PROCESS OF PRODUCTION OF 1-(5.5-DIMETHYLCYCLOHEX-1-EN-1- YUETHANONE AND 1-(5,5-DIMETHYLCYCLOHEX-6-EN-1-YL)ETHANONE
The present invention relates to an improved method for producing 1 -(5,5- dimethylcyclohex-1 -en-1 -yl)ethanone and 1 -(5,5-dimethylcyclohex-6-en-1 - yl)ethanone.
1 -(5,5-dimethylcyclohex-1 -en-1 -yl)ethanone (compound of formula (Γ)) and 1 - (5,5-dimethylcyclohex-6-en-1 -yl)ethanone (compound of formula (I"))
are versatile and important intermediates for the synthesis of various known fragrance compounds.
In the context of the present patent application the compounds of formula (Γ) and of formula (I") are summarized by the following formula (I)
Compounds of formula (I) (which means either the compound of formula (Γ) or the compound of formula (I") as such as well as any mixture of these two
compounds) can be used to produce for example the following compounds of formulae (la) to (Ig):
All these reactions to obtain the compounds of formula (la) to (Ig) from the compound of formula (I) are well known from the prior art.
The compounds of formula (I) are usually produced by using formic acid (HCOOH) as a catalyst in the rearrangement of compounds having formula (II)
This reaction is described for example in US4147672.
This method of production by using strong Bronsted acids under corrosive reaction conditions does cause some problems.
For example it is necessary to use expensive acid-resistant equipment for the production. Furthermore the handling of corrosive compounds is not easy for the production team.
Due to the importance of the compounds of formula (I), there is always a need for finding an improved method of production of the compounds of formula (I).
Surprisingly it was found that it possible to produce the compounds of formula (I) by a gold catalyzed Rupe-Kambli rearrangement.
Therefore the present invention relates to a process (A) of production of the compounds of formula (I)
by a rearrangement reaction of the compound of formula (II)
wherein the rearrangement reaction is catalyzed in the presence of at least one Au(l)-complex.
A further problem, which occurs when synthesizing the compounds of formula (I) is that there are competitive reactions occurring, such as the Meyer Schuster rearrangement :
By the use of the gold catalyst the above mentioned side reaction (which does not lead to the compounds of formula (I)) is (more or less) suppressed, which means that the yield and conversion of the new process according to the present invention are excellent.
Especially the following Au(l)-complexes are used as catalysts for the process according to the present invention:
Y Au(l) Z (III)
wherein
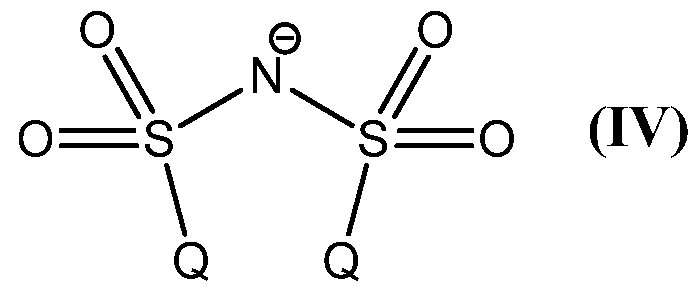
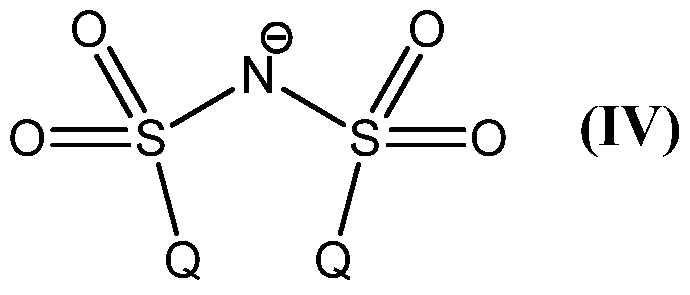
Z is an anion, which is selected from the group consisting of [BX ]", [ΡΧΘ]",
[SbF6]", [CIO4]", CF3COO", sulfonates, tetra(3,5-bis(trifluoromethyl)phenyl)borate
(BArF ), tetraphenylborate, and the anion of formula (IV);
wherein Q represents a phenyl or a Ci-s-alkyl which preferably is substituted by at least one substituent chosen from the group consisting of F, CI, NO2 and
X is a halogen atom, especially F and CI, and
Y is an organic ligand.
Therefore the present invention also relates to process (B), which is the process (A), wherein at least one Au(l) complex of the following compound of formula (III)
Y Au(l) Z (III) wherein
Z is an anion, which is selected from the group consisting of [BX ]", [ΡΧβ]",
[SbF6]", [CIO4]", CF3COO", sulfonates, tetra(3,5-bis(trifluoromethyl)phenyl)borate
(BArF ), tetraphenylborate and the anion of formula (IV)
wherein Q represents a phenyl or a Ci-s-alkyl group which preferably is substituted by at least one halogen atom, and
X is a halogen atom, especially F and CI, and
Y is an organic ligand,
is used.
Preferably Y is an organic ligand selected from the group consisting of following
Therefore the present invention also relates to process (C), which is the process (A) or (B), wherein the organic ligand Y of Au(l) complex of formula (III) is selected from the group consisting of following ligands (Y1) to (Y10):
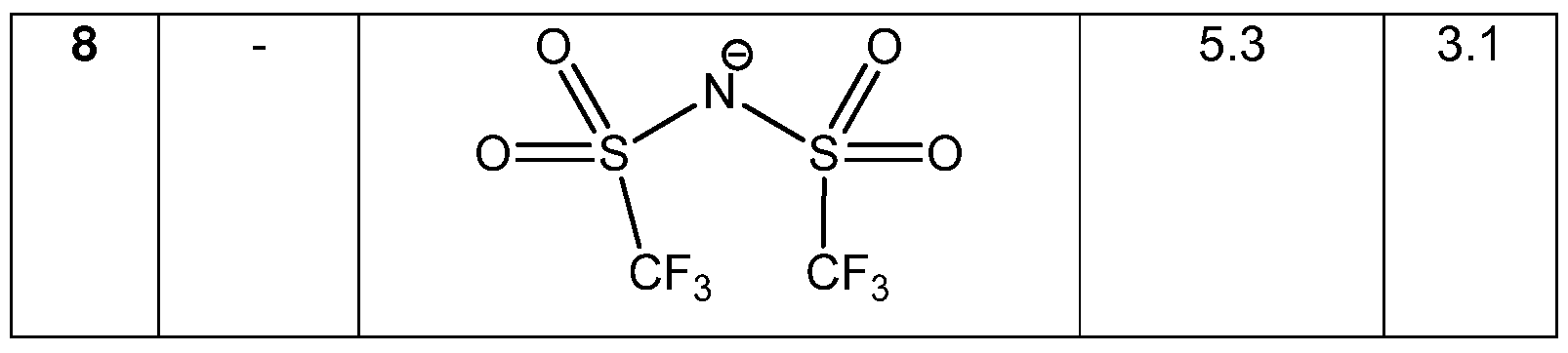
Preferably Z is an anion selected from the group consisting of following anions [BF4]", [PF6]", [SbF6]", [CIO4]~, CF3COO", sulfonates (such as triflate CF3SO3"), tetra(3,5-bis(trifluoromethyl)phenyl)borate (BArF ), tetraphenylborate, and the anion of formula (IV)
Therefore the present invention also relates to process (D), which is the process (A), (B) or (C), wherein the anion Z is selected from the group consisting of following anions [BF4]", [PF6]", [SbF6]", [CIO4]~, CF3COO", sulfonates (such as
triflate CF3SO3 "), tetra(3,5-bis(trifluoromethyl)phenyl)borate (BArF ), tetra henylborate, and the anion of formula (IV)
The catalyst (the Au(l) complex) can be added to the reaction mixture in the form, which is defined by formula (I), but it is also possible that the Au(l)-complex is formed in situ in the reaction mixture (before the starting material is added or after the starting material is added).
For example it is possible to add the organic ligand in the form of a salt (such as for example a chloride: Y-Au(l)CI) and the anion in form of a metal salt (such for example a silver salt Ag(l)Z). The Au(l) complex is then formed in situ and the resulting metal salt (for example AgCI) does not interfere negatively.
Therefore the present invention relates to a process (E), which is the process (A), (B), (C) or (D), wherein the Au(l) complex is added to reaction mixture as such.
Furthermore the present invention relates to a process (F), which is the process (A), (B), (C), (D) or (E), wherein the Au(l) complex is formed in situ in the reaction mixture.
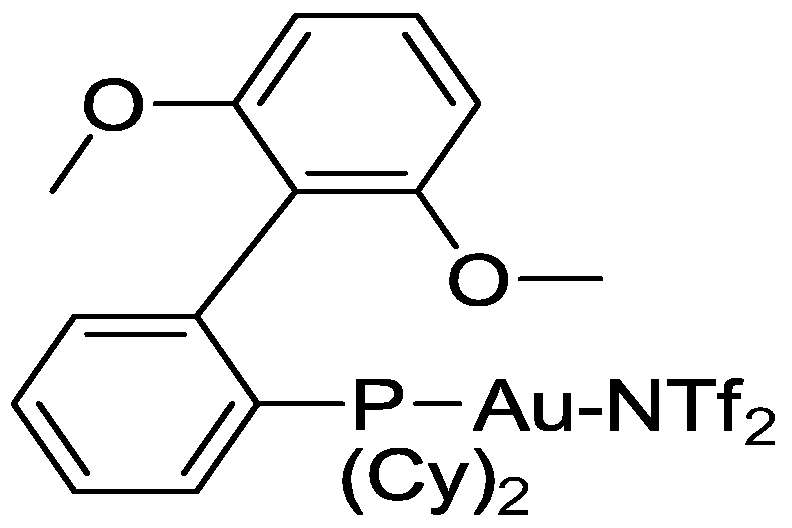
Preferred Au(l) complexes of formula (III) are the following ones.
wherein Cy is cyclohexyl, iPr is isopropyl and Tf is triflate.
Therefore a preferred process relates to a process as described above wherein at least one complex of formula (ΙΙΓ), (III") and/or (III'") is used.
The process according to the present invention is usually carried out in a solvent (or a mixture of solvents). Suitable solvents are aliphatic hydrocarbons, alcohols. Preferred solvents are alcohols, such as methanol, ethanol, 2-butanol and tert- butanol.
Therefore the present invention also relates to a process (G), which is the process (A), (B), (C), (D), (E) or (F), wherein the process is carried out in a solvent or a mixture of solvents.
Furthernnore the present invention relates to a process (G'), which is process (G) wherein the solvent is chosen from the group consisting of methanol, ethanol, 2- butanol and tert-butanol.
The process according to the present invention is usually carried out at elevated temperature, usually 30°C - 120°C. A preferred reaction temperature is between 40°C and 1 10°C, more preferred between 50°C and 100°C.
Therefore the present invention also relates to a process (H), which is the process (A), (B), (C), (D), (E), (F), (G) or (G'), wherein the process is carried out at elevated temperature.
Therefore the present invention also relates to a process (Η'), which is the process (H), wherein the process is carried out at a temperature from 30°C to 120°C.
Therefore the present invention also relates to a process (H"), which is the process (H), wherein the process is carried out at a temperature from 40°C to 1 10°C.
Therefore the present invention also relates to a process (Η"'), which is the process (H), wherein the process is carried out at a temperature from 50°C to 100°C.
Usually the Au(l) complex is present in an amount, wherein the substrate
(compound of formula (II)) to catalyst ratio is 2 : 1 to 10000 : 1 , preferred are 10 : 1 to 3000 : 1 .
Therefore the present invention also relates to a process (I), which is the process (A), (B), (C), (D), (E), (F), (G), (G'), (H), (Η'), (H") or (Η"'), wherein the substrate (compound of formula (II)) to catalyst ratio is 2 : 1 to 10000 : 1 , preferably 10 : 1 to 3000 : 1 .
The reaction time of the process (to obtain the desired product in reasonable amount) according to the present invention is usually 60 minutes to 300 minutes.
As stated above the compounds of formula (I) can be used as starting material to the synthesis of other organic compounds (especially the ones of formulae (la) -
(ig))-
The following examples serve to illustrate the invention. If not otherwise stated the temperature is given in degree Celsius and all parts are related to the weight.
Examples
Example 1 : Rearrangement of 1 -ethynyl-3,3-dimethylcyclohexanol
1 18.8 mg (0.2mmol, 0.1 equiv.) of dicyclohexylphosphine biphenyl gold(l)- chloride and 56.88 mg (0.2 mmol, 0.1 equiv.) of silver triflate were dissolved under argon at 23°C in 5.0 ml of tert.-butanol in a septum bottle. 362.6 μΙ (90%, 2 mmol, 1 equiv.) of 1 -ethynyl-3,3-dimethylcyclohexanol (90%, 2 mmol, 1 equiv.) were added. The reaction mixture was stirred for 105 min at 80°C. Afterwards the reaction mixture was cooled to 23°C. A sample was taken out and analyzed by GC and NMR.
The conversion of this reaction was 98.9%. The products of formulae (Γ and I") were obtained in a yield of 89.6 wt-%.
It is possible to separate the two isomers if desired.
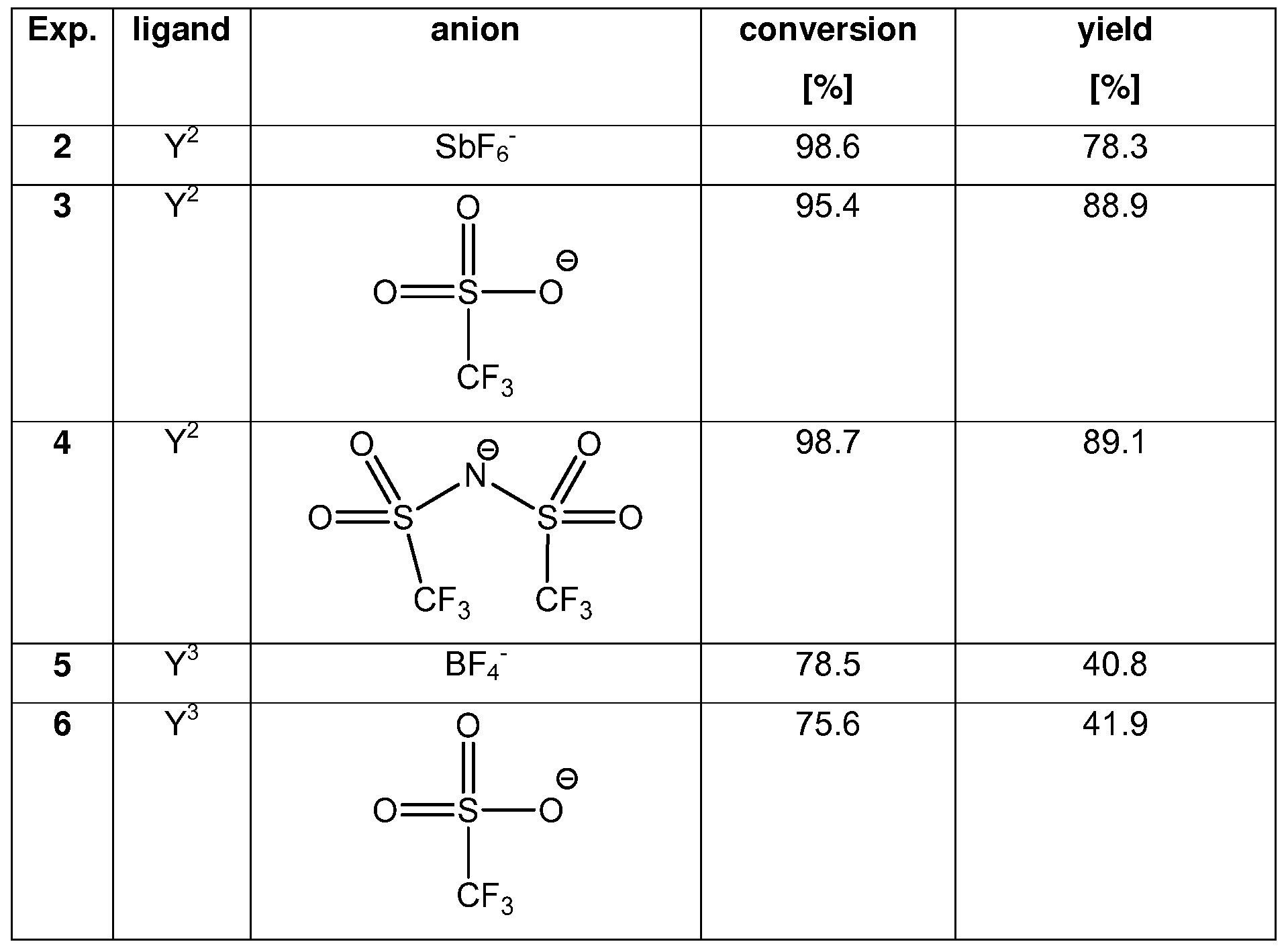
Examples 2 - 6
The following examples were carried out under the same reaction condition as in example 1 .
The ligands are defined as above and the ligands are added in the chloride form and the anions in the form of the Ag(l) salt.
Comparative examples (examples 7 - 11)
The same reaction as described in Example 1 was repeated, but Au catalysts, which do not fall under the scope of the claims were used.
Example 7 and 8 are carried out without any organic ligands. AuCI was used in combination with the silver salt
Exp. ligand anion conversion yield
[%] [%]
7 - BF4-" 16.8 0
Example 9, 10 and 11 and 8 are carried out with catalysts where Au(0) is adsorbed on a carrier.
Therefore it can be seen that the choice of the suitable (claimed) catalyst is essential.