WO2016014484A1 - Combination of broadly neutralizing hiv antibodies and viral inducers - Google Patents
Combination of broadly neutralizing hiv antibodies and viral inducers Download PDFInfo
- Publication number
- WO2016014484A1 WO2016014484A1 PCT/US2015/041272 US2015041272W WO2016014484A1 WO 2016014484 A1 WO2016014484 A1 WO 2016014484A1 US 2015041272 W US2015041272 W US 2015041272W WO 2016014484 A1 WO2016014484 A1 WO 2016014484A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- antibody
- hiv
- antibodies
- mice
- subject
- Prior art date
Links
- 239000000411 inducer Substances 0.000 title claims abstract description 70
- 230000003612 virological effect Effects 0.000 title description 43
- 230000003472 neutralizing effect Effects 0.000 title description 10
- 238000000034 method Methods 0.000 claims abstract description 61
- 230000006490 viral transcription Effects 0.000 claims abstract description 22
- 238000013518 transcription Methods 0.000 claims abstract description 11
- 230000035897 transcription Effects 0.000 claims abstract description 11
- 230000036436 anti-hiv Effects 0.000 claims abstract description 10
- 210000004027 cell Anatomy 0.000 claims description 65
- 210000001744 T-lymphocyte Anatomy 0.000 claims description 27
- 239000008194 pharmaceutical composition Substances 0.000 claims description 20
- WAEXFXRVDQXREF-UHFFFAOYSA-N vorinostat Chemical compound ONC(=O)CCCCCCC(=O)NC1=CC=CC=C1 WAEXFXRVDQXREF-UHFFFAOYSA-N 0.000 claims description 13
- 229960000237 vorinostat Drugs 0.000 claims description 13
- 230000002401 inhibitory effect Effects 0.000 claims description 12
- VUVUVNZRUGEAHB-CYBMUJFWSA-N 7-(3,5-dimethyl-4-isoxazolyl)-8-methoxy-1-[(1R)-1-(2-pyridinyl)ethyl]-3H-imidazo[4,5-c]quinolin-2-one Chemical compound C1([C@@H](C)N2C3=C4C=C(C(=CC4=NC=C3NC2=O)C2=C(ON=C2C)C)OC)=CC=CC=N1 VUVUVNZRUGEAHB-CYBMUJFWSA-N 0.000 claims description 10
- 230000037361 pathway Effects 0.000 claims description 8
- 239000003276 histone deacetylase inhibitor Substances 0.000 claims description 7
- 239000003443 antiviral agent Substances 0.000 claims description 6
- 230000003247 decreasing effect Effects 0.000 claims description 6
- 229940121372 histone deacetylase inhibitor Drugs 0.000 claims description 6
- 229940125763 bromodomain inhibitor Drugs 0.000 claims description 5
- 229940124158 Protease/peptidase inhibitor Drugs 0.000 claims description 4
- 229940125777 fusion inhibitor Drugs 0.000 claims description 4
- 229940124524 integrase inhibitor Drugs 0.000 claims description 4
- 239000002850 integrase inhibitor Substances 0.000 claims description 4
- 229940042402 non-nucleoside reverse transcriptase inhibitor Drugs 0.000 claims description 4
- 239000002726 nonnucleoside reverse transcriptase inhibitor Substances 0.000 claims description 4
- 239000000137 peptide hydrolase inhibitor Substances 0.000 claims description 4
- 241000713772 Human immunodeficiency virus 1 Species 0.000 abstract description 65
- 241000725303 Human immunodeficiency virus Species 0.000 abstract description 34
- 239000003795 chemical substances by application Substances 0.000 abstract description 15
- 241000699670 Mus sp. Species 0.000 description 89
- 206010058874 Viraemia Diseases 0.000 description 52
- 238000011225 antiretroviral therapy Methods 0.000 description 38
- 241000699666 Mus <mouse, genus> Species 0.000 description 29
- 239000000203 mixture Substances 0.000 description 28
- 230000027455 binding Effects 0.000 description 27
- 108020004414 DNA Proteins 0.000 description 25
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 25
- ZKVLEFBKBNUQHK-UHFFFAOYSA-N helium;molecular nitrogen;molecular oxygen Chemical compound [He].N#N.O=O ZKVLEFBKBNUQHK-UHFFFAOYSA-N 0.000 description 25
- 208000015181 infectious disease Diseases 0.000 description 25
- 238000011282 treatment Methods 0.000 description 24
- 108010087819 Fc receptors Proteins 0.000 description 23
- 102000009109 Fc receptors Human genes 0.000 description 23
- 239000000427 antigen Substances 0.000 description 22
- 108091007433 antigens Proteins 0.000 description 21
- 102000036639 antigens Human genes 0.000 description 21
- 108091032973 (ribonucleotides)n+m Proteins 0.000 description 20
- 239000012634 fragment Substances 0.000 description 18
- 238000002347 injection Methods 0.000 description 18
- 239000007924 injection Substances 0.000 description 18
- 238000002560 therapeutic procedure Methods 0.000 description 16
- 108010021625 Immunoglobulin Fragments Proteins 0.000 description 15
- 102000008394 Immunoglobulin Fragments Human genes 0.000 description 15
- 208000035475 disorder Diseases 0.000 description 15
- 230000000694 effects Effects 0.000 description 15
- 238000005516 engineering process Methods 0.000 description 15
- 108090000623 proteins and genes Proteins 0.000 description 15
- 102000004196 processed proteins & peptides Human genes 0.000 description 14
- 108090000765 processed proteins & peptides Proteins 0.000 description 14
- 230000001225 therapeutic effect Effects 0.000 description 14
- 238000002474 experimental method Methods 0.000 description 13
- 238000001727 in vivo Methods 0.000 description 13
- 235000018102 proteins Nutrition 0.000 description 13
- 102000004169 proteins and genes Human genes 0.000 description 13
- 230000002064 post-exposure prophylaxis Effects 0.000 description 12
- 102000005962 receptors Human genes 0.000 description 12
- 108020003175 receptors Proteins 0.000 description 12
- 125000000539 amino acid group Chemical group 0.000 description 11
- 229920001184 polypeptide Polymers 0.000 description 11
- 208000030507 AIDS Diseases 0.000 description 10
- 241000282412 Homo Species 0.000 description 10
- 238000010586 diagram Methods 0.000 description 10
- 201000010099 disease Diseases 0.000 description 10
- 239000003937 drug carrier Substances 0.000 description 10
- 208000031886 HIV Infections Diseases 0.000 description 9
- 108020000999 Viral RNA Proteins 0.000 description 9
- 241000700605 Viruses Species 0.000 description 9
- 125000003275 alpha amino acid group Chemical group 0.000 description 9
- 235000001014 amino acid Nutrition 0.000 description 9
- 238000009175 antibody therapy Methods 0.000 description 9
- 239000003814 drug Substances 0.000 description 9
- -1 i.e. Substances 0.000 description 9
- 210000003819 peripheral blood mononuclear cell Anatomy 0.000 description 9
- 210000000952 spleen Anatomy 0.000 description 9
- 101710149870 C-C chemokine receptor type 5 Proteins 0.000 description 8
- 102100035875 C-C chemokine receptor type 5 Human genes 0.000 description 8
- 241000282553 Macaca Species 0.000 description 8
- 241001465754 Metazoa Species 0.000 description 8
- 238000011577 humanized mouse model Methods 0.000 description 8
- 230000007246 mechanism Effects 0.000 description 8
- 108010047041 Complementarity Determining Regions Proteins 0.000 description 7
- 239000010282 Emodin Substances 0.000 description 7
- 108060003951 Immunoglobulin Proteins 0.000 description 7
- 239000013543 active substance Substances 0.000 description 7
- 229940024606 amino acid Drugs 0.000 description 7
- 238000003556 assay Methods 0.000 description 7
- 210000004369 blood Anatomy 0.000 description 7
- 239000008280 blood Substances 0.000 description 7
- 238000001514 detection method Methods 0.000 description 7
- RHMXXJGYXNZAPX-UHFFFAOYSA-N emodin Chemical compound C1=C(O)C=C2C(=O)C3=CC(C)=CC(O)=C3C(=O)C2=C1O RHMXXJGYXNZAPX-UHFFFAOYSA-N 0.000 description 7
- 238000000684 flow cytometry Methods 0.000 description 7
- 238000009472 formulation Methods 0.000 description 7
- 102000018358 immunoglobulin Human genes 0.000 description 7
- 238000005259 measurement Methods 0.000 description 7
- 230000004048 modification Effects 0.000 description 7
- 238000012986 modification Methods 0.000 description 7
- 239000000546 pharmaceutical excipient Substances 0.000 description 7
- 239000000523 sample Substances 0.000 description 7
- 101000738771 Homo sapiens Receptor-type tyrosine-protein phosphatase C Proteins 0.000 description 6
- 102100037422 Receptor-type tyrosine-protein phosphatase C Human genes 0.000 description 6
- 150000001413 amino acids Chemical class 0.000 description 6
- 230000007423 decrease Effects 0.000 description 6
- 230000014509 gene expression Effects 0.000 description 6
- 238000004519 manufacturing process Methods 0.000 description 6
- 230000035772 mutation Effects 0.000 description 6
- 230000002265 prevention Effects 0.000 description 6
- 239000000243 solution Substances 0.000 description 6
- 239000003981 vehicle Substances 0.000 description 6
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Chemical compound O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 6
- OUYCCCASQSFEME-QMMMGPOBSA-N L-tyrosine Chemical compound OC(=O)[C@@H](N)CC1=CC=C(O)C=C1 OUYCCCASQSFEME-QMMMGPOBSA-N 0.000 description 5
- 239000002202 Polyethylene glycol Substances 0.000 description 5
- 230000004913 activation Effects 0.000 description 5
- 230000004071 biological effect Effects 0.000 description 5
- 239000000969 carrier Substances 0.000 description 5
- 229940079593 drug Drugs 0.000 description 5
- 230000006870 function Effects 0.000 description 5
- 230000006872 improvement Effects 0.000 description 5
- 238000007912 intraperitoneal administration Methods 0.000 description 5
- 238000012423 maintenance Methods 0.000 description 5
- 229920001223 polyethylene glycol Polymers 0.000 description 5
- 241000894007 species Species 0.000 description 5
- 238000007920 subcutaneous administration Methods 0.000 description 5
- 239000000126 substance Substances 0.000 description 5
- OUYCCCASQSFEME-UHFFFAOYSA-N tyrosine Natural products OC(=O)C(N)CC1=CC=C(O)C=C1 OUYCCCASQSFEME-UHFFFAOYSA-N 0.000 description 5
- 101100112922 Candida albicans CDR3 gene Proteins 0.000 description 4
- DHMQDGOQFOQNFH-UHFFFAOYSA-N Glycine Chemical compound NCC(O)=O DHMQDGOQFOQNFH-UHFFFAOYSA-N 0.000 description 4
- 208000037357 HIV infectious disease Diseases 0.000 description 4
- 241000124008 Mammalia Species 0.000 description 4
- 238000011529 RT qPCR Methods 0.000 description 4
- 239000004480 active ingredient Substances 0.000 description 4
- 238000010171 animal model Methods 0.000 description 4
- 239000003153 chemical reaction reagent Substances 0.000 description 4
- 238000002648 combination therapy Methods 0.000 description 4
- 239000003085 diluting agent Substances 0.000 description 4
- 235000013305 food Nutrition 0.000 description 4
- 230000004927 fusion Effects 0.000 description 4
- 230000036541 health Effects 0.000 description 4
- 208000033519 human immunodeficiency virus infectious disease Diseases 0.000 description 4
- 210000000987 immune system Anatomy 0.000 description 4
- 238000000338 in vitro Methods 0.000 description 4
- 238000001990 intravenous administration Methods 0.000 description 4
- 210000004185 liver Anatomy 0.000 description 4
- 210000004698 lymphocyte Anatomy 0.000 description 4
- 230000001404 mediated effect Effects 0.000 description 4
- 230000002688 persistence Effects 0.000 description 4
- 239000002953 phosphate buffered saline Substances 0.000 description 4
- 230000000069 prophylactic effect Effects 0.000 description 4
- 230000002829 reductive effect Effects 0.000 description 4
- 238000006467 substitution reaction Methods 0.000 description 4
- 230000002459 sustained effect Effects 0.000 description 4
- VCMJCVGFSROFHV-WZGZYPNHSA-N tenofovir disoproxil fumarate Chemical compound OC(=O)\C=C\C(O)=O.N1=CN=C2N(C[C@@H](C)OCP(=O)(OCOC(=O)OC(C)C)OCOC(=O)OC(C)C)C=NC2=C1N VCMJCVGFSROFHV-WZGZYPNHSA-N 0.000 description 4
- 238000012360 testing method Methods 0.000 description 4
- 239000013598 vector Substances 0.000 description 4
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 3
- XQSPYNMVSIKCOC-NTSWFWBYSA-N Emtricitabine Chemical compound C1=C(F)C(N)=NC(=O)N1[C@H]1O[C@@H](CO)SC1 XQSPYNMVSIKCOC-NTSWFWBYSA-N 0.000 description 3
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 3
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Chemical compound OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 3
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 3
- 101100005713 Homo sapiens CD4 gene Proteins 0.000 description 3
- 108010073807 IgG Receptors Proteins 0.000 description 3
- 102000009490 IgG Receptors Human genes 0.000 description 3
- 102000006496 Immunoglobulin Heavy Chains Human genes 0.000 description 3
- 108010019476 Immunoglobulin Heavy Chains Proteins 0.000 description 3
- 102100034343 Integrase Human genes 0.000 description 3
- 108091007491 NSP3 Papain-like protease domains Proteins 0.000 description 3
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 3
- 108020005202 Viral DNA Proteins 0.000 description 3
- 230000003213 activating effect Effects 0.000 description 3
- 239000000872 buffer Substances 0.000 description 3
- 230000001413 cellular effect Effects 0.000 description 3
- 238000006243 chemical reaction Methods 0.000 description 3
- 239000002299 complementary DNA Substances 0.000 description 3
- 230000001086 cytosolic effect Effects 0.000 description 3
- 230000001419 dependent effect Effects 0.000 description 3
- 229960000366 emtricitabine Drugs 0.000 description 3
- 239000000839 emulsion Substances 0.000 description 3
- 230000001605 fetal effect Effects 0.000 description 3
- 210000004602 germ cell Anatomy 0.000 description 3
- 210000003958 hematopoietic stem cell Anatomy 0.000 description 3
- 230000008676 import Effects 0.000 description 3
- 238000001802 infusion Methods 0.000 description 3
- 238000002955 isolation Methods 0.000 description 3
- 239000000463 material Substances 0.000 description 3
- 239000003094 microcapsule Substances 0.000 description 3
- 210000005259 peripheral blood Anatomy 0.000 description 3
- 239000011886 peripheral blood Substances 0.000 description 3
- 238000002823 phage display Methods 0.000 description 3
- 230000001766 physiological effect Effects 0.000 description 3
- 230000003389 potentiating effect Effects 0.000 description 3
- 230000002028 premature Effects 0.000 description 3
- 229960004742 raltegravir Drugs 0.000 description 3
- CZFFBEXEKNGXKS-UHFFFAOYSA-N raltegravir Chemical compound O1C(C)=NN=C1C(=O)NC(C)(C)C1=NC(C(=O)NCC=2C=CC(F)=CC=2)=C(O)C(=O)N1C CZFFBEXEKNGXKS-UHFFFAOYSA-N 0.000 description 3
- 230000009467 reduction Effects 0.000 description 3
- 230000000284 resting effect Effects 0.000 description 3
- 230000002441 reversible effect Effects 0.000 description 3
- 238000012552 review Methods 0.000 description 3
- 210000004988 splenocyte Anatomy 0.000 description 3
- 208000024891 symptom Diseases 0.000 description 3
- 241001430294 unidentified retrovirus Species 0.000 description 3
- 241000238876 Acari Species 0.000 description 2
- 239000004475 Arginine Substances 0.000 description 2
- CIWBSHSKHKDKBQ-JLAZNSOCSA-N Ascorbic acid Chemical compound OC[C@H](O)[C@H]1OC(=O)C(O)=C1O CIWBSHSKHKDKBQ-JLAZNSOCSA-N 0.000 description 2
- DCXYFEDJOCDNAF-UHFFFAOYSA-N Asparagine Natural products OC(=O)C(N)CC(N)=O DCXYFEDJOCDNAF-UHFFFAOYSA-N 0.000 description 2
- 241000282693 Cercopithecidae Species 0.000 description 2
- 102000011022 Chorionic Gonadotropin Human genes 0.000 description 2
- 108010062540 Chorionic Gonadotropin Proteins 0.000 description 2
- 238000002965 ELISA Methods 0.000 description 2
- LVGKNOAMLMIIKO-UHFFFAOYSA-N Elaidinsaeure-aethylester Natural products CCCCCCCCC=CCCCCCCCC(=O)OCC LVGKNOAMLMIIKO-UHFFFAOYSA-N 0.000 description 2
- 241000588724 Escherichia coli Species 0.000 description 2
- 241000282326 Felis catus Species 0.000 description 2
- 229940126656 GS-4224 Drugs 0.000 description 2
- 239000004471 Glycine Substances 0.000 description 2
- 239000012981 Hank's balanced salt solution Substances 0.000 description 2
- 101000917826 Homo sapiens Low affinity immunoglobulin gamma Fc region receptor II-a Proteins 0.000 description 2
- 101000917824 Homo sapiens Low affinity immunoglobulin gamma Fc region receptor II-b Proteins 0.000 description 2
- 102000018071 Immunoglobulin Fc Fragments Human genes 0.000 description 2
- 108010091135 Immunoglobulin Fc Fragments Proteins 0.000 description 2
- 108700005091 Immunoglobulin Genes Proteins 0.000 description 2
- 238000012404 In vitro experiment Methods 0.000 description 2
- ODKSFYDXXFIFQN-BYPYZUCNSA-P L-argininium(2+) Chemical compound NC(=[NH2+])NCCC[C@H]([NH3+])C(O)=O ODKSFYDXXFIFQN-BYPYZUCNSA-P 0.000 description 2
- DCXYFEDJOCDNAF-REOHCLBHSA-N L-asparagine Chemical compound OC(=O)[C@@H](N)CC(N)=O DCXYFEDJOCDNAF-REOHCLBHSA-N 0.000 description 2
- ZDXPYRJPNDTMRX-VKHMYHEASA-N L-glutamine Chemical compound OC(=O)[C@@H](N)CCC(N)=O ZDXPYRJPNDTMRX-VKHMYHEASA-N 0.000 description 2
- HNDVDQJCIGZPNO-YFKPBYRVSA-N L-histidine Chemical compound OC(=O)[C@@H](N)CC1=CN=CN1 HNDVDQJCIGZPNO-YFKPBYRVSA-N 0.000 description 2
- AGPKZVBTJJNPAG-WHFBIAKZSA-N L-isoleucine Chemical compound CC[C@H](C)[C@H](N)C(O)=O AGPKZVBTJJNPAG-WHFBIAKZSA-N 0.000 description 2
- KDXKERNSBIXSRK-YFKPBYRVSA-N L-lysine Chemical compound NCCCC[C@H](N)C(O)=O KDXKERNSBIXSRK-YFKPBYRVSA-N 0.000 description 2
- COLNVLDHVKWLRT-QMMMGPOBSA-N L-phenylalanine Chemical compound OC(=O)[C@@H](N)CC1=CC=CC=C1 COLNVLDHVKWLRT-QMMMGPOBSA-N 0.000 description 2
- AYFVYJQAPQTCCC-GBXIJSLDSA-N L-threonine Chemical compound C[C@@H](O)[C@H](N)C(O)=O AYFVYJQAPQTCCC-GBXIJSLDSA-N 0.000 description 2
- QIVBCDIJIAJPQS-VIFPVBQESA-N L-tryptophane Chemical compound C1=CC=C2C(C[C@H](N)C(O)=O)=CNC2=C1 QIVBCDIJIAJPQS-VIFPVBQESA-N 0.000 description 2
- KZSNJWFQEVHDMF-BYPYZUCNSA-N L-valine Chemical compound CC(C)[C@H](N)C(O)=O KZSNJWFQEVHDMF-BYPYZUCNSA-N 0.000 description 2
- 102100029204 Low affinity immunoglobulin gamma Fc region receptor II-a Human genes 0.000 description 2
- KDXKERNSBIXSRK-UHFFFAOYSA-N Lysine Natural products NCCCCC(N)C(O)=O KDXKERNSBIXSRK-UHFFFAOYSA-N 0.000 description 2
- 239000004472 Lysine Substances 0.000 description 2
- 108010092799 RNA-directed DNA polymerase Proteins 0.000 description 2
- 241000580858 Simian-Human immunodeficiency virus Species 0.000 description 2
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 2
- AYFVYJQAPQTCCC-UHFFFAOYSA-N Threonine Natural products CC(O)C(N)C(O)=O AYFVYJQAPQTCCC-UHFFFAOYSA-N 0.000 description 2
- 239000004473 Threonine Substances 0.000 description 2
- QIVBCDIJIAJPQS-UHFFFAOYSA-N Tryptophan Natural products C1=CC=C2C(CC(N)C(O)=O)=CNC2=C1 QIVBCDIJIAJPQS-UHFFFAOYSA-N 0.000 description 2
- KZSNJWFQEVHDMF-UHFFFAOYSA-N Valine Natural products CC(C)C(N)C(O)=O KZSNJWFQEVHDMF-UHFFFAOYSA-N 0.000 description 2
- 239000000654 additive Substances 0.000 description 2
- 238000004458 analytical method Methods 0.000 description 2
- 229940124522 antiretrovirals Drugs 0.000 description 2
- 239000003903 antiretrovirus agent Substances 0.000 description 2
- 238000013459 approach Methods 0.000 description 2
- ODKSFYDXXFIFQN-UHFFFAOYSA-N arginine Natural products OC(=O)C(N)CCCNC(N)=N ODKSFYDXXFIFQN-UHFFFAOYSA-N 0.000 description 2
- 235000009582 asparagine Nutrition 0.000 description 2
- 229960001230 asparagine Drugs 0.000 description 2
- 210000003719 b-lymphocyte Anatomy 0.000 description 2
- 230000009286 beneficial effect Effects 0.000 description 2
- 230000008901 benefit Effects 0.000 description 2
- 230000037396 body weight Effects 0.000 description 2
- 210000004899 c-terminal region Anatomy 0.000 description 2
- 238000004113 cell culture Methods 0.000 description 2
- 230000022131 cell cycle Effects 0.000 description 2
- 230000008859 change Effects 0.000 description 2
- 238000010367 cloning Methods 0.000 description 2
- 230000007812 deficiency Effects 0.000 description 2
- 238000012217 deletion Methods 0.000 description 2
- 230000037430 deletion Effects 0.000 description 2
- 238000000432 density-gradient centrifugation Methods 0.000 description 2
- LOKCTEFSRHRXRJ-UHFFFAOYSA-I dipotassium trisodium dihydrogen phosphate hydrogen phosphate dichloride Chemical compound P(=O)(O)(O)[O-].[K+].P(=O)(O)([O-])[O-].[Na+].[Na+].[Cl-].[K+].[Cl-].[Na+] LOKCTEFSRHRXRJ-UHFFFAOYSA-I 0.000 description 2
- 239000006185 dispersion Substances 0.000 description 2
- 239000012636 effector Substances 0.000 description 2
- LVGKNOAMLMIIKO-QXMHVHEDSA-N ethyl oleate Chemical compound CCCCCCCC\C=C/CCCCCCCC(=O)OCC LVGKNOAMLMIIKO-QXMHVHEDSA-N 0.000 description 2
- 229940093471 ethyl oleate Drugs 0.000 description 2
- 239000013604 expression vector Substances 0.000 description 2
- 230000001815 facial effect Effects 0.000 description 2
- 238000010353 genetic engineering Methods 0.000 description 2
- ZDXPYRJPNDTMRX-UHFFFAOYSA-N glutamine Natural products OC(=O)C(N)CCC(N)=O ZDXPYRJPNDTMRX-UHFFFAOYSA-N 0.000 description 2
- HNDVDQJCIGZPNO-UHFFFAOYSA-N histidine Natural products OC(=O)C(N)CC1=CN=CN1 HNDVDQJCIGZPNO-UHFFFAOYSA-N 0.000 description 2
- 229940084986 human chorionic gonadotropin Drugs 0.000 description 2
- 210000004408 hybridoma Anatomy 0.000 description 2
- 230000028993 immune response Effects 0.000 description 2
- 230000016784 immunoglobulin production Effects 0.000 description 2
- 229940072221 immunoglobulins Drugs 0.000 description 2
- 230000001976 improved effect Effects 0.000 description 2
- CGIGDMFJXJATDK-UHFFFAOYSA-N indomethacin Chemical compound CC1=C(CC(O)=O)C2=CC(OC)=CC=C2N1C(=O)C1=CC=C(Cl)C=C1 CGIGDMFJXJATDK-UHFFFAOYSA-N 0.000 description 2
- 230000005764 inhibitory process Effects 0.000 description 2
- 238000007918 intramuscular administration Methods 0.000 description 2
- AGPKZVBTJJNPAG-UHFFFAOYSA-N isoleucine Natural products CCC(C)C(N)C(O)=O AGPKZVBTJJNPAG-UHFFFAOYSA-N 0.000 description 2
- 229960000310 isoleucine Drugs 0.000 description 2
- 210000000265 leukocyte Anatomy 0.000 description 2
- 238000012417 linear regression Methods 0.000 description 2
- 239000002502 liposome Substances 0.000 description 2
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 description 2
- 238000010369 molecular cloning Methods 0.000 description 2
- 238000006386 neutralization reaction Methods 0.000 description 2
- 231100000252 nontoxic Toxicity 0.000 description 2
- 230000003000 nontoxic effect Effects 0.000 description 2
- COLNVLDHVKWLRT-UHFFFAOYSA-N phenylalanine Natural products OC(=O)C(N)CC1=CC=CC=C1 COLNVLDHVKWLRT-UHFFFAOYSA-N 0.000 description 2
- 229920001451 polypropylene glycol Polymers 0.000 description 2
- 229920000136 polysorbate Polymers 0.000 description 2
- 239000000843 powder Substances 0.000 description 2
- 238000002360 preparation method Methods 0.000 description 2
- 239000003755 preservative agent Substances 0.000 description 2
- 238000002203 pretreatment Methods 0.000 description 2
- 239000011541 reaction mixture Substances 0.000 description 2
- 238000012163 sequencing technique Methods 0.000 description 2
- 239000011780 sodium chloride Substances 0.000 description 2
- 239000002904 solvent Substances 0.000 description 2
- 230000009870 specific binding Effects 0.000 description 2
- 210000004989 spleen cell Anatomy 0.000 description 2
- 239000003381 stabilizer Substances 0.000 description 2
- 238000010561 standard procedure Methods 0.000 description 2
- 238000007619 statistical method Methods 0.000 description 2
- 239000008223 sterile water Substances 0.000 description 2
- 238000003860 storage Methods 0.000 description 2
- 230000001629 suppression Effects 0.000 description 2
- 239000000725 suspension Substances 0.000 description 2
- 230000002195 synergetic effect Effects 0.000 description 2
- 229960004556 tenofovir Drugs 0.000 description 2
- 210000001519 tissue Anatomy 0.000 description 2
- 230000000699 topical effect Effects 0.000 description 2
- 238000012546 transfer Methods 0.000 description 2
- 230000009261 transgenic effect Effects 0.000 description 2
- 239000004474 valine Substances 0.000 description 2
- 210000003462 vein Anatomy 0.000 description 2
- MTCFGRXMJLQNBG-REOHCLBHSA-N (2S)-2-Amino-3-hydroxypropansäure Chemical compound OC[C@H](N)C(O)=O MTCFGRXMJLQNBG-REOHCLBHSA-N 0.000 description 1
- 102000040650 (ribonucleotides)n+m Human genes 0.000 description 1
- NHBKXEKEPDILRR-UHFFFAOYSA-N 2,3-bis(butanoylsulfanyl)propyl butanoate Chemical compound CCCC(=O)OCC(SC(=O)CCC)CSC(=O)CCC NHBKXEKEPDILRR-UHFFFAOYSA-N 0.000 description 1
- IDOQDZANRZQBTP-UHFFFAOYSA-N 2-[2-(2,4,4-trimethylpentan-2-yl)phenoxy]ethanol Chemical compound CC(C)(C)CC(C)(C)C1=CC=CC=C1OCCO IDOQDZANRZQBTP-UHFFFAOYSA-N 0.000 description 1
- JKMHFZQWWAIEOD-UHFFFAOYSA-N 2-[4-(2-hydroxyethyl)piperazin-1-yl]ethanesulfonic acid Chemical compound OCC[NH+]1CCN(CCS([O-])(=O)=O)CC1 JKMHFZQWWAIEOD-UHFFFAOYSA-N 0.000 description 1
- 108010088751 Albumins Proteins 0.000 description 1
- 102000009027 Albumins Human genes 0.000 description 1
- KHOITXIGCFIULA-UHFFFAOYSA-N Alophen Chemical compound C1=CC(OC(=O)C)=CC=C1C(C=1N=CC=CC=1)C1=CC=C(OC(C)=O)C=C1 KHOITXIGCFIULA-UHFFFAOYSA-N 0.000 description 1
- 229940088617 BET protein inhibitor Drugs 0.000 description 1
- VGGGPCQERPFHOB-MCIONIFRSA-N Bestatin Chemical compound CC(C)C[C@H](C(O)=O)NC(=O)[C@@H](O)[C@H](N)CC1=CC=CC=C1 VGGGPCQERPFHOB-MCIONIFRSA-N 0.000 description 1
- VGGGPCQERPFHOB-UHFFFAOYSA-N Bestatin Natural products CC(C)CC(C(O)=O)NC(=O)C(O)C(N)CC1=CC=CC=C1 VGGGPCQERPFHOB-UHFFFAOYSA-N 0.000 description 1
- 102000001805 Bromodomains Human genes 0.000 description 1
- 108050009021 Bromodomains Proteins 0.000 description 1
- 101100332641 Caenorhabditis elegans eat-4 gene Proteins 0.000 description 1
- 241000282832 Camelidae Species 0.000 description 1
- 241000700199 Cavia porcellus Species 0.000 description 1
- KRKNYBCHXYNGOX-UHFFFAOYSA-K Citrate Chemical compound [O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O KRKNYBCHXYNGOX-UHFFFAOYSA-K 0.000 description 1
- 108020004705 Codon Proteins 0.000 description 1
- 102000029816 Collagenase Human genes 0.000 description 1
- 108060005980 Collagenase Proteins 0.000 description 1
- 102000000989 Complement System Proteins Human genes 0.000 description 1
- 108010069112 Complement System Proteins Proteins 0.000 description 1
- 241000938605 Crocodylia Species 0.000 description 1
- 229920000858 Cyclodextrin Polymers 0.000 description 1
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 1
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 description 1
- FBPFZTCFMRRESA-JGWLITMVSA-N D-glucitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-JGWLITMVSA-N 0.000 description 1
- WQZGKKKJIJFFOK-QTVWNMPRSA-N D-mannopyranose Chemical compound OC[C@H]1OC(O)[C@@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-QTVWNMPRSA-N 0.000 description 1
- 102000053602 DNA Human genes 0.000 description 1
- 230000004544 DNA amplification Effects 0.000 description 1
- 102000007260 Deoxyribonuclease I Human genes 0.000 description 1
- 108010008532 Deoxyribonuclease I Proteins 0.000 description 1
- 102000016911 Deoxyribonucleases Human genes 0.000 description 1
- 108010053770 Deoxyribonucleases Proteins 0.000 description 1
- 239000004375 Dextrin Substances 0.000 description 1
- 229920001353 Dextrin Polymers 0.000 description 1
- BXZVVICBKDXVGW-NKWVEPMBSA-N Didanosine Chemical compound O1[C@H](CO)CC[C@@H]1N1C(NC=NC2=O)=C2N=C1 BXZVVICBKDXVGW-NKWVEPMBSA-N 0.000 description 1
- 101100117236 Drosophila melanogaster speck gene Proteins 0.000 description 1
- 206010013710 Drug interaction Diseases 0.000 description 1
- KCXVZYZYPLLWCC-UHFFFAOYSA-N EDTA Chemical compound OC(=O)CN(CC(O)=O)CCN(CC(O)=O)CC(O)=O KCXVZYZYPLLWCC-UHFFFAOYSA-N 0.000 description 1
- XPOQHMRABVBWPR-UHFFFAOYSA-N Efavirenz Natural products O1C(=O)NC2=CC=C(Cl)C=C2C1(C(F)(F)F)C#CC1CC1 XPOQHMRABVBWPR-UHFFFAOYSA-N 0.000 description 1
- 108010032976 Enfuvirtide Proteins 0.000 description 1
- 239000001116 FEMA 4028 Substances 0.000 description 1
- 241000282324 Felis Species 0.000 description 1
- 241000724791 Filamentous phage Species 0.000 description 1
- 238000000729 Fisher's exact test Methods 0.000 description 1
- 108010010803 Gelatin Proteins 0.000 description 1
- WHUUTDBJXJRKMK-UHFFFAOYSA-N Glutamic acid Natural products OC(=O)C(N)CCC(O)=O WHUUTDBJXJRKMK-UHFFFAOYSA-N 0.000 description 1
- 102000003886 Glycoproteins Human genes 0.000 description 1
- 108090000288 Glycoproteins Proteins 0.000 description 1
- 108010026389 Gramicidin Proteins 0.000 description 1
- 239000007995 HEPES buffer Substances 0.000 description 1
- 102100031573 Hematopoietic progenitor cell antigen CD34 Human genes 0.000 description 1
- 102000003964 Histone deacetylase Human genes 0.000 description 1
- 108090000353 Histone deacetylase Proteins 0.000 description 1
- 101000946926 Homo sapiens C-C chemokine receptor type 5 Proteins 0.000 description 1
- 101100220044 Homo sapiens CD34 gene Proteins 0.000 description 1
- 101000777663 Homo sapiens Hematopoietic progenitor cell antigen CD34 Proteins 0.000 description 1
- 102000008100 Human Serum Albumin Human genes 0.000 description 1
- 108091006905 Human Serum Albumin Proteins 0.000 description 1
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 1
- 102000013463 Immunoglobulin Light Chains Human genes 0.000 description 1
- 108010065825 Immunoglobulin Light Chains Proteins 0.000 description 1
- 108010067060 Immunoglobulin Variable Region Proteins 0.000 description 1
- 102000017727 Immunoglobulin Variable Region Human genes 0.000 description 1
- 108010061833 Integrases Proteins 0.000 description 1
- 238000012695 Interfacial polymerization Methods 0.000 description 1
- 238000012313 Kruskal-Wallis test Methods 0.000 description 1
- XUJNEKJLAYXESH-REOHCLBHSA-N L-Cysteine Chemical compound SC[C@H](N)C(O)=O XUJNEKJLAYXESH-REOHCLBHSA-N 0.000 description 1
- ONIBWKKTOPOVIA-BYPYZUCNSA-N L-Proline Chemical compound OC(=O)[C@@H]1CCCN1 ONIBWKKTOPOVIA-BYPYZUCNSA-N 0.000 description 1
- QNAYBMKLOCPYGJ-REOHCLBHSA-N L-alanine Chemical compound C[C@H](N)C(O)=O QNAYBMKLOCPYGJ-REOHCLBHSA-N 0.000 description 1
- CKLJMWTZIZZHCS-REOHCLBHSA-N L-aspartic acid Chemical compound OC(=O)[C@@H](N)CC(O)=O CKLJMWTZIZZHCS-REOHCLBHSA-N 0.000 description 1
- WHUUTDBJXJRKMK-VKHMYHEASA-N L-glutamic acid Chemical compound OC(=O)[C@@H](N)CCC(O)=O WHUUTDBJXJRKMK-VKHMYHEASA-N 0.000 description 1
- ROHFNLRQFUQHCH-YFKPBYRVSA-N L-leucine Chemical compound CC(C)C[C@H](N)C(O)=O ROHFNLRQFUQHCH-YFKPBYRVSA-N 0.000 description 1
- FFEARJCKVFRZRR-BYPYZUCNSA-N L-methionine Chemical compound CSCC[C@H](N)C(O)=O FFEARJCKVFRZRR-BYPYZUCNSA-N 0.000 description 1
- ROHFNLRQFUQHCH-UHFFFAOYSA-N Leucine Natural products CC(C)CC(N)C(O)=O ROHFNLRQFUQHCH-UHFFFAOYSA-N 0.000 description 1
- HLFSDGLLUJUHTE-SNVBAGLBSA-N Levamisole Chemical compound C1([C@H]2CN3CCSC3=N2)=CC=CC=C1 HLFSDGLLUJUHTE-SNVBAGLBSA-N 0.000 description 1
- 239000004907 Macro-emulsion Substances 0.000 description 1
- 229930195725 Mannitol Natural products 0.000 description 1
- OVRNDRQMDRJTHS-UHFFFAOYSA-N N-acelyl-D-glucosamine Natural products CC(=O)NC1C(O)OC(CO)C(O)C1O OVRNDRQMDRJTHS-UHFFFAOYSA-N 0.000 description 1
- OVRNDRQMDRJTHS-RTRLPJTCSA-N N-acetyl-D-glucosamine Chemical compound CC(=O)N[C@H]1C(O)O[C@H](CO)[C@@H](O)[C@@H]1O OVRNDRQMDRJTHS-RTRLPJTCSA-N 0.000 description 1
- MBLBDJOUHNCFQT-LXGUWJNJSA-N N-acetylglucosamine Natural products CC(=O)N[C@@H](C=O)[C@@H](O)[C@H](O)[C@H](O)CO MBLBDJOUHNCFQT-LXGUWJNJSA-N 0.000 description 1
- 230000004988 N-glycosylation Effects 0.000 description 1
- 229910019142 PO4 Inorganic materials 0.000 description 1
- 208000037581 Persistent Infection Diseases 0.000 description 1
- 206010057249 Phagocytosis Diseases 0.000 description 1
- 239000004698 Polyethylene Substances 0.000 description 1
- 241000288906 Primates Species 0.000 description 1
- ONIBWKKTOPOVIA-UHFFFAOYSA-N Proline Natural products OC(=O)C1CCCN1 ONIBWKKTOPOVIA-UHFFFAOYSA-N 0.000 description 1
- 108010076504 Protein Sorting Signals Proteins 0.000 description 1
- 238000013381 RNA quantification Methods 0.000 description 1
- 108020004511 Recombinant DNA Proteins 0.000 description 1
- 241001068263 Replication competent viruses Species 0.000 description 1
- NCDNCNXCDXHOMX-UHFFFAOYSA-N Ritonavir Natural products C=1C=CC=CC=1CC(NC(=O)OCC=1SC=NC=1)C(O)CC(CC=1C=CC=CC=1)NC(=O)C(C(C)C)NC(=O)N(C)CC1=CSC(C(C)C)=N1 NCDNCNXCDXHOMX-UHFFFAOYSA-N 0.000 description 1
- 241000283984 Rodentia Species 0.000 description 1
- 229920002684 Sepharose Polymers 0.000 description 1
- MTCFGRXMJLQNBG-UHFFFAOYSA-N Serine Natural products OCC(N)C(O)=O MTCFGRXMJLQNBG-UHFFFAOYSA-N 0.000 description 1
- 102000007562 Serum Albumin Human genes 0.000 description 1
- 108010071390 Serum Albumin Proteins 0.000 description 1
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 1
- DBMJMQXJHONAFJ-UHFFFAOYSA-M Sodium laurylsulphate Chemical compound [Na+].CCCCCCCCCCCCOS([O-])(=O)=O DBMJMQXJHONAFJ-UHFFFAOYSA-M 0.000 description 1
- 230000006044 T cell activation Effects 0.000 description 1
- 229940126530 T cell activator Drugs 0.000 description 1
- 108091008874 T cell receptors Proteins 0.000 description 1
- 102000016266 T-Cell Antigen Receptors Human genes 0.000 description 1
- SUJUHGSWHZTSEU-UHFFFAOYSA-N Tipranavir Natural products C1C(O)=C(C(CC)C=2C=C(NS(=O)(=O)C=3N=CC(=CC=3)C(F)(F)F)C=CC=2)C(=O)OC1(CCC)CCC1=CC=CC=C1 SUJUHGSWHZTSEU-UHFFFAOYSA-N 0.000 description 1
- 101710120037 Toxin CcdB Proteins 0.000 description 1
- 229920004929 Triton X-114 Polymers 0.000 description 1
- 241000251539 Vertebrata <Metazoa> Species 0.000 description 1
- 208000036142 Viral infection Diseases 0.000 description 1
- WREGKURFCTUGRC-POYBYMJQSA-N Zalcitabine Chemical compound O=C1N=C(N)C=CN1[C@@H]1O[C@H](CO)CC1 WREGKURFCTUGRC-POYBYMJQSA-N 0.000 description 1
- 230000002378 acidificating effect Effects 0.000 description 1
- 238000007792 addition Methods 0.000 description 1
- 239000002671 adjuvant Substances 0.000 description 1
- 230000002411 adverse Effects 0.000 description 1
- 230000009824 affinity maturation Effects 0.000 description 1
- 235000004279 alanine Nutrition 0.000 description 1
- LSQZJLSUYDQPKJ-NJBDSQKTSA-N amoxicillin Chemical compound C1([C@@H](N)C(=O)N[C@H]2[C@H]3SC([C@@H](N3C2=O)C(O)=O)(C)C)=CC=C(O)C=C1 LSQZJLSUYDQPKJ-NJBDSQKTSA-N 0.000 description 1
- 229960003022 amoxicillin Drugs 0.000 description 1
- 229960001830 amprenavir Drugs 0.000 description 1
- YMARZQAQMVYCKC-OEMFJLHTSA-N amprenavir Chemical compound C([C@@H]([C@H](O)CN(CC(C)C)S(=O)(=O)C=1C=CC(N)=CC=1)NC(=O)O[C@@H]1COCC1)C1=CC=CC=C1 YMARZQAQMVYCKC-OEMFJLHTSA-N 0.000 description 1
- 210000004102 animal cell Anatomy 0.000 description 1
- 239000002259 anti human immunodeficiency virus agent Substances 0.000 description 1
- 229940124411 anti-hiv antiviral agent Drugs 0.000 description 1
- 230000000798 anti-retroviral effect Effects 0.000 description 1
- 230000010056 antibody-dependent cellular cytotoxicity Effects 0.000 description 1
- 210000000612 antigen-presenting cell Anatomy 0.000 description 1
- 239000003963 antioxidant agent Substances 0.000 description 1
- 235000006708 antioxidants Nutrition 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- 238000009246 art therapy Methods 0.000 description 1
- 125000003118 aryl group Chemical group 0.000 description 1
- 229960005070 ascorbic acid Drugs 0.000 description 1
- 235000010323 ascorbic acid Nutrition 0.000 description 1
- 239000011668 ascorbic acid Substances 0.000 description 1
- 235000003704 aspartic acid Nutrition 0.000 description 1
- 238000013475 authorization Methods 0.000 description 1
- 238000002819 bacterial display Methods 0.000 description 1
- 230000004888 barrier function Effects 0.000 description 1
- 210000003651 basophil Anatomy 0.000 description 1
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 1
- OQFSQFPPLPISGP-UHFFFAOYSA-N beta-carboxyaspartic acid Natural products OC(=O)C(N)C(C(O)=O)C(O)=O OQFSQFPPLPISGP-UHFFFAOYSA-N 0.000 description 1
- WHGYBXFWUBPSRW-FOUAGVGXSA-N beta-cyclodextrin Chemical compound OC[C@H]([C@H]([C@@H]([C@H]1O)O)O[C@H]2O[C@@H]([C@@H](O[C@H]3O[C@H](CO)[C@H]([C@@H]([C@H]3O)O)O[C@H]3O[C@H](CO)[C@H]([C@@H]([C@H]3O)O)O[C@H]3O[C@H](CO)[C@H]([C@@H]([C@H]3O)O)O[C@H]3O[C@H](CO)[C@H]([C@@H]([C@H]3O)O)O3)[C@H](O)[C@H]2O)CO)O[C@@H]1O[C@H]1[C@H](O)[C@@H](O)[C@@H]3O[C@@H]1CO WHGYBXFWUBPSRW-FOUAGVGXSA-N 0.000 description 1
- 235000011175 beta-cyclodextrine Nutrition 0.000 description 1
- 229960004853 betadex Drugs 0.000 description 1
- 230000000975 bioactive effect Effects 0.000 description 1
- 238000002306 biochemical method Methods 0.000 description 1
- 230000005540 biological transmission Effects 0.000 description 1
- 238000005460 biophysical method Methods 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 230000000903 blocking effect Effects 0.000 description 1
- 210000000988 bone and bone Anatomy 0.000 description 1
- 239000008366 buffered solution Substances 0.000 description 1
- 150000001720 carbohydrates Chemical class 0.000 description 1
- 235000014633 carbohydrates Nutrition 0.000 description 1
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 1
- 239000011111 cardboard Substances 0.000 description 1
- 238000013216 cat model Methods 0.000 description 1
- 210000003855 cell nucleus Anatomy 0.000 description 1
- 108091092356 cellular DNA Proteins 0.000 description 1
- 230000002032 cellular defenses Effects 0.000 description 1
- 239000001913 cellulose Substances 0.000 description 1
- 229920002678 cellulose Polymers 0.000 description 1
- 235000010980 cellulose Nutrition 0.000 description 1
- 239000002738 chelating agent Substances 0.000 description 1
- 230000001684 chronic effect Effects 0.000 description 1
- 238000011260 co-administration Methods 0.000 description 1
- 230000004186 co-expression Effects 0.000 description 1
- 238000005354 coacervation Methods 0.000 description 1
- 229960002424 collagenase Drugs 0.000 description 1
- 230000000052 comparative effect Effects 0.000 description 1
- 230000004154 complement system Effects 0.000 description 1
- 150000001875 compounds Chemical class 0.000 description 1
- 238000010276 construction Methods 0.000 description 1
- 238000007796 conventional method Methods 0.000 description 1
- 229920001577 copolymer Polymers 0.000 description 1
- 238000004132 cross linking Methods 0.000 description 1
- 239000012228 culture supernatant Substances 0.000 description 1
- 235000018417 cysteine Nutrition 0.000 description 1
- XUJNEKJLAYXESH-UHFFFAOYSA-N cysteine Natural products SCC(N)C(O)=O XUJNEKJLAYXESH-UHFFFAOYSA-N 0.000 description 1
- 231100000433 cytotoxic Toxicity 0.000 description 1
- 229940127089 cytotoxic agent Drugs 0.000 description 1
- 239000002254 cytotoxic agent Substances 0.000 description 1
- 231100000599 cytotoxic agent Toxicity 0.000 description 1
- 230000001472 cytotoxic effect Effects 0.000 description 1
- 230000003013 cytotoxicity Effects 0.000 description 1
- 231100000135 cytotoxicity Toxicity 0.000 description 1
- 229960005107 darunavir Drugs 0.000 description 1
- CJBJHOAVZSMMDJ-HEXNFIEUSA-N darunavir Chemical compound C([C@@H]([C@H](O)CN(CC(C)C)S(=O)(=O)C=1C=CC(N)=CC=1)NC(=O)O[C@@H]1[C@@H]2CCO[C@@H]2OC1)C1=CC=CC=C1 CJBJHOAVZSMMDJ-HEXNFIEUSA-N 0.000 description 1
- 238000007405 data analysis Methods 0.000 description 1
- 230000003111 delayed effect Effects 0.000 description 1
- 210000004443 dendritic cell Anatomy 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 235000019425 dextrin Nutrition 0.000 description 1
- 229960002656 didanosine Drugs 0.000 description 1
- 230000029087 digestion Effects 0.000 description 1
- 239000000539 dimer Substances 0.000 description 1
- 150000002016 disaccharides Chemical class 0.000 description 1
- 239000012153 distilled water Substances 0.000 description 1
- 238000012377 drug delivery Methods 0.000 description 1
- XPOQHMRABVBWPR-ZDUSSCGKSA-N efavirenz Chemical compound C([C@]1(C2=CC(Cl)=CC=C2NC(=O)O1)C(F)(F)F)#CC1CC1 XPOQHMRABVBWPR-ZDUSSCGKSA-N 0.000 description 1
- 229960003804 efavirenz Drugs 0.000 description 1
- 239000002158 endotoxin Substances 0.000 description 1
- PEASPLKKXBYDKL-FXEVSJAOSA-N enfuvirtide Chemical compound C([C@@H](C(=O)N[C@@H](CO)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CO)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](C)C(=O)N[C@@H](CO)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CC=1C=CC=CC=1)C(N)=O)NC(=O)[C@@H](NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(C)=O)[C@@H](C)O)[C@@H](C)CC)C1=CN=CN1 PEASPLKKXBYDKL-FXEVSJAOSA-N 0.000 description 1
- 229960002062 enfuvirtide Drugs 0.000 description 1
- 244000309457 enveloped RNA virus Species 0.000 description 1
- 210000003979 eosinophil Anatomy 0.000 description 1
- 230000008029 eradication Effects 0.000 description 1
- 229940011871 estrogen Drugs 0.000 description 1
- 239000000262 estrogen Substances 0.000 description 1
- PYGWGZALEOIKDF-UHFFFAOYSA-N etravirine Chemical compound CC1=CC(C#N)=CC(C)=C1OC1=NC(NC=2C=CC(=CC=2)C#N)=NC(N)=C1Br PYGWGZALEOIKDF-UHFFFAOYSA-N 0.000 description 1
- 229960002049 etravirine Drugs 0.000 description 1
- 239000000284 extract Substances 0.000 description 1
- 230000002349 favourable effect Effects 0.000 description 1
- 239000012997 ficoll-paque Substances 0.000 description 1
- 238000001943 fluorescence-activated cell sorting Methods 0.000 description 1
- 210000000285 follicular dendritic cell Anatomy 0.000 description 1
- 238000012395 formulation development Methods 0.000 description 1
- 238000002825 functional assay Methods 0.000 description 1
- 108020001507 fusion proteins Proteins 0.000 description 1
- 102000037865 fusion proteins Human genes 0.000 description 1
- 229920000159 gelatin Polymers 0.000 description 1
- 239000008273 gelatin Substances 0.000 description 1
- 235000019322 gelatine Nutrition 0.000 description 1
- 235000011852 gelatine desserts Nutrition 0.000 description 1
- 238000003500 gene array Methods 0.000 description 1
- 238000007429 general method Methods 0.000 description 1
- 239000011521 glass Substances 0.000 description 1
- 239000008103 glucose Substances 0.000 description 1
- 235000013922 glutamic acid Nutrition 0.000 description 1
- 239000004220 glutamic acid Substances 0.000 description 1
- 230000013595 glycosylation Effects 0.000 description 1
- 238000006206 glycosylation reaction Methods 0.000 description 1
- 229960004905 gramicidin Drugs 0.000 description 1
- ZWCXYZRRTRDGQE-SORVKSEFSA-N gramicidina Chemical compound C1=CC=C2C(C[C@H](NC(=O)[C@@H](CC(C)C)NC(=O)[C@H](CC=3C4=CC=CC=C4NC=3)NC(=O)[C@@H](CC(C)C)NC(=O)[C@H](CC=3C4=CC=CC=C4NC=3)NC(=O)[C@@H](CC(C)C)NC(=O)[C@H](CC=3C4=CC=CC=C4NC=3)NC(=O)[C@H](C(C)C)NC(=O)[C@H](C(C)C)NC(=O)[C@@H](C(C)C)NC(=O)[C@H](C)NC(=O)[C@H](NC(=O)[C@H](C)NC(=O)CNC(=O)[C@@H](NC=O)C(C)C)CC(C)C)C(=O)NCCO)=CNC2=C1 ZWCXYZRRTRDGQE-SORVKSEFSA-N 0.000 description 1
- 229940093915 gynecological organic acid Drugs 0.000 description 1
- 210000003630 histaminocyte Anatomy 0.000 description 1
- 230000009675 homeostatic proliferation Effects 0.000 description 1
- 102000048160 human CCR5 Human genes 0.000 description 1
- 210000005260 human cell Anatomy 0.000 description 1
- 229920001477 hydrophilic polymer Polymers 0.000 description 1
- 229920003063 hydroxymethyl cellulose Polymers 0.000 description 1
- 229940031574 hydroxymethyl cellulose Drugs 0.000 description 1
- 230000001900 immune effect Effects 0.000 description 1
- 239000012642 immune effector Substances 0.000 description 1
- 230000036039 immunity Effects 0.000 description 1
- 230000002998 immunogenetic effect Effects 0.000 description 1
- 229940027941 immunoglobulin g Drugs 0.000 description 1
- 229940121354 immunomodulator Drugs 0.000 description 1
- 238000009169 immunotherapy Methods 0.000 description 1
- 229960001936 indinavir Drugs 0.000 description 1
- CBVCZFGXHXORBI-PXQQMZJSSA-N indinavir Chemical compound C([C@H](N(CC1)C[C@@H](O)C[C@@H](CC=2C=CC=CC=2)C(=O)N[C@H]2C3=CC=CC=C3C[C@H]2O)C(=O)NC(C)(C)C)N1CC1=CC=CN=C1 CBVCZFGXHXORBI-PXQQMZJSSA-N 0.000 description 1
- 229960000905 indomethacin Drugs 0.000 description 1
- 230000001939 inductive effect Effects 0.000 description 1
- 239000003112 inhibitor Substances 0.000 description 1
- 108091008042 inhibitory receptors Proteins 0.000 description 1
- 239000007928 intraperitoneal injection Substances 0.000 description 1
- 238000007919 intrasynovial administration Methods 0.000 description 1
- 238000007913 intrathecal administration Methods 0.000 description 1
- 238000005304 joining Methods 0.000 description 1
- 229960001614 levamisole Drugs 0.000 description 1
- 230000000670 limiting effect Effects 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- 238000011068 loading method Methods 0.000 description 1
- 210000001165 lymph node Anatomy 0.000 description 1
- 210000002540 macrophage Anatomy 0.000 description 1
- 235000019359 magnesium stearate Nutrition 0.000 description 1
- 239000000594 mannitol Substances 0.000 description 1
- 235000010355 mannitol Nutrition 0.000 description 1
- 229960004710 maraviroc Drugs 0.000 description 1
- GSNHKUDZZFZSJB-QYOOZWMWSA-N maraviroc Chemical compound CC(C)C1=NN=C(C)N1[C@@H]1C[C@H](N2CC[C@H](NC(=O)C3CCC(F)(F)CC3)C=3C=CC=CC=3)CC[C@H]2C1 GSNHKUDZZFZSJB-QYOOZWMWSA-N 0.000 description 1
- 210000003071 memory t lymphocyte Anatomy 0.000 description 1
- 239000002184 metal Substances 0.000 description 1
- 229910052751 metal Inorganic materials 0.000 description 1
- 229930182817 methionine Natural products 0.000 description 1
- 229920000609 methyl cellulose Polymers 0.000 description 1
- 239000001923 methylcellulose Substances 0.000 description 1
- 239000004530 micro-emulsion Substances 0.000 description 1
- 230000000813 microbial effect Effects 0.000 description 1
- 239000004005 microsphere Substances 0.000 description 1
- 239000000178 monomer Substances 0.000 description 1
- 150000002772 monosaccharides Chemical class 0.000 description 1
- 238000010172 mouse model Methods 0.000 description 1
- 238000002703 mutagenesis Methods 0.000 description 1
- 231100000350 mutagenesis Toxicity 0.000 description 1
- 239000002088 nanocapsule Substances 0.000 description 1
- 239000002105 nanoparticle Substances 0.000 description 1
- 239000007922 nasal spray Substances 0.000 description 1
- 229940097496 nasal spray Drugs 0.000 description 1
- 210000000822 natural killer cell Anatomy 0.000 description 1
- 238000007857 nested PCR Methods 0.000 description 1
- 210000000440 neutrophil Anatomy 0.000 description 1
- 239000012457 nonaqueous media Substances 0.000 description 1
- 239000002687 nonaqueous vehicle Substances 0.000 description 1
- 239000002736 nonionic surfactant Substances 0.000 description 1
- 230000009871 nonspecific binding Effects 0.000 description 1
- 239000000346 nonvolatile oil Substances 0.000 description 1
- 235000021590 normal diet Nutrition 0.000 description 1
- 108020004707 nucleic acids Proteins 0.000 description 1
- 102000039446 nucleic acids Human genes 0.000 description 1
- 150000007523 nucleic acids Chemical class 0.000 description 1
- 239000002773 nucleotide Substances 0.000 description 1
- 125000003729 nucleotide group Chemical group 0.000 description 1
- 239000004006 olive oil Substances 0.000 description 1
- 235000008390 olive oil Nutrition 0.000 description 1
- 238000005457 optimization Methods 0.000 description 1
- 238000003305 oral gavage Methods 0.000 description 1
- 239000000668 oral spray Substances 0.000 description 1
- 229940041678 oral spray Drugs 0.000 description 1
- 150000007524 organic acids Chemical class 0.000 description 1
- 235000005985 organic acids Nutrition 0.000 description 1
- 150000002895 organic esters Chemical class 0.000 description 1
- LSQZJLSUYDQPKJ-UHFFFAOYSA-N p-Hydroxyampicillin Natural products O=C1N2C(C(O)=O)C(C)(C)SC2C1NC(=O)C(N)C1=CC=C(O)C=C1 LSQZJLSUYDQPKJ-UHFFFAOYSA-N 0.000 description 1
- 239000005022 packaging material Substances 0.000 description 1
- 230000000242 pagocytic effect Effects 0.000 description 1
- 238000007911 parenteral administration Methods 0.000 description 1
- 239000003182 parenteral nutrition solution Substances 0.000 description 1
- 239000002245 particle Substances 0.000 description 1
- 244000052769 pathogen Species 0.000 description 1
- 230000008506 pathogenesis Effects 0.000 description 1
- 230000001717 pathogenic effect Effects 0.000 description 1
- 230000001575 pathological effect Effects 0.000 description 1
- 239000008188 pellet Substances 0.000 description 1
- 230000008782 phagocytosis Effects 0.000 description 1
- 230000000144 pharmacologic effect Effects 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 1
- 239000010452 phosphate Substances 0.000 description 1
- 230000036470 plasma concentration Effects 0.000 description 1
- 239000013612 plasmid Substances 0.000 description 1
- 239000004033 plastic Substances 0.000 description 1
- 229920003023 plastic Polymers 0.000 description 1
- 108700004029 pol Genes Proteins 0.000 description 1
- 101150088264 pol gene Proteins 0.000 description 1
- 229920001983 poloxamer Polymers 0.000 description 1
- 229920000642 polymer Polymers 0.000 description 1
- 229920005862 polyol Polymers 0.000 description 1
- 150000003077 polyols Chemical class 0.000 description 1
- 239000001267 polyvinylpyrrolidone Substances 0.000 description 1
- 229920000036 polyvinylpyrrolidone Polymers 0.000 description 1
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 description 1
- 230000029279 positive regulation of transcription, DNA-dependent Effects 0.000 description 1
- 231100000683 possible toxicity Toxicity 0.000 description 1
- 230000002335 preservative effect Effects 0.000 description 1
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 1
- 230000002035 prolonged effect Effects 0.000 description 1
- 230000001737 promoting effect Effects 0.000 description 1
- 238000011321 prophylaxis Methods 0.000 description 1
- 230000009993 protective function Effects 0.000 description 1
- 238000002818 protein evolution Methods 0.000 description 1
- 238000001742 protein purification Methods 0.000 description 1
- 230000002797 proteolythic effect Effects 0.000 description 1
- 230000005180 public health Effects 0.000 description 1
- 238000010926 purge Methods 0.000 description 1
- 230000007420 reactivation Effects 0.000 description 1
- 230000001105 regulatory effect Effects 0.000 description 1
- 238000009877 rendering Methods 0.000 description 1
- 230000010076 replication Effects 0.000 description 1
- 230000000717 retained effect Effects 0.000 description 1
- 238000002702 ribosome display Methods 0.000 description 1
- 229960000311 ritonavir Drugs 0.000 description 1
- NCDNCNXCDXHOMX-XGKFQTDJSA-N ritonavir Chemical compound N([C@@H](C(C)C)C(=O)N[C@H](C[C@H](O)[C@H](CC=1C=CC=CC=1)NC(=O)OCC=1SC=NC=1)CC=1C=CC=CC=1)C(=O)N(C)CC1=CSC(C(C)C)=N1 NCDNCNXCDXHOMX-XGKFQTDJSA-N 0.000 description 1
- 238000012216 screening Methods 0.000 description 1
- 210000002966 serum Anatomy 0.000 description 1
- 230000035939 shock Effects 0.000 description 1
- 125000005629 sialic acid group Chemical group 0.000 description 1
- 231100000161 signs of toxicity Toxicity 0.000 description 1
- 229910052814 silicon oxide Inorganic materials 0.000 description 1
- 238000002741 site-directed mutagenesis Methods 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 235000019333 sodium laurylsulphate Nutrition 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 239000000600 sorbitol Substances 0.000 description 1
- 230000000087 stabilizing effect Effects 0.000 description 1
- 238000010186 staining Methods 0.000 description 1
- 210000000130 stem cell Anatomy 0.000 description 1
- 150000005846 sugar alcohols Chemical class 0.000 description 1
- 230000008685 targeting Effects 0.000 description 1
- 229960004693 tenofovir disoproxil fumarate Drugs 0.000 description 1
- 235000021195 test diet Nutrition 0.000 description 1
- 229940124597 therapeutic agent Drugs 0.000 description 1
- 231100001274 therapeutic index Toxicity 0.000 description 1
- 238000011285 therapeutic regimen Methods 0.000 description 1
- 210000001541 thymus gland Anatomy 0.000 description 1
- 229960000838 tipranavir Drugs 0.000 description 1
- SUJUHGSWHZTSEU-FYBSXPHGSA-N tipranavir Chemical compound C([C@@]1(CCC)OC(=O)C([C@H](CC)C=2C=C(NS(=O)(=O)C=3N=CC(=CC=3)C(F)(F)F)C=CC=2)=C(O)C1)CC1=CC=CC=C1 SUJUHGSWHZTSEU-FYBSXPHGSA-N 0.000 description 1
- 231100000331 toxic Toxicity 0.000 description 1
- 230000002588 toxic effect Effects 0.000 description 1
- 108091006106 transcriptional activators Proteins 0.000 description 1
- 230000001052 transient effect Effects 0.000 description 1
- 238000013519 translation Methods 0.000 description 1
- 229950009811 ubenimex Drugs 0.000 description 1
- 229960005486 vaccine Drugs 0.000 description 1
- 235000015112 vegetable and seed oil Nutrition 0.000 description 1
- 239000008158 vegetable oil Substances 0.000 description 1
- 230000009264 viral breakthrough Effects 0.000 description 1
- 230000009385 viral infection Effects 0.000 description 1
- 239000000080 wetting agent Substances 0.000 description 1
- HBOMLICNUCNMMY-XLPZGREQSA-N zidovudine Chemical compound O=C1NC(=O)C(C)=CN1[C@@H]1O[C@H](CO)[C@@H](N=[N+]=[N-])C1 HBOMLICNUCNMMY-XLPZGREQSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/08—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from viruses
- C07K16/10—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from viruses from RNA viruses
- C07K16/1036—Retroviridae, e.g. leukemia viruses
- C07K16/1045—Lentiviridae, e.g. HIV, FIV, SIV
- C07K16/1063—Lentiviridae, e.g. HIV, FIV, SIV env, e.g. gp41, gp110/120, gp160, V3, PND, CD4 binding site
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/16—Amides, e.g. hydroxamic acids
- A61K31/165—Amides, e.g. hydroxamic acids having aromatic rings, e.g. colchicine, atenolol, progabide
- A61K31/167—Amides, e.g. hydroxamic acids having aromatic rings, e.g. colchicine, atenolol, progabide having the nitrogen of a carboxamide group directly attached to the aromatic ring, e.g. lidocaine, paracetamol
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/185—Acids; Anhydrides, halides or salts thereof, e.g. sulfur acids, imidic, hydrazonic or hydroximic acids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/505—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim
- A61K31/513—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim having oxo groups directly attached to the heterocyclic ring, e.g. cytosine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/66—Phosphorus compounds
- A61K31/683—Diesters of a phosphorus acid with two hydroxy compounds, e.g. phosphatidylinositols
- A61K31/685—Diesters of a phosphorus acid with two hydroxy compounds, e.g. phosphatidylinositols one of the hydroxy compounds having nitrogen atoms, e.g. phosphatidylserine, lecithin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/395—Antibodies; Immunoglobulins; Immune serum, e.g. antilymphocytic serum
- A61K39/42—Antibodies; Immunoglobulins; Immune serum, e.g. antilymphocytic serum viral
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K45/00—Medicinal preparations containing active ingredients not provided for in groups A61K31/00 - A61K41/00
- A61K45/06—Mixtures of active ingredients without chemical characterisation, e.g. antiphlogistics and cardiaca
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/505—Medicinal preparations containing antigens or antibodies comprising antibodies
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/30—Immunoglobulins specific features characterized by aspects of specificity or valency
- C07K2317/33—Crossreactivity, e.g. for species or epitope, or lack of said crossreactivity
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/50—Immunoglobulins specific features characterized by immunoglobulin fragments
- C07K2317/52—Constant or Fc region; Isotype
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/70—Immunoglobulins specific features characterized by effect upon binding to a cell or to an antigen
- C07K2317/71—Decreased effector function due to an Fc-modification
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/70—Immunoglobulins specific features characterized by effect upon binding to a cell or to an antigen
- C07K2317/76—Antagonist effect on antigen, e.g. neutralization or inhibition of binding
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/90—Immunoglobulins specific features characterized by (pharmaco)kinetic aspects or by stability of the immunoglobulin
- C07K2317/94—Stability, e.g. half-life, pH, temperature or enzyme-resistance
Definitions
- This invention relates to methods and agents for prevention or disruption of the establishment or maintenance of human immunodeficiency virus latent reservoirs.
- HIV Human immunodeficiency virus
- AIDS acquired immunodeficiency syndrome
- HIV-1 is the most common and pathogenic strain of the virus.
- ART combination anti-retroviral therapies
- ART termination follows by rapid viral rebound (Davey, et al., Proc. Natl. Acad. Sci. U.S.AA999. pp. 15109- 15114).
- This invention relates to using broadly neutralizing antibodies (bNAbs) alone or in combination with viral transcription inducers in preventing the establishment of the latent reservoirs of HIV-1 infected cells or decreasing the size of the reservoir, and thereby addresses the need mentioned above.
- bNAbs broadly neutralizing antibodies
- one aspect of this invention provides a method for decreasing the size of or preventing the establishment of a latent reservoir of HIV infected cells (e.g., a cell population comprising HIV-infected CD4 + T cells.) in a subject in need thereof.
- the method includes administering to the subject a therapeutically effective amount of an isolated anti-HIV antibody, e.g., a bNAb, and administering to the subject two or more viral transcription inducers in effective amounts to induce transcription of an HIV provirus in the cells.
- an isolated anti-HIV antibody e.g., a bNAb
- the antibody can be a human antibody, a humanized antibody, or a chimeric antibody.
- the antibody is antibody 3BNC1 17, 10-1074, or PG16, or any other described below, or combination thereof.
- two or three of the antibodies 3BNC1 17, 10-1074, and PG16 are administered to the subject.
- the antibody or antibodies can be administered to the subject within about 96 hours or earlier (e.g., 72, 48, 36, 24, or 12 hours) after exposure or suspected exposure to HIV.
- the transcription inducers can be administered to the subject for a period of about 5-14 days (e.g., 6-13, 7-12, 8, 9, 10, 1 1 , 12 days).
- transcription inducers examples include vorinostat, an HDAC inhibitor, I-BET151 , a BET bromodomain inhibitor, and aCTLA4, a T-cell inhibitory pathway blocker.
- the method can further comprise administering to the subject an antiviral agent, such as one selected from the group consisting of a non-nucleoside reverse transcriptase inhibitor, a protease inhibitor, an entry or fusion inhibitor, and an integrase inhibitor.
- the invention provides a kit comprising an isolated anti-HIV antibody, a first viral transcription inducer, and a second viral transcription inducer.
- the kit can contain one, two, three, or more of antibodies 3BNC1 17, 10-1074, PG16, and those described below.
- the antibody can be a human antibody, a humanized antibody, or a chimeric antibody.
- the transcription inducers can be selected from the group consisting of vorinostat, an HDAC inhibitor, I-BET151 , a BET bromodomain inhibitor, and aCTLA4, a T-cell inhibitory pathway blocker.
- the first viral transcription inducer and the second viral transcription inducer can be contained in one pharmaceutical composition or in two separate pharmaceutical compositions.
- the kit can further contain an antiviral agent, which can be selected from the group consisting of a non-nucleoside reverse transcriptase inhibitor, a protease inhibitor, an entry or fusion inhibitor, and an integrase inhibitor.
- an antiviral agent which can be selected from the group consisting of a non-nucleoside reverse transcriptase inhibitor, a protease inhibitor, an entry or fusion inhibitor, and an integrase inhibitor.
- the kit can be used in the method of this invention.
- the invention features a method for preventing the establishment of a latent reservoir of HIV infected cells in a subject in need thereof.
- the method includes administering to the subject a therapeutically effective amount of an isolated anti- HIV antibody.
- the antibody can be a human antibody, a humanized antibody, or a chimeric antibody.
- the antibody is antibody 3BNC1 17, 10-1074, PG16, any of those described below, or a combination thereof.
- two, three, or more of the antibodies are administered to the subject.
- the antibody or antibodies can be administered to the subject within about 96 hours or earlier (e.g., 72, 48, 36, 24, or 12 hours) after exposure or suspected exposure to HIV.
- FIG. 1A, FIG. IB, FIG. 1C, FIG. ID, FIG. IE, FIG. IF, AND FIG. 1H are a set of diagrams showing post-exposure prophylaxis with bNAbs:
- FIG. 1A Schematic timeline for the bNAb (top) and ART (bottom) experiments.
- FIG. IB Plasma viremia for untreated mice. The x-axis is in days post HIV-1 challenge. The y-axis is viral RNA copies/ml. Gray shading indicates values beneath the detection limit of 800 copies/ml. The blue line indicates the geometric mean plasma viremia.
- FIG. 1C Plasma viremia for ART -treated mice as in FIG. IB.
- FIG. ID Plasma viremia for antibody-treated mice.
- the red arrows indicate antibody tri-mix injections.
- the dashed red line shows average plasma antibody concentration for all mice in the group.
- FIG. IE Graph as in FIG. ID, for mice treated with antibody starting 8 days after HIV-1 challenge.
- FIG. IF Percentage of CD4 + T cells in the spleen at the terminal point measured by flow cytometry, organized by treatment group.
- FIG. 1G Cell-associated HIV-1 RNA measured in spleen T cells at the terminal point, plotted as the ratio of HIV-1 RNA to CCR5 copies for each mouse.
- Gray shading indicates the detection limit of 1.25xl0 ⁇ 5 copies per cell.
- FIG. 1H Cell-associated HIV-1 DNA measured in spleen T cells at the terminal point, plotted as the ratio of HIV-1 RNA to CCR5 copies for each mouse. Gray shading indicates the detection limit of 2.0x10 "5 copies per cell.
- FIG. 2A, FIG. 2B, FIG. 2C, and FIG. 2D are a set of diagrams showing that FcR nu11 antibodies suppress viremia but do not prevent rebound:
- FIG. 2A Plasma viremia as in FIG. ID for mice treated with FcR nu11 tri-mix.
- FIG. 2B The proportion of mice that were viremic at the terminal point for each treatment group (*, p ⁇ 0.05; Fisher's Exact test).
- FIG. 2C For all viremic mice, plasma antibody concentration on the day of viral rebound. Antibody levels were significantly higher in FcR nu11 tri-mix treated mice compared to wild-type tri-mix treated mice (*,p ⁇ 0.05; **, p ⁇ 0.01; Mann- Whitney test).
- FIG. 2D Sequences of gpl20 cloned from plasma. Horizontal lines denote individual clones, grouped by mouse, shown on the right. Red ticks and green ticks indicate non- synonymous and synonymous substitutions relative to gpl20yu 2 , respectively. Blue shading highlights sites of mutations that confer escape to the antibody tri-mix.
- FIG. 3A, FIG. 3B, FIG. 3C, FIG. 3D, and FIG. 3E are a set of diagrams showing rebound viremia after therapy with single inducers:
- FIG. 3A Schematic timeline of the experiment.
- FIG. 3B, FIG. 3C, and FIG. 3D Graphs show plasma viremia for individual mice on the left y-axis, geometric mean antibody level on the right y-axis among all mice in the group (red). The x-axis represents days relative to the first antibody injection. Antibody injections are indicated with red arrows. Mice that had rebound plasma viremia are shown in gray. Mice that failed to rebound are shown in black.
- FIG. 3B Mice that received tri-mix antibodies, but no inducers.
- FIG. 3C Mice that received tri-mix antibodies and vorinostat (green arrows).
- FIG. 3D Mice that received tri-mix antibodies and I-BET151 (purple shading).
- FIG. 3E Mice that received tri-mix antibodies and aCTLA4 (orange arrows).
- FIG. 4A and FIG. 4B are a set of diagrams showing that combination inducers decrease the incidence of rebound viremia:
- FIG. 4A Mice treated with tri-mix of antibodies, and a combination of three inducers. Graph, arrows, and shading are as in FIG. 3.
- FIG. 4B Graph shows the proportion of mice that showed rebound viremia for each treatment group, where all mice that received antibody tri-mix and any one of the three single inducers (shown in FIGs. 3C, 3D, and 3E) are pooled together (*, p ⁇ 0.05; Mann- Whitney test).
- FIG. 5A, FIG. 5B, FIG. 5C, FIG. 5D, FIG. 5E, FIG. 5F, and FIG. 5G are a set of diagrams showing antibody persistence and premature termination do not account for non- rebounding:
- FIG. 5A Percentage of CD4 + T cells at the terminal point measured in the spleen by flow cytometry.
- FIG. 5B Cell-associated HIV-1 RNA measured in spleen cells at terminal point, plotted as the ratio of HIV-1 RNA to CCR5 DNA copies for each mouse. Mice that had measureable HIV-1 RNA, but undetectable CCR5 DNA are plotted as 10 4 copies per cell.
- FIG. 5A Percentage of CD4 + T cells at the terminal point measured in the spleen by flow cytometry.
- FIG. 5B Cell-associated HIV-1 RNA measured in spleen cells at terminal point, plotted as the ratio of HIV-1 RNA to CCR5 DNA copies for each mouse. Mice that had measureable HIV-1 RNA, but undetectable C
- FIG. 5D The plasma antibody level at the time of viral rebound for each mouse that rebounded, organized by treatment group. The mean plasma antibody level at the time of rebound was 2.97 ⁇ g/ml for all groups.
- FIG. 5E For each mouse that rebounded, the number of days that elapsed from when the antibody level dropped below 2.97 ⁇ g/ml to the time of rebound.
- FIG. 5G Cell-associated HIV-1 DNA measured in spleen T cells at the terminal point, plotted as the ratio of HIV-1 DNA to CCR5 copies for each mouse. Mice that had measureable HIV-1 DNA, but undetectable CCR5 DNA are plotted as 10 4 copies per cell.
- FIG. 6A and FIG. 6B are set of diagrams showing viremia and antibody levels in individual mice (bNAb Post-exposure prophylaxis) as in FIG. ID: (FIG. 6 A) Each mouse shown in FIG. ID is shown individually. (FIG. 6B) Each mouse shown in FIG. IE is shown individually.
- FIG. 7 is a set of diagram showing viremia and antibody levels in individual mice (FcR nu11 tri -mix Post-exposure prophylaxis) as in FIG. ID. Each mouse shown in FIG. 2A is shown individually.
- FIG. 8A, FIG. 8B, FIG. 8C, and FIG. 8D are a set of diagram showing viremia and antibody levels in individual mice (antibody only and antibody plus single inducers) in FIGs. 3A-3D:
- FIG. 8A Each mouse shown in Fig. 3A is shown individually.
- FIG. 8B Each mouse shown in FIG. 3B is shown individually.
- FIG. 8C Each mouse shown in FIG. 3C is shown individually.
- FIG. 8D Each mouse shown in FIG. 3D is shown individually.
- FIG. 9 is a set of diagram showing viremia in individual mice (antibody plus combination inducers) as in FIG. 4A. Each mouse shown in FIG. 4A is shown individually.
- FIG. 10A, FIG. 10B, and FIG. 10 are a set of diagram showing sustained viremia in inducers treated mice without therapy:
- FIG. 10A Graphs shown as in FIG. 4A. The blue line indicates the geometric mean viremia across all mice.
- FIG. 10B Proportion of human CD45 + leukocytes in peripheral blood, measured by flow cytometry.
- FIG. IOC Percentage of CD4 + T cells in the peripheral blood measured by flow cytometry. DETAILED DESCRIPTION OF THE INVENTION
- This invention is based, at least in part, on unexpected discoveries (i) that bNAbs are more effective than ART in preventing the establishment of the latent reservoirs of HIV-1 infected cells by a mechanism that requires Fc receptor function and (ii) that bNAbs in combination with viral transcription inducers that act by independent mechanisms synergize to decrease the size of the reservoir as measured by prevention of viral rebound.
- combinations of viral transcription inducers and bNAbs constitute a therapeutic strategy that impacts the establishment and maintenance of the HIV-1 reservoir.
- HIV-1 viruses are transmitted as single-stranded, positive-sense, enveloped RNA viruses.
- the viral RNA genome Upon entry into a target host cell, the viral RNA genome is reverse transcribed into double-stranded DNA by a virally encoded reverse transcriptase that is transported along with the viral genome in the virus particle.
- the resulting viral DNA is then imported into the cell nucleus and integrated into the cellular DNA by a virally encoded integrase and host co-factors. Once integrated, the virus may become latent, allowing the virus and its host cell to avoid detection by the immune system. HIV-1 persists in infected individuals in a stable pool or reservoir of resting CD4 + T cells and other cells as a latent but replication-competent provirus.
- latent reservoir refers to cells or sites in a host that are latently infected with a microbe (e.g., HIV). Its presence and size of a HIV latent reservoir can be measured or assessed by viral rebound after terminating therapy as disclosed in the examples below.
- a microbe e.g., HIV
- Such a latent reservoir of HIV-1 infected cells cannot be cleared by combined ART and remains the major barrier to curing HIV-1 infection.
- the latent reservoir In humans, macaques and humanized mice, the latent reservoir is established within days of initial infection and persists for the lifetime of the individual, making ART a lifelong necessity. While the latent reservoir's exact cellular composition is debated, it is generally believed that it consists primarily of CD4 + memory T cells harboring replication competent provirus that is transcriptionally silenced. Because these cells have very long half-lives and may be able to undergo homeostatic proliferation, a "shock and kill" approach has been proposed to eradicate this reservoir by combining ART with inducers of viral transcription. However, all attempts at altering the HIV-1 reservoir have failed to date.
- bNAbs can be used in preventing the establishment of the reservoir by a mechanism that requires Fc receptor function.
- bNAbs plus single inducers are ineffective in decreasing the reservoir size.
- combinations of inducers and bNAbs constitute a therapeutic strategy that can be used to prevent or disrupt the establishment and maintenance of the HIV-1 reservoir in humans and related animal models.
- the invention disclosed herein involves broadly neutralizing HIV-1 antibodies. These antibodies refer to a class of neutralizing antibodies that neutralize multiple HIV-1 viral strains.
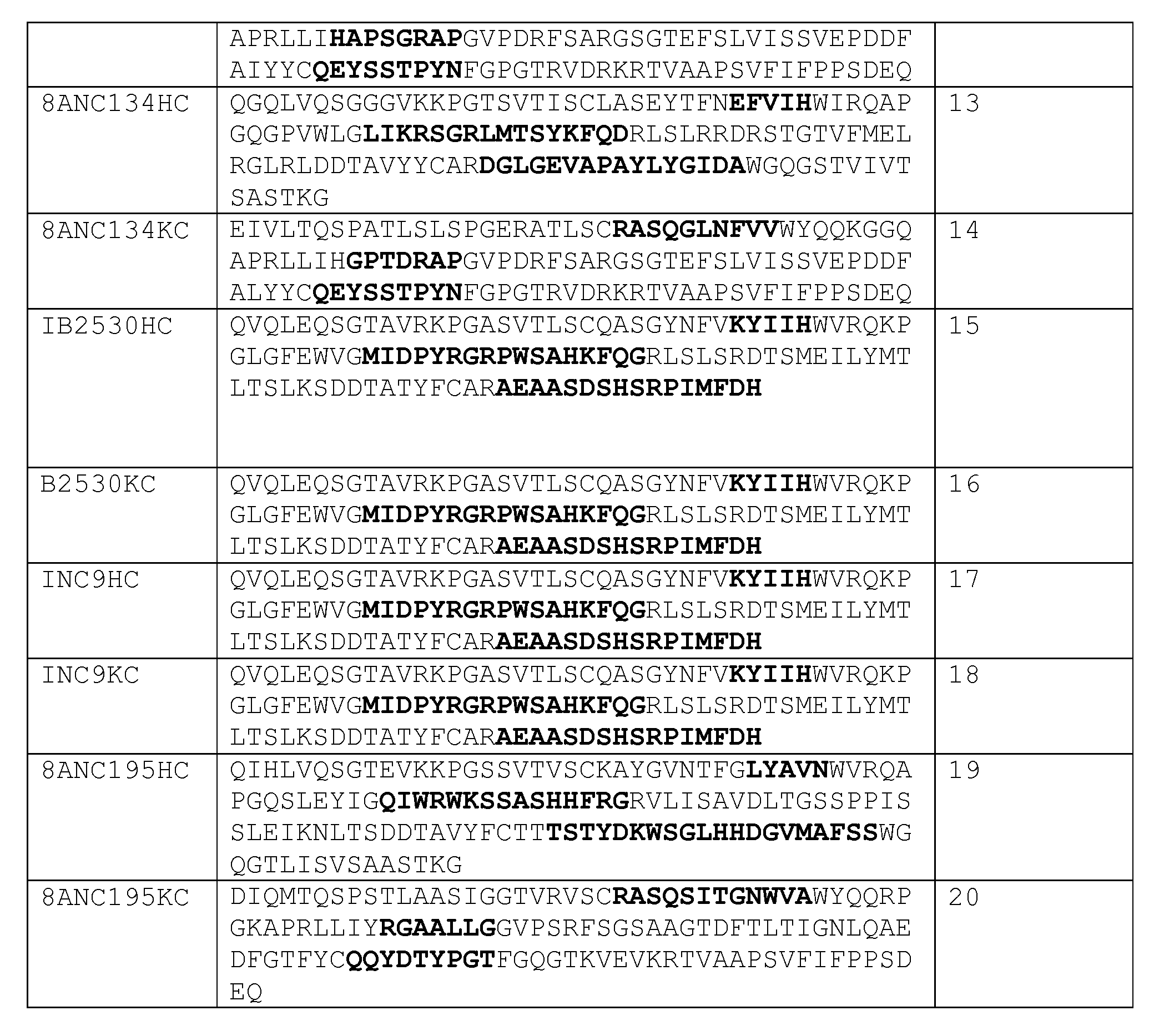
- Various bNAbs are known in the art and can be used in this invention. Examples include but are not limited to those described in U.S. Patent NO. 8673307, WO 2014063059, WO2012158948, and U.S. Provisional Application 61/934,359, including antibodies 3BNC117, 3BNC60, 12A12, 12A21, NIH45-46, bANC131, 8ANC134, IB2530, INC9, 8ANC195.
- HC heavy chain variable regions
- LC light chain variable regions
- the heavy chain variable regions (IgH) and light chain variable regions (IgL) of additional antibodies are listed below, where the CDRs are in bold.
- bNAbs can completely suppress viremia in HIV-1 infected humanized mice (Klein et al, Nature, 2012. 492(7427): p. 118-22 and Horwitz et al, Proc Natl Acad Sci U S A, 2013. 110(41): p. 16538-43) and in SHIV infected macaques (Barouch et al, Nature, 2013. 503(7475): p. 224-8 and Shingai et al, Nature, 2013. 503(7475): p. 277-80).
- antibodies can engage the host immune system by virtue of their Fc effector domains (Nimmerjahn et al, Nat Rev Immunol, 2008. 8(1): p. 34-47) and thereby accelerate clearance of cell free virus (Igarashi et al, Nat Med, 1999. 5(2): p. 211- 6), induce antibody dependent cytotoxicity to kill infected cells (Chung et al, Proc Natl Acad Sci U S A, 2011. 108(18): p. 7505-10; Sun et al, J Virol, 2011. 85(14): p. 6906- 12 Bonsignori et al, J Virol, 2012. 86(21): p.
- antibody includes monoclonal antibodies, polyclonal antibodies, multispecific antibodies (for example, bispecific antibodies and polyreactive antibodies), and antibody fragments.
- an antibody as used in any context within this specification is meant to include, but not be limited to, any specific binding member, immunoglobulin class and/or isotype (e.g., IgGl , IgG2, IgG3, IgG4, IgM, IgA, IgD, IgE and IgM); and biologically relevant fragment or specific binding member thereof, including but not limited to Fab, F(ab')2, Fv, and scFv (single chain or related entity).
- an antibody is a glycoprotein comprising at least two heavy (H) chains and two light (L) chains inter-connected by disulfide bonds, or an antigen binding portion thereof.
- a heavy chain is comprised of a heavy chain variable region (VH) and a heavy chain constant region (CHI , CH2 and CH3).
- a light chain is comprised of a light chain variable region (VL) and a light chain constant region (CL).
- the variable regions of both the heavy and light chains comprise framework regions (FWR) and complementarity determining regions (CDR).
- the four FWR regions are relatively conserved while CDR regions (CDR1 , CDR2 and CDR3) represent hypervariable regions and are arranged from NH2 terminus to the COOH terminus as follows: FWR1 , CDR1 , FWR2, CDR2, FWR3, CDR3, and FWR4.
- the variable regions of the heavy and light chains contain a binding domain that interacts with an antigen while, depending of the isotype, the constant region(s) may mediate the binding of the immunoglobulin to host tissues or factors.
- antibody as used herein are chimeric antibodies, humanized antibodies, and recombinant antibodies, human antibodies generated from a transgenic non-human animal, as well as antibodies selected from libraries using enrichment technologies available to the artisan.
- variable refers to the fact that certain segments of the variable (V) domains differ extensively in sequence among antibodies.
- the V domain mediates antigen binding and defines specificity of a particular antibody for its particular antigen.
- variability is not evenly distributed across the 1 10-amino acid span of the variable regions.
- the V regions consist of relatively invariant stretches called framework regions (FRs) of 15-30 amino acids separated by shorter regions of extreme variability called “hypervariable regions” that are each 9-12 amino acids long.
- FRs framework regions
- hypervariable regions that are each 9-12 amino acids long.
- the variable regions of native heavy and light chains each comprise four FRs, largely adopting a beta sheet configuration, connected by three hypervariable regions, which form loops connecting, and in some cases forming part of, the beta sheet structure.
- the hypervariable regions in each chain are held together in close proximity by the FRs and, with the hypervariable regions from the other chain, contribute to the formation of the antigen- binding site of antibodies (see, for example, Kabat et al., Sequences of Proteins of Immunological Interest, 5th Ed. Public Health Service, National Institutes of Health, Bethesda, Md. (1991)).
- hypervariable region refers to the amino acid residues of an antibody that are responsible for antigen binding.
- the hypervariable region generally comprises amino acid residues from a “complementarity determining region” (“CDR").
- the term "monoclonal antibody” as used herein refers to an antibody obtained from a population of substantially homogeneous antibodies, i.e., the individual antibodies comprising the population are identical except for possible naturally occurring mutations that may be present in minor amounts.
- polyclonal antibody refers to preparations that include different antibodies directed against different determinants ("epitopes").
- the monoclonal antibodies herein include "chimeric" antibodies in which a portion of the heavy and/or light chain is identical with, or homologous to, corresponding sequences in antibodies derived from a particular species or belonging to a particular antibody class or subclass, while the remainder of the chain(s) is identical with, or homologous to, corresponding sequences in antibodies derived from another species or belonging to another antibody class or subclass, as well as fragments of such antibodies, so long as they exhibit the desired biological activity (see, for example, U.S. Pat. No. 4,816,567; and Morrison et al, Proc. Natl. Acad. Sci. USA, 81 :6851-6855 (1984)).
- Chimeric antibodies include antibodies having one or more human antigen binding sequences (for example, CDRs) and containing one or more sequences derived from a non-human antibody, for example, an FR or C region sequence.
- chimeric antibodies included herein are those comprising a human variable region antigen binding sequence of one antibody class or subclass and another sequence, for example, FR or C region sequence, derived from another antibody class or subclass.
- a "humanized antibody” generally is considered to be a human antibody that has one or more amino acid residues introduced into it from a source that is non-human. These non-human amino acid residues often are referred to as "import" residues, which typically are taken from an "import” variable region.
- Humanization may be performed following the method of Winter and co-workers (see, for example, Jones et al, Nature 321 :522-525 (1986); Reichmann et al, Nature 1> ⁇ >2: ⁇ >2 ⁇ >- ⁇ >2 ⁇ (1988); Verhoeyen et al, Science 239: 1534-1536 (1988)), by substituting import hypervariable region sequences for the corresponding sequences of a human antibody. Accordingly, such "humanized" antibodies are chimeric antibodies (see, for example, U.S. Pat. No. 4,816,567), where substantially less than an intact human variable region has been substituted by the corresponding sequence from a non-human species.
- antibody fragment comprises a portion of an intact antibody, such as the antigen binding or variable region of the intact antibody.
- antibody fragments include, but are not limited to, Fab, Fab', F(ab')2, and Fv fragments; diabodies; linear antibodies (see, for example, U.S. Pat. No. 5,641,870; Zapata et al, Protein Eng. 8(10): 1057-1062 [1995]); single-chain antibody molecules; and multispecific antibodies formed from antibody fragments.
- Fv is the minimum antibody fragment that contains a complete antigen- recognition and antigen-binding site. This fragment contains a dimer of one heavy- and one light-chain variable region domain in tight, non-covalent association. From the folding of these two domains emanate six hypervariable loops (three loops each from the H and L chain) that contribute the amino acid residues for antigen binding and confer antigen binding specificity to the antibody. However, even a single variable region (or half of an Fv comprising only three CDRs specific for an antigen) has the ability to recognize and bind antigen, although at a lower affinity than the entire binding site.
- Single-chain Fv (“sFv” or “scFv”) are antibody fragments that comprise the VH and VL antibody domains connected into a single polypeptide chain.
- the sFv polypeptide can further comprise a polypeptide linker between the VH and VL domains that enables the sFv to form the desired structure for antigen binding.
- sFv Single-chain Fv
- diabodies refers to small antibody fragments prepared by constructing sFv fragments with short linkers (about 5-10 residues) between the VH and VL domains such that inter-chain but not intra-chain pairing of the V domains is achieved, resulting in a bivalent fragment, i.e., fragment having two antigen-binding sites.
- Bispecific diabodies are heterodimers of two "crossover" sFv fragments in which the VH and VL domains of the two antibodies are present on different polypeptide chains.
- Diabodies are described more fully in, for example, EP 404,097; WO 93/11161; and Hollinger et al, Proc. Natl. Acad. Sci. USA, 90:6444-6448 (1993).
- DAbs Domain antibodies
- VH and VL immunoglobulins
- DAbs are the robust variable regions of the heavy and light chains of immunoglobulins (VH and VL, respectively). They are highly expressed in microbial cell culture, show favorable biophysical properties including, for example, but not limited to, solubility and temperature stability, and are well suited to selection and affinity maturation by in vitro selection systems such as, for example, phage display.
- DAbs are bioactive as monomers and, owing to their small size and inherent stability, can be formatted into larger molecules to create drugs with prolonged serum half-lives or other pharmacological activities. Examples of this technology have been described in, for example, W09425591 for antibodies derived from Camelidae heavy chain Ig, as well in US20030130496 describing the isolation of single domain fully human antibodies from phage libraries.
- Fv and sFv are the only species with intact combining sites that are devoid of constant regions. Thus, they are suitable for reduced nonspecific binding during in vivo use.
- sFv fusion proteins can be constructed to yield fusion of an effector protein at either the amino or the carboxy terminus of an sFv. See, for example, Antibody Engineering, ed. Borrebaeck, supra.
- the antibody fragment also can be a "linear antibody", for example, as described in U.S. Pat. No. 5,641,870 for example. Such linear antibody fragments can be monospecific or bispecific.
- antibodies of the described invention are bispecific or multi-specific.
- Bispecific antibodies are antibodies that have binding specificities for at least two different epitopes.
- Exemplary bispecific antibodies can bind to two different epitopes of a single antigen.
- Other such antibodies can combine a first antigen binding site with a binding site for a second antigen.
- an anti-HIV arm can be combined with an arm that binds to a triggering molecule on a leukocyte, such as a T-cell receptor molecule (for example, CD3), or Fc receptors for IgG (Fc gamma R), such as Fc gamma RI (CD64), Fc gamma RII (CD32) and Fc gamma RIII (CD 16), so as to focus and localize cellular defense mechanisms to the infected cell.
- Bispecific antibodies also can be used to localize cytotoxic agents to infected cells. Bispecific antibodies can be prepared as full length antibodies or antibody fragments (for example, F(ab')2 bispecific antibodies). See for example, WO 96/16673, U.S. Pat. No. 5,837,234, WO98/02463, U.S. Pat. No. 5,821,337, and Mouquet et al, Nature. 467, 591-5 (2010).
- bispecific antibodies are known in the art. Traditional production of full length bispecific antibodies is based on the co-expression of two immunoglobulin heavy chain-light chain pairs, where the two chains have different specificities (see, for example, Millstein et al, Nature, 305:537-539 (1983)). Similar procedures are disclosed in, for example, WO 93/08829, Traunecker et al, EMBO J., 10:3655-3659 (1991) and see also; Mouquet et al, Nature. 467, 591-5 (2010). Techniques for generating bispecific antibodies from antibody fragments also have been described in the literature. For example, bispecific antibodies can be prepared using chemical linkage. See Brennan et al, Science, 229: 81 (1985).
- the antibodies of described in the invention can be produced using conventional hybridoma technology or made recombinantly using vectors and methods available in the art.
- Human antibodies also can be generated by in vitro activated B cells (see, for example, U.S. Pat. Nos. 5,567,610 and 5,229,275).
- General methods in molecular genetics and genetic engineering useful in the present invention are described in the current editions of Molecular Cloning: A Laboratory Manual (Sambrook, et al., Molecular Cloning: A Laboratory Manual (Fourth Edition) Cold Spring Harbor Lab. press, 2012), Gene Expression Technology (Methods in Enzymology, Vol. 185, edited by D. Goeddel, 1991.
- Human antibodies also can be produced in transgenic animals (for example, mice) that are capable of producing a full repertoire of human antibodies in the absence of endogenous immunoglobulin production.
- transgenic animals for example, mice
- JH antibody heavy-chain joining region
- F(ab')2 fragments can be isolated directly from recombinant host cell culture.
- Fab and F(ab')2 fragment with increased in vivo half-life comprising a salvage receptor binding epitope residues are described in U.S. Pat. No. 5,869,046. Other techniques for the production of antibody fragments will be apparent to the skilled practitioner.
- Phage display technology is known in the art (e.g., see technology from Cambridge Antibody Technology (CAT)) as disclosed in U.S. Patent Nos. 5,565,332; 5,733,743; 5,871,907; 5,872,215; 5,885,793; 5,962,255; 6,140,471; 6,225,447; 6,291650; 6,492,160; 6,521,404; 6,544,731; 6,555,313; 6,582,915; 6,593, 081, as well as other U.S. family members, or applications which rely on priority filing GB 9206318, filed 24 May 1992; see also Vaughn, et al. 1996, Nature Biotechnology 14: 309- 314).
- Single chain antibodies may also be designed and constructed using available recombinant DNA technology, such as a DNA amplification method (e.g., PCR), or possibly by using a respective hybridoma cDNA as a template.
- variants of the sequences recited in the application also are included within the scope of the invention.
- Further variants of the antibody sequences having improved affinity can be obtained using methods known in the art and are included within the scope of the invention.
- amino acid substitutions can be used to obtain antibodies with further improved affinity.
- codon optimization of the nucleotide sequence can be used to improve the efficiency of translation in expression systems for the production of the antibody.
- an antibody of the invention comprises a heavy chain variable region comprising CDRl, CDR2 and CDR3 sequences and a light chain variable region comprising CDRl, CDR2 and CDR3 sequences, wherein one or more of these CDR sequences comprise specified amino acid sequences based on the preferred antibodies described herein, or conservative modifications thereof, and wherein the antibodies retain the desired functional properties of neutralizing multiple HIV-1 viral strains.
- conservative sequence modifications refers to amino acid modifications that do not significantly affect or alter the binding characteristics of the antibody containing the amino acid sequence. Such conservative modifications include amino acid substitutions, additions and deletions.
- Modifications can be introduced into an antibody of the invention by standard techniques known in the art, such as site-directed mutagenesis and PCR-mediated mutagenesis.
- Conservative amino acid substitutions are ones in which the amino acid residue is replaced with an amino acid residue having a similar side chain. Families of amino acid residues having similar side chains have been defined in the art. These families include
- amino acids with basic side chains e.g., lysine, arginine, histidine
- acidic side chains e.g., aspartic acid, glutamic acid
- uncharged polar side chains e.g., glycine, asparagine, glutamine, serine, threonine, tyrosine, cysteine, tryptophan
- nonpolar side chains e.g., alanine, valine, leucine, isoleucine, proline, phenylalanine, methionine
- beta-branched side chains e.g., threonine, valine, isoleucine
- aromatic side chains e.g., tyrosine, phenylalanine, tryptophan, histidine.
- amino acid residues within the CDR regions of an antibody of the invention can be replaced with other amino acid residues from the same side chain family and the altered antibody can be tested for retained function using the functional assays described herein.
- the antibody can be linked to one of a variety of nonproteinaceous polymers, for example, polyethylene glycol, polypropylene glycol, polyoxyalkylenes, or copolymers of polyethylene glycol and polypropylene glycol.
- the antibody also can be entrapped in microcapsules prepared, for example, by coacervation techniques or by interfacial polymerization (for example, hydroxymethylcellulose or gelatin-microcapsules and poly- (methylmethacylate) microcapsules, respectively), in colloidal drug delivery systems (for example, liposomes, albumin microspheres, microemulsions, nano-particles and nanocapsules), or in macroemulsions.
- colloidal drug delivery systems for example, liposomes, albumin microspheres, microemulsions, nano-particles and nanocapsules
- macroemulsions for example, Remington's Pharmaceutical Sciences, 16th edition, Oslo, A., Ed., (1980).
- bNAbs are significantly more effective than ART in blocking the establishment of the reservoir when given early in the infection.
- One of the key differences between antibodies and ART is that antibodies can engage a variety of host immune effector pathways by way of their Fc receptors (Nimmerjahn et al., Nat Rev Immunol, 2008. 8(1): p. 34-47). Consistent with this important difference, the mechanism by which antibodies interfere with the establishment of the reservoir is dependent on their ability to bind to Fc receptors.
- Fc receptor or “FcR” are used to describe a receptor that binds to the Fc region of an antibody.
- An Fc receptor is a protein found on the surface of certain cells - including, among others, B lymphocytes, follicular dendritic cells, natural killer cells, macrophages, neutrophils, eosinophils, basophils and mast cells - that contribute to the protective functions of the immune system. Its name is derived from its binding specificity for the Fc region (fragment crystallizable region) of an antibody.
- Fc receptors bind to antibodies that are attached to infected cells or invading pathogens. Their activity stimulates phagocytic or cytotoxic cells to destroy microbes, or infected cells by antibody-mediated phagocytosis or antibody-dependent cell-mediated cytotoxicity. It was also known in the art that the Fc region of an antibody ensures that each antibody generates an appropriate immune response for a given antigen, by binding to a specific class of Fc receptors, and other immune molecules, such as complement proteins.
- FcRs are defined by their specificity for immunoglobulin isotypes: Fc receptors for IgG antibodies are referred to as FcyR, for IgE as FcsFR, for IgA as FcaR and so on.
- FcyR Fc receptors for IgG antibodies
- FcsFR FcsFR
- IgA FcaR
- Surface receptors for immunoglobulin G are present in two distinct classes-those that activate cells upon their crosslinking (“activation FcRs") and those that inhibit activation upon co-engagement (“inhibitory FcRs").
- FcyRI CD64
- FcyRII CD32
- FcyRIII CDI6
- FcylV FcylV
- FcyRII displays high affinity for the antibody constant region and restricted isotype specificity
- FcyRII and FcyRIII have low affinity for the Fc region of IgG but a broader isotype binding pattern (Ravetch and Kinet, 1991; Hulett and Hogarth, Adv Immunol 57, 1-127 (1994)).
- FcyRIV is a recently identified receptor, conserved in all mammalian species with intermediate affinity and restricted subclass specificity (Mechetina et al., Immunogenetics 54, 463-468 (2002); Davis et al, Immunol Rev 190, 123-136 (2002); Nimmerjahn et al, Immunity 23, 41-51 (2005)). Functionally there are two different classes of Fc-receptors: the activation and the inhibitory receptors, which transmit their signals via immunoreceptor tyrosine based activation (ITAM) or inhibitory motifs (ITIM), respectively (Ravetch, in Fundamental Immunology W. E. Paul, Ed.
- ITAM immunoreceptor tyrosine based activation
- ITIM inhibitory motifs
- FcR is a native sequence human FcR.
- FcR including human FcR, binds an IgG antibody (a gamma receptor) and includes receptors of the FcyRI, FcyRII, and FcyRIII subclasses, including allelic variants and alternatively spliced forms of these receptors.
- FcyRII receptors include FcyRIIA (an “activating receptor") and FcyRIIB (an "inhibiting receptor”), which have similar amino acid sequences that differ primarily in the cytoplasmic domains thereof.
- Activating receptor FcyRIIA contains an immunoreceptor tyrosine-based activation motif (ITAM) in its cytoplasmic domain.
- Inhibiting receptor FcyRIIB contains an immunoreceptor tyrosine-based inhibition motif (ITIM) in its cytoplasmic domain (see review in Daron, Annu Rev Immunol, 15, 203-234 (1997); FcRs are reviewed in Ravetch and Kinet, Annu Rev Immunol, 9, 457-92 (1991); Capel et al, Immunomethods, 4, 25-34 (1994); and de Haas et al, J Lab Clin Med, 126, 330-41 (1995), Nimmerjahn and Ravetch 2006, Ravetch Fc Receptors in Fundemental Immunology, ed William Paul 5th Ed. each of which is incorporated herein by reference).
- ITAM immunoreceptor tyrosine-based activation motif
- ITIM immunoreceptor tyrosine-based inhibition motif
- Fc fragment or "Fc region” is used to define a C- terminal region of an immunoglobulin heavy chain.
- Fc region is the tail region of an antibody that interacts with Fc receptors and some proteins of the complement system.
- the Fc region may be a native sequence Fc region or a variant Fc region.
- the human IgG heavy chain Fc region is usually defined to stretch from an amino acid residue at position Cys226, or from Pro230, to the carboxyl-terminus thereof.
- a native sequence Fc region comprises an amino acid sequence identical to the amino acid sequence of an Fc region found in nature.
- a variant Fc region as appreciated by one of ordinary skill in the art comprises an amino acid sequence which differs from that of a native sequence Fc region by virtue of at least one "amino acid modification.”
- the Fc region is composed of two identical protein fragments, derived from the second and third constant domains of the antibody's two heavy chains; IgM and IgE Fc regions contain three heavy chain constant domains (C H domains 2-4) in each polypeptide chain.
- the Fc regions of IgGs bear a highly conserved N-glycosylation site. Glycosylation of the Fc fragment is important for Fc receptor-mediated activity.
- the N-glycans attached to this site are predominantly core- fucosylated diantennary structures of the complex type. In addition, small amounts of these N-glycans also bear bisecting GlcNAc and a-2,6 linked sialic acid residues.
- the bNAb antibody of this invention can include antibody variable regions with the desired binding specificities (antibody-antigen combining sites) fused to immunoglobulin constant domain sequences.
- the fusion can be with an Ig heavy chain constant domain, comprising at least part of the hinge, CH2, and CH3 regions.
- the first heavy-chain constant region (CHI) containing the site necessary for light chain bonding is present in at least one of the fusions.
- DNAs encoding the immunoglobulin heavy chain fusions and, if desired, the immunoglobulin light chain are inserted into separate expression vectors, and are co-transfected into a suitable host cell.
- a transcription inducer refers to any agent that can induce the transcriptional activation of the HIV-1 promoter, or re-activate latent HIV-1 from the patient viral reservoir directly or indirectly ⁇ e.g., facilitating T cell activation by removing an inhibitory pathway).
- suitable transcription inducers include vorinostat, an HDAC inhibitor (Contreras et al, J Biol Chem, 2009. 284(11): p. 6782-9; Archin et al, AIDS Res Hum Retroviruses, 2009. 25(2): p. 207-12; and Archin et al, AIDS, 2009. 23(14): p.
- HDAC inhibitors in combination with the bNAbs of this invention can be used in disrupting, decreasing, or purging the latently infected reservoirs in patients, particularly patients undergoing Highly Active Antiretro viral Therapy (HAART).
- the above-described antibodies and viral transcription inducers can used in combination with one or more anti-retroviral agents for the treatment of HIV latency and/or infection. See, e.g., US 2010/0166806, US 2010/0324034, and US 2012/0203014, which are hereby incorporated in their entirety.
- compositions according to the present invention may also be administered in combination with other agents to enhance the biological activity of such agents.
- agents may include any one or more of the standard anti-HIV agents which are known in the art, including, but not limited to, azidothymidine (AZT), dideoxycytidine (ddC), and dideoxyinosine (ddl).
- AZT azidothymidine
- ddC dideoxycytidine
- ddl dideoxyinosine
- compositions in accordance to the invention include, for example, raltegravir, maraviroc, bestatin, human chorionic gonadotropin (hCG), levamisole, estrogen, efavirenz, etravirine, indomethacin, emtricitabine, tenofovir disoproxil fumarate, amprenavir, tipranavir, indinavir, ritonavir, darunavir, enfuvirtide, and gramicidin.
- raltegravir maraviroc
- bestatin human chorionic gonadotropin (hCG)
- levamisole estrogen
- efavirenz etravirine
- indomethacin emtricitabine
- tenofovir disoproxil fumarate amprenavir, tipranavir, indinavir, ritonavir, darunavir, enfuvirtide, and gramicidin.
- the present invention provides a composition comprising at least one bNAb mentioned above alone or in combination with one of the other active agent mentioned above and a pharmaceutically acceptable carrier.
- the composition may include a plurality of the antibodies having the characteristics described herein in any combination and can further include antibodies neutralizing to HIV as are known in the art.
- compositions can be a single or a combination of antibodies disclosed herein, which can be the same or different, in order to prophylactically or therapeutically treat the progression of various subtypes of HIV infection.
- an antibody or active agent When an antibody or active agent is administered to an animal or a human, it can be combined with one or more pharmaceutically acceptable carriers, excipients or adjuvants as are known to one of ordinary skilled in the art.
- mice for example, SCID, bg/nu/xid, NOD/SCID, SCID-hu, immunocompetent SCID-hu, bone marrow-ablated BALB/c
- PBMCs peripheral blood mononuclear cells
- the simian immune deficiency virus (SrV)/monkey model can be employed, as can the feline immune deficiency virus (FIV)/cat model.
- the pharmaceutical composition can contain other pharmaceuticals, in conjunction with a vector according to the invention, when used to therapeutically treat AIDS. These other pharmaceuticals can be used in their traditional fashion (i.e., as agents to treat HIV infection).
- the present invention provides an antibody- based pharmaceutical composition comprising an effective amount of an isolated bNAb of the invention, or an affinity matured version, which provides a prophylactic or therapeutic treatment choice to reduce the latent reservoir and infection of the HIV virus.
- the pharmaceutical compositions of the present invention may be formulated by any number of strategies known in the art (e.g., see McGoff and Scher, 2000, Solution Formulation of Proteins/Peptides: In McNally, E.J., ed. Protein Formulation and Delivery. New York, NY: Marcel Dekker; pp. 139-158; Akers and Defilippis, 2000, Peptides and Proteins as Parenteral Solutions. In: Pharmaceutical Formulation Development of Peptides and Proteins.
- a pharmaceutically acceptable composition suitable for patient administration will contain an effective amount of the bNAb antibody in a formulation which both retains biological activity while also promoting maximal stability during storage within an acceptable temperature range.
- the pharmaceutical compositions can also include, depending on the formulation desired, pharmaceutically acceptable diluents, pharmaceutically acceptable carriers and/or pharmaceutically acceptable excipients, or any such vehicle commonly used to formulate pharmaceutical compositions for animal or human administration. The diluent is selected so as not to affect the biological activity of the combination.
- an excipient that is useful in the pharmaceutical composition or formulation of this invention is an amount that serves to uniformly distribute the antibody throughout the composition so that it can be uniformly dispersed when it is to be delivered to a subject in need thereof. It may serve to dilute the antibody or other active agent to a concentration which provides the desired beneficial palliative or curative results while at the same time minimizing any adverse side effects that might occur from too high a concentration. It may also have a preservative effect. Thus, for an active ingredient having a high physiological activity, more of the excipient will be employed. On the other hand, for any active ingredient(s) that exhibit a lower physiological activity, a lesser quantity of the excipient will be employed.
- bNAb antibodies and antibody compositions comprising at least one or a combination of the antibodies described herein, can be administered for the prophylactic and therapeutic treatment of HIV viral infection.
- composition can be a pharmaceutical composition that contains a pharmaceutically acceptable carrier.
- pharmaceutical composition refers to the combination of an active agent with a carrier, inert or active, making the composition especially suitable for diagnostic or therapeutic use in vivo or ex vivo.
- carrier as used herein includes pharmaceutically acceptable carriers, excipients, or stabilizers that are nontoxic to the cell or mammal being exposed thereto at the dosages and concentrations employed. Often the physiologically acceptable carrier is an aqueous pH buffered solution.
- physiologically acceptable carriers include, but not limited to, buffers such as phosphate, citrate, and other organic acids; antioxidants including, but not limited to, ascorbic acid; low molecular weight (less than about 10 residues) polypeptide; proteins, such as, but not limited to, serum albumin, gelatin, or immunoglobulins; hydrophilic polymers such as, but not limited to, polyvinylpyrrolidone; amino acids such as, but not limited to, glycine, glutamine, asparagine, arginine or lysine; monosaccharides, disaccharides, and other carbohydrates including, but not limited to, glucose, mannose, or dextrins; chelating agents such as, but not limited to, EDTA; sugar alcohols such as, but not limited to, mannitol or sorbitol; salt-forming counterions such as, but not limited to, sodium; and/or nonionic surfactants such as, but not limited to, TWEEN.; polyethylene glycol (P
- pharmaceutically acceptable carrier refers to any of the standard pharmaceutical carriers, such as a phosphate buffered saline solution, water, emulsions, and various types of wetting agents.
- the compositions also can include stabilizers and preservatives.
- a pharmaceutically acceptable carrier after administered to or upon a subject, does not cause undesirable physiological effects.
- the carrier in the pharmaceutical composition must be “acceptable” also in the sense that it is compatible with the active ingredient and, preferably, capable of stabilizing it.
- One or more solubilizing agents can be utilized as pharmaceutical carriers for delivery of an active agent. Examples of other carriers include colloidal silicon oxide, magnesium stearate, cellulose, and sodium lauryl sulfate.
- a “subject” refers to a human and a non-human animal.
- a non-human animal include all vertebrates, e.g., mammals, such as non-human primates (particularly higher primates), dog, rodent (e.g., mouse or rat), guinea pig, cat, and non-mammals, such as birds, amphibians, reptiles, etc.
- the subject is a human.
- the subject is an experimental animal or animal suitable as a disease model (such as non-human primates).
- a subject to be treated can be identified by standard diagnosing techniques for the disorder.
- the present invention provides a method of reducing or preventing the establishment of a latent reservoir of HIV infected cells in a subject in need thereof (e.g., a subject infected with HIV or at risk of infection with HIV), thereby treating infection with a HIV infection, comprising administering to the subject a pharmaceutical composition comprising the HIV antibodies disclosed herein.
- the compositions of the invention can include more than one antibody having the characteristics disclosed (for example, a plurality or pool of antibodies). It also can include other HIV neutralizing antibodies and/or active agent known in the art.
- Subjects at risk for HIV-related diseases or disorders include patients who have come into contact with an infected person or who have been exposed to HIV in some other way. Administration of a prophylactic agent can occur prior to the manifestation of symptoms characteristic of HIV-related disease or disorder, such that a disease or disorder is prevented or, alternatively, delayed in its progression.
- the patient is administered or provided a pharmaceutical formulation including an HIV antibody of the invention.
- the antibodies of the invention are administered to the patient in therapeutically effective amounts (i.e., amounts that eliminate or reduce the patient's latent viral reservoir).
- the antibodies are administered to a human patient, in accord with known methods, such as intravenous administration, for example, as a bolus or by continuous infusion over a period of time, by intramuscular, intraperitoneal, intracerobrospinal, subcutaneous, intra-articular, intrasynovial, intrathecal, oral, topical, or inhalation routes.
- the antibodies can be administered parenterally, when possible, at the target cell site, or intravenously.
- antibody is administered by intravenous or subcutaneous administration.
- Therapeutic compositions of the invention may be administered to a patient or subject systemically, parenterally, or locally. The above parameters for assessing successful treatment and improvement in the disease are readily measurable by routine procedures familiar to a physician.
- the antibodies may be formulated in a unit dosage injectable form (solution, suspension, emulsion) in association with a pharmaceutically acceptable, parenteral vehicle.
- a pharmaceutically acceptable, parenteral vehicle examples include, but are not limited, water, saline, Ringer's solution, dextrose solution, and 5% human serum albumin.
- Nonaqueous vehicles include, but are not limited to, fixed oils and ethyl oleate. Liposomes can be used as carriers.
- the vehicle may contain minor amounts of additives such as substances that enhance isotonicity and chemical stability, such as, for example, buffers and preservatives.
- the antibodies can be formulated in such vehicles at concentrations of about 1 mg/ml to 10 mg/ml.
- the dose and dosage regimen depends upon a variety of factors readily determined by a physician, such as the nature of the infection, for example, its therapeutic index, the patient, and the patient's history.
- a therapeutically effective amount of an antibody is administered to a patient.
- the amount of antibody administered is in the range of about 0.1 mg/kg to about 50 mg/kg of patient body weight.
- about 0.1 mg/kg to about 50 mg/kg body weight (for example, about 0.1-15 mg/kg/dose) of antibody is an initial candidate dosage for administration to the patient, whether, for example, by one or more separate administrations, or by continuous infusion.
- the progress of this therapy is readily monitored by conventional methods and assays and based on criteria known to the physician or other persons of skill in the art.
- the above parameters for assessing successful treatment and improvement in the disease are readily measurable by routine procedures familiar to a physician.
- Other therapeutic regimens may be combined with the administration of the bNAb HIV antibody of the present invention.
- the combined administration includes co- administration, using separate formulations or a single pharmaceutical formulation, and consecutive administration in either order, wherein preferably there is a time period while both (or all) active agents simultaneously exert their biological activities.
- Such combined therapy can result in a synergistic therapeutic effect.
- the parameters for assessing successful treatment and improvement in the disease are also readily measurable by routine procedures familiar to a physician.
- treating or “treatment” or “alleviation” are used interchangeably and refer to both therapeutic treatment and prophylactic or preventative measures; wherein the object is to prevent or slow down (lessen) the targeted pathologic condition or disorder.
- it refers to administration of a compound or agent to a subject, who has a disorder (such as an HIV infection), with the purpose to cure, alleviate, relieve, remedy, delay the onset of, prevent, or ameliorate the disorder, the symptom of the disorder, the disease state secondary to the disorder, or the predisposition toward the disorder.
- a disorder such as an HIV infection
- Those in need of treatment include those already with the disorder as well as those prone to have the disorder or those in whom the disorder is to be prevented.
- a subject or mammal is successfully "treated" for an infection if, after receiving a therapeutic amount of an antibody according to the methods of the present invention, the patient shows observable and/or measurable reduction in or absence of one or more of the following: reduction in the number of infected cells or absence of the infected cells; reduction in the percent of total cells that are infected; and/or relief to some extent, one or more of the symptoms associated with the specific infection; reduced morbidity and mortality, and improvement in quality of life issues.
- the above parameters for assessing successful treatment and improvement in the disease are readily measurable by routine procedures familiar to a physician.
- Eliminating the HIV-1 reservoir in chronic infection is key to curing the disease, but direct measurement of the latent reservoir to evaluate therapeutic eradication strategies remains difficult (Siliciano et al, Curr Opin HIV AIDS, 2013. 8(4): p. 318-25).
- Quantitative viral outgrowth assays and PCR-based assays of integrated DNA yield variable results (Eriksson et al., PLoS Pathog, 2013. 9(2): p. el003174) in part because PCR cannot distinguish between inactive and permanently disabled proviruses, and outgrowth assays underestimate reservoir size (Ho et al., Cell, 2013. 155(3): p. 540-51).
- the most effective way to evaluate the reservoir in vivo is to measure viral rebound after terminating therapy as disclosed in the examples below.
- the terms "prevent,” “preventing,” “prevention,” “prophylactic treatment” and the like refer to reducing the probability of developing a disorder or condition in a subject, who does not have, but is at risk of or susceptible to developing a disorder or condition.
- a “therapeutically effective amount” refers to the amount of an agent sufficient to effect beneficial or desired results.
- a therapeutically effective amount can be administered in one or more administrations, applications or dosages and is not intended to be limited to a particular formulation or administration route.
- Administration "in combination with” one or more further therapeutic agents includes simultaneous (concurrent) and consecutive administration in any order.
- compositions of this invention may be administered to humans and other animals by a variety of methods that may include continuous or intermittent administration. Examples of methods of administration may include, but are not limited to, oral, rectal, parenteral, intracisternal, intrasternal, intravaginal, intraperitoneal, topical, transdermal, buccal, or as an oral or nasal spray. Accordingly, the pharmaceutically effective compositions may also include pharmaceutically acceptable additives, carriers or excipients. Such pharmaceutical compositions may also include the active ingredients formulated together with one or more non-toxic, pharmaceutically acceptable carriers specially formulated for oral administration in solid or liquid form, for parenteral injection or for rectal administration according to standard methods known in the art.
- parenteral administration refers to modes of administration which include intravenous, intramuscular, intraperitoneal, intracisternal, intrasternal, subcutaneous and intraarticular injection and infusion.
- injectable mixtures are known in the art and comprise pharmaceutically acceptable sterile aqueous or nonaqueous solutions, dispersions, suspensions or emulsions as well as sterile powders for reconstitution into sterile injectable solutions or dispersions just prior to use.
- aqueous and nonaqueous carriers, diluents, solvents or vehicles examples include water, ethanol, polyols (such as glycerol, propylene glycol, polyethylene glycol and the like), vegetable oils (such as olive oil), injectable organic esters (such as ethyl oleate) and suitable mixtures thereof.
- polyols such as glycerol, propylene glycol, polyethylene glycol and the like
- vegetable oils such as olive oil
- injectable organic esters such as ethyl oleate
- kits In general, kits according to the present invention comprise an isolated anti-HIV bNAb antibody, a first viral transcription inducer, and a second viral transcription inducer. Components of the kits can be provided in containers.
- the containers are provided in packaged combination in a suitable package, such as a box made of cardboard, glass, plastic, metal, or a combination thereof.
- suitable packaging materials for pharmaceutical compositions or reagents are known and widely used in the art, and thus need not be specified herein.
- kits of the invention can comprise any number of additional reagents or substances that are useful for practicing a method of the invention. Such substances include, but are not limited to: reagents (including buffers) for detecting HIV virus or components thereof or HIV-infected cells in a subject.
- reagents including buffers
- the kits of the invention can be provided at any temperature. For example, for storage of kits containing protein-based agents, it is preferred that they are provided and maintained below 0 °C, preferably at or below -20 °C, or otherwise in a frozen state.
- the kit may further comprise a software package for data analysis of the physiological status of a subject to be treated, which may include reference profiles for comparison with the relevant test profile.
- kits may also include information, such as scientific literature references, package insert materials, clinical trial results, and/or summaries of these and the like, which indicate or establish the activities and/or advantages of the composition, and/or which describe dosing, administration, side effects, drug interactions, or other information useful to the health care provider. Such information may be based on the results of various studies, for example, studies using experimental animals involving in vivo models and studies based on human clinical trials. Kits described herein can be provided, marketed and/or promoted to health providers, including physicians, nurses, pharmacists, formulary officials, and the like. Kits may also, in some embodiments, be marketed directly to the consumer.
- NOD Ragl ⁇ rg ⁇ (NOD.Cg-Ragl tmlMom I12rg tmlwjl /SzJ, NRG) mice were purchased from The Jackson Laboratory. All mice were bred and maintained at the Comparative Bioscience Center of The Rockefeller University according to guidelines established by the Institutional Animal Committee. All experiments were performed with authorization from the Institutional Review Board and the IACUC at The Rockefeller University.
- mice were generated as previously described in Klein et ah, Nature, 2012. 492(7427): p. 118-22. Briefly, human fetal livers were obtained from Advanced Bioscience Resources (ABR). Fetal livers were homogenized and incubated in HBSS media with 0.1% collagenase IV (Sigma- Aldrich), 40 mM HEPES, 2 mM CaCk and 2 U ml "1 DNAase I (Roche) for 30 minutes at 37° C. Hematopoietic Stem Cells (HSCs) were isolated from digested liver using CD34 + HSC isolation kit (Stem Cell Technologies). Neonatal NRG mice (1-5 days old) were sublethally irradiated with 100 cG and injected intrahepatically with 2 X 10 5 human CD34 + HSCs 6 h after irradiation. Mouse Screening For Humanization
- mice were screened for the presence of human lymphocytes in peripheral blood by flow cytometry. 200 ⁇ whole blood was collected by facial vein bleed and peripheral blood mononuclear cells (PBMCs) were isolated by density gradient centrifugation using Ficoll-Paque Plus (GE Healthcare Life Sciences). PBMCs were stained with antibodies to mouse CD45-PECy7, human CD45- Pacific Orange, human CD3- Pacific Blue, human CD19-APC, human CD4-PE, human CD8-FITC, and human CD16-Alexa700 for 25 min at 4 °C. Cells were washed and fixed using Cytofix/Cytoperm (BD Biosciences).
- Flow cytometry analysis was performed with a LSRFortessa (BD) and Flow Jo software (Tree Star). For each mouse, the percentage of human lymphocytes [(100 x human CD45 ) / (human CD45 + mouse CD45 + )], termed huCD45 + %, and the percentage of human CD4 + T cells (100 x human CD45 + CD3 + CD4 + / human CD45 + ), termed huCD4 + %, was calculated. Mice with at least 10% huCD45 + and 10% huCD4 + were selected for post-exposure prophylaxis experiments, and infected with two doses of HIV-1 Y U2 (150 ng p24) by i.p.
- mice with measurable human CD4 + cells by FACS were injected with two doses of HIV-1 Y U2 (150 ng), and pre-treatment viremia was measured 14-18 days after the first injection.
- Mice with plasma viral loads >3000 RNA copies/ml were selected to receive antibody therapy. After five subcutaneous antibody injections (see below), post-treatment viremias were measured. Only mice with completely suppressed plasma viremias were selected for further analysis and to receive viral inducers.
- the reaction mixture was prepared using TaqMan RNA-to-Ct 1-Step kit (Applied Biosystems), with 20 ⁇ of eluted RNA, and a sequence specific probe targeting a conserved region of the HIV-1 pol gene (/HEX/5 '-CCCACCAACARGCRGCCT TAACTG-3 VZenDQ, HXB2 nt 4603 to 4626, SEQ ID No: 44) (Integrated DNA Technologies).
- GGTGTGTAGTTCTGCCAATCAGGGAAGWAGCCTTGTG-3' (SEQ ID Nos: 47 and 48).
- Primers for the second round of PCR were 5'- T AGAAAG AGC AGAAGAC AGTGGC AATGA-3 ' and 5 ' - TC ATC AATGGTGGTGATGATGATGTTTTTCTCTCTGC ACC ACTCTTCT-3 ' (SEQ ID Nos: 47 and 48).
- Primers for the second round of PCR were 5'- T AGAAAG AGC AGAAGAC AGTGGC AATGA-3 ' and 5 ' - TC ATC AATGGTGGTGATGATGATGTTTTTCTCTCTGC ACC ACTCTTCT-3 ' (SEQ ID Nos: 47 and 48).
- Gel-purified PCR amplicons were ligated into pCR4-TOPO (Invitrogen) and transformed into One Shot TOP 10 cells. Individual colonies were sequenced using M13F and M13R primers. Sequences were aligned to gpl20yu 2 (accession number M93258) and analyzed for mutations using Los Alamos Highlighter tool (hiv.lanl.gov/content/sequence/HIGHLIGHT/ HIGHLIGHT XYPLOT/highlighter.html).
- the cellular fraction of whole blood was resuspended in 400 ⁇ PBS and PBMCs were isolated by density gradient centrifugation as described above. Lymphocytes were split into two samples, one for cell-associated HIV-1 RNA measurements, and one for cell-associated HIV-1 DNA measurements. Cell-associated RNA was extracted and quantified by the same procedures as described above for plasma viral RNA. The lower limit of detection was determined to be 10 copies viral RNA per qRT-PCR reaction. Cell- associated HIV-1 RNA is reported as the ratio of HIV- 1 RNA copies per sample to CCR5 genomic DNA copies per equivalent sample measured in DNA extract. For terminal point measurements, spleen tissue was isolated, homogenized, and filtered through 40 ⁇ mesh. Splenocytes were used to isolate HIV-1 RNA as described above.
- PBMCs peripheral blood cells were isolated from whole blood as described above. Splenocytes were isolated from spleen as described above. Total DNA was extracted using QIAmp DNA Blood Mini Kit (Qiagen) and eluted in 80 ⁇ volume. Purified DNA was quantified for HIV-1 DNA by qPCR using the primers and probe for HIV-1 RNA quantification mentioned above.
- Genomic human CCR5 DNA was quantified with primers 5'- GTTGGACC AAGCTATGCAGGT-3 ' (forward, SEQ ID No: 51) and 5'- AGAAGCGTTTGGCAATGTGC-3 ' (reverse, SEQ ID No: 52), and the sequence-specific probe /HEX/5 '-TTGGGATGACGCACTGCTGCATCAACCCCA-3 VZenDQ (SEQ ID No: 53). All qPCR reactions contained 25 ⁇ AmpliTaq Gold PCR master mix (Applied Biosystems), in 50 ⁇ reaction volume. Reaction mixtures were as previously described (Horwitz et al, Proc Natl Acad Sci U S A, 2013. 110(41): p. 16538-43). HIV-1 DNA is reported as copies per sample to CCR5 genomic copies per equivalent sample.
- lymphocytes at the terminal point were quantified from the spleen and PBMCs by flow cytometry. Isolation of PBMCs and splenocytes were as described above. Staining procedures were as described above.
- Plasma levels of passively administered antibodies were quantified by two independent methods.
- gpl20-specific ELISA was as previously described (Klein et al, Nature, 2012. 492(7427): p. 118-22), using 10-1074 and 3BNC117 monoclonal antibodies as standard controls. The detection limit was 0.05 ⁇ g/ml. Because PG16 does not bind gpl20, and endogenously produced gpl20-reactive antibodies could confound the ELISA measurement, plasma antibody levels were also quantified by TZM-bl neutralization using the Tier 2 envelopes 3301.vl .c24 and YU2. A mixture with known amounts of 3BNC117, 10-1074, and PG16 was used as standard for calibration.
- Plasma viremias immediately preceding and following viral rebound were plotted on a semi-log-y-axis versus days post initial antibody injection (x-axis) for each individual mouse.
- the linear portion of viremia was fit to a line by least-squares linear regression.
- the day that viremia crossed the 800 copies/ml quantitation limit, termed rebound day, was calculated from the viremia fit.
- the antibody concentrations (as determined by TZM- bl neutralization) spanning before and after viral rebound were plotted on a semi-log-y- axis versus days post initial antibody injection.
- the linear portion of antibody concentrations was fit to a line by least-squares linear regression, and the antibody concentration on the rebound day was calculated from the fit.
- mice were weighed daily on normal diet, then switched to ART feed and weighed daily. There were no visible signs of toxicity and mice maintained their weights. Assuming mice weigh 25 grams and eat 4 grams of food per day, the drug doses correspond to 2.88 mg/kg TFV, 83 mg/kg FTC, and 768 mg/kg RAL daily.
- Plasmids encoding 10-1074 or PG16 heavy- and light- chain Ig genes were transfected into HEK 293E cells. Antibodies were isolated from tissue-culture supernatant using Protein G Sepharose 4 Fast-Flow (GE Healthcare). Antibodies were then buffer- exchanged into PBS and sterile-filtered using Ultrafree-CL centrifugal filters (0.22 ⁇ ; Millipore). Endotoxin was removed from antibody preparations using Triton X-114 (Sigma- Aldrich) as previously described (Aida et al., J Immunol Methods, 1990. 132(2): p. 191-5), and antibodies were concentrated to 10 mg/ml. Sterile, endotoxin- free 3BNC117 (20 mg/ml) was obtained from CellDex Therapeutics. All antibodies were injected subcutaneously as described.
- Vorinostat (Selleckchem) was suspended in sterile water or sterile water plus 0.5% methylcellulose, 0.1 % Tween (v/v) and administered by oral gavage at doses of 60 mg/kg (Krejsgaard et al., 2010 Experimental dermatology 19, 1096-1102). For each mouse, three total doses were administered, spaced 48 hours apart. 100 ⁇ g doses of aCTLA4 were injected intraperitoneally (i.p.). Three total doses were administered, spaced 48 hours apart.
- I-BET was obtained from Glaxo SmithKline and dissolved in 10% beta-cyclodextrin, 5% DMSO in 0.9%> saline and injected daily for 14 days at doses of 30 mg/kg (Dawson et al, 2011 Nature 478, 529-533.).
- the ART -resistant reservoir is established early in infection as evidenced by postexposure prophylaxis experiments in humans and macaques.
- Post-exposure prophylaxis with ART or previous-generation bNAbs is only effective when administered within 24 hours of intravenous exposure.
- mice were infected with HIV-lyu 2 (150 ng) by intraperitoneal injection, and treated with either ART (raltegravir, emtricitabine, tenofovir) (Denton et al., J Virol, 2012.
- ART was administered in the food for 32-39 days starting 4 days after infection (Denton et al, J Virol, 2012. 86(1): p.
- mice in the early treatment group that failed to show detectable plasma viremia were further examined for the presence of human CD4 + T cells and cell-associated HIV-1 RNA and DNA in the spleen. It was found that mice that failed to develop sustained plasma viremia showed CD4 + T cell levels that were similar to infected controls. Therefore differences in CD4 + T cell levels are unlikely to account for the observed differences between viremic and aviremic mice (FIG. IF). Moreover, T cell-associated HIV-1 RNA levels were consistent with plasma viral loads, with mice that remained aviremic having either undetectable or lower cell-associated HIV-1 RNA than mice that developed sustained viremia (FIG. 1G).
- HIV-1 DNA was measured as an imperfect surrogate of the HIV-1 reservoir. HIV-1 DNA is thought to overestimate the reservoir because it fails to exclude damaged or incomplete viral sequences that cannot be reactivated. In addition, the overall number cells assayed in mice is limited and therefore the assay is not very sensitive. Nevertheless HIV-1 DNA measurements were found to be consistent with each mouse's rebound status (FIG. 1H). It was concluded that bNAbs can interfere with the establishment of the latent HIV-1 reservoir in hu-mice.
- bNAbs differ from ART in that they can prevent establishment of the latent HIV-1 reservoir in hu-mice at a time when ART is significantly less effective.
- FcR nu11 antibodies were far less potent than controls in vivo (FIGs. 2 and 7).
- Mice treated with FcR nu11 tri-mix initially suppressed viremia at the same rate as the wild type antibody-treated mice (FIG. 2A).
- 9 of 15 mice receiving post exposure prophylaxis with the FcR nu11 tri-mix showed viral rebound by 44 days after the last antibody injection.
- the loss of in vivo activity of the FcR nu11 tri-mix was not due to lower antibody levels in the FcR nu11 injected mice.
- Hu-mice with established HIV-lyu 2 infections (viremia ranging from 4.70x10 - 7.96xl0 5 copies/ml at 2-3 weeks after infection, FIGs. 3B-E) were treated with tri-mix bNAbs.
- plasma viremia dropped below detection, they were co-administered a viral inducer for 5-14 days, and monitored for viral rebound for an additional 47-85 days.
- the inducers tested were vorinostat, an HDAC inhibitor (Archin et al, AIDS Res Hum Retroviruses, 2009. 25(2): p. 207-12; Archin et al, AIDS, 2009. 23(14): p. 1799-806; and Contreras et al, J Biol Chem, 2009.
- vorinostat is the only one that has also been studied in HIV-1 infected humans.
- Treatment with vorinostat plus ART resulted in a transient increase in resting CD4 + T cell-associated HIV-1 RNA, but no change in plasma viremia, or the frequency of replication-competent HIV-1 within resting CD4 + T cells (Archin et al, Nature, 2012. 487(7408): p. 482-5 and Archin et al, J. Infect. Dis. 2014. p. 1-26).
- the above results in hu-mice are consistent with these findings, and extend them to additional candidate inducers, demonstrating that administration of a single inducer has no significant effect on the ability of the latent reservoir to produce rebound viremia.
- Example 5 Combination Therapy with Multiple Inducers
- assays were carried out to determine whether a combination of inducers might be more effective than a single inducer. More specifically, all three inducers mentioned above were administered simultaneously.
- antibody levels at the time of rebound and at the terminal point were calculated.
- the average plasma antibody concentration at the time of viral rebound in the 59 rebounding mice was 2.97 ⁇ g/ml (FIG. 5D). Since the antibody concentrations decayed to 2.97 ⁇ g/ml at different rates in individual mice, the number of days that elapsed from when each individual mouse's antibody levels reached 2.97 ⁇ g/ml to when the mouse showed rebound viremia were calculated. 50 of 59 mice rebounded within 10 days (FIG. 5E).
- the average antibody concentration at the terminal point was 0.44 ⁇ g/ml, with 15 out of 18 mice having antibody concentrations less than 2.97 ⁇ g/ml. Furthermore, in non-rebounding mice, an average of 20.2 days elapsed from the time antibody concentrations reached 2.97 ⁇ g/ml to termination (FIG. 5F). Thus, failure to rebound cannot be explained by antibody persistence or premature termination.
- hu-mice resembled infected humans in that they contain human cells that are infected with authentic HIV-1 (Hatziioannou et al, Nat Rev Microbiol, 2012. 10(12): p. 852-67 and Brehm et al, J Infect Dis, 2013. 208 Suppl 2: p. S 125-30).
- the kinetics of viral rebound in hu-mice after suppression of viremia with ART resembles infected humans (Horwitz et al, Proc Natl Acad Sci U S A, 2013. 110(41): p. 16538-43 and Nischang et al, PLoS ONE2012. p. e38853).
- the macaque model is valuable because it represents an immunologically intact host that may also harbor reservoirs not found in the mice.
- the infection in macaques involves non-human primate cells and SHIV or SIV, both of which differ significantly from HIV-1. While neither of the two model systems is entirely faithful, they have produced very similar results in both therapy and prevention experiments to date (West et al, Cell, 2014. 156(4): p. 633-48).
Abstract
The present invention relates to methods and agents for preventing the establishment of HIV-1 latent reservoirs or for reducing the size of the reservoirs. Specifically, the disclosure provides methods and agents for preventing the establishment of HIV-1 latent reservoirs or for reducing the size of the reservoirs, the methods comprising administering to the subject a therapeutically effective amount of an isolated anti-HIV antibody, and administering to the subject two or more viral transcription inducers in effective amounts to induce transcription of an HIV provirus in the cells. Further provided are antibodies and viral transcription inducers used in the methods.
Description
Combination of Broadly Neutralizing HIV Antibodies and Viral
Inducers
CROSS REFERENCE TO RELATED APPLICATION
This application claims priority to U.S. Provisional Application No. 62/026,915 filed on July 21, 2014. The content of the application is incorporated herein by reference in its entirety.
GOVERNMENT INTERESTS
The invention disclosed herein was made, at least in part, with Government support under Grant Nos. T32GM07739, AI100663-02, AI100148-02, AI081677-05, and #8 UL1 TR000043 from the National Institutes of Health (NIH). Accordingly, the U.S. Government has certain rights in this invention.
FIELD OF THE INVENTION
This invention relates to methods and agents for prevention or disruption of the establishment or maintenance of human immunodeficiency virus latent reservoirs. BACKGROUND OF THE INVENTION
Human immunodeficiency virus (HIV) is a retrovirus that causes acquired immunodeficiency syndrome (AIDS). HIV-1 is the most common and pathogenic strain of the virus. Although HIV-1 infection can be suppressed with any one of several different combination anti-retroviral therapies (ART), such a therapy must be maintained for the life of a patient because it does not eliminate a reservoir of latently infected cells even after years of ART (Siliciano, et al, Nat Med, 2003. 9(6): pp. 727-8.). As a result, ART termination follows by rapid viral rebound (Davey, et al., Proc. Natl. Acad. Sci. U.S.AA999. pp. 15109- 15114). To date, all attempts to alter the reservoir by intensifying ART, by including additional anti-retroviral drugs (Dinoso, et al, Proc Natl Acad Sci U S A, 2009. 106(23) pp. 9403-8 and Gandhi, et al, PLoS Med, 2010. 7(8)), or by administration of global T-cell activators or inducers of viral transcription, in the presence of ART, have failed (Archin, et al, J. Infect. Dis.2014. pp. 1-26; Dybul, et al, Infect Dis, 2002. 185(1): pp. 61-8; Lafeuillade, et al, J Acquir Immune Defic Syndr, 2001. 26(1): p. 44-55; and Prins, et al, AIDS, 1999. 13(17): p. 2405-10). Thus, there is a need for
methods and agents for prevention or disruption of the establishment or maintenance of HIV-1 latent reservoirs.
SUMMARY OF INVENTION
This invention relates to using broadly neutralizing antibodies (bNAbs) alone or in combination with viral transcription inducers in preventing the establishment of the latent reservoirs of HIV-1 infected cells or decreasing the size of the reservoir, and thereby addresses the need mentioned above.
Accordingly, one aspect of this invention provides a method for decreasing the size of or preventing the establishment of a latent reservoir of HIV infected cells (e.g., a cell population comprising HIV-infected CD4+ T cells.) in a subject in need thereof. The method includes administering to the subject a therapeutically effective amount of an isolated anti-HIV antibody, e.g., a bNAb, and administering to the subject two or more viral transcription inducers in effective amounts to induce transcription of an HIV provirus in the cells.
The antibody can be a human antibody, a humanized antibody, or a chimeric antibody. In one example, the antibody is antibody 3BNC1 17, 10-1074, or PG16, or any other described below, or combination thereof. Preferably, two or three of the antibodies 3BNC1 17, 10-1074, and PG16 are administered to the subject. The antibody or antibodies can be administered to the subject within about 96 hours or earlier (e.g., 72, 48, 36, 24, or 12 hours) after exposure or suspected exposure to HIV. The transcription inducers can be administered to the subject for a period of about 5-14 days (e.g., 6-13, 7-12, 8, 9, 10, 1 1 , 12 days). Examples of the transcription inducers include vorinostat, an HDAC inhibitor, I-BET151 , a BET bromodomain inhibitor, and aCTLA4, a T-cell inhibitory pathway blocker. The method can further comprise administering to the subject an antiviral agent, such as one selected from the group consisting of a non-nucleoside reverse transcriptase inhibitor, a protease inhibitor, an entry or fusion inhibitor, and an integrase inhibitor.
In a second aspect, the invention provides a kit comprising an isolated anti-HIV antibody, a first viral transcription inducer, and a second viral transcription inducer. The kit can contain one, two, three, or more of antibodies 3BNC1 17, 10-1074, PG16, and those described below. The antibody can be a human antibody, a humanized antibody, or a chimeric antibody. The transcription inducers can be selected from the group consisting
of vorinostat, an HDAC inhibitor, I-BET151 , a BET bromodomain inhibitor, and aCTLA4, a T-cell inhibitory pathway blocker. In the kit, the first viral transcription inducer and the second viral transcription inducer can be contained in one pharmaceutical composition or in two separate pharmaceutical compositions. The kit can further contain an antiviral agent, which can be selected from the group consisting of a non-nucleoside reverse transcriptase inhibitor, a protease inhibitor, an entry or fusion inhibitor, and an integrase inhibitor. The kit can be used in the method of this invention.
In a third aspect, the invention features a method for preventing the establishment of a latent reservoir of HIV infected cells in a subject in need thereof. The method includes administering to the subject a therapeutically effective amount of an isolated anti- HIV antibody. The antibody can be a human antibody, a humanized antibody, or a chimeric antibody. In one example, the antibody is antibody 3BNC1 17, 10-1074, PG16, any of those described below, or a combination thereof. Preferably, two, three, or more of the antibodies are administered to the subject. The antibody or antibodies can be administered to the subject within about 96 hours or earlier (e.g., 72, 48, 36, 24, or 12 hours) after exposure or suspected exposure to HIV.
The details of one or more embodiments of the invention are set forth in the description below. Other features, objectives, and advantages of the invention will be apparent from the description and from the claims. BRIEF DESCRIPTION OF THE DRAWINGS
FIG. 1A, FIG. IB, FIG. 1C, FIG. ID, FIG. IE, FIG. IF, AND FIG. 1H are a set of diagrams showing post-exposure prophylaxis with bNAbs: (FIG. 1A) Schematic timeline for the bNAb (top) and ART (bottom) experiments. (FIG. IB) Plasma viremia for untreated mice. The x-axis is in days post HIV-1 challenge. The y-axis is viral RNA copies/ml. Gray shading indicates values beneath the detection limit of 800 copies/ml. The blue line indicates the geometric mean plasma viremia. (FIG. 1C) Plasma viremia for ART -treated mice as in FIG. IB. The blue shading indicates the treatment period with ART. (FIG. ID) Plasma viremia for antibody-treated mice. The red arrows indicate antibody tri-mix injections. The dashed red line shows average plasma antibody concentration for all mice in the group. (FIG. IE) Graph as in FIG. ID, for mice treated with antibody starting 8 days after HIV-1 challenge. (FIG. IF) Percentage of CD4+ T cells
in the spleen at the terminal point measured by flow cytometry, organized by treatment group. (A = aviremic, V =viremic). (FIG. 1G) Cell-associated HIV-1 RNA measured in spleen T cells at the terminal point, plotted as the ratio of HIV-1 RNA to CCR5 copies for each mouse. Gray shading indicates the detection limit of 1.25xl0~5 copies per cell. (FIG. 1H) Cell-associated HIV-1 DNA measured in spleen T cells at the terminal point, plotted as the ratio of HIV-1 RNA to CCR5 copies for each mouse. Gray shading indicates the detection limit of 2.0x10"5 copies per cell.
FIG. 2A, FIG. 2B, FIG. 2C, and FIG. 2D are a set of diagrams showing that FcRnu11 antibodies suppress viremia but do not prevent rebound: (FIG. 2A) Plasma viremia as in FIG. ID for mice treated with FcRnu11 tri-mix. (FIG. 2B) The proportion of mice that were viremic at the terminal point for each treatment group (*, p < 0.05; Fisher's Exact test). (FIG. 2C) For all viremic mice, plasma antibody concentration on the day of viral rebound. Antibody levels were significantly higher in FcRnu11 tri-mix treated mice compared to wild-type tri-mix treated mice (*,p < 0.05; **, p < 0.01; Mann- Whitney test). (FIG. 2D) Sequences of gpl20 cloned from plasma. Horizontal lines denote individual clones, grouped by mouse, shown on the right. Red ticks and green ticks indicate non- synonymous and synonymous substitutions relative to gpl20yu2, respectively. Blue shading highlights sites of mutations that confer escape to the antibody tri-mix.
FIG. 3A, FIG. 3B, FIG. 3C, FIG. 3D, and FIG. 3E are a set of diagrams showing rebound viremia after therapy with single inducers: (FIG. 3A) Schematic timeline of the experiment. (FIG. 3B, FIG. 3C, and FIG. 3D) Graphs show plasma viremia for individual mice on the left y-axis, geometric mean antibody level on the right y-axis among all mice in the group (red). The x-axis represents days relative to the first antibody injection. Antibody injections are indicated with red arrows. Mice that had rebound plasma viremia are shown in gray. Mice that failed to rebound are shown in black. (FIG. 3B) Mice that received tri-mix antibodies, but no inducers. (FIG. 3C) Mice that received tri-mix antibodies and vorinostat (green arrows). (FIG. 3D) Mice that received tri-mix antibodies and I-BET151 (purple shading). (FIG. 3E) Mice that received tri-mix antibodies and aCTLA4 (orange arrows).
FIG. 4A and FIG. 4B are a set of diagrams showing that combination inducers decrease the incidence of rebound viremia: (FIG. 4A) Mice treated with tri-mix of
antibodies, and a combination of three inducers. Graph, arrows, and shading are as in FIG. 3. (FIG. 4B) Graph shows the proportion of mice that showed rebound viremia for each treatment group, where all mice that received antibody tri-mix and any one of the three single inducers (shown in FIGs. 3C, 3D, and 3E) are pooled together (*, p < 0.05; Mann- Whitney test).
FIG. 5A, FIG. 5B, FIG. 5C, FIG. 5D, FIG. 5E, FIG. 5F, and FIG. 5G are a set of diagrams showing antibody persistence and premature termination do not account for non- rebounding: (FIG. 5A) Percentage of CD4+ T cells at the terminal point measured in the spleen by flow cytometry. (FIG. 5B) Cell-associated HIV-1 RNA measured in spleen cells at terminal point, plotted as the ratio of HIV-1 RNA to CCR5 DNA copies for each mouse. Mice that had measureable HIV-1 RNA, but undetectable CCR5 DNA are plotted as 104 copies per cell. (FIG. 5C) Plasma viremia before therapy was initiated for each mouse, organized by treatment group and rebound status (N.R. = non-rebounder, Reb. = viral rebounder). There was no significant difference for any individual group (Kruskal- Wallis test). (FIG. 5D) The plasma antibody level at the time of viral rebound for each mouse that rebounded, organized by treatment group. The mean plasma antibody level at the time of rebound was 2.97 μg/ml for all groups. (FIG. 5E) For each mouse that rebounded, the number of days that elapsed from when the antibody level dropped below 2.97 μg/ml to the time of rebound. (FIG. 5F) For mice that did not rebound, the number of days that elapsed from when each mouse's antibody levels dropped below 2.97 μg/ml to the terminal point. (FIG. 5G) Cell-associated HIV-1 DNA measured in spleen T cells at the terminal point, plotted as the ratio of HIV-1 DNA to CCR5 copies for each mouse. Mice that had measureable HIV-1 DNA, but undetectable CCR5 DNA are plotted as 104 copies per cell.
FIG. 6A and FIG. 6B are set of diagrams showing viremia and antibody levels in individual mice (bNAb Post-exposure prophylaxis) as in FIG. ID: (FIG. 6 A) Each mouse shown in FIG. ID is shown individually. (FIG. 6B) Each mouse shown in FIG. IE is shown individually.
FIG. 7 is a set of diagram showing viremia and antibody levels in individual mice (FcRnu11 tri -mix Post-exposure prophylaxis) as in FIG. ID. Each mouse shown in FIG. 2A is shown individually.
FIG. 8A, FIG. 8B, FIG. 8C, and FIG. 8D are a set of diagram showing viremia and antibody levels in individual mice (antibody only and antibody plus single inducers) in FIGs. 3A-3D: (FIG. 8A) Each mouse shown in Fig. 3A is shown individually. (FIG. 8B) Each mouse shown in FIG. 3B is shown individually. (FIG. 8C) Each mouse shown in FIG. 3C is shown individually. (FIG. 8D) Each mouse shown in FIG. 3D is shown individually.
FIG. 9 is a set of diagram showing viremia in individual mice (antibody plus combination inducers) as in FIG. 4A. Each mouse shown in FIG. 4A is shown individually.
FIG. 10A, FIG. 10B, and FIG. 10 are a set of diagram showing sustained viremia in inducers treated mice without therapy: (FIG. 10A) Graphs shown as in FIG. 4A. The blue line indicates the geometric mean viremia across all mice. (FIG. 10B) Proportion of human CD45+ leukocytes in peripheral blood, measured by flow cytometry. (FIG. IOC) Percentage of CD4+ T cells in the peripheral blood measured by flow cytometry. DETAILED DESCRIPTION OF THE INVENTION
This invention is based, at least in part, on unexpected discoveries (i) that bNAbs are more effective than ART in preventing the establishment of the latent reservoirs of HIV-1 infected cells by a mechanism that requires Fc receptor function and (ii) that bNAbs in combination with viral transcription inducers that act by independent mechanisms synergize to decrease the size of the reservoir as measured by prevention of viral rebound. Thus, combinations of viral transcription inducers and bNAbs constitute a therapeutic strategy that impacts the establishment and maintenance of the HIV-1 reservoir.
A. HIV-1 Latent Reservoir
HIV-1 viruses are transmitted as single-stranded, positive-sense, enveloped RNA viruses. Upon entry into a target host cell, the viral RNA genome is reverse transcribed into double-stranded DNA by a virally encoded reverse transcriptase that is transported along with the viral genome in the virus particle. The resulting viral DNA is then imported into the cell nucleus and integrated into the cellular DNA by a virally encoded integrase and host co-factors. Once integrated, the virus may become latent, allowing the
virus and its host cell to avoid detection by the immune system. HIV-1 persists in infected individuals in a stable pool or reservoir of resting CD4+ T cells and other cells as a latent but replication-competent provirus. Accordingly, as used herein, the term "latent reservoir" refers to cells or sites in a host that are latently infected with a microbe (e.g., HIV). Its presence and size of a HIV latent reservoir can be measured or assessed by viral rebound after terminating therapy as disclosed in the examples below.
Such a latent reservoir of HIV-1 infected cells cannot be cleared by combined ART and remains the major barrier to curing HIV-1 infection. In humans, macaques and humanized mice, the latent reservoir is established within days of initial infection and persists for the lifetime of the individual, making ART a lifelong necessity. While the latent reservoir's exact cellular composition is debated, it is generally believed that it consists primarily of CD4+ memory T cells harboring replication competent provirus that is transcriptionally silenced. Because these cells have very long half-lives and may be able to undergo homeostatic proliferation, a "shock and kill" approach has been proposed to eradicate this reservoir by combining ART with inducers of viral transcription. However, all attempts at altering the HIV-1 reservoir have failed to date.
As disclosed herein, bNAbs can be used in preventing the establishment of the reservoir by a mechanism that requires Fc receptor function. In established infection bNAbs plus single inducers are ineffective in decreasing the reservoir size. Surprisingly, bNAbs plus a combination of inducers that act by independent mechanisms synergize to decrease the size of the reservoir as measured by prevention of viral rebound. Thus, combinations of inducers and bNAbs constitute a therapeutic strategy that can be used to prevent or disrupt the establishment and maintenance of the HIV-1 reservoir in humans and related animal models. B. Antibodies
The invention disclosed herein involves broadly neutralizing HIV-1 antibodies. These antibodies refer to a class of neutralizing antibodies that neutralize multiple HIV-1 viral strains. Various bNAbs are known in the art and can be used in this invention. Examples include but are not limited to those described in U.S. Patent NO. 8673307, WO 2014063059, WO2012158948, and U.S. Provisional Application 61/934,359, including antibodies 3BNC117, 3BNC60, 12A12, 12A21, NIH45-46, bANC131, 8ANC134,
IB2530, INC9, 8ANC195. 8ANC196, 10-259, 10-303, 10-410, 10- 847, 10-996, 10-1074, 10-1121, 10-1130, 10-1146, 10-1341, 10-1369, and 10-1074GM. Additional examples include those described in Klein et al, Nature, 2012. 492(7427): p. 118— 22, Horwitz et al, Proc Natl Acad Sci U S A, 2013. 110(41): p. 16538—43, Scheid, et al. 2011. Science, 333: 1633-1637, Scheid, et al. 2009. Nature, 458:636-640, Eroshkin et al., Nucleic Acids Res. 2014 Jan;42 (Database issue):Dl 133-9, Mascola et al. /mmunol Rev. 2013 Jul;254(l):225-44, such as those listed below.
Table 1.
Listed below are the heavy chain variable regions (HC) sequences and light chain variable regions (LC) of some of the antibodies mentioned above, where the CDRs are in bold.
The heavy chain variable regions (IgH) and light chain variable regions (IgL) of additional antibodies are listed below, where the CDRs are in bold.
I H SE UEN(ES
Shown below are the amino acid sequences of PG16 heavy chain and light chain:
PG16 Igyl
(T) MGWSCIILFLVATATGVHSQEQLVESGGGWQPGGSLRLSCLASGFTFHKYGMHWVR QAPGKGLEWVALISDDGMRKYHSDSMWGRVTISRDNSKNTLYLQFSSLKVEDTAMFFCAR
EAGGPIWHDDVKYYDFNDGYYNYHYMDVWGKGTTVTVSSASTKGPSVFPLAPSSKSTSGG
TAALGCLVKDYFPEPVTVSWNSGALTSGVHTFPAVLQSSGLYSLSSVVTVPSSSLGTQTY ICNVNHKPSNTKVDKKVEPKSCDKTHTCPPCPAPELLGGPSVFLFPPKPKDTLMI SRTPE VTCVVVDVSHEDPEVKFNWYVDGVEVHNAKTKPREEQYNSTYRVVSVLTVLHQDWLNGKE YKCKVSNKALPAPIEKTI SKAKGQPREPQVYTLPPSRDELTKNQVSLTCLVKGFYPSDIA VEWESNGQPENNYKTTPPVLDSDGSFFLYSKLTVDKSRWQQGNVFSCSVMHEALHNHYTQ KSLSLSPGK (SEQ ID No: 54)
PG16 IqX2
(T) MGWSCIILFLVATATGSVTQSALTQPASVSGSPGQTITISCNGTSSDVGGFDSVSWY QQSPGKAPKVMVFDVSHRPSGISNRFSGSKSGNTASLTISGLHIEDEGDYFCSSLTDRSH
RIFGGGTKVTVLGQPKAAPSVTLFPPSSEELQANKATLVCLI SDFYPGAVTVAWKADSSP
VKAGVETTTPSKQSNNKYAASSYLSLTPEQWKSHRSYSCQVTHEGSTVEKTVAPTECS (S
EQ ID No: 55)
Italic: leader peptides
Bold: variable domains
Underlined: constant domains
Like ART, bNAbs can completely suppress viremia in HIV-1 infected humanized mice (Klein et al, Nature, 2012. 492(7427): p. 118-22 and Horwitz et al, Proc Natl Acad Sci U S A, 2013. 110(41): p. 16538-43) and in SHIV infected macaques (Barouch et al, Nature, 2013. 503(7475): p. 224-8 and Shingai et al, Nature, 2013. 503(7475): p. 277-80). Similar to humans, termination of bNAb or ART therapy in hu-mice and macaques results in robust viral rebound, indicating persistence of a functionally silent pool of cells harboring replication-competent virus. Moreover, the relative frequency of latent CD4+ T cells as measured by ex vivo re-activation is similar in ART suppressed hu-mice and humans (Marsden et al, J Virol, 2012. 86(1): p. 339-47 and Denton et al, J Virol, 2012. 86(1): p. 630-4). Therefore, antibodies and ART contain HIV-1 infection in hu-mice but also produce a latent reservoir.
Unlike ART however, antibodies can engage the host immune system by virtue of their Fc effector domains (Nimmerjahn et al, Nat Rev Immunol, 2008. 8(1): p. 34-47) and thereby accelerate clearance of cell free virus (Igarashi et al, Nat Med, 1999. 5(2): p. 211- 6), induce antibody dependent cytotoxicity to kill infected cells (Chung et al, Proc Natl Acad Sci U S A, 2011. 108(18): p. 7505-10; Sun et al, J Virol, 2011. 85(14): p. 6906- 12 Bonsignori et al, J Virol, 2012. 86(21): p. 11521-32; Jost et al, Annu Rev Immunol, 2013. 31 : p. 163-94; Formal et al, J Virol, 2001. 75(15): p. 6953-61 and Formal et al, Curr Opin HIV AIDS, 2013. 8(5): p. 393-401) and produce immune complexes that activate dendritic cells to become potent antigen presenting cells (Dhodapkar et al, Proc Natl Acad Sci U S A, 2005. 102(8): p. 2910-5). Finally, bNAbs can prevent cell-cell transmission of HIV-1 (Malbec et al, J Exp Med, 2013. 210(13): p. 2813-21 and Abela et al, PLoS Pathog, 2012. 8(4): p. el002634), whereas ART's activity in this regard is still debated (Sigal et al, Nature, 2011. 477(7362): p. 95-8; Schiffner et al, Vaccine, 2013. 31(49): p. 5789- 97; and Agosto et al, PLoS Pathog, 2014. 10(2): p. el003982).
The term "antibody" (Ab) as used herein includes monoclonal antibodies, polyclonal antibodies, multispecific antibodies (for example, bispecific antibodies and polyreactive antibodies), and antibody fragments. Thus, the term "antibody" as used in any context within this specification is meant to include, but not be limited to, any specific binding member, immunoglobulin class and/or isotype (e.g., IgGl , IgG2, IgG3, IgG4, IgM, IgA, IgD, IgE and IgM); and biologically relevant fragment or specific binding member thereof, including but not limited to Fab, F(ab')2, Fv, and scFv (single chain or related entity). It is understood in the art that an antibody is a glycoprotein comprising at least two heavy (H) chains and two light (L) chains inter-connected by disulfide bonds, or an antigen binding portion thereof. A heavy chain is comprised of a heavy chain variable region (VH) and a heavy chain constant region (CHI , CH2 and CH3). A light chain is comprised of a light chain variable region (VL) and a light chain constant region (CL). The variable regions of both the heavy and light chains comprise framework regions (FWR) and complementarity determining regions (CDR). The four FWR regions are relatively conserved while CDR regions (CDR1 , CDR2 and CDR3) represent hypervariable regions and are arranged from NH2 terminus to the COOH terminus as follows: FWR1 , CDR1 , FWR2, CDR2, FWR3, CDR3, and FWR4. The variable regions of the heavy and light chains contain a binding domain that interacts with an antigen while, depending of the isotype, the constant region(s) may mediate the binding of the immunoglobulin to host tissues or factors.
Also included in the definition of "antibody" as used herein are chimeric antibodies, humanized antibodies, and recombinant antibodies, human antibodies generated from a transgenic non-human animal, as well as antibodies selected from libraries using enrichment technologies available to the artisan.
The term "variable" refers to the fact that certain segments of the variable (V) domains differ extensively in sequence among antibodies. The V domain mediates antigen binding and defines specificity of a particular antibody for its particular antigen. However, the variability is not evenly distributed across the 1 10-amino acid span of the variable regions. Instead, the V regions consist of relatively invariant stretches called framework regions (FRs) of 15-30 amino acids separated by shorter regions of extreme variability called "hypervariable regions" that are each 9-12 amino acids long. The
variable regions of native heavy and light chains each comprise four FRs, largely adopting a beta sheet configuration, connected by three hypervariable regions, which form loops connecting, and in some cases forming part of, the beta sheet structure. The hypervariable regions in each chain are held together in close proximity by the FRs and, with the hypervariable regions from the other chain, contribute to the formation of the antigen- binding site of antibodies (see, for example, Kabat et al., Sequences of Proteins of Immunological Interest, 5th Ed. Public Health Service, National Institutes of Health, Bethesda, Md. (1991)).
The term "hypervariable region" as used herein refers to the amino acid residues of an antibody that are responsible for antigen binding. The hypervariable region generally comprises amino acid residues from a "complementarity determining region" ("CDR").
The term "monoclonal antibody" as used herein refers to an antibody obtained from a population of substantially homogeneous antibodies, i.e., the individual antibodies comprising the population are identical except for possible naturally occurring mutations that may be present in minor amounts. The term "polyclonal antibody" refers to preparations that include different antibodies directed against different determinants ("epitopes").
The monoclonal antibodies herein include "chimeric" antibodies in which a portion of the heavy and/or light chain is identical with, or homologous to, corresponding sequences in antibodies derived from a particular species or belonging to a particular antibody class or subclass, while the remainder of the chain(s) is identical with, or homologous to, corresponding sequences in antibodies derived from another species or belonging to another antibody class or subclass, as well as fragments of such antibodies, so long as they exhibit the desired biological activity (see, for example, U.S. Pat. No. 4,816,567; and Morrison et al, Proc. Natl. Acad. Sci. USA, 81 :6851-6855 (1984)). Chimeric antibodies include antibodies having one or more human antigen binding sequences (for example, CDRs) and containing one or more sequences derived from a non-human antibody, for example, an FR or C region sequence. In addition, chimeric antibodies included herein are those comprising a human variable region antigen binding sequence of one antibody class or subclass and another sequence, for example, FR or C region sequence, derived from another antibody class or subclass.
A "humanized antibody" generally is considered to be a human antibody that has one or more amino acid residues introduced into it from a source that is non-human. These non-human amino acid residues often are referred to as "import" residues, which typically are taken from an "import" variable region. Humanization may be performed following the method of Winter and co-workers (see, for example, Jones et al, Nature 321 :522-525 (1986); Reichmann et al, Nature 1>Ί>2:Ί>2Ί>-Ί>2Ί (1988); Verhoeyen et al, Science 239: 1534-1536 (1988)), by substituting import hypervariable region sequences for the corresponding sequences of a human antibody. Accordingly, such "humanized" antibodies are chimeric antibodies (see, for example, U.S. Pat. No. 4,816,567), where substantially less than an intact human variable region has been substituted by the corresponding sequence from a non-human species.
An "antibody fragment" comprises a portion of an intact antibody, such as the antigen binding or variable region of the intact antibody. Examples of antibody fragments include, but are not limited to, Fab, Fab', F(ab')2, and Fv fragments; diabodies; linear antibodies (see, for example, U.S. Pat. No. 5,641,870; Zapata et al, Protein Eng. 8(10): 1057-1062 [1995]); single-chain antibody molecules; and multispecific antibodies formed from antibody fragments.
"Fv" is the minimum antibody fragment that contains a complete antigen- recognition and antigen-binding site. This fragment contains a dimer of one heavy- and one light-chain variable region domain in tight, non-covalent association. From the folding of these two domains emanate six hypervariable loops (three loops each from the H and L chain) that contribute the amino acid residues for antigen binding and confer antigen binding specificity to the antibody. However, even a single variable region (or half of an Fv comprising only three CDRs specific for an antigen) has the ability to recognize and bind antigen, although at a lower affinity than the entire binding site.
"Single-chain Fv" ("sFv" or "scFv") are antibody fragments that comprise the VH and VL antibody domains connected into a single polypeptide chain. The sFv polypeptide can further comprise a polypeptide linker between the VH and VL domains that enables the sFv to form the desired structure for antigen binding. For a review of sFv, see, for example, Pluckthun in The Pharmacology of Monoclonal Antibodies, vol. 113, Rosenburg and Moore eds., Springer-Verlag, New York, pp. 269-315 (1994); Borrebaeck 1995, infra.
The term "diabodies" refers to small antibody fragments prepared by constructing sFv fragments with short linkers (about 5-10 residues) between the VH and VL domains such that inter-chain but not intra-chain pairing of the V domains is achieved, resulting in a bivalent fragment, i.e., fragment having two antigen-binding sites. Bispecific diabodies are heterodimers of two "crossover" sFv fragments in which the VH and VL domains of the two antibodies are present on different polypeptide chains. Diabodies are described more fully in, for example, EP 404,097; WO 93/11161; and Hollinger et al, Proc. Natl. Acad. Sci. USA, 90:6444-6448 (1993).
Domain antibodies (dAbs), which can be produced in fully human form, are the smallest known antigen-binding fragments of antibodies, ranging from about 11 kDa to about 15 kDa. DAbs are the robust variable regions of the heavy and light chains of immunoglobulins (VH and VL, respectively). They are highly expressed in microbial cell culture, show favorable biophysical properties including, for example, but not limited to, solubility and temperature stability, and are well suited to selection and affinity maturation by in vitro selection systems such as, for example, phage display. DAbs are bioactive as monomers and, owing to their small size and inherent stability, can be formatted into larger molecules to create drugs with prolonged serum half-lives or other pharmacological activities. Examples of this technology have been described in, for example, W09425591 for antibodies derived from Camelidae heavy chain Ig, as well in US20030130496 describing the isolation of single domain fully human antibodies from phage libraries.
Fv and sFv are the only species with intact combining sites that are devoid of constant regions. Thus, they are suitable for reduced nonspecific binding during in vivo use. sFv fusion proteins can be constructed to yield fusion of an effector protein at either the amino or the carboxy terminus of an sFv. See, for example, Antibody Engineering, ed. Borrebaeck, supra. The antibody fragment also can be a "linear antibody", for example, as described in U.S. Pat. No. 5,641,870 for example. Such linear antibody fragments can be monospecific or bispecific.
In certain embodiments, antibodies of the described invention are bispecific or multi-specific. Bispecific antibodies are antibodies that have binding specificities for at least two different epitopes. Exemplary bispecific antibodies can bind to two different epitopes of a single antigen. Other such antibodies can combine a first antigen binding
site with a binding site for a second antigen. Alternatively, an anti-HIV arm can be combined with an arm that binds to a triggering molecule on a leukocyte, such as a T-cell receptor molecule (for example, CD3), or Fc receptors for IgG (Fc gamma R), such as Fc gamma RI (CD64), Fc gamma RII (CD32) and Fc gamma RIII (CD 16), so as to focus and localize cellular defense mechanisms to the infected cell. Bispecific antibodies also can be used to localize cytotoxic agents to infected cells. Bispecific antibodies can be prepared as full length antibodies or antibody fragments (for example, F(ab')2 bispecific antibodies). See for example, WO 96/16673, U.S. Pat. No. 5,837,234, WO98/02463, U.S. Pat. No. 5,821,337, and Mouquet et al, Nature. 467, 591-5 (2010).
Methods for making bispecific antibodies are known in the art. Traditional production of full length bispecific antibodies is based on the co-expression of two immunoglobulin heavy chain-light chain pairs, where the two chains have different specificities (see, for example, Millstein et al, Nature, 305:537-539 (1983)). Similar procedures are disclosed in, for example, WO 93/08829, Traunecker et al, EMBO J., 10:3655-3659 (1991) and see also; Mouquet et al, Nature. 467, 591-5 (2010). Techniques for generating bispecific antibodies from antibody fragments also have been described in the literature. For example, bispecific antibodies can be prepared using chemical linkage. See Brennan et al, Science, 229: 81 (1985).
Typically, the antibodies of described in the invention can be produced using conventional hybridoma technology or made recombinantly using vectors and methods available in the art. Human antibodies also can be generated by in vitro activated B cells (see, for example, U.S. Pat. Nos. 5,567,610 and 5,229,275). General methods in molecular genetics and genetic engineering useful in the present invention are described in the current editions of Molecular Cloning: A Laboratory Manual (Sambrook, et al., Molecular Cloning: A Laboratory Manual (Fourth Edition) Cold Spring Harbor Lab. press, 2012), Gene Expression Technology (Methods in Enzymology, Vol. 185, edited by D. Goeddel, 1991. Academic Press, San Diego, CA), "Guide to Protein Purification" in Methods in Enzymology (M.P. Deutshcer, ed., (1990) Academic Press, Inc.); PCR Protocols: A Guide to Methods and Applications (Innis, et al. 1990. Academic Press, San Diego, CA), Culture of Animal Cells: A Manual of Basic Technique, 2nd Ed. (R.I. Freshney. 1987. Liss, Inc. New York, NY), and Gene Transfer and Expression Protocols,
pp. 109-128, ed. E.J. Murray, The Humana Press Inc., Clifton, N.J.). Reagents, cloning vectors, and kits for genetic manipulation are available from commercial vendors such as BioRad, Stratagene, Invitrogen, ClonTech and Sigma- Aldrich Co.
Human antibodies also can be produced in transgenic animals (for example, mice) that are capable of producing a full repertoire of human antibodies in the absence of endogenous immunoglobulin production. For example, it has been described that the homozygous deletion of the antibody heavy-chain joining region (JH) gene in chimeric and germ-line mutant mice results in complete inhibition of endogenous antibody production. Transfer of the human germ-line immunoglobulin gene array into such germ- line mutant mice results in the production of human antibodies upon antigen challenge. See, for example, Jakobovits et al, Proc. Natl. Acad. Sci. USA, 90:2551 (1993); Jakobovits et al, Nature, 362:255-258 (1993); Bruggemann et al, Year in Immuno., 7:33 (1993); U.S. Pat. Nos. 5,545,806, 5,569,825, 5,591,669 (all of GenPharm); U.S. Pat. No. 5,545,807; and WO 97/17852. Such animals can be genetically engineered to produce human antibodies comprising a polypeptide of the described invention.
Various techniques have been developed for the production of antibody fragments. Traditionally, these fragments were derived via proteolytic digestion of intact antibodies (see, for example, Morimoto et al., Journal of Biochemical and Biophysical Methods 24: 107-117 (1992); and Brennan et al, Science, 229:81 (1985)). However, these fragments can now be produced directly by recombinant host cells. Fab, Fv and ScFv antibody fragments can all be expressed in and secreted from E. coli, thus allowing the facile production of large amounts of these fragments. Fab'-SH fragments can be directly recovered from E. coli and chemically coupled to form F(ab')2 fragments (see, for example, Carter et al., Bio/Technology 10: 163-167 (1992)). According to another approach, F(ab')2 fragments can be isolated directly from recombinant host cell culture. Fab and F(ab')2 fragment with increased in vivo half-life comprising a salvage receptor binding epitope residues are described in U.S. Pat. No. 5,869,046. Other techniques for the production of antibody fragments will be apparent to the skilled practitioner.
Other techniques that are known in the art for the selection of antibody fragments from libraries using enrichment technologies, including but not limited to phage display, ribosome display (Hanes and Pluckthun, 1997, Proc. Nat. Acad. Sci. 94: 4937-4942),
bacterial display (Georgiou, et al., 1997, Nature Biotechnology 15: 29-34) and/or yeast display (Kieke, et al., 1997, Protein Engineering 10: 1303-1310) may be utilized as alternatives to previously discussed technologies to select single chain antibodies. Single- chain antibodies are selected from a library of single chain antibodies produced directly utilizing filamentous phage technology. Phage display technology is known in the art (e.g., see technology from Cambridge Antibody Technology (CAT)) as disclosed in U.S. Patent Nos. 5,565,332; 5,733,743; 5,871,907; 5,872,215; 5,885,793; 5,962,255; 6,140,471; 6,225,447; 6,291650; 6,492,160; 6,521,404; 6,544,731; 6,555,313; 6,582,915; 6,593, 081, as well as other U.S. family members, or applications which rely on priority filing GB 9206318, filed 24 May 1992; see also Vaughn, et al. 1996, Nature Biotechnology 14: 309- 314). Single chain antibodies may also be designed and constructed using available recombinant DNA technology, such as a DNA amplification method (e.g., PCR), or possibly by using a respective hybridoma cDNA as a template.
Variant antibodies also are included within the scope of the invention. Thus, variants of the sequences recited in the application also are included within the scope of the invention. Further variants of the antibody sequences having improved affinity can be obtained using methods known in the art and are included within the scope of the invention. For example, amino acid substitutions can be used to obtain antibodies with further improved affinity. Alternatively, codon optimization of the nucleotide sequence can be used to improve the efficiency of translation in expression systems for the production of the antibody.
In certain embodiments, an antibody of the invention comprises a heavy chain variable region comprising CDRl, CDR2 and CDR3 sequences and a light chain variable region comprising CDRl, CDR2 and CDR3 sequences, wherein one or more of these CDR sequences comprise specified amino acid sequences based on the preferred antibodies described herein, or conservative modifications thereof, and wherein the antibodies retain the desired functional properties of neutralizing multiple HIV-1 viral strains. As used herein, the term "conservative sequence modifications" refers to amino acid modifications that do not significantly affect or alter the binding characteristics of the antibody containing the amino acid sequence. Such conservative modifications include amino acid substitutions, additions and deletions. Modifications can be introduced into an
antibody of the invention by standard techniques known in the art, such as site-directed mutagenesis and PCR-mediated mutagenesis. Conservative amino acid substitutions are ones in which the amino acid residue is replaced with an amino acid residue having a similar side chain. Families of amino acid residues having similar side chains have been defined in the art. These families include
amino acids with basic side chains (e.g., lysine, arginine, histidine),
acidic side chains (e.g., aspartic acid, glutamic acid),
uncharged polar side chains (e.g., glycine, asparagine, glutamine, serine, threonine, tyrosine, cysteine, tryptophan),
nonpolar side chains (e.g., alanine, valine, leucine, isoleucine, proline, phenylalanine, methionine),
beta-branched side chains (e.g., threonine, valine, isoleucine) and
aromatic side chains (e.g., tyrosine, phenylalanine, tryptophan, histidine).
Thus, one or more amino acid residues within the CDR regions of an antibody of the invention can be replaced with other amino acid residues from the same side chain family and the altered antibody can be tested for retained function using the functional assays described herein.
Other modifications of the antibody are contemplated herein. For example, the antibody can be linked to one of a variety of nonproteinaceous polymers, for example, polyethylene glycol, polypropylene glycol, polyoxyalkylenes, or copolymers of polyethylene glycol and polypropylene glycol. The antibody also can be entrapped in microcapsules prepared, for example, by coacervation techniques or by interfacial polymerization (for example, hydroxymethylcellulose or gelatin-microcapsules and poly- (methylmethacylate) microcapsules, respectively), in colloidal drug delivery systems (for example, liposomes, albumin microspheres, microemulsions, nano-particles and nanocapsules), or in macroemulsions. Such techniques are disclosed in, for example, Remington's Pharmaceutical Sciences, 16th edition, Oslo, A., Ed., (1980).
As disclosed herein, bNAbs are significantly more effective than ART in blocking the establishment of the reservoir when given early in the infection. One of the key differences between antibodies and ART is that antibodies can engage a variety of host immune effector pathways by way of their Fc receptors (Nimmerjahn et al., Nat Rev
Immunol, 2008. 8(1): p. 34-47). Consistent with this important difference, the mechanism by which antibodies interfere with the establishment of the reservoir is dependent on their ability to bind to Fc receptors.
The terms "Fc receptor" or "FcR" are used to describe a receptor that binds to the Fc region of an antibody. An Fc receptor is a protein found on the surface of certain cells - including, among others, B lymphocytes, follicular dendritic cells, natural killer cells, macrophages, neutrophils, eosinophils, basophils and mast cells - that contribute to the protective functions of the immune system. Its name is derived from its binding specificity for the Fc region (fragment crystallizable region) of an antibody.
Several antibody functions are mediated by Fc receptors. For example, Fc receptors bind to antibodies that are attached to infected cells or invading pathogens. Their activity stimulates phagocytic or cytotoxic cells to destroy microbes, or infected cells by antibody-mediated phagocytosis or antibody-dependent cell-mediated cytotoxicity. It was also known in the art that the Fc region of an antibody ensures that each antibody generates an appropriate immune response for a given antigen, by binding to a specific class of Fc receptors, and other immune molecules, such as complement proteins. FcRs are defined by their specificity for immunoglobulin isotypes: Fc receptors for IgG antibodies are referred to as FcyR, for IgE as FcsFR, for IgA as FcaR and so on. Surface receptors for immunoglobulin G are present in two distinct classes-those that activate cells upon their crosslinking ("activation FcRs") and those that inhibit activation upon co-engagement ("inhibitory FcRs").
In all mammalian species studied to date, four different classes of IgG Fc-receptors have been defined: FcyRI (CD64), FcyRII (CD32), FcyRIII (CDI6) and FcylV. Whereas FcyRI displays high affinity for the antibody constant region and restricted isotype specificity, FcyRII and FcyRIII have low affinity for the Fc region of IgG but a broader isotype binding pattern (Ravetch and Kinet, 1991; Hulett and Hogarth, Adv Immunol 57, 1-127 (1994)). FcyRIV is a recently identified receptor, conserved in all mammalian species with intermediate affinity and restricted subclass specificity (Mechetina et al., Immunogenetics 54, 463-468 (2002); Davis et al, Immunol Rev 190, 123-136 (2002); Nimmerjahn et al, Immunity 23, 41-51 (2005)).
Functionally there are two different classes of Fc-receptors: the activation and the inhibitory receptors, which transmit their signals via immunoreceptor tyrosine based activation (ITAM) or inhibitory motifs (ITIM), respectively (Ravetch, in Fundamental Immunology W. E. Paul, Ed. (Lippincott-Raven, Philadelphia, (2003); Ravetch and Lanier, Science 290, 84-89 (2000). The paired expression of activating and inhibitory molecules on the same cell is the key for the generation of a balanced immune response. Additionally, it has been appreciated that the IgG Fc-receptors show significant differences in their affinity for individual antibody isotypes rendering certain isotypes more strictly regulated than others (Nimmerjahn et al., 2005).
In one embodiment of the invention, FcR is a native sequence human FcR. In another embodiment, FcR, including human FcR, binds an IgG antibody (a gamma receptor) and includes receptors of the FcyRI, FcyRII, and FcyRIII subclasses, including allelic variants and alternatively spliced forms of these receptors. FcyRII receptors include FcyRIIA (an "activating receptor") and FcyRIIB (an "inhibiting receptor"), which have similar amino acid sequences that differ primarily in the cytoplasmic domains thereof. Activating receptor FcyRIIA contains an immunoreceptor tyrosine-based activation motif (ITAM) in its cytoplasmic domain. Inhibiting receptor FcyRIIB contains an immunoreceptor tyrosine-based inhibition motif (ITIM) in its cytoplasmic domain (see review in Daron, Annu Rev Immunol, 15, 203-234 (1997); FcRs are reviewed in Ravetch and Kinet, Annu Rev Immunol, 9, 457-92 (1991); Capel et al, Immunomethods, 4, 25-34 (1994); and de Haas et al, J Lab Clin Med, 126, 330-41 (1995), Nimmerjahn and Ravetch 2006, Ravetch Fc Receptors in Fundemental Immunology, ed William Paul 5th Ed. each of which is incorporated herein by reference).
As used herein, the term "Fc fragment" or "Fc region" is used to define a C- terminal region of an immunoglobulin heavy chain. Such an Fc region is the tail region of an antibody that interacts with Fc receptors and some proteins of the complement system. The Fc region may be a native sequence Fc region or a variant Fc region. Although the boundaries of the Fc region of an immunoglobulin heavy chain might vary, the human IgG heavy chain Fc region is usually defined to stretch from an amino acid residue at position Cys226, or from Pro230, to the carboxyl-terminus thereof. A native sequence Fc region comprises an amino acid sequence identical to the amino acid sequence of an Fc region
found in nature. A variant Fc region as appreciated by one of ordinary skill in the art comprises an amino acid sequence which differs from that of a native sequence Fc region by virtue of at least one "amino acid modification."
In IgG, IgA and IgD antibody isotypes, the Fc region is composed of two identical protein fragments, derived from the second and third constant domains of the antibody's two heavy chains; IgM and IgE Fc regions contain three heavy chain constant domains (CH domains 2-4) in each polypeptide chain. The Fc regions of IgGs bear a highly conserved N-glycosylation site. Glycosylation of the Fc fragment is important for Fc receptor-mediated activity. The N-glycans attached to this site are predominantly core- fucosylated diantennary structures of the complex type. In addition, small amounts of these N-glycans also bear bisecting GlcNAc and a-2,6 linked sialic acid residues. See, e.g., US20080286819, US20100278808, US20100189714, US 2009004179, 20080206246, 20110150867, and WO2013095966, each of which is incorporated herein by reference.
As the Fc receptor function is involved in HIV-1 latent reservoir, the bNAb antibody of this invention can include antibody variable regions with the desired binding specificities (antibody-antigen combining sites) fused to immunoglobulin constant domain sequences. The fusion can be with an Ig heavy chain constant domain, comprising at least part of the hinge, CH2, and CH3 regions. According to some embodiments, the first heavy-chain constant region (CHI) containing the site necessary for light chain bonding, is present in at least one of the fusions. DNAs encoding the immunoglobulin heavy chain fusions and, if desired, the immunoglobulin light chain, are inserted into separate expression vectors, and are co-transfected into a suitable host cell. This provides for greater flexibility in adjusting the mutual proportions of the three polypeptide fragments in embodiments when unequal ratios of the three polypeptide chains used in the construction provide the optimum yield of the desired bispecific antibody. It is, however, possible to insert the coding sequences for two or all three polypeptide chains into a single expression vector when the expression of at least two polypeptide chains in equal ratios results in high yields or when the ratios have no significant effect on the yield of the desired chain combination.
C. Inducers
Some embodiments of this invention involve using viral transcription inducers. A transcription inducer refers to any agent that can induce the transcriptional activation of the HIV-1 promoter, or re-activate latent HIV-1 from the patient viral reservoir directly or indirectly {e.g., facilitating T cell activation by removing an inhibitory pathway). Examples of suitable transcription inducers include vorinostat, an HDAC inhibitor (Contreras et al, J Biol Chem, 2009. 284(11): p. 6782-9; Archin et al, AIDS Res Hum Retroviruses, 2009. 25(2): p. 207-12; and Archin et al, AIDS, 2009. 23(14): p. 1799-806), I-BET151, a BET bromodomain inhibitor (Boehm et al, Cell Cycle, 2013. 12(3): p. 452- 62), and aCTLA4, a T-cell inhibitory pathway blocker (Alegre et al, Nat. Rev. Immunol.200\ . p. 220-228 and Krummel et al., J. Exp. Med. \995. p. 459-465. See also US 20140128391. For example, the use of HDAC inhibitors in combination with the bNAbs of this invention can be used in disrupting, decreasing, or purging the latently infected reservoirs in patients, particularly patients undergoing Highly Active Antiretro viral Therapy (HAART).
Experiments with indicator cell lines indicate that HDAC and bromodomain inhibitors show synergy when combined with conventional transcriptional activators in reactivating HIV-1 in vitro. Consistent with the in vitro experiments, the combination of these inducers appears to be synergistic in vivo since single inducers had no measurable effect above the background controls, while -57% of the hu-mice treated with antibodies plus combination inducers failed to rebound. Irrespective of the mechanism, the reservoir of HIV-1 infected cells remaining after combination inducer and antibody therapy in hu- mice is significantly decreased, establishing the principle that the HIV-1 reservoir can be altered by combination therapy with antibodies and inducers in vivo. D. Other Anti-retroviral Agents
The above-described antibodies and viral transcription inducers can used in combination with one or more anti-retroviral agents for the treatment of HIV latency and/or infection. See, e.g., US 2010/0166806, US 2010/0324034, and US 2012/0203014, which are hereby incorporated in their entirety.
Compositions according to the present invention may also be administered in combination with other agents to enhance the biological activity of such agents. Such
agents may include any one or more of the standard anti-HIV agents which are known in the art, including, but not limited to, azidothymidine (AZT), dideoxycytidine (ddC), and dideoxyinosine (ddl). Additional agents which have shown anti-HIV effects and may be combined with compositions in accordance to the invention include, for example, raltegravir, maraviroc, bestatin, human chorionic gonadotropin (hCG), levamisole, estrogen, efavirenz, etravirine, indomethacin, emtricitabine, tenofovir disoproxil fumarate, amprenavir, tipranavir, indinavir, ritonavir, darunavir, enfuvirtide, and gramicidin.
E. Treatment Compositions and Methods
In one embodiment, the present invention provides a composition comprising at least one bNAb mentioned above alone or in combination with one of the other active agent mentioned above and a pharmaceutically acceptable carrier. The composition may include a plurality of the antibodies having the characteristics described herein in any combination and can further include antibodies neutralizing to HIV as are known in the art.
It is to be understood that compositions can be a single or a combination of antibodies disclosed herein, which can be the same or different, in order to prophylactically or therapeutically treat the progression of various subtypes of HIV infection. When an antibody or active agent is administered to an animal or a human, it can be combined with one or more pharmaceutically acceptable carriers, excipients or adjuvants as are known to one of ordinary skilled in the art.
Further, with respect to determining the effective level in a patient for treatment of HIV, in particular, suitable animal models are available and have been widely implemented for evaluating the in vivo efficacy against HIV of various therapy protocols. These models include mice, monkeys and cats. Even though these animals are not naturally susceptible to HIV disease, chimeric mice models (for example, SCID, bg/nu/xid, NOD/SCID, SCID-hu, immunocompetent SCID-hu, bone marrow-ablated BALB/c) reconstituted with human peripheral blood mononuclear cells (PBMCs), lymph nodes, fetal liver/thymus or other tissues can be infected with lentiviral vector or HIV, and employed as models for HIV pathogenesis. Similarly, the simian immune deficiency virus (SrV)/monkey model can be employed, as can the feline immune deficiency virus (FIV)/cat model. The pharmaceutical composition can contain other pharmaceuticals, in
conjunction with a vector according to the invention, when used to therapeutically treat AIDS. These other pharmaceuticals can be used in their traditional fashion (i.e., as agents to treat HIV infection).
According to another embodiment, the present invention provides an antibody- based pharmaceutical composition comprising an effective amount of an isolated bNAb of the invention, or an affinity matured version, which provides a prophylactic or therapeutic treatment choice to reduce the latent reservoir and infection of the HIV virus. The pharmaceutical compositions of the present invention may be formulated by any number of strategies known in the art (e.g., see McGoff and Scher, 2000, Solution Formulation of Proteins/Peptides: In McNally, E.J., ed. Protein Formulation and Delivery. New York, NY: Marcel Dekker; pp. 139-158; Akers and Defilippis, 2000, Peptides and Proteins as Parenteral Solutions. In: Pharmaceutical Formulation Development of Peptides and Proteins. Philadelphia, PA: Talyor and Francis; pp. 145-177; Akers, et al., 2002, Pharm. Biotechnol. 14:47-127). A pharmaceutically acceptable composition suitable for patient administration will contain an effective amount of the bNAb antibody in a formulation which both retains biological activity while also promoting maximal stability during storage within an acceptable temperature range. The pharmaceutical compositions can also include, depending on the formulation desired, pharmaceutically acceptable diluents, pharmaceutically acceptable carriers and/or pharmaceutically acceptable excipients, or any such vehicle commonly used to formulate pharmaceutical compositions for animal or human administration. The diluent is selected so as not to affect the biological activity of the combination. Examples of such diluents are distilled water, physiological phosphate- buffered saline, Ringer's solutions, dextrose solution, and Hank's solution. The amount of an excipient that is useful in the pharmaceutical composition or formulation of this invention is an amount that serves to uniformly distribute the antibody throughout the composition so that it can be uniformly dispersed when it is to be delivered to a subject in need thereof. It may serve to dilute the antibody or other active agent to a concentration which provides the desired beneficial palliative or curative results while at the same time minimizing any adverse side effects that might occur from too high a concentration. It may also have a preservative effect. Thus, for an active ingredient having a high physiological activity, more of the excipient will be employed. On the other hand, for any
active ingredient(s) that exhibit a lower physiological activity, a lesser quantity of the excipient will be employed.
The above described bNAb antibodies and antibody compositions, comprising at least one or a combination of the antibodies described herein, can be administered for the prophylactic and therapeutic treatment of HIV viral infection.
The composition can be a pharmaceutical composition that contains a pharmaceutically acceptable carrier. The term "pharmaceutical composition" refers to the combination of an active agent with a carrier, inert or active, making the composition especially suitable for diagnostic or therapeutic use in vivo or ex vivo. A "carrier" as used herein includes pharmaceutically acceptable carriers, excipients, or stabilizers that are nontoxic to the cell or mammal being exposed thereto at the dosages and concentrations employed. Often the physiologically acceptable carrier is an aqueous pH buffered solution. Examples of physiologically acceptable carriers include, but not limited to, buffers such as phosphate, citrate, and other organic acids; antioxidants including, but not limited to, ascorbic acid; low molecular weight (less than about 10 residues) polypeptide; proteins, such as, but not limited to, serum albumin, gelatin, or immunoglobulins; hydrophilic polymers such as, but not limited to, polyvinylpyrrolidone; amino acids such as, but not limited to, glycine, glutamine, asparagine, arginine or lysine; monosaccharides, disaccharides, and other carbohydrates including, but not limited to, glucose, mannose, or dextrins; chelating agents such as, but not limited to, EDTA; sugar alcohols such as, but not limited to, mannitol or sorbitol; salt-forming counterions such as, but not limited to, sodium; and/or nonionic surfactants such as, but not limited to, TWEEN.; polyethylene glycol (PEG), and PLURONICS.
The term "pharmaceutically acceptable carrier" refers to any of the standard pharmaceutical carriers, such as a phosphate buffered saline solution, water, emulsions, and various types of wetting agents. The compositions also can include stabilizers and preservatives. A pharmaceutically acceptable carrier, after administered to or upon a subject, does not cause undesirable physiological effects. The carrier in the pharmaceutical composition must be "acceptable" also in the sense that it is compatible with the active ingredient and, preferably, capable of stabilizing it. One or more solubilizing agents can be utilized as pharmaceutical carriers for delivery of an active
agent. Examples of other carriers include colloidal silicon oxide, magnesium stearate, cellulose, and sodium lauryl sulfate.
A "subject" refers to a human and a non-human animal. Examples of a non-human animal include all vertebrates, e.g., mammals, such as non-human primates (particularly higher primates), dog, rodent (e.g., mouse or rat), guinea pig, cat, and non-mammals, such as birds, amphibians, reptiles, etc. In a preferred embodiment, the subject is a human. In another embodiment, the subject is an experimental animal or animal suitable as a disease model (such as non-human primates). A subject to be treated can be identified by standard diagnosing techniques for the disorder.
According to another embodiment, the present invention provides a method of reducing or preventing the establishment of a latent reservoir of HIV infected cells in a subject in need thereof (e.g., a subject infected with HIV or at risk of infection with HIV), thereby treating infection with a HIV infection, comprising administering to the subject a pharmaceutical composition comprising the HIV antibodies disclosed herein. The compositions of the invention can include more than one antibody having the characteristics disclosed (for example, a plurality or pool of antibodies). It also can include other HIV neutralizing antibodies and/or active agent known in the art.
Subjects at risk for HIV-related diseases or disorders include patients who have come into contact with an infected person or who have been exposed to HIV in some other way. Administration of a prophylactic agent can occur prior to the manifestation of symptoms characteristic of HIV-related disease or disorder, such that a disease or disorder is prevented or, alternatively, delayed in its progression.
For in vivo treatment of human and non-human patients, the patient is administered or provided a pharmaceutical formulation including an HIV antibody of the invention. When used for in vivo therapy, the antibodies of the invention are administered to the patient in therapeutically effective amounts (i.e., amounts that eliminate or reduce the patient's latent viral reservoir). The antibodies are administered to a human patient, in accord with known methods, such as intravenous administration, for example, as a bolus or by continuous infusion over a period of time, by intramuscular, intraperitoneal, intracerobrospinal, subcutaneous, intra-articular, intrasynovial, intrathecal, oral, topical, or inhalation routes. The antibodies can be administered parenterally, when possible, at the
target cell site, or intravenously. In some embodiments, antibody is administered by intravenous or subcutaneous administration. Therapeutic compositions of the invention may be administered to a patient or subject systemically, parenterally, or locally. The above parameters for assessing successful treatment and improvement in the disease are readily measurable by routine procedures familiar to a physician.
For parenteral administration, the antibodies may be formulated in a unit dosage injectable form (solution, suspension, emulsion) in association with a pharmaceutically acceptable, parenteral vehicle. Examples of such vehicles include, but are not limited, water, saline, Ringer's solution, dextrose solution, and 5% human serum albumin. Nonaqueous vehicles include, but are not limited to, fixed oils and ethyl oleate. Liposomes can be used as carriers. The vehicle may contain minor amounts of additives such as substances that enhance isotonicity and chemical stability, such as, for example, buffers and preservatives. The antibodies can be formulated in such vehicles at concentrations of about 1 mg/ml to 10 mg/ml.
The dose and dosage regimen depends upon a variety of factors readily determined by a physician, such as the nature of the infection, for example, its therapeutic index, the patient, and the patient's history. Generally, a therapeutically effective amount of an antibody is administered to a patient. In some embodiments, the amount of antibody administered is in the range of about 0.1 mg/kg to about 50 mg/kg of patient body weight. Depending on the type and severity of the infection, about 0.1 mg/kg to about 50 mg/kg body weight (for example, about 0.1-15 mg/kg/dose) of antibody is an initial candidate dosage for administration to the patient, whether, for example, by one or more separate administrations, or by continuous infusion. The progress of this therapy is readily monitored by conventional methods and assays and based on criteria known to the physician or other persons of skill in the art. The above parameters for assessing successful treatment and improvement in the disease are readily measurable by routine procedures familiar to a physician.
Other therapeutic regimens may be combined with the administration of the bNAb HIV antibody of the present invention. The combined administration includes co- administration, using separate formulations or a single pharmaceutical formulation, and consecutive administration in either order, wherein preferably there is a time period while
both (or all) active agents simultaneously exert their biological activities. Such combined therapy can result in a synergistic therapeutic effect. The parameters for assessing successful treatment and improvement in the disease are also readily measurable by routine procedures familiar to a physician.
The terms "treating" or "treatment" or "alleviation" are used interchangeably and refer to both therapeutic treatment and prophylactic or preventative measures; wherein the object is to prevent or slow down (lessen) the targeted pathologic condition or disorder. In particular, it refers to administration of a compound or agent to a subject, who has a disorder (such as an HIV infection), with the purpose to cure, alleviate, relieve, remedy, delay the onset of, prevent, or ameliorate the disorder, the symptom of the disorder, the disease state secondary to the disorder, or the predisposition toward the disorder.
Those in need of treatment include those already with the disorder as well as those prone to have the disorder or those in whom the disorder is to be prevented. A subject or mammal is successfully "treated" for an infection if, after receiving a therapeutic amount of an antibody according to the methods of the present invention, the patient shows observable and/or measurable reduction in or absence of one or more of the following: reduction in the number of infected cells or absence of the infected cells; reduction in the percent of total cells that are infected; and/or relief to some extent, one or more of the symptoms associated with the specific infection; reduced morbidity and mortality, and improvement in quality of life issues. The above parameters for assessing successful treatment and improvement in the disease are readily measurable by routine procedures familiar to a physician.
Eliminating the HIV-1 reservoir in chronic infection is key to curing the disease, but direct measurement of the latent reservoir to evaluate therapeutic eradication strategies remains difficult (Siliciano et al, Curr Opin HIV AIDS, 2013. 8(4): p. 318-25). Quantitative viral outgrowth assays and PCR-based assays of integrated DNA yield variable results (Eriksson et al., PLoS Pathog, 2013. 9(2): p. el003174) in part because PCR cannot distinguish between inactive and permanently disabled proviruses, and outgrowth assays underestimate reservoir size (Ho et al., Cell, 2013. 155(3): p. 540-51). To that end, the most effective way to evaluate the reservoir in vivo is to measure viral rebound after terminating therapy as disclosed in the examples below.
The terms "prevent," "preventing," "prevention," "prophylactic treatment" and the like refer to reducing the probability of developing a disorder or condition in a subject, who does not have, but is at risk of or susceptible to developing a disorder or condition.
A "therapeutically effective amount" refers to the amount of an agent sufficient to effect beneficial or desired results. A therapeutically effective amount can be administered in one or more administrations, applications or dosages and is not intended to be limited to a particular formulation or administration route.
Administration "in combination with" one or more further therapeutic agents includes simultaneous (concurrent) and consecutive administration in any order.
Pharmaceutically effective compositions of this invention may be administered to humans and other animals by a variety of methods that may include continuous or intermittent administration. Examples of methods of administration may include, but are not limited to, oral, rectal, parenteral, intracisternal, intrasternal, intravaginal, intraperitoneal, topical, transdermal, buccal, or as an oral or nasal spray. Accordingly, the pharmaceutically effective compositions may also include pharmaceutically acceptable additives, carriers or excipients. Such pharmaceutical compositions may also include the active ingredients formulated together with one or more non-toxic, pharmaceutically acceptable carriers specially formulated for oral administration in solid or liquid form, for parenteral injection or for rectal administration according to standard methods known in the art.
The term "parenteral" administration refers to modes of administration which include intravenous, intramuscular, intraperitoneal, intracisternal, intrasternal, subcutaneous and intraarticular injection and infusion. Injectable mixtures are known in the art and comprise pharmaceutically acceptable sterile aqueous or nonaqueous solutions, dispersions, suspensions or emulsions as well as sterile powders for reconstitution into sterile injectable solutions or dispersions just prior to use. Examples of suitable aqueous and nonaqueous carriers, diluents, solvents or vehicles include water, ethanol, polyols (such as glycerol, propylene glycol, polyethylene glycol and the like), vegetable oils (such as olive oil), injectable organic esters (such as ethyl oleate) and suitable mixtures thereof.
F. Kits
Another aspect of the invention provides kits. In general, kits according to the present invention comprise an isolated anti-HIV bNAb antibody, a first viral transcription inducer, and a second viral transcription inducer. Components of the kits can be provided in containers. The containers are provided in packaged combination in a suitable package, such as a box made of cardboard, glass, plastic, metal, or a combination thereof. Suitable packaging materials for pharmaceutical compositions or reagents are known and widely used in the art, and thus need not be specified herein.
The kits of the invention can comprise any number of additional reagents or substances that are useful for practicing a method of the invention. Such substances include, but are not limited to: reagents (including buffers) for detecting HIV virus or components thereof or HIV-infected cells in a subject. The kits of the invention can be provided at any temperature. For example, for storage of kits containing protein-based agents, it is preferred that they are provided and maintained below 0 °C, preferably at or below -20 °C, or otherwise in a frozen state.
The kit may further comprise a software package for data analysis of the physiological status of a subject to be treated, which may include reference profiles for comparison with the relevant test profile. Such kits may also include information, such as scientific literature references, package insert materials, clinical trial results, and/or summaries of these and the like, which indicate or establish the activities and/or advantages of the composition, and/or which describe dosing, administration, side effects, drug interactions, or other information useful to the health care provider. Such information may be based on the results of various studies, for example, studies using experimental animals involving in vivo models and studies based on human clinical trials. Kits described herein can be provided, marketed and/or promoted to health providers, including physicians, nurses, pharmacists, formulary officials, and the like. Kits may also, in some embodiments, be marketed directly to the consumer.
As disclosed herein, a number of ranges of values are provided. It is understood that each intervening value, to the tenth of the unit of the lower limit, unless the context clearly dictates otherwise, between the upper and lower limits of that range is also specifically disclosed. Each smaller range between any stated value or intervening value
in a stated range and any other stated or intervening value in that stated range is encompassed within the invention. The upper and lower limits of these smaller ranges may independently be included or excluded in the range, and each range where either, neither, or both limits are included in the smaller ranges is also encompassed within the invention, subject to any specifically excluded limit in the stated range. Where the stated range includes one or both of the limits, ranges excluding either or both of those included limits are also included in the invention.
The term "about" generally refers to plus or minus 10% of the indicated number. For example, "about 10%" may indicate a range of 9% to 11%, and "about 1" may mean from 0.9-1.1. Other meanings of "about" may be apparent from the context, such as rounding off, so, for example "about 1" may also mean from 0.5 to 1.4.
EXAMPLES
Example 1 Materials and Methods
This example descibes materials and methods used in Examples 2 -5 below.
Mice
NOD Ragl^rg^ (NOD.Cg-RagltmlMom I12rgtmlwjl/SzJ, NRG) mice were purchased from The Jackson Laboratory. All mice were bred and maintained at the Comparative Bioscience Center of The Rockefeller University according to guidelines established by the Institutional Animal Committee. All experiments were performed with authorization from the Institutional Review Board and the IACUC at The Rockefeller University.
Humanized Mice
Humanized mice were generated as previously described in Klein et ah, Nature, 2012. 492(7427): p. 118-22. Briefly, human fetal livers were obtained from Advanced Bioscience Resources (ABR). Fetal livers were homogenized and incubated in HBSS media with 0.1% collagenase IV (Sigma- Aldrich), 40 mM HEPES, 2 mM CaCk and 2 U ml"1 DNAase I (Roche) for 30 minutes at 37° C. Hematopoietic Stem Cells (HSCs) were isolated from digested liver using CD34+ HSC isolation kit (Stem Cell Technologies). Neonatal NRG mice (1-5 days old) were sublethally irradiated with 100 cG and injected intrahepatically with 2 X 105 human CD34+ HSCs 6 h after irradiation.
Mouse Screening For Humanization
Eight or more weeks after HSC injection, mice were screened for the presence of human lymphocytes in peripheral blood by flow cytometry. 200 μΐ whole blood was collected by facial vein bleed and peripheral blood mononuclear cells (PBMCs) were isolated by density gradient centrifugation using Ficoll-Paque Plus (GE Healthcare Life Sciences). PBMCs were stained with antibodies to mouse CD45-PECy7, human CD45- Pacific Orange, human CD3-Pacific Blue, human CD19-APC, human CD4-PE, human CD8-FITC, and human CD16-Alexa700 for 25 min at 4 °C. Cells were washed and fixed using Cytofix/Cytoperm (BD Biosciences). Flow cytometry analysis was performed with a LSRFortessa (BD) and Flow Jo software (Tree Star). For each mouse, the percentage of human lymphocytes [(100 x human CD45 ) / (human CD45 + mouse CD45+ )], termed huCD45+ %, and the percentage of human CD4+ T cells (100 x human CD45+ CD3+ CD4+ / human CD45+ ), termed huCD4+ %, was calculated. Mice with at least 10% huCD45+ and 10% huCD4+ were selected for post-exposure prophylaxis experiments, and infected with two doses of HIV-1YU2 (150 ng p24) by i.p. injections, 24 hours apart. Pre-treatment viremia was measured at 72-96 hours following the first HIV-1YU2 injection, and treatment was initiated 4 days following the first injection. For experiments assessing the effects of bNAbs and inducers on established infections, mice with measurable human CD4+ cells by FACS were injected with two doses of HIV-1YU2 (150 ng), and pre-treatment viremia was measured 14-18 days after the first injection. Mice with plasma viral loads >3000 RNA copies/ml were selected to receive antibody therapy. After five subcutaneous antibody injections (see below), post-treatment viremias were measured. Only mice with completely suppressed plasma viremias were selected for further analysis and to receive viral inducers.
Plasma Viral Load Measurements
300-500 μΐ of whole blood was collected from mice at each time point by facial vein bleed. Whole blood was spun at 300g for 10 minutes to separate plasma from the cellular fraction. Total RNA was extracted from 100 μΐ plasma using QIAmp MinElute Virus Spin Kit (Qiagen) in combination with RNase-free DNase (Qiagen), eluted in a 50 μΐ volume. HIV-1 RNA was quantified by qRT-PCR. The reaction mixture was prepared using TaqMan RNA-to-Ct 1-Step kit (Applied Biosystems), with 20 μΐ of eluted RNA,
and a sequence specific probe targeting a conserved region of the HIV-1 pol gene (/HEX/5 '-CCCACCAACARGCRGCCT TAACTG-3 VZenDQ, HXB2 nt 4603 to 4626, SEQ ID No: 44) (Integrated DNA Technologies). Forward and reverse primer sequences were 5 ' -T AATGGC AGC AATTTC ACC A-3 ' (HXB2 nt 4577-4596, SEQ ID No: 45) and 5'- GAATGCC AAATTCCTGCTTGA-3 ' (HXB2 nt 4633 to 4653, SEQ ID No: 46), respectively. Cycle threshold (Ct) values were calibrated using standard samples with known amounts of absolute viral RNA copies. The quantitation limit was previously determined to be 800 copies/ml (Klein et al, Nature, 2012. 492(7427): p. 118-22).
Gpl20 Sequencing
Gpl20 cloning and sequencing was performed as previously described (Klein et al., 2012, HIV therapy by a combination of broadly neutralizing antibodies in humanized mice. In Nature (Nature Publishing Group), pp. 118-122). Briefly, cDNA was synthesized from viral RNA using Superscript III reverse transcriptase (Invitrogen Life Technologies). cDNA was amplified with Expand Long Template PCR System (Roche) with nested PCR. Primers for the first round of PCR were 5'- GGCTTAGGC ATCTCCTATGGC AGGAAGAA-3 ' and 5'-
GGTGTGTAGTTCTGCCAATCAGGGAAGWAGCCTTGTG-3' (SEQ ID Nos: 47 and 48). Primers for the second round of PCR were 5'- T AGAAAG AGC AGAAGAC AGTGGC AATGA-3 ' and 5 ' - TC ATC AATGGTGGTGATGATGATGTTTTTCTCTCTGC ACC ACTCTTCT-3 ' (SEQ
ID Nos: 49 and 50). Gel-purified PCR amplicons were ligated into pCR4-TOPO (Invitrogen) and transformed into One Shot TOP 10 cells. Individual colonies were sequenced using M13F and M13R primers. Sequences were aligned to gpl20yu2 (accession number M93258) and analyzed for mutations using Los Alamos Highlighter tool (hiv.lanl.gov/content/sequence/HIGHLIGHT/ HIGHLIGHT XYPLOT/highlighter.html).
Cell-associated HIV-1 RNA
The cellular fraction of whole blood was resuspended in 400 μΐ PBS and PBMCs were isolated by density gradient centrifugation as described above. Lymphocytes were split into two samples, one for cell-associated HIV-1 RNA measurements, and one for cell-associated HIV-1 DNA measurements. Cell-associated RNA was extracted and
quantified by the same procedures as described above for plasma viral RNA. The lower limit of detection was determined to be 10 copies viral RNA per qRT-PCR reaction. Cell- associated HIV-1 RNA is reported as the ratio of HIV- 1 RNA copies per sample to CCR5 genomic DNA copies per equivalent sample measured in DNA extract. For terminal point measurements, spleen tissue was isolated, homogenized, and filtered through 40 μιη mesh. Splenocytes were used to isolate HIV-1 RNA as described above.
Cell-associated HIV-1 DNA
PBMCs were isolated from whole blood as described above. Splenocytes were isolated from spleen as described above. Total DNA was extracted using QIAmp DNA Blood Mini Kit (Qiagen) and eluted in 80 μΐ volume. Purified DNA was quantified for HIV-1 DNA by qPCR using the primers and probe for HIV-1 RNA quantification mentioned above. Genomic human CCR5 DNA was quantified with primers 5'- GTTGGACC AAGCTATGCAGGT-3 ' (forward, SEQ ID No: 51) and 5'- AGAAGCGTTTGGCAATGTGC-3 ' (reverse, SEQ ID No: 52), and the sequence-specific probe /HEX/5 '-TTGGGATGACGCACTGCTGCATCAACCCCA-3 VZenDQ (SEQ ID No: 53). All qPCR reactions contained 25 μΐ AmpliTaq Gold PCR master mix (Applied Biosystems), in 50 μΐ reaction volume. Reaction mixtures were as previously described (Horwitz et al, Proc Natl Acad Sci U S A, 2013. 110(41): p. 16538-43). HIV-1 DNA is reported as copies per sample to CCR5 genomic copies per equivalent sample.
Terminal Graft
The presence of human lymphocytes at the terminal point was quantified from the spleen and PBMCs by flow cytometry. Isolation of PBMCs and splenocytes were as described above. Staining procedures were as described above.
Antibody Concentrations
Plasma levels of passively administered antibodies were quantified by two independent methods. gpl20-specific ELISA was as previously described (Klein et al, Nature, 2012. 492(7427): p. 118-22), using 10-1074 and 3BNC117 monoclonal antibodies as standard controls. The detection limit was 0.05 μg/ml. Because PG16 does not bind gpl20, and endogenously produced gpl20-reactive antibodies could confound the ELISA measurement, plasma antibody levels were also quantified by TZM-bl neutralization using
the Tier 2 envelopes 3301.vl .c24 and YU2. A mixture with known amounts of 3BNC117, 10-1074, and PG16 was used as standard for calibration.
Day of Viral Rebound and Antibody Level At Rebound
Plasma viremias immediately preceding and following viral rebound were plotted on a semi-log-y-axis versus days post initial antibody injection (x-axis) for each individual mouse. The linear portion of viremia was fit to a line by least-squares linear regression. The day that viremia crossed the 800 copies/ml quantitation limit, termed rebound day, was calculated from the viremia fit. The antibody concentrations (as determined by TZM- bl neutralization) spanning before and after viral rebound were plotted on a semi-log-y- axis versus days post initial antibody injection. The linear portion of antibody concentrations was fit to a line by least-squares linear regression, and the antibody concentration on the rebound day was calculated from the fit.
Anti-retroviral Therapy
Individual tablets of tenofovir disproxil-fumarate (TDF; Gilead Sciences), emtricitabine (FTC; Gilead Sciences), and raltegravir (RAL; Merck) were crushed into fine powder and manufactured with TestDiet 5B1Q feed (Modified LabDiet 5058 with 0.12% amoxicillin) into ½" irradiated pellets. Final concentrations of ART drugs in the food were 720 mg/kg TFV, 520 mg/kg FTC, and 4800 mg/kg RAL. Doses were chosen based on suppression of viremia in humanized mice as previously published (Denton et al, J Virol, 2012. 86(1): p. 630-4 and Nischang et al, PLoS ONE2012. p. e38853), and by pharmacokinetic analysis of these drugs in humanized mice (unpublished, Speck Laboratory). To test potential toxicity, or reduced preference for drug- supplemented food, mice were weighed daily on normal diet, then switched to ART feed and weighed daily. There were no visible signs of toxicity and mice maintained their weights. Assuming mice weigh 25 grams and eat 4 grams of food per day, the drug doses correspond to 2.88 mg/kg TFV, 83 mg/kg FTC, and 768 mg/kg RAL daily.
Antibody Therapy
Plasmids encoding 10-1074 or PG16 heavy- and light- chain Ig genes were transfected into HEK 293E cells. Antibodies were isolated from tissue-culture supernatant using Protein G Sepharose 4 Fast-Flow (GE Healthcare). Antibodies were then buffer- exchanged into PBS and sterile-filtered using Ultrafree-CL centrifugal filters (0.22μιη;
Millipore). Endotoxin was removed from antibody preparations using Triton X-114 (Sigma- Aldrich) as previously described (Aida et al., J Immunol Methods, 1990. 132(2): p. 191-5), and antibodies were concentrated to 10 mg/ml. Sterile, endotoxin- free 3BNC117 (20 mg/ml) was obtained from CellDex Therapeutics. All antibodies were injected subcutaneously as described.
Inducers
Vorinostat (Selleckchem) was suspended in sterile water or sterile water plus 0.5% methylcellulose, 0.1 % Tween (v/v) and administered by oral gavage at doses of 60 mg/kg (Krejsgaard et al., 2010 Experimental dermatology 19, 1096-1102). For each mouse, three total doses were administered, spaced 48 hours apart. 100 μg doses of aCTLA4 were injected intraperitoneally (i.p.). Three total doses were administered, spaced 48 hours apart. I-BET was obtained from Glaxo SmithKline and dissolved in 10% beta-cyclodextrin, 5% DMSO in 0.9%> saline and injected daily for 14 days at doses of 30 mg/kg (Dawson et al, 2011 Nature 478, 529-533.).
Statistical Analysis
Statistical analyses were performed using GraphPad Prism 6.0 for Mac OS X.
Example 2 Post Exposure Prophylaxis with bNAbs
The ART -resistant reservoir is established early in infection as evidenced by postexposure prophylaxis experiments in humans and macaques. Post-exposure prophylaxis with ART or previous-generation bNAbs is only effective when administered within 24 hours of intravenous exposure. (Lifson et al., Journal of Virology 2000. p. 2584-2593; Wade et al., N Engl J Med, 1998. 339(20): p. 1409-14; Tsai, et al, Journal of Virology 1998. p. 4265; Tsai et al, Science 1995. p. 1-3; Landovitz et al, in N EnglJ Med 2009. p. 1-8; Nishimura et al, Proc Natl Acad Sci U S A, 2003. 100(25): p. 15131-6; and Ferrantelli et al., Virology, 2007. 358(1): p. 69-78). To determine if the current generation of more potent bNAbs can abort the establishment of a latent HIV-1 reservoir at later time points, post-exposure prophylaxis experiments were performed in humanized mice (FIG. 1A).
Mice were infected with HIV-lyu2 (150 ng) by intraperitoneal injection, and treated with either ART (raltegravir, emtricitabine, tenofovir) (Denton et al., J Virol, 2012.
86(1): p. 630-4 and Nischang et al, PLoS ONE 2012. p. e38853) or a tri-mix of bNAbs
(3BNC117, 10-1074, and PG16) (Horwitz et al, Proc Natl Acad Sci U S A, 2013. 110(41): p. 16538-43) 4 or 8 days after infection when viremia was already detectable in 51 of 70 mice. Plasma viremia varied from undetectable to 2.70x106 viral RNA copies/ml at 4 days after infection (FIGs. 1B-E and 6). In the absence of therapy, 14 out of 15 mice in the control group developed sustained plasma viremia ranging from 2.48xl03 to 4.19xl06 copies/ml (FIG. IB).
Doses of ART and antibodies were chosen on the basis of their therapeutic efficacy in chronic HIV-1 infection in hu-mice (Klein et al, Nature, 2012. 492(7427): p. 118-22; Horwitz et al, Proc Natl Acad Sci U S A, 2013. 110(41): p. 16538-43; Denton et al, J Virol, 2012. 86(1): p. 630-4; and Nischang et al, PLoS ONE2012. p. e38853). ART was administered in the food for 32-39 days starting 4 days after infection (Denton et al, J Virol, 2012. 86(1): p. 630-4; and Nischang et al, PLoS ONE2012. p. e38853). Antibodies were administered subcutaneous ly with a loading dose of 3 mg per mouse, and 3-5 subsequent doses of 1.5 mg each, spaced 3-4 days apart (FIG. 1A). Consistent with human and macaque studies, 18 of 22 mice treated with ART showed viremia after ART termination, demonstrating that this form of therapy is relatively ineffective at preventing reservoir development in hu-mice when administered 4 days after infection (FIG. 1C). Among the 18 viremic mice, viremia was first detected 28 to 84 days after ART termination (FIGs. 1C and 6). In contrast, 10 of 21 hu-mice treated with antibodies 4 days after infection showed viremia by the terminal point (p =0.027), and for 9 of these 10 viremic mice, the first detectable viremia occurred 74-107 days after the last antibody injection (FIGs. ID and 6). However, bNAb treatment after 8 days was far less effective, resulting in viremia in 10 of the 11 treated mice 44-58 days after the last antibody injection (FIG. IE).
Mice in the early treatment group that failed to show detectable plasma viremia were further examined for the presence of human CD4+ T cells and cell-associated HIV-1 RNA and DNA in the spleen. It was found that mice that failed to develop sustained plasma viremia showed CD4+ T cell levels that were similar to infected controls. Therefore differences in CD4+ T cell levels are unlikely to account for the observed differences between viremic and aviremic mice (FIG. IF). Moreover, T cell-associated HIV-1 RNA levels were consistent with plasma viral loads, with mice that remained
aviremic having either undetectable or lower cell-associated HIV-1 RNA than mice that developed sustained viremia (FIG. 1G).
Cell-associated viral DNA was measured as an imperfect surrogate of the HIV-1 reservoir. HIV-1 DNA is thought to overestimate the reservoir because it fails to exclude damaged or incomplete viral sequences that cannot be reactivated. In addition, the overall number cells assayed in mice is limited and therefore the assay is not very sensitive. Nevertheless HIV-1 DNA measurements were found to be consistent with each mouse's rebound status (FIG. 1H). It was concluded that bNAbs can interfere with the establishment of the latent HIV-1 reservoir in hu-mice.
The above results indicate that bNAbs differ from ART in that they can prevent establishment of the latent HIV-1 reservoir in hu-mice at a time when ART is significantly less effective.
Example 3 Fc Receptor Binding is Required for bNAb Activity
To determine if the efficacy of bNAbs is dependent on the antibodies' ability to engage components of the immune system through their Fc domains, the day 4 postexposure prophylaxis experiments were repeated using the same tri-mix of bNAbs carrying Fc region mutations that abrogate both human and mouse Fc-receptor binding (G236R/L328R; GRLR, herein referred to as FcRnu11) (Horton et al, 2010, Blood 116, 3004-3012). Despite equivalent neutralizing activity in TZM-bl assays (Pietzsch et al., Proc Natl Acad Sci U S A, 2012. 109(39), FcRnu11 antibodies were far less potent than controls in vivo (FIGs. 2 and 7). Mice treated with FcRnu11 tri-mix initially suppressed viremia at the same rate as the wild type antibody-treated mice (FIG. 2A). However 9 of 15 mice receiving post exposure prophylaxis with the FcRnu11 tri-mix showed viral rebound by 44 days after the last antibody injection. In contrast, 44 days after the last injection of control antibodies, only 1 of 21 mice showed rebound viremia (p = 0.0004). The loss of in vivo activity of the FcRnu11 tri-mix was not due to lower antibody levels in the FcRnu11 injected mice. In fact, antibody levels at the time of viral rebound were ~50-fold higher for mice receiving FcRnu11 tri-mix compared to wild-type tri-mix (p = 0.0035, FIG. 2C). It was concluded that effective post-exposure prophylaxis by bNAbs requires engagement of Fc- receptors.
The escape variants to the individual bNAbs in the tri-mix used in these experiments have been documented extensively (Horwitz et al, Proc Natl Acad Sci U S A, 2013. 110(41): p. 16538-43, and Klein et al, Nature, 2012. 492(7427): p. 118-22). However, inventors have never observed HIV-1 escape by mutation to the bNAb tri-mix. Rather viral rebound is usually due to a drop in antibody concentrations to sub-therapeutic levels (Horwitz et al, Proc Natl Acad Sci U S A, 2013. 110(41): p. 16538-43, and Klein et al, Nature, 2012. 492(7427): p. 118-22). Because mice receiving FcRnu11 tri-mix showed viral rebound in the presence of antibody concentrations far higher than the therapeutic threshold for wild-type antibodies (FIG. 2C), gpl20 from 9 of the rebounding mice were cloned and sequenced to examine the mechanism for viral breakthrough in the presence of FcRnu11 tri-mix (FIG. 2D). Among all 40 clones sequenced, not a single clone had the triple combination of signature mutations that confer escape to the antibody-tri-mix. It was concluded that viral rebound in FcRnu11 tri-mix treated mice is not attributable to antibody escape, but rather reduced antibody potency. Thus, FcRnu11 mutant antibodies, which cannot engage Fc receptors, are less active in suppressing infection than their wild type counterparts.
These results indicate that effective post-exposure prophylaxis by bNAbs requires engagement of Fc-receptors.
Example 4 Combination Therapy with bNAbs and Inducers
A small number (-15%) of chronically infected hu-mice and macaques treated with antibodies fail to show rebound viremia after therapy is discontinued (Barouch et al, Nature, 2013. 503(7475): p. 224-8; Horwitz et al, Proc Natl Acad Sci U S A, 2013. 110(41): p. 16538-43; Klein et al, Nature 2012, p. 118-122 ; and Shingai et al, Nature, 2013. 503(7475): p. 277-80). This suggests that antibodies may be able to decrease the size of the reservoir, or interfere with its maintenance, in established infections. To determine whether agents that induce viral transcription from latently infected cells can enhance this effect antibody therapy was combined with viral inducers (FIGs. 3A and 8).
Hu-mice with established HIV-lyu2 infections (viremia ranging from 4.70x10 - 7.96xl05 copies/ml at 2-3 weeks after infection, FIGs. 3B-E) were treated with tri-mix bNAbs. When plasma viremia dropped below detection, they were co-administered a viral inducer for 5-14 days, and monitored for viral rebound for an additional 47-85 days. The
inducers tested were vorinostat, an HDAC inhibitor (Archin et al, AIDS Res Hum Retroviruses, 2009. 25(2): p. 207-12; Archin et al, AIDS, 2009. 23(14): p. 1799-806; and Contreras et al, J Biol Chem, 2009. 284(11): p. 6782-9), I-BET151, a BET protein inhibitor (Boehm et al, Cell Cycle, 2013. 12(3): p. 452-62), and aCTLA4, a T-cell inhibitory pathway blocker (Alegre et al, Nat. Rev. Immunol 2001. p. 220-228, and Krummel et al, J. Exp. Med. 1995. p. 459-465). They were selected because of their documented abilities to induce HIV-1 transcription in vitro, as well as their safety and established pharmacokinetic properties in mice (Krejsgaard et al, Exp Dermatol, 2010. 19(12): p. 1096-102 ; Kwon et al, Proc Natl Acad Sci U S A, 1997. 94(15): p. 8099-103; and Nicodeme et al, Nature, 2010. 468(7327): p. 1119-23).
Hu-mice receiving antibodies plus vorinostat showed no significant differences in viral rebound compared to hu-mice receiving antibody alone (FIGs. 3B, 3C and 8). The same result was seen for hu-mice treated with antibodies plus I-BET151 or aCTLA4 (FIGs. 3D, 3E and 8). All 10 mice that received antibody therapy plus vorinostat showed viral rebound when the antibody dropped below therapeutic levels. Of 12 mice that received antibody therapy plus I-BET151, 11 had viral rebound, and 10 of 11 mice that received antibody plus aCTLA4 showed viral rebound. In total, of 33 mice that received antibody plus a single inducer, 31 showed viral rebound. In comparison, of 25 mice that received antibody therapy alone, 22 rebounded after the level of passively administered antibody decayed below the therapeutic threshold (p =0.64).
Of the three inducers tested, vorinostat is the only one that has also been studied in HIV-1 infected humans. Treatment with vorinostat plus ART resulted in a transient increase in resting CD4+ T cell-associated HIV-1 RNA, but no change in plasma viremia, or the frequency of replication-competent HIV-1 within resting CD4+ T cells (Archin et al, Nature, 2012. 487(7408): p. 482-5 and Archin et al, J. Infect. Dis. 2014. p. 1-26). The above results in hu-mice are consistent with these findings, and extend them to additional candidate inducers, demonstrating that administration of a single inducer has no significant effect on the ability of the latent reservoir to produce rebound viremia.
Example 5 Combination Therapy with Multiple Inducers
In this example, assays were carried out to determine whether a combination of inducers might be more effective than a single inducer. More specifically, all three inducers mentioned above were administered simultaneously.
It was found that, in the absence of antibody therapy, the combination of all three inducers was not measurably toxic and did not abort or noticeably alter active infection (FIG. 6). 23 mice that initially suppressed viremia on antibody therapy were treated with the inducer combination and followed for 62-105 days after the last antibody injection (FIG. 4A). Only 10 of the 23 showed viral rebound, and the remaining 57% of the mice failed to rebound, a significant decrease in rebound frequency compared to antibody alone (p = 0.0018), or antibody plus single inducers (p = 0.0001) (FIG. 4B).
Importantly, when compared to antibody alone, neither single inducer nor combination inducers measurably altered the frequency of CD4+ T cells remaining at the end of the experiment (FIG. 5A). Additionally, spleen T-cell associated viral RNA reflected plasma viral RNA levels at the time the experiment was terminated in that it was largely undetectable in mice that failed to rebound (FIGs. 5B).
Finally, when compared to controls, hu-mice that failed to rebound after combination antibody and inducer therapy showed similar initial plasma viremias to mice that rebounded across all experimental groups (FIG. 5C). Therefore, neither initial viremia levels, nor CD4+ T cell levels can account for the differences between the experimental groups.
To determine if antibody persistence or premature termination accounted for differing viral rebound outcomes, antibody levels at the time of rebound and at the terminal point were calculated. The average plasma antibody concentration at the time of viral rebound in the 59 rebounding mice was 2.97 μg/ml (FIG. 5D). Since the antibody concentrations decayed to 2.97 μg/ml at different rates in individual mice, the number of days that elapsed from when each individual mouse's antibody levels reached 2.97 μg/ml to when the mouse showed rebound viremia were calculated. 50 of 59 mice rebounded within 10 days (FIG. 5E). Of the 18 non-rebounding mice, the average antibody concentration at the terminal point was 0.44 μg/ml, with 15 out of 18 mice having antibody concentrations less than 2.97 μg/ml. Furthermore, in non-rebounding mice, an
average of 20.2 days elapsed from the time antibody concentrations reached 2.97 μg/ml to termination (FIG. 5F). Thus, failure to rebound cannot be explained by antibody persistence or premature termination.
Finally, inventors could not detect viral DNA at the terminal point in the majority of mice that did not rebound, whereas the majority of mice that did rebound had detectable HIV-1 DNA, with an average of 0.09 copies per T cell (FIG. 5G). This suggests that combining vorinostat, I-BET151 and aCTLA4 with immunotherapy decreases rebound viremia in hu-mice.
In the above example, hu-mice resembled infected humans in that they contain human cells that are infected with authentic HIV-1 (Hatziioannou et al, Nat Rev Microbiol, 2012. 10(12): p. 852-67 and Brehm et al, J Infect Dis, 2013. 208 Suppl 2: p. S 125-30). In addition, the kinetics of viral rebound in hu-mice after suppression of viremia with ART resembles infected humans (Horwitz et al, Proc Natl Acad Sci U S A, 2013. 110(41): p. 16538-43 and Nischang et al, PLoS ONE2012. p. e38853). The macaque model is valuable because it represents an immunologically intact host that may also harbor reservoirs not found in the mice. However, the infection in macaques involves non-human primate cells and SHIV or SIV, both of which differ significantly from HIV-1. While neither of the two model systems is entirely faithful, they have produced very similar results in both therapy and prevention experiments to date (West et al, Cell, 2014. 156(4): p. 633-48).
One of the strategies proposed to eliminate latent viruses involves inducing their expression under the cover of ART. In theory, this would kill infected cells while preventing the spread of infection (Deeks, S.G., Nature, 2012. 487(7408): p. 439-40). In vitro experiments indicate that silent proviruses can in fact be induced to become active (Ho et al, Cell, 2013. 155(3): p. 540-51 and Bullen et al, Nature, 2014, p. 1-6), but neither infected neither humans nor hu-mice show a measurable change in the reservoir after therapy with a single inducer and ART (Archin et al, J. Infect. Dis.2014. p. 1-26) or bNAbs. The results here establish the principle that only a combination of inducers and bNAbs can have a significant impact on the viral reservoir in vivo.
The foregoing examples and description of the preferred embodiments should be taken as illustrating, rather than as limiting the present invention as defined by the claims.
As will be readily appreciated, numerous variations and combinations of the features set forth above can be utilized without departing from the present invention as set forth in the claims. Such variations are not regarded as a departure from the scope of the invention, and all such variations are intended to be included within the scope of the following claims. All references cited herein are incorporated by reference in their entireties.
Claims
1. A method for decreasing the size of or preventing the establishment of a latent reservoir of HIV infected cells in a subject in need thereof, comprising
administering to said subject a therapeutically effective amount of an isolated anti-
HIV antibody, and
administering to said subject two or more viral transcription inducers in effective amounts to induce transcription of an HIV provirus in said cells.
2. The method of claim 1, wherein the antibody is a human antibody, a humanized antibody, or a chimeric antibody.
3. The method of claim 1, wherein the antibody is antibody 3BNC117, 10- 1074, or PG16.
4. The method of claim 3, wherein two or more of said antibodies 3BNC117, 10-1074, and PG16 are administered to said subject.
5. The method of claim 1, wherein one, two, or more of antibodies listed in Table 1 are administered to said subject.
6. The method of any one of claims 1-5, wherein the transcription inducers are selected from the group consisting of vorinostat, an HDAC inhibitor, I-BET151, a BET bromodomain inhibitor, aCTLA4, and a T-cell inhibitory pathway blocker.
7. The method of any one of claims 1-6, wherein said cells comprise CD4+ T cells.
8. The method of any one of claims 1-7, wherein the antibody or antibodies are administered to the subject within about 96 hours after exposure to HIV.
9. The method of any one of claims 1-8, further comprising administering to said subject an antiviral agent.
10. The method of claim 8, wherein the antiviral agent is selected from the group consisting of a non-nucleoside reverse transcriptase inhibitor, a protease inhibitor, an entry or fusion inhibitor, and an integrase inhibitor.
11. A kit comprising an isolated anti-HIV antibody, a first viral transcription inducer, and a second viral transcription inducer.
12. The kit of claim 11, wherein the antibody is antibody 3BNC117, 10-1074, or PG16.
13. The kit of claim 11-12, wherein kit comprises two or more of said antibodies 3BNC117, 10-1074, and PG16.
14. The kit of claim 11-13, wherein the antibody is a human antibody, a humanized antibody, or a chimeric antibody.
15. The kit of claim 11-14, wherein the transcription inducers are selected from the group consisting of vorinostat, an HDAC inhibitor, I-BET151, a BET bromodomain inhibitor, aCTLA4, and a T-cell inhibitory pathway blocker.
16. The kit of any one of claims 11-15, wherein the first viral transcription inducer and the second viral transcription inducer are in one pharmaceutical composition or in two separate pharmaceutical compositions.
17. The kit of any one of claims 11-16, further comprising an antiviral agent.
18. The kit of claim 17, wherein the antiviral agent is one selected from the group consisting of a non-nucleoside reverse transcriptase inhibitor, a protease inhibitor, an entry or fusion inhibitor, and an integrase inhibitor.
19. A method for preventing the establishment of a latent reservoir of HIV infected cells in a subject in need thereof, comprising administering to said subject a therapeutically effective amount of an isolated anti-HIV antibody.
20. The method of claim 19, wherein the antibody is antibody 3BNC117, 10- 1074, or PG16.
21. The method of claim 19 or 20, wherein the antibody is a human antibody, a humanized antibody, or a chimeric antibody.
22. The method of claim 20, wherein two or more of said antibodies 3BNC117,
10-1074, and PG16 are administered to said subject.
23. The method of any one of claims 19-22, wherein the antibody or antibodies are administered to the subject within about 96 hours after exposure to HIV.
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US15/327,725 US10676521B2 (en) | 2014-07-21 | 2015-07-21 | Combination of broadly neutralizing HIV antibodies and viral inducers |
| US16/885,797 US20200299365A1 (en) | 2014-07-21 | 2020-05-28 | Combination of broadly neutralizing hiv antibodies and viral inducers |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201462026915P | 2014-07-21 | 2014-07-21 | |
| US62/026,915 | 2014-07-21 |
Related Child Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US15/327,725 A-371-Of-International US10676521B2 (en) | 2014-07-21 | 2015-07-21 | Combination of broadly neutralizing HIV antibodies and viral inducers |
| US16/885,797 Division US20200299365A1 (en) | 2014-07-21 | 2020-05-28 | Combination of broadly neutralizing hiv antibodies and viral inducers |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2016014484A1 true WO2016014484A1 (en) | 2016-01-28 |
Family
ID=55163619
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2015/041272 WO2016014484A1 (en) | 2014-07-21 | 2015-07-21 | Combination of broadly neutralizing hiv antibodies and viral inducers |
Country Status (2)
| Country | Link |
|---|---|
| US (2) | US10676521B2 (en) |
| WO (1) | WO2016014484A1 (en) |
Cited By (26)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2015048770A2 (en) | 2013-09-30 | 2015-04-02 | Beth Israel Deaconess Medical Center, Inc. | Antibody therapies for human immunodeficiency virus (hiv) |
| JP2019535011A (en) * | 2016-09-30 | 2019-12-05 | セントレ ナショナル デ ラ レセルシュ シャンティフィク | Cell marker |
| WO2020010107A1 (en) | 2018-07-03 | 2020-01-09 | Gilead Sciences, Inc. | Antibodies that target hiv gp120 and methods of use |
| WO2020263830A1 (en) | 2019-06-25 | 2020-12-30 | Gilead Sciences, Inc. | Flt3l-fc fusion proteins and methods of use |
| WO2021011891A1 (en) | 2019-07-18 | 2021-01-21 | Gilead Sciences, Inc. | Long-acting formulations of tenofovir alafenamide |
| WO2021011544A1 (en) | 2019-07-16 | 2021-01-21 | Gilead Sciences, Inc. | Hiv vaccines and methods of making and using |
| WO2021130638A1 (en) | 2019-12-24 | 2021-07-01 | Carna Biosciences, Inc. | Diacylglycerol kinase modulating compounds |
| WO2021188959A1 (en) | 2020-03-20 | 2021-09-23 | Gilead Sciences, Inc. | Prodrugs of 4'-c-substituted-2-halo-2'-deoxyadenosine nucleosides and methods of making and using the same |
| US11149082B2 (en) | 2016-10-17 | 2021-10-19 | University Of Maryland, College Park | Multispecific antibodies targeting human immunodeficiency virus and methods of using the same |
| WO2021236944A1 (en) | 2020-05-21 | 2021-11-25 | Gilead Sciences, Inc. | Pharmaceutical compositions comprising bictegravir |
| JP2022500042A (en) * | 2018-09-14 | 2022-01-04 | ザ ロックフェラー ユニバーシティー | Anti-HIV antibody 10-1074 variant |
| WO2022031894A1 (en) | 2020-08-07 | 2022-02-10 | Gilead Sciences, Inc. | Prodrugs of phosphonamide nucleotide analogues and their pharmaceutical use |
| WO2022046644A1 (en) | 2020-08-25 | 2022-03-03 | Gilead Sciences, Inc. | Multi-specific antigen binding molecules targeting hiv and methods of use |
| WO2022271677A1 (en) | 2021-06-23 | 2022-12-29 | Gilead Sciences, Inc. | Diacylglyercol kinase modulating compounds |
| WO2022271650A1 (en) | 2021-06-23 | 2022-12-29 | Gilead Sciences, Inc. | Diacylglyercol kinase modulating compounds |
| WO2022271659A1 (en) | 2021-06-23 | 2022-12-29 | Gilead Sciences, Inc. | Diacylglyercol kinase modulating compounds |
| WO2022271684A1 (en) | 2021-06-23 | 2022-12-29 | Gilead Sciences, Inc. | Diacylglyercol kinase modulating compounds |
| WO2023102239A1 (en) | 2021-12-03 | 2023-06-08 | Gilead Sciences, Inc. | Therapeutic compounds for hiv virus infection |
| WO2023102523A1 (en) | 2021-12-03 | 2023-06-08 | Gilead Sciences, Inc. | Therapeutic compounds for hiv virus infection |
| WO2023102529A1 (en) | 2021-12-03 | 2023-06-08 | Gilead Sciences, Inc. | Therapeutic compounds for hiv virus infection |
| WO2023196875A1 (en) | 2022-04-06 | 2023-10-12 | Gilead Sciences, Inc. | Bridged tricyclic carbamoylpyridone compounds and uses thereof |
| US11845788B2 (en) | 2018-05-22 | 2023-12-19 | Beth Israel Deaconess Medical Center, Inc. | Antibody therapies for human immunodeficiency virus (HIV) |
| WO2024006982A1 (en) | 2022-07-01 | 2024-01-04 | Gilead Sciences, Inc. | Therapeutic compounds useful for the prophylactic or therapeutic treatment of an hiv virus infection |
| WO2024015741A1 (en) | 2022-07-12 | 2024-01-18 | Gilead Sciences, Inc. | Hiv immunogenic polypeptides and vaccines and uses thereof |
| WO2024044477A1 (en) * | 2022-08-26 | 2024-02-29 | Gilead Sciences, Inc. | Dosing and scheduling regimen for broadly neutralizing antibodies |
| WO2024076915A1 (en) | 2022-10-04 | 2024-04-11 | Gilead Sciences, Inc. | 4'-thionucleoside analogues and their pharmaceutical use |
Families Citing this family (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| MX358099B (en) * | 2011-05-17 | 2018-08-06 | Univ Rockefeller | Human immunodeficiency virus neutralizing antibodies adn methods of use thereof. |
| UY38720A (en) * | 2019-05-31 | 2020-12-31 | Viracta Therapeutics Inc | METHODS FOR TREATING VIRUS-ASSOCIATED CANCERS WITH HISTONE DEACETILASE INHIBITORS |
| EP3976198A4 (en) | 2019-05-31 | 2023-07-19 | Viracta Subsidiary, Inc. | Methods of treating virally associated cancers with histone deacetylase inhibitors |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20130189364A1 (en) * | 2004-07-09 | 2013-07-25 | Robert Sabin | Compositions and methods of potentiating adjuvant pharmaceuticals targeting latent viral infections |
| US20140017234A1 (en) * | 2012-06-21 | 2014-01-16 | California Institute Of Technology | Antibodies targeting hiv escape mutants |
| US20140199260A1 (en) * | 2011-07-29 | 2014-07-17 | The Children's Hospital Of Philadelphia | Compositions and methods for the treatment of hiv |
Family Cites Families (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5855887A (en) * | 1995-07-25 | 1999-01-05 | The Regents Of The University Of California | Blockade of lymphocyte down-regulation associated with CTLA-4 signaling |
| MX358099B (en) * | 2011-05-17 | 2018-08-06 | Univ Rockefeller | Human immunodeficiency virus neutralizing antibodies adn methods of use thereof. |
-
2015
- 2015-07-21 US US15/327,725 patent/US10676521B2/en active Active
- 2015-07-21 WO PCT/US2015/041272 patent/WO2016014484A1/en active Application Filing
-
2020
- 2020-05-28 US US16/885,797 patent/US20200299365A1/en active Pending
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20130189364A1 (en) * | 2004-07-09 | 2013-07-25 | Robert Sabin | Compositions and methods of potentiating adjuvant pharmaceuticals targeting latent viral infections |
| US20140199260A1 (en) * | 2011-07-29 | 2014-07-17 | The Children's Hospital Of Philadelphia | Compositions and methods for the treatment of hiv |
| US20140017234A1 (en) * | 2012-06-21 | 2014-01-16 | California Institute Of Technology | Antibodies targeting hiv escape mutants |
Non-Patent Citations (6)
| Title |
|---|
| CILLO ET AL.: "Quantification of HIV-1 latency reversal in resting CD 4+ T cells from patients on suppressive antiretroviral therapy.", PROC NATL ACAD SCI USA., vol. 111, no. 19, May 2014 (2014-05-01), pages 7078 - 83 * |
| HALPER-STROMBERG ET AL.: "Broadly neutralizing antibodies and viral inducers decrease rebound from HIV-1 latent reservoirs in humanized mice.", CELL, vol. 158, no. 5, August 2014 (2014-08-01), pages 989 - 99 * |
| HORWITZ ET AL.: "HIV-1 suppression and durable control by combining single broadly neutralizing antibodies and antiretroviral drugs in humanized mice.", PROC NATL ACAD SCI U S A, vol. 110, no. 41, 2013, pages 16538 - 43. * |
| LI ET AL.: "The BET bromodomain inhibitor JQ1 activates HIV latency through antagonizing Brd4 inhibition of Tat-transactivation.", NUCLEIC ACIDS RES., vol. 41, no. 1, 18 October 2012 (2012-10-18), pages 277 - 87, [retrieved on 20130000] * |
| PEJCHAL ET AL.: "Structure and function of broadly reactive antibody PG16 reveal an H3 subdomain that mediates potent neutralization of HIV-1.", PROC NATL ACAD SCI USA., vol. 107, no. 25, 2010, pages 11483 - 8, XP055113318, DOI: doi:10.1073/pnas.1004600107 * |
| SCRIPPS_NEWS_RELEASE: "Two New Antibodies Found to Cripple HIV", NEWS RELEASE, 3 September 2009 (2009-09-03), pages 1 - 6, Retrieved from the Internet <URL:https://www.scripps.edu/news/press/2009/090309b.html> * |
Cited By (34)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP3052132A4 (en) * | 2013-09-30 | 2017-05-17 | Beth Israel Deaconess Medical Center, Inc. | Antibody therapies for human immunodeficiency virus (hiv) |
| US10376583B2 (en) | 2013-09-30 | 2019-08-13 | Beth Israel Deaconess Medical Center, Inc. | Human immunodeficiency virus therapies utilizing N332-glycan-dependent antibodies and a reservoir activator |
| EP3052132B1 (en) | 2013-09-30 | 2020-07-29 | Beth Israel Deaconess Medical Center, Inc. | Antibody therapies for human immunodeficiency virus (hiv) |
| WO2015048770A2 (en) | 2013-09-30 | 2015-04-02 | Beth Israel Deaconess Medical Center, Inc. | Antibody therapies for human immunodeficiency virus (hiv) |
| US11202830B2 (en) | 2013-09-30 | 2021-12-21 | Beth Israel Deaconess Medical Center, Inc. | Human immunodeficiency virus therapies utilizing N332-glycan-dependent antibodies in subjects with low viral loads |
| JP2019535011A (en) * | 2016-09-30 | 2019-12-05 | セントレ ナショナル デ ラ レセルシュ シャンティフィク | Cell marker |
| JP7083819B2 (en) | 2016-09-30 | 2022-06-13 | セントレ ナショナル デ ラ レセルシュ シャンティフィク | Cell marker |
| US11149082B2 (en) | 2016-10-17 | 2021-10-19 | University Of Maryland, College Park | Multispecific antibodies targeting human immunodeficiency virus and methods of using the same |
| US11845788B2 (en) | 2018-05-22 | 2023-12-19 | Beth Israel Deaconess Medical Center, Inc. | Antibody therapies for human immunodeficiency virus (HIV) |
| WO2020010107A1 (en) | 2018-07-03 | 2020-01-09 | Gilead Sciences, Inc. | Antibodies that target hiv gp120 and methods of use |
| EP4257600A2 (en) | 2018-07-03 | 2023-10-11 | Gilead Sciences, Inc. | Antibodies that target hiv gp120 and methods of use |
| JP2022500042A (en) * | 2018-09-14 | 2022-01-04 | ザ ロックフェラー ユニバーシティー | Anti-HIV antibody 10-1074 variant |
| EP3849612A4 (en) * | 2018-09-14 | 2022-07-06 | The Rockefeller University | Anti-hiv antibody 10-1074 variants |
| WO2020263830A1 (en) | 2019-06-25 | 2020-12-30 | Gilead Sciences, Inc. | Flt3l-fc fusion proteins and methods of use |
| WO2021011544A1 (en) | 2019-07-16 | 2021-01-21 | Gilead Sciences, Inc. | Hiv vaccines and methods of making and using |
| WO2021011891A1 (en) | 2019-07-18 | 2021-01-21 | Gilead Sciences, Inc. | Long-acting formulations of tenofovir alafenamide |
| WO2021130638A1 (en) | 2019-12-24 | 2021-07-01 | Carna Biosciences, Inc. | Diacylglycerol kinase modulating compounds |
| WO2021188959A1 (en) | 2020-03-20 | 2021-09-23 | Gilead Sciences, Inc. | Prodrugs of 4'-c-substituted-2-halo-2'-deoxyadenosine nucleosides and methods of making and using the same |
| WO2021236944A1 (en) | 2020-05-21 | 2021-11-25 | Gilead Sciences, Inc. | Pharmaceutical compositions comprising bictegravir |
| WO2022031894A1 (en) | 2020-08-07 | 2022-02-10 | Gilead Sciences, Inc. | Prodrugs of phosphonamide nucleotide analogues and their pharmaceutical use |
| WO2022046644A1 (en) | 2020-08-25 | 2022-03-03 | Gilead Sciences, Inc. | Multi-specific antigen binding molecules targeting hiv and methods of use |
| WO2022271677A1 (en) | 2021-06-23 | 2022-12-29 | Gilead Sciences, Inc. | Diacylglyercol kinase modulating compounds |
| WO2022271684A1 (en) | 2021-06-23 | 2022-12-29 | Gilead Sciences, Inc. | Diacylglyercol kinase modulating compounds |
| WO2022271650A1 (en) | 2021-06-23 | 2022-12-29 | Gilead Sciences, Inc. | Diacylglyercol kinase modulating compounds |
| WO2022271659A1 (en) | 2021-06-23 | 2022-12-29 | Gilead Sciences, Inc. | Diacylglyercol kinase modulating compounds |
| WO2023102239A1 (en) | 2021-12-03 | 2023-06-08 | Gilead Sciences, Inc. | Therapeutic compounds for hiv virus infection |
| WO2023102523A1 (en) | 2021-12-03 | 2023-06-08 | Gilead Sciences, Inc. | Therapeutic compounds for hiv virus infection |
| WO2023102529A1 (en) | 2021-12-03 | 2023-06-08 | Gilead Sciences, Inc. | Therapeutic compounds for hiv virus infection |
| WO2023196875A1 (en) | 2022-04-06 | 2023-10-12 | Gilead Sciences, Inc. | Bridged tricyclic carbamoylpyridone compounds and uses thereof |
| EP4310087A1 (en) | 2022-04-06 | 2024-01-24 | Gilead Sciences, Inc. | Bridged tricyclic carbamoylpyridone compounds and uses thereof |
| WO2024006982A1 (en) | 2022-07-01 | 2024-01-04 | Gilead Sciences, Inc. | Therapeutic compounds useful for the prophylactic or therapeutic treatment of an hiv virus infection |
| WO2024015741A1 (en) | 2022-07-12 | 2024-01-18 | Gilead Sciences, Inc. | Hiv immunogenic polypeptides and vaccines and uses thereof |
| WO2024044477A1 (en) * | 2022-08-26 | 2024-02-29 | Gilead Sciences, Inc. | Dosing and scheduling regimen for broadly neutralizing antibodies |
| WO2024076915A1 (en) | 2022-10-04 | 2024-04-11 | Gilead Sciences, Inc. | 4'-thionucleoside analogues and their pharmaceutical use |
Also Published As
| Publication number | Publication date |
|---|---|
| US20170210786A1 (en) | 2017-07-27 |
| US10676521B2 (en) | 2020-06-09 |
| US20200299365A1 (en) | 2020-09-24 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US20200299365A1 (en) | Combination of broadly neutralizing hiv antibodies and viral inducers | |
| Parsons et al. | Fc-dependent functions are redundant to efficacy of anti-HIV antibody PGT121 in macaques | |
| US11202830B2 (en) | Human immunodeficiency virus therapies utilizing N332-glycan-dependent antibodies in subjects with low viral loads | |
| TWI735491B (en) | Human immunodeficiency virus neutralizing antibodies | |
| Halper-Stromberg et al. | Towards HIV-1 remission: potential roles for broadly neutralizing antibodies | |
| ES2789348T3 (en) | Neutralizing antibodies to GP120 and their uses | |
| US20180118816A1 (en) | Multi-valent human immunodeficiency virus antigen binding molecules and uses thereof | |
| CN110831965A (en) | Multispecific antibodies targeting HIV GP120 and CD3 | |
| US11292839B2 (en) | Treatment and sustained virologic remission of HIV infection by antibodies to CD4 in HAART stabilized patients | |
| JP2020525505A (en) | Use of anti-FAM19A5 antibody for cancer treatment | |
| KR20210060548A (en) | Anti-HIV antibody 10-1074 variant | |
| US20180028658A1 (en) | Antibody-drug conjugates for reducing the latent hiv reservoir | |
| JP7355852B2 (en) | Method of identifying HIV patients susceptible to therapy with GP120 V3 glycan-directed antibodies | |
| US20220144923A1 (en) | METHODS OF IDENTIFYING HIV PATIENTS SENSITIVE TO THERAPY WITH gp120 CD4 BINDING SITE-DIRECTED ANTIBODIES | |
| JP2017529355A (en) | Treatment and functional healing of HIV infection with monoclonal antibodies against CD4 that mediate competitive HIV entry inhibition | |
| WO2022228827A1 (en) | Human neutralizing monoclonal antibodies against sars-cov-2 and uses thereof | |
| Schriek et al. | Next-generation bNAbs for HIV-1 cure strategies |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 15824595 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 15327725 Country of ref document: US |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 15824595 Country of ref document: EP Kind code of ref document: A1 |