0.000 claims description 11
- 125000004423 acyloxy group Chemical group 0.000 claims description 11
- 125000003545 alkoxy group Chemical group 0.000 claims description 11
- 125000004414 alkyl thio group Chemical group 0.000 claims description 11
- 125000003368 amide group Chemical group 0.000 claims description 11
- 125000000446 sulfanediyl group Chemical group *S* 0.000 claims description 11
- 125000005420 sulfonamido group Chemical group S(=O)(=O)(N*)* 0.000 claims description 11
- 125000000472 sulfonyl group Chemical group *S(*)(=O)=O 0.000 claims description 11
- 239000004365 Protease Substances 0.000 claims description 10
- 125000000852 azido group Chemical group *N=[N+]=[N-] 0.000 claims description 10
- 230000002147 killing effect Effects 0.000 claims description 10
- 230000002401 inhibitory effect Effects 0.000 claims description 9
- 239000003085 diluting agent Substances 0.000 claims description 8
- 238000004519 manufacturing process Methods 0.000 claims description 8
- 102000008394 Immunoglobulin Fragments Human genes 0.000 claims description 6
- 108010021625 Immunoglobulin Fragments Proteins 0.000 claims description 6
- 230000035755 proliferation Effects 0.000 claims description 6
- 239000003937 drug carrier Substances 0.000 claims description 5
- OZXMCEGJEIIDQT-DEOSSOPVSA-N (19S)-7-amino-10-butyl-19-ethyl-19-hydroxy-17-oxa-3,13-diazapentacyclo[11.8.0.02,11.04,9.015,20]henicosa-1(21),2,4(9),5,7,10,15(20)-heptaene-14,18-dione Chemical compound C1=C(N)C=C2C(CCCC)=C(CN3C(C4=C([C@@](C(=O)OC4)(O)CC)C=C33)=O)C3=NC2=C1 OZXMCEGJEIIDQT-DEOSSOPVSA-N 0.000 claims description 4
- 238000002560 therapeutic procedure Methods 0.000 claims description 4
- 108010016626 Dipeptides Proteins 0.000 claims description 3
- 102100037486 Reverse transcriptase/ribonuclease H Human genes 0.000 claims 1
- 229940124597 therapeutic agent Drugs 0.000 abstract description 3
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 228
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 192
- DTQVDTLACAAQTR-UHFFFAOYSA-N Trifluoroacetic acid Chemical compound OC(=O)C(F)(F)F DTQVDTLACAAQTR-UHFFFAOYSA-N 0.000 description 191
- 239000000243 solution Substances 0.000 description 173
- 238000004895 liquid chromatography mass spectrometry Methods 0.000 description 164
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 164
- 229910001868 water Inorganic materials 0.000 description 157
- 239000007787 solid Substances 0.000 description 155
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 154
- PYFSLJVSCGXYAJ-UHFFFAOYSA-N methyl 2-hydroxy-4-[[3-(2-hydroxyphenyl)phenyl]sulfonylamino]benzoate Chemical compound C1=C(O)C(C(=O)OC)=CC=C1NS(=O)(=O)C1=CC=CC(C=2C(=CC=CC=2)O)=C1 PYFSLJVSCGXYAJ-UHFFFAOYSA-N 0.000 description 132
- 238000000746 purification Methods 0.000 description 130
- 125000005647 linker group Chemical group 0.000 description 119
- 229940049595 antibody-drug conjugate Drugs 0.000 description 111
- 238000005160 1H NMR spectroscopy Methods 0.000 description 98
- 239000000562 conjugate Substances 0.000 description 97
- 238000002953 preparative HPLC Methods 0.000 description 93
- 239000000611 antibody drug conjugate Substances 0.000 description 85
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 81
- 125000004103 aminoalkyl group Chemical group 0.000 description 81
- 210000004027 cell Anatomy 0.000 description 67
- 239000000203 mixture Substances 0.000 description 47
- JGFZNNIVVJXRND-UHFFFAOYSA-N N,N-Diisopropylethylamine (DIPEA) Chemical compound CCN(C(C)C)C(C)C JGFZNNIVVJXRND-UHFFFAOYSA-N 0.000 description 45
- OKKJLVBELUTLKV-VMNATFBRSA-N methanol-d1 Chemical compound [2H]OC OKKJLVBELUTLKV-VMNATFBRSA-N 0.000 description 45
- 238000006243 chemical reaction Methods 0.000 description 44
- IAZDPXIOMUYVGZ-WFGJKAKNSA-N Dimethyl sulfoxide Chemical compound [2H]C([2H])([2H])S(=O)C([2H])([2H])[2H] IAZDPXIOMUYVGZ-WFGJKAKNSA-N 0.000 description 41
- AICOOMRHRUFYCM-ZRRPKQBOSA-N oxazine, 1 Chemical compound C([C@@H]1[C@H](C(C[C@]2(C)[C@@H]([C@H](C)N(C)C)[C@H](O)C[C@]21C)=O)CC1=CC2)C[C@H]1[C@@]1(C)[C@H]2N=C(C(C)C)OC1 AICOOMRHRUFYCM-ZRRPKQBOSA-N 0.000 description 41
- NQRYJNQNLNOLGT-UHFFFAOYSA-N Piperidine Chemical compound C1CCNCC1 NQRYJNQNLNOLGT-UHFFFAOYSA-N 0.000 description 38
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 36
- SJRJJKPEHAURKC-UHFFFAOYSA-N N-Methylmorpholine Chemical compound CN1CCOCC1 SJRJJKPEHAURKC-UHFFFAOYSA-N 0.000 description 36
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 35
- 239000000543 intermediate Substances 0.000 description 30
- 239000011541 reaction mixture Substances 0.000 description 30
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 29
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 29
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 28
- 238000003756 stirring Methods 0.000 description 28
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 24
- 238000007792 addition Methods 0.000 description 23
- 239000002244 precipitate Substances 0.000 description 21
- 238000002360 preparation method Methods 0.000 description 20
- WEVYAHXRMPXWCK-FIBGUPNXSA-N acetonitrile-d3 Chemical compound [2H]C([2H])([2H])C#N WEVYAHXRMPXWCK-FIBGUPNXSA-N 0.000 description 19
- 238000001914 filtration Methods 0.000 description 19
- 238000004191 hydrophobic interaction chromatography Methods 0.000 description 19
- 230000021615 conjugation Effects 0.000 description 18
- 230000003013 cytotoxicity Effects 0.000 description 18
- 231100000135 cytotoxicity Toxicity 0.000 description 18
- 239000000843 powder Substances 0.000 description 18
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 15
- 239000000047 product Substances 0.000 description 15
- KDXKERNSBIXSRK-YFKPBYRVSA-N L-lysine Chemical compound NCCCC[C@H](N)C(O)=O KDXKERNSBIXSRK-YFKPBYRVSA-N 0.000 description 14
- KDXKERNSBIXSRK-UHFFFAOYSA-N Lysine Natural products NCCCCC(N)C(O)=O KDXKERNSBIXSRK-UHFFFAOYSA-N 0.000 description 14
- HEDRZPFGACZZDS-MICDWDOJSA-N Trichloro(2H)methane Chemical compound [2H]C(Cl)(Cl)Cl HEDRZPFGACZZDS-MICDWDOJSA-N 0.000 description 14
- DYHSDKLCOJIUFX-UHFFFAOYSA-N tert-butoxycarbonyl anhydride Chemical compound CC(C)(C)OC(=O)OC(=O)OC(C)(C)C DYHSDKLCOJIUFX-UHFFFAOYSA-N 0.000 description 14
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 13
- 239000012071 phase Substances 0.000 description 13
- 239000000725 suspension Substances 0.000 description 13
- 238000012360 testing method Methods 0.000 description 13
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 12
- 150000001412 amines Chemical class 0.000 description 12
- 235000019439 ethyl acetate Nutrition 0.000 description 12
- 239000000377 silicon dioxide Substances 0.000 description 12
- 206010006187 Breast cancer Diseases 0.000 description 11
- CKLJMWTZIZZHCS-REOHCLBHSA-N L-aspartic acid Chemical compound OC(=O)[C@@H](N)CC(O)=O CKLJMWTZIZZHCS-REOHCLBHSA-N 0.000 description 11
- 241000699670 Mus sp. Species 0.000 description 11
- 125000003277 amino group Chemical group 0.000 description 11
- 238000005516 engineering process Methods 0.000 description 11
- 150000002148 esters Chemical class 0.000 description 11
- 125000000524 functional group Chemical group 0.000 description 11
- 238000000338 in vitro Methods 0.000 description 11
- 238000001542 size-exclusion chromatography Methods 0.000 description 11
- 208000026310 Breast neoplasm Diseases 0.000 description 10
- 102000035195 Peptidases Human genes 0.000 description 10
- 238000003776 cleavage reaction Methods 0.000 description 10
- 238000004108 freeze drying Methods 0.000 description 10
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical class CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 10
- 230000002829 reductive effect Effects 0.000 description 10
- 230000007017 scission Effects 0.000 description 10
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 10
- 229960000575 trastuzumab Drugs 0.000 description 10
- KXDHJXZQYSOELW-UHFFFAOYSA-M Carbamate Chemical compound NC([O-])=O KXDHJXZQYSOELW-UHFFFAOYSA-M 0.000 description 9
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 9
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 9
- VMHLLURERBWHNL-UHFFFAOYSA-M Sodium acetate Chemical compound [Na+].CC([O-])=O VMHLLURERBWHNL-UHFFFAOYSA-M 0.000 description 9
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 9
- 239000002253 acid Substances 0.000 description 9
- 239000004480 active ingredient Substances 0.000 description 9
- 150000001413 amino acids Chemical group 0.000 description 9
- 239000000460 chlorine Substances 0.000 description 9
- 229940079593 drug Drugs 0.000 description 9
- 238000003379 elimination reaction Methods 0.000 description 9
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 9
- 229910052938 sodium sulfate Inorganic materials 0.000 description 9
- 241001465754 Metazoa Species 0.000 description 8
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical compound [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 description 8
- PZBFGYYEXUXCOF-UHFFFAOYSA-N TCEP Chemical compound OC(=O)CCP(CCC(O)=O)CCC(O)=O PZBFGYYEXUXCOF-UHFFFAOYSA-N 0.000 description 8
- 238000004458 analytical method Methods 0.000 description 8
- 239000000872 buffer Substances 0.000 description 8
- 230000000981 bystander Effects 0.000 description 8
- 150000003141 primary amines Chemical class 0.000 description 8
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 8
- 238000004007 reversed phase HPLC Methods 0.000 description 8
- 125000003396 thiol group Chemical group [H]S* 0.000 description 8
- FYSNRJHAOHDILO-UHFFFAOYSA-N thionyl chloride Chemical compound ClS(Cl)=O FYSNRJHAOHDILO-UHFFFAOYSA-N 0.000 description 8
- 230000004614 tumor growth Effects 0.000 description 8
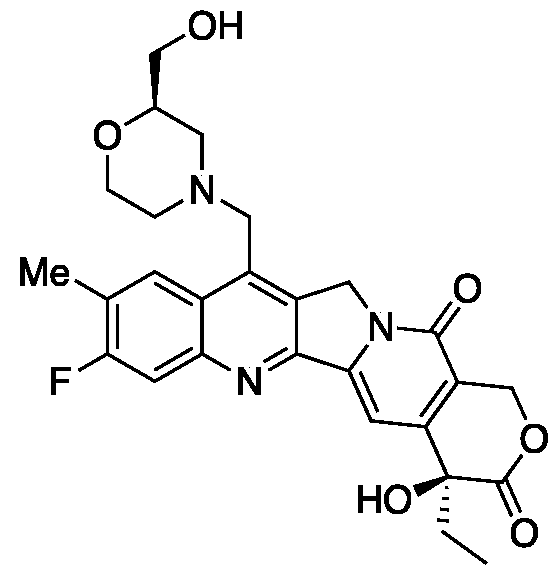
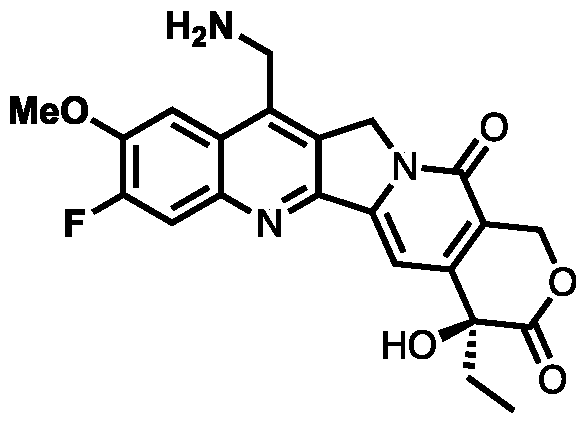
- RTHVXOWSCOVBGA-NRFANRHFSA-N CC[C@](C(C=C1N2CC3=C(CN)C(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CN)C(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O RTHVXOWSCOVBGA-NRFANRHFSA-N 0.000 description 7
- DTFQCOHTHXUPMI-FQEVSTJZSA-N CC[C@](C(C=C1N2CC3=CC(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=CC(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O DTFQCOHTHXUPMI-FQEVSTJZSA-N 0.000 description 7
- MHAJPDPJQMAIIY-UHFFFAOYSA-N Hydrogen peroxide Chemical compound OO MHAJPDPJQMAIIY-UHFFFAOYSA-N 0.000 description 7
- 239000012267 brine Substances 0.000 description 7
- 238000004128 high performance liquid chromatography Methods 0.000 description 7
- 239000000178 monomer Substances 0.000 description 7
- 239000000546 pharmaceutical excipient Substances 0.000 description 7
- 229920001223 polyethylene glycol Polymers 0.000 description 7
- 235000019419 proteases Nutrition 0.000 description 7
- 125000006239 protecting group Chemical group 0.000 description 7
- HPALAKNZSZLMCH-UHFFFAOYSA-M sodium;chloride;hydrate Chemical compound O.[Na+].[Cl-] HPALAKNZSZLMCH-UHFFFAOYSA-M 0.000 description 7
- LMDZBCPBFSXMTL-UHFFFAOYSA-N 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide Chemical compound CCN=C=NCCCN(C)C LMDZBCPBFSXMTL-UHFFFAOYSA-N 0.000 description 6
- FPIRBHDGWMWJEP-UHFFFAOYSA-N 1-hydroxy-7-azabenzotriazole Chemical compound C1=CN=C2N(O)N=NC2=C1 FPIRBHDGWMWJEP-UHFFFAOYSA-N 0.000 description 6
- HZAXFHJVJLSVMW-UHFFFAOYSA-N 2-Aminoethan-1-ol Chemical compound NCCO HZAXFHJVJLSVMW-UHFFFAOYSA-N 0.000 description 6
- VTYYLEPIZMXCLO-UHFFFAOYSA-L Calcium carbonate Chemical compound [Ca+2].[O-]C([O-])=O VTYYLEPIZMXCLO-UHFFFAOYSA-L 0.000 description 6
- 101001012157 Homo sapiens Receptor tyrosine-protein kinase erbB-2 Proteins 0.000 description 6
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 6
- YNAVUWVOSKDBBP-UHFFFAOYSA-N Morpholine Chemical compound C1COCCN1 YNAVUWVOSKDBBP-UHFFFAOYSA-N 0.000 description 6
- 241000699666 Mus <mouse, genus> Species 0.000 description 6
- QPCDCPDFJACHGM-UHFFFAOYSA-N N,N-bis{2-[bis(carboxymethyl)amino]ethyl}glycine Chemical compound OC(=O)CN(CC(O)=O)CCN(CC(=O)O)CCN(CC(O)=O)CC(O)=O QPCDCPDFJACHGM-UHFFFAOYSA-N 0.000 description 6
- 239000002202 Polyethylene glycol Substances 0.000 description 6
- 102100030086 Receptor tyrosine-protein kinase erbB-2 Human genes 0.000 description 6
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 6
- 150000001299 aldehydes Chemical class 0.000 description 6
- 238000013459 approach Methods 0.000 description 6
- 239000000796 flavoring agent Substances 0.000 description 6
- 238000009472 formulation Methods 0.000 description 6
- BDAGIHXWWSANSR-UHFFFAOYSA-N methanoic acid Natural products OC=O BDAGIHXWWSANSR-UHFFFAOYSA-N 0.000 description 6
- 239000012044 organic layer Substances 0.000 description 6
- 229960000402 palivizumab Drugs 0.000 description 6
- 229960003330 pentetic acid Drugs 0.000 description 6
- 108090000765 processed proteins & peptides Proteins 0.000 description 6
- 235000011152 sodium sulphate Nutrition 0.000 description 6
- 239000003765 sweetening agent Substances 0.000 description 6
- JOXIMZWYDAKGHI-UHFFFAOYSA-N toluene-4-sulfonic acid Chemical compound CC1=CC=C(S(O)(=O)=O)C=C1 JOXIMZWYDAKGHI-UHFFFAOYSA-N 0.000 description 6
- 210000004881 tumor cell Anatomy 0.000 description 6
- AEMRFAOFKBGASW-UHFFFAOYSA-N Glycolic acid Chemical class OCC(O)=O AEMRFAOFKBGASW-UHFFFAOYSA-N 0.000 description 5
- 229940024606 amino acid Drugs 0.000 description 5
- 235000001014 amino acid Nutrition 0.000 description 5
- 239000007900 aqueous suspension Substances 0.000 description 5
- 238000003556 assay Methods 0.000 description 5
- 238000010461 azide-alkyne cycloaddition reaction Methods 0.000 description 5
- 230000015572 biosynthetic process Effects 0.000 description 5
- 230000037396 body weight Effects 0.000 description 5
- 125000002843 carboxylic acid group Chemical group 0.000 description 5
- 239000007859 condensation product Substances 0.000 description 5
- 238000010511 deprotection reaction Methods 0.000 description 5
- 235000014113 dietary fatty acids Nutrition 0.000 description 5
- 239000002270 dispersing agent Substances 0.000 description 5
- 230000000694 effects Effects 0.000 description 5
- 230000008030 elimination Effects 0.000 description 5
- 239000000194 fatty acid Substances 0.000 description 5
- 229930195729 fatty acid Natural products 0.000 description 5
- 150000004665 fatty acids Chemical class 0.000 description 5
- 239000000706 filtrate Substances 0.000 description 5
- 235000013355 food flavoring agent Nutrition 0.000 description 5
- 235000003599 food sweetener Nutrition 0.000 description 5
- 230000006870 function Effects 0.000 description 5
- 125000005842 heteroatom Chemical group 0.000 description 5
- ALBYIUDWACNRRB-UHFFFAOYSA-N hexanamide Chemical compound CCCCCC(N)=O ALBYIUDWACNRRB-UHFFFAOYSA-N 0.000 description 5
- 238000001727 in vivo Methods 0.000 description 5
- 238000011534 incubation Methods 0.000 description 5
- 230000010354 integration Effects 0.000 description 5
- 239000000463 material Substances 0.000 description 5
- QSRRZKPKHJHIRB-UHFFFAOYSA-N methyl 4-[(2,5-dichloro-4-methylthiophen-3-yl)sulfonylamino]-2-hydroxybenzoate Chemical compound C1=C(O)C(C(=O)OC)=CC=C1NS(=O)(=O)C1=C(Cl)SC(Cl)=C1C QSRRZKPKHJHIRB-UHFFFAOYSA-N 0.000 description 5
- 238000009343 monoculture Methods 0.000 description 5
- 239000003921 oil Substances 0.000 description 5
- 235000019198 oils Nutrition 0.000 description 5
- 235000018102 proteins Nutrition 0.000 description 5
- 102000004169 proteins and genes Human genes 0.000 description 5
- 108090000623 proteins and genes Proteins 0.000 description 5
- 239000002904 solvent Substances 0.000 description 5
- 239000000375 suspending agent Substances 0.000 description 5
- 230000001225 therapeutic effect Effects 0.000 description 5
- 150000003573 thiols Chemical class 0.000 description 5
- 125000002221 trityl group Chemical group [H]C1=C([H])C([H])=C([H])C([H])=C1C([*])(C1=C(C(=C(C(=C1[H])[H])[H])[H])[H])C1=C([H])C([H])=C([H])C([H])=C1[H] 0.000 description 5
- 239000000080 wetting agent Substances 0.000 description 5
- IGKWOGMVAOYVSJ-ZDUSSCGKSA-N (4s)-4-ethyl-4-hydroxy-7,8-dihydro-1h-pyrano[3,4-f]indolizine-3,6,10-trione Chemical compound C1=C2C(=O)CCN2C(=O)C2=C1[C@](CC)(O)C(=O)OC2 IGKWOGMVAOYVSJ-ZDUSSCGKSA-N 0.000 description 4
- OIIOPWHTJZYKIL-PMACEKPBSA-N (5S)-5-[[[5-[2-chloro-3-[2-chloro-3-[6-methoxy-5-[[[(2S)-5-oxopyrrolidin-2-yl]methylamino]methyl]pyrazin-2-yl]phenyl]phenyl]-3-methoxypyrazin-2-yl]methylamino]methyl]pyrrolidin-2-one Chemical compound C1(=C(N=C(C2=C(C(C3=CC=CC(=C3Cl)C3=NC(OC)=C(N=C3)CNC[C@H]3NC(=O)CC3)=CC=C2)Cl)C=N1)OC)CNC[C@H]1NC(=O)CC1 OIIOPWHTJZYKIL-PMACEKPBSA-N 0.000 description 4
- LRMHQHUZLWXMEQ-UHFFFAOYSA-N 2-hydroxyethanesulfonyl chloride Chemical compound OCCS(Cl)(=O)=O LRMHQHUZLWXMEQ-UHFFFAOYSA-N 0.000 description 4
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 4
- VDFTXUVEZFUUDE-VWLOTQADSA-N CC[C@](C(C=C1N2CC3=C(CN4CCOCC4)C(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CN4CCOCC4)C(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O VDFTXUVEZFUUDE-VWLOTQADSA-N 0.000 description 4
- NTFVMBFNMVCYCG-QHCPKHFHSA-N CC[C@](C(C=C1N2CC3=C(CNC(C)=O)C(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CNC(C)=O)C(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O NTFVMBFNMVCYCG-QHCPKHFHSA-N 0.000 description 4
- MKIFEFMQSGQCSQ-NRFANRHFSA-N CC[C@](C(C=C1N2CC3=C(CO)C(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CO)C(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O MKIFEFMQSGQCSQ-NRFANRHFSA-N 0.000 description 4
- BWGNESOTFCXPMA-UHFFFAOYSA-N Dihydrogen disulfide Chemical compound SS BWGNESOTFCXPMA-UHFFFAOYSA-N 0.000 description 4
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 4
- IAYPIBMASNFSPL-UHFFFAOYSA-N Ethylene oxide Chemical compound C1CO1 IAYPIBMASNFSPL-UHFFFAOYSA-N 0.000 description 4
- 206010058467 Lung neoplasm malignant Diseases 0.000 description 4
- AFVFQIVMOAPDHO-UHFFFAOYSA-N Methanesulfonic acid Chemical compound CS(O)(=O)=O AFVFQIVMOAPDHO-UHFFFAOYSA-N 0.000 description 4
- 239000012359 Methanesulfonyl chloride Substances 0.000 description 4
- BAVYZALUXZFZLV-UHFFFAOYSA-N Methylamine Chemical compound NC BAVYZALUXZFZLV-UHFFFAOYSA-N 0.000 description 4
- 229910019142 PO4 Inorganic materials 0.000 description 4
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 4
- 229930006000 Sucrose Natural products 0.000 description 4
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 4
- 238000002835 absorbance Methods 0.000 description 4
- 125000002777 acetyl group Chemical group [H]C([H])([H])C(*)=O 0.000 description 4
- 150000001540 azides Chemical class 0.000 description 4
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 description 4
- FJDQFPXHSGXQBY-UHFFFAOYSA-L caesium carbonate Chemical compound [Cs+].[Cs+].[O-]C([O-])=O FJDQFPXHSGXQBY-UHFFFAOYSA-L 0.000 description 4
- 239000006285 cell suspension Substances 0.000 description 4
- 238000004587 chromatography analysis Methods 0.000 description 4
- 239000000839 emulsion Substances 0.000 description 4
- VKYKSIONXSXAKP-UHFFFAOYSA-N hexamethylenetetramine Chemical compound C1N(C2)CN3CN1CN2C3 VKYKSIONXSXAKP-UHFFFAOYSA-N 0.000 description 4
- 229910052739 hydrogen Inorganic materials 0.000 description 4
- 230000005764 inhibitory process Effects 0.000 description 4
- 125000003588 lysine group Chemical group [H]N([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])(N([H])[H])C(*)=O 0.000 description 4
- 230000014759 maintenance of location Effects 0.000 description 4
- QARBMVPHQWIHKH-UHFFFAOYSA-N methanesulfonyl chloride Chemical compound CS(Cl)(=O)=O QARBMVPHQWIHKH-UHFFFAOYSA-N 0.000 description 4
- 125000002950 monocyclic group Chemical group 0.000 description 4
- 229910052757 nitrogen Inorganic materials 0.000 description 4
- 150000007524 organic acids Chemical class 0.000 description 4
- 230000036961 partial effect Effects 0.000 description 4
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Chemical compound [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 description 4
- 239000003755 preservative agent Substances 0.000 description 4
- 230000009467 reduction Effects 0.000 description 4
- 238000010992 reflux Methods 0.000 description 4
- 229920006395 saturated elastomer Polymers 0.000 description 4
- 210000002966 serum Anatomy 0.000 description 4
- 239000011734 sodium Substances 0.000 description 4
- 229910000029 sodium carbonate Inorganic materials 0.000 description 4
- 239000012321 sodium triacetoxyborohydride Substances 0.000 description 4
- 239000012453 solvate Substances 0.000 description 4
- 241000894007 species Species 0.000 description 4
- 239000005720 sucrose Substances 0.000 description 4
- 239000003826 tablet Substances 0.000 description 4
- 230000035899 viability Effects 0.000 description 4
- VLARLSIGSPVYHX-UHFFFAOYSA-N (2,5-dioxopyrrolidin-1-yl) 6-(2,5-dioxopyrrol-1-yl)hexanoate Chemical compound O=C1CCC(=O)N1OC(=O)CCCCCN1C(=O)C=CC1=O VLARLSIGSPVYHX-UHFFFAOYSA-N 0.000 description 3
- KQRHTCDQWJLLME-XUXIUFHCSA-N (2s)-2-[[(2s)-2-[[(2s)-2-[[(2s)-2-aminopropanoyl]amino]-4-methylpentanoyl]amino]propanoyl]amino]-4-methylpentanoic acid Chemical compound CC(C)C[C@@H](C(O)=O)NC(=O)[C@H](C)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](C)N KQRHTCDQWJLLME-XUXIUFHCSA-N 0.000 description 3
- AGGWFDNPHKLBBV-YUMQZZPRSA-N (2s)-2-[[(2s)-2-amino-3-methylbutanoyl]amino]-5-(carbamoylamino)pentanoic acid Chemical compound CC(C)[C@H](N)C(=O)N[C@H](C(O)=O)CCCNC(N)=O AGGWFDNPHKLBBV-YUMQZZPRSA-N 0.000 description 3
- PFBYWNBYIPWLRG-UHFFFAOYSA-N (3-fluoroazetidin-3-yl)methanol Chemical compound OCC1(F)CNC1 PFBYWNBYIPWLRG-UHFFFAOYSA-N 0.000 description 3
- NANQJUFFKXWVJR-UHFFFAOYSA-N 1-(benzenesulfonyl)piperazine Chemical compound C=1C=CC=CC=1S(=O)(=O)N1CCNCC1 NANQJUFFKXWVJR-UHFFFAOYSA-N 0.000 description 3
- PVOAHINGSUIXLS-UHFFFAOYSA-N 1-Methylpiperazine Chemical compound CN1CCNCC1 PVOAHINGSUIXLS-UHFFFAOYSA-N 0.000 description 3
- NDKDFTQNXLHCGO-UHFFFAOYSA-N 2-(9h-fluoren-9-ylmethoxycarbonylamino)acetic acid Chemical compound C1=CC=C2C(COC(=O)NCC(=O)O)C3=CC=CC=C3C2=C1 NDKDFTQNXLHCGO-UHFFFAOYSA-N 0.000 description 3
- WEZDRVHTDXTVLT-GJZGRUSLSA-N 2-[[(2s)-2-[[(2s)-2-[(2-aminoacetyl)amino]-3-phenylpropanoyl]amino]-4-methylpentanoyl]amino]acetic acid Chemical compound OC(=O)CNC(=O)[C@H](CC(C)C)NC(=O)[C@@H](NC(=O)CN)CC1=CC=CC=C1 WEZDRVHTDXTVLT-GJZGRUSLSA-N 0.000 description 3
- HXUVTXPOZRFMOY-NSHDSACASA-N 2-[[(2s)-2-[[2-[(2-aminoacetyl)amino]acetyl]amino]-3-phenylpropanoyl]amino]acetic acid Chemical compound NCC(=O)NCC(=O)N[C@H](C(=O)NCC(O)=O)CC1=CC=CC=C1 HXUVTXPOZRFMOY-NSHDSACASA-N 0.000 description 3
- BOTJOIYABJNMEW-UHFFFAOYSA-N 3-azabicyclo[3.1.1]heptan-6-ol Chemical compound C1C2C(O)C1CNC2 BOTJOIYABJNMEW-UHFFFAOYSA-N 0.000 description 3
- OSWFIVFLDKOXQC-UHFFFAOYSA-N 4-(3-methoxyphenyl)aniline Chemical compound COC1=CC=CC(C=2C=CC(N)=CC=2)=C1 OSWFIVFLDKOXQC-UHFFFAOYSA-N 0.000 description 3
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 3
- AAQGRPOPTAUUBM-ZLUOBGJFSA-N Ala-Ala-Asn Chemical compound [H]N[C@@H](C)C(=O)N[C@@H](C)C(=O)N[C@@H](CC(N)=O)C(O)=O AAQGRPOPTAUUBM-ZLUOBGJFSA-N 0.000 description 3
- QXRNAOYBCYVZCD-BQBZGAKWSA-N Ala-Lys Chemical compound C[C@H](N)C(=O)N[C@H](C(O)=O)CCCCN QXRNAOYBCYVZCD-BQBZGAKWSA-N 0.000 description 3
- OMNVYXHOSHNURL-WPRPVWTQSA-N Ala-Phe Chemical compound C[C@H](N)C(=O)N[C@H](C(O)=O)CC1=CC=CC=C1 OMNVYXHOSHNURL-WPRPVWTQSA-N 0.000 description 3
- WEZNQZHACPSMEF-QEJZJMRPSA-N Ala-Phe-Lys Chemical compound NCCCC[C@@H](C(O)=O)NC(=O)[C@@H](NC(=O)[C@@H](N)C)CC1=CC=CC=C1 WEZNQZHACPSMEF-QEJZJMRPSA-N 0.000 description 3
- QJMCHPGWFZZRID-BQBZGAKWSA-N Asn-Lys Chemical compound NCCCC[C@@H](C(O)=O)NC(=O)[C@@H](N)CC(N)=O QJMCHPGWFZZRID-BQBZGAKWSA-N 0.000 description 3
- VHQSGALUSWIYOD-QXEWZRGKSA-N Asn-Pro-Val Chemical compound [H]N[C@@H](CC(N)=O)C(=O)N1CCC[C@H]1C(=O)N[C@@H](C(C)C)C(O)=O VHQSGALUSWIYOD-QXEWZRGKSA-N 0.000 description 3
- CPMKYMGGYUFOHS-FSPLSTOPSA-N Asp-Val Chemical compound CC(C)[C@@H](C(O)=O)NC(=O)[C@@H](N)CC(O)=O CPMKYMGGYUFOHS-FSPLSTOPSA-N 0.000 description 3
- XWKBWZXGNXTDKY-ZKWXMUAHSA-N Asp-Val-Ala Chemical compound OC(=O)[C@H](C)NC(=O)[C@H](C(C)C)NC(=O)[C@@H](N)CC(O)=O XWKBWZXGNXTDKY-ZKWXMUAHSA-N 0.000 description 3
- 241000416162 Astragalus gummifer Species 0.000 description 3
- NBPOCFCIIFOHEK-YTTGMZPUSA-N CC[C@](C(C=C1N2CC3=C(CN(CC4)CCN4C(C=C4)=CC=C4N)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CN(CC4)CCN4C(C=C4)=CC=C4N)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O NBPOCFCIIFOHEK-YTTGMZPUSA-N 0.000 description 3
- PPQRJBSWDLCYCP-HKBQPEDESA-N CC[C@](C(C=C1N2CC3=C(CN(CC4)CCN4C(OC(C)(C)C)=O)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CN(CC4)CCN4C(OC(C)(C)C)=O)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O PPQRJBSWDLCYCP-HKBQPEDESA-N 0.000 description 3
- SAXPMAYHHSDRQO-YTTGMZPUSA-N CC[C@](C(C=C1N2CC3=C(CN(CC4)CCN4S(C(C=C4)=CC=C4N)(=O)=O)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CN(CC4)CCN4S(C(C=C4)=CC=C4N)(=O)=O)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O SAXPMAYHHSDRQO-YTTGMZPUSA-N 0.000 description 3
- OHWRBAMYYKSMGR-SANMLTNESA-N CC[C@](C(C=C1N2CC3=C(CN4CCCCC4)C(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CN4CCCCC4)C(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O OHWRBAMYYKSMGR-SANMLTNESA-N 0.000 description 3
- FKVVDKIZCSODDI-NDEPHWFRSA-N CC[C@](C(C=C1N2CC3=C(CNS(C(C=C4)=CC=C4N)(=O)=O)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CNS(C(C=C4)=CC=C4N)(=O)=O)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O FKVVDKIZCSODDI-NDEPHWFRSA-N 0.000 description 3
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 3
- 239000004150 EU approved colour Substances 0.000 description 3
- 108010009504 Gly-Phe-Leu-Gly Proteins 0.000 description 3
- VLDVBZICYBVQHB-IUCAKERBSA-N His-Val Chemical compound CC(C)[C@@H](C([O-])=O)NC(=O)[C@@H]([NH3+])CC1=CN=CN1 VLDVBZICYBVQHB-IUCAKERBSA-N 0.000 description 3
- OAKJQQAXSVQMHS-UHFFFAOYSA-N Hydrazine Chemical compound NN OAKJQQAXSVQMHS-UHFFFAOYSA-N 0.000 description 3
- BBIXOODYWPFNDT-CIUDSAMLSA-N Ile-Pro Chemical compound CC[C@H](C)[C@H](N)C(=O)N1CCC[C@H]1C(O)=O BBIXOODYWPFNDT-CIUDSAMLSA-N 0.000 description 3
- BCXBIONYYJCSDF-CIUDSAMLSA-N Ile-Val Chemical compound CC[C@H](C)[C@H](N)C(=O)N[C@@H](C(C)C)C(O)=O BCXBIONYYJCSDF-CIUDSAMLSA-N 0.000 description 3
- FADYJNXDPBKVCA-UHFFFAOYSA-N L-Phenylalanyl-L-lysin Natural products NCCCCC(C(O)=O)NC(=O)C(N)CC1=CC=CC=C1 FADYJNXDPBKVCA-UHFFFAOYSA-N 0.000 description 3
- WMFOQBRAJBCJND-UHFFFAOYSA-M Lithium hydroxide Chemical compound [Li+].[OH-] WMFOQBRAJBCJND-UHFFFAOYSA-M 0.000 description 3
- VWPJQIHBBOJWDN-DCAQKATOSA-N Lys-Val-Ala Chemical compound [H]N[C@@H](CCCCN)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](C)C(O)=O VWPJQIHBBOJWDN-DCAQKATOSA-N 0.000 description 3
- IMTUWVJPCQPJEE-IUCAKERBSA-N Met-Lys Chemical compound CSCC[C@H](N)C(=O)N[C@H](C(O)=O)CCCCN IMTUWVJPCQPJEE-IUCAKERBSA-N 0.000 description 3
- 239000007832 Na2SO4 Substances 0.000 description 3
- MUBZPKHOEPUJKR-UHFFFAOYSA-N Oxalic acid Chemical compound OC(=O)C(O)=O MUBZPKHOEPUJKR-UHFFFAOYSA-N 0.000 description 3
- 235000019483 Peanut oil Nutrition 0.000 description 3
- OZILORBBPKKGRI-RYUDHWBXSA-N Phe-Arg Chemical compound NC(N)=NCCC[C@@H](C(O)=O)NC(=O)[C@@H](N)CC1=CC=CC=C1 OZILORBBPKKGRI-RYUDHWBXSA-N 0.000 description 3
- FADYJNXDPBKVCA-STQMWFEESA-N Phe-Lys Chemical compound NCCCC[C@@H](C(O)=O)NC(=O)[C@@H](N)CC1=CC=CC=C1 FADYJNXDPBKVCA-STQMWFEESA-N 0.000 description 3
- RBRNEFJTEHPDSL-ACRUOGEOSA-N Phe-Phe-Lys Chemical compound C([C@@H](C(=O)N[C@@H](CCCCN)C(O)=O)NC(=O)[C@@H](N)CC=1C=CC=CC=1)C1=CC=CC=C1 RBRNEFJTEHPDSL-ACRUOGEOSA-N 0.000 description 3
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 description 3
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 3
- 229920001615 Tragacanth Polymers 0.000 description 3
- HSRXSKHRSXRCFC-WDSKDSINSA-N Val-Ala Chemical compound CC(C)[C@H](N)C(=O)N[C@@H](C)C(O)=O HSRXSKHRSXRCFC-WDSKDSINSA-N 0.000 description 3
- XXDVDTMEVBYRPK-XPUUQOCRSA-N Val-Gln Chemical compound CC(C)[C@H](N)C(=O)N[C@H](C(O)=O)CCC(N)=O XXDVDTMEVBYRPK-XPUUQOCRSA-N 0.000 description 3
- JKHXYJKMNSSFFL-IUCAKERBSA-N Val-Lys Chemical compound CC(C)[C@H](N)C(=O)N[C@H](C(O)=O)CCCCN JKHXYJKMNSSFFL-IUCAKERBSA-N 0.000 description 3
- IOUPEELXVYPCPG-UHFFFAOYSA-N Valylglycine Chemical compound CC(C)C(N)C(=O)NCC(O)=O IOUPEELXVYPCPG-UHFFFAOYSA-N 0.000 description 3
- 230000002776 aggregation Effects 0.000 description 3
- 238000004220 aggregation Methods 0.000 description 3
- 108010054982 alanyl-leucyl-alanyl-leucine Proteins 0.000 description 3
- 108010011559 alanylphenylalanine Proteins 0.000 description 3
- 230000000259 anti-tumor effect Effects 0.000 description 3
- 239000002585 base Substances 0.000 description 3
- 125000003236 benzoyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C(*)=O 0.000 description 3
- 125000002619 bicyclic group Chemical group 0.000 description 3
- 229910000019 calcium carbonate Inorganic materials 0.000 description 3
- 235000010216 calcium carbonate Nutrition 0.000 description 3
- 239000001506 calcium phosphate Substances 0.000 description 3
- 125000000837 carbohydrate group Chemical group 0.000 description 3
- 229910052799 carbon Inorganic materials 0.000 description 3
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 3
- 239000003153 chemical reaction reagent Substances 0.000 description 3
- 229910052801 chlorine Inorganic materials 0.000 description 3
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 3
- 238000003501 co-culture Methods 0.000 description 3
- 230000008878 coupling Effects 0.000 description 3
- 238000010168 coupling process Methods 0.000 description 3
- 238000005859 coupling reaction Methods 0.000 description 3
- 238000012258 culturing Methods 0.000 description 3
- 125000000151 cysteine group Chemical class N[C@@H](CS)C(=O)* 0.000 description 3
- 238000011033 desalting Methods 0.000 description 3
- 238000001514 detection method Methods 0.000 description 3
- 235000011180 diphosphates Nutrition 0.000 description 3
- 238000009826 distribution Methods 0.000 description 3
- VHJLVAABSRFDPM-QWWZWVQMSA-N dithiothreitol Chemical compound SC[C@@H](O)[C@H](O)CS VHJLVAABSRFDPM-QWWZWVQMSA-N 0.000 description 3
- 125000005519 fluorenylmethyloxycarbonyl group Chemical group 0.000 description 3
- 229910052731 fluorine Inorganic materials 0.000 description 3
- 235000019253 formic acid Nutrition 0.000 description 3
- 239000007903 gelatin capsule Substances 0.000 description 3
- XKUKSGPZAADMRA-UHFFFAOYSA-N glycyl-glycyl-glycine Chemical compound NCC(=O)NCC(=O)NCC(O)=O XKUKSGPZAADMRA-UHFFFAOYSA-N 0.000 description 3
- 239000008187 granular material Substances 0.000 description 3
- 125000005179 haloacetyl group Chemical group 0.000 description 3
- 150000004688 heptahydrates Chemical class 0.000 description 3
- 150000003840 hydrochlorides Chemical class 0.000 description 3
- 239000001257 hydrogen Substances 0.000 description 3
- 125000004435 hydrogen atom Chemical group [H]* 0.000 description 3
- 239000005457 ice water Substances 0.000 description 3
- 150000007529 inorganic bases Chemical class 0.000 description 3
- BAUYGSIQEAFULO-UHFFFAOYSA-L iron(2+) sulfate (anhydrous) Chemical compound [Fe+2].[O-]S([O-])(=O)=O BAUYGSIQEAFULO-UHFFFAOYSA-L 0.000 description 3
- 229910000359 iron(II) sulfate Inorganic materials 0.000 description 3
- 108010078274 isoleucylvaline Proteins 0.000 description 3
- 150000002540 isothiocyanates Chemical class 0.000 description 3
- 125000000468 ketone group Chemical group 0.000 description 3
- 239000010410 layer Substances 0.000 description 3
- 229940057995 liquid paraffin Drugs 0.000 description 3
- 201000005202 lung cancer Diseases 0.000 description 3
- 208000020816 lung neoplasm Diseases 0.000 description 3
- 238000004949 mass spectrometry Methods 0.000 description 3
- 229920000609 methyl cellulose Polymers 0.000 description 3
- NQMRYBIKMRVZLB-UHFFFAOYSA-N methylamine hydrochloride Chemical compound [Cl-].[NH3+]C NQMRYBIKMRVZLB-UHFFFAOYSA-N 0.000 description 3
- 239000001923 methylcellulose Substances 0.000 description 3
- 235000010981 methylcellulose Nutrition 0.000 description 3
- 150000007522 mineralic acids Chemical class 0.000 description 3
- 238000002156 mixing Methods 0.000 description 3
- 150000004682 monohydrates Chemical class 0.000 description 3
- 231100000252 nontoxic Toxicity 0.000 description 3
- 230000003000 nontoxic effect Effects 0.000 description 3
- 239000004006 olive oil Substances 0.000 description 3
- 235000008390 olive oil Nutrition 0.000 description 3
- 229940006093 opthalmologic coloring agent diagnostic Drugs 0.000 description 3
- 239000012074 organic phase Substances 0.000 description 3
- 239000000312 peanut oil Substances 0.000 description 3
- 239000003208 petroleum Substances 0.000 description 3
- 108010018625 phenylalanylarginine Proteins 0.000 description 3
- 125000006413 ring segment Chemical group 0.000 description 3
- FVAUCKIRQBBSSJ-UHFFFAOYSA-M sodium iodide Chemical compound [Na+].[I-] FVAUCKIRQBBSSJ-UHFFFAOYSA-M 0.000 description 3
- 108090000250 sortase A Proteins 0.000 description 3
- 239000000126 substance Substances 0.000 description 3
- 238000003786 synthesis reaction Methods 0.000 description 3
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 3
- 238000004809 thin layer chromatography Methods 0.000 description 3
- ATGUDZODTABURZ-UHFFFAOYSA-N thiolan-2-ylideneazanium;chloride Chemical compound Cl.N=C1CCCS1 ATGUDZODTABURZ-UHFFFAOYSA-N 0.000 description 3
- 238000000108 ultra-filtration Methods 0.000 description 3
- 108010073969 valyllysine Proteins 0.000 description 3
- 235000015112 vegetable and seed oil Nutrition 0.000 description 3
- 239000008158 vegetable oil Substances 0.000 description 3
- UGOMMVLRQDMAQQ-UHFFFAOYSA-N xphos Chemical compound CC(C)C1=CC(C(C)C)=CC(C(C)C)=C1C1=CC=CC=C1P(C1CCCCC1)C1CCCCC1 UGOMMVLRQDMAQQ-UHFFFAOYSA-N 0.000 description 3
- DGVVWUTYPXICAM-UHFFFAOYSA-N β‐Mercaptoethanol Chemical compound OCCS DGVVWUTYPXICAM-UHFFFAOYSA-N 0.000 description 3
- PUPZLCDOIYMWBV-UHFFFAOYSA-N (+/-)-1,3-Butanediol Chemical compound CC(O)CCO PUPZLCDOIYMWBV-UHFFFAOYSA-N 0.000 description 2
- UASDKSVNENJORL-UHFFFAOYSA-N (2,5-dioxocyclopentyl) 6-(2,5-dioxopyrrol-1-yl)hexanoate Chemical compound O=C(CCCCCN1C(=O)C=CC1=O)OC1C(=O)CCC1=O UASDKSVNENJORL-UHFFFAOYSA-N 0.000 description 2
- NHUCANAMPJGMQL-ZDUSSCGKSA-N (2,5-dioxopyrrolidin-1-yl) (2s)-2-[(2-methylpropan-2-yl)oxycarbonylamino]-3-phenylpropanoate Chemical compound C([C@H](NC(=O)OC(C)(C)C)C(=O)ON1C(CCC1=O)=O)C1=CC=CC=C1 NHUCANAMPJGMQL-ZDUSSCGKSA-N 0.000 description 2
- NKUZQMZWTZAPSN-UHFFFAOYSA-N (2,5-dioxopyrrolidin-1-yl) 2-bromoacetate Chemical compound BrCC(=O)ON1C(=O)CCC1=O NKUZQMZWTZAPSN-UHFFFAOYSA-N 0.000 description 2
- VRDGQQTWSGDXCU-UHFFFAOYSA-N (2,5-dioxopyrrolidin-1-yl) 2-iodoacetate Chemical compound ICC(=O)ON1C(=O)CCC1=O VRDGQQTWSGDXCU-UHFFFAOYSA-N 0.000 description 2
- JWDFQMWEFLOOED-UHFFFAOYSA-N (2,5-dioxopyrrolidin-1-yl) 3-(pyridin-2-yldisulfanyl)propanoate Chemical compound O=C1CCC(=O)N1OC(=O)CCSSC1=CC=CC=N1 JWDFQMWEFLOOED-UHFFFAOYSA-N 0.000 description 2
- WGMMKWFUXPMTRW-UHFFFAOYSA-N (2,5-dioxopyrrolidin-1-yl) 3-[(2-bromoacetyl)amino]propanoate Chemical compound BrCC(=O)NCCC(=O)ON1C(=O)CCC1=O WGMMKWFUXPMTRW-UHFFFAOYSA-N 0.000 description 2
- PVGATNRYUYNBHO-UHFFFAOYSA-N (2,5-dioxopyrrolidin-1-yl) 4-(2,5-dioxopyrrol-1-yl)butanoate Chemical compound O=C1CCC(=O)N1OC(=O)CCCN1C(=O)C=CC1=O PVGATNRYUYNBHO-UHFFFAOYSA-N 0.000 description 2
- PMJWDPGOWBRILU-UHFFFAOYSA-N (2,5-dioxopyrrolidin-1-yl) 4-[4-(2,5-dioxopyrrol-1-yl)phenyl]butanoate Chemical compound O=C1CCC(=O)N1OC(=O)CCCC(C=C1)=CC=C1N1C(=O)C=CC1=O PMJWDPGOWBRILU-UHFFFAOYSA-N 0.000 description 2
- WCMOHMXWOOBVMZ-UHFFFAOYSA-N (2,5-dioxopyrrolidin-1-yl) 6-[3-(2,5-dioxopyrrol-1-yl)propanoylamino]hexanoate Chemical compound O=C1CCC(=O)N1OC(=O)CCCCCNC(=O)CCN1C(=O)C=CC1=O WCMOHMXWOOBVMZ-UHFFFAOYSA-N 0.000 description 2
- LOVPHSMOAVXQIH-UHFFFAOYSA-N (4-nitrophenyl) hydrogen carbonate Chemical compound OC(=O)OC1=CC=C([N+]([O-])=O)C=C1 LOVPHSMOAVXQIH-UHFFFAOYSA-N 0.000 description 2
- ZORQXIQZAOLNGE-UHFFFAOYSA-N 1,1-difluorocyclohexane Chemical compound FC1(F)CCCCC1 ZORQXIQZAOLNGE-UHFFFAOYSA-N 0.000 description 2
- TZCPCKNHXULUIY-RGULYWFUSA-N 1,2-distearoyl-sn-glycero-3-phosphoserine Chemical compound CCCCCCCCCCCCCCCCCC(=O)OC[C@H](COP(O)(=O)OC[C@H](N)C(O)=O)OC(=O)CCCCCCCCCCCCCCCCC TZCPCKNHXULUIY-RGULYWFUSA-N 0.000 description 2
- RYHBNJHYFVUHQT-UHFFFAOYSA-N 1,4-Dioxane Chemical compound C1COCCO1 RYHBNJHYFVUHQT-UHFFFAOYSA-N 0.000 description 2
- FPKVOQKZMBDBKP-UHFFFAOYSA-N 1-[4-[(2,5-dioxopyrrol-1-yl)methyl]cyclohexanecarbonyl]oxy-2,5-dioxopyrrolidine-3-sulfonic acid Chemical compound O=C1C(S(=O)(=O)O)CC(=O)N1OC(=O)C1CCC(CN2C(C=CC2=O)=O)CC1 FPKVOQKZMBDBKP-UHFFFAOYSA-N 0.000 description 2
- SDTORDSXCYSNTD-UHFFFAOYSA-N 1-methoxy-4-[(4-methoxyphenyl)methoxymethyl]benzene Chemical compound C1=CC(OC)=CC=C1COCC1=CC=C(OC)C=C1 SDTORDSXCYSNTD-UHFFFAOYSA-N 0.000 description 2
- VBICKXHEKHSIBG-UHFFFAOYSA-N 1-monostearoylglycerol Chemical compound CCCCCCCCCCCCCCCCCC(=O)OCC(O)CO VBICKXHEKHSIBG-UHFFFAOYSA-N 0.000 description 2
- IIZPXYDJLKNOIY-JXPKJXOSSA-N 1-palmitoyl-2-arachidonoyl-sn-glycero-3-phosphocholine Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCC\C=C/C\C=C/C\C=C/C\C=C/CCCCC IIZPXYDJLKNOIY-JXPKJXOSSA-N 0.000 description 2
- IZHVBANLECCAGF-UHFFFAOYSA-N 2-hydroxy-3-(octadecanoyloxy)propyl octadecanoate Chemical compound CCCCCCCCCCCCCCCCCC(=O)OCC(O)COC(=O)CCCCCCCCCCCCCCCCC IZHVBANLECCAGF-UHFFFAOYSA-N 0.000 description 2
- BMYNFMYTOJXKLE-UHFFFAOYSA-N 3-azaniumyl-2-hydroxypropanoate Chemical compound NCC(O)C(O)=O BMYNFMYTOJXKLE-UHFFFAOYSA-N 0.000 description 2
- BFWYZZPDZZGSLJ-UHFFFAOYSA-N 4-(aminomethyl)aniline Chemical compound NCC1=CC=C(N)C=C1 BFWYZZPDZZGSLJ-UHFFFAOYSA-N 0.000 description 2
- JXRGUPLJCCDGKG-UHFFFAOYSA-N 4-nitrobenzenesulfonyl chloride Chemical compound [O-][N+](=O)C1=CC=C(S(Cl)(=O)=O)C=C1 JXRGUPLJCCDGKG-UHFFFAOYSA-N 0.000 description 2
- BTJIUGUIPKRLHP-UHFFFAOYSA-N 4-nitrophenol Chemical compound OC1=CC=C([N+]([O-])=O)C=C1 BTJIUGUIPKRLHP-UHFFFAOYSA-N 0.000 description 2
- VAVOYRCCWLRTMS-UHFFFAOYSA-N 4-piperazin-1-ylaniline Chemical compound C1=CC(N)=CC=C1N1CCNCC1 VAVOYRCCWLRTMS-UHFFFAOYSA-N 0.000 description 2
- GFDCXCIQDDYTEL-UHFFFAOYSA-N 4-piperazin-1-ylsulfonylaniline Chemical compound C1=CC(N)=CC=C1S(=O)(=O)N1CCNCC1 GFDCXCIQDDYTEL-UHFFFAOYSA-N 0.000 description 2
- 244000215068 Acacia senegal Species 0.000 description 2
- 235000006491 Acacia senegal Nutrition 0.000 description 2
- 208000036832 Adenocarcinoma of ovary Diseases 0.000 description 2
- GUBGYTABKSRVRQ-XLOQQCSPSA-N Alpha-Lactose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-XLOQQCSPSA-N 0.000 description 2
- CIWBSHSKHKDKBQ-JLAZNSOCSA-N Ascorbic acid Chemical compound OC[C@H](O)[C@H]1OC(=O)C(O)=C1O CIWBSHSKHKDKBQ-JLAZNSOCSA-N 0.000 description 2
- LJZOXYJYMHNKAY-QFIPXVFZSA-N CC[C@](C(C=C1N2CC3=C(CC)C(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CC)C(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O LJZOXYJYMHNKAY-QFIPXVFZSA-N 0.000 description 2
- QBJHYMQBCSRTMX-QHCPKHFHSA-N CC[C@](C(C=C1N2CC3=C(CCOC)C(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CCOC)C(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O QBJHYMQBCSRTMX-QHCPKHFHSA-N 0.000 description 2
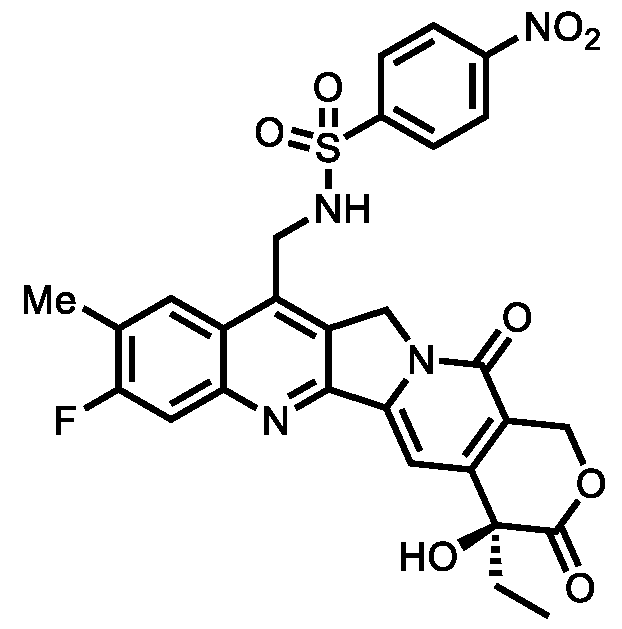
- XKWWCNGUJCEGFD-LJAQVGFWSA-N CC[C@](C(C=C1N2CC3=C(CN(C(O)=O)C(C=C4)=CC=C4[N+]([O-])=O)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CN(C(O)=O)C(C=C4)=CC=C4[N+]([O-])=O)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O XKWWCNGUJCEGFD-LJAQVGFWSA-N 0.000 description 2
- WJZRBBAPUDWJOO-DEOSSOPVSA-N CC[C@](C(C=C1N2CC3=C(CN(C)C(CO)=O)C(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CN(C)C(CO)=O)C(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O WJZRBBAPUDWJOO-DEOSSOPVSA-N 0.000 description 2
- ORAQQWSBJJQICA-QHCPKHFHSA-N CC[C@](C(C=C1N2CC3=C(CN(C)S(C)(=O)=O)C(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CN(C)S(C)(=O)=O)C(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O ORAQQWSBJJQICA-QHCPKHFHSA-N 0.000 description 2
- MIVCBCAYMISUIT-VWLOTQADSA-N CC[C@](C(C=C1N2CC3=C(CN(C4)CC4(CO)F)C(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CN(C4)CC4(CO)F)C(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O MIVCBCAYMISUIT-VWLOTQADSA-N 0.000 description 2
- HCISXLMGAJMXGP-KSKVVAMCSA-N CC[C@](C(C=C1N2CC3=C(CN(CC4)CC(CO)C4(F)F)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CN(CC4)CC(CO)C4(F)F)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O HCISXLMGAJMXGP-KSKVVAMCSA-N 0.000 description 2
- VZVIOXYNQQVQJW-YTTGMZPUSA-N CC[C@](C(C=C1N2CC3=C(CN(CC4)CCN4C(C=C4)=CC=C4N)C(C=C(C(F)=C4)OC)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CN(CC4)CCN4C(C=C4)=CC=C4N)C(C=C(C(F)=C4)OC)=C4N=C13)=C(CO1)C2=O)(C1=O)O VZVIOXYNQQVQJW-YTTGMZPUSA-N 0.000 description 2
- SAJRUTUFRZDRSE-HKBQPEDESA-N CC[C@](C(C=C1N2CC3=C(CN(CC4)CCN4S(C4=CC=CC=C4)(=O)=O)C(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CN(CC4)CCN4S(C4=CC=CC=C4)(=O)=O)C(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O SAJRUTUFRZDRSE-HKBQPEDESA-N 0.000 description 2
- SHLMZHOSZGTBDN-YTTGMZPUSA-N CC[C@](C(C=C1N2CC3=C(CN(CC4)CCN4S(C4=CC=CC=C4)(=O)=O)C(C=C(C(F)=C4)OC)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CN(CC4)CCN4S(C4=CC=CC=C4)(=O)=O)C(C=C(C(F)=C4)OC)=C4N=C13)=C(CO1)C2=O)(C1=O)O SHLMZHOSZGTBDN-YTTGMZPUSA-N 0.000 description 2
- PWOHEMBXRIJZKW-YTTGMZPUSA-N CC[C@](C(C=C1N2CC3=C(CN(CC4)CCN4S(C4=CC=CC=C4)(=O)=O)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CN(CC4)CCN4S(C4=CC=CC=C4)(=O)=O)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O PWOHEMBXRIJZKW-YTTGMZPUSA-N 0.000 description 2
- FVXPYFSOQGHWQD-LXDMYWKFSA-N CC[C@](C(C=C1N2CC3=C(CN(CC4C5)CC5C4O)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CN(CC4C5)CC5C4O)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O FVXPYFSOQGHWQD-LXDMYWKFSA-N 0.000 description 2
- UQCWGBBGZNWOFS-JDHIJPHQSA-N CC[C@](C(C=C1N2CC3=C(CN(C[C@H]4C5)C[C@H]5C4O)C(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CN(C[C@H]4C5)C[C@H]5C4O)C(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O UQCWGBBGZNWOFS-JDHIJPHQSA-N 0.000 description 2
- UDQFMOJLFXLAEI-KSKVVAMCSA-N CC[C@](C(C=C1N2CC3=C(CN4C(CO)CSCC4)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CN4C(CO)CSCC4)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O UDQFMOJLFXLAEI-KSKVVAMCSA-N 0.000 description 2
- CVCVTROMVVAEQA-BKCIKAJISA-N CC[C@](C(C=C1N2CC3=C(CN4C5(CO)CCC4CC5)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CN4C5(CO)CCC4CC5)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O CVCVTROMVVAEQA-BKCIKAJISA-N 0.000 description 2
- AVEWRPFINBCZKE-SANMLTNESA-N CC[C@](C(C=C1N2CC3=C(CN4CC(CO)C4)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CN4CC(CO)C4)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O AVEWRPFINBCZKE-SANMLTNESA-N 0.000 description 2
- RULOIEOXSYPMGN-MHZLTWQESA-N CC[C@](C(C=C1N2CC3=C(CN4CCCCC4)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CN4CCCCC4)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O RULOIEOXSYPMGN-MHZLTWQESA-N 0.000 description 2
- AAKHISJUTYVEIM-SANMLTNESA-N CC[C@](C(C=C1N2CC3=C(CN4CCN(C)CC4)C(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CN4CCN(C)CC4)C(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O AAKHISJUTYVEIM-SANMLTNESA-N 0.000 description 2
- DRIRJNRAINJZAJ-MHZLTWQESA-N CC[C@](C(C=C1N2CC3=C(CN4CCN(C)CC4)C(C=C(C(F)=C4)OC)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CN4CCN(C)CC4)C(C=C(C(F)=C4)OC)=C4N=C13)=C(CO1)C2=O)(C1=O)O DRIRJNRAINJZAJ-MHZLTWQESA-N 0.000 description 2
- HJGKPZJNCDAIJU-MHZLTWQESA-N CC[C@](C(C=C1N2CC3=C(CN4CCN(C)CC4)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CN4CCN(C)CC4)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O HJGKPZJNCDAIJU-MHZLTWQESA-N 0.000 description 2
- XASXUUJFCBRLCG-SANMLTNESA-N CC[C@](C(C=C1N2CC3=C(CN4CCOCC4)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CN4CCOCC4)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O XASXUUJFCBRLCG-SANMLTNESA-N 0.000 description 2
- CLDALAIQIBJASM-RXAIFQJESA-N CC[C@](C(C=C1N2CC3=C(CN4C[C@H](CO)OCC4)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CN4C[C@H](CO)OCC4)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O CLDALAIQIBJASM-RXAIFQJESA-N 0.000 description 2
- JTLMQJRIYJKEHT-NRFANRHFSA-N CC[C@](C(C=C1N2CC3=C(CN=[N+]=[N-])C(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CN=[N+]=[N-])C(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O JTLMQJRIYJKEHT-NRFANRHFSA-N 0.000 description 2
- JOICFTKGKVLDLN-QHCPKHFHSA-N CC[C@](C(C=C1N2CC3=C(CNC(NC)=O)C(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CNC(NC)=O)C(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O JOICFTKGKVLDLN-QHCPKHFHSA-N 0.000 description 2
- VGAPEKFEIJGPJH-DEOSSOPVSA-N CC[C@](C(C=C1N2CC3=C(CNC(NC)=O)C(C=C(C(F)=C4)OC)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CNC(NC)=O)C(C=C(C(F)=C4)OC)=C4N=C13)=C(CO1)C2=O)(C1=O)O VGAPEKFEIJGPJH-DEOSSOPVSA-N 0.000 description 2
- AROZJFZRKWAMTQ-DEOSSOPVSA-N CC[C@](C(C=C1N2CC3=C(CNC(NC)=O)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CNC(NC)=O)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O AROZJFZRKWAMTQ-DEOSSOPVSA-N 0.000 description 2
- SWSRWKFDIPHWLJ-QHCPKHFHSA-N CC[C@](C(C=C1N2CC3=C(CNC(NC)=S)C(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CNC(NC)=S)C(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O SWSRWKFDIPHWLJ-QHCPKHFHSA-N 0.000 description 2
- GAZPJFXKYDTAHT-PMERELPUSA-N CC[C@](C(C=C1N2CC3=C(CNC(NCC(C=C4)=CC=C4N)=O)C(C=C(C(F)=C4)OC)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CNC(NCC(C=C4)=CC=C4N)=O)C(C=C(C(F)=C4)OC)=C4N=C13)=C(CO1)C2=O)(C1=O)O GAZPJFXKYDTAHT-PMERELPUSA-N 0.000 description 2
- UBWSFZPEPDGLHH-PMERELPUSA-N CC[C@](C(C=C1N2CC3=C(CNC(NCC(C=C4)=CC=C4N)=O)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CNC(NCC(C=C4)=CC=C4N)=O)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O UBWSFZPEPDGLHH-PMERELPUSA-N 0.000 description 2
- XKPGJGGSJGMUQR-VWLOTQADSA-N CC[C@](C(C=C1N2CC3=C(CNC(NCCO)=O)C(C=C(C(F)=C4)OC)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CNC(NCCO)=O)C(C=C(C(F)=C4)OC)=C4N=C13)=C(CO1)C2=O)(C1=O)O XKPGJGGSJGMUQR-VWLOTQADSA-N 0.000 description 2
- KRSPCZFYSZZPMK-VWLOTQADSA-N CC[C@](C(C=C1N2CC3=C(CNC(NCCO)=O)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CNC(NCCO)=O)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O KRSPCZFYSZZPMK-VWLOTQADSA-N 0.000 description 2
- ATZINVXQHLTMHK-DEOSSOPVSA-N CC[C@](C(C=C1N2CC3=C(CNC(OC)=O)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CNC(OC)=O)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O ATZINVXQHLTMHK-DEOSSOPVSA-N 0.000 description 2
- LSZGOVQPYWLUFM-DEOSSOPVSA-N CC[C@](C(C=C1N2CC3=C(CNC([O-])=[S+]CCO)C(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CNC([O-])=[S+]CCO)C(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O LSZGOVQPYWLUFM-DEOSSOPVSA-N 0.000 description 2
- FNTFDLIOZQBLDW-QFIPXVFZSA-N CC[C@](C(C=C1N2CC3=C(CNS(C)(=O)=O)C(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CNS(C)(=O)=O)C(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O FNTFDLIOZQBLDW-QFIPXVFZSA-N 0.000 description 2
- LLDHGUXKWZFCBN-QHCPKHFHSA-N CC[C@](C(C=C1N2CC3=C(CNS(C)(=O)=O)C(C=C(C(F)=C4)OC)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CNS(C)(=O)=O)C(C=C(C(F)=C4)OC)=C4N=C13)=C(CO1)C2=O)(C1=O)O LLDHGUXKWZFCBN-QHCPKHFHSA-N 0.000 description 2
- DMGLZCKUYPGBPY-NDEPHWFRSA-N CC[C@](C(C=C1N2CC3=C(CNS(C4=CC=CC=C4)(=O)=O)C(C=C(C(F)=C4)OC)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CNS(C4=CC=CC=C4)(=O)=O)C(C=C(C(F)=C4)OC)=C4N=C13)=C(CO1)C2=O)(C1=O)O DMGLZCKUYPGBPY-NDEPHWFRSA-N 0.000 description 2
- LAMVSFYCEZZVOC-NDEPHWFRSA-N CC[C@](C(C=C1N2CC3=C(CNS(C4=CC=CC=C4)(=O)=O)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CNS(C4=CC=CC=C4)(=O)=O)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O LAMVSFYCEZZVOC-NDEPHWFRSA-N 0.000 description 2
- MMVSKEUHTITNBY-DEOSSOPVSA-N CC[C@](C(C=C1N2CC3=C(CNS(CCO)(=O)=O)C(C=C(C(F)=C4)OC)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CNS(CCO)(=O)=O)C(C=C(C(F)=C4)OC)=C4N=C13)=C(CO1)C2=O)(C1=O)O MMVSKEUHTITNBY-DEOSSOPVSA-N 0.000 description 2
- DGKYSVLKARWCOT-DEOSSOPVSA-N CC[C@](C(C=C1N2CC3=C(CNS(CCO)(=O)=O)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CNS(CCO)(=O)=O)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O DGKYSVLKARWCOT-DEOSSOPVSA-N 0.000 description 2
- XFUUCFOAMYAEIC-QFIPXVFZSA-N CC[C@](C(C=C1N2CC3=C(COC(N)=O)C(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(COC(N)=O)C(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O XFUUCFOAMYAEIC-QFIPXVFZSA-N 0.000 description 2
- ZXKKWXDYEMUEQP-QFIPXVFZSA-N CC[C@](C(C=C1N2CC3=C(COC)C(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(COC)C(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O ZXKKWXDYEMUEQP-QFIPXVFZSA-N 0.000 description 2
- RIPKQXLKOSMRBZ-FQEVSTJZSA-N CC[C@](C(C=C1N2CC3=CC(C=C(C(C(F)(F)F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=CC(C=C(C(C(F)(F)F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O RIPKQXLKOSMRBZ-FQEVSTJZSA-N 0.000 description 2
- GMUFPUIYULJWPT-QFIPXVFZSA-N CC[C@](C(C=C1N2CC3=CC(C=C(C(F)=C4)NC(C)=O)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=CC(C=C(C(F)=C4)NC(C)=O)=C4N=C13)=C(CO1)C2=O)(C1=O)O GMUFPUIYULJWPT-QFIPXVFZSA-N 0.000 description 2
- DCERHCFNWRGHLK-UHFFFAOYSA-N C[Si](C)C Chemical compound C[Si](C)C DCERHCFNWRGHLK-UHFFFAOYSA-N 0.000 description 2
- KLWPJMFMVPTNCC-UHFFFAOYSA-N Camptothecin Natural products CCC1(O)C(=O)OCC2=C1C=C3C4Nc5ccccc5C=C4CN3C2=O KLWPJMFMVPTNCC-UHFFFAOYSA-N 0.000 description 2
- 201000009030 Carcinoma Diseases 0.000 description 2
- 206010009944 Colon cancer Diseases 0.000 description 2
- 102000004190 Enzymes Human genes 0.000 description 2
- 108090000790 Enzymes Proteins 0.000 description 2
- KAJAOGBVWCYGHZ-JTQLQIEISA-N Gly-Gly-Phe Chemical compound [NH3+]CC(=O)NCC(=O)N[C@H](C([O-])=O)CC1=CC=CC=C1 KAJAOGBVWCYGHZ-JTQLQIEISA-N 0.000 description 2
- ZWZWYGMENQVNFU-UHFFFAOYSA-N Glycerophosphorylserin Natural products OC(=O)C(N)COP(O)(=O)OCC(O)CO ZWZWYGMENQVNFU-UHFFFAOYSA-N 0.000 description 2
- DHMQDGOQFOQNFH-UHFFFAOYSA-N Glycine Chemical compound NCC(O)=O DHMQDGOQFOQNFH-UHFFFAOYSA-N 0.000 description 2
- 229920000084 Gum arabic Polymers 0.000 description 2
- 101001103036 Homo sapiens Nuclear receptor ROR-alpha Proteins 0.000 description 2
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 2
- 229930194542 Keto Natural products 0.000 description 2
- COLNVLDHVKWLRT-QMMMGPOBSA-N L-phenylalanine Chemical compound OC(=O)[C@@H](N)CC1=CC=CC=C1 COLNVLDHVKWLRT-QMMMGPOBSA-N 0.000 description 2
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 2
- 235000010643 Leucaena leucocephala Nutrition 0.000 description 2
- 240000007472 Leucaena leucocephala Species 0.000 description 2
- 206010025323 Lymphomas Diseases 0.000 description 2
- 239000004472 Lysine Substances 0.000 description 2
- CSNNHWWHGAXBCP-UHFFFAOYSA-L Magnesium sulfate Chemical compound [Mg+2].[O-][S+2]([O-])([O-])[O-] CSNNHWWHGAXBCP-UHFFFAOYSA-L 0.000 description 2
- PEEHTFAAVSWFBL-UHFFFAOYSA-N Maleimide Chemical compound O=C1NC(=O)C=C1 PEEHTFAAVSWFBL-UHFFFAOYSA-N 0.000 description 2
- 241000124008 Mammalia Species 0.000 description 2
- 108010008707 Mucin-1 Proteins 0.000 description 2
- 102000007298 Mucin-1 Human genes 0.000 description 2
- 241001529936 Murinae Species 0.000 description 2
- GRYLNZFGIOXLOG-UHFFFAOYSA-N Nitric acid Chemical compound O[N+]([O-])=O GRYLNZFGIOXLOG-UHFFFAOYSA-N 0.000 description 2
- 206010033128 Ovarian cancer Diseases 0.000 description 2
- 206010061328 Ovarian epithelial cancer Diseases 0.000 description 2
- 206010061535 Ovarian neoplasm Diseases 0.000 description 2
- 108010002519 Prolactin Receptors Proteins 0.000 description 2
- 102100029000 Prolactin receptor Human genes 0.000 description 2
- NBBJYMSMWIIQGU-UHFFFAOYSA-N Propionic aldehyde Chemical compound CCC=O NBBJYMSMWIIQGU-UHFFFAOYSA-N 0.000 description 2
- 238000011579 SCID mouse model Methods 0.000 description 2
- 206010039491 Sarcoma Diseases 0.000 description 2
- PXIPVTKHYLBLMZ-UHFFFAOYSA-N Sodium azide Chemical compound [Na+].[N-]=[N+]=[N-] PXIPVTKHYLBLMZ-UHFFFAOYSA-N 0.000 description 2
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical compound [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 2
- WQDUMFSSJAZKTM-UHFFFAOYSA-N Sodium methoxide Chemical compound [Na+].[O-]C WQDUMFSSJAZKTM-UHFFFAOYSA-N 0.000 description 2
- 229920002472 Starch Polymers 0.000 description 2
- DKGAVHZHDRPRBM-UHFFFAOYSA-N Tert-Butanol Chemical compound CC(C)(C)O DKGAVHZHDRPRBM-UHFFFAOYSA-N 0.000 description 2
- 108060008539 Transglutaminase Proteins 0.000 description 2
- XSQUKJJJFZCRTK-UHFFFAOYSA-N Urea Chemical compound NC(N)=O XSQUKJJJFZCRTK-UHFFFAOYSA-N 0.000 description 2
- 238000010521 absorption reaction Methods 0.000 description 2
- 235000010489 acacia gum Nutrition 0.000 description 2
- 150000007513 acids Chemical class 0.000 description 2
- 125000002947 alkylene group Chemical group 0.000 description 2
- 150000001408 amides Chemical class 0.000 description 2
- 125000005001 aminoaryl group Chemical group 0.000 description 2
- MDFFNEOEWAXZRQ-UHFFFAOYSA-N aminyl Chemical compound [NH2] MDFFNEOEWAXZRQ-UHFFFAOYSA-N 0.000 description 2
- BFNBIHQBYMNNAN-UHFFFAOYSA-N ammonium sulfate Chemical compound N.N.OS(O)(=O)=O BFNBIHQBYMNNAN-UHFFFAOYSA-N 0.000 description 2
- 229910052921 ammonium sulfate Inorganic materials 0.000 description 2
- 150000008064 anhydrides Chemical class 0.000 description 2
- 229940009098 aspartate Drugs 0.000 description 2
- 239000012298 atmosphere Substances 0.000 description 2
- 239000011324 bead Substances 0.000 description 2
- CSKNSYBAZOQPLR-UHFFFAOYSA-N benzenesulfonyl chloride Chemical compound ClS(=O)(=O)C1=CC=CC=C1 CSKNSYBAZOQPLR-UHFFFAOYSA-N 0.000 description 2
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid Chemical compound OC(=O)C1=CC=CC=C1 WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 description 2
- 125000001584 benzyloxycarbonyl group Chemical group C(=O)(OCC1=CC=CC=C1)* 0.000 description 2
- 230000001588 bifunctional effect Effects 0.000 description 2
- 239000011230 binding agent Substances 0.000 description 2
- ACBQROXDOHKANW-UHFFFAOYSA-N bis(4-nitrophenyl) carbonate Chemical compound C1=CC([N+](=O)[O-])=CC=C1OC(=O)OC1=CC=C([N+]([O-])=O)C=C1 ACBQROXDOHKANW-UHFFFAOYSA-N 0.000 description 2
- 201000008275 breast carcinoma Diseases 0.000 description 2
- 229910000024 caesium carbonate Inorganic materials 0.000 description 2
- 229910000389 calcium phosphate Inorganic materials 0.000 description 2
- 235000011010 calcium phosphates Nutrition 0.000 description 2
- OSGAYBCDTDRGGQ-UHFFFAOYSA-L calcium sulfate Chemical compound [Ca+2].[O-]S([O-])(=O)=O OSGAYBCDTDRGGQ-UHFFFAOYSA-L 0.000 description 2
- 229940127093 camptothecin Drugs 0.000 description 2
- 125000004432 carbon atom Chemical group C* 0.000 description 2
- 239000000969 carrier Substances 0.000 description 2
- 230000015556 catabolic process Effects 0.000 description 2
- 230000010261 cell growth Effects 0.000 description 2
- 238000012054 celltiter-glo Methods 0.000 description 2
- 238000005119 centrifugation Methods 0.000 description 2
- 238000012512 characterization method Methods 0.000 description 2
- 125000003636 chemical group Chemical group 0.000 description 2
- 238000001311 chemical methods and process Methods 0.000 description 2
- 239000003795 chemical substances by application Substances 0.000 description 2
- 150000001805 chlorine compounds Chemical class 0.000 description 2
- WRJWRGBVPUUDLA-UHFFFAOYSA-N chlorosulfonyl isocyanate Chemical compound ClS(=O)(=O)N=C=O WRJWRGBVPUUDLA-UHFFFAOYSA-N 0.000 description 2
- 229910052681 coesite Inorganic materials 0.000 description 2
- 238000004440 column chromatography Methods 0.000 description 2
- 230000000295 complement effect Effects 0.000 description 2
- 229910052906 cristobalite Inorganic materials 0.000 description 2
- DMEGYFMYUHOHGS-UHFFFAOYSA-N cycloheptane Chemical compound C1CCCCCC1 DMEGYFMYUHOHGS-UHFFFAOYSA-N 0.000 description 2
- 230000001472 cytotoxic effect Effects 0.000 description 2
- 238000007405 data analysis Methods 0.000 description 2
- 230000034994 death Effects 0.000 description 2
- 231100000517 death Toxicity 0.000 description 2
- 238000006731 degradation reaction Methods 0.000 description 2
- 230000001419 dependent effect Effects 0.000 description 2
- NKLCNNUWBJBICK-UHFFFAOYSA-N dess–martin periodinane Chemical compound C1=CC=C2I(OC(=O)C)(OC(C)=O)(OC(C)=O)OC(=O)C2=C1 NKLCNNUWBJBICK-UHFFFAOYSA-N 0.000 description 2
- 238000011161 development Methods 0.000 description 2
- 201000010099 disease Diseases 0.000 description 2
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 2
- VSJKWCGYPAHWDS-UHFFFAOYSA-N dl-camptothecin Natural products C1=CC=C2C=C(CN3C4=CC5=C(C3=O)COC(=O)C5(O)CC)C4=NC2=C1 VSJKWCGYPAHWDS-UHFFFAOYSA-N 0.000 description 2
- 239000002552 dosage form Substances 0.000 description 2
- 229940088598 enzyme Drugs 0.000 description 2
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 2
- 238000011156 evaluation Methods 0.000 description 2
- 239000012530 fluid Substances 0.000 description 2
- 238000001943 fluorescence-activated cell sorting Methods 0.000 description 2
- OVBPIULPVIDEAO-LBPRGKRZSA-N folic acid Chemical compound C=1N=C2NC(N)=NC(=O)C2=NC=1CNC1=CC=C(C(=O)N[C@@H](CCC(O)=O)C(O)=O)C=C1 OVBPIULPVIDEAO-LBPRGKRZSA-N 0.000 description 2
- 125000002485 formyl group Chemical group [H]C(*)=O 0.000 description 2
- WHUUTDBJXJRKMK-VKHMYHEASA-L glutamate group Chemical group N[C@@H](CCC(=O)[O-])C(=O)[O-] WHUUTDBJXJRKMK-VKHMYHEASA-L 0.000 description 2
- ZDXPYRJPNDTMRX-UHFFFAOYSA-N glutamine Natural products OC(=O)C(N)CCC(N)=O ZDXPYRJPNDTMRX-UHFFFAOYSA-N 0.000 description 2
- 230000012010 growth Effects 0.000 description 2
- 229940093915 gynecological organic acid Drugs 0.000 description 2
- 125000005843 halogen group Chemical group 0.000 description 2
- BXWNKGSJHAJOGX-UHFFFAOYSA-N hexadecan-1-ol Chemical compound CCCCCCCCCCCCCCCCO BXWNKGSJHAJOGX-UHFFFAOYSA-N 0.000 description 2
- 239000004312 hexamethylene tetramine Substances 0.000 description 2
- 235000010299 hexamethylene tetramine Nutrition 0.000 description 2
- 239000013628 high molecular weight specie Substances 0.000 description 2
- 210000004408 hybridoma Anatomy 0.000 description 2
- 150000002430 hydrocarbons Chemical group 0.000 description 2
- 230000005917 in vivo anti-tumor Effects 0.000 description 2
- 239000004615 ingredient Substances 0.000 description 2
- 229940102223 injectable solution Drugs 0.000 description 2
- 229940102213 injectable suspension Drugs 0.000 description 2
- 239000007924 injection Substances 0.000 description 2
- 238000002347 injection Methods 0.000 description 2
- 229910052500 inorganic mineral Inorganic materials 0.000 description 2
- 238000001990 intravenous administration Methods 0.000 description 2
- 229960004768 irinotecan Drugs 0.000 description 2
- GURKHSYORGJETM-WAQYZQTGSA-N irinotecan hydrochloride (anhydrous) Chemical compound Cl.C1=C2C(CC)=C3CN(C(C4=C([C@@](C(=O)OC4)(O)CC)C=4)=O)C=4C3=NC2=CC=C1OC(=O)N(CC1)CCC1N1CCCCC1 GURKHSYORGJETM-WAQYZQTGSA-N 0.000 description 2
- 239000012948 isocyanate Substances 0.000 description 2
- 150000002513 isocyanates Chemical class 0.000 description 2
- 238000001155 isoelectric focusing Methods 0.000 description 2
- 239000008101 lactose Substances 0.000 description 2
- 239000000787 lecithin Substances 0.000 description 2
- 235000010445 lecithin Nutrition 0.000 description 2
- 229940067606 lecithin Drugs 0.000 description 2
- 239000007788 liquid Substances 0.000 description 2
- 239000013627 low molecular weight specie Substances 0.000 description 2
- 238000004020 luminiscence type Methods 0.000 description 2
- 230000002132 lysosomal effect Effects 0.000 description 2
- 210000003712 lysosome Anatomy 0.000 description 2
- 230000001868 lysosomic effect Effects 0.000 description 2
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 description 2
- 108010082117 matrigel Proteins 0.000 description 2
- 239000011159 matrix material Substances 0.000 description 2
- 230000001404 mediated effect Effects 0.000 description 2
- TWXDDNPPQUTEOV-FVGYRXGTSA-N methamphetamine hydrochloride Chemical compound Cl.CN[C@@H](C)CC1=CC=CC=C1 TWXDDNPPQUTEOV-FVGYRXGTSA-N 0.000 description 2
- 229940098779 methanesulfonic acid Drugs 0.000 description 2
- NSPJNIDYTSSIIY-UHFFFAOYSA-N methoxy(methoxymethoxy)methane Chemical compound COCOCOC NSPJNIDYTSSIIY-UHFFFAOYSA-N 0.000 description 2
- 235000010755 mineral Nutrition 0.000 description 2
- 239000011707 mineral Substances 0.000 description 2
- 239000002480 mineral oil Substances 0.000 description 2
- 235000010446 mineral oil Nutrition 0.000 description 2
- 230000004048 modification Effects 0.000 description 2
- 238000012986 modification Methods 0.000 description 2
- 239000001788 mono and diglycerides of fatty acids Substances 0.000 description 2
- HOGDNTQCSIKEEV-UHFFFAOYSA-N n'-hydroxybutanediamide Chemical compound NC(=O)CCC(=O)NO HOGDNTQCSIKEEV-UHFFFAOYSA-N 0.000 description 2
- XBXCNNQPRYLIDE-UHFFFAOYSA-M n-tert-butylcarbamate Chemical compound CC(C)(C)NC([O-])=O XBXCNNQPRYLIDE-UHFFFAOYSA-M 0.000 description 2
- 239000013642 negative control Substances 0.000 description 2
- 229910017604 nitric acid Inorganic materials 0.000 description 2
- 239000012299 nitrogen atmosphere Substances 0.000 description 2
- 208000002154 non-small cell lung carcinoma Diseases 0.000 description 2
- 239000000346 nonvolatile oil Substances 0.000 description 2
- 235000005985 organic acids Nutrition 0.000 description 2
- 150000007530 organic bases Chemical class 0.000 description 2
- 208000013371 ovarian adenocarcinoma Diseases 0.000 description 2
- 201000006588 ovary adenocarcinoma Diseases 0.000 description 2
- 239000008188 pellet Substances 0.000 description 2
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 2
- UYWQUFXKFGHYNT-UHFFFAOYSA-N phenylmethyl ester of formic acid Natural products O=COCC1=CC=CC=C1 UYWQUFXKFGHYNT-UHFFFAOYSA-N 0.000 description 2
- 235000021317 phosphate Nutrition 0.000 description 2
- XKJCHHZQLQNZHY-UHFFFAOYSA-N phthalimide Chemical compound C1=CC=C2C(=O)NC(=O)C2=C1 XKJCHHZQLQNZHY-UHFFFAOYSA-N 0.000 description 2
- 229910052697 platinum Inorganic materials 0.000 description 2
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 2
- 238000001556 precipitation Methods 0.000 description 2
- 230000002265 prevention Effects 0.000 description 2
- 102000004196 processed proteins & peptides Human genes 0.000 description 2
- QELSKZZBTMNZEB-UHFFFAOYSA-N propylparaben Chemical compound CCCOC(=O)C1=CC=C(O)C=C1 QELSKZZBTMNZEB-UHFFFAOYSA-N 0.000 description 2
- DVUZQONFTJKFPV-UHFFFAOYSA-N pyrrole-2,5-dithione Chemical compound S=C1NC(=S)C=C1 DVUZQONFTJKFPV-UHFFFAOYSA-N 0.000 description 2
- 238000004451 qualitative analysis Methods 0.000 description 2
- 238000010791 quenching Methods 0.000 description 2
- 238000011160 research Methods 0.000 description 2
- CVHZOJJKTDOEJC-UHFFFAOYSA-N saccharin Chemical compound C1=CC=C2C(=O)NS(=O)(=O)C2=C1 CVHZOJJKTDOEJC-UHFFFAOYSA-N 0.000 description 2
- 229940081974 saccharin Drugs 0.000 description 2
- 235000019204 saccharin Nutrition 0.000 description 2
- 239000000901 saccharin and its Na,K and Ca salt Substances 0.000 description 2
- ULRUOUDIQPERIJ-PQURJYPBSA-N sacituzumab govitecan Chemical compound N([C@@H](CCCCN)C(=O)NC1=CC=C(C=C1)COC(=O)O[C@]1(CC)C(=O)OCC2=C1C=C1N(C2=O)CC2=C(C3=CC(O)=CC=C3N=C21)CC)C(=O)COCC(=O)NCCOCCOCCOCCOCCOCCOCCOCCOCCN(N=N1)C=C1CNC(=O)C(CC1)CCC1CN1C(=O)CC(SC[C@H](N)C(O)=O)C1=O ULRUOUDIQPERIJ-PQURJYPBSA-N 0.000 description 2
- 229950000143 sacituzumab govitecan Drugs 0.000 description 2
- 229930195734 saturated hydrocarbon Natural products 0.000 description 2
- 238000000926 separation method Methods 0.000 description 2
- 239000002356 single layer Substances 0.000 description 2
- 239000001632 sodium acetate Substances 0.000 description 2
- 235000017281 sodium acetate Nutrition 0.000 description 2
- AKHNMLFCWUSKQB-UHFFFAOYSA-L sodium thiosulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=S AKHNMLFCWUSKQB-UHFFFAOYSA-L 0.000 description 2
- 239000001593 sorbitan monooleate Substances 0.000 description 2
- 235000011069 sorbitan monooleate Nutrition 0.000 description 2
- 229940035049 sorbitan monooleate Drugs 0.000 description 2
- 238000010561 standard procedure Methods 0.000 description 2
- 239000008107 starch Substances 0.000 description 2
- 235000019698 starch Nutrition 0.000 description 2
- 239000007858 starting material Substances 0.000 description 2
- 229910052682 stishovite Inorganic materials 0.000 description 2
- 239000011550 stock solution Substances 0.000 description 2
- YBBRCQOCSYXUOC-UHFFFAOYSA-N sulfuryl dichloride Chemical class ClS(Cl)(=O)=O YBBRCQOCSYXUOC-UHFFFAOYSA-N 0.000 description 2
- 239000000829 suppository Substances 0.000 description 2
- 230000004083 survival effect Effects 0.000 description 2
- 125000001981 tert-butyldimethylsilyl group Chemical group [H]C([H])([H])[Si]([H])(C([H])([H])[H])[*]C(C([H])([H])[H])(C([H])([H])[H])C([H])([H])[H] 0.000 description 2
- 125000005931 tert-butyloxycarbonyl group Chemical group [H]C([H])([H])C(OC(*)=O)(C([H])([H])[H])C([H])([H])[H] 0.000 description 2
- 125000001412 tetrahydropyranyl group Chemical group 0.000 description 2
- 125000002088 tosyl group Chemical group [H]C1=C([H])C(=C([H])C([H])=C1C([H])([H])[H])S(*)(=O)=O 0.000 description 2
- 102000003601 transglutaminase Human genes 0.000 description 2
- 230000032258 transport Effects 0.000 description 2
- 229940049679 trastuzumab deruxtecan Drugs 0.000 description 2
- QORWJWZARLRLPR-UHFFFAOYSA-H tricalcium bis(phosphate) Chemical compound [Ca+2].[Ca+2].[Ca+2].[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O QORWJWZARLRLPR-UHFFFAOYSA-H 0.000 description 2
- 229910052905 tridymite Inorganic materials 0.000 description 2
- 125000004044 trifluoroacetyl group Chemical group FC(C(=O)*)(F)F 0.000 description 2
- 125000000025 triisopropylsilyl group Chemical group C(C)(C)[Si](C(C)C)(C(C)C)* 0.000 description 2
- 125000000026 trimethylsilyl group Chemical group [H]C([H])([H])[Si]([*])(C([H])([H])[H])C([H])([H])[H] 0.000 description 2
- 208000029729 tumor suppressor gene on chromosome 11 Diseases 0.000 description 2
- 238000001946 ultra-performance liquid chromatography-mass spectrometry Methods 0.000 description 2
- 206010046766 uterine cancer Diseases 0.000 description 2
- 229910052720 vanadium Inorganic materials 0.000 description 2
- LEONUFNNVUYDNQ-UHFFFAOYSA-N vanadium atom Chemical compound [V] LEONUFNNVUYDNQ-UHFFFAOYSA-N 0.000 description 2
- 230000003442 weekly effect Effects 0.000 description 2
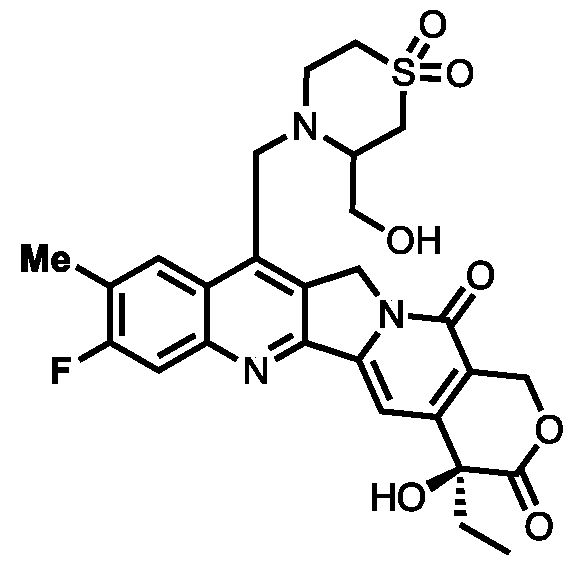
- CGBSWCOIPWWGHT-UHFFFAOYSA-N (1,1-dioxo-1,4-thiazinan-3-yl)methanol Chemical compound OCC1CS(=O)(=O)CCN1 CGBSWCOIPWWGHT-UHFFFAOYSA-N 0.000 description 1
- CRZIREHHSWTUQV-UHFFFAOYSA-N (2,3,5,6-tetrafluorophenyl) 3-[2-[2-[2-(2,5-dioxopyrrol-1-yl)ethoxy]ethoxy]ethoxy]propanoate Chemical compound FC1=CC(F)=C(F)C(OC(=O)CCOCCOCCOCCN2C(C=CC2=O)=O)=C1F CRZIREHHSWTUQV-UHFFFAOYSA-N 0.000 description 1
- QUGODPAQMQMGRN-UHFFFAOYSA-N (2,3-dinitrophenyl) hydrogen carbonate Chemical compound OC(=O)OC1=CC=CC([N+]([O-])=O)=C1[N+]([O-])=O QUGODPAQMQMGRN-UHFFFAOYSA-N 0.000 description 1
- TYKASZBHFXBROF-UHFFFAOYSA-N (2,5-dioxopyrrolidin-1-yl) 2-(2,5-dioxopyrrol-1-yl)acetate Chemical compound O=C1CCC(=O)N1OC(=O)CN1C(=O)C=CC1=O TYKASZBHFXBROF-UHFFFAOYSA-N 0.000 description 1
- YWPGJWLLWYHMEN-UHFFFAOYSA-N (2,5-dioxopyrrolidin-1-yl) 2-[[2-(9H-fluoren-9-ylmethoxycarbonylamino)acetyl]amino]acetate Chemical compound O=C(CNC(=O)OCC1c2ccccc2-c2ccccc12)NCC(=O)ON1C(=O)CCC1=O YWPGJWLLWYHMEN-UHFFFAOYSA-N 0.000 description 1
- FLCQLSRLQIPNLM-UHFFFAOYSA-N (2,5-dioxopyrrolidin-1-yl) 2-acetylsulfanylacetate Chemical compound CC(=O)SCC(=O)ON1C(=O)CCC1=O FLCQLSRLQIPNLM-UHFFFAOYSA-N 0.000 description 1
- LLXVXPPXELIDGQ-UHFFFAOYSA-N (2,5-dioxopyrrolidin-1-yl) 3-(2,5-dioxopyrrol-1-yl)benzoate Chemical compound C=1C=CC(N2C(C=CC2=O)=O)=CC=1C(=O)ON1C(=O)CCC1=O LLXVXPPXELIDGQ-UHFFFAOYSA-N 0.000 description 1
- JKHVDAUOODACDU-UHFFFAOYSA-N (2,5-dioxopyrrolidin-1-yl) 3-(2,5-dioxopyrrol-1-yl)propanoate Chemical compound O=C1CCC(=O)N1OC(=O)CCN1C(=O)C=CC1=O JKHVDAUOODACDU-UHFFFAOYSA-N 0.000 description 1
- IHVODYOQUSEYJJ-UHFFFAOYSA-N (2,5-dioxopyrrolidin-1-yl) 6-[[4-[(2,5-dioxopyrrol-1-yl)methyl]cyclohexanecarbonyl]amino]hexanoate Chemical compound O=C1CCC(=O)N1OC(=O)CCCCCNC(=O)C(CC1)CCC1CN1C(=O)C=CC1=O IHVODYOQUSEYJJ-UHFFFAOYSA-N 0.000 description 1
- BSPMWFRGZQDRIU-UHFFFAOYSA-N (2-amino-1h-imidazol-5-yl)methanol Chemical class NC1=NC(CO)=CN1 BSPMWFRGZQDRIU-UHFFFAOYSA-N 0.000 description 1
- UFGUUZVZUIXKQZ-DEOSSOPVSA-N (2S)-2-[[2-[[2-(9H-fluoren-9-ylmethoxycarbonylamino)acetyl]amino]acetyl]amino]-3-phenylpropanoic acid Chemical compound C1=CC=CC=2C3=CC=CC=C3C(C1=2)COC(=O)NCC(=O)NCC(=O)N[C@H](C(=O)O)CC1=CC=CC=C1 UFGUUZVZUIXKQZ-DEOSSOPVSA-N 0.000 description 1
- QVHJQCGUWFKTSE-YFKPBYRVSA-N (2s)-2-[(2-methylpropan-2-yl)oxycarbonylamino]propanoic acid Chemical compound OC(=O)[C@H](C)NC(=O)OC(C)(C)C QVHJQCGUWFKTSE-YFKPBYRVSA-N 0.000 description 1
- GAJBPZXIKZXTCG-VIFPVBQESA-N (2s)-2-amino-3-[4-(azidomethyl)phenyl]propanoic acid Chemical compound OC(=O)[C@@H](N)CC1=CC=C(CN=[N+]=[N-])C=C1 GAJBPZXIKZXTCG-VIFPVBQESA-N 0.000 description 1
- JPSHPWJJSVEEAX-OWPBQMJCSA-N (2s)-2-amino-4-fluoranylpentanedioic acid Chemical compound OC(=O)[C@@H](N)CC([18F])C(O)=O JPSHPWJJSVEEAX-OWPBQMJCSA-N 0.000 description 1
- ZXSBHXZKWRIEIA-JTQLQIEISA-N (2s)-3-(4-acetylphenyl)-2-azaniumylpropanoate Chemical compound CC(=O)C1=CC=C(C[C@H](N)C(O)=O)C=C1 ZXSBHXZKWRIEIA-JTQLQIEISA-N 0.000 description 1
- XTGWMTQKEVTLQD-UHFFFAOYSA-N (4,4-difluoropiperidin-3-yl)methanol Chemical compound OCC1CNCCC1(F)F XTGWMTQKEVTLQD-UHFFFAOYSA-N 0.000 description 1
- CAXRPEAFHWDFTD-UHFFFAOYSA-N (4-nitrophenyl)methanesulfonyl chloride Chemical compound [O-][N+](=O)C1=CC=C(CS(Cl)(=O)=O)C=C1 CAXRPEAFHWDFTD-UHFFFAOYSA-N 0.000 description 1
- WRIDQFICGBMAFQ-UHFFFAOYSA-N (E)-8-Octadecenoic acid Natural products CCCCCCCCCC=CCCCCCCC(O)=O WRIDQFICGBMAFQ-UHFFFAOYSA-N 0.000 description 1
- BYEAHWXPCBROCE-UHFFFAOYSA-N 1,1,1,3,3,3-hexafluoropropan-2-ol Chemical compound FC(F)(F)C(O)C(F)(F)F BYEAHWXPCBROCE-UHFFFAOYSA-N 0.000 description 1
- FUHCFUVCWLZEDQ-UHFFFAOYSA-N 1-(2,5-dioxopyrrolidin-1-yl)oxy-1-oxo-4-(pyridin-2-yldisulfanyl)butane-2-sulfonic acid Chemical compound O=C1CCC(=O)N1OC(=O)C(S(=O)(=O)O)CCSSC1=CC=CC=N1 FUHCFUVCWLZEDQ-UHFFFAOYSA-N 0.000 description 1
- TUSLTJFGSBMTHL-UHFFFAOYSA-N 1-(2-amino-4-fluoro-5-methoxyphenyl)-2-chloroethanone Chemical compound COC1=C(C=C(C(=C1)C(=O)CCl)N)F TUSLTJFGSBMTHL-UHFFFAOYSA-N 0.000 description 1
- OJQSISYVGFJJBY-UHFFFAOYSA-N 1-(4-isocyanatophenyl)pyrrole-2,5-dione Chemical compound C1=CC(N=C=O)=CC=C1N1C(=O)C=CC1=O OJQSISYVGFJJBY-UHFFFAOYSA-N 0.000 description 1
- GPAAEZIXSQCCES-UHFFFAOYSA-N 1-methoxy-2-(2-methoxyethoxymethoxymethoxy)ethane Chemical compound COCCOCOCOCCOC GPAAEZIXSQCCES-UHFFFAOYSA-N 0.000 description 1
- CBDLNOVOFXJEOB-UHFFFAOYSA-N 1-methoxy-4-(4-methoxyphenoxy)benzene Chemical compound C1=CC(OC)=CC=C1OC1=CC=C(OC)C=C1 CBDLNOVOFXJEOB-UHFFFAOYSA-N 0.000 description 1
- IXPNQXFRVYWDDI-UHFFFAOYSA-N 1-methyl-2,4-dioxo-1,3-diazinane-5-carboximidamide Chemical compound CN1CC(C(N)=N)C(=O)NC1=O IXPNQXFRVYWDDI-UHFFFAOYSA-N 0.000 description 1
- DGHHQBMTXTWTJV-BQAIUKQQSA-N 119413-54-6 Chemical compound Cl.C1=C(O)C(CN(C)C)=C2C=C(CN3C4=CC5=C(C3=O)COC(=O)[C@]5(O)CC)C4=NC2=C1 DGHHQBMTXTWTJV-BQAIUKQQSA-N 0.000 description 1
- YQTCQNIPQMJNTI-UHFFFAOYSA-N 2,2-dimethylpropan-1-one Chemical group CC(C)(C)[C]=O YQTCQNIPQMJNTI-UHFFFAOYSA-N 0.000 description 1
- 150000003923 2,5-pyrrolediones Chemical class 0.000 description 1
- UFCRQKWENZPCAD-UHFFFAOYSA-N 2-(2-aminophenyl)propanamide Chemical class NC(=O)C(C)C1=CC=CC=C1N UFCRQKWENZPCAD-UHFFFAOYSA-N 0.000 description 1
- QEVGZEDELICMKH-UHFFFAOYSA-L 2-(carboxylatomethoxy)acetate Chemical compound [O-]C(=O)COCC([O-])=O QEVGZEDELICMKH-UHFFFAOYSA-L 0.000 description 1
- IMSODMZESSGVBE-UHFFFAOYSA-N 2-Oxazoline Chemical group C1CN=CO1 IMSODMZESSGVBE-UHFFFAOYSA-N 0.000 description 1
- OWDQCSBZQVISPN-UHFFFAOYSA-N 2-[(2,5-dioxopyrrolidin-1-yl)amino]-4-(2-iodoacetyl)benzoic acid Chemical compound OC(=O)C1=CC=C(C(=O)CI)C=C1NN1C(=O)CCC1=O OWDQCSBZQVISPN-UHFFFAOYSA-N 0.000 description 1
- VRPJIFMKZZEXLR-UHFFFAOYSA-N 2-[(2-methylpropan-2-yl)oxycarbonylamino]acetic acid Chemical compound CC(C)(C)OC(=O)NCC(O)=O VRPJIFMKZZEXLR-UHFFFAOYSA-N 0.000 description 1
- PAYROHWFGZADBR-UHFFFAOYSA-N 2-[[4-amino-5-(5-iodo-4-methoxy-2-propan-2-ylphenoxy)pyrimidin-2-yl]amino]propane-1,3-diol Chemical compound C1=C(I)C(OC)=CC(C(C)C)=C1OC1=CN=C(NC(CO)CO)N=C1N PAYROHWFGZADBR-UHFFFAOYSA-N 0.000 description 1
- BGFTWECWAICPDG-UHFFFAOYSA-N 2-[bis(4-chlorophenyl)methyl]-4-n-[3-[bis(4-chlorophenyl)methyl]-4-(dimethylamino)phenyl]-1-n,1-n-dimethylbenzene-1,4-diamine Chemical compound C1=C(C(C=2C=CC(Cl)=CC=2)C=2C=CC(Cl)=CC=2)C(N(C)C)=CC=C1NC(C=1)=CC=C(N(C)C)C=1C(C=1C=CC(Cl)=CC=1)C1=CC=C(Cl)C=C1 BGFTWECWAICPDG-UHFFFAOYSA-N 0.000 description 1
- OGMADIBCHLQMIP-UHFFFAOYSA-N 2-aminoethanethiol;hydron;chloride Chemical compound Cl.NCCS OGMADIBCHLQMIP-UHFFFAOYSA-N 0.000 description 1
- DJQYYYCQOZMCRC-UHFFFAOYSA-N 2-aminopropane-1,3-dithiol Chemical group SCC(N)CS DJQYYYCQOZMCRC-UHFFFAOYSA-N 0.000 description 1
- IKCLCGXPQILATA-UHFFFAOYSA-N 2-chlorobenzoic acid Chemical class OC(=O)C1=CC=CC=C1Cl IKCLCGXPQILATA-UHFFFAOYSA-N 0.000 description 1
- 125000004493 2-methylbut-1-yl group Chemical group CC(C*)CC 0.000 description 1
- BAAIBSCYAOWMPE-UHFFFAOYSA-N 2-oxa-5-azabicyclo[2.2.1]heptan-4-ylmethanol Chemical compound OCC12COC(CN1)C2 BAAIBSCYAOWMPE-UHFFFAOYSA-N 0.000 description 1
- LQJBNNIYVWPHFW-UHFFFAOYSA-N 20:1omega9c fatty acid Natural products CCCCCCCCCCC=CCCCCCCCC(O)=O LQJBNNIYVWPHFW-UHFFFAOYSA-N 0.000 description 1
- WFOVEDJTASPCIR-UHFFFAOYSA-N 3-[(4-methyl-5-pyridin-4-yl-1,2,4-triazol-3-yl)methylamino]-n-[[2-(trifluoromethyl)phenyl]methyl]benzamide Chemical compound N=1N=C(C=2C=CN=CC=2)N(C)C=1CNC(C=1)=CC=CC=1C(=O)NCC1=CC=CC=C1C(F)(F)F WFOVEDJTASPCIR-UHFFFAOYSA-N 0.000 description 1
- MWYYNBYTVFKXSD-UHFFFAOYSA-N 3-bromo-4-(trifluoromethyl)benzaldehyde Chemical compound FC(F)(F)C1=CC=C(C=O)C=C1Br MWYYNBYTVFKXSD-UHFFFAOYSA-N 0.000 description 1
- FAHZIKXYYRGSHF-UHFFFAOYSA-N 3-bromo-4-fluorobenzaldehyde Chemical compound FC1=CC=C(C=O)C=C1Br FAHZIKXYYRGSHF-UHFFFAOYSA-N 0.000 description 1
- LJWAPDSCYTZUJU-UHFFFAOYSA-N 3-fluoro-4-methoxyaniline Chemical compound COC1=CC=C(N)C=C1F LJWAPDSCYTZUJU-UHFFFAOYSA-N 0.000 description 1
- OXGJKCALURPRCN-UHFFFAOYSA-N 3-methoxypropanal Chemical compound COCCC=O OXGJKCALURPRCN-UHFFFAOYSA-N 0.000 description 1
- BMTZEAOGFDXDAD-UHFFFAOYSA-M 4-(4,6-dimethoxy-1,3,5-triazin-2-yl)-4-methylmorpholin-4-ium;chloride Chemical compound [Cl-].COC1=NC(OC)=NC([N+]2(C)CCOCC2)=N1 BMTZEAOGFDXDAD-UHFFFAOYSA-M 0.000 description 1
- LQILVUYCDHSGEU-UHFFFAOYSA-N 4-[(2,5-dioxopyrrol-1-yl)methyl]cyclohexane-1-carboxylic acid Chemical compound C1CC(C(=O)O)CCC1CN1C(=O)C=CC1=O LQILVUYCDHSGEU-UHFFFAOYSA-N 0.000 description 1
- WCVPFJVXEXJFLB-UHFFFAOYSA-N 4-aminobutanamide Chemical class NCCCC(N)=O WCVPFJVXEXJFLB-UHFFFAOYSA-N 0.000 description 1
- PXACTUVBBMDKRW-UHFFFAOYSA-N 4-bromobenzenesulfonic acid Chemical compound OS(=O)(=O)C1=CC=C(Br)C=C1 PXACTUVBBMDKRW-UHFFFAOYSA-N 0.000 description 1
- GSDQYSSLIKJJOG-UHFFFAOYSA-N 4-chloro-2-(3-chloroanilino)benzoic acid Chemical compound OC(=O)C1=CC=C(Cl)C=C1NC1=CC=CC(Cl)=C1 GSDQYSSLIKJJOG-UHFFFAOYSA-N 0.000 description 1
- MZJKOAWTWHFDFV-UHFFFAOYSA-N 5,6-dibromopyridazine-3,4-dione Chemical compound BrC1=C(Br)C(=O)C(=O)N=N1 MZJKOAWTWHFDFV-UHFFFAOYSA-N 0.000 description 1
- UVWDWKCRJYWAGD-UHFFFAOYSA-N 5-bromo-2-nitro-4-(trifluoromethyl)benzaldehyde Chemical compound [O-][N+](C(C=C1C(F)(F)F)=C(C=O)C=C1Br)=O UVWDWKCRJYWAGD-UHFFFAOYSA-N 0.000 description 1
- LQFBETPMHZUCCK-UHFFFAOYSA-N 5-bromo-4-fluoro-2-nitrobenzaldehyde Chemical compound [O-][N+](=O)C1=CC(F)=C(Br)C=C1C=O LQFBETPMHZUCCK-UHFFFAOYSA-N 0.000 description 1
- IJRKLHTZAIFUTB-UHFFFAOYSA-N 5-nitro-2-(2-phenylethylamino)benzoic acid Chemical compound OC(=O)C1=CC([N+]([O-])=O)=CC=C1NCCC1=CC=CC=C1 IJRKLHTZAIFUTB-UHFFFAOYSA-N 0.000 description 1
- WXNSCLIZKHLNSG-MCZRLCSDSA-N 6-(2,5-dioxopyrrol-1-yl)-N-[2-[[2-[[(2S)-1-[[2-[[2-[[(10S,23S)-10-ethyl-18-fluoro-10-hydroxy-19-methyl-5,9-dioxo-8-oxa-4,15-diazahexacyclo[14.7.1.02,14.04,13.06,11.020,24]tetracosa-1,6(11),12,14,16,18,20(24)-heptaen-23-yl]amino]-2-oxoethoxy]methylamino]-2-oxoethyl]amino]-1-oxo-3-phenylpropan-2-yl]amino]-2-oxoethyl]amino]-2-oxoethyl]hexanamide Chemical compound CC[C@@]1(O)C(=O)OCC2=C1C=C1N(CC3=C1N=C1C=C(F)C(C)=C4CC[C@H](NC(=O)COCNC(=O)CNC(=O)[C@H](CC5=CC=CC=C5)NC(=O)CNC(=O)CNC(=O)CCCCCN5C(=O)C=CC5=O)C3=C14)C2=O WXNSCLIZKHLNSG-MCZRLCSDSA-N 0.000 description 1
- HBEDKBRARKFPIC-UHFFFAOYSA-N 6-(2,5-dioxopyrrol-1-yl)hexanoic acid;1-hydroxypyrrolidine-2,5-dione Chemical compound ON1C(=O)CCC1=O.OC(=O)CCCCCN1C(=O)C=CC1=O HBEDKBRARKFPIC-UHFFFAOYSA-N 0.000 description 1
- BUBVVQHRVJCXFR-UHFFFAOYSA-N 7-azabicyclo[2.2.1]heptan-4-ylmethanol Chemical compound C1CC2CCC1(CO)N2 BUBVVQHRVJCXFR-UHFFFAOYSA-N 0.000 description 1
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical compound [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 1
- IRBAWVGZNJIROV-SFHVURJKSA-N 9-(2-cyclopropylethynyl)-2-[[(2s)-1,4-dioxan-2-yl]methoxy]-6,7-dihydropyrimido[6,1-a]isoquinolin-4-one Chemical compound C1=C2C3=CC=C(C#CC4CC4)C=C3CCN2C(=O)N=C1OC[C@@H]1COCCO1 IRBAWVGZNJIROV-SFHVURJKSA-N 0.000 description 1
- QSBYPNXLFMSGKH-UHFFFAOYSA-N 9-Heptadecensaeure Natural products CCCCCCCC=CCCCCCCCC(O)=O QSBYPNXLFMSGKH-UHFFFAOYSA-N 0.000 description 1
- 102100031585 ADP-ribosyl cyclase/cyclic ADP-ribose hydrolase 1 Human genes 0.000 description 1
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 description 1
- 241000251468 Actinopterygii Species 0.000 description 1
- 239000012099 Alexa Fluor family Substances 0.000 description 1
- 239000005995 Aluminium silicate Substances 0.000 description 1
- NLXLAEXVIDQMFP-UHFFFAOYSA-N Ammonia chloride Chemical compound [NH4+].[Cl-] NLXLAEXVIDQMFP-UHFFFAOYSA-N 0.000 description 1
- QGZKDVFQNNGYKY-UHFFFAOYSA-O Ammonium Chemical compound [NH4+] QGZKDVFQNNGYKY-UHFFFAOYSA-O 0.000 description 1
- VHUUQVKOLVNVRT-UHFFFAOYSA-N Ammonium hydroxide Chemical compound [NH4+].[OH-] VHUUQVKOLVNVRT-UHFFFAOYSA-N 0.000 description 1
- 102000006306 Antigen Receptors Human genes 0.000 description 1
- 108010083359 Antigen Receptors Proteins 0.000 description 1
- 101100330723 Arabidopsis thaliana DAR2 gene Proteins 0.000 description 1
- 101100330725 Arabidopsis thaliana DAR4 gene Proteins 0.000 description 1
- 101100330727 Arabidopsis thaliana DAR6 gene Proteins 0.000 description 1
- 102000030431 Asparaginyl endopeptidase Human genes 0.000 description 1
- 102000006942 B-Cell Maturation Antigen Human genes 0.000 description 1
- 108010008014 B-Cell Maturation Antigen Proteins 0.000 description 1
- 102100038080 B-cell receptor CD22 Human genes 0.000 description 1
- 102100024222 B-lymphocyte antigen CD19 Human genes 0.000 description 1
- 102100022005 B-lymphocyte antigen CD20 Human genes 0.000 description 1
- 108010074708 B7-H1 Antigen Proteins 0.000 description 1
- 238000000035 BCA protein assay Methods 0.000 description 1
- 229910015844 BCl3 Inorganic materials 0.000 description 1
- IYHHRZBKXXKDDY-UHFFFAOYSA-N BI-605906 Chemical compound N=1C=2SC(C(N)=O)=C(N)C=2C(C(F)(F)CC)=CC=1N1CCC(S(C)(=O)=O)CC1 IYHHRZBKXXKDDY-UHFFFAOYSA-N 0.000 description 1
- 241000599985 Beijerinckia mobilis Species 0.000 description 1
- 239000005711 Benzoic acid Substances 0.000 description 1
- BVKZGUZCCUSVTD-UHFFFAOYSA-M Bicarbonate Chemical class OC([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-M 0.000 description 1
- 108010051479 Bombesin Proteins 0.000 description 1
- 229910014263 BrF3 Inorganic materials 0.000 description 1
- 208000003174 Brain Neoplasms Diseases 0.000 description 1
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical compound [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 1
- FERIUCNNQQJTOY-UHFFFAOYSA-M Butyrate Chemical compound CCCC([O-])=O FERIUCNNQQJTOY-UHFFFAOYSA-M 0.000 description 1
- 102100031650 C-X-C chemokine receptor type 4 Human genes 0.000 description 1
- 102100031658 C-X-C chemokine receptor type 5 Human genes 0.000 description 1
- 102100026094 C-type lectin domain family 12 member A Human genes 0.000 description 1
- 101710188619 C-type lectin domain family 12 member A Proteins 0.000 description 1
- BGGALFIXXQOTPY-NRFANRHFSA-N C1(=C(C2=C(C=C1)N(C(C#N)=C2)C[C@@H](N1CCN(CC1)S(=O)(=O)C)C)C)CN1CCC(CC1)NC1=NC(=NC2=C1C=C(S2)CC(F)(F)F)NC Chemical compound C1(=C(C2=C(C=C1)N(C(C#N)=C2)C[C@@H](N1CCN(CC1)S(=O)(=O)C)C)C)CN1CCC(CC1)NC1=NC(=NC2=C1C=C(S2)CC(F)(F)F)NC BGGALFIXXQOTPY-NRFANRHFSA-N 0.000 description 1
- VIMRBMFQCXMCKY-QFIPXVFZSA-N C=12C3=NC4=C(C=C(C(=C4)F)C)C(=C3CN2C(=O)C2=C(C=1)[C@@](C(=O)OC2)(O)CC)CCl Chemical compound C=12C3=NC4=C(C=C(C(=C4)F)C)C(=C3CN2C(=O)C2=C(C=1)[C@@](C(=O)OC2)(O)CC)CCl VIMRBMFQCXMCKY-QFIPXVFZSA-N 0.000 description 1
- QTGYMFHSEPDEQO-QFIPXVFZSA-N C=12C=C3C4=NC5=C(C(=C4CN3C(=O)C=1COC(=O)[C@]2(O)CC)CN)C=C(C(F)=C5)C Chemical compound C=12C=C3C4=NC5=C(C(=C4CN3C(=O)C=1COC(=O)[C@]2(O)CC)CN)C=C(C(F)=C5)C QTGYMFHSEPDEQO-QFIPXVFZSA-N 0.000 description 1
- FKALGRODVIEPOY-UHFFFAOYSA-N CC(C)(C)OC(NC(C(OC)=C1)=CC(C=O)=C1N)=O Chemical compound CC(C)(C)OC(NC(C(OC)=C1)=CC(C=O)=C1N)=O FKALGRODVIEPOY-UHFFFAOYSA-N 0.000 description 1
- IIMZECNSZRJPKA-UHFFFAOYSA-N CC(C)(C)OC(NC(C=C(C=O)C(N)=C1)=C1F)=O Chemical compound CC(C)(C)OC(NC(C=C(C=O)C(N)=C1)=C1F)=O IIMZECNSZRJPKA-UHFFFAOYSA-N 0.000 description 1
- FGRQOZGDEOPAAC-UHFFFAOYSA-N CC(C)(C)OC(NC(C=C(C=O)C([N+]([O-])=O)=C1)=C1F)=O Chemical compound CC(C)(C)OC(NC(C=C(C=O)C([N+]([O-])=O)=C1)=C1F)=O FGRQOZGDEOPAAC-UHFFFAOYSA-N 0.000 description 1
- OCHOEMREVVTGBO-UHFFFAOYSA-N CC(C)(C)OC(NC(C=C1C=O)=C(C(F)(F)F)C=C1N)=O Chemical compound CC(C)(C)OC(NC(C=C1C=O)=C(C(F)(F)F)C=C1N)=O OCHOEMREVVTGBO-UHFFFAOYSA-N 0.000 description 1
- ZEFCOVFRFWRQBI-UHFFFAOYSA-N CC(C)(C)OC(NC(C=C1C=O)=C(C(F)(F)F)C=C1[N+]([O-])=O)=O Chemical compound CC(C)(C)OC(NC(C=C1C=O)=C(C(F)(F)F)C=C1[N+]([O-])=O)=O ZEFCOVFRFWRQBI-UHFFFAOYSA-N 0.000 description 1
- UHNRLQRZRNKOKU-UHFFFAOYSA-N CCN(CC1=NC2=C(N1)C1=CC=C(C=C1N=C2N)C1=NNC=C1)C(C)=O Chemical compound CCN(CC1=NC2=C(N1)C1=CC=C(C=C1N=C2N)C1=NNC=C1)C(C)=O UHNRLQRZRNKOKU-UHFFFAOYSA-N 0.000 description 1
- GHHOYDHFWHKXCV-SANMLTNESA-N CC[C@](C(C=C1N2CC3=C(C=O)C(C=C(C(F)=C4)NC(OC(C)(C)C)=O)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(C=O)C(C=C(C(F)=C4)NC(OC(C)(C)C)=O)=C4N=C13)=C(CO1)C2=O)(C1=O)O GHHOYDHFWHKXCV-SANMLTNESA-N 0.000 description 1
- QYTOBYCRRKAYKY-SANMLTNESA-N CC[C@](C(C=C1N2CC3=C(CCl)C(C=C(C(F)=C4)NC(OC(C)(C)C)=O)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CCl)C(C=C(C(F)=C4)NC(OC(C)(C)C)=O)=C4N=C13)=C(CO1)C2=O)(C1=O)O QYTOBYCRRKAYKY-SANMLTNESA-N 0.000 description 1
- ZESULGLLOIQRDC-SANMLTNESA-N CC[C@](C(C=C1N2CC3=C(CN(C4)CC4(CO)F)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CN(C4)CC4(CO)F)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O ZESULGLLOIQRDC-SANMLTNESA-N 0.000 description 1
- GFZWUCDDPQSCTC-KSKVVAMCSA-N CC[C@](C(C=C1N2CC3=C(CN(CC4)C(CO)CS4(=O)=O)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CN(CC4)C(CO)CS4(=O)=O)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O GFZWUCDDPQSCTC-KSKVVAMCSA-N 0.000 description 1
- CPZNWJURIMZMKE-YTTGMZPUSA-N CC[C@](C(C=C1N2CC3=C(CN(CC4)CCN4S(C(C=C4)=CC=C4N)(=O)=O)C(C=C(C(F)=C4)OC)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CN(CC4)CCN4S(C(C=C4)=CC=C4N)(=O)=O)C(C=C(C(F)=C4)OC)=C4N=C13)=C(CO1)C2=O)(C1=O)O CPZNWJURIMZMKE-YTTGMZPUSA-N 0.000 description 1
- IOQFGWQHMSZOSL-SANMLTNESA-N CC[C@](C(C=C1N2CC3=C(CN)C(C=C(C(F)=C4)NC(OC(C)(C)C)=O)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CN)C(C=C(C(F)=C4)NC(OC(C)(C)C)=O)=C4N=C13)=C(CO1)C2=O)(C1=O)O IOQFGWQHMSZOSL-SANMLTNESA-N 0.000 description 1
- ZARAWTSIQUXCDW-ZVOWKAADSA-N CC[C@](C(C=C1N2CC3=C(CN4C(CO)(C5)COC5C4)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CN4C(CO)(C5)COC5C4)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O ZARAWTSIQUXCDW-ZVOWKAADSA-N 0.000 description 1
- PFXIASUZJBCXLV-SANMLTNESA-N CC[C@](C(C=C1N2CC3=C(CN4CCNCC4)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CN4CCNCC4)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O PFXIASUZJBCXLV-SANMLTNESA-N 0.000 description 1
- DUWLBFHXALUKPD-SANMLTNESA-N CC[C@](C(C=C1N2CC3=C(CN4CCOCC4)C(C=C(C(F)=C4)OC)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CN4CCOCC4)C(C=C(C(F)=C4)OC)=C4N=C13)=C(CO1)C2=O)(C1=O)O DUWLBFHXALUKPD-SANMLTNESA-N 0.000 description 1
- QWAIYPMIXZNGOD-DEOSSOPVSA-N CC[C@](C(C=C1N2CC3=C(CNC(NCCO)=O)C(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CNC(NCCO)=O)C(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O QWAIYPMIXZNGOD-DEOSSOPVSA-N 0.000 description 1
- GGHLAUGHLIXUIL-SANMLTNESA-N CC[C@](C(C=C1N2CC3=C(CNC(OC(C)(C)C)=O)C(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CNC(OC(C)(C)C)=O)C(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O GGHLAUGHLIXUIL-SANMLTNESA-N 0.000 description 1
- RFGSWYBAUAGOEM-NDEPHWFRSA-N CC[C@](C(C=C1N2CC3=C(CNC(OC(C)(C)C)=O)C(C=C(C(F)=C4)NC(CN)=O)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CNC(OC(C)(C)C)=O)C(C=C(C(F)=C4)NC(CN)=O)=C4N=C13)=C(CO1)C2=O)(C1=O)O RFGSWYBAUAGOEM-NDEPHWFRSA-N 0.000 description 1
- OOUAAHXLBAABGH-NDEPHWFRSA-N CC[C@](C(C=C1N2CC3=C(CNC(OC(C=C4)=CC=C4[N+]([O-])=O)=O)C(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CNC(OC(C=C4)=CC=C4[N+]([O-])=O)=O)C(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O OOUAAHXLBAABGH-NDEPHWFRSA-N 0.000 description 1
- IJZJLGCRABHOAW-QHCPKHFHSA-N CC[C@](C(C=C1N2CC3=C(CNC(OC)=O)C(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CNC(OC)=O)C(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O IJZJLGCRABHOAW-QHCPKHFHSA-N 0.000 description 1
- SJLICXIYNPAXHJ-VWLOTQADSA-N CC[C@](C(C=C1N2CC3=C(CNC(OCCO)=O)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CNC(OCCO)=O)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O SJLICXIYNPAXHJ-VWLOTQADSA-N 0.000 description 1
- JZFNVGJFNSBFCO-MHZLTWQESA-N CC[C@](C(C=C1N2CC3=C(CNC)C(C=C(C(F)=C4)NC(OC(C)(C)C)=O)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CNC)C(C=C(C(F)=C4)NC(OC(C)(C)C)=O)=C4N=C13)=C(CO1)C2=O)(C1=O)O JZFNVGJFNSBFCO-MHZLTWQESA-N 0.000 description 1
- NIWGFXRMELSZBQ-NDEPHWFRSA-N CC[C@](C(C=C1N2CC3=C(CNS(C(C=C4)=CC=C4N)(=O)=O)C(C=C(C(F)=C4)OC)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CNS(C(C=C4)=CC=C4N)(=O)=O)C(C=C(C(F)=C4)OC)=C4N=C13)=C(CO1)C2=O)(C1=O)O NIWGFXRMELSZBQ-NDEPHWFRSA-N 0.000 description 1
- IOMBTORHAVOHCA-NDEPHWFRSA-N CC[C@](C(C=C1N2CC3=C(CNS(C(C=C4)=CC=C4[N+]([O-])=O)(=O)=O)C(C=C(C(F)=C4)OC)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CNS(C(C=C4)=CC=C4[N+]([O-])=O)(=O)=O)C(C=C(C(F)=C4)OC)=C4N=C13)=C(CO1)C2=O)(C1=O)O IOMBTORHAVOHCA-NDEPHWFRSA-N 0.000 description 1
- ZFFIXODWBRDCQH-NDEPHWFRSA-N CC[C@](C(C=C1N2CC3=C(CNS(C(C=C4)=CC=C4[N+]([O-])=O)(=O)=O)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CNS(C(C=C4)=CC=C4[N+]([O-])=O)(=O)=O)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O ZFFIXODWBRDCQH-NDEPHWFRSA-N 0.000 description 1
- KVNGXFQGUPXCOX-QHCPKHFHSA-N CC[C@](C(C=C1N2CC3=C(CNS(C)(=O)=O)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CNS(C)(=O)=O)C(C=C(C)C(F)=C4)=C4N=C13)=C(CO1)C2=O)(C1=O)O KVNGXFQGUPXCOX-QHCPKHFHSA-N 0.000 description 1
- HKZIESYXBFFALF-QHCPKHFHSA-N CC[C@](C(C=C1N2CC3=C(CNS(CCO)(=O)=O)C(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CNS(CCO)(=O)=O)C(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O HKZIESYXBFFALF-QHCPKHFHSA-N 0.000 description 1
- DXMXBPVQZGWYQQ-QHCPKHFHSA-N CC[C@](C(C=C1N2CC3=C(CO)C(C=C(C(F)=C4)NC(CN)=O)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CO)C(C=C(C(F)=C4)NC(CN)=O)=C4N=C13)=C(CO1)C2=O)(C1=O)O DXMXBPVQZGWYQQ-QHCPKHFHSA-N 0.000 description 1
- VSUVGSOOVUABFL-SANMLTNESA-N CC[C@](C(C=C1N2CC3=C(CO)C(C=C(C(F)=C4)NC(OC(C)(C)C)=O)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(CO)C(C=C(C(F)=C4)NC(OC(C)(C)C)=O)=C4N=C13)=C(CO1)C2=O)(C1=O)O VSUVGSOOVUABFL-SANMLTNESA-N 0.000 description 1
- NTEJSVCFMIXSHC-MHZLTWQESA-N CC[C@](C(C=C1N2CC3=C(COC(N)=O)C(C=C(C(F)=C4)NC(OC(C)(C)C)=O)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(COC(N)=O)C(C=C(C(F)=C4)NC(OC(C)(C)C)=O)=C4N=C13)=C(CO1)C2=O)(C1=O)O NTEJSVCFMIXSHC-MHZLTWQESA-N 0.000 description 1
- SWSQVNUOUDHNBS-KDXMTYKHSA-N CC[C@](C(C=C1N2CC3=C(COCNC(CNC(OCC4C5=CC=CC=C5C5=C4C=CC=C5)=O)=O)C(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=C(COCNC(CNC(OCC4C5=CC=CC=C5C5=C4C=CC=C5)=O)=O)C(C=C(C(F)=C4)N)=C4N=C13)=C(CO1)C2=O)(C1=O)O SWSQVNUOUDHNBS-KDXMTYKHSA-N 0.000 description 1
- CNKGMNDQUMRMII-MHZLTWQESA-N CC[C@](C(C=C1N2CC3=CC(C=C(C(F)=C4)NC(CNC(OC(C)(C)C)=O)=O)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=CC(C=C(C(F)=C4)NC(CNC(OC(C)(C)C)=O)=O)=C4N=C13)=C(CO1)C2=O)(C1=O)O CNKGMNDQUMRMII-MHZLTWQESA-N 0.000 description 1
- ISWCUPJVOLJZFJ-UGDMGKLASA-N CC[C@](C(C=C1N2CC3=CC(C=C(C(F)=C4)NC(CNC([C@H](CC5=CC=CC=C5)N)=O)=O)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=CC(C=C(C(F)=C4)NC(CNC([C@H](CC5=CC=CC=C5)N)=O)=O)=C4N=C13)=C(CO1)C2=O)(C1=O)O ISWCUPJVOLJZFJ-UGDMGKLASA-N 0.000 description 1
- HNFOYXCLRLDVJX-VWLOTQADSA-N CC[C@](C(C=C1N2CC3=CC(C=C(C(F)=C4)NC(OC(C)(C)C)=O)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=CC(C=C(C(F)=C4)NC(OC(C)(C)C)=O)=C4N=C13)=C(CO1)C2=O)(C1=O)O HNFOYXCLRLDVJX-VWLOTQADSA-N 0.000 description 1
- UKYQTCDMOZEDHB-OIJAAMDRSA-N CC[C@](C(C=C1N2CC3=CC(C=C(C(F)=C4)NC([C@H](C)NC(OC(C)(C)C)=O)=O)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=CC(C=C(C(F)=C4)NC([C@H](C)NC(OC(C)(C)C)=O)=O)=C4N=C13)=C(CO1)C2=O)(C1=O)O UKYQTCDMOZEDHB-OIJAAMDRSA-N 0.000 description 1
- XMDFFQUKQREBIQ-ZBACMUJDSA-N CC[C@](C(C=C1N2CC3=CC(C=C(C(F)=C4)NC([C@H](C)NC([C@H](C(C)C)N)=O)=O)=C4N=C13)=C(CO1)C2=O)(C1=O)O Chemical compound CC[C@](C(C=C1N2CC3=CC(C=C(C(F)=C4)NC([C@H](C)NC([C@H](C(C)C)N)=O)=O)=C4N=C13)=C(CO1)C2=O)(C1=O)O XMDFFQUKQREBIQ-ZBACMUJDSA-N 0.000 description 1
- 102100024210 CD166 antigen Human genes 0.000 description 1
- 102000017420 CD3 protein, epsilon/gamma/delta subunit Human genes 0.000 description 1
- 108050005493 CD3 protein, epsilon/gamma/delta subunit Proteins 0.000 description 1
- 108010029697 CD40 Ligand Proteins 0.000 description 1
- 102100032937 CD40 ligand Human genes 0.000 description 1
- 108010058905 CD44v6 antigen Proteins 0.000 description 1
- 102100036008 CD48 antigen Human genes 0.000 description 1
- 102100025221 CD70 antigen Human genes 0.000 description 1
- 102100029756 Cadherin-6 Human genes 0.000 description 1
- 241000283707 Capra Species 0.000 description 1
- 229920002134 Carboxymethyl cellulose Polymers 0.000 description 1
- 102100025475 Carcinoembryonic antigen-related cell adhesion molecule 5 Human genes 0.000 description 1
- 102100025473 Carcinoembryonic antigen-related cell adhesion molecule 6 Human genes 0.000 description 1
- 102000005600 Cathepsins Human genes 0.000 description 1
- 108010084457 Cathepsins Proteins 0.000 description 1
- 206010008342 Cervix carcinoma Diseases 0.000 description 1
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 description 1
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 1
- RENMDAKOXSCIGH-UHFFFAOYSA-N Chloroacetonitrile Chemical compound ClCC#N RENMDAKOXSCIGH-UHFFFAOYSA-N 0.000 description 1
- 102100039496 Choline transporter-like protein 4 Human genes 0.000 description 1
- 208000006332 Choriocarcinoma Diseases 0.000 description 1
- JJBDYJMZKVHALC-QFIPXVFZSA-N ClCC1=C2C(=NC=3C=C(C(=CC1=3)OC)F)C1=CC3=C(C(N1C2)=O)COC([C@]3(O)CC)=O Chemical compound ClCC1=C2C(=NC=3C=C(C(=CC1=3)OC)F)C1=CC3=C(C(N1C2)=O)COC([C@]3(O)CC)=O JJBDYJMZKVHALC-QFIPXVFZSA-N 0.000 description 1
- 102100038449 Claudin-6 Human genes 0.000 description 1
- 208000001333 Colorectal Neoplasms Diseases 0.000 description 1
- 101710095920 Competence-stimulating peptide type 1 Proteins 0.000 description 1
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 1
- 229910021592 Copper(II) chloride Inorganic materials 0.000 description 1
- 229920002261 Corn starch Polymers 0.000 description 1
- 208000001528 Coronaviridae Infections Diseases 0.000 description 1
- 102000003915 DNA Topoisomerases Human genes 0.000 description 1
- 108090000323 DNA Topoisomerases Proteins 0.000 description 1
- 102100036466 Delta-like protein 3 Human genes 0.000 description 1
- 206010012438 Dermatitis atopic Diseases 0.000 description 1
- 108700022150 Designed Ankyrin Repeat Proteins Proteins 0.000 description 1
- 235000019739 Dicalciumphosphate Nutrition 0.000 description 1
- 102100029857 Dipeptidase 3 Human genes 0.000 description 1
- 102100024361 Disintegrin and metalloproteinase domain-containing protein 9 Human genes 0.000 description 1
- 101710116121 Disintegrin and metalloproteinase domain-containing protein 9 Proteins 0.000 description 1
- 101100120663 Drosophila melanogaster fs(1)h gene Proteins 0.000 description 1
- 102100026245 E3 ubiquitin-protein ligase RNF43 Human genes 0.000 description 1
- 238000002965 ELISA Methods 0.000 description 1
- 102100038083 Endosialin Human genes 0.000 description 1
- 108010055196 EphA2 Receptor Proteins 0.000 description 1
- 108010055191 EphA3 Receptor Proteins 0.000 description 1
- 102100030340 Ephrin type-A receptor 2 Human genes 0.000 description 1
- 102100030324 Ephrin type-A receptor 3 Human genes 0.000 description 1
- 102100033942 Ephrin-A4 Human genes 0.000 description 1
- 102000018651 Epithelial Cell Adhesion Molecule Human genes 0.000 description 1
- 108010066687 Epithelial Cell Adhesion Molecule Proteins 0.000 description 1
- 108010037362 Extracellular Matrix Proteins Proteins 0.000 description 1
- 102000010834 Extracellular Matrix Proteins Human genes 0.000 description 1
- 102100040834 FXYD domain-containing ion transport regulator 5 Human genes 0.000 description 1
- 102100023600 Fibroblast growth factor receptor 2 Human genes 0.000 description 1
- 101710182389 Fibroblast growth factor receptor 2 Proteins 0.000 description 1
- 102100027842 Fibroblast growth factor receptor 3 Human genes 0.000 description 1
- 101710182396 Fibroblast growth factor receptor 3 Proteins 0.000 description 1
- PXGOKWXKJXAPGV-UHFFFAOYSA-N Fluorine Chemical compound FF PXGOKWXKJXAPGV-UHFFFAOYSA-N 0.000 description 1
- 102100035139 Folate receptor alpha Human genes 0.000 description 1
- 101150030514 GPC1 gene Proteins 0.000 description 1
- 102100040510 Galectin-3-binding protein Human genes 0.000 description 1
- 102000004862 Gastrin releasing peptide Human genes 0.000 description 1
- 108090001053 Gastrin releasing peptide Proteins 0.000 description 1
- 102100036519 Gastrin-releasing peptide Human genes 0.000 description 1
- 108010010803 Gelatin Proteins 0.000 description 1
- 102000053187 Glucuronidase Human genes 0.000 description 1
- 108010060309 Glucuronidase Proteins 0.000 description 1
- 102100041003 Glutamate carboxypeptidase 2 Human genes 0.000 description 1
- 239000004471 Glycine Substances 0.000 description 1
- 244000068988 Glycine max Species 0.000 description 1
- 235000010469 Glycine max Nutrition 0.000 description 1
- AEMRFAOFKBGASW-UHFFFAOYSA-M Glycolate Chemical compound OCC([O-])=O AEMRFAOFKBGASW-UHFFFAOYSA-M 0.000 description 1
- 102000003886 Glycoproteins Human genes 0.000 description 1
- 108090000288 Glycoproteins Proteins 0.000 description 1
- 101150112082 Gpnmb gene Proteins 0.000 description 1
- 208000031886 HIV Infections Diseases 0.000 description 1
- 208000037357 HIV infectious disease Diseases 0.000 description 1
- 102100030595 HLA class II histocompatibility antigen gamma chain Human genes 0.000 description 1
- 102000006354 HLA-DR Antigens Human genes 0.000 description 1
- 108010058597 HLA-DR Antigens Proteins 0.000 description 1
- 101710113864 Heat shock protein 90 Proteins 0.000 description 1
- 102100034051 Heat shock protein HSP 90-alpha Human genes 0.000 description 1
- 208000002250 Hematologic Neoplasms Diseases 0.000 description 1
- 108010007712 Hepatitis A Virus Cellular Receptor 1 Proteins 0.000 description 1
- 102100034459 Hepatitis A virus cellular receptor 1 Human genes 0.000 description 1
- 102100034458 Hepatitis A virus cellular receptor 2 Human genes 0.000 description 1
- 101710083479 Hepatitis A virus cellular receptor 2 homolog Proteins 0.000 description 1
- 101000777636 Homo sapiens ADP-ribosyl cyclase/cyclic ADP-ribose hydrolase 1 Proteins 0.000 description 1
- 101000884305 Homo sapiens B-cell receptor CD22 Proteins 0.000 description 1
- 101000980825 Homo sapiens B-lymphocyte antigen CD19 Proteins 0.000 description 1
- 101000897405 Homo sapiens B-lymphocyte antigen CD20 Proteins 0.000 description 1
- 101000922348 Homo sapiens C-X-C chemokine receptor type 4 Proteins 0.000 description 1
- 101000922405 Homo sapiens C-X-C chemokine receptor type 5 Proteins 0.000 description 1
- 101000980840 Homo sapiens CD166 antigen Proteins 0.000 description 1
- 101000716130 Homo sapiens CD48 antigen Proteins 0.000 description 1
- 101000934356 Homo sapiens CD70 antigen Proteins 0.000 description 1
- 101000794604 Homo sapiens Cadherin-6 Proteins 0.000 description 1
- 101000914324 Homo sapiens Carcinoembryonic antigen-related cell adhesion molecule 5 Proteins 0.000 description 1
- 101000914326 Homo sapiens Carcinoembryonic antigen-related cell adhesion molecule 6 Proteins 0.000 description 1
- 101000882898 Homo sapiens Claudin-6 Proteins 0.000 description 1
- 101000928513 Homo sapiens Delta-like protein 3 Proteins 0.000 description 1
- 101000864130 Homo sapiens Dipeptidase 3 Proteins 0.000 description 1
- 101000692702 Homo sapiens E3 ubiquitin-protein ligase RNF43 Proteins 0.000 description 1
- 101000884275 Homo sapiens Endosialin Proteins 0.000 description 1
- 101000925259 Homo sapiens Ephrin-A4 Proteins 0.000 description 1
- 101000893718 Homo sapiens FXYD domain-containing ion transport regulator 5 Proteins 0.000 description 1
- 101001023230 Homo sapiens Folate receptor alpha Proteins 0.000 description 1
- 101000967904 Homo sapiens Galectin-3-binding protein Proteins 0.000 description 1
- 101000892862 Homo sapiens Glutamate carboxypeptidase 2 Proteins 0.000 description 1
- 101001082627 Homo sapiens HLA class II histocompatibility antigen gamma chain Proteins 0.000 description 1
- 101000606465 Homo sapiens Inactive tyrosine-protein kinase 7 Proteins 0.000 description 1
- 101001103039 Homo sapiens Inactive tyrosine-protein kinase transmembrane receptor ROR1 Proteins 0.000 description 1
- 101001046677 Homo sapiens Integrin alpha-V Proteins 0.000 description 1
- 101001057504 Homo sapiens Interferon-stimulated gene 20 kDa protein Proteins 0.000 description 1
- 101000960952 Homo sapiens Interleukin-1 receptor accessory protein Proteins 0.000 description 1
- 101001055144 Homo sapiens Interleukin-2 receptor subunit alpha Proteins 0.000 description 1
- 101000998120 Homo sapiens Interleukin-3 receptor subunit alpha Proteins 0.000 description 1
- 101001033312 Homo sapiens Interleukin-4 receptor subunit alpha Proteins 0.000 description 1
- 101001043809 Homo sapiens Interleukin-7 receptor subunit alpha Proteins 0.000 description 1
- 101001063456 Homo sapiens Leucine-rich repeat-containing G-protein coupled receptor 5 Proteins 0.000 description 1
- 101000777628 Homo sapiens Leukocyte antigen CD37 Proteins 0.000 description 1
- 101001018034 Homo sapiens Lymphocyte antigen 75 Proteins 0.000 description 1
- 101000634835 Homo sapiens M1-specific T cell receptor alpha chain Proteins 0.000 description 1
- 101000991061 Homo sapiens MHC class I polypeptide-related sequence B Proteins 0.000 description 1
- 101000961414 Homo sapiens Membrane cofactor protein Proteins 0.000 description 1
- 101000576802 Homo sapiens Mesothelin Proteins 0.000 description 1
- 101000623873 Homo sapiens Metaxin-3 Proteins 0.000 description 1
- 101000934338 Homo sapiens Myeloid cell surface antigen CD33 Proteins 0.000 description 1
- 101000581981 Homo sapiens Neural cell adhesion molecule 1 Proteins 0.000 description 1
- 101000724418 Homo sapiens Neutral amino acid transporter B(0) Proteins 0.000 description 1
- 101000897042 Homo sapiens Nucleotide pyrophosphatase Proteins 0.000 description 1
- 101001099381 Homo sapiens Peroxisomal biogenesis factor 19 Proteins 0.000 description 1
- 101000904173 Homo sapiens Progonadoliberin-1 Proteins 0.000 description 1
- 101000932478 Homo sapiens Receptor-type tyrosine-protein kinase FLT3 Proteins 0.000 description 1
- 101000738771 Homo sapiens Receptor-type tyrosine-protein phosphatase C Proteins 0.000 description 1
- 101000633786 Homo sapiens SLAM family member 6 Proteins 0.000 description 1
- 101000633784 Homo sapiens SLAM family member 7 Proteins 0.000 description 1
- 101000835984 Homo sapiens SLIT and NTRK-like protein 6 Proteins 0.000 description 1
- 101000936922 Homo sapiens Sarcoplasmic/endoplasmic reticulum calcium ATPase 2 Proteins 0.000 description 1
- 101000829127 Homo sapiens Somatostatin receptor type 2 Proteins 0.000 description 1
- 101000874179 Homo sapiens Syndecan-1 Proteins 0.000 description 1
- 101000634836 Homo sapiens T cell receptor alpha chain MC.7.G5 Proteins 0.000 description 1
- 101000914496 Homo sapiens T-cell antigen CD7 Proteins 0.000 description 1
- 101000635804 Homo sapiens Tissue factor Proteins 0.000 description 1
- 101000835093 Homo sapiens Transferrin receptor protein 1 Proteins 0.000 description 1
- 101000658574 Homo sapiens Transmembrane 4 L6 family member 1 Proteins 0.000 description 1
- 101000851376 Homo sapiens Tumor necrosis factor receptor superfamily member 8 Proteins 0.000 description 1
- 101001103033 Homo sapiens Tyrosine-protein kinase transmembrane receptor ROR2 Proteins 0.000 description 1
- 101000808105 Homo sapiens Uroplakin-2 Proteins 0.000 description 1
- 101000685848 Homo sapiens Zinc transporter ZIP6 Proteins 0.000 description 1
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 1
- 206010061598 Immunodeficiency Diseases 0.000 description 1
- 108060003951 Immunoglobulin Proteins 0.000 description 1
- 102100039813 Inactive tyrosine-protein kinase 7 Human genes 0.000 description 1
- 102100039615 Inactive tyrosine-protein kinase transmembrane receptor ROR1 Human genes 0.000 description 1
- 102100022337 Integrin alpha-V Human genes 0.000 description 1
- 102100027268 Interferon-stimulated gene 20 kDa protein Human genes 0.000 description 1
- 102100039880 Interleukin-1 receptor accessory protein Human genes 0.000 description 1
- 102100020793 Interleukin-13 receptor subunit alpha-2 Human genes 0.000 description 1
- 102100033493 Interleukin-3 receptor subunit alpha Human genes 0.000 description 1
- 102100039078 Interleukin-4 receptor subunit alpha Human genes 0.000 description 1
- 102100021593 Interleukin-7 receptor subunit alpha Human genes 0.000 description 1
- 108020003285 Isocitrate lyase Proteins 0.000 description 1
- 102000001399 Kallikrein Human genes 0.000 description 1
- 108060005987 Kallikrein Proteins 0.000 description 1
- 208000008839 Kidney Neoplasms Diseases 0.000 description 1
- PWKSKIMOESPYIA-BYPYZUCNSA-N L-N-acetyl-Cysteine Chemical compound CC(=O)N[C@@H](CS)C(O)=O PWKSKIMOESPYIA-BYPYZUCNSA-N 0.000 description 1
- 239000012480 LAL reagent Substances 0.000 description 1
- 108090001090 Lectins Proteins 0.000 description 1
- 102000004856 Lectins Human genes 0.000 description 1
- 102100031036 Leucine-rich repeat-containing G-protein coupled receptor 5 Human genes 0.000 description 1
- 102100031586 Leukocyte antigen CD37 Human genes 0.000 description 1
- 102000019298 Lipocalin Human genes 0.000 description 1
- 108050006654 Lipocalin Proteins 0.000 description 1
- 102100033486 Lymphocyte antigen 75 Human genes 0.000 description 1
- 102100035133 Lysosome-associated membrane glycoprotein 1 Human genes 0.000 description 1
- 101710116782 Lysosome-associated membrane glycoprotein 1 Proteins 0.000 description 1
- 102100029450 M1-specific T cell receptor alpha chain Human genes 0.000 description 1
- 102100030301 MHC class I polypeptide-related sequence A Human genes 0.000 description 1
- 102100030300 MHC class I polypeptide-related sequence B Human genes 0.000 description 1
- OFOBLEOULBTSOW-UHFFFAOYSA-L Malonate Chemical compound [O-]C(=O)CC([O-])=O OFOBLEOULBTSOW-UHFFFAOYSA-L 0.000 description 1
- 102000011716 Matrix Metalloproteinase 14 Human genes 0.000 description 1
- 108010076557 Matrix Metalloproteinase 14 Proteins 0.000 description 1
- 102100039373 Membrane cofactor protein Human genes 0.000 description 1
- 102100025096 Mesothelin Human genes 0.000 description 1
- 108010006035 Metalloproteases Proteins 0.000 description 1
- 102000005741 Metalloproteases Human genes 0.000 description 1
- 206010027476 Metastases Diseases 0.000 description 1
- 102100023139 Metaxin-3 Human genes 0.000 description 1
- BZLVMXJERCGZMT-UHFFFAOYSA-N Methyl tert-butyl ether Chemical compound COC(C)(C)C BZLVMXJERCGZMT-UHFFFAOYSA-N 0.000 description 1
- 101100369076 Mus musculus Tdgf1 gene Proteins 0.000 description 1
- 102100025243 Myeloid cell surface antigen CD33 Human genes 0.000 description 1
- OVBPIULPVIDEAO-UHFFFAOYSA-N N-Pteroyl-L-glutaminsaeure Natural products C=1N=C2NC(N)=NC(=O)C2=NC=1CNC1=CC=C(C(=O)NC(CCC(O)=O)C(O)=O)C=C1 OVBPIULPVIDEAO-UHFFFAOYSA-N 0.000 description 1
- LIMFPAAAIVQRRD-BCGVJQADSA-N N-[2-[(3S,4R)-3-fluoro-4-methoxypiperidin-1-yl]pyrimidin-4-yl]-8-[(2R,3S)-2-methyl-3-(methylsulfonylmethyl)azetidin-1-yl]-5-propan-2-ylisoquinolin-3-amine Chemical compound F[C@H]1CN(CC[C@H]1OC)C1=NC=CC(=N1)NC=1N=CC2=C(C=CC(=C2C=1)C(C)C)N1[C@@H]([C@H](C1)CS(=O)(=O)C)C LIMFPAAAIVQRRD-BCGVJQADSA-N 0.000 description 1
- POFVJRKJJBFPII-UHFFFAOYSA-N N-cyclopentyl-5-[2-[[5-[(4-ethylpiperazin-1-yl)methyl]pyridin-2-yl]amino]-5-fluoropyrimidin-4-yl]-4-methyl-1,3-thiazol-2-amine Chemical compound C1(CCCC1)NC=1SC(=C(N=1)C)C1=NC(=NC=C1F)NC1=NC=C(C=C1)CN1CCN(CC1)CC POFVJRKJJBFPII-UHFFFAOYSA-N 0.000 description 1
- UGJBHEZMOKVTIM-UHFFFAOYSA-N N-formylglycine Chemical compound OC(=O)CNC=O UGJBHEZMOKVTIM-UHFFFAOYSA-N 0.000 description 1
- CNGXKGWHKFFUBU-QFIPXVFZSA-N NCC1=C2C(=NC=3C=C(C(=CC1=3)OC)F)C1=CC3=C(C(N1C2)=O)COC([C@]3(O)CC)=O Chemical compound NCC1=C2C(=NC=3C=C(C(=CC1=3)OC)F)C1=CC3=C(C(N1C2)=O)COC([C@]3(O)CC)=O CNGXKGWHKFFUBU-QFIPXVFZSA-N 0.000 description 1
- 238000005481 NMR spectroscopy Methods 0.000 description 1
- 102100035486 Nectin-4 Human genes 0.000 description 1
- 101710043865 Nectin-4 Proteins 0.000 description 1
- 102100027347 Neural cell adhesion molecule 1 Human genes 0.000 description 1
- 102100028267 Neutral amino acid transporter B(0) Human genes 0.000 description 1
- 102100021969 Nucleotide pyrophosphatase Human genes 0.000 description 1
- RMINQIRDFIBNLE-NNRWGFCXSA-N O-[N-acetyl-alpha-neuraminyl-(2->6)-N-acetyl-alpha-D-galactosaminyl]-L-serine Chemical compound O1[C@H](OC[C@H](N)C(O)=O)[C@H](NC(=O)C)[C@@H](O)[C@@H](O)[C@H]1CO[C@@]1(C(O)=O)O[C@@H]([C@H](O)[C@H](O)CO)[C@H](NC(C)=O)[C@@H](O)C1 RMINQIRDFIBNLE-NNRWGFCXSA-N 0.000 description 1
- ILUJQPXNXACGAN-UHFFFAOYSA-N O-methylsalicylic acid Chemical class COC1=CC=CC=C1C(O)=O ILUJQPXNXACGAN-UHFFFAOYSA-N 0.000 description 1
- VCPAGPOJXJVXKO-ULXOWCQFSA-N O=C(CNC(OCC1C2=CC=CC=C2C2=C1C=CC=C2)=O)NCC(N[C@@H](CC1=CC=CC=C1)C(NCC(NCOC1C2CNCC1C2)=O)=O)=O Chemical compound O=C(CNC(OCC1C2=CC=CC=C2C2=C1C=CC=C2)=O)NCC(N[C@@H](CC1=CC=CC=C1)C(NCC(NCOC1C2CNCC1C2)=O)=O)=O VCPAGPOJXJVXKO-ULXOWCQFSA-N 0.000 description 1
- SLUMDRUFLYFIES-LJAQVGFWSA-N O=C(CNC(OCC1C2=CC=CC=C2C2=C1C=CC=C2)=O)NCC(N[C@@H](CC1=CC=CC=C1)C(NCC(NCOCC1(CNC1)F)=O)=O)=O Chemical compound O=C(CNC(OCC1C2=CC=CC=C2C2=C1C=CC=C2)=O)NCC(N[C@@H](CC1=CC=CC=C1)C(NCC(NCOCC1(CNC1)F)=O)=O)=O SLUMDRUFLYFIES-LJAQVGFWSA-N 0.000 description 1
- VFJAFDQZLDKVRU-INIZCTEOSA-N O=C(CNC(OCC1C2=CC=CC=C2C2=C1C=CC=C2)=O)NCOC[C@H]1OCCNC1 Chemical compound O=C(CNC(OCC1C2=CC=CC=C2C2=C1C=CC=C2)=O)NCOC[C@H]1OCCNC1 VFJAFDQZLDKVRU-INIZCTEOSA-N 0.000 description 1
- 239000005642 Oleic acid Substances 0.000 description 1
- ZQPPMHVWECSIRJ-UHFFFAOYSA-N Oleic acid Natural products CCCCCCCCC=CCCCCCCCC(O)=O ZQPPMHVWECSIRJ-UHFFFAOYSA-N 0.000 description 1
- 102000016387 Pancreatic elastase Human genes 0.000 description 1
- 108010067372 Pancreatic elastase Proteins 0.000 description 1
- 206010061902 Pancreatic neoplasm Diseases 0.000 description 1
- 102100038883 Peroxisomal biogenesis factor 19 Human genes 0.000 description 1
- 239000004264 Petrolatum Substances 0.000 description 1
- IGVPBCZDHMIOJH-UHFFFAOYSA-N Phenyl butyrate Chemical class CCCC(=O)OC1=CC=CC=C1 IGVPBCZDHMIOJH-UHFFFAOYSA-N 0.000 description 1
- 241000233805 Phoenix Species 0.000 description 1
- 206010035226 Plasma cell myeloma Diseases 0.000 description 1
- 241000276498 Pollachius virens Species 0.000 description 1
- 229920003171 Poly (ethylene oxide) Polymers 0.000 description 1
- 239000004698 Polyethylene Substances 0.000 description 1
- 102100024028 Progonadoliberin-1 Human genes 0.000 description 1
- 102100024216 Programmed cell death 1 ligand 1 Human genes 0.000 description 1
- 102100021923 Prolow-density lipoprotein receptor-related protein 1 Human genes 0.000 description 1
- 101710202113 Prolow-density lipoprotein receptor-related protein 1 Proteins 0.000 description 1
- OFOBLEOULBTSOW-UHFFFAOYSA-N Propanedioic acid Natural products OC(=O)CC(O)=O OFOBLEOULBTSOW-UHFFFAOYSA-N 0.000 description 1
- XBDQKXXYIPTUBI-UHFFFAOYSA-N Propionic acid Chemical class CCC(O)=O XBDQKXXYIPTUBI-UHFFFAOYSA-N 0.000 description 1
- 206010060862 Prostate cancer Diseases 0.000 description 1
- 208000000236 Prostatic Neoplasms Diseases 0.000 description 1
- 102100036467 Protein delta homolog 1 Human genes 0.000 description 1
- 101710119301 Protein delta homolog 1 Proteins 0.000 description 1
- 201000004681 Psoriasis Diseases 0.000 description 1
- 239000012980 RPMI-1640 medium Substances 0.000 description 1
- 102100020718 Receptor-type tyrosine-protein kinase FLT3 Human genes 0.000 description 1
- 102100037422 Receptor-type tyrosine-protein phosphatase C Human genes 0.000 description 1
- 108020004511 Recombinant DNA Proteins 0.000 description 1
- 206010038389 Renal cancer Diseases 0.000 description 1
- 241000315672 SARS coronavirus Species 0.000 description 1
- 102100029197 SLAM family member 6 Human genes 0.000 description 1
- 102100029198 SLAM family member 7 Human genes 0.000 description 1
- 108091007561 SLC44A4 Proteins 0.000 description 1
- 102100025504 SLIT and NTRK-like protein 6 Human genes 0.000 description 1
- 102100027732 Sarcoplasmic/endoplasmic reticulum calcium ATPase 2 Human genes 0.000 description 1
- RJFAYQIBOAGBLC-BYPYZUCNSA-N Selenium-L-methionine Chemical compound C[Se]CC[C@H](N)C(O)=O RJFAYQIBOAGBLC-BYPYZUCNSA-N 0.000 description 1
- RJFAYQIBOAGBLC-UHFFFAOYSA-N Selenomethionine Natural products C[Se]CCC(N)C(O)=O RJFAYQIBOAGBLC-UHFFFAOYSA-N 0.000 description 1
- 229920002684 Sepharose Polymers 0.000 description 1
- UIIMBOGNXHQVGW-DEQYMQKBSA-M Sodium bicarbonate-14C Chemical compound [Na+].O[14C]([O-])=O UIIMBOGNXHQVGW-DEQYMQKBSA-M 0.000 description 1
- DBMJMQXJHONAFJ-UHFFFAOYSA-M Sodium laurylsulphate Chemical compound [Na+].CCCCCCCCCCCCOS([O-])(=O)=O DBMJMQXJHONAFJ-UHFFFAOYSA-M 0.000 description 1
- 102100023802 Somatostatin receptor type 2 Human genes 0.000 description 1
- 235000021355 Stearic acid Nutrition 0.000 description 1
- 208000005718 Stomach Neoplasms Diseases 0.000 description 1
- 108010090804 Streptavidin Proteins 0.000 description 1
- 241001495137 Streptomyces mobaraensis Species 0.000 description 1
- KDYFGRWQOYBRFD-UHFFFAOYSA-N Succinic acid Natural products OC(=O)CCC(O)=O KDYFGRWQOYBRFD-UHFFFAOYSA-N 0.000 description 1
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 1
- 101000996723 Sus scrofa Gonadotropin-releasing hormone receptor Proteins 0.000 description 1
- 102100035721 Syndecan-1 Human genes 0.000 description 1
- 102100027208 T-cell antigen CD7 Human genes 0.000 description 1
- 229940126547 T-cell immunoglobulin mucin-3 Drugs 0.000 description 1
- 102100030859 Tissue factor Human genes 0.000 description 1
- 102000004338 Transferrin Human genes 0.000 description 1
- 108090000901 Transferrin Proteins 0.000 description 1
- 102100026144 Transferrin receptor protein 1 Human genes 0.000 description 1
- 102100034902 Transmembrane 4 L6 family member 1 Human genes 0.000 description 1
- 102000004142 Trypsin Human genes 0.000 description 1
- 108090000631 Trypsin Proteins 0.000 description 1
- 108060008682 Tumor Necrosis Factor Proteins 0.000 description 1
- 102000000852 Tumor Necrosis Factor-alpha Human genes 0.000 description 1
- 102100036857 Tumor necrosis factor receptor superfamily member 8 Human genes 0.000 description 1
- 102100039616 Tyrosine-protein kinase transmembrane receptor ROR2 Human genes 0.000 description 1
- 102100038851 Uroplakin-2 Human genes 0.000 description 1
- 208000006105 Uterine Cervical Neoplasms Diseases 0.000 description 1
- 208000002495 Uterine Neoplasms Diseases 0.000 description 1
- 102100023144 Zinc transporter ZIP6 Human genes 0.000 description 1
- VLAZLCVSFAYIIL-RXMQYKEDSA-N [(2r)-morpholin-2-yl]methanol Chemical compound OC[C@H]1CNCCO1 VLAZLCVSFAYIIL-RXMQYKEDSA-N 0.000 description 1
- VLAZLCVSFAYIIL-YFKPBYRVSA-N [(2s)-morpholin-2-yl]methanol Chemical compound OC[C@@H]1CNCCO1 VLAZLCVSFAYIIL-YFKPBYRVSA-N 0.000 description 1
- WERKSKAQRVDLDW-ANOHMWSOSA-N [(2s,3r,4r,5r)-2,3,4,5,6-pentahydroxyhexyl] (z)-octadec-9-enoate Chemical compound CCCCCCCC\C=C/CCCCCCCC(=O)OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO WERKSKAQRVDLDW-ANOHMWSOSA-N 0.000 description 1
- PKOKAERYVONVSL-UHFFFAOYSA-N [7-[3-(4-fluorophenoxy)-2-hydroxypropyl]-3-methyl-2,6-dioxopurin-8-yl] benzoate Chemical compound C=1C=C(F)C=CC=1OCC(O)CN1C=2C(=O)NC(=O)N(C)C=2N=C1OC(=O)C1=CC=CC=C1 PKOKAERYVONVSL-UHFFFAOYSA-N 0.000 description 1
- IMQUIIUKNXMJRT-UHFFFAOYSA-N [O-][N+](C(C=C1)=CC=C1OC(NCCOCNC(CNC(OCC1C2=CC=CC=C2C2=C1C=CC=C2)=O)=O)=O)=O Chemical compound [O-][N+](C(C=C1)=CC=C1OC(NCCOCNC(CNC(OCC1C2=CC=CC=C2C2=C1C=CC=C2)=O)=O)=O)=O IMQUIIUKNXMJRT-UHFFFAOYSA-N 0.000 description 1
- MQNSEPLSHPJLGY-UMSFTDKQSA-N [O-][N+](C(C=C1)=CC=C1OC(OCCOCNC(CNC([C@H](CC1=CC=CC=C1)NC(CNC(CNC(OCC1C2=CC=CC=C2C2=C1C=CC=C2)=O)=O)=O)=O)=O)=O)=O Chemical compound [O-][N+](C(C=C1)=CC=C1OC(OCCOCNC(CNC([C@H](CC1=CC=CC=C1)NC(CNC(CNC(OCC1C2=CC=CC=C2C2=C1C=CC=C2)=O)=O)=O)=O)=O)=O)=O MQNSEPLSHPJLGY-UMSFTDKQSA-N 0.000 description 1
- PYLKDJYQQHWVTQ-UHFFFAOYSA-N [O-][N+](C(C=C1)=CC=C1OC(OCCSSCCNC(CCN(C(C=C1)=O)C1=O)=O)=O)=O Chemical compound [O-][N+](C(C=C1)=CC=C1OC(OCCSSCCNC(CCN(C(C=C1)=O)C1=O)=O)=O)=O PYLKDJYQQHWVTQ-UHFFFAOYSA-N 0.000 description 1
- 230000002159 abnormal effect Effects 0.000 description 1
- 150000001241 acetals Chemical group 0.000 description 1
- 150000001242 acetic acid derivatives Chemical class 0.000 description 1
- CSCPPACGZOOCGX-WFGJKAKNSA-N acetone d6 Chemical compound [2H]C([2H])([2H])C(=O)C([2H])([2H])[2H] CSCPPACGZOOCGX-WFGJKAKNSA-N 0.000 description 1
- 150000008065 acid anhydrides Chemical class 0.000 description 1
- 230000002378 acidificating effect Effects 0.000 description 1
- 150000001252 acrylic acid derivatives Chemical class 0.000 description 1
- 208000009956 adenocarcinoma Diseases 0.000 description 1
- 239000002671 adjuvant Substances 0.000 description 1
- 230000002411 adverse Effects 0.000 description 1
- QBYJBZPUGVGKQQ-SJJAEHHWSA-N aldrin Chemical compound C1[C@H]2C=C[C@@H]1[C@H]1[C@@](C3(Cl)Cl)(Cl)C(Cl)=C(Cl)[C@@]3(Cl)[C@H]12 QBYJBZPUGVGKQQ-SJJAEHHWSA-N 0.000 description 1
- 235000010443 alginic acid Nutrition 0.000 description 1
- 239000000783 alginic acid Substances 0.000 description 1
- 229920000615 alginic acid Polymers 0.000 description 1
- 229960001126 alginic acid Drugs 0.000 description 1
- 150000004781 alginic acids Chemical class 0.000 description 1
- 125000001931 aliphatic group Chemical group 0.000 description 1
- 239000003513 alkali Substances 0.000 description 1
- 229910001854 alkali hydroxide Inorganic materials 0.000 description 1
- 229910001860 alkaline earth metal hydroxide Inorganic materials 0.000 description 1
- 125000003342 alkenyl group Chemical group 0.000 description 1
- 150000001345 alkine derivatives Chemical class 0.000 description 1
- 125000005237 alkyleneamino group Chemical group 0.000 description 1
- 125000000304 alkynyl group Chemical group 0.000 description 1
- MXKCYTKUIDTFLY-ZNNSSXPHSA-N alpha-L-Fucp-(1->2)-beta-D-Galp-(1->4)-[alpha-L-Fucp-(1->3)]-beta-D-GlcpNAc-(1->3)-D-Galp Chemical compound O[C@H]1[C@H](O)[C@H](O)[C@H](C)O[C@H]1O[C@H]1[C@H](O[C@H]2[C@@H]([C@@H](NC(C)=O)[C@H](O[C@H]3[C@H]([C@@H](CO)OC(O)[C@@H]3O)O)O[C@@H]2CO)O[C@H]2[C@H]([C@H](O)[C@H](O)[C@H](C)O2)O)O[C@H](CO)[C@H](O)[C@@H]1O MXKCYTKUIDTFLY-ZNNSSXPHSA-N 0.000 description 1
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 1
- 235000012211 aluminium silicate Nutrition 0.000 description 1
- SNAAJJQQZSMGQD-UHFFFAOYSA-N aluminum magnesium Chemical compound [Mg].[Al] SNAAJJQQZSMGQD-UHFFFAOYSA-N 0.000 description 1
- 239000000908 ammonium hydroxide Substances 0.000 description 1
- 230000003432 anti-folate effect Effects 0.000 description 1
- 229940125644 antibody drug Drugs 0.000 description 1
- 229940127074 antifolate Drugs 0.000 description 1
- 239000002246 antineoplastic agent Substances 0.000 description 1
- 239000003963 antioxidant agent Substances 0.000 description 1
- 230000003078 antioxidant effect Effects 0.000 description 1
- 235000006708 antioxidants Nutrition 0.000 description 1
- 239000008365 aqueous carrier Substances 0.000 description 1
- 150000001491 aromatic compounds Chemical class 0.000 description 1
- 125000003710 aryl alkyl group Chemical group 0.000 description 1
- 235000010323 ascorbic acid Nutrition 0.000 description 1
- 229960005070 ascorbic acid Drugs 0.000 description 1
- 239000011668 ascorbic acid Substances 0.000 description 1
- 108010055066 asparaginylendopeptidase Proteins 0.000 description 1
- 125000004429 atom Chemical group 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- 201000008937 atopic dermatitis Diseases 0.000 description 1
- GNVWVYIAQBJHGV-UHFFFAOYSA-N azetidin-3-ylmethanol Chemical compound OCC1CNC1 GNVWVYIAQBJHGV-UHFFFAOYSA-N 0.000 description 1
- 125000002393 azetidinyl group Chemical group 0.000 description 1
- 125000004069 aziridinyl group Chemical group 0.000 description 1
- 210000003719 b-lymphocyte Anatomy 0.000 description 1
- 230000001580 bacterial effect Effects 0.000 description 1
- 235000013871 bee wax Nutrition 0.000 description 1
- 239000012166 beeswax Substances 0.000 description 1
- LNHWXBUNXOXMRL-VWLOTQADSA-N belotecan Chemical compound C1=CC=C2C(CCNC(C)C)=C(CN3C4=CC5=C(C3=O)COC(=O)[C@]5(O)CC)C4=NC2=C1 LNHWXBUNXOXMRL-VWLOTQADSA-N 0.000 description 1
- 229950011276 belotecan Drugs 0.000 description 1
- 125000000499 benzofuranyl group Chemical group O1C(=CC2=C1C=CC=C2)* 0.000 description 1
- 235000010233 benzoic acid Nutrition 0.000 description 1
- 150000001558 benzoic acid derivatives Chemical class 0.000 description 1
- 125000001164 benzothiazolyl group Chemical group S1C(=NC2=C1C=CC=C2)* 0.000 description 1
- 125000004541 benzoxazolyl group Chemical group O1C(=NC2=C1C=CC=C2)* 0.000 description 1
- SHOMMGQAMRXRRK-UHFFFAOYSA-N bicyclo[3.1.1]heptane Chemical compound C1C2CC1CCC2 SHOMMGQAMRXRRK-UHFFFAOYSA-N 0.000 description 1
- 230000008033 biological extinction Effects 0.000 description 1
- LZZXZDMVRZJZST-UHFFFAOYSA-N bis(2,5-dioxopyrrolidin-1-yl) hexanedioate Chemical compound O=C1CCC(=O)N1OC(=O)CCCCC(=O)ON1C(=O)CCC1=O LZZXZDMVRZJZST-UHFFFAOYSA-N 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-M bisulphate group Chemical group S([O-])(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-M 0.000 description 1
- DNDCVAGJPBKION-DOPDSADYSA-N bombesin Chemical compound C([C@@H](C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCSC)C(N)=O)NC(=O)CNC(=O)[C@@H](NC(=O)[C@H](C)NC(=O)[C@H](CC=1NC2=CC=CC=C2C=1)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](CC(N)=O)NC(=O)CNC(=O)[C@H](CC(C)C)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H]1NC(=O)CC1)C(C)C)C1=CN=CN1 DNDCVAGJPBKION-DOPDSADYSA-N 0.000 description 1
- 210000001185 bone marrow Anatomy 0.000 description 1
- 201000008274 breast adenocarcinoma Diseases 0.000 description 1
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 description 1
- 229910052794 bromium Inorganic materials 0.000 description 1
- 150000001649 bromium compounds Chemical class 0.000 description 1
- 239000006172 buffering agent Substances 0.000 description 1
- KDYFGRWQOYBRFD-NUQCWPJISA-N butanedioic acid Chemical compound O[14C](=O)CC[14C](O)=O KDYFGRWQOYBRFD-NUQCWPJISA-N 0.000 description 1
- ZTQSAGDEMFDKMZ-UHFFFAOYSA-N butyric aldehyde Natural products CCCC=O ZTQSAGDEMFDKMZ-UHFFFAOYSA-N 0.000 description 1
- AXCZMVOFGPJBDE-UHFFFAOYSA-L calcium dihydroxide Chemical compound [OH-].[OH-].[Ca+2] AXCZMVOFGPJBDE-UHFFFAOYSA-L 0.000 description 1
- 239000000920 calcium hydroxide Substances 0.000 description 1
- 229910001861 calcium hydroxide Inorganic materials 0.000 description 1
- 230000005880 cancer cell killing Effects 0.000 description 1
- 238000005251 capillar electrophoresis Methods 0.000 description 1
- 239000004202 carbamide Substances 0.000 description 1
- 150000001720 carbohydrates Chemical group 0.000 description 1
- BVKZGUZCCUSVTD-UHFFFAOYSA-N carbonic acid Chemical compound OC(O)=O BVKZGUZCCUSVTD-UHFFFAOYSA-N 0.000 description 1
- 150000004649 carbonic acid derivatives Chemical class 0.000 description 1
- 125000002915 carbonyl group Chemical group [*:2]C([*:1])=O 0.000 description 1
- 125000006243 carbonyl protecting group Chemical group 0.000 description 1
- 150000003857 carboxamides Chemical class 0.000 description 1
- 150000007942 carboxylates Chemical group 0.000 description 1
- 150000001732 carboxylic acid derivatives Chemical class 0.000 description 1
- 125000006244 carboxylic acid protecting group Chemical group 0.000 description 1
- 210000000170 cell membrane Anatomy 0.000 description 1
- 230000004663 cell proliferation Effects 0.000 description 1
- 201000010881 cervical cancer Diseases 0.000 description 1
- 229960000541 cetyl alcohol Drugs 0.000 description 1
- 239000007795 chemical reaction product Substances 0.000 description 1
- 238000013375 chromatographic separation Methods 0.000 description 1
- 150000001860 citric acid derivatives Chemical class 0.000 description 1
- 229940110456 cocoa butter Drugs 0.000 description 1
- 235000019868 cocoa butter Nutrition 0.000 description 1
- 239000003240 coconut oil Substances 0.000 description 1
- 235000019864 coconut oil Nutrition 0.000 description 1
- 208000029742 colonic neoplasm Diseases 0.000 description 1
- 230000001268 conjugating effect Effects 0.000 description 1
- 229910052802 copper Inorganic materials 0.000 description 1
- 239000010949 copper Substances 0.000 description 1
- ORTQZVOHEJQUHG-UHFFFAOYSA-L copper(II) chloride Chemical compound Cl[Cu]Cl ORTQZVOHEJQUHG-UHFFFAOYSA-L 0.000 description 1
- 239000008120 corn starch Substances 0.000 description 1
- 125000001995 cyclobutyl group Chemical group [H]C1([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- ZPWOOKQUDFIEIX-UHFFFAOYSA-N cyclooctyne Chemical compound C1CCCC#CCC1 ZPWOOKQUDFIEIX-UHFFFAOYSA-N 0.000 description 1
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- 125000001559 cyclopropyl group Chemical group [H]C1([H])C([H])([H])C1([H])* 0.000 description 1
- 235000018417 cysteine Nutrition 0.000 description 1
- 125000005534 decanoate group Chemical class 0.000 description 1
- 229940127276 delta-like ligand 3 Drugs 0.000 description 1
- RAFNCPHFRHZCPS-UHFFFAOYSA-N di(imidazol-1-yl)methanethione Chemical compound C1=CN=CN1C(=S)N1C=CN=C1 RAFNCPHFRHZCPS-UHFFFAOYSA-N 0.000 description 1
- 150000004985 diamines Chemical class 0.000 description 1
- NEFBYIFKOOEVPA-UHFFFAOYSA-K dicalcium phosphate Chemical compound [Ca+2].[Ca+2].[O-]P([O-])([O-])=O NEFBYIFKOOEVPA-UHFFFAOYSA-K 0.000 description 1
- 229910000390 dicalcium phosphate Inorganic materials 0.000 description 1
- 229940038472 dicalcium phosphate Drugs 0.000 description 1
- YNLAOSYQHBDIKW-UHFFFAOYSA-M diethylaluminium chloride Chemical compound CC[Al](Cl)CC YNLAOSYQHBDIKW-UHFFFAOYSA-M 0.000 description 1
- 238000010790 dilution Methods 0.000 description 1
- 239000012895 dilution Substances 0.000 description 1
- 125000000118 dimethyl group Chemical group [H]C([H])([H])* 0.000 description 1
- 230000008034 disappearance Effects 0.000 description 1
- 239000000975 dye Substances 0.000 description 1
- 238000001962 electrophoresis Methods 0.000 description 1
- 238000010828 elution Methods 0.000 description 1
- 239000012149 elution buffer Substances 0.000 description 1
- 239000003995 emulsifying agent Substances 0.000 description 1
- 238000005538 encapsulation Methods 0.000 description 1
- 201000004202 endocervical carcinoma Diseases 0.000 description 1
- 210000001163 endosome Anatomy 0.000 description 1
- 239000002158 endotoxin Substances 0.000 description 1
- 230000009144 enzymatic modification Effects 0.000 description 1
- 102000052116 epidermal growth factor receptor activity proteins Human genes 0.000 description 1
- 108700015053 epidermal growth factor receptor activity proteins Proteins 0.000 description 1
- HHFAWKCIHAUFRX-UHFFFAOYSA-N ethoxide Chemical compound CC[O-] HHFAWKCIHAUFRX-UHFFFAOYSA-N 0.000 description 1
- 125000003916 ethylene diamine group Chemical class 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 210000002744 extracellular matrix Anatomy 0.000 description 1
- 230000002349 favourable effect Effects 0.000 description 1
- 238000003818 flash chromatography Methods 0.000 description 1
- 239000011737 fluorine Substances 0.000 description 1
- 239000011724 folic acid Substances 0.000 description 1
- 229960000304 folic acid Drugs 0.000 description 1
- 235000019152 folic acid Nutrition 0.000 description 1
- 239000004052 folic acid antagonist Substances 0.000 description 1
- 150000004675 formic acid derivatives Chemical class 0.000 description 1
- VZCYOOQTPOCHFL-OWOJBTEDSA-L fumarate(2-) Chemical class [O-]C(=O)\C=C\C([O-])=O VZCYOOQTPOCHFL-OWOJBTEDSA-L 0.000 description 1
- 206010017758 gastric cancer Diseases 0.000 description 1
- PUBCCFNQJQKCNC-XKNFJVFFSA-N gastrin-releasingpeptide Chemical compound C([C@@H](C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCSC)C(N)=O)NC(=O)CNC(=O)[C@@H](NC(=O)[C@H](C)NC(=O)[C@H](CC=1C2=CC=CC=C2NC=1)NC(=O)[C@H](CC=1N=CNC=1)NC(=O)[C@H](CC(N)=O)NC(=O)CNC(=O)[C@H](CCCN=C(N)N)NC(=O)[C@H]1N(CCC1)C(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CCSC)NC(=O)[C@H](CCCCN)NC(=O)[C@@H](NC(=O)[C@H](CC(C)C)NC(=O)[C@@H](NC(=O)[C@@H](NC(=O)CNC(=O)CNC(=O)CNC(=O)[C@H](C)NC(=O)[C@H]1N(CCC1)C(=O)[C@H](CC(C)C)NC(=O)[C@H]1N(CCC1)C(=O)[C@@H](N)C(C)C)[C@@H](C)O)C(C)C)[C@@H](C)O)C(C)C)C1=CNC=N1 PUBCCFNQJQKCNC-XKNFJVFFSA-N 0.000 description 1
- 210000001035 gastrointestinal tract Anatomy 0.000 description 1
- 239000000499 gel Substances 0.000 description 1
- 238000001502 gel electrophoresis Methods 0.000 description 1
- 229920000159 gelatin Polymers 0.000 description 1
- 239000008273 gelatin Substances 0.000 description 1
- 235000019322 gelatine Nutrition 0.000 description 1
- 235000011852 gelatine desserts Nutrition 0.000 description 1
- 229940074045 glyceryl distearate Drugs 0.000 description 1
- 229940075507 glyceryl monostearate Drugs 0.000 description 1
- XLXSAKCOAKORKW-UHFFFAOYSA-N gonadorelin Chemical compound C1CCC(C(=O)NCC(N)=O)N1C(=O)C(CCCN=C(N)N)NC(=O)C(CC(C)C)NC(=O)CNC(=O)C(NC(=O)C(CO)NC(=O)C(CC=1C2=CC=CC=C2NC=1)NC(=O)C(CC=1NC=NC=1)NC(=O)C1NC(=O)CC1)CC1=CC=C(O)C=C1 XLXSAKCOAKORKW-UHFFFAOYSA-N 0.000 description 1
- 239000003102 growth factor Substances 0.000 description 1
- 230000009036 growth inhibition Effects 0.000 description 1
- 239000001963 growth medium Substances 0.000 description 1
- 125000001188 haloalkyl group Chemical group 0.000 description 1
- 239000007902 hard capsule Substances 0.000 description 1
- 201000010536 head and neck cancer Diseases 0.000 description 1
- 208000014829 head and neck neoplasm Diseases 0.000 description 1
- 238000003505 heat denaturation Methods 0.000 description 1
- 150000002373 hemiacetals Chemical group 0.000 description 1
- MNWFXJYAOYHMED-UHFFFAOYSA-N heptanoic acid Chemical class CCCCCCC(O)=O MNWFXJYAOYHMED-UHFFFAOYSA-N 0.000 description 1
- 229940022353 herceptin Drugs 0.000 description 1
- 125000000623 heterocyclic group Chemical group 0.000 description 1
- 239000008241 heterogeneous mixture Substances 0.000 description 1
- KKLGDUSGQMHBPB-UHFFFAOYSA-N hex-2-ynedioic acid Chemical class OC(=O)CCC#CC(O)=O KKLGDUSGQMHBPB-UHFFFAOYSA-N 0.000 description 1
- FBPFZTCFMRRESA-UHFFFAOYSA-N hexane-1,2,3,4,5,6-hexol Chemical compound OCC(O)C(O)C(O)C(O)CO FBPFZTCFMRRESA-UHFFFAOYSA-N 0.000 description 1
- FUZZWVXGSFPDMH-UHFFFAOYSA-M hexanoate Chemical compound CCCCCC([O-])=O FUZZWVXGSFPDMH-UHFFFAOYSA-M 0.000 description 1
- 208000033519 human immunodeficiency virus infectious disease Diseases 0.000 description 1
- 150000007857 hydrazones Chemical class 0.000 description 1
- 150000004678 hydrides Chemical class 0.000 description 1
- XMBWDFGMSWQBCA-UHFFFAOYSA-N hydrogen iodide Chemical compound I XMBWDFGMSWQBCA-UHFFFAOYSA-N 0.000 description 1
- 229940071870 hydroiodic acid Drugs 0.000 description 1
- 230000007062 hydrolysis Effects 0.000 description 1
- 238000006460 hydrolysis reaction Methods 0.000 description 1
- 125000002768 hydroxyalkyl group Chemical group 0.000 description 1
- AVXURJPOCDRRFD-UHFFFAOYSA-N hydroxylamine group Chemical group NO AVXURJPOCDRRFD-UHFFFAOYSA-N 0.000 description 1
- 238000003384 imaging method Methods 0.000 description 1
- 150000002463 imidates Chemical class 0.000 description 1
- 229940127121 immunoconjugate Drugs 0.000 description 1
- 102000018358 immunoglobulin Human genes 0.000 description 1
- 125000003392 indanyl group Chemical group C1(CCC2=CC=CC=C12)* 0.000 description 1
- KPYKFQNTASVHIG-UHFFFAOYSA-N indolizine Chemical compound C1=CC=CC2=C=C[CH]N21 KPYKFQNTASVHIG-UHFFFAOYSA-N 0.000 description 1
- 125000001041 indolyl group Chemical group 0.000 description 1
- 239000003701 inert diluent Substances 0.000 description 1
- 238000001802 infusion Methods 0.000 description 1
- 238000009434 installation Methods 0.000 description 1
- 108040003607 interleukin-13 receptor activity proteins Proteins 0.000 description 1
- 210000000936 intestine Anatomy 0.000 description 1
- 230000003834 intracellular effect Effects 0.000 description 1
- 238000007918 intramuscular administration Methods 0.000 description 1
- 239000007928 intraperitoneal injection Substances 0.000 description 1
- 238000007913 intrathecal administration Methods 0.000 description 1
- 150000004694 iodide salts Chemical class 0.000 description 1
- 229910052740 iodine Inorganic materials 0.000 description 1
- 239000011630 iodine Substances 0.000 description 1
- 229910052742 iron Inorganic materials 0.000 description 1
- SURQXAFEQWPFPV-UHFFFAOYSA-L iron(2+) sulfate heptahydrate Chemical compound O.O.O.O.O.O.O.[Fe+2].[O-]S([O-])(=O)=O SURQXAFEQWPFPV-UHFFFAOYSA-L 0.000 description 1
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 1
- KQNPFQTWMSNSAP-UHFFFAOYSA-N isobutyric acid Chemical class CC(C)C(O)=O KQNPFQTWMSNSAP-UHFFFAOYSA-N 0.000 description 1
- QXJSBBXBKPUZAA-UHFFFAOYSA-N isooleic acid Natural products CCCCCCCC=CCCCCCCCCC(O)=O QXJSBBXBKPUZAA-UHFFFAOYSA-N 0.000 description 1
- 125000001972 isopentyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 125000002183 isoquinolinyl group Chemical group C1(=NC=CC2=CC=CC=C12)* 0.000 description 1
- NLYAJNPCOHFWQQ-UHFFFAOYSA-N kaolin Chemical compound O.O.O=[Al]O[Si](=O)O[Si](=O)O[Al]=O NLYAJNPCOHFWQQ-UHFFFAOYSA-N 0.000 description 1
- 201000010982 kidney cancer Diseases 0.000 description 1
- 150000003893 lactate salts Chemical class 0.000 description 1
- 239000002523 lectin Substances 0.000 description 1
- 208000032839 leukemia Diseases 0.000 description 1
- 230000000670 limiting effect Effects 0.000 description 1
- 229960005015 local anesthetics Drugs 0.000 description 1
- 239000007937 lozenge Substances 0.000 description 1
- 239000000314 lubricant Substances 0.000 description 1
- 201000005296 lung carcinoma Diseases 0.000 description 1
- 210000001165 lymph node Anatomy 0.000 description 1
- 210000004698 lymphocyte Anatomy 0.000 description 1
- 235000019359 magnesium stearate Nutrition 0.000 description 1
- 229910052943 magnesium sulfate Inorganic materials 0.000 description 1
- PCTLYGKHPBZRCZ-UKLTVQOFSA-N maitotoxin-3 Chemical compound C[C@H]1CC[C@H]2O[C@@]3(C)C[C@H]4O[C@H]5C[C@H]6O[C@](O)(C[C@@H](O)CO)[C@@H](O)[C@@H](OS(O)(=O)=O)[C@@H]6O[C@@H]5C=C[C@@H]4O[C@@H]3CC[C@@H]2O[C@@H]2C[C@@H]3O[C@]4(C)CC(=C)C[C@](C)(O[C@H]4C[C@H]3O[C@@H]12)[C@@H](O)CC(=O)CC\C=C(/C)C=C PCTLYGKHPBZRCZ-UKLTVQOFSA-N 0.000 description 1
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 description 1
- 239000011976 maleic acid Substances 0.000 description 1
- 150000002688 maleic acid derivatives Chemical class 0.000 description 1
- 125000005439 maleimidyl group Chemical group C1(C=CC(N1*)=O)=O 0.000 description 1
- 208000015486 malignant pancreatic neoplasm Diseases 0.000 description 1
- 150000002690 malonic acid derivatives Chemical class 0.000 description 1
- 238000000816 matrix-assisted laser desorption--ionisation Methods 0.000 description 1
- 238000001840 matrix-assisted laser desorption--ionisation time-of-flight mass spectrometry Methods 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- 230000007246 mechanism Effects 0.000 description 1
- 239000002609 medium Substances 0.000 description 1
- 201000001441 melanoma Diseases 0.000 description 1
- 125000005341 metaphosphate group Chemical group 0.000 description 1
- 230000009401 metastasis Effects 0.000 description 1
- AFVFQIVMOAPDHO-UHFFFAOYSA-M methanesulfonate group Chemical class CS(=O)(=O)[O-] AFVFQIVMOAPDHO-UHFFFAOYSA-M 0.000 description 1
- NBTOZLQBSIZIKS-UHFFFAOYSA-N methoxide Chemical compound [O-]C NBTOZLQBSIZIKS-UHFFFAOYSA-N 0.000 description 1
- WCYWZMWISLQXQU-UHFFFAOYSA-N methyl Chemical class [CH3] WCYWZMWISLQXQU-UHFFFAOYSA-N 0.000 description 1
- YGDGZDGRCWHDOU-UHFFFAOYSA-N methyl 4-[[5-chloro-4-(2-hydroxyphenyl)thiophen-2-yl]sulfonylamino]-2-hydroxybenzoate Chemical compound C1=C(O)C(C(=O)OC)=CC=C1NS(=O)(=O)C1=CC(C=2C(=CC=CC=2)O)=C(Cl)S1 YGDGZDGRCWHDOU-UHFFFAOYSA-N 0.000 description 1
- QPJVMBTYPHYUOC-UHFFFAOYSA-N methyl benzoate Chemical class COC(=O)C1=CC=CC=C1 QPJVMBTYPHYUOC-UHFFFAOYSA-N 0.000 description 1
- 150000004702 methyl esters Chemical group 0.000 description 1
- CPZBTYRIGVOOMI-UHFFFAOYSA-N methylsulfanyl(methylsulfanylmethoxy)methane Chemical compound CSCOCSC CPZBTYRIGVOOMI-UHFFFAOYSA-N 0.000 description 1
- 239000010445 mica Substances 0.000 description 1
- 229910052618 mica group Inorganic materials 0.000 description 1
- 238000012737 microarray-based gene expression Methods 0.000 description 1
- 238000012544 monitoring process Methods 0.000 description 1
- 125000002757 morpholinyl group Chemical group 0.000 description 1
- 238000012243 multiplex automated genomic engineering Methods 0.000 description 1
- 201000000050 myeloid neoplasm Diseases 0.000 description 1
- YOHYSYJDKVYCJI-UHFFFAOYSA-N n-[3-[[6-[3-(trifluoromethyl)anilino]pyrimidin-4-yl]amino]phenyl]cyclopropanecarboxamide Chemical compound FC(F)(F)C1=CC=CC(NC=2N=CN=C(NC=3C=C(NC(=O)C4CC4)C=CC=3)C=2)=C1 YOHYSYJDKVYCJI-UHFFFAOYSA-N 0.000 description 1
- OWIUPIRUAQMTTK-UHFFFAOYSA-M n-aminocarbamate Chemical compound NNC([O-])=O OWIUPIRUAQMTTK-UHFFFAOYSA-M 0.000 description 1
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000001280 n-hexyl group Chemical group C(CCCCC)* 0.000 description 1
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- PSZYNBSKGUBXEH-UHFFFAOYSA-N naphthalene-1-sulfonic acid Chemical class C1=CC=C2C(S(=O)(=O)O)=CC=CC2=C1 PSZYNBSKGUBXEH-UHFFFAOYSA-N 0.000 description 1
- 125000001624 naphthyl group Chemical group 0.000 description 1
- 229930014626 natural product Natural products 0.000 description 1
- 125000001971 neopentyl group Chemical group [H]C([*])([H])C(C([H])([H])[H])(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 125000006574 non-aromatic ring group Chemical group 0.000 description 1
- 231100000344 non-irritating Toxicity 0.000 description 1
- UMRZSTCPUPJPOJ-KNVOCYPGSA-N norbornane Chemical compound C1C[C@H]2CC[C@@H]1C2 UMRZSTCPUPJPOJ-KNVOCYPGSA-N 0.000 description 1
- GYCKQBWUSACYIF-UHFFFAOYSA-N o-hydroxybenzoic acid ethyl ester Natural products CCOC(=O)C1=CC=CC=C1O GYCKQBWUSACYIF-UHFFFAOYSA-N 0.000 description 1
- QIQXTHQIDYTFRH-UHFFFAOYSA-N octadecanoic acid Chemical compound CCCCCCCCCCCCCCCCCC(O)=O QIQXTHQIDYTFRH-UHFFFAOYSA-N 0.000 description 1
- OQCDKBAXFALNLD-UHFFFAOYSA-N octadecanoic acid Natural products CCCCCCCC(C)CCCCCCCCC(O)=O OQCDKBAXFALNLD-UHFFFAOYSA-N 0.000 description 1
- WWZKQHOCKIZLMA-UHFFFAOYSA-M octanoate Chemical class CCCCCCCC([O-])=O WWZKQHOCKIZLMA-UHFFFAOYSA-M 0.000 description 1
- ZQPPMHVWECSIRJ-KTKRTIGZSA-N oleic acid Chemical compound CCCCCCCC\C=C/CCCCCCCC(O)=O ZQPPMHVWECSIRJ-KTKRTIGZSA-N 0.000 description 1
- 238000011275 oncology therapy Methods 0.000 description 1
- 210000000056 organ Anatomy 0.000 description 1
- 125000000962 organic group Chemical group 0.000 description 1
- 239000003960 organic solvent Substances 0.000 description 1
- 125000001181 organosilyl group Chemical group [SiH3]* 0.000 description 1
- 150000002905 orthoesters Chemical group 0.000 description 1
- 150000003891 oxalate salts Chemical class 0.000 description 1
- 235000006408 oxalic acid Nutrition 0.000 description 1
- 150000002923 oximes Chemical class 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- 125000000636 p-nitrophenyl group Chemical group [H]C1=C([H])C(=C([H])C([H])=C1*)[N+]([O-])=O 0.000 description 1
- 201000002528 pancreatic cancer Diseases 0.000 description 1
- 208000008443 pancreatic carcinoma Diseases 0.000 description 1
- 239000012188 paraffin wax Substances 0.000 description 1
- 125000000538 pentafluorophenyl group Chemical group FC1=C(F)C(F)=C(*)C(F)=C1F 0.000 description 1
- 125000001147 pentyl group Chemical group C(CCCC)* 0.000 description 1
- 229940066842 petrolatum Drugs 0.000 description 1
- 235000019271 petrolatum Nutrition 0.000 description 1
- DYUMLJSJISTVPV-UHFFFAOYSA-N phenyl propanoate Chemical class CCC(=O)OC1=CC=CC=C1 DYUMLJSJISTVPV-UHFFFAOYSA-N 0.000 description 1
- WLJVXDMOQOGPHL-UHFFFAOYSA-N phenylacetic acid Chemical class OC(=O)CC1=CC=CC=C1 WLJVXDMOQOGPHL-UHFFFAOYSA-N 0.000 description 1
- 229960005190 phenylalanine Drugs 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 1
- 239000010452 phosphate Substances 0.000 description 1
- 229960004838 phosphoric acid Drugs 0.000 description 1
- 150000003013 phosphoric acid derivatives Chemical class 0.000 description 1
- 125000005498 phthalate group Chemical class 0.000 description 1
- 239000006187 pill Substances 0.000 description 1
- 125000004193 piperazinyl group Chemical group 0.000 description 1
- 125000003386 piperidinyl group Chemical group 0.000 description 1
- 229940012957 plasmin Drugs 0.000 description 1
- 229920000889 poly(m-phenylene isophthalamide) Polymers 0.000 description 1
- 229920001481 poly(stearyl methacrylate) Polymers 0.000 description 1
- 229920000573 polyethylene Polymers 0.000 description 1
- 239000000244 polyoxyethylene sorbitan monooleate Substances 0.000 description 1
- 235000010482 polyoxyethylene sorbitan monooleate Nutrition 0.000 description 1
- 229920000053 polysorbate 80 Polymers 0.000 description 1
- 239000001267 polyvinylpyrrolidone Substances 0.000 description 1
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 description 1
- 229920000036 polyvinylpyrrolidone Polymers 0.000 description 1
- 239000011736 potassium bicarbonate Substances 0.000 description 1
- 235000015497 potassium bicarbonate Nutrition 0.000 description 1
- 229910000028 potassium bicarbonate Inorganic materials 0.000 description 1
- 229910000027 potassium carbonate Inorganic materials 0.000 description 1
- 235000011181 potassium carbonates Nutrition 0.000 description 1
- TYJJADVDDVDEDZ-UHFFFAOYSA-M potassium hydrogencarbonate Chemical compound [K+].OC([O-])=O TYJJADVDDVDEDZ-UHFFFAOYSA-M 0.000 description 1
- 230000003389 potentiating effect Effects 0.000 description 1
- 239000002243 precursor Substances 0.000 description 1
- 239000000651 prodrug Substances 0.000 description 1
- 229940002612 prodrug Drugs 0.000 description 1
- KCXFHTAICRTXLI-UHFFFAOYSA-N propane-1-sulfonic acid Chemical class CCCS(O)(=O)=O KCXFHTAICRTXLI-UHFFFAOYSA-N 0.000 description 1
- UORVCLMRJXCDCP-UHFFFAOYSA-N propynoic acid Chemical class OC(=O)C#C UORVCLMRJXCDCP-UHFFFAOYSA-N 0.000 description 1
- 235000019833 protease Nutrition 0.000 description 1
- 238000002731 protein assay Methods 0.000 description 1
- 230000009145 protein modification Effects 0.000 description 1
- 238000000425 proton nuclear magnetic resonance spectrum Methods 0.000 description 1
- 125000003373 pyrazinyl group Chemical group 0.000 description 1
- MYKHANQTTIRZJK-UHFFFAOYSA-N pyridazine-3,4-dione Chemical compound O=C1C=CN=NC1=O MYKHANQTTIRZJK-UHFFFAOYSA-N 0.000 description 1
- 125000002098 pyridazinyl group Chemical group 0.000 description 1
- 125000004076 pyridyl group Chemical group 0.000 description 1
- 125000000714 pyrimidinyl group Chemical group 0.000 description 1
- 125000000719 pyrrolidinyl group Chemical group 0.000 description 1
- 125000000168 pyrrolyl group Chemical group 0.000 description 1
- 150000003242 quaternary ammonium salts Chemical class 0.000 description 1
- 125000002294 quinazolinyl group Chemical group N1=C(N=CC2=CC=CC=C12)* 0.000 description 1
- 125000002943 quinolinyl group Chemical group N1=C(C=CC2=CC=CC=C12)* 0.000 description 1
- 125000001567 quinoxalinyl group Chemical group N1=C(C=NC2=CC=CC=C12)* 0.000 description 1
- 230000009257 reactivity Effects 0.000 description 1
- 108020003175 receptors Proteins 0.000 description 1
- 102000005962 receptors Human genes 0.000 description 1
- 210000000664 rectum Anatomy 0.000 description 1
- 230000001105 regulatory effect Effects 0.000 description 1
- 230000002441 reversible effect Effects 0.000 description 1
- 206010039073 rheumatoid arthritis Diseases 0.000 description 1
- 238000007363 ring formation reaction Methods 0.000 description 1
- 229950001460 sacituzumab Drugs 0.000 description 1
- YGSDEFSMJLZEOE-UHFFFAOYSA-N salicylic acid Chemical class OC(=O)C1=CC=CC=C1O YGSDEFSMJLZEOE-UHFFFAOYSA-N 0.000 description 1
- 238000003118 sandwich ELISA Methods 0.000 description 1
- CXMXRPHRNRROMY-UHFFFAOYSA-N sebacic acid Chemical class OC(=O)CCCCCCCCC(O)=O CXMXRPHRNRROMY-UHFFFAOYSA-N 0.000 description 1
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 125000003548 sec-pentyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 150000003335 secondary amines Chemical class 0.000 description 1
- 125000000467 secondary amino group Chemical group [H]N([*:1])[*:2] 0.000 description 1
- 229960002718 selenomethionine Drugs 0.000 description 1
- 230000035945 sensitivity Effects 0.000 description 1
- 238000013207 serial dilution Methods 0.000 description 1
- 239000008159 sesame oil Substances 0.000 description 1
- 235000011803 sesame oil Nutrition 0.000 description 1
- 238000003998 size exclusion chromatography high performance liquid chromatography Methods 0.000 description 1
- 239000002002 slurry Substances 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 239000007974 sodium acetate buffer Substances 0.000 description 1
- 235000010413 sodium alginate Nutrition 0.000 description 1
- 239000000661 sodium alginate Substances 0.000 description 1
- 229940005550 sodium alginate Drugs 0.000 description 1
- 229910000030 sodium bicarbonate Inorganic materials 0.000 description 1
- 229910001467 sodium calcium phosphate Inorganic materials 0.000 description 1
- 239000011780 sodium chloride Substances 0.000 description 1
- 235000009518 sodium iodide Nutrition 0.000 description 1
- KSAVQLQVUXSOCR-UHFFFAOYSA-M sodium lauroyl sarcosinate Chemical compound [Na+].CCCCCCCCCCCC(=O)N(C)CC([O-])=O KSAVQLQVUXSOCR-UHFFFAOYSA-M 0.000 description 1
- 239000001488 sodium phosphate Substances 0.000 description 1
- VUFNRPJNRFOTGK-UHFFFAOYSA-M sodium;1-[4-[(2,5-dioxopyrrol-1-yl)methyl]cyclohexanecarbonyl]oxy-2,5-dioxopyrrolidine-3-sulfonate Chemical compound [Na+].O=C1C(S(=O)(=O)[O-])CC(=O)N1OC(=O)C1CCC(CN2C(C=CC2=O)=O)CC1 VUFNRPJNRFOTGK-UHFFFAOYSA-M 0.000 description 1
- RPENMORRBUTCPR-UHFFFAOYSA-M sodium;1-hydroxy-2,5-dioxopyrrolidine-3-sulfonate Chemical class [Na+].ON1C(=O)CC(S([O-])(=O)=O)C1=O RPENMORRBUTCPR-UHFFFAOYSA-M 0.000 description 1
- 239000007901 soft capsule Substances 0.000 description 1
- 238000004611 spectroscopical analysis Methods 0.000 description 1
- 210000000952 spleen Anatomy 0.000 description 1
- 239000007921 spray Substances 0.000 description 1
- 206010041823 squamous cell carcinoma Diseases 0.000 description 1
- 238000010186 staining Methods 0.000 description 1
- 239000008117 stearic acid Substances 0.000 description 1
- 201000011549 stomach cancer Diseases 0.000 description 1
- 239000007929 subcutaneous injection Substances 0.000 description 1
- 238000010254 subcutaneous injection Methods 0.000 description 1
- TYFQFVWCELRYAO-UHFFFAOYSA-N suberic acid Chemical class OC(=O)CCCCCCC(O)=O TYFQFVWCELRYAO-UHFFFAOYSA-N 0.000 description 1
- 238000003883 substance clean up Methods 0.000 description 1
- 238000006467 substitution reaction Methods 0.000 description 1
- 150000003890 succinate salts Chemical class 0.000 description 1
- KZNICNPSHKQLFF-UHFFFAOYSA-N succinimide Chemical class O=C1CCC(=O)N1 KZNICNPSHKQLFF-UHFFFAOYSA-N 0.000 description 1
- 235000000346 sugar Nutrition 0.000 description 1
- LSNNMFCWUKXFEE-UHFFFAOYSA-L sulfite Chemical class [O-]S([O-])=O LSNNMFCWUKXFEE-UHFFFAOYSA-L 0.000 description 1
- 229940124530 sulfonamide Drugs 0.000 description 1
- 150000003456 sulfonamides Chemical class 0.000 description 1
- 125000001273 sulfonato group Chemical group [O-]S(*)(=O)=O 0.000 description 1
- 239000011593 sulfur Substances 0.000 description 1
- 125000004434 sulfur atom Chemical group 0.000 description 1
- 229940032330 sulfuric acid Drugs 0.000 description 1
- 150000003467 sulfuric acid derivatives Chemical class 0.000 description 1
- 239000006228 supernatant Substances 0.000 description 1
- 230000002459 sustained effect Effects 0.000 description 1
- 208000024891 symptom Diseases 0.000 description 1
- 239000006188 syrup Substances 0.000 description 1
- 235000020357 syrup Nutrition 0.000 description 1
- 201000000596 systemic lupus erythematosus Diseases 0.000 description 1
- 101150047061 tag-72 gene Proteins 0.000 description 1
- 239000000454 talc Substances 0.000 description 1
- 235000012222 talc Nutrition 0.000 description 1
- 229910052623 talc Inorganic materials 0.000 description 1
- 150000003892 tartrate salts Chemical class 0.000 description 1
- YLUFRCJDHAVEIX-UHFFFAOYSA-N tert-butyl N-(5-formyl-2-methoxy-4-nitrophenyl)carbamate Chemical compound CC(C)(C)OC(NC(C=C(C=O)C([N+]([O-])=O)=C1)=C1OC)=O YLUFRCJDHAVEIX-UHFFFAOYSA-N 0.000 description 1
- AUJWRCFOXBKTFR-UHFFFAOYSA-N tert-butyl n-[2-(2-hydroxyethyldisulfanyl)ethyl]carbamate Chemical compound CC(C)(C)OC(=O)NCCSSCCO AUJWRCFOXBKTFR-UHFFFAOYSA-N 0.000 description 1
- YSGLXWZKQNQDAI-UHFFFAOYSA-N tert-butyl n-[2-(pyridin-2-yldisulfanyl)ethyl]carbamate Chemical compound CC(C)(C)OC(=O)NCCSSC1=CC=CC=N1 YSGLXWZKQNQDAI-UHFFFAOYSA-N 0.000 description 1
- CWXPZXBSDSIRCS-UHFFFAOYSA-N tert-butyl piperazine-1-carboxylate Chemical compound CC(C)(C)OC(=O)N1CCNCC1 CWXPZXBSDSIRCS-UHFFFAOYSA-N 0.000 description 1
- 125000001973 tert-pentyl group Chemical group [H]C([H])([H])C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 239000002562 thickening agent Substances 0.000 description 1
- SRVJKTDHMYAMHA-WUXMJOGZSA-N thioacetazone Chemical compound CC(=O)NC1=CC=C(\C=N\NC(N)=S)C=C1 SRVJKTDHMYAMHA-WUXMJOGZSA-N 0.000 description 1
- MXFNPNPWOIRFCE-UHFFFAOYSA-N thiomorpholin-3-ylmethanol Chemical compound OCC1CSCCN1 MXFNPNPWOIRFCE-UHFFFAOYSA-N 0.000 description 1
- 210000001541 thymus gland Anatomy 0.000 description 1
- 238000004448 titration Methods 0.000 description 1
- 230000000699 topical effect Effects 0.000 description 1
- 229960000303 topotecan Drugs 0.000 description 1
- 235000010487 tragacanth Nutrition 0.000 description 1
- 239000000196 tragacanth Substances 0.000 description 1
- 229940116362 tragacanth Drugs 0.000 description 1
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 1
- 239000012581 transferrin Substances 0.000 description 1
- PYHOFAHZHOBVGV-UHFFFAOYSA-N triazane Chemical compound NNN PYHOFAHZHOBVGV-UHFFFAOYSA-N 0.000 description 1
- 125000004306 triazinyl group Chemical group 0.000 description 1
- FAQYAMRNWDIXMY-UHFFFAOYSA-N trichloroborane Chemical compound ClB(Cl)Cl FAQYAMRNWDIXMY-UHFFFAOYSA-N 0.000 description 1
- RYFMWSXOAZQYPI-UHFFFAOYSA-K trisodium phosphate Chemical compound [Na+].[Na+].[Na+].[O-]P([O-])([O-])=O RYFMWSXOAZQYPI-UHFFFAOYSA-K 0.000 description 1
- 239000012588 trypsin Substances 0.000 description 1
- 230000004565 tumor cell growth Effects 0.000 description 1
- 239000003981 vehicle Substances 0.000 description 1
- 230000003612 virological effect Effects 0.000 description 1
- 239000011782 vitamin Substances 0.000 description 1
- 235000013343 vitamin Nutrition 0.000 description 1
- 229940088594 vitamin Drugs 0.000 description 1
- 229930003231 vitamin Natural products 0.000 description 1
- GDJZZWYLFXAGFH-UHFFFAOYSA-M xylenesulfonate group Chemical group C1(C(C=CC=C1)C)(C)S(=O)(=O)[O-] GDJZZWYLFXAGFH-UHFFFAOYSA-M 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/12—Antivirals
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/68—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment
- A61K47/6801—Drug-antibody or immunoglobulin conjugates defined by the pharmacologically or therapeutically active agent
- A61K47/6803—Drugs conjugated to an antibody or immunoglobulin, e.g. cisplatin-antibody conjugates
- A61K47/68037—Drugs conjugated to an antibody or immunoglobulin, e.g. cisplatin-antibody conjugates the drug being a camptothecin [CPT] or derivatives
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/68—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment
- A61K47/6835—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site
- A61K47/6851—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site the antibody targeting a determinant of a tumour cell
- A61K47/6855—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site the antibody targeting a determinant of a tumour cell the tumour determinant being from breast cancer cell
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/68—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment
- A61K47/6835—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site
- A61K47/6875—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site the antibody being a hybrid immunoglobulin
- A61K47/6879—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site the antibody being a hybrid immunoglobulin the immunoglobulin having two or more different antigen-binding sites, e.g. bispecific or multispecific immunoglobulin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/68—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment
- A61K47/6889—Conjugates wherein the antibody being the modifying agent and wherein the linker, binder or spacer confers particular properties to the conjugates, e.g. peptidic enzyme-labile linkers or acid-labile linkers, providing for an acid-labile immuno conjugate wherein the drug may be released from its antibody conjugated part in an acidic, e.g. tumoural or environment
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/02—Immunomodulators
- A61P37/06—Immunosuppressants, e.g. drugs for graft rejection
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D491/00—Heterocyclic compounds containing in the condensed ring system both one or more rings having oxygen atoms as the only ring hetero atoms and one or more rings having nitrogen atoms as the only ring hetero atoms, not provided for by groups C07D451/00 - C07D459/00, C07D463/00, C07D477/00 or C07D489/00
- C07D491/22—Heterocyclic compounds containing in the condensed ring system both one or more rings having oxygen atoms as the only ring hetero atoms and one or more rings having nitrogen atoms as the only ring hetero atoms, not provided for by groups C07D451/00 - C07D459/00, C07D463/00, C07D477/00 or C07D489/00 in which the condensed system contains four or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/08—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from viruses
- C07K16/10—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from viruses from RNA viruses
- C07K16/1027—Paramyxoviridae, e.g. respiratory syncytial virus
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/32—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against translation products of oncogenes
Definitions
- camptothecin is a natural product that inhibits topoisomerase I and has broad spectrum anti-tumor activity. Camptothecin, however, is poorly soluble making it unsuitable for clinical development. As such, considerable effort has been directed towards identifying analogues or derivatives of camptothecin with properties more suitable for therapeutic use.
- Irinotecan is a prodrug, which is converted in vivo into SN-38, a more potent analogue.
- a third derivative, belotecan has been approved in Korea.
- Camptothecin analogues have also been developed as payloads for antibody-drug conjugates (ADCs). Two such ADCs have been approved for treatment of cancer.
- the present disclosure relates to a compound having Formula (I): wherein: R 1 is selected from: -H, -CH 3 , -CHF 2 , -CF 3 , -F, -Br, -Cl, -OH, -OCH 3 , -OCF 3 and - NH 2 , and R 2 is selected from: -H, -CH 3 , -CF 3 , -F, -Br, -Cl, -OH, -OCH 3 and -OCF 3 , and wherein: when R 1 is -NH 2 , then R is R 3 or R 4 , and when R 1 is other than -NH 2 , then R is R 4 ; R 3 is selected from: -H, -C 1 -C 6 alkyl, -C 3 -C 8 cycloalkyl, -(C 1 -C 6 alkyl)-O-R 5 , , -CO 2 R 8 ,
- R 5 is selected from: -H, -C 1 -C 6 alkyl, -C 3 -C 8 cycloalkyl, -aryl, -heteroaryl, -aryl and –(C 1 -C 6 alkyl)-aryl;
- R 6 and R 7 are each independently selected from: -H, -C 1 -C 6 alkyl, -C 3 -C 8 cycloalkyl, -(C 1 -C 6 alkyl)-O-R 5 , -C 3 -C 8 heterocycloalkyl and -C(O)R 17 ;
- R 8 is selected from: -H, -C 1 -C 6 alkyl, -C 3 -C 8 cycloalkyl and -C 3 -C 8 heterocycloalkyl;
- each R 9 is independently selected from: -H, -C 1 -C 6 alkyl, -C 3 -C 8 cycloalky
- R 1 is NH 2
- R 2 is other than -H.
- Another aspect of the present disclosure relates to a pharmaceutical composition comprising a compound having Formula (I), and a pharmaceutically acceptable carrier or diluent.
- Another aspect of the present disclosure relates to a method of inhibiting the proliferation of cancer cells comprising contacting the cells with an effective amount of a compound according having Formula (I).
- Another aspect relates to a method of killing cancer cells comprising contacting the cells with an effective amount of a compound having Formula (I).
- Another aspect of the present disclosure relates to a use of a compound having Formula (I) in the manufacture of a medicament for the treatment of cancer, an autoimmune disease or a viral infection.
- Another aspect of the present disclosure relates to a conjugate having Formula (X): T-[L-(D)m]n (X) wherein: T is a targeting moiety; L is a linker; D is a camptothecin analogue as described herein; m is an integer between 1 and 4, and n is an integer between 1 and 10.
- T is a targeting moiety
- L is a linker
- m is an integer between 1 and 4
- n is an integer between 1 and 10
- D is a compound of Formula (IV): wherein: R 1a is selected from: -H, -CH 3 , -CHF 2 , -CF 3 , -F, -Br, -Cl, -OH, -OCH 3 , -OCF 3 and - NH 2
- R 2a is selected from: -H, -CH 3 , -CF 3 , -F, -Br, -Cl, -OH, -OCH 3 and -OCF 3
- X is -O-, -S- or -NH-
- R 4a is selected from: , , , f attachment to X, and wherein p
- T is a targeting moiety
- L is a linker
- m is an integer between 1 and 4
- n is an integer between 1 and 10
- D is a compound of Formula (V):
- R 2a is selected from: -CH 3 , -CF 3 , -F, -Br, -Cl, -OH, -OCH 3 and -OCF 3
- R 20a is selected from: -H, -C 1 -C 6 alkyl, -C 3 -C 8 cycloalkyl, -(C 1 -C 6 alkyl)-O-R 5 , , -CO 2 R 8 , -aryl, -heteroaryl, –(C 1 -C 6 alkyl)-aryl,
- R 5 is selected from: -H, -C 1 -C 6 alkyl, -C 3 -C 8 cycloalkyl, -aryl, -heteroaryl and –(C 1 - C 6 alkyl)-aryl
- R 6 and R 7 are each independently selected from: -H, -C 1 -C 6 alkyl, -C 3 -C 8
- T is a targeting moiety
- L is a linker
- m is an integer between 1 and 4
- n is an integer between 1 and 10
- D is a compound of Formula (VI):
- R 2a is selected from: -H, -CH 3 , -CF 3 , -F, -Br, -Cl, -OH, -OCH 3 and -OCF 3 ;
- X is -O-, -S- or -NH-, and
- R 25 is selected from: -C 1 -C 6 alkyl, -(C 1 -C 6 alkyl)-O-R 5a , - CO 2 R 8a , -C(O)-, -aryl, -heteroaryl,–(C 1 -C 6 alkyl)-aryl, , , wherein * is the point of attachment to X, and wherein p is 1, 2, 3 or 4; or X is O, and R 25 -X- is selected from: R 5a is selected from: -C 1 -C 6 alkyl, –C 3 -C 8 cycloalkyl, –aryl, -heteroaryl and
- Another aspect of the present disclosure relates to a pharmaceutical composition comprising a conjugate of Formula (X), and a pharmaceutically acceptable carrier or diluent.
- Another aspect of the present disclosure relates to a method of inhibiting the proliferation of cancer cells comprising contacting the cells with an effective amount of a conjugate having Formula (X).
- Another aspect relates to a method of killing cancer cells comprising contacting the cells with an effective amount of a conjugate having Formula (X).
- Another aspect of the present disclosure relates to a method of treating cancer in a subject in need thereof comprising administering to the subject an effective amount of a conjugate of Formula (X).
- Another aspect relates to a method of treating an autoimmune disease in a subject in need thereof comprising administering to the subject an effective amount of a conjugate of Formula (X).
- Another aspect relates to a method of treating a viral infection in a subject in need thereof comprising administering to the subject an effective amount of a conjugate of Formula (X).
- Another aspect of the present disclosure relates to a conjugate having Formula (X) for use in therapy.
- Another aspect relates to a conjugate having Formula (X) for use in the treatment of cancer, an autoimmune disease or a viral infection.
- Another aspect of the present disclosure relates to a use of a conjugate having Formula (X) in the manufacture of a medicament for the treatment of cancer, an autoimmune disease or a viral infection.
- Fig.1 presents schematics of general procedures that may be used in the preparation of intermediates for the synthesis of camptothecin analogues and conjugates described herein,
- A Synthetic Scheme I: General Procedure 1;
- B Synthetic Scheme II: General Procedure 2;
- C Synthetic Scheme III: General Procedure 3;
- D Synthetic Scheme IV: General Procedure 4;
- E Synthetic Scheme V: General Procedure 5;
- F Synthetic Scheme VI: General Procedure 7,
- G Synthetic Scheme VII: General Procedure 8.
- Fig. 2 shows the bystander killing effect of conjugates comprising camptothecin analogues described herein conjugated to trastuzumab at DAR 8 on HER2-negative MDA-MB- 468 cancer cells, (A) at 1nM concentration, and (B) 0.1 nM concentration.
- Fig. 3 shows the anti-tumor activity of conjugates comprising camptothecin analogues described herein conjugated to trastuzumab at DAR 8 in a JIMT-1 xenograft model of breast cancer expressing HER2 (mid).
- Fig.4 shows exemplary drug-linker (DL) structures comprising camptothecin analogues of Formula (I) with a C7 linkage (Table 4).
- Fig.5 shows exemplary drug-linker (DL) structures comprising camptothecin analogues of Formula (I) with a C10 linkage (Table 5).
- Fig.6 shows exemplary drug-linker (DL) structures comprising camptothecin analogues of Formula (I) with either a C7 or C10 linkage (Table 6).
- Fig. 7 shows exemplary conjugate (DC) structures comprising camptothecin analogues of Formula (I) with a C7 linkage (Table 7).
- Fig. 8 shows exemplary conjugate (DC) structures comprising camptothecin analogues of Formula (I) with a C10 linkage (Table 8).
- Fig. 8 shows exemplary conjugate (DC) structures comprising camptothecin analogues of Formula (I) with a C10 linkage (Table 8).
- FIG.10A-C shows the in vivo anti-tumor activities of an anti-FR ⁇ antibody v30384 (A) conjugated to the camptothecin analogues Compound 139 and Compound 141 at DAR 8 in an OV90 xenograft model, (B) conjugated to the camptothecin analogues Compound 140 and Compound 141 at DAR 8 in an OV90 xenograft model and (C) conjugated to the camptothecin analogues Compound 139, Compound 140, Compound 141 and Compound 148 at DAR 8 in a H2110 xenograft model.
- camptothecin analogues and conjugates comprising the camptothecin analogues. Camptothecin analogues and conjugates are shown to have cytotoxic activity, for example against cancer cells. Certain embodiments of the present disclosure thus relate to the use of the camptothecin analogues and conjugates as therapeutic agents, particularly in the treatment of cancer.
- compositions, use or method denotes that additional elements and/or method steps may be present, but that these additions do not materially affect the manner in which the recited composition, method or use functions.
- a composition, use or method described herein as comprising certain elements and/or steps may also, in certain embodiments consist essentially of those elements and/or steps, and in other embodiments consist of those elements and/or steps, whether or not these embodiments are specifically referred to.
- acyl refers to the group -C(O)R, where R is hydrogen, alkyl, aryl, heteroaryl, cycloalkyl or heterocycloalkyl.
- acyloxy refers to the group -OC(O)R, where R is alkyl.
- alkoxy refers to the group -OR, where R is alkyl, aryl, heteroaryl, cycloalkyl or cycloheteroalkyl.
- alkyl refers to a straight chain or branched saturated hydrocarbon group containing the specified number of carbon atoms.
- alkyl examples include, but are not limited to, methyl, ethyl, n-propyl, isopropyl, n-butyl, sec-butyl, isobutyl, t-butyl, pentyl, isopentyl, t-pentyl, neo-pentyl, 1-methylbutyl, 2-methylbutyl, n-hexyl, and the like.
- alkylaminoaryl refers to an alkyl group as defined herein substituted with one aminoaryl group as defined herein.
- alkylheterocycloalkyl refers to an alkyl group as defined herein substituted with one heterocycloalkyl group as defined herein.
- alkylthio refers to the group -SR, where R is an alkyl group.
- alkylamido refers to the group -C(O)NRR', where R and R' are independently hydrogen, alkyl, aryl, heteroaryl, cycloalkyl or heterocycloalkyl.
- amino refers to the group -NRR', where R and R' are independently hydrogen, alkyl, aryl, heteroaryl, cycloalkyl or heterocycloalkyl.
- aminoalkyl refers to an alkyl group as defined herein substituted with one or more amino groups, for example, one, two or three amino groups.
- aminoaryl refers to an aryl group as defined herein substituted with one amino group.
- aryl refers to a 6- to 12-membered mono- or bicyclic hydrocarbon ring system in which at least one ring aromatic. Examples of aryl include, but are not limited to, phenyl, naphthalenyl, 1,2,3,4-tetrahydro-naphthalenyl, 5,6,7,8-tetrahydro- naphthalenyl, indanyl, and the like.
- carboxy refers to the group -C(O)OR, where R is H, alkyl, aryl, heteroaryl, cycloalkyl or cycloheteroalkyl.
- cyano refers to the group -CN.
- cycloalkyl refers to a mono- or bicyclic saturated hydrocarbon containing the specified number of carbon atoms. Examples of cycloalkyl include, but are not limited to, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptane, bicyclo [2.2.1] heptane, bicyclo [3.1.1] heptane, and the like.
- haloalkyl refers to an alkyl group as defined herein substituted with one or more halogen atoms.
- halogen and halo refer to fluorine (F), bromine (Br), chlorine (Cl) and iodine (I).
- heteroaryl refers to a 6- to 12-membered mono- or bicyclic ring system in which at least one ring atom is a heteroatom and at least one ring is aromatic. Examples of heteroatoms include, but are not limited to, O, S and N.
- heteroaryl examples include, but are not limited to: pyridyl, benzofuranyl, pyrazinyl, pyridazinyl, pyrimidinyl, triazinyl, quinolinyl, benzoxazolyl, benzothiazolyl, isoquinolinyl, quinazolinyl, quinoxalinyl, pyrrolyl, indolyl, and the like.
- heterocycloalkyl refers to a mono- or bicyclic non-aromatic ring system containing the specified number of atoms and in which at least one ring atom is a heteroatom, for example, O, S or N.
- a heterocyclyl substituent can be attached via any of its available ring atoms, for example, a ring carbon, or a ring nitrogen.
- heterocycloalkyl include, but are not limited to, aziridinyl, azetidinyl, piperidinyl, morpholinyl, piperazinyl, pyrrolidinyl, and the like.
- hydroxy and “hydroxyl,” as used herein, refer to the group -OH.
- hydroxyalkyl refers to an alkyl group as defined herein substituted with one or more hydroxy groups.
- nitro refers to the group -NO 2 .
- sulfonyl refers to the group -S(O) 2 R, where R is H, alkyl or aryl.
- sulfonamido refers to the group -NH-S(O) 2 R, where R is H, alkyl or aryl.
- thio and “thiol,” as used herein, refer to the group -SH.
- each such reference includes both unsubstituted and substituted versions of these groups.
- reference to a “-C 1 -C 6 alkyl” includes both unsubstituted -C 1 -C 6 alkyl and - C 1 -C 6 alkyl substituted with one or more substituents.
- substituents include, but are not limited to, halogen, acyl, acyloxy, alkoxy, carboxy, hydroxy, amino, amido, nitro, cyano, azido, alkylthio, thio, sulfonyl, sulfonamido, alkyl, cycloalkyl, heterocycloalkyl, aryl or heteroaryl.
- each alkyl, cycloalkyl, heterocycloalkyl, aryl or heteroaryl group referred to herein is optionally substituted with one or more substituents selected from: halogen, acyl, acyloxy, alkoxy, carboxy, hydroxy, amino, amido, nitro, cyano, azido, alkylthio, thio, sulfonyl and sulfonamido.
- a chemical group described herein that is “substituted” may include one substituent or a plurality of substituents up to the full valence of substitution for that group.
- a methyl group may include 1, 2, or 3 substituents
- a phenyl group may include 1, 2, 3, 4, or 5 substituents.
- the substituents may be the same or they may be different.
- the terms “subject” and “patient” as used herein refer to an animal in need of treatment.
- An animal in need of treatment may be a human or a non-human animal, such as a mammal, bird or fish.
- the subject or patient is a mammal.
- the subject or patient is a human.
- an “effective amount” of a compound or conjugate described herein in respect of a particular result to be achieved is an amount sufficient to achieve the desired result.
- an “effective amount” of a compound when referred to in respect of the killing of cancer cells refers to an amount of compound sufficient to produce a killing effect.
- the positive recitation of a feature in one embodiment serves as a basis for excluding the feature in an alternative embodiment.
- a list of options is presented for a given embodiment or claim, it is to be understood that one or more option may be deleted from the list and the shortened list may form an alternative embodiment, whether or not such an alternative embodiment is specifically referred to.
- camptothecin analogue compounds of the present disclosure are compounds having Formula (I): wherein: R 1 is selected from: -H, -CH 3 , -CHF 2 , -CF 3 , -F, -Br, -Cl, -OH, -OCH 3 , -OCF 3 and - NH 2 , and R 2 is selected from: -H, -CH 3 , -CF 3 , -F, -Br, -Cl, -OH, -OCH 3 and -OCF 3 , and wherein: when R 1 is -NH 2 , then R is R 3 or R 4 , and when R 1 is other than -NH 2 , then R is R 4 ; R 3 is selected from: -H,
- the camptothecin analogues are compounds of Formula (I), with the proviso that when R 1 is NH 2 , R 2 is other than H.
- R 1 is selected from: -CH 3 , -CF 3 , - OCH 3 , -OCF 3 and NH 2 .
- R 1 is NH 2 in compounds of Formula (I).
- R 1 is selected from: -H, -CH 3 , -CF 3 , -F, -Br, -Cl, -OH, -OCH 3 and -OCF 3 .
- R 1 is selected from: -CH 3 , -CF 3 , - OCH 3 and -OCF 3 .
- R 2 is selected from: -H, -CH 3 , -CF 3 , -F, -Cl, -OCH 3 and -OCF 3 .
- R 2 is selected from: -CH 3 , -CF 3 , -F, -Cl, -OCH 3 and -OCF 3 .
- R 2 is selected from: -H, -F, -Br and -Cl.
- R 2 is selected from: -F, -Br and -Cl.
- R 3 is selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 3 -C 8 cycloalkyl, -(C 1 -C 6 -aminoaryl, -heteroaryl and –(C 1 -C 6 alkyl)- aminoaryl.
- R 4 is selected from: , , [0080]
- R 5 is selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, -C 3 -C 8 cycloalkyl, unsubstituted -aryl, -aminoaryl, -heteroaryl and –(C 1 -C 6 alkyl)-aminoaryl.
- R 6 and R 7 are each independently selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 3 -C 8 cycloalkyl, -(C 1 -C 6 alkyl)-O-R 5 , -C 3 -C 8 heterocycloalkyl and -C(O)R 17 .
- R 8 is selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, -C 3 -C 8 cycloalkyl and -C 3 -C 8 heterocycloalkyl.
- each R 9 is independently selected from: -C 1 -C 6 alkyl, -C 3 -C 8 cycloalkyl, -aryl and –(C 1 -C 6 alkyl)-aryl.
- each R 9 is independently selected from: -C 1 -C 6 alkyl and –(C 1 -C 6 alkyl)-aryl. [0085] In some embodiments, in compounds of Formula (I), each R 9 is independently selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, - C 3 -C 8 cycloalkyl, unsubstituted -aryl, -aminoaryl, -heteroaryl and –(C 1 -C 6 alkyl)-aminoaryl.
- each R 10 is independently selected from: -C 1 -C 6 alkyl, -C 3 -C 8 cycloalkyl, -NR 14 R 14’ , -aryl and –(C 1 -C 6 alkyl)-aryl.
- each R 10 is independently selected from: -C 1 -C 6 alkyl, -NR 14 R 14’ , -aryl and –(C 1 -C 6 alkyl)-aryl.
- each R 10 is independently selected from: unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, -C 3 - C 8 cycloalkyl, -NR 14 R 14’ , unsubstituted -aryl, -aminoaryl, -heteroaryl and –(C 1 -C 6 alkyl)-aryl.
- each R 10’ is independently selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, - C 3 -C 8 cycloalkyl, unsubstituted -aryl, -aminoaryl, -heteroaryl and –(C 1 -C 6 alkyl)-aryl.
- R 11 is selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl and -C 1 -C 6 aminoalkyl.
- R 12 is selected from: -H, -C 1 -C 6 alkyl, -CO 2 R 8 , -aryl, –(C 1 -C 6 alkyl)-aryl and -S(O) 2 R 16 .
- R 12 is selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, -CO 2 R 8 , unsubstituted -aryl, -aminoaryl, -heteroaryl, –(C 1 -C 6 alkyl)-aminoaryl, -S(O) 2 R 16 and .
- R 13 is selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl and -C 1 -C 6 aminoalkyl.
- R 14 and R 14’ are each independently selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, -C 3 -C 8 cycloalkyl and -C 3 -C 8 heterocycloalkyl.
- R 16 is selected from: -aryl, - heteroaryl and –(C 1 -C 6 alkyl)-aryl.
- R 16 is selected from: unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, -C 3 -C 8 cycloalkyl, unsubstituted aryl, -aminoaryl, -heteroaryl and –(C 1 -C 6 alkyl)-aminoaryl.
- R 17 is selected from: unsubstituted C 1 -C 6 alkyl, -C 1 -C 6 hydroxyalkyl, -C 3 -C 8 cycloalkyl, -C 3 -C 8 heterocycloalkyl, –(C 1 -C 6 alkyl)-C 3 - C 8 heterocycloalkyl, unsubstituted aryl, -hydroxyaryl, -aminoaryl, -heteroaryl and –(C 1 -C 6 alkyl)- aminoaryl.
- R 18 and R 19 taken together with the N atom to which they are bonded form a 4-, 5-, 6- or 7-membered ring having 0 to 3 substituents selected from: halogen, unsubstituted C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, -C 3 -C 8 cycloalkyl and -(C 1 -C 6 alkyl)-O-R 5 .
- X a and X b are each independently selected from: NH and O.
- X a and X b are each independently selected from: NH and O.
- the compound of Formula (I) has Formula (Ia): wherein: R 1 , R 2 , R 4 , R 5 , R 8 , R 9 , R 10 , R 10’ , R 11 , R 12 , R 13 , R 14 , R 14’ , R 16 , R 18 , R 19 , X a , X b and X c are as defined for Formula (I).
- R 1 is selected from: -CH 3 , -CF 3 , - OCH 3 , -OCF 3 and -NH 2 .
- R 2 is selected from: -H, -CH 3 , - CF 3 , -F, -Cl, -OCH 3 and -OCF 3 .
- R 2 is selected from: -H, -F and - Cl.
- R 4 is selected from: , [00106]
- R 5 is selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, -C 3 -C 8 cycloalkyl, unsubstituted -aryl, -aminoaryl, -heteroaryl and –(C 1 -C 6 alkyl)-aminoaryl.
- R 8 is selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, -C 3 -C 8 cycloalkyl and -C 3 -C 8 heterocycloalkyl.
- each R 9 is independently selected from: -C 1 -C 6 alkyl, -C 3 -C 8 cycloalkyl, -aryl and –(C 1 -C 6 alkyl)-aryl.
- each R 9 is independently selected from: -C 1 -C 6 alkyl and –(C 1 -C 6 alkyl)-aryl.
- each R 9 is independently selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, - C 3 -C 8 cycloalkyl, unsubstituted -aryl, -aminoaryl, -heteroaryl and –(C 1 -C 6 alkyl)-aminoaryl.
- each R 10 is independently selected from: -C 1 -C 6 alkyl, -C 3 -C 8 cycloalkyl, -NR 14 R 14’ , -aryl and –(C 1 -C 6 alkyl)-aryl.
- each R 10 is independently selected from: -C 1 -C 6 alkyl, -NR 14 R 14’ , -aryl and –(C 1 -C 6 alkyl)-aryl.
- each R 10 is independently selected from: unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, -C 3 - C 8 cycloalkyl, -NR 14 R 14’ , unsubstituted -aryl, -aminoaryl, -heteroaryl and –(C 1 -C 6 alkyl)-aryl.
- each R 10’ is independently selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, - C 3 -C 8 cycloalkyl, unsubstituted -aryl, -aminoaryl, -heteroaryl and –(C 1 -C 6 alkyl)-aryl.
- R 11 is selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl and -C 1 -C 6 aminoalkyl.
- R 12 is selected from: -H, -C 1 -C 6 alkyl, -CO 2 R 8 , -aryl and –(C 1 -C 6 alkyl)-aryl and -S(O) 2 R 16 .
- R 12 is selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, -CO 2 R 8 , unsubstituted -aryl, -aminoaryl, -heteroaryl, –(C 1 -C 6 alkyl)-aminoaryl, -S(O) 2 R 16 and .
- R 13 is selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl and -C 1 -C 6 aminoalkyl.
- R 14 and R 14’ are each independently selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, -C 3 -C 8 cycloalkyl and -C 3 -C 8 heterocycloalkyl.
- R 16 is selected from: -aryl, - heteroaryl and –(C 1 -C 6 alkyl)-aryl.
- R 16 is selected from: unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, -C 3 -C 8 cycloalkyl, unsubstituted -aryl, -aminoaryl, -heteroaryl and –(C 1 -C 6 alkyl)-aminoaryl.
- R 17 is selected from: unsubstituted C 1 -C 6 alkyl, -C 1 -C 6 hydroxyalkyl, -C 3 -C 8 cycloalkyl, -C 3 -C 8 heterocycloalkyl, –(C 1 -C 6 alkyl)-C 3 - C 8 heterocycloalkyl, unsubstituted aryl, -hydroxyaryl, -aminoaryl, -heteroaryl and –(C 1 -C 6 alkyl)- aminoaryl.
- R 18 and R 19 taken together with the N atom to which they are bonded form a 4-, 5-, 6- or 7-membered ring having 0 to 3 substituents selected from: halogen, unsubstituted C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, -C 3 -C 8 cycloalkyl and -(C 1 -C 6 alkyl)-O-R 5 .
- X a and X b are each independently selected from: NH and O.
- Combinations of any of the foregoing embodiments for compounds of Formula (Ia) are also contemplated and each combination forms a separate embodiment for the purposes of the present disclosure.
- the compound of Formula (I) has Formula (II): wherein: R 2 is selected from: -H, -CH 3 , -CF 3 , -F, -Br, -Cl, -OH, -OCH 3 and -OCF 3 ; R 20 is selected from: -H, -C 1 -C 6 alkyl, -C 3 -C 8 cycloalkyl, -(C 1 -C 6 alkyl)-O-R 5 , , -CO 2 R 8 , -aryl, -heteroaryl,–(C 1 -C 6 alkyl)-aryl, , R 5 is selected from: -H, -C 1 -C 6 alkyl, -C 3 -C 8 cycloalkyl, -aryl, -heteroaryl and –(C 1 - C 6 alkyl)-aryl; R 6 and R 7 are each independently selected from:
- R 2 is selected from: -CH 3 , -CF 3 , - F, -Br, -Cl, -OH, -OCH 3 and -OCF 3 .
- R 2 is selected from: -CH 3 , -CF 3 , - F, -Cl, -OCH 3 and -OCF 3 .
- R 2 is selected from F and Cl.
- R 20 is selected from: -H, -C 1 -C 6 [00131] In some embodiments, in compounds of Formula (II), R 20 is selected from: -H, -C 1 -C 6 , [00132] In some embodiments, in compounds of Formula (II), R 20 is selected from: -H, -C 1 -C 6 , [00133] In some embodiments, in compounds of Formula (II), R 20 is selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 3 -C 8 cycloalkyl, -(C 1 -C 6 alkyl)-O- -CO 2 R 8 , unsubstituted aryl, -aminoaryl, -heteroaryl, –(C 1 -
- R 6 and R 7 are each independently selected from: -H, -C 1 -C 6 alkyl, -C 3 -C 8 cycloalkyl and -C(O)R 17 .
- R 6 is H
- R 7 is selected from: - H, -C 1 -C 6 alkyl, -C 3 -C 8 cycloalkyl, -(C 1 -C 6 alkyl)-O-R 5 , -C 3 -C 8 heterocycloalkyl and -C(O)R 17 .
- R 6 is H
- R 7 is selected from: - H, -C 1 -C 6 alkyl, -C 3 -C 8 cycloalkyl and -C(O)R 17 .
- R 6 and R 7 are each independently selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 3 -C 8 cycloalkyl, -(C 1 -C 6 alkyl)-O-R 5 , -C 3 -C 8 heterocycloalkyl and -C(O)R 17 .
- R 8 is selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, -C 3 -C 8 cycloalkyl and -C 3 -C 8 heterocycloalkyl.
- each R 9 is independently selected from: -C 1 -C 6 alkyl, -C 3 -C 8 cycloalkyl, -aryl and –(C 1 -C 6 alkyl)-aryl.
- each R 9 is independently selected from: -C 1 -C 6 alkyl and –(C 1 -C 6 alkyl)-aryl.
- each R 9 is independently selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, - C 3 -C 8 cycloalkyl, unsubstituted -aryl, -aminoaryl, -heteroaryl and –(C 1 -C 6 alkyl)-aminoaryl.
- each R 10 is independently selected from: -C 1 -C 6 alkyl, -C 3 -C 8 cycloalkyl, -NR 14 R 14’ , -aryl and –(C 1 -C 6 alkyl)-aryl.
- each R 10 is independently selected from: -C 1 -C 6 alkyl, -NR 14 R 14’ , -aryl and –(C 1 -C 6 alkyl)-aryl.
- each R 10 is independently selected from: unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, -C 3 - C 8 cycloalkyl, -NR 14 R 14’ , unsubstituted -aryl, -aminoaryl, -heteroaryl and –(C 1 -C 6 alkyl)-aryl.
- each R 10’ is independently selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, - C 3 -C 8 cycloalkyl, unsubstituted -aryl, -aminoaryl, -heteroaryl and –(C 1 -C 6 alkyl)-aryl.
- R 11 is selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl and -C 1 -C 6 aminoalkyl.
- R 12 is selected from: -H, -C 1 -C 6 alkyl, -CO 2 R 8 , -aryl, –(C 1 -C 6 alkyl)-aryl and -S(O) 2 R 16 .
- R 12 is selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, -CO 2 R 8 , unsubstituted -aryl, -aminoaryl, -heteroaryl, –(C 1 -C 6 alkyl)-aminoaryl, -S(O) 2 R 16 and .
- R 13 is selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl and -C 1 -C 6 aminoalkyl.
- R 14 and R 14’ are each independently selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, -C 3 -C 8 cycloalkyl and -C 3 -C 8 heterocycloalkyl.
- R 16 is selected from: -aryl, - heteroaryl and –(C 1 -C 6 alkyl)-aryl.
- R 16 is selected from: unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, -C 3 -C 8 cycloalkyl, unsubstituted -aryl, -aminoaryl, -heteroaryl and –(C 1 -C 6 alkyl)-aminoaryl.
- R 17 is -C 1 -C 6 alkyl.
- R 17 is selected from: unsubstituted C 1 -C 6 alkyl, -C 1 -C 6 hydroxyalkyl, -C 3 -C 8 cycloalkyl, -C 3 -C 8 heterocycloalkyl, –(C 1 -C 6 alkyl)-C 3 - C 8 heterocycloalkyl, unsubstituted aryl, -hydroxyaryl, -aminoaryl, -heteroaryl and –(C 1 -C 6 alkyl)- aminoaryl.
- R 18 and R 19 taken together with the N atom to which they are bonded form a 4-, 5-, 6- or 7-membered ring having 0 to 3 substituents selected from: halogen, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, -C 3 -C 8 cycloalkyl and -(C 1 -C 6 alkyl)-O-R 5 .
- X a and X b are each independently selected from: NH and O.
- X a and X b are each independently selected from: NH and O.
- the compound of Formula (I) has Formula (IIa): wherein: R 20 , R 5 , R 6 , R 7 , R 8 , R 9 , R 10 , R 10’ , R 11 , R 12 , R 13 , R 14 , R 14’ , R 16 , R 17 , R 18 , R 19 , X a , X b and X c are as defined for Formula (II).
- R 20 is selected from: -H, -C 1 -C 6 alkyl, -(C 1 -C 6 alkyl)-O-R 5 , , –(C 1 -C 6 alkyl)-aryl, , .
- R 20 is selected from: -H, -C 1 -C 6 alkyl, -(C 1 -C 6 alkyl)-O-R 5 , –(C 1 -C 6 alkyl)-aryl, [00165] In some embodiments, in compounds of Formula (IIa), R 20 is selected from: -H, -C 1 -C 6 [00166] In some embodiments, in compounds of Formula (IIa), R 20 is selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 3 -C 8 cycloalkyl, -(C 1 -C 6 alkyl)-O-R 5 , -CO 2 R 8 , unsubstituted aryl, -aminoaryl, -heteroary
- R 5 is selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, -C 3 -C 8 cycloalkyl, unsubstituted -aryl, -aminoaryl, -heteroaryl and –(C 1 -C 6 alkyl)-aminoaryl.
- R 6 and R 7 are each independently selected from: -H, -C 1 -C 6 alkyl, -C 3 -C 8 cycloalkyl and -C(O)R 17 .
- R 6 is H
- R 7 is selected from: -H, -C 1 -C 6 alkyl, -C 3 -C 8 cycloalkyl, -(C 1 -C 6 alkyl)-O-R 5 , -C 3 -C 8 heterocycloalkyl and -C(O)R 17 .
- R 6 is H
- R 7 is selected from: -H, -C 1 -C 6 alkyl, -C 3 -C 8 cycloalkyl and -C(O)R 17 .
- R 6 and R 7 are each independently selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 3 -C 8 cycloalkyl, -(C 1 -C 6 alkyl)-O-R 5 , -C 3 -C 8 heterocycloalkyl and -C(O)R 17 .
- R 8 is selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, -C 3 -C 8 cycloalkyl and -C 3 -C 8 heterocycloalkyl.
- each R 9 is independently selected from: -C 1 -C 6 alkyl, -C 3 -C 8 cycloalkyl, -aryl and –(C 1 -C 6 alkyl)-aryl.
- each R 9 is independently selected from: -C 1 -C 6 alkyl and –(C 1 -C 6 alkyl)-aryl.
- each R 9 is independently selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, - C 3 -C 8 cycloalkyl, unsubstituted -aryl, -aminoaryl, -heteroaryl and –(C 1 -C 6 alkyl)-aminoaryl.
- each R 10 is independently selected from: -C 1 -C 6 alkyl, -C 3 -C 8 cycloalkyl, -NR 14 R 14’ , -aryl and –(C 1 -C 6 alkyl)-aryl.
- each R 10 is independently selected from: -C 1 -C 6 alkyl, -NR 14 R 14’ , -aryl and –(C 1 -C 6 alkyl)-aryl.
- each R 10 is independently selected from: unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, -C 3 - C 8 cycloalkyl, -NR 14 R 14’ , unsubstituted -aryl, -aminoaryl, -heteroaryl and –(C 1 -C 6 alkyl)-aryl.
- each R 10’ is independently selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, -C 3 -C 8 cycloalkyl, unsubstituted -aryl, -aminoaryl, -heteroaryl and –(C 1 -C 6 alkyl)- aryl.
- R 11 is selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl and -C 1 -C 6 aminoalkyl.
- R 12 is selected from: -H, -C 1 -C 6 alkyl, -CO 2 R 8 , -aryl, –(C 1 -C 6 alkyl)-aryl and -S(O) 2 R 16 .
- R 12 is selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, -CO 2 R 8 , unsubstituted -aryl, -aminoaryl, -heteroaryl, –(C 1 -C 6 alkyl)-aminoaryl, -S(O) 2 R 16 and .
- R 13 is selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl and -C 1 -C 6 aminoalkyl.
- R 14 and R 14’ are each independently selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, -C 3 -C 8 cycloalkyl and -C 3 -C 8 heterocycloalkyl.
- R 16 is selected from: -aryl, - heteroaryl and –(C 1 -C 6 alkyl)-aryl.
- R 16 is selected from: unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, -C 3 -C 8 cycloalkyl, unsubstituted -aryl, -aminoaryl, -heteroaryl and –(C 1 -C 6 alkyl)-aminoaryl.
- R 17 is -C 1 -C 6 alkyl.
- R 17 is selected from: unsubstituted C 1 -C 6 alkyl. -C 1 -C 6 hydroxyalkyl, -C 3 -C 8 cycloalkyl, -C 3 -C 8 heterocycloalkyl, –(C 1 -C 6 alkyl)-C 3 - C 8 heterocycloalkyl, unsubstituted -aryl, -hydroxyaryl, -aminoaryl, -heteroaryl and –(C 1 -C 6 alkyl)- aminoaryl.
- R 18 and R 19 taken together with the N atom to which they are bonded form a 4-, 5-, 6- or 7-membered ring having 0 to 3 substituents selected from: halogen, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, -C 3 -C 8 cycloalkyl and -(C 1 -C 6 alkyl)-O-R 5 .
- X a and X b are each independently selected from: NH and O.
- X a and X b are each independently selected from: NH and O.
- Combinations of any of the foregoing embodiments for compounds of Formula (IIa) are also contemplated and each combination forms a separate embodiment for the purposes of the present disclosure.
- the compound of Formula (I) has Formula (III):
- R 2 is selected from: -H, -CH 3 , -CF 3 , -F, -Br, -Cl, -OH, -OCH 3 and -OCF 3
- R 15 is selected from: -H, -CH 3 , -CHF 2 , -CF 3 , -F, -Br, -Cl, -OH, -OCH 3 and -OCF 3
- R 5 is selected from: -H, -C 1 -C 6 alkyl, -C 3 -C 8 cycloalkyl, -aryl, -heteroaryl and –(C 1 - C 6 alkyl)-aryl
- R 8 is selected from: -H, -C 1 -C 6 alkyl, -C 3 -C 8 cycloalkyl and -C 3 -C 8 heterocycloalkyl
- each R 9 is independently selected from: -H, -C 1 -C 6
- R 2 is selected from: -H, -CH 3 , - CF 3 , -F, -Cl, -OCH 3 and -OCF 3 .
- R 2 is selected from: -H, -F and - Cl.
- R 15 is selected from: -CH 3 , -CF 3 , -OCH 3 and -OCF 3 .
- R 15 is selected from: -CH 3 and - OCH 3 .
- R 2 is selected from: -H, -F and - Cl
- R 15 is selected from: -CH 3 , -CF 3 , -OCH 3 and -OCF 3 .
- R 2 is selected from: -H, -F and - Cl
- R 15 is selected from: -CH 3 and -OCH 3 .
- R 4 is selected from: , [00200]
- R 5 is selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, -C 3 -C 8 cycloalkyl, unsubstituted -aryl, -aminoaryl, -heteroaryl and –(C 1 -C 6 alkyl)-aminoaryl.
- R 8 is selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, -C 3 -C 8 cycloalkyl and -C 3 -C 8 heterocycloalkyl.
- each R 9 is independently selected from: -C 1 -C 6 alkyl, -C 3 -C 8 cycloalkyl, -aryl and –(C 1 -C 6 alkyl)-aryl.
- each R 9 is independently selected from: -C 1 -C 6 alkyl and –(C 1 -C 6 alkyl)-aryl.
- each R 9 is independently selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, - C 3 -C 8 cycloalkyl, unsubstituted -aryl, -aminoaryl, -heteroaryl and –(C 1 -C 6 alkyl)-aminoaryl.
- each R 10 is independently selected from: -C 1 -C 6 alkyl, -C 3 -C 8 cycloalkyl, -NR 14 R 14’ , -aryl and –(C 1 -C 6 alkyl)-aryl.
- each R 10 is independently selected from: -C 1 -C 6 alkyl, -NR 14 R 14’ , -aryl and –(C 1 -C 6 alkyl)-aryl.
- each R 10 is independently selected from: unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, -C 3 - C 8 cycloalkyl, -NR 14 R 14’ , unsubstituted -aryl, -aminoaryl, -heteroaryl and –(C 1 -C 6 alkyl)-aryl.
- each R 10’ is independently selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, - C 3 -C 8 cycloalkyl, unsubstituted -aryl, -aminoaryl, -heteroaryl and –(C 1 -C 6 alkyl)-aryl.
- R 11 is selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl and -C 1 -C 6 aminoalkyl.
- R 12 is selected from: -H, -C 1 -C 6 alkyl, -CO 2 R 8 , -aryl, –(C 1 -C 6 alkyl)-aryl and -S(O) 2 R 16 .
- R 12 is selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, -CO 2 R 8 , unsubstituted -aryl, -aminoaryl, -heteroaryl, –(C 1 -C 6 alkyl)-aminoaryl, -S(O) 2 R 16 and .
- R 13 is selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl and -C 1 -C 6 aminoalkyl.
- R 14 and R 14’ are each independently selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, -C 3 -C 8 cycloalkyl and -C 3 -C 8 heterocycloalkyl.
- R 16 is selected from: -aryl, - heteroaryl and –(C 1 -C 6 alkyl)-aryl.
- R 16 is selected from: unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, -C 3 -C 8 cycloalkyl, unsubstituted -aryl, -aminoaryl, -heteroaryl and –(C 1 -C 6 alkyl)-aminoaryl.
- R 18 and R 19 taken together with the N atom to which they are bonded form a 4-, 5-, 6- or 7-membered ring having 0 to 3 substituents selected from: halogen, unsubstituted C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, -C 3 -C 8 cycloalkyl and -(C 1 -C 6 alkyl)-O-R 5 .
- X a and X b are each independently selected from: NH and O.
- X a and X b are each independently selected from: NH and O.
- the compound of Formula (I) has Formula (IIIa) or (IIIb): wherein: R 4 , R 5 , R 8 , R 9 , R 10 , R 10’ , R 11 , R 12 , R 13 , R 14 , R 14’ , R 16 , R 18 , R 19 , X a , X b and X c are as defined in Formula (III).
- R 4 is selected [00221]
- R 5 is selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, - C 3 -C 8 cycloalkyl, unsubstituted -aryl, -aminoaryl, -heteroaryl and –(C 1 -C 6 alkyl)-aminoaryl.
- R 8 is selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, - C 3 -C 8 cycloalkyl and -C 3 -C 8 heterocycloalkyl.
- each R 9 is independently selected from: -C 1 -C 6 alkyl, -C 3 -C 8 cycloalkyl, -aryl and –(C 1 -C 6 alkyl)-aryl.
- each R 9 is independently selected from: -C 1 -C 6 alkyl and –(C 1 -C 6 alkyl)-aryl.
- each R 9 is independently selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, -C 3 -C 8 cycloalkyl, unsubstituted -aryl, -aminoaryl, -heteroaryl and –(C 1 -C 6 alkyl)-aminoaryl.
- each R 10 is independently selected from: -C 1 -C 6 alkyl, -C 3 -C 8 cycloalkyl, -NR 14 R 14’ , -aryl and –(C 1 -C 6 alkyl)- aryl.
- each R 10 is independently selected from: -C 1 -C 6 alkyl, -NR 14 R 14’ , -aryl and –(C 1 -C 6 alkyl)-aryl.
- each R 10 is independently selected from: unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, - C 1 -C 6 aminoalkyl, -C 3 -C 8 cycloalkyl, -NR 14 R 14’ , unsubstituted -aryl, -aminoaryl, -heteroaryl and –(C 1 -C 6 alkyl)-aryl.
- each R 10’ is independently selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, -C 3 -C 8 cycloalkyl, unsubstituted -aryl, -aminoaryl, -heteroaryl and –(C 1 -C 6 alkyl)-aryl.
- R 11 is selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl and -C 1 -C 6 aminoalkyl.
- R 12 is selected from: -H, -C 1 -C 6 alkyl, -CO 2 R 8 , -aryl, –(C 1 -C 6 alkyl)-aryl and -S(O) 2 R 16 .
- R 12 is selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, - CO 2 R 8 , unsubstituted -aryl, -aminoaryl, -heteroaryl, –(C 1 -C 6 alkyl)-aminoaryl, -S(O) 2 R 16 and .
- R 13 is selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl and -C 1 -C 6 aminoalkyl.
- R 14 and R 14’ are each independently selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, -C 3 -C 8 cycloalkyl and -C 3 -C 8 heterocycloalkyl.
- R 16 is selected from: -aryl, -heteroaryl and –(C 1 -C 6 alkyl)-aryl.
- R 16 is selected from: unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, -C 3 - C 8 cycloalkyl, unsubstituted -aryl, -aminoaryl, -heteroaryl and –(C 1 -C 6 alkyl)-aminoaryl.
- R 18 and R 19 taken together with the N atom to which they are bonded form a 4-, 5-, 6- or 7-membered ring having 0 to 3 substituents selected from: halogen, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, - C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, -C 3 -C 8 cycloalkyl and -(C 1 -C 6 alkyl)-O-R 5 .
- X a and X b are each independently selected from: NH and O.
- Combinations of any of the foregoing embodiments for each of compounds of Formula (IIIa) and Formula (IIIb) are also contemplated and each combination forms a separate embodiment for the purposes of the present disclosure.
- certain compounds of Formulae (I), (Ia), (II), (IIa), (III), (IIIa) or (IIIb) may include one or more free amino, hydroxy, carbonyl (for example, keto or aldehyde) or carboxylic acid groups.
- protecting group refers to a chemical group that, when attached to a potentially reactive functional group, masks, reduces or prevents the reactivity of the functional group. Typically, a protecting group can be selectively removed as desired during the course of a synthesis.
- Protecting groups are well-known in the art and various examples are described, for example, in “Protective Groups in Organic Chemistry” (Greene, W.
- amino protecting groups include, but are not limited to, formyl, acetyl, trifluoroacetyl, benzyl (Bn), benzoyl (Bz), benzyloxycarbonyl (CBZ), tert-butoxycarbonyl (Boc), trimethylsilyl (TMS), 2-trimethylsilyl-ethanesulfonyl (TES), trityl, substituted trityl, tosyl, phthalimide, alloxycarbonyl (Alloc) and 9-fluorenylmethyloxycarbonyl (FMOC).
- amino protecting groups include, but are not limited to, formyl, acetyl, trifluoroacetyl, benzyl (Bn), benzoyl (Bz), benzyloxycarbonyl (CBZ), tert-butoxycarbonyl (Boc), trimethylsilyl (TMS), 2-trimethylsilyl-ethanesulfonyl (TES
- hydroxy protecting groups include, but are not limited to, acetyl, benzyl (Bn), t-butyl, benzoyl (Bz), ⁇ -methoxyethoxymethyl ether (MEM), dimethoxytrityl (DMT), methoxymethyl ether (MOM), methoxytrityl [(4-methoxyphenyl)diphenylmethyl] (MMT), p-methoxybenzyl ether (PMB), p-methoxyphenyl ether (PMP), methylthiomethyl ether, pivaloyl (Piv), tetrahydropyranyl (THP), tetrahydrofuran (THF), trityl, trimethylsilyl (TMS), tert- butyldimethylsilyl (TBDMS or TBS), tri-iso-propylsilyloxymethyl (TOM), and triisopropylsilyl (TIPS).
- MEM ⁇ -
- carbonyl protecting groups include, but are not limited to, acetals, hemi- acetals and ketals.
- carboxylic acid protecting groups include, but are not limited to, methyl esters, benzyl esters, tert-butyl esters, silyl esters, orthoesters and oxazoline.
- Some embodiments relate to protected compounds of Formula (II) or (IIa) in which the free amino group at C10 is protected with a formyl, acetyl, trifluoroacetyl, benzyl (Bn), benzoyl (Bz), benzyloxycarbonyl (CBZ), tert- butoxycarbonyl (Boc), trityl, substituted trityl, tosyl, phthalimide, alloxycarbonyl (Alloc) or 9- fluorenylmethyloxycarbonyl (FMOC) group.
- Some embodiments relate to protected versions of compounds of Formula (II) or (IIa) in which the free amino group at C10 is protected with an acetyl group.
- each alkyl, cycloalkyl, heterocycloalkyl, aryl and heteroaryl group as defined in any one of Formulae (I), (Ia), (II), (IIa), (III), (IIIa) or (IIIb) is optionally substituted with one or more substituents selected from: halogen, acyl, acyloxy, alkoxy, carboxy, hydroxy, amino, amido, nitro, cyano, azido, alkylthio, thio, sulfonyl, sulfonamido, alkyl, cycloalkyl, heterocycloalkyl, aryl and heteroaryl.
- each alkyl, cycloalkyl, heterocycloalkyl, aryl and heteroaryl group as defined in any one of Formulae (I), (Ia), (II), (IIa), (III), (IIIa) or (IIIb) is optionally substituted with one or more substituents selected from: halogen, acyl, acyloxy, alkoxy, carboxy, hydroxy, amino, amido, nitro, cyano, azido, alkylthio, thio, sulfonyl and sulfonamido.
- the camptothecin analogue is a compound having Formula (I) or a protected version thereof and is selected from the compounds shown in Table 1.
- Table 1 Exemplary Camptothecin Analogues of Formula (I)
- the camptothecin analogue is a compound having Formula (II) or a protected version thereof and is selected from the compounds shown in Table 2.
- the camptothecin analogue is a compound having Formula (III) or a protected version thereof and is selected from the compounds shown in Table 3.
- Table 3 Exemplary Camptothecin Analogues of Formula (III)
- compounds of Formula (I) throughout the remainder of this disclosure, includes in various embodiments, compounds of Formula (Ia), Formula (II), Formula (IIa), Formula (III), Formula (IIIa) and Formula (IIIb), to the same extent as if embodiments reciting each of these Formulae individually were specifically recited.
- compounds of Formula (I) may possess a sufficiently acidic group, a sufficiently basic group, or both functional groups, and accordingly react with a number of organic and inorganic bases, or organic and inorganic acids, to form pharmaceutically acceptable salts.
- pharmaceutically acceptable salt refers to a salt of a compound of Formula (I), which is substantially non-toxic to living organisms.
- Typical pharmaceutically acceptable salts include those salts prepared by reaction of a compound of Formula (I) with a pharmaceutically acceptable mineral or organic acid or an organic or inorganic base. Such salts are known as acid addition and base addition salts.
- Acids commonly employed to form acid addition salts are inorganic acids including, but are not limited to, hydrochloric acid, hydrobromic acid, hydroiodic acid, sulfuric acid, phosphoric acid, and organic acids including, but not limited to, p-toluenesulfonic acid, methanesulfonic acid, oxalic acid, p-bromophenylsulfonic acid, carbonic acid, succinic acid, citric acid, benzoic acid and acetic acid.
- inorganic acids including, but are not limited to, hydrochloric acid, hydrobromic acid, hydroiodic acid, sulfuric acid, phosphoric acid
- organic acids including, but not limited to, p-toluenesulfonic acid, methanesulfonic acid, oxalic acid, p-bromophenylsulfonic acid, carbonic acid, succinic acid, citric acid, benzoic acid and acetic acid.
- Examples of pharmaceutically acceptable salts include, but are not limited to, sulfates, pyrosulfates, bisulfates, sulfites, phosphates, monohydrogenphosphates, dihydrogenphosphates, metaphosphates, pyrophosphates, bromides, iodides, acetates, propionates, decanoates, caprylates, acrylates, formates, hydrochlorides, dihydrochlorides, isobutyrates, caproates, heptanoates, propiolates, oxalates, malonates, succinates, suberates, sebacates, fumarates, maleates, butyne-1,4- dioates, hexyne-1,6-dioates, benzoates, chlorobenzoates, methylbenzoates, hydroxybenzoates, methoxybenzoates, phthalates, xylenesulfonates, phenylacetates, phenylpropionate
- Salts of amine groups may also comprise quaternary ammonium salts in which the amino nitrogen carries a suitable organic group such as an alkyl, lower alkenyl, lower alkynyl or aralkyl moiety.
- Base addition salts include those derived from inorganic bases, such as ammonium or alkali or alkaline earth metal hydroxides, carbonates, bicarbonates, and the like.
- Bases useful in preparing pharmaceutically acceptable salts include, but are not limited to, sodium hydroxide, potassium hydroxide, ammonium hydroxide, potassium carbonate, sodium carbonate, sodium bicarbonate, potassium bicarbonate, calcium hydroxide and calcium carbonate.
- the particular counterion forming a part of a pharmaceutically acceptable salt is usually not of a critical nature, so long as the salt as a whole is pharmacologically acceptable and as long as the counterion does not contribute undesired qualities to the salt as a whole.
- Certain embodiments relate to pharmaceutically acceptable solvates of a compound of Formula (I).
- solvents such as water, methanol, ethanol or acetonitrile to form pharmaceutically acceptable solvates such as the corresponding hydrate, methanolate, ethanolate or acetonitrilate.
- solvents such as water, methanol, ethanol or acetonitrile
- Other examples of solvents that may be used to prepare solvates include isopropanol, dimethyl sulfoxide, ethyl acetate, acetic acid, ethanolamine and acetone, as well as miscible formulations of solvate mixtures as would be known by the skilled artisan.
- Camptothecin analogues of Formula (I) may be prepared by standard synthetic organic chemistry methods from commercially available starting materials and reagents. See, also, Li, et al., 2019, ACS Med. Chem. Lett., 10(10): 1386 ⁇ 1392 and U.S. Patent Application Publication No. US 2004/0266803. Representative examples of suitable synthetic routes are described in detail in the Examples provided herein (see also Figure 1). One skilled in the art will recognize that alternative methods may be employed to synthesize camptothecin analogues of Formula (I), and that the approaches described herein are therefore not intended to be exhaustive.
- conjugates of compounds of Formula (I) comprising one or more compounds of Formula (I) conjugated to a targeting moiety via one or more linkers.
- the conjugates of the present disclosure may comprise one or multiple compounds of Formula (I) conjugated to the targeting moiety.
- multiple compounds of Formula (I) may be conjugated to the targeting moiety by attaching the compound at multiple different sites on the targeting moiety.
- multiple compounds of Formula (I) may be conjugated to the targeting moiety by employing one or more multivalent linkers each allowing for attachment of multiple compounds to a single site on the targeting moiety.
- certain embodiments of the present disclosure relate to conjugates of Formula (X): T-[L-(D) m ] n (X) wherein: T is a targeting moiety; L is a linker; D is a camptothecin analogue as described herein; m is an integer between 1 and 4, and n is an integer between 1 and 10.
- T is a targeting moiety
- L is a linker
- D is a camptothecin analogue as described herein
- m is an integer between 1 and 4
- n is an integer between 1 and 10.
- m is between 1 and 2.
- m is 1.
- n is between 1 and 8, for example, between 2 and 8, or between 2 and 6. In some embodiments, n is between 2 and 4.
- a targeting moiety, “T,” can be conjugated to more than one compound of Formula (I), “D.”
- D a compound of Formula (I)
- any particular targeting moiety T is conjugated to an integer number of compounds D
- analysis of a preparation of the conjugate to determine the ratio of compound D to targeting moiety T may give a non-integer result, reflecting a statistical average.
- This ratio of compound D to targeting moiety T may generally be referred to as the drug-to-antibody ratio, or “DAR.” Accordingly, conjugate preparations having non-integer DARs are intended to be encompassed by Formula (X).
- conjugates of Formula (X) comprise a camptothecin analogue as the drug moiety, D, where the camptothecin analogue is a compound of Formula (I).
- D is a compound of Formula (Ia), Formula (II), Formula (IIa), Formula (III), Formula (IIIa) or Formula (IIIb).
- D is a compound selected from the compounds shown in Tables 1-3.
- D is a compound of Formula (IV): wherein: R 1a is selected from: -H, -CH 3 , -CHF2, -CF 3 , -F, -Br, -Cl, -OH, -OCH 3 , -OCF 3 and - NH 2 ; R 2a is selected from: -H, -CH 3 , -CF 3 , -F, -Br, -Cl, -OH, -OCH 3 and -OCF 3 ; X is -O-, -S- or -NH-, and R 4a is selected from: , wherein * is the point of attachment to X, and wherein p is 1, 2, 3 or 4; or X is O, and R 4a -X- is selected from: R 5a is selected from: -C 1 -C 6 alkyl, –C 3 -C 8
- R 1a is selected from: -CH 3 , -CF 3 , -OCH 3 , -OCF 3 and -NH 2 .
- R 1a is selected from: -CH 3 , -CF 3 , -OCH 3 and -OCF 3 .
- R 1a is selected from: -CH 3 , -OCH 3 and NH 2 .
- R 1a is selected from: -CH 3 and - OCH 3 .
- R 2a is selected from: -H, -CH 3 , - CF 3 , -F, -Cl, -OCH 3 and -OCF 3 .
- R 2a is selected from: -H, -F and - Cl.
- R 2a is -F.
- each R 9a is independently selected from: -C 1 -C 6 alkyl, -C 3 -C 8 cycloalkyl, -aryl and –(C 1 -C 6 alkyl)-aryl.
- each R 9a is independently selected from: -C 1 -C 6 alkyl and –(C 1 -C 6 alkyl)-aryl.
- each R 10a is independently selected from: -C 1 -C 6 alkyl, -C 3 -C 8 cycloalkyl, -aryl, –(C 1 -C 6 alkyl)-aryl and [00276]
- each R 10a is independently selected from: -C 1 -C 6 alkyl, -aryl, –(C 1 -C 6 alkyl)-aryl and [00277]
- R 12a is selected from: -C 1 -C 6 alkyl, -aryl, –(C 1 -C 6 alkyl)-aryl and -S(O) 2 R
- R 13a is selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl and -C 1 -C 6 aminoalkyl.
- R 14a’ is selected from: H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, –C 1 -C 6 hydroxyalkyl, –C 1 -C 6 aminoalkyl, -C 3 -C 8 cycloalkyl and -C 3 -C 8 heterocycloalkyl.
- R 16a is selected from: -aryl, - heteroaryl and –(C 1 -C 6 alkyl)-aryl.
- R 22 and R 23 are each independently selected from: -H, -halogen, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 aminoalkyl, -C 1 -C 6 hydroxyalkyl and -C 3 -C 8 cycloalkyl.
- X a and X b are each independently selected from: NH and O.
- R 2a is selected from: -CH 3 , -CF 3 , - F, -Cl, -OCH 3 and -OCF 3 .
- R 2a is selected from: -CF 3 , -F, -Cl and -OCH 3 .
- R 2a is F.
- R 20a is selected from: -H, -C 1 -C 6 [00289] In some embodiments, in compounds of Formula (V), R 20a is selected from: -H, -C 1 -C 6 alkyl, -(C 1 -C 6 alkyl)-O-R 5 , –(C 1 -C 6 alkyl)-aryl, , .
- R 20a is selected from: -H, -C 1 -C 6 alkyl, -(C 1 -C 6 alkyl)-O-R 5 , , –(C 1 -C 6 alkyl)-aryl, [00291] In some embodiments, in compounds of Formula (V), R 20a is selected from: -H, -C 1 -C 6 [00292] In some embodiments, in compounds of Formula (V), R 20a is selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 3 -C 8 cycloalkyl, -(C 1 -C 6 alkyl)-O-R 5 , -CO 2 R 8 , unsubstituted -aryl, -aminoaryl, -heter
- R 6 and R 7 are each independently selected from: -H, -C 1 -C 6 alkyl, -C 3 -C 8 cycloalkyl and -C(O)R 17 .
- R 6 is H
- R 7 is selected from: - H, -C 1 -C 6 alkyl, -C 3 -C 8 cycloalkyl, -(C 1 -C 6 alkyl)-O-R 5 , -C 3 -C 8 heterocycloalkyl and -C(O)R 17 .
- R 6 is H
- R 7 is selected from: - H, -C 1 -C 6 alkyl, -C 3 -C 8 cycloalkyl and -C(O)R 17 .
- R 6 and R 7 are each independently selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 3 -C 8 cycloalkyl, -(C 1 -C 6 alkyl)-O-R 5 , -C 3 -C 8 heterocycloalkyl and -C(O)R 17 .
- R 8 is selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, -C 3 -C 8 cycloalkyl and -C 3 -C 8 heterocycloalkyl.
- each R 9 is independently selected from: -C 1 -C 6 alkyl, -C 3 -C 8 cycloalkyl, -aryl and –(C 1 -C 6 alkyl)-aryl.
- each R 9 is independently selected from: -C 1 -C 6 alkyl and –(C 1 -C 6 alkyl)-aryl.
- each R 9 is independently selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, - C 3 -C 8 cycloalkyl, unsubstituted -aryl, -aminoaryl, -heteroaryl and –(C 1 -C 6 alkyl)-aminoaryl.
- each R 10 is independently selected from: -C 1 -C 6 alkyl, -C 3 -C 8 cycloalkyl, -NR 14 R 14’ , -aryl and –(C 1 -C 6 alkyl)-aryl.
- each R 10 is independently selected from: -C 1 -C 6 alkyl, -NR 14 R 14’ , -aryl and –(C 1 -C 6 alkyl)-aryl.
- R 11 is selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl and -C 1 -C 6 aminoalkyl.
- R 12 is selected from: -H, -C 1 -C 6 alkyl, -aryl, –(C 1 -C 6 alkyl)-aryl and -S(O) 2 R 16 .
- R 12 is selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, -CO 2 R 8 , unsubstituted -aryl, -aminoaryl, -heteroaryl, –(C 1 -C 6 alkyl)-aminoaryl, -S(O) 2 R 16 and .
- R 13 is selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl and -C 1 -C 6 aminoalkyl.
- R 14 and R 14’ are each independently selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, -C 3 -C 8 cycloalkyl and -C 3 -C 8 heterocycloalkyl.
- R 16 is selected from: -aryl, - heteroaryl and –(C 1 -C 6 alkyl)-aryl.
- R 16 is selected from: unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl, -C 3 -C 8 cycloalkyl, unsubstituted -aryl, -aminoaryl, -heteroaryl and –(C 1 -C 6 alkyl)-aminoaryl.
- R 17 is selected from: unsubstituted -C 1 -C 6 alkyl, -C 3 -C 8 cycloalkyl, -C 3 -C 8 heterocycloalkyl, —(C 1 -C 6 alkyl)-C 3 -C 8 heterocycloalkyl, unsubstituted -aryl, -hydroxyaryl, -aminoaryl, -heteroaryl and –(C 1 -C 6 alkyl)-aminoaryl.
- R 18 and R 19 taken together with the N atom to which they are bonded form a 4-, 5-, 6-, or 7-membered ring having 0 to 3 substituents selected from: halogen, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 aminoalkyl, -C 1 -C 6 hydroxyalkyl, -C 3 -C 8 cycloalkyl and -(C 1 -C 6 alkyl)-O-R 5 .
- R 17 is -C 1 -C 6 alkyl.
- X a and X b are each independently selected from: NH and O.
- D is a compound of Formula (VI): wherein: R 2a is selected from: -H, -CH 3 , -CF 3 , -F, -Br, -Cl, -OH, -OCH 3 and -OCF 3 ; , wherein * is the point of attachment to X, and wherein p is 1, 2, 3 or 4; or X is O, and R 25 -X- is selected from: R 5a is selected from: -C 1 -C 6 alkyl, –C 3 -C 8 cycloalkyl, –aryl, -heteroaryl and –(C 1 -C 6 alkyl)-aryl; R 6a is selected from: -H, -C 1 -C 6 alkyl, -C 3 -C 8 cycloalkyl and -C 3 -C 8 heterocycloalkyl; R 7a is
- R 2a is selected from: -CH 3 , -CF 3 , -F, -Br, -Cl, -OH, -OCH 3 and -OCF 3 .
- R 2a is selected from: -CH 3 , -CF 3 , -F, -Cl, -OCH 3 and -OCF 3 .
- R 2a is selected from: F and Cl.
- R 2a is F.
- X is -O-, -S- or -NH-
- R 25 is selected from: -C 1 -C 6 alkyl, -(C 1 -C 6 alkyl)-O-R 5a , –(C 1 -C 6 alkyl)-aryl,
- X is -O-, -S- or -NH-
- R 25 is selected from: -C 1 -C 6 alkyl, -(C 1 -C 6 alkyl)-O-R 5a , –(C 1 -C 6 alkyl)-aryl,
- X is -O-, -S- or -NH-
- R 25 is selected from: -C 1 -C 6 alkyl, -(C 1 -C 6 alkyl)-(C 1 -C 6 alkyl)--aryl
- R 6a is H.
- R 6a is selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 3 -C 8 cycloalkyl and -C 3 -C 8 heterocycloalkyl.
- R 7a is selected from: -C 1 -C 6 alkyl, -C 3 -C 8 cycloalkyl and -C(O)R 17a .
- each R 9a is independently selected from: -C 1 -C 6 alkyl, -C 3 -C 8 cycloalkyl, -aryl and –(C 1 -C 6 alkyl)-aryl.
- each R 9a is independently selected from: -C 1 -C 6 alkyl and –(C 1 -C 6 alkyl)-aryl.
- each R 10a is independently selected from: -C 1 -C 6 alkyl, -C 3 -C 8 cycloalkyl, -aryl, –(C 1 -C 6 alkyl)-aryl and [00331] In some embodiments, in compounds of Formula (VI), each R 10a is independently selected from: -C 1 -C 6 alkyl, -aryl,–(C 1 -C 6 alkyl)-aryl and [00332] In some embodiments, in compounds of Formula (VI), R 12a is selected from: -C 1 -C 6 alkyl, -aryl, –(C 1 -C 6 alkyl)-aryl and -S(O) 2 R 16a .
- R 13a is selected from: -H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl and -C 1 -C 6 aminoalkyl.
- R 14a’ is selected from: H, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, –C 1 -C 6 hydroxyalkyl, –C 1 -C 6 aminoalkyl, -C 3 -C 8 cycloalkyl and -C 3 -C 8 heterocycloalkyl.
- R 16a is selected from: -aryl, - heteroaryl and –(C 1 -C 6 alkyl)-aryl.
- R 17a is -C 1 -C 6 alkyl.
- R 22 and R 23 are each independently selected from: -H, -halogen, unsubstituted -C 1 -C 6 alkyl, -C 1 -C 6 haloalkyl, -C 1 -C 6 hydroxyalkyl, -C 1 -C 6 aminoalkyl and -C 3 -C 8 cycloalkyl.
- X a and X b are each independently selected from: NH and O.
- each alkyl, cycloalkyl, heterocycloalkyl, aryl and heteroaryl group as defined in any one of Formulae (IV), (V) or (VI) is optionally substituted with one or more substituents selected from: halogen, acyl, acyloxy, alkoxy, carboxy, hydroxy, amino, amido, nitro, cyano, azido, alkylthio, thio, sulfonyl, sulfonamido, alkyl, cycloalkyl, heterocycloalkyl, aryl and heteroaryl.
- each alkyl, cycloalkyl, heterocycloalkyl, aryl and heteroaryl group as defined in any one of Formulae (IV), (V) or (VI) is optionally substituted with one or more substituents selected from: halogen, acyl, acyloxy, alkoxy, carboxy, hydroxy, amino, amido, nitro, cyano, azido, alkylthio, thio, sulfonyl and sulfonamido.
- the targeting moiety, T comprised by the conjugates of Formula (X) is a molecule that binds, reactively associates or complexes with a receptor, antigen or other receptive moiety associated with a given target cell population.
- the targeting moiety, T functions to deliver the camptothecin analogue, D, to the particular target cell population with which the targeting moiety, T, reacts.
- targeting moieties include, but are not limited to, proteins (such as antibodies, antibody fragments and growth factors), glycoproteins, peptides (such as bombesin and gastrin-releasing peptide), lectins, vitamins (such as folic acid) and nutrient- transport molecules (such as transferrin).
- the targeting moiety, T will be bonded to linker, L, via a heteroatom of targeting moiety, T, such as a sulfur (for example, from a sulfhydryl group), oxygen (for example, from a carbonyl, carboxyl or hydroxyl group) or nitrogen (for example, from a primary or secondary amino group).
- targeting moiety, T is an antibody.
- ADCs antibody-drug conjugates having general Formula (X) in which the targeting moiety, T, is an antibody.
- the antibody included as the targeting moiety, T may be a full-size polyclonal or monoclonal antibody, an antigen-binding antibody fragment (such as Fab, scFab, Fab', F(ab') 2 , Fv or scFv), a domain antibody (dAb) or an antibody mimetic (such as an affibody, a DARPin, an anticalin, a versabody, a duocalin, a lipocalin or an avimer).
- the antibody is typically directed to a particular antigen, for example, a disease-associated antigen such as a tumor-associated antigen, an antigen associated with an autoimmune disease or a viral antigen.
- the targeting moiety, T is a monoclonal antibody, an antigen-binding antibody fragment (such as Fab, scFab, Fab', F(ab') 2 , Fv or scFv) or a domain antibody (dAb).
- an antigen-binding antibody fragment such as Fab, scFab, Fab', F(ab') 2 , Fv or scFv
- dAb domain antibody
- monoclonal antibodies may be produced by methods including, but not limited to, the hybridoma technique originally described by Kohler and Milstein (1975, Nature 256:495- 497), the human B cell hybridoma technique (Kozbor et al., 1983, Immunology Today 4:72), the EBV-hybridoma technique (Cole et al., 1985, Monoclonal Antibodies and Cancer Therapy, Alan R. Liss, Inc., pp.
- targeting moiety is an antibody of the IgG class.
- targeting moiety, T may be a monoclonal antibody.
- the monoclonal antibody may be, for example, a non-human monoclonal antibody (such as a mouse antibody), a human monoclonal antibody, a humanized monoclonal antibody or a chimeric antibody (for example, a human-mouse antibody).
- Human monoclonal antibodies may be made by any of numerous techniques known in the art (see, for example, Teng et al., 1983, Proc. Natl. Acad. Sci. USA 80:7308-7312; Kozbor et al., 1983, Immunology Today 4:72-79; Olsson et al., 1983, Meth. Enzymol. 92:3-16; Huse et al., 1989, Science 246:1275-1281, and U.S.
- Antibodies immunospecific for a given target antigen may also be obtained commercially.
- the antibody included in the conjugate may be a bispecific or multispecific antibody.
- targeting moiety, T comprised by the conjugate is an antibody or antigen-binding antibody fragment that binds to a tumor-associated antigen (TAA).
- TAA tumor-associated antigen
- tumor-associated antigens include, but are not limited to, 5T4, ADAM-9, ALK, AMHRII, ASCT2, Axl, B7-H 3 , BCMA, C4.4a, CA6, CA9, CanAg, CD123, CD138, CD142, CD166, CD184, CD19, CD20, CD205, CD22, CD248, CD25, CD3, CD30, CD33, CD352, CD37, CD38, CD40L, CD44v6, CD45, CD46, CD48, CD51, CD56, CD7, CD70, CD71, CD74, CD79b, CDH6, CEACAM5, CEACAM6, cKIT, CLDN18.2, CLDN6, CLL-1, c-MET, Cripto, CSP-1, CXCR5, DLK-1, DLL3, DPEP3, Dysadherin, EFNA4 , EGFR, EGFRviii, ENPP3, EpCAM, EphA2, EphA3, ETBR, FGFR2, FGFR3, FL
- the conjugates of Formula (X) include a linker, L, which is a bifunctional or multifunctional moiety capable of linking one or more camptothecin analogues, D, to targeting moiety, T.
- a bifunctional (or monovalent) linker, L links a single compound D to a single site on targeting moiety, T, whereas a multifunctional (or polyvalent) linker, L, links more than one compound, D, to a single site on targeting moiety, T.
- a linker that links one compound, D, to more than one site on targeting moiety, T may also be considered to be multifunctional in certain embodiments.
- Linker, L includes a functional group capable of reacting with the target group or groups on targeting moiety, T, and at least one functional group capable of reacting with a target group on the camptothecin analogue, D.
- Suitable functional groups are known in the art and include those described, for example, in Bioconjugate Techniques (G.T. Hermanson, 2013, Academic Press).
- Groups on targeting moiety, T, and the camptothecin analogue, D, that may serve as target groups for linker attachment include, but are not limited to, thiol, hydroxyl, carboxyl, amine, aldehyde and ketone groups.
- Non-limiting examples of functional groups capable of reacting with thiols include maleimide, haloacetamide, haloacetyl, activated esters (such as succinimide esters, 4-nitrophenyl esters, pentafluorophenyl esters and tetrafluorophenyl esters), anhydrides, acid chlorides, sulfonyl chlorides, isocyanates and isothiocyanates.
- activated esters such as succinimide esters, 4-nitrophenyl esters, pentafluorophenyl esters and tetrafluorophenyl esters
- anhydrides acid chlorides, sulfonyl chlorides, isocyanates and isothiocyanates.
- self-stabilizing maleimides as described in Lyon et al., 2014, Nat. Biotechnol., 32:1059-1062.
- Non-limiting examples of functional groups capable of reacting with amines include activated esters (such as N-hydroxysuccinamide (NHS) esters, sulfo-NHS esters, imido esters such as Traut’s reagent, tetrafluorophenyl (TFP) esters and sulfodichlorophenyl esters), isothiocyanates, aldehydes and acid anhydrides (such as diethylenetriaminepentaacetic anhydride (DTPA)).
- activated esters such as N-hydroxysuccinamide (NHS) esters, sulfo-NHS esters, imido esters such as Traut’s reagent, tetrafluorophenyl (TFP) esters and sulfodichlorophenyl esters
- isothiocyanates such as N-hydroxysuccinamide (NHS) esters, sulfo-NHS esters, imido esters such
- Non-limiting examples of functional groups capable of reacting with an electrophilic group such as an aldehyde or ketone carbonyl group include hydrazide, oxime, amino, hydrazine, thiosemicarbazone, hydrazine carboxylate and arylhydrazide.
- linker, L may include a functional group that allows for bridging of two interchain cysteines on the antibody, such as a ThioBridge TM linker (Badescu et al., 2014, Bioconjug. Chem.25:1124–1136), a dithiomaleimide (DTM) linker (Behrens et al., 2015, Mol. Pharm. 12:3986–3998), a dithioaryl(TCEP)pyridazinedione-based linker (Lee et al., 2016, Chem.
- a functional group that allows for bridging of two interchain cysteines on the antibody such as a ThioBridge TM linker (Badescu et al., 2014, Bioconjug. Chem.25:1124–1136), a dithiomaleimide (DTM) linker (Behrens et al., 2015, Mol. Pharm. 12:3986–3998), a dithioaryl(
- targeting moiety, T may be modified to include a non-natural reactive group, such as an azide, that allows for conjugation to the linker via a complementary reactive group on the linker.
- conjugation of the linker to the targeting moiety may make use of click chemistry reactions (see, for example, Chio & Bane, 2020, Methods Mol.
- AAC azide-alkyne cycloaddition
- the AAC reaction may be a copper-catalyzed AAC (CuAAC) reaction, which involves coupling of an azide with a linear alkyne, or a strain-promoted AAC (SPAAC) reaction, which involves coupling of an azide with a cyclooctyne.
- CuAAC copper-catalyzed AAC
- SPAAC strain-promoted AAC
- Linker, L may be a cleavable or a non-cleavable linker.
- a cleavable linker is a linker that is susceptible to cleavage under specific conditions, for example, intracellular conditions (such as in an endosome or lysosome) or within the vicinity of a target cell (such as in the tumor microenvironment).
- Examples include linkers that are protease-sensitive, acid-sensitive or reduction-sensitive.
- Non-cleavable linkers by contrast, rely on the degradation of the antibody in the cell, which typically results in the release of an amino acid-linker-drug moiety.
- Examples of cleavable linkers include, for example, linkers comprising an amino acid sequence that is a cleavage recognition sequence for a protease. Many such cleavage recognition sequences are known in the art.
- conjugates that are not intended to be internalized by a cell for example, an amino acid sequence that is recognized and cleaved by a protease present in the extracellular matrix in the vicinity of a target cell, such as a cancer cell, may be employed.
- extracellular tumor-associated proteases include, for example, plasmin, matrix metalloproteases (MMPs), elastase and kallikrein-related peptidases.
- linker, L may comprise an amino acid sequence that is recognized and cleaved by an endosomal or lysosomal protease.
- Cleavage recognition sequences may be, for example, dipeptides, tripeptides or tetrapeptides.
- Non-limiting examples of dipeptide recognition sequences that may be included in cleavable linkers include, but are not limited to, Ala-(D)Asp, Ala-Lys, Ala-Phe, Asn-Lys, Asn- (D)Lys, Asp-Val, His-Val, Ile-Cit, Ile-Pro, Ile-Val, Leu-Cit, Me3Lys-Pro, Met-Lys, Met-(D)Lys, NorVal-(D)Asp, Phe-Arg, Phe-Cit, Phe-Lys, PhenylGly-(D)Lys, Pro-(D)Lys, Trp-Cit, Val-Ala, Val-(D)Asp, Val-Cit, Val-Gly, Val-Gln and Val-Lys.
- tri- and tetrapeptide cleavage sequences include, but are not limited to, Ala-Ala-Asn, Ala-Val-Cit, (D)Ala-Phe-Lys, Asp-Val- Ala, Asp-Val-Cit, Gly-Cit-Val, Lys-Val-Ala, Lys-Val-Cit, Met-Cit-Val, (D)Phe-Phe-Lys, Asn- Pro-Val, Ala-Leu-Ala-Leu, Gly-Phe-Leu-Gly, Gly-Gly-Phe-Gly and Gly-Phe-Gly-Gly.
- cleavable linkers include disulfide-containing linkers such as N- succinimydyl-4-(2-pyridyldithio) butanoate (SPDB) and N-succinimydyl-4-(2-pyridyldithio)-2- sulfo butanoate (sulfo-SPDB).
- Disulfide-containing linkers may optionally include additional groups to provide steric hindrance adjacent to the disulfide bond in order to improve the extracellular stability of the linker, for example, inclusion of a geminal dimethyl group.
- cleavable linkers include linkers hydrolyzable at a specific pH or within a pH range, such as hydrazone linkers. Linkers comprising combinations of these functionalities may also be useful, for example, linkers comprising both a hydrazone and a disulfide are known in the art.
- a further example of a cleavable linker is a linker comprising a ⁇ -glucuronide, which is cleavable by ⁇ -glucuronidase, an enzyme present in lysosomes and tumor interstitium (see, for example, De Graaf et al., 2002, Curr. Pharm. Des. 8:1391–1403, and International Patent Publication No.
- linker, L may also function to improve the hydrophilicity of linker, L.
- linker Another example of a linker that is cleaved internally within a cell and improves hydrophilicity is a linker comprising a pyrophosphate diester moiety (see, for example, Kern et al., 2016, J Am Chem Soc., 138:2430-1445).
- the linker, L, comprised by the conjugate of Formula (X) is a cleavable linker.
- linker, L comprises a cleavage recognition sequence.
- linker may comprise an amino acid sequence that is recognized and cleaved by a lysosomal protease.
- Cleavable linkers may optionally further comprise one or more additional functionalities such as self-immolative and self-elimination groups, stretchers or hydrophilic moieties.
- Self-immolative and self-elimination groups that find use in linkers include, for example, p-aminobenzyl (PAB) and p-aminobenzyloxycarbonyl (PABC) groups, and methylated ethylene diamine (MED).
- PAB p-aminobenzyl
- PABC p-aminobenzyloxycarbonyl
- MED methylated ethylene diamine
- self-immolative groups include, but are not limited to, aromatic compounds that are electronically similar to the PAB or PABC group such as heterocyclic derivatives, for example 2-aminoimidazol-5-methanol derivatives as described in U.S. Patent No. 7,375,078.
- Other examples include groups that undergo cyclization upon amide bond hydrolysis, such as substituted and unsubstituted 4-aminobutyric acid amides (Rodrigues et al., 1995, Chemistry Biology 2:223-227) and 2-aminophenylpropionic acid amides (Amsberry, et al., 1990, J. Org. Chem. 55:5867-5877).
- Self-immolative/self-elimination groups are typically attached to an amino or hydroxyl group on the compound, D.
- Self-immolative/self-elimination groups alone or in combination are often included in peptide-based linkers, but may also be included in other types of linkers.
- Stretchers that find use in linkers for drug conjugates include, for example, alkylene groups and stretchers based on aliphatic acids, diacids, amines or diamines, such as diglycolate, malonate, caproate and caproamide.
- Other stretchers include, for example, glycine-based stretchers and polyethylene glycol (PEG) or monomethoxy polyethylene glycol (mPEG) stretchers.
- PEG and mPEG stretchers can also function as hydrophilic moieties within a linker.
- PEG or mPEG may be included in a linker either “in-line” or as pendant groups to increase the hydrophilicity of the linker (see, for example, U.S. Patent Application Publication No. US 2016/0310612).
- Various PEG-containing linkers are commercially available from companies such as Quanta BioDesign, Ltd (Plain City, OH).
- Other hydrophilic groups that may optionally be incorporated into linker, L include, for example, ⁇ -glucuronide, sulfonate groups, carboxylate groups and pyrophosphate diesters.
- conjugates of Formula (X) may comprise a cleavable linker. In some embodiments, conjugates of Formula (X) may comprise a peptide-containing linker. In some embodiments, conjugates of Formula (X) may comprise a protease-cleavable linker.
- linker, L is a cleavable linker having Formula (XI): wherein: Z is a linking group that joins the linker to a target group on targeting moiety, T; Str is a stretcher; AA 1 and AA 2 are each independently an amino acid, wherein AA 1 -[AA 2 ] r forms a protease cleavage site; X is a self-immolative group; q is 0 or 1; r is 1, 2 or 3; s is 0, 1 or 2; # is the point of attachment to targeting moiety, T, and % is the point of attachment to the camptothecin analogue, D.
- Z is a linking group that joins the linker to a target group on targeting moiety, T
- Str is a stretcher
- AA 1 and AA 2 are each independently an amino acid, wherein AA 1 -[AA 2 ] r forms a protease cleavage site
- X is a self-immolative group
- in linkers of Formula (XI) q is 1.
- in linkers of Formula (XI) s is 1.
- in linkers of Formula (XI) s is 0.
- Str is selected from: wherein: R is H or C 1 -C 6 alkyl; t is an integer between 2 and 10, and u is an integer between 1 and 10.
- AA1-[AA2]r has a sequence selected from: Ala-(D)Asp, Ala-Lys, Ala-Phe, Asn-Lys, Asn-(D)Lys, Asp-Val, His-Val, Ile-Cit, Ile-Pro, Ile-Val, Leu-Cit, Me3Lys-Pro, Met-Lys, Met-(D)Lys, NorVal-(D)Asp, Phe-Arg, Phe-Cit, Phe-Lys, PhenylGly-(D)Lys, Pro-(D)Lys, Trp-Cit, Val-Ala, Val-(D)
- tri- and tetrapeptide cleavage sequences include, but are not limited to, Ala- Ala-Asn, Ala-Val-Cit, (D)Ala-Phe-Lys, Asp-Val-Ala, Asp-Val-Cit, Gly-Cit-Val, Lys-Val-Ala, Lys-Val-Cit, Met-Cit-Val, (D)Phe-Phe-Lys, Asn-Pro-Val, Ala-Leu-Ala-Leu, Gly-Phe-Leu-Gly, Gly-Gly-Phe-Gly and Gly-Phe-Gly-Gly.
- linker, L in conjugates of Formula (X), is 1, and linker, L, has Formula (XI).
- linker, L is a cleavable linker having Formula (XII): wherein: Z is a linking group that joins the linker to a target group on targeting moiety, T; Str is a stretcher; AA1 and AA2 are each independently an amino acid, wherein AA1-[AA2]r forms a protease cleavage site; Y is -NH-CH 2 - or -NH-CH 2 -C(O)-; q is 0 or 1; r is 1, 2 or 3; v is 0 or 1; # is the point of attachment to targeting moiety, T, and % is the point of attachment to the camptothecin analogue, D.
- linkers of Formula (XII) q is 1.
- s is 0.
- in linkers of Formula (XII) s is 1.
- Z is where # is the point of attachment to T, and * is the point of attachment to the remainder of the linker.
- Str is selected from: wherein: R is H or C 1 -C 6 alkyl; t is an integer between 2 and 10, and u is an integer between 1 and 10.
- AA1-[AA2]r has a sequence selected from: Ala-(D)Asp, Ala-Lys, Ala-Phe, Asn-Lys, Asn-(D)Lys, Asp-Val, His-Val, Ile-Cit, Ile-Pro, Ile-Val, Leu-Cit, Me 3 Lys-Pro, Met-Lys, Met-(D)Lys, NorVal-(D)Asp, Phe-Arg, Phe-Cit, Phe-Lys, PhenylGly-(D)Lys, Pro-(D)Lys, Trp-Cit, Val-Ala, Val-(
- tri- and tetrapeptide cleavage sequences include, but are not limited to, Ala- Ala-Asn, Ala-Val-Cit, (D)Ala-Phe-Lys, Asp-Val-Ala, Asp-Val-Cit, Gly-Cit-Val, Lys-Val-Ala, Lys-Val-Cit, Met-Cit-Val, (D)Phe-Phe-Lys, Asn-Pro-Val, Ala-Leu-Ala-Leu, Gly-Phe-Leu-Gly, Gly-Gly-Phe-Gly and Gly-Phe-Gly-Gly.
- conjugates of Formula (X) in conjugates of Formula (X), m is 1, and linker, L, has Formula (XII). [00386] In some embodiments, conjugates of Formula (X) may comprise a disulfide-containing linker.
- linker, L is a cleavable linker having Formula (XIII): wherein: Z is a linking group that joins the linker to a target group on targeting moiety, T; Q is –(CH 2 )p- or –(CH 2 CH 2 O)q-, wherein p and q are each independently an integer between 1 and 10; each R is independently H or C 1 -C 6 alkyl; n is 1, 2 or 3; # is the point of attachment to targeting moiety, T, and % is the point of attachment to the camptothecin analogue, D.
- Z is a linking group that joins the linker to a target group on targeting moiety, T
- Q is –(CH 2 )p- or –(CH 2 CH 2 O)q-, wherein p and q are each independently an integer between 1 and 10
- each R is independently H or C 1 -C 6 alkyl
- n is 1, 2 or 3
- # is the point of attachment to targeting moiety, T
- conjugates of Formula (X) in conjugates of Formula (X), m is 1, and linker, L, has Formula (XIII).
- conjugates of Formula (X) may comprise a ⁇ -glucuronide- containing linker.
- Various non-cleavable linkers are known in the art for linking drugs to targeting moieties and may be useful in the conjugate compositions of the present disclosure in certain embodiments.
- non-cleavable linkers include linkers having an N-succinimidyl ester or N- sulfosuccinimidyl ester moiety for reaction with the cell binding agent, as well as a maleimido- or haloacetyl-based moiety for reaction with the drug, or vice versa.
- An example of such a non- cleavable linker is based on sulfosuccinimidyl-4-[N-maleimidomethyl]cyclohexane-1-carboxylate (sulfo-SMCC).
- Sulfo-SMCC conjugation typically occurs via a maleimide group which reacts with sulfhydryls (thiols, —SH) on compound D, while the sulfo-NHS ester is reactive toward primary amines (as found in lysine and at the N-terminus of proteins or peptides) on targeting moiety T.
- thiols thiols, —SH
- linkers include those based on N-succinimidyl 4- (maleimidomethyl)cyclohexanecarboxylate (SMCC), N-succinimidyl-4-(N-maleimidomethyl)- cyclohexane-1-carboxy-(6-amidocaproate) (“long chain” SMCC or LC-SMCC), K- maleimidoundecanoic acid N-succinimidyl ester (KMUA), ⁇ -maleimidobutyric acid N- succinimidyl ester (GMBS), ⁇ -maleimidocaproic acid N-hydroxysuccinimide ester (EMCS), m- maleimidobenzoyl-N-hydroxysuccinimide ester (MBS), N-( ⁇ -maleimidoacetoxy)-succinimide ester (AMAS), succinimidyl-6-( ⁇ -maleimidopropionamido)hex
- SMCC N
- Non-limiting examples of drug-linkers comprising camptothecin analogues of Formula (I) are shown in Table 4 (Fig.4), Table 5 (Fig.5) and Table 6 (Fig.6).
- conjugates comprising these drug-linkers are shown in Table 7 (Fig.7), Table 8 (Fig.8) and Table 9 (Fig.9).
- the conjugate of Formula (X) comprises a drug-linker selected from the drug-linkers shown in Tables 4, 5 and 6.
- the conjugate of Formula (X) is selected from the conjugates shown in Tables 7, 8 and 9, where T is the targeting moiety and n is an integer between 1 and 10.
- the conjugate of Formula (X) is selected from the conjugates shown in Tables 7, 8 and 9, where T is the targeting moiety and n is an integer between 2 and 8.
- the conjugate of Formula (X) is selected from the conjugates shown in Tables 7, 8 and 9, where T is an antibody or antigen-binding antibody fragment.
- Conjugates of Formula (X) may be prepared by standard methods known in the art (see, for example, Bioconjugate Techniques (G.T. Hermanson, 2013, Academic Press)). Various linkers and linker components are commercially available or may be prepared using standard synthetic organic chemistry techniques (see, for example, March’s Advanced Organic Chemistry (Smith & March, 2006, Sixth Ed., Wiley); Toki et al., (2002) J. Org. Chem. 67:1866-1872; Frisch et al., (1997) Bioconj. Chem.
- preparation of the conjugates comprises first preparing a drug-linker, D-L, comprising one or more camptothecin analogues of Formula (I) and linker L, and then conjugating the drug-linker, D-L, to an appropriate group on targeting moiety, T.
- Linker, L, to targeting moiety, T, and subsequent ligation of the targeting moiety-linker, T-L, to one or more camptothecin analogues of Formula (I), D is an alternative approach that may be employed in some embodiments.
- Suitable groups on compounds of Formula (I), D, for attachment of linker, L, in either of the above approaches include, but are not limited to, thiol groups, amine groups, carboxylic acid groups and hydroxyl groups.
- linker, L is attached to a compound of Formula (I), D, via a hydroxyl or amine group on the compound.
- Suitable groups on targeting moiety, T, for attachment of linker, L, in either of the above approaches include sulfhydryl groups (for example, on the side-chain of cysteine residues), amino groups (for example, on the side-chain of lysine residues), carboxylic acid groups (for example, on the side-chains of aspartate or glutamate residues), and carbohydrate groups.
- targeting moiety T may comprise one or more naturally occurring sulfhydryl groups allowing targeting moiety, T, to bond to linker, L, via the sulfur atom of a sulfhydryl group.
- targeting moiety, T may comprise one or more lysine residues that can be chemically modified to introduce one or more sulfhydryl groups.
- Reagents that can be used to modify lysine residues include, but are not limited to, N-succinimidyl S-acetylthioacetate (SATA), N-succinimidyl-3-(2-pyridyldithio)propionate (“SPDP”) and 2-iminothiolane hydrochloride (Traut’s Reagent).
- SATA N-succinimidyl S-acetylthioacetate
- SPDP N-succinimidyl-3-(2-pyridyldithio)propionate
- 2-iminothiolane hydrochloride Trimethoxys Reagent
- targeting moiety, T may comprise one or more carbohydrate groups that can be chemically modified to include one or more sulfhydryl groups.
- Carbohydrate groups on targeting moiety, T may also be oxidized to provide an aldehyde ( ⁇ CHO) group (see, for example, Laguzza et al., 1989, J. Med. Chem.32(3):548-55), which could subsequently be reacted with linker, L, for example, via a hydrazine or hydroxylamine group on linker, L.
- Targeting moiety, T may also be modified to include additional cysteine residues (see, for example, U.S. Patent Nos.
- non-natural amino acids that provide reactive handles, such as selenomethionine, p-acetylphenylalanine, formylglycine or p-azidomethyl-L-phenylalanine (see, for example, Hofer et al., 2009, Biochemistry, 48:12047- 12057; Axup et al., 2012, PNAS, 109:16101-16106; Wu et al., 2009, PNAS, 106:3000-3005; Zimmerman et al., 2014, Bioconj. Chem., 25:351-361), to allow for site-specific conjugation.
- selenomethionine p-acetylphenylalanine
- formylglycine or p-azidomethyl-L-phenylalanine
- targeting moiety, T may be modified to include a non-natural reactive group, such as an azide, that allows for conjugation to the linker via a complementary reactive group on the linker, for example, for example, by click chemistry (see, for example, Chio & Bane, 2020, Methods Mol. Biol., 2078:83-97).
- a non-natural reactive group such as an azide
- a complementary reactive group on the linker for example, for example, by click chemistry (see, for example, Chio & Bane, 2020, Methods Mol. Biol., 2078:83-97).
- Other protocols for the modification of proteins for the attachment or association of linker, L are known in the art and include those described in Coligan et al., Current Protocols in Protein Science, vol.2, John Wiley & Sons (2002).
- targeting moiety is an antibody
- several different reactive groups on the antibody may function as a conjugation site, including ⁇ -amino groups on lysine residues, pendant carbohydrate moieties, side-chain carboxylic acid groups on aspartate or glutamate residues, cysteine-cysteine disulfide groups and cysteine thiol groups.
- the amino acids used for conjugation may be part of the natural sequence of the antibody, or they may be introduced by site-specific engineering techniques known in the art, as noted above.
- antibody-drug conjugates may be prepared using the enzyme transglutaminase, for example, bacterial transglutaminase (BTG) from Streptomyces mobaraensis (see, for example, Jeger et al., 2010, Angew. Chem. Int. Ed., 49:9995-9997).
- BTG forms an amide bond between the side chain carboxamide of a glutamine (the amine acceptor, typically on the antibody) and an alkyleneamino group (the amine donor, typically on the drug-linker), which can be, for example, the ⁇ -amino group of a lysine or a 5-amino-n-pentyl group.
- Antibodies may also be modified to include a glutamine containing peptide, or “tag,” which allows BTG conjugation to be used to conjugate the antibody to a drug-linker (see, for example, U.S. Patent Application Publication No. US 2013/0230543 and International (PCT) Publication No. WO 2016/144608).
- a similar conjugation approach utilizes the enzyme sortase A.
- the antibody is typically modified to include the sortase A recognition motif (LPXTG, where X is any natural amino acid) and the drug-linker is designed to include an oligoglycine motif (typically GGG) to allow for sortase A-mediated transpeptidation (see, for example, Beerli, et al., 2015, PLos One, 10:e0131177; Chen et al., 2016, Nature:Scientific Reports, 6:31899).
- GGG oligoglycine motif
- the “drug-to-antibody ratio” or DAR) may be determined by standard techniques such as UV/VIS spectroscopic analysis, ELISA-based techniques, chromatography techniques such as hydrophobic interaction chromatography (HIC), UV-MALDI mass spectrometry (MS) and MALDI-TOF MS.
- chromatography techniques such as hydrophobic interaction chromatography (HIC), UV-MALDI mass spectrometry (MS) and MALDI-TOF MS.
- distribution of drug-linked forms for example, the fraction of targeting moiety, T, containing zero, one, two, three, etc. compounds of Formula (I), D
- T the fraction of targeting moiety
- T containing zero, one, two, three, etc. compounds of Formula (I), D
- compositions may be prepared by known procedures using well- known and readily available ingredients.
- Pharmaceutical compositions may be formulated for administration to a subject by, for example, oral (including, for example, buccal or sublingual), topical, parenteral, rectal or vaginal routes, or by inhalation or spray.
- parenteral as used herein includes subcutaneous injection, and intradermal, intra-articular, intravenous, intramuscular, intravascular, intrasternal, intrathecal injection or infusion.
- compositions intended for oral use may be prepared in either solid or fluid unit dosage forms.
- Fluid unit dosage forms may be prepared according to procedures known in the art for the manufacture of pharmaceutical compositions and such compositions may contain one or more agents such as sweetening agents, flavouring agents, colouring agents and preserving agents in order to provide pharmaceutically elegant and palatable preparations.
- An elixir may be prepared by using a hydroalcoholic (for example, ethanol) carrier with suitable sweeteners such as sugar and/or saccharin, together with an aromatic flavoring agent.
- Suspensions may be prepared with an aqueous carrier and a suspending agent such as acacia, tragacanth, methylcellulose and the like.
- Solid formulations, such as tablets, contain the active ingredient in admixture with non- toxic pharmaceutically acceptable excipients that are suitable for the manufacture of tablets.
- excipients may be for example, inert diluents, such as calcium carbonate, sodium carbonate, lactose, calcium phosphate or sodium phosphate; granulating and disintegrating agents for example, corn starch, or alginic acid; binding agents, for example starch, gelatin or acacia, and/or lubricating agents, for example magnesium stearate, stearic acid or talc, as well as other conventional ingredients such as dicalcium phosphate, magnesium aluminum silicate, calcium sulfate, starch, lactose, methylcellulose, and functionally similar materials.
- inert diluents such as calcium carbonate, sodium carbonate, lactose, calcium phosphate or sodium phosphate
- granulating and disintegrating agents for example, corn starch, or alginic acid
- binding agents for example starch, gelatin or acacia
- lubricating agents for example magnesium stearate, stearic acid or talc, as
- the tablets may be uncoated or they may be coated by known techniques, for example, in order to delay disintegration and absorption in the gastrointestinal tract and thereby provide a sustained action over a longer period.
- a time delay material such as glyceryl monostearate or glyceryl distearate may be employed.
- Formulations for oral use may also be presented as hard gelatin capsules in which the active ingredient is mixed with an inert solid diluent, for example, calcium carbonate, calcium phosphate or kaolin, or as soft gelatin capsules in which the active ingredient is mixed with water or an oil medium, for example peanut oil, liquid paraffin or olive oil.
- Soft gelatin capsules are typically prepared by machine encapsulation of a slurry of the active ingredient with an acceptable vegetable oil, light liquid petrolatum or other inert oil.
- Aqueous suspensions contain the active ingredient in admixture with excipients suitable for the manufacture of aqueous suspensions.
- excipients include suspending agents, for example sodium carboxylmethylcellulose, methyl cellulose, hydropropylmethylcellulose, sodium alginate, polyvinylpyrrolidone, gum tragacanth and gum acacia, and dispersing or wetting agents.
- Dispersing and wetting agents include, for example, naturally-occurring phosphatides (for example, lecithin), condensation products of an alkylene oxide with fatty acids (for example, polyoxyethylene stearate), condensation products of ethylene oxide with long chain aliphatic alcohols (for example, hepta-decaethyleneoxycetanol), condensation products of ethylene oxide with partial esters derived from fatty acids and a hexitol (for example, polyoxyethylene sorbitol monooleate), or condensation products of ethylene oxide with partial esters derived from fatty acids and hexitol anhydrides (for example, polyethylene sorbitan monooleate).
- naturally-occurring phosphatides for example, lecithin
- condensation products of an alkylene oxide with fatty acids for example, polyoxyethylene stearate
- condensation products of ethylene oxide with long chain aliphatic alcohols for example, hepta-decaethyleneoxycetanol
- the aqueous suspensions may also contain one or more preservatives (for example ethyl, or n-propyl-p- hydroxybenzoate), one or more colouring agents, one or more flavouring agents and/or one or more sweetening agents (for example, sucrose or saccharin).
- Oily suspensions may be formulated by suspending the active ingredient in a vegetable oil, for example peanut oil, olive oil, sesame oil or coconut oil, or in a mineral oil such as liquid paraffin.
- the oily suspensions may contain a thickening agent, for example beeswax, hard paraffin or cetyl alcohol. Sweetening agents such as those set forth above, and flavouring agents may be added to provide palatable oral preparations.
- Dispersible powders and granules suitable for preparation of an aqueous suspension by the addition of water typically provide the active ingredient in admixture with a dispersing or wetting agent, suspending agent and one or more preservatives. Suitable dispersing or wetting agents and suspending agents are exemplified by those already mentioned above. One or more additional excipients, for example sweetening, flavouring and/or colouring agents, may also be present.
- Pharmaceutical compositions may also be in the form of oil-in-water emulsions.
- the oil phase may be a vegetable oil, for example olive oil or peanut oil, or a mineral oil, for example liquid paraffin, or mixtures of such oils.
- Suitable emulsifying agents for inclusion in oil-in-water emulsions include, for example, naturally-occurring gums (for example, gum acacia or gum tragacanth), naturally-occurring phosphatides (for example, soy bean, lecithin), or esters or partial esters derived from fatty acids and hexitol anhydrides (for example, sorbitan monooleate) or condensation products of such partial esters with ethylene oxide (for example polyoxyethylene sorbitan monooleate).
- the emulsions may also optionally contain sweetening and/or flavoring agents.
- the pharmaceutical compositions may be in the form of a sterile injectable aqueous or oleaginous solution or suspension. Such suspensions may be formulated using suitable dispersing or wetting agents and suspending agents such as those described above.
- the sterile injectable solution or suspension may comprise the active ingredient in a non-toxic parentally acceptable carrier or diluent. Acceptable carriers and diluents that may be employed include, for example, 1,3-butanediol, water, Ringer’s solution or isotonic sodium chloride solution.
- sterile, fixed oils may be employed as carriers. For this purpose, various bland fixed oils may be employed including synthetic mono- or diglycerides.
- compositions may also be formulated as suppositories for rectal administration. These compositions can be prepared by mixing the active ingredient with a suitable non-irritating excipient which is solid at ordinary temperatures but liquid at physiological temperature and will therefore melt in the rectum to release the drug. Such materials include cocoa butter and polyethylene glycols.
- compositions and methods of preparing pharmaceutical compositions are known in the art and are described, for example, in “Remington: The Science and Practice of Pharmacy” (formerly “Remingtons Pharmaceutical Sciences”); Gennaro, A., Lippincott, Williams & Wilkins, Philadelphia, PA (2000).
- METHODS OF USE Certain embodiments of the present disclosure relate to the therapeutic use of camptothecin analogues of Formula (I) and conjugates comprising these compounds, such as conjugates of Formula (X). Some embodiments relate to the use of compounds of Formula (I) or conjugates of Formula (X) as therapeutic agents.
- Camptothecin analogues of Formula (I) show cytotoxic activity against cancer cells, and compounds of Formula (I) and conjugates comprising these compounds, such as conjugates of Formula (X), are thus useful for inhibiting abnormal cancer cell or tumor cell growth; inhibiting cancer cell or tumor cell proliferation, or treating cancer in a patient.
- compounds of general Formula (I) and conjugates of Formula (X) may be used to treat cancer.
- Some embodiments of the present disclosure thus relate to the use of compounds of general Formula (I) and conjugates of general Formula (X) as anti-cancer agents.
- Certain embodiments of the present disclosure relate to methods of inhibiting the proliferation of cancer or tumor cells comprising contacting the cells with a compound of Formula (I) or a conjugate of Formula (X). Some embodiments relate to a method of killing cancer or tumor cells comprising contacting the cells with a compound of Formula (I) or a conjugate of Formula (X). [00418] Some embodiments relate to methods of treating a subject having a cancer by administering to the subject a compound of Formula (I) or a conjugate of Formula (X).
- treatment with a compound of Formula (I) or a conjugate of Formula (X) may result in one or more of a reduction in the size of a tumor, the slowing or prevention of an increase in the size of a tumor, an increase in the disease-free survival time between the disappearance or removal of a tumor and its reappearance, prevention of a subsequent occurrence of a tumor (for example, metastasis), an increase in the time to progression, reduction of one or more adverse symptom associated with a tumor, and/or an increase in the overall survival time of a subject having cancer.
- Certain embodiments relate to the use of a compound of Formula (I) or a conjugate of Formula (X) in a method of inhibiting tumor growth in a subject.
- Some embodiments relate to the use of a compound of Formula (I) or a conjugate of Formula (X) in a method of inhibiting proliferation of and/or killing cancer cells in vitro. Some embodiments relate to the use of a compound of Formula (I) or a conjugate of Formula (X) in a method of inhibiting proliferation of and/or killing cancer cells in vivo in a subject having a cancer.
- Examples of cancers which may be treated in certain embodiments include hematologic neoplasms, including leukemias, myelomas and lymphomas; carcinomas, including adenocarcinomas and squamous cell carcinomas; melanomas and sarcomas.
- Solid tumors Carcinomas and sarcomas are also frequently referred to as “solid tumors.”
- Examples of commonly occurring solid tumors that may be treated in certain embodiments include, but are not limited to, brain cancer, breast cancer, cervical cancer, colon cancer, head and neck cancer, kidney cancer, lung cancer, ovarian cancer, pancreatic cancer, prostate cancer, stomach cancer, uterine cancer, non-small cell lung cancer (NSCLC) and colorectal cancer.
- NSCLC non-small cell lung cancer
- Various forms of lymphoma also may result in the formation of a solid tumor and, therefore, may also be considered to be solid tumors in certain situations.
- Certain embodiments relate to the use of a compound of Formula (I) or a conjugate of Formula (X) in the treatment of an autoimmune disease, such as atopic dermatitis, rheumatoid arthritis, psoriasis or systemic lupus erythematosus.
- an autoimmune disease such as atopic dermatitis, rheumatoid arthritis, psoriasis or systemic lupus erythematosus.
- Certain embodiments relate to the use of a compound of Formula (I) or a conjugate of Formula (X) in the treatment of a viral infection, such as an HIV infection or SARS coronavirus infection.
- a pharmaceutical composition comprising a compound of Formula (I) or a conjugate of Formula (X) may be provided as part of a pharmaceutical kit or pack.
- kits include, for example, bottles, blister packs, intravenous solution bags, vials and the like, depending on the formulation of the pharmaceutical composition.
- the container may be in a form allowing for administration to a subject, for example, an inhaler, syringe, pipette, eye dropper, pre-soaked gauze or pad, or other such like apparatus, from which the contents may be administered to the subject.
- the kit may further comprise a label or package insert on or associated with the container(s).
- the term “package insert” is used to refer to instructions customarily included in commercial packages of therapeutic products that contain information about the indications, usage, dosage, administration, contraindications and/or warnings concerning the use of such therapeutic products.
- the label or package insert may further include a notice in the form prescribed by a governmental agency regulating the manufacture, use or sale of pharmaceuticals or biological products, which notice reflects approval by the agency of manufacture, for use or sale for human or animal administration.
- the label or package insert typically indicates that the compound or conjugate is for use to treat the condition of choice, for example, cancer.
- kits may be lyophilized or provided in a dry form, such as a powder or granules, and the kit can additionally contain a suitable solvent for reconstitution of the lyophilized or dried component(s).
- a suitable solvent for reconstitution of the lyophilized or dried component(s) may be provided for illustrative purposes and are not intended to limit the scope of the invention in any way.
- starting components may be obtained from commercial sources such as Sigma Aldrich (Merck KGaA), Alfa Aesar and Maybridge (Thermo Fisher Scientific Inc.), Matrix Scientific, Tokyo Chemical Industry Ltd. (TCI) and Fluorochem Ltd., or synthesized according to sources known to those skilled in the art (see, for example, March’s Advanced Organic Chemistry: Reactions, Mechanisms, and Structure, 7th edition, John Wiley & Sons, Inc., 2013) or prepared as described herein.
- BCA bicinchonic acid
- Boc di-tert-butyl dicarbonate
- CE-SDS capillary electrophoresis sodium dodecyl sulfate
- DCM dichloromethane
- DTPA diethylenetriamine pentaacetic acid
- DIPEA N,N-diisopropylethylamine
- DMF dimethylformamide
- DMMTM (4- (4,6-dimethoxy-1,3,5-triazin-2-yl)-4-methyl-morpholinium chloride
- EDC 1-ethyl-3-(3- dimethylaminopropyl)carbodiimide
- Fmoc fluorenylmethyloxycarbonyl
- HATU hexafluorophosphate azabenzotriazole tetramethyl uronium
- HIC hydrophobic interaction chromatography
- HOA hydrophobic interaction chromatography
- Step 1 To a stirring solution of amine compound in dichloromethane or dimethylformamide (0.05 – 0.1 M) was added p-nitrophenyl carbonate (1 eq.) then triethylamine (2 eq.). Upon completion (determined by LCMS typically 1 – 4 h), the reaction mixture was concentrated to dryness then was purified by reverse-phase HPLC to provide the desired PNP- carbamate intermediate after lyophilization. This intermediate can either used to generate a single analogue or be divided into multiple batches in order to generate multiple analogues in the second step.
- Step 2 To the PNP-carbamate intermediate in dimethylformamide (0.1 – 0.2 M) was added the appropriate primary amine (3 eq.). Upon completion (determined by LCMS, typically 1 h), the reaction mixture was purified by reverse-phase HPLC to provide the desired product after lyophilization.
- General Procedure 5 Conversion of amine to carbamate (Synthetic Scheme V; Fig.1E) [00435] To a stirring solution of amine compound in dichloromethane or dimethylformamide (0.05 – 0.1 M) was added p-nitrophenyl carbonate (1 eq.) then triethylamine (2 eq.).
- reverse-phase flash purification was conducting using Biotage® Snap Ultra C18 columns (12, 30, 60, or 120 g), eluting with linear gradients of 0.1% TFA in acetonitrile/ 0.1% TFA in water. Purified compounds were isolated by either removal of organic solvents by rotavap or lyophilization of acetonitrile/water mixtures.
- Preparative HPLC Reverse-phase HPLC of crude compounds was performed using a Luna® 5- ⁇ m C18100 ⁇ (150 ⁇ 30 mm) column (Phenomenex, Torrance, CA) on an Agilent 1260 Infinity II preparative LC/MSD system (Agilent Technologies, Inc., Santa Clara, CA), and eluting with linear gradients of 0.1% TFA in acetonitrile/ 0.1% TFA in water. Purified compounds were isolated by lyophilization of acetonitrile/water mixtures.
- LC/MS Reactions were monitored for completion and purified compounds were analyzed using a Kinetex® 2.6- ⁇ m C18100 ⁇ (30 ⁇ 3 mm) column (Phenomenex, Torrance, CA) on an Agilent 1290 HPLC/ 6120 single quad LC/MS system (Agilent Technologies, Inc., Santa Clara, CA), eluting with a linear 10 to 100% gradient 0.1% formic acid in acetonitrile/ 0.1% formic acid in water.
- NMR 1 H NMR spectra were collected with a Bruker AVANCE III 300 Spectrometer (300 MHz) (Bruker Corporation, Billerica, MA).
- Flash purification was accomplished as described in General Procedure 9, using a 12 g column C18 column and eluting with a 10 to 50% CH 3 CN/H 2 O + 0.1% TFA gradient to give the title compound as an off-white solid (14 mg, 53% yield).
- EXAMPLE 2 PREPARATION OF CAMPTOTHECIN ANALOGUES HAVING METHOXY AT THE C10 POSITION 2.1: 1-(2-amino-4-fluoro-5-methoxyphenyl)-2-chloroethan-1-one (Compound 2.1) [00529] A solution of 3-fluoro-4-methoxyaniline (10 g, 71 mmol) in DCM (100 mL) was cooled to 0 oC.
- EXAMPLE 3 PREPARATION OF CAMPTOTHECIN ANALOGUES HAVING AMINO AT THE C10 POSITION 3.1: 5-bromo-4-fluoro-2-nitrobenzaldehyde (Compound 3.1) [00575] To a stirring solution of HNO 3 (121.2 mL, 67% purity, 2.0 eq.) in H 2 SO 4 (500 mL) at 0 °C was added 3-bromo-4-fluorobenzaldehyde (180 g, 1.0 eq.). After the addition was complete, the ice bath was removed, and the reaction was allowed to stir for 5 h at 25 °C. The mixture was poured into ice (5 L), filtered and then dried under vacuum.
- the reaction mixture was heated at 65 °C while H 2 O 2 (24 mL, 30% purity) was added dropwise over 30 min and then stirred 0.5 h.
- the reaction solution was cooled to 25 °C, then filtered to provide the title compound as a yellow solid (1.53 g, 33.2% yield).
- H 2 O 400 mL
- the pH was adjusted to 7-8 with saturated aqueous Na 2 CO 3 then the solution was concentrated and filtered.
- the solid was triturated with MeOH (30 mL) at 55 °C for 1 h, then filtered, to provide a second batch of the title compound as a brown solid (1.09 g, 26% yield).
- Example 3.12 (S)-9-amino-4-ethyl-8-fluoro-4-hydroxy-11-(morpholinomethyl)-1,12-dihydro- 14H-pyrano[3',4':6,7]indolizino[1,2-b]quinoline-3,14(4H)-dione (Compound 3.12) [00605]
- the title compound was prepared according to General Procedure 1 starting from Compound 3.9 (150 mg) and morpholine.
- Preparative HPLC purification was accomplished as described in General Procedure 9, eluting with a 10 to 60% CH 3 CN/H 2 O + 0.1% TFA gradient to give the title compound as a red solid (TFA salt, 103 mg, 52% yield).
- the Boc protecting group was cleaved in neat TFA (2 mL) followed by precipitation in Et 2 O (100 mL).
- the solid was collected by filtration and added to a solution of 2,5-dioxopyrrolidin-1-yl (2S)-2- [(tert-butoxycarbonyl)amino]-3-phenylpropanoate (600 mg, 1.1 equiv) and N- ethyldiisopropylamine (300 uL) in DMF (7 mL). This solution was stirred at room temperature for 30 min then pipetted into Et2O (100 mL). The precipitate was collected by filtration, dried under vacuum then dissolved in neat TFA (2 mL).
- EXAMPLE 5 In vitro CYTOTOXICITY OF CAMPTOTHECIN ANALOGUES [00843] Cytotoxicity of the camptothecin analogues was assessed in vitro on the following cancer cell lines: SK-BR-3 (breast cancer), SKOV-3 (ovarian cancer), Calu-3 (lung cancer), ZR-75-1 (breast cancer) and MDA-MB-468 (breast cancer). [00844] Briefly, serial dilutions of camptothecin analogues shown in Table 5.1 were prepared in RPMI 1640 + 10% FBS, and 20 uL of each dilution was added to 384-well plates.
- the excess TCEP was removed using appropriate size 40 kD ZebaTM Spin Desalting Columns (Thermo Fisher Scientific, Waltham, MA) equilibrated with 10 mM sodium acetate buffer, pH 4.5.
- the reduced antibody solution was buffer exchanged into PBS, pH 7.4 or into A5Su (10 mM acetate pH 5, 5% sucrose).
- Maleimide functionalized drug-linkers (15 eq) as 10 mM DMSO stocks were added together with as much as 10% DMSO (v/v) in two intervals (7.5 eq each) to the column-purified reduced trastuzumab solution.
- ADCs were prepared using an appropriate size 40 kD ZebaTM Spin Desalting Column (ThermoFisher Scientific, Waltham, MA) pre-equilibrated with 10 mM sodium acetate, pH 4.5. Alternatively, in some instances, ADCs were buffer exchanged to PBS, pH 7.4 or A5Su (10mM sodium acetate, pH 5.0, 9% sucrose).
- the purified conjugates were stored at 4°C and analyzed for total protein content with a BCA assay (either Pierce BCA Protein Assay (catalogue #23225) or Pierce microBCA Protein Assay (catalog #23235; ThermoFisher Scientific, Waltham, MA).
- EXAMPLE 7 CHARACTERIZATION OF ANTI-HER2 ADCS [00848]
- the ADCs from Example 6 were characterized by HPLC-HIC, SEC, CE-SDS and RP- UPLC-MS as described below.
- the average drug-to-antibody ratio (DAR) and DAR distribution were derived from interpretation of the HIC and LC-MS data.
- HIC chromatograms were integrated using appropriate parameters that provided complete, baseline-to-baseline integration of each peak, followed by integration of each peak showing reasonable separation.
- unconjugated trastuzumab was run on the same gradient to obtain the HIC retention time of DAR 0 species.
- Table 7.1 Gradient used for HIC DAR Determination by LC/MS [00852] ADCs were deglycosylated with Endo S for 1 h at room temperature.
- the deglycosylated ADCs were reduced with 50 mM TCEP for 1 h at room temperature and injected onto an Agilent 1290 Infinity II LC coupled to an Agilent 6545 Quadrupole Time of Flight (Q-TOF) mass spectrometer (Agilent Technologies, Inc., Santa Clara, CA). Heavy and light chains were separated using a PLRP-S column (1000 ⁇ , 8 uM, 50 x 2.1 mm) at a flow rate of 0.3 ml/min and a linear gradient of 20 to 40% Mobile Phase A/Mobile Phase B.
- Mobile Phase A 0.1% FA, 0.025% TFA and 10% IPA in water.
- Mobile Phase B 0.1% FA and 10% IPA in acetonitrile.
- Chromatograms were integrated to provide complete, baseline-to-baseline integration of each peak, with reasonably placed separation between partially resolved peaks.
- the peak corresponding to the major component for IgG (approximate retention time 3.3 min) was reported as the monomer based on the SEC profile of unmodified trastuzumab. Any peak occurring prior to 3.3 min was designated as HMWS (high molecular weight species), and any peak occurring after 3.3 min was designated as LMWS (low molecular weight species), excluding solvent peaks (over 5.2 min).
- CE-SDS Analysis Initially, all ADC samples were diluted to 1 mg/mL before preparing the samples in a 96- well PCR plate following manufacturer’s protocol (Protein Express Assay LabChipTM; PerkinElmer, Inc., Waltham, MA). Briefly, 2 ⁇ g of ADC was mixed with 7 uL Protein Express buffer in the presence (reducing) or absence (non-reducing) of 400 mM dithiothreitol (DTT), followed by heat denaturation at 95 °C for 5 minutes. Samples were then diluted in dH 2 O at a 1:2 ratio before data acquisition.
- EXAMPLE 8 In vitro CYTOTOXICITY OF ANTI-HER2 ADCs [00856] In vitro cytotoxicity of select ADCs from Example 6 was tested in SKBR-3 (breast cancer), Calu3 (lung cancer) and MDA-MB-468 (breast cancer) cells using the procedure described in Example 5. The results are shown in Table 8.1. Table 8.1: In vitro Cytotoxicity of ADCs (pIC50)
- EXAMPLE 9 BYSTANDER ACTIVITY OF ANTI-HER2 ADCs [00857] The ability of select ADCs from Example 6 to exert a bystander killing effect on cancer cells was assessed as described below. Bystander killing most commonly occurs after specific uptake of an ADC into an antigen-positive cell. Trafficking and degradation of the ADC results in release of free drug, which then crosses the cell membrane to kill nearby (bystander) cells.
- the ADCs tested were: T-MT-GGFG-AM-Compound 136, T-MT-GGFG-AM- Compound 129, T-MT-GGFG-AM-Compound 139, T-MT-GGFG-Compound 141, T-MT- GGFG-AM-Compound 141, T-MT-GGFG-Compound 145, T-MT-GGFG-Compound 148, T- MT-GGFG-Compound 140, and controls T-MC-GGFG-AM-DXd and T-MT-GGFG-AM-DXd. [00859] Also included was the ADC T-ME-PEG2-GGFG-DXd2.
- the ADC has been shown to lack bystander activity (see Ogitani, et al., 2016, Cancer Sci., 107:1039-1046) and was included as a negative control.
- the ADC comprises trastuzumab (T) conjugated to the drug-linker shown below: ME-PEG2-GGFG-DXd2
- SK-BR3 (HER2+) and MDA-MB-468 (HER2-) cells were seeded either as mono- cultures or co-cultures in a 24-well plate at 30,000 cells and 10,000 cells, respectively, in 250 uL assay media (McCoy’s + 10% FBS).
- ADCs were diluted to 2 nM and 0.2 nM in assay media and 250 uL was added to the cell-containing plates (1 and 0.1 nM final ADC concentration). Cells were incubated with test ADCs for 4 d at 37 °C and detached by TrypLETM Express Enzyme (ThermoFisher Scientific, Waltham, MA).
- YO-PRO®-1 ThermoFisher Scientific, Waltham, MA
- Alexa Fluor® 647 Biolegend, Inc., San Diego, CA; Catalogue # 324412. After 20 min incubation at room temperature, cells were washed in FACS buffer and resuspended in 100 uL FACS buffer per well.50 uL were analyzed on the BD FortessaTM flow cytometer (BD Biosciences, San Jose, CA). Dead cells were gated out by YO-PRO®-1 staining.
- the number of SK-BR3 and MDA-MB- 468 cells was then determined by the number of events in the HER2+ and HER2- gates, respectively. % viability was calculated as the number of cells treated divided by the number of cells untreated. [00861] The results for are shown in Fig. 2. Bystander effect was evaluated by comparing the viability of HER2- MDA-MB-468 cells treated as a monoculture (black bars) with that of the cells treated as a co-culture with HER2+ SK-BR-3 cells (grey bars). A greater decrease in viability in co-culture compared with monoculture indicates a higher bystander effect.
- EXAMPLE 10 PLASMA STABILITY OF ANTI-HER2 ADCs
- the stability of select ADCs from Example 6 was tested in mouse plasma as follows. ADCs were diluted in mouse plasma (BioIVT, Westbury, NY) to 0.5 mg/mL and incubated in a 37 °C water bath. Aliquots were taken out at 10 min, 1.5 h, 8 h, 24 h, 72 h and 7 d and frozen at - 80 °C. Once all aliquots were collected, they were thawed and prepared for the affinity capture coupled to LC-MS analysis.
- Biotinylated goat anti-human IgG F(ab’) 2 (Jackson ImmunoResearch Laboratories, Inc., West Grove, PA) was coupled to Streptavidin Mag Sepharose® beads (GE Healthcare Bio Sciences, Uppsala, Sweden) for 30 min at room temperature prior to use.
- Mouse plasma samples containing approximately 2 ug ADC were diluted in PBS and deglycosylated with EndoS for 1 h at room temperature. Capture antibody-streptavidin bead mixture was added to the deglycosylated sample and incubated for 1.5 h at room temperature.
- EXAMPLE 10 In vivo EVALUATION OF ANT-HER2 ADCs [00865] The anti-tumor activity of select ADCs from Example 6 was investigated in a JIMT-1 xenograft model of breast cancer expressing HER2 (mid) as described below.
- the ADCs evaluated were: T-MT-GGFG-AM-Compound 136, T-MT-GGFG-AM-Compound 129, T-MT-GGFG-AM- Compound 139, T-MT-GGFG-Compound 140, T-MC-GGFG-AM-Compound 141, T-MT- GGFG-AM-Compound 141, T-MT-GGFG-Compound 141, T-MT-GGFG-Compound 145 and T- MT-GGFG-Compound 148, and control T-MC-GGFG-AM-DXd.
- Tumor cell suspensions (5 x 10 6 cells in 0.1 mL PBS) were implanted subcutaneously into female CB17/scid mice.
- EXAMPLE 12 PREPARATION OF ANTI-FR ⁇ ANTIBODY-DRUG CONJUGATES [00869] ADCs comprising select drug-linkers prepared as described in Example 4 were conjugated to two anti-folate receptor alpha (FR ⁇ ) antibodies (v36675 and v30384; see Table 12.1). Exemplary conjugation protocols are provided below.
- FR ⁇ anti-folate receptor alpha
- v36675-MC-GGFG-AM-DXd1 A solution (83.5 mL) of the anti-FR ⁇ antibody v36675 (1.5 g) in PBS, pH 7.4 was reduced by addition of 5 mM DTPA (24 mL in PBS, pH adjusted to 7.4) and 10 mM of an aqueous TCEP solution (12.5 mL, 12 eq.).
- the reduced antibody was diluted to 125 mL with PBS and purified using a Pellicon® XL Ultrafiltration Module (Ultracel 30 kDa 0.005m 2 ; MilliporeSigma, Burlington, MA; PXC030C50) with approximately 5 diavolumes of 10 mM NaOAc, pH 5.5.
- the purified antibody (1133 mg) was diluted to a final volume of 211 mL using 10 mM NaOAc, pH 5.5.
- To the antibody solution was added 6.4 mL of DMSO and an excess of drug-linker (9.43 mL; 12 eq.) from a 10 mM DMSO stock solution.
- EXAMPLE 13 PURIFICATION AND CHARACTERIZATION OF ANTI-FR ⁇ ADCs
- the quenched ADC solution from Example 12 was diluted to approximately 5 mg/mL with 10 mM NaOAc, pH 5.5 and purified using a Pellicon® XL Ultrafiltration Module (Ultracel 30 kDa 0.005m 2 ; MilliporeSigma, Burlington, MA; PXC030C50) with 11 diavolumes of 10 mM NaOAc, pH 4.5, followed by 4 diavolumes of 10 mM NaOAc, pH 4.5 with 9% (v/v) sucrose.
- the purified ADC was then sterile filtered (0.2 ⁇ m).
- the concentration of the ADCs was determined by a BCA assay with reference to a standard curve generated using the antibody v36675, estimated by measurement of absorption at 280 nm using extinction coefficients taken from the literature (European Patent No.3342785, for MC-GGFG-AM-DXd1), or determined experimentally (for the remaining drug- linkers).
- ADCs were also characterized by hydrophobic interaction chromatography (HIC) and size exclusion chromatography (SEC) as described below. Hydrophobic Interaction Chromatography [00876]
- HIC hydrophobic interaction chromatography
- SEC size exclusion chromatography
- Antibody and ADCs were analyzed by HIC to estimate the drug-to-antibody ratio (DAR).
- KB-HeLa endocervical carcinoma
- JEG-3 choriocarcinoma
- T-47D breast carcinoma
- MDA-MB-468 breast adenocarcinoma
- % cytotoxicity values were calculated and plotted against test article concentration using GraphPad Prism 9 software (GraphPad Software, San Diego, CA). The results are shown in Table 14.1.
- v30384 ADCs displayed significant cytotoxicity in the FR ⁇ -expressing cell lines KB-HeLa, JEG-3 and T-47D, yielding single-digit nM or lower EC50 values after the 4-day treatment. In the FR ⁇ -negative cell line, MDA-MB-468, the ADCs did not show target-dependent cytotoxicity. Both v30384 and control (palivizumab) ADCs showed comparable potency in this cell line.
- Table 14.1 In vitro Cytotoxicity – 2D Monolayer
- EXAMPLE 15 IN VITRO CYTOTOXICITY OF ANTI-FR ⁇ ADCS – 3D SPHEROIDS
- the cytotoxicity capabilities of select ADCs from Example 12 were assessed in a panel of FR ⁇ -expressing cell line spheroids as described below.
- Cell lines used were IGROV-1 (ovarian adenocarcinoma), T-47D (breast carcinoma), OVCAR-3 (ovarian adenocarcinoma), HEC-1-A (uterine adenocarcinoma) and EBC-1 (lung carcinoma; FR ⁇ -negative).
- ADCs comprising the antibody palivizumab (v21995) were used as non-targeted controls.
- tumor cell suspensions (1 x10 7 cells in 0.1 ml 50% Matrigel®) were implanted subcutaneously into female CB.17 SCID mice.
- mean tumor volume reached 100-150 mm 3
- tumor cell suspensions (1 x10 7 cells in 0.1 ml 50% Matrigel®) were implanted subcutaneously into CB.17 SCID mice.
- the ADCs v30384-MT-GGFG-AM-Compound 139, v30384-MT- GGFG-AM-Compound 141 and v30384-MT-GGFG-Compound 141 all resulted in superior inhibition of tumor growth rate compared to v30384-MC-GGFG-AM-DXd (p ⁇ 0.01).
- v30384-MT-GGFG-Compound 140, v30384-MC-GGFG-Compound 140 and v30384-MT-GGFG- Compound 148 all resulted in stasis of tumor growth for approximately 2 weeks post-dose, which represented a statistically significant inhibition of tumor growth rate compared to each of control, v30384-MC-GGFG-AM-DXd and non-targeted v21995 ADCs (p ⁇ 0.01).
- Test article concentrations were measured in mouse serum by sandwich ELISA utilizing an anti-human IgG1 Fc capture antibody (Jackson Immuno Research Labs, West Grove, PA; Cat.709-005-098) and a HRP-conjugated anti- IgG1 Fab detection antibody (Jackson Immuno Research Labs; Cat. 109-035-097) for total IgG levels. Absorbance at 450nM was measured using a SynergyTM H1 Hybrid Multi-Mode Plate Reader (BioTek Instruments, Winooski, VT). Pharmacokinetics parameters were calculated from non-compartmental analysis using Phoenix WinNonlinTM software (Certara, Princeton, NJ).
- EXAMPLE 18 MURINE TOLERABILITY OF ANTI-FR ⁇ ADCS [00892] Select ADCs from Example 12 were assessed for tolerability in mice at single doses of 60 and 200 mg/kg as follows. Test articles were administered to mice (Balb/c, female, 6-8 weeks old, ⁇ 20g) via 20 ml/kg intraperitoneal injections at 60 and 200 mg/kg. From each dose group, 3 mice were subject to planned observation for 3 weeks post-dose. An additional 3 mice were subject to planned observation for 1 week post-dose, followed by termination and examination of formalin-fixed, paraffin-embedded organs. Mice were euthanized if body weight fell by ⁇ 20 % from pre-dose levels.
- ADCs v30384-MT-GGFG-Compound 140, v30384-MT-GGFG-Compound 148 and v30384-MC- GGFG-Compound 140 resulted in rapid body weight loss, mortality or sacrifice due to moribund condition between 3-6 days post dose (see Table 18.1).
- Macroscopic changes considered related to the ADCs were present in preterminal animals treated with 60 and/or 200 mg/kg of v30384-MT- GGFG-Compound 140, v30384-MT-GGFG-Compound 148 and v30384-MC-GGFG-Compound 140.
- Microscopic changes considered related to administration of ADCs v30384-MT-GGFG-Compound 140, v30384-MT-GGFG-Compound 148 and v30384- MC-GGFG-Compound 140 at ⁇ 60 mg/kg dose were present in intestine, bone marrow, thymus, spleen and mesenteric lymph node.
Abstract
Description
Claims
Priority Applications (7)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| BR112023024433A BR112023024433A2 (en) | 2021-05-27 | 2022-05-27 | ANALOGS, CONJUGATES AND METHODS OF USE OF CAMPTOTECIN |
| IL308734A IL308734A (en) | 2021-05-27 | 2022-05-27 | Camptothecin analogues, conjugates and methods of use |
| CA3177067A CA3177067A1 (en) | 2021-05-27 | 2022-05-27 | Camptothecin analogues, conjugates and methods of use |
| CN202280052312.XA CN117715914A (en) | 2021-05-27 | 2022-05-27 | Camptothecin analogs, conjugates, and methods of use |
| EP22810026.9A EP4347601A1 (en) | 2021-05-27 | 2022-05-27 | Camptothecin analogues, conjugates and methods of use |
| KR1020237044336A KR20240031235A (en) | 2021-05-27 | 2022-05-27 | Camptothecin analogues, conjugates and methods of use |
| AU2022282813A AU2022282813A1 (en) | 2021-05-27 | 2022-05-27 | Camptothecin analogues, conjugates and methods of use |
Applications Claiming Priority (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202163194138P | 2021-05-27 | 2021-05-27 | |
| US63/194,138 | 2021-05-27 | ||
| US202163203667P | 2021-07-27 | 2021-07-27 | |
| US63/203,667 | 2021-07-27 | ||
| US202163290587P | 2021-12-16 | 2021-12-16 | |
| US63/290,587 | 2021-12-16 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2022246576A1 true WO2022246576A1 (en) | 2022-12-01 |
Family
ID=84229147
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/CA2022/050864 WO2022246576A1 (en) | 2021-05-27 | 2022-05-27 | Camptothecin analogues, conjugates and methods of use |
Country Status (7)
| Country | Link |
|---|---|
| EP (1) | EP4347601A1 (en) |
| KR (1) | KR20240031235A (en) |
| AU (1) | AU2022282813A1 (en) |
| BR (1) | BR112023024433A2 (en) |
| CA (1) | CA3177067A1 (en) |
| IL (1) | IL308734A (en) |
| WO (1) | WO2022246576A1 (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2023216956A1 (en) * | 2022-05-13 | 2023-11-16 | 四川科伦博泰生物医药股份有限公司 | Camptothecin compound, preparation method therefor and use thereof |
| WO2023178289A3 (en) * | 2022-03-17 | 2023-12-14 | Seagen Inc. | Camptothecin conjugates |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1995022549A1 (en) * | 1994-02-16 | 1995-08-24 | Pharmacia S.P.A. | Water-soluble camptothecin derivatives, process for their preparation and their use as antitumor agents |
| WO2018175994A1 (en) * | 2017-03-24 | 2018-09-27 | Seattle Genetics, Inc. | Process for the preparation of glucuronide drug-linkers and intermediates thereof |
| CN110128501A (en) * | 2019-05-21 | 2019-08-16 | 北京海美源医药科技有限公司 | A kind of camptothecine compounds and its preparation method and application targeting FAP enzyme |
-
2022
- 2022-05-27 BR BR112023024433A patent/BR112023024433A2/en unknown
- 2022-05-27 AU AU2022282813A patent/AU2022282813A1/en active Pending
- 2022-05-27 CA CA3177067A patent/CA3177067A1/en active Pending
- 2022-05-27 KR KR1020237044336A patent/KR20240031235A/en unknown
- 2022-05-27 WO PCT/CA2022/050864 patent/WO2022246576A1/en active Application Filing
- 2022-05-27 EP EP22810026.9A patent/EP4347601A1/en active Pending
- 2022-05-27 IL IL308734A patent/IL308734A/en unknown
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1995022549A1 (en) * | 1994-02-16 | 1995-08-24 | Pharmacia S.P.A. | Water-soluble camptothecin derivatives, process for their preparation and their use as antitumor agents |
| WO2018175994A1 (en) * | 2017-03-24 | 2018-09-27 | Seattle Genetics, Inc. | Process for the preparation of glucuronide drug-linkers and intermediates thereof |
| CN110128501A (en) * | 2019-05-21 | 2019-08-16 | 北京海美源医药科技有限公司 | A kind of camptothecine compounds and its preparation method and application targeting FAP enzyme |
Non-Patent Citations (4)
| Title |
|---|
| BURKE, P. J. ET AL.: "Design, Synthesis, and Biological Evaluation of Antibody-Drug Conjugates Comprised of Potent Camptothecin Analogues", BIOCONJUGATE CHEMISTRY, vol. 10, 2009, pages 1242 - 1250, XP055079987, DOI: 10.1021/bc9001097 * |
| GUIOTTO ANDREA, CANEVARI MIRTA, ORSOLINI PIERO, LAVANCHY OLIVIER, DEUSCHEL CHRISTINE, KANEDA NORIMASA, KURITA AKINOBU, MATSUZAKI T: "Efficient and chemoselective N-acylation of 10-amino-7-ethyl camptothecin with poly(ethylene glycol)", BIOORGANIC & MEDICINAL CHEMISTRY LETTERS, ELSEVIER, AMSTERDAM NL, vol. 14, no. 7, 1 April 2004 (2004-04-01), Amsterdam NL , pages 1803 - 1805, XP093011086, ISSN: 0960-894X, DOI: 10.1016/j.bmcl.2003.12.098 * |
| GUITTO, A. ET AL.: "Synthesis, Characterization, and Preliminary in Vivo Tests of New Poly(ethylene glycol) Conjugates of the Antitumor agent 10-Amino-7-ethylcamptothecin", JOURNAL OF MEDICINAL CHEMISTRY, vol. 47, 2004, pages 1280 - 1289, XP009121059, DOI: 10.1021/jm031072e * |
| SONG Y L: "Flexible Molecular Docking Studies of Antineoplastic Camptothecin Derivatives on DNA-topoisomerase I Complex", ACTA CHIMICA SINICA, vol. 61, no. 11, 1 January 2003 (2003-01-01), XP093011089 * |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2023178289A3 (en) * | 2022-03-17 | 2023-12-14 | Seagen Inc. | Camptothecin conjugates |
| WO2023216956A1 (en) * | 2022-05-13 | 2023-11-16 | 四川科伦博泰生物医药股份有限公司 | Camptothecin compound, preparation method therefor and use thereof |
Also Published As
| Publication number | Publication date |
|---|---|
| EP4347601A1 (en) | 2024-04-10 |
| BR112023024433A2 (en) | 2024-02-20 |
| IL308734A (en) | 2024-01-01 |
| CA3177067A1 (en) | 2022-12-01 |
| KR20240031235A (en) | 2024-03-07 |
| AU2022282813A1 (en) | 2023-11-30 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6598821B2 (en) | Novel maytansinoid derivatives having peptide linkers and conjugates thereof | |
| US9249186B2 (en) | Cytotoxic peptides and antibody drug conjugates thereof | |
| RU2669807C2 (en) | Bifunctional cytotoxic agents | |
| CN113766954A (en) | Camptothecin derivatives | |
| JP2018502131A (en) | Heteroarylene-bridged benzodiazepine dimers, conjugates thereof, and methods of making and using | |
| CA2973354A1 (en) | Benzodiazepine dimers, conjugates thereof, and methods of making and using | |
| US11365263B2 (en) | Bifunctional cytotoxic agents containing the CTI pharmacophore | |
| WO2022246576A1 (en) | Camptothecin analogues, conjugates and methods of use | |
| CA2837327A1 (en) | Dna minor groove binding agent immunoconjugates, composition containing them and methods of making and use | |
| CA2833477A1 (en) | Novel binder-drug conjugates (adcs) and their use | |
| US20220111067A1 (en) | Maytansinoid derivatives with self-immolative peptide linkers and conjugates thereof | |
| US11712480B2 (en) | Heteroaryl sulfone-based conjugation handles, methods for their preparation, and their use in synthesizing antibody drug conjugates | |
| ES2955886T3 (en) | Method and molecules | |
| TW202400137A (en) | Camptothecin conjugates | |
| JP2023520605A (en) | Camptothecin derivatives and their conjugates | |
| JP2024511360A (en) | Selective drug release from internalization complexes of bioactive compounds | |
| CN110312730A (en) | Cytotoxin and conjugate, its Use and preparation method | |
| CN117715914A (en) | Camptothecin analogs, conjugates, and methods of use | |
| EP3271365B1 (en) | Bifunctional cytotoxic agents containing the cti pharmacophore | |
| WO2024020684A1 (en) | Immunomodulator purine-derived compounds, conjugates thereof, and methods of use thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 22810026 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2022282813 Country of ref document: AU Ref document number: AU2022282813 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: MX/A/2023/013738 Country of ref document: MX |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 308734 Country of ref document: IL |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2023572596 Country of ref document: JP |
|
| ENP | Entry into the national phase |
Ref document number: 2022282813 Country of ref document: AU Date of ref document: 20220527 Kind code of ref document: A |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: 112023024433 Country of ref document: BR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 202393362 Country of ref document: EA |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2022810026 Country of ref document: EP |
|
| ENP | Entry into the national phase |
Ref document number: 2022810026 Country of ref document: EP Effective date: 20240102 |
|
| ENP | Entry into the national phase |
Ref document number: 112023024433 Country of ref document: BR Kind code of ref document: A2 Effective date: 20231122 |