Treatment of Cancer with Heterocyclic Inhibitors of Glutaminase
Related Applications
This application claims the benefit of priority to U.S. Provisional Patent Application No. 61/732,755, filed December 3, 2012, U.S. Provisional Patent Application No. 61/749,016, filed January 4, 2013, U.S. Provisional Patent
Application No. 61/784,984, filed March 14, 2013, U.S. Provisional Patent
Application No. 61/809,795, filed April 8, 2013, and U.S. Provisional Patent
Application No. 61/824,513, filed May 17, 2013, which applications are hereby incorporated by reference in their entirety. Background
Glutamine supports cell survival, growth and proliferation through metabolic and non-metabolic mechanisms. In actively proliferating cells, the metabolism of glutamine to lactate, also referred to as "glutaminolysis" is a major source of energy in the form of NADPH. The first step in glutaminolysis is the deamination of glutamine to form glutamate and ammonia, which is catalyzed by the glutaminase enzyme (GLS). Thus, deamination via glutaminase is a control point for glutamine metabolism.
Ever since Warburg's observation that ascites tumor cells exhibited high rates of glucose consumption and lactate secretion in the presence of oxygen (Warburg, 1956), researchers have been exploring how cancer cells utilize metabolic pathways to be able to continue actively proliferating. Several reports have demonstrated how glutamine metabolism supports macromolecular synthesis necessary for cells to replicate (Curthoys, 1995; DeBardinis, 2008).
Thus, glutaminase has been theorized to be a potential therapeutic target for the treatment of diseases characterized by actively proliferating cells, such as cancer. The lack of suitable glutaminase inhibitors has made validation of this target impossible. Therefore, the creation of glutaminase inhibitors that are specific and capable of being formulated for in vivo use could lead to a new class of therapeutics.
Summary of Invention
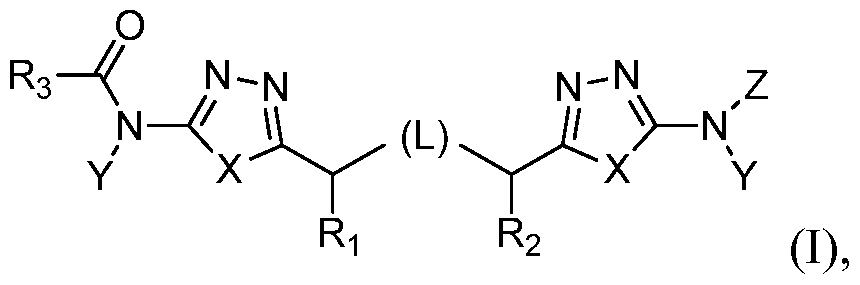
The present invention provides a method of treating or preventing cancer comprising administering a compound of formula I,
or a pharmaceutically acceptable salt thereof, wherein:
L represents CH2SCH2, CH2CH2, CH2CH2CH2, CH2, CH2S, SCH2, CH2NHCH2,
CH=CH, or ^ ^ , preferably CH2CH2, wherein any hydrogen atom of a CH or CH2 unit may be replaced by alkyl or alkoxy, any hydrogen of an NH unit may be replaced by alkyl, and any hydrogen atom of a CH2 unit of CH2CH2, CH2CH2CH2 or CH2 may be replaced by hydroxy;
X, independently for each occurrence, represents S, O or CH=CH, preferably S or
CH=CH, wherein any hydrogen atom of a CH unit may be replaced by alkyl;
Y, independently for each occurrence, represents H or CH20(CO)R7 ;
R7, independently for each occurrence, represents H or substituted or unsubstituted alkyl, alkoxy, aminoalkyl, alkylaminoalkyl, heterocyclylalkyl, arylalkyl, or heterocyclylalkoxy;
Z represents H or R3(CO);
Ri and R2 each independently represent H, alkyl, alkoxy or hydroxy;
R3, independently for each occurrence, represents substituted or unsubstituted alkyl, hydroxyalkyl, aminoalkyl, acylaminoalkyl, alkenyl, alkoxy, alkoxyalkyl, aryl, arylalkyl, aryloxy, aryloxyalkyl, cycloalkyl, cycloalkylalkyl, heterocyclyl, heterocyclylalkyl, heteroaryl, heteroarylalkyl, heteroaryloxy,
heteroaryloxyalkyl or C(Rg)(R9)(Rio), N(R4)(R5) or OR5, wherein any free hydroxyl group may be acylated to form C(0)R7;
R4 and R5 each independently represent H or substituted or unsubstituted alkyl,
hydroxyalkyl, acyl, aminoalkyl, acylaminoalkyl, alkenyl, alkoxyalkyl, aryl, arylalkyl, aryloxy, aryloxyalkyl, cycloalkyl, cycloalkylalkyl, heterocyclyl, heterocyclylalkyl, heteroaryl, heteroarylalkyl, heteroaryloxy, or
heteroaryloxyalkyl, wherein any free hydroxyl group may be acylated to form C(0)R7;
Re, independently for each occurrence, represents substituted or unsubstituted alkyl, hydroxyalkyl, aminoalkyl, acylaminoalkyl, alkenyl, alkoxyalkyl, aryl, arylalkyl, aryloxy, aryloxyalkyl, cycloalkyl, cycloalkylalkyl, heterocyclyl, heterocyclylalkyl, heteroaryl, heteroarylalkyl, heteroaryloxy, or
heteroaryloxyalkyl, wherein any free hydroxyl group may be acylated to form C(0)R7; and
Rg, R9 and Rio each independently represent H or substituted or unsubstituted alkyl, hydroxy, hydroxyalkyl, amino, acylamino, aminoalkyl, acylaminoalkyl, alkoxycarbonyl, alkoxycarbonylamino, alkenyl, alkoxy, alkoxyalkyl, aryl, arylalkyl, aryloxy, aryloxyalkyl, cycloalkyl, cycloalkylalkyl, heterocyclyl, heterocyclylalkyl, heteroaryl, heteroarylalkyl, heteroaryloxy, or
heteroaryloxyalkyl, or Rg and R
9 together with the carbon to which they are attached, form a carbocyclic or heterocyclic ring system, wherein any free hydroxyl group may be acylated to form C(0)R
7, and wherein at least two of
In certain embodiments, the cancer is selected from breast cancer, colorectal cancer, endocrine cancer, melanoma, renal cancer and B cell malignancy. In certain such embodiments wherein the cancer is breast cancer, the breast cancer comprises basal-type breast cancer cells, triple-negative breast cancer cells or claudin-low breast cancer cells. In certain embodiments wherein the cancer is endocrine cancer, the endocrine cancer is selected from adrenal cortex adenoma, adrenal cortex carcicnoma, adrenal gland pheochromocytoma and parathyroid gland adenoma. In certain embodiments wherein the cancer is a B cell malignancy, the B cell malignancy is selected from multiple myeloma, leukemia, such as acute lymphoblastic leukemia or chronic lymphoblastic leukemia, and lymphoma, such as Burkitt's lymphoma, Diffuse large B cell lymphoma, follicular lymphoma or Hodgkin's lymphoma.
In certain embodiments, the present invention provides a pharmaceutical preparation suitable for use in a human patient in the treatment or prevention of cancer, such as breast cancer, colorectal cancer, endocrine cancer, melanoma, renal cancer or B cell malignancy, comprising an effective amount of any of the compounds described herein (e.g., a compound of the invention, such as a compound of formula
I), and one or more pharmaceutically acceptable excipients. In certain embodiments, the pharmaceutical preparations may be for use in treating or preventing a condition or disease as described herein. In certain embodiments, the pharmaceutical preparations have a low enough pyrogen activity to be suitable for intravenous use in a human patient.
Detailed Description of the Drawings
Figure 1 shows the correlation between glutamine-dependence and antiproliferative effect of compound 670 for a panel of breast tumor cell lines.
Figure 2 shows the differential expression of glutaminase and glutamine synthetase in triple-negative breast cancer subtype.
Figure 3 shows single-agent compound 402 treatment of MDA-MB-231 orthotopic xenograft model.
Figure 4 shows a combination study with compound 389 and paclitaxel in MDA-MB-231 orthotopic xenograft model.
Figure 5 shows results of the median glutaminase: glutamine synthetase expression ratio in various cancer types, including colorectal cancer, renal cancer, lymphoma, melanoma and myeloma.
Figure 6 shows that the glutaminase: glutamine synthetase expression ratio varies by subtypes in endocrine cancers.
Figure 7 depicts the median glutaminase: glutamine synthetase expression ratio in acute lymphoblastic leukemia (ALL) and chronic lymphocytic leukemia (CLL).
Figure 8 shows the glutaminase: glutamine synthetase expression ratio for several subtypes of lymphomas within the B cell malignancy category.
Figure 9 shows the correlation between the antiproliferative effect of compound 670 and the glutamate:glutamine concentration ratios for a panel of breast tumor cell lines.
Figure 10 shows the correlation between the glutamate: glutamine
concentration ratios to glutaminase: glutamine synthetase expression ratios and to glutaminase specific activity in a variety of primary tumor xenografts.
Figure 11 shows that intraperitoneal administration of compound 188 to mice results in reduced tumor size in a HCT116 colon carcinoma xenograft model.
Figure 12 shows that oral administration of compound 670 to mice results in reduced tumor size in a H2122 lung adenocarcinoma xenograft model.
Figure 13 shows the mRNA expression levels of GLS (KGA or GAC), GS, and the ratio of KGA:GS and GAC:GS in TNBC vs. HR+ or Her2+ cell lines. The "box" depicts the 2nd and 3rd quartiles with the median corresponding to the horizontal line; "whiskers" span the 10th and 90th percentile with data outside this range shown as individual data points.
Figure 14 shows correlation between the sensitivity to Compound 670 and mRNA expression levels of GLS, GS, or expression ratios. For each bivariate graph, the Compound 670 sensitivity is plotted on the x-axis and the expression parameter is plotted on the y-axis with each point representing an individual cell line.
Figure 15 shows western analysis of KGA, GAC and GS in breast cancer cell lines. Blots were probed with antibodies recognizing KGA, GAC and GS. The CAG antibody also recognizes KGA and the two are distinguishable on the blot by their molecular weight differences. Blots were stripped and re-probed with GAPDH as a loading control.
Figure 16 shows the correlation between the glutamate:glutamine
concentration ratios to sensitivity to glutaminase inhibitor compound 670.
Figure 17 shows that oral administration of compound 670 to mice results in reduced tumor size in a RPMI-8226 multiple myeloma xenograft model.
Figure 18 shows that compound 670 synergizes with pomalidomide or dexamethasone to produce an anti-tumor effect in multiple myeloma cells.
Detailed Description of the Invention
The present invention provides a method of treating or preventing cancer comprising administering a compound of formula I,
or a pharmaceutically acceptable salt thereof, wherein:
L represents CH2SCH2, CH2CH2, CH2CH2CH2, CH2, CH2S, SCH2, CH2NHCH2,
CH=CH, or ^ .A ^, , preferably CH2CH2, wherein any hydrogen atom of a
CH or CH2 unit may be replaced by alkyl or alkoxy, any hydrogen of an NH unit may be replaced by alkyl, and any hydrogen atom of a CH2 unit of
CH2CH2, CH2CH2CH2 or CH2 may be replaced by hydroxy;
X, independently for each occurrence, represents S, O or CH=CH, preferably S or
CH=CH, wherein any hydrogen atom of a CH unit may be replaced by alkyl; Y, independently for each occurrence, represents H or CH20(CO)R7 ;
R7, independently for each occurrence, represents H or substituted or unsubstituted alkyl, alkoxy, aminoalkyl, alkylaminoalkyl, heterocyclylalkyl, arylalkyl, or heterocyclylalkoxy;
Z represents H or R3(CO);
Ri and R2 each independently represent H, alkyl, alkoxy or hydroxy;
R3, independently for each occurrence, represents substituted or unsubstituted alkyl, hydroxyalkyl, aminoalkyl, acylaminoalkyl, alkenyl, alkoxy, alkoxyalkyl, aryl, arylalkyl, aryloxy, aryloxyalkyl, cycloalkyl, cycloalkylalkyl, heterocyclyl, heterocyclylalkyl, heteroaryl, heteroarylalkyl, heteroaryloxy,
heteroaryloxyalkyl or C(Rg)(R9)(Rio), N(R )(R5) or O 5, wherein any free hydroxyl group may be acylated to form C(0)R7;
R4 and R5 each independently represent H or substituted or unsubstituted alkyl,
hydroxyalkyl, acyl, aminoalkyl, acylaminoalkyl, alkenyl, alkoxyalkyl, aryl, arylalkyl, aryloxy, aryloxyalkyl, cycloalkyl, cycloalkylalkyl, heterocyclyl, heterocyclylalkyl, heteroaryl, heteroarylalkyl, heteroaryloxy, or
heteroaryloxyalkyl, wherein any free hydroxyl group may be acylated to form
C(0)R7;
5, independently for each occurrence, represents substituted or unsubstituted alkyl, hydroxyalkyl, aminoalkyl, acylaminoalkyl, alkenyl, alkoxyalkyl, aryl, arylalkyl, aryloxy, aryloxyalkyl, cycloalkyl, cycloalkylalkyl, heterocyclyl, heterocyclylalkyl, heteroaryl, heteroarylalkyl, heteroaryloxy, or
heteroaryloxyalkyl, wherein any free hydroxyl group may be acylated to form C(0)R7; and
Rg, R and Rio each independently represent H or substituted or unsubstituted alkyl, hydroxy, hydroxyalkyl, amino, acylamino, aminoalkyl, acylaminoalkyl, alkoxycarbonyl, alkoxycarbonylamino, alkenyl, alkoxy, alkoxyalkyl, aryl, arylalkyl, aryloxy, aryloxyalkyl, cycloalkyl, cycloalkylalkyl, heterocyclyl, heterocyclylalkyl, heteroaryl, heteroarylalkyl, heteroaryloxy, or
heteroaryloxyalkyl, or Rg and R
9 together with the carbon to which they are attached, form a carbocyclic or heterocyclic ring system, wherein any free hydroxyl group may be acylated to form C(0)R
7, and wherein at least two of
In certain embodiments wherein alkyl, hydroxyalkyl, amino, acylamino, aminoalkyl, acylaminoalkyl, alkenyl, alkoxy, alkoxyalkyl, aryl, arylalkyl, aryloxy, aryloxyalkyl, cycloalkyl, cycloalkylalkyl, heterocyclyl, heterocyclylalkyl, heteroaryl, heteroarylalkyl, heteroaryloxy, or heteroaryloxyalkyl are substituted, they are substituted with one or more substituents selected from substituted or unsubstituted alkyl, such as perfluoroalkyl (e.g., trifluoromethyl), alkenyl, alkoxy, alkoxyalkyl, aryl, aralkyl, arylalkoxy, aryloxy, aryloxyalkyl, hydroxyl, halo, alkoxy, such as
perfluoroalkoxy (e.g., trifluoromethoxy), alkoxyalkoxy, hydroxyalkyl,
hydroxyalkylamino, hydroxyalkoxy, amino, aminoalkyl, alkylamino,
aminoalkylalkoxy, aminoalkoxy, acylamino, acylaminoalkyl, such as perfluoro acylaminoalkyl (e.g., trifluoromethylacylaminoalkyl), acyloxy, cycloalkyl, cycloalkylalkyl, cycloalkylalkoxy, heterocyclyl, heterocyclylalkyl, heterocyclyloxy, heterocyclylalkoxy, heteroaryl, heteroarylalkyl, heteroarylalkoxy, heteroaryloxy, heteroaryloxyalkyl, heterocyclylaminoalkyl, heterocyclylaminoalkoxy, amido, amidoalkyl, amidine, imine, oxo, carbonyl (such as carboxyl, alkoxycarbonyl, formyl, or acyl, including perfluoroacyl (e.g., C(0)CF3)), carbonylalkyl (such as
carboxyalkyl, alkoxycarbonylalkyl, formylalkyl, or acylalkyl, including
perfluoroacylalkyl (e.g., -alkylC(0)CF3)), carbamate, carbamatealkyl, urea, ureaalkyl, sulfate, sulfonate, sulfamoyl, sulfone, sulfonamide, sulfonamidealkyl, cyano, nitro, azido, sulfhydryl, alkylthio, thiocarbonyl (such as thioester, thioacetate, or
thioformate), phosphoryl, phosphate, phosphonate or phosphinate.
In certain embodiments, L represents CH2SCH2, CH2CH2, CH2CH2CH2, CH2, CH2S, SCH2, or CH2NHCH2, wherein any hydrogen atom of a CH2 unit may be replaced by alkyl or alkoxy, and any hydrogen atom of a CH2 unit of CH2CH2,
CH2CH2CH2 or CH2 may be replaced by hydroxyl. In certain embodiments, L represents CH2SCH2, CH2CH2, CH2S or SCH2. In certain embodiments, L represents CH2CH2. In certain embodiments, L is not CH2SCH2.
In certain embodiments, Y represents H.
In certain embodiments, X represents S or CH=CH. In certain embodiments, one or both X represents CH=CH. In certain embodiments, each X represents S. In certain embodiments, one X represents S and the other X represents CH=CH.
In certain embodiments, Z represents R3(CO). In certain embodiments wherein Z is R3(CO), each occurrence of R3 is not identical (e.g., the compound of formula I is not symmetrical).
In certain embodiments, Ri and R2 each represent H.
In certain embodiments, R3 represents arylalkyl, heteroarylalkyl, cycloalkyl or heterocycloalkyl. In certain embodiments, R3 represents C(Rg)(R9)(Rio), wherein R8 represents aryl, arylalkyl, heteroaryl or heteroaralkyl, such as aryl, arylalkyl or heteroaryl, R9 represents H, and Rio represents hydroxy, hydroxyalkyl, alkoxy or alkoxyalkyl, such as hydroxy, hydroxyalkyl or alkoxy.
In certain embodiments, L represents CH2SCH2, CH2CH2, CH2S or SCH2, such as CH2CH2, CH2S or SCH2, Y represents H, X represents S, Z represents R3(CO), Ri and R2 each represent H, and each R3 represents arylalkyl, heteroarylalkyl, cycloalkyl or heterocycloalkyl. In certain such embodiments, each occurrence of R3 is identical.
In certain embodiments, L represents CH2SCH2, CH2CH2, CH2S or SCH2, Y represents H, X represents S, Z represents R3(CO), Ri and R2 each represent H, and each R3 represents C(Rg)(R9)(Rio), wherein R8 represents aryl, arylalkyl, heteroaryl or heteroaralkyl, such as aryl, arylalkyl or heteroaryl, R9 represents H, and Rio represents hydroxy, hydroxyalkyl, alkoxy or alkoxyalkyl, such as hydroxy, hydroxyalkyl or alkoxy. In certain such embodiments, each occurrence of R3 is identical.
In certain embodiments, L represents CH2CH2, Y represents H, X represents S or CH=CH, Z represents R3(CO), Ri and R2 each represent H, and each R3 represents substituted or unsubstituted arylalkyl, heteroarylalkyl, cycloalkyl or heterocycloalkyl. In certain such embodiments, each X represents S. In other embodiments, one or both occurrences of X represents CH=CH, such as one occurrence of X represents S and the other occurrence of X represents CH=CH. In certain embodiments of the
foregoing, each occurrence of R3 is identical. In other embodiments of the foregoing wherein one occurrence of X represents S and the other occurrence of X represents CH=CH, the two occurrences of R3 are not identical.
In certain embodiments, L represents CH2CH2, Y represents H, X represents S, Z represents R3(CO), Ri and R2 each represent H, and each R3 represents
C(Rg)(R9)(Rio), wherein R8 represents aryl, arylalkyl or heteroaryl, R9 represents H, and Rio represents hydroxy, hydroxyalkyl or alkoxy. In certain such embodiments, Rs represents aryl and Rio represents hydroxyalkyl. In certain such embodiments, each occurrence of R3 is identical.
In certain embodiments wherein L represents CH2, CH2CH2CH2 or CH2CH2, X represents O, and Z represents R3(CO), both R3 groups are not alkyl, such as methyl, or C(R8)(R9)(Rio), wherein R8, R9 andRio are each independently hydrogen or alkyl.
In certain embodiments wherein L represents CH2CH2, X represents S, and Z represents R3(CO), both R3 groups are not phenyl or heteroaryl, such as 2-furyl.
In certain embodiments wherein L represents CH2CH2, X represents O, and Z represents R3(CO), both R3 groups are not N(R4)(R5) wherein R4 is aryl, such as phenyl, and R5 is H.
In certain embodiments wherein L represents CH2SCH2, X represents S, and Z represents R3(CO), both R3 groups are not aryl, such as optionally substituted phenyl, aralkyl, such as benzyl, heteroaryl, such as 2-furyl, 2-thienyl or 1,2,4-trizole, substituted or unsubstituted alkyl, such as methyl, chloromethyl, dichloromethyl, n- propyl, n-butyl, t-butyl or hexyl, heterocyclyl, such as pyrimidine-2,4(lH,3H)-dione, or alkoxy, such as methoxy, pentyloxy or ethoxy.
In certain embodiments wherein L represents CH2SCH2, X represents S, and Z represents R3(CO), both R3 groups are not N(R4)(R5) wherein R4 is aryl, such as substituted or unsubstituted phenyl (e.g., phenyl, 3-tolyl, 4-tolyl, 4-bromophenyl or 4- nitrophenyl), and R5 is H.
In certain embodiments wherein L represents CH2CH2CH2, X represents S, and Z represents R3(CO), both R3 groups are not alkyl, such as methyl, ethyl, or propyl, cycloalkyl, such as cyclohexyl, or C(Rg)(R9)(Rio), wherein any of Rg, R9 and Rio together with the C to which they are attached, form any of the foregoing.
In certain embodiments, the compound is not one of the following:
11
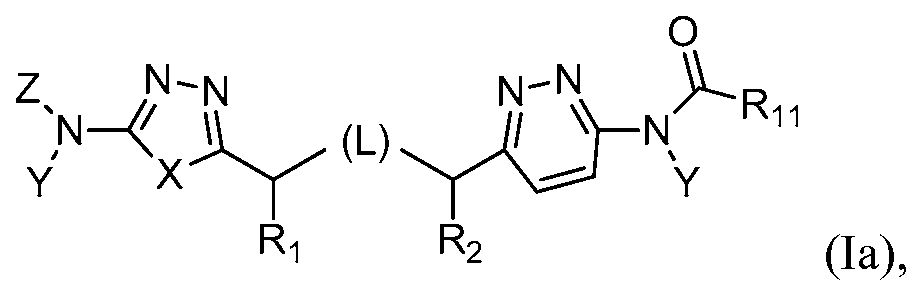
The present invention further provides a method of treating or preventing cancer comprising administering a compound of formula la,
or a pharmaceutically acceptable salt thereof, wherein:
L repre H
2, CH
2CH
2CH
2, CH
2, CH
2S, SCH
2, CH
2NHCH
2,
, preferably CH
2CH
2, wherein any hydrogen atom of a
CH or CH2 unit may be replaced by alkyl or alkoxy, any hydrogen of an NH unit may be replaced by alkyl, and any hydrogen atom of a CH2 unit of CH2CH2, CH2CH2CH2 or CH2 may be replaced by hydroxy;
X represents S, O or CH=CH, preferably S or CH=CH, wherein any hydrogen atom of a CH unit may be replaced by alkyl;
Y, independently for each occurrence, represents H or CH20(CO)R7 ;
R7, independently for each occurrence, represents H or substituted or unsubstituted alkyl, alkoxy, aminoalkyl, alkylaminoalkyl, heterocyclylalkyl, arylalkyl, or heterocyclylalkoxy;
Z represents H or R3(CO);
Ri and R2 each independently represent H, alkyl, alkoxy or hydroxy, preferably H;
R3 represents substituted or unsubstituted alkyl, hydroxyalkyl, aminoalkyl,
acylaminoalkyl, alkenyl, alkoxy, alkoxyalkyl, aryl, arylalkyl, aryloxy, aryloxyalkyl, cycloalkyl, cycloalkylalkyl, heterocyclyl, heterocyclylalkyl, heteroaryl, heteroarylalkyl, heteroaryloxy, heteroaryloxyalkyl or
C(R8)(R9)(Rio), N(R )(R5) or OR6, wherein any free hydroxyl group may be acylated to form C(0)R7;
R4 and R5 each independently represent H or substituted or unsubstituted alkyl,
hydroxyalkyl, acyl, aminoalkyl, acylaminoalkyl, alkenyl, alkoxyalkyl, aryl, arylalkyl, aryloxy, aryloxyalkyl, cycloalkyl, cycloalkylalkyl, heterocyclyl, heterocyclylalkyl, heteroaryl, heteroarylalkyl, heteroaryloxy, or
heteroaryloxyalkyl, wherein any free hydroxyl group may be acylated to form
C(0)R7;
5, independently for each occurrence, represents substituted or unsubstituted alkyl, hydroxyalkyl, aminoalkyl, acylaminoalkyl, alkenyl, alkoxyalkyl, aryl, arylalkyl, aryloxy, aryloxyalkyl, cycloalkyl, cycloalkylalkyl, heterocyclyl,
heterocyclylalkyl, heteroaryl, heteroarylalkyl, heteroaryloxy, or
heteroaryloxyalkyl, wherein any free hydroxyl group may be acylated to form C(0)R7; and
R8, R9 and R10 each independently represent H or substituted or unsubstituted alkyl, hydroxy, hydroxyalkyl, amino, acylamino, aminoalkyl, acylaminoalkyl, alkoxycarbonyl, alkoxycarbonylamino, alkenyl, alkoxy, alkoxyalkyl, aryl, arylalkyl, aryloxy, aryloxyalkyl, cycloalkyl, cycloalkylalkyl, heterocyclyl, heterocyclylalkyl, heteroaryl, heteroarylalkyl, heteroaryloxy, or
heteroaryloxyalkyl, or Rg and R9 together with the carbon to which they are attached, form a carbocyclic or heterocyclic ring system, wherein any free hydroxyl group may be acylated to form C(0)R7, and wherein at least two of R8, R9 and R10 are not H;
R11 represents substituted or unsubstituted aryl, arylalkyl, aryloxy, aryloxyalkyl,
heteroaryl, heteroarylalkyl, heteroaryloxy, or heteroaryloxyalkyl, or
C(Ri2)(Ri3)(Ri4), N(Rzt)(R14) or OR14, wherein any free hydroxyl group may be acylated to form C(0)R7;
Ri2 and R13 each independently respresent H or substituted or unsubstituted alkyl, hydroxy, hydroxyalkyl, amino, acylamino, aminoalkyl, acylaminoalkyl, alkoxycarbonyl, alkoxycarbonylamino, alkenyl, alkoxy, alkoxyalkyl, aryl, arylalkyl, aryloxy, aryloxyalkyl, cycloalkyl, cycloalkylalkyl, heterocyclyl, heterocyclylalkyl, heteroaryl, heteroarylalkyl, heteroaryloxy, or
heteroaryloxyalkyl, wherein any free hydroxyl group may be acylated to form C(0)R7, and wherein both of Ri2 and R13 are not H; and
Ri4 represents substituted or unsubstituted aryl, arylalkyl, aryloxy, aryloxyalkyl,
heteroaryl, heteroarylalkyl, heteroaryloxy, or heteroaryloxyalkyl.
In certain embodiments wherein alkyl, hydroxyalkyl, amino, acylamino, aminoalkyl, acylaminoalkyl, alkenyl, alkoxy, alkoxyalkyl, aryl, arylalkyl, aryloxy, aryloxyalkyl, cycloalkyl, cycloalkylalkyl, heterocyclyl, heterocyclylalkyl, heteroaryl, heteroarylalkyl, heteroaryloxy, or heteroaryloxyalkyl are substituted, they are substituted with one or more substituents selected from substituted or unsubstituted alkyl, such as perfluoroalkyl (e.g., trifluoromethyl), alkenyl, alkoxy, alkoxyalkyl, aryl, aralkyl, arylalkoxy, aryloxy, aryloxyalkyl, hydroxyl, halo, alkoxy, such as
perfluoroalkoxy (e.g., trifluoromethylalkoxy), alkoxyalkoxy, hydroxyalkyl,
hydroxyalkylamino, hydroxyalkoxy, amino, aminoalkyl, alkylamino, aminoalkylalkoxy, aminoalkoxy, acylamino, acylaminoalkyl, such as perfluoro acylaminoalkyl (e.g., trifluoromethylacylaminoalkyl), acyloxy, cycloalkyl, cycloalkylalkyl, cycloalkylalkoxy, heterocyclyl, heterocyclylalkyl, heterocyclyloxy, heterocyclylalkoxy, heteroaryl, heteroarylalkyl, heteroarylalkoxy, heteroaryloxy, heteroaryloxyalkyl, heterocyclylaminoalkyl, heterocyclylaminoalkoxy, amido, amidoalkyl, amidine, imine, oxo, carbonyl (such as carboxyl, alkoxycarbonyl, formyl, or acyl, including perfluoroacyl (e.g., C(0)CF3)), carbonylalkyl (such as
carboxyalkyl, alkoxycarbonylalkyl, formylalkyl, or acylalkyl, including
perfluoroacylalkyl (e.g., -alkylC(0)CF3)), carbamate, carbamatealkyl, urea, ureaalkyl, sulfate, sulfonate, sulfamoyl, sulfone, sulfonamide, sulfonamidealkyl, cyano, nitro, azido, sulfhydryl, alkylthio, thiocarbonyl (such as thioester, thioacetate, or
thioformate), phosphoryl, phosphate, phosphonate or phosphinate.
In certain embodiments, Rn represents substituted or unsubstituted arylalkyl, such as substituted or unsubstituted benzyl.
In certain embodiments, L represents CH2SCH2, CH2CH2, CH2CH2CH2, CH2, CH2S, SCH2, or CH2NHCH2, wherein any hydrogen atom of a CH2 unit may be replaced by alkyl or alkoxy, and any hydrogen atom of a CH2 unit of CH2CH2, CH2CH2CH2 or CH2 may be replaced by hydroxyl. In certain embodiments, L represents CH2SCH2, CH2CH2, CH2S or SCH2, preferably CH2CH2. In certain embodiments, L is not CH2SCH2.
In certain embodiments, each Y represents H. In other embodiments, at least one Y is CH20(CO)R7.
In certain embodiments, X represents S or CH=CH. In certain embodiments, X represents S.
In certain embodiments, Ri and R2 each represent H.
In certain embodiments, Z represents R3(CO). In certain embodiments wherein Z is R3(CO), R3 and Rn are not identical (e.g., the compound of formula I is not symmetrical).
In certain embodiments, Z represents R3(CO) and R3 represents arylalkyl, heteroarylalkyl, cycloalkyl or heterocycloalkyl. In certain embodiments, Z represents R3(CO) and R3 represents C(Rg)(R9)(Rio), wherein R8 represents aryl, arylalkyl, heteroaryl or heteroaralkyl, such as aryl, arylalkyl or heteroaryl, R9 represents H, and
Rio represents hydroxy, hydroxyalkyl, alkoxy or alkoxyalkyl, such as hydroxy, hydroxyalkyl or alkoxy. In certain embodiments, Z represents R3(CO) and R3 represents heteroarylalkyl.
In certain embodiments, L represents CH2SCH2, CH2CH2, CH2S or SCH2, such as CH2CH2, Y represents H, X represents S, Z represents R3(CO), Ri and R2 each represent H, R3 represents arylalkyl, heteroarylalkyl, cycloalkyl or heterocycloalkyl, and Rii represents arylalkyl. In certain such embodiments, R3 represents
heteroarylalkyl.
In certain embodiments, L represents CH2SCH2, CH2CH2, CH2S or SCH2, such as CH2CH2, Y represents H, X represents S, Z represents R3(CO), Ri and R2 each represent H, and R3 represents C(Rg)(R9)(Rio), wherein R8 represents aryl, arylalkyl, heteroaryl or heteroaralkyl, such as aryl, arylalkyl or heteroaryl, R9 represents H, and Rio represents hydroxy, hydroxyalkyl, alkoxy or alkoxyalkyl, such as hydroxy, hydroxyalkyl or alkoxy, and Rn represents arylalkyl. In certain such embodiments, Rs represents heteroaryl.
In certain embodiments, L represents CH2CH2, Y represents H, X represents S or CH=CH, such as S, Z represents R3(CO), Ri and R2 each represent H, R3 represents substituted or unsubstituted arylalkyl, heteroarylalkyl, cycloalkyl or heterocycloalkyl, and Rn represents arylalkyl. In certain such embodiments, R3 represents
heteroarylalkyl.
In certain embodiments, L represents CH2CH2, Y represents H, X represents S, Z represents R3(CO), Ri and R2 each represent H, R3 represents C(Rg)(R9)(Rio), wherein R8 represents aryl, arylalkyl or heteroaryl, R9 represents H, and Rio represents hydroxy, hydroxyalkyl or alkoxy, and Rn represents arylalkyl. In certain such embodiments, Rg represents aryl and Rio represents hydroxyalkyl. In certain other embodiments, Rs represents heteroaryl.
In certain embodiments, the cancer is selected from breast cancer, colorectal cancer, endocrine cancer, melanoma, renal cancer and B cell malignancy. In certain such embodiments wherein the cancer is breast cancer, the breast cancer comprises basal-type breast cancer cells, triple-negative breast cancer cells or claudin-low breast cancer cells. In certain embodiments wherein the cancer is endocrine cancer, the endocrine cancer is selected from adrenal cortex adenoma, adrenal cortex carcicnoma, adrenal gland pheochromocytoma and parathyroid gland adenoma. In certain
embodiments wherein the cancer is a B cell malignancy, the B cell malignancy is selected from multiple myeloma, leukemia, such as acute lymphoblastic leukemia or chronic lymphoblastic leukemia, and lymphoma, such as Burkitt's lymphoma, Diffuse large B cell lymphoma, follicular lymphoma or Hodgkin's lymphoma.
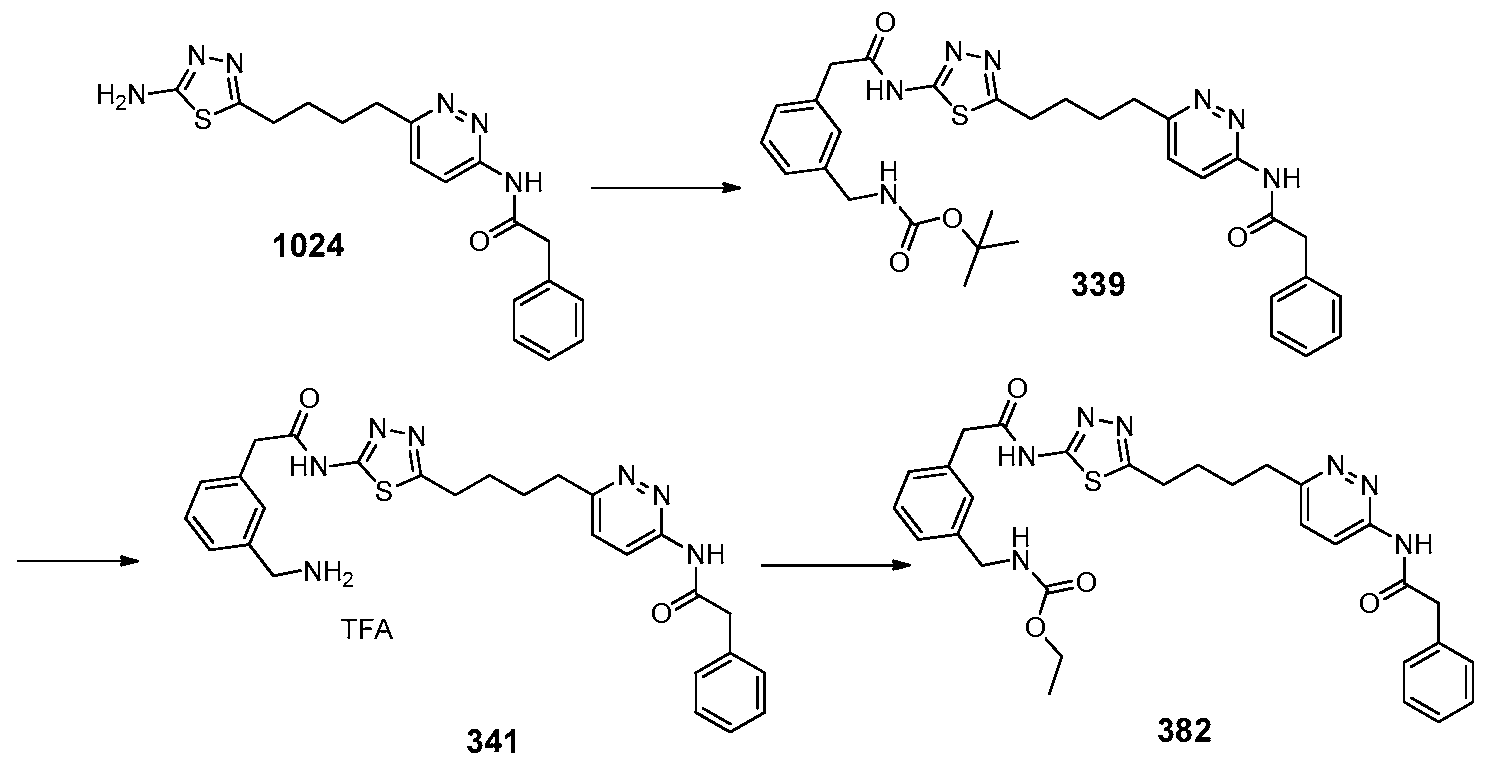
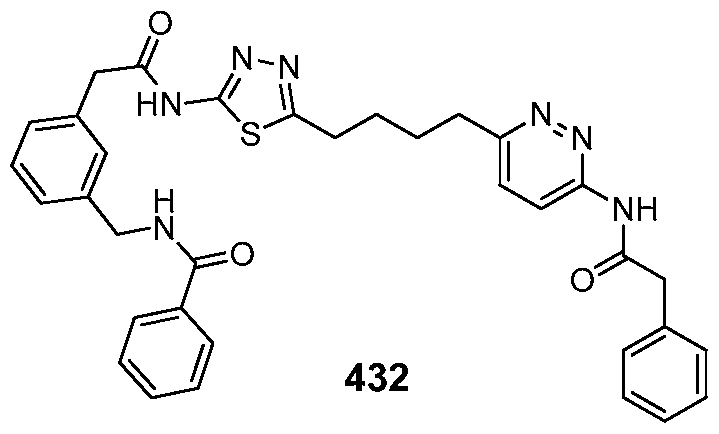
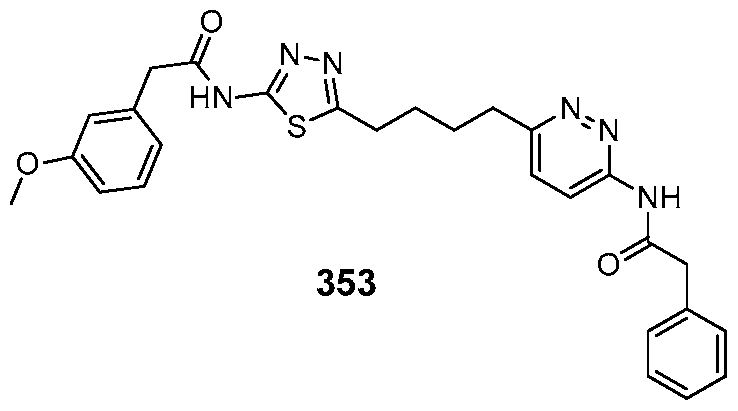
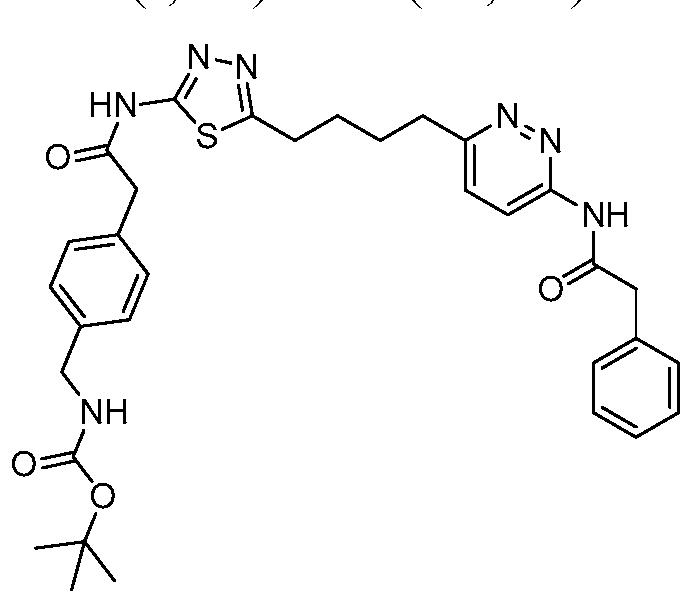
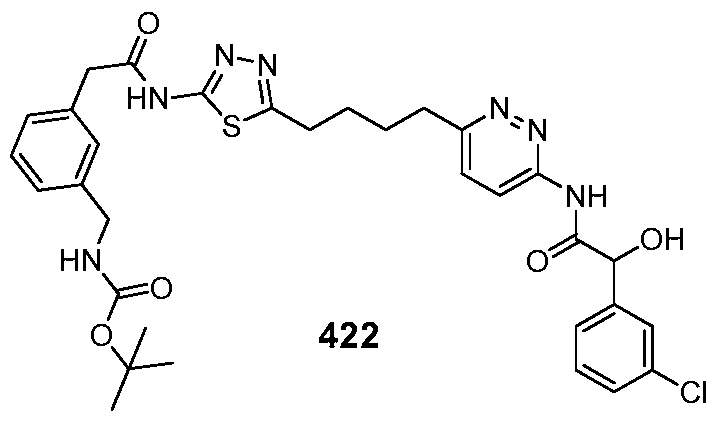
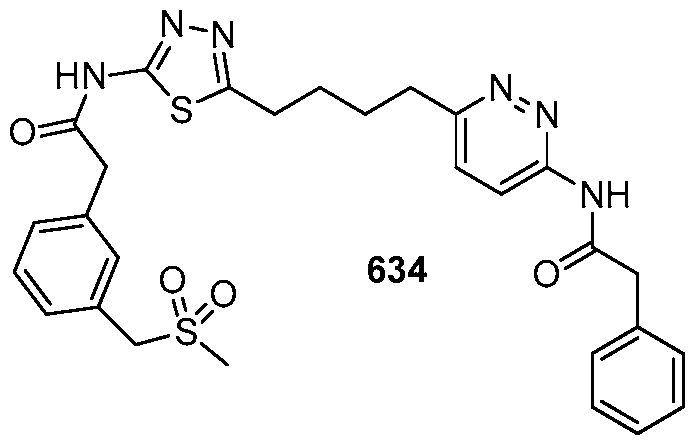
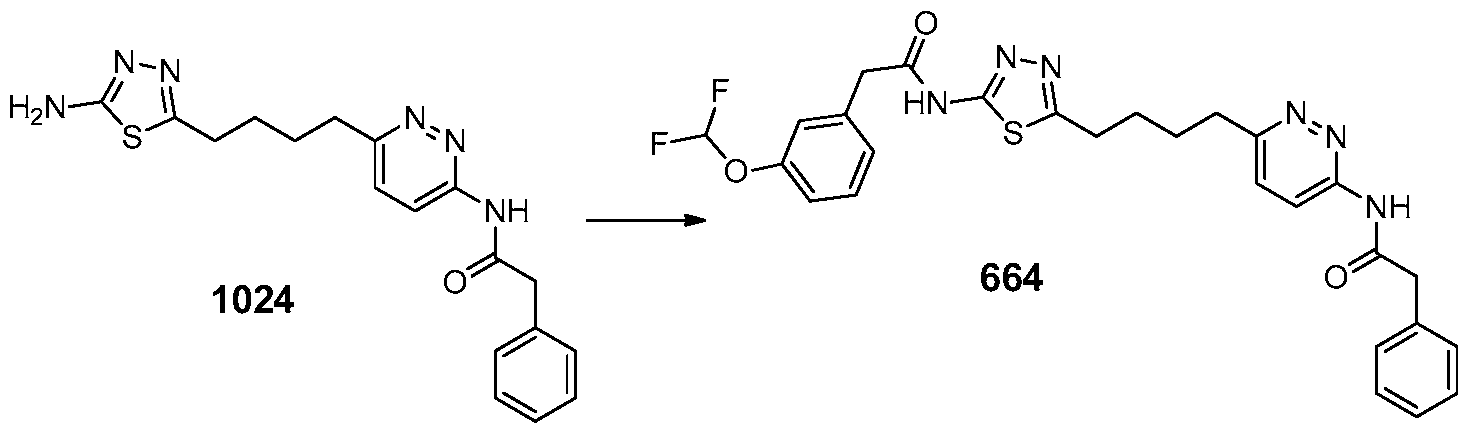
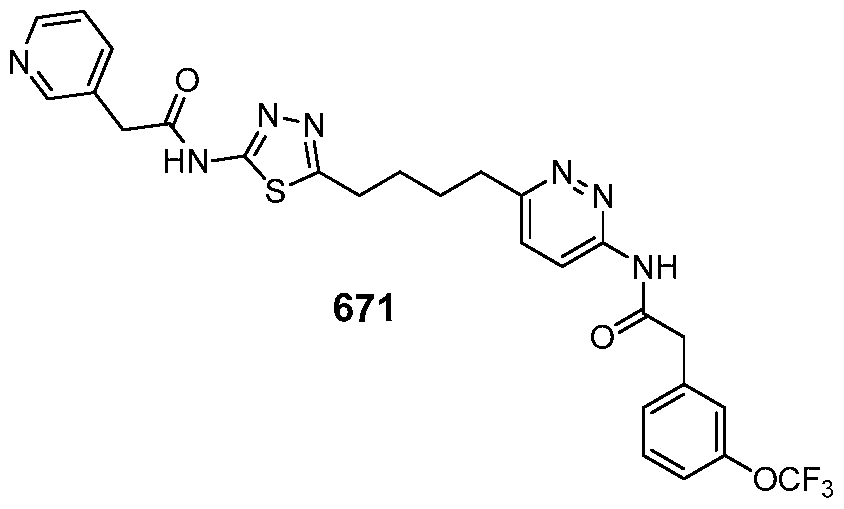
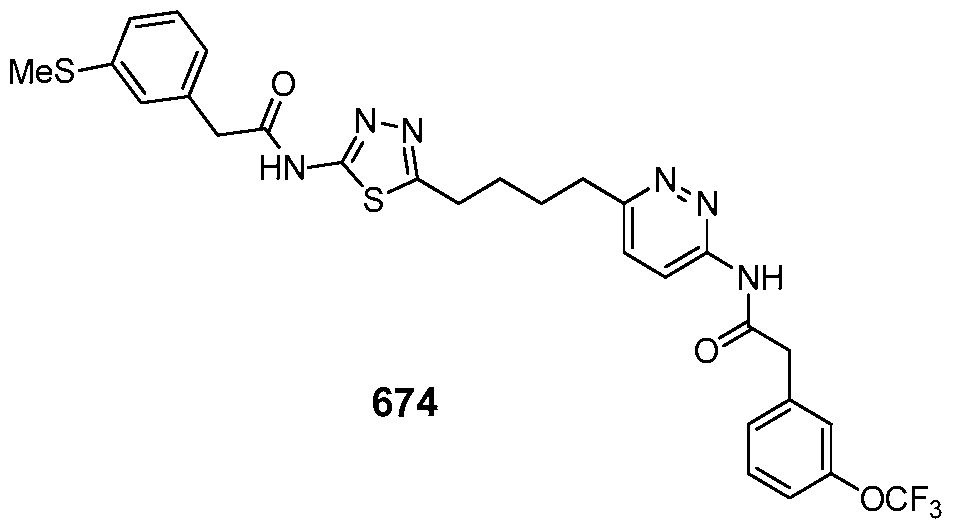
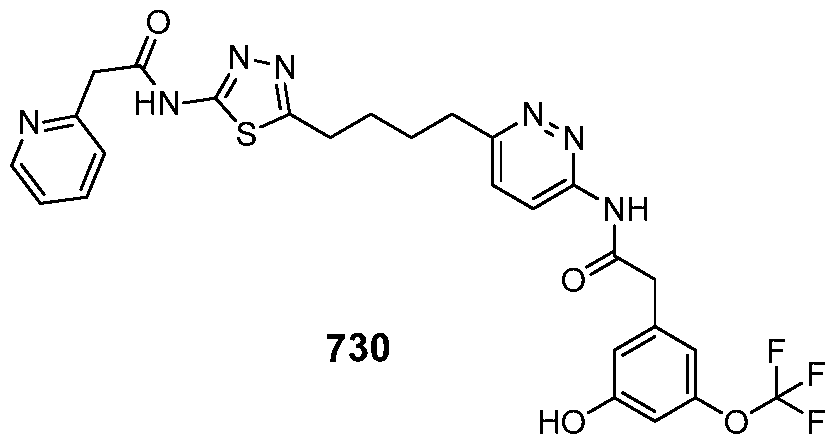
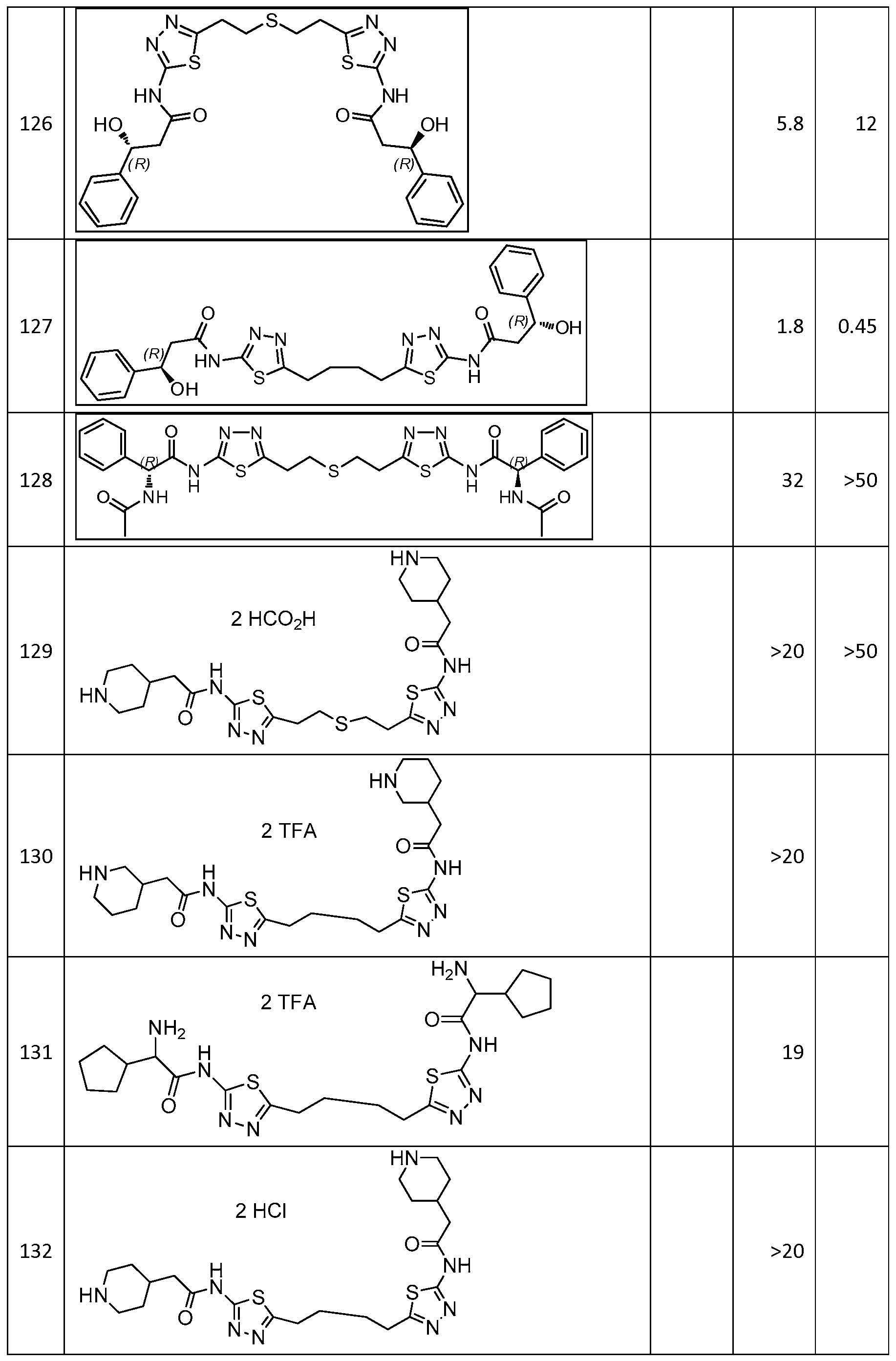
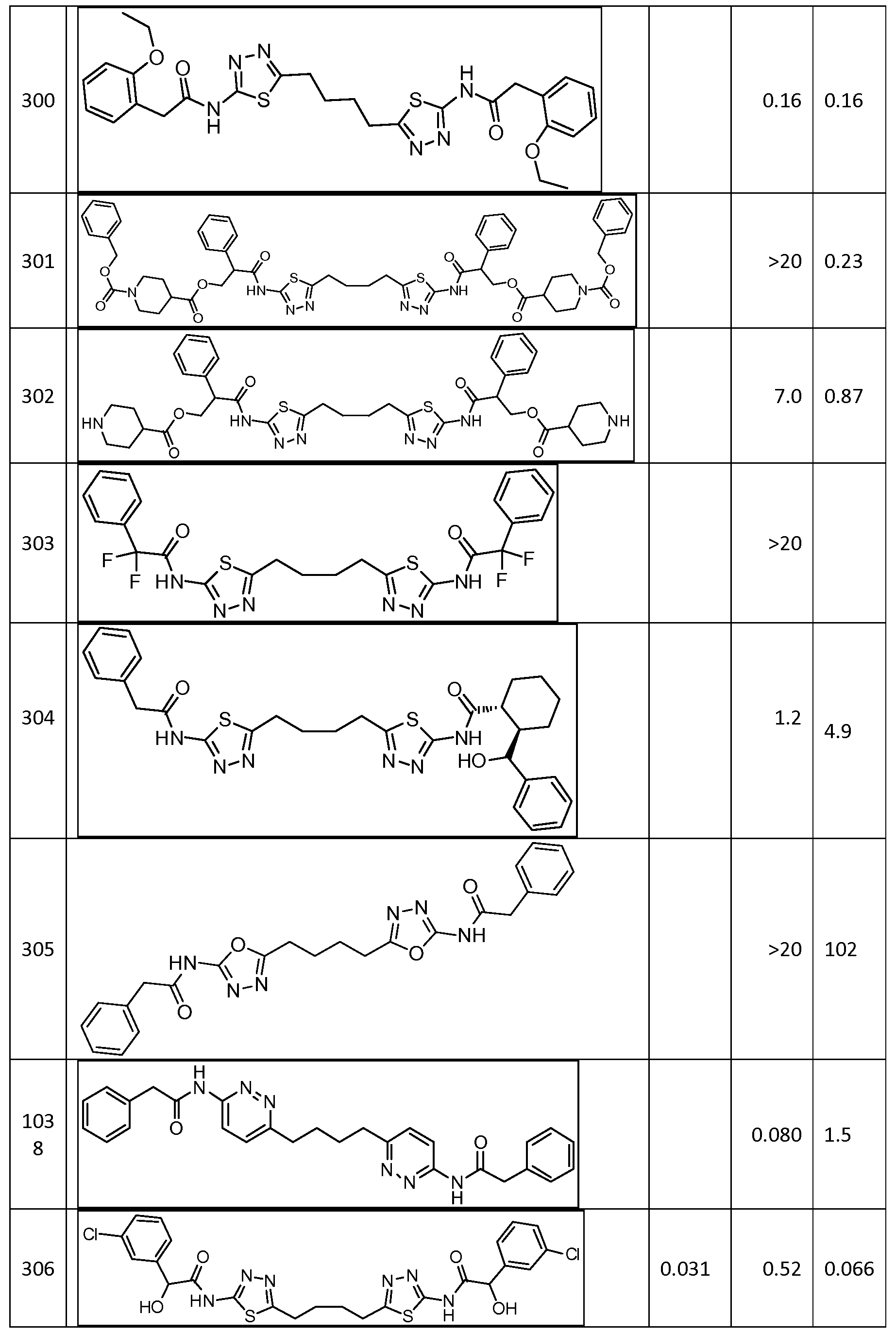
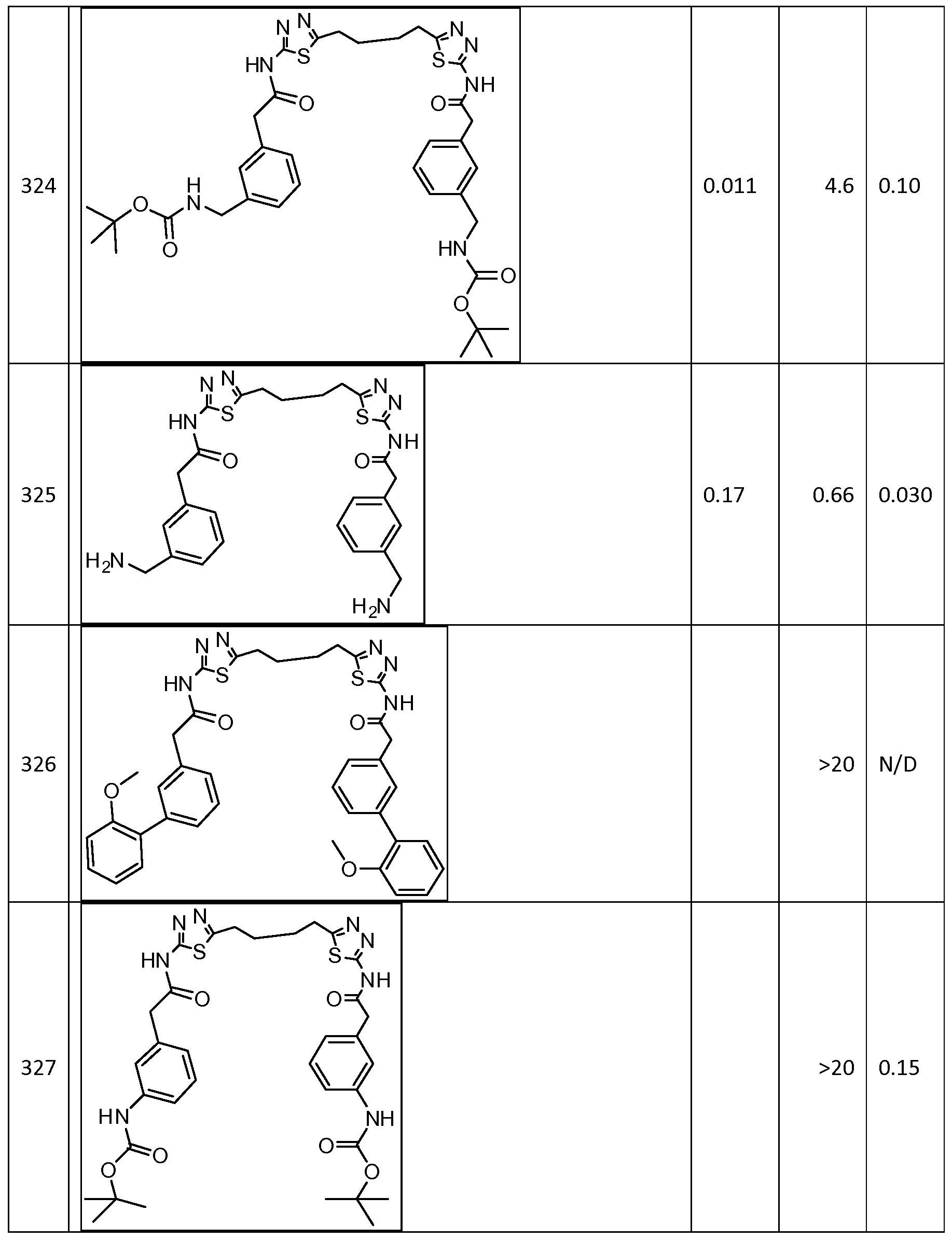
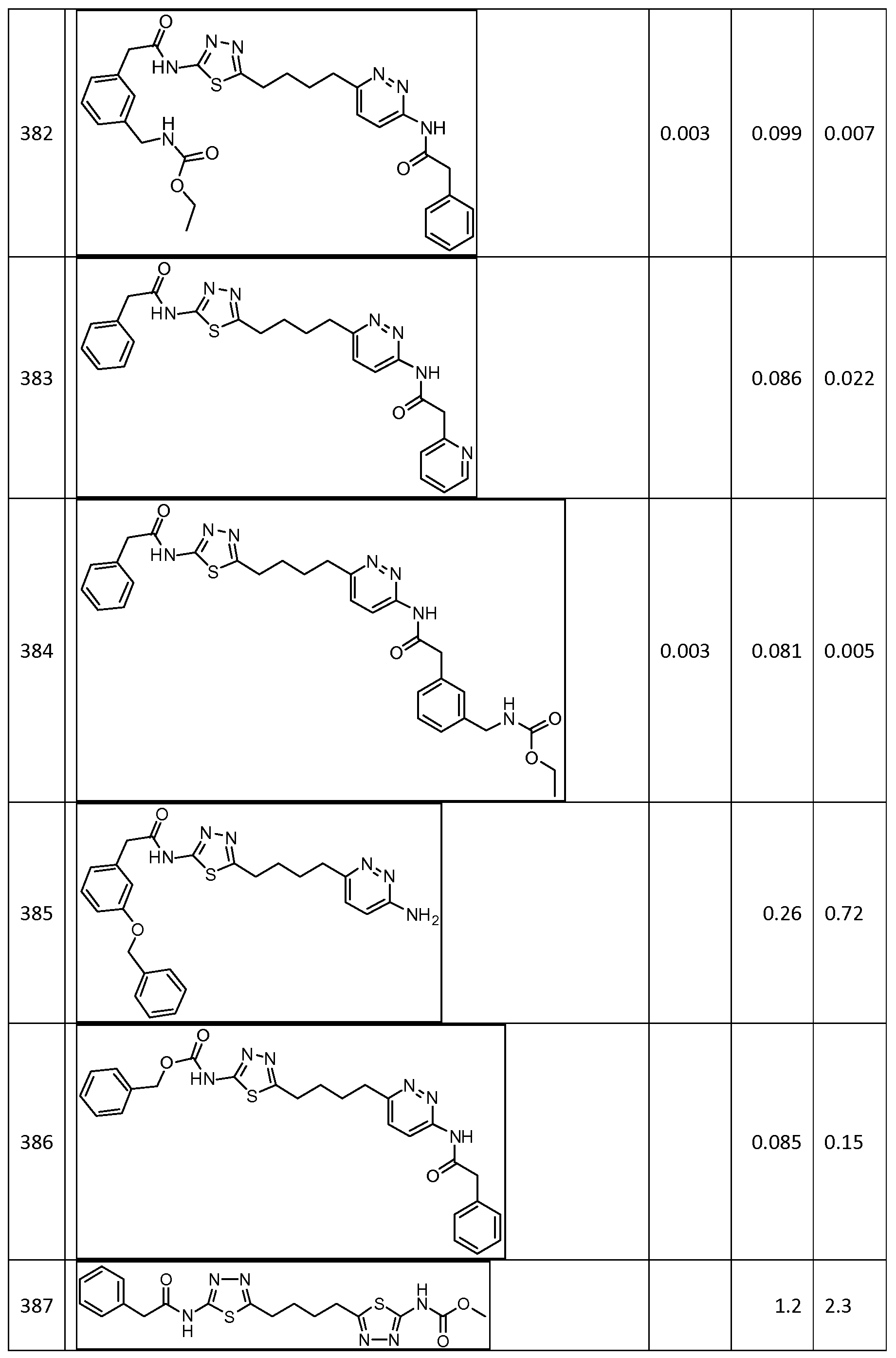
In certain embodiments, the compound is selected from any one of the compounds disclosed in Table 3. Preferably, the compound is selected from compound 1, 2, 6, 7, 8, 11, 13, 14, 15, 16, 17, 18, 19, 20, 21, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 35, 36, 38, 39, 40, 41, 43, 44, 47, 48, 50, 51, 52, 54, 55, 58, 63, 64, 65, 67, 68, 69, 70, 71, 72, 73, 77, 78, 79, 80, 81, 82, 83, 84, 85, 86, 87, 88, 92, 93, 94, 95, 97, 99, 100, 102, 105, 107, 111, 112, 114, 115, 116, 117, 118, 120, 121, 122, 123, 126, 127, 133, 135, 136, 138, 140, 141, 143, 146, 147, 148, 152, 153, 155, 156, 157, 158, 159, 160, 161, 162, 163, 164, 165, 166, 168, 169, 170, 172, 173, 174, 175, 176, 177, 178, 179, 180, 181, 182, 185, 186, 187, 188, 189, 190, 193, 194, 195, 196, 197, 198, 199, 200, 201, 202, 203, 204, 205, 208, 210, 211, 213, 214, 216, 217, 219, 220, 226, 227, 228, 229, 231, 232, 234, 235, 236, 237, 239, 240, 241, 242, 243, 244, 245, 246, 247, 248, 249, 250, 251, 252, 255, 256, 257, 258, 259, 260, 261, 262, 263, 264, 265, 266, 267, 268, 269, 270, 271, 273, 274, 275, 276, 278, 279, 280, 281, 282, 283, 285, 286, 287, 288, 290, 291, 292, 293, 294, 295, 296, 297, 298, 299, 300, 302, 304, 1038, 306, 307, 308, 309, 310, 311, 313, 314, 315, 316, 317, 318, 319, 320, 321, 322, 323, 324, 325, 327, 329, 332, 333, 334, 335, 336, 337, 338, 339, 340, 341, 342, 343, 344, 345, 346, 527, 347, 348, 349, 350, 351, 352, 353, 354, 355, 358, 359, 360, 361, 362, 363, 364, 365, 366, 367, 368, 369, 370, 371, 372, 373, 374, 375, 376, 377, 378, 379, 380, 381, 382, 383, 384, 385, 386, 387, 388, 389, 390, 391, 392, 393, 394, 395, 396, 397, 398, 399, 400, 401, 402, 403, 404, 405, 406, 407, 408, 409, 410, 411, 412, 413, 414, 415, 416, 417, 418, 419, 420, 421, 422, 423, 424, 425, 426, 427, 428, 429, 430, 431, 432, 433, 434, 435, 436, 437, 438, 439, 440, 441, 442, 443, 444, 445, 446, 447, 448, 449, 450, 451, 452, 453, 454, 455, 456, 457, 458, 459, 460, 461, 462, 463, 464, 465, 466, 467, 468, 469, 470, 471, 472, 473, 474, 475, 476, 477, 478, 479, 480, 481, 482, 483, 484, 485, 486, 487, 488, 489, 490, 491, 492, 493, 494, 495, 496, 497, 498, 499, 500, 501, 502, 503, 504, 505, 506, 507, 508, 509, 510, 511, 512, 513, 514, 515, 516, 517, 518, 519, 520, 521, 522, 523, 528, 529, 530, 531, 532, 533, 534, 535, 536, 537, 538, 539, 540, 541, 542, 543, 544, 545, 546, 547, 548, 549, 550, 551, 552, 553, 554, 555, 556, 557, 558, 559, 560, 561, 562, 563, 564, 565, 566, 567, 568, 569,
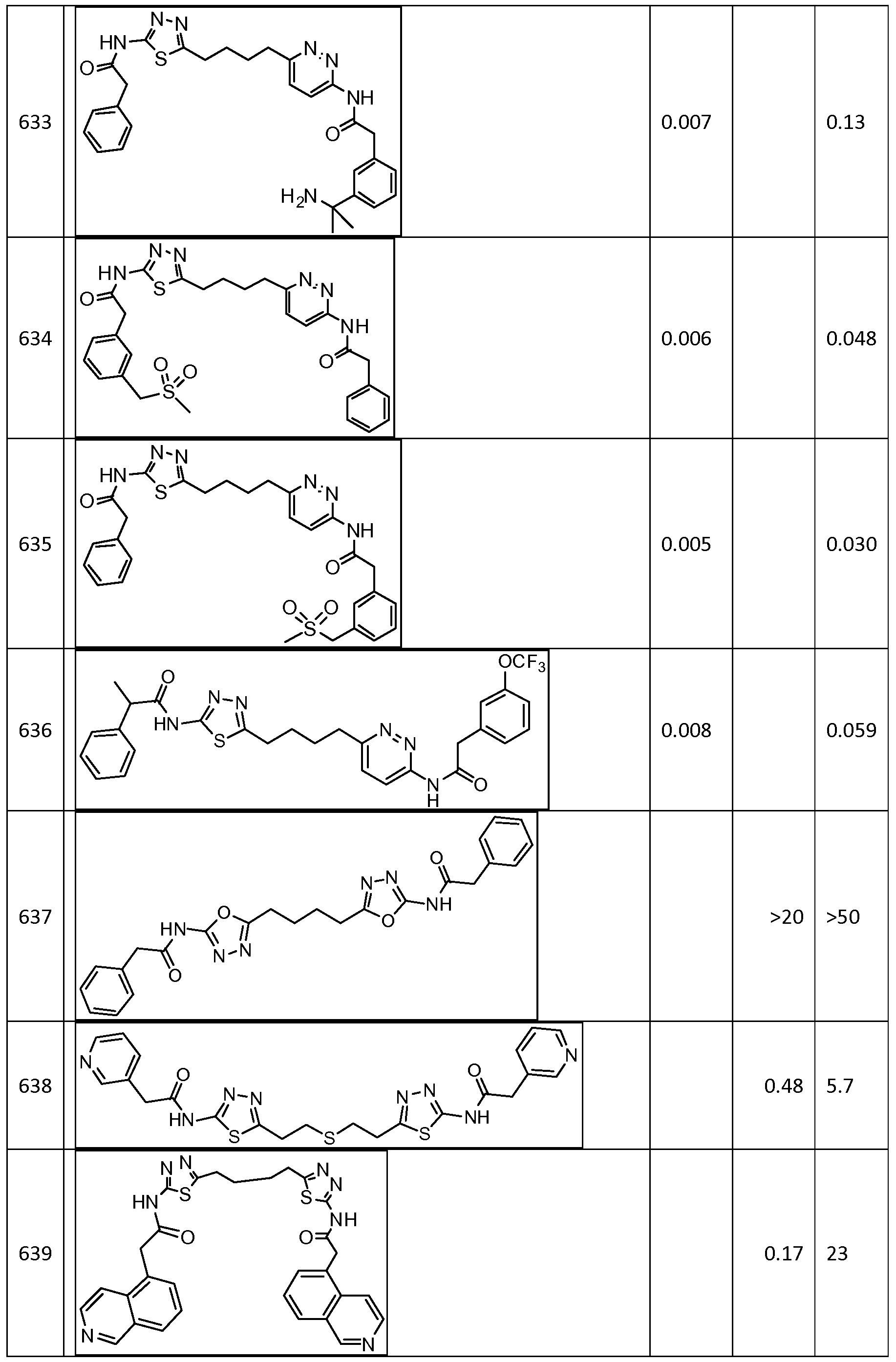
570, 571, 572, 573, 574, 575, 576, 577, 578, 579, 580, 581, 582, 583, 584, 585, 586, 587, 588, 589, 590, 591, 592, 593, 594, 595, 596, 597, 598, 599, 600, 601, 602, 603, 604, 605, 606, 607, 608, 609, 610, 611, 612, 613, 614, 615, 616, 617, 618, 619, 620, 621, 622, 623, 624, 625, 626, 627, 628, 629, 630, 631, 632, 633, 634, 635, 636, 638, 639, 640, 641, 644, 645, 646, 647, 648, 649, 650, 651, 652, 653, 654, 655, 656, 657, 658, 659, 660, 661, 662, 663, 664, 665, 666, 667, 668, 669, 670, 671, 672, 673, 674, 675, 676, 677, 678, 679, 680, 681, 682, 683, 684, 685, 686, 687, 688, 689, 690, 692, 693, 694, 695, 696, 697, 698, 699, 700, 701, 702, 703, 704, 705, 707, 708, 709, 715, 716, 717, 718, 719, 720, 721, 722, 723, 724, 725, 726, 727, 728, 729, or 730.
In certain embodiments, compounds of the invention may be prodrugs of the compounds of formula I or la, e.g., wherein a hydroxyl in the parent compound is presented as an ester or a carbonate, or carboxylic acid present in the parent compound is presented as an ester. In certain such embodiments, the prodrug is metabolized to the active parent compound in vivo (e.g., the ester is hydrolyzed to the corresponding hydroxyl, or carboxylic acid).
In certain embodiments, compounds of the invention may be racemic. In certain embodiments, compounds of the invention may be enriched in one
enantiomer. For example, a compound of the invention may have greater than 30% ee, 40% ee, 50% ee, 60% ee, 70% ee, 80% ee, 90% ee, or even 95% or greater ee. In certain embodiments, compounds of the invention may have more than one stereocenter. In certain such embodiments, compounds of the invention may be enriched in one or more diastereomer. For example, a compound of the invention may have greater than 30% de, 40% de, 50% de, 60% de, 70% de, 80% de, 90% de, or even 95% or greater de.
In certain embodiments, the present invention relates to methods of treating or preventing cancer, such as breast cancer, colorectal cancer, endocrine cancer, melanoma, renal cancer or B cell malignancy, with a compound of formula I or la, or a pharmaceutically acceptable salt thereof. In certain embodiments, the therapeutic preparation may be enriched to provide predominantly one enantiomer of a compound (e.g., of formula I or la). An enantiomerically enriched mixture may comprise, for example, at least 60 mol percent of one enantiomer, or more preferably at least 75, 90, 95, or even 99 mol percent. In certain embodiments, the compound enriched in one enantiomer is substantially free of the other enantiomer, wherein substantially free
means that the substance in question makes up less than 10%, or less than 5%, or less than 4%, or less than 3%, or less than 2%, or less than 1% as compared to the amount of the other enantiomer, e.g., in the composition or compound mixture. For example, if a composition or compound mixture contains 98 grams of a first enantiomer and 2 grams of a second enantiomer, it would be said to contain 98 mol percent of the first enantiomer and only 2% of the second enantiomer.
In certain embodiments, the therapeutic preparation may be enriched to provide predominantly one diastereomer of a compound (e.g., of formula I or la). A diastereomerically enriched mixture may comprise, for example, at least 60 mol percent of one diastereomer, or more preferably at least 75, 90, 95, or even 99 mol percent.
In certain embodiments, the present invention provides a pharmaceutical preparation suitable for use in a human patient, comprising any of the compounds shown above (e.g., a compound of the invention, such as a compound of formula I or la), and one or more pharmaceutically acceptable excipients. In certain embodiments, the pharmaceutical preparations may be for use in treating or preventing a condition or disease as described herein. In certain embodiments, the pharmaceutical preparations have a low enough pyrogen activity to be suitable for use in a human patient.
Compounds of any of the above structures may be used in the manufacture of medicaments for the treatment of any diseases or conditions disclosed herein.
Uses of enzyme inhibitors
Glutamine plays an important role as a carrier of nitrogen, carbon, and energy.
It is used for hepatic urea synthesis, for renal ammoniagenesis, for gluconeogenesis, and as respiratory fuel for many cells. Cells get their glutamine by either synthesizing it internally via an enzyme called glutamine synthetase (GS) or exogenously from the environment.
The conversion of glutamine into glutamate is initiated by the mitochondrial enzyme, glutaminase. There are two major forms of the enzyme, K-type and L-type, which are distinguished by their Km values for glutamine and response to glutamate, wherein the Km value, or Michaelis constant, is the concentration of substrate required to reach half the maximal velocity. The L-type, also known as "liver-type" or GLS2, has a high Km for glutamine and is glutamate resistant. The K-type, also
known as "kidney-type" or GLS1 or "KGA", has a low Km for glutamine and is inhibited by glutamate. An alternative splice form of GLS1, referred to as
glutaminase C or "GAC", has recently been identified.
In addition to serving as the basic building blocks of protein synthesis, amino acids have been shown to contribute to many processes critical for growing and dividing cells, and this is particularly true for cancer cells. Nearly all definitions of cancer include reference to dysregulated proliferation. Numerous studies on glutamine metabolism in cancer indicate that many tumors are avid glutamine consumers (Souba, Ann. Surg., 1993; Collins et al, J. Cell. Physiol, 1998; Medina, J. Nutr., 2001; Shanware et al, J. Mol. Med., 2011), and this includes, but not limited to breast cancer. Certain embodiments of the invention relate to the use of the compounds described herein for the treatment of breast cancer.
While many cancer cells depend on exogenous glutamine for survival, the degree of glutamine dependence among tumor cell subtypes may make a population of cells more susceptible to the reduction of glutamine. As an example, gene expression analysis of breast cancers has identified five intrinsic subtypes (luminal A, luminal B, basal, HER2+, and normal-like) (Sorlie et al., Proc Natl Acad Sci USA, 2001). Although glutamine deprivation has an impact on cell growth and viability, basal-like cells appear to be more sensitive to the reduction of exogenous glutamine (Kung et al, PLoS Genetics, 2011). This supports the concept that glutamine is a very important energy source in basal-like breast cancer cell lines, and suggests that inhibition of the glutaminase enzyme would be beneficial in the treatment of breast cancers comprised of basal-like cells. Figure 1 further supports the correlation that cells dependent on exogenous glutamine are susceptible to the presence of a glutaminase inhibitor. Certain embodiments of the present invention relate to the method of treating basal-like breast cancer cells comprising administering a glutaminase inhibitor of the present application.
Enzyme expression levels can be determined in multiple manners, and quantitation is relative, based on a specific standard for each assay. The results can be used to provide a genetic profile, where the levels of certain genes, mRNAs or resulting expression products form a signature pattern that can used to characterize cell types. Kung et al, demonstrated that the basal-like breast cancer cells that showed glutamine dependency exhibited a genetic profile in which GLS expression was
relatively high and GS expression was relatively low. Furthermore, the expression level of GLS2 was relatively low. Analysis of primary breast tumors mRNA expression dataset (The Cancer Genome Atlas; N=756) support that basal-type cells generally have high GLS expression relative to GS expression.
Triple-negative breast cancer (TNBC) is characterized by a lack of estrogen receptor (ER), progesterone receptor (PR) and human epidermal growth factor receptor 2 (HER2) expression. It has a higher rate of relapse following
chemotherapy, and a poorer prognosis than with the other breast cancer subtypes (Dent et al., Clin Cancer Res, 2007). Interestingly, there appears to be significant similarities in metabolic profiling between TNBC cells and basal-like breast cancer cells. In particular, TNBC cells appear to have a similar genetic signature of high GLS expression and low GS expression (Figure 2). A more specific analysis of GLS expression in breast cancer cell lines revealed that TNBC cells express higher levels of both splice variants of GLS1, KGA and GAC, as well as significantly lower levels of GS, when compared to hormone receptor (HR)-positive, or Her2 -positive cell lines (Figures 13 and 15). An aspect of the present invention provides a method for treating breast cancer comprising TNBC cells comprising administering a glutaminase inhibitor of the present application.
More recently, another breast cancer cell type has been identified, called claudin-low (Prat et al., Breast Cancer Res, 2010). The genetic profile of this cell type also exhibits relatively high GLS expression and low GS expression. Analysis of several claudin-low breast cancer cell lines revealed that these cells were generally dependent on exogenous glutamine and also susceptible to glutaminase inhibition. An aspect of the present invention provides a method for treating breast cancer comprising claudin-low cells comprising administering a glutaminase inhibitor of the present application.
Another aspect of the invention is the use of the compounds described herein for the treatment of breast cancer comprising cells selected from basal-type breast cancer cells, triple-negative breast cancer cells, and claudin-low breast cancer cells.
This led to the hypothesis that the high GLS expression and low GS expression profile may serve as a genetic signature to identify other cancers that may be particularly dependent on exogenous glutamine, and therefore susceptible to glutaminase inhibition. Upon analysis of a vast number of primary human cancers
from a commercial database, several cancers exhibited high GLS to low GS expression patterns. In addition to the breast cancers previously noted, colorectal cancer, endocrine cancers, lung cancer, melanoma, mesothelioma, renal cancer and B cell malignancies had notably high GLS/GS ratios (Figures 5 and 10). Certain embodiments of the invention relate to the use of the compounds described herein for the treatment of cancers selected from colorectal cancer, endocrine cancer, lung cancer, melanoma, mesothelioma, renal cancer and B cell malignancies.
As with breast cancer, certain subtypes of some of these cancers appear to have a more prevalent GLS/GS expression ratio. For example, of the endocrine cancers, adrenal cortex adenoma, adrenal cortex carcinoma, adrenal gland
pheochromocytoma and parathyroid gland adenoma had ratios greater than three times that of endometrial entometrioid adenocarcinoma (Figure 6).
Within the data set, B cell malignancies included such cancers as multiple myeloma, leukemia (including acute lymphoblastic leukemia (ALL) and chronic lymphoblastic leukemia (CLL)) and lymphoma (including Burkitt's lymphoma, diffuse large B cell lymphoma, follicular lymphoma and Hodgkin's lymphoma). All these cancers displayed a genetic profile comprising high GLS/GS expression level ratios, further suggesting that these cancers would be susceptible to glutaminase inhibition (Figures 7 and 8). Figure 17 demonstrates that administration of glutaminase inhibitor compound reduced tumor size in a multiple myeloma xenograft model, further supporting this concept. Certain embodiments of the invention relate to the use of the compounds described herein for the treatment of multiple myeloma, leukemia and lymphoma.
In some embodiments, the method of treating or preventing cancer, such as breast cancer, colorectal cancer, endocrine cancer, melanoma, renal cancer or B cell malignancy, may comprise administering a compound of the invention conjointly with one or more other chemotherapeutic agent(s). Chemotherapeutic agents that may be conjointly administered with compounds of the invention include: ABT-263, aminoglutethimide, amsacrine, anastrozole, asparaginase, beg, bicalutamide, bleomycin, bortezomib, buserelin, busulfan, campothecin, capecitabine, carboplatin, carfilzomib, carmustine, chlorambucil, chloroquine, cisplatin, cladribine, clodronate, colchicine, cyclophosphamide, cyproterone, cytarabine, dacarbazine, dactinomycin, daunorubicin, demethoxyviridin, dexamethasone, dichloroacetate, dienestrol,
diethylstilbestrol, docetaxel, doxorubicin, epirubicin, estradiol, estramustine, etoposide, everolimus, exemestane, filgrastim, fludarabine, fludrocortisone, f uorouracil and 5-f uorouracil, f uoxymesterone, f utamide, gemcitabine, genistein, goserelin, hydroxyurea, idarubicin, ifosfamide, imatinib, interferon, irinotecan, ironotecan, lenalidomide, letrozole, leucovorin, leuprolide, levamisole, lomustine, lonidamine, mechlorethamine, medroxyprogesterone, megestrol, melphalan, mercaptopurine, mesna, metformin, methotrexate, mitomycin, mitotane,
mitoxantrone, nilutamide, nocodazole, octreotide, oxaliplatin, paclitaxel, pamidronate, pentostatin, perifosine, PF-04691502, plicamycin, pomalidomide, porfimer, procarbazine, raltitrexed, rituximab, romidepsin, sorafenib, streptozocin, sunitinib, suramin, tamoxifen, temozolomide, temsirolimus, teniposide, testosterone, thalidomide, thioguanine, thiotepa, titanocene dichloride, topotecan, trastuzumab, tretinoin, vinblastine, vincristine, vindesine, vinorelbine, and vorinostat (SAHA). For example, chemotherapeutic agents that may be conjointly administered with compounds of the invention include: aminoglutethimide, amsacrine, anastrozole, asparaginase, beg, bicalutamide, bleomycin, bortezomib, buserelin, busulfan, campothecin, capecitabine, carboplatin, carfilzomib, carmustine, chlorambucil, chloroquine, cisplatin, cladribine, clodronate, colchicine, cyclophosphamide, cyproterone, cytarabine, dacarbazine, dactinomycin, daunorubicin, demethoxyviridin, dichloroacetate, dienestrol, diethylstilbestrol, docetaxel, doxorubicin, epirubicin, estradiol, estramustine, etoposide, everolimus, exemestane, filgrastim, fludarabine, fludrocortisone, fluorouracil, fluoxymesterone, flutamide, gemcitabine, genistein, goserelin, hydroxyurea, idarubicin, ifosfamide, imatinib, interferon, irinotecan, ironotecan, lenalidomide, letrozole, leucovorin, leuprolide, levamisole, lomustine, lonidamine, mechlorethamine, medroxyprogesterone, megestrol, melphalan, mercaptopurine, mesna, metformin, methotrexate, mitomycin, mitotane,
mitoxantrone, nilutamide, nocodazole, octreotide, oxaliplatin, paclitaxel, pamidronate, pentostatin, perifosine, plicamycin, pomalidomide, porfimer, procarbazine, raltitrexed, rituximab, sorafenib, streptozocin, sunitinib, suramin, tamoxifen, temozolomide, temsirolimus, teniposide, testosterone, thalidomide, thioguanine, thiotepa, titanocene dichloride, topotecan, trastuzumab, tretinoin, vinblastine, vincristine, vindesine, and vinorelbine. In other embodiments, chemotherapeutic agents that may be conjointly administered with compounds of the invention include:
ABT-263, dexamethasone, 5-fluorouracil, PF-04691502, romidepsin, and vorinostat (SAHA). In certain embodiments of the methods of the invention described herein, the chemotherapeutic agent conjointly administered with compounds of the invention is a taxane chemotherapeutic agent, such as paclitaxel or docetaxel. In certain embodiments of the methods of the invention described herein, the chemotherapeutic agent conjointly administered with compounds of the invention is doxorubicin. In certain embodiments of the methods of the invention described herein, a compound of the invention is administered conjointly with a taxane chemotherapeutic agent (e.g., paclitaxel) and doxorubicin.
Many combination therapies have been developed for the treatment of cancer.
In certain embodiments, compounds of the invention may be conjointly administered with a combination therapy. Examples of combination therapies with which compounds of the invention may be conjointly administered are included in Table 1. Table 1 : Exemplary combinatorial therapies for the treatment of cancer.
BOMP Bleomycin, Vincristine, Cisplatin, Mitomycin
CA Cytarabine, Asparaginase
CABO Cisplatin, Methotrexate, Bleomycin, Vincristine
CAF Cyclophosphamide, Doxorubicin, Fluorouracil
CAL-G Cyclophosphamide, Daunorubicin, Vincristine,
Prednisone, Asparaginase
CAMP Cyclophosphamide, Doxorubicin, Methotrexate,
Procarbazine
CAP Cyclophosphamide, Doxorubicin, Cisplatin
CaT Carboplatin, Paclitaxel
CAV Cyclophosphamide, Doxorubicin, Vincristine
CAVE ADD CAV and Etoposide
CA-VP16 Cyclophosphamide, Doxorubicin, Etoposide
CC Cyclophosphamide, Carboplatin
CDDP/VP-16 Cisplatin, Etoposide
CEF Cyclophosphamide, Epirubicin, Fluorouracil
CEPP(B) Cyclophosphamide, Etoposide, Prednisone, with or
without/ Bleomycin
CEV Cyclophosphamide, Etoposide, Vincristine
CF Cisplatin, Fluorouracil or Carboplatin Fluorouracil
CHAP Cyclophosphamide or Cyclophosphamide, Altretamine,
Doxorubicin, Cisplatin
ChlVPP Chlorambucil, Vinblastine, Procarbazine, Prednisone
CHOP Cyclophosphamide, Doxorubicin, Vincristine, Prednisone
CHOP-BLEO Add Bleomycin to CHOP
CISCA Cyclophosphamide, Doxorubicin, Cisplatin
CLD-BOMP Bleomycin, Cisplatin, Vincristine, Mitomycin
CMF Methotrexate, Fluorouracil, Cyclophosphamide
CMFP Cyclophosphamide, Methotrexate, Fluorouracil,
Prednisone
CMFVP Cyclophosphamide, Methotrexate, Fluorouracil,
Name Therapeutic agents
Vincristine, Prednisone
CMV Cisplatin, Methotrexate, Vinblastine
CNF Cyclophosphamide, Mitoxantrone, Fluorouracil
CNOP Cyclophosphamide, Mitoxantrone, Vincristine, Prednisone
COB Cisplatin, Vincristine, Bleomycin
CODE Cisplatin, Vincristine, Doxorubicin, Etoposide
COMLA Cyclophosphamide, Vincristine, Methotrexate,
Leucovorin, Cytarabine
COMP Cyclophosphamide, Vincristine, Methotrexate, Prednisone
Cooper Regimen Cyclophosphamide, Methotrexate, Fluorouracil,
Vincristine, Prednisone
COP Cyclophosphamide, Vincristine, Prednisone
COPE Cyclophosphamide, Vincristine, Cisplatin, Etoposide
COPP Cyclophosphamide, Vincristine, Procarbazine, Prednisone
CP(Chronic Chlorambucil, Prednisone
lymphocytic leukemia)
CP (Ovarian Cancer) Cyclophosphamide, Cisplatin
CT Cisplatin, Paclitaxel
CVD Cisplatin, Vinblastine, Dacarbazine
CVI Carboplatin, Etoposide, Ifosfamide, Mesna
CVP Cyclophosphamide, Vincristine, Prednisome
CVPP Lomustine, Procarbazine, Prednisone
CYVADIC Cyclophosphamide, Vincristine, Doxorubicin,
Dacarbazine
DA Daunorubicin, Cytarabine
DAT Daunorubicin, Cytarabine, Thioguanine
DAV Daunorubicin, Cytarabine, Etoposide
DCT Daunorubicin, Cytarabine, Thioguanine
DHAP Cisplatin, Cytarabine, Dexamethasone
DI Doxorubicin, Ifosfamide
DTIC/Tamoxifen Dacarbazine, Tamoxifen
Name Therapeutic agents
DVP Daunorubicin, Vincristine, Prednisone
EAP Etoposide, Doxorubicin, Cisplatin
EC Etoposide, Carboplatin
EFP Etoposie, Fluorouracil, Cisplatin
ELF Etoposide, Leucovorin, Fluorouracil
EM A 86 Mitoxantrone, Etoposide, Cytarabine
EP Etoposide, Cisplatin
EVA Etoposide, Vinblastine
FAC Fluorouracil, Doxorubicin, Cyclophosphamide
FAM Fluorouracil, Doxorubicin, Mitomycin
FAMTX Methotrexate, Leucovorin, Doxorubicin
FAP Fluorouracil, Doxorubicin, Cisplatin
F-CL Fluorouracil, Leucovorin
FEC Fluorouracil, Cyclophosphamide, Epirubicin
FED Fluorouracil, Etoposide, Cisplatin
FL Flutamide, Leuprolide
FZ Flutamide, Goserelin acetate implant
HDMTX Methotrexate, Leucovorin
Hexa-CAF Altretamine, Cyclophosphamide, Methotrexate,
Fluorouracil
ICE-T Ifosfamide, Carboplatin, Etoposide, Paclitaxel, Mesna
IDMTX/6-MP Methotrexate, Mercaptopurine, Leucovorin
IE Ifosfamide, Etoposie, Mesna
IfoVP Ifosfamide, Etoposide, Mesna
IPA Ifosfamide, Cisplatin, Doxorubicin
M-2 Vincristine, Carmustine, Cyclophosphamide, Prednisone,
Melphalan
MAC-III Methotrexate, Leucovorin, Dactinomycin,
Cyclophosphamide
MACC Methotrexate, Doxorubicin, Cyclophosphamide,
Lomustine
Name Therapeutic agents
MACOP-B Methotrexate, Leucovorin, Doxorubicin,
Cyclophosphamide, Vincristine, Bleomycin, Prednisone
MAID Mesna, Doxorubicin, Ifosfamide, Dacarbazine
m-BACOD Bleomycin, Doxorubicin, Cyclophosphamide, Vincristine,
Dexamethasone, Methotrexate, Leucovorin
MBC Methotrexate, Bleomycin, Cisplatin
MC Mitoxantrone, Cytarabine
MF Methotrexate, Fluorouracil, Leucovorin
MICE Ifosfamide, Carboplatin, Etoposide, Mesna
MINE Mesna, Ifosfamide, Mitoxantrone, Etoposide
mini-BEAM Carmustine, Etoposide, Cytarabine, Melphalan
MOBP Bleomycin, Vincristine, Cisplatin, Mitomycin
MOP Mechlorethamme, Vincristine, Procarbazine
MOPP Mechlorethamme, Vincristine, Procarbazine, Prednisone
MOPP/ABV Mechlorethamme, Vincristine, Procarbazine, Prednisone,
Doxorubicin, Bleomycin, Vinblastine
MP (multiple Melphalan, Prednisone
myeloma)
MP (prostate cancer) Mitoxantrone, Prednisone
MTX/6-MO Methotrexate, Mercaptopurine
MTX/6-MP/VP Methotrexate, Mercaptopurine, Vincristine, Prednisone
MTX-CDDPAdr Methotrexate, Leucovorin, Cisplatin, Doxorubicin
MV (breast cancer) Mitomycin, Vinblastine
MV (acute myelocytic Mitoxantrone, Etoposide
leukemia)
M-VAC Methotrexate Vinblastine, Doxorubicin, Cisplatin
MVP Mitomycin Vinblastine, Cisplatin
MVPP Mechlorethamme, Vinblastine, Procarbazine, Prednisone
NFL Mitoxantrone, Fluorouracil, Leucovorin
NOVP Mitoxantrone, Vinblastine, Vincristine
OPA Vincristine, Prednisone, Doxorubicin
Name Therapeutic agents
OPPA Add Procarbazine to OPA.
PAC Cisplatin, Doxorubicin
PAC-I Cisplatin, Doxorubicin, Cyclophosphamide
PA-CI Cisplatin, Doxorubicin
PC Paclitaxel, Carboplatin or Paclitaxel, Cisplatin
PCV Lomustine, Procarbazine, Vincristine
PE Paclitaxel, Estramustine
PFL Cisplatin, Fluorouracil, Leucovorin
POC Prednisone, Vincristine, Lomustine
ProMACE Prednisone, Methotrexate, Leucovorin, Doxorubicin,
Cyclophosphamide, Etoposide
ProMACE/cytaBOM Prednisone, Doxorubicin, Cyclophosphamide, Etoposide,
Cytarabine, Bleomycin, Vincristine, Methotrexate, Leucovorin, Cotrimoxazole
PRoMACE/MOPP Prednisone, Doxorubicin, Cyclophosphamide, Etoposide,
Mechlorethamme, Vincristine, Procarbazine, Methotrexate, Leucovorin
Pt/VM Cisplatin, Teniposide
PVA Prednisone, Vincristine, Asparaginase
PVB Cisplatin, Vinblastine, Bleomycin
PVDA Prednisone, Vincristine, Daunorubicin, Asparaginase
SMF Streptozocin, Mitomycin, Fluorouracil
TAD Mechlorethamme, Doxorubicin, Vinblastine, Vincristine,
Bleomycin, Etoposide, Prednisone
TCF Paclitaxel, Cisplatin, Fluorouracil
TIP Paclitaxel, Ifosfamide, Mesna, Cisplatin
TTT Methotrexate, Cytarabine, Hydrocortisone
Topo/CTX Cyclophosphamide, Topotecan, Mesna
VAB-6 Cyclophosphamide, Dactinomycin, Vinblastine, Cisplatin,
Bleomycin
VAC Vincristine, Dactinomycin, Cyclophosphamide
Name Therapeutic agents
VACAdr Vincristine, Cyclophosphamide, Doxorubicin,
Dactinomycin, Vincristine
VAD Vincristine, Doxorubicin, Dexamethasone
VATH Vinblastine, Doxorubicin, Thiotepa, Flouxymesterone
VBAP Vincristine, Carmustine, Doxorubicin, Prednisone
VBCMP Vincristine, Carmustine, Melphalan, Cyclophosphamide,
Prednisone
VC Vinorelbine, Cisplatin
VCAP Vincristine, Cyclophosphamide, Doxorubicin, Prednisone
VD Vinorelbine, Doxorubicin
VelP Vinblastine, Cisplatin, Ifosfamide, Mesna
VIP Etoposide, Cisplatin, Ifosfamide, Mesna
VM Mitomycin, Vinblastine
VMCP Vincristine, Melphalan, Cyclophosphamide, Prednisone
VP Etoposide, Cisplatin
V-TAD Etoposide, Thioguanine, Daunorubicin, Cytarabine
5 + 2 Cytarabine, Daunorubicin, Mitoxantrone
7 + 3 Cytarabine with/, Daunorubicin or Idarubicin or
Mitoxantrone
"8 in 1" Methylprednisolone, Vincristine, Lomustine,
Procarbazine, Hydroxyurea, Cisplatin, Cytarabine, Dacarbazine
The proliferation of cancer cells requires lipid synthesis. Normally, acetyl- coA used for lipid synthesis is formed from a mitochondrial pool of pyruvate that is derived from glycolysis. Yet under hypoxic conditions, such as those normally found in a tumor environment, the conversion of pyruvate to acetyl-coA within the mitochondria is downregulated. Recent studies from Metallo et al. (2011) and Mullen et al. (2011) revealed that under such hypoxic conditions, cells instead largely switch to using a pathway involving the reductive carboxylation of alpha-ketoglutarate to make acetyl-coA for lipid synthesis. The first step in this pathway involves converting glutamine to glutamate via glutaminase enzymes. Subsequently, glutamate is
converting to alpha-ketoglutarate, and the resulting alpha-ketoglutarate is converted to isocitrate in a reductive carboxylation step mediated by the isocitrate dehydrogenase enzymes. A switch to this reductive carboxylation pathway also occurs in some renal carcinoma cell lines that contain either impaired mitochondria or an impaired signal for induction of the enzyme responsible for converting glycolytic pyruvate to acetyl- coA (Mullen et al 2011). A similar switch occurs in cells exposed to mitochondrial respiratory chain inhibitors such as metformin, rotenone, and antimycin (Mullen at al. 2011). Therefore, in some embodiments of this invention, we propose using combinations of mitochondrial respiratory chain inhibitors and glutaminase inhibitors to simultaneously increase cancer cells' dependence on glutaminase-dependent pathways for lipid synthesis while inhibiting those very pathways.
The increased dependence on glycolysis in tumor cells is likely because the hypoxic tumor environment impairs mitochondrial respiration. Furthermore, depletion of glucose induces apoptosis in cells transformed with the MYC oncogene. These findings suggest that inhibiting glycolysis would have a therapeutic value in preventing cancer cell proliferation. There are currently many documented glycolytic inhibitors (Pelicano et al. 2006). However, as pointed out by Zhao et al. (2012), "available glycolytic inhibitors are generally not very potent, and high doses are required, which may cause high levels of systemic toxicity." Since cancer cells typically use both glucose and glutamine at higher levels than normal cells, impairing utilization of each of those metabolites will likely have a synergistic effect.
Therefore, in some embodiments of this invention, we propose using combinations of glycolytic pathway inhibitors and glutaminase inhibitors. Such glycolytic inhibitors include 2-deoxyglucose, lonidamine, 3-bromopyruvate, imatinib, oxythiamine, rapamycin, and their pharmacological equivalents. Glycolysis can be inhibited indirectly by depleting NAD+ via DNA damage induced by DNA alkylating agents through a pathway activated by poly(ADP-ribose) polymerase (Zong et al. 2004). Therefore, in some embodiments of this invention, we propose using a combination of DNA alkylating agents and glutaminase inhibitors. Cancer cells use the pentose phosphate pathway along with the glycolytic pathway to create metabolic
intermediates derived from glucose. Therefore, in some embodiments of this
invention, we propose using a combination of pentose phosphate inhibitors such as 6- aminonicotinamide along with glutaminase inhibitors.
In certain embodiments, a compound of the invention may be conjointly administered with non-chemical methods of cancer treatment. In certain
embodiments, a compound of the invention may be conjointly administered with radiation therapy. In certain embodiments, a compound of the invention may be conjointly administered with surgery, with thermoablation, with focused ultrasound therapy, with cryotherapy, or with any combination of these.
In certain embodiments, different compounds of the invention may be conjointly administered with one or more other compounds of the invention.
Moreover, such combinations may be conjointly administered with other therapeutic agents, such as other agents suitable for the treatment of cancer, immunological or neurological diseases, such as the agents identified above. In certain embodiments, conjointly administering one or more additional chemotherapeutic agents with a compound of the invention provides a synergistic effect, such as shown in Figure 18. In certain embodiments, conjointly administering one or more additional
chemotherapeutics agents provides an additive effect.
In certain embodiments, the present invention provides a kit comprising: a) one or more single dosage forms of a compound of the invention; b) one or more single dosage forms of a chemotherapeutic agent as mentioned above; and c) instructions for the administration of the compound of the invention and the chemotherapeutic agent for the treatment of cancer, wherein the cancer is selected from breast cancer, colorectal cancer, endocrine cancer, lung cancer, melanoma, mesothelioma, renal cancer and B cell malignancy.
The present invention provides a kit comprising:
a) a pharmaceutical formulation (e.g., one or more single dosage forms)
comprising a compound of the invention; and
b) instructions for the administration of the pharmaceutical formulation, e.g., for treating or preventing cancer, such as breast cancer, colorectal cancer, endocrine cancer, lung cancer, melanoma, mesothelioma, renal cancer or B cell malignancy.
The present invention provides a kit comprising:
a) a pharmaceutical formulation (e.g., one or more single dosage forms) comprising a compound of the invention; and
b) instructions for the administration of the pharmaceutical formulation, e.g., for treating or preventing breast cancer, wherein the breast cancer comprises basal-type breast cancer cells, triple-negative breast cancer cells, or claudin- low breast cancer cells .
In certain embodiments, the kit further comprises instructions for the administration of the pharmaceutical formulation comprising a compound of the invention conjointly with a chemotherapeutic agent as mentioned above. In certain embodiments, the kit further comprises a second pharmaceutical formulation (e.g., as one or more single dosage forms) comprising a chemotherapeutic agent as mentioned above.
Both dependency on exogenous glutamine and the expression profile of high glutaminase (GLS) and low glutamine synthetase (GS) levels have been shown to correlate with a cancer cell's sensitivity to glutaminase inhibition. Utilizing this information, one may theorize that the amount of metabolic metabolites within a cancer cell may be used as a way to predict its sensitivity to glutaminase inhibition. Testing out this theory, glutamate and glutamine levels were determined in TNBC cells previously shown to be glutamine dependent and sensitive to glutaminase inhibition (Figure 9). Concentrations of glutamate and glutamine were determined by liquid chromatography tandem spectrometry (LC-MS/MS); however, any method of determining metabolite concentrations could be utilized. The cells with
glutamate: glutamine ratios greater than or equal to 1.5 did appear to be sensitive to glutaminase inhibition. The correlation was even stronger when the
glutamate: glutamine ratio was greater than or equal to 2. This result provides a means to identify cancer patients that may benefit from treatment with a glutaminase inhibitor.
Analysis of several primary tumor xenografts show that expression and metabolite correlation extends to other tumor types, such as lung and mesothelioma, in addition to those cancers previously discussed (Figure 10). Xenograft studies using HCT116 colon carcinoma cells (Figure 11) and H2122 lung adenocarcinoma cells (Figure 12) show that treatment with a glutaminase inhibitor described herein resulted in reduced tumor size.
In certain embodiments, the invention provides a method of identifying a cancer patient that may benefit from treatment with a glutaminase inhibitor comprising determining the ratio of glutamate to glutamine in cancer cells of the cancer patient, wherein a ratio greater than or equal to 1.5, such as greater than or equal to 1.6, greater than or equal to 1.7, greater than or equal to 1.8, greater than or equal to 1.9, or greater than or equal to 2.0, indicates the patient may benefit from treatment with a glutaminase inhibitor. In certain such embodiments, the method of determining the ratio includes measuring the amounts of glutamate and glutamine in the cancer cells of the cancer patient. In certain embodiments, the ratio is greater than or equal to 2.0. In certain embodiments of the foregoing, the glutaminase inhibitor is a compound described herein (e.g., a compound of formula I or la). In certain embodiments, the cancer is selected from B cell malignancy, breast cancer, colorectal cancer, endocrine cancer, lung cancer, melanoma, mesothelioma and renal cancer.
In certain embodiments, the invention provides a method of treating a cancer patient comprising 1) determining the ratio of glutamate to glutamine in cancer cells of the cancer patient; and 2) if the ratio of glutamate to glutamine is greater than or equal to 1.5, such as greater than or equal to 1.6, greater than or equal to 1.7, greater than or equal to 1.8, greater than or equal to 1.9, or greater than or equal to 2.0, treating the patient with a compound of formula I or la. In certain such embodiments, the method of determining the ratio includes measuring the amounts of glutamate and glutamine in the cancer cells of the cancer patient. In certain embodiments, the ratio of glutamate to glutamine is greater than or equal to 2.0. In certain embodiments, the cancer is selected from B cell malignancy, breast cancer, colorectal cancer, endocrine cancer, lung cancer, melanoma, mesothelioma and renal cancer.
As mentioned above, high glutaminase (GLS) and low glutamine synthetase
(GS) expression levels have been shown to correlate with a cancer cell's sensitivity to glutaminase inhibition. One may therefore theorize that the levels of GLS and GS within a cancer cell could be used as a way to predict its sensitivity to glutaminase inhibition. Testing out this theory, GLS (both KGA and GAC) levels and GS levels were determined in TNBC cells, which are known to be more sensitive to glutaminase inhibition, and in HR+ or Her2+ cells, which known to be less sensitive to
glutaminase inhibition (Figure 14 and Table 7).
Correlations were observed between glutaminase inhibitor sensitivity and expression of the GAC isoform of GLS. Cells expressing GAC appeared to be more sensitive to glutaminase inhibition. Thus, a cell with a detectable level of GAC would be sensitive to a glutaminase inhibitor such as the compounds described herein. A correlation was also observed for cells expressing a level of GAC that is equal or higher than KGA. Accordingly, in certain embodiments, the invention provides a method of identifying a cancer patient that may benefit from treatment with a glutaminase inhibitor, comprising determining the level of GAC and KGA expression in a cancer cell of the cancer patient, wherein an expression level of GAC is greater than, or equal to the expression level of KGA, indicates that the patient may benefit from treatment with a glutaminase inhibitor.
Significant correlations were observed between glutaminase inhibitor sensitivity and the ratio of GAC:GS. Cells with a GAC:GS ratios greater than or equal to 0.05 appeared to be sensitive to glutaminase inhibition. The correlation was even stronger for cells having a GAC:GS ratio greater than or equal to 1. This result provides a means to identify cancer patients that may benefit from treatment with a glutaminase inhibitor.
In certain embodiments, the invention provides a method of identifying a cancer patient that may benefit from treatment with a glutaminase inhibitor, comprising determining the ratio of glutaminase to glutamine synthetase in cancer cells of the cancer patient, wherein a ratio greater than or equal to 0.05, such as greater than or equal to 0.06, greater than or equal to 0.07, greater than or equal to 0.08, greater than or equal to 0.9, or greater than or equal to 1.0, indicates the patient may benefit from treatment with a glutaminase inhibitor. In certain such
embodiments, the method of determining the ratio includes measuring the levels of glutaminase and glutamine synthetase in the cancer cells of the cancer patient. In certain embodiments, the ratio is greater than or equal to 1. In certain embodiments of the foregoing, the glutaminase inhibitor is a compound described herein (e.g., a compound of formula I or la). In certain embodiments, the glutaminase is both KGA and GAC. In certain embodiments, the glutaminase is KGA. In prefered
embodiments, the glutaminase is GAC.
In certain embodiments, the invention provides a method of treating a cancer patient comprising 1) determining the ratio of glutaminase to glutamine synthetase in
cancer cells of the cancer patient; and 2) if the ratio of glutaminase to glutamine synthetase is greater than or equal to 0.05, such as greater than or equal to 0.06, greater than or equal to 0.07, greater than or equal to 0.08, greater than or equal to 0.9, or greater than or equal to 1.0, indicates the patient may benefit from treatment with a glutaminase inhibitor. In certain such embodiments, the method of determining the ratio includes measuring the amounts of glutaminase and glutamine synthetase in the cancer cells of the cancer patient. In certain embodiments, the ratio is greater than or equal to 1. In certain embodiments, the glutaminase is both KGA and GAC. In certain embodiments, the glutaminase is KGA. In prefered embodiments, the glutaminase is GAC.
In certain embodiments, the cancer is selected from B cell malignancy, breast cancer, colorectal cancer, endocrine cancer, lung cancer, melanoma, mesothelioma and renal cancer.
The level of a GLS (e.g., KGA and/or GAC) and GS can be measured using any suitable method. Some methods involve measuring protein levels, and others involve measuring levels of mR A.
Protein amounts can be measured using antibodies. Antibodies suitable for use in the methods disclosed herein are commercially available, or can be prepared routinely. Methods for preparing and using antibodies in assays for proteins of interest are conventional, and are described in, for example, Green et al., Production of Polyclonal Antisera, in Immunochemical Protocols (Manson, ed.), (Humana Press 1992); Coligan et al, in Current Protocols in Immunology, Sec. 2.4.1 (1992); Kohler & Milstein (1975), Nature 256, 495; Coligan et al, sections 2.5.1-2.6.7; and Harlow et al., Antibodies: A Laboratory Manual, page 726 (Cold Spring Harbor Laboratory Pub. 1988).
Any of a variety of antibodies can be used in methods of the invention. Such antibodies include, for example, polyclonal, monoclonal (mAbs), recombinant, humanized or partially humanized, single chain, Fab, and fragments thereof. The antibodies can be of any isotype, e.g., IgM, various IgG isotypes such as IgGl, IgG2a, etc., and they can be from any animal species that produces antibodies, including goat, rabbit, mouse, chicken or the like. The term "an antibody specific for" a protein means that the antibody recognizes a defined sequence of amino acids, or epitope, in the protein, and binds selectively to the protein and not generally to proteins
unintended for binding to the antibody. The parameters required to achieve specific binding can be determined routinely, using conventional methods in the art.
In some embodiments of the invention, antibodies specific for KGA, GAC and/or GS are immobilized on a surface (e.g., are reactive elements on an array, such as a microarray, or are on another surface, such as used for surface plasmon resonance (SPR)-based technology, such as Biacore), and proteins in the sample are detected by virtue of their ability to bind specifically to the antibodies. Alternatively, proteins in the sample can be immobilized on a surface, and detected by virtue of their ability to bind specifically to the antibodies. Methods of preparing the surfaces and performing the analyses, including conditions effective for specific binding, are conventional and well-known in the art.
Among the many types of suitable immunoassays are immunohistochemical staining, ELISA, Western blot (immunoblot), immunoprecipitation,
radioimmunoassay (RIA), fluorescence-activated cell sorting (FACS), etc. Assays used in methods of the invention can be based on colorimetric readouts, fluorescent readouts, mass spectroscopy, visual inspection, etc.
As mentioned above, expression levels of GLS (KGA and/or GAC) and GS can be measured by measuring mRNA amounts. The amount of an mRNA encoding a KGA, GAC and/or GS can be measured using any suitable method. Examples of such methods include, for example, reverse transcriptase-polymerase chain reaction (RT- PCR), including real time PCR, microarray analysis, nanostring, Northern blot analysis, differential hybridization, and ribonuclease protection assay. Such methods are well-known in the art and are described in, for example, Sambrook et al.,
Molecular Cloning: A Laboratory Manual, current edition, Cold Spring Harbor Laboratory, Cold Spring Harbor, N.Y., and Ausubel et al, Current Protocols in Molecular Biology, John Wiley & sons, New York, N.Y.
In some embodiments of the invention, a histological sample is obtained from a subject (e.g., from a tumor biopsy), using any method known in the art, and include, but are not limited to, tissue section, needle biopsy, and the like. Frequently the sample will be a "clinical sample", which is a sample derived from a patient, including sections of tissues such as frozen sections or paraffin sections taken for histological purposes. The sample can also be derived from supernatants (of cells) or the cells themselves from cell cultures, cells from tissue culture and other media.
Protein or mRNA is then obtained brom the sample, and used to quantitate the amounts of GLS (KGA and/or GAC) and GS.
An alternative way of viewing the correlation between glutaminase activity and sensitivity to glutaminase inhibitor is shown in Figure 16, wherein a glutaminase activity of 0.005 μmol/min/mg of protein predicts sensitivity to glutaminase inhibitor. In certain embodiments, the invention provides a method of identifying a cancer patient that may benefit from treatment with a glutaminase inhibitor comprising determining glutaminase activity in cancer cells of the cancer patient, wherein an activity greater than or equal to 0.005 μmol/min/mg of protein, such as greater than or equal to 0.006 μmol/min/mg of protein, greater than or equal to 0.007 μmol/min/mg of protein, greater than or equal to 0.008 μmol/min/mg of protein, greater than or equal to 0.009 μmol/min/mg of protein, or greater than or equal to 0.010
μmol/min/mg of protein, indicates the patient may benefit from treatment with a glutaminase inhibitor. In certain such embodiments, the method of determining the glutaminase activity includes measuring the glutaminase activity in the cancer cells of the cancer patient. In certain embodiments, the glutaminase activity is greater than or equal to 0.010. In certain embodiments of the foregoing, the glutaminase inhibitor is a compound described herein (e.g., a compound of formula I or la). In certain embodiments, the cancer is selected from B cell malignancy, breast cancer, colorectal cancer, endocrine cancer, lung, melanoma, mesothelioma and renal cancer.
In certain embodiments, the invention provides a method of treating a cancer patient comprising 1) determining glutaminase activity in cancer cells of the cancer patient; and 2) and wherein an activity greater than or equal to 0.005 μmol/min/mg of protein, such as greater than or equal to 0.006 μmol/min/mg of protein, greater than or equal to 0.007 μmol/min/mg of protein, greater than or equal to 0.008 μmol/min/mg of protein, greater than or equal to 0.009 μmol/min/mg of protein, or greater than or equal to 0.010 μmol/min/mg of protein, treating the patient with a compound of formula I or la. In certain such embodiments, the method of determining determining glutaminase activity in the cancer cells of the cancer patient. In certain embodiments, the ratio of glutamate to glutamine is greater than or equal to 2.0. In certain embodiments, the cancer is selected from B cell malignancy, breast cancer, colorectal cancer, endocrine cancer, lung, melanoma, mesothelioma and renal cancer.
The disclosure also provides kits for detecting whether a subject having a cancer is likely to be responsive to glutaminase inhibitors. The kit may include one or more agents for detecting the amount of expression of a protein of the invention [e.g., the amount of the protein, and/or the amount of a nucleic acid (e.g., an mRNA) encoding the protein]. The agents in the kit can encompass, for example, antibodies specific for the proteins, or probes specific for the mRNA that can be used to hybridize to the RNA (or to a cDNA generated from it) or to perform RT-PCR. The kit may also include additional agents suitable for detecting, measuring and/or quantitating the amount of protein or nucleic acid. Among other uses, kits of the invention can be used in experimental applications. A skilled worker will recognize components of kits suitable for carrying out a method of the invention.
Optionally, a kit of the invention may comprise instructions for performing the method. Optional elements of a kit of the invention include suitable buffers, containers, or packaging materials. The reagents of the kit may be in containers in which the reagents are stable, e.g., in lyophilized form or stabilized liquids. The reagents may also be in single use form, e.g., for the performance of an assay for a single subject.
Definitions
The term "acyl" is art-recognized and refers to a group represented by the general formula hydrocarbylC(O)-, preferably alkylC(O)-.
The term "acylamino" is art-recognized and refers to an amino group substituted with an acyl group and may be represented, for example, by the formula hydrocarbylC(0)NH-.
The term "acyloxy" is art-recognized and refers to a group represented by the general formula hydrocarbylC(0)0-, preferably alkylC(0)0-.
The term "alkoxy" refers to an alkyl group, preferably a lower alkyl group, having an oxygen attached thereto. Representative alkoxy groups include methoxy, ethoxy, propoxy, tert-butoxy and the like.
The term "alkoxyalkyl" refers to an alkyl group substituted with an alkoxy group and may be represented by the general formula alkyl-O-alkyl.
The term "alkenyl", as used herein, refers to an aliphatic group containing at least one double bond and is intended to include both "unsubstituted alkenyls" and "substituted alkenyls", the latter of which refers to alkenyl moieties having
substituents replacing a hydrogen on one or more carbons of the alkenyl group. Such substituents may occur on one or more carbons that are included or not included in one or more double bonds. Moreover, such substituents include all those
contemplated for alkyl groups, as discussed below, except where stability is prohibitive. For example, substitution of alkenyl groups by one or more alkyl, carbocyclyl, aryl, heterocyclyl, or heteroaryl groups is contemplated.
An "alkyl" group or "alkane" is a straight chained or branched non-aromatic hydrocarbon which is completely saturated. Typically, a straight chained or branched alkyl group has from 1 to about 20 carbon atoms, preferably from 1 to about 10 unless otherwise defined. Examples of straight chained and branched alkyl groups include methyl, ethyl, n-propyl, iso-propyl, n-butyl, sec-butyl, tert-butyl, pentyl, hexyl, pentyl and octyl. A Ci-C6 straight chained or branched alkyl group is also referred to as a "lower alkyl" group.
Moreover, the term "alkyl" (or "lower alkyl") as used throughout the specification, examples, and claims is intended to include both "unsubstituted alkyls" and "substituted alkyls", the latter of which refers to alkyl moieties having
substituents replacing a hydrogen on one or more carbons of the hydrocarbon backbone. Such substituents, if not otherwise specified, can include, for example, a halogen, a hydroxyl, a carbonyl (such as a carboxyl, an alkoxycarbonyl, a formyl, or an acyl), a thiocarbonyl (such as a thioester, a thioacetate, or a thioformate), an alkoxyl, a phosphoryl, a phosphate, a phosphonate, a phosphinate, an amino, an amido, an amidine, an imine, a cyano, a nitro, an azido, a sulfhydryl, an alkylthio, a sulfate, a sulfonate, a sulfamoyl, a sulfonamido, a sulfonyl, a heterocyclyl, an aralkyl, or an aromatic or heteroaromatic moiety. It will be understood by those skilled in the art that the moieties substituted on the hydrocarbon chain can themselves be substituted, if appropriate. For instance, the substituents of a substituted alkyl may include substituted and unsubstituted forms of amino, azido, imino, amido, phosphoryl (including phosphonate and phosphinate), sulfonyl (including sulfate, sulfonamido, sulfamoyl and sulfonate), and silyl groups, as well as ethers, alkylthios, carbonyls (including ketones, aldehydes, carboxylates, and esters), -CF3, -CN and the like. Exemplary substituted alkyls are described below. Cycloalkyls can be further substituted with alkyls, alkenyls, alkoxys, alkylthios, aminoalkyls, carbonyl- substituted alkyls, -CF3, -CN, and the like.
The term "Cx_y" when used in conjunction with a chemical moiety, such as, acyl, acyloxy, alkyl, alkenyl, alkynyl, or alkoxy is meant to include groups that contain from x to y carbons in the chain. For example, the term "Cx_yalkyl" refers to substituted or unsubstituted saturated hydrocarbon groups, including straight-chain alkyl and branched-chain alkyl groups that contain from x to y carbons in the chain, including haloalkyl groups such as trifluoromethyl and 2,2,2-tirfluoroethyl, etc. Co alkyl indicates a hydrogen where the group is in a terminal position, a bond if internal. The terms "C2-yalkenyl" and "C2_yalkynyl" refer to substituted or unsubstituted unsaturated aliphatic groups analogous in length and possible substitution to the alkyls described above, but that contain at least one double or triple bond respectively.
The term "alkylamino", as used herein, refers to an amino group substituted with at least one alkyl group.
The term "alkylthio", as used herein, refers to a thiol group substituted with an alkyl group and may be represented by the general formula alkylS-.
The term "alkynyl", as used herein, refers to an aliphatic group containing at least one triple bond and is intended to include both "unsubstituted alkynyls" and "substituted alkynyls", the latter of which refers to alkynyl moieties having
substituents replacing a hydrogen on one or more carbons of the alkynyl group. Such substituents may occur on one or more carbons that are included or not included in one or more triple bonds. Moreover, such substituents include all those contemplated for alkyl groups, as discussed above, except where stability is prohibitive. For example, substitution of alkynyl groups by one or more alkyl, carbocyclyl, aryl, heterocyclyl, or heteroaryl groups is contemplated.
The term "amide", as used herein, refers to a group
wherein each R independently represent a hydrogen or hydrocarbyl group, or two R10 are taken together with the N atom to which they are attached complete a heterocycle having from 4 to 8 atoms in the ring structure.
The terms "amine" and "amino" are art-recognized and refer to both unsubstituted and substituted amines and salts thereof, e.g., a moiety that can be represented by
wherein each R10 independently represents a hydrogen or a hydrocarbyl group, or two R10 are taken together with the N atom to which they are attached complete a heterocycle having from 4 to 8 atoms in the ring structure.
The term "aminoalkyl", as used herein, refers to an alkyl group substituted with an amino group.
The term "aralkyl", as used herein, refers to an alkyl group substituted with an aryl group.
The term "aryl" as used herein include substituted or unsubstituted single-ring aromatic groups in which each atom of the ring is carbon. Preferably the ring is a 5- to 7-membered ring, more preferably a 6-membered ring. The term "aryl" also includes polycyclic ring systems having two or more cyclic rings in which two or more carbons are common to two adjoining rings wherein at least one of the rings is aromatic, e.g., the other cyclic rings can be cycloalkyls, cycloalkenyls, cycloalkynyls, aryls, heteroaryls, and/or heterocyclyls. Aryl groups include benzene, naphthalene, phenanthrene, phenol, aniline, and the like.
The term "carbamate" is art-recognized and refers to a group
wherein R9 and R10 independently represent hydrogen or a hydrocarbyl group, such as an alkyl group, or R9 and R10 taken together with the intervening atom(s) complete a heterocycle having from 4 to 8 atoms in the ring structure.
The terms "carbocycle", and "carbocyclic", as used herein, refers to a saturated or unsaturated ring in which each atom of the ring is carbon. The term carbocycle includes both aromatic carbocycles and non-aromatic carbocycles. Non- aromatic carbocycles include both cycloalkane rings, in which all carbon atoms are saturated, and cycloalkene rings, which contain at least one double bond.
"Carbocycle" includes 5-7 membered monocyclic and 8-12 membered bicyclic rings.
Each ring of a bicyclic carbocycle may be selected from saturated, unsaturated and aromatic rings. Carbocycle includes bicyclic molecules in which one, two or three or
more atoms are shared between the two rings. The term "fused carbocycle" refers to a bicyclic carbocycle in which each of the rings shares two adjacent atoms with the other ring. Each ring of a fused carbocycle may be selected from saturated, unsaturated and aromatic rings. In an exemplary embodiment, an aromatic ring, e.g., phenyl, may be fused to a saturated or unsaturated ring, e.g., cyclohexane,
cyclopentane, or cyclohexene. Any combination of saturated, unsaturated and aromatic bicyclic rings, as valence permits, is included in the definition of
carbocyclic. Exemplary "carbocycles" include cyclopentane, cyclohexane, bicyclo[2.2.1 Jheptane, 1 ,5-cyclooctadiene, 1 ,2,3,4-tetrahydronaphthalene,
bicyclo[4.2.0]oct-3-ene, naphthalene and adamantane. Exemplary fused carbocycles include decalin, naphthalene, 1,2,3,4-tetrahydronaphthalene, bicyclo[4.2.0]octane, 4,5,6,7-tetrahydro-lH-indene and bicyclo[4.1.0]hept-3-ene. "Carbocycles" may be susbstituted at any one or more positions capable of bearing a hydrogen atom.
A "cycloalkyl" group is a cyclic hydrocarbon which is completely saturated. "Cycloalkyl" includes monocyclic and bicyclic rings. Typically, a monocyclic cycloalkyl group has from 3 to about 10 carbon atoms, more typically 3 to 8 carbon atoms unless otherwise defined. The second ring of a bicyclic cycloalkyl may be selected from saturated, unsaturated and aromatic rings. Cycloalkyl includes bicyclic molecules in which one, two or three or more atoms are shared between the two rings. The term "fused cycloalkyl" refers to a bicyclic cycloalkyl in which each of the rings shares two adjacent atoms with the other ring. The second ring of a fused bicyclic cycloalkyl may be selected from saturated, unsaturated and aromatic rings. A
"cycloalkenyl" group is a cyclic hydrocarbon containing one or more double bonds.
The term "carbocyclylalkyl", as used herein, refers to an alkyl group substituted with a carbocycle group.
The term "carbonate" is art-recognized and refers to a group -OCO2-R10, wherein R10 represents a hydrocarbyl group.
The term "carboxy", as used herein, refers to a group represented by the formula -C02H.
The term "ester", as used herein, refers to a group -C(0)OR10 wherein R10 represents a hydrocarbyl group.
The term "ether", as used herein, refers to a hydrocarbyl group linked through an oxygen to another hydrocarbyl group. Accordingly, an ether substituent of a
hydrocarbyl group may be hydrocarbyl-O-. Ethers may be either symmetrical or unsymmetrical. Examples of ethers include, but are not limited to, heterocycle-O- heterocycle and aryl-O-heterocycle. Ethers include "alkoxyalkyl" groups, which may be represented by the general formula alkyl-O-alkyl.
The terms "halo" and "halogen" as used herein means halogen and includes chloro, fluoro, bromo, and iodo.
The terms "hetaralkyl" and "heteroaralkyl", as used herein, refers to an alkyl group substituted with a hetaryl group.
The term "heteroalkyl", as used herein, refers to a saturated or unsaturated chain of carbon atoms and at least one heteroatom, wherein no two heteroatoms are adjacent.
The terms "heteroaryl" and "hetaryl" include substituted or unsubstituted aromatic single ring structures, preferably 5- to 7-membered rings, more preferably 5- to 6-membered rings, whose ring structures include at least one heteroatom, preferably one to four heteroatoms, more preferably one or two heteroatoms. The terms "heteroaryl" and "hetaryl" also include polycyclic ring systems having two or more cyclic rings in which two or more carbons are common to two adjoining rings wherein at least one of the rings is heteroaromatic, e.g., the other cyclic rings can be cycloalkyls, cycloalkenyls, cycloalkynyls, aryls, heteroaryls, and/or heterocyclyls. Heteroaryl groups include, for example, pyrrole, furan, thiophene, imidazole, oxazole, thiazole, pyrazole, pyridine, pyrazine, pyridazine, and pyrimidine, and the like.
The term "heteroatom" as used herein means an atom of any element other than carbon or hydrogen. Preferred heteroatoms are nitrogen, oxygen, and sulfur.
The terms "heterocyclyl", "heterocycle", and "heterocyclic" refer to substituted or unsubstituted non-aromatic ring structures, preferably 3- to 10- membered rings, more preferably 3- to 7-membered rings, whose ring structures include at least one heteroatom, preferably one to four heteroatoms, more preferably one or two heteroatoms. The terms "heterocyclyl" and "heterocyclic" also include polycyclic ring systems having two or more cyclic rings in which two or more carbons are common to two adjoining rings wherein at least one of the rings is heterocyclic, e.g., the other cyclic rings can be cycloalkyls, cycloalkenyls,
cycloalkynyls, aryls, heteroaryls, and/or heterocyclyls. Heterocyclyl groups include,
for example, piperidine, piperazine, pyrrolidine, morpholine, lactones, lactams, and the like.
The term "heterocyclylalkyl", as used herein, refers to an alkyl group substituted with a heterocycle group.
The term "hydrocarbyl", as used herein, refers to a group that is bonded through a carbon atom that does not have a =0 or =S substituent, and typically has at least one carbon-hydrogen bond and a primarily carbon backbone, but may optionally include heteroatoms. Thus, groups like methyl, ethoxy ethyl, 2-pyridyl, and trifluoromethyl are considered to be hydrocarbyl for the purposes of this application, but substituents such as acetyl (which has a =0 substituent on the linking carbon) and ethoxy (which is linked through oxygen, not carbon) are not. Hydrocarbyl groups include, but are not limited to aryl, heteroaryl, carbocycle, heterocyclyl, alkyl, alkenyl, alkynyl, and combinations thereof.
The term "hydroxyalkyl", as used herein, refers to an alkyl group substituted with a hydroxy group.
The term "lower" when used in conjunction with a chemical moiety, such as, acyl, acyloxy, alkyl, alkenyl, alkynyl, or alkoxy is meant to include groups where there are ten or fewer non-hydrogen atoms in the substituent, preferably six or fewer. A "lower alkyl", for example, refers to an alkyl group that contains ten or fewer carbon atoms, preferably six or fewer. In certain embodiments, acyl, acyloxy, alkyl, alkenyl, alkynyl, or alkoxy substituents defined herein are respectively lower acyl, lower acyloxy, lower alkyl, lower alkenyl, lower alkynyl, or lower alkoxy, whether they appear alone or in combination with other substituents, such as in the recitations hydroxyalkyl and aralkyl (in which case, for example, the atoms within the aryl group are not counted when counting the carbon atoms in the alkyl substituent).
The terms "polycyclyl", "polycycle", and "polycyclic" refer to two or more rings (e.g., cycloalkyls, cycloalkenyls, cycloalkynyls, aryls, heteroaryls, and/or heterocyclyls) in which two or more atoms are common to two adjoining rings, e.g., the rings are "fused rings". Each of the rings of the polycycle can be substituted or unsubstituted. In certain embodiments, each ring of the polycycle contains from 3 to 10 atoms in the ring, preferably from 5 to 7.
The term "silyl" refers to a silicon moiety with three hydrocarbyl moieties attached thereto.
The term "substituted" refers to moieties having substituents replacing a hydrogen on one or more carbons of the backbone. It will be understood that "substitution" or "substituted with" includes the implicit proviso that such substitution is in accordance with permitted valence of the substituted atom and the substituent, and that the substitution results in a stable compound, e.g., which does not spontaneously undergo transformation such as by rearrangement, cyclization, elimination, etc. As used herein, the term "substituted" is contemplated to include all permissible substituents of organic compounds. In a broad aspect, the permissible substituents include acyclic and cyclic, branched and unbranched, carbocyclic and heterocyclic, aromatic and non-aromatic substituents of organic compounds. The permissible substituents can be one or more and the same or different for appropriate organic compounds. For purposes of this invention, the heteroatoms such as nitrogen may have hydrogen substituents and/or any permissible substituents of organic compounds described herein which satisfy the valences of the heteroatoms.
Substituents can include any substituents described herein, for example, a halogen, a hydroxyl, a carbonyl (such as a carboxyl, an alkoxycarbonyl, a formyl, or an acyl), a thiocarbonyl (such as a thioester, a thioacetate, or a thioformate), an alkoxyl, a phosphoryl, a phosphate, a phosphonate, a phosphinate, an amino, an amido, an amidine, an imine, a cyano, a nitro, an azido, a sulfhydryl, an alkylthio, a sulfate, a sulfonate, a sulfamoyl, a sulfonamido, a sulfonyl, a heterocyclyl, an aralkyl, or an aromatic or heteroaromatic moiety. It will be understood by those skilled in the art that substituents can themselves be substituted, if appropriate. Unless specifically stated as "unsubstituted," references to chemical moieties herein are understood to include substituted variants. For example, reference to an "aryl" group or moiety implicitly includes both substituted and unsubstituted variants.
The term "sulfate" is art-recognized and refers to the group -OSO3H, or a pharmaceutically acceptable salt thereof.
The term "sulfonamide" is art-recognized and refers to the group represented by the general formulae
wherein R
9 and R
10 independently represents hydrogen or hydrocarbyl, such as alkyl, or R
9 and R
10 taken together with the intervening atom(s) complete a heterocycle having from 4 to 8 atoms in the ring structure.
The term "sulfoxide" is art-recognized and refers to the group -S(0)-R10, wherein R10 represents a hydrocarbyl.
The term "sulfonate" is art-recognized and refers to the group SO3H, or a pharmaceutically acceptable salt thereof.
The term "sulfone" is art-recognized and refers to the group -S(0)2-R10, wherein R10 represents a hydrocarbyl.
The term "thioalkyl", as used herein, refers to an alkyl group substituted with a thiol group.
The term "thioester", as used herein, refers to a group -C(0)SR10 or -SC(0)R10 wherein R10 represents a hydrocarbyl.
The term "thioether", as used herein, is equivalent to an ether, wherein the oxygen is replaced with a sulfur.
The term "urea" is art-recognized and may be represented by the general formula
R9 R9
wherein R9 and R10 independently represent hydrogen or a hydrocarbyl, such as alkyl, or either occurrence of R9 taken together with R10 and the intervening atom(s) complete a heterocycle having from 4 to 8 atoms in the ring structure.
"Protecting group" refers to a group of atoms that, when attached to a reactive functional group in a molecule, mask, reduce or prevent the reactivity of the functional group. Typically, a protecting group may be selectively removed as desired during the course of a synthesis. Examples of protecting groups can be found in
Greene and Wuts, Protective Groups in Organic Chemistry, 3rd Ed., 1999, John Wiley & Sons, NY and Harrison et al, Compendium of Synthetic Organic Methods, Vols. 1- 8, 1971-1996, John Wiley & Sons, NY. Representative nitrogen protecting groups include, but are not limited to, formyl, acetyl, trifluoroacetyl, benzyl,
benzyloxycarbonyl ("CBZ"), tert-butoxycarbonyl ("Boc"), trimethylsilyl ("TMS"), 2- trimethylsilyl-ethanesulfonyl ("TES"), trityl and substituted trityl groups,
allyloxycarbonyl, 9-fluorenylmethyloxycarbonyl ("FMOC"), nitro- veratryloxycarbonyl ("NVOC") and the like. Representative hydroxylprotecting groups include, but are not limited to, those where the hydroxyl group is either acylated (esterified) or alkylated such as benzyl and trityl ethers, as well as alkyl ethers, tetrahydropyranyl ethers, trialkylsilyl ethers (e.g., TMS or TIPS groups), glycol ethers, such as ethylene glycol and propylene glycol derivatives and allyl ethers.
The term "healthcare providers" refers to individuals or organizations that provide healthcare services to a person, community, etc. Examples of "healthcare providers" include doctors, hospitals, continuing care retirement communities, skilled nursing facilities, subacute care facilities, clinics, multispecialty clinics, freestanding ambulatory centers, home health agencies, and HMO's.
As used herein, a therapeutic that "prevents" a disorder or condition refers to a compound that, in a statistical sample, reduces the occurrence of the disorder or condition in the treated sample relative to an untreated control sample, or delays the onset or reduces the severity of one or more symptoms of the disorder or condition relative to the untreated control sample.
The term "treating" includes prophylactic and/or therapeutic treatments. The term "prophylactic or therapeutic" treatment is art-recognized and includes administration to the host of one or more of the subject compositions. If it is administered prior to clinical manifestation of the unwanted condition (e.g., disease or other unwanted state of the host animal) then the treatment is prophylactic (i.e., it protects the host against developing the unwanted condition), whereas if it is administered after manifestation of the unwanted condition, the treatment is therapeutic, (i.e., it is intended to diminish, ameliorate, or stabilize the existing unwanted condition or side effects thereof).
The term "prodrug" is intended to encompass compounds which, under physiologic conditions, are converted into the therapeutically active agents of the present invention (e.g., a compound of formula I). A common method for making a prodrug is to include one or more selected moieties which are hydro lyzed under physiologic conditions to reveal the desired molecule. In other embodiments, the prodrug is converted by an enzymatic activity of the host animal. For example, esters or carbonates (e.g., esters or carbonates of alcohols or carboxylic acids) are preferred
prodrugs of the present invention. In certain embodiments, some or all of the compounds of formula I in a formulation represented above can be replaced with the corresponding suitable prodrug, e.g., wherein a hydroxyl in the parent compound is presented as an ester or a carbonate or carboxylic acid present in the parent compound is presented as an ester.
Pharmaceutical Compositions
The compositions and methods of the present invention may be utilized to treat an individual in need thereof. In certain embodiments, the individual is a mammal such as a human, or a non-human mammal. When administered to an animal, such as a human, the composition or the compound is preferably administered as a pharmaceutical composition comprising, for example, a compound of the invention and a pharmaceutically acceptable carrier. Pharmaceutically acceptable carriers are well known in the art and include, for example, aqueous solutions such as water or physiologically buffered saline or other solvents or vehicles such as glycols, glycerol, oils such as olive oil, or injectable organic esters. In a preferred
embodiment, when such pharmaceutical compositions are for human administration, particularly for invasive routes of administration (i.e., routes, such as injection or implantation, that circumvent transport or diffusion through an epithelial barrier), the aqueous solution is pyrogen-free, or substantially pyrogen-free. The excipients can be chosen, for example, to effect delayed release of an agent or to selectively target one or more cells, tissues or organs. The pharmaceutical composition can be in dosage unit form such as tablet, capsule (including sprinkle capsule and gelatin capsule), granule, lyophile for reconstitution, powder, solution, syrup, suppository, injection or the like. The composition can also be present in a transdermal delivery system, e.g., a skin patch. The composition can also be present in a solution suitable for topical administration, such as an eye drop.
A pharmaceutically acceptable carrier can contain physiologically acceptable agents that act, for example, to stabilize, increase solubility or to increase the absorption of a compound such as a compound of the invention. Such physiologically acceptable agents include, for example, carbohydrates, such as glucose, sucrose or dextrans, antioxidants, such as ascorbic acid or glutathione, chelating agents, low molecular weight proteins or other stabilizers or excipients. The choice of a pharmaceutically acceptable carrier, including a physiologically acceptable agent,
depends, for example, on the route of administration of the composition. The preparation or pharmaceutical composition can be a selfemulsifying drug delivery system or a selfmicroemulsifying drug delivery system. The pharmaceutical composition (preparation) also can be a liposome or other polymer matrix, which can have incorporated therein, for example, a compound of the invention. Liposomes, for example, which comprise phospholipids or other lipids, are nontoxic, physiologically acceptable and metabolizable carriers that are relatively simple to make and administer.
The phrase "pharmaceutically acceptable" is employed herein to refer to those compounds, materials, compositions, and/or dosage forms which are, within the scope of sound medical judgment, suitable for use in contact with the tissues of human beings and animals without excessive toxicity, irritation, allergic response, or other problem or complication, commensurate with a reasonable benefit/risk ratio.
The phrase "pharmaceutically acceptable carrier" as used herein means a pharmaceutically acceptable material, composition or vehicle, such as a liquid or solid filler, diluent, excipient, solvent or encapsulating material. Each carrier must be "acceptable" in the sense of being compatible with the other ingredients of the formulation and not injurious to the patient. Some examples of materials which can serve as pharmaceutically acceptable carriers include: (1) sugars, such as lactose, glucose and sucrose; (2) starches, such as corn starch and potato starch; (3) cellulose, and its derivatives, such as sodium carboxymethyl cellulose, ethyl cellulose and cellulose acetate; (4) powdered tragacanth; (5) malt; (6) gelatin; (7) talc; (8) excipients, such as cocoa butter and suppository waxes; (9) oils, such as peanut oil, cottonseed oil, safflower oil, sesame oil, olive oil, corn oil and soybean oil; (10) glycols, such as propylene glycol; (11) polyols, such as glycerin, sorbitol, mannitol and polyethylene glycol; (12) esters, such as ethyl oleate and ethyl laurate; (13) agar; (14) buffering agents, such as magnesium hydroxide and aluminum hydroxide; (15) alginic acid; (16) pyrogen- free water; (17) isotonic saline; (18) Ringer's solution; (19) ethyl alcohol; (20) phosphate buffer solutions; and (21) other non-toxic compatible substances employed in pharmaceutical formulations.
A pharmaceutical composition (preparation) can be administered to a subject by any of a number of routes of administration including, for example, orally (for example, drenches as in aqueous or non-aqueous solutions or suspensions, tablets,
capsules (including sprinkle capsules and gelatin capsules), boluses, powders, granules, pastes for application to the tongue); absorption through the oral mucosa (e.g., sublingually); anally, rectally or vaginally (for example, as a pessary, cream or foam); parenterally (including intramuscularly, intravenously, subcutaneously or intrathecally as, for example, a sterile solution or suspension); nasally;
intraperitoneally; subcutaneously; transdermally (for example as a patch applied to the skin); and topically (for example, as a cream, ointment or spray applied to the skin, or as an eye drop). The compound may also be formulated for inhalation. In certain embodiments, a compound may be simply dissolved or suspended in sterile water. Details of appropriate routes of administration and compositions suitable for same can be found in, for example, U.S. Pat. Nos. 6,110,973, 5,763,493, 5,731,000, 5,541,231, 5,427,798, 5,358,970 and 4,172,896, as well as in patents cited therein.
The formulations may conveniently be presented in unit dosage form and may be prepared by any methods well known in the art of pharmacy. The amount of active ingredient which can be combined with a carrier material to produce a single dosage form will vary depending upon the host being treated, the particular mode of administration. The amount of active ingredient that can be combined with a carrier material to produce a single dosage form will generally be that amount of the compound which produces a therapeutic effect. Generally, out of one hundred percent, this amount will range from about 1 percent to about ninety-nine percent of active ingredient, preferably from about 5 percent to about 70 percent, most preferably from about 10 percent to about 30 percent.
Methods of preparing these formulations or compositions include the step of bringing into association an active compound, such as a compound of the invention, with the carrier and, optionally, one or more accessory ingredients. In general, the formulations are prepared by uniformly and intimately bringing into association a compound of the present invention with liquid carriers, or finely divided solid carriers, or both, and then, if necessary, shaping the product.
Formulations of the invention suitable for oral administration may be in the form of capsules (including sprinkle capsules and gelatin capsules), cachets, pills, tablets, lozenges (using a flavored basis, usually sucrose and acacia or tragacanth), lyophile, powders, granules, or as a solution or a suspension in an aqueous or nonaqueous liquid, or as an oil-in- water or water-in-oil liquid emulsion, or as an elixir or
syrup, or as pastilles (using an inert base, such as gelatin and glycerin, or sucrose and acacia) and/or as mouth washes and the like, each containing a predetermined amount of a compound of the present invention as an active ingredient. Compositions or compounds may also be administered as a bolus, electuary or paste.
To prepare solid dosage forms for oral administration (capsules (including sprinkle capsules and gelatin capsules), tablets, pills, dragees, powders, granules and the like), the active ingredient is mixed with one or more pharmaceutically acceptable carriers, such as sodium citrate or dicalcium phosphate, and/or any of the following: (1) fillers or extenders, such as starches, lactose, sucrose, glucose, mannitol, and/or silicic acid; (2) binders, such as, for example, carboxymethylcellulose, alginates, gelatin, polyvinyl pyrrolidone, sucrose and/or acacia; (3) humectants, such as glycerol; (4) disintegrating agents, such as agar-agar, calcium carbonate, potato or tapioca starch, alginic acid, certain silicates, and sodium carbonate; (5) solution retarding agents, such as paraffin; (6) absorption accelerators, such as quaternary ammonium compounds; (7) wetting agents, such as, for example, cetyl alcohol and glycerol monostearate; (8) absorbents, such as kaolin and bentonite clay; (9) lubricants, such a talc, calcium stearate, magnesium stearate, solid polyethylene glycols, sodium lauryl sulfate, and mixtures thereof; (10) complexing agents, such as, modified and unmodified cyclodextrins; and (11) coloring agents. In the case of capsules (including sprinkle capsules and gelatin capsules), tablets and pills, the pharmaceutical compositions may also comprise buffering agents. Solid compositions of a similar type may also be employed as fillers in soft and hard-filled gelatin capsules using such excipients as lactose or milk sugars, as well as high molecular weight polyethylene glycols and the like.
A tablet may be made by compression or molding, optionally with one or more accessory ingredients. Compressed tablets may be prepared using binder (for example, gelatin or hydroxypropylmethyl cellulose), lubricant, inert diluent, preservative, disintegrant (for example, sodium starch glycolate or cross-linked sodium carboxymethyl cellulose), surface-active or dispersing agent. Molded tablets may be made by molding in a suitable machine a mixture of the powdered compound moistened with an inert liquid diluent.
The tablets, and other solid dosage forms of the pharmaceutical compositions, such as dragees, capsules (including sprinkle capsules and gelatin capsules), pills and
granules, may optionally be scored or prepared with coatings and shells, such as enteric coatings and other coatings well known in the pharmaceutical-formulating art. They may also be formulated so as to provide slow or controlled release of the active ingredient therein using, for example, hydroxypropylmethyl cellulose in varying proportions to provide the desired release profile, other polymer matrices, liposomes and/or microspheres. They may be sterilized by, for example, filtration through a bacteria-retaining filter, or by incorporating sterilizing agents in the form of sterile solid compositions that can be dissolved in sterile water, or some other sterile injectable medium immediately before use. These compositions may also optionally contain opacifying agents and may be of a composition that they release the active ingredient(s) only, or preferentially, in a certain portion of the gastrointestinal tract, optionally, in a delayed manner. Examples of embedding compositions that can be used include polymeric substances and waxes. The active ingredient can also be in micro-encapsulated form, if appropriate, with one or more of the above-described excipients.
Liquid dosage forms useful for oral administration include pharmaceutically acceptable emulsions, lyophiles for reconstitution, microemulsions, solutions, suspensions, syrups and elixirs. In addition to the active ingredient, the liquid dosage forms may contain inert diluents commonly used in the art, such as, for example, water or other solvents, cyclodextrins and derivatives thereof, solubilizing agents and emulsifiers, such as ethyl alcohol, isopropyl alcohol, ethyl carbonate, ethyl acetate, benzyl alcohol, benzyl benzoate, propylene glycol, 1,3-butylene glycol, oils (in particular, cottonseed, groundnut, corn, germ, olive, castor and sesame oils), glycerol, tetrahydrofuryl alcohol, polyethylene glycols and fatty acid esters of sorbitan, and mixtures thereof.
Besides inert diluents, the oral compositions can also include adjuvants such as wetting agents, emulsifying and suspending agents, sweetening, flavoring, coloring, perfuming and preservative agents.
Suspensions, in addition to the active compounds, may contain suspending agents as, for example, ethoxylated isostearyl alcohols, polyoxyethylene sorbitol and sorbitan esters, microcrystalline cellulose, aluminum metahydroxide, bentonite, agar- agar and tragacanth, and mixtures thereof.
Formulations of the pharmaceutical compositions for rectal, vaginal, or urethral administration may be presented as a suppository, which may be prepared by mixing one or more active compounds with one or more suitable nonirritating excipients or carriers comprising, for example, cocoa butter, polyethylene glycol, a suppository wax or a salicylate, and which is solid at room temperature, but liquid at body temperature and, therefore, will melt in the rectum or vaginal cavity and release the active compound.
Formulations of the pharmaceutical compositions for administration to the mouth may be presented as a mouthwash, or an oral spray, or an oral ointment.
Alternatively or additionally, compositions can be formulated for delivery via a catheter, stent, wire, or other intraluminal device. Delivery via such devices may be especially useful for delivery to the bladder, urethra, ureter, rectum, or intestine.
Formulations which are suitable for vaginal administration also include pessaries, tampons, creams, gels, pastes, foams or spray formulations containing such carriers as are known in the art to be appropriate.
Dosage forms for the topical or transdermal administration include powders, sprays, ointments, pastes, creams, lotions, gels, solutions, patches and inhalants. The active compound may be mixed under sterile conditions with a pharmaceutically acceptable carrier, and with any preservatives, buffers, or propellants that may be required.
The ointments, pastes, creams and gels may contain, in addition to an active compound, excipients, such as animal and vegetable fats, oils, waxes, paraffins, starch, tragacanth, cellulose derivatives, polyethylene glycols, silicones, bentonites, silicic acid, talc and zinc oxide, or mixtures thereof.
Powders and sprays can contain, in addition to an active compound, excipients such as lactose, talc, silicic acid, aluminum hydroxide, calcium silicates and polyamide powder, or mixtures of these substances. Sprays can additionally contain customary propellants, such as chlorofluorohydrocarbons and volatile unsubstituted hydrocarbons, such as butane and propane.
Transdermal patches have the added advantage of providing controlled delivery of a compound of the present invention to the body. Such dosage forms can be made by dissolving or dispersing the active compound in the proper medium. Absorption enhancers can also be used to increase the flux of the compound across
the skin. The rate of such flux can be controlled by either providing a rate controlling membrane or dispersing the compound in a polymer matrix or gel.
Ophthalmic formulations, eye ointments, powders, solutions and the like, are also contemplated as being within the scope of this invention. Exemplary ophthalmic formulations are described in U.S. Publication Nos. 2005/0080056, 2005/0059744, 2005/0031697 and 2005/004074 and U.S. Patent No. 6,583,124, the contents of which are incorporated herein by reference. If desired, liquid ophthalmic formulations have properties similar to that of lacrimal fluids, aqueous humor or vitreous humor or are compatable with such fluids. A preferred route of administration is local
administration (e.g., topical administration, such as eye drops, or administration via an implant).
The phrases "parenteral administration" and "administered parenterally" as used herein means modes of administration other than enteral and topical
administration, usually by injection, and includes, without limitation, intravenous, intramuscular, intraarterial, intrathecal, intracapsular, intraorbital, intracardiac, intradermal, intraperitoneal, transtracheal, subcutaneous, subcuticular, intraarticular, subcapsular, subarachnoid, intraspinal and intrasternal injection and infusion.
Pharmaceutical compositions suitable for parenteral administration comprise one or more active compounds in combination with one or more pharmaceutically acceptable sterile isotonic aqueous or nonaqueous solutions, dispersions, suspensions or emulsions, or sterile powders which may be reconstituted into sterile injectable solutions or dispersions just prior to use, which may contain antioxidants, buffers, bacteriostats, solutes which render the formulation isotonic with the blood of the intended recipient or suspending or thickening agents.
Examples of suitable aqueous and nonaqueous carriers that may be employed in the pharmaceutical compositions of the invention include water, ethanol, polyols (such as glycerol, propylene glycol, polyethylene glycol, and the like), and suitable mixtures thereof, vegetable oils, such as olive oil, and injectable organic esters, such as ethyl oleate. Proper fluidity can be maintained, for example, by the use of coating materials, such as lecithin, by the maintenance of the required particle size in the case of dispersions, and by the use of surfactants.
These compositions may also contain adjuvants such as preservatives, wetting agents, emulsifying agents and dispersing agents. Prevention of the action of
microorganisms may be ensured by the inclusion of various antibacterial and antifungal agents, for example, paraben, chlorobutanol, phenol sorbic acid, and the like. It may also be desirable to include isotonic agents, such as sugars, sodium chloride, and the like into the compositions. In addition, prolonged absorption of the injectable pharmaceutical form may be brought about by the inclusion of agents that delay absorption such as aluminum monostearate and gelatin.
In some cases, in order to prolong the effect of a drug, it is desirable to slow the absorption of the drug from subcutaneous or intramuscular injection. This may be accomplished by the use of a liquid suspension of crystalline or amorphous material having poor water solubility. The rate of absorption of the drug then depends upon its rate of dissolution, which, in turn, may depend upon crystal size and crystalline form. Alternatively, delayed absorption of a parenterally administered drug form is accomplished by dissolving or suspending the drug in an oil vehicle.
Injectable depot forms are made by forming microencapsulated matrices of the subject compounds in biodegradable polymers such as polylactide-polyglycolide. Depending on the ratio of drug to polymer, and the nature of the particular polymer employed, the rate of drug release can be controlled. Examples of other biodegradable polymers include poly(orthoesters) and poly(anhydrides). Depot injectable formulations are also prepared by entrapping the drug in liposomes or microemulsions that are compatible with body tissue.
For use in the methods of this invention, active compounds can be given per se or as a pharmaceutical composition containing, for example, 0.1 to 99.5% (more preferably, 0.5 to 90%) of active ingredient in combination with a pharmaceutically acceptable carrier.
Methods of introduction may also be provided by rechargeable or
biodegradable devices. Various slow release polymeric devices have been developed and tested in vivo in recent years for the controlled delivery of drugs, including proteinacious biopharmaceuticals. A variety of biocompatible polymers (including hydrogels), including both biodegradable and non-degradable polymers, can be used to form an implant for the sustained release of a compound at a particular target site.
Actual dosage levels of the active ingredients in the pharmaceutical compositions may be varied so as to obtain an amount of the active ingredient that is
effective to achieve the desired therapeutic response for a particular patient, composition, and mode of administration, without being toxic to the patient.
The selected dosage level will depend upon a variety of factors including the activity of the particular compound or combination of compounds employed, or the ester, salt or amide thereof, the route of administration, the time of administration, the rate of excretion of the particular compound(s) being employed, the duration of the treatment, other drugs, compounds and/or materials used in combination with the particular compound(s) employed, the age, sex, weight, condition, general health and prior medical history of the patient being treated, and like factors well known in the medical arts.
A physician or veterinarian having ordinary skill in the art can readily determine and prescribe the therapeutically effective amount of the pharmaceutical composition required. For example, the physician or veterinarian could start doses of the pharmaceutical composition or compound at levels lower than that required in order to achieve the desired therapeutic effect and gradually increase the dosage until the desired effect is achieved. By "therapeutically effective amount" is meant the concentration of a compound that is sufficient to elicit the desired therapeutic effect. It is generally understood that the effective amount of the compound will vary according to the weight, sex, age, and medical history of the subject. Other factors which influence the effective amount may include, but are not limited to, the severity of the patient's condition, the disorder being treated, the stability of the compound, and, if desired, another type of therapeutic agent being administered with the compound of the invention. A larger total dose can be delivered by multiple administrations of the agent. Methods to determine efficacy and dosage are known to those skilled in the art (Isselbacher et al. (1996) Harrison's Principles of Internal Medicine 13 ed., 1814-1882, herein incorporated by reference).
In general, a suitable daily dose of an active compound used in the
compositions and methods of the invention will be that amount of the compound that is the lowest dose effective to produce a therapeutic effect. Such an effective dose will generally depend upon the factors described above.
If desired, the effective daily dose of the active compound may be
administered as one, two, three, four, five, six or more sub-doses administered separately at appropriate intervals throughout the day, optionally, in unit dosage
forms. In certain embodiments of the present invention, the active compound may be administered two or three times daily. In preferred embodiments, the active compound will be administered once daily.
The patient receiving this treatment is any animal in need, including primates, in particular humans, and other mammals such as equines, cattle, swine and sheep; and poultry and pets in general.
In certain embodiments, compounds of the invention may be used alone or conjointly administered with another type of therapeutic agent. As used herein, the phrase "conjoint administration" refers to any form of administration of two or more different therapeutic compounds such that the second compound is administered while the previously administered therapeutic compound is still effective in the body (e.g., the two compounds are simultaneously effective in the patient, which may include synergistic effects of the two compounds). For example, the different therapeutic compounds can be administered either in the same formulation or in a separate formulation, either concomitantly or sequentially. In certain embodiments, the different therapeutic compounds can be administered within one hour, 12 hours, 24 hours, 36 hours, 48 hours, 72 hours, or a week of one another. Thus, an individual who receives such treatment can benefit from a combined effect of different therapeutic compounds.
In certain embodiments, conjoint administration of compounds of the invention with one or more additional therapeutic agent(s) (e.g., one or more additional chemotherapeutic agent(s)) provides improved efficacy relative to each individual administration of the compound of the invention (e.g., compound of formula I or la) or the one or more additional therapeutic agent(s). In certain such embodiments, the conjoint administration provides an additive effect, wherein an additive effect refers to the sum of each of the effects of individual administration of the compound of the invention and the one or more additional therapeutic agent(s).
This invention includes the use of pharmaceutically acceptable salts of compounds of the invention in the compositions and methods of the present invention. In certain embodiments, contemplated salts of the invention include, but are not limited to, alkyl, dialkyl, trialkyl or tetra-alkyl ammonium salts. In certain
embodiments, contemplated salts of the invention include, but are not limited to, L- arginine, benenthamine, benzathine, betaine, calcium hydroxide, choline, deanol,
diethanolamine, diethylamine, 2-(diethylamino)ethanol, ethanolamine,
ethylenediamine, N-methylglucamine, hydrabamine, lH-imidazole, lithium, L-lysine, magnesium, 4-(2-hydroxyethyl)morpholine, piperazine, potassium, l-(2- hydroxyethyl)pyrrolidine, sodium, triethanolamine, tromethamine, and zinc salts. In certain embodiments, contemplated salts of the invention include, but are not limited to, Na, Ca, K, Mg, Zn or other metal salts.
The pharmaceutically acceptable acid addition salts can also exist as various solvates, such as with water, methanol, ethanol, dimethylformamide, and the like. Mixtures of such solvates can also be prepared. The source of such solvate can be from the solvent of crystallization, inherent in the solvent of preparation or crystallization, or adventitious to such solvent.
Wetting agents, emulsifiers and lubricants, such as sodium lauryl sulfate and magnesium stearate, as well as coloring agents, release agents, coating agents, sweetening, flavoring and perfuming agents, preservatives and antioxidants can also be present in the compositions.
Examples of pharmaceutically acceptable antioxidants include: (1) water- soluble antioxidants, such as ascorbic acid, cysteine hydrochloride, sodium bisulfate, sodium metabisulfite, sodium sulfite and the like; (2) oil-soluble antioxidants, such as ascorbyl palmitate, butylated hydroxyanisole (BHA), butylated hydroxytoluene (BHT), lecithin, propyl gallate, alpha-tocopherol, and the like; and (3) metal-chelating agents, such as citric acid, ethylenediamine tetraacetic acid (EDTA), sorbitol, tartaric acid, phosphoric acid, and the like.
In certain embodiments, the invention relates to a method for conducting a pharmaceutical business, by manufacturing a formulation of a compound of the invention, or a kit as described herein, and marketing to healthcare providers the benefits of using the formulation or kit for treating or preventing any of the diseases or conditions as described herein.
In certain embodiments, the invention relates to a method for conducting a pharmaceutical business, by providing a distribution network for selling a formulation of a compound of the invention, or kit as described herein, and providing instruction material to patients or physicians for using the formulation for treating or preventing any of the diseases or conditions as described herein.
In certain embodiments, the invention comprises a method for conducting a pharmaceutical business, by determining an appropriate formulation and dosage of a compound of the invention for treating or preventing any of the diseases or conditions as described herein, conducting therapeutic profiling of identified formulations for efficacy and toxicity in animals, and providing a distribution network for selling an identified preparation as having an acceptable therapeutic profile. In certain embodiments, the method further includes providing a sales group for marketing the preparation to healthcare providers.
In certain embodiments, the invention relates to a method for conducting a pharmaceutical business by determining an appropriate formulation and dosage of a compound of the invention for treating or preventing any of the disease or conditions as described herein, and licensing, to a third party, the rights for further development and sale of the formulation.
Examples
Example 1 : Synthetic Protocols
Synthesis of linker cores:
5,5'-(butane-l,4-diyl)-bis(l,3,4-thiadiazol-2-amine) (1001)
1001
A mixture of adiponitrile (8.00 g, 73.98 mmol) and thiosemicarbazide (13.48 g, 147.96 mmol) in trifluoroacetic acid (TFA) (75 mL) was heated at 80 °C for 17 hours. The reaction was cooled to room temperature and poured into a mixture of ice and water. Sodium hydroxide pellets were added to the mixture until it was basic (pH 14). The white precipitate was collected by suction filtration, rinsed with water and dried to provide 5,5'-(butane-l,4-diyl)-bis(l,3,4-thiadiazol-2-amine) (1001, 13.07 g). 1H NMR (300 MHz, DMSO-d6) δ 7.00 (s, 4H), 2.84 (bs, 4H), 1.68 (bs, 4H).
Synthesis of 5,5'-(thiobis(ethane-2,l-diyl))bis(l,3,4-thiadiazol-2-amine) (1002)
1002
Compound 1002 was prepared as described in US/2002/0115698 Al
5,5'-(2-methylbutane-l,4-diyl)-bis(l,3,4-thiadiazol-2-amine) (1003)
1003
A mixture of 3-methyl adipic acid (5.00 g, 31.22 mmol) and thiosemicarbazide (5.69 g, 62.43 mmol) in POCI3 (45 mL) was heated at 90 °C for 4 h. The reaction was cooled to room temperature and poured into a mixture of ice and water. Sodium hydroxide pellets were added to the mixture until it was basic (pH 14). The white precipitate was collected by suction filtration, rinsed with water and dried to provide
5,5*-(2-methylbutane-l,4-diyl)-bis(l,3,4-thiadiazol-2-amine) (1003, 8.97 g). 1H NMR (300 MHz, DMSO-de) δ 7.00 (s, 4H), 2.89-2.81 (m, 3H), 2.89-2.81 (m, 3H), 2.69 (dd, J= 7.6, 7.6 Hz, 1H), 1.89-1.46 (m, 3H), 0.94 (d, J= 6.6 Hz, 3H).
5,5'-(propane-l,3-diyl)-bis(l,3,4-thiadiazol-2-amine) (1004)
1004 A mixture of glutaronitrile (5.00 g, 53.13 mmol) and thiosemicarbazide (9.68 g,
106.26 mmol) in TFA (50 mL) was heated at 85 °C for 4 h. The reaction was cooled to room temperature and poured into a mixture of ice and water. Sodium hydroxide pellets were added to the mixture until it was basic (pH 14). The white precipitate was collected by suction filtration, rinsed with water and dried to provide 5,5'-(propane- l,3-diyl)-bis(l,3,4-thiadiazol-2-amine) (1004, 13.72 g). 1H NMR (300 MHz, DMSO- de) δ 7.06-7.03 (s, 4H), 2.87 (t, J= 7.5 Hz, 4H), 2.02 - 1.95 (m, 2H).
5-(2-((2-(5-amino-l,3,4-thiadiazol-2-yl)ethyl)amino)ethyl)-l,3,4-thiadiazol-2-amine
1005
A mixture of 3,3'-iminodipropionitrile (1.50 g, 12.18 mmol) and thiosemicarbazide (2.22 g, 24.36 mmol) in TFA (10 mL) was heated at 85 for 4.5 h. The reaction was cooled to room temperature and poured into a mixture of ice and water. Sodium hydroxide pellets were added to the mixture until it was basic (pH 14). The white precipitate was collected by suction filtration, rinsed with water and dried to provide 5-(2-((2-(5-amino-l,3,4-thiadiazol-2-yl)ethyl)amino)ethyl)-l,3,4-thiadiazol-2-amine
(1005, 1.47 g). 1H NMR (300 MHz, DMSO-d6) δ 6.95 (s, 4H), 2.90 (d, J = 6.0 Hz, 4H), 2.83 (d, J= 6.3 Hz, 4H).
LiOH.H20
To a solution of methyl 3-((2-methoxy-2-oxoethyl)thio)propanoate (5.0 g, 26 mmol) in THF/MeOH/water (60mL, 4: 1 : 1) was added lithium hydroxide monohydrate (4.375 g, 101 mmol). The resulting mixture was stirred at room temperature overnight before it was concentrated under reduced pressure. The residue obtained was diluted with water (-lOOmL) and the resulting solution was acidified with 6N HC1. The mixture was partitioned between water and ethyl acetate. The organic extract was washed with more water, separated, dried over sodium sulfate, filtered and evaporated to afford 3- ((carboxymethyl)thio)propanoic acid (3.64g, 85%) as a white solid. 1H NMR
(300MHz, DMSO-d6) δ ppm 2.55-2.57 (t, 2H) 2.75-2.79 (t, 2H) 3.27 (s, 2H) 12.41 (s, 2H)
To a mixture of 3-((carboxymethyl)thio)propanoic acid (3.64g, 22.2 mmol) and thiosemicarbazide (4.1g, 45 mmol) was added phosphorus oxychloride (25mL) slowly. The resulting mixture was stirred at 90°C for 3hr before it was poured over crushed ice slowly. The solid separated was filtered and the filtrate was basified to
pH~13 by solid sodium hydroxide. The solid separated was filtered, washed with water and dried at 45°C under vacuum overnight to afford 1006 (~3g, 50%) as a tan solid. 1H NMR (300MHz, DMSO-d6) δ ppm 2.79-2.83 (t, 2H) 3.06-3.10 (t, 2H) 3.99 (s, 2H) 7.04 (s, 2H) 7.16 (s, 2H)
O O S N-N N-N
HCT ^ H2N^NHNH2 S^^b^^S
1007
A mixture of 2,2'-Thiodiacetic acid (5.00 g, 33.3 mmol) and thiosemicarbazide (6.07 g, 66.6 mmol) in POCl
3 (40 mL) was heated at 90 °C for 5 h. The reaction was cooled to room temperature and carefully poured it onto a mixture of ice and water. Sodium hydroxide pellets were added to the mixture until it was basic (pH 14). The white precipitate was collected by suction filtration, rinsed with water and dried to afford 1007. 1H NMR (300 MHz, DMSO-d
6) δ 7.18 (s, 4H), 3.96 (s, 4H).
A mixture of 1,5-dicyanopentane (1.00 g, 8.19 mmol) and thiosemicarbazide (1.5 g, 16.40 mmol) in TFA (3 mL) was heated at 85 °C for 5 h. The reaction was cooled to room temperature and poured into a mixture of ice and water. Sodium hydroxide pellets were added to the mixture until it was basic (pH 14). The white precipitate was collected by suction filtration, rinsed with water and dried to afford 1008. 1H NMR (300 MHz, DMSO-de) δ 6.98 (s, 4H), 2.81 (t, 4H), 1.67 (m, 4H), 1.20 (m, 2H).
Acylation of diamino core: Method A: via acid chloride
N,N- [5 ,5 '-(butane- 1 ,4-diyl)-bis( 1 ,3 ,4-thiadiazole-5 ,2-diyl)] -bis(2-phenylacetamide) (21)
1001 21
To a suspension of 1001 (8.00 g, 31.21 mmol) in l-Methyl-2-pyrrolidinone (NMP)
100 mL) at 0 °C was added phenylacetyl chloride (10.25 mL, 77.54 mmol) dropwise. The resulting mixture was stirred at 0 °C for 1 h before it was quenched by addition of water (-200 mL). The white precipitate was collected by suction filtration, rinsed with water and dried to provide N,N'-[5,5'-(butane-l,4-diyl)-bis(l,3,4-thiadiazole-5,2- diyl)]-bis(2-phenylacetamide) (21, 14.02 g). 1H NMR (300 MHz, DMSO-d6) δ 12.66 (s, 2H), 7.34 (m, 10H), 3.81 (s, 4H), 3.01 (bs, 4H), 1.76 (bs, 4H).
Compound 43 was prepared following Method A using phenoxyacetyl chloride. 1H NMR (300 MHz, DMSO-d6) δ 12.68 (s, 2H), 7.35-7.30 (m, 4H), 6.99-6.97 (m, 6H), 4.90 (s, 4H), 3.05 (bs, 4H), 1.79 (bs, 4H).
Compound 100 was prepared following Method A. 1H NMR (300 MHz, DMSO-d
6) δ 12.42 (s, 2H), 3.64 (t, J= 5.6 Hz, 4H), 3.24 (s, 6H), 3.01 (bs, 4H), 2.72 (t, J= 6.2 Hz, 4H), 1.79 (bs, 4H).
Compound 5 was prepared according to Method A: 1H NMR (300 MHz, DMSO-d
6) δ 12.66(s, 4H), 3.27(t, J=6.99 Hz, 4H), 2.95(t, J=7.02 Hz, 4H), 2.12(s, 6H).
To a suspension of 1001 (200 mg, 0.78 mmol) in NMP (2 mL) at 0 °C was added O- acetylmandelic acid chloride (0.44 mL, 1.95 mmol) dropwise. The resulting mixture was stirred at 0 °C for 1.5 h before it was quenched by addition of water (-10 mL). The white precipitate was collected by suction filtration, rinsed with more water and dried. The crude material was purified by recrystallization with a mixture of DMSO and MeOH to afford 173.
A flask was charged with 173 and 2N ammonia in MeOH (3 ml) and the resulting mixture was stirred at room temperature for 6 h. The solvent was removed and the resulting material was dried in the oven to afford 174. 1H NMR (300 MHz, DMSO- d6) δ 12.42 (s, 2H), 7.53-7.31 (m, 10H), 6.35 (s, 2H), 5.34 (d, J= 1.14 Hz, 2H), 3.01 (bs, 4H), 1.76 (bs, 4H).
Compound 306 was prepared according to the procedure for compound 174 above.
To a suspension of 1001 (400 mg, 1.56 mmol) in NMP (4 mL) at 0 °C was added (R)-(-)-0-formylmandeloyl chloride (0.61 mL, 3.90 mmol) dropwise. The resulting mixture was stirred at 0 °C for 1.5 h before it was quenched by addition of water (~10 mL). The white precipitate was collected by suction filtration, rinsed with more water
and dried. The crude material was purified by recrystallization with a mixture of DMSO and MeOH to afford 68.
A flask was charged with 68 and 2N ammonia in MeOH (5 ml) and the resulting mixture was stirred at room temperature for 2 h. The solvent was removed and the resulting material was dried in the oven to afford 80. 1H NMR (300 MHz, DMSO-d
6) δ 7.53-7.31 (m, 10H), 6.34 (s, 2H), 5.33 (s, 2H), 3.01 (bs, 4H), 1.75 (bs, 4H).
To a suspension of 1002 (544 mg, 1.89 mmol) in NMP (13 mL) at -15°C was added phenylacetyl chloride (0.249 mL, 1.89 mmol) dropwise. The resulting mixture was stirred at 0°C for 1 h and quenched by the addition of water (54 mL). The white precipitate was collected by suction filtration, rinsed with water (27 mL) and ethyl acetate (3x27 mL). The filtrate was basified to pH 11 using 2.5M NaOH. The layers were separated and the aqueous layer extracted with dichloromethane (3x54 mL). The combined organic layers were dried over magnesium sulfate and concentrated to afford N-(5-(2-((2-(5-amino-l,3,4-thiadiazol-2-yl)ethyl)thio)ethyl)-l,3,4-thiadiazol-2- yl)-2-phenylacetamide (17, 56 mg) 1H NMR (300 MHz, DMSO-d6) δ 12.71(s, 1H), 7.32(s, 5H), 3.81(s, 2H), 3.25(t, J=7.61 Hz, 2H) 3.06(t, J=7.25 Hz, 2H), 2.92(t,
J=6.90 Hz, 2H), 2.85(t, J=6.86 Hz, 2H)
Phenylacetyl chloride (0.134 mL, 1.01 mmol) and acetoxyacetyl chloride (0.109 mL, 1.01 mmol) were mixed together in NMP (0.5 mL). This mixture was slowly added to a suspension of 1002 (292 mg, 1.01 mmol) in NMP (7 mL) at RT. The resulting mixture was stirred at RT for 1 h and quenched by the addition of water (20 mL). The white precipitate was collected by suction filtration, rinsed with water and dried under high vacuum. The crude material was purified by preparative HPLC. Compound 26: 1H NMR (300 MHz, DMSO-d
6) δ 12.69(s, 2H), 7.34(3, 5H), 4.81(s, 2H), 3.82(s, 2H), 2.96(bs, 4H), 2.14(s, 3H).
44
Compound 44 was prepared following the procedure for compound 21 described previously. 1H NMR (300 MHz, DMSO-d6) δ 12.66 (s, 2H), 7.34-7.28 (m, 10H), 3.81 (s, 4H), 3.05-3.00 (m, 3H), 2.87 (dd, J= 7.9, 8.2 Hz, 1H), 1.95-1.77 (m, 3H), 0.94 (d, J= 6.5 Hz, 3H).
Compound 72 was prepared following the procedure for compound 21 described previously. To a suspension of diamine 1004 (0.70 g, 3.07 mmol) in NMP (15 mL) at 0 °C was added phenylacetyl chloride (811 μί, 6.13 mmol) dropwise. The resulting mixture was stirred at 0 °C for 1 h before it was quenched by addition of water. The white precipitate was collected by suction filtration, rinsed with water and dried to provide NJV-[5 ,5 '-(propane- 1 ,3 -diyl)-bis( 1 ,3 ,4-thiadiazole-5 ,2-diyl)]-bis(2- phenylacetamide) (72, 1.37 g). 1H NMR (300 MHz, DMSO-d6) δ 12.68 (s, 2H), 7.38-7.27 (m, 10H), 3.82 (s, 4H), 3.06 (t, J= 7.2 Hz, 4H), 2.17-2.12 (m, 2H).
To a suspension of compound 1005 (100 mg, 0.37 mmol) in DMF (12 mL) at room temperature was added a solution of (t-Boc)
20 (88 mg, 0.41 mmol) in DMF (2 mL). The mixture was stirred at room temperature for 24 h. To this reaction mixture was added NMP (2 mL) and followed by addition of phenylacetyl chloride (97 μί, 0.74 mmol). The reaction was stirred for 1 h before it was poured into a mixture of ice- water. The solid was collected by suction filtration, rinsed with water and dried to provide 1010 (180 mg).
The above product 1010 (160 mg, 0.26 mmol) in a mixture of TFA (1.5 mL) and CH2CH2 (10 mL) was stirred at room temperature for 4 h before it was concentrated. The residue was re-taken up in CH2C12 (3x) and concentrated to provide N,N"-(5,5'- (azanediyl-bis(ethane-2, 1 -diyl))-bis( 1 ,3 ,4-thiadiazole-5 ,2-diyl))-bis(2- phenylacetamide) trifluoroacetic acid (149, 122 mg). 1H NMR (300 MHz, DMSO-d6) δ 12.81 (s, 2H), 8.75 (bs, 2H), 7.38-7.27 (m, 10H), 3.84 (s, 4H), 3.45 (d, J= 2.9 Hz, 4H), 3.39 (d, J = 6.0 Hz, 4H).
To a suspension of 1006 (0.274g, lmmol) in NMP (5mL) was added phenyl acetyl chloride (0.263mL, 2mmol) dropwise. The mixture was stirred at room temperature for lhr and afterwards it was diluted with water. Solid separated was filtered, washed with more water and dried. The crude material was purified by prep HPLC to afford
199 as a white solid. 1H NMR (300MHz, Dimethylsulfoxide-d6) δ ppm 2.87-2.91 (t, 2H) 3.25-3.29 (t, 2H) 3.82 (s, 4H) 4.19 (s, 2H) 7.26-7.33 (m, 10H) 12.71-12.72 (br s,
2H).
Method B: via acid using peptide coupling reagents
0'"~V| O N— N N— N o
H H
12
To a flask containing 5,5'-(thiobis(ethane-2,l-diyl))bis(l,3,4-thiadiazol-2-amine) (1002) (0.69 mmol, 0.20 g, 1.0 equiv.) was added 2-morpholinoacetic acid (1.52 mmol, 0.22 g, 2.2 equiv.), 0-(Benzotriazol-l-yl)-N,N,N',N'-tetramethyluronium hexafluorophosphate (HBTU) (2.20 mmol, 0.83 g, 3.2 equiv.), 1-
Hydroxybenzotriazole (HOBT) (2.2 mmol, 0.29 g, 3.2 equiv.) 5 mL of DMF followed by Ν,Ν-Diisopropylethylamine (DIEA) (5.52 mmol, 0.71 g, 0.960 mL, 8.0 equiv.). The mixture was stirred overnight at room temperature and then diluted with 15 mL water. The mixture was extracted with EtOAc and the organic layers combined, washed with water, brine and dried over Na2S04. The Na2S04 was removed by filtration and the volatiles removed under reduced pressure to give 0.04 g of compound 12. 1FiNMR (300 MHz, CDC13) Compound 12: δ 3.80 (broad multiplet, 4H), 3.34 (dd, 4H, J= 7.2 Hz), 3.28 (s, 4 H), 3.00 (dd, 4H, J= 7.1 Hz), 2.63 (broad multiplet, 4H).
2) 4M HCI / dioxane
To a flask containing 5,5'-(butane-l,4-diyl)bis(l,3,4-thiadiazol-2-amine) (1101) (3.9 mmol, 1.0 g, 1.0 equiv.) was added (S)-2-((tert-butoxycarbonyl)amino)-2- phenylacetic acid (8.58 mmol, 2.15 g, 2.2 equiv.), HBTU (12.48 mmol, 4.73 g, 3.2 equiv.), HOBt (12.48 mmol, 1.69 g, 3.2 equiv.) 25 mL of DMF followed by DIEA (31.2 mmol, 4.03 g, 5.43 mL, 8.0 equiv.). The mixture was stirred overnight and poured into 150 mL water. The white solids that formed were collected by vacuum filtration, washed with water and dried under vacuum giving 2.47 g of the bis-Boc protected intermediate.
To a slurry of the bis-Boc protected intermediate (2.76 mmol, 2.0 g, 1.0 equiv.) in 20 mL of dichloromethane (DCM) was added 4 M HC1 in dioxane (40 mmol, 10 mL) with vigorous stirring. The mixture briefly became clear and homogeneous then a white precipitate formed. The mixture was stirred overnight and diluted with 20 mL diethyl ether. The solids were collected by vacuum filtration washed with additional diethyl ether and dried under vacuum giving 0.9 g 187.
1HNMR (300 MHz, DMSO, d
6) Compound 187: δ 9.13 (s, 4H), 7.61 (m, 4H), 7.48 (m, 6H), 6.2 (broad singlet, 4H), 5.32 (s, 2H), 3.04 (broad multiplet, 4H), 1.77 (broad multiplet, 4H).
To a solution of 2,2-bis(hydroxymethyl)propionic acid (5.00 g, 37.28 mmol) in acetone (80 mL) at room temperature was added 2,2-dimethoxypropane (6.88 mL, 55.92 mmol) and /?-TsOH H
20 (0.36 g, 1.86 mmol). The reaction was stirred for 2 h before it was quenched with Et
3N (0.30 mL). The organic volatile was removed under reduced pressure. The residue was partitioned between EtOAc and water. The organic layer was washed with brine, dried (MgS0
4) and concentrated to provide the desired product 1011 (5.17 g) as a white solid.
To a suspension of diamine 1001 (500 mg, 1.95 mmol), 3-fluorophenylacetic acid (361 mg, 2.34 mmol) and acid 1011 (442 mg, 2.54 mmol) in DMF (20 mL) at 0 °C was added HOBt (791 mg, 5.85 mmol) and followed by N-(3-Dimethylaminopropyl)- N'-ethylcarbodiimide hydrochloride (EDC) (1.12 g, 5.85 mmol). The mixture was stirred from 0 °C to room temperature over 18 h before it was diluted with water. The
precipitate was collected by suction filtration, washed with water and dried. The crude product was purified by silica gel chromatography eluting with 1-10% MeOH in CH2CI2 to provide N-(5-(4-(5-(2-(3-f uorophenyl)acetamido)-l ,3,4-thiadiazol-2- yl)butyl)-l ,3,4-thiadiazol-2-yl)-2,2,5-trimethyl-l ,3-dioxane-5-carboxamide (1012, 208 mg).
The above product 1012 (87 mg, 0.16 mmol) and TFA (2 mL) in a mixture of THF (8 mL) and water (2 mL) was heated at 50 °C for 5 h before it was concentrated under reduced pressure. The crude residue was purified by HPLC to provide N,N-(5-(4-(5- (2-(3-fluorophenyl)acetamido)-l ,3,4-thiadiazol-2-yl)butyl)-l ,3,4-thiadiazol-2-yl)-3- hydroxy-2-(hydroxymethyl)-2-methylpropanamide (152). 1H NMR (300 MHz,
DMSO-de) δ 12.68 (s, 1H), 1 1.77 (s, 1H), 7.04-7.38 (m, 1H), 7.18-7.09 (m, 4H), 4.98 (s, 2H), 3.86 (s, 2H), 3.62 (dd, J = 10.7, 29.0 Hz, 4H), 3.03 (bs, 4H), 1.77 (bs, 4H), 1.14 (s, 3H).
To a suspension of diamine 1001 (400 mg, 1.56 mmol), 3-fluorophenylacetic acid (313 mg, 2.03 mmol), (i?)-(-)-2,2-dimethyl-5-oxo-l ,3-dioxolane-4-acetic acid (353 mg, 2.03 mmol) and Et
3N (200 μ∑) in DMF (20 mL) at 0 °C was added HOBt (633 mg, 4.68 mmol) and followed by EDC (897 mg, 4.68 mmol). The mixture was stirred from 0 °C to room temperature over 18 h before it was diluted with water. The precipitate was collected by suction filtration and washed with water. The solid was further rinsed with a mixture of hot MeOH-THF. The combined filtrate was concentrated and purified by silica gel chromatography eluting with 1-10% MeOH in CH
2C1
2 to provide (i?)-N-(5-(4-(5-(2-(3-fluorophenyl)acetamido)-l ,3,4-thiadiazol-2- yl)butyl)-l ,3,4-thiadiazol-2-yl)-3,4-dihydroxybutanamide (1013, 93 mg). The above product 1013 (87 mg, 0.16 mmol) and TFA (2 mL) in a mixture of THF (8 mL) and water (2 mL) was heated at 50 °C for 5 h before it was concentrated under
reduced pressure. The crude residue was purified by HPLC to provide (i?)-N-(5-(4- (5-(2-(3-fluorophenyl)acetamido)-l ,3,4-thiadiazol-2-yl)butyl)-l ,3,4-thiadiazol-2-yl)- 3,4-dihydroxybutanamide (153). 1H NMR (300 MHz, DMSO-d
6) δ 12.67 (s, 1H), 12.43 (s, 1H), 7.41-7.38 (m, 1H), 7.20-7.12 (m, 4H), 4.45-4.40 (m, 1H), 3.86 (s, 2H), 3.03 (bs, 4H), 2.85-2.77 (m, 2H), 1.78 (bs, 4H).
To a suspension of (S)-(+)-0-acetylmandelic acid (666 mg, 3.43 mmol) and 0-(7- Azabenzotriazol- 1 -yl)-N,N,N',N'-tetramethyluronium hexafluorophosphate (H ATU) (1.47 g, 3.86 mmol) in DMF (4 mL) was added DIEA (0.672 ml, 3.86 mmol) followed by 1001 (400 mg, 1.56 mmol). The resulting mixture was stirred at room temperature overnight before it was quenched by addition of water (~10 mL). The white precipitate was collected by suction filtration, rinsed with more water and dried. The crude material was purified by recrystallization with a mixture of DMSO and MeOH to afford 66. A flask was charged with 66 and 2N ammonia in MeOH (5 ml) and the resulting mixture was stirred at room temperature for 6 h. The solvent was removed and the resulting material was dried in the oven to afford 92. 1H NMR (300 MHz, DMSO-d
6) δ 12.42 (s, 2H), 7.53-7.31 (m, 10H), 6.35 (s, 2H), 5.33 (s, 2H), 3.01 (bs, 4H), 1.76 (bs, 4H).
A flask was charged with 1001 (200 mg, 0.78 mmol), DL-3-phenyllactic acid (285 mg, 1.716 mmol), and HOBT (527 mg, 3.9 mmol) in DMF (3 ml) was added EDC
(897 mg, 4.68 mmol) followed by triethylamine (0.87 ml, 6.24 mmol). The resulting mixture was stirred at room temperature overnight before it was quenched by addition of water (~5 mL). The mixture was partitioned between water and EtOAc. The organic extract was washed with water, dried over sodium sulfate, filtered and evaporated. The crude material was purified by silica gel chromatography eluting with 0-6% MeOH in CH
2C1
2 to afford 69. 1H NMR (300 MHz, DMSO-d
6) δ 12.20 (s, 2H), 7.24 (m, 10H), 5.75 (d, J= 6.87 Hz, 2H), 4.43 (m, 2H), 3.10 (m, 6H), 2.89-2.81 (m, 2H), 1.80 (bs, 4H).
169 A flask was charged with 1001 (200 mg, 0.78 mmol), D-(+)-3-phenyllactic acid (285 mg, 1.716 mmol), and HOBt (464 mg, 3.43 mmol) in DMF (3 ml) was added EDC (822 mg, 4.28 mmol) followed by triethylamine (0.718 ml, 5.15 mmol). The resulting mixture was stirred at room temperature overnight before it was quenched by addition of water (~5 mL). The mixture was partitioned between water and EtOAc. The organic extract was washed with water, dried over sodium sulfate, filtered and evaporated. The crude material was purified by silica gel chromatography eluting with 0-6% MeOH in CH
2C1
2 to afford 169. 1H NMR (300 MHz, DMSO-d
6) δ 12.20 (s, 2H), 7.24 (m, 10H), 5.75 (d, J= 6.87 Hz, 2H), 4.43 (m, 2H), 3.03 (m, 6H), 2.89- 2.81 (m, 2H), 1.80 (bs, 4H).
146
A flask was charged with 1001 (200 mg, 0.78 mmol), L-(-)-3-phenyllactic acid (285 mg, 1.716 mmol), and HOBt (464 mg, 3.43 mmol) in DMF (3 ml) was added EDC (822 mg, 4.28 mmol) followed by triethylamine (0.718 ml, 5.15 mmol). The resulting mixture was stirred at room temperature overnight before it was quenched by addition of water (~5 mL). The mixture was partitioned between water and EtOAc. The organic extract was washed with more water, dried over sodium sulfate, filtered and
evaporated. The crude material was purified by silica gel chromatography eluting with 0-6% MeOH in CH2C12 to afford 146. 1H NMR (300 MHz, DMSO-d6) δ 12.27 (s, 2H), 7.31 (m, 10H), 5.78 (m, 2H), 4.44 (m, 2H), 3.05 (m, 6H), 2.87 (m, 2H), 1.79 (bs, 4H).
127
To a suspension of (R)-(+)-3-hydroxy-3-phenylpropionic acid (285 mg, 1.72 mmol) and HATU (719 mg, 1.89 mmol) in DMF (3 mL) was added DIEA (0.329 ml, 1.89 mmol) followed by 1001 (200 mg, 0.78 mmol). The resulting mixture was stirred at room temperature overnight before it was quenched by addition of water (~10 mL).
The white precipitate was collected by suction filtration, rinsed with more water and dried. The crude material was purified by recrystallization with DMSO and MeOH to afford 127. 1H NMR (300 MHz, DMSO-d6) δ 12.38 (s, 2H), 7.34 (m, 10H), 5.56 (m,
143
To a suspension of (R)-2-hydroxy-2-phenylbutyric acid (310 mg, 1.72 mmol) and
HATU (719 mg, 1.89 mmol) in DMF (3 mL) was added DIEA (0.329 ml, 1.89 mmol) followed by 1001 (200 mg, 0.78 mmol). The resulting mixture was stirred at room temperature overnight before it was quenched by addition of water (~10 mL). The crude material was purified by HPLC to afford 143. 1H NMR (300 MHz, DMSO-d6) δ 7.61 (d, J= 7.65 Hz, 4H), 7.34 (m, 6H), 2.99 (bs, 4H), 2.26 (m, 2H), 2.10 (m, 2H) 1.74 (bs, 4H), 0.80 (t, 6H).
To a suspension of 3-Oxo-l-indancarboxylic acid (604 mg, 3.43 mmol) and HATU (1.47g, 3.86 mmol) in DMF (5 mL) was added DIEA (0.672 ml, 3.86 mmol) followed by 1001 (400 mg, 1.56 mmol). The resulting mixture was stirred at room temperature overnight before it was quenched by addition of water (~10 mL). The light brown precipitate was collected by suction filtration, rinsed with water and dried. The crude material was purified by recrystallization with a mixture of DMSO and MeOH to afford 64.
To a suspension of 64 (100 mg, 0.175 mmol) in EtOH (20 ml) at 0 °C was added NaBH4 (15 mg, 0.384 mmol) and the resulting mixture was stirred for 1 h before it was quenched by IN HC1. The mixture was partitioned between IN HC1 and EtOAc, the organic extract was dried over sodium sulfate, filtered and evaporated. The crude material was purified by silica gel chromatography eluting with 0-6% MeOH in CH2CI2 and further purified by recrystallization with a mixture of DMSO and MeOH to afford 94. 1H NMR (300 MHz, DMSO-d6) δ 12.81 (s, 2H), 7.34 (m, 8H), 5.56 (m, 2H), 5.11 (t, 2H), 4.15 (t, 2H), 3.05 (bs, 4H), 2.70 (m, 2H), 2.15 (m, 2H), 1.80 (bs, 4H).
To a solution of DL-mandelic acid (1 g, 6.57 mmol) in DMF (10 ml) at 0 °C was added NaH ( 700 mg, 19.7 mmol) and allowed the mixture to stir for 20 minutes before 2-bromoethyl methyl ether (1.24 ml, 13.1 mmol) was added dropwise. The resulting mixture was stirred at 0 °C and slowly warmed up to room temperature overnight before it was quenched by IN HCl. The mixture was partitioned between IN HCl and EtOAc, the organic extract was washed with water, dried over sodium sulfate, filtered and evaporated to afford 1014.
To a suspension of 1014 (500 mg, 2.37 mmol) and HATU (995 mg, 2.62 mmol) in DMF (3 mL) was added DIEA (0.456 ml, 2.62 mmol) followed by 1001 (277 mg,
1.08 mmol). The resulting mixture was stirred at room temperature overnight before it was quenched by addition of water (~6 mL). The mixture was partitioned between water and EtOAc. The organic extract was washed with water, dried over sodium sulfate, filtered and evaporated. The crude material was purified by HPLC to afford 203. 1H NMR (300 MHz, DMSO-d6) δ 12.58 (s, 2H), 7.49-7.37 (m, 10H), 5.22 (s, 2H), 3.66-3.54 (m, 8H), 3.27 (s, 6H), 3.01 (bs, 4H), 1.75 (bs, 4H).
HCI
To a suspension of 2-(4-Boc-piperazinyl)-2-phenylacetic acid (1.1 g, 3.43 mmol) and HATU (1.47g, 3.86 mmol) in DMF (5 mL) was added DIEA (0.672 ml, 3.86 mmol) followed by 1001 (400 mg, 1.56 mmol). The resulting mixture was stirred at room temperature overnight before it was quenched by addition of water (~10 mL). The white precipitate was collected by suction filtration, rinsed with water and dried. The crude material was purified by recrystallization with DMSO and MeOH to afford 63.
A flask was charged with 63 and 4N HCI in 1,4-dioxane (6 ml) and the resulting mixture was stirred at room temperature for 3 h. The precipitation was collected by filtration, rinse with EtOAc/CH2Cl2 and dried to afford 77. 1H NMR (300 MHz, DMSO-de) δ 9.10 (bs, 4H), 7.51-7.41 (m, 10H), 4.90 (bs, 2H), 4.62 (s, 2H), 3.15 (bs, 8H), 3.03 (bs, 4H), 2.73 (bs, 8H), 1.76 (bs, 4H).
To a suspension of (R)-(+)-3-hydroxy-3-phenylpropionic acid (254 mg, 1.53 mmol) and HATU (640 mg, 1.68 mmol) in DMF (3 mL) was added DIEA (0.292 ml, 1.68 mmol) followed by 1002 (200 mg, 0.693 mmol). The resulting mixture was stirred at room temperature overnight before it was quenched by addition of water (~10 mL). The white precipitate was collected by suction filtration, rinsed with water and dried. The crude material was purified by recrystallization with a mixture of DMSO and
MeOH to afford 126. 1H NMR (300 MHz, DMSO-d
6) δ 12.40 (s, 2H), 7.38 (m, 10H), 5.55 (m, 2H), 5.09 (m, 2H), 3.27 (t, 4H), 2.95 (t, 4H), 2.82 (m, 4H).
76
A flask was charged with 1002 (200 mg, 0.693 mmol), 2-(4-Boc-piperazinyl)-2- phenylacetic acid (244 mg, 0.763 mmol), and HOBt (187 mg, 1.39 mmol) in DMF (3 ml) was added EDC (332 mg, 1.73 mmol) followed by triethylamine (0.290 ml, 2.08 mmol). The resulting mixture was stirred at room temperature overnight before phenylacetyl chloride (0.037 ml, 0.277 mmol) was added dropwise at 0 °C and stirred for 1 h before it was quenched by addition of water (~10 mL). The white precipitate was collected by suction filtration, rinsed with water and dried. The crude material was purified by HPLC to afford 70 and 76.
A flask was charged with 70 and 4N HCI in 1,4-dioxane (6 ml) and the resulting mixture was stirred at room temperature for 3 h. The precipitation was collected by filtration, rinse with EtOAc/CH
2Cl
2 and dried to afford 78. 1H NMR (300 MHz, DMSO-de) δ 12.70 (s, 2H), 8.97 (bs, 2H), 7.50-7.29 (m, 10H), 4.72 (bs, 1H), 4.59 (s, 1H), 3.82 (s, 2H), 3.27 (t, 4H), 3.15 (bs, 4H), 2.92 (t, 4H), 2.70 (bs, 4H).
A flask was charged with 76 and 4N HC1 in 1,4-dioxane (6 ml) and the resulting mixture was stirred at room temperature for 3 h. The precipitation was collected by filtration, rinse with EtOAc/CH2Cl2 and dried to afford 79. 1H NMR (300 MHz, DMSO-de) δ 12.87 (s, 2H), 9.03 (bs, 4H), 7.50-7.40 (m, 10H), 4.67 (bs, 2H), 4.59 (s, 2H), 3.28 (t, 4H), 3.14 (bs, 8H), 2.97 (t, 4H), 2.71 (bs, 8H).
Amide Coupling General Procedure (used for following examples): To a 0.2 molar concentration suspension of carboxylic acid (2 equivalents) in DMF was added HATU (2 equivalents) and stirred till reaction mixture is clear followed by the addition of an amine (1 equivalent) and DIPEA(4 equivalents). The resulting mixture was stirred at room temperature overnight before it was quenched by the addition of water. The solid separated was filtered, washed with water and dried.
39: 1H NMR (300MHz, Dimethylsulfoxide-d6) δ ppm 1.89-2.01 (m, 6H) 2.18-2.29 (m, 2H) 2.95-3 (m, 4H) 3.79-3.86 (m, 2H) 3.94-4.02 (m, 2H) 4.55-4.6 (m, 2H) 12.29 (brs, 2H).
41: 1H NMR (300MHz, Dimethylsulfoxide-d6) δ ppm 2.93-2.98 (m, 4H) 3.27-3.32 (m, -5.2 (br s, 2H) 6.88-7.03 (m, 8H) 12.87-12.92 (br s, 2H).
51: 1H NMR (300MHz, Dimethylsulfoxide-d6) δ ppm 1.78 (br s, 4H) 3.05-3.06 (br s, 4H), 3.38-3.40 (m, 2H) 3.54-3.63 (m, 2H) 5.44-5.50 (m, 2H) 6.92-7.26 (m, 8H) 12.78
54: 1H NMR (300MHz, Dimethylsulfoxide-d6) δ ppm 1.92-2.03 (m, 10H) 2.17-2.28 (m, 2H) 3.05 (br s, 4H) 3.79-3.85 (m, 2H) 3.94-4.01 (m, 2H) 4.55-4.59 (m, 2H) 12.27(br s, 2H).
60: 1H NMR (300MHz, Dimethylsulfoxide-d6) δ ppm 1.77 (br s, 4H) 3.04 (br s, 4H) 5. 7.79 (br s, 2H) 12.80 (br s, 2H).
85: 1H NMR (300MHz, Dimethylsulfoxide-d6) δ ppm 0.20-0.21 (br s, 4H) 0.48-0.50 (br s, 4H) 1.79 (br s, 4H) 2.35-2.38 (br s, 4H) 3.04 (br s, 4H) 12.32 (br s, 2H).
87: 1H NMR (300MHz, Dimethylsulfoxide-d6) 5 ppm 1.78 (br s, 4H) 3.03 (br s, 4H) 4.0 -7.44 (m, 2H) 12.68 (br s, 2H).
114: 1H NMR (300MHz, Dimethylsulfoxide-d6) δ ppm 1.01-1.12 (m, 4H) 1 ,40 (s, 18H) 1.61-1.65 (m, 4H) 1.78 (br s, 4H) 1.95 (br s, 2H) 3.84 (m, 4H) 2.65-2.75 (m,
-3.93 (m, 4H) 12.39 (br s, 2H).
123: 1H NMR (300MHz, Dimethylsulfoxide-d6) δ ppm 1.43 (s, 6H) 1.79-1.94 (m, 10H) -2.31 (m, 2H) 3.05 (br s, 4H) 3.85-4.01 (m, 4H) 11.85 (br s, 2H).
133: 1H NMR (300MHz, Dimethylsulfoxide-d6) 6 ppm 2.92-2.97 (m, 4H) 3.26-3.30 (m, 4H) 4.61-4.87 (m, 6H) 6.83-6.89 (m, 4H) 7.16-7.21 (m, 2H) 7.36-7.38 (m, 2H) 12.95 (br s, 2H).
135: 1H NMR (300MHz, Dimethylsulfoxide-d6) δ ppm 1.77 (br s, 4H) 3.03 (br s, 4H) 4.60-4.87 (m, 6H) 6.83-6.89 (m, 4H) 7.16-7.22 (m, 2H) 7.36-7.38 (m, 2H) 12.92 (br s, 2H).
114: 1H NMR (300MHz, Dimethylsulfoxide-d6) δ ppm 1.01-1.12 (m, 4H) 1,40 (s, 18H) 1.61-1.65 (m, 4H) 1.78 (br s, 4H) 1.95 (br s, 2H) 3.84 (m, 4H) 2.65-2.75 (m,
-3.93 (m, 4H) 12.39 (br s, 2H).
323: 1H NMR (300MHz, Dimethylsulfoxide-d6) δ ppm 1.76 (brs, 4H) 3.01(brs, 4H) 4.02 (s, 4H) 6.56 (s, 2H) 6.94-7.05 (m, 4H) 7.31-7.33 (m, 4H) 11.12 (brs, 2H) 12.69
397: 1H NMR (300MHz, Dimethylsulfoxide-d6) δ ppm 1.75 (brs, 4H) 2.90 (brs, 2H) 3.02 (brs, 2H) 3.67-3.82 (m, 10H) 6.85-7.03 (m, 4H) 7.26-7.36 (m, 5H) 7.55-7.58 (d, -8.21 (d, 1H) 1 1.26 (s, 1H) 12.65 (brs, 1H).
398: 1H NMR (300MHz, Dimethylsulfoxide-d6) δ ppm ppm 1.75 (brs, 4H) 2.90 (brs, 2H) 3.02 (brs, 2H) 3.72-3.78 (m, 10H) 6.42-6.51 (m, 4H) 7.36 (m, 5H) 7.54-7.58 (d, 1 -8.21 (d, 1H) 1 1.26 (s, 1H) 12.65 (brs, 1H).
399: 1H NMR (300MHz, Dimethylsulfoxide-d6) δ ppm 1.48 (s, 9H) 1.75 (brs, 4H) 2.90 (brs, 2H) 3.02 (brs, 2H) 3.74-3.78 (m, 4H) 6.92-6.94 (m,lH) 7.20-7.36 (m, 7H) -7.58 (m, 2H) 8.18-8.21 (d, 1H) 9.34 (s, 1H) 11.26 (s, 1H) 12.65 (brs, 1H).
400: 1H NMR (300MHz, Dimethylsulfoxide-d6) δ ppm 1.48 (s, 9H) 1.75 (brs, 4H) 2.90 (brs, 2H) 3.02 (brs, 2H) 3.71-3.78 (m, 4H) 7.18-7.42 (m, 9H) 7.54-7.58 (m, 2H) 8.18-8.21 (d, 1H) 9.34 (s, 1H) 11.26 (s, 1H) 12.65 (brs, 1H).
324: 1H NMR (300MHz, Dimethylsulfoxide-d6) δ ppm 1.39 (s, 18H) 1.76 (brs, 4H) 3.01(brs, 4H) 3.79 (s, 4H) 4.11-4.13 (brs, 4H) 7.13-7.38 (m, 8H) 12.65 (s, 2H).
Method C: via aluminum amide coupling with esters/lactones
To a suspension of 1002 (288 mg, 1.00 mmol) in toluene (9 mL) was added 3- isochromanone (311 mg, 2.10 mmol) followed by trimethyl aluminum (2M in toluene, 1.0 mL, 2.00 mmol). The resulting mixture was stirred at 75°C for 15 h, cooled to room temperature and diluted with ethyl acetate (50 mL). The organic layer was washed with water (3x20 mL), 10% sodium chloride solution (10 mL), dried
(magnesium sulfate) and concentrated under reduced pressure. The crude product was purified by HPLC to afford N,N*-(5,5*-(thiobis(ethane-2,l-diyl))bis(l,3,4- thiadiazole-5,2-diyl))bis(2-(2-(hydroxymethyl)phenyl)acetamide) (181, 78 mg). 1H NMR (300 MHz, DMSO-d6) δ 7.42(d, J=6.84 Hz, 2H), 7.26(bs, 6H), 4.57(s, 4H), 3.90(s, 4H), 3.27(t, J =6.62 Hz, 4H), 2.94(t, J =6.44 Hz, 4H)
To a suspension of 1001 (256 mg, 1.00 mmol) in toluene (8 mL) was added 3- isochromanone (311 mg, 2.10 mmol) followed by trimethyl aluminum (2M in toluene, 1.0 mL, 2.00 mmol). The resulting mixture was stirred at 75°C 15 h, cooled to room temperature and diluted with ethyl acetate (50 mL). The organic layer was washed with water (3x20 mL), 10% sodium chloride solution (10 mL), dried (magnsesium sulfate) and concentrated under reduced pressure. The crude product was purified by HPLC to afford N,N
*-(5,5
*-(thiobis(ethane-2,l-diyl))bis(l,3,4-thiadiazole-5,2- diyl))bis(2-(2-(hydroxymethyl)phenyl)acetamide) (208, 62 mg). 1H NMR (300 MHz, DMSO-de) δ 7.4 l(s, 2H), 7.26(s, 6H), 4.56(s, 4H), 3.01(bs, 4H), 1.76(bs, 4H)
To a solution of 1015 (3.2g, 19.5mmol) in carbon tetrachloride (150mL) was added N-bromosuccinimide (3.47g, 19.6mmol) and benzoyl peroxide (lOmg, catalytic). The resulting mixture was refluxed overnight before it was filtered hot. The filtrate was concentrated under reduced pressure and the residue obtained was purified by silica gel chromatography eluting with 20% ethylacetate/hexane to afford 1016 (2g, 42% yield) as an oil. 1H NMR (300MHz, Chloroform-d) δ ppm 3.66 (s, 2H) 3.74 (s, 3H) 4.5 l(s, 2H) 7.35 (m, 4H).
To a solution of 1016 (0.243g, lmmol) in acetone (lOmL) was added 2-methyl imidazole (0.4 lg, 5mmol). The resulting mixture was refluxed overnight before it was concentrated under reduced pressure and the residue obtained was diluted with water (-lOOmL). The resulting solution was partitioned between water and ethyl acetate. The organic extract was washed with more water, separated, dried over sodium
sulfate, filtered and evaporated. The residue obtained was purified by silica gel chromatography eluting with MeOH/dichloromethane to afford 1017 (0.17g, 69% yield) as an oil. 1H NMR (300MHz, Chloroform-d) δ ppm 2.37 (s, 3H) 3.63 (s, 2H) 3.72 (s, 3H) 5.07 (s, 2H) 6.87 (s, 1H) 6.96-7.02 9m, 2H) 7.23-7.33 (m, 3H) To a solution of 1017 (0.17g, 0.69mmol) in THF/MeOH/Water (lOmL, 2mL, 2mL) was added lithium hydroxide monohydrate (0.06g, 1.42mmol). The resulting mixture was stirred at room temperature overnight before it was concentrated under reduced pressure. The residue obtained was diluted with water (~20mL) and the resulting solution was acidified with acetic acid. The aqueous layer was concentrated and the product was isolated by prep HPLC. The residue obtained was dissolved in water (5 mL) and concentrated hydrochloric acid (83 μί) was added to it before it was concentrated and dried to afford 1018 (0.15gm) as a hydrochloride salt.
To a suspension of carboxylic acid 1018 (105mg, 0.39mmol) in DMF (3mL) was added HATU (150mg, 0.39mmol) and stirred till reaction mixture is clear followed by the addition of an amine 1001 (50.5mg, 0.197mmol) and DIPEA (0.14mL, 0.8mmol). The resulting mixture was stirred at room temperature overnight before it was quenched by the addition of water. The solid separated was filtered, washed with water and dried to afford 296 (112mg, 83%). 1H NMR (300MHz, Dimethylsulfoxide- d6) δ ppm 1.76 (brs, 4H) 2.38 (s, 6H) 3.01(brs, 4H) 3.82 (s, 4H) 5.25 (s, 4H) 7.09- 7.38 (m, 12H) 12.64-12.67 (brs, 2H).
To a suspension of 1019 (1.5 g, 6.8 mmol) in CH2C12 (15 mL) at 0 °C was added Et3N (1.9 ml, 13.6 mmol) dropwise followed by phenyl acetyl chloride (1.07 ml, 8.1 mmol) dropwise. The resulting mixture was stirred at 0 °C and then slowly warmed up to room temperature for 2 days. The crude material was purified by silica gel chromatography eluting with 0-25% EtOAc in hexane to afford 1020.
To a solution of 4-bromo-l-butyne (7 g, 53 mmol) in DMSO (30 ml) at 0 °C was added Nal (7.94 g, 53 mmol). The mixture was stirred at room temperature for 2 h before it was cooled to 0 °C and followed by addition of NaCN (5.2 g, 106 mmol). The resulting mixture was heated at 80 °C for 2.5 h and then stirred at room temperature overnight. The mixture was partitioned between water and EtOAc. The organic extract was washed with water, dried over sodium sulfate, filtered and evaporated to afford 1021.
To a mixture of 1020 (400 mg, 1.18 mmol), PdCl2(PPh3)2 (41 mg, 0.059 mmol) and Cul (11 mg, 0.059 mmol) in Et3N (3 ml) and THF (6 ml) under argon atmosphere was added 1021 (187 mg, 2.36 mmol), then heated at 60 °C overnight. After removal of
the solvent, the residue was purified by silica gel chromatography eluting with 0-60% EtOAc in Hexane to afford 1022.
To a solution of 1022 (118 mg, 0.406 mmol) in the mixture of EtOAc (60 ml) and EtOH (15 ml) was added Pd(OH)2/C (50 mg, 0.356 mmol). Hydrogen was bubbled through the resulting mixture and stirred for 1 h. The Pd catalyst was filterd off and the filtrate was concentrated to afford 1023.
A mixture of 1023 (127 mg, 0.431 mmol) and thiosemicarbazide (51 mg, 0.561 mmol) in TFA (3 mL) was heated at 85 °C for 5 h. The reaction was cooled to room temperature and poured onto a mixture of ice-water. The mixture was basified with NaOH pellets (pH 10). The crude material was purified by silica gel chromatography eluting with 0-6% MeOH in CH2C12 to afford 1024.
To a solution of 1024 (38.4 mg, 0.104 mmol) in NMP (1 mL) at 0 °C was added phenyl acetyl chloride (0.017 mL, 0.125 mmol) dropwise. The resulting mixture was stirred at 0 °C for 1.5 h before it was quenched by addition of water (~10 mL). The mixture was partitioned between water and EtOAc. The organic extract was washed with water, dried over sodium sulfate, filtered and evaporated. The crude material was purified by silica gel chromatography eluting with 0-6% MeOH in CH2C12 to afford 295. 1H NMR (300 MHz, DMSO-d6) δ 12.65 (s, 1H), 11.26 (s, 1H), 8.22-8.19 (d, J= 8.82 Hz, 1H), 7.58-7.54 (d, J= 9.72 Hz, 1H), 7.36-7.28 (m, 10H), 3.81-3.78 (d, J= 8.43 Hz, 4H), 3.01 (bs, 2H), 2.90 (bs, 2H), 1.73 (bs, 4H).
Compound 1024 can also be prepared according to the following procedure:
To a solution of 3-amino-6-chloropyridazine (11.14 g, 86.0 mmol) in NMP (279 mL) at 19°C was added phenylacetyl chloride (18.2 mL, 137.6 mmol) dropwise over 5 minutes with the internal temperature of the solution maintained T
z < 28 °C. The resulting mixture was stirred at 19°C for 90 minutes and poured into ice water (557 mL). The white precipitate was collected by suction filtration, rinsed with water
(2x110 mL) and diethyl ether (110 mL). The product was dried overnight under high vacuum to afford N-(6-chloropyridazin-3-yl)-2-phenylacetamide (xxx, 18.8 g). 1H NMR (300 MHz, DMSO-d6) δ 11.57(s, 1H), 8.40(d, J=9.636 Hz, 1H), 7.90(d, J=9.516 Hz, 1H), 7.36(m, 5H) 3.82(s, 2H) A 1000 mL three-neck flask fitted with internal temperature probe and addition funnel was flushed with Α%). Under positive Argon pressure 4-cyanobutylzinc bromide (0.5M in THF, 500mL, 250 mmol) was charged into the addition funnel then added to the reaction vessel at room temperature. Solid N-(6-chloropyridazin-3-yl)-2- phenylacetamide (20.6 g, 83.3 mmol) was added to the stirred solution at RT under Ar(g) flow, followed by the addition of NiCl2(dppp) (4.52 g, 8.33 mmol). The resulting mixture was stirred at 19°C for 240 minutes and then quenched with ethanol (120 mL). Water (380mL) added to the stirred red solution, giving a thick precipitate. Ethyl acetate (760 mL) added and stirred well for 30 minutes. The solids were removed by filtration through a pad of celite. The mother liquor was then transferred to a separatory funnel and the organic layer was washed with H20 (380mL), 0.5% ethylenediammetetraacetic acid solution (380 mL) and again with H20 (380mL). The organic layer was concentrated by rotoevaporation. Resulting red oil was redissolved in EtOAc (200 mL) and 1M HC1 (380 mL) was added to the well stirred flask. After 30 minutes the mixture was transferred to separatory funnel and the aqueous layer collected. The organic layer was extracted with 1M HC1 (2x380mL). The aqueous layer's pH was then adjusted to ~7 using 7.5% sodium bicarbonate solution and the pale yellow precipitate was collected by suction filtration, rinsed with water (200 mL) and diethyl ether (2x200mL). The solid was dried overnight under high vacuum to afford N-(6-(4-cyanobutyl)pyridazin-3-yl)- 2-phenylacetamide (1023, 14.76 g). 1H NMR (300 MHz, DMSO-d6) δ 11.29(s, 1H), 8.23(d, J=9.036 Hz, 1H), 7.59(d,
J=9.246 Hz, 1H), 7.32(m, 5H), 3.79(s, 2H), 2.90(t, J= 7.357 Hz, 2H), 2.56(t, J= 7.038 Hz, 2H), 1.79(t, J= 7.311 Hz, 2H), 1.63(t, J= 7.01 Hz, 2H)
N-(6-(4-cyanobutyl)pyridazin-3-yl)-2-phenylacetamide (14.7 g, 50.2 mmol) was charged into a 250 mL round bottom flask fitted with an open top reflux condenser. To the flask was added thiosemicarbazide (5.03 g, 55.2 mmol) and trifluoroacetic acid (88 mL). The reaction slurry was heated in a 65°C bath for 2 h. After cooling to RT, H20 (150 mL) was added and stirred for 30 minutes. The mixture was then slowly transferred to a stirred 7.5% sodium bicarbonate solution (1400mL) cooled in a 0°C bath. The precipitate was collected by suction filtration, rinsed with water (2x200 mL), diethyl ether (2x200mL) and dried under high vacuum overnight. The off- white solid was slurried in DMSO (200 mL) and heated in an 80°C bath until the internal temperature reached 65°C. DMSO (105 mL) was used to rinse sides of flask. H20 (120 mL) was slowly added until the solution became slightly cloudy and then the mixture was removed from heat bath and allowed to cool to ambient temperature while stirring. The pale green precipitate was collected by suction filtration, rinsed with water (200 mL) and diethyl ether (2x200mL). The solid was dried overnight under high vacuum to provide N-(6-(4-(5-amino-l,3,4-thiadiazol-2- yl)butyl)pyridazin-3-yl)-2-phenylacetamide (1024, 15.01 g). 1H NMR (300 MHz, DMSO-de) δ 11.28(s, 1H), 8.23(d, J=8.916 Hz, 1H), 7.59(d, J=8.826 Hz, 1H), 7.36(m, 5H), 7.07(s, 2H), 3.78(s, 2H), 2.87(t, J= 6.799 Hz, 4H), 1.69(bm, 4H)
To a solution of dimethyl adipate (28.7 mmol, 5.0 g, 4.7 mL, 1.0 equiv.) in 20 mL of MeOH was added anhydrous hydrazine (229.6 mmol, 7.36 g, 7.51 mL, 8.0 equiv.) and the mixture heated to 50°C, giving a white precipitate. The mixture was heated for one hour and then allowed to cool to room temperature. The white solid was collected by filtration and washed with additional MeOH then dried under high vacuum giving 4.6 g of adipohydrizide.
1HNMR (300 MHz, DMSO-d
6) δ 8.91 (s, 2H), 4.14 (s, 4H), 2.00 (br s, 4H), 1.46 (br s, 4H).
To a 0°C cooled slurry of adipohydrizide (12.49 mmol, 4.0 g, 1.0 equiv.), potassium bicarbonate (15.61 mmol, 1.56 g, 1.25 equiv.) in 25 mL of MeOH was added solid cyanogen bromide (13.74 mmol, 1.44 g, 1.1 equiv.) in one portion. This mixture was stirred at 0°C and allowed to warm to RT over one hour and then stirred overnight. The volatiles were removed under reduced pressure and the solids diluted with water. The pH was adjusted to 12 with 2.5 N NaOH and the solids collected by filtration. The white solid was washed with water and dried under high vacuum to give 1.73 g of oxadiazole 1025.
1HNMR (300 MHz, DMSO-d
6) δ 6.85 (s, 4H), 2.68 (s, 4H), 1.68 (s, 4H).
To a suspension of oxadiazole 1025 (181 mg, 0.81 mmol) in NMP (9 mL) was added triethylamine (0.564 mL, 4.05 mmol) and the mixture warmed to 70°C. The mixture was allowed to stir for 30 minutes followed by the addition of phenylacetyl chloride (0.234 mL, 1.77 mmol). The reaction temperature was held at 70°C for 15 hours then allowed to cool to room temperature. The crude reaction mixture was purified by reverse phase HPLC giving 305 (0.015 g). 1HNMR (300 MHz, DMSO-d6) δ 11.74(s, 2H), 7.33(s, 10H), 3.74(s, 4H), 2.85(s, 4H), 1.76(s, 4H).
Functionalization of diacylated cores:
To a suspension of 21 (2.25 g, 4.57 mmol) in a mixture of THF (250 mL) and H
20 (20 mL) at room temperature was added NaOH (1.83 g, 45.67 mmol) and
formaldehyde solution (37% in water, 14.83 mL, 182.70 mmol). The resulting mixture was heated at 60 °C for 7 h before it was cooled to 0 °C and acidified to pH 7 with aq. HCl solution. The white precipitate was collected by suction filtration, rinsed
with water and dried to provide N,N'-[5,5'-(butane-l,4-diyl)-bis(l,3,4-thiadiazole-5,2- diyl)]-bis(3-hydroxy-2-phenylpropanamide) (36, 624 mg). The 2
nd precipitation from the filtrate provided additional product (1.29 g). 1H NMR (300 MHz, DMSO-d
6) δ 12.65 (bs, 2H), 7.35-7.30 (m, 10H), 5.09 (bs, 2H), 4.10-4.02 (m, 4H), 3.61 (d, J= 8.1 Hz, 2H), 3.02 (bs, 4H), 1.76 (bs, 4H).
To a suspension of 199 (300 mg, 0.572 mmol) in a mixture of THF (50 mL) and MeOH (5 ml) was added potassium carbonate (158 mg, 1.144 mmol) and
formaldehyde solution (37% in water, 2 mL). The resulting mixture was stirred at room temperature for 48 h before it was cooled to 0 °C and acidified to pH 7 with aq, HC1 solution. The white precipitate was collected by suction filtration, rinsed with water and dried. The crude material was purified by HPLC to afford 29. 1H NMR (300 MHz, DMSO-d6) δ 7.34-7.26 (m, 10H), 4.13-4.02 (m, 2H), 3.81 (s, 2H), 3.62 (m, 2H), 3.24 (t, 4H), 2.93 (t, 4H).
To a suspension of 199 (2.0 g, 3.81 mmol) in a mixture of THF (250 mL) and MeOH (20 ml)H20 (20 mL) at room temperature was added IN NaOH (20 ml) and formaldehyde solution (37% in water, 15 mL). The resulting mixture was heated at 50 °C overnight before it was cooled to 0 °C and acidified to pH 7 with aq. HC1 solution. The white precipitate was collected by suction filtration, rinsed with water and dried. The crude material was purified by HPLC to afford 24. 1H NMR (300 MHz, DMSO-de) δ 12.67 (bs, 2H), 7.36-7.30 (m, 10H), 5.10 (bs, 2H), 4.10-4.02 (m, 4H), 3.61 (d, 2H), 3.27 (t, 4H), 2.95 (t, 4H).
Prodrugs:
To a flask containing N,N'-(5,5'-(thiobis(ethane-2,l-diyl))bis(l,3,4-thiadiazole-5,2- diyl))bis(2-phenylacetamide) (1) (9.4 mmol, 5.0 g, 1.0 equiv.) was added 100 mL DMF, K2C03 (20.98 mmol, 2.89 g, 2.2 equiv.), and chloromethyl butyrate (20.98 mmol, 2.86 g, 2.62 mL, 2.2 equiv.). The mixture stirred at room temperature for 15 hours then diluted with 200 mL water and 200 mL EtOAc. The layers were separated and the aqueous layer extracted with EtOAc (2 x 100 mL) and the organic layers combined, washed with water, brine and dried over Na2S04. The Na2S04 was removed by filtration and the volatiles removed under reduced pressure. The compounds were purified by reverse phase chromatography (MeCN, H20) giving 0.235 g of compound 8 and 0.126 g of compound 7.
1HNMR (300 MHz, DMSO, d6) Compound 8: δ 7.31 (m, 10H), 6.18 (s, 4H), 3.82 (s, 4H), 3.17 (dd, 2H, J=6.8 Hz), 2.92 (dd, 2H, J=6.8 Hz), 2.93 (m, 4H), 2.32 (dd, 2H, J=7.2 Hz), 1.54 (dt, 2H, J=7.2, 7.4 Hz), 0.87 (t, 3H, J= 7.4Hz).
1HNMR (300 MHz, DMSO, d6) Compound 7: δ 12.68 (s, 1H), 7.32 (m, 10H), 6.18 (s, 2H), 3.82 (s, 4H), 3.26 (dd, 2H, J=7.0 Hz), 3.17 (dd, 2H, J=6.8 Hz), 2.93 (m, 4H), 2.32 (dd, 2H, J =7.2 Hz), 1.54 (dt, 2H, J =7.2, 7.4 Hz), 0.87 (t, 3H, J= 7.4Hz).
To a suspension of 3-morpholin-4-yl-propionic acid hydrochloride (500 mg, 2.56
mmol) in DMF (20 mL) at 0 °C was added N-(3-dimethylaminopropyl)-N'- ethylcarbodiimide hydrochloride (534 mg, 2.79 mmol). The resulting mixture was stirred at 0 °C for 40 min and followed by addition of diol 36 (642 mg, 1.16 mmol) and 4-DMAP (454 mg, 3.72 mmol). The resulting mixture was stirred from 0 °C to room temperature over a period of 3.5 h before it was diluted with EtO Ac and cold water. The organic layer was separated and washed with water (3><50 mL), brine, dried (MgS04) and concentrated. The crude product was purified by silica gel chromatography eluting with 10-25% MeOH in EtO Ac to provide { [5, 5 '-(butane- 1, 4- diyl)-bis(l,3,4-thiadiazole-5,2-diyl)]-bis(azanediyl)}-bis(3-oxo-2-phenylpropane-3,l- diyl)-bis(3-morpholinopropanoate) (188, 340 mg) and a less polar product, 3-((5-{4- [5-(3-hydroxy-2-phenylpropanamido)-l,3,4-thiadiazol-2-yl]butyl}-l,3,4-thiadiazol-2- yl)amino)-3-oxo-2-phenylpropyl 3-morpholinopropanoate (228, 103 mg). 188: 1H NMR (300 MHz, DMSO-d6) δ 12.80 (s, 2H), 7.39 (m, 10H), 4.62 (t, J= 9.6 Hz, 2H), 4.33-4.27 (m, 4H), 3.48 (bs, 8H), 3.02 (bs, 4H), 2.45 (bs, 8H), 2.25 (bs, 8H), 1.76 (bs, 4H).
228: 1H NMR (300 MHz, MeOD-d
4) δ 7.43-7.37 (m, 10H), 4.71 (t, J= 10.5 Hz, 1H), 4.41 (m, 1H), 4.30-4.24 (m, 2H), 4.06-4.03 (m, 1H), 3.80-3.76 (m, 1H), 3.62 (bs, 4H), 3.11 (bs, 4H), 2.63-2.52 (m, 4H), 2.40 (bs, 4H), 1.90 (bs, 4H).
s
U
1035 1041
To a solution of diethyl trans- 1 ,2-cyclopropanedicarboxylate (5.00 g, 26.85 mmol) in THF (20 mL) at 0 °C was added a solution of LAH (67.13 mL, 1.0 M in THF, 67.13 mmol) dropwise. The resulting mixture was stirred at 0 °C for 1.5 h before it was quenched with H20 (20 mL), 2N aq. NaOH (20 mL) and H20 (20 mL). The mixture was stirred vigorously for 1 h at room temperature before it was filtered through a
plug of celite. The filtrate was dried (MgSC^) and concentrated to provide the desired diol (2.73 g) as a colorless oil.
A mixture of the diol (2.00 g, 19.58 mmol) in CH2C12 (75 mL) at 0 °C was added pyridine (6.34 mL, 78.33 mmol) and followed by MsCl (3.33 mL, 43.08 mmol) dropwise. The resulting mixture was stirred 0 °C for 1 h before it was warmed up to room temperature. The reaction was quenched with H20 and diluted with ether. The organic layer was washed with brine, dried (MgSC^) and concentrated to provide 1039. This crude product was dissolved in DMSO (75 mL), and added NaCN (2.88 g, 58.75 mmol) and Nal (294 mg, 1.96 mmol). The resulting mixture was heated at 45 °C for 8 h before it was allowed to cool to room temperature and diluted with EtOAc and H20. The organic layer was separated, washed with brine, dried (MgSC^) and concentrated to provide the crude product 1040 which was used in the following step without purification.
A mixture of 1040 and thiosemicarbazide (3.75 g, 41.12 mmol) in trifluoroacetic acid (TFA) (20 mL) was heated at 80 °C for 5 h. The reaction was cooled to room temperature and poured into a mixture of ice and water. Sodium hydroxide pellets were added to the mixture until it was basic (pH 14). The white precipitate was collected by suction filtration, rinsed with water, ether and dried to provide 1041 (472 mg). To a suspension of 1041 (70 mg, 0.26 mmol) in l-Methyl-2-pyrrolidinone (NMP) (5 mL) at 0 °C was added phenylacetyl chloride (72 μί, 0.55 mmol) dropwise. The resulting mixture was stirred at 0 °C for 1 h before it was quenched by addition of water (~3 mL). The white precipitate was collected by suction filtration, rinsed with water and dried to provide 1035 (37 mg). 1H NMR (300 MHz, DMSO-d6) δ 12.65 (s, 2H), 7.34-7.27 (m, 10H), 3.82 (s, 4H), 3.04 - 2.75 (m, 4H), 1.14-1.12 (m, 2H), 0.63- 0.59 (m, 2H).
1038 1037
To a solution of 1020 (1.50 g, 4.42 mmol), ethynyltrimethylsilane (813 uL, 5.75 mmol), PdCl2(PPh3)2 (310 mg, 0.44 mmol) and Cul (59 mg, 0.31 mmol) in THF (20 mL) under argon atmosphere at room temperature was added Et3N (6.16 mL, 44.23 mmol). The resulting mixture was heated at 50 °C for 5 h before it was allowed to cool to room temperature and filtered through a plug of celite. The filtrate was concentrated and the crude residue was purified by flash column chromatography over silica gel eluting with 10-50% EtOAc in hexanes to provide the desired product (1.21 g) as a solid. A mixture of the foregoing intermediate (1.07 g, 3.48 mmol) and K2C03 (0.40 g, 2.90 mmol) in MeOH (100 mL) was stirred at room temperature for 5 h before it was concentrated under reduced pressure. The residue was re-dissolved in a mixture of EtOAc and H20, and was neutralized with IN aq. HCl solution to pH 7. The organic layer was separated, washed with brine, dried (MgS04) and concentrated. The crude residue was purified by flash column chromatography over silica gel eluting with 10- 50% EtOAc in hexanes to provide the desired alkyne 1036 (0.48 g) as a white solid.
To a solution of alkyne 1036 (52 mg, 0.22 mmol) in pyridine (5 mL) at room temperature was added CuCl (4.3 mg, 0.04 mmol). The resulting mixture was stirred under a stream of air for 40 min as all of the starting material was consumed. The reaction mixture was diluted with saturated aq. NH4C1 solution (~2 mL). The off- white precipitate was collected by suction filtration, washed with H20 and dried. This crude bis-acetylene product 1037 (52 mg) was used in the following step without further purification.
A mixture of 1037 (52 mg) and Pd(OH)2/C (100 mg) in a mixture of DMF (5 mL) and
THF (10 mL) was stirred at room temperature under 1 atmosphere of H2 for 3 h as all
of the starting material was consumed. The palladium catalyst was filtered off and the filtrate was concentrated. The crude residue was purified by column chromatography over silica gel eluting with 1-10% MeOH in CH2CI2 to provide the desired product 1038 (18 mg) as a solid. 1H NMR (300 MHz, DMSO-d6) δ 11.26 (s, 2H), 8.20 (d, J = 8.97 Hz, 2H), 7.56 (d, J= 8.77 Hz, 2H), 7.36-7.24 (m, 10H), 3.78 (s, 4H), 2.90 (bs,
To a solution of adiponitrile (19.02 g, 175.8 mmol) in TFA (50 mL) was added thiosemicarbazide (16.02 g, 175.8 mmol) and the mixture heated to 70°C for 4 hours under an atmosphere of Argon. The mixture was allowed to cool to room temperature and the volatiles removed under reduced pressure. The residue was diluted with water (200 mL) and the pH adjusted to 7 with solid NaOH giving a white precipitate that was collected by filtration and washed with water. The solids were dried under high vacuum giving 9.22 g of 1081. 1HNMR (DMSO, d6): δ 7.02 (br s, 2H) 2.84 (m, 2H), 2.55 (m, 2H), 1.67 (m, 4H).
To a solution of 1081 (0.625 g, 2.87 mmol) in NMP (12.5 mL) was added
phenylacetyl chloride (0.487 g, 0.42 mL, 3.15 mmol) dropwise and the mixture stirred at room temperature for one hour under an atmosphere of Argon. The mixture was poured into water (100 mL) and the solids collected by filtration. The solids were washed with water and dried under high vacuum to give 0.805 g of 1082. 1HNMR
(DMSO, d6): δ 12.65 (s, 1H) 7.31 (m, 5H), 3.80 (s, 2H), 3.00 (t, 2H, J= 7.3 Hz), 2.53 (t, 2H, J= 7.1 Hz), 1.78 (dq, 2H, J= 7.3, 7.1 Hz), 1.61 (dq, 2H, j= 7.3, 7.1 Hz).
To a solution of 1082 (0.49 g, 1.33 mmol) in TFA (10 mL) was added
thiosemicarbazide (0.23 g, 1.46 mmol) and the mixture heated at 70°C overnight under an atmosphere of Argon. The mixture was allowed to cool to room temperature and the volatiles removed under reduced pressure. The residue was diluted with water (50 mL) and the pH adjusted to 7 with solid NaOH giving a white precipitate that was collected by filtration and washed with water. The solids were dried under high vacuum giving 0.367 g of 1083. 1HNMR (DMSO, d6): δ 12.70 (s, 1H) 7.34 (br s, 5H), 7.16 (s, 2H), 3.82 (s, 2H), 3.01 (s, 2H), 2.84 (S, 2H), 1.71 (br s, 4H).
To a solution of 1083 (0.10 g, 0.267 mmol), 2,4-difluoro-3-methoxyphenylacetic acid (0.058 g, 0.267 mmol), EDC (0.127 g, 0.667 mmol), HOBt (0.090 g, 0.667 mmol) in DMF (4 mL) was added DIEA (0.171 g, 0.231 mL, 1.335 mmol) and the mixture stirred overnight under an atmosphere of Argon. The mixture was poured into water (20 mL) and the solids formed were collected by filtration, washed with water and dried under high vacuum. The crude 1084 was used in the following step without purification. To a solution of 1084 (0.050 g, 0.091 mmol) in dichloromethane (1 mL) was added BBr3 (1.0 mL, 1 mmol, 1.0 M in dichloromethane) and the mixture stirred for 4 hours at room temperature under an atmosphere of Argon. The volatiles were removed under reduced pressure and the residue diluted with dichloromethane (5 mL). The volatiles were removed under reduced pressure and the residue diluted with water (15 mL) and the pH adjusted to 12. The aqueous layer was washed with dichloromethane (4 x 5 mL) and the pH adjusted to 4. The solids were collected by filtration, washed with water and dried under high vacuum giving 0.029 g of 346. 1HNMR (DMSO, d6): δ 12.66 (s, 2H), 10.12 (s, 1H), 7.33 (s, 5H), 7.00 (m, 1H), 6.80 (m, 1H), 3.84 (s, 2H), 3.81 (s, 2H), 3.02 (br s, 4H), 1.76 (br s, 4H).
To a solution of 1083 (0.05 g, 0.133 mmol), Boc-3-aminomethyl-phenylacetic acid (0.035 g, 0.133 mmol), EDC (0.064 g, 0.332 mmol), HOBt (0.045 g, 0.332 mmol) in DMF (8 mL) was added DIEA (0.086 g, 0.115 mL, 0.665 mmol) and the mixture stirred overnight under an atmosphere of Argon. The mixture was poured into water (20 mL) and the solids formed were collected by filtration, washed with water and dried under high vacuum to give 0.023 g of 375.
1HNMR (DMSO, d
6): δ 12.66 (s, 2H), 7.27 (m, 10H), 4.11 (br s, 2H), 3.81 (s, 2H), 3.79 (s, 2H), 3.01(br s, 4H), 1.76 (br
A flask was charged with 1024 (100 mg, 0.27 mmol), tropic acid (54 mg, 0.326 mmol) in DMF (2 ml) at 0 °C was added HOBT (88 mg, 0.652 mmol) followed by EDCI (156 mg, 0.815 mmol). The resulting mixture was slowly warmed up to room temperature and stirred for 3 h before it was quenched by addition of water (~10 mL) The white precipitate was collected by suction filtration, rinsed with more water and dried to afford 314. 1H NMR (300 MHz, DMSO-d6) δ 12.65 (s, 1H), 11.26 (s, 1H), 8.22-8.19 (d, J= 8.82 Hz, 1H), 7.58-7.54 (d, J= 9.72 Hz, 1H), 7.36-7.28 (m, 10H), 4.10-4.05 (m, 2H), 3.78 (s, 3H), 3.65 (s, 1H), 3.01 (bs, 2H), 2.90 (bs, 2H), 1.73 (bs,
A flask was charged with 1024 (500 mg, 1.36 mmol), DL-mandelic acid (248 mg, 1.63 mmol) in DMF (10 ml) at 0 °C was added HOBT (441 mg, 3.26 mmol) followed
by EDCI (781 mg, 4.08 mmol). The resulting mixture was stirred at 0 C for 10 minutes then warmed up to room temperature and stirred for 10 minutes before it was quenched by addition of water (-50 mL) at 0 °C. The white precipitate was collected by suction filtration, rinsed with more water and dried to afford 315. 1H NMR (300 MHz, DMSO-de) δ 12.65 (s, 1H), 11.26 (s, 1H), 8.22-8.19 (d, J= 8.82 Hz, 1H), 7.58- 7.50 (m, 3H), 7.36-7.28 (m, 8H), 6.35 (s, 1H), 5.32 (s, 1H), 3.78 (s, 2H), 3.01 (bs, 2H), 2.90 (bs, 2H), 1.73 (bs, 4H).
To a suspension of 3-morpholin-4-yl-propionic acid hydrochloride (209 mg, 1.07 mmol) in DMF (10 ml) was added EDCI (308 mg, 1.61 mmol). The resulting mixture was stirred at 0 °C for 1 hour and followed by addition of 315 (447 mg, 0.889 mmol) and 4-DMAP (261 mg, 2.14 mmol). The resulting mixture was stirred from 0 °C to room temperature over a period of 6 h before it was quenched by addition of ice water (~50mL). The white precipitate was collected by suction filtration, rinsed with more water. The crude material was purified by silica gel chromatography eluting with 0- 6% MeOH in EtOAc to afford 334. 1H NMR (300 MHz, DMSO-d6) δ 12.95 (s, 1H), 11.26 (s, 1H), 8.22-8.19 (d, J= 9.45 Hz, 1H), 7.58-7.26 (m, 11H), 6.14 (s, 1H), 3.78 (s, 2H), 3.54 (bs, 4H), 3.01 (bs, 2H), 2.90 (bs, 2H), 2.63 (bs, 4H), 2.38 (bs, 4H), 1.73
Compound 317 was prepared according to the procedure above for compound 315.
1H NMR (300 MHz, DMSO-d6) δ 12.40 (s, 1H), 11.26 (s, 1H), 8.22-8.19 (d, J= 9.03 Hz, 1H), 7.58-7.54 (d, J= 9.72 Hz, 1H), 7.36-6.87 (m, 9H), 6.35 (bs, 1H), 5.30 (s, 1H), 3.78 (m, 5H), 3.01 (bs, 2H), 2.90 (bs, 2H), 1.73 (bs, 4H).
Compound 318 was prepared according to the procedure above for compound 315.
1H NMR (300 MHz, DMSO-d6) δ 12.50 (s, 1H), 11.26 (s, 1H), 8.22-8.19 (d, J= 9.43 Hz, 1H), 7.60-7.27 (m, 10H), 6.51 (bs, 1H), 5.35 (s, 1H), 3.78 (s, 2H), 3.01 (bs, 2H), 2.90 (bs, 2H), 1.73 (bs, 4H).
A flask was charged with 1024 (50 mg, 0.135 mmol), 3-chlorophenylacetic acid (28 mg, 0.163 mmol) in DMF (1 ml) at 0 °C was added HOBT (44 mg, 0.326 mmol) followed by EDCI (78 mg, 0.408 mmol). The resulting mixture was slowly warmed up to room temperature and stirred for 1 h before it was quenched by addition of water (~5 mL). The white precipitate was collected by suction filtration, rinsed with more water and ether then dried to afford 335. 1H NMR (300 MHz, DMSO-d6) δ 12.65 (s, 1H), 11.26 (s, 1H), 8.22-8.19 (d, J= 8.82 Hz, 1H), 7.58-7.54 (d, J= 9.72 Hz, 1H), 7.36-7.28 (m, 9H), 3.84 (s, 2H), 3.78 (s, 2H), 3.01 (bs, 2H), 2.90 (bs, 2H), 1.73 (bs, 4H).
Compound 337 was prepared according to the procedure above for compound 335.
1H NMR (300 MHz, DMSO-d
6) δ 12.65 (s, 1H), 11.26 (s, 1H), 9.38 (s, 1H), 8.22-8.19 (d, J= 8.37 Hz, 1H), 7.58-7.54 (d, J= 9.63 Hz, 1H), 7.36-7.09 (m, 6H), 6.75-6.65 (m, 3H), 3.78 (s, 2H), 3.70 (s, 2H), 3.01 (bs, 2H), 2.90 (bs, 2H), 1.73 (bs, 4H).
339, 341, 382: A flask was charged with 1024 (100 mg, 0.27 mmol), Boc-3- aminomethyl-phenylacetic acid (86 mg, 0.325 mmol) in DMF (2 ml) at 0 °C was added HOBT (88 mg, 0.65 mmol) followed by EDCI (156 mg, 0.812 mmol). The resulting mixture was stirred at 0 °C for 5 minutes then warmed up to room
temperature and stirred for 1.5 h before it was quenched by addition of water (~10 mL) at 0 °C. The white precipitate was collected by suction filtration, rinsed with more water and ether then dried to afford 339. 1H NMR (300 MHz, DMSO-d6) δ 12.65 (s, 1H), 11.26 (s, 1H), 8.22-8.19 (d, J= 8.82 Hz, 1H), 7.58-7.54 (d, J= 9.42 Hz, 1H), 7.36-7.13 (m, 9H), 4.13-4.11 (d, J= 10.62, 2H), 3.78 (s, 4H), 3.01 (bs, 2H), 2.90 (bs, 2H), 1.73 (bs, 4H), 1.38 (s, 9H).
To a suspension of 339 (50 mg, 0.081 mmol) in dichloromethane (2 ml) was added TFA (2 ml) at 0 °C. The resulting mixture was stirred at room temperature for 20 minutes before it was evaporated under vacuo to dryness. Ether was added and the white precipitate was collected by suction filtration, rinsed with more ether and dichloromethane then dried to afford 341. 1H NMR (300 MHz, DMSO-d6) δ 12.65 (s, 1H), 11.26 (s, 1H), 8.22-8.19 (d, J= 8.82 Hz, 1H), 8.14-8.11 (bs, 2H), 7.58-7.54 (d, J = 9.42 Hz, 1H), 7.36-7.13 (m, 9H), 4.06-4.03 (m, 2H), 3.84 (s, 2H), 3.78 (s, 2H), 3.01 (bs, 2H), 2.90 (bs, 2H), 1.73 (bs, 4H). To a solution of 341 (10 mg, 0.0159mmol) in DMF (1 ml) at 0 °C was added triethylamine (4.4 ul, 0.0317 mmol) drop wise followed by ethyl chloroformate (1.8 ul, 0.0191 mmol) drop wise. The resulting mixture was slowly warmed up to room temperature and stirred for 30 minutes before it was quenched by addition of water
(~1 mL) at 0 C. The mixture was partitioned between water and EtOAc. The organic extract was washed with water, dried over sodium sulfate, filtered and evaporated. The crude material was purified by silica gel chromatography eluting with 0-6% MeOH in CH2C12 to afford 382. 1H NMR (300 MHz, DMSO-d6) δ 12.65 (s, 1H), 11.26 (s, 1H), 8.22-8.19 (d, J= 8.82 Hz, 1H), 7.67-7.58 (bs, 1H), 7.58-7.54 (d, J= 9.42 Hz, 1H), 7.36-7.13 (m, 9H), 4.18-4.16 (m, 2H), 4.06-4.0 (q, 2H), 3.78 (s, 4H), 3.01 (bs, 2H), 2.90 (bs, 2H), 1.73 (bs, 4H), 1.19-1.13 (t, 3H).
Compound 431 was prepared according to the procedure above for compound 382 with the appropriate reagents. 1H NMR (300 MHz, DMSO-d6) δ 12.65 (s, 1H), 11.26 (s, 1H), 8.35 (s, 1H), 8.22-8.19 (d, J= 8.88 Hz, 1H), 7.57-7.54 (d, J= 9.51 Hz, 1H), 7.38-7.15 (m, 9H), 4.25-4.24 (d, J= 5.64 Hz, 2H), 3.76 (s, 4H), 3.01 (bs, 2H), 2.90 (bs, 2H), 1.87 (s, 3H), 1.73 (bs, 4H).
Compound 432 was prepared according to the procedure above for compound 382 with the appropriate reagents. 1H NMR (300 MHz, DMSO-d6) δ 12.63 (s, 1H), 11.26 (s, 1H), 9.04-9.01 (m, 1H), 8.22-8.19 (d, J= 8.91 Hz, 1H), 7.93-7.89 (d, J= 9.51 Hz, 2H), 7.58-7.25 (m, 13H), 4.50-4.48 (d, J= 5.91 Hz, 2H), 3.78 (s, 4H), 3.01 (bs, 2H), 2.90 (bs, 2H), 1.73 (bs, 4H).
Compound 433 was prepared according to the procedure above for compound 382 with the appropriate reagents. 1H NMR (300 MHz, DMSO-d
6) δ 12.63 (s, 1H), 11.26 (s, 1H), 8.31-8.21 (m, 1H), 8.20-8.19 (d, J= 9.57 Hz, 1H), 7.57-7.54 (d, J= 8.73 Hz, 1H), 7.35-7.13 (m, 9H), 4.26-4.24 (d, J= 5.52 Hz, 2H), 3.78 (s, 4H), 3.01 (bs, 2H),
-0.85 (d, J= 3.99 Hz, 6H).
To a solution of 341 (70 mg, 0.11 lmmol) in DMF (1 ml) at 0 UC was added triethylamine (31 ul, 0.22 mmol) drop wise followed by 5-bromovaleryl chloride (12 ul, 0.122 mmol) drop wise. The resulting mixture was slowly warmed up to room temperature and stirred for lh. Potassium tert-butoxide (50 mg, 0.445 mmol) was then added to the reaction mixture at 0 °C. The resulting mixture was slowly warmed up to room temperature and stirred for overnight before it was quenched by addition of water (~2 mL) at 0 °C. The mixture was partitioned between water and EtOAc. The organic extract was washed with water, dried over sodium sulfate, filtered and evaporated. The crude material was purified by silica gel chromatography eluting with 0-6% MeOH in CH2C12 to afford 476. 1H NMR (300 MHz, DMSO-d6) δ 12.65
(s, 1H), 11.26 (s, 1H), 8.22-8.19 (d, J= 8.82 Hz, 1H), 7.58-7.54 (d, J= 9.42 Hz, 1H), 7.36-7.13 (m, 9H), 4.50 (s, 2H), 3.78 (s, 4H), 3.35 (bs, 2H), 3.20 (bs, 2H), 3.01 (bs,
, 1.68-1.80 (d, 6H).
Compound 340 was prepared according to the procedure above for compound 315 with the appropriate reagents. 1H NMR (300 MHz, DMSO-d
6) δ 12.50 (s, 1H), 11.26 (s, 1H), 8.22-8.19 (d, J= 9.24 Hz, 1H), 7.60-7.27 (m, 10H), 6.51 (bs, 1H), 5.35 (s, 1H), 3.78 (s, 2H), 3.01 (bs, 2H), 2.90 (bs, 2H), 1.73 (bs, 4H).
Compound 349 was prepared according to the procedure above for compound 315 with the appropriate reagents. 1H NMR (300 MHz, DMSO-d6) δ 12.41 (s, 1H), 1 1.26 (s, 1H), 8.22-8.19 (d, J = 8.76 Hz, 1H), 7.58-7.27 (m, 1 1H), 6.36 (s, 1H), 5.34 (s, 1H),
(bs, 2H), 1.73 (bs, 4H).
Compound 350 was prepared according to the procedure above for compound 315 with the appropriate reagents. 1H NMR (300 MHz, DMSO-d6) δ 12.41 (s, 1H), 11.26 (s, 1H), 8.22-8.19 (d, J= 8.67 Hz, 1H), 7.58-7.27 (m, 11H), 6.34 (s, 1H), 5.34 (s, 1H),
2H), 1.73 (bs, 4H).
Compound 351 was prepared according to the procedure above for compound 315 with the appropriate reagents. 1H NMR (300 MHz, DMSO-d
6) δ 12.50 (s, 1H), 1 1.26 (s, 1H), 8.21-8.18 (d, J= 8.67 Hz, 1H), 7.58-7.54 (d, J= 9.72 Hz, 1H), 7.36-7.23 (m, 8H), 6.67 (s, 1H), 5.40 (s, 1H), 3.78 (s, 2H), 3.01 (bs, 2H), 2.90 (bs, 2H), 1.73 (bs, 4H).
To a solution of 1024 (50 mg, 0.136 mmol) in DMF (1 ml) at 0 UC was added triethylamine (38 ul, 0.271 mmol) drop wise followed by benzyl isocyanate (20 ul, 0.163 mmol) drop wise. The resulting mixture was slowly warmed up to room temperature and stirred for 40 minutes before it was quenched by addition of water (~5 mL) at 0 °C. The white precipitate was collected by suction filtration, rinsed with more water. The crude material was purified by silica gel chromatography eluting with 0-6% MeOH in CH2C12 to afford 352. 1H NMR (300 MHz, DMSO-d6) δ 11.26 (s, 1H), 10.82 (s, 1H), 8.22-8.19 (d, J= 9.42 Hz, 1H), 7.58-7.54 (d, J= 8.79 Hz, 1H), 7.36-7.31 (m, 10H), 7.06 (bs, 1H), 4.37-4.35 (d, J= 5.22 Hz, 2H), 3.78 (s, 2H), 2.99- 2.90 (m, 4H), 1.73 (bs, 4H).
Compound 353 was prepared according to the procedure above for the preparation of compound 335. 1H NMR (300 MHz, DMSO-d6) δ 12.57 (s, 1H), 11.26 (s, 1H), 8.22- 8.19 (d, J= 9.45 Hz, 1H), 7.57-7.54 (d, J= 9.48 Hz, 1H), 7.36-7.25 (m, 6H), 6.91-
A flask was charged with 1024 (50 mg, 0.135 mmol), 2-pyridine acetic acid hydrochloride (27 mg, 0.156 mmol) in DMF (1 ml) at 0 °C was added
propylphosphonic anhydride solution (91 ul) followed by triethylamine (54 ul, 0.39 mmol). The resulting mixture was slowly warmed up to room temperature and stirred
for 1 h before it was quenched by addition of water (~5 mL). The white precipitate was collected by suction filtration, rinsed with more water and ether then dried to afford 354. 1H NMR (300 MHz, DMSO-d6) δ 12.65 (s, 1H), 11.26 (s, 1H), 8.51 (s, 1H), 8.22-8.19 (d, J= 8.97 Hz, 1H), 7.81-7.76 (m, 1H), 7.58-7.54 (d, J= 9.06 Hz, 1H), 7.42-7.26 (m, 7H), 4.02 (s, 2H), 3.78 (s, 2H), 3.01 (bs, 2H), 2.90 (bs, 2H), 1.73 (bs, 4H).
Compound 355 was prepared according to the procedure above for the preparation of compound 354. 1H NMR (300 MHz, DMSO-d6) δ 12.70 (s, 1H), 11.26 (s, 1H), 8.53- 8.49 (m, 1H), 8.22-8.19 (d, J= 9.0 Hz, 1H), 7.77-7.73 (d, J= 8.46 Hz, 1H), 7.58-7.54 (d, J= 9.48 Hz, 1H), 7.38-7.26 (m, 7H), 3.88 (s, 2H), 3.78 (s, 2H), 3.01 (bs, 2H), 2.90 (bs, 2H), 1.73 (bs, 4H).
Compounds 309 and 310 were prepared according to the procedure above for the preparation of compound 354.
To a solution of 1043 (3.2g, 19.5mmol) in carbon tetrachloride (150mL) was added N-bromosuccinimide (3.47g, 19.6mmol) and benzoyl peroxide (lOmg, catalytic). The resulting mixture was refluxed overnight before it was filtered hot. The filtrate was concentrated under reduced pressure and the residue obtained was purified by silica gel chromatography eluting with 20% ethylacetate/hexane to afford 1044 (2g, 42% yield) as an oil. 1H NMR (300MHz, Chloroform-d) δ ppm 3.66 (s, 2H) 3.74 (s, 3H) 4.5 l(s, 2H) 7.35 (m, 4H)
To a solution of 1044 (0.243g, lmmol) in acetone (lOmL) was added 2-methyl imidazole (0.4 lg, 5mmol). The resulting mixture was refluxed overnight before it was concentrated under reduced pressure and the residue obtained was diluted with water (-lOOmL). The resulting solution was partitioned between water and ethyl acetate. The organic extract was washed with more water, separated, dried over sodium sulfate, filtered and evaporated. The residue obtained was purified by silica gel chromatography eluting with MeOH/dichloromethane to afford 1045 (0.17g, 69% yield) as an oil. 1H NMR (300MHz, Chloroform-d) δ ppm 2.37 (s, 3H) 3.63 (s, 2H) 3.72 (s, 3H) 5.07 (s, 2H) 6.87 (s, 1H) 6.96-7.02 9m, 2H) 7.23-7.33 (m, 3H)
To a solution of 1045 (0.17g, 0.69mmol) in THF/MeOH/Water (lOmL, 2mL, 2mL) was added lithium hydroxide monohydrate (0.06g, 1.42mmol). The resulting mixture was stirred at room temperature overnight before it was concentrated under reduced pressure. The residue obtained was diluted with water (~20mL) and the resulting solution was acidified with acetic acid. The aqueous layer was concentrated and the product was isolated by prep HPLC. The residue obtained was dissolved in water (mL) and concentrated hydrochloric acid (mL) was added to it before it was concentrated and dried to afford 1046 (0.15gm) as a hydrochloride salt. To a suspension of carboxylic acid 1046 (41.8mg, 0.157mmol) in DMF (3mL) was added HATU (61.3mg, 0.16 lmmol) and stirred till reaction mixture is clear followed by the addition of an amine 1024 (52.5mg, 0.142mmol) and DIPEA (50ul,
0.29mmol). The resulting mixture was stirred at room temperature overnight before it was quenched by the addition of water. The resulting solution was partitioned between water and ethyl acetate. The organic extract was washed with more water, separated, dried over sodium sulfate, filtered and evaporated. The residue obtained
was triturated with ether. The solid separated was filtered, washed with ether and dried to afford 380 (40mg, 48%). 1H NMR (300MHz, Dimethylsulfoxide-d6) δ ppm 1.74 (brs, 4H) 2.91-3.02 (brs, 4H) 3.78-3.83 (m, 4H) 5.34 (s, 2H) 7.16-7.57 (m, 12H) 8.19-8.22 (d, 1H) 11.26 (s, 1H) 12.65 (brs, 1H)
-CI
O
' 1050
To an ice cold solution of 1048(5 g, 0.033mol) in methanol (50mL) was added thionyl chloride (0.2mL) and the resulting mixture was stirred at room temperature overnight before it was concentrated under reduced pressure. The residue obtained was dried at high vacuum overnight to afford 1049 (5gm) as an oil and was used as such for the next step. 1H NMR (300MHz, Chloroform-d) δ ppm 3.62 (s, 2H) 3.74 (s, 3H) 6.76- 6.87 (m, 3H) 7.18-7.21(m, 1H).
To a solution of 1049 (lg, 6mmol) in DMF (20mL) was added potassium carbonate (2.08g, 15mmol), 1050 (1.225g, 6.62mmol) and sodium iodide (lOmg). The resulting mixture was stirred at 80 °C overnight before it was diluted with water (-lOOmL). The resulting solution was partitioned between water and ethyl acetate. The organic extract was washed with more water, separated, dried over sodium sulfate, filtered and evaporated. The residue obtained was purified by silica gel chromatography eluting with MeOH/dichloromethane to afford 1051 (lg, 60% yield) as an oil. 1H NMR (300MHz, Chloroform-d) δ ppm 2.61 (s, 4H) 2.83 (t, 2H) 3.62 (s, 2H) 3.63 (s, 3H) 3.73-3.77 (m, 4H) 4.14 (t, 2H) 6.88-6.91 (m, 3H) 7.26-7.29 (m, 1H)
To a solution of 1051 (lg, 3.57mmol) in THF/MeOH/Water (30mL, 5mL, 5mL) was added lithium hydroxide monohydrate (0.3g, 7.14mmol). The resulting mixture was
stirred at room temperature overnight before it was concentrated under reduced pressure. The residue obtained was diluted with water (~50mL) and the resulting solution was acidified with IN hydrochloric acid. The aqueous layer was concentrated and the product was isolated by prep HPLC. The residue obtained was dissolved in water (mL) and concentrated hydrochloric acid (mL) was added to it before it was concentrated and dried to afford 1052 as a hydrochloride salt.
To a suspension of carboxylic acid 1052 (47.4mg, 0.157mmol) in DMF (3mL) was added HATU (61.3mg, 0.161mmol) and stirred till reaction mixture is clear followed by the addition of an amine 1024 (52.5mg, 0.142mmol) and DIPEA (50ul,
0.29mmol). The resulting mixture was stirred at room temperature overnight before it was quenched by the addition of water. The resulting solution was partitioned between water and ethyl acetate. The organic extract was washed with more water, separated, dried over sodium sulfate, filtered and evaporated. The residue obtained was purified by silica gel chromatography eluting with MeOH/dichloromethane to afford 381 (40mg, 46% yield). 1H NMR (300MHz, Dimethylsulfoxide-d6) δ ppm 1.74 (brs, 4H) 2.72 (t, 2H) 2.89-2.9 (m, 4H) 3.02 (brs, 4H) 3.336 (m, 2H) 3.76-3.78 (m,2H) 4.09 (m, 2H) 6.88-6.93 (m, 3H) 7.24-7.36 (m, 6H) 7.54-7.58 (d, 1H) 8.18- 8.21 (d, 1H) 11.26 (s, 1H) 12.65 (brs, 1H).
Pyrazole
a solution of 1044 (2.29g, O.Olmol) in DMF (lOOmL) was added potassium
carbonate (1.38g, O.Olmmol) and pyrazole (0.68g, O.Olmol). The resulting mixture was stirred at 70 °C for 5hr before it was diluted with water (~100mL). The resulting solution was partitioned between water and ethyl acetate. The organic extract was washed with more water, separated, dried over sodium sulfate, filtered and
evaporated. The residue obtained was purified by silica gel chromatography eluting with EtOAc/Hexane to afford 1053 (lg, 50% yield). 1H NMR (300MHz,
Chloroform-d) δ ppm 3.94 (s, 3H) 5.40 (s, 2H) 6.33 (s, 1H) 7.42-7.48 (m, 3H) 7.58 (s, 1H) 7.95 (s, 1H) 8.00-8.02 (m, 1H)
To an ice cold solution of 1053 (lg, 4.62mmol) in THF (20mL) was added lithium aluminum hydride (2.5mL, 2M/THF) drop wise and the resulting reaction mixture was stirred at 0 °C for 5hr before it was quenched with saturated Rochelle salt solution. The resulting solution was partitioned between water and ethyl acetate. The organic extract was washed with more water, separated, dried over sodium sulfate, filtered and evaporated to afford 1054 (0.8g, 92% yield). 1H NMR (300MHz, Chloroform-d) δ ppm 4.71 (s, 2H) 5.35 (s, 2H) 6.30 (s, 1H) 7.15-7.43 (m, 5H) 7.58 (s, 1H)
To a solution of 1054 (0.8g, 4.2mmol) in dichloromethane (20mL) was added thionyl chloride and the resulting mixture was stirred at room temperature for 5hr before it was concentrated under the reduced pressure. The residue obtained was dried at high vacuum overnight to afford 1055 (lg, 97% yield) as a HC1 salt. 1H NMR (300MHz, Dimethylsulfoxide-d6) δ ppm 4.75 (s, 2H) 5.38 (s, 2H) 6.30 (s, 1H) 7.19-7.50 (m, 5H) 7.86 (s, 1H) 11.49-11.60 (brs, 1H)
To a solution of 1055 (lg, 4.1mmol) in DMF (20mL) was added sodium cyanide (0.625g, 12.7mmol) and sodium iodide (20mg) and the resulting reaction mixture was stirred at 70 °C for 2hr before it was diluted with water. The resulting solution was partitioned between water and ethyl acetate. The organic extract was washed with more water, separated, dried over sodium sulfate, filtered and evaporated. The residue obtained was purified by silica gel chromatography eluting with EtOAc/Hexane to afford 1056 (0.664g, 83% yield). 1H NMR (300MHz, Chloroform-d) δ ppm 3.76 (s, 2H) 5.38 (s, 2H) 6.35 (s, 1H) 7.19-7.46 (m, 5H) 7.61 (s, 1H)
To a solution of 1056 (0.664g, 3.3mmol) in dioxane (5mL) was added concentrated hydrochloric acid (5mL) and the resulting reaction mixture was stirred at 90 °C overnight before it was concentrated under the reduced pressure. The residue obtained was purified through prep HPLC and was converted to HC1 salt to afford 1057 (0.5g, 40% yield). 1H NMR (300MHz, Dimethylsulfoxide-d6) δ ppm 3.55 (s, 2H) 5.33 (s, 2H) 6.29 (s, 1H) 7.14-7.20 (m, 4H) 7.48 (s, 1H) 7.84 (s, 1H) 11.97-11.99 (brs, 1H)
To a suspension of carboxylic acid 1057 (19.8mg, 0.0785mmol) in DMF (2mL) was added HATU (30.6mg, 0.08mmol) and stirred till reaction mixture is clear followed by the addition of an amine 1024 (26.25mg, 0.07mmol) and DIPEA (25ul,
0.15mmol). The resulting mixture was stirred at room temperature overnight before it was quenched by the addition of water. The solid separated was filtered, washed with water and dried to afford 395 (18mg, 45%yield). 1H NMR (300MHz,
Dimethylsulfoxide-d6) δ ppm 1.74 (brs, 4H) 2.89-3.04 (m, 4H) 3.78 (s, 4H) 5.33 (s, 2H) 6.27-6.28 (s, 1H) 7.09-7.58 (m, 11H) 7.82 (s, 1H) 8.19-8.21 (d, 1H) 11.26 (s, 1H) 12.65 (brs, 1H)
To a solution of 1044 (lg, 4.1mmol) in THF(5mL) was added 2M/THF methyl amine solution (2mL) and the resulting reaction mixture was stirred at room temperature overnight before it was concentrated under the reduced pressure. The residue obtained
was partitioned between water and ethyl acetate. The organic extract was washed with more water, separated, dried over sodium sulfate, filtered and evaporated. The residue obtained was purified by silica gel chromatography eluting with
MeOH/dichloromethane to afford 1058 (0.26g, 33% yield). 1H NMR (300MHz, Chloroform-d) δ ppm 2.49 (s, 3H) 3.66 (s, 2H) 3.73 (s, 3H) 3.79 (s, 2H) 7.2-7.33 (m, 4H).
To a solution of 1058 (0.26g, 1.35mmol) in dichloromethane (5mL) was added boc anhydride (0.293g, 1.35mmol) and the resulting reaction mixture was stirred at room temperature for 4hr before it was purified by silica gel chromatography eluting with EtOAc/Hexane to afford 1059 (0.3g, 77% yield). 1H NMR (300MHz, Chloroform-d) δ ppm 1.5 (s, 9H) 2.84 (s, 3H) 3.66 (s, 2H) 3.73 (s, 3H) 4.44 (s, 2H) 7.17-7.32 (m, 4H).
To an ice cold solution of 1059 (0.3g, 1.02mmol) in dioxane (3mL) and water (2mL) was added lithium hydroxide monohydrate (0.086g, 2.04mmol) and the resulting reaction mixture was stirred at 0 °C for 3hr before it was acidified with IN HC1. The resulting solution was partitioned between water and ethyl acetate. The organic extract was washed with more water, separated, dried over sodium sulfate, filtered and evaporated. The residue obtained was dried at high vacuum overnight to afford 1060 (0.2g, 70%yield). 1H NMR (300MHz, Chloroform-d) δ ppm 1.5 (s, 9H) 2.84 (s, 3H) 3.66 (s, 2H) 4.43 (s, 2H) 7.17-7.32 (m, 4H)
To a suspension of carboxylic acid 1060 (51.1mg, 0.183mmol) in DMF (3mL) was added HATU (69.7mg, 0.183mmol) and stirred till reaction mixture is clear followed by the addition of an amine 1024 (61.3mg, 0.166mmol) and DIPEA (58ul,
0.33mmol). The resulting mixture was stirred at room temperature overnight before it was quenched by the addition of water. The resulting solution was partitioned between water and ethyl acetate. The organic extract was washed with more water, separated, dried over sodium sulfate, filtered and evaporated. The residue obtained was purified by silica gel chromatography eluting with MeOH/dichloromethane to afford 445 (0.06g, 57% yield). 1H NMR (300MHz, Dimethylsulfoxide-d6) δ ppm 1.37-1.38 (s, 9H) 1.74 (brs, 4H) 2.76 (s,3H) 2.89 (brs, 2H) 3.02 (brs, 2H)3.78-3.80
(m, 4H) 4.36 (s, 2H) 7.11-7.36 (m, 9H) 7.54-7.57 (d, 1H) 8.18-8.21 (d, 1H) 11.26 (s, 1H) 12.65 (brs, 1H).
Prep of 445 via 396 deprotection to 408 and re-acylation:
To an ice cold solution of 408 (26mg, 0.04mmol) in DMF (lmL) was added triethylamine (12.3uL, 0.088mmol) and acetyl chloride (3.16uL, 0.044mmol). The resulting mixture was stirred at room temperature for 2hr before it was diluted with water. The solid separated was filtered, washed with water and dried at high vacuum overnight to afford 445 (1 Omg, 48% yield). 1H NMR (300MHz, Dimethylsulfoxide- d6) δ ppm 1.74 (brs, 4H) 2.05 (m, 3H) 2.91-3.02 (m,7H) 3.78-3.82 (m, 4H) 4.49-4.56 (m, 2H) 7.18-7.36 (m, 9H) 7.55-7.58 (d, 1H) 8.18-8.21 (d, 1H) 8.75-8.7 (brs, 2H) 11.26 (s, 1H) 12.65 (brs, 1H).
Compound 401 was prepared according to the procedure above for the preparation of compound 339. 1H NMR (300MHz, Dimethylsulfoxide-d6) δ ppm 1.40 (s, 9H) 1.75 (brs, 4H) 2.87 (brs, 2H) 2.89 (brs, 2H) 3.78 (s, 4H) 4.09-4.11 (brs, 2H) 7.18-7.36 (m, -7.58 (d, 1H) 8.18-8.21 (d, 1H) 11.26 (s, 1H) 12.65 (brs, 1H)
Compound 413 was prepared according to the procedure above for the preparation of compound 315. 1H NMR (300 MHz, DMSO-d6) δ 12.68 (bs, 1H), 11.26 (s, 1H), 8.20
(d, J= 9.46 Hz, 1H), 7.58-7.26 (m, 10H), 3.90 (s, 2H), 3.78 (s, 2H), 3.02 (bs, 2H),
Compound 415 was prepared according to the procedure above for the preparation of compound 315.: 1H NMR (300 MHz, DMSO-d6) δ 12.48 (s, 1H), 11.26 (s, 1H), 8.20 (d, J= 8.95 Hz, 1H), 7.75 (s, 1H), 7.58-7.26 (m, 9H), 6.52 (m, 1H), 5.35 (m, 1H), 3.78 (s, 2H), 3.02 (m, 2H), 2.90 (m, 2H), 1.74 (bs, 4H).
To a solution of 1063 (6.3 lg, 24.9mmol) in ethanol was added lithium hydroxide monohydrate (1.048g, 24.9mmol) and the resulting reaction mixture was stirred at room temperature for 3hr before it was concentrated under the reduced pressure. The residue obtained was diluted with water and was acidified with 6N HCl. The solution was extracted with ethyl acetate. The organic extract was washed with more water, separated, dried over sodium sulfate, filtered and evaporated. The residue obtained was purified by silica gel chromatography eluting with EtOAc/hexane to afford 1064 (3g, 53% yield).
To a suspension of carboxylic acid 1064 (O. lg, 0.44mmol) in DMF (2mL) was added HATU (0.17g, 0.44mmol) and stirred till reaction mixture is clear followed by the addition of an amine 1024 (0.15g, 0.4mmol) and DIPEA (0.14mL, 0.8mmol). The resulting mixture was stirred at room temperature overnight before it was quenched by the addition of water. The solid separated was filtered, washed with water and dried to afford 456 (0.2, 86%yield). 1H NMR (300MHz, Dimethylsulfoxide-d6) δ ppm 1.18 (t, 3H) 1.74 (brs, 4H) 2.88-2.90 (m,2H) 3.01-3.04 (m, 2H) 3.66 (s, 2H) 3.78 (s, 4H) 4.05-4.12 (q, 2H) 7.19-7.36 (m, 9H) 7.55-7.58 (m, 1H) 8.18-8.21 (d, 1H) 11.26 (s, 1H) 12.65 (brs, 1H).
To a solution of 456 (0.205g, 0.358mmol) in Dioxane/Water (20mL/ 6mL) was added lithium hydroxide monohydrate (0.06g, 1.42mmol). The resulting mixture was stirred at room temperature for 3hr before it was acidified with acetic acid. The solution was concentrated under reduced pressure and the residue obtained was diluted with water. The solid separated was filtered, washed with water and dried at high vacuum overnight. The residue obtained was purified by silica gel chromatography eluting with MeOH/dichloromethane to afford 465 (0.15g, 77% yield). 1H NMR (300MHz, Dimethylsulfoxide-d6) δ ppm 1.74 (brs, 4H) 2.90 (brs, 2H) 3.01 (brs, 2H) 3.5 (s, 2H) 3.78 (s, 4H) 7.19-7.36 (m, 9H) 7.55-7.58 (m, 1H) 8.18-8.21 (d, 1H) 1 1.26 (s, 1H) 12.32 (brs, 1H) 12.65 (s, 1H). To a suspension of carboxylic acid 465 (25mg, 0.046mmol) in DMF (lmL) was added HATU (19.2mg, 0.05mmol) and stirred till reaction mixture is clear followed by the addition of an Ν,Ν-dimethylamine (2M/THF, 30uL, 0.05mmol) and DIPEA (16uL, 0.092mmol). The resulting mixture was stirred at room temperature for 3hr before it was quenched by the addition of water. The solid separated was filtered, washed with water and dried to afford 472 (19mg, 73%yield). 1H NMR (300MHz, Dimethylsulfoxide-d6) δ ppm 1.74 (brs, 4H) 2.83-2.90 (brs, 6H) 3.01 (brs, 4H) 3.68 (s, 2H) 3.78 (s, 4H) 7.14-7.36 (m, 9H) 7.55-7.58 (d, 1H) 8.18-8.21 (d, 1H) 11.26 (s, 1H) 12.65 (brs, 1H).
To a solution of 1049 (lg, 6mmol) in DMF (20mL) was added potassium carbonate
(1.662g, 12mmol) and (2.16g, 9mmol). The resulting mixture was stirred at 70 C overnight before it was diluted with water (-lOOmL). The resulting solution was partitioned between water and ethyl acetate. The organic extract was washed with more water, separated, dried over sodium sulfate, filtered and evaporated. The residue obtained was purified by silica gel chromatography eluting with EtOAc/Hexane to afford 1065 (1.78g, 91% yield) as an oil. 1H NMR (300MHz, Chloroform-d) δ ppm 0.13 (s, 6H) 0.95 (s, 9H) 3.63 (s, 2H) 3.73 (s, 2H) 3.99-4.06 (m, 4H) 6.87 (m, 3H) 7.3 (m, 1H).
To a solution of 1065 (1.78g, 5.5mmol) in THF/MeOH/Water (30mL, 3mL, 3mL) was added lithium hydroxide monohydrate (0.46g, 10.9mmol). The resulting mixture was stirred at room temperature overnight before it was concentrated under reduced pressure. The residue obtained was diluted with water (~20mL) and the resulting solution was acidified with 6N hydrochloric acid. The solution was partitioned between water and ethyl acetate. The organic extract was washed with more water, separated, dried over sodium sulfate, filtered and evaporated. The residue obtained was purified by silica gel chromatography eluting with EtOAc/Hexane to afford 1065 and 1066. 1H NMR (300MHz, Dimethylsulfoxide-d6) δ ppm 3.54 (s, 2H) 3.72 (brs, 2H) 3.96-3.98 (brs, 2H) 4.85 (brs, 1H) 6.82-6.85 (m, 3H) 7.0-7.22 (m, 1H) 12.3 (brs, 1H).
To a suspension of carboxylic acid 1065 (27mg, 0.137mmol) in DMF (2mL) was added HATU (52.2mg, 0.137mmol) and stirred till reaction mixture is clear followed
by the addition of an amine 1024 (46mg, 0.125mmol) and DIPEA (44ul, 0.25mmol). The resulting mixture was stirred at room temperature overnight before it was quenched by the addition of water. The solid separated was filtered, washed with water and dried. The solid obtained was purified by prep HPLC to afford 427 (16mg, 23%yield). 1H NMR (300MHz, Dimethylsulfoxide-d6) δ ppm 1.75 (brs, 4H) 2.90 (brs, 2H) 3.02 (brs, 2H) 3.71-3.78 (m, 6H) 3.98-3.99 (brs, 2H) 4.84-4.87 (brs, 1H) 6.83-6.92 (m,3H) 7.21-7.36 (m, 6H) 7.54-7.58 (d, 1H) 8.2-8.23 (d, 1H) 11.26 (s, 1H) 12.65 (brs, 1H).
To a solution of 1049 (lg, 6mmol) in acetone (50mL) was added cesium carbonate (2.545g, 7.83mmol), 2- bromoethyl methyl ether(0.92g, 6.62mmol) and sodium iodide(lOmg). The resulting mixture was stirred at 50 °C overnight before it was filtered. The filtrate was evaporated and the residue obtained was purified by silica gel chromatography eluting with EtOAc/Hexane to afford 1075 (0.97g, 72% yield) as oil. 1H NMR (300MHz, Chloroform-d) δ ppm 3.48 (s, 3H) 3.63 (s, 2H) 3.72(brs, 2H) 4.14-4.15 (t, 2H) 6.86-6.9 (m, 3H) 7.26-7.29 (m, 1H).
The remainder of the preparation for compound 428 followed the procedure above for compound 427. 428: 1H NMR (300MHz, Dimethylsulfoxide-d6) δ ppm 1.75 (brs, 4H) 2.90 (brs, 2H) 3.02 (brs, 2H) 3.32 (s, 3H) 3.66 (brs,2H) 3.78 (brs, 4H) 4.08 (brs, 2H) 6.88-6.92 (m,3H) 7.25-7.27 (m, 6H) 7.54-7.58 (d, 1H) 8.2-8.23 (d, 1H) 11.26 (s, 1H) 12.65 (brs, 1H).
To an ice cold solution of 1068 (6g, 30.9mmoL) in ethanol (50mL) was added thionyl chloride (2mL) and the resulting reaction mixture was stirred at room temperature overnight before it was concentrated under the reduced pressure. The residue obtained was partitioned between water and ethyl acetate. The organic extract was washed with more water, separated, dried over sodium sulfate, filtered and evaporated to afford 1063 (6gm).
To a stirred solution of 1063 (3.35g, 13.4mmol) in THF (50mL) was added CDI (2.44g, 15mmol)and the resulting mixture was stirred for 2hr followed by the addition of water (13mL). The reaction mixture was cooled to 0 °C and sodium borohydride (2.87g, 76mmol) was added portionwise. The stirring was continued at room temperature for 3hr before it was diluted with ethyl acetate and acidifed with 6N HCl. The organic layer was separated, dried over sodium sulfate, filtered and evaporated. The residue obtained was purified by silica gel chromatography eluting with
EtOAc/Hexane to afford 1069 (0.563g, 20% yield) as an oil. 1H NMR (300MHz,
Chloroform-d) δ ppm 1.27-1.31 (q, 3H) 2.87-2.92 (d, 2H) 3.63 (s, 2H) 3.87-3.92 (t, 2H) 4.18-4.2 (q, 2H) 7.19-7.31 (m, 4H).
To an ice cold solution of 1069 (0.563g, 2.7mmol) in dichloromethane (40mL) and triethylamine (0.47mL, 3.3mmol) was added methane sulfonylchloride (0.23mL, 3.3mmol) and the resulting mixture was stirred at 0 °C for 2hr and at room
temperature for lhr before it was diluted with saturated aqueous sodium bicarbonate solution. The solution was extracted with ethyl acetate. The organic extract was washed with more water, separated, dried over sodium sulfate, filtered and evaporated to afford 1070 (0.78g, 100%yield). 1H NMR (300MHz, Chloroform-d) δ ppm 1.27- 1.31 (q, 3H) 2.87 (s, 3H) 3.08 (t, 2H) 3.63 (s, 2H) 4.18-4.2 (t, 2H) 4.45 (q, 2H) 7.19- 7.31 (m, 4H).
To a solution of 1070 (0.787g, 2.7mmol) in DMF (6mL) was added sodium azide (0.358g, 5.5mmol) and the resulting reaction mixture was stirred at 60 °C for 3hr before it was partitioned between water and ethyl acetate. The organic extract was washed with more water, separated, dried over sodium sulfate, filtered and evaporated. The residue obtained was purified by silica gel chromatography eluting with EtOAc/Hexane to afford 1071 (0.5g, 78% yield) as an oil. 1H NMR (300MHz, Chloroform-d) δ ppm 1.27-1.31 (q, 3H) 2.92 (t, 2H) 3.54 (t, 2H) 3.63 (s, 2H) 4.18-4.2 (q, 2H) 7.19-7.29 (m, 4H). To a solution of 1071 (0.5g, 2.1mmol) in THF (25mL) was added triphenylphosphine (0.787g, 3mmol) and the reaction mixture was stirred at room temperature under argon for overnight before it was diluted with lmL of water. The reaction was continued at 50 °C for lhr before it was concentrated under the reduced pressure. The residue was partitioned between saturated sodium bicarbonate solution and dichloromethane. The organic layer was separated, dried over sodium sulfate, filtered and evaporated. The residue obtained was purified by silica gel chromatography eluting with MeOH/dichloromethane to afford 1072 (0.43g, 100% yield) as an oil. 1H NMR (300MHz, Chloroform-d) δ ppm 1.27-1.31 (q, 3H) 2.75-2.79 (t, 2H) 2.98- 3.02 (t, 2H) 3.63 (s, 2H) 4.18-4.2 (q, 2H) 7.13-7.29 (m, 4H). To a solution of 1072 (0.427g, 2mmol) in dichloromethane (30mL) was added di-tert- butyl dicarbonate ( 0.447g, 2mmol) and the reaction mixture was stirred at room
temperature for 5hr before it was purified by silica gel chromatography eluting with EtOAc/Hexane to afford 1073 (0.577g, 91% yield) as an oil. 1H NMR (300MHz, Chloroform-d) δ ppm 1.27-1.31 (q, 3H) 1.59 (s, 9H) 2.82 (t, 2H) 3.4 (m, 2H) 3.63 (s, 2H) 4.18 (q, 2H) 7.13-7.29 (m, 4H). To a solution of 1073 (0.577g, 1.8mmol) in Dioxane/Water (lOmL/ 3mL) was added lithium hydroxide monohydrate (0.158g, 3.6mmol). The resulting mixture was stirred at room temperature overnight before it was concentrated under reduced pressure. The residue obtained was diluted with water (~20mL) and the resulting solution was acidified with IN hydrochloric acid. The solution was partitioned between water and ethyl acetate. The organic extract was washed with more water, separated, dried over sodium sulfate, filtered and evaporated to afford 1074 (0.35g, 67%yield). 1H NMR (300MHz, Chloroform-d) δ ppm 2.82 (m, 2H) 3.4 (m, 2H) 3.63 (s, 2H) 4.6 (brs, 1H) 7.13-7.29 (m, 4H).
To a suspension of carboxylic acid 1074 (43.8mg, 0.157mmol) in DMF (2mL) was added HATU (61.3mg, 0.16 lmmol) and stirred till reaction mixture is clear followed by the addition of an amine 1024 (52.5mg, 0.142mmol) and DIPEA (50ul,
0.287mmol). The resulting mixture was stirred at room temperature overnight before it was quenched by the addition of water. The solid separated was filtered, washed with water and dried to afford 429 (60mg, 67%yield). 1H NMR (300MHz,
Dimethylsulfoxide-d6) δ ppm 1.37-1.38 (s, 9H) 1.74 (brs, 4H) 2.69-2.71 (m,2H) 2.87- 2.88 (m, 2H) 2.9-3.15 (m, 4H) 3.78 (s, 4H) 7.09 (brs, 1H) 7.12-7.36 (m, 9H) 7.54- 7.57 (d, 1H) 8.18-8.21 (d, 1H) 11.26 (s, 1H) 12.65 (brs, 1H).
To a suspension of 429 (50mg, 79.5mmol) in dichloromethane (5mL) was added TFA (lmL) and the reaction mixture was stirred at room temperature for overnight before it was concentrated under the reduced pressure. The residue obtained was triturated with ether. The solid separated was filtered, washed with ether and dried at high vacuum overnight to afford 441 (45mg, 88%yield) as a TFA salt. 1H NMR
(300MHz, Dimethylsulfoxide-d6) δ ppm 1.74 (brs, 4H) 2.86-3.02 (m, 8H) 3.78-3.80 (s, 4H) 7.12-7.36 (m, 8H) 7.58 (d, 1H) 7.78 (brs, 3H) 8.18-8.21 (d, 1H) 1 1.26 (s, 1H) 12.65 (brs, 1H).
To an ice cold solution of 441 (23mg, 0.035mmol) in DMF (lmL) was added triethylamine (l luL, 0.079mmol) and acetyl chloride (2.8uL, 0.038mmol). The resulting mixture was stirred at room temperature for 2hr before it was diluted with water. The solid separated was filtered, washed with water and dried at high vacuum overnight to afford 454 (lOmg, 50% yield). 1H NMR (300MHz, Dimethylsulfoxide- d6) δ ppm 1.75-1.79 (m, 7H) 2.67-2.70 (m, 2H) 2.9 (brs, 2H) 3.00-3.02 (m, 2H) 3.21- 3.26 (m, 2H) 3.78 (s, 4H) 7.12-7.36 (m, 9H) 7.58 (d, 1H) 7.9 (brs, 1H) 8.18-8.21 (d,
Compound 409 was prepared via TFA deprotection of compound 399 according to the procedure above for the preparation of compound 441. 1H NMR (300MHz,
Dimethylsulfoxide-d6) δ ppm 1.75 (brs, 4H) 2.90 (brs, 2H) 3.02 (brs, 2H) 3.78 (brs, 4H) 6.89-6.98 (m,4H) 7.25-7.36 (m, 7H) 7.51-7.58 (d, 1H) 8.2-8.23 (d, 1H) 9.34 (s,
1H).
Compound 457 was prepared by acylation of 409 according to the amide coupling procedure above for the preparation of compound 39. 1H NMR (300MHz,
Dimethylsulfoxide-d6) δ ppm 1.74 (brs, 4H) 2.32 (s, 6H) 2.89 (m, 2H) 3.02 (m, 2H) 3.13 (s, 2H) 3.78 (s, 4H) 7.01-7.04 (m, 1H) 7.25-7.38 (m, 6H) 7.54-7.58 (m, 3H) 8.18-8.21 (d, 1H) 9.77 (s, 1H) 11.26 (s, 1H) 12.65 (brs, 1H)
348
To a suspension of 295 (30 mg, 0.0617 mmol) in MeOH (2 ml) at 0 C was added 2N NaOH (2 ml) solution. The resulting mixture was stirred at room temperature overnight. The solvent was evaporated under vacuo and the mixture was acidified with IN HCl to pH 6. The white precipitate was collected by suction filtration, rinsed with more water and dried to afford 348. 1H NMR (300 MHz, DMSO-d6) δ 7.32-7.24 (m, 5H), 7.15-7.12 (d, J= 9.57 Hz, 1H), 6.72-6.69 (d, J= 9.15 Hz, 1H), 6.09 (s, 2H),
-2.96 (bs, 2H), 2.76-2.70 (bs, 2H), 1.70 (bs, 4H).
366: 1H NMR (300 MHz, DMSO-d6) δ 12.65 (s, 1H), 11.26 (s, 1H), 8.22-8.19 (d, J = 8.82 Hz, 1H), 7.58-7.54 (d, J= 9.32 Hz, 1H), 7.33-7.25 (m, 6H), 6.95-6.82 (m, 3H), 3.81 (s. 3H), 3.75 (s, 4H), 3.01 (bs, 2H), 2.90 (bs, 2H), 1.73 (bs, 4H).
367: A flask was charged with 348 (100 mg, 0.27 mmol), Boc-3-aminomethyl- phenylacetic acid (86 mg, 0.325 mmol) in DMF (2 ml) at 0 °C was added HOBT (88 mg, 0.65 mmol) followed by EDCI (156 mg, 0.812 mmol). The resulting mixture was stirred at 0 °C for 5 minutes then warmed up to room temperature overnight before it was quenched by addition of water (-10 mL) at 0 °C. The white precipitate was collected by suction filtration, rinsed with more water. The crude material was purified by silica gel chromatography eluting with 0-6% MeOH in CH
2CI
2 to afford 367.
Compound 368 was prepared via the deprotection of compound 367 according to the procedure above for compound 341. 1H NMR (300 MHz, DMSO-d6) δ 12.65 (s, 1H),
11.26 (s, 1H), 8.22-8.16 (m, 3H), 7.58-7.54 (d, J= 9.27 Hz, 1H), 7.40-7.28 (m, 9H),
4.04 (s, 2H), 3.81 (s. 4H), 3.01 (bs, 2H), 2.90 (bs, 2H), 1.73 (bs, 4H).
Compound 383 was prepared from compound 348 according to the procedure above for the preparation of compound 354. 1H NMR (300 MHz, DMSO-d6) δ 12.65 (s,
1H), 11.26 (s, 1H), 8.51 (s, 1H), 8.22-8.19 (d, J= 9.09 Hz, 1H), 7.81-7.76 (m, 1H),
7.58-7.54 (d, J= 9.12 Hz, 1H), 7.42-7.26 (m, 7H), 4.0 (s, 2H), 3.81 (s, 2H), 3.01 (bs,
2H), 2.90 (bs, 2H), 1.73 (bs, 4H).
To a solution of 348 (56.5 mg, 0.153 mmol) in DMF (1 ml) at 0 C was added triethylamine (43 ul, 0.306 mmol) drop wise followed by benzyl isocyanate (23 ul, 0.184 mmol) drop wise. The resulting mixture was slowly warmed up to room temperature and stirred for 6 h before it was quenched by addition of water (~5 mL) at 0 °C. The white precipitate was collected by suction filtration, rinsed with more water and ether and dichloromethane then dried to afford 405. 1H NMR (300 MHz, DMSO- d6) δ 12.65 (s, 1H), 9.57 (s, 1H), 8.25 (bs, 1H), 7.74-7.71 (d, J= 8.61 Hz, 1H), 7.50-
7.47 (d, J= 9.42 Hz, 1H), 7.34-7.27 (m, 10H), 4.42-4.40 (d, J = 5.46 Hz, 2H), 3.80 (s,
To a suspension of 339 (1 g, 1.62 mmol) in MeOH (10 ml) at 0 C was added 2N NaOH (10 ml) solution. The resulting mixture was stirred at room temperature overnight. The solvent was evaporated under vacuo and the mixture was acidified with 6N HCl to pH 6 at 0 °C. The mixture was triturated with EtOAc and the white precipitate was collected by suction filtration, rinsed with more EtOAc and dried to afford 412. 1H NMR (300 MHz, DMSO-d6) δ 12.66 (s, 1H), 7.29-7.22 (m, 2H), 7.19-7.13 (m, 4H), 6.72 (d, J= 8.86 Hz, 1H), 6.12 (bs, 2H), 4.12 (d, J= 6.09 Hz, 2H), 3.79 (s, 2H), 3.01 (m, 2H), 2.71 (m, 2H), 1.70 (bs, 4H), 1.39 (s, 9H).
To a solution of 412 (60 mg, 0.121 mmol) in DMF (1 ml) at 0 °C was added triethylamine (34 ul, 0.242 mmol) drop wise followed by ethyl isocyanate (11 ul, 0.145 mmol) drop wise. The resulting mixture was slowly warmed up to room temperature and stirred for 6 h before it was quenched by addition of water (~5 mL) at 0 °C. The white precipitate was collected by suction filtration. The crude material was purified by silica gel chromatography eluting with 0-6% MeOH in CH
2CI
2 to afford 420. 1H NMR (300 MHz, DMSO-d
6) δ 12.65 (s, 1H), 11.27 (s, 1H), 9.42 (s, 1H), 8.22-8.19 (d, J= 8.61 Hz, 1H), 7.77-7.13 (m, 5H), 6.56-6.53 (bs, 1H), 4.12-4.11 (d, 2H), 3.78 (s, 2H), 3.23-3.16 (m, 2H), 3.01 (bs, 2H), 2.90 (bs, 2H), 1.73 (bs, 4H), 1.38 (s, 9H), 1.10-1.07 (t, 3H).
422: 1H NMR (300 MHz, DMSO-d6) δ 12.65 (s, 1H), 10.74 (s, 1H), 8.18-8.15 (d, J = 9.51 Hz, 1H), 7.61-7.12 (m, 9H), 6.62 (s, 1H), 5.33 (s, 1H), 4.13-4.11 (d, J= 5.58 Hz,
), 2.90 (bs, 2H), 1.73 (bs, 4H), 1.38 (s, 9H).
To a solution of 412 (40 mg, 0.0804 mmol) in DMF (1 ml) at 0 UC was added triethylamine (17 ul, 0.121 mmol) drop wise followed by acetic anhydride (8 ul, 0.0844 mmol) drop wise. The resulting mixture was slowly warmed up to room temperature and stirred overnight before it was quenched by addition of water (~5 mL) at 0 °C. The mixture was partitioned between water and EtO Ac. The organic extract was washed with water, dried over sodium sulfate, filtered and evaporated. The crude material was purified by silica gel chromatography eluting with 0-6% MeOH in CH2C12 to afford 424. 1H NMR (300 MHz, DMSO-d6) δ 12.65 (s, 1H), 11.01 (s, 1H), 8.23-8.20 (d, J= 8.61 Hz, 1H), 7.57-7.55 (d, J= 8.16 Hz, 1H), 7.38- 7.12 (m, 4H), 4.13-4.11 (d, J = 5.76 Hz, 2H), 3.78 (s, 2H), 3.01 (bs, 2H), 2.90 (bs,
4H), 1.39 (s, 9H).
425
To a suspension of 424 (10 mg, 0.018 mmol) in dichloromethane (1 ml) was added
TFA (1 ml) at 0 °C. The resulting mixture was stirred at room temperature for 1 h before it was evaporated under vacuo to dryness. Ether was added and the white precipitate was collected by suction filtration, rinsed with more ether and dried to afford 425. 1H NMR (300 MHz, DMSO-d6) δ 12.70 (s, 1H), 11.0 (s, 1H), 8.22-8.19
(d, J= 8.82 Hz, 1H), 8.16-8.08 (bs, 2H), 7.58-7.54 (d, J= 9.42 Hz, 1H), 7.39-7.30 (m, 4H), 4.06-4.03 (m, 2H), 3.84 (s, 2H), 3.01 (bs, 2H), 2.90 (bs, 2H), 2.14 (s, 3H), 1.75 (bs, 4H).
To a solution of 1076(1.8g, lOmmmol) in ethanol/water (40mL/20mL) was added sodium cyanide (0.98g, 20mmol). The resulting mixture was stirred at 90 °C for 4hr before it was cooled to 0 °C. Solid separated was filtered, washed with water and dried at high vacuum overnight to afford 1077(1.5g, 85% yield).
To an ice cold solution of 1077(1 g, 5.68mmmol) in ethanol (50mL) was added sodium borohydride (0.86g, 22.72mmol) followed by the addition of bismuth chloride (2g, 6.248mmol) portionwise. The resulting mixture was stirred at room temperature for 3hr before it was filtered through the celite pad. Filtrate was concentrated and the residue obtained was partitioned between aq sodium bicarbonate solution and ethyl acetate. The organic extract was separated, dried over sodium sulfate, filtered and evaporated to afford 1078 (0.82g, 100% yield). 1H NMR (300MHz, Chloroform-d) δ ppm 2.17(s, 3H) 3.69-3.71 (brs, 4H) 6.71-6.74 (d, 1H) 6.80-6.83(d, 1H) 7.04-7.09 (m, 1H).
To a solution of 1078 (0.3g, 2mmmol) in toluene (lOmL) was added potassium acetate (0.2g, 2.04mmol) and acetic anhydride (0.55mL, 5.83mmol). The resulting mixture was stirred at 80 °C for lhr followed by the addition of isoamyl nitrite (0.4mL, 3mmol). Stirring was continued at 80 °C overnight before it was cooled to room temperature. The solution was partitioned between water and ethyl acetate. The organic extract was washed with more water, separated, dried over sodium sulfate, filtered and evaporated. The residue obtained was purified by silica gel
chromatography eluting with EtOAc/Hexane to afford 1079 (0.22g, 54% yield). 1H NMR (300MHz, Chloroform-d) δ ppm 2.85(s, 3H) 4.09 (s, 2H) 7.39-7.41 (d, 1H) 7.58-7.63(m, 1H) 8.28 (s, 1H) 8.48-8.5 l(d, 1H)
To a solution of 1079 (0.44g, 2.21mmmol) in ethanol (5mL) was added 20% aqueous sodium hydroxide (5mL). The resulting mixture was stirred at 90 0 overnight before it was concentrated. The residue obtained was diluted with water, acidified with acetic acid and extracted with ethyl acetate. The organic extract was separated, dried over sodium sulfate, filtered and evaporated to afford 1080 (0.1 g, 51 % yield). lU NMR (300MHz, Dimethylsulfoxide-d6) δ ppm 3.89 (s, 2H) 6.98-7.0 (d, 1H) 7.27-7.32(m, 1H) 7.43-7.46 (d, 1H) 8.10(s, 1H) 12.3-13.2(broad doublet, 2H)
To a suspension of carboxylic acid 1080 (60mg, 0.34mmol) in DMF (2mL) was added HATU (130mg, 0.34mmol) and stirred till reaction mixture is clear followed by the addition of an amine 1024 (114mg, 0.31mmol) and DIPEA (108uL, 0.62mmol). The resulting mixture was stirred at room temperature for 3hr before it was quenched by the addition of water. The solid separated was filtered, washed with water and dried. The residue obtained was purified by silica gel chromatography eluting with MeOH/dichloromethane to afford 512 (14mg, 9%yield). 1H NMR (300MHz,
Dimethylsulfoxide-d6) δ ppm 1.74 (brs, 4H) 2.89 (brs, 2H) 2.91 (brs, 2H) 3.78 (s, 2H) 4.13 (s, 2H) 7.05-7.08 (m, 1H) 7.27-7.57 (m, 8H) 8.19 (d, 2H) 11.26 (s, 1H) 12.76- 12.80 (brs, 1H) 13.11 (s, 1H).
Compound 389 was prepared according to the procedure above for the preparation of compound 334. 1H NMR (300 MHz, DMSO-d6) δ 12.95 (s, 1H), 11.26 (s, 1H), 8.22- 8.19 (d, J= 8.91 Hz, 1H), 7.61-7.26 (m, 10H), 6.17 (s, 1H), 3.78 (s, 2H), 3.54 (bs,
2H), 2.67-2.62 (m, 4H), 2.38 (bs, 4H), 1.73 (bs, 4H).
Compound 404 was prepared according to the procedure above for the preparation of compound 334. 1H NMR (300 MHz, DMSO-d6) δ 12.95 (s, 1H), 11.26 (s, 1H), 8.22- 8.19 (d, J= 9.60 Hz, 1H), 7.58-7.54 (d, J= 9.03 Hz, 1Η),7.39-7.26 (m, 6H), 7.12 (s, 2H), 7.01-6.98 (m, 1H), 6.10 (s, 1H), 3.78 (s, 5H), 3.54 (bs, 4H), 3.01 (bs, 2H), 2.90
1.74 (bs, 4H).
To a flask was added K
2CO
3 (0.28 g, 2.06 mmol), compound 295 (0.5 g, 1.03 mmol) followed by 25 mL of DMF. The mixture was stirred for 15 minutes and chloromethyl butyrate (0.17 g, 1.23 mmol) was added and the reaction placed under an atmosphere of argon. The mixture was heated to 80°C for 1.5 hours, allowed to cool to room temperature and poured into 200 ml water. The mixture was transferred to a
separatory funnel, extracted with EtOAc (3x100 mL), the organic layers separated and washed with water (3x50 mL), brine (2x50 ml) and dried over Na
2S0
4. The Na
2S0
4 was removed by filtration and the volatiles removed under reduced pressure. The crude material was purified by reverse-phase chromatography giving 0.15 g of compound 402.
318 439
To a solution of 318 (100 mg, 0.19 mmol) in CH2C12 (5 mL) at 0 °C was added pyridine (300 μί) and followed by addition of a solution of butyryl chloride (43 mL, 0.41 mmol) in CH2C12 (5 mL) dropwise. The resulting mixture was stirred at 0 °C for 1 h before it was partitioned between EtOAc and H20. The organic layer was separated, dried (MgS04) and concentrated. The residue was purified by flash column chromatography over silica gel eluting with 1-10% MeOH in CH2C12 to provide the desired product 439 (117 mg). 1H NMR (300 MHz, CDC13) δ 13.01 (bs, 1H), 10.12 (s, 1H), 8.49 (d, J= 9.64 Hz, 1H), 7.77 (s, 1H), 7.57 (d, J= 7.11 Hz, 1H), 7.40-7.30 (m, 8H), 6.57 (s, 1H), 3.97 (s, 2H), 3.09 (bs, 2H), 3.00 (bs, 2H), 2.48 (m,
-1.62 (m, 2H), 0.98 (t, J= 7.07 Hz, 3H).
To a solution of sodium thiomethoxide (0.266g, 3.8mmol) in DMF(lOmL) was added a solution of 1016 (0.657g, 2.7mmol) in DMF and the resulting mixture was stirred at room temperature for overnight. The solution was partitioned between water and ethyl acetate. The organic extract was washed with more water, separated, dried over sodium sulfate, filtered and evaporated. The residue obtained was purified by silica gel chromatography eluting with EtOAc/Hexane to afford 1085 (0.4 lg, 72% yield).
1H NMR (300MHz, Chloroform-d) δ ppm 2.03-2.04(s, 3H) 3.66-3.73(m, 7H) 7.21- 7.32(m, 4H).
To a solution of 1085 (0.503g, 2.39mmol) in dichloromethane was added MCPBA (1.338g, 7.78mmol) and the resulting mixture was stirred at room temperature for 4hr before it was diluted with aq. Sodium thiosulfate solution. Organic layer was separated, washed with saturated aq. Sodium bicarbonate solution and water, dried over sodium sulfate, filtered and concentrated. The residue obtained was purified by silica gel chromatography eluting with EtOAc/Hexane to afford 1086 (0.5g, 86% yield). 1H NMR (300MHz, Chloroform-d) δ ppm 2.8(s, 3H) 3.7-3.74(m, 5H) 4.27(s, 2H) 7.30-7.4(m, 4H).
To an ice cold solution of 1086 (0.5g, 2.06mmol) in dioxane (lOmL) and water (lOmL) was added lithium hydroxide monohydrate (0.26g, 6.19mmol) and the resulting reaction mixture was stirred at room temperature for overnight before it was concentrated. The residue obtained was diluted with water and was acidified with acetic acid. The resulting solution was partitioned between water and ethyl acetate. The organic extract was washed with more water, separated, dried over sodium sulfate, filtered and evaporated. The residue obtained was triturated with ether. The solid separated was filtered, washed with ether and dried at high vacuum overnight to afford 1087 (0.3g, 64%yield). 1H NMR (300MHz, Dimethylsulfoxide-d6) δ ppm 2 2(s, 3H) 3.61(s, 2H) 4.48(s, 2H) 7.31-7.35(m, 4H) 12.37(s, 1H).
Compound 634 was prepared using procedures analogous to those above. 1H NMR (300MHz, Dimethylsulfoxide-d6) δ ppm 1.74 (brs, 4H) 2.91 (brs, 5H) 3.03(brs, 2H) 3.78 (s, 2H) 3.85 (s, 2H) 4.49 (s, 2H) 7.32-7.40 (m, 9H) 7.55-7.58 (d, 1H) 8.19 (d, 1H) 11.26 (s, 1H) 12.69 (s, 1H).
Compound 635 was prepared using procedures analogous to those above. 1H NMR (300MHz, Dimethylsulfoxide-d6) δ ppm 1.75 (brs, 4H) 2.91 (brs, 5H) 3.03(brs, 2H) 3.82 (s, 4H) 4.49 (s, 2H) 7.32-7.40 (m, 9H) 7.55-7.58 (d, 1H) 8.19 (d, 1H) 11.26 (s,
To a solution of 1,3-bromo chloropropane (1.57g, lOmmol) in DMF (lOmL) was added sodium thiomethoxide (0.63g, 9mmol) and the resulting reaction mixture was stirred at room temperature overnight and at 70 °C for another day. The solution was partitioned between water and ethyl acetate. The organic extract was washed with more water, separated, dried over sodium sulfate, filtered and evaporated to afford 1088 (1.3gm) which is used for the next step without purification.
To a solution of 1088 (1.3g, 7.7mmol) in dichloromethane (lOOmL) was added MCPBA(5.15g, 23.34mmol) and the resulting mixture was stirred at room temperature for overnight before it was diluted with aq. Sodium thiosulfate solution.
Organic layer was separated, washed with saturated aq. Sodium bicarbonate solution and water, dried over sodium sulfate, filtered and concentrated. The residue obtained was purified by silica gel chromatography eluting with EtOAc/Hexane to afford 1089 (0.3gm). 1H NMR (300MHz, Chloroform-d) δ ppm 2.38-2.49(m, 2H) 2.99(s, 3H) 3.22-3.27(m, 2H) 3.57-3.77(m, 2H).
To a solution of 1092 (0.525g, 3.16mmol) in DMF (15mL) was added potassium carbonate (0.873g, 6.32mmol), 1089 (0.74g, 4.74mmol) and sodium iodide (lOmg). The resulting mixture was stirred at 70 °C overnight before it was diluted with water (-lOOmL). The resulting solution was partitioned between water and ethyl acetate. The organic extract was washed with more water, separated, dried over sodium sulfate, filtered and evaporated. The residue obtained was purified by silica gel chromatography eluting with EtOAc/Hexane to afford 1090 (0.53g, 59% yield). 1H NMR (300MHz, Chloroform-d) δ ppm 2.35-2.40(m, 2H) 2.99(s, 3H) 3.26-3.3 l(m, 2H) 3.63(s, 2H) 3.73(s, 3H) 4.16(t, 2H) 6.81-6.93(m, 3H) 7.25(m, 1H).
To a solution of 1090 (0.53g, 1.85mmol) in dioxane (8mL) and water (4mL) was added lithium hydroxide monohydrate (0.156g, 3.71mmol) and the resulting reaction mixture was stirred at room temperature for 5hr before it was acidified with acetic acid. The resulting solution was partitioned between water and ethyl acetate. The organic extract was washed with more water, separated, dried over sodium sulfate, filtered and evaporated. The residue obtained was triturated with ether. The solid separated was filtered, washed with ether and dried at high vacuum overnight to afford 1091 (0.2g, 40%yield). 1H NMR (300MHz, Chloroform-d) δ ppm 2.32- 2.42(m, 2H) 2.99(s, 3H) 3.26-3.31(m, 2H) 3.66(s, 2H) 4.12-4.16(t, 2H) 6.83-6.94(m -7.3 l(m, 1H).
Compound 583 was prepared by coupling of 1091 with 1024 using procedure described for Amide Coupling General Procedure. 1H NMR (300MHz,
Dimethylsulfoxide-d6) δ ppm 1.74 (brs, 4H) 2.15-2.19(m, 2H) 2.90-3.03(m, 7H) 3.27-
3.39 (m, 2H) 3.78(s, 4H) 4.07-4.11 (t, 2H) 6.90-6.93 (m, 3H) 7.24-7.37 (m, 6H) 7.55- 7 (s, 1H) 12.69 (s, 1H).
Compound 623 was prepared by coupling of 11 with 348 using procedure described for Amide Coupling General Procedure. 1H NMR (300MHz, Dimethylsulfoxide-d6) δ ppm 1.74 (brs, 4H) 2.15-2.19(m, 2H) 2.90-3.03(m, 7H) 3.27-3.39 (m, 2H) 3.75- 3.78(m, 4H) 4.07-4.11 (t, 2H) 6.90-6.97 (m, 3H) 7.26-7.34 (m, 6H) 7.58(d, 1H) 8.19 (d, 1H) 11.26 (s, 1H) 12.69 (s, 1H).
To a solution of 3-hydroxyphenylacetic acid (1 g, 0.00657 mol) in MeOH (10 ml) at 0 °C was added (Trimethylsilyl) diazomethane solution (2 M in hexanes, 20 ml) dropwise. The resulting mixture was stirred at room temperature for 30 minutes before it was evaporated to dryness. The crude material was purified by silica gel chromatography eluting with 0-25% EtOAc in Hexanes to afford 1093.
1094 was made using procedure described for compound 1119.
1095 was made using procedure described for compound 1102.
646 was made using procedure described for compound 666. 1H NMR (300 MHz, CDCls) δ 10.32 (s, 1H), 8.50-8.47 (d, J= 8.52 Hz, 1H), 7.90-7.70 (m, 1H), 7.40-7.36 (m, 6H), 7.03-6.86 (m, 3H), 4.72 (s, 2H), 4.02 (s, 2H), 3.90 (s, 2H), 3.44-3.39 (m, 4H), 3.09-2.96 (d, 4H), 1.87 (bs, 4H), 1.24-1.16 (m, 6H).
647 was made using procedure described for compound 666. 1H NMR (300 MHz, DMSO-de) δ 12.61 (s, 1H), 11.22 (s, 1H), 8.22-8.19 (d, J= 9.18 Hz, 1H), 8.02-8.10 (t, 1H), 7.58-7.55 (d, J= 9.12 Hz, 1H), 7.36-7.24 (m, 5H), 6.99-6.84 (m, 3H), 4.48 (s, 2H), 3.82 (s, 2H), 3.75 (s, 2H), 3.50 (s, 2H), 3.01-2.90 (m, 5H), 1.73 (bs, 4H), 0.82- = 6.69 Hz, 6H).
1096
A solution of hydroxylamine (50% in water, 7.4 mL) was added to acetonitrile (60 mL) and the mixture heated to 90°C for 16 hours. The mixture was cooled to room temperature then cooled in a wet-ice bath giving a precipitate. The solids were collected by filtration and rinsed with cold acetonitrile (10 mL) and dried under high vacuum giving 4.47 g of N'-hydroxyacetimidamide 1096. See Zemolka, S. et al PCT Int Appl 2009118174. 1H NMR 300 MHz CDC13: δ 4.57 (br s, 2H), 1.89 (s, 3H).
1097 1098
A flask was charged with N'-hydroxyacetimidamide 1096 (0.45 g, 6.17 mmol) followed by THF (25 mL), NaH (60% in oil, 0.246 g, 6.17 mmol), 4A molecular sieves (4.5 g) and the mixture heated to 60°C under an atmosphere of argon for 1 hour. A solution of ethyl 2-(3-bromophenyl)acetate 1097 (1.5 g, 6.17 mmol) in THF (12.5 mL) was added to the N'-hydroxyacetimidamide mixture and heated at 60°C for 16 hours. The mixture was diluted with water (100 mL) and extracted with EtOAc (2 x 25 mL). The organic layers were combined, washed with water (25 mL), brine (2 x
25 mL) and dried over Na2S04. The Na2S04 was removed by filtration and the volatiles removed under reduced pressure. The crude material was purified by normal phase chromatography 0-30% EtOAc / hexanes giving 0.56 g of 5-(3-bromobenzyl)- 3-methyl-l,2,4-oxadiazole 1098. 1H NMR 300 MHz CDC13: δ 7.48-7.42 (m, 2H), 7.26-7.24 (m, 2H), 4.15 (s, 2H), 2.38 (s, 3H).
1098 1099
To a solution of 5-(3-bromobenzyl)-3-methyl-l,2,4-oxadiazole 1098 (0.50 g, 1.97 mmol) in dioxane (1 mL), under an atmosphere of Argon, was added Bis(tri-t- butylphosphine)palladium(O) (0.15 g, 0.295 mmol) followed by the addition of 2-tert- butoxy-2-oxoethylzinc chloride (0.5 M in diethyl ether, 4.92 mmol, 9.84 mL). The mixture was allowed to stir under argon for 20 hours and the volatiles were removed under reduced pressure. The residue was taken up in EtOAc (10 mL) and washed with water (2 x 5 mL), brine (2 x 5 mL) and dried over Na2S04. The Na2S04 was removed by filtration and the volatiles removed under reduced pressure. The crude material was purified by normal phase chromatography 0-50% EtOAc / Hexanes to give 0.300 g tert-butyl 2-(3-((3-methyl-l,2,4-oxadiazol-5-yl)methyl)phenyl)acetate 1099. 1H NMR 300 MHz CDC13: δ 7.40-7.18 (m, 4H), 4.17 (s, 2H), 3.51 (s, 2H), 2.36 (s, 3H), 1.43 (s, 9H).
1099 1100
To a mixture of tert-butyl 2-(3-((3-methyl-l,2,4-oxadiazol-5-yl)methyl)phenyl)acetate 1099 (0.127 g, 0.44 mmol) in dioxane (3 mL) was added 4N HCI in dioxane (1 mL) and stirred under an atmosphere of argon for 2 hours. The volatiles were removed under reduced pressure and the residue diluted with water (5 mL) and the pH adjusted to 12 with 2.5 N NaOH. The mixture was washed with dichloromethane (4 x 2 mL) and the pH adjusted to 6 with 1 N HCI. The mixture was extracted with EtOAc (3 x 2
mL) and the organic layers combined, washed with brine and dried over Na2S04. The Na2S04 was removed by filtration and the volatiles removed under reduced pressure to give 0.041 g of 2-(3-((3-methyl-l,2,4-oxadiazol-5-yl)methyl)phenyl)acetic acid 1100. 1H NMR 300 MHz CDC13: δ 7.40-7.18 (m, 4H), 4.18 (s, 2H), 3.63 (s, 2H),
To a solution of N-(5-(4-(6-aminopyridazin-3-yl)butyl)-l,3,4-thiadiazol-2-yl)-2- phenylacetamide 348 (0.061 g, 0.0165 mmol), 2-(3-((3-methyl-l,2,4-oxadiazol-5- yl)methyl)phenyl)acetic acid 1100 (0.040 g, 0.18 mmol), l-ethyl-3-(3- dimethylaminopropyl) carbodiimide (0.078 g, 0.41 mmol), 1-hydroxybenzotriazole (0.055 g, 0.41 mmol) in DMF (3 mL) was added DIEA (0.085 g, 0.115 mL, 0.66 mmol) and the mixture stirred for 16 hours. The mixture was diluted with water (20 mL) and extracted with EtOAc (3 x 20 mL).The organic layers were combined, washed with water (3 x 20 mL), brine (2 x 20 mL) and dried over Na2S04. The Na2S04 was removed by filtration and the volatiles removed under reduced pressure. The crude material was purified by normal phase chromatography 0-5% MeOH / dichloromethane giving 0.003 g of 2-(3-((3-methyl-l,2,4-oxadiazol-5- yl)methyl)phenyl)-N-(6-(4-(5-(2-phenylacetamido)-l,3,4-thiadiazol-2- yl)butyl)pyridazin-3-yl)acetamide 648. 1H NMR 300 MHz CDC13: δ 12.59 (s, 1H), 10.53 (s, 1H), 8.45 (d, 1H, J= 12.2 Hz), 7.4-7.1 (m, 10H), 4.15 (s, 2H), 4.03 (s, 2H),
1101 1102
1101 was made using procedure described for compound 1119.
To a solution of 1101 (470 mg, 1.41 mmol) in MeOH (5 ml) and H20 (5 ml) at 0 °C was added lithium hydroxide monohydrate (296 mg, 7.05 mmol). The resulting mixture was stirred at room temperature for 3 days before it was evaporated to dryness. The mixture was then acidified with IN HCl (pH 4), and it was partitioned between water and EtOAc. The organic extract was washed with water, dried over sodium sulfate, filtered and evaporated to afford 1102.
608 was made using procedure described for compound 664. 1H NMR (300 MHz, DMSO-de) δ 12.71 (s, 1H), 11.32 (s, 1H), 8.22-8.19 (d, J= 9.15 Hz, 1H), 7.58-7.54 (d, J= 9.27 Hz, 1H), 7.38-7.28 (m, 8H), 4.63 (bs, 4H), 3.82 (s, 2H), 3.78 (s, 2H),
-1.44 (d, J = 5.93 Hz, 9H).
612 was made using procedure described for compound 666. 1H NMR (300 MHz, DMSO-de) δ 11.32 (s, 1H), 8.22-8.19 (d, J= 9.78 Hz, 1H), 7.58-7.54 (d, J = 9.72 Hz, 1H), 7.48-7.28 (m, 7H), 4.67-4.61 (m, 4H), 3.88 (s, 2H), 3.80 (s, 2H), 3.01 (bs, 2H),
-1.44 (d, J= 9.93 Hz, 9H).
649 was made using procedure described for compound 695. 1H NMR (300 MHz,
DMSO-de) δ 11.36 (s, 1H), 8.20-8.17 (d, J= 9.78 Hz, 1H), 7.60-7.57 (d, J = 8.92 Hz,
1H), 7.52-7.32 (m, 7H), 4.61-4.56 (d, J= 16.99 Hz, 4H), 3.91 (s, 2H), 3.87 (s, 2H), 3.01 (bs, 2H), 2.90 (bs, 2H), 1.73 (bs, 4H).
650 was made using procedure described for compound 695. 1H NMR (300 MHz, DMSO-de) δ 12.71 (s, 1H), 11.32 (s, 1H), 9.40 (bs, 1H), 8.22-8.19 (d, J= 9.09 Hz, 1H), 7.58-7.54 (d, J= 9.36 Hz, 1H), 7.38-7.28 (m, 8H), 4.63 (bs, 4H), 3.82 (s, 2H), 3.78 (s, 2H), 3.01 (bs, 2H), 2.90 (bs, 2H), 1.73 (bs, 4H).
To a solution of 650 (30 mg, 0.0468 mmol) in DMF (1 ml) at 0 UC was added triethylamine (13 ul, 0.0936 mmol) dropwise followed by acetic anhydride (4.64 ul, 0.0491 mmol) dropwise. The resulting mixture was stirred at 0 °C for 20 minutes before it was quenched by addition of ice water (~5 mL). The white precipitate was collected by suction filtration, rinsed with more water. The crude material was purified by silica gel chromatography eluting with 0-6% MeOH in CH2CI2 to afford 651. 1H NMR (300 MHz, DMSO-d6) δ 12.71 (s, 1H), 11.32 (s, 1H), 8.22-8.19 (d, J = 9.27 Hz, 1H), 7.58-7.54 (d, J= 9.00 Hz, 1H), 7.38-7.28 (m, 8H), 4.88 (bs, 2H), 4.67 (bs, 2H), 3.82 (s, 2H), 3.78 (s, 2H), 3.01 (bs, 2H), 2.90 (bs, 2H), 2.11 (s, 3H), 1.73
1103 1097
To a solution of 2-(3-bromophenyl)acetic acid 1103 (10.0 g, 46.5 mmol) in 100 mL EtOH was added cone. H2SO4 (10 drops) and the mixture heated to relux temperature for 3 hours. The mixture was allowed to cool to room temperature and the volatiles were removed under reduced pressure. The residue was taken up in EtOAc (100 mL) and washed with water (2 x 50 mL), saturated NaHC03 (1 x 25 mL), brine (2 x 25 mL) and dried over Na2S04. The Na2S04 was removed by filtration and the volatiles removed under reduced pressure to give ethyl 2-(3-bromophenyl)acetate 1097 (11.1 grams) as a liquid). 1H NMR 300 MHz CDC13: δ 7.41 (m, 2 H), 7.20 (m, 2H), 4.14 (q, 2H, J= 9.5 Hz), 3.57 (s, 2H), 1.25 (t, 3H, J= 9.5 Hz).
To a solution of ethyl 2-(3-bromophenyl)acetate 1097 (1.5 g, 6.17 mmol) in MeOH (20 mL) was added hydrazine (0.79 g, 24.7 mmol) and the mixture heated to reflux temperature for 4 hours. The mixture was allowed to cool to room temperature giving rise to a white precipitate which was collected by filtration and rinsed with MeOH (10 mL). After drying under reduced pressure 1.4 grams of 2-(3- bromophenyl)acetohydrazide 1104 was isolated. 1H NMR 300 MHz CDC13: δ 7.42 (s, 2H), 7.20 (s, 2H), 6.73 (br s, 1H), 3.51 (s, 2H), 1.81 (br s, 2H).
To a solution of 2-(3-bromophenyl)acetohydrazide 1104 (1.0 g, 4.37 mmol) in AcOH (10 mL) was added trimethylorthoacetate (2.62 g, 21.83 mmol) and the mixture heated to 115°C for 18 hours. The volatiles were removed under reduced pressure and the residue purified by reverse phase chromatography to give 0.59 g of 2-(3- bromobenzyl)-5-methyl-l,3,4-oxadiazole 1105. 1H NMR 300 MHz CDC1
3: δ 7.45 (m, 2H), 7.23 (m, 2H), 4.12 (s, 2H), 2.49 (s, 3H).
1105 1106
To a solution of 2-(3-bromobenzyl)-5 -methyl- 1,3, 4-oxadiazole 1105 (0.50 g, 1.97 mmol) in dioxane (1 mL), under an atmosphere of Argon, was added Bis(tri-t- butylphosphine)palladium(O) (0.15 g, 0.295 mmol) followed by the addition of 2-tert- butoxy-2-oxoethylzinc chloride (0.5 M in diethyl ether, 4.92 mmol, 9.84 mL). The mixture was allowed to stir under Argon for 20 hours and the volatiles were removed under reduced pressure. The residue was taken up in EtOAc (10 mL) and washed with water (2 x 5 mL), brine (2 x 5 mL) and dried over Na2S04. The Na2S04 was removed by filtration and the volatiles removed under reduced pressure. The crude material was purified by normal phase chromatography 0-50% EtOAc / Hexanes to give 0.338 g of tert-butyl 2-(3-((5-methyl-l,3,4-oxadiazol-2-yl)methyl)phenyl)acetate 1106. 1H NMR 300 MHz CDC13: δ 7.24 (m, 4H), 4.12 (s, 2H), 3.51 (s, 2H), 2.46 (s, 3H), 1.43
1106 1107
To a mixture of tert-butyl 2-(3 -((5 -methyl- 1 ,3 ,4-oxadiazol-2-yl)methyl)phenyl)acetate 1106 (0.127 g, 0.44 mmol) in dioxane (3 mL) was added 4N HCI in dioxane (1 mL) and stirred under an atmosphere of Argon for 2 hours. The volatiles were removed under reduced pressure and the residue diluted with water (5 mL) and the pH adjusted to 12 with 2.5 N NaOH. The mixture was washed with dichloromethane (4 x 2 mL) and the pH adjusted to 6 with 1 N HCI. The mixture was extracted with EtOAc (3 x 2 mL) and the organic layers combined, washed with brine and dried over Na2S04. The Na2S04 was removed by filtration and the volatiles removed under reduced pressure to give 0.023 g of 2-(3-((5-methyl-l,3,4-oxadiazol-2-yl)methyl)phenyl)acetic acid 1107.
A solution of N-(5-(4-(6-aminopyridazin-3-yl)butyl)-l ,3,4-thiadiazol-2-yl)-2- phenylacetamide 348 (0.035 g, 0.094 mmol), 2-(3 -((5 -methyl- 1,3, 4-oxadiazol-2- yl)methyl)phenyl)acetic acid 1107 (0.023 g, 0.094 mmol), l-ethyl-3-(3- dimethylaminopropyl) carbodiimide (0.045 g, 0.235 mmol), 1-hydroxybenzotriazole (0.032 g, 0.235 mmol) in DMF (1.75 mL) was stirred for 16 hours and diluted with water (20 mL). The mixture was extracted with EtOAc (3 x 20 mL) the organic layers combined, washed with water (3 x 20 mL), brine (2 x 20 mL) and dried over Na2S04. The Na2S04 was removed by filtration and the volatiles removed under reduced pressure. The crude material was purified by reverse phase chromatography giving 0.004g of 2-(3-((5-methyl-l,3,4-oxadiazol-2-yl)methyl)phenyl)-N-(6-(4-(5-(2- phenylacetamido)-l,3,4-thiadiazol-2-yl)butyl)pyridazin-3-yl)acetamide 652. 1HNMR 300 MHz DMSO-d6: δ 12.62 (s, 1H), 11.24 (s, 1H), 8.16 (d, 1H, J=12.2 Hz), 7.54 (d, 1H, J= 12.2 Hz), 7.3-7.1 (m, 9H), 4.20 (s, 2H), 3.78 (s, 2H), 3.74 (s, 2H), 2.99 (m, 2H), 2.87 (m, 2H), 2.41 (s, 3H), 1.72 (m, 4H).
559
A mixture of 3-bromoacetophenone (5g, 25.1mmol) in formic acid (6gm) and formamide (25mL) was heated to 170 °C for overnight before it was extracted with toluene. Organic layer was separated and concentrated. The residue obtained was diluted with 3N HCl and the resulting mixture was refluxed overnight before it was cooled to room temperature. The solution was extracted with ether. Aqueous layer was separated, basified with aq. Sodium hydroxide solution and extracted with ether. Organic layer was separated, dried over sodium sulfate, filtered and concentrated to afford 1108 (3g, 60% yield). 1H NMR (300MHz, Chloroform-d) δ ppm 1.22-1.25(d, 3H) 3.97-3.99(q, 1H) 7.23-7.4(m, 3H) 7.6(s, 1H).
To a solution of 1108 (2.945g, 14.7mmol) in dichloromethane (lOOmL) was added boc anhydride (3.21g, 14.7mmol) and the reaction mixture was stirred at room temperature overnight before it was concentrated and purified by silica gel chromatography eluting with EtOAc/Hexane to afford 1109 (3g, 68% yield). 1H NMR (300MHz, Dimethylsulfoxide-d6) δ ppm 1.29-1.3 l(d, 3H) 1.38(s, 9H) 4.61- 4.63(q, 1H) 7.3(brs, 2H) 7.41-7.5(m, 3H).
To a degassed solution of 1109 (0.5g, 1.66mmol) and bis(tri-tert- butylphosphine)palladium(O) (0.085g, 0.166mmol) in dioxane(3mL) was added 2-
tert-Butoxy-2-oxoethylzinc chloride (8.5mL, 4.15mmol) under Argon and the resulting reaction mixture was stirred at room temperature for 4hr before it was quenched with saturated aqueous ammonium chloride solution. The resulting solution was partitioned between water and ethyl acetate. The organic extract was washed with more water, separated, dried over sodium sulfate, filtered and evaporated. The residue obtained was purified by silica gel chromatography eluting with EtOAc/Hexane to afford 1110 (0.35g, 62% yield). 1H NMR (300MHz,
Dimethylsulfoxide-d6) δ ppm 1.29-1.31(d, 3H) 1.388-1.42(brs, 18H) 3.53(s, 2H) 4.59-4.63(q, 1H) 7.09 (brs, 1H) 7.12-7.20(brs, 2H) 7.25-7.27(m, 1H) 7.27-7.30(m, 1H).
To a solution of 1110 (0.44g, 1.3mmol) in methanol (40mL) and water (lOmL) was added lithium hydroxide monohydrate (0.4gm) and the resulting reaction mixture was stirred at room temperature for 2days before it was concentrated. The residue obtained was diluted with ice cold water and acidified with acetic acid. The resulting solution was partitioned between water and ethyl acetate. The organic extract was washed with more water, separated, dried over sodium sulfate, filtered and evaporated. The residue obtained was purified by silica gel chromatography eluting with EtOAc/Hexane to afford 1111 (0.316g, 86% yield). 1H NMR (300MHz, Dimethylsulfoxide-d6) δ ppm 1 -1.39(m, 12H) 3.55(s, 2H) 4.58-4.63(q, 1H) 7.1 l-7.38(m, 5H) 12.29(s, 1H).
1H NMR (300MHz, Dimethylsulfoxide-d6) δ ppm 1.43 (m, 12H) 1.89 (brs, 4H) 2.97- 3.08 (m, 4H) 3.95-4.03 (m, 4H) 4.71-4.77 (q, 1H) 7.24-7.43 (m, 11H) 8.45-8.48 (d, 1H) 10.99 (s, 1H) 12.4 (brs, 1H).
1H NMR (300MHz, Dimethylsulfoxide-d6) δ ppm 1.43 (m, 12H) 1.89 (brs, 4H) 2.97- 3.08 (m, 4H) 3.95-4.03 (m, 4H) 4.71-4.77 (q, IH) 7.24-7.43 (m, 1 IH) 8.45-8.48 (d,
.
1H NMR (300MHz, Dimethylsulfoxide-d6) δ ppm 1.5-1.52 (d, 3H) 1.75 (brs, 4H) 2.88-2.93 (m, 2H) 3.03-3.05 (m, 2H) 3.79(s, 2H) 3.86(s, 2H) 4.38-4.44 (q, IH) 7.27- (m, lOH) 8.20-8.23 (m, 4H) 1 1.27 (s, IH) 12.71 (s, IH).
1H NMR (300MHz, Dimethylsulfoxide-d6) δ ppm 1.5-1.52 (d, 3H) 1.75 (brs, 4H) 2.88-2.93 (m, 2H) 3.03-3.05 (m, 2H) 3.86(s, 4H) 4.38-4.44 (q, IH) 7.27-7.59 (m, 1 H) 8.20-8.23 (m, 4H) 11.27 (s, IH) 12.71 (s, IH).
1H NMR (300MHz, Dimethylsulfoxide-d6) δ ppm 1.5-1.52 (d, 3H) 1.75 (brs, 4H) 2.88-2.93 (m, 2H) 3.03-3.05 (m, 2H) 3.78(s, 2H) 3.82(s, 2H) 4.91-4.96 (q, IH) 7.20- 7.35 (m, 9H) 7.55-7.58(d, IH) 8.20-8.23(d, IH) 8.68-8.71 (m, IH) 1 1.27 (s, IH)
To an ice cold solution of l-(5-bromo-2-fluorophenyl)ethanone (4.5g, 20.7mmol) in methanol(lOOmL) was added ammonium acetate(32g, 414.7mmol) and sodium cyanoborohydride(6.15g, 28.98mmol). The reaction mixture was stirred at room temperatue over the weekend before it was concentrated. The residue obtained was diluted with water, basified to pH~13 wih IN NaOH and extracted with
dichloromethane. The organic extract was separated, dried over sodium sulfate, filtered and evaporated. The residue obtained was purified by silica gel
chromatography eluting with EtOAc/Hexane to afford 1112 (1.8g, 40% yield). 1H NMR (300MHz, Dimethylsulfoxide-d6) δ ppm 1.24-1.26(d, 3H) 4.22-4.24(q, IH) 7.1- 7.16(t, IH) 7.41-7.46(m, IH) 7.76(m, IH).
To a solution of 1112 (1.97g, 9mmol) in dichloromethane (lOOmL) was added boc anhydride (1.97g, 9mmol) and the reaction mixture was stirred at room temperature overnight before it was concentrated and purified by silica gel chromatography
eluting with EtOAc/Hexane to afford 1113 (2.4g, 83% yield). 1H NMR (300MHz, Dimethylsulfoxide-d6) δ ppm 1.29-1.32(d, 3H) 1.39(s, 9H) 4.87(q, 1H) 7.14-7.21(t, 1H) 7.46-7.58(m, 3H).
To a degassed solution of 1113 (2.4g, 7.54mmol) and bis(tri-tert- butylphosphine)palladium(O) (0.77g, 1.508mmol) in dioxane(12mL) was added 2- tert-Butoxy-2-oxoethylzinc chloride (38mL, 18.85mmol) under Argon and the resulting reaction mixture was stirred at room temperature for 4hr before it was quenched with saturated aqueous ammonium chloride solution. The resulting solution was partitioned between water and ethyl acetate. The organic extract was washed with more water, separated, dried over sodium sulfate, filtered and
evaporated. The residue obtained was purified by silica gel chromatography eluting with EtOAc/Hexane to afford 1114 (2g, 75% yield). 1H NMR (300MHz,
Dimethylsulfoxide-d6) δ ppm 1.29-1.32(d, 3H) 1.38-1.41(m, 18H) 3.53(s, 2H) 4.87(q, 1H) 7.05-7.16(m, 2H) 7.26-7.29(m, 1H) 7.48(m, 1H).
To a solution of 1114 (2g, 5.66mmol) in methanol (lOOmL) and water (25mL) was added lithium hydroxide monohydrate (2gm) and the resulting reaction mixture was stirred at room temperature for 2days before it was concentrated. The residue obtained was diluted with ice cold water and acidified with acetic acid. The resulting solution was partitioned between water and ethyl acetate. The organic extract was washed with more water, separated, dried over sodium sulfate, filtered and evaporated. The residue obtained was purified by silica gel chromatography eluting with EtOAc/Hexane to afford 1115 (1.5g, 89% yield). 1H NMR (300MHz, Dimethylsulfoxide-d6) δ ppm 1.29-1.3 l(d, 3H) 1.38 (s, 9H) 3.53(s, 2H) 4.87(q, 1H) 7.05-7.19(m, 2H) 7.26-7.29(m, 1H 7.45- 7.48(m, 1H) 12.32(s, 1H).
1H NMR (300MHz, Dimethylsulfoxide-d6) δ ppm 1.30-1.33 (m, 12H) 1.74 (brs, 4H) 2.89(m, 2H) 3.02 (m, 2H) 3.78 (s, 4H) 4.85 (q, IH) 7.10-7.57 (m, 11H) 8.19-8.22 (d, IH 11.26 (s, IH) 12.64 (s, IH).
1H NMR (300MHz, Dimethylsulfoxide-d6) δ ppm 1.28-1.32 (m, 12H) 1.73-1.75 (brs, 4H) 2.87(m, 2H) 2.89 (m, 2H) 3.75 (s, 2H) 3.81(s, 2H) 4.85 (q, IH) 7.06-7.57 (m, 11H 8.18-8.21(d, IH) 11.26 (s, IH) 12.64 (s, IH).
1H NMR (300MHz, Dimethylsulfoxide-d6) δ ppm 1.51-1.53 (m, 3H) 1.75 (brs, 4H) 2.90(m, 2H) 3.02 (m, 2H) 3.78 (s, 2H) 3.85(s, 2H) 4.65 (q, IH) 7.25-7.61 (m, 10H) 8.21-8.25 d, IH) 8.33-8.35(brs, 3H) 11.29 (s, IH) 12.68 (s, IH).
1H NMR (300MHz, Dimethylsulfoxide-d6) δ ppm 1.54 (d, 3H) 1.75-1.76 (brs, 4H) 2.91(m, 2H) 3.02 (m, 2H) 3.81-3.83(m, 4H) 4.65 (q, IH) 7.24-7.63 (m, 10H) 8.22- 8.25 (d, IH) 8.36(brs, 3H) 11.35 (s, IH) 12.66 (s, IH).
To a mixture of 413 (1.62 g) in MeOH (25 mL), THF (10 mL) and H20 (10 mL) at room temperature was added IN aq. NaOH (8 mL). This mixture was stirred for 24 h before the organic volatile was removed under reduced pressure. The residue was neutralized to pH 7 with IN aq. HC1 solution and extracted with EtOAc (2x20 mL). The combined extract was dried (MgS04) and concentrated. The crude was purified by silica gel chromatography eluting with 1-15% MeOH in dichloromethane to afford amine 1116. The resulting amine 1116 was converted to 660 as described for 335. 1H NMR (300 MHz, DMSO-d6) δ 12.68 (bs, 1H), 11.31 (s, 1H), 8.20 (d, J= 9.2 Hz, 1H), 7.57 (d, J= 8.8 Hz, 1H), 7.52-7.21 (m, 8H), 3.90 (s, 2H), 3.87 (s, 2H), 3.06-2.86 (m,
3-Amino-6-chloropyridazine (55.5 g, 0.428 mol) and 3-
(Trifluoromethoxy)phenylacetic acid (1.1 equiv., 0.471 mol, 104 g) were dissolved in DMF (30.0 vol., 1.66 L) in a 3000 mL three neck round-bottom flask. Addition of DIEA (1.1 equiv., 0.471 mol, 82 mL) via addition funnel was done over 5 minutes. Propylphosphonic anhydride solution (300 mL of a 50%> solution in DMF, 1.1 equiv., 0.471 mol, ) was charged into a 500 mL addition funnel and added dropwise to reaction solution (keeping reaction temperature < +30 °C). The reaction usually goes to completion after 3 hours (TLC: 6:4 hexanes-ethyl acetate). Reaction mixture was then poured into 7.5%> sodium bicarbonate (80.0 vol., 4.4 L) which was chilled in an ice bath. Off-white crystalline powder was filtered through a Buchner funnel, rinsed with water (20.0 vol., 1.1 L). Dried in a 50 °C vacuum to a constant weight to afford N-(6-chloropyridazin-3-yl)-2-(3-(trifluoromethoxy)phenyl)acetamide 1117: yield of
119.6 g (77%). 1H NMR (300 MHz, DMSO-d6) δ 11.63 (s, 1H), 8.38(d, J=9.4 Hz, =9.4 Hz, 1H), 7.52 - 7.27(m, 4H), 3.90(s, 2H).
4-Cyanobutylzinc bromide solution (3.0 equiv., 0.50 mol, 1.0 L) was charged into an argon gas purged 5000 mL 3 neck round bottom flask. Argon(g) purge for 5 minutes followed by the addition of 1117 (1.0 equiv., 0.167 mol, 55.3 g) and NiCl2(dppp) (0.15 equiv., 0.0251 mol, 13.6 g) under a blanket of argon(g). The reaction usually goes to completion after 4 hours (TLC: 1 : 1 hexanes-ethyl acetate). EtOAc (15 vol., 832 mL) added to deep red solution. Water (15 vol., 832 mL) was added, thick slurry formed. IN HCl added until slurry breaks to pale blue layer (~6 vol., 333 mL).
Transferred to separatory funnel and organic layer was washed with IN HCl (2x500 mL), dried (MgS04) and concentrated by rotary evaporation (bath < 30 °C) to a solid reddish oil. Oil dissolved in dichloromethane (15 vol., 832 mL), silica gel (100g) was slurried into red solution, this was concentrated by rotary evaporation (bath < 30 °C) to a solid reddish powder. Loaded onto a bed of silica gel (5 cm x 11 cm), flushed with 25% hexanes in ethyl acetate (3 L), combined organics concentrated by rotary evaporation (bath < 30 °C). Dried under high vacuum to a constant weight to afford N-(6-(4-cyanobutyl)pyridazin-3 -yl)-2-(3 -(trifluoromethoxy)phenyl)acetamide 1118: yield of 58.2 g (92%). 1H NMR (300 MHz, DMSO-d6) δ 11.41 (s, 1H), 8.28(d, J=9.2 Hz, 1H), 7.65(d, J=9.2 Hz, 1H), 7.52 - 7.27(m, 4H), 3.89(s, 2H), 2.92(t, J=7.5 Hz,
=7.0 Hz, 2H), 1.80 (m, 2H), 1.61 (m, 2H).
1118 (1.0 equiv., 0.154 mol, 58.2 g) was charged into a 500 mL round bottom flask along with thiosemicarbazide (1.2 equiv., 0.184 mol, 16.8 g). TFA (5 vol., 291 mL) slowly added to reaction vessel while stirring. The reaction slurry was heated in a 65°C bath with an open top reflux condenser. The reaction usually goes to completion after 5 hours (determined by LC/MS). Toluene (10 vol., 582 mL) added
to deep red solution, azeotroped by rotary evaporation (bath < 30 °C) to a red oil. Slowly transferred oil to a well stirred 6000 mL Erlenmeyer flask containing 7.5% sodium bicarbonate solution (69 vol., 4.0 L) cooled in a 0°C bath. The crystals were filtered through a Buchner funnel and rinsed twice with diethyl ether (5 vol., 2x250 mL). Dried under high vacuum to a constant weight to afford N-(6-(4-(5-amino- l,3,4-thiadiazol-2-yl)butyl)pyridazin-3-yl)-2-(3-(trifluoromethoxy)phenyl)acetamide 657; yield of 55.7 g (80%). 1H NMR (300 MHz, DMSO-d6) δ 11.33 (s, 1H), 8.2 l(d, J=9.2 Hz, 1H), 7.58(d, J=9.2 Hz, 1H), 7.51 - 7.26(m, 4H), 6.99(s, 2H), 3.88(s, 2H),
To a solution of 657 (50 mg, 0.11 mmol) in DMF (3 mL) at 0 °C was added 4- fluorophenyl acetic acid (22 mg, 0.14 mmol), HOBt (30 mg, 0.22 mmol) and EDCI (42 mg, 0.22 mmol). The resulting mixture was stirred at room temperature for 1.5 h before it was cooled to 0 °C and quenched with H20. The precipitate was collected by suction filtration and further purified by silica gel chromatography eluting with 1- 10% MeOH in dichloromethane to afford 661. 1H NMR (300 MHz, DMSO-d6) δ 12.65 (bs, 1H), 11.31 (s, 1H), 8.20 (d, J= 9.1 Hz, 1H), 7.57 (d, J= 9.4 Hz, 1H), 7.49- 7.14 (m, 8H), 3.87 (s, 2H), 3.81 (s, 2H), 3.06-2.86 (m, 4H), 1.77-1.72 (m, 4H).
662 was prepared by the procedure as described for compound 661. 1H NMR (300 MHz, DMSO-d6) δ 12.67 (bs, 1H), 11.31 (s, 1H), 8.20 (d, J= 9.1 Hz, 1H), 7.57 (d, J
= 9.1 Hz, 1H), 7.51-7.07 (m, 7H), 3.89 (s, 2H), 3.87 (s, 2H), 3.06-2.86 (m, 4H), -1.72 (m, 4H).
663 was prepared by the procedure as described for compound 661. 1H NMR (300 MHz, DMSO-de) δ 12.74 (bs, 1H), 11.31 (s, 1H), 8.20 (d, J= 9.2 Hz, 1H), 7.57 (d, J = 9.2 Hz, 1H), 7.51-7.19 (m, 7H), 3.97 (s, 2H), 3.87 (s, 2H), 3.06-2.86 (m, 4H), -1.72 (m, 4H).
1119 1120
To a mixture of l-bromo-3-(difluoromethoxy) benzene (1 g, 4.5 mmol), bis(tri-tert- butylphosphine) palladium(O) (460 mg, 0.9 mmol) in 1,4-dioxane (30 ml) under argon atmosphere was added 0.5 M of 2-tert-butoxy-2-oxoethyl zinc chloride in ether (22.5 ml). The resulting mixture was stirred at room temperature overnight. The mixture was partitioned between saturated NH4CI and EtOAc. The organic extract was washed with brine, dried over sodium sulfate, filtered and evaporated. The crude material was purified by silica gel chromatography eluting with 0-10% EtOAc in Hexane to afford 1119.
To a solution of 1119 (300 mg, 1.16 mmol) in dichloromethane (5 ml) at 0 °C was added TFA (3 ml) dropwise. The resulting mixture was stirred at room temperature overnight before it was evaporated to dryness then triturated the residue with ether to afford 1120.
1121
1121 was made using procedure described for compound 1120 from l-Bromo-3- (2,2,2-trifluoroethoxy)benzene.
A flask was charged with 1024 (50 mg, 0.135 mmol), 1120 (28 mg, 0.142 mmol) in DMF (1 ml) at 0 °C was added HOBT (39 mg, 0.285 mmol) followed by EDCI (68 mg, 0.356 mmol). The resulting mixture was slowly warmed up to room temperature and stirred for 2 h before it was quenched by addition of ice water (~5 mL). The white precipitate was collected by suction filtration, rinsed with more water to afford 664. 1H NMR (300 MHz, DMSO-d6) δ 12.71 (s, 1H), 11.32 (s, 1H), 8.22-8.19 (d, J = 9.12 Hz, 1H), 7.58-7.54 (d, J= 9.03 Hz, 1H), 7.48-6.99 (m, 10H), 3.85 (s, 2H), 3.78 (s, 2H), 3.01 (bs, 2H), 2.90 (bs, 2H), 1.73 (bs, 4H).
665 was made using procedure described for compound 664. 1H NMR (300 MHz, DMSO-de) δ 12.71 (s, 1H), 11.32 (s, 1H), 8.22-8.19 (d, J= 9.12 Hz, 1H), 7.58-7.54 (d, J= 9.03 Hz, 1H), 7.38-7.28 (m, 6H), 7.03-6.97 (m, 3H), 4.77-4.74 (q, 2H), 3.80- 3.78 (d, J= 5.82 Hz, 4H), 3.01 (bs, 2H), 2.90 (bs, 2H), 1.73 (bs, 4H).
A flask was charged with 348 (50 mg, 0.135 mmol), 1120 (28 mg, 0.142 mmol) in DMF (1 ml) at 0 °C was added HOBT (39 mg, 0.285 mmol) followed by EDCI (68 mg, 0.356 mmol). The resulting mixture was slowly warmed up to room temperature
and stirred overnight before it was quenched by addition of ice water (~5 mL). The white precipitate was collected by suction filtration, rinsed with more water. The crude material was purified by silica gel chromatography eluting with 0-6% MeOH in dichloromethane to afford 666. 1H NMR (300 MHz, DMSO-d6) δ 12.71 (s, 1H), 11.32 (s, 1H), 8.22-8.19 (d, J= 9.12 Hz, 1H), 7.58-7.54 (d, J= 9.03 Hz, 1H), 7.48- 6.98 (m, 10H), 3.81 (bs, 4H), 3.01 (bs, 2H), 2.90 (bs, 2H), 1.73 (bs, 4H).
667 was made using procedure described for compound 666. 1H NMR (300 MHz, DMSO-de) δ 12.71 (s, 1H), 11.32 (s, 1H), 8.22-8.19 (d, J= 9.12 Hz, 1H), 7.58-7.54 (d, J= 8.97 Hz, 1H), 7.35-7.28 (m, 6H), 7.03-6.97 (m, 3H), 4.77-4.74 (q, 2H), 3.87 (s, 2H), 3.78 (s, 2H), 3.01 (bs, 2H), 2.90 (bs, 2H), 1.73 (bs, 4H).
668 was made using procedure described for compound 675. 1H NMR (300 MHz, DMSO-d6) δ 12.71 (s, 1H), 11.32 (s, 1H), 8.22-8.19 (d, J= 9.15 Hz, 1H), 7.58-6.99 (m, 10H), 3.87-3.84 (d, 4H), 3.01 (bs, 2H), 2.90 (bs, 2H), 1.73 (bs, 4H).
669 was made using procedure described for compound 675. 1H NMR (300 MHz, DMSO-de) δ 12.71 (s, 1H), 11.32 (s, 1H), 8.22-8.19 (d, J= 9.09 Hz, 1H), 7.58-7.54
(d, J= 9.37 Hz, 1H), 7.48-7.28 (m, 6H), 7.03-6.97 (m, 2H), 4.77-4.74 (q, 2H), 3.87
A flask was charged with 657 (50 mg, 0.111 mmol), 2-pyridine acetic acid hydrochloride (20 mg, 0.116 mmol) in DMF (1 ml) at 0 °C was treated with propylphosphonic anhydride solution (91 ul) followed by triethylamine (40 ul, 0.29 mmol). The resulting mixture was slowly warmed up to room temperature and stirred for 1 h before it was quenched by addition of ice water (~5 mL). The yellow precipitate was collected by suction filtration, rinsed with more water. The crude material was purified by silica gel chromatography eluting with 0-6% MeOH in dichloromethane to afford 670. 1H NMR (300 MHz, DMSO-d6) δ 12.67 (s, 1H), 11.32 (s, 1H), 8.53-8.49 (m, 1H), 8.22-8.19 (d, J= 9.12 Hz, 1H), 7.78-7.76 (t, 1H), 7.58-7.26 (m, 7H), 4.01 (s, 2H), 3.87 (s, 2H), 3.01 (bs, 2H), 2.90 (bs, 2H), 1.73 (bs,
671 was made using procedure described for compound 670. 1H NMR (300 MHz, DMSO-de) δ 12.70 (s, 1H), 11.32 (s, 1H), 8.53-8.48 (m, 2H), 8.22-8.19 (d, J= 9.12 Hz, 1H), 7.76-7.26 (m, 7H), 3.87 (s, 4H), 3.01 (bs, 2H), 2.90 (bs, 2H), 1.73 (bs, 4H).
672 was made using procedure described for compound 670. 1H NMR (300 MHz, DMSO-de) δ 11.32 (s, IH), 8.53-8.52 (bs, 2H), 8.22-8.19 (d, J= 9.12 Hz, IH), 7.58- 7.26 (m, 7H), 3.87 (s, 4H), 3.01 (bs, 2H), 2.90 (bs, 2H), 1.73 (bs, 4H).
673 was prepared by the procedure as described for compound 661. 1H NMR (300 MHz, DMSO-d6) δ 12.69 (bs, IH), 11.31 (s, IH), 8.20 (d, J= 9.1 Hz, IH), 7.57 (d, J = 9.1 Hz, IH), 7.51-7.21 (m, 8H), 3.90 (s, 2H), 3.87 (s, 2H), 3.06-2.86 (m, 4H), -1.72 (m, 4H).
674 was prepared by the procedure as described for compound 661. 1H NMR (300 MHz, DMSO-de) δ 12.63 (bs, IH), 11.32 (s, IH), 8.20 (d, J= 9.2 Hz, IH), 7.57 (d, J = 9.2 Hz, IH), 7.51-7.38 (m, 3H), 7.33-7.09 (m, 5H), 3.87 (s, 2H), 3.79 (s, 2H), 3.06-2.86 (m, 4H), 2.48 (s, 3H), 1.77-1.72 (m, 4H).
A flask was charged with 657 (70 mg, 0.155 mmol), 5-pyrimidineacetic acid (22 mg, 0.162 mmol) in DMF (1 ml) at 0 °C was added HOBT (44 mg, 0.326 mmol) followed by EDCI (78 mg, 0.408 mmol). The resulting mixture was slowly warmed up to room temperature and stirred for overnight before it was quenched by addition of ice water (~5 mL). The white precipitate was collected by suction filtration, rinsed with more water. The crude material was purified by silica gel chromatography eluting with 0- 6% MeOH in dichloromethane to afford 675. 1H NMR (300 MHz, DMSO-d6) δ
12.75 (s, 1H), 11.32 (s, 1H), 9.11 (s, 1H), 8.76 (s, 1H), 8.22-8.19 (d, J= 9.12 Hz, 1H), 7.59-7.26 (m, 6H), 3.94 (s, 2H), 3.87 (s, 2H), 3.01 (bs, 2H), 2.90 (bs, 2H), 1.73 (bs, 4H).
676 was made using procedure described for compound 675. 1H NMR (300 MHz, DMSO-de) δ 12.75 (s, 1H), 11.32 (s, 1H), 8.70 (s, 1H), 8.61-8.57 (m, 2H), 8.22-8.19 (d, J= 9.36 Hz, 1H), 7.59-7.26 (m, 5H), 4.11 (s, 2H), 3.87 (s, 2H), 3.01 (bs, 2H), 2.90 (bs, 2H), 1.73 (bs, 4H).
677 was made using procedure described for compound 675. 1H NMR (300 MHz, DMSO-de) δ 12.75 (s, 1H), 11.32 (s, 1H), 8.89 (s, 1H), 8.22-8.19 (d, J= 9.15 Hz, 1H),
7.59-7.26 (m, 5H), 6.62 (s, IH), 3.99 (s, 2H), 3.87 (s, 2H), 3.01 (bs, 2H), 2.90 (bs,
2H), 1.73 (bs, 4H).
678 was made using procedure described for compound 675. 1H NMR (300 MHz, DMSO-d6) δ 12.75 (s, IH), 11.32 (s, IH), 9.06 (s, IH), 8.22-8.19 (d, J= 9.21 Hz, IH), 7.59-7.26 (m, 6H), 4.03 (s, 2H), 3.87 (s, 2H), 3.01 (bs, 2H), 2.90 (bs, 2H), 1.73 (bs,
679 was prepared by the procedure as described for compound 661. 1H NMR (300 MHz, DMSO-d6) δ 12.67 (bs, IH), 11.31 (s, IH), 8.20 (d, J= 9.2 Hz, IH), 7.57 (d, J = 9.2 Hz, IH), 7.51-7.36 (m, 4H), 7.29-7.12 (m, 4H), 3.87 (s, 2H), 3.85 (s, 2H),
-2.86 (m, 4H), 1.77-1.72 (m, 4H).
680 was prepared by the procedure as described for compound 661. 1H NMR (300 MHz, DMSO-d
6) δ 12.67 (bs, IH), 11.31 (s, IH), 8.20 (d, J= 9.3 Hz, IH), 7.57 (d, J = 9.0 Hz, IH), 7.51-7.28 (m, 8H), 3.87 (s, 2H), 3.84 (s, 2H), 3.06-2.86 (m, 4H), 1.77-1.72 (m, 4H).
To a solution of 674 (100 mg, 0.16 mmol) in dichloromethane at -78 °C was added m- CPBA (60 mg, 0.24 mmol) in 4 portions. The resulting mixture was stirred at that temperature for 1 h before it was slowly warmed up to -10 °C and quenched with 25% aq. Na2S203 solution. The reaction was diluted with EtOAc, washed with saturated aq. NaHC03 (3x 10 mL). The combined organic layer was separated, washed with brine, dried (MgS04) and concentrated. The crude was purified by HPLC to afford 682. 1H NMR (300 MHz, DMSO-d6) δ 12.72 (bs, 1H), 11.31 (s, 1H), 8.20 (d, J= 9.0 Hz, 1H), 7.68 (m, 1H), 7.60-7.26 (m, 8H), 3.91 (s, 2H), 3.87 (s, 2H), -2.86 (m, 4H), 2.76 (s, 3H), 1.77-1.72 (m, 4H).
681 was prepared from 657 and 3-methylsulphonylphenyl acetic acid by the procedure as described for compound 661. 1H NMR (300 MHz, DMSO-d6) δ 12.72
(bs, 1H), 11.31 (s, 1H), 8.20 (d, J= 9.0 Hz, 1H), 7.92 - 7.83 (m, 2H), 7.70-7.26 (m,
7H), 3.93 (s, 2H), 3.87 (s, 2H), 3.23 (s, 3H), 3.06-2.86 (m, 4H), 1.77-1.72 (m, 4H).
683 was made using procedure described for compound 675. 1H NMR (300 MHz, DMSO-de) δ 12.75 (s, 1H), 11.32 (s, 1H), 8.36 (s, 1H), 8.21-8.18 (d, J= 9.18 Hz, 1H),
7.84-7.80 (d, J= 9.36 Hz, 1H), 7.59-7.26 (m, 6H), 3.90-3.87 (d, 4H), 3.01 (bs, 2H),
684 was made using procedure described for compound 675. 1H NMR (300 MHz, DMSO-de) δ 12.75 (s, 1H), 11.32 (s, 1H), 8.57 (s, 1H), 8.51-8.49 (d, J= 9.18 Hz, 1H), 8.21-8.18 (d, J= 9.06 Hz, 1H), 7.79-7.75 (d, J= 9.36 Hz, 1H), 7.59-7.26 (m, 6H), 4.07 (t, 2H), 3.87 (s, 2H), 3.30-3.28 (m, 1H), 3.19 (s, 3H), 3.01 (bs, 2H), 2.90 (bs,
1.73 (bs, 4H).
685 was prepared by the procedure as described for compound 661. 1H NMR (300 MHz, DMSO-de) δ 12.52 (bs, 1H), 11.31 (s, 1H), 8.20 (d, J= 9.1 Hz, 1H), 7.61-7.25 (m, 7H), 3.87 (s, 2H), 3.80 (s, 3H), 3.62 (s, 2H), 3.06-2.86 (m, 4H), 1.77-1.72 (m,
686 was prepared by the procedure as described for compound 661. 1H NMR (300
MHz, DMSO-de) δ 12.53 (bs, 1H), 11.32 (s, 1H), 8.20 (d, J= 9.1 Hz, 1H), 7.58 (d, J
= 9.2 Hz, 1H), 7.52-7.26 (m, 4H), 5.96 (s, 1H), 3.87 (s, 2H), 3.67 (s, 2H), 3.64 (s, -2.86 (m, 4H), 2.21 (s, 3H), 1.77-1.72 (m, 4H).
687 was prepared by the procedure as described for compound 661. 1H NMR (300 MHz, DMSO-de) δ 12.56 (bs, 1H), 11.32 (s, 1H), 8.20 (d, J= 9.3 Hz, 1H), 7.61-7.38 (m, 6H), 6.17 (d, J= 2.2 Hz, 1H), 3.87 (s, 2H), 3.79 (s, 3H), 3.75 (s, 2H), 3.03-2.90
-1.72 (m, 4H).
688 was prepared by the procedure as described for compound 661. 1H NMR (300 MHz, DMSO-dg) δ 12.61 (bs, 1H), 11.32 (s, 1H), 8.20 (d, J= 9.3 Hz, 1H), 7.58 (d, J = 9.3 Hz, 1H), 7.51-7.26 (m, 4H), 3.87 (s, 2H), 3.84 (s, 2H), 3.07-2.86 (m, 4H), -1.72 (m, 4H).
To a solution of 657 (200 mg, 0.44 mmol) in DMF (4 mL) at 0 °C was added mandelic acid (124 mg, 0.66 mmol), HOBt (119 mg, 0.88 mmol) and EDCI (170 mg, 0.88 mmol). The resulting mixture was stirred at room temperature for 1.5 h before it was cooled to 0 °C and quenched with H
20. The precipitate was collected by suction filtration and further purified by silica gel chromatography eluting with 1-10%
MeOH in dichloromethane to afford 690 and a more polar 689. 689: 1H NMR (300 MHz, DMSO-de) δ 12.42 (bs, 1H), 11.31 (s, 1H), 8.20 (d, J= 9.2 Hz, 1H), 7.58-7.27 (m, 10H), 6.35 (d, J= 4.4 Hz, 1H), 5.34 (d, J= 4.3 Hz, 1H), 3.87 (s, 2H), 3.03-2.89 (m, 4H), 1.77-1.73 (m, 4H). 690: 1H NMR (300 MHz, DMSO-d6) δ 13.05 (bs, 1H), 11.31 (s, 1H), 8.20 (d, J= 9.0 Hz, 1H), 7.59-7.26 (m, 15H), 6.26 (d, J= 5.5 Hz, 1H), 6.11 (s, 1H), 5.38 (d, J= 5.3 Hz, 1H), 3.87 (s, 2H), 3.03-2.88 (m, 4H), 1.76-1.73 (m, 4H).
447 was prepared from 657 and 3-chloromandelic acid by the procedure as described for compound 689. 1H NMR (300 MHz, DMSO-d6) δ 12.48 (bs, 1H), 11.31 (s, 1H), 8.20 (d, J= 9.2 Hz, 1H), 7.59-7.26 (m, 9H), 6.53 (m, 1H), 5.36 (t, J= 0.7 Hz, 1H), 3.87 (s, 2H), 3.03-2.90 (m, 4H), 1.75-1.71 (m, 4H).
692 was made using procedure described for compound 675. 1H NMR (300 MHz, DMSO-de) δ 12.75 (s, 1H), 11.32 (s, 1H), 8.21-8.18 (d, J= 9.18 Hz, 1H), 7.80-7.26 (m, 9H), 3.92 (s, 2H), 3.87 (s, 2H), 3.01 (bs, 2H), 2.90 (bs, 2H), 1.73 (bs, 4H).
693 was made using procedure described for compound 675. 1H NMR (300 MHz, DMSO-de) δ 12.75 (s, 1H), 11.32 (s, 1H), 8.22-8.19 (d, J= 9.06 Hz, 1H), 7.79 (s, 1H), 7.59-7.26 (m, 6H), 6.31 (s, 1H), 5.20 (s, 2H), 3.87 (s, 2H), 3.01 (bs, 2H), 2.90 (bs, 2H), 1.73 (bs, 4H).
694 was made using procedure described for compound 675. 1H NMR (300 MHz, DMSO-de) δ 12.71 (s, 1H), 11.32 (s, 1H), 8.22-8.18 (d, J= 9.15 Hz, 1H), 7.58-7.54 (d, J= 9.18 Hz, 1H), 7.48-7.26 (m, 4H), 3.87 (s, 2H), 3.63 (s, 2H), 3.01 (bs, 2H), 2.90 (bs, 2H), 2.39 (s, 3H), 2.13 (s, 3H), 1.73 (bs, 4H), 1.57 (s, 9H).
To a solution of 694 (50 mg, 0.081 mmol) in dichloromethane (2 ml) was added TFA (2 ml) at 0 °C. The resulting mixture was stirred at room temperature for 1 h before it was evaporated under vacuo to dryness. Ether was added and the white precipitate was collected by suction filtration, rinsed with more ether to afford 695. 1H NMR (300 MHz, DMSO-d
6) δ 12.71 (s, 1H), 11.32 (s, 1H), 8.22-8.19 (d, J= 9.36 Hz, 1H), 7.60-7.57 (d, J= 9.27 Hz, 1H), 7.51-7.28 (m, 4H), 3.88 (s, 2H), 3.57 (s, 2H), 3.01 (bs, 2H), 2.90 (bs, 2H), 2.45 (s, 3H), 2.15 (s, 3H), 1.73 (bs, 4H).
696 was made using procedure described for compound 695. 1H NMR (300 MHz, DMSO-de) δ 12.71 (s, 1H), 11.32 (s, 1H), 8.22-8.19 (d, J= 9.30 Hz, 1H), 8.15 (s, 1H), 7.58-7.54 (d, J= 9.30 Hz, 1H), 7.48-7.28 (m, 5H), 3.87 (s, 2H), 3.76 (s, 2H), 3.01
1.59 (s, 9H).
697 was made using procedure described for compound 695. 1H NMR (300 MHz, DMSO-de) δ 14.22 (s, 1H), 12.71 (s, 1H), 11.32 (s, 1H), 9.01 (s, 1H), 8.22-8.19 (d, J = 9.15 Hz, 1H), 7.59-7.26 (m, 6H), 4.04 (s, 2H), 3.87 (s, 2H), 3.01 (bs, 2H), 2.90 (bs,
To a suspension of
acid hydrochloride (113 mg, 0.58 mmol) in DMF (8 mL) at 0 °C was added N-(3-dimethylaminopropyl)-N'- ethylcarbodiimide hydrochloride (130 mg, 0.67 mmol). The resulting mixture was stirred at 0 °C for 40 min and followed by addition of 689 (300 mg, 0.48 mmol) and 4-DMAP (165 mg, 1.35 mmol). The resulting mixture was stirred from 0 °C to room temperature over a period of 3.5 h before it was diluted with EtOAc and cold water. The organic layer was separated and washed with water (3x 15 mL), brine, dried (MgS0
4) and concentrated. The crude product was purified by silica gel
chromatography eluting with 0-15% MeOH in CH2CI2 to provide 711 (297 mg) as white solid. 1H NMR (300 MHz, CDC13) δ 10.75 (bs, 1H), 8.49 (d, J= 9.0 Hz, 1H),
10<
7.64 (s, 1H), 7.50-7.26 (m, 7H), 7.16-7.15 (m, 1H), 6.51 (s, 1H), 4.04 (s, 2H), 3.80- 3.72 (m, 4H), 3.88-2.81 (m, 8H), 2.75-2.71 (m, 5H), 1.89 (m, 4H).
A mixture of 1117 (4.00 g, 12.06 mmol), 4-pentynenitrile (2.11 mL, 24.12 mmol), PdCl
2(PPh
3)
2 (847 mg, 1.21 mmol), Cul (184 mg, 0.96 mmol) and Et
3N (13.44 mL, 96.48 mmoL) in DMF (18 mL) was heated at 55 °C for 5 h. The reaction was cooled to room temperature and poured into a mixture of ice-water. The precipitate was collected by suction filtration and air dried. The crude product was further recrystallized from a mixture of i-PrOH-H
20 first and then from i-PrOH to provide alkyne 1131.
A mixture of alkyne 1131 (6.00 g) and Pd(OH)2/C (1.00 g) in a mixture of EtOAc (150 mL), THF (75 mL) and MeOH (75 mL) was stirred under 1 atm of D2 at room temperature for 3 h before the catalyst was filtered off a short plug of Si02 and rinsed with EtOAc. The filtrate was concentrated to provide the crude product which was further recrystallized from a mixture of EtOAc and ether to give the desired alkane 1132 as off-white solid (6.01 g)
A mixture of nitrile 1132 (5.20 g, 13.61 mmol) and thiosemicarbazide (1.61 g, 17.69 mmol) in TFA (75 mL) was heated at 80 °C for 4 h. The reaction was cooled to room temperature and poured into a mixture of ice-water. The mixture was basified with NaOH pellets (pH 14). The white precipitate was collected by suction filtration, rinsed with water and dried to provide 726 (5.87 g).
To a solution of 726 (1.40 g, 3.07 mmol) and 2-pyridylacetic acid HCl salt (1.49 g, 8.59 mmol) in DMF (20 mL) at 0 °C was added Et3N (1.50 mL, 10.73 mmol) and followed by 1-propanephosphonic anhydride (2.73 mL, 50% in DMF, 4.29 mmol). This mixture was stirred for 2.5 h at room temperature before it was cooled back to 0 °C and quenched with ice-H20. The precipitate was collected by suction filtration and air dried. This crude product was further purified by silica gel chromatography eluting with 0-15% MeOH in DCM to afford 727 (0.97 g). 1H NMR (300 MHz, DMSO-de) δ 12.67 (s, 1H), 11.31 (s, 1H), 8.52-8.50 (m, 1H), 8.20 (d, J = 9.2 Hz, 1H), 7.78 (dt, J = 1.8, 7.6 Hz, 1H), 7.58 (d, J = 9.1 Hz, 1H), 7.51-7.26 (m, 6H), 4.02
= 7.4 Hz, 2H), 1.73 (t, J= 7.4 Hz, 2H).
Compound 710 was prepared from compound 447 using a procedure analogous to that employed for the preparation of compound 711. 1H NMR (300 MHz, DMSO-d6) δ 11.32 (s, 1H), 8.21-8.18 (d, J= 9.06 Hz, 1H), 7.62-7.26 (m, 9H), 6.16 (s, 1H), 3.87 (s, 2H), 3.52-3.50 (d, 2H), 3.01 (bs, 2H), 2.90 (bs, 2H), 2.80-2.71(m, 11H), 1.73 (bs, 4H).
Compound 712 was prepared from compound 447 using a procedure analogous to that employed for the preparation of compound 711. 1H NMR (300 MHz, DMSO-d
6) δ 11.32 (s, 1H), 8.21-8.18 (d, J= 9.06 Hz, 1H), 7.62-7.26 (m, 9H), 6.16 (s, 1H), 3.87 (s, 2H), 3.38-3.36 (d, 2H), 3.01 (bs, 2H), 2.90 (bs, 2H), 2.29 (s, 6H), 1.73 (bs, 4H).
Compound 713 was prepared from compound 447 using a procedure analogous to that employed for the preparation of compound 711. 1H NMR (300 MHz, DMSO-d6) δ 13.11 (bs, 1H), 11.32 (s, 1H), 8.21-8.18 (d, J= 9.06 Hz, 1H), 7.62-7.26 (m, 9H), 6.16 (s, 1H), 3.87 (s, 2H), 3.60-3.57 (m, 4H), 3.44-3.42 (d, 2H), 3.01 (bs, 2H), 2.90 (bs,
-2.51 (m, 4H), 1.73 (bs, 4H).
Compound 714 was prepared from compound 447 using a procedure analogous to that employed for the preparation of compound 711. 1H NMR (300 MHz, DMSO-d6) δ 11.32 (s, 1H), 8.21-8.18 (d, J= 9.06 Hz, 1H), 7.62-7.26 (m, 9H), 6.16 (s, 1H), 3.87 (s, 2H), 3.38-3.31 (d, 2H), 3.01 (bs, 2H), 2.90 (bs, 2H), 2.49-2.47 (m, 4H), 1.93 (bs, 4H),
To a suspension of 670 (3 g, 5.24 mmol) in MeOH (50 ml) at 0 UC was added 2N NaOH (20 ml) solution. The resulting mixture was stirred at room temperature overnight. The solvent was evaporated under vacuo and the mixture was acidified
with IN HCl to pH 6. The white precipitate was collected by suction filtration, rinsed with more water and dried to afford 1121a. 1H NMR (300 MHz, DMSO-d6) δ 12.66 (s, 1H), 8.51-8.50 (m, 1H), 7.81-7.76 (m, 1H), 7.42-7.28 (m, 2H), 7.16-7.13 (d, 1H), -6.70 (d, 1H), 6.10 (s, 2H), 4.0 (s, 2H), 3.01 (bs, 2H), 2.71 (bs, 2H), 1.70 (bs, 4H).
To a solution of 1121a (20 mg, 0.054 mmol) in DMF (1 ml) at 0 UC was added triethylamine (11 ul, 0.081 mmol) drop wise followed by o-acetylmandelic acid chloride (15 ul, 0.065 mmol) drop wise. The resulting mixture was slowly warmed up to room temperature and stirred for 1 h before it was quenched by addition of water (~3 mL) at 0 °C. The mixture was partitioned between water and EtOAc. The organic extract was washed with brine, dried over sodium sulfate, filtered and evaporated. The crude material was purified by silica gel chromatography eluting with 0-5% MeOH in DCM to afford 1122.
A flask was charged with 1122 (20 mg, 0.037 mmol) and 2N ammonia in MeOH (5 ml). The mixture was stirred at room temperature for 2 hours. The solvent was evaporated under vacuo and the mixture was triturated with ether. The white precipitate was collected by suction filtration, rinsed with ether and dried to afford
715. 1H NMR (300 MHz, DMSO-d
6) δ 12.66 (s, 1H), 10.61 (s, 1H), 8.51-8.50 (m, 1H), 8.21-8.18 (d, J= 9.06 Hz, 1H), 7.81-7.76 (m, 1H), 7.61-7.53 (m, 3H), 7.42-7.28 (m, 5H), 6.49-6.47 (d, 1H), 5.30-5.28 (d, 1H), 4.0 (s, 2H), 3.02 (bs, 2H), 2.91 (bs, 2H), 1.75 (bs, 4H).
Compound 719 was prepared using a procedure analogous to that employed for the preparation of compound 670. 1H NMR (300 MHz, DMSO-d6) δ 12.66 (s, 1H), 11.32 (s, 1H), 8.51-8.50 (m, 1H), 8.21-8.18 (d, J= 9.06 Hz, 1H), 7.79-7.76 (m, 1H), 7.59- 3.01 (bs, 2H), 2.90 (bs, 2H), 1.75 (bs, 4H).
Compound 720 was prepared using a procedure analogous to that employed for the preparation of compound 670. 1H NMR (300 MHz, DMSO-d6) δ 12.66 (s, 1H), 11.32
(s, 1H), 8.51-8.50 (m, 1H), 8.19-8.16 (d, J= 9.06 Hz, 1H), 7.79-7.76 (m, 1H), 7.59-
7.30 (m, 6H), 4.01 (s, 2H), 3.95 (s, 2H), 3.03 (bs, 2H), 2.91 (bs, 2H), 1.76 (bs, 4H).
Compound 721 was prepared using a procedure analogous to that employed for the preparation of compound 670. 1H NMR (300 MHz, DMSO-d
6) δ 12.66 (s, 1H), 11.32 (s, 1H), 8.51-8.50 (m, 1H), 8.21-8.16 (d, J= 9.06 Hz, 1H), 7.81-7.28 (m, 7H), 4.01 (s, 2H), 3.89 (s, 2H), 3.03 (bs, 2H), 2.91 (bs, 2H), 1.76 (bs, 4H).
Compound 717 was prepared using a procedure analogous to that employed for the preparation of compound 670. 1H NMR (300 MHz, DMSO-d6) δ 12.66 (s, IH), 11.17 (s, IH), 8.52-8.50 (m, IH), 8.19-8.16 (d, J= 9.06 Hz, IH), 7.81-7.76 (m, IH), 7.58- 7.55 (d, IH), 7.42-7.09 (m, 4H), 7.08-7.06 (d, IH), 4.01 (s, 2H), 3.83 (s, 2H), 3.79 (s, 3H), 3.03 (bs, 2H), 2.91 (bs, 2H), 1.76 (bs, 4H).
To a solution of 717 (10 mg, 0.017 mmol) in DCM (3 ml) at 0 C was added boron tribromide solution (IN in DCM) (2 ml) drop wise. The resulting mixture was slowly warmed up to room temperature and stirred for 4.5 h before it was quenched by addition of water (~3 mL). The mixture was then basified with IN NaOH to pH 8. The mixture was partitioned between water and DCM. The organic extract was washed with brine, dried over sodium sulfate, filtered and evaporated. The crude material was purified by silica gel chromatography eluting with 0-10% MeOH in
DCM to afford 718. 1H NMR (300 MHz, DMSO-d6) δ 11.17 (s, IH), 8.52-8.50 (m, IH), 8.21-8.18 (d, J= 9.06 Hz, IH), 7.81-7.76 (m, IH), 7.58-7.55 (d, IH), 7.51-7.09 (m, 4H), 6.88-6.85 (d, IH), 4.0 (s, 2H), 3.79 (s, 2H), 3.03 (bs, 2H), 2.91 (bs, 2H), 1.76 (bs, 4H).
Compound 1128 was prepared from 4-bromo-2-trifluoromethoxyanisole using a procedure analogous to that for compound 1124 below.
Compound 722 was prepared using compound 1128 with a procedure analogous to that for compound 670. 1H NMR (300 MHz, DMSO-d6) δ 12.66 (s, 1H), 11.17 (s, 1H), 8.52-8.50 (m, 1H), 8.21-8.18 (d, J= 9.06 Hz, 1H), 7.81-7.76 (m, 1H), 7.58-7.55 (d, 1H), 7.42-7.19 (m, 5H), 4.0 (s, 2H), 3.85 (s, 3H), 3.79 (s, 2H), 3.03 (bs, 2H), 2.91 (bs, 2H), 1.76 (bs, 4H).
Compound 723 was prepared from compound 722 using a procedure analogous to that for the preparation of compound 718 above. 1H NMR (300 MHz, DMSO-d6) δ 12.66 (s, 1H), 11.17 (s, 1H), 10.06 (s, 1H), 8.52-8.50 (m, 1H), 8.21-8.18 (d, J= 9.06 Hz, 1H), 7.81-7.76 (m, 1H), 7.58-7.55 (d, 1H), 7.42-7.19 (m, 4H), 6.99-6.96 (d, 1H), 4.0 (s, 2H), 3.70 (s, 2H), 3.03 (bs, 2H), 2.91 (bs, 2H), 1.76 (bs, 4H).
Compound 1129 was prepared from 3-bromo-5-trifluoromethoxyanisole using a procedure analogous to that for compound 1126 below.
Compound 729 was prepared using compound 1129 with a procedure analogous to that for compound 670. 1H NMR (300 MHz, DMSO-d6) δ 12.66 (s, 1H), 11.28 (s, 1H), 8.52-8.50 (m, 1H), 8.21-8.18 (d, J= 9.06 Hz, 1H), 7.81-7.76 (m, 1H), 7.58-7.55 (d, 1H), 7.42-7.29 (m, 2H), 6.99-6.95 (m, 2H), 6.84 (s, 1H), 4.0 (s, 2H), 3.80 (m, 5H),
4H).
Compound 730 was prepared from compound 729 using a procedure analogous to that for the preparation of compound 718 above. 1H NMR (300 MHz, DMSO-d6) δ 12.66 (s, 1H), 11.28 (s, 1H), 10.04 (s, 1H), 8.52-8.50 (m, 1H), 8.21-8.18 (d, J= 9.06 Hz, 1H), 7.81-7.76 (m, 1H), 7.58-7.55 (d, 1H), 7.42-7.29 (m, 2H), 6.81-6.78 (m, 2H), 6.61 (s, 1H), 4 4H).
To a mixture of 6-(di-Boc-amino)-2-bromopyridine (1 g, 2.9 mmol), bis(tri-tert- butylphosphine) palladium(O) (300 mg, 0.59 mmol) in 1 ,4-dioxane (30 ml) under argon atmosphere was added 0.5 M of 2-tert-butoxy-2-oxoethyl zinc chloride in ether (15 ml). The resulting mixture was stirred at room temperature overnight. The mixture was partitioned between saturated NH4C1 and EtOAc. The organic extract was washed with brine, dried over sodium sulfate, filtered and evaporated. The crude
material was purified by silica gel chromatography eluting with 0-20% EtOAc in Hexane to afford 1123.
To a solution of 1123 (150 mg, 0.37 mmol) in MeOH (6 ml) and water (2 ml) at 0 °C was added Lithium hydroxide monohydrate (100 mg, 2.38 mmol). The resulting mixture was stirred at room temperature for 2 days before it was evaporated to dryness. The mixture was then acidified with IN HCl (pH 4), and it was partitioned between water and EtOAc. The organic extract was washed with water, dried over sodium sulfate, filtered and evaporated to afford 1124.
A flask was charged with 657 (105 mg, 0.232 mmol), 1124 (90 mg, 0.255 mmol) in DMF (1 ml) at 0 °C was added propylphosphonic anhydride solution (300 ul) followed by triethylamine (89 ul, 0.64 mmol). The resulting mixture was slowly warmed up to room temperature and stirred for 3 h before it was quenched by addition of ice water (~5 mL). The precipitate was collected by suction filtration, rinsed with more water. The crude material was purified by silica gel chromatography eluting with 0-6% MeOH in DCM to afford 724. 1H NMR (300 MHz, DMSO-d6) δ 12.67 (s, 1H), 11.32 (s, 1H), 9.69 (s, 1H), 8.22-8.19 (d, J= 9.12 Hz, 1H), 7.72-7.01 (m, 8H), 3.91-3.87 (d, 4H), 3.01 (bs, 2H), 2.90 (bs, 2H), 1.75 (bs, 4H) 1.47 (s, 9H).
To a solution of 724 (50 mg, 0.07 mmol) in DCM (3 ml) at 0 °C was added TFA (3 ml) dropwise. The resulting mixture was stirred at room temperature for 3 h before it was evaporated to dryness then triturated the residue with ether to afford 725. 1H NMR (300 MHz, DMSO-d6) δ 12.67 (s, 1H), 11.32 (s, 1H), 8.22-8.19 (d, J= 9.12 Hz, 1H), 7.88-7.77 (m, 3H), 7.59-7.26 (m, 5H), 6.90-6.80 (m, 2H), 4.05 (s, 2H), 3.87 (s, 2H), 3.01 (bs, 2H), 2.90 (bs, 2H), 1.75 (bs, 4H).
Q I if ^ Q
N CI ^ N ^-^ O ^ ^ N v OH
TFA salt
1125 1126
To a stirred solution of tert-butyl acetate (789 ul, 5.88 mmol), 2-chloro-6- methylpyridine (428 ul, 3.92 mmol), chloro(2-di-t-butylphosphino-2',4',6'-tri-l- propyl-l, -bi-phenyl)[2-(2-aminoethyl)phenyl]palladium(II) (27 mg, 0.039 mmol) in toluene (10 ml) at 0 °C under argon was added a solution of LHMDS (1M in toluene) (12 ml, 12 mmol) pre-cooled to 0 °C. The resulting mixture was stirred for 1 h. The mixture was partitioned between saturated NH4C1 and EtOAc. The organic extract was washed with brine, dried over sodium sulfate, filtered and evaporated. The crude material was purified by silica gel chromatography eluting with 0-15% EtOAc in Hexane to afford 1125.
To a solution of 1125 (267 mg, 1.29 mmol) in DCM (3 ml) at 0 °C was added TFA (1.5 ml) dropwise. The resulting mixture was stirred at room temperature overnight before it was evaporated to dryness then triturated the residue with ether to afford
A flask was charged with 657 (50 mg, 0.111 mmol), 1126 (35 mg, 0.133 mmol) in DMF (1 ml) at 0 °C was added propylphosphonic anhydride solution (155 ul) followed by triethylamine (57 ul, 0.4 mmol). The resulting mixture was slowly warmed up to room temperature and stirred for 3 h before it was quenched by addition of ice water (~5 mL). The precipitate was collected by suction filtration, rinsed with more water. The crude material was purified by silica gel chromatography eluting with 0-6% MeOH in DCM to afford 728. 1H NMR (300 MHz, DMSO-d
6) δ 12.67 (s, 1H), 11.32 (s, 1H), 8.22-8.19 (d, J= 9.12 Hz, 1H), 7.69-7.15 (m, 8H), 3.96 (s, 2H), 3.87 (s, 2H), 3
To a solution of ethyl 2-pyridyl acetate (1 g, 6.05 mmol) in DCM (20 ml) at 0 °C was added MCPBA (77% max) (1.77 g, 10.2 mmol). The resulting mixture was warmed up to room temperature for 3 h before it was partitioned between saturated sodium
bicarbonate and DCM. The organic extract was washed with brine, dried over sulfate, filtered and evaporated. The crude material was purified by silica gel chromatography eluting with 0-12% MeOH in EtOAc to afford 1127.
To a suspension of 657 (331 mg, 0.73 mmol) in toluene was added 1127 (278 mg, 1.53 mmol) followed by trimethylaluminum (2M in toluene) (732 ul, 1.46 mmol). The resulting mixture was stirred at 60 °C overnight. The reaction mixture was partitioned between water and DCM. The organic extract was washed with brine, dried over sodium sulfate, filtered and evaporated. The crude material was purified by silica gel chromatography eluting with 0-5% MeOH in DCM then 0-15% MeOH in EtOAc to afford 716. 1H NMR (300 MHz, DMSO-d6) δ 12.67 (s, 1H), 11.32 (s, 1H), 8.29-8.27 (m, 1H), 8.21-8.19 (d, J = 9.12 Hz, 1H), 7.61-7.26 (m, 8H), 4.03 (s, 2H), 3.87 (s, 2H), 3.01 (bs, 2H), 2.90 (bs, 2H), 1.75 (bs, 4H).
Preparative HPLC Purification
All reverse phase preparative HPLC purifications were performed using a Shimadzu Prominence Preparative Liquid Chromatograph with the column at ambient temperature. Mobile phases A and B consisted of 0.1% formic acid in water and 0.1%) formic acid in acetonitrile, respectively. Crude product mixtures were dissolved in DMF, DMSO or mixtures thereof at concentrations of approximately 100 mg/mL and chromatographed according to the methods described in Table 2. Appropriate chromatographic fractions were then evaporated under high vacuum at 45° C using a Savant Speed Vac Plus Model SC210A to yield purified products.
TABLE 2: Preparative HPLC Method Descriptions
2 20 5
3 70 5
14 100 5
1 0 20 2 11.5
1 20 2
2 20 5
3 70 5
14 100 5
1 0 40 1 6
1 40 2
3.5 40 4
4 40 4.73
10 90 4.73
2 0 40 2 7.7
1 40 3
2 40 18.9
13 50 18.9
2 0 32 3 12.1
0.5 32 5
1 32 18.9
13 35 18.9
2 0 50 3 9.1
1 50 3
2 50 18.9
5 50 18.9
15 80 18.9
2 0 35 3 6.2
1 35 3
2 35 18.9
4 35 18.9
14 75 18.9
199 2 0 45 3 8.3
1 45 3
2 45 18.9
3 45 18.9
13 65 18.9
203 2 0 50 3 9.6
1 50 3
2 50 18.9
5 50 18.9
15 60 18.9
208 2 0 35 3 7.6
1 35 3
2 35 18.9
4 35 18.9
14 50 18.9
The following representative synthetic protocols may also be used for producing compounds of the invention.
1028 1029
3,6-Dichloropyridazine is treated with di-tertbutyl malonate and sodium hydride in THF or DMF to give 1026. Intermediate 1026 is then treated with sodium hydride in THF or DMF followed by bis-(chloromethyl)sulfide to give 1027. Intermediate 1027
is treated with TFA in dichloromethane to give 1028. Intermediate 1028 is treated with ammonia to give 1029. Intermediate 1028 is also converted to 1029 by sequential treatment with 2, 4-dimethoxybenzyl amine and TFA. The bis-amino intermediate 1029 may be converted to acylated products analogous to those described in Table 3 using the methods described in Synthetic Protocols section above for acylation of 1001-1008.
MsCI, pyridine
OH DCM MsO' OMs NaCN, DMSO
HO
1031 1032
Both trans- and czs-cyclopropane-l,2-diyldimethanols are converted into the corresponding bis-nitrile 1031 via bis-mesylated intermediate 1030. The bismesylate intermediate 1030 is prepared by treating the diol with methanesulfonyl chloride in the presence of pyridine or triethylamine in dichloromethane. The bisnitrile 1031 is prepared by treating 1030 with sodium cyanide in DMSO or ethanol/water. Using a procedure similar to that described for the preparation 1001, bis-nitrile 1031 undergoes cyclization with thiosemicarbazide in TFA to provide bis-amino intermediate 1032. The bis-amino intermediate 1032 may be converted to acylated products analogous to those described in Table 3 using the methods described in Synthetic Protocols section above for acylation of 1001-1008.
The alkene analog 1033 is prepared from trans-3-hexenedinitrile using a procedure similar to that described for the preparation 1001. The bis-amino intermediate 1033
may be converted to acylated products analogous to those described in Table 3 (for example, 1034) using the methods described in Synthetic Protocols section above for acylation of 1001-1008. The products may be further converted to cyclopropyl analogs (exemplified by 1035) under the Simmons-Smith conditions (Et2Zn, CH2I2,l,2-dimethoxyethane).
Example 2: Compound Assays
Compounds were assayed in both an in vitro biochemical assay and a cell proliferation assay as follows. The IC50 results are provided in Table 3.
Recombinant Enzyme assay
Compounds were assessed for their ability to inhibit the enzymatic activity of a recombinant form of Glutaminase 1 (GAC) using a biochemical assay that couples the production of glutamate (liberated by GAC) to glutamate dehydrogenase (GDH) and measuring the change in absorbance for the reduction of NAD+ to NADH.
Substrate solution was prepared (50 mM Tris-HCl pH 8.0, 0.2 mM EDTA, 150 mM K2HPO4, 0.1 mg/ml BSA, 1 mM DTT, 20mM L-glutamine, 2 mM NAD+, and 10 ppm antifoam) and 50 added to a 96-well half area clear plate (Corning #3695).
Compound (2 μΕ) was added to give a final DMSO concentration of 2% at 2X the desired concentration of compound. Enzymatic reaction was started with the addition of 50 μΕ of enzyme solution (50 mM Tris-HCl pH 8.0, 0.2 mM EDTA, 150 mM K2HPO4, 0.1 mg/ml BSA, 1 mM DTT, 10 ppm antifoam, 4 units/ml GDH, 4 mM adenosine diphosphate, and 4 nM GAC) and read in a Molecular Devices M5 plate reader at 20°C. The plate reader was configured to read absorbance (λ=340 nm) in kinetic mode for 15 minutes. Data was recorded as milli-absorbance units per minute and slopes were compared to a control compound and a DMSO-only control on the same plate. Compounds with slopes less than the DMSO control were considered inhibitors and plate variability was assessed using the control compound.
Results from this assay for several compounds of the invention are shown in Tables 3a and 3b, expressed as IC50, or half maximal inhibitory concentration, wherein IC50 is a quantitative measure indicating how much compound is needed to inhibit a given biological activity by half.
Recombinant Enzyme assay - Time Dependence
Compounds were assessed for their ability to inhibit the enzymatic activity of a recombinant form of Glutaminase 1 (GAC) using a biochemical assay that couples the production of glutamate (liberated by GAC) to glutamate dehydrogenase (GDH) and measuring the change in absorbance for the reduction of NAD+ to NADH.
Enzyme solution was prepared (50 mM Tris-HCl pH 8.0, 0.2 mM EDTA, 150 mM K2HP04, 0.1 mg/ml BSA, 1 mM DTT, 10 ppm antifoam, 4 units/ml GDH, 4 mM adenosine diphosphate, and 4 nM GAC) and 50 added to a 96-well half area clear plate (Corning #3695). Compound (2 μΕ) was added to give a final DMSO concentration of 2% at 2X the desired concentration of compound. The
enzyme/compound mix was sealed with sealing foil (USA Scientific) and allowed to incubate, with mild agitation, for 60 minutes at 20°C. Enzymatic reaction was started with the addition of 50 of substrate solution (50 mM Tris-HCl pH 8.0, 0.2 mM EDTA, 150 mM K2HP04, 0.1 mg/ml BSA, 1 mM DTT, 20mM L-glutamine, 2 mM NAD+, and 10 ppm antifoam) and read in a Molecular Devices M5 plate reader at 20°C. The plate reader was configured to read absorbance (λ=340 nm) in kinetic mode for 15 minutes. Data was recorded as milli-absorbance units per minute and slopes were compared to a control compound and a DMSO-only control on the same plate. Compounds with slopes less than the DMSO control were considered inhibitors and plate variability was assessed using the control compound.
Results from this assay for several compounds of the invention are shown in
Tables 3a and 3b, expressed as IC50, or half maximal inhibitory concentration, wherein IC50 is a quantitative measure indicating how much compound is needed to inhibit a given biological activity by half.
Cell proliferation assay
P493-6 (myc "on") cells were maintained in growth media (RPMI-1640,
10%FBS, 2mM glutamine, 100 units/ml Penicillin and 100μg/ml streptomycin) at 37°C with 5% C02. For compound assay, P493-6 cells were plated in 96-well V- bottom plates on the day of compound addition in 50 μΐ of growth media at a cell density of 200,000 cells/ml (10,000 cells/well). Compounds were serially diluted in 100% DMSO at 200-times the final concentration. Compounds were diluted 100- fold into growth media and then 50 μΐ of this mixture was added to cell plates making the final concentration of DMSO 0.5%. Cells were incubated with compound for 72 hrs at 37°C with 5% C02 and analyzed for antiproliferative effects either by Cell Titer
Glo (Promega) or FACS analysis using the Viacount (Millipore) kit on the Guava instrument.
Results from this assay for several compounds of the invention are shown in Tables 3a and 3b, expressed as IC50, or half maximal inhibitory concentration, wherein IC50 is a quantitative measure indicating how much compound is needed to inhibit a given biological activity by half.
Modified Recombinant Enzyme assay - Time Dependence
Compounds were assessed for their ability to inhibit the enzymatic activity of a recombinant form of glutaminase using a biochemical assay that couples the production of Glu (liberated by glutaminase) to GDH and measures the increase in fluorescence due to the reduction of NADP+ to NADPH.
Assay Set-up: Glutaminase reaction buffer was prepared [50 mM Tris-HCl pH 8.8, 150 mM K2HP04, 0.25 mM EDTA, 0.1 mg/ml BSA (Calbiochem no. 2960), 1 mM DTT, 2 mM NADP+ (Sigma Aldrich no. N5755), and 0.01% TX-100] and used to make 3x-enzyme-containing solution, 3x-substrate-containing solution, and 3x- inhibitor-containing solution (see below). Inhibitor-containing solution was made by diluting DMSO stocks of compounds into the glutaminase reaction buffer to create a 3x inhibitor solution containing 6% DMSO. 3x-enzyme-containing solution was made by diluting recombinant glutaminase and GDH from Proteus species (Sigma Aldrich no. G4387) into glutaminase buffer to create a 6 iiM glutaminase plus 18 units/mL GDH solution. A 3x substrate solution containing either Gin, Glu, or NADPH was made by diluting a stock of Gin (Sigma Aldrich no. 49419), Glu (Sigma Aldrich no. 49449), or NADPH (Sigma Aldrich no. N1630) into glutaminase reaction buffer to create a 3x-substrate solution. Reactions were assembled in a 384-well low- volume black microtiter plates (Molecular Devices no. 0200-5202) by mixing 5 μΐ^ of inhibitor-containing solution with 5 μΐ^ of substrate-containing solution followed by 5 μΐ. of enzyme-containing solution when no preincubation was required. When time- dependent effects of compound inhibition were tested, enzyme-containing solution was treated with inhibitor-containing solution for the indicated time prior to addition of substrate-containing solution.
Measurement of glutaminase activity: Following the mixture of all three components, fluorescence increase (Ex: 340 nM, Em:460 nm) was recorded for 15 min at room temperature using the Spectromax M5e (Molecular Devices).
IC50 Determination: The initial velocities of each progress curve were calculated using a straight line equation (Y=Yintercept + (slope) * X). Initial velocity values were plotted against compound concentration and fit to a four parameter dose response equation (% activity =Bottom + (Top-Bottom)/(l+10A((LogIC50- X)*HillSlope))) to calculate an IC50 value.
Results from this assay for several compounds are shown in Tables 3a and 3b, expressed as IC50, or half maximal inhibitory concentration, wherein IC50 is a quantitative measure indicating how much compound is needed to inhibit a given biological activity by half.
Table 3a:
N'N N-fvj ^J?
ı94
Example 3 : Caco-2 Permeability Assay
Caco-2 cells are commonly used in a confluent monolayer on a cell culture insert filter. When cultured in this format and under specific conditions, the cells become differentiated and polarized such that their phenotype, morphologically and functionally resembles the enterocytes lining the small intestine. The cell monolayer provides a physical and biochemical barrier to the passage of small molecules, and is widely used across the pharmaceutical industry as an in vitro model of the human small intestinal mucosa to predict the absorption of orally administered drugs
(Hidalgo et al, Gastroenterology, 1989; Artursson, J. Pharm. Sci., 1990). The correlation between the in vitro apparent permeability (P-'app) across Caco-2 monolayers and the in vivo absorption is well established (Artursson et al, Biochem. Biophys. Res. Comm., 1991).
The present assay was used to determine the bidirectional permeability of the compounds of the invention through Caco-2 cell monolayers. Caco-2 cells were grown in confluent monolayers where the media of both the apical (A) and basolateral
(B) sides were at pH 7.4. Compounds were dosed at ΙμΜ in the presence of 200μΜ Lucifer Yellow, on the apical side (A→B) or the basolateral side (B→A) for assessment, in duplicate. Samples from both A and B sides were taken after 120 minutes exposure, and compound concentration (reported as percent recovery) was determined using a generic LC-MS/MS method with a minimum four-point calibration curve.
The absorption potential of compounds were classified as either Low (P-app < 1X10"6 cm/s) or High (P-app > 1X10"6 cm/s). The efflux ratio was calculated as (Papp B→A)/(Papp A→B), with efflux ratios being significant when greater than or equal to 3 when the Papp (B→A) was greater than or equal to 1X10"6 cm/s. Results for certain compounds of the invention are shown in Table 4.
Table 4: Caco-2 Permeability Results
Cmpd Direction Recovery Papp Efflux Permeability Significant
(%) (avg.) Ratio Classification Efflux
533 A→B 41 4.94 7.6 High Yes
B→A 52 37.5
585 A→B 42 7.52 3.1 High Yes
B→A 53 23.4
616 A→B 65 8.23 6.0 High Yes
B→A 76 49.5
295 A→B 89 8.17 7.3 High Yes
B→A 96 59.8
318 A→B 73 2.45 18 High Yes
B→A 82 44.5
339 A→B 73 2.39 17 High Yes
B→A 80 41.6
354 A→B 117 1.38 33 High Yes
B→A 101 45.0
436 A→B 44 3.75 6.6 High Yes
B→A 57 24.7
660 A→B 56 0.61 3.9 Low Yes
B→A 68 2.37
670 A→B 70 9.64 6.2 High Yes
B→A 72 59.6
679 A→B 34 7.59 2.6 High No
B→A 42 19.6
447 A→B 71 7.76 3.5 High Yes
B→A 56 27.2
703 A→B 51 6.26 6.6 High Yes
B→A 66 41.0
705 A→B 60 8.52 6.0 High Yes
B→A 67 51.0
Example 4: Solubility
Ca. 1 mg portions of test article were combined with 120 μΐ, solvent in wells of a 96 x 2 mL polypropylene plate. The plate was vigorously vortex mixed at room temperature (ca. 20 C) for 18 hr and each well checked visually for undissolved solid; wells containing no visible solid were charged with additional solid test article and vortex mixed another 6 hr at room temperature after which all wells showed visible solid. The contents of all wells were then filtered through a 0.45 μιη GHP filter plate to yield clear filtrates. 5 of each filtrate was diluted into 100 DMF and vortex mixed to yield HPLC samples. Duplicate quantitation standards for each test article were prepared by diluting weighed portions of solid test article in measured volumes of DMF. 2 μΐ^ of each HPLC sample and quantitation standard were analyzed by HPLC using the method outlined in Table 5. Dissolved test article concentrations were calculated by peak area ratio against the appropriate quantitation standards. Solubility results are presented in Table 6. Table 5: Outline of HPLC Method
8.6 20
9.6 END
Table 6: Measured Solubilities
0.9% NaCl < 0.002 0.005 < 0.004
0.1 M HC1 0.005 < 0.004 < 0.004
50 mM Cit
pH 2.3 0.066 < 0.004 < 0.004
50 mM Cit
pH 3.3 0.003 < 0.004 < 0.004
50 mM Cit
pH 4.4 < 0.002 < 0.004 < 0.004
50 mM Cit
pH 5.4 < 0.002 < 0.004 < 0.004
PBS < 0.002 < 0.004 < 0.004
0.1 M
NaOH 0.227 0.192 0.656
10% PS80 /
50 mM cit 1.204 0.851 0.378
10% CrEL
/ 50 mM cit 0.458 0.732 0.309
20%
SBECD /
50 mM cit 5.256 2.718 0.476
20%
HPBCD /
50 mM cit 9.685 2.177 0.651
Labrasol 5.042 77.164 20.727
Capryol
PGMC 1.519 7.916 3.683
Capryol 90 1.974 11.114 7.409
canola oil 0.012 0.071 0.014
PEG400 9.901 57.334 22.419
PG 2.569 8.265 4.698
EtOH 0.964 3.921 2.645
Example 5 : Anti-proliferation and glutamine dependency assay.
Breast cell lines were tested in vitro for their ability to grow in the absence of glutamine and for their sensitivity to compound 670 in glutamine containing media. The cells were maintained in growth media (RPMI-1640, 10%FBS, 100 units/ml penicillin and lOOAg/ml streptomycin, 0.25 μg/mL amphotericin) supplemented with 2mM glutamine at 37°C with 5% C02.
To determine glutamine dependence, cells were seeded in 96-well plates at a density of 3000-5000 cells/well depending on cell size and their growth characteristics. The appropriate plating density was selected to ensure that the cells did not become confluent during the 72 hour assay period. Twenty- four hours after seeding, the
plating media was removed and the cells were washed 2 times with glutamine-free growth media and then 100 uL of glutamine-free media or glutamine containing (2mM) growth media was added back to the wells. Cells were incubated for 72 hrs at 37°C with 5% C02 and analyzed for antiproliferative effects by Cell Titer Glo (Promega). Cell proliferation (% of DMSO control) was determined by comparing the Cell Titer Glo signal (rfu) on the day of glutamine withdrawal (t=0) measured in parallel plates with the signal observed after the 72 hour incubation period by the following formula ((rfu of cells grown in glutamine-free media for 72 hours - rfu at t=0)/(rfu of cells grown in 2mM glutamine for 72hrs - rfu at t=0)). Cell lost was determined by the following formula: (100 x rfu at 72 hours in glutamine-free media/rfu at t=0) - 100.
Sensitivity to compound 670 was determined by treating cells in 96-well plate seeded as described above. Twenty four hours after seeding, the cells were washed with growth media with 2mM glutamine and 50 uL of growth media with 2 mM glutamine was added to the well. A 10 mM DMSO stock of compound 670 was diluted into
100% DMSO at 200 uM. This was further diluted to 2 uM in growth media with 2mM glutamine. 50 ul of this mixture was added to cell plates making the final
concentration of 670 uM to be 1 uM. Parallel control wells were treated with DMSO only. Cells were incubated for 72 hours at 37 °C with 5% C02 and analyzed for antiproliferative effects by Cell Titer Glo. Cell proliferation was calculated in a manner similar to that described above with the following modifications: cell proliferation ((rfu of cells grown in 1 uM compound 670 for 72 hours - rfu at t=0)/(rfu of DMSO control at 72hrs - rfu at t=0)), cell lost (100 x rfu at 72 hours in 1 uM compound 670/rfu at t=0) - 100. The results from these assays are shown in Figure 1.
Example 6: Differential expression of glutaminase and glutamine synthetase in triple- negative breast cancer subtype.
Primary breast tumor and cell line expression datasets were downloaded [The Cancer Genome Atlas from https://genome-cancer.ucsc.edu (breast invasive carcinoma/gene expression/R AseqV2 data) and The Cell Line Encyclopedia from
http://www.broadinstitute.org/ccle/home (gene-centric RMA-normalized mRNA expression/aAffymetrix U133+2 arrays)] and the expression level within each dataset
for the following genes was evaluated: estrogen receptor (ER), progesterone receptor (PR) and Her2 (ERBB2), glutaminase (GLS) and glutamine synthetase (GLUL). The relative expression level for a given gene in each sample was calculated by comparison to the median expression of the gene in the entire dataset. Samples with the lowest relative levels of ER, PR, and Her2 ("triple-negative") were identified by analysis of individual expression distributions for the three marker genes and the relative levels of glutaminase and glutamine synthetase within this population and the non-triple-negative population was assessed. Figure 2 represents a heatmap illustrating the relatively high expression (red) of glutaminase and low expression (green) of glutamine synthetase in the triple-negative population.
Example 7: Single-agent compound 402 treatment of MDA-MB-231 orthotopic xenograft model.
Female scig/beige mice (n=20) age 6-8 weeks were implanted in the inguinal mammary fat pad with 1 x 107 MDA-MB-231 cells mixed 1 : 1 with matrigel. When tumors reached a volume of 100-150 mm3, mice were randomized into the following two groups of n=10 mice/group: 1) Vehicle control (Gelucire) dosed PO BID for 35 days, and 2) compound 402 at 100 mg/kg (formulated at 10 mg/mL in Gelucire) dosed IP BID for 35 days. Tumors were measured with calipers twice weekly for 35 days and tumor volume calculated using the formula tumor volume (mm3) = (a x b2/2) where 'b' is the smallest diameter and 'a' is the largest diameter. 24 hours after the final dose, mice were sacrificed, lungs were excised, and lung metastases quantified by percent lung metastases coverage (textured lung exterior). Figure 3 shows measurement of tumor volume and metastases upon treatment with compound 402 compared to vehicle. Example 8: Combination study with compound 389 and paclitaxel in MDA-MB-231 orthotopic xenograft model.
Female scig/beige mice (n=40) age 6-8 weeks were implanted in the inguinal mammary fat pad with 1 x 107 MDA-MB-231 cells mixed 1 : 1 with matrigel. When tumors reached a volume of 100-150 mm3, mice were randomized into the following four groups of n=10 mice/group: 1) Vehicle control (20% HPBCD/lOmM Citrate buffer pH 4.0) dosed IP BID for 35 days, 2) compound 389 at 50 mg/kg (formulated
at 5 mg/mL in 20% HPBCD/lOmM Citrate buffer pH 4.0) dosed IP BID for 35 days, 3) Paclitaxel at 10 mg/kg (clinical formulation diluted to 1 mg/mL in saline) dosed IP QD x 5 days, and 4) compound 389 at 50 mg/kg IP BID x 35 days plus paclitaxel at 10 mg/kg dosed IP QD x 5 days. Tumors were measured with calipers twice weekly for 35 days and tumor volume calculated using the formula tumor volume (mm3) = (a x b2/2) where 'b' is the smallest diameter and 'a' is the largest diameter. Figure 4 shows measurement of tumor volume upon treatment with a combination of compound 389 and paclitaxel compared to vehicle and each agent alone.
Example 9: Determination of glutamate and glutamine in cell samples by liquid chromatography tandem mass spectrometry.
Sensitivity to compound 670 was determined as described in Example 5.
Untreated cells were examined for metabolite levels. Concentrations of glutamine and glutamate were determined by liquid chromatography tandem mass spectrometry (LC-MS/MS). Cell pellets from in vitro cell assays were washed by PBS, mixed with methanol: water (50:50) containing internal standard (IS) for extraction of glutamine and glutamate and then stored at-70°C until analysis. The extracted cellular samples were vortexed, centrifuged and/or filtered and 10 of the extracts was injected for LC-MS/MS analysis. Glutamine and glutamate were quantified by comparing the peak area ratios of the analyte to IS in study samples to the standard calibration samples. The LC-MS/MS system comprised an API 4000 mass spectrometer (ABSCIEX, Foster City, CA) equipped with Shimadzu LC- lOADvp pumps
(Shimadzu, Columbia, MD) and Leap PAL HTC-xt autosampler. Chromatographic separation was achieved on an Phenomenex Luna NH2 column (2.1 x 50 mm, 3.5 μιη particle size) using gradient elution. The mobile phases were (A) 10 mM ammonium acetate and 5 mM ammonium hydroxide in water and (B) 50:50 methanokacetontrile. Mass spectrometric detection was accomplished using the Turbo ionspray interface in the negative ionization mode by MRM of the selective m/z transitions: 145.9— »101.8 for glutamate and 144.7— »108.8 for glutamine. The results from these assays are shown in Figure 9. Example 10: Determination of glutaminase: lutamine synthetase ratios
Gene expression data were from the Barretina Cell Line dataset in Oncomine.
Expression levels for each glutaminase and glutamine synthetase transcript for each primary tumor sample were quantile normalized. In any given sample, a log2 copy number of 0 indicates that the gene in question is expressed at the median expression level relative to 12,000 genes across all datasets and samples analyzed. The horizontal line indicates the ratio of the median expression of each transcript within the number of clinical samples shown. The results are represented in Figures 5, 6, 7 and 8.
Example 11 : Expression and metabolite correlations extend to other tumor types Primary tumor xenografts were provided by a commercial clinical research organization, along with microarray data for glutaminase and glutamine synthetase expression. Glutamate and glutamine cncentrations were determined as described in Example 9. Glutaminase activity was determined essentially as described in Curthoys and Bellemann (Exp Cell Res, 1979). Figure 10 shows the correlation between glutamate: glutamine ratios and glutaminase: glutamine synthetase expression ratios or glutaminase activity.
Example 12: Colon carcinoma xenograft efficacy study
Female scid/bg mice, approximately 6 weeks of age, were implanted subcutaneously on the right flank with 5 x 106 HCT116 cells per mouse in a volume of 100 uL of sterile PBS. When tumors reached a volume of 50 - 100mm3, mice were randomized to groups of n=10 to receive either vehicle or test compound delivered twice daily by intraperitoneal injection. Tumors were measured three times per week using Vernier calipers and tumor volume calculated using the formula: Volume = (Length x Width2/2), where length and width are the longest perpendicular sides of the tumor. Dosing continued twice daily until control tumors reached a size of 2000mm3.
Statistical comparisons were made using a 2-way ANOVA with Bonferroni post-test. Figure 11 shows that intraperitoneal administration of compound 188 to mice results in reduced tumor size in this HCT116 colon carcinoma xenograft model.
Example 13: Lung adenocarcinoma xenograft efficacy study
Female scid/beige mice (n=20) age 6-8 weeks were implanted subcutaneously with 1 x 107 H2122 lung adenocarcinoma cells per mouse suspended in PBS. Mice were randomized into the following two groups of n=10 mice/group: 1) Vehicle control (25% Hydroxypropyl-P-cyclodextrin) and 2) Compound 670 dosed orally at 200 mg/kg (formulated at 20 mg/mL in 25% ΗΡ-β-CD). For both groups, dosing was initiated 24 hours post-implant and continued orally BID for 23 days. Tumors were measured with calipers three times per week and tumor volume calculated using the formula tumor volume (mm3) = (a x b2/2) where 'b' is the smallest diameter and 'a' is the largest perpendicular diameter. **P-value < 0.01 (Two-sided T-test). Results are shown in Figure 12.
Example 14: mRNA expression of glutaminase and glutamine synthetase in TNBC and HR+/Her2+ breast tumor cell lines
Two publicly available databases were queried to determine the mRNA levels of glutaminase (GLS) and glutamine synthetase (GS):
- Microarray expression data for a panel of 51 breast cancer cell lines
published by Neve et al, (Cancer Cell 10(6):515-27 (Dec 2006)) of which 20 were evaluated in the present example, and
The Cancer Cell Line Encyclopedia (CCLE; Barretina et al., Nature 483, 603-607 (29 March 2012)) which included expression data for 58 breast cancer cell lines, 25 of which were used in this example.
The Neve et al. publication included annotation of hormone and growth factor status for each cell line in the data set (25 triple negative, 26 HR+ or Her2+). For the CCLE dataset, hormone and growth factor receptor status was evaluated based on mRNA expression levels for estrogen receptor (ESR, 20/58 positive), progesterone receptor (PGR, 10/58 positive) and Her2 (ERBB2, 13/58 positive). Based on this analysis, a total of 31 cell lines were classified as TNBC and 27 as HR+ or Her2+. For the 33 cell lines represented in both datasets, there was good concordance in the hormone and growth factor receptor status assignment (32/33). The present example was carried out in a panel of cell lines that included 22 triple negative and 7 HR+ or Her2+.
The log2 transfomred mRNA expression values for the GLS splice variants KGA (probeset 203159_at) and GAC (probeset 221510_s_at) as well as GS (probeset
215001_s_at) in each cell line were median-centered based on the median expression value for all probesets across all samples in the dataset (median of 5.583 for the Neve et al. dataset and median of 4.809 for the CCLE dataset). For calculation of the GLS:GS ratio, the log2 transformed expression values for KGA, GAC, and GS were first converted back to their corresponding untransformed values. The expression levels of GLS (KGA and GAC), GS and the ratio of GLS (KGA or GAC) to GS were compared in the TNBC cell lines vs. the HR+/Her2+ cell lines. Significant differences were determined using an un-paired Student's T-test (Prism). Differences between the TNBC cell lines and HR+/Her2+ cell lines are depicted graphically in Figure 13. For both datasets, there was significantly higher expression of the GLS splice variants KGA and GAC in the TNBC as compared to the HR+ or Her2+ cell lines. The magnitude of difference and statistical significance was greater for the GAC splice variant. For glutamine synthetase (GS), there was significantly lower expression in the TNBC cell lines relative to the HR+ or Her2+ subset for both datasets. The ratio of either KGA to GS and of GAC to GS was also significantly higher in the TNBC cell lines.
Example 15: Correlation between sensitivity to Compound 670 and expression of GLS and GS.
The cell proliferation and cell loss observed as a result of Compound 670 treatment was compared to the expression levels of glutaminase (KGA and GAC), glutamine synthetase (GS), and the ratio of glutaminase to glutamine synthetase. The
antiproliferation effect of Compound 670 was determined as described in Example 5. Figure 14 displays a series of bivariate graphs plotting the Compound 670 sensitivty against each expression parameter for all tested cell lines (from either the Neve et al. or the CCLE datasets) while Table 7 summarizes the corresponding Spearman rank order correlation coefficients (and P values). For both expression datasets, significant correlations were observed between Compound 670 sensitivity and the expression of the GAC isoform of glutaminase, the expression of glutamine synthetase (GS), and the ratio of GAC:GS. The most significant correlation in each dataset was with GAC expression alone. These results support the hypothesis that cells with an elevated
GAC expression or a GAC:GS ratio are sensitive to GLS inhibition with glutaminase inhibitors. This phenotype is observed in the majority of TNBC cell lines and the minority of receptor positive breast cell lines. Table 7. Correlation between sensitivity to Compound 670 and GLS mR A expression, GS mRNA expression, or expression ratios1.
Spearman rank order correlation coefficients and associated P-values for the data plotted in the bivariate graphs from Figure 14. The full panel of breast cancer cell lines (TNBC, HR+, Her2+) were combined for each correlation analysis.
Example 15: Protein expression of Gin-utilizing enzymes in breast cancer cell lines
Western analysis of extracts prepared from the panel of breast cancer cell lines was used to monitor, at the protein level, expression of GLS (GAC and KGA splice variants) and glutamine synthetase. As shown in Figure 15, consistent with the microarray mRNA expression analysis for these genes, the majority of TNBC cell lines express GAC and KGA, while GAC and KGA are expressed at lower levels (or undetectable levels) in most of the receptor-positive lines. GAC, in particular, is expressed at relatively high levels in nearly all of the TNBC cell lines tested
(compared to the HR+/Her2+ cell lines). The expression of glutamine synthetase was more variable and, in contrast to the microarray data, did not display a clear distinction between TNBC and receptor-positive cells across this panel of cell lines.
Cell lysates prepared for the Western blotting were analyzed for glutaminase activity according to the method described in Example 11. Results show that the level of KGA and GAC protein corresponded with higher glutaminase activity.
Example 16: Sensitivity to glutaminase inhibitor and metabolite levels.
Glutamate and glutamine concentrations were determined as described in Example 9. Sensitivity to glutaminase inhibitor was determined as described in Example 5.
Figure 16 shows the correlation between glutamate: glutamine ratios and sensitivity to compound 670.
Example 17: Multiple Myeloma xenograft study. Female CB.17 SCID mice (n=20) age 8-12 weeks were implanted subcutaneously with 1 x 107 RPMI-8226 myeloma cells per mouse mixed 1 : 1 with Matrigel. Mice were randomized into the following two groups of n=10 mice/group: 1) Vehicle control (25% Hydroxypropyl-P-cyclodextrin) and 2) Compound 670 dosed at orally at 200 mg/kg (formulated at 20 mg/mL in 25% ΗΡ-β-CD). For both groups, dosing was initiated when tumors reached a volume of 100- 150mm3 and continued orally BID for 28 days. Tumors were measured with calipers two times per week and tumor volume calculated using the formula tumor volume (mm3) = (a x b2/2) where 'b' is the smallest diameter and 'a' is the largest perpendicular diameter. **P-value < 0.01 (Two-sided T-test). Results are shown in Figure 17.
Example 18: Treatment of Multiple Myeloma cells with a combination of drugs.
As shown in Figure 18, MM1S cells (panels A & B) and RPMI-8226 cells (panels C & D) were treated with a dose titration of either compound 670, pomalidomide or a mixture thereof (panels A & C) or compound 670, dexamethsone or a mixture thereof (panels B & D) for 72 hours in growth media. At the end of the incubation, cell viability was measured using Cell Titer Glo as per manufacturer's protocol (Promega, Madison, WI). Measured values for compound-treated cells were normalized to
DMSO-treated cells and data is reported as a cell survival ratio with a value of 1 (one) corresponding to maximum cell survival and a value of 0 (zero) corresponding to no cell survival. Cell survival ratios for all compound treatments are represented as bar graphs. Combination indices were calculated using the Calcusyn program
(biosoft.com) and reported for individual mixtures of compound 670 and
pomalidomide [POM] (panels A &C) and individual mixtures of compound 670 and dexamethasone [DEX] (panels B & D). Compound mixtures that produced a synergistic anti-tumor activity are highlighted.
Incorporation by Reference All publications and patents mentioned herein are hereby incorporated by reference in their entirety as if each individual publication or patent was specifically and individually indicated to be incorporated by reference. In case of conflict, the present application, including any definitions herein, will control. The compounds, synthetic methods, and experimental protocols and results of U.S. Application Number 13/680,582, filed November 19, 2012, are hereby incorporated by reference.
Equivalents
While specific embodiments of the subject invention have been discussed, the above specification is illustrative and not restrictive. Many variations of the invention will become apparent to those skilled in the art upon review of this specification and the claims below. The full scope of the invention should be determined by reference to the claims, along with their full scope of equivalents, and the specification, along with such variations.