WO2017191453A1 - Anti-microbial compositions - Google Patents
Anti-microbial compositions Download PDFInfo
- Publication number
- WO2017191453A1 WO2017191453A1 PCT/GB2017/051243 GB2017051243W WO2017191453A1 WO 2017191453 A1 WO2017191453 A1 WO 2017191453A1 GB 2017051243 W GB2017051243 W GB 2017051243W WO 2017191453 A1 WO2017191453 A1 WO 2017191453A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- edta
- compound
- suitably
- biofilm
- ppm
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N55/00—Biocides, pest repellants or attractants, or plant growth regulators, containing organic compounds containing elements other than carbon, hydrogen, halogen, oxygen, nitrogen and sulfur
- A01N55/02—Biocides, pest repellants or attractants, or plant growth regulators, containing organic compounds containing elements other than carbon, hydrogen, halogen, oxygen, nitrogen and sulfur containing metal atoms
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N37/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom having three bonds to hetero atoms with at the most two bonds to halogen, e.g. carboxylic acids
- A01N37/44—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom having three bonds to hetero atoms with at the most two bonds to halogen, e.g. carboxylic acids containing at least one carboxylic group or a thio analogue, or a derivative thereof, and a nitrogen atom attached to the same carbon skeleton by a single or double bond, this nitrogen atom not being a member of a derivative or of a thio analogue of a carboxylic group, e.g. amino-carboxylic acids
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N59/00—Biocides, pest repellants or attractants, or plant growth regulators containing elements or inorganic compounds
- A01N59/16—Heavy metals; Compounds thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L15/00—Chemical aspects of, or use of materials for, bandages, dressings or absorbent pads
- A61L15/16—Bandages, dressings or absorbent pads for physiological fluids such as urine or blood, e.g. sanitary towels, tampons
- A61L15/42—Use of materials characterised by their function or physical properties
- A61L15/44—Medicaments
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/02—Drugs for dermatological disorders for treating wounds, ulcers, burns, scars, keloids, or the like
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C229/00—Compounds containing amino and carboxyl groups bound to the same carbon skeleton
- C07C229/02—Compounds containing amino and carboxyl groups bound to the same carbon skeleton having amino and carboxyl groups bound to acyclic carbon atoms of the same carbon skeleton
- C07C229/04—Compounds containing amino and carboxyl groups bound to the same carbon skeleton having amino and carboxyl groups bound to acyclic carbon atoms of the same carbon skeleton the carbon skeleton being acyclic and saturated
- C07C229/06—Compounds containing amino and carboxyl groups bound to the same carbon skeleton having amino and carboxyl groups bound to acyclic carbon atoms of the same carbon skeleton the carbon skeleton being acyclic and saturated having only one amino and one carboxyl group bound to the carbon skeleton
- C07C229/10—Compounds containing amino and carboxyl groups bound to the same carbon skeleton having amino and carboxyl groups bound to acyclic carbon atoms of the same carbon skeleton the carbon skeleton being acyclic and saturated having only one amino and one carboxyl group bound to the carbon skeleton the nitrogen atom of the amino group being further bound to acyclic carbon atoms or to carbon atoms of rings other than six-membered aromatic rings
- C07C229/16—Compounds containing amino and carboxyl groups bound to the same carbon skeleton having amino and carboxyl groups bound to acyclic carbon atoms of the same carbon skeleton the carbon skeleton being acyclic and saturated having only one amino and one carboxyl group bound to the carbon skeleton the nitrogen atom of the amino group being further bound to acyclic carbon atoms or to carbon atoms of rings other than six-membered aromatic rings to carbon atoms of hydrocarbon radicals substituted by amino or carboxyl groups, e.g. ethylenediamine-tetra-acetic acid, iminodiacetic acids
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C229/00—Compounds containing amino and carboxyl groups bound to the same carbon skeleton
- C07C229/76—Metal complexes of amino carboxylic acids
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F1/00—Compounds containing elements of Groups 1 or 11 of the Periodic Table
- C07F1/005—Compounds containing elements of Groups 1 or 11 of the Periodic Table without C-Metal linkages
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F1/00—Compounds containing elements of Groups 1 or 11 of the Periodic Table
- C07F1/08—Copper compounds
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F1/00—Compounds containing elements of Groups 1 or 11 of the Periodic Table
- C07F1/10—Silver compounds
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F3/00—Compounds containing elements of Groups 2 or 12 of the Periodic Table
- C07F3/003—Compounds containing elements of Groups 2 or 12 of the Periodic Table without C-Metal linkages
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F3/00—Compounds containing elements of Groups 2 or 12 of the Periodic Table
- C07F3/06—Zinc compounds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61L—METHODS OR APPARATUS FOR STERILISING MATERIALS OR OBJECTS IN GENERAL; DISINFECTION, STERILISATION OR DEODORISATION OF AIR; CHEMICAL ASPECTS OF BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES; MATERIALS FOR BANDAGES, DRESSINGS, ABSORBENT PADS OR SURGICAL ARTICLES
- A61L2300/00—Biologically active materials used in bandages, wound dressings, absorbent pads or medical devices
- A61L2300/40—Biologically active materials used in bandages, wound dressings, absorbent pads or medical devices characterised by a specific therapeutic activity or mode of action
- A61L2300/404—Biocides, antimicrobial agents, antiseptic agents
Definitions
- the present invention relates to compounds, compositions, methods and uses for sanitising and/or substantially removing biofilms and microorganisms living within or around said biofilm.
- the invention relates to metal-EDTA compounds which can be used in wound treatments, wound dressings, medical devices, water treatments, food processing and dental care to treat biofilms and the pathogenic microorganisms which live within and around said biofilms.
- EPS extracellular polymeric substances
- EPS is composed of polysaccharides, proteins, extracellular DNA and other biological and inorganic material.
- EPS forms the matrix of a biofilm and helps to protect microorganisms from immune cells and anti-microbial agents, reducing the efficacy of anti-microbial agents (e.g. antibiotics and biocides, in particular antiseptics). Therefore it is desirable to disrupt the biofilm and expose the microorganisms to anti-microbial agents applied to the biofilm and in doing so reduce the concentration of such anti-microbial agents needed to effectively treat the microorganisms living in the biofilm.
- anti-microbial agents e.g. antibiotics and biocides, in particular antiseptics
- Ethylenediaminetetra-acetic acid added as the di-sodium or calcium di-sodium salts has been used to treat topical infections or to treat hard surfaces such as catheters.
- WO03/047341 describes the use of EDTA, for example as an additive for a toothpaste.
- EDTA is also used as a formulation agent to reduce the effects of water hardness and generally as a chelating agent.
- EDTA combinations with other antibiotic agents have been disclosed, for example in US 5998488 where EDTA is used in combination with an anti-microbial preservative in a solution for ophthalmic use.
- compositions consisting essentially of or “consists essentially of” means including the components specified but excluding other components except for materials present as impurities, unavoidable materials present as a result of processes used to provide the components, and components added for a purpose other than achieving the technical effect of the invention.
- a composition consisting essentially of a set of components will comprise less than 5% by weight, typically less than 3% by weight, more typically less than 1 % by weight of non-specified components.
- consisting of or “consists of means including the components specified but excluding the addition of other components.
- anti-microbial refers to a compound or a composition that may kill and/or inhibit and/or stop the growth of any one or more types of microorganisms, including, viruses, prions, protozoa, amoeba, bacteria, fungi and yeasts, or any one or more of specific species of microorganism.
- biofilm refers to both a monocultured and a polymicrobial community of microorganisms enclosed in an extracellular polymeric matrix (composed of EPS), and attached to a biotic or an abiotic surface.
- biofilm formation refers to the attachment of microorganisms to surfaces and the subsequent development of multiple layers of cells within an EPS matrix.
- anti-biofilm refers to the inhibition of microbial biofilm formation and/or disruption and/or dispersal of biofilms and/or detachment and/or dispersion and/or breakdown of EPS of a biofilm.
- anti-inflammatory refers to the property of a substance or treatment that reduces inflammation or swelling, typically in a wound.
- infection refers to the invasion and multiplication of microorganisms such as bacteria, viruses, fungi, yeasts and parasites that are not normally present within the body.
- An infection may cause no symptoms and be subclinical, or it may cause symptoms and be clinically apparent.
- An infection may remain localized, or it may spread through the blood or lymphatic vessels to become systemic (body wide). Microorganisms that live naturally in the body are not considered infections.
- wound refers to a type of injury in which skin is torn, cut or punctured (an open wound), or where blunt force trauma causes a contusion (a closed wound).
- Acute wound refers to those wounds which are new and in the first phase of healing. Acute wounds are characterized by skin layers that have been punctured or broken by an external force or object. Any acute wound can progress to a chronic wound if it does not heal within the expected time frame or as a result of a poor supply of blood, oxygen, nutrients or through poor hygiene. Acute wounds should be properly treated to avoid infection and/or inflammation. Acute wounds are categorized based on causes such as lacerations, abrasions, punctures, incisions, gunshots, burns and according to their size and depth (superficial or deep).
- chronic wound refers to a wound that will not repair itself over time. Chronic wounds are often thought to be "stuck” in one of the phases of wound healing, and are most often seen in the older adult population. Typically, if a wound is not healing as expected within 2-3 months, it is considered chronic. Chronic wounds include pressure ulcers (e.g. bed sores), arterial and venous leg ulcers, and diabetic ulcers.
- pressure ulcers e.g. bed sores
- arterial and venous leg ulcers e.g. diabetic ulcers.
- n is an integer from 2 to 4; each M is a metal ion; and
- M n comprises at least two different metal ions selected from Ag, Al, Au, Ba, Bi, Tl, Ce, Co, Cu, Fe, Ga, Ir, Mo, Rh, Ru, Ti, and Zn ions.
- the compound of formula M n (EDTA) may be described as a metal-EDTA compound.
- metal-EDTA compound refers to any type of chemical species which comprises at least one metal and at least one EDTA molecule, whether said metal and said EDTA compound are present as salts, ions or neutral species and therefore encompasses metal-EDTA salts, chelates and co-ordination complexes.
- the compound of formula M n (EDTA) may be a salt and/or a chelate and/or a co-ordination complex.
- the compound of formula M n (EDTA) is a salt comprising ionic bonds between the metal atoms and the EDTA molecule.
- the compound of formula M n (EDTA) is a neutral compound. It may be assumed that where no charge information is given in a particular formula, the compound represented by that formula has no overall charge.
- the compound may be in the form of a solid.
- the compound may be present in a solution or a suspension, suitably an aqueous solution or an aqueous suspension, suitably an aqueous solution.
- n is an integer from 2 to 4. Therefore the compound may be a di-metal-EDTA compound, a tri- metal-EDTA compound or a tetra-metal-EDTA compound.
- the compound may be a mixture of compounds of formula M n (EDTA).
- the mixture of compounds of formula M n (EDTA) may comprise different numbers of metal ions, for example at least two of the following species: M 2 (EDTA), M 3 (EDTA) and M 4 (EDTA).
- M 4 (EDTA) each metal ion has a 1 + charge and the compound has no overall charge
- the EDTA component has no free carboxylic acid groups.
- the formula is M 3 (EDTA)
- each metal ion has a 1 + charge and the compound has no overall charge
- the EDTA component has one free carboxylic acid group.
- each metal ion has a 1 + charge and werein the compound has no overall charge, the EDTA component has two free carboxylic acid groups.
- the number of metal ions (M) which the compound comprises may depend on the pH of a solution in which the compound may be present. For example an acidic pH may reduce the number of metal ions (M) present in the compound. If any of the metal ions (M) present in the compound of formula M n (EDTA) have a higher than 1 + charge, for example Zn 2+ and wherein the compound has no overall charge, then the number of metal ions (M) which can be present in the compound is reduced (i.e. n is 2 or 3) and the number of free carboxylic acids present in the compound will be reduced accordingly.
- M n comprises at least two different metal ions selected from Ag, Al, Au, Ba, Bi, Tl, Ce, Co, Cu, Fe, Ga, Ir, Mo, Rh, Ru, Ti, and Zn ions.
- M n comprises at least two different metal ions selected from Ag, Au, Ce, Cu, Ga and Zn ions.
- M n comprises at least two different metal ions selected from Ag, Al, Au, Ba, Bi, Tl, Ce, Co, Cu, Fe, Ga, Ir, Mo, Rh, Ru, Ti, and Zn ions
- the compound may be considered to comprise EDTA, at least a first metal ion M and at least a second metal ion M 2 , wherein M and M 2 are different and are each selected from Ag, Al, Au, Ba, Bi, Tl, Ce, Co, Cu, Fe, Ga, Ir, Mo, Rh, Ru, Ti, and Zn ions.
- M n can comprise up to 4 metal ions.
- M n may comprise other metal ions in addition to the at least two different metal ions selected from Ag, Al, Au, Ba, Bi, Tl, Ce, Co, Cu, Fe, Ga, Ir, Mo, Rh, Ru, Ti, and Zn ions.
- the other metal ions which M n comprises are Na ions.
- M n comprises only metal ions selected from Ag, Al, Au, Ba, Bi, Tl, Ce, Co, Cu, Fe, Ga, Ir, Mo, Rh, Ru, Ti, and Zn ions.
- M n may comprise two, three or four different metal ions selected from Ag, Al, Au, Ba, Bi, Tl, Ce, Co, Cu, Fe, Ga, Ir, Mo, Rh, Ru, Ti, and Zn ions.
- M n may comprise more than one of any specific metal ion selected from Ag, Al, Au, Ba, Bi, Tl, Ce, Co, Cu, Fe, Ga, Ir, Mo, Rh, Ru, Ti, and Zn ions, for example more than one Ag ion.
- n 3 or 4 and M n comprises two Ag ions.
- the compound of the formula Mn(EDTA) also comprises at least one metal ion selected from Al, Au, Ba, Bi, Tl, Ce, Co, Cu, Fe, Ga, Ir, Mo, Rh, Ru, Ti, and Zn ions.
- M n comprises at least one Ag ion.
- M n comprises at least one Ag ion and at least one metal ion selected from Cu, Ga and Zn ions.
- M n comprises at least two Ag ions and at least one metal ion selected from Cu, Ga and Zn ions.
- M n comprises at least one Ag ion and one Zn ion.
- n is 3 and M n comprises two Ag ions and one Zn ion.
- the compound of this first aspect has the formula Ag 2 Zn(EDTA).
- n is 2 and M n comprises one Ag ions and one Zn ion.
- the compound of this first aspect has the formula AgZnNa(EDTA).
- Zn ions have anti-inflammatory properties and therefore when the compound of the formula M n (EDTA) includes one or more Zn ions, the Zn ions may provide a beneficial anti-inflammatory effect to a wound on which the compound of this first aspect may be applied, in addition to any antimicrobial and/or anti-biofilm effects of the compound.
- M n comprises at least one Ag ion and one Cu ion.
- n is 3 and M n comprises two Ag ions and one Cu ion.
- the compound of this first aspect has the formula Ag 2 Cu(EDTA).
- n is 2 and M n comprises one Ag ions and one Cu ion.
- the compound of this first aspect has the formula AgCuNa(EDTA).
- n is 2 and M n comprises a Cu ion and a Zn ion.
- the compound has the formula CuZn(EDTA).
- M n comprises a Cu ion and a Zn ion
- the Cu ions in combination with the Zn ions may exhibit a synergistic anti-microbial effect, which may allow a lower and therefore less cytotoxic concentration of a compound of this first aspect to be used than may otherwise be possible.
- the compounds of this first aspect may be synthesised according to the methods described in the Examples.
- the compounds of this first aspect can be synthesised by reacting a tetra sodium salt of EDTA (Na 4 (EDTA)), which is commercially available, with a mixture of salts of at least two different metal ions (M) selected from Ag, Al, Au, Ba, Bi, Tl, Ce, Co, Cu, Fe, Ga, Ir, Mo, Rh, Ru, Ti, and Zn ions.
- metal ions include sulphates and chlorides.
- the inventors have found that compounds of this first aspect may exhibit useful biological activity.
- the compounds may exhibit any one or more of anti-microbial, anti-biofilm and antiinflammatory activities in use.
- the compounds are preferably safe for human administration and are biocompatible and non-corrosive.
- the compounds exhibit anti-microbial and/or anti-biofilm and/or anti-inflammatory activities in use, for example when incorporated into a wound dressing or a medical device.
- the compounds of this first aspect may be useful in treating or combatting biofilms, for example disrupting and/or dispersing biofilms and providing an anti-microbial action on the microbes living on and within said biofilm and/or breaking down the EPS of the biofilm.
- the compounds of this first aspect may be used in a wound dressing, a medical device, on skin or on other abiotic or biotic surfaces important in water treatment (including water storage and distribution), food preparation and processing or dentistry.
- the compounds of this first aspect may be used in the form of a solution or suspension, for example for decontaminating surfaces, food preparation and processing equipment and/or food products, in particular meat processing equipment and meat products.
- Such a solution may be used as a mastitis dip solution, an ophthalmic solution, for wound irrigation or in dentistry.
- the compounds of this first aspect may be used in an endodontic irrigating solution and/or for cleaning/disinfecting dental water lines.
- the compounds of this first aspect may be used as a medicament, for example in a cream, paste or lotion.
- the use of compounds according to the first aspect when applied to a wound may react with Ca and/or Mg ions present in a biofilm associated with the wound. This reaction may remove the Ca and/or Mg ions from the biofilm which then disrupts the integrity of the biofilm. It is believed that Ca and Mg ions are important for the maintenance of a biofilm's structural integrity as these ions bind together the EPS which makes up the matrix of the biofilm. This reaction may also cause the metal ions (M) to be displaced from the compound and therefore be released into the wound and/or biofilm to produce desirable biological activity.
- M metal ions
- the reaction with the Ca and/or Mg present in a biofilm may release Ag ions into the wound and/or biofilm to provide a beneficial anti-microbial effect and/or may release Zn ions into the wound and/or biofilm to provide a beneficial anti-inflammatory effect.
- Zn ions are believed to provide an antiinflammatory effect.
- the use of the compound of this first aspect may therefore improve wound healing by simultaneously preventing and treating microbial infections and/or reducing wound inflammation.
- a composition comprising one or more compounds of formula M n (EDTA) according to the first aspect.
- the compound of formula M n (EDTA) in the composition of the second aspect may have any of the suitable features and/or advantageous properties described herein in relation to the first aspect.
- composition of this second aspect may be provided in any suitable form, such as in the form of a solution or a suspension or in a dry form.
- composition is a fluid.
- the composition may be an aqueous solution, a lotion, a cream, a balm, a gel, a paste or a solid, suitably powdered, composition.
- the composition of this second aspect is a solution of the compound of formula M n (EDTA).
- the compound of formula M n (EDTA) is suitably dissolved in a suitable solvent.
- the solution may comprise an aqueous solution such as water or saline, or another biocompatible solution in which the compound of formula M n (EDTA) is soluble.
- the solution may comprise an alcohol, for example ethanol.
- the composition is a solution of the compound of formula M n (EDTA) in a mixture of water and ethanol.
- solutions may be highly efficacious and may be prepared by making a concentrated stock solution of the compound of formula M n (EDTA) in water and then introducing the desired concentration of ethanol.
- the solution of the compound of formula M n (EDTA) is preferably provided in a sterile and non- pyrogenic form and may be packaged in any convenient fashion.
- the solution of the compound of formula M n (EDTA) may be provided in connection with or as part of a medical device, such as in a pre-filled syringe or another medical device.
- the solution of the compound of formula M n (EDTA) may be prepared under sterile, aseptic conditions, or may be sterilized following preparation and/or packaging using any of a variety of suitable sterilization techniques.
- Single use vials, syringes or containers of the solution of the compound of formula M n (EDTA) may be provided. Multiple use vials, syringes or containers may also be provided.
- the composition of this second aspect is provided in a substantially dry form, such as a substantially dry coating on a surface of tubing, or a conduit, or a medical or industrial device such as a catheter or a container and the like.
- a substantially dry form of the composition of this second aspect may be provided in a powder or lyophilized form that may be reconstituted to form a solution with the addition of a solvent.
- Substantially dry forms of the composition may alternatively be provided as a coating, or may be incorporated in a gel or another type of carrier, or encapsulated or otherwise packaged and provided on a surface as a coating or in a container.
- Such substantially dry forms of the composition of this second aspect are formulated such that in the presence of a solvent, the substantially dry composition forms a solution or suspension of the compound of formula M n (EDTA) having the properties described above.
- EDTA compound of formula M n
- different encapsulation or storage techniques may be employed such that effective time release of the compound of formula M n (EDTA) is accomplished upon extended exposure to solutions.
- the substantially dry composition of this second aspect may provide anti-microbial and/or anti-biofilm and/or antiinflammatory activity over an extended period of time and/or upon multiple exposures to solutions.
- composition is an aqueous solution.
- the pH of the composition is up to 12.5.
- the pH of the composition is in the range from 4.0 to 12.0, suitably in the range from 4.0 to 10.0, suitably from 4.5 to 8.0.
- the pH of the composition is in the range from 4.0 to 7.0, suitably in the range from 4.0 to 6.0, suitably from 4.0 to 5.0.
- the composition is an aqueous solution having a pH in the range from 4.0 to 12.0, suitably in the range from 4.0 to 1 1 .0, suitably in the range from 4.0 to 10.0, suitably from 4.5 to 8.0.
- the pH of the composition is in the range from 4.0 to 7.0, suitably in the range from 4.0 to 6.0, suitably from 4.0 to 5.0.
- the compound of formula M n (EDTA) is present in the composition in an amount of at least 0.01 ppm, suitably at least 0.1 ppm, suitably at least 1 .0 ppm, suitably at least 10 ppm, suitably at least 100 ppm, suitably at least 1 ,000 ppm, suitably at least 5,000 ppm.
- the compound of formula M n (EDTA) is present in the composition in an amount of up to 100,000 ppm, suitably up to 10,000 ppm, suitably up to 5,000 ppm.
- the compound of formula M n (EDTA) is present in the composition in an amount of from 0.01 ppm to 100,000 ppm, suitably from 0.1 ppm to 10,000 ppm, suitably from 1 .0 ppm to 10,000 ppm, suitably from 10 ppm to 100 ppm, suitably from 100 ppm to 10,000 ppm, suitably from 1 ,000 ppm to 10,000 ppm.
- the composition comprises EDTA and at least a first metal ion M and a second metal ion M 2 , wherein M and M 2 are different and are each selected from Ag, Al, Au, Ba, Bi, Tl, Ce, Co, Cu, Fe, Ga, Ir, Mo, Rh, Ru, Ti, and Zn ions.
- EDTA is present in the composition in an amount of from at least 0.01 ppm, suitably at least 0.1 ppm, suitably at least 1 .0 ppm, suitably at least 10 ppm, suitably at least 100 ppm, suitably at least 1 ,000 ppm.
- EDTA is present in the composition in an amount of up to 100,000 ppm, suitably up to 10,000 ppm, suitably up to 5,000 ppm.
- EDTA is present in the composition in an amount of from 0.01 ppm to 100,000 ppm, suitably from 0.1 ppm to 10,000 ppm, suitably from 1 .0 ppm to 10,000 ppm, suitably from 10 ppm to 100 ppm, suitably from 100 ppm to 10,000 ppm, suitably from 1 ,000 ppm to 10,000 ppm.
- the first metal ion M is present in the composition in an amount of from at least 0.01 ppm, suitably at least 0.1 ppm, suitably at least 1 .0 ppm, suitably at least 10 ppm, suitably at least 100 ppm, suitably at least 1 ,000 ppm.
- the first metal ion M is present in the composition in an amount of up to 100,000 ppm, suitably up to 10,000 ppm, suitably up to 5,000 ppm.
- the first metal ion M is present in the composition in an amount of from 0.01 ppm to 100,000 ppm, suitably from 0.1 ppm to 10,000 ppm, suitably from 1 .0 ppm to 10,000 ppm, suitably from 10 ppm to 100 ppm, suitably from 100 ppm to 10,000 ppm, suitably from 1 ,000 ppm to 10,000 ppm.
- the second metal ion M 2 is present in the composition in an amount of from at least 0.01 ppm, suitably at least 0.1 ppm, suitably at least 1 .0 ppm, suitably at least 10 ppm, suitably at least 100 ppm, suitably at least 1 ,000 ppm.
- the second metal ion M 2 is present in the composition in an amount of up to 100,000 ppm, suitably up to 10,000 ppm, suitably up to 5,000 ppm.
- the second metal ion M 2 is present in the composition in an amount of from 0.01 ppm to 100,000 ppm, suitably from 0.1 ppm to 10,000 ppm, suitably from 1 .0 ppm to 10,000 ppm, suitably from 10 ppm to 100 ppm, suitably from 100 ppm to 10,000 ppm, suitably from 1 ,000 ppm to 10,000 ppm.
- the suitable concentration of the first and/or second and/or any further metal ion may depend on the specific metal ion present. For example, a silver metal ion may be present in a relatively low concentration and still be effective in a given application.
- the composition may comprise other metal ions in addition to the at least two different metal ions selected from Ag, Al, Au, Ba, Bi, Tl, Ce, Co, Cu, Fe, Ga, Ir, Mo, Rh, Ru, Ti, and Zn ions.
- Such other metal ions may be any suitable metal ion, for example Na ions.
- the composition may comprise more than two different metal ions selected from Ag, Al, Au, Ba, Bi, Tl, Ce, Co, Cu, Fe, Ga, Ir, Mo, Rh, Ru, Ti, and Zn ions.
- the composition may comprise a carrier and/or an excipient, suitably a pharmaceutically acceptable carrier and/or excipient.
- Suitable carriers and/or an excipients may be selected from water, ethanol, polypropylene glycol, glycerol, sorbitol, hydrocolloids, polyoxyethtylene block copolymers, carboxy methyl cellulose, pluronic F-127, cotton, chitosan, silicone, polyurethanes, acrylics, hydrogels, bamboo, soya, oils/fats, micelles, emulsions, paints, sodium alginate, polyethylene glycol, thickening agents such as CarbopolTM and mixtures thereof.
- the composition of this second aspect may be in the form of a water-based gel.
- the composition is a hydrogel which comprises the one or more compounds of formula M n (EDTA).
- EDTA compounds of formula M n
- such hydrogels can maintain a moist wound healing environment and promote wound healing when said composition is applied to a wound, for example as part of a wound dressing.
- Such hydrogels may flow into the wound when applied to said wound to form an intimate contact with the wound bed and provide anti-microbial and/or anti-inflammatory effects to the whole wound.
- the hydrogel has a high enough viscosity that it does not flow out of wounds on areas of the body that are or become non-horizontal through movement of the patient.
- the hydrogel comprises a buffer, suitably to buffer the pH of the hydrogel to between 5.5 to 12.0. Suitable buffers are known in the art.
- the composition may comprise a non-metal ion anti-microbial agent.
- Suitable non-metal ion anti-microbial agents may be selected from any one or more of a chlorhexidine, a chlorhexadine salt, a triclosan, a polymoxin, a tetracycline, an amino glycoside (e.g.
- gentamicin or Tobramycin(TM) a rifampicin, a bacitracin, an erythromycin, a neomycin, a chloramphenicol, a miconazole, a quinolone, a penicillin, a nonoxynol 9, a fusidic acid, a cephalosporin, a mupirocin, a metronidazole, a secropin, a protegrin, a bacteriolcin, a defensin, a nitrofurazone, a mafenide, a acyclovir, a vanocmycin, a clindamycin, a lincomycin, a sulfonamide, a norfloxacin, a pefloxacin, a nalidizic acid, an oxalic acid, an enoxacin acid, a ciprofloxacin, a biguanide, i
- the non-metal ion anti-microbial agent may provide a beneficial anti-microbial which is additive to and/or synergistic with any anti-microbial effect provided by the compound of formula Mn(EDTA).
- the composition may comprise a surfactant.
- a suitable surfactant may be sodium hexametaphosphate or a quaternary ammonium compound.
- a surfactant may improve the anti-biofilm effectiveness of the composition by removing matter from a biofilm during and/or after the action of the compound of formula Mn(EDTA) to disrupt the biofilm.
- the compound of formula M n (EDTA) may act to disrupt the biofilm and in doing so produce flocculated cells.
- the surfactant may act to at least partially solubilise and remove these cells and prevent them from re-adhering to the biofilm and in doing so assist with the break-up and removal of the biofilm.
- the composition may comprise an anti-biofilm agent, suitably in addition to any anti-biofilm properties of the compound of formula M n (EDTA) and/or any non-metal ion anti-microbial agent, if present, and/or any surfactant, if present.
- the composition may comprise an anti- biofilm agent selected from any one or more of DisperinB, DNase 1 , ethylene glycol tetraacetic acid (EGTA), Proteinase K, apyrase, cis-2-decenoic acid, alginate lyase, lactoferrin, gallium, cellulose, and 5-fluorouracil.
- the composition comprises fibres which are in contact with the one or more compounds of formula M n (EDTA).
- compositions comprising fibres may be formed by impregnating the composition into and/or coating the composition onto fibres.
- Suitable fibres may be selected from natural fibres, synthetic fibres and combinations thereof.
- Suitable fibres may be selected from any one or more fibres of cellulose, alginates, cotton, chitosan, soya, bamboo, carboxymethylcellulose, Rayon, Nylon, acrylic, polyester, polyurethane, polyurethane foam and combinations thereof.
- compositions comprising fibres may be incorporated into and/or used to form a wound dressing.
- a wound dressing may have the advantage that the one or more compounds of formula M n (EDTA) present in the composition are delivered to the wound, producing the beneficial effects referred to in relation to the first aspect.
- the wound dressing may exhibit any one or more of anti-microbial, anti-biofilm and anti-inflammatory activities in use and therefore promote wound healing whilst combatting infection, if present.
- a wound dressing comprising a compound according to the first aspect or a composition according to the second aspect.
- the wound dressing may have any of the suitable features and advantages described in relation to the first and second aspects.
- the wound dressing comprises the compound of formula M n (EDTA) in a wound contact layer which has been impregnated, coated, dipped, laminated and/or sprayed with the compound of formula M n (EDTA).
- the wound dressing may comprise the compound of formula M n (EDTA) in an absorbant layer that is attached to a wound contact layer.
- the wound dressing may comprise the compound of formula M n (EDTA) in adhesive which contacts the skin in use.
- a medical device comprising a compound according to the first aspect or a composition according to the second aspect.
- the medical device may be a catheter.
- the medical device may be an intubation tube.
- the medical device may be a medical tube, a conduit, an intravascular device, an implanted medical device, a medical or veterinary instrument, a contact lens, an optical implant or a dental, orthodontic or periodontal device.
- the compound according to the first aspect or the composition according to the second aspect is coated onto at least a part of a surface of the medical device, suitably a surface which is intended to contact a part of a patient's body, in use.
- a surface of the medical device suitably a surface which is intended to contact a part of a patient's body, in use.
- the medical device of this fourth aspect has a reduced capacity for biofilm formation than a comparable medical device of the prior art which does not comprise such a compound or composition.
- the medical device of this fourth aspect may therefore reduce or substantially prevent infections caused by biofilm formation and pathogenic microorganism growth on the medical device.
- a use of a compound according to the first aspect or a composition according to the second aspect to sanitise and/or substantially remove a biofilm from a substrate.
- a method of sanitising and/or substantially removing a biofilm from a substrate comprising treating the substrate with a compound according to the first aspect or a composition according to the second aspect.
- the substrate may be any surface where biofilm treatment and/or removal is required.
- the substrate may be a wound on a human or animal body, the wound being of any of the types described above.
- the substrate may be a part of a medical device. In some embodiments the substrate may be a part of food preparation and processing equipment or a food product, for example meat processing equipment or meat products.
- the use and/or method at least disrupts and/or disperses the biofilm.
- the use and/or method increases the susceptibility of the biofilm and the microorganisms within it to attack by the compound of the first aspect and/or any metal ions released from the compound and/or any additional agents (such as non-metal ion anti-microbial agents and/or anti-biofilm agents) present in the composition according to the second aspect.
- the use and/or method completely removes the biofilm from the substrate.
- the use and/or method sanitises the substrate.
- the use and/or method completely removes the biofilm from the substrate and sanitises the substrate.
- the use and/or method carried out on a wound may advantageously facilitate wound healing and/or treat infections and/or reduce inflammation.
- the use and/or method carried out on a medical device may advantageously clean and/or sanitise the medical device and therefore prevent infections caused by medical devices comprising biofilms harbouring pathogenic microorganisms.
- the use and/or method carried out on a food product may advantageously slow or preferably stop the growth of pathogenic microorganisms on the food product and therefore prevent spoilage of the food product and food poisoning which may result from ingesting such food contaminated with pathogenic microorganisms.
- a compound according to the first aspect or a composition according to the second aspect for use as a medicament.
- the compound or composition is for use to sanitise and/or substantially remove a biofilm from a wound.
- the compound or composition for use in the sixth aspect may be used to treat infections of cuts, bruises, surgical sites, lacerations, abrasions, punctures, incisions, gunshots, burns, pyoderma, atopic dermatitis, eczema, pressure ulcers, venous and artery leg ulcers, diabetic foot ulcers, cystic fibrosis (CF)-associated infections, mastitis, otitis, community or hospital acquired infections or food-borne diseases.
- CF cystic fibrosis
- a method of producing a compound of the first aspect comprising reacting a metal-EDTA compound with a salt of a first metal ion selected from Ag, Al, Au, Ba, Bi, Tl, Ce, Co, Cu, Fe, Ga, Ir, Mo, Rh, Ru, Ti, and Zn ions and a salt of a second metal ion selected from Ag, Al, Au, Ba, Bi, Tl, Ce, Co, Cu, Fe, Ga, Ir, Mo, Rh, Ru, Ti, and Zn ions; wherein the first and second metal ions are different.
- the method involves reacting the metal-EDTA complex with the salt of the first metal ion and the salt of the second metal ion at the same time.
- the method of this seventh aspect comprises the steps of: a) reacting a metal-EDTA complex with a salt of a first metal ion selected from Ag, Al, Au, Ba, Bi, Tl, Ce, Co, Cu, Fe, Ga, Ir, Mo, Rh, Ru, Ti, and Zn ions; and b) reacting the EDTA complex comprising one metal ion selected from Ag, Al, Au, Ba, Bi, Tl, Ce, Co, Cu, Fe, Ga, Ir, Mo, Rh, Ru, Ti, and Zn ions formed in step a) with a salt of a second metal ion selected from Ag, Al, Au, Ba, Bi, Tl, Ce, Co, Cu, Fe, Ga, Ir, Mo, Rh, Ru, Ti, and Zn ions; wherein the first and second metal ions are different.
- the steps of the method are carried out in the order step a) followed by step b).
- kits comprising a compound according to the first aspect or a composition according to the second aspect and a medical device or wound dressing.
- the kit comprises a solution or suspension of the compound of formula M n (EDTA), suitably contained in a pre-filled syringe or another medical device.
- EDTA the compound of formula M n
- the medical device is a catheter or an intubation tube.
- an eighth aspect of the present invention there is provided a use of a compound according to the first aspect or a composition according to the second aspect to coat at least a part of a medical device. According to a further aspect of the present invention there is provided a use of a compound according to the first aspect or a composition according to the second aspect in a wound dressing.
- a ninth aspect of the present invention there is provided a use of a compound according to the first aspect or a composition according to the second aspect to provide anti- biofilm and/or anti-microbial and/or anti-inflammatory activity in a wound treatment or a medical device.
- Fe 3+ reacts with thiocyanate producing red complexes.
- Na 4 (EDTA) tetra sodium EDTA
- Fe 3+ reacts with the Fe 3+ resulting in a red colour that decreases as the levels of Na 4 EDTA increase.
- Fe 3+ solution - formed by dissolving ammonium iron(lll) sulfate dodecahydrate in a small amount of HCI, adding sodium acetate buffer and then adding to water.
- Figure 1 shows the standard correlation curve for tetra sodium EDTA.
- the results show there was a linear correlation between OD (optical density) value and the amount of tetra sodium EDTA in the solution at concentration from 0 to 1 1 .9 mM, which means this method can be used to measure quantitatively tetra sodium EDTA as solid or in the solution.
- the coated coverslips and plastic discs are then immersed in 2 ml deionized water for 1 week.
- the PU films on glass coverslips were named G1 , G2, G3, G4, and G5 correlating to different concentrations of tetra silver EDTA complex.
- the elution results are shown in table 2 below.
- Silver nitrate was reacted with a tetra-sodium salt of EDTA in a 4:1 molar ratio under vigorous stirring.
- the resulting solid was filtered off, washed with cold deionized water 3 times, washed with 50 % ethanol, 70 % ethanol and 100 % ethanol one time each; and then dried in a vacuum oven at 50 °C in a dark flask to avoid exposure to direct light.
- a white powder of Ag 4 (EDTA) was produced in the following yield.
- Figs. 2A-C The FTIR of EDTA, Na 4 (EDTA) and Ag 4 (EDTA) are shown in Figs. 2A-C.
- the absorption band centred at 1740 cm “1 in Fig. 2A (EDTA) attributed to the stretching vibration of carbonyl in COOH is not present in the spectra of Figs 2B and 2C (Na 4 EDTA and Ag 4 EDTA respectively).
- the spectrum of the pure Na 4 EDTA (Fig. 2B) is characterized by an absorption band centred at around 1580 cm "1 which is attributed to the asymmetric stretching vibration of carbonyl in the four ionic carboxylate groups (v as coo " ). The corresponding symmetric stretching vibration occurs close to 1465 cm “1 (v s coo " ).
- the asymmetric stretching vibration adsorption band of carbonyl in the Ag 4 EDTA (Fig. 2C) is much stronger than the respective peak in Na 4 EDTA and the band centre moves to around 1550 cm “1 .
- the corresponding symmetric stretching vibration band centred at 1465 cm “1 which is a less intense absorption band than the one at 1550 cm “1 .
- the Ag 4 (EDTA) powders were suspended in deionised water at 1 mg/ml. A 100 ⁇ portion of the suspension was dropped onto a first glass coverslip. A 50 ⁇ portion of the suspension was added to a 50 ⁇ portion of a 1 .7 % NaCI solution and dropped onto a second glass coverslip. A 50 ⁇ portion of the suspension was mixed with a 50 ⁇ portion of a PU solution and then dropped onto a third glass coverslip. All the coverslips were dried in vacuum oven at 50 °C in a dark flask to avoid exposure to direct light. The results showed that Ag 4 (EDTA) can be coated on glass or as films of polymers (certainly at least polyurethane). Displacement experiment
- the suspension quickly precipitated a solid (AgCI).
- the precipitate was filtered off, washed with deionized water 4 times; and then dried in a vacuum oven at 50 °C in a dark flask to avoid exposure to direct light.
- Fig. 3 shows the ATR-FTIR spectrum of the dark blue powder of Ag 2 Cu(EDTA).
- Examples - silver and zinc EDTA complex synthesis - compounds 3 and 4 These compounds were prepared by first dissolving 38 g of ethylenediaminetetraacetic acid tetrasodium salt hydrate (Tetra sodium EDTA) in 1000 ml H 2 0 to provide solution 1 . Then 68 g of silver nitrate (AgN0 3 ) was dissolved in 1000 ml H 2 0 to provide solution 2. 18.6 g of zinc sulfate monohydrate (ZnS0 4 « H 2 0) was dissolved in 1000ml H 2 0 to provide solution 4.
- Tetra sodium EDTA ethylenediaminetetraacetic acid tetrasodium salt hydrate
- the EDTA compounds 1 -4 were added to collagen films and then investigated for elution of these complexes.
- Hydrogels were loaded with EDTA compounds and investigated for elution of the silver, zinc and EDTA.
- Carbopol ETD 2020 was obtained from Lubrizol Advanced Materials, Inc. USA. 1 .2 g carbopol power was dissolved in 42.36 g deionized water for 1 hour to fully hydrate. Then 8.4 ml of the EDTA complexes and 4.8 g glycerine were added into carbopol solution with vigorous stirring; finally, 3 g 50 % triethanolamine was dropped into the suspension to obtain final products: carbopol hydrogel with EDTA complexes.
- Table 1 1 shows that all the hydrogels belong to 1 b or 1 c type. That means all the hydrogels are donation type.
- the tetra sodium EDTA only hydrogel was found to be in the 1 c hydrogel group (donated the most fluid); other gels were characterised as 1 b gels, which donated 5 to 10 % water from hydrogel to gelatin.
- Table 11 Water donation/absorption results
- MIC Minimum inhibitory concentration
- MMC minimum biocidal concentration
- MBEC minimum biofilm eradication
- results for the antimicrobial effectiveness of the dressing were expressed as microbicidal (Log 0 Reduction > 3) or microbistatic (Log 0 Reduction ⁇ 3). SD represents standard deviation. Experiment was performed on triplicate dressings.
- results for the antimicrobial effectiveness of the dressing were expressed as microbicidal (Log 0 Reduction > 3) or microbistatic (Log 0 Reduction ⁇ 3). SD represents standard deviation. Experiment was performed on triplicate dressings.
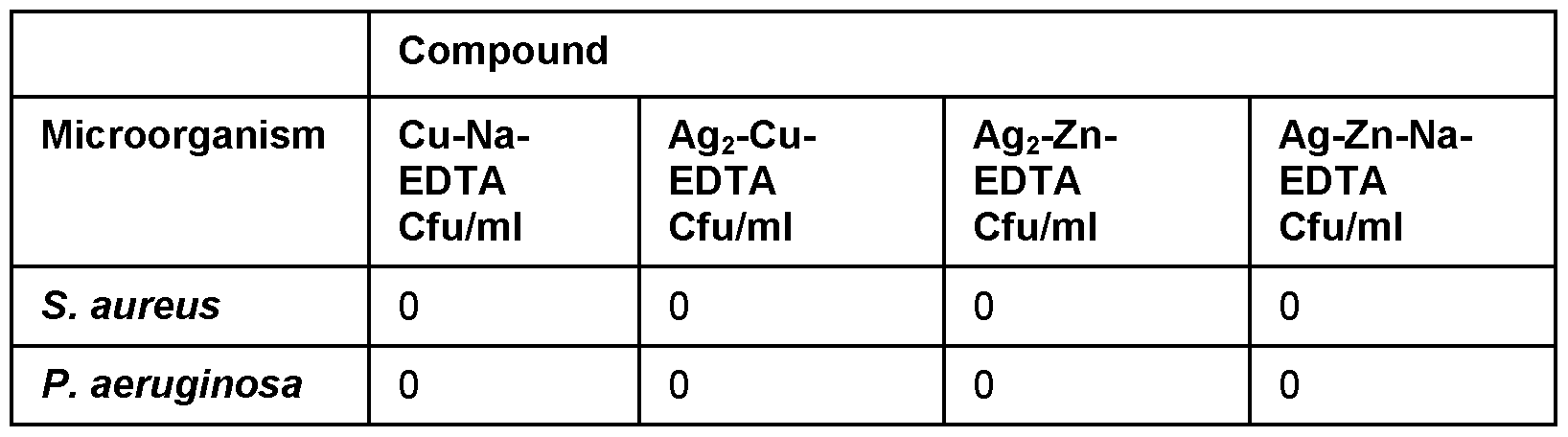
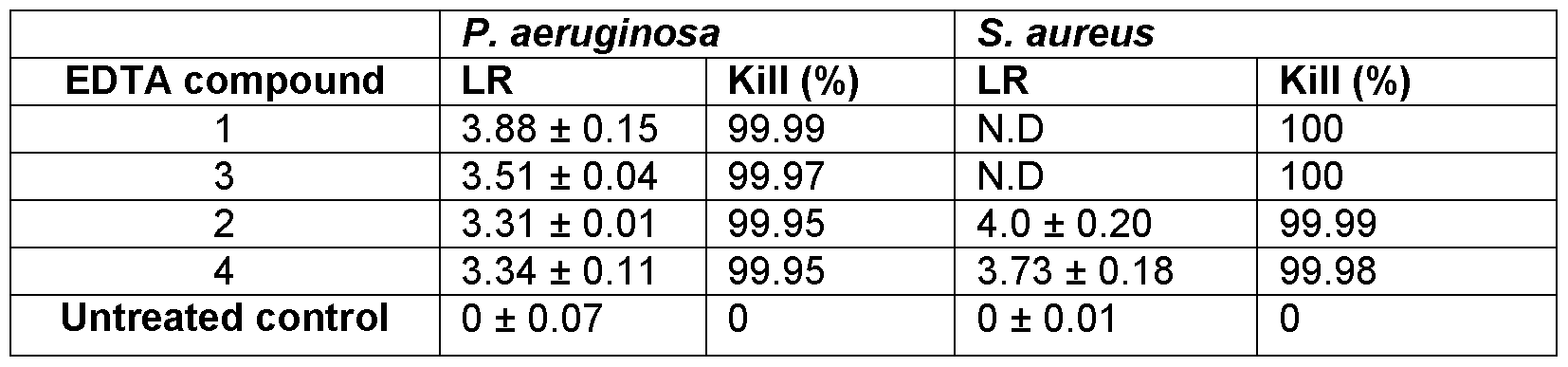
- EDTA compounds 1 -4 (prepared as described above) were tested against P. aeruginosa and S. aureus biofilms at a concentration of 100ppm silver, to which the compounds Ag/Cu/NaEDTA and Ag/Zn/NaEDTA showed 100% kill in S. aureus and at a 3.5 log reduction in P. aeruginosa (Table 18).
- the anti-biofilm efficacy of the EDTA-Ag-Zn compound as a liquid was evaluated using the MBEC model. 100% kill of P. aeruginosa biofilms (no viable colonies detected) following treatment with various concentrations of the complexes (1000ppm-62.5ppm) was observed (Table 19). Table 19. Log reduction values related to the treatment of S. aureus and P. aeruginosa 48-hour biofilms with EDTA-Ag 2 -Zn compound (EDTA compound 4)
- Biofilms were formed using the MBEC plate method. Log reduction (LR) values are expressed with standard error. N.D (not determined) is stated when no colonies were detected upon enumeration.
- the MBEC model (ASTM E2799) was used to assess the anti-biofilm efficacy of the liquid formulas. Biofilms were grown on polystyrene pegs in the MBEC model for either 24 or 48 hours before treatment. Using the liquid form of the EDTA complexes 24- and 48-hour biofilms were tested (Table 20). The results show that the complexes and silver solution (50ppm and 100ppm) caused 100% kill of 24- and 48-hour biofilms. Similarly, 4% tetrasodium EDTA caused 100% kill in 24 and 48 hours on S. aureus, MRSA, S. epidermidis and E. faecalis biofilms. However for P.
- EDTA-Ag 2 -Zn (compound 4) 50ppm 6.44 100 6.50 100 S. aureus MRSA BAA-43 control 0 ⁇ 0 0 0 ⁇ 0 0 tEDTA (4%) 5.40 100 6.43 100
- Gauze impregnated with the EDTA-Ag 2 -Zn compound 4 showed complete biofilm kill after 24 hours (see Table 21), which correlated with elution data whereby the largest amount of silver and EDTA eluted at 24 hours. Furthermore, gauze containing the EDTA-Ag 2 -Zn compound 4 was more effective than tetrasodium EDTA at 2% and 4%. There was a greater log reduction in biofilms when treated with gauze containing 4% EDTA than 2% EDTA. The collagen film containing the compound was less effective than the gauze.
- Biofilms (48-hours) were exposed to various platforms for 24 hours.
- the compound used to impregnate the platforms was EDTA-Ag 2 -Zn (compound 4). Samples were run in triplicate. Mean Log values were calculated and the standard error (SE) of the mean LR was determined (LR ⁇ SE). In cases whereby no colonies were detected following treatment, 'N.D' for 'not determined' is used, which represents 100% kill. Table 21 provides evidence that the complexes of the present invention demonstrate potential synergy between agents and out performed tetra sodium EDTA.
- the CDC biofilm bioreactor model (ASTM E2871) was used to test anti-biofilm efficacy of the hydrogel formulations. Biofilms were grown on polycarbonate coupons in the bioreactor for 48 hours (48-hour) biofilm. The formulation of a hydrogel containing the complexes showed complete biofilm kill in the CDC model after 24 hours of treatment (see Table 22). The hydrogel containing the compound 4 outperformed the hydrogel containing silver alone.
- Biofilms (48-hours) were exposed to hydrogel for 24 hours.
- the compound used to impregnate the platforms was EDTA-Ag 2 -Zn (compound 4). Samples were run in triplicate. Mean Log values were calculated and the standard error (SE) of the mean LR was determined (LR ⁇ SE). In cases whereby no colonies were detected following treatment, 'N.D' for 'not determined' is used, which represents 100% kill. Table 22 demonstrates evidence that the complex of EDTA-Ag 2 -Zn outperformed a silver hydrogel alone in antibiofilm activity.
- hydrogel EDTA-Ag 2 -Zn compound 4 To test the anti-biofilm efficacy of the hydrogel EDTA-Ag 2 -Zn compound 4, we tested the hydrogel against five microorganisms in biofilm form using the CDC biofilm bioreactor. The hydrogel control showed a slight log reduction in microbial numbers. Hydrogel containing silver resulted in 100% kill in S. aureus, MRSA, S. epidermidis and E. faecalis. However the silver hydrogel only caused a 3.16 ⁇ 1 .83 log reduction in P. aeruginosa (Table 23). The hydrogel containing zinc caused between a 1 .24 to 1 .84 log reduction respectively in all microorganisms except for S. epidermidis, whereby 100% kill was recorded.
- hydrogel containing tetrasodium EDTA caused a slight log reduction in most microorganisms, however caused a 100% kill in S. epidermidis.
- the hydrogel formulation containing a low concentration of the compound 4 did not have a significant reduction in most microorganisms but did cause a 100% kill in S. epidermidis.
- the high concentration of the compound 4 incorporated into the hydrogel caused 100% kill in all microbial biofilms tested.
- Table 23. Log reduction (LR) and percentage kill (%) values of 48-hour biofilms following treatment with hydrogel formulations in the CDC model. Biofilms (48-hours) were exposed to hydrogel for 24 hours at 37°C ⁇ 2°C.
- the compound used to impregnate the platforms was EDTA-Ag 2 -Zn (compound 4) at a low and high concentration. Samples were run in triplicate. Mean Log values were calculated and the standard error (SE) of the mean LR was determined (LR ⁇ SE). The mean log density (mean log density ⁇ standard deviation) of each microorganism is stated.
- Table 23 demonstrates that the compound 4 (EDTA-Ag 2 -Zn) outperformed a silver hydrogel alone, a zinc hydrogel alone and to tetra sodium EDTA alone, in antibiofilm activity.
- Results showed that increasing concentrations of tetrasodium EDTA were cytotoxic to cells with percentage viability between 3.87% and 14.23%. All concentrations of Compounds 1 and 3 were classed as cytotoxic, with viability ranging from 52.57% to 53.66% and 46.46% to 63.08%, respectively. Compound 2 at all concentrations was interpreted as non-cytotoxic with percentage viability ranging from 72.01 % and 72.21 %. Compound 4 at 50ppm was also non-cytotoxic, will 72.93% viability, however other concentrations were cytotoxic (Table 24). The agar diffusion method for indirect cytotoxicity allows for the qualitative assessment of cytotoxicity.
- DMEM (Negative control) 100 ⁇ 0 Non cytotoxic DMEM (Negative control) 100 ⁇ 0 Non cytotoxic DMEM (Negative control) 100 ⁇ 0 Non cytotoxic DMEM (Negative control) 100 ⁇ 0 Non cytotoxic DMEM (Negative control) 100 ⁇ 0 Non cytotoxic DMEM (Negative control) 100 ⁇ 0 Non cytotoxic
- Table 24 demonstrates that the compounds of the present invention were significantly lower in toxicity when compared to tetra sodium EDTA alone.
- Zone index and lysis index for indirect cytotoxicity test of EDTA compounds 1 -4 Zone index measures the clear zone in which cells do not stain with neutral red.
- the lysis index measures the number of cells affected within the zone of toxicity. All samples were tested in triplicate.
- Table 25 demonstrates that the complexes were significantly lower in toxicity when compared to tetra sodium EDTA alone.
- the efficacy of the EDTA-Ag 2 -Zn compound (compound 4) in a wound biofilm model At day 6, the untreated S. aureus wound biofilm was present in more than 50% of the tissue. After treatment with 100ppm of the Ag-Zn 2 -EDTA (compound 4), there appeared to be a reduced amount of microorganisms within the tissues, however there was no identifiable epidermis and the nuclei of fibroblasts in the dermal layer were faint.
- Enzyme-linked immunosorbent assay was used to assess the secreted levels of the inflammatory cytokine interleukin-6 (IL-6) according to previously published methods (Foster, A. M., Baliwag, J., Chen, C. S., Guzman, A. M., Stoll, S. W., Gudjonsson, J. E., Ward, N. L. & Johnston, A. 2014, IL-36 promotes myeloid cell infiltration, activation, and inflammatory activity in skin, The Journal of Immunology, 192, 6053-6061).
- ELISA detection of secreted IL-6 in the same experiment showed a reduction in IL-6 in the S. aureus wound biofilm control (Figure 4). Treatment of wound biofilm with the Ag-Zn 2 -EDTA (compound 4), was shown to reduce the presence of microorganisms upon histological examination. The IL-6 levels were reduced further when compared to the S. aureus wound biofilm control.
- Figure 4 shows IL-6 cytokine secretion from wounded, biofilm-containing Labskin following treatment with liquid Ag-Zn 2 -EDTA (compound 4).
- ELISA was used to assess the levels of IL-6 in conditioned cell culture medium. Culture medium was changed at day 4, which accounts for the reduction in IL-6. Biofilms were treated with either 50ppm or 100ppm of EDTA compound 4. Tests were performed in duplicate. Error bars indicate standard deviation.
- Figure 4 demonstrates that the Ag-Zn 2 -EDTA (compound 4) at 100ppm caused a significant reduction in the inflammatory marker.
- the present invention provides a metal-EDTA compound/complex for combatting biofilms and/or treating wounds.
- the compound/complex comprises EDTA and from two to four metal ions. Of those two to four metal ions, at least two are different metal ions selected from Ag, Al, Au, Ba, Bi, Tl, Ce, Co, Cu, Fe, Ga, Ir, Mo, Rh, Ru, Ti, and Zn ions.
- the metal- EDTA compound/complex may exhibit any one or more of anti-microbial, anti-biofilm and antiinflammatory activities in use and may increase the susceptibility of a biofilm and the microorganisms within said biofilm to attack by anti-microbial agents, helping to remove and sanitise the biofilm.
- a composition, wound dressing and medical device comprising the metal- EDTA complex are also provided.
- Uses of the metal-EDTA compound/complex as a medicament and/or to sanitise and/or substantially remove a biofilm from a substrate are also disclosed.
Landscapes
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- General Health & Medical Sciences (AREA)
- Wood Science & Technology (AREA)
- Zoology (AREA)
- Agronomy & Crop Science (AREA)
- Pest Control & Pesticides (AREA)
- Plant Pathology (AREA)
- Dentistry (AREA)
- Environmental Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Inorganic Chemistry (AREA)
- Hematology (AREA)
- Materials Engineering (AREA)
- Epidemiology (AREA)
- Medicinal Chemistry (AREA)
- Dermatology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Pharmacology & Pharmacy (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
- Materials For Medical Uses (AREA)
- Medicinal Preparation (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
Abstract
Description
Claims
Priority Applications (8)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201780042081.3A CN109414010B (en) | 2016-05-04 | 2017-05-04 | Antimicrobial composition |
| EP17723475.4A EP3451829B1 (en) | 2016-05-04 | 2017-05-04 | Anti-microbial compositions |
| DK17723475.4T DK3451829T3 (en) | 2016-05-04 | 2017-05-04 | ANTIMICROBIAL COMPOSITIONS |
| ES17723475T ES2836299T3 (en) | 2016-05-04 | 2017-05-04 | Antimicrobial compositions |
| JP2019510479A JP7020700B2 (en) | 2016-05-04 | 2017-05-04 | Antibacterial composition |
| US16/098,629 US10874108B2 (en) | 2016-05-04 | 2017-05-04 | Anti-microbial compositions |
| AU2017259304A AU2017259304B2 (en) | 2016-05-04 | 2017-05-04 | Anti-microbial compositions |
| SG11201809526YA SG11201809526YA (en) | 2016-05-04 | 2017-05-04 | Anti-microbial compositions |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| GB1607814.9 | 2016-05-04 | ||
| GBGB1607814.9A GB201607814D0 (en) | 2016-05-04 | 2016-05-04 | Anti-microbial compositions |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2017191453A1 true WO2017191453A1 (en) | 2017-11-09 |
Family
ID=56234390
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/GB2017/051243 Ceased WO2017191453A1 (en) | 2016-05-04 | 2017-05-04 | Anti-microbial compositions |
Country Status (10)
| Country | Link |
|---|---|
| US (1) | US10874108B2 (en) |
| EP (1) | EP3451829B1 (en) |
| JP (1) | JP7020700B2 (en) |
| CN (1) | CN109414010B (en) |
| AU (1) | AU2017259304B2 (en) |
| DK (1) | DK3451829T3 (en) |
| ES (1) | ES2836299T3 (en) |
| GB (1) | GB201607814D0 (en) |
| SG (1) | SG11201809526YA (en) |
| WO (1) | WO2017191453A1 (en) |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2019172741A1 (en) * | 2018-03-09 | 2019-09-12 | Instituto Tecnológico José Mario Molina Pasquel y Henriquez | Bimetal complexes with divalent cations with an inhibiting effect on hsv replication |
| EP3556214A1 (en) * | 2018-04-19 | 2019-10-23 | Intracare B.V. | Methods for the prevention of infections of epithelial cells in an animal |
| WO2020154762A1 (en) * | 2019-01-28 | 2020-08-06 | Rr Medsciences Pty Ltd | Anti-inflammatory compositions and methods |
| WO2022023724A1 (en) | 2020-07-27 | 2022-02-03 | 5D Health Protection Group Ltd | Anti-microbial compositions |
| DE102022117861A1 (en) | 2022-07-18 | 2024-01-18 | Paul Hartmann Ag | Hydrogel to reduce biofilms |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN113600827B (en) * | 2021-07-30 | 2022-07-19 | 山东大学 | Au/Ga2O3/AuGa2Multiphase composite material and preparation method and application thereof |
| DE102024106518A1 (en) | 2024-03-07 | 2025-09-11 | Paul Hartmann Ag | Wound contact layer with anti-infective properties |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20020162800A1 (en) * | 2001-05-03 | 2002-11-07 | Back Dwight D. | Formulation and method for treating wetted surface elements in climate control systems |
| US20050048124A1 (en) * | 2003-08-26 | 2005-03-03 | Shantha Sarangapani | Antimicrobial composition for medical articles |
Family Cites Families (42)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5141803A (en) | 1988-06-29 | 1992-08-25 | Sterling Drug, Inc. | Nonwoven wipe impregnating composition |
| US5149354A (en) | 1991-01-10 | 1992-09-22 | Delaney Brendan J | Composition for treating swimming pools |
| US5362754A (en) | 1992-11-12 | 1994-11-08 | Univ. Of Tx Md Anderson Cancer Center | M-EDTA pharmaceutical preparations and uses thereof |
| US5688516A (en) | 1992-11-12 | 1997-11-18 | Board Of Regents, The University Of Texas System | Non-glycopeptide antimicrobial agents in combination with an anticoagulant, an antithrombotic or a chelating agent, and their uses in, for example, the preparation of medical devices |
| JP3297969B2 (en) | 1994-12-26 | 2002-07-02 | ライオン株式会社 | Eye drops |
| US5602180A (en) | 1995-03-31 | 1997-02-11 | World Health Group | Method of administering EDTA complexes |
| US6165484A (en) | 1997-08-26 | 2000-12-26 | Wake Forest University | EDTA and other chelators with or without antifungal antimicrobial agents for the prevention and treatment of fungal infections |
| US6056939A (en) | 1998-08-28 | 2000-05-02 | Desreux; Jean F. | Self-assembling heteropolymetallic chelates as imaging agents and radiopharmaceuticals |
| JP2002529545A (en) | 1998-11-06 | 2002-09-10 | ユニベルシテ ドゥ モントリオール | Improved germicidal and non-sterile solutions for removing biofilms |
| EP1593387B1 (en) | 1999-06-23 | 2008-11-19 | Institut Pasteur | Compositions for the treatment of impaired interpersonal and behavioral disorders |
| BR0011858A (en) | 1999-06-25 | 2002-07-02 | Arch Chem Inc | Antimicrobial composition |
| ATE424181T1 (en) | 1999-06-25 | 2009-03-15 | Procter & Gamble | TOPICAL ANTIMICROBIAL COMPOSITIONS |
| US6303039B1 (en) | 2000-03-06 | 2001-10-16 | Mainstream Engineering Corporation | Method for treating water over an extended time using tablets and packets |
| AU7999401A (en) * | 2000-08-28 | 2002-03-13 | Gavin Derek Mitchell | Trace elements |
| DE10051628B4 (en) | 2000-10-18 | 2007-06-06 | Fresenius Hemocare Beteiligungs Gmbh | Single-stranded oligonucleotide and its use |
| AU2002353061B2 (en) | 2001-12-05 | 2008-07-10 | Aseptica, Inc. | Anti-microbial systems and methods |
| US8541472B2 (en) | 2001-12-05 | 2013-09-24 | Aseptica, Inc. | Antiseptic compositions, methods and systems |
| WO2003086332A1 (en) | 2002-04-11 | 2003-10-23 | Dvm Pharmaceuticals, Inc. | Antimicrobial wash and carrier solutions, and uses thereof |
| EP1551429A4 (en) * | 2002-09-25 | 2008-04-16 | Edwin Odell Miner | Antiseptic solutions containing silver chelated with polypectate and edta |
| JP4244129B2 (en) | 2002-10-28 | 2009-03-25 | 株式会社メニコン | Disinfection method |
| US20080124368A1 (en) | 2004-08-25 | 2008-05-29 | Shantha Sarangapani | Composition |
| US20060045899A1 (en) | 2004-08-25 | 2006-03-02 | Shantha Sarangapani | Antimicrobial composition for medical articles |
| US8415280B2 (en) | 2005-01-18 | 2013-04-09 | Bestline International Research, Inc. | Universal synthetic penetrating lubricant, method and product-by-process |
| US8679537B2 (en) | 2005-08-24 | 2014-03-25 | Actamaz Surgical Materials, LLC | Methods for sealing an orifice in tissue using an aldol-crosslinked polymeric hydrogel adhesive |
| GB0525504D0 (en) | 2005-12-14 | 2006-01-25 | Bristol Myers Squibb Co | Antimicrobial composition |
| JP5153005B2 (en) | 2006-04-11 | 2013-02-27 | タイコ ヘルスケア グループ リミテッド パートナーシップ | Wound dressing containing antibacterial and chelating agents |
| AU2007355106A1 (en) | 2006-11-29 | 2008-12-18 | Foamix Ltd. | Foamable waterless compositions with modulating agents |
| EP2120561A4 (en) | 2007-02-19 | 2012-11-21 | Plurogen Therapeutics Inc | Compositions for treating biofilms and methods for using same |
| US20110189260A1 (en) | 2008-05-30 | 2011-08-04 | University Of Cincinnati | Use of zinc chelators to inhibit biofilm formation |
| AU2009279525B2 (en) | 2008-08-08 | 2015-04-09 | Smith & Nephew Inc. | Wound dressing of continuous fibers |
| GB201000916D0 (en) | 2010-01-21 | 2010-03-10 | Adv Med Solutions Ltd | Treatment of biofilms |
| US20140135372A1 (en) | 2010-02-02 | 2014-05-15 | Elliott Farber | Compositions and methods of treatment of inflammatory skin conditions using allantoin |
| US9089140B2 (en) | 2011-02-23 | 2015-07-28 | Sanikleen, Llc | Cleaning and sanitizing film |
| GB201105829D0 (en) | 2011-04-06 | 2011-05-18 | Convatec Technologies Inc | Antimicrobial compositions |
| EP2941128B1 (en) | 2012-12-20 | 2023-06-28 | Rajiv Bhushan | Antimicrobial compositions |
| WO2014134731A1 (en) | 2013-03-07 | 2014-09-12 | Kane Biotech Inc. | Antimicrobial-antibiofilm compositions and methods of use thereof |
| US9821063B2 (en) | 2013-04-16 | 2017-11-21 | University Of Cincinnati | Antimicrobial compositions of aminoglycosidic antibiotics and zinc ion chelators specifically formulated for enhanced inhibition of bacterial colonization and antibacterial efficacy |
| GB201308770D0 (en) * | 2013-05-15 | 2013-06-26 | Convatec Technologies Inc | Wound Dressing Comprising an Antimicrobial Composition |
| MX2016008795A (en) | 2014-01-03 | 2017-03-23 | Scioderm Inc | Allantoin compositions for treating inflammatory skin conditions. |
| WO2015123503A1 (en) | 2014-02-14 | 2015-08-20 | Scioderm, Inc. | Methods for treating burns using allantoin |
| US20160375078A1 (en) | 2015-06-24 | 2016-12-29 | Amphora Holdings, Inc. | Topical composition comprising natural ingredients for bruising or hematoma healing on skin and use thereof |
| EP3231452A1 (en) | 2016-04-11 | 2017-10-18 | DiCosmo, Frank | Wound irrigation solutions |
-
2016
- 2016-05-04 GB GBGB1607814.9A patent/GB201607814D0/en not_active Ceased
-
2017
- 2017-05-04 JP JP2019510479A patent/JP7020700B2/en active Active
- 2017-05-04 CN CN201780042081.3A patent/CN109414010B/en active Active
- 2017-05-04 WO PCT/GB2017/051243 patent/WO2017191453A1/en not_active Ceased
- 2017-05-04 ES ES17723475T patent/ES2836299T3/en active Active
- 2017-05-04 US US16/098,629 patent/US10874108B2/en active Active
- 2017-05-04 AU AU2017259304A patent/AU2017259304B2/en active Active
- 2017-05-04 EP EP17723475.4A patent/EP3451829B1/en active Active
- 2017-05-04 SG SG11201809526YA patent/SG11201809526YA/en unknown
- 2017-05-04 DK DK17723475.4T patent/DK3451829T3/en active
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20020162800A1 (en) * | 2001-05-03 | 2002-11-07 | Back Dwight D. | Formulation and method for treating wetted surface elements in climate control systems |
| US20050048124A1 (en) * | 2003-08-26 | 2005-03-03 | Shantha Sarangapani | Antimicrobial composition for medical articles |
Cited By (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2019172741A1 (en) * | 2018-03-09 | 2019-09-12 | Instituto Tecnológico José Mario Molina Pasquel y Henriquez | Bimetal complexes with divalent cations with an inhibiting effect on hsv replication |
| EP3556214A1 (en) * | 2018-04-19 | 2019-10-23 | Intracare B.V. | Methods for the prevention of infections of epithelial cells in an animal |
| NL2020799B1 (en) * | 2018-04-19 | 2019-10-28 | Intracare Bv | Methods for the prevention of mastitis |
| WO2020154762A1 (en) * | 2019-01-28 | 2020-08-06 | Rr Medsciences Pty Ltd | Anti-inflammatory compositions and methods |
| US12472207B2 (en) | 2019-01-28 | 2025-11-18 | Rr Medsciences Pty Ltd. | Anti-inflammatory compositions and methods |
| AU2020215661B2 (en) * | 2019-01-28 | 2025-12-18 | Rr Medsciences Pty Ltd | Anti-inflammatory compositions and methods |
| WO2022023724A1 (en) | 2020-07-27 | 2022-02-03 | 5D Health Protection Group Ltd | Anti-microbial compositions |
| DE102022117861A1 (en) | 2022-07-18 | 2024-01-18 | Paul Hartmann Ag | Hydrogel to reduce biofilms |
| WO2024017804A1 (en) | 2022-07-18 | 2024-01-25 | Paul Hartmann Ag | Hydrogel for the reduction of biofilms |
Also Published As
| Publication number | Publication date |
|---|---|
| ES2836299T3 (en) | 2021-06-24 |
| JP2019515961A (en) | 2019-06-13 |
| US10874108B2 (en) | 2020-12-29 |
| JP7020700B2 (en) | 2022-02-16 |
| US20190133131A1 (en) | 2019-05-09 |
| SG11201809526YA (en) | 2018-11-29 |
| GB201607814D0 (en) | 2016-06-15 |
| DK3451829T3 (en) | 2020-12-21 |
| CN109414010A (en) | 2019-03-01 |
| EP3451829B1 (en) | 2020-09-30 |
| EP3451829A1 (en) | 2019-03-13 |
| AU2017259304B2 (en) | 2021-04-29 |
| CN109414010B (en) | 2021-09-24 |
| AU2017259304A1 (en) | 2018-11-15 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US10874108B2 (en) | Anti-microbial compositions | |
| Zhou et al. | Antibacterial and wound healing–promoting effect of sponge-like chitosan-loaded silver nanoparticles biosynthesized by iturin | |
| Konop et al. | Certain aspects of silver and silver nanoparticles in wound care: a minireview | |
| Bigliardi et al. | An Asian perspective on povidone iodine in wound healing | |
| WO2020043665A1 (en) | Antiseptic gel | |
| CN113144281B (en) | Wound surface disinfection gel and preparation method thereof | |
| US20220347138A1 (en) | Wound care product | |
| US9723843B2 (en) | Family of silver (I) periodate compounds having broad microbial properties | |
| CA2692094C (en) | Antimicrobial compositions | |
| US20230312610A1 (en) | Anti-Microbial Compositions | |
| Wang et al. | Antimicrobial efficacy of composite irrigation solution against dominant pathogens in seawater immersion wound and in vivo wound healing assessment | |
| Qin | Antimicrobial textile dressings to manage wound infection | |
| RU2426558C1 (en) | Wound bandage with antimicrobial properties | |
| HK40005577A (en) | Anti-microbial compositions | |
| HK40005577B (en) | Anti-microbial compositions | |
| Reśliński et al. | The influence of octenidine dihydrochloride on bacterial biofilm on the surface of a polypropylene mesh | |
| Ip | Antimicrobial dressings | |
| US20260115372A1 (en) | Wound care product | |
| WO2025006425A2 (en) | Disinfectants with iodine and copper complexes for biofilm eradication and prevention | |
| WO2024138043A2 (en) | Zeolitic materials for the inhibition and disruption of biofilms, deactivation of viruses, and inhibition of fungal growth | |
| Edwards‐Jones | Antimicrobial dressings | |
| HK1094320B (en) | Antiseptic compositions, methods and systems |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| ENP | Entry into the national phase |
Ref document number: 2019510479 Country of ref document: JP Kind code of ref document: A |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2017259304 Country of ref document: AU Date of ref document: 20170504 Kind code of ref document: A |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 17723475 Country of ref document: EP Kind code of ref document: A1 |
|
| ENP | Entry into the national phase |
Ref document number: 2017723475 Country of ref document: EP Effective date: 20181204 |