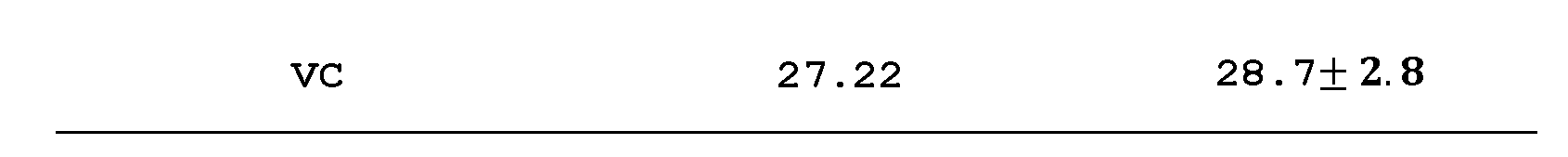WO2022109685A1 - Complex materials - Google Patents
Complex materials Download PDFInfo
- Publication number
- WO2022109685A1 WO2022109685A1 PCT/AU2021/051429 AU2021051429W WO2022109685A1 WO 2022109685 A1 WO2022109685 A1 WO 2022109685A1 AU 2021051429 W AU2021051429 W AU 2021051429W WO 2022109685 A1 WO2022109685 A1 WO 2022109685A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- complex
- carbides
- metal
- carbide
- complex carbide
- Prior art date
Links
- 239000011365 complex material Substances 0.000 title description 3
- 229910052751 metal Inorganic materials 0.000 claims abstract description 118
- 239000002184 metal Substances 0.000 claims abstract description 113
- 229910052723 transition metal Inorganic materials 0.000 claims abstract description 41
- 150000003624 transition metals Chemical class 0.000 claims abstract description 40
- 229910052500 inorganic mineral Inorganic materials 0.000 claims abstract description 21
- 239000011707 mineral Substances 0.000 claims abstract description 21
- 238000005065 mining Methods 0.000 claims abstract description 21
- 238000012545 processing Methods 0.000 claims abstract description 21
- 150000001247 metal acetylides Chemical class 0.000 claims description 225
- -1 7C3 carbides Chemical class 0.000 claims description 69
- 229910052721 tungsten Inorganic materials 0.000 claims description 56
- 239000000919 ceramic Substances 0.000 claims description 44
- 229910052804 chromium Inorganic materials 0.000 claims description 44
- 229910052720 vanadium Inorganic materials 0.000 claims description 44
- 229910052750 molybdenum Inorganic materials 0.000 claims description 27
- 229910052719 titanium Inorganic materials 0.000 claims description 25
- 238000000034 method Methods 0.000 claims description 22
- 229910052726 zirconium Inorganic materials 0.000 claims description 17
- 229910052758 niobium Inorganic materials 0.000 claims description 16
- 229910052715 tantalum Inorganic materials 0.000 claims description 15
- 229910052727 yttrium Inorganic materials 0.000 claims description 14
- 229910052748 manganese Inorganic materials 0.000 claims description 11
- 229910052706 scandium Inorganic materials 0.000 claims description 11
- 229910001092 metal group alloy Inorganic materials 0.000 claims description 10
- 229910052735 hafnium Inorganic materials 0.000 claims description 9
- 229910052742 iron Inorganic materials 0.000 claims description 9
- 239000000758 substrate Substances 0.000 claims description 7
- 238000005552 hardfacing Methods 0.000 claims description 6
- 239000002245 particle Substances 0.000 claims description 6
- 229910052725 zinc Inorganic materials 0.000 claims description 5
- 238000005266 casting Methods 0.000 claims description 4
- 238000005253 cladding Methods 0.000 claims description 2
- 239000002923 metal particle Substances 0.000 claims description 2
- 239000010936 titanium Substances 0.000 description 60
- 239000000463 material Substances 0.000 description 37
- 239000010937 tungsten Substances 0.000 description 35
- 125000004429 atom Chemical group 0.000 description 33
- 230000015572 biosynthetic process Effects 0.000 description 30
- 239000013078 crystal Substances 0.000 description 27
- 150000004767 nitrides Chemical class 0.000 description 27
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 22
- 238000004364 calculation method Methods 0.000 description 22
- 239000011780 sodium chloride Substances 0.000 description 21
- 238000010801 machine learning Methods 0.000 description 20
- 150000002739 metals Chemical class 0.000 description 20
- 239000000203 mixture Substances 0.000 description 20
- 235000002639 sodium chloride Nutrition 0.000 description 20
- 229910052581 Si3N4 Inorganic materials 0.000 description 18
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 17
- 235000010755 mineral Nutrition 0.000 description 17
- 238000012360 testing method Methods 0.000 description 17
- 230000006399 behavior Effects 0.000 description 16
- 239000010439 graphite Substances 0.000 description 16
- 229910002804 graphite Inorganic materials 0.000 description 16
- 230000000875 corresponding effect Effects 0.000 description 15
- 239000000523 sample Substances 0.000 description 14
- 238000002441 X-ray diffraction Methods 0.000 description 13
- 239000011159 matrix material Substances 0.000 description 13
- 239000007787 solid Substances 0.000 description 13
- 238000003775 Density Functional Theory Methods 0.000 description 12
- 238000005275 alloying Methods 0.000 description 12
- 229910052799 carbon Inorganic materials 0.000 description 12
- 150000001875 compounds Chemical class 0.000 description 12
- 238000002844 melting Methods 0.000 description 11
- 230000008018 melting Effects 0.000 description 11
- 239000003607 modifier Substances 0.000 description 11
- 238000002149 energy-dispersive X-ray emission spectroscopy Methods 0.000 description 10
- 239000000843 powder Substances 0.000 description 10
- 230000002787 reinforcement Effects 0.000 description 10
- 238000009826 distribution Methods 0.000 description 9
- 238000005457 optimization Methods 0.000 description 9
- 238000000576 coating method Methods 0.000 description 8
- 239000002131 composite material Substances 0.000 description 8
- 238000013459 approach Methods 0.000 description 7
- 230000007423 decrease Effects 0.000 description 7
- 239000000126 substance Substances 0.000 description 7
- 238000004458 analytical method Methods 0.000 description 6
- 239000000470 constituent Substances 0.000 description 6
- 230000003247 decreasing effect Effects 0.000 description 6
- 238000013461 design Methods 0.000 description 6
- 238000004519 manufacturing process Methods 0.000 description 6
- OAXLZNWUNMCZSO-UHFFFAOYSA-N methanidylidynetungsten Chemical compound [W]#[C-] OAXLZNWUNMCZSO-UHFFFAOYSA-N 0.000 description 6
- 230000002596 correlated effect Effects 0.000 description 5
- 230000003993 interaction Effects 0.000 description 5
- 238000012986 modification Methods 0.000 description 5
- LEONUFNNVUYDNQ-UHFFFAOYSA-N vanadium atom Chemical group [V] LEONUFNNVUYDNQ-UHFFFAOYSA-N 0.000 description 5
- 102100021164 Vasodilator-stimulated phosphoprotein Human genes 0.000 description 4
- 230000004888 barrier function Effects 0.000 description 4
- 230000008901 benefit Effects 0.000 description 4
- 238000006243 chemical reaction Methods 0.000 description 4
- 230000006870 function Effects 0.000 description 4
- 230000007246 mechanism Effects 0.000 description 4
- 230000004048 modification Effects 0.000 description 4
- 230000008569 process Effects 0.000 description 4
- 239000011253 protective coating Substances 0.000 description 4
- 238000012827 research and development Methods 0.000 description 4
- 238000001878 scanning electron micrograph Methods 0.000 description 4
- 238000012546 transfer Methods 0.000 description 4
- WFKWXMTUELFFGS-UHFFFAOYSA-N tungsten Chemical compound [W] WFKWXMTUELFFGS-UHFFFAOYSA-N 0.000 description 4
- 229910045601 alloy Inorganic materials 0.000 description 3
- 239000000956 alloy Substances 0.000 description 3
- 230000006835 compression Effects 0.000 description 3
- 238000007906 compression Methods 0.000 description 3
- 150000002500 ions Chemical class 0.000 description 3
- 238000004372 laser cladding Methods 0.000 description 3
- 230000003647 oxidation Effects 0.000 description 3
- 238000007254 oxidation reaction Methods 0.000 description 3
- 239000008188 pellet Substances 0.000 description 3
- 238000012216 screening Methods 0.000 description 3
- 239000007858 starting material Substances 0.000 description 3
- 238000006467 substitution reaction Methods 0.000 description 3
- UONOETXJSWQNOL-UHFFFAOYSA-N tungsten carbide Chemical compound [W+]#[C-] UONOETXJSWQNOL-UHFFFAOYSA-N 0.000 description 3
- 238000007514 turning Methods 0.000 description 3
- 238000009827 uniform distribution Methods 0.000 description 3
- 108010054220 vasodilator-stimulated phosphoprotein Proteins 0.000 description 3
- 229920001479 Hydroxyethyl methyl cellulose Polymers 0.000 description 2
- 229910000990 Ni alloy Inorganic materials 0.000 description 2
- 238000010314 arc-melting process Methods 0.000 description 2
- 230000003190 augmentative effect Effects 0.000 description 2
- 239000002585 base Substances 0.000 description 2
- 125000004432 carbon atom Chemical group C* 0.000 description 2
- 150000004696 coordination complex Chemical class 0.000 description 2
- RKTYLMNFRDHKIL-UHFFFAOYSA-N copper;5,10,15,20-tetraphenylporphyrin-22,24-diide Chemical group [Cu+2].C1=CC(C(=C2C=CC([N-]2)=C(C=2C=CC=CC=2)C=2C=CC(N=2)=C(C=2C=CC=CC=2)C2=CC=C3[N-]2)C=2C=CC=CC=2)=NC1=C3C1=CC=CC=C1 RKTYLMNFRDHKIL-UHFFFAOYSA-N 0.000 description 2
- 238000005520 cutting process Methods 0.000 description 2
- 238000011161 development Methods 0.000 description 2
- 238000005553 drilling Methods 0.000 description 2
- 238000002474 experimental method Methods 0.000 description 2
- 230000002349 favourable effect Effects 0.000 description 2
- 230000005283 ground state Effects 0.000 description 2
- 101150107144 hemC gene Proteins 0.000 description 2
- 238000007373 indentation Methods 0.000 description 2
- 238000011835 investigation Methods 0.000 description 2
- 230000004807 localization Effects 0.000 description 2
- 230000033001 locomotion Effects 0.000 description 2
- 238000005259 measurement Methods 0.000 description 2
- QSHDDOUJBYECFT-UHFFFAOYSA-N mercury Chemical compound [Hg] QSHDDOUJBYECFT-UHFFFAOYSA-N 0.000 description 2
- 239000011156 metal matrix composite Substances 0.000 description 2
- 238000003801 milling Methods 0.000 description 2
- 230000003287 optical effect Effects 0.000 description 2
- 230000000704 physical effect Effects 0.000 description 2
- 238000002360 preparation method Methods 0.000 description 2
- 230000005855 radiation Effects 0.000 description 2
- 230000002829 reductive effect Effects 0.000 description 2
- 230000003014 reinforcing effect Effects 0.000 description 2
- 230000002441 reversible effect Effects 0.000 description 2
- 238000004626 scanning electron microscopy Methods 0.000 description 2
- 238000004088 simulation Methods 0.000 description 2
- 238000002490 spark plasma sintering Methods 0.000 description 2
- 238000003786 synthesis reaction Methods 0.000 description 2
- 230000002194 synthesizing effect Effects 0.000 description 2
- INZDTEICWPZYJM-UHFFFAOYSA-N 1-(chloromethyl)-4-[4-(chloromethyl)phenyl]benzene Chemical compound C1=CC(CCl)=CC=C1C1=CC=C(CCl)C=C1 INZDTEICWPZYJM-UHFFFAOYSA-N 0.000 description 1
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 1
- 239000004593 Epoxy Substances 0.000 description 1
- 229910015503 Mo5Si3 Inorganic materials 0.000 description 1
- 229910020968 MoSi2 Inorganic materials 0.000 description 1
- 229910017263 Mo—C Inorganic materials 0.000 description 1
- 244000137852 Petrea volubilis Species 0.000 description 1
- 238000000441 X-ray spectroscopy Methods 0.000 description 1
- 238000000367 ab initio method Methods 0.000 description 1
- 229910001514 alkali metal chloride Inorganic materials 0.000 description 1
- 229910001515 alkali metal fluoride Inorganic materials 0.000 description 1
- 229910000287 alkaline earth metal oxide Inorganic materials 0.000 description 1
- 239000012300 argon atmosphere Substances 0.000 description 1
- SKKMWRVAJNPLFY-UHFFFAOYSA-N azanylidynevanadium Chemical compound [V]#N SKKMWRVAJNPLFY-UHFFFAOYSA-N 0.000 description 1
- 150000001768 cations Chemical class 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- 238000005094 computer simulation Methods 0.000 description 1
- 239000004020 conductor Substances 0.000 description 1
- 230000008602 contraction Effects 0.000 description 1
- 229910052802 copper Inorganic materials 0.000 description 1
- 239000010949 copper Substances 0.000 description 1
- 238000005260 corrosion Methods 0.000 description 1
- 230000007797 corrosion Effects 0.000 description 1
- 239000013310 covalent-organic framework Substances 0.000 description 1
- 238000005336 cracking Methods 0.000 description 1
- 238000002790 cross-validation Methods 0.000 description 1
- 230000001419 dependent effect Effects 0.000 description 1
- 238000009792 diffusion process Methods 0.000 description 1
- 239000006185 dispersion Substances 0.000 description 1
- 230000000694 effects Effects 0.000 description 1
- 230000005489 elastic deformation Effects 0.000 description 1
- 238000001493 electron microscopy Methods 0.000 description 1
- 238000004612 electronic band structure model Methods 0.000 description 1
- 238000002362 energy-dispersive X-ray chemical map Methods 0.000 description 1
- 230000001747 exhibiting effect Effects 0.000 description 1
- 150000002222 fluorine compounds Chemical class 0.000 description 1
- 238000007731 hot pressing Methods 0.000 description 1
- 230000006872 improvement Effects 0.000 description 1
- 230000001788 irregular Effects 0.000 description 1
- 230000000670 limiting effect Effects 0.000 description 1
- 229910052976 metal sulfide Inorganic materials 0.000 description 1
- 229910052759 nickel Inorganic materials 0.000 description 1
- 229910052757 nitrogen Inorganic materials 0.000 description 1
- 238000009828 non-uniform distribution Methods 0.000 description 1
- 229910052755 nonmetal Inorganic materials 0.000 description 1
- 230000006911 nucleation Effects 0.000 description 1
- 238000010899 nucleation Methods 0.000 description 1
- 229910052760 oxygen Inorganic materials 0.000 description 1
- 230000036961 partial effect Effects 0.000 description 1
- 230000000737 periodic effect Effects 0.000 description 1
- 239000004417 polycarbonate Substances 0.000 description 1
- 230000002028 premature Effects 0.000 description 1
- 239000013074 reference sample Substances 0.000 description 1
- 230000004044 response Effects 0.000 description 1
- 238000005070 sampling Methods 0.000 description 1
- 229910052710 silicon Inorganic materials 0.000 description 1
- HQVNEWCFYHHQES-UHFFFAOYSA-N silicon nitride Chemical compound N12[Si]34N5[Si]62N3[Si]51N64 HQVNEWCFYHHQES-UHFFFAOYSA-N 0.000 description 1
- 238000005245 sintering Methods 0.000 description 1
- 239000002002 slurry Substances 0.000 description 1
- 230000009897 systematic effect Effects 0.000 description 1
- 239000013077 target material Substances 0.000 description 1
- 238000007751 thermal spraying Methods 0.000 description 1
- 150000003568 thioethers Chemical class 0.000 description 1
- 238000012549 training Methods 0.000 description 1
- 238000005230 valence electron density Methods 0.000 description 1
- 238000010200 validation analysis Methods 0.000 description 1
- 238000003466 welding Methods 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C29/00—Alloys based on carbides, oxides, nitrides, borides, or silicides, e.g. cermets, or other metal compounds, e.g. oxynitrides, sulfides
- C22C29/02—Alloys based on carbides, oxides, nitrides, borides, or silicides, e.g. cermets, or other metal compounds, e.g. oxynitrides, sulfides based on carbides or carbonitrides
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B35/00—Shaped ceramic products characterised by their composition; Ceramics compositions; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products
- C04B35/515—Shaped ceramic products characterised by their composition; Ceramics compositions; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products based on non-oxide ceramics
- C04B35/56—Shaped ceramic products characterised by their composition; Ceramics compositions; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products based on non-oxide ceramics based on carbides or oxycarbides
- C04B35/5607—Shaped ceramic products characterised by their composition; Ceramics compositions; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products based on non-oxide ceramics based on carbides or oxycarbides based on refractory metal carbides
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01B—NON-METALLIC ELEMENTS; COMPOUNDS THEREOF; METALLOIDS OR COMPOUNDS THEREOF NOT COVERED BY SUBCLASS C01C
- C01B32/00—Carbon; Compounds thereof
- C01B32/90—Carbides
- C01B32/907—Oxycarbides; Sulfocarbides; Mixture of carbides
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B33—ADDITIVE MANUFACTURING TECHNOLOGY
- B33Y—ADDITIVE MANUFACTURING, i.e. MANUFACTURING OF THREE-DIMENSIONAL [3-D] OBJECTS BY ADDITIVE DEPOSITION, ADDITIVE AGGLOMERATION OR ADDITIVE LAYERING, e.g. BY 3-D PRINTING, STEREOLITHOGRAPHY OR SELECTIVE LASER SINTERING
- B33Y70/00—Materials specially adapted for additive manufacturing
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B33—ADDITIVE MANUFACTURING TECHNOLOGY
- B33Y—ADDITIVE MANUFACTURING, i.e. MANUFACTURING OF THREE-DIMENSIONAL [3-D] OBJECTS BY ADDITIVE DEPOSITION, ADDITIVE AGGLOMERATION OR ADDITIVE LAYERING, e.g. BY 3-D PRINTING, STEREOLITHOGRAPHY OR SELECTIVE LASER SINTERING
- B33Y80/00—Products made by additive manufacturing
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01B—NON-METALLIC ELEMENTS; COMPOUNDS THEREOF; METALLOIDS OR COMPOUNDS THEREOF NOT COVERED BY SUBCLASS C01C
- C01B32/00—Carbon; Compounds thereof
- C01B32/90—Carbides
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01B—NON-METALLIC ELEMENTS; COMPOUNDS THEREOF; METALLOIDS OR COMPOUNDS THEREOF NOT COVERED BY SUBCLASS C01C
- C01B32/00—Carbon; Compounds thereof
- C01B32/90—Carbides
- C01B32/914—Carbides of single elements
- C01B32/921—Titanium carbide
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01B—NON-METALLIC ELEMENTS; COMPOUNDS THEREOF; METALLOIDS OR COMPOUNDS THEREOF NOT COVERED BY SUBCLASS C01C
- C01B32/00—Carbon; Compounds thereof
- C01B32/90—Carbides
- C01B32/914—Carbides of single elements
- C01B32/949—Tungsten or molybdenum carbides
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B35/00—Shaped ceramic products characterised by their composition; Ceramics compositions; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products
- C04B35/515—Shaped ceramic products characterised by their composition; Ceramics compositions; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products based on non-oxide ceramics
- C04B35/56—Shaped ceramic products characterised by their composition; Ceramics compositions; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products based on non-oxide ceramics based on carbides or oxycarbides
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B35/00—Shaped ceramic products characterised by their composition; Ceramics compositions; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products
- C04B35/622—Forming processes; Processing powders of inorganic compounds preparatory to the manufacturing of ceramic products
- C04B35/64—Burning or sintering processes
- C04B35/645—Pressure sintering
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C29/00—Alloys based on carbides, oxides, nitrides, borides, or silicides, e.g. cermets, or other metal compounds, e.g. oxynitrides, sulfides
- C22C29/02—Alloys based on carbides, oxides, nitrides, borides, or silicides, e.g. cermets, or other metal compounds, e.g. oxynitrides, sulfides based on carbides or carbonitrides
- C22C29/06—Alloys based on carbides, oxides, nitrides, borides, or silicides, e.g. cermets, or other metal compounds, e.g. oxynitrides, sulfides based on carbides or carbonitrides based on carbides, but not containing other metal compounds
-
- G—PHYSICS
- G06—COMPUTING; CALCULATING OR COUNTING
- G06N—COMPUTING ARRANGEMENTS BASED ON SPECIFIC COMPUTATIONAL MODELS
- G06N20/00—Machine learning
- G06N20/10—Machine learning using kernel methods, e.g. support vector machines [SVM]
-
- G—PHYSICS
- G16—INFORMATION AND COMMUNICATION TECHNOLOGY [ICT] SPECIALLY ADAPTED FOR SPECIFIC APPLICATION FIELDS
- G16C—COMPUTATIONAL CHEMISTRY; CHEMOINFORMATICS; COMPUTATIONAL MATERIALS SCIENCE
- G16C60/00—Computational materials science, i.e. ICT specially adapted for investigating the physical or chemical properties of materials or phenomena associated with their design, synthesis, processing, characterisation or utilisation
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B22—CASTING; POWDER METALLURGY
- B22F—WORKING METALLIC POWDER; MANUFACTURE OF ARTICLES FROM METALLIC POWDER; MAKING METALLIC POWDER; APPARATUS OR DEVICES SPECIALLY ADAPTED FOR METALLIC POWDER
- B22F10/00—Additive manufacturing of workpieces or articles from metallic powder
- B22F10/20—Direct sintering or melting
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B22—CASTING; POWDER METALLURGY
- B22F—WORKING METALLIC POWDER; MANUFACTURE OF ARTICLES FROM METALLIC POWDER; MAKING METALLIC POWDER; APPARATUS OR DEVICES SPECIALLY ADAPTED FOR METALLIC POWDER
- B22F5/00—Manufacture of workpieces or articles from metallic powder characterised by the special shape of the product
- B22F2005/001—Cutting tools, earth boring or grinding tool other than table ware
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2002/00—Crystal-structural characteristics
- C01P2002/50—Solid solutions
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2002/00—Crystal-structural characteristics
- C01P2002/70—Crystal-structural characteristics defined by measured X-ray, neutron or electron diffraction data
- C01P2002/72—Crystal-structural characteristics defined by measured X-ray, neutron or electron diffraction data by d-values or two theta-values, e.g. as X-ray diagram
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2002/00—Crystal-structural characteristics
- C01P2002/70—Crystal-structural characteristics defined by measured X-ray, neutron or electron diffraction data
- C01P2002/77—Crystal-structural characteristics defined by measured X-ray, neutron or electron diffraction data by unit-cell parameters, atom positions or structure diagrams
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2002/00—Crystal-structural characteristics
- C01P2002/80—Crystal-structural characteristics defined by measured data other than those specified in group C01P2002/70
- C01P2002/85—Crystal-structural characteristics defined by measured data other than those specified in group C01P2002/70 by XPS, EDX or EDAX data
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2004/00—Particle morphology
- C01P2004/01—Particle morphology depicted by an image
- C01P2004/03—Particle morphology depicted by an image obtained by SEM
-
- C—CHEMISTRY; METALLURGY
- C01—INORGANIC CHEMISTRY
- C01P—INDEXING SCHEME RELATING TO STRUCTURAL AND PHYSICAL ASPECTS OF SOLID INORGANIC COMPOUNDS
- C01P2006/00—Physical properties of inorganic compounds
- C01P2006/90—Other properties not specified above
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/02—Composition of constituents of the starting material or of secondary phases of the final product
- C04B2235/30—Constituents and secondary phases not being of a fibrous nature
- C04B2235/40—Metallic constituents or additives not added as binding phase
- C04B2235/404—Refractory metals
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/02—Composition of constituents of the starting material or of secondary phases of the final product
- C04B2235/30—Constituents and secondary phases not being of a fibrous nature
- C04B2235/42—Non metallic elements added as constituents or additives, e.g. sulfur, phosphor, selenium or tellurium
- C04B2235/422—Carbon
- C04B2235/425—Graphite
-
- C—CHEMISTRY; METALLURGY
- C04—CEMENTS; CONCRETE; ARTIFICIAL STONE; CERAMICS; REFRACTORIES
- C04B—LIME, MAGNESIA; SLAG; CEMENTS; COMPOSITIONS THEREOF, e.g. MORTARS, CONCRETE OR LIKE BUILDING MATERIALS; ARTIFICIAL STONE; CERAMICS; REFRACTORIES; TREATMENT OF NATURAL STONE
- C04B2235/00—Aspects relating to ceramic starting mixtures or sintered ceramic products
- C04B2235/65—Aspects relating to heat treatments of ceramic bodies such as green ceramics or pre-sintered ceramics, e.g. burning, sintering or melting processes
- C04B2235/66—Specific sintering techniques, e.g. centrifugal sintering
- C04B2235/666—Applying a current during sintering, e.g. plasma sintering [SPS], electrical resistance heating or pulse electric current sintering [PECS]
-
- E—FIXED CONSTRUCTIONS
- E02—HYDRAULIC ENGINEERING; FOUNDATIONS; SOIL SHIFTING
- E02F—DREDGING; SOIL-SHIFTING
- E02F9/00—Component parts of dredgers or soil-shifting machines, not restricted to one of the kinds covered by groups E02F3/00 - E02F7/00
- E02F9/28—Small metalwork for digging elements, e.g. teeth scraper bits
- E02F9/2808—Teeth
- E02F9/285—Teeth characterised by the material used
-
- E—FIXED CONSTRUCTIONS
- E21—EARTH OR ROCK DRILLING; MINING
- E21B—EARTH OR ROCK DRILLING; OBTAINING OIL, GAS, WATER, SOLUBLE OR MELTABLE MATERIALS OR A SLURRY OF MINERALS FROM WELLS
- E21B10/00—Drill bits
- E21B10/46—Drill bits characterised by wear resisting parts, e.g. diamond inserts
-
- E—FIXED CONSTRUCTIONS
- E21—EARTH OR ROCK DRILLING; MINING
- E21C—MINING OR QUARRYING
- E21C35/00—Details of, or accessories for, machines for slitting or completely freeing the mineral from the seam, not provided for in groups E21C25/00 - E21C33/00, E21C37/00 or E21C39/00
- E21C35/18—Mining picks; Holders therefor
- E21C35/183—Mining picks; Holders therefor with inserts or layers of wear-resisting material
- E21C35/1835—Chemical composition or specific material
-
- G—PHYSICS
- G06—COMPUTING; CALCULATING OR COUNTING
- G06N—COMPUTING ARRANGEMENTS BASED ON SPECIFIC COMPUTATIONAL MODELS
- G06N20/00—Machine learning
-
- G—PHYSICS
- G16—INFORMATION AND COMMUNICATION TECHNOLOGY [ICT] SPECIALLY ADAPTED FOR SPECIFIC APPLICATION FIELDS
- G16C—COMPUTATIONAL CHEMISTRY; CHEMOINFORMATICS; COMPUTATIONAL MATERIALS SCIENCE
- G16C10/00—Computational theoretical chemistry, i.e. ICT specially adapted for theoretical aspects of quantum chemistry, molecular mechanics, molecular dynamics or the like
-
- G—PHYSICS
- G16—INFORMATION AND COMMUNICATION TECHNOLOGY [ICT] SPECIALLY ADAPTED FOR SPECIFIC APPLICATION FIELDS
- G16C—COMPUTATIONAL CHEMISTRY; CHEMOINFORMATICS; COMPUTATIONAL MATERIALS SCIENCE
- G16C20/00—Chemoinformatics, i.e. ICT specially adapted for the handling of physicochemical or structural data of chemical particles, elements, compounds or mixtures
- G16C20/30—Prediction of properties of chemical compounds, compositions or mixtures
-
- G—PHYSICS
- G16—INFORMATION AND COMMUNICATION TECHNOLOGY [ICT] SPECIALLY ADAPTED FOR SPECIFIC APPLICATION FIELDS
- G16C—COMPUTATIONAL CHEMISTRY; CHEMOINFORMATICS; COMPUTATIONAL MATERIALS SCIENCE
- G16C20/00—Chemoinformatics, i.e. ICT specially adapted for the handling of physicochemical or structural data of chemical particles, elements, compounds or mixtures
- G16C20/70—Machine learning, data mining or chemometrics
Definitions
- the present invention relates to complex materials .
- the present invention relates particularly, although by no means exclusively, to complex carbides for use in mining and mineral processing applications.
- the present invention also relates to mining and mineral processing equipment that is subject to wear that is formed from or includes the above-described hard complex carbides.
- the present invention also relates to complex nitrides, complex borides, complex oxides, complex carbonitrides and other combinations of carbides, borides, oxides and nitrides for use in mining and mineral processing applications.
- the present invention also relates to mining and mineral processing equipment that is subject to wear that is formed from or includes the above-described complex nitrides, complex borides, complex oxides, complex carbonitrides and other combinations.
- the equipment includes, for example, slurry pumps and pipelines, mill liners, crushers, transfer chutes and ground-engaging tools.
- Metal carbides such as TiC, VC, and WC, are known for their superior mechanical and thermal properties. Examples include tungsten carbides or titanium carbides for material surface protection against wear and zirconium carbides and tantalum carbides for high-temperature protection.
- TiC and VC are often used to make cutting tools (turning, milling, drilling), wear-resistant coatings, and strengthen composites and alloys as a reinforcement.
- tungsten monocarbide is often used as a reinforcement in wear- resistant coatings and alloys, such as in cutting tools (turning, milling, drilling) or as a reinforcing phase in protective coatings to protect metallic substrates from wear, e.g., metal-matrix coatings containing WC particles made by thermal spraying, spray-fuse and welding processes.
- Such composite coatings provide an effective, economic and flexible technique for surface protection, which are widely used to protect machinery and facilities in various industrial operations.
- WC particles are not homogeneously distributed in metal-matrix hard facing overlays, which negatively influences the overall performance, such as wear resistance, of the overlays .
- High-entropy ceramics such as high-entropy carbides, another form of metal carbides, are widely used as reinforcing phases for hard coatings, diffusion barriers, and thermal protective coatings in various technological fields because of their excellent mechanical properties, high thermal stability, and corrosion resistance.
- Many industrial ceramics, such as the rock-salt-structured TiC have high hardness and Young's modulus, but their fracture toughness is generally low, leading to the risk of premature failure and, thus, limiting their applications in harsh environments or in specific industrial operating conditions.
- there are no reliable and effective methods pinpointing the appropriate constituents for the wished-for mechanical properties.
- the development of high-entropy ceramics is largely based on the costly trial-and-error approach because of an insufficient understanding of the key factors that govern the mechanical properties of the materials, especially the roles that different types of bonds have in determining the mechanical behaviour.
- the invention is concerned with providing an alternative to known metal carbides.
- the required properties include, for example, density matching, mechanical properties, and wear performance .
- the complex carbides of the invention are an alternative to known metal carbides used for the applications .
- the work carried out by the applicant also indicates that the modelling approach is not confined to complex carbides and also applies to complex nitrides, complex borides, complex oxides, complex carbonitrides and other combinations.
- complex carbide is understood herein to mean a carbide that includes at least two metals as part of the complex carbide.
- complex nitrides complex borides, complex oxides, complex carbonitrides, etc
- complex carbide complex carbide set out above.
- the specific complex carbides of the invention have been identified through modelling work are complex carbides that have favourable physical properties over typical carbides used in industry, particularly in their hardness, Young’s modulus, and toughness.
- the modelling work has further identified that these complex carbides are feasible to fabricate and stable in their fabricated form.
- An example of a complex carbide is a carbide of the formula (M,X)C, where "M” and “X" are metals.
- the invention includes a method of selecting a complex carbide for an end-use application in mining and mineral processing applications, comprising: modelling properties of complex carbides, determining the required properties for the end- use application, and selecting a modelled complex carbide that meets the required properties for the end-use application.
- the invention also includes a complex carbide for an end-use application in mining and mineral processing applications that are subject to severe wear.
- the complex carbide may be a carbide that includes a main metal, i.e. a metal that is a substantial proportion by weight of the metals in the carbide, and at least one additional metal, with the additional metal being a transition metal.
- the complex carbide may be any suitable (M,X)C carbide, where "M” is the main metal and “X” is the transition metal.
- the complex carbide may be a
- the complex carbide may be any suitable (M 1 - x X x ) 7 C 3 carbide, where the lower case x is 0 ⁇ x ⁇ 1 , ; where "M” is the main metal and "X” is the transition metal, noting that (M 1-x X x ) 7 C 3 may also be described as (M 7-X X X )C 3 ) .
- the complex carbide may include further metal (s) , i.e. (M,X,Y) 7 C 3 , where "Y" is a further metal.
- the complex carbide may be high-entropy ceramics (HECs) of any one of the formula M 1 M 2 M 3 C, M 1 M 2 M 3 M 4 C, M 1 M 2 M 3 M 4 M 5 C, or M 1 M 2 M 3 M 4 M 5 M 6 C , where each M x element is unique in the complex carbide and selected from any one of Ti, V, Cr, Zr, Nb, Mo, Hf, Ta, and W.
- HECs high-entropy ceramics
- the main metal may be any suitable metal.
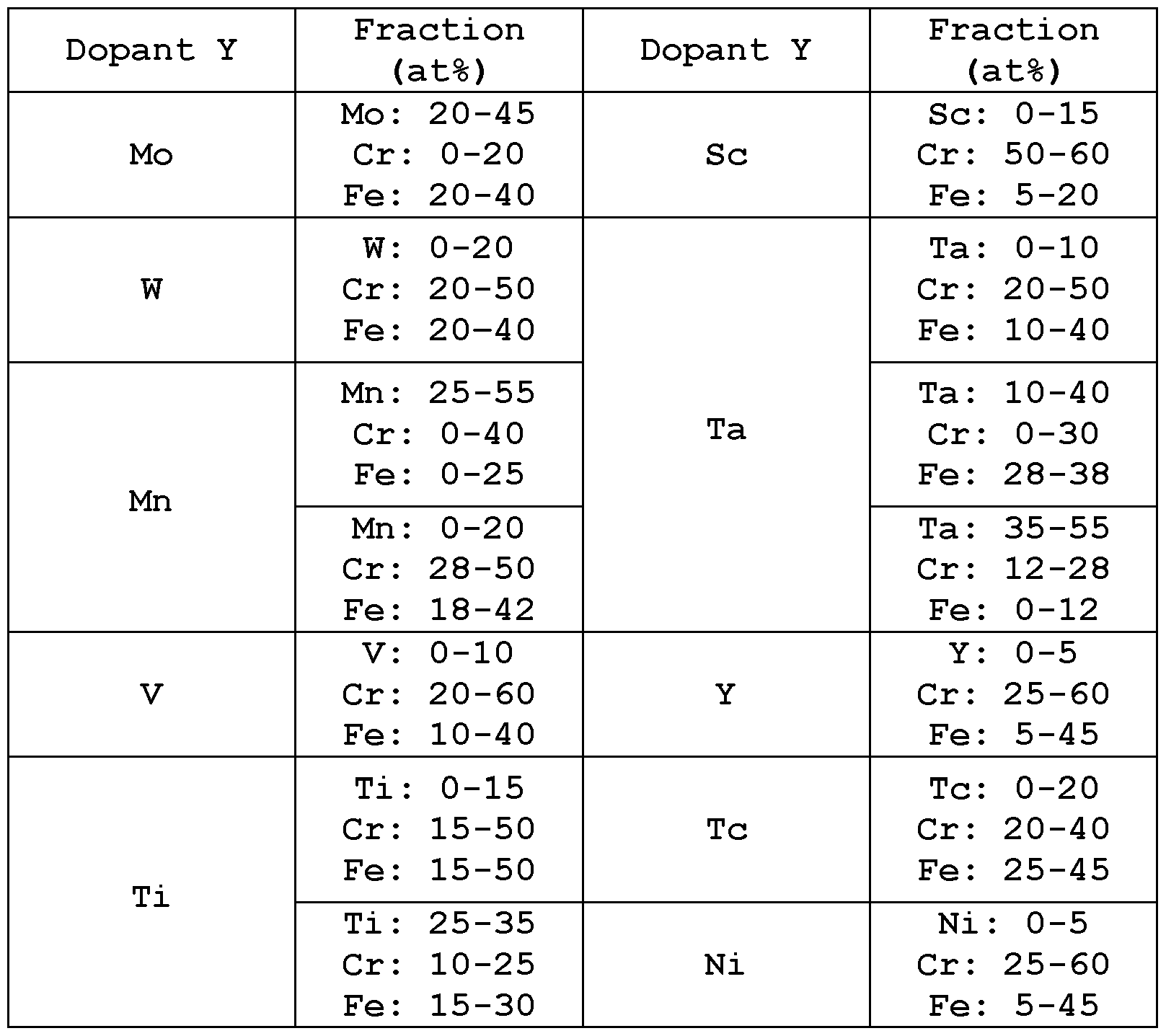
- the main metal may be any one or more than one of Al, Co, Cr, Cu, Hf, Sc, Ti, W, Zr, Fe, Mn, Mo, Nb, Ta, V, Zn, and Y.
- the transition metal may be any suitable metal.
- the transition metal may be a 3d-6d transition metal.
- the further metal(s) may be any suitable metal.
- the further metal(s) may be a 3d-6d transition metal.
- the further metal(s) may be any one or more than one of Al, Co, Cr, Cu, Hf, Sc, Ti, W, Zr, Fe, Mn, Mo, Nb, Ta, V, Zn, and Y.
- the transition metal may a 3d or a 4d transition metal.
- the transition metal may another 3d-6d transition metal.
- the main metal may be at least 15%, typically at least 20% by weight of the total weight of the complex carbide.
- High-entropy ceramics of any one of the formula M 1 M 2 M 3 C, M 1 M 2 M 3 M 4 C, M 1 M 2 M 3 M 4 M 5 C, or M 1 M 2 M 3 M 4 M 5 M 6 C, where each M x element is unique in the complex carbide and selected from any one of Ti, V, Cr, Zr, Nb, Mo, Hf, Ta, and W.
- embodiments of the invention relate to one or more of:
- HECs High-entropy ceramics of any one of the formula M 1 M 2 M 3 C, M 1 M 2 M 3 M 4 C, M 1 M 2 M 3 M 4 M 5 C, or M 1 M 2 M 3 M 4 M 5 M 6 C, where each M x element is unique in the complex carbide and selected from any one of Ti, V, Cr, Zr, Nb, Mo, Hf, Ta, and W.
- the M 1-x X x C (0 ⁇ x ⁇ 1) complex carbides may include any one or more of the following
- M and X are selected from combinations of Al, Co, Cr, Cu, Hf, Sc, Ti, W, Zr, Fe, Mn, Mo, Nb, Ta, V, Zn, and Y including but not limited to the following carbides, typically having negative formation energy and cohesive energy and improved properties (Young's modulus "E”, Hardness "H”, and Pugh's ratio) compared to the corresponding mono- metal carbides:
- Hf, Ti, Zr, Nb, Ta, and V metals in general improve the properties, when compared to the properties of the corresponding mono-carbides. Although most of the carbides have Pugh's ratio lower than 1.75, they are close to a critical value (i.e. in range of 1.4 - 1.7).
- the category 3 complex (Fe,Cr,Y) 7 C 3 carbides may be any one or more than one of the following carbides: (Fe,Cr,Mo) 7 C 3 ,
- the (Fe,Cr,Y) 7 C 3 complex carbides may have a hardness in a range of 9.0 - 15.5 GPa, typically 10.0
- the (Fe,Cr,Y) 7 C 3 complex carbides may have a Young's Modulus in a range of 290 - 410 GPa, typically 310 - 400 GPa.
- the (Fe,Cr,Y) 7 C 3 complex carbides may have a toughness (B/G) in a range of 1.70 - 2.40, typically 1.50 - 1.90.
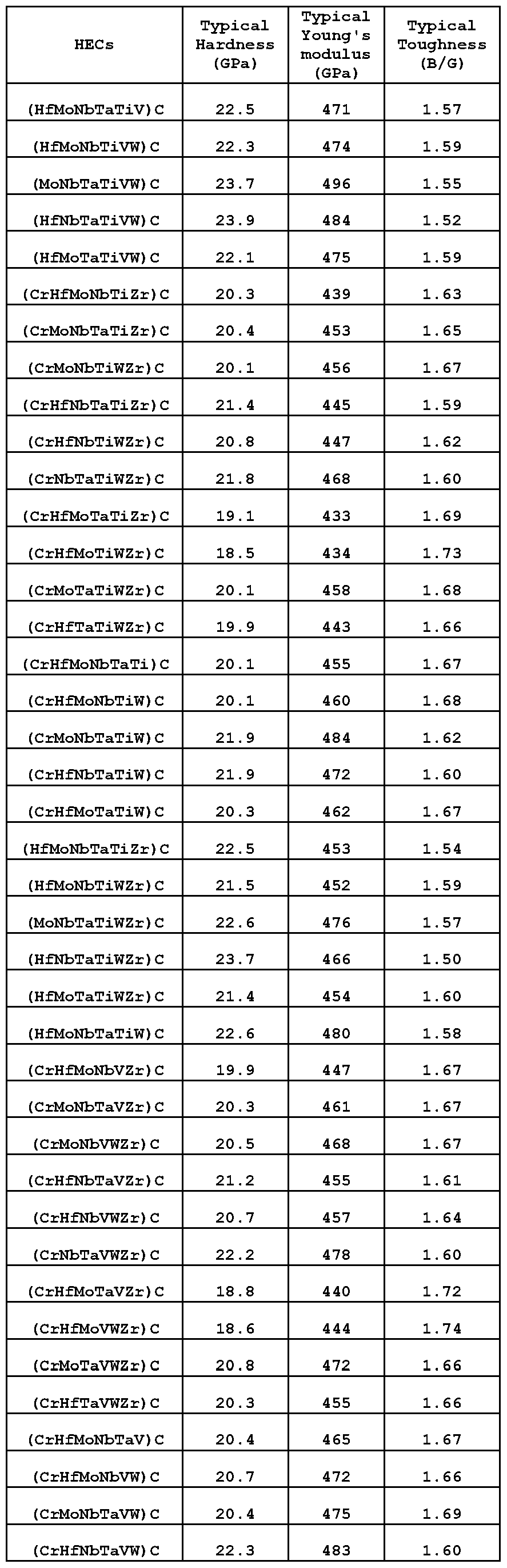
- the category 5 high-entropy complex carbides may be any one or more than one of the following carbides of general formula M 1 M 2 M 3 C, M 1 M 2 M 3 M 4 C, M 1 M 2 M 3 M 4 M 5 C, or M 1 M 2 M 3 M 4 M 5 M 6 C, with 4-6 metal elements, each in a uniform distribution, and with these high-entropy complex carbides having been modelled and found to have the following typical improved properties:
- the category 5 high-entropy complex carbides may have a hardness in a range of 15.0 - 27.0 GPa, typically 15.5 - 26.0 GPa.
- the category 5 high-entropy complex carbides may have a Young's Modulus in a range of 420 - 515 GPa, typically 435 - 500 GPa.
- the category 5 high-entropy complex carbides may have a toughness (B/G) in a range of 1.40 - 1.95, typically 1.50 - 1.90.
- the category 5 high entropy complex carbides may be any one or more than one of the following carbides of general formula M 1 M 2 M 3 C, M 1 M 2 M 3 M 4 C, and M 1 M 2 M 3 M 4 M 5 C, noting that there is some overlap with the carbides mentioned in the previous table:
- Complex carbides can be produced separately from a metal product - e.g. producing complex carbides and then laser cladding to reinforce the metal product.
- Complex carbides can be used in a wide range of products for mining and mineral processing industries .
- Production methods for forming complex carbides for mining and mineral processing applications may include the following options: ⁇ Small scale, including:
- the invention also provides mining and mineral processing equipment that is subject to wear that is formed from or includes the above-described complex carbide dispersed in or formed as a layer on a metal or a metal alloy.
- the equipment may be in the form of a casting of the complex carbide and the metal or the metal alloy.
- the equipment may be in the form of a layer of the complex carbide on a substrate of the metal or the metal alloy.
- the layer may be formed, for example, as a hard- facing on the substrate or a cladding on the substrate.
- the equipment may be in the form of sintered particles of the complex carbide and particles of the metal or the metal alloy.
- the equipment may be additively manufactured from the complex carbide and the metal or the metal alloy.
- Embodiments of the invention take advantage of a machine-learning accelerated strategy involving first-principle calculations based on density function theory (DFT) to design complex metal carbides with the desired mechanical properties.
- DFT density function theory
- these embodiments take advantage of a machine-learning tool developed by the inventors that describes the correlations between properties, which show good prediction accuracy, as verified by computational and experimental data.
- the invention also includes a complex nitride for an end-use application in mining and mineral processing applications that are subject to severe wear.
- the invention also includes a complex boride for an end-use application in mining and mineral processing applications that are subject to severe wear.
- the invention also includes a complex oxide for an end-use application in mining and mineral processing applications that are subject to severe wear.
- the invention also includes a complex carbonitride and other combinations of carbides, borides, oxides and nitrides for an end-use application in mining and mineral processing applications that are subject to severe wear.
- the model mentioned above is based on density functional theory (DFT) calculations using commercially available software VASP.
- DFT density functional theory
- the model includes the following main stages/steps .
- Stage 1 Determines geometry relaxation of structure, formation and stability of the complex carbide.
- Stage 2 (Step 3): Determines mechanical properties of the complex carbide.
- Stage 3 (Steps 4-5): Building database and identifying suitable complex carbides using the identified criterion.
- the model includes the following steps.
- Step 1 Determining Geometry relaxation of simulated atomic structures of selected carbide compositions.
- An initial atomic model was built for the selected carbide composition. This initial model typically has an unrelaxed structure, i.e. having inner stress in the initial model and the lattice size and positions of atoms were away from the equilibrium position.
- Geometry relaxation for the selected carbide composition was determined by releasing the inner stress step by step by moving atoms and modifying the lattice size (i.e., atom position), until the inner stress was smaller than the preset convergence criteria (e.g. 0.01 eV/ ⁇ ) and the change of energy of the system between each step was smaller than the preset convergence criteria (e.g. 10e -4 eV).
- the preset convergence criteria e.g. 0.01 eV/ ⁇
- Formation energy ( EF) of A m B n This energy is needed to determine if the material can be made without barrier).
- EAmBn ( EAmBn — mEA — nEB) /(m + n)
- EAmBn is the energy of A m B n per chemical formula
- EA and EB are energies per atom of simple substances A and B
- EAmBn — mE A ' — nE B ' EAmBn — mE A ' — nE B ' ) /(m + n)
- EAmBn is the energy of A m B n per chemical formula
- E A ' andE B ' are energies of A and B atoms .
- the database may be screened with the following criteria:
- Negative cohesive energy Carbides with negative cohesive energies are chemically stable.
- Criterion 3 Carbides with relatively better mechanical properties in the property distribution maps and having negative formation and cohesive energies are screened.
- Figures 1-4 are microstructures and element maps of Mo-complex carbide samples
- Figure 6 is microhardness values of the fabricated WC and Mo-complex carbides
- Figure 7 is friction coefficients of WC and Mo- complex carbides sliding at applied load of 20 N for 3600s;
- Figure 8 is wear track images of WC and Mo- complex tungsten carbides
- Figure 9 shows SEM images of worn surfaces of the WC and Mo-complex tungsten carbides
- Figure 10 shows typical Young's moduli and densities of Ti-, Cr- and Mo-complex carbides versus the metal concentrations
- Figure 11 presents E/ ⁇ ratios of WC complex carbides with metal concentrations
- Figure 12 presents typical HI and p values of the metal-complex carbides
- Figure 13 shows Pugh's ratios of metal-complex carbides with metal concentrations
- Figure 14 shows Poisson's ratios of metal- complex carbides with metal concentrations
- Figure 15 is an XRD comparison between 5 samples of TiC and VC monocarbides
- Figure 16 is BSI and EDX maps of Tio.25Vo.75C.
- Figure 17 is a series of plots of density of states (DOS) and crystal orbital Hamilton population (COHP);
- Figure 18 is Supercell structures of WC and metal complex tungsten carbides based on hexagonal WC;
- Figure 19 is a graph of toughness v hardness for high-entropy complex carbides (HECs).
- Figure 20 is a schematic illustration of the design strategy for selecting HECs
- Figure 21 is a series of graphs that illustrate bond properties and mechanical properties of rock- salt mono carbides, nitrides, and carbonitrides;
- Figure 22 is a series of graphs that illustrate the influence of alloying on atomic bond strengths and mechanical properties
- Figure 23 is a series of graphs that illustrate scaling mechanical properties from bond properties.
- Figure 24 is a series of graphs that illustrate predicting mechanical properties using the machine- learning models.
- High-entropy ceramics of any one of the formula M 1 M 2 M 3 C, M 1 M 2 M 3 M 4 C, M 1 M 2 M 3 M 4 M 5 C, or M 1 M 2 M 3 M 4 M 5 M 6 C, where each Mx element is unique in the complex carbide and selected from any one of Ti, V, Cr, Zr, Nb, Mo, Hf, Ta, and W.
- Section 1 relates to complex tungsten carbides (W ,X) carbides.
- Section 2 relates to complex (Ti,V) carbides.
- Section 3 relates to high-entropy ceramics (HECs).
- the complex carbides were designed and studied through first-principle calculations based on the density function theory (DFT).
- DFT density function theory
- WC can be tailored by element modifiers resulting in stable complex (W,X)C carbides that have desired mechanical properties with modifiable density.
- the designed (W,X)C carbides were ranked and selected for fabrication using an arc melting technique, and their structure, hardness and wear behaviour were investigated using SEM, EDS, XRD, microhardness and wear testing instruments, respectively .
- tungsten monocarbide For tungsten monocarbide (WC), the experimentally determined crystal structure was used as a base configuration for optimization and element modifiers to form complex carbides.
- Metal complex tungsten carbides with concentrations of 25 at.%, 50 at.%, and 75 at.% metals were modelled using 2x2x1 supercells based on W 4 C 4 configuration containing 4 tungsten atoms and 4 carbon atoms.
- the valence electron configurations for W, Sc, Ti, V, Cr, Mn, Fe, Mo and C correspond to :3P 6 3d 2 4s 2 , 3P 6 3d 3 4s 2 , 3P 6 3d 5 4s 1 , 3P 6 4d 2 5s 2 , 3P 6 4d 4 5s 1 , 4P 6 4d 5 4s 1 , 5d 4 6s 2 and 2s 2 2p 2 .
- the plane wave cut-off energy was set as 600 eV.
- a convergence criterion of 10-6 eV/atom was used for the electronic self-consistency loop, k-point meshes for the Brillouin zone sampling were constructed through the Gamma scheme.
- the 11 x 11 x 11 and 9 x 9 x 11 k-points grids were used for WC and metal complex tungsten carbides, respectively.
- the unit cell of the carbides at the zero pressure was optimized by full relaxation with respect to the volume, shape, and internal atomic positions until the atomic forces were less than 10-2 eV/A.
- the crystal structures of WC and complex carbides were represented using MS visualizer and the VESTA software .
- the strain-stress relationship method was used to determine elastic constants from the optimized unit cells under zero pressure, as implemented in the VASP.
- the elastic constants were defined as the first derivatives of the stresses with respect to the strain tensor.
- the elastic tensor was determined by performing six finite distortions of the lattice and deriving the elastic constants from the strain-stress relationship.
- the elastic tensor was calculated for rigid ions and an allowance was made for relaxation of the ions. Ionic contributions were determined by inverting the ionic Hessian matrix and multiplying with the internal strain tensor.
- Final elastic constants include both the contributions from distortions with rigid ions and the contributions from the ionic relaxations.
- the WC bulk carbide samples were fabricated with WC powder.
- the complex (W,Mo)C bulk samples were fabricated with pure metals (W and Mo) powders and graphite powder.
- Microstructural features of the specimens and the compositions of various phases were examined by scanning electron microscopy (SEM, EVO-MA 10) equipped with an X-ray energy dispersive spectroscopy (EDS) system.
- SEM scanning electron microscopy
- EDS X-ray energy dispersive spectroscopy
- the range of glancing angles is 20-100°.
- the test voltage and current were 40 kV and 30 mA, respectively.
- the hardness of fabricated WC and Mo-complex carbides was measured using a Microhardness (FISHER) tester at load of 500 mN for 60 s. For each kind of carbide, three to five positions were tested at the area without graphite and averaged.
- FISHER Microhardness
- the density of WC can be markedly changed after modifying with other metals to form complex carbides, which is vital for its application in metal-matrix hard facing overlays and composites (such as Co-matrix, Ni alloy matrix, iron-matrix, and , etc.) ⁇
- the density of complex tungsten carbides e.g., W 1 Ti 3 C 4 , could be as low as 7.22 g/cm 3 .
- Total Free energy of the complex carbides can be obtained directly by geometry optimization calculation. However, these free energies cannot be directly used to compare the stability of each carbide.
- Mo-complex carbides possessed the lowest formation energy, indicating that this series of carbides are easier to be formed with higher stability.
- Ef is the formation energy of a carbide
- x represent the number of metal atoms in a cell of the carbide
- E tot is the total energy of a cell of the carbide at the optimized geometries
- E w solid E M solid and E graphite solid represent the energy of single atom W, complex-metals and C in the solid state, respectively.
- the complex carbides with negative formation energies are more stable than the mixture of single metals and graphite (C) and they would be easy to be formed using single pure metals and graphite(C).
- Ti-, V-, Cr- and Mo-complex carbides are stable.
- W 1 Mo 3 C 4 complex carbide is the most stable one among the calculated metal-complex tungsten carbides.
- Mo is a strong candidate for an alloying element to modify WC carbide.
- experimental work on Mo-complex tungsten carbides reported.
- Typical Mo-complex tungsten carbides were prepared by melting a mixture of pure metal and graphite powders using an arc melting furnace.
- WC bulk sample was also fabricated by melting WC powder using the same arc melting procedure.
- Fig. 6 shows microhardness values of the fabricated WC and Mo-complex carbides.
- the WC carbide ( ⁇ 34 GPa) was harder than the Mo-complex carbides.
- all the Mo-complex carbides had their microhardness values higher than 30 GPa, close to that of WC, consistent with the theoretical prediction.
- Mo-complex tungsten carbides are promising to be used as substitutes for WC in metal- matrix hard facing overlays and composites with improved performance.
- pin-on-disk wear tests were performed under normal loads of 20 N for 30 minutes .
- Fig. 8 show wear track images of the WC and Mo-complex tungsten carbides, which are rather shallow, and wear rates of the carbides are obviously small. This makes it difficult to measure the volume loss of the carbides from their wear track dimensions.
- the wear resistances of the carbides were evaluated using an indirect approach by measuring the volume loss of counter-body (Si 3 N 4 ball) based on the geometry of the worn area on the Si 3 N 4 ball (see Fig.8 (e)).
- the fabricated W 3 Mo 1 C 4 carbide had the highest wear resistance.
- the wear resistances of the carbides are thus ranked as W 3 Mo 1 C 4 > WC > W 2 Mo 2 C 4 > W 1 Mo 3 C 4 .
- Fig. 9 shows the SEM images of worn surfaces of the WC and Mo-complex tungsten carbides.
- Elastic constants and elastic anisotropy are fundamental properties in understanding mechanical properties ranging from stress-strain behaviour, dislocation motion, crack nucleation, crack propagation, etc.
- the elastic constants of a crystal system need to satisfy the generalized mechanical stability criterion.
- the criterion is represented by the following conditions : C 44 >0,C 11 -IC 12 I>0,C 33 (C 11 +C 12 )-2C 13 2 > 0
- C44 is related to the resistance to shear deformation
- C11- IC12I reflects the resistance to expansion along the spindle axis during the contraction of the other directions perpendicular to the spindle axis
- C33(C11 + C12)-2C132 represents the resistance to the volumetric deformation of the crystal.
- the elastic constants are of importance to the understanding of the crystals' reversible elastic deformation or the response to corresponding stresses .
- C11, C22 and C33 are the measure of resistance to linear compression along x, y and z directions, respectively.
- the calculated C11 of the metal-complex tungsten carbides were very large except those of Sc- and Ti-complex ones.
- the results suggest that there is a very high linear compression resistance along x direction.
- the corresponding values were higher than 600 GPa (C33 as higher as 850 GPa), indicating a very high linear compression resistance along x, y and z directions .
- Fig. 10 show typical Young's moduli and densities of Ti-, Cr- and Mo-complex carbides versus the metal concentrations.
- Fig.11 presents E/ ⁇ ratios of WC carbides before and after metal modifying. As shown, WC has its ratio of E/ ⁇ equal to 45 GPa/(g/cm 3 ), which was increased by modifying with Cr, Mn and Mo.
- metal complex tungsten carbides would have a good potential for aerospace applications.
- the calculated moduli of WC are in good agreement with available reported data.
- B values for most of the complex carbides are higher than 300 GPa, especially, the Cr- and Mo-complex carbides have their B higher than 340 GPa, very close to that of WC, which indicates the strong atomic bonding strength.
- G values of the Cr- and Mo-complex carbides are higher than 250 GPa, also very close to the value of WC, indicating that these carbides possess high resistance to reversible shear deformation.
- the calculated E values also show that the Cr- and Mo-complex tungsten carbides can have their mechanical strength close to that of the mono- WC.
- the element modifying provided various combinations of E and p, which help select appropriate metal modifiers for specific applications .
- Ti-, V- and Zr-complex carbides had relatively low densities and their Young's moduli were also low.
- W 1 Cr 3 C 4 and W 1 Mo 3 C 4 carbide possess a relative lower density (as low as 9.04 g/cm 3 ) but their Young's modulus of (higher than 610 GPa) are close or comparable to that of WC.
- Hardness is the ability of a material to resist deformation under a mechanical load.
- the shear modulus is sensitive to the nonuniform distribution of valence electron density corresponding to the kind of directional bonds which in turn act as barriers to dislocation movement.
- Bulk modulus depends on the spatially averaged electron density within the three dimensional densely packed networks without respect to the type of bonds formed, i.e., metallic, ionic or covalent one. Therefore, the shear modulus is a better qualitative predictor of hardness than the bulk modulus, that is, the shear modulus is more pertinent to hardness than the bulk modulus.
- G/B2G Another parameter, (G/B)2G, related to both elasticity and plasticity is also often used to predict the hardness of materials, known as the Hardness indicating factor (HI).
- Chen et al proposed another empirical formula as shown by the following equation, which is often adopted for calculating the Vickers hardness (HV) of materials,
- This equation is used in the present work to calculate hardness of metal-complex tungsten carbides. Typical HI and p values of the metal- complex carbides are shown in Fig.12.
- the calculated hardness of metal-complex tungsten carbides is usually lower than that of WC but this is dependent on the type of complex metals. As shown in Fig.12, hardness values of Cr-, Mn- and Mo-complex carbides only slightly decreased while their densities were considerably lower. Thus, the carbides modified with these metals were markedly lighter while retaining comparable hardness would be valuable reinforcements for fabricating different metal-matrix composite materials and coatings. Compared to these metal modifiers, Sc- and Ti- complex carbides have very low densities but their HI values also decrease sharply, making them less valuable in terms of modification of tungsten carbides for developing lighter reinforcements.
- the ductility or brittleness of materials is crucial to the resistance of carbides to cracking under impact force or stress with larger fluctuations in magnitude.
- materials can be classified based on their ductility or brittleness for practical applications in resisting material failure.
- Ductility is of technologically importance.
- Pugh's ratio (B/G) is a parameter often used to estimate the ductility of a material. If the value of Pugh's ratio of a material is higher than 1.75, the material is considered to be ductile; otherwise, it is brittle.
- Fig. 14 shows Poisson's ratios of metal-complex carbides changed with the modifier's concentration.
- v value also provides the information on the atomic bonding nature. Ionic solids have their values of v around 0.25, while covalent materials' value is about 0.10. In the present work, the v values of WC and Cr-, Mn- and Mo-complex carbides were in the range from 0.21 to 0.23, suggesting that these carbides may have mixtures of ionic and covalent bonds but dominated by the former. Besides, a material is considered to be a central-force solid if the value of v lies in the range 0.25-0.5. Most of calculated Poisson's ratios of Sc- and Ti-complex carbides fall into this category, suggesting that the interatomic forces of these carbides are mainly the type of central force.
- Cauchy pressure is also used as an indicator to judge the ductile/brittle behaviour of materials, which may describe the angular character of atomic bonding in solids. According to Pettifor's rule, a material having more metallic bonds is more ductile if it has a larger positive Cauchy pressure, or it has angular bonds and thus brittle if the material has a negative value of Cauchy pressure.
- Cauchy pressure is defined as (C13-C44) and (C12-C66).
- the Cauchy pressures is defined as C23-C44 for the (100) plane, C13-C55 for the (010) plane, and C12-C66 for the (001) plane.
- the calculated Cauchy pressures for Sc- and Ti- complex carbides are positive, meaning that Sc- and Ti-complex carbides are ductile. Other complex carbides had negative Cauchy pressures i.e. they are brittle. The result of the Cauchy pressure is consistent with those of the Pugh's ratio and Poisson's ratio analyses.
- the Cauchy pressure can also provide the information on the type atomic bonding in solids. Positive Cauchy pressure usually corresponds to metallic bonding, while negative Cauchy pressure characterizes the directional covalent bonding.
- the calculated Cauchy pressure of metal-complex carbides suggests the presence of covalent and/or ionic bonding in metal- complex carbides except for Sc- and Ti-complex carbides .
- TDOS total density of states
- PDOS partial density of states
- ELF electron local function
- DOS helps looking into the electron energy and describe the dispersion of a given electronic band over the space of energy.
- the positive values of DOS at the Fermi level (EF) indicates the metallicity and electronic conductivity of a crystal structure.
- the bonding in the metal-complex carbides are mixture of metallic, covalent, and ionic bonds.
- the sharp decreases in Young's moduli and hardness of WC by Sc-modification should be ascribed to the increase in metallic bonding component and decrease in the generally stronger covalent bonding or ionic bonding components .
- the stability, atomic bonding and electronic states of the complex carbide were analyzed in order to understand the mechanisms responsible for the improvement in properties of the complex carbide for further optimization.
- the synthesized samples were characterized with X-ray diffraction analysis, scanning electron microscopy, and electron dispersive x-ray spectroscopy . Hardness and wear resistance of the samples were evaluated and compared to those of TiC and VC monocarbides .
- TiC was modified by V substitution to form complex (Ti,V)C.
- TiC has a good combination of mechanical and thermal properties, which has found many applications such as wear-resistant coatings and high-temperature vessels.
- a closed neighbour of Ti in the periodic table is vanadium, which has its atomic structures similar to that of Ti.
- Previous work on the elastic modulus of TiC:VC alloyed carbide was reported in the literature, but the mechanism for the changes in properties needs to be better understood.
- metal carbides are used as the reinforcement to make wear-resistant metal-matrix composites and coatings
- wear behaviour of metal carbides themselves has not been well studied.
- the information on the wear behaviour of bulk carbides is also valuable for understanding their role as an reinforcement and for properly selecting effective carbides wear-resistant composites and protective coatings .
- Synthesizing metal carbides is commonly achieved by reactions of compressed fine powders of different components at high temperatures and pressures.
- the most commonly used processes are the spark plasma sintering (SPS) and hot pressing, both of which are straightforward.
- SPS spark plasma sintering
- Another method is the arc melting process, which is less time consuming but less tried, is used for synthesizing refractory carbides.
- the arc melting method is usually used to make metals or alloy, it can be used to fabricate metal carbides, since the arc can reach 3000 - 4000 degrees which is sufficient for synthesis of carbides .
- the fabricated complex carbide samples were characterized by X-ray diffraction (XRD) at a scanning rate of 4 degree/min in the 2-theta range of 20-100° .
- XRD X-ray diffraction
- the XRD analysis was carried out using a Bruker D8 Discovery diffraction system equipped with LynxEye 1-dementional detector and a Copper radiation source .
- the wear behavior of the complex carbides was evaluated by sliding wear test on a Rtec tribometer with a rotary module. A normal load of 30N load was applied on a Si 3 N 4 ball of 6 mm in diameter, which was pressed onto the sample under testing. The ball moved along a circle track at a speed of 200 rpm. The wear track was analyzed with a Zygo ZeGage 3D optical profilometer to measure the volume loss. The morphology and detailed surface information was characterized with Zeiss EVO M10 Secondary Electron Microscopy (SEM). The information about the elemental distribution on the worn surface was collected by the Energy Dispersive X-ray analysis (EDX).
- EDX Energy Dispersive X-ray analysis
- the elastic properties and band structures were calculated using the Vienna ab-initio Simulation Package.
- a 13x13x13 K-point grids for a face-centered cubic structure was selected for calculations.
- a Generalized gradient approximation (GGA) with Perdew Burke-Ernzerhof (PBE) and projector augmented wave method (PAW) were used.
- the structure model used for calculation is a typical rock salt structure, Fm3 m.
- a suitable energy cut-off is adjusted to be 600 eV for the pseudopotential sets. Only relaxed lattice structure was used calculate the ground-state energy. The convergence of energy calculation was set to be 1 ⁇ 10-5 eV.
- E total is the total energy of the complex with the optimized unit cell
- a, b, and c are the numbers of atoms for each element in the unit cell
- the elastic modulus was calculated with Voigt- Reuss-Hill approximation (VRH). For cubic structures, three independent elastic constant C11, C12, and C44, were used for the calculation. For the tetragonal structure, six independent elastic constants are C11, C12, C13, C33, C44, and C66. With calculated bulk and shear modulus, Young's modulus can be calculated as: where B represents the bulk modulus and G the shear modulus. The Poisson's ratio is expressed as:
- Hardness was estimated using the following equation: where k is the Pugh's ratio, G/B.
- Ti 1 - x V x C series has 5 compounds with x in the range of 0 - 1.
- the carbides have a cubic unit cell after optimization except Ti 0.5 V 0.5 C which has a tetragonal unit cell due to the arrangement of replaced atom position.
- the interatomic distance changes and the elastic moduli increase as more Ti is replaced by V.
- the moduli increased as Ti was replaced by V.
- the calculated hardness showed an increase from 25 GPa to about 28 GPa as the substitute V was added.
- Ti 0.5 V 0.5 C had the highest hardness value, 28.21 GPa, and Ti 0.75 V 0.25 C had the second highest hardness value of 27.82.
- Carbides having the five compositions as listed in Table 9 were made. Carbide samples were made successfully using elements as starting materials, except Ti 0.5 V 0.5 C and Ti 0 . 25 V 0 . 75 C .
- Results of the experimental analyses are given in Table 10. The results are in agreement with the calculations.
- the experimental unit cell parameters were calculated through WinCSD and compared with the optimized unit cell calculated by VASP. As more vanadium substitute was added to replace Ti, the cell parameters decreased and the unit cells shrunk.
- the back scattered image shows the uniform distribution of rod-like graphite particles.
- a map of EDX confirmed unreacted graphite in the bulk samples as well - see Fig. 16.
- the sample surface is rough due to the quick reaction. Particles do not have enough time to grow and form fine grains.
- composition of Ti 1 -xV x C samples was determined by EDX. Since carbon is a light element, the variance of result may be large.
- hardness is a very important property of carbides used as reinforcement for composites and protective coatings.
- the mirco-hardness of carbide samples was determined using the following formula:
- HV 2sin ((136°)/2)-F/d ⁇ 2 where F is kilograms-force and d is the average diagonal width of indentation.
- micro-hardness tests were not only performed on specific smooth areas, but also flat rough areas. Three or more tests with 500mN force were performed for each sample. Results of the experimentally measured hardness are in good agreement with the calculated hardness, as Table 7 illustrates.
- the wear damage to the carbides caused by the silicon nitride ball was small and not sufficiently accurate to be used for ranking their wear resistances.
- the wear resistances of the carbides were evaluated using an indirect approach by measuring the volume loss of counter-body (Si 3 N 4 ball) based on the geometry of the worn area on the Si 3 N 4 ball.
- the fermi level of all samples crossed the broad band starts from -6 -eV.
- Ti 3s/3p/3d/4s, V 3s/3p/3d/4s, and C 2s/2p states are mixed within the band.
- V states are more localized near -5 eV and 1 eV; Ti states are more localized near -5 eV and 3 eV when Ti and V are both exist in the compound.
- M-C bonding information is revealed from COHP results.
- the paper specifically explores the relationship between electron work function (EWF) and bulk modulus (B) of transition metal substituted MC carbides, which could assist in material selection of the substituted MC carbides.
- EWF electron work function
- B bulk modulus
- Section 3 high-entropy carbides (HECs)
- the selected high-entropy carbides were found to have balanced hardness, Young's modulus and toughness, compared to commonly used mono-carbides (see the Fig. 19).
- Rock-salt ceramics including rock-salt carbides, nitrides, and carbo-nitrides, were used as representative examples and conduct a systematic work based on the density-functional theory (DFT) calculations to evaluate contributions of various types of atomic bond to their mechanical properties. It was found that mechanical properties of multi- element ceramics have clear correlations with bond parameters, such as the bond order, bond ionicity, and bond length, which can be determined by those of the involved constituents. Based on the theoretically determined bond-mechanical property correlations, machine-learning models are trained to build the bridge between the bond parameters and the mechanical properties, and they perform well in predicting mechanical properties of multi-element rock-salt ceramics, consistent with computational and experimental data.
- DFT density-functional theory
- the design strategy is schematically illustrated in Fig. 20.
- prediction models correlating mechanical properties and bond parameters are trained through machine-learning.
- the machine-learning models were used to predict mechanical properties of high-entropy ceramics from their bond parameters, which were weighted from those of the involved constituents according to their atomic concentrations .
- the data-base containing mechanical properties of multi- element carbides covering millions of combinations of constituents can be quickly obtained; from which, potential candidates with desired mechanical properties were identified.
- the design strategy provided an effective and reliable approach for screening ceramics with the wished-for mechanical properties, and it particularly accelerated designing high-entropy ceramics by identifying the optimal candidates from a huge number of potential choices.
- Rock-salt carbides, nitrides, and carbonitrides have similar structures, in which metal atoms occupy all sites of the face-centered cubic (FCC) lattice, whereas non-metal atoms (C or N) occupy all the octahedral sites.
- the representative structures vanadium carbide [VC], vanadium nitride [VN], and vanadium carbonitride [V(CN)]
- Fig. 21A along with their charge density (CD) distributions, electron localization functions (ELFs),28 and densities of state (DOSs).
- the relatively higher CD between V and C(N) atoms indicates the covalency of V-C (N) bonds, which can also be reflected by the pseudo-gaps in DOSs and bond orders from density- derived electrostatic and chemical (DDEC)29 analysis. Electron localizations around the C(N) atoms are high, whereas those around the V atoms are low, showing obvious ionic characteristics, and the net charges from DDEC analysis also show the charge transfer from the V atoms to the C(N) atoms.
- the CD and ELF results and the DDEC analysis demonstrated delocalized electrons shared by metal atoms, indicating the existence of metallic bonds in the systems, corresponding to continuous valence and conduction bands in the DOSs.
- the rock-salt carbides, nitrides, and carbonitrides have mixed covalent, ionic, and metallic bonds, which synergistically determine their mechanical properties.
- Close correlations between bond strengths and mechanical properties of the mono-carbides are illustrated in Figs. 21C and 21D.
- Young's and shear moduli show similar trends with respect to the materials, which are close to that of the ionic-bond strength, while the trend of bulk modulus is similar to those of the metallic and covalent bond strengths. These suggest that the ionic bond has a predominant role in determining Young's and shear moduli of simple rock-salt ceramics, whereas the metallic and covalent bonds are more responsible to the bulk modulus .
- rock-salt ceramics can be classified into three groups (I, II, and III): compounds in group I have strong ionic bonds and weak covalent/metallic bonds, compounds in group II have strong ionic bonds and stronger covalent/metallic bonds, and those in group III have weak ionic bonds.
- groups II, and III
- groups compounds in group I have strong ionic bonds and weak covalent/metallic bonds
- compounds in group II have strong ionic bonds and stronger covalent/metallic bonds
- those in group III have weak ionic bonds.
- which group a compound belongs to depends on the type of metal elements in the corn-pound.
- the compounds in group I contain group-IIIB elements; compounds in group II contain group-IVB, VB, and VIB elements; and those in group III contain group-VIIB, VIII, IB, and IIB elements.
- Such classification provides guidance for selecting appropriate alloying elements to modify mechanical properties of rock-salt ceramics with greater effectiveness .
- High-entropy ceramics contain multi-principle alloying elements. To determine the effects of the alloying elements on the bond strength of ceramics and, consequently, the overall mechanical properties, bond strengths and mechanical properties of VC alloyed with different elements were calculated. To minimize the influence of changes in ionic bond on mechanical properties, alloying elements that had similar electro-negativities with V were selected to make the overall ionicity of alloyed carbides close to that of the VC (Fig. 22A), and structures of the alloyed carbides were initially un-relaxed to keep the bond length unchanged after alloying.
- ionic, covalent, and metallic bonds all have contributions to Young's and shear moduli at different levels, it is possible to evaluate them based on the properties of their atomic bonds. For a stronger ionic bond, more charges are transferred between the adjacent atoms with shorter bonds. For a stronger covalent bond, more electrons are expected to be shared by adjacent atoms with smaller bond lengths. For a stronger metallic bond, the density of the delocalized electrons is larger, corresponding to more electrons shared by metal atoms with smaller cell volumes. More charge transfer means a larger net charge of a cation, a smaller cell volume corresponds to a shorter bond length, and electrons shared by atoms can be reflected by the parameter SBO.
- Such descriptors also work for scaling the mechanical properties of rock-salt alkaline-earth metal oxides and sulfides and rock-salt alkali metal chlorides and fluorides. It is thus feasible to scale the mechanical properties of rock-salt ceramics using these descriptors, which are related to bond properties.
- mechanical properties of multi-element carbides, nitrides, and carbonitrides can be scaled using their bond parameters, which can be obtained from those of involved mono-carbides and nitrides.
- NbC and TaC have high Young's moduli and relatively high brittleness
- MoC and WC showed high bulk moduli but relatively low brittleness.
- the HEC NbTaMoWC4 containing Nb-C, Ta-C, Mo-C, and W-C bonds have balanced properties: high Young's and bulk moduli and lower brittleness, indicating that optimally balanced mechanical properties can be achieved from appropriate combinations of different bonds in high-entropy ceramics by alloying multi-elements.
- Bond-parameter-derived descriptors can describe trends of variations in different mechanical properties of the target materials with respect to their compositions. Machine-learning algorithms made it is feasible to predict mechanical properties from bond parameters, instead of analytically determining their complex inner correlations.
- HEMCs HE carbides
- HEMNs HE nitrides
- HMCNs HE carbonitrides
- Figs. 24D and 24E The mapped distributions of hardness, Young's modulus, and ductility of rock-salt carbides, nitrides, and carbonitrides (Figs. 24D and 24E) were obtained from the database included 436,494 ceramics from binary compounds (metal carbides “MCs” and metal nitrides “MNs”) to octonary high-entropy compounds (HEM[CN]s) based on ergodic combinations for 23 alloying metal elements.
- the distributions followed the common rule that harder materials have both greater Young's moduli and greater brittleness. Substituting C with N reduced the brittleness but also decreased the hardness. However, the hardness and brittleness are not simply correlated in a linear manner.
- Combinations of greater hardness and less brittleness are possible. Among materials having similar hardness values, those possessing greater Young's moduli are less brittle. This shows a direction for designing materials having high Young's modulus and hardness but low brittleness.
- a combination of high hardness and low brittleness is particularly desirable for ceramics, but the wished-for balance between these two properties cannot be achieved for mono-carbides, nitrides and carbonitrides because there is a large gap between materials having high hardness and brittleness and those having low hardness and brittleness (see Fig. 24E). This gap can be reduced by turning the mono-systems into multi-element ones with alloying elements, providing alternatives with the desired combinations of high hardness and high ductility or toughness.
- Densities of rock-salt carbides, nitrides, and carbonitrides can be modified when the materials contain multi-elements. Because bond lengths of multi-element ceramics can be determined from those of the constituents involved, their cell volumes and densities can be calculated based on the bond lengths determined. The modifiable density and mechanical properties certainly help optimize the hard materials for widened applications, e.g., achieving homogeneous distribution of hard ceramics as reinforcements for hard facing overlays and composites. Materials can be screened and selected, e.g., from the dashed rectangular box in Fig. 24D and their properties can be compared with those shown in Fig. 24F; from which, candidates with desired mechanical properties and densities can be identified.
Landscapes
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Organic Chemistry (AREA)
- Materials Engineering (AREA)
- Ceramic Engineering (AREA)
- Manufacturing & Machinery (AREA)
- Inorganic Chemistry (AREA)
- Theoretical Computer Science (AREA)
- Computing Systems (AREA)
- Structural Engineering (AREA)
- Metallurgy (AREA)
- Mechanical Engineering (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Bioinformatics & Computational Biology (AREA)
- Life Sciences & Earth Sciences (AREA)
- Software Systems (AREA)
- Medical Informatics (AREA)
- Data Mining & Analysis (AREA)
- Evolutionary Computation (AREA)
- Computer Vision & Pattern Recognition (AREA)
- Artificial Intelligence (AREA)
- Physics & Mathematics (AREA)
- General Engineering & Computer Science (AREA)
- General Physics & Mathematics (AREA)
- Mathematical Physics (AREA)
- Carbon And Carbon Compounds (AREA)
- Ceramic Products (AREA)
- Manufacture Of Alloys Or Alloy Compounds (AREA)
Abstract
Description
Claims
Priority Applications (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| PE2023001702A PE20231232A1 (en) | 2020-11-30 | 2021-11-30 | COMPLEX MATERIALS |
| CA3196754A CA3196754A1 (en) | 2020-11-30 | 2021-11-30 | Complex materials |
| CN202180076733.1A CN116568838A (en) | 2020-11-30 | 2021-11-30 | Composite material |
| EP21895963.3A EP4225717A4 (en) | 2020-11-30 | 2021-11-30 | Complex materials |
| US18/034,366 US20230406713A1 (en) | 2020-11-30 | 2021-11-30 | Complex Materials |
| AU2021386468A AU2021386468A1 (en) | 2020-11-30 | 2021-11-30 | Complex materials |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| AU2020904431A AU2020904431A0 (en) | 2020-11-30 | Hard Complex Carbides | |
| AU2020904431 | 2020-11-30 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2022109685A1 true WO2022109685A1 (en) | 2022-06-02 |
Family
ID=81753649
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/AU2021/051429 WO2022109685A1 (en) | 2020-11-30 | 2021-11-30 | Complex materials |
Country Status (8)
| Country | Link |
|---|---|
| US (1) | US20230406713A1 (en) |
| EP (1) | EP4225717A4 (en) |
| CN (1) | CN116568838A (en) |
| AU (1) | AU2021386468A1 (en) |
| CA (1) | CA3196754A1 (en) |
| CL (1) | CL2023001382A1 (en) |
| PE (1) | PE20231232A1 (en) |
| WO (1) | WO2022109685A1 (en) |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3859057A (en) * | 1970-03-16 | 1975-01-07 | Kennametal Inc | Hardfacing material and deposits containing tungsten titanium carbide solid solution |
| WO2009082180A2 (en) * | 2007-12-26 | 2009-07-02 | Seoul National University Industry Foundation | Solid-solution carbide/carbonitride powder and method for preparing thereof |
| JP2010115657A (en) * | 2008-11-04 | 2010-05-27 | Japan New Metals Co Ltd | Binderless powder for surface hardening |
| CN103382555A (en) * | 2013-07-12 | 2013-11-06 | 河海大学 | Precursor carbonization plasma cladding reaction synthesized WC reinforced metal based alloy coating and preparation |
| CN104120360B (en) * | 2014-07-28 | 2016-07-13 | 宁国市宁武耐磨材料有限公司 | A kind of high chromium lining sheet for ball mill |
| KR101822293B1 (en) * | 2016-08-17 | 2018-01-26 | 현대자동차주식회사 | High strength special steel |
Family Cites Families (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101743091B (en) * | 2007-08-31 | 2012-12-05 | 六号元素(产品)(控股)公司 | Polycrystalline diamond composites |
| CN107001051B (en) * | 2014-09-25 | 2020-02-07 | 德雷塞尔大学 | Physical form of MXene materials exhibiting novel electrical and optical properties |
| MA44552B1 (en) * | 2016-06-24 | 2020-11-30 | Weir Minerals Australia Ltd | Erosion and corrosion resistant white cast iron |
| JP2018035020A (en) * | 2016-08-30 | 2018-03-08 | 住友電気工業株式会社 | Aqueous solution composition and method for producing the same, oxide powder and method for producing the same, carbide powder and method for producing the same, and cemented carbide and method for producing the same |
| WO2018084184A1 (en) * | 2016-11-01 | 2018-05-11 | 新日鐵住金株式会社 | Titanium material, constituent member for cells, cell, and solid polymer fuel cell |
| CN109180189B (en) * | 2018-10-08 | 2021-03-30 | 中南大学 | High-entropy carbide ultra-high temperature ceramic powder and preparation method thereof |
| CN111471268B (en) * | 2020-03-12 | 2021-03-26 | 中国科学院化学研究所 | Carbide high-entropy ceramic precursor, high-entropy ceramic and preparation method |
| CN111943678A (en) * | 2020-08-14 | 2020-11-17 | 西北工业大学 | HfxZr1-xC ceramic solid solution nanowire and preparation method thereof |
-
2021
- 2021-11-30 US US18/034,366 patent/US20230406713A1/en active Pending
- 2021-11-30 EP EP21895963.3A patent/EP4225717A4/en active Pending
- 2021-11-30 WO PCT/AU2021/051429 patent/WO2022109685A1/en active Application Filing
- 2021-11-30 CA CA3196754A patent/CA3196754A1/en active Pending
- 2021-11-30 AU AU2021386468A patent/AU2021386468A1/en active Pending
- 2021-11-30 PE PE2023001702A patent/PE20231232A1/en unknown
- 2021-11-30 CN CN202180076733.1A patent/CN116568838A/en active Pending
-
2023
- 2023-05-12 CL CL2023001382A patent/CL2023001382A1/en unknown
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3859057A (en) * | 1970-03-16 | 1975-01-07 | Kennametal Inc | Hardfacing material and deposits containing tungsten titanium carbide solid solution |
| WO2009082180A2 (en) * | 2007-12-26 | 2009-07-02 | Seoul National University Industry Foundation | Solid-solution carbide/carbonitride powder and method for preparing thereof |
| JP2010115657A (en) * | 2008-11-04 | 2010-05-27 | Japan New Metals Co Ltd | Binderless powder for surface hardening |
| CN103382555A (en) * | 2013-07-12 | 2013-11-06 | 河海大学 | Precursor carbonization plasma cladding reaction synthesized WC reinforced metal based alloy coating and preparation |
| CN104120360B (en) * | 2014-07-28 | 2016-07-13 | 宁国市宁武耐磨材料有限公司 | A kind of high chromium lining sheet for ball mill |
| KR101822293B1 (en) * | 2016-08-17 | 2018-01-26 | 현대자동차주식회사 | High strength special steel |
Non-Patent Citations (13)
| Title |
|---|
| DATABASE Registry CAS; 17 September 1988 (1988-09-17), "Chromium iron manganese carbide ((Cr,Fe,Mn)7C3) ", XP055943046, retrieved from STN Database accession no. RN 116392-15-5 * |
| DATABASE Registry CAS; 21 November 1987 (1987-11-21), "Chromium iron vanadium carbide ((Cr,Fe,V)7C3) ", XP055943047, retrieved from STN Database accession no. RN 111465-50-0 * |
| DATABASE Registry CAS; 24 March 2011 (2011-03-24), "Chromium iron nickel carbide ((Cr,Fe,Ni)7C3)", XP055943051, retrieved from STN Database accession no. RN 1269813-06-0 * |
| DATABASE Registry CAS; 28 December 2010 (2010-12-28), "Chromium iron molybdenum carbide ((Cr,Fe,Mo)7C3)", XP055943042, retrieved from STN Database accession no. RN 1257655-61-0 * |
| DATABASE Registry CAS; 29 September 2011 (2011-09-29), "Chromium iron tungsten carbide ((Cr,Fe,W)7C3)", XP055943043, retrieved from STN Database accession no. RN 1333980-57-6 * |
| DATABASE Registry CAS; 4 October 2011 (2011-10-04), "Chromium iron titanium carbide ((Cr,Fe,Ti)7C3) ", XP055943050, retrieved from STN Database accession no. RN 1334305-62-2 * |
| HARRINGTON, T. ET AL.: "Phase stability and mechanical properties of novel high entropy transition metal carbides", ACTA MATERIALIA, vol. 166, 2019, pages 271 - 280, XP085595360, DOI: 10.1016/j.actamat.2018.12.054 * |
| KUČEROVÁ LUDMILA, ZETKOVÁ IVANA, JENÍČEK ŠTĚPÁN, BURDOVÁ KAROLÍNA: "Hybrid parts produced by deposition of 18Ni300 maraging steel via selective laser melting on forged and heat treated advanced high strength steel", ADDITIVE MANUFACTURING, vol. 32, 1 March 2020 (2020-03-01), NL , pages 101108, XP055943031, ISSN: 2214-8604, DOI: 10.1016/j.addma.2020.101108 * |
| SARKER, P. ET AL.: "High-entropy high-hardness metal carbides discovered by entropy descriptors", NAT COMMUN., vol. 9, 2018, pages 1 - 10, XP055943024 * |
| See also references of EP4225717A4 * |
| TĘCZA G, SOBULA S: "Effect of Heat Treatment on Change Microstructure of Cast High-manganese Hadfield Steel with Elevated Chromium Content", ARCHIVES OF FOUNDRY ENGINEERING, vol. 14, no. special issue 3, 1 January 2014 (2014-01-01), PL , pages 67 - 70, XP055943029, ISSN: 1897-3310 * |
| VECCHIO KENNETH S ET AL.: "MODELING AND SYNTHESIS OF HIGH-ENTROPY REFRACTORY CARBIDES, NITRIDES, AND CARBONITRIDES", 1 September 2017 (2017-09-01), XP055943027, Retrieved from the Internet <URL:https://dc.engconfintl.org/uhtc_iv/32/> * |
| ZHANG HUI, CHONG KAI, ZHAO WEI, SUN ZHIPING: "Laser cladding in-situ micro and sub-micro/nano Ti-V carbides reinforced Fe-based layers by optimizing initial alloy powders size", MATERIALS LETTERS, vol. 220, 1 June 2018 (2018-06-01), AMSTERDAM, NL , pages 44 - 46, XP055943034, ISSN: 0167-577X, DOI: 10.1016/j.matlet.2018.02.130 * |
Also Published As
| Publication number | Publication date |
|---|---|
| CL2023001382A1 (en) | 2024-01-05 |
| EP4225717A4 (en) | 2024-04-24 |
| CA3196754A1 (en) | 2022-06-02 |
| US20230406713A1 (en) | 2023-12-21 |
| PE20231232A1 (en) | 2023-08-21 |
| CN116568838A (en) | 2023-08-08 |
| AU2021386468A1 (en) | 2023-06-01 |
| EP4225717A1 (en) | 2023-08-16 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Harrington et al. | Phase stability and mechanical properties of novel high entropy transition metal carbides | |
| Csanádi et al. | Strength enhancement and slip behaviour of high-entropy carbide grains during micro-compression | |
| Cui et al. | Strengthening mechanism in two-phase FeCoCrNiMnAl high entropy alloy coating | |
| Kindlund et al. | A review of the intrinsic ductility and toughness of hard transition-metal nitride alloy thin films | |
| Munoz et al. | Adhesion at metal–ZrO2 interfaces | |
| Wang et al. | High-entropy carbide ceramics: a perspective review | |
| WO2011035193A1 (en) | Compositions and methods for determining alloys for thermal spray, weld overlay, thermal spray post processing applications, and castings | |
| Esmaily et al. | High-temperature oxidation behaviour of Al x FeCrCoNi and AlTiVCr compositionally complex alloys | |
| Lou et al. | Development of robust surfaces for harsh service environments from the perspective of phase formation and transformation | |
| Srivastava et al. | Microstructural and fractographic analysis of A359/Si3N4 surface composite produced by friction stir processing | |
| Butola et al. | Fabrication and multi-objective optimization of friction stir processed aluminium based surface composites using Taguchi approach | |
| Gu et al. | Solving strength–toughness dilemma in superhard transition-metal diborides via a distinct chemically tuned solid solution approach | |
| Garg et al. | Improving the high temperature oxidation resistance of high entropy alloy by surface modification | |
| Gao et al. | The improvement of room temperature plasticity of refractory high entropy alloy based on different first principles calculation models and experiment verification | |
| Chen et al. | Effects of the proportions of carbon on the microstructure and properties of NbC-reinforced Ni-WC composite coatings by laser cladding in-situ synthesis | |
| Hugosson et al. | The connection between the electronic structure and the properties of binderless tungsten carbides | |
| Rokanopoulou et al. | Titanium carbide/duplex stainless steel (DSS) metal matrix composite coatings prepared by the plasma transferred arc (PTA) technique: microstructure and wear properties | |
| US20230406713A1 (en) | Complex Materials | |
| Saxena et al. | Investigation of microstructural and wear behavior of Al6061 surface composites fabricated by friction stir process using Taguchi approach | |
| Węglewski et al. | Anomalous size effect in thermal residual stresses in pressure sintered alumina-chromium composites | |
| Yousfi et al. | Creep of un-doped and Cr-doped WC-Co at high temperature and high load | |
| Saha et al. | Attainment of high specific hardness and specific modulus in spark plasma sintered aluminium‐copper‐silicon carbide‐titanium carbide hybrid composite | |
| Salarpour et al. | Characterization of 1050Al/Al3Ni/ZrO2 hybrid surface composites fabricated by friction stir processing | |
| Zhang et al. | V-substituted Ti1-xVxC carbides in comparison with Mono-TiC carbide–A study combining first-principle calculations and experiments | |
| Adapa et al. | High throughput structure–property relationship for additively manufactured 316L/IN625 alloy mixtures leveraging 2-step Bayesian estimation |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 21895963 Country of ref document: EP Kind code of ref document: A1 |
|
| DPE1 | Request for preliminary examination filed after expiration of 19th month from priority date (pct application filed from 20040101) | ||
| ENP | Entry into the national phase |
Ref document number: 3196754 Country of ref document: CA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 18034366 Country of ref document: US |
|
| ENP | Entry into the national phase |
Ref document number: 2021895963 Country of ref document: EP Effective date: 20230509 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 202180076733.1 Country of ref document: CN |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: 112023008660 Country of ref document: BR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 001702-2023 Country of ref document: PE |
|
| ENP | Entry into the national phase |
Ref document number: 2021386468 Country of ref document: AU Date of ref document: 20211130 Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 112023008660 Country of ref document: BR Kind code of ref document: A2 Effective date: 20230505 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |