WO2021233786A1 - Herbicidal cinnoline derivatives - Google Patents
Herbicidal cinnoline derivatives Download PDFInfo
- Publication number
- WO2021233786A1 WO2021233786A1 PCT/EP2021/062884 EP2021062884W WO2021233786A1 WO 2021233786 A1 WO2021233786 A1 WO 2021233786A1 EP 2021062884 W EP2021062884 W EP 2021062884W WO 2021233786 A1 WO2021233786 A1 WO 2021233786A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- phenyl
- oxo
- cinnoline
- trifluoromethoxy
- heterocyclyl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 0 *C(c1cccc(*(C2)(C2*=C2C(*)=O)c(cc3)ccc3O)c1C2=O)=O Chemical compound *C(c1cccc(*(C2)(C2*=C2C(*)=O)c(cc3)ccc3O)c1C2=O)=O 0.000 description 7
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D237/00—Heterocyclic compounds containing 1,2-diazine or hydrogenated 1,2-diazine rings
- C07D237/26—Heterocyclic compounds containing 1,2-diazine or hydrogenated 1,2-diazine rings condensed with carbocyclic rings or ring systems
- C07D237/28—Cinnolines
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N25/00—Biocides, pest repellants or attractants, or plant growth regulators, characterised by their forms, or by their non-active ingredients or by their methods of application, e.g. seed treatment or sequential application; Substances for reducing the noxious effect of the active ingredients to organisms other than pests
- A01N25/32—Ingredients for reducing the noxious effect of the active substances to organisms other than pests, e.g. toxicity reducing compositions, self-destructing compositions
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/48—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with two nitrogen atoms as the only ring hetero atoms
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/48—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with two nitrogen atoms as the only ring hetero atoms
- A01N43/58—1,2-Diazines; Hydrogenated 1,2-diazines
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/64—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with three nitrogen atoms as the only ring hetero atoms
- A01N43/647—Triazoles; Hydrogenated triazoles
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/72—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with nitrogen atoms and oxygen or sulfur atoms as ring hetero atoms
- A01N43/74—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with nitrogen atoms and oxygen or sulfur atoms as ring hetero atoms five-membered rings with one nitrogen atom and either one oxygen atom or one sulfur atom in positions 1,3
- A01N43/76—1,3-Oxazoles; Hydrogenated 1,3-oxazoles
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/72—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with nitrogen atoms and oxygen or sulfur atoms as ring hetero atoms
- A01N43/80—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with nitrogen atoms and oxygen or sulfur atoms as ring hetero atoms five-membered rings with one nitrogen atom and either one oxygen atom or one sulfur atom in positions 1,2
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/72—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with nitrogen atoms and oxygen or sulfur atoms as ring hetero atoms
- A01N43/84—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with nitrogen atoms and oxygen or sulfur atoms as ring hetero atoms six-membered rings with one nitrogen atom and either one oxygen atom or one sulfur atom in positions 1,4
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01P—BIOCIDAL, PEST REPELLANT, PEST ATTRACTANT OR PLANT GROWTH REGULATORY ACTIVITY OF CHEMICAL COMPOUNDS OR PREPARATIONS
- A01P13/00—Herbicides; Algicides
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D403/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00
- C07D403/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings
- C07D403/04—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings directly linked by a ring-member-to-ring-member bond
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D405/00—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom
- C07D405/02—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing two hetero rings
- C07D405/04—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing two hetero rings directly linked by a ring-member-to-ring-member bond
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D413/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D413/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings
- C07D413/04—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings directly linked by a ring-member-to-ring-member bond
Definitions
- the present invention relates to herbicidal cinnoline derivatives, e.g., as active ingredients, which have herbicidal activity.
- the invention also relates to agrochemical compositions which comprise at least one of the cinnoline derivatives, to processes of preparation of these compounds and to uses of the cinnoline derivatives or compositions in agriculture or horticulture for controlling weeds, in particular in crops of useful plants.
- EP0273325, EP0274717, and US5183891 describe cinnoline derivatives as herbicidal agents.
- X is O, NR 13 or S
- R 1 is phenyl optionally substituted with 1 , 2, 3, or 4 groups, which may be the same or different, represented by R 7 ;
- R 3 is hydrogen, Ci-C6alkyl, Ci-C6haloalkyl, C3-C6cycloalkyl, C3-C6cycloalkylCi-C6alkyl, Ci- C6alkoxyCi-C6alkyl, C2-C6alkenyl, C2-C6alkynyl, phenyl, or phenylCi-C3alkyl, wherein the phenyl moieties may be optionally substituted with 1 , 2, 3 or 4 groups, which may be the same or different, represented by R 12 ;
- R 4 , R 5 , and R 6 are each independently selected from hydrogen, halogen, cyano, Ci-C6alkyl, Ci- C6alkoxy, Ci-C6haloalkyl, Ci-C6haloalkoxy, Ci-C6alkylsulfanyl, Ci-Cealkylsulfinyl, and Ci- C6alkylsulfonyl;
- R 7 is halogen, cyano, Ci-C6alkyl, Ci-C6alkoxy, Ci-C6haloalkyl, Ci-C6haloalkoxy, Ci- Cealkylsulfanyl, or Ci-C6alkylsulfonyl; or any two adjacent R 7 groups together with the carbon atoms to which they are attached, may form a 5- or 6-membered heterocyclyl ring, comprising 1 or 2 heteroatoms selected from O and N, or any two adjacent R 7 groups together with the carbon atoms to which they are attached, may form
- R 8 and R 9 are each independently selected from halogen, Ci-C3alkyl, Ci-C3haloalkyl, and Ci-
- R 10 and R 11 are each independently selected from hydrogen and Ci-C3alkyl
- R 12 is halogen, cyano, Ci-C3alkyl, or Ci-C3alkoxy;
- R 13 is hydrogen, Ci-C3alkyl, or Ci-C3alkoxy; or a salt or an N-oxide thereof.
- novel compounds of Formula (I) have, for practical purposes, a very advantageous level of herbicidal activity.
- an agrochemical composition comprising a herbicidally effective amount of a compound of Formula (I) according to the present invention.
- Such an agricultural composition may further comprise at least one additional active ingredient and/or an agrochemically-acceptable diluent or carrier.
- a method of controlling weeds at a locus comprising applying to the locus a weed controlling amount of a composition comprising a compound of Formula (I).
- Ci-Csalkyl substituted by 1 , 2 or 3 halogens may include, but not be limited to, -CH2CI, -CHCI2, -CCI3, -CH2F, -CHF2, -CF3, -CH2CF3 or -CF2CH3 groups.
- Ci-C6alkoxy substituted by 1 , 2 or 3 halogens may include, but not limited to, CH2CIO-, CHCI2O-, CCI3O-, CH2FO-, CHF2O-, CF3O-, CF3CH2O- or CH3CF2O- groups.
- cyano means a -CN group.
- halogen refers to fluorine (fluoro), chlorine (chloro), bromine (bromo) or iodine (iodo).
- hydroxy means an -OH group
- acetyl means a -C(0)CH3 group.
- nitro means an NO2 group.
- Ci-C6alkyl refers to a straight or branched hydrocarbon chain radical consisting solely of carbon and hydrogen atoms, containing no unsaturation, having from one to six carbon atoms, and which is attached to the rest of the molecule by a single bond. “Ci-C 4 alkyl” and “Ci- C3alkyl” are to be construed accordingly. Examples of Ci-C6alkyl include, but are not limited to, methyl, ethyl, n-propyl, and the isomers thereof, for example, iso-propyl.
- Ci-C6alkylene refers to the corresponding definition of Ci-C6alkyl, except that such radical is attached to the rest of the molecule by two single bonds.
- the term “Ci-C2alkylene” is to be construed accordingly.
- Examples of Ci-C6alkylene include, but are not limited to, -CH2-, -CH2CH2- and -(CH2)3-.
- Ci-C6haloalkyl refers a Ci-C6alkyl radical as generally defined above substituted by one or more of the same or different halogen atoms.
- Examples of Ci-C6haloalkyl include, but are not limited to trifluoromethyl.
- Ci-C6di-haloalkyl refers a Ci-C6alkyl radical as generally defined above substituted by two of the same or different halogen atoms.
- Examples of Ci-C6di-haloalkyl include, but are not limited to difluoromethyl.
- cyanoCi-Cealkyl refers to a Ci-C6alkyl radical as generally defined above substituted by one or more cyano groups, as defined above.
- cyanoCi-Cealkyl include, but are not limited to 2-cyanomethyl and 2-cyanoethyl.
- Ci-C6haloalkoxy refers to a Ci-C6alkoxy radical as generally defined above substituted by one or more of the same or different halogen atoms.
- Examples of Ci-C6haloalkoxy include, but are not limited to trifluoromethoxy.
- Ci-C6alkoxy refers to a radical of the formula -OR a where R a is a Ci- C6alkyl radical as generally defined above.
- R a is a Ci- C6alkyl radical as generally defined above.
- Ci-C 4 alkoxy and “Ci-C3alkoxy” are to be construed accordingly.
- Examples of Ci-C6alkoxy include, but are not limited to, methoxy, ethoxy, 1- methylethoxy (iso-propoxy), and propoxy.
- C2-C6alkenyl refers to a straight or branched hydrocarbon chain radical group consisting solely of carbon and hydrogen atoms, containing at least one double bond that can be of either the (E)- or ( ⁇ -configuration, having from two to six carbon atoms, which is attached to the rest of the molecule by a single bond.
- C 2 -C3alkenyl is to be construed accordingly. Examples of C2-C6alkenyl include, but are not limited to, ethenyl (vinyl), prop-1 -enyl, prop-2-enyl (allyl), but-1-enyl.
- C2-C6alkynyl refers to a straight or branched hydrocarbon chain radical group consisting solely of carbon and hydrogen atoms, containing at least one triple bond, having from two to six carbon atoms, and which is attached to the rest of the molecule by a single bond.
- C 2 -C3alkynyl is to be construed accordingly. Examples of C2-C6alkynyl include, but are not limited to, ethynyl, prop-1 -ynyl, but-1-ynyl.
- Ci-C6alkoxyCi-C6alkyl refers to a radical of the formula RbORa- wherein Rb is a Ci-C6alkyl radical as generally defined above, and R a is a Ci-C6alkylene radical as generally defined above.
- cyanoCi-C6alkyl“ refers to a Ci-C6alkyl radical as generally defined above substituted by one or more cyano groups, as defined above.
- Examples of cyanoCi-Cealkyl include, but are not limited to 2-cyanoethyl.
- C3-C6cycloalkyl refers to a radical which is a monocyclic saturated ring system and which contains 3 to 6 carbon atoms.
- the terms “Cs-Cscycloalkyl” and “C3-C 4 cycloalkyl” are to be construed accordingly.
- Examples of C3-C6cycloalkyl include, but are not limited to, cyclopropyl, cyclobutyl, cyclopentyl and cyclohexyl.
- C3-C6cycloalkylCi-C6alkyl refers to a C3-C6cycloalkyl ring attached to the rest of the molecule by a Ci-C6alkylene linker as defined above.
- Ci-C6alkoxyC2-C6alkenyl“ refers to a a radical of the formula RbORa- wherein Rb is a Ci-C6alkyl radical as generally defined above, and R a is a Ci-C6alkene radical as generally defined above.
- Examples of Ci-C6alkoxyC2-C6alkenyl include, but are not limited to 1- methoxyvinyl and 1-ethoxyvinyl.
- C2-C6alkenyloxyCi-C6alkyl“ refers to a radical of the formula RbORa- wherein Rb is a C2-C6alkenyl radical as generally defined above, and R a is a Ci-C6alkylene radical as generally defined above.
- oxo-Ci-C6alkyl“ refers to a radical of the formula -R a CHO, wherein R a is a Ci-C6alkene radical as generally defined above.
- R a is a Ci-C6alkene radical as generally defined above.
- Examples of “oxo-Ci-C6alkyl“ include, but are not limited to 2-oxoethyl.
- phenoxy refers to a phenyl ring attached to the rest of the molecule through an oxygen atom.
- phenylCi-C3alkyl refers to a phenyl ring attached to the rest of the molecule by a Ci-C3alkylene linker as defined above.
- heterocyclyl refers to a stable 4-, 5- or 6-membered non-aromatic monocyclic ring which comprises 1 or 2 heteroatoms, wherein the heteroatoms are individually selected from nitrogen, oxygen, and sulfur.
- the heterocyclyl radical may be bonded to the rest of the molecule via a carbon atom or heteroatom.
- heterocyclyl examples include, but are not limited to, azetidinyl, oxetanyl, thietanyl, tetrahydrofuranyl, pyrrolidinyl, pyrazolidinyl, imidazolidnyl, piperidinyl, piperazinyl, morpholinyl, dioxolanyl, dithiolanyl and thiazolidinyl.
- heterocyclyloxy refers to a heterocyclyl ring attached to the rest of the molecule through an oxygen atom.
- heteroaryl refers to a 5- or 6-membered aromatic monocyclic ring radical which comprises 1 , 2, 3 or 4 heteroatoms individually selected from nitrogen, oxygen, and sulfur.
- heteroaryl include, but are not limited to, furanyl, pyrrolyl, thienyl, pyrazolyl, imidazolyl, thiazolyl, isothiazolyl, oxazolyl, isoxazolyl, triazolyl, tetrazolyl, pyrazinyl, pyridazinyl, pyrimidyl or pyridyl.
- Ci-C6alkylcarbonyl refers to a radical of the formula -C(0)R a , where R a is a Ci-C6alkyl radical as generally defined above.
- Ci-C6alkylsulfanyl refers to a radical of the formula -SR a , where R a is a Ci-C6alkyl radical as generally defined above.
- R a is a Ci-C6alkyl radical as generally defined above.
- Ci-C 4 alkylsulfanyl and “Ci-C3alkylsulfanyl”, are to be construed accordingly.
- Examples of Ci-C6alkylsulfanyl include, but are not limited to methylsulfanyl.
- Ci-Cealkylsulfinyl refers to a radical of the formula -S(0)R a , where R a is a Ci-C6alkyl radical as generally defined above.
- R a is a Ci-C6alkyl radical as generally defined above.
- Ci-C 4 alkylsulfinyl and “Ci-C3alkylsulfinyl”, are to be construed accordingly.
- Examples of Ci-Cealkylsulfinyl include, but are not limited to methylsulfinyl.
- Ci-C6alkylsulfonyl refers to a radical of the formula -S(0) 2 R a , where R a is a Ci-C6alkyl radical as generally defined above.
- R a is a Ci-C6alkyl radical as generally defined above.
- Ci-C 4 alkylsulfonyl and “Ci- C3alkylsulfonyl”, are to be construed accordingly.
- Examples of Ci-C6alkylsolfanyl include, but are not limited to methylsulfonyl.
- the presence of one or more possible stereogenic elements in a compound of formula (I) means that the compounds may occur in optically isomeric forms, i.e., enantiomeric or diastereomeric forms. Also, atropisomers may occur as a result of restricted rotation about a single bond.
- Formula (I) is intended to include all those possible isomeric forms and mixtures thereof. The present invention includes all those possible isomeric forms and mixtures thereof for a compound of formula (I).
- formula (I) is intended to include all possible tautomers. The present invention includes all possible tautomeric forms for a compound of formula (I).
- the compounds of formula (I) according to the invention are in free form, in oxidized form as an N-oxide, or in salt form, e.g., an agronomically usable salt form.
- Salts that the compounds of Formula (I) may form with amines including primary, secondary and tertiary amines (for example ammonia, dimethylamine and triethylamine), alkali metal and alkaline earth metal bases, transition metals or quaternary ammonium bases are preferred.
- N-oxides are oxidized forms of tertiary amines or oxidized forms of nitrogen-containing heteroaromatic compounds. They are described for instance in the book “Heterocyclic N-oxides” by A. Albini and S. Pietra, CRC Press, Boca Raton (1991).
- X is O, N or S. In one set of embodiments, X is O. In another set of embodiments, X is S. In a further set of embodiments, X is N.
- R 1 is phenyl optionally substituted with 1 , 2, 3, or 4 groups, which may be the same or different, represented by R 7 .
- R 1 is phenyl optionally substituted with 1 , 2, or 3 groups, which may be the same or different, represented by R 7 .
- R 1 is phenyl optionally substituted with 1 or 2 groups, which may be the same or different, represented by R 7 .
- R 1 is phenyl optionally substituted with a single group represented by R 7 .
- R 1 is phenyl subsitututed in the para position by a single group represented by R 7 .
- R 1 is 4-(trifluoromethoxy)phenyl, 4-chlorophenyl, 3,4- dimethoxyphenyl, 4-methylsulfanylphenyl, 3-chloro-5-methyl-phenyl, 4-(trifluoromethyl)phenyl, 4- cyanophenyl, 7-quinolyl, 2,2-difluoro-1 ,3-benzodioxol-5-yl, 1-methylindazol-6-yl, 2,2,3,3-tetrafluoro-1 ,4- benzodioxin-6-yl, 3-chloro-4-methyl-phenyl, 3,4-dichlorophenyl, 3-cyanophenyl, 3-chlorophenyl, or 3- chloro-4-fluoro-phenyl.
- R 1 is 4-(trifluoromethoxy)phenyl, 4-chlorophenyl, or 3,4- dimethoxyphenyl. In a further set of embodiments, R 1 is 4-(trifluoromethoxy)phenyl, or 4-chlorophenyl.
- R 8 is halogen (preferably bromo
- R 2 is cyano, cyanomethyl, difluoromethyl, acetyl, vinyl, 1-methoxyvinyl, 1- ethoxyvinyl, N-methoxy-C-methylcarbonimidoyl, N-hydroxy-C-methyl-carbonimidoyl, 2-oxo-ethyl, nitro, phenyl, phenoxy, oxazolyl, isoxazolyl, pyrazolyl, triazolyl, pyrrolidinyl, piperidinyl, morpholino, and wherein the phenyl, phenoxy, oxazolyl, isoxazolyl, pyrazolyl, and triazolyl, moieties may each be optionally substituted with a single substituent represented by R 8 .
- R 2 is cyano, cyanomethyl, difluoromethyl, acetyl, vinyl, 1- methoxyvinyl, 1-ethoxyvinyl, N-methoxy-C-methylcarbonimidoyl, N-hydroxy-C-methyl-carbonimidoyl, 2- oxo-ethyl, nitro, phenoxy, 5-oxazol-2-yl, isoxazol-3-yl, pyrazol-1-yl, triazol-1-yl, triazol-2-yl, 1 -piperidinyl, morpholino, and wherein the phenoxy, isoxazolyl, pyrazolyl, and triazolyl, moieties may each be optionally substituted with a single substituent represented by R 8 .
- R 2 is cyano, cyanomethyl, difluoromethyl, acetyl, vinyl, 1- methoxyvinyl, 1-ethoxyvinyl, N-methoxy-C-methylcarbonimidoyl, N-hydroxy-C-methyl-carbonimidoyl, 2- oxo-ethyl, nitro, phenoxy, 5-oxazol-2-yl, 5-(difluoromethyl)isoxazol-3-yl, 4-chloropyrazol-1-yl, 4- (trifluoromethyl)triazol-l-yl, 4-(trifluoromethyl)triazol-2-yl, 1 -piperidinyl, or morpholino.
- R 2 is cyano, acetyl, propanoyl, 2-methylpropanoyl, 1-methoxyvinyl, 1- ethoxyvinyl, N-methoxy-C-methylcarbonimidoyl, N-ethoxy-C-methylcarbonimidoyl, phenyl, phenoxy, heteroaryl, wherein the heteroaryl moiety is a 5-membered aromatic monocyclic ring comprising 1 or 2 heteroatoms individually selected from N and O, or heterocyclyl, wherein the heterocyclyl moiety is a 5- or 6-membered non-aromatic monocyclic ring comprising 1 or 2 heteroatoms individually selected from N and O, and wherein the phenyl and phenoxy moieties may each be optionally substituted with a single group represented by R 8 .
- R 2 is cyano, acetyl, 1-methoxyvinyl, 1-ethoxyvinyl, N-methoxy-C- methylcarbonimidoyl, 4-fluorophenyl, 4-fluorophenoxy, or oxazol-2-yl.
- R 2 is cyanomethyl, difluoromethyl, vinyl, 1- methoxyvinyl, 1-ethoxyvinyl, N-methoxy-C-methylcarbonimidoyl, N-hydroxy-C-methyl-carbonimidoyl, 2- oxo-ethyl, phenoxy, 5-oxazol-2-yl, 5-(difluoromethyl)isoxazol-3-yl, 4-chloropyrazol-1-yl, 4- (trifluoromethyl)triazol-l-yl, 4-(trifluoromethyl)triazol-2-yl, 1-piperidinyl, or morpholino.
- R 3 is hydrogen, Ci-C6alkyl, Ci-C6haloalkyl, C3-C6cycloalkyl, C3-C6cycloalkylCi-C6alkyl, Ci- CealkoxyCi-Cealkyl, C2-C6alkenyl, C2-C6alkynyl, phenyl, or phenylCi-C3alkyl, wherein the phenyl moieties may be optionally substituted with 1 , 2, 3 or 4 groups, which may be the same or different, represented by R 12 .
- R 3 is hydrogen, Ci-C6alkyl, Ci-C6haloalkyl, C3-C6cycloalkyl, C3- C6cycloalkylCi-C3alkyl, Ci-C 4 alkoxyCi-C3alkyl, C2-C6alkenyl, C2-C6alkynyl, phenyl, or phenylCi-C2alkyl, wherein the phenyl moieties may be optionally substituted with 1 , 2, 3 or 4 groups, which may be the same or different, represented by R 12 .
- R 3 is hydrogen, Ci-C6alkyl, Ci-C 4 haloalkyl, C3- C6cycloalkyl, C3-C6cycloalkylCi-C3alkyl, Ci-C 4 alkoxyCi-C3alkyl, C2-Csalkenyl, C2-Csalkynyl, phenyl, or benzyl, wherein the phenyl moieties may be optionally substituted with 1 , 2, or 3 groups, which may be the same or different, represented by R 12 .
- R 3 is hydrogen, Ci-C6alkyl, Ci- C 4 haloalkyl, C3-C6cycloalkyl, C3-C6cycloalkylCi-C3alkyl, Ci-C 4 alkoxyCi-C3alkyl, phenyl, or benzyl, wherein the phenyl moieties may be optionally substituted with 1 or 2 groups, which may be the same or different, represented by R 12 .
- R 3 is hydrogen or Ci-C6alkyl.
- R 3 is hydrogen or Ci-C 4 alkyl, more preferably, hydrogen or Ci-C3alkyl.
- R 3 is hydrogen, methyl, or ethyl. More preferably still, R 3 is hydrogen or methyl.
- R 4 and R 5 are each independently selected from hydrogen, halogen, cyano, Ci-C6alkyl, Ci- Cealkoxy, Ci-C6haloalkyl, Ci-C6haloalkoxy, Ci-C6alkylsulfanyl, Ci-Cealkylsulfinyl, and Ci- Cealkylsulfonyl, and R 6 is hydrogen.
- R 4 and R 5 are each independently selected from hydrogen, halogen, cyano, Ci- C 4 alkyl, Ci-C 4 alkoxy, Ci-C 4 haloalkyl, Ci-C 4 haloalkoxy, Ci-C 4 alkylsulfanyl, Ci-C 4 alkylsulfinyl, and Ci- C 4 alkylsulfonyl, and R 6 is hydrogen.
- R 4 and R 5 are each independently selected from hydrogen, halogen, cyano, Ci- C 4 alkyl, Ci-C3alkoxy, Ci-C3haloalkyl, Ci-C3haloalkoxy, Ci-C3alkylsulfanyl, Ci-C3alkylsulfinyl, and Ci- C3alkylsulfonyl, and R 6 is hydrogen.
- R 4 and R 5 are each independently selected from hydrogen, fluoro, bromo, cyano, Ci-C 4 alkyl, methoxy, ethoxy, trifluoromethyl, trifluoromethoxy, methylsulfanyl, and methylsulfonyl, and R 6 is hydrogen.
- R 4 and R 5 are each independently selected from hydrogen, fluoro, bromo, cyano, methyl, isobutyl, methoxy, and trifluoromethyl, and R 6 is hydrogen.
- R 4 , R 5 , and R 6 are each independently selected from hydrogen, halogen, cyano, Ci-C6alkyl, Ci-C6alkoxy, Ci-C6haloalkyl, Ci-C6haloalkoxy, Ci-C6alkylsulfanyl, Ci-Cealkylsulfinyl, and Ci-C6alkylsulfonyl.
- R 4 , R 5 , and R 6 are each independently selected from hydrogen, halogen, cyano, Ci-C 4 alkyl, Ci-C 4 alkoxy, Ci-C 4 haloalkyl, Ci-C 4 haloalkoxy, Ci-C 4 alkylsulfanyl, Ci- C 4 alkylsulfinyl, and Ci-C 4 alkylsulfonyl.
- R 4 , R 5 , and R 6 are each independently selected from hydrogen, halogen, cyano, Ci-C 4 alkyl, Ci-C3alkoxy, Ci-C3haloalkyl, Ci-C3haloalkoxy, Ci- C3alkylsulfanyl, Ci-C3alkylsulfinyl, and Ci-C3alkylsulfonyl. More preferably still, R 4 , R 5 , and R 6 are each independently selected from hydrogen, fluoro, bromo, cyano, Ci-C 4 alkyl, methoxy, ethoxy, trifluoromethyl, trifluoromethoxy, methylsulfanyl, and methylsulfonyl. Even more preferably, R 4 , R 5 , and R 6 are each independently selected from hydrogen, fluoro, bromo, cyano, methyl, isobutyl, methoxy, and trifluoromethyl.
- R 4 and R 5 are each independently selected from hydrogen, fluoro, bromo, cyano, methyl, isobutyl, methoxy, and trifluoromethyl, and R 6 is hydrogen. In a further set of embodiments, R 4 , R 5 , and R 6 are all hydrogen.
- R 7 is halogen, cyano, Ci-C6alkyl, Ci-C6alkoxy, Ci-C6haloalkyl, Ci-C6haloalkoxy, Ci- Cealkylsulfanyl, or Ci-C6alkylsulfonyl; or any two adjacent R 7 groups together with the carbon atoms to which they are attached, may form a 5- or 6-membered heterocyclyl ring, comprising 1 or 2 heteroatoms selected from O and N, or any two adjacent R 7 groups together with the carbon atoms to which they are attached, may form a 5- or 6- membered heteroaryl ring, comprising 1 or 2 heteroatoms selected from O and N, and wherein the heterocyclyl or heteroaryl rings may be optionally substituted with 1 , 2, 3 or 4 groups, which may be the same or different, represented by R 9 .
- R 7 is halogen, cyano, Ci-C3alkyl, Ci-C3alkoxy, Ci-C3haloalkyl, Ci-C3haloalkoxy, Ci-C3alkylsulfanyl, or Ci-C3alkylsulfonyl; or any two adjacent R 7 groups together with the carbon atoms to which they are attached, may form a 5- or 6-membered heterocyclyl ring, comprising 1 or 2 heteroatoms selected from O and N, or any two adjacent R 7 groups together with the carbon atoms to which they are attached, may form a 5- or 6- membered heteroaryl ring, comprising 1 or 2 heteroatoms selected from O and N, and wherein the heterocyclyl or heteroaryl rings may be optionally substituted with 1 , 2, 3 or 4 groups, which may be the same or different, represented by R 9 .
- R 7 is halogen, cyano, Ci-C3alkyl, Ci-C3alkoxy, Ci-C3haloalkyl, Ci- C3haloalkoxy, or Ci-C3alkylsulfanyl; or any two adjacent R 7 groups together with the carbon atoms to which they are attached, may form a 5- or 6-membered heterocyclyl ring, comprising 1 or 2 oxygen atoms, or any two adjacent R 7 groups together with the carbon atoms to which they are attached, may form a 5- or 6-membered heteroaryl ring, comprising 1 or 2 nitrogen atoms, and wherein the heterocyclyl or heteroaryl rings may be optionally substituted with 1 , 2, 3 or 4 groups, which may be the same or different, represented by R 9 .
- R 7 is chloro, cyano, methyl, methoxy, trifluoroalkyl, trifluoromethoxy, methylsulfanyl, or any two adjacent R 7 groups together with the carbon atoms to which they are attached form a quinolyl, indazolyl, 1 ,3-benzoxadiozolyl, or 1 ,4-benzodioxinyl group, and wherein the quinolyl, indazolyl, 1 ,3-benzoxadiozolyl, or 1 ,4-benzodioxinyl groups may be optionally substituted with 1 2, 3 or 4 groups, which may be the same or different, represented by R 9 .
- R 7 is halogen, cyano, Ci-C6alkyl, Ci-C6alkoxy, Ci-C6haloalkyl, Ci- Cehaloalkoxy, Ci-C6alkylsulfanyl, or Ci-C6alkylsulfonyl; or any two adjacent R 7 groups together with the carbon atoms to which they are attached, may form a 5- or 6-membered heterocyclyl ring, comprising 1 or 2 heteroatoms selected from O and N, and wherein the heterocyclyl ring may be optionally substituted with 1 , 2, 3 or 4 groups, which may be the same or different, represented by R 9 .
- R 7 is halogen, cyano, Ci-C3alkyl, Ci-C3alkoxy, Ci-C3haloalkyl, Ci-C3haloalkoxy, Ci- C3alkylsulfanyl, Ci-C3alkylsulfinyl, or Ci-C3alkylsulfonyl; or any two adjacent R 7 groups together with the carbon atoms to which they are attached, may form a 5- or 6-membered heterocyclyl ring, comprising 1 or 2 heteroatoms selected from O and N, and wherein the heterocyclyl ring may be optionally substituted with 1 , 2 or 3 groups, which may be the same or different, represented by R 9 .
- R 7 is halogen, cyano, Ci-C3alkyl, Ci-C3alkoxy, Ci-C3haloalkyl, Ci-C3haloalkoxy, Ci-C3alkylsulfanyl, Ci-C3alkylsulfinyl, or Ci-C3alkylsulfonyl.
- R 7 is fluoro, bromo, chloro, cyano, methyl, ethyl, isopropyl, methoxy, ethoxy, trifluoromethyl, trifluoromethoxy, methylsulfanyl, methylsulfinyl, or methylsulfonyl; or any two adjacent R 7 groups together with the carbon atoms to which they are attached, may form a 5- or 6-membered heterocyclyl ring, comprising 1 or 2 heteroatoms selected from O and N, and wherein the heterocyclyl ring may be optionally substituted with 1 or 2 groups, which may be the same or different, represented by R 9 .
- R 7 is fluoro, bromo, chloro, cyano, methyl, methoxy, trifluoromethyl, or trifluoromethoxy. More preferably still, R 7 is chloro, methoxy, or trifluoromethoxy. Even more preferably still, R 7 is chloro or trifluoromethoxy.
- R 7 is Ci-C6haloalkoxy.
- R 7 is Ci-C 4 haloalkoxy, more preferably, R 7 is Ci-C3haloalkoxy, Even more preferably, R 7 is Ci-C3fluoroalkoxy, and more preferably still, R 7 is trifluoromethoxy.
- R 8 and R 9 are each independently selected from halogen, Ci-C3alkyl, Ci-C3haloalkyl, and Ci- C3alkoxy.
- each R 8 is independently selected from halogen and Ci-C3haloalkyl. More preferably, each R 8 is independently selected from halogen and Ci-C3fluoroalkyl. Even more preferably, each R 8 is independently selected from fluoro, chloro, difluoromethyl, and trifluoromethyl.
- each R 9 is independently selected from halogen and Ci-C3alkyl. More preferably, each R 9 is independently selected from halogen and methyl. Even more preferably, each R 9 is independently selected from fluoro and methyl.
- R 8 and R 9 are each independently selected from halogen, Ci-C3alkyl, and Ci-C3alkoxy.
- R 8 and R 9 are each independently selected from halogen, methyl, ethyl, n- propyl, isopropyl, methoxy, ethoxy, or isopropoxy. More preferably, R 8 and R 9 are each independently selected from halogen, methyl, or methoxy. More preferably still, R 8 and R 9 are each independently selected from fluoro, chloro, methyl, or methoxy. In one set of embodiments, R 8 is fluoro.
- R 10 and R 11 are each independently selected from hydrogen and Ci-C3alkyl.
- R 10 and R 11 are each independently selected from hydrogen, methyl and ethyl. In one set of embodiments, R 10 and R 11 are both methyl.
- R 12 is halogen, cyano, Ci-C3alkyl, or Ci-C3alkoxy.
- R 12 is bromo, chloro, fluoro, cyano, methyl or methoxy.
- R 13 is hydrogen, Ci-C3alkyl, orCi-C3alkoxy.
- R 13 is hydrogen, methyl, or methoxy. More preferably, R 13 is hydrogen.
- X is O, N or S
- R 1 is phenyl optionally substituted with 1 or 2 groups, which may be the same or different, represented by R 7 ;
- R 2 is cyano, acetyl, propanoyl, 2-methylpropanoyl, 1-methoxyvinyl, 1-ethoxyvinyl, N-methoxy-C- methylcarbonimidoyl, N-ethoxy-C-methylcarbonimidoyl, phenyl, phenoxy, heteroaryl, wherein the heteroaryl moiety is a 5- or 6-membered aromatic monocyclic ring comprising 1 or 2 heteroatoms individually selected from N and O, or heterocyclyl, wherein the heterocyclyl moiety is a 5- or 6- membered non-aromatic monocyclic ring comprising 1 or 2 heteroatoms individually selected from N and O, and wherein the phenyl and phenoxy moieties may each be optionally substituted with a single group represented by R 8 ;
- R 3 is hydrogen or Ci-C3alkyl
- R 4 , R 5 , and R 6 are all hydrogen
- R 7 is halogen or Ci-C3haloalkoxy; and R 8 is halogen.
- X is O
- R 1 is phenyl optionally substituted with a single group represented by R 7 ;
- R 3 is hydrogen, methyl, or ethyl
- R 4 , R 5 , and R 6 are all hydrogen
- R 7 is halogen or Ci-C3haloalkoxy; and R 8 is halogen.
- X is O
- R 1 is phenyl optionally substituted with a single group represented by R 7 ;
- R 2 is cyano, acetyl, 1-methoxyvinyl, 1-ethoxyvinyl, N-methoxy-C-methylcarbonimidoyl, 4- fluorophenyl, 4-fluorophenoxy, or oxazol-2-yl;
- R 3 is hydrogen, methyl, or ethyl
- R 4 , R 5 , and R 6 are all hydrogen
- R 7 is halogen or Ci-C3haloalkoxy.
- X is O
- R 1 is phenyl optionally substituted with a single group represented by R 7 ;
- R 3 is hydrogen, methyl, or ethyl
- R 4 , R 5 , and R 6 are all hydrogen
- R 7 is Ci-C3haloalkoxy
- R 8 is fluoro, chloro, difluoromethyl, and trifluoromethyl.
- R 10 is hydrogen or methyl; and R 11 is methyl.
- X is O
- R 1 is phenyl optionally substituted with a single group represented by R 7 ;
- R 3 is hydrogen, methyl, or ethyl
- R 4 , R 5 , and R 6 are all hydrogen
- R 7 is chloro, methoxy, or trifluoromethoxy
- R 8 is fluoro, chloro, difluoromethyl, and trifluoromethyl.
- R 10 is hydrogen or methyl; and R 11 is methyl.
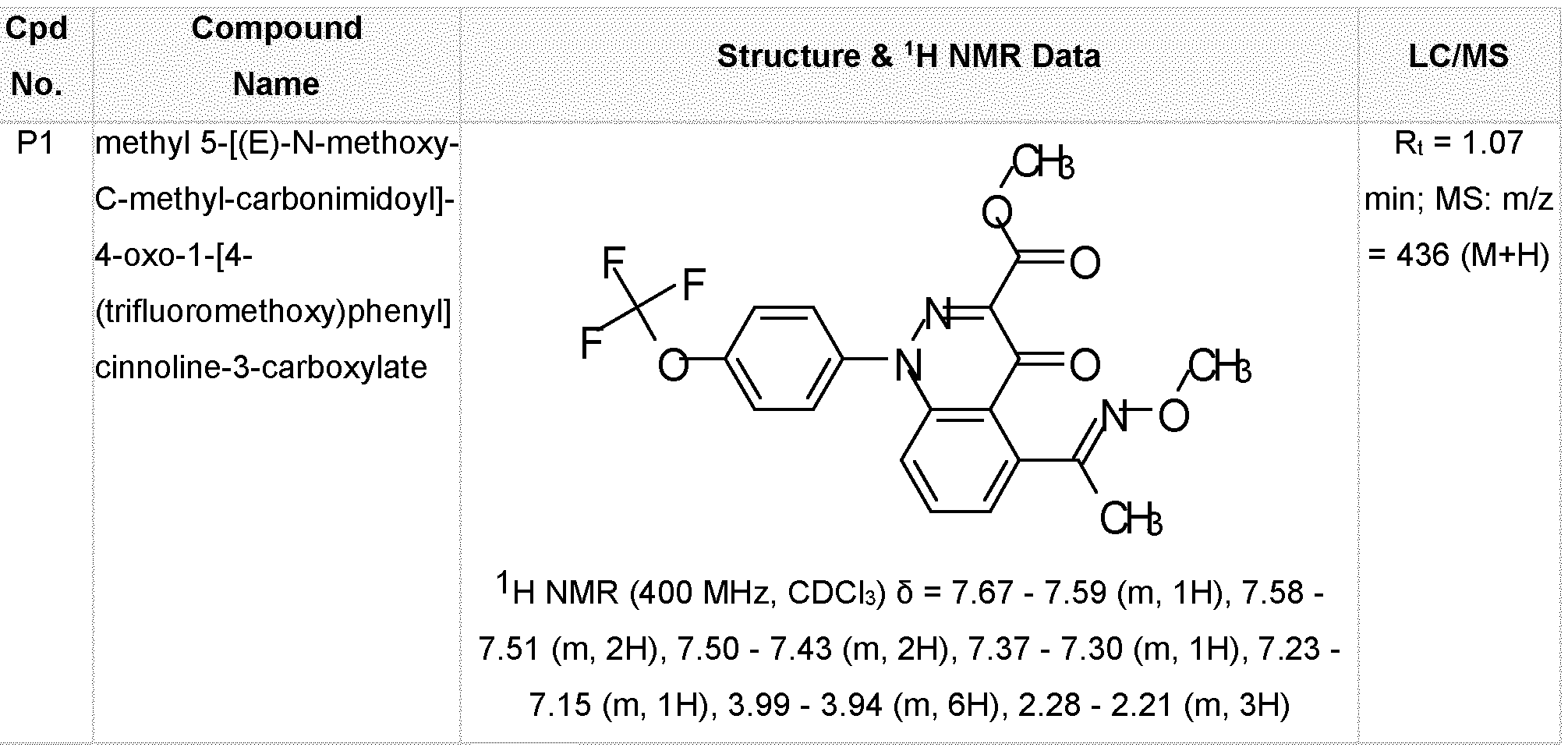
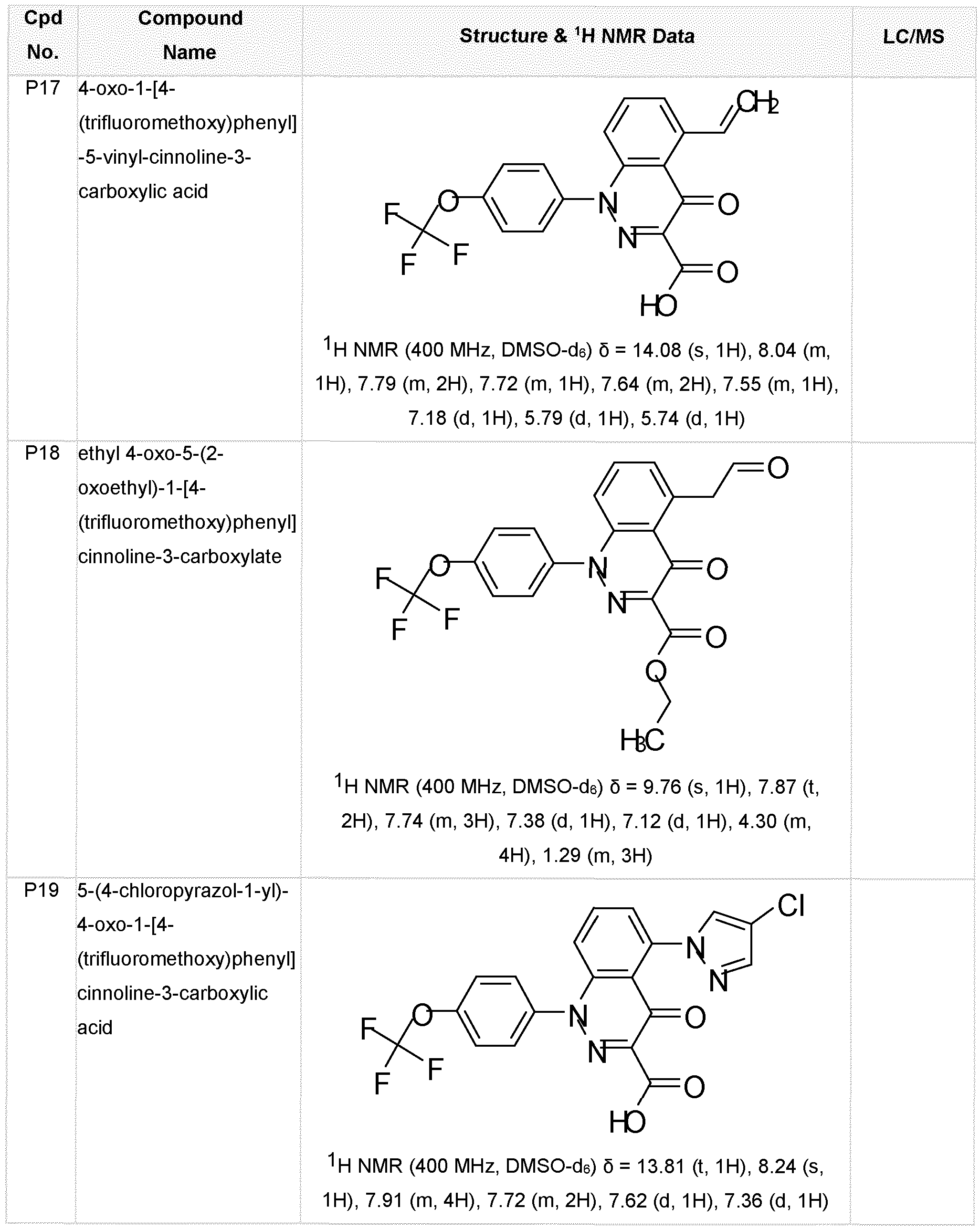
- the compound of Formula (I) is selected from: methyl 5-[(E)-N-methoxy-C-methyl-carbonimidoyl]-4-oxo-1-[4-(trifluoromethoxy)phenyl]cinnoline- 3-carboxylate (P1), 5-[(E)-N-methoxy-C-methyl-carbonimidoyl]-4-oxo-1-[4-
- the compound of Formula (I) is selected from: methyl 5-[(E)-N-methoxy-C-methyl-carbonimidoyl]-4-oxo-1-[4-(trifluoromethoxy)phenyl]cinnoline- 3-carboxylate (P1), 5-[(E)-N-methoxy-C-methyl-carbonimidoyl]-4-oxo-1-[4-
- the compound of Formula (I) is selected from: methyl 5-[(E)-N-methoxy-C-methyl-carbonimidoyl]-4-oxo-1-[4-(trifluoromethoxy)phenyl]cinnoline- 3-carboxylate (P1), 5-[(E)-N-methoxy-C-methyl-carbonimidoyl]-4-oxo-1-[4-
- a compound of Formula (I) wherein X is oxygen and R 3 is hydrogen may be prepared by hydrolysis of a compound of Formula (I) wherein X is oxygen and R 3 is not hydrogen, but any other R 3 group as defined above, with a suitable base (such as sodium hydroxide or lithium hydroxide), or with a suitable acid (such as trifluoroacetic acid, hydrochloric acid, formic acid or sulfuric acid), in a suitable solvent (such as methanol, ethanol, dichloromethane, chloroform, ethyl acetate ortetrahydrofuran), with an optional co-solvent (such as water).
- a suitable base such as sodium hydroxide or lithium hydroxide
- a suitable acid such as trifluoroacetic acid, hydrochloric acid, formic acid or sulfuric acid
- a suitable solvent such as methanol, ethanol, dichloromethane, chloroform, ethyl acetate ortetrahydrofuran
- an optional co-solvent
- Compounds of Formula (I) may be prepared from a compound Formula (B) wherein Y is F, Cl, Br or I.
- compounds of Formula (I) may be prepared by reaction with an appropriately substituted phenol or heterocyclyl alcohol under SnAr conditions in analogy to literature conditions. Typically the reaction is performed in the presence of a base (such as potassium carbonate), in an organic solvent (such as dimethylacetamide or N,N-dimethylformamide), at elevated temperature (such as 100°C to 170°C). This is shown in Scheme 2 above.
- a base such as potassium carbonate
- organic solvent such as dimethylacetamide or N,N-dimethylformamide
- Formula (B) Formula (I) Compounds of Formula (I) maybe additionally be prepared from a compound of Formula (B) wherein Y is Cl, Br or I.
- compounds of Formula (I) may be prepared in a Stille reaction by reaction with a stannane reagent in the presence of a palladium catalyst (such as dichlorobis(triphenylphosphine)palladium(ll), tetrakis(triphenylphosphine)palladium) or dichloro(1 ,1'-bis(diphenylphosphanyl)ferrocene)palladium(ll) dichloromethane adduct), with or without a base (such as triethylamine), in a suitable organic solvent (such as toluene, 1 ,4-dioxane or N,N-dimethylformamide), at an elevated reaction temperature (e.g. 120°
- a palladium catalyst such as dichlorobis(triphenylphosphin
- compounds of Formula (I) wherein R 2 is Ci-C6alkylcarbonyl may be prepared from compounds of Formula (I) wherein R 2 is C2-C6alkenyloxy by a hydrolysis reaction.
- the reaction is performed by treatment with aqueous acid (such as hydrochloric acid), optionally in a suitable organic solvent (such as acetone, 1 ,4-dioxane or tetrahydrofuran), and at a suitable temperature (20°C to 60°C). This is shown in Scheme 4 above.
- the reaction is performed with a compound with the formula H2NOR 10 as either the freebase or the hydrochloride salt, with or without the addition of a base (such as sodium acetate, pyridine or aqueous potassium hydroxide), in a suitable organic solvent (such as ethanol, dimethylsulfoxide, tetrahydrofuran, dimethylether or methanol) with or without additional water at elevated temperature.
- a base such as sodium acetate, pyridine or aqueous potassium hydroxide
- a suitable organic solvent such as ethanol, dimethylsulfoxide, tetrahydrofuran, dimethylether or methanol
- a compound of Formula (B) wherein Y is Br may be converted to a compound of Formula (I) wherein R 2 is a C-linked heterocycle (such as oxazol-2-yl), by reaction under Stille conditions with, for instance, a heterocyclic stannane in the presence of a catalyst (such as Pd- PEPPSI IPent), and a base (such as cesium fluoride), in a suitable solvent (such as 1 ,4-dioxane), at elevated temperature (for example 150°C).
- a catalyst such as Pd- PEPPSI IPent
- a base such as cesium fluoride
- Scheme 7 In another transformation, a compound of Formula (B) wherein Y is Br may be converted to a compound of Formula (I) wherein R 2 is alkyl or phenyl under Suzuki-Miyaura cross-coupling conditions in analogy to literature conditions.
- reaction is performed by reaction of a compound of Formula (B) with R 2 -boronic acid or boroxine in the presence of a suitable catalyst (such as dichlorobis(triphenylphosphine)palladium(ll), tetrakis(triphenylphosphine)palladium), tris(dibenzylideneacetone)dipalladium, or dichloro(1 ,T-bis(diphenylphosphanyl)ferrocene)palladium(ll) dichloromethane adduct), or palladium diacetate optionally with a ligand (such as 2- dicyclohexylphosphino-2',6'-dimethoxybiphenyl) in the presence of a base (such as potassium or cesium carbonate or tripotassium phosphate) in a suitable organic solvent (such as 1 ,4-dioxane, toluene or tetrahydrofuran) optionally in the presence of
- a compound of Formula (B) wherein Y is Br may be converted to a compound of Formula (I) wherein R 2 is nitrile under Negishi cross-coupling conditions in analogy to literature conditions.
- the reaction is performed by reaction of a compound of Formula (B) with dicyanozinc in the presence of a suitable catalyst (such as dichlorobis(triphenylphosphine)palladium(ll), tetrakis(triphenylphosphine)palladium), tris(dibenzylideneacetone)dipalladium, or dichloro(1 , 1 bis(diphenylphosphanyl)ferrocene)palladium(ll) dichloromethane adduct), or palladium diacetate optionally with a ligand (such as 2-dicyclohexylphosphino-2',6'-dimethoxybiphenyl) in a suitable organic solvent (such as dimethylformamide), at elevated temperature
- a base such as a metal hydride e.g. sodium hydride, or potassium carbonate
- a suitable solvent such as 1 ,4-dioxane, tetrahydrofuran or N,N-dimethylformamide
- a compound of Formula (C), wherein Y is Br and wherein LG ia a suitable leaving group (such as F, Cl or Br), may be prepared from reaction of b-keto esters of Formula (D) with an arene diazonium salt.
- the arene diazonium salts can be prepared in situ by diazotisation of anilines of Formula (E) with sodium nitrite in the presence of acid (such as hydrochloric acid), in water followed by reaction with compounds of Formula (D) in the presence of a suitable base (such as sodium or potassium acetate or potassium carbonate), in a suitable solvent (such as water, methanol or ethanol), at temperatures between 0°C and 25°C.
- acid such as hydrochloric acid
- a suitable base such as sodium or potassium acetate or potassium carbonate
- a suitable solvent such as water, methanol or ethanol
- a dicarbonyl compound of Formula (D) wherein Y is Br and wherein LG ia a suitable leaving group (such as F, Cl or Br), may be prepared from a methyl ketone compound of Formula (F) and a diester of Formula (G) via a Claisen condensation by treatment of the methyl ketone with a suitable base (such as potassium t-butoxide or sodium hydride), in a suitable solvent (such as tetrahydrofuran, N,N- dimethylformamide, toluene or 1 ,4-dioxane), followed by reaction of the mixture with a carbonate ester (such as dimethylcarbonate or diethylcarbonate), at temperatures between 0°C to 110°C.
- a suitable base such as potassium t-butoxide or sodium hydride
- a suitable solvent such as tetrahydrofuran, N,N- dimethylformamide, toluene or 1 ,4-dioxane
- the present invention still further provides a method of controlling weeds at a locus said method comprising application to the locus of a weed controlling amount of a composition comprising a compound of Formula (I).
- the present invention may further provide a method of selectively controlling weeds at a locus comprising useful (crop) plants and weeds, wherein the method comprises application to the locus of a weed controlling amount of a composition according to the present invention.
- Controlling means killing, reducing or retarding growth or preventing or reducing germination. It is noted that the compounds of the present invention show a much improved selectivity compared to know, structurally similar compounds. Generally the plants to be controlled are unwanted plants (weeds).
- Locus means the area in which the plants are growing or will grow. The application may be applied to the locus pre-emergence and/or postemergence of the crop plant. Some crop plants may be inherently tolerant to herbicidal effects of compounds of Formula (I).
- the rates of application of compounds of Formula (I) may vary within wide limits and depend on the nature of the soil, the method of application (pre- or post-emergence; seed dressing; application to the seed furrow; no tillage application etc.), the crop plant, the weed(s) to be controlled, the prevailing climatic conditions, and other factors governed by the method of application, the time of application and the target crop.
- the compounds of Formula I according to the invention are generally applied at a rate of from 10 to 2500 g/ha, especially from 25 to 1000 g/ha, more especially from 25 to 250 g/ha.
- the application is generally made by spraying the composition, typically by tractor mounted sprayer for large areas, but other methods such as dusting (for powders), drip or drench can also be used.
- useful plants is to be understood as also including useful plants that have been rendered tolerant to herbicides like bromoxynil or classes of herbicides such as, for example, 4- Hydroxyphenylpyruvate dioxygenase (HPPD) inhibitors, ALS inhibitors, for example primisulfuron, prosulfuron and trifloxysulfuron, 5-enol-pyrovyl-shikimate-3-phosphate-synthase (EPSPS) inhibitors, glutamine synthetase (GS) inhibitors or protoporphyrinogen-oxidase (PPO) inhibitors as a result of conventional methods of breeding or genetic engineering.
- HPPD 4- Hydroxyphenylpyruvate dioxygenase
- ALS inhibitors for example primisulfuron, prosulfuron and trifloxysulfuron
- 5-enol-pyrovyl-shikimate-3-phosphate-synthase (EPSPS) inhibitors glutamine syntheta
- An example of a crop that has been rendered tolerant to imidazolinones, e.g. imazamox, by conventional methods of breeding (mutagenesis) is Clearfield® summer rape (Canola).
- crops that have been rendered tolerant to herbicides or classes of herbicides by genetic engineering methods include glyphosate- and glufosinate-resistant maize varieties commercially available under the trade names RoundupReady®, Herculex I® and LibertyLink®.
- useful plants is to be understood as also including useful plants which have been so transformed by the use of recombinant DNA techniques that they are capable of synthesising one or more selectively acting toxins, such as are known, for example, from toxin-producing bacteria, especially those of the genus Bacillus.
- YieldGard® (maize variety that expresses a CrylA(b) toxin); YieldGard Rootworm® (maize variety that expresses a CrylllB(bl) toxin); YieldGard Plus® (maize variety that expresses a CrylA(b) and a Cryll IB(b1 ) toxin); Starlink® (maize variety that expresses a Cry9(c) toxin); Herculex I® (maize variety that expresses a CrylF(a2) toxin and the enzyme phosphinothricine N-acetyltransferase (PAT) to achieve tolerance to the herbicide glufosinate ammonium); NuCOTN 33B® (cotton variety that expresses a CrylA(c) toxin); Bollgard I® (cotton variety that expresses a CrylA(c) toxin); Bollgard II® (cotton variety that
- Plant crops or seed material thereof can be both resistant to herbicides and, at the same time, resistant to insect feeding (“stacked” transgenic events).
- seed can have the ability to express an insecticidal Cry3 protein while at the same time being tolerant to glyphosate.
- Crop plants are also to be understood to include those which are obtained by conventional methods of breeding or genetic engineering and contain so-called output traits (e.g. improved storage stability, higher nutritional value and improved flavour).
- output traits e.g. improved storage stability, higher nutritional value and improved flavour.
- the compounds of Formula (I) can be used to control unwanted plants (collectively, ‘weeds’).
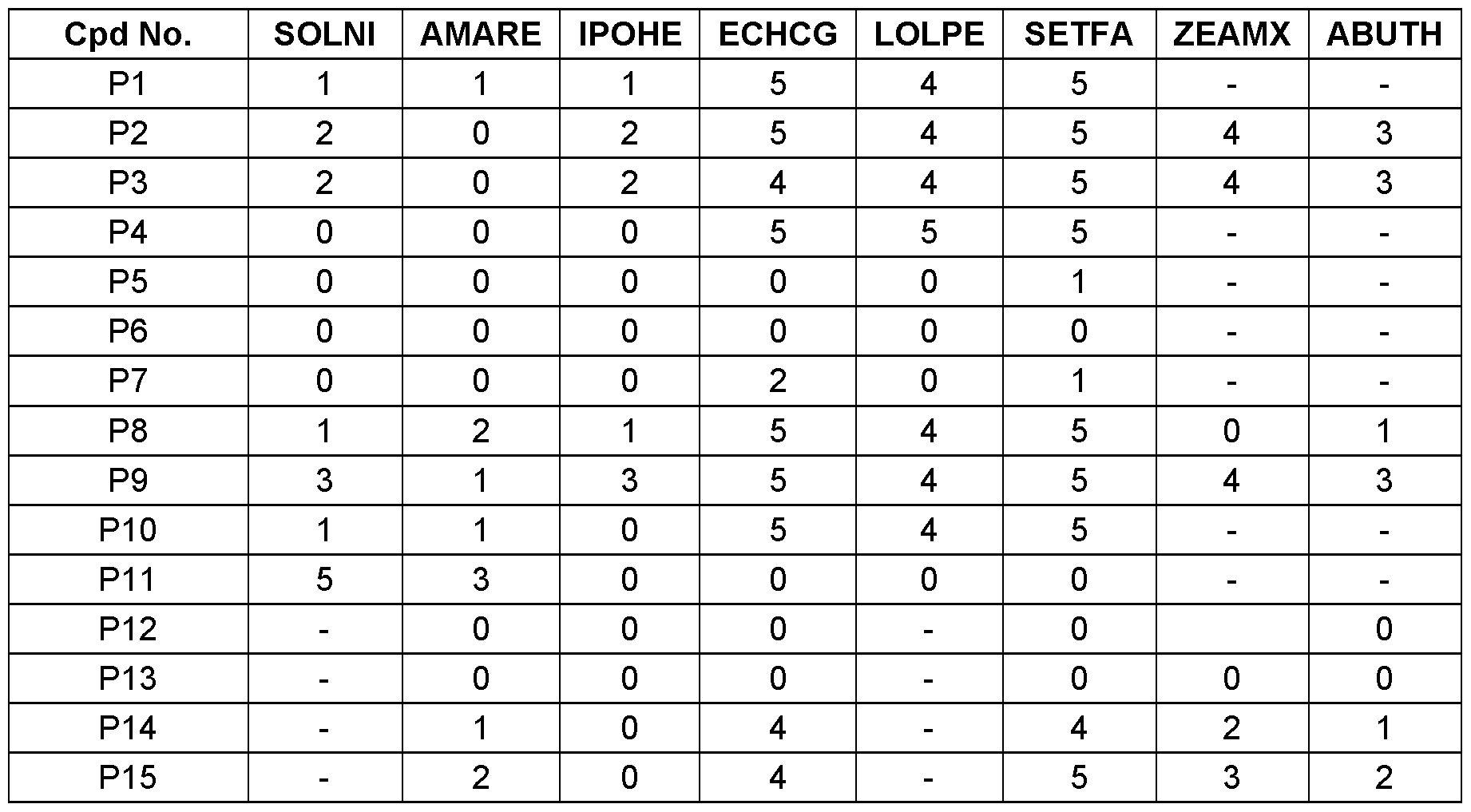
- weeds to be controlled may be both monocotyledonous species, for example Agrostis, Alopecurus, Avena, Brachiaria, Bromus, Cenchrus, Cyperus, Digitaha, Echinochloa, Eleusine, Lolium, Monochoria, Rottboellia, Sagittaria, Scirpus, Setaria and Sorghum, and dicotyledonous species, for example Abutilon, Amaranthus, Ambrosia, Chenopodium, Chrysanthemum, Conyza, Galium, Ipomoea, Nasturtium, Sida, Sinapis, Solanum, Stellaria, Veronica, Viola andXanthium.
- Agrostis Alopecurus
- Avena Brachiaria
- Bromus Cenchrus
- Cyperus Digitaha
- Echinochloa Eleusine
- Lolium Monochori
- Compounds of Formula (I) may be used in unmodified form or, preferably, together with the adjuvants conventionally employed in the art of formulation to provide herbicidal compositions, using formulation adjuvants, such as carriers, solvents, and surface-active agents (SAA).
- formulation adjuvants such as carriers, solvents, and surface-active agents (SAA).
- SAA surface-active agents
- the invention therefore further provides a herbicidal composition, comprising at least one compound Formula (I) and an agriculturally acceptable carrier and optionally an adjuvant.
- An agricultural acceptable carrier is for example a carrier that is suitable for agricultural use. Agricultural carriers are well known in the art.
- the herbicidal compositions generally comprise from 0.1 to 99 % by weight, especially from 0.1 to 95 % by weight, compounds of Formula I and from 1 to 99.9 % by weight of a formulation adjuvant which preferably includes from 0 to 25 % by weight of a surface-active substance.
- compositions can be chosen from a number of formulation types. These include an emulsion concentrate (EC), a suspension concentrate (SC), a suspo-emulsion (SE), a capsule suspension (CS), a water dispersible granule (WG), an emulsifiable granule (EG), an emulsion, water in oil (EO), an emulsion, oil in water (EW), a micro-emulsion (ME), an oil dispersion (OD), an oil miscible flowable (OF), an oil miscible liquid (OL), a soluble concentrate (SL), an ultra-low volume suspension (SU), an ultra-low volume liquid (UL), a technical concentrate (TK), a dispersible concentrate (DC), a soluble powder (SP), a wettable powder (WP) and a soluble granule (SG).
- formulation type chosen in any instance will depend upon the particular purpose envisaged and the physical, chemical, and biological properties of the compound of Formula (I).
- Soluble powders may be prepared by mixing a compound of Formula (I) with one or more water-soluble inorganic salts (such as sodium bicarbonate, sodium carbonate or magnesium sulphate) or one or more water-soluble organic solids (such as a polysaccharide) and, optionally, one or more wetting agents, one or more dispersing agents or a mixture of said agents to improve water dispersibility/solubility. The mixture is then ground to a fine powder. Similar compositions may also be granulated to form water soluble granules (SG).
- water-soluble inorganic salts such as sodium bicarbonate, sodium carbonate or magnesium sulphate
- water-soluble organic solids such as a polysaccharide
- WP Wettable powders
- WG Water dispersible granules
- Granules may be formed either by granulating a mixture of a compound of Formula (I) and one or more powdered solid diluents or carriers, or from pre-formed blank granules by absorbing a compound of Formula (I) (or a solution thereof, in a suitable agent) in a porous granular material (such as pumice, attapulgite clays, fuller's earth, kieselguhr, diatomaceous earths or ground corn cobs) or by adsorbing a compound of Formula (I) (ora solution thereof, in a suitable agent) on to a hard core material (such as sands, silicates, mineral carbonates, sulphates or phosphates) and drying if necessary.
- a hard core material such as sands, silicates, mineral carbonates, sulphates or phosphates
- Agents which are commonly used to aid absorption or adsorption include solvents (such as aliphatic and aromatic petroleum solvents, alcohols, ethers, ketones and esters) and sticking agents (such as polyvinyl acetates, polyvinyl alcohols, dextrins, sugars and vegetable oils).
- solvents such as aliphatic and aromatic petroleum solvents, alcohols, ethers, ketones and esters
- sticking agents such as polyvinyl acetates, polyvinyl alcohols, dextrins, sugars and vegetable oils.
- One or more other additives may also be included in granules (for example an emulsifying agent, wetting agent or dispersing agent).
- DC Dispersible Concentrates
- a compound of Formula (I) may be prepared by dissolving a compound of Formula (I) in water or an organic solvent, such as a ketone, alcohol or glycol ether.
- organic solvent such as a ketone, alcohol or glycol ether.
- surface active agent for example to improve water dilution or prevent crystallisation in a spray tank.
- Emulsifiable concentrates or oil-in-water emulsions (EW) may be prepared by dissolving a compound of Formula (I) in an organic solvent (optionally containing one or more wetting agents, one or more emulsifying agents or a mixture of said agents).
- Suitable organic solvents for use in ECs include aromatic hydrocarbons (such as alkylbenzenes or alkylnaphthalenes, exemplified by SOLVESSO 100, SOLVESSO 150 and SOLVESSO 200; SOLVESSO is a Registered Trade Mark), ketones (such as cyclohexanone or methylcyclohexanone) and alcohols (such as benzyl alcohol, furfuryl alcohol or butanol), N-alkylpyrrolidones (such as N-methylpyrrolidone or N-octylpyrrolidone), dimethyl amides of fatty acids (such as Cs-Cio fatty acid dimethylamide) and chlorinated hydrocarbons.
- An EC product may spontaneously emulsify on addition to water, to produce an emulsion with sufficient stability to allow spray application through appropriate equipment.
- Preparation of an EW involves obtaining a compound of Formula (I) either as a liquid (if it is not a liquid at room temperature, it may be melted at a reasonable temperature, typically below 70°C) or in solution (by dissolving it in an appropriate solvent) and then emulsifying the resultant liquid or solution into water containing one or more SAAs, under high shear, to produce an emulsion.
- Suitable solvents for use in EWs include vegetable oils, chlorinated hydrocarbons (such as chlorobenzenes), aromatic solvents (such as alkylbenzenes or alkylnaphthalenes) and other appropriate organic solvents which have a low solubility in water.
- Microemulsions may be prepared by mixing water with a blend of one or more solvents with one or more SAAs, to produce spontaneously a thermodynamically stable isotropic liquid formulation.
- a compound of Formula (I) is present initially in either the water or the solvent/SAA blend.
- Suitable solvents for use in MEs include those hereinbefore described for use in in ECs or in EWs.
- An ME may be either an oil-in-water or a water-in-oil system (which system is present may be determined by conductivity measurements) and may be suitable for mixing water-soluble and oil-soluble pesticides in the same formulation.
- An ME is suitable for dilution into water, either remaining as a microemulsion or forming a conventional oil-in-water emulsion.
- SC Suspension concentrates
- SCs may comprise aqueous or non-aqueous suspensions of finely divided insoluble solid particles of a compound of Formula (I).
- SCs may be prepared by ball or bead milling the solid compound of Formula (I) in a suitable medium, optionally with one or more dispersing agents, to produce a fine particle suspension of the compound.
- One or more wetting agents may be included in the composition and a suspending agent may be included to reduce the rate at which the particles settle.
- a compound of Formula (I) may be dry milled and added to water, containing agents hereinbefore described, to produce the desired end product.
- Aerosol formulations comprise a compound of Formula (I) and a suitable propellant (for example n-butane).
- a compound of Formula (I) may also be dissolved or dispersed in a suitable medium (for example water or a water miscible liquid, such as n-propanol) to provide compositions for use in non- pressurised, hand-actuated spray pumps.
- Capsule suspensions (CS) may be prepared in a manner similar to the preparation of EW formulations but with an additional polymerisation stage such that an aqueous dispersion of oil droplets is obtained, in which each oil droplet is encapsulated by a polymeric shell and contains a compound of Formula (I) and, optionally, a carrier or diluent therefor.
- the polymeric shell may be produced by either an interfacial polycondensation reaction or by a coacervation procedure.
- the compositions may provide for controlled release of the compound of Formula (I) and they may be used for seed treatment.
- a compound of Formula (I) may also be formulated in a biodegradable polymeric matrix to provide a slow, controlled release of the compound.
- the composition may include one or more additives to improve the biological performance of the composition, for example by improving wetting, retention or distribution on surfaces; resistance to rain on treated surfaces; or uptake or mobility of a compound of Formula (I).
- additives include surface active agents (SAAs), spray additives based on oils, for example certain mineral oils or natural plant oils (such as soy bean and rape seed oil), modified plant oils such as methylated rape seed oil (MRSO), and blends of these with other bio-enhancing adjuvants (ingredients which may aid or modify the action of a compound of Formula (I).
- wetting agents, dispersing agents and emulsifying agents may be SAAs of the cationic, anionic, amphoteric or non-ionic type.
- Suitable SAAs of the cationic type include quaternary ammonium compounds (for example cetyltrimethyl ammonium bromide), imidazolines and amine salts.
- Suitable anionic SAAs include alkali metals salts of fatty acids, salts of aliphatic monoesters of sulphuric acid (for example sodium lauryl sulphate), salts of sulphonated aromatic compounds (for example sodium dodecylbenzenesulphonate, calcium dodecylbenzenesulphonate, butylnaphthalene sulphonate and mixtures of sodium di-/sopropyl- and tri-/sopropyl-naphthalene sulphonates), ether sulphates, alcohol ether sulphates (for example sodium laureth-3-sulphate), ether carboxylates (for example sodium laureth-3-carboxylate), phosphate esters (products from the reaction between one or more fatty alcohols and phosphoric acid (predominately mono-esters) or phosphorus pentoxide (predominately di-esters), for example the reaction between lauryl alcohol and tetraphosphoric acid
- Suitable SAAs of the amphoteric type include betaines, propionates and glycinates.
- Suitable SAAs of the non-ionic type include condensation products of alkylene oxides, such as ethylene oxide, propylene oxide, butylene oxide or mixtures thereof, with fatty alcohols (such as oleyl alcohol or cetyl alcohol) or with alkylphenols (such as octylphenol, nonylphenol or octylcresol); partial esters derived from long chain fatty acids or hexitol anhydrides; condensation products of said partial esters with ethylene oxide; block polymers (comprising ethylene oxide and propylene oxide); alkanolamides; simple esters (for example fatty acid polyethylene glycol esters); amine oxides (for example lauryl dimethyl amine oxide); lecithins and sorbitans and esters thereof, alkyl polyglycosides and tristyrylphenols.
- alkylene oxides such as ethylene oxide, propylene oxide, butylene oxide or mixtures thereof
- fatty alcohols such as oleyl
- Suitable suspending agents include hydrophilic colloids (such as polysaccharides, polyvinylpyrrolidone or sodium carboxymethylcellulose) and swelling clays (such as bentonite or attapulgite).
- hydrophilic colloids such as polysaccharides, polyvinylpyrrolidone or sodium carboxymethylcellulose
- swelling clays such as bentonite or attapulgite.
- the compounds of present invention can also be used in mixture with one or more additional herbicides and/or plant growth regulators.
- additional herbicides or plant growth regulators include acetochlor, acifluorfen (including acifluorfen- sodium), aclonifen, ametryn, amicarbazone, aminopyralid, aminotriazole, atrazine, beflubutamid- M, benquitrione, bensulfuron (including bensulfuron-methyl), bentazone, bicyclopyrone, bilanafos, bipyrazone, bispyribac-sodium, bixlozone, bromacil, bromoxynil, butachlor, butafenacil, carfentrazone (including carfentrazone-ethyl), cloransulam (including cloransulam- methyl), chlorimuron (including chlorimuron-ethyl), chlorotoluron, chlorsulfuron, cinmethylin, clacyfos, clethodim, clodinafop (including clodinafop
- the compounds or mixtures of the present invention can also be used in combination with one or more herbicide safeners.
- herbicide safeners include benoxacor, cloquintocet (including cloquintocet-mexyl), cyprosulfamide, dichlormid, fenchlorazole (including fenchlorazole- ethyl), fenclorim, fluxofenim, furilazole, isoxadifen (including isoxadifen- ethyl), mefenpyr (including mefenpyr-diethyl), metcamifen and oxabetrinil.
- the mixing partners of the compound of Formula (I) may also be in the form of esters or salts, as mentioned e.g. in The Pesticide Manual, Sixteenth Edition, British Crop Protection Council, 2012.
- the mixing ratio of the compound of Formula (I) to the mixing partner is preferably from 1 : 100 to 1000:1 .
- mixtures can advantageously be used in the above-mentioned formulations (in which case "active ingredient” relates to the respective mixture of compound of Formula (I) with the mixing partner).
- the compounds or mixtures of the present invention can also be used in combination with one or more herbicide safeners.
- herbicide safeners include benoxacor, cloquintocet (including cloquintocet-mexyl), cyprosulfamide, dichlormid, fenchlorazole (including fenchlorazole-ethyl), fenclorim, fluxofenim, furilazole, isoxadifen (including isoxadifen-ethyl), mefenpyr (including mefenpyr- diethyl), metcamifen and oxabetrinil.
- mixtures of a compound of Formula (I) with cyprosulfamide, isoxadifen-ethyl, cloquintocet-mexyl and/or metcamifen are particularly preferred.
- the safeners of the compound of Formula (I) may also be in the form of esters or salts, as mentioned e.g. in The Pesticide Manual, 16 th Edition (BCPC), 2012.
- the reference to cloquintocet-mexyl also applies to a lithium, sodium, potassium, calcium, magnesium, aluminium, iron, ammonium, quaternary ammonium, sulfonium or phosphonium salt thereof as disclosed in WO 02/34048.
- the mixing ratio of compound of Formula (I) to safener is from 100:1 to 1 :10, especially from 20:1 to 1 :1.
- the compounds of Formula (I) are normally used in the form of agrochemical compositions and can be applied to the crop area or plant to be treated, simultaneously or in succession with further compounds.
- further compounds can be e.g. fertilizers or micronutrient donors or other preparations, which influence the growth of plants. They can also be selective herbicides or non- selective herbicides as well as insecticides, fungicides, bactericides, nematicides, molluscicides or mixtures of several of these preparations, if desired together with further carriers, surfactants or application promoting adjuvants customarily employed in the art of formulation.
- locus means fields in or on which plants are growing, or where seeds of cultivated plants are sown, or where seed will be placed into the soil. It includes soil, seeds, and seedlings, as well as established vegetation.
- plants refers to all physical parts of a plant, including seeds, seedlings, saplings, roots, tubers, stems, stalks, foliage, and fruits.
- plant propagation material is understood to denote generative parts of the plant, such as seeds, which can be used for the multiplication of the latter, and vegetative material, such as cuttings or tubers, for example potatoes. There may be mentioned for example seeds (in the strict sense), roots, fruits, tubers, bulbs, rhizomes, and parts of plants. Germinated plants and young plants which are to be transplanted after germination or after emergence from the soil, may also be mentioned. These young plants may be protected before transplantation by a total or partial treatment by immersion. Preferably “plant propagation material” is understood to denote seeds.
- Pesticidal agents referred to herein using their common name are known, for example, from “The Pesticide Manual”, 15th Ed., British Crop Protection Council 2009.
- the compounds of formula (I) may be used in unmodified form or, preferably, together with the adjuvants conventionally employed in the art of formulation. To this end, they may be conveniently formulated in known manner to emulsifiable concentrates, coatable pastes, directly sprayable or dilutable solutions or suspensions, dilute emulsions, wettable powders, soluble powders, dusts, granulates, and also encapsulations e.g. in polymeric substances. As with the type of the compositions, the methods of application, such as spraying, atomising, dusting, scattering, coating or pouring, are chosen in accordance with the intended objectives and the prevailing circumstances. The compositions may also contain further adjuvants such as stabilizers, antifoams, viscosity regulators, binders or tackifiers as well as fertilizers, micronutrient donors or other formulations for obtaining special effects.
- Suitable carriers and adjuvants can be solid or liquid and are substances useful in formulation technology, e.g. natural or regenerated mineral substances, solvents, dispersants, wetting agents, tackifiers, thickeners, binders, or fertilizers.
- Such carriers are for example described in WO 97/33890.
- the compounds of Formula (I) are normally used in the form of compositions and can be applied to the crop area or plant to be treated, simultaneously or in succession with further compounds.
- further compounds can be, e.g., fertilizers or micronutrient donors or other preparations, which influence the growth of plants. They can also be selective herbicides or non-selective herbicides as well as insecticides, fungicides, bactericides, nematicides, molluscicides or mixtures of several of these preparations, if desired together with further carriers, surfactants or application promoting adjuvants customarily employed in the art of formulation.
- the compound of Formula (I) may be the sole active ingredient of a composition or it may be admixed with one or more additional active ingredients such as a pesticide, fungicide, synergist, herbicide or plant growth regulator where appropriate.
- An additional active ingredient may, in some cases, result in unexpected synergistic activities.
- the formulations include from 0.01 to 90% by weight of active agent, from 0 to 20% agriculturally acceptable surfactant and 10 to 99.99% solid or liquid formulation inerts and adjuvant(s), the active agent consisting of at least the compound of formula (I) together with component (B) and (C), and optionally other active agents, particularly microbiocides or conservatives or the like.
- Concentrated forms of compositions generally contain in between about 2 and 80%, preferably between about 5 and 70% by weight of active agent.
- Application forms of formulation may for example contain from 0.01 to 20% by weight, preferably from 0.01 to 5% by weight of active agent.
- commercial products will preferably be formulated as concentrates, the end user will normally employ diluted formulations.
- Table A-1 provides 48 compounds A-1 .001 to A.1 .048 of Formula (I) wherein X is oxygen, and R 1 , R 2 , R 3 , R 4 , R 5 , and R 6 are as defined in Table 1 .
- Formulation Examples
- Wettable powders a) b) c) active ingredient [compound of formula (I)] 25 % 50 % 75 % sodium lignosulfonate 5 % 5 % sodium lauryl sulfate 3 % 5 % sodium diisobutylnaphthalenesulfonate 6 % 10 % phenol polyethylene glycol ether 2 % (7-8 mol of ethylene oxide) highly dispersed silicic acid 5 % 10 % 10 % Kaolin 62 % 27 %
- the active ingredient is thoroughly mixed with the adjuvants and the mixture is thoroughly ground in a suitable mill, affording wettable powders that can be diluted with waterto give suspensions of the desired concentration.
- Powders for dry seed treatment a) b) c) active ingredient [compound of formula (I)] 25 % 50 % 75 % light mineral oil 5 % 5 % 5 % highly dispersed silicic acid 5 % 5 %
- the active ingredient is thoroughly mixed with the adjuvants and the mixture is thoroughly ground in a suitable mill, affording powders that can be used directly for seed treatment.
- Emulsifiable concentrate active ingredient [compound of formula (I)] 10 % octylphenol polyethylene glycol ether 3 %
- Emulsions of any required dilution which can be used in plant protection, can be obtained from this concentrate by dilution with water.
- Active ingredient [compound of formula (I)] 5 % 6 % 4 % talcum 95 %
- Kaolin 94 % mineral filler 96 % Ready-for-use dusts are obtained by mixing the active ingredient with the carrier and grinding the mixture in a suitable mill. Such powders can also be used for dry dressings for seed. Extruder granules
- Active ingredient 15 % sodium lignosulfonate 2 % carboxymethylcellulose 1 %
- the active ingredient is mixed and ground with the adjuvants, and the mixture is moistened with water.
- the mixture is extruded and then dried in a stream of air.
- Active ingredient 8 % polyethylene glycol (mol. wt. 200) 3 %
- the finely ground active ingredient is uniformly applied, in a mixer, to the kaolin moistened with polyethylene glycol. Non-dusty coated granules are obtained in this manner.
- Suspension concentrate active ingredient [compound of formula (I)] 40 % propylene glycol 10 % nonylphenol polyethylene glycol ether (15 mol of ethylene oxide) 6 %
- the finely ground active ingredient is intimately mixed with the adjuvants, giving a suspension concentrate from which suspensions of any desired dilution can be obtained by dilution with water.
- a suspension concentrate from which suspensions of any desired dilution can be obtained by dilution with water.
- living plants as well as plant propagation material can be treated and protected against infestation by microorganisms, by spraying, pouring or immersion.
- Flowable concentrate for seed treatment active ingredient [compound of formula (I)] 40 % propylene glycol 5 % copolymer butanol PO/EO 2 % tristyrenephenole with 10-20 moles EO 2 %
- Silicone oil (in the form of a 75 % emulsion in water) 0.2 %
- the finely ground active ingredient is intimately mixed with the adjuvants, giving a suspension concentrate from which suspensions of any desired dilution can be obtained by dilution with water.
- a suspension concentrate from which suspensions of any desired dilution can be obtained by dilution with water.
- living plants as well as plant propagation material can be treated and protected against infestation by microorganisms, by spraying, pouring or immersion.
- 28 parts of a combination of the compound of formula (I) are mixed with 2 parts of an aromatic solvent and 7 parts of toluene diisocyanate/polymethylene-polyphenylisocyanate-mixture (8:1).
- This mixture is emulsified in a mixture of 1 .2 parts of polyvinyl alcohol, 0.05 parts of a defoamer and 51 .6 parts of water until the desired particle size is achieved.
- To this emulsion a mixture of 2.8 parts 1 ,6-diaminohexane in 5.3 parts of water is added. The mixture is agitated until the polymerization reaction is completed.
- the obtained capsule suspension is stabilized by adding 0.25 parts of a thickener and 3 parts of a dispersing agent.
- the capsule suspension formulation contains 28% of the active ingredients.
- the medium capsule diameter is 8-15 microns.
- the resulting formulation is applied to seeds as an aqueous suspension in an apparatus suitable for that purpose.
- Step 1 Synthesis of methyl 3-(2-bromo-6-fluoro-phenyl)-3-oxo-propanoate
- Step 2 Synthesis of methyl (2E)-3-(2-bromo-6-fluoro-phenyl)-3-oxo-2-[[4-(trifluoromethoxy) phenyl]hydrazono]propanoate
- 4-(trifluoromethoxy)aniline (2.12 g, 12.0 mmol) in hydrochloric acid (10.0 ml_, 60.1 mmol, 6 mol/L) at 0°C was added dropwise a solution of sodium nitrite (0.921 g, 13.2 mmol) in water (2.4 ml_, 12.0 mmol).
- the reaction mixture was stirred for 30 minutes at 0°C before being added portionwise to a suspension of methyl 3-(2-bromo-6-fluoro-phenyl)-3-oxo-propanoate (3.34 g, 12.0 mmol) and potassium acetate (6.0 g, 60.1 mmol) in water (2.4 ml_) resulting in formation of a yellow solid.
- the reaction mixture was stirred for 45 minutes and then allowed to warm to room temperature. After 90 minutes, the reaction mixture was filtered and the solid was collected by filtration to give the desired product as a yellow solid (3.5 g, 7.6 mmol, 64%).
- Step 3 Synthesis of methyl 5-bromo-4-oxo-1-[4-(trifluoromethoxy)phenyl]cinnoline-3- carboxylate
- reaction mixture was evaporated to dryness under reduced pressure to afford a brown gum which was purified by flash chromatography on silica gel using a gradient of 5-100% ethyl acetate in cyclohexane as eluent to give the desired product as an off-white solid (0.43 g, 0.99 mmol, 70%).
- Step 5 Synthesis of methyl 5-acetyl-4-oxo-1-[4-(trifluoromethoxy)phenyl]cinnoline-3- carboxylaxylate (Compound P9)
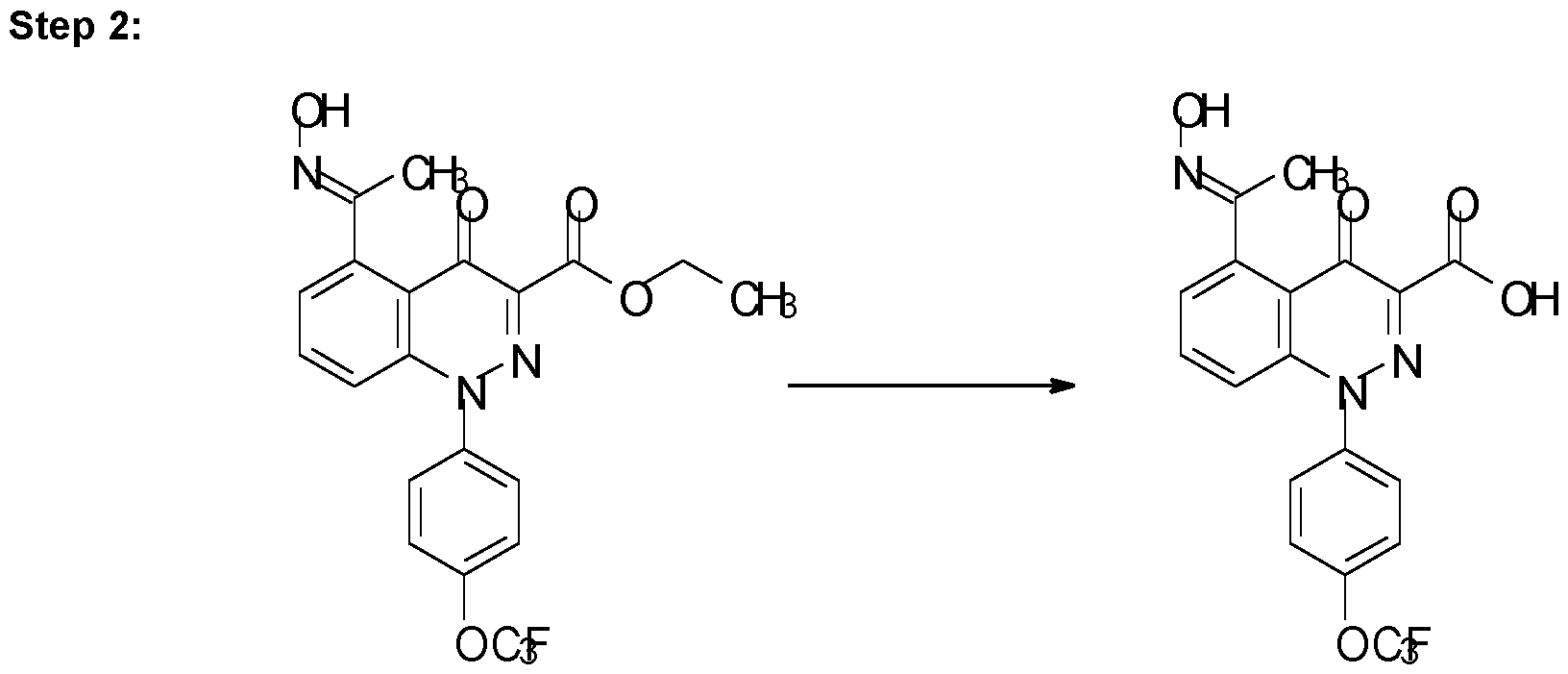
- Step 6 Synthesis of methyl 5-(N-methoxy-C-methyl-carbonimidoyl)-4-oxo-1-[4-(trifluoro methoxy)phenyl]cinnoline-3-carboxylate (Compound P1)
- the cooled reaction mixture was diluted with 2M aqueous hydrochloric acid and the precipitated solid was collected by filtration.
- the solid was purified by flash chromatography on silica gel using a gradient of 5-100% ethyl acetate in cyclohexane as eluent to give the desired product as a white solid (0.17 g, 0.39 mmol, 34%).
- Step 7 Synthesis of 5-(N-methoxy-C-methyl-carbonimidoyl)-4-oxo-1-[4-(trifluoromethoxy) phenyl]cinnoline-3-carboxylic acid (Compound P2)
- reaction mixture was diluted with 2M hydrochloric acid and extracted with ethyl acetate. The combined organics were concentrated to dryness under reduced pressure. The crude residue was purified by mass-directed reverse phase HPLC and then triturated with diethyl ether to give the desired product as a pale-yellow powder (0.026 g, 0.063 mmol, 14%).
- Example 4 Synthesis of methyl 5-(1-methoxyvinyl)-4-oxo-1-[4-(trifluoromethoxy)phenyl] cinnoline-3-carboxylate (Compound P4) To dichlorobis(triphenylphosphine)palladium(ll) (0.024 g, 0.034 mmol) was added a solution of methyl 5-bromo-4-oxo-1-[4-(trifluoromethoxy)phenyl]cinnoline-3-carboxylate (0.30 g, 0.68 mmol) in toluene (5 ml_).
- the mixture was degassed with nitrogen for 5 minutes after which tributyl(1- methoxyvinyl)stannane (0.71 g, 2.03 mmol) was added.
- the reaction mixture was heated under microwave irradiation at 120°C for 45 minutes.
- the cooled reaction mixture was filtered through diatomaceous earth and the filtrated was concentrated to dryness under reduced pressure.
- the crude residue was purified by flash chromatography on silica gel using a gradient of 5 - 100% ethyl acetate in cyclohexane as eluent to give the desired product as an off-white solid (0.086 g, 0.20 mmol, 30%).
- Step 1 Synthesis of ethyl 3-(2,6-difluorophenyl)-3-oxo-propanoate
- the reaction mixture was evaporated under reduced pressure and azeotroped with toluene.
- the residue was suspended in ethyl acetate (50 ml_) and 2M aqueous hydrochloric acid.
- the phases were separated and the aqueous was re-extracted twice with ethyl acetate.
- the combined organic extracts were dried over magnesium sulfate and evaporated to dryness under reduced pressure to give the crude desired product (mixture of tautomers) as a pale-yellow liquid (4.5 g, 20 mmol).
- Step 2 Synthesis of ethyl (2E)-2-[(4-chlorophenyl)hydrazono]-3-(2,6-difluorophenyl)-3-oxo- propanoate
- Step 3 Synthesis of ethyl 1-(4-chlorophenyl)-5-fluoro-4-oxo-cinnoline-3-carboxylate
- Step 4 Synthesis of 1-(4-chlorophenyl)-5-fluoro-4-oxo-cinnoline-3-carboxylic acid
- Step 5 Synthesis of 1-(4-chlorophenyl)-5-(4-fluorophenoxy)-4-oxo-cinnoline-3-carboxylic acid
- Example 6 Synthesis of methyl 5-cyano-4-oxo-1-[4-(trifluoromethoxy)phenyl]cinnoline-3- carboxylate and 5-cyano-4-oxo-1-[4-(trifluoromethoxy)phenyl]cinnoline-3-carboxylic acid (Compounds 1.017 and 1.018)
- Example 8 Synthesis of ethyl 5-[(E)-N-hydroxy-C-methyl-carbonimidoyl]-4-oxo-1-[4- (trifluoromethoxy)phenyl]cinnoline-3-carboxylate (compound P14) and 5-[(E)-N-hydroxy-C- methyl-carbonimidoyl]-4-oxo-1 -[4-(trifluoromethoxy)phenyl]cinnoline-3-carboxylic acid
- Step 1 To a stirring solution of ethyl 5-acetyl-4-oxo-1-[4-(trifluoromethoxy)phenyl]cinnoline-3-carboxylate (50.0 mg, 0.119 mmol) in methanol (10 ml_) and water (2.5 ml_) at 0°C was added sodium acetate (0.088 g, 1.07 mmol) and hydroxylamine chloride (0.074 g, 1.07 mmol). The reaction mixture was stirred for 5 minutes then heated at 80°C for 6 hours. The cooled reaction mixture was poured onto ice water, neutralized with 2M aqueous hydrochloric acid and filtered. The filtrate was evaporated to dryness under reduced pressure.
- Step 1 To a stirring solution of ethyl 5-acetyl-4-oxo-1-[4-(trifluoromethoxy)phenyl]cinnoline-3-carboxylate (1.00 g, 2.38 mmol) in tetrahydrofuran (25 ml_) was added a solution of sodium methoxide (0.129 g, 2.38 mmol) and methyl 2,2-difluoroacetate (0.393 g, 3.57 mmol) in methanol. The reaction mixture was stirred at room temperature for 10 hours. The reaction mixture was quenched by addition of 1 M aqueous hydrochloric acid and extracted into ethyl acetate. The organic extracts were evaporated to dryness under reduced pressure to give crude product (0.900 g, 1 .91 mmol, 80%) as a brown solid.
- Step 2 To a stirring solution of 5-(4,4-difluoro-3-oxo-butanoyl)-4-oxo-1-[4-(trifluoromethoxy)phenyl]cinnoline-3- carboxylic acid (0.650 g, 1.38 mmol) in ethanol (5.30 ml_) was added 1 M aqueous sodium hydroxide solution (0.75 ml_) followed by hydroxylamine hydrochloride (0.144 g, 2.07 mmol). The reaction mixture was stirred at room temperature 2 hours.
- reaction mixture was diluted with brine, extracted into ethyl acetate and the combined organic extracts were evaporated to dryness under reduced pressure to give 5-[5-(difluoromethyl)-5-hydroxy-4H-isoxazol-3-yl]-4-oxo-1-[4-(trifluoromethoxy)phenyl]cinnoline- 3-carboxylic acid (0.600 g, 1.24 mmol, 90%).
- Example 10 Synthesis of ethyl 4-oxo-1-[4-(trifluoromethoxy)phenyl]-5-vinyl-cinnoline-3- carboxylate (Compound P16) and 4-oxo-1-[4-(trifluoromethoxy)phenyl]-5-vinyl-cinnoline-3- carboxylic acid (Compound P17)
- Step 1
- Step 1 To a solution of ethyl 5-fluoro-4-oxo-1-[4-(trifluoromethoxy)phenyl]cinnoline-3-carboxylate (0.500 g, 1 .26 mmol) in N,N-dimethylformamide (5 ml_) at 0°C and under argon was added potassium carbonate (124 mg, 1 .26 mmol) followed by 4-chloro-1 H-pyrazole (0.194 g, 1 .89 mmol). The reaction mixture was stirred at 110°C for 5 hours. The cooled reaction mixture was poured into water and extracted into ethyl acetate.
- Step 2 To a stirring solution of ethyl 5-(4-chloro-2H-pyrrol-2-yl)-4-oxo-1-[4-(trifluoromethoxy)phenyl]cinnoline- 3-carboxylate (180 mg, 0.358 mmol) in a mixture of tetrahydrofuran (5 ml_) and water (1.5 ml_) at room temperature was added lithium hydroxide monohydrate (0.060 g, 1 .43 mmol). The reaction mixture was stirred at room temperature for 2 hours. The reaction mixture was acidified with 2M aqueous hydrochloric acid (2 ml_) and extracted into dichloromethane.
- 2M aqueous hydrochloric acid 2 ml_
- reaction mixture was degassed under argon for 2 minutes then heated at 100°C for 5 hours in a sealed tube.
- the cooled reaction mixture was filtered through diatomaceous earth and the filtrate was evaporated to dryness under reduced pressure.
- the crude residue was purified by flash chromatography on silica gel using a gradient of 20-30% ethyl acetate in hexanes as eluent to give ethyl 5-(cyanomethyl)- 4-oxo-1-[4-(trifluoromethoxy)phenyl]cinnoline-3-carboxylate (0.050 g, 0.120 mmol, 61%).
- Example 15 Synthesis of ethyl 5-nitro-4-oxo-1-[4-(trifluoromethoxy)phenyl]cinnoline-3- carboxylate (Compound P39) and 5-nitro-4-oxo-1-[4-(trifluoromethoxy)phenyl]cinnoline-3- carboxylic acid (Compound P40)
- Step 1
- reaction mixture was stirred at 0°C for 2 hours then allowed to warm to room temperature.
- the reaction mixture was diluted with water and extracted into ethyl acetate (x2).
- the combined organic extracts were washed with water and brine, dried over sodium sulfate, filtered and evaporated to dryness under reduced pressure to give crude ethyl (2Z)-3-(2-fluoro-6-nitro-phenyl)- 3-oxo-2-[[4-(trifluoromethoxy)phenyl]hydrazono]propanoate (5.00 g, 11.3 mmol, 63%) as a red liquid.
- Seeds of a variety of test species are sown in standard soil in pots ( Amaranthus retoflexus (AMARE), Solanum nigrum (SOLNI), Setaria faberi (SETFA), Lolium perenne (LOLPE), Echinochloa crus-galli (ECHCG), Ipomoea hederacea (IPOHE)).
- AMARE Amaranthus retoflexus
- SOLNI Solanum nigrum
- SETFA Setaria faberi
- LELPE Lolium perenne
- EHCG Echinochloa crus-galli
- IPHE Ipomoea hederacea
- the plants After 8 days cultivation under controlled conditions in a glasshouse (at 24 °C /16 °C, day/night; 14 hours light; 65 % humidity), the plants are sprayed with an aqueous spray solution derived from the formulation of the technical active ingredient in acetone / water (50:50) solution containing 0.5% Tween 20 (polyoxyethelyene sorbitan monolaurate, CAS RN 9005-64-5). Compounds are applied at 1000 g/ha unless otherwise stated. The test plants are then grown in a glasshouse under controlled conditions in a glasshouse (at 24 °C/16 °C, day/night; 14 hours light; 65 % humidity) and watered twice daily. After 13 days the test is evaluated for the percentage damage caused to the plant.
Landscapes
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- General Health & Medical Sciences (AREA)
- Pest Control & Pesticides (AREA)
- Plant Pathology (AREA)
- Wood Science & Technology (AREA)
- Zoology (AREA)
- Environmental Sciences (AREA)
- Dentistry (AREA)
- Agronomy & Crop Science (AREA)
- General Chemical & Material Sciences (AREA)
- Toxicology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Plural Heterocyclic Compounds (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
- Heterocyclic Carbon Compounds Containing A Hetero Ring Having Nitrogen And Oxygen As The Only Ring Hetero Atoms (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
Abstract
Description
Claims
Priority Applications (7)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CA3178605A CA3178605A1 (en) | 2020-05-19 | 2021-05-14 | Herbicidal cinnoline derivatives |
| AU2021276067A AU2021276067A1 (en) | 2020-05-19 | 2021-05-14 | Herbicidal cinnoline derivatives |
| US17/999,249 US20230303498A1 (en) | 2020-05-19 | 2021-05-14 | Herbicidal cinnoline derivatives |
| EP21726383.9A EP4152932A1 (en) | 2020-05-19 | 2021-05-14 | Herbicidal cinnoline derivatives |
| JP2022570595A JP7770340B2 (en) | 2020-05-19 | 2021-05-14 | Herbicidal shinorine derivatives |
| BR112022023491A BR112022023491A2 (en) | 2020-05-19 | 2021-05-14 | HERBICIDIAL CINOLINE DERIVATIVES |
| CN202180047977.7A CN115802892B (en) | 2020-05-19 | 2021-05-14 | Herbicidal cinnoline derivatives |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| GBGB2007419.1A GB202007419D0 (en) | 2020-05-19 | 2020-05-19 | Herbicidal derivatives |
| GB2007419.1 | 2020-05-19 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2021233786A1 true WO2021233786A1 (en) | 2021-11-25 |
Family
ID=71135198
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2021/062884 Ceased WO2021233786A1 (en) | 2020-05-19 | 2021-05-14 | Herbicidal cinnoline derivatives |
Country Status (11)
| Country | Link |
|---|---|
| US (1) | US20230303498A1 (en) |
| EP (1) | EP4152932A1 (en) |
| JP (1) | JP7770340B2 (en) |
| CN (1) | CN115802892B (en) |
| AR (1) | AR122103A1 (en) |
| AU (1) | AU2021276067A1 (en) |
| BR (1) | BR112022023491A2 (en) |
| CA (1) | CA3178605A1 (en) |
| GB (1) | GB202007419D0 (en) |
| UY (1) | UY39218A (en) |
| WO (1) | WO2021233786A1 (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2023088921A1 (en) | 2021-11-18 | 2023-05-25 | Syngenta Crop Protection Ag | Herbicidal derivatives |
| WO2024160989A1 (en) | 2023-02-03 | 2024-08-08 | Syngenta Crop Protection Ag | Herbicide resistant plants |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2026014533A1 (en) * | 2024-07-11 | 2026-01-15 | 住友化学株式会社 | Cinnoline derivative and composition containing same |
Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4729782A (en) * | 1983-09-14 | 1988-03-08 | Lafarge Coppee | Pollen suppressant comprising a fused pyridazinone |
| EP0273325A2 (en) | 1986-12-25 | 1988-07-06 | Sumitomo Chemical Company, Limited | Cinnoline derivative, process for preparing the same and herbicidal composition containing the same |
| EP0274717A2 (en) | 1986-12-26 | 1988-07-20 | Sumitomo Chemical Company, Limited | Plant male sterilant |
| EP0320793A2 (en) * | 1987-12-17 | 1989-06-21 | Sumitomo Chemical Company, Limited | Cinnoline derivative, process for preparing the same and herbicidal composition containing the same |
| US5129939A (en) * | 1988-09-13 | 1992-07-14 | Orsan | Pollen suppressant comprising a 5-oxy- or amino-substituted cinnoline |
| US5183891A (en) | 1989-11-09 | 1993-02-02 | Orsan | Method for the preparation of substituted 1,4-dihydro-4-oxo-cinnoline-3-carboxylic acid, esters and salts thereof, and intermediates used in their preparation |
| WO1997033890A1 (en) | 1996-03-11 | 1997-09-18 | Novartis Ag | Pyrimidin-4-one derivatives as pesticide |
| WO2002034048A1 (en) | 2000-10-23 | 2002-05-02 | Syngenta Participations Ag | Agrochemical compositions with quinoline safeners |
Family Cites Families (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4604134A (en) * | 1983-09-14 | 1986-08-05 | Lafarge Coppee | Pollen suppressant comprising a fused pyridazine |
| US4756740A (en) * | 1983-09-14 | 1988-07-12 | Lafarge Coppee | Pollen suppressant comprising a fused pyridazine |
| EP0197226A1 (en) * | 1985-04-03 | 1986-10-15 | Lafarge Coppee | Chemical hybridization agents comprising derivatives of cinnoline-3-carboxylic acids, and their application |
| JPS63264464A (en) * | 1986-12-25 | 1988-11-01 | Sumitomo Chem Co Ltd | Cinnoline derivative, production thereof and herbicide comprising said derivative as active ingredient |
| JPS63264405A (en) * | 1986-12-26 | 1988-11-01 | Sumitomo Chem Co Ltd | Male sterilization agent for plant |
| JPH01250362A (en) * | 1987-12-17 | 1989-10-05 | Sumitomo Chem Co Ltd | Cinnoline derivative, its production and herbicide containing the same as an active ingredient |
| US5332716A (en) * | 1988-09-13 | 1994-07-26 | Jeffrey Labovitz | Pollen suppressant comprising a 5-oxy- or amino-substituted cinnoline |
-
2020
- 2020-05-19 GB GBGB2007419.1A patent/GB202007419D0/en not_active Ceased
-
2021
- 2021-05-14 WO PCT/EP2021/062884 patent/WO2021233786A1/en not_active Ceased
- 2021-05-14 BR BR112022023491A patent/BR112022023491A2/en unknown
- 2021-05-14 EP EP21726383.9A patent/EP4152932A1/en active Pending
- 2021-05-14 AU AU2021276067A patent/AU2021276067A1/en active Pending
- 2021-05-14 JP JP2022570595A patent/JP7770340B2/en active Active
- 2021-05-14 AR ARP210101338A patent/AR122103A1/en unknown
- 2021-05-14 CN CN202180047977.7A patent/CN115802892B/en active Active
- 2021-05-14 US US17/999,249 patent/US20230303498A1/en active Pending
- 2021-05-14 UY UY0001039218A patent/UY39218A/en unknown
- 2021-05-14 CA CA3178605A patent/CA3178605A1/en active Pending
Patent Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4729782A (en) * | 1983-09-14 | 1988-03-08 | Lafarge Coppee | Pollen suppressant comprising a fused pyridazinone |
| EP0273325A2 (en) | 1986-12-25 | 1988-07-06 | Sumitomo Chemical Company, Limited | Cinnoline derivative, process for preparing the same and herbicidal composition containing the same |
| EP0274717A2 (en) | 1986-12-26 | 1988-07-20 | Sumitomo Chemical Company, Limited | Plant male sterilant |
| EP0320793A2 (en) * | 1987-12-17 | 1989-06-21 | Sumitomo Chemical Company, Limited | Cinnoline derivative, process for preparing the same and herbicidal composition containing the same |
| US5129939A (en) * | 1988-09-13 | 1992-07-14 | Orsan | Pollen suppressant comprising a 5-oxy- or amino-substituted cinnoline |
| US5183891A (en) | 1989-11-09 | 1993-02-02 | Orsan | Method for the preparation of substituted 1,4-dihydro-4-oxo-cinnoline-3-carboxylic acid, esters and salts thereof, and intermediates used in their preparation |
| WO1997033890A1 (en) | 1996-03-11 | 1997-09-18 | Novartis Ag | Pyrimidin-4-one derivatives as pesticide |
| WO2002034048A1 (en) | 2000-10-23 | 2002-05-02 | Syngenta Participations Ag | Agrochemical compositions with quinoline safeners |
Non-Patent Citations (2)
| Title |
|---|
| "The Pesticide Manual", 2009, BRITISH CROP PROTECTION COUNCIL |
| CAS, no. 9005-64-5 |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2023088921A1 (en) | 2021-11-18 | 2023-05-25 | Syngenta Crop Protection Ag | Herbicidal derivatives |
| AU2022392750B2 (en) * | 2021-11-18 | 2025-03-13 | Syngenta Crop Protection Ag | Herbicidal derivatives |
| WO2024160989A1 (en) | 2023-02-03 | 2024-08-08 | Syngenta Crop Protection Ag | Herbicide resistant plants |
Also Published As
| Publication number | Publication date |
|---|---|
| AR122103A1 (en) | 2022-08-10 |
| JP7770340B2 (en) | 2025-11-14 |
| EP4152932A1 (en) | 2023-03-29 |
| CN115802892A (en) | 2023-03-14 |
| GB202007419D0 (en) | 2020-07-01 |
| CN115802892B (en) | 2025-08-08 |
| UY39218A (en) | 2021-12-31 |
| US20230303498A1 (en) | 2023-09-28 |
| AU2021276067A1 (en) | 2022-12-15 |
| BR112022023491A2 (en) | 2022-12-20 |
| CA3178605A1 (en) | 2021-11-25 |
| JP2023526085A (en) | 2023-06-20 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2022117446A1 (en) | Herbicidal derivatives | |
| EP4448509A1 (en) | Herbicidal pyridone derivatives | |
| EP4532468A1 (en) | Herbicidal derivatives | |
| JP7770340B2 (en) | Herbicidal shinorine derivatives | |
| WO2024115438A1 (en) | Herbicidal derivatives | |
| AU2023281844A1 (en) | Herbicidal derivatives | |
| WO2023232674A1 (en) | Herbicidal derivatives | |
| EP4433467B1 (en) | HERBICIDE DERIVATIVES | |
| WO2023156323A1 (en) | Herbicidal quinolone derivatives | |
| CA3248515A1 (en) | Herbicidal 2-oxo-nicotinic acid derivatives | |
| AU2024263972A1 (en) | Herbicidal derivatives | |
| WO2026002882A1 (en) | Herbicidal derivatives | |
| WO2024047202A1 (en) | Herbicidal pyridone derivatives | |
| WO2025119663A1 (en) | Herbicidal derivatives |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 21726383 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 3178605 Country of ref document: CA |
|
| ENP | Entry into the national phase |
Ref document number: 2022570595 Country of ref document: JP Kind code of ref document: A |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: 112022023491 Country of ref document: BR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 202217071254 Country of ref document: IN |
|
| ENP | Entry into the national phase |
Ref document number: 2021276067 Country of ref document: AU Date of ref document: 20210514 Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 112022023491 Country of ref document: BR Kind code of ref document: A2 Effective date: 20221118 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2021726383 Country of ref document: EP Effective date: 20221219 |
|
| WWG | Wipo information: grant in national office |
Ref document number: 202180047977.7 Country of ref document: CN |